Abstract
Background: The purpose of this study is to determine the incidence and significance of hypouricemia in patients with severe acute respiratory syndrome (SARS). Pulmonary lesions in patients with SARS are thought to result from proinflammatory cytokine dysregulation. Acute renal failure has been reported in patients with SARS, but whether cytokines can injure renal tubules is unknown. Methods: Sixty patients diagnosed with SARS in Taiwan in April 2003 were studied. Patients were identified as hypouricemic when their serum uric acid (UA) level was less than 2.5 mg/dL (<149 μmol/L) within 15 days after fever onset. Urine UA and creatinine levels were available for 43 patients; the serum cytokines interleukin-6 (IL-6), IL-8, and tumor necrosis factor-α (TNF-α) were measured in 16 patients. Results: Sixteen patients (26.7%) had hypouricemia (UA, 1.68 ± 0.52 mg/dL [100 ± 31 μmol/L]). No differences in age, sex, symptoms, vital signs, hemogram, or other biochemistry data existed between the hypouricemic and normouricemic groups. Fractional excretion (FE) of UA (FEUA) in 12 hypouricemic patients was 39.6% ± 23.4%, significantly greater than that of 31 normouricemic patients (16.4% ± 11.4%; P < 0.0001). After adjustments for age and sex, high FEUA was significantly associated with the lowest blood oxygenation (P = 0.001; r = −0.624). The number of catastrophic outcomes (endotracheal intubation and/or death) adjusted for older age and sex showed that hypouremic patients had an odds ratio of 10.57 (confidence interval, 2.33 to 47.98; P = 0.002). Kaplan-Meier curves for catastrophic outcome–free results showed significant differences between patients with normouricemia or hypouricemia (P = 0.01). Serum IL-8 levels correlated significantly with FEUA (P < 0.001; r = 0.785) and inversely with serum UA level (P = 0.044; r = −0.509); neither IL-6 nor TNF-α level showed such correlations. Conclusion: One fourth of patients with SARS developed hypouricemia, which might result from a defect in renal UA handling and was associated with a high serum IL-8 level. Renal hypouricemia is an ominous sign in patients with SARS.
Index words: Hypouricemia, severe acute respiratory syndrome (SARS), uric acid (UA), fraction excretion (FE), renal tubule
PATIENTS WITH severe acute respiratory syndrome (SARS) caused by a new coronavirus (CoV) present with fever and respiratory symptoms.1 Although the virus can be found in urine from patients,2 renal function usually is preserved and acute renal failure (ARF) is uncommon.3 Causes of ARF in the patients with SARS we studied were multifactorial, eg, infections, rhabdomyolysis, antiviral agents, and hypoxemia. However, we observed that the elevation in serum creatinine levels in patients with ARF was preceded by increased serum cytokine levels, indicating that proinflammatory cytokine-related renal lesions may have an important role.4
Direct and indirect associations of viral infections with human renal diseases have been noted for years,5 and several pathogenic mechanisms have been postulated as contributing to the renal diseases associated with viral infections.6 Most virus-related renal diseases mainly involve glomeruli. However, lesions of the renal tubules and interstitium have been reported infrequently.7 We showed interstitial infiltrations of inflammatory cells in renal histological specimens obtained at necropsy for 2 patients with SARS complicated by ARF.4 Taking these observations together, we speculate that significant elevations in proinflammatory cytokine levels may have a pivotal role in renal injury in patients with SARS.
Proximal tubular injury is observed commonly in patients with interstitial inflammation and clinically manifested by defects in organic acid and electrolyte transport. Hypouricemia caused by uricosuria has been shown in patients with acute and chronic tubulointerstitial nephritis, but rarely has been reported in patients with systemic infections without frank renal disease.7, 8 Although the mechanism is not elucidated, renal hypouricemia in patients with systemic infections has been shown to be a factor in poor prognosis.7 In the present study, we examine the prevalence of hypouricemia in patients with SARS and its significance to the outcomes of these patients.
Methods
Patients
Seventy-four patients with probable SARS were admitted to this hospital from April 19 to May 16, 2003, when endemic SARS occurred on this island.9 The diagnosis of probable SARS was made in the presence of fever greater than 38°C, respiratory symptoms, contact history, and either pneumonic patch on chest film or positive test for CoV, a definition for probable SARS by the Centers for Disease Control and Prevention and World Health Organization.2, 10, 11 Seven patients were excluded from this study because diuretics were administered during blood sampling; another 7 patients were excluded because they had only 1 test of serum uric acid (UA) within 15 days after the onset of fever.
SARS-CoV had been detected in 18 patients (30%) by means of reverse-transcriptase polymerase chain reaction of throat swabs. Another 36 patients who had negative throat swab results for SARS-CoV had seroconversion detected by means of immunofluorescent assay in 26 patients (43.3%) and by means of enzyme-linked immunosorbent assay in 10 patients (16.7%). The other 6 patients (10%), who had no direct microbiological or serological evidence of SARS-CoV infection by the end of the study, had clinical courses compatible with those of probable SARS. They had a clear relationship, as well as exposure, to initial immigrant clusters and later intrahospital outbreaks in Taiwan.12 The diagnosis of SARS in these patients had been confirmed by a committee of the Center for Disease Control (Taiwan).
Definition of hypouricemia and inappropriate uricosuria
Serum UA was measured at least twice in all patients included in this study. However, because blood sampling was not performed on a fixed schedule, data were clustered in 3 periods after the onset of fever. If more than 2 measurements were obtained in any period, the lowest level in that period was used for analysis. When multiple daily measurements were performed, data obtained closest to 8:00 am was analyzed. Hypouricemia is defined when a serum UA level within 15 days after onset of fever was less than 2.5 mg/dL (<149 μmol/L), which is more than 2 SDs less than the mean UA level measured in this hospital. A standard formula was used to calculate fractional excretion (FE) of UA (FEUA);
In hypouricemic patients, FEUA greater than 10% is defined as inappropriate uricosuria.13, 14
Laboratory measurements
Biochemical parameters were measured using the Toshiba TBA-200FR Clinical Chemistry analyzer (Toshiba, Tochigi-ken, Japan); UA was measured using the uricase method.15 Serum levels of tumor necrosis factor-α (TNF-α), interleukin 6 (IL-6), and IL-8 were determined by using an immunometric assay (Immulite; Diagnostic Products Corp, Los Angeles, CA).
Blood oxygen saturation of hospitalized patients was automatically recorded 3 times every day by using pulse oximetry (Agilent, Andover, MA). The lowest value on the day renal excretion of UA was studied was used for analysis.
Statistical analysis
Results are expressed as mean ± SD unless otherwise specified. Unpaired Student’s t-test was applied to analyze continuous data, and either chi-square test or Fisher’s exact test was applied to analyze categorical data. Statistical analyses were performed using SPSS for Windows, version 10.0 (SPSS Inc, Chicago, IL). Comparison of data between groups was made using a t-test for random data. In addition, repeated measurements of UA variables with hypouremic and normouremic patients were analyzed by using analysis of variance. If results differed significantly, a post hoc comparison was performed using a t-test for paired data and Scheffé test.
A catastrophic outcome is defined as death or need for mechanical ventilation.16 Time to discharge and time to catastrophic outcome were examined by using survival analysis, with follow-up for all patients starting at fever onset and ending after discharge. Data were censored if, at the end of follow-up, patients were still in the hospital (for time to discharge), were still alive (from time to death), and did not have a catastrophic outcome. The Kaplan-Meier (product-limit) method was used to estimate survival and plot time to event between the hypouricemia and normouricemia groups, and log-rank test, to compare differences between groups.13
P less than 0.05 is considered statistically significant.
Results
Patient characteristics
Patient age was 47 ± 17 years, and there were 30 women and 30 men. Thirty-five, 54, and 55 patients had serum UA levels measured at 1 to 5, 6 to 9, and 10 to 15 days after fever onset, respectively. Sixteen patients (26.7%) were hypouricemic, with a serum UA level of 1.68 ± 0.59 mg/dL (100 ± 35 μmol/L); the remainder had a serum UA level of 4.56 ± 1.41 mg/dL (271 ± 84 μmol/L). As shown in Fig 1, at each period, hypouricemic patients had lower serum UA levels than normouricemic patients. In hypouricemic patients, the lowest serum UA level was observed 6 to 9 days after the onset of fever (P = 0.001 compared with days 1 to 5) and returned to baseline 10 to 15 days after fever onset (P = 0.565 compared with days 1 to 5). Conversely, serum UA levels in normouricemic patients were unchanged during this time (P = 0.770). Eleven hypouricemic patients had serum UA levels return to normal (UA > 2.5 mg/dL) after fever onset (day 12.3 ± 1.9).2 Four patients remained hypouricemic after day 16, and 1 patient was without UA data after day 12. Patients with persistent hypouricemia showed a graver prognosis than patients with corrected hypouricemia (intubated or dead, P = 0.016).
Fig 1.
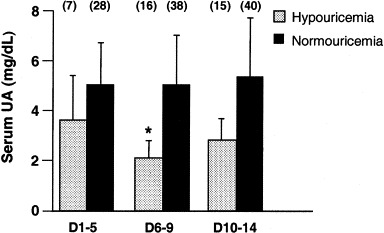
Serum UA levels in patients with SARS measured in different periods after fever: 1 to 5, 6 to 9, and 10 to 14 days. Sixteen patients were identified as hypouricemic (dotted bar), and 44 patients, normouricemic (black bar). Numbers in parentheses indicate the number of patients with serum UA measurements in that period. *P = 0.001.
There were no differences in age, sex, symptoms (ie, sore throat, runny nose, cough, myalgia, and diarrhea), or vital signs on admission between the 2 groups (Table 1). Prevalences of hypertension, diabetes, and ischemic heart disease were the same between the 2 groups (Table 1).
Table 1.
Characteristics of Patients
| Hypouricemic Group (n = 16) | Normouricemic Group (n = 44) | P | |
|---|---|---|---|
| Sex (M/F) | 7/9 | 23/21 | 0.386 |
| Age (y) | 50.2 ± 16.7 | 46.3 ± 17.5 | 0.448 |
| Vital signs at admission | |||
| Systolic blood pressure (mm Hg) | 136.7 ± 23.4 | 133.9 ± 25.9 | 0.732 |
| Diastolic blood pressure (mm Hg) | 84.2 ± 16.7 | 77.02 ± 12.2 | 0.098 |
| Heart rate (beats/min) | 94.1 ± 15.1 | 97.7 ± 15.5 | 0.477 |
| Temperature (°C) | 38.4 ± 1.0 | 37.9 ± 0.97 | 0.152 |
| Symptoms | |||
| Sore throat | 1 (7.69) | 7 (15.91) | 0.287 |
| Cough | 7 (43.75) | 18 (40.91) | 0.459 |
| Myalgia | 8 (50.00) | 23 (52.27) | 0.487 |
| Diarrhea | 8 (50.00) | 14 (31.81) | 0.193 |
| Comorbidities | |||
| Hypertension | 1 (6.25) | 5 (11.36) | 0.655 |
| Ischemic heart disease | 0 (0) | 2 (4.55) | 0.547 |
| Diabetes | 2 (12.50) | 7 (15.91) | 0.551 |
NOTE. Results expressed as number (%) or mean ± SD.
Hemograms and biochemical parameter measurements at admission are listed in Table 2. There were no differences between the 2 groups in white blood cell count, hematocrit value, platelet count, or levels of urea nitrogen, serum creatinine, creatine kinase, and electrolytes.
Table 2.
Hemogram and Biochemical Parameters in Normouricemic and Hypouricemic Patients With SARS
| Hypouricemic (n = 16) | Normouricemic (n = 44) | P | |
|---|---|---|---|
| Lowest count of | |||
| White blood cells (/μL) | 3,909 ± 2,265 | 4,468 ± 3,191 | 0.963 |
| Absolute neutrophils (/μL) | 367 ± 191 | 450 ± 243 | 0.237 |
| Peak levels of | |||
| White blood cells (/μL) | 12,681 ± 6,190 | 12,582 ± 7,603 | 0.524 |
| Blood urea nitrogen (mg/dL) | 33.8 ± 23.6 | 33.7 ± 33.1 | 0.992 |
| Serum creatinine (mg/dL) | 1.3 ± 1.1 | 1.5 ± 1.6 | 0.617 |
| Creatinine kinase (U/L) | 23,159 ± 85,438 | 646 ± 2,239 | 0.342 |
| Aspartate aminotransferase (U/L) | 88 ± 76 | 92 ± 78 | 0.855 |
| Alanine aminotransferase (U/L) | 121 ± 151 | 112 ± 159 | 0.862 |
| Lactate dehydrogenase (U/L) | 1,104 ± 646 | 1,330 ± 1,403 | 0.554 |
| C-reactive protein (mg/dL) | 6.6 ± 4.3 | 6.5 ± 4.7 | 0.969 |
| Electrolytes and FE | |||
| Sodium (mEq/L) | 137.5 ± 4.6 | 136.6 ± 5.4 | 0.586 |
| Potassium (mEq/L) | 4.0 ± 0.4 | 4.0 ± 0.62 | 0.857 |
| Calcium (mg/dL) | 7.6 ± 0.4 | 7.6 ± 0.8 | 0.523 |
| Phosphate (mg/dL) | 3.3 ± 0.4 | 3.4 ± 0.7 | 0.860 |
| FEUA (%) | 40.14 ± 23.60 | 16.11 ± 11.38 | 0.005 |
| FE of sodium (%) | 0.68 ± 0.55 | 0.65 ± 0.94 | 0.895 |
| FE of potassium (%) | 9.20 ± 4.50 | 8.04 ± 5.30 | 0.472 |
| Urine osmolarity (mOsmol/kgH2O) | 431 ± 168 | 457 ± 211 | 0.710 |
NOTE. Data represent the lowest observed level of serum UA. Values are expressed as means ± SD. To convert blood urea nitrogen in mg/dL to mmol/L, multiply by 0.357; serum creatinine in mg/dL to μmol/L, multiply by 88.4; sodium and potassium in mEq/L to mmol/L, multiply by 1; calcium in mg/dL to mmol/L, multiply by 0.2495; phosphate in mg/dL to mmol/L, multiply by 0.3229.
Excretion of uric acid
Forty-three patients (12 hypouricemic, 31 normouricemic) had urine samples available to calculate FEUA and electrolyte levels days 5 to 11 after fever onset. FEUA of the hypouricemic group was significantly greater than that of the normouricemic group (39.6% ± 23.4% versus 16.4% ± 11.4%; P < 0.0001). All hypouricemic patients had inappropriate uricosuria (FEUA > 10%). The inverse correlation of FEUA with serum UA level was significant (r = −0.565; P < 0.001; Fig 2). Three hypouricemic patients had a positive urine glucose test result detected with Multistix (Bayer, Taiwan), including 2 patients with diabetes with plasma glucose levels greater than 200 mg/dL (>11.1 mmol/L).
Fig 2.
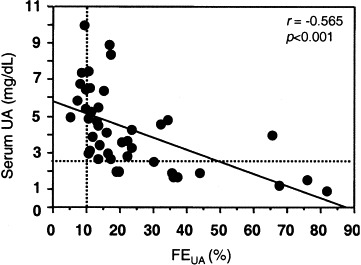
The relationship between serum UA level and FEUA in 43 patients with SARS. Dashed lines indicate FEUA of 10% (horizontal) and serum UA level of 2.5 mg/dL (149 μmol/L; vertical). To convert UA in mg/dL to μmol/L, multiply by 59.48.
Effects of uric acid on clinical outcomes
Most patients were treated with ribavirin, 1,200 mg/d; methylprednisolone, 2 mg/kg/d for 5 days; and, depending on the patient’s condition, intravenous immunoglobulin, 1 g/kg/d for 2 days.9 There was no difference in treatment regimens between the 2 groups (Table 3). Logistic regression analysis did not show a significant correlation between serum UA levels and medication, except for a modest correlation between ribavirin treatment and FEUA (P = 0.074). There were no differences between the 2 groups in duration of fever, hospital stay, or incidence of ARF. However, more hypouricemic patients needed endotracheal intubation for ventilation support than normouricemic patients (P = 0.026; Table 3). Twenty patients underwent endotracheal intubation for mechanical ventilation, and their FEUA was greater than that of patients who did not undergo mechanical ventilation (P = 0.023; Fig 3).
Table 3.
Treatment and Clinical Outcomes
| Hypouricemia (n = 16) | Normouricemia (n = 44) | P | |
|---|---|---|---|
| Treatment | |||
| Ribavirin | 12 (75) | 41 (93.18) | 0.074 |
| Methylprednisolone | 15 (93.75) | 39 (88.64) | 0.488 |
| Intravenous immunoglobulin | 11 (68.75) | 25 (56.81) | 0.299 |
| Clinical outcomes | |||
| Duration of fever (d) | 8.1 ± 4.7 | 9.6 ± 3.8 | 0.224 |
| Hospital stay (d) | 35.9 ± 31.4 | 26.2 ± 24.7 | 0.261 |
| ARF | 3 (18.75) | 8 (18.18) | 0.614 |
| Death | 5 (31.25) | 9 (20.45) | 0.292 |
| Endotracheal intubation | 9 (56.3) | 11 (25) | 0.026 |
| Death and/or intubation | 11 (69.8) | 12 (27.3) | 0.005 |
NOTE. Results expressed as number (percent) or mean ± SD.
Fig 3.
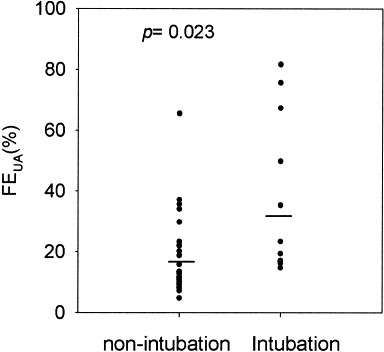
FEUA of patients who did versus did not need endotracheal intubation. Lines indicate mean values for the 2 groups.
Five hypouricemic patients and 9 normouricemic patients died. Twenty-three patients in our cohort (38.3%) had catastrophic outcomes (endotracheal intubation and/or death). The number of catastrophic outcomes adjusted for older age and sex showed that hypouremic patients had an odds ratio of 10.57 (confidence interval, 2.33 to 47.98; P = 0.002). Kaplan-Meier curves for catastrophic outcome–free results showed significant differences between patients with normouricemia or hypouricemia (log rank P = 0.01; Fig 4).
Fig 4.
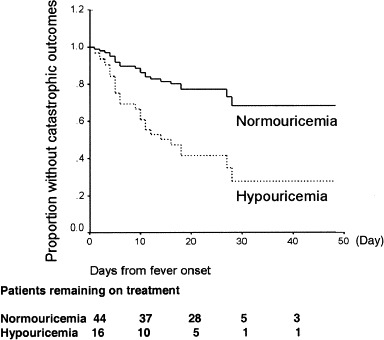
Kaplan-Meier survival estimates of patients with SARS according to hypouricemic and normouricemic groups; P = 0.01 by log-rank test.
Factors associated with increased uric acid excretion
FEUA correlated with peak creatine kinase level (P = 0.014; r = 0.376) measured within the first 15 days and with need for endotracheal intubation for supportive ventilation (P = 0.003; r = 0.448), but correlated negatively with blood oxygen saturation (P = 0.001; r = −0.624). After adjustments for age and sex, FEUA remained significantly associated with blood oxygen saturation (P = 0.001; r = −0.624; adjusted r 2 = 0.389). Moreover, FEUA was greater in patients who needed endotracheal intubation for ventilation support than in those who did not (P = 0.003; Fig 4).
Serum levels of the proinflammatory cytokines IL-6, IL-8, and TNF-α were measured in 16 patients (6 hypouricemic, 10 normouricemic) during their UA excretion studies. IL-8 levels correlated significantly with FEUA (P < 0.001; r = 0.785) and inversely with serum UA level (P = 0.044; r = −0.509; Fig 5). However, similar correlations were not observed for either IL-6 or TNF-α level.
Fig 5.
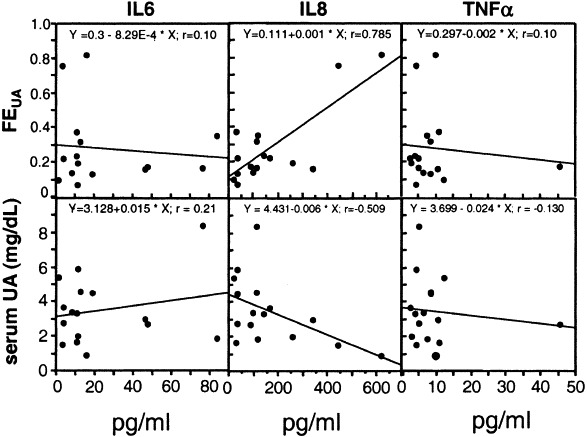
Relationships between FEUA and serum IL-6, IL-8, and TNF-α levels in 16 patients with SARS.
Discussion
In our study, more than one fourth of patients with SARS had hypouricemia, a clinical feature not reported previously. Considering that ribavirin treatment may increase serum UA levels,17 the incidence of hypouricemia in patients with SARS may be underestimated. All hypouricemic patients in the present study had inappropriate uricosuria. The inverse correlation between serum UA level and FEUA indicates that the hypouricemia in patients with SARS resulted from an abnormal increase in UA excretion during SARS-CoV infection. Although SARS-CoV could be detected in patients’ urine, most of the patients with SARS we studied had normal renal function. Acute interstitial nephritis has been associated with various viral infections, but not reported in SARS-CoV infection. We had reported 2 necropsy cases of patients with SARS with ARF and did not find glomerular or tubular lesions.4
Several diseases or factors may cause renal hypouricemia, eg, obstructive jaundice, solid or hematologic neoplasias, diabetes mellitus, and drugs affecting urate homeostasis.14 Renal hypouricemia associated with infections has been reported infrequently.18, 19 Although a substantial proportion of patients with acquired immunodeficiency syndrome have presented with renal hypouricemia, co-trimoxazole was thought to be a contributory factor.19, 20 Causes of renal hypouricemia in patients with other systemic infections are not known.
Four complementary DNAs that express proteins transporting urate recently have been cloned.21 Mutational defects of URAT1 (SLC22A12) have been identified in patients with renal hypouricemia,22, 23 but the mechanisms of urate transport regulation are not fully understood. Although proinflammatory cytokine data were obtained from a small proportion of our patients with SARS, it is interesting that serum IL-8 level correlated significantly with FEUA. It has been shown that proinflammatory cytokines can influence UA excretion24 and modulate the activities of channels and transporters by various mechanisms.25, 26 To our knowledge, the effect of cytokines on urate transport has not been investigated. Previous reports indicated that the mean time for immunoglobulin G seroconversion of SARS-CoV occurs on day 10,2 accompanied by a decline in serum levels of such proinflammatory cytokines as IL-6, IL-8, and TNF-α in the convalescent stage.27 Our study showed that the lowest serum UA level occurred days 7 to 9 after fever onset, when the cytokine storm of patients with SARS usually occurred.27
Hypouricemia is a poor prognostic indicator in patients with intra-abdominal sepsis18 or radiating pneumonitis.28 In patients with acquired immunodeficiency syndrome with infections of the central nervous system, renal hypouricemia also is an ominous sign.19 The present study shows that hypouricemic patients with SARS had a poor outcome, especially in terms of respiratory failure, compared with normouricemic patients. Moreover, Kaplan-Meier curves for catastrophic outcome–free results showed significant differences between patients with normouricemia or hypouricemia. In addition, FEUA correlated inversely with blood oxygenation. UA is the main metabolite of adenosine triphosphate degradation, and increased UA excretion has been observed in patients with respiratory failure.29 However, in such patients, serum UA levels were not decreased or changed by treatment,29 implicating increased UA production, rather than abnormal UA renal handling, in hypoxic patients. We speculate that both hypoxia and hypouricemia in patients with SARS indicate the severity of SARS-CoV infection, which affects both ventilation and UA excretion. In vivo and in vitro studies30, 31 have shown UA to be an important antioxidant, contributing as much as 60% of free-radical scavenging in human serum.32 A prospective, randomized, controlled clinical trial showed that a nucleotide-supplemented diet could significantly reduce complications and shorten hospital stay after admission to an intensive care unit,33 suggesting serum UA as a possible surrogate of antioxidant capacity.34 It is not clear whether poor outcomes of hypouricemic patients with SARS are caused in part by a shortage of antioxidants.
In summary, hypouricemia resulting from abnormal renal urate handling is not rare in patients with SARS-CoV infection and may reflect the severity of disease and predict poor patient outcomes. The significance of the relationship between IL-8 level and FEUA needs further investigation.
Acknowledgment
The authors thank Ming-Yi Chung for technical support and Dr Tzi-Yi Chuang for help in data collection.
Footnotes
The SARS Research Group of National Taiwan University College of Medicine and National Taiwan University Hospital includes the following members: Ding-Shinn Chen, Yuan-Teh Lee, Che-Ming Teng, Pan-Chyr Yang, Hong-Nerng Ho, Pei-Jer Chen, Ming-Fu Chang, Jin-Town Wang, Shan-Chwen Chang, Chuan-Liang Kao, Wei-Kung Wang, Cheng-Hsiang Hsiao, and Po-Ren Hsueh.
Supported in part by the Improving Dialysis Quality Research Funds and Ta-Tung Kidney Foundation.
References
- 1.Ksiazek T.G., Erdman D., Goldsmith C.S. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953–1966. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 2.Peiris J.S., Chu C.M., Cheng V.C. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: A prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tsai H.B., Huang J.W., Chen K.Y. Acute renal failure and renal replacement therapy in SARS patients. Presented at the Annual Committee on American Society of Nephrology, San Diego, CA, November 12–17. 2003. (abstr) [Google Scholar]
- 4.Wu V.C., Hsueh P.R., Lin W.C. Acute renal failure in SARS patients: More than rhabdomyolysis. 2003. Presented at the 20th Annual Committee on Taiwan Society of Nephrology, Taipei, Taiwan, December 13–14 (abstr) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vas S.I. Primary and secondary role of viruses in chronic renal failure. Kidney Int Suppl. 1991;35:S2–S4. [PubMed] [Google Scholar]
- 6.Glassock R.J. Immune complex-induced glomerular injury in viral diseases: An overview. Kidney Int Suppl. 1991;35:S5–S7. [PubMed] [Google Scholar]
- 7.Maesaka J.K., Cusano A.J., Thies H.L. Hypouricemia in acquired immunodeficiency syndrome. Am J Kidney Dis. 1990;15:252–257. doi: 10.1016/s0272-6386(12)80770-0. [DOI] [PubMed] [Google Scholar]
- 8.Ogino K., Hisatome I., Saitoh M. Clinical significance of hypouricemia in hospitalized patients. J Med. 1991;22:76–82. [PubMed] [Google Scholar]
- 9.Wang J.T., Wang J.L., Fang C.T., Chang S.C. Early defervescence and SARS recovery. Emerg Infect Dis. 2004;10:544–545. doi: 10.3201/eid1003.030500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Health Organization . Case Definitions for Surveillance of Severe Acute Respiratory Syndrome (SARS) 2003. Available at: http://www.who.int/csr/don/2003_04_19/en/. Accessed: May 5. [Google Scholar]
- 11.Centers for Disease Control and Prevention . SARS Coronavirus Sequencing. 2003. Available at: http://www.cdc.gov.ncidod/sars/sequence.htm. Accessed: April 14. [Google Scholar]
- 12.Wang J.T., Sheng W.H., Fang C.T. Clinical manifestations, laboratory findings, and treatment outcomes of SARS patients. Emerg Infect Dis. 2004;10:818–824. doi: 10.3201/eid1005.030640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Maesaka J.K., Fishbane S. Regulation of renal urate excretion: A critical review. Am J Kidney Dis. 1998;32:917–933. doi: 10.1016/s0272-6386(98)70067-8. [DOI] [PubMed] [Google Scholar]
- 14.Bairaktari E.T., Kakafika A.I., Pritsivelis N. Hypouricemia in individuals admitted to an inpatient hospital-based facility. Am J Kidney Dis. 2003;41:1225–1232. doi: 10.1016/s0272-6386(03)00355-x. [DOI] [PubMed] [Google Scholar]
- 15.Bairaktari E., Liamis G., Tsolas O., Elisaf M. Partially reversible renal tubular damage in patients with obstructive jaundice. Hepatology. 2001;33:1365–1369. doi: 10.1053/jhep.2001.25089. [DOI] [PubMed] [Google Scholar]
- 16.Booth C.M., Matukas L.M., Tomlinson G.A. Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area. JAMA. 2003;289:2801–2809. doi: 10.1001/jama.289.21.JOC30885. [DOI] [PubMed] [Google Scholar]
- 17.Moreno-Monteagudo J.A., Fernandez-Bermejo M., Garcia-Buey L. Interferon alpha with ribavirin for the treatment of chronic hepatitis C in non-responders or relapsers to interferon monotherapy. Aliment Pharmacol Ther. 1998;12:717–723. doi: 10.1046/j.1365-2036.1998.00359.x. [DOI] [PubMed] [Google Scholar]
- 18.Abou-Mourad N.N., Chamberlain B.E., Ackerman N.B. Poor prognosis of patients with intra-abdominal sepsis and hypouricemia. Surg Gynecol Obstet. 1979;148:358–360. [PubMed] [Google Scholar]
- 19.Collazos J., Blanco M.S., Guerra E. Sequential evaluation of serum urate concentrations in AIDS patients with infections of the central nervous system. Clin Chem Lab Med. 2000;38:1293–1296. doi: 10.1515/CCLM.2000.204. [DOI] [PubMed] [Google Scholar]
- 20.Chertow G.M., Seifter J.L., Christiansen C.L., O’Donnell W.J. Trimethoprim-sulfamethoxazole and hypouricemia. Clin Nephrol. 1996;46:193–198. [PubMed] [Google Scholar]
- 21.Rafey M.A., Lipkowitz M.S., Leal-Pinto E., Abramson R.G. Uric acid transport. Curr Opin Nephrol Hypertens. 2003;12:511–516. doi: 10.1097/00041552-200309000-00005. [DOI] [PubMed] [Google Scholar]
- 22.Enomoto A., Kimura H., Chairoungdua A. Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature. 2002;417:447–452. doi: 10.1038/nature742. [DOI] [PubMed] [Google Scholar]
- 23.Ichida K., Hosoyamada M., Hisatome I. Clinical and molecular analysis of patients with renal hypouricemia in Japan—Influence of URAT1 gene on urinary urate excretion. J Am Soc Nephrol. 2004;15:164–173. doi: 10.1097/01.asn.0000105320.04395.d0. [DOI] [PubMed] [Google Scholar]
- 24.Urano W., Yamanaka H., Tsutani H. The inflammatory process in the mechanism of decreased serum uric acid concentrations during acute gouty arthritis. J Rheumatol. 2002;29:1950–1953. [PubMed] [Google Scholar]
- 25.Puma C., Danik M., Quirion R. The chemokine interleukin-8 acutely reduces Ca(2+) currents in identified cholinergic septal neurons expressing CXCR1 and CXCR2 receptor mRNAs. J Neurochem. 2001;78:960–971. doi: 10.1046/j.1471-4159.2001.00469.x. [DOI] [PubMed] [Google Scholar]
- 26.Qiu M.R., Campbell T.J., Breit S.N. A potassium ion channel is involved in cytokine production by activated human macrophages. Clin Exp Immunol. 2002;130:67–74. doi: 10.1046/j.1365-2249.2002.01965.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hsueh P.R., Chen P.J., Hsiao C.H. Patient data, early SARS epidemic, Taiwan. Emerg Infect Dis. 2004;10:489–493. doi: 10.3201/eid1003.030571. [DOI] [PubMed] [Google Scholar]
- 28.Wang J.Y., Chen K.Y., Wang J.T. Outcome and prognostic factors for patients with non-small-cell lung cancer and severe radiation pneumonitis. Int J Radiat Oncol Biol Phys. 2002;54:735–741. doi: 10.1016/s0360-3016(02)02994-2. [DOI] [PubMed] [Google Scholar]
- 29.Sahebjami H. Changes in urinary uric acid excretion in obstructive sleep apnea before and after therapy with nasal continuous positive airway pressure. Chest. 1998;113:1604–1608. doi: 10.1378/chest.113.6.1604. [DOI] [PubMed] [Google Scholar]
- 30.Hink H.U., Santanam N., Dikalov S. Peroxidase properties of extracellular superoxide dismutase: Role of uric acid in modulating in vivo activity. Arterioscler Thromb Vasc Biol. 2002;22:1402–1408. doi: 10.1161/01.atv.0000027524.86752.02. [DOI] [PubMed] [Google Scholar]
- 31.Ames B.N., Cathcart R., Schwiers E., Hochstein P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: A hypothesis. Proc Natl Acad Sci U S A. 1981;78:6858–6862. doi: 10.1073/pnas.78.11.6858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Maxwell S.R., Thomason H., Sandler D. Antioxidant status in patients with uncomplicated insulin-dependent and non-insulin-dependent diabetes mellitus. Eur J Clin Invest. 1997;27:484–490. doi: 10.1046/j.1365-2362.1997.1390687.x. [DOI] [PubMed] [Google Scholar]
- 33.Bower R.H., Cerra F.B., Bershadsky B. Early enteral administration of a formula (Impact) supplemented with arginine, nucleotides, and fish oil in intensive care unit patients: Results of a multicenter, prospective, randomized, clinical trial. Crit Care Med. 1995;23:436–449. doi: 10.1097/00003246-199503000-00006. [DOI] [PubMed] [Google Scholar]
- 34.Hsu S.P., Pai M.F., Peng Y.S. Serum uric acid levels show a ’J-shaped’ association with all-cause mortality in haemodialysis patients. Nephrol Dial Transplant. 2004;19:457–462. doi: 10.1093/ndt/gfg563. [DOI] [PubMed] [Google Scholar]


