Abstract
The human respiratory tract virome is defined here as the viruses present in the human respiratory tract that can infect human cells. Sensitive, culture-independent molecular assays (polymerase chain reaction and high-throughput sequencing) reveal that in addition to common viruses that cause acute, symptomatic infections the virome also includes viruses that do not cause clinical symptoms, have unknown pathogenic effect, or cause symptoms but are not among the most common viral respiratory tract pathogens. These molecular tools provide means for better defining the virome and studying the effects of viral infections on the dynamics of chronic lung diseases.
Keywords: Virus, Infection, Lung, Culture independent, Chronic lung disease, Diagnostics, High-throughput sequencing
Key points
-
•
Culture-independent molecular assays detect viral pathogens with great sensitivity and can be used to define the virome in the upper and lower respiratory tract.
-
•
The respiratory tract virome is defined by very common pathogens (rhinoviruses, paramyxoviruses) as well as viruses that occur less frequently and those with unknown pathogenicity.
-
•
Viruses with the potential for pathogenicity are detected in both symptomatic and asymptomatic people.
-
•
Monitoring emerging respiratory pathogens is important, and high-throughput sequencing can be used as a tool to complement epidemiologic studies and to design diagnostics.
-
•
In the future, comprehensive pathogen detection and host response may be coupled to create better assays for research studies and diagnostics.
Introduction
Viral infections of the respiratory tract are very common. In a recent study of 26 households in Utah that were followed weekly over 1 year, modern molecular methods were used to detect respiratory viruses in the anterior nares.1 This study found that children less than 5 years old had about 12 viral episodes in the respiratory tract each year, whereas adults averaged about 6 per year. These numbers are higher than previous studies,2 which is likely explained by the use of molecular assays instead of culture- and serology-based tests and the discovery of new respiratory viruses in intervening years that would not have been assessed in older studies.
Modern molecular methods for virus detection are highly sensitive and specific. Polymerase chain reaction (PCR) assays are also rapid and generally inexpensive. High-throughput nucleic acid sequencing (HTS) methods are slower but have the potential to be more comprehensive because there is no need to select specific targets beforehand, and the method can detect genomes with substantial sequence variation compared with known reference genomes (Fig. 1 ). With these tools in hand, we can begin to think about characterizing the virome of the respiratory tract, herein defined as all of the viruses in the respiratory tract that can infect and replicate in human cells, which includes known pathogens and viruses with unknown pathogenicity. We have begun to learn about the virome of the respiratory tract through studies of patients with acute infections, chronic lung diseases, and undergoing lung transplantation, among others. The author reviews some of these studies in addition to recent technological developments, which will improve characterization of the respiratory virome and diagnostics in coming years.
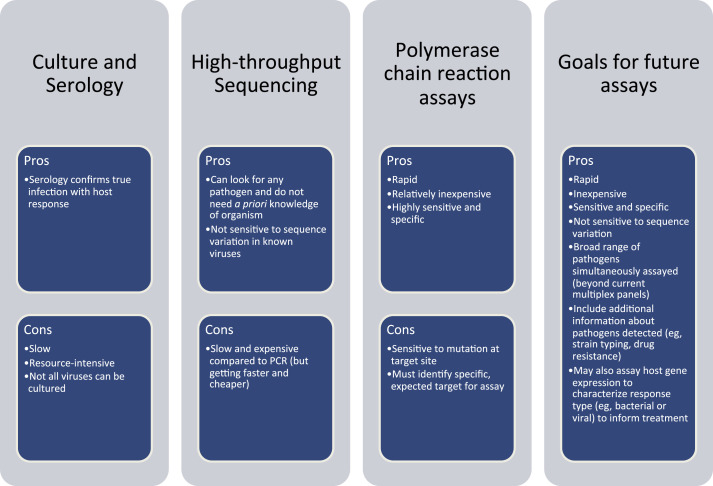
Fig. 1.
Methods for characterizing viruses in the respiratory tract. Current molecular methods, such as PCR and HTS, have clear advantages over older methods (culture and serology) in terms of cost, speed, and sensitivity. Future assays for research and diagnostics will be aimed at capturing and improving on the best features of the current methods.
Overview of the virome in the respiratory tract
Defining the virome in the respiratory tract and understanding the implications of the viruses detected are significant challenges. The work is complicated by several factors. First, the lower airway is not easily accessible and sometimes requires invasive sampling. For instance, bronchoalveolar lavage samples are often only available from symptomatic individuals who are having lavage performed for diagnostic testing and not from asymptomatic controls. Second, a study of the viruses in the lungs of patients with cystic fibrosis (CF) showed that the viral populations were distinct in different regions of the lung.3 This variation within the respiratory tract and lung means it can be difficult to get a clear, or complete, view of the virome. Third, only recently have relatively unbiased approaches to identifying viruses become available in the form of HTS assays. With that said, a great deal of progress has been made in defining the human virome in the respiratory tract (summarized in Table 1 ).
Table 1.
Common viruses detected in the respiratory tract virome
| Virus Groups | Species or Types | References from this Review |
|---|---|---|
| Picornaviruses | Rhinoviruses A, B, and/or C | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Colvin et al,8 2012; Wylie et al,9 2012; Flight et al,13 2014; Goffard et al,14 2014; Wat et al,15 2008; Graf et al,24 2016; Thorburn et al,25 2015; Zoll et al,26 2015 |
| Enteroviruses | Colvin et al,8 2012; Wylie et al,9 2012; Wylie et al,22 2015; Wylie et al,23 2015; Graf et al,24 2016; Thorburn et al,25 2015; Zoll et al,26 2015 | |
| Parechovirus | Wylie et al,9 2012 | |
| Paramyxoviruses | Respiratory syncytial virus | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Flight et al,13 2014; Wat et al,15 2008; Graf et al,24 2016; Thorburn et al,25 2015; Zoll et al,26 2015 |
| Parainfluenzaviruses 1–4 | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Colvin et al,8 2012; Wylie et al,9 2012; Flight et al,13 2014; Goffard et al,14 2014; Wat et al,15 2008; Graf et al,24 2016; Thorburn et al,25 2015 | |
| Metapneumovirus | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Colvin et al,8 2012; Graf et al,24 2016; Thorburn et al,25 2015; Zoll et al,26 2015 | |
| Measles virus | Lysholm et al,4 2012; Wang et al,5 2016; Wylie et al,9 2012; Flight et al,13 2014; Graf et al,24 2016 | |
| Pneumovirus | Wylie et al,9 2012 | |
| Orthomyxoviruses | Influenzavirus A, B, and/or C | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Colvin et al,8 2012; Wylie et al,9 2012; Flight et al,13 2014; Goffard et al,14 2014; Wat et al,15 2008; Graf et al,24 2016; Thorburn et al,25 2015 |
| Coronaviruses | HKU1, OC43, 229E, and/or NL63 | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Colvin et al,8 2012; Wylie et al,9 2012; Goffard et al,14 2014; Wat et al,15 2008; Graf et al,24 2016; Thorburn et al,25 2015 |
| Adenoviruses | Adenovirus C or untyped | Lysholm et al,4 2012; Wang et al,5 2016; Jain et al,6 2015; Jain et al,7 2015; Colvin et al,8 2012; Wylie et al,9 2012; Flight et al,13 2014; Graf et al,24 2016; Thorburn et al,25 2015 |
| Parvoviruses | Bocavirus or unclassified | Lysholm et al,4 2012; Wang et al,5 2016; Colvin et al,8 2012; Wylie et al,9 2012; Willner et al,16 2009; Young et al,17 2015; Graf et al,24 2016; Zoll et al,26 2015 |
| Herpesviruses | Cytomegalovirus, Epstein-Barr virus, Roseolovirus, and/or Kaposi sarcomavirus | Wang et al,5 2016; Wylie et al,9 2012; Willner et al,16 2009; Young et al,17 2015; Graf et al,24 2016 |
| Anelloviruses | Torque teno virus, torque teno midi virus, and/or torque teno mini virus or untyped | Lysholm et al,4 2012; Wang et al,5 2016; Wylie et al,9 2012 |
| Papillomaviruses | Various | Wang et al,5 2016; Willner et al,16 2009; Young et al,17 2015 |
| Polyomaviruses | KI and/or WU | Lysholm et al,4 2012; Colvin et al,8 2012; Wylie et al,9 2012 |
The Virome in Patients with Respiratory Tract Infections and Controls
One cost-effective approach to broadly identify viruses associated with the respiratory tract is to pool samples and screen for a comprehensive set of viruses. The downside to this approach is that one cannot determine the frequency at which any individual virus occurs among patients. However, as characterization of the respiratory tract virome using molecular methods is a relatively new area of exploration, these studies can be useful in order to determine if viruses beyond the common, known respiratory pathogens are detected.
In one study, 210 adults and children with severe lower respiratory tract infections were sampled.4 Nasopharyngeal aspirates were collected and samples were combined, creating 13 pools of 8 to 24 samples per pool. Virus particles were enriched, and DNA and RNA viruses were assessed using HTS. Thirty-nine viral species were observed in these samples, giving a broad view of the scope of the respiratory tract virome during infection. Based on read counts, the most abundant viruses in the data set were the paramyxoviruses (including human respiratory syncytial virus, human metapneumovirus). Picornaviruses were the next most abundant (primarily rhinoviruses A and C). Orthomyxoviruses were the third most abundant (influenza viruses A, B, and C). There were several rare and/or unexpected viruses represented at low abundance. These viruses included bocavirus, KI polyomavirus, picobirnavirus, measles virus, and anelloviruses. In a study from China, a similar sequencing-based approach was taken to characterize the virome in children less than 6 years old with severe acute respiratory illness and 15 controls without respiratory illness.5 Nasopharyngeal swabs were pooled into 9 pools of 15 samples each; virus particles were enriched; and HTS data were generated to assess both RNA and DNA viruses. The most highly represented viruses included the paramyxoviruses (primarily human respiratory syncytial virus), and the other common viruses detected in the study described earlier were detected. This study also detected human coronaviruses, bocaviruses, picornaviruses, influenza viruses, adenoviruses, and anelloviruses. Rare sequences included those from metapneumovirus, measles, hepatitis B and C, papillomavirus, and others. These two studies demonstrate the power of HTS compared with targeted PCR assays for defining the virome. In both studies, common and expected respiratory pathogens were detected. However, other viruses, some with unknown pathogenicity in the respiratory tract, were also detected. Detection of these viruses is valuable as we aim to fully understand the biology of the respiratory tract. Although these studies were exploratory, they raise questions about whether some of the viruses with unclear pathogenicity could be contributing to the presentation of symptoms, complicating the course of infection with other pathogens, or are biomarkers for infection or host response. These questions remain to be addressed in future studies.
The Virome in Children and Adults with Pneumonia
Viruses are clearly an important cause of pneumonia, particularly in the postpneumococcal vaccine era. A prospective multicenter study sponsored by the Centers for Disease Control and Prevention focused on the Etiology of Pneumonia in the Community (EPIC).6, 7 This study used extensive diagnostic testing (culture, serology, molecular testing) to understand the causes of pneumonia in more than 2000 adults in the United States, in the time after the implementation of the pneumococcal vaccine. In this study, a pathogen was detected in 38% of the samples. At least 1 virus was detected in 23% of the samples available for testing, and 3% had both bacterial and viral pathogens detected. Rhinoviruses and influenza viruses were the most common pathogens detected in 9% and 6% of patients, respectively. The EPIC study took a similar approach to study the cause of community-acquired pneumonia in children. Again, more than 2000 subjects were enrolled. The children were less than 18 years old, with a median age of 2 years. A microbial pathogen was detected in 81% of samples. At least one virus was detected in 66% of the samples, and both bacterial and viral pathogens were detected in 7% of the children. In children, respiratory syncytial virus, rhinovirus, metapneumovirus, and adenovirus were the most common pathogens. These studies identify viruses as key pathogens in pneumonia since the implementation of the pneumococcal vaccine. Furthermore, it is important to note that no pathogen was detected in 62% of adults and 19% of children, suggesting the possibility that pneumonia was caused by viruses not included in the set of targeted assays used in this study. Identification of the etiologic agent of pneumonia may benefit from unbiased HTS assays to detect unexpected or rare pathogens that are not be included in standard clinical testing.
The Virome in Children with Unexplained Fever and Asymptomatic Controls
In a study of children with unexplained fever, both targeted PCR assays8 and HTS9 were used to characterize the respiratory tract virome in individual samples from subjects. Children with fever were compared with afebrile children who were in the hospital for same-day surgery. Nasopharyngeal swabs from 75 febrile children and 116 afebrile children were tested with a panel of PCR assays that targeted common respiratory pathogens and viruses of interest, including influenza A, parainfluenza virus, metapneumovirus, rhinovirus, enterovirus, coronavirus, adenovirus, bocavirus, and the recently discovered KI and WU polyomaviruses. HTS was performed on 50 samples from febrile children and 81 samples from afebrile controls. Using sequencing, 17 viral genera were detected overall. These genera included viruses listed earlier; cytomegalovirus, parechovirus and others were detected in febrile children, whereas Roseolovirus was detected in nasopharyngeal swabs from both febrile and afebrile children. Although febrile children were more likely to have a virus present in the sample compared with controls, afebrile children still carried viruses asymptomatically. Rhinoviruses/enteroviruses and anelloviruses were particularly common in asymptomatic children in this study. However, anelloviruses, particularly torque teno virus, were associated with fever.10 This study demonstrates 2 points very well. First, even with a panel of respiratory virus PCR assays that extends far beyond targets that would be used for clinical testing, additional viruses were detected in the respiratory tract only by sequencing. This finding emphasizes the potential to improve viral diagnostic testing by broadening the set of viruses evaluated. Second, viruses are commonly found in the respiratory tracts of asymptomatic children and many of these viruses have potential to cause symptomatic infection. This point means that pathogen detection may not always be enough to make a clear diagnosis. Other information may be needed, as discussed in more detail later in this review.
The Respiratory Tract Virome in Patients with Cystic Fibrosis
Although many viral infections are mild or resolve without complication in generally healthy individuals, viruses in the respiratory tract are associated with exacerbations of chronic lung diseases, including CF, chronic obstructive pulmonary disease, and asthma (reviewed in the following11, 12). Exploration of the virome in the respiratory tracts of patients with chronic lung diseases has emphasized the prevalence of common pathogens and also further defines the scope of the respiratory tract virome.
To illustrate this point, the author discusses a few studies of the virome in patients with CF. Molecular methods of detection, specifically PCR assays, have increased the association of viral infection with pulmonary exacerbation. Incidence of viral infections in adult patients with CF was estimated at 1 to 2 viral infections per year based on a study of 100 patients who were sampled every 2 months for a year and on exacerbation.13 Samples from the respiratory tract (sputum, nose and throat swabs) were tested using PCR assays for common respiratory pathogens, including respiratory syncytial virus, rhinovirus, influenza virus, and others. Rhinovirus and metapneumovirus were the most commonly detected viruses, accounting for 72.5% and 13.2% of virus-positive samples, respectively. Viral infection was associated with pulmonary exacerbation, with virus detected in 40% of exacerbation samples compared with 24% of samples collected on regular visits. In another study, sputum was collected from 46 adult patients and viruses were screened using PCR assays.14 In this study, rhinoviruses and coronaviruses were the most common viruses, and rhinoviruses were associated with exacerbation. In children with CF, exacerbation has also been associated with viral infection. In a study of 71 patients, viruses were assessed in nasal swabs using a panel of targeted PCR assays for common respiratory pathogens.15 Viruses were detected in 46% of samples collected during exacerbation but only 17% of samples collected when asymptomatic. Influenza A, influenza B, and rhinovirus were all associated with exacerbation.
The virome of patients with CF has also been characterized using HTS. One study aimed to study the virome in sputum from 5 patients with CF and 5 healthy controls.16 Virus particles were enriched, and DNA viruses were sequenced. This study was particularly interesting because the use of HTS instead of targeted PCR assays resulted in the detection of viruses that would not have been included in typical PCR panels evaluating respiratory viruses. In the patients with CF, reticuloendotheliosis virus, Epstein-Barr virus, human herpesvirus 6B, and human herpesvirus 8 were detected. In control patients, human papillomaviruses were detected in 2 samples. Several single-stranded DNA viruses were also detected in patients and/or controls, including geminiviruses and circoviruses. In a second study, the virome was assessed in explanted lungs from patients with CF undergoing lung transplant and lungs obtained post mortem.3 Anelloviruses, papillomaviruses, and herpesviruses were detected; viruses were detected in distinct regions of the lung rather than diffusely throughout. These studies were valuable because many of the viruses detected were not those that would have been included in PCR screens for common respiratory pathogens, yet they may impact disease progression in the lung by creating or promoting inflammation in the respiratory tract.
The Respiratory Tract Virome after Lung Transplantation
In one study, the virome was studied in bronchoalveolar lavage and oral washes from lung transplant patients within the first year of transplant, human immunodeficiency virus (HIV) positive subjects without respiratory symptoms, and healthy volunteers.17 Anelloviruses, papillomaviruses, and herpesviruses were detected. The striking observation in this study was that the diversity and abundance of anelloviruses was highly increased in the transplant patients compared with patients with HIV and healthy controls. This finding was true in both the lower airway lavage samples and the upper airway oral washes. Although anelloviruses are not known to be pathogenic, they seem to be a marker of immunosuppression.18 The effects of their dysregulation on engraftment or outcome, if any, are not known at this time.
Emerging Respiratory Viruses
Since the discovery of severe acute respiratory syndrome coronavirus in 2003,19 other novel respiratory pathogens have emerged, including coronaviruses NL63, HKU1, and MERS (reviewed in20) and influenza viruses H1N1 pandemic strain and H7N9.21 In each case, the molecular tools to adequately survey the spread of the virus had to be developed. Once assays become available, the transmission of these viruses can be tracked and recommendations for protecting public health can be made. HTS assays can be useful to survey outbreaks of emerging viruses. A recent example of this occurred in the fall of 2014, when the United States experienced a widespread outbreak of enterovirus D68 in 49 states and the District of Columbia. This virus had previously been observed rarely. In multiplex PCR panels, the virus was either undetected or broadly typed only as an enterovirus/rhinovirus, which limited the study of the outbreak. Typing of the virus initially involved a labor-intensive and slow method in which an amplicon was generated and sequenced, and the sequence was compared with reference strains for typing. The genomes of the outbreak strains were sequenced22(and also by the Centers for Disease Control and Prevention), and subsequently a highly specific molecular assay was developed in order to aid in detection and typing of the outbreak strain.23 In this case, sequencing provided specific viral typing and allowed for genomic characterization and design of a specific laboratory developed test. This model could be useful for future outbreaks.
High-throughput sequencing in the clinic
Sequencing Assays Compared with Standard Clinical Tests
The workhorses for clinical testing of respiratory viruses are multiplex PCR panels that target the most common respiratory viruses. These assays are highly sensitive and yield rapid results. However, PCR-based assays can have limitations. In most multiplex panels, the assays cannot be used to subtype viruses, identify drug-resistant alleles, or identify viruses not targeted by the panel. Evolving viruses may also mutate in the region targeted by the PCR primers and be missed by the assay. For these reasons, HTS-based assays could have a role in the clinic.
Recently, several groups have compared the results from HTS with standard clinical assays for detection of viruses in the respiratory tract. One study found that RNA sequencing and the GenMark eSensor Respiratory Virus Panel (RVP) had an 86% correlation rate on one set of 42 known positive nasopharyngeal swab samples and 93% correlation on a second set of 67 samples.24 High-throughput RNA sequencing detected 12 viruses that were either not included in the RVP panel or whose sequence was divergent from the RVP target and, thus, not detected. Furthermore, viral subtypes were determined for influenza A (as well as identification of the oseltamivir resistance mutation), respiratory syncytial viruses, and rhinoviruses. Another study tested 89 nasopharyngeal swabs from adults with upper respiratory tract infections using reverse transcription (RT)-PCR assays for a series of common viruses, including human rhinoviruses, coronaviruses, influenza viruses and others, and by RNA sequencing.25 The HTS assay had a sensitivity of 77% compared with the PCR assays. The viruses that were not detected by HTS had higher cycle threshold (Ct) values in the real-time RT-PCR assays, indicating there were lower levels of viral nucleic acid present in those samples. Again, HTS had the advantage of providing additional subtyping information, in this case for human enteroviruses, rhinoviruses, metapneumovirus, and respiratory syncytial virus. A third study demonstrated that HTS could be used to detect a pathogen (rhinovirus C) in a sample from a child with respiratory symptoms in which no virus had been detected by PCR.26 Taken together, these data show that in some cases HTS could be advantageous compared with PCR assays, but sensitivity of sequencing can be a limitation.
Improving Sensitivity of High-Throughput Nucleic Acid Sequencing for Virus Detection
The studies comparing clinical tests with HTS demonstrate that sequencing can add information to clinical assays in terms of typing viruses and detecting resistance mutations. Importantly, they illustrate that viruses not included in the PCR panels are sometimes present. In each case, despite the slightly different method used for sample collection and preparation and the different patient cohorts used, sensitivity for detection was an issue. Recently 2 groups developed an approach to enrich viral nucleic acids from a comprehensive set of viruses before sequencing.27, 28 This approach uses targeted sequence capture, a hybridization-based approach for selecting targets of interest. Probes or baits are made using target sequences, and these are hybridized to the nucleic acid in the sample of interest. The baits are then captured along with the sequences that hybridized to them and washed; the result is an enrichment of the target nucleic acids in the sequencing assays. These newly developed target-based enrichment strategies do not target specific viruses or viral families that are expected to be associated with a disease, but rather they include targets for all viruses that are known to infect vertebrates, allowing for a comprehensive screen of both expected and unexpected viruses in the same assay. This kind of approach greatly improves sequencing sensitivity, with the percentage of viral reads increasing from approximately 10 to approximately 10,000 fold using targeted sequence capture compared with standard HTS.27, 28 As a result, virus targeted sequence capture may be particularly useful in helping to diagnose infections where no virus has been detected by routine methods. Failures of routine tests occur because the virus has divergent sequence from the target in the PCR panel, the virus is an emerging infectious disease, or the virus is not one of the prominent causes of respiratory infection.
Host Response to Infection
Interestingly, sensitive molecular methods currently used for diagnostics and many research studies demonstrate that viruses can frequently be detected in asymptomatic individuals. In the Utah study of 26 households mentioned earlier, bocaviruses and rhinoviruses could frequently be detected in asymptomatic children and adults.1 A Missouri study that used both PCR assays8 and HTS9 demonstrated that viral nucleic acid could be detected in nasopharyngeal swabs from asymptomatic children, in particular enteroviruses/rhinoviruses. It is necessary to appreciate that the detection of viral nucleic acid with molecular methods does not necessarily indicate the virus had infected the cell and/or was successfully replicating or that symptoms are necessarily resultant from the particular virus that was detected.
Additional information regarding the host response can be used to determine whether symptoms are caused by viral or bacterial pathogens (Fig. 2 ). Using a set of 30 samples from febrile children and 22 samples from afebrile controls, Hu and colleagues29 demonstrated that there were distinct host gene expression patterns in the blood that distinguished viral and bacterial infections; furthermore, symptomatic and asymptomatic infections could be clearly delineated. Similarly, in 118 adults with lower respiratory tract infections and 40 healthy controls, host gene expression in the blood could distinguish viral and bacterial infections with 95% sensitivity and 92% specificity.30 Bacterial-viral coinfections could also be distinguished. Tsalik and colleagues used publicly available microarray data to develop a host gene expression classifier that could distinguish bacterial, viral, and noninfectious illnesses with 87% accuracy.31 Both the Suarez and colleagues30 and Tsalik and colleagues studies showed that gene expression profiling performed better than procalcitonin. In the future, one might imagine that diagnostics may couple pathogen detection with host response to provide clinicians clear results that indicate whether there is a need for antibiotics in each case. In fact, approaches that couple pathogen detection and host response are being put forth as highly effective diagnostic approach; software tools are being developed to rapidly provide reports that may in the very near future be used by clinicians for diagnostic purposes.32
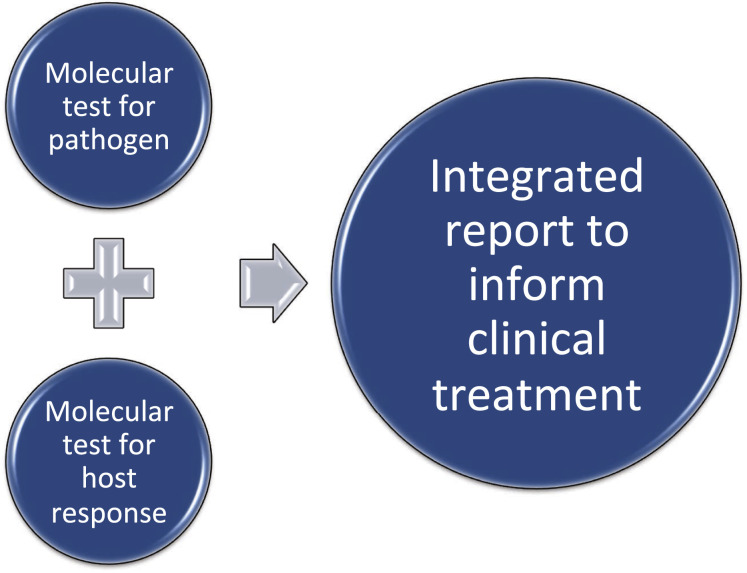
Fig. 2.
Future diagnostics. In the future, respiratory tract infections may be diagnosed by merging pathogen detection (the current method for diagnostics) with host response measures that further define the cause of the symptoms (viral, bacterial, coinfections, not pathogenic). This merger will help clarify diagnoses and define appropriate treatment measures.
Summary
We are beginning to define the scope of the human respiratory tract virome. The prevalence of individual viruses vary from study to study (see Table 1), likely due to differences in seasonality of sample collection, variation in local virus circulation, and methodological choices (eg, sample type, sample preparation). The studies reviewed here demonstrate that HTS and expanded panels of PCR assays can identify rare viral pathogens that might not be included in multiplex diagnostic panels or PCR panels in which only the most common pathogens or viruses of interest are selected. The relatively unbiased sequencing approach can also reveal viruses that may not directly cause respiratory illness but whose presence may impact the trajectory of illness through mechanisms we do not yet understand. Methodological improvements to virus detection will help us better define the respiratory tract virome and monitor outbreaks. In the future, clinical tests may include both pathogen detection and an assessment of host response in order to more clearly distinguish viral and bacterial infections.
Footnotes
Disclosure: The author has nothing to disclose.
References
- 1.Byington C.L., Ampofo K., Stockmann C. Community surveillance of respiratory viruses among families in the Utah better identification of germs-longitudinal viral epidemiology (BIG-LoVE) study. Clin Infect Dis. 2015;61(8):1217–1224. doi: 10.1093/cid/civ486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Monto A.S. Studies of the community and family: acute respiratory illness and infection. Epidemiol Rev. 1994;16(2):351–373. doi: 10.1093/oxfordjournals.epirev.a036158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Willner D., Haynes M.R., Furlan M. Case studies of the spatial heterogeneity of DNA viruses in the cystic fibrosis lung. Am J Respir Cell Mol Biol. 2012;46(2):127–131. doi: 10.1165/rcmb.2011-0253OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lysholm F., Wetterbom A., Lindau C. Characterization of the viral microbiome in patients with severe lower respiratory tract infections, using metagenomic sequencing. PLoS One. 2012;7(2):e30875. doi: 10.1371/journal.pone.0030875. Highlander SK. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang Y., Zhu N., Li Y. Metagenomic analysis of viral genetic diversity in respiratory samples from children with severe acute respiratory infection in China. Clin Microbiol Infect. 2016;22(5):458.e1-e9. doi: 10.1016/j.cmi.2016.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jain S., Finelli L., CDC EPIC Study Team Community-acquired pneumonia among U.S. children. N Engl J Med. 2015;372(22):2167–2168. doi: 10.1056/NEJMc1504028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jain S., Self W.H., Wunderink R.G. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373(5):415–427. doi: 10.1056/NEJMoa1500245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Colvin J.M., Muenzer J.T., Jaffe D.M. Detection of viruses in young children with fever without an apparent source. Pediatrics. 2012;130(6):e1455–e1462. doi: 10.1542/peds.2012-1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wylie K.M., Mihindukulasuriya K.A., Sodergren E. Sequence analysis of the human virome in febrile and afebrile children. PLoS One. 2012;7(6):e27735. doi: 10.1371/journal.pone.0027735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McElvania TeKippe E., Wylie K.M., Deych E. Increased prevalence of anellovirus in pediatric patients with fever. PLoS One. 2012;7(11):e50937. doi: 10.1371/journal.pone.0050937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hendricks M.R., Bomberger J.M. Digging through the obstruction: insight into the epithelial cell response to respiratory virus infection in patients with cystic fibrosis. J Virol. 2016;90(9):4258–4261. doi: 10.1128/JVI.01864-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hewitt R., Farne H., Ritchie A. The role of viral infections in exacerbations of chronic obstructive pulmonary disease and asthma. Ther Adv Respir Dis. 2016;10(2):158–174. doi: 10.1177/1753465815618113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Flight W.G., Bright-Thomas R.J., Tilston P. Incidence and clinical impact of respiratory viruses in adults with cystic fibrosis. Thorax. 2014;69(3):247–253. doi: 10.1136/thoraxjnl-2013-204000. [DOI] [PubMed] [Google Scholar]
- 14.Goffard A., Lambert V., Salleron J. Virus and cystic fibrosis: rhinoviruses are associated with exacerbations in adult patients. J Clin Virol. 2014;60(2):147–153. doi: 10.1016/j.jcv.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wat D., Gelder C., Hibbitts S. The role of respiratory viruses in cystic fibrosis. J Cyst Fibros. 2008;7(4):320–328. doi: 10.1016/j.jcf.2007.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Willner D., Furlan M., Haynes M. Metagenomic analysis of respiratory tract DNA viral communities in cystic fibrosis and non-cystic fibrosis individuals. PLoS One. 2009;4(10):e7370. doi: 10.1371/journal.pone.0007370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Young J.C., Chehoud C., Bittinger K. Viral metagenomics reveal blooms of anelloviruses in the respiratory tract of lung transplant recipients. Am J Transplant. 2015;15(1):200–209. doi: 10.1111/ajt.13031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.De Vlaminck I., Khush K.K., Strehl C. Temporal response of the human virome to immunosuppression and antiviral therapy. Cell. 2013;155(5):1178–1187. doi: 10.1016/j.cell.2013.10.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang D., Urisman A., Liu Y.-T. Viral discovery and sequence recovery using DNA microarrays. PLoS Biol. 2003;1(2):E2. doi: 10.1371/journal.pbio.0000002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Berry M., Gamieldien J., Fielding B.C. Identification of new respiratory viruses in the new millennium. Viruses. 2015;7(3):996–1019. doi: 10.3390/v7030996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Trombetta C., Piccirella S., Perini D. Emerging influenza strains in the last two decades: a threat of a new pandemic? Vaccines (Basel) 2015;3(1):172–185. doi: 10.3390/vaccines3010172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wylie K.M., Wylie T.N., Orvedahl A. Genome sequence of enterovirus D68 from St Louis, Missouri, USA. Emerg Infect Dis. 2015;21(1):184–186. doi: 10.3201/eid2101.141605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wylie T.N., Wylie K.M., Buller R.S. Development and evaluation of an enterovirus D68 real-time reverse transcriptase PCR assay. J Clin Microbiol. 2015;53(8):2641–2647. doi: 10.1128/JCM.00923-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Graf E.H., Simmon K.E., Tardif K.D. Unbiased detection of respiratory viruses by use of RNA sequencing-based metagenomics: a systematic comparison to a commercial PCR panel. J Clin Microbiol. 2016;54(4):1000–1007. doi: 10.1128/JCM.03060-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Thorburn F., Bennett S., Modha S. The use of next generation sequencing in the diagnosis and typing of respiratory infections. J Clin Virol. 2015;69:96–100. doi: 10.1016/j.jcv.2015.06.082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zoll J., Rahamat-Langendoen J., Ahout I. Direct multiplexed whole genome sequencing of respiratory tract samples reveals full viral genomic information. J Clin Virol. 2015;66:6–11. doi: 10.1016/j.jcv.2015.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wylie T.N., Wylie K.M., Herter B.N. Enhanced virome sequencing using targeted sequence capture. Genome Res. 2015;25(12):1910–1920. doi: 10.1101/gr.191049.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Briese T., Kapoor A., Mishra N. Virome capture sequencing enables sensitive viral diagnosis and comprehensive virome analysis. MBio. 2015;6(5):e01491–e01515. doi: 10.1128/mBio.01491-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hu X., Yu J., Crosby S.D. Gene expression profiles in febrile children with defined viral and bacterial infection. Proc Natl Acad Sci USA. 2013;110(31):12792–12797. doi: 10.1073/pnas.1302968110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Suarez N.M., Bunsow E., Falsey A.R. Superiority of transcriptional profiling over procalcitonin for distinguishing bacterial from viral lower respiratory tract infections in hospitalized adults. J Infect Dis. 2015;212(2):213–222. doi: 10.1093/infdis/jiv047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tsalik E.L., Henao R., Nichols M. Host gene expression classifiers diagnose acute respiratory illness etiology. Sci Transl Med. 2016;8(322):322ra11. doi: 10.1126/scitranslmed.aad6873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Flygare S., Simmon K., Miller C. Taxonomer: an interactive metagenomics analysis portal for universal pathogen detection and host mRNA expression profiling. Genome Biol. 2016;17(1):111. doi: 10.1186/s13059-016-0969-1. [DOI] [PMC free article] [PubMed] [Google Scholar]