Abstract
The tumor microenvironment is complex with the cancer stem cell (CSC) as a member within its community. This population possesses the capacity to self-renew and to cause cellular heterogeneity of the tumor. CSCs are resistant to conventional anti-proliferative drugs. In order to be curative, it is imperative that CSCs must be eliminated by cancer therapy. A variety of dietary phytochemicals and repositioned drugs can act synergistically with conventional anti-cancer agents. In this review, we advocate the development of a novel approach, namely combination therapy by incorporating both phytochemicals and repositioned drugs to target CSCs. We cover select dietary phytochemicals (curcumin, resveratrol, EGCG, genistein) and repurposed drugs (metformin, niclosamide, thioridazine, chloroquine). Five of the eight (curcumin, resveratrol, EGCG, genistein, metformin) are listed in “The Halifax Project”, that explores “the concept of a low-toxicity ‘broad-spectrum’ therapeutic approach that could simultaneously target many key pathways and mechanisms” [1]. For these compounds, we discuss their mechanisms of action, in which models their anti-CSC activities were identified, as well as advantages, challenges and potentials of combination therapy.
Keywords: Curcumin, Metformin, Niclosamide, Resveratrol, Repurposing
Highlights
-
•
Cancer stem cells (CSCs) are the culprits for cancer drug resistance and cancer relapse.
-
•
Dietary phytochemicals (curcumin, resveratrol, epigallocatechin gallate) target CSCs.
-
•
Repositioned drugs (metformin, niclosamide, thioridazine) target CSCs.
-
•
Phytochemical-drug combinations targeting CSCs may exhibit synergistic effects.
1. Introduction to cancer and phytochemical-drug pairing
In the course of its lifespan, a somatic cell undergoes changes in genome and epigenome due to intrinsic (cell-developmental) and extrinsic (environmental) factors. When accumulated changes disrupt its regulation of cell growth and death, the resulting uncharted growth leads to cancer. Carcinogenesis is a multi-step process: initiation, promotion, progression and metastasis. The “hallmarks of cancer” denote alterations in cancer cell physiology [2]. Indeed, only a very limited number of genes (140 or so), covering a dozen signaling pathways on the processes of cell fate, cell survival and genome maintenance, are relevant and designated as “driver” mutations [3]. Histologically, a tumor is heterogeneous, consisting of cancer cells at various stages of differentiation, as well as other cell types (fibroblasts, immune cells, endothelial cells). These cells behave as members of a community; the tumor microenvironment (niche) is a complex interactive network [4].
Cancer stem cell (CSC) is the cell within a tumor that possesses the dual capacity to self-renew and cause the heterogeneous lineages of cancer cells that comprise the tumor [5]. Conventional chemotherapy targets the bulk of proliferating cancer cells. Examples are paclitaxel for several cancer types and imatinib (tyrosine kinase inhibitor) for chronic myelogenous leukemia (CML). However, CSCs are resistant to these anti-cancer drugs [6]. A curative cancer therapy must acknowledge CSC plasticity and their complete elimination.
Many CSC-targeting dietary phytochemicals can act synergistically with conventional anti-cancer agents. Recently, investigators discovered CSC-targeting repurposed drugs [7]. We advocate here a novel approach: combination therapy with the pairing of CSC-targeting phytochemicals and repositioned drugs. Our interest in ovarian cancer led us to phytochemical-drug combinations [8,9]. Anti-cancer studies have demonstrated benefits of specific phytochemical combinations over individual compounds [10]. Dietary phytochemicals and repositioned drugs should provide easier access towards clinical use, because of the former's “Generally Regarded as Safe” (GRAS) status and the latter's prior Food and Drug Administration (FDA) approval. This innovative approach is advantageous: both compounds target CSCs, an activity insurmountable by current anti-cancer agents.
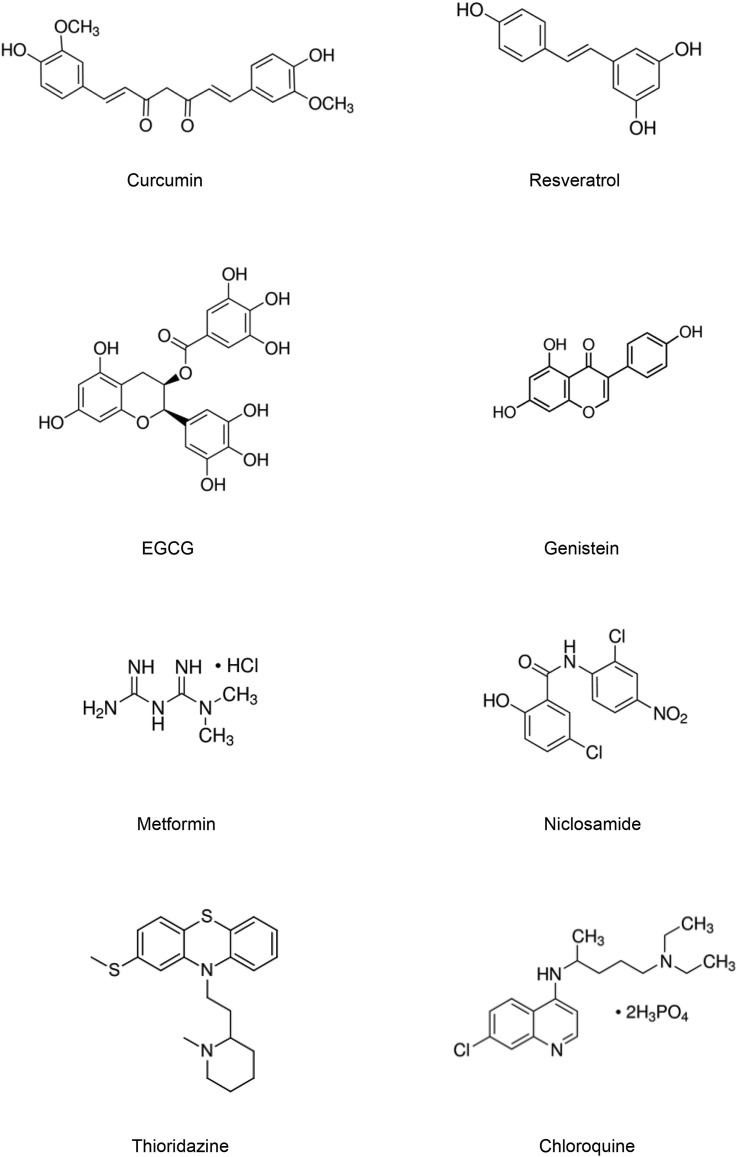
For CSC targeting, we recommend four dietary phytochemicals (curcumin, resveratrol, EGCG, genistein) and four repurposed drugs (metformin, niclosamide, thioridazine, chloroquine). (See Fig. 1 for molecular structure.) These eight are selected based on literature survey and our own experience. In our view they are the most promising for applications of CSC-targeting phytochemical-drug combinations. Furthermore, five (curcumin, resveratrol, EGCG, genistein, metformin) are listed by an international task force of 180 clinicians and scientists of “The Halifax Project”, with dedicated objective to “explore the concept of a low-toxicity ‘broad-spectrum’ therapeutic approach that could simultaneously target many key pathways and mechanisms” for cancer treatment [1]. Block et al. (2015) stated: “among approaches, curcumin, genistein, resveratrol and EGCG boast a wealth of fundamental research” [1]. (We are aware investigators may disagree with our selection of phytochemicals; others will also be cited.) All eight have efficacy, are low cost and free of intellectual property constraints. These attributes make sure the combinations can be widely affordable, and are economical compared to current chemo- and targeted therapies.
Fig. 1.
Chemical structures of selected phytochemicals and drugs.
Different from conventional anti-cancer drugs with unique molecular targets, dietary phytochemicals and repurposed drugs are pleiotropic. Each has a multitude of cellular targets (as shown later in compound description). Multilateral targeting of molecular and cellular pathways will inhibit CSC growth or induce CSC differentiation, and hinder the CSC's ability to develop resistance. In this case, “promiscuous drugs” is a virtue; “dirty” might be better [11,12]. A phytochemical-drug combination is advantageous for synergistic effects. Such pairing allows lower effective doses than using a single compound for activity. Lower doses mean less potential toxicity and fewer side effects. For a particular cancer, various combinations can be explored to look for the ideal pair at optimal doses. For clinical use, the combination can be used alone (if both CSCs and bulk cancer cells are eliminated), or as adjuvant to conventional cancer therapy (the combination targets CSCs and conventional therapy eliminates the bulk cancer cells).
If the approach of CSC-targeting dietary phytochemical-repositioned drug combination is important, what are the reasons for its being overlooked? The gap in knowledge exists because cancer investigators in different areas may not be familiar with contributions of each other. Phytochemicals for cancer chemoprevention and therapy have a long history; current interests extend to CSCs. On the other hand, drug repurposing, especially regarding CSC-targeting, is new. Pairings of dietary phytochemicals or repurposed drugs with cancer chemotherapeutics have been around, but we propose the pairing of CSC-targeting dietary phytochemicals and repurposed drugs. This novel combination is an innovative concept worthy of promotion.
To present a more comprehensive picture, we start with considerations to CSC methodologies on which various observations were made, in the “evolving concept” of CSC [13,14]. We follow with compound description, and end with challenges and the need of applying CSC-targeting phytochemical-drug combinations in the current climate of cancer therapy.
2. Identification and drug resistance of cancer stem cells
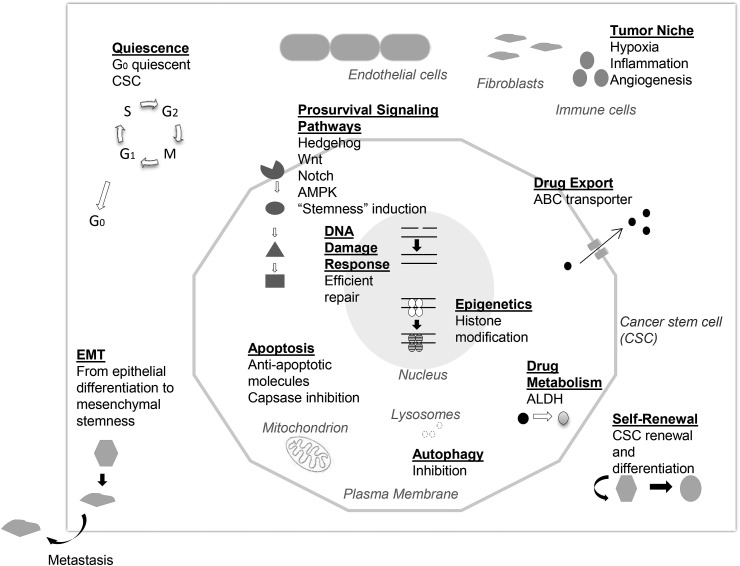
Human CSCs were first discovered in leukemia and then characterized in solid tumors. Utilizing an array of mechanisms, CSCs are insensitive to conventional cancer treatments, but these mechanisms can be targets of phytochemicals and drugs (as described later) [[15], [16], [17], [18], [19]]. (See Fig. 2 for key mechanisms of CSC resistance to therapy.) CSCs may be quiescent and exhibit a slow proliferating nature. They may possess an efficient DNA repair system, activated by genomic instability and regulated by epigenetic modifications via histone deacetylases. Pro-survival signaling pathways may be activated (Hedgehog, Wnt, Notch, PI3K, NFκB). CSCs may have high levels of anti-apoptotic molecules. For drug resistance, both the activation of drug transporters, with high level expression of ATP-binding cassette (ABC) membrane transporters for drug efflux, and the enzyme aldehyde dehydrogenase (ALDH), for drug metabolic activities, have been detected. In addition, their niche has less reactive oxygen species (ROS), thus less susceptible to radiation therapy.
Fig. 2.
Key mechanisms of cancer stem cell resistance to therapy.
One notable CSC-associated feature is epithelial-mesenchymal transition (EMT), a process when epithelial cells lose polarity and become less adhesive [20]. With these changes the cells become invasive, migratory and mesenchymal-like with “stemness” properties. The process involves the activation of transcription factors (Snail, Twist) and change in marker proteins (E-to N-cadherin). First seen in embryogenesis, EMT has been viewed as reappearance of embryonic features in cancer cells. The process is reversible and has been linked to CSCs for tumor heterogeneity and metastasis. EMT involvement with CSCs is under active research [21].
CSCs can be identified by in vitro and in vivo methods. They are isolated via fluorescence-activated cell sorting (FACS) by their surface antigens or detoxification capacity. CSCs express identifying cell surface antigens, for example, ovarian CSCs express CD133 (prominin-1); these molecules can serve as biomarkers allowing their isolation by FACS. ALDH is a detoxification enzyme that oxidizes aldehydes to carboxylic acids, for further metabolism or excretion via the liver. Its activity has been utilized for CSC selection by FACS, using a fluorescent substrate (ALDEFLUOR) [22]. The “side population” (SP) assay identifies CSCs apart from the main population of cancer cells in flow cytometry, by their fast expulsion of a fluorescent dye (Hoechst 33342). However, cell surface markers for CSC selection are also present in normal stem cells and other cell types, they are not unique to CSCs [6]. Both SP and ALDH selections were originally developed for hematopoietic stem cells (HSCs) and then adapted to CSCs.
Spheroid culture is another method for identifying CSCs. A cancer cell (transformed due to mutations) exhibits anchorage independence, a characteristic allowing it to form a cell colony in the absence of substratum, as shown by soft agar colony formation assay (normal cells die in the absence of attachment via the process of anoikis). For serum-free spheroid cultures (spheres), cancer cells grow as spheroids in suspension (in untreated plastic ware) when provided with specific growth factors (insulin, epidermal growth factor, basic fibroblast growth factor), unlike ordinary tissue culture where cells usually grow as a monolayer (with fetal bovine serum for growth factors and other components). Spheroids mimic the three-dimensional nature of tumors with limited oxygen available to the internal cells. This hypoxic condition favors expression of pluripotency maintaining transcription factors, thus promoting the differentiation to CSCs and facilitating the state of “stemness” [6]. Spheroids can form secondary and tertiary spheres in subsequent cultures. CSCs known as “neoplastic sphere-forming cells” are measured by enumerating secondary spheres generated when re-plating the spheres, based on the assumption that the secondary spheres are clones [23]. The technique of spheroid culture was originally developed for neurobiology and then adapted to CSCs [24].
Apart from in vitro methods, the “gold standard” for CSC is “neoplasia-initiating cells” that regenerate detectable neoplastic populations as xenografts in immunodeficient mice in vivo [23]. CSCs, isolated from either a fresh tumor or an established cell line, are transplanted to initiate tumor growth in immunocompromised recipient animals. Different models are available: athymic nude mice, non-obese diabetic/severe combined immunodeficiency (NOD/SCID) mice, and NOD/SCID interleukin-2 receptor gamma chain null (Il2rg −/−) (NSG) mice. The abundance of CSCs in a tumor sample can be estimated as the xenotransplantation yield, which confirms the presence of CSCs by limiting dilution analysis [6].
Many compounds (natural, natural-derived, synthetic) have been reported as CSC-targeting [19,25,26]. Two strategies are available for compound identification: a pathway-specific approach and a general high throughput, mass screening approach. The first yielded cyclopamine from the plant corn lily Veratrum californicum; it targets glioblastoma CSCs via the Hedgehog pathway but can act as a teratogen that causes cyclopia. The second yielded salinomycin from the bacterium Streptomyces albus; it is toxic to humans so its use is confined to the poultry industry, as a coccidiostat for treating a parasitic protozoan disease [6]. For clinical uses, medicinal chemists must modify to remove teratogenicity or toxicity. Identified compounds must target only CSCs and not normal stem cells. Even though CSCs differ from the bulk cancer cells, they may share gene expression and signaling pathways with normal stem cells. CSC-targeting phytochemicals and drugs are discussed in the following sections.
3. Targeting of cancer stem cells with dietary phytochemicals
Dietary phytochemicals have advantages over other compounds [27]: (1) they usually have very low or no toxicity, in contrast to most chemotherapeutic drugs; (2) they are present in commonly consumed food that is readily available to most people in daily life; (3) they have shown potential as adjuvants to chemotherapy. Originally, they were studied for cancer chemo-prevention [28]. As initiation (chemoprevention) and growth (cancer chemotherapy) may share common molecular mechanisms, they were applied to cancer therapy. Furthermore, phytochemicals can overcome drug resistance in ovarian and other cancers [29]. They act on genes and non-coding gene regulatory pathways (microRNA) [30]. Most importantly, phytochemicals target CSCs [[31], [32], [33], [34], [35], [36]]. Commenting on CSCs and dietary phytochemicals, Kim et al. (2012) wrote: “a diet-induced shift from deregulation to regulation in cancer stem cells could have profound influence on cancer relapses and therefore is of immense societal importance” [36]. Instead of diet, here we propose the use of CSC-targeting phytochemical-drug combinations for cancer therapy.
Our laboratories have been studying the phytochemicals curcumin, quercetin and EGCG. These polyphenols act synergistically with cisplatin (conventional anti-cancer drug) in growth inhibition of drug-resistant ovarian cancer cell lines [8,9]. These compounds are active against CSCs in vitro, as well as against cancer and other diseases in animal models. Some are in clinical trials, though to date none has been approved for cancer treatment. Here, we discuss four CSC-targeting phytochemicals: curcumin, resveratrol, EGCG and genistein. (See Table 1 for select cancer clinical trials of the four phytochemicals, from https://clinicaltrials.gov/.)
Table 1.
Sample clinical trials of selected phytochemicals.
| Cancer Type | Identifier no. | Year | No. | Trial title | Intervention | Phase |
|---|---|---|---|---|---|---|
| Breast Cancer | NCT00852332 | 2009 | 100 | Docetaxel With or Without a Phytochemical in Treating Patients With Breast Cancer | Curcumin Taxotere | II |
| Colorectal Cancer | NCT01333917 | 2010 | 40 | Curcumin Biomarkers | Curcumin C3 | I |
| Colonic cancer | NCT01490996 | 2012 | 51 | Combining Curcumin With FOLFOX Chemotherapy in Patients With Inoperable Colorectal Cancer (CUFOX) | Curcumin C3 Chemotherapy |
I/II |
| Prostate cancer | NCT02095717 | 2014 | 50 | Multicenter Study Comparing Taxotere Plus Curcumin Versus Taxotere Plus Placebo Combination in First-line Treatment of Prostate Cancer Metastatic Castration Resistant (CURTAXEL) | Curcumin Taxotere | II |
| Prostate Cancer | NCT02064673 | 2014 | 600 | Adjuvant Curcumin to Assess Recurrence Free Survival in Patients Who Have Had a Radical Prostatectomy | Curcumin | III |
| Colorectal Cancer | NCT02439385 | 2015 | 44 | Avastin/FOLFIRI in Combination With Curcumin in Colorectal Cancer Patients With Unresectable Metastasis | Curcumin Avastin/FOLFIRI | II |
| Neoplasms | NCT02944578 | 2017 | 40 | Topical Curcumin for Precancer Cervical Lesions | Curcumin | II |
| Solid Tumor | NCT00098969 | 2004 | 40 | UMCC 2003-064 Resveratrol in Preventing Cancer in Healthy Participants (IRB, 2004–535) | Resveratrol | I |
| Colon Cancer | NCT00256334 | 2005 | 11 | Resveratrol for Patients With Colon Cancer | Resveratrol | I |
| Adenocarcinoma of the Colon | NCT00433576 | 2006 | 20 | Resveratrol in Treating Patients With Colorectal Cancer That Can Be Removed By Surgery | Resveratrol | I |
| Breast Cancer | NCT00676793 | 2004 | 32 | A Study of the Effect of Polyphenon E (Green Tea Extract) on Breast Cancer Progression | Polyphenon E | II |
| Cervical Cancer | NCT00303823 | 2005 | 98 | Green Tea Extract in Preventing Cervical Cancer in Patients With Human Papillomavirus and Low-Grade Cervical Intraepithelial Neoplasia | Tea catechin extract | II |
| Barrett Esophagus | NCT00233935 | 2005 | 55 | Defined Green Tea Catechin Extract in Preventing Esophageal Cancer in Patients with Barrett's Esophagus | Catechin Extract | I |
| Carcinoma | NCT02029352 | 2014 | 42 | Topical Green Tea Ointment in Treatment of Superficial Skin Cancer | Sinecatechins 10% | II/III |
| Lung Neoplasms | NCT02577393 | 2015 | 53 | Study of Epigallocatechin-3-gallate (EGCG) for Esophagus Protection in Patients With Lung Cancer Receiving Radial Radiotherapy | EGCG | II |
| Colon Cancer | NCT02891538 | 2017 | 50 | Chemopreventive Effects of Epigallocatechin Gallate (EGCG) in Colorectal Cancer (CRC) Patients | EGCG | I |
| Bladder Cancer | NCT00118040 | 2005 | 60 | Phase II Study of Isoflavone G-2535 (Genistein) in Patients With Bladder Cancer | Genistein | II |
| Breast Cancer | NCT00290758 | 2006 | 126 | Genistein in Preventing Breast Cancer in Women at High Risk for Breast Cancer | Genistein | II |
| Prostate Cancer | NCT01036321 | 2009 | 71 | Phase II Clinical Trial of Purified Isoflavones in Prostate Cancer: Comparing Safety, Effectiveness | Purified isoflavones | II |
| Non-small cell lung cancer | NCT01628471 | 2012 | 20 | MTD Determination, Safety and Efficacy of the Decitabine-Genistein Drug Combination in Advanced Solid Tumors and Non-Small Cell Lung Cancer | Genistein Decitabine | I/II |
| Carcinoma | NCT02567799 | 2015 | 36 | BIO 300 Non-Small Cell Lung Cancer Study (NSCLC) | BIO300 Paclitaxel Carboplatin Radiotherapy | I/II |
From https://clinicaltrials.gov/including year started and number of participants.
3.1. Curcumin
Curcumin, a diferuloylmethane, is the biological principle from the Indian spice turmeric that gives curry powder the yellow color. Turmeric is produced from rhizome of the plant Curcuma longa; it has multiple utilities and health benefits in traditional Indian medicine (from insect bites to wound healing). A widely studied property of curcumin is anti-inflammation. Curcumin inhibits many pro-inflammatory gene products, including enzymes (inducible nitric oxide synthase, iNOS), transcription factors (nuclear factor kappa B, NFκB), cytokines (tumor necrosis factor, TNFα) and chemokines (interleukin-8, IL-8) [6]. Using the lipopolysaccharide (LPS)-induced murine sepsis model, we reported curcumin ingestion by gavage down-regulated iNOS gene expression in the murine liver [37]. Curcumin may suppresses carcinogenesis by down-regulating inflammation, which supports cancer cell survival, proliferation and invasion [2].
Another widely studied anti-cancer property of curcumin is CSC-targeting [[38], [39], [40], [41], [42]]. (See Table 2 for select cellular targets of phytochemicals and drugs in CSCs.) CSCs that resist conventional anti-cancer drugs are susceptible to curcumin. Curcumin acts on stem cell signaling pathways implicated in the process of carcinogenesis, including Wnt, Notch, Hedgehog, and signal transduction and activator (STAT). For example, in hepatocellular carcinoma CSCs, Tsai et al. (2015) reported that curcumin inhibited SP, invasion, EMT and reduced tumor size and lung metastasis in a nude mice xenograft model. Immunoblots revealed that the sphingosine 1-phosphate receptor 3 (SIPR3) signaling pathway was inhibited [43]. In cell culture, Subramaniam et al. (2012) reported that curcumin-induced apoptosis of esophageal cancer cells and decreased esophageal CSC spheroid size and number. The molecular mechanism was inhibition of the Notch pathway, seen as a decrease in RNA and protein expression of γ secretase, Notch-1 protein and its ligand Jaggard-1 [44]. The investigators also found down-regulated oncomir miRNA (miR-21, miR-34a) and up-regulated tumor suppressor miRNA (let-7a miRNA) in the curcumin-treated esophageal CSC spheroids. Similar to the esophageal example, for colorectal CSCs, Ramasamy et al. (2015) reported curcumin-induced epigenetic modifications, including the methylation of epidermal growth factor receptor (EGFR) promoter, expression of microRNA oncomirs relevant to metastasis (for EMT), as well as suppression of CSC markers (CD133, ALDH+) [42]. Thus, curcumin modifies epigenetically by inducing specific methylation changes and regulating microRNA expression. Huminiecki et al. (2017) have reviewed the functional genomic studies of curcumin from microarray, methylation array, microRNA array to RNA-seq and concluded that curcumin has “powerful effects” on gene expression, including “genes involved in cell signaling, apoptosis, and the control of cell cycle” [45].
Table 2.
Select cellular targets for dietary phytochemicals and repositioned drugs in cancer stem cells (CSCs).
| Phytochemicals |
Drugs |
|||||||
|---|---|---|---|---|---|---|---|---|
| Curcumin | Resveratrol | EGCG | Genistein | Metformin | Niclosamide | Thioridazine | Chloroquine | |
| Signaling pathway | ||||||||
| Hedgehog | + | + | + | + | ||||
| Wnt | + | + | + | + | + | |||
| Notch | + | + | + | + | + | |||
| Kinases | + | + | + | + | + | + | + | |
| Inflammation | ||||||||
| NFκB | + | + | + | + | + | + | ||
| IL-6 | + | + | ||||||
| STAT 3 | + | + | + | + | + | + | ||
| Epigenetics | ||||||||
| Micro RNA | + | + | + | |||||
| SIRTUIN | + | + | ||||||
| Drug resistance | ||||||||
| Efflux | + | + | + | + | ||||
| ALDH | + | + | ||||||
| Metabolism | ||||||||
| AMPK | + | + | + | + | ||||
| ROS | + | + | + | + | ||||
| No Anchorage | ||||||||
| Spheroids | + | + | + | + | + | + | + | + |
| EMT | + | + | + | + | + | |||
| Metastasis | + | + | + | |||||
| Apoptosis | + | + | + | + | + | + | + | |
| Autophagy | + | + | + | + | + | |||
| Differentiation | + | + | + | + | + | |||
| Drug Synergy | + | + | + | + | + | + | + | |
Curcumin can induce CSC differentiation [41]. Zhuang et al. (2012) reported that curcumin promoted differentiation of glioma CSCs and induced autophagy, using immunofluorescence to show decrease in stemness markers (CD133) and increase in differentiation markers (βIII tubulin) [46].
Curcumin can alter chemo-sensitivity. It synergizes with conventional drugs and has specificity for CSCs. In vitro, curcumin enhanced cisplatin effects on non-small cell lung CSC [47]. We found that curcumin (5 μM daily for 10 days via medium change) reduced SP of the C6 rat glioma cells, possibly via inhibition of the drug transporter [48]. In vivo, curcumin enhanced paclitaxel effects on brain tumor CSCs; it sensitized breast CSCs to mitomycin C in a nude mice xenograft model [49].
Several reasons have been proposed to explain the CSC-specific action and low adversity towards normal stem cells [41]. Curcumin uptake may be greater in CSCs, and curcumin may target the CSC microenvironment which obviously differs from those of normal stem cells. For example, Buhrmann et al. (2014) reported that curcumin sensitized colorectal CSCs towards fluorouracil (5-FU) and suppressed the crosstalk between CSCs and stromal fibroblasts in the tumor niche, using co-culture and assaying EMT markers by immunoblotting [50].
3.2. Resveratrol
Resveratrol is a stilbene (3,4′,5-tri-hydroxy-trans-stilbene) from grapes, peanuts and pine nuts, and is present in red, but not white, wine because it is associated with the skin of grapes. It is a pleiotropic agent with multiple cellular targets. For example, it inhibits the mammalian target of rapamycin (mTOR) pathways and cyclooxygenase (COX) enzymes to play roles in inflammatory diseases, diabetes, obesity, cardiovascular diseases, neurological disorders and cancer [[51], [52], [53]]. Resveratrol also activates AMP-activated protein kinase (AMPK, a key nutrient sensor) and sirtuin deacetylases, leading to lifespan extension in yeasts, nematodes, fruit flies, fish and obese mice (but not normal mice) [54]. It retards the ageing process in mammals, similar to caloric restriction. Furthermore, resveratrol is a phytoalexin (compound produced by plants against pathogens, such as fungal infections to the grape plant) and we found this property can be harnessed for inhibiting the growth of human cutaneous fungal species, thus potentially useful for tinea (ringworm, athlete's foot) [55].
In cancer, resveratrol can target CSCs. Park and Pezzuto (2015) have reviewed the molecular alterations resulting from resveratrol intervention in different breast, prostate, lung and colorectal cancer models [51]. The general approach in these studies is to isolate the CSCs, assay the effects of resveratrol in vitro and in vivo (comparing CSCs with the bulk cancer cells) and determine which molecular mechanisms are modulated. For example, Fu et al. (2014) reported resveratrol cytotoxicity to breast CSCs in vitro [56]. Mechanistically, immunofluorescence and immunoblot of microtubule-associated protein light chain 3 (LC3) and β catenin proteins showed that it induced autophagy and suppressed the Wnt pathway. Resveratrol also inhibited spheroid formation, decreased ALDH+ cells and decreased tumor size in murine xenografts. Shankar et al. (2011) reported resveratrol inhibited pancreatic CSC spheroid formation via the induction of apoptosis, inhibition of mRNA expression of the pluripotency maintenance transcription factors (Oct 4, Nanog), as well as interference with EMT [57]. Similar inhibition was also observed in a transgenic mouse model of pancreatic ductal adenocarcinoma. With glioblastoma multiforme, Yang et al. (2012) found that CSCs pre-treated with resveratrol in vitro were less tumorigenic, as the xenografted SCID mice survive better than the untreated counterpart [58]. The investigators also performed microarray analyses and shRNA confirmation to compare the effects of resveratrol on CSCs (CD133+) and non-CSCs (CD133-). Resveratrol inhibited “stemness” gene expression and induced CSC differentiation via suppression of the STAT3 pathway.
Resveratrol, like curcumin, can be CSC-specific and spare the normal stem cells. Pandey et al. (2011) reported that resveratrol blocked gene expression of fatty acid synthase and inhibited breast CSC growth, but the enzyme in normal human mammary epithelial cells was not susceptible [59]. Fu et al. (2014) reported resveratrol was cytotoxic to breast CSCs but not normal breast epithelial cells [56]. Similarly, Sayd et al. (2014) reported that resveratrol blocked enzymatic induction of sirtuin activity and inhibited the growth of glioma CSCs, but had no effect on normal neural stem cells (from fetal brains) [60].
3.3. EGCG
Epigallocatechin gallate (EGCG), a polyphenol (flavone-3-ol) from the plant Camellia sinensis, is the major component in green tea. Its many health benefits include cancer chemoprevention and treatment, reduction of atherosclerosis, hypercholesterolemia, Alzheimer's and other ageing-related diseases. Before CSCs, EGCG was first studied for its effect on normal stem cells. Chen et al. (2003) reported tea components, including EGCG, inhibited normal rat neurosphere adhesion and migration [61]. EGCG has pleiotropic activity on multiple cellular targets, both genetic and epigenetic (including DNA methyltransferase, NFκB, AMPK signaling pathway) [[62], [63], [64]]. It can act on its target with very impressive sensitivity, KD of 39.9 nM for the 67 Kd laminin receptor [65].
In cancer, EGCG can target CSCs. It inhibited ALDH activity, spheroid formation, and expression of “stemness” genes (Oct 4 and Nanog) in human neuroblastoma CSCs [66]. Similar results were observed in breast CSCs. EGCG treatment significantly decreased the expression of Nanog, the number of ALDH+ cells, and the tumor volume in mouse xenografts [67]. Furthermore, Chen et al. (2017) reported EGCG inhibited the spheroid formation of colorectal CSCs, the mechanism being inhibition of the Wnt pathway, as shown by a decrease in β catenin in immunoblot [68].
EGCG works in combination with other phytochemicals and conventional drugs to enhance anti-cancer effects, targeting bulk cancer cells and CSCs [69]. It synergized with quercetin to decrease spheroids, inhibited activation of EMT and increased apoptosis in prostate CSCs [70]. It synergized with temozolomide to decrease spheroids and P-glycoprotein synthesis in glioma CSCs [71]. We found that EGCG acted synergistically with cisplatin to inhibit the cisplatin-resistant ovarian cancer cell line C200 in cell culture [9]. In vivo, the EGCG and cisplatin combination decreased tumor formation in xenografts of head and neck, and nasopharyngeal CSCs [72,73]. It induced chemo-sensitivity to cisplatin by enhancing apoptosis and inhibiting the phosphorylation of STAT3 in nasopharyngeal CSCs [74]. These findings suggest its use as an adjuvant in cancer therapy [75].
3.4. Genistein
Genistein, an isoflavone (4,5,7-trihydroxyisoflavone) from the legume plant Glycine max, is found in soybeans. It has been classified as a phytoestrogen because it binds estrogen receptor. Genistein is the active ingredient in soy-rich food that contributes to the lower rates of prostate and breast cancers in China and Japan, as compared to Western countries. Besides as anti-cancer agent, genistein has other health benefits, including osteoporosis, heart diseases and cognition [[76], [77], [78], [79]].
Genistein has multiple cellular targets and acts on a spectrum of protein tyrosine kinases and DNA topoisomerase II. It targets CSCs in solid tumors. Huang et al. (2014) reported genistein inhibited spheroids, “stemness” (Oct 4 and Nanog gene expression) and reduced xenograft tumor volume of gastric CSCs [80]. Genistein also inhibited drug transporter and extracellular signal-regulated kinase (ERK) pathway in these CSCs. In both breast and prostate CSCs, genistein down-regulated the Hedgehog pathway (reducing Gli 1 gene expression) and decreased spheroid formation in cell culture and tumor volume in xenografts [81,82]. In chronic myelogenous leukemia (CML), genistein reduced the leukemic progenitor cells by inhibiting expression of the tyrosine kinase coded from the breakpoint cluster region/Abelson murine leukemia viral oncogene homolog (BCR/ABL) fusion gene [83]. However, as a protein tyrosine kinase inhibitor, genistein targets both leukemic stem cells and normal stem cells.
Genistein acts synergistically with conventional anti-cancer agents; it also targets the CSC microenvironment. It overcame docetaxel resistance, and decreased the tumor size of docetaxel-resistant prostate CSCs (than either compound administered separately) [82]. Breast adipose tissue contributes to breast cancer development. Montales et al. (2013) found that genistein acted on the CSC niche and inhibited the differentiation of mammary stromal fibroblast-like cells to adipocytes [84]. The mechanism was inhibition of PPARγ and fatty acid synthase gene expression. Interestingly, the investigators discovered genistein's biphasic effect, only seen in low dose (40 nM) but not high dose (2 μM). Montales et al. (2012) also found the effect of genistein to be transferrable [85]. When fed to mice at concentrations present in soy protein isolate (250 mg/kg food), their sera could inhibit the spheroid formation of human breast CSCs when added at 1–5% in cell culture. This suggests the presence of “CSC inhibiting factors” in the circulation after genistein consumption. Furthermore, genistein has transgenerational effect. Maternal dietary supplementation with genistein leads to DNA hyper-methylation in the embryo, and this methylation state is maintained until adulthood. The color change in the fur of genistein-fed agouti mice indicates an epigenetic biosensor for nutritional and environmental alterations on the fetal genome. Phytochemicals such as genistein modify the epigenome and the effect starts early in embryogenesis [86].
4. Targeting of cancer stem cells with repositioned drugs
FDA-approved small-molecule drugs for some diseases/disorders have been shown to target CSCs by the process of drug repurposing. The NIH National Center for Advancing Translational Sciences (NCATS) defines: “Repurposing generally refers to studying drugs that are already approved to treat one disease or condition to see if they are safe and effective for treating other diseases”. The rationale is to expedite drug discovery. NCAT estimates 14 years for a new drug development. Therefore, the strategy is “to reduce this time frame, decrease costs and improve success rates …. Many agents approved for other uses already have been tested in humans, so detailed information is available on their pharmacology, formulation and potential toxicity. Because repurposing builds upon previous research and development efforts, new candidate therapies could be ready for clinical trials quickly …” (https://ncats.nih.gov/preclinical/repurpose#learn-more). Drug repurposing, also known as drug repositioning, drug re-tasking, drug re-profiling or therapeutic switching, is a way to obtain fast and cost-efficient drug discovery, since this strategy effectively enables the preclinical studies to be bypassed. Resources (DRUGSURV, Drug Repurposing Hub) are available for this “new tricks for old drugs” [7,[87], [88], [89]]. Investigators can perform experimentation in silico, in vitro or in vivo in search for new targets of existing drugs. For CSCs, drug libraries are screened in assays of CSC inhibition, as shown below for niclosamide and thioridazine. Here, we discuss four CSC-targeting drugs: metformin, niclosamide, thioridazine and chloroquine.
4.1. Metformin
Probably the most promising discovery is metformin, a synthetic biguanide derived from the herb French lily (Galega officianalis) [90]. It is an inexpensive and long-approved drug for type 2 diabetes, prescribed to an estimated 150 million individuals worldwide. It modulates energy metabolism by targeting AMPK and other AMPK-independent effects. Besides diabetes, metformin has been shown to be applicable to cardiovascular and neurodegenerative diseases [[91], [92], [93], [94], [95]]. It is in a clinical trial for longevity and ageing [96].
Metformin was explored for CSC-targeting because it targets multiple signaling pathways (Wnt, AMPK, NFκB) [93,95]. Metformin has been used in combination therapy. For example, it synergized with the conventional combination of 5-fluorouracil (5-FU), epirubicin and cyclophosphamide (FEC chemotherapy) in inhibiting breast CSCs derived from spheroids [97]. It enhanced the effect of denosumab, an antibody against RANKL (receptor activator of NFκB ligand, a cytokine), in inhibiting breast CSC spheroids [98]. It enhanced the sensitivity of pancreatic CSCs to gemcitabine in vivo, significantly reducing the tumor volume in murine xenografts [99]. When given in combination, metformin and doxorubicin showed the advantage of targeting both breast CSCs and non-stem cancer cells, by reducing the overall tumor growth and tumor remission in nude mice [100]. Synergy was found with paclitaxel and carboplatin in prostate and lung CSCs murine xenografts [101]. For phytochemicals, Montales et al. (2015) reported that metformin and genistein targeted colon CSC spheroid formation and proliferation, and the combination increased the effectiveness of 5-FU [102]. Ning et al. (2016) reported that both curcumin and metformin individually inhibited pancreatic CSC spheroids [103], the two compounds should be tested as a combination. These examples suggest a combinational strategy for metformin to target CSCs and cancers [104].
4.2. Niclosamide
The next most promising existing drug is niclosamide, an anthelminthic against tapeworms [105]. It is also active against viruses (severe acute respiratory syndrome, SARS virus), possibly because it blocks proton carriers. Niclosamide was explored for CSC-targeting based on the general association that a developmental stage of tapeworm, the metacestode, resembles cancer in having uncontrolled proliferation, invasion and metastasis, and can be difficult to kill without collateral damage to the surrounding host tissues [106].
Niclosamide can block multiple CSC signaling pathways, including Wnt, Notch, STAT3 and NFκB [107]. Wieland et al. (2013) selected niclosamide as the lead for glioblastoma targeting after screening the 160 member Killer Plate compound library [108]. Simultaneous inhibition of multiple pathways (Wnt, Notch, mTOR, NFκB) was indicated when niclosamide pretreatment inhibited glioblastoma xenograft development in nude mice. From 1258 drugs in the LOPAC chemical library, Wang et al. (2013) [109] and Yo et al. (2012) [110] selected niclosamide for its inhibition of breast and ovarian CSCs, respectively. Niclosamide inhibited spheroid formation and reduced tumor volume of xenografts in NOD/SCID mice. It acted on cisplatin-resistant ovarian CSCs. Spheroids from patient-derived CD133+ and ALDH+ ovarian cancer were susceptible to niclosamide, with inhibition of the Wnt pathway (shown by β catenin immunoblot) [111]. Furthermore, it complements platinum drugs. Niclosamide in combination with carboplatin inhibited primary ovarian CSCs [111] and in combination with cisplatin inhibited breast CSC spheroid formation and reduced tumor size of xenografts in nude mice [112].
4.3. Thioridazine
Thioridazine, a phenothiazine, is dopamine receptor antagonist originally prescribed as an anti-psychotic drug for schizophrenia in the 1960s. Withdrawn by Norvatis in 2005 due to cardiac side effects, the generic version is still available. Interest in thioridazine has recently been revived as anti-microbial against methicillin-resistant Staphylococcus aureus (MRSA) and multidrug-resistant Mycobacterium tuberculosis [113].
Thioridazine targets CSCs. It can inhibit CSCs from a spectrum of origins: myeloid leukemia, glioblastoma, lung, liver, ovarian and breast cancers [[114], [115], [116], [117], [118]]. It inhibits CSC spheroid formation and induces apoptosis in vitro, and the treated cells show reduced xenograft tumor volume in mice. Sachlos et al. (2012) selected thioridazine in their screening for acute myeloid leukemia (AML) CSC-specific but not normal pluripotent stem cell-reactive molecules from 590 compounds of the NIH Clinical Collection and Canadian Collection [119]. Thioridazine was found to induce AML differentiation; it also synergized with cytosine arabinoside (cytarabine, AraC) in AML CSC killing. In glioblastoma CSCs, it induced autophagy (seen as expression of biomarker LC3) [114]. In lung, hepatoma and breast CSCs, it induced apoptosis in association with activation of caspases, inhibition of “stemness” gene expression (Oct4, CD133), and inhibition of the mTOR pathway, respectively [[115], [116], [117]]. Synergy with conventional drug has been shown: thioridazine was co-delivered with doxorubicin in mixed polymeric micelles to target both breast CSCs and bulk cancer cells [118]. We found that thioridazine and curcumin may act synergistically in inhibiting spheroids from ovarian cancer cells, with the combination more effective than either compound alone. (See Fig. 3 for inhibition of spheroids by thioridazine and curcumin).
Fig. 3.
Inhibition of growth of SKOV3 spheroids in the presence of thioridazine and curcumin at the indicated concentrations after 5 days. Combination was more effective than either alone.
4.4. Chloroquine
Chloroquine, a 4-aminoquinoline, is an anti-malarial [120]. First discovered in 1936 b y Hans Andersag at Bayer, it was ignored until rediscovery by US Army during World War II. Highly effective and well tolerated, chloroquine remains the drug of choice for malaria. Its accumulation within lysosomes raises organelles' pH and inhibits lysosomal function in the malaria parasite. Chloroquine is also used for immunosuppression in rheumatoid arthritis.
Chloroquine, either independently or in combination with conventional drugs, has efficacy against several cancers. It inhibits CSCs from a spectrum of origins: glioma, liver, pancreatic, urothelial, ovarian and breast cancer [[121], [122], [123], [124], [125], [126], [127], [128]]. It inhibits autophagy and spheroid formation; it induces CSC apoptosis. These effects have been demonstrated in primary glioma CSCs, CD133+ liver CSCs enriched by deprivation of oxygen and nutrients, pancreatic CSCs, urothelial carcinoma CSCs, and breast CSCs.
The CSC-targeting chloroquine shows synergistic effects with conventional therapy [127]. It sensitizes radiation and chemotherapeutic agents in cancer [91,120]. For example, chloroquine increased the cytotoxicity of gemcitabine-mitomycin combination against urothelial carcinoma CSCs [124]. It synergized with gemcitabine to decrease tumor volume in pancreatic CSC xenografts [123], and with carboplatin in breast CSC xenografts [126].
5. Additional CSC-targeting phytochemicals and repurposed drugs
We discuss four phytochemicals and four drugs that target CSCs, and view them as the most promising for a novel approach of phytochemical-drug combination. There are others.
Exploring CSC-targeting phytochemicals is a growing field. Other dietary phytochemicals include quercetin, a flavonol found in common fruits and vegetables such as apples, cranberries and onions; sulforaphane, an isothiocyanate found in cruciferous such as broccoli, cauliflower and kale; indole-3-carbinol, another compound from cruciferous vegetables; and cucurbitacin I, from cucumber. Non-dietary phytochemicals include parthenolide from herbal medicine feverfew plant (Tanacetum parthenium), celastrol from traditional Chinese medicine thunder god vine (Tripterygium wilfordii), berberine from herbal medical plant Chinese goldthread (Coptis chinensis), oxymatrine from Chinese herbal medical plant Sophora flavescens, silybin (silibinin) from milk thistle (Silybum marianum), and gossypol from cotton plant (genus Gossypium) [6].
Similarly, the list of CSC-targeting existing drugs is expanding. Other repurposed drugs include anti-allergy drug tranilast, cholesterol-lowering statins (simvastatin and lovastatin), and anti-alcoholism drug disulfiram [6,19,25,128,129]. We anticipate more to come.
6. Challenges on CSC-targeting phytochemical-drug combinations
For challenges we start with the safety issue. With GRAS status dietary phytochemicals are apparently safe. For example, when curcumin was given to cancer patients at 3600 mg/day for 4 months, only minor adverse effects were observed. Daily oral doses of EGCG, for 4 weeks at 800 mg/day in 40 volunteers, caused only minor adverse effects. A single oral dose of resveratrol at 5 g in 10 volunteers caused only minor adverse effects [6]. With prior FDA approval, repurposed drugs are also presumably safe. Although individual compounds are safe, for the selected synergistic CSC-targeting phytochemical-drug combination, pharmacokinetics and pharmacodynamics features, toxicity profiles, are still needed because this information is lacking. Current clinical trials may yield useful data. (See Table 1 for phytochemical clinical trials.)
Molecular targets of phytochemicals are under scrutiny. Recent findings suggest that promiscuity (multiple targets) results from perturbation of cellular membranes, leading to alteration of multiple membrane protein functions [130]. This multi-target nature of polyphenolic phytochemicals hamper therapeutic development because, in high throughput drug screening assays, phytochemicals (including curcumin, resveratrol, EGCG and genistein) are known as “pan-assay interference compounds” (PAINS) and “invalid metabolic panaceas” (IMPs) for their non-specific inhibitions [[131], [132], [133]]. Whereas there may be truth to the designation, the fact that phytochemicals (such as curcumin) will cause global changes in gene expression shows there are more than mere non-specific protein interactions [45]. Nonetheless, multi-target interactions, specific or non-specific, at the protein or nucleic acid level, will be a plus in CSC-targeting as long as normal stem cells are spared.
Additional concerns are phytochemical bioavailability in vivo and product quality. For some polyphenolic compounds, effects in vivo, although significant, are less prominent than ones observed in vitro [134]. The bioavailability issue has been addressed with enhancing compounds, such as piperine from the spice black pepper (Piper nigrum), which amplifies the effect of phytochemicals (including curcumin). Bioavailability can be aided by formulations and special delivery systems, such as nanoparticles for curcumin and micelles for niclosamide [38,[135], [136], [137]]. Curcumin is an example which we managed to devise a measure to enhance bioavailability. By oral gavage to mice on empty stomach, we demonstrated in vivo effect: curcumin inhibition of the inflammatory iNOS in murine liver (beneficial outcome) and exacerbation of murine visceral leishmaniasis, a protozoan parasitic disease (detrimental outcome) [37,138]. Another concern is reliability of the source of phytochemicals. Consistent and unadulterated phytochemicals are essential, for example, curcumin as Curcumin C3 Complex from Sabinsa and EGCG as Polyphenon E from Mitsui Norin (as used in some clinical trials, see Table 1) [6]. (Repurposed drugs, as synthetic compounds, do not have this problem.) Yet another approach is the synthesis of better analogs and derivatives (such as difluorinated-curcumin and various chloroquine analogs), but they are beyond the scope of this review [32,40,54,120].
Discrepancies in phytochemical research results are another challenge. Whereas we found curcumin inhibited SP in rat glioma [48], Shi et al. (2015) found curcumin induced human glioma CSCs [139]. Kakarala et al. (2010) reported curcumin inhibited both breast CSCs and normal human breast stem cells, thus affecting both CSCs and normal stem cells [140]. These differences can be attributed to variations in dosage, length of treatment periods, grade and stability of compounds used, or other experimental conditions [133]. Polyphenolic phytochemicals have antioxidant capacities against reactive oxygen species (ROS), but under special conditions they exhibit pro-oxidant capacity [141]. Whereas ROS are harmful in general, the removal of too much ROS interferes with bodily functions, as seen in the warning presented by scientists to the food industry [142].
Related to dosage is the concept of hormesis [143], which suggests the fundamental nature of the dose-response curve is neither linear nor threshold, but U- or J-shaped. A low dose stimulatory response is the hormetic effect, representing overcompensation in response to disruptions in homeostasis. A biphasic dose response is observed. At high concentrations, phytochemicals can be toxic, whereas sub-toxic doses may induce adaptive stress responses. Hormetic mechanisms of action have been proposed to underlie many of the health benefits of phytochemicals [6]. Protective effect of resveratrol is only observed in low dose but not high dose in a mouse colon cancer model [144]. Similar dose effects are seen in genistein [84].
For repurposed drugs, most research results indicate CSC-targeting, there are discordance. Sancho et al. (2015) reported metformin-resistant pancreatic CSCs [145] and Xin et al. (2016) reported metformin-resistant liver CSCs [146]. In these studies, the effective dose of metformin is much higher than other drugs or phytochemicals. Moreover, Asiedu et al. (2018) reported heterogeneity in metformin response in both patient-derived and cancer cell lines [147]. Chloroquine is “a double-edged sword of autophagy”, causing severe kidney damage [148]. Whether it is the compound's pleiotropic nature that leads to unexpected adverse effects is still unclear. For repurposed drugs, studies are needed to determine that they can reach the CSC niche [7].
7. Conclusion
Combinational treatment has a long history in cancer chemotherapy. The first successful example is the treatment of childhood leukemia with “VAMP”, four drug combination (vincristine, amethopterin, 6-mercaptopurine, prednisone) in 1963 [149]. Our approach continues this tradition. We recommend four phytochemicals (curcumin, resverestrol, EGCG, genistein) and four drugs (metformin, niclosamide, thioridazine, chloroquine). This choice is in sync with selections by the Halifax Project, aiming at multi-targeted compounds for cancer prophylaxis and treatment [1]. We advocate a novel approach of CSC-targeting dietary phytochemical-repurposed drug combinations and look forward towards clinical applications.
8. Future perspectives
In the United States, cancer drug prices are getting out of hand. The March 2018 report to President Donald Trump stated: “The President's Cancer Panel concluded that addressing the dramatic rise of cancer drug prices must be made a national priority. … Innovative drugs offer new hope for patients to achieve long-term remissions—even cures—but virtually all new cancer drugs enter the market with a price tag that exceeds $100,000 per year and, increasingly, much higher” (https://PresCancerPanel.cancer.gov/report/drugvalue). Instead of new cancer drugs, the older repurposed drugs in combination with dietary phytochemicals are inexpensive and may work just as well.
Cancer is a worldwide problem affecting all nations rich and poor. On this topic, Sullivan et al. (2017) warned: “Treating cancer with the latest drugs and techniques is costly and will not improve survival globally” [150]. They asserted: “Cancer is on the rise” and concluded the focus should be “on building infrastructure, and delivering affordable, equitable and effective care”. Our idea of CSC-targeting phytochemical-drug combination fits seamlessly in this scenario as effective and affordable cancer treatment for the global population.
For World Health Organization (WHO), cancer is non-communicable chronic disease, and the increase in cancer is due to an aging population and lifestyle changes. There is an urgent need for affordable cancer treatment. Whether independently administered or in combination (as adjuvant with current therapy), development of a CSC-targeting dietary phytochemical-repurposed drug combination will be affordable, effective, and low toxicity. Our goal is to increase the scientific community's awareness and build a momentum towards conducting well-controlled clinical trials, done under uniform experimental conditions and in a double-blinded manner, to fully validate this novel combinatorial approach. Since we propose to combine dietary phytochemicals and repurposed drugs, i.e. non-patentable natural products and patent-expired older drugs, we envision financial supports for this endeavor have to come from governmental and philanthropic resources. The novel approach is ready for action.
Conflicts of interest
None declared.
Acknowledgements
We thank Drs. David Axelrod and Dongming Sun for helpful comments. We are grateful to Claire Chen, Chase Christensen, Nana Haruna, Avery Lee and Tanjida Tasmin for assistance in manuscript preparation. MMC received grant support from American Institute for Cancer Research. RC was the recipient of undergraduate research fund from Aresty Research Center at Rutgers University. DF received grant support from New Jersey Health Foundation (PC55-16).
References
- 1.Block K.I., Gyllenhaal C., Lowe L., Amedei A., Amin A.R.M.R., Amin A. Designing a broad-spectrum integrative approach for cancer prevention and treatment. Semin. Canc. Biol. 2015;35(Suppl):S276–S304. doi: 10.1016/j.semcancer.2015.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hanahan D., Weinberg R.A. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 3.Vogelstein B., Papadopoulos N., Velculescu V.E., Zhou S., Diaz L.A., Jr., Kinzler K.W. Cancer genome landscapes. Science. 2013;339:1546–1558. doi: 10.1126/science.1235122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tabassum D.P., Polyak K. Tumorigenesis: it takes a village. Nat. Rev. Canc. 2015;15:473–483. doi: 10.1038/nrc3971. [DOI] [PubMed] [Google Scholar]
- 5.Clarke M.F., Dick J.E., Dirks P.B., Eaves C.J., Jamieson C.H., Jones D.L. Cancer stem cells-perspectives on current status and future directions: AACR workshop on cancer stem cells. Canc. Res. 2006;66:9339–9344. doi: 10.1158/0008-5472.CAN-06-3126. [DOI] [PubMed] [Google Scholar]
- 6.Fong D., Chan M.M. Dietary phytochemicals target cancer stem cells for cancer chemoprevention. In: Chandra D., editor. Mitochondria as Targets for Phytochemicals in Cancer Prevention and Theraphy. Springer; New York: 2013. pp. 85–125. [Google Scholar]
- 7.Lv J., Shim J.S. Existing drugs and their application in drug discovery targeting cancer stem cells. Arch. Pharm. Res. (Seoul) 2015;38:1617–1626. doi: 10.1007/s12272-015-0628-1. [DOI] [PubMed] [Google Scholar]
- 8.Chan M.M., Fong D., Soprano K.J., Holmes W.F., Heverling H. Inhibition of growth and sensitization to cisplatin-mediated killing of ovarian cancer cells by polyphenolic chemopreventive agents. J. Cell. Physiol. 2003;194:63–70. doi: 10.1002/jcp.10186. [DOI] [PubMed] [Google Scholar]
- 9.Chan M.M., Soprano K.J., Weinstein K., Fong D. Epigallocatechin-3-gallate delivers hydrogen peroxide to induce death of ovarian cancer cells and enhances their cisplatin susceptibility. J. Cell. Physiol. 2006;207:389–396. doi: 10.1002/jcp.20569. [DOI] [PubMed] [Google Scholar]
- 10.de Kok T.M., van Breda S.G., Manson M.M. Mechanisms of combined action of different chemopreventive dietary compounds: a review. Eur. J. Nutr. 2008;47(2):51–59. doi: 10.1007/s00394-008-2006-y. [DOI] [PubMed] [Google Scholar]
- 11.Mencher S.K., Wang L.G. Promiscuous drugs compared to selective drugs (promiscuity can be a virtue) BMC Clin. Pharmacol. 2005;5 doi: 10.1186/1472-6904-5-3. 3–6904-5-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fojo T. Commentary: novel therapies for cancer: why dirty might be better. Oncology. 2008;13:277–283. doi: 10.1634/theoncologist.2007-0090. [DOI] [PubMed] [Google Scholar]
- 13.Nguyen L.V., Vanner R., Dirks P., Eaves C.J. Cancer stem cells: an evolving concept. Nat. Rev. Canc. 2012;12:133–143. doi: 10.1038/nrc3184. [DOI] [PubMed] [Google Scholar]
- 14.Nassar D., Blanpain C. Cancer stem cells: basic concepts and therapeutic implications. Annu. Rev. Pathol. 2016;11:47–76. doi: 10.1146/annurev-pathol-012615-044438. [DOI] [PubMed] [Google Scholar]
- 15.Donnenberg V.S., Donnenberg A.D. Multiple drug resistance in cancer revisited: the cancer stem cell hypothesis. J. Clin. Pharmacol. 2005;45:872–877. doi: 10.1177/0091270005276905. [DOI] [PubMed] [Google Scholar]
- 16.Zhou B.B., Zhang H., Damelin M., Geles K.G., Grindley J.C., Dirks P.B. Tumour-initiating cells: challenges and opportunities for anticancer drug discovery. Nat. Rev. Drug Discov. 2009;8:806–823. doi: 10.1038/nrd2137. [DOI] [PubMed] [Google Scholar]
- 17.Vidal S.J., Rodriguez-Bravo V., Galsky M., Cordon-Cardo C., Domingo-Domenech J. Targeting cancer stem cells to suppress acquired chemotherapy resistance. Oncogene. 2014;33:4451–4463. doi: 10.1038/onc.2013.411. [DOI] [PubMed] [Google Scholar]
- 18.Phi L.T.H., Sari I.N., Yang Y.G., Lee S.H., Jun N., Kim K.S. Cancer stem cells (CSCs) in drug resistance and their therapeutic implications in cancer treatment. Stem Cell. Int. 2018;2018 doi: 10.1155/2018/5416923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sotiropoulou P.A., Christodoulou M.S., Silvani A., Herold-Mende C., Passarella D. Chemical approaches to targeting drug resistance in cancer stem cells. Drug Discov. Today. 2014;19:1547–1562. doi: 10.1016/j.drudis.2014.05.002. [DOI] [PubMed] [Google Scholar]
- 20.Sato R., Semba T., Saya H., Arima Y. Concise review: stem cells and epithelial-mesenchymal transition in cancer: biological implications and therapeutic targets. Stem Cell. 2016;34:1997–2007. doi: 10.1002/stem.2406. [DOI] [PubMed] [Google Scholar]
- 21.Brabletz T., Kalluri R., Nieto M.A., Weinberg R.A. EMT in cancer. Nat. Rev. Canc. 2018;18:128–134. doi: 10.1038/nrc.2017.118. [DOI] [PubMed] [Google Scholar]
- 22.Xu X., Chai S., Wang P., Zhang C., Yang Y., Yang Y. Aldehyde dehydrogenases and cancer stem cells. Canc. Lett. 2015;369:50–57. doi: 10.1016/j.canlet.2015.08.018. [DOI] [PubMed] [Google Scholar]
- 23.Valent P., Bonnet D., De Maria R., Lapidot T., Copland M., Melo J.V. Cancer stem cell definitions and terminology: the devil is in the details. Nat. Rev. Canc. 2012;12:767–775. doi: 10.1038/nrc3368. [DOI] [PubMed] [Google Scholar]
- 24.Pastrana E., Silva-Vargas V., Doetsch F. Eyes wide open: a critical review of sphere-formation as an assay for stem cells. Cell. Stem Cell. 2011;8:486–498. doi: 10.1016/j.stem.2011.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Naujokat C., Laufer S. Targeting cancer stem cells with defined compounds and drugs. J. Canc. Res. Updates. 2013:36–67. [Google Scholar]
- 26.Taylor W.F., Jabbarzadeh E. The use of natural products to target cancer stem cells. Am. J. Cancer. Res. 2017;7:1588–1605. [PMC free article] [PubMed] [Google Scholar]
- 27.Li Y., Wicha M.S., Schwartz S.J., Sun D. Implications of cancer stem cell theory for cancer chemoprevention by natural dietary compounds. J. Nutr. Biochem. 2011;22:799–806. doi: 10.1016/j.jnutbio.2010.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Surh Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Canc. 2003;3:768–780. doi: 10.1038/nrc1189. [DOI] [PubMed] [Google Scholar]
- 29.Chan M.M., Fong D. Overcoming drug resistance by phytochemicals. In: Siddik Z., Mehta K., editors. Drug Resistance in Cancer Cells. Springer; New York: 2009. pp. 314–344. [Google Scholar]
- 30.Budisan L., Gulei D., Zanoaga O.M., Irimie A.I., Sergiu C., Braicu C. Dietary intervention by phytochemicals and their role in modulating coding and non-coding genes in cancer. Int. J. Mol. Sci. 2017;18 doi: 10.3390/ijms18061178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kawasaki B.T., Hurt E.M., Mistree T., Farrar W.L. Targeting cancer stem cells with phytochemicals. Mol. Interv. 2008;8:174–184. doi: 10.1124/mi.8.4.9. [DOI] [PubMed] [Google Scholar]
- 32.Dandawate P., Padhye S., Ahmad A., Sarkar F.H. Novel strategies targeting cancer stem cells through phytochemicals and their analogs. Drug Deliv. Transl. Res. 2013;3:165–182. doi: 10.1007/s13346-012-0079-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Scarpa E.S., Ninfali P. Phytochemicals as innovative therapeutic tools against cancer stem cells. Int. J. Mol. Sci. 2015;16:15727–15742. doi: 10.3390/ijms160715727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Oh J., Hlatky L., Jeong Y.S., Kim D. Therapeutic effectiveness of anticancer phytochemicals on cancer stem cells. Toxins. 2016;8 doi: 10.3390/toxins8070199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Singh A.K., Sharma N., Ghosh M., Park Y.H., Jeong D.K. Emerging importance of dietary phytochemicals in fight against cancer: role in targeting cancer stem cells. Crit. Rev. Food Sci. Nutr. 2017;57:3449–3463. doi: 10.1080/10408398.2015.1129310. [DOI] [PubMed] [Google Scholar]
- 36.Kim Y.S., Farrar W., Colburn N.H., Milner J.A. Cancer stem cells: potential target for bioactive food components. J. Nutr. Biochem. 2012;23:691–698. doi: 10.1016/j.jnutbio.2012.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chan M.M., Huang H.I., Fenton M.R., Fong D. In vivo inhibition of nitric oxide synthase gene expression by curcumin, a cancer preventive natural product with anti-inflammatory properties. Biochem. Pharmacol. 1998;55:1955–1962. doi: 10.1016/s0006-2952(98)00114-2. [DOI] [PubMed] [Google Scholar]
- 38.Panda A.K., Chakraborty D., Sarkar I., Khan T., Sa G. New insights into therapeutic activity and anticancer properties of curcumin. J. Exp. Pharmacol. 2017;9:31–45. doi: 10.2147/JEP.S70568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Norris L., Karmokar A., Howells L., Steward W.P., Gescher A., Brown K. The role of cancer stem cells in the anti-carcinogenicity of curcumin. Mol. Nutr. Food Res. 2013;57:1630–1637. doi: 10.1002/mnfr.201300120. [DOI] [PubMed] [Google Scholar]
- 40.Li Y., Zhang T. Targeting cancer stem cells by curcumin and clinical applications. Canc. Lett. 2014;346:197–205. doi: 10.1016/j.canlet.2014.01.012. [DOI] [PubMed] [Google Scholar]
- 41.Sordillo P.P., Helson L. Curcumin and cancer stem cells: curcumin has asymmetrical effects on cancer and normal stem cells. Anticancer Res. 2015;35:599–614. [PubMed] [Google Scholar]
- 42.Ramasamy T.S., Ayob A.Z., Myint H.H., Thiagarajah S., Amini F. Targeting colorectal cancer stem cells using curcumin and curcumin analogues: insights into the mechanism of the therapeutic efficacy. Canc. Cell Int. 2015;15 doi: 10.1186/s12935-015-0241-x. 96–9015-0241-x. eCollection 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tsai C.F., Hsieh T.H., Lee J.N., Hsu C.Y., Wang Y.C., Kuo K.K. Curcumin suppresses phthalate-induced metastasis and the proportion of cancer stem cell (CSC)-like cells via the inhibition of AhR/ERK/SK1 signaling in hepatocellular carcinoma. J. Agric. Food Chem. 2015;63:10388–10398. doi: 10.1021/acs.jafc.5b04415. [DOI] [PubMed] [Google Scholar]
- 44.Subramaniam D., Ponnurangam S., Ramamoorthy P., Standing D., Battafarano R.J., Anant S. Curcumin induces cell death in esophageal cancer cells through modulating notch signaling. PLoS One. 2012;7 doi: 10.1371/journal.pone.0030590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Huminiecki L., Horbanczuk J., Atanasov A.G. The functional genomic studies of curcumin. Semin. Canc. Biol. 2017;46:107–118. doi: 10.1016/j.semcancer.2017.04.002. [DOI] [PubMed] [Google Scholar]
- 46.Zhuang W., Long L., Zheng B., Ji W., Yang N., Zhang Q. Curcumin promotes differentiation of glioma-initiating cells by inducing autophagy. Canc. Sci. 2012;103:684–690. doi: 10.1111/j.1349-7006.2011.02198.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Baharuddin P., Satar N., Fakiruddin K.S., Zakaria N., Lim M.N., Yusoff N.M. Curcumin improves the efficacy of cisplatin by targeting cancer stem-like cells through p21 and cyclin D1-mediated tumour cell inhibition in non-small cell lung cancer cell lines. Oncol. Rep. 2016;35:13–25. doi: 10.3892/or.2015.4371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fong D., Yeh A., Naftalovich R., Choi T.H., Chan M.M. Curcumin inhibits the side population (SP) phenotype of the rat C6 glioma cell line: towards targeting of cancer stem cells with phytochemicals. Canc. Lett. 2010;293:65–72. [Google Scholar]
- 49.Zhou Q., Ye M., Lu Y., Zhang H., Chen Q., Huang S. Curcumin improves the tumoricidal effect of mitomycin C by suppressing ABCG2 expression in stem cell-like breast cancer cells. PLoS One. 2015;10 doi: 10.1371/journal.pone.0136694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Buhrmann C., Kraehe P., Lueders C., Shayan P., Goel A., Shakibaei M. Curcumin suppresses crosstalk between colon cancer stem cells and stromal fibroblasts in the tumor microenvironment: potential role of EMT. PLoS One. 2014;9 doi: 10.1371/journal.pone.0107514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Park E.J., Pezzuto J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta. 2015;1852:1071–1113. doi: 10.1016/j.bbadis.2015.01.014. [DOI] [PubMed] [Google Scholar]
- 52.Kulkarni S.S., Canto C. The molecular targets of resveratrol. Biochim. Biophys. Acta. 2015;1852:1114–1123. doi: 10.1016/j.bbadis.2014.10.005. [DOI] [PubMed] [Google Scholar]
- 53.McCubrey J.A., Lertpiriyapong K., Steelman L.S., Abrams S.L., Yang L.V., Murata R.M. Effects of resveratrol, curcumin, berberine and other nutraceuticals on aging, cancer development, cancer stem cells and microRNAs. Aging (N Y) 2017;9:1477–1536. doi: 10.18632/aging.101250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bhullar K.S., Hubbard B.P. Lifespan and healthspan extension by resveratrol. Biochim. Biophys. Acta. 2015;1852:1209–1218. doi: 10.1016/j.bbadis.2015.01.012. [DOI] [PubMed] [Google Scholar]
- 55.Chan M.M. Antimicrobial effect of resveratrol on dermatophytes and bacterial pathogens of the skin. Biochem. Pharmacol. 2002;63:99–104. doi: 10.1016/s0006-2952(01)00886-3. [DOI] [PubMed] [Google Scholar]
- 56.Fu Y., Chang H., Peng X., Bai Q., Yi L., Zhou Y. Resveratrol inhibits breast cancer stem-like cells and induces autophagy via suppressing Wnt/beta-catenin signaling pathway. PLoS One. 2014;9 doi: 10.1371/journal.pone.0102535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shankar S., Nall D., Tang S.N., Meeker D., Passarini J., Sharma J. Resveratrol inhibits pancreatic cancer stem cell characteristics in human and KrasG12D transgenic mice by inhibiting pluripotency maintaining factors and epithelial-mesenchymal transition. PLoS One. 2011;6 doi: 10.1371/journal.pone.0016530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yang Y.P., Chang Y.L., Huang P.I., Chiou G.Y., Tseng L.M., Chiou S.H. Resveratrol suppresses tumorigenicity and enhances radiosensitivity in primary glioblastoma tumor initiating cells by inhibiting the STAT3 axis. J. Cell. Physiol. 2012;227:976–993. doi: 10.1002/jcp.22806. [DOI] [PubMed] [Google Scholar]
- 59.Pandey P.R., Okuda H., Watabe M., Pai S.K., Liu W., Kobayashi A. Resveratrol suppresses growth of cancer stem-like cells by inhibiting fatty acid synthase. Breast Canc. Res. Treat. 2011;130:387–398. doi: 10.1007/s10549-010-1300-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sayd S., Thirant C., El-Habr E.A., Lipecka J., Dubois L.G., Bogeas A. Sirtuin-2 activity is required for glioma stem cell proliferation arrest but not necrosis induced by resveratrol. Stem Cell Rev. 2014;10:103–113. doi: 10.1007/s12015-013-9465-0. [DOI] [PubMed] [Google Scholar]
- 61.Chen C.N., Liang C.M., Lai J.R., Tsai Y.J., Tsay J.S., Lin J.K. Capillary electrophoretic determination of the anine, caffeine, and catechins in fresh tea leaves and oolong tea and their effects on rat neurosphere adhesion and migration. J. Agric. Food Chem. 2003;51:7495–7503. doi: 10.1021/jf034634b. [DOI] [PubMed] [Google Scholar]
- 62.Khan N., Mukhtar H. Multitargeted therapy of cancer by green tea polyphenols. Canc. Lett. 2008;269:269–280. doi: 10.1016/j.canlet.2008.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kim H.S., Quon M.J., Kim J.A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2014;2:187–195. doi: 10.1016/j.redox.2013.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Gan R.Y., Li H.B., Sui Z.Q., Corke H. Absorption, metabolism, anti-cancer effect and molecular targets of epigallocatechin gallate (EGCG): an updated review. Crit. Rev. Food Sci. Nutr. 2018;58:924–941. doi: 10.1080/10408398.2016.1231168. [DOI] [PubMed] [Google Scholar]
- 65.Kumazoe M., Tachibana H. Anti-cancer effect of EGCG and its mechanisms. 2016;6:70–78. [Google Scholar]
- 66.Nishimura N., Hartomo T.B., Pham T.V., Lee M.J., Yamamoto T., Morikawa S. Epigallocatechin gallate inhibits sphere formation of neuroblastoma BE(2)-C cells. Environ. Health Prev. Med. 2012;17:246–251. doi: 10.1007/s12199-011-0239-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mineva N.D., Paulson K.E., Naber S.P., Yee A.S., Sonenshein G.E. Epigallocatechin-3-gallate inhibits stem-like inflammatory breast cancer cells. PLoS One. 2013;8 doi: 10.1371/journal.pone.0073464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Chen Y., Wang X.Q., Zhang Q., Zhu J.Y., Li Y., Xie C.F. (-)-Epigallocatechin-3-gallate inhibits colorectal cancer stem cells by suppressing Wnt/beta-catenin pathway. Nutrients. 2017;9 doi: 10.3390/nu9060572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Fujiki H., Sueoka E., Watanabe T., Suganuma M. Synergistic enhancement of anticancer effects on numerous human cancer cell lines treated with the combination of EGCG, other green tea catechins, and anticancer compounds. J. Canc. Res. Clin. Oncol. 2015;141:1511–1522. doi: 10.1007/s00432-014-1899-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tang S.N., Fu J., Nall D., Rodova M., Shankar S., Srivastava R.K. Inhibition of sonic hedgehog pathway and pluripotency maintaining factors regulate human pancreatic cancer stem cell characteristics. Int. J. Canc. 2012;131:30–40. doi: 10.1002/ijc.26323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhang Y., Wang S.X., Ma J.W., Li H.Y., Ye J.C., Xie S.M. EGCG inhibits properties of glioma stem-like cells and synergizes with temozolomide through downregulation of P-glycoprotein inhibition. J. Neuro Oncol. 2015;121:41–52. doi: 10.1007/s11060-014-1604-1. [DOI] [PubMed] [Google Scholar]
- 72.Lee S.H., Nam H.J., Kang H.J., Kwon H.W., Lim Y.C. Epigallocatechin-3-gallate attenuates head and neck cancer stem cell traits through suppression of notch pathway. Eur. J. Canc. 2013;49:3210–3218. doi: 10.1016/j.ejca.2013.06.025. [DOI] [PubMed] [Google Scholar]
- 73.Li Y.J., Wu S.L., Lu S.M., Chen F., Guo Y., Gan S.M. (-)-Epigallocatechin-3-gallate inhibits nasopharyngeal cancer stem cell self-renewal and migration and reverses the epithelial-mesenchymal transition via NF-kappaB p65 inactivation. Tumour Biol. 2015;36:2747–2761. doi: 10.1007/s13277-014-2899-4. [DOI] [PubMed] [Google Scholar]
- 74.Lin C.H., Chao L.K., Hung P.H., Chen Y.J. EGCG inhibits the growth and tumorigenicity of nasopharyngeal tumor-initiating cells through attenuation of STAT3 activation. Int. J. Clin. Exp. Pathol. 2014;7:2372–2381. [PMC free article] [PubMed] [Google Scholar]
- 75.Lecumberri E., Dupertuis Y.M., Miralbell R., Pichard C. Green tea polyphenol epigallocatechin-3-gallate (EGCG) as adjuvant in cancer therapy. Clin. Nutr. 2013;32:894–903. doi: 10.1016/j.clnu.2013.03.008. [DOI] [PubMed] [Google Scholar]
- 76.Banerjee S., Li Y., Wang Z., Sarkar F.H. Multi-targeted therapy of cancer by genistein. Canc. Lett. 2008;269:226–242. doi: 10.1016/j.canlet.2008.03.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Taylor C.K., Levy R.M., Elliott J.C., Burnett B.P. The effect of genistein aglycone on cancer and cancer risk: a review of in vitro, preclinical, and clinical studies. Nutr. Rev. 2009;67:398–415. doi: 10.1111/j.1753-4887.2009.00213.x. [DOI] [PubMed] [Google Scholar]
- 78.Russo M., Russo G.L., Daglia M., Kasi P.D., Ravi S., Nabavi S.F. Understanding genistein in cancer: the “good” and the “bad” effects: a review. Food Chem. 2016;196:589–600. doi: 10.1016/j.foodchem.2015.09.085. [DOI] [PubMed] [Google Scholar]
- 79.Wang Q., Ge X., Tian X., Zhang Y., Zhang J., Zhang P. Soy isoflavone: the multipurpose phytochemical. Biomed. Rep. 2013;1:697–701. doi: 10.3892/br.2013.129. (review) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Huang W., Wan C., Luo Q., Huang Z., Luo Q. Genistein-inhibited cancer stem cell-like properties and reduced chemoresistance of gastric cancer. Int. J. Mol. Sci. 2014;15:3432–3443. doi: 10.3390/ijms15033432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Fan P., Fan S., Wang H., Mao J., Shi Y., Ibrahim M.M. Genistein decreases the breast cancer stem-like cell population through hedgehog pathway. Stem Cell Res. Ther. 2013;4:146. doi: 10.1186/scrt357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zhang L., Li L., Jiao M., Wu D., Wu K., Li X. Genistein inhibits the stemness properties of prostate cancer cells through targeting hedgehog-Gli1 pathway. Canc. Lett. 2012;323:48–57. doi: 10.1016/j.canlet.2012.03.037. [DOI] [PubMed] [Google Scholar]
- 83.Carlo-Stella C., Dotti G., Mangoni L., Reggazi E., Garau D., Bonati A. Selection of myeloid progenitors lacking BCR/ABL mRNA in chronic myelogenous leukemia patients after in vitro treatment with the tyrosine kinase inhibitor genistein. Blood. 1996;88:3091–3100. [PubMed] [Google Scholar]
- 84.Montales M.T., Rahal O.M., Nakatani H., Matsuda T., Simmen R.C. Repression of mammary adipogenesis by genistein limits mammosphere formation of human MCF-7 cells. J. Endocrinol. 2013;218:135–149. doi: 10.1530/JOE-12-0520. [DOI] [PubMed] [Google Scholar]
- 85.Montales M.T., Rahal O.M., Kang J., Rogers T.J., Prior R.L., Wu X. Repression of mammosphere formation of human breast cancer cells by soy isoflavone genistein and blueberry polyphenolic acids suggests diet-mediated targeting of cancer stem-like/progenitor cells. Carcinogenesis. 2012;33:652–660. doi: 10.1093/carcin/bgr317. [DOI] [PubMed] [Google Scholar]
- 86.Dolinoy D.C., Weidman J.R., Waterland R.A., Jirtle R.L. Maternal genistein alters coat color and protects avy mouse offspring from obesity by modifying the fetal epigenome. Environ. Health Perspect. 2006;114:567–572. doi: 10.1289/ehp.8700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Nosengo N. Can you teach old drugs new tricks? Nature. 2016;534:314–316. doi: 10.1038/534314a. [DOI] [PubMed] [Google Scholar]
- 88.Amelio I., Gostev M., Knight R.A., Willis A.E., Melino G., Antonov A.V. DRUGSURV: a resource for repositioning of approved and experimental drugs in oncology based on patient survival information. Cell Death Dis. 2014;5 doi: 10.1038/cddis.2014.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Corsello S.M., Bittker J.A., Liu Z., Gould J., McCarren P., Hirschman J.E. The drug repurposing hub: a next-generation drug library and information resource. Nat. Med. 2017;23:405–408. doi: 10.1038/nm.4306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Vazquez-Martin A., Lopez-Bonetc E., Cufi S., Oliveras-Ferraros C., Del Barco S., Martin-Castillo B. Repositioning chloroquine and metformin to eliminate cancer stem cell traits in pre-malignant lesions. Drug Resist. Updates. 2011;14:212–223. doi: 10.1016/j.drup.2011.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pryor R., Cabreiro F. Repurposing metformin: an old drug with new tricks in its binding pockets. Biochem. J. 2015;471:307–322. doi: 10.1042/BJ20150497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kim T.H., Suh D.H., Kim M.K., Song Y.S. Metformin against cancer stem cells through the modulation of energy metabolism: special considerations on ovarian cancer. BioMed Res. Int. 2014;2014:132702. doi: 10.1155/2014/132702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lei Y., Yi Y., Liu Y., Liu X., Keller E.T., Qian C.N. Metformin targets multiple signaling pathways in cancer. Chin. J. Canc. 2017;36 doi: 10.1186/s40880-017-0184-9. 17–017-0184-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Gantois I., Khoutorsky A., Popic J., Aguilar-Valles A., Freemantle E., Cao R. Metformin ameliorates core deficits in a mouse model of fragile X syndrome. Nat. Med. 2017;23:674–677. doi: 10.1038/nm.4335. [DOI] [PubMed] [Google Scholar]
- 95.Saini N., Yang X. Metformin as an anti-cancer agent: actions and mechanisms targeting cancer stem cells. Acta Biochim. Biophys. Sin. 2018;50:133–143. doi: 10.1093/abbs/gmx106. [DOI] [PubMed] [Google Scholar]
- 96.Hall S.S. A trial for the ages. Science. 2015;349:1274–1278. doi: 10.1126/science.349.6254.1274. [DOI] [PubMed] [Google Scholar]
- 97.Soo J.S., Ng C.H., Tan S.H., Malik R.A., Teh Y.C., Tan B.S. Metformin synergizes 5-fluorouracil, epirubicin, and cyclophosphamide (FEC) combination therapy through impairing intracellular ATP production and DNA repair in breast cancer stem cells. Apoptosis. 2015;20:1373–1387. doi: 10.1007/s10495-015-1158-5. [DOI] [PubMed] [Google Scholar]
- 98.Cuyas E., Martin-Castillo B., Bosch-Barrera J., Menendez J.A. Metformin inhibits RANKL and sensitizes cancer stem cells to denosumab. Cell Cycle. 2017;16:1022–1028. doi: 10.1080/15384101.2017.1310353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Chai X., Chu H., Yang X., Meng Y., Shi P., Gou S. Metformin increases sensitivity of pancreatic cancer cells to gemcitabine by reducing CD133+ cell populations and suppressing ERK/P70S6K signaling. Sci. Rep. 2015;5:14404. doi: 10.1038/srep14404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hirsch H.A., Iliopoulos D., Tsichlis P.N., Struhl K. Metformin selectively targets cancer stem cells, and acts together with chemotherapy to block tumor growth and prolong remission. Canc. Res. 2009;69:7507–7511. doi: 10.1158/0008-5472.CAN-09-2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Iliopoulos D., Hirsch H.A., Struhl K. Metformin decreases the dose of chemotherapy for prolonging tumor remission in mouse xenografts involving multiple cancer cell types. Canc. Res. 2011;71:3196–3201. doi: 10.1158/0008-5472.CAN-10-3471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Montales M.T., Simmen R.C., Ferreira E.S., Neves V.A., Simmen F.A. Metformin and soybean-derived bioactive molecules attenuate the expansion of stem cell-like epithelial subpopulation and confer apoptotic sensitivity in human colon cancer cells. Genes Nutr. 2015;10 doi: 10.1007/s12263-015-0499-6. 49–4015-0499-6. Epub 2015 Oct 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ning X., Du Y., Ben Q., Huang L., He X., Gong Y. Bulk pancreatic cancer cells can convert into cancer stem cells(CSCs) in vitro and 2 compounds can target these CSCs. Cell Cycle. 2016;15:403–412. doi: 10.1080/15384101.2015.1127471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zhang H.H., Guo X.L. Combinational strategies of metformin and chemotherapy in cancers. Canc. Chemother. Pharmacol. 2016;78:13–26. doi: 10.1007/s00280-016-3037-3. [DOI] [PubMed] [Google Scholar]
- 105.Li Y., Li P.K., Roberts M.J., Arend R.C., Samant R.S., Buchsbaum D.J. Multi-targeted therapy of cancer by niclosamide: a new application for an old drug. Canc. Lett. 2014;349:8–14. doi: 10.1016/j.canlet.2014.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Tsai I.J., Zarowiecki M., Holroyd N., Garciarrubio A., Sanchez-Flores A., Brooks K.L. The genomes of four tapeworm species reveal adaptations to parasitism. Nature. 2013;496:57–63. doi: 10.1038/nature12031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Pan J.X., Ding K., Wang C.Y. Niclosamide, an old antihelminthic agent, demonstrates antitumor activity by blocking multiple signaling pathways of cancer stem cells. Chin. J. Canc. 2012;31:178–184. doi: 10.5732/cjc.011.10290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wieland A., Trageser D., Gogolok S., Reinartz R., Hofer H., Keller M. Anticancer effects of niclosamide in human glioblastoma. Clin. Canc. Res. 2013;19:4124–4136. doi: 10.1158/1078-0432.CCR-12-2895. [DOI] [PubMed] [Google Scholar]
- 109.Wang Y.C., Chao T.K., Chang C.C., Yo Y.T., Yu M.H., Lai H.C. Drug screening identifies niclosamide as an inhibitor of breast cancer stem-like cells. PLoS One. 2013;8 doi: 10.1371/journal.pone.0074538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Yo Y.T., Lin Y.W., Wang Y.C., Balch C., Huang R.L., Chan M.W. Growth inhibition of ovarian tumor-initiating cells by niclosamide. Mol. Canc. Therapeut. 2012;11:1703–1712. doi: 10.1158/1535-7163.MCT-12-0002. [DOI] [PubMed] [Google Scholar]
- 111.Arend R.C., Londono-Joshi A.I., Samant R.S., Li Y., Conner M., Hidalgo B. Inhibition of Wnt/beta-catenin pathway by niclosamide: a therapeutic target for ovarian cancer. Gynecol. Oncol. 2014;134:112–120. doi: 10.1016/j.ygyno.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 112.Liu J., Chen X., Ward T., Pegram M., Shen K. Combined niclosamide with cisplatin inhibits epithelial-mesenchymal transition and tumor growth in cisplatin-resistant triple-negative breast cancer. Tumour Biol. 2016;37:9825–9835. doi: 10.1007/s13277-015-4650-1. [DOI] [PubMed] [Google Scholar]
- 113.Thanacoody H.K. Thioridazine: resurrection as an antimicrobial agent? Br. J. Clin. Pharmacol. 2007;64:566–574. doi: 10.1111/j.1365-2125.2007.03021.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Cheng H.W., Liang Y.H., Kuo Y.L., Chuu C.P., Lin C.Y., Lee M.H. Identification of thioridazine, an antipsychotic drug, as an antiglioblastoma and anticancer stem cell agent using public gene expression data. Cell Death Dis. 2015;6 doi: 10.1038/cddis.2015.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Shen J., Ma B., Zhang X., Sun X., Han J., Wang Y. Thioridazine has potent antitumor effects on lung cancer stem-like cells. Oncol. Lett. 2017;13:1563–1568. doi: 10.3892/ol.2017.5651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lu M., Li J., Luo Z., Zhang S., Xue S., Wang K. Roles of dopamine receptors and their antagonist thioridazine in hepatoma metastasis. OncoTargets Ther. 2015;8:1543–1552. doi: 10.2147/OTT.S77373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Park M.S., Dong S.M., Kim B.R., Seo S.H., Kang S., Lee E.J. Thioridazine inhibits angiogenesis and tumor growth by targeting the VEGFR-2/PI3K/mTOR pathway in ovarian cancer xenografts. Oncotarget. 2014;5:4929–4934. doi: 10.18632/oncotarget.2063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ke X.Y., Lin Ng V.W., Gao S.J., Tong Y.W., Hedrick J.L., Yang Y.Y. Co-delivery of thioridazine and doxorubicin using polymeric micelles for targeting both cancer cells and cancer stem cells. Biomaterials. 2014;35:1096–1108. doi: 10.1016/j.biomaterials.2013.10.049. [DOI] [PubMed] [Google Scholar]
- 119.Sachlos E., Risueno R.M., Laronde S., Shapovalova Z., Lee J.H., Russell J. Identification of drugs including a dopamine receptor antagonist that selectively target cancer stem cells. Cell. 2012;149:1284–1297. doi: 10.1016/j.cell.2012.03.049. [DOI] [PubMed] [Google Scholar]
- 120.Solomon V.R., Lee H. Chloroquine and its analogs: a new promise of an old drug for effective and safe cancer therapies. Eur. J. Pharmacol. 2009;625:220–233. doi: 10.1016/j.ejphar.2009.06.063. [DOI] [PubMed] [Google Scholar]
- 121.Firat E., Weyerbrock A., Gaedicke S., Grosu A.L., Niedermann G. Chloroquine or chloroquine-PI3K/Akt pathway inhibitor combinations strongly promote gamma-irradiation-induced cell death in primary stem-like glioma cells. PLoS One. 2012;7 doi: 10.1371/journal.pone.0047357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Song Y.J., Zhang S.S., Guo X.L., Sun K., Han Z.P., Li R. Autophagy contributes to the survival of CD133+ liver cancer stem cells in the hypoxic and nutrient-deprived tumor microenvironment. Canc. Lett. 2013;339:70–81. doi: 10.1016/j.canlet.2013.07.021. [DOI] [PubMed] [Google Scholar]
- 123.Yang M.C., Wang H.C., Hou Y.C., Tung H.L., Chiu T.J., Shan Y.S. Blockade of autophagy reduces pancreatic cancer stem cell activity and potentiates the tumoricidal effect of gemcitabine. Mol. Canc. 2015;14 doi: 10.1186/s12943-015-0449-3. 179–015-0449-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Ojha R., Jha V., Singh S.K. Gemcitabine and mitomycin induced autophagy regulates cancer stem cell pool in urothelial carcinoma cells. Biochim. Biophys. Acta. 2016;1863:347–359. doi: 10.1016/j.bbamcr.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 125.Pagotto A., Pilotto G., Mazzoldi E.L., Nicoletto M.O., Frezzini S., Pasto A. Autophagy inhibition reduces chemoresistance and tumorigenic potential of human ovarian cancer stem cells. Cell Death Dis. 2017;8 doi: 10.1038/cddis.2017.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Liang D.H., Choi D.S., Ensor J.E., Kaipparettu B.A., Bass B.L., Chang J.C. The autophagy inhibitor chloroquine targets cancer stem cells in triple negative breast cancer by inducing mitochondrial damage and impairing DNA break repair. Canc. Lett. 2016;376:249–258. doi: 10.1016/j.canlet.2016.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Pascolo S. Time to use a dose of chloroquine as an adjuvant to anti-cancer chemotherapies. Eur. J. Pharmacol. 2016;771:139–144. doi: 10.1016/j.ejphar.2015.12.017. [DOI] [PubMed] [Google Scholar]
- 128.Ahmed M., Chaudhari K., Babaei-Jadidi R., Dekker L.V., Shams Nateri A. Concise review: emerging drugs targeting epithelial cancer stem-like cells. Stem Cell. 2017;35:839–850. doi: 10.1002/stem.2579. [DOI] [PubMed] [Google Scholar]
- 129.Renz B.W., D'Haese J.G., Werner J., Westphalen C.B., Ilmer M. Repurposing established compounds to target pancreatic cancer stem cells (CSCs) Med. Sci. 2017;5 doi: 10.3390/medsci5020014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ingolfsson H.I., Thakur P., Herold K.F., Hobart A., Ramsey N.B., Periole X. Phytochemicals perturb membranes and promiscuously alter protein function. ACS Chem. Biol. 2014;9:1788–1798. doi: 10.1021/cb500086e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Baell J., Walters M.A. Chemistry: chemical con artists foil drug discovery. Nature. 2014;513:481–483. doi: 10.1038/513481a. [DOI] [PubMed] [Google Scholar]
- 132.Bisson J., McAlpine J.B., Friesen J.B., Chen S.N., Graham J., Pauli G.F. Can invalid bioactives undermine natural product-based drug discovery? J. Med. Chem. 2016;59:1671–1690. doi: 10.1021/acs.jmedchem.5b01009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Nelson K.M., Dahlin J.L., Bisson J., Graham J., Pauli G.F., Walters M.A. The essential medicinal chemistry of curcumin. J. Med. Chem. 2017;60:1620–1637. doi: 10.1021/acs.jmedchem.6b00975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Williamson G., Manach C. Bioavailability and bioefficacy of polyphenols in humans. II. review of 93 intervention studies. Am. J. Clin. Nutr. 2005;81:243S–255S. doi: 10.1093/ajcn/81.1.243S. [DOI] [PubMed] [Google Scholar]
- 135.Aras A., Khokhar A.R., Qureshi M.Z., Silva M.F., Sobczak-Kupiec A., Pineda E.A. Targeting cancer with nano-bullets: curcumin, EGCG, resveratrol and quercetin on flying carpets. Asian Pac. J. Cancer Prev. APJCP. 2014;15:3865–3871. doi: 10.7314/apjcp.2014.15.9.3865. [DOI] [PubMed] [Google Scholar]
- 136.Misra S.K., Jensen T.W., Pan D. Enriched inhibition of cancer and stem-like cancer cells via STAT-3 modulating niclocelles. Nanoscale. 2015;7:7127–7132. doi: 10.1039/c5nr00403a. [DOI] [PubMed] [Google Scholar]
- 137.Mukherjee S., Baidoo J.N.E., Sampat S., Mancuso A., David L., Cohen L.S. A synergistic combination of curcumin, epicatechin gallate and resveratrol, repolarizes tumor-associated Microglia/Macrophages, and eliminates glioblastoma (GBM) and GBM stem cells. Molecules. 2018;23 doi: 10.3390/molecules23010201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Adapala N., Chan M.M. Long-term use of an antiinflammatory, curcumin, suppressed type 1 immunity and exacerbated visceral leishmaniasis in a chronic experimental model. Lab. Invest. 2008;88:1329–1339. doi: 10.1038/labinvest.2008.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Shi L., Wang Z., Sun G. Curcumin induces glioma stem-like cell formation. Neuroreport. 2015;26:167–172. doi: 10.1097/WNR.0000000000000320. [DOI] [PubMed] [Google Scholar]
- 140.Kakarala M., Brenner D.E., Korkaya H., Cheng C., Tazi K., Ginestier C., Liu S. Targeting breast stem cells with the cancer preventive compounds curcumin and piperine. Breast Canc. Res. Treat. 2010;122:777–785. doi: 10.1007/s10549-009-0612-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Russo G.L., Tedesco I., Spagnuolo C., Russo M. Antioxidant polyphenols in cancer treatment: friend, foe or foil? Semin. Canc. Biol. 2017;46:1–13. doi: 10.1016/j.semcancer.2017.05.005. [DOI] [PubMed] [Google Scholar]
- 142.Finley J.W., Kong A.N., Hintze K.J., Jeffery E.H., Ji L.L., Lei X.G. Antioxidants in foods: state of the science important to the food industry. J. Agric. Food Chem. 2011;59:6837–6846. doi: 10.1021/jf2013875. [DOI] [PubMed] [Google Scholar]
- 143.Calabrese E.J. Hormesis: from marginalization to mainstream: a case for hormesis as the default dose-response model in risk assessment. Toxicol. Appl. Pharmacol. 2004;197:125–136. doi: 10.1016/j.taap.2004.02.007. [DOI] [PubMed] [Google Scholar]
- 144.Cai H., Scott E., Kholghi A., Andreadi C., Ruffini A., Karmokar A. Cancer chemoprevention: evidence of a nonlinear dose response for the protective effects of resveratrol in humans and mice. Sci. Transl. Med. 2015;7 doi: 10.1126/scitranslmed.aaa7619. 298ra117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Sancho P., Burgos-Ramos E., Tavera A., Bou Kheir T., Jagust P., Schoenhals M. MYC/PGC-1alpha balance determines the metabolic phenotype and plasticity of pancreatic cancer stem cells. Cell Metabol. 2015;22:590–605. doi: 10.1016/j.cmet.2015.08.015. [DOI] [PubMed] [Google Scholar]
- 146.Xin H.W., Ambe C.M., Miller T.C., Chen J., Wiegand G.W., Anderson A.J. Liver label retaining cancer cells are relatively resistant to the reported anti-cancer stem cell drug metformin. J. Canc. 2016;7:1142–1151. doi: 10.7150/jca.10047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Asiedu M.K., Barron M., Aubry M.C., Wigle D.A. Patient- and cell type-specific heterogeneity of metformin response. Basic Clin. Pharmacol. Toxicol. 2018;122:214–222. doi: 10.1111/bcpt.12898. [DOI] [PubMed] [Google Scholar]
- 148.Kimura T., Takabatake Y., Takahashi A., Isaka Y. Chloroquine in cancer therapy: a double-edged sword of autophagy. Canc. Res. 2013;73:3–7. doi: 10.1158/0008-5472.CAN-12-2464. [DOI] [PubMed] [Google Scholar]
- 149.DeVita V.T., Jr., Chu E. A history of cancer chemotherapy. Canc. Res. 2008;68:8643–8653. doi: 10.1158/0008-5472.CAN-07-6611. [DOI] [PubMed] [Google Scholar]
- 150.Sullivan R., Pramesh C.S., Booth C.M. Cancer patients need better care, not just more technology. Nature. 2017;549:325–328. doi: 10.1038/549325a. [DOI] [PubMed] [Google Scholar]