Abstract
Diarrhoea caused by Cryptosporidium parvum is a major problem in calves younger than 4 weeks of age. To date only a few compounds have been approved for prophylactic and none for therapeutic use. Nitazoxanide (NTZ) has proven its efficacy in vitro against C. parvum and is approved by FDA for the treatment of human cryptosporidiosis.
In a first experimental study, 3 uninfected calves were treated with NTZ and pharmacokinetics was followed through blood samples. Serum samples of uninfected treated calves contained both NTZ metabolites (tizoxanide and tizoxanide glucuronide) and oral administration at 12 h intervals was considered as optimal. Three groups of three calves (1–3 days old) were then each inoculated with 1 × 107 oocysts of C. parvum (cattle genotype): the prophylactic group received 15 mg/kg body weight NTZ twice daily orally in milk from 1 day before to 8 days postinoculation (dpi). The therapeutic group received the same dosage of NTZ for 10 days from the appearance of diarrhoea (between 1 and 5 dpi). The control group was left untreated. All calves were monitored daily from day −1 to 28 dpi and faecal samples were collected for evaluation of consistency and for determination of oocyst numbers per gram (OPG) of faeces.
Diarrhoea was observed in all calves within the first week. Neither prophylactic nor therapeutic use of NTZ improved the clinical appearance and calves of the therapeutic showed a longer diarrheic episode (p < 0.05) with strong altered faecal consistence compared to the untreated control group.
The number of days with oocyst excretion did not differ significantly between the groups. In 5 out of 6 infected and treated calves oocyst excretion stopped only after discontinuation of treatment. In the prophylactic and in the control group mean values of the sum of the daily OPG per calf (8.5 × 106 and 8.0 × 106, respectively) and of the mean daily number of OPG (0.3 × 106 and 0.3 × 106, respectively) were similar, while the therapeutic group showed significantly lower values (1.9 × 106 and 0.06 × 106, respectively, p < 0.05). However oocyst determinations in this group may have been altered by the severe diarrhoea, diluting oocyst densities in the analysed faecal samples. In conclusion, these preliminary results about the first prophylactic and therapeutic use of NTZ in calves did not show the expected positive effect on the course of the Cryptosporidium-infection, neither on reducing the clinical severity, nor on oocyst excretion.
Keywords: Cryptosporidium parvum, Calves, Nitazoxanide, Prophylaxis, Treatment
1. Introduction
Cryptosporidium parvum is a protozoan parasite that can cause gastrointestinal disease in a wide variety of mammals, including humans, cattle, sheep, goat and pigs (Olson et al., 2004). Bovine cryptosporidiosis occurs worldwide with prevalences of 20–80% in young calves in different European countries (Appelbee et al., 2005). Studies performed in the United States showed that particularly calves at 2 weeks of age were excreting C. parvum oocysts (Santin et al., 2008) with prevalences of 45.8% in calves of 1–8 weeks of age. Prevalences in milking cows were significantly lower and mainly not concerning C. parvum, representing the only zoonotic species and also the most pathogenic species for cattle (Fayer et al., 2007). Although C. parvum infection is often detected in combination with other enteropathogens such as Rotavirus, Coronavirus, Escherichia coli K99 or Salmonella species, many experimental studies and investigations in the field have shown that Cryptosporidium may act as a primary pathogen (Tzipori et al., 1983, Naciri et al., 1999). More than 200 substances have been tested against cryptosporidiosis (O’Donoghue, 1995, Stockdale et al., 2008) and some such as halofuginone and decoquinate are reported to exhibit promising effects (Lefay et al., 2001, Lallemand et al., 2006). Prophylactic administration of paromomycin inhibited oocyst shedding and reduced the number of days with diarrhoea in experimentally infected calves (Fayer and Ellis, 1993), but in a field trial oocyst shedding and diarrhoea started with withdrawal of the drug and no reduction of the disease was obtained (Grinberg et al., 2002). Reports on the outcomes of halofuginone treatment are controversial: in some studies, calves showed significantly reduced oocyst excretion and decreased diarrhoea (Joachim et al., 2003) preventing mortality (Naciri et al., 1993). In another study treatments with halofuginone or decoquinate had no effect on the levels of diarrhoea or dehydration, but halofuginone significantly reduced the excretion of oocysts and decoquinate increased the average daily weight gain of calves (Lallemand et al., 2006). In a further study the administration of halofuginone during the first 7 days of life inhibited oocyst excretion until 2 weeks of age, but the incidence of diarrhoea was only delayed for 3.1 days and there was no significant difference compared to the control group concerning weight gain (Jarvie et al., 2005). Halofuginone is the only drug currently available for the treatment of bovine cryptosporidiosis, but it is known to be relatively toxic and care must be taken not to exceed the therapeutic dose. Nitazoxanide (NTZ) is an antimicrobial substance that has been applied for antiparasitic treatments in dogs, cats, sheep and goats (Euzeby et al., 1980, Gookin et al., 2001, Viel et al., 2007). NTZ has been FDA-approved for human use for the treatment of cryptosporidiosis and giardiasis in children and adults, and also for the treatment of equine protozoan myeloencephalitis caused by Sarcocystis neurona. The goal of the present pilot study was to investigate the efficacy of NTZ for the prophylaxis and treatment of cryptosporidiosis in calves.
2. Materials and methods
2.1. Housing and handling of animals and disease-monitoring
Animal experiments were approved and supervised by the Cantonal Veterinary Office of Zürich and the respective Animal Experimentation Ethics Committee. The study was carried out during May–December 2005 in experimental barns which are part of the veterinary hospital of the University of Zurich, Switzerland. Calves were purchased from surrounding farms 1–3 days after birth and all calves had received colostrum. They were housed in pens (8.5–10.5 m2) in groups of up to 5 animals with a litter of straw over a concrete base. The pens were cleaned daily with hot vapour pressure. Calves were fed with a commercial milk replacer supplied by an automatic milk deliverer; animals with reduced appetite were additionally hand-fed with milk and/or electrolyte solution. Water was supplied ad libitum and from the third week hay was also available. Calves were neither vaccinated, nor did they receive any supplementary vitamins during the experiment. All animals were checked twice daily clinically including faecal consistency and appetite. Weight was recorded daily. At the first manifestation of diarrhoea, faeces from all calves were tested once for antigens of bovine Coronavirus (FASTest® BCV), Rotavirus (FASTest® Rota), E. coli main pilar K99 (FASTest® E. coli-K99) and against C. parvum (FASTest® Crypto), all from MegaCor, Austria.
Calves were euthanized between 29 and 42 dpi. As part of the postmortem analysis, they were additionally examined by antigen detection in skin biopsies (Hilbe et al., 2007) for the presence of bovine virus diarrhoea (BVD). This was to exclude persistent and primary postnatal infections with BVD Virus resulting in immunosuppression, thereby enhancing the vulnerability of the calves to secondary infections (Potgieter, 1995).
2.2. Origin of Cryptosporidium oocysts
The Cryptosporidium-isolate used in this study originated from an affected outpatient calf of the animal hospital. Oocyst production was achieved in an experimentally inoculated calf (1 × 107 oocysts). Faeces from this calf had been collected by means of a bag that was attached through an appliance around the belly of the animal. Oocysts were purified on Ficoll gradients, and C. parvum “cattle genotype” was identified (Ward et al., 2002). These oocysts were stored for a maximum of 175 days at 4 °C in Phosphate Buffered Saline until to be used for infection experiments.
2.3. Experimental groups
At first NTZ was fed to three calves, representing the uninfected treatment group, for a period of 10 days starting from 1 day after their arrival at the experimental farm for the determination of NTZ metabolites. Afterwards, upon arrival at the experimental barns, 9 calves were randomly assigned to three experimental groups. On day 0 animals were inoculated with 1 × 107 C. parvum oocysts. In a first group, the prophylactic group, NTZ was administered prior to inoculation (day −1) and then daily for a period of 10 days. NTZ (kindly provided by Cambrex Profarmaco Landen NV; Belgium) was fed through a suckling bottle with milk replacer at 15 mg/kg bodyweight (BW) twice daily.
The three calves of a second group, the therapeutic group, received NTZ starting at the time point of the first manifestation of diarrhoea and then daily for a period of 10 days. The three calves of the third group, the infection control group, were left untreated.
2.4. Pharmacokinetics of NTZ metabolites
Blood samples were taken prior to drug treatment and at 1, 2, 4, 6, 8, 12, 24 and 48 h after the first administration of NTZ from 3 calves of the uninfected treatment group. Samples were sent to SGS Life Science Services, Wavre, Belgium, for the determination of the active metabolites tizoxanide and tizoxanide glucuronide as previously described (Stettler et al., 2004).
2.5. Assessment of faeces consistency and oocyst numbers
Faecal samples were directly collected in containers of about 2 dl from the rectum from all calves every day; each sample was labelled, sealed and transported to the laboratory on the day of collection. A single faecal smear was prepared from each mixed sample, stained with Ziehl–Neelsen (Eckert et al., 2005) for visualisation of oocysts and entirely examined by microscopy. If oocysts were present, samples were analysed by a modified method of Grinberg et al. (2002) to determine the number of cryptosporidial oocyst per gram (OPG) of faeces. Briefly, 1 g of the mixed faecal sample was mixed with 10 mL of tap water, passed through a 100-mesh sieve, the suspension centrifuged and the sediment resuspended in 4 mL of normal saline. Afterwards, 10 μl of this suspension was poured as a drop on a slide, air-dried and stained with Ziehl–Neelsen. The entire area of smear was examined with a 400× objective lens and oocysts counted. The OPG was then calculated by multiplying the result by 500 (or 5000, if there were too many oocysts to be counted and the sample had previously been diluted 10-fold). Results were not corrected for dilution in diarrhoeic stools. The mean OPG for each calf was the arithmetic mean of daily OPG from day −1 to 28 dpi. The number of oocysts shed was the sum of the daily OPG of each calf. To determine the effective total number of oocysts shed would have required the collection of all faeces on all days, this was not possible.
Faecal consistency was based on 3 scores: faeces solid, brownish and with adhesion at the plastic container, faeces semi-solid, yellowish and without adhesion at the plastic container (sample spread across the bottom of the container but not liquid), and liquid diarrhoea, as previously described (Grinberg et al., 2002). Differences in oocyst excretion intensities were statistically compared by maximum likelihood estimation based on the negative binomial distribution (Torgerson et al., 2003) and were considered significant with values p < 0.05. The number of days with diarrhoea or semi-solid faecal consistency was compared between treatment and control groups using the Student's t-test.
3. Results, discussion
3.1. Pharmacokinetics
Blood samples from uninfected NTZ treated calves contained both NTZ metabolites (Fig. 1 ). Tizoxanide (TIZ) represents the deacetylated primary metabolite, and was first detected within 1–2 h after administration (limit of traceability: 0.0500 μg/mL). Highest TIZ-levels were recorded between 2 and 8 h (0.941–1.57 μg/mL), followed by a decrease until the next administration. Tizoxanide glucuronide (TIG), was first detected slightly after TIZ (limit of traceability: 0.200 μg/mL) and highest levels were also seen between 2 and 8 h (0.239–1.11 μg/mL). TIG was no longer detected after 12 h. After oral administration in humans, maximum plasma concentrations of both metabolites had also been observed within 1–4 h, but reached levels up to 8 μg/mL (Broekhuysen et al., 2000). Oral administration with milk or milk replacer at 12 h intervals seemed to be an appropriate way for NTZ administration.
Fig. 1.
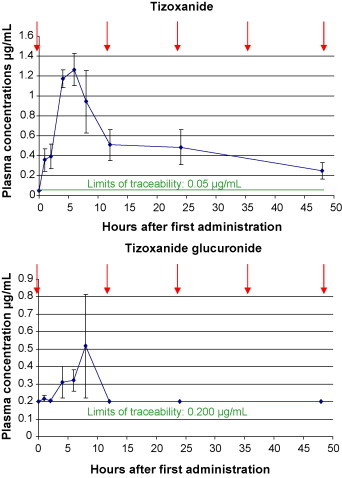
Mean and standard deviation of nitazoxanide metabolite-concentrations in blood samples of 3 calves prior to drug treatment (↓) and at 1, 2, 4, 6, 8, 12, 24 and 48 h after the first administration of NTZ through a suckling bottle with milk replacer at 15 mg/kg bodyweight (BW) every 12 h (↓).
3.2. Effects of NTZ treatments
Cryptosporidium-infection took place in all 9 infected animals. None of the calves, including the uninfected NTZ treated animals, was positive for viral or bacterial agents tested (C. parvum, Rota- and Coronaviruses, E. coli K99), so that other infectious agents potentially causing diarrhoea could be excluded. Skin biopsies postmortem were negative for BVD Virus.
Altered faecal consistency and particularly diarrhoea, main symptom of cryptosporidiosis in calves, was observed in all animals, but there were differences in duration and severity (Table 1 ). In the therapeutic group, animals showed a significantly higher (p < 0.05) number of days with liquid diarrhoea if compared with the infected but untreated control group. Bloody faeces were present for 3.33 and 2.66 days in the prophylactic and the therapeutic group respectively, while the infected untreated control group had a mean of 2 days with bloody faeces, but differences were not significant. Also calves from the uninfected group receiving NTZ developed diarrhoea for a mean duration of 15.3 days. Blood was present in faeces from one calf on days 3 and 4 of treatment, from the second calf on days 9 and 15 and from the third calf on days 8 and 9, for a mean of 2 days per animal. Thus the diarrhoea in this uninfected group was possibly caused by NTZ treatment.
Table 1.
Diarrhoea in calves experimentally infected with Cryptosporidium oocysts (P-, T- and C-group) and/or treated with nitazoxanide during the first 28 days of the experiment (uninfected NTZ-, P- and T-group).
| Animal number | Number of days with semi-solid faecal consistency | Number of days with liquid diarrhoea | |
|---|---|---|---|
| Uninfected NTZ-group | 1 | 5 | 5 |
| 2 | 14 | 4 | |
| 3 | 13 | 5 | |
| Mean (S.D.) | 10.7 (4.9) | 4.6 (0.6) | |
| Prophylactic group | 4 | 4 | 6 |
| 5 | 13 | 3 | |
| 6 | 9 | 6 | |
| Mean (S.D.) | 8.7 (4.5) | 5 (1.7) | |
| Therapeutic group | 7 | 10 | 8 |
| 8 | 10 | 5 | |
| 9 | 10 | 5 | |
| Mean (S.D.) | 10 (0) | 6a(1.7) | |
| Control group | 10 | 5 | 3 |
| 11 | 6 | 1 | |
| 12 | 15 | 1 | |
| Mean (S.D.) | 8.7 (5.2) | 1.7 (1.2) | |
p < 0.05: paired t test with control group.
In the untreated control group conspicuous liquid diarrhoea was detected in only 16.3% (1.7/10.4 days) of the days with altered faecal consistency. This indicated less pathology compared to the treatment groups. The percentages in the uninfected, the prophylactic and the therapeutic group were twice as much: 30.1% (4.6/15.3), 36.5% (5/13.7) and 37.5% (6/16), respectively. Additionally, 80% (4/5 days) and respectively 72.2% (4.3/6 days) of the days with liquid diarrhoea in the prophylactic and the therapeutic group occurred during the NTZ administration, suggesting a cumulative negative effect of NTZ in combination with parasite infection.
Results of daily OPG determination of infected calves are presented in Table 2 and the dynamic of oocyst excretion is shown in Fig. 2 . Oocyst excretion started in all infected animals within 2–4 dpi (days postinoculation) and highest excretion of oocysts took place at 4–7 dpi. In both infected treatment groups faecal consistency changed just before the start of oocyst excretion. After discontinuation of the medication in the prophylactic and the therapeutic group, the mean duration of oocyst excretion was 1.6 and 1 day, respectively. Consequently calves of these two groups excreted oocysts mainly during the treatment days (91.3% and 98.9%, respectively).
Table 2.
Number of days with oocyst excretion, mean OPG based on daily faecal samples (1–28 dpi) and number of oocysts shed (sum of the daily OPG per calf) on days 1–28 postinfection for each calf of the prophylactic, the therapeutic and the control group.
| Animal number | Number of days with oocyst excretion | Mean OPG excreted | Number of oocysts shed | |
|---|---|---|---|---|
| Prophylactic group | 4a | 24 | 0.4 × 106 | 12.8 × 106 |
| 5 | 9 | 0.2 × 106 | 7.4 × 106 | |
| 6 | 9 | 0.2 × 106 | 5.4 × 106 | |
| Mean (S.D.) | 14 (8.7) | 0.3 × 106 (0.12 × 106) | 8.5 × 106 (3.8 × 106) | |
| Therapeutic group | 7 | 5 | 0.02 × 106 | 0.7 × 106 |
| 8 | 11 | 0.07 × 106 | 2.7 × 106 | |
| 9 | 9 | 0.05 × 106 | 2.1 × 106 | |
| Mean (S.D.) | 8.3 (3.1) | 0.06 × 106b (0.03 × 106) | 1.9 × 106b (1.0 × 106) | |
| Control group | 10a | 17 | 0.4 × 106 | 11.7 × 106 |
| 11 | 9 | 0.2 × 106 | 8.7 × 106 | |
| 12 | 15 | 0.1 × 106 | 3.6 × 106 | |
| Mean (S.D.) | 13.6 (4.2) | 0.3 × 106 (0.14 × 106) | 8.0 × 106 (4.1 × 106) | |
Chronic carrier.
p < 0.05: maximum likelihood estimate based on negative binomial distribution.
Fig. 2.
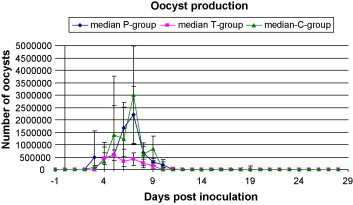
Median oocyst production and standard deviation in the prophylactic, therapeutic and control group from day −1 to 28 days postinoculation.
The number of oocysts shed and the daily mean OPG value was significantly lower in the therapeutic group. However, oocyst counts in this group may be altered by the severe diarrhoea, diluting oocyst densities in the faecal samples.
One chronic carrier was noted in each the prophylactic and the control group and none in the therapeutic group. However, the number of excreted oocysts from 11 dpi on in chronic carriers was very low. Also the faecal consistency of these two animals was not altered from 10 dpi. After 12 dpi all other calves were no longer excreting oocysts, except one animal excreting until 17 dpi. Nevertheless these animals continued having altered faecal consistency for several days.
Horses treated for S. neurona infection receive 5.15 mg/kg BW for 5 days, then 10.30 mg/kg BW for 23 days once a day (see U.S. Food and Drug Administration, Navigator®, Idexx Laboratories). In previous experiments other animals such as mice, rats, pigs and cats had been treated with higher dosages of NTZ, such as 100–150 mg/kg BW for 5 days (Blagburn et al., 1998), 50–200 mg/kg BW for 7 days (Li et al., 2003), 125–250 mg/kg for 11 days (Theodos et al., 1998) and 25–75 mg/kg BW for 5–28 days (Gookin et al., 2001). Euzeby et al. (1980) described the use of NTZ in adult dogs, cats and sheep with dosages from 75 to 400 mg/kg BW and observed that general tolerance was excellent. However, in all 3 species a softened faecal consistency with almost sub-diarrhoeic patterns appeared 6–8 h after administration of NTZ. This mild diarrhoea lasted for approximately 24–36 h. No enteric lesions have been observed during postmortem analysis, with the exception of a mild catarrhal inflammation in sheep. The oral administration of NTZ with a dosage of 15 mg/kg BW bid to the calves in this study was twice the usual dosage for human patients with protozoan infections (7.5 mg/kg BW every 12 h for 3 consecutive days as recommended by Romero Cabello et al., 1997), but seemed to be an appropriate dosage for this preliminary study. In clinical studies with paediatric patients (Alinia®, Nitazoxanide 003576, www.mosbysdrugconsult.com/DrugConsult/003576.html), the most frequent adverse events reported, regardless of causality assessment, were abdominal pain (7.8%) and diarrhoea (2.1%), followed by vomiting (1.1%) and headache (1.1%). Nevertheless, symptoms in children were mild and transient, and did not differ significantly from the placebo-group.
Initially the study groups had been designed to consist of 6 animals each. However, the experiment was terminated with groups of 3 animals since these preliminary results did not show the expected positive effect on the course of the Cryptosporidium-infection. On the contrary, the data from the uninfected but treated group suggested that NTZ had a direct negative effect on mucous membranes of gastrointestinal tracts of calves, a problem which was also seen in piglets (Theodos et al., 1998) and cats (Gookin et al., 2001). Our results were supported by Viel et al. (2007), reporting about the use of NTZ (Navigator®, equine paste, 32%, IDEXX Laboratories) in 2–4-day-old kids experimentally infected with C. parvum. An acute toxicity and similar results concerning oocyst excretion and clinical outcome were observed in an experiment with totally 47 animals. The drug was described to be also active against microaerophilic and anaerobic bacteria of the gastrointestinal flora (Euzeby et al., 1980). This could therefore cause significant problems in the gastrointestinal tract of ruminants, which in turn could be more serious than the disease itself (Fox and Saravolatz, 2005, Hemphill et al., 2006). It has been speculated that gastrointestinal problems could be due to the action of the enzyme pyruvate ferredoxin oxidoreductase (PFOR) or other nitro-reductases which have been hypothesized to reduce the thiazole ring-associated nitro group, and thus kill the intestinal bacterial flora through the production of free radicals (Sisson et al., 2002, Hoffman et al., 2007; reviewed in Hemphill et al., 2006). A concomitant C. parvum-infection or other infectious agents damaging the superficial intestinal epithelium may enhance the potential toxicity of treatments substances due to augmented resorption. As a consequence the severity of diarrhoea may increase, augmenting also dehydration and mortality of the animals, particularly of this delicate age group.
This is the first report about the use of NTZ against Cryptosporidiosis in calves. In conclusion, neither prophylactic nor therapeutic use of NTZ in new born calves infected with C. parvum improved clinical appearance in this preliminary experiment or diminished intensity or duration of oocyst excretion. Treatment with NTZ did not show the expected anti-cryptosporidial effect and appeared to fail. Histopathology would be required to support the hypothesis that NTZ has a negative impact on the integrity of the intestinal epithelium or on the normal flora in the calves, but in consideration of animal welfare reasons the results of this study were not sufficiently encouraging to justify the prosecution of experiments.
Acknowledgements
The authors thank Hanspeter Müller for his intensive care of the animals and Dr. Paul Torgerson for his support in statistics. The study was conducted according to ethical guidelines and was approved by the Ethics Committee and the Cantonal Veterinary Office from the Canton of Zürich, permission number 28/2005.
References
- Appelbee A.J., Thompson R.C.A., Olson M.E. Giardia and Cryptosporidium in mammalian wildlife—current status and future needs. Trends Parasitol. 2005;21(8):370–376. doi: 10.1016/j.pt.2005.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blagburn B.L., Drain K.L., Land T.M., Kinard R.G., Moore P.H., Lindsay D.S., Patrick D.A., Boykin D.W., Tidwell R.R. Comparative efficacy evaluation of dicationic carbazole compounds, nitazoxanide, and paromomycin against Cryptosporidium parvum infections in a neonatal mouse model. Antimicrob. Agents Chemother. 1998;42:2877–2882. doi: 10.1128/aac.42.11.2877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broekhuysen J., Stockis A., Lins R.L., De Graeve J., Rossignol J.F. Nitazoxanide: pharmacokinetics and metabolism in man. Int. J. Clin. Pharmacol. Ther. 2000;38(8):387–394. doi: 10.5414/cpp38387. [DOI] [PubMed] [Google Scholar]
- Eckert J., Friedhoff K.T., Zahner H., Deplazes P. Lehrbuch der Parasitologie für Tiermedizin. Enke Verlag; Stuttgart: 2005. Ordnung cryptosporiida. pp. 91–95. [Google Scholar]
- Euzeby J., Prom Tep S., Rossignol J.F. Expérimentation des propriétés anthelminthiques de la nitazoxanide chez le chien, le chat et les ovins. Rev. Méd. Vet. 1980;131:687–696. [Google Scholar]
- Fayer R., Ellis W. Paromomycin is effective as prophylaxis for cryptosporidiosis in dairy calves. J. Parasitol. 1993;79:771–774. [PubMed] [Google Scholar]
- Fayer R., Santin M., Trout J.M., Fayer R., Santin M., Trout J.M. Prevalence of Cryptosporidium species and genotypes in mature dairy cattle on farms in eastern United States compared with younger cattle from the same locations. Vet. Parasitol. 2007;145:260–266. doi: 10.1016/j.vetpar.2006.12.009. [DOI] [PubMed] [Google Scholar]
- Fox L.M., Saravolatz L.D. Nitazoxanide: a new thiazolide antiparasitic agent. Rev. Anti Infect. Agents. 2005;40:1173–1180. doi: 10.1086/428839. [DOI] [PubMed] [Google Scholar]
- Gookin J.L., Levy M.G., Law J.M., Papich M.G., Poore M.F., Breitschwerdt E.B. Experimental infection of cats with Tritrichomonas foetus. Am. J. Vet. Res. 2001;62:1690–1697. doi: 10.2460/ajvr.2001.62.1690. [DOI] [PubMed] [Google Scholar]
- Grinberg A., Markovics A., Galindez J., Lopez-Villalobos N., Kosak A., Tranquillo V.M. Controlling the onset of natural cryptosporidiosis in calves with paromomycin sulphate. Vet. Rec. 2002;151:606–608. doi: 10.1136/vr.151.20.606. [DOI] [PubMed] [Google Scholar]
- Hemphill A., Mueller J., Esposito M. Nitazoxanide, a broad-spectrum thiazolide anti-infective agent for the treatment of gastrointestinal infections. Expert Opin. Pharmacother. 2006;7(7):953–964. doi: 10.1517/14656566.7.7.953. [DOI] [PubMed] [Google Scholar]
- Hilbe M., Arquint A., Schaller P., Zlinsky K., Braun U., Peterhans E., Ehrensperger F. Immunohistochemical diagnosis of persistent infection with Bovine Viral Diarrhoea Virus (BVDV) on skin biopsies. Schweiz. Arch. Tierheilk. 2007;149:337–344. doi: 10.1024/0036-7281.149.8.337. [DOI] [PubMed] [Google Scholar]
- Hoffman P.S., Sisson G., Croxen M.A., Welch K., Harman W.D., Cremades N., Morash M.G. Antiparasitic drug nitazoxanide inhibits the pyruvate oxidoreductases of Helicobacter pylori, selected anaerobic bacteria and parasites, and Campylobacter jejuni. Antimicrob. Agents Chemother. 2007;51(30):868–876. doi: 10.1128/AAC.01159-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarvie B.D., Trotz-Williams L.A., McKnight D.R., Leslie K.E., Wallace M.M., Todd C.G., Sharpe P.H., Peregrine A.S., Jarvie B.D., Trotz-Williams L.A., McKnight D.R., Leslie K.E., Wallace M.M., Todd C.G., Sharpe P.H., Peregrine A.S. Effect of halofuginone lactate on the occurrence of Cryptosporidium parvum and growth of neonatal dairy calves. J. Dairy Sci. 2005;88:1801–1806. doi: 10.3168/jds.S0022-0302(05)72854-X. [DOI] [PubMed] [Google Scholar]
- Joachim A., Krull T., Schwarzkopf J., Daugschies A. Prevalence and control of bovine cryptosporidiosis in German dairy herds. Vet. Parasitol. 2003;112:277–288. doi: 10.1016/S0304-4017(03)00006-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lallemand M., Villeneuve A., Belda J., Dubreuil P. Field study of the efficacy of halofuginone and decoquinate in the treatment of cryptosporidiosis in veal calves. Vet. Rec. 2006;159:672–676. doi: 10.1136/vr.159.20.672. [DOI] [PubMed] [Google Scholar]
- Lefay D., Naciri M., Poirier P., Chermette R. Efficacy of halofuginone lactate in the prevention of cryptosporidiosis in suckling calves. Vet. Rec. 2001;148:108–112. doi: 10.1136/vr.148.4.108. [DOI] [PubMed] [Google Scholar]
- Li X., Brasseur P., Agnamey P., Lemeteil D., Favennec L., Ballet J.J., Rossignol J.F. Long-lasting anticryptosporidial activity of nitazoxanide in an immunosuppressed rat model. Folia Parasitol. 2003;50:19–22. doi: 10.14411/fp.2003.003. [DOI] [PubMed] [Google Scholar]
- Naciri M., Lefay M.P., Mancassola R., Poirier P., Chermette R. Role of Cryptosporidium parvum as a pathogen in neonatal diarrhoea complex in suckling and dairy calves in France. Vet. Parasitol. 1999;85:245–257. doi: 10.1016/S0304-4017(99)00111-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naciri M., Mancassola R., Yvore P., Peeters J.E. The effect of halofuginone lactate on experimental Cryptosporidium parvum infections in calves. Vet. Parasitol. 1993;45:199–207. doi: 10.1016/0304-4017(93)90075-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Donoghue P.J. Cryptosporidium and cryptosporidiosis in man and animals. Int. J. Parasitol. 1995;25:139–195. doi: 10.1016/0020-7519(94)e0059-v. [DOI] [PubMed] [Google Scholar]
- Olson M.E., O’Handley R.M., Ralston B.J., McAllister T.A., Thompson R.C. Update on Cryptosporidium and Giardia infections in cattle. Trends Parasitol. 2004;20:185–191. doi: 10.1016/j.pt.2004.01.015. [DOI] [PubMed] [Google Scholar]
- Potgieter L.N. Immunology of bovine viral diarrhoea virus. Vet. Clin. North Am. Food Anim. Pract. 1995;11:501–520. doi: 10.1016/s0749-0720(15)30464-3. [DOI] [PubMed] [Google Scholar]
- Romero Cabello R., Guerrero L.R., Munoz Garcia M.R., Geyne Cruz A. Nitazoxanide for the treatment of intestinal protozoan and helminthic infections in Mexico. Trans. R. Soc. Trop. Med. Hyg. 1997;91:701–703. doi: 10.1016/s0035-9203(97)90531-9. [DOI] [PubMed] [Google Scholar]
- Santin M., Trout J.M., Fayer R., Santin M., Trout J.M., Fayer R. A longitudinal study of cryptosporidiosis in dairy cattle from birth to 2 years of age. Vet. Parasitol. 2008;155:15–23. doi: 10.1016/j.vetpar.2008.04.018. [DOI] [PubMed] [Google Scholar]
- Sisson G., Goodwin A., Raudonikiene A., Hughes N.J., Mukhopadhyay A.K., Berg D.A., Hoffmann P.S. Enzymes associated with reductive activation and action of nitazoxanide, nitrofurans and metronidazole in Helicobacter pylori. Antimicrob. Agents Chemother. 2002;46:2116–2323. doi: 10.1128/AAC.46.7.2116-2123.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stettler M., Rossignol J.F., Fink R., Walker M., Gottstein B., Merli M., Theurillat R., Thormann W., Dricot E., Segers R., Hemphill A. Secondary and primary murine alveolar echinococcosis: combined albendazole/nitazoxanide chemotherapy exhibits profound anti-parasitic activity. Int. J. Parasitol. 2004;34:615–624. doi: 10.1016/j.ijpara.2004.01.006. [DOI] [PubMed] [Google Scholar]
- Stockdale H.D., Spencer J.A., Blagburn B.L. Prophylaxis and chemotherapy. In: Fayer R., Xiao L., editors. Cryptosporidium and Cryptosporidiosis. CRC Press Inc; Boca Raton, FL: 2008. pp. 255–288. [Google Scholar]
- Theodos C.M., Griffiths J.K., D’Onfro J., Fairfield A., Tzipori S. Efficacy of nitazoxanide against Cryptosporidium parvum in cell culture and in animal models. Antimicrob. Agents Chemother. 1998;42:1959–1965. doi: 10.1128/aac.42.8.1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torgerson P.R., Shaikenov B.S., Rysmukhambetova A.T., Abdybekova A.M., Usenbayev A.E., Baitursinov K.K. Modelling the transmission dynamics of Echinococcus granulosus in cattle and sheep in Kazakhstan. Vet. Parasitol. 2003;114:143–153. doi: 10.1016/s0304-4017(03)00136-5. [DOI] [PubMed] [Google Scholar]
- Tzipori S., Smith M., Halpin C., Angus K.W., Sherwood D., Campbell I. Experimental cryptosporidiosis in calves: clinical manifestations and pathological findings. Vet. Rec. 1983;112:116–120. doi: 10.1136/vr.112.6.116. [DOI] [PubMed] [Google Scholar]
- Viel H., Rocques H., Martin J., Chartier C. Efficacy of nitazoxanide against experimental cryptosporidiosis in goat neonates. Proceedings of the 21st International Conference of the W.A.A.V.P; Gent, Belgium; 2007. p. 490. [DOI] [PubMed] [Google Scholar]
- Ward P.I., Deplazes P., Regli W., Rinder H., Mathis A. Detection of eight Cryptosporidium genotypes in surface and waste waters in Europe. Parasitology. 2002;124:359–368. doi: 10.1017/s0031182001001317. [DOI] [PubMed] [Google Scholar]


