Research highlights
▶ The classical MOG35-55-EAE is complemented by novel cortical and toxic MS models. ▶ Putative autoantigens of EAE/MS include myelin and axonal glycoproteins and lipids. ▶ MS risk genes involve DRB1*1501 and more than 10 novel candidate genes. ▶ Treg and Th17 cells deserve close attention in EAE/MS pathogenesis. ▶ Oral disease-modifiers and monoclonal antibodies show great promise for MS therapy.
Abbreviations: APC, antigen-presenting cell; AT-EAE, adoptive transfer EAE; BBB, blood–brain barrier; BDNF, brain-derived neurotrophic factor; CD, cluster of differentiation; CNS, central nervous system; CNTF, ciliary neurotrophic factor; EAE, experimental autoimmune encephalomyelitis; HLA, human leukocyte antigen; Ig, immunoglobulin; IL, interleukin; IFN, interferon; IVIg, intravenous immunoglobulin; mAb, monoclonal antibody; MBP, myelin basic protein; MHC, major histocompatibility complex; MOG, myelin oligodendrocyte glycoprotein; MP, methylprednisolone; MRI, magnetic resonance imaging; MS, multiple sclerosis; NK, natural killer; ODC, oligodendrocyte; QTL, quantitative trait locus; PLP, proteolipid protein; Tc, cytotoxic T cell; TCR, T cell receptor; TGF, transforming growth factor; Th cell, helper T cell; TNF, tumor necrosis factor
Keywords: Animal model, Autoimmunity, Experimental autoimmune encephalomyelitis, Immunogenetics, Immunomodulatory therapy, Multiple sclerosis
Abstract
Experimental autoimmune encephalomyelitis (EAE) is still the most widely accepted animal model of multiple sclerosis (MS). Different types of EAE have been developed in order to investigate pathogenetic, clinical and therapeutic aspects of the heterogenic human disease. Generally, investigations in EAE are more suitable for the analysis of immunogenetic elements (major histocompatibility complex restriction and candidate risk genes) and for the study of histopathological features (inflammation, demyelination and degeneration) of the disease than for screening of new treatments. Recent studies in new EAE models, especially in transgenic ones, have in connection with new analytical techniques such as microarray assays provided a deeper insight into the pathogenic cellular and molecular mechanisms of EAE and potentially of MS. For example, it was possible to better delineate the role of soluble pro-inflammatory (tumor necrosis factor-α, interferon-γ and interleukins 1, 12 and 23), anti-inflammatory (transforming growth factor-β and interleukins 4, 10, 27 and 35) and neurotrophic factors (ciliary neurotrophic factor and brain-derived neurotrophic factor). Also, the regulatory and effector functions of distinct immune cell subpopulations such as CD4+ Th1, Th2, Th3 and Th17 cells, CD4+FoxP3+ Treg cells, CD8+ Tc1 and Tc2, B cells and γδ+ T cells have been disclosed in more detail. The new insights may help to identify novel targets for the treatment of MS. However, translation of the experimental results into the clinical practice requires prudence and great caution.
1. Introduction
1.1. Origins of EAE: from primates to rodents
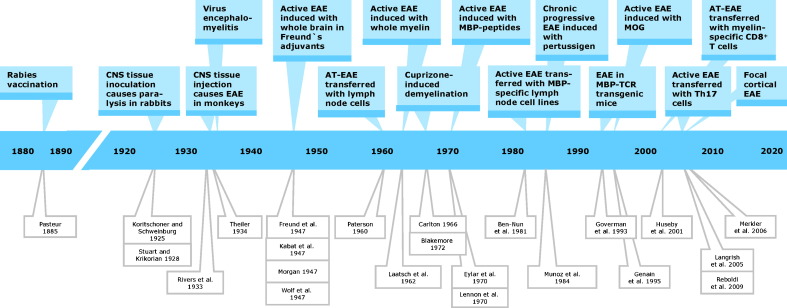
Trials to investigate pathogenetic, diagnostic and therapeutic aspects of multiple sclerosis (MS) in animal models date back to the first half of the 20th century (Lindsey, 2005) (Fig. 1 ). Before, Louis Pasteur's rabies vaccination (Pasteur and Illo, 1996) gave first hints to the possibility that immunization of humans with xenogenic nervous tissue induces ascending paralysis. Specifically, inoculation of rabies patients with desiccated spinal cord of rabies-infected rabbits caused pareses of limb, neck and facial muscles resulting in gait, swallowing and breathing problems (Baxter, 2007). Conversely, it was shown by Koritschoner and Schweinburg (1925) and Stuart and Krikorian (1928) that injection of human spinal cord or sheep brain into rabbits leads to limb paralysis (clumsy gait and muscle weakness). Rivers et al. (1933) first demonstrated in monkeys immunized with rabbit brain or brain extracts that paralysis was associated with perivascular infiltrates and demyelination in the brain and spinal cord. He called the disease acute disseminated encephalomyelitis, a term that was later changed to experimental allergic or autoimmune encephalomyelitis (EAE). Since the frequency and severity of paralyses was correlated to the titer of anti-brain antibodies, researchers in subsequent trials boosted the humoral immune response with Freund's adjuvant (CFA) (Freund and McDermott, 1942), later complemented by pertussis toxin (Munoz et al., 1984). Thereby they could induce oscillatory symptoms and relapsing–remitting courses of the disease accompanied by perivascular leukocyte infiltration in acute lesions and gliosis in chronic lesions both reminiscent of MS pathology (Wolf et al., 1947). Experiments were performed first in guinea pigs (Freund et al., 1947) and monkeys (Kabat et al., 1947, Morgan, 1947, Wolf et al., 1947) and later in various other species including mice (Olitzky and Yager, 1949) and rats (Lipton and Freund, 1952) enabling more extensive immunogenetic, histopathological and therapeutic studies. It turned out that the histopathology and the clinical course of the disease varied significantly reflecting in part the heterogeneity of its human counterpart dependent on the genetic background of the animals, the source of the antigenic material and the mode of application of the antigen (Hartung et al., 2005, Olsson et al., 2000, Wekerle et al., 1994).
Fig. 1.
Timeline of milestones in the history of animal models of MS. Abbreviations: AT-EAE, adoptive transfer EAE; CD, cluster of differentiation; CNS, central nervous system; MBP, myelin basic protein; MOG, myelin oligodendrocyte glycoprotein; TCR, T cell receptor; Genain et al., 1995, Huseby et al., 2001 T helper cell.
1.2. Different types of EAE
Importantly, by stepwise reduction of the complexity of the antigenic material from crude brain tissue and protein extracts through various central myelin proteins such as
-
(i)
myelin basic protein (MBP) (Einstein et al., 1962, Laatsch et al., 1962),
-
(ii)
myelin oligodendrocyte (ODC) glycoprotein (MOG) (Lebar et al., 1986),
-
(iii)
proteolipid protein (PLP) (Tuohy et al., 1988),
-
(iv)
myelin-associated oligodendrocytic basic protein and 2′,3′-cyclic nucleotide 3′-phosphodiesterase (Määttä et al., 1998)
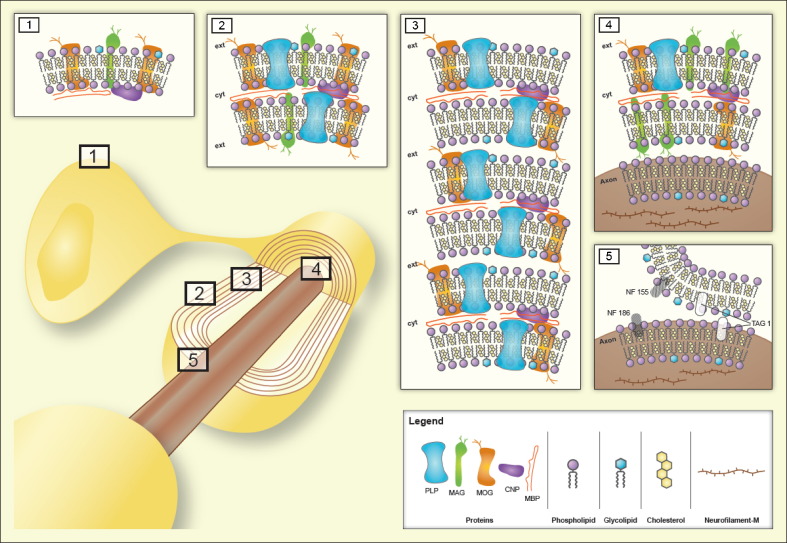
to small encephalitogenic peptides (Eylar et al., 1970, Lennon et al., 1970) such as MBP1–37, MBP1–11, MBP1–9, MBP83–99, MOG55–75 and PLP139–151, more reproducible EAE models became available that mirror different features of MS (Wekerle et al., 1994). More recently a variety of additional antigens have been supposed to be involved in autoimmune reaction in MS and EAE (Table 1 ). Some of them are myelin constituents such as neurofascin NF 155 (Mathey et al., 2007), others are expressed on myelin and axons such as contactin-2/transient axonal glycoprotein-1 (TAG-1) (Derfuss et al., 2009) and some are entirely non-myelin antigens such as the neuronal membrane protein neurofascin NF 186 (Mathey et al., 2007), the neuronal cytoskeletal protein neurofilament-M (Krishnamoorthy et al., 2009) and the astrocyte-typical Ca2+-binding protein S100β (Kojima et al., 1997). The neurofascins NF 155 and NF 186 and the adhesion molecule contactin-2/TAG-1have been identified as putative MS auto-antigens by a proteomics-based screening approach of MS sera and were subsequently shown to promote the autoimmune pathogenesis of EAE in rat models (Derfuss et al., 2009, Derfuss et al., 2010, Mathey et al., 2007). Antibodies to neurofascins, in particular to NF 186, caused axonal injury without enhancing inflammation and demyelination in MOG–EAE (Mathey et al., 2007). In contrast to MOG–EAE, contactin-2/TAG-1-specific T cells induced inflammatory lesions preferentially in the cerebral cortex and the spinal cord white and gray matter (Derfuss et al., 2009). However, while these cells were unable to cause demyelination by themselves they opened the blood–brain barrier (BBB) thereby allowing access of anti-MOG antibodies to the central nervous system (CNS) where they could trigger demyelination. The EAE variants induced by axonal antigens may reflect special features of MS subtypes, e.g. those characterized by cortical lesions or by histological patterns II and IV according to the classification of Lucchinetti et al. (2000). The schematic drawing in Fig. 2 indicates the molecular localisation of currently known putative auto-antigens in EAE. Proteins, glycoproteins and lipoproteins possessing encephalitogenic epitopes may be exposed to the outer surface of ODCs (1) or myelin (2) or they may reside in the compact myelin zone (3), at the myelin–axon interface (4) or the node of Ranvier (5). As to the last localisation the three adhesion molecules NF 155 and NF 186 contactin-2/TAG-1 are differentially expressed. Whereas NF 155 is localised at the paranodal myelin loop and interact with contactin-1/F3 and the contactin-associated protein 2 (caspr 2) on the axoplasm in a ternary complex (Charles et al., 2002), NF 186 is exposed in the non-myelinated part of the node of Ranvier and interacts with the neuron-glia-related cell adhesion molecule (NrCAM) and voltage-gated Na+ channels (Hedstrom et al., 2007). Contactin-2/TAG-1 is also expressed juxtaparanodal by both the myelin and axonal membrane forming dimers or molecular zippers (Mörtl et al., 2007, Shimoda and Watanabe, 2009). Whether lipids, glycolipids and phospholipids play also a role as auto-antigens in EAE and MS is not clear, although several evidences from experimental and clinical studies support such a role (Podbielska and Hogan, 2009). Candiate myelin lipid auto-antigens in MS and/or EAE are shown in Table 1. In a lipid microarray study of Kanter et al. (2006) co-immunization with sulfatides and co-application of sulfatide-specific antibodies worsened the clinical course of PLP139–151–EAE in SJL/J mice, and Quintana et al. (2008b) found auto-antibodies to lipids such as oxidized cholesterol derivatives in the serum of MS patients with the immunopathologic pattern II according to Lucchinetti et al. (2000). However, the oxidized cholesterol derivatives did not affect the specific humoral and T cell response in MOG55–75–EAE.
Table 1.
Putative protein and lipid auto-antigens in EAE and/or MS.
| Antigens | Results in MS and/or EAE | References |
|---|---|---|
| Myelin basic protein | T and B cell response in EAE and MS | Einstein et al. (1962), Laatsch et al. (1962) |
| MOG | T and B cell response in EAE and MS | Lebar et al. (1986) |
| PLP | T and B cell response in EAE and MS | Tuohy et al. (1988) |
| 2′,3′-CNP | T and B cell response in EAE and MS | Määttä et al. (1998) |
| NF 155 | Antibodies recognize the extracellular domain in MS and cause axonal injury in EAE, but only in preexisting demyelinated lesions | Charles et al. (2002), Derfuss et al. (2010), Mathey et al. (2007) |
| NF 186 | Antibodies recognize the extracellular domain in MS, inhibit axonal conduction in a complement-dependent manner and cause axonal injury in EAE | Derfuss et al. (2010), Hedstrom et al. (2007), Mathey et al. (2007) |
| Neurofilament-M | Neurofilament-M-specific T cells induce severe clinical EAE with confluent demyelination and massive axonal loss | Krishnamoorthy et al. (2009) |
| Contactin-2/TAG-1 | Contactin-2/TAG-1-specific T cells induce inflammatory lesions in the cortex and white and gray matter thereby opening locally the BBB and causing occasionally clinical EAE | Derfuss et al., 2009, Derfuss et al., 2010, Mörtl et al. (2007), Shimoda and Watanabe (2009) |
| S100β | Strong T cell response in EAE | Kojima et al. (1997) |
| Phosphatidylserine | Promotion of demyelination in marmoset EAE | Ohler et al. (2004) |
| Sulfatides | T and B cell response in EAE | Kanter et al. (2006) |
| Oxidized phosphatidylcholine | Strong antibody reactivity in MS brain and EAE spinal cord | Qin et al. (2007) |
| Ganglioside GM1, sulfatide and galactosylceramide | Increased reactivity of pro-inflammatory cytokine secreting CD8+ T cells in MS patients | Shamshiev et al. (1999) |
| Gangliosides GM3 and GQ1b | Increased T cell response in primary progressive MS patients | Pender et al. (2003) |
| Ganglioside GD1a | Increased antibodies in serum and cerebrospinal fluid of patients with MS and optic neuritis | Matà et al. (1999) |
| Lactosylceramide and l-α-lysophosphatidylserine | Strong antibody reactivity in serum and cerebrospinal fluid of MS patients | Kanter et al. (2006), Quintana et al. (2008b) |
Fig. 2.
Putative auto-antigens in EAE with indication of their preferential localisation. Insets refer to the ODC membrane (inset 1), myelin surface zone (inset 2), compact myelin zone (inset 3), myelin/axon interface zone (inset 4), and nodal and paranodal zone of node of Ranvier (inset 5). Abbreviations: CNP, 2′,3′-cyclic nucleotide-3′-phosphodiesterase; cyt, cytoplasm; ext, extracellular space; MAG, myelin-associated glycoprotein; MBP, myelin basic protein; MOG, myelin oligodendrocyte glycoprotein; NF, neurofascin; PLP, proteolipid protein; TAG, transient axonal glycoprotein.
Several studies have shown that actively induced EAE models can reproduce the typical temporal maturation of MS lesions from inflammation with or without deposition of immunoglobulin through demyelination and axonal damage to gliosis and partial remyelination. However, special phenotypes of MS pathology such as primary neuronal degeneration, shift of CD4+ T helper cells to CD8+ cytotoxic T cells and lesions in cortical areas are rarely reproduced by actively induced EAE models (Aktas et al., 2007, Gold et al., 2006, Herrero-Herranz et al., 2008, Linker et al., 2005, Pomeroy et al., 2005). Hitherto it is not clear to which extent cortical lesions account for the brain atrophy in MS. Merkler et al. (2006) could induce focal demyelinating lesions in the cortex of MOG–EAE rats by stereotactical injection of the pro-inflammatory cytokines tumor necrosis factor-α (TNF-α) and interferon-γ (IFN-γ). Corresponding to findings in MS the focal cortical lesions were rapidly remyelinated. Accordingly, in marmoset MOG–EAE focal cortical lesions were not the major cause of diffuse cortical atrophy (Pomeroy et al., 2008). The potential pathomechanisms of CNS atrophy in MS are complex and may include several mechanisms of neuronal damage such as anterograde Wallerian degeneration, neuronal dying-back and neuronal soma and dendritic shrinkage (Dziedzic et al., 2010, Siffrin et al., 2010). Currently, disclosure of the exact mechanisms of axon damage is a major challenge of MS research, since it appears to be the main cause of clinical disability and may be the result of immune and/or non-immune attacks on neurons rather than a consequence of immune-mediated demyelination (Siffrin et al., 2010).
Concerning the aetiopathogenesis of MS the role of infections is still a matter of controversy. In mice lymphocytic choriomenigitis virus protein expression could elicit chronic autoimmune inflammation and demyelination in the CNS (Evans et al., 1996). A Chlamydia pneumonia-specific peptide sharing an immunodominant epitope with MBP-induced severe clinical and histological EAE (Lenz et al., 2001), whereas intestinal parasites conveyed resistance to EAE in EAE-susceptible Lewis rats (Zorzella et al., 2007). Also, human herpes virus type 6, Epstein–Barr virus, measles virus and retroviruses have been implicated in the aetiopathogenesis of MS, but currently available experimental and clinical data are not convincing enough to justify anti-viral or antibiotic therapy in MS. However, experimental demyelinating diseases induced by Theiler's virus (Theiler, 1934, Ure and Rodriguez, 2005), coronavirus (Lavi, 2005) or canine distemper virus (Seehusen and Baumgärtner, 2010) are suitable models for the investigation of some aspects of MS pathology.
Milestones of the development of increasingly specific EAE models were (Fig. 1):
-
(i)
Induction of EAE by transfer of total lymph node cells (Paterson, 1960), isolated MBP-specific T cell-line cells (Ben-Nun et al., 1981) or interleukin-23 (IL-23)-dependent PLP-specific CD4+ T helper IL-17 (Th17) cells (Langrish et al., 2005) into naive rats or mice, respectively, establishing distinct forms of adoptive transfer EAE (AT-EAE) and
-
(ii)
generation of transgenic mice with deletion (knock-out leading to loss-of-function) or over-expression (knock-in leading to gain of function) of pathogenetically relevant genes (reviewed in Krishnamoorthy et al., 2007). Examples of such genes are those encoding T cell receptors (TCRs) (Bettelli et al., 2006, Goverman et al., 1993, Lafaille et al., 1994, Mendel et al., 2004), major histocompatibility complex (MHC) molecules (Friese et al., 2008, Khare et al., 2005, Linker et al., 2005, Mangalam et al., 2008), cytokines (reviewed in Campbell et al., 1998, Owens et al., 2001) as well as neurotrophic factors (Linker et al., 2005, Linker et al., 2008a, Linker et al., 2009b, Mirowska-Guzel, 2009) and their receptors (Dallenga et al., 2009, Linker et al., 2009b).
The constitutive knock-out or knock-in of cytokine genes throughout the whole development and adulthood of an animal implies serious drawbacks due to redundancy and feed-back loops of cytokine signal pathways (Owens et al., 2001, Steinman, 1997). This constraint can been overcome by approaches with inducible spatially and temporally restricted gene targeting, which has been only recently introduced for studies directed to the CNS (Gavériaux-Ruff and Kieffer, 2007, Hövelmeyer et al., 2005).
Despite the obvious importance of autoimmune processes in the pathogenesis of MS, there is evidence for a non-immune origin of at least some subtypes of MS. For example, even aggressive immunosuppression is not sufficient to treat progressive MS. Therefore, additional experimental models are needed, especially for the study of non-immune-mediated demyelination and axonal loss via ODC degeneration. For this purpose a toxic model of demyelination has been developed that is based on the selective toxicity for ODCs of the copper chelator biscyclohexanone oxaldihydrazone (cuprizone) (Blakemore, 1972, Carlton, 1966). If young mice are fed with cuprizone, focal demyelination occurs predominantly in the cerebellar cortex and peduncle. After withdrawal of the toxin spontaneous remyelination can be seen predominantly in the rostral regions (Skripuletz et al., 2010, Torkildsen et al., 2008). Thereby the cuprizone-model correlates well with histopathological features of MS, especially in the subtype or variant classified as histological pattern III according to Lucchinetti et al. (2000), which renders it a useful tool for MS research (Einstein et al., 2009, Kipp et al., 2009). Paradoxically, according to a recent report of Herder et al. (2009) cuprizone ameliorates Theiler's murine encephalomyelitis suggesting that it might have immunomodulatory and/or anti-viral properties in addition to its toxic effects.
1.3. Current EAE models
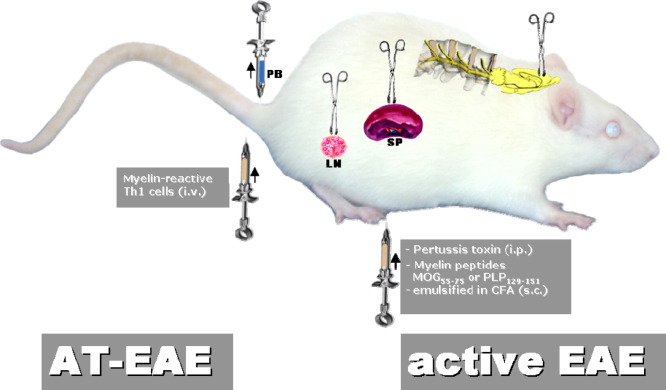
Currently, the most common mode of EAE induction is based on the injection of an encephalitogenic peptide, mostly MOG35–55 or PLP139–151, which is emulsified in CFA containing mineral oil and Mycobacterium tuberculosis strain H37RA, followed by intraperitoneal injections of pertussis toxin (Fig. 3 ). The resulting phenotype depends mainly on the antigen source and the genetic background of the animal species and strains used. For example, PLP139–151 induces a relapsing–remitting EAE in SJL mice, whereas MOG35–55 triggers chronic-progressive EAE in C57BL mice that are the most favored mice for transgenic experiments (Gold et al., 2006). Crossing of C57BL mice, which over-express MOG-TCR and MOG-specific B cells, resulted in a severe form of EAE that closely replicated Devic's variant of MS with inflammatory lesions of optic nerves and spinal cord (Bettelli et al., 2006). MOG-TCR transgenic mice backcrossed to SJL/J background develop a relapsing–remitting form of EAE with episodes altering between optic nerve, cerebellum and spinal cord. Evolution of this model depends on an intact B cell compartment. Apparently, MOG-TCR transgenic T cells expand endogenous auto-reactive B cells that manufacture pathogenic demyelinating auto-antibodies to a conformational epitope on native MOG protein whilst not recognizing the T cell target MOG peptide (Pöllinger et al., 2009). The authors claim to have generated the first spontaneous animal model for relapsing–remitting MS. Current types of AT-EAE include those induced by Th17 cells suggesting that these newly detected effector cells may in part be responsible for the pathological heterogeneity of MS lesions (Afzali et al., 2007, Gold and Lühder, 2008, Hofstetter et al., 2007, Hofstetter et al., 2009, Jäger et al., 2009, Korn et al., 2007, Quintana et al., 2008a, Reboldi et al., 2009). All EAE models are directly accessible to investigation of the immune and nervous system (Fig. 3), which interact during the pathogenesis of the disease and which are both targeted by established and experimental therapies.
Fig. 3.
Most common animal models of MS with indication of the compartments investigated for analysis of systemic and local disease processes. For active immunization antigens are preferentially applied to the flank or toe pad of the animal, since draining lymph nodes of these areas mediate a highly effective systemic immune response to the putative auto-antigens as a first step for induction of autoimmune processes in the CNS. Abbreviations: AT-EAE, adoptive transfer EAE; CFA, complete Freund's adjuvant; i.p., intraperitoneal; i.v., intravenous; LN, lymph node; MOG, myelin oligodendrocyte glycoprotein; PB, peripheral blood; PLP, proteolipid protein; s.c., subcutaneous; SP, spleen, Th1 cells, T helper type 1 cells.
2. Investigation of immunogenetic and pathogenetic features
2.1. Immunogenetics of EAE and MS
Since MS appears to be a polygenetically determined disease, efforts have been undertaken by linkage and association studies to define chromosomal regions, quantitative trait loci (QTL), that control the susceptibility to the disease and to compare these QTL with the susceptibility loci in the animal model EAE (Serrano-Fernández et al., 2004). The high frequency of intergenomic EAE/MS consensus genes supports the value of EAE for studying of MS features. Backcross (offspring-parent mating) and intercross (sibling mating) experiments with EAE susceptible and resistant animal strains of mice, rats, guinea pigs and hamsters revealed mainly MHC-linked QTL (Encinas et al., 1996, Olsson et al., 2000). Combined-cross analysis could enhance the detection of QTL with moderate effects in rats (Jagodic and Olsson, 2006). Genotyping of 150 microsatellite markers in F2 intercross populations of EAE-susceptible SJL/L mice and EAE-resistant C57BL/10.S mice identified QTL linked to increased latency of cortical motor evoked potentials in non-immunized animals, which correlated with earlier onset of the disease (Mazón Peláez et al., 2005). This finding points to a role of myelin composition and synaptic transmission in susceptibility to EAE and provides a chance to detect individuals with high risk for autoimmune demyelination by QTL analysis before the disease is manifested. In MS, some immune response-related genes have been identified by genome-wide association studies using single nucleotide polymorphism analysis with microarray technique as being heritable risk factors of the disease, although their individual contribution is clearly modest with odds ratios for most not exceeding 1.2 (IMSGC, 2007). These genes include the interleukin (IL)-2α and IL-7α receptor genes on chromosomes 10 and 5, respectively, and some human leukocyte antigens (HLA) belonging to MHC class II molecules on chromosome 6 (Table 2 ) (Gregory et al., 2007, Hafler et al., 2007). More recently, the genes encoding the following proteins have been confirmed as novel MS risk genes (explanation of abbreviations in Table 2) (Aulchenko et al., 2008, Dabbeekeh et al., 2007, De Jager et al., 2009a, De Jager et al., 2009b, Hafler et al., 2009, Hoppenbrouwers et al., 2008, Hoppenbrouwers et al., 2009, Hafler et al., 2007, Johnson et al., 2010, Mero et al., 2010, Rubio et al., 2008, Sarrias et al., 2007):
-
-
EV15, CD58, KIF1B, RGS1 and RPL5 (on chromosome 1),
-
-
IL-12A (on chromosome 3),
-
-
PTGER4 (on chromosome 5),
-
-
OLIG3-TNFAIP3 (on chromosome 6),
-
-
CD6 (on chromosome 11),
-
-
TNFRSF1A (on chromosome 12),
-
-
IRF8 and CLEC16A (on chromosome 16),
-
-
CD226 (on chromosome 18), and
-
-
TYK2 (on chromosome 19).
Table 2.
Susceptibility genes of MS.
| Gene | Function | References |
|---|---|---|
| HLA-DRB1*1501 | Antigen presentation | Barcellos et al. (2006), Chao et al. (2008), IMSGC (2007), Khare et al. (2005) |
| IL-2α receptor | T and B cell activation | IMSGC (2007) |
| IL-7α receptor | T cell survival, differentiation and homeostasis, B cell development | Gregory et al. (2007), IMSGC (2007) |
| EV15 | GTPase activation | Dabbeekeh et al. (2007), Hoppenbrouwers et al. (2008) |
| CD58 (lymphocyte-associated antigen 3, LFA-3) | Ligand of CD2, costimulatory molecule for T cells enhancing FoxP3 expression in Treg cells | De Jager et al. (2009a), Hoppenbrouwers et al. (2009) |
| CLEC16A (C-type lectin domain family 16, member A) | Unknown function, but highly expressed in dendritic cells, B cells and NK cells… | Hoppenbrouwers et al. (2009), Johnson et al. (2010), Rubio et al. (2008) |
| CD6 | ||
| Bacterial molecular pattern recognition and suppressing TNF-α, IL-6 and IL-1β | De Jager et al. (2009b), Sarrias et al. (2007) | |
| IRF8 (IFN regulatory factor 8) | Activation or repressing of IFN type I transcription | De Jager et al. (2009b), Johnson et al. (2010) |
| TNFRSF1A (tumor necrosis factor receptor superfamily, member 1A) | Pro-inflammatory and proapoptotic activity | De Jager et al. (2009b) |
| OLIG3-TNFAIP3 (oligodendrocyte transcription factor 3—NF-α-induced protein 3) | Development of neuronal cells, tumor suppression and anti-inflammation | De Jager et al. (2009b) |
| IL-12A (IL-12p35) | Growth factor for activated T and NK cells, enhancing the lytic activity of NK/lymphokine-activated killer cells | Rubio et al. (2008) |
| PTGER4 (prostaglandin E receptor 4) | Activation of T cell factor signaling | De Jager et al. (2009b) |
| RGS1 (regulator of G-protein signaling 1) | B cell activation | Johnson et al. (2010) |
| TYK-2 (tyrosine kinase 2) | Intracellular signal transduction of type I IFNs | Johnson et al. (2010), Mero et al. (2010) |
| CD226 | Intercellular adhesion, lymphocyte signaling, cytotoxicity and lymphokine secretion mediated by cytotoxic T cells and NK cells | Hafler et al. (2009) |
| KIF1B (kinesin family member 1B) | Motor protein transporting mitochondria and synaptic vesicle precursors | Aulchenko et al. (2008) |
| RPL5 (ribosomal protein L5) | Transport of nonribosome-associated cytoplasmic 5S rRNA to the nucleolus for assembly into ribosomes. | Rubio et al. (2008) |
The strongest association to MS susceptibility, although not to the age of onset and severity of the disease, was found for the HLA-DRB1*1501 allel (Barcellos et al., 2006, Chao et al., 2008), whereas transgenic mice over-expressing the human HLA-DRB1*1502 allel developed a severe MOG–EAE (Khare et al., 2005). In contrast, genes related to antigen processing can also slow down disease progression as reflected by a milder course of the disease in MOG35–55–EAE of congenic NOR/LtJ mice compared to the wild-type NOD mice (Mayo and Quinn, 2007). Similarly, congenic mapping of the rat genome confirmed that a chromosomal region homologous to a human MS susceptibility region confers protection against MOG–EAE (Jagodic et al., 2001). Also, transgenic MOG35–55–EAE mice over-expressing the TCR for MOG35–55 showed protective rather than pathogenic effects (Mendel et al., 2004). In contrast, transgenic mice over-expressing MBP-specific TCR developed high frequency spontaneous EAE (Goverman et al., 1993, Lafaille et al., 1994). The relevance of these findings for the human disease remains to be elucidated. Research in EAE also yielded hints for the existence of chromosomal loci that control disease susceptibility in dependence of age and season pointing to a role of chronobiology in autoimmunity (Teuscher et al., 2006). Moreover, EAE susceptibility can vary even between different colonies of the same animal strain as demonstrated for Lewis rats purchased from different animal facilities (Gould et al., 1994). Since the genetic background of these animals is almost identical, other mechanisms, preferentially those involving gene regulating, may be responsible for differences in disease susceptibility and progression. Transcriptome and proteome analyses are powerful new tools for the elucidation of those mechanisms (Elkabes and Li, 2007, Fernald et al., 2005, Goertsches and Zettl, 2007, Ibrahim et al., 2001, Ibrahim and Gold, 2005).
2.2. Gene expression studies by microarray analysis
An integrated analysis of available data from genome-wide genetic screens and high-throughput gene expression studies in MS and EAE revealed that differentially expressed genes appear mainly in clusters rather than in uniform distribution throughout the genome (Fernald et al., 2005). The hereby included hypothesis-neutral gene expression studies are mainly performed with microarray technique (RNA profiling) and applied in MS patients to peripheral blood mononuclear cells or brain tissue and in EAE animals to lymphatic and nervous tissue (Goertsches and Zettl, 2007, Tajouri et al., 2007). They deliver an increasing wealth of data, which implies a great challenge for bioinformatic analysis that may include pathway analyses using background information from the Gene Ontology project and other data bases as well as data mining approaches. Results of gene expression studies are generally hampered by methodical problems such as the heterogeneity of the material from different individuals and the difficulties in defining a reasonable threshold for differentially expressed genes. Nevertheless, meaningful data have already been obtained from expression studies in EAE that point to new EAE-QTL and susceptibility genes, dissect new pathogenic and protective pathways and identify new therapeutic targets (Comabella and Martin, 2007, Jelinsky et al., 2005, Matejuk et al., 2003, Mazón Peláez et al., 2005, Mix et al., 2002, Mix et al., 2006, Paintlia et al., 2004). As an example, the comparison of the EAE-resistant strain C57BL/10.S with the EAE-susceptible strain C57BL/6 in the MOG35–55–EAE model strongly supported EAE-resistance to be an active process revealing new target genes for therapeutic intervention (Mix et al., 2004). Moreover, data of Baranzini et al. (2005) derived from the MOG35–55–EAE model led to new conclusions such as
-
(i)
early non-specific BBB impairment (mainly neutrophil-related) secondary to immunization with CFA,
-
(ii)
transition from innate to adaptive immune responses before onset of EAE,
-
(iii)
identification of at least 3 discrete EAE phases (early EAE, peak EAE and early recovery) with characteristic gene expression patterns, and
-
(iv)
early neuronal damage.
Therefore, gene expression studies can provide new insights into the dynamics of discrete early and progressive phases of EAE on the transcriptional level, which are consistent with the histological and clinical phenotype. They can serve as a tool for fingerprinting of individual disease processes in man. As a practical consequence new therapeutic targets surface. RNA profiling has also enabled researchers to identify genes which support maintenance of the hematopoietic stem cell niche synapse as a source for therapeutic mesenchymal stem cells that can ameliorate EAE (Pedemonte et al., 2007). On the other hand, EAE can also serve as a tool to validate new targets derived from gene-microarray analysis (Lock et al., 2002).
Despite the clear benefit of the transcriptome approach there are voices advising caution concerning overinterpretation of results of global gene expression analysis. In any case, due to its obvious limitations transcriptomics should be employed only with a clear and specific question in mind and carefully in view of the regulatory settings and potential pitfalls of the technique (Casciano and Woodcock, 2006, Fathallah-Shaykh, 2005). The same is true for the proteome approach (reviewed in Elkabes and Li, 2007, Linker et al., 2009a). In EAE, it has served to generate differential protein expression profiles (Duzhak et al., 2003, Liu et al., 2007), to monitor the diversity of autoantibody responses (Robinson et al., 2003) and to validate new therapeutic targets derived from laser-capture micro-dissections of MS lesions (Han et al., 2008). Also putative lipid auto-antigens could be identified by the microarray approach in MS and EAE (Kanter et al., 2006, Quintana et al., 2008b).
2.3. Immunopathogenesis
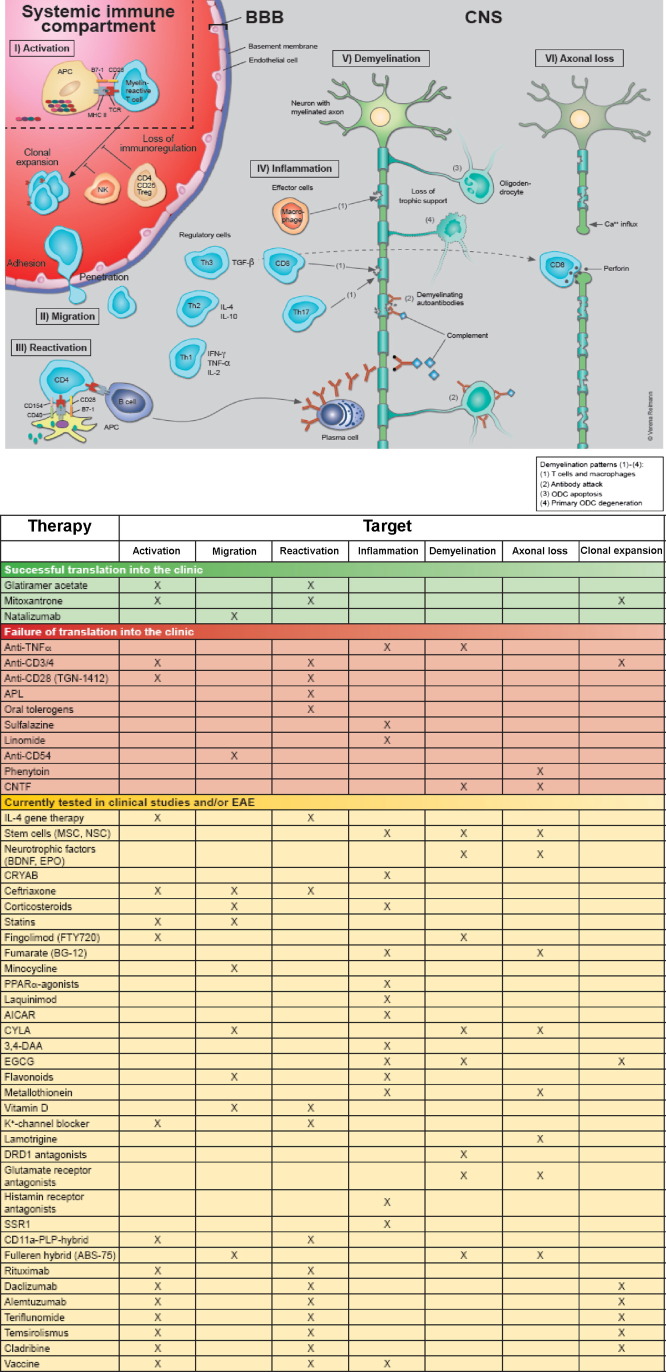
There is still consensus amongst most researchers that immunological processes play a pivotal role in the pathogenesis and progression of MS (Gold et al., 2006, Linker et al., 2008c, Steinman and Zamvil, 2006, Weiner, 2009), although the aetiological factors may vary between different subtypes of MS and may not primarily affect the immune system, especially in cases with the histopathologic patterns III and IV according to Lucchinetti et al. (2000). Moreover, in early MS with histologic patterns I–III Wallerian degeneration seems to contribute significantly to axonal loss in the plaques and periplaque white matter (Dziedzic et al., 2010). The current concept of pathogenetic processes in MS is schematically depicted in Fig. 4 , which indicates also therapeutic interventions and their putative targets. Recent paradigm shifts relate to the recognition of new roles for CD8+ T cells (Friese and Fugger, 2009), B cells (Franciotta et al., 2008), innate immunity (Weiner, 2009) and emerging pathogenic pathways causing neuronal damage (Aktas et al., 2010, Centonze et al., 2010, Dziedzic et al., 2010, Herz et al., 2009).
Fig. 4.
Putative pathogenic mechanisms of MS. Auto-reactive lymphocytes may be recruited from peripheral lymphoid organs and after migration through the BBB reactivated in the CNS, where an inflammatory cascade is initiated leading to subsequent damage of myelin and axons. Alternatively, primary oligodendroglial and axonal degeneration may be followed by an inflammatory autoimmune process. The adjacent table depicts the putative pathogenic processes that are targeted by established and experimental therapies. Treatments are grouped according to the contribution made by EAE to their development, i.e. they are either successfully translated into the clinic (green), only successful in EAE (red) or currently tested in EAE and/or MS (yellow). Abbreviations: AICAR, 5-aminoimidazole-4-carboxamide-1-β-d-ribofuranoside; APC, antigen-presenting cell; APL, altered peptide ligand; BBB, blood–brain barrier; BDNF, brain-derived neurotrophic factor; CD, cluster of differentiation; CNS, central nervous system; CNTF, ciliary neurotrophic factor; CRYAB, αB-crystallin; CYLA, Calpain inhibitor; 3,4-DAA, N-(3,4,-dimethoxycinnamoyl) anthranilic acid; DRD1, dopamine receptor type 1; EGCG, (−)-epigallocatechin-3-gallate; IL, interleukin; IFN-γ, interferon-γ; major histocompatibility complex; MOG, myelin oligodendrocyte glycoprotein; MP, methylprednisolone; MRI, magnetic resonance imaging; NK, natural killer; ODC; oligodendrocyte; PLP, proteolipid protein; PPAR-α, peroxisome proliferator-activated receptor-α; SSRI, selective serotonin reuptake inhibitor; Tc, cytotoxic T cell; TCR, T cell receptor; TGF-β, transforming growth factor-β; Th cell, helper T cell; TNF-α, tumor necrosis factor-α.
EAE models have considerably contributed to our understanding of immune regulatory processes in the pathogenesis of MS (Gold et al., 2006, Wekerle et al., 1994). While traditionally regulatory (suppressive) activity in autoimmune processes was primarily attributed to CD8+ T cells (Zozulya and Wiendl, 2008), more recently CD4+CD25+FoxP3+ regulatory T cells (Yi et al., 2006) have attracted interest in this respect (Ephrem et al., 2008, Paintlia et al., 2008, Tischner et al., 2006). They seem to be important as antagonists of CD4+ Th17 cells (Afzali et al., 2007, Littman and Rudensky, 2010, Quintana et al., 2008a) that are supposed to be effector cells in EAE (Hofstetter et al., 2007, Huppert et al., 2010, Liu et al., 2010, Steinman, 2007) and MS (Gold and Lühder, 2008). However, the pathogenic role of Th17 cells and of IL-17 in EAE is controversial. According to findings of Haak et al. (2009) in transgenic mice the in vivo function of IL-17 in the CNS may be redundant and the members of the IL-17 family IL-17A and IL-17F may contribute only marginally to the autoimmune pathogenesis of MOG35–55–EAE. On the other hand, Huppert et al. (2010) found that IL-17A promotes breakdown of the BBB, a crucial step in the development of EAE. Moreover, findings of Nowak et al. (2009) point to a pro-inflammatory role of the Th17-derived IL-9 in MOG35–55–EAE. Th17 cells are driven by IL-21 (Korn et al., 2007) and IL-23 (Langrish et al., 2005, McKenzie et al., 2006) and suppressed by IL-27 (Fitzgerald et al., 2007, Wang et al., 2008). Recently, the IL-7-IL-7R pathway has been implicated in the survival and expansion of effector/memory Th17 cells. Blockade of IL-7R rendered differentiated Th17 cells susceptible to apoptosis which led to attenuation of MOG–EAE (Liu et al., 2010). Corresponding IL-17+ CD8+ Tc cells show impaired cytotoxicity and IFN-γ production, but may through their excessive IL-17 production contribute to inflammatory processes in EAE and MS (Huber et al., 2009). Recently, Sobottka et al. (2009) could demonstrate in brain slices of transgenic mice that myelin-directed CD8+ Tc cells cause extensive damage not only of the myelin sheath, but also of the axons. Consequently, these cells may contribute to axonal loss in EAE and probably also in MS with the immunopathologic pattern I according to Lucchinetti et al. (2000).
B cells are involved in the immunopathogenesis of MS and EAE at least by two functional activities, i.e. as antigen-presenting cells (APCs) and as antibody producers (Franciotta et al., 2008, Goverman, 2009, Hohlfeld et al., 2008, Martin Mdel and Monson, 2007, Weiner, 2009, Ziemssen and Ziemssen, 2005). In addition they may co-stimulate T cells, facilitate recruitment of inflammatory cells to the CNS and myelin opsonization, but also promote remyelination, and take part in immunoregulatory processes. Anti-myelin antibodies are supposed to be major components of the histopathologic MS pattern II according to Lucchinetti et al. (2000). A special role as targets for antibodies in distinct types of EAE and MS is ascribed to the myelin molecules MOG (Haase et al., 2001) and the axonal molecule neurofascin (Hohlfeld et al., 2008).
Another distinct lymphocyte subpopulation that has been implicated in CNS autoimmunity is the γδ+ T cell subset bearing TCRγδ. These fetal-type T cells are increased in number in MS cerebrospinal fluid (Mix et al., 1990) and may exert pathogenic and regulatory functions (reviewed in Blink and Miller, 2009 and Goverman, 2009). Interestingly, the majority of IL-17 producing host cells in a Th1-mediated AT-EAE belonged to the γδ+ T cell type (Lees et al., 2008).
Adding to the complexity, there is an obvious pathogenic role for EAE and MS of innate immunity mediated by dendritic cells, monocytes and microglia (Furtado et al., 2006). Whereas the adaptive immune system seems to drive mainly relapses of MS, abnormalities of the innate immune system may prevail in the progressive stage of the disease (Weiner, 2009).
An important aspect of MS pathogenesis is the apoptotic activity in the lymphatic and nervous system. Therefore, apoptotic processes have been analysed in EAE. While apoptosis plays obviously a pivotal pathogenic role when affecting ODCs (Hövelmeyer et al., 2005), it appears to be protective when eliminating myelin-reactive and bystander T cells (Zettl et al., 1997). This process is augmented by therapeutic drugs such as methylprednisolone (MP) (Schmidt et al., 2000) and resveratrol (Singh et al., 2007).
The role of cytokines and neurotrophic factors for the pathogenesis of MS and EAE has been investigated in a plethora of studies and the results have been reviewed elsewhere (Goverman, 2009, Imitola et al., 2005, Link, 1998, Linker et al., 2009b, Mirowska-Guzel, 2009, Owens et al., 2001, Ozenci et al., 2002). For details the reader is referred to these reviews.
In addition to the mentioned immunopathogenic pathways a direct crosstalk between the immune and nervous system may influence the pathogenesis of MS and EAE (Kerschensteiner et al., 2009, Mix et al., 2007). This involves direct effects of cytokines and chemokines on nerve cells and modulation of immune cell activity by neurotrophins and neurotransmitters implicating new treatment approaches, e.g. by associative conditioning (Jones et al., 2008). Disclosure of the complex mechanisms of neuro-immune interactions requires further investigations in the EAE model.
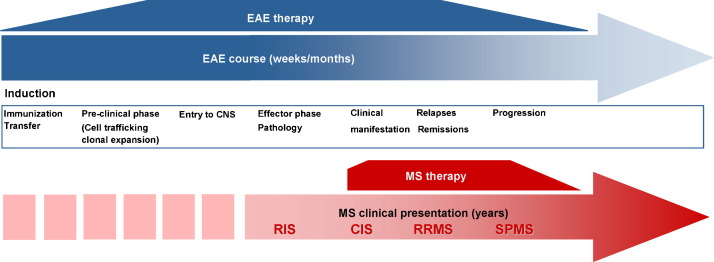
Many other aspects of MS research are investigated in the animal model. Among them monitoring of disease activity by traditional magnetic resonance imaging (MRI) (Morrissey et al., 1996) and new bioluminescence techniques (Luo et al., 2008) deserve special attention. Identification of reliable surrogate markers for diagnostic and prognostic purposes (responder detection for specific therapies, disease follow-up) will be a prominent task of the future. However, a consistent limitation for the translation of experimental results into the clinic is the different situation in EAE and MS concerning the timeline of detection of clinical signs and of therapeutic interventions (Fig. 5 ). Whereas in EAE pathological processes can be observed from the beginning and treatment approaches can be started at the early pre-clinical phase, in MS diagnostic measures will commonly not be initiated before first clinical signs are present and the intensity of treatment increases usually until late progression of the disease.
Fig. 5.
Timeline of the pathophysiological and clinical course of EAE and MS. In EAE, the complete pathological course including the pre-clinical phase is detected and immune therapeutic interventions start early and decrease usually with the proceeding time. In MS, there is an opposite situation. First radiological signs remain usually undetected and immunomodulatory treatment starts only with first clinical signs and is usually intensified until late progression of the disease. Abbreviations: CIS, clinical isolated syndrome; CNS, central nervous system; RIS, radiologic isolated syndrome; RRMS, relapsing–remitting MS; SPMS, secondary progressive MS.
3. Development and validation of novel therapies
3.1. Therapies developed primarily in EAE
Despite extensive screening for new targets of MS therapy in EAE so far only a few of the established MS therapies have been developed in the animal model. Examples are glatiramer acetate, mitoxantrone and natalizumab (Kieseier and Hartung, 2003, Steinman and Zamvil, 2006).
The glatiramer acetate preparation is a random polymer consisting of repeated sequences of the four amino acids glutamic acid, lysine, alanine and tyrosine that occur in MBP in a specific molar ratio. It was primarily called copolymer 1 and tested first for its encephalitogenic potency and subsequently for its influence on guinea pig EAE (Teitelbaum et al., 1971). Surprisingly, it turned out that copolymer 1 suppressed rather than induced EAE, most probably via stimulation of Th2/Th3-mediated anti-MBP immune response (Aharoni et al., 1997, Aharoni et al., 2008). Recent studies utilising sophisticated immunologic techniques point to a more complex mechanism of action of glatiramer acetate, including modification and killing of APCs, generation of regulatory T cells and turning the polyclonal CD8+ T cell response into an oligoclonal one (Racke et al., 2010).
Mitoxantrone has first been proven to be a powerful immunosuppressive drug in EAE (Ridge et al., 1985) and it is now a second-line component of escalating MS therapy (Hartung et al., 2002, Krapf et al., 2005, Neuhaus et al., 2006a, Neuhaus et al., 2006b, Rieckmann et al., 2004). Its mechanism of action relies most probably on cytotoxic effects on lymphocytes and induction of apoptosis of APC such as monocytes and dendritic cells (Neuhaus et al., 2005, Vollmer et al., 2010).
Natalizumab is a monoclonal antibody (mAb) that inhibits the transmigration of immune cells into the inflamed parenchyma of lymphatic organs and the CNS. It binds to α4β1-integrin (CD49dCD29, very late activation antigen-4) on lymphocytes and blocks the interaction with the integrin ligand CD106 (vascular cell adhesion molecule-1) on endothelia cells thereby being effective in preventing EAE (Rice et al., 2005, Yednock et al., 1992). It is the first mAb approved for therapeutic trials in MS (Polman et al., 2006) now belonging to second-line MS therapeutics, although it carries the risk to activate the polyoma virus JC leading to progressive multifocal leukoencephalopathy, especially if applied in combination with IFN-β (Clifford et al., 2010, Kleinschmidt-DeMasters and Tyler, 2005, Langer-Gould et al., 2005, Stüve and Bennett, 2007, Yousry et al., 2006). Recently, even cases of progressive multifocal leukoencephalopathy on natalizumab monotherapy have been reported (Clifford et al., 2010, Hartung, 2009, Hartung et al., 2009, Lindå et al., 2009, Wenning et al., 2009).
3.2. Therapies investigated secondarily in EAE
A number of established MS therapies have subsequently been investigated in the EAE model. The aims are:
-
(i)
to get a deeper insight into the mechanisms of action including disclosure of the specific targets of the therapies and
-
(ii)
to improve regimens of old therapies and to develop new therapies targeting the same pathogenic mechanisms, but being more convenient for clinical practice (low frequency of application, oral application) and avoiding adverse side-effects.
For example, with respect to methylprednisolone therapy for MS relapses Schmidt et al. (2000) could unravel a switch from cytoplasmic to nuclear effects accompanied by enhanced T cell apoptosis with increasing steroid dosage. Moreover, liposome encapsulated MP revealed a dose-dependently increased therapeutic efficiency compared to free MP in EAE (Linker et al., 2008b). In IFN-β treated EAE, interruption of therapy caused disease exacerbation (van der Meide et al., 1998). In an attempt to explore the influence of intravenous immunoglobulin (IVIg) therapy on the local immune response in the CNS, Jørgensen et al. (2007) found accumulation of IVIg only in active CNS lesions with BBB breakdown limiting its prospects for repair processes. However, natural CD4+CD25+FoxP3+ regulatory T cells were enhanced by prophylactic application of IVIg in an EAE study of Ephrem et al. (2008) suggesting a potential benefit of this therapy in early onset MS.
In other therapeutic trials, two treatments have been combined in order to achieve synergistic effects and/or to reduce adverse side-effects of single agents. For example, combined application of MP and erythropoietin protected neurons and axons of retinal ganglion cells and optic nerve of rats with MOG-induced EAE from morphological and functional impairment, whereas monotherapy caused only isolated neuronal or axonal protection without clinical benefit (Diem et al., 2005). In a MBP-induced active rat EAE model, the anti-inflammatory, anti-demyelinating and neuroprotective effects of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitory statins could be improved by combination with the selective phosphodiesterase-4 inhibitor rolipram (Paintlia et al., 2008, Paintlia et al., 2009) or the protein kinase A activating substance 5-aminoimidazole-4-carboxamide-1-β-d-ribofuranoside (Paintlia et al., 2006), even when the statin was applied in suboptimal doses. For rolipram alone, beneficial effects in the animal model could previously not be reproduced in the human system (Zhu et al., 2001). In actively induced chronic murine EAE a synergistic therapeutic effect of IFN-β and the immunomodulatory drug laquinimod was observed (Runström et al., 2006). In a patient study, the IFN-β-mediated up-regulation of the anti-inflammatory cytokine IL-10 was enhanced by additive administration of the non-selective phosphodiesterase inhibitor pentoxiphylline (Weber et al., 1998). Pentoxiphylline also reduced side-effects of IFN-β therapy such as myalgia, fever and injection site reactions (Rieckmann et al., 1996). IVIg decreased T cell apoptosis and liver damage, but increased ODC apoptosis in high-dose MBP-treated rats with AT-EAE induced by MBP-specific T cells (Weishaupt et al., 2002). But there are also perilous combination effects, which cannot always be foreseen and thereby prevented in the animal model as illustrated by the hazardous combination of IFN-β with natalizumab (Hartung et al., 2009, Kleinschmidt-DeMasters and Tyler, 2005, Langer-Gould et al., 2005, Stüve and Bennett, 2007) or statin (Birnbaum et al., 2008).
While disease-modifying agents act largely through modulation of peripheral immune mechanisms, some of them appear to act additionally or even predominantly locally within the CNS, e.g. at the BBB like IFN-β (Dhib-Jalbut and Marks, 2010) and natalizumab (Rice et al., 2005), on resident auto-reactive T cells and ODC like fingolimod (Miron et al., 2008, Miron et al., 2010, Papadopoulos et al., 2010). When entering the CNS via a locally disrupted BBB at lesion site, IFN-β may also directly act on astrocytes (Boutros et al., 1997) and microglia (Prinz et al., 2008) and even exert direct protective effects on neurons (Plioplys and Massimini, 1995).
3.3. Therapy failures in MS
On the other hand, there are several examples of compounds which were quite effective in curtailing disease activity in the animal model but turned out to lack therapeutic utility or proved to generate inacceptable adverse effect in MS patients. These inconsistencies prompted Sriram and Steiner (2005) to consider EAE a “misleading model of MS”. Examples for therapy failures in MS are given in Table 3 . Reasons for the discrepant result obtained in the animal and human systems could be manifold. Their nature may be genetic (species differences, peculiarities of inbred animal strains), pathogenetic (individual variability between MS patients) or kinetic (different ontogeny and biorhythms, temporal differences of immune reactivity and response to therapy). Additionally, in MS the BBB may be insufficiently disrupted as compared to EAE thereby preventing therapeutic molecules to reach their target within the CNS. This seems to be relevant especially when targeting cytokines in MS. For example, the mAb to IL-12 p40 ustekinumab failed to improve relapsing–remitting MS despite promising results in rodent and marmoset EAE (Segal et al., 2008; reviewed in Steinman, 2010). Other promising therapeutic principles revealed already in the animal model limited benefit or adverse effects precluding their use as MS therapeutics. Examples are:
-
(i)
the neuroprotective polypeptide hormone ciliary neurotrophic factor (CNTF), which elicited an acute-phase response in rat liver (Dittrich et al., 1994),
-
(ii)
the anti-adhesion molecule mAb anti-CD54, which revealed no MRI effect in AT-EAE (Morrissey et al., 1996),
-
(iii)
the Na+-channel blocker phenytoin, which potentially protects demyelinated axons, but resulted in exacerbation of MOG–EAE after withdrawal (Black et al., 2007),
-
(iv)
the phosphodiesterase-4 inhibitor rolipram, while very effective in suppressing EAE, failed to suppress inflammatory activity as gleaned through magnetic resonance imaging in a pilot trial in patients with relapsing–remitting MS (Bielekova et al., 2009),
-
(v)
the immunosuppressive drug cyclosporin A prevented BBB disruption and suppressed the development of EAE, but was classified as unacceptable for treatment of MS based on risk/benefit consideration due to low efficacy and frequent adverse reactions (Goodin et al., 2002, Kappos et al., 1988, Kieseier and Hartung, 2003, McCombe et al., 1999, Paul and Bolton, 1995).
Table 3.
Failure of translation of experimental therapies from the animal model to the clinical practice (selected examples).
| Therapy | Results in MS patients | References |
|---|---|---|
| Anti-TNF-α mAb infliximab | Increased MRI activity | van Oosten et al. (1996) |
| Anti-CD3 and anti-CD4 antibodies | No significant clinical effect | Wiendl and Hohlfeld (2002) |
| Anti-CD28 mAb TGN-1412 | Cytokine storm causing multiple organ failure | Hünig (2007) |
| Altered peptide ligands | Anaphylactic reactions and exacerbation of MS | Bielekova et al. (2000), Kappos et al. (2000) |
| Oral tolerogens | No significant clinical effect | Faria and Weiner (2006) |
| Sulfasalazine | Only transient clinical effect | Noseworthy et al. (1998) |
| Linomide | Cardiopulmonary toxicity | Noseworthy et al. (2000) |
The table is reproduced from Mix et al. (2008) with permission of the publisher (Springer).
If one therefore considers only the therapeutic trials conducted in EAE and their translation into the clinic, it may well be regarded as a misleading model of MS. However, several aspects of the aetiopathogenesis of MS such as susceptibility genes, immunoregulatory circuits, mechanisms of immune cell activation, migration and elimination as well as of nervous tissue destruction and repair have been successfully studied in EAE rendering it a useful model of MS (Hemmer and Hartung, 2007, Schreiner et al., 2009). EAE will be of continued utility in the future if one capitalizes on the availability of distinct types of EAE, including those induced in transgenic and knockout animals, to explore pathogenic pathways and strategies of intervention in different subtypes or variants of MS.
4. Perspectives
As outlined before, several therapeutic interventions have been successful exclusively in EAE, but not in MS. Other therapeutic agents have shown proven benefit in both EAE and MS. A survey of these agents is given in Table 4 . There are also few instances where therapeutic agents for the treatment of MS have been clinically developed without prior evaluation in the animal model. Examples of such drugs including their proposed mechanism of action are listed in Table 5 .
Table 4.
Therapeutic agents effective in both MS and EAE.
Table 5.
Experimental therapies for MS evaluated in clinical trials without prior investigation in the animal model (selected examples).
| Therapy | Proposed mechanism of action | References |
|---|---|---|
| Monoclonal antibodies | Bielekova and Becker (2010), McLaughlin and Wucherpfennig (2008), Linker et al. (2008c) | |
| - Rituximab,ocrelizumab, ofatumumab | Anti-CD20 inhibits B cells. | Hauser et al. (2008), Hawker et al. (2009) |
| - Daclizumab | Anti-CD25 inhibits lymphocyte activation and expands subpopulation of regulatory T cells. | Rose et al. (2007), Wynn et al. (2010) |
| - Alemtuzumab | Anti-CD52 depletes lymphocytes. | CAMMS 223 Trial Investigators (2008), Jones and Coles (2008) |
| Teriflunomide | Dihydro-orotate dehydrogenase inhibitor disrupts the immunologic synapse. | O’Connor et al. (2006), Zeyda et al. (2005) |
| Temsirolimus | Antifungal antibiotic rapamycin acts immunosuppressive. | Carlson et al. (1993), Keever-Taylor et al. (2007) |
| Cladribine | 2-Chloro-2′-deoxyadenosine alters binding of transcription factors to the gene regulatory AT-rich sequences; accumulated cladribine nucleotides disrupt DNA synthesis and repair and suppress CD4+ and CD8+ T cells. | Foley et al. (2004), Giovannoni et al. (2010), Hartung et al. (2010), Sipe et al. (1996) |
The table is reproduced from Mix et al. (2008) with slight modification with permission of the publisher (Springer).
Nonetheless, an increasing number of emerging therapies for MS are currently being tested in pre-clinical phases by making use of the EAE model (Cohen and Rieckmann, 2007, Linker et al., 2008c, Weiner, 2009). The most promising experimental therapies rely on gene transfer, stem cell transplantation, oral administration of small molecular weight disease-modifying drugs and intravenous or subcutaneous application of mAb targeting cells or molecules crucial in the pathogenesis of the disease (Aktas et al., 2010, Bielekova and Becker, 2010, Butti et al., 2008, Einstein et al., 2009, Hauser et al., 2008, Hawker et al., 2009, Hemmer and Hartung, 2007, Kieseier and Wiendl, 2007, Pluchino and Martino, 2008, Wynn et al., 2010). Table 6 provides a survey of experimental therapeutic approaches that are currently being investigated, some of which being already approved for phase I-III clinical trials. Table 6 also includes proposed mechanisms of action of the new therapies. Putative targets of established and experimental MS therapies with and without prior testing in EAE are indicated in Fig. 4. Other experimental approaches for MS are based on vaccination, e.g. with pathogenic T cells, TCRs, dendritic cells pulsed with antigen, DNA vaccine encoding MBP, axonal growth inhibitors associated with myelin or pro-inflammatory cytokines. These approaches are extensively reviewed elsewhere (Correale et al., 2008). Recently, a new therapeutic target for a more selective treatment of EAE and MS compared to available therapies has been proposed, i.e. the IL-7-IL-7R pathway that affects pathogenic Th17 cells, but spares regulatory T cells and unrelated immune cells (Liu et al., 2010).
Table 6.
Experimental therapies for MS as tested in EAE.
| Therapeutic approach | Proposed mechanism of action | References |
|---|---|---|
| Gene therapy | Furlan et al. (2003) | |
| - IL-4 | Inhibits Th1 cell activation. | Broberg et al. (2004), Butti et al. (2008) |
| - IFN-β | Inhibits local autoimmune reaction in the CNS. | Makar et al. (2008a) |
| Stem cell transplantation | Scolding (2006) | |
| - Mesenchymal stem cells | Modulate T cell function, decrease IL-17 via IL-23 secretion. | Pedemonte et al. (2007), Wang et al. (2008) |
| - Neural stem cells | Down-regulate inflammation, stimulate the endogenous brain repair system. | Aharonowiz et al. (2008), Einstein et al., 2006, Einstein et al., 2009, Martino and Pluchino (2007), Pluchino and Martino (2008) |
| Neurotrophic factors | Mirowska-Guzel (2009) | |
| - BDNF | Reduces inflammation and apoptosis. | Makar et al. (2008b) |
| - Erythropoietin | Activates the neuroprotective phosphatidylinositol 3-kinase/Akt pathway, down-regulates glial MHC class II. | Agnello et al. (2002), Sättler et al. (2004), Yuan et al. (2008) |
| Monoclonal antibodies | Buttmann and Rieckmann (2008), Lutterotti and Martin (2008), Rose et al. (2008) | |
| Natalizumab | Anti-CD49d inhibits lymphocyte adhesion. | Rice et al. (2005), Stüve and Bennett (2007) |
| Anti-cytokines | Small molecular weight drug suppresses pro-inflammatory cytokines. | Karpus et al. (2008) |
| CRYAB | Stress protein αB-crystallin has an anti-inflammatory effect. | Ousman et al. (2007) |
| Beta-lactam antibiotic | Ceftriaxone modulates myelin antigen presentation and impairs antigen-specific T cell migration into the CNS. | Melzer et al. (2008) |
| Steroids | Estradiol and progesterone increase BDNF and myelination. | Garay et al. (2008) |
| Statins | 3-Hydroxy-3-methylglutaryl-coenzymeA-reductase inhibitors prevent geranyl-geranylation of RhoA GTPase and its tethering to the membrane and thereby inhibit T cell activation and infiltration into the CNS. | Aktas et al. (2003), Mix et al. (2006), Stanislaus et al. (1999), Waiczies et al. (2008), Youssef et al. (2002) |
| Fingolimod (FTY720) | Sphingosine-1-phosphate agonist reduces systemic T and B cell response as well as auto-reactive T cells in the CNS and it promotes remyelination by stimulation of ODC function. | Balatoni et al. (2007), Foster et al. (2009), Kappos et al. (2006b), Miron et al., 2008, Miron et al., 2010, Papadopoulos et al. (2010) |
| Fumarate (BG-12) | Fumaric acid esters increase the anti-inflammatory cytokine IL-10. | Schilling et al. (2006) |
| Minocycline | Inhibits matrix metalloproteinases and thereby T cell transmigration. | Brundula et al. (2002) |
| Gemfibrozile, fenofibrate, ciprofibra | Peroxisome proliferator-activated receptor (PPAR)-α agonists increase the anti-inflammatory cytokine IL-4. | Lovett-Racke et al. (2004) |
| Laquinimod | Linomide-derivative ABR-215062 changes the cytokine balance towards the anti-inflammatory cytokines IL-4, IL-10 and TGF-β | Brunmark et al. (2002), Comi et al. (2008), Runström et al. (2006), Wegner et al. (2009), Yang et al. (2004) |
| AICAR | Protein kinase A activating 5-aminoimidazole-4-carboxamide-1-β-d-ribofuranoside inhibits the pro-inflammatory cytokines IFN-γ and TNF-α and induces the anti-inflammatory cytokines IL-4 and IL-10. | Nath et al. (2005) |
| CYLA | Calpain inhibitor reduces inflammatory infiltration, demyelination and axonal injury. | Hassen et al. (2008) |
| 3,4-DAA | Derivative of tryptophan metabolite N-(3,4,-Dimethoxycinnamoyl) anthranilic acid inhibits pro-inflammatory cytokines. | Platten et al. (2005) |
| EGCG | Green tea constituent (−)-epigallocatechin-3-gallate blocks proteasome complex, proliferation and TNF-α production of encephalitogenic T cells and formation of neurotoxic reactive oxygen species. | Aktas et al. (2004) |
| Flavonoids | Luteoline scavenges oxygen radicals, inhibits RhoA GTPase and prevents monocyte infiltration into the CNS. | Hendriks et al. (2004) |
| Metallothionein I and II | Antioxidant proteins act anti-inflammatory and neuroprotective. | Espejo et al. (2005) |
| Vitamin D | 1,25-Dihydroxyvitamin D3 declines inducible nitric oxide synthase, chemokines and monocyte recruitment into the CNS and stimulates activated CD4+ T cell apoptosis in the CNS. | Pedersen et al. (2007) |
| K+-channel blocker | Alkoxypsoralens, kaliotoxin, charybdotoxin, psora-4, bupivacaine, anandamide, spermine and ruthenium red inhibit T cell activation. | Beeton et al. (2001), Meuth et al. (2008), Strauss et al. (2000), Wulff et al. (2009) |
| Na+-channel blocker | Phenytoin, flecainide and lamotrigine prevent axonal degeneration. | Bechtold et al. (2004), Bechtold et al. (2006), Lo et al. (2003) |
| Dopamine receptor antagonists | DRD1 antagonist SCH23390 blocks dopamine receptors on Th17 cells. | Nakano et al. (2008) |
| Glutamate receptor antagonists | AMPA/kainate antagonists NBQX and MPQX prevent glutamate-mediated demyelination and neuronal death. | Smith et al. (2000) |
| Histamine receptor antagonists | Histamine-1 receptor antagonist hydroxyzine blocks mast cell degranulation. | Dimitriadou et al. (2000) |
| Serotonin reuptake inhibitors | Venlafaxine suppresses pro-inflammatory cytokines. | Vollmar et al. (2008) |
| Bifunctional hybrid molecules | ||
| Bifunctional peptide inhibitor (BPI) | Hybrid peptides made of integrin CD11a237–246 and antigenic epitopes PLP139–151 or glutamic acid decarboxylase GAD208–217 block the immunologic synapse. | Kobayashi et al. (2008) |
| Fulleren hybrid molecule (ABS-75) | Hybrid molecules made of an antioxidant carboxy-fullerene moiety and NMDA receptor-targeting adamantyl groups inhibit oxidative injury, chemokine expression, CD11b+ cell infiltration, demyelination and axonal loss. | Basso et al. (2008) |
The table is reproduced from Mix et al. (2008) with slight modification with permission of the publisher (Springer).
An important field of therapeutic approaches in EAE and MS that will deserve more attention in the future is the enhancement of remyelination. So far experimental trials have involved transplantation of neural stem cells, ODC precursor cells, Schwann cells and olfactory ensheathing cells and application of growth factors such as platelet derived growth factor and epidermal growth factor (Franklin and Ffrench-Constant, 2008). However, these trials are hampered by the fact that the demyelinating lesions are regularly wide-spread and dispersed within the CNS and that they, additionally, lack stimulating factors of ODC precursor cell recruitment and differentiation, or even contain inhibitory factors of remyelination like the neurite outgrowth inhibitor-A (Nogo-A) and its receptor component leucin-rich repeat and Ig domain containing-1 (Lingo-1) (Pernet et al., 2008). Therefore, current efforts to enhance remyelination in EAE and MS rely mainly on established therapies with remyelinating potency such as IVIg, especially polyclonal IgM (Bieber et al., 2000, Trebst and Stangel, 2006, Wright et al., 2009), glatiramer acetate (Aharoni et al., 2008, Arnon and Aharoni, 2009, Racke et al., 2010) and fingolimod (Miron et al., 2008, Miron et al., 2010) or on the application of antagonists of the remyelination inhibitors Nogo-A and Lingo-1 (Bourquin et al., 2008, Mi et al., 2007, Mi et al., 2009, Rudick et al., 2008, Yang et al., 2010).
This review is restricted to the EAE model of MS and a short reference to the toxic cuprizone-mediated model of demyelination. Further information on advantages and disadvantages of EAE and additionally of virus-mediated demyelinating diseases can be derived from the excellent monograph “Experimental models of multiple sclerosis” edited by Lavi and Constantinescu (2005).
In summary, specific questions of MS genetics, pathogenesis and therapy require investigations in different available and forthcoming EAE models. A comparison of the most important immunopathological, clinical and therapeutic features of EAE and MS is given in Table 7 . Thereby it becomes obvious that great precaution is advisable when translating the results of experimental therapeutic trials into clinical practice.
Table 7.
Comparison of immunopathological, clinical and therapeutic features of EAE and MS.
| EAE | MS | |
|---|---|---|
| Genetics | Susceptible and resistant animal strains and colonies, e.g. C57BL/6 vs C57BL/10.S mice and different colonies of Lewis rat | Weak evidence of association (confirmed only for HLA-DRB1*15), risk alleles: IL-2RA, IL-7RA and EV15 |
| Pathology | ||
| - Inflammation | Dominant (CD4+ T cells and macrophages) | Rare (type I/II, CD4+/CD8+ T cells, CD20+ B cells, macrophages) |
| - Demyelination | Rare (anti-MOG–EAE) | Strong |
| - Degeneration | Late (murine EAE) | Early (type III/IV) |
| - Cortical lesions | Rare (MOG–EAE in marmosets) | Rare |
| Clinical course | ||
| - Acute | Frequent (active EAE) | Rare (Marburg type) |
| - Primary chronic-progrssive | Rare (MOG–EAE, AT-EAE) | Rare (<10%) |
| - Relapsing–remitting | Rare (PLP139–151–EAE, pertussis toxin-EAE) | Frequent (>90%) |
| - Immunotherapy | ||
| - Immunosuppression/immunomodulation | Azathioprine, IFN-β, glatiramer acetate, gene therapy, stem cell transplantation, mitoxantrone, mAb, small molecular weight disease-modifying drugs | Azathioprine, IFN-β, glatiramer acetate, plasma exchange, immunoadsorption, mitoxantrone, mAb, IVIg |
| - Anti-inflammatory | Methylprednisolone | Methylprednisolone |
| - Antigen specific | Altered peptide ligands, bifunctional peptide inhibitors, oral and nasal tolerance, DNA vaccines | |
| - Neuroprotective | CNTF, BDNF, erythropoietin | |
Conflict of interest
Eilhard Mix, Hans Meyer-Rienecker, Hans-Peter Hartung and Uwe K. Zettl have no conflict of interest related to this review to declare.
Acknowledgements
The authors are most grateful to Gabriele Gillwaldt and Verena Reimann for providing the artwork.
References
- Afzali B., Lombardi G., Lechler R.I., Lord G.M. The role of T helper 17 (Th17) and regulatory T cells (Treg) in human organ transplantation and autoimmune disease. Clin. Exp. Immunol. 2007;148:32–46. doi: 10.1111/j.1365-2249.2007.03356.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agnello D., Bigini P., Villa P., Mennini T., Cerami A., Brines M.L., Ghezzi P. Erythropoietin exerts an anti-inflammatory effect on the CNS in a model of experimental autoimmune encephalomyelitis. Brain Res. 2002;952:128–134. doi: 10.1016/s0006-8993(02)03239-0. [DOI] [PubMed] [Google Scholar]
- Aharoni R., Teitelbaum D., Sela M., Arnon R. Copolymer 1 induces T cells of the T helper type 2 that crossreact with myelin basic protein and suppress experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. U. S. A. 1997;94:10821–10826. doi: 10.1073/pnas.94.20.10821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aharoni R., Herschkovitz A., Eilam R., Blumberg-Hazan M., Sela M., Brück W., Arnon R. Demyelination arrest and remyelination induced by glatiramer acetate treatment of experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. U. S. A. 2008;105:11358–11363. doi: 10.1073/pnas.0804632105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aharonowiz M., Einstein O., Fainstein N., Lassmann H., Reubinoff B., Ben-Hur T. Neuroprotective effect of transplanted human embryonic stem cell-derived neural precursors in an animal model of multiple sclerosis. PLoS One. 2008;3:e3145. doi: 10.1371/journal.pone.0003145. (1–10) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aktas O., Waiczies S., Smorodchenko A., Dorr J., Seeger B., Prozorovski T., Sallach S., Endres M., Brocke S., Nitsch R., Zipp F. Treatment of relapsing paralysis in experimental encephalomyelitis by targeting Th1 cells through atorvastatin. J. Exp. Med. 2003;197:725–733. doi: 10.1084/jem.20021425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aktas O., Prozorovski T., Smorodchenko A., Savaskan N.E., Lauster R., Kloetzel P.M., Infante-Duarte C., Brocke S., Zipp F. Green tea epigallocatechin-3-gallate mediates T cellular NF-kappa B inhibition and exerts neuroprotection in autoimmune encephalomyelitis. J. Immunol. 2004;173:5794–5800. doi: 10.4049/jimmunol.173.9.5794. [DOI] [PubMed] [Google Scholar]
- Aktas O., Waiczies S., Zipp F. Neurodegeneration in autoimmune demyelination: recent mechanistic insights reveal novel therapeutic targets. J. Neuroimmunol. 2007;184:17–26. doi: 10.1016/j.jneuroim.2006.11.026. [DOI] [PubMed] [Google Scholar]
- Aktas O., Kieseier B., Hartung H.P. Neuroprotection, regeneration and immunomodulation: broadening the therapeutic repertoire in multiple sclerosis. Trends Neurosci. 2010;33:140–152. doi: 10.1016/j.tins.2009.12.002. [DOI] [PubMed] [Google Scholar]
- Arnon R., Aharoni R. Neuroprotection and neurogeneration in MS and its animal model EAE effected by glatiramer acetate. J. Neural. Transm. 2009;116:1443–1449. doi: 10.1007/s00702-009-0272-3. [DOI] [PubMed] [Google Scholar]
- Aulchenko Y.S., Hoppenbrouwers I.A., Ramagopalan S.V., Broer L., Jafari N., Hillert J., Link J., Lundström W., Greiner E., Sadovnick D.A., Goossens D., van Broeckhoven C., del Favero J., Ebers G.C., Oostra B.A., van Duijn C.M., Hintzen R.Q. Genetic variation in the KIF1B locus influences susceptibility to multiple sclerosis. Nat. Genet. 2008;40:1402–1403. doi: 10.1038/ng.251. [DOI] [PubMed] [Google Scholar]
- Balatoni B., Storch M.K., Swoboda E.M., Schönborn V., Koziel A., Lambrou G.N., Hiestand P.C., Weissert R., Foster C.A. FTY720 sustains and restores neuronal function in the DA rat model of MOG-induced experimental autoimmune encephalomyelitis. Brain Res. Bull. 2007;74:307–316. doi: 10.1016/j.brainresbull.2007.06.023. [DOI] [PubMed] [Google Scholar]
- Barcellos L.F., Sawcer S., Ramsay P.P., Baranzini S.E., Thomson G., Briggs F., Cree B.C., Begovich A.B., Villoslada P., Montalban X., Uccelli A., Savettieri G., Lincoln R.R., DeLoa C., Haines J.L., Pericak-Vance M.A., Compston A., Hauser S.L., Oksenberg J.R. Heterogeneity at the HLA-DRB1 locus and risk for multiple sclerosis. Hum. Mol. Genet. 2006;15:2813–2824. doi: 10.1093/hmg/ddl223. [DOI] [PubMed] [Google Scholar]
- Baranzini S.E., Bernard C.C., Oksenberg J.R. Modular transcriptional activity characterizes the initiation and progression of autoimmune encephalomyelitis. J. Immunol. 2005;174:7412–7422. doi: 10.4049/jimmunol.174.11.7412. [DOI] [PubMed] [Google Scholar]
- Basso A.S., Frenkel D., Quintana F.J., Costa-Pinto F.A., Petrovic-Stojkovic S., Puckett L., Monsonego A., Bar-Shir A., Engel Y., Gozin M., Weiner H.L. Reversal of axonal loss and disability in a mouse model of progressive multiple sclerosis. J. Clin. Invest. 2008;118:1532–1543. doi: 10.1172/JCI33464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baxter A.G. The origin and application of experimental autoimmune encephalomyelitis. Nat. Rev. Immunol. 2007;7:904–912. doi: 10.1038/nri2190. [DOI] [PubMed] [Google Scholar]
- Bechtold D.A., Kapoor R., Smith K.J. Axonal protection using flecainide in experimental autoimmune encephalomyelitis. Ann. Neurol. 2004;55:607–616. doi: 10.1002/ana.20045. [DOI] [PubMed] [Google Scholar]
- Bechtold D.A., Miller S.J., Dawson A.C., Sun Y., Kapoor R., Berry D., Smith K.J. Axonal protection achieved in a model of multiple sclerosis using lamotrigine. J. Neurol. 2006;253:1542–1551. doi: 10.1007/s00415-006-0204-1. [DOI] [PubMed] [Google Scholar]
- Beeton C., Barbaria J., Giraud P., Devaux J., Benoliel A.M., Gola M., Sabatier J.M., Bernard D., Crest M., Béraud E. Selective blocking of voltage-gated K+ channels improves experimental autoimmune encephalomyelitis and inhibits T cell activation. J. Immunol. 2001;166:936–944. doi: 10.4049/jimmunol.166.2.936. [DOI] [PubMed] [Google Scholar]
- Ben-Nun A., Wekerle H., Cohen I.R. The rapid isolation of clonable antigen-specific T lymphocyte lines capable of mediating autoimmune encephalomyelitis. Eur. J. Immunol. 1981;11:195–199. doi: 10.1002/eji.1830110307. [DOI] [PubMed] [Google Scholar]
- Bettelli E., Baeten D., Jäger A., Sobel R.A., Kuchroo V.K. Myelin oligodendrocyte glycoprotein-specific T and B cells cooperate to induce a Devic-like disease in mice. J. Clin. Invest. 2006;116:2393–2402. doi: 10.1172/JCI28334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bieber A., Asakura K., Warrington A., Kaveri S.V., Rodriguez M. Antibody-mediated remyelination: relevance to multiple sclerosis. Mult. Scler. 2000;6(Suppl. 2):S1–S5. [PubMed] [Google Scholar]
- Bielekova B., Goodwin B., Richert N., Cortese I., Kondo T., Afshar G., Gran B., Eaton J., Antel J., Frank J.A., McFarland H.F., Martin R. Encephalitogenic potential of the myelin basic protein peptide (amino acids 83–99) in multiple sclerosis: results of a phase II clinical trial with an altered peptide ligand. Nat. Med. 2000;6:1167–1175. doi: 10.1038/80516. [DOI] [PubMed] [Google Scholar]
- Bielekova B., Richert N., Howard T., Packer A.N., Blevins G., Ohayon J., McFarland H.F., Stürzebecher C.S., Martin R. Treatment with the phosphodiesterase type-4 inhibitor rolipram fails to inhibit blood–brain barrier disruption in multiple sclerosis. Mult. Scler. 2009;15:1206–1214. doi: 10.1177/1352458509345903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bielekova B., Becker B.L. Monoclonal antibodies in MS: mechanisms of action. Neurology. 2010;74(Suppl. 1):S31–S40. doi: 10.1212/WNL.0b013e3181c97ed3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnbaum G., Cree B., Altafullah I., Zinser M., Reder A.T. Combining beta interferon and atorvastatin may increase disease activity in multiple sclerosis. Neurology. 2008;71:1390–1395. doi: 10.1212/01.wnl.0000319698.40024.1c. [DOI] [PubMed] [Google Scholar]
- Black J.A., Liu S., Carrithers M., Carrithers L.M., Waxman S.G. Exacerbation of experimental autoimmune encephalomyelitis after withdrawal of phenytoin and carbamazepine. Ann. Neurol. 2007;62:21–33. doi: 10.1002/ana.21172. [DOI] [PubMed] [Google Scholar]
- Blakemore W.F. Observations on oligodendrocyte degeneration, the resolution of status spongiosus and remyelination in cuprizone intoxication in mice. J. Neurocytol. 1972;1:413–426. doi: 10.1007/BF01102943. [DOI] [PubMed] [Google Scholar]
- Błaszczyk B., Giełdanowski J., Karakoz I. Experimental allergic encephalomyelitis in chickens. Arch. Immunol. Ther. Exp. (Warsz) 1978;26:743–746. [PubMed] [Google Scholar]
- Blink S.E., Miller S.D. The contribution of gammadelta T cells to the pathogenesis of EAE and MS. Curr. Mol. Med. 2009;9:15–22. doi: 10.2174/156652409787314516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourquin C., van der Haar M.E., Anz D., Sandholzer N., Neumaier I., Endres S., Skerra A., Schwab M.E., Linington C. DNA vaccination efficiently induces antibodies to Nogo-A and does not exacerbate experimental autoimmune encephalomyelitis. Eur. J. Pharmacol. 2008;588:99–105. doi: 10.1016/j.ejphar.2008.04.026. [DOI] [PubMed] [Google Scholar]
- Boutros T., Croze E., Yong V.W. Interferon-beta is a potent promoter of nerve growth factor production by astrocytes. J. Neurochem. 1997;69:939–946. doi: 10.1046/j.1471-4159.1997.69030939.x. [DOI] [PubMed] [Google Scholar]
- Broberg E.K., Salmi A.A., Hukkanen V. IL-4 is the key regulator in herpes simplex virus-based gene therapy of BALB/c experimental autoimmune encephalomyelitis. Neurosci. Lett. 2004;364:173–178. doi: 10.1016/j.neulet.2004.04.059. [DOI] [PubMed] [Google Scholar]
- Brundula V., Rewcastle N.B., Metz L.M., Bernard C.C., Yong V.W. Targeting leukocyte MMPs and transmigration: minocycline as a potential therapy for multiple sclerosis. Brain. 2002;125:1297–1308. doi: 10.1093/brain/awf133. [DOI] [PubMed] [Google Scholar]
- Brunmark C., Runström A., Ohlsson L., Sparre B., Brodin T., Aström M., Hedlund G. The new orally active immunoregulator laquinimod (ABR-215062) effectively inhibits development and relapses of experimental autoimmune encephalomyelitis. J. Neuroimmunol. 2002;130:163–172. doi: 10.1016/s0165-5728(02)00225-4. [DOI] [PubMed] [Google Scholar]
- Butti E., Bergami A., Recchia A., Brambilla E., del Carro U., Amadio S., Cattalini A., Esposito M., Stornaiuolo A., Comi G., Pluchino S., Mavilio F., Martino G., Furlan R. IL4 gene delivery to the CNS recruits regulatory T cells and induces clinical recovery in mouse models of multiple sclerosis. Gene Ther. 2008;15:504–1515. doi: 10.1038/gt.2008.10. [DOI] [PubMed] [Google Scholar]
- Buttmann M., Rieckmann P. Treating multiple sclerosis with monoclonal antibodies. Expert Rev. Neurother. 2008;8:433–455. doi: 10.1586/14737175.8.3.433. [DOI] [PubMed] [Google Scholar]
- CAMMS 223 Trial Investigators, Coles, A.J., Compston, D.A., Selmaj, K.W., Lake, S.L., Moran, S., Margolin, D.H., Norris, K., Tandon, P.K., 2008. Alemtuzumab vs interferon beta-1a in early multiple sclerosis. N. Engl. J. Med. 359, 1786–1801. [DOI] [PubMed]
- Campbell I.L., Stalder A.K., Akwa Y., Pagenstecher A., Asensio V.C. Transgenic models to study the actions of cytokines in the central nervous system. Neuroimmunomodulation. 1998;5:126–135. doi: 10.1159/000026329. [DOI] [PubMed] [Google Scholar]
- Carlson R.P., Baeder W.L., Caccese R.G., Warner L.M., Sehgal S.N. Effects of orally administered rapamycin in animal models of arthritis and other autoimmune diseases. Ann. N. Y. Acad. Sci. 1993;685:86–113. doi: 10.1111/j.1749-6632.1993.tb35855.x. [DOI] [PubMed] [Google Scholar]
- Carlton W.W. Response of mice to the chelating agents sodium diethyldithiocarbamate, alpha-benzoinoxime, and biscyclohexanone oxaldihydrazone. Toxicol. Appl. Pharmacol. 1966;8:512–521. doi: 10.1016/0041-008x(66)90062-7. [DOI] [PubMed] [Google Scholar]
- Casciano D.A., Woodcock J. Empowering microarrays in the regulatory setting. Nat. Biotechnol. 2006;24:1103. doi: 10.1038/nbt0906-1103. [DOI] [PubMed] [Google Scholar]
- Centonze D., Muzio L., Rossi S., Furlan R., Bernardi G., Martino G. The link between inflammation, synaptic transmission and neurodegeneration in multiple sclerosis. Cell Death Differ. 2010;17:1083–1091. doi: 10.1038/cdd.2009.179. [DOI] [PubMed] [Google Scholar]
- Chao M.J., Barnardo M.C., Lincoln M.R., Ramagopalan S.V., Herrera B.M., Dyment D.A., Montpetit A., Sadovnick A.D., Knight J.C., Ebers G.C. HLA class I alleles tag HLA-DRB1*1501 haplotypes for differential risk in multiple sclerosis susceptibility. Proc. Natl. Acad. Sci. U. S. A. 2008;105:13069–13074. doi: 10.1073/pnas.0801042105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Charles P., Tait S., Faivre-Sarrailh C., Barbin G., Gunn-Moore F., Denisenko-Nehrbass N., Guennoc A.M., Girault J.A., Brophy P.J., Lubetzki C. Neurofascin is a glial receptor for the paranodin/Caspr-contactin axonal complex at the axoglial junction. Curr. Biol. 2002;12:217–220. doi: 10.1016/s0960-9822(01)00680-7. [DOI] [PubMed] [Google Scholar]
- Clifford D.B., de Luca A., Simpson D.M., Arendt G., Giovannoni G., Nath A. Natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: lessons from 28 cases. Lancet Neurol. 2010;9:438–446. doi: 10.1016/S1474-4422(10)70028-4. [DOI] [PubMed] [Google Scholar]
- Cohen B.A., Rieckmann P. Emerging oral therapies for multiple sclerosis. Int. J. Clin. Pract. 2007;61:1922–1930. doi: 10.1111/j.1742-1241.2007.01561..x. [DOI] [PubMed] [Google Scholar]
- Cohen J.A., Barkhof F., Comi G., Hartung H.P., Khatri B.O., Montalban X., Pelletier J., Capra R., Gallo P., Izquierdo G., Tiel-Wieck K., de Vera A., Jin J., Stites T., Wi S., Aradhye S., Kappos L., TRANSFORMS Study Group Oral fingolimod or intramuscular interferon for relapsing multiple sclerosis. N. Engl. J. Med. 2010;362:402–415. doi: 10.1056/NEJMoa0907839. [DOI] [PubMed] [Google Scholar]
- Comabella M., Martin R. Genomics in multiple sclerosis—current state and future directions. J. Neuroimmunol. 2007;187:1–8. doi: 10.1016/j.jneuroim.2007.02.009. [DOI] [PubMed] [Google Scholar]
- Comi G., Filippi M., Barkhof F., Durelli L., Edan G., Fernández O., Hartung H.-P., Seeldrayers P., Sørensen P.S., Rovaris M., Martinelli V., Hommes O.R., Early Treatment of Multiple Sclerosis Study Group Effect of early interferon treatment on conversion to definite multiple sclerosis: a randomized study. Lancet. 2001;357:1576–1582. doi: 10.1016/s0140-6736(00)04725-5. [DOI] [PubMed] [Google Scholar]
- Comi G., Filippi M., Wolinsky J.S. European/Canadian mutlicenter, double-blind, randomized, placebo-controlled study of the effects of glatiramer acetate on magnetic resonance imaging-measured disease activity and burden in patients with relapsing multiple sclerosis. European/Canadian Glatiramer Acetate Study Group. Ann. Neurol. 2001;49:290–297. [PubMed] [Google Scholar]
- Comi G., Pulizzi A., Rovaris M., Abramsky O., Arbizu T., Boiko A., Gold R., Havrdova E., Komoly S., Sharrack B., Filippi M., LAQ/5062 Study Group Effect of laquinimod on MRI-monitored disease activity in patients with relapsing–remitting multiple sclerosis: a multicentre, randomised, double-blind, placebo-controlled phase IIb study. Lancet. 2008;371:2085–2092. doi: 10.1016/S0140-6736(08)60918-6. [DOI] [PubMed] [Google Scholar]
- Comi G., Martinelli V., Rodegher M., Moiala L., Bajenaru O., Varra A., Elovaara I., Fazekas F., Hartung H.P., Hillert J., King J., Komoly S., Lubetzki C., Montalban X., Myhr K.M., Ravnborg M., Rieckmann P., Wynn D., Young C., Filippi M., PreCISe Study Group Effect of glatiramer acetate on conversion to clinically definite multiple sclerosis in patients with clinically isolated syndrome (PreCISe study) a randomized, double-blind, placebo-controlled trial. Lancet. 2009;374:1503–1511. doi: 10.1016/S0140-6736(09)61259-9. [DOI] [PubMed] [Google Scholar]
- Correale J., Farez M., Gilmore W. Vaccines for multiple sclerosis: progress to date. CNS Drugs. 2008;22:175–198. doi: 10.2165/00023210-200822030-00001. [DOI] [PubMed] [Google Scholar]
- Dabbeekeh J.T., Faitar S.L., Dufresne C.P., Cowell J.K. The EVI5 TBC domain provides the GTPase-activating protein motif for RAB11. Oncogene. 2007;26:2804–2808. doi: 10.1038/sj.onc.1210081. [DOI] [PubMed] [Google Scholar]
- Dallenga T., Bittner A., Jäger W., Vollmar P., Oertel W.H., Sommer N., Möller J.C., Hemmer B., Stadelmann C., Nessler S. Increased axonal damage in p75NTR-knockout mice after induction of experimental autoimmune encephalomyelitis by adoptive transfer (Abstract) Acta Neuropathol. 2009;118:436. [Google Scholar]
- De Jager P.L., Baecher-Allan C., Maier L.M., Arthur A.T., Ottoboni L., Barcellos L., McCauley J.L., Sawcer S., Goris A., Saarela J., Yelensky R., Price A., Leppa V., Patterson N., de Bakker P.I., Tran D., Aubin C., Pobywajlo S., Rossin E., Hu X., Ashley C.W., Choy E., Rioux J.D., Pericak-Vance M.A., Ivinson A., Booth D.R., Stewart G.J., Palotie A., Peltonen L., Dubois B., Haines J.L., Weiner H.L., Compston A., Hauser S.L., Daly M.J., Reich D., Oksenberg J.R., Hafler D.A. The role of the CD58 locus in multiple sclerosis. Proc. Natl. Acad. Sci. U. S. A. 2009;106:5264–5269. doi: 10.1073/pnas.0813310106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Jager P.L., Jia X., Wang J., de Bakker P.I., Ottoboni L., Aggarwal N.T., Piccio L., Raychaudhuri S., Tran D., Aubin C., Briskin R., Romano S., International M.S., Genetics Consortium, Baranzini S.E., McCauley J.L., Pericak-Vance M.A., Haines J.L., Gibson R.A., Naeglin Y., Uitdehaag B., Matthews P.M., Kappos L., Polman C., McArdle W.L., Strachan D.P., Evans D., Cross A.H., Daly M.J., Compston A., Sawcer S.J., Weiner H.L., Hauser S.L., Hafler D.A., Oksenberg J.R. Meta-analysis of genome scans and replication identify CD6,IRF8 and TNFRSF1A as new multiple sclerosis susceptibility loci. Nat. Genet. 2009;41:776–782. doi: 10.1038/ng.401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derfuss T., Parikh K., Velhin S., Braun M., Mathey E., Krumbholz M., Kümpfel T., Moldenhauer A., Rader C., Sonderegger P., Pöllmann W., Tiefenthaller C., Bauer J., Lassmann H., Wekerle H., Karagogeos D., Hohlfeld R., Linington C., Meinl E. Contactin-2/TAG-1-directed autoimmunity is identified in multiple sclerosis patients and mediates gray matter pathology in animals. Proc. Natl. Acad. Sci. U. S. A. 2009;106:8302–8307. doi: 10.1073/pnas.0901496106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derfuss T., Linington C., Hohlfeld R., Meinl E. Axo-glial antigens as targets in multiple sclerosis: implications for axonal and grey matter injury. J. Mol. Med. 2010;(May 6) doi: 10.1007/s00109-010-0632-3. [DOI] [PubMed] [Google Scholar]
- Diem R., Sättler M.B., Merkler D., Demmer I., Maier K., Stadelmann C., Ehrenreich H., Bähr M. Combined therapy with methylprednisolone and erythropoietin in a model of multiple sclerosis. Brain. 2005;128:375–385. doi: 10.1093/brain/awh365. [DOI] [PubMed] [Google Scholar]
- Dimitriadou V., Pang X., Theoharides T.C. Hydroxyzine inhibits experimental allergic encephalomyelitis (EAE) and associated brain mast cell activation. Int. J. Immunopharmacol. 2000;22:673–684. doi: 10.1016/s0192-0561(00)00029-1. [DOI] [PubMed] [Google Scholar]
- Dittrich F., Thoenen H., Sendtner M. Ciliary neurotrophic factor: pharmacokinetics and acute-phase response in rat. Ann. Neurol. 1994;35:151–163. doi: 10.1002/ana.410350206. [DOI] [PubMed] [Google Scholar]
- Dhib-Jalbut S., Marks S. Interferon-beta mechanisms of action in multiple sclerosis. Neurology. 2010;74(Suppl. 1):S17–S24. doi: 10.1212/WNL.0b013e3181c97d99. [DOI] [PubMed] [Google Scholar]
- Duzhak T., Emerson M.R., Chakrabarty A., Alterman M.A., Levine S.M. Analysis of protein induction in the CNS of SJL mice with experimental allergic encephalomyelitis by proteomic screening and immunohistochemistry. Cell. Mol. Biol. (Noisy-le-grand) 2003;49:723–732. [PubMed] [Google Scholar]
- Dziedzic T., Metz I., Dallenga T., König F.B., Müller S., Stadelmann C., Brück W. Wallerian degeneration: a major component of early axonal pathology in multiple sclerosis. Brain Pathol. 2010;(April 14) doi: 10.1111/j.1750-3639.2010.00401.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edan G., Morrissey S.P., Hartung H.P. Use of mitoxantrone to treat multiple sclerosis. In: Cohen J.A., Rudick R.A., editors. Multiple Sclerosis Therapeutics. Informa Healthcare; London: 2007. pp. 457–480. [Google Scholar]
- Einstein E.R., Robertson D.M., DiCaprio J.M., Moore W. The isolation from bovine spinal cord of a homogeneous protein with encephalitogenic activity. J. Neurochem. 1962;9:353–361. doi: 10.1111/j.1471-4159.1962.tb09461.x. [DOI] [PubMed] [Google Scholar]
- Einstein O., Grigoriadis N., Mizrachi-Kol R., Reinhartz E., Polyzoidou E., Lavon I., Milonas I., Karussis D., Abramsky O., Ben-Hur T. Transplanted neural precursor cells reduce brain inflammation to attenuate chronic experimental autoimmune encephalomyelitis. Exp. Neurol. 2006;198:275–284. doi: 10.1016/j.expneurol.2005.11.007. [DOI] [PubMed] [Google Scholar]
- Einstein O., Friedman-Levi Y., Grigoriadis N., Ben-Hur T. Transplanted neural precursors enhance host brain-derived myelin regeneration. J. Neurosci. 2009;29:15694–15702. doi: 10.1523/JNEUROSCI.3364-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elkabes S., Li H. Proteomic strategies in multiple sclerosis and its animal models. Proteomics Clin. Appl. 2007;1:1393–1405. doi: 10.1002/prca.200700315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Encinas J.A., Weiner H.L., Kuchroo V.K. Inheritance of susceptibility to experimental autoimmune encephalomyelitis. J. Neurosci. Res. 1996;45:655–669. doi: 10.1002/(SICI)1097-4547(19960915)45:6<655::AID-JNR2>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- Ephrem A., Chamat S., Miquel C., Fisson S., Mouthon L., Caligiuri G., Delignat S., Elluru S., Bayry J., Lacroix-Desmazes S., Cohen J.L., Salomon B.L., Kazatchkine M.D., Kaveri S.V., Misra N. Expansion of CD4+CD25+ regulatory T cells by intravenous immunoglobulin: a critical factor in controlling experimental autoimmune encephalomyelitis. Blood. 2008;111:715–722. doi: 10.1182/blood-2007-03-079947. [DOI] [PubMed] [Google Scholar]
- Espejo C., Penkowa M., Demestre M., Montalban X., Martínez-Cáceres E.M. Time-course expression of CNS inflammatory, neurodegenerative tissue repair markers and metallothioneins during experimental autoimmune encephalomyelitis. Neuroscience. 2005;132:1135–1349. doi: 10.1016/j.neuroscience.2005.01.057. [DOI] [PubMed] [Google Scholar]
- Evans C.F., Horwitz M.S., Hobbs M.V., Oldstone M.B. Viral infection of transgenic mice expressing a viral protein in oligodendrocytes leads to chronic central nervous system autoimmune disease. J. Exp. Med. 1996;184:2371–2384. doi: 10.1084/jem.184.6.2371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eylar E.H., Caccam J., Jackson J.J., Westall F.C., Robinson A.B. Experimental allergic encephalomyelitis: synthesis of disease-inducing site of the basic protein. Science. 1970;168:1220–1223. doi: 10.1126/science.168.3936.1220. [DOI] [PubMed] [Google Scholar]
- Faria A.M., Weiner H.L. Oral tolerance: therapeutic implications for autoimmune diseases. Clin. Dev. Immunol. 2006;13:143–157. doi: 10.1080/17402520600876804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fathallah-Shaykh H.M. Microarrays: applications and pitfalls. Arch. Neurol. 2005;62:1669–1672. doi: 10.1001/archneur.62.11.1669. [DOI] [PubMed] [Google Scholar]
- Fernald G.H., Yeh R.F., Hauser S.L., Oksenberg J.R., Baranzini S.E. Mapping gene activity in complex disorders: integration of expression and genomic scans for multiple sclerosis. J. Neuroimmunol. 2005;167:157–169. doi: 10.1016/j.jneuroim.2005.06.032. [DOI] [PubMed] [Google Scholar]
- Fitzgerald D.C., Ciric B., Touil T., Harle H., Grammatikopolou J., Das Sarma J., Gran B., Zhang G.X., Rostami A. Suppressive effect of IL-27 on encephalitogenic Th17 cells and the effector phase of experimental autoimmune encephalomyelitis. J. Immunol. 2007;179:3268–3275. doi: 10.4049/jimmunol.179.5.3268. [DOI] [PubMed] [Google Scholar]
- Foley T.T., Hentosh P., Walters D.E. 2-Chloro-2′-deoxyadenosine: alteration of DNA:TATA element binding protein (TBP) interactions. J. Mol. Model. 2004;10:32–37. doi: 10.1007/s00894-003-0163-8. [DOI] [PubMed] [Google Scholar]
- Foster C.A., Mechtcheriakova D., Storch M.K., Balatoni B., Howard L.M., Bornancin F., Wlachos A., Sobanov J., Kinnunen A., Baumruker T. FTY720 rescue therapy in the Dark Agouti rat model of experimental autoimmune encephalomyelitis: expression of central nervous system genes and reversal of blood–brain-barrier damage. Brain Pathol. 2009;19:254–266. doi: 10.1111/j.1750-3639.2008.00182.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox R.J., Kinkel R.P. High-dose methylprednisolone in the treatment of multiple sclerosis. In: Cohen J.A., Rudick R.A., editors. Multiple Sclerosis Therapeutics. Informa Healthcare; London: 2007. pp. 515–533. [Google Scholar]
- Franciotta D., Salvetti M., Lolli F., Serafini B., Aloisi F. B cells and multiple sclerosis. Lancet Neurol. 2008;7:852–858. doi: 10.1016/S1474-4422(08)70192-3. [DOI] [PubMed] [Google Scholar]
- Franklin R.J., Ffrench-Constant C. Remyelination in the CNS: from biology to therapy. Nat. Rev. Neurosci. 2008;9:839–855. doi: 10.1038/nrn2480. [DOI] [PubMed] [Google Scholar]
- Freund J., McDermott K. Sensitisation to horse serum by means of adjuvants. Proc. Soc. Exp. Biol. 1942;49:548–553. [Google Scholar]
- Freund J., Stern E.R., Pisani T.M. Isoallergic encephalomyelitis and radiculitis in guinea pigs after one injection of brain and mycobacteria in water-in-oil emulsion. J. Immunol. 1947;57:179–194. [PubMed] [Google Scholar]
- Friese M.A., Jakobsen K.B., Friis L., Etzensperger R., Craner M.J., McMahon R.M., Jensen L.T., Huygelen V., Jones E.Y., Bell J.I., Fugger L. Opposing effects of HLA class I molecules in tuning autoreactive CD8+ T cells in multiple sclerosis. Nat. Med. 2008;14:1227–1235. doi: 10.1038/nm.1881. [DOI] [PubMed] [Google Scholar]
- Friese M.A., Fugger L. Pathogenic CD8+ T cells in multiple sclerosis. Ann. Neurol. 2009;66:132–141. doi: 10.1002/ana.21744. [DOI] [PubMed] [Google Scholar]
- Furlan R., Pluchino S., Martino G. Gene therapy-mediated modulation of immune processes in the central nervous system. Curr. Pharm. Des. 2003;9:2002–2008. doi: 10.2174/1381612033454315. [DOI] [PubMed] [Google Scholar]
- Furtado G.C., Piña B., Tacke F., Gaupp S., van Rooijen N., Moran T.M., Randolph G.J., Ransohoff R.M., Chensue S.W., Raine C.S., Lira S.A. A novel model of demyelinating encephalomyelitis induced by monocytes and dendritic cells. J. Immunol. 2006;177:6871–6879. doi: 10.4049/jimmunol.177.10.6871. [DOI] [PubMed] [Google Scholar]
- Garay L., Gonzalez Deniselle M.C., Gierman L., Meyer M., Lima A., Roig P., De Nicola A.F. Steroid protection in the experimental autoimmune encephalomyelitis model of multiple sclerosis. Neuroimmunomodulation. 2008;15:76–83. doi: 10.1159/000135627. [DOI] [PubMed] [Google Scholar]
- Gauthier S.A., Weiner H.L. Use of cyclophosphamide and other immunosuppressants to treat multiple sclerosis. In: Cohen J.A., Rudick R.A., editors. Multiple Sclerosis Therapeutics. Informa Healthcare; London: 2007. pp. 481–498. [Google Scholar]
- Gavériaux-Ruff C., Kieffer B.L. Conditional gene targeting in the mouse nervous system: Insights into brain function and diseases. Pharmacol. Ther. 2007;113:619–634. doi: 10.1016/j.pharmthera.2006.12.003. [DOI] [PubMed] [Google Scholar]
- Genain C.P., Nguyen M.H., Letvin N.L., Pearl R., Davis R.L., Adelman M., Lees M.B., Linington C., Hauser S.L. Antibody facilitation of multiple sclerosis-like lesions in a nonhuman primate. J. Clin. Invest. 1995;96:2966–2974. doi: 10.1172/JCI118368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giovannoni G., Comi G., Cook S., Rammohan K., Rieckmann P., Soelberg Sorensen P., Vermersch P., Chang P., Hamlett A., Musch B., Greenberg S.J., CLARITY Study Group A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis. N. Engl. J. Med. 2010;362:416–426. doi: 10.1056/NEJMoa0902533. [DOI] [PubMed] [Google Scholar]
- Goertsches R., Zettl U.K. MS therapy research applying genome-wide RNA profiling of peripheral blood. Int. MS J. 2007;14:98–107. [PubMed] [Google Scholar]
- Gold R., Linington C., Lassmann H. Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalomyelitis research. Brain. 2006;129:1953–1971. doi: 10.1093/brain/awl075. [DOI] [PubMed] [Google Scholar]
- Gold R., Lühder F. Interleukin-17—extended features of a key player in multiple sclerosis. Am. J. Pathol. 2008;172:8–10. doi: 10.2353/ajpath.2008.070862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodin D.S., Frohman E.M., Garmany G.P., Halper J., Likosky W.H., Lublin F.D., Silberberg D.H., Stuart W.H., van den Noort S., Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology, the MS Council for Clinical Practice Guidelines Disease modifying therapies in multiple sclerosis: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the MS Council for Clinical Practice Guidelines. Neurology. 2002;58:169–178. doi: 10.1212/wnl.58.2.169. [DOI] [PubMed] [Google Scholar]
- Goodkin D.E., Rudick R.A., VanderBrug Medendorp S., Daughtry M.M., Schwetz K.M., Fischer J., Van Dyke C. Low-dose (7.5 mg) oral methotrexate reduces the rate of progression in chronic progressive multiple sclerosis. Ann. Neurol. 1995;37:30–40. doi: 10.1002/ana.410370108. [DOI] [PubMed] [Google Scholar]
- Gould K.E., Stepaniak J.A., Swanborg R.H. Variable susceptibility of Lewis rats to experimental autoimmune encephalomyelitis. J. Neuroimmunol. 1994;54:145–146. doi: 10.1016/0165-5728(94)90240-2. [DOI] [PubMed] [Google Scholar]
- Goverman J., Woods A., Larson L., Weiner L.P., Hood L., Zaller D.M. Transgenic mice that express a myelin basic protein-specific T cell receptor develop spontaneous autoimmunity. Cell. 1993;72:551–560. doi: 10.1016/0092-8674(93)90074-z. [DOI] [PubMed] [Google Scholar]
- Goverman J. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 2009;9:393–407. doi: 10.1038/nri2550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory S.G., Schmidt S., Seth P., Oksenberg J.R., Hart J., Prokop A., Caillier S.J., Ban M., Goris A., Barcellos L.F., Lincoln R., McCauley J.L., Sawcer S.J., Compston D.A., Dubois B., Hauser S.L., Garcia-Blanco M.A., Pericak-Vance M.A., Haines J.L., Multiple Sclerosis Genetics Group Interleukin 7 receptor alpha chain (IL7R) shows allelic and functional association with multiple sclerosis. Nat. Genet. 2007;39:1083–1091. doi: 10.1038/ng2103. [DOI] [PubMed] [Google Scholar]
- Haak S., Croxford A.L., Kreymborg K., Heppner F.L., Pouly S., Becher B., Waisman A. IL-17A and IL-17F do not contribute vitally to autoimmune neuro-inflammation in mice. J. Clin. Invest. 2009;119:61–69. doi: 10.1172/JCI35997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haase C.G., Guggenmos J., Brehm U., Andersson M., Olsson T., Reindl M., Schneidewind J.M., Zettl U.K., Heidenreich F., Berger T., Wekerle H., Hohlfeld R., Linington C. The fine specificity of the myelin oligodendrocyte glycoprotein autoantibody response in patients with multiple sclerosis and normal healthy controls. J. Neuroimmunol. 2001;114:220–225. doi: 10.1016/s0165-5728(00)00462-8. [DOI] [PubMed] [Google Scholar]
- Hafler, J.P., Maier, L.M., Cooper, J.D., Plagnol, V., Hinks, A., Simmonds, M.J., Stevens, H.E., Walker, N.M., Healy, B., Howson, J.M., Maisuria, M., Duley, S., Coleman, G., Gough, S.C., International Multiple Sclerosis Genetics Consortium (IMSGC), Worthington, J., Kuchroo, V.K., Wicker, L.S., Todd, J.A., 2009. [226CD] Gly307Ser association with multiple autoimmune diseases. Genes Immun. 10, 5–10. [DOI] [PMC free article] [PubMed]
- Han M.H., Hwang S.I., Roy D.B., Lundgren D.H., Price J.V., Ousman S.S., Fernald G.H., Gerlitz B., Robinson W.H., Baranzini S.E., Grinnell B.W., Raine C.S., Sobel R.A., Han D.K., Steinman L. Proteomic analysis of active multiple sclerosis lesions reveals therapeutic targets. Nature. 2008;451:1076–1081. doi: 10.1038/nature06559. [DOI] [PubMed] [Google Scholar]
- Hartung H.P., Gonsette R., König N., Kwiecinski H., Guseo A., Morrissey S.P., Krapf H., Zwingers T., Mitoxantrone in Multiple Sclerosis Study Group (MIMS) Mitoxantrone in progressive multiple sclerosis: a placebo-controlled, double-blind, randomised, multicentre trial. Lancet. 2002;360:2018–2025. doi: 10.1016/S0140-6736(02)12023-X. [DOI] [PubMed] [Google Scholar]
- Hartung H.P., Kieseier B.C., Hemmer B. Purely systemically active anti-inflammatory treatments are adequate to control multiple sclerosis. J. Neurol. 2005;252(Suppl. 5):v30–v37. doi: 10.1007/s00415-005-5006-3. [DOI] [PubMed] [Google Scholar]
- Hartung H.P. New cases of progressive multifocal leukoencephalopathy after treatment with natalizumab. Lancet Neurol. 2009;8:28–31. doi: 10.1016/S1474-4422(08)70281-3. [DOI] [PubMed] [Google Scholar]
- Hartung H.P., Warnke C., Hohlfeld R., Kieseier B.C. Progressive multifocal leukoencephalopathy. Undesirable side effect of immunotherapy (German) Nervenarzt. 2009;80:1143–1144. doi: 10.1007/s00115-009-2701-9. 1146–1148, 1150–1153. [DOI] [PubMed] [Google Scholar]
- Hartung H.P., Aktas O., Kieseier B., Comi G. Development of oral cladribine for the treatment of multiple sclerosis. J. Neurol. 2010;257:207–211. doi: 10.1007/s00415-009-5359-0. [DOI] [PubMed] [Google Scholar]
- Hassen G.W., Feliberti J., Kesner L., Stracher A., Mokhtarian F. Prevention of axonal injury using calpain inhibitor in chronic progressive experimental autoimmune encephalomyelitis. Brain Res. 2008;1236:206–215. doi: 10.1016/j.brainres.2008.07.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauser S.L., Waubant E., Arnold D.L., Vollmer T., Antel J., Fox R.J., Bar-Or A., Panzara M., Sarkar N., Agarwal S., Langer-Gould A., Smith C.H., HERMES Trial Group B-cell depletion with rituximab in relapsing–remitting multiple sclerosis. N. Engl. J. Med. 2008;358:676–688. doi: 10.1056/NEJMoa0706383. [DOI] [PubMed] [Google Scholar]
- Hawker K., O‘Connor P., Freedman M.S., Calabresi P.A., Antel J., Simon J., Hauser S., Waubant E., Vollmer T., Panitch H., Zhang J., Chin P., Smith C.H., OLYMPUS Trial Group Rituximab in patients with primary progressive muoltiple sclerosis: results of a randomized, double-blind, placebo-controlled multicentre trial. Ann. Neurol. 2009;66:460–471. doi: 10.1002/ana.21867. [DOI] [PubMed] [Google Scholar]
- Hedstrom K.L., Xu X., Ogawa Y., Frischknecht R., Seidenbecher C.I., Shrager P., Rasband M.N. Neurofascin assembles a specialized extracellular matrix at the axon initial segment. J. Cell Biol. 2007;178:875–886. doi: 10.1083/jcb.200705119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemmer B., Hartung H.P. Toward the development of rational therapies in multiple sclerosis: what is on the horizon? Ann. Neurol. 2007;62:314–326. doi: 10.1002/ana.21289. [DOI] [PubMed] [Google Scholar]
- Hendriks J.J., Alblas J., van der Pol S.M., van Tol E.A., Dijkstra C.D., de Vries H.E. Flavonoids influence monocytic GTPase activity and are protective in experimental allergic encephalitis. J. Exp. Med. 2004;200:1667–1672. doi: 10.1084/jem.20040819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herder V., Hansmann F., Stangel M., Baumgärtner W., Beineke A. Cuprizone ameliorates spinal cord inflammation in a viral model of multiple sclerosis. Acta Neuropathol. 2009;118:443–444. (abstract) [Google Scholar]
- Herrero-Herranz E., Pardo L.A., Gold R., Linker R.A. Pattern of axonal injury in murine myelin oligodendrocyte glycoprotein induced experimental autoimmune encephalomyelitis: implications for multiple sclerosis. Neurobiol. Dis. 2008;30:162–173. doi: 10.1016/j.nbd.2008.01.001. [DOI] [PubMed] [Google Scholar]
- Herz J., Zipp F., Siffrin V. Neurodegeneration in autoimmune CNS inflammation. Exp. Neurol. 2009;(December 1) doi: 10.1016/j.expneurol.2009.11.019. [DOI] [PubMed] [Google Scholar]
- Hofstetter H.H., Grau C., Buttmann M., Forsthuber T.G., Gaupp S., Toyka K.V., Gold R. The PLP-specific T-cell population promoted by pertussis toxin is characterized by high frequencies of IL-17-producing cells. Cytokine. 2007;40:35–43. doi: 10.1016/j.cyto.2007.07.192. [DOI] [PubMed] [Google Scholar]
- Hofstetter H.H., Gold R., Hartung H.P. Th17 cells in MS and experimental autoimmune encephalomyelitis. Int. MS J. 2009;16:12–18. [PubMed] [Google Scholar]
- Hohlfeld R., Meinl E., Dornmair K. B- and T-cell responses in multiple sclerosis: novel approaches offer new insights. J. Neurol. Sci. 2008;274:5–8. doi: 10.1016/j.jns.2008.07.006. [DOI] [PubMed] [Google Scholar]
- Hoppenbrouwers I.A., Aulchenko Y.S., Ebers G.C., Ramagopalan S.V., Oostra B.A., van Duijn C.M., Hintzen R.Q. EVI5 is a risk gene for multiple sclerosis. Genes Immun. 2008;9:334–337. doi: 10.1038/gene.2008.22. [DOI] [PubMed] [Google Scholar]
- Hoppenbrouwers I.A., Aulchenko Y.S., Janssens A.C., Ramagopalan S.V., Broer L., Kayser M., Ebers G.C., Oostra B.A., van Duijn C.M., Hintzen R.Q. Replication of CD58 and CLEC16A as genome-wide significant risk genes for multiple sclerosis. J. Hum. Genet. 2009;54:676–680. doi: 10.1038/jhg.2009.96. [DOI] [PubMed] [Google Scholar]
- Hövelmeyer N., Hao Z., Kranidioti K., Kassiotis G., Buch T., Frommer F., von Hoch L., Kramer D., Minichiello L., Kollias G., Lassmann H., Waisman A. Apoptosis of oligodendrocytes via Fas and TNF-R1 is a key event in the induction of experimental autoimmune encephalomyelitis. J. Immunol. 2005;175:5875–5884. doi: 10.4049/jimmunol.175.9.5875. [DOI] [PubMed] [Google Scholar]
- Huber M., Heink S., Grothe H., Guralnik A., Reinhard K., Elflein K., Hünig T., Mittrücker H.W., Brüstle A., Kamradt T., Lohoff M. A Th17-like developmental process leads to CD8+ Tc17 cells with reduced cytotoxic activity. Eur. J. Immunol. 2009;39:1716–1725. doi: 10.1002/eji.200939412. [DOI] [PubMed] [Google Scholar]
- Hünig T. Manipulation of regulatory T-cell number and function with CD28-specific monoclonal antibodies. Adv. Immunol. 2007;95:111–148. doi: 10.1016/S0065-2776(07)95004-X. [DOI] [PubMed] [Google Scholar]
- Huppert J., Closhen D., Croxford A., White R., Kulig P., Pietrowski E., Bechmann I., Becher B., Luhmann H.J., Waisman A., Kuhlmann C.R. Cellular mechanisms of IL-17-induced blood–brain barrier disruption. FASEB J. 2010;24:1023–1034. doi: 10.1096/fj.09-141978. [DOI] [PubMed] [Google Scholar]
- Huseby E.S., Liggitt D., Brabb T., Schnabel B., Ohlén C., Goverman J. A pathogenic role for myelin-specific CD8+ T cells in a model for multiple sclerosis. J. Exp. Med. 2001;194:669–676. doi: 10.1084/jem.194.5.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ibrahim S.M., Mix E., Böttcher T., Koczan D., Gold R., Rolfs A., Thiesen H.J. Gene expression profiling of the nervous system in murine experimental autoimmune encephalomyelitis. Brain. 2001;124:1927–1938. doi: 10.1093/brain/124.10.1927. [DOI] [PubMed] [Google Scholar]
- Ibrahim S.M., Gold R. Genomics, proteomics, metabolomics: what is in a word for multiple sclerosis? Curr. Opin. Neurol. 2005;18:231–235. doi: 10.1097/01.wco.0000169738.06664.3b. [DOI] [PubMed] [Google Scholar]
- Imitola J., Chitnis T., Khoury S.J. Cytokines in multiple sclerosis: from bench to bedside. Pharmacol. Ther. 2005;106:163–177. doi: 10.1016/j.pharmthera.2004.11.007. [DOI] [PubMed] [Google Scholar]
- Hafler D.A., Compston A., Sawcer S., Lander E.S., Daly M.J., De Jager P.L., de Bakker P.I., Gabriel S.B., Mirel D.B., Ivinson A.J., Pericak-Vance M.A., Gregory S.G., Rioux J.D., McCauley J.L., Haines J.L., Barcellos L.F., Cree B., Oksenberg J.R., Hauser S.L., IMSGC International Multiple Sclerosis Genetics Consortium Risk alleles for multiple sclerosis identified by a genomewide study. N. Engl. J. Med. 2007;357:851–862. doi: 10.1056/NEJMoa073493. [DOI] [PubMed] [Google Scholar]
- Jacobs L.D., Beck R.W., Simon J.H., Kinkel R.P., Brownscheidle C.M., Murray T.J., Simonian N.A., Slasor P.J., Sandrock A.W. Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. CHAMPS Study Group. N. Engl. J. Med. 2000;343:898–904. doi: 10.1056/NEJM200009283431301. [DOI] [PubMed] [Google Scholar]
- Jäger A., Dardalhon V., Sobel R.A., Bettelli E., Kuchroo V.K. Th1, Th17, and Th9 effector cells induce experimental autoimmune encephalomyelitis with different pathological phenotypes. J. Immunol. 2009;183:7169–7177. doi: 10.4049/jimmunol.0901906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jagodic M., Kornek B., Weissert R., Lassmann H., Olsson T., Dahlman I. Congenic mapping confirms a locus on rat chromosome 10 conferring strong protection against myelin oligodendrocyte glycoprotein-induced experimental autoimmune encephalomyelitis. Immunogenetics. 2001;53:410–415. doi: 10.1007/s002510100342. [DOI] [PubMed] [Google Scholar]
- Jagodic M., Olsson T. Combined-cross analysis of genome-wide linkage scans for experimental autoimmune encephalomyelitis in rat. Genomics. 2006;88:737–744. doi: 10.1016/j.ygeno.2006.08.013. [DOI] [PubMed] [Google Scholar]
- Jelinsky S.A., Miyashiro J.S., Saraf K.A., Tunkey C., Reddy P., Newcombe J., Oestreicher J.L., Brown E., Trepicchio W.L., Leonard J.P., Marusic S. Exploiting genotypic differences to identify genes important for EAE development. J. Neurol. Sci. 2005;239:81–93. doi: 10.1016/j.jns.2005.08.008. [DOI] [PubMed] [Google Scholar]
- Johnson B.A., Wang J., Taylor E.M., Caillier S.J., Herbert J., Khan O.A., Cross A.H., de Jager P.L., Gourraud P.A., Cree B.C., Hauser S.L., Oksenberg J.R. Multiple sclerosis susceptibility alleles in African Americans. Genes Immun. 2010;11:343–350. doi: 10.1038/gene.2009.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones J.L., Coles A.J. Campath-1H treatment of multiple sclerosis. Neurodegener. Dis. 2008;5:27–31. doi: 10.1159/000109935. [DOI] [PubMed] [Google Scholar]
- Jones R.E., Moes N., Zwickey H., Cunningham C.L., Gregory W.L., Oken B. Treatment of experimental autoimmune encephalomyelitis with alpha lipoic acid and associative conditioning. Brain Behav. Immun. 2008;22:538–543. doi: 10.1016/j.bbi.2007.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jørgensen S.H., Storm N., Jensen P.E., Laursen H., Sørensen P.S. IVIG enters the central nervous system during treatment of experimental autoimmune encephalomyelitis and is localised to inflammatory lesions. Exp. Brain Res. 2007;178:462–469. doi: 10.1007/s00221-006-0752-8. [DOI] [PubMed] [Google Scholar]
- Kabat E.A., Wolf A., Bezer A.E. The rapid production of acute disseminated encephalomyelitis in rhesus monkeys by injection of heterologous and homologous brain tissue with adjuvants. J. Exp. Med. 1947;85:117–130. doi: 10.1084/jem.85.1.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanter J.L., Narayana S., Ho P.P., Catz I., Warren K.G., Sobel R.A., Steinman L., Robinson W.H. Lipid microarrays identify key mediators of autoimmune brain inflammation. Nat. Med. 2006;12:138–143. doi: 10.1038/nm1344. [DOI] [PubMed] [Google Scholar]
- Kappos L., Patzold U., Dommasch D., Poser S., Haas J., Krauseneck P., Malin J.P., Fierz W., Graffenried B.U., Gugerli U.S. Cyclosporine versus azathioprine in the long-term treatment of multiple sclerosis—results of the German multicenter study. Ann. Neurol. 1988;23:56–63. doi: 10.1002/ana.410230110. [DOI] [PubMed] [Google Scholar]
- Kappos L., Comi G., Panitch H., Oger J., Antel J., Conlon P., Steinman L. Induction of a non-encephalitogenic type 2 T helper-cell autoimmune response in multiple sclerosis after administration of an altered peptide ligand in a placebo-controlled, randomized phase II trial. The Altered Peptide Ligand in Relapsing MS Study Group. Nat. Med. 2000;6:1176–1182. doi: 10.1038/80525. [DOI] [PubMed] [Google Scholar]
- Kappos L., Polman C.H., Freedman M.S., Edan G., Hartung H.P., Miller D.H., Montalban X., Barkhof F., Bauer L., Jakobs P., Pohl C., Sandbrink R. Treatment with interferon beta-1b delays conversion to clinically definite and McDonald MS in patients with clinically isolated syndromes. Neurology. 2006;67:1242–1249. doi: 10.1212/01.wnl.0000237641.33768.8d. [DOI] [PubMed] [Google Scholar]
- Kappos L., Antel J., Comi G., Montalban X., O’Connor P., Polman C.H., Haas T., Korn A.A., Karlsson G., Radue E.W., FTY720 D2201 Study Group Oral fingolimod (FTY720) for relapsing multiple sclerosis. N. Engl. J. Med. 2006;355:1124–1140. doi: 10.1056/NEJMoa052643. [DOI] [PubMed] [Google Scholar]
- Kappos L., Lindberg R.L.P. Interferons in relapsing–remitting multiple sclerosis. In: Cohen J.A., Rudick R.A., editors. Multiple Sclerosis Therapeutics. Informa Healthcare; London: 2007. pp. 373–392. [Google Scholar]
- Kappos L., Freedman M.S., Polman C.H., Edan G., Hartung H.P., Miller D.H., Montalbán X., Barkhof F., Radü E.W., Bauer L., Dahms S., Lanius V., Pohl C., Sandbrink R., BENEFIT Study Group Effect of early versus delayed interferon beta-1b treatment on disability after a first clinical event suggestive of multiple sclerosis: a 3-year follow-up analysis of the BENEFIT study. Lancet. 2007;370:389–397. doi: 10.1016/S0140-6736(07)61194-5. [DOI] [PubMed] [Google Scholar]
- Kappos L., Gold R., Miller D.H., Macmanus D.G., Havrdova E., Limmroth V., Polman C.H., Schmierer K., Yousry T.A., Yang M., Eraksoy M., Meluzinova E., Rektor I., Dawson K.T., Sandrock A.W., O’Neill G.N., BG-12 Phase IIb Study Investigators Efficacy and safety of oral fumarate in patients with relapsing–remitting multiple sclerosis: a multicentre, randomised, double-blind, placebo-controlled phase IIb study. Lancet. 2008;372:1463–1472. doi: 10.1016/S0140-6736(08)61619-0. [DOI] [PubMed] [Google Scholar]
- Karpus W.J., Reynolds N., Behanna H.A., Van Eldik L.J., Watterson D.M. Inhibition of experimental autoimmune encephalomyelitis by a novel small molecular weight proinflammatory cytokine suppressing drug. J. Neuroimmunol. 2008;203:73–78. doi: 10.1016/j.jneuroim.2008.06.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keever-Taylor C.A., Browning M.B., Johnson B.D., Truitt R.L., Bredeson C.N., Behn B., Tsao A. Rapamycin enriches for CD4+CD25+CD27+Foxp3+ regulatory T cells in ex vivo-expanded CD25-enriched products from healthy donors and patients with multiple sclerosis. Cytotherapy. 2007;9:144–157. doi: 10.1080/14653240601145223. [DOI] [PubMed] [Google Scholar]
- Kerschensteiner M., Meinl E., Hohlfeld R. Neuro-immune crosstalk in CNS diseases. Neuroscience. 2009;158:1122–1132. doi: 10.1016/j.neuroscience.2008.09.009. [DOI] [PubMed] [Google Scholar]
- Khare M., Mangalam A., Rodriguez M., David C.S. HLA DR and DQ interaction in myelin oligodendrocyte glycoprotein-induced experimental autoimmune encephalomyelitis in HLA class II transgenic mice. J. Neuroimmunol. 2005;169:1–12. doi: 10.1016/j.jneuroim.2005.07.023. [DOI] [PubMed] [Google Scholar]
- Kieseier B.C., Hartung H.P. Current disease-modifying therapies in multiple sclerosis. Sem. Neurol. 2003;23:133–146. doi: 10.1055/s-2003-41138. [DOI] [PubMed] [Google Scholar]
- Kieseier B.C., Wiendl H. Oral disease-modifying treatments for multiple sclerosis: the story so far. CNS Drugs. 2007;21:483–502. doi: 10.2165/00023210-200721060-00005. [DOI] [PubMed] [Google Scholar]
- Kipp M., Clarner T., Dang J., Copray S., Beyer C. The cuprizone animal model: new insights into an old story. Acta Neuropathol. 2009;118:723–736. doi: 10.1007/s00401-009-0591-3. [DOI] [PubMed] [Google Scholar]
- Kleinschmidt-DeMasters B.K., Tyler K.L. Progressive multifocal leukoencephalopathy complicating treatment with natalizumab and interferon beta-1a for multiple sclerosis. N. Engl. J. Med. 2005;353:369–374. doi: 10.1056/NEJMoa051782. [DOI] [PubMed] [Google Scholar]
- Kobayashi N., Kiptoo P., Kobayashi H., Ridwan R., Brocke S., Siahaan T.J. Prophylactic and therapeutic suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor. Clin. Immunol. 2008;129:69–79. doi: 10.1016/j.clim.2008.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kojima K., Wekerle H., Lassmann H., Berger T., Linington C. Induction of experimental autoimmune encephalomyelitis by CD4+ T cells specific for an astrocyte protein, S100 beta. J. Neural. Transm. Suppl. 1997;49:43–51. doi: 10.1007/978-3-7091-6844-8_5. [DOI] [PubMed] [Google Scholar]
- Koritschoner R., Schweinburg F. Klinsche und experimentelle Beobachtungen über Lähmungen nach Wutschutzimpfung. Z. Immunitäts-Forsch. 1925;42:217–283. [Google Scholar]
- Korn T., Bettelli E., Gao W., Awasthi A., Jäger A., Strom T.B., Oukka M., Kuchroo V.K. IL-21 initiates an alternative pathway to induce proinflammatory TH17 cells. Nature. 2007;448:484–487. doi: 10.1038/nature05970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krapf H., Morrissey S.P., Zenker O., Zwingers T., Gonsette R., Hartung H.P., MIMS Study Group Effect of mitoxantrone on MRI in progressive MS: results of the MIMS trial. Neurology. 2005;65:690–695. doi: 10.1212/01.wnl.0000174439.70369.7a. [DOI] [PubMed] [Google Scholar]
- Krishnamoorthy G., Holz A., Wekerle H. Experimental models of spontaneous autoimmune disease in the central nervous system. J. Mol. Med. 2007;85:1161–1173. doi: 10.1007/s00109-007-0218-x. [DOI] [PubMed] [Google Scholar]
- Krishnamoorthy G., Saxena A., Mars L.T., Domingues H.S., Mentele R., Ben-Nun A., Lassmann H., Dornmair K., Kurschus F.C., Liblau R.S., Wekerle H. Myelin-specific T cells also recognize neuronal autoantigen in a transgenic mouse model of multiple sclerosis. Nat. Med. 2009;15:626–632. doi: 10.1038/nm.1975. [DOI] [PubMed] [Google Scholar]
- Laatsch R.H., Kies M.W., Gordon S., Alvord E.C. The encephalomyelitic activity of myelin isolated by ultracentrifugation. J. Exp. Med. 1962;115:77–88. doi: 10.1084/jem.115.4.777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lafaille J.J., Nagashima K., Katsuki M., Tonegawa S. High incidence of spontaneous autoimmune encephalomyelitis in immunodeficient anti-myelin basic protein T cell receptor transgenic mice. Cell. 1994;78:399–408. doi: 10.1016/0092-8674(94)90419-7. [DOI] [PubMed] [Google Scholar]
- Lange F., Bajtner E., Rintisch C., Nandakumar K.S., Sack U., Holmdahl R. Methotrexate ameliorates T cell dependent autoimmune arthritis and encephalomyelitis but not antibody induced or fibroblast induced arthritis. Ann. Rheum. Dis. 2005;64:599–5605. doi: 10.1136/ard.2004.026120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langer-Gould A., Atlas S.W., Green A.J., Bollen A.W., Pelletier D. Progressive multifocal leukoencephalopathy in a patient treated with natalizumab. N. Engl. J. Med. 2005;353:375–381. doi: 10.1056/NEJMoa051847. [DOI] [PubMed] [Google Scholar]
- Langrish C.L., Chen Y., Blumenschein W.M., Mattson J., Basham B., Sedgwick J.D., McClanahan T., Kastelein R.A., Cua D.J. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 2005;201:233–240. doi: 10.1084/jem.20041257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavi E. Histopathology in coronavirus-induced demyelination. In: Lavi E., Constantinescu C.S., editors. Experimental Models of Multiple Sclerosis. Springer; New York: 2005. pp. 711–716. [Google Scholar]
- Lavi E., Constantinescu C.S., editors. Experimental Models of Multiple Sclerosis. Springer; New York: 2005. [Google Scholar]
- Lebar R., Lubetzki C., Vincent C., Lombrail P., Boutry J.M. The M2 autoantigen of central nervous system myelin, a glycoprotein present in oligodendrocyte membrane. Clin. Exp. Immunol. 1986;66:423–434. [PMC free article] [PubMed] [Google Scholar]
- Lees J.R., Iwakura Y., Russell J.H. Host T cells are the main producers of IL-17 within the central nervous system during initiation of experimental autoimmune encephalomyelitis induced by adoptive transfer of Th1 cell lines. J. Immunol. 2008;180:8066–8072. doi: 10.4049/jimmunol.180.12.8066. [DOI] [PubMed] [Google Scholar]
- Lennon V.A., Wilks A.V., Carnegie P.R. Immunologic properties of the main encephalitogenic peptide from the basic protein of human myelin. J. Immunol. 1970;105:1223–1230. [PubMed] [Google Scholar]
- Lenz D.C., Lu L., Conant S.B., Wolf N.A., Gérard H.C., Whittum-Hudson J.A., Hudson A.P., Swanborg R.H. A Chlamydia pneumoniae-specific peptide induces experimental autoimmune encephalomyelitis in rats. J. Immunol. 2001;167:1803–1808. doi: 10.4049/jimmunol.167.3.1803. [DOI] [PubMed] [Google Scholar]
- Lindsey J.W. EAE: history, clinical signs, and disease course. In: Lavi E., Constantinescu C.S., editors. Experimental Models of Multiple Sclerosis. Springer; New York: 2005. pp. 1–9. [Google Scholar]
- Lindå H., von Heijne A., Major E.O., Ryschkewitsch C., Berg J., Olsson T., Martin C. Progressive multifocal leukoencephalopathy after natalizumab monotherapy. N. Engl. J. Med. 2009;361:1081–1087. doi: 10.1056/NEJMoa0810316. [DOI] [PubMed] [Google Scholar]
- Link H. The cytokine storm in multiple sclerosis. Mult. Scler. 1998;4:12–15. doi: 10.1177/135245859800400104. [DOI] [PubMed] [Google Scholar]
- Linker R.A., Sendtner M., Gold R. Mechanisms of axonal degeneration in EAE—lessons from CNTF and MHC I knockout mice. J. Neurol. Sci. 2005;233:167–172. doi: 10.1016/j.jns.2005.03.021. [DOI] [PubMed] [Google Scholar]
- Linker R.A., Kruse N., Israel S., Wei T., Seubert S., Hombach A., Holtmann B., Lühder F., Ransohoff R.M., Sendtner M., Gold R. Leukemia inhibitory factor deficiency modulates the immune response and limits autoimmune demyelination: a new role for neurotrophic cytokines in neuroinflammation. J. Immunol. 2008;180:2204–2213. doi: 10.4049/jimmunol.180.4.2204. [DOI] [PubMed] [Google Scholar]
- Linker R.A., Weller C., Lühder F., Mohr A., Schmidt J., Knauth M., Metselaar J.M., Gold R. Liposomal glucocorticosteroids in treatment of chronic autoimmune demyelination: long-term protective effects and enhanced efficacy of methylprednisolone formulations. Exp. Neurol. 2008;211:397–406. doi: 10.1016/j.expneurol.2008.02.005. [DOI] [PubMed] [Google Scholar]
- Linker R.A., Kieseier B.C., Gold R. Identification and development of new therapeutics for multiple sclerosis. Trends Pharmacol. Sci. 2008;29:558–565. doi: 10.1016/j.tips.2008.07.012. [DOI] [PubMed] [Google Scholar]
- Linker R.A., Brechlin P., Jesse S., Steinacker P., Lee D.H., Asif A.R., Jahn O., Tumani H., Gold R., Otto M. Proteome profiling in murine models of multiple sclerosis: identification of stage specific markers and culprits for tissue damage. PLoS One. 2009;4:e7624. doi: 10.1371/journal.pone.0007624. (1–9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linker R.A., Gold R., Lühder F. Function of neurotrophic factors beyond the nervous system: inflammation and autoimmune demyelination. Crit. Rev. Immunol. 2009;29:43–68. doi: 10.1615/critrevimmunol.v29.i1.20. [DOI] [PubMed] [Google Scholar]
- Lipton M.M., Freund J. Encephalomyelitis in the rat following intracutaneous injection of central nervous system tissue with adjuvant. Proc. Soc. Exp. Biol. Med. 1952;81:260–261. doi: 10.3181/00379727-81-19840. [DOI] [PubMed] [Google Scholar]
- Littman D.R., Rudensky A.Y. Th17 and regulatory T cells in mediating and restraining inflammation. Cell. 2010;140:845–858. doi: 10.1016/j.cell.2010.02.021. [DOI] [PubMed] [Google Scholar]
- Liu T., Donahue K.C., Hu J., Kurnellas M.P., Grant J.E., Li H., Elkabes S. Identification of differentially expressed proteins in experimental autoimmune encephalomyelitis (EAE) by proteomic analysis of the spinal cord. J. Proteome Res. 2007;6:2565–2575. doi: 10.1021/pr070012k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X., Leung S., Wang C., Tan Z., Wang J., Guo T.B., Fang L., Zhao Y., Wan B., Qin X., Lu L., Li R., Pan H., Song M., Liu A., Hong J., Lu H., Zhang J.Z. Crucial role of interleukin-7 in T helper type 17 survival and expansion in autoimmune disease. Nat. Med. 2010;16:191–197. doi: 10.1038/nm.2077. [DOI] [PubMed] [Google Scholar]
- Lo A.C., Saab C.Y., Black J.A., Waxman S.G. Phenytoin protects spinal cord axons and preserves axonal conduction and neurological function in a model of neuroinflammation in vivo. J. Neurophysiol. 2003;90:3566–3571. doi: 10.1152/jn.00434.2003. [DOI] [PubMed] [Google Scholar]
- Lock C., Hermans G., Pedotti R., Brendolan A., Schadt E., Garren H., Langer-Gould A., Strober S., Cannella B., Allard J., Klonowski P., Austin A., Lad N., Kaminski N., Galli S.J., Oksenberg J.R., Raine C.S., Heller R., Steinman L. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat. Med. 2002;8:500–508. doi: 10.1038/nm0502-500. [DOI] [PubMed] [Google Scholar]
- Lovett-Racke A.E., Hussain R.Z., Northrop S., Choy J., Rocchini A., Matthes L., Chavis J.A., Diab A., Drew P.D., Racke M.K. Peroxisome proliferator-activated receptor α agonists as therapy for autoimmune disease. J. Immunol. 2004;172:5790–5798. doi: 10.4049/jimmunol.172.9.5790. [DOI] [PubMed] [Google Scholar]
- Lucchinetti C., Brück W., Parisi J., Scheithauer B., Rodriguez M., Lassmann H. Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann. Neurol. 2000;47:707–717. doi: 10.1002/1531-8249(200006)47:6<707::aid-ana3>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- Lühder F., Reichardt H.M. Traditional concepts and future avenues of glucocorticoid action in experimental autoimmune encephalomyelitis and multiple sclerosis therapy. Crit. Rev. Immunol. 2009;29:255–273. doi: 10.1615/critrevimmunol.v29.i3.50. [DOI] [PubMed] [Google Scholar]
- Luo J., Ho P., Steinman L., Wyss-Coray T. Bioluminescence in vivo imaging of autoimmune encephalomyelitis predicts disease. J. Neuroinflammation. 2008;5:6. doi: 10.1186/1742-2094-5-6. (1–6) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lutterotti A., Martin R. Getting specific: monoclonal antibodies in multiple sclerosis. Lancet Neurol. 2008;7:538–547. doi: 10.1016/S1474-4422(08)70110-8. [DOI] [PubMed] [Google Scholar]
- Makar T.K., Trisler D., Bever C.T., Goolsby J.E., Sura K.T., Balasubramanian S., Sultana S., Patel N., Ford D., Singh I.S., Gupta A., Valenzuela R.M., Dhib-Jalbut S. Stem cell based delivery of IFN-β reduces relapses in experimental autoimmune encephalomyelitis. J. Neuroimmunol. 2008;196:67–81. doi: 10.1016/j.jneuroim.2008.02.014. [DOI] [PubMed] [Google Scholar]
- Makar T.K., Trisler D., Sura K.T., Sultana S., Patel N., Bever C.T. Brain derived neurotrophic factor treatment reduces inflammation and apoptosis in experimental allergic encephalomyelitis. J. Neurol. Sci. 2008;270:70–76. doi: 10.1016/j.jns.2008.02.011. [DOI] [PubMed] [Google Scholar]
- Mangalam A.K., Rajagopalan G., Taneja V., David C.S. HLA class II transgenic mice mimic human inflammatory diseases. Adv. Immunol. 2008;97:65–147. doi: 10.1016/S0065-2776(08)00002-3. [DOI] [PubMed] [Google Scholar]
- Mangano K., Nicoletti A., Patti F., Donia M., Malaguarnera L., Signorelli S., Magro G., Muzio V., Greco B., Zaratin P., Meroni P., Zappia M., Nicoletti F. Variable effects of cyclophosphamide in rodent models of experimental allergic encephalomyelitis. Clin. Exp. Immunol. 2010;159:159–168. doi: 10.1111/j.1365-2249.2009.04050.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marrie R.A., Cohen J.A. Interferons in secondary progressive multiple sclerosis. In: Cohen J.A., Rudick R.A., editors. Multiple Sclerosis Therapeutics. Informa Healthcare; London: 2007. pp. 393–407. [Google Scholar]
- Martin Mdel P., Monson N.L. Potential role of humoral immunity in the pathogenesis of multiple sclerosis (MS) and experimental autoimmune encephalomyelitis (EAE) Front. Biosci. 2007;12:2735–2749. doi: 10.2741/2268. [DOI] [PubMed] [Google Scholar]
- Martino G., Pluchino S. Neural stem cells: guardians of the brain. Nat. Cell Biol. 2007;9:1031–1034. doi: 10.1038/ncb0907-1031. [DOI] [PubMed] [Google Scholar]
- Määttä J.A., Käldman M.S., Sakoda S., Salmi A.A., Hinkkanen A.E. Encephalitogenicity of myelin-associated oligodendrocytic basic protein and 2′,3′-cyclic nucleotide 3′-phosphodiesterase for BALB/c and SJL mice. Immunology. 1998;95:383–388. doi: 10.1046/j.1365-2567.1998.00605.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matà S., Lolli F., Söderström M., Pinto F., Link H. Multiple sclerosis is associated with enhanced B cell responses to the ganglioside GD1a. Mult. Scler. 1999;5:379–388. doi: 10.1177/135245859900500i603. [DOI] [PubMed] [Google Scholar]
- Matejuk A., Hopke C., Dwyer J., Subramanian S., Jones R.E., Bourdette D.N., Vandenbark A.A., Offner H. CNS gene expression pattern associated with spontaneous experimental autoimmune encephalomyelitis. J. Neurosci. Res. 2003;73:667–678. doi: 10.1002/jnr.10689. [DOI] [PubMed] [Google Scholar]
- Mathey E.K., Derfuss T., Storch M.K., Williams K.R., Hales K., Woolley D.R., Al-Hayani A., Davies S.N., Rasband M.N., Olsson T., Moldenhauer A., Velhin S., Hohlfeld R., Meinl E., Linington C. Neurofascin as a novel target for autoantibody-mediated axonal injury. J. Exp. Med. 2007;204:2363–2372. doi: 10.1084/jem.20071053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayo S., Quinn A. Altered susceptibility to EAE in congenic NOD mice: Altered processing of the encephalitogenic MOG35–55 peptide by NOR/LtJ mice. Clin. Immunol. 2007;122:91–100. doi: 10.1016/j.clim.2006.08.001. [DOI] [PubMed] [Google Scholar]
- Mazón Peláez I., Vogler S., Strauss U., Wernhoff P., Pahnke J., Brockmann G., Moch H., Thiesen H.J., Rolfs A., Ibrahim S.M. Identification of quantitative trait loci controlling cortical motor evoked potentials in experimental autoimmune encephalomyelitis: correlation with incidence, onset and severity of disease. Hum. Mol. Genet. 2005;14:1977–1989. doi: 10.1093/hmg/ddi203. [DOI] [PubMed] [Google Scholar]
- McCombe P.A., Harness J., Pender M.P. Effects of cyclosporin A treatment on clinical course and inflammatory cell apoptosis in experimental autoimmune encephalomyelitis induced in Lewis rats by inoculation with myelin basic protein. J. Neuroimmunol. 1999;97:60–69. doi: 10.1016/s0165-5728(99)00047-8. [DOI] [PubMed] [Google Scholar]
- McKenzie B.S., Kastelein R.A., Cua D.J. Understanding the IL-23-IL-17 immune pathway. Trends Immunol. 2006;27:17–23. doi: 10.1016/j.it.2005.10.003. [DOI] [PubMed] [Google Scholar]
- McLaughlin K.A., Wucherpfennig K.W. B cells and autoantibodies in the pathogenesis of multiple sclerosis and related inflammatory demyelinating diseases. Adv. Immunol. 2008;98:121–149. doi: 10.1016/S0065-2776(08)00404-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melzer N., Meuth S.G., Torres-Salazar D., Bittner S., Zozulya A.L., Weidenfeller C., Kotsiari A., Stangel M., Fahlke C., Wiendl H. A β-lactam antibiotic dampens excitotoxic inflammatory CNS damage in a mouse model of multiple sclerosis. PLoS One. 2008;3:e3149. doi: 10.1371/journal.pone.0003149. (1–12) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mendel I., Natarajan K., Ben-Nun A., Shevach E.M. A novel protective model against experimental allergic encephalomyelitis in mice expressing a transgenic TCR-specific for myelin oligodendrocyte glycoprotein. J. Neuroimmunol. 2004;149:10–21. doi: 10.1016/j.jneuroim.2003.12.007. [DOI] [PubMed] [Google Scholar]
- Merkler D., Ernsting T., Kerschensteiner M., Brück W., Stadelmann C. A new focal EAE model of cortical demyelination: multiple sclerosis-like lesions with rapid resolution of inflammation and extensive remyelination. Brain. 2006;129:1972–1983. doi: 10.1093/brain/awl135. [DOI] [PubMed] [Google Scholar]
- Mero I.L., Lorentzen A.R., Ban M., Smestad C., Celius E.G., Aarseth J.H., Myhr K.M., Link J., Hillert J., Olsson T., Kockum I., Masterman T., Oturai A.B., Søndergaard H.B., Sellebjerg F., Saarela J., Kemppinen A., Elovaara I., Spurkland A., Dudbridge F., Lie B.A., Harbo H.F. A rare variant of the TYK2 gene is confirmed to be associated with multiple sclerosis. Eur. J. Hum. Genet. 2010;18:502–504. doi: 10.1038/ejhg.2009.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meuth S.G., Bittner S., Meuth P., Simon O.J., Budde T., Wiendl H. TWIK-related acid-sensitive K+ channel 1 (TASK1) and TASK3 critically influence T lymphocyte effector functions. J. Biol. Chem. 2008;283:14559–14570. doi: 10.1074/jbc.M800637200. [DOI] [PubMed] [Google Scholar]
- Mi S., Hu B., Hahm K., Luo Y., Kam Hui E.S., Yuan Q., Wong W.M., Wang L., Su H., Chu T.H., Guo J., Zhang W., So K.F., Pepinsky B., Shao Z., Graff C., Garber E., Jung V., Wu E.X., Wu W. LINGO-1 antagonist promotes spinal cord remyelination and axonal integrity in MOG-induced experimental autoimmune encephalomyelitis. Nat. Med. 2007;13:1228–1233. doi: 10.1038/nm1664. [DOI] [PubMed] [Google Scholar]
- Mi S., Miller R.H., Tang W., Lee X., Hu B., Wu W., Zhang Y., Shields C.B., Zhang Y., Miklasz S., Shea D., Mason J., Franklin R.J., Ji B., Shao Z., Chédotal A., Bernard F., Roulois A., Xu J., Jung V., Pepinsky B. Promotion of central nervous system remyelination by induced differentiation of oligodendrocyte precursor cells. Ann. Neurol. 2009;65:304–315. doi: 10.1002/ana.21581. [DOI] [PubMed] [Google Scholar]
- Miron V.E., Schubart A., Antel J.P. Central nervous system-directed effects of FTY720 (fingolimod) J. Neurol. Sci. 2008;274:13–17. doi: 10.1016/j.jns.2008.06.031. [DOI] [PubMed] [Google Scholar]
- Miron V.E., Ludwin S.K., Darlington P.J., Jarjour A.A., Soliven B., Kennedy T.E., Antel J.P. Fingolimod (FTY720) enhances remyelination following demyelination of organotypic cerebellar slices. Am. J. Pathol. 2010;176:2682–2694. doi: 10.2353/ajpath.2010.091234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirowska-Guzel D. The role of neurotrophic factors in the pathology and treatment of multiple sclerosis. Immunopharmacol. Immunotoxicol. 2009;31:32–38. doi: 10.1080/08923970802379819. [DOI] [PubMed] [Google Scholar]
- Mix E., Olsson T., Correale J., Kostulas V., Link H. CD4+, CD8+, and CD4−CD8− T cells in CSF and blood of patients with multiple sclerosis and tension headache. Scand. J. Immunol. 1990;31:493–501. doi: 10.1111/j.1365-3083.1990.tb02797.x. [DOI] [PubMed] [Google Scholar]
- Mix E., Pahnke J., Ibrahim S.M. Gene-expression profiling of experimental autoimmune encephalomyelitis. Neurochem. Res. 2002;27:1157–1163. doi: 10.1023/a:1020925425780. [DOI] [PubMed] [Google Scholar]
- Mix E., Ibrahim S., Pahnke J., Koczan D., Sina C., Böttcher T., Thiesen H.J., Rolfs A. Gene-expression profiling of the early stages of MOG-induced EAE proves EAE-resistance as an active process. J. Neuroimmunol. 2004;151:158–170. doi: 10.1016/j.jneuroim.2004.03.007. [DOI] [PubMed] [Google Scholar]
- Mix E., Ibrahim S.M., Pahnke J., Glass A., Mazón-Peláez I., Lemcke S., Koczan D., Gimsa U., Bansemer S., Scheel T., Karopka T., Böttcher T., Müller J., Dazert E., Antipova V., Hoffrogge R., Wree A., Zschiesche M., Strauss U., Kundt G., Warzok R., Gierl L., Rolfs A. 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitor Atorvastatin mediated effects depend on the activation status of target cells in PLP–EAE. J. Autoimmun. 2006;27:251–265. doi: 10.1016/j.jaut.2006.09.006. [DOI] [PubMed] [Google Scholar]
- Mix E., Goertsches R., Zettl U.K. Immunology and neurology. J. Neurol. 2007;254(Suppl. 2):II2–II7. doi: 10.1007/s00415-007-2002-9. [DOI] [PubMed] [Google Scholar]
- Mix E., Meyer-Rienecker H., Zettl U.K. Animal models of multiple sclerosis for the development and validation of novel therapies. Potentials and limitations. J. Neurol. 2008;255(Suppl. 6):7–14. doi: 10.1007/s00415-008-6003-0. [DOI] [PubMed] [Google Scholar]
- Mörtl M., Sonderegger P., Diederichs K., Welte W. The crystal structure of the ligand-binding module of human TAG-1 suggests a new mode of homophilic interaction. Protein Sci. 2007;16:2174–2183. doi: 10.1110/ps.072802707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan I.M. Allergic encephalomyelitis in monkeys in response to injection of normal monkey nervous tissue. J. Exp. Med. 1947;85:131–140. doi: 10.1084/jem.85.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrissey S.P., Stodal H., Zettl U., Simonis C., Jung S., Kiefer R., Lassmann H., Hartung H.P., Haase A., Toyka K.V. In vivo MRI and its histological correlates in acute adoptive transfer experimental allergic encephalomyelitis. Quantification of inflammation and oedema. Brain. 1996;119:239–248. doi: 10.1093/brain/119.1.239. [DOI] [PubMed] [Google Scholar]
- Munoz J.J., Bernard C.C., Mackay I.R. Elicitation of experimental allergic encephalomyelitis (EAE) in mice with the aid of pertussigen. Cell. Immunol. 1984;83:92–100. doi: 10.1016/0008-8749(84)90228-4. [DOI] [PubMed] [Google Scholar]
- Nakano K., Higashi T., Hashimoto K., Takagi R., Tanaka Y., Matsushita S. Antagonizing dopamine D1-like receptor inhibits Th17 cell differentiation: preventive and therapeutic effects on experimental autoimmune encephalomyelitis. Biochem. Biophys. Res. Commun. 2008;373:286–291. doi: 10.1016/j.bbrc.2008.06.012. [DOI] [PubMed] [Google Scholar]
- Nath N., Giri S., Prasad R., Salem M.L., Singh A.K., Singh I. 5-aminoimidazole-4-carboxamide ribonucleoside: a novel immunomodulator with therapeutic efficacy in experimental autoimmune encephalomyelitis. J. Immunol. 2005;175:566–574. doi: 10.4049/jimmunol.175.1.566. [DOI] [PubMed] [Google Scholar]
- Neuhaus O., Wiendl H., Kieseier B.C., Archelos J.J., Hemmer B., Stüve O., Hartung H.P. Multiple sclerosis: mitoxantrone promotes differential effects in immunocompetent cells in vitro. J. Neuroimmunol. 2005;168:128–137. doi: 10.1016/j.jneuroim.2005.01.024. [DOI] [PubMed] [Google Scholar]
- Neuhaus O., Kieseier B.C., Hartung H.P. Therapeutic role of mitoxantrone in multiple sclerosis. Pharmacol. Ther. 2006;109:198–209. doi: 10.1016/j.pharmthera.2005.07.002. [DOI] [PubMed] [Google Scholar]
- Neuhaus O., Kieseier B.C., Hartung H.P. Mitoxantrone in multiple sclerosis. Adv. Neurol. 2006;98:293–302. [PubMed] [Google Scholar]
- Neuhaus O., Kieseier B.C., Hartung H.P. Immunosuppressive agents in multiple sclerosis. Neurotherapeutics. 2007;4:654–660. doi: 10.1016/j.nurt.2007.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noseworthy J.H., O’Brien P., Erickson B.J., Lee D., Sneve D., Ebers G.C., Rice G.P., Auty A., Hader W.J., Kirk A., Duquette P., Carter J., Francis G., Metz L., Shuster E. The Mayo Clinic-Canadian Cooperative trial of sulfasalazine in active multiple sclerosis. Neurology. 1998;51:1342–1352. doi: 10.1212/wnl.51.5.1342. [DOI] [PubMed] [Google Scholar]
- Noseworthy J.H., Wolinsky J.S., Lublin F.D., Whitaker J.N., Linde A., Gjorstrup P., Sullivan H.C. Linomide in relapsing and secondary progressive MS: part I: trial design and clinical results. North American Linomide Investigators. Neurology. 2000;54:1726–1733. doi: 10.1212/wnl.54.9.1726. [DOI] [PubMed] [Google Scholar]
- Nowak E.C., Weaver C.T., Turner H., Begum-Haque S., Becher B., Schreiner B., Coyle A.J., Kasper L.H., Noelle R.J. IL-9 as a mediator of Th17-driven inflammatory disease. J. Exp. Med. 2009;206:1653–1660. doi: 10.1084/jem.20090246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Connor P.W., Li D., Freedman M.S., Bar-Or A., Rice G.P., Confavreux C., Paty D.W., Stewart J.A., Scheyer R., Teriflunomide Multiple Sclerosis Trial Group, University of British Columbia MS/MRI Research Group A Phase II study of the safety and efficacy of teriflunomide in multiple sclerosis with relapses. Neurology. 2006;66:894–900. doi: 10.1212/01.wnl.0000203121.04509.31. [DOI] [PubMed] [Google Scholar]
- Ohler B., Graf K., Bragg R., Lemons T., Coe R., Genain C., Israelachvili J., Husted C. Role of lipid interactions in autoimmune demyelination. Biochim. Biophys. Acta. 2004;1688:10–17. doi: 10.1016/j.bbadis.2003.10.001. [DOI] [PubMed] [Google Scholar]
- Olitzky P.K., Yager R.H. Experimental disseminated encephalomyelitis in white mice. J. Exp. Med. 1949;90:213–224. doi: 10.1084/jem.90.3.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsson T., Dahlman I., Wallström E., Weissert R., Piehl F. Genetics of rat neuroinflammation. J. Neuroimmunol. 2000;107:191–200. doi: 10.1016/s0165-5728(00)00224-1. [DOI] [PubMed] [Google Scholar]
- Ousman S.S., Tomooka B.H., van Noort J.M., Wawrousek E.F., O’Connor K.C., Hafler D.A., Sobel R.A., Robinson W.H., Steinman L. Protective and therapeutic role for αB-crystallin in autoimmune demyelination. Nature. 2007;448:474–479. doi: 10.1038/nature05935. [DOI] [PubMed] [Google Scholar]
- Owens T., Wekerle H., Antel J. Genetic models for CNS inflammation. Nat. Med. 2001;7:161–166. doi: 10.1038/84603. [DOI] [PubMed] [Google Scholar]
- Ozenci V., Kouwenhoven M., Link H. Cytokines in multiple sclerosis: methodological aspects and pathogenic implications. Mult. Scler. 2002;8:396–404. doi: 10.1191/1352458502ms837rr. [DOI] [PubMed] [Google Scholar]
- Paintlia A.S., Paintlia M.K., Singh A.K., Stanislaus R., Gilg A.G., Barbosa E., Singh I. Regulation of gene expression associated with acute experimental autoimmune encephalomyelitis by Lovastatin. J. Neurosci. Res. 2004;77:63–81. doi: 10.1002/jnr.20130. [DOI] [PubMed] [Google Scholar]
- Paintlia A.S., Paintlia M.K., Singh I., Singh A.K. Immunomodulatory effect of combination therapy with lovastatin and 5-aminoimidazole-4-carboxamide-1-beta-d-ribofuranoside alleviates neurodegeneration in experimental autoimmune encephalomyelitis. Am. J. Pathol. 2006;169:1012–1025. doi: 10.2353/ajpath.2006.051309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paintlia A.S., Paintlia M.K., Singh I., Singh A.K. Combined medication of lovastatin with rolipram suppresses severity of experimental autoimmune encephalomyelitis. Exp. Neurol. 2008;214:168–180. doi: 10.1016/j.expneurol.2008.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paintlia A.S., Paintlia M.K., Singh I., Skoff R.B., Singh A.K. Combination therapy of lovastatin and rolipram provides neuroprotection and promotes neurorepair in inflammatory demyelination model of multiple sclerosis. Glia. 2009;57:182–193. doi: 10.1002/glia.20745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papadopoulos D., Rundle J., Patel R., Marshall I., Stretton J., Eaton R., Richardson J.C., Gonzalez M.I., Philpott K.L., Reynolds R. FTY720 ameliorates MOG-induced experimental autoimmune encephalomyelitis by suppressing both cellular and humoral immune responses. J. Neurosci. Res. 2010;88:346–359. doi: 10.1002/jnr.22196. [DOI] [PubMed] [Google Scholar]
- Pasteur L., Illo J. Pasteur and rabies: an interview of 1882. Med. Hist. 1996;40:373–377. doi: 10.1017/s0025727300061354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paterson P.Y. Transfer of allergic encephalomyelitis in rats by means of lymph node cells. J. Exp. Med. 1960;111:119–136. doi: 10.1084/jem.111.1.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul C., Bolton C. Inhibition of blood–brain barrier disruption in experimental allergic encephalomyelitis by short-term therapy with dexamethasone or cyclosporin A. Int. J. Immunopharmacol. 1995;17:497–503. doi: 10.1016/0192-0561(95)00034-y. [DOI] [PubMed] [Google Scholar]
- Pedemonte E., Benvenuto F., Casazza S., Mancardi G., Oksenberg J.R., Uccelli A., Baranzini S.E. The molecular signature of therapeutic mesenchymal stem cells exposes the architecture of the hematopoietic stem cell niche synapse. BMC Genomics. 2007;8:65. doi: 10.1186/1471-2164-8-65. (1–14) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pender M.P., Csurhes P.A., Wolfe N.P., Hooper K.D., Good M.F., McCombe P.A., Greer J.M. Increased circulating T cell reactivity to GM3 and GQ1b gangliosides in primary progressive multiple sclerosis. J. Clin. Neurosci. 2003;10:63–66. doi: 10.1016/s0967-5868(02)00270-9. [DOI] [PubMed] [Google Scholar]
- Pedersen L.B., Nashold F.E., Spach K.M., Hayes C.E. 1,25-dihydroxyvitamin D3 reverses experimental autoimmune encephalomyelitis by inhibiting chemokine synthesis and monocyte trafficking. J. Neurosci. Res. 2007;85:2480–2490. doi: 10.1002/jnr.21382. [DOI] [PubMed] [Google Scholar]
- Pernet V., Joly S., Christ F., Dimou L., Schwab M.E. Nogo-A and myelin-associated glycoprotein differently regulate oligodendrocyte maturation and myelin formation. J. Neurosci. 2008;28:7435–7444. doi: 10.1523/JNEUROSCI.0727-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platten M., Ho P.P., Youssef S., Fontoura P., Garren H., Hur E.M., Gupta R., Lee L.Y., Kidd B.A., Robinson W.H., Sobel R.A., Selley M.L., Steinman L. Treatment of autoimmune neuroinflammation with a synthetic tryptophan metabolite. Science. 2005;310:850–855. doi: 10.1126/science.1117634. [DOI] [PubMed] [Google Scholar]
- Plioplys A.V., Massimini N. Alpha/beta interferon is a neuronal growth factor. Neuroimmunomodulation. 1995;2:31–35. doi: 10.1159/000096838. [DOI] [PubMed] [Google Scholar]
- Pluchino S., Martino G. The therapeutic plasticity of neural stem/precursor cells in multiple sclerosis. J. Neurol. Sci. 2008;265:105–110. doi: 10.1016/j.jns.2007.07.020. [DOI] [PubMed] [Google Scholar]
- Podbielska M., Hogan E.L. Molecular and immunogenic features of myelin lipids: incitants or modulators of multiple sclerosis? Mult. Scler. 2009;15:1011–1029. doi: 10.1177/1352458509106708. [DOI] [PubMed] [Google Scholar]
- Pöllinger B., Krishnamoorthy G., Berer K., Lassmann H., Bösl M.R., Dunn R., Domingues H.S., Holz A., Kurschus F.C., Wekerle H. Spontaneous relapsing–remitting EAE in the SJL/J mouse: MOG-reactive transgenic T cells recruit endogenous MOG-specific B cells. J. Exp. Med. 2009;206:1303–1316. doi: 10.1084/jem.20090299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polman C.H., O‘Connor P.W., Havrdova E., Hutchinson M., Kappos L., Miller D.H., Phillips J.T., Lublin F.D., Giovanonni G., Wajgt A., Toal M., Lynn F., Panzara M.A., Sandrock A.W., AFFIRM Investigators A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N. Engl. J. Med. 2006;354:899–910. doi: 10.1056/NEJMoa044397. [DOI] [PubMed] [Google Scholar]
- Pomeroy I.M., Matthews P.M., Frank J.A., Jordan E.K., Esiri M.M. Demyelinated neocortical lesions in marmoset autoimmune encephalomyelitis mimic those in multiple sclerosis. Brain. 2005;128:2713–2721. doi: 10.1093/brain/awh626. [DOI] [PubMed] [Google Scholar]
- Pomeroy I.M., Jordan E.K., Frank J.A., Matthews P.M., Esiri M.M. Diffuse cortical atrophy in a marmoset model of multiple sclerosis. Neurosci. Lett. 2008;437:121–124. doi: 10.1016/j.neulet.2008.03.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prinz M., Schmidt H., Mildner A., Knobeloch K.P., Hanisch U.K., Raasch J., Merkler D., Detje C., Gutcher I., Mages J., Lang R., Martin R., Gold R., Becher B., Brück W., Kalinke U. Distinct and nonredundant in vivo functions of IFNAR on myeloid cells limit autoimmunity in the central nervous system. Immunity. 2008;28:675–686. doi: 10.1016/j.immuni.2008.03.011. [DOI] [PubMed] [Google Scholar]
- Qin J., Goswami R., Balabanov R., Dawson G. Oxidized phosphatidylcholine is marker for euroinflammation in multiple sclerosis brain. J. Neurosci. Res. 2007;85:977–984. doi: 10.1002/jnr.21206. [DOI] [PubMed] [Google Scholar]
- Quintana F.J., Basso A.S., Iglesias A.H., Korn T., Farez M.F., Bettelli E., Caccamo M., Oukka M., Weiner H.L. Control of Treg and TH17 cell differentiation by the aryl hydrocarbon receptor. Nature. 2008;453:65–71. doi: 10.1038/nature06880. [DOI] [PubMed] [Google Scholar]
- Quintana F.J., Farez M.F., Viglietta V., Iglesias A.H., Merbl Y., Izquierdo G., Lucas M., Basso A.S., Khoury S.J., Lucchinetti C.F., Cohen I.R., Weiner H.L. Antigen microarrays identify unique serum autoantibody signatures in clinical and pathologic subtypes of multiple sclerosis. Proc. Natl. Acad. Sci. U. S. A. 2008;105:18889–18894. doi: 10.1073/pnas.0806310105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Racke M.K., Lovett-Racke A.E., Karandikar N.J. The mechanism of action of glatiramer acetate treatment in multiple sclerosis. Neurology. 2010;74(Suppl. 1):S25–S30. doi: 10.1212/WNL.0b013e3181c97e39. [DOI] [PubMed] [Google Scholar]
- Reboldi A., Coisne C., Baumjohann D., Benvenuto F., Bottinelli D., Lira S., Uccelli A., Lanzavecchia A., Engelhardt B., Sallusto F. C-C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 2009;10:514–523. doi: 10.1038/ni.1716. [DOI] [PubMed] [Google Scholar]
- Rice G.P., Hartung H.P., Calabresi P. Anti-alpha4 integrin therapy for multiple sclerosis: mechanisms and rationale. Neurology. 2005;64:1336–1342. doi: 10.1212/01.WNL.0000158329.30470.D0. [DOI] [PubMed] [Google Scholar]
- Ridge S.C., Sloboda A.E., McReynolds R.A., Levine S., Oronsky A.L., Kerwar S.S. Suppression of experimental allergic encephalomyelitis by mitoxantrone. Clin. Immunol. Immunopathol. 1985;35:35–42. doi: 10.1016/0090-1229(85)90075-3. [DOI] [PubMed] [Google Scholar]
- Rieckmann P., Weber F., Günther A., Poser S. The phosphodiesterase inhibitor pentoxifylline reduces early side effects of interferon-beta 1b treatment in patients with multiple sclerosis. Neurology. 1996;47:604. doi: 10.1212/wnl.47.2.604. [DOI] [PubMed] [Google Scholar]
- Rieckmann P., Toyka K.V., Bassetti C., Beer K., Beer S., Buettner U., Chofflon M., Götschi-Fuchs M., Hess K., Kappos L., Kesselring J., Goebels N., Ludin H.P., Mattle H., Schluep M., Vaney C., Baumhackl U., Berger T., Deisenhammer F., Fazekas F., Freimüller M., Kollegger H., Kristoferitsch W., Lassmann H., Markut H., Strasser-Fuchs S., Vass K., Altenkirch H., Bamborschke S., Baum K., Benecke R., Brück W., Dommasch D., Elias W.G., Gass A., Gehlen W., Haas J., Haferkamp G., Hanefeld F., Hartung H.P., Heesen C., Heidenreich F., Heitmann R., Hemmer B., Hense T., Hohlfeld R., Janzen R.W., Japp G., Jung S., Jügelt E., Koehler J., Kölmel W., König N., Lowitzsch K., Manegold U., Melms A., Mertin J., Oschmann P., Petereit H.F., Pette M., Pöhlau D., Pohl D., Poser S., Sailer M., Schmidt S., Schock G., Schulz M., Schwarz S., Seidel D., Sommer N., Stangel M., Stark E., Steinbrecher A., Tumani H., Voltz R., Weber F., Weinrich W., Weissert R., Wiendl H., Wiethölter H., Wildemann U., Zettl U.K., Zipp F., Zschenderlein R., Izquierdo G., Kirjazovas A., Packauskas L., Miller D., Koncan Vracko B., Millers A., Orologas A., Panellus M., Sindic C.J., Bratic M., Svraka A., Vella N.R., Stelmasiak Z., Selmaj K., Bartosik-Psujik H., Mitosek-Szewczyk K., Belniak E., Mochecka A., Bayas A., Chan A., Flachenecker P., Gold R., Kallmann B., Leussink V., Mäurer M., Ruprecht K., Stoll G., Weilbach F.X., Multiple Sclerosis Therapy Consensus Group Escalating immunotherapy of multiple sclerosis—new aspects and practical application. J. Neurol. 2004;251:1329–1339. doi: 10.1007/s00415-004-0537-6. [DOI] [PubMed] [Google Scholar]
- Rivers T.M., Sprunt D.H., Berry G.P. Observations on attempts to produce acute disseminated encephalomyelitis in monkeys. J. Exp. Med. 1933;58:39–53. doi: 10.1084/jem.58.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson W.H., Fontoura P., Lee B.J., de Vegvar H.E., Tom J., Pedotti R., DiGennaro C.D., Mitchell D.J., Fong D., Ho P.P., Ruiz P.J., Maverakis E., Stevens D.B., Bernard C.C., Martin R., Kuchroo V.K., van Noort J.M., Genain C.P., Amor S., Olsson T., Utz P.J., Garren H., Steinman L. Protein microarrays guide tolerizing DNA vaccine treatment of autoimmune encephalomyelitis. Nat. Biotechnol. 2003;21:1033–1039. doi: 10.1038/nbt859. [DOI] [PubMed] [Google Scholar]
- Rose J.W., Burns J.B., Bjorklund J., Klein J., Watt H.E., Carlson N.G. Daclizumab phase II trial in relapsing and remitting multiple sclerosis: MRI and clinical results. Neurology. 2007;69:785–789. doi: 10.1212/01.wnl.0000267662.41734.1f. [DOI] [PubMed] [Google Scholar]
- Rose J.W., Foley J., Carlson N. Monoclonal antibody treatments for multiple sclerosis. Curr. Neurol. Neurosci. Rep. 2008;8:419–426. doi: 10.1007/s11910-008-0065-3. [DOI] [PubMed] [Google Scholar]
- Rosenthale M.E., Gluckman M.I. Immunopharmacologic activity of 1-aminocyclopentane-1-carboxylic acid. Experientia. 1968;24:1229–1230. doi: 10.1007/BF02146638. [DOI] [PubMed] [Google Scholar]
- Rubio J.P., Stankovich J., Field J., Tubridy N., Marriott M., Chapman C., Bahlo M., Perera D., Johnson L.J., Tait B.D., Varney M.D., Speed T.P., Taylor B.V., Foote S.J., Butzkueven H., Kilpatrick T.J. Replication of KIAA0350, IL2RA, RPL5 and CD58 as multiple sclerosis susceptibility genes in Australians. Genes Immun. 2008;9:624–630. doi: 10.1038/gene.2008.59. [DOI] [PubMed] [Google Scholar]
- Rudick R.A., Mi S., Sandrock A.W. LINGO-1 antagonists as therapy for multiple sclerosis: in vitro and in vivo evidence. Expert Opin. Biol. Ther. 2008;8:1561–1570. doi: 10.1517/14712598.8.10.1561. [DOI] [PubMed] [Google Scholar]
- Runström A., Leanderson T., Ohlsson L., Axelsson B. Inhibition of the development of chronic experimental autoimmune encephalomyelitis by laquinimod (ABR-215062) in IFN-beta k.o. and wild type mice. J. Neuroimmunol. 2006;173:69–78. doi: 10.1016/j.jneuroim.2005.11.023. [DOI] [PubMed] [Google Scholar]
- Ruuls S.R., de Labie M.C., Weber K.S., Botman C.A., Groenestein R.J., Dijkstra C.D., Olsson T., van der Meide P.H. The length of treatment determines whether IFN-beta prevents or aggravates experimental autoimmune encephalomyelitis in Lewis rats. J. Immunol. 1996;157:5721–5757. [PubMed] [Google Scholar]
- Sättler M.B., Merkler D., Maier K., Stadelmann C., Ehrenreich H., Bähr M., Diem R. Neuroprotective effects and intracellular signaling pathways of erythropoietin in a rat model of multiple sclerosis. Cell Death Differ. 2004;11(Suppl. 2):S181–S192. doi: 10.1038/sj.cdd.4401504. [DOI] [PubMed] [Google Scholar]
- Sarrias M.R., Farnós M., Mota R., Sánchez-Barbero F., Ibáñez A., Gimferrer I., Vera J., Fenutría R., Casals C., Yélamos J., Lozano F. CD6 binds to pathogen-associated molecular patterns and protects from LPS-induced septic shock. Proc. Natl. Acad. Sci. U. S. A. 2007;104:11724–11729. doi: 10.1073/pnas.0702815104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schilling S., Goelz S., Linker R., Lühder F., Gold R. Fumaric acid esters are effective in chronic experimental autoimmune encephalomyelitis and suppress macrophage infiltration. Clin. Exp. Immunol. 2006;145:101–107. doi: 10.1111/j.1365-2249.2006.03094.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt J., Gold R., Schönrock L., Zettl U.K., Hartung H.P., Toyka K.V. T-cell apoptosis in situ in experimental autoimmune encephalomyelitis following methylprednisolone pulse therapy. Brain. 2000;123:1431–1441. doi: 10.1093/brain/123.7.1431. [DOI] [PubMed] [Google Scholar]
- Schreiner B., Heppner F.L., Becher B. Modeling multiple sclerosis in laboratory animals. Semin. Immunopathol. 2009;31:479–495. doi: 10.1007/s00281-009-0181-4. [DOI] [PubMed] [Google Scholar]
- Scolding N. Stem cell therapy in patients with multiple sclerosis. Mult. Scler. 2006;12:677–678. doi: 10.1177/1352458506073176. [DOI] [PubMed] [Google Scholar]
- Seehusen F., Baumgärtner W. Axonal pathology and loss precede demyelination and accompany chronic lesions in a spontaneously occurring animal model of multiple sclerosis. Brain Pathol. 2010;20:551–559. doi: 10.1111/j.1750-3639.2009.00332.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segal B.M., Constantinescu C.S., Raychaudhuri A., Kim L., Fidelus-Gort R., Kasper L.H., Ustekinumab MS Investigators Repeated subcutaneous injections of IL12/23 p40 neutralising antibody, ustekinumab, in patients with relapsing–remitting multiple sclerosis: a phase II, double-blind, placebo-controlled, randomised, dose-ranging study. Lancet Neurol. 2008;7:796–804. doi: 10.1016/S1474-4422(08)70173-X. [DOI] [PubMed] [Google Scholar]
- Serrano-Fernández P., Ibrahim S.M., Zettl U.K., Thiesen H.J., Gödde R., Epplen J.T., Möller S. Intergenomic consensus in multifactorial inheritance loci: the case of multiple sclerosis. Genes Immun. 2004;5:615–620. doi: 10.1038/sj.gene.6364134. [DOI] [PubMed] [Google Scholar]
- Shamshiev A., Donda A., Carena I., Mori L., Kappos L., De Libero G. Self glycolipids as T-cell autoantigens. Eur. J. Immunol. 1999;29:1667–1675. doi: 10.1002/(SICI)1521-4141(199905)29:05<1667::AID-IMMU1667>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- Shimoda Y., Watanabe K. Contactins: emerging key roles in the development and function of the nervous system. Cell Adh. Migr. 2009;3:64–70. doi: 10.4161/cam.3.1.7764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siffrin V., Vogt J., Radbruch H., Nitsch R., Zipp F. Multiple sclerosis—candidate mechanisms underlying CNS atrophy. Trends Neurosci. 2010;33:202–210. doi: 10.1016/j.tins.2010.01.002. [DOI] [PubMed] [Google Scholar]
- Singh N.P., Hegde V.L., Hofseth L.J., Nagarkatti M., Nagarkatti P. Resveratrol (trans-3,5,4′-trihydroxystilbene) ameliorates experimental allergic encephalomyelitis, primarily via induction of apoptosis in T cells involving activation of aryl hydrocarbon receptor and estrogen receptor. Mol. Pharmacol. 2007;72:1508–1521. doi: 10.1124/mol.107.038984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sipe J.C., Romine J.S., Koziol J.A., McMillan R., Zyroff J., Beutler E. Development of cladribine treatment in multiple sclerosis. Mult. Scler. 1996;1:343–347. doi: 10.1177/135245859600100612. [DOI] [PubMed] [Google Scholar]
- Skripuletz T., Bussmann J.H., Gudi V., Koutsoudaki P.N., Pul R., Moharregh-Khiabani D., Lindner M., Stangel M. Cerebellar cortical demyelination in the murine cuprizone model. Brain Pathol. 2010;20:301–312. doi: 10.1111/j.1750-3639.2009.00271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith T., Groom A., Zhu B., Turski L. Autoimmune encephalomyelitis ameliorated by AMPA antagonists. Nat. Med. 2000;6:62–66. doi: 10.1038/71548. [DOI] [PubMed] [Google Scholar]
- Sobottka B., Harrer M.D., Ziegler U., Fischer K., Wiendl H., Hünig T., Becher B., Goebels N. Collateral bystander damage by myelin-directed CD8+ T cells causes axonal loss. Am. J. Pathol. 2009;175:1160–1166. doi: 10.2353/ajpath.2009.090340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sriram S., Steiner I. Experimental allergic encephalomyelitis: a misleading model of multiple sclerosis. Ann. Neurol. 2005;58:939–945. doi: 10.1002/ana.20743. [DOI] [PubMed] [Google Scholar]
- Stanislaus R., Pahan K., Singh A.K., Singh I. Amelioration of experimental allergic encephalomyelitis in Lewis rats by lovastatin. Neurosci. Lett. 1999;269:71–74. doi: 10.1016/s0304-3940(99)00414-0. [DOI] [PubMed] [Google Scholar]
- Steinman L. Some misconceptions about understanding autoimmunity through experiments with knockouts. J. Exp. Med. 1997;185:2039–2041. doi: 10.1084/jem.185.12.2039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinman L., Zamvil S.S. How to successfully apply animal studies in experimental allergic encephalomyelitis to research on multiple sclerosis. Ann. Neurol. 2006;60:12–21. doi: 10.1002/ana.20913. [DOI] [PubMed] [Google Scholar]
- Steinman L. A brief history of TH17, the first major revision in the TH1/TH2 hypothesis of T cell-mediated tissue damage. Nat. Med. 2007;13:139–145. doi: 10.1038/nm1551. [DOI] [PubMed] [Google Scholar]
- Steinman L. Mixed results with modulation of Th17 cells in human autoimmune disease. Nat. Immunol. 2010;11:41–44. doi: 10.1038/ni.1803. [DOI] [PubMed] [Google Scholar]
- Strauss U., Wissel K., Jung S., Wulff H., Hänsel W., Zhu J., Rolfs A., Mix E. K+ channel-blocking alkoxypsoralens inhibit the immune response of encephalitogenic T line cells and lymphocytes from Lewis rats challenged for experimental autoimmune encephalomyelitis. Immunopharmacology. 2000;48:51–63. doi: 10.1016/s0162-3109(00)00177-6. [DOI] [PubMed] [Google Scholar]
- Stuart G., Krikorian K.S. The neuro-paralytic accidents of anti-rabies treatment. Ann. Trop. Med. 1928;22:327–377. [Google Scholar]
- Stüve O., Bennett J.L. Pharmacological properties, toxicology and scientific rationale for the use of natalizumab (Tysabri) in inflammatory diseases. CNS Drug Rev. 2007;13:79–95. doi: 10.1111/j.1527-3458.2007.00003.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tajouri L., Fernandez F., Griffiths L.R. Gene expression studies in multiple sclerosis. Curr. Genomics. 2007;8:181–189. doi: 10.2174/138920207780833829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teitelbaum D., Meshorer A., Hirshfeld T., Arnon R., Sela M. Suppression of experimental allergic encephalomyelitis by a synthetic polypeptide. Eur. J. Immunol. 1971;1:242–248. doi: 10.1002/eji.1830010406. [DOI] [PubMed] [Google Scholar]
- Teuscher C., Doerge R.W., Fillmore P.D., Blankenhorn E.P. eae36, a locus on mouse chromosome 4, controls susceptibility to experimental allergic encephalomyelitis in older mice and mice immunized in the winter. Genetics. 2006;172:1147–1153. doi: 10.1534/genetics.105.049049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theiler M. Spontaneous encephalomyelitis of mice—a new virus disease. Science. 1934;80:122. doi: 10.1126/science.80.2066.122-a. [DOI] [PubMed] [Google Scholar]
- Tischner D., Weishaupt A., van den Brandt J., Müller N., Beyersdorf N., Ip C.W., Toyka K.V., Hünig T., Gold R., Kerkau T., Reichardt H.M. Polyclonal expansion of regulatory T cells interferes with effector cell migration in a model of multiple sclerosis. Brain. 2006;129:2635–2647. doi: 10.1093/brain/awl213. [DOI] [PubMed] [Google Scholar]
- Torkildsen O., Brunborg L.A., Myhr K.M., Bø L. The cuprizone model for demyelination. Acta Neurol. Scand. Suppl. 2008;188:72–76. doi: 10.1111/j.1600-0404.2008.01036.x. [DOI] [PubMed] [Google Scholar]
- Trebst C., Stangel M. Promotion of remyelination by immunoglobulins: implications for the treatment of multiple sclerosis. Curr. Pharm. Des. 2006;12:241–249. doi: 10.2174/138161206775193118. [DOI] [PubMed] [Google Scholar]
- Tuohy V.K., Lu Z.J., Sobel R.A., Laursen R.A., Lees M.B. A synthetic peptide from myelin proteolipid protein induces experimental allergic encephalomyelitis. J. Immunol. 1988;141:1126–1130. [PubMed] [Google Scholar]
- Ure D.R., Rodriguez M. Histopathology in the Theiler's virus model of demyelination. In: Lavi E., Constantinescu C.S., editors. Experimental Models of Multiple Sclerosis. Springer; New York: 2005. pp. 579–592. [Google Scholar]
- van der Meide P.H., de Labie M.C., Ruuls S.R., Groenestein R.J., Botman C.A., Olsson T., Dijkstra C.D. Discontinuation of treatment with IFN-beta leads to exacerbation of experimental autoimmune encephalomyelitis in Lewis rats. Rapid reversal of the antiproliferative activity of IFN-beta and excessive expansion of autoreactive T cells as disease promoting mechanisms. J. Neuroimmunol. 1998;84:14–23. doi: 10.1016/s0165-5728(97)00207-5. [DOI] [PubMed] [Google Scholar]
- van Oosten B.W., Barkhof F., Truyen L., Boringa J.B., Bertelsmann F.W., von Blomberg B.M., Woody J.N., Hartung H.P., Polman C.H. Increased MRI activity and immune activation in two multiple sclerosis patients treated with the monoclonal anti-tumor necrosis factor antibody cA2. Neurology. 1996;47:1531–1534. doi: 10.1212/wnl.47.6.1531. [DOI] [PubMed] [Google Scholar]
- Vollmar P., Nessler S., Kalluri S.R., Hartung H.P., Hemmer B. The antidepressant venlafaxine ameliorates murine experimental autoimmune encephalomyelitis by suppression of pro-inflammatory cytokines. Int. J. Neuropsychopharmacol. 2008;16:1–12. doi: 10.1017/S1461145708009425. [DOI] [PubMed] [Google Scholar]
- Vollmer T., Stewart T., Baxter N. Mitoxantrone and cytotoxic drugs mechanisms of action. Neurology. 2010;74(Suppl. 1):S41–S46. doi: 10.1212/WNL.0b013e3181c97f5a. [DOI] [PubMed] [Google Scholar]
- Waiczies S., Bendix I., Zipp F. Geranylgeranylation but not GTP-loading of Rho GTPases determines T cell function. Sci. Signal. 2008;1:pt3. doi: 10.1126/stke.112pt3. (1–3) [DOI] [PubMed] [Google Scholar]
- Wang J., Wang G., Sun B., Li H., Mu L., Wang Q., Li G., Shi L., Jin L., Kostulas N. Interleukin-27 suppresses experimental autoimmune encephalomyelitis during bone marrow stromal cell treatment. J. Autoimmun. 2008;30:222–229. doi: 10.1016/j.jaut.2007.10.001. [DOI] [PubMed] [Google Scholar]
- Weber F., Polak T., Günther A., Kubuschok B., Janovskaja J., Bitsch A., Poser S., Rieckmann P. Synergistic immunomodulatory effects of ‘1b and the phosphodiesterase inhibitor pentoxifylline in patients with relapsing–remitting multiple sclerosis. Ann. Neurol. 1998;44:27–34. doi: 10.1002/ana.410440109. [DOI] [PubMed] [Google Scholar]
- Wegner C., Stadelmann C., Brück W. Therapeutic treatment with Laquinimod reduces inflammation, demyelination and axonal damage in experimental autoimmune encephalomyelitis. Acta Neuropathol. 2009;118:464. (abstract) [Google Scholar]
- Weiner H.L. The challenge of multiple sclerosis: how do we cure a chronic heterogeneous disease? Ann. Neurol. 2009;65:239–248. doi: 10.1002/ana.21640. [DOI] [PubMed] [Google Scholar]
- Weishaupt A., Kuhlmann T., Schönrock L.M., Toyka K.V., Brück W., Gold R. Effects of intravenous immunoglobulins on T cell and oligodendrocyte apoptosis in high-dose antigen therapy in experimental autoimmune encephalomyelitis. Acta Neuropathol. 2002;104:385–390. doi: 10.1007/s00401-002-0568-y. [DOI] [PubMed] [Google Scholar]
- Wekerle H., Kojima K., Lannes-Vieira J., Lassmann H., Linington C. Animal models. Ann. Neurol. 1994;36(Suppl.):S47–S53. doi: 10.1002/ana.410360714. [DOI] [PubMed] [Google Scholar]
- Wenning W., Haghikia A., Laubenberger J., Clifford D.B., Behrens P.F., Chan A., Gold R. Treatment of progressive multifocal leukoencephalopathy associated with natalizumab. N. Engl. J. Med. 2009;361:1075–1080. doi: 10.1056/NEJMoa0810257. [DOI] [PubMed] [Google Scholar]
- Wiendl H., Hohlfeld R. Therapeutic approaches in multiple sclerosis: lessons from failed and interrupted treatment trials. BioDrugs. 2002;16:183–200. doi: 10.2165/00063030-200216030-00003. [DOI] [PubMed] [Google Scholar]
- Wolf A., Kabat E.A., Bezer A.E. The pathology of acute disseminated encephalomyelitis produced experimentally in the rhesus monkey and its resemblance to human demyelinating disease. J. Neuropath. Exp. Neurol. 1947;6:333–357. doi: 10.1097/00005072-194710000-00003. [DOI] [PubMed] [Google Scholar]
- Wright B.R., Waarington A.E., Rodriguez M. Cellular mechanisms of central nervous system repair by natural autoreactive antibodies. Arch. Neurol. 2009;66:1456–1459. doi: 10.1001/archneurol.2009.262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wulff H., Castle N.A., Pardo L.A. Voltage-gated potassium channels as therapeutic targets. Nat. Rev. Drug Discov. 2009;8:982–1001. doi: 10.1038/nrd2983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wynn D., Kaufman M., Montalban X., Vollmer T., Simon J., Elkins J., O‘Neill G., Neyer L., Sheridan J., Wang C., Fong A., Rose J.W., CHOICE Investigators Daclizumab in active relapsing multiple sclerosis (CHOICE study): a phase 2, randomised, double-blin, placebo-controlled, add-on trial with interferon beta. Lancet Neurol. 2010;9:381–390. doi: 10.1016/S1474-4422(10)70033-8. [DOI] [PubMed] [Google Scholar]
- Yang J.S., Xu L.Y., Xiao B.G., Hedlund G., Link H. Laquinimod (ABR-215062) suppresses the development of experimental autoimmune encephalomyelitis, modulates the Th1/Th2 balance and induces the Th3 cytokine TGF-beta in Lewis rats. J. Neuroimmunol. 2004;156:3–9. doi: 10.1016/j.jneuroim.2004.02.016. [DOI] [PubMed] [Google Scholar]
- Yang Y., Liu Y., Wei P., Peng H., Winger R., Hussain R.Z., Ben L.H., Cravens P.D., Gocke A.R., Puttaparthi K., Racke M.K., McTigue D.M., Lovett-Racke A.E. Silencing Nogo-A promotes functional recovery in demyelinating disease. Ann. Neurol. 2010;67:498–507. doi: 10.1002/ana.21935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yednock T.A., Cannon C., Fritz L.C., Sanchez-Madrid F., Steinman L., Karin N. Prevention of experimental autoimmune encephalomyelitis by antibodies against α4β1 integrin. Nature. 1992;356:63–66. doi: 10.1038/356063a0. [DOI] [PubMed] [Google Scholar]
- Yi H., Zhen Y., Jiang L., Zheng J., Zhao Y. The phenotypic characterization of naturally occurring regulatory CD4+CD25+ T cells. Cell. Mol. Immunol. 2006;3:189–195. [PubMed] [Google Scholar]
- Yousry T.A., Major E.O., Ryschkewitsch C., Fahle G., Fischer S., Hou J., Curfman B., Miszkiel K., Mueller-Lenke N., Sanchez E., Barkhof F., Radue E.W., Jäger H.R., Clifford D.B. Evaluation of patients treated with natalizumab for progressive multifocal leukoencephalopathy. N. Engl. J. Med. 2006;354:924–933. doi: 10.1056/NEJMoa054693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youssef S., Stüve O., Patarroyo J.C., Ruiz P.J., Radosevich J.L., Hur E.M., Bravo M., Mitchell D.J., Sobel R.A., Steinman L., Zamvil S.S. The HMG-CoA reductase inhibitor, atorvastatin, promotes a Th2 bias and reverses paralysis in central nervous system autoimmune disease. Nature. 2002;420:78–84. doi: 10.1038/nature01158. [DOI] [PubMed] [Google Scholar]
- Yuan R., Maeda Y., Li W., Lu W., Cook S., Dowling P. Erythropoietin: a potent inducer of peripheral immuno/inflammatory modulation in autoimmune EAE. PLoS One. 2008;3:e1924. doi: 10.1371/journal.pone.0001924. (1–18) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yudkin P.L., Ellison G.W., Ghezzi A., Goodkin D.E., Hughes R.A., McPherson K., Mertin J., Milanese C. Overview of azathioprine treatment in multiple sclerosis. Lancet. 1991;338:1051–1055. doi: 10.1016/0140-6736(91)91909-e. [DOI] [PubMed] [Google Scholar]
- Zettl U.K., Mix E., Zielasek J., Stangel M., Hartung H.P., Gold R. Apoptosis of myelin-reactive T cells induced by reactive oxygen and nitrogen intermediates in vitro. Cell. Immunol. 1997;178:1–8. doi: 10.1006/cimm.1997.1113. [DOI] [PubMed] [Google Scholar]
- Zeyda M., Poglitsch M., Geyeregger R., Smolen J.S., Zlabinger G.J., Hörl W.H., Waldhäusl W., Stulnig T.M., Säemann M.D. Disruption of the interaction of T cells with antigen-presenting cells by the active leflunomide metabolite teriflunomide: involvement of impaired integrin activation and immunologic synapse formation. Arthritis Rheum. 2005;52:2730–2739. doi: 10.1002/art.21255. [DOI] [PubMed] [Google Scholar]
- Zhu J., Mix E., Winblad B. The antidepressant and antiinflammatory effects of rolipram in the central nervous system. CNS Drug Rev. 2001;7:387–398. doi: 10.1111/j.1527-3458.2001.tb00206.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziemssen T., Ziemssen F. The role of the humoral immune system in multiple sclerosis (MS) and its animal model experimental autoimmune encephalomyelitis (EAE) Autoimmun. Rev. 2005;4:460–467. doi: 10.1016/j.autrev.2005.03.005. [DOI] [PubMed] [Google Scholar]
- Zorzella S.F., Seger J., Martins D.R., Pelizon A.C., Sartori A. Resistance to experimental autoimmune encephalomyelitis development in Lewis rats from a conventional animal facility. Mem. Inst. Oswaldo Cruz. 2007;102:931–936. doi: 10.1590/s0074-02762007000800007. [DOI] [PubMed] [Google Scholar]
- Zozulya A.L., Wiendl H. The role of CD8 suppressors versus destructors in autoimmune central nervous system inflammation. Hum. Immunol. 2008;69:780–797. doi: 10.1016/j.humimm.2008.07.014. [DOI] [PubMed] [Google Scholar]