Abstract
Porcine reproductive and respiratory syndrome (PRRS) is a chronic viral disease of pigs, has been posing a huge economic concern to pig industry worldwide. In this study, we developed biodegradable PLGA [poly(d,l-lactide-co-glycolide)] nanoparticle-entrapped killed PRRSV vaccine (Nano-KAg), and administered intranasally to pigs once and evaluated the immune correlates. In Nano-KAg vaccinated homologous virus challenged pigs, complete clearance of viremia was observed in 2 weeks, associated with a significant increase in virus neutralizing titers only in the lungs, compared to both unvaccinated and killed vaccine vaccinated pigs. The lung homogenate and sera of Nano-KAg vaccinated pigs had higher levels of IFN-γ and lower levels of TGF-β than control groups. Restimulation of mononuclear cells isolated from the lungs, blood, BAL, and TBLN of Nano-KAg vaccinated pigs’ secreted significantly increased levels of Th1 cytokines, IFN-γ and IL-12. In addition, higher frequencies of CD3+CD8+, CD4+CD8+, and γδ T cells, and reduced frequency of Foxp3+ T-regulatory cells were observed in Nano-KAg vaccinated pigs. Thus, intranasal delivery of Nano-KAg vaccine may be a suitable strategy to elicit anti-PRRSV immune response required to better clear viremia in pigs.
Keywords: PRRS virus, Killed PRRSV vaccine, PLGA nanoparticles, Mucosal immune response, Protection
1. Introduction
Porcine reproductive and respiratory syndrome (PRRS) was first reported in the US in 1987 (Keffaber, 1989). PRRS is endemic in most pork-producing countries and it is responsible for major economic losses to the swine industry, with an estimated annual loss of $664 million to the US economy (Holtkamp and Kliebenstein, 2011). Clinical signs of PRRS in pigs include fever, anorexia, and respiratory disease; and poor reproductive performance in pregnant pigs is characterized by abortions, mummified fetus, and weak-born piglets (Done et al., 1996). The causal agent is PRRS virus (PRRSV) and it is an enveloped, positive sense, single-strand RNA virus, belongs to the family Arteriviridae (King et al., 2011).
PRRSV modulates the pig immune system as early as two-days, and continues several weeks post-infection (Dwivedi et al., 2012, Mateu and Diaz, 2008). In PRRSV infected pigs production of an important Th1 cytokine IFN-γ is delayed and in reduced amounts. The immune modulation caused by PRRSV is attributed to virus-mediated suppression of host IFN-α production (Albina et al., 1998). In addition, the virus helps in induction of secretion of interleukin (IL)-10 and transforming growth factor-β (TGF-β), and upregulation of Foxp3+ T-regulatory cells (Tregs) (Silva-Campa et al., 2012). Virus neutralizing (VN) antibody is important for viral clearance, but the role of VN antibodies in clearing the PRRSV in infected pigs is unclear (Diaz et al., 2005). However, passive transfer of VN antibodies has shown to provide protection against PRRSV challenge (Osorio et al., 2002). To control PRRS outbreaks modified live PRRSV vaccine (PRRS-MLV) is widely used and it confers protection against homologous virus, but incomplete protection against reinfections and heterologous viruses (Mengeling et al., 2003). There are limitations of PRRS-MLV, as there are reports of appearance of revertant vaccine virus in certain pigs (Li et al., 2009, Mengeling et al., 2002, Nielsen et al., 2001). Available killed vaccines are safe, but their efficacy is poor (Bassaganya-Riera et al., 2004, Charerntantanakul, 2009, Piras et al., 2005).
Stimulating the immune system systemically results mainly in systemic protection with low mucosal immune responses. In contrast, optimum stimulation of the mucosal immune system provides both mucosal and systemic immunity, resulting in inhibition of entry of pathogens into the body (Holmgren et al., 1992). Nanotechnology based vaccine delivery system is an innovation of the 21st century (Panyam and Labhasetwar, 2003). Nanoparticles offer the advantage of increasing the efficiency of drug and vaccine delivery, and also possess adjuvant properties (Gupta et al., 1998). Poly(d,l-lactide-co-glycolide) (PLGA) nanoparticle-mediated delivery of vaccines has shown to be effective in eliciting protective immune response, administered by either mucosal and systemic routes (Eldridge et al., 1989, Spiers et al., 2000). PLGA is biocompatible, biodegradable, and protects the entrapped vaccine from proteases mediated degradation at mucosal surfaces; and it is a FDA approved agent (Lu et al., 2009). PLGA helps in sustained release of entrapped vaccine, and thus limits the need of booster doses (Thomasin et al., 1996). Therefore, the goal of this study was to develop PLGA nanoparticle-encapsulated PRRSV killed vaccine and investigate its efficacy in pigs vaccinated by intranasal route. Our results suggest that our strategy has the potential to generate anti-PRRSV immune response and in better clearance of viremia.
2. Materials and methods
2.1. Cells, PRRSV, and polymer
A stable Mycoplasma-free MARC-145 cells (African Green monkey kidney cell line) which supports the growth of PRRSV (Kim et al., 2007) were used to prepare PRRSV stocks, killed viral antigens, and in immunological assays. Cells were maintained in Dulbecco's minimum essential medium (DMEM, Lonza) supplemented with 10% fetal bovine serum (Atlanta Biologicals), 37 °C with 5% CO2. For virus infection DMEM supplemented with 2% horse serum was used. For preparation of nanoparticles, PLGA [poly(d,l-lactide-co-glycolide)] 50:50 (MW 40,000–75,000), polyvinyl alcohol (MW 30,000–70,000) (Sigma–Aldrich), dicholoro methane (Acros Organics), and BCA (bicinchoninic acid) protein assay kit (Pierce) were used.
2.2. Animals
Conventional Large White–Duroc crossbred weaned specific-pathogen-free pigs at 3–4 weeks of age were received and housed in our BSL-2 facility. The swine herd (source of piglets) was confirmed seronegative for PRRSV, porcine respiratory coronavirus, transmissible gastroenteritis virus, and porcine circovirus 2. Blood samples collected from piglets were confirmed negative for PRRSV antibodies. Pigs were allowed to acclimate for 1 week before initiation of the experiments. Throughout the duration of the study animals received food and water ad libitum.
2.3. Preparation of PRRSV antigens
MARC-145 cell-monolayer (>90% confluent) was infected with the North American prototype PRRSV strain VR2332 (Kim et al., 2007) (provided by Dr. Eric Nelson, South Dakota State University) which was attenuated (passaged several times (∼50) in vitro) and was used at 0.001 multiplicity of infection in roller bottles or T150 tissue culture flasks, and freeze-thawed three times when cells were showing >80% cytopathic effect. Harvested infected cell culture fluid was clarified to remove cell debris at 2000 × g for 15 min and subjected to ultracentrifugation with a 20% sucrose overlay at 100,000 × g for 2 h at 4 °C. Pooled semi-purified PRRSV antigen was suspended in sterile PBS and titrated to determine the viral titer. PRRSV was inactivated using 254 nm UV-irradiation [EL series UV lamps, UVP, LLC (CA); 8 W/115 V–60 Hz/0.32 A] for 1 h and the inactivation was confirmed by cell culture indirect immunofluorescence assay in MARC-145 cells. Semipurified virus was sonicated (probe sonicator at 80% amplitude, 30 s for 3 cycles), the protein content was estimated using BCA assay kit (Pierce), and aliquots were stored at −70 °C. Control antigen was prepared in the same manner using uninfected MARC-145 cells.
2.4. Preparation of PLGA nanoparticles
Nanoparticles were prepared by a standard double emulsion solvent evaporation technique (Cao and Schoichet, 1999). Briefly, 15% of PLGA 50/50 (750 mg) was dissolved in 5 ml of dichloromethane, and 100 μl of killed VR2332 proteins (5 mg) was added. The mixture was homogenized for 90 s using a Brinkman Polytron homogenizer at 6000 rpm. The homogenized mixture was added to 60 ml of aqueous solution of polyvinyl alcohol (10% PVA), and homogenized for 5 min. Finally, the preparation was stirred overnight at room temperature (RT) to allow solvent evaporation. The nanoparticles were washed in distilled water three times by centrifugation at 10,000 rpm for 30 min. After the final wash the nanoparticles were freeze-dried and stored at 4 °C until used for immunization.
2.5. Determination of size and morphology of nanoparticles
Size and morphology of nanoparticles was determined by scanning electron microscopy (Hitachi S-3500 N). Briefly, freeze-dried nanoparticles were mounted on an adhesive stub, coated with gold/palladium under vacuum using an ion coater. The coated specimen was examined under the microscope at 10 kV.
2.6. Determination of entrapment efficiency of PRRSV proteins in nanoparticles
The amount of entrapped PRRSV antigens in the nanoparticles was determined as described previously (Rajapaksa et al., 2010). Briefly, freeze-dried nanoparticles (10 mg) were dissolved in 1 ml of 0.1 N sodium hydroxide and incubated for 1 h at 37 °C with constant stirring. The mixture was vortexed and centrifuged at 10,000 rpm for 5 min. The supernatant was collected and analyzed for the protein content with the help of series of BSA standards prepared in 0.1 N NaOH and using BCA protein assay kit (Pierce, USA).
2.7. Pigs and inoculations
Pigs (n = 12) were divided into four groups (n = 3 pigs per group). Group I: mock pigs; Group II: inoculated with normal saline; Group III: inoculated with killed VR2332 antigens (K-Ag); Group IV: inoculated with nanoparticle-entrapped killed VR2332 antigens (Nano-KAg). The amount of PRRSV antigens in each dose of Nano-KAg and KAg was one mg, which was equivalent to approximately 5 × 106 TCID50 of inactivated virus. All inoculations including the challenge virus (1 × 106 TCID50 in 2 ml per pig) were administered once, intranasally. Groups II, III, and IV were challenged on day post-immunization (DPI) 21 and euthanized on day post-challenge (DPC or PC) 15. Mock-inoculated pigs were euthanized separately prior to killing of any infected animals. All the pigs were housed and maintained in our large animal BSL-2 facility. Clinical samples from pigs were collected and animals euthanized as per the protocols approved by the Institutional Animal Care and use Committee, and biological agents were handled as per the Institutional Biosafety Committee, The Ohio State University, Ohio.
2.8. Collection of blood and lung samples for analysis
For evaluation of viremia and for titration of PRRSV specific VN antibodies, 3–5 ml of blood samples were collected at DPI 0 and 21, and DPC 7 and 15. Serum was separated from the clotted blood and aliquots were stored at −20 °C. We and others have found suitability of lung homogenates (lysates) instead of BAL fluid for the cytokine and virus evaluation in the pig lungs (Barbe et al., 2011, Renukaradhya et al., 2010). Briefly, 2–5 g of lung tissue samples collected in DMEM were minced and blended for 5 min in a Stomacher-400 laboratory blender (Seward). Clarified supernatant (1 g/ml) was aliquoted and stored at −20 °C.
2.9. Isolation of PBMC, lung MNC, BAL, TBLN cells
For isolation of PBMC, blood was collected in acid citrate dextrose solution from euthanized pigs. Lung mononuclear cells (lung MNC/LMNC) from individual pigs were isolated as described (Dwivedi et al., 2011a). The airways were lavaged to collect BAL fluid using sterile ice cold PBS containing EDTA (0.03%); and (Dwivedi et al., 2011a) samples of tracheobronchial lymph nodes (TBLN) were collected in DMEM, and MNCs were isolated as described (Dwivedi et al., 2011a).
2.10. Virus titration and virus neutralizing test (VNT)
PRRSV titer in serum and VN antibody titer in serum and lung homogenate were analyzed by indirect immunofluorescence assay (IFA) (Christopher-Hennings et al., 2001). Briefly, for virus titration confluent monolayer of MARC-145 cells in 96-well microtiter plate was treated with 10-fold dilution of serum for 48 h. For VNT, serum and lung homogenate were heat inactivated (56 °C for 30 min), twofold diluted, and incubated with equal volume of PRRSV (VR2332) 250 TCID50 per well for 2 h at 37 °C. The suspension was transferred into microtiter plates containing confluent monolayer of MARC-145 cells and incubated for 48 h at 37 °C. Followed by treatment with anti-PPRSV nucleocapsid protein specific mAb (SDOW-17), Alexa-488 conjugated anti-mouse IgG (H + L) secondary antibody, and mounted with glycerol–PBS (6:4 ratio); the virus induced cytopathic effect was examined under a fluorescent microscope. For VNT, the reciprocal of the dilution in which greater than 95% reduction in fluorescence foci compared to virus control (250 TCID50) was considered as the titer.
2.11. PRRSV isotype specific antibody analysis in the lungs and blood
The presence of PRRSV specific IgA antibodies in the lung homogenate was analyzed by ELISA as described (Dwivedi et al., 2011b). Briefly, ELISA plates were coated with pre-titrated semipurified PRRSV (VR2332) antigens (10 μg/ml) or recombinant PRRSV nucleocapsid (N′) protein (5 μg/ml) (Baker et al., 2007, Johnson et al., 2007) (kind gift from Dr. Murtaugh, University of Minnesota) in carbonate–bicarbonate buffer (pH 9.6) overnight at 4 °C, and plates were washed and treated with blocking buffer (1% BSA and 0.1% Tween 20 in PBS) for 2 h at RT. Serum and lung homogenate samples (1:100 diluted) were added and incubated for 2 h at RT. The bound virus specific isotype antibody was detected using affinity purified (to ensure class specificity) Goat anti-pig IgA secondary antibody conjugated with HRP (Cat# A100-102P, Bethyl Laboratories Inc.). Finally, plates were developed using chromogen TMB (3,3′, 5,5″-tetramethylbenzidine) (KPL) and the reaction was stopped using 1 M phosphoric acid, and the plate was read at 450 nm. To eliminate the background activity, we also included non-PRRSV specific antigen coated control plate, blocked and treated exactly as above with test samples side-by-side. The OD values obtained from control plates were subtracted from the experimental plate to obtain corrected absorbance values. For lung homogenate the mean OD value of 10 PRRSV negative pig lung homogenate samples plus 2 Standard Deviation was considered as positive–negative cut-off, and it was 0.20.
2.12. PRRSV specific recall/memory immune response
Five million pig PBMC, TBLN MNC, and lung MNC were restimulated ex vivo in a 24-well tissue culture plate in the absence or presence of killed semipurified VR2332 Ags (50 μg/ml) in enriched RPMI for 48 h at 37 °C. The harvested culture supernatant was analyzed for cytokines by ELISA. Amounts of cytokines secreted by the cells cultured in the absence of any Ags were subtracted from the respective restimulated experimental well values.
2.13. Analysis of cytokine response
Serum samples, culture supernatants, and lung homogenates were analyzed for Th1 (IFN-γ and IL-12), Th2 (IL-4), pro-inflammatory (IL-6), and immunosuppressive (IL-10 and TGF-β) cytokines by ELISA (Renukaradhya et al., 2010). Briefly, ELISA plates were coated with cytokine specific capture antibodies in carbonate–bicarbonate buffer (pH 9.6), and blocked with blocking buffer. Test samples and cytokine standards were added and incubated overnight at 4 °C. After washing, respective cytokine specific detection antibodies were added and incubated for 2 h at RT. Plates were washed and treated with HRP-conjugated streptavidin (Biosource) and incubated for 1 h at RT. After washing, plates were developed by adding TMB (KPL) and the reaction was stopped using 1 M phosphoric acid. The plates were read in a microtiter plate reader at OD450 nm within 30 min. Cytokine levels were determined by their comparison to standard curves generated from OD values of known concentrations of swine recombinant cytokines. Amount of cytokines present in the lung homogenate was converted to pg per gm of lung tissue.
2.14. Flow cytometry study of different immune cell populations
Flow cytometry analysis was performed to determine the phenotype and the frequency of different immune cells by a multicolor immunoassay as described (Dwivedi et al., 2011a, Manickam et al., 2013). Briefly, single cell suspensions (PBMC, LMNC, BAL, TBLN and ILN) were resuspended in fluorescence-activated cell sorting (FACS) buffer and plated (1 × 106) in U-bottom 96-well plates and treated with 2% pig serum to block the Fc receptors. Cells were treated with fluorochrome tagged, purified, or biotin labeled pig specific mAbs, CD3ɛ, CD172 (Southern Biotech), CD4α, CD8α/β, CD11c (BD Biosciences), CD25, TcR1N4 (VMRD), SLAII (Serotech), Foxp3 (eBioscience) or their respective isotype control mAb. Cells were washed and treated with streptavidin-conjugated fluorochrome or respective anti-species isotype specific secondary antibody conjugated with fluorochrome. Finally, cells were fixed and washed in FACS buffer. For intracellular Foxp3 staining, the cells were stained first for cell surface CD4 and CD25, and then treated with permeabilization buffer (PBS containing 0.1% Saponin and 3% Formaldehyde) and stained with fluorochrome-conjugated pig Foxp3 cross-reactive anti-rat Foxp3 mAb. Immunostained cells were acquired using a FACS Aria II (BD Biosciences) flow cytometer. The analysis was done to determine different immune cell populations based on the phenotypes: T-helper cells (CD3+CD4+CD8−); CD8+ T cells (CD3+CD4−CD8+); T-helper/memory cells (CD3+CD4+CD8+); γδ T cells (CD8α+TcR1N4+); T-regulatory cells (CD4+CD25+Foxp3+); CD172+ (myeloid cells); and CD172+CD11 C+SLAII+ (dendritic cells rich fraction), analyzed using FlowJo software (Tree Star Inc., OR, USA). Frequencies of individual immune cells were analyzed from a total 50,000 events.
2.15. Statistical analysis
All data were expressed as the mean ± SEM of three pigs. Statistical analyses were performed using one way analysis of variance (ANOVA) followed by Tukey's HSD post-test using GraphPad InStat (software version 5.0 for windows) to establish differences between different experimental groups. Statistical significance was assessed as P < 0.05. Alphabets ‘a’, ‘b’ and ‘c’ in the figures represent statistically significant difference between: ‘a’ – unvaccinated vs K-Ag; ‘b’ – unvaccinated vs Nano-KAg; ‘c’ – K-Ag vs Nano-KAg pigs.
3. Results
3.1. Characterization of PRRSV antigens entrapped nanoparticles
Nanoparticles used in our study were characterized for their size, morphology, yield, and protein encapsulation efficiency. The size of both sham as well as PRRSV antigens entrapped nanoparticles was in the range of 200–600 nm (Fig. 1a). The yield of prepared nanoparticles was calculated considering the amount of viral antigens used and it was 82.32 ± 3.3%. The surface morphology of the Nano-Kg was spherical with no surface discontinuity as per SEM pictures (Fig. 1a). The entrapment efficiency of PRRSV antigens in Nano-Kg was 50–55%, calculated based on the amount of total PRRSV protein used in the entrapment.
Fig. 1.
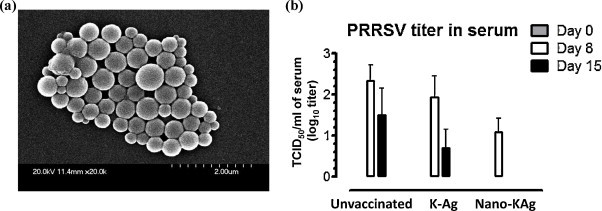
(a) Morphology of PRRSV Ags entrapped PLGA nanoparticles. Scanning electronic photomicrograph of PLGA nanoparticles prepared by a standard multiple emulsion method. The size of the nanoparticles appears to be variable ranging 200–600 nm. (b) Mucosal vaccination of pigs with PRRSV Ags entrapped in nanoparticles (Nano-KAg) cleared the viremia by PC 15. Pigs were unvaccinated (n = 3) or vaccinated with either killed PRRSV antigens (K-Ag) (n = 3) or Nano-KAg (n = 3) once, intranasally, and on DPI 21 challenged with PRRSV VR2332 strain. Serum samples collected on indicated post-challenge day (PC) were analyzed to measure PRRSV titers. Each bar represents the geometric mean of three pigs ± SEM.
3.2. Viral load in serum
Immunofluorescence assay was performed to determine the PRRSV titer (TCID50/ml) in serum samples collected at PC 0, 7, and 15. In Nano-KAg vaccinated pigs reduced viremia by greater than 1 log at PC 8 and complete viral clearance by PC 15 was observed (Fig. 1b). In the K-Ag vaccinated pigs there was a marginal reduction in viremia compared to unvaccinated virus challenged pigs (Fig. 1b).
3.3. Humoral immune response in serum and the lungs
Nano-KAg vaccinated pigs had increased levels of VN titers in serum with significantly higher levels only at PC 7 compared to K-Ag vaccinated or unvaccinated pigs (Fig. 2a), but the VN titer in serum was not increased (rather decreased) at PC 15 (Fig. 2a). However, in the lung homogenate of Nano-KAg received pigs significantly increased VN titers compared to both unvaccinated and K-Ag groups was observed (Fig. 2b). Interestingly, both the K-Ag and Nano-KAg vaccinated pigs showed significantly increased PRRSV specific IgA levels in the lungs compared to unvaccinated virus challenged animals (Fig. 2c). Since the PRRSV Ags used in vaccine preparation and in ELISA was a semipurified PRRSV, it contains high levels of MARC-145 cell components. Therefore, we wanted to analyze the PRRSV specific response in the lung homogenate of all the pigs used in this study, using the recombinant PRRSV N′ protein in IgA ELISA. Our results suggested that virus specific response was elicited in both K-Ag and Nano-KAg vaccinated pigs (Fig. 2d).
Fig. 2.
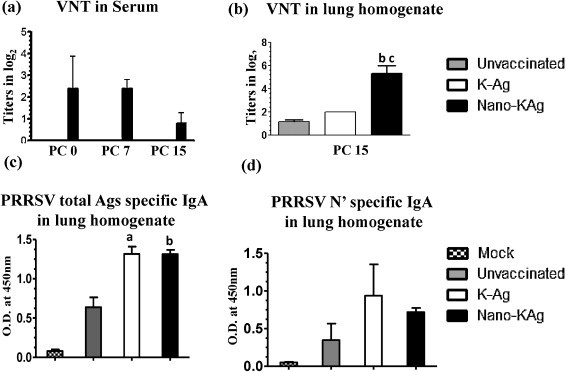
Enhanced IgA and neutralizing antibody titers in serum and lungs of Nano-KAg vaccinated virus challenged pigs. Analysis was performed to determine PRRSV neutralizing antibody response in: (a) serum; (b) lung homogenate by immunofluorescence assay; (c) PRRSV IgA response in the lung homogenate against PRRSV total Ags; (d) PRRSV IgA response in the lung homogenate against PRRSV N′ protein by ELISA. Each bar in the graph represents geometric mean VN titer (log2 values) or optical density value from three pigs ± SEM. Alphabet ‘a’, ‘b’ and ‘c’ represents the statistical significant difference (p < 0.05) between unvaccinated vs K-Ag, unvaccinated vs Nano-KAg, and K-Ag vs Nano-KAg pigs, respectively.
3.4. Secretion of increased Th1, reduced Th2 and immunosuppressive cytokines in serum and lungs of Nano-KAg vaccinated pigs
The Th1 cytokine, IFN-γ, in serum of unvaccinated pigs was undetectable, and it was secreted at lower levels in K-Ag received virus challenged pigs. But, in Nano-KAg vaccinated pigs, a significantly increased IFN-γ response was detected at PC 7 and 15 (Fig. 3a). Similarly, Nano-KAg immunized pig lung homogenate had significantly higher levels of IFN-γ compared to both the control group pigs at PC 15 (Fig. 3c).
Fig. 3.
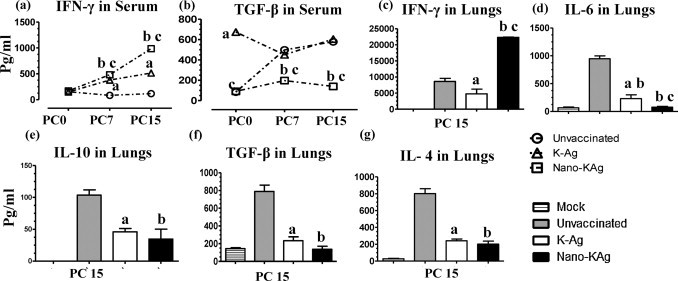
Cytokine response in serum and lungs of pigs intranasally vaccinated with Nano-KAg vaccine. Serum samples collected at indicated PCs were analyzed for cytokines, (a) IFN-γ and (b) TGF-β by ELISA. Lung homogenates prepared on the day of necropsy (PC 15) were analyzed for: (c) IFN-γ; (d) IL-6; (e) IL-10; (f) TGF-β; and (g) IL-4 by ELISA. Each bar or data point in the graph represents geometric mean cytokine amount from three pigs ± SEM. Alphabet ‘a’, ‘b’ and ‘c’ represents the statistical significant difference (p < 0.05) between unvaccinated vs K-Ag, unvaccinated vs Nano-KAg, and K-Ag Vs Nano-KAg pigs, respectively.
K-Ag vaccinated pigs had significantly increased TGF-β levels in serum compared to other experimental groups at PC 0. Unvaccinated virus challenged pigs also had an increased trend in serum TGF-β levels at both PC 7 and PC 15 (Fig. 3b). However, Nano-KAg vaccinated pigs had significantly reduced TGF-β levels in serum at PC 7 and 15 (Fig. 3b). The proinflammatory cytokine, IL-6, in the lung homogenate was significantly reduced in Nano-KAg immunized and K-Ag immunized compared to unvaccinated pigs. Moreover, reduction in IL-6 in Nano-KAg inoculated pigs was significantly lower than K-Ag immunized pigs (Fig. 3d). Both the immunosuppressive cytokines (IL-10 and TGF-β) and a Th2 cytokine, IL-4, in the lung homogenate of Nano-KAg and K-Ag immunized pigs were significantly reduced compared to unvaccinated pigs at PC 15 (Fig. 3e–g).
3.5. Enhanced Th1 and reduced proinflammatory recall cytokines response in Nano-KAg vaccinated pigs
Mononuclear cells isolated from the lungs, lymph nodes, and blood was restimulated in the presence of semi-purified killed VR2332 antigens to assess the recall cytokine response. In the culture supernatant of PBMC, BAL, and TBLN of Nano-KAg immunized pigs a significantly increased Th1 cytokines (IL-12 and IFN-γ) compared to both K-Ag and unvaccinated virus challenged pigs was observed (Fig. 4 b–d, f–h). While, LMNC of Nano-KAg immunized pigs secreted significantly higher Th1 cytokines in comparison to unvaccinated (but not to K-Ag vaccinated) pigs (Fig. 4a and e).
Fig. 4.
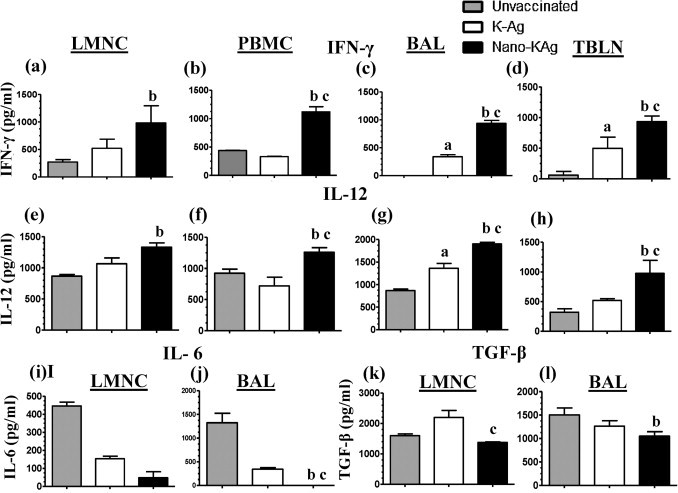
Analysis of PRRSV specific recall cytokine response. Indicated mononuclear cells were restimulated and the harvested culture supernatant was analyzed for cytokines by ELISA: (a–d) IFN-γ; (e–h) IL-12; (i and j) IL-6; and (k and l) TGF-β. Cytokines secreted by immune cells cultured in the absence of PRRSV antigens was subtracted from the test values. Each bar in the graph represents geometric mean cytokine amount from three pigs ± SEM. Alphabet ‘a’, ‘b’ and ‘c’ represents the statistical significant difference (p < 0.05) between unvaccinated vs K-Ag, unvaccinated vs Nano-KAg, and K-Ag vs Nano-KAg pigs, respectively.
Secretion of proinflammatory cytokine, IL-6, in LMNC and BAL of Nano-KAg immunized pigs was significantly reduced compared to unvaccinated and K-Ag immunized pigs (Fig. 4i and j). The Nano-KAg immunized pigs secreted significantly less of TGF-β in LMNC cultures compared to K-Ag immunized group (Fig. 4k) and in BAL compared to unvaccinated pigs (Fig. 4l). We detected production of IL-10 by LMNC in the recall response, but the levels were not significantly different (data not shown).
3.6. Phenotypic analysis of different immune cells
The flow cytometric analysis was performed once on the day of euthanasia (PC 15). The frequency of total lymphocyte population (CD3+) was significantly increased in BAL-MNC of Nano-KAg and K-Ag immunized compared to unvaccinated pigs (Fig. 5c). In the LMNC of Nano-KAg immunized pigs, a significantly higher frequency of CD4+, CD8+, and CD4CD8 double positive T cells compared to unvaccinated virus challenged pigs was detected (Fig. 5e, i, and m). In BAL-MNC of Nano-KAg received pigs a similar significant increase in lymphocyte subsets was observed (Fig. 5c, g, and o); in addition, an increase in CD3+CD4+ T cells was significantly higher than both the control pig groups (Fig. 5k).
Fig. 5.
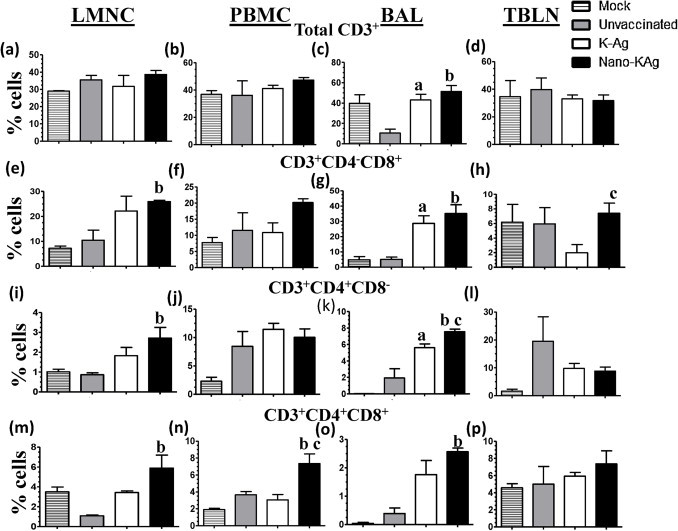
Flow cytometric analysis of CD and CD8 positive T cell subsets. Indicated mononuclear cells were immunostained to analyze the frequency of immune cells: (a–d) CD3+ cells; (e–h) CD8+ T cells; (i–l) CD4+ T cells; and (m–p) CD4+CD8+ T cells. Each bar in the graph represents geometric mean of percent of immune cells from three pigs ± SEM. Alphabet ‘a’, ‘b’ and ‘c’ represents the statistical significant difference (p < 0.05) between unvaccinated vs K-Ag, unvaccinated vs Nano-KAg, and K-Ag Vs Nano-KAg pigs, respectively.
In Nano-KAg and K-Ag received pigs, γδ T cells frequency in LMNC, PBMC, BAL, and TBLN-MNC was significantly higher compared to unvaccinated pigs (Fig. 6a–d). In addition, γδ T cell population in LMNC and BAL-MNC of Nano-KAg pigs was significantly higher than K-Ag vaccinated group (Fig. 6a and c). Myeloid cells in Nano-KAg pigs were increased significantly in LMNC compared to unvaccinated virus challenged group (Fig. 6e) and in PBMC compared to K-Ag immunized pigs (Fig. 6f). DCs rich fraction in PBMC and LMNC of Nano-KAg vaccinated pigs was significantly higher than both unvaccinated and K-Ag vaccinated, virus challenged pigs (Fig. 6i and j). However, we did not find any significant changes in myeloid cells and DCs rich fraction in BAL and TBLN MNCs in tested pigs (Fig. 6g, h, and k).
Fig. 6.
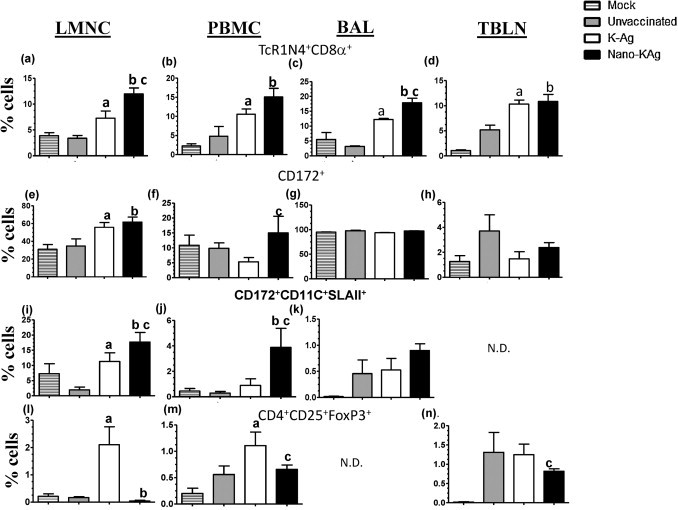
Flow cytometry analysis of innate and regulatory T cells. Indicated mononuclear cells were immunostained to determine the frequency of immune cells: (a–d) γδ T cells; (e–h) myeloid cells; (i–k) dendritic cells rich fraction; and (l–n) Tregs. Each bar in the graph represents geometric mean of percent of immune cells from three pigs ± SEM. Alphabet ‘a’, ‘b’ and ‘c’ represents the statistical significant difference (p < 0.05) between unvaccinated vs K-Ag, unvaccinated vs Nano-KAg, and K-Ag vs Nano-KAg pigs, respectively.
Frequency of Tregs (CD4+CD25+Foxp3+) in LMNC was significantly reduced in Nano-KAg immunized compared to unvaccinated pigs (Fig. 6l). While in pigs immunized with K-Ag, a significantly higher Tregs population in LMNC than unvaccinated pigs was observed (Fig. 6l). The Tregs frequency in PBMC was significantly reduced in Nano-KAg vaccinated, and significantly increased in K-Ag vaccinated compared to unvaccinated pigs (Fig. 6m). In addition, TBLN of Nano-KAg vaccinated pigs had significantly higher population of Tregs compared to K-Ag received pigs (Fig. 6n).
4. Discussion
So far researchers have made several efforts to develop protective killed PRRSV vaccine, but with limited success (Charerntantanakul, 2009). The possible reasons include: (i) inability to deliver killed PRRSV vaccine to pig immune system; (ii) like live PRRSV, killed virus may also be immunosuppressive; (iii) the antigenic mass used in killed vaccine may be insufficient to induce protective immunity; and (iv) the administered killed PRRSV vaccine failed to elicit adequate cell-mediated immune response. This suggests the need of potent adjuvant and/or novel delivery system to improve the efficacy of killed PRRSV vaccine.
Clinically, in VR2332 challenged pigs disease symptoms were absent. We used the VR2332 strain which was passaged several times (∼50) in vitro, and the lack of obvious clinical disease may have been due to attenuation. Macroscopically, typical PRRS lung lesions were not very distinct; therefore, in this paper we did not make any comparative lung lesion analyses, but reported the comprehensive immunological data in relation to viremia. PRRSV neutralizing antibodies protect pigs against viremia, virus replication in the lungs, and transplacental spread of the virus (Lopez et al., 2007, Lopez and Osorio, 2004), but the commercial killed vaccines induce negligible VN antibody response (Bassaganya-Riera et al., 2004, Vanhee et al., 2009). A study demonstrated that it is possible to elicit increased VN titers to a killed PRRSV vaccine by inactivating the virus using UV or binary ethylenimine and by coadministering the vaccine with a potent adjuvant (Vanhee et al., 2009). In that study amount of inactivated PRRSV in each vaccine dose was 1 × 108 TCID50 (Vanhee et al., 2009). In our study, each vaccine dose had approximately 5 × 106 TCID50 of UV-treated virus, suggesting PLGA nanoparticle-based intranasal delivery of killed PRRSV vaccine needs significantly reduced virus amount to elicit anti-PRRSV immune response. Recently, it was shown that PLGA nanoparticle-based tumor antigenic peptides vaccine delivery could reduce the required vaccine dose by 63 times (Ma et al., 2012); indicating that such a delivery system has the potential to significantly reduce the required amount of killed PRRSV antigens in each dose.
PRRSV VN antibody in the serum was shown to be important for viral clearance (Lopez et al., 2007, Lopez and Osorio, 2004, Osorio et al., 2002), but it needs to be further investigated due to lack of complete virus clearance even in the presence of VN antibodies in pigs (Diaz et al., 2005) (Michael Murtaugh, personal communication). Although porcine lung Ig is primarily IgG, generation of protective IgA response is essential to reduce the entry of pathogens whose principle port of entry is through mucosal surfaces (McGhee et al., 1992). Mucosal immunization elicits production of IgA antibodies (Ogra and Karzon, 1969), and the IgA antibody is protective in clearing the virus infection by their virus neutralization activity (Liew et al., 1984). But, we lack convincing studies about the role of lung IgA in PRRSV clearance in infected or vaccinated pigs. In this study the levels of PRRSV specific IgA antibody in the lungs of both Nano-KAg and K-Ag vaccinated pigs was found to be high and comparable; but, significant increase in lung VN titers was detected only in Nano-KAg received pigs. Further, in Nano-KAg immunized pigs increased levels of IFN-γ response was observed both in the lungs and blood. In the serum of pigs unvaccinated or vaccinated with K-Ag and virus challenged, detectable VN titers were absent. But, Nano-KAg vaccine induced low levels of VN titers (1:2 and 1:4) in serum, and it might be due to nonspecific effects, as such low titers did not boost upon the viral challenge. This suggests the need of a potent adjuvant and/or a booster dose of the Nano-KAg vaccine to induce high PRRS VN titers in the serum. Further analysis in the lymphoid tissues and lungs is necessary to confirm the effect of Nano-KAg vaccine in viral clearance in challenged animals.
Nanoparticles prepared from PLGA containing hepatitis B, rotavirus, influenza, or parainfluenza viruses delivered to mucosal sites of mice generated protective immune response (Thomas et al., 2011). Biodegradable and biocompatible PLGA nanoparticles are free from any toxicity, safe to use, and are approved by the US Food and Drug Administration (Duncan, 2005). In our study, PLGA nanoparticles did not induce any toxic symptoms in vaccinated pigs. Further, due to particulate nature of nanoparticles and the inherent ability of professional antigen presenting cells (APCs) to passively phagocytoze particulate antigens (Inaba et al., 1993), nanoparticle-based delivery of vaccines to mucosal sites is advantageous. To note, abundant populations of APCs are present at mucosal tissues and lymphoid organs; and in BAL fluid of mice, humans, and pigs greater than 90% of cells are macrophages, suggesting that intranasally administered Nano-KAg was probably phagocytozed by alveolar macrophages. Earlier studies with killed influenza virus vaccine entrapped in nanoparticles administered with an adjuvant intranasally to mice, rabbits, and pigs elicited protective immune response and the response induced in pigs was significantly better than by intramuscular vaccination (Singh et al., 2001). Therefore, innovative nanotechnology based intranasal vaccine delivery is an attractive alternative approach to deliver killed PRRSV vaccine to pigs.
Insufficient anti-PRRSV immunity in vaccinated and PRRSV-infected pigs was also linked to low levels of IFN-γ production (Suradhat et al., 2003). Enhanced IFN-γ response and antigen specific T-cell response (CD4+ and CD8+ T cells) is important for anti-PRRSV immunity (Xiao et al., 2004). We have detected a significant increase in IFN-γ levels and CD4+ and CD8+ T cell population in the lungs and blood of Nano-KAg vaccinated pigs. In pigs, T lymphocyte subset with CD4+CD8+ phenotype are abundant and they possess memory, T-helper, and cytolytic properties (Zuckermann, 1999). In Nano-KAg immunized pigs, there was a significant increase in CD4+CD8+ T cells frequency in the lungs and blood. In pigs infected with the European strain of PRRSV a negative correlation between the frequency of CD4+CD8+ T cells and viremia was reported, suggesting the protective role of CD4+CD8+ T cells in PRRSV immunity (Gomez-Laguna et al., 2009).
Recall cytokine response in LMNC, PBMC, BAL, and TBLN-MNC of Nano-KAg immunized pigs revealed secretion of significantly increased Th1 cytokines. The cytokine IL-12 plays an important role in host defense against viral infections by inducing IFN-γ production by NK cells, and also helps in differentiation of Th1 cell phenotype (Kaplan et al., 1996). Co-administration of IL-12 and PRRS-MLV enhanced the frequency of IFN-γ secreting cells (Foss et al., 2002). Enhanced secretion of Th1 cytokines is associated with protective cell-mediated immune response in mice delivered intranasally with PLGA-based hepatitis B virus vaccine (Jaganathan and Vyas, 2006). In addition, we also observed significantly increased myeloid and dendritic cell rich population in LMNC and PBMC of Nano-KAg compared to K-Ag vaccinated pigs. Unlike other non-lymphoid tissues the lung contains a large fraction of effector and central memory CD8+ T lymphocytes (Anderson et al., 2012, Unsoeld and Pircher, 2005); in fact, even naïve lymphocytes are present in the perfused lung (Cose et al., 2006). Since we included LMNC to analyze phenotypic analysis of immune cells our study represented the mucosal immune responses in the pig lungs against PRRSV.
A significant reduction in immunosuppressive cytokines (IL-10 and TGF-β) in the lung homogenate of Nano-KAg immunized pigs was associated with enhanced IFN-γ production. In pigs, the cytokine IL-10 inhibits IFN-γ production by T cells (Waters et al., 1999), and its immunosuppressive function with reduced IFN-γ production was reported in PRRSV infected pigs (Charerntantanakul et al., 2006). Earlier studies by others (Barbe et al., 2011) and us (Renukaradhya et al., 2010) have confirmed that pig lung homogenate provides reproducible and comparable data obtained by BAL fluid. Unlike the lung homogenate, the method of collection and the dilution factor in the BAL fluid may significantly alter the data from lab to lab, and thus lung homogenate may be a suitable sample for lung immune analysis with minimal variations. At this point due to absence of data from BAL fluid we could not conclude which sample is better in PRRSV vaccinated and/or challenged animals.
Infiltration of Tregs in infected pig lung microenvironment contributes to secretion of high levels of IL-10 and TGF-β (Didierlaurent et al., 2007). An increased frequency of Tregs in PRRSV infected pigs is reported (Silva-Campa et al., 2009). In Nano-KAg (but not K-Ag) vaccinated pigs reduction in Tregs population in LMNC, PBMC, and TBLN was observed, signifying that our vaccination strategy dampened the Tregs response and favored the PRRSV specific Th1 and Th2 responses. We performed the immune cell analyses at a single time point and our data although supported the viral clearance, considering the nature of macrophage, neutrophil and lymphocyte trafficking and recruiting, it is difficult to precisely document the direct correlation of our immune cell phenotypic data with the observed response.
Pigs possess relatively a large population of γδ T cells compared to other species, and it is considered as an important innate immune cell at mucosal sites (Olin et al., 2005). In Nano-KAg vaccinated pigs there was an increased frequency of γδ T cells in the lungs, blood, and TBLN, suggesting the possible protective role played by γδ T cells in PRRSV immunity. In conclusion, our study for the first time demonstrated the efficacy of PLGA nanoparticle-encapsulated PRRSV killed vaccine in inducing anti-PRRSV specific humoral and cell-mediated immune responses, which concertedly contributed in better clearance of viremia compared to control killed vaccine. Further studies are required to evaluate anti-PRRSV cross-protective immunity in vaccinated pigs. In summary, intranasal delivery of Nano-KAg vaccine has a promise, but needs further investigation and improvement.
Acknowledgments
This work was supported by National Pork Board (NPB# 09-213) and USDA-NIFA PRRSCAP2 (2008-55620-19132) awards to GJR. Salaries and research support were provided by state and federal funds appropriated to Ohio Agricultural Research and Development Center, The Ohio State University. We would like to thank Dr. Eric Nelson (South Dakota State University) for providing VR2332 strain of PRRSV and control serum samples, and Dr. Murtaugh (University of Minnesota) has provided the PRRSV N′ protein. We would like to thank Ms. Ruthi Patterson, Drs. Mahesh Khatri and Juliette Hanson, and Andrew Wright for their help in animal studies.
References
- Albina E., Carrat C., Charley B. Interferon-alpha response to swine arterivirus (PoAV), the porcine reproductive and respiratory syndrome virus. J. Interferon Cytokine Res. 1998;18:485–490. doi: 10.1089/jir.1998.18.485. [DOI] [PubMed] [Google Scholar]
- Anderson K.G., Sung H., Skon C.N., Lefrancois L., Deisinger A., Vezys V., Masopust D. Cutting edge: intravascular staining redefines lung CD8 T cell responses. J. Immunol. 2012;189:2702–2706. doi: 10.4049/jimmunol.1201682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker R.B., Yu W., Fuentes M., Johnson C.R., Peterson L., Rossow K., Daniels C.S., Daniels A.M., Polson D., Murtaugh M.P. Prairie dog (Cynomys ludovicianus) is not a host for porcine reproductive and respiratory syndrome virus. J. Swine Health. Prod. 2007;15:22–29. [Google Scholar]
- Barbe F., Atanasova K., Van Reeth K. Cytokines and acute phase proteins associated with acute swine influenza infection in pigs. Vet. J. 2011;187:48–53. doi: 10.1016/j.tvjl.2009.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassaganya-Riera J., Thacker B.J., Yu S., Strait E., Wannemuehler M.J., Thacker E.L. Impact of immunizations with porcine reproductive and respiratory syndrome virus on lymphoproliferative recall responses of CD8+ T cells. Viral Immunol. 2004;17:25–37. doi: 10.1089/088282404322875430. [DOI] [PubMed] [Google Scholar]
- Cao X., Schoichet M.S. Delivering neuroactive molecules from biodegradable microspheres for application in central nervous system disorders. Biomaterials. 1999;20:329–339. doi: 10.1016/s0142-9612(98)00172-0. [DOI] [PubMed] [Google Scholar]
- Charerntantanakul W. Adjuvants for porcine reproductive and respiratory syndrome virus vaccines. Vet. Immunol. Immunopathol. 2009;129:1–13. doi: 10.1016/j.vetimm.2008.12.018. [DOI] [PubMed] [Google Scholar]
- Charerntantanakul W., Platt R., Roth J.A. Effects of porcine reproductive and respiratory syndrome virus-infected antigen-presenting cells on T cell activation and antiviral cytokine production. Viral Immunol. 2006;19:646–661. doi: 10.1089/vim.2006.19.646. [DOI] [PubMed] [Google Scholar]
- Christopher-Hennings J., Holler L.D., Benfield D.A., Nelson E.A. Detection and duration of porcine reproductive and respiratory syndrome virus in semen, serum, peripheral blood mononuclear cells, and tissues from Yorkshire, Hampshire, and Landrace boars. J. Vet. Diagn. Invest. 2001;13:133–142. doi: 10.1177/104063870101300207. [DOI] [PubMed] [Google Scholar]
- Cose S., Brammer C., Khanna K.M., Masopust D., Lefrancois L. Evidence that a significant number of naive T cells enter non-lymphoid organs as part of a normal migratory pathway. Eur. J. Immunol. 2006;36:1423–1433. doi: 10.1002/eji.200535539. [DOI] [PubMed] [Google Scholar]
- Diaz I., Darwich L., Pappaterra G., Pujols J., Mateu E. Immune responses of pigs after experimental infection with a European strain of Porcine reproductive and respiratory syndrome virus. J. Gen. Virol. 2005;86:1943–1951. doi: 10.1099/vir.0.80959-0. [DOI] [PubMed] [Google Scholar]
- Didierlaurent A., Goulding J., Hussell T. The impact of successive infections on the lung microenvironment. Immunology. 2007;122:457–465. doi: 10.1111/j.1365-2567.2007.02729.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Done S.H., Paton D.J., White M.E. Porcine reproductive and respiratory syndrome (PRRS): a review, with emphasis on pathological, virological and diagnostic aspects. Br. Vet. J. 1996;152:153–174. doi: 10.1016/S0007-1935(96)80071-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan R. Nanomedicine gets clinical. Mater. Today. 2005;8:16–17. [Google Scholar]
- Dwivedi V., Manickam C., Binjawadagi B., Linhares D., Murtaugh M.P., Renukaradhya G.J. Evaluation of immune responses to porcine reproductive and respiratory syndrome virus in pigs during early stage of infection under farm conditions. Virol. J. 2012;9:45. doi: 10.1186/1743-422X-9-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dwivedi V., Manickam C., Patterson R., Dodson K., Murtaugh M., Torrelles J.B., Schlesinger L.S., Renukaradhya G.J. Cross-protective immunity to porcine reproductive and respiratory syndrome virus by intranasal delivery of a live virus vaccine with a potent adjuvant. Vaccine. 2011;29:4058–4066. doi: 10.1016/j.vaccine.2011.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dwivedi V., Manickam C., Patterson R., Dodson K., Weeman M., Renukaradhya G.J. Intranasal delivery of whole cell lysate of Mycobacterium tuberculosis induces protective immune responses to a modified live porcine reproductive and respiratory syndrome virus vaccine in pigs. Vaccine. 2011;29:4067–4076. doi: 10.1016/j.vaccine.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eldridge J.H., Gilley R.M., Staas J.K., Moldoveanu Z., Meulbroek J.A., Tice T.R. Biodegradable microspheres: vaccine delivery system for oral immunization. Curr. Top. Microbiol. Immunol. 1989;146:59–66. doi: 10.1007/978-3-642-74529-4_6. [DOI] [PubMed] [Google Scholar]
- Foss D.L., Zilliox M.J., Meier W., Zuckermann F., Murtaugh M.P. Adjuvant danger signals increase the immune response to porcine reproductive and respiratory syndrome virus. Viral Immunol. 2002;15:557–566. doi: 10.1089/088282402320914502. [DOI] [PubMed] [Google Scholar]
- Gomez-Laguna J., Salguero F.J., De Marco M.F., Pallares F.J., Bernabe A., Carrasco L. Changes in lymphocyte subsets and cytokines during European porcine reproductive and respiratory syndrome: increased expression of IL-12 and IL-10 and proliferation of CD4(−)CD8(high) Viral Immunol. 2009;22:261–271. doi: 10.1089/vim.2009.0003. [DOI] [PubMed] [Google Scholar]
- Gupta R.K., Chang A.C., Siber G.R. Biodegradable polymer microspheres as vaccine adjuvants and delivery systems. Dev. Biol. Stand. 1998;92:63–78. [PubMed] [Google Scholar]
- Holmgren J., Czerkinsky C., Lycke N., Svennerholm A.M. Mucosal immunity: implications for vaccine development. Immunobiology. 1992;184:157–179. doi: 10.1016/S0171-2985(11)80473-0. [DOI] [PubMed] [Google Scholar]
- Holtkamp, D., Kliebenstein, J., 2011. PRRS Costs Industry $664 Million Annually. Pork Checkoff Study, http://www.pork.org/News/1265/PRRSCostsIndustry664Million.aspx.
- Inaba K., Inaba M., Naito M., Steinman R.M. Dendritic cell progenitors phagocytose particulates, including bacillus Calmette–Guerin organisms, and sensitize mice to mycobacterial antigens in vivo. J. Exp. Med. 1993;178:479–488. doi: 10.1084/jem.178.2.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaganathan K.S., Vyas S.P. Strong systemic and mucosal immune responses to surface-modified PLGA microspheres containing recombinant hepatitis B antigen administered intranasally. Vaccine. 2006;24:4201–4211. doi: 10.1016/j.vaccine.2006.01.011. [DOI] [PubMed] [Google Scholar]
- Johnson C.R., Yu W., Murtaugh M.P. Cross-reactive antibody responses to nsp1 and nsp2 of Porcine reproductive and respiratory syndrome virus. J. Gen. Virol. 2007;88:1184–1195. doi: 10.1099/vir.0.82587-0. [DOI] [PubMed] [Google Scholar]
- Kaplan M.H., Sun Y.L., Hoey T., Grusby M.J. Impaired IL-12 responses and enhanced development of Th2 cells in Stat4-deficient mice. Nature. 1996;382:174–177. doi: 10.1038/382174a0. [DOI] [PubMed] [Google Scholar]
- Keffaber K.K. Reproductive failure of unknown etiology. Am. Assoc. Swine Pract. Newsl. 1989;1:1–9. [Google Scholar]
- Kim W.I., Lee D.S., Johnson W., Roof M., Cha S.H., Yoon K.J. Effect of genotypic and biotypic differences among PRRS viruses on the serologic assessment of pigs for virus infection. Vet. Microbiol. 2007;123:1–14. doi: 10.1016/j.vetmic.2007.03.007. [DOI] [PubMed] [Google Scholar]
- King A.M.Q., Lefkowitz E., Adams M.J., Carstens E.B. Ninth Report of the International Committee on Taxonomy of Viruses International Union of Microbiological Societies, Virology Division. 2011. Virus taxonomy. [Google Scholar]
- Li B., Fang L., Xu Z., Liu S., Gao J., Jiang Y., Chen H., Xiao S. Recombination in vaccine and circulating strains of porcine reproductive and respiratory syndrome viruses. Emerg. Infect. Dis. 2009;15:2032–2035. doi: 10.3201/eid1512.090390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew F.Y., Russell S.M., Appleyard G., Brand C.M., Beale J. Cross-protection in mice infected with influenza A virus by the respiratory route is correlated with local IgA antibody rather than serum antibody or cytotoxic T cell reactivity. Eur. J. Immunol. 1984;14:350–356. doi: 10.1002/eji.1830140414. [DOI] [PubMed] [Google Scholar]
- Lopez O.J., Oliveira M.F., Garcia E.A., Kwon B.J., Doster A., Osorio F.A. Protection against porcine reproductive and respiratory syndrome virus (PRRSV) infection through passive transfer of PRRSV-neutralizing antibodies is dose dependent. Clin. Vaccine Immunol. 2007;14:269–275. doi: 10.1128/CVI.00304-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez O.J., Osorio F.A. Role of neutralizing antibodies in PRRSV protective immunity. Vet. Immunol. Immunopathol. 2004;102:155–163. doi: 10.1016/j.vetimm.2004.09.005. [DOI] [PubMed] [Google Scholar]
- Lu J.M., Wang X., Marin-Muller C., Wang H., Lin P.H., Yao Q., Chen C. Current advances in research and clinical applications of PLGA-based nanotechnology. Expert Rev. Mol. Diagn. 2009;9:325–341. doi: 10.1586/erm.09.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma W., Chen M., Kaushal S., McElroy M., Zhang Y., Ozkan C., Bouvet M., Kruse C., Grotjahn D., Ichim T., Minev B. PLGA nanoparticle-mediated delivery of tumor antigenic peptides elicits effective immune responses. Int. J. Nanomed. 2012;7:1475–1487. doi: 10.2147/IJN.S29506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manickam C., Dwivedi V., Patterson R., Papenfuss T., Renukaradhya G.J. Porcine reproductive and respiratory syndrome virus induces pronounced immune modulatory responses at mucosal tissues in the parental vaccine strain VR2332 infected pigs. Vet. Microbiol. 2013;162:68–77. doi: 10.1016/j.vetmic.2012.08.021. [DOI] [PubMed] [Google Scholar]
- Mateu E., Diaz I. The challenge of PRRS immunology. Vet. J. 2008;177:345–351. doi: 10.1016/j.tvjl.2007.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGhee J.R., Mestecky J., Dertzbaugh M.T., Eldridge J.H., Hirasawa M., Kiyono H. The mucosal immune system: from fundamental concepts to vaccine development. Vaccine. 1992;10:75–88. doi: 10.1016/0264-410x(92)90021-b. [DOI] [PubMed] [Google Scholar]
- Mengeling W.L., Clouser D.F., Vorwald A.C., Lager K.M. The potential role of genetic recombination in the evolution of new strains of porcine reproductive and respiratory syndrome virus (PRRSV) J Swine Health Prod. 2002;10:273–275. [Google Scholar]
- Mengeling W.L., Lager K.M., Vorwald A.C., Koehler K.J. Strain specificity of the immune response of pigs following vaccination with various strains of porcine reproductive and respiratory syndrome virus. Vet. Microbiol. 2003;93:13–24. doi: 10.1016/s0378-1135(02)00427-3. [DOI] [PubMed] [Google Scholar]
- Nielsen H.S., Oleksiewicz M.B., Forsberg R., Stadejek T., Botner A., Storgaard T. Reversion of a live porcine reproductive and respiratory syndrome virus vaccine investigated by parallel mutations. J. Gen. Virol. 2001;82:1263–1272. doi: 10.1099/0022-1317-82-6-1263. [DOI] [PubMed] [Google Scholar]
- Ogra P.L., Karzon D.T. Distribution of poliovirus antibody in serum, nasopharynx and alimentary tract following segmental immunization of lower alimentary tract with poliovaccine. J. Immunol. 1969;102:1423–1430. [PubMed] [Google Scholar]
- Olin M.R., Hwa Choi K., Lee J., Molitor T.W. Gammadelta T-lymphocyte cytotoxic activity against Mycobacterium bovis analyzed by flow cytometry. J. Immunol. Methods. 2005;297:1–11. doi: 10.1016/j.jim.2004.10.002. [DOI] [PubMed] [Google Scholar]
- Osorio F.A., Galeota J.A., Nelson E., Brodersen B., Doster A., Wills R., Zuckermann F., Laegreid W.W. Passive transfer of virus-specific antibodies confers protection against reproductive failure induced by a virulent strain of porcine reproductive and respiratory syndrome virus and establishes sterilizing immunity. Virology. 2002;302:9–20. doi: 10.1006/viro.2002.1612. [DOI] [PubMed] [Google Scholar]
- Panyam J., Labhasetwar V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv. Drug Deliv. Rev. 2003;55:329–347. doi: 10.1016/s0169-409x(02)00228-4. [DOI] [PubMed] [Google Scholar]
- Piras F., Bollard S., Laval F., Joisel F., Reynaud G., Charreyre C., Andreoni C., Juillard V. Porcine reproductive and respiratory syndrome (PRRS) virus-specific interferon-gamma(+) T-cell responses after PRRS virus infection or vaccination with an inactivated PRRS vaccine. Viral Immunol. 2005;18:381–389. doi: 10.1089/vim.2005.18.381. [DOI] [PubMed] [Google Scholar]
- Rajapaksa T.E., Bennett K.M., Hamer M., Lytle C., Rodgers V.G., Lo D.D. Intranasal M cell uptake of nanoparticles is independently influenced by targeting ligands and buffer ionic strength. J. Biol. Chem. 2010;285:23739–23746. doi: 10.1074/jbc.M110.126359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renukaradhya G.J., Alekseev K., Jung K., Fang Y., Saif L.J. Porcine reproductive and respiratory syndrome virus-induced immunosuppression exacerbates the inflammatory response to porcine respiratory coronavirus in pigs. Viral Immunol. 2010;23:457–466. doi: 10.1089/vim.2010.0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva-Campa E., Cordoba L., Fraile L., Flores-Mendoza L., Montoya M., Hernandez J. European genotype of porcine reproductive and respiratory syndrome (PRRSV) infects monocyte-derived dendritic cells but does not induce Treg cells. Virology. 2009;396:264–271. doi: 10.1016/j.virol.2009.10.024. [DOI] [PubMed] [Google Scholar]
- Silva-Campa E., Mata-Haro V., Mateu E., Hernandez J. Porcine reproductive and respiratory syndrome virus induces CD4(+)CD8(+)CD25(+)Foxp3(+) regulatory T cells (Tregs) Virology. 2012;430:73–80. doi: 10.1016/j.virol.2012.04.009. [DOI] [PubMed] [Google Scholar]
- Singh M., Briones M., O‘Hagan D.T. A novel bioadhesive intranasal delivery system for inactivated influenza vaccines. J. Control. Release. 2001;70:267–276. doi: 10.1016/s0168-3659(00)00330-8. [DOI] [PubMed] [Google Scholar]
- Spiers I.D., Eyles J.E., Baillie L.W., Williamson E.D., Alpar H.O. Biodegradable microparticles with different release profiles: effect on the immune response after a single administration via intranasal and intramuscular routes. J. Pharm. Pharmacol. 2000;52:1195–1201. doi: 10.1211/0022357001777324. [DOI] [PubMed] [Google Scholar]
- Suradhat S., Thanawongnuwech R., Poovorawan Y. Upregulation of IL-10 gene expression in porcine peripheral blood mononuclear cells by porcine reproductive and respiratory syndrome virus. J. Gen. Virol. 2003;84:453–459. doi: 10.1099/vir.0.18698-0. [DOI] [PubMed] [Google Scholar]
- Thomas C., Rawat A., Hope-Weeks L., Ahsan F. Aerosolized PLA and PLGA nanoparticles enhance humoral, mucosal and cytokine responses to hepatitis B vaccine. Mol. Pharm. 2011;8:405–415. doi: 10.1021/mp100255c. [DOI] [PubMed] [Google Scholar]
- Thomasin C., Corradin G., Men Y., Merkle H.P., Gander B. Tetanus toxoid and malaria antigen containing poly(lactide)/poly(lactide-co-glycolide) microspheres: importance of polymer degradation antigen release for immune response. J. Control. Release. 1996;41:131–145. [Google Scholar]
- Unsoeld H., Pircher H. Complex memory T-cell phenotypes revealed by coexpression of CD62L and CCR7. J. Virol. 2005;79:4510–4513. doi: 10.1128/JVI.79.7.4510-4513.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanhee M., Delputte P.L., Delrue I., Geldhof M.F., Nauwynck H.J. Development of an experimental inactivated PRRSV vaccine that induces virus-neutralizing antibodies. Vet. Res. 2009;40:63. doi: 10.1051/vetres/2009046. [DOI] [PubMed] [Google Scholar]
- Waters W.R., Sacco R.E., Dorn A.D., Hontecillas R., Zuckermann F.A., Wannemuehler M.J. Systemic and mucosal immune responses of pigs to parenteral immunization with a pepsin-digested Serpulina hyodysenteriae bacterin. Vet. Immunol. Immunopathol. 1999;69:75–87. doi: 10.1016/s0165-2427(99)00043-4. [DOI] [PubMed] [Google Scholar]
- Xiao Z., Batista L., Dee S., Halbur P., Murtaugh M.P. The level of virus-specific T-cell and macrophage recruitment in porcine reproductive and respiratory syndrome virus infection in pigs is independent of virus load. J. Virol. 2004;78:5923–5933. doi: 10.1128/JVI.78.11.5923-5933.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuckermann F.A. Extrathymic CD4/CD8 double positive T cells. Vet. Immunol. Immunopathol. 1999;72:55–66. doi: 10.1016/s0165-2427(99)00118-x. [DOI] [PubMed] [Google Scholar]


