Abstract
Coinfection with two or more pathogens is a common occurrence in respiratory diseases of most species. The manner in which multiple pathogens interact is not always straightforward, however. Bordetella bronchiseptica and porcine respiratory coronavirus (PRCV) are respiratory pathogens of pigs whose relatives, B. pertussis and the SARS virus, cause respiratory disease in humans. In an initial experiment, the effect of coinfection of PRCV and B. bronchiseptica was examined in thirty, 4-week-old pigs (10 pigs/group) that were infected with either PRCV or B. bronchiseptica, or both PRCV and B. bronchiseptica. An additional 10 pigs served as sham infected controls. Five pigs from each group were euthanized at 4 and 10 days post-infection. Gross and histopathological lung lesions were more severe in the coinfected group as compared to the groups infected with B. bronchiseptica or PRCV alone. In order to investigate the potential role of proinflammatory cytokines in disease severity after coinfection, a second experiment was performed to examine cytokine transcription in alveolar macrophages from single and dually infected pigs. A total of 48 pigs were divided equally into groups as above, but 4 pigs from each group were euthanized at 1, 4 and 10 days post-infection. Coinfected pigs showed a greater and more sustained transcription of proinflammatory cytokines, especially IL-6 and MCP-1, than pigs infected with either PRCV or B. bronchiseptica alone. Thus, there appears to be a synergistic effect between PRCV and B. bronchiseptica with regards to proinflammatory cytokine transcription that may partially explain the increased severity of pneumonia in coinfected pigs.
Keywords: Porcine respiratory coronavirus, Bordetella bronchiseptica, Porcine respiratory disease complex
1. Introduction
Respiratory disease in swine, as with other species, is generally considered a multifactorial problem caused by a combination of viral and bacterial infectious agents, as well as adverse environmental conditions. The list of infectious agents that cause respiratory disease in swine is extensive and includes both viral agents, such as porcine reproductive and respiratory syndrome virus (PRRSV), swine influenza virus (SIV) and porcine respiratory coronavirus (PRCV); and bacterial agents, such as Mycoplasma hyopneumoniae, Bordetella bronchiseptica, Pasteurella multocida and Actinobacillus pleuropneumoniae. Although any one of these pathogens can potentially cause disease on its own, more serious and chronic respiratory disease results, and more economic losses are incurred, when infection with multiple pathogens occurs. Although the multifactorial nature of respiratory disease is well accepted, the specific mechanisms by which respiratory pathogens interact with each other or the host to cause more severe disease are poorly understood.
Coronaviruses are enveloped, single-stranded, RNA viruses which cause respiratory and enteric disease in animals, including the common cold and severe acute respiratory syndrome (SARS) in humans. Porcine respiratory coronavirus is a naturally occurring deletion mutant of transmissible gastroenteritis virus (TGE), an enteric coronavirus of swine (Rasschaert et al., 1990, Britton et al., 1991, Wesley et al., 1991). A deletion in the spike gene of TGE has altered the tropism of PRCV from primarily enteric to primarily respiratory in nature. Although experimentally PRCV has been shown to cause bronchointersitial pneumonia, the clinical relevance of this virus has been debated, since many farms seroconvert without overt clinical signs of disease. What role, if any, PRCV plays in multifactorial respiratory disease is still being investigated. Prior work has shown that exposure to PRCV decreases replication of SIV, in vivo, and mixed results have been reported with coinfections of PRCV and PRRSV (Van Reeth and Pensaert, 1994, Van Reeth and Nauwynck, 2000). However, experimental exposure of pigs to bacterial lipopolysaccharide (LPS) following PRCV infection led to more severe respiratory disease and increased tumor necrosis factor-alpha (TNF-alpha) and interleukin (IL)-1 levels than exposure to either LPS or PRCV alone (Van Reeth et al., 2000). Thus, infection with PRCV and gram-negative bacteria may result in a more severe disease.
The species belonging to the genus Bordetella are closely related gram-negative bacteria which cause respiratory disease in animals. Bordetella pertussis is the obligatory human pathogen that causes whooping cough, and B. bronchiseptica is the more promiscuous pathogen that is the etiologic agent of respiratory disease in a number of animals including swine. Although B. bronchiseptica can cause pneumonia in neonatal pigs and rhinitis and bronchitis in older swine on its own, its role in multifactorial disease is also under investigation. Prior infection with B. bronchiseptica in swine has led to increased colonization and more severe disease with other bacterial agents such as P. multocida, Streptococcus suis and Haemophilus parasuis (Chanter et al., 1989, Vecht et al., 1992, Brockmeier et al., 2001, Brockmeier, 2004), and coinfection of B. bronchiseptica with PRRSV in swine exacerbated clinical disease and induced B bronchiseptica pneumonia (Brockmeier et al., 2000).
The innate immune response of the lung is not only important for initial protection from infectious agents and stimulation of the adaptive immune response, but the nature of the innate response can dictate the character of the pathology observed and the outcome of infection. Proinflammatory cytokines, such as TNF-alpha, IL-1 and IL-6, as well as the chemokines IL-8 and monocyte chemotactic protein-1 (MCP-1) are often produced after recognition of pathogen associated molecular patterns for early host protection and recruitment of effector cells. Exposure to multiple infectious agents may result in distinct cytokine patterns and thus alter the severity of disease compared to that observed during infection with individual agents. In this report we determine whether coinfection with PRCV and B. bronchiseptica leads to increased severity of respiratory disease, and if so, whether or not the local proinflammatory cytokine response helps to elucidate the mechanism of this interaction. The data reported here demonstrates that coinfection results in a proinflammatory response that is different from each individual infection, and this proinflammatory response likely contributes to the differences in disease pathology observed during infection with PRCV and B. bronchiseptica.
2. Materials and methods
2.1. PRCV and B. bronchiseptica inocula
The PRCV isolate used was isolated from a pig on a farm in Iowa that was experiencing respiratory disease. This isolate of PRCV has a 648-nucleotide deletion after base 99 of the spike gene compared to the Purdue strain of TGE (data not shown). PRCV was isolated on swine testicular (ST) cells, passaged two times and frozen at −80 °C. Pigs were inoculated intranasally with 4 ml (2 ml/nostril) of cell culture medium containing 106 median cell culture infectious dose (CCID50)/ml of PRCV. Sham inoculum was prepared from uninfected ST cells.
B. bronchiseptica strain KM22 is a virulent phase I swine isolate cultured from a herd with atrophic rhinitis. To prepare the inoculum, B. bronchiseptica was cultured on Bordet-Gengou agar supplemented with 10% sheep's blood at 37 °C for 40 h and suspended in phosphate buffered saline (PBS). Pigs were inoculated intranasally with 1 ml of PBS (0.5 ml/nostril) containing 106 colony forming units (CFU)/ml of B. bronchiseptica.
2.2. Experimental design
Two experiments were performed. At 3 weeks of age, 40 pigs (experiment 1) and 48 pigs (experiment 2) were transferred from herds (a different herd in each experiment), which were negative for antibody to PRCV, PRRSV and SIV, to an animal isolation facility at the National Animal Disease Center. No medications or vaccinations were given to the piglets. Nasal swabs were obtained from all pigs before the start of the experiment, and neither B. bronchiseptica nor PRCV was isolated. The pigs were randomly divided into 4 groups with 10 pigs/group (experiment 1) and 12 pigs/group (experiment 2). After 1 week of acclimation, on day 0 of the experiment, the pigs in group 1 were intranasally inoculated with sham PBS and cell culture medium from uninfected ST cells, pigs in group 2 were inoculated intranasally with B. bronchiseptica and sham cell culture medium, the pigs in group 3 were intranasally inoculated with sham PBS and PRCV, the pigs in group 4 were inoculated with PRCV followed by B. bronchiseptica. In experiment 1, 5 pigs from each group were euthanized on days 4 and 10 of the experiment. In experiment 2, 4 pigs from each group were euthanized on days 1, 4 and 10 of the experiment.
2.3. Pathological evaluation
At necropsy, an estimate of gross lung involvement was assigned based on the percentage of each lung lobe affected and the percentage of total lung volume each lobe represented. Percentage of total lung volume of each lobe was estimated as 5% for the left cranial, 6% for the left middle, 29% for the left caudal, 11% for the right cranial, 10% for the right middle, 34% for the right caudal and 5% for the intermediate lung lobes (Christensen and Mousing, 1992).
Sections from the lung were taken for microscopic evaluation. All tissues were fixed in 10% neutral buffered formalin for 24 h and then placed in 90% ethanol. All sections were routinely processed and embedded in paraffin, sectioned and stained with hematoxylin and eosin.
2.4. Virus and bacterial isolation
A sample of nasal turbinate, trachea and lung from each pig was weighed and ground individually in PBS to make a 10% weight to volume suspension. The number of CFU of B. bronchiseptica/g of tissue was determined by plating serial 10-fold dilutions of homogenates on duplicate selective blood agar plates containing 20 μg/ml penicillin, 10 μg/ml amphotericin B, 10 μg/ml streptomycin and 10 μg/ml spectinomycin. In both experiments, fluid from bronchoalveolar lavage (BALF) was obtained for virus isolation, and in addition, nasal swabs collected in 1 ml of PBS, and samples of ground turbinate, ground trachea and ground lung were obtained from the second experiment for virus isolation and frozen at −80 °C. Isolation of PRCV was performed by inoculation of 100 μl of sample onto a monolayer of ST cells in a 24-well plate. The cells were examined for cytopathic effect for 1 week.
2.5. Alveolar macrophage isolation
The lungs were filled with 200 ml of sterile PBS, gently massaged, and following aspiration, approximately 100 ml of the PBS was recovered. This BALF was centrifuged at 450 × g for 15 min and alveolar macrophages (AM) were isolated by plastic adherence. BALF cells were cultured in flat-bottom, 6-well plates for 2-h in cell culture media [RPMI 1640, 2 mM l-glutamine, 1× antibiotic/antimycotic (all from Invitrogen), 25 mM HEPES, 100 μg/ml Gentamicin and 5% swine sera]. After 2-h the non-adherent cells were washed away. The adherent cell population was collected in RLT buffer and stored at −80 °C for later RNA isolation.
2.6. Cytokine gene expression
RNA was isolated from cells collected in RLT buffer (Qiagen RNeasy Mini kit). Reverse-transcription for cDNA synthesis was done using random primers and SuperScript II Reverse Transcriptase according to the manufacturer's recommendations (Invitrogen). SYBR green based real-time PCR was carried out for various mRNA targets according to manufacturer's recommendations (SYBR Green Master Mix, Applied Biosystems). Briefly, mRNA, SYBR Green Master Mix and primers (final concentration 600 nM) were mixed in a 20 μl reaction and cycled as follows: 95 °C for 15 min, 95 °C for 15 s followed by 50 °C for 1 min (45 cycles) and a final dissociation step. Levels of mRNA were calculated using the 2−ΔΔCt method, which expresses mRNA from the cells of infected pigs relative to the sham infected pigs after normalizing to beta-actin (Livak and Schmittgen, 2001). Primers are listed in Table 1 , and PCR products were less than 100 base pairs in size. Primers were generated using Primer Express, purchased from Integrated DNA Technologies (IDT), and primer pair efficiency confirmed using the method described by Livak and Schmittgen (2001).
Table 1.
Real-time PCR cytokine primers
| Gene | Accession numbers | Forward primer (5′ → 3′) | Reverse primer (5′ → 3′) |
|---|---|---|---|
| β-Actin | AJ312193 | CTCCTTCCTGGGCATGGA | CGCACTTCATGATCGAGTTGA |
| TNF-alpha | M29079 | CCACGTTGTAGCCAATGTC | CTGGGAGTAGATGAGGTACAG |
| IL-6 | AF518322 | GGCTGTGCAGATTAGTACC | CTGTGACTGCAGCTTATCC |
| MCP-1 | NM_214214 | GCTGATGAGCTACAGAAGAG | GCGATGGTCTTGAAGATCAC |
| IL-8 | X61151 | CTGGCTGTTGCCTTCTTG | CACTGGCATCGAAGTTCTG |
| IL-1βa | NM_214055 | TTGAATTCGAGTCTGCCCTGT | CCCAGGAAGACGGGCTTT |
NCBI accession numbers used to derive primers.
2.7. Statistics
Statistical analysis was done using Student's t-test assuming unequal variance and a significance level of p ≤ 0.05, comparing the mean percentage of lung affected between single and dual infected groups, the number of CFU's of B. bronchiseptica isolated from different tissues between single and dual infected groups, and the fold change in cytokine message expression between single and dual infected groups.
3. Results
3.1. Macroscopic and microscopic analysis of lesions
Clinical signs were generally mild in all infected groups, and consisted of sneezing, coughing, lethargy and pigs that were quicker to fatigue. There was a general impression that coinfected pigs were slightly more affected since clinical signs were recorded earlier and there were more of the above mentioned signs recorded for this group in both experiments.
The number of pigs/group with macroscopic lesions of pneumonia, and the mean percentage of lung affected by pneumonia for each group, for both experiments is summarized in Table 2 . Gross pneumonic lesions, irrespective of agent given, were characterized by bilateral, multifocal red consolidation which tended to have a more cranial–ventral distribution (Fig. 1 ). In the first experiment, the mean percentage of lung involvement was greatest for the group infected with both PRCV and B. bronchiseptica as compared to either agent alone. This difference was greatest and statistically significant 10 days after challenge, when the mean percentage of lung involvement was 19% for the coinfected group, but only 1.4 and 5.5% for the groups infected with B. bronchiseptica or PRCV, respectively. The mean percentage of lung affected was greater at 10 days than 4 days after challenge for all infected groups. Most pigs in all groups had gross evidence of ascarid migration in the liver.
Table 2.
Mean percentage of lung affected by pneumonia (number of pigs with lesions) in groups infected with Bordetella bronchiseptica (Bb), porcine respiratory coronavirus (PRCV), or Bb and PRCV (Bb/PRCV) on the various days post-infection
| Experiment | Day | Group |
||
|---|---|---|---|---|
| Bb | PRCV | Bb/PRCV | ||
| 1 | 4 | 0.6 (3/5) | 2.2 (5/5) | 3.6 (5/5) |
| 10 | 1.4 (2/5) | 5.5 (5/5) | 19 (5/5) | |
| 2 | 1 | 0 (0/4) | 0.25 (1/4) | 4.75 (4/4) |
| 4 | 0.25 (1/4) | 8.25 (3/4) | 6.25 (4/4) | |
| 10 | 3.0 (2/4) | 4.5 (3/4) | 12.5 (4/4) | |
Fig. 1.
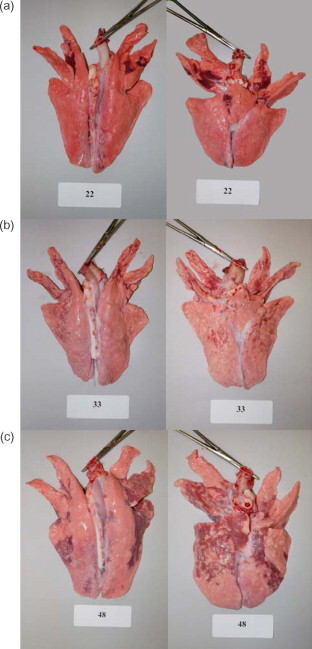
Macroscopic lesions of the lung from pigs infected with: (a) B. bronchiseptica only; (b) PRCV only or (c) coinfected with both B. bronchiseptica. Photos are of the lung of the most severely affected pig of each group 10 days post-infection in the second experiment.
Gross lesions for the second experiment were similar in character to the first experiment. The mean percentage of lung involvement for the coinfected group was again greater than the single agent infected groups at days 1 and 10 post-infection, but at day 4 the mean percentage of lung involvement was greatest for the group infected with PRCV alone. The variances were too great for any of the differences to be statistically significant, however. All 12 pigs in the coinfected group had pneumonic lesions, whereas only 7 pigs in the PRCV group and 3 pigs in the B. bronchiseptica group had gross lesions; the largest difference being at the 1-day time point. Again, lesions were more severe, in general, at 10 days post-infection, except for the group of pigs infected with PRCV alone, in which lesions peaked at 4 days post-infection.
In experiment 1, not all pigs infected with B. bronchiseptica alone had microscopic lesions. However, in the pigs with microscopic lesions, the lesions consisted of mild multifocal areas of interstitial thickening of the alveoli with mononuclear cells and occasional focal accumulations of neutrophils in the bronchi and alveoli (Fig. 2 ). Microscopic lesions of the lung of pigs infected with PRCV alone typically consisted of multifocal areas of moderate to severe interstitial thickening of alveoli with mononuclear cells and lymphocytic perivascular and peribronchiolar infiltrates. These changes tended to be more pronounced on day 10 after challenge as compared to day 4, but at day 4 there were mild infiltrates of neutrophils in some of the airways that were not present on day 10 (Fig. 2). Microscopic lesions in coinfected pigs included changes seen in pigs infected with single agents but also included additional changes and tended to be more severe than pigs infected with either agent alone. Typical microscopic lesions of the lung of pigs infected with both PRCV and B. bronchiseptica consisted of multifocal areas of moderate to severe interstitial thickening of the alveoli with mononuclear cells, lymphocytic perivascular and peribronchiolar cuffing, intra-alveolar epithelial cell necrosis with type II pneumocyte hyperplasia, and syncycial cell formation. Intra-alveolar aggregates of neutrophils, macrophages, epithelial cells and necrotic debris were also present in coinfected pigs, as well as, accumulations of neutrophils in the bronchi and bronchioles, terminal bronchiolar epithelial cell necrosis and flattening, interlobular edema and occasional hemorrhage (Fig. 2). Again, these changes tended to be more pronounced on day 10 post-infection. Three of the control animals had mild microscopic lesions consisting of peribronchial and perivascular infiltrates of lymphocytes, macrophages and eosinophils. Bronchoalveolar lavage was tested by PCR for the presence of Mycoplasma, and none was detected (data not shown).
Fig. 2.
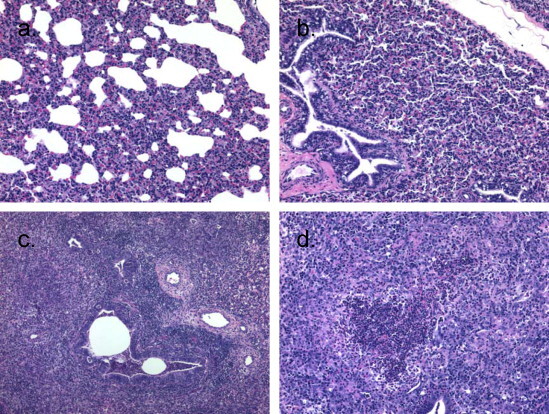
Microscopic lesions of the lungs from pigs infected with: (a) B. bronchiseptica only, showing mild areas of interstitial thickening of the alveoli with mononuclear cells; (b) PRCV only, showing areas of moderate interstitial thickening of alveoli with mononuclear cells or (c and d) coinfected with both B. bronchiseptica and PRCV, showing areas of severe interstitial thickening of the alveoli with mononuclear cells, lymphocytic perivascular and peribronchiolar cuffing, and accumulations of neutrophils in the bronchioles (c); intra-alveolar aggregates of neutrophils, macrophages, epithelial cells, and necrotic debris (d). Sections are from the lungs of pigs shown in Fig. 1.
The microscopic lesions seen in the second experiment were similar to the first experiment at days 4 and 10 post-infection except that there were fewer epithelial cell changes seen. At day 1 post-infection, the lung of 1 pig infected with PRCV and all the coinfected pigs had interstitial thickening, alveoli containing macrophages, neutrophils and fibrin, and mild peribronchiolar and perivascular lymphocytic infiltrates.
3.2. B. bronchiseptica and PRCV isolation
B. bronchiseptica was isolated from the turbinate and/or trachea of all pigs from both experiments that were challenged with the bacterium in numbers that were similar between the two groups (Table 3 ). In the first experiment, B. bronchiseptica was isolated from the lung of 8 of 10 challenged with the bacteria alone and 6 of 10 coinfected pigs (Table 3) and in similar numbers. B. bronchiseptica was isolated from the lung of more coinfected pigs than single infected pigs in the second experiment. Eight of 12 coinfected pigs had B. bronchiseptica in their lungs, whereas only 3 of 12 pigs challenged with B. bronchiseptica alone did (Table 3).
Table 3.
Number of pigs from which Bordetella bronchiseptica (geometric mean log10 titer) was isolated on the various days post-infection
| Experiment | Day | Sample | Group |
|
|---|---|---|---|---|
| Bb | Bb/PRCV | |||
| 1 | 4 | Turbinate | 4/5 (5.77) | 5/5 (4.66) |
| Trachea | 5/5 (5.73) | 5/5 (5.48) | ||
| Lung | 3/5 (4.84) | 3/5 (4.89) | ||
| 10 | Turbinate | 5/5 (6.85) | 4/5 (5.86) | |
| Trachea | 5/5 (5.21) | 5/5 (5.73) | ||
| Lung | 5/5 (6.29) | 3/5 (3.79) | ||
| 2 | 1 | Turbinate | 4/4 (5.03) | 4/4 (5.27) |
| Trachea | 4/4 (4.26) | 4/4 (3.52) | ||
| Lung | 0/4 (<2) | 2/4 (3.37) | ||
| 4 | Turbinate | 4/4 (7.26) | 4/4 (7.30) | |
| Trachea | 4/4 (7.18) | 4/4 (5.87) | ||
| Lung | 1/4 (2.83) | 3/4 (5.03) | ||
| 10 | Turbinate | 4/4 (6.99) | 4/4 (7.38) | |
| Trachea | 4/4 (6.53) | 4/4 (7.05) | ||
| Lung | 2/4 (3.94) | 3/4 (3.64) | ||
In the first experiment, PRCV was isolated from the BALF on day 4 post-infection from 9 of 10 pigs given the virus, but the virus had been cleared from all pigs euthanized on day 10 (Table 4 ). PRCV was isolated from the BALF or lung of more coinfected pigs than single infected pigs in the second experiment. Seven of 12 coinfected pigs had PRCV in their lung or BALF, whereas only 2 of 12 pigs that were challenged with PRCV alone did (Table 4). PRCV was isolated from the nasal swab, turbinate or trachea of all pigs challenged with the virus that were euthanized on days 1 and 4 post-infection, indicating that all pigs were indeed successfully infected (Table 4). Again, the virus was largely cleared by day 10 post-infection, as PRCV was not detected in any of the samples tested except the BALF from 1 coinfected pig.
Table 4.
Number of pigs from which porcine respiratory coronavirus (PRCV) was isolated on the various days post-infection
| Experiment | Day | Sample | Group |
|
|---|---|---|---|---|
| PRCV | Bb/PRCV | |||
| 1 | 4 | BAL | 5/5 | 4/5 |
| 10 | BAL | 0/5 | 0/5 | |
| 2 | 1 | Nasal swab | 2/4 | 3/4 |
| Turbinate | 2/4 | 4/4 | ||
| Trachea | 1/4 | 0/4 | ||
| Lung | 1/4 | 2/4 | ||
| BAL | 2/4 | 3/4 | ||
| 4 | Nasal swab | 4/4 | 3/4 | |
| Turbinate | 4/4 | 4/4 | ||
| Trachea | 2/4 | 0/4 | ||
| Lung | 0/4 | 2/4 | ||
| BAL | 0/4 | 3/4 | ||
| 10 | Nasal swab | 0/4 | 0/4 | |
| Turbinate | 0/4 | 0/4 | ||
| Trachea | 0/4 | 0/4 | ||
| Lung | 0/4 | 0/4 | ||
| BAL | 0/4 | 1/4 | ||
3.3. Cytokine gene expression
Cytokine production after infection likely plays a role in pathological changes observed during infection. Alveolar macrophages reside in the airway space of the lungs and are one of the first cells encountered during respiratory disease. Thus, during the second experiment we investigated changes in cytokine and chemokine mRNA levels in AM isolated from infected pigs. Fig. 3 summarizes changes in mRNA levels of proinflammatory cytokines of infected pigs relative to sham treated. In general, pigs challenged with B. bronchiseptica alone had increased mRNA levels of all the proinflammatory cytokines genes measured (TNF-alpha, IL-6, MCP-1, IL-8 and IL-1beta) at day 1 post-infection, with the majority of pigs responding with 4-fold or greater increases in transcript. B. bronchiseptica was not isolated from the lung of any of these pigs nor did any of the pigs have lesions either macroscopically or microscopically. By day 4 post-infection, cytokine mRNA levels in alveolar macrophages collected from pigs infected with B. bronchiseptica alone were near or below levels observed in uninfected animals. Fifty percent or more of the pigs had a greater than 2-fold reduction in transcript detected for IL-6, MCP-1, IL-8 and IL-1beta. The cytokine mRNA levels in B. bronchiseptica infected pigs were essentially equal to sham infected pigs by 10 days post-infection with one exception. One pig infected with B. bronchiseptica alone, which had lung lesions involving a significant portion of the lung (see Fig. 1a), had a 2-fold or greater increase in transcription of TNF-alpha, IL-6 and MCP-1.
Fig. 3.
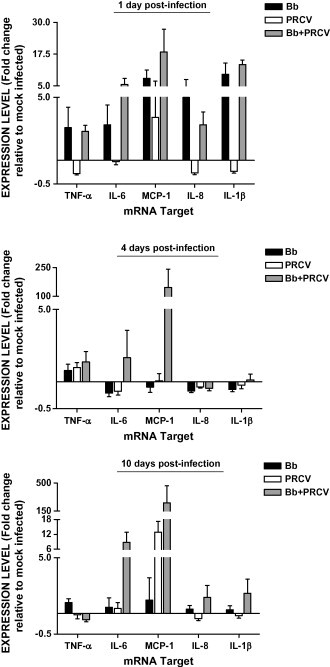
Alveolar macrophage (AM) cytokine response during infection with Bordetella bronchiseptica (Bb), porcine respiratory coronavirus (PRCV), or Bb and PRCV. AM's were isolated from infected animals, RNA isolated, and mRNA levels were assessed by real-time PCR. Data is expressed as the mean ± S.E.M. fold change in gene expression relative to mock infected animals.
With the exception of MCP-1, mRNA levels of the cytokines examined on day 1 post-infection in animals infected with PRCV alone were the same as sham infected pigs (IL-6) or were 2-fold or more below levels examined for sham infected pigs (TNF-alpha, IL-8 and IL-1beta). The one pig that had increased MCP-1 transcript levels (13-fold) in this group at this time was the pig with the microscopic lesions described above on day 1. This trend continued in the pigs infected with PRCV alone, as mRNA levels of IL-6, MCP-1, IL-8 and IL-1beta were at or below the levels measured in sham treated animals on day 4. MCP-1 mRNA levels in PRCV infected pigs showed marked elevation on day 10 post-infection with an average 12-fold increase. The level of MCP-1 was negatively correlated (R = −0.904) with the percentage of lung macroscopically affected with lesions. Three of the pigs at this time point had increases in MCP-1 transcript of 12- to 20-fold and had gross lesions affecting 0–3% of their lung, while the pig with 14% affected lung had no increase in MCP-1 transcript compared to sham treated pigs. TNF-alpha, IL-6, IL-8 and IL-1beta mRNA levels remained at levels observed in control animals in this group 10 days post-infection.
Cytokine changes observed on day 1 post-infection in coinfected pigs were similar to those observed in pigs infected with B. bronchiseptica alone. Coinfected pigs exhibited at least a 2-fold increase in mRNA levels of TNF-alpha and IL-8, a 5-fold increase of IL-6 and IL-1beta, and nearly 15-fold increase of MCP-1. Although the increase in mRNA levels were similar to that of pigs infected with B. bronchiseptica alone, the mRNA levels of TNF-alpha, IL-8, IL-6 and IL-1beta were significantly greater in coinfected pigs when compared to PRCV infected pigs. While none of the AM collected from pigs receiving PRCV or B. bronchiseptica alone showed an increase in MCP-1 mRNA levels 4 days post-infection, 3 of the 4 pigs in the coinfected group had a 55-fold or greater increase of MCP-1 mRNA, which made the coinfected group's mRNA levels of this cytokine statistically greater than for either of the single infected groups. These three pigs had the largest percentage of lung affected by macroscopic lesions, more severe microscopic lesions, and both B. bronchiseptica and PRCV were isolated from the lung, while the one pig that did not have an increase in MCP-1 transcript had the fewest lesions and no B. bronchiseptica or PRCV isolated from its lungs. At 10 days post-infection, the majority of coinfected pigs had increased mRNA levels for all the proinflammatory cytokines tested except TNF-alpha, for which all 4 pigs had a greater than 2-fold reduction of mRNA. The coinfected group had significantly less TNF-alpha transcript and significantly more MCP-1 transcript, than the group infected with B. bronchiseptica alone. The coinfected group had close to significantly greater (0.5 ≤ p ≤ 0.1) IL-6, MCP-1, IL-8 and IL-1beta transcripts, than the group infected with PRCV alone. Overall, at 10 days post-infection more coinfected pigs responded with at least a 2-fold increase of any cytokine compared to pigs infected with a single pathogen. For all timepoints, coinfected pigs exhibited enhanced mRNA levels of proinflammatory cytokines 60% of the time as compared to 23% for pigs infected with B. bronchiseptica alone and 13% for pigs infected with PRCV alone. With the exception of TNF-alpha at 10 days post-infection, coinfected pigs also had no apparent down regulation of examined cytokines.
4. Discussion
Because of its unique role in gas exchange and constant exposure to the environment, the respiratory tract is one of the most common sights for pathogen entry. Although any pathogen by itself may cause disease, it is not uncommon for multiple agents to be involved in respiratory disease. Interactions among multiple pathogens often appear to generate a more severe or chronic outcome than is observed with the individual pathogens by themselves, but little is known about the specific interactions. In both experiments performed in this study, coinfection with PRCV and B. bronchiseptica resulted in a higher percentage of the lung, on average, affected by pneumonia and an increased severity of microscopic lesions of pneumonia as compared to pigs infected with either pathogen by itself. The increased severity of gross and microscopic lesions at day 1 post-infection indicate that lesions developed earlier in coinfected pigs than in pigs infected with either agent alone. In addition, the increased severity of lesions in coinfected pigs at day 10 post-infection, a time when lesions should be resolving, suggests there was also a delay in resolution of the lesions compared to pigs infected with virus or bacteria alone. It is unlikely that lesions observed in coinfected animals were solely an additive effect, since the percentage of lung affected in the coinfected group was greater than the combined effect of the single infected groups. Furthermore, microscopic lesions in coinfected pigs were not just a combination of those induced by PRCV or B. bronchiseptica alone, as new lesions not observed in pigs infected with either pathogen alone were observed.
The isolation of both B. bronchiseptica and PRCV from the lungs of coinfected pigs was greater, as compared to singly infected pigs, in the second experiment. These results may help explain the increased severity of lesions seen in coinfected pigs. This was not the case in the first experiment, where the isolation rate was roughly the same for both single and dual infected pigs. However, there were only minor, if any, gross or microscopic lesions that accompanied the isolation of B. bronchiseptica from the pigs infected with bacteria alone in the first experiment. Although isolation of B. bronchiseptica from the turbinates and trachea is common in the age of pig used for these experiments, isolation from the lung in pigs challenged with B. bronchiseptica alone is not typical of prior experiments with this isolate (Brockmeier et al., 2000).
There was evidence of ascarid infection and migration through the liver and lung of the pigs in the first experiment. Previous studies have indicated that parasitic infections can alter the response to bacterial and viral infections. For example, aerosol exposure to P. multocida in mice with migrating Ascaris suum larvae developed more severe pneumonia than Ascaris free mice (Tjornehoj et al., 1992), and coinfection with Schistosoma mansoni and vaccinia virus resulted in a decreased antiviral Th1 response and delayed viral clearance in mice (Actor et al., 1993). A. suum, the probable cause of the lesions seen in the pigs in the first experiment, has also been shown to skew the adaptive immune response to a Th2 type response (Dawson et al., 2005). Thus, the increased isolation of B. bronchiseptica and PRCV, the appearance of mild microscopic lesions in control pigs, and the increased severity of microscopic lesions in coinfected pigs that were all seen in the first experiment as compared to the second, may be attributed to the resolving ascarid migration that was evident in these pigs. These results illustrate the difficulties in reproducing the effects of experimental combined infections in out bred conventionally raised animals such as pigs.
Alveolar macrophages are first-responders during pulmonary disease and take action against invading pathogens through the production of proinflammatory cytokines and chemokines. In addition, alveolar macrophages can recruit effector cells to the site of infection and act as antigen-presenting cells. As well as pathogen burden, another explanation for the increased disease severity observed in coinfected pigs may be, in part, the result of enhanced proinflammatory cytokine expression in alveolar macrophages. Although cytokines were only examined at the transcriptional level, and enhanced transcription does not assure the cytokines are secreted and functional, an increase in transcription of at least one proinflammatory cytokine was detected in 100% of the coinfected pigs at each of the various time points, whereas only 50% of the pigs from each of the groups infected with a single agent responded. Quantitatively, IL-6 and MCP-1 showed the largest transcriptional changes in coinfected pigs when compared to pigs infected with either agent alone.
In general, pigs infected with B. bronchiseptica alone had an increase of mRNA of all cytokines tested at day 1 post-infection, even though no lesions were seen nor was bacteria recovered from the samples of lung tested. This was likely an acute response to B. bronchiseptica LPS in the inoculum. Little increase in cytokine transcriptional response was noted after day 1 in these pigs. In fact, at day 4 there appeared to be a down regulation of most of the cytokines examined. It is likely that the bacteria were largely cleared from the lungs of these pigs and few, if any, lesions were seen. The one exception was a pig on day 10 that had significant lung lesions affecting an estimated 11% of the lung and had increased TNF-alpha, IL-6, and MCP-1 mRNA. In this regard, at days 4 and 10 post-infection, lesions correlated to increased cytokine responses. Previous research in mice shows that IL-6 and TNF-alpha are induced in bone marrow derived macrophages 24 h after exposure, as well as in lung homogenates 12 h after exposure, to B. bronchiseptica in a toll-like receptor-4 (TLR4) dependent manner (Mann et al., 2004a). Furthermore, this early TNF-alpha response appears vital for limiting the extent of bacterial growth and pneumonia, permitting recovery (Mann et al., 2004b).
Pigs infected with PRCV alone displayed a limited cytokine response, that was basically restricted to an increase in MCP-1. If anything, there appeared to largely be a down regulation of most of the cytokines examined. These results agree with those of a previous study which found no increase in the bioactive levels of TNF-alpha or IL-1 in the BALF of PRCV infected pigs up to 4 days after infection (Van Reeth et al., 1999). The results reported here are also similar to a recent report describing responses to SARS coronavirus in humans (Law et al., 2005). Human dendritic cells responded to SARS coronavirus with low expression of the antiviral cytokines (IFN-alpha, IFN-beta, IFN-gamma and IL-12) and proinflammatory cytokines (IL-6 and TNF-alpha), but large increases in CXC chemokines that preferentially act on mononuclear cells (MIP-1α, RANTES, IP-10 and MCP-1). Of these CXC chemokines, induction was strongest for MCP-1 and IP-10. Additionally, MCP-1 was also found to be elevated in SARS patients as well (Huang et al., 2005). MCP-1 is important for the recruitment and activation of macrophages and has been shown to influence viral load in an influenza model (Dawson et al., 2000). The increase in MCP-1 in 1 pig at day 1 seemed to be positively correlated with microscopic lesions seen in that pig, while at 10 days post-infection the pig with the largest percentage of lung affected had no increase in MCP-1, and the other 3 pigs all had significant increases in this cytokine and little to no lung lesions. Thus, lesions at 10 days post-infection seemed to be inversely correlated with the level of MCP-1 produced.
On day 1 post-infection, the cytokine response in coinfected pigs was most like that of pigs infected with B. bronchiseptica alone, in that there was a general increase in the transcriptional response to all cytokines tested. Again, this may largely be due to the response to bacterial LPS. However, unlike the pigs that were challenged with bacteria alone, bacteria were isolated from some of the coinfected pigs and lesions were seen in all of the coinfected pigs as compared to pigs infected with bacteria or virus alone. The coinfected pigs responded with an increase to more cytokines per pig and quantitatively greater, on average, to IL-6 and MCP-1, than did pigs infected with the bacteria alone. This might explain the increase in the severity of lesions observed early in the coinfected pigs compared to the pigs infected with bacteria or virus alone. Whether or not this early cytokine response in coinfected pigs, which was not typical of pigs infected with the virus alone, might have altered the host response to PRCV resulting in the decreased clearance of the virus observed in the second experiment, warrants further investigation.
The increased transcriptional response of MCP-1 on day 4, and IL-6, MCP-1, IL-8 and IL-1beta on day 10 post-infection in coinfected pigs demonstrates an enhanced response of proinflammatory cytokines compared to what is seen with either of the pathogens alone. A previously proposed hypothesis suggested that proinflammatory cytokines, particularly TNF-alpha, produced during exposure to PRCV and bacterial LPS might be responsible for more severe clinical respiratory signs in pigs (Van Reeth et al., 2000). Although the results reported here do not point specifically to TNF-alpha, the lung lesions in coinfected pigs support this hypothesis. In another study, exposing human macrophages to SARS coronavirus and subsequent suboptimal LPS led to a dramatic increase in IL-6 over what was observed with cells exposed to either virus or LPS alone (Tseng et al., 2005). Overall, the data presented here does not implicate a single cytokine or chemokine in causing more severe lung disease in coinfected animals than in PRCV or B. bronchiseptica infection alone.
Ultimately, the early interaction of pathogens with the innate immune system and its subsequent influence on the adaptive immune response is critical in determining the outcome of infection. The host has evolved specific mechanisms to deal with diverse classes of pathogens in order to induce the most effective response. In addition to the innate mechanisms, such as epithelial barriers, muco-ciliary clearance, and antimicrobial substances, that are ubiquitously and constitutively present, innate responses induced through TLR receptors, which recognize conserved molecular patterns present in microorganisms, can stimulate specific responses to different pathogens. For example, neutrophils usually dominate the initial inflammatory infiltrate subsequent to bacterial infections, while viral infection is often characterized by primarily mononuclear infiltration. In this study, the cytokine response in coinfected pigs was unique compared to the cytokine response in pigs infected with either agent by itself, suggesting that the alteration in the innate response, which has developed over time to deal with these pathogens, may be one mechanism through which coinfection alters the disease outcome.
Acknowledgements
The authors wish to thank William Mengeling and Ann Vorwald for providing the PRCV isolate and Kim Driftmier, Theresa Waters, Michael Mullins and Don Hackbarth for their excellent technical assistance.
References
- Actor J.K., Shirai M., Kullberg M.C., Buller R.L., Sher A., Berzofsky J.A. Helminth infection results in decreased virus-specific CD8+ cytotoxic T-cell and Th1 cytokine responses as well as delayed virus clearance. Proc. Natl. Acad. Sci. 1993;90:948–952. doi: 10.1073/pnas.90.3.948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Britton P., Mawditt K.L., Page K.W. The cloning and sequencing of the virion protein genes from a British isolate of porcine respiratory coronavirus: comparison with transmissible gastroenteritis virus genes. Virus Res. 1991;21:181–198. doi: 10.1016/0168-1702(91)90032-Q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brockmeier S.L. Prior infection with Bordetella bronchiseptica increases colonization with Haemophilus parasuis in swine. Vet. Microbiol. 2004;99:75–78. doi: 10.1016/j.vetmic.2003.08.013. [DOI] [PubMed] [Google Scholar]
- Brockmeier S.L., Palmer M.V., Bolin S.R. Effects of intranasal inoculation of porcine reproductive and respiratory syndrome virus, Bordetella bronchiseptica, alone or in combination, in pigs. Am. J. Vet. Res. 2000;61:892–899. doi: 10.2460/ajvr.2000.61.892. [DOI] [PubMed] [Google Scholar]
- Brockmeier S.L., Palmer M.V., Bolin S.R., Rimler R.B. Effects of intranasal inoculation with Bordetella bronchiseptica, porcine reproductive and respiratory syndrome virus, or a combination of both organisms on subsequent infection with Pasteurella multocida in pigs. Am. J. Vet. Res. 2001;62:521–525. doi: 10.2460/ajvr.2001.62.521. [DOI] [PubMed] [Google Scholar]
- Chanter N., Magyar T., Rutter J.M. Interactions between Bordetella bronchiseptica and toxigenic Pasteurella multocida in atrophic rhinitis of pigs. Res. Vet. Sci. 1989;47:48–53. [PubMed] [Google Scholar]
- Christensen G., Mousing J. In: Diseases of Swine. seventh ed. Leman A.D., Straw B.E., Mengeling W.L., editors. Iowa State University Press; Ames, Iowa: 1992. Respiratory system; pp. 139–162. [Google Scholar]
- Dawson T.C., Beck M.A., Kuziel W.A., Henderson F., Maeda N. Contrasting effects of CCR5 and CCR2 deficiency in the pulmonary inflammatory response to influenza A virus. Am. J. Pathol. 2000;156:1951–1959. doi: 10.1016/S0002-9440(10)65068-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson H.D., Beshah E., Nishi S., Solano-Aguilar G., Morimoto M., Zhau A.K., Madden K.B., Ledbetter T.K., Dubey J.P., Shea-Donohue T., Lunney J.K., Urban J.F. Localized multigene expression patterns support an evolving Th1/Th2-like paradigm in response to infections with Toxoplasma gondii and Ascaris suum. Infect. Immun. 2005;73:1116–1128. doi: 10.1128/IAI.73.2.1116-1128.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J.-L., Huang J., Duan Z.-H., Wei J., Min J., Luo X.-H., Li J.-G., Tan W.-P., Wu L.-Z., Liu R.-Y., Li Y., Shao J., Huang B.-J., Zeng Y.-X., Huang W. Th2 predominance and CD8+ memory T cell depletion in patients with severe acute respiratory syndrome. Microbes Infect. 2005;7:427–436. doi: 10.1016/j.micinf.2004.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law H.K.W., Cheung C.Y., Ng H.Y., Sia S.F., Chan Y.O., Luk W., Nicholls J.M., Malik Peiris J.S., Lau Y.L. Chemokine up-regulation in SARS-coronavirus-infected, monocyte-derived human dendritic cells. Blood. 2005;106:2366–2374. doi: 10.1182/blood-2004-10-4166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livak K.J., Schmittgen T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Mann P.B., Kennett M.J., Harvill E.T. Toll-like receptor 4 is critical to innate host defense in a murine model of Bordetellosis. J. Infect. Dis. 2004;189:833–836. doi: 10.1086/381898. [DOI] [PubMed] [Google Scholar]
- Mann P.B., Elder K.D., Kennett M.J., Harvill E.T. Toll-like receptor 4-dependent early elicited tumor necrosis factor alpha expression is critical for innate host defense against Bordetella bronchiseptica. Infect. Immun. 2004;72:6650–6658. doi: 10.1128/IAI.72.11.6650-6658.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Royaee A.R., Husmann R.J., Dawson H.D., Calzada-Nova G., Schnitzlein W.M., Zuckermann F.A., Lunney J.K. Deciphering the involvement of innate immune factors in the development of the host response to PRRSV vaccination. Vet. Immunol. Immunopathol. 2004;102:199–216. doi: 10.1016/j.vetimm.2004.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasschaert D., Duarte M., Laude H. Porcine respiratory coronavirus differs from transmissible gastroenteritis virus by a few genomic deletions. J. Gen. Virol. 1990;71:2599–2607. doi: 10.1099/0022-1317-71-11-2599. [DOI] [PubMed] [Google Scholar]
- Tjornehoj K., Eriksen L., Aalbaek B., Nansen P. Interaction between Ascaris suum and Pasteurella multocida in the lungs of mice. Parasitol. Res. 1992;78:525–528. doi: 10.1007/BF00931575. [DOI] [PubMed] [Google Scholar]
- Tseng C-T.K., Perrone L.A., Zhu H., Makino S., Peters C.J. Severe acute respiratory syndrome and the innate immune responses: modulation of effector cell function without productive infection. J. immunol. 2005;174:7977–7985. doi: 10.4049/jimmunol.174.12.7977. [DOI] [PubMed] [Google Scholar]
- Van Reeth K., Pensaert M.B. Porcine respiratory coronavirus-mediated interference against influenza virus replication in the respiratory tract of feeder pigs. Am. J. Vet. Res. 1994;55:1275–1281. [PubMed] [Google Scholar]
- Van Reeth K., Nauwynck H. Proinflammatory cytokines and viral respiratory disease in pigs. Vet. Res. 2000;31:187–213. doi: 10.1051/vetres:2000113. [DOI] [PubMed] [Google Scholar]
- Van Reeth K., Labarque G., Nauwynck H., Pensaert M. Differential production of proinflammatory cytokines in the pig lung during different respiratory virus infections: correlations with pathogenicity. Res. Vet. Sci. 1999;67:47–52. doi: 10.1053/rvsc.1998.0277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Reeth K., Nauwynck H., Pensaert M. A potential role for tumour necrosis factor-alpha in synergy between porcine respiratory coronavirus and bacterial lipopolysaccharide in the induction of respiratory disease in pigs. J. Med. Microbiol. 2000;49:613–620. doi: 10.1099/0022-1317-49-7-613. [DOI] [PubMed] [Google Scholar]
- Vecht U., Wisselink H.J., van Dijk J.E., Smith H.E. Virulence of Streptococcus suis type 2 strains in newborn germfree pigs depends on phenotype. Infect. Immun. 1992;60:550–556. doi: 10.1128/iai.60.2.550-556.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wesley R.D., Woods R.D., Cheung A.K. Genetic analysis of porcine respiratory coronavirus, an attenuated variant of transmissible gastroenteritis virus. J. Virol. 1991;65:3369–3373. doi: 10.1128/jvi.65.6.3369-3373.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]


