Gastrointestinal (GI) disease is common in pet ferrets. According to Delaney, unique ferret anatomy and physiology, including rapid GI transit time, and short, simple GI tract predisposes to GI disease [1]. Etiologies include bacteria, including helicobacter infections, virus, parasites, inflammatory conditions, neoplasia, foreign body-related diseases, and stress. Some practitioners report improvement in some refractory cases with diet modification, which suggests dietary allergen or intolerance. A thorough workup is critical for distinction between etiologies typically producing similar clinical signs and symptoms [1], [2].
Diarrhea is one of the most common presenting complaints, and can represent a myriad of underlying and multifactorial causes ( Fig. 1). Table 1 includes common and uncommon causes of diarrhea in pet ferrets. History can be helpful in determining underlying cause. Ferrets under 1 year of age are more prone to foreign body ingestion, coccida and proliferative colitis, while coronavirus and Helicobacter mustelae typically affect older ferrets [1], [2].
Fig. 1.

Various forms of diarrhea in the ferret.
Table 1.
Diseases producing intermittent or chronic diarrhea in ferrets
| Disease | Diagnosis | Treatment |
|---|---|---|
| Bacterial, primary or secondary | Culture and sensitivity | Appropriate antimicrobial therapy, preferably based on culture and sensitivity |
| Helicobacter | Helicobacter PCR, histopathology | |
| Lawsoni/Desulfovibrio | Biopsy and histopathology | |
| Campylobacter jejuni | Culture difficult | |
| Bacterial, uncommon Mycobacteriosis | Histopathology PCR | Appropriate antimicrobial therapy; treatment for mycobacteriosis is controversial due to potential zoonosis |
| Viral; | Coronavirus isolation | Supportive care |
| Ferret Enteric coronavirus (FEVC) | PCR | |
| Rotavirus canine distemper virus | PCR | |
| PCR | ||
| Coccidiosis | Fecal floatation, direct smear | Anticoccidial drugs |
| Giardiasis | ||
| Helicobacter mustelae | PCR—gastric swab Histopathology—gastric | Specific Helicobacter therapy, usually triple drug therapy; traditional therapy includes metronidazole, amoxicillin and bismuth subsalicylate; other drugs including proton-pump inhibitors have been utilized |
| Histopathology—gastric | ||
| “Inflammatory bowel disease” | Histopathology | Some suggest anti-inflammatory drugs; caution in ferrets with possible Helicobacter or underlying bacterial or viral disease |
| Gastrointestinal neoplasia | Histopathology | Surgical excision |
| Chemotherapy | ||
| Foreign body ingestion | PE, radiographs, exploratory surgery | Surgery |
| Stress—medical or psychologic | History | Correction of underlying medical disorder or psychological stress |
| Detection of underlying medical condition | ||
| Idiopathic megaesophagus | Radiology | Unrewarding |
Diarrhea occurring soon after introduction of a new ferret into the household suggests an infectious etiology, such as coronavirus, coccidia, or possibly giardia. Infectious agents such as Lawsonia and Mycobacteria, however, tend to affect only a few members of a group [1], [2].
Common causes of gastrointestinal disease in ferrets
Foreign body ingestion
Ingestion of foreign material, in particular objects made of rubber or rubber-like materials, is common in ferrets under 2 years of age. Symptoms vary depending on volume of material ingested and presence of complete or partial blockage. The most common symptoms are sudden onset of anorexia, vomiting, nausea, and lethargy, which is sometimes accompanied by stress-related diarrhea. Palpation often reveals gastric distention, GI pain, or in many cases, presence of the foreign material itself. Laboratory analysis is nonspecific, but may reflect dehydration, stress, and starvation in chronic cases. Plain radiography often reveals characteristic GI gas patterns typical of obstruction in other small animals. In some cases, definitive diagnosis is made at exploratory surgery [2].
It should be noted that the author has discovered foreign material within the stomach of ferrets undergoing abdominal surgery for reasons unrelated to GI disease. Therefore, any exploratory surgery in a ferret should include careful examination and palpation of the stomach and intestinal tract, and collection of appropriate biopsy samples.
Bacterial diseases
Ferrets are prone to both primary and secondary bacterial infections. Secondary bacterial overgrowth is not uncommon in ferrets with other diseases affecting the GI tract. Ferrets can be exposed to Salmonella and Campylobacter from ingestion of raw meat. Escherichia coli has been cultured from the feces of ferrets with diarrhea [2].
H mustelae is a common inhabitant of the ferret stomach, and some authors suggest prevalence in pet ferrets to be 100%. Helicobacter apparently does not cause disease in all ferrets, but is linked to severe lymphocytic plasmacytic inflammatory gastric lesions, diarrhea, anemia, and chronic wasting. Diarrhea can be black and tarry when Helicobacter-associated ulceration is present ( Fig. 2). Helicobacter can be difficult to completely eradicate. As reinfection is common, the organism must be eliminated from all contact animals as well [3], [4]. Gastric biopsies of ferrets that have undergone Helicobacter treatment can reveal healing ulcers, fibrosis, and other evidence of gastric damage that may explain why treatment of Helicobacter does not always result in complete resolution of clinical signs (D. Reavill and R. Schmidt, personal communication, Zoo and Exotic Pathology Service, West Sacramento, CA).
Fig. 2.

Melena in a ferret with confirmed gastric ulceration.
Recent work in humans and laboratory animal models indicates that Helicobacter is not confined to the stomach and duodenum, and may play a role in inflammatory disease along the entire GI tract. In mice, inflammatory bowel disease is directly linked to the presence of certain subspecies of Helicobacter. It is currently unknown whether this link exists in the ferret, but must be considered [5], [6], [7].
Proliferative bowel disease is thought to be caused by an organism similar to that causing proliferative ileitis of pigs and hamsters. Older literature refers to the causative agent as a Campylobacter-like organism [2]. However, experimental infection with Campylobacter jejunum failed to produce lesions of proliferative bowel disease [8]. The organism was later tentatively identified as Lawsonia intracellularis. However, a more recent study showed that lesions typical of proliferative bowel disease could be produced by coinfection of ferrets with an intracellular Desulfovirbio species and coccidia [9]. Typical symptoms include chronic diarrhea with or without blood or mucus, tenesmus, and possible rectal prolapse. Weight loss and dehydration typically follow [2].
Campylobacter jejuni does occasionally cause diarrhea in ferrets younger than 6 months of age. It is usually self-limiting, and experimental studies show the disease to typically be asymptomatic. A more serious concern, however, is the potential zoonotic risk of Campylobacter species [2].
Mycobacteria have been isolated from the GI tract of ferrets, including granulomatous enteritis caused by M avium, and a disseminated gastric lesion produced by M celatum (type 3). Granulomatous enteritis can produce vomiting, anorexia, and diarrhea in the affected ferret [10], [11].
Diagnosis of bacterial diseases is often not straightforward ( Table 2). Many of the organisms mentioned above are not readily identified using standard culture and sensitivity methods.
Table 2.
Diagnosis of bacteria producing gastrointestinal diseases in ferrets
| Bacterial organism | Definitive diagnosis |
|---|---|
| Salmonella sp, E coli, Enterobacter sp, other | Standard aerobic culture and sensitivity; consider anaerobic culture as well |
| Helicobacter mustelae | Culture difficult |
| Polymerase chain reaction (PCR) gastric mucosal, colonic, and mouth swabs | |
| Histopathology of gastric samples | |
| Lawsonia intracellularis (Desulfovibrio spp) | Histopathology of colonic biopsy samples |
| Campylobacter jejuni | Special culture and sensitivity requirements; contact diagnostic laboratory |
| Dark-field microscopy of fresh feces to observe motility characteristics of organism | |
| Mycobacterium spp | Biopsy of gastrointestinal lesions |
| Acid-fast staining of suspected lesions | |
| PCR of suspected lesions |
Data from Lennox AM. Working up mystery anemia in ferrets. Exotic DVM 2004;6(3):22–6; and Williams BH. Ferret microbiology and virology. In: Fudge A, editor. Laboratory medicine, avian and exotic pets. Philadelphia: WB Saunders; 2000.
Viral diseases
Viruses producing GI disease in ferrets include coronavirus, rotavirus, and canine distemper virus [12]. It should be kept in mind that any viral disease that does not primarily affect the GI tract may produce diarrhea secondary to stress and general debilitation.
Rotavirus has been documented in several large ferret facilities worldwide, and typically affects animals 4 to 6 weeks of age. Morbidity is high in this age group, and low in adult ferrets. Younger ferrets may die of secondary complications including dehydration. Rotavirus does not appear to be a commonly diagnosed disease of ferrets in the pet population but must be considered a potential diagnosis in very young animals [2]. Polymerase chain reaction (PCR) testing is currently available to detect Rotavirus in suspect ferrets (M. Kuipel and R. Maes, personal communication, Michigan State University, Department of Pathobiology and Diagnostic Investigation; kiupel@dcpah.musu.edu).
Canine distemper virus can produce diarrhea in affected ferrets, along with more typical symptoms including ocular and nasal discharge, characteristic skin rash, and so-called “hard pad” [2]. PCR, immunohistochemistry, and fluorescent antibody staining, as well as virus isolation are commercially available for the postmortem diagnosis of canine distemper virus (M. Kuipel and R. Maes, personal communication, Michigan State University, Department of Pathobiology and Diagnostic Investigation; kiupel@dcpah.musu.edu) [13]. PCR product can also be sequenced to differentiate field strains from vaccine strains [13]. Canine distemper virus can also be identified through immunohistochemistry of ante-mortem intestinal biopsy specimens.
In 1993, breeders and veterinarians recognized an outbreak of diarrhea later termed epizootic catarrhal enteritis (ECE). Typical presentation included profuse green diarrhea with excessive mucus. Although morbidity was typically high, mortality was generally low with supportive care. Older ferrets with a concurrent disease were often most severely affected. Typical history included an onset of diarrhea in existing pets shortly after introduction of new, apparently normal, ferrets into the household. Incubation period is typically 48 to 72 hours. Older ferrets that recover from ECE often developed chronic diarrhea and wasting [14].
Coronavirus particles were identified by transmission electron microscopy in feces and jejunal specimens, and by immunohistochemical staining of jejunal sections. Histopathologic lesions of intestine also were typical of coronaviral infection [14]. Recent work has now classified the virus as a novel ferret coronavirus, and suggested the name ferret enteric coronavirus (FECV). PCR testing is now commercially available, and has been shown to reliably identify FECV in the feces, saliva, and intestinal biopsy specimens of infected ferrets [15].
Inflammatory gastrointestinal disease
By definition, inflammatory GI disease is the accumulation of inflammatory cells within samples of the GI system or organs. Classification largely depends on the types and proportions of cells identified, and location of the inflammatory process. Some pathologists classify lymphocytic/plasmacytic inflammation of the intestine as “inflammatory bowel disease.” The presence of moderate to large numbers of eosinophils may warrant a histologic diagnosis of “eosinophilic gastroenteritis” [2]. It is uncertain whether or not specific etiologies produce specific patterns of inflammatory infiltrate allowing definitive diagnosis. Several authors believe eosinophilic inflammatory infiltrate to be more indicative of an allergic response, perhaps to dietary antigen [2], [5]. Much work remains to be done on the etiology and pathogenesis of inflammatory GI disease in ferrets.
Of 135 cases of ferret GI organ and lymph node biopsies submitted to an exotic animal pathology laboratory, 92% reported mild to severe inflammation (Zoo and Exotic Pathology Service, Drs. Reavill and Schmidt, personal communication). Of these submissions, 73% reported symptoms related primarily to the GI tract, such as vomiting and diarrhea. The rest reported less specific symptoms such as weight loss and anorexia, or no symptoms recognized at all. In the latter cases, samples were most often collected during exploratory surgery to investigate adrenal disease, or other non-GI indication. Severity of the lesion did not always correlate with severity of clinical symptoms. Spiral organisms consistent with Helicobacter sp. were identified in gastric samples with varying types and severities of inflammatory infiltration [5]. It has been speculated by Johnson-Delaney that ferrets may have aberrations with one or more tumor suppressor genes. The ferret's response to infection with Helicobacter may be further explained through examination of the genome and enteric immune responses. Research is currently underway to explore this possibility.
Neoplasia
GI neoplasia commonly presents as vague GI symptoms. The author has seen disseminated intestinal lymphoma on several occasions, and symptoms have ranged from acute death due to intestinal perforation without previous symptoms, to chronic or intermittent diarrhea ( Fig. 3).
Fig. 3.
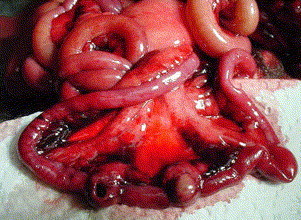
Intestinal lymphoma and bowel perforation. This ferret had experienced mild intermittent diarrhea for several weeks before presenting with acute depression and anorexia.
GI neoplasia has been linked to Helicobacter sp. infections in both humans and ferrets, and includes lymphoma, adenocarcinoma, and recently colonic adenomas in humans [3], [4], [16]. Recent cases of pyloric adenocarcinoma in ferrets presented as intermittent vomiting, and most advanced cases have palpably enlarged stomachs [17], [18]. A case of adenocarcinoma of the colon presented as rectal prolapse [5].
Changes suggesting lymphoid neoplasia are commonly reported in enlarged gastric and mesenteric lymph nodes submitted for histopathology. In some cases differentiation of reactive versus neoplastic lymph nodes is difficult.
Miscellaneous
Megaesophagus has been diagnosed in ferrets. Clinical signs include difficulty swallowing, decreased appetite, and regurgitation. Treatment included fluids, antibiotics and agents directed against possible primary causes of megaesophagus, and were reported as ineffective. Nine ferrets ultimately submitted for necropsy and histopathology had no etiology determined for megaesophagus [19].
Cryptococcus has been reported in the literature as a cause of GI disease in a ferret. The organism was identified in a section of intestine of a ferret that exhibited vague GI symptoms [20].
Diagnosis of gastrointestinal disease
Clinical pathology
Complete blood count (CBC) and chemistry panel can provide clues to diagnosis and help evaluate overall patient condition. In many cases biochemical changes simply reflect patient dehydration, anorexia, and wasting. Anemia can occur from chronic inflammation and blood loss Table 3 lists typical complete blood count and biochemistry testing changes expected with typical GI diseases of ferrets [21].
Table 3.
Ferret gastrointestinal diseases and common clinical pathologic changes
| Disease | Clinical pathology change |
|---|---|
| Bacterial or viral enteritis | Changes depend on chronicity of disease: also commonly see elevated ALP, AST, and ALT. |
| Changes can also reflect anorexia, starvation, and dehydration. | |
| Gastric and/or intestinal foreign body | Changes typically reflect anorexia, starvation, stress, and dehydration: leukocytosis, neutrophilia, lymphocytosis. |
| Elevated hematocrit and BUN may reflect dehydration | |
| Gastritis | Same as above |
| Gastric ulceration | Same as above: anemia is common, positive occult fecal blood test |
| Proliferative bowel disease | Changes depend on chronicity of disease but often reflect dehydration, possible anemia, and hypoalbuminemia |
| Eosinophilic gastroenteritis | Can see marked eosinophilia, as high as 35%. Changes can also reflect anorexia, starvation, and dehydration |
| Megaesophagus | Changes reflect anorexia, starvation, and dehydration, depending on chronicity of disease. |
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Modified from Jenkins JR. Rabbit and ferret liver and gastrointestinal testing. In: Fudge A, editor. Laboratory medicine, avian and exotic pets. Philadelphia: WB Saunders; 2000; with permission.
Culture and sensitivity can help identify primary or secondary bacterial pathogens. H mustelae is a common pathogen of ferrets, but cannot be identified on culture. Biopsy and histopathology of gastric samples, or PCR of gastric, colonic, or mouth swabs provide more specific diagnosis. As H mustelae is thought to be present in nearly all ferrets, and does not always produce disease, the most useful application of PCR is to monitor response to therapy and detect reinfection after successful therapy [4].
Radiography
Radiography is useful in the diagnosis of GI disease. Abnormal gas patterns can suggest foreign body obstruction or enteritis.
Contrast media have been used to evaluate the GI tract. A recent study on results of the use of barium sulfate, 8 to 13 mL/kg, indicated that small intestinal transit time was less than 2 hours. Visualization of the barium-filled small bowel was optimal at 20 and 40 minutes postbarium administration. Small bowel loops in these normal ferrets did not exceed 5 to 7 mm in width. The use of ketamine and diazepam to facilitate radiography did not significantly slow GI transit time in these ferrets [22].
Ultrasound
Abdominal ultrasonography can be an extremely effective diagnostic tool for evaluation of abdominal organs in dogs, cats, and other mammals. Information on organ size, shape, density, and even GI motility can be beneficial. The most common reported usage in the pet ferret is for evaluation of adrenal glands in ferrets with suspected adrenal neoplasia, but GI ultrasound should provide similar benefits to those reported in canine and feline practice.
Endoscopy
Rigid or flexible endoscopy can aid diagnosis of GI disease in dogs and cats. Few reports exist documenting its usefulness in ferrets, but can be expected to provide similar benefits. The author has reported use of a semiflexible 1.2-mm endoscope with air insufflation to document gastric ulceration in a ferret [4].
Laparoscopy has been described in the ferret. The technique is relatively simple, but primary use is for visualization and biopsy of those organs available for collection via simple clamshell or punch-style biopsy forceps or True-cut needles, such as liver, pancreas, and kidney [23]. Full-thickness GI biopsies in the ferret must be obtained via traditional laparotomy.
Gastrointestinal biopsy
Biopsy of GI tract remains the most specific test for confirmation of etiology, especially in those cases refractory to treatment. Diagnosis of GI neoplasia, inflammatory GI disease, and disease secondary to unusual forms of bacteria is extremely difficult without biopsy. Biopsy and histopathology are extremely useful for monitoring response to treatment. ECE virus and canine distemper virus can also be detected in biopsy samples of the small intestine through immunohistochemistry [12], [13].
In human medicine, GI biopsy remains the gold standard for diagnosis of Helicobacter-related gastroenteritis, as simple presence of the organism does not always correlate well with disease [24].
Gastric or colonic mucosal biopsy samples can be collected via semiflexible or flexible endoscopy. Surgical biopsy during laparotomy, however, gives the added advantage of allowing visualization and targeting of grossly abnormal sections, and collection of full-thickness samples.
Miscellaneous testing
Some GI diseases such as Helicobacter-related gastric ulceration produce hematochezia. Fecal occult blood testing can help confirm the presence of blood. As some diseases produce only intermittent bleeding, a single negative occult fecal blood test does not rule out GI hemorrhage [3], [4].
The Hemoccult Fecal Occult Blood Test (Beckman Coulter, www.coulter.com) has been evaluated in clinical practice for use in ferrets. Elimination of dietary heme must precede testing, and can be accomplished by feeding a nonheme-containing diet. Johnson-Delaney and Lennox demonstrated success with the use of two carnivore critical care diets (Quantum Series Enteral Carnivore Diet, Walkabout Farms, Pembroke, Virginia, www.herpnutrition.com; Oxbow Pet Products, Murdock, Nebraska, www.oxbowhay.com) [1], [4].
Treatment of gastrointestinal disease
Treatment of GI disease is ideally based upon a thorough workup and a confirmed diagnosis, or at least a tentative diagnosis based on physical exam findings, and laboratory analysis. It should be noted that treatments suggested for one GI disease might exacerbate another condition. For example, drugs suggested for inflammatory bowel disease may be contraindicated when active viral or bacterial agents are present. Table 4 lists drugs commonly used to treat GI disease in ferrets.
Table 4.
Formulary of drugs used to treat gastrointestinal disease in ferrets
| Drug and dosage | Indication | Comments |
|---|---|---|
| Amoxicillin: 20 mg/kg bid × 28 days | Helicobacter “triple therapy” | Clinical trials on ferrets proven effective |
| Metronidazole: 30 mg/kg bid × 28 days | ||
| Pepto Bismal: 15 mg/kg bid × 28 days | ||
| Loperamide hydrochloride 0.2 mg/kg q12h | Supportive care | Often ineffective |
| Azathioprine: 0.9 mg/kg po q72ha | Inflammatory bowel disease | Immune suppressor; contraindicated in cases of infectious disease; monitor CBC/Chem during therapy |
| Enrofloxacin 5–10 mg/kg po bid | Susceptible microorganisms | |
| Sulfadimethoxine 50 mg/kg po, then 25 mg/kg q24h × 9 days | Coccidia | |
| Amprolium 19 mg/kg po q24h | Coccidia | |
| Metronidazole 15–20 mg/kg po q12h × 14 days | Gastrointestinal protozoa | |
| Metoclopramide 0.2–1.0 mg/kg q6–8h po, sq, im | Antiemetic, motility enhancer | |
| Cimetidine 5–10 mg/kg po sq im q8h | H2 blocker; inhibits acid secretion; GI ulcers | Give iv slowly |
| Famotidine 0.25–0.50 mg/kg po iv q24h | Inhibits acid secretion; GI ulcers | |
| Sucralfate 25 mg/kg po q8h | Gastrointestinal ulcers |
Modified from Carpenter JW, Mishima TY, et al. Exotic animal formulary. 2nd edition. Philadelphia: WB Saunders; 2001; with permission.
Data from Burgess M, Garner M. Clinical aspects of inflammatory bowel disease in ferrets. Exotic DVM 2002;4(2):29–33.
Supportive care includes replacing fluid needs, and ensuring adequate nutrition to prevent weight loss and loss of muscle mass that often accompanies chronic diarrhea. A number of hand-feeding products have been suggested, including high-protein commercial foods such as Oxbow Carnivore Critical Care (Oxbow Pet Products, Murdock, Nebraska, www.oxbowhay.com), Hill's a/d, strained turkey, or chicken baby food.
Antidiarrheal drugs such as loperamide hydrocloride are sometimes helpful in slowing diarrhea as an adjunct to treatment of the primary underlying cause [25].
A number of protocols have been suggested for treatment of neoplasia in ferrets, in particular GI lymphoma [25], [26]. It should be noted that in humans, there is strong evidence that treatment to eradicate Helicobacter pylori alone may cure low-grade gastric lymphoma [27]. Therefore, clinicians treating GI neoplasia, especially those tentatively linked with Helicobacter sp., should consider therapy targeting at this organism as well.
GI disease is a common clinical manifestation in pet ferrets. Numerous disease conditions can produce primary GI disease, or secondary disease due to debilitation and stress. A careful and thorough workup is necessary to determine etiology and to help develop an optimal therapeutic plan.
References
- 1.Johnson-Delaney C.A. A clinician's perspective on ferret diarrhea. Exotic DVM. 2004;6(3):27–28. [Google Scholar]
- 2.Hoefer H, Bell J. Gastrointestinal diseases. In: Quesenberry K, Carpenter J, editors. Ferrets, rabbits and rodents, clinical medicine and surgery. 2nd edition. St. Louis (MO): Saunders.
- 3.Fox J.G., Marini R.P. Helicobacter mustelae infection in ferrets: pathogenesis, epizootiology, diagnosis and treatment. Sem Avian Exotic Pet Med. 2001;10:36–42. [Google Scholar]
- 4.Lennox A.M. Working up mystery anemia in ferrets. Exotic DVM. 2004;6(3):22–26. [Google Scholar]
- 5.Cardonna L., Armati L. Enteric bacteria, lipopolysaccharides and related cytokines in inflammatory bowel disease: biological and clinical significance. J Endotoxin Res. 2000;6(3):205–214. [PubMed] [Google Scholar]
- 6.Franklin C.L., Riley L.K., Livingston R.S. Enteric lesions in SCID mice infected with “Helicobacter typlonicus,” a novel urease-negative Helicobacter species. Lab Anim Sci. 1999;49(5):496–505. [PubMed] [Google Scholar]
- 7.Saunders K.E., Shen Z. Novel intestinal Helicobacter species isolated from cotton-top tamarins (Saguinus oedipus) with chronic colitis. J Clin Microbiol. 1999;37(1):146–151. doi: 10.1128/jcm.37.1.146-151.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bell J.A., Manning D. Evaluation of Campylobacter jejuni colonization of the domestic ferret intestine as a model of proliferative colitis. Am J Vet Res. 1991;52(6):826. [PubMed] [Google Scholar]
- 9.Li X., Pang J., Fox J.G. Coinfection with intracellular Desulfovibrio species and coccidia in ferrets with proliferative bowel disease. Lab Anim Sci. 1996;16(5):569–571. [PubMed] [Google Scholar]
- 10.Valheim M., Djonne B., Heiene R. Disseminated Mycobacterium celatum (type 3) infection in a domestic ferret (Mustela putorius furo) Vet Pathol. 2001;38(4):460–463. doi: 10.1354/vp.38-4-460. [DOI] [PubMed] [Google Scholar]
- 11.Schultheiss P.C., Dolginow S.Z. Granulomatous enteritis caused by Mycobacterium avium in a ferret. J Am Vet Med Assoc. 1994;204(8):1217–1218. [PubMed] [Google Scholar]
- 12.Williams B.H. Ferret microbiology and virology. In: Fudge A., editor. Laboratory medicine, avian and exotic pets. WB Saunders; Philadelphia: 2000. [Google Scholar]
- 13.Ramos-Vara J.A., Kiupel M. Diagnostic immunohistochemistry of infectious diseases in dogs and cats. J Histotech. 2002;25:201–214. [Google Scholar]
- 14.Williams B., Kiupel M., West K.H. Coronavirus-associated epizootic catarrhal enteritis in ferrets. JAVMA. 2000;217(4):526–530. doi: 10.2460/javma.2000.217.526. [DOI] [PubMed] [Google Scholar]
- 15.Wise A., Kieupel M. Development and evaluation of molecular techniques for the diagnosis of epizootic catarrhal enteritis infection of ferrets. Verh Ver Erkrg Zootiere. 2003;41:427–432. [Google Scholar]
- 16.Breuer-Katschinshi B., Nemes K., Marr A. Helicobacter pylori and the risk of colonic adenomas. Colorectal Adenoma Study Group. Digestion. 1999;60(3):210–215. doi: 10.1159/000007661. [DOI] [PubMed] [Google Scholar]
- 17.Rice L.E., Stahl S.J., McLeod C.G., Jr. Pyloric adenocarcinoma in a ferret. J Am Vet Med Assoc. 1992;200(8):1117–1118. [PubMed] [Google Scholar]
- 18.Sleeman V., Clyde V.L., Jones M.P. Two cases of pyloric adenocarcinoma in the ferret (Mustela putorius furo) Vet Rec. 1995;127(11):272–273. doi: 10.1136/vr.137.11.272. [DOI] [PubMed] [Google Scholar]
- 19.Blanco M.C., Fox J.G., Rosenthal K. Megaesophagus in nine ferrets. J Am Vet Med Assoc. 1994;205(3):444–447. [PubMed] [Google Scholar]
- 20.Malik R., Alderton B., Finlaison D. Cryptococcosis in ferrets: a diverse spectrum of clinical disease. Aust Vet J. 2002;80(12):749–755. doi: 10.1111/j.1751-0813.2002.tb11343.x. [DOI] [PubMed] [Google Scholar]
- 21.Jenkins J.R. Rabbit and ferret liver and gastrointestinal testing. In: Fudge A., editor. Laboratory medicine, avian and exotic pets. WB Saunders; Philadelphia: 2000. [Google Scholar]
- 22.Schwarz L.A., Solano M., Manning A. The normal upper gastrointestinal examination in the ferret. Vet Radiol Ultrasound. 2003;44(2):165–172. doi: 10.1111/j.1740-8261.2003.tb01265.x. [DOI] [PubMed] [Google Scholar]
- 23.Murray M.J. Laparoscopy in the domestic ferret. Proc Int Conf Exot. 2002;4:3. [Google Scholar]
- 24.Munson L. Emerging helicobacter diseases. Proc Am Assoc Zoo Vet. 1996:490. [Google Scholar]
- 25.Carpenter J.W., Mishima T.Y. 2nd edition. WB Saunders; Philadelphia: 2001. Exotic animal formulary. [Google Scholar]
- 26.Fisher P., Lennox A. Therapeutic options for ferret lymphoma: a review. J Exot Mam Med Surg. 2003;1:2. [Google Scholar]
- 27.Medscape Reuters Health Information Helicobacter pylori eradication may cure low-grade gastric lymphoma. Gut. 2004;53:34–37. ( www.medscape.com/viewarticle/466949) [Google Scholar]


