Health Care Infection Control Practices Advisory Committee (HICPAC)
Chair
Patrick J. Brennan, MD, Professor of Medicine, Division of Infectious Diseases, University of Pennsylvania Medical School
Executive Secretary
Michael Bell, MD, Division of Health Care Quality Promotion, National Center for Infectious Diseases, Centers for Disease Control and Prevention
Members
Vicki L. Brinsko, RN, BA, Infection Control Coordinator, Vanderbilt University Medical Center
E. Patchen Dellinger, MD, Professor of Surgery, University of Washington School of Medicine
Jeffrey Engel, MD, Head, General Communicable Disease Control Branch, North Carolina State Epidemiologist
Steven M. Gordon, MD, Chairman, Department of Infections Diseases, Hospital Epidemiologist, Cleveland Clinic Foundation
Lizzie J. Harrell, PhD, D(ABMM), Research Professor of Molecular Genetics, Microbiology and Pathology, Associate Director, Clinical Microbiology, Duke University Medical Center
Carol O'Boyle, PhD, RN, Assistant Professor, School of Nursing, University of Minnesota
David Alexander Pegues, MD, Division of Infectious Diseases, David Geffen School of Medicine at UCLA
Dennis M. Perrotta, PhD, CIC, Adjunct Associate Professor of Epidemiology, University of Texas School of Public Health, Texas A&M University School of Rural Public Health
Harriett M. Pitt, MS, CIC, RN, Director, Epidemiology, Long Beach Memorial Medical Center
Keith M. Ramsey, MD, Professor of Medicine, Medical Director of Infection Control, Brody School of Medicine, East Carolina University
Nalini Singh, MD, MPH, Professor of Pediatrics, Epidemiology and International Health, George Washington University Children's National Medical Center
Kurt Brown Stevenson, MD, MPH Division of Infectious Diseases, Department of Internal Medicine, Ohio State University Medical Center
Philip W. Smith, MD, Chief, Section of Infectious Diseases, Department of Internal Medicine, University of Nebraska Medical Center
HICPAC membership (past)
Robert A. Weinstein, MD (Chair), Cook County Hospital, Chicago, IL
Jane D. Siegel, MD (Co-Chair), University of Texas Southwestern Medical Center, Dallas, TX
Michele L. Pearson, MD (Executive Secretary), Centers for Disease Control and Prevention, Atlanta, GA
Raymond Y.W. Chinn, MD, Sharp Memorial Hospital, San Diego, CA
Alfred DeMaria, Jr, MD, Massachusetts Department of Public Health, Jamaica Plain, MA
James T. Lee, MD, PhD, University of Minnesota, Minneapolis, MN
William A. Rutala, PhD, MPH, University of North Carolina Health Care System, Chapel Hill, NC
William E. Scheckler, MD, University of Wisconsin, Madison, WI
Beth H. Stover, RN, Kosair Children's Hospital, Louisville, KY
Marjorie A. Underwood, RN, BSN CIC, Mt Diablo Medical Center, Concord, CA
HICPAC Liaisons
William B. Baine, MD, Liaison to the Agency for Health Care Quality Research
Joan Blanchard, RN, MSN, CNOR, Liaison to the Association of Perioperative Registered Nurses
Patrick J. Brennan, MD, Liaison to the Board of Scientific Counselors
Nancy Bjerke, RN, MPH, CIC, Liaison to the Association of Professionals in Infection Prevention and Control
Jeffrey P. Engel, MD, Liaison to the Advisory Committee on Elimination of Tuberculosis
David Henderson, MD, Liaison to the National Institutes of Health
Lorine J. Jay, MPH, RN, CPHQ, Liaison to the Health Care Resources Services Administration
Stephen F. Jencks, MD, MPH, Liaison to the Center for Medicare and Medicaid Services
Sheila A. Murphey, MD, Liaison to the Food and Drug Administration
Mark Russi, MD, MPH, Liaison to the American College of Occupational and Environmental Medicine
Rachel L. Stricof, MPH, Liaison to the Advisory Committee on Elimination of Tuberculosis
Michael L. Tapper, MD, Liaison to the Society for Health Care Epidemiology of America
Robert A. Wise, MD, Liaison to the Joint Commission on the Accreditation of Health Care Organizations
Authors' Associations
Jane D. Siegel, MD, Professor of Pediatrics, Department of Pediatrics, University of Texas Southwestern Medical Center
Emily Rhinehart, RN, MPH, CIC, CPHQ, Vice President, AIG Consultants, Inc
Marguerite Jackson, RN, PhD, CIC, Director, Administrative Unit, National Tuberculosis Curriculum Consortium, Department of Medicine, University of California San Diego
Linda Chiarello, RN, MS, Division of Health Care Quality Promotion, National Center for Infectious Diseases, Centers for Disease Control and Prevention
Table of contents
Executive Summary
Abbreviations
Part I: Review of the Scientific Data Regarding Transmission of Infectious Agents in Health Care Settings
-
I.A.
Evolution of the 2007 Document
-
I.B.Rationale for Standard and Transmission-Based Precautions in Health Care Settings
-
I.B.1.Source of Infectious Agents
-
I.B.2.Susceptible Hosts
-
I.B.3.Modes of Transmission
-
I.B.3.a.Contact Transmission
-
I.B.3.a.i.Direct Contact Transmission
-
I.B.3.a.ii.Indirect Contact Transmission
-
I.B.3.a.i.
-
I.B.3.b.Droplet Transmission
-
I.B.3.c.Airborne Transmission
-
I.B.3.d.Emerging Issues and Controversies Concerning Bioaerosols and Airborne Transmission of Infectious Agents
-
I.B.3.d.i.Transmission From Patients
-
I.B.3.d.ii.Transmission From the Environment
-
I.B.3.d.i.
-
I.B.3.e.Other Sources of Infection
-
I.B.3.a.
-
I.B.1.
-
I.C.Infectious Agents of Special Infection Control Interest for Health Care Settings
-
I.C.1.Epidemiologically Important Organisms
-
I.C.1.a.Clostridium difficile
-
I.C.1.b.Multidrug-Resistant Organisms
-
I.C.1.a.
-
I.C.2.Agents of Bioterrorism
-
I.C.3.Prions
-
I.C.4.Severe Acute Respiratory Syndrome (SARS)
-
I.C.5.Monkeypox
-
I.C.6.Noroviruses
-
I.C.7.Hemorrhagic Fever Viruses
-
I.C.1.
-
I.D.Transmission Risks Associated With Specific Types of Health Care Settings
-
I.D.1.Hospitals
-
I.D.1.a.Intensive Care Units
-
I.D.1.b.Burn Units
-
I.D.1.c.Pediatrics
-
I.D.1.a.
-
I.D.2.Nonacute Care Settings
-
I.D.2.a.Long-Term Care
-
I.D.2.b.Ambulatory Care
-
I.D.2.c.Home Care
-
I.D.2.d.Other Sites of Health Care Delivery
-
I.D.2.a.
-
I.D.1.
-
I.E.Transmission Risks Associated With Special Patient Populations
-
I.E.1.Immunocompromised Patients
-
I.E.2.Cystic Fibrosis Patients
-
I.E.1.
-
I.F.New Therapies With Potential Transmissible Infectious Agents
-
I.F.1.Gene Therapy
-
I.F.2.Infections Transmitted Through Blood, Organs and Tissues
-
I.F.3.Xenotransplantation and Tissue Allografts
-
I.F.1.
Part II. Fundamental Elements to Prevent Transmission of Infectious Agents in Health Care Settings
-
II.A.Health Care System Components That Influence the Effectiveness of Precautions to Prevent Transmission
-
II.A.1.Administrative Measures
-
II.A.1.a.Scope of Work and Staffing Needs for Infection Control Professionals
-
II.A.1.a.i.Infection Control Liaison Nurse
-
II.A.1.a.i.
-
II.A.1.b.Bedside Nurse Staffing
-
II.A.1.c.Clinical Microbiology Laboratory Support
-
II.A.1.a.
-
II.A.2.Institutional Safety Culture and Organizational Characteristics
-
II.A.3.Adherence of Health Care Workers to Recommended Guidelines
-
II.A.1.
-
II.B.
Surveillance for Health Care–Associated Infections
-
II.C.
Education of Health Care Workers, Patients, and Families
-
II.D.
Hand Hygiene
-
II.E.Personal Protective Equipment for Health Care Workers
-
II.E.1.Gloves
-
II.E.2.Isolation Gowns
-
II.E.3.Face Protection: Masks, Goggles, Face Shields
-
II.E.3.a.Masks
-
II.E.3.b.Goggles and Face Shields
-
II.E.3.a.
-
II.E.4.Respiratory Protection
-
II.E.1.
-
II.F.Safe Work Practices to Prevent Health Care Worker Exposure to Bloodborne Pathogens
-
II.F.1.Prevention of Needlesticks and Other Sharps-Related Injuries
-
II.F.2.Prevention of Mucous Membrane Contact
-
II.F.2.a.Precautions During Aerosol-Generating Procedures
-
II.F.2.a.
-
II.F.1.
-
II.G.Patient Placement
-
II.G.1.Hospitals and Long-Term Care Settings
-
II.G.2.Ambulatory Care Settings
-
II.G.3.Home Care
-
II.G.1.
-
II.H.
Transport of Patients
-
II.I.
Environmental Measures
-
II.J.
Patient Care Equipment, Instruments/Devices
-
II.K.
Textiles and Laundry
-
II.L.
Solid Waste
-
II.M.
Dishware and Eating Utensils
-
II.N.Adjunctive Measures
-
II.N.1.Chemoprophylaxis
-
II.N.2.Immunoprophylaxis
-
II.N.3.Management of Visitors
-
II.N.3.a.Visitors as Sources of Infection
-
II.N.3.b.Use of Barrier Precautions by Visitors
-
II.N.3.a.
-
II.N.1.
Part III. HICPAC Precautions to Prevent Transmission of Infectious Agents
-
III.A.Standard Precautions
-
III.A.1.New Standard Precautions for Patients
-
III.A.1.a.Respiratory Hygiene/Cough Etiquette
-
III.A.1.b.Safe Injection Practices
-
III.A.1.c.Infection Control Practices for Special Lumbar Puncture Procedures
-
III.A.1.a.
-
III.A.1.
-
III.B.Transmission-Based Precautions
-
III.B.1.Contact Precautions
-
III.B.2.Droplet Precautions
-
III.B.3.Airborne Infection Isolation Precautions
-
III.B.1.
-
III.C.
Syndromic or Empiric Application of Transmission-Based Precautions
-
III.D.
Discontinuation of Precautions
-
III.E.
Application of Transmission-Based Precautions in Ambulatory and Home Care Settings
-
III.F.
Protective Environment
Part IV: Recommendations
Appendix A: Type and Duration of Precautions Needed for Selected Infections and Conditions
Glossary
References
Table 1. Recent history of guidelines for prevention of health care–associated infections
Table 2. Clinical syndromes or conditions warranting additional empiric transmission-based precautions pending confirmation of diagnosis
Table 3. Infection control considerations for high-priority (CDC category A) diseases that may result from bioterrorist attacks or are considered bioterrorist threats
Table 4. Recommendations for application of Standard Precautions for the care of all patients in all health care settings
Table 5. Components of a protective environment
Fig 1. Sequence for donning and removing personal protective equipment
Executive Summary
The Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Health Care Settings 2007 updates and expands the 1996 Guideline for Isolation Precautions in Hospitals. The following developments led to these revisions of the 1996 guideline:
-
1.
The transition of health care delivery from primarily acute care hospitals to other health care settings (eg, home care, ambulatory care, free-standing specialty care sites, long-term care) created a need for recommendations that can be applied in all health care settings using common principles of infection control practice, yet can be modified to reflect setting-specific needs. Accordingly, the revised guideline addresses the spectrum of health care delivery settings. Furthermore, the term “nosocomial infections“ is replaced by “health care–associated infections” (HAIs), to reflect the changing patterns in health care delivery and difficulty in determining the geographic site of exposure to an infectious agent and/or acquisition of infection.
-
2.
The emergence of new pathogens (eg, severe acute respiratory syndrome coronavirus [SARS-CoV] associated with SARS avian influenza in humans), renewed concern for evolving known pathogens (eg, Clostridium difficile, noroviruses, community-associated methicillin-resistant Staphylococcus aureus [CA-MRSA]), development of new therapies (eg, gene therapy), and increasing concern for the threat of bioweapons attacks, necessitates addressing a broader scope of issues than in previous isolation guidelines.
-
3.
The successful experience with Standard Precautions, first recommended in the 1996 guideline, has led to a reaffirmation of this approach as the foundation for preventing transmission of infectious agents in all health care settings. New additions to the recommendations for Standard Precautions are respiratory hygiene/cough etiquette and safe injection practices, including the use of a mask when performing certain high-risk, prolonged procedures involving spinal canal punctures (eg, myelography, epidural anesthesia). The need for a recommendation for respiratory hygiene/cough etiquette grew out of observations during the SARS outbreaks, when failure to implement simple source control measures with patients, visitors, and health care workers (HCWs) with respiratory symptoms may have contributed to SARS-CoV transmission. The recommended practices have a strong evidence base. The continued occurrence of outbreaks of hepatitis B and hepatitis C viruses in ambulatory settings indicated a need to reiterate safe injection practice recommendations as part of Standard Precautions. The addition of a mask for certain spinal injections grew from recent evidence of an associated risk for developing meningitis caused by respiratory flora.
-
4.
The accumulated evidence that environmental controls decrease the risk of life-threatening fungal infections in the most severely immunocompromised patients (ie, those undergoing allogeneic hematopoietic stem cell transplantation [HSCT]) led to the update on the components of the protective environment (PE).
-
5.
Evidence that organizational characteristics (eg, nurse staffing levels and composition, establishment of a safety culture) influence HCWs' adherence to recommended infection control practices, and thus are important factors in preventing transmission of infectious agents, led to a new emphasis and recommendations for administrative involvement in the development and support of infection control programs.
-
6.
Continued increase in the incidence of HAIs caused by multidrug-resistant organisms (MDROs) in all health care settings and the expanded body of knowledge concerning prevention of transmission of MDROs created a need for more specific recommendations for surveillance and control of these pathogens that would be practical and effective in various types of health care settings.
This document is intended for use by infection control staff, health care epidemiologists, health care administrators, nurses, other health care providers, and persons responsible for developing, implementing, and evaluating infection control programs for health care settings across the continuum of care. The reader is referred to other guidelines and websites for more detailed information and for recommendations concerning specialized infection control problems.
Parts I, II, and III: Review of the Scientific Data Regarding Transmission of Infectious Agents in Health Care Settings
Part I reviews the relevant scientific literature that supports the recommended prevention and control practices. As in the 1996 guideline, the modes and factors that influence transmission risks are described in detail. New to the section on transmission are discussions of bioaerosols and of how droplet and airborne transmission may contribute to infection transmission. This became a concern during the SARS outbreaks of 2003, when transmission associated with aerosol-generating procedures was observed. Also new is a definition of “epidemiologically important organisms” that was developed to assist in the identification of clusters of infections that require investigation (ie multidrug-resistant organisms, C difficile). Several other pathogens of special infection control interest (ie, norovirus, SARS, Centers for Disease Control and Prevention [CDC] category A bioterrorist agents, prions, monkeypox, and the hemorrhagic fever viruses) also are discussed, to present new information and infection control lessons learned from experience with these agents. This section of the guideline also presents information on infection risks associated with specific health care settings and patient populations.
Part II updates information on the basic principles of hand hygiene, barrier precautions, safe work practices, and isolation practices that were included in previous guidelines. However, new to this guideline is important information on health care system components that influence transmission risks, including those components under the influence of health care administrators. An important administrative priority that is described is the need for appropriate infection control staffing to meet the ever-expanding role of infection control professionals in the complex modern health care system. Evidence presented also demonstrates another administrative concern: the importance of nurse staffing levels, including ensuring numbers of appropriately trained nurses in intensive care units (ICUs) for preventing HAIs. The role of the clinical microbiology laboratory in supporting infection control is described, to emphasize the need for this service in health care facilities. Other factors that influence transmission risks are discussed, including the adherence of HCWs to recommended infection control practices, organizational safety culture or climate, and education and training.
Discussed for the first time in an isolation guideline is surveillance of health care–associated infections. The information presented will be useful to new infection control professionals as well as persons involved in designing or responding to state programs for public reporting of HAI rates.
Part III describes each of the categories of precautions developed by the Health Care Infection Control Practices Advisory Committee (HICPAC) and the CDC and provides guidance for their application in various health care settings. The categories of Transmission-Based Precautions are unchanged from those in the 1996 guideline: Contact, Droplet, and Airborne. One important change is the recommendation to don the indicated personal protective equipment (PPE—gowns, gloves, mask) on entry into the patient's room for patients who are on Contact and/or Droplet Precautions, because the nature of the interaction with the patient cannot be predicted with certainty, and contaminated environmental surfaces are important sources for transmission of pathogens. In addition, the PE for patients undergoing allogeneic HSCT, described in previous guidelines, has been updated.
Tables, Appendices, and Other Information
Five tables summarize important information. Table 1 provides a summary of the evolution of this document. Table 2 gives guidance on using empiric isolation precautions according to a clinical syndrome. Table 3 summarizes infection control recommendations for CDC category A agents of bioterrorism. Table 4 lists the components of Standard Precautions and recommendations for their application, and Table 5 lists components of the PE.
Table 1.
History of guidelines for isolation precautions in hospitals∗
| Year (reference) | Document issued | Comments |
|---|---|---|
| 19701095 | Isolation Techniques for Use in Hospitals, 1st ed | • Introduced 7 isolation precaution categories with color-coded cards: strict, respiratory, protective, enteric, wound and skin, discharge, and blood. |
| • No user decision making required. | ||
| • Simplicity a strength; overisolation prescribed for some infections. | ||
| 19751100 | Isolation Techniques for Use in Hospitals, 2nd ed | • Same conceptual framework as first edition. |
| 19831096, 1097 | Guideline for Isolation Precautions in Hospitals | • Provided 2 systems for isolation: category-specific and disease-specific. |
| • Protective isolation eliminated; blood precautions expanded to include body fluids. | ||
| • Categories included strict, contact, respiratory, acid-fast bacteria, enteric, drainage/secretion, blood and body fluids. | ||
| • Emphasized decision making by users. | ||
| 1985-88778, 894 | Universal Precautions | • Developed in response to the HIV/AIDS epidemic. |
| • Dictated application of blood and body fluid precautions to all patients, regardless of infection status. | ||
| • Did not apply to feces, nasal secretions, sputum, sweat, tears, urine, or vomitus unless contaminated by visible blood. | ||
| • Added personal protective equipment to protect health care workers from mucous membrane exposures. | ||
| • Handwashing recommended immediately after glove removal. | ||
| • Added specific recommendations for handling needles and other sharp devices; concept became integral to the OSHA's 1991 rule on occupational exposure to blood-borne pathogens in health care settings. | ||
| 19871098 | Body Substance Isolation | • Emphasized avoiding contact with all moist and potentially infectious body substances except sweat even if blood not present. |
| • Shared some features with Universal Precautions. | ||
| • Weak on infections transmitted by large droplets or by contact with dry surfaces. | ||
| • Did not emphasize need for special ventilation to contain airborne infections. | ||
| • Handwashing after glove removal not specified in the absence of visible soiling. | ||
| 19961 | Guideline for Isolation Precautions in Hospitals | • Prepared by the Healthcare Infection Control Practices Advisory Committee. |
| • Melded major features of Universal Precautions and body substance isolation into Standard Precautions to be used with all patients at all times. | ||
| • Included 3 transmission-based precaution categories: Airborne, Droplet, and Contact. | ||
| • Listed clinical syndromes that should dictate use of empiric isolation until an etiologic diagnosis is established. |
Derived from Garner and Simmons.1099
Table 2.
Clinical syndromes or conditions warranting empiric transmission-based precautions in addition to Standard Precautions pending confirmation of diagnosis∗
| Clinical syndrome or condition† | Potential pathogens‡ | Empiric precautions (always includes Standard Precautions) |
|---|---|---|
| Diarrhea | ||
| Acute diarrhea with a likely infectious cause in an incontinent or diapered patient | Enteric pathogens§ | Contact Precautions (pediatrics and adult) |
| Meningitis | Neisseria meningitidis | Droplet Precautions for first 24 hours of antimicrobial therapy; mask and face protection for intubation |
| Enteroviruses | Contact Precautions for infants and children | |
| Mycobacterium tuberculosis | Airborne Precautions if pulmonary infiltrate present | |
| Airborne Precautions plus Contact Precautions if potentially infectious draining body fluid present | ||
| Rash or exanthems, generalized, etiology unknown | ||
| Petechial/ecchymotic with fever (general) | Neisseria meningitides | Droplet Precautions for the first 24 hours of antimicrobial therapy |
| Positive history of travel to an area with an ongoing outbreak of VHF in the 10 days before onset of fever | Ebola, Lassa, Marburg viruses | Droplet Precautions plus Contact Precautions, with face/eye protection, emphasizing safety sharps and Barrier Precautions when blood exposure likely. N95 or higher-level respiratory protection when aerosol-generating procedure performed |
| Vesicular | Varicella-zoster, herpes simplex, variola (smallpox), vaccinia viruses | Airborne plus Contact Precautions |
| Vaccinia virus | Contact Precautions only if herpes simplex, localized zoster in an immunocompetent host, or vaccinia virus likely | |
| Maculopapular with cough, coryza, and fever | Rubeola (measles) virus | Airborne Precautions |
| Respiratory infections | ||
| Cough/fever/upper lobe pulmonary infiltrate in an HIV-negative patient or a patient at low risk for HIV infection | M. tuberculosis, respiratory viruses, Streptococcus pneumoniae, Staphylococcus aureus (MSSA or MRSA) | Airborne Precautions plus Contact Precautions |
| Cough/fever/pulmonary infiltrate in any lung location in an HIV-infected patient or a patient at high risk for HIV infection | M tuberculosis, respiratory viruses, S pneumoniae, S aureus (MSSA or MRSA) | Airborne Precautions plus Contact Precautions; eye/face protection if aerosol-generating procedure performed or contact with respiratory secretions anticipated; Droplet Precautions instead of Airborne Precautions if tuberculosis unlikely and airborne infection isolation room and/or respirator unavailable (tuberculosis more likely in HIV-infected than in HIV-negative individuals) |
| Cough/fever/pulmonary infiltrate in any lung location in a patient with a history of recent travel (10 to 21 days) to countries with active outbreaks of SARS, avian influenza | M tuberculosis, severe acute respiratory syndrome virus (SARS-CoV), avian influenza | Airborne plus Contact Precautions plus eye protection; Droplet Precautions instead of Airborne Precautions if SARS and tuberculosis unlikely |
| Respiratory infections, particularly bronchiolitis and pneumonia, in infants and young children | Respiratory syncytial virus, parainfluenza virus, adenovirus, influenza virus, human metapneumovirus | Contact plus Droplet Precautions; discontinue Droplet Precautions if adenovirus and influenza ruled out |
| Skin or wound infection | ||
| Abscess or draining wound that cannot be covered | S aureus (MSSA or MRSA), group A streptococcus | Contact Precautions, plus Droplet Precautions for the first 24 hours of appropriate antimicrobial therapy if invasive group A streptococcal disease suspected |
Infection control professionals should modify or adapt this table according to local conditions. To ensure that appropriate empiric precautions are implemented always, hospitals must have systems in place to evaluate patients routinely according to these criteria as part of their preadmission and admission care.
Patients with the syndromes or conditions listed below may present with atypical signs or symptoms (eg, neonates and adults with pertussis may not have paroxysmal or severe cough). The clinician's index of suspicion should be guided by the prevalence of specific conditions in the community, as well as clinical judgment.
The organisms listed under the column “Potential Pathogens” are not intended to represent the complete, or even most likely, diagnoses, but rather possible etiologic agents that require additional precautions beyond Standard Precautions until they can be ruled out.
These pathogens include enterohemorrhagic Escherichia coli O157:H7, Shigella spp, hepatitis A virus, noroviruses, rotavirus, and Clostridium difficile.
Table 3.
Infection control considerations for high-priority (CDC category A) diseases that may result from bioterrorist attacks or are considered bioterrorist threats (see http://www.bt.cdc.gov)
| Disease | Anthrax |
|---|---|
| Site(s) of infection; transmission mode | Cutaneous (contact with spores); RT (inhalation of spores); GIT (ingestion of spores [rare]) |
| Cutaneous and inhalation disease have occurred in past bioterrorist incidents | Comment: Spores can be inhaled into the lower respiratory tract. The infectious dose of Bacillus anthracis in humans by any route is not precisely known. In primates, the LD50 for an aerosol challenge with B anthracis is estimated to be 8,000 to 50,000 spores; the infectious dose may be as low as 1 to 3 spores. |
| Incubation period | Cutaneous: 1 to 12 days; RT: Usually 1 to 7 days, but up to 43 days reported; GIT: 15 to 72 hours |
| Clinical features | Cutaneous: Painless, reddish papule that develops a central vesicle or bulla in 1 to 2 days; over the next 3 to 7 days, the lesion becomes pustular and then necrotic, with black eschar and extensive surrounding edema |
| RT: Initial flu-like illness for 1 to 3 days with headache, fever, malaise, cough; by day 4, severe dyspnea and shock. Usually fatal (85% to 90%) if untreated; meningitis develops in 50% of RT cases. | |
| GIT: In intestinal form, necrotic, ulcerated edematous lesions develop in intestines with fever, nausea, and vomiting and progression to hematemesis and bloody diarrhea; 25% to 60% mortality | |
| Diagnosis | Cutaneous: Swabs of lesion (under eschar) for IHC, PCR, and culture; punch biopsy for IHC, PCR, and culture; vesicular fluid aspirate for Gram's stain and culture; blood culture if systemic symptoms present; acute and convalescent sera for ELISA serology |
| RT: CXR or CT demonstrating wide mediastinal widening and/or pleural effusion and hilar abnormalities; blood for culture and PCR; pleural effusion for culture, PCR, and IHC; CSF (if meningeal signs present) for IHC, PCR, and culture; acute and convalescent sera for ELISA serology; pleural and/or bronchial biopsy specimens for IHC | |
| GIT: Blood and ascites fluid, stool samples, rectal swabs, and swabs of oropharyngeal lesions, if present, for culture, PCR, and IHC | |
| Infectivity | Cutaneous: Person-to-person transmission from contact with lesion of untreated patient is possible but rare |
| RT and GIT: Person-to-person transmission does not occur | |
| Aerosolized powder, environmental exposures: Highly infectious if aerosolized | |
| Recommended precautions | Cutaneous: Standard Precautions; Contact Precautions if uncontained copious drainage present |
| RT and GIT: Standard Precautions. | |
| Aerosolized powder, environmental exposures: Respirator (N95 mask or powered air-purifying respirator), protective clothing; decontamination of persons with powder on them (see http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5135a3.htm) | |
| Hand hygiene: Handwashing for 30 to 60 seconds with soap and water or 2% chlorhexidene gluconate after spore contact; alcohol hand rubs are inactive against spores.981 | |
| Postexposure prophylaxis after environmental exposure: A 60-day course of antimicrobials (doxycycline, ciprofloxacin, or levofloxacin) and postexposure vaccine under IND. |
| Disease | Botulism |
|---|---|
| Site(s) of infection; transmission mode | GIT: Ingestion of toxin-containing food; RT: Inhalation of toxin containing aerosol. Comment: Toxin ingested or potentially delivered by aerosol in bioterrorist incidents. LD50 for type A is 0.001 μg/mL/kg. |
| Incubation period | 1 to 5 days. |
| Clinical features | Ptosis, generalized weakness, dizziness, dry mouth and throat, blurred vision, diplopia, dysarthria, dysphonia, and dysphagia, followed by symmetrical descending paralysis and respiratory failure. |
| Diagnosis | Clinical diagnosis: identification .of toxin in stool, serology, unless toxin-containing material available for toxin neutralization bioassays. |
| Infectivity | Not transmitted from person to person; exposure to toxin necessary for disease. |
| Recommended precautions | Standard Precautions. |
| Disease | Ebola Hemorrhagic Fever |
|---|---|
| Site(s) of infection; transmission mode | As a rule, infection develops after exposure of mucous membranes or RT, or through broken skin or percutaneous injury. |
| Incubation period | 2 to 19 days, usually 5 to 10 days |
| Clinical features | Febrile illnesses with malaise, myalgias, headache, vomiting, and diarrhea that are rapidly complicated by hypotension, shock, and hemorrhagic features. Massive hemorrhage in < 50% of patients. |
| Diagnosis | Etiologic diagnosis can be made using reverse-transcription-PCR, serologic detection of antibody and antigen, pathologic assessment with immunohistochemistry, and viral culture with electromicroscopic confirmation of morphology, |
| Infectivity | Person-to-person transmission occurs primarily through unprotected contact with blood and body fluids; percutaneous injuries (eg, needlestick) are associated with a high rate of transmission. Transmission in health care settings has been reported but can be prevented by use of Barrier Precautions. |
| Recommended precautions | Hemorrhagic fever–specific Barrier Precautions: If disease is believed to be related to intentional release of a bioweapon, then the epidemiology of transmission is unpredictable pending observation of disease transmission. Until the nature of the pathogen is understood and its transmission pattern confirmed, Standard, Contact, and Airborne Precautions should be used. Once the pathogen is characterized, if the epidemiology of transmission is consistent with natural disease, then Droplet Precautions can be substituted for Airborne Precautions. Emphasize the following: (1) use of sharps safety devices and safe work practices, (2) proper hand hygiene, (3) barrier protection against blood and body fluids on entry into room (single gloves and fluid-resistant or impermeable gown, face/eye protection with masks, goggles or face shields), and (4) appropriate waste handling. Use N95 or higher respirators when performing aerosol-generating procedures. In settings where AIIRs are unavailable or the large numbers of patients cannot be accommodated by existing AIIRs, observe Droplet Precautions (plus Standard and Contact Precautions) and segregate patients from those not suspected as having VHF infection. Limit blood draws to those essential to care. See the text for discussion and Appendix A for recommendations for naturally occurring VHFs. |
| Disease | Plague∗ |
|---|---|
| Site(s) of infection; transmission mode | RT: Inhalation of respiratory droplets. Comment: Pneumonic plague is most likely when used as a biological weapon, but some cases of bubonic and primary septicemia also may occur. Infective dose, 100 to 500 bacteria. |
| Incubation period | 1 to 6 days, usually 2 to 3 days. |
| Clinical features | Pneumonic: Fever, chills, headache, cough, dyspnea, rapid progression of weakness, and, in later stages, hemoptysis, circulatory collapse, and bleeding diathesis. |
| Diagnosis | Presumptive is diagnosis from Gram's stain or Wayson's stain of sputum, blood, or lymph node aspirate; definitive diagnosis is from cultures of same material or paired acute/convalescent serology. |
| Infectivity | Person-to-person transmission occurs through respiratory droplets. Risk of transmission is low during the first 20 to 24 hours of illness and requires close contact. Respiratory secretions probably are not infectious within a few hours after initiation of appropriate therapy. |
| Recommended precautions | Standard and Droplet Precautions until patients have received 48 hours of appropriate therapy. Chemoprophylaxis: Consider antibiotic prophylaxis for HCWs with close contact exposure. |
| Disease | Smallpox |
|---|---|
| Site(s) of infection; transmission mode | RT Inhalation of droplet or, rarely, aerosols; and skin lesions (contact with virus). |
| Comment: If used as a biological weapon, natural disease (which has not occurred since 1977) likely will result. | |
| Incubation period | 7 to 19 days (mean, 12 days). |
| Clinical features | Fever, malaise, backache, headache, and often vomiting for 2 to 3 days, followed by generalized papular or maculopapular rash (more on face and extremities), which becomes vesicular (on day 4 or 5) and then pustular; lesions all in same stage. |
| Diagnosis | Electron microscopy of vesicular fluid or culture of vesicular fluid by a World Health Organization–approved laboratory (CDC); detection by PCR available only at select LRN laboratories, the CDC, and US Army Medical Research Institute of Infectious Diseases. |
| Infectivity | Secondary attack rates up to 50% in unvaccinated persons. Infected persons may transmit disease from the time that rash appears until all lesions have crusted over (about 3 weeks). Infectivity is greatest during the first 10 days of rash. |
| Recommended precautions | Combined use of Standard, Contact, and Airborne Precautions should be maintained until all scabs have separated (3 to 4 weeks).† Only immune HCWs should care for patients. Postexposure vaccine should be provided within 4 days. |
| Vaccinia‡: HCWs to cover vaccination site with gauze and semipermeable dressing until scab separates (≥ 21 days). Hand hygiene should be observed. | |
| Adverse events with virus-containing lesions: Standard Precautions plus Contact Precautions until all lesions are crusted. |
| Disease | Tularemia |
|---|---|
| Site(s) of infection; transmission mode | RT: Inhalation of aerosolized bacteria; GIT: Ingestion of food or drink contaminated with aerosolized bacteria. |
| Comment: Pneumonic or typhoidal disease likely to occur after bioterrorist event using aerosol delivery. Infective dose, 10 to 50 bacteria. | |
| Incubation period | 2 to 10 days; usually 3 to 5 days. |
| Clinical features | Pneumonic: malaise, cough, sputum production, dyspnea. Typhoidal: fever, prostration, weight loss and frequently an associated pneumonia. |
| Diagnosis | Diagnosis usually made with serology on acute and convalescent serum specimens; bacterium can be detected by PCR (LRN) or isolated from blood and other body fluids on cysteine-enriched media or mouse inoculation. |
| Infectivity | Person-to-person spread is rare. Laboratory workers who encounter/handle cultures of this organism are at high risk for disease if exposed. |
| Recommended precautions | Standard Precautions |
AIIR, airborne infection isolation room; BSL, biosafety level; CSF, cerebrospinal fluid; CT, computed tomography; CXR, chest x-ray; ELISA, enzyme-linked immunosorbent assay; GIT, gastrointestinal tract; HCW, health care worker; IHC, immunohistochemistry; LD50, lethal dose for 50% of experimental animals; LRN, Laboratory Response Network; PAPR, powered air-purifying respirator; PCR, polymerase chain reaction; RT, respiratory tract; VHF, viral hemorrhagic fever.
Pneumonic plague is not as contagious as is often thought. Historical accounts and contemporary evidence indicate that persons with plague usually transmit the infection only when the disease is in the end stage. These persons cough copious amounts of bloody sputum that contains many plague bacteria. Patients in the early stage of primary pneumonic plague (approximately the first 20 to 24 hours) apparently pose little risk (Wu L-T. A treatise on pneumonic plague. Geneva, Switzerland: League of Nations; 1926; Kool JL. Risk of person-to-person transmission of pneumonic plague. Clin Infect Dis 2005;40:1166-72). Antibiotic medication rapidly clears the sputum of plague bacilli, so that a patient generally is not infective within hours after initiation of effective antibiotic treatment (Butler TC. Plague and other Yersinia infections. In: Greenough WB, editor. Current topics in infectious disease. New York: Plenum; 1983). This means that in modern times, many patients will never reach a stage where they pose a significant risk to others. Even in the end stage of disease, transmission occurs only after close contact. Simple protective measures, such as wearing masks, maintaining good hygiene, and avoiding close contact, have been effective in interrupting transmission during many pneumonic plague outbreaks; in the United States, the last known case of person-to-person transmission of pneumonic plague occurred in 1925 (Kool JL. Risk of person-to-person transmission of pneumonic plague. Clin Infect Dis 2005;40:1166-72).
Transmission by the airborne route is a rare event. Airborne Precautions are recommended when possible, but in the event of mass exposures, Barrier Precautions and containment within a designated area are most important.204, 212
Vaccinia adverse events with lesions containing infectious virus include inadvertent autoinoculation, ocular lesions (blepharitis, conjunctivitis), generalized vaccinia, progressive vaccinia, and eczema vaccinatum. Bacterial superinfection also requires addition of Contact Precautions if exudates cannot be contained.216, 217
Table 4.
Recommendations for application of Standard Precautions for the care of all patients in all healthcare settings (see Sections II.D to II.J and III.A.1)
| Component | Recommendations |
|---|---|
| Hand hygiene | After touching blood, body fluids, secretions, excretions, contaminated items; immediately after removing gloves; between patient contacts |
| Personal protective equipment (PPE) | |
| Gloves | For touching blood, body fluids, secretions, excretions, contaminated items, mucous membranes, and nonintact skin |
| Gown | During procedures and patient care activities when contact of clothing/exposed skin with blood/body fluids, secretions, and excretions is anticipated |
| Mask, eye protection (goggles), face shield∗ | During procedures and patient care activities likely to generate splashes or sprays of blood, body fluids, secretions, especially suctioning, endotracheal intubation |
| Soiled patient care equipment | Handle in a manner that prevents transfer of microorganisms to others and to the environment; wear gloves if visibly contaminated; perform hand hygiene |
| Environmental control | Develop procedures for routine care, cleaning, and disinfection of environmental surfaces, especially frequently touched surfaces in patient care areas |
| Textiles and laundry | Handle in a manner that prevents transfer of microorganisms to others and to the environment |
| Needles and other sharps | Do not recap, bend, break, or hand-manipulate used needles; if recapping is required, use a one-handed scoop technique only; use safety features when available; place used sharps in puncture-resistant container |
| Patient resuscitation | Use mouthpiece, resuscitation bag, other ventilation devices to prevent contact with mouth and oral secretions |
| Patient placement | Prioritize for single-patient room if patient is at increased risk of transmission, is likely to contaminate the environment, does not maintain appropriate hygiene, or is at increased risk of acquiring infection or developing adverse outcome after infection |
| Respiratory hygiene/cough etiquette (source containment of infectious respiratory secretions in symptomatic patients, beginning at initial point of encounter, eg, triage and reception areas in emergency departments and physician offices) | Instruct symptomatic persons to cover mouth/nose when sneezing/coughing; use tissues and dispose in no-touch receptacle; observe hand hygiene after soiling of hands with respiratory secretions; wear surgical mask if tolerated or maintain spatial separation, >3 feet if possible. |
During aerosol-generating procedures on patients with suspected or proven infections transmitted by respiratory aerosols (eg, severe acute respiratory syndrome), wear a fit-tested N95 or higher respirator in addition to gloves, gown, and face/eye protection.
Table 5.
Components of a protective environment
| I. Patients: allogeneic hematopoeitic stem cell transplantation only |
| • Maintain in protective environment (PE) room except for required diagnostic or therapeutic procedures that cannot be performed in the room (eg, radiology, surgery) |
| • Respiratory protection (eg, N95 respirator) for the patient when leaving PE during periods of construction |
| II. Standard and Expanded Precautions |
| • Hand hygiene observed before and after patient contact |
| • Gown, gloves, mask not required for health care workers (HCWs) or visitors for routine entry into the room |
| • Use of gown, gloves, and mask by HCWs and visitors according to Standard Precautions and as indicated for suspected or proven infections for which transmission-based precautions are recommended |
| III. Engineering |
| • Central or point-of-use high-efficiency particulate air (HEPA) filters (99.97% efficiency) filters capable of removing particles 0.3 μm in diameter in supply (incoming) air |
| • Well-sealed rooms: |
| - Proper construction of windows, doors, and intake and exhaust ports |
| - Ceilings: smooth, free of fissures, open joints, crevices |
| - Walls sealed above and below the ceiling |
| - If leakage detected, locate source and make necessary repairs |
| • Ventilation to maintain ≥12 air changes/hour |
| • Directed air flow; air supply and exhaust grills located so that clean, filtered air enters from one side of the room, flows across the patient's bed, and exits on opposite side of the room |
| • Positive room air pressure in relation to the corridor; pressure differential of >2.5 Pa (0.01-inch water gauge) |
| • Air flow patterns monitored and recorded daily using visual methods (eg, flutter strips, smoke tubes) or a hand-held pressure gauge |
| • Self-closing door on all room exits |
| • Back-up ventilation equipment (eg, portable units for fans or filters) maintained for emergency provision of ventilation requirements for PE areas, with immediate steps taken to restore the fixed ventilation system |
| • For patients who require both a PE and an airborne infection isolation room (AIIR), use an anteroom to ensure proper air balance relationships and provide independent exhaust of contaminated air to the outside, or place a HEPA filter in the exhaust duct. If an anteroom is not available, place patient in an AIIR and use portable ventilation units, industrial-grade HEPA filters to enhance filtration of spores. |
| IV. Surfaces |
| • Daily wet-dusting of horizontal surfaces using cloths moistened with EPA-registered hospital disinfectant/detergent |
| • Avoid dusting methods that disperse dust |
| • No carpeting in patient rooms or hallways |
| • No upholstered furniture and furnishings |
| V. Other |
| • No flowers (fresh or dried) or potted plants in PE rooms or areas |
| • Vacuum cleaner equipped with HEPA filters when vacuum cleaning is necessary |
Adapted from Centers for Disease Control and Prevention.11
A glossary of definitions used in this guideline also is provided. New to this edition of the guideline is a figure showing the recommended sequence for donning and removing PPE used for isolation precautions to optimize safety and prevent self-contamination during removal.
Appendix A: Type and Duration of Precautions Recommended for Selected Infections and Conditions
Appendix A provides an updated alphabetical list of most infectious agents and clinical conditions for which isolation precautions are recommended. A preamble to the appendix provides a rationale for recommending the use of 1 or more Transmission-Based Precautions in addition to Standard Precautions, based on a review of the literature and evidence demonstrating a real or potential risk for person-to-person transmission in health care settings. The type and duration of recommended precautions are presented, with additional comments concerning the use of adjunctive measures or other relevant considerations to prevent transmission of the specific agent. Relevant citations are included.
Prepublication of the Guideline on Preventing Transmission of MDROs
New to this guideline is a comprehensive review and detailed recommendations for prevention of transmission of MDROs. This portion of the guideline was published electronically in October 2006 and updated in November 2006 (Siegel JD, Rhinehart E, Jackson M, Chiarello L and HICPAC. Management of multidrug-resistant organisms in health care settings, 2006; available from http://www.cdc.gov/ncidod/dhqp/pdf/ar/mdroGuideline2006.pdf), and is considered a part of the Guideline for Isolation Precautions. This section provides a detailed review of the complex topic of MDRO control in health care settings and is intended to provide a context for evaluation of MDRO at individual health care settings. A rationale and institutional requirements for developing an effective MDRO control program are summarized.
Although the focus of this guideline is on measures to prevent transmission of MDROs in health care settings, information concerning the judicious use of antimicrobial agents also is presented, because such practices are intricately related to the size of the reservoir of MDROs, which in turn influences transmission (eg, colonization pressure). Two tables summarize recommended prevention and control practices using 7 categories of interventions to control MDROs: administrative measures, education of HCWs, judicious antimicrobial use, surveillance, infection control precautions, environmental measures, and decolonization. Recommendations for each category apply to and are adapted for the various health care settings.
With the increasing incidence and prevalence of MDROs, all health care facilities must prioritize effective control of MDRO transmission. Facilities should identify prevalent MDROs at the facility, implement control measures, assess the effectiveness of control programs, and demonstrate decreasing MDRO rates. A set of intensified MDRO prevention interventions is to be added if the incidence of transmission of a target MDRO is not decreasing despite implementation of basic MDRO infection control measures, and when the first case of an epidemiologically important MDRO is identified within a health care facility.
Summary
This updated guideline responds to changes in health care delivery and addresses new concerns about transmission of infectious agents to patients and HCWs in the United States and infection control. The primary objective of the guideline is to improve the safety of the nation's health care delivery system by reducing the rates of HAIs.
Abbreviations Used in the Guideline
- AIA
American Institute of Architects
- AIIR
Airborne infection isolation room
- CDC
Centers for Disease Control and Prevention
- CF
Cystic fibrosis
- CJD
Creutzfeld-Jakob Disease
- ESBL
Extended-spectrum beta-lactamase
- FDA
Food and Drug Administration
- HAI
Health care–associated infection
- HBV
Hepatitis B virus
- HCV
Hepatitis C virus
- HEPA
High-efficiency particulate air
- HICPAC
Health Care Infection Control Practices Advisory Committee
- HIV
Human immunodeficiency virus
- HCW
Health care worker
- HFV
Hemorrhagic fever virus
- HSCT
Hematopoetic stem cell transplantation
- ICP
Infection prevention and control professional
- ICU
Intensive care unit
- LTCF
Long-term care facility
- MDR-GNB
Multidrug-resistant gram-negative bacilli
- MDRO
Multidrug-resistant organism
- MRSA
Methicillin-resistant Staphylococcus aureus
- MSSA
Methicillin-susceptible Staphylococcus aureus
- NICU
Neonatal intensive care unit
- NIOSH
National Institute for Occupational Safety and Health
- NNIS
National Nosocomial Infection Surveillance
- NSSP
Nonsusceptible Streptococcus pneumoniae
- OSHA
Occupational Safety and Health Administration
- PCR
Polymerase chain reaction
- PE
Protective environment
- PFGE
Pulsed-field gel electrophoresis
- PICU
Pediatric intensive care unit
- PPE
Personal protective equipment
- RSV
Respiratory syncytial virus
- SARS
Severe acute respiratory syndrome
- vCJD
variant Creutzfeld-Jakob disease
- VISA
Vancomycin-intermediate/resistannt Staphylococcus aureus
- VRE
Vancomycin-resistant enterococci
- VRSA
Vancomycin-resistant Staphylococcus aureus
- WHO
World Health Organization
Part I: Review of Scientific Data Regarding Transmission of Infectious Agents in Health Care Settings
I.A. Evolution of the 2007 Document
The Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Health care Settings 2007 builds on a series of isolation and infection prevention documents promulgated since 1970. These previous documents are summarized and referenced in Table 1 and in Part I of the 1996 Guideline for Isolation Precautions in Hospitals.1
I.A.1. Objectives and Methods
The objectives of this guideline are to (1) provide infection control recommendations for all components of the health care delivery system, including hospitals, long-term care facilities, ambulatory care, home care, and hospice; (2) reaffirm Standard Precautions as the foundation for preventing transmission during patient care in all health care settings; (3) reaffirm the importance of implementing Transmission-Based Precautions based on the clinical presentation or syndrome and likely pathogens until the infectious etiology has been determined (Table 2); and (4) provide epidemiologically sound and, whenever possible, evidence-based recommendations.
This guideline is designed for use by individuals who are charged with administering infection control programs in hospitals and other health care settings. The information also will be useful for other HCWs, health care administrators, and anyone needing information about infection control measures to prevent transmission of infectious agents. Commonly used abbreviations are provided, and terms used in the guideline are defined in the Glossary.
Medline and PubMed were used to search for relevant studies published in English, focusing on those published since 1996. Much of the evidence cited for preventing transmission of infectious agents in health care settings is derived from studies that used “quasi-experimental designs,” also referred to as nonrandomized preintervention and postintervention study designs.2 Although these types of studies can provide valuable information regarding the effectiveness of various interventions, several factors decrease the certainty of attributing improved outcome to a specific intervention. These include: difficulties in controlling for important confounding variables, the use of multiple interventions during an outbreak, and results that are explained by the statistical principle of regression to the mean (eg, improvement over time without any intervention).3 Observational studies remain relevant and have been used to evaluate infection control interventions.4, 5 The quality of studies, consistency of results, and correlation with results from randomized controlled trials, when available, were considered during the literature review and assignment of evidence-based categories (see Part IV: Recommendations) to the recommendations in this guideline. Several authors have summarized properties to consider when evaluating studies for the purpose of determining whether the results should change practice or in designing new studies.2, 6, 7
I.A.2. Changes or Clarifications in Terminology
This guideline contains 4 changes in terminology from the 1996 guideline:
-
1.
The term “nosocomial infection” is retained to refer only to infections acquired in hospitals. The term “health care–associated infection” (HAI) is used to refer to infections associated with health care delivery in any setting (eg, hospitals, long-term care facilities, ambulatory settings, home care). This term reflects the inability to determine with certainty where the pathogen was acquired, because patients may be colonized with or exposed to potential pathogens outside of the health care setting before receiving health care, or may develop infections caused by those pathogens when exposed to the conditions associated with delivery of health care. In addition, patients frequently move among the various settings within the health care system.8
-
2.
A new addition to the practice recommendations for Standard Precautions is respiratory hygiene/cough etiquette. Whereas Standard Precautions generally apply to the recommended practices of HCWs during patient care, respiratory hygiene/cough etiquette applies broadly to all persons who enter a health care setting, including HCWs, patients, and visitors. These recommendations evolved from observations during the SARS epidemic that failure to implement basic source control measures with patients, visitors, and HCWs with signs and symptoms of respiratory tract infection may have contributed to SARS-CoV transmission. This concept has been incorporated into CDC planning documents for SARS and pandemic influenza.9, 10
-
3.
The term “Airborne Precautions” has been supplemented by the term “Airborne Infection Isolation Room” (AIIR), to achieve consistency with the Guidelines for Environmental Infection Control in Health Care Facilities,11 the Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health Care Settings 2005,12 and the American Institute of Architects (AIA) 2006 guidelines for design and construction of hospitals.13
-
4.
A set of prevention measures known as the protective environment (PE) has been added to the precautions for preventing HAIs. These measures, which have been defined in previous guidelines, consist of engineering and design interventions aimed at decreasing the risk of exposure to environmental fungi for severely immunocompromised patients undergoing allogeneic HSCT during the times of highest risk, usually the first 100 days posttransplantation or longer in the presence of graft-versus-host disease.11, 13, 14, 15 Recommendations for a PE apply only to acute care hospitals that provide care to patients undergoing HSCT.
I.A.3. Scope
This guideline, like its predecessors, focuses primarily on interactions between patients and health care providers. The Guidelines for the Prevention of MDRO Infection were published separately in November 2006 and are available online at http://www.cdc.gov/ncidod/dhqp/index.html. Several other HICPAC guidelines to prevent transmission of infectious agents associated with health care delivery are cited, including Guideline for Hand Hygiene, Guideline for Environmental Infection Control, Guideline for Prevention of Health Care–Associated Pneumonia, and Guideline for Infection Control in Health Care Personnel.11, 14, 16, 17 In combination, these provide comprehensive guidance on the primary infection control measures for ensuring a safe environment for patients and HCWs.
This guideline does not discuss in detail specialized infection control issues in defined populations that are addressed elsewhere (eg, Recommendations for Preventing Transmission of Infections Among Chronic Hemodialysis Patients, Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health Care Facilities 2005, Guidelines for Infection Control in Dental Health Care Settings, and Infection Control Recommendations for Patients With Cystic Fibrosis.12, 18, 19, 20 An exception has been made by including abbreviated guidance for a PE used for allogeneic HSCT recipients, because components of the PE have been defined more completely since publication of the Guidelines for Preventing Opportunistic Infections Among HSCT Recipients in 2000 and the Guideline for Environmental Infection Control in Health Care Facilities.11, 15
I.B. Rationale for Standard and Transmission-Based Precautions in Health Care Settings
Transmission of infectious agents within a health care setting requires 3 elements: a source (or reservoir) of infectious agents, a susceptible host with a portal of entry receptive to the agent, and a mode of transmission for the agent. This section describes the interrelationship of these elements in the epidemiology of HAIs.
I.B.1. Sources of Infectious Agents
Infectious agents transmitted during health care derive primarily from human sources but inanimate environmental sources also are implicated in transmission. Human reservoirs include patients,20, 21, 22, 23, 24, 25, 26, 27, 28 HCWs,17, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39 and household members and other visitors.40, 41, 42, 43, 44, 45 Such source individuals may have active infections, may be in the asymptomatic and/or incubation period of an infectious disease, or may be transiently or chronically colonized with pathogenic microorganisms, particularly in the respiratory and gastrointestinal tracts. Other sources of HAIs are the endogenous flora of patients (eg, bacteria residing in the respiratory or gastrointestinal tract).46, 47, 48, 49, 50, 51, 52, 53, 54
I.B.2. Susceptible Hosts
Infection is the result of a complex interrelationship between a potential host and an infectious agent. Most of the factors that influence infection and the occurrence and severity of disease are related to the host. However, characteristics of the host–agent interaction as it relates to pathogenicity, virulence, and antigenicity also are important, as are the infectious dose, mechanisms of disease production, and route of exposure.55 There is a spectrum of possible outcomes after exposure to an infectious agent. Some persons exposed to pathogenic microorganisms never develop symptomatic disease, whereas others become severely ill and even die. Some individuals are prone to becoming transiently or permanently colonized but remain asymptomatic. Still others progress from colonization to symptomatic disease either immediately after exposure or after a period of asymptomatic colonization. The immune state at the time of exposure to an infectious agent, interaction between pathogens, and virulence factors intrinsic to the agent are important predictors of an individual's outcome. Host factors such as extremes of age and underlying disease (eg, diabetes56, 57, human immunodeficiency virus/acquired immune deficiency syndrome [HIV/AIDS],58, 59 malignancy, and transplantation18, 60, 61) can increase susceptibility to infection, as can various medications that alter the normal flora (eg, antimicrobial agents, gastric acid suppressors, corticosteroids, antirejection drugs, antineoplastic agents, immunosuppressive drugs). Surgical procedures and radiation therapy impair defenses of the skin and other involved organ systems. Indwelling devices, such as urinary catheters, endotracheal tubes, central venous and arterial catheters,62, 63, 64 and synthetic implants, facilitate development of HAIs by allowing potential pathogens to bypass local defenses that ordinarily would impede their invasion and by providing surfaces for development of biofilms that may facilitate adherence of microorganisms and protect from antimicrobial activity.65 Some infections associated with invasive procedures result from transmission within the health care facility; others arise from the patient's endogenous flora.46, 47, 48, 49, 50 High-risk patient populations with noteworthy risk factors for infection are discussed further in Sections I.D, I.E, and I.F.
I.B.3. Modes of Transmission
Several classes of pathogens can cause infection, including bacteria, viruses, fungi, parasites, and prions. The modes of transmission vary by type of organism, and some infectious agents may be transmitted by more than 1 route. Some are transmitted primarily by direct or indirect contact, (eg, herpes simplex virus [HSV], respiratory syncytial virus, S aureus), others by the droplet, (eg, influenza virus, Bordetella pertussis) or airborne routes (eg, Mycobacterium tuberculosis). Other infectious agents, such as bloodborne viruses (eg, hepatitis B virus [HBV], hepatitis C virus [HCV], HIV), are rarely transmitted in health care settings through percutaneous or mucous membrane exposure. Importantly, not all infectious agents are transmitted from person to person; these are listed in Appendix A. The 3 principal routes of transmission—contact, droplet, and airborne—are summarized below.
I.B.3.a. Contact Transmission
The most common mode of transmission, contact transmission is divided into 2 subgroups: direct contact and indirect contact.
I.B.3.a.i. Direct Contact Transmission
Direct transmission occurs when microorganisms are transferred from an infected person to another person without a contaminated intermediate object or person. Opportunities for direct contact transmission between patients and HCWs have been summarized in HICPAC's Guideline for Infection Control in Health Care Personnel, 1998 17 and include the following:
-
•
Blood or other blood-containing body fluids from a patient directly enters a HCW's body through contact with a mucous membrane66 or breaks (ie, cuts, abrasions) in the skin.67
-
•
Mites from a scabies-infested patient are transferred to a HCW's skin while he or she is in direct ungloved contact with the patient's skin.68, 69
-
•
A HCW develops herpetic whitlow on a finger after contact with HSV when providing oral care to a patient without using gloves, or HSV is transmitted to a patient from a herpetic whitlow on an ungloved hand of a HCW.70, 71
I.B.3.a.ii. Indirect Contact Transmission
Indirect transmission involves the transfer of an infectious agent through a contaminated intermediate object or person. In the absence of a point-source outbreak, it is difficult to determine how indirect transmission occurs. However, extensive evidence cited in the Guideline for Hand Hygiene in Health Care Settings suggests that the contaminated hands of HCWs are important contributors to indirect contact transmission.16 Examples of opportunities for indirect contact transmission include the following:
-
•
A HCWs' hands may transmit pathogens after touching an infected or colonized body site on 1 patient or a contaminated inanimate object, if hand hygiene is not performed before touching another patient.72, 73
-
•
Patient-care devices (eg, electronic thermometers, glucose monitoring devices) may transmit pathogens if devices contaminated with blood or body fluids are shared between patients without cleaning and disinfecting between patients.74, 75, 76, 77
-
•
Shared toys may become a vehicle for transmitting respiratory viruses (eg, respiratory syncytial virus [RSV]24, 78, 79 or pathogenic bacteria (eg, Pseudomonas aeruginosa 80) among pediatric patients.
-
•
Instruments that are inadequately cleaned between patients before disinfection or sterilization (eg, endoscopes or surgical instruments)81, 82, 83, 84, 85 or that have manufacturing defects that interfere with the effectiveness of reprocessing86, 87 may transmit bacterial and viral pathogens.
Clothing, uniforms, laboratory coats, or isolation gowns used as PPE may become contaminated with potential pathogens after care of a patient colonized or infected with an infectious agent, (eg, MRSA,88 vancomycin-resistant enterococci [VRE],89 and C difficile 90). Although contaminated clothing has not been implicated directly in transmission, the potential exists for soiled garments to transfer infectious agents to successive patients.
I.B.3.b. Droplet Transmission
Droplet transmission is technically a form of contact transmission; some infectious agents transmitted by the droplet route also may be transmitted by direct and indirect contact routes. However, in contrast to contact transmission, respiratory droplets carrying infectious pathogens transmit infection when they travel directly from the respiratory tract of the infectious individual to susceptible mucosal surfaces of the recipient, generally over short distances, necessitating facial protection. Respiratory droplets are generated when an infected person coughs, sneezes, or talks91, 92 or during such procedures as suctioning, endotracheal intubation,93, 94, 95, 96 cough induction by chest physiotherapy,97 and cardiopulmonary resuscitation.98, 99 Evidence for droplet transmission comes from epidemiologic studies of disease outbreaks,100, 101, 102, 103 from experimental studies,104 and from information on aerosol dynamics.91, 105 Studies have shown that the nasal mucosa, conjunctivae, and, less frequently, the mouth are susceptible portals of entry for respiratory viruses.106 The maximum distance for droplet transmission is currently unresolved; pathogens transmitted by the droplet route have not been transmitted through the air over long distances, in contrast to the airborne pathogens discussed below. Historically, the area of defined risk has been a distance of < 3 feet around the patient, based on epidemiologic and simulated studies of selected infections.103, 104 Using this distance for donning masks has been effective in preventing transmission of infectious agents through the droplet route. However, experimental studies with smallpox107, 108 and investigations during the global SARS outbreaks of 2003101 suggest that droplets from patients with these 2 infections could reach persons located 6 feet or more from their source. It is likely that the distance that droplets travel depends on the velocity and mechanism by which respiratory droplets are propelled from the source, the density of respiratory secretions, environmental factors (eg, temperature, humidity), and the pathogen's ability to maintain infectivity over that distance.105 Thus, a distance of < 3 feet around the patient is best considered an example of what is meant by “a short distance from a patient” and should not be used as the sole criterion for determining when a mask should be donned to protect from droplet exposure. Based on these considerations, it may be prudent to don a mask when within 6 to 10 feet of the patient or on entry into the patient's room, especially when exposure to emerging or highly virulent pathogens is likely. More studies are needed to gain more insight into droplet transmission under various circumstances.
Droplet size is another variable under investigation. Droplets traditionally have been defined as being > 5 μm in size. Droplet nuclei (ie, particles arising from desiccation of suspended droplets) have been associated with airborne transmission and defined as < 5 μm in size,105 a reflection of the pathogenesis of pulmonary tuberculosis that is not generalizeable to other organisms. Observations of particle dynamics have demonstrated that a range of droplet sizes, including those of diameter ≥ 30 μm, can remain suspended in the air.109 The behavior of droplets and droplet nuclei affect recommendations for preventing transmission. Whereas fine airborne particles containing pathogens that are able to remain infective may transmit infections over long distances, requiring AIIR to prevent its dissemination within a facility; organisms transmitted by the droplet route do not remain infective over long distances and thus do not require special air handling and ventilation. Examples of infectious agents transmitted through the droplet route include B pertussis,110 influenza virus,23 adenovirus,111 rhinovirus,104 Mycoplasma pneumoniae,112 SARS-CoV,21, 96, 113 group A streptococcus,114 and Neisseria meningitides.95, 103, 115 Although RSV may be transmitted by the droplet route, direct contact with infected respiratory secretions is the most important determinant of transmission and consistent adherence to Standard Precautions plus Contact Precautions prevents transmission in health care settings.24, 116, 117
Rarely, pathogens that are not transmitted routinely by the droplet route are dispersed into the air over short distances. For example, although S aureus is transmitted most frequently by the contact route, viral upper respiratory tract infection has been associated with increased dispersal of S aureus from the nose into the air for a distance of 4 feet under both outbreak and experimental conditions; this is known as the “cloud baby” and “cloud adult” phenomenon.118, 119, 120
I.B.3.c. Airborne Transmission
Airborne transmission occurs by dissemination of either airborne droplet nuclei or small particles in the respirable size range containing infectious agents that remain infective over time and distance (eg, spores of Aspergillus spp and M tuberculosis). Microorganisms carried in this manner may be dispersed over long distances by air currents and may be inhaled by susceptible individuals who have not had face-to-face contact with (or even been in the same room with) the infectious individual.121, 122, 123, 124 Preventing the spread of pathogens that are transmitted by the airborne route requires the use of special air handling and ventilation systems (eg, AIIRs) to contain and then safely remove the infectious agent.11, 12 Infectious agents to which this applies include M tuberculosis,124, 125, 126, 127 rubeola virus (measles),122 and varicella-zoster virus (chickenpox).123 In addition, published data suggest the possibility that variola virus (smallpox) may be transmitted over long distances through the air under unusual circumstances, and AIIRs are recommended for this agent as well; however, droplet and contact routes are the more frequent routes of transmission for smallpox.108, 128, 129 In addition to AIIRs, respiratory protection with a National Institute for Occupational Safety and Health (NIOSH)-certified N95 or higher-level respirator is recommended for HCWs entering the AIIR, to prevent acquisition of airborne infectious agents such as M tuberculosis.12
For certain other respiratory infectious agents, such as influenza130, 131 and rhinovirus,104 and even some gastrointestinal viruses (eg, norovirus132 and rotavirus133), there is some evidence that the pathogen may be transmitted through small-particle aerosols under natural and experimental conditions. Such transmission has occurred over distances > 3 feet but within a defined air space (eg, patient room), suggesting that it is unlikely that these agents remain viable on air currents that travel long distances. AIIRs are not routinely required to prevent transmission of these agents. Additional issues concerning small-particle aerosol transmission of agents that are most frequently transmitted by the droplet route are discussed below.
I.B.3.d. Emerging Issues Concerning Airborne Transmission of Infectious Agents
I.B.3.d.i. Transmission From Patients
The emergence of SARS in 2002, the importation of monkeypox into the United States in 2003, and the emergence of avian influenza present challenges to the assignment of isolation categories due to conflicting information and uncertainty about possible routes of transmission. Although SARS-CoV is transmitted primarily by contact and/or droplet routes, airborne transmission over a limited distance (eg, within a room) has been suggested, although not proven.134, 135, 136, 137, 138, 139, 140, 141 This is true of other infectious agents as well, such as influenza virus130 and noroviruses.132, 142, 143 Influenza viruses are transmitted primarily by close contact with respiratory droplets,23, 102 and acquisition by HCWs has been prevented by Droplet Precautions, even when positive-pressure rooms were used in one center.144 However, inhalational transmission could not be excluded in an outbreak of influenza in the passengers and crew of an aircraft.130 Observations of a protective effect of ultraviolet light in preventing influenza among patients with tuberculosis during the influenza pandemic of 1957–1958 have been used to suggest airborne transmission.145, 146
In contrast to the strict interpretation of an airborne route for transmission (ie, long distances beyond the patient room environment), short-distance transmission by small-particle aerosols generated under specific circumstances (eg, during endotracheal intubation) to persons in the immediate area near the patient also has been demonstrated. Aerosolized particles < 100 μm in diameter can remain suspended in air when room air current velocities exceed the terminal settling velocities of the particles.109 SARS-CoV transmission has been associated with endotracheal intubation, noninvasive positive pressure ventilation, and cardiopulmonary resuscitation.93, 94, 96, 98, 141 Although the most frequent routes of transmission of noroviruses are contact and foodborne and waterborne routes, several reports suggest that noroviruses also may be transmitted through aerosolization of infectious particles from vomitus or fecal material.142, 143, 147, 148 It is hypothesized that the aerosolized particles are inhaled and subsequently swallowed.
Roy and Milton have proposed a new classification for aerosol transmission when evaluating routes of SARS transmission:
-
•
Obligate. Under natural conditions, disease occurs after transmission of the agent only through inhalation of small-particle aerosols (eg, tuberculosis).
-
•
Preferential. Natural infection results from transmission through multiple routes, but small-particle aerosols are the predominant route (eg, measles, varicella).
-
•
Opportunistic. Under special circumstances, agents that naturally cause disease through other routes may be transmitted through small-particle aerosols.149
This conceptual framework can explain rare occurrences of airborne transmission of agents that are transmitted most frequently by other routes (eg, smallpox, SARS, influenza, noroviruses). Concerns about unknown or possible routes of transmission of agents associated with severe disease and no known treatment often result in the adoption of overextreme prevention strategies, and recommended precautions may change as the epidemiology of an emerging infection becomes more well defined and controversial issues are resolved.
I.B.3.d.ii. Transmission From the Environment
Some airborne infectious agents are derived from the environment and do not usually involve person-to-person transmission; for example, anthrax spores present in a finely milled powdered preparation can be aerosolized from contaminated environmental surfaces and inhaled into the respiratory tract.150, 151 Spores of environmental fungi (eg, Aspergillus spp) are ubiquitous in the environment and may cause disease in immunocompromised patients who inhale aerosolized spores (through, eg, construction dust).152, 153 As a rule, neither of these organisms is subsequently transmitted from infected patients; however, there is 1 well-documented report of person-to-person transmission of Aspergillus sp in the ICU setting that was most likely due to the aerosolization of spores during wound debridement.154 The PE involves isolation practices designed to decrease the risk of exposure to environmental fungal agents in allogeneic HSCT patients.11, 14, 15, 155, 156, 157, 158
Environmental sources of respiratory pathogens (eg, Legionella) transmitted to humans through a common aerosol source is distinct from direct patient-to-patient transmission.
I.B.3.e. Other Sources of Infection
Sources of infection transmission other than infectious individuals include those associated with common environmental sources or vehicles (eg, contaminated food, water, or medications, such as intravenous fluids). Although Aspergillus spp have been recovered from hospital water systems,159 the role of water as a reservoir for immunosuppressed patients remains unclear. Vectorborne transmission of infectious agents from mosquitoes, flies, rats, and other vermin also can occur in health care settings. Prevention of vectorborne transmission is not addressed in this document.
I.C. Infectious Agents of Special Infection Control Interest for Health Care Settings
This section discusses several infectious agents with important infection control implications that either were not discussed extensively in previous isolation guidelines or have emerged only recently. Included are epidemiologically important organisms (eg, C difficile), agents of bioterrorism, prions, SARS-CoV, monkeypox, noroviruses, and the hemorrhagic fever viruses (HFVs). Experience with these agents has broadened the understanding of modes of transmission and effective preventive measures. These agents are included for information purposes and, for some (ie, SARS-CoV, monkeypox), to highlight the lessons that have been learned about preparedness planning and responding effectively to new infectious agents.
I.C.1. Epidemiologically Important Organisms
Under defined conditions, any infectious agent transmitted in a health care setting may become targeted for control because it is epidemiologically important. C difficile is specifically discussed below because of its current prevalence and seriousness in US health care facilities. In determining what constitutes an “epidemiologically important organism,” the following criteria apply:
-
•
A propensity for transmission within health care facilities based on published reports and the occurrence of temporal or geographic clusters of more than 2 patients, (eg, C difficile, norovirus, RSV, influenza, rotavirus, Enterobacter spp, Serratia spp, group A streptococcus). A single case of health care–associated invasive disease caused by certain pathogens (eg, group A streptococcus postoperatively,160 in a burn unit,161 or in a LTCF;162 Legionella spp,14, 163 Aspergillus spp164) is generally considered a trigger for investigation and enhanced control measures because of the risk of additional cases and the severity of illness associated with these infections. Antimicrobial resistance can have the following characteristics:
-
•
Resistance to first-line therapies (eg, MRSA, vancomycin-intermediate/resistannt S aureus [VISA], vancomycin-resistant S aureus [VRSA], VRE, extended-spectrum beta-lactamase [ESBL]-producing organisms)
-
•
Common and uncommon microorganisms with unusual patterns of resistance within a facility (eg, the first isolate of Burkholderia cepacia complex or Ralstonia spp in non-CF patients or a quinolone-resistant strain of P aeruginosa in a facility)
-
•
Difficult to treat because of innate or acquired resistance to multiple classes of antimicrobial agents (eg, Stenotrophomonas maltophilia, Acinetobacter spp)
-
•
Association with serious clinical disease, and increased morbidity and mortality (eg, MRSA and methicillin-susceptible S aureus [MSSA], group A streptococcus)
-
•
A newly discovered or reemerging pathogen.
I.C.1.a. Clostridium difficile
C difficile is a spore-forming gram-positive anaerobic bacillus that was first isolated from stools of neonates in 1935165 and identified as the most frequent causative agent of antibiotic-associated diarrhea and pseudomembranous colitis in 1977.166 This pathogen is a major cause of health care–associated diarrhea and has been responsible for many large outbreaks in health care settings that have proven extremely difficult to control. Important factors contributing to health care–associated outbreaks include environmental contamination, persistence of spores for prolonged periods, resistance of spores to routinely used disinfectants and antiseptics, hand carriage by HCWs to other patients, and exposure of patients to frequent courses of antimicrobial agents.167 Antimicrobials most frequently associated with increased risk of C difficile include third-generation cephalosporins, clindamycin, vancomycin, and fluoroquinolones.
Since 2001, outbreaks and sporadic cases of C difficile with increased morbidity and mortality have occurred in several US states, Canada, England, and the Netherlands.168, 169, 170, 171, 172 The same strain of C difficile has been implicated in all of these outbreaks;173 this strain, toxinotype III, North American pulsed-field gel electrophoresis (PFGE) type 1, and polymerase chain reaction (PCR)-ribotype 027 (NAP1/027), has been found to hyperproduce toxin A (a 16-fold increase) and toxin B (a 23-fold increase) compared with isolates from 12 other PFGE types. A recent survey of US infectious disease physicians found that 40% of the respondents perceived recent increases in the incidence and severity of C difficile disease.174 Standardization of testing methodology and surveillance definitions is needed for accurate comparisons of trends in rates among hospitals.175 It is hypothesized that the incidence of disease and apparent heightened transmissibility of this new strain may be due, at least in part, to the greater production of toxins A and B, increasing the severity of diarrhea and producing more environmental contamination. Considering the greater morbidity, mortality, length of stay, and costs associated with C difficile disease in both acute care and long-term care facilities, control of this pathogen is becoming increasingly important.
Prevention of transmission focuses on syndromic application of Contact Precautions for patients with diarrhea, accurate identification of affected patients, environmental measures (eg, rigorous cleaning of patient rooms), and consistent hand hygiene. Using soap and water rather than alcohol-based handrubs for mechanical removal of spores from hands and using a bleach-containing disinfectant (5000 ppm) for environmental disinfection may be valuable in cases of transmission in health care facilities. Appendix A provides for recommendations.
I.C.1.b. Multidrug-Resistant Organisms
In general, MDROs are defined as microorganisms—predominantly bacteria—that are resistant to 1 or more classes of antimicrobial agents.176 Although the names of certain MDROs suggest resistance to only a single agent (eg, MRSA, VRE), these pathogens are usually resistant to all but a few commercially available antimicrobial agents. This latter feature defines MDROs that are considered to be epidemiologically important and deserve special attention in health care facilities.177 Other MDROs of current concern include multidrug-resistant Streptococcus pneumoniae, which is resistant to penicillin and other broad-spectrum agents such as macrolides and fluroquinolones, multidrug-resistant gram-negative bacilli (MDR-GNB), especially those producing ESBLs; and strains of S aureus that are intermediate or resistant to vancomycin (ie, VISA and VRSA).178, 179, 180, 181, 182, 183, 184, 185, 186, 187, 188, 189, 190, 191, 192, 193, 194, 195, 196, 197, 198
MDROs are transmitted by the same routes as antimicrobial susceptible infectious agents. Patient-to-patient transmission in health care settings, usually via hands of HCWs, has been a major factor accounting for the increase in MDRO incidence and prevalence, especially for MRSA and VRE in acute care facilities.199, 200, 201 Preventing the emergence and transmission of these pathogens requires a comprehensive approach that includes administrative involvement and measures (eg, nurse staffing, communication systems, performance improvement processes to ensure adherence to recommended infection control measures), education and training of medical and other HCWs, judicious antibiotic use, comprehensive surveillance for targeted MDROs, application of infection control precautions during patient care, environmental measures (eg, cleaning and disinfection of the patient care environment and equipment, dedicated single-patient use of noncritical equipment), and decolonization therapy when appropriate.
The prevention and control of MDROs is a national priority, one that requires that all health care facilities and agencies assume responsibility and participate in community-wide control programs.176, 177 A detailed discussion of this topic and recommendations for prevention published in 2006 is available at http://www.cdc.gov/ncidod/dhqp/pdf/ar/mdroGuideline2006.pdf.
I.C.2. Agents of Bioterrorism
The CDC has designated the agents that cause anthrax, smallpox, plague, tularemia, viral hemorrhagic fevers, and botulism as category A (high priority), because these agents can be easily disseminated environmentally and/or transmitted from person to person, can cause high mortality and have the potential for major public health impact, might cause public panic and social disruption, and necessitate special action for public health preparedness.202 General information relevant to infection control in health care settings for Category A agents of bioterrorism is summarized in Table 3. (See http://www.bt.cdc.gov for additional, updated Category A agent information as well as information concerning Category B and C agents of bioterrorism and updates.) Category B and C agents are important but are not as readily disseminated and cause less morbidity and mortality than Category A agents.
Health care facilities confront a different set of issues when dealing with a suspected bioterrorism event compared with other communicable diseases. An understanding of the epidemiology, modes of transmission, and clinical course of each disease, as well as carefully drafted plans that specify an approach and relevant websites and other resources for disease-specific guidance to health care, administrative, and support personnel, are essential for responding to and managing a bioterrorism event. Infection control issues to be addressed include (1) identifying persons who may be exposed or infected; (2) preventing transmission among patients, HCWs, and visitors; (3) providing treatment, chemoprophylaxis, or vaccine to potentially large numbers of people; (4) protecting the environment, including the logistical aspects of securing sufficient numbers of AIIRs or designating areas for patient cohorts when an insufficient number of AIIRs is available; (5) providing adequate quantities of appropriate PPE; and (6) identifying appropriate staff to care for potentially infectious patients (eg, vaccinated HCWs for care of patients with smallpox). The response is likely to differ for exposures resulting from an intentional release compared with a naturally occurring disease because of the large number of persons that can be exposed at the same time and possible differences in pathogenicity.
Various sources offer guidance for the management of persons exposed to the most likely agents of bioterrorism. Federal agency websites (eg, http://www.usamriid.army.mil/publications/index.html and http://www.bt.cdc.gov) and state and county health department websites should be consulted for the most up-to-date information. Sources of information on specific agents include anthrax,203 smallpox,204, 205, 206 plague,207, 208 botulinum toxin,209 tularemia,210 and hemorrhagic fever viruses.211, 212
I.C.2.a. Pre-Event Administration of Smallpox (Vaccinia) Vaccine to Health Care Workers
Vaccination of HCWsl in preparation for a possible smallpox exposure has important infection control implications.213, 214, 215 These include the need for meticulous screening for vaccine contraindications in persons at increased risk for adverse vaccinia events; containment and monitoring of the vaccination site to prevent transmission in the health care setting and at home; and management of patients with vaccinia-related adverse events.216, 217 The pre-event US smallpox vaccination program of 2003 is an example of the effectiveness of carefully developed recommendations for both screening potential vaccinees for contraindications and vaccination site care and monitoring. Between December 2002 and February 2005, approximately 760,000 individuals were vaccinated in the Department of Defense and 40,000 in the civilian or public health populations, including approximately 70,000 who worked in health care settings. No cases of eczema vaccinatum, progressive vaccinia, fetal vaccinia, or contact transfer of vaccinia were reported in health care settings or in military workplaces.218, 219 Outside the health care setting, there were 53 cases of contact transfer from military vaccinees to close personal contacts (eg, bed partners or contacts during participation in sports such as wrestling220). All contact transfers were from individuals who were not following recommendations to cover their vaccination sites. Vaccinia virus was confirmed by culture or PCR in 30 cases, 2 of which resulted from tertiary transfer. All recipients, including 1 breast-fed infant, recovered without complications. Subsequent studies using viral culture and PCR techniques have confirmed the effectiveness of semipermeable dressings to contain vaccinia.221, 222, 223, 224 This experience emphasizes the importance of ensuring that newly vaccinated HCWs adhere to recommended vaccination site care, especially those caring for high-risk patients. Recommendations for pre-event smallpox vaccination of HCWs and vaccinia-related infection control recommendations are published in the Morbidity and Mortality Weekly Report,216, 225 with updates posted on the CDC's bioterrorism website.205
I.C.3. Prions
Creutzfeldt-Jakob disease (CJD) is a rapidly progressive, degenerative neurologic disorder of humans, with an incidence in the United States of approximately 1 person/million population/year.226, 227 CJD is believed to be caused by a transmissible proteinaceous infectious agent known as a prion. Infectious prions are isoforms of a host-encoded glycoprotein known as the prion protein. The incubation period (ie, time between exposure and and onset of symptoms) varies from 2 years to many decades. However, death typically occurs within 1 year of the onset of symptoms. Approximately 85% of CJD cases occur sporadically with no known environmental source of infection, and 10% of cases are familial. Iatrogenic transmission has occurred, with most cases resulting from treatment with human cadaver pituitary-derived growth hormone or gonadotropin,228, 229 from implantation of contaminated human dura mater grafts,230 or from corneal transplants.231 Transmission has been linked to the use of contaminated neurosurgical instruments or stereotactic electroencephalogram electrodes.232, 233, 234, 235
Prion diseases in animals include scrapie in sheep and goats, bovine spongiform encephalopathy (BSE, or “mad cow disease”) in cattle, and chronic wasting disease in deer and elk.236 BSE, first recognized in the United Kingdom in 1986, was associated with a major epidemic among cattle that had consumed contaminated meat and bone meal. The possible transmission of BSE to humans causing variant CJD (vCJD) was first described in 1996 and was subsequently found to be associated with consumption of BSE-contaminated cattle products primarily in the United Kingdom. There is strong epidemiologic and laboratory evidence for a causal association between the causative agent of BSE and vCJD.237 Although most cases of vCJD have been reported from the United Kingdom, a few cases also have been reported from Europe, Japan, Canada, and the United States. Most persons affected with vCJD worldwide lived in or visited the United Kingdom during the years of a large outbreak of BSE (1980–1996) and may have consumed contaminated cattle products during that time (see http://www.cdc.gov/ncidod/diseases/cjd/cjd.htm). Although there has been no indigenously acquired vCJD in the United States, the sporadic occurrence of BSE in cattle in North America has heightened awareness of the possibility that such infections could occur and have led to increased surveillance activities. Updated information may be found at http://www.cdc.gov/ncidod/diseases/cjd/cjd.htm. The public health impact of prion diseases has been reviewed previously.238
vCJD in humans has different clinical and pathologic characteristics than sporadic or classic CJD,239 including (1) younger median age at death (28 [range, 16 to 48] vs 68 years), (2) longer median duration of illness (14 months vs 4 to 6 months), (3) increased frequency of sensory symptoms and early psychiatric symptoms with delayed onset of frank neurologic signs; and (4) detection of prions in tonsillar and other lymphoid tissues, not present in sporadic CJD.240 Similar to sporadic CJD, there have been no reported cases of direct human-to-human transmission of vCJD by casual or environmental contact, droplet, or airborne routes. Ongoing blood safety surveillance in the United States has not detected sporadic CJD transmission through blood transfusion;241, 242, 243 however, bloodborne transmission of vCJD is believed to have occurred in 2 patients in the Uited Kingdom.244, 245 The following FDA websites provide information on steps currently being taken in the United States to protect the blood supply from CJD and vCJD: http://www.fda.gov/cber/gdlns/cjdvcjd.htm and http://www.fda.gov/cber/gdlns/cjdvcjdq&a.htm.
Standard Precautions are used when caring for patients with suspected or confirmed CJD or vCJD. However, special precautions are recommended for tissue handling in the histology laboratory and for conducting an autopsy, embalming, and coming into contact with a body that has undergone autopsy.246 Recommendations for reprocessing surgical instruments to prevent transmission of CJD in health care settings have been published by the World Health Organization (WHO) and are currently under review at the CDC.
Questions may arise concerning notification of patients potentially exposed to CJD or vCJD through contaminated instruments and blood products from patients with CJD or vCJD or at risk of having vCJD. The risk of transmission associated with such exposures is believed to be extremely low but may vary based on the specific circumstance. Therefore, consultation on appropriate options is advised. The United Kingdom has developed several documents that clinicians and patients in the United States may find useful (see http://www.hpa.org.uk/infections/topics_az/cjd/information_documents.htm).
I.C.4. Severe Acute Respiratory Syndrome
SARS is a newly discovered respiratory disease that emerged in China late in 2002 and spread to several countries.135, 140 In particular, mainland China, Hong Kong, Hanoi, Singapore, and Toronto have been significantly affected. SARS is caused by SARS-CoV, a previously unrecognized member of the coronavirus family.247, 248 The incubation period from exposure to the onset of symptoms is typically 2 to 7 days, but can be as long as 10 days and in rare cases even longer.249 The illness is initially difficult to distinguish from other common respiratory infections. Signs and symptoms usually include fever above 38.0°C and chills and rigors, sometimes accompanied by headache, myalgia, and mild to severe respiratory symptoms. A radiographic profile of atypical pneumonia is an important clinical indicator of possible SARS. Compared with adults, children are affected less frequently, have milder disease, and are less likely to transmit SARS-CoV.135, 249, 250, 251 The overall case fatality rate is approximately 6%; underlying disease and advanced age increase the risk of mortality (see http://www.who.int/csr/sarsarchive/2003_05_07a/en/).
Outbreaks in health care settings, with transmission to large numbers of HCWs and patients, haa been a striking feature of SARS; undiagnosed infectious patients and visitors have been important initiators of these outbreaks.21, 252, 253, 254 The relative contribution of potential modes of transmission is not known precisely. There is ample evidence for droplet and contact transmission;96, 101, 113 however, opportunistic airborne transmission cannot be excluded.101, 135, 136, 137, 138, 139, 149, 254 For example, exposure to aerosol-generating procedures (eg, endotracheal intubation, suctioning) has been associated with transmission of infection to large numbers of HCWs outside of the United States.93, 94, 96, 98, 253 Therefore, aerosolization of small infectious particles generated during these and other similar procedures could be a risk factor for transmission to others within a multibed room or shared airspace. A review of the infection control literature generated from the SARS outbreaks of 2003 concluded that the greatest risk of transmission is to those who have close contact, are not properly trained in use of protective infection control procedures, and do not consistently use PPE, and that N95 or higher-level respirators may offer additional protection to those exposed to aerosol-generating procedures and high-risk activities.255, 256 Organizational and individual factors that affect adherence to infection control practices for SARS also were identified.256
Control of SARS requires a coordinated, dynamic response by multiple disciplines in a health care setting.9 Early detection of cases is accomplished by screening persons with symptoms of a respiratory infection for history of travel to areas experiencing community transmission or contact with SARS patients, followed by implementation of respiratory hygiene/cough etiquette (ie, placing a mask over the patient's nose and mouth) and physical separation from other patients in common waiting areas. The precise combination of precautions to protect HCWs has not yet been determined. At the time of this publication, the CDC recommends Standard Precautions, with emphasis on the use of hand hygiene; Contact Precautions, with emphasis on environmental cleaning due to the detection of SARS-CoV RNA by PCR on surfaces in rooms occupied by SARS patients;138, 254, 257 and Airborne Precautions, including use of fit-tested NIOSH-approved N95 or higher-level respirators and eye protection.258 In Hong Kong, the use of Droplet and Contact Precautions, including the use of a mask but not a respirator, was effective in protecting HCWs.113 However, in Toronto, consistent use of an N95 respirator was found to be slightly more protective than a mask.93 It is noteworthy that no transmission of SARS-CoV to public hospital workers occurred in Vietnam despite inconsistent use of infection control measures, including use of PPE, which suggests other factors (eg, severity of disease, frequency of high-risk procedures or events, environmental features) may influence opportunities for transmission.259
SARS-CoV also has been transmitted in the laboratory setting through breaches in recommended laboratory practices. Research laboratories in which SARS-CoV was under investigation were the source of most cases reported after the first series of outbreaks in the winter and spring of 2003.260, 261 Studies of the SARS outbreaks of 2003 and transmissions occurring in the laboratory reaffirm the effectiveness of recommended infection control precautions and highlight the importance of consistent adherence to these measures.
Lessons learned from the SARS outbreaks are useful in devising plans to respond to future public health crises, such as pandemic influenza and bioterrorism events. Surveillance for cases among patients and HCWs, ensuring availability of adequate supplies and staffing, and limiting access to health care facilities were important factors in the response to SARS.9 Guidance for infection control precautions in various settings is available at http://www.cdc.gov/ncidod/sars.
I.C.5. Monkeypox
Monkeypox is a rare viral disease found mostly in the rain forest countries of Central and West Africa. The disease is caused by an orthopoxvirus that is similar in appearance to smallpox but causes a milder disease. The only recognized outbreak of human monkeypox in the United States was detected in June 2003, after several people became ill after contact with sick pet prairie dogs. Infection in the prairie dogs was subsequently traced to their contact with a shipment of animals from Africa, including giant Gambian rats.262 This outbreak demonstrates the importance of recognition and prompt reporting of unusual disease presentations by clinicians to enable prompt identification of the etiology, as well as the potential of epizootic diseases to spread from animal reservoirs to humans through personal and occupational exposure.263
Only limited data on transmission of monkeypox are available. Transmission from infected animals and humans is believed to occur primarily through direct contact with lesions and respiratory secretions; airborne transmission from animals to humans is unlikely but cannot be excluded, and may have occurred in veterinary practices (eg, during administration of nebulized medications to ill prairie dogs264). In humans, 4 instances of monkeypox transmission in hospitals have been reported in Africa among children, usually related to sharing the same ward or bed.265, 266 Additional recent literature documents transmission of Congo Basin monkeypox in a hospital compound for an extended number of generations.267
There has been no evidence of airborne or any other person-to-person transmission of monkeypox in the United States, and no new cases of monkeypox have been identified since the outbreak in June 2003.268 The outbreak strain is a clade of monkeypox distinct from the Congo Basin clade and may have different epidemiologic properties (including human-to-human transmission potential) from monkeypox strains of the Congo Basin;269 this awaits further study. Smallpox vaccine is 85% protective against Congo Basin monkeypox.270 Because there is an associated case fatality rate of < 10%, administration of smallpox vaccine within 4 days to individuals who have had direct exposure to patients or animals with monkeypox is a reasonable policy.271 For the most current information on monkeypox, see http://www.cdc.gov/ncidod/monkeypox/clinicians.htm.
I.C.6. Noroviruses
Noroviruses, formerly referred to as Norwalk-like viruses, are members of the Caliciviridae family. These agents are transmitted via contaminated food or water and from person to person, causing explosive outbreaks of gastrointestinal disease.272 Environmental contamination also has been documented as a contributing factor in ongoing transmission during outbreaks.273, 274 Although noroviruses cannot be propagated in cell culture, DNA detection by molecular diagnostic techniques has brought a greater appreciation of their role in outbreaks of gastrointestinal disease.275 Reported outbreaks in hospitals,132, 142, 276 nursing homes,274, 277, 278, 279, 280, 281, 282 cruise ships,283, 284, hotels,143, 147 schools,148 and large crowded shelters established for hurricane evacuees285 has demonstrated their highly contagious nature, their potentially disruptive impact in health care facilities and the community, and the difficulty of controlling outbreaks in settings in which people share common facilites and space. Of note, there is nearly a 5-fold increase in the risk to patients in outbreaks when a patient is the index case compared with exposure of patients during outbreaks when a staff member is the index case.286
The average incubation period for gastroenteritis caused by noroviruses is 12 to 48 hours, and the clinical course lasts 12 to 60 hours.272 Illness is characterized by acute onset of nausea, vomiting, abdominal cramps, and/or diarrhea. The disease is largely self-limited; rarely, death due to severe dehydration can occur, particularly in elderly persons with debilitating health conditions.
The epidemiology of norovirus outbreaks shows that even though primary cases may result from exposure to a fecally contaminated food or water, secondary and tertiary cases often result from person-to-person transmission facilitated by contamination of fomites272, 287 and dissemination of infectious particles, especially during the process of vomiting.132, 142, 143, 147, 148, 272, 278, 279 Widespread, persistent, and inapparent contamination of the environment and fomites can make outbreaks extremely difficult to control.147, 274, 283 These clinical observations and the detection of norovirus DNA on horizontal surfaces 5 feet above the level that might be touched normally suggest that under certain circumstances, aerosolized particles may travel distances beyond 3 feet.147 It is hypothesized that infectious particles may be aerosolized from vomitus, inhaled, and swallowed. In addition, individuals who are responsible for cleaning the environment may be at increased risk of infection. Development of disease and transmission may be facilitated by the low infectious dose (ie, < 100 viral particles)288 and the resistance of these viruses to the usual cleaning and disinfection agents (ie, they may survive < 10 ppm chlorine).289, 290, 291 An alternate phenolic agent that was shown to be effective against feline calicivirus was used for environmental cleaning in one outbreak.275, 292 There are insufficient data to determine the efficacy of alcohol-based hand rubs against noroviruses when the hands are not visibly soiled.293 Absence of disease in certain individuals during an outbreak may be explained by protection from infection conferred by the B histo-blood group antigen.294 Consultation on outbreaks of gastroenteritis is available through the CDC's Division of Viral and Rickettsial Diseases.295
I.C.7. Hemorrhagic Fever Viruses
HFV is a mixed group of viruses that cause serious disease with high fever, skin rash, bleeding diathesis, and, in some cases, high mortality; the resulting disease is referred to as viral hemorrhagic fever (VHF). Among the more commonly known HFVs are Ebola and Marburg viruses (Filoviridae), Lassa virus (Arenaviridae), Crimean-Congo hemorrhagic fever and Rift Valley Fever virus (Bunyaviridae), and Dengue and Yellow fever viruses (Flaviviridae).212, 296 These viruses are transmitted to humans through contact with infected animals or via arthropod vectors. Although none of these viruses is endemic in the United States, outbreaks in affected countries provide potential opportunities for importation by infected humans and animals. Furthermore, there is a concern that some of these agents could be used as bioweapons.212 Person-to-person transmission has been documented for Ebola, Marburg, Lassa, and Crimean-Congo HFVs. In resource-limited health care settings, transmission of these agents to HCWs, patients, and visitors has been described and in some outbreaks has accounted for a large proportion of cases.297, 298, 299 Transmission within households also has been documented in individuals who had direct contact with ill persons or their body fluids, but not in those who did not have such contact.300
Evidence concerning the transmission of HFVs has been summarized previously.212, 301 Person-to-person transmission is associated primarily with direct blood and body fluid contact. Percutaneous exposure to contaminated blood carries a particularly high risk for transmission and increased mortality.302, 303 The finding of large numbers of Ebola viral particles in the skin and the lumina of sweat glands has raised concerns that transmission could occur from direct contact with intact skin, although epidemiologic evidence to support this is lacking.304 Postmortem handling of infected bodies is an important risk for transmission.300, 305, 306 In rare situations, cases in which the mode of transmission was unexplained among individuals with no known direct contact have led to speculation that airborne transmission could have occurred.297 However, airborne transmission of naturally occurring HFVs in humans has not been documented. A study of airplane passengers exposed to an in-flight index case of Lassa fever found no transmission to any passengers.307
In the laboratory setting, animals have been infected experimentally with Marburg or Ebola virus through direct inoculation of the nose, mouth, and/or conjunctiva308, 309 and by using mechanically generated virus-containing aerosols.310, 311 Transmission of Ebola virus among laboratory primates in an animal facility has been described.312 The secondarily infected animals were in individual cages separated by approximately 3 meters. Although the possibility of airborne transmission was suggested, the investigators were not able to exclude droplet or indirect contact transmission in this incidental observation.
Guidance on infection control precautions for HVFs transmitted person-to-person have been published by the CDC1, 211 and by the Johns Hopkins Center for Civilian Biodefense Strategies.212 The most recent recommendations at the time of publication of this document were posted on the CDC website on May 19, 2005.313 Inconsistencies among the various recommendations have raised questions about the appropriate precautions to use in US hospitals. In less developed countries, outbreaks of HFVs have been controlled with basic hygiene, barrier precautions, safe injection practices, and safe burial practices.298, 305 The preponderance of evidence on HFV transmission indicates that Standard, Contact, and Droplet Precautions with eye protection are effective in protecting HCWs and visitors coming in contact with an infected patient. Single gloves are adequate for routine patient care; double-gloving is advised during invasive procedures (eg, surgery) that pose an increased risk of blood exposure. Routine eye protection (ie goggles or face shield) is particularly important. Fluid-resistant gowns should be worn for all patient contact. Airborne Precautions are not required for routine patient care; however, use of AIIRs is prudent when procedures that could generate infectious aerosols are performed (eg, endotracheal intubation, bronchoscopy, suctioning, autopsy procedures involving oscillating saws). N95 or higher-level respirators may provide added protection for individuals in a room during aerosol-generating procedures (Table 3, Appendix A). When a patient with a syndrome consistent with hemorrhagic fever also has a history of travel to an endemic area, precautions are initiated on presentation and then modified as more information is obtained (Table 2). Patients with hemorrhagic fever syndrome in the setting of a suspected bioweapons attack should be managed using Airborne Precautions, including AIIRs, because the epidemiology of a potentially weaponized hemorrhagic fever virus is unpredictable.
I.D. Transmission Risks Associated With Specific Types of Health Care Settings
Numerous factors influence differences in transmission risks among the various health care settings. These factors include the population characteristics (eg, increased susceptibility to infections, type and prevalence of indwelling devices), intensity of care, exposure to environmental sources, length of stay, and frequency of interaction between patients/residents with each other and with HCWs. These factors, as well as organizational priorities, goals, and resources, influence how different health care settings adapt transmission prevention guidelines to meet their specific needs.314, 315 Infection control management decisions are informed by data regarding institutional experience/Epidemiology; trends in community and institutional HAIs; local, regional, and national Epidemiology; and emerging infectious disease threats.
I.D.1. Hospitals
Infection transmission risks are present in all hospital settings. However, certain hospital settings and patient populations have unique conditions that predispose patients to infection and merit special mention. These are often sentinel sites for the emergence of new transmission risks that may be unique to that setting or present opportunities for transmission to other settings in the hospital.
I.D.1.a. Intensive Care Units
Intensive care units (ICUs) serve patients who are immunocompromised by disease state and/or by treatment modalities, as well as patients with major trauma, respiratory failure, and other life-threatening conditions (eg, myocardial infarction, congestive heart failure, overdose, stroke, gastrointestinal bleeding, renal failure, hepatic failure, multiorgan system failure, and extremes of age). Although ICUs account for a relatively small proportion of hospitalized patients, infections acquired in these units account for > 20% of all HAIs.316 In the National Nosocomial Infection Surveillance (NNIS) system, 26.6% of HAIs were reported from ICU and high-risk nursery (neonatal ICU [NICU]) patients in 2002 (NNIS, unpublished data). This patient population has increased susceptibility to colonization and infection, especially with MDROs and Candida spp,317, 318 because of underlying diseases and conditions, the invasive medical devices and technology used in their care (eg central venous catheters and other intravascular devices, mechanical ventilators, extracorporeal membrane oxygenation, hemodialysis/filtration, pacemakers, implantable left-ventricular assist devices), the frequency of contact with HCWs, prolonged lengths of stay, and prolonged exposure to antimicrobial agents.319, 320, 321, 322, 323, 324, 325, 326, 327, 328, 329, 330 Furthermore, adverse patient outcomes in this setting are more severe and are associated with a higher mortality.331 Outbreaks associated with various bacterial, fungal, and viral pathogens due to common-source and person-to-person transmissions are frequent in adult ICUs and pediatric ICUs (PICUs).31, 332, 333, 334, 335, 336, 337
I.D.1.b. Burn Units
Burn wounds can provide optimal conditions for colonization, infection, and transmission of pathogens; infection acquired by burn patients is a frequent cause of morbidity and mortality.319, 338, 339 The risk of invasive burn wound infection is particularly high in patients with a burn injury involving > 30% of the total body surface area (TBSA).340, 341 Infections occurring in patients with burn injuries involving < 30% of the TBSA are usually associated with the use of invasive devices. MSSA, MRSA, enterococci (including VRE), gram-negative bacteria, and Candida spp are prevalent pathogens in burn infections,53, 339, 342, 343, 344, 345, 346, 347, 348, 349 and outbreaks of these organisms have been reported.350, 351, 352, 353 Shifts over time in the predominance of pathogens causing infections in burn patients often lead to changes in burn care practices.342, 354, 355, 356, 357 Burn wound infections caused by Aspergillus spp or other environmental molds may result from exposure to supplies contaminated during construction358 or to dust generated during construction or other environmental disruption.359
Hydrotherapy equipment is an important environmental reservoir of gram-negative organisms. Its use in burn care is discouraged based on demonstrated associations between the use of contaminated hydrotherapy equipment and infections. Burn wound infections and colonization, as well as bloodstream infections, caused by multidrug-resistant P aeruginosa,360 Acinetobacter baumannii,361 and MRSA351 have been associated with hydrotherapy; thus, excision of burn wounds in operating rooms is the preferred approach.
Advances in burn care (specifically, early excision and grafting of the burn wound, use of topical antimicrobial agents, and institution of early enteral feeding) have led to decreased infectious complications. Other advances have included prophylactic antimicrobial use, selective digestive decontamination, and use of antimicrobial-coated catheters; however, few epidemiologic studies and no efficacy studies have been performed to investigate the relative benefit of these measures.356
There is no consensus on the most effective infection control practices to prevent transmission of infections to and from patients with serious burns (eg, single-bed rooms,357 laminar flow,362 and high-efficiency particulate air [HEPA] filtration,359 or maintaining burn patients in a separate unit with no exposure to patients or equipment from other units363). There also is controversy regarding the need for and type of barrier precautions in the routine care of burn patients. One retrospective study demonstrated the efficacy and cost-effectiveness of a simplified barrier isolation protocol for wound colonization, emphasizing handwashing and use of gloves, caps, masks, and impermeable plastic aprons (rather than isolation gowns) for direct patient contact.364 However, to date no studies have determined the most effective combination of infection control precautions for use in burn settings. Prospective studies in this area are needed.
I.D.1.c. Pediatrics
Studies of the epidemiology of HAIs in children have identified unique infection control issues in this population.63, 64, 365, 366, 367, 368, 369 Pediatric ICU patients and the lowest birth weight babies in the NICU monitored in the NNIS system have had high rates of central venous catheter–associated bloodstream infections.64, 319, 368, 369, 370, 371 In addition, there is a high prevalence of community-acquired infections among hospitalized infants and young children who have not yet become immune either by vaccination or by natural infection. This results in more patients and their sibling visitors with transmissible infections in pediatric health care settings, especially during seasonal epidemics (eg, pertussis;36, 40, 41 respiratory viral infections. including those caused by RSV,24 influenza viruses,372 parainfluenza virus,373 human metapneumovirus,374 and adenoviruses;375 rubeola [measles];34 varicella [chickenpox];376 and rotavirus38, 377).
Close physical contact between HCWs and infants and young children (eg. cuddling, feeding, playing, changing soiled diapers, and cleaning copious uncontrolled respiratory secretions) provides abundant opportunities for transmission of infectious material. Such practices and behaviors as congregation of children in play areas where toys and bodily secretions are easily shared and rooming-in of family members with pediatric patients can further increase the risk of transmission. Pathogenic bacteria have been recovered from toys used by hospitalized patients;378 contaminated bath toys were implicated in an outbreak of multidrug-resistant P. aeruginosa on a pediatric oncology unit.80 In addition, several patient factors increase the likelihood that infection will result from exposure to pathogens in health care settings (eg, immaturity of the neonatal immune system, lack of previous natural infection and resulting immunity, prevalence of patients with congenital or acquired immune deficiencies, congenital anatomic anomalies, and use of life-saving invasive devices in NICUs and PICUs).63 There are theoretical concerns that infection risk will increase in association with innovative practices used in the NICU for the purpose of improving developmental outcomes, Such factors include co-bedding379 and kangaroo care,380 which may increase opportunity for skin-to-skin exposure of multiple gestation infants to each other and to their mothers, respectively; although the risk of infection actually may be reduced among infants receiving kangaroo care.381 Children who attend child care centers382, 383 and pediatric rehabilitation units384 may increase the overall burden of antimicrobial resistance by contributing to the reservoir of CA-MRSA.385, 386, 387, 388, 389, 390 Patients in chronic care facilities may have increased rates of colonization with resistant garm-negative bacilli and may be sources of introduction of resistant organisms to acute care settings.50
I.D.2. Nonacute Health Care Settings
Health care is provided in various settings outside of hospitals, including long-term care facilities (LTCFs) (eg nursing homes), homes for the developmentally disabled, behavioral health service settings, rehabilitation centers, and hospices.391 In addition, health care may be provided in non–health care settings, such as workplaces with occupational health clinics, adult day care centers, assisted-living facilities, homeless shelters, jails and prisons, school clinics, and infirmaries. Each of these settings has unique circumstances and population risks that must be considered when designing and implementing an infection control program. Several of the most common settings and their particular challenges are discussed below. Although this guideline does not address each setting, the principles and strategies provided herein may be adapted and applied as appropriate.
I.D.2.a. Long-Term Care
The designation LTCF applies to a diverse group of residential settings, ranging from institutions for the developmentally disabled to nursing homes for the elderly and pediatric chronic care facilities.392, 393, 394 Nursing homes for the elderly predominate numerically and frequently represent long-term care as a group of facilities. Approximately 1.8 million Americans reside in the nation's 16,500 nursing homes.395 Estimates of HAI rates of 1.8 to 13.5 per 1000 resident-care days have been reported, with a range of 3 to 7 per 1000 resident-care days in the more rigorous studies.396, 397, 398, 399, 400 The infrastructure described in the Department of Veterans Affairs' nursing home care units is a promising example for the development of a nationwide HAI surveillance system for LTCFs.401
LCTFs are different from other health care settings in that elderly patients at increased risk for infection are brought together in one setting and remain in the facility for extended periods; for most residents, it is their home. An atmosphere of community is fostered, and residents share common eating and living areas and participate in various facility-sponsored activities.402, 403 Because able residents interact freely with each other, controlling infection transmission in this setting can be challenging.404 A residents who is colonized or infected with certain microorganisms are in some cases restricted to his or her room. However, because of the psychosocial risks associated with such restriction, balancing psychosocial needs with infection control needs is important in the LTCF setting.405, 406, 407, 408 Documented LTCF outbreaks have been caused by various viruses (eg, influenza virus,35, 409, 410, 411 rhinovirus,412 adenovirus [conjunctivitis],413 norovirus274, 277, 278, 280) and bacteria, including group A streptococcus,162, B pertussis,414 nonsusceptible S pneumoniae,197, 198 other MDROs, and C difficile 415). These pathogens can lead to substantial morbidity and mortality, as well as increased medical costs; prompt detection and implementation of effective control measures are needed.
Risk factors for infection are prevalent among LTCF residents.394, 416, 417 Age-related declines in immunity may affect the response to immunizations for influenza and other infectious agents and increase the susceptibility to tuberculosis. Immobility, incontinence, dysphagia, underlying chronic diseases, poor functional status, and age-related skin changes increase susceptibility to urinary, respiratory, and cutaneous and soft tissue infections, whereas malnutrition can impair wound healing.418, 419, 420, 421, 422 Medications (eg, drugs that affect level of consciousness, immune function, gastric acid secretions, and normal flora, including antimicrobial therapy) and invasive devices (eg, urinary catheters and feeding tubes) heighten the susceptibility to infection and colonization in LTCF residents.423, 424, 425 Finally, limited functional status and total dependence on HCWs for activities of daily living have been identified as independent risk factors for infection400, 416, 426 and for colonization with MRSA427, 428 and ESBL-producing Klebsiella pneumoniae.429 Several position papers and review articles provide guidance on various aspects of infection control and antimicrobial resistance in LTCFs.405, 406, 407, 430, 431, 432, 433, 434, 435 The Centers for Medicare and Medicaid Services has established regulations for the prevention of infection in LTCFs.436
Because residents of LTCFs are hospitalized frequently, they can transfer pathogens between LTCFs and health care facilities in which they receive care.8, 437, 438, 439, 440 This also is true for pediatric long-term care populations. Pediatric chronic care facilities have been associated with the importation of extended-spectrum cephalosporin-resistant, gram-negative bacilli into a PICU.50 Children from pediatric rehabilitation units may contribute to the reservoir of community-associated MRSA.384, 388, 389, 390
I.D.2.b. Ambulatory Care
Over the past decade, health care delivery in the United States has shifted from the acute, inpatient hospital to various ambulatory and community-based settings, including the home. Ambulatory care is provided in hospital-based outpatient clinics, nonhospital-based clinics and physicians' offices, public health clinics, free-standing dialysis centers, ambulatory surgical centers, urgent care centers, and other setting. In 2000, there were 83 million visits to hospital outpatient clinics and more than 823 million visits to physicians' offices;441 ambulatory care now accounts for most patient encounters with the health care system.442 Adapting transmission prevention guidelines to these settings is challenging, because patients remain in common areas for prolonged periods waiting to be seen by a health care provider or awaiting admission to the hospital, examination or treatment rooms are turned around quickly with limited cleaning, and infectious patients may not be recognized immediately. Furthermore, immunocompromised patients often receive chemotherapy in infusion rooms, where they stay for extended periods along with other types of patients.
Little data exist on the risk of HAIs in ambulatory care settings, with the exception of hemodialysis centers.18, 443, 444 Transmission of infections in outpatient settings has been reviewed in 3 studies.445, 446, 447 Goodman and Solomon445 summarized 53 clusters of infections associated with the outpatient setting between 1961 and 1990. Overall, 29 clusters were associated with common source transmission from contaminated solutions or equipment, 14 were associated with person-to-person transmission from or involving HCWs, and 10 were associated with airborne or droplet transmission among patients and health care workers. Transmission of bloodborne pathogens (ie, HBV, HCV, and, rarely, HIV) in outbreaks, sometimes involving hundreds of patients, continues to occur in ambulatory settings. These outbreaks often are related to common source exposures, usually a contaminated medical device, multidose vial, or intravenous solution.82, 448, 449, 450, 451, 452 In all cases, transmission has been attributed to failure to adhere to fundamental infection control principles, including safe injection practices and aseptic technique. This subject has been reviewed, and recommended infection control and safe injection practices have been summarized.453
Airborne transmission of M tuberculosis and measles in ambulatory settings, most often emergency departments, has been reported.34, 127, 445, 447, 454, 455, 456 Measles virus was transmitted in physicians' offices and other outpatient settings during an era when immunization rates were low and measles outbreaks in the community were occurring regularly.34, 122, 457 Rubella has been transmitted in the outpatient obstetric setting;33 there are no published reports of varicella transmission in the outpatient setting. In the ophthalmology setting, adenovirus type 8 epidemic keratoconjunctivitis has been transmitted through incompletely disinfected ophthalmology equipment and/or from HCWs to patients, presumably by contaminated hands.17, 445, 447, 458, 459, 460, 461
Preventing transmission in outpatient settings necessitates screening for potentially infectious symptomatic and asymptomatic individuals, especially those at possible risk for transmitting airborne infectious agents (eg, M tuberculosis, varicella-zoster virus, rubeola [measles]), at the start of the initial patient encounter. On identification of a potentially infectious patient, implementation of prevention measures, including prompt separation of potentially infectious patients and implementation of appropriate control measures (eg, respiratory hygiene/cough etiquette and Transmission-Based Precautions) can decrease transmission risks.9, 12 Transmission of MRSA and VRE in outpatient settings has not been reported, but the association of CA-MRSA in HCWs working in an outpatient HIV clinic with environmental CA-MRSA contamination in that clinic suggests the possibility of transmission in that setting.462 Patient-to-patient transmission of Burkholderia spp and P aeruginosa in outpatient clinics for adults and children with cystic fibrosis has been confirmed.463, 464
I.D.2.c. Home Care
Home care in the United States is delivered by more than 20,000 provider agencies, including home health agencies, hospices, durable medical equipment providers, home infusion therapy services, and personal care and support services providers. Home care is provided to patients of all ages with both acute and chronic conditions. The scope of services ranges from assistance with activities of daily living and physical and occupational therapy to the care of wounds, infusion therapy, and chronic ambulatory peritoneal dialysis.
The incidence of infection in home care patients, other than that associated with infusion therapy, has not been well studied.465, 466, 467, 468, 469, 470 However, data collection and calculation of infection rates have been done for central venous catheter–associated bloodstream infections in patients receiving home infusion therapy469, 470, 471, 472, 473 and for the risk of blood contact through percutaneous or mucosal exposures, demonstrating that surveillance can be performed in this setting.474 Draft definitions for home care–associated infections have been developed.475
Transmission risks during home care are presumed to be minimal. The main transmission risks to home care patients are from an infectious home care provider or contaminated equipment; a provider also can be exposed to an infectious patient during home visits. Because home care involves patient care by a limited number of personnel in settings without multiple patients or shared equipment, the potential reservoir of pathogens is reduced. Infections of home care providers that could pose a risk to home care patients include infections transmitted by the airborne or droplet routes (eg, chickenpox, tuberculosis, influenza), skin infestations (eg, scabies69 and lice), and infections transmitted by direct or indirect contact (eg, impetigo). There are no published data on indirect transmission of MDROs from one home care patient to another, although this is theoretically possible if contaminated equipment is transported from an infected or colonized patient and used on another patient. Of note, investigations of the first case of VISA in home care186 and the first 2 reported cases of VRSA178, 180, 181, 183 found no evidence of transmission of VISA or VRSA to other home care recipients. Home health care also may contribute to antimicrobial resistance; a review of outpatient vancomycin use found that 39% of recipients did not receive prescribed antibiotics according to recommended guidelines.476
Although most home care agencies implement policies and procedures aimed at preventing transmission of organisms, the current approach is based on the adaptation of the 1996 Guideline for Isolation Precautions in Hospitals,1 as well as other professional guidance.477, 478 This issue has proven very challenging to the home care industry, and practice has been inconsistent and frequently not evidence-based. For example, many home health agencies continue to observe “nursing bag technique,” a practice that prescribes the use of barriers between the nursing bag and environmental surfaces in the home.479 Although the home environment may not always appear clean, the use of barriers between 2 noncritical surfaces has been questioned.480, 481 Opportunites exist to conduct research in home care related to infection transmission risks.482
I.D.2.d. Other Sites of Health Care Delivery
Facilities that are not primarily health care settings but in which health care is delivered include clinics in correctional facilities and shelters. Both of these settings can have suboptimal features, such as crowded conditions and poor ventilation. Economically disadvantaged individuals who may have chronic illnesses and health care problems related to alcoholism, injected drug use, poor nutrition, and/or inadequate shelter often receive their primary health care at such sites.483 Infectious diseases of special concern for transmission include tuberculosis, scabies, respiratory infections (eg, N meningitides, S pneumoniae), sexually transmitted and bloodborne diseases (eg, HIV, HBV, HCV, syphilis, gonorrhea), hepatitis A virus, diarrheal agents such as norovirus, and foodborne diseases.285, 484, 485, 486, 487 A high index of suspicion for tuberculosis and CA-MRSA in these populations is needed; outbreaks in these settings or among the populations they serve have been reported.488, 489, 490, 491, 492, 493, 494, 495, 496
Patient encounters in these types of facilities provide an opportunity to deliver recommended immunizations and screen for M tuberculosis infection, along with diagnosing and treating acute illnesses.497 Recommended infection control measures in these nontraditional areas designated for health care delivery are the same as for other ambulatory care settings. Therefore, these settings must be equipped to observe Standard Precautions and, when indicated, Transmission-Based Precautions.
I.E. Transmission Risks Associated With Special Patient Populations
As new treatments emerge for complex diseases, unique infection control challenges associated with special patient populations must be addressed.
I.E.1. Immunocompromised Patients
Patients who have congenital primary immune deficiencies or acquired disease (eg. treatment-induced immune deficiencies) are at increased risk for numerous types of infections while receiving health care; these patients may be located throughout the health care facility. The specific immune system defects determine the types of infections most likely to be acquired (eg, viral infections are associated with T cell defects, and fungal and bacterial infections occur in patients who are neutropenic). As a general group, immunocompromised patients can be cared for in the same environment as other patients; however, it is always advisable to minimize exposure to other patients with transmissible infections, such as influenza and other respiratory viruses.498, 499 The use of more intense chemotherapy regimens for treatment of childhood leukemia may be associated with prolonged periods of neutropenia and suppression of other components of the immune system, extending the period of infection risk and raising the concern that additional precautions may be indicated for select groups.500, 501 With the application of newer and more intense immunosuppressive therapies for various medical conditions (eg, rheumatologic disease,502, 503 inflammatory bowel disease504), immunosuppressed patients are likely to be more widely distributed throughout a health care facility rather than localized to single patient units (eg, hematology-oncology). Guidelines for preventing infections in certain groups of immunocompromised patients have been published previously.15, 505, 506
Published data provide evidence to support placing patients undergoing allogeneic HSCT in a PE.15, 157, 158 In addition, guidelines have been developed that address the special requirements of these immunocompromised patients, including use of antimicrobial prophylaxis and engineering controls to create a PE for the prevention of infections caused by Aspergillus spp and other environmental fungi.11, 14, 15 As more intense chemotherapy regimens associated with prolonged periods of neutropenia or graft-versus-host disease are implemented, the period of risk and duration of environmental protection may need to be prolonged beyond the traditional 100 days.507
I.E.2. Cystic Fibrosis Patients
Patients with cystic fibrosis (CF) require special consideration when developing infection control guidelines. Compared with other patients, CF patients require additional protection to prevent transmission from contaminated respiratory therapy equipment.508, 509, 510, 511, 512 Such infectious agents as B cepacia complex and P aeruginosa.463, 464, 513, 514 have unique clinical and prognostic significance. In CF patients, B cepacia infection has been associated with increased morbidity and mortality,515, 516, 517 whereas delayed acquisition of chronic P aeruginosa infection may be associated with an improved long-term clinical outcome.518, 519
Person-to-person transmission of B cepacia complex has been demonstrated among children516 and adults520 with CF in health care settings463, 521 and from various social contacts,522 most notably attendance at camps for patients with CF523 and among siblings with CF.524 Successful infection control measures used to prevent transmission of respiratory secretions include segregation of CF patients from each other in ambulatory and hospital settings (including use of private rooms with separate showers), environmental decontamination of surfaces and equipment contaminated with respiratory secretions, elimination of group chest physiotherapy sessions, and disbanding of CF camps.97, 525 The Cystic Fibrosis Foundation has published a consensus document with evidence-based recommendations for infection control practices in CF patients.20
I.F. New Therapies Associated With Potentially Transmissible Infectious Agents
I.F.1. Gene Therapy
Gene therapy has has been attempted using various viral vectors, including nonreplicating retroviruses, adenoviruses, adeno-associated viruses, and replication-competent strains of poxviruses. Unexpected adverse events have restricted the prevalence of gene therapy protocols.
The infectious hazards of gene therapy are theoretical at this time but require meticulous surveillance due to the possible occurrence of in vivo recombination and the subsequent emergence of a transmissible genetically altered pathogen. The greatest concern attends the use of replication-competent viruses, especially vaccinia. To date, no reports have described transmission of a vector virus from a gene therapy recipient to another individual, but surveillance is ongoing. Recommendations for monitoring infection control issues throughout the course of gene therapy trials have been published.526, 527, 528
I.F.2. Infections Transmitted Through Blood, Organs, and Other Tissues
The potential hazard of transmitting infectious pathogens through biologic products is a small but ever-present risk, despite donor screening. Reported infections transmitted by transfusion or transplantation include West Nile virus infection,529 cytomegalovirus infection,530 CJD,230 hepatitis C,531 infections with Clostridium spp532 and group A streptococcus,533 malaria,534 babesiosis,535 Chagas disease,536 lymphocytic choriomeningitis,537 and rabies.538, 539 Therefore, it is important to consider receipt of biologic products when evaluating patients for potential sources of infection.
I.F.3. Xenotransplantation
Transplantation of nonhuman cells, tissues, and organs into humans potentially exposes patients to zoonotic pathogens. Transmission of known zoonotic infections (eg, trichinosis from porcine tissue) is of concern. Also of concern is the possibility that transplantation of nonhuman cells, tissues, or organs may transmit previously unknown zoonotic infections (xenozoonoses) to immunosuppressed human recipients. Potential infections that potentially could accompany transplantation of porcine organs have been described previously.540 Guidelines from the US Public Health Service address many infectious diseases and infection control issues that surround the developing field of xenotransplantation;541 work in this area is ongoing.
Part II: Fundamental Elements Needed to Prevent Transmission of Infectious Agents in Health Care Settings
II.A. Health Care System Components That Influence the Effectiveness of Precautions to Prevent Transmission
II.A.1. Administrative Measures
Health care organizations can demonstrate a commitment to preventing transmission of infectious agents by incorporating infection control into the objectives of the organization's patient and occupational safety programs.542, 543, 544, 545, 546 An infrastructure designed to guide, support, and monitor adherence to Standard Precautions and Transmission-Based Precautions434, 547, 548 will facilitate fulfillment of the organization's mission and achievement of the Joint Commission on Accreditation of Health Care Organizations' patient safety goal to decrease HAIs.549 Policies and procedures that explain how Standard Precautions and Transmission-Based Precautions are applied, including systems used to identify and communicate information on patients with potentially transmissible infectious agents, are essential to ensure the success of these measures. These policies and procedures may vary according to the characteristics of the organization.
A key administrative measure is the provision of fiscal and human resources for maintaining infection control and occupational health programs that are responsive to emerging needs. Specific components include bedside nurse550 and infection prevention and control professional (ICP) staffing levels,551 inclusion of ICPs in facility construction and design decisions,11 clinical microbiology laboratory support,552, 553 adequate supplies and equipment including facility ventilation systems,11 adherence monitoring,554 assessment and correction of system failures that contribute to transmission,555, 556 and provision of feedback to HCWs and senior administrators.433, 547, 548, 557 The positive influence of institutional leadership has been demonstrated repeatedly in studies of HCWs' adherence to recommended hand hygiene practices.176, 177, 433, 547, 548, 558, 559, 560, 561, 562, 563 Health care administrators' involvement in the infection control processes can improve their awareness of the rationale and resource requirements for following recommended infection control practices.
Several administrative factors may affect the transmission of infectious agents in health care settings, including the institutional culture, individual HCW behavior, and the work environment. Each of these areas is suitable for performance improvement monitoring and incorporation into the organization's patient safety goals.542, 543, 545, 564
II.A.1.a. Scope of Work and Staffing Needs for Infection Control Professionals
The effectiveness of infection surveillance and control programs in preventing nosocomial infections in USt hospitals was assessed by the CDC through the Study on the Efficacy of Nosocomial Infection Control (SENIC Project) conducted between 1970 and 1976.565 In a representative sample of US general hospitals, those with a trained infection control physician or microbiologist involved in an infection control program and at least 1 infection control nurse per 250 beds were associated with a 32% lower rate of the 4 infections studied (CVC-associated bloodstream infections, ventilator-associated pneumonias, catheter-related urinary tract infections, and surgical site infections).
Since the publication of that landmark study, responsibilities of ICPs have expanded commensurate with the growing complexity of the health care system, the patient populations served, and the increasing numbers of medical procedures and devices used in all types of health care settings. The scope of work of ICPs was first assessed in 1982566, 567, 568 by the Certification Board of Infection Control, and has been reassessed every 5 years since that time.557, 569, 570, 571 The findings of these analyses have been used to develop and update the Infection Control Certification Examination, which was first offered in 1983. With each new survey, it becomes increasingly apparent that the role of the ICP is growing in complexity and scope beyond traditional infection control activities in acute care hospitals. Activities currently assigned to ICPs in response to emerging challenges include (1) surveillance and infection prevention at facilities other than acute care hospitals (eg, ambulatory clinics, day surgery centers, LTCFs, rehabilitation centers, home care); (2) oversight of employee health services related to infection prevention (eg, assessment of risk and administration of recommended treatment after exposure to infectious agents, tuberculosis screening, influenza vaccination, respiratory protection fit testing, and administration of other vaccines as indicated, such as smallpox vaccine in 2003); (3) preparedness planning for annual influenza outbreaks, pandemic influenza, SARS, and bioweapons attacks; (4) adherence monitoring for selected infection control practices; (5) oversight of risk assessment and implementation of prevention measures associated with construction and renovation; (6) prevention of transmission of MDROs; (7) evaluation of new medical products that could be associated with increased infection risk (eg, intravenous infusion materials); (8) communication with the public, facility staff, and state and local health departments concerning infection control–related issues; and (9) participation in local and multicenter research projects.433, 548, 551, 557, 572, 573
None of the Certification Board of Infection Control job analyses addressed specific staffing requirements for the identified tasks, although the surveys did include information about hours worked; the 2001 survey included the number of ICPs assigned to the responding facilities.557 There is agreement in the literature that a ratio of 1 ICP per 250 acute care beds is no longer adequate to meet current infection control needs; a Delphi project that assessed staffing needs of infection control programs in the 21st century concluded that a ratio of 0.8 to 1.0 ICP per 100 occupied acute care beds is an appropriate staffing level.551 A survey of participants in the NNIS system found an average daily patient census of 115 per ICP.315 Results of other studies have been similar: 3 per 500 beds for large acute care hospitals, 1 per 150 to 250 beds in LTCFs, and 1.56 per 250 in small rural hospitals.572, 574 The foregoing demonstrates that infection control staffing no longer can be based on patient census alone, but rather must be determined by the scope of the program, characteristics of the patient population, complexity of the health care system, tools available to assist personnel to perform essential tasks (eg, electronic tracking and laboratory support for surveillance), and unique or urgent needs of the institution and community.551 Furthermore, appropriate training is required to optimize the quality of work performed.557, 571, 575
II.A.1.a.i. Infection Control Nurse Liaison
Designating a bedside nurse on a patient care unit as an infection control liaison or “link nurse” is reported to be an effective adjunct to enhance infection control at the unit level.576, 577, 578, 579, 580, 581 Such individuals receive training in basic infection control and have frequent communication with ICPs, but maintain their primary role as bedside caregiver on their units. The infection control nurse liaison increases the awareness of infection control at the unit level. He or she is especially effective in implementating new policies or control interventions because of the rapport with individuals on the unit, an understanding of unit-specific challenges, and ability to promote strategies that are most likely to be successful in that unit. This position is an adjunct to, not a replacement for, fully trained ICPs. Furthermore, the infection control liaison nurses should not be counted when considering ICP staffing.
II.A.1.b. Bedside Nurse Staffing
There is increasing evidence that the level of bedside nurse staffing influences the quality of patient care.582, 583 Adequate nursing staff makes it more likely that infection control practices, including hand hygiene, Standard Precautions, and Transmission-Based Precautions, will be given appropriate attention and applied correctly and consistently.551 A national multicenter study reported strong and consistent inverse relationships between nurse staffing and 5 adverse outcomes in medical patients, 2 of which were HAIs (urinary tract infections and pneumonia).582 The association of nursing staff shortages with increased rates of HAI has been demonstrated in several outbreaks in hospitals and LTCFs, and with increased transmission of hepatitis C virus in dialysis units.22, 417, 550, 584, 585, 586, 587, 588, 589, 590, 591, 592, 593, 594, 595, 596 In most cases, when staffing was improved as part of a comprehensive control intervention, the outbreak ended or the HAI rate declined. In 2 studies,589, 595 the composition of the nursing staff (“pool” or “float” vs regular staff nurses) influenced the rate of primary bloodstream infections, with an increased infection rate occurring when the proportion of regular nurses decreased and that of pool nurses increased.
II.A.1.c. Clinical Microbiology Laboratory Support
The critical role of the clinical microbiology laboratory in infection control and health care epidemiology has been well described552, 553, 597, 598, 599 and is supported by the Infectious Disease Society of America's policy statement on the consolidation of clinical microbiology laboratories published in 2001.552 The clinical microbiology laboratory contributes to preventing transmission of infectious diseases in health care settings by promptly detecting and reporting epidemiologically important organisms, identifying emerging patterns of antimicrobial resistance, and assessing the effectiveness of recommended precautions to limit transmission during outbreaks.597 Outbreaks of infections may be recognized first by laboratorians.161 Health care organizations need to ensure the availability of the recommended scope and quality of laboratory services, a sufficient number of appropriately trained laboratory staff members, and systems to promptly communicate epidemiologically important results to those who will take action (eg, providers of clinical care, infection control staff, health care epidemiologists, and infectious disease consultants).600 As concerns about emerging pathogens and bioterrorism grow, the role of the clinical microbiology laboratory assumes ever-greater importance. For health care organizations that outsource microbiology laboratory services (eg, ambulatory care, home care, LTCFs, smaller acute care hospitals), it is important to specify by contract the types of services (eg, periodic institution-specific aggregate susceptibility reports) required to support infection control.
Several key functions of the clinical microbiology laboratory are relevant to this guideline:
-
•
Antimicrobial susceptibility by testing and interpretation in accordance with current guidelines developed by the National Committee for Clinical Laboratory Standards, known as the Clinical and Laboratory Standards Institute since 2005,601 for the detection of emerging resistance patterns602, 603 and for the preparation, analysis, and distribution of periodic cumulative antimicrobial susceptibility summary reports.604, 605, 606 Although not required, clinical laboratories ideally should have access to rapid genotypic identification of bacteria and their antibiotic resistance genes.607
-
•
Performance of surveillance cultures when appropriate (including retention of isolates for analysis), to assess patterns of infection transmission and effectiveness of infection control interventions at the facility or organization. Microbiologists assist in decision making regarding the indications for initiating and discontinuing active surveillance programs and optimizing the use of laboratory resources.
-
•
Molecular typing, onsite or outsourced, to investigate and control health care–associated outbreaks.608
-
•
Application of rapid diagnostic tests to support clinical decisions involving patient treatment, room selection, and implementation of control measures, including barrier precautions and use of vaccine or chemoprophylaxis agents (eg, influenza,609, 610, 611 B pertussis,612, RSV,613, 614 and enteroviruses615). The microbiologist provides guidance to limit rapid testing to clinical situations in which rapid results influence patient management decisions, and also provides oversight of point-of-care testing performed by nonlaboratory HCWs.616
-
•
Detection and rapid reporting of epidemiologically important organisms, including those that are reportable to public health agencies.
-
•
Implementation of a quality control program to ensure that testing services are appropriate for the population being served and are stringently evaluated for sensitivity, specificity, applicability, and feasibility.
-
•
Participation in a multidisciplinary team to develop and maintain an effective institutional program for the judicious use of antimicrobial agents.617, 618
II.A.2. Institutional Safety Culture and Organizational Characteristics
Safety culture (or safety climate) refers to a work environment in which a shared commitment to safety on the part of management and the workforce is understood and maintained.558, 619, 620 The authors of the Institute of Medicine's report titled To Err is Human 542 acknowledged that causes of medical error are multifaceted but emphasized the pivotal role of system failures and the benefits of a safety culture. A safety culture is created through (1) the actions that management takes to improve patient and worker safety, (2) worker participation in safety planning, (3) the availability of appropriate PPE, (4) the influence of group norms regarding acceptable safety practices, and (5) the organization's socialization process for new personnel. Safety and patient outcomes can be enhanced by improving or creating organizational characteristics within patient care units, as demonstrated by studies of surgical ICUs.621, 622 Each of these factors has a direct bearing on adherence to transmission prevention recommendations.256 Measurement of an institution's culture of safety is useful in designing improvements in health care.623, 624 Several hospital-based studies have linked measures of safety culture with both employee adherence to safe practices and reduced exposures to blood and body fluids.625, 626, 627, 628, 629, 630, 631 One study of hand hygiene practices concluded that improved adherence requires integration of infection control into the organization's safety culture.560 Several hospitals that are part of the Veterans Administration health care system have taken specific steps toward improving the safety culture, including error-reporting mechanisms, root cause analyses of identified problems, safety incentives, and employee education.632, 633, 634
II.A.3. Adherence of Health Care Workers to Recommended Guidelines
HCWs' adherence to recommended infection control practices decreases the transmission of infectious agents in health care settings.116, 561, 635, 636, 637, 638, 639 Several observational studies have shown limited adherence to recommended practices by HCWs.558, 639, 640, 641, 642, 643, 644, 645, 646, 647, 648, 649, 650, 651, 652, 653, 654, 655, 656 Observed adherence to universal precautions ranged from 43% to 89%.640, 641, 648, 650, 651 The degree of adherence often depended on the specific practice that was assessed and, for glove use, the circumstance in which the practice was applied. Observed rates of appropriate glove use has ranged from a low of 15%644 to a high of 82%.649 However, 92% and 98% adherence with glove use have been reported during arterial blood gas collection and resuscitation, respectively, procedures in which considerable blood contact may occur.642, 655 Differences in observed adherence have been reported among occupational groups in the same health care facility640 and between experienced and nonexperienced professionals.644 In surveys of HCWs, self-reported adherence was generally higher than actual adherence found in observational studies. Furthermore, where an observational component was included with a self-reported survey, self-perceived adherence was often greater than observed adherence.656 Among nurses and physicians, increasing years of experience is a negative predictor of adherence.644, 650 Education to improve adherence is the primary intervention that has been studied. Whereas positive changes in knowledge and attitude have been demonstrated,639, 657 no or only limited accompanying changes in behavior often have been found.641, 643 Self-reported adherence is higher in groups that received an educational intervention.629, 658 In one study, educational interventions that incorporated videotaping and performance feedback were successful in improving adherence during the study period, but the long-term effect of such interventions is not known.653 The use of videotaping also served to identify system problems (eg, communication and access to PPE) that otherwise may not have been recognized.
Interest is growing in the use of engineering controls and facility design concepts for improving adherence. Whereas the introduction of automated sinks was found to have a negative impact on consistent adherence to handwashing in one study,659 the use of electronic monitoring and voice prompts to remind HCWs to perform hand hygiene and improving accessibility to hand hygiene products increased adherence and contributed to a decrease in HAIs in another study.660 More information is needed regarding ways in which technology might improve adherence.
Improving adherence to infection control practices requires a multifaceted approach that incorporates continuous assessment of both the individual and the work environment.558, 560 Using several behavioral theories, Kretzer and Larson concluded that a single intervention (eg, a handwashing campaign or putting up new posters about transmission precautions) likely would be ineffective in improving HCWs adherence.661 Improvement requires the organizational leadership to make prevention an institutional priority and integrate infection control practices into the organization's safety culture.560 A recent review of the literature concluded that variations in organizational factors (eg, safety climate, policies and procedures, education and training) and individual factors (eg, knowledge, perceptions of risk, past experience) were determinants of adherence to infection control guidelines for protection against SARS and other respiratory pathogens.256
II.B. Surveillance for Health Care-Associated Infections
Surveillance is an essential tool for case finding of single patients or clusters of patients who are infected or colonized with epidemiologically important organisms (eg, susceptible bacteria such as S aureus, S pyogenes [group A streptococcus] or Enterobacter-Klebsiella spp; MRSA, VRE, and other MDROs; C difficile; RSV; influenza virus) for which transmission-based precautions may be required. Surveillance is defined as the ongoing systematic collection, analysis, interpretation, and dissemination of data regarding a health-related event for use in public health action to reduce morbidity and mortality and to improve health.662 The work of Ignaz Semmelweis delineating the role of person-to-person transmission in puerperal sepsis is the earliest example of the use of surveillance data to reduce transmission of infectious agents.663 Surveillance of both process measures and the infection rates to which they are linked is important in evaluating the effectiveness of infection prevention efforts and identifying indications for change.554, 664, 665, 666, 667
The Study on the Efficacy of Nosocomial Infection Control (SENIC) found that different combinations of infection control practices resulted in reduced rates of nosocomial surgical site infections, pneumonia, urinary tract infections, and bacteremia in acute care hospitals;565 however, surveillance was the only component essential for reducing all 4 types of HAIs. Although a similar study has not been conducted in other health care settings, a role for surveillance and the need for novel strategies in LTCFs397, 433, 668, 669 and in home care469, 470, 471, 472 have been described. The essential elements of a surveillance system are (1) standardized definitions, (2) identification of patient populations at risk for infection, (3) statistical analysis (eg, risk adjustment, calculation of rates using appropriate denominators, trend analysis using such methods as statistical process control charts), and (4) feedback of results to the primary caregivers.670, 671, 672, 673, 674, 675 Data gathered through surveillance of high-risk populations, device use, procedures, and facility locations (eg, ICUs) are useful in detecting transmission trends.670, 671, 672 Identification of clusters of infections should be followed by a systematic epidemiologic investigation to determine commonalities in persons, places, and time and to guide implementation of interventions and evaluation of the effectiveness of those interventions.
Targeted surveillance based on the highest-risk areas or patients has been preferred over facility-wide surveillance for the most effective use of resources.672, 675 However, for certain epidemiologically important organisms, surveillance may need to be facility-wide. Surveillance methods will continue to evolve as health care delivery systems change391, 676 and user-friendly electronic tools for electronic tracking and trend analysis become more widely available.673, 677, 678 Individuals with experience in health care epidemiology and infection control should be involved in selecting software packages for data aggregation and analysis, to ensure that the need for efficient and accurate HAI surveillance will be met. Effective surveillance is increasingly important as legislation requiring public reporting of HAI rates is passed and states work to develop effective systems to support such legislation.679
II.C. Education of Health Care Workers, Patients, and Families
The education and training of HCWs is a prerequisite for ensuring that policies and procedures for Standard and Transmission-Based Precautions are understood and practiced. Understanding the scientific rationale for the precautions will allow HCWs to apply procedures correctly, as well as to safely modify precautions based on changing requirements, resources, or health care settings.14, 654, 680, 681, 682, 683, 684, 685, 686, 687 One study found that the likelihood of HCWs developing SARS was strongly associated with less than 2 hours of infection control training and poor understanding of infection control procedures.688 Education regarding the important role of vaccines (eg, influenza, measles, varicella, pertussis, pneumococcal) in protecting HCWs, their patients, and family members can help improve vaccination rates.689, 690, 691, 692
Education on the principles and practices for preventing transmission of infectious agents should begin during training in the health professions and be provided to anyone who has an opportunity for contact with patients or medical equipment (eg, nursing and medical staff; therapists and technicians, including respiratory, physical, occupational, radiology, and cardiology personnel; phlebotomists; housekeeping and maintenance staff; and students). In health care facilities, education and training on Standard and Transmission-Based Precautions are typically provided at the time of orientation and should be repeated as necessary to maintain competency; updated education and training are necessary when policies and procedures are revised or when a special circumstance occurs, such as an outbreak that requires modification of current practice or adoption of new recommendations. Education and training materials and methods appropriate to the HCW's level of responsibility, individual learning habits, and language needs can improve the learning experience.657, 693, 694, 695, 696, 697, 698, 699, 700, 701
Education programs for HCWs have been associated with sustained improvement in adherence to best practices and a related decrease in device-associated HAIs in teaching and nonteaching settings638, 702 and in medical and surgical ICUs (Coopersmith, 2002 #2149; Babcock, 2004 #2126; Berenholtz, 2004 #2289; http://www.ihi.org/IHI/Programs/Campaign, #2563). Several studies have shown that in addition to targeted education to improve specific practices, periodic assessment and feedback of the HCW's knowledge and adherence to recommended practices are necessary to achieve the desired changes and identify continuing education needs.561, 703, 704, 705, 706, 707 The effectiveness of this approach for isolation practices has been demonstrated in the control of RSV.116, 683
Patients, family members, and visitors can be partners in preventing transmission of infections in health care settings.9, 42, 708, 709, 710 Information on Standard Precautions, especially hand hygiene, respiratory hygiene/cough etiquette, vaccination (especially against influenza), and other routine infection prevention strategies, may be incorporated into patient information materials provided on admission to the health care facility. Additional information on Transmission-Based Precautions is best provided when these precautions are initiated. Fact sheets, pamphlets, and other printed material may include information on the rationale for the additional precautions, risks to household members, room assignment for Transmission-Based Precautions purposes, explanation of the use of PPE by HCWs, and directions for use of such equipment by family members and visitors. Such information may be particularly helpful in the home environment, where household members often have the primary responsibility for adherence to recommended infection control practices. HCWs must be available and prepared to explain this material and answer questions as needed.
II.D. Hand Hygiene
Hand hygiene has been frequently cited as the single most important practice to reduce the transmission of infectious agents in health care settings558, 711, 712 and is an essential element of Standard Precautions. The term “hand hygiene” includes both handwashing with either plain or antiseptic-containing soap and water and the use of alcohol-based products (gels, rinses, foams) that do not require water. In the absence of visible soiling of hands, approved alcohol-based products for hand disinfection are preferred over antimicrobial or plain soap and water because of their superior microbiocidal activity, reduced drying of the skin, and convenience.558 Improved hand hygiene practices have been associated with a sustained decrease in the incidence of MRSA and VRE infections primarily in ICUs.560, 561, 713, 714, 715, 716 The scientific rationale, indications, methods, and products for hand hygiene have been summarized in previous publications.558, 716
The effectiveness of hand hygiene can be reduced by the type and length of fingernails.558, 717, 718 Individuals wearing artificial nails have been shown to harbor more pathogenic organisms, especially gram-negative bacilli and yeasts, on the nails and in the subungual area compared with individuals with native nails.719, 720 In 2002, the CDC/HICPAC recommended (Category IA) that artificial fingernails and extenders not be worn by HCWs who have contact with high-risk patients (eg, those in ICUs and operating rooms), due to the association with outbreaks of gram-negative bacillus and candidal infections as confirmed by molecular typing of isolates.30, 31, 558, 721, 722, 723, 724 The need to restrict the wearing of artificial fingernails by all HCWs who provide direct patient care and those who have contact with other high-risk groups (eg, oncology and cystic fibrosis patients) has not been studied but has been recommended by some experts.20 Currently, such decisions are at the discretion of an individual facility's infection control program. There is less evidence indicating that jewelry affects the quality of hand hygiene. Although hand contamination with potential pathogens is increased with ring-wearing,558, 725 no studies have related this practice to HCW-to-patient transmission of pathogens.
II.E. Personal Protective Equipment for Health Care Workers
PPE refers to various barriers and respirators used alone or in combination to protect mucous membranes, airways, skin, and clothing from contact with infectious agents. The choice of PPE is based on the nature of the patient interaction and/or the likely mode(s) of transmission. Specific guidance on the use of PPE is provided in Part III of this guideline. A suggested procedure for donning and removing PPE aimed at preventing skin or clothing contamination is presented in Figure 1. Designated containers for used disposable or reusable PPE should be placed in a location convenient to the site of removal, to facilitate disposal and containment of contaminated materials. Hand hygiene is always the final step after removing and disposing of PPE. The following sections highlight the primary uses of and criteria for selecting this equipment.
Fig 1.

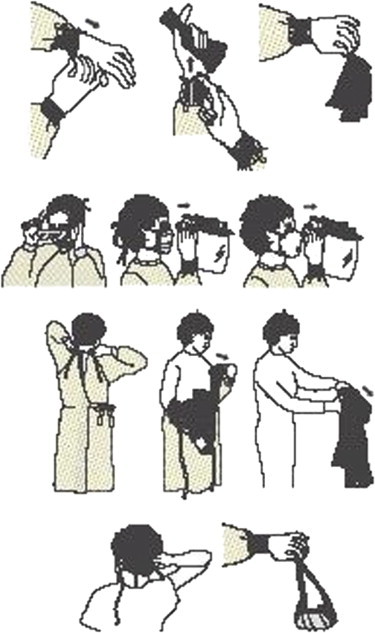
Example of safe donning and removal of PPE.
II.E.1. Gloves
Gloves are used to prevent contamination of HCW hands when (1) anticipating direct contact with blood or body fluids, mucous membranes, nonintact skin and other potentially infectious material; (2) having direct contact with patients who are colonized or infected with pathogens transmitted by the contact route (eg, VRE, MRSA, RSV558, 726, 727); or (3) handling or touching visibly or potentially contaminated patient care equipment and environmental surfaces.72, 73, 558 Gloves can protect both patients and HCWs from exposure to infectious material that may be carried on hands.73 The extent to which gloves will protect HCWs from transmission of bloodborne pathogens (eg, HIV, HBV, HCV) after a needlestick or other puncture that penetrates the glove barrier has not yet been determined. Although gloves may reduce the volume of blood on the external surface of a sharp by 46% to 86%,728 the residual blood in the lumen of a hollow-bore needle would not be affected; therefore, the effect on transmission risk is unknown.
Gloves manufactured for health care purposes are subject to FDA evaluation and clearance.729 Nonsterile disposable medical gloves made of various materials (eg, latex, vinyl, nitrile) are available for routine patient care.730 The selection of glove type for nonsurgical use is based on various factors, including the task to be performed, anticipated contact with chemicals and chemotherapeutic agents, latex sensitivity, sizing, and facility policies for creating a latex-free environment.17, 731, 732, 733 For contact with blood and body fluids during nonsurgical patient care, a single pair of gloves generally provides adequate barrier protection.733 However, there is considerable variability among gloves; both the quality of the manufacturing process and type of material influence their barrier effectiveness.734 Whereas there is little difference in the barrier properties of unused intact gloves,735 studies have shown repeatedly that vinyl gloves have higher failure rates than latex or nitrile gloves when tested under simulated and actual clinical conditions.730, 734, 735, 736, 737 For this reason, either latex or nitrile gloves are preferable for clinical procedures that require manual dexterity or will involve more than brief patient contact. A facility may need to stock gloves in several sizes. Heavier, reusable utility gloves are indicated for non–patient care activities, such as handling or cleaning contaminated equipment or surfaces.11, 14, 738
During patient care, transmission of infectious organisms can be reduced by adhering to the principles of working from “clean” to “dirty” and confining or limiting contamination to those surfaces directly needed for patient care. It may be necessary to change gloves during the care of a single patient to prevent cross-contamination of body sites.558, 739 It also may be necessary to change gloves if the patient interaction also involves touching portable computer keyboards or other mobile equipment transported from room to room. Discarding gloves between patients is necessary to prevent transmission of infectious material. Gloves must not be washed for subsequent reuse, because microorganisms cannot be removed reliably from glove surfaces, and continued glove integrity cannot be ensured. Furthermore, glove reuse has been associated with transmission of MRSA and gram-negative bacilli.740, 741, 742
When gloves are worn in combination with other PPE, they are put on last. Gloves that fit snugly around the wrist are preferred for use with an isolation gown, because they will cover the gown cuff and provide a more reliable continuous barrier for the arms, wrists, and hands. Proper glove removal will prevent hand contamination (Fig 1). Hand hygiene after glove removal further ensures that the hands will not carry potentially infectious material that might have penetrated through unrecognized tears or that could have contaminated the hands during glove removal.558, 727, 740
II.E.2. Isolation Gowns
Isolation gowns are used as specified by Standard and Transmission-Based Precautions to protect the HCW's arms and exposed body areas and prevent contamination of clothing with blood, body fluids, and other potentially infectious material.24, 88, 261, 743, 744, 745 The need for and the type of isolation gown selected is based on the nature of the patient interaction, including the anticipated degree of contact with infectious material and potential for blood and body fluid penetration of the barrier. The wearing of isolation gowns and other protective apparel is mandated by the Occupational Safety and Health Administration's (OSHA) Bloodborne Pathogens Standard.738 Clinical and laboratory coats or jackets worn over personal clothing for comfort and/or purposes of identity are not considered PPE.
When applying Standard Precautions, an isolation gown is worn only if contact with blood or body fluid is anticipated. However, when Contact Precautions are used (ie, to prevent transmission of an infectious agent that is not interrupted by Standard Precautions alone and is associated with environmental contamination), donning of both gown and gloves on room entry is indicated, to prevent unintentional contact with contaminated environmental surfaces.54, 72, 73, 88 The routine donning of isolation gowns on entry into an ICU or other high-risk area does not prevent or influence potential colonization or infection of patients in those areas, however.364, 746, 747, 748, 749
Isolation gowns are always worn in combination with gloves, and with other PPE when indicated. Gowns are usually the first piece of PPE to be donned. Full coverage of the arms and body front, from neck to the mid-thigh or below, will ensure protection of clothing and exposed upper body areas. Several gown sizes should be available in a health care facility to ensure appropriate coverage for staff members. Isolation gowns should be removed before leaving the patient care area to prevent possible contamination of the environment outside the patient's room. Isolation gowns should be removed in a manner that prevents contamination of clothing or skin (Fig 1); the outer, “contaminated” side of the gown is turned inward and rolled into a bundle, and then discarded into a designated container for waste or linen to contain contamination.
II.E.3. Face Protection: Masks, Goggles, and Face Shields
II.E.3.a. Masks
Masks are used for 3 primary purposes in health care settings: (1) placed on HCWs to protect them from contact with infectious material from patients (eg, respiratory secretions and sprays of blood or body fluids), consistent with Standard Precautions and Droplet Precautions; (2) placed on HCWs engaged in procedures requiring sterile technique, to protect patients from exposure to infectious agents carried in the HCW's mouth or nose; and (3) placed on coughing patients to limit potential dissemination of infectious respiratory secretions from the patient to others (ie, respiratory hygiene/cough etiquette). Masks may be used in combination with goggles to protect the mouth, nose, and eyes, or, alternatively, a face shield may be used instead of a mask and goggles to provide more complete protection for the face, as discussed below. Masks should not be confused with particulate respirators used to prevent inhalation of small particles that may contain infectious agents transmitted through the airborne route, as described below.
The mucous membranes of the mouth, nose, and eyes are susceptible portals of entry for infectious agents; other skin surfaces also may be portals if skin integrity is compromised (by, eg, acne, dermatitis).66, 750, 751, 752, 753 Therefore, use of PPE to protect these body sites is an important component of Standard Precautions. The protective effect of masks for exposed HCWs has been demonstrated previously.93, 113, 754, 755 Procedures that generate splashes or sprays of blood, body fluids, secretions, or excretions (eg, endotracheal suctioning, bronchoscopy, invasive vascular procedures) require either a face shield (disposable or reusable) or a mask and goggles.93, 94, 95, 96, 113, 115, 261, 738, 756 The wearing of masks, eye protection, and face shields in specified circumstances when blood or body fluid exposure is likely is mandated by OSHA's Bloodborne Pathogens Standard.738 Appropriate PPE should be selected based on the anticipated level of exposure.
Two mask types are available for use in health care settings: surgical masks that are cleared by the FDA and required to have fluid-resistant properties, and procedure or isolation masks.757 ,#2688 To date, no studies comparing mask types to determine whether one mask type provides better protection than another have been published. Because procedure/isolation masks are not regulated by the FDA, they may be more variable in terms of quality and performance than surgical masks. Masks come in various shapes (eg, molded and nonmolded), sizes, filtration efficiency, and method of attachment (eg, ties, elastic, ear loops). Health care facilities may find that different types of masks are needed to meet individual HCW needs.
II.E.3.b. Goggles and Face Shields
Guidance on eye protection for infection control has been published.758 The eye protection chosen for specific work situations (eg, goggles or face shield) depends on the circumstances of exposure, other PPE used, and personal vision needs. Personal eyeglasses and contact lenses are not considered adequate eye protection (see http://www.cdc.gov/niosh/topics/eye/eye-infectious.html). NIOSH guidelines specify that eye protection must be comfortable, allow for sufficient peripheral vision, and adjustable to ensure a secure fit. A health care facility may need to provide several different types, styles, and sizes of eye protection equipment. Indirectly vented goggles with a manufacturer's antifog coating may provide the most reliable practical eye protection from splashes, sprays, and respiratory droplets from multiple angles. Newer styles of goggles may provide better indirect airflow properties to reduce fogging, as well as better peripheral vision and more size options for fitting goggles to different workers. Many styles of goggles fit adequately over prescription glasses with minimal gaps. Although effective as eye protection, goggles do not provide splash or spray protection to other parts of the face.
The role of goggles in addition to a mask in preventing exposure to infectious agents transmitted through respiratory droplets has been studied only for RSV. Reports published in the mid-1980s demonstrated that eye protection reduced occupational transmission of RSV.759, 760 Whether this was due to the prevention hand–eye contact or the prevention of respiratory droplet–eye contact has not been determined. However, subsequent studies demonstrated that RSV transmission is effectively prevented by adherence to Standard Precautions plus Contact Precautions and that routine use of goggles is not necessary for this virus.24, 116, 117, 683, 761 It is important to remind HCWs that even if Droplet Precautions are not recommended for a specific respiratory tract pathogen, protection for the eyes, nose, and mouth using a mask and goggles or a face shield alone is necessary when a splash or spray of any respiratory secretions or other body fluids is likely to occur, as defined in Standard Precautions.
Disposable or nondisposable face shields may be used as an alternative to goggles.758 Compared with goggles, a face shield can provide protection to other facial areas besides the eyes. Face shields extending from the chin to crown provide better face and eye protection from splashes and sprays; face shields that wrap around the sides may reduce splashes around the edge of the shield.
Removal of a face shield, goggles, and mask can be performed safely after gloves have been removed and hand hygiene performed. The ties, earpieces, and/or headband used to secure the equipment to the head are considered “clean” and thus safe to touch with bare hands. The front of a mask, goggles, and face shield are considered contaminated (Fig 1).
II.E.4. Respiratory Protection
The subject of respiratory protection as it applies to preventing transmission of airborne infectious agents, including the need for and frequency of fit testing is under scientific review and was the subject of a 2004 CDC workshop.762 Respiratory protection currently requires the use of a respirator with N95 or higher-level filtration to prevent inhalation of infectious particles. Information about respirators and respiratory protection programs is summarized in the Guideline for Preventing Transmission of Mycobacterium tuberculosis in Health Care Settings.12
Respiratory protection is broadly regulated by OSHA under the general industry standard for respiratory protection (29 CFR 1910.134),763 which requires that US employers in all employment settings implement a program to protect employees from inhalation of toxic materials. OSHA program components include medical clearance to wear a respirator; provision and use of appropriate respirators, including fit-tested NIOSH-certified N95 and higher-level particulate filtering respirators; education on respirator use, and periodic reevaluation of the respiratory protection program. When selecting particulate respirators, models with inherently good fit characteristics (ie, those expected to provide protection factors of ≥ 10% to 95% of wearers) are preferred and theoretically could preclude the need for fit testing.764, 765 Issues pertaining to respiratory protection remain the subject of ongoing debate. Information on various types of respirators is available at http://www.cdc.gov/niosh/npptl/respirators/respsars.html and in several previously published studies.764, 766, 767 A user-seal check (formerly called a “fit check”) should be performed by the wearer of a respirator each time that the respirator is donned, to minimize air leakage around the face piece.768 The optimal frequency of fit testing has not been determined; retesting may be indicated if there is a change in wearer's facial features, onset of a medical condition that would affect respiratory function in the wearer, or a change in the model or size of the respirator that was initially assigned.12
Respiratory protection was first recommended for protection of US HCWs from exposure to M tuberculosis in 1989. That recommendation has been maintained in 2 successive revisions of the Guidelines for Prevention of Transmission of Tuberculosis in Hospitals and Other Health Care Settings.12, 126 The incremental benefit from respirator use, in addition to administrative and engineering controls (ie, AIIRs, early recognition of patients likely to have tuberculosis and prompt placement in an AIIR, and maintenance of a patient with suspected tuberculosis in an AIIR until no longer infectious), for preventing transmission of airborne infectious agents (eg, M tuberculosis) remains undetermined. Although some studies have demonstrated effective prevention of M tuberculosis transmission in hospitals in which surgical masks instead of respirators were used in conjunction with other administrative and engineering controls.636, 769, 770 the CDC currently recommends N95 or higher-level respirators for personnel exposed to patients with suspected or confirmed tuberculosis. Currently, this recommendation also holds for other diseases that could be transmitted through the airborne route, including SARS261 and smallpox,108, 129, 771 until inhalational transmission is better defined or health care-specific PPE more suitable for preventing infection is developed. Wearing of respirators is also currently recommended during the performance of aerosol-generating procedures (eg, intubation, bronchoscopy, suctioning) in patients with SARS-CoV infection, avian influenza, and pandemic influenza (see Appendix A).
Although Airborne Precautions are recommended for preventing airborne transmission of measles and varicella-zoster viruses, no data are available on which to base a recommendation for respiratory protection to protect susceptible personnel against these 2 infections. Transmission of varicella-zoster virus has been prevented among pediatric patients using negative-pressure isolation alone.772 Whether respiratory protection (ie, wearing a particulate respirator) will enhance protection from these viruses has not yet been studied. Because most HCWs have natural or acquired immunity to these viruses, only immune personnel generally care for patients with these infections.773, 774, 775, 776 Although there is no evidence suggesting that masks are not adequate to protect HCWs in these settings, for purposes of consistency and simplicity, or because of difficulties in ascertaining immunity, some facilities may require the use of respirators for entry into all AIIRs, regardless of the specific infectious agent present.
Procedures for safe removal of respirators are provided in Figure 1. In some health care settings, particulate respirators used to provide care for patients with M tuberculosis are reused by the same HCW. This is an acceptable practice providing that the respirator is not damaged or soiled, the fit is not compromised by a change in shape, and the respirator has not been contaminated with blood or body fluids. No data are available on which to base a recommendation regarding the length of time that a respirator may be safely reused.
II.F. Safe Work Practices to Prevent Health Care Worker Exposure to Bloodborne Pathogens
II.F.1. Prevention of Needlesticks and Other Sharps-Related Injuries
Injuries due to needles and other sharps have been associated with transmission of HBV, HCV, and HIV to HCWs.777, 778 The prevention of sharps injuries has always been an essential element of Universal Precautions and is now an aspect of Standard Precautions.1, 779 These include measures to handle needles and other sharp devices in a manner that will prevent injury to the user and to others who may encounter the device during or after a procedure. These measures apply to routine patient care and do not address the prevention of sharps injuries and other blood exposures during surgical and other invasive procedures addressed elsewhere.780, 781, 782, 783, 784
Since 1991, when OSHA first issued its Bloodborne Pathogens Standard to protect HCWs from blood exposure, the focus of regulatory and legislative activity has been on implementing a hierarchy of control measures. This has included focusing attention on removing sharps hazards through the development and use of engineering controls. The federal Needlestick Safety and Prevention Act, signed into law in November 2000, authorized OSHA's revision of its Bloodborne Pathogens Standard to more explicitly require the use of safety-engineered sharps devices.785 The CDC has provided guidance on sharps injury prevention,786, 787 including guidelines for the design, implementation and evaluation of a comprehensive sharps injury prevention program.788
II.F.2. Prevention of Mucous Membrane Contact
Exposure of mucous membranes of the eyes, nose, and mouth to blood and body fluids has been associated with the transmission of bloodborne viruses and other infectious agents to HCWs.66, 751, 753, 778 The prevention of mucous membrane exposures has always been an element of Universal Precautions and is now an element of Standard Precautions for routine patient care1, 752 and is subject to OSHA bloodborne pathogen regulations. Safe work practices, in addition to wearing PPE, are designed to protect mucous membranes and nonintact skin from contact with potentially infectious material. These include keeping contaminated gloved and ungloved hands from touching the mouth, nose, eyes, or face and positioning patients to direct sprays and splatter away from the caregiver's face. Careful placement of PPE before patient contact will help avoid the need to make adjustments to PPE and prevent possible face or mucous membrane contamination during use.
In areas where the need for resuscitation is unpredictable, mouthpieces, pocket resuscitation masks with 1-way valves, and other ventilation devices provide an alternative to mouth-to-mouth resuscitation, preventing exposure of the caregiver's nose and mouth to oral and respiratory fluids during the procedure.
II.F.2.a. Precautions During Aerosol-Generating Procedures
The performance of procedures that can generate small-particle aerosols (aerosol-generating procedures), such as bronchoscopy, endotracheal intubation, and open suctioning of the respiratory tract, have been associated with transmission of infectious agents to HCWs, including M tuberculosis,789 SARS-CoV,93, 94, 98 and N meningitidis.95 Protection of the eyes, nose, and mouth, in addition to gown and gloves, is recommended during performance of these procedures in accordance with Standard Precautions. The use of a particulate respirator is recommended during aerosol-generating procedures when the aerosol is likely to contain M tuberculosis, SARS-CoV, or avian or pandemic influenza viruses.
II.G. Patient Placement
II.G.1. Hospitals and Long-Term Care Facilities
Options for patient placement include single-patient rooms, 2-patient rooms, and multibed wards. Of these, single-patient rooms are preferred when transmission of an infectious agent is of concern. Although some studies have failed to demonstrate the efficacy of single-patient rooms in preventing HAIs,790 other published studies, including one commissioned by the AIA and the Facility Guidelines Institute, have documented a beneficial relationship between private rooms and reduced infectious and noninfectious adverse patient outcomes.791, 792 The AIA notes that private rooms are the trend in hospital planning and design. However, most hospitals and LTCFs have multibed rooms and must consider many competing priorities when determining the appropriate room placement for patients (eg, reason for admission; patient characteristics, such as age, gender, and mental status; staffing needs; family requests; psychosocial factors; reimbursement concerns). In the absence of obvious infectious diseases that require specified airborne infection isolation rooms (eg, tuberculosis, SARS, chickenpox), the risk of transmission of infectious agents is not always considered when making placement decisions.
When only a limited number of single-patient rooms is available, it is prudent to prioritize room assignments for those patients with conditions that facilitate transmission of infectious material to other patients (eg, draining wounds, stool incontinence, uncontained secretions) and those at increased risk of acquisition and adverse outcomes resulting from HAIs (due to, eg, immunosuppression, open wounds, indwelling catheters, anticipated prolonged length of stay, total dependence on HCWs for activities of daily living).15, 24, 43, 429, 793, 794
Single-patient rooms are always indicated for patients placed on Airborne Precautions in a PE and are preferred for patients requiring Contact or Droplet Precautions.23, 24, 409, 434, 795, 796 During a suspected or proven outbreak caused by a pathogen whose reservoir is the gastrointestinal tract, the use of single-patient rooms with private bathrooms limits opportunities for transmission, especially when the colonized or infected patient has poor personal hygiene habits or fecal incontinence, or cannot be expected to assist in maintaining procedures that prevent transmission of microorganisms (eg, infants, children, and patients with altered mental status or developmental delay). In the absence of continued transmission, it is not necessary to provide a private bathroom for patients colonized or infected with enteric pathogens as long as personal hygiene practices and Standard Precautions (especially hand hygiene and appropriate environmental cleaning) are maintained. Assignment of a dedicated commode to a patient, and cleaning and disinfecting fixtures and equipment that may have fecal contamination (eg, bathrooms, commodes,797 scales used for weighing diapers) and the adjacent surfaces with appropriate agents may be especially important when a single-patient room cannot be assigned, because environmental contamination with intestinal tract pathogens is likely from both continent and incontinent patients.54, 798 The results of several studies that investigated the benefit of a single-patient room in preventing transmission of C difficile were inconclusive.167, 799, 800, 801 Some studies have shown that being in the same room with a colonized or infected patient is not necessarily a risk factor for transmission;790, 802, 803, 804 however, for children, the risk of health care–associated diarrhea is increased with the increased number of patients per room.805 These findings demonstrate that patient factors are important determinants of infection transmission risks. The need for a single-patient room and/or private bathroom for any patient is best determined on a case-by-case basis.
Cohorting is the practice of grouping together patients who are colonized or infected with the same organism to confine their care to a single area and prevent contact with other patients. Cohorts are created based on clinical diagnosis, microbiologic confirmation (when available), epidemiology, and mode of transmission of the infectious agent. Avoiding placing severely immunosuppressed patients in rooms with other patients is generally preferred. Cohorting has been extensively used for managing outbreaks of MDROs, including MRSA,22, 806 VRE,637, 807, 808 MDR-ESBL,809 P aeruginosa,29 MSSA,810 RSV,811, 812 adenovirus keratoconjunctivitis,813 rotavirus,814 and SARS.815 Modeling studies provide additional support for cohorting patients to control outbreaks;816, 817, 818 however, cohorting often is implemented only after routine infection control measures have failed to control an outbreak.
Assigning or cohorting HCWs to care only for patients infected or colonized with a single target pathogen limits further transmission of the target pathogen to uninfected patients,739, 818 but is difficult to achieve in the face of current staffing shortages in hospitals582 and residential health care sites.819, 820, 821 However, cohorting of HCWs may be beneficial when transmission continues after implementing routine infection control measures and creating patient cohorts.
During periods when RSV, human metapneumovirus,822 parainfluenza, influenza, other respiratory viruses,823 and rotavirus are circulating in the community, cohorting based on the presenting clinical syndrome is often a priority in facilities that care for infants and young children.824 For example, during the respiratory virus season, infants may be cohorted based solely on the clinical diagnosis of bronchiolitis, due to the logistical difficulties and costs associated with requiring microbiologic confirmation before room placement and the predominance of RSV during most of the season. However, when available, single-patient rooms are always preferred, because a common clinical presentation (eg, bronchiolitis), can be caused by more than 1 infectious agent.822, 823, 825 Furthermore, the inability of infants and children to contain body fluids, and the close physical contact associated with their care, increases the risk of infection transmission for patients and personnel in this setting.24, 794
II.G.2. Ambulatory Care Settings
Patients actively infected with or incubating transmissible infectious diseases are frequently seen in ambulatory settings (eg, outpatient clinics, physicians' offices, emergency departments) and potentially expose HCWs and other patients, family members, and visitors.21, 34, 127, 135, 142, 826 In response to the global outbreak of SARS in 2003 and in preparation for pandemic influenza, HCWs working in outpatient settings are urged to implement source containment measures (eg, asking coughing patients to wear a surgical mask or cover coughing with tissues) to prevent transmission of respiratory infections, beginning at the initial patient encounter,9, 261, 827 as described in Section III.A.1.a. Signs can be posted at the facility's entrance or at the reception or registration desk requesting that the patient or individuals accompanying the patient promptly inform the receptionist of any symptoms of respiratory infection (eg, cough, flu-like illness, increased production of respiratory secretions). The presence of diarrhea, skin rash, or known or suspected exposure to a transmissible disease (eg, measles, pertussis, chickenpox, tuberculosis) also could be added. Prompt placement of a potentially infectious patient in an examination room limits the number of exposed individuals in the common waiting area.
In waiting areas, maintaining a distance between symptomatic and nonsymptomatic patients (eg, > 3 feet), in addition to source control measures, may limit exposures. However, infections transmitted through the airborne route (eg, M tuberculosis, measles, chickenpox) require additional precautions.12, 125, 828 Patients suspected of having such an infection can wear a surgical mask for source containment, if tolerated, and should be placed in an examination room (preferably an AIIR) as soon as possible. If this is not possible, then having the patient wear a mask and segregating the patient from other patients in the waiting area will reduce the risk of exposing others. Because the person(s) accompanying the patient also may be infectious, application of the same infection control precautions may be extended to these persons if they are symptomatic.21, 251, 829 Family members accompanying children admitted with suspected M tuberculosis have been found to have unsuspected pulmonary tuberculosis with cavitary lesions, even when asymptomatic.42, 830
Patients with underlying conditions that increase their susceptibility to infection (eg, immunocompromised status43, 44 or cystic fibrosis20) require special efforts to protect them from exposure to infected patients in common waiting areas. Informing the receptionist of their infection risk on arrival allows appropriate steps to further protect these patients from infection. In some cystic fibrosis clinics, to avoid exposure to other patients who could be colonized with B cepacia, patients have been given beepers on registration so that they may leave the area and receive notification to return when an examination room becomes available.831
II.G.3. Home Care
In home care, patient placement concerns focus on protecting others in the home from exposure to an infectious household member. For individuals who are especially vulnerable to adverse outcomes associated with certain infections, it may be beneficial to either remove them from the home or segregate them within the home. Persons who are not part of the household may need to be prohibited from visiting during the period of infectivity. For example, in a situation where a patient with pulmonary tuberculosis is contagious and being cared for at home, very young children (age under 4 years)832 and immunocompromised persons who have not yet been infected should be removed or excluded from the household. During the SARS outbreak of 2003, segregation of infected persons during the communicable phase of the illness was found to be beneficial in preventing household transmission.249, 833
II.H. Transport of Patients
Several principles guide the transport of patients requiring Transmission-Based Precautions. In the inpatient and residential settings, these include the following:
1. Limiting transport of such patients to essential purposes, such as diagnostic and therapeutic procedures that cannot be performed in the patient's room.
2. When transport is necessary, applying appropriate barriers on the patient (eg, mask, gown, wrapping in sheets or use of impervious dressings to cover the affected areas) when infectious skin lesions or drainage are present, consistent with the route and risk of transmission.
3. Notifying HCWs in the receiving area of the patient's impending arrival and of the necessary precautions to prevent transmission.
4. For patients being transported outside the facility, informing the receiving facility and the medi-van or emergency vehicle personnel in advance about the type of Transmission-Based Precautions being used.
For tuberculosis, additional precautions may be needed in a small shared air space, such as in an ambulance.12
II.I. Environmental Measures
Cleaning and disinfecting noncritical surfaces in patient care areas is an aspect of Standard Precautions. In general, these procedures do not need to be changed for patients on Transmission-Based Precautions. The cleaning and disinfection of all patient care areas is important for frequently touched surfaces, especially those closest to the patient, which are most likely to be contaminated (eg, bedrails, bedside tables, commodes, doorknobs, sinks, surfaces and equipment in close proximity to the patient).11, 72, 73, 834 The frequency or intensity of cleaning may need to be changed, based on the patient's level of hygiene and the degree of environmental contamination and for certain infectious agents with reservoirs in the intestinal tract.54 This may be particularly important in LTCFs and pediatric facilities, where patients with stool and urine incontinence are encountered more frequently. In addition, increased frequency of cleaning may be needed in a PE to minimize dust accumulation.11 Special recommendations for cleaning and disinfecting environmental surfaces in dialysis centers have been published previously.18 In all health care settings, administrative, staffing, and scheduling activities should prioritize the proper cleaning and disinfection of surfaces that could be implicated in transmission. During a suspected or proven outbreak in which an environmental reservoir is suspected, routine cleaning procedures should be reviewed, and the need for additional trained cleaning staff should be assessed. Adherence should be monitored and reinforced to promote consistent and correct cleaning.
US Environmental Protection Agency–registered disinfectants or detergents/disinfectants that best meet the overall needs of the health care facility for routine cleaning and disinfection should be selected.11, 835 In general, use of the existing facility detergent/disinfectant according to the manufacturer's recommendations for amount, dilution, and contact time is sufficient to remove pathogens from surfaces of rooms where colonized or infected individuals were housed. This includes those pathogens that are resistant to multiple classes of antimicrobial agents (eg, C difficile, VRE, MRSA, MDR-GNB11, 24, 88, 434, 745, 795, 836). Most often, environmental reservoirs of pathogens during outbreaks are related to a failure to follow recommended procedures for cleaning and disinfection, rather than to the specific cleaning and disinfectant agents used.837, 838, 839, 840
Certain pathogens (eg, rotavirus, noroviruses, C difficile) may be resistant to some routinely used hospital disinfectants.274, 291, 841, 842, 843, 844, 845, 846 The role of specific disinfectants in limiting transmission of rotavirus has been demonstrated experimentally.841 Also, because C difficile may display increased levels of spore production when exposed to non–chlorine-based cleaning agents, and because these spores are more resistant than vegetative cells to commonly used surface disinfectants, some investigators have recommended the use of a 1:10 dilution of 5.25% sodium hypochlorite (household bleach) and water for routine environmental disinfection of rooms of patients with C difficile when there is continued transmission.843, 847 One study found an association between the use of a hypochlorite solution and decreased rates of C difficile infections.846 The need to change disinfectants based on the presence of these organisms can be determined in consultation with the infection control committee.11, 846, 847
Detailed recommendations for disinfection and sterilization of surfaces and medical equipment that have been in contact with prion-containing tissue or high risk body fluids, and for cleaning of blood and body substance spills, are available in the Guidelines for Environmental Infection Control in Health Care Facilities 11 and in the Guideline for Disinfection and Sterilization.847
II.J. Patient Care Equipment and Instruments/Devices
Medical equipment and instruments/devices must be cleaned and maintained according to the manufacturers' instructions to prevent patient-to-patient transmission of infectious agents.86, 87, 324, 848 Cleaning to remove organic material always must precede high-level disinfection and sterilization of critical and semicritical instruments and devices, because residual proteinacous material reduces the effectiveness of the disinfection and sterilization processes.835, 847 Noncritical equipment, such as commodes, intravenous pumps, and ventilators, must be thoroughly cleaned and disinfected before being used on another patient. All such equipment and devices should be handled in a manner that will prevent HCW and environmental contact with potentially infectious material. It is important to include computers and personal digital assistants used in patient care in policies for cleaning and disinfection of noncritical items. The literature on contamination of computers with pathogens has been summarized,849 and 2 reports have linked computer contamination to colonization and infections in patients.850, 851 Although keyboard covers and washable keyboards that can be easily disinfected are available, the infection control benefit of these items and their optimal management have not yet been determined.
In all health care settings, providing patients who are on Transmission-Based Precautions with dedicated noncritical medical equipment (eg, stethoscope, blood pressure cuff, electronic thermometer) has proven beneficial for preventing transmission.74, 89, 739, 852, 853 When this is not possible, disinfection of this equipment after each use is recommended. Other previously published guidelines should be consulted for detailed guidance in developing specific protocols for cleaning and reprocessing medical equipment and patient care items in both routine and special circumstances.11, 14, 18, 20, 739, 835, 847
In home care, it is preferable to remove visible blood or body fluids from durable medical equipment before it leaves the home. Equipment can be cleaned onsite using a detergent/disinfectant and, when possible, should be placed in a plastic bag for transport to the reprocessing location.20, 738
II.K. Textiles and Laundry
Although soiled textiles, including bedding, towels, and patient or resident clothing, may be contaminated with pathogenic microorganisms, the risk of disease transmission is negligible if these textiles are handled, transported, and laundered in a safe manner.11, 854, 855 Key principles for handling soiled laundry are (1) avoiding shaking the items or handling them in any way that may aerosolize infectious agents, (2) avoiding contact of one's body and personal clothing with the soiled items being handled, and (3) containing soiled items in a laundry bag or designated bin. If a laundry chute is used, it must be maintained to minimize dispersion of aerosols from contaminated items.11 Methods of handling, transporting, and laundering soiled textiles are determined by organizational policy and any applicable regulations;738 guidance is provided in the Guidelines for Environmental Infection Control in Health Care Facilities.11 Rather than rigid rules and regulations, hygienic and common sense storage and processing of clean textiles is recommended.11, 856 When laundering is done outside of a health care facility, the clean items must be packaged or completely covered and placed in an enclosed space during transport to prevent contamination with outside air or construction dust that could contain infectious fungal spores that pose a risk for immunocompromised patients.11
Institutions are required to launder garments used as PPE and uniforms visibly soiled with blood or infective material.738 Little data exist on the safety of home laundering of HCW uniforms, but no increase in infection rates was observed in the one published study,857 and no pathogens were recovered from home- or hospital-laundered scrubs in another study.858 In the home, textiles and laundry from patients with potentially transmissible infectious pathogens do not require special handling or separate laundering and may be washed with warm water and detergent.11, 857, 858
II.L. Solid Waste
The management of solid waste emanating from the health care environment is subject to federal and state regulations for medical and nonmedical waste.859, 860 No additional precautions are needed for nonmedical solid waste removed from rooms of patients on Transmission-Based Precautions. Solid waste may be contained in a single bag of sufficient strength.861
II.M. Dishware and Eating Utensils
The combination of hot water and detergents used in dishwashers is sufficient to decontaminate dishware and eating utensils. Therefore, no special precautions are needed for dishware (eg, dishes, glasses, cups) or eating utensils. Reusable dishware and utensils may be used for patients requiring Transmission-Based Precautions. In the home and other communal settings, eating utensils and drinking vessels should not be shared, consistent with principles of good personal hygiene and to help prevent transmission of respiratory viruses, herpes simplex virus, and infectious agents that infect the gastrointestinal tract and are transmitted by the fecal/oral route (eg, hepatitis A virus, noroviruses). If adequate resources for cleaning utensils and dishes are not available, then disposable products may be used.
II.N. Adjunctive Measures
Important adjunctive measures that are not considered primary components of programs to prevent transmission of infectious agents but nonetheless improve the effectiveness of such programs include (1) antimicrobial management programs, (2) postexposure chemoprophylaxis with antiviral or antibacterial agents, (3) vaccines used both for pre-exposure and postexposure prevention, and (4) screening and restricting visitors with signs of transmissible infections. Detailed discussion of judicious use of antimicrobial agents is beyond the scope of this document; however, this topic has been addressed in a previous CDC guideline (http://www.cdc.gov/ncidod/dhqp/pdf/ar/mdroGuideline2006.pdf).
II.N.1. Chemoprophylaxis
Antimicrobial agents and topical antiseptics may be used to prevent infection and potential outbreaks of selected agents. Infections for which postexposure chemoprophylaxis is recommended under defined conditions include B pertussis,17, 862 N meningitides,863 B anthracis after environmental exposure to aeosolizable material,864 influenza virus,610 HIV,865 and group A streptococcus.160 Orally administered antimicrobials also may be used under defined circumstances for MRSA decolonization of patients or HCWs.866
Another form of chemoprophylaxis involves the use of topical antiseptic agents. For example, triple dye is routinely used on the umbilical cords of term newborns to reduce the risk of colonization, skin infections, and omphalitis caused by S aureus, including MRSA, and group A streptococcus.867, 868 Extension of the use of triple dye to low birth weight infants in a NICU was one component of a program that controlled a long-standing MRSA outbreak.22 Topical antiseptics (eg, mupirocin) also are used for decolonization of HCWs or selected patients colonized with MRSA, as discussed in the MDRO guideline866, 869, 870, 871, 872
II.N.2. Immunoprophylaxis
Certain immunizations recommended for susceptible HCWs have decreased the risk of infection and the potential for transmission in health care facilities.17, 873 The OSHA mandate requiring employers to offer HBV vaccination to HCWs has played a substantial role in the sharp decline in incidence of occupational HBV infection.777, 874 The routine administration of varicella vaccine to HCWs has decreased the need to place susceptible HCWs on administrative leave after exposure to patients with varicella.774 In addition, reports of health care–associated transmission of rubella in obstetric clinics33, 875 and measles in acute care settings34 demonstrate the importance of immunization of susceptible HCWs against childhood diseases. Many states have requirements for vaccination of HCWs for measles and rubella in the absence of evidence of immunity. Annual influenza vaccine campaigns targeted at patients and HCWs in LTCFs and acute care settings have been instrumental in preventing or limiting institutional outbreaks; consequently, increasing attention is being directed toward improving influenza vaccination rates in HCWs.35, 610, 689, 876, 877, 878
Transmission of B pertussis in health care facilities has been associated with large and costly outbreaks that include both HCWs and patients.17, 36, 41, 100, 682, 826, 879, 880 HCWs in close contact with infants with pertussis are at particularly high risk because of waning immunity and, until 2005, the absence of a vaccine appropriate for adults. But 2 acellular pertussis vaccines were licensed in the United States in 2005, 1 for use in individuals age 11 to 18 years and the other for use in those age 10 to 64 years.881 Current Advisory Committee on Immunization Practices provisional recommendations include immunization of adolescents and adults, especially those in contact with infants under age 12 months and HCWs with direct patient contact.882, 883
Immunization of children and adults will help prevent the introduction of vaccine-preventable diseases into health care settings. The recommended immunization schedule for children is published annually in the January issues of the Morbidity and Mortality Weekly Report, with interim updates as needed.884, 885 An adult immunization schedule also is available for healthy adults and those with special immunization needs due to high-risk medical conditions.886
Some vaccines are also used for postexposure prophylaxis of susceptible individuals, including varicella,887 influenza,610 hepatitis B,777 and smallpox225 vaccines.17, 873 In the future, administration of a newly developed S aureus conjugate vaccine (still under investigation) to selected patients may provide a novel method of preventing health care–associated S aureus (including MRSA) infections in high-risk groups (eg, hemodialysis patients and candidates for selected surgical procedures).888, 889
Immune globulin preparations also are used for postexposure prophylaxis of certain infectious agents under specified circumstances (eg, varicella-zoster virus, HBV, rabies, measles and hepatitis A virus17, 832, 873). The RSV monoclonal antibody preparation palivizumab may have contributed to controlling a nosocomial outbreak of RSV in one NICU, but there is insufficient evidence to support a routine recommendation for its use in this setting.890
II.N.3. Management of Visitors
II.N.3.a. Visitors as Sources of Infection
Visitors have been identified as the source of several types of HAIs (eg, pertussis,40, 41 M tuberculosis,42, 891 influenza and other respiratory viruses24, 43, 44, 372 and SARS21, 252, 253, 254). Effective methods for visitor screening in health care settings have not yet been studied, however. Visitor screening is especially important during community outbreaks of infectious diseases and for high-risk patient units. Sibling visits are often encouraged in birthing centers, postpartum rooms, pediatric inpatient units, PICUs, and residential settings for children; in hospital settings, a child visitor should visit only his or her own sibling. Screening of visiting siblings and other children before they are allowed into clinical areas is necessary to prevent the introduction of childhood illnesses and common respiratory infections. Screening may be passive, through the use of signs to alert family members and visitors with signs and symptoms of communicable diseases not to enter clinical areas. More active screening may include the completion of a screening tool or questionnaire to elicit information related to recent exposures or current symptoms. This information is reviewed by the facility staff, after which the visitor is either permitted to visit or is excluded.832
Family and household members visiting pediatric patients with pertussis and tuberculosis may need to be screened for a history of exposure, as well as signs and symptoms of current infection. Potentially infectious visitors are excluded until they receive appropriate medical screening, diagnosis, or treatment. If exclusion is not considered to be in the best interest of the patient or family (ie, primary family members of critically or terminally ill patients), then the symptomatic visitor must wear a mask while in the health care facility and remain in the patient's room, avoiding exposure to others, especially in public waiting areas and the cafeteria.
Visitor screening is used consistently on HSCT units.15, 43 However, considering the experience during the 2003 SARS outbreaks and the potential for pandemic influenza, developing effective visitor screening systems will be beneficial.9 Education concerning respiratory hygiene/cough etiquette is a useful adjunct to visitor screening.
II.N.3.b. Use of Barrier Precautions by Visitors
The use of gowns, gloves, and masks by visitors in health care settings has not been addressed specifically in the scientific literature. Some studies included the use of gowns and gloves by visitors in the control of MDROs but did not perform a separate analysis to determine whether their use by visitors had a measurable impact.892, 893, 894 Family members or visitors who are providing care to or otherwise are in very close contact with the patient (eg, feeding, holding) may also have contact with other patients and could contribute to transmission in the absence of effective barrier precautions. Specific recommendations may vary by facility or by unit and should be determined by the specific level of interaction.
Part III: Precautions to Prevent Transmission of Infectious Agents
There are 2 tiers of HICPAC/CDC precautions to prevent transmission of infectious agents, Standard Precautions and Transmission-Based Precautions. Standard Precautions are intended to be applied to the care of all patients in all health care settings, regardless of the suspected or confirmed presence of an infectious agent. Implementation of Standard Precautions constitutes the primary strategy for the prevention of health care–associated transmission of infectious agents among patients and HCWs. Transmission-Based Precautions are for patients who are known or suspected to be infected or colonized with infectious agents, including certain epidemiologically important pathogens, which require additional control measures to effectively prevent transmission. Because the infecting agent often is not known at the time of admission to a health care facility, Transmission-Based Precautions are used empirically, according to the clinical syndrome and the likely etiologic agents at the time, and then modified when the pathogen is identified or a transmissible infectious etiology is ruled out. Examples of this syndromic approach are presented in Table 2. The HICPAC/CDC Guidelines also include recommendations for creating a Protective Environment for allogeneic HSCT patients.
The specific elements of Standard and Transmission-Based Precautions are discussed in Part II of this guideline. In Part III, the circumstances in which Standard Precautions, Transmission-Based Precautions, and a Protective Environment are applied are discussed. Table 4, Table 5 summarize the key elements of these sets of precautions
III.A. Standard Precautions
Standard Precautions combine the major features of Universal Precautions779, 895 and Body Substance Isolation639 and are based on the principle that all blood, body fluids, secretions, excretions except sweat, nonintact skin, and mucous membranes may contain transmissible infectious agents. Standard Precautions include a group of infection prevention practices that apply to all patients, regardless of suspected or confirmed infection status, in any setting in which health care is delivered (Table 4). These include hand hygiene; use of gloves, gown, mask, eye protection, or face shield, depending on the anticipated exposure; and safe injection practices. Also, equipment or items in the patient environment likely to have been contaminated with infectious body fluids must be handled in a manner to prevent transmission of infectious agents (eg, wear gloves for direct contact, contain heavily soiled equipment, properly clean and disinfect or sterilize reusable equipment before use on another patient).
The application of Standard Precautions during patient care is determined by the nature of the HCW–patient interaction and the extent of anticipated blood, body fluid, or pathogen exposure. For some interactions (eg, performing venipuncture), only gloves may be needed; during other interactions (eg, intubation), use of gloves, gown, and face shield or mask and goggles is necessary. Education and training on the principles and rationale for recommended practices are critical elements of Standard Precautions because they facilitate appropriate decision-making and promote adherence when HCWs are faced with new circumstances.654, 680, 681, 682, 683, 684, 685 An example of the importance of the use of Standard Precautions is intubation, especially under emergency circumstances when infectious agents may not be suspected, but later are identified (eg, SARS-CoV, N meningitides). The application of Standard Precautions is described below and summarized in Table 4. Guidance on donning and removing gloves, gowns and other PPE is presented in Figure 1.
Standard Precautions are also intended to protect patients by ensuring that HCWs do not carry infectious agents to patients on their hands or via equipment used during patient care.
III.A.1. New Elements of Standard Precautions
Infection control problems that are identified in the course of outbreak investigations often indicate the need for new recommendations or reinforcement of existing infection control recommendations to protect patients. Because such recommendations are considered a standard of care and may not be included in other guidelines, they are added here to Standard Precautions. Three such areas of practice that have been added are respiratory hygiene/cough etiquette, safe injection practices, and use of masks for insertion of catheters or injection of material into spinal or epidural spaces through lumbar puncture procedures (eg, myelogram, spinal or epidural anesthesia). Although most elements of Standard Precautions evolved from Universal Precautions that were developed for protection of HCWs, these new elements of Standard Precautions focus on protection of patients.
III.A.1.a. Respiratory Hygiene/Cough Etiquette
The transmission of SARS-CoV in emergency departments by patients and their family members during the widespread SARS outbreaks in 2003 highlighted the need for vigilance and prompt implementation of infection control measures at the first point of encounter within a health care setting (eg, reception and triage areas in emergency departments, outpatient clinics, and physician offices).21, 254, 896 The strategy proposed has been termed respiratory hygiene/cough etiquette9, 827 and is intended to be incorporated into infection control practices as a new component of Standard Precautions. The strategy is targeted at patients and accompanying family members and friends with undiagnosed transmissible respiratory infections, and applies to any person with signs of illness including cough, congestion, rhinorrhea, or increased production of respiratory secretions when entering a health care facility.40, 41, 43 The term cough etiquette is derived from recommended source control measures for M tuberculosis.12, 126
The elements of respiratory hygiene/cough etiquette include (1) education of health care facility staff, patients, and visitors; (2) posted signs, in language(s) appropriate to the population served, with instructions to patients and accompanying family members or friends; (3) source control measures (eg, covering the mouth/nose with a tissue when coughing and prompt disposal of used tissues, using surgical masks on the coughing person when tolerated and appropriate); (4) hand hygiene after contact with respiratory secretions; and (5) spatial separation, ideally >3 feet, of persons with respiratory infections in common waiting areas when possible. Covering sneezes and coughs and placing masks on coughing patients are proven means of source containment that prevent infected persons from dispersing respiratory secretions into the air.107, 145, 897, 898 Masking may be difficult in some settings, (eg, pediatrics), in which case the emphasis by necessity may be on cough etiquette.899 Physical proximity of < 3 feet has been associated with an increased risk for transmission of infections through the droplet route (eg, N meningitidis 103 and group A streptococcus114) and thus supports the practice of distancing infected persons from others who are not infected. The effectiveness of good hygiene practices, especially hand hygiene, in preventing transmission of viruses and reducing the incidence of respiratory infections both within and outside900, 901, 902 health care settings is summarized in several reviews.558, 716, 903
These measures should be effective in decreasing the risk of transmission of pathogens contained in large respiratory droplets (eg, influenza virus,23 adenovirus,111 B pertussis,826 and M pneumoniae 112). Although fever will be present in many respiratory infections, patients with pertussis and mild upper respiratory tract infections are often afebrile. Therefore, the absence of fever does not always exclude a respiratory infection. Patients who have asthma, allergic rhinitis, or chronic obstructive lung disease also may be coughing and sneezing. Although these patients often are not infectious, cough etiquette measures are prudent.
HCWs are advised to observe Droplet Precautions (ie, wear a mask) and hand hygiene when examining and caring for patients with signs and symptoms of a respiratory infection. HCWs who have a respiratory infection are advised to avoid direct patient contact, especially with high-risk patients. If this is not possible, then a mask should be worn while providing patient care.
III.A.1.b. Safe Injection Practices
The investigation of 4 large outbreaks of HBV and HCV among patients in ambulatory care facilities in the United States identified a need to define and reinforce safe injection practices.452 The 4 outbreaks occurred in a private medical practice, a pain clinic, an endoscopy clinic, and a hematology/oncology clinic. The primary breaches in infection control practice that contributed to these outbreaks were reinsertion of used needles into a multiple-dose vial or solution container (eg, saline bag) and use of a single needle/syringe to administer intravenous medication to multiple patients. In 1 of these outbreaks, preparation of medications in the same workspace where used needle/syringes were dismantled also may have been a contributing factor. These and other outbreaks of viral hepatitis could have been prevented by adherence to basic principles of aseptic technique for the preparation and administration of parenteral medications.452, 453 These include the use of a sterile, single-use, disposable needle and syringe for each injection given and prevention of contamination of injection equipment and medication. Whenever possible, use of single-dose vials is preferred over multiple-dose vials, especially when medications will be administered to multiple patients.
Outbreaks related to unsafe injection practices indicate that some HCWs are unaware of, do not understand, or do not adhere to basic principles of infection control and aseptic technique. A survey of US health care workers who provide medication through injection found that 1% to 3% reused the same needle and/or syringe on multiple patients.904 Among the deficiencies identified in recent outbreaks were a lack of oversight of personnel and failure to follow up on reported breaches in infection control practices in ambulatory settings. Therefore, to ensure that all HCWs understand and adhere to recommended practices, principles of infection control and aseptic technique need to be reinforced in training programs and incorporated into institutional polices that are monitored for adherence.453
III.A.1.c. Infection Control Practices for Special Lumbar Puncture Procedures
In 2004, the CDC investigated 8 cases of postmyelography meningitis that either were reported to the CDC or identified through a survey of the Emerging Infections Network of the Infectious Disease Society of America. Blood and/or cerebrospinal fluid of all 8 cases yielded streptococcal species consistent with oropharyngeal flora and there were changes in the CSF indices and clinical status indicative of bacterial meningitis. Equipment and products used during these procedures (eg, contrast media) were excluded as probable sources of contamination. Procedural details available for 7 cases determined that antiseptic skin preparations and sterile gloves had been used. However, none of the clinicians wore a face mask, giving rise to the speculation that droplet transmission of oralpharyngeal flora was the most likely explanation for these infections. Bacterial meningitis after myelography and other spinal procedures (eg, lumbar puncture, spinal and epidural anesthesia, intrathecal chemotherapy) has been reported previously.905, 906, 907, 908, 909, 910, 911, 912, 913, 914 As a result, the question of whether face masks should be worn to prevent droplet spread of oral flora during spinal procedures (eg, myelography, lumbar puncture, spinal anesthesia) has been debated.915, 916 Face masks are effective in limiting the dispersal of oropharyngeal droplets917 and are recommended for the placement of central venous catheters.918 In October 2005, HICPAC reviewed the evidence and concluded that there is sufficient experience to warrant the additional protection of a face mask for the individual placing a catheter or injecting material into the spinal or epidural space.
III.B. Transmission-Based Precautions
There are 3 categories of Transmission-Based Precautions: Contact Precautions, Droplet Precautions, and Airborne Precautions. Transmission-Based Precautions are used when the route(s) of transmission is (are) not completely interrupted using Standard Precautions alone. For some diseases that have multiple routes of transmission (eg, SARS), more than 1 Transmission-Based Precautions category may be used. When used either singly or in combination, they are always used in addition to Standard Precautions. See Appendix A for recommended precautions for specific infections. When Transmission-Based Precautions are indicated, efforts must be made to counteract possible adverse effects on patients (ie, anxiety, depression and other mood disturbances,919, 920, 921 perceptions of stigma,922 reduced contact with clinical staff,923, 924, 925 and increases in preventable adverse events564) to improve acceptance by the patients and adherence by HCWs.
III.B.1. Contact Precautions
Contact Precautions are intended to prevent transmission of infectious agents, including epidemiologically important microorganisms, which are spread by direct or indirect contact with the patient or the patient's environment as described in Section I.B.3.a. The specific agents and circumstance for which Contact Precautions are indicated are found in Appendix A. The application of Contact Precautions for patients infected or colonized with MDROs is described in the 2006 HICPAC/CDC MDRO guideline.926 Contact Precautions also apply where the presence of excessive wound drainage, fecal incontinence, or other discharges from the body suggest an increased potential for extensive environmental contamination and risk of transmission. A single-patient room is preferred for patients who require Contact Precautions. When a single-patient room is not available, consultation with infection control personnel is recommended to assess the various risks associated with other patient placement options (eg, cohorting, keeping the patient with an existing roommate). In multipatient rooms, ≥ 3 feet spatial separation between beds is advised to reduce the opportunities for inadvertent sharing of items between the infected/colonized patient and other patients. HCWs caring for patients on Contact Precautions wear a gown and gloves for all interactions that may involve contact with the patient or potentially contaminated areas in the patient's environment. Donning PPE on room entry and discarding before exiting the patient room is done to contain pathogens, especially those that have been implicated in transmission through environmental contamination (eg, VRE, C difficile, noroviruses and other intestinal tract pathogens, RSV).54, 72, 73, 78, 273, 274, 739
III.B.2. Droplet Precautions
Droplet Precautions are intended to prevent transmission of pathogens spread through close respiratory or mucous membrane contact with respiratory secretions as described in Section I.B.3.b. Because these pathogens do not remain infectious over long distances in a health care facility, special air handling and ventilation are not required to prevent droplet transmission. Infectious agents for which Droplet Precautions are indicated are listed in Appendix A and include B pertussis, influenza virus, adenovirus, rhinovirus, N meningitides, and group A streptococcus (for the first 24 hours of antimicrobial therapy). A single-patient room is preferred for patients who require Droplet Precautions. When a single-patient room is not available, consultation with infection control personnel is recommended to assess the various risks associated with other patient placement options (eg, cohorting, keeping the patient with an existing roommate). Spatial separation of ≥ 3 feet and drawing the curtain between patient beds is especially important for patients in multibed rooms with infections transmitted by the droplet route. HCWs wear a mask (a respirator is not necessary) for close contact with infectious patient; the mask is generally donned on room entry. Patients on Droplet Precautions who must be transported outside of the room should wear a mask if tolerated and follow respiratory hygiene/cough etiquette.
III.B.3. Airborne Precautions
Airborne Precautions prevent transmission of infectious agents that remain infectious over long distances when suspended in the air (eg, rubeola virus [measles], varicella virus [chickenpox], M tuberculosis, and possibly SARS-CoV), as described in Section I.B.3.c and Appendix A. The preferred placement for patients who require Airborne Precautions is in an AIIR, a single-patient room equipped with special air handling and ventilation capacity that meet the AIA/Facility Guidelines Institute standards for AIIRs (ie, monitored negative pressure relative to the surrounding area; 12 air exchanges per hour for new construction and renovation and 6 air exchanges per hour for existing facilities; air exhausted directly to the outside or recirculated through HEPA filtration before return).12, 13 Some states require the availability of such rooms in hospitals, emergency departments, and nursing homes that care for patients with M tuberculosis. A respiratory protection program that includes education about use of respirators, fit testing, and user seal checks is required in any facility with AIIRs. In settings where Airborne Precautions cannot be implemented due to limited engineering resources (eg, physician offices), masking the patient, placing the patient in a private room (eg, office examination room) with the door closed, and providing N95 or higher-level respirators or masks if respirators are not available for HCWs will reduce the likelihood of airborne transmission until the patient is either transferred to a facility with an AIIR or returned to the home environment, as deemed medically appropriate. HCWs caring for patients on Airborne Precautions wear a mask or respirator, depending on the disease-specific recommendations (see Section II.E.4, Table 2, and Appendix A), that is donned before room entry. Whenever possible, nonimmune HCWs should not care for patients with vaccine-preventable airborne diseases (eg, measles, chickenpox, smallpox).
III.C. Syndromic and Empiric Applications of Transmission-Based Precautions
Diagnosis of many infections requires laboratory confirmation. Because laboratory tests, especially those that depend on culture techniques, often require 2 or more days for completion, Transmission-Based Precautions must be implemented while test results are pending, based on the clinical presentation and likely pathogens. Use of appropriate Transmission-Based Precautions at the time a patient develops symptoms or signs of transmissible infection, or arrives at a health care facility for care, reduces transmission opportunities. Although it is not possible to identify prospectively all patients needing Transmission-Based Precautions, certain clinical syndromes and conditions carry a sufficiently high risk to warrant their use empirically while confirmatory tests are pending (see Table 2). ICPs are encouraged to modify or adapt this table according to local conditions.
III.D. Discontinuation of Transmission-Based Precautions
Transmission-Based Precautions remain in effect for limited periods (ie, while the risk for transmission of the infectious agent persists or for the duration of the illness (see Appendix A). For most infectious diseases, this duration reflects known patterns of persistence and shedding of infectious agents associated with the natural history of the infectious process and its treatment. For some diseases (eg, pharyngeal or cutaneous diphtheria, RSV), Transmission-Based Precautions remain in effect until culture or antigen-detection test results document eradication of the pathogen and, for RSV, symptomatic disease is resolved. For other diseases (eg, M tuberculosis), state laws and regulations and health care facility policies may dictate the duration of precautions.12 In immunocompromised patients, viral shedding can persist for prolonged periods of time (many weeks to months) and transmission to others may occur during that time; therefore, the duration of contact and/or droplet precautions may be prolonged for many weeks.499, 927, 928, 929, 930, 931, 932
The duration of Contact Precautions for patients who are colonized or infected with MDROs remains undefined. MRSA is the only MDRO for which effective decolonization regimens are available.866 However, carriers of MRSA who have negative nasal cultures after a course of systemic or topical therapy may resume shedding MRSA in the weeks after therapy.933, 934 Although early guidelines for VRE suggested discontinuation of Contact Precautions after 3 stool cultures obtained at weekly intervals proved negative,739 subsequent experiences have indicated that such screening may fail to detect colonization that can persist for > 1 year.27, 935, 936, 937 Likewise, available data indicate that colonization with VRE, MRSA,938 and possibly MDR-GNB can persist for many months, especially in the presence of severe underlying disease, invasive devices, and recurrent courses of antimicrobial agents.
It may be prudent to assume that MDRO carriers are colonized permanently and manage them accordingly. Alternatively, an interval free of hospitalizations, antimicrobial therapy, and invasive devices (eg, 6 or 12 months) before reculturing patients to document clearance of carriage may be used. Determination of the best strategy awaits the results of additional studies. See the 2006 HICPAC/CDC MDRO guideline926 for a discussion of possible criteria to discontinue Contact Precautions for patients colonized or infected with MDROs.
III.E. Application of Transmission-Based Precautions in Ambulatory and Home Care Settings
Although Transmission-Based Precautions generally apply in all health care settings, exceptions exist. For example, in home care, AIIRs are not available. Furthermore, family members already exposed to diseases such as varicella and tuberculosis would not use masks or respiratory protection, but visiting HCWs would need to use such protection. Similarly, management of patients colonized or infected with MDROs may necessitate Contact Precautions in acute care hospitals and in some LTCFs when there is continued transmission, but the risk of transmission in ambulatory care and home care has not been defined. Consistent use of Standard Precautions may suffice in these settings, but more information is needed.
III.F. Protective Environment
A PE is designed for allogeneic HSCT patients to minimize fungal spore counts in the air and reduce the risk of invasive environmental fungal infections (see Table 5 for specifications).11, 13, 14, 15 The need for such controls has been demonstrated in studies of aspergillosis outbreaks associated with construction.11, 14, 15, 157, 158 As defined by the AIA13 and presented in detail in the CDC's 2003 Guideline for Environmental Infection Control in Health Care Facilities,11, 860 air quality for HSCT patients is improved through a combination of environmental controls that include (1) HEPA filtration of incoming air, (2) directed room air flow, (3) positive room air pressure relative to the corridor, (4) well-sealed rooms (including sealed walls, floors, ceilings, windows, electrical outlets) to prevent flow of air from the outside, (5) ventilation to provide ≥ 12 air changes per hour, (6) strategies to minimize dust (eg, scrubbable surfaces rather than upholstery939 and carpet,940 and routinely cleaning crevices and sprinkler heads), and (7) prohibiting dried and fresh flowers and potted plants in the rooms of HSCT patients. The latter is based on molecular typing studies that have found indistinguishable strains of Aspergillus terreus in patients with hematologic malignancies and in potted plants in the vicinity of the patients.941, 942, 943 The desired quality of air may be achieved without incurring the inconvenience or expense of laminar airflow.15, 157 To prevent inhalation of fungal spores during periods when construction, renovation, or other dust-generating activities that may be ongoing in and around the health care facility, it has been recommended that severely immunocompromised patients wear a high-efficiency respiratory protection device (eg, an N95 respirator) when they leave the PE.11, 14, 944 The use of masks or respirators by HSCT patients when they are outside of the PE for prevention of environmental fungal infections in the absence of construction has not been evaluated. A PE does not include the use of barrier precautions beyond those indicated for Standard Precuations and Transmission-Based Precautions. No published reports support the benefit of placing patients undergoing solid organ transplantation or other immunocompromised patients in a PE.
Part IV: Recommendations
These recommendations are designed to prevent transmission of infectious agents among patients and HCWs in all settings where health care is delivered. As in other CDC/HICPAC guidelines, each recommendation is categorized on the basis of existing scientific data, theoretical rationale, applicability, and, when possible, economic impact. The CDC/HICPAC system for categorizing recommendations is as follows:
Category IA. Strongly recommended for implementation and strongly supported by well-designed experimental, clinical, or epidemiologic studies.
Category IB. Strongly recommended for implementation and supported by some experimental, clinical, or epidemiologic studies and a strong theoretical rationale.
Category IC. Required for implementation, as mandated by federal and/or state regulation or standard.
Category II. Suggested for implementation and supported by suggestive clinical or epidemiologic studies or a theoretical rationale.
No recommendation; unresolved issue. Practices for which insufficient evidence or no consensus regarding efficacy exists.
I. Administrative Responsibilities
Health care organization administrators should ensure the implementation of recommendations specified in this section.
-
I.A.
Incorporate preventing transmission of infectious agents into the objectives of the organization's patient and occupational safety programs.542, 543, 544, 545, 560, 629, 625, 945 Category IB/IC
-
I.B.Make preventing transmission of infectious agents a priority for the health care organization. Provide administrative support, including fiscal and human resources for maintaining infection control programs.433, 547, 548, 551, 558, 560, 561, 562, 563, 565, 661, 945 Category IB/IC
-
I.B.1.Ensure that individuals with training in infection control are employed by or are available by contract to all health care facilities, so that the infection control program is managed by 1 or more qualified individuals.315, 551, 565, 572, 575, 574, 945, 946 Category IB/IC
-
I.B.1.a.Determine the specific infection control full-time equivalents according to the scope of the infection control program, the complexity of the health care facility or system, the characteristics of the patient population, the unique or urgent needs of the facility and community, and proposed staffing levels based on survey results and recommendations from professional organizations.315, 433, 548, 551, 565, 568, 572, 574, 947, 948 Category IB
-
I.B.1.a.
- I.B.2.
- I.B.3.
- I.B.4.
- I.B.5.
-
I.B.6.Ensure availability of human and fiscal resources to provide clinical microbiology laboratory support, including a sufficient number of medical technologists trained in microbiology, appropriate to the health care setting, for monitoring transmission of microorganisms, planning and conducting epidemiologic investigations, and detecting emerging pathogens. Identify resources for performing surveillance cultures, rapid diagnostic testing for viral and other selected pathogens, preparation of antimicrobial susceptibility summary reports, trend analysis, and molecular typing of clustered isolates (performed either onsite or in a reference laboratory) and use these resources according to facility-specific epidemiologic needs, in consultation with clinical microbiologists.552, 553, 597, 598, 602, 604, 605, 606, 608, 609, 611, 613, 614, 615, 616, 953 Category IB
- I.B.7.
- I.B.8.
- I.B.9.
-
I.B.1.
-
I.C.
Develop and implement processes to ensure oversight of infection control activities appropriate to the health care setting and assign responsibility for oversight of infection control activities to an individual or group within the health care organization that is knowledgeable about infection control.433, 548, 565 Category II
-
I.D.
Develop and implement systems for early detection and management (eg, use of appropriate infection control measures, including isolation precautions, PPE) of potentially infectious persons at initial points of patient encounter in outpatient settings (eg, triage areas, emergency departments, outpatient clinics, physician offices) and at the time of admission to hospitals and LTCFs.9, 122, 134, 253, 826 Category IB
-
I.E.
Develop and implement policies and procedures to limit patient visitation by persons with signs or symptoms of a communicable infection. Screen visitors to high-risk patient care areas (eg, oncology units, HSCT units, intensive care units, other severely immunocompromised patients) for possible infection.24, 41, 43, 960, 961 Category IB
-
I.F.
Identify performance indicators of the effectiveness of organization-specific measures to prevent transmission of infectious agents (Standard Precautions and Transmission-Based Precautions), establish processes to monitor adherence to those performance measures, and provide feedback to staff members.554, 665, 666, 667, 703, 704, 739, 962 Category IB
II. Education and Training
-
II.A.Provide job- or task-specific education and training on preventing transmission of infectious agents associated with health care during orientation to the health care facility; update information periodically during ongoing education programs. Target all HCWs for education and training, including but not limited to medical, nursing, clinical technicians, and laboratory staff; property service (housekeeping), laundry, maintenance and dietary workers; students; contract staff; and volunteers. Document competency initially and repeatedly, as appropriate, for the specific staff positions. Develop a system to ensure that HCWs employed by outside agencies meet these education and training requirements through programs offered by the agencies or by participation in the health care facility's program designed for full-time personnel.126, 558, 560, 561, 654, 680, 681, 682, 683, 685, 687, 688, 701, 892, 918, 963 Category IB
- II.A.1.
- II.A.2.
-
II.B.
Provide instructional materials for patients and visitors on recommended hand hygiene and respiratory hygiene/cough etiquette practices and the application of Transmission-Based Precautions.9, 708, 709, 961 Category II
III. Surveillance
-
III.A.
Monitor the incidence of epidemiologically important organisms and targeted HAIs that have a substantial impact on outcome and for which effective preventive interventions are available. Use information collected through surveillance of high-risk populations, procedures, devices, and highly transmissible infectious agents to detect transmission of infectious agents in the health care facility.565, 670, 671, 672, 674, 686, 918, 965, 966, 967, 968 Category IA
-
III.B.Apply the following epidemiologic principles of infection surveillance:662, 663, 670, 672, 965, 967 Category IB
-
•Use standardized definitions of infection.
-
•Use laboratory-based data (when available).
-
•Collect epidemiologically important variables (eg, patient locations and/or clinical service in hospitals and other large multiunit facilities, population-specific risk factors [eg, low birth weight neonates], underlying conditions that predispose to serious adverse outcomes).
-
•Analyze data to identify trends that may indicated increased rates of transmission.
-
•Feedback information on trends in the incidence and prevalence of HAIs, probable risk factors, and prevention strategies and their impact to the appropriate health care providers, organization administrators, and as required by local and state health authorities.
-
•
-
III.C.
Develop and implement strategies to reduce risks for transmission and evaluate effectiveness565, 672, 683, 961, 968, 969 Category IB
-
III.D.
When transmission of epidemiologically important organisms continues despite implementation and documented adherence to infection prevention and control strategies, obtain consultation from persons knowledgeable in infection control and health care epidemiology to review the situation and recommend additional measures for control.247, 566, 686 Category IB
-
III.E.
Periodically review information on community or regional trends regarding the incidence and prevalence of epidemiologically important organisms (eg, influenza, RSV, pertussis, invasive group A streptococcal disease, MRSA, VRE) (including in other health care facilities) that may affect transmission of organisms within the facility.397, 686, 970, 971, 972 Category II
IV. Standard Precautions
Assume that every person is potentially infected or colonized with an organism that could be transmitted in the health care setting and apply the following infection control practices during the delivery of health care.
-
IV.A.Hand Hygiene
-
IV.A.1.During the delivery of health care, avoid unnecessary touching of surfaces in close proximity to the patient to prevent both contamination of clean hands from environmental surfaces and transmission of pathogens from contaminated hands to surfaces.72, 73, 738, 799, 973 {CDC, 2001 #970. Category IB/IC
-
IV.A.2.When hands are visibly dirty, contaminated with proteinaceous material, or visibly soiled with blood or body fluids, wash hands with either a nonantimicrobial soap and water or an antimicrobial soap and water.558 Category IA
-
IV.A.3.If hands are not visibly soiled, or after removing visible material with nonantimicrobial soap and water, decontaminate hands in the clinical situations described in IV.A.2.a–f. The preferred method of hand decontamination is with an alcohol-based hand rub.561, 974 Alternatively, hands may be washed with an antimicrobial soap and water. Frequent use of an alcohol-based hand rub immediately after handwashing with nonantimicrobial soap may increase the frequency of dermatitis.558 Category IB
- Perform hand hygiene:
- IV.A.3.a.
-
IV.A.3.b.After contact with blood, body fluids or excretions, mucous membranes, nonintact skin, or wound dressings.663 Category IA
- IV.A.3.c.
-
IV.A.3.d.If hands will be moving from a contaminated body site to a clean body site during patient care. Category II
- IV.A.3.e.
- IV.A.3.f.
-
IV.A.4.Wash hands with nonantimicrobial soap and water or with antimicrobial soap and water if contact with spores (eg, C difficile or B anthracis) is likely to have occurred. The physical action of washing and rinsing hands under such circumstances is recommended because alcohols, chlorhexidine, iodophors, and other antiseptic agents have poor activity against spores.558, 954, 981 Category II
-
IV.A.5.Do not wear artificial fingernails or extenders if duties include direct contact with patients at high risk for infection and associated adverse outcomes (eg, those in ICUs or operating rooms).30, 31, 558, 721, 722, 723 Category IA
-
IV.A.5.a.Develop an organizational policy on the wearing of nonnatural nails by HCWs who have direct contact with patients outside of the groups specified above.982 Category II
-
IV.A.5.a.
-
IV.A.1.
-
IV.B.Personal protective equipment (see Fig 1)
-
IV.B.1.Observe the following principles of use:
- IV.B.1.a.
-
IV.B.1.b.Prevent contamination of clothing and skin during the process of removing PPE (see Fig 1). Category II
- IV.B.1.c.
-
IV.B.2.Gloves
- IV.B.2.a.
-
IV.B.2.b.Wear gloves with fit and durability appropriate to the task.558, 730, 731, 738, 984, 985 Category IB
-
IV.B.2.b.i.Wear disposable medical examination gloves for providing direct patient care.
-
IV.B.2.b.ii.Wear disposable medical examination gloves or reusable utility gloves for cleaning the environment or medical equipment.
-
IV.B.2.b.i.
-
IV.B.2.c.Remove gloves after contact with a patient and/or the surrounding environment (including medical equipment) using proper technique to prevent hand contamination (see Fig 1). Do not wear the same pair of gloves for the care of more than 1 patient. Do not wash gloves for the purpose of reuse, because this practice has been associated with transmission of pathogens.558, 727, 740, 741, 742, 986 Category IB
-
IV.B.2.d.Change gloves during patient care if the hands will move from a contaminated body site (eg, perineal area) to a clean body site (eg, face). Category II
-
IV.B.3.Gowns
- IV.B.3.a.
-
IV.B.3.b.Do not reuse gowns, even for repeated contacts with the same patient. Category II
- IV.B.3.c.
-
IV.B.4.Mouth, nose, and eye protection
-
IV.B.4.a.Use PPE to protect the mucous membranes of the eyes, nose, and mouth during procedures and patient care activities that are likely to generate splashes or sprays of blood, body fluids, secretions, and excretions. Select masks, goggles, face shields, and combinations of these according to the need anticipated by the task to be performed.113, 738, 779, 895 Category IB/IC
-
IV.B.4.a.
-
IV.B.5.During aerosol-generating procedures (eg, bronchoscopy, suctioning of the respiratory tract [if not using in-line suction catheters], endotracheal intubation) in patients who are not suspected of being infected with an agent for which respiratory protection is otherwise recommended (eg, M tuberculosis, SARS, or hemorrhagic fever viruses), wear one of the following: a face shield that fully covers the front and sides of the face, a mask with attached shield, or a mask and goggles (in addition to gloves and gown).93, 94, 95, 96, 113, 126, 134 Category IB
-
IV.B.1.
-
IV.C.Respiratory hygiene/cough etiquette
-
IV.C.1.Educate HCWs on the importance of source control measures to contain respiratory secretions, to prevent droplet and fomite transmission of respiratory pathogens, especially during seasonal outbreaks of viral respiratory tract infections (eg, influenza, RSV, adenovirus, parainfluenza virus) in communities.10, 14, 24, 261, 683 Category IB
-
IV.C.2.Implement the following measures to contain respiratory secretions in patients and accompanying individuals who have signs and symptoms of a respiratory infection, beginning at the point of initial encounter in a health care setting (eg, triage, reception and waiting areas in emergency departments, outpatient clinics, and physicians' offices).20, 24, 145, 901, 987
-
IV.C.2.a.Post signs at entrances and in strategic places (eg, elevators, cafeterias) within ambulatory and inpatient settings with instructions to patients and other persons with symptoms of respiratory infection to cover their mouths and noses when coughing or sneezing, use and dispose of tissues, and perform hand hygiene after hands have been in contact with respiratory secretions. Category II
-
IV.C.2.b.Provide tissues and no-touch receptacles (eg, foot pedal–operated lid or open, plastic-lined wastebasket) for disposal of tissues.20 Category II
- IV.C.2.c.
-
IV.C.2.d.During periods of increased prevalence of respiratory infections in the community (as indicated by, eg, increased school absenteeism, increased number of patients seeking care for respiratory infection), offer masks to coughing patients and other symptomatic persons (eg, persons who accompany ill patients) on entry into the facility or medical office126, 898, 899 and encourage them to maintain special separation (ideally, at least 3 feet) from others in common waiting areas.20, 23, 103, 111, 114, 134 Category IB
-
IV.C.2.d.i.Some facilities may find it logistically easier to institute this recommendation year-round as a standard of practice. Category II
-
IV.C.2.d.i.
-
IV.C.2.a.
-
IV.C.1.
-
IV.D.Patient placement
-
IV.D.1.Include the potential for transmission of infectious agents in patient placement decisions. Place patients who pose a risk for transmission to others (eg, those with uncontained secretions, excretions, or wound drainage; infants with suspected viral respiratory or gastrointestinal infections) in a single-patient room when available.24, 409, 429, 434, 792, 795, 796, 805, 988 Category IB
-
IV.D.2.Determine patient placement based on the following factors:
-
•Route(s) of transmission of the known or suspected infectious agent
-
•Risk factors for transmission in the infected patient
-
•Risk factors for adverse outcomes resulting from an HAI in other patients in the area or room being considered for patient placement
-
•Availability of single-patient rooms
-
•Patient options for room sharing (eg, cohorting patients with the same infection) Category II
-
•
-
IV.D.1.
-
IV.E.Patient care equipment and instruments/devices954
- IV.E.1.
- IV.E.2.
- IV.E.3.
-
IV.F.Care of the environment11
-
IV.F.1.Establish policies and procedures for routine and targeted cleaning of environmental surfaces as indicated by the level of patient contact and degree of soiling.11 Category II
-
IV.F.2.Clean and disinfect surfaces likely to be contaminated with pathogens, including those in close proximity to the patient (eg, bed rails, over bed tables) and frequently touched surfaces in the patient care environment (eg, door knobs, surfaces in and surrounding toilets in patient rooms) on a more frequent schedule compared with that for other surfaces (eg, horizontal surfaces in waiting rooms).11, 72, 73, 739, 745, 799, 833, 991, 992, 993 Category IB
- IV.F.3.
- IV.F.4.
- Consider the following principles when developing this policy and procedures: Category II
-
•Select play toys that can be easily cleaned and disinfected.
-
•Do not permit use of stuffed furry toys if they will be shared.
-
•Clean and disinfect large stationary toys (eg, climbing equipment) at least weekly and whenever visibly soiled.
-
•If toys are likely to be mouthed, rinse with water after disinfection; alternatively, wash in a dishwasher.
-
•When a toy requires cleaning and disinfection, do so immediately or store in a designated labeled container separate from toys that are clean and ready for use.
-
•
-
IV.F.5.Include multiuse electronic equipment in policies and procedures for preventing contamination and for cleaning and disinfection, especially those items that are used by patients, those used during delivery of patient care, and mobile devices that are moved in and out of patient rooms frequently (eg, daily).849, 850, 851, 995 Category IB
-
IV.F.5.a.No recommendation for use of removable protective covers or washable keyboards. Unresolved issue
-
IV.F.5.a.
-
IV.F.1.
-
IV.G.Textiles and laundry
- IV.G.1.
- IV.G.2.
-
IV.H.
Safe injection practices
- The following recommendations apply to the use of needles, cannulas that replace needles, and, where applicable, intravenous delivery systems:453
- IV.H.1.
-
IV.H.2.Do not administer medications from a syringe to multiple patients, even if the needle or cannula on the syringe is changed. Needles, cannulae, and syringes are sterile, single-use items; they should not be reused for another patient or to access a medication or solution that might be used for a subsequent patient.452, 918, 1002, 1003 Category IA
-
IV.H.3.Use fluid infusion and administration sets (ie, intravenous bags, tubing and connectors) for one patient only and dispose of appropriately after use. Consider a syringe or needle/cannula to be contaminated once it has been used to enter or connect to a patient's intravenous infusion bag or administration set.452 Category IB
-
IV.H.4.Use single-dose vials for parenteral medications whenever possible.452 Category IA
- IV.H.5.
- IV.H.6.
- IV.H.7.
- IV.H.8.
-
IV.I.
Infection control practices for special lumbar puncture procedures
Wear a surgical mask when placing a catheter or injecting material into the spinal canal or subdural space (ie, during myelograms, lumbar puncture and spinal or epidural anesthesia).904, 905, 906, 907, 908, 909, 910, 911, 912, 916, 1005 Category IB
-
IV.J.
Worker safety
Adhere to federal and state requirements for protection of HCWs from exposure to bloodborne pathogens.738 Category IC
V. Transmission-Based Precautions
-
V.A.General principles
-
V.A.1.In addition to Standard Precautions, use Transmission-Based Precautions for patients with documented or suspected infection or colonization with highly transmissible or epidemiologically important pathogens for which additional precautions are needed to prevent transmission (see Appendix A).24, 93, 126, 141, 305, 805, 1006 Category IA
- V.A.2.
-
V.A.1.
-
V.B.Contact Precautions
-
V.B.1.Use Contact Precautions as recommended in Appendix A for patients with known or suspected infections or evidence of syndromes that represent an increased risk for contact transmission. For specific recommendations for use of Contact Precautions for colonization or infection with MDROs, consult the MDRO guideline, available at http://www.cdc.gov/ncidod/dhqp/pdf/ar/mdroGuideline2006.pdf.869
-
V.B.2.Patient placement
- V.B.2.a.
-
V.B.2.b.When single-patient rooms are in short supply, apply the following principles for making decisions on patient placement:
-
•Prioritize patients with conditions that may facilitate transmission (eg, uncontained drainage, stool incontinence) for single-patient room placement. Category II
- •
-
•If it becomes necessary to place a patient requiring Contact Precautions in a room with a patient who is not infected or colonized with the same infectious agent:
-
-Avoid placing patients on Contact Precautions in the same room with patients who have conditions that may increase the risk of adverse outcome from infection or that may facilitate transmission (eg, those who are immunocompromised, have open wounds, or have anticipated prolonged lengths of stay). Category II
-
-Ensure that patients are physically separated (ie, >3 feet apart) from each other. Draw the privacy curtain between beds to minimize opportunities for direct contact. Category II
- -
-
-
-
•
-
V.B.2.c.In long-term care and other residential settings, make decisions regarding patient placement on a case-by-case basis, balancing infection risks to other patients in the room, the presence of risk factors that increase the likelihood of transmission, and the potential adverse psychological impact on the infected or colonized patient.919, 920 Category II
-
V.B.2.d.In ambulatory settings, place patients who require Contact Precautions in an examination room or cubicle as soon as possible.20 Category II
-
V.B.3.Use of PPE
-
V.B.3.a.Gloves
-
V.B.3.b.Gowns
-
V.B.3.b.i.Wear a gown whenever it is anticipated that clothing will come in direct contact with the patient or potentially contaminated environmental surfaces or equipment in close proximity to the patient. Don a gown on entry into the room or cubicle. Remove the gown and observe hand hygiene before leaving the patient care environment.24, 88, 134, 744, 836 Category IB
- V.B.3.b.ii.
-
V.B.3.b.i.
-
V.B.3.a.
-
V.B.4.Patient transport
-
V.B.4.a.In acute care hospitals and long-term care and other residential settings, limit transport and movement of patients outside of the room to medically necessary purposes. Category II
-
V.B.4.b.When transport or movement in any health care setting is necessary, ensure that infected or colonized areas of the patient's body are contained and covered. Category II
-
V.B.4.c.Remove and dispose of contaminated PPE and perform hand hygiene before transporting patients on Contact Precautions. Category II
-
V.B.4.d.Don clean PPE to handle the patient at the transport destination. Category II
-
V.B.4.a.
-
V.B.5.Patient care equipment and instruments/devices
- V.B.5.a.
-
V.B.5.b.In acute care hospitals and long-term care and other residential settings, use disposable noncritical patient care equipment (eg, blood pressure cuffs) or implement patient-dedicated use of such equipment. If common use of equipment for multiple patients is unavoidable, clean and disinfect such equipment before use on another patient.24, 88, 795, 835, 836, 853, 1014 Category IB
-
V.B.5.c.In-home care settings
-
V.B.5.c.i.Limit the amount of nondisposable patient care equipment brought into the home of a patient on Contact Precautions. Whenever possible, leave patient care equipment in the home until discharge from home care services. Category II
-
V.B.5.c.ii.If noncritical patient care equipment (eg, stethoscope) cannot remain in the home, clean and disinfect items before taking them from the home using a low- to intermediate-level disinfectant. Alternatively, place contaminated reusable items in a plastic bag for transport and subsequent cleaning and disinfection. Category II
-
V.B.5.c.i.
-
V.B.5.d.In ambulatory settings, place contaminated reusable noncritical patient care equipment in a plastic bag for transport to a soiled utility area for reprocessing. Category II
-
V.B.6.Environmental measures
- Ensure that rooms of patients on Contact Precautions are prioritized for frequent cleaning and disinfection (eg, at least daily) with a focus on frequently touched surfaces (eg, bed rails, overbed table, bedside commode, lavatory surfaces in patient bathrooms, doorknobs) and equipment in the immediate vicinity of the patient.11, 24, 88, 745, 836 Category IB
-
V.B.7.Discontinue Contact Precautions after signs and symptoms of the infection have resolved or according to pathogen-specific recommendations in Appendix A. Category IB
-
V.B.1.
-
V.C.Droplet Precautions
-
V.C.1.Use Droplet Precautions as recommended in Appendix A for patients known or suspected infection with pathogens transmitted by respiratory droplets (ie, droplets > 5 μ) generated by a patient who is coughing, sneezing, or talking,14, 23, Steinberg, 1969 #1708,41, 95, 103, 111, 112, 754, 755, 987, 1015 Category IB
-
V.C.2.Patient placement
-
V.C.2.a.In acute care hospitals, place patients who require Droplet Precautions in a single-patient room when available Category II
- When single-patient rooms are in short supply, apply the following principles when making decisions on patient placement:
-
•Prioritize patients who have excessive cough and sputum production for single-patient room placement. Category II
- •
-
•If it becomes necessary to place patients who require Droplet Precautions in a room with a patient who does not have the same infection:
-
-Avoid placing patients on Droplet Precautions in the same room with patients who have conditions that may increase the risk of adverse outcome from infection or that may facilitate transmission (eg, those who are immunocompromised or have anticipated prolonged lengths of stay). Category II
- -
- -
-
-
-
•
-
V.C.2.b.In long-term care and other residential settings, make decisions regarding patient placement on a case-by-case basis after considering infection risks to other patients in the room and available alternatives.409 Category II
- V.C.2.c.
-
V.C.2.a.
-
V.C.3.Use of PPE
- V.C.3.a.
-
V.C.3.b.No recommendation for routinely wearing eye protection (eg, goggle or face shield) in addition to a mask, for close contact with patients who require Droplet Precautions. Unresolved issue
-
V.C.3.c.For patients with suspected or proven SARS, avian influenza or pandemic influenza, refer to the following websites for the most current recommendations: http://www.cdc.gov/ncidod/sars/; http://www.cdc.gov/flu/avian/; and http://www.pandemicflu.gov/.134, 1016, 1017
-
V.C.4.Patient transport
-
V.C.4.a.In acute care hospitals and long-term care and other residential settings, limit transport and movement of patients outside of the room to medically necessary purposes. Category II
-
V.C.4.b.If transport or movement in any health care setting is necessary, instruct the patient to wear a mask and follow respiratory hygiene/cough etiquette (see http://www.cdc.gov/flu/professionals/infectioncontrol/resphygiene.htm). Category IB
-
V.C.4.c.No mask is required for persons transporting patients on Droplet Precautions. Category II
-
V.C.4.d.Discontinue Droplet Precautions after signs and symptoms have resolved or according to pathogen-specific recommendations in Appendix A. Category IB
-
V.C.4.a.
-
V.C.1.
-
V.D.Airborne Precautions
- V.D.1.
-
V.D.2.Patient placement
-
V.D.2.a.In acute care hospitals and long-term care settings, place patients who require Airborne Precautions in an AIIR that has been constructed in accordance with current guidelines.11, 12, 13 Category IA/IC
-
V.D.2.a.i.Provide at least 6 (in an existing facility) or 12 (in new construction/renovation) air changes per hour.
-
V.D.2.a.ii.Direct exhaust of air to the outside. If it is not possible to exhaust air from an AIIR directly to the outside, the air may be returned to the air-handling system or adjacent spaces if all air is directed through HEPA filters.
- V.D.2.a.iii.
-
V.D.2.a.iv.Keep the AIIR door closed when not required for entry and exit.
-
V.D.2.a.i.
-
V.D.2.b.When an AIIR is not available, transfer the patient to a facility that has an available AIIR.12 Category II
-
V.D.2.c.In the event of an outbreak or exposure involving large numbers of patients who require Airborne Precautions:
-
•Consult an ICP before patient placement to determine the safety of an alternative room that does not meet engineering requirements for an AIIR.
-
•Place patients who are presumed to have the same infection (based on clinical presentation and diagnosis when known) together (cohort) in areas of the facility away from other patients, especially patients at increased risk for infection (eg, immunocompromised patients).
-
•Use temporary portable solutions (eg, exhaust fan) to create a negative-pressure environment in the converted area of the facility. Discharge air directly to the outside, away from people and air intakes, or direct all of the air through HEPA filters before it is introduced to other air spaces.12 Category II
-
•
-
V.D.2.d.In ambulatory settings:
- V.D.2.d.i.
-
V.D.2.d.ii.Place the patient in an AIIR as soon as possible. If an AIIR is not available, place a surgical mask on the patient and place the patient in an examination room. Once the patient leaves, the room should remain vacant for the appropriate time (generally 1 hour) to allow for a full exchange of air.11, 12, 122 Category IB/IC
- V.D.2.d.iii.
-
V.D.2.a.
-
V.D.3.Personnel restrictions
-
V.D.4.Use of PPE
-
V.D.4.a.Wear a fit-tested NIOSH-approved N95 or higher-level respirator for respiratory protection when entering the room or home of a patient when the following diseases are suspected or confirmed:
- •
-
•Smallpox (vaccinated and unvaccinated). Respiratory protection is recommended for all HCWs, including those with a documented “take” after smallpox vaccination due to the risk of a genetically engineered virus against which the vaccine may not provide protection, or of exposure to a very large viral load (from, eg, high-risk aerosol-generating procedures, immunocompromised patients, hemorrhagic or flat smallpox).108, 129 Category II
-
V.D.4.b.No recommendation is made regarding the use of PPE by HCWs who are presumed to be immune to measles (rubeola) or varicella-zoster based on history of disease, vaccine, or serologic testing when caring for an individual with known or suspected measles, chickenpox, or disseminated zoster due to difficulties in establishing definite immunity.1025, 1026 Unresolved issue
-
V.D.4.c.No recommendation is made regarding the type of PPE (ie, surgical mask or respiratory protection with a N95 or higher-level respirator) to be worn by susceptible HCWs who must have contact with patients with known or suspected measles, chickenpox, or disseminated herpes zoster. Unresolved issue
-
V.D.4.a.
-
V.D.5.Patient transport
-
V.D.5.a.In acute care hospitals and long-term care and other residential settings, limit transport and movement of patients outside of the room to medically necessary purposes. Category II
-
V.D.5.b.If transport or movement outside an AIIR is necessary, instruct the patient to wear a surgical mask, if possible, and to observe respiratory hygiene/cough etiquette.12 Category II
- V.D.5.c.
-
V.D.5.d.An HCW transporting a patient on Airborne Precautions does not need to wear a mask or respirator during transport if the patient is wearing a mask and infectious skin lesions are covered. Category II
-
V.D.5.a.
-
V.D.6.Exposure management
- Immunize or provide the appropriate immune globulin to susceptible persons as soon as possible after unprotected contact (ie, exposure) to a patient with measles, varicella, or smallpox: Category IA
- •
-
•Administer varicella vaccine to exposed susceptible persons within 120 hours after the exposure or administer varicella immune globulin (VZIG or an alternative product), when available, within 96 hours for high-risk persons in whom vaccine is contraindicated (eg, immunocompromised patients, pregnant women, newborns whose mother's varicella onset was < 5 days before or within 48 hours after delivery).887, 1033, 1034, 1035
- •
-
V.D.7.Discontinue Airborne Precautions according to pathogen-specific recommendations in Appendix A. Category IB
-
V.D.8.Consult the Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health Care Settings, 2005 12 and the Guideline for Environmental Infection Control in Health Care Facilities 11 for additional guidance on environment strategies for preventing transmission of tuberculosis in health care settings. The environmental recommendations in these guidelines may be applied to patients with other infections that necessitate Airborne Precautions.
VI. Protective Environment (see Table 4)
-
VI.A.
Place allogeneic HSCT patients in a PE as described in the Guideline to Prevent Opportunistic Infections in HSCT Patients,15 Guideline for Environmental Infection Control in Health Care Facilities,11 and Guidelines for Preventing Health Care–Associated Pneumonia, 200314 to reduce exposure to environmental fungi (eg, Aspergillus spp).157, 158 Category IB
-
VI.B.
No recommendation for placing patients with other medical conditions associated with increased risk for environmental fungal infections (eg, aspergillosis) in a PE.11 Unresolved issue
-
VI.C.For patients who require a PE, implement the following (see Table 5):11, 15
-
VI.C.1.Environmental controls
-
VI.C.1.a.Filtered incoming air using central or point-of-use HEPA filters capable of removing 99.97% of particles ≥ 0.3 μm in diameter.13 Category IB
-
VI.C.1.b.Directed room airflow with the air supply on one side of the room that moves air across the patient bed and out through an exhaust on the opposite side of the room.13 Category IB
- VI.C.1.c.
-
VI.C.1.d.Well-sealed rooms that prevent infiltration of outside air.13 Category IB
-
VI.C.1.e.At least 12 air changes per hour.13 Category IB
-
VI.C.1.a.
- VI.C.2.
-
VI.C.3.Avoid carpeting in hallways and patient rooms in areas.940 Category IB
- VI.C.4.
-
VI.C.1.
-
VI.D.
Minimize the time that patients who require a PE are outside their rooms for diagnostic procedures and other activities.11, 158, 944 Category IB
-
VI.E.During periods of construction, to prevent inhalation of respirable particles that could contain infectious spores, provide respiratory protection (eg, N95 respirator) to patients who are medically fit to tolerate a respirator when they are required to leave the PE.158, 944 Category II
-
VI.E.1.a.No recommendation for fit testing of patients who are using respirators. Unresolved issue
-
VI.E.1.b.No recommendation for use of particulate respirators when leaving the PE in the absence of construction. Unresolved issue
-
VI.E.1.a.
-
VI.F.Use of Standard and Transmission-Based Precautions in a PE
-
VI.F.1.Use Standard Precautions as recommended for all patient interactions. Category IA
- VI.F.2.
-
VI.F.3.Barrier precautions, (eg, masks, gowns, gloves) are not required for HCWs in the absence of suspected or confirmed infection in the patient or if they are not indicated according to Standard Precautions.15 Category II
-
VI.F.4.Implement Airborne Precautions for patients who require a PE and who also have an airborne infectious disease (eg, pulmonary or laryngeal tuberculosis, acute varicella-zoster). Category IA
-
VI.F.1.
GLOSSARY
Airborne infection isolation room (AIIR). Formerly known as a negative-pressure isolation room, an AIIR is a single-occupancy patient care room used to isolate persons with a suspected or confirmed airborne infectious disease. Environmental factors are controlled in AIIRs to minimize the transmission of infectious agents that are usually transmitted from person to person by droplet nuclei associated with coughing or aerosolization of contaminated fluids. AIIRs should provide negative pressure in the room (so that air flows under the door gap into the room), an air flow rate of 6 to 12 air changes per hour (ACH) (6 ACH for existing structures, 12 ACH for new construction or renovation), and direct exhaust of air from the room to the outside of the building or recirculation of air through a high-efficiency particulate air filter before returning to circulation. (MMWR 2003; 52 [RR-10]; MMWR 1994; 43 [RR-13).]
American Institute of Architects (AIA). A professional organization that has developed standards for building ventilation, the 2001Guidelines for Design and Construction of Hospital and Health Care Facilities, the development of which was supported by the AIA, Academy of Architecture for Health, and Facilities Guideline Institute, with assistance from the US Department of Health and Human Services and the National Institutes of Health, is the primary source of guidance for creating airborne infection isolation rooms and protective environments (http://www.aia.org/aah).
Ambulatory care setting. A facility that provides health care to patients who do not remain overnight; examples include hospital-based outpatient clinics, non–hospital-based clinics and physician offices, urgent care centers, surgicenters, free-standing dialysis centers, public health clinics, imaging centers, ambulatory behavioral health and substance abuse clinics, physical therapy and rehabilitation centers, and dental practices.
Bioaerosol. An airborne dispersion of particles containing whole or parts of biological entities, including bacteria, viruses, dust mites, fungal hyphae, and fungal spores. Such aerosols usually consist of a mixture of monodispersed and aggregate cells, spores, or viruses carried by other materials, such as respiratory secretions and/or inert particles. Infectious bioaerosols (ie, those containing biological agents capable of causing an infectious disease) can be generated from human sources (eg, expulsion from the respiratory tract during coughing, sneezing, talking, singing, suctioning, or wound irrigation), wet environmental sources (eg, high-volume air consitioning and cooling tower water with Legionella) or dry sources (eg, construction dust with spores produced by Aspergillus spp). Bioaerosols include large respiratory droplets and small droplet nuclei (Cole EC. AJIC 1998;26: 453-64).
Caregiver.. Any person who is not an employee of an organization, is not paid, and provides or assists in providing health care to a patient (eg, family member, friend) and acquire technical training as needed based on the tasks that must be performed.
Cohorting. In the context of this guideline, this term applies to the practice of grouping patients infected or colonized with the same infectious agent together to confine their care to one area and prevent contact with susceptible patients (cohorting patients). During outbreaks, health care personnel may be assigned to a cohort of patients to further limit opportunities for transmission (cohorting staff).
Colonization. Proliferation of microorganisms on or within body sites without detectable host immune response, cellular damage, or clinical expression. The presence of a microorganism within a host may occur with varying durations but may become a source of potential transmission. In many instances, colonization and carriage are synonymous.
Droplet nuclei. Microscopic particles < 5 μm in size that are the residue of evaporated droplets and are produced when a person coughs, sneezes, shouts, or sings. These particles can remain suspended in the air for prolonged periods and can be carried on normal air currents in a room or beyond, to adjacent spaces or areas receiving exhaust air.
Engineering controls. Removal or isolation of a workplace hazard through technology. An airborne infection isolation room, a protective environment, engineered sharps injury prevention device, and a sharps container are examples of engineering controls.
Epidemiologically important pathogen. An infectious agent that has one or more of the following characteristics: (1) readily transmissible, (2) a proclivity toward causing outbreaks, (3) possible association with a severe outcome, and (4) difficult to treat. Examples include Acinetobacter spp, Aspergillus spp, Burkholderia cepacia, Clostridium difficile, Klebsiella or Enterobacter spp, extended-spectrum beta-lactamase–producing gram-negative bacilli, methicillin-resistant Staphylococcus aureus, Pseudomonas aeruginosa, vancomycin-resistant enterococci, vancomycin-resistant Staphylococcus aureus, influenza virus, respiratory syncytial virus, rotavirus, severe acute respiratory syndrome coronavirus, noroviruses, and the hemorrhagic fever viruses.
Hand hygiene. A general term that applies to any one of the following: (1) handwashing with plain (nonantimicrobial) soap and water, (2) antiseptic handwashing (soap containing antiseptic agents and water), (3) antiseptic handrub (waterless antiseptic product, most often alcohol-based, rubbed on all surfaces of hands), or (4) surgical hand antisepsis (antiseptic handwash or antiseptic handrub performed preoperatively by surgical personnel to eliminate transient hand flora and reduce resident hand flora).558
Health care–associated infection (HAI). An infection that develops in a patient who is cared for in any setting where health care is delivered (eg, acute care hospital, chronic care facility, ambulatory clinic, dialysis center, surgicenter, home) and is related to receiving health care (ie, was not incubating or present at the time health care was provided). In ambulatory and home settings, HAI refers to any infection that is associated with a medical or surgical intervention. Because the geographic location of infection acquisition is often uncertain, the preferred term is considered to be health care-associated rather than health care-acquired.
Healthcare epidemiologist. A person whose primary training is medical (MD, DO) and/or masters- or doctorate-level epidemiology who has received advanced training in health care epidemiology. Typically these professionals direct or provide consultation to an infection control program in a hospital, long-term care facility, or health care delivery system (also see Infection control professional).
Health care personnel, health care worker (HCW). Any paid or unpaid person who works in a health care setting (eg, any person who has professional or technical training in a health care–related field and provides patient care in a health care setting or any person who provides services that support the delivery of health care such as dietary, housekeeping, engineering, maintenance personnel).
Hematopoietic stem cell transplantation (HSCT). Any transplantation of blood- or bone marrow–derived hematopoietic stem cells, regardless of donor type (eg, allogeneic or autologous) or cell source (eg, bone marrow, peripheral blood, or placental/umbilical cord blood), associated with periods of severe immunosuppression that vary with the source of the cells, the intensity of chemotherapy required, and the presence of graft versus host disease (MMWR 2000; 49: RR-10).
High-efficiency particulate air (HEPA) filter. An air filter that removes >99.97% of particles > 0.3 μm (the most penetrating particle size) at a specified flow rate of air. HEPA filters may be integrated into the central air handling systems, installed at the point of use above the ceiling of a room, or used as portable units (MMWR 2003; 52: RR-10).
Home care. A wide range of medical, nursing, rehabilitation, hospice, and social services delivered to patients in their place of residence (eg, private residence, senior living center, assisted living facility). Home health care services include care provided by home health aides and skilled nurses, respiratory therapists, dieticians, physicians, chaplains, and volunteers; provision of durable medical equipment; home infusion therapy; and physical, speech, and occupational therapy.
Immunocompromised patient. A patient whose immune mechanisms are deficient because of a congenital or acquired immunologic disorder (eg, human immunodeficiency virus infection, congenital immune deficiency syndromes), chronic diseases such as diabetes mellitus, cancer, emphysema, or cardiac failure, intensive care unit care, malnutrition, and immunosuppressive therapy of another disease process [eg, radiation, cytotoxic chemotherapy, anti–graft rejection medication, corticosteroids, monoclonal antibodies directed against a specific component of the immune system]). The type of infections for which an immunocompromised patient has increased susceptibility is determined by the severity of immunosuppression and the specific component(s) of the immune system that is affected. Patients undergoing allogeneic hematopoietic stem cell transplantation and those with chronic graft versus host disease are considered the most vulnerable to health care–associated infections. Immunocompromised states also make it more difficult to diagnose certain infections (eg, tuberculosis) and are associated with more severe clinical disease states than persons with the same infection and a normal immune system.
Infection. The transmission of microorganisms into a host after evading or overcoming defense mechanisms, resulting in the organism's proliferation and invasion within host tissue(s). Host responses to infection may include clinical symptoms or may be subclinical, with manifestations of disease mediated by direct organisms pathogenesis and/or a function of cell-mediated or antibody responses that result in the destruction of host tissues.
Infection control and prevention professional (ICP). A person whose primary training is in either nursing, medical technology, microbiology, or epidemiology and who has acquired specialized training in infection control. Responsibilities may include collection, analysis, and feedback of infection data and trends to health care providers; consultation on infection risk assessment, prevention, and control strategies; performance of education and training activities; implementation of evidence-based infection control practices or those mandated by regulatory and licensing agencies; application of epidemiologic principles to improve patient outcomes; participation in planning renovation and construction projects (eg, to ensure appropriate containment of construction dust); evaluation of new products or procedures on patient outcomes; oversight of employee health services related to infection prevention; implementation of preparedness plans; communication within the health care setting, with local and state health departments, and with the community at large concerning infection control issues; and participation in research. Certification in infection control is available through the Certification Board of Infection Control and Epidemiology.
Infection control and prevention program. A multidisciplinary program that includes a group of activities to ensure that recommended practices for the prevention of health care–associated infections are implemented and followed by health care workers, making the health care setting safe from infection for patients and health care personnel. The Joint Commission on Accreditation of Healthcare Organizations requires the following 5 components of an infection control program for accreditation: (1) surveillance: monitoring patients and health care personnel for acquisition of infection and/or colonization; (2) investigation: identification and analysis of infection problems or undesirable trends; (3) prevention: implementation of measures to prevent transmission of infectious agents and to reduce risks for device- and procedure-related infections; (4) control: evaluation and management of outbreaks; and (5) reporting: provision of information to external agencies as required by state and federal laws and regulations (see http://www.jcaho.org). The infection control program staff has the ultimate authority to determine infection control policies for a health care organization with the approval of the organization's governing body.
Long-term care facility (LTCF). A residential or outpatient facility designed to meet the biopsychosocial needs of persons with sustained self-care deficits. These include skilled nursing facilities, chronic disease hospitals, nursing homes, foster and group homes, institutions for the developmentally disabled, residential care facilities, assisted living facilities, retirement homes, adult day health care facilities, rehabilitation centers, and long-term psychiatric hospitals.
Mask. A term that applies collectively to items used to cover the nose and mouth and includes both procedure masks and surgical masks (see http://www.fda.gov/cdrh/ode/guidance/094.html#4).
Multidrug-resistant organism (MDRO). In general, a bacterium (excluding Mycobacterium tuberculosis) that is resistant to 1 or more classes of antimicrobial agents and usually is resistant to all but 1 or 2 commercially available antimicrobial agents (eg, methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, extended-spectrum beta-lactamase–producing or intrinsically resistant gram-negative bacilli).176
Nosocomial infection. Derived from 2 Greek words, “nosos” (disease) and “komeion” (to take care of), refers to any infection that develops during or as a result of an admission to an acute care facility (hospital) and was not incubating at the time of admission.
Personal protective equipment (PPE). A variety of barriers used alone or in combination to protect mucous membranes, skin, and clothing from contact with infectious agents. PPE includes gloves, masks, respirators, goggles, face shields, and gowns.
Procedure mask. A covering for the nose and mouth that is intended for use in general patient care situations. These masks generally attach to the face with ear loops rather than ties or elastic. Unlike surgical masks, procedure masks are not regulated by the Food and Drug Administration.
Protective environment. A specialized patient care area, usually in a hospital, with a positive air flow relative to the corridor (ie, air flows from the room to the outside adjacent space). The combination of high-efficiency particulate air filtration, high numbers (>12) of air changes per hour, and minimal leakage of air into the room creates an environment that can safely accommodate patients with a severely compromised immune system (eg, those who have received allogeneic hemopoietic stem cell transplantation) and decrease the risk of exposure to spores produced by environmental fungi. Other components include use of scrubbable surfaces instead of materials such as upholstery or carpeting, cleaning to prevent dust accumulation, and prohibition of fresh flowers or potted plants.
Quasi-experimental study. A study undertaken to evaluate interventions but do not use randomization as part of the study design. These studies are also referred to as nonrandomized, pre-/postintervention study designs. These studies aim to demonstrate causality between an intervention and an outcome but cannot achieve the level of confidence concerning an attributable benefit obtained through a randomized controlled trial. In hospitals and public health settings, randomized control trials often cannot be implemented due to ethical, practical, and urgency reasons; therefore, quasi-experimental design studies are commonly used. However, even if an intervention appears to be effective statistically, the question can be raised as to the possibility of alternative explanations for the result. Such a study design is used when it is not logistically feasible or ethically possible to conduct a randomized controlled trial, (eg, during outbreaks). Within the classification of quasi-experimental study designs, there is a hierarchy of design features that may contribute to validity of results (Harris et al. CID 2004:38: 1586).
Residential care setting. A facility in which people live, minimal medical care is delivered, and the psychosocial needs of the residents are provided for.
Respirator. A personal protective device worn by health care personnel over the nose and mouth to protect them from acquiring airborne infectious diseases due to inhalation of infectious airborne particles < 5 μm in size. These include infectious droplet nuclei from patients with Mycobacterium tuberculosis, variola virus [smallpox], or severe acute respiratory syndrome and dust particles that contain infectious particles, such as spores of environmental fungi (eg, Aspergillus spp). The Centers for Disease Control and Prevention's National Institute for Occupational Safety and Health (NIOSH) certifies respirators used in health care settings (see http://www.cdc.gov/niosh/topics/respirators/). The N95 disposable particulate, air-purifying respirator is the type used most commonly by health care personnel. Other respirators used include N-99 and N-100 particulate respirators, powered air-purifying respirators with high-efficiency filters, and nonpowered full-facepiece elastomeric negative pressure respirators. A listing of NIOSH-approved respirators can be found at http://www.cdc.gov/niosh/npptl/respirators/disp_part/particlist.html. Respirators must be used in conjunction with a complete respiratory protection program, as required by the Occupational Safety and Health Administration, which includes fit testing, training, proper selection of respirators, medical clearance, and respirator maintenance.
Respiratory hygiene/cough etiquette. A combination of measures designed to minimize the transmission of respiratory pathogens through droplet or airborne routes in health care settings. The components of respiratory hygiene/cough etiquette are (1) covering the mouth and nose during coughing and sneezing, (2) using tissues to contain respiratory secretions with prompt disposal into a no-touch receptacle, (3) offering a surgical mask to persons who are coughing to decrease contamination of the surrounding environment, and (4) turning the head away from others and maintaining spatial separation (ideally >3 feet) when coughing. These measures are targeted to all patients with symptoms of respiratory infection and their accompanying family members or friends beginning at the point of initial encounter with a health care setting (eg, reception/triage in emergency departments, ambulatory clinics, health care provider offices).126 (Srinivasin A ICHE 2004; 25: 1020; http://www.cdc.gov/flu/professionals/infectioncontrol/resphygiene.htm).
Safety culture. Shared perceptions of workers and management regarding the level of safety in the work environment. A hospital safety climate includes the following organizational components: (1) senior management support for safety programs, (2) absence of workplace barriers to safe work practices, (3) cleanliness and orderliness of the worksite, (4) minimal conflict and good communication among staff members, (5) frequent safety-related feedback/training by supervisors, and (6) availability of PPE and engineering controls.618
Source control. The process of containing an infectious agent either at the portal of exit from the body or within a confined space. The term is applied most frequently to containment of infectious agents transmitted by the respiratory route but could apply to other routes of transmission, (eg, a draining wound, vesicular or bullous skin lesions). Respiratory hygiene/cough etiquette that encourages individuals to “cover your cough” and/or wear a mask is a source control measure. The use of enclosing devices for local exhaust ventilation (eg, booths for sputum induction or administration of aerosolized medication) is another example of source control.
Standard precautions. A group of infection prevention practices that apply to all patients, regardless of suspected or confirmed diagnosis or presumed infection status. Standard precautions represents a combination and expansion of universal precautions778 and body substance isolation.1109 Standard precautions are based on the principle that all blood, body fluids, secretions, excretions except sweat, nonintact skin, and mucous membranes may contain transmissible infectious agents. Standard precautions include hand hygiene and, depending on the anticipated exposure, use of gloves, gown, mask, eye protection, or face shield. In addition, equipment or items in the patient environment likely to have been contaminated with infectious fluids must be handled in a manner to prevent transmission of infectious agents (eg, wear gloves for handling, contain heavily soiled equipment, properly clean and disinfect or sterilize reusable equipment before use on another patient).
Surgical mask. A device worn over the mouth and nose by operating room personnel during surgical procedures to protect both surgical patients and operating room personnel from transfer of microorganisms and body fluids. Surgical masks also are used to protect health care personnel from contact with large infectious droplets (> 5 μm in size). According to draft guidance issued by the Food and Drug Administration on May 15, 2003, surgical masks are evaluated using standardized testing procedures for fluid resistance, bacterial filtration efficiency, differential pressure (air exchange), and flammability to mitigate the risks to health associated with the use of surgical masks. These specifications apply to any masks that are labeled surgical, laser, isolation, or dental or medical procedure (http://www.fda.gov/cdrh/ode/guidance/094.html#4). Surgical masks do not protect against inhalation of small particles or droplet nuclei and should not be confused with particulate respirators that are recommended for protection against selected airborne infectious agents (eg, Mycobacterium tuberculosis).
Acknowledgments
The authors and HICPAC gratefully acknowledge Dr Larry Strausbaugh for his many contributions and valued guidance in the preparation of this guideline.
Appendix A. Type and Duration of Precautions Recommended for Selected Infections And conditions
Preamble
The mode(s) and risk of transmission for each specific disease agent listed in this appendix were reviewed. Principle sources consulted for the development of disease-specific recommendations for the appendix included infectious disease manuals and textbooks.831, 1039, 1040 The published literature was searched for evidence of person-to-person transmission in health care and non–health care settings with a focus on reported outbreaks that would assist in developing recommendations for all settings where health care is delivered. The following criteria were used to assign transmission-based precautions categories:
-
•
A transmission-based precautions category was assigned if there was strong evidence for person-to-person transmission via droplet, contact, or airborne routes in health care or non–health care settings and/or if patient factors (eg, diapered infants, diarrhea, draining wounds) increased the risk of transmission.
-
•
Transmission-based precautions category assignments reflect the predominant mode(s) of transmission.
-
•
If there was no evidence for person-to-person transmission by droplet, contact or airborne routes, then Standard Precautions were assigned.
-
•
If there was a low risk for person-to-person transmission and no evidence of health care-associated transmission, then Standard Precautions were assigned.
-
•
Standard precautions were assigned for bloodborne pathogens (eg, HBV, HCV, HIV) in accordance with CDC recommendations for universal precautions issued in 1988.778 Subsequent experience has confirmed the efficacy of Standard Precautions to prevent exposure to infected blood and body fluid.776, 777, 863
Additional information relevant to use of precautions was added in the comments column to assist the caregiver in decision-making. Citations were added as needed to support a change in or provide additional evidence for recommendations for a specific disease and for new infectious agents (eg, SARS-CoV, avian influenza) that have been added to Appendix A. The reader may refer to more detailed discussion concerning modes of transmission and emerging pathogens in the background text and for MDRO control in the MDRO Guideline.
| Precaution |
|||
|---|---|---|---|
| Infection/Condition | Type∗ | Duration† | Comments |
| Abscess | |||
| Draining, major | C | DI | No dressing or containment of drainage; until drainage stops or can be contained by dressing. |
| Draining, minor or limited | S | Dressing to cover and contain drainage. | |
| AIDS/HIV | S | Postexposure chemoprophylaxis for some blood exposures.865 | |
| Actinomycosis | S | Not transmitted from person to person. | |
| Adenovirus infection (see agent-specific guidance under gastroenteritis, conjuctivitis, pneumonia) | |||
| Amebiasis | S | Person-to-person transmission is rare. Transmission in settings for the mentally challenged and in a family group has been reported.1041 Use care when handling diapered infants and mentally challenged persons.1042 | |
| Anthrax | S | Infected patients do not generally pose a transmission risk. | |
| Cutaneous | S | Transmission through nonintact skin contact with draining lesions possible; thus, use Contact Precautions if a large amount of uncontained drainage is present. Handwashing with soap and water is preferable to the use of waterless alcohol-based antiseptics, because alcohol does not have sporicidal activity.979 | |
| Pulmonary | S | Not transmitted from person to person. | |
| Environmental: aerosolizable spore-containing powder or other substance | DE | Until decontamination of environment complete.203 Wear respirator (N95 mask or PAPR), protective clothing; decontaminate persons with powder on them (http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5135a3.htm). | |
| Hand hygiene: Handwashing for 30 to 60 seconds with soap and water or 2% chlorhexidene gluconate after spore contact. (Alcohol handrubs are inactive against spores.)979 | |||
| Postexposure prophylaxis after environmental exposure: 60 days of antimicrobials (either doxycycline, ciprofloxacin, or levofloxacin) and postexposure vaccine under IND. | |||
| Antibiotic-associated colitis (see Clostridium difficile) | |||
| Arthropod-borne viral encephalitides (eastern, western, Venezuelan equine encephalomyelitis; St Louis, California encephalitis; west Nile virus) and viral fevers (dengue, yellow fever, Colorado tick fever) | S | Not transmitted from person to person except rarely by transfusion, and for West Nile virus by organ transplant, breastmilk or transplacentally.528, 1043 Install screens in windows and doors in endemic areas. | |
| Use DEET-containing mosquito repellants and clothing to cover extremities. | |||
| Ascariasis | S | Not transmitted from person to person. | |
| Aspergillosis | S | Institute Contact Precautions and Airborne Precautions if massive soft tissue infection with copious drainage and repeated irrigations required.154 | |
| Avian influenza (see influenza, avian below) | |||
| Babesiosis | S | Not transmitted from person to person except rarely by transfusion. | |
| Blastomycosis, North American, cutaneous or pulmonary | S | Not transmitted from person to person. | |
| Botulism | S | Not transmitted from person to person. | |
| Bronchiolitis (see respiratory infections in infants and young children) | C | DI | Use mask according to Standard Precautions. |
| Brucellosis (undulant, Malta, Mediterranean fever) | S | Not transmitted from person to person except rarely through banked spermatozoa and sexual contact.1044, 1045 Provide antimicrobial prophylaxis following laboratory exposure.1046 | |
| Campylobacter gastroenteritis (see gastroenteritis) | |||
| Candidiasis, all forms, including mucocutaneous | S | ||
| Cat-scratch fever (benign inoculation lymphoreticulosis) | S | Not transmitted from person to person. | |
| Cellulitis | S | ||
| Chancroid (soft chancre) (Haemophilus ducreyi) | S | Transmitted sexually from person to person. | |
| Chickenpox (see varicella) | |||
| Chlamydia trachomatis | |||
| Conjunctivitis | S | ||
| Genital (lymphogranuloma venereum) | S | ||
| Pneumonia (infants ≤ 3 mos. of age)) | S | ||
| Chlamydia pneumoniae | S | Outbreaks in institutionalized populations are rarely reported.1047, 1048 | |
| Cholera (see gastroenteritis) | |||
| Closed-cavity infection | |||
| Open drain in place; limited or minor drainage | S | Contact Precautions if copious uncontained drainage is present. | |
| No drain or closed drainage system in place | S | ||
| Clostridium spp | |||
| C botulinum | S | Not transmitted from person to person. | |
| C difficile (see gastroenteritis, C difficile) | C | DI | |
| C perfringens | |||
| Food poisoning | S | Not transmitted from person to person. | |
| Gas gangrene | S | Transmission from person to person is rare; 1 outbreak in a surgical setting has been reported.1053 Use Contact Precautions if wound drainage is extensive. | |
| Coccidioidomycosis (valley fever) | |||
| Draining lesions | S | Not transmitted from person to person except under extraordinary circumstances, because the infectious arthroconidial form of Coccidioides immitis is not produced in humans.1049, 1050 | |
| Pneumonia | S | Not transmitted from person to person except under extraordinary circumstances (eg, inhalation of aerosolized tissue phase endospores during necropsy, transplantation of infected lung), because the infectious arthroconidial form of C immitis is not produced in humans.1050, 1051 | |
| Colorado tick fever | S | Not transmitted from person to person. | |
| Congenital rubella | C | Until age 1 year | Standard Precautions if nasopharyngeal and urine cultures are repeatedly negative after age 3 months. |
| Conjunctivitis | |||
| Acute bacterial | S | ||
| Chlamydial | S | ||
| Gonococcal | S | ||
| Acute viral (acute hemorrhagic) | C | DI | Adenovirus most common; enterovirus 70,1052 Coxsackie virus A241054 also associated with community outbreaks. Highly contagious; outbreaks in eye clinics, pediatric and neonatal settings, institutional settings reported. Eye clinics should follow Standard Precautions when handling patients with conjunctivitis. Routine use of infection control measures in the handling of instruments and equipment will prevent the occurrence of outbreaks in this and other settings.458, 459, 812, 1054, 1055, 1056 |
| Corona virus associated with SARS (SARS-CoV) (see severe acute respiratory syndrome) | |||
| Coxsackie virus disease (see enteroviral infection) | |||
| Creutzfeldt-Jakob disease (CJD, vCJD) | S | Use disposable instruments or special sterilization/disinfection for surfaces and objects contaminated with neural tissue if CJD or vCJD has not been ruled out; no special burial procedures.1057 | |
| Croup (see respiratory infections in infants and young children) | |||
| Crimean-Congo Fever (see viral hemorrhagic fever) | S | ||
| Cryptococcosis | S | Not transmitted from person to person, except rarely through tissue and corneal transplantation.1058, 1059 | |
| Cryptosporidiosis (see gastroenteritis) | |||
| Cysticercosis | S | Not transmitted from person to person. | |
| Cytomegalovirus infection, including in neonates and immunosuppressed patients | S | No additional precautions for pregnant HCWs. | |
| Decubitus ulcer (see Pressure ulcer) | |||
| Dengue fever | S | Not transmitted from person to person. | |
| Diarrhea, acute-infective etiology suspected (see gastroenteritis) | |||
| Diphtheria | |||
| Cutaneous | C | CN | Until 2 cultures obtained 24 hours apart are negative. |
| Pharyngeal | D | CN | Until 2 cultures obtained 24 hours apart are negative. |
| Ebola virus (see viral hemorrhagic fevers) | |||
| Echinococcosis (hydatidosis) | S | Not transmitted from person to person. | |
| Echovirus (see enteroviral infection) | |||
| Encephalitis or encephalomyelitis (see specific etiologic agents) | |||
| Endometritis (endomyometritis) | S | ||
| Enterobiasis (pinworm disease, oxyuriasis) | S | ||
| Enterococcus spp (see multidrug-resistant organisms if epidemiologically significant or vancomycin-resistant) | |||
| Enterocolitis, Clostridium difficile (see C difficile, gastroenteritis) | |||
| Enteroviral infections (ie, group A and B Coxsackie viruses and Echo viruses) (excludes polio virus) | S | Use Contact Precautions for diapered or incontinent children for duration of illness and to control institutional outbreaks. | |
| Epiglottitis, due to Haemophilus influenzae type b | D | U 24 hours | (See specific disease agents for epiglottitis due to other etiologies.) |
| Epstein-Barr virus infection, including infectious mononucleosis | S | ||
| Erythema infectiosum (also see parvovirus B19) | |||
| Escherichia coli gastroenteritis (see gastroenteritis) | |||
| Food poisoning | |||
| Botulism | S | Not transmitted from person to person. | |
| Clostridium perfringens or C welchii | S | Not transmitted from person to person. | |
| Staphylococcal | S | Not transmitted from person to person. | |
| Furunculosis, staphylococcal | S | Contact if drainage not controlled. Follow institutional policies if MRSA. | |
| Infants and young children | C | DI | |
| Gangrene (gas gangrene) | S | Not transmitted from person to person. | |
| Gastroenteritis | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks for gastroenteritis caused by all of the agents listed below. | |
| Adenovirus | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Campylobacter spp | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Cholera (Vibrio cholerae) | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Clostridium difficile | C | DI | Discontinue antibiotics if appropriate. Do not share electronic thermometers;851, 852 ensure consistent environmental cleaning and disinfection. Hypochlorite solutions may be required for cleaning if transmission continues.845 Handwashing with soap and water is preferred because of the absence of sporicidal activity of alcohol in waterless antiseptic handrubs.979 |
| Cryptosporidium spp | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Escherichia coli | |||
| Enteropathogenic O157:H7 and other shiga toxin–producing strains | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Other species | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Giardia lamblia | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Noroviruses | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. Persons who clean areas heavily contaminated with feces or vomitus may benefit from wearing masks, because virus can be aerosolized from these body substances;142, 147, 148 ensure consistent environmental cleaning and disinfection with focus on restrooms even when apparently unsoiled.272, 1060 Hypochlorite solutions may be required when there is continued transmission.289, 290, 291 Alcohol is less active, but there is no evidence that alcohol antiseptic handrubs are not effective for hand decontamination.293 Cohorting of affected patients to separate airs paces and toilet facilities may help interrupt transmission during outbreaks. | |
| Rotavirus | C | DI | Ensure consistent environmental cleaning and disinfection and frequent removal of soiled diapers. Prolonged shedding may occur in both immunocompetent and immunocompromised children and the elderly.930, 931 |
| Salmonella species (including S typhi) | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Shigella species (bacillary dysentery) | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Vibrio parahaemolyticus | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Viral (if not covered elsewhere) | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| Yersinia enterocolitica | S | Use Contact Precautions for diapered or incontinent persons for the duration of illness or to control institutional outbreaks. | |
| German measles (see rubella; see congenital rubella) | |||
| Giardiasis (see gastroenteritis) | |||
| Gonococcal ophthalmia neonatorum (gonorrheal ophthalmia, acute conjunctivitis of newborn) | S | ||
| Gonorrhea | S | ||
| Granuloma inguinale (donovanosis, granuloma venereum) | S | ||
| Guillain-Barré syndrome | S | Not an infectious condition. | |
| Haemophilus influenzae (see disease-specific recommendations) | |||
| Hand, foot, and mouth disease (see enteroviral infection) | |||
| Hansen's disease (see leprosy) | |||
| Hantavirus pulmonary syndrome | S | Not transmitted from person to person. | |
| Helicobacter pylori | S | ||
| Hepatitis, viral | |||
| Type A | S | Provide hepatitis A vaccine postexposure as recommended.1061 | |
| Diapered or incontinent patients | C | Maintain Contact Precautions for the duration of hospitalization in infants and children under age 3 years, for 2 weeks after onset of symptoms in children age 3 to 14 years, and for 1 week after onset of symptoms in those over age 14 year.831, 1062, 1063 | |
| Type B-HBsAg positive; acute or chronic | S | See specific recommendations for care of patients in hemodialysis centers.776 | |
| Type C and other unspecified non-A, non-B | S | See specific recommendations for care of patients in hemodialysis centers.776 | |
| Type D (seen only with hepatitis B) | S | ||
| Type E | S | Use Contact Precautions for diapered or incontinent individuals for the duration of illness.1064 | |
| Type G | S | ||
| Herpangina (see enteroviral infection) | |||
| Hookworm | S | ||
| Herpes simplex (Herpesvirus hominis) | |||
| Encephalitis | S | ||
| Mucocutaneous, disseminated or primary, severe | C | Until lesions dry and crusted | |
| Mucocutaneous, recurrent (skin, oral, genital) | S | ||
| Neonatal | C | Until lesions dry and crusted | Also for asymptomatic, exposed infants delivered vaginally or by C-section and if mother has active infection and membranes have been ruptured for more than 4 to 6 hours until infant surface cultures obtained at 24 to 36 hours of age negative after 48 hours of incubation.1065, 1066 |
| Herpes zoster (varicella-zoster) (shingles) | |||
| Disseminated disease in any patient | A,C | DI | Susceptible HCWs should not enter room if immune caregivers are available; no recommendation for protection of immune HCWs; no recommendation for type of protection (ie surgical mask or respirator) for susceptible HCWs. |
| Localized disease in immunocompromised patient until disseminated infection ruled out | |||
| Localized in patient with intact immune system with lesions that can be contained/covered | S | DI | Susceptible HCWs should not provide direct patient care when other immune caregivers are available. |
| Histoplasmosis | S | Not transmitted from person to person. | |
| Human immunodeficiency virus (HIV) | S | Postexposure chemoprophylaxis for some blood exposures.864 | |
| Human metapneumovirus | C | DI | HAI reported,1067 but the route of transmission is not established.821 Assumed to be contact transmission as for RSV since the viruses are closely related and have similar clinical manifestations and epidemiology. Wear masks according to Standard Precautions. |
| Impetigo | C | U 24 hours | |
| Infectious mononucleosis | S | ||
| Influenza | |||
| Human (seasonal influenza) | D | 5 days except DI in immuno- compromised persons | Single patient room when available or cohort; avoid placement with high-risk patients; mask patient when transported out of room; chemoprophylaxis/vaccine to control/prevent outbreaks.609 Use of gown and gloves according to Standard Precautions may be especially important in pediatric settings. Duration of precautions for immunocompromised patients cannot be defined; prolonged duration of viral shedding (ie for several weeks) has been observed; implications for transmission are unknown.928 |
| Avian (eg, H5N1, H7, H9 strains) | See http://www.cdc.gov/flu/avian/professional/infect-control.htm for current avian influenza guidance. | ||
| Pandemic influenza (also a human influenza virus) | D | 5 days from onset of symptoms | See http://www.pandemicflu.gov for current pandemic influenza guidance. |
| Kawasaki syndrome | S | Not an infectious condition. | |
| Lassa fever (see viral hemorrhagic fevers) | |||
| Legionnaires' disease | S | Not transmitted from person to person. | |
| Leprosy | S | ||
| Leptospirosis | S | Not transmitted from person to person; see | |
| Lice | http://www.cdc.gov/ncidod/dpd/parasites/lice/default.htm. | ||
| Head (pediculosis) | C | U 4 hours | |
| Body | S | Transmitted person to person through infested clothing. Wear gown and gloves when removing clothing; bag and wash clothes according to CDC guidance. | |
| Pubic | S | Transmitted person to person through sexual contact. | |
| Listeriosis (Listeria monocytogenes) | S | Person-to-person transmission rare; cross-transmission in neonatal settings reported.1068, 1069, 1070, 1071 | |
| Lyme disease | S | Not transmitted from person to person. | |
| Lymphocytic choriomeningitis | S | Not transmitted from person to person. | |
| Lymphogranuloma venereum | S | ||
| Malaria | S | Not transmitted from person to person except rarely through transfusion and due to failure to follow Standard Precautions during patient care.1072, 1073, 1074, 1075 Install screens in windows and doors in endemic areas. Use DEET-containing mosquito repellants and clothing to cover extremities. | |
| Marburg virus disease (see viral hemorrhagic fevers) | |||
| Measles (rubeola) | A | 4 days after onset of rash; DI in immune compromised | Susceptible HCWs should not enter room if immune care providers are available; no recommendation for face protection for immune HCW; no recommendation for type of face protection for susceptible HCWs (ie, mask or respirator).1023, 1025 For exposed susceptible HCWs, postexposure vaccine within 72 hours or immune globulin within 6 days when available.17, 1028, 1030 Place exposed susceptible patients on Airborne Precautions and exclude susceptible HCWs from duty from day 5 after first exposure to day 21 after last exposure, regardless of postexposure vaccine.17 |
| Melioidosis, all forms | S | Not transmitted from person to person. | |
| Meningitis | |||
| Aseptic (nonbacterial or viral; also see enteroviral infections) | S | Contact for infants and young children. | |
| Bacterial, gram-negative enteric, in neonates | S | ||
| Fungal | S | ||
| Haemophilus influenzae, type b known or suspected | D | U 24 hours | |
| Listeria monocytogenes (See listeriosis) | S | ||
| Neisseria meningitidis (meningococcal) known or suspected | D | U 24 hours | See meningococcal disease below. |
| Streptococcus pneumoniae | S | ||
| Mycobacterium tuberculosis | S | Concurrent, active pulmonary disease or draining cutaneous lesions may necessitate addition of Contact and/or Airborne Precautions. | |
| For children, airborne precautions until active tuberculosis ruled out in visiting family members (see tuberculosis below).42 | |||
| Other diagnosed bacterial | S | ||
| Meningococcal disease: sepsis, pneumonia, meningitis | D | U 24 hours | Postexposure chemoprophylaxis for household contacts, HCWs exposed to respiratory secretions; postexposure vaccine only to control outbreaks.15, 17 |
| Molluscum contagiosum | S | ||
| Monkeypox | A,C | A-Until monkeypox confirmed and smallpox excluded | See http://www.cdc.gov/ncidod/monkeypox for most current recommendations. Transmission in hospital settings unlikely.267 Preexposure and postexposure smallpox vaccine recommended for exposed HCWs. |
| C-Until lesions crusted | |||
| Mucormycosis | S | ||
| Multidrug-resistant organisms (MDROs), infection or colonization (eg, MRSA, VRE, VISA/VRSA, ESBLs, resistant S. pneumoniae) | S/C | MDROs judged by the infection control program, based on local, state, regional, or national recommendations, to be of clinical and epidemiologic significance. Contact Precautions recommended in settings with evidence of ongoing transmission, acute care settings with increased risk for transmission or wounds that cannot be contained by dressings. See recommendations for management options in Management of Multidrug-Resistant Organisms In Health care Settings, 2006.868 Contact state health department for guidance regarding new or emerging MDROs. | |
| Mumps (infectious parotitis) | D | U 9 days | After onset of swelling; susceptible HCWs should not provide care if immune caregivers are available. (Note: Recent assessment of outbreaks in healthy 18- to 24-year-olds has indicated that salivary viral shedding occurred early in the course of illness and that 5 days of isolation after onset of parotitis may be appropriate in community settings; however, the implications for health care personnel and high-risk patient populations remain to be clarified.) |
| Mycobacteria, nontuberculosis (atypical) | Not transmitted person-to-person. | ||
| Pulmonary | S | ||
| Wound | S | ||
| Mycoplasma pneumonia | D | DI | |
| Necrotizing enterocolitis | S | Contact Precautions when cases clustered temporally.1076, 1077, 1078, 1079 | |
| Nocardiosis, draining lesions, or other presentations | S | Not transmitted person-to-person. | |
| Norovirus (see gastroenteritis) | |||
| Norwalk agent gastroenteritis (see gastroenteritis) | |||
| Orf | S | ||
| Parainfluenza virus infection, respiratory in infants and young children | C | DI | Viral shedding may be prolonged in immunosuppressed patients.1005, 1006 Reliability of antigen testing to determine when to remove patients with prolonged hospitalizations from Contact Precautions uncertain. |
| Parvovirus B19 (Erythema infectiosum) | D | Maintain precautions for duration of hospitalization when chronic disease occurs in immunocompromised patients. For patients with transient aplastic crisis or red cell crisis, maintain precautions for 7 days. Duration of precautions for immunosuppressed patients with persistently positive PCR not defined, but transmission has occurred.927 | |
| Pediculosis (lice) | C | U 24 hours after treatment | |
| Pertussis (whooping cough) | D | U 5 days | Single patient room preferred. Cohorting an option. Postexposure chemoprophylaxis for household contacts and HCWs with prolonged exposure to respiratory secretions.861 Recommendations for Tdap vaccine in adults under development. |
| Pinworm infection (Enterobiasis) | S | ||
| Plague (Yersinia pestis) | |||
| Bubonic | S | ||
| Pneumonic | D | U 48 hours | Antimicrobial prophylaxis for exposed HCW.207 |
| Pneumonia | |||
| Adenovirus | D, C | DI | Outbreaks in pediatric and institutional settings reported.375, 1080, 1081, 1082 In immunocompromised hosts, extend duration of Droplet and Contact Precautions due to prolonged shedding of virus.929 |
| Bacterial not listed elsewhere (including gram-negative bacterial) | S | ||
| B cepacia in patients with CF, including respiratory tract colonization | C | Unknown | Avoid exposure to other persons with CF; private room preferred. Criteria for D/C precautions not established. See the Cystic Fibrosis Foundation guidelines.20 |
| B cepacia in patients without CF (see multidrug-resistant organisms) | |||
| Chlamydia | S | ||
| Fungal | S | ||
| Haemophilus influenzae, type b | |||
| Adults | S | ||
| Infants and children | D | U 24 hours | |
| Legionella spp | S | ||
| Meningococcal | D | U 24 hours | See meningococcal disease above. |
| Multidrug-resistant bacterial (see multidrug-resistant organisms) | |||
| Mycoplasma (primary atypical pneumonia) | D | DI | |
| Pneumococcal pneumonia | S | Use Droplet Precautions if evidence of transmission within a patient care unit or facility.196, 197, 198, 1083 | |
| Pneumocystis jiroveci (Pneumocystis carinii) | S | Avoid placement in the same room with an immunocompromised patient. | |
| Staphylococcus aureus | S | For MRSA, see MDROs. | |
| Streptococcus, group A | |||
| Adults | D | U 24 hours | See streptococcal disease (group A streptococcus) below. |
| Contact precautions if skin lesions present. | |||
| Infants and young children | D | U 24 hours | Contact Precautions if skin lesions present. |
| Varicella-zoster (see varicella-zoster) | |||
| Viral | |||
| Adults | S | ||
| Infants and young children (see respiratory infectious disease, acute, or specific viral agent) | |||
| Poliomyelitis | C | DI | |
| Pressure ulcer (decubitus ulcer, pressure sore) infected | |||
| Major | C | DI | If no dressing or containment of drainage; until drainage stops or can be contained by dressing. |
| Minor or limited | S | If dressing covers and contains drainage. | |
| Prion disease (See Creutzfeld-Jacob Disease) | |||
| Psittacosis (ornithosis) (Chlamydia psittaci) | S | Not transmitted from person to person. | |
| Q fever | S | ||
| Rabies | S | Person-to-person transmission is rare; transmission via corneal, tissue and organ transplants has been reported.537, 1084 If patient has bitten another individual or saliva has contaminated an open wound or mucous membrane, wash exposed area thoroughly and administer postexposure prophylaxis.1085 | |
| Rat-bite fever (Streptobacillus moniliformis disease, Spirillum minus disease) | S | Not transmitted from person to person. | |
| Relapsing fever | S | Not transmitted from person to person. | |
| Resistant bacterial infection or colonization (see multidrug-resistant organisms) | |||
| Respiratory infectious disease, acute (if not covered elsewhere) | |||
| Adults | S | ||
| Infants and young children | C | DI | Also see syndromes or conditions listed in Table 2. |
| Respiratory syncytial virus infection, in infants, young children and immunocompromised adults | C | DI | Wear mask according to Standard Precautions24, 116, 117 In immunocompromised patients, extend the duration of Contact Precautions due to prolonged shedding.926 Reliability of antigen testing to determine when to remove patients with prolonged hospitalizations from Contact Precautions uncertain. |
| Reye's syndrome | S | Not an infectious condition. | |
| Rheumatic fever | S | Not an infectious condition. | |
| Rhinovirus | D | DI | Droplet most important route of transmission.104, 1086 Outbreaks have occurred in NICUs and LTCFs.411, 1087, 1088 Add Contact Precautions if copious moist secretions and close contact likely to occur (eg, young infants).111, 831 |
| Rickettsial fevers, tickborne (Rocky Mountain spotted fever, tickborne typhus fever) | S | Not transmitted from person to person except rarely through transfusion. | |
| Rickettsialpox (vesicular rickettsiosis) | S | Not transmitted from person to person. | |
| Ringworm (dermatophytosis, dermatomycosis, tinea) | S | Rarely, outbreaks have occurred in health care settings, (eg, NICU,1089 rehabilitation hospital1090). Use Contact Precautions for outbreak. | |
| Ritter's disease (staphylococcal scalded skin syndrome) | C | DI | See staphylococcal disease and scalded skin syndrome below. |
| Rocky Mountain spotted fever | S | Not transmitted from person to person except rarely through transfusion. | |
| Roseola infantum (exanthem subitum; caused by HHV-6) | S | ||
| Rotavirus infection (see gastroenteritis) | |||
| Rubella (German measles) (also see congenital rubella) | D | U 7 days after onset of rash | Susceptible HCWs should not enter room if immune caregivers are available. No recommendation for wearing face protection (eg, a surgical mask) if immune. Pregnant women who are not immune should not care for these patients.17, 33 Administer vaccine within 3 days of exposure to nonpregnant susceptible individuals. Place exposed susceptible patients on Droplet Precautions; exclude susceptible health care personnel from duty from day 5 after first exposure to day 21 after last exposure, regardless of postexposure vaccine. |
| Rubeola (see measles) | |||
| Salmonellosis (see gastroenteritis) | |||
| Scabies | C | U 24 | |
| Scalded skin syndrome, staphylococcal | C | DI | See staphylococcal disease and scalded skin syndrome below. |
| Schistosomiasis (bilharziasis) | S | ||
| Severe acute respiratory syndrome (SARS) | A, D,C | DI plus 10 days after resolution of fever, provided respiratory symptoms are absent or improving | Airborne Precautions preferred; D if AIIR unavailable. N95 or higher-level respiratory protection; surgical mask if N95 is unavailable; eye protection (goggles, face shield); aerosol-generating procedures and “supershedders” are at highest risk for transmission through small droplet nuclei and large droplets.93, 94, 96Vigilant environmental disinfection necessary (see http://www.cdc.gov/ncidod/sars). |
| Shigellosis (see gastroenteritis) | |||
| Smallpox (variola; see vaccinia for management of vaccinated persons) | A,C | DI | Until all scabs have crusted and separated (3 to 4 weeks). Nonvaccinated HCWs should not provide care when immune HCWs are available; N95 or higher-level respiratory protection for susceptible and successfully vaccinated individuals; postexposure vaccine within 4 days of exposure protective.108, 129, 1034, 1035, 1036 |
| Sporotrichosis | S | ||
| Spirillum minor disease (rat-bite fever) | S | Not transmitted from person to person. | |
| Staphylococcal disease (S. aureus) | |||
| Skin, wound, or burn | |||
| Major | C | DI | No dressing, or dressing does not adequately contain drainage. |
| Minor or limited | S | Dressing adequatelys cover and contain drainage. | |
| Enterocolitis | S | Use Contact Precautions for diapered or incontinent children for duration of illness. | |
| Multidrug-resistant (see multidrug-resistant organisms) | |||
| Pneumonia | S | ||
| Scalded skin syndrome | C | DI | Consider health care personnel as potential source of nursery, NICU outbreak.1091 |
| Toxic shock syndrome | S | ||
| Streptobacillus moniliformis disease (rat-bite fever) | S | Not transmitted from person to person. | |
| Streptococcal disease (group A streptococcus) | |||
| Skin, wound, or burn | |||
| Major | C,D | U 24 hours | No dressing, or dressing does not adequately contain drainage. |
| Minor or limited | S | Dressing covers and adequately contains drainage. | |
| Endometritis (puerperal sepsis) | S | ||
| Pharyngitis in infants and young children | D | U 24 hours | |
| Pneumonia | D | U 24 hours | |
| Scarlet fever in infants and young children | D | U 24 hours | |
| Serious invasive disease | D | U24 hours | Outbreaks of serious invasive disease have occurred secondary to transmission among patients and HCWs.162, 968, 1092, 1093, 1094 |
| Contact Precautions for draining wound as above; follow recommendations for antimicrobial prophylaxis in selected conditions.160 | |||
| Streptococcal disease (group B streptococcus), neonatal | S | ||
| Streptococcal disease (not group A or B) unless covered elsewhere | S | ||
| Multidrug-resistant (see multidrug-resistant organisms) | |||
| Strongyloidiasis | S | ||
| Syphilis | |||
| Latent (tertiary) and seropositivity without lesions | S | ||
| Skin and mucous membrane, including congenital, primary, secondary | S | ||
| Tapeworm disease | |||
| Hymenolepis nana | S | Not transmitted from person to person. | |
| Taenia solium (pork) | S | ||
| Other | S | ||
| Tetanus | S | Not transmitted from person to person. | |
| Tinea (eg, dermatophytosis, dermatomycosis, ringworm) | S | Rare episodes of person-to-person transmission. | |
| Toxoplasmosis | S | Transmission from person to person is rare; vertical transmission from mother to child, transmission through organs and blood transfusion rare. | |
| Toxic shock syndrome (staphylococcal disease, streptococcal disease) | S | Droplet Precautions for the first 24 hours after implementation of antibiotic therapy if group A streptococcus is a likely etiology. | |
| Trachoma, acute | S | ||
| Transmissible spongiform encephalopathy (see Creutzfeld-Jacob disease, CJD, vCJD) | |||
| Trench mouth (Vincent's angina) | S | ||
| Trichinosis | S | ||
| Trichomoniasis | S | ||
| Trichuriasis (whipworm disease) | S | ||
| Tuberculosis (M. tuberculosis) | |||
| Extrapulmonary, draining lesion) | A,C | Discontinue precautions only when patient is improving clinically and drainage has ceased or there are 3 consecutive negative cultures of continued drainage.1021, 1022 Examine for evidence of active pulmonary tuberculosis. | |
| Extrapulmonary, no draining lesion, meningitis | S | Examine for evidence of pulmonary tuberculosis. For infants and children, use Airborne Precautions until active pulmonary tuberculosis in visiting family members ruled out.42 | |
| Pulmonary or laryngeal disease, confirmed | A | Discontinue precautions only when patient on effective therapy is improving clinically and has three consecutive sputum smears negative for acid-fast bacilli collected on separate days (see http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5417a1.htm?s_cid=rr5417a1_e).12 | |
| Pulmonary or laryngeal disease, suspected | A | Discontinue precautions only when the likelihood of infectious TB disease is deemed negligible, and either there is another diagnosis that explains the clinical syndrome or the results of three sputum smears for AFB are negative. The 3 sputum specimens should be collected 8 to 24 hours apart, and at least 1 specimen should be an early-morning specimen | |
| Skin-test positive with no evidence of current active disease | S | ||
| Tularemia | |||
| Draining lesion | S | Not transmitted from person to person. | |
| Pulmonary | S | Not transmitted from person to person. | |
| Typhoid (Salmonella typhi) fever (see gastroenteritis) | |||
| Typhus | |||
| Rickettsia prowazekii (Epidemic or Louse-borne typhus) | S | Transmitted from person to person through close personal or clothing contact. | |
| Rickettsia typhi | S | Not transmitted from person to person. | |
| Urinary tract infection (including pyelonephritis), with or without urinary catheter | S | ||
| Vaccinia (vaccination site, adverse events after vaccination)∗ | Only vaccinated HCWs have contact with active vaccination sites and care for persons with adverse vaccinia events; if unvaccinated, only HCWs without contraindications to vaccine may provide care. | ||
| Vaccination site care (including autoinoculated areas) | S | Vaccination recommended for vaccinators; for newly vaccinated HCWs: semipermeable dressing over gauze until scab separates, with dressing change as fluid accumulates, ∼3 to 5 days; gloves, hand hygiene for dressing change; vaccinated HCW or HCW without contraindication to vaccine for dressing changes.205, 221, 225 | |
| Eczema vaccinatum | C | Until lesions dry and crusted, scabs separated | For contact with virus-containing lesions and exudative material. |
| Fetal vaccinia | C | ||
| Generalized vaccinia | C | ||
| Progressive vaccinia | C | ||
| Postvaccinia encephalitis | S | ||
| Blepharitis or conjunctivitis | S/C | Use Contact Precautions if copious drainage is present. | |
| Iritis or keratitis | S | ||
| Vaccinia-associated erythema multiforme (Stevens-Johnson syndrome) | S | Not an infectious condition. | |
| Secondary bacterial infection (eg, S. aureus, group A beta hemolytic streptococcus | S/C | Follow organism-specific (streptococcal and staphylococcal most frequent) recommendations and consider magnitude of drainage. | |
| Varicella zoster | A,C | Until lesions dry and crusted | Susceptible HCWs should not enter room if immune caregivers are available; no recommendation for face protection of immune HCWs; no recommendation for type of protection (ie, surgical mask or respirator) for susceptible HCWs. In an immunocompromised host with varicella pneumonia, prolong the duration of precautions for duration of illness. Postexposure prophylaxis: Provide postexposure vaccine as soon as possible but within 120 hours; for susceptible exposed persons for whom vaccine is contraindicated (immunocompromised persons, pregnant women, newborns whose mother's varicella onset is ≤ 5 days before delivery or within 48 hours after delivery) provide VZIG, when available, within 96 hours; if unavailable, use IVIG. Provide Airborne Precautions for exposed susceptible persons and exclude exposed susceptible health care workers beginning 8 days after first exposure until 21 days after last exposure or 28 if received VZIG, regardless of postexposure vaccination.1032 |
| Variola (see smallpox) | |||
| Vibrio parahaemolyticus (see gastroenteritis) | |||
| Vincent's angina (trench mouth) | S | ||
| Viral hemorrhagic fevers due to Lassa, Ebola, Marburg, Crimean-Congo fever viruses | S, D, C | DI | Single-patient room preferred. Emphasize: use of sharps safety devices and safe work practices, hand hygiene; barrier protection against blood and body fluids on entry into room (single gloves and fluid-resistant or impermeable gown, face/eye protection with masks, goggles or face shields), and appropriate waste handling. Use N95 or higher-level respirator when performing aerosol-generating procedures. Largest viral load in final stages of illness when hemorrhage may occur; additional PPE, including double gloves, leg and shoe coverings may be used, especially in resource-limited settings where options for cleaning and laundry are limited. Notify public health officials immediately if Ebola is suspected.212, 313, 738, 770 Also see Table 3 for Ebola as a bioterrorism agent. |
| Viral respiratory diseases (not covered elsewhere) | |||
| Adults | S | ||
| Infants and young children (see respiratory infectious disease, acute) | |||
| Whooping cough (see pertussis) | |||
| Wound infections | |||
| Major | C | DI | No dressing or dressing does not contain drainage adequately. |
| Minor or limited | S | Dressing covers and contains drainage adequately. | |
| Yersinia enterocolitica gastroenteritis (see gastroenteritis) | |||
| Zoster (varicella-zoster) (see herpes zoster) | |||
| Zygomycosis (phycomycosis, mucormycosis) | S | Not transmitted person to person. | |
Type of precautions: A, airborne precautions; C, contact; D, droplet; S, standard; when A, C, and D are specified, also use S.
Duration of precautions: CN, until off antimicrobial treatment and culture-negative; DI, duration of illness (with wound lesions, DI means until wounds stop draining); DE, until environment completely decontaminated; U, until time specified in hours (hrs) after initiation of effective therapy; Unknown: criteria for establishing eradication of pathogen has not been determined
References
- 1.Garner J.S. Guideline for isolation precautions in hospitals. The Hospital Infection Control Practices Advisory Committee. Infect Control Hosp Epidemiol. 1996;17:53–80. doi: 10.1086/647190. [DOI] [PubMed] [Google Scholar]
- 2.Harris A.D., Bradham D.D., Baumgarten M., Zuckerman I.H., Fink J.C., Perencevich E.N. The use and interpretation of quasi-experimental studies in infectious diseases. Clin Infect Dis. 2004;38:1586–1591. doi: 10.1086/420936. [DOI] [PubMed] [Google Scholar]
- 3.Morton V., Torgerson D.J. Effect of regression to the mean on decision making in health care. BMJ. 2003;326:1083–1084. doi: 10.1136/bmj.326.7398.1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pocock S.J., Elbourne D.R. Randomized trials or observational tribulations? N Engl J Med. 2000;342:1907–1909. doi: 10.1056/NEJM200006223422511. [DOI] [PubMed] [Google Scholar]
- 5.Ioannidis J.P., Haidich A.B., Pappa M. Comparison of evidence of treatment effects in randomized and nonrandomized studies. JAMA. 2001;286:821–830. doi: 10.1001/jama.286.7.821. [DOI] [PubMed] [Google Scholar]
- 6.Bent S., Shojania K.G., Saint S. The use of systematic reviews and meta-analyses in infection control and hospital epidemiology. Am J Infect Control. 2004;32:246–254. doi: 10.1016/j.ajic.2003.10.004. [DOI] [PubMed] [Google Scholar]
- 7.Harris A.D., Lautenbach E., Perencevich E. A systematic review of quasi-experimental study designs in the fields of infection control and antibiotic resistance. Clin Infect Dis. 2005;41:77–82. doi: 10.1086/430713. [DOI] [PubMed] [Google Scholar]
- 8.Evans R., Lloyd J.F., Abouzelof R.H., Taylor C.W., Anderson V.R., Samore M.H. System-wide surveillance for clinical encounters by patients previously identified with MRSA and VRE. Medinfo. 2004;2004:212–216. [PubMed] [Google Scholar]
- 9.Srinivasan A., McDonald L.C., Jernigan D. Foundations of the severe acute respiratory syndrome preparedness and response plan for healthcare facilities. Infect Control Hosp Epidemiol. 2004;25:1020–1025. doi: 10.1086/502338. [DOI] [PubMed] [Google Scholar]
- 10.www.cdc.gov/flu/avian/professional/infect-control.htm.
- 11.Centers for Disease Control and Prevention Guidelines for environmental infection control in health-care facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 2003;52:1–42. [PubMed] [Google Scholar]
- 12.Centers for Disease Control and Prevention Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. MMWR Recomm Rep. 2005;54:1–141. [PubMed] [Google Scholar]
- 13.American Institute of Architects . American Institute of Architects Press; Washington, DC: 2006. Guidelines for design and construction of hospital and health care facilities. [Google Scholar]
- 14.Centers for Disease Control and Prevention Guidelines for preventing health care–associated pneumonia, 2003. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee. MMWR Recomm Rep. 2004;53:1–40. [PubMed] [Google Scholar]
- 15.Centers for Disease Control and Prevention Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. Recommendations of CDC, the Infectious Disease Society of America, and the American Society of Blood and Marrow Transplantation. MMWR Recomm Rep. 2000;49:1–125. [PubMed] [Google Scholar]
- 16.Boyce J.M., Pittet D. Guideline for hand hygiene in health-care settings. Recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force, Society for Healthcare Epidemiology of America/Association for Professionals in Infection Control/Infectious Diseases Society of America. MMWR Recomm Rep. 2002;51:1–45. [PubMed] [Google Scholar]
- 17.Bolyard E.A., Tablan O.C., Williams W.W., Pearson M.L., Shapiro C.N., Deitchmann S.D. Guideline for infection control in healthcare personnel, 1998. Hospital Infection Control Practices Advisory Committee. Infect Control Hosp Epidemiol. 1998;19:407–463. doi: 10.1086/647840. [DOI] [PubMed] [Google Scholar]
- 18.Centers for Disease Control and Prevention Recommendations for preventing transmission of infections among chronic hemodialysis patients. MMWR Recomm Rep. 2001;50:1–43. [PubMed] [Google Scholar]
- 19.Kohn W.G., Collins A.S., Cleveland J.L., Harte J.A., Eklund K.J., Malvitz D.M. Guidelines for infection control in dental health-care settings, 2003. MMWR Recomm Rep. 2003;52:1–61. [PubMed] [Google Scholar]
- 20.Saiman L., Siegel J. Infection control recommendations for patients with cystic fibrosis: microbiology, important pathogens, and infection control practices to prevent patient-to-patient transmission. Infect Control Hosp Epidemiol. 2003;24(5 Suppl):S6–S52. doi: 10.1086/503485. [DOI] [PubMed] [Google Scholar]
- 21.Varia M., Wilson S., Sarwal S. Investigation of a nosocomial outbreak of severe acute respiratory syndrome (SARS) in Toronto, Canada. CMAJ. 2003;169:285–292. [PMC free article] [PubMed] [Google Scholar]
- 22.Haley R.W., Cushion N.B., Tenover F.C. Eradication of endemic methicillin-resistant Staphylococcus aureus infections from a neonatal intensive care unit. J Infect Dis. 1995;171:614–624. doi: 10.1093/infdis/171.3.614. [DOI] [PubMed] [Google Scholar]
- 23.Bridges C.B., Kuehnert M.J., Hall C.B. Transmission of influenza: implications for control in health care settings. Clin Infect Dis. 2003;37:1094–1101. doi: 10.1086/378292. [DOI] [PubMed] [Google Scholar]
- 24.Hall C.B. Nosocomial respiratory syncytial virus infections: the “cold war” has not ended. Clin Infect Dis. 2000;31:590–596. doi: 10.1086/313960. [DOI] [PubMed] [Google Scholar]
- 25.Campbell J.R., Zaccaria E., Mason E.O., Jr., Baker C.J. Epidemiological analysis defining concurrent outbreaks of Serratia marcescens and methicillin-resistant Staphylococcus aureus in a neonatal intensive-care unit. Infect Control Hosp Epidemiol. 1998;19:924–928. [PubMed] [Google Scholar]
- 26.Pena C., Pujol M., Ardanuy C. Epidemiology and successful control of a large outbreak due to Klebsiella pneumoniae producing extended-spectrum beta-lactamases. Antimicrob Agents Chemother. 1998;42:53–58. doi: 10.1128/aac.42.1.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bonten M.J., Slaughter S., Ambergen A.W. The role of “colonization pressure” in the spread of vancomycin-resistant enterococci: an important infection control variable. Arch Intern Med. 1998;158:1127–1132. doi: 10.1001/archinte.158.10.1127. [DOI] [PubMed] [Google Scholar]
- 28.Jensenius M., Ringertz S.H., Berild D., Bell H., Espinoza R., Grinde B. Prolonged nosocomial outbreak of hepatitis A arising from an alcoholic with pneumonia. Scand J Infect Dis. 1998;30:119–123. doi: 10.1080/003655498750003474. [DOI] [PubMed] [Google Scholar]
- 29.Zawacki A., O'Rourke E., Potter-Bynoe G., Macone A., Harbarth S., Goldmann D. An outbreak of Pseudomonas aeruginosa pneumonia and bloodstream infection associated with intermittent otitis externa in a healthcare worker. Infect Control Hosp Epidemiol. 2004;25:1083–1089. doi: 10.1086/502348. [DOI] [PubMed] [Google Scholar]
- 30.Foca M., Jakob K., Whittier S. Endemic Pseudomonas aeruginosa infection in a neonatal intensive care unit. N Engl J Med. 2000;343:695–700. doi: 10.1056/NEJM200009073431004. [DOI] [PubMed] [Google Scholar]
- 31.Gupta A., Della-Latta P., Todd B. Outbreak of extended-spectrum beta-lactamase–producing Klebsiella pneumoniae in a neonatal intensive care unit linked to artificial nails. Infect Control Hosp Epidemiol. 2004;25:210–215. doi: 10.1086/502380. [DOI] [PubMed] [Google Scholar]
- 32.Boyce J.M., Opal S.M., Potter-Bynoe G., Medeiros A.A. Spread of methicillin-resistant Staphylococcus aureus in a hospital after exposure to a health care worker with chronic sinusitis. Clin Infect Dis. 1993;17:496–504. doi: 10.1093/clinids/17.3.496. [DOI] [PubMed] [Google Scholar]
- 33.Fliegel P.E., Weinstein W.M. Rubella outbreak in a prenatal clinic: management and prevention. Am J Infect Control. 1982;10:29–33. doi: 10.1016/0196-6553(82)90037-2. [DOI] [PubMed] [Google Scholar]
- 34.Atkinson W.L., Markowitz L.E., Adams N.C., Seastrom G.R. Transmission of measles in medical settings, United States, 1985–1989. Am J Med. 1991;91:320S–324S. doi: 10.1016/0002-9343(91)90389-f. [DOI] [PubMed] [Google Scholar]
- 35.Carman W.F., Elder A.G., Wallace L.A. Effects of influenza vaccination of health-care workers on mortality of elderly people in long-term care: a randomised controlled trial. Lancet. 2000;355:93–97. doi: 10.1016/S0140-6736(99)05190-9. [DOI] [PubMed] [Google Scholar]
- 36.Centers for Disease Control and Prevention Outbreaks of pertussis associated with hospitals: Kentucky, Pennsylvania, and Oregon, 2003. MMWR Morb Mortal Wkly Rep. 2005;54:67–71. [PubMed] [Google Scholar]
- 37.Mermel L.A., McKay M., Dempsey J., Parenteau S. Pseudomonas surgical-site infections linked to a healthcare worker with onychomycosis. Infect Control Hosp Epidemiol. 2003;24:749–752. doi: 10.1086/502125. [DOI] [PubMed] [Google Scholar]
- 38.Barnes G.L., Callaghan S.L., Kirkwood C.D., Bogdanovic-Sakran N., Johnston L.J., Bishop R.F. Excretion of serotype G1 rotavirus strains by asymptomatic staff: a possible source of nosocomial infection. J Pediatr. 2003;142:722–725. doi: 10.1067/mpd.2003.240. [DOI] [PubMed] [Google Scholar]
- 39.Wang J.T., Chang S.C., Ko W.J. A hospital-acquired outbreak of methicillin-resistant Staphylococcus aureus infection initiated by a surgeon carrier. J Hosp Infect. 2001;47:104–109. doi: 10.1053/jhin.2000.0878. [DOI] [PubMed] [Google Scholar]
- 40.Valenti W.M., Pincus P.H., Messner M.K. Nosocomial pertussis: possible spread by a hospital visitor. Am J Dis Child. 1980;134:520–521. doi: 10.1001/archpedi.1980.02130170070023. [DOI] [PubMed] [Google Scholar]
- 41.Christie C.D., Glover A.M., Willke M.J., Marx M.L., Reising S.F., Hutchinson N.M. Containment of pertussis in the regional pediatric hospital during the greater Cincinnati epidemic of 1993. Infect Control Hosp Epidemiol. 1995;16:556–563. doi: 10.1086/647008. [DOI] [PubMed] [Google Scholar]
- 42.Munoz F.M., Ong L.T., Seavy D., Medina D., Correa A., Starke J.R. Tuberculosis among adult visitors of children with suspected tuberculosis and employees at a children's hospital. Infect Control Hosp Epidemiol. 2002;23:568–572. doi: 10.1086/501972. [DOI] [PubMed] [Google Scholar]
- 43.Garcia R., Raad I., Abi-Said D. Nosocomial respiratory syncytial virus infections: prevention and control in bone marrow transplant patients. Infect Control Hosp Epidemiol. 1997;18:412–416. doi: 10.1086/647640. [DOI] [PubMed] [Google Scholar]
- 44.Whimbey E., Champlin R.E., Couch R.B. Community respiratory virus infections among hospitalized adult bone marrow transplant recipients. Clin Infect Dis. 1996;22:778–782. doi: 10.1093/clinids/22.5.778. [DOI] [PubMed] [Google Scholar]
- 45.Saiman L., O'Keefe M., Graham P.L. Hospital transmission of community-acquired methicillin-resistant Staphylococcus aureus among postpartum women. Clin Infect Dis. 2003;37:1313–1319. doi: 10.1086/379022. [DOI] [PubMed] [Google Scholar]
- 46.Bonten M.J., Slaughter S., Hayden M.K., Nathan C., van Voorhis J., Weinstein R.A. External sources of vancomycin-resistant enterococci for intensive care units. Crit Care Med. 1998;26:2001–2004. doi: 10.1097/00003246-199812000-00029. [DOI] [PubMed] [Google Scholar]
- 47.Flynn D.M., Weinstein R.A., Nathan C., Gaston M.A., Kabins S.A. Patients' endogenous flora as the source of “nosocomial” Enterobacter in cardiac surgery. J Infect Dis. 1987;156:363–368. doi: 10.1093/infdis/156.2.363. [DOI] [PubMed] [Google Scholar]
- 48.Olson B., Weinstein R.A., Nathan C., Chamberlin W., Kabins S.A. Epidemiology of endemic Pseudomonas aeruginosa: why infection control efforts have failed. J Infect Dis. 1984;150:808–816. doi: 10.1093/infdis/150.6.808. [DOI] [PubMed] [Google Scholar]
- 49.Perl T.M., Cullen J.J., Wenzel R.P. Intranasal mupirocin to prevent postoperative Staphylococcus aureus infections. N Engl J Med. 2002;346:1871–1877. doi: 10.1056/NEJMoa003069. [DOI] [PubMed] [Google Scholar]
- 50.Toltzis P., Hoyen C. Factors that predict preexisting colonization with antibiotic-resistant Gram-negative bacilli in patients admitted to a pediatric intensive care unit. Pediatrics. 1999;103(4 Pt 1):719–723. doi: 10.1542/peds.103.4.719. [DOI] [PubMed] [Google Scholar]
- 51.Sarginson R.E., Taylor N., Reilly N., Baines P.B., Van Saene H.K. Infection in prolonged pediatric critical illness: a prospective four-year study based on knowledge of the carrier state. Crit Care Med. 2004;32:839–847. doi: 10.1097/01.ccm.0000117319.17600.e8. [DOI] [PubMed] [Google Scholar]
- 52.Silvestri L., Monti Bragadin C., Milanese M. Are most ICU infections really nosocomial? A prospective observational cohort study in mechanically ventilated patients. J Hosp Infect. 1999;42:125–133. doi: 10.1053/jhin.1998.0550. [DOI] [PubMed] [Google Scholar]
- 53.Heggers J.P., Phillips L.G., Boertman J.A. The epidemiology of methicillin-resistant Staphylococcus aureus in a burn center. J Burn Care Rehabil. 1988;9:610–612. doi: 10.1097/00004630-198811000-00007. [DOI] [PubMed] [Google Scholar]
- 54.Donskey C.J. The role of the intestinal tract as a reservoir and source for transmission of nosocomial pathogens. Clin Infect Dis. 2004;39:219–226. doi: 10.1086/422002. [DOI] [PubMed] [Google Scholar]
- 55.Osterholm MT, Hedberg CW, Moore KA. The epidemiology of infectious diseases. In: G.L. M, Jr DRG, J.E. B, editors. Principles and practice of infectious diseases. 5th ed. Philadelphia: Churchill Livingstone; 2000. p 161–3.
- 56.Thomsen R.W., Hundborg H.H., Lervang H.H., Johnsen S.P., Schonheyder H.C., Sorensen H.T. Risk of community-acquired pneumococcal bacteremia in patients with diabetes: a population-based case-control study. Diabetes Care. 2004;27:1143–1147. doi: 10.2337/diacare.27.5.1143. [DOI] [PubMed] [Google Scholar]
- 57.Carton J.A., Maradona J.A., Nuno F.J., Fernandez-Alvarez R., Perez-Gonzalez F., Asensi V. Diabetes mellitus and bacteraemia: a comparative study between diabetic and non-diabetic patients. Eur J Med. 1992;1:281–287. [PubMed] [Google Scholar]
- 58.Hirschtick R.E., Glassroth J., Jordan M.C. Bacterial pneumonia in persons infected with the human immunodeficiency virus. Pulmonary Complications of HIV Infection Study Group. N Engl J Med. 1995;333:845–851. doi: 10.1056/NEJM199509283331305. [DOI] [PubMed] [Google Scholar]
- 59.Rosenberg A.L., Seneff M.G., Atiyeh L., Wagner R., Bojanowski L., Zimmerman J.E. The importance of bacterial sepsis in intensive care unit patients with acquired immunodeficiency syndrome: implications for future care in the age of increasing antiretroviral resistance. Crit Care Med. 2001;29:548–556. doi: 10.1097/00003246-200103000-00013. [DOI] [PubMed] [Google Scholar]
- 60.Malone J.L., Ijaz K., Lambert L. Investigation of healthcare-associated transmission of Mycobacterium tuberculosis among patients with malignancies at three hospitals and at a residential facility. Cancer. 2004;101:2713–2721. doi: 10.1002/cncr.20698. [DOI] [PubMed] [Google Scholar]
- 61.Fishman J.A., Rubin R.H. Infection in organ transplant recipients. N Engl J Med. 1998;338:1741–1751. doi: 10.1056/NEJM199806113382407. [DOI] [PubMed] [Google Scholar]
- 62.Safdar N., Kluger D.M., Maki D.G. A review of risk factors for catheter-related bloodstream infection caused by percutaneously inserted, noncuffed central venous catheters: implications for preventive strategies. Medicine (Baltimore) 2002;81:466–479. doi: 10.1097/00005792-200211000-00007. [DOI] [PubMed] [Google Scholar]
- 63.Jarvis W.R., Robles B. Nosocomial infections in pediatric patients. Adv Pediatr Infect Dis. 1996;12:243–259. [PubMed] [Google Scholar]
- 64.Yogaraj J.S., Elward A.M., Fraser V.J. Rate, risk factors, and outcomes of nosocomial primary bloodstream infection in pediatric intensive care unit patients. Pediatrics. 2002;110:481–485. doi: 10.1542/peds.110.3.481. [DOI] [PubMed] [Google Scholar]
- 65.Donlan R.M. Biofilms: microbial life on surfaces. Emerg Infect Dis. 2002;8:881–890. doi: 10.3201/eid0809.020063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rosen H.R. Acquisition of hepatitis C by a conjunctival splash. Am J Infect Control. 1997;25:242–247. doi: 10.1016/s0196-6553(97)90011-0. [DOI] [PubMed] [Google Scholar]
- 67.Beltrami E.M., Kozak A., Williams I.T. Transmission of HIV and hepatitis C virus from a nursing home patient to a health care worker. Am J Infect Control. 2003;31:168–175. doi: 10.1067/mic.2003.27. [DOI] [PubMed] [Google Scholar]
- 68.Obasanjo O.O., Wu P., Conlon M. An outbreak of scabies in a teaching hospital: lessons learned. Infect Control Hosp Epidemiol. 2001;22:13–18. doi: 10.1086/501818. [DOI] [PubMed] [Google Scholar]
- 69.Andersen B.M., Haugen H., Rasch M., Heldal Haugen A., Tageson A. Outbreak of scabies in Norwegian nursing homes and home care patients: control and prevention. J Hosp Infect. 2000;45:160–164. doi: 10.1053/jhin.1999.0716. [DOI] [PubMed] [Google Scholar]
- 70.Avitzur Y., Amir J. Herpetic whitlow infection in a general pediatrician: an occupational hazard. Infection. 2002;30:234–236. doi: 10.1007/s15010-002-2155-5. [DOI] [PubMed] [Google Scholar]
- 71.Adams G., Stover B.H., Keenlyside R.A. Nosocomial herpetic infections in a pediatric intensive care unit. Am J Epidemiol. 1981;113:126–132. doi: 10.1093/oxfordjournals.aje.a113076. [DOI] [PubMed] [Google Scholar]
- 72.Bhalla A., Pultz N.J., Gries D.M. Acquisition of nosocomial pathogens on hands after contact with environmental surfaces near hospitalized patients. Infect Control Hosp Epidemiol. 2004;25:164–167. doi: 10.1086/502369. [DOI] [PubMed] [Google Scholar]
- 73.Duckro A.N., Blom D.W., Lyle E.A., Weinstein R.A., Hayden M.K. Transfer of vancomycin-resistant enterococci via health care worker hands. Arch Intern Med. 2005;165:302–307. doi: 10.1001/archinte.165.3.302. [DOI] [PubMed] [Google Scholar]
- 74.Brooks S.E., Veal R.O., Kramer M., Dore L., Schupf N., Adachi M. Reduction in the incidence of Clostridium difficile–associated diarrhea in an acute care hospital and a skilled nursing facility following replacement of electronic thermometers with single-use disposables. Infect Control Hosp Epidemiol. 1992;13:98–103. doi: 10.1086/646480. [DOI] [PubMed] [Google Scholar]
- 75.Centers for Disease Control and Prevention Nosocomial hepatitis B virus infection associated with reusable fingerstick blood sampling devices, Ohio and New York City, 1996. MMWR Morb Mortal Wkly Rep. 1997;46:217–221. [PubMed] [Google Scholar]
- 76.Desenclos J.C., Bourdiol-Razes M., Rolin B. Hepatitis C in a ward for cystic fibrosis and diabetic patients: possible transmission by spring-loaded finger-stick devices for self-monitoring of capillary blood glucose. Infect Control Hosp Epidemiol. 2001;22:701–707. doi: 10.1086/501849. [DOI] [PubMed] [Google Scholar]
- 77.Centers for Disease Control and Prevention Transmission of hepatitis B virus among persons undergoing blood glucose monitoring in long-term care facilities, Mississippi, North Carolina, and Los Angeles County, California, 2003–2004. MMWR Morb Mortal Wkly Rep. 2005;54:220–223. [PubMed] [Google Scholar]
- 78.Hall C.B., Douglas R.G., Jr. Modes of transmission of respiratory syncytial virus. J Pediatr. 1981;99:100–103. doi: 10.1016/s0022-3476(81)80969-9. [DOI] [PubMed] [Google Scholar]
- 79.Hall C.B., Douglas R.G., Jr., Geiman J.M. Possible transmission by fomites of respiratory syncytial virus. J Infect Dis. 1980;141:98–102. doi: 10.1093/infdis/141.1.98. [DOI] [PubMed] [Google Scholar]
- 80.Buttery J.P., Alabaster S.J., Heine R.G. Multiresistant Pseudomonas aeruginosa outbreak in a pediatric oncology ward related to bath toys. Pediatr Infect Dis J. 1998;17:509–513. doi: 10.1097/00006454-199806000-00015. [DOI] [PubMed] [Google Scholar]
- 81.Agerton T., Valway S., Gore B. Transmission of a highly drug-resistant strain (strain W1) of Mycobacterium tuberculosis: community outbreak and nosocomial transmission via a contaminated bronchoscope. JAMA. 1997;278:1073–1077. [PubMed] [Google Scholar]
- 82.Bronowicki J.P., Venard V., Botte C. Patient-to-patient transmission of hepatitis C virus during colonoscopy. N Engl J Med. 1997;337:237–240. doi: 10.1056/NEJM199707243370404. [DOI] [PubMed] [Google Scholar]
- 83.Michele T.M., Cronin W.A., Graham N.M. Transmission of Mycobacterium tuberculosis by a fiberoptic bronchoscope: identification by DNA fingerprinting. JAMA. 1997;278:1093–1095. [PubMed] [Google Scholar]
- 84.Schelenz S., French G. An outbreak of multidrug-resistant Pseudomonas aeruginosa infection associated with contamination of bronchoscopes and an endoscope washer-disinfector. J Hosp Infect. 2000;46:23–30. doi: 10.1053/jhin.2000.0800. [DOI] [PubMed] [Google Scholar]
- 85.Weber D.J., Rutala W.A. Lessons from outbreaks associated with bronchoscopy. Infect Control Hosp Epidemiol. 2001;22:403–408. doi: 10.1086/501924. [DOI] [PubMed] [Google Scholar]
- 86.Kirschke D.L., Jones T.F., Craig A.S. Pseudomonas aeruginosa and Serratia marcescens contamination associated with a manufacturing defect in bronchoscopes. N Engl J Med. 2003;348:214–220. doi: 10.1056/NEJMoa021791. [DOI] [PubMed] [Google Scholar]
- 87.Srinivasan A., Wolfenden L.L., Song X. An outbreak of Pseudomonas aeruginosa infections associated with flexible bronchoscopes. N Engl J Med. 2003;348:221–227. doi: 10.1056/NEJMoa021808. [DOI] [PubMed] [Google Scholar]
- 88.Boyce J.M., Potter-Bynoe G., Chenevert C., King T. Environmental contamination due to methicillin-resistant Staphylococcus aureus: possible infection control implications. Infect Control Hosp Epidemiol. 1997;18:622–627. [PubMed] [Google Scholar]
- 89.Zachary K.C., Bayne P.S., Morrison V.J., Ford D.S., Silver L.C., Hooper D.C. Contamination of gowns, gloves, and stethoscopes with vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 2001;22:560–564. doi: 10.1086/501952. [DOI] [PubMed] [Google Scholar]
- 90.Perry C., Marshall R., Jones E. Bacterial contamination of uniforms. J Hosp Infect. 2001;48:238–241. doi: 10.1053/jhin.2001.0962. [DOI] [PubMed] [Google Scholar]
- 91.Papineni R.S., Rosenthal F.S. The size distribution of droplets in the exhaled breath of healthy human subjects. J Aerosol Med. 1997;10:105–116. doi: 10.1089/jam.1997.10.105. [DOI] [PubMed] [Google Scholar]
- 92.Wells W.F. On airborne infection. Study II: droplets and droplet nuclei. Am J Hygiene. 1934;20:611–618. [Google Scholar]
- 93.Loeb M., McGeer A., Henry B. SARS among critical care nurses, Toronto. Emerg Infect Dis. 2004;10:251–255. doi: 10.3201/eid1002.030838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Fowler R.A., Guest C.B., Lapinsky S.E. Transmission of severe acute respiratory syndrome during intubation and mechanical ventilation. Am J Respir Crit Care Med. 2004;169:1198–1202. doi: 10.1164/rccm.200305-715OC. [DOI] [PubMed] [Google Scholar]
- 95.Gehanno J.F., Kohen-Couderc L., Lemeland J.F., Leroy J. Nosocomial meningococcemia in a physician. Infect Control Hosp Epidemiol. 1999;20:564–565. doi: 10.1086/501672. [DOI] [PubMed] [Google Scholar]
- 96.Scales D. Illness in intensive-care staff after brief exposure to severe acute respiratory syndrome. Emerg Infect Dis. 2003;9:1205–1210. doi: 10.3201/eid0910.030525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ensor E., Humphreys H., Peckham D., Webster C., Knox A.J. Is Burkholderia (Pseudomonas) cepacia disseminated from cystic fibrosis patients during physiotherapy? J Hosp Infect. 1996;32:9–15. doi: 10.1016/s0195-6701(96)90160-7. [DOI] [PubMed] [Google Scholar]
- 98.Christian M.D., Loutfy M., McDonald L.C. Possible SARS coronavirus transmission during cardiopulmonary resuscitation. Emerg Infect Dis. 2004;10:287–293. doi: 10.3201/eid1002.030700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Valenzuela T.D., Hooton T.M., Kaplan E.L., Schlievert P. Transmission of “toxic strep” syndrome from an infected child to a firefighter during CPR. Ann Emerg Med. 1991;20:90–92. doi: 10.1016/s0196-0644(05)81129-1. [DOI] [PubMed] [Google Scholar]
- 100.Bassinet L., Matrat M., Njamkepo E., Aberrane S., Housset B., Guiso N. Nosocomial pertussis outbreak among adult patients and healthcare workers. Infect Control Hosp Epidemiol. 2004;25:995–997. doi: 10.1086/502332. [DOI] [PubMed] [Google Scholar]
- 101.Wong T.W., Lee C.K., Tam W. Cluster of SARS among medical students exposed to single patient, Hong Kong. Emerg Infect Dis. 2004;10:269–276. doi: 10.3201/eid1002.030452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Pachucki C.T., Pappas S.A., Fuller G.F., Krause S.L., Lentino J.R., Schaaff D.M. Influenza A among hospital personnel and patients: implications for recognition, prevention, and control. Arch Intern Med. 1989;149:77–80. [PubMed] [Google Scholar]
- 103.Feigin R.D., Baker C.J., Herwaldt L.A., Lampe R.M., Mason E.O., Whitney S.E. Epidemic meningococcal disease in an elementary school classroom. N Engl J Med. 1982;307:1255–1257. doi: 10.1056/NEJM198211113072007. [DOI] [PubMed] [Google Scholar]
- 104.Dick E.C., Jennings L.C., Mink K.A., Wartgow C.D., Inhorn S.L. Aerosol transmission of rhinovirus colds. J Infect Dis. 1987;156:442–448. doi: 10.1093/infdis/156.3.442. [DOI] [PubMed] [Google Scholar]
- 105.Duguid J.P. The size and duration of air-carriage of respiratory droplets and droplet nucleii. J Hyg (Lond) 1946;44:471–479. doi: 10.1017/s0022172400019288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hall C.B., Douglas R.G., Jr., Schnabel K.C., Geiman J.M. Infectivity of respiratory syncytial virus by various routes of inoculation. Infect Immun. 1981;33:779–783. doi: 10.1128/iai.33.3.779-783.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Downie A.W., Meiklejohn M., St Vincent L., Rao A.R., Sundara Babu B.V., Kempe C.H. The recovery of smallpox virus from patients and their environment in a smallpox hospital. Bull World Health Organ. 1965;33:615–622. [PMC free article] [PubMed] [Google Scholar]
- 108.Fenner F., Henderson D.A., Arita I., Jezek Z., Ladnyi I.D. Smallpox and its eradication. World Health Organization; Geneva, Switzerland:: 1988. The epidemiology of smallpox. [Google Scholar]
- 109.Cole E.C., Cook C.E. Characterization of infectious aerosols in health care facilities: an aid to effective engineering controls and preventive strategies. Am J Infect Control. 1998;26:453–464. doi: 10.1016/S0196-6553(98)70046-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Christie C., Mazon D., Hierholzer W., Jr., Patterson J.E. Molecular heterogeneity of Acinetobacter baumanii isolates during seasonal increase in prevalence. Infect Control Hosp Epidemiol. 1995;16:590–594. doi: 10.1086/647013. [DOI] [PubMed] [Google Scholar]
- 111.Musher D.M. How contagious are common respiratory tract infections? N Engl J Med. 2003;348:1256–1266. doi: 10.1056/NEJMra021771. [DOI] [PubMed] [Google Scholar]
- 112.Steinberg P., White R.J., Fuld S.L., Gutekunst R.R., Chanock R.M., Senterfit L.B. Ecology of Mycoplasma pneumoniae infections in marine recruits at Parris Island, South Carolina. Am J Epidemiol. 1969;89:62–73. doi: 10.1093/oxfordjournals.aje.a120916. [DOI] [PubMed] [Google Scholar]
- 113.Seto W.H., Tsang D., Yung R.W. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS) Lancet. 2003;361:1519–1520. doi: 10.1016/S0140-6736(03)13168-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hamburger M., Robertson O.H. Expulsion of group A haemolytic streptocicci in droplets and droplet nuclei by sneezing, coughing and talking. Am J Med. 1948;4:690. doi: 10.1016/s0002-9343(48)90392-1. [DOI] [PubMed] [Google Scholar]
- 115.Centers for Disease Control and Prevention Nosocomial meningococcemia. MMWR Morb Mortal Wkly Rep. 1978;27:358. [Google Scholar]
- 116.LeClair J.M., Freeman J., Sullivan B.F., Crowley C.M., Goldmann D.A. Prevention of nosocomial respiratory syncytial virus infections through compliance with glove and gown isolation precautions. N Engl J Med. 1987;317:329–334. doi: 10.1056/NEJM198708063170601. [DOI] [PubMed] [Google Scholar]
- 117.Madge P., Paton J.Y., McColl J.H., Mackie P.L. Prospective controlled study of four infection-control procedures to prevent nosocomial infection with respiratory syncytial virus. Lancet. 1992;340:1079–1083. doi: 10.1016/0140-6736(92)93088-5. [DOI] [PubMed] [Google Scholar]
- 118.Bassetti S., Bischoff W.E., Walter M. Dispersal of Staphylococcus aureus into the air associated with a rhinovirus infection. Infect Control Hosp Epidemiol. 2005;26:196–203. doi: 10.1086/502526. [DOI] [PubMed] [Google Scholar]
- 119.Eichenwald H.F., Kotsevalov O., Fasso L.A. The “cloud baby”: an example of bacterial–viral interaction. Am J Dis Child. 1960;100:161–173. doi: 10.1001/archpedi.1960.04020040163003. [DOI] [PubMed] [Google Scholar]
- 120.Sheretz R.J., Reagan D.R., Hampton K.D. A cloud adult: the Staphylococcus aureus–virus interaction revisited. Ann Intern Med. 1996;124:539–547. doi: 10.7326/0003-4819-124-6-199603150-00001. [DOI] [PubMed] [Google Scholar]
- 121.Coronado V.G., Beck-Sague C.M., Hutton M.D. Transmission of multidrug-resistant Mycobacterium tuberculosis among persons with human immunodeficiency virus infection in an urban hospital: epidemiologic and restriction fragment length polymorphism analysis. J Infect Dis. 1993;168:1052–1055. doi: 10.1093/infdis/168.4.1052. [DOI] [PubMed] [Google Scholar]
- 122.Bloch A.B., Orenstein W.A., Ewing W.M. Measles outbreak in a pediatric practice: airborne transmission in an office setting. Pediatrics. 1985;75:676–683. [PubMed] [Google Scholar]
- 123.LeClair J.M., Zaia J.A., Levin M.J., Congdon R.G., Goldmann D.A. Airborne transmission of chickenpox in a hospital. N Engl J Med. 1980;302:450–453. doi: 10.1056/NEJM198002213020807. [DOI] [PubMed] [Google Scholar]
- 124.Riley R.L., Mills C.C., Nyka W. Aerial dissemination of pulmonary tuberculosis: a two-year study of contagion in a tuberculosis ward. Am J Hygiene. 1959;70:185–196. doi: 10.1093/oxfordjournals.aje.a117542. [DOI] [PubMed] [Google Scholar]
- 125.Beck-Sague C., Dooley S.W., Hutton M.D. Hospital outbreak of multidrug-resistant Mycobacterium tuberculosis infections: factors in transmission to staff and HIV-infected patients. JAMA. 1992;268:1280–1286. doi: 10.1001/jama.1992.03490100078031. [DOI] [PubMed] [Google Scholar]
- 126.Centers for Disease Control and Prevention Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care facilities, 1994. MMWR Recomm Rep. 1994;43:1–132. [PubMed] [Google Scholar]
- 127.Haley C.E., McDonald R.C., Rossi L., Jones W.D., Jr., Haley R.W., Luby J.P. Tuberculosis epidemic among hospital personnel. Infect Control Hosp Epidemiol. 1989;10:204–210. doi: 10.1086/646003. [DOI] [PubMed] [Google Scholar]
- 128.Wehrle P.F., Posch J., Richter K.H., Henderson D.A. An airborne outbreak of smallpox in a German hospital and its significance with respect to other recent outbreaks in Europe. Bull World Health Organ. 1970;43:669–679. [PMC free article] [PubMed] [Google Scholar]
- 129.Gelfand H.M., Posch J. The recent outbreak of smallpox in Meschede, West Germany. Am J Epidemiol. 1971;93:234–237. doi: 10.1093/oxfordjournals.aje.a121251. [DOI] [PubMed] [Google Scholar]
- 130.Moser M.R., Bender T.R., Margolis H.S., Noble G.R., Kendal A.P., Ritter D.G. An outbreak of influenza aboard a commercial airliner. Am J Epidemiol. 1979;110:1–6. doi: 10.1093/oxfordjournals.aje.a112781. [DOI] [PubMed] [Google Scholar]
- 131.Alford R.H., Kasel J.A., Gerone P.J., Knight V. Human influenza resulting from aerosol inhalation. Proc Soc Exp Biol Med. 1966;122:800–804. doi: 10.3181/00379727-122-31255. [DOI] [PubMed] [Google Scholar]
- 132.Chadwick P.R., McCann R. Transmission of a small round structured virus by vomiting during a hospital outbreak of gastroenteritis. J Hosp Infect. 1994;26:251–259. doi: 10.1016/0195-6701(94)90015-9. [DOI] [PubMed] [Google Scholar]
- 133.Prince D.S., Astry C., Vonderfecht S., Jakab G., Shen F.M., Yolken R.H. Aerosol transmission of experimental rotavirus infection. Pediatr Infect Dis. 1986;5:218–222. doi: 10.1097/00006454-198603000-00012. [DOI] [PubMed] [Google Scholar]
- 134.Centers for Disease Control and Prevention. Available from www.cdc.gov/ncidod/sars. Accessed.
- 135.Peiris J.S., Yuen K.Y., Osterhaus A.D., Stohr K. The severe acute respiratory syndrome. N Engl J Med. 2003;349:2431–2441. doi: 10.1056/NEJMra032498. [DOI] [PubMed] [Google Scholar]
- 136.Olsen S.J., Chang H.L., Cheung T.Y. Transmission of the severe acute respiratory syndrome on aircraft. N Engl J Med. 2003;349:2416–2422. doi: 10.1056/NEJMoa031349. [DOI] [PubMed] [Google Scholar]
- 137.Wilder-Smith A., Leong H.N., Villacian J.S. In-flight transmission of severe acute respiratory syndrome (SARS): a case report. J Travel Med. 2003;10:299–300. doi: 10.2310/7060.2003.2737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Booth T.F., Kournikakis B., Bastien N. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J Infect Dis. 2005;191:1472–1477. doi: 10.1086/429634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Yu I.T., Li Y., Wong T.W. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med. 2004;350:1731–1739. doi: 10.1056/NEJMoa032867. [DOI] [PubMed] [Google Scholar]
- 140.Centers for Disease Control and Prevention Update: outbreak of severe acute respiratory syndrome worldwide. MMWR Morb Mortal Wkly Rep. 2003;52:241–248. [PubMed] [Google Scholar]
- 141.Centers for Disease Control and Prevention Cluster of severe acute respiratory syndrome cases among protected health-care workers, Toronto, Canada, April 2003. MMWR Morb Mortal Wkly Rep. 2003;52:433–436. [PubMed] [Google Scholar]
- 142.Sawyer L.A., Murphy J.J., Kaplan J.E. 25- to 30-nm virus particles associated with a hospital outbreak of acute gastroenteritis with evidence for airborne transmission. Am J Epidimiol. 1988;127:1261–1271. doi: 10.1093/oxfordjournals.aje.a114918. [DOI] [PubMed] [Google Scholar]
- 143.Marks P.J., Vipond I.B., Carlisle D., Deakin D., Fey R.E., Caul E.O. Evidence for airborne transmission of Norwalk-like virus (NLV) in a hotel restaurant. Epidemiol Infect. 2000;124:481–487. doi: 10.1017/s0950268899003805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Salgado C.D., Farr B.M., Hall K.K., Hayden F.G. Influenza in the acute hospital setting. Lancet Infect Dis. 2002;2:145–155. doi: 10.1016/s1473-3099(02)00221-9. [DOI] [PubMed] [Google Scholar]
- 145.Riley R.L. Airborne infection. Am J Med. 1974;57:466–475. doi: 10.1016/0002-9343(74)90140-5. [DOI] [PubMed] [Google Scholar]
- 146.McLean R. General discussion. Am Rev Respir Dis. 1961;83:36–38. [Google Scholar]
- 147.Cheesbrough J.S., Green J., Gallimore C.I., Wright P.A., Brown D.W. Widespread environmental contamination with Norwalk-like viruses (NLV) detected in a prolonged hotel outbreak of gastroenteritis. Epidemiol Infect. 2000;125:93–98. doi: 10.1017/s095026889900432x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Marks P.J., Vipond I.B., Regan F.M., Wedgwood K., Fey R.E., Caul E.O. A school outbreak of Norwalk-like virus: evidence for airborne transmission. Epidemiol Infect. 2003;131:727–736. doi: 10.1017/s0950268803008689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Roy C.J., Milton D.K. Airborne transmission of communicable infection: the elusive pathway. N Engl J Med. 2004;350:1710–1712. doi: 10.1056/NEJMp048051. [DOI] [PubMed] [Google Scholar]
- 150.Dull P.M., Wilson K.E., Kournikakis B. Bacillus anthracis aerosolization associated with a contaminated mail-sorting machine. Emerg Infect Dis. 2002;8:1044–1047. doi: 10.3201/eid0810.020356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Weis C.P., Intrepido A.J., Miller A.K. Secondary aerosolization of viable Bacillus anthracis spores in a contaminated US Senate Office. JAMA. 2002;288:2853–2858. doi: 10.1001/jama.288.22.2853. [DOI] [PubMed] [Google Scholar]
- 152.Patterson J.E., Zidouh A., Miniter P., Andriole V.T., Patterson T.F. Hospital epidemiologic surveillance for invasive aspergillosis: patient demographics and the utility of antigen detection. Infect Control Hosp Epidemiol. 1997;18:104–108. doi: 10.1086/647563. [DOI] [PubMed] [Google Scholar]
- 153.Arnow P.M., Andersen R.L., Mainous P.D., Smith E.J. Pumonary aspergillosis during hospital renovation. Am Rev Respir Dis. 1978;118:49–53. doi: 10.1164/arrd.1978.118.1.49. [DOI] [PubMed] [Google Scholar]
- 154.Pegues D.A., Lasker B.A., McNeil M.M., Hamm P.M., Lundal J.L., Kubak B.M. Cluster of cases of invasive aspergillosis in a transplant intensive care unit: evidence of person-to-person airborne transmission. Clin Infect Dis. 2002;34:412–416. doi: 10.1086/338025. [DOI] [PubMed] [Google Scholar]
- 155.Buffington J., Reporter R., Lasker B.A. Investigation of an epidemic of invasive aspergillosis: utility of molecular typing with the use of random amplified polymorphic DNA probes. Pediatr Infect Dis J. 1994;13:386–393. [PubMed] [Google Scholar]
- 156.Krasinski K., Holzman R.S., Hanna B., Greco M.A., Graff M., Bhogal M. Nosocomial fungal infection during hospital renovation. Infect Control. 1985;6:278–282. doi: 10.1017/s0195941700061750. [DOI] [PubMed] [Google Scholar]
- 157.Humphreys H. Positive-pressure isolation and the prevention of invasive aspergillosis: what is the evidence? J Hosp Infect. 2004;56:93–100. doi: 10.1016/j.jhin.2003.10.011. [DOI] [PubMed] [Google Scholar]
- 158.Thio C.L., Smith D., Merz W.G. Refinements of environmental assessment during an outbreak investigation of invasive aspergillosis in a leukemia and bone marrow transplant unit. Infect Control Hosp Epidemiol. 2000;21:18–23. doi: 10.1086/501691. [DOI] [PubMed] [Google Scholar]
- 159.Anaissie E.J., Stratton S.L., Dignani M.C. Pathogenic Aspergillus species recovered from a hospital water system: a 3-year prospective study. Clin Infect Dis. 2002;34:780–789. doi: 10.1086/338958. [DOI] [PubMed] [Google Scholar]
- 160.Centers for Disease Control and Prevention Prevention of invasive group A streptococcal disease among household contacts of case patients and among postpartum and postsurgical patients: recommendations from the Centers for Disease Control and Prevention. Clin Infect Dis. 2002;35:950–959. doi: 10.1086/342692. [DOI] [PubMed] [Google Scholar]
- 161.Gruteke P., van Belkum A., Schouls L.M. Outbreak of group A streptococci in a burn center: use of pheno- and genotypic procedures for strain tracking. J Clin Microbiol. 1996;34:114–118. doi: 10.1128/jcm.34.1.114-118.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Greene C.M., Van Beneden C.A., Javadi M. Cluster of deaths from group A streptococcus in a long-term care facility, Georgia, 2001. Am J Infect Control. 2005;33:108–113. doi: 10.1016/j.ajic.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 163.Sabria M., Campins M. Legionnaires' disease: update on epidemiology and management options. Am J Respir Med. 2003;2:235–243. doi: 10.1007/BF03256652. [DOI] [PubMed] [Google Scholar]
- 164.Bille J., Marchetti O., Calandra T. Changing face of health care–associated fungal infections. Curr Opin Infect Dis. 2005;18:314–319. doi: 10.1097/01.qco.0000171924.39828.fb. [DOI] [PubMed] [Google Scholar]
- 165.Hall I.C., O'Toole E. Intestinal flora in newborn infants with a description of a new pathogenic anaerobe, Bacillus difficilis. Am J Dis Child. 1935;49:390–402. [Google Scholar]
- 166.George W.L., Sutter V.L., Finegold S.M. Antimicrobial agent–induced diarrhea: a bacterial disease. J Infect Dis. 1977;136:822–828. doi: 10.1093/infdis/136.6.822. [DOI] [PubMed] [Google Scholar]
- 167.McFarland L.V., Mulligan M.E., Kwok R.Y., Stamm W.E. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320:204–210. doi: 10.1056/NEJM198901263200402. [DOI] [PubMed] [Google Scholar]
- 168.Pepin J., Valiquette L., Alary M.E. Clostridium difficile–associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity. CMAJ. 2004;171:466–472. doi: 10.1503/cmaj.1041104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Agency H.P. Outbreak of Clostridium difficile infection in a hospital in southeast England. Commun Dis Rep CDR Wkly. 2005;31 [Google Scholar]
- 170.Koopmans M., Wilbrink B., Conyn M. Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet. 2004;363:587–593. doi: 10.1016/S0140-6736(04)15589-X. [DOI] [PubMed] [Google Scholar]
- 171.McDonald L.C., Killgore G.E., Thompson A. An epidemic, toxin gene-variant strain of Clostridium difficile. N Engl J Med. 2005;353:2433–2441. doi: 10.1056/NEJMoa051590. [DOI] [PubMed] [Google Scholar]
- 172.Loo V.G., Poirier L., Miller M.A. A predominantly clonal multi-institutional outbreak of Clostridium difficile–associated diarrhea with high morbidity and mortality. N Engl J Med. 2005;353:2442–2449. doi: 10.1056/NEJMoa051639. [DOI] [PubMed] [Google Scholar]
- 173.Warny M., Pepin J., Fang A. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366:1079–1084. doi: 10.1016/S0140-6736(05)67420-X. [DOI] [PubMed] [Google Scholar]
- 174.Layton BAML, Gerding DN, Liedtke LA, Strausbaugh LJ. Perceived increases in the incidence and severity of Clostridium difficile disease: an emerging threat that continues to unfold. Presented at the 15th Annual Scientific Meeting of the Society for Healthcare Epidemiology of America, Los Angeles, CA, 2005.
- 175.Sohn S., Climo M., Diekema D. Varying rates of Clostridium difficile–associated diarrhea at prevention epicenter hospitals. Infect Control Hosp Epidemiol. 2005;26:676–679. doi: 10.1086/502601. [DOI] [PubMed] [Google Scholar]
- 176.Harrison P.F., Lederberg J., editors. Institute of Medicine. Antimicrobial resistance: issues and options. National Academy Press; Washington, DC: 1998. pp. 8–74. [PubMed] [Google Scholar]
- 177.Shlaes D.M., Gerding D.N., John J.F., Jr. Society for Healthcare Epidemiology of America and Infectious Diseases Society of America Joint Committee on the Prevention of Antimicrobial Resistance guidelines for the prevention of antimicrobial resistance in hospitals. Infect Control Hosp Epidemiol. 1997;18:275–291. doi: 10.1086/647610. [DOI] [PubMed] [Google Scholar]
- 178.Whitener C.J., Park S.Y., Browne F.A. Vancomycin-resistant Staphylococcus aureus in the absence of vancomycin exposure. Clin Infect Dis. 2004;38:1049–1055. doi: 10.1086/382357. [DOI] [PubMed] [Google Scholar]
- 179.Centers for Disease Control and Prevention Staphylococcus aureus with reduced susceptibility to vancomycin, United States. MMWR Morb Mortal Wkly Rep. 1997;46:765–766. [PubMed] [Google Scholar]
- 180.Centers for Disease Control and Prevention Staphylococcus aureus resistant to vancomycin, United States, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:565–567. [PubMed] [Google Scholar]
- 181.Centers for Disease Control and Prevention Public health dispatch: vancomycin-resistant Staphylococcus aureus, Pennsylvania, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:902. [PubMed] [Google Scholar]
- 182.Centers for Disease Control and Prevention Vancomycin-resistant Staphylococcus aureus, New York, 2004. MMWR Morb Mortal Wkly Rep. 2004;53:322–323. [PubMed] [Google Scholar]
- 183.Chang S., Sievert D.M., Hageman J.C. Infection with vancomycin-resistant Staphylococcus aureus containing the vanA resistance gene. N Engl J Med. 2003;348:1342–1347. doi: 10.1056/NEJMoa025025. [DOI] [PubMed] [Google Scholar]
- 184.Fridkin S.K., Hageman J., McDougal L.K. Epidemiological and microbiological characterization of infections caused by Staphylococcus aureus with reduced susceptibility to vancomycin, United States, 1997–2001. Clin Infect Dis. 2003;36:429–439. doi: 10.1086/346207. [DOI] [PubMed] [Google Scholar]
- 185.Gold H.S., Moellering R.C., Jr. Antimicrobial drug resistance. N Engl J Med. 1996;335:1445–1453. doi: 10.1056/NEJM199611073351907. [DOI] [PubMed] [Google Scholar]
- 186.Hageman J.C., Pegues D.A., Jepson C. Vancomycin-intermediate Staphylococcus aureus in a home health care patient. Emerg Infect Dis. 2001;7:1023–1025. doi: 10.3201/eid0706.010618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Harwell J.I., Brown R.B. The drug-resistant pneumococcus: clinical relevance, therapy, and prevention. Chest. 2000;117:530–541. doi: 10.1378/chest.117.2.530. [DOI] [PubMed] [Google Scholar]
- 188.Jones R.N. Resistance patterns among nosocomial pathogens: trends over the past few years. Chest. 2001;119(2 Suppl):397S–404S. doi: 10.1378/chest.119.2_suppl.397s. [DOI] [PubMed] [Google Scholar]
- 189.Murray B.E. Vancomycin-resistant enterococcal infections. N Engl J Med. 2000;342:710–721. doi: 10.1056/NEJM200003093421007. [DOI] [PubMed] [Google Scholar]
- 190.Neuhauser M.M., Weinstein R.A., Rydman R., Danziger L.H., Karam G., Quinn J.P. Antibiotic resistance among Gram-negative bacilli in US intensive care units: implications for fluoroquinolone use. JAMA. 2003;289:885–888. doi: 10.1001/jama.289.7.885. [DOI] [PubMed] [Google Scholar]
- 191.Pitout J.D., Sanders C.C., Sanders W.E., Jr. Antimicrobial resistance with focus on beta-lactam resistance in Gram-negative bacilli. Am J Med. 1997;103:51–59. doi: 10.1016/s0002-9343(97)00044-2. [DOI] [PubMed] [Google Scholar]
- 192.Rotun S.S., McMath V., Schoonmaker D.J. Staphylococcus aureus with reduced susceptibility to vancomycin isolated from a patient with fatal bacteremia. Emerg Infect Dis. 1999;5:147–149. doi: 10.3201/eid0501.990118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Smith T.L., Pearson M.L., Wilcox K.R. Emergence of vancomycin resistance in Staphylococcus aureus. Glycopeptide-Intermediate Staphylococcus aureus Working Group. N Engl J Med. 1999;340:493–501. doi: 10.1056/NEJM199902183400701. [DOI] [PubMed] [Google Scholar]
- 194.Srinivasan A., Dick J.D., Perl T.M. Vancomycin resistance in staphylococci. Clin Microbiol Rev. 2002;15:430–438. doi: 10.1128/CMR.15.3.430-438.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Hyle E.P., Lipworth A.D., Zaoutis T.E. Risk factors for increasing multidrug resistance among extended-spectrum ß-lactamase–producing Escheria coli and Klebsiella species. Clin Infect Dis. 2005;40:1317–1324. doi: 10.1086/429239. [DOI] [PubMed] [Google Scholar]
- 196.Gleich S., Morad Y., Echague R. Streptococcus pneumoniae serotype 4 outbreak in a home for the aged: report and review of recent outbreaks. Infect Control Hosp Epidemiol. 2000;21:711. doi: 10.1086/501717. [DOI] [PubMed] [Google Scholar]
- 197.Fry A.M., Udeagu C.C., Soriano-Gabarro M. Persistence of fluoroquinolone-resistant, multidrug-resistant Streptococcus pneumoniae in a long-term-care facility: efforts to reduce intrafacility transmission. Infect Control Hosp Epidemiol. 2005;26:239–247. doi: 10.1086/502533. [DOI] [PubMed] [Google Scholar]
- 198.Carter R.J., Sorenson G., Heffernan R. Failure to control an outbreak of multidrug-resistant Streptococcus pneumoniae in a long-term-care facility: emergence and ongoing transmission of a fluoroquinolone-resistant strain. Infect Control Hosp Epidemiol. 2005;26:248–255. doi: 10.1086/502534. [DOI] [PubMed] [Google Scholar]
- 199.Blok H.E., Troelstra A., Kamp-Hopmans T.E. Role of healthcare workers in outbreaks of methicillin-resistant Staphylococcus aureus: a 10-year evaluation from a Dutch university hospital. Infect Control Hosp Epidemiol. 2003;24:679–685. doi: 10.1086/502275. [DOI] [PubMed] [Google Scholar]
- 200.Muto C.A., Jernigan J.A., Ostrowsky B.E. SHEA guideline for preventing nosocomial transmission of multidrug-resistant strains of Staphylococcus aureus and enterococcus. Infect Control Hosp Epidemiol. 2003;24:362–386. doi: 10.1086/502213. [DOI] [PubMed] [Google Scholar]
- 201.Tammelin A., Klotz F., Hambraeus A., Stahle E., Ransjo U. Nasal and hand carriage of Staphylococcus aureus in staff at a Department for Thoracic and Cardiovascular Surgery: endogenous or exogenous source? Infect Control Hosp Epidemiol. 2003;24:686–689. doi: 10.1086/502277. [DOI] [PubMed] [Google Scholar]
- 202.Centers for Disease Control and Prevention Biological and chemical terrorism: strategic plan for preparedness and response. Recommendations of the CDC Strategic Planning Workgroup. MMWR Recomm Rep. 2000;49:1–14. [PubMed] [Google Scholar]
- 203.Inglesby T.V., O'Toole T., Henderson D.A. Anthrax as a biological weapon, 2002: updated recommendations for management. JAMA. 2002;287:2236–2252. doi: 10.1001/jama.287.17.2236. [DOI] [PubMed] [Google Scholar]
- 204.Henderson D.A., Inglesby T.V., Bartlett J.G. Smallpox as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA. 1999;281:2127–2137. doi: 10.1001/jama.281.22.2127. [DOI] [PubMed] [Google Scholar]
- 205.www.bt.cdc.gov/agent/smallpox/.
- 206.www.who.int/csr/disease/smallpox/en/.
- 207.Kool J.L. Risk of person-to-person transmission of pneumonic plague. Clin Infect Dis. 2005;40:1166–1172. doi: 10.1086/428617. [DOI] [PubMed] [Google Scholar]
- 208.Inglesby T.V., Dennis D.T., Henderson D.A. Plague as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA. 2000;283:2281–2290. doi: 10.1001/jama.283.17.2281. [DOI] [PubMed] [Google Scholar]
- 209.Arnon S.S., Schechter R., Inglesby T.V. Botulinum toxin as a biological weapon: medical and public health management. JAMA. 2001;285:1059–1070. doi: 10.1001/jama.285.8.1059. [DOI] [PubMed] [Google Scholar]
- 210.Dennis D.T., Inglesby T.V., Henderson D.A. Tularemia as a biological weapon: medical and public health management. JAMA. 2001;285:2763–2773. doi: 10.1001/jama.285.21.2763. [DOI] [PubMed] [Google Scholar]
- 211.Centers for Disease Control and Prevention Notice to readers update: management of patients with suspected viral hemorrhagic fever, United States. MMWR Recomm Rep. 1995;44:475–479. [PubMed] [Google Scholar]
- 212.Borio L., Inglesby T., Peters C.J. Hemorrhagic fever viruses as biological weapons: medical and public health management. JAMA. 2002;287:2391–2405. doi: 10.1001/jama.287.18.2391. [DOI] [PubMed] [Google Scholar]
- 213.Neff J.M., Lane J.M., Fulginiti V.A., Henderson D.A. Contact vaccinia: transmission of vaccinia from smallpox vaccination. JAMA. 2002;288:1901–1905. doi: 10.1001/jama.288.15.1901. [DOI] [PubMed] [Google Scholar]
- 214.Sepkowitz K.A. How contagious is vaccinia? N Engl J Med. 2003;348:439–446. doi: 10.1056/NEJMra022500. [DOI] [PubMed] [Google Scholar]
- 215.Lane J.M., Fulginiti V.A. Transmission of vaccinia virus and rationale for measures for prevention. Clin Infect Dis. 2003;37:281–284. doi: 10.1086/377236. [DOI] [PubMed] [Google Scholar]
- 216.Centers for Disease Control and Prevention Smallpox vaccination and adverse reactions: guidance for clinicians. MMWR Morb Mortal Wkly Rep. 2003;52:1–28. [PubMed] [Google Scholar]
- 217.Fulginiti V.A., Papier A., Lane J.M., Neff J.M., Henderson D.A. Smallpox vaccination: a review. Part II: adverse events. Clin Infect Dis. 2003;37:251–271. doi: 10.1086/375825. [DOI] [PubMed] [Google Scholar]
- 218.www.smallpox.mil./event/SPSafetySum.asp.
- 219.Centers for Disease Control and Prevention Update: adverse events following civilian smallpox vaccination, United States, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:419–420. [PubMed] [Google Scholar]
- 220.Centers for Disease Control and Prevention Secondary and tertiary transfer of vaccinia virus among US military personnel, United States and worldwide, 2002–2004. MMWR Morb Mortal Wkly Rep. 2004;53:103–105. [PubMed] [Google Scholar]
- 221.Talbot T.R., Ziel E., Doersam J.K., LaFleur B., Tollefson S., Edwards K.M. Risk of vaccinia transfer to the hands of vaccinated persons after smallpox immunization. Clin Infect Dis. 2004;38:536–541. doi: 10.1086/381205. [DOI] [PubMed] [Google Scholar]
- 222.Hepburn M.J., Dooley D.P., Murray C.K. Frequency of vaccinia virus isolation on semipermeable versus nonocclusive dressings covering smallpox vaccination sites in hospital personnel. Am J Infect Control. 2004;32:126–130. doi: 10.1016/j.ajic.2003.08.006. [DOI] [PubMed] [Google Scholar]
- 223.Waibel K.H., Ager E.P., Topolski R.L., Walsh D.S. Randomized trial comparing vaccinia on the external surfaces of 3 conventional bandages applied to smallpox vaccination sites in primary vaccinees. Clin Infect Dis. 2004;39:1004–1007. doi: 10.1086/423967. [DOI] [PubMed] [Google Scholar]
- 224.Tenorio AR, Peeples M, Patri M, et al. Quantitative vaccinia cultures and evolution of vaccinia-specific CD8+ cytotoxic T-lymphocyte (CTL) responses in revaccinees [abstract 823]. Presented at the Infectious Diseases Society of America 41st annual meeting, San Diego, CA, October 2003.
- 225.Wharton M., Strikas R.A., Harpaz R. Recommendations for using smallpox vaccine in a pre-event vaccination program: supplemental recommendations of the Advisory Committee on Immunization Practices (ACIP) and the Healthcare Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 2003;52:1–16. [PubMed] [Google Scholar]
- 226.Centers for Disease Control and Prevention Surveillance for Creutzfeldt-Jakob disease, United States. MMWR Morb Mortal Wkly Rep. 1996;45:665–668. [PubMed] [Google Scholar]
- 227.Johnson R.T., Gibbs C.J., Jr. Creutzfeldt-Jakob disease and related transmissible spongiform encephalopathies. N Engl J Med. 1998;339:1994–2004. doi: 10.1056/NEJM199812313392707. [DOI] [PubMed] [Google Scholar]
- 228.Brown P., Gajdusek D.C., Gibbs C.J., Jr., Asher D.M. Potential epidemic of Creutzfeldt-Jakob disease from human growth hormone therapy. N Engl J Med. 1985;313:728–731. doi: 10.1056/NEJM198509193131205. [DOI] [PubMed] [Google Scholar]
- 229.Frasier S.D., Foley T.P., Jr. Clinical review 58: Creutzfeldt-Jakob disease in recipients of pituitary hormones. J Clin Endocrinol Metab. 1994;78:1277–1279. doi: 10.1210/jcem.78.6.8200924. [DOI] [PubMed] [Google Scholar]
- 230.Centers for Disease Control and Prevention Update: Creutzfeldt-Jakob disease associated with cadaveric dura mater grafts, Japan, 1979–2003. MMWR Morb Mortal Wkly Rep. 2003;52(48):1179–1181. [PubMed] [Google Scholar]
- 231.Lang C.J., Heckmann J.G., Neundorfer B. Creutzfeldt-Jakob disease via dural and corneal transplants. J Neurol Sci. 1998;160:128–139. doi: 10.1016/s0022-510x(98)00226-3. [DOI] [PubMed] [Google Scholar]
- 232.el Hachimi K.H., Chaunu M.P., Cervenakova L., Brown P., Foncin J.F. Putative neurosurgical transmission of Creutzfeldt-Jakob disease with analysis of donor and recipient: agent strains. C R Acad Sci Ser III. 1997;320:319–328. doi: 10.1016/s0764-4469(97)82774-6. [DOI] [PubMed] [Google Scholar]
- 233.Will R.G., Matthews W.B. Evidence for case-to-case transmission of Creutzfeldt-Jakob disease. J Neurol Neurosurg Psychiatry. 1982;45:235–238. doi: 10.1136/jnnp.45.3.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Bernoulli C., Siegfried J., Baumgartner G. Danger of accidental person-to-person transmission of Creutzfeldt-Jakob disease by surgery. Lancet. 1977;1:478–479. doi: 10.1016/s0140-6736(77)91958-4. [DOI] [PubMed] [Google Scholar]
- 235.Rutala W.A., Weber D.J. Creutzfeldt-Jakob disease: recommendations for disinfection and sterilization. Clin Infect Dis. 2001;32:1348–1356. doi: 10.1086/319997. [DOI] [PubMed] [Google Scholar]
- 236.Belay E.D., Maddox R.A., Williams E.S., Miller M.W., Gambetti P., Schonberger L.B. Chronic wasting disease and potential transmission to humans. Emerg Infect Dis. 2004;10:977–984. doi: 10.3201/eid1006.031082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Collinge J., Sidle K.C., Meads J., Ironside J., Hill A.F. Molecular analysis of prion strain variation and the aetiology of “new variant” CJD. Nature. 1996;383:685–690. doi: 10.1038/383685a0. [DOI] [PubMed] [Google Scholar]
- 238.Belay E.D., Schonberger L.B. The public health impact of prion diseases. Annu Rev Public Health. 2005;26:191–212. doi: 10.1146/annurev.publhealth.26.021304.144536. [DOI] [PubMed] [Google Scholar]
- 239.Belay E.D., Schonberger L.B. Variant Creutzfeldt-Jakob disease and bovine spongiform encephalopathy. Clin Lab Med. 2002;22:849–862. doi: 10.1016/s0272-2712(02)00024-0. [DOI] [PubMed] [Google Scholar]
- 240.Hill A.F., Butterworth R.J., Joiner S. Investigation of variant Creutzfeldt-Jakob disease and other human prion diseases with tonsil biopsy samples. Lancet. 1999;353:183–189. doi: 10.1016/s0140-6736(98)12075-5. [DOI] [PubMed] [Google Scholar]
- 241.Evatt B. Creutzfeldt-Jakob disease and haemophilia: assessment of risk. Haemophilia. 2000;6(Suppl 1):94–99. doi: 10.1046/j.1365-2516.2000.00052.x. [DOI] [PubMed] [Google Scholar]
- 242.Chamberland M.E. Emerging infectious agents: do they pose a risk to the safety of transfused blood and blood products? Clin Infect Dis. 2002;34:797–805. doi: 10.1086/338787. [DOI] [PubMed] [Google Scholar]
- 243.Available from. Accessed.
- 244.Llewelyn C.A., Hewitt P.E., Knight R.S. Possible transmission of variant Creutzfeldt-Jakob disease by blood transfusion. Lancet. 2004;363:417–421. doi: 10.1016/S0140-6736(04)15486-X. [DOI] [PubMed] [Google Scholar]
- 245.Peden A.H., Head M.W., Ritchie D.L., Bell J.E., Ironside J.W. Preclinical vCJD after blood transfusion in a PRNP codon 129 heterozygous patient. Lancet. 2004;364:527–529. doi: 10.1016/S0140-6736(04)16811-6. [DOI] [PubMed] [Google Scholar]
- 246.Brown P. Guidelines for high-risk autopsy cases: special precautions for Creutzfeldt-Jakob disease. In: Hutchins G., editor. Autopsy performance and reporting. College of American Pathologists; Northfield (IL): 1990. pp. 68–74. [Google Scholar]
- 247.Drosten C., Gunther S., Preiser W. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967–1976. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- 248.Ksiazek T.G., Erdman D., Goldsmith C.S. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953–1966. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 249.Chan W.M., Kwan Y.W., Wan H.S., Leung C.W., Chiu M.C. Epidemiologic linkage and public health implication of a cluster of severe acute respiratory syndrome in an extended family. Pediatr Infect Dis J. 2004;23:1156–1159. [PubMed] [Google Scholar]
- 250.Leung C.W., Kwan Y.W., Ko P.W. Severe acute respiratory syndrome among children. Pediatrics. 2004;113:e535–e543. doi: 10.1542/peds.113.6.e535. [DOI] [PubMed] [Google Scholar]
- 251.Bitnun A., Allen U., Heurter H. Children hospitalized with severe acute respiratory syndrome–related illness in Toronto. Pediatrics. 2003;112:e261. doi: 10.1542/peds.112.4.e261. [DOI] [PubMed] [Google Scholar]
- 252.Chow K.Y., Lee C.E., Ling M.L., Heng D.M., Yap S.G. Outbreak of severe acute respiratory syndrome in a tertiary hospital in Singapore, linked to an index patient with atypical presentation: epidemiological study. BMJ. 2004;328:195. doi: 10.1136/bmj.37939.465729.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Shen Z., Ning F., Zhou W. Super-spreading SARS events, Beijing, 2003. Emerg Infect Dis. 2004;10:256–260. doi: 10.3201/eid1002.030732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Chen Y.-C., Huang L.-M., Chan C.-C., Su C.-P., Chang S.-C., Chang Y.-Y. SARS in hospital emergency room. Emerg Infect Dis. 2004;10:782–788. doi: 10.3201/eid1005.030579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Gamage B., Moore D., Copes R., Yassi A., Bryce E. Protecting health care workers from SARS and other respiratory pathogens: a review of the infection control literature. Am J Infect Control. 2005;33:114–121. doi: 10.1016/j.ajic.2004.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Moore D., Gamage B., Bryce E., Copes R., Yassi A. Protecting health care workers from SARS and other respiratory pathogens: organizational and individual factors that affect adherence to infection control guidelines. Am J Infect Control. 2005;33:88–96. doi: 10.1016/j.ajic.2004.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Dowell S.F., Simmerman J.M., Erdman D.D. Severe acute respiratory syndrome coronavirus on hospital surfaces. Clin Infect Dis. 2004;39:652–657. doi: 10.1086/422652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Public Health Guidance for Community-Level Preparedness and Response to Severe Acute Respiratory Syndrome (SARS). 2004. Available from http://www.cdc.gov/ncidod/sars/guidance/I/occupational.htm.
- 259.Le D.H., Bloom S.A., Nguyen Q.H. Lack of SARS transmission among public hospital workers. Vietnam. Emerg Infect Dis. 2004;10:265–268. doi: 10.3201/eid1002.030707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Lim P.L., Kurup A., Gopalakrishna G. Laboratory-acquired severe acute respiratory syndrome. N Engl J Med. 2004;350:1740–1745. doi: 10.1056/NEJMoa032565. [DOI] [PubMed] [Google Scholar]
- 261.Centers for Disease Control and Prevention. 2003. Available from www.cdc.gov/ncidod/sars.
- 262.Reed K.D., Melski J.W., Graham M.B. The detection of monkeypox in humans in the Western Hemisphere. N Engl J Med. 2004;350:342–350. doi: 10.1056/NEJMoa032299. [DOI] [PubMed] [Google Scholar]
- 263.Anderson M.G., Frenkel L.D., Homann S., Guffey J. A case of severe monkeypox virus disease in an American child: emerging infections and changing professional values. Pediatr Infect Dis J. 2003;22:1093–1096. doi: 10.1097/01.inf.0000101821.61387.a5. [DOI] [PubMed] [Google Scholar]
- 264.Jezek Z., Fenner F. Human monkey pox. In: Melnick J.L., editor. Vol. 17. S Karger; Basel, Switzerland: 1988. pp. 81–102. (Monographs in virology). [Google Scholar]
- 265.Marennikova S.S., Jezek Z., Szczeniowski M., Mbudi P.M., Vernette M. Contagiousness of monkey pox for humans: results of an investigation of 2 outbreaks of the infection in Zaire. Zh Mikrobiol Epidemiol Immunobiol. 1985:38–43. [PubMed] [Google Scholar]
- 266.Jezek Z., Arita I., Mutombo M., Dunn C., Nakano J.H., Szczeniowski M. Four generations of probable person-to-person transmission of human monkeypox. Am J Epidemiol. 1986;123:1004–1012. doi: 10.1093/oxfordjournals.aje.a114328. [DOI] [PubMed] [Google Scholar]
- 267.Learned L.A., Reynolds M.G., Wassa D.W. Extended interhuman transmission of monkeypox in a hospital community in the Republic of the Congo, 2003. Am J Trop Med Hyg. 2005;73:428–434. [PubMed] [Google Scholar]
- 268.Fleischauer A.T., Kile J.C., Davidson M. Evaluation of human-to-human transmission of monkeypox from infected patients to health care workers. Clin Infect Dis. 2005;40:689–694. doi: 10.1086/427805. [DOI] [PubMed] [Google Scholar]
- 269.Likos A.M., Sammons S.A., Olson V.A. A tale of two clades: monkeypox viruses. J Gen Virol. 2005;86(Pt 10):2661–2672. doi: 10.1099/vir.0.81215-0. [DOI] [PubMed] [Google Scholar]
- 270.Fine P.E., Jezek Z., Grab B., Dixon H. The transmission potential of monkeypox virus in human populations. Int J Epidemiol. 1988;17:643–650. doi: 10.1093/ije/17.3.643. [DOI] [PubMed] [Google Scholar]
- 271.Jezek Z., Grab B., Paluku K.M., Szczeniowski M.V. Human monkeypox: disease pattern, incidence and attack rates in a rural area of northern Zaire. Trop Geogr Med. 1988;40:73–83. [PubMed] [Google Scholar]
- 272.Centers for Disease Control Norwalk-like viruses: public health consequences and outbreak management. MMWR Recomm Rep. 2001;50:1–18. [PubMed] [Google Scholar]
- 273.Evans M.R., Meldrum R., Lane W. An outbreak of viral gastroenteritis following environmental contamination at a concert hall. Epidemiol Infect. 2002;129:355–360. doi: 10.1017/s0950268802007446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Wu H.M., Fornek M., Kellogg J.S. A norovirus outbreak at a long-term-care facility: the role of environmental surface contamination. Infect Control Hosp Epidemiol. 2005;26:802–810. doi: 10.1086/502497. [DOI] [PubMed] [Google Scholar]
- 275.Duizer E., Schwab K.J., Neill F.H., Atmar R.L., Koopmans M.P., Estes M.K. Laboratory efforts to cultivate noroviruses. J Gen Virol. 2004;85(Pt 1):79–87. doi: 10.1099/vir.0.19478-0. [DOI] [PubMed] [Google Scholar]
- 276.Zingg W., Colombo C., Jucker T., Bossart W., Ruef C. Impact of an outbreak of norovirus infection on hospital resources. Infect Control Hosp Epidemiol. 2005;26:263–267. doi: 10.1086/502537. [DOI] [PubMed] [Google Scholar]
- 277.Calderon-Margalit R., Sheffer R., Halperin T., Orr N., Cohen D., Shohat T. A large-scale gastroenteritis outbreak associated with norovirus in nursing homes. Epidemiol Infect. 2005;133:35–40. doi: 10.1017/s0950268804003115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Marx A., Shay D.K., Noel J.S. An outbreak of acute gastroenteritis in a geriatric long-term-care facility: combined application of epidemiological and molecular diagnostic methods. Infect Control Hosp Epidemiol. 1999;20:306–311. doi: 10.1086/501622. [DOI] [PubMed] [Google Scholar]
- 279.Gellert G.A., Waterman S.H., Ewert D. An outbreak of acute gastroenteritis caused by a small round structured virus in a geriatric convalescent facility. Infect Control Hosp Epidemiol. 1990;11:459–464. doi: 10.1086/646212. [DOI] [PubMed] [Google Scholar]
- 280.Cooper E., Blamey S. A norovirus gastroenteritis epidemic in a long-term-care facility. Infect Control Hosp Epidemiol. 2005;26:256. doi: 10.1086/502535. [DOI] [PubMed] [Google Scholar]
- 281.Navarro G., Sala R.M., Segura F. An outbreak of norovirus infection in a long-term-care facility. Infect Control Hosp Epidemiol. 2005;26:259. doi: 10.1086/502536. [DOI] [PubMed] [Google Scholar]
- 282.Green K.Y., Belliot G., Taylor J.L. A predominant role for Norwalk-like viruses as agents of epidemic gastroenteritis in Maryland nursing homes for the elderly. J Infect Dis. 2002;185:133–146. doi: 10.1086/338365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Widdowson M.A., Cramer E.H., Hadley L. Outbreaks of acute gastroenteritis on cruise ships and on land: identification of a predominant circulating strain of norovirus, United States, 2002. J Infect Dis. 2004;190:27–36. doi: 10.1086/420888. [DOI] [PubMed] [Google Scholar]
- 284.Centers for Disease Control and Prevention Outbreaks of gastroenteritis associated with noroviruses on cruise ships, United States, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:1112–1115. [PubMed] [Google Scholar]
- 285.Centers for Disease Control and Prevention Norovirus outbreak among evacuees from hurricane Katrina, Houston, Texas, September 2005. MMWR Morb Mortal Wkly Rep. 2005;54:1016–1018. [PubMed] [Google Scholar]
- 286.Mattner F., Mattner L., Borck H.U., Gastmeier P. Evaluation of the impact of the source (patient versus staff) on nosocomial norovirus outbreak severity. Infect Control Hosp Epidemiol. 2005;26:268–272. doi: 10.1086/502538. [DOI] [PubMed] [Google Scholar]
- 287.Isakbaeva E.T., Bulens S.N., Beard R.S. Norovirus and child care: challenges in outbreak control. Pediatr Infect Dis J. 2005;24:561–563. doi: 10.1097/01.inf.0000164764.58715.23. [DOI] [PubMed] [Google Scholar]
- 288.Kapikian A.Z., Estes M.K., Chanock R.M. Norwalk group of viruses. In: Fields B.N., Knipe D.M., Howley P.M., editors. Fields virology. 3rd ed. Lippincott-Raven; Philadelphia: 1996. pp. 783–810. [Google Scholar]
- 289.Duizer E., Bijkerk P., Rockx B., De Groot A., Twisk F., Koopmans M. Inactivation of caliciviruses. Appl Environ Microbiol. 2004;70:4538–4543. doi: 10.1128/AEM.70.8.4538-4543.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Doultree J.C., Druce J.D., Birch C.J., Bowden D.S., Marshall J.A. Inactivation of feline calicivirus, a Norwalk virus surrogate. J Hosp Infect. 1999;41:51–57. doi: 10.1016/s0195-6701(99)90037-3. [DOI] [PubMed] [Google Scholar]
- 291.Barker J., Vipond I.B., Bloomfield S.F. Effects of cleaning and disinfection in reducing the spread of norovirus contamination via environmental surfaces. J Hosp Infect. 2004;58:42–49. doi: 10.1016/j.jhin.2004.04.021. [DOI] [PubMed] [Google Scholar]
- 292.Gulati B.R., Allwood P.B., Hedberg C.W., Goyal S.M. Efficacy of commonly used disinfectants for the inactivation of calicivirus on strawberry, lettuce, and a food-contact surface. J Food Prot. 2001;64:1430–1434. doi: 10.4315/0362-028x-64.9.1430. [DOI] [PubMed] [Google Scholar]
- 293.Gehrke C., Steinmann J., Goroncy-Bermes P. Inactivation of feline calicivirus, a surrogate of norovirus (formerly Norwalk-like viruses), by different types of alcohol in vitro and in vivo. J Hosp Infect. 2004;56:49–55. doi: 10.1016/j.jhin.2003.08.019. [DOI] [PubMed] [Google Scholar]
- 294.Hutson A.M., Atmar R.L., Graham D.Y., Estes M.K. Norwalk virus infection and disease is associated with ABO histo-blood group type. J Infect Dis. 2002;185:1335–1337. doi: 10.1086/339883. [DOI] [PubMed] [Google Scholar]
- 295.National Center for Infectious Diseases, Division of Viral and Rickettsial Diseases. Available from. Accessed.
- 296.LeDuc J.W. Epidemiology of hemorrhagic fever viruses. Rev Infect Dis. 1989;11(Suppl 4):S730–S735. doi: 10.1093/clinids/11.supplement_4.s730. [DOI] [PubMed] [Google Scholar]
- 297.Roels T.H., Bloom A.S., Buffington J. . Ebola hemorrhagic fever, Kikwit, Democratic Republic of the Congo, 1995: risk factors for patients without a reported exposure. J Infect Dis. 1999;179(Suppl 1):S92–S97. doi: 10.1086/514286. [DOI] [PubMed] [Google Scholar]
- 298.Suleiman M.N., Muscat-Baron J.M., Harries J.R. Congo/Crimean haemorrhagic fever in Dubai: an outbreak at the Rashid Hospital. Lancet. 1980;21:939–941. [PubMed] [Google Scholar]
- 299.Monath T.P., Mertens P.E., Patton R. A hospital epidemic of Lassa fever in Zorzor, Liberia, March–April 1972. Am J Trop Med Hyg. 1973;22:773–779. doi: 10.4269/ajtmh.1973.22.773. [DOI] [PubMed] [Google Scholar]
- 300.Dowell S.F., Mukunu R., Ksiazek T.G., Khan A.S., Rollin P.E., Peters C.J. Transmission of Ebola hemorrhagic fever: a study of risk factors in family members, Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis. 1999;179(Suppl 1):S87–S91. doi: 10.1086/514284. [DOI] [PubMed] [Google Scholar]
- 301.Peters C.J. Marburg and Ebola: arming ourselves against the deadly filoviruses. N Engl J Med. 2005;352:2571–2573. doi: 10.1056/NEJMp058109. [DOI] [PubMed] [Google Scholar]
- 302.Ebola haemorrhagic fever in Zaire, 1976. Bull World Health Organ. 1978;56:271–293. [PMC free article] [PubMed] [Google Scholar]
- 303.Emond R.T., Evans B., Bowen E.T., Lloyd G. A case of Ebola virus infection. Br Med J. 1977;2:541–544. doi: 10.1136/bmj.2.6086.541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Zaki S.R., Shieh W.J., Greer P.W. A novel immunohistochemical assay for the detection of Ebola virus in skin: implications for diagnosis, spread, and surveillance of Ebola hemorrhagic fever. Commission de Lutte contre les Epidemies a Kikwit. J Infect Dis. 1999;179(Suppl 1):S36–S47. doi: 10.1086/514319. [DOI] [PubMed] [Google Scholar]
- 305.Khan A.S., Tshioko F.K., Heymann D.L. The reemergence of Ebola hemorrhagic fever, Democratic Republic of the Congo, 1995. Commission de Lutte contre les Epidemies a Kikwit. J Infect Dis. 1999;179(Suppl 1):S76–S86. doi: 10.1086/514306. [DOI] [PubMed] [Google Scholar]
- 306.Muyembe-Tamfum J.J., Kipasa M., Kiyungu C., Colebunders R. Ebola outbreak in Kikwit, Democratic Republic of the Congo: discovery and control measures. J Infect Dis. 1999;179(Suppl 1):S259–S262. doi: 10.1086/514302. [DOI] [PubMed] [Google Scholar]
- 307.Haas W.H., Breuer T., Pfaff G. Imported Lassa fever in Germany: surveillance and management of contact persons. Clin Infect Dis. 2003;36:1254–1258. doi: 10.1086/374853. [DOI] [PubMed] [Google Scholar]
- 308.Simpson D.I. Marburg agent disease in monkeys. Trans R Soc Trop Med Hyg. 1969;63:303–309. doi: 10.1016/0035-9203(69)90002-9. [DOI] [PubMed] [Google Scholar]
- 309.Jaax N.K., Davis K.J., Geisbert T.J. Lethal experimental infection of rhesus monkeys with Ebola-Zaire (Mayinga) virus by the oral and conjunctival route of exposure. Arch Pathol Lab Med. 1996;120:140–155. [PubMed] [Google Scholar]
- 310.Stephenson E.H., Larson E.W., Dominik J.W. Effect of environmental factors on aerosol-induced Lassa virus infection. J Med Virol. 1984;14:295–303. doi: 10.1002/jmv.1890140402. [DOI] [PubMed] [Google Scholar]
- 311.Johnson E., Jaax N., White J., Jahrling P. Lethal experimental infections of rhesus monkeys by aerosolized Ebola virus. Int J Exp Pathol. 1995;76:227–236. [PMC free article] [PubMed] [Google Scholar]
- 312.Jaax N., Jahrling P., Geisbert T. Transmission of Ebola virus (Zaire strain) to uninfected control monkeys in a biocontainment laboratory. Lancet. 1995;346:1669–1671. doi: 10.1016/s0140-6736(95)92841-3. [DOI] [PubMed] [Google Scholar]
- 313.www.bt.cdc.gov/agent/vhf/.
- 314.Nguyen G.T., Proctor S.E., Sinkowitz-Cochran R.L., Garrett D.O., Jarvis W.R. Status of infection surveillance and control programs in the United States, 1992–1996. Association for Professionals in Infection Control and Epidemiology. Am J Infect Control. 2000;28:392–400. doi: 10.1067/mic.2000.110298. [DOI] [PubMed] [Google Scholar]
- 315.Richards C., Emori T.G., Edwards J., Fridkin S., Tolson J., Gaynes R. Characteristics of hospitals and infection control professionals participating in the National Nosocomial Infections Surveillance System 1999. Am J Infect Control. 2001;29:400–403. doi: 10.1067/mic.2001.118408. [DOI] [PubMed] [Google Scholar]
- 316.Wenzel R.P., Thompson R.L., Landry S.M. Hospital-acquired infections in intensive care unit patients: an overview with emphasis on epidemics. Infect Control. 1983;4:371–375. doi: 10.1017/s0195941700059774. [DOI] [PubMed] [Google Scholar]
- 317.Wenzel R.P., Gennings C. Bloodstream infections due to Candida species in the intensive care unit: identifying especially high-risk patients to determine prevention strategies. Clin Infect Dis. 2005;41(Suppl 6):S389–S393. doi: 10.1086/430923. [DOI] [PubMed] [Google Scholar]
- 318.San Miguel L.G., Cobo J., Otheo E., Sanchez-Sousa A., Abraira V., Moreno S. Secular trends of candidemia in a large tertiary-care hospital from 1988 to 2000: emergence of Candida parapsilosis. Infect Control Hosp Epidemiol. 2005;26:548–552. doi: 10.1086/502582. [DOI] [PubMed] [Google Scholar]
- 319.National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control. 2004;32:470–485. doi: 10.1016/S0196655304005425. [DOI] [PubMed] [Google Scholar]
- 320.Richards M.J., Edwards J.R., Culver D.H., Gaynes R.P. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit Care Med. 1999;27:887–892. doi: 10.1097/00003246-199905000-00020. [DOI] [PubMed] [Google Scholar]
- 321.Richards M.J., Edwards J.R., Culver D.H., Gaynes R.P. Nosocomial infections in combined medical-surgical intensive care units in the United States. Infect Control Hosp Epidemiol. 2000;21:510–515. doi: 10.1086/501795. [DOI] [PubMed] [Google Scholar]
- 322.Hugonnet S., Eggimann P., Borst F., Maricot P., Chevrolet J.C., Pittet D. Impact of ventilator-associated pneumonia on resource utilization and patient outcome. Infect Control Hosp Epidemiol. 2004;25:1090–1096. doi: 10.1086/502349. [DOI] [PubMed] [Google Scholar]
- 323.O'Neill J.M., Schutze G.E., Heulitt M.J., Simpson P.M., Taylor B.J. Nosocomial infections during extracorporeal membrane oxygenation. Intensive Care Med. 2001;27:1247–1253. doi: 10.1007/s001340101029. [DOI] [PubMed] [Google Scholar]
- 324.Villegas M.V., Hartstein A.I. Acinetobacter outbreaks, 1977–2000. Infect Control Hosp Epidemiol. 2003;24:284–295. doi: 10.1086/502205. [DOI] [PubMed] [Google Scholar]
- 325.Gordon S.M., Schmitt S.K., Jacobs M. Nosocomial bloodstream infections in patients with implantable left ventricular assist devices. Ann Thorac Surg. 2001;72:725–730. doi: 10.1016/s0003-4975(01)02888-0. [DOI] [PubMed] [Google Scholar]
- 326.Giamarellou H. Nosocomial cardiac infections. J Hosp Infect. 2002;50:91–105. doi: 10.1053/jhin.2001.1144. [DOI] [PubMed] [Google Scholar]
- 327.Fridkin S.K. Increasing prevalence of antimicrobial resistance in intensive care units. Crit Care Med. 2001;29(4 Suppl):N64–N68. doi: 10.1097/00003246-200104001-00002. [DOI] [PubMed] [Google Scholar]
- 328.Kollef M.H., Fraser V.J. Antibiotic resistance in the intensive care unit. Ann Intern Med. 2001;134:298–314. doi: 10.7326/0003-4819-134-4-200102200-00014. [DOI] [PubMed] [Google Scholar]
- 329.Fridkin S.K., Edwards J.R., Courval J.M. The effect of vancomycin and third-generation cephalosporins on prevalence of vancomycin-resistant enterococci in 126 US adult intensive care units. Ann Intern Med. 2001;135:175–183. doi: 10.7326/0003-4819-135-3-200108070-00009. [DOI] [PubMed] [Google Scholar]
- 330.Crnich C.J., Safdar N., Maki D.G. The role of the intensive care unit environment in the pathogenesis and prevention of ventilator-associated pneumonia. Respir Care. 2005;50:813–838. [PubMed] [Google Scholar]
- 331.Knaus W.A., Wagner D.P., Zimmerman J.E., Draper E.A. Variations in mortality and length of stay in intensive care units. Ann Intern Med. 1993;118:753–761. doi: 10.7326/0003-4819-118-10-199305150-00001. [DOI] [PubMed] [Google Scholar]
- 332.Villarino M.E., Stevens L.E., Schable B. Risk factors for epidemic Xanthomonas maltophilia infection/colonization in intensive care unit patients. Infect Control Hosp Epidemiol. 1992;13:201–206. doi: 10.1086/646510. [DOI] [PubMed] [Google Scholar]
- 333.Sanchez V., Vazquez J.A., Barth-Jones D., Dembry L., Sobel J.D., Zervos M.J. Nosocomial acquisition of Candida parapsilosis: an epidemiologic study. Am J Med. 1993;94:577–582. doi: 10.1016/0002-9343(93)90207-6. [DOI] [PubMed] [Google Scholar]
- 334.Husni R.N., Goldstein L.S., Arroliga A.C. Risk factors for an outbreak of multidrug-resistant Acinetobacter nosocomial pneumonia among intubated patients. Chest. 1999;115:1378–1382. doi: 10.1378/chest.115.5.1378. [DOI] [PubMed] [Google Scholar]
- 335.McDonald L.C., Walker M., Carson L. Outbreak of Acinetobacter spp bloodstream infections in a nursery associated with contaminated aerosols and air conditioners. Pediatr Infect Dis J. 1998;17:716–722. doi: 10.1097/00006454-199808000-00011. [DOI] [PubMed] [Google Scholar]
- 336.Trick W.E., Kioski C.M., Howard K.M. Outbreak of Pseudomonas aeruginosa ventriculitis among patients in a neurosurgical intensive care unit. Infect Control Hosp Epidemiol. 2000;21:204–208. doi: 10.1086/501745. [DOI] [PubMed] [Google Scholar]
- 337.Guidry G.G., Black-Payne C.A., Payne D.K., Jamison R.M., George R.B., Bocchini J.A., Jr. Respiratory syncytial virus infection among intubated adults in a university medical intensive care unit. Chest. 1991;100:1377–1384. doi: 10.1378/chest.100.5.1377. [DOI] [PubMed] [Google Scholar]
- 338.Wurtz R., Karajovic M., Dacumos E., Jovanovic B., Hanumadass M. Nosocomial infections in a burn intensive care unit. Burns. 1995;21:181–184. doi: 10.1016/0305-4179(95)80005-9. [DOI] [PubMed] [Google Scholar]
- 339.Rodgers G.L., Mortensen J., Fisher M.C., Lo A., Cresswell A., Long S.S. Predictors of infectious complications after burn injuries in children. Pediatr Infect Dis J. 2000;19:990–995. doi: 10.1097/00006454-200010000-00010. [DOI] [PubMed] [Google Scholar]
- 340.Pruitt B.A., Jr., McManus A.T., Kim S.H., Goodwin C.W. Burn wound infections: current status. World J Surg. 1998;22:135–145. doi: 10.1007/s002689900361. [DOI] [PubMed] [Google Scholar]
- 341.Weber J., editor. Epidemiology of infections and strategies for control in burn care and therapy. Mosby; St. Louis: 1998. [Google Scholar]
- 342.Heggers J.P., McCoy L., Reisner B., Smith M., Edgar P., Ramirez R.J. Alternate antimicrobial therapy for vancomycin-resistant enterococci burn wound infections. J Burn Care Rehabil. 1998;19:399–403. doi: 10.1097/00004630-199809000-00007. [DOI] [PubMed] [Google Scholar]
- 343.Sheridan R.L., Weber J., Benjamin J., Pasternack M.S., Tompkins R.G. Control of methicillin-resistant Staphylococcus aureus in a pediatric burn unit. Am J Infect Control. 1994;22:340–345. doi: 10.1016/0196-6553(94)90032-9. [DOI] [PubMed] [Google Scholar]
- 344.Matsumura H., Yoshizawa N., Narumi A., Harunari N., Sugamata A., Watanabe K. Effective control of methicillin-resistant Staphylococcus aureus in a burn unit. Burns. 1996;22:283–286. doi: 10.1016/0305-4179(95)00145-x. [DOI] [PubMed] [Google Scholar]
- 345.McGregor J.C. Profile of the first four years of the Regional Burn Unit based at St. John's Hospital, West Lothian (1992–1996) J R Coll Surg Edinb. 1998;43:45–48. [PubMed] [Google Scholar]
- 346.Desai M.H., Rutan R.L., Heggers J.P., Herndon D.N. Candida infection with and without nystatin prophylaxis: an 11-year experience with patients with burn injury. Arch Surg. 1992;127:159–162. doi: 10.1001/archsurg.1992.01420020041006. [DOI] [PubMed] [Google Scholar]
- 347.Ekenna O., Sherertz R.J., Bingham H. Natural history of bloodstream infections in a burn patient population: the importance of candidemia. Am J Infect Control. 1993;21:189–195. doi: 10.1016/0196-6553(93)90030-8. [DOI] [PubMed] [Google Scholar]
- 348.Bowser-Wallace B.H., Graves D.B., Caldwell F.T. An epidemiological profile and trend analysis of wound flora in burned children: 7 years' experience. Burns Incl Therm Inj. 1984;11:16–25. doi: 10.1016/0305-4179(84)90156-6. [DOI] [PubMed] [Google Scholar]
- 349.Tredget E.E., Shankowsky H.A., Rennie R., Burrell R.E., Logsetty S. Pseudomonas infections in the thermally injured patient. Burns. 2004;30:3–26. doi: 10.1016/j.burns.2003.08.007. [DOI] [PubMed] [Google Scholar]
- 350.Edgar P., Mlcak R., Desai M., Linares H.A., Phillips L.G., Heggers J.P. Containment of a multiresistant Serratia marcescens outbreak. Burns. 1997;23:15–18. doi: 10.1016/s0305-4179(97)81117-5. [DOI] [PubMed] [Google Scholar]
- 351.Embil J.M., McLeod J.A., Al-Barrak A.M. An outbreak of methicillin-resistant Staphylococcus aureus on a burn unit: potential role of contaminated hydrotherapy equipment. Burns. 2001;27:681–688. doi: 10.1016/s0305-4179(01)00045-6. [DOI] [PubMed] [Google Scholar]
- 352.Meier P.A., Carter C.D., Wallace S.E., Hollis R.J., Pfaller M.A., Herwaldt L.A. A prolonged outbreak of methicillin-resistant Staphylococcus aureus in the burn unit of a tertiary medical center. Infect Control Hosp Epidemiol. 1996;17:798–802. doi: 10.1086/647239. [DOI] [PubMed] [Google Scholar]
- 353.Snyder L.L., Wiebelhaus P., Boon S.E., Morin R.A., Goering R. Methicillin-resistant Staphylococcus aureus eradication in a burn center. J Burn Care Rehabil. 1993;14(2 Pt 1):164–168. doi: 10.1097/00004630-199303000-00007. [DOI] [PubMed] [Google Scholar]
- 354.May A.K., Melton S.M., McGwin G., Cross J.M., Moser S.A., Rue L.W. Reduction of vancomycin-resistant enterococcal infections by limitation of broad-spectrum cephalosporin use in a trauma and burn intensive care unit. Shock. 2000;14:259–264. doi: 10.1097/00024382-200014030-00003. [DOI] [PubMed] [Google Scholar]
- 355.Sheridan R.L., Weber J.M., Budkevich L.G., Tompkins R.G. Candidemia in the pediatric patient with burns. J Burn Care Rehabil. 1995;16:440–443. doi: 10.1097/00004630-199507000-00011. [DOI] [PubMed] [Google Scholar]
- 356.Mayhall C.G. The epidemiology of burn wound infections: then and now. Clin Infect Dis. 2003;37:543–550. doi: 10.1086/376993. [DOI] [PubMed] [Google Scholar]
- 357.McManus A.T., Mason A.D., Jr., McManus W.F., Pruitt B.A., Jr. A decade of reduced Gram-negative infections and mortality associated with improved isolation of burned patients. Arch Surg. 1994;129:1306–1309. doi: 10.1001/archsurg.1994.01420360096013. [DOI] [PubMed] [Google Scholar]
- 358.Bryce E.A., Walker M., Scharf S. An outbreak of cutaneous aspergillosis in a tertiary care hospital. Infect Control Hosp Epidemiol. 1996;17:170–172. doi: 10.1086/647266. [DOI] [PubMed] [Google Scholar]
- 359.Levenson C., Wohlford P., Djou J., Evans S., Zawacki B. Preventing postoperative burn wound aspergillosis. J Burn Care Rehabil. 1991;12:132–135. doi: 10.1097/00004630-199103000-00008. [DOI] [PubMed] [Google Scholar]
- 360.Tredget E.E., Shankowsky H.A., Joffe A.M. Epidemiology of infections with Pseudomonas aeruginosa in burn patients: the role of hydrotherapy. Clin Infect Dis. 1992;15:941–949. doi: 10.1093/clind/15.6.941. [DOI] [PubMed] [Google Scholar]
- 361.Wisplinghoff H., Perbix W., Seifert H. Risk factors for nosocomial bloodstream infections due to Acinetobacter baumannii: a case-control study of adult burn patients. Clin Infect Dis. 1999;28:59–66. doi: 10.1086/515067. [DOI] [PubMed] [Google Scholar]
- 362.Weber J.M., Sheridan R.L., Schulz J.T., Tompkins R.G., Ryan C.M. Effectiveness of bacteria-controlled nursing units in preventing cross-colonization with resistant bacteria in severely burned children. Infect Control Hosp Epidemiol. 2002;23:549–551. doi: 10.1086/502106. [DOI] [PubMed] [Google Scholar]
- 363.Bayat A., Shaaban H., Dodgson A., Dunn K.W. Implications for burn unit design following outbreak of multi-resistant Acinetobacter infection in an ICU and burn unit. Burns. 2003;29:303–306. doi: 10.1016/s0305-4179(03)00011-1. [DOI] [PubMed] [Google Scholar]
- 364.Lee J.J., Marvin J.A., Heimbach D.M., Grube B.J., Engrav L.H. Infection control in a burn center. J Burn Care Rehabil. 1990;11:575–580. doi: 10.1097/00004630-199011000-00018. [DOI] [PubMed] [Google Scholar]
- 365.Raymond J., Aujard Y. Nosocomial infections in pediatric patients: a European, multicenter prospective study. European Study Group. Infect Control Hosp Epidemiol. 2000;21:260–263. doi: 10.1086/501755. [DOI] [PubMed] [Google Scholar]
- 366.Campins M., Vaque J., Rossello J. Nosocomial infections in pediatric patients: a prevalence study in Spanish hospitals. EPINE Working Group. Am J Infect Control. 1993;21:58–63. doi: 10.1016/0196-6553(93)90225-s. [DOI] [PubMed] [Google Scholar]
- 367.Allen U., Ford-Jones E.L. Nosocomial infections in the pediatric patient: an update. Am J Infect Control. 1990;18:176–193. doi: 10.1016/0196-6553(90)90183-s. [DOI] [PubMed] [Google Scholar]
- 368.Grohskopf L.A., Sinkowitz-Cochran R.L., Garrett D.O. A national point-prevalence survey of pediatric intensive care unit–acquired infections in the United States. J Pediatr. 2002;140:432–438. doi: 10.1067/mpd.2002.122499. [DOI] [PubMed] [Google Scholar]
- 369.Sohn A.H., Garrett D.O., Sinkowitz-Cochran R.L. Prevalence of nosocomial infections in neonatal intensive care unit patients: results from the first national point-prevalence survey. J Pediatr. 2001;139:821–827. doi: 10.1067/mpd.2001.119442. [DOI] [PubMed] [Google Scholar]
- 370.Gaynes R.P., Edwards J.R., Jarvis W.R., Culver D.H., Tolson J.S., Martone W.J. Nosocomial infections among neonates in high-risk nurseries in the United States. National Nosocomial Infections Surveillance System. Pediatrics. 1996;98(3 Pt 1):357–361. [PubMed] [Google Scholar]
- 371.Richards M.J., Edwards J.R., Culver D.H., Gaynes R.P. Nosocomial infections in pediatric intensive care units in the United States. National Nosocomial Infections Surveillance System. Pediatrics. 1999;103:e39. doi: 10.1542/peds.103.4.e39. [DOI] [PubMed] [Google Scholar]
- 372.Maltezou H.C., Drancourt M. Nosocomial influenza in children. J Hosp Infect. 2003;55:83–91. doi: 10.1016/s0195-6701(03)00262-7. [DOI] [PubMed] [Google Scholar]
- 373.Moisiuk S.E., Robson D., Klass L. Outbreak of parainfluenza virus type 3 in an intermediate care neonatal nursery. Pediatr Infect Dis J. 1998;17:49–53. doi: 10.1097/00006454-199801000-00011. [DOI] [PubMed] [Google Scholar]
- 374.Mullins J.A., Erdman D.D., Weinberg G.A. Human metapneumovirus infection among children hospitalized with acute respiratory illness. Emerg Infect Dis. 2004;10:700–705. doi: 10.3201/eid1004.030555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Hatherill M., Levin M., Lawrenson J., Hsiao N.Y., Reynolds L., Argent A. Evolution of an adenovirus outbreak in a multidisciplinary children's hospital. J Paediatr Child Health. 2004;40:449–454. doi: 10.1111/j.1440-1754.2004.00426.x. [DOI] [PubMed] [Google Scholar]
- 376.Langley J.M., Hanakowski M. Variation in risk for nosocomial chickenpox after inadvertent exposure. J Hosp Infect. 2000;44:224–226. doi: 10.1053/jhin.1999.0664. [DOI] [PubMed] [Google Scholar]
- 377.Ratner A.J., Neu N., Jakob K. Nosocomial rotavirus in a pediatric hospital. Infect Control Hosp Epidemiol. 2001;22:299–301. doi: 10.1086/501904. [DOI] [PubMed] [Google Scholar]
- 378.Avila-Aguero M.L., German G., Paris M.M., Herrera J.F. Toys in a pediatric hospital: are they a bacterial source? Am J Infect Control. 2004;32:287–290. doi: 10.1016/j.ajic.2003.10.018. [DOI] [PubMed] [Google Scholar]
- 379.Nyqvist K.H., Lutes L.M. Co-bedding twins: a developmentally supportive care strategy. J Obstet Gynecol Neonatal Nurs. 1998;27:450–456. doi: 10.1111/j.1552-6909.1998.tb02669.x. [DOI] [PubMed] [Google Scholar]
- 380.Feldman R., Eidelman A.I., Sirota L., Weller A. Comparison of skin-to-skin (kangaroo) and traditional care: parenting outcomes and preterm infant development. Pediatrics. 2002;110(1 Pt 1):16–26. doi: 10.1542/peds.110.1.16. [DOI] [PubMed] [Google Scholar]
- 381.Conde-Agudelo A., Diaz-Rossello J.L., Belizan J.M. Kangaroo mother care to reduce morbidity and mortality in low birthweight infants. Cochrane Database Syst Rev. 2003 doi: 10.1002/14651858.CD002771. CD002771. [DOI] [PubMed] [Google Scholar]
- 382.Adcock P.M., Pastor P., Medley F., Patterson J.E., Murphy T.V. Methicillin-resistant Staphylococcus aureus in two child care centers. J Infect Dis. 1998;178:577–580. doi: 10.1086/517478. [DOI] [PubMed] [Google Scholar]
- 383.Shahin R., Johnson I.L., Jamieson F., McGeer A., Tolkin J., Ford-Jones E.L. Methicillin-resistant Staphylococcus aureus carriage in a child care center following a case of disease. Toronto Child Care Center Study Group. Arch Pediatr Adolesc Med. 1999;153:864–868. doi: 10.1001/archpedi.153.8.864. [DOI] [PubMed] [Google Scholar]
- 384.Stover B.H., Duff A., Adams G., Buck G., Hancock G., Rabalais G. Emergence and control of methicillin-resistant Staphylococcus aureus in a children's hospital and pediatric long-term care facility. Am J Infect Control. 1992;20:248–255. doi: 10.1016/s0196-6553(05)80198-1. [DOI] [PubMed] [Google Scholar]
- 385.Herold B.C., Immergluck L.C., Maranan M.C. Community-acquired methicillin-resistant Staphylococcus aureus in children with no identified predisposing risk. JAMA. 1998;279:593–598. doi: 10.1001/jama.279.8.593. [DOI] [PubMed] [Google Scholar]
- 386.Centers for Disease Control and Prevention Four pediatric deaths from community-acquired methicillin-resistant Staphylococcus aureus, Minnesota and North Dakota, 1997–1999. MMWR Morb Mortal Wkly Rep. 1999;48:707–710. [PubMed] [Google Scholar]
- 387.Abi-Hanna P., Frank A.L., Quinn J.P. Clonal features of community-acquired methicillin-resistant Staphylococcus aureus in children. Clin Infect Dis. 2000;30:630–631. doi: 10.1086/313706. [DOI] [PubMed] [Google Scholar]
- 388.Fergie J.E., Purcell K. Community-acquired methicillin-resistant Staphylococcus aureus infections in south Texas children. Pediatr Infect Dis J. 2001;20:860–863. doi: 10.1097/00006454-200109000-00007. [DOI] [PubMed] [Google Scholar]
- 389.Sattler C.A., Mason E.O., Jr., Kaplan S.L. Prospective comparison of risk factors and demographic and clinical characteristics of community-acquired, methicillin-resistant versus methicillin-susceptible Staphylococcus aureus infection in children. Pediatr Infect Dis J. 2002;21:910–917. doi: 10.1097/00006454-200210000-00005. [DOI] [PubMed] [Google Scholar]
- 390.Kaplan S.L., Hulten K.G., Gonzalez B.E. Three-year surveillance of community-acquired Staphylococcus aureus infections in children. Clin Infect Dis. 2005;40:1785–1791. doi: 10.1086/430312. [DOI] [PubMed] [Google Scholar]
- 391.Jarvis W.R. Infection control and changing health-care delivery systems. Emerg Infect Dis. 2001;7:170–173. doi: 10.3201/eid0702.010202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Garibaldi R.A. Residential care and the elderly: the burden of infection. J Hosp Infect. 1999;43(Suppl):S9–S18. doi: 10.1016/s0195-6701(99)90061-0. [DOI] [PubMed] [Google Scholar]
- 393.Hsu K., Harris J.A. Control of infections in nonacute care pediatric settings. Semin Pediatr Infect Dis. 2001;12:92–99. [Google Scholar]
- 394.Strausbaugh L.J., Joseph C.L. The burden of infection in long-term care. Infect Control Hosp Epidemiol. 2000;21:674–679. doi: 10.1086/501712. [DOI] [PubMed] [Google Scholar]
- 395.Centers for Medicare and Medicaid Services. Healthcare industry market update. Available from http://wwwcmshhsgov/reports/hcimu/hcimu_05202003pdf, May 2003.
- 396.Lee Y.L., Thrupp L.D., Friis R.H., Fine M., Maleki P., Cesario T.C. Nosocomial infection and antibiotic utilization in geriatric patients: a pilot prospective surveillance program in skilled nursing facilities. Gerontology. 1992;38:223–232. doi: 10.1159/000213332. [DOI] [PubMed] [Google Scholar]
- 397.Stevenson K.B. Regional data set of infection rates for long-term care facilities: description of a valuable benchmarking tool. Am J Infect Control. 1999;27:20–26. doi: 10.1016/s0196-6553(99)70070-2. [DOI] [PubMed] [Google Scholar]
- 398.Jackson M.M., Fierer J., Barrett-Connor E. Intensive surveillance for infections in a three-year study of nursing home patients. Am J Epidemiol. 1992;135:685–696. doi: 10.1093/oxfordjournals.aje.a116348. [DOI] [PubMed] [Google Scholar]
- 399.Darnowski S.B., Gordon M., Simor A.E. Two years of infection surveillance in a geriatric long-term care facility. Am J Infect Control. 1991;19:185–190. doi: 10.1016/0196-6553(91)90002-t. [DOI] [PubMed] [Google Scholar]
- 400.Hoffman N., Jenkins R., Putney K. Nosocomial infection rates during a one-year period in a nursing home care unit of a Veterans Administration hospital. Am J Infect Control. 1990;18:55–63. doi: 10.1016/0196-6553(90)90082-4. [DOI] [PubMed] [Google Scholar]
- 401.Tsan L., Hojlo C., Kearns M.A. Infection surveillance and control programs in the Department of Veterans Affairs nursing home care units: a preliminary assessment. Am J Infect Control. 2006;34:80–83. doi: 10.1016/j.ajic.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 402.Kane R.A., Caplan A.L., Urv-Wong E.K., Freeman I.C., Aroskar M.A., Finch M. Everyday matters in the lives of nursing home residents: wish for and perception of choice and control. J Am Geriatr Soc. 1997;45:1086–1093. doi: 10.1111/j.1532-5415.1997.tb05971.x. [DOI] [PubMed] [Google Scholar]
- 403.Libow L.S., Starer P. Care of the nursing home patient. N Engl J Med. 1989;321:93–96. doi: 10.1056/NEJM198907133210206. [DOI] [PubMed] [Google Scholar]
- 404.Perls T.T., Herget M. Higher respiratory infection rates on an Alzheimer's special care unit and successful intervention. J Am Geriatr Soc. 1995;43:1341–1344. doi: 10.1111/j.1532-5415.1995.tb06611.x. [DOI] [PubMed] [Google Scholar]
- 405.Bradley S.F. Issues in the management of resistant bacteria in long-term-care facilities. Infect Control Hosp Epidemiol. 1999;20:362–366. doi: 10.1086/501637. [DOI] [PubMed] [Google Scholar]
- 406.Crossley K. Vancomycin-resistant enterococci in long-term-care facilities. Infect Control Hosp Epidemiol. 1998;19:521–525. doi: 10.1086/647864. [DOI] [PubMed] [Google Scholar]
- 407.Strausbaugh L.J., Crossley K.B., Nurse B.A., Thrupp L.D. Antimicrobial resistance in long-term-care facilities. Infect Control Hosp Epidemiol. 1996;17:129–140. doi: 10.1086/647257. [DOI] [PubMed] [Google Scholar]
- 408.Richards C.L. Infections in Long-Term-Care Facilities: Screen or Clean? Infect Control Hosp Epidemiol. 2005;26:800–801. doi: 10.1086/502496. [DOI] [PubMed] [Google Scholar]
- 409.Drinka P.J., Krause P., Nest L., Goodman B.M., Gravenstein S. Risk of acquiring influenza A in a nursing home from a culture-positive roommate. Infect Control Hosp Epidemiol. 2003;24:872–874. doi: 10.1086/502153. [DOI] [PubMed] [Google Scholar]
- 410.Falsey A.R., Treanor J.J., Betts R.F., Walsh E.E. Viral respiratory infections in the institutionalized elderly: clinical and epidemiologic findings. J Am Geriatr Soc. 1992;40:115–119. doi: 10.1111/j.1532-5415.1992.tb01929.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Ellis S.E., Coffey C.S., Mitchel E.F., Jr., Dittus R.S., Griffin M.R. Influenza- and respiratory syncytial virus–associated morbidity and mortality in the nursing home population. J Am Geriatr Soc. 2003;51:761–767. doi: 10.1046/j.1365-2389.2003.51254.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Louie J.K., Yagi S., Nelson F.A. Rhinovirus outbreak in a long-term care facility for elderly persons associated with unusually high mortality. Clin Infect Dis. 2005;41:262–265. doi: 10.1086/430915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.Piednoir E., Bureau-Chalot F., Merle C., Gotzamanis A., Wuibout J., Bajolet O. Direct costs associated with a nosocomial outbreak of adenoviral conjunctivitis infection in a long-term care institution. Am J Infect Control. 2002;30:407–410. doi: 10.1067/mic.2002.125193. [DOI] [PubMed] [Google Scholar]
- 414.Addiss D.G., Davis J.P., Meade B.D. A pertussis outbreak in a Wisconsin nursing home. J Infect Dis. 1991;164:704–710. doi: 10.1093/infdis/164.4.704. [DOI] [PubMed] [Google Scholar]
- 415.Gaynes R., Rimland D., Killum E. Outbreak of Clostridium difficile infection in a long-term care facility: association with gatifloxacin use. Clin Infect Dis. 2004;38:640–645. doi: 10.1086/381551. [DOI] [PubMed] [Google Scholar]
- 416.High K.P., Bradley S., Loeb M., Palmer R., Quagliarello V., Yoshikawa T. A new paradigm for clinical investigation of infectious syndromes in older adults: assessment of functional status as a risk factor and outcome measure. Clin Infect Dis. 2005;40:114–122. doi: 10.1086/426082. [DOI] [PubMed] [Google Scholar]
- 417.Loeb M.B., Craven S., McGeer A.J. Risk factors for resistance to antimicrobial agents among nursing home residents. Am J Epidemiol. 2003;157:40–47. doi: 10.1093/aje/kwf173. [DOI] [PubMed] [Google Scholar]
- 418.Vergis E.N., Brennen C., Wagener M., Muder R.R. Pneumonia in long-term care: a prospective case-control study of risk factors and impact on survival. Arch Intern Med. 2001;161:2378–2381. doi: 10.1001/archinte.161.19.2378. [DOI] [PubMed] [Google Scholar]
- 419.Loeb M., McGeer A., McArthur M., Walter S., Simor A.E. Risk factors for pneumonia and other lower respiratory tract infections in elderly residents of long-term care facilities. Arch Intern Med. 1999;159:2058–2064. doi: 10.1001/archinte.159.17.2058. [DOI] [PubMed] [Google Scholar]
- 420.Brandeis G.H., Ooi W.L., Hossain M., Morris J.N., Lipsitz L.A. A longitudinal study of risk factors associated with the formation of pressure ulcers in nursing homes. J Am Geriatr Soc. 1994;42:388–393. doi: 10.1111/j.1532-5415.1994.tb07486.x. [DOI] [PubMed] [Google Scholar]
- 421.Allard J.P., Aghdassi E., McArthur M. Nutrition risk factors for survival in the elderly living in Canadian long-term care facilities. J Am Geriatr Soc. 2004;52:59–65. doi: 10.1111/j.1532-5415.2004.52011.x. [DOI] [PubMed] [Google Scholar]
- 422.Pick N., McDonald A., Bennett N. Pulmonary aspiration in a long-term care setting: clinical and laboratory observations and an analysis of risk factors. J Am Geriatr Soc. 1996;44:763–768. doi: 10.1111/j.1532-5415.1996.tb03731.x. [DOI] [PubMed] [Google Scholar]
- 423.Nicolle L.E. The chronic indwelling catheter and urinary infection in long-term care facility residents. Infect Control Hosp Epidemiol. 2001;22:316–321. doi: 10.1086/501908. [DOI] [PubMed] [Google Scholar]
- 424.Pien E.C., Hume K.E., Pien F.D. Gastrostomy tube infections in a community hospital. Am J Infect Control. 1996;24:353–358. doi: 10.1016/s0196-6553(96)90022-x. [DOI] [PubMed] [Google Scholar]
- 425.Gomes G.F., Pisani J.C., Macedo E.D., Campos A.C. The nasogastric feeding tube as a risk factor for aspiration and aspiration pneumonia. Curr Opin Clin Nutr Metab Care. 2003;6:327–333. doi: 10.1097/01.mco.0000068970.34812.8b. [DOI] [PubMed] [Google Scholar]
- 426.Bula C.J., Ghilardi G., Wietlisbach V., Petignat C., Francioli P. Infections and functional impairment in nursing home residents: a reciprocal relationship. J Am Geriatr Soc. 2004;52:700–706. doi: 10.1111/j.1532-5415.2004.52205.x. [DOI] [PubMed] [Google Scholar]
- 427.Bradley S.F., Terpenning M.S., Ramsey M.A. Methicillin-resistant Staphylococcus aureus: colonization and infection in a long-term care facility. Ann Intern Med. 1991;115:417–422. doi: 10.7326/0003-4819-115-6-417. [DOI] [PubMed] [Google Scholar]
- 428.Washio M., Nishisaka S., Kishikawa K. Incidence of methicillin-resistant Staphylococcus aureus (MRSA) isolation in a skilled nursing home: a third report on the risk factors for the occurrence of MRSA infection in the elderly. J Epidemiol. 1996;6:69–73. doi: 10.2188/jea.6.69. [DOI] [PubMed] [Google Scholar]
- 429.Trick W.E., Weinstein R.A., DeMarais P.L. Colonization of skilled-care facility residents with antimicrobial-resistant pathogens. J Am Geriatr Soc. 2001;49:270–276. doi: 10.1046/j.1532-5415.2001.4930270.x. [DOI] [PubMed] [Google Scholar]
- 430.Nicolle L.E., Garibaldi R.A. Infection control in long-term care facilities. Infect Control Hosp Epidemiol. 1995;16:348–353. doi: 10.1086/647124. [DOI] [PubMed] [Google Scholar]
- 431.Crossley K. Long-term care facilities as sources of antibiotic-resistant nosocomial pathogens. Curr Opin Infect Dis. 2001;14:455–459. doi: 10.1097/00001432-200108000-00010. [DOI] [PubMed] [Google Scholar]
- 432.Smith P.W., Rusnak P.G. Infection prevention and control in the long-term-care facility. SHEA Long-Term-Care Committee and APIC Guidelines Committee. Infect Control Hosp Epidemiol. 1997;18:831–849. [PubMed] [Google Scholar]
- 433.Friedman C., Barnette M., Buck A.S. Requirements for infrastructure and essential activities of infection control and epidemiology in out-of-hospital settings: a consensus panel report. Association for Professionals in Infection Control and Epidemiology and Society for Healthcare Epidemiology of America. Infect Control Hosp Epidemiol. 1999;20:695–705. doi: 10.1086/501569. [DOI] [PubMed] [Google Scholar]
- 434.Nicolle L.E. Infection control in long-term care facilities. Clin Infect Dis. 2000;31:752–756. doi: 10.1086/314010. [DOI] [PubMed] [Google Scholar]
- 435.Bradley S.F. Methicillin-resistant Staphylococcus aureus: long-term care concerns. Am J Med. 1999;106:2S–10S. doi: 10.1016/s0002-9343(98)00349-0. [DOI] [PubMed] [Google Scholar]
- 436.www.cms.hhs.gov/manuals/Downloads/som107ap_pp_guidelines_ltcf.
- 437.Mylotte J.M., Goodnough S., Tayara A. Antibiotic-resistant organisms among long-term care facility residents on admission to an inpatient geriatrics unit: retrospective and prospective surveillance. Am J Infect Control. 2001;29:139–144. doi: 10.1067/mic.2001.114225. [DOI] [PubMed] [Google Scholar]
- 438.Strausbaugh L.J., Jacobson C., Yost T. Methicillin-resistant Staphylococcus aureus in a nursing home and affiliated hospital: a four-year perspective. Infect Control Hosp Epidemiol. 1993;14:331–336. doi: 10.1086/646752. [DOI] [PubMed] [Google Scholar]
- 439.Wiener J., Quinn J.P., Bradford P.A. Multiple antibiotic-resistant Klebsiella and Escherichia coli in nursing homes. JAMA. 1999;281:517–523. doi: 10.1001/jama.281.6.517. [DOI] [PubMed] [Google Scholar]
- 440.Pop-Vicas A.E., D'Agata E.M. The rising influx of multidrug-resistant Gram-negative bacilli in tertiary care hospitals. Clin Infect Dis. 2005;40:1792–1798. doi: 10.1086/430314. [DOI] [PubMed] [Google Scholar]
- 441.Ly N., McCaig L.F. National Hospital Ambulatory Medical Care Survey: 2000 outpatient department summary. Adv Data. 2002:1–27. [PubMed] [Google Scholar]
- 442.Cherry D.K., Woodwell D.A. National Ambulatory Medical Care Survey: 2000 summary. Adv Data. 2002:1–32. [PubMed] [Google Scholar]
- 443.Finelli L., Miller J.T., Tokars J.I., Alter M.J., Arduino M.J. National surveillance of dialysis-associated diseases in the United States. Semin Dial. 2002;2005(18):52–61. doi: 10.1111/j.1525-139X.2005.18108.x. [DOI] [PubMed] [Google Scholar]
- 444.D'Agata E.M. Antimicrobial-resistant, Gram-positive bacteria among patients undergoing chronic hemodialysis. Clin Infect Dis. 2002;35:1212–1218. doi: 10.1086/344282. [DOI] [PubMed] [Google Scholar]
- 445.Goodman R.A., Solomon S.L. Transmission of infectious diseases in outpatient health care settings. JAMA. 1991;265:2377–2381. [PubMed] [Google Scholar]
- 446.Nafziger D.A., Lundstrom T., Chandra S., Massanari R.M. Infection control in ambulatory care. Infect Dis Clin North Am. 1997;11:279–296. doi: 10.1016/s0891-5520(05)70356-1. [DOI] [PubMed] [Google Scholar]
- 447.Herwaldt L.A., Smith S.D., Carter C.D. Infection control in the outpatient setting. Infect Control Hosp Epidemiol. 1998;19:41–74. doi: 10.1086/647706. [DOI] [PubMed] [Google Scholar]
- 448.Hlady W.G., Hopkins R.S., Ogilby T.E., Allen S.T. Patient-to-patient transmission of hepatitis B in a dermatology practice. Am J Public Health. 1993;83:1689–1693. doi: 10.2105/ajph.83.12.1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 449.Birnie G.G., Quigley E.M., Clements G.B., Follet E.A., Watkinson G. Endoscopic transmission of hepatitis B virus. Gut. 1983;24:171–174. doi: 10.1136/gut.24.2.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Chant K., Lowe D., Rubin G. Patient-to-patient transmission of HIV in private surgical consulting rooms. Lancet. 1993;342:1548–1549. doi: 10.1016/s0140-6736(05)80112-6. [DOI] [PubMed] [Google Scholar]
- 451.Chant K., Kociuba K., Munro R. Investigation of possible patient-to-patient transmission of hepatitis C in a hospital. NSW Public Health Bull. 1994;5:47–51. [Google Scholar]
- 452.Centers for Disease Control and Prevention Transmission of hepatitis B and C viruses in outpatient settings, New York, Oklahoma, and Nebraska, 2000–2002. MMWR Morb Mortal Wkly Rep. 2003;52:901–906. [PubMed] [Google Scholar]
- 453.Williams I.T., Perz J.F., Bell B.P. Viral hepatitis transmission in ambulatory health care settings. Clin Infect Dis. 2004;38:1592–1598. doi: 10.1086/420935. [DOI] [PubMed] [Google Scholar]
- 454.Couldwell D.L., Dore G.J., Harkness J.L. Nosocomial outbreak of tuberculosis in an outpatient HIV treatment room. AIDS. 1996;10:521–525. doi: 10.1097/00002030-199605000-00011. [DOI] [PubMed] [Google Scholar]
- 455.Centers for Disease Control and Prevention Mycobacterium tuberculosis transmission in a health clinic, Florida. MMWR Morb Mortal Wkly Rep. 1989;38:256–258. [PubMed] [Google Scholar]
- 456.Calder R.A., Duclos P., Wilder M.H., Pryor V.L., Scheel W.J. Mycobacterium tuberculosis transmission in a health clinic. Bull Int Union Tuberc Lung Dis. 1991;66:103–106. [PubMed] [Google Scholar]
- 457.Istre G.R., McKee P.A., West G.R. Measles spread in medical settings: an important focus of disease transmission? Pediatrics. 1987;79:356–358. [PubMed] [Google Scholar]
- 458.Dawson C., Darrell R. Infections due to adenovirus type 8 in the United States. I: an outbreak of epidemic keratoconjunctivitis originating in a physician's office. N Engl J Med. 1963;268:1031–1034. doi: 10.1056/NEJM196305092681901. [DOI] [PubMed] [Google Scholar]
- 459.Montessori V., Scharf S., Holland S., Werker D.H., Roberts F.J., Bryce E. Epidemic keratoconjunctivitis outbreak at a tertiary referral eye care clinic. Am J Infect Control. 1998;26:399–405. doi: 10.1016/s0196-6553(98)70035-5. [DOI] [PubMed] [Google Scholar]
- 460.Jernigan J.A., Lowry B.S., Hayden F.G. Adenovirus type 8 epidemic keratoconjunctivitis in an eye clinic: risk factors and control. J Infect Dis. 1993;167:1307–1313. doi: 10.1093/infdis/167.6.1307. [DOI] [PubMed] [Google Scholar]
- 461.Buehler J.W., Finton R.J., Goodman R.A. Epidemic keratoconjunctivitis: report of an outbreak in an ophthalmology practice and recommendations for prevention. Infect Control. 1984;5:390–394. doi: 10.1017/s0195941700062238. [DOI] [PubMed] [Google Scholar]
- 462.Johnston CP, Cooper L, Ruby W, Teeter T, et al. Community-associated methicillin resistant Staphyloccoccus aureus skin infections among outpatient healthcare workers and its isolation in the clinic environment [abstract 132]. Presented at the 15th Annual Scientific Meeting of the Society for Healthcare Epidemiology of America (SHEA), Los Angeles, CA, April 10, 2005.
- 463.Biddick R., Spilker T., Martin A., LiPuma J.J. Evidence of transmission of Burkholderia cepacia, Burkholderia multivorans, and Burkholderia dolosa among persons with cystic fibrosis. FEMS Microbiol Lett. 2003;228:57–62. doi: 10.1016/S0378-1097(03)00724-9. [DOI] [PubMed] [Google Scholar]
- 464.Griffiths A.L., Jamsen K., Carlin J.B. Effects of segregation on an epidemic Pseudomonas aeruginosa strain in a cystic fibrosis clinic. Am J Respir Crit Care Med. 2005;171:1020–1025. doi: 10.1164/rccm.200409-1194OC. [DOI] [PubMed] [Google Scholar]
- 465.Danzig L.E., Short L.J., Collins K. Bloodstream infections associated with a needleless intravenous infusion system in patients receiving home infusion therapy. JAMA. 1995;273:1862–1864. [PubMed] [Google Scholar]
- 466.Kellerman S., Shay D.K., Howard J. Bloodstream infections in home infusion patients: the influence of race and needleless intravascular access devices. J Pediatr. 1996;129:711–717. doi: 10.1016/s0022-3476(96)70154-3. [DOI] [PubMed] [Google Scholar]
- 467.Do A.N., Ray B.J., Banerjee S.N. Bloodstream infection associated with needleless device use and the importance of infection-control practices in the home health care setting. J Infect Dis. 1999;179:442–448. doi: 10.1086/314592. [DOI] [PubMed] [Google Scholar]
- 468.Tokars J.I., Cookson S.T., McArthur M.A., Boyer C.L., McGeer A.J., Jarvis W.R. Prospective evaluation of risk factors for bloodstream infection in patients receiving home infusion therapy. Ann Intern Med. 1999;131:340–347. doi: 10.7326/0003-4819-131-5-199909070-00004. [DOI] [PubMed] [Google Scholar]
- 469.Manangan L.P., Pearson M.L., Tokars J.I., Miller E., Jarvis W.R. Feasibility of national surveillance of health care–associated infections in home-care settings. Emerg Infect Dis. 2002;8:233–236. doi: 10.3201/eid0803.010098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 470.Shah S.S., Manning M.L., Leahy E., Magnusson M., Rheingold S.R., Bell L.M. Central venous catheter–associated bloodstream infections in pediatric oncology home care. Infect Control Hosp Epidemiol. 2002;23:99–101. doi: 10.1086/502015. [DOI] [PubMed] [Google Scholar]
- 471.Gorski L.A. Central venous access device outcomes in a homecare agency: a 7-year study. J Infus Nurs. 2004;27:104–111. doi: 10.1097/00129804-200403000-00006. [DOI] [PubMed] [Google Scholar]
- 472.Rosenheimer L., Embry F.C., Sanford J., Silver S.R. Infection surveillance in home care: device-related incidence rates. Am J Infect Control. 1998;26:359–363. doi: 10.1016/s0196-6553(98)80018-7. [DOI] [PubMed] [Google Scholar]
- 473.White M.C., Ragland K.E. Surveillance of intravenous catheter–related infections among home care clients. Am J Infect Control. 1994;22:231–235. doi: 10.1016/0196-6553(94)99002-6. [DOI] [PubMed] [Google Scholar]
- 474.Beltrami E.M., McArthur M.A., McGeer A. The nature and frequency of blood contacts among home healthcare workers. Infect Control Hosp Epidemiol. 2000;21:765–770. doi: 10.1086/501730. [DOI] [PubMed] [Google Scholar]
- 475.Embry F.C., Chinnes L.F. Draft definitions for surveillance of infections in home health care. Am J Infect Control. 2000;28:449–453. doi: 10.1067/mic.2000.112150. [DOI] [PubMed] [Google Scholar]
- 476.Fraser T.G., Stosor V., Wang Q., Allen A., Zembower T.R. Vancomycin and home health care. Emerg Infect Dis. 2005;11:1558–1564. doi: 10.3201/eid1110.050336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 477.Carrico R.M., Niner S. Multidrug-resistant organisms—VRE and MRSA: practical home care tips. Home Healthcare Nurse. 2002;20:23–28. doi: 10.1097/00004045-200201000-00006. [DOI] [PubMed] [Google Scholar]
- 478.Friedman M.M., Rhinehart E. Improving infection control in home care: from ritual to science-based practice. Home Healthcare Nurse. 2000;18:99–105. doi: 10.1097/00004045-200002000-00007. [DOI] [PubMed] [Google Scholar]
- 479.Friedman M.M., Rhinehart E. Putting infection control principles into practice in home care. Nurs Clin North Am. 1999;34:463–482. [PubMed] [Google Scholar]
- 480.Davis P.L., Madigan E.A. Evidence-based practice and the home care nurse's bag. Home Healthcare Nurse. 1999;17:295–299. [PubMed] [Google Scholar]
- 481.Sitzman K.L., Pett M.A., Bloswick D.S. An exploratory study of nurse bag use by home visiting nurses. Home Healthcare Nurse. 2002;20:237–243. doi: 10.1097/00004045-200204000-00009. [DOI] [PubMed] [Google Scholar]
- 482.Anderson M.A., Madigan E.A., Helms L.B. Nursing research in home health care: endangered species? Home Care Provid. 2001;6:200–204. doi: 10.1067/mhc.2001.120983. [DOI] [PubMed] [Google Scholar]
- 483.White M.C. Identifying infectious diseases in prisons: surveillance, protection, and intervention. West J Med. 1999;170:177. [PMC free article] [PubMed] [Google Scholar]
- 484.Puisis M. Update on public health in correctional facilities. West J Med. 1998;169:374. [PMC free article] [PubMed] [Google Scholar]
- 485.Levy M.H., Lerwitworapong J. Issues facing TB control (3.1). Tuberculosis in prisons. Scott Med J. 2000;45(5 Suppl):30–33. doi: 10.1177/00369330000450S114. [DOI] [PubMed] [Google Scholar]
- 486.Parece M.S., Herrera G.A., Voigt R.F., Middlekauff S.L., Irwin K.L. STD testing policies and practices in US city and county jails. Sex Transm Dis. 1999;26:431–437. doi: 10.1097/00007435-199909000-00003. [DOI] [PubMed] [Google Scholar]
- 487.Cieslak P.R., Curtis M.B., Coulombier D.M., Hathcock A.L., Bean N.H., Tauxe R.V. Preventable disease in correctional facilities: desmoteric foodborne outbreaks in the United States, 1974–1991. Arch Intern Med. 1996;156:1883–1888. doi: 10.1001/archinte.156.16.1883. [DOI] [PubMed] [Google Scholar]
- 488.Centers for Disease Control and Prevention Public health dispatch: tuberculosis outbreak in a homeless population, Portland, Maine, 2002–2003. MMWR Morb Mortal Wkly Rep. 2003;52:1184. [PubMed] [Google Scholar]
- 489.Centers for Disease Control and Prevention. Public health dispatch: tuberculosis outbreak among homeless persons, King County, Washington, 2002–2003. MMWR Morb Mortal Wkly Rep. 2003;52:1209–1210. [PubMed] [Google Scholar]
- 490.Centers for Disease Control and Prevention. Tuberculosis transmission in a homeless shelter population, New York, 2000–2003. MMWR Morb Mortal Wkly Rep. 2005;54:149–152. [PubMed] [Google Scholar]
- 491.Baillargeon J., Kelley M.F., Leach C.T., Baillargeon G., Pollock B.H. Methicillin-resistant Staphylococcus aureus infection in the Texas prison system. Clin Infect Dis. 2004;38:e92–e95. doi: 10.1086/383146. [DOI] [PubMed] [Google Scholar]
- 492.Young D.M., Harris H.W., Charlebois E.D. An epidemic of methicillin-resistant Staphylococcus aureus soft tissue infections among medically underserved patients. Arch Surg. 2004;139:947–953. doi: 10.1001/archsurg.139.9.947. [DOI] [PubMed] [Google Scholar]
- 493.Pan E.S., Diep B.A., Carleton H.A. Increasing prevalence of methicillin-resistant Staphylococcus aureus infection in California jails. Clin Infect Dis. 2003;37:1384–1388. doi: 10.1086/379019. [DOI] [PubMed] [Google Scholar]
- 494.Centers for Disease Control and Prevention Drug-susceptible tuberculosis outbreak in a state correctional facility housing HIV-infected inmates, South Carolina, 1999–2000. MMWR Morb Mortal Wkly Rep. 2000;49:1041–1044. [PubMed] [Google Scholar]
- 495.Centers for Disease Control and Prevention Methicillin-resistant Staphylococcus aureus skin or soft tissue infections in a state prison, Mississippi, 2000. MMWR Morb Mortal Wkly Rep. 2001;50:919–922. [PubMed] [Google Scholar]
- 496.Mohle-Boetani J.C., Miguelino V., Dewsnup D.H. Tuberculosis outbreak in a housing unit for human immunodeficiency virus–infected patients in a correctional facility: transmission risk factors and effective outbreak control. Clin Infect Dis. 2002;34:668–676. doi: 10.1086/338815. [DOI] [PubMed] [Google Scholar]
- 497.Centers for Disease Control and Prevention Prevention and control of tuberculosis in correctional racilities: recommendations of the Advisory Council for the Elimination of Tuberculosis. MMWR Recomm Rep. 1996;45:1–37. [Google Scholar]
- 498.Whimbey E., Englund J.A., Couch R.B. Community respiratory virus infections in immunocompromised patients with cancer. Am J Med. 1997;102:10–18. doi: 10.1016/S0002-9343(97)80004-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 499.Zambon M., Bull T., Sadler C.J., Goldman J.M., Ward K.N. Molecular epidemiology of two consecutive outbreaks of parainfluenza 3 in a bone marrow transplant unit. J Clin Microbiol. 1998;36:2289–2293. doi: 10.1128/jcm.36.8.2289-2293.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 500.Gamis A.S., Howells W.B., DeSwarte-Wallace J., Feusner J.H., Buckley J.D., Woods W.G. Alpha-hemolytic streptococcal infection during intensive treatment for acute myeloid leukemia: a report from the Children's Cancer Group Study CCG-2891. J Clin Oncol. 2000;18:1845–1855. doi: 10.1200/JCO.2000.18.9.1845. [DOI] [PubMed] [Google Scholar]
- 501.Ek T., Mellander L., Andersson B., Abrahamsson J. Immune reconstitution after childhood acute lymphoblastic leukemia is most severely affected in the high-risk group. Pediatr Blood Cancer. 2005;44:461–468. doi: 10.1002/pbc.20255. [DOI] [PubMed] [Google Scholar]
- 502.Pascual V., Allantaz F., Arce E., Punaro M., Banchereau J. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J Exp Med. 2005;201:1479–1486. doi: 10.1084/jem.20050473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 503.Marchesoni A., Puttini P.S., Gorla R. Cyclosporine in addition to infliximab and methotrexate in refractory rheumatoid arthritis. Clin Exp Rheumatol. 2005;23:916–917. [PubMed] [Google Scholar]
- 504.Isaacs K.L., Lewis J.D., Sandborn W.J., Sands B.E., Targan S.R. State of the art: IBD therapy and clinical trials in IBD. Inflamm Bowel Dis. 2005;11(Suppl 1):S3–S12. doi: 10.1097/01.mib.0000184852.84558.b2. [DOI] [PubMed] [Google Scholar]
- 505.Centers for Disease Control and Prevention Guidelines for preventing opportunitic infections among HIV-infected persons. MMWR Recomm Rep. 2002;51:1–52. [PubMed] [Google Scholar]
- 506.Kusne S., Krystofak S. Infection control issues after solid organ transplantation. In: Bowden R.A., Ljungman P., Paya C.V., editors. Transplant infections. 2nd ed. Lippincott Williams & Wilkins; Philadelphia: 2003. [Google Scholar]
- 507.Anderson D., DeFor T., Burns L. A comparison of related donor peripheral blood and bone marrow transplants: importance of late-onset chronic graft-versus-host disease and infections. Biol Blood Marrow Transplant. 2003;9:52–59. doi: 10.1053/bbmt.2003.50000. [DOI] [PubMed] [Google Scholar]
- 508.Pitchford K.C., Corey M., Highsmith A.K. Pseudomonas species contamination of cystic fibrosis patients' home inhalation equipment. J Pediatr. 1987;111:212–216. doi: 10.1016/s0022-3476(87)80069-0. [DOI] [PubMed] [Google Scholar]
- 509.Hamill R.J., Houston E.D., Georghiou P.R. An outbreak of Burkholderia (formerly Pseudomonas) cepacia respiratory tract colonization and infection associated with nebulized albuterol therapy. Ann Intern Med. 1995;122:762–766. doi: 10.7326/0003-4819-122-10-199505150-00005. [DOI] [PubMed] [Google Scholar]
- 510.Hutchinson G.R., Parker S., Pryor J.A. Home-use nebulizers: a potential primary source of Burkholderia cepacia and other colistin-resistant, Gram-negative bacteria in patients with cystic fibrosis. J Clin Microbiol. 1996;34:584–587. doi: 10.1128/jcm.34.3.584-587.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Jakobsson B.M., Onnered A.B., Hjelte L., Nystrom B. Low bacterial contamination of nebulizers in home treatment of cystic fibrosis patients. J Hosp Infect. 1997;36:201–207. doi: 10.1016/s0195-6701(97)90195-x. [DOI] [PubMed] [Google Scholar]
- 512.Rosenfeld M., Joy P., Nguyen C.D., Krzewinski J., Burns J.L. Cleaning home nebulizers used by patients with cystic fibrosis: is rinsing with tap water enough? J Hosp Infect. 2001;49:229–230. doi: 10.1053/jhin.2001.1083. [DOI] [PubMed] [Google Scholar]
- 513.Govan J.R. Infection control in cystic fibrosis: methicillin-resistant Staphylococcus aureus, Pseudomonas aeruginosa and the Burkholderia cepacia complex. J R Soc Med. 2000;93(Suppl 38):40–45. [PMC free article] [PubMed] [Google Scholar]
- 514.Frederiksen B., Koch C., Hoiby N. Changing epidemiology of Pseudomonas aeruginosa infection in Danish cystic fibrosis patients (1974–1995) Pediatr Pulmonol. 1999;28:159–166. doi: 10.1002/(sici)1099-0496(199909)28:3<159::aid-ppul1>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 515.Isles A., Maclusky I., Corey M. Pseudomonas cepacia infection in cystic fibrosis: an emerging problem. J Pediatr. 1984;104:206–210. doi: 10.1016/s0022-3476(84)80993-2. [DOI] [PubMed] [Google Scholar]
- 516.LiPuma J.J. Burkholderia cepacia: Management issues and new insights. Clin Chest Med. 1998;19:473–486. doi: 10.1016/s0272-5231(05)70094-0. [DOI] [PubMed] [Google Scholar]
- 517.Tablan O.C., Chorba T.L., Schidlow D.V. Pseudomonas cepacia colonization in patients with cystic fibrosis: risk factors and clinical outcome. J Pediatr. 1985;107:382–387. doi: 10.1016/s0022-3476(85)80511-4. [DOI] [PubMed] [Google Scholar]
- 518.Hudson V.L., Wielinski C.L., Regelmann W.E. Prognostic implications of initial oropharyngeal bacterial flora in patients with cystic fibrosis diagnosed before the age of two years. J Pediatr. 1993;122:854–860. doi: 10.1016/s0022-3476(09)90007-5. [DOI] [PubMed] [Google Scholar]
- 519.Farrell P.M., Li Z., Kosorok M.R. Bronchopulmonary disease in children with cystic fibrosis after early or delayed diagnosis. Am J Respir Crit Care Med. 2003;168:1100–1108. doi: 10.1164/rccm.200303-434OC. [DOI] [PubMed] [Google Scholar]
- 520.Smith D.L., Gumery L.B., Smith E.G., Stableforth D.E., Kaufmann M.E., Pitt T.L. Epidemic of Pseudomonas cepacia in an adult cystic fibrosis unit: evidence of person-to-person transmission. J Clin Microbiol. 1993;31:3017–3022. doi: 10.1128/jcm.31.11.3017-3022.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.Pegues D.A., Schidlow D.V., Tablan O.C., Carson L.A., Clark N.C., Jarvis W.R. Possible nosocomial transmission of Pseudomonas cepacia in patients with cystic fibrosis. Arch Pediatr Adolesc Med. 1994;148:805–812. doi: 10.1001/archpedi.1994.02170080035006. [DOI] [PubMed] [Google Scholar]
- 522.Govan J.R., Brown P.H., Maddison J. Evidence for transmission of Pseudomonas cepacia by social contact in cystic fibrosis. Lancet. 1993;342:15–19. doi: 10.1016/0140-6736(93)91881-l. [DOI] [PubMed] [Google Scholar]
- 523.Pegues D.A., Carson L.A., Tablan O.C. Acquisition of Pseudomonas cepacia at summer camps for patients with cystic fibrosis. Summer Camp Study Group. J Pediatr. 1994;124(5 Pt 1):694–702. doi: 10.1016/s0022-3476(05)81357-5. [DOI] [PubMed] [Google Scholar]
- 524.Tablan O.C., Martone W.J., Doershuk C.F. Colonization of the respiratory tract with Pseudomonas cepacia in cystic fibrosis. risk factors and outcomes. Chest. 1987;91:527–532. doi: 10.1378/chest.91.4.527. [DOI] [PubMed] [Google Scholar]
- 525.Thomassen M.J., Demko C.A., Doershuk C.F., Stern R.C., Klinger J.D. Pseudomonas cepacia: decrease in colonization in patients with cystic fibrosis. Am Rev Respir Dis. 1986;134:669–671. doi: 10.1164/arrd.1986.134.4.669. [DOI] [PubMed] [Google Scholar]
- 526.Weber D.J., Rutala W.A. Gene therapy: a new challenge for infection control. Infect Control Hosp Epidemiol. 1999;20:530–532. doi: 10.1086/501664. [DOI] [PubMed] [Google Scholar]
- 527.Evans M.E., Lesnaw J.A. Infection control for gene therapy: a busy physician's primer. Clin Infect Dis. 2002;35:597–605. doi: 10.1086/342194. [DOI] [PubMed] [Google Scholar]
- 528.Strausbaugh L.J. Gene therapy and infection control: more light on the way. Infect Control Hosp Epidemiol. 2000;21:630–632. doi: 10.1086/501714. [DOI] [PubMed] [Google Scholar]
- 529.Centers for Disease Control and Prevention West Nile virus infections in organ transplant recipients, New York and Pennsylvania, August–September, 2005. MMWR Morb Mortal Wkly Rep. 2005;54:1021–1023. [PubMed] [Google Scholar]
- 530.Lawson C.A. Cytomegalovirus after kidney transplantation: a case review. Prog Transplant. 2005;15:157–160. doi: 10.1177/152692480501500208. [DOI] [PubMed] [Google Scholar]
- 531.Tugwell B.D., Patel P.R., Williams I.T. Transmission of hepatitis C virus to several organ and tissue recipients from an antibody-negative donor. Ann Intern Med. 2005;143:648–654. doi: 10.7326/0003-4819-143-9-200511010-00008. [DOI] [PubMed] [Google Scholar]
- 532.Kainer M.A., Linden J.V., Whaley D.N. Clostridium infections associated with musculoskeletal tissue allografts. N Engl J Med. 2004;350:2564–2571. doi: 10.1056/NEJMoa023222. [DOI] [PubMed] [Google Scholar]
- 533.Centers for Disease Control and Prevention Invasive Streptococcus pyogenes after allograft implantation, Colorado, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:1174–1176. [PubMed] [Google Scholar]
- 534.Mungai M., Tegtmeier G., Chamberland M., Parise M. Transfusion-transmitted malaria in the United States from 1963 through 1999. N Engl J Med. 2001;344:1973–1978. doi: 10.1056/NEJM200106283442603. [DOI] [PubMed] [Google Scholar]
- 535.Lux J.Z., Weiss D., Linden J.V. Transfusion-associated babesiosis after heart transplant. Emerg Infect Dis. 2003;9:116–119. doi: 10.3201/eid0901.020149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 536.Centers for Disease Control and Prevention Chagas disease after organ transplantation, United States, 2001. MMWR Morb Mortal Wkly Rep. 2002;51:210–212. [PubMed] [Google Scholar]
- 537.Centers for Disease Control and Prevention Lymphocytic choriomeningitis virus infection in organ transplant recipients, Massachusetts and Rhode Island, 2005. MMWR Morb Mortal Wkly Rep. 2005;54:537–539. [PubMed] [Google Scholar]
- 538.Srinivasan A., Burton E.C., Kuehnert M.J. Transmission of rabies virus from an organ donor to four transplant recipients. N Engl J Med. 2005;352:1103–1111. doi: 10.1056/NEJMoa043018. [DOI] [PubMed] [Google Scholar]
- 539.Gottesdiener K.M. Transplanted infections: donor-to-host transmission with the allograft. Ann Intern Med. 1989;110:1001–1016. doi: 10.7326/0003-4819-110-12-1001. [DOI] [PubMed] [Google Scholar]
- 540.Borie D.C., Cramer D.V., Phan-Thanh L. Microbiological hazards related to xenotransplantation of porcine organs into man. Infect Control Hosp Epidemiol. 1998;19:355–365. doi: 10.1086/647830. [DOI] [PubMed] [Google Scholar]
- 541.Centers for Disease Control and Prevention US Public Health Service guideline on infectious disease issues in xenotransplantation. MMWR Recomm Rep. 2001;50:1–46. [PubMed] [Google Scholar]
- 542.Institute of Medicine . National Academy Press; Washington DC: 1999. To err is human: building a safer health system. [Google Scholar]
- 543.Gerberding J.L. Hospital-onset infections: a patient safety issue. Ann Intern Med. 2002;137:665–670. doi: 10.7326/0003-4819-137-8-200210150-00011. [DOI] [PubMed] [Google Scholar]
- 544.Leape L.L., Berwick D.M., Bates D.W. What practices will most improve safety? Evidence-based medicine meets patient safety. JAMA. 2002;288:501–507. doi: 10.1001/jama.288.4.501. [DOI] [PubMed] [Google Scholar]
- 545.Burke J.P. Patient safety. Infection control: a problem for patient safety. N Engl J Med. 2003;348:651–656. doi: 10.1056/NEJMhpr020557. [DOI] [PubMed] [Google Scholar]
- 546.Shulman L., Ost D. Managing infection in the critical care unit: how can infection control make the ICU safe? Crit Care Clin. 2005;21:111–128. doi: 10.1016/j.ccc.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 547.Goldmann D.A., Weinstein R.A., Wenzel R.P. Strategies to prevent and control the emergence and spread of antimicrobial-resistant microorganisms in hospitals: a challenge to hospital leadership. JAMA. 1996;275:234–240. [PubMed] [Google Scholar]
- 548.Scheckler W.E., Brimhall D., Buck A.S. Requirements for infrastructure and essential activities of infection control and epidemiology in hospitals: a consensus panel report. Society for Healthcare Epidemiology of America. Infect Control Hosp Epidemiol. 1998;19:114–124. doi: 10.1086/647779. [DOI] [PubMed] [Google Scholar]
- 549.www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/.
- 550.Jackson M., Chiarello L.A., Gaynes R.P., Gerberding J.L. Nurse staffing and health care–associated infections: proceedings from a working group meeting. Am J Infect Control. 2002;30:199–206. doi: 10.1067/mic.2002.123416. [DOI] [PubMed] [Google Scholar]
- 551.O'Boyle C., Jackson M., Henly S.J. Staffing requirements for infection control programs in US health care facilities: Delphi project. Am J Infect Control. 2002;30:321–333. doi: 10.1067/mic.2002.127930. [DOI] [PubMed] [Google Scholar]
- 552.Peterson L.R., Hamilton J.D., Baron E.J. Role of clinical microbiology laboratories in the management and control of infectious diseases and the delivery of health care. Clin Infect Dis. 2001;32:605–611. doi: 10.1086/318725. [DOI] [PubMed] [Google Scholar]
- 553.McGowan J.E., Jr., Tenover F.C. Confronting bacterial resistance in healthcare settings: a crucial role for microbiologists. Nat Rev Microbiol. 2004;2:251–258. doi: 10.1038/nrmicro845. [DOI] [PubMed] [Google Scholar]
- 554.Available from. Accessed.
- 555.Curtis J.R., Cook D.J., Wall R.J. Intensive care unit quality improvement: a “how-to” guide for the interdisciplinary team. Crit Care Med. 2006;34:211–218. doi: 10.1097/01.ccm.0000190617.76104.ac. [DOI] [PubMed] [Google Scholar]
- 556.Pronovost P.J., Nolan T., Zeger S., Miller M., Rubin H. How can clinicians measure safety and quality in acute care? Lancet. 2004;363:1061–1067. doi: 10.1016/S0140-6736(04)15843-1. [DOI] [PubMed] [Google Scholar]
- 557.Goldrick B.A., Dingle D.A., Gilmore G.K., Curchoe R.M., Plackner C.L., Fabrey L.J. Practice analysis for infection control and epidemiology in the new millennium. Am J Infect Control. 2002;30:437–448. doi: 10.1067/mic.2002.127706. [DOI] [PubMed] [Google Scholar]
- 558.Centers for Disease Control and Prevention Guideline for hand hygiene in health-care settings: recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. MMWR Recomm Rep. 2002;51:1–44. [PubMed] [Google Scholar]
- 559.Bonomo R.A., Rice L.B. Emerging issues in antibiotic resistant infections in long-term care facilities. J Gerontol A Biol Sci Med Sci. 1999;54:B260–B267. doi: 10.1093/gerona/54.6.b260. [DOI] [PubMed] [Google Scholar]
- 560.Larson E.L., Early E., Cloonan P., Sugrue S., Parides M. An organizational climate intervention associated with increased handwashing and decreased nosocomial infections. Behav Med. 2000;26:14–22. doi: 10.1080/08964280009595749. [DOI] [PubMed] [Google Scholar]
- 561.Pittet D., Hugonnet S., Harbarth S. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Infection Control Programme. Lancet. 2000;356:1307–1312. doi: 10.1016/s0140-6736(00)02814-2. [DOI] [PubMed] [Google Scholar]
- 562.Murthy R. Implementation of strategies to control antimicrobial resistance. Chest. 2001;119(2 Suppl):405S–411S. doi: 10.1378/chest.119.2_suppl.405s. [DOI] [PubMed] [Google Scholar]
- 563.Rondeau K.V., Wagar T.H. Organizational learning and continuous quality improvement: examining the impact on nursing home performance. Health Care Manage Forum. 2002;15:17–23. doi: 10.1016/S0840-4704(10)60576-5. [DOI] [PubMed] [Google Scholar]
- 564.Stelfox H.T., Bates D.W., Redelmeier D.A. Safety of patients isolated for infection control. JAMA. 2003;290:1899–1905. doi: 10.1001/jama.290.14.1899. [DOI] [PubMed] [Google Scholar]
- 565.Haley R.W., Culver D.H., White J.W. The efficacy of infection surveillance and control programs in preventing nosocomial infections in US hospitals. Am J Epidemiol. 1985;121:182–205. doi: 10.1093/oxfordjournals.aje.a113990. [DOI] [PubMed] [Google Scholar]
- 566.McArthur B.J., Pugliese G., Weinstein S. A national task analysis of infection control practitioners, 1982. Part One: methodology and demography. Am J Infect Control. 1984;12:88–95. doi: 10.1016/0196-6553(84)90022-1. [DOI] [PubMed] [Google Scholar]
- 567.Shannon R., McArthur B.J., Weinstein S. A national task analysis of infection control practitioners, 1982. Part Two: tasks, knowledge, and abilities for practice. Am J Infect Control. 1984;12:187–196. doi: 10.1016/0196-6553(84)90096-8. [DOI] [PubMed] [Google Scholar]
- 568.Pugliese G., McArthur B.J., Weinstein S. A national task analysis of infection control practitioners, 1982. Part Three: the relationship between hospital size and tasks performed. Am J Infect Control. 1984;12:221–227. doi: 10.1016/0196-6553(84)90112-3. [DOI] [PubMed] [Google Scholar]
- 569.Larson E., Eisenberg R., Soule B.M. Validating the certification process for infection control practice. Am J Infect Control. 1988;16:198–205. doi: 10.1016/0196-6553(88)90060-0. [DOI] [PubMed] [Google Scholar]
- 570.Bjerke N.B., Fabrey L.J., Johnson C.B. Job analysis 1992: infection control practitioner. Am J Infect Control. 1993;21:51–57. doi: 10.1016/0196-6553(93)90224-r. [DOI] [PubMed] [Google Scholar]
- 571.Turner J.G., Kolenc K.M., Docken L. Job analysis 1996: infection control professional. Certification Board in Infection Control and Epidemiology Inc, 1996 Job Analysis Committee. Am J Infect Control. 1999;27:145–157. doi: 10.1016/s0196-6553(99)70091-x. [DOI] [PubMed] [Google Scholar]
- 572.Health Canada, Nosocomial and Occupational Infections Section Development of a resource model for infection prevention and control programs in acute, long-term, and home care settings: conference proceedings of the Infection Prevention and Control Alliance. Am J Infect Cont. 2004;32:2–6. doi: 10.1016/j.ajic.2003.10.002. [DOI] [PubMed] [Google Scholar]
- 573.Lee T.H., Meyer G.S., Brennan T.A. A middle ground on public accountability. N Engl J Med. 2004;350:2409–2412. doi: 10.1056/NEJMsb041193. [DOI] [PubMed] [Google Scholar]
- 574.Stevenson K.B., Murphy C.L., Samore M.H. Assessing the status of infection control programs in small rural hospitals in the western United States. Am J Infect Control. 2004;32:255–261. doi: 10.1016/j.ajic.2003.10.016. [DOI] [PubMed] [Google Scholar]
- 575.Simonds D.N., Horan T.C., Kelley R., Jarvis W.R. Detecting pediatric nosocomial infections: how do infection control and quality assurance personnel compare? Am J Infect Control. 1997;25:202–208. doi: 10.1016/s0196-6553(97)90005-5. [DOI] [PubMed] [Google Scholar]
- 576.Dawson S.J. The role of the infection control link nurse. J Hosp Infect. 2003;54:251–257. doi: 10.1016/s0195-6701(03)00131-2. [DOI] [PubMed] [Google Scholar]
- 577.Wright J., Stover B.H., Wilkerson S., Bratcher D. Expanding the infection control team: development of the infection control liaison position for the neonatal intensive care unit. Am J Infect Control. 2002;30:174–178. doi: 10.1067/mic.2002.119927. [DOI] [PubMed] [Google Scholar]
- 578.Teare E.L., Peacock A. The development of an infection control link-nurse programme in a district general hospital. J Hosp Infect. 1996;34:267–278. doi: 10.1016/s0195-6701(96)90107-3. [DOI] [PubMed] [Google Scholar]
- 579.Ching T.Y., Seto W.H. Evaluating the efficacy of the infection control liaison nurse in the hospital. J Adv Nurs. 1990;15:1128–1131. doi: 10.1111/j.1365-2648.1990.tb01703.x. [DOI] [PubMed] [Google Scholar]
- 580.Amundsen J., Drennan D.P. An infection control nurse-advisor program. Am J Infect Control. 1983;11:20–23. doi: 10.1016/s0196-6553(83)80009-1. [DOI] [PubMed] [Google Scholar]
- 581.Ross K.A. A program for infection surveillance utilizing an infection control liaison nurse. Am J Infect Control. 1982;10:24–28. doi: 10.1016/0196-6553(82)90036-0. [DOI] [PubMed] [Google Scholar]
- 582.Needleman J., Buerhaus P., Mattke S., Stewart M., Zelevinsky K. Nurse-staffing levels and the quality of care in hospitals. N Engl J Med. 2002;346:1715–1722. doi: 10.1056/NEJMsa012247. [DOI] [PubMed] [Google Scholar]
- 583.Dimick J.B., Swoboda S.M., Pronovost P.J., Lipsett P.A. Effect of nurse-to-patient ratio in the intensive care unit on pulmonary complications and resource use after hepatectomy. Am J Crit Care. 2001;10:376–382. [PubMed] [Google Scholar]
- 584.Mayhall C.G., Lamb V.A., Gayle W.E., Jr., Haynes B.W., Jr. Enterobacter cloacae septicemia in a burn center: epidemiology and control of an outbreak. J Infect Dis. 1979;139:166–171. doi: 10.1093/infdis/139.2.166. [DOI] [PubMed] [Google Scholar]
- 585.Goldmann D.A., Durbin W.A., Jr., Freeman J. Nosocomial infections in a neonatal intensive care unit. J Infect Dis. 1981;144:449–459. doi: 10.1093/infdis/144.5.449. [DOI] [PubMed] [Google Scholar]
- 586.Arnow P., Allyn P.A., Nichols E.M., Hill D.L., Pezzlo M., Bartlett R.H. Control of methicillin-resistant Staphylococcus aureus in a burn unit: role of nurse staffing. J Trauma. 1982;22:954–959. doi: 10.1097/00005373-198211000-00012. [DOI] [PubMed] [Google Scholar]
- 587.Haley R.W., Bregman D.A. The role of understaffing and overcrowding in recurrent outbreaks of staphylococcal infection in a neonatal special-care unit. J Infect Dis. 1982;145:875–885. doi: 10.1093/infdis/145.6.875. [DOI] [PubMed] [Google Scholar]
- 588.Fridkin S.K., Pear S.M., Williamson T.H., Galgiani J.N., Jarvis W.R. The role of understaffing in central venous catheter–associated bloodstream infections. Infect Control Hosp Epidemiol. 1996;17:150–158. doi: 10.1086/647262. [DOI] [PubMed] [Google Scholar]
- 589.Robert J., Fridkin S.K., Blumberg H.M. The influence of the composition of the nursing staff on primary bloodstream infection rates in a surgical intensive care unit. Infect Control Hosp Epidemiol. 2000;21:12–17. doi: 10.1086/501690. [DOI] [PubMed] [Google Scholar]
- 590.Li J., Birkhead G.S., Strogatz D.S., Coles F.B. Impact of institution size, staffing patterns, and infection control practices on communicable disease outbreaks in New York State nursing homes. Am J Epidemiol. 1996;143:1042–1049. doi: 10.1093/oxfordjournals.aje.a008668. [DOI] [PubMed] [Google Scholar]
- 591.Archibald L.K., Manning M.L., Bell L.M., Banerjee S., Jarvis W.R. Patient density, nurse-to-patient ratio and nosocomial infection risk in a pediatric cardiac intensive care unit. Pediatr Infect Dis J. 1997;16:1045–1048. doi: 10.1097/00006454-199711000-00008. [DOI] [PubMed] [Google Scholar]
- 592.Harbarth S., Sudre P., Dharan S., Cadenas M., Pittet D. Outbreak of Enterobacter cloacae related to understaffing, overcrowding, and poor hygiene practices. Infect Control Hosp Epidemiol. 1999;20:598–603. doi: 10.1086/501677. [DOI] [PubMed] [Google Scholar]
- 593.Vicca A.F. Nursing staff workload as a determinant of methicillin-resistant Staphylococcus aureus spread in an adult intensive therapy unit. J Hosp Infect. 1999;43:109–113. doi: 10.1053/jhin.1999.0246. [DOI] [PubMed] [Google Scholar]
- 594.Stegenga J., Bell E., Matlow A. The role of nurse understaffing in nosocomial viral gastrointestinal infections on a general pediatrics ward. Infect Control Hosp Epidemiol. 2002;23:133–136. doi: 10.1086/502022. [DOI] [PubMed] [Google Scholar]
- 595.Alonso-Echanove J., Edwards J.R., Richards M.J. Effect of nurse staffing and antimicrobial-impregnated central venous catheters on the risk for bloodstream infections in intensive care units. Infect Control Hosp Epidemiol. 2003;24:916–925. doi: 10.1086/502160. [DOI] [PubMed] [Google Scholar]
- 596.Petrosillo N., Gilli P., Serraino D. Prevalence of infected patients and understaffing have a role in hepatitis C virus transmission in dialysis. Am J Kidney Dis. 2001;37:1004–1010. doi: 10.1016/s0272-6386(05)80017-4. [DOI] [PubMed] [Google Scholar]
- 597.Pfaller M.A., Herwaldt L.A. The clinical microbiology laboratory and infection control: emerging pathogens, antimicrobial resistance, and new technology. Clin Infect Dis. 1997;25:858–870. doi: 10.1086/515557. [DOI] [PubMed] [Google Scholar]
- 598.Simor A.E. The role of the laboratory in infection prevention and control programs in long-term-care facilities for the elderly. Infect Control Hosp Epidemiol. 2001;22:459–463. doi: 10.1086/501935. [DOI] [PubMed] [Google Scholar]
- 599.Weinstein R.A., Mallison G.F. The role of the microbiology laboratory in surveillance and control of nosocomial infections. Am J Clin Pathol. 1978;69:130–136. doi: 10.1093/ajcp/69.2.130. [DOI] [PubMed] [Google Scholar]
- 600.Kolmos H.J. Interaction between the microbiology laboratory and clinician: what the microbiologist can provide. J Hosp Infect. 1999;43(Suppl):S285–S291. doi: 10.1016/s0195-6701(99)90101-9. [DOI] [PubMed] [Google Scholar]
- 601.Clinical and Laboratory Standards Institute. www.clsi.org/.
- 602.Ginocchio C.C. Role of NCCLS in antimicrobial susceptibility testing and monitoring. Am J Health Syst Pharm. 2002;59(8 Suppl. 3):S7–S11. doi: 10.1093/ajhp/59.suppl_3.S7. [DOI] [PubMed] [Google Scholar]
- 603.National Committee for Clinical Laboratory Standards . National Committee for Clinical Laboratory Standards; Wayne (PA): 2002. Performance standards for antimicrobial susceptibility testing: twelfth informational supplement. Document M100–S12. [Google Scholar]
- 604.National Committee for Clinical Laboratory Standards . National Committee for Clinical Laboratory Standards; Wayne (PA): 2002. Analysis and presentation of cumulative antimicrobial susceptibility test data: approved guideline. Document M39-A. [Google Scholar]
- 605.Halstead D.C., Gomez N., McCarter Y.S. Reality of developing a community-wide antibiogram. J Clin Microbiol. 2004;42:1–6. doi: 10.1128/JCM.42.1.1-6.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 606.Ernst E.J., Diekema D.J., BootsMiller B.J. Are United States hospitals following national guidelines for the analysis and presentation of cumulative antimicrobial susceptibility data? Diagn Microbiol Infect Dis. 2004;49:141–145. doi: 10.1016/j.diagmicrobio.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 607.Bergeron M.G., Ouellette M. Preventing antibiotic resistance through rapid genotypic identification of bacteria and of their antibiotic resistance genes in the clinical microbiology laboratory. J Clin Microbiol. 1998;36:2169–2172. doi: 10.1128/jcm.36.8.2169-2172.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 608.Hacek D.M., Suriano T., Noskin G.A., Kruszynski J., Reisberg B., Peterson L.R. Medical and economic benefit of a comprehensive infection control program that includes routine determination of microbial clonality. Am J Clin Pathol. 1999;111:647–654. doi: 10.1093/ajcp/111.5.647. [DOI] [PubMed] [Google Scholar]
- 609.Rodriguez W.J., Schwartz R.H., Thorne M.M. Evaluation of diagnostic tests for influenza in a pediatric practice. Pediatr Infect Dis J. 2002;21:193–196. doi: 10.1097/00006454-200203000-00006. [DOI] [PubMed] [Google Scholar]
- 610.Centers for Disease Control and Prevention Prevention and control of influenza: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2005;54:1–40. [PubMed] [Google Scholar]
- 611.Uyeki T.M. Influenza diagnosis and treatment in children: a review of studies on clinically useful tests and antiviral treatment for influenza. Pediatr Infect Dis J. 2003;22:164–177. doi: 10.1097/01.inf.0000050458.35010.b6. [DOI] [PubMed] [Google Scholar]
- 612.Chan E.L., Antonishyn N., McDonald R. The use of TaqMan PCR assay for detection of Bordetella pertussis infection from clinical specimens. Arch Pathol Lab Med. 2002;126:173–176. doi: 10.5858/2002-126-0173-TUOTPA. [DOI] [PubMed] [Google Scholar]
- 613.Barenfanger J., Drake C., Kacich G. Clinical and financial benefits of rapid bacterial identification and antimicrobial susceptibility testing. J Clin Microbiol. 1999;37:1415–1418. doi: 10.1128/jcm.37.5.1415-1418.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 614.Barenfanger J., Drake C., Leon N., Mueller T., Troutt T. Clinical and financial benefits of rapid detection of respiratory viruses: an outcomes study. J Clin Microbiol. 2000;38:2824–2828. doi: 10.1128/jcm.38.8.2824-2828.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 615.Ramers C., Billman G., Hartin M., Ho S., Sawyer M.H. Impact of a diagnostic cerebrospinal fluid enterovirus polymerase chain reaction test on patient management. JAMA. 2000;283:2680–2685. doi: 10.1001/jama.283.20.2680. [DOI] [PubMed] [Google Scholar]
- 616.Mackie P.L., Joannidis P.A., Beattie J. Evaluation of an acute point-of-care system screening for respiratory syncytial virus infection. J Hosp Infect. 2001;48:66–71. doi: 10.1053/jhin.2001.0942. [DOI] [PubMed] [Google Scholar]
- 617.Guillemot D., Courvalin P. Better control of antibiotic resistance. Clin Infect Dis. 2001;33:542–547. doi: 10.1086/322583. [DOI] [PubMed] [Google Scholar]
- 618.Paterson D.L. The role of antimicrobial management programs in optimizing antibiotic prescribing within hospitals. Clin Infect Dis. 2006;42(Suppl 2):S90–S95. doi: 10.1086/499407. [DOI] [PubMed] [Google Scholar]
- 619.Lundstrom T., Pugliese G., Bartley J., Cox J., Guither C. Organizational and environmental factors that affect worker health and safety and patient outcomes. Am J Infect Control. 2002;30:93–106. doi: 10.1067/mic.2002.119820. [DOI] [PubMed] [Google Scholar]
- 620.www.patientsafety.com/vision.html.
- 621.Pronovost P.J., Jenckes M.W., Dorman T. Organizational characteristics of intensive care units related to outcomes of abdominal aortic surgery. JAMA. 1999;281:1310–1317. doi: 10.1001/jama.281.14.1310. [DOI] [PubMed] [Google Scholar]
- 622.Pronovost P.J., Angus D.C., Dorman T., Robinson K.A., Dremsizov T.T., Young T.L. Physician staffing patterns and clinical outcomes in critically ill patients: a systematic review. JAMA. 2002;288:2151–2162. doi: 10.1001/jama.288.17.2151. [DOI] [PubMed] [Google Scholar]
- 623.Pronovost P.J., Weast B., Holzmueller C.G. Evaluation of the culture of safety: survey of clinicians and managers in an academic medical center. Qual Saf Health Care. 2003;12:405–410. doi: 10.1136/qhc.12.6.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 624.Nieva V.F., Sorra J. Safety culture assessment: a tool for improving patient safety in healthcare organizations. Qual Saf Health Care. 2003;12(Suppl 2):ii17–ii23. doi: 10.1136/qhc.12.suppl_2.ii17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 625.Clarke S.P., Rockett J.L., Sloane D.M., Aiken L.H. Organizational climate, staffing, and safety equipment as predictors of needlestick injuries and near-misses in hospital nurses. Am J Infect Control. 2002;30:207–216. doi: 10.1067/mic.2002.123392. [DOI] [PubMed] [Google Scholar]
- 626.Rivers D.L., Aday L.A., Frankowski R.F., Felknor S., White D., Nichols B. Predictors of nurses' acceptance of an intravenous catheter safety device. Nurs Res. 2003;52:249–255. doi: 10.1097/00006199-200307000-00007. [DOI] [PubMed] [Google Scholar]
- 627.Gershon R.R., Karkashian C.D., Grosch J.W. Hospital safety climate and its relationship with safe work practices and workplace exposure incidents. Am J Infect Control. 2000;28:211–221. doi: 10.1067/mic.2000.105288. [DOI] [PubMed] [Google Scholar]
- 628.Gershon R.R., Vlahov D., Felknor S.A. Compliance with universal precautions among health care workers at three regional hospitals. Am J Infect Control. 1995;23:225–236. doi: 10.1016/0196-6553(95)90067-5. [DOI] [PubMed] [Google Scholar]
- 629.Michalsen A., Delclos G.L., Felknor S.A. Compliance with universal precautions among physicians. J Occup Environ Med. 1997;39:130–137. doi: 10.1097/00043764-199702000-00010. [DOI] [PubMed] [Google Scholar]
- 630.Vaughn T.E., McCoy K.D., Beekmann S.E., Woolson R.E., Torner J.C., Doebbeling B.N. Factors promoting consistent adherence to safe needle precautions among hospital workers. Infect Control Hosp Epidemiol. 2004;25:548–555. doi: 10.1086/502438. [DOI] [PubMed] [Google Scholar]
- 631.Grosch J.W., Gershon R.R., Murphy L.R., DeJoy D.M. Safety climate dimensions associated with occupational exposure to blood-borne pathogens in nurses. Am J Ind Med. 1999;(Suppl 1):122–124. doi: 10.1002/(sici)1097-0274(199909)36:1+<122::aid-ajim43>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 632.Piotrowski M.M., Hinshaw D.B. The safety checklist program: creating a culture of safety in intensive care units. Jt Comm J Qual Improv. 2002;28:306–315. doi: 10.1016/s1070-3241(02)28030-x. [DOI] [PubMed] [Google Scholar]
- 633.Weeks W.B., Bagian J.P. Developing a culture of safety in the Veterans Health Administration. Eff Clin Pract. 2000;3:270–276. [PubMed] [Google Scholar]
- 634.Bagian J.P., Gosbee J.W. Developing a culture of patient safety at the VA. Ambul Outreach. 2000:25–29. [PubMed] [Google Scholar]
- 635.Tokars J.I., McKinley G.F., Otten J. Use and efficacy of tuberculosis infection control practices at hospitals with previous outbreaks of multidrug-resistant tuberculosis. Infect Control Hosp Epidemiol. 2001;22:449–455. doi: 10.1086/501933. [DOI] [PubMed] [Google Scholar]
- 636.Maloney S.A., Pearson M.L., Gordon M.T., Del Castillo R., Boyle J.F., Jarvis W.R. Efficacy of control measures in preventing nosocomial transmission of multidrug-resistant tuberculosis to patients and health care workers. Ann Intern Med. 1995;122:90–95. doi: 10.7326/0003-4819-122-2-199501150-00002. [DOI] [PubMed] [Google Scholar]
- 637.Montecalvo M.A., Jarvis W.R., Uman J. Infection-control measures reduce transmission of vancomycin-resistant enterococci in an endemic setting. Ann Intern Med. 1999;131:269–272. doi: 10.7326/0003-4819-131-4-199908170-00006. [DOI] [PubMed] [Google Scholar]
- 638.Sherertz R.J., Ely E.W., Westbrook D.M. Education of physicians-in-training can decrease the risk for vascular catheter infection. Ann Intern Med. 2000;132:641–648. doi: 10.7326/0003-4819-132-8-200004180-00007. [DOI] [PubMed] [Google Scholar]
- 639.Lynch P., Cummings M.J., Roberts P.L., Herriott M.J., Yates B., Stamm W.E. Implementing and evaluating a system of generic infection precautions: body substance isolation. Am J Infect Control. 1990;18:1–12. doi: 10.1016/0196-6553(90)90204-6. [DOI] [PubMed] [Google Scholar]
- 640.Kelen G.D., DiGiovanna T.A., Celentano D.D. Adherence to universal (barrier) precautions during interventions on critically ill and injured emergency department patients. J Acquir Immune Defic Syndr. 1990;3:987–994. [PubMed] [Google Scholar]
- 641.Courington K.R., Patterson S.L., Howard R.J. Universal precautions are not universally followed. Arch Surg. 1991;126:93–96. doi: 10.1001/archsurg.1991.01410250099016. [DOI] [PubMed] [Google Scholar]
- 642.Kaczmarek R.G., Moore R.M., Jr., McCrohan J. Glove use by health care workers: results of a tristate investigation. Am J Infect Control. 1991;19:228–232. doi: 10.1016/s0196-6553(05)80253-6. [DOI] [PubMed] [Google Scholar]
- 643.Freeman S.W., Chambers C.V. Compliance with universal precautions in a medical practice with a high rate of HIV infection. J Am Board Fam Pract. 1992;5:313–318. [PubMed] [Google Scholar]
- 644.Friedland L.R., Joffe M., Wiley J.F., 2nd, Schapire A., Moore D.F. Effect of educational program on compliance with glove use in a pediatric emergency department. Am J Dis Child. 1992;146:1355–1358. doi: 10.1001/archpedi.1992.02160230113030. [DOI] [PubMed] [Google Scholar]
- 645.Henry K., Campbell S., Maki M. A comparison of observed and self-reported compliance with universal precautions among emergency department personnel at a Minnesota public teaching hospital: implications for assessing infection control programs. Ann Emerg Med. 1992;21:940–946. doi: 10.1016/s0196-0644(05)82932-4. [DOI] [PubMed] [Google Scholar]
- 646.Henry K., Campbell S., Collier P., Williams C.O. Compliance with universal precautions and needle handling and disposal practices among emergency department staff at two community hospitals. Am J Infect Control. 1994;22:129–137. doi: 10.1016/0196-6553(94)90001-9. [DOI] [PubMed] [Google Scholar]
- 647.Eustis T.C., Wright S.W., Wrenn K.D., Fowlie E.J., Slovis C.M. Compliance with recommendations for universal precautions among prehospital providers. Ann Emerg Med. 1995;25:512–515. doi: 10.1016/s0196-0644(95)70268-7. [DOI] [PubMed] [Google Scholar]
- 648.DiGiacomo J.C., Hoff W.S., Rotondo M.F. Barrier precautions in trauma resuscitation: real-time analysis utilizing videotape review. Am J Emerg Med. 1997;15:34–39. doi: 10.1016/s0735-6757(97)90044-9. [DOI] [PubMed] [Google Scholar]
- 649.Thompson B.L., Dwyer D.M., Ussery X.T., Denman S., Vacek P., Schwartz B. Handwashing and glove use in a long-term-care facility. Infect Control Hosp Epidemiol. 1997;18:97–103. doi: 10.1086/647562. [DOI] [PubMed] [Google Scholar]
- 650.Helfgott A.W., Taylor-Burton J., Garcini F.J., Eriksen N.L., Grimes R. Compliance with universal precautions: knowledge and behavior of residents and students in a department of obstetrics and gynecology. Infect Dis Obstet Gynecol. 1998;6:123–128. doi: 10.1002/(SICI)1098-0997(1998)6:3<123::AID-IDOG5>3.0.CO;2-Y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 651.Moore S., Goodwin H., Grossberg R., Toltzis P. Compliance with universal precautions among pediatric residents. Arch Pediatr Adolesc Med. 1998;152:554–557. doi: 10.1001/archpedi.152.6.554. [DOI] [PubMed] [Google Scholar]
- 652.Akduman D., Kim L.E., Parks R.L. Use of personal protective equipment and operating room behaviors in four surgical subspecialties: personal protective equipment and behaviors in surgery. Infect Control Hosp Epidemiol. 1999;20:110–114. doi: 10.1086/501601. [DOI] [PubMed] [Google Scholar]
- 653.Brooks A.J., Phipson M., Potgieter A., Koertzen H., Boffard K.D. Education of the trauma team: video evaluation of the compliance with universal barrier precautions in resuscitation. Eur J Surg. 1999;165:1125–1128. doi: 10.1080/110241599750007621. [DOI] [PubMed] [Google Scholar]
- 654.Kidd F., Heitkemper P., Kressel A.B. A comprehensive educational approach to improving patient isolation practice. Clin Perform Qual Health Care. 1999;7:74–76. [PubMed] [Google Scholar]
- 655.Madan A.K., Rentz D.E., Wahle M.J., Flint L.M. Noncompliance of health care workers with universal precautions during trauma resuscitations. South Med J. 2001;94:277–280. [PubMed] [Google Scholar]
- 656.Madan A.K., Raafat A., Hunt J.P., Rentz D., Wahle M.J., Flint L.M. Barrier precautions in trauma: is knowledge enough? J Trauma. 2002;52:540–543. doi: 10.1097/00005373-200203000-00020. [DOI] [PubMed] [Google Scholar]
- 657.Jeffe D.B., Mutha S., Kim L.E., Evanoff B.A., Fraser V.J. Evaluation of a preclinical, educational and skills-training program to improve students' use of blood and body fluid precautions: one-year follow-up. Prev Med. 1999;29:365–373. doi: 10.1006/pmed.1999.0571. [DOI] [PubMed] [Google Scholar]
- 658.Williams C.O., Campbell S., Henry K., Collier P. Variables influencing worker compliance with universal precautions in the emergency department. Am J Infect Control. 1994;22:138–148. doi: 10.1016/0196-6553(94)90002-7. [DOI] [PubMed] [Google Scholar]
- 659.Larson E., McGeer A., Quraishi Z.A. Effect of an automated sink on handwashing practices and attitudes in high-risk units. Infect Control Hosp Epidemiol. 1991;12:422–428. doi: 10.1086/646372. [DOI] [PubMed] [Google Scholar]
- 660.Swoboda S.M., Earsing K., Strauss K., Lane S., Lipsett P.A. Electronic monitoring and voice prompts improve hand hygiene and decrease nosocomial infections in an intermediate care unit. Crit Care Med. 2004;32:358–363. doi: 10.1097/01.CCM.0000108866.48795.0F. [DOI] [PubMed] [Google Scholar]
- 661.Kretzer E.K., Larson E.L. Behavioral interventions to improve infection control practices. Am J Infect Control. 1998;26:245–253. doi: 10.1016/s0196-6553(98)80008-4. [DOI] [PubMed] [Google Scholar]
- 662.Centers for Disease Control and Prevention Updated guidelines for evaluating public health surveillance systems: recommendations from the Guidelines Working Group. MMWR Recomm Rep. 2001;50:1–35. [PubMed] [Google Scholar]
- 663.Semmelweiss IP. Die aetiologie, der begriff und die prophylaxis des kindbettfiebers. Pest, Wein, und Leipzig: CA Harleben's Verlags-Expedition; 1861.
- 664.Bratzler D.W., Houck P.M. Antimicrobial prophylaxis for surgery: an advisory statement from the National Surgical Infection Prevention Project. Clin Infect Dis. 2004;38:1706–1715. doi: 10.1086/421095. [DOI] [PubMed] [Google Scholar]
- 665.Bloom B.T., Craddock A., Delmore P.M. Reducing acquired infections in the NICU: observing and implementing meaningful differences in process between high and low acquired infection rate centers. J Perinatol. 2003;23:489–492. doi: 10.1038/sj.jp.7210981. [DOI] [PubMed] [Google Scholar]
- 666.Braun B.I., Kritchevsky S.B., Wong E.S. Preventing central venous catheter–associated primary bloodstream infections: characteristics of practices among hospitals participating in the Evaluation of Processes and Indicators in Infection Control (EPIC) study. Infect Control Hosp Epidemiol. 2003;24:926–935. doi: 10.1086/502161. [DOI] [PubMed] [Google Scholar]
- 667.Baker O.G. Process surveillance: an epidemiologic challenge for all health care organizations. Am J Infect Cont. 1997;25:96–101. doi: 10.1016/s0196-6553(97)90034-1. [DOI] [PubMed] [Google Scholar]
- 668.Loeb M., McGeer A., McArthur M., Peeling R.W., Petric M., Simor A.E. Surveillance for outbreaks of respiratory tract infections in nursing homes. CMAJ. 2000;162:1133–1137. [PMC free article] [PubMed] [Google Scholar]
- 669.Nicolle L.E. Preventing infections in non-hospital settings: long-term care. Emerg Infect Dis. 2001;7:205–207. doi: 10.3201/eid0702.010210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 670.Pottinger J.M., Herwaldt L.A., Perl T.M. Basics of surveillance: an overview. Infect Control Hosp Epidemiol. 1997;18:513–527. doi: 10.1086/647659. [DOI] [PubMed] [Google Scholar]
- 671.Lee T.B., Baker O.G., Lee J.T., Scheckler W.E., Steele L., Laxton C.E. Recommended practices for surveillance. Association for Professionals in Infection Control and Epidemiology Inc, Surveillance Initiative Working Group. Am J Infect Control. 1998;26:277–288. doi: 10.1016/s0196-6553(98)80013-8. [DOI] [PubMed] [Google Scholar]
- 672.Haley R.W. The scientific basis for using surveillance and risk factor data to reduce nosocomial infection rates. J Hosp Infect. 1995;30(Suppl):3–14. doi: 10.1016/0195-6701(95)90001-2. [DOI] [PubMed] [Google Scholar]
- 673.Benneyan J.C., Lloyd R.C., Plsek P.E. Statistical process control as a tool for research and healthcare improvement. Qual Saf Health Care. 2003;12:458–464. doi: 10.1136/qhc.12.6.458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 674.Lemmen S.W., Zolldann D., Gastmeier P., Lutticken R. Implementing and evaluating a rotating surveillance system and infection control guidelines in 4 intensive care units. Am J Infect Control. 2001;29:89–93. doi: 10.1067/mic.2001.111415. [DOI] [PubMed] [Google Scholar]
- 675.Gaynes R., Richards C., Edwards J. Feeding back surveillance data to prevent hospital-acquired infections. Emerg Infect Dis. 2001;7:295–298. doi: 10.3201/eid0702.010230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 676.Tokars J.I., Richards C., Andrus M. The changing face of surveillance for health care–associated infections. Clin Infect Dis. 2004;39:1347–1352. doi: 10.1086/425000. [DOI] [PubMed] [Google Scholar]
- 677.Sands K.E., Yokoe D.S., Hooper D.C. Detection of postoperative surgical-site infections: comparison of health plan–based surveillance with hospital-based programs. Infect Control Hosp Epidemiol. 2003;24:741–743. doi: 10.1086/502123. [DOI] [PubMed] [Google Scholar]
- 678.Jodra V.M., Rodela A.R., Martinez E.M., Fresnena N.L. Standardized infection ratios for three general surgery procedures: a comparison between Spanish hospitals and US centers participating in the National Nosocomial Infections Surveillance System. Infect Control Hosp Epidemiol. 2003;24:744–748. doi: 10.1086/502124. [DOI] [PubMed] [Google Scholar]
- 679.McKibben L., Horan T., Tokars J.I. Guidance on public reporting of healthcare–associated infections: recommendations of the Healthcare Infection Control Practices Advisory Committee. Am J Infect Control. 2005;33:217–226. doi: 10.1016/j.ajic.2005.04.001. [DOI] [PubMed] [Google Scholar]
- 680.Gould D., Chamberlain A. The use of a ward-based educational teaching package to enhance nurses' compliance with infection control procedures. J Clin Nurs. 1997;6:55–67. doi: 10.1111/j.1365-2702.1997.tb00284.x. [DOI] [PubMed] [Google Scholar]
- 681.Calabro K., Weltge A., Parnell S., Kouzekanani K., Ramirez E. Intervention for medical students: effective infection control. Am J Infect Control. 1998;26:431–436. doi: 10.1016/s0196-6553(98)70041-0. [DOI] [PubMed] [Google Scholar]
- 682.Haiduven D.J., Hench C.P., Simpkins S.M., Stevens D.A. Standardized management of patients and employees exposed to pertussis. Infect Control Hosp Epidemiol. 1998;19:861–864. doi: 10.1086/647748. [DOI] [PubMed] [Google Scholar]
- 683.Macartney K.K., Gorelick M.H., Manning M.L., Hodinka R.L., Bell L.M. Nosocomial respiratory syncytial virus infections: the cost-effectiveness and cost benefit of infection control. Pediatrics. 2000;106:520–526. doi: 10.1542/peds.106.3.520. [DOI] [PubMed] [Google Scholar]
- 684.Beekmann S.E., Vaughn T.E., McCoy K.D. Hospital bloodborne pathogens programs: program characteristics and blood and body fluid exposure rates. Infect Control Hosp Epidemiol. 2001;22:73–82. doi: 10.1086/501867. [DOI] [PubMed] [Google Scholar]
- 685.Sokas R.K., Simmens S., Scott J. A training program in universal precautions for second-year medical students. Acad Med. 1993;68:374–376. doi: 10.1097/00001888-199305000-00022. [DOI] [PubMed] [Google Scholar]
- 686.Ostrowsky B.E., Trick W.E., Sohn A.H. Control of vancomycin-resistant enterococcus in health care facilities in a region. N Engl J Med. 2001;344:1427–1433. doi: 10.1056/NEJM200105103441903. [DOI] [PubMed] [Google Scholar]
- 687.Bonten M.J., Kollef M.H., Hall J.B. Risk factors for ventilator-associated pneumonia: from epidemiology to patient management. Clin Infect Dis. 2004;38:1141–1149. doi: 10.1086/383039. [DOI] [PubMed] [Google Scholar]
- 688.Lau J.T., Fung K.S., Wong T.W. SARS transmission among hospital workers in Hong Kong. Emerg Infect Dis. 2004;10:280–286. doi: 10.3201/eid1002.030534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 689.Talbot T.R., Bradley S.E., Cosgrove S.E., Ruef C., Siegel J.D., Weber D.J. Influenza vaccination of healthcare workers and vaccine allocation for healthcare workers during vaccine shortages. Infect Control Hosp Epidemiol. 2005;26:882–890. doi: 10.1086/502512. [DOI] [PubMed] [Google Scholar]
- 690.Harbarth S., Siegrist C.A., Schira J.C., Wunderli W., Pittet D. Influenza immunization: improving compliance of healthcare workers. Infect Control Hosp Epidemiol. 1998;19:337–342. doi: 10.1086/647825. [DOI] [PubMed] [Google Scholar]
- 691.Bryant K.A., Stover B., Cain L., Levine G.L., Siegel J., Jarvis W.R. Improving influenza immunization rates among healthcare workers caring for high-risk pediatric patients. Infect Control Hosp Epidemiol. 2004;25:912–917. doi: 10.1086/502319. [DOI] [PubMed] [Google Scholar]
- 692.Martinello R.A., Jones L., Topal J.E. Correlation between healthcare workers' knowledge of influenza vaccine and vaccine receipt. Infect Control Hosp Epidemiol. 2003;24:845–847. doi: 10.1086/502147. [DOI] [PubMed] [Google Scholar]
- 693.Goldrick B., Gruendemann B., Larson E. Learning styles and teaching/learning strategy preferences: implications for educating nurses in critical care, the operating room, and infection control. Heart Lung. 1993;22:176–182. [PubMed] [Google Scholar]
- 694.Davis D., O'Brien M.A., Freemantle N., Wolf F.M., Mazmanian P., Taylor-Vaisey A. Impact of formal continuing medical education: do conferences, workshops, rounds, and other traditional continuing education activities change physician behavior or health care outcomes? JAMA. 1999;282:867–874. doi: 10.1001/jama.282.9.867. [DOI] [PubMed] [Google Scholar]
- 695.Carr H., Hinson P. 2nd edition. Association for Professionals in Infection Control and Epidemiology; Washington, DC: 2005. Education and training. In APIC Infection Control and Epidemiology. p 11-1. [Google Scholar]
- 696.Caffarella R.S. 2nd ed. Jossey-Bass; San Francisco: 2001. Planning programs for adult learners: a practical guide for educators, trainers, and staff developers. [Google Scholar]
- 697.Sargeant J., Curran V., Jarvis-Selinger S. Interactive on-line continuing medical education: physicians' perceptions and experiences. J Contin Educ Health Prof. 2004;24:227–236. doi: 10.1002/chp.1340240406. [DOI] [PubMed] [Google Scholar]
- 698.Van Harrison R. Systems-based framework for continuing medical education and improvements in translating new knowledge into physicians' practices. J Contin Educ Health Prof. 2004;24(Suppl 1):S50–S62. doi: 10.1002/chp.1340240508. [DOI] [PubMed] [Google Scholar]
- 699.Cole T.B., Glass R.M. Learning associated with participation in journal-based continuing medical education. J Contin Educ Health Prof. 2004;24:205–212. doi: 10.1002/chp.1340240404. [DOI] [PubMed] [Google Scholar]
- 700.Diekema D.J., Albanese M.A., Schuldt S.S., Doebbeling B.N. Blood and body fluid exposures during clinical training: relation to knowledge of universal precautions. J Gen Intern Med. 1996;11:109–111. doi: 10.1007/BF02599587. [DOI] [PubMed] [Google Scholar]
- 701.Diekema D.J., Schuldt S.S., Albanese M.A., Doebbeling B.N. Universal precautions training of preclinical students: impact on knowledge, attitudes, and compliance. Prev Med. 1995;24:580–585. doi: 10.1006/pmed.1995.1092. [DOI] [PubMed] [Google Scholar]
- 702.Warren D.K., Zack J.E., Cox M.J., Cohen M.M., Fraser V.J. An educational intervention to prevent catheter-associated bloodstream infections in a nonteaching, community medical center. Crit Care Med. 2003;31:1959–1963. doi: 10.1097/01.CCM.0000069513.15417.1C. [DOI] [PubMed] [Google Scholar]
- 703.Dubbert P.M., Dolce J., Richter W., Miller M., Chapman S.W. Increasing ICU staff handwashing: effects of education and group feedback. Infect Control Hosp Epidemiol. 1990;11:191–193. doi: 10.1086/646148. [DOI] [PubMed] [Google Scholar]
- 704.Avila-Aguero M.L., Umana M.A., Jimenez A.L., Faingezicht I., Paris M.M. Handwashing practices in a tertiary-care, pediatric hospital and the effect on an educational program. Clin Perform Qual Health Care. 1998;6:70–72. [PubMed] [Google Scholar]
- 705.Lai K.K., Fontecchio S.A., Kelley A.L., Melvin Z.S. Knowledge of the transmission of tuberculosis and infection control measures for tuberculosis among healthcare workers. Infect Control Hosp Epidemiol. 1996;17:168–170. doi: 10.1086/647265. [DOI] [PubMed] [Google Scholar]
- 706.Koenig S., Chu J. Senior medical students' knowledge of universal precautions. Acad Med. 1993;68:372–374. doi: 10.1097/00001888-199305000-00021. [DOI] [PubMed] [Google Scholar]
- 707.Babcock H.M., Zack J.E., Garrison T. An educational intervention to reduce ventilator-associated pneumonia in an integrated health system: a comparison of effects. Chest. 2004;125:2224–2231. doi: 10.1378/chest.125.6.2224. [DOI] [PubMed] [Google Scholar]
- 708.McGuckin M., Taylor A., Martin V., Porten L., Salcido R. Evaluation of a patient education model for increasing hand hygiene compliance in an inpatient rehabilitation unit. Am J Infect Control. 2004;32:235–238. doi: 10.1016/j.ajic.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 709.Cirone N. Patient-education handbook. Nursing. 1997;27:44–45. [PubMed] [Google Scholar]
- 710.Chase T.M. Learning styles and teaching strategies: enhancing the patient education experience. SCI Nursing. 2001;18:138–141. [PubMed] [Google Scholar]
- 711.Jarvis W.R. Handwashing: the Semmelweis lesson forgotten? Lancet. 1994;344:1311–1312. doi: 10.1016/s0140-6736(94)90687-4. [DOI] [PubMed] [Google Scholar]
- 712.Daniels I.R., Rees B.I. Handwashing: simple, but effective. Ann R Coll Surg Engl. 1999;81:117–118. [PMC free article] [PubMed] [Google Scholar]
- 713.Webster J., Faoagali J.L., Cartwright D. Elimination of methicillin-resistant Staphylococcus aureus from a neonatal intensive care unit after hand washing with triclosan. J Paediatr Child Health. 1994;30:59–64. doi: 10.1111/j.1440-1754.1994.tb00568.x. [DOI] [PubMed] [Google Scholar]
- 714.Zafar A.B., Butler R.C., Reese D.J., Gaydos L.A., Mennonna P.A. Use of 0.3% triclosan (Bacti-Stat) to eradicate an outbreak of methicillin-resistant Staphylococcus aureus in a neonatal nursery. Am J Infect Control. 1995;23:200–208. doi: 10.1016/0196-6553(95)90042-x. [DOI] [PubMed] [Google Scholar]
- 715.Malik R.K., Montecalvo M.A., Reale M.R. Epidemiology and control of vancomycin-resistant enterococci in a regional neonatal intensive care unit. Pediatr Infect Dis J. 1999;18:352–356. doi: 10.1097/00006454-199904000-00009. [DOI] [PubMed] [Google Scholar]
- 716.Pittet D., Boyce J.M. Hand hygiene and patient care: pursuing the Semmelweis legacy. Lancet Infect Dis. 2001:9–20. [Google Scholar]
- 717.Lin C.M., Wu F.M., Kim H.K., Doyle M.P., Michael B.S., Williams L.K. A comparison of hand-washing techniques to remove Escherichia coli and caliciviruses under natural or artificial fingernails. J Food Prot. 2003;66:2296–2301. doi: 10.4315/0362-028x-66.12.2296. [DOI] [PubMed] [Google Scholar]
- 718.Edel E., Houston S., Kennedy V., LaRocco M. Impact of a 5-minute scrub on the microbial flora found on artificial, polished, or natural fingernails of operating room personnel. Nurs Res. 1998;47:54–59. doi: 10.1097/00006199-199801000-00010. [DOI] [PubMed] [Google Scholar]
- 719.Pottinger J., Burns S., Manske C. Bacterial carriage by artificial versus natural nails. Am J Infect Control. 1989;17:340–344. doi: 10.1016/0196-6553(89)90003-5. [DOI] [PubMed] [Google Scholar]
- 720.Hedderwick S.A., McNeil S.A., Lyons M.J., Kauffman C.A. Pathogenic organisms associated with artificial fingernails worn by healthcare workers. Infect Control Hosp Epidemiol. 2000;21:505–509. doi: 10.1086/501794. [DOI] [PubMed] [Google Scholar]
- 721.Passaro D.J., Waring L., Armstrong R. Postoperative Serratia marcescens wound infections traced to an out-of- hospital source. J Infect Dis. 1997;175:992–995. doi: 10.1086/514008. [DOI] [PubMed] [Google Scholar]
- 722.Moolenaar R.L., Crutcher J.M., San Joaquin V.H. A prolonged outbreak of Pseudomonas aeruginosa in a neonatal intensive care unit: did staff fingernails play a role in disease transmission? Infect Control Hosp Epidemiol. 2000;21:80–85. doi: 10.1086/501739. [DOI] [PubMed] [Google Scholar]
- 723.Parry M.F., Grant B., Yukna M. Candida osteomyelitis and diskitis after spinal surgery: an outbreak that implicates artificial nail use. Clin Infect Dis. 2001;32:352–357. doi: 10.1086/318487. [DOI] [PubMed] [Google Scholar]
- 724.Boszczowski I., Nicoletti C., Puccini D.M. Outbreak of extended spectrum beta-lactamase–producing Klebsiella pneumoniae infection in a neonatal intensive care unit related to onychomycosis in a health care worker. Pediatr Infect Dis J. 2005;24:648–650. doi: 10.1097/01.inf.0000168844.55898.8f. [DOI] [PubMed] [Google Scholar]
- 725.Trick W.E., Vernon M.O., Hayes R.A. Impact of ring wearing on hand contamination and comparison of hand hygiene agents in a hospital. Clin Infect Dis. 2003;36:1383–1390. doi: 10.1086/374852. [DOI] [PubMed] [Google Scholar]
- 726.Pittet D., Dharan S., Touveneau S., Sauvan V., Perneger T.V. Bacterial contamination of the hands of hospital staff during routine patient care. Arch Intern Med. 1999;159:821–826. doi: 10.1001/archinte.159.8.821. [DOI] [PubMed] [Google Scholar]
- 727.Tenorio A.R., Badri S.M., Sahgal N.B. Effectiveness of gloves in the prevention of hand carriage of vancomycin-resistant enterococcus species by health care workers after patient care. Clin Infect Dis. 2001;32:826–829. doi: 10.1086/319214. [DOI] [PubMed] [Google Scholar]
- 728.Mast S.T., Woolwine J.D., Gerberding J.L. Efficacy of gloves in reducing blood volumes transferred during simulated needlestick injury. J Infect Dis. 1993;168:1589–1592. doi: 10.1093/infdis/168.6.1589. [DOI] [PubMed] [Google Scholar]
- 729.Medical Glove Guidance Manual. Available from. Accessed.
- 730.Korniewicz D.M., El-Masri M., Broyles J.M., Martin C.D., O'Connell K.P. Performance of latex and nonlatex medical examination gloves during simulated use. Am J Infect Control. 2002;30:133–138. doi: 10.1067/mic.2002.119512. [DOI] [PubMed] [Google Scholar]
- 731.Korniewicz D.M., McLeskey S.W. Latex allergy and gloving standards. Semin Perioper Nurs. 1998;7:216–221. [PubMed] [Google Scholar]
- 732.Ranta P.M., Ownby D.R. A review of natural-rubber latex allergy in health care workers. Clin Infect Dis. 2004;38:252–256. doi: 10.1086/380789. [DOI] [PubMed] [Google Scholar]
- 733.Korniewicz D.M., Kirwin M., Cresci K. Barrier protection with examination gloves: double versus single. Am J Infect Control. 1994;22:12–15. doi: 10.1016/0196-6553(94)90085-x. [DOI] [PubMed] [Google Scholar]
- 734.Korniewicz D.M., Kirwin M., Cresci K., Larson E. Leakage of latex and vinyl exam gloves in high- and low-risk clinical settings. Am Ind Hyg Assoc J. 1993;54:22–26. doi: 10.1080/15298669391354261. [DOI] [PubMed] [Google Scholar]
- 735.Rego A., Roley L. In-use barrier integrity of gloves: latex and nitrile superior to vinyl. Am J Infect Control. 1999;27:405–410. doi: 10.1016/s0196-6553(99)70006-4. [DOI] [PubMed] [Google Scholar]
- 736.Kotilainen H.R., Brinker J.P., Avato J.L., Gantz N.M. Latex and vinyl examination gloves. Quality control procedures and implications for health care workers. Arch Intern Med. 1989;149:2749–2753. doi: 10.1001/archinte.149.12.2749. [DOI] [PubMed] [Google Scholar]
- 737.Korniewicz D.M., Laughon B.E., Butz A., Larson E. Integrity of vinyl and latex procedure gloves. Nurs Res. 1989;38:144–146. [PubMed] [Google Scholar]
- 738.Occupational Safety and Health Administration Occupational exposure to bloodborne pathogens: final rule. 29 CFR Part 1910:1030. Federal Register. 1991;56:64003–64182. Revised 2001 CFR 66 2001:5317-25. [PubMed] [Google Scholar]
- 739.Centers for Disease Control and Prevention Recommendations for preventing the spread of vancomycin resistance: recommendations of the Hospital Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 1995;44:1–13. [PubMed] [Google Scholar]
- 740.Olsen R.J., Lynch P., Coyle M.B., Cummings J., Bokete T., Stamm W.E. Examination gloves as barriers to hand contamination in clinical practice. JAMA. 1993;270:350–353. [PubMed] [Google Scholar]
- 741.Doebbeling B.N., Pfaller M.A., Houston A.K., Wenzel R.P. Removal of nosocomial pathogens from the contaminated glove: implications for glove reuse and handwashing. Ann Intern Med. 1988;109:394–398. doi: 10.7326/0003-4819-109-5-394. [DOI] [PubMed] [Google Scholar]
- 742.Maki DG, McCormick RD, Zilz MA, et al. A MRSA outbreak in an SICU during universal precautions: new epidemiology for nosocomial MRSA [abstract 473]. Presented at the 30th Annual Meeting of the Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), Chicago, IL, October 21–24, 1990.
- 743.Boyce J.M., Jackson M.M., Pugliese G. Methicillin-resistant Staphylococcus aureus (MRSA): a briefing for acute care hospitals and nursing facilities. The AHA Technical Panel on Infections Within Hospitals. Infect Control Hosp Epidemiol. 1994;15(2):105–115. doi: 10.1086/646870. [DOI] [PubMed] [Google Scholar]
- 744.Boyce J.M., Mermel L.A., Zervos M.J. Controlling vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 1995;16:634–637. doi: 10.1086/647028. [DOI] [PubMed] [Google Scholar]
- 745.Gerding D.N., Johnson S., Peterson L.R., Mulligan M.E., Silva J., Jr. Clostridium difficile–associated diarrhea and colitis. Infect Control Hosp Epidemiol. 1995;16:459–477. doi: 10.1086/648363. [DOI] [PubMed] [Google Scholar]
- 746.Cloney D.L., Donowitz L.G. Overgrown use for infection control in nurseries and neonatal intensive care units. Am J Dis Child. 1986;140:680–683. doi: 10.1001/archpedi.1986.02140210078030. [DOI] [PubMed] [Google Scholar]
- 747.Pelke S., Ching D., Easa D., Melish M.E. Gowning does not affect colonization or infection rates in a neonatal intensive care unit. Arch Pediatr Adolesc Med. 1994;148:1016–1020. doi: 10.1001/archpedi.1994.02170100014004. [DOI] [PubMed] [Google Scholar]
- 748.Slaughter S., Hayden M.K., Nathan C. A comparison of the effect of universal use of gloves and gowns with that of glove use alone on acquisition of vancomycin-resistant enterococci in a medical intensive care unit. Ann Intern Med. 1996;125:448–456. doi: 10.7326/0003-4819-125-6-199609150-00004. [DOI] [PubMed] [Google Scholar]
- 749.Duquette-Petersen L., Francis M.E., Dohnalek L., Skinner R., Dudas P. The role of protective clothing in infection prevention in patients undergoing autologous bone marrow transplantation. Oncol Nurs Forum. 1999;26:1319–1324. [PubMed] [Google Scholar]
- 750.Sartori M., La Terra G., Aglietta M., Manzin A., Navino C., Verzetti G. Transmission of hepatitis C via blood splash into conjunctiva. Scand J Infect Dis. 1993;25:270–271. doi: 10.3109/00365549309008497. [DOI] [PubMed] [Google Scholar]
- 751.Hosoglu S., Celen M.K., Akalin S., Geyik M.F., Soyoral Y., Kara I.H. Transmission of hepatitis C by blood splash into conjunctiva in a nurse. Am J Infect Control. 2003;31:502–504. doi: 10.1016/j.ajic.2003.03.005. [DOI] [PubMed] [Google Scholar]
- 752.Centers for Disease Control and Prevention Update: human immunodeficiency virus infections in health care workers exposed to blood of infected patients. MMWR Morb Mortal Wkly Rep. 1987;36:285–289. [PubMed] [Google Scholar]
- 753.Keijman J., Tjhie J., Olde Damink S., Alink M. Unusual nosocomial transmission of Mycobacterium tuberculosis. Eur J Clin Microbiol Infect Dis. 2001;20:808–809. doi: 10.1007/s100960100606. [DOI] [PubMed] [Google Scholar]
- 754.Weaver G.H. Value of the face mask and other measures. JAMA. 1918;70:76. [Google Scholar]
- 755.Weaver G.H. Droplet infection and its prevention by the face mask. J Infect Dis. 1919;24:218–230. [Google Scholar]
- 756.Davidson I.R., Crisp A.J., Hinwood D.C., Whitaker S.C., Gregson R.H. Eye splashes during invasive vascular procedures. Br J Radiol. 1995;68:39–41. doi: 10.1259/0007-1285-68-805-39. [DOI] [PubMed] [Google Scholar]
- 757.Guidance for industry and FDA staff: surgical masks. Premarket notification [510(k)] submissions; guidance for industry and FDA. http://www.fda.gov/cdrh/ode/guidance/094.html.
- 758.National Institute for Occupational Health and Safety. Eye protection for infection control. http://www.cdc.gov/niosh/topics/eye/eye-infectious.html.
- 759.Gala C.L., Hall C.B., Schnabel K.C. The use of eye-nose goggles to control nosocomial respiratory syncytial virus infection. JAMA. 1986;256:2706–2708. [PubMed] [Google Scholar]
- 760.Agah R., Cherry J.D., Garakian A.J., Chapin M. Respiratory syncytial virus (RSV) infection rate in personnel caring for children with RSV infections: routine isolation procedure versus routine procedure supplemented by use of masks and goggles. Am J Dis Child. 1987;141:695–697. doi: 10.1001/archpedi.1987.04460060111049. [DOI] [PubMed] [Google Scholar]
- 761.Thorburn K., Kerr S., Taylor N., van Saene H.K. RSV outbreak in a paediatric intensive care unit. J Hosp Infect. 2004;57:194–201. doi: 10.1016/j.jhin.2004.03.013. [DOI] [PubMed] [Google Scholar]
- 762.http://a257.g.akamaitech.net/7/257/2422/06jun20041800/edocket.access.gpo.gov/2004/04-25183.htm.
- 763.Occupational Safety and Health Administration. Respiratory protection. www.osha.gov/dcsp/ote/trng-materials/respirators/respirators.html.
- 764.Campbell D.L., Coffey C.C., Lenhart S.W. Respiratory protection as a function of respirator fitting characteristics and fit-test accuracy. Am Ind Hyg Assoc J. 2001;62:36–44. doi: 10.1080/15298660108984607. [DOI] [PubMed] [Google Scholar]
- 765.Lee K., Slavcev A., Nicas M. Respiratory protection against Mycobacterium tuberculosis: quantitative fit test outcomes for five type N95 filtering-facepiece respirators. J Occup Environ Hyg. 2004;1:22–28. doi: 10.1080/15459620490250026. [DOI] [PubMed] [Google Scholar]
- 766.Coffey C.C., Campbell D.L., Zhuang Z. Simulated workplace performance of N95 respirators. Am Ind Hyg Assoc J. 1999;60:618–624. doi: 10.1080/00028899908984481. [DOI] [PubMed] [Google Scholar]
- 767.Coffey C.C., Lawrence R.B., Zhuang Z., Campbell D.L., Jensen P.A., Myers W.R. Comparison of five methods for fit-testing N95 filtering-facepiece respirators. Appl Occup Environ Hyg. 2002;17:723–730. doi: 10.1080/10473220290107002. [DOI] [PubMed] [Google Scholar]
- 768.National Personal Protective Technology Laboratory. Available from www.cdc.gov/niosh/npptl/. Accessed.
- 769.McGowan J.E., Jr. Nosocomial tuberculosis: new progress in control and prevention. Clin Infect Dis. 1995;21:489–505. doi: 10.1093/clinids/21.3.489. [DOI] [PubMed] [Google Scholar]
- 770.Jarvis W.R. Nosocomial transmission of multidrug-resistant Mycobacterium tuberculosis. Am J Infect Control. 1995;23:146–151. doi: 10.1016/0196-6553(95)90259-7. [DOI] [PubMed] [Google Scholar]
- 771.Centers for Disease Control and Prevention. Emergency preparedness and response. Available from wwwbtcdcgov 2003. Accessed.
- 772.Anderson J.D., Bonner M., Scheifele D.W., Schneider B.C. Lack of nosocomial spread of varicella in a pediatric hospital with negative-pressure ventilated patient rooms. Infect Control. 1985;6:120–121. doi: 10.1017/s0195941700062792. [DOI] [PubMed] [Google Scholar]
- 773.Brunell P.A., Wood D. Varicella serological status of healthcare workers as a guide to whom to test or immunize. Infect Control Hosp Epidemiol. 1999;20:355–357. doi: 10.1086/501635. [DOI] [PubMed] [Google Scholar]
- 774.Saiman L., LaRussa P., Steinberg S.P. Persistence of immunity to varicella-zoster virus after vaccination of healthcare workers. Infect Control Hosp Epidemiol. 2001;22:279–283. doi: 10.1086/501900. [DOI] [PubMed] [Google Scholar]
- 775.Willy M.E., Koziol D.E., Fleisher T. Measles immunity in a population of healthcare workers. Infect Control Hosp Epidemiol. 1994;15:12–17. doi: 10.1086/646811. [DOI] [PubMed] [Google Scholar]
- 776.Wright L.J., Carlquist J.F. Measles immunity in employees of a multihospital healthcare provider. Infect Control Hosp Epidemiol. 1994;15:8–11. doi: 10.1086/646810. [DOI] [PubMed] [Google Scholar]
- 777.Centers for Disease Control and Prevention Updated US Public Health Service Guidelines for the management of occupational exposures to HBV, HCV, and HIV. MMWR Recomm Rep. 2001;50:1–52. [PubMed] [Google Scholar]
- 778.Do A.N., Ciesielski C.A., Metler R.P., Hammett T.A., Li J., Fleming P.L. Occupationally acquired human immunodeficiency virus (HIV) infection: national case surveillance data during 20 years of the HIV epidemic in the United States. Infect Control Hosp Epidemiol. 2003;24:86–96. doi: 10.1086/502178. [DOI] [PubMed] [Google Scholar]
- 779.Centers for Disease Control and Prevention Update: universal precautions for prevention of transmission of human immunodeficiency virus, hepatitis B virus, and other bloodborne pathogens in health-care settings. MMWR Morb Mortal Wkly Rep. 1988;37:377–388. [PubMed] [Google Scholar]
- 780.Davis M.S. Occupational hazards of operating: opportunities for improvement. Infect Control Hosp Epidemiol. 1996;17:691–693. doi: 10.1086/647205. [DOI] [PubMed] [Google Scholar]
- 781.Gerberding J.L. Procedure-specific infection control for preventing intraoperative blood exposures. Am J Infect Control. 1993;21:364–367. doi: 10.1016/0196-6553(93)90403-q. [DOI] [PubMed] [Google Scholar]
- 782.Fry D.E., Telford G.L., Fecteau D.L., Sperling R.S., Meyer A.A. Prevention of blood exposure: body and facial protection. Surg Clin North Am. 1995;75:1141–1157. doi: 10.1016/s0039-6109(16)46787-3. [DOI] [PubMed] [Google Scholar]
- 783.Hansen M.E. Bloodborne pathogens and procedure safety in interventional radiology. Semin Ultrasound CT MR. 1998;19:209–214. doi: 10.1016/s0887-2171(98)90062-2. [DOI] [PubMed] [Google Scholar]
- 784.Holodnick C.L., Barkauskas V. Reducing percutaneous injuries in the OR by educational methods. AORN J. 2000;72:461–476. doi: 10.1016/s0001-2092(06)61278-7. [DOI] [PubMed] [Google Scholar]
- 785.www.osha.gov/SLTC/bloodbornepathogens/index.html.
- 786.www.cdc.gov/niosh/2000-108.html.
- 787.National Insititute for Occupational Health and Safety. Safer medical device implementation in health care facilities. http://www.cdc.gov/niosh/topics/bbp/safer/.
- 788.Centers for Disease Control and Prevention. Available from www.cdc.gov/sharpssafety/resources.html.
- 789.Catanzaro A. Nosocomial tuberculosis. Am Rev Respir Dis. 1982;125:559–562. doi: 10.1164/arrd.1982.125.5.559. [DOI] [PubMed] [Google Scholar]
- 790.Cepeda J.A., Whitehouse T., Cooper B. Isolation of patients in single rooms or cohorts to reduce spread of MRSA in intensive-care units: prospective two-centre study. Lancet. 2005;365:295–304. doi: 10.1016/S0140-6736(05)17783-6. [DOI] [PubMed] [Google Scholar]
- 791.Mulin B., Rouget C., Clement C. Association of private isolation rooms with ventilator-associated Acinetobacter baumanii pneumonia in a surgical intensive-care unit. Infect Control Hosp Epidemiol. 1997;18:499–503. doi: 10.1086/647655. [DOI] [PubMed] [Google Scholar]
- 792.www.aia.org/aah_gd_hospcons.
- 793.Raad I., Abbas J., Whimbey E. Infection control of nosocomial respiratory viral disease in the immunocompromised host. Am J Med. 1997;102:48–52. doi: 10.1016/s0002-9343(97)00011-9. [DOI] [PubMed] [Google Scholar]
- 794.Isaacs D., Dickson H., O'Callaghan C., Sheaves R., Winter A., Moxon E.R. Handwashing and cohorting in prevention of hospital acquired infections with respiratory syncytial virus. Arch Dis Child. 1991;66:227–231. doi: 10.1136/adc.66.2.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 795.Chang V.T., Nelson K. The role of physical proximity in nosocomial diarrhea. Clin Infect Dis. 2000;31:717–722. doi: 10.1086/314030. [DOI] [PubMed] [Google Scholar]
- 796.Byers K.E., Anglim A.M., Anneski C.J. A hospital epidemic of vancomycin-resistant Enterococcus: risk factors and control. Infect Control Hosp Epidemiol. 2001;22:140–147. doi: 10.1086/501880. [DOI] [PubMed] [Google Scholar]
- 797.Dassut B. The implementation of a commode cleaning and identification system. Nurs Times. 2004;100:47. [PubMed] [Google Scholar]
- 798.Mayer R.A., Geha R.C., Helfand M.S., Hoyen C.K., Salata R.A., Donskey C.J. Role of fecal incontinence in contamination of the environment with vancomycin-resistant enterococci. Am J Infect Control. 2003;31:221–225. doi: 10.1067/mic.2003.45. [DOI] [PubMed] [Google Scholar]
- 799.Samore M.H., Venkataraman L., DeGirolami P.C., Arbeit R.D., Karchmer A.W. Clinical and molecular epidemiology of sporadic and clustered cases of nosocomial Clostridium difficile diarrhea. Am J Med. 1996;100:32–40. doi: 10.1016/s0002-9343(96)90008-x. [DOI] [PubMed] [Google Scholar]
- 800.Clabots C.R., Johnson S., Olson M.M., Peterson L.R., Gerding D.N. Acquisition of Clostridium difficile by hospitalized patients: evidence for colonized new admissions as a source of infection. J Infect Dis. 1992;166:561–567. doi: 10.1093/infdis/166.3.561. [DOI] [PubMed] [Google Scholar]
- 801.Samore M.H. Epidemiology of nosocomial Clostridium difficile diarrhoea. J Hosp Infect. 1999;43(Suppl):S183–S190. doi: 10.1016/s0195-6701(99)90085-3. [DOI] [PubMed] [Google Scholar]
- 802.Tokars J.I., Satake S., Rimland D. The prevalence of colonization with vancomycin-resistant Enterococcus at a Veterans' Affairs institution. Infect Control Hosp Epidemiol. 1999;20:171–175. doi: 10.1086/501606. [DOI] [PubMed] [Google Scholar]
- 803.Cone R., Mohan K., Thouless M., Corey L. Nosocomial transmission of rotavirus infection. Pediatr Infect Dis J. 1988;7:103–109. doi: 10.1097/00006454-198802000-00008. [DOI] [PubMed] [Google Scholar]
- 804.Bruce B.B., Blass M.A., Blumberg H.M., Lennox J.L., del Rio C., Horsburgh C.R., Jr. Risk of Cryptosporidium parvum transmission between hospital roommates. Clin Infect Dis. 2000;31:947–950. doi: 10.1086/318147. [DOI] [PubMed] [Google Scholar]
- 805.Ford-Jones E.L., Mindorff C.M., Gold R., Petric M. The incidence of viral-associated diarrhea after admission to a pediatric hospital. Am J Epidemiol. 1990;131:711–718. doi: 10.1093/oxfordjournals.aje.a115555. [DOI] [PubMed] [Google Scholar]
- 806.Murray-Leisure K.A., Geib S., Graceley D. Control of epidemic methicillin-resistant Staphylococcus aureus. Infect Control Hosp Epidemiol. 1990;11:343–350. doi: 10.1086/646185. [DOI] [PubMed] [Google Scholar]
- 807.Jochimsen E.M., Fish L., Manning K. Control of vancomycin-resistant enterococci at a community hospital: efficacy of patient and staff cohorting. Infect Control Hosp Epidemiol. 1999;20:106–109. doi: 10.1086/501598. [DOI] [PubMed] [Google Scholar]
- 808.Sample M.L., Gravel D., Oxley C., Toye B., Garber G., Ramotar K. An outbreak of vancomycin-resistant enterococci in a hematology-oncology unit: control by patient cohorting and terminal cleaning of the environment. Infect Control Hosp Epidemiol. 2002;23:468–470. doi: 10.1086/502088. [DOI] [PubMed] [Google Scholar]
- 809.Podnos Y.D., Cinat M.E., Wilson S.E., Cooke J., Gornick W., Thrupp L.D. Eradication of multidrug-resistant Acinetobacter from an intensive care unit. Surg Infect (Larchmt) 2001;2:297–301. doi: 10.1089/10962960152813331. [DOI] [PubMed] [Google Scholar]
- 810.Graham P.L., 3rd, Morel A.S., Zhou J. Epidemiology of methicillin-susceptible Staphylococcus aureus in the neonatal intensive care unit. Infect Control Hosp Epidemiol. 2002;23:677–682. doi: 10.1086/501993. [DOI] [PubMed] [Google Scholar]
- 811.Doherty J.A., Brookfield D.S., Gray J., McEwan R.A. Cohorting of infants with respiratory syncytial virus. J Hosp Infect. 1998;38:203–206. doi: 10.1016/s0195-6701(98)90275-4. [DOI] [PubMed] [Google Scholar]
- 812.Hall C.B., Geiman J.M., Douglas R.G., Jr., Meagher M.P. Control of nosocomial respiratory syncytial viral infections. Pediatrics. 1978;62:728–732. [PubMed] [Google Scholar]
- 813.Buffington J., Chapman L.E., Stobierski M.G. Epidemic keratoconjunctivitis in a chronic care facility: risk factors and measures for control. J Am Geriatr Soc. 1993;41:1177–1181. doi: 10.1111/j.1532-5415.1993.tb07299.x. [DOI] [PubMed] [Google Scholar]
- 814.Grehn M., Kunz J., Sigg P., Slongo R., Zbinden R. Nosocomial rotavirus infections in neonates: means of prevention and control. J Perinat Med. 1990;18:369–374. doi: 10.1515/jpme.1990.18.5.369. [DOI] [PubMed] [Google Scholar]
- 815.Tan Y.M., Chow P.K., Tan B.H. Management of inpatients exposed to an outbreak of severe acute respiratory syndrome (SARS) J Hosp Infect. 2004;58:210–215. doi: 10.1016/j.jhin.2004.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 816.Talon D., Vichard P., Muller A., Bertin M., Jeunet L., Bertrand X. Modelling the usefulness of a dedicated cohort facility to prevent the dissemination of MRSA. J Hosp Infect. 2003;54:57–62. doi: 10.1016/s0195-6701(03)00086-0. [DOI] [PubMed] [Google Scholar]
- 817.Hotchkiss J.R., Strike D.G., Simonson D.A., Broccard A.F., Crooke P.S. An agent-based and spatially explicit model of pathogen dissemination in the intensive care unit. Crit Care Med. 2005;33:168–176. doi: 10.1097/01.ccm.0000150658.05831.d2. [DOI] [PubMed] [Google Scholar]
- 818.Austin D.J., Bonten M.J., Weinstein R.A., Slaughter S., Anderson R.M. Vancomycin-resistant enterococci in intensive-care hospital settings: transmission dynamics, persistence, and the impact of infection control programs. Proc Natl Acad Sci U S A. 1999;96:6908–6913. doi: 10.1073/pnas.96.12.6908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 819.Kovner C.T., Harrington C. Counting nurses: data show many nursing homes to be short-staffed. Am J Nurs. 2000;100:53–54. doi: 10.1097/00000446-200009000-00039. [DOI] [PubMed] [Google Scholar]
- 820.Mueller C. Staffing problems in long-term care: let's do something about it! J Gerontol Nurs. 2003;29:3–4. doi: 10.3928/0098-9134-20030301-03. [DOI] [PubMed] [Google Scholar]
- 821.Stats & facts: nursing staff shortages in long-term care facilities. Manag Care Interface. 2000;13:46–47. [PubMed] [Google Scholar]
- 822.Mejias A., Chavez-Bueno S., Ramilo O. Human metapneumovirus: a not-so-new virus. Pediatr Infect Dis J. 2004;23:1–7. doi: 10.1097/01.inf.0000105103.60288.0e. [DOI] [PubMed] [Google Scholar]
- 823.Iwane M.K., Edwards K.M., Szilagyi P.G. Population-based surveillance for hospitalizations associated with respiratory syncytial virus, influenza virus, and parainfluenza viruses among young children. Pediatrics. 2004;113:1758–1764. doi: 10.1542/peds.113.6.1758. [DOI] [PubMed] [Google Scholar]
- 824.Ong G.M., Wyatt D.E., O'Neill H.J., McCaughey C., Coyle P.V. A comparison of nested polymerase chain reaction and immunofluorescence for the diagnosis of respiratory infections in children with bronchiolitis, and the implications for a cohorting strategy. J Hosp Infect. 2001;49:122–128. doi: 10.1053/jhin.2001.1044. [DOI] [PubMed] [Google Scholar]
- 825.von Linstow M.L., Larsen H.H., Eugen-Olsen J. Human metapneumovirus and respiratory syncytial virus in hospitalized Danish children with acute respiratory tract infection. Scand J Infect Dis. 2004;36:578–584. doi: 10.1080/00365540410018166. [DOI] [PubMed] [Google Scholar]
- 826.Gehanno J.F., Pestel-Caron M., Nouvellon M., Caillard J.F. Nosocomial pertussis in healthcare workers from a pediatric emergency unit in France. Infect Control Hosp Epidemiol. 1999;20:549–552. doi: 10.1086/501667. [DOI] [PubMed] [Google Scholar]
- 827.www.cdc.gov/flu/professionals/infectioncontrol/resphygiene.htm.
- 828.Edlin B.R., Tokars J.I., Grieco M.H. An outbreak of multidrug-resistant tuberculosis among hospitalized patients with the acquired immunodeficiency syndrome. N Engl J Med. 1992;326:1514–1521. doi: 10.1056/NEJM199206043262302. [DOI] [PubMed] [Google Scholar]
- 829.Centers for Disease Control and Prevention Update: severe acute respiratory syndrome, Toronto, Canada, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:547–550. [PubMed] [Google Scholar]
- 830.Starke J.R. Transmission of Mycobacterium tuberculosis to and from children and adolescents. Semin Pediatr Infect Dis. 2001;12:115–123. [Google Scholar]
- 831.Saiman L., Macdonald N., Burns J.L., Hoiby N., Speert D.P., Weber D. Infection control in cystic fibrosis: practical recommendations for the hospital, clinic, and social settings. Am J Infect Control. 2000;28:381–385. doi: 10.1067/mic.2000.106337. [DOI] [PubMed] [Google Scholar]
- 832.24th ed. American Academy of Pediatrics; Elk Grove Village (IL): 2003. American Academy of Pediatrics. Red Book 2003 Report of the Committee on Infectious Diseases. [Google Scholar]
- 833.Lau J.T., Lau M., Kim J.H., Tsui H.Y., Tsang T., Wong T.W. Probable secondary infections in households of SARS patients in Hong Kong. Emerg Infect Dis. 2004;10:235–243. doi: 10.3201/eid1002.030626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 834.Hota B. Contamination, disinfection, and cross-colonization: are hospital surfaces reservoirs for nosocomial infection? Clin Infect Dis. 2004;39:1182–1189. doi: 10.1086/424667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 835.Rutala W.A., Weber D.J. Disinfection and sterilization in health care facilities: what clinicians need to know. Clin Infect Dis. 2004;39:702–709. doi: 10.1086/423182. [DOI] [PubMed] [Google Scholar]
- 836.Boyce J.M., Opal S.M., Chow J.W. Outbreak of multidrug-resistant Enterococcus faecium with transferable vanB class vancomycin resistance. J Clin Microbiol. 1994;32:1148–1153. doi: 10.1128/jcm.32.5.1148-1153.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 837.Engelhart S., Krizek L., Glasmacher A., Fischnaller E., Marklein G., Exner M. Pseudomonas aeruginosa outbreak in a haematology-oncology unit associated with contaminated surface cleaning equipment. J Hosp Infect. 2002;52:93–98. doi: 10.1053/jhin.2002.1279. [DOI] [PubMed] [Google Scholar]
- 838.Denton M., Wilcox M.H., Parnell P. Role of environmental cleaning in controlling an outbreak of Acinetobacter baumannii on a neurosurgical intensive care unit. J Hosp Infect. 2004;56:106–110. doi: 10.1016/j.jhin.2003.10.017. [DOI] [PubMed] [Google Scholar]
- 839.Hollyoak V., Allison D., Summers J. Pseudomonas aeruginosa wound infection associated with a nursing home's whirlpool bath. Commun Dis Rep CDR Rev. 1995;5:R100–R102. [PubMed] [Google Scholar]
- 840.Malik R.E., Cooper R.A., Griffith C.J. Use of audit tools to evaluate the efficacy of cleaning systems in hospitals. Am J Infect Control. 2003;31:181–187. doi: 10.1067/mic.2003.34. [DOI] [PubMed] [Google Scholar]
- 841.Ansari S.A., Springthorpe V.S., Sattar S.A. Survival and vehicular spread of human rotaviruses: possible relation to seasonality of outbreaks. Rev Infect Dis. 1991;13:448–461. doi: 10.1093/clinids/13.3.448. [DOI] [PubMed] [Google Scholar]
- 842.Kaatz G.W., Gitlin S.D., Schaberg D.R. Acquisition of Clostridium difficile from the hospital environment. Am J Epidemiol. 1988;127:1289–1294. doi: 10.1093/oxfordjournals.aje.a114921. [DOI] [PubMed] [Google Scholar]
- 843.Mayfield J.L., Leet T., Miller J., Mundy L.M. Environmental control to reduce transmission of Clostridium difficile. Clin Infect Dis. 2000;31:995–1000. doi: 10.1086/318149. [DOI] [PubMed] [Google Scholar]
- 844.Dennehy P.H. Transmission of rotavirus and other enteric pathogens in the home. Pediatr Infect Dis J. 2000;19(10 Suppl):S103–S105. doi: 10.1097/00006454-200010001-00003. [DOI] [PubMed] [Google Scholar]
- 845.Dennehy P. Rotavirus infections in infection control reference service. In: Abrutyn E., Goldmann D., Scheckler W., editors. Saunders; Philadelphia: 2001. pp. 821–823. [Google Scholar]
- 846.Wilcox M.H., Fawley W.N., Wigglesworth N., Parnell P., Verity P., Freeman J. Comparison of the effect of detergent versus hypochlorite cleaning on environmental contamination and incidence of Clostridium difficile infection. J Hosp Infect. 2003;54:109–114. doi: 10.1016/s0195-6701(02)00400-0. [DOI] [PubMed] [Google Scholar]
- 847.Rutala WA, Weber DJ, Healthcare Infection Control Practices Advisory Committee Committee (HICPAC). Guideline for disinfection and sterilization in health care facilities. 2007. in press.
- 848.Bernards A.T., Harinck H.I., Dijkshoorn L., van der Reijden T.J., van den Broek P.J. Persistent Acinetobacter baumannii? Look inside your medical equipment. Infect Control Hosp Epidemiol. 2004;25:1002–1004. doi: 10.1086/502335. [DOI] [PubMed] [Google Scholar]
- 849.Neely A.N., Weber J.M., Daviau P. Computer equipment used in patient care within a multihospital system: recommendations for cleaning and disinfection. Am J Infect Control. 2005;33:233–237. doi: 10.1016/j.ajic.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 850.Neely A.N., Maley M.P., Warden G.D. Computer keyboards as reservoirs for Acinetobacter baumannii in a burn hospital. Clin Infect Dis. 1999;29:1358–1360. doi: 10.1086/313463. [DOI] [PubMed] [Google Scholar]
- 851.Bures S., Fishbain J.T., Uyehara C.F., Parker J.M., Berg B.W. Computer keyboards and faucet handles as reservoirs of nosocomial pathogens in the intensive care unit. Am J Infect Control. 2000;28:465–471. doi: 10.1067/mic.2000.107267. [DOI] [PubMed] [Google Scholar]
- 852.Brooks S., Khan A., Stoica D. Reduction in vancomycin-resistant Enterococcus and Clostridium difficile infections following change to tympanic thermometers. Infect Control Hosp Epidemiol. 1998;19:333–336. doi: 10.1086/647824. [DOI] [PubMed] [Google Scholar]
- 853.Jernigan J.A., Siegman-Igra Y., Guerrant R.C., Farr B.M. A randomized crossover study of disposable thermometers for prevention of Clostridium difficile and other nosocomial infections. Infect Control Hosp Epidemiol. 1998;19:494–499. doi: 10.1086/647855. [DOI] [PubMed] [Google Scholar]
- 854.Weinstein S.A., Gantz N.M., Pelletier C., Hibert D. Bacterial surface contamination of patients' linen: isolation precautions versus standard care. Am J Infect Control. 1989;17:264–267. doi: 10.1016/0196-6553(89)90173-9. [DOI] [PubMed] [Google Scholar]
- 855.Pugliese G. Isolating and double-bagging laundry: is it really necessary? Health Facil Manag. 1989;2:16–21. [PubMed] [Google Scholar]
- 856.Available from. Accessed .
- 857.Kiehl E., Wallace R., Warren C. Tracking perinatal infection: is it safe to launder your scrubs at home? MCN Am J Matern Child Nurs. 1997;22:195–197. doi: 10.1097/00005721-199707000-00007. [DOI] [PubMed] [Google Scholar]
- 858.Jurkovich P. Home- versus hospital-laundered scrubs: a pilot study. MCN Am J Matern Child Nurs. 2004;29:106–110. doi: 10.1097/00005721-200403000-00009. [DOI] [PubMed] [Google Scholar]
- 859.US Environmental Protection Agency. Medical waste. Available from www.epa.gov/epaoswer/other/medical/. [PubMed]
- 860.www.cdc.gov/ncidod/dhqp/gl_environinfection.html.
- 861.Maki D.G., Alvarado C., Hassemer C. Double-bagging of items from isolation rooms is unnecessary as an infection control measure: a comparative study of surface contamination with single- and double-bagging. Infect Control. 1986;7:535–537. doi: 10.1017/s0195941700065279. [DOI] [PubMed] [Google Scholar]
- 862.Centers for Disease Control and Prevention Recommended antimicrobial agents for the treatment and postexposure prophylaxis of pertussis: 2005 CDC guidelines. MMWR Recomm Rep. 2005;54:1–16. [PubMed] [Google Scholar]
- 863.Centers for Disease Control and Prevention Prevention and control of meningococcal disease: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2000;49:1–10. [PubMed] [Google Scholar]
- 864.Centers for Disease Control and Prevention Notice to readers: additional options for preventive treatment for persons exposed to inhalational anthrax. MMWR Morb Mortal Wkly Rep. 2001;50:1142–1151. [Google Scholar]
- 865.Centers for Disease Control and Prevention Updated US Public Health Service guidelines for the management of occupational exposures to HIV and recommendations for postexposure prophylaxis. MMWR Recomm Rep. 2005;54:1–17. [PubMed] [Google Scholar]
- 866.Boyce J.M. MRSA patients: proven methods to treat colonization and infection. J Hosp Infect. 2001;48(Suppl A):S9–S14. doi: 10.1016/s0195-6701(01)90005-2. [DOI] [PubMed] [Google Scholar]
- 867.Barrett F.F., Mason E.O., Jr., Fleming D. Brief clinical and laboratory observations. J Pediatr. 1979;94:796–800. doi: 10.1016/s0022-3476(79)80159-6. [DOI] [PubMed] [Google Scholar]
- 868.4th ed. American Academy of Pediatrics; Elk Grove Village (IL): 2002. American Academy of Pediatrics and American Academy of Obstetricians and Gynecologists. Guidelines for perinatal care. [Google Scholar]
- 869.Centers for Disease Control and Prevention. Management of multidrug-resistant organisms in health care settings, 2006. Available from. Accessed.
- 870.Kallen A.J., Wilson C.T., Larson R.J. Perioperative intranasal mupirocin for the prevention of surgical-site infections: systematic review of the literature and meta-analysis. Infect Control Hosp Epidemiol. 2005;26:916–922. doi: 10.1086/505453. [DOI] [PubMed] [Google Scholar]
- 871.Carrier M., Marchand R., Auger P. Methicillin-resistant Staphylococcus aureus infection in a cardiac surgical unit. J Thorac Cardiovasc Surg. 2002;123:40–44. doi: 10.1067/mtc.2002.118505. [DOI] [PubMed] [Google Scholar]
- 872.Tacconelli E., Carmeli Y., Aizer A., Ferreira G., Foreman M.G., D'Agata E.M. Mupirocin prophylaxis to prevent Staphylococcus aureus infection in patients undergoing dialysis: a meta-analysis. Clin Infect Dis. 2003;37:1629–1638. doi: 10.1086/379715. [DOI] [PubMed] [Google Scholar]
- 873.Centers for Disease Control and Prevention Immunization of health-care workers: recommendations of the Advisory Committee on Immunization Practices (ACIP) and the Hospital Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 1997;46:1–42. [PubMed] [Google Scholar]
- 874.Mahoney F.J., Stewart K., Hu H., Coleman P., Alter M.J. Progress toward the elimination of hepatitis B virus transmission among health care workers in the United States. Arch Intern Med. 1997;157:2601–2605. [PubMed] [Google Scholar]
- 875.Gladstone J.L., Millian S.J. Rubella exposure in an obstetric clinic. Obstet Gynecol. 1981;57:182–186. [PubMed] [Google Scholar]
- 876.Wilde J.A., McMillan J.A., Serwint J., Butta J., O'Riordan M.A., Steinhoff M.C. Effectiveness of influenza vaccine in health care professionals: a randomized trial. JAMA. 1999;281:908–913. doi: 10.1001/jama.281.10.908. [DOI] [PubMed] [Google Scholar]
- 877.Potter J., Stott D.J., Roberts M.A. Influenza vaccination of health care workers in long-term care hospitals reduces the mortality of elderly patients. J Infect Dis. 1997;175:1–6. doi: 10.1093/infdis/175.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 878.Pearson M.L., Bridges C.B., Harper S.A. Influenza vaccination of health-care personnel: recommendations of the Healthcare Infection Control Practices Advisory Committee (HICPAC) and the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2006;55:1–16. [PubMed] [Google Scholar]
- 879.Wright S.W., Decker M.D., Edwards K.M. Incidence of pertussis infection in healthcare workers. Infect Control Hosp Epidemiol. 1999;20:120–123. doi: 10.1086/501593. [DOI] [PubMed] [Google Scholar]
- 880.Calugar A., Ortega-Sanchez I.R., Tiwari T., Oakes L., Jahre J.A., Murphy T.V. Nosocomial pertussis: costs of an outbreak and benefits of vaccinating health care workers. Clin Infect Dis. 2006;42:981–988. doi: 10.1086/500321. [DOI] [PubMed] [Google Scholar]
- 881.www.fda.gov.
- 882.Campins-Marti M., Cheng H.K., Forsyth K. Recommendations are needed for adolescent and adult pertussis immunisation: rationale and strategies for consideration. Vaccine. 2001;20:641–646. doi: 10.1016/s0264-410x(01)00393-0. [DOI] [PubMed] [Google Scholar]
- 883.www.cdc.gov/nip/recs/provisional_recs/default.htm.
- 884.Centers for Disease Control and Prevention Recommended childhood and adolescent immunization schedule, United States, 2006. MMWR Morb Mortal Wkly Rep. 2006;54:Q1–Q4. [PubMed] [Google Scholar]
- 885.Centers for Disease Control and Prevention Recommended childhood and adolescent immunization schedule, United States, 2006. Pediatrics. 2006;117:239–240. doi: 10.1542/peds.2005-2790. [DOI] [PubMed] [Google Scholar]
- 886.Centers for Disease Control and Prevention Recommended adult immunization schedule, United States, October 2005–September 2006. MMWR Morb Mortal Wkly Rep. 2005;54:Q1–Q4. [PubMed] [Google Scholar]
- 887.Centers for Disease Control and Prevention Prevention of varicella: updated recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 1999;48:1–5. [PubMed] [Google Scholar]
- 888.McKenney D., Pouliot K.L., Wang Y. Broadly protective vaccine for Staphylococcus aureus based on an in vivo-expressed antigen. Science. 1999;284:1523–1527. doi: 10.1126/science.284.5419.1523. [DOI] [PubMed] [Google Scholar]
- 889.Shinefield H., Black S., Fattom A. Use of a Staphylococcus aureus conjugate vaccine in patients receiving hemodialysis. N Engl J Med. 2002;346:491–496. doi: 10.1056/NEJMoa011297. [DOI] [PubMed] [Google Scholar]
- 890.Abadesso C., Almeida H.I., Virella D., Carreiro M.H., Machado M.C. Use of palivizumab to control an outbreak of syncytial respiratory virus in a neonatal intensive care unit. J Hosp Infect. 2004;58:38–41. doi: 10.1016/j.jhin.2004.04.024. [DOI] [PubMed] [Google Scholar]
- 891.George R.H., Gully P.R., Gill O.N., Innes J.A., Bakhshi S.S., Connolly M. An outbreak of tuberculosis in a children's hospital. J Hosp Infect. 1986;8:129–142. doi: 10.1016/0195-6701(86)90039-3. [DOI] [PubMed] [Google Scholar]
- 892.Simor A.E., Lee M., Vearncombe M. An outbreak due to multiresistant Acinetobacter baumannii in a burn unit: risk factors for acquisition and management. Infect Control Hosp Epidemiol. 2002;23:261–267. doi: 10.1086/502046. [DOI] [PubMed] [Google Scholar]
- 893.Puzniak L.A., Leet T., Mayfield J., Kollef M., Mundy L.M. To gown or not to gown: the effect on acquisition of vancomycin-resistant enterococci. Clin Infect Dis. 2002;35:18–25. doi: 10.1086/340739. [DOI] [PubMed] [Google Scholar]
- 894.Hanna H., Umphrey J., Tarrand J., Mendoza M., Raad I. Management of an outbreak of vancomycin-resistant enterococci in the medical intensive care unit of a cancer center. Infect Control Hosp Epidemiol. 2001;22:217–219. doi: 10.1086/501892. [DOI] [PubMed] [Google Scholar]
- 895.Centers for Disease Control and Prevention Recommendations for preventing transmission of infection with human T-lymphotropic virus type III/lymphadenopathy-associated virus in the workplace. MMWR Morb Mortal Wkly Rep. 1985;34:681–695. [PubMed] [Google Scholar]
- 896.Centers for Disease Control and Prevention Severe acute respiratory syndrome, Taiwan, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:461–466. [PubMed] [Google Scholar]
- 897.Capps J.A. Measures for the prevention and control of respiratory infections in military camps. JAMA. 1918;71:448–451. [Google Scholar]
- 898.Thomas C. Efficiency of surgical masks in use in hospital wards: report to the Control of Infection Subcommittee. Guys Hosp Rep. 1961;110:157–167. [Google Scholar]
- 899.Beck M., Antle B.J., Berlin D. Wearing masks in a pediatric hospital: developing practical guidelines. Can J Public Health. 2004;95:256–257. doi: 10.1007/BF03405126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 900.Ryan M.A., Christian R.S., Wohlrabe J. Handwashing and respiratory illness among young adults in military training. Am J Prev Med. 2001;21:79–83. doi: 10.1016/s0749-3797(01)00323-3. [DOI] [PubMed] [Google Scholar]
- 901.Roberts L., Smith W., Jorm L., Patel M., Douglas R.M., McGilchrist C. Effect of infection control measures on the frequency of upper respiratory infection in child care: a randomized, controlled trial. Pediatrics. 2000;105(4 Pt 1):738–742. doi: 10.1542/peds.105.4.738. [DOI] [PubMed] [Google Scholar]
- 902.White C., Kolble R., Carlson R. The effect of hand hygiene on illness rate among students in university residence halls. Am J Infect Control. 2003;31:364–370. doi: 10.1016/s0196-6553(03)00041-5. [DOI] [PubMed] [Google Scholar]
- 903.Aiello A.E., Larson E.L. What is the evidence for a causal link between hygiene and infections? Lancet Infect Dis. 2002;2:103–110. doi: 10.1016/s1473-3099(02)00184-6. [DOI] [PubMed] [Google Scholar]
- 904.American Association of Nurse Anesthesists. Reuse of needles and syringes by healthcare providers put patients at risk. Available from www.aana.com/news.aspx?ucNavMenu_TSMenuTargetID=171&ucNavMenu_TSMenuTargetType=4&ucNavMenu_TSMenuID=6&id=1613. Accessed November 10, 2007.
- 905.Watanakunakorn C., Stahl C. Streptococcus salivarius meningitis following myelography. Infect Control Hosp Epidemiol. 1992;13:454. doi: 10.1086/646571. [DOI] [PubMed] [Google Scholar]
- 906.Gelfand M.S., Abolnik I.Z. Streptococcal meningitis complicating diagnostic myelography: three cases and review. Clin Infect Dis. 1995;20:582–587. doi: 10.1093/clinids/20.3.582. [DOI] [PubMed] [Google Scholar]
- 907.Schlesinger J.J., Salit I.E., McCormack G. Streptococcal meningitis after myelography. Arch Neurol. 1982;39:576–577. doi: 10.1001/archneur.1982.00510210046010. [DOI] [PubMed] [Google Scholar]
- 908.Yaniv L.G., Potasman I. Iatrogenic meningitis: an increasing role for resistant viridans streptococci? Case report and review of the last 20 years. Scand J Infect Dis. 2000;32:693–696. doi: 10.1080/003655400459658. [DOI] [PubMed] [Google Scholar]
- 909.Schlegel L., Merlet C., Laroche J.M., Fremaux A., Geslin P. Iatrogenic meningitis due to Abiotrophia defectiva after myelography. Clin Infect Dis. 1999;28:155–156. doi: 10.1086/517189. [DOI] [PubMed] [Google Scholar]
- 910.Schneeberger P.M., Janssen M., Voss A. Alpha-hemolytic streptococci: a major pathogen of iatrogenic meningitis following lumbar puncture. Case reports and a review of the literature. Infection. 1996;24:29–33. doi: 10.1007/BF01780647. [DOI] [PubMed] [Google Scholar]
- 911.Veringa E., van Belkum A., Schellekens H. Iatrogenic meningitis by Streptococcus salivarius following lumbar puncture. J Hosp Infect. 1995;29:316–318. doi: 10.1016/0195-6701(95)90283-x. [DOI] [PubMed] [Google Scholar]
- 912.Couzigou C., Vuong T.K., Botherel A.H., Aggoune M., Astagneau P. Iatrogenic Streptococcus salivarius meningitis after spinal anaesthesia: need for strict application of standard precautions. J Hosp Infect. 2003;53:313–314. doi: 10.1053/jhin.2002.1366. [DOI] [PubMed] [Google Scholar]
- 913.Torres E., Alba D., Frank A., Diez-Tejedor E. Iatrogenic meningitis due to Streptococcus salivarius following a spinal tap. Clin Infect Dis. 1993;17:525–526. doi: 10.1093/clinids/17.3.525. [DOI] [PubMed] [Google Scholar]
- 914.Trautmann M., Lepper P.M., Schmitz F.J. Three cases of bacterial meningitis after spinal and epidural anesthesia. Eur J Clin Microbiol Infect Dis. 2002;21:43–45. doi: 10.1007/s10096-001-0643-7. [DOI] [PubMed] [Google Scholar]
- 915.Baer E.T. Iatrogenic meningitis: the case for face masks. Clin Infect Dis. 2000;31:519–521. doi: 10.1086/313991. [DOI] [PubMed] [Google Scholar]
- 916.Black S.R., Weinstein R.A. The case for face masks: zorro or zero? Clin Infect Dis. 2000;31:522–523. doi: 10.1086/313956. [DOI] [PubMed] [Google Scholar]
- 917.Philips B.J., Fergusson S., Armstrong P., Anderson F.M., Wildsmith J.A. Surgical face masks are effective in reducing bacterial contamination caused by dispersal from the upper airway. Br J Anaesth. 1992;69:407–408. doi: 10.1093/bja/69.4.407. [DOI] [PubMed] [Google Scholar]
- 918.Centers for Disease Control and Prevention Guidelines for the prevention of intravascular catheter–related infections. MMWR Recomm Rep. 2002;51:1–26. [PubMed] [Google Scholar]
- 919.Catalano G., Houston S.H., Catalano M.C. Anxiety and depression in hospitalized patients in resistant organism isolation. South Med J. 2003;96:141–145. doi: 10.1097/01.SMJ.0000050683.36014.2E. [DOI] [PubMed] [Google Scholar]
- 920.Tarzi S., Kennedy P., Stone S., Evans M. Methicillin-resistant Staphylococcus aureus: psychological impact of hospitalization and isolation in an older adult population. J Hosp Infect. 2001;49:250–254. doi: 10.1053/jhin.2001.1098. [DOI] [PubMed] [Google Scholar]
- 921.Kelly-Rossini L., Perlman D.C., Mason D.J. The experience of respiratory isolation for HIV-infected persons with tuberculosis. J Assoc Nurses AIDS Care. 1996;7:29–36. doi: 10.1016/S1055-3290(96)80035-5. [DOI] [PubMed] [Google Scholar]
- 922.Knowles H.E. The experience of infectious patients in isolation. Nurs Times. 1993;89:53–56. [PubMed] [Google Scholar]
- 923.Evans H.L., Shaffer M.M., Hughes M.G. Contact isolation in surgical patients: a barrier to care? Surgery. 2003;134:180–188. doi: 10.1067/msy.2003.222. [DOI] [PubMed] [Google Scholar]
- 924.Kirkland K.B., Weinstein J.M. Adverse effects of contact isolation. Lancet. 1999;354:1177–1178. doi: 10.1016/S0140-6736(99)04196-3. [DOI] [PubMed] [Google Scholar]
- 925.Saint S., Higgins L.A., Nallamothu B.K., Chenoweth C. Do physicians examine patients in contact isolation less frequently? A brief report. Am J Infect Control. 2003;31:354–356. doi: 10.1016/s0196-6553(02)48250-8. [DOI] [PubMed] [Google Scholar]
- 926.Management of multidrug-resistant organisms in healthcare settings, 2006. Available from www.cdc.gov/ncidod/dhqp/pdf/ar/mdroGuideline2006.pdf.
- 927.Hall C.B., Powell K.R., MacDonald N.E. Respiratory syncytial viral infection in children with compromised immune function. N Engl J Med. 1986;315:77–81. doi: 10.1056/NEJM198607103150201. [DOI] [PubMed] [Google Scholar]
- 928.Lui S.L., Luk W.K., Cheung C.Y., Chan T.M., Lai K.N., Peiris J.S. Nosocomial outbreak of parvovirus B19 infection in a renal transplant unit. Transplantation. 2001;71:59–64. doi: 10.1097/00007890-200101150-00010. [DOI] [PubMed] [Google Scholar]
- 929.Weinstock D.M., Gubareva L.V., Zuccotti G. Prolonged shedding of multidrug-resistant influenza A virus in an immunocompromised patient. N Engl J Med. 2003;348:867–868. doi: 10.1056/NEJM200302273480923. [DOI] [PubMed] [Google Scholar]
- 930.van Tol M.J., Claas E.C., Heemskerk B. Adenovirus infection in children after allogeneic stem cell transplantation: diagnosis, treatment and immunity. Bone Marrow Transplant. 2005;35(Suppl 1):S73–S76. doi: 10.1038/sj.bmt.1704852. [DOI] [PubMed] [Google Scholar]
- 931.Wood D.J., David T.J., Chrystie I.L., Totterdell B. Chronic enteric virus infection in two T-cell–immunodeficient children. J Med Virol. 1988;24:435–444. doi: 10.1002/jmv.1890240410. [DOI] [PubMed] [Google Scholar]
- 932.Mori I., Matsumoto K., Sugimoto K. Prolonged shedding of rotavirus in a geriatric inpatient. J Med Virol. 2002;67:613–615. doi: 10.1002/jmv.10147. [DOI] [PubMed] [Google Scholar]
- 933.Cederna J.E., Terpenning M.S., Ensberg M., Bradley S.F., Kauffman C.A. Staphylococcus aureus nasal colonization in a nursing home: eradication with mupirocin. Infect Control Hosp Epidemiol. 1990;11:13–16. doi: 10.1086/646072. [DOI] [PubMed] [Google Scholar]
- 934.Kauffman C.A., Terpenning M.S., He X. Attempts to eradicate methicillin-resistant Staphylococcus aureus from a long-term-care facility with the use of mupirocin ointment. Am J Med. 1993;94:371–378. doi: 10.1016/0002-9343(93)90147-h. [DOI] [PubMed] [Google Scholar]
- 935.Montecalvo M.A., de Lencastre H., Carraher M. Natural history of colonization with vancomycin-resistant Enterococcus faecium. Infect Control Hosp Epidemiol. 1995;16:680–685. doi: 10.1086/647041. [DOI] [PubMed] [Google Scholar]
- 936.D'Agata E.M. High rate of false-negative results of the rectal swab culture method in detection of gastrointestinal colonization with vancomycin-resistant enterococci. Clin Infect Dis. 2002;34:167–172. doi: 10.1086/338234. [DOI] [PubMed] [Google Scholar]
- 937.Donskey C.J., Hoyen C.K., Das S.M., Helfand M.S., Hecker M.T. Recurrence of vancomycin-resistant Enterococcus stool colonization during antibiotic therapy. Infect Control Hosp Epidemiol. 2002;23:436–440. doi: 10.1086/502081. [DOI] [PubMed] [Google Scholar]
- 938.Scanvic A., Denic L., Gaillon S., Giry P., Andremont A., Lucet J.C. Duration of colonization by methicillin-resistant Staphylococcus aureus after hospital discharge and risk factors for prolonged carriage. Clin Infect Dis. 2001;32:1393–1398. doi: 10.1086/320151. [DOI] [PubMed] [Google Scholar]
- 939.Noskin G.A., Bednarz P., Suriano T., Reiner S., Peterson L.R. Persistent contamination of fabric-covered furniture by vancomycin-resistant enterococci: implications for upholstery selection in hospitals. Am J Infect Control. 2000;28:311–313. doi: 10.1067/mic.2000.108129. [DOI] [PubMed] [Google Scholar]
- 940.Gerson S.L., Parker P., Jacobs M.R., Creger R., Lazarus H.M. Aspergillosis due to carpet contamination. Infect Control Hosp Epidemiol. 1994;15(4 Pt 1):221–223. [PubMed] [Google Scholar]
- 941.Taplin D., Mertz P.M. Flower vases in hospitals as reservoirs of pathogens. Lancet. 1973;2:1279–1281. doi: 10.1016/s0140-6736(73)92866-3. [DOI] [PubMed] [Google Scholar]
- 942.Walsh T.J., Dixon D.M. Nosocomial aspergillosis: environmental microbiology, hospital epidemiology, diagnosis and treatment. Eur J Epidemiol. 1989;5:131–142. doi: 10.1007/BF00156818. [DOI] [PubMed] [Google Scholar]
- 943.Lass-Florl C., Rath P., Niederwieser D. Aspergillus terreus infections in haematological malignancies: molecular epidemiology suggests association with in-hospital plants. J Hosp Infect. 2000;46:31–35. doi: 10.1053/jhin.2000.0799. [DOI] [PubMed] [Google Scholar]
- 944.Raad I., Hanna H., Osting C. Masking of neutropenic patients on transport from hospital rooms is associated with a decrease in nosocomial aspergillosis during construction. Infect Control Hosp Epidemiol. 2002;23:41–43. doi: 10.1086/501967. [DOI] [PubMed] [Google Scholar]
- 945.www.cms.hhs.gov/CLIA.
- 946.Emori T.G., Haley R.W., Stanley R.C. The infection control nurse in US hospitals, 1976–1977: characteristics of the position and its occupant. Am J Epidemiol. 1980;111:592–607. doi: 10.1093/oxfordjournals.aje.a112936. [DOI] [PubMed] [Google Scholar]
- 947.Richet H.M., Benbachir M., Brown D.E. Are there regional variations in the diagnosis, surveillance, and control of methicillin-resistant Staphylococcus aureus? Infect Control Hosp Epidemiol. 2003;24:334–341. doi: 10.1086/502216. [DOI] [PubMed] [Google Scholar]
- 948.Anderson DJ, Kirkland KB, McDonald JR, et al. Results of a survey of work duties of 56 infection control professionals (ICPs): are new guidelines needed for the staffing of infection control (IC) programs? [abstract 146]. Presented at the 16th annual meeting of the Society for Healthcare Epidemiology of America, Chicago, IL, 2006.
- 949.Harvey M.A. Critical care unit bedside design and furnishing: impact on nosocomial infections. Infect Control Hosp Epidemiol. 1998;19:597–601. doi: 10.1086/647881. [DOI] [PubMed] [Google Scholar]
- 950.Srinivasan A., Beck C., Buckley T. The ability of hospital ventilation systems to filter Aspergillus and other fungi following a building implosion. Infect Control Hosp Epidemiol. 2002;23:520–524. doi: 10.1086/502100. [DOI] [PubMed] [Google Scholar]
- 951.Maragakis L.L., Bradley K.L., Song X. Increased catheter-related bloodstream infection rates after the introduction of a new mechanical valve intravenous access port. Infect Control Hosp Epidemiol. 2006;27:67–70. doi: 10.1086/499166. [DOI] [PubMed] [Google Scholar]
- 952.Joint Commision on Accreditation of Healthcare Organizations . Joint Commision on Accreditation of Healthcare Organizations; Oakbrook Terrace (IL): 2007. Comprehensive accredication manual for hospitals: the official handbook. [PubMed] [Google Scholar]
- 953.Peterson L.R., Noskin G.A. New technology for detecting multidrug-resistant pathogens in the clinical microbiology laboratory. Emerg Infect Dis. 2001;7:306–311. doi: 10.3201/eid0702.010233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 954.Diekema D.J., Doebbeling B.N. Employee health and infection control. Infect Control Hosp Epidemiol. 1995;16:292–301. doi: 10.1086/647110. [DOI] [PubMed] [Google Scholar]
- 955.Weems J.J., Jr. Nosocomial outbreak of Pseudomonas cepacia associated with contamination of reusable electronic ventilator temperature probes. Infect Control Hosp Epidemiol. 1993;14:583–586. doi: 10.1086/646642. [DOI] [PubMed] [Google Scholar]
- 956.Berthelot P., Grattard F., Mahul P. Ventilator temperature sensors: an unusual source of Pseudomonas cepacia in nosocomial infection. J Hosp Infect. 1993;25:33–43. doi: 10.1016/0195-6701(93)90006-l. [DOI] [PubMed] [Google Scholar]
- 957.Centers for Disease Control and Prevention Bronchoscopy-related infections and pseudoinfections, New York, 1996 and 1998. MMWR Morb Mortal Wkly Rep. 1999;48:557–560. [PubMed] [Google Scholar]
- 958.Heeg P., Roth K., Reichl R., Cogdill C.P., Bond W.W. Decontaminated single-use devices: an oxymoron that may be placing patients at risk for cross-contamination. Infect Control Hosp Epidemiol. 2001;22:542–549. doi: 10.1086/501949. [DOI] [PubMed] [Google Scholar]
- 959.www.fda.gov/cdrh/reprocessing/.
- 960.Centers for Disease Control and Prevention Prevention and control of influenza: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2003;52:1–36. [PubMed] [Google Scholar]
- 961.Weinstock D.M., Eagan J., Malak S.A. Control of influenza A on a bone marrow transplant unit. Infect Control Hosp Epidemiol. 2000;21:730–732. doi: 10.1086/501726. [DOI] [PubMed] [Google Scholar]
- 962.Cromer A.L., Hutsell S.O., Latham S.C. Impact of implementing a method of feedback and accountability related to contact precautions compliance. Am J Infect Control. 2004;32:451–455. doi: 10.1016/j.ajic.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 963.Eveillard M., Eb F., Tramier B. Evaluation of the contribution of isolation precautions in prevention and control of multi-resistant bacteria in a teaching hospital. J Hosp Infect. 2001;47:116–124. doi: 10.1053/jhin.2000.0877. [DOI] [PubMed] [Google Scholar]
- 964.Pfeiffer J., Gilmore G. The text as an orientation tool. In: Pfeiffer J., editor. APIC Text of Infection Control and Epidemiology. Association for Professionals in Infection Control and Epidemiology; Washington, DC: 2000. 7/1–8. [Google Scholar]
- 965.Gaynes R.P., Emori T.G. Surveillance for nosocomial infections. In: Abrutyn E., Goldmann D.A., Scheckler W.E., editors. Saunders Infection Control Reference. Saunders; Philadelphia, PA: 2001. pp. 40–44. [Google Scholar]
- 966.Centers for Disease Control and Prevention Monitoring hospital-acquired infections to promote patient safety, United States, 1990-1999. MMWR Morb Mortal Wkly Rep. 2000;49:149–153. [PubMed] [Google Scholar]
- 967.Curran E.T., Benneyan J.C., Hood J. Controlling methicillin-resistant Staphylococcus aureus: a feedback approach using annotated statistical process control charts. Infect Control Hosp Epidemiol. 2002;23:13–18. doi: 10.1086/501961. [DOI] [PubMed] [Google Scholar]
- 968.Lanotte P., Cantagrel S., Mereghetti L. Spread of Stenotrophomonas maltophilia colonization in a pediatric intensive care unit detected by monitoring tracheal bacterial carriage and molecular typing. Clin Microbiol Infect. 2003;9:1142–1147. doi: 10.1046/j.1469-0691.2003.00785.x. [DOI] [PubMed] [Google Scholar]
- 969.Coopersmith C.M., Zack J.E., Ward M.R. The impact of bedside behavior on catheter-related bacteremia in the intensive care unit. Arch Surg. 2004;139:131–136. doi: 10.1001/archsurg.139.2.131. [DOI] [PubMed] [Google Scholar]
- 970.O'Brien K.L., Beall B., Barrett N.L. Epidemiology of invasive group a streptococcus disease in the United States, 1995–1999. Clin Infect Dis. 2002;35:268–276. doi: 10.1086/341409. [DOI] [PubMed] [Google Scholar]
- 971.Nicolle L.E., Dyck B., Thompson G. Regional dissemination and control of epidemic methicillin-resistant Staphylococcus aureus. Manitoba Chapter of CHICA-Canada. Infect Control Hosp Epidemiol. 1999;20(3):202–205. doi: 10.1086/501613. [DOI] [PubMed] [Google Scholar]
- 972.Seybold U., Kourbatova E.V., Johnson J.G. Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care–associated blood stream infections. Clin Infect Dis. 2006;42:647–656. doi: 10.1086/499815. [DOI] [PubMed] [Google Scholar]
- 973.Bond W.W., Favero M.S., Petersen N.J., Gravelle C.R., Ebert J.W., Maynard J.E. Survival of hepatitis B virus after drying and storage for one week. Lancet. 1981;1:550–551. doi: 10.1016/s0140-6736(81)92877-4. [DOI] [PubMed] [Google Scholar]
- 974.Ehrenkranz N.J., Alfonso B.C. Failure of bland soap handwash to prevent hand transfer of patient bacteria to urethral catheters. Infect Control Hosp Epidemiol. 1991;12:654–662. doi: 10.1086/646261. [DOI] [PubMed] [Google Scholar]
- 975.Winnefeld M., Richard M.A., Drancourt M., Grob J.J. Skin tolerance and effectiveness of two hand decontamination procedures in everyday hospital use. Br J Dermatol. 2000;143:546–550. doi: 10.1111/j.1365-2133.2000.03708.x. [DOI] [PubMed] [Google Scholar]
- 976.Widmer A.F. Replace hand washing with use of a waterless alcohol hand rub? Clin Infect Dis. 2000;31:136–143. doi: 10.1086/313888. [DOI] [PubMed] [Google Scholar]
- 977.Mortimer E.A., Jr., Lipsitz P.J., Wolinsky E., Gonzaga A.J., Rammelkamp C.H., Jr. Transmission of staphylococci between newborns: importance of the hands to personnel. Am J Dis Child. 1962;104:289–295. doi: 10.1001/archpedi.1962.02080030291012. [DOI] [PubMed] [Google Scholar]
- 978.Casewell M., Phillips I. Hands as route of transmission for Klebsiella species. Br Med J. 1977;2:1315–1317. doi: 10.1136/bmj.2.6098.1315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 979.Ojajarvi J. Effectiveness of hand washing and disinfection methods in removing transient bacteria after patient nursing. J Hyg (Lond) 1980;85:193–203. doi: 10.1017/s0022172400063221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 980.Otter J, Havill N, Adams N, Joyce J. Extensive environmental contamination associated with patients with loose stools and MRSA colonization of the gastrointestinal tract [abstract 159]. Presented at the 16th annual scientific meeting of the Society for Healthcare Epidemiology of America, Chicago, IL, 2006.
- 981.Weber D.J., Sickbert-Bennett E., Gergen M.F., Rutala W.A. Efficacy of selected hand hygiene agents used to remove Bacillus atrophaeus (a surrogate of Bacillus anthracis) from contaminated hands. JAMA. 2003;289:1274–1277. doi: 10.1001/jama.289.10.1274. [DOI] [PubMed] [Google Scholar]
- 982.Saiman L., Lerner A., Saal L. Banning artificial nails from health care settings. Am J Infect Control. 2002;30:252–254. doi: 10.1067/mic.2002.122102. [DOI] [PubMed] [Google Scholar]
- 983.Johnson S., Gerding D.N., Olson M.M. Prospective, controlled study of vinyl glove use to interrupt Clostridium difficile nosocomial transmission. Am J Med. 1990;88:137–140. doi: 10.1016/0002-9343(90)90462-m. [DOI] [PubMed] [Google Scholar]
- 984.Neal J.G., Jackson E.M., Suber F., Edlich R.F. Latex glove penetration by pathogens: a review of the literature. J Long Term Eff Med Implants. 1998;8:233–240. [PubMed] [Google Scholar]
- 985.Broyles J.M., O'Connell K.P., Korniewicz D.M. PCR-based method for detecting viral penetration of medical exam gloves. J Clin Microbiol. 2002;40:2725–2728. doi: 10.1128/JCM.40.8.2725-2728.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 986.Patterson J.E., Vecchio J., Pantelick E.L. Association of contaminated gloves with transmission of Acinetobacter calcoaceticus var. anitratus in an intensive care unit. Am J Med. 1991;91:479–483. doi: 10.1016/0002-9343(91)90183-x. [DOI] [PubMed] [Google Scholar]
- 987.Goldmann D.A. Epidemiology and prevention of pediatric viral respiratory infections in health-care institutions. Emerg Infect Dis. 2001;7:249–253. doi: 10.3201/eid0702.010220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 988.Gaggero A., Avendano L.F., Fernandez J., Spencer E. Nosocomial transmission of rotavirus from patients admitted with diarrhea. J Clin Microbiol. 1992;30:3294–3297. doi: 10.1128/jcm.30.12.3294-3297.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 989.Merritt K., Hitchins V.M., Brown S.A. Safety and cleaning of medical materials and devices. J Biomed Mater Res. 2000;53:131–136. doi: 10.1002/(sici)1097-4636(2000)53:2<131::aid-jbm1>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 990.Kampf G., Bloss R., Martiny H. Surface fixation of dried blood by glutaraldehyde and peracetic acid. J Hosp Infect. 2004;57:139–143. doi: 10.1016/j.jhin.2004.02.004. [DOI] [PubMed] [Google Scholar]
- 991.Weber D.J., Rutala W.A. Role of environmental contamination in the transmission of vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 1997;18:306–309. doi: 10.1086/647616. [DOI] [PubMed] [Google Scholar]
- 992.Byers K.E., Durbin L.J., Simonton B.M., Anglim A.M., Adal K.A., Farr B.M. Disinfection of hospital rooms contaminated with vancomycin-resistant Enterococcus faecium. Infect Control Hosp Epidemiol. 1998;19:261–264. doi: 10.1086/647806. [DOI] [PubMed] [Google Scholar]
- 993.Martinez J.A., Ruthazer R., Hansjosten K., Barefoot L., Snydman D.R. Role of environmental contamination as a risk factor for acquisition of vancomycin-resistant enterococci in patients treated in a medical intensive care unit. Arch Intern Med. 2003;163:1905–1912. doi: 10.1001/archinte.163.16.1905. [DOI] [PubMed] [Google Scholar]
- 994.US Environmental Protection Agency. Federal Insecticide, Fungicide, and Rodenticidal Act 7 USC 136 et seq.
- 995.Devine J., Cooke R.P., Wright E.P. Is methicillin-resistant Staphylococcus aureus (MRSA) contamination of ward-based computer terminals a surrogate marker for nosocomial MRSA transmission and handwashing compliance? J Hosp Infect. 2001;48:72–75. doi: 10.1053/jhin.2001.0955. [DOI] [PubMed] [Google Scholar]
- 996.Sattar S.A., Springthorpe S., Mani S. Transfer of bacteria from fabrics to hands and other fabrics: development and application of a quantitative method using Staphylococcus aureus as a model. J Appl Microbiol. 2001;90:962–970. doi: 10.1046/j.1365-2672.2001.01347.x. [DOI] [PubMed] [Google Scholar]
- 997.Shiomori T., Miyamoto H., Makishima K. Evaluation of bedmaking-related airborne and surface methicillin-resistant Staphylococcus aureus contamination. J Hosp Infect. 2002;50:30–35. doi: 10.1053/jhin.2001.1136. [DOI] [PubMed] [Google Scholar]
- 998.Whyte W., Baird G., Annand R. Bacterial contamination on the surface of hospital linen chutes. J Hyg (Lond) 1969;67:427–435. doi: 10.1017/s002217240004184x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 999.Michaelsen G.S. Designing linen chutes to reduce spread of infectious organisms. Hospitals. 1965;39:116–119. [PubMed] [Google Scholar]
- 1000.Plott R.T., Wagner R.F., Jr., Tyring S.K. Iatrogenic contamination of multidose vials in simulated use: a reassessment of current patient injection technique. Arch Dermatol. 1990;126:1441–1444. [PubMed] [Google Scholar]
- 1001.Samandari T., Malakmadze N., Balter S. A large outbreak of hepatitis B virus infections associated with frequent injections at a physician's office. Infect Control Hosp Epidemiol. 2005;26:745–750. doi: 10.1086/502612. [DOI] [PubMed] [Google Scholar]
- 1002.Comstock R.D., Mallonee S., Fox J.L. A large nosocomial outbreak of hepatitis C and hepatitis B among patients receiving pain remediation treatments. Infect Control Hosp Epidemiol. 2004;25:576–583. doi: 10.1086/502442. [DOI] [PubMed] [Google Scholar]
- 1003.Germain J.M., Carbonne A., Thiers V. Patient-to-patient transmission of hepatitis C virus through the use of multidose vials during general anesthesia. Infect Control Hosp Epidemiol. 2005;26:789–792. doi: 10.1086/502618. [DOI] [PubMed] [Google Scholar]
- 1004.Macedo de Oliveira A., White K.L., Leschinsky D.P. An outbreak of hepatitis C virus infections among outpatients at a hematology/oncology clinic. Ann Intern Med. 2005;142:898–902. doi: 10.7326/0003-4819-142-11-200506070-00007. [DOI] [PubMed] [Google Scholar]
- 1005.Hsu J., Jensen B., Arduino M. Streptococcal meningitis following myelogram procedures. Infect Control Hosp Epidemiol. 2007;28:614–617. doi: 10.1086/513496. [DOI] [PubMed] [Google Scholar]
- 1006.Srinivasan A., Song X., Ross T., Merz W., Brower R., Perl T.M. A prospective study to determine whether cover gowns in addition to gloves decrease nosocomial transmission of vancomycin-resistant enterococci in an intensive care unit. Infect Control Hosp Epidemiol. 2002;23:424–428. doi: 10.1086/502079. [DOI] [PubMed] [Google Scholar]
- 1007.Nichols W.G., Corey L., Gooley T., Davis C., Boeckh M. Parainfluenza virus infections after hematopoietic stem cell transplantation: risk factors, response to antiviral therapy, and effect on transplant outcome. Blood. 2001;98:573–578. doi: 10.1182/blood.v98.3.573. [DOI] [PubMed] [Google Scholar]
- 1008.Elizaga J., Olavarria E., Apperley J., Goldman J., Ward K. Parainfluenza virus 3 infection after stem cell transplant: relevance to outcome of rapid diagnosis and ribavirin treatment. Clin Infect Dis. 2001;32:413–418. doi: 10.1086/318498. [DOI] [PubMed] [Google Scholar]
- 1009.Oishi I., Kimura T., Murakami T. Serial observations of chronic rotavirus infection in an immunodeficient child. Microbiol Immunol. 1991;35:953–961. doi: 10.1111/j.1348-0421.1991.tb01617.x. [DOI] [PubMed] [Google Scholar]
- 1010.Fierobe L., Lucet J.C., Decre D. An outbreak of imipenem-resistant Acinetobacter baumannii in critically ill surgical patients. Infect Control Hosp Epidemiol. 2001;22:35–40. doi: 10.1086/501822. [DOI] [PubMed] [Google Scholar]
- 1011.Montesinos I., Salido E., Delgado T., Lecuona M., Sierra A. Epidemiology of methicillin-resistant Staphylococcus aureus at a university hospital in the Canary Islands. Infect Control Hosp Epidemiol. 2003;24:667–672. doi: 10.1086/502276. [DOI] [PubMed] [Google Scholar]
- 1012.Poutanen S.M., Vearncombe M., McGeer A.J., Gardam M., Large G., Simor A.E. Nosocomial acquisition of methicillin-resistant Staphylococcus aureus during an outbreak of severe acute respiratory syndrome. Infect Control Hosp Epidemiol. 2005;26:134–137. doi: 10.1086/502516. [DOI] [PubMed] [Google Scholar]
- 1013.Yap F.H., Gomersall C.D., Fung K.S. Increase in methicillin-resistant Staphylococcus aureus acquisition rate and change in pathogen pattern associated with an outbreak of severe acute respiratory syndrome. Clin Infect Dis. 2004;39:511–516. doi: 10.1086/422641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1014.Layton M.C., Perez M., Heald P., Patterson J.E. An outbreak of mupirocin-resistant Staphylococcus aureus on a dermatology ward associated with an environmental reservoir. Infect Control Hosp Epidemiol. 1993;14:369–375. doi: 10.1086/646764. [DOI] [PubMed] [Google Scholar]
- 1015.Gilmore A., Stuart J., Andrews N. Risk of secondary meningococcal disease in health-care workers. Lancet. 2000;356:1654–1655. doi: 10.1016/S0140-6736(00)03163-9. [DOI] [PubMed] [Google Scholar]
- 1016.www.cdc.gov/flu/avian/index.htm.
- 1017.www.hhs.gov/pandemicflu/plan/pdf/S04.pdf.
- 1018.Ehresmann K.R., Hedberg C.W., Grimm M.B., Norton C.A., MacDonald K.L., Osterholm M.T. An outbreak of measles at an international sporting event with airborne transmission in a domed stadium. J Infect Dis. 1995;171:679–683. doi: 10.1093/infdis/171.3.679. [DOI] [PubMed] [Google Scholar]
- 1019.Gustafson T.L., Lavely G.B., Brawner E.R., Jr., Hutcheson R.H., Jr., Wright P.F., Schaffner W. An outbreak of airborne nosocomial varicella. Pediatrics. 1982;70:550–556. [PubMed] [Google Scholar]
- 1020.Hyams P.J., Stuewe M.C., Heitzer V. Herpes zoster causing varicella (chickenpox) in hospital employees: cost of a casual attitude. Am J Infect Control. 1984;12:2–5. doi: 10.1016/0196-6553(84)90064-6. [DOI] [PubMed] [Google Scholar]
- 1021.Pavelchak N., DePersis R.P., London M. Identification of factors that disrupt negative air pressurization of respiratory isolation rooms. Infect Control Hosp Epidemiol. 2000;21:191–195. doi: 10.1086/501742. [DOI] [PubMed] [Google Scholar]
- 1022.Rice N., Streifel A., Vesley D. An evaluation of hospital special ventilation room pressures. Infect Control Hosp Epidemiol. 2001;22:19–23. doi: 10.1086/501819. [DOI] [PubMed] [Google Scholar]
- 1023.Hutton M.D., Stead W.W., Cauthen G.M., Bloch A.B., Ewing W.M. Nosocomial transmission of tuberculosis associated with a draining abscess. J Infect Dis. 1990;161:286–295. doi: 10.1093/infdis/161.2.286. [DOI] [PubMed] [Google Scholar]
- 1024.Frampton M.W. An outbreak of tuberculosis among hospital personnel caring for a patient with a skin ulcer. Ann Intern Med. 1992;117:312–313. doi: 10.7326/0003-4819-117-4-312. [DOI] [PubMed] [Google Scholar]
- 1025.Ammari L.K., Bell L.M., Hodinka R.L. Secondary measles vaccine failure in healthcare workers exposed to infected patients. Infect Control Hosp Epidemiol. 1993;14:81–86. doi: 10.1086/646687. [DOI] [PubMed] [Google Scholar]
- 1026.Behrman A., Schmid D.S., Crivaro A., Watson B. A cluster of primary varicella cases among healthcare workers with false-positive varicella zoster virus titers. Infect Control Hosp Epidemiol. 2003;24:202–206. doi: 10.1086/502187. [DOI] [PubMed] [Google Scholar]
- 1027.Josephson A., Gombert M.E. Airborne transmission of nosocomial varicella from localized zoster. J Infect Dis. 1988;158:238–241. doi: 10.1093/infdis/158.1.238. [DOI] [PubMed] [Google Scholar]
- 1028.Brodkin R.H. Zoster-causing varicella: current dangers of contagion without isolation. Arch Dermatol. 1963;88:322–324. doi: 10.1001/archderm.1963.01590210080012. [DOI] [PubMed] [Google Scholar]
- 1029.Suzuki K., Yoshikawa T., Tomitaka A., Matsunaga K., Asano Y. Detection of aerosolized varicella-zoster virus DNA in patients with localized herpes zoster. J Infect Dis. 2004;189:1009–1012. doi: 10.1086/382029. [DOI] [PubMed] [Google Scholar]
- 1030.Ruuskanen O., Salmi T.T., Halonen P. Measles vaccination after exposure to natural measles. J Pediatr. 1978;93:43–46. doi: 10.1016/s0022-3476(78)80597-6. [DOI] [PubMed] [Google Scholar]
- 1031.Berkovich S., Starr S. Use of live measles virus vaccine to abort an expected outbreak of measles within a closed population. N Engl J Med. 1963;269:75–77. doi: 10.1056/NEJM196307112690204. [DOI] [PubMed] [Google Scholar]
- 1032.Centers for Disease Control and Prevention Measles, mumps, and rubella vaccine use and strategies for elimination of measles, rubella, and congenital rubella syndrome and control of mumps: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 1998;47:1–57. [PubMed] [Google Scholar]
- 1033.Centers for Disease Control and Prevention General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2006;55:1–48. [PubMed] [Google Scholar]
- 1034.Watson B., Seward J., Yang A. Postexposure effectiveness of varicella vaccine. Pediatrics. 2000;105(1 Pt 1):84–88. doi: 10.1542/peds.105.1.84. [DOI] [PubMed] [Google Scholar]
- 1035.Salzman M.B., Garcia C. Postexposure varicella vaccination in siblings of children with active varicella. Pediatr Infect Dis J. 1998;17:256–257. doi: 10.1097/00006454-199803000-00020. [DOI] [PubMed] [Google Scholar]
- 1036.Centers for Disease Control and Preverntion Vaccinia (smallpox) vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2001. MMWR Recomm Rep. 2001;50:1–25. [PubMed] [Google Scholar]
- 1037.Fulginiti V.A., Papier A., Lane J.M., Neff J.M., Henderson D.A. Smallpox vaccination: a review. Part I: background, vaccination technique, normal vaccination and revaccination, and expected normal reactions. Clin Infect Dis. 2003;37:241–250. doi: 10.1086/375824. [DOI] [PubMed] [Google Scholar]
- 1038.Dixon C.W. Smallpox in Tripolitania, 1946: an epidemiological and clinical study of 500 cases, including trials of penicillin treatment. J Hyg (Lond) 1948;46:351–377. doi: 10.1017/s0022172400036536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1039.Murray W.A., Streifel A.J., O'Dea T.J., Rhame F.S. Ventilation for protection of immune-compromised patients. ASHRAE Trans. 1988;94:1185. [Google Scholar]
- 1040.Rutala W.A., Jones S.M., Worthington J.M., Reist P.C., Weber D.J. Efficacy of portable filtration units in reducing aerosolized particles in the size range of Mycobacterium tuberculosis. Infect Control Hosp Epidemiol. 1995;16:391–398. doi: 10.1086/647136. [DOI] [PubMed] [Google Scholar]
- 1041.Mandell G.L., Bennett J.E., Dolin R., editors. Mandell, Douglas and Bennett's principles and practice of infectious diseases. 5th ed. Churchill Livingstone; Philadelphia: 2000. [Google Scholar]
- 1042.Heymann D.L., editor. Control of communicable diseases manual. 18th ed. American Public Health Association; Washington, DC: 2005. [Google Scholar]
- 1043.Vreden S.G., Visser L.G., Verweij J.J. Outbreak of amebiasis in a family in the Netherlands. Clin Infect Dis. 2000;31:1101–1104. doi: 10.1086/318153. [DOI] [PubMed] [Google Scholar]
- 1044.Thacker S.B., Kimball A.M., Wolfe M., Choi K., Gilmore L. Parasitic disease control in a residential facility for the mentally retarded: failure of selected isolation procedures. Am J Public Health. 1981;71:303–305. doi: 10.2105/ajph.71.3.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1045.Sampathkumar P. West Nile virus: epidemiology, clinical presentation, diagnosis, and prevention. Mayo Clin Proc. 2003;78:1137–1143. doi: 10.4065/78.9.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1046.Ruben B., Band J.D., Wong P., Colville J. Person-to-person transmission of Brucella melitensis. Lancet. 1991;337:14–15. doi: 10.1016/0140-6736(91)93332-4. [DOI] [PubMed] [Google Scholar]
- 1047.Vandercam B., Zech F., de Cooman S., Bughin C., Gigi J., Wauters G. Isolation of Brucella melitensis from human sperm. Eur J Clin Microbiol Infect Dis. 1990;9:303–304. doi: 10.1007/BF01968069. [DOI] [PubMed] [Google Scholar]
- 1048.Robichaud S., Libman M., Behr M., Rubin E. Prevention of laboratory-acquired brucellosis. Clin Infect Dis. 2004;38:e119–e122. doi: 10.1086/421024. [DOI] [PubMed] [Google Scholar]
- 1049.Troy C.J., Peeling R.W., Ellis A.G. Chlamydia pneumoniae as a new source of infectious outbreaks in nursing homes. JAMA. 1997;277:1214–1218. [PubMed] [Google Scholar]
- 1050.Ekman M.R., Grayston J.T., Visakorpi R., Kleemola M., Kuo C.C., Saikku P. An epidemic of infections due to Chlamydia pneumoniae in military conscripts. Clin Infect Dis. 1993;17:420–425. doi: 10.1093/clinids/17.3.420. [DOI] [PubMed] [Google Scholar]
- 1051.Eickhoff T.C. An outbreak of surgical wound infections due to Clostridium perfringens. Surg Gynecol Obstet. 1962;114:102–108. [PubMed] [Google Scholar]
- 1052.Kohn G.J., Linne S.R., Smith C.M., Hoeprich P.D. Acquisition of coccidioidomycosis at necropsy by inhalation of coccidioidal endospores. Diagn Microbiol Infect Dis. 1992;15:527–530. doi: 10.1016/0732-8893(92)90103-z. [DOI] [PubMed] [Google Scholar]
- 1053.Wright P.W., Pappagianis D., Wilson M. Donor-related coccidioidomycosis in organ transplant recipients. Clin Infect Dis. 2003;37:1265–1269. doi: 10.1086/378741. [DOI] [PubMed] [Google Scholar]
- 1054.Maitreyi R.S., Dar L., Muthukumar A. Acute hemorrhagic conjunctivitis due to enterovirus 70 in India. Emerg Infect Dis. 1999;5:267–269. doi: 10.3201/eid0502.990212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1055.Centers for Disease Control and Prevention Acute hemorrhagic conjunctivitis outbreak caused by Coxsackievirus A24, Puerto Rico, 2003. MMWR Morb Mortal Wkly Rep. 2004;53:632–634. [PubMed] [Google Scholar]
- 1056.Faden H., Wynn R.J., Campagna L., Ryan R.M. Outbreak of adenovirus type 30 in a neonatal intensive care unit. J Pediatr. 2005;146:523–527. doi: 10.1016/j.jpeds.2004.11.032. [DOI] [PubMed] [Google Scholar]
- 1057.Chaberny I.E., Schnitzler P., Geiss H.K., Wendt C. An outbreak of epidemic keratoconjunctivtis in a pediatric unit due to adenovirus type 8. Infect Control Hosp Epidemiol. 2003;24:514–519. doi: 10.1086/502247. [DOI] [PubMed] [Google Scholar]
- 1058.Warren D., Nelson K.E., Farrar J.A. A large outbreak of epidemic keratoconjunctivitis: problems in controlling nosocomial spread. J Infect Dis. 1989;160:938–943. doi: 10.1093/infdis/160.6.938. [DOI] [PubMed] [Google Scholar]
- 1059.www.cdc.gov/ncidod/dvrd/cjd/qa_cjd_infection_control.htm.
- 1060.Wang C.Y., Wu H.D., Hsueh P.R. Nosocomial transmission of cryptococcosis. N Engl J Med. 2005;352:1271–1272. doi: 10.1056/NEJM200503243521225. [DOI] [PubMed] [Google Scholar]
- 1061.Beyt B.E., Jr., Waltman S.R. Cryptococcal endophthalmitis after corneal transplantation. N Engl J Med. 1978;298:825–826. doi: 10.1056/NEJM197804132981506. [DOI] [PubMed] [Google Scholar]
- 1062.Widdowson M.A., Glass R., Monroe S. Probable transmission of norovirus on an airplane. JAMA. 2005;293:1859–1860. doi: 10.1001/jama.293.15.1859. [DOI] [PubMed] [Google Scholar]
- 1063.Centers for Disease Control and Prevention Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 1999;48:1–37. [PubMed] [Google Scholar]
- 1064.Rosenblum L.S., Villarino M.E., Nainan O.V. Hepatitis A outbreak in a neonatal intensive care unit: risk factors for transmission and evidence of prolonged viral excretion among preterm infants. J Infect Dis. 1991;164:476–482. doi: 10.1093/infdis/164.3.476. [DOI] [PubMed] [Google Scholar]
- 1065.Carl M., Kantor R.J., Webster H.M., Fields H.A., Maynard J.E. Excretion of hepatitis A virus in the stools of hospitalized hepatitis patients. J Med Virol. 1982;9:125–129. doi: 10.1002/jmv.1890090207. [DOI] [PubMed] [Google Scholar]
- 1066.Robson S.C., Adams S., Brink N., Woodruff B., Bradley D. Hospital outbreak of hepatitis E. Lancet. 1992;339:1424–1425. doi: 10.1016/0140-6736(92)91250-c. [DOI] [PubMed] [Google Scholar]
- 1067.Arvin A., Whitley R. Herpes simplex virus infections. In: Remington J.S., Klein J.O., editors. Infectious diseases of the fetus and newborn infant. 5th ed. Saunders; Philadelphia: 2001. [Google Scholar]
- 1068.Enright A.M., Prober C.G. Neonatal herpes infection: diagnosis, treatment and prevention. Semin Neonatol. 2002;7:283–291. doi: 10.1016/s1084-2756(02)90115-6. [DOI] [PubMed] [Google Scholar]
- 1069.Esper F., Boucher D., Weibel C., Martinello R.A., Kahn J.S. Human metapneumovirus infection in the United States: clinical manifestations associated with a newly emerging respiratory infection in children. Pediatrics. 2003;111(6 Pt 1):1407–1410. doi: 10.1542/peds.111.6.1407. [DOI] [PubMed] [Google Scholar]
- 1070.Colodner R., Sakran W., Miron D., Teitler N., Khavalevsky E., Kopelowitz J. Listeria moncytogenes cross-contamination in a nursery. Am J Infect Control. 2003;31:322–324. doi: 10.1067/mic.2003.25. [DOI] [PubMed] [Google Scholar]
- 1071.Farber J.M., Peterkin P.I., Carter A.O., Varughese P.V., Ashton F.E., Ewan E.P. Neonatal listeriosis due to cross-infection confirmed by isoenzyme typing and DNA fingerprinting. J Infect Dis. 1991;163:927–928. doi: 10.1093/infdis/163.4.927. [DOI] [PubMed] [Google Scholar]
- 1072.Schuchat A., Lizano C., Broome C.V., Swaminathan B., Kim C., Winn K. Outbreak of neonatal listeriosis associated with mineral oil. Pediatr Infect Dis J. 1991;10:183–189. doi: 10.1097/00006454-199103000-00003. [DOI] [PubMed] [Google Scholar]
- 1073.Pejaver R.K., Watson A.H., Mucklow E.S. Neonatal cross-infection with Listeria monocytogenes. J Infect. 1993;26:301–303. doi: 10.1016/0163-4453(93)95519-o. [DOI] [PubMed] [Google Scholar]
- 1074.Jain S.K., Persaud D., Perl T.M. Nosocomial malaria and saline flush. Emerg Infect Dis. 2005;11:1097–1099. doi: 10.3201/eid1107.050092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1075.Abulrahi H.A., Bohlega E.A., Fontaine R.E., al-Seghayer S.M., al-Ruwais A.A. Plasmodium falciparum malaria transmitted in hospital through heparin locks. Lancet. 1997;349:23–25. doi: 10.1016/s0140-6736(96)03508-8. [DOI] [PubMed] [Google Scholar]
- 1076.Al-Saigul A.M., Fontaine R.E., Haddad Q. Nosocomial malaria from contamination of a multidose heparin container with blood. Infect Control Hosp Epidemiol. 2000;21:329–330. doi: 10.1086/501765. [DOI] [PubMed] [Google Scholar]
- 1077.Piro S., Sammud M., Badi S., Al Ssabi L. Hospital-acquired malaria transmitted by contaminated gloves. J Hosp Infect. 2001;47:156–158. doi: 10.1053/jhin.2000.0907. [DOI] [PubMed] [Google Scholar]
- 1078.Book L.S., Overall J.C., Jr., Herbst J.J., Britt M.R., Epstein B., Jung A.L. Clustering of necrotizing enterocolitis: interruption by infection-control measures. N Engl J Med. 1977;297:984–986. doi: 10.1056/NEJM197711032971805. [DOI] [PubMed] [Google Scholar]
- 1079.Rotbart H.A., Levin M.J. How contagious is necrotizing enterocolitis? Pediatr Infect Dis. 1983;2:406–413. doi: 10.1097/00006454-198309000-00019. [DOI] [PubMed] [Google Scholar]
- 1080.Rotbart H.A., Levin M.J., Yolken R.H., Manchester D.K., Jantzen J. An outbreak of rotavirus-associated neonatal necrotizing enterocolitis. J Pediatr. 1983;103:454–459. doi: 10.1016/S0022-3476(83)80427-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1081.Gerber A.R., Hopkins R.S., Lauer B.A., Curry-Kane A.G., Rotbart H.A. Increased risk of illness among nursery staff caring for neonates with necrotizing enterocolitis. Pediatr Infect Dis. 1985;4:246–249. doi: 10.1097/00006454-198505000-00008. [DOI] [PubMed] [Google Scholar]
- 1082.Sanchez M.P., Erdman D.D., Torok T.J., Freeman C.J., Matyas B.T. Outbreak of adenovirus 35 pneumonia among adult residents and staff of a chronic care psychiatric facility. J Infect Dis. 1997;176:760–763. doi: 10.1086/517295. [DOI] [PubMed] [Google Scholar]
- 1083.Singh-Naz N., Brown M., Ganeshananthan M. Nosocomial adenovirus infection: molecular epidemiology of an outbreak. Pediatr Infect Dis J. 1993;12:922–925. [PubMed] [Google Scholar]
- 1084.Uemura T., Kawashitam T., Ostuka Y., Tanaka Y., Kusubae R., Yoshinaga M. A recent outbreak of adenovirus type 7 infection in a chronic inpatient facility for the severely handicapped. Infect Control Hosp Epidemiol. 2000;21:559–560. doi: 10.1086/503238. [DOI] [PubMed] [Google Scholar]
- 1085.Nuorti J.P., Butler J.C., Crutcher J.M. An outbreak of multidrug-resistant pneumococcal pneumonia and bacteremia among unvaccinated nursing home residents. N Engl J Med. 1998;338:1861–1868. doi: 10.1056/NEJM199806253382601. [DOI] [PubMed] [Google Scholar]
- 1086.Houff S.A., Burton R.C., Wilson R.W. Human-to-human transmission of rabies virus by corneal transplant. N Engl J Med. 1979;300:603–604. doi: 10.1056/NEJM197903153001105. [DOI] [PubMed] [Google Scholar]
- 1087.Centers for Disease Control and Prevention Human rabies prevention, United States, 1999: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 1999;48:1–21. [PubMed] [Google Scholar]
- 1088.Hayden F.G. Rhinovirus and the lower respiratory tract. Rev Med Virol. 2004;14:17–31. doi: 10.1002/rmv.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 1089.Valenti W.M., Clarke T.A., Hall C.B., Menegus M.A., Shapiro D.L. Concurrent outbreaks of rhinovirus and respiratory syncytial virus in an intensive care nursery: epidemiology and associated risk factors. J Pediatr. 1982;100:722–726. doi: 10.1016/s0022-3476(82)80571-4. [DOI] [PubMed] [Google Scholar]
- 1090.Chidekel A.S., Rosen C.L., Bazzy A.R. Rhinovirus infection associated with serious lower respiratory illness in patients with bronchopulmonary dysplasia. Pediatr Infect Dis J. 1997;16:43–47. doi: 10.1097/00006454-199701000-00010. [DOI] [PubMed] [Google Scholar]
- 1091.Drusin L.M., Ross B.G., Rhodes K.H., Krauss A.N., Scott R.A. Nosocomial ringworm in a neonatal intensive care unit: a nurse and her cat. Infect Control Hosp Epidemiol. 2000;21:605–607. doi: 10.1086/501814. [DOI] [PubMed] [Google Scholar]
- 1092.Lewis S.M., Lewis B.G. Nosocomial transmission of Trichophyton tonsurans tinea corporis in a rehabilitation hospital. Infect Control Hosp Epidemiol. 1997;18:322–325. doi: 10.1086/647619. [DOI] [PubMed] [Google Scholar]
- 1093.Saiman L., Jakob K., Holmes K.W. Molecular epidemiology of staphylococcal scalded skin syndrome in premature infants. Pediatr Infect Dis J. 1998;17:329–334. doi: 10.1097/00006454-199804000-00012. [DOI] [PubMed] [Google Scholar]
- 1094.Ramage L., Green K., Pyskir D., Simor A.E. An outbreak of fatal nosocomial infections due to group A streptococcus on a medical ward. Infect Control Hosp Epidemiol. 1996;17:429–431. doi: 10.1086/647335. [DOI] [PubMed] [Google Scholar]
- 1095.Kakis A., Gibbs L., Eguia J. An outbreak of group A streptococcal infection among health care workers. Clin Infect Dis. 2002;35:1353–1359. doi: 10.1086/344279. [DOI] [PubMed] [Google Scholar]
- 1096.Schwartz B., Elliott J.A., Butler J.C. Clusters of invasive group A streptococcal infections in family, hospital, and nursing home settings. Clin Infect Dis. 1992;15:277–284. doi: 10.1093/clinids/15.2.277. [DOI] [PubMed] [Google Scholar]
- 1097.National Communicable Disease Center . US Government Printing Office; Washington, DC: 1970. Isolation techniques for use in hospitals. [Google Scholar]
- 1098.Centers for Disease Control and Prevention . 2nd ed. US Government Printing Office; Washington, DC: 1975. Isolation techniques for use in hospitals. [Google Scholar]
- 1099.Garner J.S., Simmons B.P. US Department of Health and Human Services, Public Health Service, Centers for Disease Control; Atlanta (GA): 1983. CDC guideline for isolation precautions in hospitals. [Google Scholar]
- 1100.Lynch P., Jackson M.M., Cummings M.J., Stamm W.E. Rethinking the role of isolation practices in the prevention of nosocomial infections. Ann Intern Med. 1987;107:243–246. doi: 10.7326/0003-4819-107-2-243. [DOI] [PubMed] [Google Scholar]


