Abstract
The early cytokines interferon-α (IFN-α), tumour necrosis factor-α (TNF-α), interleukin-1, -6 and -8 (IL-1, -6, -8) are produced during the most early stage of an infection. The activities of these cytokines have been studied extensively in vitro and in rodents, but in vivo studies on the role of these cytokines in infectious diseases of food animals are few. This review concentrates on in vivo studies of cytokine involvement in infectious respiratory diseases of swine, with an emphasis on viral infections. First evidence for the role of early cytokines in pneumonia in swine came from experimental infections with Mycoplasma hyopneumoniae and Actinobacillus pleuropneumoniae. The role of TNF-α and IL-1 in the symptoms and pathology of porcine pleuropneumonia has recently been proven by use of an adenovirus vector expressing the anti-inflammatory IL-10. In the authors’ laboratory, studies were undertaken to investigate the relationship between viral respiratory disease and bioactive lung lavage levels of IFN-α, TNF-α, IL-1 and IL-6. Out of three respiratory viruses—porcine respiratory coronavirus (PRCV), porcine reproductive and respiratory syndrome virus (PRRSV) and swine influenza virus (SIV)—only SIV induced acute respiratory disease and severe lung damage by itself. Disease and lung pathology were tightly associated with the simultaneous production of IFN-α, TNF-α, IL-1 and IL-6. In challenge studies of SIV-vaccinated pigs, levels of IFN-α, TNF-α and IL-6, but not IL-1 were correlated with clinical and virological protection. Multifactorial respiratory disease was reproduced by combined inoculations with PRCV or PRRSV followed by LPS from Escherichia coli. In comparison with the respective single inoculations, which were subclinical, there was a true potentiation of disease and production of TNF-α, IL-1 and IL-6. TNF-α and IL-6 were best correlated with disease. In further studies, we will use more specific strategies to dissect the role of cytokines during viral infections.
Keywords: Pathogenesis, Early cytokines, Viral respiratory disease, Swine
1. Introduction
Interferon-α (IFN-α), tumour necrosis factor-α (TNF-α), interleukin-1-α and β (IL-1-α and β), interleukin-6 (IL-6) and interleukin-8 (IL-8) are produced during the most early stage of an infection, before the specific immune response comes into play. Each of these cytokines has a number of local and systemic effects, among others: upregulation of adhesion molecules on the vascular endothelium and leukocytes, activation of neutrophil and macrophage antimicrobial activities, fever, induction of acute phase proteins, catabolic effects on muscle and fat cells (for reviews see Murtaugh et al., 1996, Bogdan, 2000, Dinarello, 2000). TNF-α and IL-1, the classical “proinflammatory” cytokines, stimulate nearly all local and systemic inflammatory responses. IL-6, which can be induced by TNF-α and IL-1, is pyrogenic, induces the release of acute phase reactants by the liver, and in turn switches off proinflammatory cytokine production. While TNF-α and IL-1 mediate the initial adhesive reaction of neutrophils to the endothelium, IL-8 appears to be essential for the directed migration of leukocytes into the infected tissue (Strieter and Kunkel, 1994). IFN-α, though best known for its antiviral effects, can exert all of the systemic effects mentioned here and induces adhesion molecules in some in vitro systems. Due to their diverse actions, cytokines such as IFN-α and IL-6 have been classified as “proinflammatory” by some researchers and as “anti-inflammatory” by others. The term “early cytokines” applies to all cytokines mentioned here and will be used further.
The amount of in vitro studies on cytokine actions and interactions is impressive, but there are rather few in vivo studies on cytokines in infectious disease models of food animals. Still, the response of cells to cytokines can be markedly affected by the “context”, and the expression in vivo veritas is more valid for cytokine studies than for any other type of research. Here we focus on in vivo research of cytokine involvement in infectious respiratory diseases of swine. This type of research encounters some particular problems. First, cytokine production during pulmonary infections is mainly restricted to the lung and diffusion of cytokines across the blood–alveolar barrier is rather limited (Nelson et al., 1989). Samples of the lower respiratory tract, such as lung tissue, bronchoalveolar lavage (BAL) cells or fluid, are thus needed to study cytokine production, and these are difficult to obtain from living animals. For cytokine studies in particular, it is problematic to perform serial in vivo lung lavages because the procedure in itself causes lung damage with neutrophil infiltration which can influence cytokine production. Also, the amount of cells and fluid recovered in this way is rather small and not representative of the whole lung. Second, the possibilities for intervention in the form of treatment with specific cytokine-blocking antibodies or antagonists are limited. The major problem here is the delivery of such molecules to the deeper lung over a prolonged time period. This is the reason why most studies have only demonstrated associations between lung cytokines and other parameters, without proving a true cause–effect relationship. Third, respiratory infections occur with a high prevalence in swine, and the upper respiratory tract harbors commensal bacteria that can easily colonize the lungs in the presence of predisposing events. Also, some of the early cytokines are induced readily by a variety of stimuli. Thus, pigs of a high-health status are required for the study of cytokines at the lung level.
2. Cytokines mediate mycoplasmal and bacterial pneumonia of swine
First evidence for the involvement of early cytokines in pneumonia in swine came from experimental infections with mycoplasma and bacteria. Mycoplasma hyopneumoniae is the causative agent of enzootic pneumonia, a mild chronic pneumonia with few clinical consequences if uncomplicated. Characteristic peribronchiolar lymphoid hyperplasia and gross lung lesions develop by 3 and 4 weeks after experimental infection. Lesions were associated with increased levels of bioactive TNF-α, IL-1 and IL-6 in BAL fluids (Asai et al., 1993, Asai et al., 1994). Based on this association, it is believed that these cytokines mediate lymphocyte infiltration and activation in the pneumonic lung.
Porcine pleuropneumonia is caused by Actinobacillus pleuropneumoniae and is characterized by a necrotizing fibrinous pneumonia with prominent neutrophil infiltration and high morbidity and mortality. Dyspnoea, increased respiratory rate, fever, depression and inappetence occur as early as 2 h after endotracheal inoculation of A. pleuropneumoniae (Baarsch et al., 2000). TNF-α, IL-1, IL-6 and IL-8 mRNA levels in the lung increased at the same time and cytokine expression was localized to the periphery of lung lesions (Baarsch et al., 1995, Choi et al., 1999, Huang et al., 1999). The studies by Baarsch et al., 1995, Baarsch et al., 2000 illustrate perfectly that the results of cytokine measurements are dramatically affected by the sampling compartment and the detection method used, as well as by the time of sampling. IL-6 mRNA, for example, was detected by Northern blotting in lung parenchyma but not in BAL cells, whereas in situ hybridization (ISH) on BAL cells was clearly positive. Similarly, TNF-α mRNA in BAL cells appeared to be increased in ISH, but TNF-α bioactivity in BAL fluids was undetectable in a standard L929 cytotoxicity assay. In later experiments by the same group, however, elevated TNF-α levels were found in BAL fluids by ELISA (Morrison et al., 2000). In summary, TNF-α as well as bioactive IL-1 and IL-6 have been detected in lung lavage fluids and the role of the first two cytokines has recently been proven by administration to the lung of a recombinant replication-deficient adenovirus which expressed the anti-inflammatory cytokine IL-10. IL-10 expression reduced IL-1 and TNF-α secretion, and this was associated with clinical protection and a significant reduction of lung pathology (Morrison et al., 2000).
In a few studies, cytokines in serum were found to be suitable indicators of ongoing bacterial infections or other disease conditions. Indeed, haematogenic spread of bacteria may elicit a systemic cytokine response, and limited diffusion of cytokines from the lungs to the circulation is also possible. Serum IL-6, but not TNF-α or IFNs, were detectable during the acute stage of infection with A. pleuropneumoniae (Fossum et al., 1998). Other experimental infection studies with A. pleuropneumoniae found elevated serum TNF-α and IL-1 levels (Huang et al., 1999) or no cytokines at all (Baarsch et al., 1995). Changes in serum TNF-α have been associated with weight loss, depression and respiratory disease in high-health boars entering conventional swine facilities (Harding et al., 1997). Though the exact etiology of this so-called post-arrival disease is undefined, the pathogenesis seemingly involves changes in plasma or serum levels of TNF-α and acute phase reactants.
3. Viruses can induce the concurrent production of multiple early cytokines: the example of swine influenza virus
At the Laboratory of Veterinary Virology, Ghent University, we are studying the significance of cytokines during viral infections of the lungs of swine. We have previously demonstrated different cytokine profiles in BAL fluids of gnotobiotic pigs infected with porcine respiratory coronavirus (PRCV), porcine reproductive and respiratory syndrome virus (PRRSV) or swine influenza virus (SIV) (Van Reeth et al., 1999). We chose to detect bioactive cytokines in these studies because bioassays have the greatest biological significance. The major finding was that cytokines may provide a clue for the remarkable differences in pathogenicity between the three pulmonary viruses. After intratracheal inoculation of gnotobiotic pigs with high virus doses, both PRCV and PRRSV were unable to induce overt respiratory signs. The infection with PRCV, which replicates in alveolar epithelial cells, was asymptomatic, though gross lung consolidation consistently occurred. Microscopic lung inflammation and airway epithelial necrosis, on the other hand, remained very mild. Of the four cytokines (IFN-α, TNF-α, IL-1 and IL-6) examined, only IFN-α was found at high and sustained levels. PRRSV, a macrophage-tropic virus, caused a transient fever, anorexia and lethargy without respiratory disease. There was massive infiltration of alveolar septa and spaces with macrophage-like cells. Lung epithelial damage, neutrophil infiltration and gross lung consolidation, on the contrary, were minimal. Only IL-1 was consistently recovered from BAL fluids of PRRSV-infected pigs.
SIV, in contrast, is a primary respiratory pathogen of swine. The disease is characterized by high fever, depression, anorexia, tachypnoea and a labored abdominal respiration. The hallmarks of the infection are a fulminant virus replication in bronchial, bronchiolar and alveolar epithelia, degeneration and desquamation of epithelial cells, neutrophil infiltration, and the appearance of well-demarkated, dark red areas of gross pneumonia. Neutrophils are the predominant cell type in BAL fluids and they are thought to be responsible for the typical breathing difficulties and lung damage. Fig. 1 illustrates the time course of pathogenetic events and the corresponding cytokine profile after intratracheal inoculation of gnotobiotic pigs with SIV. Disease, peak virus titres and neutrophil infiltration developed as early as 18–24 h after inoculation. Clinical signs and neutrophil infiltration resolved very rapidly and virus titres had decreased significantly by 72 h. Excessive amounts of bioactive IFN-α, TNF-α, IL-1 and IL-6 were found in BAL fluids of these pigs. There was a tight correlation between cytokines on the one hand and disease, neutrophil infiltration and peak virus titres on the other. As clinical signs and lung inflammation subsided, IFN-α and IL-6 titres decreased and TNF-α and IL-1 became undetectable. Because of the synergistic interactions between these cytokines, the cytokine “quartet” is likely to have much more potent effects than each cytokine on its own. Furthermore, cytokines other than those examined here may also contribute to the pathogenesis. One candidate cytokine is IL-8, the primary neutrophil chemoattractant. IL-8 is one of the more difficult cytokines to detect by bioassay and commercial ELISAs for porcine IL-8 have only recently become available. Consequently, the secretion of IL-8 in body fluids of pigs has never been demonstrated so far and its significance in diseases of swine remains to be proven.
Fig. 1.
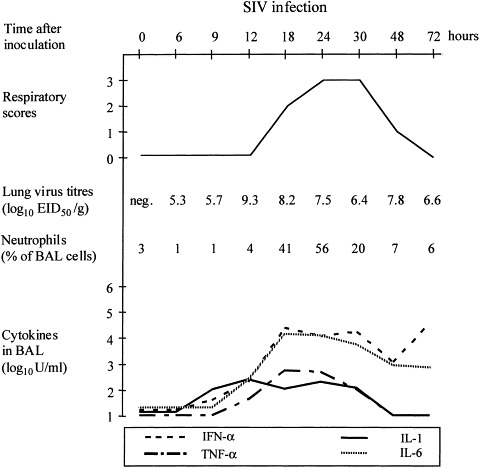
Kinetics of respiratory signs, lung virus titres, neutrophils and cytokines in BAL fluids after intratracheal inoculation of gnotobiotic pigs with the Sw/Belgium/1/83 (H1N1) SIV.
There are some striking parallels between SIV and A. pleuropneumoniae with regard to clinicopathological manifestations and cytokine profile. With both pathogens, the clinical outcomes of an experimental infection depend on the inoculation route and dose, and cytokines are only found in clinically severe infections. In our hands, characteristic “swine flu” symptoms can only be reproduced by direct intratracheal inoculation of 7.0–7.5 log10 EID50 virus. Intranasal or aerosol inoculation of a similar virus dose induced only mild clinical signs and pathology, and virus titres in the lungs were approximately 100 times lower. Interestingly, the aerosol inoculation also failed to induce detectable TNF-α and IL-1, while IFN-α and IL-6 levels were about 100 times lower than after intratracheal inoculation.
Further support for a role of cytokines in influenza symptoms and pneumonia comes from examinations of cytokine profiles in SIV vaccination-challenge studies (Van Reeth et al., 2001). In these studies, conventional SIV-negative pigs were vaccinated twice with different inactivated SIV vaccines and then challenged intratracheally with virulent SIV. At 24 h after the challenge, mean clinical scores, lung virus titres, neutrophil counts, and IFN-α, TNF-α and IL-6 levels were clearly lower in the vaccinates than in unvaccinated challenge controls. All three cytokines were directly correlated with the viral load and with disease severity. This type of study however does not really prove the role of cytokines in disease. Because replication of the challenge virus was significantly reduced in the vaccinated pigs, direct pathogenic effects of the virus could probably not occur. Still, our data raise questions about the role of IL-1 and neutrophils, since both parameters showed little correlation with infectious virus production or disease.
4. Viruses can sensitize the lungs for a massive production of proinflammatory cytokines in response to LPS: the examples of PRCV and PRRSV
PRCV and PRRSV fail to induce multiple cytokines or respiratory disease by itself, but there are strong indications for their involvement in multifactorial respiratory disease. PRRSV in particular is considered a major player in the etiology of the so-called “porcine respiratory disease complex (PRDC)”. Unexpectedly, most experimental dual infections with PRRSV followed by bacteria were clinically mild or subclinical. In other dual infection studies, the clinical effects were extremely variable. This was also true for infections with PRRSV followed by a second virus, such as PRCV or SIV (reviewed in Van Reeth and Nauwynck, 2000). A critical evaluation shows that all of the published dual infection studies with PRRSV result in additive effects and not in a true potentiation of one infection by another.
In an attempt to study interactions between respiratory viruses and secondary agents in a reproducible way, we have performed subsequent inoculations of pigs with either PRCV or PRRSV followed by bacterial lipopolysaccharide (LPS). LPS is the biologically active component of endotoxin, a major constituent of the cell wall of Gram-negative bacteria. The choice of LPS was based on the following:
-
1.
Pigs are continually exposed to endotoxins in the respirable fraction of dust in swine confinement units. In addition, endotoxins are released locally in the lungs during pulmonary infections with Gram-negative bacteria.
-
2.
LPS does not multiply and standardized LPS preparations are commercially available. Combined inoculations with virus and LPS therefore may avoid the variability resulting from interference of a first virus with replication of a second virus or bacteria.
-
3.
LPS is an extremely potent cytokine inducer and exerts most of its biological effects through cytokines. Nearly all LPS effects are strictly dose-related, and relatively high LPS doses are required to induce cytokines and decreased lung function upon inhalation. Low LPS doses, on the other hand, are insufficient to induce cytokines and disease. It was our working hypothesis that PRCV or PRRSV may prime lung cells for enhanced secretion of early cytokines in response to minute amounts of LPS and thereby initiate disease.
In experiments with PRCV, gnotobiotic pigs were inoculated intratracheally with PRCV and 24 h later with LPS from E. coli 0111: B4 (Van Reeth et al., 2000). Fig. 2 compares the effects of the combined PRCV–24 h–LPS inoculation with those of the respective single inoculations. All pigs were euthanatized between 3 and 72 h after LPS, or at corresponding times after the single PRCV inoculation. The effects of separate virus or LPS inoculations were subclinical and failed to induce high and sustained cytokine levels. The combination of both agents, on the contrary, resulted in marked labored breathing, dullness and loss of appetite during the first 10–12 h after LPS. Prior infection with PRCV truly potentiated the cytokine response to LPS, with 10–100 times higher titres of TNF-α, IL-1 and IL-6 than after the respective single inoculations. Despite very high IFN-α titres in the dually inoculated pigs, similar titres may also occur after a single PRCV infection (Van Reeth et al., 1999). Experiments with different time intervals between PRCV and LPS inoculations have demonstrated a distinct time frame post-viral infection during which the synergy between virus and LPS is maximal. That is, typical respiratory disease was seen with 8–32 h intervals between virus and LPS, but not with shorter or longer intervals. Titres of TNF-α and IL-6 were also critically dependent on the time interval, and those cytokines were best correlated with disease. IL-1 and IFN-α, on the other hand, were not affected by the time interval and appear to play a minor if any role. The time interval experiments suggest that one virus replication cycle in the lungs must be completed at the time of LPS inoculation for enhanced disease and TNF-α/IL-6 production to occur.
Fig. 2.

Comparative profiles of respiratory signs, lung virus titres, neutrophils and cytokines in BAL fluids after inoculation with PRCV followed 24 h later by LPS, or with PRCV or LPS only.
Unexpectedly, there was no clear synergistic effect between PRCV and LPS with respect to lung pathological changes. Excessive neutrophil infiltration, oedema and haemorrhages were seen in both singly LPS and PRCV–LPS inoculated pigs, to an almost similar degree. Also, there was little correlation between these pathological features and disease or cytokines. These observations were surprising because a direct relationship between neutrophil sequestration in the lung and respiratory disorders has been demonstrated in many studies (Lentsch and Ward, 2001). One attractive hypothesis is that not the inflammation or structural lung damage but functional lung disturbances, such as bronchoconstriction or bronchial hyperresponsiveness, contribute to respiratory disease. Interestingly, TNF-α has been implicated in bronchial hyperresponsiveness in rats (Kips et al., 1992).
Our clinicopathological, virological and cytokine findings have been remarkably similar for the combination PRRSV and LPS. Still, PRRSV and PRCV differ completely from a pathogenetic point of view. Unlike PRCV, PRRSV has a specific tropism for lung macrophages and the infection produces a massive and long-lasting infiltration of the lungs with macrophage-like cells. Macrophages are pre-eminent producer cells of TNF-α and IL-1 upon in vitro LPS exposure. Because absolute numbers of macrophage-like cells increase approximately 5-fold after PRRSV infection, one would assume that the lungs contain more potentially LPS-responsive cells than after any other virus infection. Still, TNF-α, IL-1, as well as IL-6 profiles were remarkably similar to those seen in PRCV–LPS experiments. This raises questions about the importance of macrophages relative to other lung cells in virus–LPS induced cytokine responses. The in vivo situation is probably much more complicated than any in vitro system, and several types of lung cells may be involved. In contrast with the cytokines just mentioned, IFN-α levels were some 1000 times lower than in the PRCV–LPS model. PRRSV is a very poor inducer of IFN-α in itself (Albina et al., 1998, Van Reeth et al., 1999), and the subsequent LPS inoculation apparently did not affect IFN-α titres. This confirms the idea that IFN-α is less important in the pathogenesis of virus–LPS interactions.
Perhaps the most important difference between PRCV and PRRSV infection is the time frame during which the lungs are more responsive to LPS. Unlike PRCV, PRRSV persists in the lungs during at least 3–4 weeks. Experimental PRRSV–LPS inoculations with intervals varying from 3 up to 14 days all resulted in disease and cytokine production. It seems logical therefore that PRRSV will have a greater chance to interact with bacterial components in the field than PRCV. Consequently, we will use the PRRSV–LPS model to test preventive or therapeutic measures and study the role of cytokines in more detail. In ongoing experiments, we are studying the effect of treatment with pentoxifylline (PTX) on the clinical response to PRRSV–LPS. PTX, a phosphodiesterase inhibitor, has been documented to block the production of TNF-α, and may also reduce production of IL-1 and IL-6 (Neuner et al., 1994). Like for SIV, however, the true role of cytokines in PRRSV–LPS induced disease needs to be examined by specific anti-cytokine strategies.
5. Conclusive remarks
Though our studies have been merely descriptive, we were the first to demonstrate different cytokine patterns during different viral respiratory infections of swine and, most important, an association between cytokines and disease. The next step is to use more specific strategies to dissect the role of cytokines during specific viral infections. One important issue is that the same cytokines mediating disease may also regulate disease resistance. In fact, IFN-α has extremely potent antiviral effects, and in vitro antiviral effects have also been attributed to TNF-α and IL-1. Fever and neutrophils are part of the innate immune response to pathogens, and their suppression has proven deleterious in many instances. With the exception of IL-8, the early cytokines are also involved in specific immunity by activation of T cells, B cells and macrophages. In the case of SIV, virus clearance from the lungs and the development of a specific immune response occur within days. It is conceivable therefore that cytokines mediate both clinicopathological manifestations and the resolution of a SIV infection. It remains to be seen whether selected anti-cytokine strategies can improve disease without damaging normal immune mechanisms.
Swine provide a unique animal model for the study of cytokines in viral respiratory diseases of humans. The pathogenesis of influenza, for example, is very similar in swine and humans. Moreover, the same cytokines associated with flu symptoms in swine have been demonstrated in nasal lavage fluids of experimentally infected human volunteers (Hayden et al., 1998). The role of cytokines in pneumonia or lower respiratory tract disease, however, cannot be studied in humans. Also, the clinicopathological manifestations of influenza in the mouse model differ widely from those in natural virus hosts, and pigs are an alternative and valuable animal model. Interactions between respiratory viruses and endotoxins, though given very little attention, may also occur in humans. Respiratory coronaviruses, for example, cause subclinical infections of the respiratory tract of humans, and the inhalation of endotoxins has similar effects in humans and swine (Thorn, 2001). Finally, we can study some of the most exciting questions in pigs, such as: How do viruses induce the production of different cytokines? Which cells make cytokines during different virus infections and where? How do cytokines released in the lung exert systemic effects?
Acknowledgements
The research described here was financially supported by the Belgian Ministry of Agriculture. Kristien Van Reeth and Steven Van Gucht are fellows of the Fund for Scientific Research-Flanders (FWO-Vlaanderen). The authors thank Lieve Sys and Fernand De Backer for expert technical assistance.
References
- Albina E., Carrat C., Charley B. Interferon-alpha response to swine arterivirus (PoAV), the porcine reproductive and respiratory syndrome virus. J. Interf. Cytok. Res. 1998;18:485–490. doi: 10.1089/jir.1998.18.485. [DOI] [PubMed] [Google Scholar]
- Asai T., Okada M., Ono M., Irisawa T., Mori Y., Yokomizo Y., Sato S. Increased levels of tumor necrosis factor and interleukin-1 in bronchoalveolar lavage fluids from pigs infected with Mycoplasma hyopneumoniae. Vet. Immunol. Immunopathol. 1993;38:253–260. doi: 10.1016/0165-2427(93)90085-i. [DOI] [PubMed] [Google Scholar]
- Asai T., Okada M., Ono M., Mori Y., Yokomizo Y., Sato S. Detection of interleukin-6 and prostaglandin E2 in bronchoalveolar lavage fluids of pigs experimentally infected with Mycoplasma hyopneumoniae. Vet. Immunol. Immunopathol. 1994;44:97–102. doi: 10.1016/0165-2427(94)90172-4. [DOI] [PubMed] [Google Scholar]
- Baarsch M.J., Scamurra R.W., Burger K., Foss D.L., Maheswaran S.K., Murtaugh M.P. Inflammatory cytokine expression in swine experimentally infected with Actinobacillus pleuropneumoniae. Infect. Immun. 1995;63:3587–3594. doi: 10.1128/iai.63.9.3587-3594.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baarsch M.J., Foss D.L., Murtaugh M.P. Pathophysiologic correlates of acute porcine pleuropneumonia. Am. J. Vet. Res. 2000;61:684–690. doi: 10.2460/ajvr.2000.61.684. [DOI] [PubMed] [Google Scholar]
- Bogdan C. The function of type I interferons in antimicrobial immunity. Curr. Opin. Immunol. 2000;12:419–424. doi: 10.1016/s0952-7915(00)00111-4. [DOI] [PubMed] [Google Scholar]
- Choi C., Kwon D., Min K., Chae C. In situ hybridization for the detection of inflammatory cytokines (IL-1, TNF-α and IL-6) in pigs naturally infected with Actinobacillus pleuropneumoniae. J. Comp. Pathol. 1999;121:349–356. doi: 10.1053/jcpa.1999.0332. [DOI] [PubMed] [Google Scholar]
- Dinarello C.A. Proinflammatory cytokines. Chest. 2000;118:503–508. doi: 10.1378/chest.118.2.503. [DOI] [PubMed] [Google Scholar]
- Fossum C., Wattrang E., Fuxler L., Jensen K.T., Wallgren P. Evaluation of various cytokines (IL-6, IFN-α, IFN-γ, TNF-α) as markers for acute bacterial infection in swine—a possible role for serum interleukin-6. Vet. Immunol. Immunopathol. 1998;64:161–172. doi: 10.1016/s0165-2427(98)00126-3. [DOI] [PubMed] [Google Scholar]
- Harding J.C., Baarsch M.J., Murtaugh M.P. Association of tumour necrosis factor and acute phase reactant changes with post-arrival disease in swine. J. Vet. Med. B. 1997;44:405–413. doi: 10.1111/j.1439-0450.1997.tb00991.x. [DOI] [PubMed] [Google Scholar]
- Hayden F.G., Fritz R.S., Lobo M.C., Alvord W.G., Strober W., Straus S.E. Local and systemic cytokine responses during experimental human influenza A virus infection. J. Clin. Invest. 1998;101:643–649. [Google Scholar]
- Huang H., Potter A.A., Campos M., Leighton F.A., Willson P.J., Haines D.M., Yates W.D.G. Pathogenesis of porcine Actinobacillus pleuropneumonia. Part II. Roles of proinflammatory cytokines. Can. J. Vet. Res. 1999;63:69–78. [PMC free article] [PubMed] [Google Scholar]
- Kips J.C., Tavernier J., Pauwels R.A. Tumor necrosis factor causes bronchial hyperresponsiveness in rats. Am. Rev. Resp. Dis. 1992;145:332–336. doi: 10.1164/ajrccm/145.2_Pt_1.332. [DOI] [PubMed] [Google Scholar]
- Lentsch A.B., Ward P.A. Regulation of experimental lung inflammation. Resp. Physiol. 2001;128:17–22. doi: 10.1016/s0034-5687(01)00260-2. [DOI] [PubMed] [Google Scholar]
- Morrison D.F., Foss D.L., Murtaugh M.P. Interleukin-10 gene therapy-mediated amelioration of bacterial pneumonia. Infect. Immun. 2000;68:4752–4758. doi: 10.1128/iai.68.8.4752-4758.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murtaugh M.P., Baarsch M.J., Zhou Y., Scamurra R.W., Lin G. Inflammatory cytokines in animal health and disease. Vet. Immunol. Immunopathol. 1996;54:45–55. doi: 10.1016/s0165-2427(96)05698-x. [DOI] [PubMed] [Google Scholar]
- Nelson S., Bagby G.J., Bainton B.G., Wilson N.A., Thompson J.J., Summer W.R. Compartmentalization of intraalveolar and systemic lipopolysaccharide-induced tumor necrosis factor and the pulmonary inflammatory response. J. Infect. Dis. 1989;159:189–194. doi: 10.1093/infdis/159.2.189. [DOI] [PubMed] [Google Scholar]
- Neuner P., Klosner G., Schauer E., Pourmojib M., Macheiner W., Grunwald C., Knobler R., Schwarz A., Luger T.A., Schwarz T. Pentoxyfylline in vivo down-regulates the release of IL-1 beta, IL-6, IL-8 and tumour necrosis factor-alpha by human peripheral blood mononuclear cells. Immunology. 1994;83:262–267. [PMC free article] [PubMed] [Google Scholar]
- Strieter R.M., Kunkel S.L. Acute lung injury: the role of cytokines in the elicitation of neutrophils. J. Invest. Med. 1994;42:640–651. [PubMed] [Google Scholar]
- Thorn J. The inflammatory response in humans after inhalation of bacterial endotoxin: a review. Inflamm. Res. 2001;50:254–261. doi: 10.1007/s000110050751. [DOI] [PubMed] [Google Scholar]
- Van Reeth K., Nauwynck H. Proinflammatory cytokines and viral respiratory disease in pigs. Vet. Res. 2000;31:187–213. doi: 10.1051/vetres:2000113. [DOI] [PubMed] [Google Scholar]
- Van Reeth K., Labarque G., Nauwynck H., Pensaert M. Differential production of proinflammatory cytokines in the pig lung during different respiratory virus infections: correlations with pathogenicity. Res. Vet. Sci. 1999;67:47–52. doi: 10.1053/rvsc.1998.0277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Reeth K., Nauwynck H., Pensaert M. A potential role for tumour necrosis factor-α in synergy between porcine respiratory coronavirus and bacterial lipopolysaccharide in the induction of respiratory disease in pigs. J. Med. Microbiol. 2000;49:613–620. doi: 10.1099/0022-1317-49-7-613. [DOI] [PubMed] [Google Scholar]
- Van Reeth K., Labarque G., De Clercq S., Pensaert M. Efficacy of vaccination of pigs with different H1N1 swine influenza viruses using a recent challenge strain and different parameters of protection. Vaccine. 2001;19:4479–4486. doi: 10.1016/s0264-410x(01)00206-7. [DOI] [PubMed] [Google Scholar]


