Abstract
For more than 20 years, sepsis has been defined as symptoms associated with the response to microorganism infection, which was more specifically called systemic inflammatory response syndrome (SIRS). With the evidence of organ failure, it was called severe sepsis, and this could lead to hypotension (septic shock). However, with the deep understanding of the pathophysiology of sepsis, sepsis has been known as both inflammatory and anti-inflammatory. Additionally, the classic use of SIRS could lead to overestimation of sepsis. For example, usual common cold could be identified as sepsis in classic definition. With this background, new sepsis definition, Sepsis 3, was introduced and sepsis was defined as a “life-threatening organ dysfunction caused by a dysregulated host response to infection.” The management of sepsis has been changed dramatically, with the development of Surviving Sepsis Campaign, which substantially increased the survival of sepsis. However, this is not with the help of a new drug, but the implementation of a treatment system. Unfortunately, no specific drug for sepsis has survived in clinical use even though many candidate drugs have been successfully investigated in preclinical setting, and this leads to the new approach to the sepsis.
Introduction
Definition
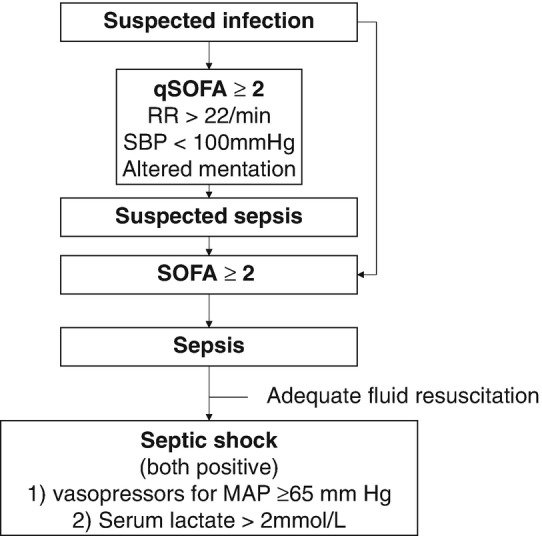
More than 20 years, sepsis was defined as symptoms associated with the response to microorganism infection, which was more specifically called systemic inflammatory response syndrome (SIRS). With the evidence of organ failure, it was called severe sepsis, and this could lead to hypotension (septic shock) [1]. However, with the deep understanding of the pathophysiology of sepsis, sepsis has been known as both inflammatory and anti-inflammatory. Additionally, the classic use of SIRS could lead to overestimation of sepsis. For example, usual common cold could be identified as sepsis in classic definition [2]. With this background, new sepsis definition, Sepsis 3, was introduced with expert consensus, literature review, and finally big data analysis. Sepsis was defined as a “life-threatening organ dysfunction caused by a dysregulated host response to infection.” They recommended the use of sequential organ failure assessment (SOFA) score as clinical criteria for sepsis in ICU encounters. More specifically, increase in SOFA score of two points or more should be used. In non-ICU encounters, quick SOFA score was recommended as screening tool, and qSOFA score of two or more was set to identify high-risk patients. The concept of classical severe sepsis was eliminated. Septic shock was newly defined as sepsis with fluid-unresponsive hypotension, serum lactate level greater than 2 mmol/L, and need for vasopressors to maintain mean arterial pressure of 65 mmHg or greater (Fig. 5.1) [3, 4].
Fig. 5.1.
Sepsis-3 Definition of sepsis and septic shock [4]
Epidemiology
Annual worldwide incidence of sepsis was estimated from 15–31 million, but this wide estimation stems from various methods used in calculation. This reflects vagueness and difficulty in sepsis definition. However, the incidence has been investigated to increase as consistently, and aging and more comorbid disease (cancer, diabetes, etc.) may affect this increase [5, 6].
No Magic Bullet as a Drug in Septic Shock Treatment
High disease burden in respect to the incidence, morbidity, and mortality has led to the extensive research about the septic shock, but unfortunately no drug has survived from clinical trials. This could mean the poor understanding of pathophysiology, different subphenotypes, diverse phases, etc. [5]. With more knowledge of septic shock, it is rapidly changing how to manage the septic shock, which would be briefly discussed.
Introduction to This Chapter
In this chapter, we introduce the pathophysiology, diagnostic approach, initial management, and future of septic shock. Considering the purpose of this book, i.e., scenario based, we do not cover all aspects of septic shock, but focus on the early management of septic shock.
Pathophysiology of Septic Shock
Introduction
Although septic shock is one of the distributive shocks, the pathophysiology of septic shock is different from other distributive shock diseases. The pathophysiology and the clinical course of septic shock are more complex and vary over the course of the disease, with variable degrees of intravascular volume depletion, peripheral vasodilation, and cardiac dysfunction.
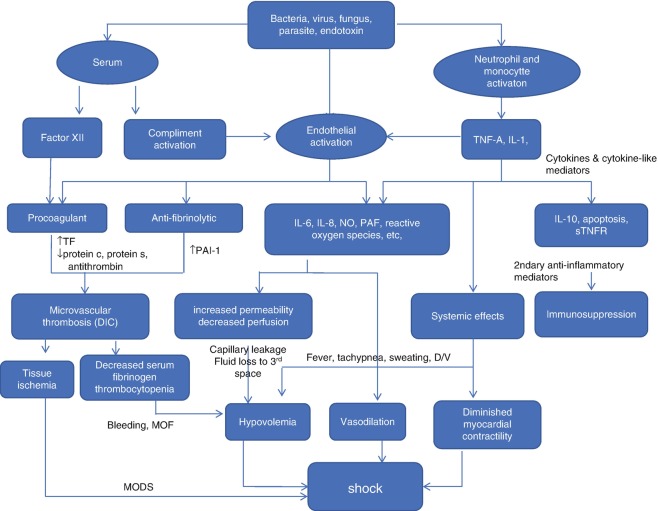
Septic shock can be produced by any microbial infection (bacteria, virus, fungus, parasites, etc.), even if no organism is identified at nearly 50% of cases of septic shock. Lipopolysaccharide (LPS) of gram-negative [G (−)] bacteria and lipoteichoic acid and peptidoglycan of gram-positive [G (+)] organisms are well-known mediators of sepsis.
Pathophysiology
Mediators
Against invading organism or its toxins, the early humoral responses are the complement system involvement and immune cell [monocytes/macrophages and polymorphonuclear neutrophils (PMNs), etc.] activation. Immune cells not only are able to recognize invading organisms and their products so they can destroy them but also release a series of mediators that can activate other cells. Toll-like receptors, among cell membrane receptors, are implicated in the recognition of pathogenic agents. Cellular stimulation activates intracellular signaling and results largely in the activation of transcriptional factors (including nuclear factor kappa B), which initiate proinflammatory reactions in turn. A number of cytokines, mainly tumor necrosis factor alpha (TNF-α) and interleukin (IL)-1, are released by macrophages and other cells. TNF-α and IL-1 are particularly important proinflammatory cytokines that can reproduce all features of septic shock including hypotension and development of multiple-organ failure (MOF) in animal studies. Anti-inflammatory mediators including IL-4 and IL-10 are also released during the sepsis response, which limit the effects of proinflammatory mediators and can lead to a state of relative immunosuppression sometimes called immunoparalysis [7]. When sepsis is diagnosed, many patients are already immunosuppressed [8].
Immune cells also release secondary host mediators including lipid mediators, oxygen free radicals, proteases, and arachidonic acid metabolites. Vasodilator substances such as nitric oxide (NO) and prostaglandins are released by endothelial cells and are responsible for the early hemodynamic changes of sepsis. The formation of large quantities of NO can also have secondary toxic effects on cells. NO can block mitochondrial respiration, directly by inhibiting cytochrome a and a3 and reacting with superoxide radicals, resulting in the production of peroxynitrite, which inhibits various phases of mitochondrial respiration [9] (Fig. 5.2).
Fig. 5.2.
The pathophysiology of septic shock
Hemodynamic Changes
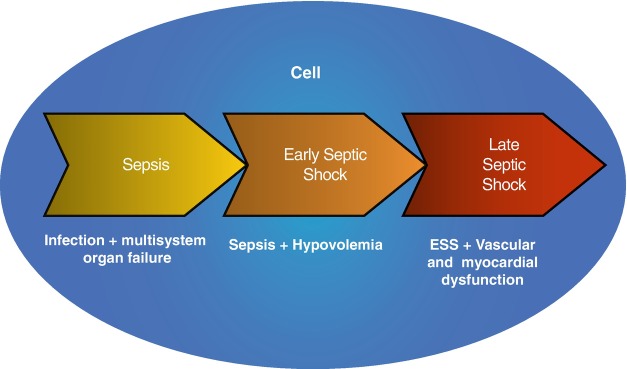
Widespread systemic inflammation likely plays a role in the development and persistence of multisystem organ failure in sepsis through microvascular and mitochondrial dysfunction. Although microcirculatory and mitochondrial dysfunction in sepsis and its links with organ dysfunction are not fully understood, they are likely to be key locus of hemodynamic compromise in septic shock [10, 11]. Septic shock is often due to three major causes: intravascular volume depletion, cardiac dysfunction, and peripheral vasodilation. And they also may be associated with microcirculatory and mitochondrial dysfunction (Fig. 5.2). Therefore, septic shock was recently classified, according to pathophysiological background, into two stages as follows: the early hypovolemic and the late vascular and myocardial circulatory dysfunction stage [12] (Fig. 5.3).
Fig. 5.3.
The phase of septic shock
Intravascular Volume Depletion
Sepsis produces hypovolemia, and the hypovolemia contributes partially to deterioration into septic shock. Hypovolemia in septic shock mainly results from gastrointestinal volume loss (diarrhea, vomiting), tachypnea, sweating, and decreased fluid intake during development of the illness.
The other auxiliary cause of hypovolemia in septic shock results from increasing capillary leak and resultant loss of intravascular volume into third spaces. In septic shock, adhesion molecules on vascular endothelium and circulating cells (platelets, PMNs, and monocytes) are expressed by inflammatory reaction. Activated leukocytes adhere to vascular endothelium and migrate to subendothelial tissues. Alterations in intercellular endothelial junctions result in increased capillary permeability and generalized edema [13].
Alterations in coagulation and fibrinolysis complete the picture, with proinflammatory mediators creating a procoagulant state. Briefly, the activation of tissue factor on the surface of various cells, particularly monocytes and endothelial cells, initiates the coagulation system [13]. Endotoxin, TNF-α, and IL-1 are the key mediators. The plasma levels of natural anticoagulants such as protein C, protein S, and antithrombin are significantly reduced in sepsis by reducing their synthesis and increasing their consumption and clearance. In early sepsis, thrombolysis is also stimulated with an increase in the levels of plasminogen activator inhibitor-1 (PAI-1). The net result is a balance in favor of procoagulant processes, often leading to DIC and participating in the microcirculatory disorder that leads to MOF in many patients with sepsis. This sequence of events leads to consumption of coagulation factors and the fibrinolytic system is suppressed in late sepsis and that promotes capillary leakage, bleeding, and third-space fluid shift (Fig. 5.2).
Cardiac Dysfunction
Cardiovascular dysfunction and failure arise from direct myocardial depression and distributive shock condition. Any microbes and killed organisms can cause myocardial depression. The direct effects of the toxic mediators as well as the knock-on effects of host mediators of sepsis produce a septic shock. Septic shock causes direct myocardial depression. Even in the hyperdynamic stages, cardiac contractility becomes impaired during the early phase of septic shock. Circulating mediators, inflammatory myocardial cellular injury, and deranged metabolism interact synergistically to injure the heart during septic shock, and specific cytokines (most notably TNF-α and IL-lß), overproduction of NO, and possibly impairment in mitochondrial respiration may contribute to the cardiovascular dysfunction.
Early in sepsis, although the cardiac output (CO) is increased, it is at the expense of ventricular dilation and decreased ejection fraction (EF). After vascular filling as a result of volume resuscitation, the hemodynamic status in septic shock is characterized by a fall in vascular tone associated with reduced systemic vascular resistance and a raised CO. Ejection volume and, particularly, CO may be maintained by an increase in diastolic volumes. Therefore, myocardial depression or dysfunction without any true cardiac failure would be developed and that may be associated with reduced cardiac output.
Peripheral Vasodilation
NO is a powerful vasodilator acting on vascular smooth muscle. Increased NO production in sepsis is essentially due to the induction of inducible NO synthase (iNOS) by proinflammatory cytokines. NO regulates vascular tone by an indirect effect on smooth muscle cells and that is important role in septic shock. NO also contributes to platelet adhesion, insulin secretion, neurotransmission, tissue injury, and cytotoxicity, and it seems to be a key mediator of septic shock although its mechanisms of action are not well understood. Enhanced NO production is thought to contribute to the profound vasodilation found in patients in septic shock.
Vasopressin is a naturally occurring hormone that is essential for cardiovascular stability. It is produced as a prohormone in the hypothalamus. The hormone is stored in the pituitary gland and released in response to stressors such as pain, hypoxia, hypovolemia, and hyperosmolality. There is a brief rise in circulating vasopressin levels in sepsis and followed by a prolonged and severe suppression in septic shock.
Septic Shock: Initial Approach and Diagnosis
Recognition of Sepsis Candidate
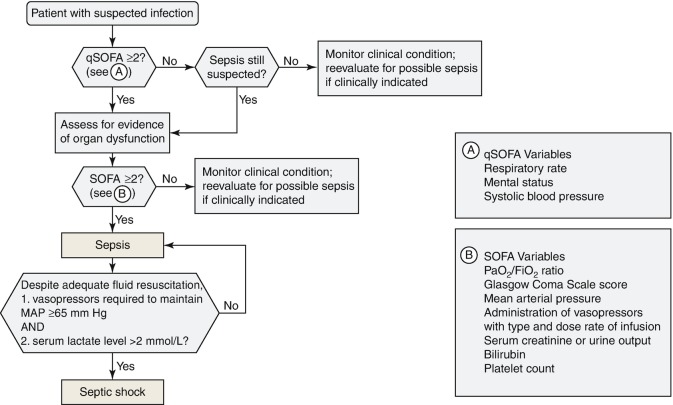
According to the new definition updated in 2016 published by “The Third International Consensus Definitions for Sepsis and Septic Shock” (Sepsis-3) in 2016, sepsis is a life-threatening organ dysfunction caused by a dysregulated host response to infection [4]. A clinical algorithm to identify patient with sepsis and septic shock was also proposed (Fig. 5.4). Sepsis screening is associated with earlier treatment because a lack of its recognition may prevent timely therapy [14]. A significant challenge in recognizing sepsis may be the complexity of the sepsis presentation. Notably, sepsis screening has been associated with survival improvement [15].
Fig. 5.4.
Operationalization of clinical criteria identifying patients with sepsis and septic shock. SOFA: Sepsis-related Organ Function Assessment. Modified from ref. [4]
Patients with Suspected Infection
The above algorithm starts from patient with suspected infection, which may include a wide range of patients. A prospective cohort study showed that 13% of patients admitted to the intensive care unit (ICU) with suspected sepsis had in fact no infection identified [16]. Such sepsis mimickers are consisted of adverse drug reaction, acute mesenteric ischemia, malignancies, and various sterile inflammatory diseases [17]. The presence of an infectious process is by definition necessary to discriminate between sepsis and sepsis mimickers. However, patients with sepsis mimickers have a clinical phenotype that resembles that of patients with sepsis, so that antimicrobial agents are usually administrated within the first hours of recognition of shock, until infection is ruled out and an alternative noninfectious diagnosis is made.
Quick SOFA Score
The quick Sepsis-related Organ Function Assessment (qSOFA) score was introduced as an initial screening tool to screen sepsis, which comprises a respiratory rate of 22/min or greater, altered mentation, or systolic blood pressure of 100 mmHg or less. If a patient has at least two of these criteria present, he or she could be identified as being more likely to have poor outcome of sepsis [4]. A patient with a qSOFA score of 2 or higher had a 3- to 14-fold increase in hospital mortality compared with a patient with a qSOFA score lower than 2 [18]. Compared with previous SIRS criteria, qSOFA had better discriminative value and hazard ratio for predicting death, ICU admission, and ICU stay longer than 72 h. The strong prognostic accuracy of qSOFA for mortality was confirmed with an area under the receiver operating characteristic (AUROC) of 0.80, which was greater than that of SIRS and severe sepsis (AUROC 0.65 for both) [19]. However, a recent prospective study reported that a qSOFA score of two or higher had high specificity for organ dysfunction but poor sensitivity (96.1% vs. 29.9%, respectively) [20].
Other Screening Tools
Many other potential clinical scoring systems could be used as a screening tool including simple triage scoring system (STSS), rapid emergency medicine score (REMS), modified early warning score (MEWS), and national early warning score (NEWS). They are easy to use because they only require simple physiological measures such as heart rate, blood pressure, respiratory rate, oxygen saturation, mental status, and urine output. These variables can easily be measured in primary care setting.
Several studies based on electronic screening tools have been reported in order to recognize sepsis without delay. A prospective observational study showed that an electronic alert was sent to the care team if two or more SIRS criteria were detected in patients older than 70 years. The system had a sensitivity of 14% and a specificity of 98% for detecting an infection [21]. Nelson et al. used an automated messaging system that alerted the care team if a patient presented to the ED with two or more SIRS criteria in addition to two systolic blood pressure readings of <90 mmHg. Their system had a sensitivity of 64% and specificity of 99% [22]. Sepsis performance improvement programs can be aimed at earlier recognition of sepsis via a formal screening effort and improved management of patients once they are identified as being septic.
Organ Dysfunction Assessment
The Sepsis-3 algorithm recommends to assess for evidence of organ dysfunction using the SOFA score for a patient with suspected sepsis who has a qSOFA score of two or higher. The SOFA score is based on six different scores, consisted of the respiratory, cardiovascular, hepatic, coagulation, renal, and neurological systems. A SOFA score of two points or more is associated with an in-hospital mortality greater than 10% and is therefore considered indicative for a patient with suspected infection having sepsis.
Besides SOFA score, there are some potential scores to assess organ dysfunctions in the emergency department setting such as Mortality in Emergency Department, Sepsis (MEDS); Confusion, Urea nitrogen, Respiratory rate, Blood pressure, 65 years and older (CURB-65); predisposition, infection, response, and organ dysfunction (PIRO); Acute Physiology and Chronic Health Evaluation (APACHE) scores; and Simplified Acute Physiology Score (SAPS) are more complicated scores to calculate, as they require for example the measurement of arterial oxygenation; therefore they are usually used in critical care settings. These scores could be used as a prognostic indicator to estimate the mortality of critically ill patients.
Clinical Manifestations
The septic patient may manifest signs of systemic infection. The symptoms and signs of sepsis are nonspecific but may include the following: temperature >38.3 or <36 °C, heart rate >90 beats/min, respiratory rate >20 breaths/min, and arterial hypotension (systolic blood pressure <90 mmHg or mean arterial pressure <70 mmHg). Symptoms and signs may be various and specific to an infectious source. For example, cough or dyspnea may suggest pneumonia, and pain and purulent exudate in a surgical wound may suggest an underlying abscess.
Warm, flushed skin may be present in the early phases of sepsis. As sepsis progresses to shock, the skin may become cool due to redirection of blood flow to core organs. Decreased capillary refill, cyanosis, or mottling may indicate shock. Additional signs of hypoperfusion include altered mental status, obtundation or restlessness, and oliguria or anuria. Ileus or absent bowel sounds are often an end-stage sign of hypoperfusion. These findings may be modified by preexisting disease or medications. For example, older patients, diabetic patients, and patients who take beta-blockers may not exhibit an appropriate tachycardia as blood pressure falls. In contrast, younger patients frequently develop a severe and prolonged tachycardia and fail to become hypotensive until acute decompensation later occurs. Patients with chronic hypertension may develop critical hypoperfusion at a higher blood pressure than healthy patients (relative hypotension). Importantly, the presentation is nonspecific such that many other conditions such as pancreatitis and acute respiratory distress syndrome may present similarly.
Adequate Fluid Resuscitation
By definition of Sepsis-3, the identification of patients with septic shock includes the requirement of vasopressors to maintain a mean arterial pressure of 65 mmHg or greater and the presence of serum lactate levels greater than 2 mmol/L despite adequate fluid resuscitation [4]. Therefore, we need to administrate adequate fluid to identify the patients with septic shock as well as shock management.
Controversy for EGDT
Early effective fluid resuscitation is crucial for stabilization of sepsis-induced tissue hypoperfusion or septic shock. Sepsis-induced hypoperfusion may be manifested by acute organ dysfunction and/or decreased blood pressure and increased serum lactate. Previous guidelines have recommended a protocolized quantitative resuscitation, also known as early goal-directed therapy (EGDT) [23]. This approach has been challenged following the failure to show a mortality reduction in large RCTs such as ARISE, PROCESS, and PROMISE trial [24–26]. Although, the EGDT protocol cannot now be recommended from its evidence base, bedside clinicians still need guidance as to how to approach patients suspicious of septic shock who have significant mortality and morbidity.
Volume of Fluid for Initial Resuscitation
Surviving Sepsis Campaign (SSC) guideline recommended that these patients be viewed as having a medical emergency that necessitates urgent assessment and management [27]. As part of this, guideline recommends that initial fluid resuscitation begin with 30 mL/kg of crystalloid within the first 3 h. This fixed volume of fluid enables clinicians to initiate resuscitation while obtaining more specific information about the patient and while awaiting more precise measurements of hemodynamic status. The average volume of fluid before randomization given in the PROCESS and ARISE trials was approximately 30 mL/kg, and approximately 2 L in the PROMISE trial [24–26].
Optimal Fluid Type for Initial Resuscitation
The optimal fluid in the initial resuscitation of sepsis is not yet clear. Solutions containing water and freely permeable ions, mainly sodium and chloride, are classified as crystalloids. Balanced solutions are usually defined as intravenous fluids having an electrolyte composition close to that of plasma. Balanced crystalloids such as Plasma-Lyte solution are probably superior to normal saline, but further prospective studies are warranted [28].
Albumin appears to be equivalent to crystalloids in terms of outcomes, but should be second-line due to higher cost. Among patients with sepsis, several randomized trials and meta-analyses have reported no difference in mortality when albumin was compared with crystalloids, although one meta-analysis suggested benefit in those with septic shock [29–31]. In the Saline versus Albumin Fluid Evaluation (SAFE) trial performed in critically ill patients, there was no benefit to albumin compared with saline even in the subgroup with severe sepsis, who comprised 18% of the total group [32]. Hydroxyethyl starches (HES) appear to increase mortality and acute kidney injury in critically ill septic patients and are no longer indicated in the treatment of this patient population. In the Scandinavian Starch for Severe Sepsis and Septic Shock (6S) trial, compared with Ringer’s acetate, use of HES resulted in increased mortality (51% vs. 43%) and renal replacement therapy (22% vs. 16%) [33]. There is very limited clinical data regarding the use of hypertonic saline and no data regarding the use of plasma in sepsis.
Assessment of Fluid Responsiveness
Unnecessary fluid administration in the treatment of shock can increase morbidity and mortality, whereas selective yet timely use of fluids has shown to be beneficial. For adequate fluid resuscitation, physicians should assess the fluid responsiveness which is the ability of cardiac output to increase in response to a fluid infusion. Static measures of preload such as filling pressures and volumes are poor predictors of fluid responsiveness. The use of CVP alone to guide fluid resuscitation can no longer be justified because the ability to predict a response to a fluid challenge when the CVP is within a relatively normal range (8–12 mmHg) is limited [34].
Therefore, dynamic indices which measure the hemodynamic response of the cardiovascular system to a controlled variation in preload have been introduced in clinical practice. The most investigated dynamic indices such as the pulse pressure variation measure the response to preload variations induced by mechanical ventilation. They are good predictors of fluid responsiveness, but they lose their value in patients with spontaneous breathing activity and arrhythmias.
Passive Leg-Raising Test
To overcome the above limitations, the passive leg raising (PLR) has been proposed as an alternative preload-modifying maneuver. PLR induces a rapid, reversible increase in biventricular preload through an increase in venous return mimicking fluid administration. The reported amount of volume “autotransfused” by PLR ranges from 250 to 350 mL. Although PLR induces an increase in cardiac preload with its maximum effect at approximately 1 min, the effect is not sustained and vanishes completely when the legs are returned to the horizontal position. Thus, the hemodynamic effects of PLR must be assessed during a time frame of 30–90 s with a fast-responding method. No difference in diagnostic performance of PLR was seen in spontaneously breathing patients compared with controlled mechanically ventilated patients. Therefore, PLR has been proposed as an attractive way to predict fluid responsiveness and showed good diagnostic accuracy in meta-analysis [35]. The pooled sensitivity and specificity of PLR-induced changes in cardiac output were 89.4% (84.1–93.4%) and 91.4% (85.9–95.2%), respectively.
Serum Lactate Measurement
According to the Sepsis-3 definition, presence of serum lactate levels greater than 2 mmol/L is a component to diagnose septic shock. The 3-h bundle protocol recommends measurement of the serum lactate level to complete within 3 h. Serum lactic acid levels have long been identified as a diagnostic tool for global tissue hypoxia and therefore can serve in identifying patients with sepsis. However, increases in the serum lactate level may represent accelerated aerobic glycolysis driven by excess beta-adrenergic stimulation, or other causes including liver failure. Regardless of the source, increased lactate levels are associated with worse outcomes [36]. Several randomized controlled trials have evaluated lactate-guided resuscitation of patients with septic shock [27, 37, 38]. A significant reduction in mortality was seen in lactate-guided resuscitation compared to resuscitation without lactate monitoring (RR 0.67; 95% CI, 0.53–0.84). There was no evidence for difference in ICU length of stay (mean difference −1.51 days; 95% CI, −3.65 to 0.62).
Lactate Clearance
Another meta-analysis demonstrated reduction in mortality when an early lactate clearance strategy was used, compared with either usual care or a ScvO2 normalization strategy [39, 40]. The lactate clearance was defined by the equation [(lactateinitial − lactatedelayed)/lactateinitial] × 100%, for which lactateinitial was the measurement at the start of the resuscitation and lactatedelayed was another measurement after a minimum of 2 h after resuscitation was initiated [38]. It was shown that lactate clearance greater than 10% from initial measurement during the first 2–6 h of resuscitation predicted survival from septic shock and that protocols targeting lactate clearance of at least 10% produced similar short-term survival rates to protocols using ScvO2 monitoring [38, 41]. Moreover, it was demonstrated that for every 10% increase in lactate clearance, there was a corresponding 11% decrease in in-hospital mortality [42]. Similarly, septic patients with lactate clearance of greater than 20% during the initial 8 h of resuscitation had a 22% decline in the relative risk of mortality, compared with patients having lactate clearances of less than 20% [37]. However, there was no association between degree of lactate clearance and change in microcirculatory blood flow in patients with septic shock [43].
Laboratory Findings of Sepsis
An ideal biomarker can be objectively measured and could play a role in sepsis screening, early diagnosis, risk stratification, critical assessment, and prognosis prediction. Numerous biomarkers have been studied for use in diagnosis or prognosis for sepsis. They consist of cytokines, cell markers, receptors, coagulation markers, acute-phase proteins, markers related to vascular endothelial damage, vasodilation and organ dysfunction, and so on [44]. Until now more than 170 different biomarkers have been assessed for potential use in sepsis. Several studies have proposed that measurement of multiple cytokines such as TNF-α, IL-1β, and IL-6 correlates well with disease severity and prognosis of sepsis. However, most of the cytokines and inflammatory mediators were of limited use in clinical setting.
C-Reactive Protein
The C-reactive protein (CRP) was identified as a protein responsible for precipitating C polysaccharide during the acute phase of Streptococcus pneumonia infection. Its response is stronger in acutely ill patients; levels decrease as patients recover. These characteristics make CRP a member of the class of acute-phase reactants. CRP is an old biomarker and widely used in clinical settings. It is a nonspecific marker of inflammation that also increases after surgery, burns, myocardial infarctions, and rheumatic diseases. According to a meta-analysis, the sensitivity and specificity of CRP as a marker for bacterial infections are 81% and 67%, respectively [45]. Its low specificity and inability to differentiate bacterial infections from noninfectious causes of inflammation make CRP of limited diagnostic value. However, CRP shows promise for evaluating sepsis severity and prognosis. CRP plasma levels have been shown to correlate with the severity of infection [46]. A rapid decrease in CRP levels has been reported to correlate with good response to initial antimicrobial therapy in septic patients [47]. CRP is a useful biomarker to monitor treatment response. However, CRP is not recommended to use in the day of sepsis diagnosis as a marker of prognosis and risk stratification [48].
Procalcitonin
Procalcitonin (PCT) is a precursor of calcitonin, a calcium-regulatory hormone secreted from thyroid tissue in healthy individuals. In infectious conditions, PCT is released from all tissues including lung, liver, kidney, pancreas, and adipose tissues. In a systematic review, PCT was found to be more specific (specificity 81%) than CRP (67%) for differentiating bacterial infection among hospitalized patients [45]. The cutoff value of PCT for diagnosis of sepsis has not yet been fully determined. PCT values need to be further evaluated according to different sites of infection, hosts, and pathogens. Another recent meta-analysis showed that PCT is a useful marker for early diagnosis of sepsis in critically ill patients, with sensitivity and specificity of 77% (95% CI: 72–81%) and 79% (95% CI: 74–84%), respectively [49]. PCT levels are also elevated after surgery, cardiogenic shock, heat shock, acute graft-versus-host disease, and immunotherapy such as granulocyte transfusion, which could limit its usefulness as a sepsis biomarker.
Antibiotic Stewardship with Procalcitonin
PCT has also drawn attention because it can be used for guidance of antibiotic stewardship to reduce inappropriate use of antibiotics [50]. Procalcitonin-guided therapy is defined as initiation of antibiotic treatment using PCT measurements, usually using a suggested treatment algorithm based on the height of the PCT measurement. Many studies used a cutoff of 0.25 ng/mL to suggest or encourage the initiation of antibiotics [51]. Multiple studies have investigated whether a PCT-guided algorithm can optimize the therapeutic approach in sepsis patients, mainly by monitoring PCT kinetics and stopping antibiotics once PCT has dropped to levels <0.5 ng/mL or by at least 80–90% of the peak in combination with clinical improvement. A meta-analysis including 1075 patients with sepsis or septic shock found overall reduced antibiotic treatment courses (6 days vs. 8 days) when PCT was used to guide therapy compared to routine care. There was no increase in 28-day or in-hospital mortality or in length of stay in the ICU or the hospital. The authors do stress that there was heterogeneity in PCT protocols across trials with regard to different cutoff values or different algorithms for medical or surgical patients [52]. A large RCT evaluated the use of PCT to de-escalate and stop antibiotics in critically ill patients who had received antibiotics <24 h before inclusion in the study for an assumed or a proven infection. The study found that the PCT-guided protocol shortened length of antibiotic treatment (5 days vs. 7 days in the first 28 days of admission) and lowered 28-day mortality from 25% to 19.6% [53].
Microbiologic Cultures
Blood cultures should be obtained prior to initiating antimicrobial therapy if cultures can be obtained in a timely manner. Several retrospective studies have suggested that obtaining cultures prior to antimicrobial therapy is associated with improved outcome [54]. However, the identification of an organism in culture in a patient with suspected sepsis is highly supportive of the diagnosis but is not necessary. The rationale behind its lack of inclusion in the diagnostic criteria for sepsis is that a culprit organism is frequently not identified in up to 50% of patients who present with sepsis nor is a positive culture required to make a decision regarding treatment with empiric antibiotics. Therefore, the desire to obtain cultures prior to initiating antimicrobial therapy should be balanced against the mortality risk of delaying a definitive therapy in patients with suspected sepsis or septic shock who are at significant risk of death [55].
Appropriate microbiologic cultures should be obtained before initiation of antimicrobial therapy from all sites considered to be potential sources of infection if it results in no substantial delay in the start of antimicrobials. This may include blood, cerebrospinal fluid, urine, wounds, respiratory secretions, and other body fluids. Two or more sets (aerobic and anaerobic) of blood cultures are recommended before initiation of any new antimicrobial in all patients with suspected sepsis [56]. All necessary blood cultures may be drawn together on the same occasion. Blood culture yield has not been shown to be improved with sequential draws or timing to temperature spikes [57].
Management of Septic Shock
Initial Resuscitation
- Basal concept
- To optimize tissue perfusion within 6 h post-diagnosis
- To acquire the balance between the oxygen consumption (VO2) and the oxygen delivery (DO2) within 6 h post-diagnosis, but there have been no methods to estimate VO2 during the acute phase of sepsis [27]. Therefore, the acute management of sepsis should be focused to increase DO2 [58]. The formula to calculate DO2 is as follows:
- DO2 ≈ CO × 1.34 × [Hb] × SaO2
- To increase DO2, cardiac output, hemoglobin concentration, and arterial oxygen saturation should be increased [59]. When adequate oxygen is provided, an increase in cardiac output can be a primary target for an increase in DO2.
- Cardiac output is calculated by stroke volume multiplied by heart rate. And the three components which contribute to make a stroke volume are preload, afterload, and myocardial contractility. Therefore, the basal concept of initial management to acquire optimal cardiac output is to maintain optimal preload, afterload, and contractility with an appropriate range of heart rate within 6 h post-diagnosis [23].
- First Preload Optimization
- The definition of preload for shock management is the end-diastolic volume of left ventricle. Preload is affected by venous pressure and venous return [60].
- To increase venous return, first of all, adequate hydration (30 mL/kg of crystalloid) should be provided within 3 h [27].
- To estimate volume status, many parameters are monitored. Because the direct measurement of left ventricular volume is impossible, most of the parameters monitor pressure. Commonly used parameters are static pressures which reflect left ventricle end-diastolic pressure. If the pulmonary circulation system would be intact, central venous pressure (CVP) is well correlated with pulmonary capillary wedge pressure (PCWP) and left ventricular end-diastolic pressure [61]. Because of the ease of accessibility and safety, CVP is the most commonly used parameter for static volume status in clinical setting [62]. However, many of the critically ill patients have structural or functional problems in pulmonary circulation system; CVP plays a limited role in the estimation of acute-phase volume status in patients with septic shock [63, 64]. And thus, alternative parameters including the diameter and/or collapsibility of inferior vena cava (IVC) are tested [65, 66].
- Initial hydration is the most important process to optimize preload and acquire appropriate tissue perfusion. But overhydration which induces fluid overload resulting in tissue edema and additional microcirculatory dysfunction should be avoided [67, 68]. Previous studies reported that fluid overload contributed to an increase in the mortality of patients with septic shock [69]. Therefore, the repeated measurements of dynamic fluid responsiveness should be combined with the measurement of static volume status.
- The parameters monitoring static volume status have another limitation. They cannot reflect dynamic fluid responsiveness during initial resuscitation [61]. Therefore, adjunctive methods to predict dynamic fluid responsiveness, such as pulse pressure variation (PPV > 13%), stroke volume variation (SVV > 12%), passive leg raising (PLR), and mini-fluid challenge (stroke volume index >6% after 100 mL of fluid challenge), have been developed and applied [70–76]. Among them, PPV and SVV should be measured in patients with full sedation and positive pressure ventilation and without dysrhythmia, and the use of them in emergency department is limited [71–74].
- The estimation of volume status and fluid responsiveness during fluid resuscitation is not easy in clinical setting. The combination of various monitoring parameters using all of the presently available devices may be helpful to estimate volume status more accurately and to optimize preload in patients with septic shock.
- Second After Optimization
- The definition of afterload for shock management is the pressure in the wall of the left ventricle during ejection. If aortic valvular function is maintained a systemic vascular resistance contributes to afterload to maintain blood flow to peripheral tissues.
- To maintain systemic vascular resistance, the infusion of vasopressors is recommended.
- To estimate afterload, mean arterial pressure (MAP) is commonly used. To measure more reliable MAP values, a routine intra-arterial catheterization in radial artery is recommended in patients with septic shock [27].
- Third consider myocardial contractility augmentation with an appropriate range of heart rate
- Myocardial contractility is the innate ability of the myocardium and an energy-consuming process. Furthermore, tachycardia also aggravates the energy-consuming process in myocardium. These energy-consuming processes need high oxygen supply to maintain the balance between VO2 and DO2.
- However, in patients who have acquired optimal preload and afterload but still have low cardiac output, the use of inotropic agents may be considered under the monitoring of stepwise changes in tissue perfusion with inotropic agents and heart rate [27].
- Hemoglobin
- To transport oxygen to peripheral tissues, adequate hemoglobin level should be maintained. There have been controversies about the optimal target hemoglobin level. When hemoglobin concentration is 15.0 g/dL, only a half of circulating hemoglobin participates in oxygen transport [81]. Furthermore, recent large clinical studies have shown that a transfusion threshold of the hemoglobin level of 7.0 g/dL was not different from 9.0 g/dL in the mortality of patients with septic shock [25, 82].
Surviving Sepsis Campaign Bundle
To optimize tissue perfusion with 6 h post-diagnosis, the current guideline recommends bundle therapies which should be completed within 3 h and 6 h, respectively (Table 5.1) [27, 84].
Table 5.1.
Surviving sepsis campaign bundle
| To be completed within 3 h | Measure lactate level |
| Obtain blood culture prior to administration of antibiotics | |
| Administer broad-spectrum antibiotics | |
| Administer 30 mL/kg crystalloid for hypotension or lactate ≥4 mmol/L | |
| To be completed within 6 h | Apply vasopressors for hypotension that does not respond to initial fluid resuscitation to maintain MAP ≥65 mmHg |
| Remeasure lactate if initial lactate was elevated | |
| In persistent arterial hypotension despite volume resuscitation or initial lactate ≥4 mmol/L, RE-access volume status and tissue perfusion and document findings according to | |
| Repeat focused exam with respect to vital signs including mental status, cardiopulmonary including urination, capillary refill, pulse including tachycardia, and skin findings such as mottled skin | |
|
Or two of the following: (1) Measure CVP: at least ≥8 mmHg (2) Measure ScvO2: ≥70% (3) Bedside cardiovascular ultrasound (4) Dynamic fluid responsiveness with PLR or mini-fluid challenge |
Fluid Therapy
Until now, the current guideline recommends the use of crystalloid for initial fluid resuscitation in patients with septic shock rather than colloid. Many previous studies reported that the use of colloid during initial resuscitation induces acute kidney injury and increases the mortality in patients with septic shock [33, 85].
The current guideline recommends the use of crystalloid, both the 0.9% saline and the balanced crystalloids, such as Ringer’s lactate solution and Plasma-Lyte. However, some studies showed that because of hyperchloremic metabolic acidosis, balanced crystalloids may be better than 0.9% saline for initial resuscitation (Table 5.2) [86, 87].
To optimize preload, adequate fluid therapy should be provided. However, a sustained positive fluid balance after initial resuscitation has been reported to be harmful [88, 89]. Therefore, after preload optimization, fluid therapy should be cautiously performed with various monitoring devices to estimate the present volume status of patients.
Although there have been no data supporting the amount of fluid volume after the 30 mL/kg of initial fluid resuscitation in patients with septic shock, the 30 mL/kg of initial fluid resuscitation, check volume status, additional fluid challenge (500 mL q 30 min) till CVP reaches ≥8 mmHg, recheck volume status with 100 mL of mini-fluid challenge, 150 mL of fluid infusion, and repeating the 100 mL of mini-fluid challenge and 150 mL of fluid infusion may be helpful to acquire optimal volume status.
Recent clinical studies have shown the benefits of 5% albumin administration [32, 90]. Albumin administration can reduce fluid balance in septic shock patients who require substantial amounts of crystalloids. However, in patients with severe pneumonia or brain pathologies, the routine use of albumin should be avoided because the leakage of albumin can induce edema in lung or brain resulting in clinical deterioration [32].
Table 5.2.
Common crystalloids
| Plasma | 0.9% saline | Ringer’s lactate | Plasma-Lyte | |
|---|---|---|---|---|
| Sodium (mmol/L) | 136–145 | 154 | 130 | 140 |
| Potassium (mmol/L) | 3.5–5.0 | 0 | 4 | 5 |
| Magnesium (mmol/L) | 0.8–1.0 | 0 | 0 | 1.5 |
| Calcium (mmol/L) | 2.2–2.6 | 0 | 3 | 0 |
| Chloride (mmol/L) | 98–106 | 154 | 109 | 98 |
| Lactate (mmol/L) | 0 | 0 | 28 | 0 |
Vasoactive Agents
After optimized preload, to maintain afterload (MAP ≥65 mmHg) and cardiac contractility, vasoactive agents can be added. The commonly used vasoactive agents and their properties are described in Table 5.3 [91].
The current guideline recommends to use norepinephrine as the first-choice vasopressor [27]. In previous studies, dopamine might induce tachyarrhythmia and deteriorated clinical outcomes in critically ill patients [92–94]. Therefore, dopamine can be considered for highly selected patients with low risk of tachyarrhythmia and absolute or relative bradycardia.
If with the maximal dose of norepinephrine MAP failed to reach >65 mmHg, low doses of vasopressin or epinephrine can be added to norepinephrine [95, 96]. Vasopressin acts not to α-receptor but to V-receptor, and the deficiency of vasopressin may contribute to the vasodilation of septic shock [97]. The use of vasopressin can be helpful to decrease norepinephrine dosage.
The use of dobutamine may be considered only in patients with persistent hypoperfusion despite adequate administrations of fluids and vasopressors [24, 25, 80].
Table 5.3.
Common vasoactive agents
| Agents | Heart | Vasculature | Cardiac output | MAP | HR | Dose range | |
|---|---|---|---|---|---|---|---|
| β1 | α1 | α2 | (μg/kg/min) | ||||
| Dopamine | 0–3+ | 0–3+ | 0–2+ | ↑ | ↑ | ↑↑ | 2–20 |
| Dobutamine | 4+ | 1+ | 2+ | ↑↑ | ↔ | ↑ | 2.5–10.0 |
| Epinephrine | 4+ | 2–4+ | 1–3+ | ↑↑ | ↑↑ | ↑↑ | 0.005–0.200 |
| Norepinephrine | 2+ | 4+ | 1+ | ↔ | ↑↑ | ↑ | 0.04–1.00 |
| Vasopressin | Vasopressin receptor | ↓ | ↑ | ↔ | 0.01–0.04 U/min | ||
Antimicrobial Therapy
Antibiotics are the most important factor to improve survival in patients with septic shock. Particularly, IV antibiotics should be administered within 1 h after presenting symptoms and signs of septic shock [98, 99].
- We suggest the following three principles for the use of antibiotics in the emergency department for patients with septic shock.
- Blood culture samples should be acquired before antibiotics administration. And thus, blood culture should be done within 45 min after presenting symptoms and signs of septic shock so as NOT TO delay antibiotics administration [27].
- Accelerating appropriate antimicrobial therapy may cause a delay in the start of antibiotic therapy [100]. The combination therapy of two or more of broad-spectrum antibiotics should be provided as an empiric antimicrobial therapy. Then, according to the identified pathogens and clinical improvement, the empiric antimicrobial therapy should be de-escalated.
- Imaging studies should be considered to find infection source, and if indicated invasive source control including radiologic interventions and/or surgery should be performed as soon as possible (within 12 h presenting symptoms and signs of septic shock).
Others
- Glucose control:
- To avoid hypoglycemic events, the target upper blood glucose level should be maintained ≤180 mg/dL [101].
- Corticosteroid:
- In recent large clinical studies, a routine use of corticosteroid failed to improve survival in patients with septic shock [102, 103]. Corticosteroid showed a benefit in the reversal of shock in patients who did not achieve hemodynamic stability in spite of adequate fluid resuscitation and vasopressor therapy [103].
Summary
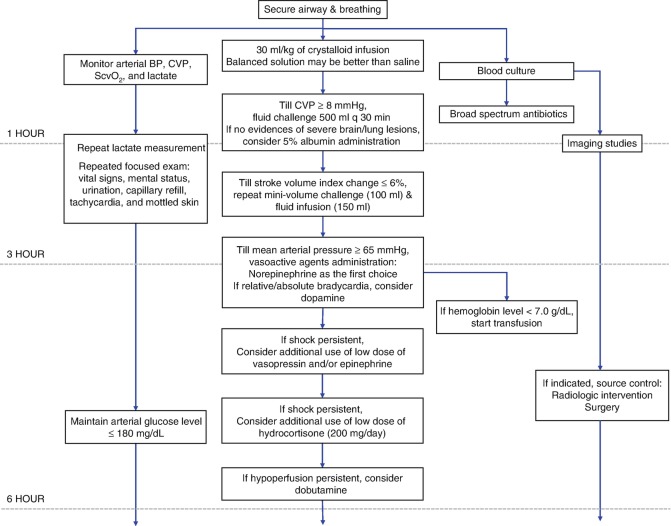
See Fig. 5.5
Fig. 5.5.
Initial management of septic shock
Future of Sepsis Management
Introduction: Current Problem of Sepsis Management
Definition/Epidemiology
The challenge for the management of sepsis might be originated from the “vagueness” of definition of sepsis. For a long time, the sepsis has been considered as one identity, which proved to be wrong. The sepsis could not be a “disease,” but a simple symptom or response to more specific diseases, e.g., pneumonia, urinary tract infection, and biliary infection. Antibiotics treatment for sepsis should be different according to the severity, site of infection, or organisms of resistance. Other treatments of sepsis, such as vasopressor, steroid, and immune-targeting drugs, should also be chosen to specific conditions of sepsis. Taken together, sepsis should not be managed with one magic bullet even though some basic management (for example, early fluid management) could be same.
The wide epidemiologic range of sepsis over the world or even in same country could also be explained by various definitions of sepsis, which could make it difficult to see the trend or effect of treatment when tested in large clinical trials.
Preclinical Study: In Vitro/In Vivo Study
No drug has survived in final clinical trials, although many preclinical studies showed promising results. The gap could be explained in many aspects. The species-specific pathway of sepsis could be one reason. Usually, mouse has been used to see the effects of drugs or pathway of sepsis, and although many pathways are same in both rodents and humans there are different pathways or their significances between rodents and humans, which could make little translation into clinical outcome [106]. Also, the design of preclinical study should be mentioned. For example, lipopolysaccharide (LPS) model has little chance to be translated into human study [107]. No patients visit to the emergency department with LPS injection. With this background, recently the cecal ligation and puncture model has gained popularity. However, the diverse method used in this model could affect the translation, and efforts to standardize the method are under progress (Minimum Quality Threshold in Pre-clinical Sepsis Studies, MQTiPSS). Considering cecal ligation and puncture model, the animals usually get the fluid and antibiotics [107]. In real clinical situation, however, we perform source control and administer vasopressor, or ventilator care, if necessary as well as just one-shot antibiotics and fluid [108]. These affect clinical course dramatically. In these days, a few research centers run mouse or rat ICU, and this might help translation of preclinical research to clinical practice.
Phase and Severity
As mentioned above, the phase and severity of sepsis are definitely different among sepsis patients. One drug could be beneficial in phase A, but not or even harmful in phase B. One good example could be steroid [109]. If patients are in hyperinflammatory status, it could work, but in opposite situation it could be harmful. The use of vasopressin in septic shock is also interesting. As shown in VAAST trial, vasopressin could be beneficial in less severe septic shock without effects in more severe one [95].
Clinical Trial Design
Recently, previous clinical trial design has been challenged. Clinical trial design should reflect the real world, not just digging the statistics or so. With this background, more realistic design is emerging, i.e., platform trial or REMAP trial [110]. ACCESS trial has already adopted this design, and we hope this shows paradigm shift in the design of clinical trial.
Protocol-Based Bundle Management
Early Recognition: EMS/ED
Previously, and maybe nowadays, the management of sepsis has been ICU based. Now we understand that the early management of sepsis is mandatory. Supranormal oxygen delivery concept by Dr. Shoemaker failed [111], but the nearly same concept, early goal-directed therapy (EGDT) by Dr. Rivers, has saved many septic patients all over the world [23]. Actually, EGDT is the early application of supranormal oxygen delivery. We should manage the patients not in irreversible condition, which could already occur in ICU. Likewise, the management of sepsis could start in EMS or even at home. To do that, we should implement the early recognition and management protocol, which could be used in prehospital setting, or at home.
Fluid Management
Appropriate Fluid
Fluid resuscitation in sepsis is the mainstay in sepsis management. However, the issues regarding which fluid is more appropriate should be determined. Usually balanced crystalloids are favored [86, 87, 112, 113], but the pitfall of these fluids should be overcome, e.g., potassium containment. The role of albumin should be more specified since the current guideline is a little confusing (albumin could be used after “substantial” amount of crystalloid, but what is the definition of “substantial”?) [27]. However, this could be hard to prove in current clinical design [114], and again we need another platform of clinical study.
How Much Amount of Fluid?
Classically, preload has been measured with central venous pressure or pulmonary capillary wedge pressure, which has proved to be inappropriate parameters. Current recommendation for determining preload status (passive leg raising, dynamic parameters, such as pulse pressure variation, stroke volume variation, ultrasonographic parameters, or small volume challenges) has pros and cons [115], and the optimal and feasible method to see the fluid responsiveness should be explored with the more advanced technology.
Target Goal of Hemodynamics
Recently, the important clinical study showed that the target blood pressure in sepsis is between mean arterial pressure of 65 and 85 mmHg [116]. However, shown in this study, in chronic hypertension patients, the 80–85 mmHg target could be more optimal. Simple and unique target of blood pressure in sepsis has little chance of reflecting the optimal tissue perfusion, and in that sense individualized approach is ideal, which is not available in current practice. To achieve this, new technologies to monitor the optimal tissue perfusion directly should be investigated and validated.
Antibiotics
Initial Appropriate Antibiotics: Organism Isolation
Early appropriate administration of antibiotics is of paramount importance in sepsis management [27, 117]. The choice of appropriate antibiotics needs the identification of the specific microorganism. Currently, the isolation of organism takes a long time. With classical blood or body fluid culture, it takes at least 2–3 days [118], which could determine the fate of septic patients. More disappointingly, the yield of blood culture is not high [119]. Recently, the genome-based detection method has been introduced, but the diagnostic performance was not yet satisfied. A new method to isolate the specific microorganism or even resistant species is being investigated, but currently not in clinical use.
Maintenance Duration
The use of antibiotics has disadvantages such as rising resistant organisms and antibiotics-associated secondary infection. Determining the optimal duration of antibiotics is of great concern in this aspect. Currently, predetermined duration of antibiotics administration or the use of clinical information, e.g., fever, or some biomarker (CPR, procalcitonin), has been advocated [120–122], but not in a sufficient way. New biomarker-based approach is of need to be developed.
New Antibiotics
Recently, and rapidly, the multidrug-resistant bacteria have emerged over the world. The new super-antibiotics for these nagging bacteria are desperately necessary. Also, various, life-threatening viral infections need to have attention, but there are little antiviral agents.
Biomarker or Phenotype-Driven Management: Personalized Management
Sepsis is one of the perfect targets for the precision medicine. Sepsis is a group of diverse infected patients, and has dynamic change during short time. This means it needs a different approach from other diseases, such as cancer. In cancer, single measurement of subphenotype could lead to the specific treatment, but, in sepsis, the status could be different between today and tomorrow, or even change within hours. Considering that, the immediately available biomarker for specific conditions is of paramount importance. Even though not in sepsis, the opposite response to treatment according to the subphenotype of ARDS is very intriguing and could show the insight into sepsis management [123]. The effect of high PEEP or conservative fluid management was opposite with different subphenotypes in ARDS study, and this was validated using another big network sample [124]. Interestingly, the different subphenotypes were determined with inflammatory/anti-inflammatory cytokines, which have been used to see the phase of sepsis for a long time. Taken together, the same approach should be implemented in sepsis, and this approach could have huge impact on future sepsis management.
Promising Novel Therapeutic Strategies
New Vasopressor
Vasopressin
In VAAST trial, the use of vasopressin compared to norepinephrine showed no difference in survival. However, in less shock patients, the use of vasopressin showed lower mortality [95]. In sub-study, the combined use of steroid proved to be more beneficial. Vasopressin in sepsis should be more investigated in two aspects. One is in less severe septic shock, and another is to use vasopressin as hormone, not just as vasopressor. Vasopressin showed bimodal secretion in many stressful situations, such as shock [125]. Vasopressin has many functions depending on the type of receptors. Sepsis is a stressful condition, and the role of vasopressin as stress hormone could have more effects than only as vasopressor. Regarding this, the strategy to use the continuous constant use of vasopressin as hormone and optimizing norepinephrine as vasopressor could be beneficial in sepsis.
Selepressin
Selepressin is the new selective V1a receptor agonist, and this drug has been investigated in septic shock, showing promising results. Currently phase 2b/3 clinical trial has been in progress [126].
Angiotensin II
Recent randomized clinical trial has been successfully performed to see the effects of angiotensin II in refractory to high-dose vasopressor. The primary outcome (MAP response) was achieved, and without statistical significance the trend to better survival was shown [127]. A larger trial would be anticipated.
Endothelial Homeostasis
Endothelial homeostasis has been known to be important in sepsis. Global increased permeability syndrome represents endothelial breakage and this could lead to multiple-organ dysfunction [128], which is the main concern of sepsis. The drug to maintain the endothelial homeostasis could lead to less organ injury, and finally survival benefit. For example, recently developed angiopoietin II antagonist showed dramatic effects on sepsis outcomes in preclinical study [129].
Immune Suppressor/Enhancer
Historically, the drugs to suppress cytokine storm have been used to treat sepsis. However, all drugs failed [130, 131]. This might not mean that there is no role in immune suppressor, but specific time point to use immune suppressor is mandatory. With biomarkers, we could define the status of septic patients whether the patients are under hyper-inflammation or immune paralysis. With this information, we could use immune suppressor or enhancer as appropriate. Recent study showed beneficial effect of immunostimulants [132] and many trials to use these agents are under investigation.
Mitochondrial Target
Mitochondria have gained much attention in sepsis. Mitochondria are very important in both bioenergetics and ROS production. Mitochondrial dysfunction or cytopathic hypoxia is one of the most important pathophysiologies of late sepsis [133]. Mitochondria-targeted treatment needs monitoring of mitochondrial function, and this could be real time based since the cellular status of sepsis changes dynamically, or superdynamically. There is no clinically available device for this purpose, but it is under investigation. The target of drug to maintain the function of mitochondria could be in control of mPTP opening, mitochondrial ROS scavenger, specific respiratory complex chain, etc.
Sympathetic/Parasympathetic Intervention
Brain and immune system has been known to be closely connected, especially via cholinergic pathway. Autonomic interventions to treat sepsis have been intensively investigated [134], but no drug or intervention has clinical translation. However, in the near future, it could be used. For example, esmolol in sepsis had promising clinical results [135], and we could see the results of large clinical trial.
Stem Cell
Likewise in other areas of medicine, stem cell research has been extensively under way in sepsis. For example, mesenchymal stromal cell has been studied and showed some beneficial effects [136], and the various mechanisms were proposed (direct effects, paracrine effects, extracellular vesicles, etc.). More extensive research is expected to investigate the effects of stem cells on the outcomes in sepsis.
Sepsis: From Vital Sign to Brain
Sepsis-associated long-term cognitive dysfunction has been investigated and the results are a little shocking [137]. Mild Alzheimer disease-level dysfunction has been shown, and the effects are diverse from medical to socioeconomic aspect. To target the brain resuscitation in sepsis, the extensive pathophysiologic studies should be done, and this area is underdeveloped. Recently, the hippocampus could be an important area in sepsis-associated cognitive impairment, and less dendrite in CA1 was implicated as an important pathophysiology [138].
References
- 1.American College of Chest Physicians Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20(6):864–874. doi: 10.1097/00003246-199206000-00025. [DOI] [PubMed] [Google Scholar]
- 2.Jacob JA. New sepsis diagnostic guidelines shift focus to organ dysfunction. JAMA. 2016;315(8):739–740. doi: 10.1001/jama.2016.0736. [DOI] [PubMed] [Google Scholar]
- 3.Shankar-Hari M, Phillips GS, Levy ML, Seymour CW, Liu VX, Deutschman CS, et al. Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for sepsis and septic shock (sepsis-3) JAMA. 2016;315(8):775–787. doi: 10.1001/jama.2016.0289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3) JAMA. 2016;315(8):801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cohen J, Vincent J-L, Adhikari NKJ, Machado FR, Angus DC, Calandra T, et al. Sepsis: a roadmap for future research. Lancet Infect Dis. 2015;15(5):581–614. doi: 10.1016/S1473-3099(15)70112-X. [DOI] [PubMed] [Google Scholar]
- 6.Adhikari NK, Fowler RA, Bhagwanjee S, Rubenfeld GD. Critical care and the global burden of critical illness in adults. Lancet. 2010;376(9749):1339–1346. doi: 10.1016/S0140-6736(10)60446-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hamers L, Kox M, Pickkers P. Sepsis-induced immunoparalysis: mechanisms, markers, and treatment options. Minerva Anestesiol. 2015;81:426–439. [PubMed] [Google Scholar]
- 8.Davenport EE, Burnham KL, Radhakrishnan J, Humburg P, Hutton P, Mills TC, Rautanen A, Gordon AC, Garrard C, Hill AVS, Hinds CJ, Knight JC. Genomic landscape of the individual host response and outcomes in severe sepsis. Lancet Respir Med. 2016;4:259–271. doi: 10.1016/S2213-2600(16)00046-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Radi R. Peroxynitrite, a stealthy biological oxidant. J Biol Chem. 2013;288:26464–26472. doi: 10.1074/jbc.R113.472936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moore JP, Dyson A, Singer M, Fraser J. Microcirculatory dysfunction and resuscitation: why, when, and how. Br J Anaesth. 2015;115(3):366–375. doi: 10.1093/bja/aev163. [DOI] [PubMed] [Google Scholar]
- 11.Spronk PE, Zandstra DF, Ince C. Bench-to-bedside review: sepsis is a disease of the microcirculation. Crit Care. 2004;8:462–468. doi: 10.1186/cc2894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hernandez G, Bruhn A, Castro R, Regueira T. The holistic view on perfusion monitoring in septic shock. Curr Opin Crit Care. 2012;18(3):280–286. doi: 10.1097/MCC.0b013e3283532c08. [DOI] [PubMed] [Google Scholar]
- 13.Simmons J, Pittet JF. The coagulopathy of acute sepsis. Curr Opin Anaesthesiol. 2015;28:227–236. doi: 10.1097/ACO.0000000000000163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gatewood MO, Wemple M, Greco S, Kritek PA, Durvasula R. A quality improvement project to improve early sepsis care in the emergency department. BMJ Qual Saf. 2015;24:787–795. doi: 10.1136/bmjqs-2014-003552. [DOI] [PubMed] [Google Scholar]
- 15.Jones SL, Ashton CM, Kiehne L, et al. Reductions in sepsis mortality and costs after design and implementation of a nurse-based early recognition and response program. Jt Comm J Qual Patient Saf. 2015;41:483–491. doi: 10.1016/S1553-7250(15)41063-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Klein Klouwenberg PM, Cremer OL, van Vught LA, et al. Likelihood of infection in patients with presumed sepsis at the time of intensive care unit admission: a cohort study. Crit Care. 2015;19:319. doi: 10.1186/s13054-015-1035-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Contou D, Roux D, Jochmans S, et al. Septic shock with no diagnosis at 24 hours: a pragmatic multicenter prospective cohort study. Crit Care. 2016;20:360. doi: 10.1186/s13054-016-1537-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of clinical criteria for sepsis: for the third international consensus definitions for sepsis and septic shock (sepsis-3) JAMA. 2016;315:762–774. doi: 10.1001/jama.2016.0288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Freund Y, Lemachatti N, Krastinova E, et al. Prognostic accuracy of sepsis-3 criteria for in-hospital mortality among patients with suspected infection presenting to the emergency department. JAMA. 2017;317:301–308. doi: 10.1001/jama.2016.20329. [DOI] [PubMed] [Google Scholar]
- 20.Williams JM, Greenslade JH, McKenzie JV, Chu K, Brown AF, Lipman J. Systemic inflammatory response syndrome, quick sequential organ function assessment, and organ dysfunction: insights from a prospective database of ED patients with infection. Chest. 2017;151:586–596. doi: 10.1016/j.chest.2016.10.057. [DOI] [PubMed] [Google Scholar]
- 21.Meurer WJ, Smith BL, Losman ED, et al. Real-time identification of serious infection in geriatric patients using clinical information system surveillance. J Am Geriatr Soc. 2009;57:40–45. doi: 10.1111/j.1532-5415.2008.02094.x. [DOI] [PubMed] [Google Scholar]
- 22.Nelson JL, Smith BL, Jared JD, Younger JG. Prospective trial of real-time electronic surveillance to expedite early care of severe sepsis. Ann Emerg Med. 2011;57:500–504. doi: 10.1016/j.annemergmed.2010.12.008. [DOI] [PubMed] [Google Scholar]
- 23.Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368–1377. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- 24.Peake SL, Delaney A, Bailey M, et al. Goal-directed resuscitation for patients with early septic shock. N Engl J Med. 2014;371:1496–1506. doi: 10.1056/NEJMoa1404380. [DOI] [PubMed] [Google Scholar]
- 25.Yealy DM, Kellum JA, Huang DT, et al. A randomized trial of protocol-based care for early septic shock. N Engl J Med. 2014;370:1683–1693. doi: 10.1056/NEJMoa1401602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mouncey PR, Osborn TM, Power GS, et al. Trial of early, goal-directed resuscitation for septic shock. N Engl J Med. 2015;372:1301–1311. doi: 10.1056/NEJMoa1500896. [DOI] [PubMed] [Google Scholar]
- 27.Rhodes A, Evans LE, Alhazzani W, et al. Surviving sepsis campaign: International guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45:486–552. doi: 10.1097/CCM.0000000000002255. [DOI] [PubMed] [Google Scholar]
- 28.Raghunathan K, Bonavia A, Nathanson BH, et al. Association between initial fluid choice and subsequent in-hospital mortality during the resuscitation of adults with septic shock. Anesthesiology. 2015;123:1385–1393. doi: 10.1097/ALN.0000000000000861. [DOI] [PubMed] [Google Scholar]
- 29.Caironi P, Tognoni G, Masson S, et al. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med. 2014;370:1412–1421. doi: 10.1056/NEJMoa1305727. [DOI] [PubMed] [Google Scholar]
- 30.Xu JY, Chen QH, Xie JF, et al. Comparison of the effects of albumin and crystalloid on mortality in adult patients with severe sepsis and septic shock: a meta-analysis of randomized clinical trials. Crit Care. 2014;18:702. doi: 10.1186/s13054-014-0702-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jiang L, Jiang S, Zhang M, Zheng Z, Ma Y. Albumin versus other fluids for fluid resuscitation in patients with sepsis: a meta-analysis. PLoS One. 2014;9:e114666. doi: 10.1371/journal.pone.0114666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Finfer S, Bellomo R, Boyce N, French J, Myburgh J, Norton R. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med. 2004;350:2247–2256. doi: 10.1056/NEJMoa040232. [DOI] [PubMed] [Google Scholar]
- 33.Perner A, Haase N, Guttormsen AB, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med. 2012;367:124–134. doi: 10.1056/NEJMoa1204242. [DOI] [PubMed] [Google Scholar]
- 34.Eskesen TG, Wetterslev M, Perner A. Systematic review including re-analyses of 1148 individual data sets of central venous pressure as a predictor of fluid responsiveness. Intensive Care Med. 2016;42:324–332. doi: 10.1007/s00134-015-4168-4. [DOI] [PubMed] [Google Scholar]
- 35.Cavallaro F, Sandroni C, Marano C, et al. Diagnostic accuracy of passive leg raising for prediction of fluid responsiveness in adults: systematic review and meta-analysis of clinical studies. Intensive Care Med. 2010;36:1475–1483. doi: 10.1007/s00134-010-1929-y. [DOI] [PubMed] [Google Scholar]
- 36.Casserly B, Phillips GS, Schorr C, et al. Lactate measurements in sepsis-induced tissue hypoperfusion: results from the Surviving Sepsis Campaign database. Crit Care Med. 2015;43:567–573. doi: 10.1097/CCM.0000000000000742. [DOI] [PubMed] [Google Scholar]
- 37.Jansen TC, van Bommel J, Schoonderbeek FJ, et al. Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial. Am J Respir Crit Care Med. 2010;182:752–761. doi: 10.1164/rccm.200912-1918OC. [DOI] [PubMed] [Google Scholar]
- 38.Jones AE, Shapiro NI, Trzeciak S, Arnold RC, Claremont HA, Kline JA. Lactate clearance vs central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial. JAMA. 2010;303:739–746. doi: 10.1001/jama.2010.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Simpson SQ, Gaines M, Hussein Y, Badgett RG. Early goal-directed therapy for severe sepsis and septic shock: a living systematic review. J Crit Care. 2016;36:43–48. doi: 10.1016/j.jcrc.2016.06.017. [DOI] [PubMed] [Google Scholar]
- 40.Gu WJ, Zhang Z, Bakker J. Early lactate clearance-guided therapy in patients with sepsis: a meta-analysis with trial sequential analysis of randomized controlled trials. Intensive Care Med. 2015;41:1862–1863. doi: 10.1007/s00134-015-3955-2. [DOI] [PubMed] [Google Scholar]
- 41.Arnold RC, Shapiro NI, Jones AE, et al. Multicenter study of early lactate clearance as a determinant of survival in patients with presumed sepsis. Shock. 2009;32:35–39. doi: 10.1097/SHK.0b013e3181971d47. [DOI] [PubMed] [Google Scholar]
- 42.Nguyen HB, Rivers EP, Knoblich BP, et al. Early lactate clearance is associated with improved outcome in severe sepsis and septic shock. Crit Care Med. 2004;32:1637–1642. doi: 10.1097/01.CCM.0000132904.35713.A7. [DOI] [PubMed] [Google Scholar]
- 43.Puskarich MA, Shapiro NI, Massey MJ, Kline JA, Jones AE. Lactate clearance in septic shock is not a surrogate for improved microcirculatory flow. Acad Emerg Med. 2016;23:690–693. doi: 10.1111/acem.12928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pierrakos C, Vincent JL. Sepsis biomarkers: a review. Crit Care. 2010;14:R15. doi: 10.1186/cc8872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Simon L, Gauvin F, Amre DK, Saint-Louis P, Lacroix J. Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: a systematic review and meta-analysis. Clin Infect Dis. 2004;39:206–217. doi: 10.1086/421997. [DOI] [PubMed] [Google Scholar]
- 46.Povoa P, Coelho L, Almeida E, et al. C-reactive protein as a marker of infection in critically ill patients. Clin Microbiol Infect. 2005;11:101–108. doi: 10.1111/j.1469-0691.2004.01044.x. [DOI] [PubMed] [Google Scholar]
- 47.Schmit X, Vincent JL. The time course of blood C-reactive protein concentrations in relation to the response to initial antimicrobial therapy in patients with sepsis. Infection. 2008;36:213–219. doi: 10.1007/s15010-007-7077-9. [DOI] [PubMed] [Google Scholar]
- 48.Silvestre J, Povoa P, Coelho L, et al. Is C-reactive protein a good prognostic marker in septic patients? Intensive Care Med. 2009;35:909–913. doi: 10.1007/s00134-009-1402-y. [DOI] [PubMed] [Google Scholar]
- 49.Wacker C, Prkno A, Brunkhorst FM, Schlattmann P. Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13:426–435. doi: 10.1016/S1473-3099(12)70323-7. [DOI] [PubMed] [Google Scholar]
- 50.Kopterides P, Siempos II, Tsangaris I, Tsantes A, Armaganidis A. Procalcitonin-guided algorithms of antibiotic therapy in the intensive care unit: a systematic review and meta-analysis of randomized controlled trials. Crit Care Med. 2010;38:2229–2241. doi: 10.1097/CCM.0b013e3181f17bf9. [DOI] [PubMed] [Google Scholar]
- 51.van der Does Y, Rood PP, Haagsma JA, Patka P, van Gorp EC, Limper M. Procalcitonin-guided therapy for the initiation of antibiotics in the ED: a systematic review. Am J Emerg Med. 2016;34:1286–1293. doi: 10.1016/j.ajem.2016.03.065. [DOI] [PubMed] [Google Scholar]
- 52.Prkno A, Wacker C, Brunkhorst FM, Schlattmann P. Procalcitonin-guided therapy in intensive care unit patients with severe sepsis and septic shock—a systematic review and meta-analysis. Crit Care. 2013;17:R291. doi: 10.1186/cc13157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.de Jong E, van Oers JA, Beishuizen A, et al. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis. 2016;16:819–827. doi: 10.1016/S1473-3099(16)00053-0. [DOI] [PubMed] [Google Scholar]
- 54.Cardoso T, Carneiro AH, Ribeiro O, Teixeira-Pinto A, Costa-Pereira A. Reducing mortality in severe sepsis with the implementation of a core 6-hour bundle: results from the Portuguese community-acquired sepsis study (SACiUCI study) Crit Care. 2010;14:R83. doi: 10.1186/cc9008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ferrer R, Artigas A, Suarez D, et al. Effectiveness of treatments for severe sepsis: a prospective, multicenter, observational study. Am J Respir Crit Care Med. 2009;180:861–866. doi: 10.1164/rccm.200812-1912OC. [DOI] [PubMed] [Google Scholar]
- 56.Weinstein MP, Reller LB, Murphy JR, Lichtenstein KA. The clinical significance of positive blood cultures: a comprehensive analysis of 500 episodes of bacteremia and fungemia in adults. I. Laboratory and epidemiologic observations. Rev Infect Dis. 1983;5:35–53. doi: 10.1093/clinids/5.1.35. [DOI] [PubMed] [Google Scholar]
- 57.Baron EJ, Miller JM, Weinstein MP, et al. A guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2013 recommendations by the Infectious Diseases Society of America (IDSA) and the American Society for Microbiology (ASM)(a) Clin Infect Dis. 2013;57:e22–e121. doi: 10.1093/cid/cit278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vincent JL, De Backer D. Circulatory shock. N Engl J Med. 2013;369(18):1726–1734. doi: 10.1056/NEJMra1208943. [DOI] [PubMed] [Google Scholar]
- 59.Baigorri F, Russell JA. Oxygen delivery in critical illness. Crit Care Clin. 1996;12(4):971–994. doi: 10.1016/S0749-0704(05)70287-6. [DOI] [PubMed] [Google Scholar]
- 60.Luecke T, Roth H, Herrmann P, Joachim A, Weisser G, Pelosi P, Quintel M. Assessment of cardiac preload and left ventricular function under increasing levels of positive end-expiratory pressure. Intensive Care Med. 2004;30(1):119–126. doi: 10.1007/s00134-003-1993-7. [DOI] [PubMed] [Google Scholar]
- 61.Marik PE, Monnet X, Teboul JL. Hemodynamic parameters to guide fluid therapy. Ann Intensive Care. 2011;1(1):1. doi: 10.1186/2110-5820-1-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb SA, Beale RJ, Vincent JL, Moreno R, Surviving Sepsis Campaign Guidelines Committee including the Pediatric Subgroup Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580–637. doi: 10.1097/CCM.0b013e31827e83af. [DOI] [PubMed] [Google Scholar]
- 63.Osman D, Ridel C, Ray P, Monnet X, Anguel N, Richard C, Teboul JL. Cardiac filling pressures are not appropriate to predict hemodynamic response to volume challenge. Crit Care Med. 2007;35(1):64–68. doi: 10.1097/01.CCM.0000249851.94101.4F. [DOI] [PubMed] [Google Scholar]
- 64.Marik PE, Cavallazzi R. Does the central venous pressure predict fluid responsiveness? An updated meta-analysis and a plea for some common sense. Crit Care Med. 2013;41(7):1774–1781. doi: 10.1097/CCM.0b013e31828a25fd. [DOI] [PubMed] [Google Scholar]
- 65.Prekker ME, Scott NL, Hart D, Sprenkle MD, Leatherman JW. Point-of-care ultrasound to estimate central venous pressure: a comparison of three techniques. Crit Care Med. 2013;41(3):833–841. doi: 10.1097/CCM.0b013e31827466b7. [DOI] [PubMed] [Google Scholar]
- 66.Muller L, Bobbia X, Toumi M, Louart G, Molinari N, Ragonnet B, Quintard H, Leone M, Zoric L, Lefrant JY, AzuRea Group Respiratory variations of inferior vena cava diameter to predict fluid responsiveness in spontaneously breathing patients with acute circulatory failure: need for a cautious use. Crit Care. 2012;16(5):R188. doi: 10.1186/cc11672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wang CH, Hsieh WH, Chou HC, Huang YS, Shen JH, Yeo YH, Chang HE, Chen SC, Lee CC. Liberal versus restricted fluid resuscitation strategies in trauma patients: a systematic review and meta-analysis of randomized controlled trials and observational studies. Crit Care Med. 2014;42(4):954–961. doi: 10.1097/CCM.0000000000000050. [DOI] [PubMed] [Google Scholar]
- 68.Jozwiak M, Silva S, Persichini R, Anguel N, Osman D, Richard C, Teboul JL, Monnet X. Extravascular lung water is an independent prognostic factor in patients with acute respiratory distress syndrome. Crit Care Med. 2013;41(2):472–480. doi: 10.1097/CCM.0b013e31826ab377. [DOI] [PubMed] [Google Scholar]
- 69.Micek ST, McEvoy C, McKenzie M, Hampton N, Doherty JA, Kollef MH. Fluid balance and cardiac function in septic shock as predictors of hospital mortality. Crit Care. 2013;17(5):R246. doi: 10.1186/cc13072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mackenzie DC. Noble VE2. Assessing volume status and fluid responsiveness in the emergency department. Clin Exp Emerg Med. 2014;1(2):67–77. doi: 10.15441/ceem.14.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Marik PE, Cavallazzi R, Vasu T, Hirani A. Dynamic changes in arterial waveform derived variables and fluid responsiveness in mechanically ventilated patients: a systematic review of the literature. Crit Care Med. 2009;37(9):2642–2647. doi: 10.1097/CCM.0b013e3181a590da. [DOI] [PubMed] [Google Scholar]
- 72.Guinot PG, de Broca B, Abou Arab O, Diouf M, Badoux L, Bernard E, Lorne E, Dupont H. Ability of stroke volume variation measured by oesophageal Doppler monitoring to predict fluid responsiveness during surgery. Br J Anaesth. 2013;110(1):28–33. doi: 10.1093/bja/aes301. [DOI] [PubMed] [Google Scholar]
- 73.De Backer D, Heenen S, Piagnerelli M, Koch M, Vincent JL. Pulse pressure variations to predict fluid responsiveness: influence of tidal volume. Intensive Care Med. 2005;31(4):517–523. doi: 10.1007/s00134-005-2586-4. [DOI] [PubMed] [Google Scholar]
- 74.Soubrier S, Saulnier F, Hubert H, Delour P, Lenci H, Onimus T, Nseir S, Durocher A. Can dynamic indicators help the prediction of fluid responsiveness in spontaneously breathing critically ill patients? Intensive Care Med. 2007;33(7):1117–1124. doi: 10.1007/s00134-007-0644-9. [DOI] [PubMed] [Google Scholar]
- 75.Biais M, de Courson H, Lanchon R, Pereira B, Bardonneau G, Griton M, Sesay M, Nouette-Gaulain K. Mini-fluid challenge of 100 ml of crystalloid predicts fluid responsiveness in the operating room. Anesthesiology. 2017;127(3):450–456. doi: 10.1097/ALN.0000000000001753. [DOI] [PubMed] [Google Scholar]
- 76.Guinot PG, Bernard E, Defrancq F, Petiot S, Majoub Y, Dupont H, Lorne E. Mini-fluid challenge predicts fluid responsiveness during spontaneous breathing under spinal anaesthesia: An observational study. Eur J Anaesthesiol. 2015;32(9):645–649. doi: 10.1097/EJA.0000000000000175. [DOI] [PubMed] [Google Scholar]
- 77.Leisman DE, Goldman C, Doerfler ME, Masick KD, Dries S, Hamilton E, Narasimhan M, Zaidi G, D’Amore JA, D’Angelo JK. Patterns and outcomes associated with timeliness of initial crystalloid resuscitation in a prospective sepsis and septic shock cohort. Crit Care Med. 2017;45(10):1596–1606. doi: 10.1097/CCM.0000000000002574. [DOI] [PubMed] [Google Scholar]
- 78.Leisman D, Wie B, Doerfler M, Bianculli A, Ward MF, Akerman M, D’Angelo JK, Zemmel D'Amore JA. Association of fluid resuscitation initiation within 30 minutes of severe sepsis and septic shock recognition with reduced mortality and length of stay. Ann Emerg Med. 2016;68(3):298–311. doi: 10.1016/j.annemergmed.2016.02.044. [DOI] [PubMed] [Google Scholar]
- 79.Sperry JL, Minei JP, Frankel HL, West MA, Harbrecht BG, Moore EE, Maier RV, Nirula R. Early use of vasopressors after injury: caution before constriction. J Trauma. 2008;64(1):9–14. doi: 10.1097/TA.0b013e31815dd029. [DOI] [PubMed] [Google Scholar]
- 80.Mouncey PR, Osborn TM, Power GS, Harrison DA, Sadique MZ, Grieve RD, Jahan R, Harvey SE, Bell D, Bion JF, Coats TJ, Singer M, Young JD, Rowan KM. ProMISe Trial Investigators. Trial of early, goal-directed resuscitation for septic shock. N Engl J Med. 2015;372(14):1301–1311. doi: 10.1056/NEJMoa1500896. [DOI] [PubMed] [Google Scholar]
- 81.Weiskopf RB, Viele MK, Feiner J, Kelley S, Lieberman J, Noorani M, Leung JM, Fisher DM, Murray WR, Toy P, Moore MA. Human cardiovascular and metabolic response to acute, severe isovolemic anemia. JAMA. 1998;279(3):217–221. doi: 10.1001/jama.279.3.217. [DOI] [PubMed] [Google Scholar]
- 82.Holst LB, Haase N, Wetterslev J, Wernerman J, Guttormsen AB, Karlsson S, Johansson PI, Aneman A, Vang ML, Winding R, Nebrich L, Nibro HL, Rasmussen BS, Lauridsen JR, Nielsen JS, Oldner A, Pettilä V, Cronhjort MB, Andersen LH, Pedersen UG, Reiter N, Wiis J, White JO, Russell L, Thornberg KJ, Hjortrup PB, Müller RG, Møller MH, Steensen M, Tjäder I, Kilsand K, Odeberg-Wernerman S, Sjøbø B, Bundgaard H, Thyø MA, Lodahl D, Mærkedahl R, Albeck C, Illum D, Kruse M, Winkel P, Perner A, TRISS Trial Group; Scandinavian Critical Care Trials Group Lower versus higher hemoglobin threshold for transfusion in septic shock. N Engl J Med. 2014;371(15):1381–1391. doi: 10.1056/NEJMoa1406617. [DOI] [PubMed] [Google Scholar]
- 83.Hébert PC, Yetisir E, Martin C, Blajchman MA, Wells G, Marshall J, Tweeddale M, Pagliarello G, Schweitzer I, Transfusion Requirements in Critical Care Investigators for the Canadian Critical Care Trials Group Is a low transfusion threshold safe in critically ill patients with cardiovascular diseases? Crit Care Med. 2001;29(2):227–234. doi: 10.1097/00003246-200102000-00001. [DOI] [PubMed] [Google Scholar]
- 84.Surviving Sepsis Campaign Bundles-Revised 4/2015 by the SSC Executive Committee. http://www.survivingsepsis.org/SiteCollectionDocuments/SSC_Bundle.pdf.
- 85.Myburgh JA, Finfer S, Bellomo R, Billot L, Cass A, Gattas D, Glass P, Lipman J, Liu B, McArthur C, McGuinness S, Rajbhandari D, Taylor CB, Webb SA, CHEST Investigators; Australian and New Zealand Intensive Care Society Clinical Trials Group Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367(20):1901–1911. doi: 10.1056/NEJMoa1209759. [DOI] [PubMed] [Google Scholar]
- 86.Yunos NM, Bellomo R, Hegarty C, Story D, Ho L, Bailey M. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. JAMA. 2012;308(15):1566–1572. doi: 10.1001/jama.2012.13356. [DOI] [PubMed] [Google Scholar]
- 87.Rochwerg B, Alhazzani W, Sindi A, Heels-Ansdell D, Thabane L, Fox-Robichaud A, Mbuagbaw L, Szczeklik W, Alshamsi F, Altayyar S, Ip WC, Li G, Wang M, Wludarczyk A, Zhou Q, Guyatt GH, Cook DJ, Jaeschke R, Annane D, Fluids in Sepsis and Septic Shock Group Fluid resuscitation in sepsis: a systematic review and network meta-analysis. Ann Intern Med. 2014;161(5):347–355. doi: 10.7326/M14-0178. [DOI] [PubMed] [Google Scholar]
- 88.Acheampong A, Vincent JL. A positive fluid balance is an independent prognostic factor in patients with sepsis. Crit Care. 2015;19:251. doi: 10.1186/s13054-015-0970-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Mitchell KH, Carlbom D, Caldwell E, Leary PJ, Himmelfarb J, Hough CL. Volume overload: prevalence, risk factors, and functional outcome in survivors of septic shock. Ann Am Thorac Soc. 2015;12(12):1837–1844. doi: 10.1513/AnnalsATS.201504-187OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Delaney AP, Dan A, McCaffrey J, Finfer S. The role of albumin as a resuscitation fluid for patients with sepsis: a systematic review and meta-analysis. Crit Care Med. 2011;39(2):386–391. doi: 10.1097/CCM.0b013e3181ffe217. [DOI] [PubMed] [Google Scholar]
- 91.Zhang Z, Chen K. Vasoactive agents for the treatment of sepsis. Ann Transl Med. 2016;4(17):333. doi: 10.21037/atm.2016.08.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.De Backer D, Biston P, Devriendt J, Madl C, Chochrad D, Aldecoa C, Brasseur A, Defrance P, Gottignies P, Vincent JL. SOAP II Investigators. Comparison of dopamine and norepinephrine in the treatment of shock. N Engl J Med. 2010;362(9):779–789. doi: 10.1056/NEJMoa0907118. [DOI] [PubMed] [Google Scholar]
- 93.Marik PE, Mohedin M. The contrasting effects of dopamine and norepinephrine on systemic and splanchnic oxygen utilization in hyperdynamic sepsis. JAMA. 1994;272(17):1354–1357. doi: 10.1001/jama.1994.03520170064037. [DOI] [PubMed] [Google Scholar]
- 94.Patel GP, Grahe JS, Sperry M, Singla S, Elpern E, Lateef O, Balk RA. Efficacy and safety of dopamine versus norepinephrine in the management of septic shock. Shock. 2010;33(4):375–380. doi: 10.1097/SHK.0b013e3181c6ba6f. [DOI] [PubMed] [Google Scholar]
- 95.Russell JA, Walley KR, Singer J, Gordon AC, Hébert PC, Cooper DJ, Holmes CL, Mehta S, Granton JT, Storms MM, Cook DJ, Presneill JJ, Ayers D, VASST Investigators Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med. 2008;358(9):877–887. doi: 10.1056/NEJMoa067373. [DOI] [PubMed] [Google Scholar]
- 96.Gordon AC, Mason AJ, Thirunavukkarasu N, Perkins GD, Cecconi M, Cepkova M, Pogson DG, Aya HD, Anjum A, Frazier GJ, Santhakumaran S, Ashby D, Brett SJ, VANISH Investigators Effect of early vasopressin vs norepinephrine on kidney failure in patients with septic shock: the vanish randomized clinical trial. JAMA. 2016;316(5):509–518. doi: 10.1001/jama.2016.10485. [DOI] [PubMed] [Google Scholar]
- 97.Landry DW, Levin HR, Gallant EM, Ashton RC, Jr, Seo S, D'Alessandro D, Oz MC, Oliver JA. Vasopressin deficiency contributes to the vasodilation of septic shock. Circulation. 1997;95(5):1122–1125. doi: 10.1161/01.CIR.95.5.1122. [DOI] [PubMed] [Google Scholar]
- 98.Ferrer R, Martin-Loeches I, Phillips G, Osborn TM, Townsend S, Dellinger RP, Artigas A, Schorr C, Levy MM. Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: results from a guideline-based performance improvement program. Crit Care Med. 2014;42(8):1749–1755. doi: 10.1097/CCM.0000000000000330. [DOI] [PubMed] [Google Scholar]
- 99.Gaieski DF, Mikkelsen ME, Band RA, Pines JM, Massone R, Furia FF, Shofer FS, Goyal M. Impact of time to antibiotics on survival in patients with severe sepsis or septic shock in whom early goal-directed therapy was initiated in the emergency department. Crit Care Med. 2010;38(4):1045–1053. doi: 10.1097/CCM.0b013e3181cc4824. [DOI] [PubMed] [Google Scholar]
- 100.Funk DJ, Kumar A. Antimicrobial therapy for life-threatening infections: speed is life. Crit Care Clin. 2011;27(1):53–76. doi: 10.1016/j.ccc.2010.09.008. [DOI] [PubMed] [Google Scholar]
- 101.NICE-SUGAR Study Investigators. Finfer S, Chittock DR, Su SY, Blair D, Foster D, Dhingra V, Bellomo R, Cook D, Dodek P, Henderson WR, Hébert PC, Heritier S, Heyland DK, McArthur C, McDonald E, Mitchell I, Myburgh JA, Norton R, Potter J, Robinson BG, Ronco JJ. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283–1297. doi: 10.1056/NEJMoa0810625. [DOI] [PubMed] [Google Scholar]
- 102.Keh D, Trips E, Marx G, Wirtz SP, Abduljawwad E, Bercker S, Bogatsch H, Briegel J, Engel C, Gerlach H, Goldmann A, Kuhn SO, Hüter L, Meier-Hellmann A, Nierhaus A, Kluge S, Lehmke J, Loeffler M, Oppert M, Resener K, Schädler D, Schuerholz T, Simon P, Weiler N, Weyland A, Reinhart K, Brunkhorst FM, SepNet–Critical Care Trials Group Effect of hydrocortisone on development of shock among patients with severe sepsis: the HYPRESS randomized clinical trial. JAMA. 2016;316(17):1775–1785. doi: 10.1001/jama.2016.14799. [DOI] [PubMed] [Google Scholar]
- 103.Sprung CL, Annane D, Keh D, Moreno R, Singer M, Freivogel K, Weiss YG, Benbenishty J, Kalenka A, Forst H, Laterre PF, Reinhart K, Cuthbertson BH, Payen D, Briegel J, CORTICUS Study Group Hydrocortisone therapy for patients with septic shock. N Engl J Med. 2008;358(2):111–124. doi: 10.1056/NEJMoa071366. [DOI] [PubMed] [Google Scholar]
- 104.Annane D, Sébille V, Charpentier C, Bollaert PE, François B, Korach JM, Capellier G, Cohen Y, Azoulay E, Troché G, Chaumet-Riffaud P, Bellissant E. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA. 2002;288(7):862–871. doi: 10.1001/jama.288.7.862. [DOI] [PubMed] [Google Scholar]
- 105.Weber-Carstens S, Deja M, Bercker S, Dimroth A, Ahlers O, Kaisers U, Keh D. Impact of bolus application of low-dose hydrocortisone on glycemic control in septic shock patients. Intensive Care Med. 2007;33(4):730–733. doi: 10.1007/s00134-007-0540-3. [DOI] [PubMed] [Google Scholar]
- 106.Mestas J, Hughes CC. Of mice and not men: differences between mouse and human immunology. J Immunol. 2004;172(5):2731–2738. doi: 10.4049/jimmunol.172.5.2731. [DOI] [PubMed] [Google Scholar]
- 107.Dyson A, Singer M. Animal models of sepsis: why does preclinical efficacy fail to translate to the clinical setting? Crit Care Med. 2009;37(1 Suppl):S30–S37. doi: 10.1097/CCM.0b013e3181922bd3. [DOI] [PubMed] [Google Scholar]
- 108.Fink MP, Warren HS. Strategies to improve drug development for sepsis. Nat Rev Drug Discov. 2014;13(10):741–758. doi: 10.1038/nrd4368. [DOI] [PubMed] [Google Scholar]
- 109.Minneci PC, Deans KJ, Eichacker PQ, Natanson C. The effects of steroids during sepsis depend on dose and severity of illness: an updated meta-analysis. Clin Microbiol Infect. 2009;15(4):308–318. doi: 10.1111/j.1469-0691.2009.02752.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Berry SM, Connor JT, Lewis RJ. The platform trial: an efficient strategy for evaluating multiple treatments. JAMA. 2015;313(16):1619–1620. doi: 10.1001/jama.2015.2316. [DOI] [PubMed] [Google Scholar]
- 111.Gattinoni L, Brazzi L, Pelosi P, Latini R, Tognoni G, Pesenti A, et al. A trial of goal-oriented hemodynamic therapy in critically ill patients. SvO2 Collaborative Group. N Engl J Med. 1995;333(16):1025–1032. doi: 10.1056/NEJM199510193331601. [DOI] [PubMed] [Google Scholar]
- 112.Raghunathan K, Shaw A, Nathanson B, Sturmer T, Brookhart A, Stefan MS, et al. Association between the choice of IV crystalloid and in-hospital mortality among critically ill adults with sepsis*. Crit Care Med. 2014;42(7):1585–1591. doi: 10.1097/CCM.0000000000000305. [DOI] [PubMed] [Google Scholar]
- 113.Zampieri FG, Ranzani OT, Azevedo LC, Martins ID, Kellum JA, Liborio AB. Lactated ringer is associated with reduced mortality and less acute kidney injury in critically ill patients: a retrospective cohort analysis. Crit Care Med. 2016;44(12):2163–2170. doi: 10.1097/CCM.0000000000001948. [DOI] [PubMed] [Google Scholar]
- 114.Patel A, Laffan MA, Waheed U, Brett SJ. Randomised trials of human albumin for adults with sepsis: systematic review and meta-analysis with trial sequential analysis of all-cause mortality. BMJ. 2014;349:g4561. doi: 10.1136/bmj.g4561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Monnet X, Teboul JL. Assessment of volume responsiveness during mechanical ventilation: recent advances. Crit Care. 2013;17(2):217. doi: 10.1186/cc12526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Asfar P, Meziani F, Hamel JF, Grelon F, Megarbane B, Anguel N, et al. High versus low blood-pressure target in patients with septic shock. N Engl J Med. 2014;370(17):1583–1593. doi: 10.1056/NEJMoa1312173. [DOI] [PubMed] [Google Scholar]
- 117.Seymour CW, Gesten F, Prescott HC, Friedrich ME, Iwashyna TJ, Phillips GS, et al. Time to treatment and mortality during mandated emergency care for sepsis. N Engl J Med. 2017;376(23):2235–2244. doi: 10.1056/NEJMoa1703058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ziegler R, Johnscher I, Martus P, Lenhardt D, Just HM. Controlled clinical laboratory comparison of two supplemented aerobic and anaerobic media used in automated blood culture systems to detect bloodstream infections. J Clin Microbiol. 1998;36(3):657–661. doi: 10.1128/jcm.36.3.657-661.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bochud PY, Bonten M, Marchetti O, Calandra T. Antimicrobial therapy for patients with severe sepsis and septic shock: an evidence-based review. Crit Care Med. 2004;32(11 Suppl):S495–S512. doi: 10.1097/01.CCM.0000143118.41100.14. [DOI] [PubMed] [Google Scholar]
- 120.Bouadma L, Luyt CE, Tubach F, Cracco C, Alvarez A, Schwebel C, et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375(9713):463–474. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 121.Jensen JU, Hein L, Lundgren B, Bestle MH, Mohr TT, Andersen MH, et al. Procalcitonin-guided interventions against infections to increase early appropriate antibiotics and improve survival in the intensive care unit: a randomized trial. Crit Care Med. 2011;39(9):2048–2058. doi: 10.1097/CCM.0b013e31821e8791. [DOI] [PubMed] [Google Scholar]
- 122.Layios N, Lambermont B, Canivet JL, Morimont P, Preiser JC, Garweg C, et al. Procalcitonin usefulness for the initiation of antibiotic treatment in intensive care unit patients. Crit Care Med. 2012;40(8):2304–2309. doi: 10.1097/CCM.0b013e318251517a. [DOI] [PubMed] [Google Scholar]
- 123.Calfee CS, Delucchi K, Parsons PE, Thompson BT, Ware LB, Matthay MA, et al. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med. 2014;2(8):611–620. doi: 10.1016/S2213-2600(14)70097-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Famous KR, Delucchi K, Ware LB, Kangelaris KN, Liu KD, Thompson BT, et al. Acute respiratory distress syndrome subphenotypes respond differently to randomized fluid management strategy. Am J Respir Crit Care Med. 2017;195(3):331–338. doi: 10.1164/rccm.201603-0645OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Delmas A, Leone M, Rousseau S, Albanese J, Martin C. Clinical review: Vasopressin and terlipressin in septic shock patients. Crit Care. 2005;9(2):212–222. doi: 10.1186/cc2945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Asfar P, Russell JA, Tuckermann J, Radermacher P. Selepressin in septic shock: a step toward decatecholaminization? Crit Care Med. 2016;44(1):234–236. doi: 10.1097/CCM.0000000000001441. [DOI] [PubMed] [Google Scholar]
- 127.Khanna A, English SW, Wang XS, Ham K, Tumlin J, Szerlip H, et al. Angiotensin II for the treatment of vasodilatory shock. N Engl J Med. 2017;377(5):419–430. doi: 10.1056/NEJMoa1704154. [DOI] [PubMed] [Google Scholar]
- 128.Cordemans C, De Laet I, Van Regenmortel N, Schoonheydt K, Dits H, Huber W, et al. Fluid management in critically ill patients: the role of extravascular lung water, abdominal hypertension, capillary leak, and fluid balance. Ann Intensive Care. 2012;2(Suppl 1 Diagnosis and management of intra-abdominal hyperten):S1. doi: 10.1186/2110-5820-2-S1-S1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Han S, Lee SJ, Kim KE, Lee HS, Oh N, Park I, et al. Amelioration of sepsis by TIE2 activation-induced vascular protection. Sci Transl Med. 2016;8(335):335ra55. doi: 10.1126/scitranslmed.aad9260. [DOI] [PubMed] [Google Scholar]
- 130.Opal SM, Laterre PF, Francois B, LaRosa SP, Angus DC, Mira JP, et al. Effect of eritoran, an antagonist of MD2-TLR4, on mortality in patients with severe sepsis: the ACCESS randomized trial. JAMA. 2013;309(11):1154–1162. doi: 10.1001/jama.2013.2194. [DOI] [PubMed] [Google Scholar]
- 131.Ranieri VM, Thompson BT, Barie PS, Dhainaut JF, Douglas IS, Finfer S, et al. Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med. 2012;366(22):2055–2064. doi: 10.1056/NEJMoa1202290. [DOI] [PubMed] [Google Scholar]
- 132.Meisel C, Schefold JC, Pschowski R, Baumann T, Hetzger K, Gregor J, et al. Granulocyte-macrophage colony-stimulating factor to reverse sepsis-associated immunosuppression: a double-blind, randomized, placebo-controlled multicenter trial. Am J Respir Crit Care Med. 2009;180(7):640–648. doi: 10.1164/rccm.200903-0363OC. [DOI] [PubMed] [Google Scholar]
- 133.Fink MP. Cytopathic hypoxia. Mitochondrial dysfunction as mechanism contributing to organ dysfunction in sepsis. Crit Care Clin. 2001;17(1):219–237. doi: 10.1016/S0749-0704(05)70161-5. [DOI] [PubMed] [Google Scholar]
- 134.Schmidt H, Muller-Werdan U, Hoffmann T, Francis DP, Piepoli MF, Rauchhaus M, et al. Autonomic dysfunction predicts mortality in patients with multiple organ dysfunction syndrome of different age groups. Crit Care Med. 2005;33(9):1994–2002. doi: 10.1097/01.CCM.0000178181.91250.99. [DOI] [PubMed] [Google Scholar]
- 135.Morelli A, Ertmer C, Westphal M, Rehberg S, Kampmeier T, Ligges S, et al. Effect of heart rate control with esmolol on hemodynamic and clinical outcomes in patients with septic shock: a randomized clinical trial. JAMA. 2013;310(16):1683–1691. doi: 10.1001/jama.2013.278477. [DOI] [PubMed] [Google Scholar]
- 136.Tyndall A, Pistoia V. Mesenchymal stem cells combat sepsis. Nat Med. 2009;15(1):18–20. doi: 10.1038/nm0109-18. [DOI] [PubMed] [Google Scholar]
- 137.Pandharipande PP, Girard TD, Jackson JC, Morandi A, Thompson JL, Pun BT, et al. Long-term cognitive impairment after critical illness. N Engl J Med. 2013;369(14):1306–1316. doi: 10.1056/NEJMoa1301372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Heming N, Mazeraud A, Verdonk F, Bozza FA, Chretien F, Sharshar T. Neuroanatomy of sepsis-associated encephalopathy. Crit Care. 2017;21(1):65. doi: 10.1186/s13054-017-1643-z. [DOI] [PMC free article] [PubMed] [Google Scholar]