Abstract
Influenza viruses are members of the family Orthomyxoviridae and include influenza virus types A, B, and C. This introduction provides an overview of influenza virus classification, structure, and life cycle. We also include a brief review of the clinical manifestations of influenza and the molecular determinants for virulence. The genetic diversity of influenza A viruses and their capability to successfully infect an array of hosts, including avian and mammalian species, are highlighted in a discussion about host range and evolution. The importance of viral receptor-binding hemagglutinins and host sialic acid distribution in species-restricted binding of viruses is underscored. Finally, recent advances in our understanding of the seasonality and transmission of influenza viruses are described, and their importance for the control of the spread of these viruses is discussed.
Keywords: Influenza Virus, Sialic Acid, H1N1 Virus, Avian Influenza Virus, Pandemic Influenza
Introduction
Influenza has had significant historical impact and continues to pose a considerable threat to public health. Since the transmission of H5N1 avian influenza from birds to humans in 1997, virologists and public health officials alike anticipated global human spread of this virus. More recently, however, pandemic spread of a novel H1N1 influenza virus arose from an unpredicted source; precursors of the pandemic influenza A (H1N1) 2009 virus have been circulating among pigs for over a decade [1, 2]. Additional reassortment events have led to the current pandemic influenza A (H1N1) 2009 virus. Features observed in past pandemics, including atypical seasonality and shifting of the burden of disease to younger populations, are evident during the influenza pandemic of 2009.
Our understanding of the biology of influenza virus and its effect on the host has advanced considerably in recent decades. Recent events in influenza virus research have contributed to this progress [3]. These include the development of plasmid-based reverse genetics systems [4, 5], the generation of the 1918 pandemic H1N1 influenza virus [6], improved access to biosafety level 3 facilities, the establishment of international influenza virus sequence databases, and bioinformatics [7, 8]. Advances have also led to the production of FDA-approved antivirals for influenza, and a heightened understanding of host–virus interactions resulted in the exploration of novel therapies including immunodulatory approaches [9]. New vaccine technologies such as the use of live-attenuated vaccines [10–13] and the development of novel vaccine production methods, including cell culture-based approaches, are the benefits of scientific progress. Continued acceleration of influenza virus research has direct implications for the development of improved vaccines, infection control, and clinical management during pandemic and interpandemic periods.
Overview and Classification
Influenza viruses are members of the family Orthomyxoviridae and include influenza virus types A, B, and C. Influenza viruses possess seven (influenza C) or eight (influenza A and B) genome segments composed of negative sense single-stranded RNA. These types differ in various aspects, the most important of which include antigenicity, host range, pathogenicity, transmission, and seasonality. Standard nomenclature for human influenza viruses includes type, geographic location of isolation, isolate number, and year of isolation. For example, an influenza A virus isolated in Panama in 1999 would be referred to as A/Panama/2002/1999. Subtypes of influenza A viruses are described by hemagglutinin (HA) and neuraminidase (NA) designations. To date, 16 HA and 9 NA subtypes have been described.
Influenza A viruses are mostly responsible for seasonal epidemics, global pandemics, and the burden of disease attributable to influenza. Clinical disease includes systemic and respiratory manifestations, and rarely may be complicated by central nervous system involvement, toxic shock, or multiorgan system failure [14, 15]. Circulating strains of influenza A viruses are targets for annual vaccination to mitigate morbidity and mortality imparted by these viruses. In addition to infecting humans, influenza A viruses circulate in other mammals, including swine and horses. Waterfowl harbor several lineages of influenza A viruses and serve as a reservoir. Transmission among wild and domestic fowl and mammalian species is an important characteristic of influenza A, enabling viral reassortment and emergence of novel subtypes in susceptible human populations.
In contrast, influenza B virus has a restricted host range, circulating only in humans, although the virus has been isolated in seals [16]. Influenza B virus demonstrates seasonality and is responsible for human disease, although the clinical manifestations are generally less severe compared with influenza A virus-associated illness. Nonetheless, rare cases of encephalitis and septic shock have been described in children [17, 18]. At present, the two major lineages are represented by influenza B/Victoria/2/1987 and B/Yamagata/16/1988 viruses [19]. Re-emergence of the Victoria lineage after a decade of absence was associated with an outbreak during the 2001–2002 influenza season, affecting healthy but immunologically naive children [20]. Influenza B virus is included in inactivated and live-attenuated annual influenza vaccines.
Unlike influenza A and B, influenza C virus lacks neuraminidase and codes for a single-surface hemagglutinin–esterase–fusion (HEF) glycoprotein. This virus does not demonstrate marked seasonality and is not included in the annual influenza vaccine, although it has been responsible for occasional outbreaks, predominantly in children [21]. Illness in humans is generally mild and consists of an upper respiratory tract infection. Influenza C has also been isolated in swine, raising the possibility that this species may serve as a reservoir [22].
Structure and Genomic Organization
Influenza viruses are enveloped, deriving the lipid bilayer from the host cell membrane during the process of budding. Viral particles are pleomorphic in nature and may be spherical or filamentous, ranging in size from 100 to over 300nm [3]. Spikes consisting of HA and NA project from the surface of virions at a ratio of roughly 4:1 in influenza A viruses (Fig.1) [3]. The viral envelope is also associated with the matrix (M2) protein which forms a tetrameric ion channel.
Fig.1.
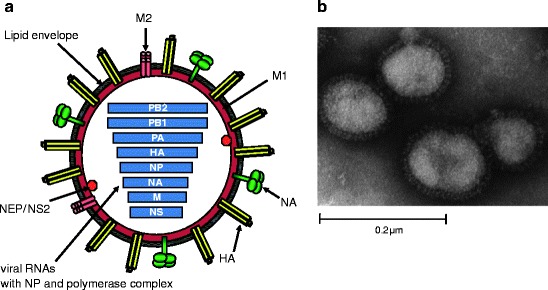
Schematic structure and electron micrograph of influenza virus A. (a) The viral envelop anchors the HA and NA glycoproteins and M2 protein and is derived from the host cell during the process of budding. M1 lies beneath the viral envelope. NEP/NS1 and the core of the virion are contained within. The core consists of eight segments of viral RNA associated with the polymerase complex (PB2, PB1, and PA) and NP. Adapted from [1] and kindly provided by M.L. Shaw. (b) Negatively stained electron micrograph of mouse-adapted influenza A WSN/33. Glycoprotein spikes are visible on the surface of the virion. Kindly provided by M.L. Shaw
The polymerase proteins PB1, PB2, and PA, the nucleoprotein (NP), and the virion RNA comprise the ribonucleoprotein (RNP) complex. This complex is present in the core of virions, which also includes the nuclear export and nonstructural protein (NEP/NS1). Influenza virus genes, gene products, and primary functions are summarized in Table1.
Table1.
Influenza A genes and primary functions of their encoded proteins
| Genome segmenta | Length in nucleotides | Encoded proteins | Protein size in amino acids | Function |
|---|---|---|---|---|
| 1 | 2341 | PB2 | 759 | Polymerase subunit, mRNA cap recognition |
| 2 | 2341 | PB1 | 757 | Polymerase subunit, endonuclease activity, RNA elongation |
| PB1-F2b | 87 | Proapoptotic activity | ||
| 3 | 2233 | PA | 716 | Polymerase subunit, protease activity, assembly of polymerase complex |
| 4 | 1778 | HA | 550 | Surface glycoprotein, receptor binding, fusion activity, major viral antigen |
| 5 | 1565 | NP | 498 | RNA binding activity, required for replication, regulates RNA nuclear import |
| 6 | 1413 | NA | 454 | Surface glycoprotein with neuraminidase activity, virus release |
| 7 | 1027 | M1 | 252 | Matrix protein, interacts with vRNPs and glycoproteins, regulates RNA nuclear export, viral budding |
| M2c | 97 | Integral membrane protein, ion channel activity, uncoating, virus assembly | ||
| 8 | 890 | NS1 | 230 | Interferon antagonist activity, regulates host gene expression |
| NEP/NS2c | 121 | Nuclear export of RNA |
aInfluenza A/Puerto Rico/8/1934
bEncoded by an alternate open reading frame
cTranslated from an alternatively spliced transcript
Influenza Virus Life Cycle
Attachment, Entry, and Nuclear Import
In humans, influenza viruses are transmitted by the respiratory route. Host cellular receptors consist of oligosaccharides residing on the surface of respiratory epithelial cells. Specificity of binding is imparted by the linkage of the penultimate galactose (Gal) to N-acetylsialic acid (SA). α2,6 linkage (SAα2,6Gal) is distributed in the human respiratory tract and is associated with binding to human influenza virus HA. In contrast, avian hosts including waterfowl and domestic poultry harbor sialic acid with α2,3 linkage (SAα2,3Gal) which is distributed in the gastrointestinal tract, reflecting the fecal-oral mode of transmission of avian influenza strains in these species [23]. Specificity of viral HA binding is imparted by the receptor-binding pocket on the surface of the HA molecule (Fig.2). The HA is a rod-shaped trimer anchored in the virion’s envelope and contains three primary ligand-binding sites on a globular head [24, 25]. Specificity of binding has been linked to certain amino acid residues in the HA receptor-binding domain. In H3 subtypes, amino acid 226 is one such residue, where the presence of leucine allows binding of SAα2,6Gal, whereas the presence of glutamine at this position permits binding of SAα2,3Gal. Amino acid changes in the HA of other subtypes, such as H1 viruses (including the H1N1 virus responsible for the 1918 pandemic), have been associated with adaptations in receptor-binding specificity, translating into a switch in host specificity with disastrous consequences [26, 27]. Specifically, changes at amino acid position 225 impart the ability of A/New York/1/18 to bind both avian and human host influenza virus receptors [26]. Strains of the 2009 pandemic H1N1 influenza viruses retain amino acids (aspartic acids) at positions 190 and 225 of the HA consistent with human sialic acid receptor-binding specificity, although conflicting data exist regarding binding specificity for these viruses. One approach utilizing carbohydrate microarrays suggests that dual (human and avian) sialic acid receptor binding occurs [28]; data obtained using a different approach, namely biotinylated α2,3- and α2,6-sialylated glycans, suggest currently circulating pandemic viruses preferentially bind human sialic acid receptors with α2-6 linkage [29]. The importance of these amino acid residues to respiratory droplet transmission has recently been described using the ferret transmission model. H1N1 viruses containing aspartic acids at residues 190 and 225 were capable of aerosol transmission. This contrasted with H1N1 viruses with glutamic acid and glycine at residues 190 and 225, respectively (consistent with avian sialic acid receptor-binding specificity), which did not transmit through the air [30]. Furthermore, other changes in the HA (and NA) of an avian H9N2 after adaptation in the ferret conferred a more efficient respiratory transmission phenotype [31].
Fig.2.
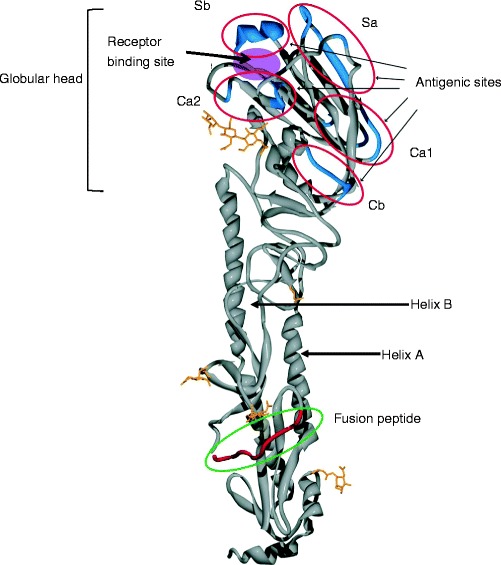
Ribbon structure of the 1918 influenza virus hemagglutinin. The sialic acid receptor-binding site and the five antigenic sites are located on the globular head. This structure also possesses a cleavage site where HA is cleaved into HA1 and HA2 for fusion of viral and endosomal membranes and subsequent uncoating. Adapted from [1] and kindly provided by J. Stevens and I. Wilson
Several possible pathways for the entry of influenza viruses into host cells have been postulated and recently reviewed [32]. Endocytosis is a multistep process consisting of surface receptor-mediated binding, internalization, and intracellular trafficking. Clathrin-mediated and clathrin-independent internalization via caveolae and caveolae-independent endocytosis have been demonstrated [33, 34]. An initial acidification step in early endosomes is followed by trafficking to low-pH late endosomes, a process mediated by members of the Rab host protein family. Fusion of influenza virus to the endosome is triggered by low pH conditions and mediated by the fusion peptide of HA2 after cleavage of HA, creating a pore in the endosome through fusion of viral and endosomal membranes (Fig.3) [3].
Fig.3.
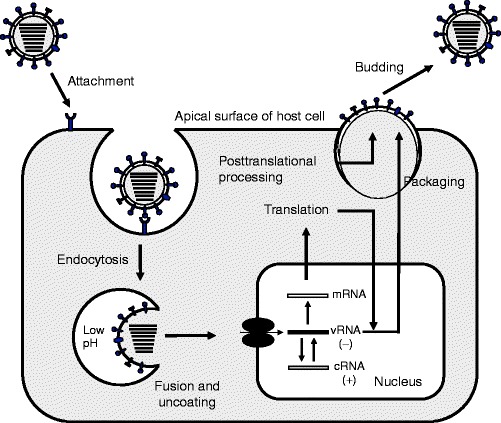
Influenza virus replication cycle. The virus is endocytosed after initial binding of the HA to host cell sialic acid receptors. Acidification of the cleaved HA facilitates approximation of viral and endosomal membranes and release of RNP. Transcription follows importation of RNPs into the nucleus. Assembly occurs at the apical surface of the host cell where budding and release occur. Adapted from [1] and kindly provided by M.L. Shaw. See text for detail
Subsequent steps in the uncoating process involve the influenza virus tetrameric M2 protein, which is involved in the release of RNP into the host cell cytoplasm through ion channel activity [35, 36]. Viral RNA (vRNA) synthesis occurs in the nucleus, and viral RNPs must therefore be imported. This process is primarily mediated by viral NP, which coats viral RNA and possesses nuclear localization signals (NLSs), including an unconventional NLS which binds host karyopherin-α and is essential for energy-dependent RNP nuclear import [37, 38].
Transcription, Replication, and Nuclear Export
Viral RNA serves as a template for the production of messenger RNA (mRNA) and subsequent transcription, as well as for the generation of complementary RNA (cRNA), which is positive sense and functions as a template for the generation of more vRNA (viral replication). RNA segments are coated by NP through nonspecific interactions between the arginine-rich positively charged NP and the negatively charged RNA phosphate backbone [3]. The viral polymerase complex consists of tightly associated PB1, PB2, and PA and associates with NP-coated RNA without disrupting this interaction [39]. PB1 is an endonuclease involved in both replication and transcription and binds the promoter region of RNA segments [40]. It functions as an RNA-dependent RNA polymerase and catalyzes RNA chain elongation. Interaction with PA is required for this function and viral replication [41]. PB2 binds both NP and PB1 via separate binding sites [42]. Initiation of transcription is reliant on PB2, which binds the cap on host pre-mRNA, and this cap serves as a primer for transcription [43, 44]. In addition, interactions between PB2 and host proteins may be species specific and potentially plays a role in restricting host range [45]. PA is a component of the polymerase heterotrimer, is cotransported into the nucleus with PB1, and is thus important in the formation of this complex [46, 47].
Synthesis of mRNA begins with a host cell 5′-capped primer, generated by host cell RNA polymerase II and obtained from host pre-mRNA [44]. Transcription is thus initiated and synthesis on the template occurs in a 3′ to 5′ direction. A polyadenylation signal consisting of 5–7 uridines at the 5′ end of vRNA prematurely terminates transcription after inducing stuttering of the viral polymerase [48–50]. The generation of NP and NS1 tends to occur earlier after infection compared with the generation of surface glycoprotein and M1 mRNAs [3]. Mechanisms for the regulation of gene expression remain evasive, although NP has been implicated in the control of gene expression [51].
Viral replication requires the synthesis of vRNA, which is primer independent and occurs through a cRNA intermediate. Nascent cRNA is therefore not capped or polyadenylated upon termination. The notion that cRNA synthesis is initiated after a switch from mRNA synthesis has been challenged [52].
RNP complexes subsequently associate with M1 at its C-terminal domain, and aggregation of this complex leads to inhibition of transcription [53]. M1 also interacts with NEP at its C-terminal domain [38, 54]. NEP, in turn, associates with host nuclear export receptor Crm1 via the NEP N-terminal domain [54], thus orchestrating the export of viral RNP from the nucleus.
Viral Assembly, Budding, and Release
Posttranslational modification of the HA consists of glycosylation in the Golgi apparatus [55]. Along with viral RNP, protein components of the virion are coordinately trafficked to the apical surface of the host cell for assembly into progeny virus.
Two models for the packaging of viral RNA segments exist and include the random incorporation [56, 57] and the selective incorporation models [58, 59]. The latter implies that each RNA segment possesses a packaging signal, resulting in virions with exactly eight segments. Putative packaging signals in coding regions of polymerase genes, spike glycoprotein genes, and the NS gene have been identified [58, 60–63].
Viral assembly is coordinated by the M1 protein, which associates with the cytoplasmic tails of the viral glycoproteins [19, 64, 65], as well as RNP and NEP, as described above. Lipid rafts navigate viral membrane glycoproteins to the apical surface of the host cell [66, 67]. In addition, there is evidence that targeting of NP and polymerase proteins to the apical surface also involves lipid rafts [68].
Genomic packaging and viral assembly occurs at the apical membrane and is associated with accumulation of M1 and the formation of lipid rafts. The M1 protein has also been implicated in viral morphology [69, 70]. Because the HA binds cell surface sialic acid receptors, virions must be released. The NA functions as a sialidase and cleaves sialic acids from the host cell and viral glycoproteins to minimize viral aggregation at the cell surface [71]. Balance between the HA and NA is thus required for optimal receptor binding and destruction [64, 72]. In addition to its receptor-destroying activity, NA is a viral spike glycoprotein and important surface antigen [73].
Evolution
Among the influenza virus types, influenza A demonstrates the most genetic diversity and is capable of successfully infecting an array of hosts, including avian and mammalian species. Influenza A viruses exhibit an evolutionary pattern, which is complex and consists of antigenic drift and shift. Drift occurs on an annual basis and has been attributed to low fidelity of the RNA polymerase and subsequent selection from immune pressure exerted by the host [74]. This results in antigenic diversity of the hemagglutinin and neuraminidase glycoproteins and is one of the major challenges to vaccine production, requiring annual changes to vaccine components. The HA1 domain contains several epitopes and is the most dynamic as a consequence, demonstrating clusters of antigenic variance over time [75]. Antigenic shift results after a viral reassortment event where exchange of one or more of the viral segments with that of another strain may result in a novel serotype, potentially diversifying the host range of the virus. It is in this setting that pandemic strains have emerged in immunologically naïve populations in the past, including the H2N2 (with new HA, NA, and PB1 segment) subtype in 1957 and the H3N2 influenza virus (with new HA and PB1 segments) which caused a pandemic in 1968 (Fig.4).
Fig.4.
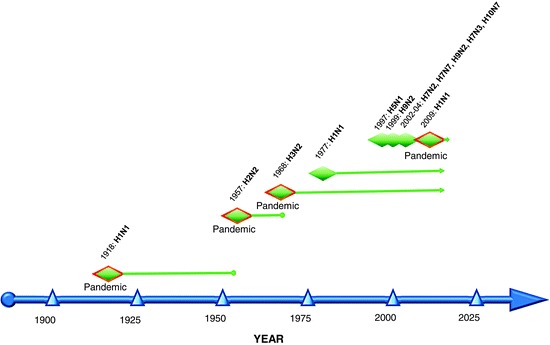
Influenza A virus subtypes in humans. Three pandemics occurred during the twentieth century, including the “Spanish” influenza pandemic of 1918, the “Asian” pandemic in 1958, and the “Hong Kong” pandemic in 1968. H1N1 viruses re-emerged in 1977 and continue to circulate in the human population, along with the H3N2 subtype. In addition, H5N1 viruses have been reported to infect humans throughout Asia and Africa. Several other avian viruses have also recently caused sporadic infection in humans. A swine-origin influenza virus (pandemic influenza A H1N1 2009 virus) emerged during the spring of 2009 and spread globally, inciting the World Health Organization to declare a pandemic in June of 2009. Adapted from [68]
Since 1997, several avian influenza viruses, including H5N1, H7N2, H7N3, H7N7, H9N2, and H10N7 subtypes, have infected humans [76], though limited evidence for person to person spread exists [77, 78]. Lack of transmission among humans remains a barrier to pandemic spread of these viruses. The H5N1 subtype isolated from avian species has undergone genetic reassortment, and several genotypes exist. Genotypes Z and V are largely responsible for outbreaks of highly pathogenic influenza viruses (HPAI) in domestic birds in Southeast Asia beginning in 2003 [77]. H5N1 viruses may also be divided into clades based on the genomic analysis of the HA genes, and clade 2 is further divided into subclades; up to ten clades have been identified in avian species, four of which have infected humans [79, 80]. Less than 1% divergence from avian isolates has been reported in viruses isolated from humans in Asia [7].
The pandemic influenza A (H1N1) 2009 virus has been described as a “triple reassortant” of swine, human, and avian influenza viruses; the H1 gene from this virus has been circulating among swine for decades, with limited drift compared with genes of H1 viruses that have been circulating in humans, and is thus antigenically different from seasonal human H1N1 viruses. The pandemic influenza A (H1N1) 2009 virus is composed of six segments from the triple reassortant, including a human PB1 segment, classical swine-origin HA, NP, and NS, and avian-origin PB2 and PA segments that have been circulating in swine since approximately 1998. The NA and M segments originate from a Eurasian lineage of swine influenza viruses [1, 2, 81] (Fig.5).
Fig.5.
Origins of pandemic influenza A H1N1 2009 virus. Swine (classical), human, and avian influenza viruses reassorted in North America in 1998 to produce an H3N2 virus which circulated in swine. Further reassortment with a Eurasian lineage of swine influenza virus resulted in the current pandemic influenza virus which has spread globally in humans
In order to tackle the challenge of understanding the evolution of influenza virus, large-scale collaborative efforts such as the Influenza Genome Sequencing Project have been undertaken. The presence of several cocirculating clades in the human population has been described, accounting for reassortment. This can result in limited vaccine effectiveness, as seen with A/Fujian/411/2002-like virus during the 2003–2004 season [8]. Genetic evolution appears to be a relatively gradual process; however, antigenic changes in the HA1 domain tend to cluster [75]. Ongoing changes of the H3 hemagglutinin in the human population result from selective pressure exerted by the host immune system. In contrast, the H3 lineage in birds has remained relatively stable [82]. The rate of change of the H3 subtype is greater when compared with H1 viruses and influenza B, with estimated nucleotide changes per site per year of 0.0037 for H3, 0.0018 for H1, and 0.0013 for influenza B [83]. As greater numbers of influenza virus genome sequences become available and we gain insight into antigenic patterns of change, this knowledge may be applied to annual vaccine development. Prediction of future influenza sequences could lead to more timely development of effective vaccines [84] though modeling methods have yet to be validated.
Host Range
Influenza A virus is a zoonotic pathogen capable of infecting birds (waterfowl and chickens), swine, horses, felines, and other species. Host range restriction of different types of influenza viruses is observed. Species-restricted binding of viruses is mediated by different types of receptor-binding hemaglutinins [85–89]. The distribution of different types of SA linkages has recently been elucidated in humans though the type of cell infected (ciliated vs. nonciliated) is under debate [90, 91]. SA with α2,6Gal linkage predominates on epithelial cells of the upper airway, including nasal mucosa, sinuses, bronchi, and bronchioles [92]. In human tracheobronchial epithelial (HTBE) cells, oligosaccharides with SA with α2,6Gal linkage predominate on nonciliated epithelial cells [91] although these oligosaccharides have been described on ciliated and goblet cells in the human airway [93]. Lower airways contain SA with mostly α2,3Gal linkage, in addition to SA with α2,6Gal linkage [92, 94].
Host restriction is not absolute, and human infections with avian influenza viruses (including H5N1, H9N2, and H7N7 viruses) have been extensively described [95–100]. H5N1 binds type II pneumocytes and macrophages of the lower respiratory tract in humans [92, 94, 101]. H5N1 infection of ciliated cells in HTBE cell culture with limited cell-to-cell spread [90] and of human nasopharyngeal, adenoid, and tonsillar ex vivo cell cultures has been shown [102]. Binding of H5N1 viruses to saccharides terminating in α2,6Gal SA linkage has been achieved by mutating HA amino acid residues at positions 182 and 192, suggesting potential for adaptation to the human host [103].
Differences in influenza virus receptors among avian species have been described and are reflected in differential binding of different types of avian influenza viruses. Although chicken and duck influenza viruses preferentially bind α2,3Gal-linked SA, viruses from chickens had greater affinity for SA where the third sugar moiety was a β(1-4)GlcNAc-containing synthetic sialylglycopolymer. Duck viruses preferred β(1-3)GalNAc sugar moieties in the third position [104]. Distribution of influenza virus receptors reflects the sites of replication. In chickens and waterfowl, SA with α2,3Gal linkage is found in the upper respiratory tract and intestines. Some species demonstrate the ability to support replication of both human and avian influenza viruses. The respiratory tract and intestines of quail contain both α2,3Gal- and α2,6Gal-linked terminal sialic acids [105]. In swine, oligosaccharides with both types of linkages may be found and suggest this species serves as a mixing vessel where human, avian, and swine influenza viruses can reassort [106, 107].
Clinical Manifestations, Pathogenesis, and Virulence
Clinical Manifestations
Uncomplicated influenza in humans is an upper respiratory tract infection characterized by cough, headache, malaise, and fever (influenza-like illness). These symptoms are nonspecific and are not predictive of influenza virus infection, particularly in individuals <60years old [108]. Pulmonary and extrapulmonary complications may arise. The latter consist of central nervous system involvement (encephalitis, acute necrotizing encephalopathy, Reye’s syndrome, and myelitis) [14], myositis/rhabdomyositis [109], myocarditis [109, 110], increased cardiovascular events [111], disseminated intravascular coagulation [109], and toxic and septic shock (bacterial and nonbacterial) [15, 18, 109]. Pulmonary complications include primary viral pneumonia, secondary bacterial pneumonia (see below), and exacerbation of chronic lung disease [109, 112]. Acute lung injury (ALI)/acute respiratory distress syndrome (ARDS), multiorgan failure, profound lymphopenia, and hemophagocytosis have been associated with H5N1 infection and carry high mortality rates [15, 95, 113–115].
Bacterial pneumonia following influenza virus infection is a well-recognized complication of influenza since the pandemic of 1918 [116]. More recently, pediatric deaths have been attributed to copathogenesis between influenza virus and Staphylococcus aureus, accounting for 34% of pediatric deaths reported to the CDC during the 2006–2007 influenza season [117]. In one case series, 43% of coinfected cases involved methicillin-resistant S. aureus, thus contributing to management challenges for these patients. Coinfection was also associated with a worse prognosis compared with influenza virus or S. aureus infection alone [118].
To date, secondary bacterial lower respiratory tract infection has not been a dominant feature in adults during the current 2009 pandemic but has been described in children [119]. Severe pandemic 2009 influenza has been predominantly associated with viral pneumonitis and subsequent ALI, particularly in pregnant women in their third trimester [120] and indigeous people including Aborigines in Australia [121], Maoris and Pacific Islanders in New Zealand [122], and First Nations People in Canada [123].
Pathogenesis
Few human histopathological studies of uncomplicated influenza exist. Pathological findings from postmortem examination of 47 fatal pediatric influenza A cases included major airway congestion (90%), inflammation (73%), and necrosis (50%) [112]. Lower airway pathology included hyaline membranes (67%), interstitial cellular infiltrates (67%), and diffuse alveolar damage (DAD). Secondary pneumonia, intraalveolar hemorrhage, and viral pneumonitis were noted in a quarter of cases [112]. Fulminant DAD with acute alveolar hemorrhage and necrosis followed by paucicellular fibrosis and hyaline membrane formation is observed in H5N1-infected human lungs [124]. Extrapulmonary pathology includes reactive hemophagocytosis in the hilar lymph nodes, bone marrow, liver, and spleen [125]; white matter demyelination [124] and cerebral necrosis [101]; and acute tubular necrosis of the kidneys [113]. Despite the presence of diarrhea and H5N1 virus replication in the gastrointestinal tract of humans, no pathological lesions have been described in the bowel [101, 114]. Immune dysregulation has been implicated in the pathogenesis of ARDS and reactive hemophagocytosis. Elevated levels of neutrophil, monocyte, and macrophage chemoattractants (IL-8, IP-10, MIG, and MCP-1) and proinflammatory cytokines (IL-10, IL-6, and IFN-γ) are observed in H5N1-infected humans [95]. In addition, increased levels of IL-2 (in a human case) [113] and RANTES (in primary human alveolar and bronchial epithelial cells) [126] have also been reported. Contribution of proinflammatory mediators to lung pathology has also been demonstrated using Toll-like receptor 3 knockout mice infected with mouse-adapted WSN influenza A virus. These mice demonstrated enhanced survival despite higher virus replication and lower levels of RANTES, IL-6, and IL-12p40/p70 compared with wild-type mice [127].
Likewise, host response has been implicated in the copathogenesis of bacterial pneumonia post-influenza virus infection. Specifically, sensitization by type I interferons [128], induction of IL-10 [129], and upregulation of interferon-α [130] have been linked to secondary bacterial pneumonia after influenza virus infection. Viral determinants for copathogenesis have also been elucidated and include PB1-F2 and viral neuraminidase [131, 132].
Virological Determinants of Virulence
The HA, PA, PB1, PB2, PB1-F2, NA, and NS1 gene products have been implicated in virulence. Virulence determinants have been explored using the reverse genetic system for influenza viruses and mammalian (ferret and mouse) models for influenza virus pathogenicity.
The polymerase gene complex, consisting of PA, PB1, and PB2 genes, is involved in replication and transcriptional activity. A single-gene reassortant containing the PB2 from A/Hong Kong/483/97 (H5N1, which is fatal in mice) in the background of A/Hong Kong/486/97 (H5N1, causing mild respiratory infection in mice) demonstrated a lethal phenotype in this animal model [133]. In addition, reassortants containing polymerase complex genes from A/chicken/Vietnam/C58/04 (H5N1), a nonlethal virus, in the background of A/Vietnam/1203/04 (H5N1) influenza virus isolated from a fatal human case were attenuated in an animal model [134]. When a single point mutation K627E in the PB2 gene was generated in A/Vietnam/1203/04 [134] and in A/Hong Kong/483/97 [133], virulence was reduced in mice, although in other studies this substitution did not reduce virulence substantially [135]. The molecular mechanism(s) responsible for virulence have yet to be completely elucidated. Enhanced replication of viruses retaining a lysine at position 627 in PB2 at the lower temperatures of the upper respiratory tract (33°C) [136] may be responsible for robust transmission in mammals [137]. This theory is supported by recent work demonstrating that replacement of the lysine at position 627 with glutamic acid (avian consensus sequence) abrogates aerosol transmission of a 1918 influenza A virus [30]. Currently circulating strains of pandemic H1N1 influenza virus have a glutamine in PB2 at position 627. This may account for reduced efficiency of aerosol transmission of this virus in ferrets, compared with a seasonal H1N1 virus [29].
PB1-F2 is the gene product arising from a second reading frame of the PB1 gene and has been implicated in immune cell apoptosis through the VDAC1 and ANT3 mitochondrial pathways [138]. Knockout of PB1-F2 did not alter viral replication, but enhanced clearance of the virus and reduced lethality in mice was demonstrated, suggesting that PB1-F2 may play a role in viral pathogenesis [139]. Enhanced pathogenicity was observed in mice infected with recombinant influenza virus containing the PB1-F2 gene from a highly pathogenic H5N1 virus isolated from a fatal human case in Hong Kong in 1997 [139]. Currently circulating strains of the pandemic influenza A H1N1 2009 virus do not express PB1-F2.
Evasion of the host immune response is a key virulence determinant, permitting viruses to establish sustainable infection. The innate immune system is the first line of host defense, and the influenza virus possesses the ability to interfere with this response. Type I interferons (IFN-α/β) are central to establishing an antiviral state in host cells. Interferon antagonism has been primarily attributed to the NS1 protein of influenza virus, which plays a multifunctional role in preventing the activation of IFN transcription factors (for review, see [140, 141]).
The effect of avian influenza virus NS1 on IFN production has also been explored. A/goose/Guangdong/1/96 virus with an NS1 that differs by one amino acid from A/goose/Guangdong/2/96 at position 149 is lethal in chickens and antagonizes IFNα/β [142]. In addition, the C-terminus of the NS1 protein contains a PDZ ligand domain, capable of binding PDZ protein interaction domains of host proteins, thus potentially disrupting host cellular pathways. Viruses causing pathogenic infection in humans between 1997 and 2003 contained avian motifs at the NS1 PDZ ligand-binding site. These and the motif found in the 1918 influenza virus NS1 had stronger binding affinities to PDZ domains of human cellular proteins compared with low pathogenicity influenza viruses [143].
Neurovirulence has been associated with glycosylation of the NA glycoprotein [144]. The HA glycoprotein has also been associated with virulence. Although cleavability of the HA gene has been primarily implicated in pathogenicity in chickens, lethality has also been demonstrated in mice. Basic amino acids at the HA cleavage site are determinants for HA cleavage and HA2 fusion activity [145]. Enhanced cleavage of the HA by ubiquitous host proteases is made possible by the presence of a polybasic cleavage site, contributing to the virulence of highly pathogenic avian influenza viruses [146, 147]. Replacement of the polybasic cleavage site in a high pathogenicity H5N1 virus from Hong Kong (HK483) with an amino acid sequence typical of low pathogenicity viruses resulted in attenuation [133]. Pandemic influenza A H1N1 2009 virus strains do not appear to have the polybasic cleavage site.
Virulence determinants for the pandemic 2009 H1N1 virus are currently investigation. Data obtained from mammalian models early in the course of the spread of this virus indicate that compared with a seasonal H1N1 influenza virus, strains of the pandemic virus replicate more efficiently in the lower respiratory tract, and are stronger inducers of proinflammatory mediators, and induce bronchopneumonia [148].
Seasonality and Transmission
Influenza A and B viruses exhibit marked seasonality, and this pattern dictates the annual vaccination schedule. Several theories with respect to the mechanism(s) responsible for this seasonal pattern have been proposed (for review, see [149]). Year-round human influenza virus activity in equatorial regions may be a reservoir for annual outbreaks in the northern and southern hemispheres. As research progresses in this area, factors determining seasonality may be exploited for the control of the spread of influenza virus [150].
Transmission of influenza virus among humans is poorly understood and the mode(s) of spread are currently under debate [151, 152]. It is widely accepted that influenza virus is transmitted by the respiratory route in humans, though the contribution of small particle aerosols relative to large respiratory droplets is unknown. In addition, the role of fomites is questionable. Until recently, ferrets have served as the only animal model for the study of influenza virus transmission. A novel mammalian model using the guinea pig has recently been developed to overcome the limitations of the ferret model. Guinea pigs are highly susceptible to infection with an unadapted human H3N3 (A/Panama/2002/1999, or Pan99) influenza virus, with a 50% infectious dose of 5 PFU, and this virus grows to high titers in the upper respiratory tract and to moderate titers in the lungs. Transmission of Pan99 by direct contact and aerosol in this system is 100% (Fig.6) [153]; however, transmission efficiency may vary among influenza virus subtypes [154]. Environmental factors such as temperature and relative humidity also appear to play a substantial role [155, 156]. Control of influenza virus spread during interpandemic and pandemic periods through vaccination [157] and physical means will be paramount to abrogating person-to-person transmission and is crucial where viruses are resistant to currently available antivirals.
Fig.6.
Close range transmission of human influenza A among guinea pigs. Inoculated animals placed in proximity to uninoculated animals (without direct contact) spread Pan99 to all exposed animals. Adapted from [109]
Perspectives
Effective and timely vaccine development depends on in-depth understanding of influenza virus biology. Although recent advances have been made, ongoing research will be required to fulfill this goal. Identification and characterization of the molecular signatures required for transmission will be of utmost importance to preventing further influenza virus pandemics. Globalization of H1N1 infection in humans requires parallel efforts on behalf of virologists in conjunction with epidemiologists and other members of the public health community to translate the growing body of knowledge into means by which influenza spread can be controlled.
Acknowledgments
The work completed in this laboratory was partially supported by the W.M. Keck Foundation, National Institutes of Health grants P01 AI158113, the Northeast Biodefense Center U54 AI057158, the Center for Investigating Viral Immunity and Antagonism (CIVIA) U19 AI62623. S.M. is grateful for the Ruth L. Kirschstein Physician Scientist Research Training in Pathogenesis of Viral Diseases Award (5T32A1007623-07) and support from Sunnybrook Health Sciences Center, Toronto, ON, Canada.
Contributor Information
Rino Rappuoli, Email: rino.rappuoli@novartis.com.
Giuseppe Del Giudice, Email: giuseppe.del_giudice@novartis.com.
Samira Mubareka, Email: samira.mubareka@sunnybrook.ca.
Peter Palese, Email: peter.palese@mssm.edu.
References
- 1.Garten RJ, Davis CT, Russell CA, Shu B, Lindstrom S, Balish A, Sessions WM, Xu X, Skepner E, Deyde V, et al. Antigenic and genetic characteristics of swine-origin 2009 A(H1N1) influenza viruses circulating in humans. Science. 2009;325:197–201. doi: 10.1126/science.1176225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shinde V, Bridges CB, Uyeki TM, Shu B, Balish A, Xu X, Lindstrom S, Gubareva LV, Deyde V, Garten RJ, et al. Triple-reassortant swine influenza A (H1) in humans in the United States, 2005–2009. N Engl J Med. 2009;360:2616–2625. doi: 10.1056/NEJMoa0903812. [DOI] [PubMed] [Google Scholar]
- 3.Shaw ML, Palese P. Orthomyxoviridae: the viruses and their replication. In: Knipe DM, Howley PM, editors. Fields virology. 5. Philadelphia: Lippincott Williams & Wilkins; 2007. pp. 1647–1689. [Google Scholar]
- 4.Fodor E, Devenish L, Engelhardt OG, Palese P, Brownlee GG, Garcia-Sastre A. Rescue of influenza A virus from recombinant DNA. J Virol. 1999;73:9679–9682. doi: 10.1128/jvi.73.11.9679-9682.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Neumann G, Watanabe T, Ito H, Watanabe S, Goto H, Gao P, Hughes M, Perez DR, Donis R, Hoffmann E, et al. Generation of influenza A viruses entirely from cloned cDNAs. Proc Natl Acad Sci USA. 1999;96:9345–9350. doi: 10.1073/pnas.96.16.9345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tumpey TM, Basler CF, Aguilar PV, Zeng H, Solorzano A, Swayne DE, Cox NJ, Katz JM, Taubenberger JK, Palese P, et al. Characterization of the reconstructed 1918 Spanish influenza pandemic virus. Science. 2005;310:77–80. doi: 10.1126/science.1119392. [DOI] [PubMed] [Google Scholar]
- 7.World Health Organization Global Influenza Program Surveillance Network Evolution of H5N1 avian influenza viruses in Asia. Emerg Infect Dis. 2005;11:1515–1521. doi: 10.3201/eid1110.050644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ghedin E, Sengamalay NA, Shumway M, Zaborsky J, Feldblyum T, Subbu V, Spiro DJ, Sitz J, Koo H, Bolotov P, et al. Large-scale sequencing of human influenza reveals the dynamic nature of viral genome evolution. Nature. 2005;437:1162–1166. doi: 10.1038/nature04239. [DOI] [PubMed] [Google Scholar]
- 9.Kugel D, Kochs G, Obojes K, Roth J, Kobinger GP, Kobasa D, Haller O, Staeheli P, von Messling V. Intranasal administration of alpha interferon reduces seasonal influenza A virus morbidity in ferrets. J Virol. 2009;83:3843–3851. doi: 10.1128/JVI.02453-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Steel J, Lowen AC, Pena L, Angel M, Solorzano A, Albrecht R, Perez DR, Garcia-Sastre A, Palese P. Live attenuated influenza viruses containing NS1 truncations as vaccine candidates against H5N1 highly pathogenic avian influenza. J Virol. 2009;83:1742–1753. doi: 10.1128/JVI.01920-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hai R, Martinez-Sobrido L, Fraser KA, Ayllon J, Garcia-Sastre A, Palese P. Influenza B virus NS1-truncated mutants: live-attenuated vaccine approach. J Virol. 2008;82:10580–10590. doi: 10.1128/JVI.01213-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Suguitan AL, Jr, McAuliffe J, Mills KL, Jin H, Duke G, Lu B, Luke CJ, Murphy B, Swayne DE, Kemble G, et al. Live, attenuated influenza A H5N1 candidate vaccines provide broad cross-protection in mice and ferrets. PLoS Med. 2006;3:e360. doi: 10.1371/journal.pmed.0030360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Murphy BR, Coelingh K. Principles underlying the development and use of live attenuated cold-adapted influenza A and B virus vaccines. Viral Immunol. 2002;15:295–323. doi: 10.1089/08828240260066242. [DOI] [PubMed] [Google Scholar]
- 14.Studahl M. Influenza virus and CNS manifestations. J Clin Virol. 2003;28:225–232. doi: 10.1016/S1386-6532(03)00119-7. [DOI] [PubMed] [Google Scholar]
- 15.Sion ML, Hatzitolios AI, Toulis EN, Mikoudi KD, Ziakas GN. Toxic shock syndrome complicating influenza A infection: a two-case report with one case of bacteremia and endocarditis. Intensive Care Med. 2001;27:443. doi: 10.1007/s001340000803. [DOI] [PubMed] [Google Scholar]
- 16.Osterhaus AD, Rimmelzwaan GF, Martina BE, Bestebroer TM, Fouchier RA. Influenza B virus in seals. Science. 2000;288:1051–1053. doi: 10.1126/science.288.5468.1051. [DOI] [PubMed] [Google Scholar]
- 17.Newland JG, Romero JR, Varman M, Drake C, Holst A, Safranek T, Subbarao K. Encephalitis associated with influenza B virus infection in 2 children and a review of the literature. Clin Infect Dis. 2003;36:e87–e95. doi: 10.1086/368184. [DOI] [PubMed] [Google Scholar]
- 18.Jaimovich DG, Kumar A, Shabino CL, Formoli R. Influenza B virus infection associated with non-bacterial septic shock-like illness. J Infect. 1992;25:311–315. doi: 10.1016/0163-4453(92)91659-Y. [DOI] [PubMed] [Google Scholar]
- 19.Chen JM, Guo YJ, Wu KY, Guo JF, Wang M, Dong J, Zhang Y, Li Z, Shu YL. Exploration of the emergence of the Victoria lineage of influenza B virus. Arch Virol. 2007;152:415–422. doi: 10.1007/s00705-006-0852-6. [DOI] [PubMed] [Google Scholar]
- 20.Hite LK, Glezen WP, Demmler GJ, Munoz FM. Medically attended pediatric influenza during the resurgence of the Victoria lineage of influenza B virus. Int J Infect Dis. 2007;11:40–47. doi: 10.1016/j.ijid.2005.10.008. [DOI] [PubMed] [Google Scholar]
- 21.Matsuzaki Y, Abiko C, Mizuta K, Sugawara K, Takashita E, Muraki Y, Suzuki H, Mikawa M, Shimada S, Sato K, et al. A nationwide epidemic of influenza C virus infection in Japan in 2004. J Clin Microbiol. 2007;45:783–788. doi: 10.1128/JCM.01555-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yuanji G, Desselberger U. Genome analysis of influenza C viruses isolated in 1981/82 from pigs in China. J Gen Virol. 1984;65(Pt 11):1857–1872. doi: 10.1099/0022-1317-65-11-1857. [DOI] [PubMed] [Google Scholar]
- 23.Wright PF, Neumann G, Kawaoka Y. Orthomyxoviruses. In: Knipe DM, Howley PM, editors. Fields virology. 5. Philadephia: Lippincott Williams & Wilkins; 2007. pp. 1714–1715. [Google Scholar]
- 24.Eisen MB, Sabesan S, Skehel JJ, Wiley DC. Binding of the influenza A virus to cell-surface receptors: structures of five hemagglutinin-sialyloligosaccharide complexes determined by X-ray crystallography. Virology. 1997;232:19–31. doi: 10.1006/viro.1997.8526. [DOI] [PubMed] [Google Scholar]
- 25.Skehel JJ, Wiley DC. Receptor binding and membrane fusion in virus entry: the influenza hemagglutinin. Annu Rev Biochem. 2000;69:531–569. doi: 10.1146/annurev.biochem.69.1.531. [DOI] [PubMed] [Google Scholar]
- 26.Glaser L, Stevens J, Zamarin D, Wilson IA, Garcia-Sastre A, Tumpey TM, Basler CF, Taubenberger JK, Palese P. A single amino acid substitution in 1918 influenza virus hemagglutinin changes receptor binding specificity. J Virol. 2005;79:11533–11536. doi: 10.1128/JVI.79.17.11533-11536.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stevens J, Blixt O, Tumpey TM, Taubenberger JK, Paulson JC, Wilson IA. Structure and receptor specificity of the hemagglutinin from an H5N1 influenza virus. Science. 2006;312:404–410. doi: 10.1126/science.1124513. [DOI] [PubMed] [Google Scholar]
- 28.Childs RA, Palma AS, Wharton S, Matrosovich T, Liu Y, Chai W, Campanero-Rhodes MA, Zhang Y, Eickmann M, Kiso M, et al. Receptor-binding specificity of pandemic influenza A (H1N1) 2009 virus determined by carbohydrate microarray. Nat Biotechnol. 2009;27:797–799. doi: 10.1038/nbt0909-797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Maines TR, Jayaraman A, Belser JA, Wadford DA, Pappas C, Zeng H, Gustin KM, Pearce MB, Viswanathan K, Shriver ZH, et al. Transmission and pathogenesis of swine-origin 2009 A(H1N1) influenza viruses in ferrets and mice. Science. 2009;325:484–487. doi: 10.1126/science.1177238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Van Hoeven N, Pappas C, Belser JA, Maines TR, Zeng H, Garcia-Sastre A, Sasisekharan R, Katz JM, Tumpey TM. Human HA and polymerase subunit PB2 proteins confer transmission of an avian influenza virus through the air. Proc Natl Acad Sci USA. 2009;106:3366–3371. doi: 10.1073/pnas.0813172106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sorrell EM, Wan H, Araya Y, Song H, Perez DR. Minimal molecular constraints for respiratory droplet transmission of an avian-human H9N2 influenza A virus. Proc Natl Acad Sci USA. 2009;106:7565–7570. doi: 10.1073/pnas.0900877106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sieczkarski SB, Whittaker GR. Viral entry. Curr Top Microbiol Immunol. 2005;285:1–23. doi: 10.1007/3-540-26764-6_1. [DOI] [PubMed] [Google Scholar]
- 33.Matlin KS, Reggio H, Helenius A, Simons K. Infectious entry pathway of influenza virus in a canine kidney cell line. J Cell Biol. 1981;91:601–613. doi: 10.1083/jcb.91.3.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nunes-Correia I, Eulalio A, Nir S, Pedroso de Lima MC. Caveolae as an additional route for influenza virus endocytosis in MDCK cells. Cell Mol Biol Lett. 2004;9:47–60. [PubMed] [Google Scholar]
- 35.Takeda M, Pekosz A, Shuck K, Pinto LH, Lamb RA. Influenza a virus M2 ion channel activity is essential for efficient replication in tissue culture. J Virol. 2002;76:1391–1399. doi: 10.1128/JVI.76.3.1391-1399.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pinto LH, Holsinger LJ, Lamb RA. Influenza virus M2 protein has ion channel activity. Cell. 1992;69:517–528. doi: 10.1016/0092-8674(92)90452-I. [DOI] [PubMed] [Google Scholar]
- 37.Wang P, Palese P, O’Neill RE. The NPI-1/NPI-3 (karyopherin alpha) binding site on the influenza a virus nucleoprotein NP is a nonconventional nuclear localization signal. J Virol. 1997;71:1850–1856. doi: 10.1128/jvi.71.3.1850-1856.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cros JF, Garcia-Sastre A, Palese P. An unconventional NLS is critical for the nuclear import of the influenza A virus nucleoprotein and ribonucleoprotein. Traffic. 2005;6:205–213. doi: 10.1111/j.1600-0854.2005.00263.x. [DOI] [PubMed] [Google Scholar]
- 39.Area E, Martin-Benito J, Gastaminza P, Torreira E, Valpuesta JM, Carrascosa JL, Ortin J. 3D structure of the influenza virus polymerase complex: localization of subunit domains. Proc Natl Acad Sci USA. 2004;101:308–313. doi: 10.1073/pnas.0307127101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jung TE, Brownlee GG. A new promoter-binding site in the PB1 subunit of the influenza A virus polymerase. J Gen Virol. 2006;87:679–688. doi: 10.1099/vir.0.81453-0. [DOI] [PubMed] [Google Scholar]
- 41.Perez DR, Donis RO. Functional analysis of PA binding by influenza a virus PB1: effects on polymerase activity and viral infectivity. J Virol. 2001;75:8127–8136. doi: 10.1128/JVI.75.17.8127-8136.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Poole E, Elton D, Medcalf L, Digard P. Functional domains of the influenza A virus PB2 protein: identification of NP- and PB1-binding sites. Virology. 2004;321:120–133. doi: 10.1016/j.virol.2003.12.022. [DOI] [PubMed] [Google Scholar]
- 43.Fechter P, Mingay L, Sharps J, Chambers A, Fodor E, Brownlee GG. Two aromatic residues in the PB2 subunit of influenza A RNA polymerase are crucial for cap binding. J Biol Chem. 2003;278:20381–20388. doi: 10.1074/jbc.M300130200. [DOI] [PubMed] [Google Scholar]
- 44.Krug RM, Bouloy M, Plotch SJ. RNA primers and the role of host nuclear RNA polymerase II in influenza viral RNA transcription. Philos Trans R Soc Lond B Biol Sci. 1980;288:359–370. doi: 10.1098/rstb.1980.0012. [DOI] [PubMed] [Google Scholar]
- 45.Labadie K, Dos Santos AE, Rameix-Welti MA, van der Werf S, Naffakh N. Host-range determinants on the PB2 protein of influenza A viruses control the interaction between the viral polymerase and nucleoprotein in human cells. Virology. 2007;362:271–282. doi: 10.1016/j.virol.2006.12.027. [DOI] [PubMed] [Google Scholar]
- 46.Kawaguchi A, Naito T, Nagata K. Involvement of influenza virus PA subunit in assembly of functional RNA polymerase complexes. J Virol. 2005;79:732–744. doi: 10.1128/JVI.79.2.732-744.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fodor E, Smith M. The PA subunit is required for efficient nuclear accumulation of the PB1 subunit of the influenza A virus RNA polymerase complex. J Virol. 2004;78:9144–9153. doi: 10.1128/JVI.78.17.9144-9153.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li X, Palese P. Characterization of the polyadenylation signal of influenza virus RNA. J Virol. 1994;68:1245–1249. doi: 10.1128/jvi.68.2.1245-1249.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Luo GX, Luytjes W, Enami M, Palese P. The polyadenylation signal of influenza virus RNA involves a stretch of uridines followed by the RNA duplex of the panhandle structure. J Virol. 1991;65:2861–2867. doi: 10.1128/jvi.65.6.2861-2867.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zheng H, Lee HA, Palese P, Garcia-Sastre A. Influenza A virus RNA polymerase has the ability to stutter at the polyadenylation site of a viral RNA template during RNA replication. J Virol. 1999;73:5240–5243. doi: 10.1128/jvi.73.6.5240-5243.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ye Q, Krug RM, Tao YJ. The mechanism by which influenza A virus nucleoprotein forms oligomers and binds RNA. Nature. 2006;444:1078–1082. doi: 10.1038/nature05379. [DOI] [PubMed] [Google Scholar]
- 52.Vreede FT, Brownlee GG. Influenza virion-derived viral ribonucleoproteins synthesize both mRNA and cRNA in vitro. J Virol. 2007;81:2196–2204. doi: 10.1128/JVI.02187-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Baudin F, Petit I, Weissenhorn W, Ruigrok RW. In vitro dissection of the membrane and RNP binding activities of influenza virus M1 protein. Virology. 2001;281:102–108. doi: 10.1006/viro.2000.0804. [DOI] [PubMed] [Google Scholar]
- 54.Akarsu H, Burmeister WP, Petosa C, Petit I, Muller CW, Ruigrok RW, Baudin F. Crystal structure of the M1 protein-binding domain of the influenza A virus nuclear export protein (NEP/NS2) EMBO J. 2003;22:4646–4655. doi: 10.1093/emboj/cdg449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gallagher PJ, Henneberry JM, Sambrook JF, Gething MJ. Glycosylation requirements for intracellular transport and function of the hemagglutinin of influenza virus. J Virol. 1992;66:7136–7145. doi: 10.1128/jvi.66.12.7136-7145.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Enami M, Sharma G, Benham C, Palese P. An influenza virus containing nine different RNA segments. Virology. 1991;185:291–298. doi: 10.1016/0042-6822(91)90776-8. [DOI] [PubMed] [Google Scholar]
- 57.Bancroft CT, Parslow TG. Evidence for segment-nonspecific packaging of the influenza a virus genome. J Virol. 2002;76:7133–7139. doi: 10.1128/JVI.76.14.7133-7139.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Watanabe T, Watanabe S, Noda T, Fujii Y, Kawaoka Y. Exploitation of nucleic acid packaging signals to generate a novel influenza virus-based vector stably expressing two foreign genes. J Virol. 2003;77:10575–10583. doi: 10.1128/JVI.77.19.10575-10583.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.de Wit E, Spronken MI, Rimmelzwaan GF, Osterhaus AD, Fouchier RA. Evidence for specific packaging of the influenza A virus genome from conditionally defective virus particles lacking a polymerase gene. Vaccine. 2006;24:6647–6650. doi: 10.1016/j.vaccine.2006.06.001. [DOI] [PubMed] [Google Scholar]
- 60.Fujii K, Fujii Y, Noda T, Muramoto Y, Watanabe T, Takada A, Goto H, Horimoto T, Kawaoka Y. Importance of both the coding and the segment-specific noncoding regions of the influenza A virus NS segment for its efficient incorporation into virions. J Virol. 2005;79:3766–3774. doi: 10.1128/JVI.79.6.3766-3774.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liang Y, Hong Y, Parslow TG. Cis-acting packaging signals in the influenza virus PB1, PB2, and PA genomic RNA segments. J Virol. 2005;79:10348–10355. doi: 10.1128/JVI.79.16.10348-10355.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Fujii Y, Goto H, Watanabe T, Yoshida T, Kawaoka Y. Selective incorporation of influenza virus RNA segments into virions. Proc Natl Acad Sci USA. 2003;100:2002–2007. doi: 10.1073/pnas.0437772100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gog JR, Afonso ED, Dalton RM, Leclercq I, Tiley L, Elton D, von Kirchbach JC, Naffakh N, Escriou N, Digard P. Codon conservation in the influenza A virus genome defines RNA packaging signals. Nucleic Acids Res. 2007;35:1897–1907. doi: 10.1093/nar/gkm087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Schmitt AP, Lamb RA. Influenza virus assembly and budding at the viral budozone. Adv Virus Res. 2005;64:383–416. doi: 10.1016/S0065-3527(05)64012-2. [DOI] [PubMed] [Google Scholar]
- 65.Chen BJ, Takeda M, Lamb RA. Influenza virus hemagglutinin (H3 subtype) requires palmitoylation of its cytoplasmic tail for assembly: M1 proteins of two subtypes differ in their ability to support assembly. J Virol. 2005;79:13673–13684. doi: 10.1128/JVI.79.21.13673-13684.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang J, Pekosz A, Lamb RA. Influenza virus assembly and lipid raft microdomains: a role for the cytoplasmic tails of the spike glycoproteins. J Virol. 2000;74:4634–4644. doi: 10.1128/JVI.74.10.4634-4644.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Barman S, Adhikary L, Chakrabarti AK, Bernas C, Kawaoka Y, Nayak DP. Role of transmembrane domain and cytoplasmic tail amino acid sequences of influenza a virus neuraminidase in raft association and virus budding. J Virol. 2004;78:5258–5269. doi: 10.1128/JVI.78.10.5258-5269.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Carrasco M, Amorim MJ, Digard P. Lipid raft-dependent targeting of the influenza A virus nucleoprotein to the apical plasma membrane. Traffic. 2004;5:979–992. doi: 10.1111/j.1600-0854.2004.00237.x. [DOI] [PubMed] [Google Scholar]
- 69.Bourmakina SV, Garcia-Sastre A. Reverse genetics studies on the filamentous morphology of influenza A virus. J Gen Virol. 2003;84:517–527. doi: 10.1099/vir.0.18803-0. [DOI] [PubMed] [Google Scholar]
- 70.Elleman CJ, Barclay WS. The M1 matrix protein controls the filamentous phenotype of influenza A virus. Virology. 2004;321:144–153. doi: 10.1016/j.virol.2003.12.009. [DOI] [PubMed] [Google Scholar]
- 71.Palese P, Tobita K, Ueda M, Compans RW. Characterization of temperature sensitive influenza virus mutants defective in neuraminidase. Virology. 1974;61:397–410. doi: 10.1016/0042-6822(74)90276-1. [DOI] [PubMed] [Google Scholar]
- 72.Mitnaul LJ, Matrosovich MN, Castrucci MR, Tuzikov AB, Bovin NV, Kobasa D, Kawaoka Y. Balanced hemagglutinin and neuraminidase activities are critical for efficient replication of influenza A virus. J Virol. 2000;74:6015–6020. doi: 10.1128/JVI.74.13.6015-6020.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Colman PM. Influenza virus neuraminidase: structure, antibodies, and inhibitors. Protein Sci. 1994;3:1687–1696. doi: 10.1002/pro.5560031007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fitch WM, Leiter JM, Li XQ, Palese P. Positive Darwinian evolution in human influenza A viruses. Proc Natl Acad Sci USA. 1991;88:4270–4274. doi: 10.1073/pnas.88.10.4270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Smith DJ, Lapedes AS, de Jong JC, Bestebroer TM, Rimmelzwaan GF, Osterhaus AD, Fouchier RA. Mapping the antigenic and genetic evolution of influenza virus. Science. 2004;305:371–376. doi: 10.1126/science.1097211. [DOI] [PubMed] [Google Scholar]
- 76.National Institute of Allergy and Infectious Diseases NIH (2007) http://www3.niaid.nih.gov/news/focuson/flu/illustrations/timeline/
- 77.Horimoto T, Kawaoka Y. Influenza: lessons from past pandemics, warnings from current incidents. Nat Rev Microbiol. 2005;3:591–600. doi: 10.1038/nrmicro1208. [DOI] [PubMed] [Google Scholar]
- 78.Wang H, Feng Z, Shu Y, Yu H, Zhou L, Zu R, Huai Y, Dong J, Bao C, Wen L, et al. Probable limited person-to-person transmission of highly pathogenic avian influenza A (H5N1) virus in China. Lancet. 2008;371:1427–1434. doi: 10.1016/S0140-6736(08)60493-6. [DOI] [PubMed] [Google Scholar]
- 79.Webster RG, Govorkova EA. H5N1 influenza-continuing evolution and spread. N Engl J Med. 2006;355:2174–2177. doi: 10.1056/NEJMp068205. [DOI] [PubMed] [Google Scholar]
- 80.Uyeki TM. Global epidemiology of human infections with highly pathogenic avian influenza A (H5N1) viruses. Respirology. 2008;13(Suppl 1):S2–S9. doi: 10.1111/j.1440-1843.2008.01246.x. [DOI] [PubMed] [Google Scholar]
- 81.Dawood FS, Jain S, Finelli L, Shaw MW, Lindstrom S, Garten RJ, Gubareva LV, Xu X, Bridges CB, Uyeki TM. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med. 2009;360:2605–2615. doi: 10.1056/NEJMoa0903810. [DOI] [PubMed] [Google Scholar]
- 82.Bean WJ, Schell M, Katz J, Kawaoka Y, Naeve C, Gorman O, Webster RG. Evolution of the H3 influenza virus hemagglutinin from human and nonhuman hosts. J Virol. 1992;66:1129–1138. doi: 10.1128/jvi.66.2.1129-1138.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ferguson NM, Galvani AP, Bush RM. Ecological and immunological determinants of influenza evolution. Nature. 2003;422:428–433. doi: 10.1038/nature01509. [DOI] [PubMed] [Google Scholar]
- 84.Plotkin JB, Dushoff J, Levin SA. Hemagglutinin sequence clusters and the antigenic evolution of influenza A virus. Proc Natl Acad Sci USA. 2002;99:6263–6268. doi: 10.1073/pnas.082110799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Suzuki Y, Ito T, Suzuki T, Holland RE, Jr, Chambers TM, Kiso M, Ishida H, Kawaoka Y. Sialic acid species as a determinant of the host range of influenza A viruses. J Virol. 2000;74:11825–11831. doi: 10.1128/JVI.74.24.11825-11831.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Vines A, Wells K, Matrosovich M, Castrucci MR, Ito T, Kawaoka Y. The role of influenza A virus hemagglutinin residues 226 and 228 in receptor specificity and host range restriction. J Virol. 1998;72:7626–7631. doi: 10.1128/jvi.72.9.7626-7631.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rogers GN, D’Souza BL. Receptor binding properties of human and animal H1 influenza virus isolates. Virology. 1989;173:317–322. doi: 10.1016/0042-6822(89)90249-3. [DOI] [PubMed] [Google Scholar]
- 88.Rogers GN, Paulson JC. Receptor determinants of human and animal influenza virus isolates: differences in receptor specificity of the H3 hemagglutinin based on species of origin. Virology. 1983;127:361–373. doi: 10.1016/0042-6822(83)90150-2. [DOI] [PubMed] [Google Scholar]
- 89.Rogers GN, Pritchett TJ, Lane JL, Paulson JC. Differential sensitivity of human, avian, and equine influenza A viruses to a glycoprotein inhibitor of infection: selection of receptor specific variants. Virology. 1983;131:394–408. doi: 10.1016/0042-6822(83)90507-X. [DOI] [PubMed] [Google Scholar]
- 90.Thompson CI, Barclay WS, Zambon MC, Pickles RJ. Infection of human airway epithelium by human and avian strains of influenza a virus. J Virol. 2006;80:8060–8068. doi: 10.1128/JVI.00384-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Matrosovich MN, Matrosovich TY, Gray T, Roberts NA, Klenk HD. Human and avian influenza viruses target different cell types in cultures of human airway epithelium. Proc Natl Acad Sci USA. 2004;101:4620–4624. doi: 10.1073/pnas.0308001101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Shinya K, Ebina M, Yamada S, Ono M, Kasai N, Kawaoka Y. Avian flu: influenza virus receptors in the human airway. Nature. 2006;440:435–436. doi: 10.1038/440435a. [DOI] [PubMed] [Google Scholar]
- 93.Ibricevic A, Pekosz A, Walter MJ, Newby C, Battaile JT, Brown EG, Holtzman MJ, Brody SL. Influenza virus receptor specificity and cell tropism in mouse and human airway epithelial cells. J Virol. 2006;80:7469–7480. doi: 10.1128/JVI.02677-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.van Riel D, Munster VJ, de Wit E, Rimmelzwaan GF, Fouchier RA, Osterhaus AD, Kuiken T. H5N1 virus attachment to lower respiratory tract. Science. 2006;312:399. doi: 10.1126/science.1125548. [DOI] [PubMed] [Google Scholar]
- 95.de Jong MD, Simmons CP, Thanh TT, Hien VM, Smith GJ, Chau TN, Hoang DM, Chau NV, Khanh TH, Dong VC, et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med. 2006;12:1203–1207. doi: 10.1038/nm1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kandun IN, Wibisono H, Sedyaningsih ER, Yusharmen HW, Purba W, Santoso H, Septiawati C, Tresnaningsih E, Heriyanto B, et al. Three Indonesian clusters of H5N1 virus infection in 2005. N Engl J Med. 2006;355:2186–2194. doi: 10.1056/NEJMoa060930. [DOI] [PubMed] [Google Scholar]
- 97.Oner AF, Bay A, Arslan S, Akdeniz H, Sahin HA, Cesur Y, Epcacan S, Yilmaz N, Deger I, Kizilyildiz B, et al. Avian influenza A (H5N1) infection in eastern Turkey in 2006. N Engl J Med. 2006;355:2179–2185. doi: 10.1056/NEJMoa060601. [DOI] [PubMed] [Google Scholar]
- 98.Butt KM, Smith GJ, Chen H, Zhang LJ, Leung YH, Xu KM, Lim W, Webster RG, Yuen KY, Peiris JS, et al. Human infection with an avian H9N2 influenza A virus in Hong Kong in 2003. J Clin Microbiol. 2005;43:5760–5767. doi: 10.1128/JCM.43.11.5760-5767.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Koopmans M, Wilbrink B, Conyn M, Natrop G, van der Nat H, Vennema H, Meijer A, van Steenbergen J, Fouchier R, Osterhaus A, et al. Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet. 2004;363:587–593. doi: 10.1016/S0140-6736(04)15589-X. [DOI] [PubMed] [Google Scholar]
- 100.Fouchier RA, Schneeberger PM, Rozendaal FW, Broekman JM, Kemink SA, Munster V, Kuiken T, Rimmelzwaan GF, Schutten M, Van Doornum GJ, et al. Avian influenza A virus (H7N7) associated with human conjunctivitis and a fatal case of acute respiratory distress syndrome. Proc Natl Acad Sci USA. 2004;101:1356–1361. doi: 10.1073/pnas.0308352100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Uiprasertkul M, Puthavathana P, Sangsiriwut K, Pooruk P, Srisook K, Peiris M, Nicholls JM, Chokephaibulkit K, Vanprapar N, Auewarakul P. Influenza A H5N1 replication sites in humans. Emerg Infect Dis. 2005;11:1036–1041. doi: 10.3201/eid1107.041313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Nicholls JM, Chan MC, Chan WY, Wong HK, Cheung CY, Kwong DL, Wong MP, Chui WH, Poon LL, Tsao SW, et al. Tropism of avian influenza A (H5N1) in the upper and lower respiratory tract. Nat Med. 2007;13:147–149. doi: 10.1038/nm1529. [DOI] [PubMed] [Google Scholar]
- 103.Yamada S, Suzuki Y, Suzuki T, Le MQ, Nidom CA, Sakai-Tagawa Y, Muramoto Y, Ito M, Kiso M, Horimoto T, et al. Haemagglutinin mutations responsible for the binding of H5N1 influenza A viruses to human-type receptors. Nature. 2006;444:378–382. doi: 10.1038/nature05264. [DOI] [PubMed] [Google Scholar]
- 104.Gambaryan AS, Tuzikov AB, Bovin NV, Yamnikova SS, Lvov DK, Webster RG, Matrosovich MN. Differences between influenza virus receptors on target cells of duck and chicken and receptor specificity of the 1997 H5N1 chicken and human influenza viruses from Hong Kong. Avian Dis. 2003;47:1154–1160. doi: 10.1637/0005-2086-47.s3.1154. [DOI] [PubMed] [Google Scholar]
- 105.Wan H, Perez DR. Quail carry sialic acid receptors compatible with binding of avian and human influenza viruses. Virology. 2006;346:278–286. doi: 10.1016/j.virol.2005.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shu LL, Lin YP, Wright SM, Shortridge KF, Webster RG. Evidence for interspecies transmission and reassortment of influenza A viruses in pigs in southern China. Virology. 1994;202:825–833. doi: 10.1006/viro.1994.1404. [DOI] [PubMed] [Google Scholar]
- 107.Castrucci MR, Donatelli I, Sidoli L, Barigazzi G, Kawaoka Y, Webster RG. Genetic reassortment between avian and human influenza A viruses in Italian pigs. Virology. 1993;193:503–506. doi: 10.1006/viro.1993.1155. [DOI] [PubMed] [Google Scholar]
- 108.Call SA, Vollenweider MA, Hornung CA, Simel DL, McKinney WP. Does this patient have influenza? JAMA. 2005;293:987–997. doi: 10.1001/jama.293.8.987. [DOI] [PubMed] [Google Scholar]
- 109.Bhat N, Wright JG, Broder KR, Murray EL, Greenberg ME, Glover MJ, Likos AM, Posey DL, Klimov A, Lindstrom SE, et al. Influenza-associated deaths among children in the United States, 2003–2004. N Engl J Med. 2005;353:2559–2567. doi: 10.1056/NEJMoa051721. [DOI] [PubMed] [Google Scholar]
- 110.Nolte KB, Alakija P, Oty G, Shaw MW, Subbarao K, Guarner J, Shieh WJ, Dawson JE, Morken T, Cox NJ, et al. Influenza A virus infection complicated by fatal myocarditis. Am J Forensic Med Pathol. 2000;21:375–379. doi: 10.1097/00000433-200012000-00016. [DOI] [PubMed] [Google Scholar]
- 111.Davis MM, Taubert K, Benin AL, Brown DW, Mensah GA, Baddour LM, Dunbar S, Krumholz HM. Influenza vaccination as secondary prevention for cardiovascular disease: a science advisory from the American Heart Association/American College of Cardiology. J Am Coll Cardiol. 2006;48:1498–1502. doi: 10.1016/j.jacc.2006.09.004. [DOI] [PubMed] [Google Scholar]
- 112.Guarner J, Paddock CD, Shieh WJ, Packard MM, Patel M, Montague JL, Uyeki TM, Bhat N, Balish A, Lindstrom S, et al. Histopathologic and immunohistochemical features of fatal influenza virus infection in children during the 2003–2004 season. Clin Infect Dis. 2006;43:132–140. doi: 10.1086/505122. [DOI] [PubMed] [Google Scholar]
- 113.Chan PK. Outbreak of avian influenza A(H5N1) virus infection in Hong Kong in 1997. Clin Infect Dis. 2002;34:S58–S64. doi: 10.1086/338820. [DOI] [PubMed] [Google Scholar]
- 114.Beigel JH, Farrar J, Han AM, Hayden FG, Hyer R, de Jong MD, Lochindarat S, Nguyen TK, Nguyen TH, Tran TH, et al. Avian influenza A (H5N1) infection in humans. N Engl J Med. 2005;353:1374–1385. doi: 10.1056/NEJMra052211. [DOI] [PubMed] [Google Scholar]
- 115.Chotpitayasunondh T, Ungchusak K, Hanshaoworakul W, Chunsuthiwat S, Sawanpanyalert P, Kijphati R, Lochindarat S, Srisan P, Suwan P, Osotthanakorn Y, et al. Human disease from influenza A (H5N1), Thailand, 2004. Emerg Infect Dis. 2005;11:201–209. doi: 10.3201/eid1102.041061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Morens DM, Taubenberger JK, Fauci AS. Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness. J Infect Dis. 2008;198:962–970. doi: 10.1086/591708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Finelli L, Fiore A, Dhara R, Brammer L, Shay DK, Kamimoto L, Fry A, Hageman J, Gorwitz R, Bresee J, et al. Influenza-associated pediatric mortality in the United States: increase of Staphylococcus aureus coinfection. Pediatrics. 2008;122:805–811. doi: 10.1542/peds.2008-1336. [DOI] [PubMed] [Google Scholar]
- 118.Reed C, Kallen AJ, Patton M, Arnold KE, Farley MM, Hageman J, Finelli L. Infection with community-onset Staphylococcus aureus and influenza virus in hospitalized children. Pediatr Infect Dis J. 2009;28:572–576. doi: 10.1097/INF.0b013e31819d8b71. [DOI] [PubMed] [Google Scholar]
- 119.CDC Surveillance for pediatric deaths associated with 2009 pandemic influenza A (H1N1) virus infection – United States, April–August 2009. MMWR Morb Mortal Wkly Rep. 2009;58:941–947. [PubMed] [Google Scholar]
- 120.Jamieson DJ, Honein MA, Rasmussen SA, Williams JL, Swerdlow DL, Biggerstaff MS, Lindstrom S, Louie JK, Christ CM, Bohm SR, et al. H1N1 2009 influenza virus infection during pregnancy in the USA. Lancet. 2009;374:451–458. doi: 10.1016/S0140-6736(09)61304-0. [DOI] [PubMed] [Google Scholar]
- 121.Massey PD, Pearce G, Taylor KA, Orcher L, Saggers S, Durrheim DN. Reducing the risk of pandemic influenza in Aboriginal communities. Rural Remote Health. 2009;9:1290. [PubMed] [Google Scholar]
- 122.Baker MG, Wilson N, Huang QS, Paine S, Lopez L, Bandaranayake D, Tobias M, Mason K, Mackereth GF, Jacobs M, et al. Pandemic influenza A(H1N1)v in New Zealand: the experience from August 2009. Euro Surveill. 2009;14(34):19319. doi: 10.2807/ese.14.34.19319-en. [DOI] [PubMed] [Google Scholar]
- 123.Public Health Agency of Canada (2009) http://www.phac-aspc.gc.ca/fluwatch/08-09/w33_09/index-eng.php. Accessed 3 Sept 2009
- 124.Ng WF, To KF, Lam WW, Ng TK, Lee KC. The comparative pathology of severe acute respiratory syndrome and avian influenza A subtype H5N1 – a review. Hum Pathol. 2006;37:381–390. doi: 10.1016/j.humpath.2006.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.To KF, Chan PK, Chan KF, Lee WK, Lam WY, Wong KF, Tang NL, Tsang DN, Sung RY, Buckley TA, et al. Pathology of fatal human infection associated with avian influenza A H5N1 virus. J Med Virol. 2001;63:242–246. doi: 10.1002/1096-9071(200103)63:3<242::AID-JMV1007>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- 126.Chan MC, Cheung CY, Chui WH, Tsao SW, Nicholls JM, Chan YO, Chan RW, Long HT, Poon LL, Guan Y, et al. Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells. Respir Res. 2005;6:135. doi: 10.1186/1465-9921-6-135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Le Goffic R, Balloy V, Lagranderie M, Alexopoulou L, Escriou N, Flavell R, Chignard M, Si-Tahar M. Detrimental contribution of the Toll-like receptor (TLR)3 to influenza A virus-induced acute pneumonia. PLoS Pathog. 2006;2:e53. doi: 10.1371/journal.ppat.0020053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Shahangian A, Chow EK, Tian X, Kang JR, Ghaffari A, Liu SY, Belperio JA, Cheng G, Deng JC. Type I IFNs mediate development of postinfluenza bacterial pneumonia in mice. J Clin Invest. 2009;119:1910–1920. doi: 10.1172/JCI35412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.van der Sluijs KF, van Elden LJ, Nijhuis M, Schuurman R, Pater JM, Florquin S, Goldman M, Jansen HM, Lutter R, van der Poll T. IL-10 is an important mediator of the enhanced susceptibility to pneumococcal pneumonia after influenza infection. J Immunol. 2004;172:7603–7609. doi: 10.4049/jimmunol.172.12.7603. [DOI] [PubMed] [Google Scholar]
- 130.Sun K, Metzger DW. Inhibition of pulmonary antibacterial defense by interferon-gamma during recovery from influenza infection. Nat Med. 2008;14:558–564. doi: 10.1038/nm1765. [DOI] [PubMed] [Google Scholar]
- 131.McAuley JL, Hornung F, Boyd KL, Smith AM, McKeon R, Bennink J, Yewdell JW, McCullers JA. Expression of the 1918 influenza A virus PB1-F2 enhances the pathogenesis of viral and secondary bacterial pneumonia. Cell Host Microbe. 2007;2:240–249. doi: 10.1016/j.chom.2007.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Peltola VT, Murti KG, McCullers JA. Influenza virus neuraminidase contributes to secondary bacterial pneumonia. J Infect Dis. 2005;192:249–257. doi: 10.1086/430954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Hatta M, Gao P, Halfmann P, Kawaoka Y. Molecular basis for high virulence of Hong Kong H5N1 influenza A viruses. Science. 2001;293:1840–1842. doi: 10.1126/science.1062882. [DOI] [PubMed] [Google Scholar]
- 134.Salomon R, Franks J, Govorkova EA, Ilyushina NA, Yen HL, Hulse-Post DJ, Humberd J, Trichet M, Rehg JE, Webby RJ, et al. The polymerase complex genes contribute to the high virulence of the human H5N1 influenza virus isolate A/Vietnam/1203/04. J Exp Med. 2006;203:689–697. doi: 10.1084/jem.20051938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Maines TR, Lu XH, Erb SM, Edwards L, Guarner J, Greer PW, Nguyen DC, Szretter KJ, Chen LM, Thawatsupha P, et al. Avian influenza (H5N1) viruses isolated from humans in Asia in 2004 exhibit increased virulence in mammals. J Virol. 2005;79:11788–11800. doi: 10.1128/JVI.79.18.11788-11800.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Massin P, van der Werf S, Naffakh N. Residue 627 of PB2 is a determinant of cold sensitivity in RNA replication of avian influenza viruses. J Virol. 2001;75:5398–5404. doi: 10.1128/JVI.75.11.5398-5404.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Steel J, Lowen AC, Mubareka S, Palese P. Transmission of influenza virus in a mammalian host is increased by PB2 amino acids 627K or 627E/701N. PLoS Pathog. 2009;5:e1000252. doi: 10.1371/journal.ppat.1000252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zamarin D, Garcia-Sastre A, Xiao X, Wang R, Palese P. Influenza virus PB1-F2 protein induces cell death through mitochondrial ANT3 and VDAC1. PLoS Pathog. 2005;1:e4. doi: 10.1371/journal.ppat.0010004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zamarin D, Ortigoza MB, Palese P. Influenza A virus PB1-F2 protein contributes to viral pathogenesis in mice. J Virol. 2006;80:7976–7983. doi: 10.1128/JVI.00415-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Garcia-Sastre A, Biron CA. Type 1 interferons and the virus-host relationship: a lesson in detente. Science. 2006;312:879–882. doi: 10.1126/science.1125676. [DOI] [PubMed] [Google Scholar]
- 141.Hale BG, Randall RE, Ortin J, Jackson D. The multifunctional NS1 protein of influenza A viruses. J Gen Virol. 2008;89:2359–2376. doi: 10.1099/vir.0.2008/004606-0. [DOI] [PubMed] [Google Scholar]
- 142.Li Z, Jiang Y, Jiao P, Wang A, Zhao F, Tian G, Wang X, Yu K, Bu Z, Chen H. The NS1 gene contributes to the virulence of H5N1 avian influenza viruses. J Virol. 2006;80:11115–11123. doi: 10.1128/JVI.00993-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Obenauer JC, Denson J, Mehta PK, Su X, Mukatira S, Finkelstein DB, Xu X, Wang J, Ma J, Fan Y, et al. Large-scale sequence analysis of avian influenza isolates. Science. 2006;311:1576–1580. doi: 10.1126/science.1121586. [DOI] [PubMed] [Google Scholar]
- 144.Li S, Schulman J, Itamura S, Palese P. Glycosylation of neuraminidase determines the neurovirulence of influenza A/WSN/33 virus. J Virol. 1993;67:6667–6673. doi: 10.1128/jvi.67.11.6667-6673.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Kawaoka Y, Webster RG. Sequence requirements for cleavage activation of influenza virus hemagglutinin expressed in mammalian cells. Proc Natl Acad Sci USA. 1988;85:324–328. doi: 10.1073/pnas.85.2.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Horimoto T, Kawaoka Y. Reverse genetics provides direct evidence for a correlation of hemagglutinin cleavability and virulence of an avian influenza A virus. J Virol. 1994;68:3120–3128. doi: 10.1128/jvi.68.5.3120-3128.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Senne DA, Panigrahy B, Kawaoka Y, Pearson JE, Suss J, Lipkind M, Kida H, Webster RG. Survey of the hemagglutinin (HA) cleavage site sequence of H5 and H7 avian influenza viruses: amino acid sequence at the HA cleavage site as a marker of pathogenicity potential. Avian Dis. 1996;40:425–437. doi: 10.2307/1592241. [DOI] [PubMed] [Google Scholar]
- 148.Itoh Y, Shinya K, Kiso M, Watanabe T, Sakoda Y, Hatta M, Muramoto Y, Tamura D, Sakai-Tagawa Y, Noda T, et al. In vitro and in vivo characterization of new swine-origin H1N1 influenza viruses. Nature. 2009;460:1021–1025. doi: 10.1038/nature08260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Lofgren E, Fefferman N, Naumov YN, Gorski J, Naumova EN. Influenza seasonality: underlying causes and modeling theories. J Virol. 2006;81:5429–5436. doi: 10.1128/JVI.01680-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Stone L, Olinky R, Huppert A. Seasonal dynamics of recurrent epidemics. Nature. 2007;446:533–536. doi: 10.1038/nature05638. [DOI] [PubMed] [Google Scholar]
- 151.Tellier R. Review of aerosol transmission of influenza A virus. Emerg Infect Dis. 2006;12:1657–1662. doi: 10.3201/eid1211.060426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Brankston G, Gitterman L, Hirji Z, Lemieux C, Gardam M. Transmission of influenza A in human beings. Lancet Infect Dis. 2007;7:257–265. doi: 10.1016/S1473-3099(07)70029-4. [DOI] [PubMed] [Google Scholar]
- 153.Lowen AC, Mubareka S, Tumpey TM, Garcia-Sastre A, Palese P. The guinea pig as a transmission model for human influenza viruses. Proc Natl Acad Sci USA. 2006;103:9988–9992. doi: 10.1073/pnas.0604157103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Mubareka S, Lowen AC, Steel J, Coates AL, Garcia-Sastre A, Palese P. Transmission of influenza virus via aerosols and fomites in the guinea pig model. J Infect Dis. 2009;199:858–865. doi: 10.1086/597073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Lowen AC, Mubareka S, Steel J, Palese P. Influenza virus transmission is dependent on relative humidity and temperature. PLoS Pathog. 2007;3:1470–1476. doi: 10.1371/journal.ppat.0030151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Lowen AC, Steel J, Mubareka S, Palese P. High temperature (30°C) blocks aerosol but not contact transmission of influenza virus. J Virol. 2008;82:5650–5652. doi: 10.1128/JVI.00325-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Lowen AC, Steel J, Mubareka S, Carnero E, Garcia-Sastre A, Palese P. Blocking interhost transmission of influenza virus by vaccination in the guinea pig model. J Virol. 2009;83:2803–2818. doi: 10.1128/JVI.02424-08. [DOI] [PMC free article] [PubMed] [Google Scholar]


