Abstract
Laboratory tests are an important part of the workup of small intestinal diseases in dogs and cats. Especially in chronic cases, when extragastrointestinal causes need to be ruled out, it is important to adhere to a systematic workup. This article details the newest available data on tests to aid this diagnostic process. Once the diagnosis of a chronic enteropathy is made, there are many laboratory tests that can help in monitoring the disease and providing prognostic information. Several new tests being evaluated for clinical usefulness are discussed.
Keywords: Diagnostic workup, Chronic diarrhea, Small intestine, Laboratory tests
Key points
-
•
A serum albumin concentration of less than 2 g/L is an indicator of poor prognosis in dogs with inflammatory bowel disease (IBD).
-
•
Cobalamin should be supplemented in all cases with decreased serum cobalamin concentrations.
-
•
Increased canine pancreatic lipase in dogs with IBD is associated with a worse outcome.
-
•
In cases of suspected intestinal lymphoma, polymerase chain reaction for antigen receptor rearrangements and immunophenotyping by flow cytometry or immunohistochemistry should be used in conjunction with clinical signs to help establish a diagnosis.
-
•
Evaluation of intestinal biopsies for expression of CD11c using immunofluorescence may be a helpful diagnostic test for IBD in dogs.
-
•
Genetic testing for mutations in innate immunity receptors is available for German Shepherd dogs, and could become a useful test for other breeds of dogs in the future.
Introduction: diagnostic workup of small intestinal disorders
The last decade has brought numerous advances in our knowledge about the pathogenesis of chronic intestinal disorders in people, particularly regarding inflammatory bowel disease (IBD), which comprises Crohn disease and ulcerative colitis. Specifically, the interplay of innate immunity receptors with commensals of the intestinal microbiome plays an important role in the disease pathogenesis. Molecular studies have identified specific disbalances in the microbiome of people with IBD. In addition, genetic polymorphisms that are associated with an increased risk of development of IBD have been identified. These data promise to be helpful in the development of new diagnostic options and targeted molecular treatment strategies for IBD. New findings in chronic enteropathies in dogs and cats suggest a pathogenesis similar to that in people with IBD. Recent studies have detected disbalances in expression of innate immunity receptors (so-called toll-like receptors [TLRs]) in the intestines of dogs with IBD1, 2 that are similar to those seen in people with IBD. The expression of some of these receptors also has been correlated with severity of clinical disease in dogs with IBD, which makes it likely that they are causally implicated in the pathogenesis.3 In addition, disbalances in the microbiome (so-called dysbiosis) have been identified using molecular methods in dogs and cats with IBD.4, 5, 6 These findings point toward a pathogenesis of IBD in dogs and cats similar to that in people, even if the clinical manifestations of these diseases are different. There is hope that similar advances regarding diagnostic options and new therapeutic modalities will be made for canine and feline IBD as has been done for IBD in humans.
A thorough history is important in the evaluation of small animal patients exhibiting signs of intestinal disorder. The first differentiation should be to establish whether the disease is acute or chronic. Diarrhea, vomiting, dehydration, weight loss, lethargy, and melena all can be signs of small intestinal disease. The disease is acute if clinical signs have been present for only a few days. However, if clinical signs persist for more than 3 weeks or are intermittently present for more than 3 weeks, the disease is defined as chronic.
If the animal has diarrhea, the next step is to determine whether it has small intestinal, large intestinal, or a combination of small and large intestinal diarrhea (Table 1 ).
Table 1.
Differentiation of small-bowel and large-bowel diarrhea
| Small | Large | |
|---|---|---|
| Volume | +++ | + |
| Mucus | − | +++ |
| Frequency | + | +++ |
| Tenesmus | − | +++ |
| Dyschezia | − | + |
| Weight loss | ++ | + |
| Vomiting | + | + |
| General condition | + | − |
Differential Diagnoses for Acute Small Intestinal Diseases
Systemically well
-
•
Dietary indiscretion
-
•
Intestinal parasites (Ancylostoma caninum, Toxocara, Giardia, Tritrichomonas fetus)
Systemically unwell, abnormal abdominal palpation, severe diarrhea with hematochezia, melena, and frequent vomiting
-
•
Dietary indiscretion
-
•
Toxicity
-
•
Viral infection (parvovirus, coronavirus, distemper, feline leukemia virus [FeLV]/feline immunodeficiency virus [FIV])
-
•
Bacterial infection (Salmonella, Campylobacter, Clostridium)
-
•
Intestinal parasites (Giardia, Tritrichomonas)
-
•
Acute pancreatitis
-
•
Intestinal obstruction
-
•
Hypoadrenocorticism (Addison disease)
Diagnostic workup of chronic small intestinal diarrhea
Once clinical signs persist for more than 3 weeks, additional workup is required to establish the diagnosis. In chronic cases it is more common for systemic, nongastrointestinal problems to cause the signs, and therefore the first diagnostic steps must be directed at excluding these extragastrointestinal causes.
Causes of Chronic Small Intestinal Disease
Extragastrointestinal (Metabolic) Causes
-
•
Hepatic disease (portosystemic shunt)
-
•
Hyperthyroidism (cats)
-
•
Hypoadrenocorticism (Addison disease) (dogs)
-
•
Renal insufficiency
-
•
Pancreatitis (acute or chronic)
-
•
Exocrine pancreatic insufficiency (EPI)
Gastrointestinal Causes
-
•
Intestinal parasites (Giardia infection, Tritrichomonas infection) (cats)
-
•
Chronic partial obstruction of the small intestine
-
•
Lymphangiectasia
-
•
Neoplasia: lymphosarcoma
-
•
Food intolerance/food allergy
-
•Chronic enteropathies/IBD
-
○Eosinophilic enteritis
-
○Lymphoplasmacytic enteritis
-
○
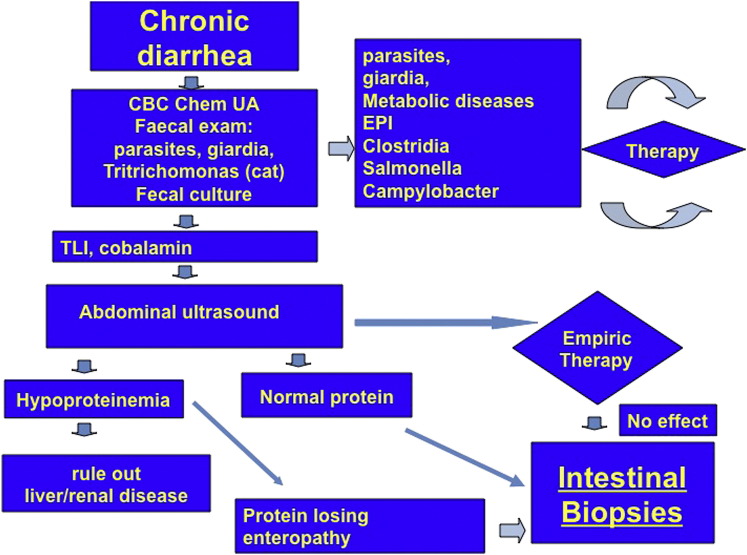
The diagnosis of chronic gastrointestinal causes is one of exclusion, and a full diagnostic workup needs to be done first to rule out all known causes of extragastrointestinal inflammation. This workup commonly involves a complete blood cell count, serum biochemical analysis, urinalysis, and fecal analysis for helminth and protozoal parasites (Giardia and Tritrichomonas in cats). Further tests are indicated if none of these tests are abnormal: trypsin-like immunoreactivity to exclude EPI, canine pancreatic lipase immunoreactivity (cPLI) to assess the possibility of pancreatitis or pancreatic tumors, corticotropin stimulation test or basal cortisol concentration to exclude hypoadrenocorticism, and cobalamin concentrations to assess the absorptive function of the distal small intestine. Total thyroxine (T4) and FeLV/FIV infection also should be assessed in cats. Abdominal ultrasonography will be more helpful than endoscopy in determining whether the small and/or large intestine is affected and whether there are mass lesions that need surgical intervention. If the results of these tests do not determine the cause for the clinical signs and the patient is stable (ie, has a normal appetite, is not lethargic, there is no or minimal weight loss, the serum protein concentration is normal, and there is no intestinal thickening on diagnostic imaging), a well-conducted therapeutic trial with an elimination diet or hydrolyzed diet for at least 2 weeks can be performed. If there is no response to a well-conducted dietary trial within 2 weeks after starting the diet, it is unlikely that the patient is suffering from food-responsive disease (FRD) (food allergy or food intolerance).7 If the dietary trial is unsuccessful, antimicrobials (metronidazole, 10–15 mg/kg by mouth twice a day or tylosin, 10 mg/kg by mouth once to twice a day) for 2 to 3 weeks can be administered. Intestinal biopsies for histopathology are collected from those patients that either fail to respond to empiric therapy or have worsening of their clinical signs. Most patients with chronic enteropathies can be diagnosed by obtaining endoscopic biopsies, as long as at least 12 to 15 biopsies from the small intestine are taken (Fig. 1 ). In rare cases, a diagnosis of lymphoma can be missed if no full-thickness biopsies are obtained, especially in cats, and if the ileum has not been sampled.
Fig. 1.
Diagnostic workup for dogs and cats presenting with signs of chronic small intestinal disease. CBC, complete blood cell count; Chem, serum biochemical profile; EPI, exocrine pancreatic insufficiency; UA, urinalysis.
Interpretation of laboratory tests to aid the diagnosis of chronic small intestinal diarrhea
Serum Albumin Concentrations
Dogs
Decreased serum albumin concentration has been described as a negative prognostic indicator in retrospective and prospective studies of IBD in dogs. Protein-losing enteropathy (PLE) accounts for the loss of albumin through the gut mucosa in severely affected dogs with IBD. PLE in dogs can be associated with severe lymphoplasmacytic IBD, intestinal lymphoma, or, rarely, primary lymphangiectasia. In one study of dogs with IBD, 12 of 80 (16%) dogs had hypoalbuminemia and 4 of 80 (5%) had panhypoproteinemia.8 Seven of 12 dogs with hypoalbuminemia had to be euthanized for intractable IBD, identifying decreased serum albumin concentration as a major risk factor associated with a worse outcome. In another recent prospective study of dogs with IBD, 12 of 58 (21%) dogs initially presented with hypoalbuminemia.7 Of these 12 dogs, 7 were panhypoproteinemic with severe hypoalbuminemia (mean albumin level 11 g/L), and 3 of these dogs eventually had to be euthanized. However, it must be noted that relatively mild reductions in serum albumin (<2 g/L) previously had been associated with an increased risk of refractoriness to treatment. At this level, most patients will not yet show any clinical signs of hypoalbuminemia, such as ascites, peripheral edema, or pleural effusion.
Furthermore, another study found that severely hypoalbuminemic dogs that failed to improve on immunosuppressive doses of steroids were successfully treated with cyclosporine.9 This finding suggests that early aggressive treatment in hypoalbuminemic dogs may potentially decrease mortality rates in severely ill animals. Serum albumin concentration also can be used to monitor patients, as improvement of serum albumin concentrations higher than 2 g/L usually indicates treatment success, even if clinical improvement can be seen earlier in some cases. It is therefore recommended to evaluate serum albumin concentrations every 2 to 3 weeks to assess when treatment can be tapered off or discontinued.
Cats
There is not much published information regarding serum albumin concentrations in cats with chronic intestinal disease. PLE as a clinical syndrome does not exist in cats, as clinical signs such as ascites and peripheral edema do not usually occur in cats with hypoalbuminemia caused by intestinal disease. In addition, the hypoalbuminemia seen in such cases is usually mild. In cats with IBD, the prevalence of hypoalbuminemia ranged from 5% to 24%.10, 11, 12 However, there is evidence that cats with chronic intestinal disease and decreased serum albumin concentrations may have concurrent pancreatic disease.
In one recent retrospective study, cats with IBD and serum feline pancreatic lipase (fPLI) concentrations of 2.0 μg/L or higher had a lower median serum albumin concentration than cats with IBD and a normal fPLI.13 However, hypoalbuminemia was not a negative predictor of survival in this study. Another study found that cats with moderate to severe pancreatitis were significantly more likely to be hypoalbuminemic than were healthy cats and cats with mild pancreatitis.14
Therefore, hypoalbuminemia in cats with chronic intestinal disease should prompt the clinician to measure fPLI concentrations and/or to perform abdominal ultrasonographic examination to determine if there is concurrent pancreatitis. Depending on the severity of the hypoalbuminemia, the clinician’s approach to treatment might be altered.
Serum Cobalamin Concentrations in Dogs and Cats
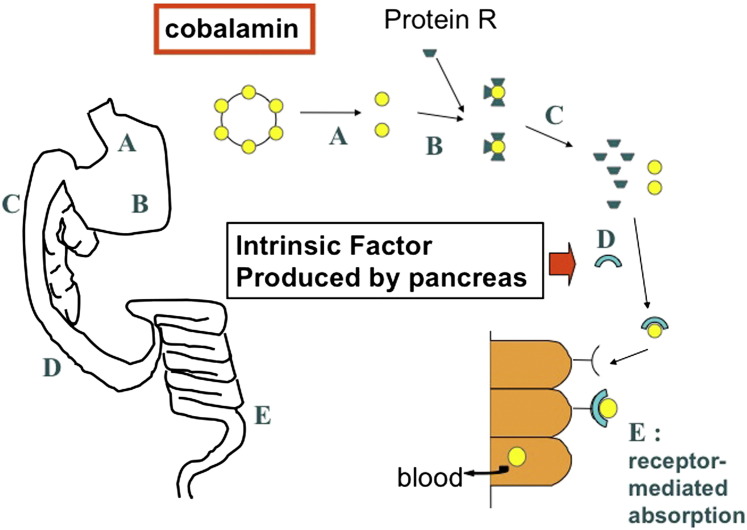
Serum cobalamin concentrations should be measured in any small animal patient with chronic small intestinal disease. Cobalamin absorption is receptor-mediated in the ileum, and decreased serum cobalamin concentrations are most commonly seen when this part of the small intestine is affected. However, absorption of cobalamin also involves intrinsic factor, which in dogs and cats is produced primarily in the pancreas. For this reason most small animals with EPI have low serum cobalamin concentrations (Fig. 2 ). The author’s group7 has shown recently that serum cobalamin also is very important for prognosis in dogs with chronic enteropathies. If cobalamin serum concentration is below the reference interval, the risk for later euthanasia increases by a factor of 10. It is therefore important to supplement dogs with hypocobalaminemia while they undergo treatment of IBD, as this “risk of euthanasia” can be reversed by cobalamin supplementation.
Fig. 2.
Absorption of cobalamin is a complex process involving several steps. Cobalamin is released from food protein in the stomach (A) and immediately bound to R-binder proteins (B). In the proximal small intestine, the cobalamin-R-binder complex is cleaved after digestion of the R-binder by pancreatic proteases (C). Free cobalamin can now bind to intrinsic factor (IF) (D), the majority of which is secreted by the pancreas in cats and dogs. This cobalamin-IF complex is subsequently absorbed by specialized receptors in the ileum (E).
Cats
Serum cobalamin concentration has long been known to be an important negative prognostic factor in cats with chronic enteropathies.15 The prevalence of decreased serum cobalamin concentrations in cats with chronic gastrointestinal signs has been reported to be up to 16.5%.16 In cats, it has also been reported that cobalamin supplementation can improve clinical signs regardless of the underlying diagnosis, and even if given as the sole treatment for their disease.15 It is therefore recommended that cats with chronic intestinal disease are supplemented with cobalamin regardless of whether a specific cause for the disease can be identified.
Supplementation recommendations for dogs and cats
Supplementation of cobalamin should be given parenterally (subcutaneously) as a weekly injection for at least 6 weeks. Exact dosages are not reported, as it is a water-soluble vitamin and cannot be overdosed. For tested recommendations, please visit the Web site of the Texas GI Laboratory (http://vetmed.tamu.edu/gilab).
Canine Pancreatic Lipase
cPLI has recently become available as a commercial test and is useful in the assessment of pancreatitis in dogs.17 However, cPLI also can be elevated in dogs with chronic enteropathy. In a retrospective study of 50 dogs with IBD, the author’s group18 evaluated clinical signs, age, serum lipase and amylase activities, albumin and cobalamin concentrations, abdominal ultrasonography results, histopathologic review of intestinal biopsies, management of IBD, and follow-up in dogs with IBD, either with or without concurrent chronic pancreatitis. Sixteen dogs with increased cPLI and 32 dogs with normal cPLI values were compared. No significant differences were found for clinical activity score, serum amylase activity, serum lipase activity, serum cobalamin concentration, serum albumin concentration, abdominal ultrasonography scores, and histopathology scores for IBD. There was no difference in the frequency of steroid treatment between the groups. Dogs with IBD and concurrent elevated cPLI were significantly older than dogs without elevated cPLI. Moreover, dogs with elevated cPLI had a higher risk of a poor follow-up score and were significantly more likely to be euthanized at follow-up. These data show that elevated cPLI in canine IBD may indicate that a subset of these patients could also have chronic subclinical pancreatitis. In patients that have been diagnosed with IBD and also have elevated cPLI without overt imaging evidence of acute pancreatitis, it is recommended to discuss treatment options for IBD that also will treat possible autoimmune pancreatitis. The author has had anecdotal success with cyclosporine at 5 mg/kg daily for 8 weeks in such cases.
C-Reactive Protein
C-reactive protein (CRP) is a serum acute-phase protein that can be elevated in many different diseases. In people with IBD, several calculated indices of clinical activity of disease incorporate measurements of CRP.19 In dogs, a similar correlation between the canine IBD activity index (CIBDAI) and serum CRP concentration has been found in one large study of 58 dogs.20 CRP was elevated in the 28 dogs with CIBDAI scores greater than 5 (which comprises mild to moderate disease activity) in comparison with normal dogs, and CRP decreased significantly after treatment.20
In the author’s experience, CRP is not very helpful when assessing dogs with chronic enteropathies. CRP was measured in 21 dogs with IBD before treatment and in 18 dogs after treatment.7 CRP was elevated in only 6 of 21 dogs before treatment, and did not correlate with CIBDAI or histologic scoring.
A large percentage of dogs with IBD do not show any elevations in CRP. Interpretation of elevated levels also may be hampered by increases related to diseases other than IBD.
Fecal α1-Proteinase Inhibitor
Fecal α1-proteinase inhibitor (α1-PI) can be used as a test for dogs in which the clinician suspects PLE although the clinical signs are not yet overtly visible. α1-PI is a plasma protein similar in size to albumin. If the intestinal mucosal barrier is compromised and loss of protein into the intestinal lumen occurs, α1-PI is lost at approximately the same rate as albumin. Unlike albumin, however, its proteinase inhibitor properties protect α1-PI from degradation by intestinal proteases, and can be measured in feces. The test recently has been validated for dogs.21
Prompt diagnosis of PLE in a patient with IBD is important because hypoalbuminemia is a risk factor for negative outcome,7 and the cause should be treated aggressively to improve survival. The α1-PI assay is especially valuable in patients with intestinal disease that have concurrent renal or hepatic disease. In these patients, measurement of fecal α1-PI can help assess which portion, if any, of the protein loss can be attributed to the intestine. This test is available only at the Texas GI Laboratory. Ideally, 3 consecutive fresh fecal samples should be submitted to improve test accuracy, which means that fecal α1-PI is not a useful test for practitioners outside North America.
Histology and World Small Animal Veterinary Association (WSAVA) Scoring of Intestinal Biopsies
Sampling of intestinal biopsies is an essential step in the evaluation of small intestinal disorders, to exclude neoplastic causes and confirm the presence of intestinal inflammation. However, interpretation of intestinal biopsies is difficult and subject to controversy. In several recent studies looking at conventional histologic interpretation of intestinal biopsies, there was no correlation between clinical activity and histologic grading either before or after therapy.7, 20, 22 In addition, total lymphocyte counts as well as the number of infiltrating CD3 cells in the lamina propria cannot be used as markers for clinical activity of disease, as there is no difference in cell counts before and after treatment.23 These findings suggest that the type and degree of histologic infiltrates in canine IBD may not be as helpful as in human medicine, in which clinical scores correlate very well with histologic grading. Therefore, a new grading scheme for the histologic interpretation of endoscopically obtained biopsies from dogs and cats with IBD has recently been published by the WSAVA Working Group.24 The findings in this study suggest that microarchitectural changes seem to be much more important than cellular infiltrates when assessing histologic severity. However, there is limited information on how well this new grading system correlates with clinical disease. In one retrospective study, the interpathologist variability was still very high even when using the picture guide from the original publication.25 In addition, it is of concern that the only parameter that correlated with clinical disease was the presence of lymphangiectasia and hypoalbuminemia. Further prospective studies are warranted before the WSAVA scoring can be adopted as a useful tool for clinicians.
PARR in Intestinal Biopsies
The polymerase chain reaction for antigen receptor rearrangements (PARR) amplifies the highly variable T- or B-cell antigen receptor genes, and is used to detect the presence of a clonally expanded population of lymphocytes. This test has been advocated as useful when applied on endoscopically sampled biopsies if a diagnosis of intestinal lymphoma is suspected but not confirmed by conventional histopathology. In a study at the Royal Veterinary College, the author prospectively evaluated the accuracy of PARR for the diagnosis of intestinal lymphoma in biopsies obtained endoscopically from dogs in a comparison with the gold standard of histopathology and clinical outcome determined by follow-up information of at least 2 years. Samples from 39 dogs were included. PARR results indicated a clonal expansion in 7 of 36 dogs. However, these dogs were clinically healthy after dietary treatment 2 years after the endoscopy, so they clearly did not have lymphoma. The data from this study indicate a false-positive rate of almost 20% for PARR when performed on endoscopic biopsies. Another recent study has confirmed these findings, showing that in dogs with IBD, PARR results showed at least one oligoclonal pattern in 38% of dogs, and an immunoglobulin (7 of 47; 14.9%) or T-cell receptor (1 of 47: 2.1%) monoclonal pattern in 17% of dogs.26 The conclusion that a positive PARR test on an endoscopic biopsy means a diagnosis of lymphoma must therefore be made cautiously in a clinical situation, and clinical signs, response to treatment, and immunohistochemistry must also be taken into account.
Perinuclear Antineutrophilic Cytoplasmic Antibodies
Perinuclear antineutrophilic cytoplasmic antibodies (pANCA) have been useful in the diagnosis of human IBD for decades.27 These antibodies are serum autoantibodies similar to antinuclear antibodies (ANA), but seem to be more specific for intestinal disease than for ANA. pANCA are detected by immunofluorescence by visualizing a typical pattern of perinuclear staining.
In the first study to assess the clinical usefulness of pANCA in dogs with IBD, sensitivity for pANCA was 0.51 and specificity ranged between 0.56 and 0.95. pANCA proved to be a highly specific marker for IBD in dogs when the group of dogs with chronic diarrhea of other causes were tested against dogs with IBD (specificity 0.95).28 This finding is in agreement with reports from human medicine that show a specificity of up to 94% for pANCA when distinguishing between IBD and healthy controls, as well as patients with non–IBD-related diarrhea from other causes.29 When pANCA were tested in a group of dogs with FRD and compared with pANCA in dogs with steroid-responsive disease, a positive pANCA titer was significantly associated with FRD.30
The pANCA assay might be helpful in differentiating dogs with chronic diarrhea caused by FRD or IBD: If the result is positive, a food-responsive chronic enteropathy is highly likely, however, if the result is negative, IBD cannot be excluded.
pANCA also may be associated with the syndrome of familial PLE in soft-coated wheaten terriers (SCWT).31 pANCA were detectable in the serum of dogs an average 1 to 2 years before the onset of clinical disease, and were highly correlated with hypoalbuminemia. This test could be a useful screening test for this specific disease in SCWT.
Care must be taken in interpreting a positive pANCA test result if other inflammatory or immune-mediated diseases are present. A recent study showed that many dogs with various vector-borne diseases or immune-mediated hemolytic anemia were positive for pANCA.32
Calprotectin and S100A12
Calprotectin and S100A12 are calcium-binding proteins that are abundant in the granules of neutrophils and macrophages. In people with IBD, serum and fecal concentrations of these proteins are increased in comparison with healthy people. In addition, fecal concentrations of calprotectin correlate very well with clinical disease activity in children with IBD.33
An immunoassay for measurement of canine calprotectin in serum and fecal samples is available.34 A serum calprotectin concentration of 296.0 μg/L or higher has sensitivity of 82.4% and specificity of 68.4% for distinguishing dogs with idiopathic IBD from healthy dogs. However, calprotectin concentrations were not significantly correlated with clinical severity, serum CRP concentration, or severity of histopathologic changes. The clinical usefulness of this test needs further evaluation.
Immunohistochemistry for P-Glycoprotein on Intestinal Biopsies
P-glycoprotein (P-gp) is a transmembrane protein that functions as a drug-efflux pump in the intestinal epithelium. Human patients with IBD who fail to respond to treatment with glucocorticosteroids express high levels of P-gp in lamina propria lymphocytes.35 Two research groups have evaluated P-gp expression in biopsies of dogs with IBD. In one study,36 duodenal biopsies from 48 dogs were evaluated by immunohistochemistry. Biopsies were evaluated after treatment with prednisolone in 15 dogs and after dietary therapy alone in 16 dogs. Dogs treated with prednisolone showed significantly higher P-gp expression in lamina propria lymphocytes after treatment compared with expression before treatment. By contrast, the group treated solely with an elimination diet showed no difference in P-gp scores before and after treatment. Moreover, a statistically significant association between refractoriness to steroid treatment and high P-gp expression was found in the glucocorticosteroid-treated group.36 In another recent study, P-gp expression was higher in duodenal epithelial cells of dogs with IBD compared with healthy control dogs.37 However, there was no difference in P-gp expression in colonic epithelial cells between IBD and control groups. These results indicate that epithelial and lamina propria lymphocyte expression of P-gp is upregulated in dogs with IBD, and they are even higher after prednisolone treatment. In addition, high P-gp expression could indicate possible multidrug resistance and should be taken into account when managing dogs that have failed steroid treatment previously.
Immunohistochemistry for CD11c in Intestinal Biopsies
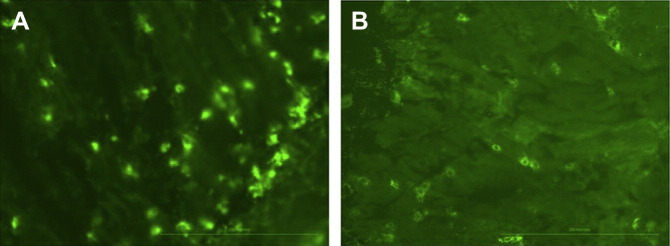
CD11c is a marker of human and murine dendritic cells (DCs), and cells expressing this marker have been shown to have similar morphologic and functional characteristics in dogs. DCs are important in determining the outcome of an immune reaction in the gut, that is, whether a pathogen will elicit a massive immune response or whether a commensal will induce tolerance.38 Specific subsets of inducible DCs are decreased in the diseased tissues of people with IBD.39 It is plausible that the number of DCs in the intestine could be used as a surrogate marker of inflammation in dogs with IBD. In one recent study, endoscopic biopsies from the duodenum, ileum, and colon were obtained from dogs with IBD and healthy dogs.40 CD11c expression was assessed by immunofluorescence using a canine monoclonal antibody (Fig. 3 ). The number of CD11c-positive cells in the duodenum, ileum, and colon of dogs with IBD was significantly reduced in comparison with controls. There was a significant negative correlation between the number of CD11c-positive cells in the colon of dogs with IBD and clinical severity. This marker therefore holds promise as a useful test to assess histologic samples. However, additional prospective studies are needed to evaluate the clinical utility of this test.
Fig. 3.
Immunofluorescence for CD11c on intestinal biopsies from (A) a healthy dog and (B) a dog with IBD. CD11c expression in the intestinal mucosa is more abundant in healthy dogs than in dogs with IBD (original magnification ×100).
Genetic Testing
Over the last decade, numerous genes have been associated with an increased risk of development of IBD in humans, many of them implicated in the innate immune response in the intestine.41 Dogs with IBD may have a similar genetic component, especially because there are breeds predisposed to certain forms of IBD. Boxers are predisposed to histiocytic ulcerative colitis, and German shepherd dogs (GSD) are predisposed to lymphoplasmacytic IBD.42 The author’s group43 recently performed a mutational analysis of the canine genes for TLR2, TLR4, TLR5, and NOD2 in GSD with IBD, and then further evaluated these in a case-control study with more than 50 cases and healthy GSD controls. Several mutations in TLR4 and TLR5 were found to be significantly associated with an increased risk of development of IBD. Moreover, these results were replicated in 38 other non-GSD breeds for the TLR5 mutation.44 A follow-up study showed that peripheral blood cells of dogs carrying the mutation are hyperresponsive to flagellin, which is the natural ligand for TLR5.45 This finding proves for the first time that a genetic mutation implicated in the pathogenesis of dogs with IBD has functional consequences at the protein level. Taken together, these findings make it very likely that TLR5 mutations are causally associated with canine IBD. Genetic testing for these polymorphisms currently is available only at the Royal Veterinary College. Such tests could become important for breeders and practitioners in the future. However, it is likely that in a multifactorial disease such as IBD in dogs, other genetic mutations and environmental factors also play a role in the pathogenesis. With the advent of genome-wide association studies, it is possible that more causative mutations will be identified.
Summary
Many laboratory tests are available to aid the diagnostic workup of cats and dogs with chronic small intestinal disorders. Some of these have been available for many years, such as serum albumin and cobalamin concentrations, as well as canine pancreatic lipase, and new data now show that these tests also may be prognostic indicators in animals with chronic enteropathy. Other tests have only relatively recently become available to practitioners, such as serum CRP, fecal α1-PI, WSAVA standardization of histopathology readings, and PARR. The value of these tests needs to be evaluated in every clinical situation. New tests that are not yet widely available, such as pANCA, calprotectin, CD11c immunofluorescence, and genetic testing, may become very useful tests in the future.
Footnotes
Current Funding Sources: Morris Animal Foundation, British Biotechnology and Bioscience Research Fund, Probiotics Ltd UK, Laboklin GmbH Germany.
Conflict of Interests: None.
References
- 1.Burgener I.A., Konig A., Allenspach K. Upregulation of toll-like receptors in chronic enteropathies in dogs. J Vet Intern Med. 2008;22:553–560. doi: 10.1111/j.1939-1676.2008.0093.x. [DOI] [PubMed] [Google Scholar]
- 2.Allenspach K., House A., Smith K. Evaluation of mucosal bacteria and histopathology, clinical disease activity and expression of Toll-like receptors in German shepherd dogs with chronic enteropathies. Vet Microbiol. 2010;146:326–335. doi: 10.1016/j.vetmic.2010.05.025. [DOI] [PubMed] [Google Scholar]
- 3.McMahon L.A., House A.K., Catchpole B. Expression of Toll-like receptor 2 in duodenal biopsies from dogs with inflammatory bowel disease is associated with severity of disease. Vet Immunol Immunopathol. 2010;135:158–163. doi: 10.1016/j.vetimm.2009.11.012. [DOI] [PubMed] [Google Scholar]
- 4.Janeczko S., Atwater D., Bogel E. The relationship of mucosal bacteria to duodenal histopathology, cytokine mRNA, and clinical disease activity in cats with inflammatory bowel disease. Vet Microbiol. 2008;128:178–193. doi: 10.1016/j.vetmic.2007.10.014. [DOI] [PubMed] [Google Scholar]
- 5.Xenoulis P.G., Palculict B., Allenspach K. Molecular-phylogenetic characterization of microbial communities imbalances in the small intestine of dogs with inflammatory bowel disease. FEMS Microbiol Ecol. 2008;66:579–589. doi: 10.1111/j.1574-6941.2008.00556.x. [DOI] [PubMed] [Google Scholar]
- 6.Suchodolski J.S., Camacho J., Steiner J.M. Analysis of bacterial diversity in the canine duodenum, jejunum, ileum, and colon by comparative 16S rRNA gene analysis. FEMS Microbiol Ecol. 2008;66(3):567–578. doi: 10.1111/j.1574-6941.2008.00521.x. [DOI] [PubMed] [Google Scholar]
- 7.Allenspach K., Wieland B., Grone A. Chronic enteropathies in dogs: evaluation of risk factors for negative outcome. J Vet Intern Med. 2007;21:700–708. doi: 10.1892/0891-6640(2007)21[700:ceideo]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 8.Craven M., Simpson J.W., Ridyard A.E. Canine inflammatory bowel disease: retrospective analysis of diagnosis and outcome in 80 cases (1995-2002) J Small Anim Pract. 2004;45:336–342. doi: 10.1111/j.1748-5827.2004.tb00245.x. [DOI] [PubMed] [Google Scholar]
- 9.Allenspach K., Rufenacht S., Sauter S. Pharmacokinetics and clinical efficacy of cyclosporine treatment of dogs with steroid-refractory inflammatory bowel disease. J Vet Intern Med. 2006;20:239–244. doi: 10.1892/0891-6640(2006)20[239:paceoc]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 10.Jergens A.E. Feline idiopathic inflammatory bowel disease: What we know and what remians to be unraveled. J Fel Med Surg. 2012;14(7):445–458. doi: 10.1177/1098612X12451548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dennis J.S., Kruger J.M., Mullaney T.P. Lymphocytic/plasmacytic colitis in cats: 14 cases (1985-1990) J Am Vet Med Assoc. 1993;202:313–318. [PubMed] [Google Scholar]
- 12.Baez J.L., Hendrick M.J., Walker L.M. Radiographic, ultrasonographic, and endoscopic findings in cats with inflammatory bowel disease of the stomach and small intestine: 33 cases (1990-1997) J Am Vet Med Assoc. 1999;215:349–354. [PubMed] [Google Scholar]
- 13.Bailey S., Benigni L., Eastwood J. Comparisons between cats with normal and increased fPLI concentrations in cats diagnosed with inflammatory bowel disease. J Small Anim Pract. 2010;51:484–489. doi: 10.1111/j.1748-5827.2010.00973.x. [DOI] [PubMed] [Google Scholar]
- 14.Forman M.A., Marks S.L., De Cock H.E. Evaluation of serum feline pancreatic lipase immunoreactivity and helical computed tomography versus conventional testing for the diagnosis of feline pancreatitis. J Vet Intern Med. 2004;18:807–815. doi: 10.1892/0891-6640(2004)18<807:eosfpl>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 15.Ruaux C.G., Steiner J.M., Williams D.A. Early biochemical and clinical responses to cobalamin supplementation in cats with signs of gastrointestinal disease and severe hypocobalaminemia. J Vet Intern Med. 2005;19:155–160. doi: 10.1892/0891-6640(2005)19<155:ebacrt>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 16.Reed N., Gunn-Moore D., Simpson K. Cobalamin, folate and inorganic phosphate abnormalities in ill cats. J Feline Med Surg. 2007;9:278–288. doi: 10.1016/j.jfms.2007.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mansfield C. Acute panceatitis in dogs: Advances in understanding, diagnostics and treatment. Top Compan Anim Med. 2012;27(3):123–132. doi: 10.1053/j.tcam.2012.04.003. [DOI] [PubMed] [Google Scholar]
- 18.Kathrani A., Steiner J.M., Suchodolski J. Elevated canine pancreatic lipase immunoreactivity concentration in dogs with inflammatory bowel disease is associated with a negative outcome. J Small Anim Pract. 2009;50:126–132. doi: 10.1111/j.1748-5827.2008.00693.x. [DOI] [PubMed] [Google Scholar]
- 19.Nielsen O.H., Vainer B., Madsen S.M. Established and emerging biological activity markers of inflammatory bowel disease. Am J Gastroenterol. 2000;95:359–367. doi: 10.1111/j.1572-0241.2000.t01-1-01790.x. [DOI] [PubMed] [Google Scholar]
- 20.Jergens A.E., Schreiner C.A., Frank D.E. A scoring index for disease activity in canine inflammatory bowel disease. J Vet Intern Med. 2003;17:291–297. doi: 10.1111/j.1939-1676.2003.tb02450.x. [DOI] [PubMed] [Google Scholar]
- 21.Heilmann R.M., Paddock C.G., Ruhnke I. Development and analytical validation of a radioimmunoassay for the measurement of alpha1-proteinase inhibitor concentrations in feces from healthy puppies and adult dogs. J Vet Diagn Invest. 2011;23:476–485. doi: 10.1177/1040638711404152. [DOI] [PubMed] [Google Scholar]
- 22.Garcia-Sancho M., Rodriguez-Franco F., Sainz A. Evaluation of clinical, macroscopic, and histopathologic response to treatment in nonhypoproteinemic dogs with lymphocytic-plasmacytic enteritis. J Vet Intern Med. 2007;21:11–17. doi: 10.1892/0891-6640(2007)21[11:eocmah]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 23.Schreiner N.M., Gaschen F., Grone A. Clinical signs, histology, and CD3-positive cells before and after treatment of dogs with chronic enteropathies. J Vet Intern Med. 2008;22:1079–1083. doi: 10.1111/j.1939-1676.2008.0153.x. [DOI] [PubMed] [Google Scholar]
- 24.Day M.J., Bilzer T., Mansell J. Histopathological standards for the diagnosis of gastrointestinal inflammation in endoscopic biopsy samples from the dog and cat: a report from the World Small Animal Veterinary Association Gastrointestinal Standardization Group. J Comp Pathol. 2008;138(Suppl 1):S1–S43. doi: 10.1016/j.jcpa.2008.01.001. [DOI] [PubMed] [Google Scholar]
- 25.Willard M., Mansell J. Correlating clinical activity and histopathologic assessment of gastrointestinal lesion severity: current challenges. Vet Clin North Am Small Anim Pract. 2011;41:457–463. doi: 10.1016/j.cvsm.2011.01.005. [DOI] [PubMed] [Google Scholar]
- 26.Olivero D., Turba M.E., Gentilini F. Reduced diversity of immunoglobulin and T-cell receptor gene rearrangements in chronic inflammatory gastrointestinal diseases in dogs. Vet Immunol Immunopathol. 2011;144:337–345. doi: 10.1016/j.vetimm.2011.08.011. [DOI] [PubMed] [Google Scholar]
- 27.Vermeire S., Peeters M., Rutgeerts P. Diagnostic approach to IBD. Hepatogastroenterology. 2000;47:44–48. [PubMed] [Google Scholar]
- 28.Allenspach K., Luckschander N., Styner M. Evaluation of assays for perinuclear antineutrophilic cytoplasmic antibodies and antibodies to Saccharomyces cerevisiae in dogs with inflammatory bowel disease. Am J Vet Res. 2004;65:1279–1283. doi: 10.2460/ajvr.2004.65.1279. [DOI] [PubMed] [Google Scholar]
- 29.Dubinsky M.C., Ofman J.J., Urman M. Clinical utility of serodiagnostic testing in suspected pediatric inflammatory bowel disease. Am J Gastroenterol. 2001;96:758–765. doi: 10.1111/j.1572-0241.2001.03618.x. [DOI] [PubMed] [Google Scholar]
- 30.Luckschander N., Allenspach K., Hall J. Perinuclear antineutrophilic cytoplasmic antibody and response to treatment in diarrheic dogs with food responsive disease or inflammatory bowel disease. J Vet Intern Med. 2006;20:221–227. doi: 10.1892/0891-6640(2006)20[221:pacaar]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 31.Allenspach K., Lomas B., Wieland B. Evaluation of perinuclear anti-neutrophilic cytoplasmic autoantibodies as an early marker of protein-losing enteropathy and protein-losing nephropathy in soft coated wheaten terriers. Am J Vet Res. 2008;69:1301–1304. doi: 10.2460/ajvr.69.10.1301. [DOI] [PubMed] [Google Scholar]
- 32.Karagianni A.E., Solano-Gallego L., Breitschwerdt E.B. Perinuclear antineutrophil cytoplasmic autoantibodies in dogs infected with various vector-borne pathogens and in dogs with immune-mediated hemolytic anemia. Am J Vet Res. 2012;73:1403–1409. doi: 10.2460/ajvr.73.9.1403. [DOI] [PubMed] [Google Scholar]
- 33.Aadland E., Fagerhol M.K. Faecal calprotectin: a marker of inflammation throughout the intestinal tract. Eur J Gastroenterol Hepatol. 2002;14:823–825. doi: 10.1097/00042737-200208000-00002. [DOI] [PubMed] [Google Scholar]
- 34.Heilmann R.M., Jergens A.E., Ackermann M.R. Serum calprotectin concentrations in dogs with idiopathic inflammatory bowel disease. Am J Vet Res. 2012;73:1900–1907. doi: 10.2460/ajvr.73.12.1900. [DOI] [PubMed] [Google Scholar]
- 35.Farrell R.J., Menconi M.J., Keates A.C. P-glycoprotein-170 inhibition significantly reduces cortisol and cyclosporin efflux from human intestinal epithelial cells and T lymphocytes. Aliment Pharmacol Ther. 2002;16:1021–1031. doi: 10.1046/j.1365-2036.2002.01238.x. [DOI] [PubMed] [Google Scholar]
- 36.Allenspach K., Bergman P.J., Sauter S. P-glycoprotein expression in lamina propria lymphocytes of duodenal biopsy samples in dogs with chronic idiopathic enteropathies. J Comp Pathol. 2006;134:1–7. doi: 10.1016/j.jcpa.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 37.Van der Heyden S., Vercauteren G., Daminet S. Expression of P-glycoprotein in the intestinal epithelium of dogs with lymphoplasmacytic enteritis. J Comp Pathol. 2011;145:199–206. doi: 10.1016/j.jcpa.2011.01.003. [DOI] [PubMed] [Google Scholar]
- 38.Ng S.C., Benjamin J.L., McCarthy N.E. Relationship between human intestinal dendritic cells, gut microbiota, and disease activity in Crohn's disease. Inflamm Bowel Dis. 2011;17:2027–2037. doi: 10.1002/ibd.21590. [DOI] [PubMed] [Google Scholar]
- 39.Ng S.C., Kamm M.A., Stagg A.J. Intestinal dendritic cells: their role in bacterial recognition, lymphocyte homing, and intestinal inflammation. Inflamm Bowel Dis. 2010;16:1787–1807. doi: 10.1002/ibd.21247. [DOI] [PubMed] [Google Scholar]
- 40.Kathrani A., Schmitz S., Priestnall S.L. CD11c+ cells are significantly decreased in the duodenum, ileum and colon of dogs with inflammatory bowel disease. J Comp Pathol. 2011;145:359–366. doi: 10.1016/j.jcpa.2011.03.010. [DOI] [PubMed] [Google Scholar]
- 41.Lees C.W., Barrett J.C., Parkes M. New IBD genetics: common pathways with other diseases. Gut. 2011;60:1739–1753. doi: 10.1136/gut.2009.199679. [DOI] [PubMed] [Google Scholar]
- 42.Kathrani A., Werling D., Allenspach K. Canine breeds at high risk of developing inflammatory bowel disease in the south-eastern UK. Vet Rec. 2011;169:635. doi: 10.1136/vr.d5380. [DOI] [PubMed] [Google Scholar]
- 43.Kathrani A., House A., Catchpole B. Polymorphisms in the TLR4 and TLR5 gene are significantly associated with inflammatory bowel disease in German shepherd dogs. PLoS One. 2010;5:e15740. doi: 10.1371/journal.pone.0015740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kathrani A., House A., Catchpole B. Breed-independent toll-like receptor 5 polymorphisms show association with canine inflammatory bowel disease. Tissue Antigens. 2011;78:94–101. doi: 10.1111/j.1399-0039.2011.01707.x. [DOI] [PubMed] [Google Scholar]
- 45.Kathrani A., Holder A., Catchpole B. TLR5 Risk-associated haplotype for canine inflammatory bowel disease confers hyper-responsiveness to flagellin. PLoS One. 2012;7:e30117. doi: 10.1371/journal.pone.0030117. [DOI] [PMC free article] [PubMed] [Google Scholar]