Astroviruses (AstVs) are small nonenveloped icosahedral viruses. Their genome is composed of plus-sense, single-stranded RNA with a 3′ polyadenylated [poly(A)] tail. AstVs have been identified from numerous animal species, including humans, and mainly associated with enteric disease.1 Nonenteric disease has been also described in avian species.2 Even more interestingly, AstVs have been detected in the central nervous system tissue of mink with neurologic disease3 and of an immunocompromised patient with encephalitis.4 Canine AstVs were first identified in the early 1980s,5 but they have been recently “rediscovered” and characterized at the molecular level as a distinct species within the Mamastrovirus genus.6, 7, 8
Causes
AstVs (family Astroviridae) are small-rounded viruses (SRVs) with a peculiar starlike shape when observed on electron microscopy (EM), although this conformation is not always readily recognizable. AstV genome is composed of a plus-sense single-stranded RNA of 6.4 of 7.3 kb in size, containing 3 open reading frames (ORFs) and with a 3′ poly(A) tail.1 Two ORFs, located at the 5′ end of the genome (ORF1a and ORF1b), encode nonstructural proteins, while ORF2, located at the 3′ end, encodes the capsid protein.1 AstVs were first identified by EM in 1975 in Scotland in the stools of infants hospitalized with diarrhea.9 Subsequently, similar SRVs have been identified from several mammalian and avian species,10, 11, 12, 13, 14, 15, 16, 17, 18 including bats,19 rats,20 and aquatic mammals.21 AstV infection is associated with gastroenteritis in most animal species, and human AstVs are regarded as the second or third most common cause of viral diarrhea in children.1 AstVs have also been associated with extraintestinal diseases, such as nephritis in chicken,2 hepatitis in ducks,16 and shaking syndrome in mink.3
Based on the species of origin of the virus, the genome structure and genetic homology, 2 genera have been defined within the family Astroviridae: Mamastrovirus, which infect mammals, and Avastrovirus, which include viruses detected from avian species. As viruses genetically highly diverse can circulate in a given host species,22 a novel classification system based on comparison of the full length ORF2, the capsid precursor, has been proposed (http://talk.ictvonline.org/files/proposals/taxonomy_proposals_vertebrate1/m/vert01/2287.aspx). In this classification, 2 genogroups (GI and II) have been defined in Mamastrovirus genus. AstV strains within the same species display a p-distance lower than 0.312 and species are assigned consecutive letters. In this system, human AstVs type 1 to type 8 are GI.A, feline AstV is GI.B, and canine AstVs are GI.E.
Novel AstVs have been recently detected in humans, although only in a sporadic fashion. These novel human AstVs are distantly related to “classic” human AstV types 1 to 8, and more similar genetically to some animal AstVs.23, 24, 25 Antibodies to one such novel AstV, strain MAstV/GII.B/Hu/THA/2001/NE-3010, have been detected in 20% to 36% of the sera of children.26 These findings suggest recent interspecies transmission events of AstVs from animals to humans and, more importantly, widespread exposure of humans to these viruses.
Astroviruses in Dogs
AstV-like particles have been detected only occasionally in dogs on EM. In some cases, due to their morphologic similarities (about 25 to 35 nm, rounded, absence of envelope) with caliciviruses and picornaviruses, they have generically been referred to as SRVs. AstV-like particles were first detected on EM in beagle pups with diarrheal disease in the United States in 1980, in mixed infection with canine parvovirus type-2 (CPV2) and canine coronaviruses (CCoV).5 AstV-like particles were also detected on EM in 3 of 157 normal fecal samples (but not in 29 diarrheal samples) in a survey in Australia in 1984.27 In a large EM-based survey in Germany, SRVs were identified in 41 of 4044 (about 1%) feces of dogs with diarrhea.28 More recently, AstVs have been identified in dogs with enteric signs and characterized molecularly, suggesting that the detected viruses may represent a distinct AstV species.6, 7, 8
While early evidence suggested that AstV infection in dogs is rare or occasional, recent investigations based on sensitive and specific assays indicate that AstVs are indeed widespread in the canine population. By screening on RT-PCR with canine AstV-specific primers 625F-1 and 626R-1 (Table 1) in a collection of fecal samples obtained from 1- to 6-month-old pups with gastroenteric signs in Italy in 2007, AstV RNA was detected in 27 of 110 samples (24.5%), either alone (10 of 110, 9.0%) or in mixed infections with canine parvovirus type 2 (5 of 110, 4.5%) and canine coronavirus (8 of 110, 7.3%) or both (4 of 110, 3.6%). Also, AstV RNA was detected in 7 of 75 swabs (9.3%) obtained from asymptomatic young dogs.7
Table 1.
List of primers used for diagnostic of canine AstVs
| Primer | Sequence (5′ to 3′) | Sense | Target | Reference | Assay |
|---|---|---|---|---|---|
| CaAstV2Pb | 6FAM-ATATGTACTTTTGCCATCAGGAGAG-BHQ1 | + | ORF1b | Unpublished | qRT-PCR |
| CaAstV2-F | ATTACCACGATGTTGYTCTGTR | + | ORF1b | Unpublished | qRT-PCR |
| CaAstV2-R | CATGATTGGTATGTTGAAAAYYTG | − | ORF1b | Unpublished | qRT-PCR |
| CaAstV1Pb | 6FAM-TACTGTGCTACTTCCATCTGGCGAG-BHQ1 | + | ORF1b | Unpublished | qRT-PCR |
| CaAstV1-F | ATTACCACGATGTTGTTCTGTA | + | ORF1b | Unpublished | qRT-PCR |
| CaAstV1-R | CATGATTGGTATGTTGAAAATCTG | − | ORF1b | Unpublished | qRT-PCR |
| panAstVFor1 | GARTTYGATTGGRCKCGKTAYGA | + | ORF1b | (19) | RT-PCR |
| panAstVFor2 | GARTTYGATTGGRCKAGGTAYGA | + | ORF1b | (19) | RT-PCR |
| panAstVRev | GGYTTKACCCACATNCCRAA | − | ORF1b | (19) | RT-PCR |
| 625F-1 | GTACTATACCRTCTGATTTAATT | + | ORF1b | (7) | RT-PCR |
| 626R-1 | AGACCAARGTGTCATAGTTCAG | − | ORF1b | (7) | RT-PCR |
| L5 | CAANTCACAACCCAAAACAAA | + | ORF2 | (8) | RT-PCR |
| R5 | TTTTNACNATCACTGCTAGNG | − | ORF2 | (8) | RT-PCR |
Unpublished data by Martella and colleagues, 2011.
In a survey in Shangai, China, in 2008, using canine AstV-specific primers L5 and R5 (Table 1), AstVs have been detected on RT-PCR in 22 of 183 (12.02%) dogs with enteric signs but in 0 of 138 asymptomatic dogs (0%). The 3′ end portion of the genome of a Chinese virus was sequenced and found to be distantly related (76.9% to 78.3% nucleotide [nt] identity in the full-length ORF2) to other canine AstVs, providing evidence for genetic heterogeneity in canine AstVs.8
Antibodies specific for canine AstV (virus Bari/08/ITA) have been detected by an indirect immunofluorescence assay in 32 of 54 (59.0%) serum samples. The majority (14 of 22, 63.6%) of the serum samples testing negative were from pups aged less than 3 months, while only 3 of 32 (9.4%) of the positive sera were from dogs aged less than 3 months.7 This age-related pattern would be consistent with the fact that by 2 to 3 months of age, pups is susceptible to infectious agents as maternally derived immunity tends to wane.29
Cultivation in Vitro
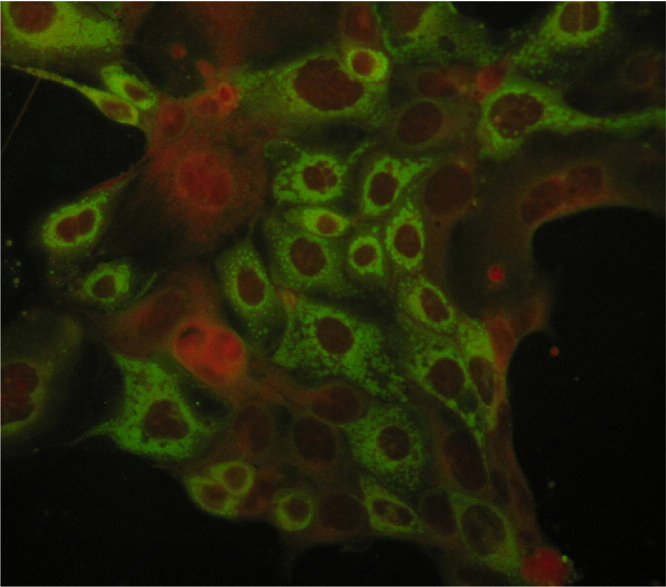
A canine AstV, strain Bari/08/ITA, has been isolated in a canine kidney cell line (Madin-Darby canine kidney [MDCK]). Likewise other mammalian AstVs, cultivation required supplementation with trypsin.7 Virus replication in MDCK cells triggered a clear cytopathic effect consisting in enlargement and/or detaching of cells and appearance of fine granules in the cytoplasm. Viral antigens were observed in immunefluorescence in the cells as fine granules dispersed in the cytoplasm, aggregating in perinuclear position (Fig. 1). No significant mutation was mapped in the ORF2 and 3′ noncoding region (3′ -UTR) between the field virus and the tissue-adapted (third serial passage) isolate Bari/08/ITA.
Fig. 1.
Indirect immunofluorescence on 24-hour infected MDCK cells.
Genome Organization of Canine Astroviruses
The full-length genome sequence of canine AstVs has not been determined yet, while the sequence of the 3′ end of the genome (the 3′ end of ORF1b, the full-length ORF2, and the 3′-UTR) is available for the isolate ITA/2008/Bari and for strain ITA/2010/Zoid. In addition, the full-length ORF2 sequences of an additional 6 canine strains from Italy and China are available in the databases. In these strains, ORF2 ranges from 765 to 774 amino acids in length, while the 3′-UTR ranges from 77 to 87 nt. There is an 8-nt overlap between the termination codon of ORF1b and the initiation codon of ORF2. The highly conserved nucleotide stretch upstream of ORF2, 5′-ATTTGGAGNGGNGGACCNAAN5–8ATGNC-3′, believed to be part of a promoter region for synthesis of subgenomic RNA,30 is nearly completely conserved in the sequence of strains ITA/2008/Bari and ITA/2010/Zoid. Overall, these features mirror those observed in other mammalian AstVs.1
By comparison of the capsid protein, the canine AstV strains exhibit more than 70.% amino acid identity to each other and less than 33% amino acid identity to noncanine AstVs, thus constituting a genetically well-defined group (Fig. 2). The range of variation among canine AstVs is similar to those defined for human GI.A AstV types 1 to 8.31 By analyzing the capsid protein of human AstVs, a high degree of conservation can be observed in the N-terminal portion (amino acids 1 to 415) of the capsid protein, while downstream the conserved 415 residues, considerable variability is seen among strains of different serotypes.1 Likewise, among the canine AstV strains, a high degree of variation occurs between aa 422 and 668 (<50% aa identity), whilst the capsid sequence is more conserved at the NH3- and COOH-ends. The hypervariable region is believed to form the spikes of the virion and to interact with the cell receptors32 as neutralizing monoclonal antibodies have been mapped to this variable domain.33, 34 These findings may be predictive of marked antigenic differences among the various canine strains.
Fig. 2.
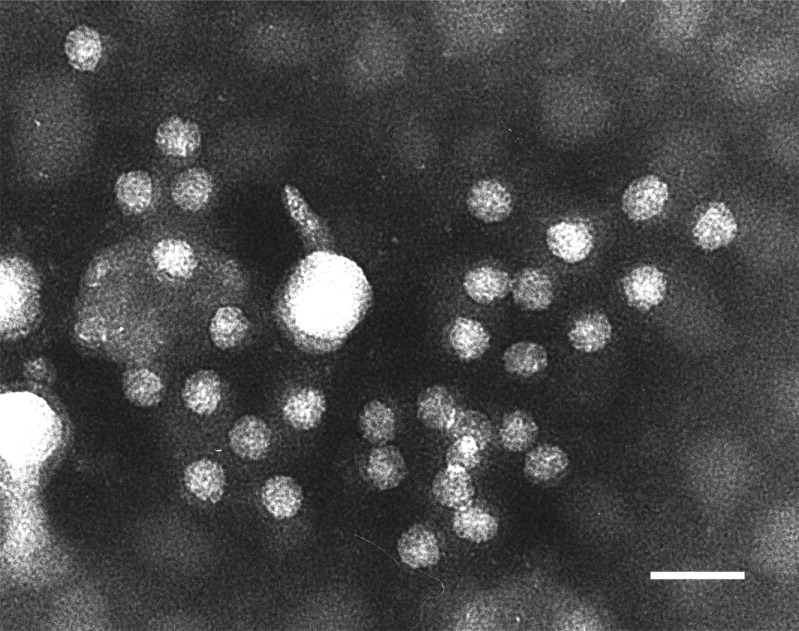
EM observation of negatively stained AstV particles, aggregated by an AstV-specific antiserum. Scale bar 50 nm.
(Courtesy of Dr Antonio Lavazza, Istituto Zooprofilattico Sperimentale di Lombardia ed Emilia Romagna, Sezione di Brescia, Italy.)
Pathogenesis
Using specific primers in RT-PCR, canine AstVs RNA has been detected exclusively in the stool and in the intestinal content of pups and no evidence for extraintestinal localization has been obtained thus far screening the tissues of animals dead from severe gastroenteritis.
AstV shedding was detectable in RT-PCR for at least 10 days in a 3-month-old pup hospitalized with watery diarrhea and severe dehydration. Clinical signs in the pup lasted 4 days, with viral shedding continuing for 8 days after the pup recovered from the disease. The pup tested negative to all other canine pathogens, suggesting that AstV was actually the causative agent of the disease.7 By quantification of viral load in real time RT-PCR (qRT-PCR), a positive correlation between the clinical signs and the virus titers was observed with the highest viral loads occurring during the acute symptomatic phase. Also, specific IgGs were not detected in the serum sample collected at the time of hospitalization, but they were detectable 2 weeks after hospitalization, indicating seroconversion.7 Interesting pieces of information have been gathered by monitoring an outbreak of gastroenteric disease associated with canine AstV, affecting 2 household dogs (aged 2 years and 2 months, respectively). The AstV strain, ITA/2010/Zoid, displayed limited amino acid identity (70.3% to 73.9%) to other canine AstV strains in the full-length capsid protein. Antibodies specific for the prototype canine astrovirus isolate ITA/2008/Bari were not detected in the convalescent sera of the animals, suggesting limited antigenic relatedness. In the 2 animals, virus shedding (up to 107 to 108 copies of genome equivalents/gr of feces) was correlated with clinical signs, with the disease being severer and virus shedding being more prolonged (more than 1 month) in the young pup (Martella and colleagues, unpublished information, 2011). Prolonged virus shedding after acute infection and resistance in the environments35, 36 could be factors facilitating virus diffusion in susceptible population.
Diagnostics
EM observation has long been the only assay able to reveal the presence of SRVs in the stools of dogs (Fig. 3). The low prevalence rates on EM, compared to the high rates revealed in molecular assays, suggest AstV titers in the feces are above the EM detection limits for a restricted time period.7 Also, EM is not always able to characterize firmly SRVs, as the peculiar morphology of AstVs is often lost during sample conservation and/or processing.9 Several primer sets have been developed to recognize effectively canine AstVs in RT-PCR (Table 1). The primers have been designed in highly conserved regions of the ORF1b (RdRp) and ORF2 (capsid protein). Also, a quantitative RT-PCR (qRT-PCR) has been set up (Table 1). The qRT-PCR assay was able to detect AstV strains that markedly differed in the capsid gene and proved to be highly sensitive (Martella and colleagues, unpublished data, 2011). Using the strain Bari/08/ITA has antigen, an indirect immunofluorescence assay has been developed to identify canine AstV-specific IgGs.7
Fig. 3.
Phylogenetic tree based on the full-length amino acid sequence of the capsid protein of AstVs of avian and mammalian origin. The tree was elaborated with the neighbor-joining method without any distance correction.
Summary
Based on the present, yet limited, literature, canine AstVs appear to be widespread in different geographical areas. AstV prevalence seems to be significantly higher in pups with gastroenteric disease than in asymptomatic animals. In some cases AstVs can be detected alone (not in coinfection with other enteric pathogens) from animals with enteric disease and virus shedding can be correlated with the onset of gastroenteric signs. Interestingly, canine AstVs appear to be genetically heterogeneous. Animal experiments will be pivotal to investigate the pathogenic role of canine AstVs and understand whether prophylaxis tools are required. Also, the canine homologous model could offer valuable information for the study of human AstVs.
Footnotes
This work was granted by the project from the Italian Ministry of Health, Ricerca corrente 2009, project IZS VE 21/09 RC “Definizione di una procedura validata per la selezione di cani per programmi di Interventi Assistiti dagli Animali (IAA)”.
The authors have nothing to disclose.
References
- 1.Mendez E., Arias C.F. In: Fields Virology. 5th ed. Knipe D.M., Howley P.M., editors. Lippincott Willliams & Wilkins; Philadelphia: 2007. Astroviruses; pp. 981–1000. [Google Scholar]
- 2.Imada T., Yamaguchi S., Mase M., et al. Avian nephritis virus (ANV) as a new member of the family Astroviridae and construction of infectious ANV cDNA. J Virol. 2000;74:8487–8493. doi: 10.1128/jvi.74.18.8487-8493.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blomström A.-L., Widén F., Hammer A.-S., et al. Detection of a novel astrovirus in brain tissue of mink suffering from shaking mink syndrome by use of viral metagenomics. J Clin Microbiol. 2010;48:4392–4396. doi: 10.1128/JCM.01040-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Quan P.L., Wagner T.A., Briese T., et al. Astrovirus encephalitis in boy with X-linked agammaglobulinemia. Emerging Infect Dis. 2010;16:918–925. doi: 10.3201/eid1606.091536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Williams F.P., Jr Astrovirus-like, coronavirus-like, and parvovirus-like particles detected in the diarrheal stools of beagle pups. Arch Virol. 1980;66:215–226. doi: 10.1007/BF01314735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Toffan A., Jonassen C.M., De Battisti C., et al. Genetic characterization of a new astrovirus detected in dogs suffering from diarrhoea. Vet Microbiol. 2009;139:147–152. doi: 10.1016/j.vetmic.2009.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Martella V., Moschidou P., Lorusso E., et al. Detection and characterization of astroviruses in dogs. J Gen Virol. 2011;92(Pt 8):1880–1887. doi: 10.1099/vir.0.029025-0. [DOI] [PubMed] [Google Scholar]
- 8.Zhu A.L., Zhao W., Yin H., et al. Isolation and characterization of canine astrovirus in China. Arch Virol. 2011;156(9):1671–1675. doi: 10.1007/s00705-011-1022-z. [DOI] [PubMed] [Google Scholar]
- 9.Madeley C.R., Cosgrove B.P. Letter: 28 nm particles in faeces in infantile gastroenteritis. Lancet. 1975;2:451–452. doi: 10.1016/s0140-6736(75)90858-2. [DOI] [PubMed] [Google Scholar]
- 10.Bridger J.C. Detection by electron microscopy of caliciviruses, astroviruses and rotavirus-like particles in the faeces of piglets with diarrhoea. Vet Rec. 1980;107:532–533. [PubMed] [Google Scholar]
- 11.Englund L., Chriél M., Dietz H.H., et al. Astrovirus epidemiologically linked to pre-weaning diarrhoea in mink. Vet Microbiol. 2002;85:1–11. doi: 10.1016/s0378-1135(01)00472-2. [DOI] [PubMed] [Google Scholar]
- 12.Hoshino Y., Zimmer J.F., Moise N.S., et al. Detection of astroviruses in feces of a cat with diarrhea: Brief report. Arch Virol. 1981;70:373–376. doi: 10.1007/BF01320252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kjeldsberg E., Hem A. Detection of astroviruses in gut contents of nude and normal mice: Brief report. Arch Virol. 1985;84:135–140. doi: 10.1007/BF01310560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McNulty M.S., Curran W.L., McFerran J.B. Detection of astroviruses in turkey faeces by direct electron microscopy. Vet Rec. 1980;106:561. doi: 10.1136/vr.106.26.561. [DOI] [PubMed] [Google Scholar]
- 15.Snodgrass D.R., Gray E.W. Detection and transmission of 30 nm virus particles (astroviruses) in faeces of lambs with diarrhoea. Arch Virol. 1977;55:287–291. doi: 10.1007/BF01315050. [DOI] [PubMed] [Google Scholar]
- 16.Todd D., Smyth V.J., Ball N.W., et al. Identification of chicken enterovirus-like viruses, duck hepatitis virus type 2 and duck hepatitis virus type 3 as astroviruses. Avian Pathol. 2009;38(1):21–30. doi: 10.1080/03079450802632056. [DOI] [PubMed] [Google Scholar]
- 17.Tzipori S., Menzies J.D., Gray E.W. Detection of astrovirus in the faeces of red deer. Vet Rec. 1981;108(13):286. doi: 10.1136/vr.108.13.286. [DOI] [PubMed] [Google Scholar]
- 18.Woode G.N., Bridger J.C. Isolation of small viruses resembling astroviruses and caliciviruses from acute enteritis of calves. J Med Microbiol. 1978;11(4):441–452. doi: 10.1099/00222615-11-4-441. [DOI] [PubMed] [Google Scholar]
- 19.Chu D.K.W., Poon L.L.M., Guan Y., et al. Novel astroviruses in insectivorous bats. J Virol. 2008;82(18):9107–9114. doi: 10.1128/JVI.00857-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chu D.K.W., Chin A.W.H., Smith G.J., et al. Detection of novel astroviruses in urban brown rats and previously known astroviruses in humans. J Gen Virol. 2010;91(Pt 10):2457–2462. doi: 10.1099/vir.0.022764-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rivera R., Nollens H.H., Venn-Watson S., et al. Characterization of phylogenetically diverse astroviruses of marine mammals. J Gen Virol. 2010;91(Pt 1):166–173. doi: 10.1099/vir.0.015222-0. [DOI] [PubMed] [Google Scholar]
- 22.Finkbeiner S.R., Holtz L.R., Jiang Y., et al. Human stool contains a previously unrecognized diversity of novel astroviruses. Virol J. 2009;6:161. doi: 10.1186/1743-422X-6-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bányai K., Meleg E., Moschidou P., et al. Detection of newly described astrovirus MLB1 in stool samples from children. Emerg Infect Dis. 2010;16(1):169. doi: 10.3201/eid1601.091120. author reply 169–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Finkbeiner S.R., Le B.M., Holtz L.R., et al. Detection of newly described astrovirus MLB1 in stool samples from children. Emerg Infect Dis. 2009;15(3):441–444. doi: 10.3201/1503.081213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kapoor A., Li L., Victoria J., et al. Multiple novel astrovirus species in human stool. J Gen Virol. 2009;90(Pt 12):2965–2972. doi: 10.1099/vir.0.014449-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Burbelo P.D., Ching K.H., Esper F., et al. Serological studies confirm the novel astrovirus HMOAstV-C as a highly prevalent human infectious agent. PLoS ONE. 2011;6(8):e22576. doi: 10.1371/journal.pone.0022576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Marshall J.A., Healey D.S., Studdert M.J., et al. Viruses and virus-like particles in the faeces of dogs with and without diarrhoea. Aust Vet J. 1984;61(2):33–38. doi: 10.1111/j.1751-0813.1984.tb07186.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vieler E., Herbst W. [Electron microscopic demonstration of viruses in feces of dogs with diarrhea] Tierarztl Prax. 1995;23(1):66–69. [PubMed] [Google Scholar]
- 29.Pollock R.V., Carmichael L.E. Maternally derived immunity to canine parvovirus infection: transfer, decline, and interference with vaccination. J Am Vet Med Assoc. 1982;180(1):37–42. [PubMed] [Google Scholar]
- 30.Walter J.E., Briggs J., Guerrero M.L., et al. Molecular characterization of a novel recombinant strain of human astrovirus associated with gastroenteritis in children. Arch Virol. 2001;146(12):2357–2367. doi: 10.1007/s007050170008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Méndez-Toss M., Romero-Guido P., Munguía M.E., et al. Molecular analysis of a serotype 8 human astrovirus genome. J Gen Virol. 2000;81(Pt 12):2891–2897. doi: 10.1099/0022-1317-81-12-2891. [DOI] [PubMed] [Google Scholar]
- 32.Krishna N.K. Identification of structural domains involved in astrovirus capsid biology. Viral Immunol. 2005;18(1):17–26. doi: 10.1089/vim.2005.18.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bass D.M., Upadhyayula U. Characterization of human serotype 1 astrovirus-neutralizing epitopes. J Virol. 1997;71(11):8666–8671. doi: 10.1128/jvi.71.11.8666-8671.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sanchez-Fauquier A., Carrascosa A.L., Carrascosa J.L., et al. Characterization of a human astrovirus serotype 2 structural protein (VP26) that contains an epitope involved in virus neutralization. Virology. 1994;201(2):312–320. doi: 10.1006/viro.1994.1296. [DOI] [PubMed] [Google Scholar]
- 35.Abad F.X., Villena C., Guix S., et al. Potential role of fomites in the vehicular transmission of human astroviruses. Appl Environ Microbiol. 2001;67(9):3904–3907. doi: 10.1128/AEM.67.9.3904-3907.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Abad F.X., Pintó R.M., Villena C., et al. Astrovirus survival in drinking water. Appl Environ Microbiol. 1997;63(8):3119–3122. doi: 10.1128/aem.63.8.3119-3122.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]