Summary
Institutional and health-care system approaches complement bedside strategies to improve care of the critically ill. Focusing on the USA and the UK, we discuss seven approaches: education (especially of non-clinical managers, policy-makers, and the public), organisational guidelines, performance reporting, financial and sociobehavioural incentives to health-care professionals and institutions, regulation, legal requirements, and health-care system reorganisation. No single action is likely to have sustained effect and we recommend a combination of approaches. Several recent initiatives that hold promise tie performance reporting to financial incentives. Though performance reporting has been hampered by concerns over cost and accuracy, it remains an essential component and we recommend continued effort in this area. We also recommend more public education and use of organisational guidelines, such as admission criteria and staffing levels in intensive care units. Even if these endeavours are successful, with rising demand for services and continuing pressure to control costs, optimum care of the critically ill will not be realised without a fundamental reorganisation of services. In both the USA and UK, we recommend exploration of regionalised care, akin to US state trauma systems, and greater use of physician-extenders, such as nurse practitioners, to provide enhanced access to specialist care for critical illness.
This article is the last in a series of five focusing on improving care and safety of the critically ill. Previous articles introduced the concepts of critical illness, quality of care, and patients' safety1 and reviewed approaches to identify patients' safety concerns,2 measure quality of care,3 and improve care through modification of caregiver behaviour.4 Three themes emerged: care of the critically ill is fraught with potential hazards for patients; errors of omission (ie, failure to provide contemporary, evidence-based care) might be a greater threat to patients' safety than errors of commission; and improvement of care needs the appropriate cultural milieu, a system-wide commitment, active engagement of all relevant stakeholders, and constant measurement and feedback.
Rationale for approaches at institutional and health-care system levels
Implicit in the third theme is the notion that improvement of care might be difficult if left entirely to the goodwill and enthusiasm of the bedside clinician. This is because a clinician might recognise a problem but be unable to fix it without the support of his or her institution, whereas at other times the problem might be invisible to the clinician (eg, if a patient who should have been transferred to an intensive care unit [ICU] dies before transfer, the ICU clinician will not know). Additionally, clinicians are usually unaware of the extent to which the overall provision of resources is adequate for a population.
The relevance of approaches at the institutional and health-care system levels is even clearer when the effect of factors at these levels is considered. There is increasing evidence that the way critical care is organised and delivered has a great effect on outcomes.5, 6, 7 The capability of hospital wards to handle sick patients will determine transfer to and from the ICU. Financial pressures faced by the hospital will affect the level of provision of ICU services. Also, the prevailing professional culture can affect who is admitted to the ICU, how they are treated, and how the ICU is staffed.
Of course, care for the critically ill extends beyond the ICU (figure ) and changes at any point in the chain of care can have important consequences. For example, ICU “outreach” teams have the potential to improve care through earlier detection and intervention of acutely deteriorating patients on the wards.4, 8, 9 Yet, the launch, maintenance, and success of an ICU outreach programme is dependent on such institutional factors as the hospital budget, the perceived value of the programme, the level of co-operation between ICU and ward staff, and the ability to measure improvements in outcome.
Figure.
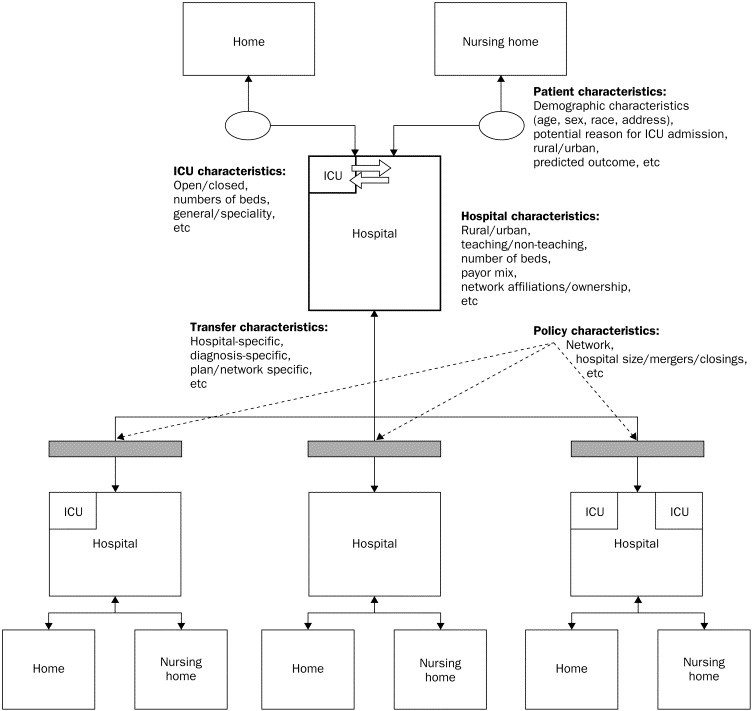
The chain of care for the critically ill
The care for a critically ill patient extends beyond the specific ICU that takes care of her. It is a chain of actions stretching from the location where the critical illness first occurred (eg, home) to the location where the critical illness has resolved. Reproduced with permission from Mark S Roberts, University of Pittsburgh, USA.
Characteristics of the health-care system are also important. These might operate at a regional level: local competition from other hospitals might lead hospital managers to build or upgrade ICUs in an effort to market technological superiority; swings in the strength of the local economy and labour market might affect staff recruitment and retention; and local news stories can sway public opinion, engage local politicians, and pressure hospital managers to change the provision of services. System influences can also originate from a national level. For example, the availability of adequately trained staff is affected by the number of training programmes the government chooses to fund and the training, accreditation, and certification standards developed by national professional associations.
In this article, we focus on approaches at the institutional and health-care system levels that can complement the bedside strategies to improve care presented in an earlier article in this Lancet series.4 Throughout, we will draw upon examples from two countries, the USA and the UK, which together illustrate a wide range of health-care organisational models. For clarity, we split these approaches into seven areas, summarised in the table . However, each works better in combination. For example, a performance report can stimulate an educational initiative which, to be effective, might need a supplementary incentive, such as its incorporation in an accreditation process.
Table.
Institutional and health-care system approaches to improving care of the critically ill
| Example | Advantages | Disadvantages | |
|---|---|---|---|
| Approach | |||
| Education | Clinical education for managers and policy-makers | Inexpensive; improve relationship between clinician and lay manager | Difficulty for managers to find time; lack of rigorous evidence of benefit |
| Public education (informed patients/lay carers) | Explicit, transparent; aids performance assessment and benchmarking | Lack of interest until health care needed | |
| Guidelines | Organisational guidelines (admission, discharge, staffing levels etc) | Inexpensive; explicit and transparent; take context into account | Difficulty achieving consensus; limited scientific evidence available; may encounter clinician resistance |
| Performance reporting | Risk-adjusted outcomes and processes (eg, CHAI in UK) | Objective; allows meaningful comparisons of providers | Uncertainty as to adequacy of adjustment |
| Public disclosure of information | Empowers public; increases accountability to public | Limited impact; little patients' choice feasible for acute illnesses | |
| Financial and sociobehavioural incentives | Reward based on outcomes | Providers have little option but to respond to financial incentives | Dependent on accurate, risk-adjusted measures; may encourage gaming such as patient selection; financial instability |
| Disclosure of performance to peers | Clinicians want to be seen by peers to be providing good-quality care | “Bad apples” may not be bothered by peers' views | |
| Regulation | Accreditation (eg, JCAHO in US) | Relatively easy as focused on inputs or structural factors | Lack of association between inputs and outcomes; lack of evidence of effectiveness |
| Inspection (eg, CHAI) | Allows in-depth assessment of structures, processes and outcomes Acts as strong incentive for clinicians and providers to use guidelines | Expensive; may damage staff morale if seen as unfair May encourage defensive medicine; huge additional financial cost to health care providers | |
| Legal requirements | Litigation | Establishes direct link between poor quality care and consequences | May encourage defensive medicine; little evidence of effect on quality of care; expensive for health system |
| Reorganisation of service delivery | Staff substitution | Increase efficiency, staff satisfaction, and morale | Resistance from professions who feel threatened |
| Increased availability and flexibility of services | Strong support from clinicians; no requirement for behaviour change by clinicians | Expensive; complexity of management of flexible services; supplier-induced demand | |
| Regionalisation by levels of care | Consistent with future staffing restrictions (eg, working hours, training) | Resistance from clinicians providing low-level care; more patients transferred |
CHAI=Commission for Healthcare Audit and Inspection. JCAHO=Joint Commission on Accreditation of Healthcare Organizations.
Education
Education to modify clinician behaviour, and thereby improve the quality of care, is important.4 However, clinicians generally know what constitutes good care and a knowledge deficit is rarely the sole cause of poor quality care.10 By contrast, some key non-clinical players (hospital managers, purchasers, and payers) often have little knowledge of how best a service should be provided. One might expect improved care if other staff, such as managers, were better informed about key clinical issues. Clinical education programmes for non-clinical staff have been started in the UK but we are unaware of any formal evaluation of such initiatives. Similarly, there are no national standards or requirements for clinical education of non-clinical staff in the USA. Within the health-care system, it is hard to imagine any large-scale change if the relevant managers are not appropriately appraised of the current situation.
Outwith the health-care system, there are two reasons to think that better education of society (in particular, patients, the media, and politicians) regarding critical illness and care options might also enhance care. First, patients and families are integral to the care process and may contribute more if better educated. For example, the morbidity and mortality of meningococcal septicaemia in children might be reduced if parents have greater awareness of the need for prompt admission to hospital and aggressive resuscitation.11, 12, 13 Similarly, a public well-educated about the severe acute respiratory syndrome (SARS) might comply better with preventive strategies and quarantine procedures.14, 15
The second reason relates to the health-care market. A major thrust of health-care policy in recent years, both in the USA and the UK, has been to promote greater competition both among providers (clinicians and hospitals) and among purchasers. The premise is that a more competitive marketplace provides better quality care at lower cost. However, “efficient” markets require educated consumers.16 If the consumer (patients, or the purchaser as their agent) cannot distinguish between a high and low quality product, then they cannot choose wisely.
There are many examples of patient and public education initiatives, including leaflets, books, TV programmes, and magazines.17 The internet has also led to some promising innovations. Several non-governmental patient education foundations and support groups exist in the USA and the UK. However, most of these focus on chronic illnesses, such as diabetes or cystic fibrosis. There are exceptions: in the UK, the Directory of Patients' Experiences (DIPEX) (www.dipex.org), which allows patients and their lay caregivers access to other people who have faced the same problems and decisions, plans to include critical care.18 In the USA, there are support groups for survivors of acute illnesses, such as the ARDS (acute respiratory distress syndrome) Foundation (www.ardsusa.org).
A major impediment to public education is that stimulating interest in critical illness is difficult. When healthy, we tend to avoid thoughts of disease, when critically ill, we are not in a position to learn about the disease and if we recover we often cannot, or do not want to, remember the experience. As such, public awareness is often limited to sensationalised headlines, such as “flesh-eating bacteria force amputation of woman's arm”.19 Yet, public awareness and advocacy can be a potent tool for driving change.20 For this reason, the International Sepsis Forum, a coalition of researchers and pharmaceutical companies, has launched the Surviving Sepsis Campaign. This campaign is intended to increase public awareness about sepsis (www.survivingsepsis.org) using a variety of public education and media relations tools.
Guidelines
Organisational, rather than clinical, guidelines for such issues as referral within and between hospitals, staffing levels, and management arrangements are another way to improve care. Guidelines should help to develop standardisation of the capabilities of an ICU. For example, the transition into and out of the ICU needs to be better delineated both to ensure efficient use of resources and to provide a better continuum of care. In the UK, the National Service Frameworks, focused either on a diagnostic or a demographic group, are a form of organisational guideline.21 The national review of adult critical care services22 (partly prompted by adverse media coverage) provide clear guidance, based on best available research evidence and professional opinion, on how services should be modernised. Endorsement of the recommendations by government was associated with a major increase in funding for critical care.
Performance reporting
The mechanisms for collection, analysis, and reporting of data for processes and outcome of care to clinicians and institutions were discussed in two preceding articles.2, 3 Such data have also been reported to provider managers, the public, purchasers, and payers. Although the public disclosure of performance seems to result in improved outcomes,23 the mechanism of action is unclear.24 Provider managers seem to respond with internal changes, especially those operating in a competitive environment. Despite this, clinicians remain sceptical and consider such disclosure has little use.24
In England and Wales, the Audit Commission relied heavily on risk-adjusted outcome data from the Intensive Care National Audit and Research Centre (ICNARC) Case Mix Programme when undertaking a nationwide audit of critical care.25, 26 However, the report recognised that even more management information was needed. In the early 1990s, the Greater Cleveland Health Quality and Choice Commission, a local coalition of purchasers, mandated all ICUs to report risk-adjusted mortality. The data were to be used to drive purchasing decisions and, through their public reporting, stimulate improvements in health-care quality.27 Although there might have been an improvement in outcome over time, it was unclear whether this was driven by the data collection and reporting,28 and after 4 years the programme was shut down because of cost, lack of clear benefit, and reluctance of some institutions to continue participating.
Despite these problems, efforts to collect and report ICU outcomes in many countries are continuing. In the USA, the Joint Commission of Accreditation Healthcare Organizations (JCAHO) is considering making collection of risk-adjusted outcome data mandatory for all hospitals wishing to participate in the Medicare programme, although how this information will be used to deny or approve accreditation is not yet clear. Similar developments in the UK are being led by the Commission for Healthcare Audit and Inspection (CHAI). The Commission currently depends on a largely administrative database, Hospital Episode Statistics, which is often incomplete, inaccurate, and does not permit adequate risk adjustment. However, the development of specialised clinical databases offers an alternative and their use has been facilitated by the creation of a national Directory of Clinical Databases (www.docdat.org).29
Currently, ICU reporting relies on specialist risk adjustment tools but their application is limited to care within the ICU. The complexity of extended critical care by multiple providers makes outcomes difficult to interpret and appropriate changes difficult to target. Despite these challenges, outcome assessment is potentially useful in identifying areas of concern. Together with monitoring specific processes, a more complete picture can be built up to aid clinicians and managers to improve patients' care and safety.
Financial and sociobehavioural incentives
Incentives can either be financial or sociobehavioural (appealing to professionals' concerns about peers' views of their competence and performance). Incentives might operate through recognition and reward for good performance (positive) or by penalising poor performance (negative). Although there are nearly always a large number of negative and positive incentives surrounding any decision or course of action, the use of deliberate incentive schemes in the UK to drive a particular behaviour is uncommon and often controversial. The Department of Health's use of a star-rating system for hospitals in England has largely been based on process and outcome indicators derived from data of doubtful quality.30 It takes no account of the heterogeneity of the performance of different units or departments within a hospital—an excellent ICU could be nested in a hospital of generally low quality.31
Health care in the USA has been much more closely associated with financial incentives. The way in which physicians have responded to the fee-for-service insurance model that prevailed for the majority of the second half of the 20th century has been the focus of much debate. One argument is that this model drives physicians to provide more services, whether they are needed or not, creating a supply-driven market characterised by overuse of services and rising health-care costs.32, 33, 34, 35 Others suggest that physicians do not respond solely to financial incentives and will modify their behaviour on the basis of their patients' interests.36 This is in contrast, for example, to a car salesman, who is not expected to prioritise his customers' needs over his own.
One solution would be to provide financial rewards for better, rather than more, service. If measures of quality of care were deemed sufficiently valid and reliable, tying them to financial incentives (and disincentives) is likely be a powerful, though controversial, modifier of physician and institutional behaviour. The financial incentives could be administered by a single payer, such as a national government, thus offering an alternative to policymakers who want to promote better care without creating a true free market. Under the US Medicare Prescription Drug, Improvement, and Modernization Act of 2003, the Centers for Medicare and Medicaid Services will assess such an approach through demonstration projects of alternative financial incentives designed to promote improved care and safety.
The Leapfrog group (www.leapfroggroup.org), a consortium of US Fortune 500 companies who purchase health care for 34 million employees and their families, also hopes financial incentives can improve quality of care and safety and reduce unnecessary health-care expenditures.37, 38 In particular, the group explicitly intends to “alert the health-care industry that big leaps in patient safety and customer value will be recognized and rewarded with preferential use and other intensified market reinforcements” by their member companies (www.leapfroggroup.org/about.htm). One of their first initiatives is to pressure hospitals to ensure that all ICU patients are cared for by intensive care specialist physicians (intensivists) with appropriate 24 h coverage. Despite numerous studies suggesting better outcome with intensivist-directed care,5 less than 10% of ICUs have such staffing.39, 40 Leapfrog has embarked on regional “roll-outs”, and claims that 21% of the ICUs in these regions have now adopted the intensivist staffing model, and another 5% will do so in the coming year.41
Regulation
Regulation, both voluntary and compulsory, is increasingly popular with governments and other payers. Types of regulation include hospital and physician credentialling, professional licensure, specialty certification and re-certification, and certificates of need. In the USA, the largest hospital regulator is JCAHO, which undertakes regular audits of all hospitals that wish to be considered eligible for the provision of Medicare services. The incentive is clear—if a hospital fails to maintain JCAHO accreditation, it cannot bill for Medicare services. In addition to considering collection of risk-adjusted outcome data, JCAHO has proposed other ICU quality measures, including catheter-related infection rates, ICU physician staffing patterns, and compliance with several evidence-based ICU processes of care, such as ventilator weaning protocols.42 This level of detail, measuring actual patients' care processes and outcomes, is part of a broader initiative by JCAHO to develop and implement rigorous quality assessments. In view of the importance of JCAHO accreditation, the use of these measures could have profound, and hopefully beneficial, effects on ICU care in the US. However, the measures are yet to be enforced, there is controversy over their interpretation, and some will be burdensome to collect.
In the UK, CHAI inspects every hospital at least once every 3 years. The inspection includes consideration of patient outcomes, including evidence of poor safety. The adoption of such an approach has been prompted by the findings of a major public inquiry into the performance of the cardiac surgical service at the Bristol Royal Infirmary.43
Legal requirements
Hospitals are subject to numerous legal requirements with respect to employment, health and safety, fire prevention and control, and laws to protect individual patients from harm. One notable example of US legislation that greatly affected care of critically ill patients was the 1986 Emergency Medical Treatment and Active Labor Act (EMTALA). Before 1986, hospitals did not have to treat all patients who sought care. Thus, a hospital might refuse to provide care to a sick patient if he was likely to consume considerable resources and had no ability to pay. Under EMTALA, such actions became illegal: EMTALA obligated all Medicare-participating hospitals with emergency departments to provide initial care and stabilisation followed by hospital admission or transfer as appropriate to all critically ill persons arriving at their door.
Perhaps the most visible intersection between quality of care and the law is medical malpractice, especially in the USA. As articulated in earlier articles in this series, medical error mainly arises from problems of the system, not the individual.44 Thus, a culture of blame should be avoided in favour of promotion of open disclosure, and greater teamwork and collaboration. The courtroom, which is explicitly about assigning blame, would seem to have little role in this framework. Yet, an interesting phenomenon of medical malpractice in the USA has been the evolving definition of a professional standard of care. Over the last two centuries, the courts have moved from a definition that was locally-defined and based on subjective expert testimony to one that is nationally representative and grounded in strong scientific evidence.45 As such, the courts increasingly represent a venue in which, theoretically, best evidence-based medicine could be promoted and enforced, and some have argued that legal deterrence is essential in the promotion of health-care quality and patient safety.45
However, using legal deterrence as a cornerstone of health-care improvement could be ineffective and unpalatable, and might even encourage poor quality care.46, 47 Thus far, there is little evidence of litigation having either a positive or negative effect on health-care quality.46, 48, 49 In the USA, only few individuals affected by malpractice claim compensation,50 settlements are disproportionate and inequitable,51, 52 and the system is very expensive.53 Reform, therefore, seems essential if this powerful tool is to be used effectively to improve care. There are numerous proposed solutions, ranging from caps on payment for damages, to a complete overhaul and adoption of a no-fault system, where compensation for injury is not dependent on individual blame.47 But no reform is under any serious consideration in the USA. Although the UK does not face a similar “tort crisis”, there is increasing concern that errors are prolific in UK hospitals, and that the current legal system does not help, and might worsen, error reduction. Therefore, the government has proposed separation of systems designed to achieve accountability for adverse events from those designed to compensate patients.54
Reorganisation
There are several ways in which improvements in care and safety can be achieved through changing the way services are organised and delivered, either at the level of the institution or by changes to the entire health-care system. First, the responsibility for clinical processes can be shifted between professions, most commonly involving nurses taking on tasks traditionally performed by doctors. At present, the lack of high quality research on the effect of such changes in the care of the critically ill patient precludes clear policy advice.55 Responsibilities can also be transferred by introducing new categories of staff. Physician assistants, developed in the 1960s for primary care, now work in secondary care as well.56 Although their potential role in care of the critically ill is unclear at present, substitution of one profession for another offers another potential approach to improving patients' safety.57
Second, changes can be made in the availability of care, in particular the balance of services (eg, ratio of number of intensive care to high dependency beds). Again, such operational planning is severely limited by our knowledge of the appropriate uses of different services. For that, we need to understand more about the costs and benefits of each option. For example, the clinical indications for stepping up or stepping down care and for inter-hospital transfers.
A third strategy is the redeployment of staff, such as the establishment of medical emergency teams to standardise and improve pre-ICU care.8, 9, 58 Another example is a post-ICU follow-up clinic for a more gradual “hand-off” of the sequelae of critical illnesses to the primary care providers.59, 60
A fourth strategy involves changing the relations between providers. This might range from informal collaborations and referral networks to regionalised care, involving a major reconfiguration of services. The rationale for regionalisation is that scarce, expensive resources can be consolidated in a regional centre where their use can be optimised and staff can gain the most concentrated training and expertise. There are two potential models for regionalisation: close small hospitals and consolidate services in a few, large institutions or create different levels of ICU on the basis of resources and expertise, such as those proposed recently by the American College of Critical Care,61 and triage patients such that the sickest patients are transferred to the highest level ICUs. The second model is more politically acceptable and has already been implemented in neonatal intensive care and trauma care with evidence of improved outcomes.62, 63, 64, 65, 66, 67, 68, 69, 70, 71 The British Paediatric Association has recommended a similar model for care of critically ill children (>1 month of age) in the UK, although their conclusions can be challenged for lacking a good evidence base.72, 73
There are barriers to regionalisation, however, and implementation is often incomplete. In the USA, 40% of all neonates who are ventilated and die do so without transfer to a tertiary care centre.74 In the UK, one attempt to regionalise trauma care was stymied by the reluctance of clinicians in hospitals providing the lowest intensity care to refer their complex “more interesting” patients to the regional centre.75 Furthermore, ICU patients are admitted from several sources (home, hospital ward, operating room, etc), which presents significant logistical problems for a regionalised system.
Despite such potential problems, greater regionalisation of services may be the most practical approach in the current environment of rising demand76 and scarce resources, to ensure care is best matched to patient need, thus optimising quality of care and improving patient safety.77 Such a strategy should be based on better estimates of the need for ICUs, as derived from appropriate epidemiological models.78 Implementation would need a broad patient classification system that determines need for ICU care,79, 80, 81, 82 and a common communications system that will know of all potential patients in time to triage their care. Newer technology, such as telemedicine, might also help by extending intensivist expertise.83 Irrespective of approach, regionalisation is unlikely to happen without the appropriate political will.
Conclusions
Much can be achieved in improving care of the critically ill through action at the institutional and healthcare system levels. Such measures will complement any bedside approaches. No individual action in isolation is likely to have much sustained effect. Instead, a package of measures is needed. All approaches have advantages and disadvantages, and these have to be weighed up before deciding on a package. The relative importance of the advantages and disadvantages will depend to some extent on the context. For example, if equity of use is highly valued it may not be politically feasible to pursue measures that increase efficiency. Yet, the opportunities for improvement are remarkably similar in the US and UK, despite the large differences in the underlying financing, organisation, and delivery of the countries' healthcare.
In view of the increasing need to be accountable not only to patients but also to payers and purchasers of health care, it is essential that meaningful measures of clinical performance are developed and implemented. Without these, it will prove impossible for clinicians and hospital managers to justify the resources they need now and in the future. Efforts to tie performance measures to financial incentives, such as the JCAHO efforts, hold promise. Greater public education and awareness will be important for generating advocacy and political will for change. Ultimately, however, significant and sustainable improvements in patients' care and safety will demand fairly large changes in the way professions and institutions inter-relate. In particular, greater use of regionalised care, akin to US state trauma systems, and greater use of physician-extenders, such as nurse practitioners, seems warranted. These changes are necessary not only to meet current demands on health care but also to meet the growing challenges of technological advances and pressures on creating and maintaining a high quality health-care workforce and delivery system.76, 77, 84
Search strategy
This article is not a systematic review, and does not report an exhaustive search of scientific publications. Rather, cited reports are intended as illustrative examples. We obtained these citations through directed MEDLINE searches, manual searches of our own files, informal critique and feedback from colleagues, and feedback from The Lancet peer review process.
Acknowledgments
Acknowledgments
D C Angus is supported by grants from the Agency for Healthcare Research and Quality, the National Institutes of Health (NHLBI, NIGMS), and the Robert Wood Johnson Foundation. The funding sources had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Conflict of interest statement
None declared.
References
- 1.Bion JF, Heffner JE. Challenges in the care of the acutely ill. Lancet. 2004;363:970–977. doi: 10.1016/S0140-6736(04)15793-0. [DOI] [PubMed] [Google Scholar]
- 2.Pronovost PJ, Nolan T, Zeger S, Miller M, Rubin H. How can clinicians measure safety and quality in acute care? Lancet. 2004;363:1061–1067. doi: 10.1016/S0140-6736(04)15843-1. [DOI] [PubMed] [Google Scholar]
- 3.Lilford RJ, Mohammed MA, Spiegelhalter D, Thomson R. Use and misuse of process and outcome data in managing performance of acute medical care: avoiding institutional stigma. Lancet. 2004;363:1147–1154. doi: 10.1016/S0140-6736(04)15901-1. [DOI] [PubMed] [Google Scholar]
- 4.Cook DJ, Montori VM, McMullin JP, Finfer SR, Rocker GM. Improving patients' safety locally: changing clinician behaviour. Lancet. 2004;363:1224–1230. doi: 10.1016/S0140-6736(04)15955-2. [DOI] [PubMed] [Google Scholar]
- 5.Pronovost PJ, Angus DC, Dorman T, Robinson KA, Dremsizov TT, Young TL. Physician staffing patterns and clinical outcomes in critically ill patients: a systematic review. JAMA. 2002;288:2151–2162. doi: 10.1001/jama.288.17.2151. [DOI] [PubMed] [Google Scholar]
- 6.Tarnow-Mordi WO, Hau C, Warden A, Shearer AJ. Hospital mortality in relation to staff workload: a 4-year study in an adult intensive-care unit. Lancet. 2000;356:185–189. doi: 10.1016/s0140-6736(00)02478-8. [DOI] [PubMed] [Google Scholar]
- 7.Goldfrad C, Rowan K. Consequences of discharges from intensive care at night. Lancet. 2000;355:1138–1142. doi: 10.1016/S0140-6736(00)02062-6. [DOI] [PubMed] [Google Scholar]
- 8.Bellomo R, Goldsmith D, Uchino S. A prospective before-and-after trial of a medical emergency team. Med J Aust. 2003;179:283–287. doi: 10.5694/j.1326-5377.2003.tb05548.x. [DOI] [PubMed] [Google Scholar]
- 9.Bellomo R, Goldsmith D, Uchino S. A prospective controlled trial of the effect of the medical emergency team on post-operative morbidity and mortality. Crit Care Med. 2004;32:916–921. doi: 10.1097/01.ccm.0000119428.02968.9e. [DOI] [PubMed] [Google Scholar]
- 10.Cabana MD, Rand CS, Powe NR. Why don't physicians follow clinical practice guidelines? A framework for improvement. JAMA. 1999;282:1458–1465. doi: 10.1001/jama.282.15.1458. [DOI] [PubMed] [Google Scholar]
- 11.Pollard AJ, Britto J, Nadel S, DeMunter C, Habibi P, Levin M. Emergency management of meningococcal disease. Arch Dis Child. 1999;80:290–296. doi: 10.1136/adc.80.3.290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Han YY, Carcillo JA, Dragotta MA. Early reversal of pediatric/neonatal septic shock by community physicians is associated with improved outcome. Pediatrics. 2003;112:793–799. doi: 10.1542/peds.112.4.793. [DOI] [PubMed] [Google Scholar]
- 13.Carcillo JA. Pediatric septic shock and multiple organ failure. Crit Care Clin. 2003;19:413–440. doi: 10.1016/s0749-0704(03)00013-7. [DOI] [PubMed] [Google Scholar]
- 14.Leung GM, Lam TH, Ho LM. The impact of community psychological responses on outbreak control for severe acute respiratory syndrome in Hong Kong. J Epidemiol Community Health. 2003;57:857–863. doi: 10.1136/jech.57.11.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tse MM, Pun SP, Benzie IF. Experiencing SARS: perspectives of the elderly residents and health care professionals in a Hong Kong nursing home. Geriatr Nurs. 2003;24:266–269. doi: 10.1016/S0197-4572(03)00251-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Arrow KJ. Uncertainty and the welfare economics of medical care. Am Econ Rev. 1963;53:941–973. [Google Scholar]
- 17.Coulter A. Partnerships with patients: the pros and cons of shared clinical decision-making. J Health Serv Res Policy. 1997;2:112–121. doi: 10.1177/135581969700200209. [DOI] [PubMed] [Google Scholar]
- 18.Herxheimer A, Ziebland S. DIPEx: fresh insights for medical practice. J R Soc Med. 2003;96:209–210. doi: 10.1258/jrsm.96.5.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Flesh-eating bacteria force amputation of woman's arm. The Associated Press. http://www.news8.net/news/stories/1203/114545.html (accessed Dec 19, 2003)
- 20.Domenighetti G, Luraschi P, Casabianca A. Effect of information campaign by the mass media on hysterectomy rates. Lancet. 1988;2:1470–1473. doi: 10.1016/s0140-6736(88)90943-9. [DOI] [PubMed] [Google Scholar]
- 21.National Service Frameworks http://www.doh.gov.uk/nsf/ (accessed June 26, 2003.)
- 22.UK Department of Health Comprehensive Critical Care. Review of adult critical care services. http://www.doh.gov.uk/compcritcare/compcritcarevw3.htm (accessed June 26, 2003).
- 23.Hannan EL. Measuring hospital outcomes: don't make perfect the enemy of good! J Health Serv Res Policy. 1998;3:67–69. doi: 10.1177/135581969800300202. [DOI] [PubMed] [Google Scholar]
- 24.Marshall MN, Shekelle PG, Leatherman S, Brook RH. The public release of performance data: What do we expect to gain? a review of the evidence. JAMA. 2000;283:1866–1874. doi: 10.1001/jama.283.14.1866. [DOI] [PubMed] [Google Scholar]
- 25.Audit Commission . Critical to success: the place of efficient and effective critical care services within the acute hospital. Audit Commission; London: 1999. [Google Scholar]
- 26.Rowan K, Black N. A bottom-up approach to performance indicators through clinician networks. Health Care UK. 2000;Spring:42–46. [Google Scholar]
- 27.Sirio CA, Angus DC, Rosenthal GE. Cleveland Health Quality Choice (CHQC): an ongoing collaborative, community-based outcomes assessment program. New Horiz. 1994;2:321–325. [PubMed] [Google Scholar]
- 28.Sirio CA, Shepardson LB, Rotondi AJ. Community-wide assessment of intensive care outcomes using a physiologically-based prognostic measure: implications for critical care delivery from Cleveland Health Quality Choice. Chest. 1999;115:793–801. doi: 10.1378/chest.115.3.793. [DOI] [PubMed] [Google Scholar]
- 29.Black N, Payne M. Directory of clinical databases: improving and promoting their use. Qual Saf Health Care. 2004;12:348–352. doi: 10.1136/qhc.12.5.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Department of Health . NHS Performance Ratings. Department of Health; London: 2002. [Google Scholar]
- 31.Rowan KR, Harrison D, Brady A, Black NA. Do ‘star ratings’ of hospitals reflect clinical outcomes? an ecological study. BMJ. (doi:10.1136/bmj.38007.694745.F7 published Jan 23, 2004). [DOI] [PMC free article] [PubMed]
- 32.Schroeder SA. Managing the U.S. health care workforce: creating policy amidst uncertainty. Inquiry. 1994;31:266–275. [PubMed] [Google Scholar]
- 33.McGuire TG, Pauly MV. Physician response to fee changes with multiple payers. J Health Econ. 1991;10:385–410. doi: 10.1016/0167-6296(91)90022-f. [DOI] [PubMed] [Google Scholar]
- 34.Evans RG. The supply and demand of health care. Hosp Trustee. 1978;2:6–7. [PubMed] [Google Scholar]
- 35.Evans RG. Supplier-induced demand: some empirical evidence and implications. In: Perlman M, editor. The economics of health and medical care. Wiley; New York: 1974. pp. 162–173. [Google Scholar]
- 36.Reinhardt UE. The economist's model of physician behavior. JAMA. 1999;281:462–465. doi: 10.1001/jama.281.5.462. [DOI] [PubMed] [Google Scholar]
- 37.Birkmeyer JD, Stukel TA, Siewers AE, Goodney PP, Wennberg DE, Lucas FL. Surgeon volume and operative mortality in the United States. N Engl J Med. 2003;349:2117–2127. doi: 10.1056/NEJMsa035205. [DOI] [PubMed] [Google Scholar]
- 38.Galvin R, Milstein A. Large employers' new strategies in health care. N Engl J Med. 2002;347:939–942. doi: 10.1056/NEJMsb012850. [DOI] [PubMed] [Google Scholar]
- 39.Groeger JS, Strosberg MA, Halpern NA. Descriptive analysis of critical care units in the United States. Crit Care Med. 1992;20:846–863. doi: 10.1097/00003246-199206000-00024. [DOI] [PubMed] [Google Scholar]
- 40.The Leapfrog Group Factsheet: ICU Physician staffing. http://www.leapfroggroup.org/FactSheets/ICU_FactSheet.pdf (accessed Mar 24, 2004)
- 41.The Leapfrog Group Factsheet: Leapfrog's regional roll-outs. http://www.leapfroggroup.org/factsheet/RRO_FactSheet.pdf (accessed Mar 24, 2004)
- 42.JCAHO National Patient Safety Goals. 2004. http://www.jcaho.org/accredited+organizations/patient+safety/04+npsg/index.htm (accessed Mar 24, 2004)
- 43.UK Department of Health . Learning from Bristol: The Department of Health's response to the report of the public inquiry into children's heart surgery at the Bristol Royal Infirmary 1984–1995. The Stationery Office; London, UK: 2002. [Google Scholar]
- 44.Committee on Quality of Health Care in America . To err is human: building a safer health system. Institute of Medicine; Washington DC: 1999. [Google Scholar]
- 45.Rosenbaum S. The impact of United States law on medicine as a profession. JAMA. 2003;289:1546–1556. doi: 10.1001/jama.289.12.1546. [DOI] [PubMed] [Google Scholar]
- 46.Black N. Medical litigation and the quality of care. Lancet. 1990;335:35–37. doi: 10.1016/0140-6736(90)90151-t. [DOI] [PubMed] [Google Scholar]
- 47.Brennan TA, Mello MM. Patient safety and medical malpractice: a case study. Ann Intern Med. 2003;139:267–273. doi: 10.7326/0003-4819-139-4-200308190-00009. [DOI] [PubMed] [Google Scholar]
- 48.Entman SS, Glass CA, Hickson GB, Githens PB, Whetten-Goldstein K, Sloan FA. The relationship between malpractice claims history and subsequent obstetric care. JAMA. 1994;272:1588–1591. [PubMed] [Google Scholar]
- 49.Sloan FA, Whetten-Goldstein K, Githens PB, Entman SS. Effects of the threat of medical malpractice litigation and other factors on birth outcomes. Med Care. 1995;33:700–714. doi: 10.1097/00005650-199507000-00006. [DOI] [PubMed] [Google Scholar]
- 50.Localio AR, Lawthers AG, Brennan TA. Relation between malpractice claims and adverse events due to negligence: results of the Harvard medical practice study III. N Engl J Med. 1991;325:245–251. doi: 10.1056/NEJM199107253250405. [DOI] [PubMed] [Google Scholar]
- 51.Brennan TA, Leape LL, Laird NM. Incidence of adverse events and negligence in hospitalized patients: results of the Harvard medical practice study I. N Engl J Med. 1991;324:370–376. doi: 10.1056/NEJM199102073240604. [DOI] [PubMed] [Google Scholar]
- 52.Brennan TA, Sox CM, Burstin HR. Relation between negligent adverse events and the outcomes of medical-malpractice litigation. N Engl J Med. 1996;335:1963–1967. doi: 10.1056/NEJM199612263352606. [DOI] [PubMed] [Google Scholar]
- 53.Schwartz GT. Reality in the economic analysis of tort law: does tort law really deter? UCLA Law Review. 1994;42:377. [Google Scholar]
- 54.Towse A, Fenn P, Gray A, Rickman N, Salinas R. Reducing harm to patients in the NHS. Will the government's compensation proposals help? Office of Health Economics; London, UK: 2003. [Google Scholar]
- 55.Sibbald B, Shen J, McBride A. Changing the skill mix of the health care workforce. J Health Serv Res Policy. 2004;9(suppl):28–38. doi: 10.1258/135581904322724112. [DOI] [PubMed] [Google Scholar]
- 56.Cawley JF, Hooker RS. Physician assistants: does the US experience have anything to offer other countries? J Health Serv Res Policy. 2003;8:65–67. doi: 10.1258/135581903321466012. [DOI] [PubMed] [Google Scholar]
- 57.Snyder JV, Sirio CA, Angus DC. Trial of nurse practitioners in intensive care. New Horiz. 1994;2:296–304. [PubMed] [Google Scholar]
- 58.Angus DC, Carlet J. on behalf of the 2002 Brussels Roundtable Participants: Surviving intensive care: a report from the 2002 Brussels Roundtable. Intensive Care Med. 2003;29:368–377. doi: 10.1007/s00134-002-1624-8. [DOI] [PubMed] [Google Scholar]
- 59.Young C, Millo JL, Salmon J. Reduction in post-ICU, in-hospital mortality following the introduction of an ICU nursing outreach service (abstract) Critical Care. 2002;6(suppl 1):117. [Google Scholar]
- 60.McMullin J, Cook DJ. Changing ICU behavior to focus on long-term outcomes. In: Angus DC, Carlet J, editors. Surviving intensive care: update in intensive care and emergency medicine No 39. Springer-Verlag; Berlin: 2003. pp. 287–296. [Google Scholar]
- 61.Guidelines for intensive care unit admission, discharge, and triage. Task Force of the American College of Critical Care Medicine, Society of Critical Care Medicine. Crit Care Med. 1999;27:633–638. [PubMed] [Google Scholar]
- 62.Nathens AB, Jurkovich GJ. The effect of organized systems of trauma care on motor vehicle crash mortality. JAMA. 2000;283:1990–1994. doi: 10.1001/jama.283.15.1990. [DOI] [PubMed] [Google Scholar]
- 63.Pearson G, Barry P, Timmins C, Stickley J, Hocking M. Changes in the profile of paediatric intensive care associated with centralisation. Intensive Care Med. 2001;27:1670–1673. doi: 10.1007/s001340101072. [DOI] [PubMed] [Google Scholar]
- 64.Nathens AB, Jurkovich GJ, Maier RV. Relationship between trauma center volume and outcomes. JAMA. 2001;285:1164–1171. doi: 10.1001/jama.285.9.1164. [DOI] [PubMed] [Google Scholar]
- 65.Nathens AB, Jurkovich GJ, Rivara FP, Maier RV. Effectiveness of state trauma systems in reducing injury-related mortality: a national evaluation. J Trauma. 2000;48:25–30. doi: 10.1097/00005373-200001000-00005. [DOI] [PubMed] [Google Scholar]
- 66.Sampalis JS, Lavoie A, Boukas S. Trauma center designation: initial impact on trauma-related mortality. J Trauma. 1995;39:232–237. doi: 10.1097/00005373-199508000-00008. [DOI] [PubMed] [Google Scholar]
- 67.Dudley RA, Johansen KL, Brand R, Rennie DJ, Milstein A. Selective referral to high-volume hospitals: estimating potentially avoidable deaths. JAMA. 2000;283:1159–1166. doi: 10.1001/jama.283.9.1159. [DOI] [PubMed] [Google Scholar]
- 68.Pearson G, Shann F, Barry P. Should paediatric intensive care be centralised? Trent versus Victoria. Lancet. 1997;349:1213–1217. doi: 10.1016/S0140-6736(96)12396-5. [DOI] [PubMed] [Google Scholar]
- 69.Mullins RJ, Veum-Stone J, Helfand M. Outcome of hospitalized injured patients after institution of a trauma system in an urban area. JAMA. 1994;271:1919–1924. doi: 10.1001/jama.1994.03510480043032. [DOI] [PubMed] [Google Scholar]
- 70.Smith JS, Jr, Martin LF, Young WW, Macioce DP. Do trauma centers improve outcome over non-trauma centers: the evaluation of regional trauma care using discharge abstract data and patient management categories. J Trauma. 1990;30:1533–1538. [PubMed] [Google Scholar]
- 71.Mullins RJ, Mann NC. Population-based research assessing the effectiveness of trauma systems. J Trauma. 1999;47:59–66. doi: 10.1097/00005373-199909001-00013. [DOI] [PubMed] [Google Scholar]
- 72.Ratcliffe J. Provision of intensive care for children. A geographically integrated service may now be achieved. BMJ. 1998;316:1547–1548. doi: 10.1136/bmj.316.7144.1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Nicholl J. Provision of intensive care for children: evidence does not support tertiary care. BMJ. 1998;317:1320–1321. [PubMed] [Google Scholar]
- 74.Angus DC, Linde-Zwirble WT, Griffin M, Clermont G, Clark RH. Epidemiology of neonatal respiratory failure in the US: projections from California and New York. Am J Respir Crit Care Med. 2001;164:1154–1160. doi: 10.1164/ajrccm.164.7.2012126. [DOI] [PubMed] [Google Scholar]
- 75.Nicholl J, Turner J. Effectiveness of a regional trauma system in reducing mortality from major trauma: before and after study. BMJ. 1997;315:1349–1354. doi: 10.1136/bmj.315.7119.1349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Angus DC, Kelley MA, Schmitz RJ, White A, Popovich JJ. Current and projected workforce requirements for care of the critically ill and patients with pulmonary disease: can we meet the requirements of an aging population? JAMA. 2000;284:2762–2770. doi: 10.1001/jama.284.21.2762. [DOI] [PubMed] [Google Scholar]
- 77.Kelley MA, Angus DC, Chalfin DB, et al. The critical care crisis in the United States: a report from the profession. Chest (in press) [DOI] [PubMed]
- 78.Sanderson CF, Hunter DJ, McKee CM, Black NA. Limitations of epidemiologically based needs assessment: the case of prostatectomy. Med Care. 1997;35:669–685. doi: 10.1097/00005650-199707000-00002. [DOI] [PubMed] [Google Scholar]
- 79.Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: A severity of disease classification system. Crit Care Med. 1985;13:818–829. [PubMed] [Google Scholar]
- 80.Knaus WA, Wagner DP, Draper EA. The APACHE III prognostic system: risk prediction of hospital mortality for critically ill hospitalized adults. Chest. 1991;100:1619–1636. doi: 10.1378/chest.100.6.1619. [DOI] [PubMed] [Google Scholar]
- 81.Lemeshow S, Teres D, Klar J, Avrunin JS, Gehlbach SH, Rapoport J. Mortality Probability Models (MPM II) based on an international cohort of intensive care unit patients. JAMA. 1993;270:2478–2486. [PubMed] [Google Scholar]
- 82.Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;270:2957–2963. doi: 10.1001/jama.270.24.2957. [DOI] [PubMed] [Google Scholar]
- 83.Rosenfeld BA, Dorman T, Breslow MJ. Intensive care unit telemedicine: alternate paradigm for providing continuous intensivist care. Crit Care Med. 2000;28:3925–3931. doi: 10.1097/00003246-200012000-00034. [DOI] [PubMed] [Google Scholar]
- 84.Black N, Rafferty AM, West E, Gough P. Health care workforce research: identifying the agenda. J Health Serv Res Policy. 2004;9(suppl):62–64. doi: 10.1258/135581904322724158. [DOI] [PubMed] [Google Scholar]


