Abstract
Purpose
To study the contribution of embryo chromosomal abnormalities in primary and secondary recurrent pregnancy loss (RPL) and to analyze the recurrence of chromosomal constitution in miscarriages from the same couple.
Methods
Retrospective study of abortion karyotypes in RPL families based on the mother’s primary or secondary RPL status (563 embryo specimens, 335 samples from primary, and 228 samples from secondary RPL). RPL was defined as two or more consecutive miscarriages. One hundred eight cases of recurrent embryo/fetal loss in 51 families were analyzed to assess the probability of having the same karyotype pattern (recurrent normal or recurrent abnormal) in both previous and subsequent pregnancy loss. The karyotypes of abortions were established using standard cytogenetic analysis, as well as interphase fluorescence in situ hybridization (FISH) and comparative genomic hybridization (CGH).
Results
The frequency of aberrations was 43.9% in abortions from primary RPL versus 52.6% in secondary RPL (p = 0.041). Women 35 years of age or older were the main contributors to this difference. The odds ratio of a subsequent abortion having the same karyotype pattern (normal or abnormal) as the previous one was 6.98 (p = 0.0013).
Conclusion
The frequency of abnormalities is higher in abortions from the secondary RPL versus primary RPL group, and this difference is due to the relative deficiency of miscarriages with abnormal karyotypes in older women with primary RPL. The probability of having the same karyotype pattern (recurrent normal or recurrent abnormal) in the previous and subsequent abortion is increased significantly compared with chance.
Keywords: Primary and secondary recurrent pregnancy loss, Chromosomal abnormalities, Miscarriage, Cytogenetics
Introduction
Recurrent pregnancy loss (RPL) is the loss of two or more consecutive pregnancies and affects up to 5% of all couples trying to conceive. The importance of genetic factors in the etiology of RPL is emphasized by the fact that the frequency of RPL among first-degree relatives of patients with idiopathic RPL is several times higher than that in the general population [1, 2]. The most common causes of RPL include endocrine imbalances, uterine abnormalities, autoimmune factors, thrombophilic disorders, and chromosomal rearrangements in either partner. However, a significant proportion of RPL cases are caused by anomalies of the embryo’s karyotype. The results of some miscarriage studies have suggested that the frequency of embryos with abnormal karyotypes is significantly lower in RPL than sporadic abortions [3–6]. At the same time, analyses of preimplantation embryos have revealed a higher frequency of embryo aneuploidy in females with RPL than in control groups [7–10]. RPL is classified as primary when all previous pregnancies ended by miscarriage and secondary when at least one of the pregnancies resulted in a live birth or progressed to more than 22 weeks. Studies of the differences in etiology and prognosis in subsequent pregnancies in primary and secondary RPL have shown contradictory results [3, 11–14]. Different mechanisms are disrupted in groups with primary and secondary RPL, so different approaches to examination and treatment would be most effective.
In this study, we analyzed the frequencies of abnormal karyotypes of spontaneous abortions in primary and secondary RPL to shed light on the genetic heterogeneity of these groups. Then, we studied the loss of several embryos/fetuses in the same family and estimated the probability of a recurrence of the normal or abnormal karyotype of abortions from a single couple. Data on previous pregnancy outcomes may be helpful, for example, for a personalized estimation of the probability of aneuploidy to decide on the use of preimplantation genetic testing for aneuploidy (PGT-A) in a specific couple.
Material and methods
A total of 563 karyotyped spontaneous abortions from women with RPL, studied in the Cytogenetic Laboratory from 1987 to 2018, were examined retrospectively. Aborted material was obtained from city hospitals, along with information regarding the patient’s age, their obstetric and gynecological history, and the number and outcomes of her previous pregnancies. This study was approved by the local ethics committee. Informed consent was obtained from all patients.
RPL was defined as two or more consecutive miscarriages. Depending on the obstetric history of the mother, the embryos and fetuses were classified into two main groups:
-
(i).
335 abortions from women with primary RPL (only miscarriages prior to gestational week 22)
-
(ii).
228 abortions from women with secondary RPL (at least one pregnancy progressing to ≥ 22 weeks resulting in a live birth or stillbirth, followed by two or more consecutive pregnancy losses)
Abortion karyotypes were established using standard trypsin-Giemsa G-band analysis after long-term fibroblast culture (479 samples) or direct preparation of the chorionic villi (17 samples). Conventional comparative genomic hybridization (CGH) (59 samples) and interphase fluorescence in situ hybridization (FISH) with centromere-enumeration probes (8 samples) were performed in cases where traditional cytogenetic analysis failed. Tissue culture, cytogenetic techniques, and interphase FISH and CGH were performed as described previously [15, 16].
Analysis of the recurrence of abortion karyotypes was carried out on 108 embryos from 51 families: 2 abortions were analyzed for 46 couples, 3 abortions for 4 couples, and 4 abortions in 1 family. The frequencies of abnormal and normal embryo karyotypes with reference to the karyotype of previous miscarriages were calculated. Patients were evaluated for propensities to RPL (hereditary thrombophilia, antiphospholipid syndrome, uterine anomalies, endocrine imbalances, and chromosomal abnormalities) that could cause repeated miscarriages, which were designated as “maternal factor.”
Variables in the groups were compared by χ2 analyses and Fisher’s exact test. As appropriate, comparisons between groups were performed using the nonparametric Mann-Whitney test. Multivariate logistic regression analysis was performed to estimate the risk for chromosomal abnormalities. Maternal age and anamnesis (primary or secondary pregnancy loss) were used as covariables in the analysis of covariance (ANCOVA). A significance level of p < 0.05 was applied for all tests. The odds ratio (OR) of the loss of the next abortion having the same karyotype pattern (normal or abnormal) as the previous abortion was calculated. Since the sample included some families with 3 and 4 abortions, the OR calculation was performed in two ways: (I) previous abortion versus all subsequent abortions or (II) all combinations of embryo karyotypes were evaluated.
Results
This study included 563 cases of miscarriage: 335 embryos and fetuses from patients with primary RPL and 228 embryos and fetuses from patients with secondary RPL. Table 1 shows the distribution of the patients with regard to parental age, gestational age of embryos/fetuses, and gravidity. The age of the women and the number of pregnancies were significantly higher in the group of mothers with secondary RPL than in those with primary RPL. Since the secondary RPL group consists of patients with repeated pregnancy losses after live birth or stillbirth, the increased age of these women is because this sample includes women with a higher number of pregnancies in anamnesis. There are different age distributions with medians shifted on 5 years toward older women in the secondary RPL group (Fig. 1).
Table 1.
Comparison of the demographic characteristics of the RPL study populations
| Total, n = 563 | Primary RPL n = 335 | Secondary RPL n = 228 | p | |
|---|---|---|---|---|
| Maternal age (y) | 29.2 ± 6.0 (17–56; 29) | 27.1 ± 5.5 (17–56; 26) | 32.2 ± 5.5 (19–44; 32) | 0.0002 |
| Paternal age (y) | 31.8 ± 6.9 (19–60; 31) | 30.1 ± 6.7 (19–60; 29) | 34.3 ± 6.4 (21–54; 34) | 0.08 |
| Gestational age (weeks) | 9.6 ± 2.8 (3.1–22; 9.3) | 9.5 ± 2.6 (3.1–18; 9.1) | 9.8 ± 3.0 (3.4–22; 9.3) | 0.85 |
| No. of pregnancies | 4.0 ± 2.3 (2–21; 3) | 3.0 ± 1.4 (2–12; 3) | 5.3 ± 2.6 (3–21; 5) | 0.007 |
| No. of miscarriages | 2.7 ± 1.2 (2–12; 2) | 2.6 ± 1.2 (2–12; 2) | 2.7 ± 1.1 (2–9; 2) | 0.56 |
Mean ± standard deviation (minimum and maximum values; median); gestational ages were calculated based on the last menstrual period; p was calculated by comparing the primary and secondary RPL
Fig. 1.
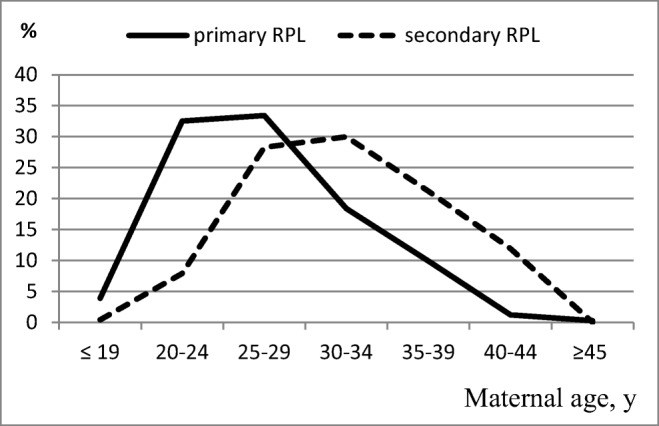
Distribution of maternal age in groups with primary and secondary RPL
In total, 52.6% (296/563) of recurrent abortions had normal karyotypes (176 with karyotypes 46,XX and 120 with karyotypes 46,XY (sex ratio, SR = 0.68)). There were no significant differences in the SR values between primary and secondary RPL groups (0.66 and 0.71, respectively, p = 0.76) (Table 2).
Table 2.
Karyotypes of spontaneous abortions in the primary and secondary RPL groups
| Karyotypes | Primary RPL | Secondary RPL | Total RPL | ||||
|---|---|---|---|---|---|---|---|
| n = 335 | n = 228 | n = 563 | |||||
| n | (%) | n | (%) | n | (%) | ||
| Total normal karyotypes | 188 | 56.1 | 108 | 47.4 | 296 | 52.6 | |
| 46,XX | 113 | 60.1 | 63 | 58.3 | 176 | 59.5 | |
| 46,XY | 75 | 39.9 | 45 | 41.7 | 120 | 40.5 | |
| Total abnormal karyotypes | 147 | 43.9 | 120 | 52.6 | 267 | 47.4 | |
| Single autosomal trisomy | 62 | 42.2 | 47 | 39.2 | 109 | 40.8 | |
| Double trisomy | 1 | 0.7 | 13 | 10.8 | 14 | 5.2 | |
| Sex chromosome trisomies | 4 | 2.7 | 5 | 4.2 | 9 | 3.4 | |
| X monosomy | 13 | 8.8 | 7 | 5.8 | 20 | 7.5 | |
| Autosomal monosomy | 24 | 16.3 | 11 | 9.2 | 35 | 13.1 | |
| Triploidy | 23 | 15.6 | 16 | 13.3 | 39 | 14.6 | |
| Tetraploidy | 2 | 1.4 | 3 | 2.5 | 5 | 1.9 | |
| Structural rearrangements | 2 | 1.4 | 1 | 0.8 | 3 | 1.1 | |
| Combineda | 15 | 10.2 | 10 | 8.3 | 25 | 9.4 | |
| Othersb | 1 | 0.7 | 7 | 5.8 | 8 | 3.0 | |
aCombined – combination of different types of abnormalities (trisomy/polyploidy/monosomy/structural)
bOthers – 46,XX/46,XY chimeras and complete hydatidiform moles with normal karyotypes. Significantly different rates are in bold
Abnormal karyotypes were found in 47.4% (267/563) of recurrent miscarriages. The overall rate of chromosomal abnormalities in the secondary RPL group was significantly increased compared with the primary RPL group (52.6% (120/228) and 43.9% (147/335), respectively, p = 0.041). By multivariate logistic regression analysis, maternal age was the only statistically significant risk factor for chromosomal aberrations (p = 0.0003), whereas the impact of obstetric history (primary versus secondary pregnancy loss) was not significant (p = 0.7593). Since the maternal age affects the incidence of aneuploidy in embryos, we stratified women into three age subgroups (up to 24, 25–34, and over 35 years old) to directly compare primary and secondary RPL. We observed that the frequency of embryos with abnormal karyotypes increases significantly in women in the older age group (35 years of age or older) with secondary RPL but not with primary RPL (Fig. 2).
Fig. 2.
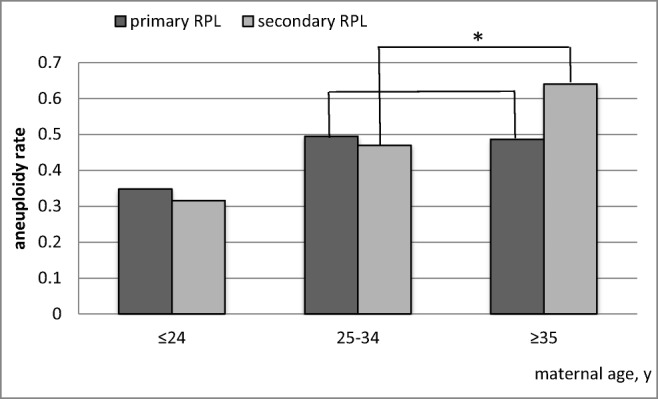
Comparison of the frequencies of the abnormal karyotypes of abortions between groups with primary and secondary RPL stratified by maternal age. *Statistically significant increase in frequency compared to younger women with secondary RPL
We found similar rates of the different types of chromosomal abnormalities in the primary and secondary RPL groups (Table 2). The most common chromosomal abnormalities were autosomal trisomies, which accounted for 40.8% of abnormal karyotypes, and another 5.2% were double trisomies. Autosome monosomies occurred in three embryos (two on chromosome 22 and one on chromosome 13), and all were mosaic with a normal cell line. We found similar rates of miscarriages with monosomy X, polyploidy, structural chromosomal rearrangements, and combined chromosomal anomalies in the primary and secondary RPL groups. Abortions with double trisomy occurred significantly more frequently in women with secondary RPL (13/228 cases), whereas only one such abortion (1/335) was detected in the group with primary RPL (p = 0.0002). The average age of women having embryos with double trisomy was higher than the average maternal age in the sample (38.6 versus 29.2 years, p = 0.000002). Another class of karyotypes was significantly more frequent in women with secondary RPL and included 46,XX/46,XY chimeras and complete hydatidiform moles with a normal karyotype, which were included in the “Others” category (Table 2).
There were 132 trisomy cases, of which 9 had X/Y chromosome trisomy and were assigned to “sex chromosomal abnormality” and 14 were double trisomies. In total, trisomies for 19 of the 22 autosomes were observed, while those for chromosomes 1, 5, and 19 were not detected. Trisomy 16 was the most common and was present in 39% of trisomic abortions, followed by trisomies 21, 15, 18, and 22 (frequency of 10–20%). Trisomies 2, 7, 9, 13, and 20 were found in approximately 5%, and abortions were rarely observed with trisomies of chromosomes 3, 4, 6, 8, 10, 12, 14, and 17. There were no significant differences in the frequency of individual trisomy between the primary and secondary RPL groups.
The analysis of the recurrence of miscarriage karyotypes was carried out for 26 women with primary and 25 women with secondary RPL. Overall, 108 embryos from 51 families were examined: the karyotypes of 2 abortions were found for 46 couples, 3 abortions for 4 couples, and 4 abortions for 1 family. The karyotype of the embryo was classified as a “normal karyotype” (46,XX or 46,XY with the exception of complete hydatidiform moles) or “abnormal karyotype” (all types of chromosomal abnormalities). Depending on the combination of abortions, women were assigned to one of four cohorts:
AA – both abortions with abnormal karyotypes
AN – previous abortion(s) with abnormal karyotype followed by abortion with normal karyotype
NA – previous abortion(s) with normal karyotype followed by abortion with abnormal karyotype
NN – both (all) abortions with a normal karyotype
After a miscarriage with an abnormal karyotype, the next abortion more often had an abnormal karyotype than a normal karyotype (72.0% and 28.0%, respectively). If the previous abortion had a normal chromosomal complement, the next one was also normal in most cases (73.1%), while the abnormal karyotype occurred in 26.9% of cases. The OR of the loss of the next embryo with the same karyotype pattern (either normal or abnormal) as the previous abortion was estimated to be 6.98 (95% CI: 2.04–23.88; p = 0.0013) according to method I and 7.84 (95% CI: 2.48–24.80; p = 0.0002) according to method II. Thus, there is an increased probability that a subsequent abortion will have the same karyotype pattern (either normal or abnormal) as the previous one.
In 37.3% (19/51) of the women, all spontaneous abortions had a normal karyotype (cohort NN). Of these, the “maternal factor” was observed in only 7 patients, 10 women did not have known miscarriage causes, including 1 woman with 3, and 1 with 4 abortions with a normal karyotype (Table 3, data are absent for 2 women). In addition, patients from this cohort were more likely to have a primary RPL (Table 3). In our study, primary RPL significantly increased the chance of having both miscarriages with normal karyotypes (OR = 3.69; 95% CI: 1.11–12.25; p = 0.029).
Table 3.
Combinations of karyotypes of spontaneous abortions from couples with recurrent pregnancy losses
| Cohorts | AA | AN | NA | NN |
|---|---|---|---|---|
| Karyotype of previous abortion | Abnormal | Normal | ||
| Karyotype of subsequent abortion | Abnormal | Normal | Abnormal | Normal |
| Number of women (% of all) | 18 (35.3%) | 7 (13.7%) | 7 (13.7%) | 19 (37.3%) |
| Number of abortions | 36 | 15 | 16 | 41 |
| Women age | 30.4 ± 5.4 | 30.7 ± 5.1 | 33.1 ± 6.2 | 27.5 ± 5.3a |
| RPL: primary/secondary | 9 / 9 | 4 / 10 | 13 / 6b | |
| Mean number of pregnancies | 3.5 ± 1.9 | 5.1 ± 2.2 | 3.3 ± 1.5 | |
| Mean number of miscarriages | 2.1 ± 1.4c | 2.9 ± 1.6 | 2.5 ± 1.0 | |
| Maternal factor: yes/not | 5/12 | 6/8 | 7/10 | |
aSignificant differences in age from cohorts AA and NA
bSignificant differences in the live birth from mixed cohort (AN+NA)
cSignificant differences in the number of miscarriages from mixed cohort (AN+NA)
There were 18 RPL women with abnormal karyotypes in both miscarriages (35%, 18/51), and the most common combination of abnormalities (6 families) was recurrent trisomies on different chromosomes (heterotrisomy). In one family, both abortions had trisomy 16. Eleven cases of recurrent abnormal miscarriages were combinations of different types of anomalies: trisomy/triploidy (4 couples), trisomy/tetraploidy (4 couples), trisomy/X monosomy (2 couples), and mosaic autosomal monosomy/tetraploidy (1 couple). Within the AA cohort, the average age of women with two trisomic miscarriages was significantly higher than the average age of women having abortions with different types of anomalies (33.1 ± 3.45 and 28.7 ± 5.78 years, respectively, p = 0.025).
Since the AN and NA cohorts included abortions with normal and abnormal karyotypes together and all the characteristics between these groups did not differ, we combined them into a mixed group. On average, women from this group had a greater number of pregnancies and a greater number of miscarriages than others (Table 3).
Overall, 59% (30/51) of women had the “maternal factor,” and 35% (18/51) were healthy, while for 6% (3/51), no data were available. Interestingly, no significant differences in the distribution of maternal factors between the cohorts were detected (Table 3). Combinations of repeated abnormal miscarriages with the “maternal factor” in the same women were quite common. For example, in group AA, 5 (out of 17) women had this pathology, including 1 with hereditary thrombophilia and 8 spontaneous abortions in her medical history; both of her karyotyped embryos had autosomal trisomy.
Discussion
The karyotyping method affects the rate of aneuploidy detection in products of conception, and the phenomenon of maternal cell contamination is an especially substantial factor influencing the results of cytogenetic analysis of spontaneous abortions [17–19]. To balance the influence of this factor, a comparison of the karyotypes of spontaneous abortions from different groups should be carried out within the same study. Such comparative studies of primary and secondary RPL are rare, and their results are contradictory (Table 4). In this study, we analyzed abortion karyotype rates in primary and secondary RPL in the largest total sample size of recurrent miscarriages compared with published data. The frequency of abnormal abortion karyotypes in the primary RPL group was lower than that in the secondary RPL group (43.9% and 52.6%, respectively, p = 0.041), and multivariate logistic regression analysis revealed that the age of women, not the obstetric history, makes the main contribution to the frequency of chromosomal abnormalities in repeated spontaneous abortions. Therefore, considering the 5-year difference in mean maternal age between the two analyzed RPL groups, the observed difference in abnormality rates is expected. Surprisingly, the rate of abnormal embryos increased significantly in older women (≥ 35 years) with secondary RPL but not with a primary RPL. In a large sample of preimplantation embryos, the proportion of aneuploid embryos remained roughly constant between 24 and 35 years of age, with a striking increase thereafter [24]. Therefore, our results imply that the lower contribution of chromosomal abnormalities to embryonic death in primary RPL is due to the “lack” of aneuploid abortions in older primary RPL women (Fig. 2).
Table 4.
Comparative frequencies of chromosomal abnormalities in spontaneous abortions from couples with primary and secondary RPL
| Population | Aneuploidy rate | Statistical significance | Age of mother (differences between groups) | Reference | |
|---|---|---|---|---|---|
| Primary RPL (n) | Secondary RPL (n) | ||||
| Israel | 26.7% (75) | 32% (50) | p > 0.05 | 31.6 ± 5.7 (n.d.) | [20] |
| Japan | 39.8% (404) | 47.4% (78) | p = 0.044 | 32.4 ± 4.45 (n.d.) | [21] |
| Austria | 56.4% (95) | 81.3% (50) | p < 0.05 | 33.9 ± 5.6 vs 35.1 ± 6.1 (no differences) | [22] |
| Poland | 64.7% (204) | 74.4% (164) | p = 0.046* | 34.2 ± 4.9 (n.d) | [23] |
| Russia | 43.9% (335) | 52.6% (228) | p = 0.041 |
27.1 ± 5.5 vs 32.2 ± 5.5 (significant differences) |
This study |
*Without correction for maternal age
n sample size; n.d. no data
In the present study, a significant difference in the rate of double trisomy was found in abortions from women with primary and secondary RPL (0.7% and 10.8%, p = 0.0002). In previous studies, double trisomies were detected significantly more often in RPL than in sporadic abortions [3, 5]. One of the possible reasons for this may be that the mother’s carrier status of mutations of some genes affected the aneuploidy rate [25]. An alternative explanation may be the age of the woman: the average age of mothers with abortions with double trisomy in our sample was 38.6 years, which is similar to that reported in other work [26, 27]. Advanced maternal age increases the risk of meiotic disturbances in oogenesis [28] and decreases endometrial selection against abnormal embryos [29]. However, this does not explain why double aneuploidy occurred almost exclusively in women with secondary RPL in the present work. Perhaps, this is a combined effect of an advanced maternal age and a greater contribution of aneuploidy to secondary RPL.
We analyzed the recurrence of abortion karyotypes in 26 women with primary RPL and 25 women with secondary RPL (a total of 108 embryos and fetuses from 51 families). Abnormal karyotypes cause an average of approximately 50% of first trimester spontaneous abortions in humans [30, 31]. Therefore, regardless of the karyotype of the first miscarriage, the second should have a normal karyotype in approximately half of the cases. In general, we found an increased likelihood for the subsequent abortion to have the same karyotype pattern (either normal or abnormal) as the previous one: the OR of losing a second embryo with the same karyotype pattern as that of the first abortion was OR = 6.98 (OR = 7.84, depending on the method of calculation).
In our study, 62.6% of patients with RPL had the same karyotype pattern in both their abortions (recurrent normal or recurrent abnormal), which is consistent with the data [21, 32] (73% and 73.7% of karyotype recurrence). These data argue that the repeatability of abortion karyotypes is more than what is expected by chance, at least in some women.
In our study, after an abortion with a normal karyotype, the next one was normal in 73.1% of cases, which agrees with data from earlier observations [33, 34] that the normal chromosomal status of a miscarriage significantly increases the likelihood of subsequent abortion to be with a normal karyotype. In our study, women with recurrent abortions with a normal karyotype were younger than women of other groups (Table 3), and primary RPL women had a significantly increased chance of having both miscarriages with normal karyotypes (OR = 3.69; 95% CI: 1.11–12.25; p = 0.029). As reported previously, the karyotype of normal abortion is associated with a large number of RPL etiological factors of the mothers [35, 36].
The recurrent loss of the embryos with normal karyotypes indicates a permanent unfavorable maternal factor, which explains that in RPL women, the normal karyotype of a previous abortion is associated with an unfavorable prognosis for subsequent pregnancy [3, 37].
In our study, in 35% (18/51) of women, both pregnancy losses were caused by anomalies of the embryo karyotype. A comparison of our results with the findings reported by other studies that analyzed karyotypes of multiple abortions from the same woman is provided in Table 5. There are some previously reported cases in which a couple had three or more abortions with karyotype anomalies, most often trisomy on different chromosomes, but there were also polyploidies [39–41]. It is possible that some women have a genetic predisposition to an embryo karyotype imbalance. Repeated cases of abortions with chromosomal abnormalities in one family can be the result of the following:
-
i).
Random recurrence due to increased risk of chromosomal nondisjunction with advanced maternal age [34, 40]. However, in our study, the average age of mothers with recurrent abnormal karyotypes of spontaneous abortions was 30.4 ± 5.4 years (Table 3).
-
ii).
Gonadal mosaicism in the mother. The presence of an aneuploid clone in the gonads can lead to the repeated appearance of embryos with the same chromosomal anomalies. Considering that reported cases of homotrisomy usually include chromosomes 16, 22, and 15 [40], the most frequent in spontaneous abortions [31], the possibility of random coincidence cannot be ruled out.
-
iii).
Increased risk of meiotic nondisjunction. This is possible due to genetic variability in the meiotic recombination rate, mutations in the genes regulating the process of meiosis, and variability of ovarian aging, which can differ among women of the same biological age. For example, a relationship was found between certain genetic variants of the MTHFR gene, which is involved in folic acid metabolism, and the SYCP3 gene, which encodes a component of the synaptonemal complex in parents with embryos with abnormal karyotypes [42, 43]. However, a large-scale whole-genome study did not reveal associations of the frequency of chromosome nondisjunction in oogenesis with the maternal genotype [44].
-
iv).
Increased risk of mitotic nondisjunction in early embryogenesis. A high frequency of chromosomal mosaicism is inherent to early human preimplantation development, including morphologically normal blastocysts [45, 46]. There is interindividual variability in the rate of somatic chromosomal nondisjunction in progeny: some patients produced “chaotic” embryos repeatedly in several IVF cycles, whereas other patients did not produce such embryos at all [47]. Recently, an association was found between the embryo karyotype aberrations that occurred in the first mitotic divisions and the mother’s carrier status of a variant of the PLK4 gene, which plays a role in centriole duplication [44].
Table 5.
Frequency of recurrent abnormal karyotypes in RPL abortions
| Reference | Population | Frequency of recurrent abortions with abnormal karyotype |
|---|---|---|
| Warburton et al. [34] | USA, New York | 19.2% (24/125) |
| Warburton et al. [34] | USA, Honolulu | 27.7% (41/148) |
| Sullivan et al. [4] | USA, Utah | 10% (3/30) |
| Skrzypczak et al. [38] | Poland | 13% (3/23) |
| Sugiura-Ogasawara et al. [21] | Japan | 33.6% (32/95) |
| Feichtinger et al. [32] | Austria, Vienna | 56% (42/75) |
| This study | Russia | 35.3% (18/51) |
Data from prenatal and preimplantation genetic testing for aneuploidy (PGT-A) suggest that an increased rate of aneuploidy in the next conception exists in women who previously had embryo aneuploidy [10, 48, 49], especially for young patients (< 35 years) [50]. These studies support the hypothesis that in some women, abnormal abortion karyotypes occur more often than what is expected by chance.
Thus, the high frequency of recurrent losses of product of conceptions with abnormal karyotypes found in our study, as well as in others [21, 32], may be caused by the predisposition of some women to form embryos with chromosomal abnormalities. The mechanism of such predisposition is unclear. Combinations of different types of aberrations are likely random because different mechanisms are involved in their formation—nondisjunction in trisomy, anaphase lag in monosomy X, dispermy or impaired meiosis in triploidy and failure of mitosis in tetraploidy. Repeated cases of trisomy in the family may be due to a predisposition for nondisjunction in oocytes or in early embryogenesis.
Conclusions
The frequency of chromosomal abnormalities was higher after a previous abortion with an abnormal karyotype and in abortions from the secondary RPL women. Therefore, the underlying mechanisms of recurrent aneuploid pregnancy losses are likely different from recurrent miscarriages with a normal karyotype. A previous abortion with an abnormal karyotype or secondary RPL may be a marker of an increased likelihood of aneuploidy in subsequent pregnancy. PGT-A should be more effective in such families than in primary RPL women or those who had a previous abortion with a normal karyotype. Thus, cytogenetic information about previous pregnancy outcomes allows for a personalized assessment of aneuploidy probability and may be valuable in determining when PGT-A may be useful.
Authors’ contributions
NTV designed the study, performed sample collection and cell culture, conducted data analysis and interpretation, and drafted the manuscript. SEA and SNN performed cytogenetic analysis of G-banded metaphase chromosomes. ZDI contributed to CGH analysis, and TEN performed cell culture. LIN contributed to sample collection and analysis and revised the article for important intellectual content.
Funding information
The study was supported by the Ministry of Science and Education of the Russian Federation (budgetary project of the Research Institute of Medical Genetics, TNRMC, # AAAA-A19–11902089005-5).
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Scientific Ethics Committee of the Research Institute of Medical Genetics of the Tomsk NRMC and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kolte AM, Nielsen HS, Moltke I, Degn B, Pedersen B, Sunde L, Nielsen FC, Christiansen OB. A genome-wide scan in affected sibling pairs with idiopathic recurrent miscarriage suggests genetic linkage. Mol Hum Reprod. 2011;17:379–385. doi: 10.1093/molehr/gar003. [DOI] [PubMed] [Google Scholar]
- 2.Miskovic S, Culic V, Konjevoda P, Pavelic J. Positive reproductive family history for spontaneous abortion: predictor for recurrent miscarriage in young couples. Eur J Obstet Gynecol Reprod Biol. 2012;161:182–186. doi: 10.1016/j.ejogrb.2011.12.027. [DOI] [PubMed] [Google Scholar]
- 3.Ogasawara M, Aoki K, Okada S, Suzumori K. Embryonic karyotype of abortuses in relation to the number of previous miscarriages. Fertil Steril. 2000;73:300–304. doi: 10.1016/s0015-0282(99)00495-1. [DOI] [PubMed] [Google Scholar]
- 4.Sullivan AE, Silver RM, LaCoursiere DY, Porter TF, Branch DW. Recurrent fetal aneuploidy and recurrent miscarriage. Obstet Gynecol. 2004;104:784–788. doi: 10.1097/01.AOG.0000137832.86727.e2. [DOI] [PubMed] [Google Scholar]
- 5.Nikitina TV, Sazhenova EA, Tolmacheva EN, Sukhanova NN, Kashevarova AA, Skryabin NA, Vasilyev SA, Nemtseva TN, Yuriev SY, Lebedev IN. Comparative cytogenetic analysis of spontaneous abortions in recurrent and sporadic pregnancy losses. Biomed Hub. 2016;1:1–11. doi: 10.1159/000446099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ozawa N, Ogawa K, Sasaki A, Mitsui M, Wada S, Sago H. Maternal age, history of miscarriage, and embryonic/fetal size are associated with cytogenetic results of spontaneous early miscarriages. J Assist Reprod Genet. 2019;36:749–757. doi: 10.1007/s10815-019-01415-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bianco K, Caughey AB, Shaffer BL, Davis R, Norton ME. History of miscarriage and increased incidence of fetal aneuploidy in subsequent pregnancy. Obstet Gynecol. 2006;107:1098–1102. doi: 10.1097/01.AOG.0000215560.86673.22. [DOI] [PubMed] [Google Scholar]
- 8.Rubio C, Buendia P, Rodrigo L, Mercader A, Mateu E, Peinado V, Delgado A, Milan M, Mir P, Simon C, Remohi J, Pellicer A. Prognostic factors for preimplantation genetic screening in repeated pregnancy loss. Reprod BioMed Online. 2009;18:687–693. doi: 10.1016/s1472-6483(10)60015-6. [DOI] [PubMed] [Google Scholar]
- 9.Hodes-Wertz B, Grifo J, Ghadir S, Kaplan B, Laskin CA, Glassner M, Munne S. Idiopathic recurrent miscarriage is caused mostly by aneuploid embryos. Fertil Steril. 2012;98:675–680. doi: 10.1016/j.fertnstert.2012.05.025. [DOI] [PubMed] [Google Scholar]
- 10.Kort JD, McCoy RC, Demko Z, Lathi RB. Are blastocyst aneuploidy rates different between fertile and infertile populations? J Assist Reprod Genet. 2018;35:403–408. doi: 10.1007/s10815-017-1060-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Alberman E. The epidemiology of repeated abortion. In: Beard RW, Sharp F, editors. Early pregnancy loss: mechanisms and treatment. London: Springer-Verlag; 1988. pp. 9–17. [Google Scholar]
- 12.Brigham SA, Conlon C, Farquharson RG. A longitudinal study of pregnancy outcome following idiopathic recurrent miscarriage. Hum Reprod. 1999;14:2868–2871. doi: 10.1093/humrep/14.11.2868. [DOI] [PubMed] [Google Scholar]
- 13.Shapira E, Ratzon R, Shoham-Vardi I, Serjienko R, Mazor M, Bashiri A. Primary vs. secondary recurrent pregnancy loss--epidemiological characteristics, etiology, and next pregnancy outcome. J Perinat Med. 2012;40:389–396. doi: 10.1515/jpm-2011-0315. [DOI] [PubMed] [Google Scholar]
- 14.Egerup P, Kolte AM, Larsen EC, Krog M, Nielsen HS, Christiansen OB. Recurrent pregnancy loss: what is the impact of consecutive versus non-consecutive losses? Hum Reprod. 2016;31:2428–2434. doi: 10.1093/humrep/dew169. [DOI] [PubMed] [Google Scholar]
- 15.Ostroverkhova NV, Nazarenko SA, Lebedev IN, Cheremnykh AD, Nikitina TV, Sukhanova NN. Detection of aneuploidy in spontaneous abortions using the comparative hybridization method. Genetika. 2002;38:1690–1698. [PubMed] [Google Scholar]
- 16.Lebedev IN, Ostroverkhova NV, Nikitina TV, Sukhanova NN, Nazarenko SA. Features of chromosomal abnormalities in spontaneous abortion cell culture failures detected by interphase FISH analysis. Eur J Hum Genet. 2004;12:513–520. doi: 10.1038/sj.ejhg.5201178. [DOI] [PubMed] [Google Scholar]
- 17.Bell KA, van Deerlin PG, Haddad BR, Feinberg RF. Cytogenetic diagnosis of “normal 46,XX” karyotypes in spontaneous abortions frequently may be misleading. Fertil Steril. 1999;71:334–341. doi: 10.1016/s0015-0282(98)00445-2. [DOI] [PubMed] [Google Scholar]
- 18.Jarrett KL, Michaelis RC, Phelan MC, Vincent VA, Best RG. Microsatellite analysis reveals a high incidence of maternal cell contamination in 46,XX products of conception consisting of villi or a combination of villi and membranous material. Am J Obstet Gynecol. 2001;185:198–203. doi: 10.1067/mob.2001.114692. [DOI] [PubMed] [Google Scholar]
- 19.Nikitina TV, Lebedev IN, Sukhanova NN, Sazhenova EA, Nazarenko SA. A mathematical model for evaluation of maternal cell contamination in cultured cells from spontaneous abortions: significance for cytogenetic analysis of prenatal selection factors. Fertil Steril. 2005;83:964–972. doi: 10.1016/j.fertnstert.2004.12.009. [DOI] [PubMed] [Google Scholar]
- 20.Carp H, Toder V, Aviram A, Daniely M, Mashiach S, Barkai G. Karyotype of the abortus in recurrent miscarriage. Fertil Steril. 2001;75:678–682. doi: 10.1016/s0015-0282(00)01801-x. [DOI] [PubMed] [Google Scholar]
- 21.Sugiura-Ogasawara M, Ozaki Y, Katano K, Suzumori N, Kitaori T, Mizutani E. Abnormal embryonic karyotype is the most frequent cause of recurrent miscarriage. Hum Reprod. 2012;27:2297–2303. doi: 10.1093/humrep/des179. [DOI] [PubMed] [Google Scholar]
- 22.Feichtinger M, Wallner E, Hartmann B, Reiner A, Philipp T. Transcervical embryoscopic and cytogenetic findings reveal distinctive differences in primary and secondary recurrent pregnancy loss. Fertil Steril. 2017;107:144–149. doi: 10.1016/j.fertnstert.2016.09.037. [DOI] [PubMed] [Google Scholar]
- 23.Massalska D, Bijok J, Kucinska-Chahwan A. Chromosomal abnormalities in the first-trimester spontaneous pregnancy loss. Austin J Obstet Gynecol. 2018;5:1116. [Google Scholar]
- 24.Demko ZP, Simon AL, McCoy RC, Petrov DA, Rabinowitz M. Effects of maternal age on euploidy rates in a large cohort of embryos analyzed with 24-chromosome single-nucleotide polymorphism-based preimplantation genetic screening. Fertil Steril. 2016;105:1307–1313. doi: 10.1016/j.fertnstert.2016.01.025. [DOI] [PubMed] [Google Scholar]
- 25.Lenzi ML, Smith J, Snowden T, Kim M, Fishel R, Poulos BK, Cohen PE. Extreme heterogeneity in the molecular events leading to the establishment of chiasmata during meiosis I in human oocytes. Am J Hum Genet. 2005;76:112–127. doi: 10.1086/427268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Menasha J, Levy B, Hirschhorn K, Kardon NB. Incidence and spectrum of chromosome abnormalities in spontaneous abortions: new insights from a 12-year study. Genet Med. 2005;7:251–263. doi: 10.1097/01.gim.0000160075.96707.04. [DOI] [PubMed] [Google Scholar]
- 27.Subramaniyam S, Pulijaal VR, Mathew S. Double and multiple chromosomal aneuploidies in spontaneous abortions: a single institutional experience. J Hum Reprod Sci. 2014;7:262–268. doi: 10.4103/0974-1208.147494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kurahashi H, Tsutsumi M, Nishiyama S, Kogo H, Inagaki H, Ohye T. Molecular basis of maternal age-related increase in oocyte aneuploidy. Congenit Anom (Kyoto) 2012;52:8–15. doi: 10.1111/j.1741-4520.2011.00350.x. [DOI] [PubMed] [Google Scholar]
- 29.Coulam C. What about superfertility, decidualization, and natural selection? J Assist Reprod Genet. 2016;33:577–580. doi: 10.1007/s10815-016-0658-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.van den Berg MM, van Maarle MC, van Wely M, Goddijn M. Genetics of early miscarriage. Biochim Biophys Acta. 1822;2012:1951–1959. doi: 10.1016/j.bbadis.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 31.Sahoo T, Dzidic N, Strecker MN, Commander S, Travis MK, Doherty C, Tyson RW, Mendoza AE, Stephenson M, Dise CA, Benito CW, Ziadie MS, Hovanes K. Comprehensive genetic analysis of pregnancy loss by chromosomal microarrays: outcomes, benefits, and challenges. Genet Med. 2017;19:83–89. doi: 10.1038/gim.2016.69. [DOI] [PubMed] [Google Scholar]
- 32.Feichtinger M, Reiner A, Hartmann B, Philipp T. Embryoscopy and karyotype findings of repeated miscarriages in recurrent pregnancy loss and spontaneous pregnancy loss. J Assist Reprod Genet. 2018;35:1401–1406. doi: 10.1007/s10815-018-1226-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hassold TJ. A cytogenetic study of repeated spontaneous abortions. Am J Hum Genet. 1980;32:723–730. [PMC free article] [PubMed] [Google Scholar]
- 34.Warburton D, Kline J, Stein Z, Hutzler M, Chin A, Hassold T. Does the karyotype of a spontaneous abortion predict the karyotype of a subsequent abortion? Evidence from 273 women with two karyotyped spontaneous abortions. Am J Hum Genet. 1987;41:465–483. [PMC free article] [PubMed] [Google Scholar]
- 35.Maslow BS, Budinetz T, Sueldo C, Anspach E, Engmann L, Benadiva C, Nulsen JC., 3rd Single-nucleotide polymorphism-microarray ploidy analysis of paraffin-embedded products of conception in recurrent pregnancy loss evaluations. Obstet Gynecol. 2015;126:175–181. doi: 10.1097/AOG.0000000000000904. [DOI] [PubMed] [Google Scholar]
- 36.Popescu F, Jaslow CR, Kutteh WH. Recurrent pregnancy loss evaluation combined with 24-chromosome microarray of miscarriage tissue provides a probable or definite cause of pregnancy loss in over 90% of patients. Hum Reprod. 2018;33:579–587. doi: 10.1093/humrep/dey021. [DOI] [PubMed] [Google Scholar]
- 37.Stephenson MD, Awartani KA, Robinson WP. Cytogenetic analysis of miscarriages from couples with recurrent miscarriage: a case-control study. Hum Reprod. 2002;17:446–451. doi: 10.1093/humrep/17.2.446. [DOI] [PubMed] [Google Scholar]
- 38.Skrzypczak J, Kwinecka-Dmitriew B, Zakrzewska M, Latos-Bielenska A. Do chromosomal abnormalities reappear in subsequent pregnancies and how often? Ginekol Pol. 2010;81:681–686. [PubMed] [Google Scholar]
- 39.Ulm JE. Recurrent trisomies: chance or inherited predisposition? J Genet Couns. 1999;8:109–117. doi: 10.1023/A:1022842931704. [DOI] [PubMed] [Google Scholar]
- 40.Robinson WP, McFadden DE, Stephenson MD. The origin of abnormalities in recurrent aneuploidy/polyploidy. Am J Hum Genet. 2001;69:1245–1254. doi: 10.1086/324468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Filges I, Manokhina I, Peñaherrera MS, McFadden DE, Louie K, Nosova E, Friedman JM, Robinson WP. Recurrent triploidy due to a failure to complete maternal meiosis II: whole-exome sequencing reveals candidate variants. Mol Hum Reprod. 2015;21:339–346. doi: 10.1093/molehr/gau112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Enciso M, Sarasa J, Xanthopoulou L, Bristow S, Bowles M, Fragouli E, Delhanty J, Wells D. Polymorphisms in the MTHFR gene influence embryo viability and the incidence of aneuploidy. Hum Genet. 2016;135:555–568. doi: 10.1007/s00439-016-1652-z. [DOI] [PubMed] [Google Scholar]
- 43.Sazegary A, Kalantar SM, Pashaiefar H, Mohtaram S, Honarvar N, Feizollahi Z, Ghasemi N. The T657C polymorphism on the SYCP3 gene is associated with recurrent pregnancy loss. J Assist Reprod Genet. 2014;31(10):1377–1381. doi: 10.1007/s10815-014-0272-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.McCoy RC, Demko Z, Ryan A, Banjevic M, Hill M, Sigurjonsson S, Rabinowitz M, Fraser HB, Petrov DA. Common variants spanning PLK4 are associated with mitotic-origin aneuploidy in human embryos. Hum Genet. 2015;348(6231):235–238. doi: 10.1126/science.aaa3337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mertzanidou A, Wilton L, Cheng J, Spits C, Vanneste E, Moreau Y, Vermeesch JR, Sermon K. Microarray analysis reveals abnormal chromosomal complements in over 70% of 14 normally developing human embryos. Hum Reprod. 2013;28(1):256–264. doi: 10.1093/humrep/des362. [DOI] [PubMed] [Google Scholar]
- 46.Taylor TH, Gitlin SA, Patrick JL, Crain JL, Wilson JM, Griffin DK. The origin, mechanisms, incidence and clinical consequences of chromosomal mosaicism in humans. Hum Reprod Update. 2014;20(4):571–581. doi: 10.1093/humupd/dmu016. [DOI] [PubMed] [Google Scholar]
- 47.Delhanty JD, Harper JC, Ao A, Handyside AH, Winston RM. Multicolour FISH detects frequent chromosomal mosaicism and chaotic division in normal preimplantation embryos from fertile patients. Hum Genet. 1997;99(6):755–760. doi: 10.1007/s004390050443. [DOI] [PubMed] [Google Scholar]
- 48.Warburton D, Dallaire L, Thangavelu M, Ross L, Levin B, Kline J. Trisomy recurrence: a reconsideration based on North American data. Am J Hum Genet. 2004;75:376–385. doi: 10.1086/423331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Al-Asmar N, Peinado V, Vera M, Remohi J, Pellicer A, Simon C, Hassold T, Rubio C. Chromosomal abnormalities in embryos from couples with a previous aneuploid miscarriage. Fertil Steril. 2012;98:145–150. doi: 10.1016/j.fertnstert.2012.03.035. [DOI] [PubMed] [Google Scholar]
- 50.De Souza E, Halliday J, Chan A, Bower C, Morris JK. Recurrence risks for trisomies 13, 18, and 21. Am J Med Genet A. 2009;149a:2716–2722. doi: 10.1002/ajmg.a.33099. [DOI] [PubMed] [Google Scholar]


