Abstract
Background
The impact of viral infections on acute exacerbations in idiopathic pulmonary fibrosis (IPF) and/or non-IPF interstitial lung disease (ILDs) has been scarcely described.
Objectives
To elucidate the frequency of virus infections in patients with IPF or non-IPF ILDs including idiopathic interstitial pneumonia (IIP) or connective tissue disease (CTD)-associated pneumonia, and its influence on their short-term mortality.
Methods
We prospectively enrolled adult patients with acute exacerbation of IPF and non-IPF ILDs who were admitted to the hospital during the last 3 years, and examined the respiratory samples obtained from nasopharyngeal, sputum, and bronchoalveolar lavage fluid.
Results
A total of 78 patients were identified, consisting of 27 patients with acute exacerbation of IPF and 51 patients with non-IPF ILDs (IIP: n = 27, CTD-associated IP: n = 24). Of all patients, 15 (19.2%) had viruses detected in their respiratory samples including the human herpesvirus 7 (HHV7; n = 4) and cytomegalovirus (CMV) plus HHV7 (n = 3). The proportion of virus infections in the IPF and non-IPF ILDs groups was comparable. The Kaplan-Meier survival curves over 60 days revealed a lower survival probability in the virus positive group (n = 15, 60%) than in the virus negative group (n = 60, 83.3%, p < 0.05). However, the virus infection itself could not predict the 60-day survival probability using simple logistic regression analysis.
Conclusions
Viral infections, mostly CMV or HHV7, were identified in both patients with acute exacerbation of IPF and non-IPF ILDs, but the clinical significance on short-term mortality or isolation itself from respiratory samples remains to be determined.
Keywords: Acute exacerbation, Idiopathic interstitial pneumonia, Virus detection, Short term mortality, IPF, Non-IPF interstitial lung disease
Abbreviations: BALF, bronchoalveolar lavage fluid; CMV, cytomegalovirus; CTD, connective tissue disease; HHV, human herpes virus; HMPV, human metapneumovirus; HPIV, human parainfluenza viruses; HRSV, human respiratory syncytial virus; HRV, human rhinovirus; ILDs, interstitial lung diseases; IIP, idiopathic interstitial pneumonia; IPF, idiopathic pulmonary fibrosis; PCR, polymerase chain reaction; UIP, usual interstitial pneumonia
1. Introduction
To date, few studies have addressed the etiology of acute exacerbation of idiopathic pulmonary fibrosis (IPF) [[1], [2], [3], [4]], and the impact of infections has yielded mixed results. Furthermore, the significance of viral infections in other types of interstitial pneumonia for triggering acute exacerbation remains to be determined. Therefore, we prospectively examined the relationship between virus infection and acute exacerbation of interstitial pneumonia including IPF, other types of idiopathic interstitial pneumonia (IIP), and connective tissue disease (CTD)-associated interstitial pneumonia.
2. Material and methods
2.1. Patients and study design
We prospectively enrolled adult hospitalized patients having acute exacerbation of interstitial pneumonia admitted at Kyorin University Hospital from August 2012 to August 2015. The definition of acute exacerbation of IIP was based on a previous report [1] as follows: (1) unexplained development of dyspnea within 30 days; (2) presence of new, bilateral pulmonary ground glass abnormalities, consolidation superimposed on a background of a reticular and/or honeycomb pattern on chest computed tomography; (3) acute respiratory symptoms; (4) no pathogenic bacterial in the bronchoalveolar lavage fluid (BALF); and (5) exclusion of alternative causes such as left heart failure and pulmonary embolism. We approved the patients who had no usual interstitial pneumonia (UIP) pattern on their radiological background. Therefore, we also enrolled patients with acute exacerbation of CTD-associated interstitial pneumonia.
Regarding these criteria, only the bronchoalveolar lavage procedure was considered to be not essential for patients to be enrolled in this study. If patients previously or concurrently fulfilled the consensus criteria of the American Thoracic Society/European Respiratory Society for a diagnosis of IPF [5], they were diagnosed with an acute exacerbation of IPF.
2.2. Samples and clinical data collection
The respiratory samples were obtained from sputum or nasopharyngeal swab or BALF within the first 48 h of admission to the hospital. Clinical data were obtained at the day of admission. Disease severity of patients was evaluated by GAP score performed at the timing prior to an acute exacerbation of interstitial lung disease [6].
2.3. RNA extraction and reverse transcription polymerase chain reaction (RT-PCR)
Samples were centrifuged at 3000 × g at 41 °C for 30 min. Viral RNA and DNA were extracted from supernatants using the QIAamp Viral RNA Mini Kit (Qiagen, Valencia, CA, USA). Reverse transcription (RT) was performed using PrimeScript™ RT reagent Kit (Takara Bio, Otsu, Japan), according to the manufacturer's instructions. We used polymerase chain reaction (PCR) to detect various respiratory viruses, such as human metapneumovirus (HMPV), human rhinovirus (HRV), enterovirus, human respiratory syncytial virus (HRSV), influenza viruses A, B, and C, human parainfluenza viruses (HPIV), human coronavirus, adenovirus, cytomegalovirus (CMV), human herpes virus (HHV) 6, 7, and 8, human parvovirus B19, varicella zoster virus, and human bocavirus, together with Mycoplasma pneumoniae and Chlamydophila pneumoniae. The PCR products were purified using MonoFas DNA Purification Kit I (GL Sciences Inc., Shinjuku, Tokyo, Japan).
2.4. Ethical approval
Samples were collected after written informed consent was obtained from the participants or their legal representatives. The study protocol was approved by the Ethics Committee on Human Research of Kyorin University Hospital (H24-021) on July 31, 2012. The protocols were carried out in accordance with approved guidelines.
2.5. Statistical analysis
Statistical comparisons of nonparametric data were performed using the Mann-Whitney test or Wilcoxon signed-rank test. Comparisons of categorical data were performed using Pearson's chi-squared test. All tests were two-sided. A value of p < 0.05 was considered statistically significant. Data were analyzed using SPSS version 20.0 software for Windows.
3. Results
3.1. Baseline characteristics: IPF vs non-IPF ILDs group
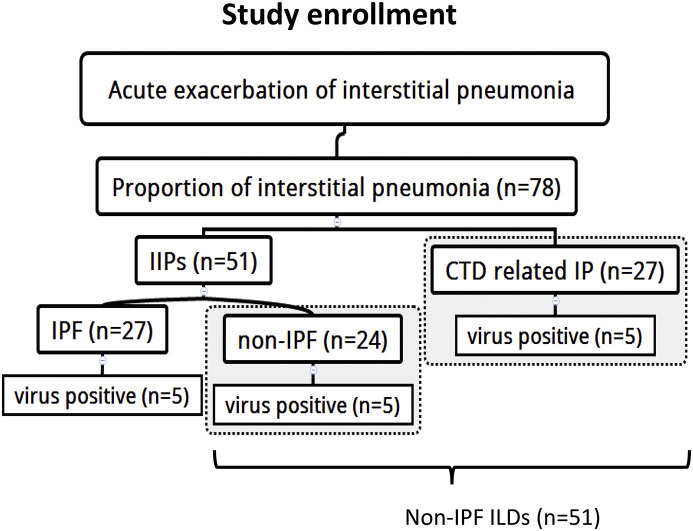
We examined a total of 78 patients with acute exacerbation of interstitial pneumonia in the study period. The number of patients with IPF was 27 and those with non-IPF ILDs was 51 (Fig. 1 ) (Table 1 ). The latter group consisted of 27 patients with IIPs without a usual interstitial pneumonia (UIP) pattern and 24 patients with CTD such as rheumatoid arthritis (n = 20), microscopic polyangitis (n = 3), scleroderma (n = 3), Sjogren's syndrome (n = 2), polymyositis (n = 1), and dermatomyositis (n = 1). The male-to-female ratio and age were comparable between the IPF (Male: n = 18, 66.7%) (median: 74 years, interquartile range[IQR] 70–84) and non-IPF ILDs group (Male: n = 28, 54.9%, p = 0.229) (median: 75years, IQR 67–80, p = 0.474), while the smoking status was significantly higher in the IPF (n = 16, 59.2%) group than in the non-IPF ILDs group (n = 16, 39%, p = 0.041). Comorbidity in the two groups was comparable, but the proportion of COPD was significantly higher in the IPF group than in the non-IPF ILDs group (25.9% vs 5.9%, p = 0.027). Regarding the thoracic high-resolution computed tomography patterns, one-quarter of the non-IPF ILDs group exhibited a UIP pattern (n = 12; 23.5%) (Table 1).
Fig. 1.
Study enrollment.
IPF: idiopathic pulmonary fibrosis.
CTD related IP: connective tissue disease related interstitial pneumonia.
Non-IPF ILDs defined as non-IPF patients (n = 24) plus CTD related IP (n = 27).
Table 1.
Characteristics of the patients.
| All patients | IPF | Non-IPF ILDs | p value | |
|---|---|---|---|---|
| Number of patients | 78 (100) | 27 | 51 | |
| Age | 74.5(68.0–82.0) | 74.0 (70.0–84.0) | 75.0 (67.0–80.0) | 0.474 |
| Male | 46 (59.0) | 18 (66.7) | 28 (54.9) | 0.229 |
| Smoking status | ||||
| Ex or Current | 32 (49.2) | 16 (59.2) | 16 (39.0) | 0.041 |
| Never | 33 (50.7) | 8 (33.3) | 25 (61.0) | 0.022 |
| Pack-years | 0 (0–40.0) | 23.5 (0–50.0) | 0 (0–20.0) | 0.007 |
| Comorbidity | ||||
| Asthma | 4 (5.1) | 1 (3.7) | 3 (5.9) | 1.0 |
| COPD | 10 (12.8) | 7 (25.9) | 3 (5.9) | 0.027 |
| Chronic heart disease | 8 (10.3) | 1 (3.7) | 7 (13.7) | 0.25 |
| Old MI or angina | 14 (17.9) | 3 (11.1) | 11 (21.6) | 0.357 |
| Diabetes mellitus | 14 (17.9) | 8 (29.6) | 6 (11.8) | 0.066 |
| Maintenance hemodialysis | 1 (1.3) | 0 (0) | 1 (2.0) | 0.346 |
| Malignancy | 11 (14.1) | 6 (22.2) | 5 (9.8) | 0.175 |
| Other lung disease | 3 (3.8) | 1 (3.7) | 2 (3.9) | 1.0 |
| Collagen vascular diseases | 26 (33.3) | 0 (0) | 26 (51) | <0.001 |
| Rheumatoid arthritis | 20 (25.6) | 0 | 20 (39.2) | |
| Microscopic polyangitis | 3 (3.8) | 0 | 3 (5.9) | |
| Scleroderma | 2 (2.6) | 0 | 2 (3.9) | |
| Sjögren's syndrome | 2 (2.6) | 0 | 2 (3.9) | |
| Polymyositis | 1 (1.3) | 0 | 1(2.0) | |
| Dermatomyositis | 1 (1.3) | 0 | 1(2.0) | |
| UIP pattern on HRCT | 39 (50) | 27 (100) | 12 (23.5) | <0.001 |
| Portion of the retrieved respiratory samples | ||||
| Sputum | 10 (12.8) | 5 (18.5) | 5 (9.8) | 0.302 |
| BALF | 5 (6.4) | 0 (0) | 5 (9.8) | 0.157 |
| Nasal swab | 63 (80.8) | 22 (81.5) | 41 (80.4) | 1.0 |
BALF, bronchoalveolar lavage fluid; COPD, chronic obstructive pulmonary disease; HRCT, high resolution computed tomography; IPF/UIP, idiopathic pulmonary fibrosis/usual interstitial pneumonia; MI, myocardial infarction; UIP, usual interstitial pneumonia.
3.2. Distribution of the detected pathogens
Among all the enrolled patients, 15 (19.2%) had respiratory viruses such as HHV7 (n = 4), CMV plus HHV7 (n = 3), Influenza virus (n = 3), HPIV (n = 2), influenza virus AH3 plus HHV7 (n = 1), CMV (n = 1), HMPV (n = 1), and HRV (n = 1) (Table 2 ). Only two cases had methicillin-susceptible Staphylococcus aureus or hemophilus influenzae, but the sputum samples could not be considered as sufficient material for evaluation. Furthermore, neither Mycoplasma pneumoniae nor Chlamydophila pneumoniae were isolated from all respiratory samples. The proportion of the isolated respiratory viruses was comparable in the IPF and non-IPF ILDs group.
Table 2.
Distribution of the detected pathogens.
| All patients (N = 78) | IPF (N = 27) | Non-IPF ILDs (N = 51) | p value | |
|---|---|---|---|---|
| Any respiratory virus (%) | 15 (19.2) | 5 (18.5) | 10 (19.6) | 0.762 |
| HHV7 (%) | 4 (5.1) | 2 (7.4) | 2 (3.9) | |
| CMV/HHV7 (%) | 3 (3.8) | 1 (3.7) | 2 (3.9) | |
| Influenza virus alone (%) | 3 (3.8) | 0 | 3 (5.9) | |
| AH3 | 2 (2.6) | 0 | 2 (3.9) | |
| H1N1 | 1 (1.3) | 0 | 1 (2.0) | |
| HPIV (%) | 2 (2.6) | 1 (3.7) | 1 (2.0) | |
| Influenza virus AH3/HHV7 | 1 (1.2) | 0 (0) | 1 (2.0) | |
| CMV (%) | 1 (1.3) | 1 (3.7) | 0 (0) | |
| HMPV (%) | 1 (1.3) | 0 (0) | 1 (2.0) | |
| HRV (%) | 0 (0) | 0 (0) | 0 (0) | |
| Adenovirus (%) | 0 (0) | 0 (0) | 0 (0) | |
| Enterovirus (%) | 0 (0) | 0 (0) | 0 (0) | |
| Coronavirus (%) | 0 (0) | 0 (0) | ||
| Parbovirus B19 (%) | 0 (0) | 0 (0) | 0 (0) | |
| Bocavirus (%) | 0 (0) | 0 (0) | 0 (0) | |
| Varicella zoster virus (%) | 0 (0) | 0 (0) | 0 (0) |
CMV, cytomegalovirus; HMPV, human metapneumovirus; HPIV, human parainfluenza virus; HRV, human rhinovirus; RSV, respiratory syncytial virus.
3.3. Comparison of the clinical findings between IPF and non-IPF ILDs
The proportion of respiratory failure in the IPF and non-IPF ILDs group was similar (77.8% vs 76.5%) on admission (Table 3 ). However, the respiratory rate was significantly higher in the IPF group (median 29 breaths/min, [IQR] 24–34) than in the non-IPF ILDs group (median 22 breaths/min, IQR 20–30, p = 0.034; Table 3). Additionally, the number of patients with home oxygen therapy was significantly higher in the IPF group than in the non-IPF ILDs group (33.3% vs 9.8%, p = 0.011), and the time of initial onset to admission was significantly shorter in the IPF group (median 5.0, IQR 3–7 days) than in the non-IPF ILDs group (median 8.0 days, IQR 4–14, p = 0.019). The initial treatment in each group did not differ, and the mortality rate at 14 days, 30 days, and 60 days were comparable. The serum laboratory data on admission were comparable between the IPF and non-IPF ILDs group, but the value of KL-6, a serum marker for alveolar injury, was significantly higher in the IPF (median 1203 U/mL, IQR 798–2006) than in the non-IPF ILDs group (median 704.5 U/mL, IQR 437–1,189, p = 0.003).
Table 3.
Comparison of the clinical findings between IPF and non-IPF ILDs cases.
| All patients (N = 78) | IPF (N = 27) | Non-IPF ILDs (N = 51) | p value | |
|---|---|---|---|---|
| Respiratory failure | 60 (76.9) | 21 (77.8) | 39 (76.5) | 1.0 |
| Body temperature (°C)* | 37.0 (36.4–37.9) | 37.1 (36.8–37.5) | 36.8 (36.3–38.0) | 0.190 |
| Respiratory rate (breaths/min)*# | 24.0 (20.0–32.0) | 29.0 (24.0–34.0) | 22.0 (20.0–30.0) | 0.034* |
| Systolic blood pressure (mmHg)* | 123.5 (110.0–138.0) | 125.5 (120.0–145.5) | 119.0 (102.0–135.0) | 0.083 |
| Heart rate (beats/min)* | 100 (84.8–119.3) | 106.0 (87.0–119.5) | 94.0 (84.5–118.0) | 0.207 |
| Mortality within 14 days# | 11 (14.3) | 5 (18.5) | 6 (12.0) | 1.0 |
| Mortality within 30 days# | 13 (16.9) | 6 (22.2) | 7 (14.0) | 0.752 |
| Mortality within 60 days# | 16 (21.3) | 8 (32.0) | 8 (16.0) | 0.365 |
| Home oxygen therapy at first visit | 14 (17.9) | 9 (33.3) | 5 (9.8) | 0.011 |
| Initial onset to admission (days)* | 7.0 (3.8–14.0) | 5.0 (3.0–7.0) | 8.0 (4.0–14.0) | 0.019* |
| Admission to death*# | 10.0 (3.8–34.8) | 5.0 (3.0–28.5) | 12.5 (8.0–41.7) | 0.525 |
| Undertreatment with antifibrotic agent | 3 (3.8) | 3 (11.1) | 0 (0) | 0.034* |
| Initial treatment | ||||
| CS oral only | 5 (6.4) | 2 (7.4) | 4 (7.8) | 1.0 |
| CS pulse | 62 (79.5) | 21(77.8) | 41(80.4) | 1.0 |
| IVCY | 13 (16.7) | 4 (14.8) | 9 (17.6) | 1.0 |
| CS (pulse and/or oral)+CsA/CY | 8 (10.2) | 4 (14.8) | 4 (7.8) | 0.430 |
| Laboratory data | ||||
| WBC (106 cells/mL)* | 10,500 (7600–13,600) | 11,400 (8400–18,560) | 9100 (7100–12,800) | 0.106 |
| CRP (mg/dL)* | 9.0 (3.2–15.4) | 12.0 (3.7–18.9) | 7.4 (3.0–13.9) | 0.292 |
| LDH* (IU/L) | 340 (258.0–444.5) | 358.0 (296.0–433.0) | 330.5 (254.8–456.3) | 0.996 |
| BNP*(pg/mL) | 113 (35.2–310.5) | 69.8 (26.3–314.0) | 118.5 (46.2–285.5) | 0.413 |
| Procalcitonin* (ng/mL) | 0.13 (0.08–0.32) | 0.12 (0.09–0.20) | 0.14 (0.08–0.72) | 0.404 |
| KL-6* (U/mL) | 864.0 (530.5–1545.5) | 1203 (798.0–2006.0) | 704.5 (437.0–1189.0) | 0.003* |
| SP-D* (ng/mL) | 192.0 (109.3–356.0) | 201.0 (118.0–332.0) | 180.0 (107.0–383.5) | 0.759 |
Data are expressed as number (percentage) unless otherwise stated. *Data are expressed as median (interquartile range). # Data are expressed as number of deaths/N (percentage) and missing date was excluded.
CS, corticosteroid; CRP, C-reactive protein; CsA, Cyclosporine; CY, Cyclophosphamide; IPF/UIP, idiopathic pulmonary fibrosis/usual interstitial pneumonia; IVCY, intravenous cyclophosphamide; WBC, white blood cell count; NS, not significant.
3.4. Sixty-day mortality rate associated with virus infections and other factors
The Kaplan-Meier survival curves over 60 days demonstrated that the virus negative group (n = 60) exhibited a significantly higher survival probability (83.3%) than did the virus positive group (n = 15, 60%, p < 0.05) on the log-rank test (Fig. 2 A). However, the virus infection itself could not predict the poor 60-day survival probability on the simple logistic regression analysis between the two groups. Similarly, when focusing on the IPF group, the 60-day survival probability in the virus negative patients (n = 22) and positive patients (n = 5) was significantly different (77.3% vs 40%, p = 0.05; Fig. 2B), but the virus infection itself could not predict the poor 60-day survival probability on the simple logistic regression analysis. Comparison of the 60-day survival probability between the IPF (n = 27) and non-IPF ILDs groups (n = 51) was comparable (p = 0.132; Fig. 2C). Interestingly, the survival probability was significantly lower in the UIP group (n = 39) than in the non-UIP group (n = 39, p = 0.022; Fig. 2D); however, the simple logistic regression analysis did not predict the 60-day poor survival rate in patients with UIP pattern. In addition, the presence of COPD, home oxygen therapy, and smoking status could not predict the 60-day mortality rate.
Fig. 2.
Comparison of the 60-day survival probabilities on Kaplan Meier plot in the two groups divided by various factors.
All data were evaluated by the log-rank test.
3.5. Comparison of GAP index in IPF, non-IPF, and CTD associated IP
Among all enrolled patients, few patients performed 6 min walk tests (6MWT), and the data of pulmonary function test were available only in 40.7% (n = 11) of IPF, 29.2% (n = 7) of non-IPF, and 33.3% (n = 9) of CTD associated IP. In the perspective of disease severity, comparison of the GAP score between IPF and non-IPF ILDs showed that the IPF was significantly higher score (median 5, IQR: 5–6, p = 0.001) than that of non-IPF ILDs (median 1, IQR: 0.25–2.0). Among the non-IPF ILDs, the GAP score of non-UIP and CTD-associated disease was comparable (median 1, IQR 0–3 vs median 1, IQR: 0.5–2.0, p = 0.685). Comparison between IPF and CTD-associated disease showed that the IPF had significantly higher score than in the CTD associated IP (p < 0.001). Further analysis demonstrated that high GAP score itself did not predict the poor 60-day survival probability on the simple logistic regression analysis.
4. Discussion
The present study clearly demonstrated that respiratory virus infections can be seen in 19.2% of patients with acute exacerbation of interstitial pneumonia. The proportion of virus isolation did not differ between the IPF and non-IPF ILDs groups. Additionally, the virus-positive patients displayed a lower 60-day survival probability, as assessed using the Kaplan-Meier test, but the virus infection itself could not predict the 60-day mortality by simple logistic regression analysis.
On the other hand, our results raised further clinical questions in understanding the role of virus infections in acute exacerbation of interstitial pneumonia because most of the isolated respiratory viruses consisted of HHV7, CMV, or HHV7/CMV co-detection. Tang et al. [2] reported that 97% of stable patients with IPF had one or more herpes viruses (CMV, EBV, HHV7, and HHV8) in their biopsied specimens, which might have contributed to the chronic irritation for antigenic stimulation. Indeed, regardless of the type of interstitial pneumonia, CMV/HHV7 accounted for up to 46.7% (n = 7) of the total isolated respiratory viruses (n = 15) in our study. This has raised concerns about whether or not those viruses truly reflected the current infection or hallmark of latent infection/re-activation. This hypothesis for latent infection or re-activation was reinforced by our results demonstrating (1) no elevation of serum atypical lymphocytes, (2) no definite evidence of inclusion body in the BALF, and (3) absence of pp65 CMV antigenemia in due clinical course. Ushiki et al. [3] reported on 14 cases of IPF in Japan acute exacerbation and found evidence of RSV in one case and CMV in two cases. In Brazil, Santos et al. [7] demonstrated that 10% of IPF acute exacerbation cases exhibited measles or CMV infection Thus, the role of virus infections both in IPF and non-IPF ILDs cases remains obscure; further studies with a higher number of these cases may be required in the future.
Other respiratory viruses such as influenza viruses, HPIV, and HRV have been reported to cause lower respiratory symptoms and/or community-acquired pneumonia in adults [8] Recently, we reported the prevalence of viral infections in adult asthma attack patients conducted in the same period of this study that the frequency of HRV and RSV infections seemed to be high in autumn, while HMPV was likely to occur in spring, and influenza virus infections were most common in winter and spring [9]. However, regarding with acute exacerbation of interstitial pneumonia, the detected pathogens differ from those of asthma attack patients [9], and it seems that respiratory samples whose acute exacerbation of interstitial pneumonia cannot be influenced by respiratory tract viral infections. The present study was conducted throughout the period of 3 years; thereby, the results can be considered as representative data from a single tertiary referral center in Japan that was not influenced by seasonal variations.
As evidenced by the serum data on admission, the procalcitonin levels were low (Table 3), which indicated that the exacerbated respiratory status would be associated with virus infections but not bacteria, if the infection can be considered as a cause of acute exacerbation of interstitial pneumonia.
There are three main limitations to our study. First, the study lacked data on the torque teno virus in BALF, which has been previously suggested as a possible cause of acute exacerbation of IPF [4]. Second, the paucity of sampling data from BALF is another limitation. However, the present study reported the first evidence of viral respiratory infections in both IPF and non-IPF ILDs cases with acute exacerbation that required hospital admission. Further accumulation of such cases may be required in future studies to elucidate that the virus detection either indicates latent infection and/or re-activation in the respiratory systems or is a true trigger of acute exacerbation. Thirdly, regarding with disease severity, few patients performed 6MWT, and the GAP score could not predict the 60-day mortality on single logistic regression analysis, but it should be evaluated in further study.
In conclusion, we suggest that viral infections, mostly CMV and HHV7, were detected from patients with IPF and non-IPF ILDs with acute exacerbation, but the clinical significance of the results remains to be determined.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure of conflict of interests
The authors state that they have no conflict of interest.
Acknowledgments
None.
References
- 1.Collard H.R., Moore B.B., Flaherty K.R., Brown K.K., Kaner R.J., King T.E., Jr., Lasky J.A., Loyd J.E., Noth I., Olman M.A., Raghu G., Roman J., Ryu J.H., Zisman D.A., Hunninghake G.W., Colby T.V., Egan J.J., Hansell D.M., Johkoh T., Kaminski N., Kim D.S., Kondoh Y., Lynch D.A., Muller-Quernheim J., Myers J.L., Nicholson A.G., Selman M., Toews G.B., Wells A.U., Martinez F.J. Idiopathic pulmonary fibrosis clinical research network, acute exacerbations of idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2007;176:636–643. doi: 10.1164/rccm.200703-463PP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tang Y.W., Johnson J.E., Browning P.J., Cruz-Gervis R.A., Davis A., Graham B.S., Brigham K.L., Oates J.A., Jr., Loyd J.E., Stecenko A.A. Herpesvirus DNA is consistently detected in lungs of patients with idiopathic pulmonary fibrosis. J. Clin. Microbiol. 2003;41:2633–2640. doi: 10.1128/JCM.41.6.2633-2640.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ushiki A., Yamazaki Y., Hama M., Yasuo M., Hanaoka M., Kubo K. Viral infections in patients with an acute exacerbation of idiopathic interstitial pneumonia. Respir. Investig. 2014;52:65–70. doi: 10.1016/j.resinv.2013.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wootton S.C., Kim D.S., Kondoh Y., Chen E., Lee J.S., Song J.W., Huh J.W., Taniguchi H., Chiu C., Boushey H., Lancaster L.H., Wolters P.J., DeRisi J., Ganem D., Collard H.R. Viral infection in acute exacerbation of idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2011;183:1698–1702. doi: 10.1164/rccm.201010-1752OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Collard H.R., Ryerson C.J., Corte T.J., Jenkins G., Kondoh Y., Lederer D.J., Lee J.S., Maher T.M., Wells A.U., Antoniou K.M., Behr J., Brown K.K., Cottin V., Flaherty K.R., Fukuoka J., Hansell D.M., Johkoh T., Kaminski N., Kim D.S., Kolb M., Lynch D.A., Myers J.L., Raghu G., Richeldi L., Taniguchi H., Martinez F.J. Acute exacerbation of idiopathic pulmonary fibrosis. An international working group report. Am. J. Respir. Crit. Care Med. 2016;194:265–275. doi: 10.1164/rccm.201604-0801CI. [DOI] [PubMed] [Google Scholar]
- 6.Ryerson C.J., Vittinghoff E., Ley B., Lee J.S., Mooney J.J., Jones K.D., Elicker B.M., Wolters P.J., Koth L.L., King T.E., Jr., Collard H.R. Predicting survival across chronic interstitial lung disease: the ILD-GAP model. Chest. 2014;145:723–728. doi: 10.1378/chest.13-1474. [DOI] [PubMed] [Google Scholar]
- 7.Santos G.C., Parra E.R., Stegun F.W., Cirqueira C.S., Capelozzi V.L. Immunohistochemical detection of virus through its nuclear cytopathic effect in idiopathic interstitial pneumonia other than acute exacerbation. Braz. J. Med. Biol. Res. 2013;46:985–992. doi: 10.1590/1414-431X20132885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kurai D., Sasaki Y., Saraya T., Ishii H., Tsukagoshi H., Kozawa K., Ryo A., Ishioka T., Kuroda M., Oishi K., Takizawa H., Kimura H. Pathogen profiles and molecular epidemiology of respiratory viruses in Japanese inpatients with community-acquired pneumonia. Respir. Investig. 2016;54:255–263. doi: 10.1016/j.resinv.2016.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Saraya T., Kimura H., Kurai D., Ishii H., Takizawa H. The molecular epidemiology of respiratory viruses associated with asthma attacks: a single-center observational study in Japan. Medicine (Baltim.) 2017;96 doi: 10.1097/MD.0000000000008204. [DOI] [PMC free article] [PubMed] [Google Scholar]