Abstract
A wide variety of disorders affecting the intestinal tract in cattle may require surgery. Among those disorders the more common are: intestinal volvulus, jejunal hemorrhage syndrome and more recently the duodenal sigmoid flexure volvulus. Although general principles of intestinal surgery can be applied, cattle has anatomical and behavior particularities that must be known before invading the abdomen. This article focuses on surgical techniques used to optimize outcomes and discusses specific disorders of small intestine. Diagnoses and surgical techniques presented can be applied in field conditions.
Keywords: Cattle, Surgery, Intestine, Jejunum, Colon, Enterectomy
Key points
-
•
The short mesentery in cattle makes intestinal exteriorization challenging.
-
•
Duodenal sigmoid flexure volvulus is suspected if there is extreme hypochloremic alkalosis.
-
•
Cattle with jejunal hemorrhage syndrome (JHS) have a poor prognosis.
-
•
Intraoperative clot fragmentation should be favored with JHS rather than enterectomy.
-
•
The prognosis is better for animals with ileal flange volvulus rather than mesentery root torsion.
Intestinal surgery in cattle is challenging because it is often done with the animal standing under sedation. Because of the abdominal pain and state of shock, affected animals will be prone to move constantly or even lay down. Moreover, it is technically demanding and can rarely be performed without assistance, unlike most common surgeries in cattle. Decision-making is crucial and appropriate technical technique is essential to improve success rate.
Decision-making
Doing an exploratory laparotomy in cattle is easy, fast, and inexpensive. There are less consequences if revealed to be unnecessary compared to other species. However, an unnecessary laparotomy could be harmful to a critical patient that could have been medically treated. Before making the decision to favor a surgical or medical approach, information must be gathered and the case approached in a logical manner: (1) review the possible causes of abdominal pain relevant to the particular case; (2) recognize the indications for immediate surgery; (3) determine if surgery is an option given the cost, facilities, and surgical abilities; (4) establish medical treatment before or during surgery; (5) if surgery is postponed, determine in advance a precise time and list criteria to be monitored to help decision-making; and (6) establish and present to the client a most realistic prognosis and cost estimate. A systematic approach based on adequate signalment and history, complete physical examination, and judicious choice of ancillary tests are the tools available to clinicians.
Frequent clinical signs and physical findings include colic, distended abdomen, tachycardia, and shock. A very distended abdomen requires rapid assistance. A stomach tube should be passed to decompress the rumen. If no free gas is coming out of the tube, it might be a frothy bloat or intestinal volvulus. Abnormal findings at rectal palpation are presence of fresh blood, absence of feces, tension band, distended small bowel, distended cecum, or a mass among small bowels. Ileus and peracute enteritis can be easily confused with jejunal obstruction. Usually, the small bowel will be distended primarily with fluids and easily palpable per rectum. Tension bands may also be palpated. In volvulus, intestines are severely distended with gas occupying the pelvic cavity. Intussusception is difficult to identify precisely but usually is suspected if a hard mass is felt among distended small bowels. Jejunal hemorrhage syndrome (JHS) might have a similar rectal examination finding except that the rectum is filled with ripe raspberry-like feces in variable quantity. Animals with enteritis may have a fever and show other clinical signs or laboratory results compatible with it (ie, leukopenia). Finally, the heart rate is a good indicator of the severity of the condition but should be correlated with other findings. A cow with mildly distended small bowel and a heart rate of 80 beats per minute (bpm) does not need immediate assistance. However, a cow with a distended organ and heart rate of 100 bpm and dehydrated needs immediate assistance.
Ultrasound imaging is very helpful to evaluate the abdomen in cattle.1, 2 Presence of severely distended and empty intestines at the ultrasound examination indicates obstruction (Fig. 1 ). Surgery is indicated even if the cause if not clearly identified at this point.
Fig. 1.
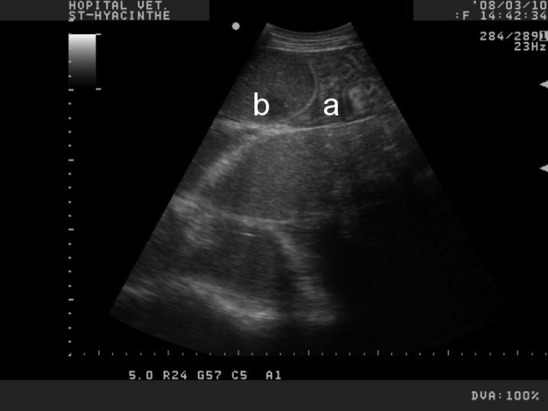
Transabdominal ultrasound of the ventral right abdomen with a curvilinear probe on an adult Holstein cow. The jejunal loops are distended, occupying most of the ultrasound field (b). The presence of empty jejunal loops (a) adjacent to distended loops is compatible with intestinal obstruction.
Preoperative considerations
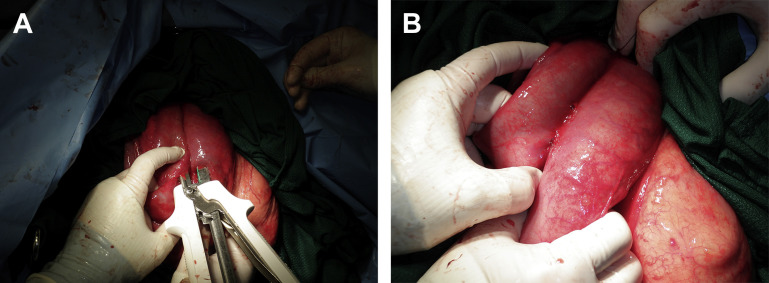
The surgeon must decide if the procedure will be performed standing, or on lateral or sternal recumbency. If the animal is down, the decision is easy. If the animal is standing, the decision is often based on surgeon’s training and experience. If the animal is weak, the surgery is performed with the animal in left lateral recumbency. Sternal recumbency is an option but the animal must be secured to prevent it from standing during the surgery (Fig. 2 ). Doing the surgery on a secured recumbent animal has the main advantage of doing the manipulation without worrying if the animal lies down, kicks, or moves constantly while suturing the intestines. However, securing an animal on lateral recumbency is difficult in a field setting. Moreover, exteriorization of bowels is more difficult on a laterally recumbent animal.
Fig. 2.
This adult Holstein cow was unable to stand because of electrolytes imbalance. Intestinal obstruction was suspected. The animals is secured in sternal recumbency and surgically prepared for a right flank laparotomy.
Preoperative analgesia is provided to the patient before invading the abdomen. Flunixin meglumin, ketoprofen, butorphanol, meloxicam, or ketamine stun3 is frequently used for standing gastrointestinal (GI) surgery in cattle. If the surgery is performed standing, sedation must be used cautiously so the animal will not lie down. The authors rarely use sedation unless the behavior of the animal is aggressive or will prevent safe manipulation of the bowels. Broad spectrum antibiotics must be given before the surgery and repeated during the procedure if it lasts longer than 2 hours. Regional block or paravertebral block is preferred, allowing the surgeon to extend the incision at his or her convenience. If the animal is in shock and there is electrolytes imbalance, fluids are given before and during the procedure. Blood transfusion must be considered in some severe cases of JHS. Avoid any high-volume oral fluids if the abdomen is distended.
Principles of bovine intestinal surgery
The cranial part of the descending duodenum courses cranially from the pylorus to the ansa sigmoidea duodenalis medial to the liver. The descending duodenum then courses caudally, wraps around the omental curtain, and turns cranially to the ascending duodenum. This portion of the duodenum courses cranially to the root of the mesentery medial to the omentum and joins the jejunum. The entire duodenum is contained by 2 mesenteries: mesoduodenum and omentum. The jejunum is restricted by a short mesentery except for the distal third of the jejunum and proximal segment of the ileum, which are suspended by a long mesenteric segment often referred to as the jejunoileal flange. This segment can be easily exteriorized from the abdomen but the remainder of the jejunum and ileum are poorly exteriorized because of the short mesentery. Although the demeanor of cattle allows for standing paralumbar fossa laparotomy, excessive tension on the mesentery will likely stimulate the patient to lie down and interfere with surgical procedures.
Enterectomy
A cow has a short mesentery, therefore, traction on it is painful and the animal may go down at any moment. This short mesentery precludes adequate exteriorization of some segments of the small bowel. Only the portion to be resected should be exteriorized to avoid excessive traction and contamination during the resection-anastomosis (Fig. 3 ). Infiltration of 2% lidocaine into the mesentery where resection is planned may decrease the pain of traction. The mesenteric vessels (arteries and veins) are ligated using mass ligation with absorbable suture material (United States Pharmacopeia [USP] 3 chromic gut, USP 1 polyglactin 910), being sure not to compromise the blood supply to the intestine to be preserved. Mass ligation is required because cattle do not have a well-defined arcuate vascular anatomy (as do horses) and the fatty mesentery renders vessel identification impossible.4 The sutures are placed in an overlapping pattern such that double ligation of the vessels is accomplished (Fig. 4A). This technique may be performed rapidly. However, it is impossible to effectively ligate all the vessels. It will be completed on complete resection of the intestinal segment. Stapling instrumentation is highly unreliable for occlusion of mesenteric vessels because of the large amount of fat normally found in the intestinal mesentery of cattle. Ligasure (Covidien, Medtronics, Minneapolis, MN, USA) is an electrocautery device that will seal vessels through a specially designed forceps. It seals vessels up to 7 mm in diameter. It is efficient and safe but expensive to use. Moreover, the machine is not meant to be used in a field situation.
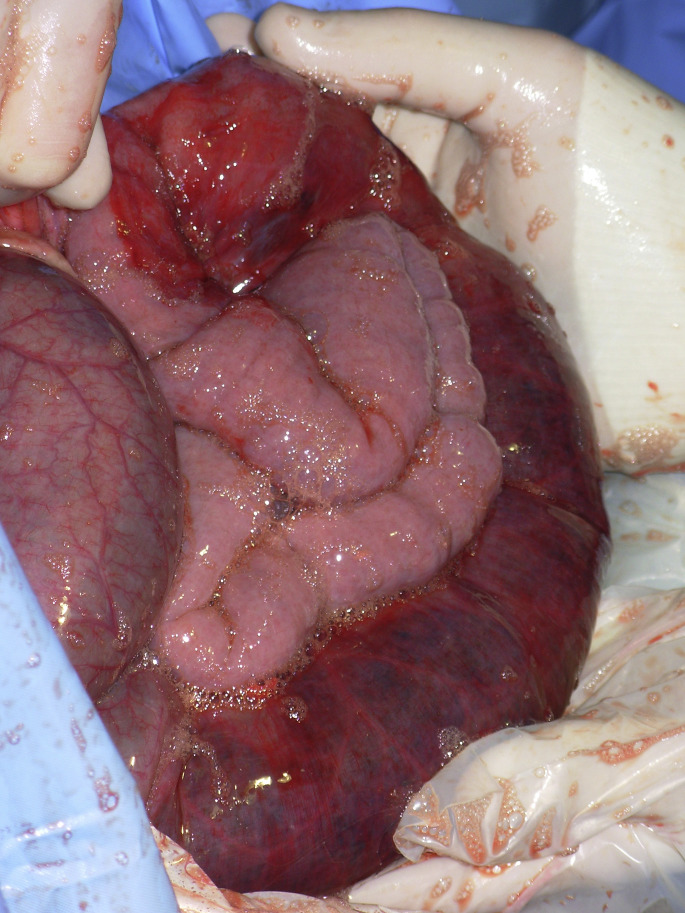
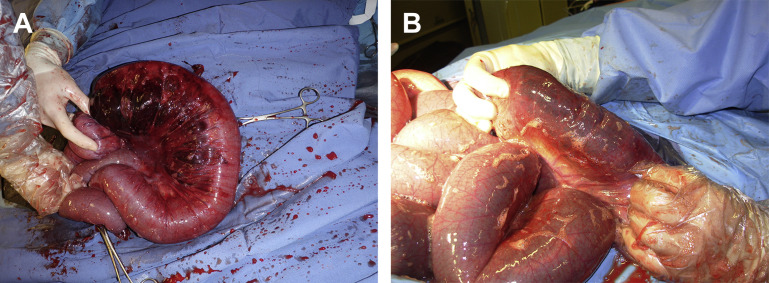
Fig. 3.
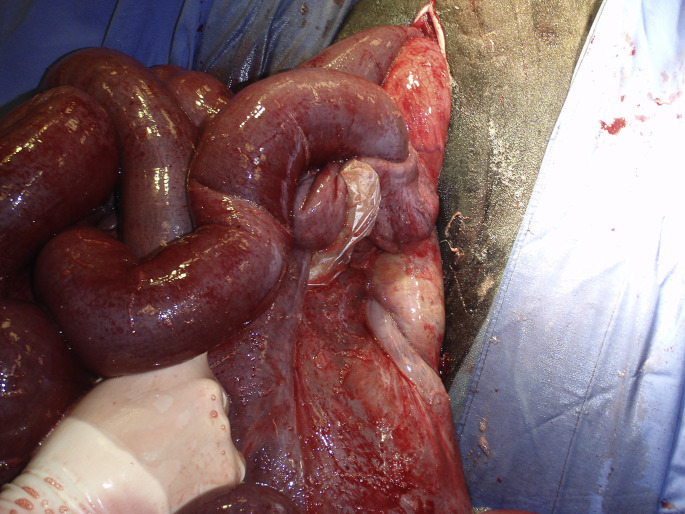
Loops of jejunum are exteriorized through a right flank laparotomy. Pink empty loops are adjacent to an obstructed reddish segment of jejunum. The obstruction was caused by a large hematoma typically found in the JHS.
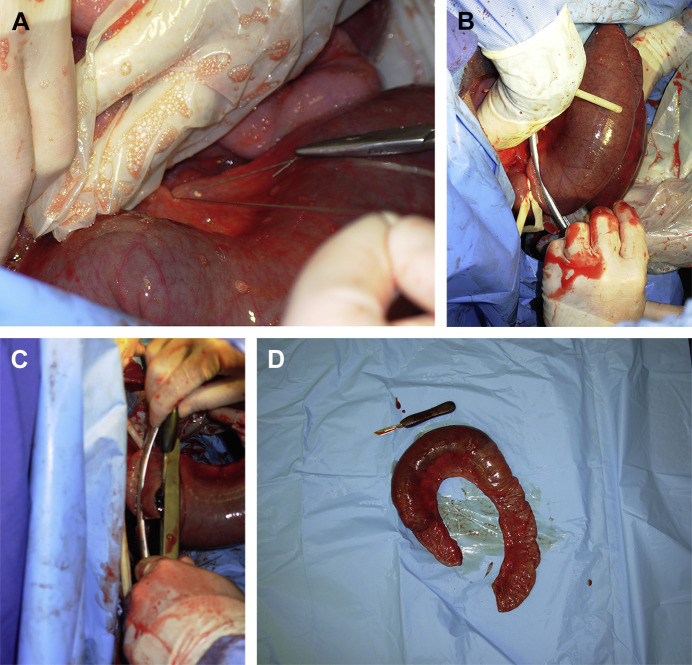
Fig. 4.
(A) Ligatures are blindly placed in the fatty mesentery to decrease excessive bleeding during intestinal resection. (B) A Penrose drain and a Doyen forceps are placed on the sound portion of the jejunum. (C) A second Doyen forceps is placed on the resected portion. The resection is performed by incising between the 2 Doyen forceps. (D) The resected loop of jejunum and the hematoma within.
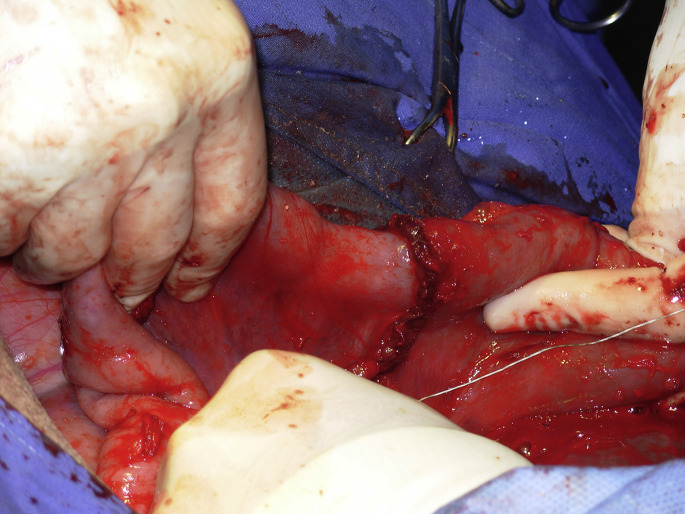
After completion of mesentery ligation and transection, Doyen intestinal forceps are used to occlude the lumen of the normal and abnormal bowel (Fig. 4B–D). When exteriorization allows it, thirty centimeters of healthy bowel adjacent to the affected portion are also resected. The proximal segment of bowel is carefully exteriorized to its maximum length and the Doyen forceps are removed. Distended orad intestine can be decompressed through the enterectomy site with special care to avoid contamination of the incision or abdomen with ingesta. This procedure will lessen the severity of postoperative ileus and shorten convalescence. The 2 segments of intestine are reunited by end-to-end or side-to-side anastomosis with an absorbable suture material (USP 2–0 for adult, USP 3–0 to 4–0 for calves with polyglactin 910 or polydioxanone) using a single or 2-layer suture pattern.5 A simple full-thickness, continuous suture provides better apposition and less lumen diameter reduction (Fig. 5 ). The anastomosis is performed in 2 to 3 overlapping simple continuous suture lines, each placed in one-half of one-third of the circumference, so that a purse-string effect is not created. A 2-layer technique consists of a simple continuous suture involving the mucosa and submucosa, followed by a Cushing or Lembert pattern. The initial suture line should be placed at the mesenteric attachment because this is the most likely site for leakage to occur. Some surgeons prefer using a full-thickness, simple interrupted suture when segments to anastomose are of significantly different diameter (Fig. 6 ). After the anastomosis, the mesentery is sutured in a continuous pattern with a USP 0 absorbable suture material (see Fig. 6). The affected intestine is thoroughly washed with sterile isotonic fluids, checked for the presence of leakage, and replaced into the abdomen. Carboxymethylcellulose gel can be applied over the anastomosis site to prevent adhesions.
Fig. 5.
Two overlapping full-thickness simple continuous sutures (180° each) were used for the anastomosis.
Fig. 6.
Simple interrupted sutures were favored by the surgeon to do the anastomosis. The mesentery is sutured in a simple continuous manner.
Enterotomy
A simple enterotomy will allow removal of a foreign body into the lumen or to take a full-thickness biopsy at a specific area of the GI tract. Two suturing techniques for closure of a jejunal enterotomy were compared in cattle. Although the stapling technique was faster (TA 55 with 3.5 mm staples), enterotomy suture with 1-layer interrupted Gambee technique resulted in fewer adhesions and thickening of the intestinal wall. At 13-weeks postsurgery, the lumen diameter reduction was similar between both suture techniques.6 As previously described, a simple continuous suture followed by a Lembert or Cushing will provide a watertight suture with minimal lumen reduction.
General guideline for postoperative management
All intestinal surgery is classified as clean-contaminated surgery and some are clearly infected because of a peritonitis. The antibiotics regimen will vary according to this classification. The authors usually recommend 5 days of broad spectrum antibiotics after any intestinal resection. Antibiotics can be continued for 2 weeks if there is a peritonitis. Dehydration and electrolytes imbalances should be corrected rapidly with intravenous (IV) fluids. Any oral fluids administration must be avoided until the animal is passing feces.
Pain management is often neglected postoperatively in cattle. Ileus is caused by electrolytes imbalance and pain, and will delay normal recovery. A variety of nonsteroidal antiinflammatory drugs (NSAIDs) are available depending on the countries. They are frequently used to control pain after abdominal surgeries in cattle. To the authors’ knowledge, there are no publications on the effect of NSAIDs on intestinal motility. However, preoperative flunixin (2.2 mg/kg) has improved postoperative recovery after an omentopexy.7 In the authors’ clinics, it is routine procedure to administer NSAIDs preoperatively and the day after. It should be repeated cautiously on anorexic animals because of potential abomasal ulceration. Butorphanol is opioid with less effect on the abomasum. Although expensive, it can be given at a dosage of 0.05 mg/kg 3 times a day. Lidocaine drip has been used in horses to treat postoperative ileus. The authors use a bolus of 1.3 mg/kg of lidocaine at a rate of 0.05 mg/kg per minute.8
Duodenum Outflow Problems
The first portion of the duodenum is the cranial part leaving the pylorus going toward the liver cranially and dorsally. It forms a cranial sigmoid flexure at the level of the gall bladder and continues caudally to the descending duodenum. At the caudal aspect of the greater omentum, it forms the caudal flexure while running cranially and medially to the ascending duodenum. Exteriorisation of the duodenum is limited to the cranial and descending duodenum. The duodenum can be affected in many ways, from simple ileus to perforation.9, 10
Any intervention on the duodenum is difficult because only the oral part of the cranial duodenum and a portion of the descending duodenum can be exteriorized. Three types of obstruction are reported: functional, intraluminal or extraluminal, and strangulation. Phytobezoar or trichobezoar, and foreign bodies, were reported to cause intraluminal obstruction, whereas adhesions and liver abscesses may cause extraluminal obstruction.9, 10, 11, 12, 13, 14, 15 Strangulation of the caudal flexure of the duodenum was reported in 3 cows during late pregnancy because the uterus had passed through a rent in the mesoduodenum.16, 17 A duodenal obstruction caused by a malposition of the gallbladder was also reported.18
Clinical Signs
The clinical signs are variable depending on the origin of obstruction. If there is no strangulation of the duodenum, the onset of clinical signs is slow with progressive distension of the ventral abdomen. This distension is caused by the enlargement of the abomasum and the rumen. Those animals are usually severely dehydrated, with an elevated heart rate, and transabdominal rectal palpation is unrewarding except from the lack of feces. A small ping behind the last right ribs has been identified in volvulus of the sigmoid flexure.19 Ultrasound examination specifies which segment of the duodenum is obstructed and locates the origin of the obstruction. The cranial duodenum is often severely distended (8 cm of diameter) with an enlarged abomasum but the rest of the GI tract is empty.
Clinical Pathologic Findings
Duodenal obstruction should be suspected if there is a severe hypochloremic hypokalemic metabolic alkalosis: serum chloride of 70 mmol/L (normal around 100 mmol/L), total serum carbon dioxide of 44 mmol/L (normal 30 mmol/L), and serum potassium of 3 mmol/L (normal of 4–5 mmol/L).5, 9, 19, 20 Depending on the location of the obstruction, liver enzymes may also be increased.18, 19 Chloride ruminal contents are elevated (>30 mmol/L).20
Surgery
If the cause cannot be determined, a temporary diagnosis of ileus is given and a large volume of IV fluids is administered for 12 to 24 hours. Depending on the dehydration, up to 40 to 60 L of physiologic saline are administered intravenously. Hypokalemia is treated with 100 to 200 g or oral potassium chloride. The animal is observed by monitoring the heart rate, fecal output, and GI motility with ultrasound.
Duodenal problems are approached through a standard right laparotomy. The length of the incision may vary from 20 to 40 cm depending on the procedure. A simple enterotomy can be performed on the cranial and descending duodenum to remove a foreign body (Fig. 7 ) or trichobezoar. However, any obstruction involving the sigmoid flexure of the duodenum is difficult because it cannot be exteriorized. A laterolateral bypass between the cranial and descending duodenum is the only alternative (Fig. 8 ).19, 20
Fig. 7.
Gravels and a 14-gauge hypodermic needle were recovered through an enterotomy of the duodenum on an adult Holstein cow.
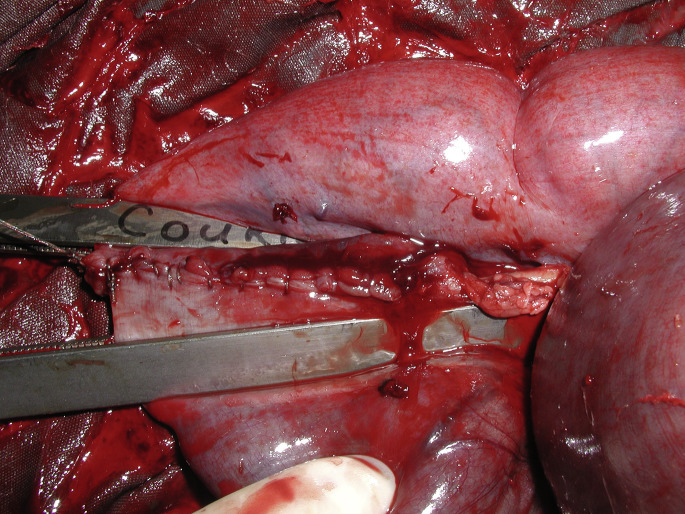
Fig. 8.
(A) Surgical stapling (Gastrointestinal anastomosis, Medtronic, Minneapolis, MN) is being used to create a laterolateral bypass between the cranial and descending duodenum. (B) The duodenoduodenal bypass is finished and ready to be replaced in the abdomen.
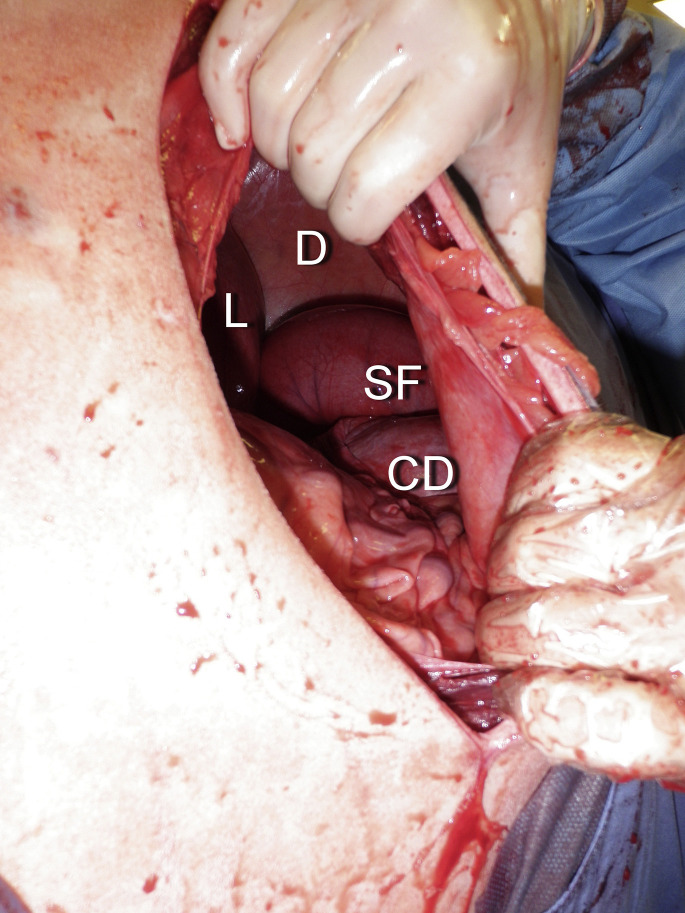
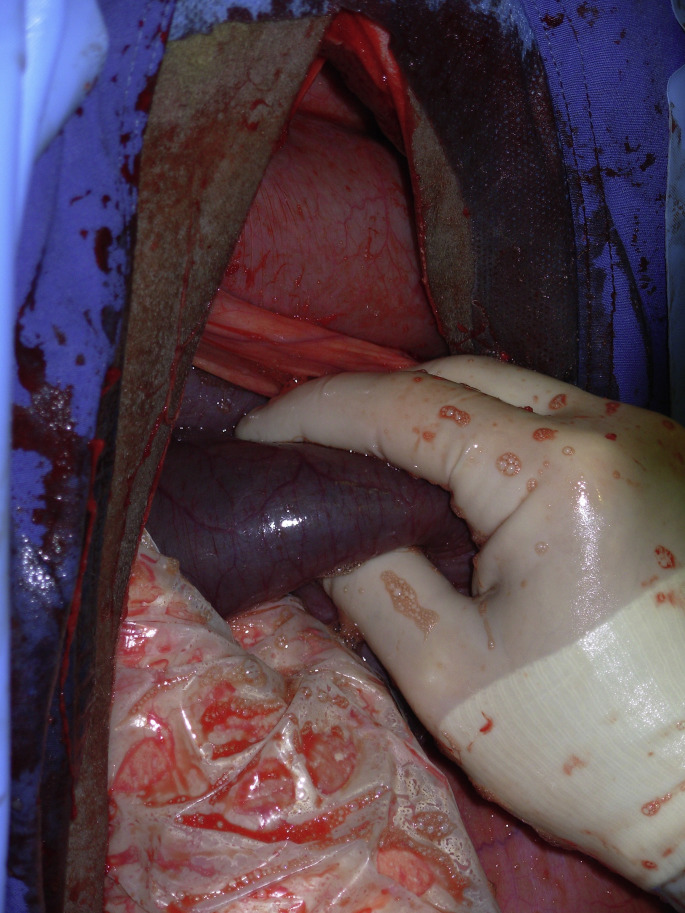
Recently, a new condition was described as a volvulus of the sigmoid flexure of the duodenum.19 The origin of the condition is unknown. A similar presentation was reported by van der Velden in the 1980s.20 Asynchronous motility of the cranial duodenum was suspected. This is a surgical condition and diagnosis is confirmed during the laparotomy. Surgical findings are an enlarged abomasum and cranial duodenum. The sigmoid flexure is severely distended with gas and a volvulus can be palpated at its root, close to the neck of the gall bladder, on the visceral part of the liver (Fig. 9 ). The volvulus is reduced and cranial duodenal content is milked through the flexure to ensure that it is functional. If the viability of the sigmoid flexure is in doubt, a laterolateral duodenal bypass must be performed between the cranial duodenum and the descending duodenum.19 The prognosis for any duodenal obstruction is generally good after cause and electrolytes imbalance are corrected.
Fig. 9.
Volvulus of the sigmoid flexure of the duodenum identified through a right flank incision on a standing adult cow. CD, cranial duodenum; D, diaphragm; L, liver; SF, sigmoid flexure.
Intussusception
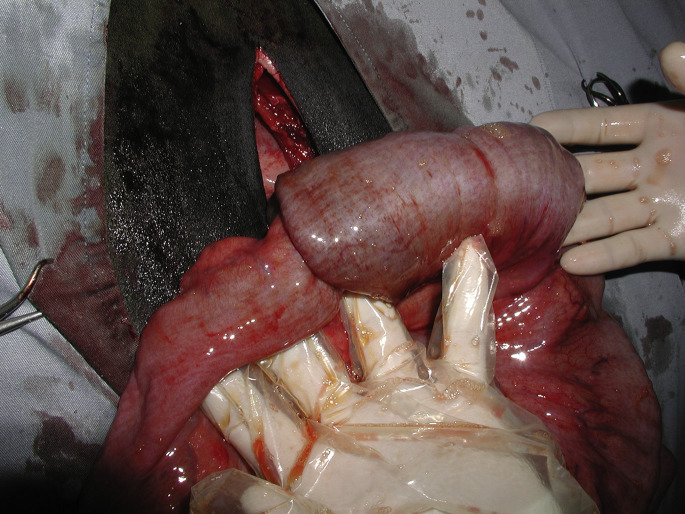
Intussusception refers to the invagination of 1 segment of intestine into an adjacent segment of intestine. The invaginated portion of intestine is termed the intussusceptum, and the outer, or receiving, segment of intestine is termed the intussuscipiens (Fig. 10 ). Intussusception occurs sporadically in cattle of all ages, breeds, and gender and may be seen at any time during the year.21, 22 However, in a case-control epidemiologic study of 336 cattle, intussusception occurred most commonly in calves less than 2 months old, Brown Swiss cattle seemed to be overrepresented and Hereford cattle seemed to be underrepresented compared with Holstein cattle.21 Although the inciting cause is rarely identified, intussusception may occur secondary to enteritis, intestinal parasitism, sudden changes in diet, mural granuloma, or abscess (Fig. 11 ), intestinal neoplasia (especially adenocarcinoma), mural hematoma, heavy work load,23 and administration of drugs that affect intestinal motility.24, 25, 26, 27, 28 Any focal disturbance of intestinal motility may facilitate the invagination of an orad segment into an aborad segment of intestine. Intussusception involving the caecum was reported in 51 calves, of which 40 had diarrhea.29 Intussusception occurs most commonly in the distal portion of the jejunum but intussusception has been found affecting the proximal jejunum, ileum, cecum, and spiral colon.21, 24, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37 In a review of 336 intussusceptions in cattle, 281 affected the small intestine, 7 were ileocolic, 12 were cecocolic, and 36 were colocolic.21
Fig. 10.
A jejunal intussusception is exteriorized through a right flank incision on a standing adult cow.
Fig. 11.
Intussusception was reduced by gently pulling on the segments. However, a granuloma is present and the segment must be resected.
Clinical Signs
Cattle affected with intussusception demonstrate clinical signs of abdominal pain (restlessness, kicking at the abdomen, lying down, getting up frequently, and assuming abnormal posture) for up to 24 hours after the onset of disease. Cattle are frequently anorectic, lethargic, and reluctant to walk. After the initial signs of abdominal pain subside, affected cattle become progressively lethargic, recumbent, and show apparent depression. Abdominal distention becomes apparent after 24 to 48 hours duration. This is caused by gas and fluid distention of the forestomach and intestines, and sequestration of ingesta within the gastrointestinal tract results in progressive dehydration and electrolyte depletion. Heart rate will increase proportionally to abdominal pain, intestinal necrosis, and dehydration. Fecal production may be normal for up to 12 hours after the occurrence of the intussusception but minimal fecal production is noted after 24 hours duration. Passage of dark scant feces or only mucus is often observed with intussusception.
Clinical Pathologic Findings
Hemoconcentration is usually present (increased packed cell volume and total proteins), and an inflammatory leukogram may be seen if ischemic necrosis of the intussusceptum has occurred. Often, changes in the white blood cell count and differential are minimal, and changes in peritoneal fluid constituents are not seen because the intussusceptum is isolated by the intussuscipiens. Hypochloremic, metabolic alkalosis is found with serum biochemistry analysis. Hyponatremia, hypokalemia, hypocalcemia, azotemia, and hyperglycemia also may be found. The magnitude of these changes depends on the location and duration of the lesion. Increased blood urea nitrogen (BUN) can be observed the more orad the intussusception. Proximal jejunal intussusception causes rapid and severe dehydration, electrolyte sequestration, and metabolic alkalosis. Most lesions occur in the distal jejunum and may require more than 48 hours to develop these changes. Elevation of rumen chloride concentration (>30 mEq/l) may be found if fluid distention of the rumen is present.
Diagnosis
Diagnosis of intussusception is usually confirmed during exploratory laparotomy. The intussusception can be felt as a hard sausage-like structure during rectal palpation34 but distention of multiple loops of small intestine is most commonly identified. These rectal palpation findings are similar to those found in JHS. In the authors’ experiences, an intussusception may be present for 48 hours or more in adult cattle without intestinal distention being found during rectal palpation. It depends on its location and the severity of the obstruction. Some animals will still pass a certain amount of feces. In calves and small ruminants, percutaneous palpation and ultrasonographic examination of the abdomen may be used to identify intestinal distention and, possibly, the intussusception.29, 38 Transabdominal and rectal ultrasound can also be used in adult cattle with the appropriate equipment. The intussusception has a typical target-like image on ultrasound with distended orad bowels and adjacent free fluid.33, 39 It should be suspected in cattle with a history of abdominal pain and abdominal distention, scant feces consisting of blood and mucus, and palpable distention of the intestine. Differential diagnoses include JHS, functional ileus, trichobezoar, foreign bodies, intestinal incarceration or strangulation, vagal syndrome, intestinal neoplasia, fat necrosis, and jejunoileal flange volvulus. Abomasal ulcers might be included in the differential if dark feces are observed.
Treatment
Affected cattle must be stabilized before surgical intervention is performed. Fluid therapy should be aimed at replacing fluid and electrolyte deficits. Surgical correction may proceed after the patient has been assessed as a suitable candidate. Most of these animals will have some degree of peritonitis. Broad spectrum antibiotics against gram-negative or gram-positive, as well as anaerobic, bacteria must be administered preoperatively. The regimen can be adjusted postoperatively related to surgical findings. Intra-abdominal surgical manipulations are painful demand appropriate preoperative analgesia. NSAIDs are given preoperatively, along with butorphanol (0.05 mg/kg S.C.). Right paralumbar fossa exploratory laparotomy is the surgical approach of choice for treatment of intussusception. Most small intestines of cattle have a short mesentery, preventing adequate exteriorization of the intussusception through a ventral midline incision. Also, the attachments of the greater omentum limit exposure with this approach. Most often, diagnostic exploratory laparotomy is performed with the cow standing after regional anesthesia. Tension on the mesentery of the small intestine results in pain and cattle may attempt to lie down during the procedure. Of 35 cattle having standing, right paralumbar fossa laparotomy for resection of intussusception, 14% became recumbent and 26% attempted to become recumbent during the surgery.21 Preoperative planning should include anticipation of this possibility. When intussusception is suspected and the animal is of high perceived economic value, right paralumbar fossa celiotomy may be performed with the patient under general anesthesia and in left lateral recumbency. The intussusception may be more difficult to elevate through the incision in recumbent cattle because the fluid-filled bowel gravitates away from the surgical site. However, isolation and resection of the intussusception can be done without risk of the animal lying down during the procedure and with minimal risk of contamination of the abdomen.
Surgical removal by resection and anastomosis is the treatment of choice for intussusception. The intussusception is exteriorized from the abdomen and isolated using a barrier drape and moistened towels. Manual reduction of the intussusception is not recommended because of the risk for rupture of the intestine during manipulation, probable ischemic necrosis of the intestine after surgery, possible reoccurrence of the intussusception, and prolonged ileus caused by motility disturbance and swelling in the affected segment of bowel. However, if the intussusception is in the spiral colon, resection and anastomosis in this area is difficult. Manual reduction only can be successful.32 Successful resection of a segment of the proximal loop of the ascending colon is reported.40 The affected segment was resected and a side-to-side anastomosis was performed between the base of the caecum and the spiral colon.
Postoperative management should be directed to prevent dehydration, maintain optimal blood electrolyte concentration, control for infection and inflammation, and stimulate appetite. IV fluids are beneficial during the first 24 hours after surgery. The authors routinely perform rumen transfaunation 12 to 24 hours after surgery to stimulate forestomach motility and appetite. Large volume of oral drenching must avoided until normal output of feces is observed. Food should not be withheld after surgery. Administration of butorphanol tartrate (0.02–0.04 mg/kg, IV) may help with pain-induced ileus by providing mild visceral analgesia without direct adverse effects on intestinal motility. Lidocaine drip for the first 24 to 48 hours must be considered to improve gut motility and control pain.8
Prognosis
The prognosis for return to productivity after surgical correction of intussusception is variable and depends somewhat on the duration of the lesion and its location. Cattle respond favorably to surgery if operated on within 48 hours of the onset of the disease. Cattle presenting with severe dehydration (>12%), tachycardia (heart rate>120 bpm), severe decrease in serum chloride concentration (Cl) less than 80 mEq/l, and severe abdominal distention are considered to have a poor prognosis for survival. If viscera rupture is present at the time of surgery, the prognosis is grave. In a case-control, multicenter study in which surgery was attempted, 85 of 143 cattle with small intestinal intussusception, 0 of 4 with ileocolic, 10 of 11 with cecocolic, and 10 of 20 with colocolic were discharged from the hospital.21 Overall, 31% of the animals diagnosed with intussusception were discharged from the hospital. In a case series, only 4 out of 20 animals diagnosed with jejunal intussusception were discharged from the hospital.28 However, many animals were presented in an unfavorable clinical state demanding immediate euthanasia. Eleven underwent surgery and 4 were discharged 8 days after the surgery.28 The extent and long duration of the condition before presentation negatively influenced the outcome.28 In another case series on 12 adult bullocks, all survived right flank resection and anastomosis of jejunum or ileal intussusception. They passed feces within 24 hours.23 Early recurrence has been reported in a calf.38
Hemorrhagic jejunal syndrome
This syndrome has been recognized in adult dairy cows involving intraluminal and intramural intestinal hemorrhage, necrosis with subsequent clot formation, and intestinal obstruction. It is referred to as JHS, hemorrhagic bowel syndrome, intraluminal-intramural hematoma, bloody gut, or hemorrhagic enteritis.41, 42, 43, 44
In the search for the cause of this disease, much speculation has fallen on Clostridium perfringens type A.41, 42 C perfringens type A was suspected as a cause or contributing factor in this disease because this organism has been cultured from the intestinal contents or feces of a large number of cows affected with JHS.42, 45, 46, 47 However, evidence regarding pathogenicity of these isolates has not been produced that links C perfringens type A to development of JHS. Moreover, C perfringens type A is also found in the gastrointestinal tract of cattle without causing clinically apparent disease.48, 49
At present, it is thought that JHS is a multifactorial disease.41, 42, 46 Proposed risk factors include feeding of silage or total mixed ration (TMR), feeding finely ground corn in the ration, early stage of lactation, high level of production, and free-choice feeding. These factors may create an environment in which stress, excess gastrointestinal starch, and subclinical rumen acidosis allow overgrowth of C perfringens type A bacteria in the gastrointestinal tract. C perfringens type A produce α-toxin, and some may produce a beta2 toxin.50, 51 In a retrospective study on 37 cattle, C perfringens type A was isolated of all the 20 samples (feces, blood clot, intestine).47 A subsample of 10 was further analyzed for toxins in which α-toxin was isolated in all and beta2 toxin in 1 sample.47 Excess grain or starch in the small intestine is more likely to lead to sudden bacterial overgrowth.41, 42, 48, 49, 52 This passage of grain particles into the small intestine may be a factor in the pathogenesis of JHS in dairy cows fed high grain TMR. Excess mixing of TMR causing finely powdered corn was suspected to be related to an outbreak of clinical cases of JHS on 1 farm.42
Aspergillus fumigatus has been incriminated as the primary agent of JHS. A fumigatus is a fungus that sporulates abundantly causing no harm in immunocompetent animals. It was isolated from affected animals but not from normal animals. Moreover, A fumigatus seems to cause a similar disease in humans.53 A gliotoxin was detected in animals infected with A fumigatus. It is hypothesized that the gliotoxin can potentiate A fumigatus.52
Clinical Signs
The physical presentation is variable depending on the onset of clinical signs. The condition has been reported in beef and dairy breeds, with a predilection for Holstein and Brown Swiss.46, 47, 54, 55, 56 Brown Swiss were significantly overrepresented in 1 study.56 Clinical signs of this peracute disease include recumbency, dehydration, shock, abdominal distension, anorexia, abdominal pain, and lack of feces or the production of tarry feces with blood clots. In many cases, the cow is found dead. Affected cows are usually in early lactation, producing large quantities of milk, and are fed silage or TMR.
The most common clinical findings are: hemorrhagic diarrhea with clots of dark blood or absence of feces, colic, tachycardia, white mucosa, right ventral distension, rumen atony, lack of intestinal motility, and variable right-sided ping.46, 47, 56, 57 Abdominal examination per rectum may reveal distended jejunum, depending on the location of the blood clot. A mass can also be palpated in some animals.
Clinical Pathologic Findings
Results are variable depending on the stage and severity of the disease. On hematology, a left-shift leukogram is often observed. Hematocrit and total protein are usually within normal limit even though a significant amount of blood may have been lost.46, 47, 54 The most significant changes are on the chemistry profile. Typically, the affected animals will have a hypochloremic, hypokalemic, metabolic alkalosis accompanied by an elevated BUN (mean = 12.6 mmol/L, n = 1.67–6.51 mmol/L) with a normal creatinine and an elevated glucose (mean = 11.7 mmol/L, n = 2.6–4.9).46, 47, 52 Liver enzymes (Succinate dehydrogenase, Lactate dehydrogenase, Gamma-glutamyl transferase) can also be increased because of the absorption of toxins from intestinal damage.46
Diagnostics
The history, the clinical findings and laboratory results are usually enough to confirm the diagnosis. The differential should include winter dysenteriae (coronavirus), intussusception, and abomasal ulcers. Transabdominal ultrasound examination of the intestinal tract is helpful to confirm the diagnosis but even more helpful to find an obstruction. Braun and colleagues55 performed ultrasound on 63 cows with hemorrhagic bowel syndrome (HBS). Their findings included fluid and fibrin between jejunal loops, increased jejunal diameter (mean: 6.76 cm, n = 2–4 cm)1 with postobstruction empty loops, absence of motility, heterogeneous hyperechoic intraluminal material corresponding to blood clots, and abomasal distension.55 Similar findings were reported by other investigators (Fig. 12 ).46, 47
Fig. 12.
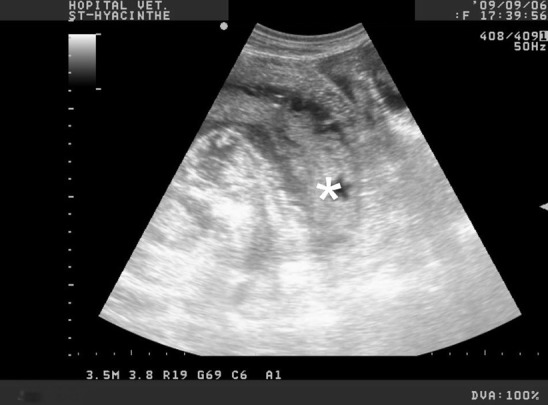
Transabdominal ultrasound of the right ventral abdomen with a curvilinear abdominal probe on an adult Holstein cow. The animal was later diagnosed with JHS. The intraluminal hematoma in the jejunum (asterisk).
Treatment
Before any surgical intervention, the animal’s hemodynamic status and electrolytes imbalance must be stabilized. Large-volume (20–40L) isotonic solution or blood transfusion (6–12L) can be administered before surgery. Hypokalemia is a common finding. Some of these animals are recumbent before or after the surgery with a S-shaped neck which is typical of the hypokalemic syndrome in cattle. It can certainly worsen the ileus. However, the surgery cannot be delayed because it may takes a few days to correct the hypokalemia.
Medical treatment is achieved if the animal is still passing feces and there are no signs of obstruction at the ultrasound. Specific treatment of the condition includes oral administration of mineral oil (4L Semel in die up to 3 days) and intramuscular or IV B-lactam. Nonsteroidal anti-inflammatory drugs are routinely given to control visceral pain. IV slow lidocaine infusion has been used in cattle to control visceral pain and improve GI motility. It is part of the authors’ medical treatment protocol and postoperative treatment.8 If the animal stops passing feces or an obstruction is suspected at the palpation per rectum or transabdominal ultrasound examination, surgery is elected.
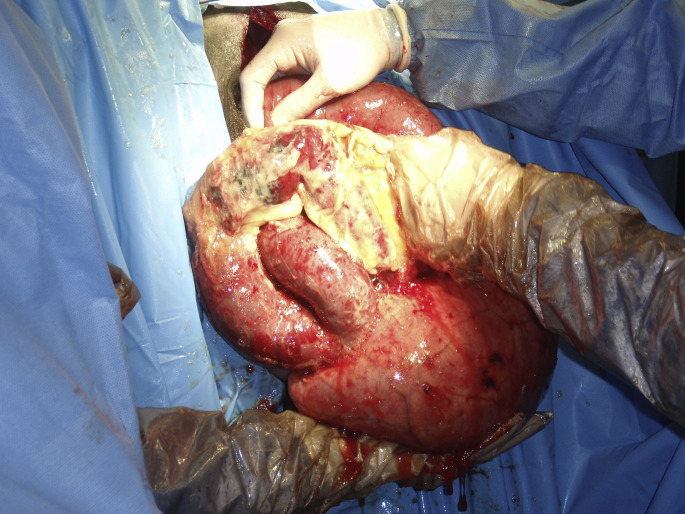
Through a right paralumbar fossa approach, the affected segment of jejunum is easily identified. A firm, distended, intrajejunal mass of variable length will obstruct the lumen with orad jejunal distension (see Fig. 3). There are 3 surgical options: aborad fragmentation and massage of the clot, enterotomy, and resection anastomosis. The decision is based on the length of the clot and the integrity of the jejunal wall (Fig. 13 ). Prompt laparotomy and manual massage must be favored, especially in field conditions (Fig. 14 ).
Fig. 13.
JHS in an adult cow. (A) The exteriorized segment is compromised and must be resected. (B) The bluish color of the affected segment is from the intraluminal hematoma. The serosa and the mesentery are relatively sound. Therefore, manual breakage and progressive massage of the hematoma must be attempted to relieve the obstruction.
Fig. 14.
Manual massage of the hematoma secondary to a JHS.
Enterotomy is rarely done by itself because the clot is adhered to the submucosa over a significant length (20–30 cm). If manual massage failed or the affected segment is disrupted, then resection-anastomosis must be performed as described previously. The intestine diameter discrepancy between preobstruction and postobstruction can be considerable. The enterectomy site must be of equal diameter before achieving the anastomosis. To achieve this, the angle of the jejunal incision may vary between the preobstruction and postobstruction segment. Any restriction at the anastomosis site will prevent orad blood clot to pass through and increase recurrence (Fig. 15 ). A laterolateral anastomosis should be considered if the surgeon is concerned about the expected anastomosis diameter.
Fig. 15.
Second laparotomy following a recurrence of obstruction of a JHS 72 hours after a jejunal resection anastomosis. Fibrin is covering distended loops of jejunum which are already adhered.
Postoperative Care and Prognosis
Fluids or blood are usually continued until the animal is passing feces in large amount or to correct previous blood analysis abnormalities. Antibiotics (B-lactam) are given for at least 5 days. Depending on contamination during the procedure or the presence of peritonitis, it can be combined with an antibiotic effective against gram-negative bacteria. Mineral oil will be administered daily postoperatively until the animal is passing feces. NSAIDs are given at least 24 hours after the procedure and can be prolonged if needed. IV lidocaine drip will also be given for 24 to 48 hours to improve motility. In general, the animal will pass feces 24 to 36 hours after the procedure.
The prognosis for survival is fair and recurrence frequent. In a study on 11 beef and dairy cattle, exploratory laparotomy was performed on 7 animals but treatment was not pursued because of economic restriction or severity of the condition. Ten were euthanized and 1 died.54 Twenty-two dairy cows form the same herd were diagnosed and treated for HBS.46 Seven out of 8 animals treated medically died. Of the 13 that underwent surgery, 4 were euthanized because of the extent of the disease. Manual clot fragmentation, enterotomy, and resection anastomosis was attempted on the remaining 9 but only 5 survived for an overall survival rate of 23%.46 In another study, medical treatment was successful in 2 out 5 animals and 18 out of 30 animals survived after a surgical procedure; resection anastomosis (14 out of 20), clot fractionation (4 out of 18), and intraoperative euthanasia (2).47 Peek and colleagues56 described the surgical findings and outcomes of 31 dairy cattle. Eighteen of the 31 (58%) that were surgically treated were discharged. Recurrence of JHS was observed on the 5 animals that died within a year. According to the investigators, clot fractionation was associated with a better prognosis than enterotomy or resection anastomosis.56 In another study on 65 cows with JHS that underwent right-flank laparotomy, 23 survived.57 All these results clearly show that prognosis is fair to grave depending on the severity of the disease. Although resection anastomosis seems appealing to resolve the problem, it is clearly not a guarantee of success with a significant chance of recurrence.
Intestinal volvulus
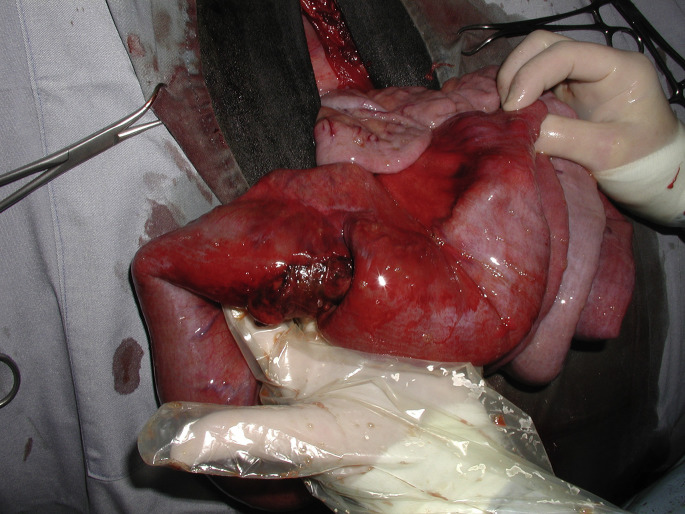
Volvulus refers to the rotation of viscera around its mesenteric attachment. Torsion refers to the rotation of viscera around its own (or long) axis. Although torsion of the abomasum and uterus are found in cattle, torsion of the small intestine is rare. Small intestinal volvulus may occur in different forms.58, 59, 60 The most severe forms of intestinal volvulus originate from the root of the mesentery and involve the entirety of the small intestine and mesenteries (Fig. 16 ). Volvulus of the root of the mesentery causes obstruction of venous outflow and arterial blood supply to the intestines. Ischemic necrosis of the intestine occurs rapidly, which causes metabolic acidosis, shock, and death. Volvulus of the jejunoileal flange refers to volvulus of the mid-to-distal jejunum and proximal ileum in which the mesentery is long (Fig. 17 ). This long mesentery and associated bowel has been termed the flange and it may rotate about its mesentery without involving the remaining small intestine. Often, arterial occlusion is not found with volvulus of the jejunoileal flange, possibly because extensive fat deposits within the mesentery may prevent compression of the muscular wall of the arteries until the volvulus becomes severe. However, obstruction of outflow of venous blood may be equally detrimental because of mural edema, shunting of blood away from the mucosa, and progressive ischemia.
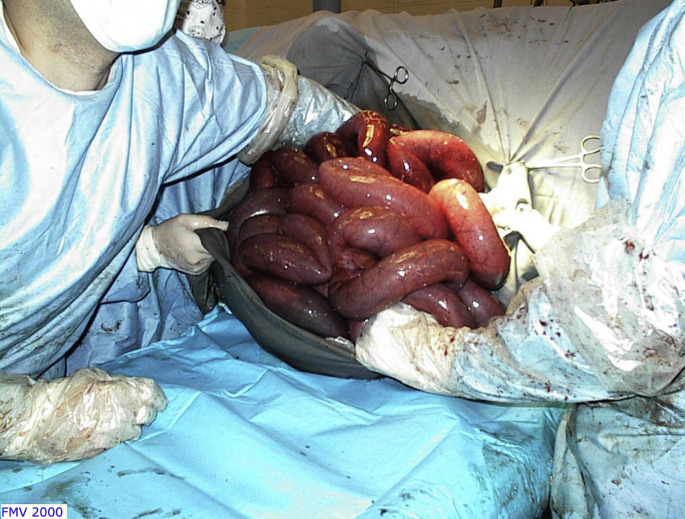
Fig. 16.
Jejunal volvulus through a right flank laparotomy. All the loops of the jejunum are severely distended with gas because of the mesenteric root torsion. It is exteriorized to allow intraabdominal manipulation and reduction of the intestinal volvulus.
Fig. 17.
Volvulus of the jejunoileal flange. Only a portion of the jejunum is distended and the its long mesentery predisposing to volvulus can be observed.
Cattle of any breed, age, or sex may be affected by intestinal volvulus at any time during the year. In a review of 190 cattle having intestinal volvulus, dairy breeds were at a higher risk of developing volvulus compared with beef breeds.58 This difference was thought to be associated with differences in management. Neither lactation nor gestation was identified as a risk factor, and calves were not found to be at an increased risk compared with adult cattle. In a separate study of 100 cattle having intestinal volvulus, 86 were calves between 1 week and 6 months old.61
Clinical Signs
Cattle having volvulus of the root of the mesentery may be found dead with severe abdominal distention. Early in the course of the disease, affected cattle demonstrate acute, severe abdominal pain (kicking at the abdomen, rolling, lying down and getting up frequently, grunting) and have marked elevation in heart rate (>120 bpm) and respiratory rate (>80 bpm). The rapid progression of the disease precludes development of significant dehydration but cardiovascular shock is usually present.
Cattle having volvulus of the jejunoileal flange may present similarly to cattle having volvulus at the root of the mesentery. However, these cattle often demonstrate clinical signs consistent with acute intestinal obstruction rather than cardiovascular shock. Cattle show signs of abdominal pain, are tachycardic (80–120 bpm), and pass minimal feces. Cattle may be dehydrated at the time of examination.
Clinical Pathologic Findings
Because of the rapid onset and progress, cattle having intestinal volvulus may not demonstrate change in serum biochemistry or hematology data. The changes expected with intestinal volvulus are consistent with intestinal obstruction, stress, and dehydration, including azotemia, hypocalcemia, hyperglycemia, and a leukocytosis with a mild left shift.58 In the early stages of the disease, cattle develop alkalemia with normal serum potassium concentration. As cardiovascular compromise and intestinal ischemia proceed, cattle develop metabolic acidosis and hyperkalemia. Cattle having the shift to acidosis and hyperkalemia have a poor prognosis for survival.58
Diagnosis
Diagnosis of intestinal volvulus is by exploratory laparotomy. Rectal palpation reveals multiple loops of distended intestine filling the caudal abdomen and excessive tension on the intestinal mesentery. Simultaneous auscultation and percussion of the abdomen yields multifocal pings of variable pitch and location. Findings of scant feces, abdominal pain, sudden onset of abdominal distention, and multiple loops of distended intestine on rectal palpation in cattle are highly suggestive of intestinal volvulus. Differential diagnoses include intussusception, cecal volvulus, abomasal volvulus, intraluminal obstruction, and severe indigestion.
Treatment
Immediate surgical correction is the treatment of choice. IV fluids should be administered to treat cardiovascular shock but preparation for surgery should not be delayed. The volvulus must be corrected before irreversible ischemic injury or thrombosis of the mesenteric arteries has occurred. A right paralumbar fossa laparotomy with the cow standing is the approach of choice. Restoration of normal anatomic position of the intestines is more easily done with the patient standing. Cattle that are thought to be at great risk of becoming recumbent during surgery should be placed under general anesthesia, in left lateral recumbency, and the laparotomy performed through the right paralumbar fossa. The presence of the volvulus and the direction of the twist are assessed by palpating the root of the mesentery and, in the case of jejunoileal flange volvulus, following this ventrally to the location of the twist. The intestinal mass is gently derotated, being careful not to cause rupture of the viscera. This procedure may require exteriorization of various portions of the intestinal mass. After correction of the volvulus, the intestinal tract should be examined for evidence of nonviable bowel. If the intestine is compromised (arterial thrombosis, blackened serosa, friable wall of the affected segment, mural edema), then intestinal resection and anastomosis is indicated (see previous discussion of intussusception). Also, exploration of the abdomen should be done to rule out the presence of a second lesion (eg, abomasal displacement, fecalith, intussusception, anomalous fibrovascular bands, and peritonitis).
Postoperative management is directed toward maintaining optimal hydration, electrolyte, and acid-base status. Antibiotics and anti-inflammatory drugs are indicated. Ileus may be seen during the first 48 hours after surgery but the use of prokinetic drugs should be weighed against the risk of leakage at the site of the anastomosis if intestinal resection was performed. Passage of large volumes of diarrhea within 24 hours after surgery is considered to be a favorable prognostic indicator.
Prognosis
Prognosis varies with the severity and duration of the lesion. Prognosis for survival for cattle having volvulus of the root of the mesentery (44%) is less than for volvulus of the jejunoileal flange (86%).58 Overall, dairy cattle have a better prognosis for survival (63%) than beef cattle (22%). This difference is presumed to be because dairy cattle are observed more frequently and, therefore, treatment sought earlier in the progression of the disease. Of 92 cattle in which surgical correction of intestinal volvulus was attempted, 13 were euthanatized during surgery, 25 died within 24 hours after surgery, 13 died between 2 and 7 days, and 41 (45%) survived.61
Trichobezoars
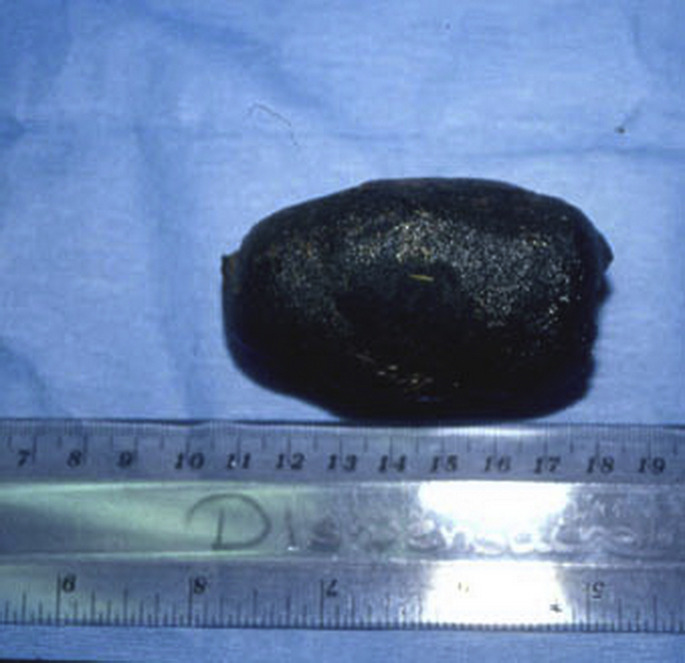
Intraluminal obstruction of the intestinal tract of cattle, sheep, and goats is most commonly caused by a trichobezoar, phytobezoar, or enterolith.62, 63 These foreign bodies form in the rumen or abomasum and may pass into the intestinal tract where they become lodged within the small intestine or spiral colon. Hairballs (trichobezoar) are caused by frequent ingestion of hair. This is seen most commonly in cattle infested with lice or mange, or during the spring when shedding of the winter hair coat occurs. Phytobezoars and enteroliths form around undigested materials (eg, nylon fibers, cotton fabric). In a necropsy survey of 166 dead calves less than 90-days-old in Western Canada, 56 calves died because of perforation of an abomasal ulcer.64 Calves having an abomasal ulcer were 2.74 times more likely to have an abomasal hairball. Calves less than 31 days old and having an abomasal ulcer were 3.81 times more likely to have an abomasal hairball. However, the investigators were unable to establish a causative relationship between the presence of abomasal hairballs and a perforating ulcer. During a study of confined cattle being fed a roughage-limited diet, cows began biting hair from each other’s hair coats and developed multiple ruminal hairballs (2–10 hairballs weighing 0.2–3.8 kg each).65 The investigators speculated that the cows began grazing hair because of the lack of roughage in the diet, boredom, and high stocking density. One report describes clinical findings in 2 sheep having 107 individual hairballs.66 The investigators speculated that pruritus or some unknown dietary deficiency was the cause of excessive ingestion of the wool.
Clinical Signs
Animals affected with ruminal or abomasal bezoars may be observed to have decreased appetite, weight loss, decreased fecal production, lethargy, and apparent depression. Multiple bezoars present in the rumen or abomasum of calves, sheep, and goats may be found during transabdominal palpation or on abdominal radiographs. When an obstruction of the small intestine or spiral colon occurs, affected animals initially show clinical signs of abdominal pain (restlessness, kicking at the abdomen, lying down and getting up frequently, arching the back, and stretching out of the legs while standing) and progress to recumbency and apparent depression. Progressive bloat or abdominal distention and lack of fecal production are noted.
Clinical Pathologic Findings
Serum biochemistry analysis reveals hypokalemic, hypochloremic, metabolic alkalosis, the severity of which depends on the duration and location of the lesion. These changes are most severe with proximal intestinal obstruction and become more severe with increasing duration. If ischemic necrosis of the intestinal wall has occurred, an inflammatory leukogram with increased numbers of immature neutrophils may be seen. As peritonitis develops and organic acids are released into the blood stream, the serum biochemistry changes to a metabolic acidosis with relative hyperkalemia. These changes are consistent with a poor prognosis. Perforation of an abomasal ulcer or rupture of the intestine and contamination of the abdomen with ingesta carries a poor to grave prognosis.
Diagnosis
In affected cattle, serum biochemistry changes are consistent with intestinal obstruction. Rumen chloride concentration may be elevated (rumen Cl > 30 mEq/l). The cause of intraluminal obstruction is rarely palpable per rectum but small intestinal distention may be palpable. Ultrasonographic examination of the abdomen may be useful in calves and small ruminants. Intraluminal intestinal obstruction should be suspected in cattle with recurrent rumen tympanites, which is transiently responsive to decompression and is associated with minimal fecal production. Differential diagnoses include intussusception, vagus indigestion syndrome, intestinal lymphosarcoma, fat necrosis, intestinal entrapment around anomalous fibrovascular bands, and volvulus of the jejunoileal flange.
Treatment
Trichobezoars, phytobezoars, or enteroliths located within the rumen are unlikely to cause clinical signs unless the number and magnitude of the foreign bodies is severe (eg, 2 sheep in which hairballs accounted for >10% of the animals’ body weight).66 A cow suffered esophageal obstruction after suspected attempted regurgitation of a rumen trichobezoar.67 Ruminal foreign bodies are removed via a left paralumbar fossa celiotomy and rumenotomy. Abomasal hairballs may cause pyloric obstruction, which leads to rapid onset of abdominal distention. The authors prefer to perform a right paramedian or ventral paracostal laparotomy to exteriorize the abomasum. An abomasotomy is performed along the greater curvature of the abomasum, the foreign bodies removed, and the abomasum is closed with absorbable synthetic suture material (eg, 0 polydioxanone, polyglycolic 910) using 2 layers of an inverting suture pattern. When obstruction of the duodenum, jejunum, or spiral colon is suspected, a right paralumbar fossa celiotomy and exploration of the abdomen should be performed. The foreign body is found by exteriorizing a segment of normal or distended intestine and tracing this segment oral or aboral, respectively, until the obstruction is found. This segment of intestine is exteriorized from the abdomen, isolated using moistened surgical towels, and an enterotomy performed (Fig. 18 ). After removal of the foreign body, the enterotomy is closed with absorbable suture material (eg, 2–0 polydioxanone, polyglactin 910) using 2 lines of an inverting suture pattern. The enterotomy may be closed transversely to maximize the lumen of the affected segment of intestine and minimize the tension endured by the suture line during contraction of the intestinal wall. When the perceived economic value of the affected cow is high, surgery may be performed with the patient under general anesthesia. This will minimize the risk of ingesta contamination of the abdomen during surgery.
Fig. 18.
A trichobezoar removed from proximal jejunum on an Angus heifer.
IV fluid therapy is based on the clinical estimate of dehydration, severity of intestinal lesions identified at surgery, and severity of serum biochemistry changes. In general, cattle should receive 20 to 60 L of isotonic saline, intravenously, over 12 hours. The authors routinely add calcium (1 mL of 23% calcium gluconate/kg body weight) and dextrose (to create a 1.25% solution) to the IV fluids. NSAIDs (banamine, 1 mg/kg body weight, IV, every 12 hours for 3 days) and antibiotics (for 3–5 days) also are administered.
Prognosis
The prognosis for return to productive use is based on the animal’s body condition, presence of visceral perforation or peritonitis, and ability to perform surgical removal of the foreign body without contaminating the abdomen.
Incarceration, entrapment, internal herniation
A loop of jejunum or any organs can be trapped in a rent through the mesentery, mesocolon, around the umbilical vein, or simply caught between adhesions. The outcome is variable depending on the severity of the obstruction. It can be a simple ileus to a strangulated segment of intestines. There are many case reports in the literature showing the infinite possibilities of internal herniation, including incarceration of the jejunum in the epiploic foramen,68 prolapse of the jejunum through a rectal tear,69 jejunal incarceration in a partially everted urinary bladder,70 strangulation of the jejunum by a persistent urachus,71 or around a persistent round ligament of the liver.72 Strangulation of the caudal sigmoid flexure of the duodenum was described in 2 late-pregnant cows. The uterus was trapped through the mesoduodenum caudally causing an oral obstruction without significant vascular strangulation.16 It was hypothesized that the mesoduodenal defect was developmental. Manual reduction was impossible. Transection and end-to-end anastomosis were successfully performed on both animals.16
Gut tie is a jejunal incarceration around the ductus deferens in castrated steers.73, 74, 75 It could be related to excessive manual traction on the testicle during castration creating a rent in the mesoductus and allowing jejunum to wrap around the ductus deferens causing strangulation. The clinical signs are similar to any intestinal obstruction. Abdominal palpation per rectum will show mucus or a small amount of feces, distended jejunum, and a small-diameter tight band crossing the caudal abdominal cavity toward the inguinal rings. Although manual rupture of this band per rectum has been reported,75 surgery is often indicated to precisely cut the ductus and evaluate intestinal viability. Prognosis is generally good, depending on the viability of the affected portion of the jejunum.
The ileum is rarely affected or rarely needs surgical attention. Jejunoileal or ileocecal intussusception,29, 33 volvulus76 and persistent vitellointestinal duct in a calf77 have been reported. Biopsy of the ileum for the diagnosis of paratuberculosis78 or its cannulation to study digestion of different nutrient has been reported.79, 80 Ileal impaction has been described in 22 cows.81 The cause is unknown, however, because animals were mainly affected during fall and winter, it is hypothesized that winter type of feeding is a significant contributing factor. The affected animals had signs of mild colics, absent or scant feces, and dilated jejunum at rectal palpation and ultrasound. Exploratory laparotomy was necessary to confirm the diagnosis. In 19 animals, the ingesta was broken down and massaged aborally into the caecum. In a few animals, typhlotomy or enterotomy was necessary to decompress and remove the impaction. A side-to-side ileocecal anastomosis had to be performed in 6 animals to prevent recurrence. The short-term and long-term prognosis was considered good.
Footnotes
The authors have nothing to disclose.
References
- 1.Braun U. Ultrasound as a decision-making tool in abdominal surgery in cows. Vet Clin North Am Food Anim Pract. 2005;21:33–53. doi: 10.1016/j.cvfa.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 2.Braun U., Nuss K., Knubben-Schweizer G. [The use of ultrasonography for diagnosing the cause of colic in cows. A review] Tierarztl Prax Ausg G Grosstiere Nutztiere. 2011;39:289–298. [in German] [PubMed] [Google Scholar]
- 3.Coetzee J.F., Gehring R., Tarus-Sang J. Effect of sub-anesthetic xylazine and ketamine ('ketamine stun') administered to calves immediately prior to castration. Vet Anaesth Analg. 2010;37:566–578. doi: 10.1111/j.1467-2995.2010.00573.x. [DOI] [PubMed] [Google Scholar]
- 4.Levine S.A., Smith D.F., Wilsman N.J. Arterial and venous supply to the bovine jejunum and proximal part of the ileum. Am J Vet Res. 1987;48:1295–1299. [PubMed] [Google Scholar]
- 5.Fubini S.L., Trent A.M. Small intestine surgery in cattle. In: Fubini S.L., Ducharme N.G., editors. Farm animal surgery. Elsevier; St Louis (MO): 2004. pp. 240–248. [Google Scholar]
- 6.Steiner A., Waldvogel A., Wicki P. Comparison of staple and Gambee techniques for enterotomy closure in the normal bovine jejunum. Zentralbl Veterinarmed A. 1992;39:193–203. doi: 10.1111/j.1439-0442.1992.tb00173.x. [DOI] [PubMed] [Google Scholar]
- 7.Gieseler T., Wittek T., Furll M. Effects of preoperative flunixin meglumine in cows with left abomasal displacement (LDA) Tierarztl Prax Ausg G Grosstiere Nutztiere. 2008;36:15–19. [in German] [Google Scholar]
- 8.Nichols S. Postoperative assessment: ileus and pain management. 2011 ACVS Veterinary Symposium. Illinois, November 3–5, 2011.
- 9.Braun U., Steiner A., Gotz M. Clinical signs, diagnosis and treatment of duodenal ileus in cattle. Schweiz Arch Tierheilkd. 1993;135:345–355. [PubMed] [Google Scholar]
- 10.Weaver A.D. Duodenal perforation and abdominal abscess in a cow. J Am Vet Med Assoc. 1989;195:1603–1605. [PubMed] [Google Scholar]
- 11.Steiner A., Muller L., Pabst B. An unusual complication after the partial resection of the ascending duodenum of a cow. Tierarztl Prax. 1989;17:17–20. [in German] [PubMed] [Google Scholar]
- 12.Cebra C.K., Cebra M.L., Garry F.B. Gravel obstruction in the abomasum or duodenum of two cows. J Am Vet Med Assoc. 1996;209:1294–1296. [PubMed] [Google Scholar]
- 13.Anderson D.E. Surgical diseases of the small intestine. Vet Clin North Am Food Anim Pract. 2008;24:383–401. doi: 10.1016/j.cvfa.2008.02.004. viii. [DOI] [PubMed] [Google Scholar]
- 14.Mullowney P.C., Whitlock R.H. Traumatic duodenitis in a dairy cow. Vet Rec. 1978;103:557–558. doi: 10.1136/vr.103.25.557. [DOI] [PubMed] [Google Scholar]
- 15.Braun U., Schnetzler C., Previtali M. Duodenal ileus caused by a calf feeding nipple in a cow. BMC Vet Res. 2011;7:2. doi: 10.1186/1746-6148-7-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Koller U., Lischer C., Geyer H. Strangulation of the duodenum by the uterus during late pregnancy in two cows. Vet J. 2001;162:33–37. doi: 10.1053/tvjl.2000.0543. [DOI] [PubMed] [Google Scholar]
- 17.Muggli E., Lesser M., Braun U. Herniation of the gravid uterus through a mesoduodenal defect and concurrent omental hernia in a cow. Vet Surg. 2014;43:91–94. doi: 10.1111/j.1532-950X.2013.12087.x. [DOI] [PubMed] [Google Scholar]
- 18.Boerboom D., Mulon P.Y., Desrochers A. Duodenal obstruction caused by malposition of the gallbladder in a heifer. J Am Vet Med Assoc. 2003;223:1475–1477. doi: 10.2460/javma.2003.223.1475. 1435. [DOI] [PubMed] [Google Scholar]
- 19.Vogel S.R., Nichols S., Buczinski S. Duodenal obstruction caused by duodenal sigmoid flexure volvulus in dairy cattle: 29 cases (2006-2010) J Am Vet Med Assoc. 2012;241:621–625. doi: 10.2460/javma.241.5.621. [DOI] [PubMed] [Google Scholar]
- 20.van der Velden M.A. Functional stenosis of the sigmoid curve of the duodenum in cattle. Vet Rec. 1983;112:452–453. doi: 10.1136/vr.112.19.452. [DOI] [PubMed] [Google Scholar]
- 21.Constable P.D., St Jean G., Hull B.L. Intussusception in cattle: 336 cases (1964-1993) J Am Vet Med Assoc. 1997;210:531–536. [PubMed] [Google Scholar]
- 22.Pearson H. Intussusception in cattle. Vet Rec. 1971;89:426–437. doi: 10.1136/vr.89.16.426. [DOI] [PubMed] [Google Scholar]
- 23.Yadav G.U., Bhikane A.U., Aher V.D. Standardization of diagnostic procedures and operative methodology in clinical cases of intussusception in bovines. Intas Polivet. 2009;10:4–7. [Google Scholar]
- 24.Archer R.M., Cooley A.J., Hinchcliff K.W. Jejunojejunal intussusception associated with a transmural adenocarcinoma in an aged cow. J Am Vet Med Assoc. 1988;192:209–211. [PubMed] [Google Scholar]
- 25.Milnes E.L., McLachlan A. Surgical management of small intestinal intussusception associated with jejunal adenocarcinoma in a dairy cow. N Z Vet J. 2015;63:288–290. doi: 10.1080/00480169.2014.999843. [DOI] [PubMed] [Google Scholar]
- 26.Okamoto M., Itoh H., Koiwa M. Intussusception of the spiral colon associated with fibroserous granulation in a heifer. Vet Rec. 2007;160:376–378. doi: 10.1136/vr.160.11.376. [DOI] [PubMed] [Google Scholar]
- 27.Payton J. Perforating duodenal sarcoma and intussusception in a cow. J Am Vet Med Assoc. 1954;124:351–352. [PubMed] [Google Scholar]
- 28.Silva Filho A.P., Afonso J.A.B., Souza J.C.D.A. Clinical and pathological analysis in 20 cases of intussusception in cattle. Veterinaria e Zootecnia. 2010;17:422–431. [in Portuguese] [Google Scholar]
- 29.Doll K., Klee W., Dirksen G. Cecal intussusception in calves. Tierarztl Prax Ausg G Grosstiere Nutztiere. 1998;26:247–253. [in German] [PubMed] [Google Scholar]
- 30.Dabas V.S., Mistry J.N., Suthar D.N. Intussusception in cross-bred cows: a review of 10 cases. Indian J Vet Surg. 2014;35:50–52. [Google Scholar]
- 31.Dharmaceelan S., Senthilkumar S., Jayakumar K. Jejuno-jejunal intussusception in kangeyam bullocks: a report of two cases. Intas Polivet. 2008;9:149–150. [Google Scholar]
- 32.Hamilton G.F., Tulleners E.P. Intussusception involving the spiral colon in a calf. Can Vet J. 1980;21:32. [PMC free article] [PubMed] [Google Scholar]
- 33.Karapinar T., Kom M. Transrectal ultrasonographic diagnosis of jejunoileal intussusception in a cow. Irish Vet J. 2007;60:422–424. doi: 10.1186/2046-0481-60-7-422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kushwaha R.B., Gupta A.K., Bhadwal M.S. Intestinal obstruction due to intussusception in cattle: a clinical study of twenty cases. Indian J Vet Surg. 2012;33:63–65. [Google Scholar]
- 35.Lee D.B., Shin S.M., Lee K.C. Surgical management of an ileocecocolic intussusception in a Korean native calf: a case report. Veterinarni Medicina. 2013;58:645–649. [Google Scholar]
- 36.Shinde M.M. Intussusception in cross-bred cattle: a study of nine cases. Indian J Vet Surg. 1996;17:52. [Google Scholar]
- 37.Strand E., Welker B., Modransky P. Spiral colon intussusception in a three-year-old bull. J Am Vet Med Assoc. 1993;202:971–972. [PubMed] [Google Scholar]
- 38.Pravettoni D., Morandi N., Rondena M. Repeated occurrence of jejuno-jejunal intussusception in a calf. Can Vet J. 2009;50:287–290. [PMC free article] [PubMed] [Google Scholar]
- 39.Sheikh I., Tyagi S.P., Adarsh K. Usefulness and limitation of ultrasonography in the diagnosis of intestinal intussusception in cows. Vet Med Int. 2011;2011:584387. doi: 10.4061/2011/584387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Horne M.M. Colonic intussusception in a Holstein calf. Can Vet J. 1991;32:493–495. [PMC free article] [PubMed] [Google Scholar]
- 41.Godden S., Frank R., Ames T. Survey of Minnesota dairy veterinarians on the occurrence of and potential risk factors for jejunal hemorrhage syndrome in adult dairy cows. Bov Pract. 2001;35:97–103. [Google Scholar]
- 42.Kirkpatrick M.A., Timms L.L., Kersting K.W. Case report - jejunal hemorrhage syndrome of dairy cattle. Bov Pract. 2001;35:104–116. [Google Scholar]
- 43.st-Jean G., Anderson D.E. Intraluminal-intramural hemorrhage of the small intestine in cattle. In: Howard J.E., Smith R.A., editors. Current veterinary therapy; food animal practice. 4th edition. WB Saunders Co; Philadelphia: 1999. p. 539. [Google Scholar]
- 44.Anderson B.C. 'Point source' haemorrhages in cows. Vet Rec. 1991;128:619–620. doi: 10.1136/vr.128.26.619. [DOI] [PubMed] [Google Scholar]
- 45.Ceci L., Paradies P., Sasanelli M. Haemorrhagic bowel syndrome in dairy cattle: possible role of Clostridium perfringens type A in the disease complex. J Vet Med Ser A. 2006;53:518–523. doi: 10.1111/j.1439-0442.2006.00884.x. [DOI] [PubMed] [Google Scholar]
- 46.Dennison A.C., VanMetre D.C., Callan R.J. Hemorrhagic bowel syndrome in dairy cattle: 22 cases (1997-2000) J Am Vet Med Assoc. 2002;221:686–689. doi: 10.2460/javma.2002.221.686. [DOI] [PubMed] [Google Scholar]
- 47.Francoz D., Babkine M., Couture Y. Haemorrhagic intestinal syndrome in cattle: retrospective study of 37 cases. (Gastro-enterologie) [French] Medecin Veterinaire du Quebec. 2005;35:65–72. [Google Scholar]
- 48.Songer J.G. Clostridial enteric diseases of domestic animals. Clin Microbiol Rev. 1996;9:216–234. doi: 10.1128/cmr.9.2.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Songer JG. Clostridium perfringens type A infection in cattle. Proceedings of the Thirty Second Annual Conference American Association of Bovine Practitioners. Nashville (TN): 1999.
- 50.Bunting M., Lorant D.E., Bryant A.E. Alpha toxin from Clostridium perfringens induces proinflammatory changes in endothelial cells. J Clin Invest. 1997;100:565–574. doi: 10.1172/JCI119566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gustafson C., Tagesson C. Phospholipase C from Clostridium perfringens stimulates phospholipase A2-mediated arachidonic acid release in cultured intestinal epithelial cells (INT 407) Scand J Gastroenterol. 1990;25:363–371. doi: 10.3109/00365529009095500. [DOI] [PubMed] [Google Scholar]
- 52.Elhanafy M.M., French D.D., Braun U. Understanding jejunal hemorrhage syndrome. J Am Vet Med Assoc. 2013;243:352–358. doi: 10.2460/javma.243.3.352. [DOI] [PubMed] [Google Scholar]
- 53.Sockett DC. Hemorrhagic bowel syndrome. Mid Atlantic Nutrition Conference. Maryland, March 24–25, 2004. p. 139–145.
- 54.Abutarbush S.M., Radostits O.M. Jejunal hemorrhage syndrome in dairy and beef cattle: 11 cases (2001 to 2003) Can Vet J. 2005;46:711–715. [PMC free article] [PubMed] [Google Scholar]
- 55.Braun U., Forster E., Steininger K. Ultrasonographic findings in 63 cows with haemorrhagic bowel syndrome. Vet Rec. 2010;166:79–81. doi: 10.1136/vr.c178. [DOI] [PubMed] [Google Scholar]
- 56.Peek S.F., Santschi E.M., Livesey M.A. Surgical findings and outcome for dairy cattle with jejunal hemorrhage syndrome: 31 cases (2000-2007) J Am Vet Med Assoc. 2009;234:1308–1312. doi: 10.2460/javma.234.10.1308. [DOI] [PubMed] [Google Scholar]
- 57.Braun U., Schmid T., Muggli E. Clinical findings and treatment in 63 cows with haemorrhagic bowel syndrome. Schweiz Arch Tierheilkd. 2010;152:515–522. doi: 10.1024/0036-7281/a000117. [DOI] [PubMed] [Google Scholar]
- 58.Anderson D.E., Constable P.D., St Jean G. Small-intestinal volvulus in cattle: 35 cases (1967-1992) J Am Vet Med Assoc. 1993;203:1178–1183. [PubMed] [Google Scholar]
- 59.Fubini S.L., Smith D.F., Tithof P.K. Volvulus of the distal part of the jejunoileum in four cows. Vet Surg. 1986;15:150–152. [Google Scholar]
- 60.Tulleners E.P. Surgical correction of volvulus of the root of the mesentery in calves. J Am Vet Med Assoc. 1981;179:998–999. [PubMed] [Google Scholar]
- 61.Rademacher G., Dirksen G., Klee W. Diagnosis, treatment and prognosis of intestinal torsion in cattle. Tierarztl Umsch. 1995;50:271–276. [in German] [Google Scholar]
- 62.Pearson H. The treatment of surgical disorders of the bovine abdomen. Vet Rec. 1973;92:245–254. doi: 10.1136/vr.92.10.245. [DOI] [PubMed] [Google Scholar]
- 63.Pearson H., Pinsent P.J.N. Intestinal obstruction in cattle. Vet Rec. 1977;101:162–166. doi: 10.1136/vr.101.9.162. [DOI] [PubMed] [Google Scholar]
- 64.Jelinski M.D., Ribble C.S., Campbell J.R. Investigating the relationship between abomasal hairballs and perforating abomasal ulcers in unweaned beef calves. Can Vet J. 1996;37:23–26. [PMC free article] [PubMed] [Google Scholar]
- 65.Cockrill J.M., Beasley J.N., Selph R.A. Trichobezoars in four Angus cows. Vet Med Small Anim Clin. 1978;73:1441–1442. [PubMed] [Google Scholar]
- 66.Ramadan R.O. Massive formation of trichobezoars in sheep. Agri Pract. 1995;16:26–28. [Google Scholar]
- 67.Patel J.H., Brace D.M. Esophageal obstruction due to a trichobezoar in a cow. Can Vet J. 1995;36:774–775. [PMC free article] [PubMed] [Google Scholar]
- 68.Deprez P., Hoogewijs M., Vlaminck L. Incarceration of the small intestine in the epiploic foramen of three calves. Vet Rec. 2006;158:869–870. doi: 10.1136/vr.158.25.869. [DOI] [PubMed] [Google Scholar]
- 69.Charmillot B. Prolapse of the jejunum through perforated rectum in a cow. Enterectomy. Schweizer Archiv fur Tierheilkunde. 1976;118:553–556. [in French] [PubMed] [Google Scholar]
- 70.Frazer G.S. Uterine torsion followed by jejunal incarceration in a partially everted urinary bladder of a cow. Aust Vet J. 1988;65:24–25. doi: 10.1111/j.1751-0813.1988.tb14925.x. [DOI] [PubMed] [Google Scholar]
- 71.Mesaric M., Modic T. Strangulation of the small intestine in a cow by a persistent urachal remnant. Vet Rec. 2003;153:688–689. doi: 10.1136/vr.153.22.688. [DOI] [PubMed] [Google Scholar]
- 72.Ducharme N.G., Smith D.F., Koch D.B. Small intestinal obstruction caused by a persistent round ligament of the liver in a cow. J Am Vet Med Assoc. 1982;180:1234–1236. [PubMed] [Google Scholar]
- 73.Lores M., Haruna J.A., Ortenburger A. Bilateral 'gut-tie' in a recently castrated steer. Can Vet J. 2006;47:155–157. [PMC free article] [PubMed] [Google Scholar]
- 74.Norman T. 'Gut-tie' in steers. Vet Rec. 1997;140:687–688. [PubMed] [Google Scholar]
- 75.Wolfe D.F., Mysinger P.W., Carson R.L. Incarceration of a section of small intestine by remnants of the ductus deferens in steers. J Am Vet Med Assoc. 1987;191:1597–1598. [PubMed] [Google Scholar]
- 76.Cecen C., Celimli N., Kabakaya G.U. Volvulus of the distal jejunum and ileum and mesenteric torsion in a calf. Cattle Pract. 2007;15:97–99. [Google Scholar]
- 77.Strachan W.D., Tremaine W.H., Holdsworth D.M. Persistent vitellointestinal duct in a calf. Vet Rec. 1997;140:629–630. doi: 10.1136/vr.140.24.629. [DOI] [PubMed] [Google Scholar]
- 78.Gilardoni L.R., Paolicchi F.A., Mundo S.L. Bovine paratuberculosis: a review of the advantages and disadvantages of different diagnostic tests. Rev Argent Microbiol. 2012;44:201–215. [PubMed] [Google Scholar]
- 79.Allen A.J., Park K.T., Barrington G.M. Development of a bovine ileal cannulation model to study the immune response and mechanisms of pathogenesis of paratuberculosis. Clin Vaccin Immunol. 2009;16:453–463. doi: 10.1128/CVI.00347-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Harmon D.L., Richards C.J. Considerations for gastrointestinal cannulations in ruminants. J Anim Sci. 1997;75(8):2248–2255. doi: 10.2527/1997.7582248x. [DOI] [PubMed] [Google Scholar]
- 81.Nuss K., Lejeune B., Lischer C. Ileal impaction in 22 cows. Vet J. 2006;171:456–461. doi: 10.1016/j.tvjl.2005.02.024. [DOI] [PubMed] [Google Scholar]