Abstract
West Nile virus was recognized in the United States for the first time in 1999, when it caused an epidemic of encephalitis and meningitis in New York City, NY. Since then, the disease has been steadily moving westward, and human cases were recognized in 39 states and the District of Columbia in 2002. The infection is caused by a flavivirus that is transmitted from birds to humans through the bite of culicine mosquitoes. Most infections are mild, with symptoms primarily being fever, headache, and myalgias. People older than 50 years are at highest risk of severe disease, which may include encephalomyelitis. In 2002, 5 new modes of transmission were recognized: blood product transfusion, organ transplantation, breast-feeding, transplacental transmission, and occupational exposure in laboratory workers. The transmission season was long, with cases occurring into December in some parts of the United States. Currently, there is no specific drug treatment or vaccine against the infection, and avoiding mosquito bites is the best way to protect against the disease.
Keywords: CDC, Centers for Disease Control and Prevention; CNS, central nervous system; CSF, cerebrospinal fluid; ELISA, enzyme-linked immunosorbent assay; JE, Japanese encephalitis; MRI, magnetic resonance imaging; PCR, polymerase chain reaction; SLE, St Louis encephalitis; WNV, West Nile virus; WNVME, WNV meningoencephalitis
The West Nile virus (WNV) was first isolated in 1937 from the blood of an infected woman in the West Nile province of what is now known as Uganda.1 Further investigations in Egypt identified the virus in birds and animals in addition to humans. The virus was recognized as causing severe meningoencephalitis during a 1957 outbreak in nursing home residents in Israel. Subsequent outbreaks of WNV infection reported from Israel and Africa were mild and consisted of a flulike illness. In the mid 1990s more frequent and more severe outbreaks were reported from Romania,2 Russia,3, 4 and Israel,5 and meningoencephalitis in humans was again a prominent feature. The WNV first appeared in the United States in August 1999 and since then has spread nationwide.
EPIDEMIOLOGY
In August 1999, an unusual cluster of cases of meningoencephalitis associated with muscle weakness was noticed in New York City, NY. Initial epidemiological and environmental investigations suggested an arthropod-borne viral cause (arbovirus), and both serologic testing and viral testing were performed to identify such a virus. The first 8 patients tested positive for antibodies against St Louis encephalitis (SLE), a mosquito-borne illness, and mosquito-control measures were instituted. Concurrently, an unusual number of deaths were noted in birds, primarily crows, and in some exotic captive bird species in the Bronx Zoo in New York City. Since SLE does not cause deaths in birds, the human and bird illnesses were thought to be unrelated. Autopsies performed on captive birds that died in the Bronx Zoo revealed varying degrees of meningoencephalitis and severe myocarditis. Tissue specimens were negative for common avian pathogens and the equine encephalitis viruses. However, the National Veterinary Services Laboratories in Ames, Iowa, isolated a virus from the birds’ tissues and forwarded this to the Centers for Disease Control and Prevention (CDC). Testing by polymerase chain reaction (PCR) and DNA sequencing at the CDC and the University of California at Irvine indicated that this virus was closely related to WNV and that it was identical to virus from human brain tissue from 4 encephalitis cases in New York. On retesting, all initial serum/cerebrospinal fluid (CSF) specimens reactive to SLE were positive for WNV by IgM enzyme-linked immunosorbent assay (ELISA), with higher positive-negative ratios than to SLE. An additional 18 patient samples that were either borderline or negative for SLE were positive for antibody to WNV, confirming that WNV was indeed the cause of the illness in these patients.6
The discovery in 2000 that WNV survived in overwintering Culex mosquitoes in New York dashed hopes that the virus would not do well in temperate climates and heralded the westward spread of the disease.7 Since then the infection has been steadily spreading westward. From 1999 through 2001, there were 149 cases of WNV human illness reported in the United States and 18 deaths (Table 1 ). In 2002 reported cases increased dramatically, with 4156 laboratory-confirmed human cases and 284 deaths (Table 1, Table 2 ). Illinois, Indiana, Ohio, Michigan, Louisiana, and Texas reported the largest number of cases. In 2003, the first confirmed human case was reported from South Carolina on July 7. As of August 11, 2003, the CDC has received reports of a total of 182 human cases of WNV infection from 17 states with 5 reported deaths. At the same time in 2002, 112 human cases from 4 states had been reported. In 2002 the vast majority of cases occurred after the first week of August, and if the same pattern holds true in 2003, an even larger outbreak is expected this year. Surveillance on animal populations by the CDC supports this hypothesis. The number of infected horses and infected mosquito pools far outpaces the number reported at this time in 2002.9
Table 1.
West Nile Virus in Humans, 1999–2002
| Year | No. of states | No. of deaths/cases | Case fatality (%) | Date range |
|---|---|---|---|---|
| 1999 | 1 | 7/62 | 11.3 | August 2-September 24 |
| 2000 | 3 | 2/21 | 9.5 | July 29-September 27 |
| 2001 | 10 | 9/66 | 13.6 | July 13-December 7 |
| 2002 | 39* | 284/4156 | 6.8 | May 19-December 14 |
Plus Washington, DC. Data from reference 8.
Table 2.
Features of the 2002 Epidemic of West Nile Virus
|
VIROLOGY
The WNV is a flavivirus and a member of the Japanese encephalitis (JE) virus serocomplex, which contains several viruses also associated with human encephalitis: JE, SLE, Murray Valley encephalitis, and Kunjin (a subtype of WNV). All flaviviruses are closely related antigenically, which accounts for the serologic cross-reactions observed. Members of the JE complex are even more closely related; often, specialized tests are needed to differentiate the infecting flavivirus. The WNV is a spherical particle that is approximately 50 nm in diameter. It is an enveloped single-stranded RNA virus. The viral membrane has 2 integral glycoproteins—E (viral envelope) and M (membrane). The E-glycoprotein, the viral hemagglutinin, mediates virushost cell binding. It elicits most of the virus-neutralizing antibodies.10, 11
Genetic analysis indicates that the virus responsible for the 1999 outbreak was extremely similar (99.8% homology) to the WNV isolated from a dead goose in Israel in 1998.12 This suggests that the virus probably originated in the Middle East. It is unclear how the virus arrived in the United States. The most likely explanations are that it was carried by infected migrating birds or possibly by infected adult mosquitoes or larvae inadvertently transported on an airplane. Because humans have extremely low-level viremias, an infected international traveler is unlikely to have been the source.
TRANSMISSION
The most common route of WNV infection to humans is through the bite of an infected culicine mosquito.13, 14 Mosquitoes become infected when they feed on infected birds that have high levels of WNV in their blood. Infected mosquitoes can then transmit WNV when they feed on humans or other animals. People, horses, and most other mammals do not develop high-level viremias and hence are traditionally thought of as “dead-end” hosts.
The WNV has been detected in dead birds of at least 138 species. Birds of the family Corvidae (crows and blue jays) appear to be particularly susceptible to WNV. People at risk of exposure to WNV include those working outdoors when mosquitoes are actively biting, such as farmers, foresters, landscapers, groundskeepers, and construction and other outdoor workers. The WNV is not transmitted from person to person by mosquitoes. However, 5 new modes of transmission were recognized in 2002 and are described subsequently. Recognition of these new modes of transmission invalidates the theory that humans are dead-end hosts.
Blood Transfusion
Between August 2002 and January 2003, there were 20 confirmed cases of WNV transmission associated with blood product transfusion, linked to 14 infectious donor units. Transmission of WNV has been documented to occur via red blood cell, plasma, and platelet transfusions. Five of 14 infected donors and 7 of the 20 confirmed cases were asymptomatic.15
In 2003, the Food and Drug Administration recommended that between June 1 and November 30 (peak transmission season) all blood banks should screen potential blood donors. Those who have had a headache with fever in the past week should be deferred from donation for 28 days.16 Cases of WNV infection in persons who have received blood transfusions in the 4 weeks before they became ill should be reported to the blood collection agency and to the CDC. In addition, public health departments and blood banks will cooperate to identify blood products from donors who develop a West Nile viral illness after giving blood. Prompt reporting of these cases will help facilitate withdrawal of potentially infected blood components. However, because symptoms occur in only 20% of infected patients, clinical screening will have limited effectiveness. Hence, the Food and Drug Administration also recommends screening donor units for WNV by using nucleic acid testing.
Organ Transplantation
In August 2002, 4 transplant recipients who had received organs from a common donor developed fever; 3 had meningoencephalitis.17, 18 WNV infection was diagnosed in all 4 recipients. The serum and plasma samples drawn from the organ donor at the time of organ recovery were positive for WNV by PCR and virus culture. The organ donor had received blood product transfusions from 63 blood donors, and further testing identified 1 donor who had viremia at the time of donation. Blood from this blood donor had also been transfused into the patient described below.
Breast-feeding
In September 2002, a woman developed WNV meningoencephalitis (WNVME) 11 days after receiving a postpartum blood transfusion from a WNV-infected donor. Her CSF sample was positive for WNV-specific IgM. She had been breast-feeding her newborn, and a sample of her breast milk obtained 16 days after delivery was positive for WNV by PCR and for WNV-specific IgG and IgM antibodies. The newborn remained afebrile and healthy, but a serum sample from the newborn on day 25 showed WNV-specific IgM antibodies. The newborn had little outdoor or other exposure to mosquitoes, suggesting that the infection most likely came from the mother through breast-feeding.19
Because the benefits of breast-feeding are well established and the risk of transmission of WNV via breastfeeding remains to be quantified, this report should not influence an individual decision to breast-feed. Between 1999 and 2002, WNV infection has been reported in only 4 infants, 3 of whom were breast-fed. There have been no reported adverse consequences of insect repellent use in breast-feeding women, and this would be the best way to prevent exposure not only to WNV but also to other insect-borne illnesses.
Transplacental Transmission
Adverse pregnancy outcomes have been reported previously with intrapartum infection with JE and dengue but not with WNV. In August 2002, a 20-year-old previously healthy woman in the 27th week of pregnancy was admitted to the hospital because of symptoms compatible with WNVME, and her serum and CSF samples were positive for WNV-specific IgM. She was delivered of a live newborn 5 weeks later. The infant had serologic evidence of WNV infection, bilateral chorioretinitis, and magnetic resonance imaging (MRI) evidence of severe bilateral white matter loss in the temporal and occipital lobes.20
Pregnant women should take precautions to reduce their risk for WNV and other arboviral infections by avoiding mosquitoes and by using protective clothing and insect repellents. When WNV transmission is occurring in an area, pregnant women who have an illness that is clinically consistent with acute WNV infection should undergo appropriate diagnostic testing. Screening of asymptomatic pregnant women or newborns for evidence of WNV infection is not recommended because no specific treatment for this infection is available, WNV-IgM antibody might persist for more than a year, and tests of a single serum sample cannot accurately determine the timing of infection. The CDC has initiated a voluntary registry to monitor birth outcomes among WNV-infected women. In the event of an adverse birth outcome, maternal and fetal or newborn samples should be submitted to a state public health laboratory or the CDC for testing.20
Laboratory Acquisition
In 2002, there were 2 reported cases of occupationally acquired infection among laboratory workers associated with percutaneous injury while performing autopsies on animals for WNV surveillance.21 Both patients had no other risk factors for acquiring disease. The illness was mild and self-limited, and recovery was complete.
Since the 2002 WNV epidemic, the number of laboratories and laboratory workers involved in arboviral diagnostic activities has increased dramatically, increasing the potential for further laboratory-acquired infections. Laboratory workers should take precautions to minimize exposure to potentially infected tissue and body fluids and use biosafety level 3 containment when working with live WNV. If unintentional exposure to WNV occurs, the area should be washed thoroughly with soap and water, and a baseline serum sample should be obtained and stored. If the worker has an illness within the next 2 weeks, medical evaluation, consultation with public health officials, and collection of additional serum samples for testing are recommended.
WNV IN ANIMALS
Cases of WNV disease in horses have been documented extensively.22, 23, 24 More than 14,000 cases occurred in 2003, with a mortality rate of approximately 35%. Horses most likely become infected with WNV in the same manner as humans, ie, by the bite of infectious mosquitoes. In locations where WNV is circulating, horses should be protected from mosquito bites as much as possible. Horses vaccinated against other arboviruses are not protected against WNV infection. An inactivated WNV equine vaccine (Innovator, Fort Dodge Animal Health Inc, Overland Park, Kan) received a full license in 2003; it has an estimated 95% efficacy after administration of 2 doses 3 to 6 weeks apart. An annual booster at the start of the mosquito season is recommended.
The WNV does not appear to cause illness in dogs or cats, although serosurveys have indicated that dogs are infected frequently.25, 26 Through December 2001, the CDC also received a small number of reports of WNV infection in bats, a chipmunk, a skunk, a squirrel, and a domestic rabbit. No animal-to-human transmission has been reported. There is 1 report of fatal WNV illness in farmed alligators traced to contaminated meat from horses infected with WNV.27
CLINICAL PRESENTATION
AfterWNV infection, the viralincubation period in humans ranges from 2 to 15 days (usual period, 2-6 days).28, 29, 30 Most human infections are clinically inapparent. On the basis of seroepidemiological studies, 1 in 5 infected persons will have a febrile illness characterized by abrupt onset of fever, headache, backache, myalgia, and anorexia that typically lasts 3 to 6 days. A roseolar or maculopapular rash involving primarily the face and trunk occurs in about half of the patients and can last up to a week. Generalized lymphadenopathy is common. Other serious nonneurologic complications in humans occur rarely and include myocarditis, pancreatitis, and fulminant hepatitis.
Neurologic Complications
Approximately 1 in 150 infected persons will have a severe illness with central nervous system (CNS) involvement.11 Advanced age is the most important risk factor for neurologic disease. People older than 50 years have a 10fold higher risk of developing neurologic symptoms; the risk is 43 times higher in patients older than 80 years.31, 32, 33, 34 Meningoencephalitis is the most common type of CNS presentation, but isolated encephalitis or meningitis also has occurred. West Nile virus–associated acute flaccid paralysis has been documented extensively.35, 36, 37, 38 Patients with this presentation are relatively young, the weakness is asymmetrical, and there is no sensory involvement.
This weakness was previously attributed to a peripheral demyelinating process (Guillain-Barré syndrome), but new studies show that the damage is localized to spinal anterior horn cells resulting in a poliomyelitis-like syndrome.38 Most patients had little or no improvement on short-term follow-up. In contrast to patients with Guillain-Barré syndrome, in which there is an albuminocytologic dissociation, these patients have elevation of both protein and cells in the CSF. Electromyography and nerve conduction studies can also help make the distinction.
Newer syndromes reported after the 2002 outbreak38 include movement disorders with tremors and myoclonus, parkinsonism, and rhabdomyolysis. Other less common neurologic manifestations include ataxia, cranial nerve involvement, optic neuritis, and polyradiculopathy. Seizures were reported in one third of patients with encephalitis in earlier outbreaks but were not a common feature in the 2000 and 2001 outbreaks in the United States.
Although the exact mechanism is uncertain, CNS infection presumably occurs when the virus crosses the blood-brain barrier by endothelial replication or axonal transport through olfactory neurons. Factors that enhance progression of CNS infections among elderly persons may include those that disrupt the blood-brain barrier (eg, hypertension) or increase the duration and level of viremia (eg, immune dysfunction).
Patients without neurologic involvement generally have an uneventful recovery. Involvement of the CNS is associated with mortality of up to 10% and with long-term morbidity. In the New York City outbreak in 1999, only 35% of patients with encephalitis had complete recovery at 1 year. The others reported varying degrees of neurologic impairment, including fatigue, chronic headaches, memory loss, difficulty walking, muscle weakness, and depression.32
Radiological Findings
In identifying CNS inflammation, MRI is more helpful than computed tomography. Computed tomographic scans in patients with WNV encephalitis usually show normal findings or preexisting lesions and long-term changes. In 30% of patients, MRI shows leptomeningeal or periventricular enhancement. Some authors suggest that high signal intensities on T2-weighted images in the thalamus and the basal ganglia may be an early indicator that a patient has WNV encephalitis.39
Pathologic Findings
In fatal cases, pathologic findings are diffuse inflammation of the brain and spinal cord with small hemorrhages, perivascular cuffing, and extensive neuronal degeneration.
The brainstem, especially the medulla, is involved most extensively, and cranial nerve roots may show a mononuclear infiltrate.37, 40
Laboratory Findings
Patients may have either a mild leukocytosis or a mild leukopenia. Mild hyponatremia may be present in some patients, especially those with encephalitis. Results of other laboratory studies are usually normal. In patients with WNVME, CSF findings include mild pleocytosis (usually 30-100 cells/µL; range, 0-1800 cells/µL) with lymphocytic predominance, mild to moderate protein elevation, and normal CSF glucose levels.
Infection is confirmed by detecting virus or antibodies against the virus (Table 3 ). Attempts at virus isolation are usually unsuccessful because the level of viremia is extremely low and clears rapidly (Figure 1 ). Viremia may last longer in patients with advanced malignancies or those taking immunosuppressive drugs.
Table 3.
Surveillance Case Definitions of WNV Infection*
|
CSF = cerebrospinal fluid; ELISA = enzyme-linked immunosorbent assay; WNV = West Nile virus.
Detected by IgM capture ELISA.
The ELISA for IgG is sensitive but nonspecific and should be confirmed with plaque reduction neutralization testing.
Figure 1.
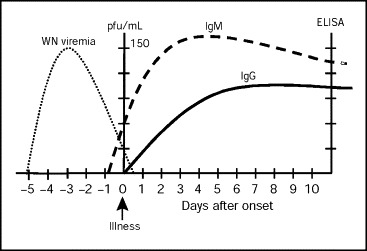
Laboratory diagnosis of West Nile virus (WNV) infection. ELISA = enzyme-linked immunosorbent assay; pfu = plaque-forming units. Data from reference 8.
Serologic tests can be performed on serum or CSF. West Nile virus–specific IgM can be detected in serum or CSF by IgM capture ELISA in at least 90% of patients on or after the eighth day of illness. Once IgM antibodies appear, they persist for more than 6 months after illness in most patients and may persist for as long as 500 days. Hence, contrary to most other infections, the presence of IgM antibodies is not suggestive of acute infection unless it is associated with a recent compatible illness. IgM antibody does not cross the blood-brain barrier, and thus positive findings on CSF are specific for CNS infection except in the case of CSF contamination with blood during a traumatic spinal tap.
IgG antibodies appear by the seventh day of illness and can be detected by 3 weeks after infection in most patients. In acute infection, IgG titers should increase between days 7 and 21 after infection. False-positive serologic results may occur in patients who have received the yellow fever or JE vaccines or have been infected with these viruses or other flavivirus such as dengue. Therefore, a travel history and immunization history are important to interpret serologic results correctly. There is also cross-reactivity with the SLE virus, which occurs widely in the United States. Positive IgG results by ELISA should be confirmed with a more specific test—the plaque reduction neutralization test.41
Viral RNA can be detected in clinical specimens with use of PCR. In a study of patients with serologically confirmed acute WNVME, the sensitivity of the reverse-tran scriptase–PCR (TaqMan) in CSF and serum was 57% and 14%, respectively.42 In fatal WNVME, WNV can be detected in brain tissue by immunohistochemistry or PCR and culture techniques.43
MANAGEMENT
There is no established treatment for WNV infection. Patients with WNVME should be hospitalized, and treatable CNS infections or conditions should be ruled out. Patients with profound muscle weakness may require intubation and mechanical ventilation. In vitro studies have shown activity of ribavirin, interferon alfa, and pyrazidine nucleosides against WNV, but no clinical data support the use of these agents at present. Intravenous immunoglobulin that has WNV-specific antibodies has been shown to have prophylactic and therapeutic efficacy for WNV infection in mice.44
There is anecdotal evidence of efficacy of intravenous immunoglobulin in humans.45 However, supportive care including ventilatory support when necessary and treatment of cerebral edema are the key management measures.
PREVENTION
Reducing contact between humansand potentially infected mosquitoes is the only way to reduce infection rates, morbidity, and mortality due to WNV infection. This can be done with specific personal protection behaviors and with mosquito-control activities (Table 4 ).
Table 4.
Preventive Measures Against West Nile Virus Infection
|
Personal protective measures that reduce the risk of infection include the following: avoiding outdoor activity in areas when potentially infected mosquitoes are present; eliminating conditions that support mosquito breeding (eg, eliminate standing water, clean rain gutters); maintaining window and door screens; wearing long-sleeved shirts and long pants when outdoors; applying insect repellents containing DEET (N, N-diethyl-m-toluamide) or permethrin to clothes; and applying DEET-containing repellents to exposed skin.
DEET is a broad-spectrum repellent that is effective against many species of mosquitoes and other biting insects. The duration of protection provided by DEET is proportional to its concentration, to a maximum of 50%. Concentrations greater than 50% provide no additional benefit. DEET should be applied to clothes and on all exposed areas and washed off when returning indoors. Overall, DEET has a good safety profile and is safe to use in infants older than 2 months and in pregnant women.46 Currently available non-DEET repellents do not provide satisfactory or reliable protection against mosquitoes.
Permethrin, a synthetic pyrethroid, can be used as an adjunct to DEET-containing insect repellent. Permethrin clothing applications were originally developed in cooperation with and for use by the US military. Permethrin is odorless after drying and binds tightly to fabric. One application to clothing can last up to 6 weeks despite several launderings. An additional benefit is that permethrin is active against ticks and other biting insects. It is inactivated on contact with skin, and hence DEET application to exposed skin is neccessary.
In areas where there has been WNV activity in birds, animals, or humans, local government agencies should establish mosquito-control programs, including applying larvicides to water collections and spraying for adult mosquito control.47
Two candidate vaccines against WNV are being evaluated. One vaccine (Acambis, Cambridge, Mass) is based on the yellow fever vaccine. The other vaccine was developed by the National Institute for Allergy and Infectious Diseases and uses the dengue virus as a backbone to carry WNV genes. Both these vaccines are expected to be tested in phase 1 human trials in late 2003.
CONCLUSION
Infections of WNV have resulted in pronounced human and economic losses since WNV first appeared in the United States in 1999. The year 2002 saw a dramatic increase in the number of cases, widening geographic distribution, and an increase in the period when cases occurred (Table 2). Also, several new modes of transmission and new clinical presentations were recognized. The illness causes substantial morbidity in elderly persons, immunosuppressed persons, and pregnant women. These groups should take special precautions against mosquito bites. Management consists of supportive care; no specific treatment or vaccine is currently available.
Questions About WNV
-
1.Which one of the following belongs to the Flaviviridae family besides WNV?
-
a.Influenza virus
-
b.JE
-
c.Herpes simplex virus
-
d.Varicella virus
-
e.SARS (severe acute respiratory syndrome)
-
a.
-
2.Which one of the following modes of transmission of WNV has not been reported?
-
a.Blood transfusion
-
b.Organ transplantation
-
c.Animals to humans
-
d.Mosquito bites
-
e.Breast-feeding
-
a.
-
3.Which one of the following is most likely to be seen in a patient with WNVM?
-
a.CSF pleocytosis and elevated CSF protein level
-
b.CSF pleocytosis and normal CSF protein level
-
c.CSF glucose level that is less than one third of concomitant serum glucose level
-
d.Meningeal enhancement on computed tomographic scan
-
e.Loss of sensation in lower extremities
-
a.
-
4.Which one of the following is most likely due to acute WNV infection?
-
a.WNV-IgM in CSF in an asymptomatic person
-
b.WNV-IgG in serum in a 44-year-old missionary with fever and chills who has recently returned from a 4-year stay in rural China
-
c.WNV-IgM in CSF in a 75-year-old man with asymmetrical upper extremity weakness
-
d.WNV-IgG in serum in a 45-year-old man with fever who received the yellow fever vaccine 4 months previously before a planned trip to Tanzania
-
e.WNV-IgM in CSF in a 70-year-old man with chronic headaches and left upper extremity weakness after a febrile illness 8 months previously
-
a.
-
5.Which one of the following measures is not appropriate to prevent WNV infection?
-
a.Avoiding being outdoors at dawn and dusk
-
b.Using DEET-containing insect repellent
-
c.Using permethrin clothing spray
-
d.Checking for and draining collections of standing water
-
e.Avoiding close contact with persons in whom WNV infection has been diagnosed
-
a.
Correct answers:
1. b, 2. c, 3. a, 4. c, 5. e
REFERENCES
- 1.Smithburn KC, Hughes TP, Burke AW, Paul JH. Neurotropic virus isolated from blood of native of Uganda. Am J Trop Med. 1940;20:471–492. [Google Scholar]
- 2.Tsai TF, Popovici F, Cernescu C, Campbell GL, Nedelcu NI. West Nile encephalitis epidemic in southeastern Romania. Lancet. 1998;352:767–771. doi: 10.1016/s0140-6736(98)03538-7. [DOI] [PubMed] [Google Scholar]
- 3.Platonov AE, Shipulin GA, Shipulina OY. Outbreak of West Nile virus infection, Volgograd Region, Russia, 1999. Emerg Infect Dis. 2001;7:128–132. doi: 10.3201/eid0701.010118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lvov DK, Butenko AM, Gromashevsky VL. Isolation of two strains of West Nile virus during an outbreak in southern Russia, 1999. Emerg Infect Dis. 2000;6:373–376. doi: 10.3201/eid0604.000408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chowers MY, Lang R, Nassar F. Clinical characteristics of the West Nile fever outbreak, Israel, 2000. Emerg Infect Dis. 2001;7:675–678. doi: 10.3201/eid0704.010414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Centers for Disease Control and Prevention Outbreak of West Nile-like viral encephalitis—New York, 1999. MMWR Morb Mortal Wkly Rep. 1999;48:845–849. [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention Update: surveillance for West Nile virus in overwintering mosquitoes—New York, 2000. MMWR Morb Mortal Wkly Rep. 2000;49:178–179. [PubMed] [Google Scholar]
- 8.Slide presentations at: Fourth National Conference on West Nile Virus in the United States; New Orleans, La; February 9-11, 2003.
- 9.Centers for Disease Control and Prevention Statistics, surveillance, and control: West Nile virus in the United States as of August 11, 2003. Available at: http://www.cdc.gov/ncidod/dvbid/westnile/surv&control03Maps.htm Accessibility verified August 12, 2003.
- 10.Brinton MA. The molecular biology of West Nile Virus: a new invader of the western hemisphere. Annu Rev Microbiol. 2002;56:371–402. doi: 10.1146/annurev.micro.56.012302.160654. [DOI] [PubMed] [Google Scholar]
- 11.Petersen LR, Marfin AA. West Nile virus: a primer for the clinician. Ann Intern Med. 2002;137:173–179. doi: 10.7326/0003-4819-137-3-200208060-00009. [DOI] [PubMed] [Google Scholar]
- 12.Lanciotti RS, Roehrig JT, Deubel V. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science. 1999;286:2333–2337. doi: 10.1126/science.286.5448.2333. [DOI] [PubMed] [Google Scholar]
- 13.Turell MJ, O'Guinn ML, Dohm DJ, Jones JW. Vector competence of North American mosquitoes (Diptera: Culicidae) for West Nile virus. J Med Entomol. 2001;38:130–134. doi: 10.1603/0022-2585-38.2.130. [DOI] [PubMed] [Google Scholar]
- 14.Dohm DJ, Sardelis MR, Turell MJ. Experimental vertical transmission of West Nile virus by Culex pipiens (Diptera: Culicidae) J Med Entomol. 2002;39:640–644. doi: 10.1603/0022-2585-39.4.640. [DOI] [PubMed] [Google Scholar]
- 15.Centers for Disease Control and Prevention Investigations of West Nile virus infections in recipients of blood transfusions. MMWR Morb Mortal Wkly Rep. 2002;51:973–974. [PubMed] [Google Scholar]
- 16.Food and Drug Administration Revised Recommendations for the Assessment of Donor Suitability and Blood and Blood Product Safety in Cases of Known or Suspected West Nile Virus Infection. May 2003. Available at: http://www.fda.gov/cber/gdlns/wnvguid.pdf Accessibility verified July 30, 2003.
- 17.Centers for Disease Control and Prevention Update: investigations of West Nile virus infections in recipients of organ transplantation and blood transfusion—Michigan, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:879. [PubMed] [Google Scholar]
- 18.Iwamoto M, Jernigan DB, Guasch A, West Nile Virus in Transplant Recipients Investigation Team Transmission of West Nile virus from an organ donor to four transplant recipients. N Engl J Med. 2003;348:2196–2203. doi: 10.1056/NEJMoa022987. [DOI] [PubMed] [Google Scholar]
- 19.Centers for Disease Control and Prevention Possible West Nile virus transmission to an infant through breast-feeding—Michigan, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:877–878. [PubMed] [Google Scholar]
- 20.Centers for Disease Control and Prevention Intrauterine West Nile virus infection—New York, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:1135–1136. [PubMed] [Google Scholar]
- 21.Centers for Disease Control and Prevention Laboratory-acquired West Nile virus infections—United States, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:1133–1135. [PubMed] [Google Scholar]
- 22.McLean RG, Ubico SR, Bourne D, Komar N. West Nile virus in livestock and wildlife. Curr Top Microbiol Immunol. 2002;267:271–308. doi: 10.1007/978-3-642-59403-8_14. [DOI] [PubMed] [Google Scholar]
- 23.Trock SC, Meade BJ, Glaser AL. West Nile virus outbreak among horses in New York State, 1999 and 2000. Emerg Infect Dis. 2001;7:745–747. doi: 10.3201/eid0704.010427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Murgue B, Murri S, Zientara S, Durand B, Durand JP, Zeller H. West Nile outbreak in horses in southern France, 2000: the return after 35 years. Emerg Infect Dis. 2001;7:692–696. doi: 10.3201/eid0704.010417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Komar N, Panella NA, Boyce E. Exposure of domestic mammals to West Nile virus during an outbreak of human encephalitis, New York City, 1999. Emerg Infect Dis. 2001;7:736–738. doi: 10.3201/eid0704.010424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Blackburn NK, Reyers F, Berry WL, Shepherd AJ. Susceptibility of dogs to West Nile virus: a survey and pathogenicity trial. J Comp Pathol. 1989;100:59–66. doi: 10.1016/0021-9975(89)90090-x. [DOI] [PubMed] [Google Scholar]
- 27.Miller DL, Mauel MJ, Baldwin C. West Nile virus in farmed alligators. Emerg Infect Dis[serial online] 2003;9:794–799. doi: 10.3201/eid0907.030085. Available at: http://www.cdc.gov/ncidod/EID/vol9no7/03-0085.htm Accessibility verified July 30, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nash D, Mostashari F, Fine A, 1999 West Nile Outbreak Response Working Group The outbreak of West Nile virus infection in the New York City area in 1999. N Engl J Med. 2001;344:1807–1814. doi: 10.1056/NEJM200106143442401. [DOI] [PubMed] [Google Scholar]
- 29.Asnis DS, Conetta R, Teixeira AA, Waldman G, Sampson BA. The West Nile Virus outbreak of 1999 in New York: the Flushing Hospital experience [published correction appears in Clin Infect Dis. 2000;30:841] Clin Infect Dis. 2000;30:413–418. doi: 10.1086/313737. [DOI] [PubMed] [Google Scholar]
- 30.Solomon T, Vaughn DW. Pathogenesis and clinical features of Japanese encephalitis and West Nile virus infections. Curr Top Microbiol Immunol. 2002;267:171–194. doi: 10.1007/978-3-642-59403-8_9. [DOI] [PubMed] [Google Scholar]
- 31.Klein C, Kimiagar I, Pollak L. Neurological features of West Nile virus infection during the 2000 outbreak in a regional hospital in Israel. J Neurol Sci. 2002;200:63–66. doi: 10.1016/s0022-510x(02)00135-1. [DOI] [PubMed] [Google Scholar]
- 32.Weiss D, Carr D, Kellachan J, West Nile Virus Outbreak Response Working Group Clinical findings of West Nile virus infection in hospitalized patients, New York and New Jersey, 2000. Emerg Infect Dis. 2001;7:654–658. doi: 10.3201/eid0704.010409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ostlund EN, Andresen JE, Andresen M. West Nile encephalitis. Vet Clin North Am Equine Pract. 2000;16:427–441. doi: 10.1016/s0749-0739(17)30087-1. [DOI] [PubMed] [Google Scholar]
- 34.Han LL, Popovici F, Alexander JP., Jr Risk factors for West Nile virus infection and meningoencephalitis, Romania, 1996. J Infect Dis. 1999;179:230–233. doi: 10.1086/314566. [DOI] [PubMed] [Google Scholar]
- 35.Centers for Disease Control and Prevention Acute flaccid paralysis syndrome associated with West Nile virus infection—Mississippi and Louisiana, July-August 2002. MMWR Morb Mortal Wkly Rep. 2002;51:825–828. [PubMed] [Google Scholar]
- 36.Glass JD, Samuels O, Rich MM. Poliomyelitis due to West Nile virus [letter] N Engl J Med. 2002;347:1280–1281. doi: 10.1056/NEJM200210173471616. [DOI] [PubMed] [Google Scholar]
- 37.Kelley TW, Prayson RA, Ruiz AI, Isada CM, Gordon SM. The neuropathology of West Nile virus meningoencephalitis: a report of two cases and review of the literature. Am J Clin Pathol. 2003;119:749–753. doi: 10.1309/PU4R-76JJ-MG1F-81RP. [DOI] [PubMed] [Google Scholar]
- 38.Sejvar JJ, Stokic DS, Ven Gerpen JA. Acute flaccid paralysis and West Nile virus infection. Emerg Infect Dis [serial online] 2003;9:788–793. doi: 10.3201/eid0907.030129. Available at: http://www.cdc.gov/ncidod/EID/vol9no7/03-0129.htm Accessibility verified July 30, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Solomon T, Ooi MH, Beasley DW, Mallewa M. West Nile encephalitis. BMJ. 2003;326:865–869. doi: 10.1136/bmj.326.7394.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sampson BA, Armbrustmacher V. West Nile encephalitis: the neuropathology of four fatalities. Ann N Y Acad Sci. 2001;951:172–178. [PubMed] [Google Scholar]
- 41.Campbell GL, Marfin AA, Lanciotti RS, Gubler DJ. West Nile virus. Lancet Infect Dis. 2002;2:519–529. doi: 10.1016/s1473-3099(02)00368-7. [DOI] [PubMed] [Google Scholar]
- 42.Lanciotti RS, Kerst AJ, Nasci RS. Rapid detection of West Nile virus from human clinical specimens, field-collected mosquitoes, and avian samples by a TaqMan reverse transcriptase-PCR assay. J Clin Microbiol. 2000;38:4066–4071. doi: 10.1128/jcm.38.11.4066-4071.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Huang C, Slater B, Rudd R. First isolation of West Nile virus from a patient with encephalitis in the United States. Emerg Infect Dis. 2002;8:1367–1371. doi: 10.3201/eid0812.020532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ben-Nathan D, Lustig S, Tam G, Robinzon S, Segal S, Rager-Zisman B. Prophylactic and therapeutic efficacy of human intravenous immunoglobulin in treating West Nile virus infection in mice. J Infect Dis. 2003;188:5–12. doi: 10.1086/376870. [DOI] [PubMed] [Google Scholar]
- 45.Shimoni Z, Niven MJ, Pitlick S, Bulvik S. Treatment of West Nile virus encephalitis with intravenous immunoglobulin [letter] Emerg Infect Dis. 2001;7:759. doi: 10.3201/eid0704.010432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fradin MS, Day JF. Comparative efficacy of insect repellents against mosquito bites. N Engl J Med. 2002;347:13–18. doi: 10.1056/NEJMoa011699. [DOI] [PubMed] [Google Scholar]
- 47.Centers for Disease Control and Prevention Epidemic/Epizootic West Nile Virus in the United States: Revised Guidelines for Surveillance, Prevention, and Control. April 2001:1-111. Available at: http://www.cdc.gov/ncidod/dvbid/westnile/resources/wnv-guidelines-apr-2001.pdf Accessibility verified July 30, 2003.


