Abstract
This article summarizes the state of the art in vaccine research against parasitic helminths in sheep and cattle. Optimization of antigen production (eg, recombinant expression) and antigen delivery, followed by extensive field testing, is still required for further development of vaccines. Helminth vaccines should sufficiently reduce parasite transmission to protect vaccinated animals against parasite-induced disorders and production loss. Vaccine efficacy requirements depend on the parasite’s epidemiology and farm management, both of which vary in different geographic regions and are influenced by climate. Vaccination is likely to be part of integrated worm control, together with other worm control measures.
Keywords: Gastrointestinal nematodes, Lungworm, Liver fluke, Vaccines
Key points
-
•
Only a few vaccines against parasitic helminths of livestock are currently on the market; namely those against Dictyocaulus viviparus in cattle, Haemonchus contortus in sheep, and the tapeworm Echinococcus granulosus in sheep.
-
•
Research is ongoing to develop vaccines based on purified or recombinantly expressed worm proteins for Ostertagia ostertagi and Cooperia oncophora in cattle, for Teladorsagia circumcincta and H contortus in sheep, and for Fasciola hepatica in ruminants.
-
•
Recombinant expression of worm antigens with the correct conformation to induce protective immunity is still a major challenge in vaccine development.
-
•
Vaccination is likely to be part of integrated worm control, together with other control measures, such as anthelmintic treatments, biological control, and grazing management.
-
•
Interdisciplinary research, including social sciences, should investigate drivers of farmers’ and veterinarians’ decision making, to optimize uptake of vaccination by the end users.
Introduction
The major helminth infections in ruminants include gastrointestinal (GI) nematodes, liver fluke, and lungworms. At present, they are mainly controlled by regular treatments with anthelmintic drugs. However, decades of intensive use of anthelmintics have led to the development of anthelmintic resistance. High levels of resistance against all classes of anthelmintics are reported in GI nematodes of small ruminants worldwide. Resistance against macrocyclic lactones is also emerging in cattle parasites, mainly in Cooperia spp and, to a lesser extent, Ostertagia ostertagi. Resistance against flukicides has been reported in Fasciola hepatica in several countries.
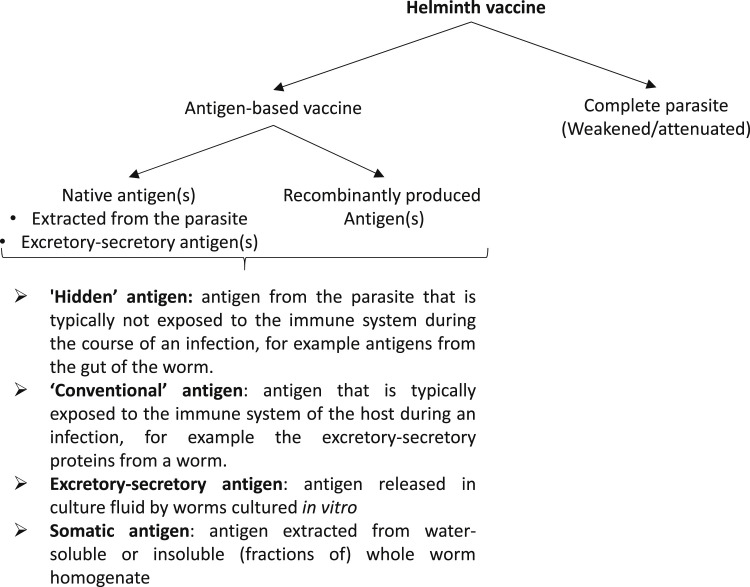
Together with a more sustainable use of anthelmintics, potential alternative (or complementary) control methods include bioactive forages, selective breeding for host resistance or resilience, nematophagous fungi, and vaccines.1 Vaccines are considered a favorable option, because of the durable protection they can provide and a lack of chemical residues in animal products and the environment.2 The principle of vaccination is to induce immunologic protection in a host against a subsequent infection. For this, the immune system can be stimulated with either the weakened or killed pathogen or proteins/antigens from it, the so-called subunit vaccines (Fig. 1 ). Subunit vaccines can be based on a single antigen or a mixture of antigens, which can be purified from the parasites or produced through recombinant DNA technology. The approach of producing a commercial vaccine straight from worm material is not applicable for most parasite species because it is practically difficult or even impossible to obtain large enough quantities of parasite material. For this reason, recombinant vaccines have been evaluated against a range of helminths. However, maintaining the correct conformation of target antigens in recombinant form is a major challenge,3 and only a few recombinant vaccine antigens have been shown to be protective. The challenge for further development lies in delivering these promising subunit vaccines in a manner feasible for large-scale commercial production, testing their efficacy in more extensive field trials, and in making them fit for purpose for commercial and end-user uptake.2
Fig. 1.
Different types of helminth vaccine antigens.
Despite strong efforts to develop antiparasitic vaccines, only 3 vaccines are currently on the market (Table 1 for specifications): (1) a live attenuated vaccine for control of the bovine lungworm, Dictyocaulus viviparus (Bovilis Huskvac, MSD Animal Health); (2) a native gut antigen-based vaccine, recently launched for the control of haemonchosis in sheep in Australia (Barbervax, Wormvax Australia Pty Ltd) and South Africa (Wirevax, Afrivet Business Management Pty Ltd); and (3) a cestode vaccine (Providean Hidatil EG95, Tecnovax) is available on the commercial market in parts of South America for the control of Echinococcus granulosus in sheep and goats.
Table 1.
Commercialized vaccines against helminth parasites in ruminants
| Brand Name | Parasite | Animal Species | Antigen | Adjuvant | Administration Route | Vaccination Schedule | Level of Protection | Withdrawal Period |
|---|---|---|---|---|---|---|---|---|
| Bovilis Huskvac | Dictyocaulus viviparus | Cattle | Live irradiated third-stage larvae | None | Oral | Basic scheme: 2 vaccinations with 4-wk interval before turnout Revaccination: single dose before turnout |
95%–98% (worm burden) | None |
| Barbervax, Wirevax | Haemonchus contortus | Sheep | Gut membrane proteins | Saponin | Subcutaneous | Basic scheme: 3 vaccinations with interval of 3–4 wk before Haemonchus season Revaccination: 6-wk intervals |
93%–95% (egg counts) 72%–94% (worm burden) |
None |
| Providean Hidatil EG95 | Echinococcus granulosus | Cattle, sheep, goats, camelids | EG95 | Montanide and saponin | Subcutaneous or intramuscular | 2 vaccinations with 1-mo interval | 95%–100% (cysts) | None |
This article summarizes the state of the art in vaccine research against sheep and cattle helminths, including the lungworm D viviparus, the GI nematodes Haemonchus contortus, Teladorsagia circumcincta, O ostertagi, and Cooperia oncophora, and the liver fluke F hepatica. Furthermore, safety and efficacy requirements for implementation of helminth vaccines in field conditions are discussed, as well as factors that may affect uptake of vaccines by the end users.
State of the art in vaccine development
Dictyocaulus viviparus
A commercial lungworm vaccine (Bovilis Huskvac, MSD Animal Health), composed of live irradiated third-stage (L3) D viviparus larvae, was developed decades ago.4 Two doses of 1000 viable irradiated larvae are delivered orally with an interval of approximately 4 weeks, and the full vaccination schedule should be completed at least 2 weeks before turnout of the vaccinated animals on pasture. Vaccinated cattle should not be treated with anthelmintics until at least 2 weeks after the second vaccination. The vaccine can be used in healthy cattle of 8 weeks of age or older, which includes grazing young stock and adult cows. Although vaccine-induced protection is generally good,5 the vaccine has disadvantages associated with live vaccines, such as ethical issues (production of larvae in donor animals), batch heterogeneity, and a short shelf life. In an attempt to overcome these issues, several attempts were made to develop recombinant subunit vaccines, with limited success. Vaccination with recombinant acetylcholinesterase,6 paramyosin,7 , 8 or asparaginyl peptidase legumain-19 with different adjuvants did not result in significant and/or reproducible reduction in worm numbers or larval shedding.
Haemonchus contortus
An H contortus vaccine based on a worm gut membrane antigen mixture was recently commercialized for sheep (Barbervax, Wirevax) and has quickly become an invaluable tool to control haemonchosis in areas where anthelmintic resistance is rampant.10 The vaccine is only registered in Australia and South Africa, but it can be used in the United Kingdom under Special Treatment Certificate and veterinary prescription. Vaccination with the vaccine also conferred protection against Haemonchus placei in grazing calves,11 but the vaccine has not been registered for use in cattle. Vaccination of young goats did not sufficiently protect against H contortus infections on pasture.12 For the production of this vaccine, the native antigens need to be extracted and purified locally from adult H contortus derived from infected sheep. A vaccine based on recombinantly expressed antigens would have advantages compared with a native antigen vaccine for reproducibility of product batches, biosafety, and global distribution. Many efforts have been made to identify and express a range of antigens from H contortus, but most recombinant proteins were unsuccessful in eliciting protective immunity.2 , 10 Recently, a gene encoding an Hc23 somatic antigen was identified and expressed in Escherichia coli. Lambs vaccinated with the recombinant Hc23 antigen were significantly protected against an artificial challenge infection, with more than 80% reduction in fecal egg counts (FECs) and average abomasal parasite burdens, compared with challenge controls.13 This work is a promising step toward a recombinant Haemonchus vaccine, but further experiments are needed to confirm these results and to test the vaccine in natural infection conditions.
Teladorsagia circumcincta
Similar to H contortus, over the years several antigens have been evaluated as vaccine candidates against the sheep abomasal parasite T circumcincta.2 The most promising results have recently been obtained with an experimental vaccine that is based on a cocktail of 8 recombinant proteins. The native versions of these antigens were originally identified either as immunodominant excretory/secretory proteins or selected because of their potential immune modulatory role or as homologues of vaccine candidates in related nematode species.2 All proteins were expressed in E coli or the yeast Pichia pastoris. Vaccination with this antigen cocktail with Quil A adjuvant protected 6- month-old to 7-month-old lambs and periparturient ewes against a challenge infection, resulting in significantly reduced FECs (73%–92% reduction in lambs, 45% in ewes) and worm burdens (56%–75% reduction in lambs).14 , 15 Work is ongoing to refine the protein cocktail by testing fewer recombinant protein combinations in vaccine trials.16 Further, its protective capacity will be tested in newly weaned lambs and in field trials.
Ostertagia ostertagi
Over the last 40 years, several antigens and antigen mixtures have been evaluated against O ostertagi (reviewed in Rinaldi and Geldhof17). At present the most promising experimental vaccine against O ostertagi is based on activation-associated secreted proteins (ASPs).2 , 18 Vaccination with a fraction of the adult excretory-secretory products containing ASP or with the purified protein has consistently resulted in a significantly reduced worm egg output (55%–62% reduction) after artificial trickle challenge infections.18, 19, 20, 21 Attempts to generate similar levels of protection with a recombinant ASP protein have been unsuccessful so far. Recombinant proteins, expressed in different expression systems, such as E coli, insect cells, and P pastoris, were either insoluble or did not protect vaccinated calves against challenge infection.3 , 22 Current research focuses on eliciting differences in protein folding and glycosylation between the native and recombinant ASP proteins, to identify epitopes that are important to induce a protective immune response.23 , 24
Cooperia oncophora
Based on the promising results obtained with an ASP-based vaccine against O ostertagi, a double-domain ASP (ddASP) protein purified from the excretory-secretory material of adult C oncophora worms was recently successfully evaluated as a vaccine candidate.25 , 26 Immunization with this antigen resulted in a significant reduction (91%) in cumulative FEC in an experimental challenge experiment. In a subsequent field trial, C oncophora cumulative FECs in vaccinates were reduced by 59%, resulting in 65% less infective C oncophora larvae on plots grazed by vaccinated calves and a significant reduction of 82% in worm counts in the vaccinates at housing.26 Vaccination with the C oncophora ddASP also protected calves against a challenge infection with an Uruguayan isolate of mixed C oncophora and Cooperia punctata, suggesting that the protective epitopes are conserved between Cooperia species/isolates in different parts of the world. In contrast, the C oncophora ddASP vaccine did not protect sheep against Cooperia curticei (unpublished results). As with O ostertagi, the research is currently focused on the recombinant expression of the ASP in an immune-active form.
Fasciola Spp
An overview of individual antigens and combinations thereof that have been tested against the liver flukes F hepatica and Fasciola gigantica in cattle and sheep is given by Toet and colleagues.27 Some antigens, such as cathepsin L1 and leucine aminopeptidase (LAP), induced significant protection levels in sheep and/or cattle, both as native, purified antigen and as recombinant protein. However, protection levels were highly variable, hence the need to test each vaccine in multiple vaccine trials to show consistent protection.27 Combining different antigens, such as cathepsins, with LAP or hemoglobin, or vaccination with a chimeric protein composed of leucine aminopeptidase and cathepsin L1 has not substantially improved vaccine efficacy.27 , 28 Despite multiple individual studies showing promising results for various DNA vaccine candidates against fascioliasis in animal models, a systematic review and meta-analysis did not show a significant pooled efficacy for all vaccine candidates against fasciola infection.29
Vaccine safety, efficacy, and use of vaccines in parasite control
The ideal parasitic vaccine should be safe, have a high efficacy, and ideally this activity should extent to a wide range of parasites.30
Vaccine Safety
Safety is an absolute requirement for any new vaccine. To avoid unwanted contamination with other proteins or pathogens and to ensure batch reproducibility, recombinant protein or DNA vaccines would be preferable to vaccines consisting of native proteins, purified from worms that are collected from animals. Vaccination with a (recombinant) protein (cocktail) in combination with a well-defined commercial adjuvant is not expected to raise important safety issues. Nevertheless, the development of a human hookworm vaccine based on ASPs caused allergic reactions in vaccinated preexposed individuals.31 Several vaccines currently under experimental evaluation for livestock helminths also contain ASPs (O ostertagi, C oncophora, T circumcincta). No side effects were observed in calves that were vaccinated with a double-domain ASP from C oncophora before turnout on pasture,26 but it has not been tested yet whether this vaccine is safe to use in regions with year-long grazing, where animals are likely to be infected before vaccination. However, Nisbet and colleagues15 observed no adverse reactions in grazing ewes that were vaccinated with a recombinant T circumcincta vaccine containing ASP.
Vaccine Efficacy
The major aim in controlling helminth infections by vaccination is to reduce parasite transmission by decreasing the number of viable eggs that are excreted into the environment, because these determine the number of infective larvae on the pasture later in the grazing season. The fecundity of Ostertagia is highly regulated by host immunity,32 and fecal egg output can be strongly reduced without a reduction in worm numbers.33 In contrast, the fecundity of Haemonchus is not regulated by the intensity of the infection and there is a good correlation between total daily FECs and mature female worm burden.34 Consequently, to prevent the buildup of a high pasture infection level, a Haemonchus vaccine needs to reduce the number of adult worms present in the animals early in the grazing season, either by reducing the establishment of infective larvae or by increasing the mortality of established worms.30
An advantage of vaccines based on hidden antigens (see Fig. 1) is that they can produce protective effects in situations in which natural immunity is either weak or ineffective, such as in young lambs or in periparturient ewes. A disadvantage is that vaccine-induced immunity is not boosted by a challenge infection, because the hidden antigens in the ingested worms are not exposed to the host’s immune system. As a consequence, immunity induced by vaccination with hidden antigens is typically short lasting and animals need to be revaccinated regularly. This need is shown by the vaccination schedules for H contortus in sheep. When lambs first receive Barbervax, 3 doses of the vaccine with intervals of 3 to 4 weeks are required to reach an effective level of antibodies, whereas subsequent vaccinations are given each 6 weeks until H contortus infections are no longer a risk (www.wormboss.com.au).
Animals vaccinated with conventional antigens (see Fig. 1) benefit from restimulation of their immune systems by the corresponding antigens from the challenge infection. The level and duration of protection required depend on the local grazing management and climate conditions.35
In sheep, both lambs and their dams need to be vaccinated. The ewes contribute significantly to the pasture infection because of their periparturient increase in egg shedding, and the lambs are vulnerable because of a lack of acquired immunity. Therefore, experimental T circumcincta vaccines are tested in both these age categories,14 , 15 and separate vaccine schedules were designed for lambs and ewes for the commercial H contortus vaccine (www.barbervax.com.au).
In dairy cattle, calves are typically separated from their dams at birth and the heifer calves are raised as a separate group of replacement stock. First grazing season calves are the most susceptible to infections with GI nematodes, and parasitic gastroenteritis is mainly seen in this age class when no appropriate preventive measures have been taken. In many parts of Europe, there is a well-defined grazing period, from spring to autumn. Helminth-naive calves should be vaccinated before turnout and the vaccine should protect them until natural immunity has sufficiently developed. Natural immunity against C oncophora develops within the first months of grazing, but immunity development against O ostertagi is slow,36 and vaccination should protect calves during their entire first grazing season. Because the peak egg output occurs around 2 months after turnout,37 it can be anticipated that reduction of worm egg shedding in the first 2 to 3 months after turnout may be sufficient to prevent accumulation of infective larvae on pasture and to protect vaccinated animals until housing in autumn.35 The authors previously suggested that a vaccine that reduces the mean fecal egg output by around 60% during the first 2 months after turnout would sufficiently protect calves against GI nematodes during their first grazing season and allow them to develop a natural immunity without production loss.30 For C oncophora, this has been shown to be an achievable goal,26 but whether this assumption holds for O ostertagi in field conditions remains to be confirmed. Moreover, this expectation does not take into account individual variability of each calves’ response to vaccination.
Required levels of vaccine efficacy and duration of protection depend on the host-parasite system and the grazing management, both of which are influenced by climate. Short-term protection may suffice for GI nematodes of cattle in regions with a restricted grazing season, such as in Europe.26 In regions with continuous grazing throughout the year (eg, South America and New Zealand), the required duration of protection offered by vaccination may be similar for parasites that rapidly induce natural immunity, such as Cooperia, but longer protection by vaccination may be needed for parasites with a slower immune development, such as Ostertagia and Fasciola.
F hepatica infection levels greater than 30 to 40 flukes in cattle and 30 to 54 flukes in sheep have been associated with production losses.38, 39, 40 In order to reduce fluke burdens below these threshold values, and taking into account reported fluke burdens in different countries, vaccine efficacy against Fasciola should reach 50% to 80% according to Toet and colleagues.27 However, it was not specified whether this reduction in fluke burden should be obtained through a direct effect of vaccination on worm viability or indirectly by reducing worm egg output in vaccinated animals. Estimating an indirect effect of reduced egg shedding on adult fluke burdens is complicated by the role of the intermediate snail host in parasite transmission. Because a snail infected with a single miracidium can produce several hundred cercariae, the snail biology has a significant impact on the outcome of vaccination.
Vaccines Against Multiple Pathogens
In most regions, grazing ruminants are coinfected with several pathogenic worm species; for example, H contortus, T circumcincta, and Trichostrongylus spp in sheep, and in cattle O ostertagi and Cooperia spp, plus others at lower levels. Multivalent vaccines, protecting against multiple GI nematodes and/or lungworms and liver fluke, could have an obvious advantage compared with single-species vaccines.2 However, monovalent vaccines may be useful in situations in which a single parasite species dominates (eg, H contortus in warmer regions), when other parasites are controlled by alternative measures, or in regions where the risk for other parasites is low (eg, in dry regions with a low risk for liver fluke infection).2 Little or no research on multispecies helminth vaccines has been conducted, to our knowledge. No cross-protection was observed against O ostertagi when cattle were vaccinated against C oncophora with a ddASP vaccine26 despite the similarity of the ASP antigens of both worm species.
Helminth vaccines could also be combined with vaccines against other pathogens. However, vaccines against multiple pathogens are particularly useful when they tackle a common disease complex, such as neonatal bovine diarrhea (a combined vaccine against E coli, rotavirus, and coronavirus) or bovine respiratory disease (eg, vaccine against bovine respiratory syncytial virus, parainfluenza virus, and Mannheimia haemolytica). Parasitic gastroenteritis and lungworm disease are well-defined disease entities in grazing ruminants, and combining a GI nematode or lungworm vaccine with bacterial or viral antigens may have little advantage.1
Vaccination and Other Worm Control Measures
In many cases, vaccination is not a stand-alone solution, especially if vaccines are targeted at single parasite species and/or confer only partial protection. However, vaccination, even if only partially effective, could become an important component of integrated worm control programs.1 Anthelmintic treatment at particular time points during the vaccination schedule with the Barbervax vaccine is recommended in sheep, to cope with concurrent trichostrongylus infections or early haemonchus infections (www.barbervax.com.au). Because there is a large variability of immune responses to vaccination between individual animals,14 animals that respond poorly to vaccination may need to be identified and either treated or removed from the group. Likewise, herd immunity leads to reduced levels of pasture contamination that then helps protect those few animals that respond poorly. It would be an impossible task to test the huge number of possible scenarios arising from the combination of (multispecies) vaccines with other worm control measures in different management systems and climatic conditions in field vaccine trials. In contrast, helminth transmission models can be a useful tool for evaluating different scenarios in silico, and to select the most promising (combinations of) control methods for field testing.41 , 42 Barnes and colleagues43 modeled the population dynamics of Trichostrongylus colubriformis in sheep to predict the effect of different vaccines on worm population dynamics in grazing lambs. They concluded that, with vaccines based on conventional antigens, substantial benefits can be obtained with a decrease of 60% in larval establishment in 80% of the flock. These simulations have been referred to in several articles to argue that it is not essential for a vaccine against GI nematodes in ruminants to obtain 100% efficacy (or even >90%) in all animals. However, these results cannot be readily extrapolated to other host-parasite systems; for example, when suppressed egg production is the main effect of vaccine-induced immunity rather than decreased larval establishment.35 Turner and colleagues44 developed a mathematical model to simulate the effectiveness of liver fluke vaccines under field conditions. The model output suggests that current vaccine candidates have the potential to reduce the mean total fluke burden by 43% and mean daily egg output by as much as 99% under field conditions. However, to be effective, a vaccine should protect at least 90% of the animals during the whole grazing season. It seems unlikely that this level and duration of protection can be achieved with vaccination alone, but vaccines could contribute substantially toward fasciolosis control, reducing usage of anthelmintics and thus delaying the spread of anthelmintic resistance.44
Potential uptake of vaccines by end users
It is unlikely that the use of helminth vaccines will be imposed by policy makers, because helminth infections in livestock are considered as production diseases, without importance for public health or international trade.45 Consequently, the decision to vaccinate will be the farmer’s responsibility and among other things will depend on the vaccine’s performance and cost-effectiveness in comparison with alternative control measures, as shown by the success of the Barbervax vaccine. Despite the need to vaccinate sheep on a regular basis and to combine vaccination with anthelmintic treatments, farmers are willing to use the vaccine because anthelmintics alone have become insufficient to control haemonchus infections in regions with high levels of anthelmintic resistance. In contrast, the lungworm vaccine is not used extensively in many countries, despite good vaccine efficacy. Practical concerns, such as a short shelf life, and the availability of anthelmintics with a persistent efficacy against Dictyocaulus, apparently have made the vaccine a less attractive control option.
Although ultimately it may be the farmer’s decision to vaccinate, veterinarians have an important role to implement and improve vaccination strategies.46 More sociopsychological studies to identify drivers of farmers’ and veterinarians’ decision making will be crucial to optimize uptake of novel parasite control tools, such as vaccines.47
Future prospects and summary
Expressing recombinant vaccine antigens in the right conformation to induce a protective immune response remains an issue for many nematode vaccines. Studies on allelic variability of the target genes and on the protein conformation and secondary modifications of the antigens should help to steer the recombinant expression work and ultimately lead to the development of protective recombinant antigens. In addition, increasing knowledge on vaccine-induced immune responses will lead to optimized antigen-adjuvant combinations and improved vaccine delivery.2 The sustainability of vaccines, like anthelmintics, will depend on parasite evolution, and the ability of helminths to develop resistance to vaccine-induced host responses remains an open question.41 Vaccines containing multiple antigens of a single parasite species could slow down adaptation of the parasites to the vaccine, such as an experimental Teladorsagia vaccine in sheep that comprises multiple recombinant proteins.14 , 16
The effect of the level and duration of vaccine efficacy on parasite transmission and animal productivity and the combined use of vaccines and other control methods in different management systems and climate regions should be simulated using mathematical models to inform vaccine developers and regulatory authorities about possible outcome scenarios.
In parallel, consumer expectations about helminth vaccines should be investigated and drivers and inhibitors of farmers’ and veterinarians’ uptake of parasite vaccines should be identified to ensure that vaccines, once they are ready for commercialization, will be effectively used and incorporated into routine farm management and disease control.
Acknowledgments
This article is based on work funded by the European Union’s Horizon 2020 Research and Innovation Programme under grant agreement no. 635408EU (PARAGONE) and COST Action COMBAR CA16230, supported by COST (European Cooperation in Science and Technology).
Disclosure
The research on vaccine development against O ostertagi and C oncophora described in this article is financially supported by Zoetis.
References
- 1.Vercruysse J., Charlier J., van Dijk J. Control of helminth ruminant infections by 2030. Parasitology. 2018;45(13):1655–1664. doi: 10.1017/S003118201700227X. [DOI] [PubMed] [Google Scholar]
- 2.Matthews J.B., Geldhof P., Tzelos T. Progress in the development of subunit vaccines for gastrointestinal nematodes of ruminants. Parasite Immunol. 2016;38:744–753. doi: 10.1111/pim.12391. [DOI] [PubMed] [Google Scholar]
- 3.Geldhof P., De Maere V., Vercruysse J. Recombinant expression systems: the obstacle to helminth vaccines? Trends Parasitol. 2007;23:527–532. doi: 10.1016/j.pt.2007.08.012. [DOI] [PubMed] [Google Scholar]
- 4.Jarrett W.F., Jennings F.W., McIntyre W.I. Immunological studies on Dictyocaulus viviparus infection: active immunization with whole worm vaccine. Immunology. 1960;3:135–144. [PMC free article] [PubMed] [Google Scholar]
- 5.Benitez-Usher C., Armour J., Urquhart G.M. Studies on immunisation of suckling calves with dictol. Vet Parasitol. 1976;2:209–222. [Google Scholar]
- 6.Matthews J.B., Davidson A.J., Freeman K.L. Immunisation of cattle with recombinant acetylcholinesterase from Dictyocaulus viviparus and with adult worm ES products. Int J Parasitol. 2001;31:307–317. doi: 10.1016/s0020-7519(00)00157-0. [DOI] [PubMed] [Google Scholar]
- 7.Joekel D., Hinse P., Raulf M.K. Vaccination of calves with yeast- and bacterial-expressed paramyosin from the bovine lungworm Dictyocaulus viviparus. Parasite Immunol. 2015;37:614–623. doi: 10.1111/pim.12280. [DOI] [PubMed] [Google Scholar]
- 8.Strube C., Haake C., Sager H. Vaccination with recombinant paramyosin against the bovine lungworm Dictyocaulus viviparus considerably reduces worm burden and larvae shedding. Parasit Vectors. 2015;8:119. doi: 10.1186/s13071-015-0733-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Holzhausen J., Haake C., Schicht S. Biological function of Dictyocaulus viviparus asparaginyl peptidase legumain-1 and its suitability as a vaccine target. Parasitology. 2018;145:387–392. doi: 10.1017/S0031182017001573. [DOI] [PubMed] [Google Scholar]
- 10.Nisbet A.J., Meeusen E.N., Gonzalez J.F. Immunity to Haemonchus contortus and vaccine development. Adv Parasitol. 2016;93:353–396. doi: 10.1016/bs.apar.2016.02.011. [DOI] [PubMed] [Google Scholar]
- 11.Bassetto C.C., Silva M.R.L., Newlands G.F.J. Vaccination of grazing calves with antigens from the intestinal membranes of Haemonchus contortus: effects against natural challenge with Haemonchus placei and Haemonchus similis. Int J Parasitol. 2014;44:697–702. doi: 10.1016/j.ijpara.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 12.Meier L., Torgerson P.R., Hertzberg H. Vaccination of goats against Haemonchus contortus with the gut membrane proteins H11/H-gal-GP. Vet Parasitol. 2016;229:15–21. doi: 10.1016/j.vetpar.2016.08.024. [DOI] [PubMed] [Google Scholar]
- 13.Fawzi E.M., González-Sánchez M.E., Corral M.J. Vaccination of lambs with the recombinant protein rHc23 elicits significant protection against Haemonchus contortus challenge. Vet Parasitol. 2015;211:54–59. doi: 10.1016/j.vetpar.2015.04.029. [DOI] [PubMed] [Google Scholar]
- 14.Nisbet A.J., McNeilly T.N., Wildblood L.A. Successful immunization against a parasitic nematode by vaccination with recombinant proteins. Vaccine. 2013;31(37):4017–4023. doi: 10.1016/j.vaccine.2013.05.026. 23. [DOI] [PubMed] [Google Scholar]
- 15.Nisbet A.J., McNeilly T.N., Greer A.W. Protection of ewes against Teladorsagia circumcincta infection in the periparturient period by vaccination with recombinant antigens. Vet Parasitol. 2016;228:130–136. doi: 10.1016/j.vetpar.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 16.Nisbet A.J., McNeilly T.N., Price D.R.G. The rational simplification of a recombinant cocktail vaccine to control the parasitic nematode Teladorsagia circumcincta. Int J Parasitol. 2019;49:257–265. doi: 10.1016/j.ijpara.2018.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rinaldi M., Geldhof P. Immunologically based control strategies for ostertagiosis in cattle: where do we stand? Parasite Immunol. 2012;34:254–264. doi: 10.1111/j.1365-3024.2011.01313.x. [DOI] [PubMed] [Google Scholar]
- 18.Geldhof P., Vercauteren I., Gevaert K. Activation-associated secreted proteins are the most abundant proteins in a host protective fraction of Ostertagia ostertagi. Mol Biochem Parasitol. 2003;128:111–114. doi: 10.1016/s0166-6851(03)00044-6. [DOI] [PubMed] [Google Scholar]
- 19.Geldhof P., Claerebout E., Knox D.P. Vaccination of calves against Ostertagia ostertagi with cysteine proteinase enriched protein fractions. Parasite Immunol. 2002;24:263–270. doi: 10.1046/j.1365-3024.2002.00461.x. [DOI] [PubMed] [Google Scholar]
- 20.Geldhof P., Vercauteren I., Vercruysse J. Validation of the protective capacity of the Ostertagia ostertagi ES-thiol antigens with different adjuvantia. Parasite Immunol. 2004;26:37–43. doi: 10.1111/j.0141-9838.2004.00681.x. [DOI] [PubMed] [Google Scholar]
- 21.Meyvis Y., Geldhof P., Gevaert K. Vaccination against Ostertagia ostertagi with subfractions of the protective ES-thiol fraction. Vet Parasitol. 2007;149:239–245. doi: 10.1016/j.vetpar.2007.08.014. [DOI] [PubMed] [Google Scholar]
- 22.Geldhof P., Meyvis Y., Vercruysse J. Vaccine testing of a recombinant activation-associated secreted protein (ASP1) from Ostertagia ostertagi. Parasite Immunol. 2008;30:57–60. doi: 10.1111/j.1365-3024.2007.01001.x. [DOI] [PubMed] [Google Scholar]
- 23.Borloo J., Geldhof P., Peelaers I. Structure of Ostertagia ostertagi ASP-1: insights into disulfide mediated cyclization and dimerization. Acta Crystallogr D Biol Crystallogr. 2013;69:493–503. doi: 10.1107/S0907444912050019. [DOI] [PubMed] [Google Scholar]
- 24.Meyvis Y., Callewaert N., Gevaert K. Hybrid N-glycans on the host protective activation-associated secreted proteins of Ostertagia ostertagi and their importance in immunogenicity. Mol Biochem Parasitol. 2008;161:67–71. doi: 10.1016/j.molbiopara.2008.05.004. [DOI] [PubMed] [Google Scholar]
- 25.Borloo J., Claerebout E., De Graef J. In-depth proteomic and glycomic analysis of the adult-stage Cooperia oncophora excretome/secretome. J Proteome Res. 2013;12(9):3900–3911. doi: 10.1021/pr400114y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Vlaminck J., Borloo J., Vercruysse J. Vaccination of calves against Cooperia oncophora with a double-domain activation-associated secreted protein reduces parasite egg output and pasture contamination. Int J Parasitol. 2015;45:209–213. doi: 10.1016/j.ijpara.2014.11.001. [DOI] [PubMed] [Google Scholar]
- 27.Toet H., Piedrafita D.M., Spithill T.W. Liver fluke vaccines in ruminants: strategies, progress and future opportunities. Int J Parasitol. 2014;44:915–927. doi: 10.1016/j.ijpara.2014.07.011. [DOI] [PubMed] [Google Scholar]
- 28.Ortega-Vargas S., Espitia C., Sahagún-Ruiz A. Moderate protection is induced by a chimeric protein composed of leucine aminopeptidase and cathepsin L1 against Fasciola hepatica challenge in sheep. Vaccine. 2019;37:3234–3240. doi: 10.1016/j.vaccine.2019.04.067. [DOI] [PubMed] [Google Scholar]
- 29.Jayaraj R., Kumarasamy C., Norbury L. Protective efficacy of liver fluke DNA vaccines: a systematic review and meta-analysis: Guiding novel vaccine development. Vet Parasitol. 2019;267:90–98. doi: 10.1016/j.vetpar.2019.01.010. [DOI] [PubMed] [Google Scholar]
- 30.Claerebout E., Knox D.P., Vercruysse J. Current research and future prospects in the development of vaccines against gastrointestinal nematodes in cattle. Expert Rev Vaccines. 2003;2:147–157. doi: 10.1586/14760584.2.1.147. [DOI] [PubMed] [Google Scholar]
- 31.Diemert D.J., Pinto A.G., Freire J. Generalized urticaria induced by the Na-ASP-2 hookworm vaccine: implications for the development of vaccines against helminths. J Allergy Clin Immunol. 2012;130:169–176. doi: 10.1016/j.jaci.2012.04.027. [DOI] [PubMed] [Google Scholar]
- 32.Smith G., Grenfell B.T., Anderson R.M. The regulation of Ostertagia ostertagi populations in calves: density-dependent control of fecundity. Parasitology. 1987;95:373–388. doi: 10.1017/s0031182000057814. [DOI] [PubMed] [Google Scholar]
- 33.Gasbarre L.C. Effects of gastrointestinal nematode infection on the ruminant immune system. Vet Parasitol. 1997;72:327–343. doi: 10.1016/s0304-4017(97)00104-0. [DOI] [PubMed] [Google Scholar]
- 34.Coyne M.J., Smith G. Trichostrongylid parasites of domestic ruminants. In: Scott M.E., Smith G., editors. Parasitic and infectious diseases: epidemiology and ecology. Academic Press; San Diego (CA): 1994. pp. 235–256. [Google Scholar]
- 35.Vercruysse J., Claerebout E. Assessment of the efficacy of helminth vaccines. J Parasitol. 2003;89(Suppl):S202–S209. [Google Scholar]
- 36.Claerebout E., Vercruysse J. The immune response and the evaluation of acquired immunity against gastrointestinal nematodes in cattle: a review. Parasitology. 2000;120:S25–S42. doi: 10.1017/s0031182099005776. [DOI] [PubMed] [Google Scholar]
- 37.Shaw D.J., Vercruysse J., Claerebout E. Gastrointestinal nematode infections of first-season grazing calves in Western Europe: general patterns and the effect of chemoprophylaxis. Vet Parasitol. 1998;75:115–131. doi: 10.1016/s0304-4017(97)00214-8. [DOI] [PubMed] [Google Scholar]
- 38.Dargie J.D. The impact on production and mechanisms of pathogenesis of trematode infections in cattle and sheep. Int J Parasitol. 1987;17:453–463. doi: 10.1016/0020-7519(87)90121-4. [DOI] [PubMed] [Google Scholar]
- 39.Hawkins C.D., Morris R.S. Depression of productivity in sheep infected with Fasciola hepatica. Vet Parasitol. 1978;4:341–351. [Google Scholar]
- 40.Hope Cawdery M.J., Strickland K.L., Conway A. Production effects of liver fluke in cattle. I. The effects of infection on liveweight gain, feed intake and food conversion efficiency in beef cattle. Br Vet J. 1977;133:145–159. doi: 10.1016/s0007-1935(17)34136-2. [DOI] [PubMed] [Google Scholar]
- 41.Morgan E.R., Aziz N.-A.A., Blanchard A. 100 important research questions in livestock helminthology. Trends Parasitol. 2018;35(1):52–71. doi: 10.1016/j.pt.2018.10.006. [DOI] [PubMed] [Google Scholar]
- 42.Verschave S.H., Charlier J., Rose H. Cattle and nematodes under global change: transmission models as an ally. Trends Parasitol. 2016;32(9):724–738. doi: 10.1016/j.pt.2016.04.018. [DOI] [PubMed] [Google Scholar]
- 43.Barnes E.H., Dobson R.J., Barger I.A. Worm control and anthelmintic resistance: adventures with a model. Parasitol Today. 1995;11:56–63. doi: 10.1016/0169-4758(95)80117-0. [DOI] [PubMed] [Google Scholar]
- 44.Turner J., Howell A., McCann C. A model to assess the efficacy of vaccines for control of liver fluke infection. Sci Rep. 2016;6:23345. doi: 10.1038/srep23345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Charlier J., Vande Velde F., van der Voort M. ECONOHEALTH: placing helminth infections of livestock in an economic and social context. Vet Parasitol. 2015;212:62–67. doi: 10.1016/j.vetpar.2015.06.018. [DOI] [PubMed] [Google Scholar]
- 46.Cresswell E., Brennan M.L., Barkema H.W. A questionnaire-based survey on the uptake and use of cattle vaccines in the UK. Vet Rec Open. 2014;1:e000042. doi: 10.1136/vropen-2014-000042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vande Velde F., Charlier J., Claerebout E. Farmer behaviour and gastrointestinal nematodes in ruminant livestock–uptake of sustainable control approaches. Front Vet Sci. 2018;5:255. doi: 10.3389/fvets.2018.00255. [DOI] [PMC free article] [PubMed] [Google Scholar]