Abstract
HIV infects the central nervous system (CNS) during primary infection and persists in resident macrophages. CNS infection initiates a strong local immune response that fails to control the virus but is responsible for by-stander lesions involved in neurocognitive disorders. Although highly active anti-retroviral therapy now offers an almost complete control of CNS viral proliferation, low-grade CNS inflammation persists. This review focuses on HIV-induced intrathecal immunoglobulin (Ig) synthesis. Intrathecal Ig synthesis early occurs in more than three-quarters of patients in response to viral infection of the CNS and persists throughout the course of the disease. Viral antigens are targeted but this specific response accounts for < 5% of the whole intrathecal synthesis. Although the nature and mechanisms leading to non-specific synthesis are unknown, this prominent proportion is comparable to that observed in various CNS viral infections. Cerebrospinal fluid-floating antibody-secreting cells account for a minority of the whole synthesis, which mainly takes place in perivascular inflammatory infiltrates of the CNS parenchyma. B-cell traffic and lineage across the blood–brain-barrier have not yet been described. We review common technical pitfalls and update the pending questions in the field. Moreover, since HIV infection is associated with an intrathecal chronic oligoclonal (and mostly non-specific) Ig synthesis and associates with low-grade axonal lesions, this could be an interesting model of the chronic intrathecal synthesis occurring during multiple sclerosis.
Keywords: Human immunodeficiency virus, Encephalopathy, Cerebrospinal fluid, Immunoglobulins, Intrathecal synthesis
Highlights
-
•
Intrathecal Ig synthesis occurs early after HIV infection and never subsides.
-
•
Only a small fraction of intrathecal Ig is directed against HIV antigens (~ 5%).
-
•
Non-specific Igs are directed against unknown targets but are poorly stidied.
-
•
CNS B-cells are compartmentalized but their traffic and maturation are poorly understood.
-
•
HIV infection may be a valuable model of chronic CSF inflammation.
1. Introduction
Brain invasion by HIV occurs as an early event, taking place during the primary infection (Davis et al., 1992, Gray et al., 1992, Gray et al., 1996, Hassine et al., 1995). It first involves HIV-infected CD4 T cells crossing the blood–brain-barrier (BBB) (Davis et al., 1992, Martin-Blondel et al., 2011) and HIV then infects the perivascular macrophages and microglia (Koenig et al., 1986, Martin-Blondel et al., 2011). Inflammatory cytokines secreted by these infected cells increase the permeability of the BBB. Two main mechanisms may be involved in central nervous system (CNS) complications: 1) the direct viral infection of CNS cells which initiates a cascade of immune reactions to control viral replication and leads to by-stander CNS lesions; and 2) opportunistic infections and lymphomas that complicate the prolonged immune suppression and evolve on their own. As a consequence, a large range of cognitive disorders was associated with HIV before appropriate drugs were made available. The proportion of involved patients markedly increases with the disease stage (Antinori et al., 2007). For example, 15% of asymptomatic HIV-infected patients suffer from neuropsychological (NP) impairment (Antinori et al., 2007), but during CDC stages A, B and C, the proportion of cognitive impairment rises from 30 to 50% (Antinori et al., 2007). Highly active anti-retroviral therapy (HAART), a combination of several drugs, was introduced at the end of the 90s and offered a new way of controlling viral infection, thereby preventing further complications of prolonged immune suppression from occurring and dramatically modifying the course of the disease.
In pre-HAART era, HIV-associated neurocognitive disorders (HAND) mainly occurred in patients with a CD4 count below 200/mm3 owing to confounding opportunistic complications. Moreover, the high burden of opportunistic infectious complications made their analysis difficult. However, even in the HAART era, HAND may occur in patients having a normal CD4 count and low plasma viral load (Antinori et al., 2007, Heaton et al., 2010). Although the risk of developing HAND decreased in the HAART era, the prevalence of HIV infection also increased owing to the longer survival and the increasing age of surviving patients.
The occurrence of brain lesions in HAART-treated patients with a well-controlled CD4 count appeared paradoxical and led to the notion of short CNS escapes (‘blips’). However, cognitive impairment also occurs in excess in cohorts of aviremic patients highly selected to exclude any confounding factors (i.e., illicit drug abuse, psychiatric and opportunistic history) (Simioni et al., 2010). Even in more recent cohorts of well-controlled patients, progressive atrophy occurs early and is still evident in the basal ganglia and the cortex, and the brain metabolites are diffusely changed (Chiang et al., 2007, Becker et al., 2012, Kallianpur et al., 2012, Kallianpur et al., 2013, Ragin et al., 2012, Towgood et al., 2012, Gongvatana et al., 2013).
The burden of cognitive impairment even in patients under HAART without (demonstrable) viral replication in the cerebrospinal fluid (CSF) suggests that a specific mechanism is triggered in the CNS. Several trials assessing neuroprotective drugs targeting various pathways have been set up, even in optimally controlled HIV-infected patients (review in (Simioni et al., 2010, Simioni et al., 2011)). HIV is a non-cytopathic virus that replicates in host cells without interfering with the processes essential for cellular survival and mostly failing to infect cells of CNS lineage (oligodendrocytes, astrocytes and neurons). Instead, CNS disease is mostly due to a chronic immune activation and response to infection, as commonly observed in many animal models of neurotropic viral infections (Subak-Sharpe et al., 1993, Mokhtarian et al., 2003, Mecha et al., 2013). Furthermore, this compartmentalized immune reaction triggered by HIV infection shares many aspects with the reaction observed during MS such as intrathecal secretion of oligoclonal antibodies, local proliferation of lymphocytes and microglial activation. Progressive brain atrophy is a salient feature in both disorders, and although progressive MS and HIV HAND dramatically differ as to their causes, it appears plausible that they may trigger a non-specific compartmentalized immune reaction, finally leading to an (at least partly) common degenerative pathway (Table 4 ). However, the full cascade of mechanisms leading to HAND remains to be elucidated (Hong and Banks, 2015).
Table 4.
Mechanisms of HIV neuropathology.
| Demonstrated mechanisms | |
|---|---|
| Confounding factors | HIV-related factors |
| Psycho-social conditions –mood disorders –psychoactive drug abuse –lack of education |
CNS cell infection (macrophages, microglia, ± astrocytes) |
| Antiviral drugs toxicity | BBB disruption and loss of pericytes |
| Aging | Neurotoxicity of HIV proteins (gp120, Nef) |
| Cerebrovascular disease | Neurotoxicity of resident macrophages/microglia via cytokine release (TNFα) |
| Nutritional and liver deficiencies | Infiltrating T-cells (exacerbated during IRIS) |
| Co-infections (CMV, JCV) and IRIS | |
| Hypothetic mechanisms |
|---|
| HIV-triggered autoimmune reaction? Local CNS lesions related to meningeal/perivascular tertiary lymphoid organs? Autoantibody neurotoxicity? Autoimmune T-cell reaction? Triggering of non-disease-specific neurodegenerative mechanisms? |
In common neurological practice, the most obvious aspect of HIV infection in CNS is the early triggered and lifelong sustained intrathecal immune reaction, which is indistinguishable from the intrathecal synthesis observed in a purely autoimmune disorder like multiple sclerosis (MS). This review focuses on evidence concerning intrathecal B-cell reaction and Ig synthesis during HIV infection. It then examines some of these unanswered questions in the light of the knowledge obtained from the field of CNS autoimmunity.
1.1. HIV invades CNS early and persists in resident cells
CNS infection occurs as an early event, probably by the entry of Trojan horse CD4-cells hijacked by the virus. Brain macrophages and microglia are then infected and serve as a sanctuary. CSF viral load strongly correlates with the expansion of perivascular macrophages both in human and animals (Fischer-Smith et al., 2008) and negatively correlates with CD8 + cell infiltration, contrary to lymphoid tissues (McCrossan et al., 2006). HIV-infected cells remain rare or undetectable in brain in most asymptomatic patients (Tavazzi et al., 2014).
Phylogenetic studies of brain viruses reveal that both canonical mutations underlying drug resistance and non-canonical mutations may be compartmentalized to brain areas, with HIV clones virtually private to some areas (Smit et al., 2004, Stam et al., 2013). This phenomenon is observed in all patients, independently of underlying neurological signs. CSF HIV decay following initiation of therapy combines two main mechanisms: a rapid decay (half-life: 2 days) corresponding to the end of the immigration of infected short-lived CD4 + T-cells coming from blood (in the absence of detectable CNS-compartmentalized viral production). Then in chronically infected patients, a slower decay (half-life: 14 days) corresponds to the clearance of perivascular macrophages infected with compartmentalized HIV variants (Harrington et al., 2005, Schnell et al., 2009).
However, CSF ‘escape’ may occur in otherwise well-controlled HIV infection (undetectable plasma HIV RNA) under HAART regimens and may co-occur with progressive neurologic involvement (Peluso et al., 2012). In such cases, high levels of HIV RNA copies are found in the CSF, often associated with multiple resistance mutations (Canestri et al., 2010, Peluso et al., 2012). MRI reveals non-specific sub-cortical or cerebellar lesions. Brain biopsy confirms the presence of dense, perivascular lymphocytic infiltrates in the white matter with extension to the surrounding parenchyma, intermingling mature and immature B and T lymphocytes, with CD8 + predominance (Peluso et al., 2012). When follow-up is available, clinical and radiological symptoms improve in all patients in whom CSF HIV RNA levels decrease in few weeks following drug adjustment (Canestri et al., 2010, Peluso et al., 2012). However, CSF escape is also observed in patients receiving drugs with good CNS penetration. The latter cases are due to an independent acquisition of drug resistance in CSF and autonomous local replication of resistant strains (Canestri et al., 2010, Eden et al., 2010), as illustrated by a longer time under HAART in CSF escape patients or treatment interruptions (Eden et al., 2010).
1.2. Blood–brain barrier impairment
BBB impairment is found in 24% of treated patients and 40% of untreated patients (Calcagno et al., 2014). The main predictors are a low CD4 nadir, a high compartmental viral replication and a higher intrathecal IgG synthesis (Calcagno et al., 2014), given that the two latter parameters are intimately linked. Plasma HIV load does not correlate with intrathecal immune parameters (Yilmaz et al., 2008). Since the proportion of BBB impairment increases along with HIV encephalitis and opportunistic complications, by-stander lesions of BBB by an inflammatory response may occur. Infection of pericytes and decreased expression of tight junctions disrupting endothelial layers have been reported, but a complete microanatomical explanation of BBB impairment is still lacking (Awan et al., 2014, Hill et al., 2014).
Whatever the exact mechanism, the main biological consequence of BBB impairment is that the concentration of blood-borne IgG in the CSF is increased, so the measurement of intrathecally synthesized IgG in the CSF is difficult (see Fig. 1 ). As widely described in various pathologies, BBB impairment should always be assessed in CSF studies (Reiber and Lange, 1991, Reiber and Peter, 2001). Direct measurement of CSF levels of various products (unless completely absent from serum) without normalization by BBB permeability would give inappropriate results and this technical pitfall should be avoided (Table 1).
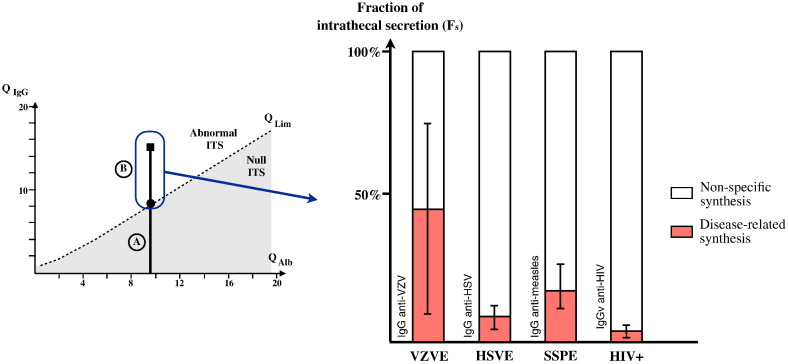
Fig. 1.
Specific and non-specific intrathecal synthesis (ITS).
Left panel. Null values of ITS are located in the shaded area. Much of the IgG level in CSF comes from a passive transudation from blood (A), proportional to BBB permeability (approximated by QAlb = AlbCSF / AlbSer). QLim(IgG) = (0.93 ∗ (QAlb2 − 6)0.5 − 1.7) ∗ 10− 3 is the maximal level of blood-borne CSF IgG. Levels of CSF IgG higher than QLim are intrathecally synthesized (B). (QAlb and QIgG are multiplied with 103).
Right panel. The fraction of intrathecally synthesized IgG (Fs) against specific viruses is plotted (mean and extreme values) among the whole intrathecally synthesized IgG in various CNS infections (Bonnan, 2014). Fs are commonly low in CNS infections and Fs against HIV is the lowest of the CNS infections. Hence, most of the intrathecally synthesized IgG in HIV infection are non-disease-specific. HSVE: HSV-encephalitis; SSPE: subacute sclerosis panencephalitis; VZVE: VZV-encephalitis.
Table 1.
Technical recommendations.
| *Specific Ig levels in CSF should be translated to standard dilution curves for antibody concentration but should no longer be expressed as optical density (OD) (Bukasa et al., 1988, Reiber and Lange, 1991). Given that QIgG cannot be correctly calculated by OD ratios, the simultaneous assay of CSF and serum samples to express the CSF/serum ratio circumvents the arbitrary use of units. |
| *Ig level in the CSF depends on BBB permeability, so most of the IgG detected in CSF are blood-borne due to passive diffusion, even at basal level. Intrathecal IgG synthesis should always be assessed by using QIgG and QAlb (Reiber and Peter, 2001). |
| *Specific intrathecal synthesis should be assessed by using Qspec. This allows the calculation of antibody index (AI) and specific fraction (Fs) (Jacobi et al., 2007). |
| *Control patients for intrathecal synthesis should be completely devoid of pathologies associated with intrathecal synthesis (i.e., multiple sclerosis). For example, up to 10% of MS patients display intrathecal synthesis against T. gondii which is part of a non-specific polyclonal reaction (Wildemann et al., 2010). |
1.3. Intrathecal IgG synthesis
IgG index is elevated in about 56–73% of patients (Chiodi et al., 1988a, Kaiser et al., 1989, McArthur et al., 1989, Singer et al., 1994, Gisslen et al., 1999a). In a recent study, mean IgG index was normal (0.63 ± 0.15) in HIV asymptomatic patients and elevated in HAND patients (0.97 ± 0.39) (Lackner et al., 2010). However, even if the median IgG index is normal in cohorts, the proportion of naïve patients with an abnormal IgG index is about 56% (Abdulle et al., 2005). The IgG index is globally stable (Elovaara et al., 1993b) or slowly increases in three stages: at less than one year, the mean IgG index is mainly normal, at 1–3 years, it increases to 1.05, and at more than 3 years it reaches 1.16 (Andersson et al., 1988, Marshall et al., 1989, Van Wielink et al., 1990). A possible decrease or reversion of intrathecal secretion may occur during late profound immunosuppression (Elovaara et al., 1987, Marshall et al., 1988, Singer et al., 1994, Gisslen et al., 1999a), unless patients are neurologically symptomatic (Elovaara et al., 1988). However, in cross-sectional studies, the proportion of patients with an abnormal IgG index or secretion of oligoclonal bands (OCB) remained roughly stable throughout the Walter Reed stages (Marshall et al., 1988).
Intrathecal synthesis is more polyclonal than oligoclonal with a stronger IgG staining in the CSF than in matching sera (Resnick et al., 1985, Chiodi et al., 1988a). OCB (≥ 2) are present in most HIV-infected patients when sensitive methods are used (Chiodi et al., 1988a, McArthur et al., 1989, Singer et al., 1994, Gisslen et al., 1999a). Serial examinations over years revealed a one-year latency from infection to the appearance of OCB (Goudsmit et al., 1986, Andersson et al., 1988). The number of OCB increases with the duration of HIV infection (Andersson et al., 1988) and varies in relation to the IgG index (Chiodi et al., 1988a, Hagberg et al., 1992).
Daily intrathecal IgG synthesis (Tourtellotte) is about 15.6 ± 18.5 mg to 43.7 ± 72.2 mg and is positively correlated with cognitive impairment (Singer et al., 1994). These results are in line with those obtained in MS where the intrathecal IgG synthesis rate is about 42 ± 24 mg/day (Tourtellotte et al., 1980) and the IgG index commonly ranges from 0.7 to 1.0 (Reiber et al., 2009).
Free light chains are more often elevated (85%) than IgG index or OCB, with conflicting results for prevalence of Lambda or Kappa FLC ratio (Gallo et al., 1990, Grimaldi et al., 1991). Levels of free light chains in the CSF was comparable or even higher than those in MS patients (Fagnart et al., 1988, Elovaara et al., 1991), and they remained stable throughout the stages before HAART (Elovaara et al., 1991). β2-Microglobulin (β2M), a component of MHC-I molecules, is a marker of immune activation, which is mainly driven by B-cell load and is observed at high levels in untreated HIV-infection. The CSF level increases linearly (from 1.3 to 1.7 mg/L) as the CD4 cell count decreases from > 1000 to 200 and abruptly rises when the latter drops below 200 (Lucey et al., 1991). β2-m CSF levels quickly normalize after HAART initiation in all patients (Abdulle et al., 2002). IgG index and CSF IgG concentration are proportional to CSF β2-microglobulin levels (Lucey et al., 1991).
In therapy-naïve patients, the intrathecally produced IgG fraction correlates with HIV load in the CSF (Cepok et al., 2007). In patients followed-up longitudinally up to 2 years after antiretroviral initiation, the proportion of elevated IgG index slightly decreases from 56% to 41% (Abdulle et al., 2005) or less (Yilmaz et al., 2006) while CD4 count is normalized and blood HIV RNA is lowered. However, HIV clearance from the CSF was incomplete in these studies since the median log of CSF HIV RNA decreased from 3.9–4.2 to only 1.6–1.7 (Abdulle et al., 2005), and older methods lacking sensitivity were used in other cases (Eden et al., 2007). Longitudinal data acquired with ultra-sensitive PCR upon intrathecal IgG synthesis in HIV patients free of CSF HIV replication for years are still lacking. The complete suppression of intrathecal IgG synthesis during long-term follow-up of HIV-RNA CSF-free patients may be expected but no data is yet available (Table 2).
Table 2.
HIV infection: toward a human model of chronic inflammation?
| Unique human infection associated with chronic CNS inflammation |
| Chronic long-standing intrathecal IgG synthesis with oligoclonal bands |
| Most HIV-induced CNS inflammation is non-specific to HIV |
| Natural history of CD4 flushes in CNS compartment |
| Complete control of infection under HAART |
1.4. Intrathecal HIV-specific IgG synthesis is frequently observed during HIV infection
Intrathecal secretion against specific HIV proteins has been found with variable frequencies (Goudsmit et al., 1987, Chiodi et al., 1988a, Chiodi et al., 1988b, Grimaldi et al., 1988, Kaiser et al., 1989, Lolli et al., 1990, Goswami et al., 1991, Fainardi et al., 2001). Intrathecal IgG OCB synthesis against HIV occurs in up to 70% of patients (Bukasa et al., 1988, Chiodi et al., 1988a, McArthur et al., 1989, Singer et al., 1994, Gisslen et al., 1999a, Fainardi et al., 2001) but OCB against HIV antigens are detected by immunoblotting in up to 87% (Bukasa et al., 1988, Dorries et al., 1989, Kaiser et al., 1990). IgGHIV antibody index is elevated (> 1.5) in the majority (75–85%) of patients from whom HIV-RNA is recovered from the CSF. IgGHIV specific index correlates weakly with IgG index but correlates better with advanced stages of the disease (Chiodi et al., 1988a, Emskoetter et al., 1989, Sonnerborg et al., 1989, Fainardi et al., 2001). In a series of 27 unselected HIV-infected patients, intrathecal synthesis was demonstrated in 86% of them (OCB, IEF-AMI against ≥ 1 antigen, AI-HIV) (Kaiser et al., 1989).
Intrathecal synthesis against env was detected in 81% of patients (9/11) and a high level of synthesis may occur in association with normal IgG index (Goudsmit et al., 1987). Local synthesis of anti-tat antibodies is detected in the CSF of HIV patients in 70–86% of cases, as early of the first year, but local secretion has not been calculated (Rodriguez et al., 2006, Bachani et al., 2013). Multiple serum samples over 6 years demonstrated a mostly stable anti-tat response: 60% and 33% persistently remained either positive or negative, whereas 7% changed their reactivity over time (Rodriguez et al., 2006). Anti-tat level is correlated very positively to normal cognition whereas lower anti-tat levels are associated with HAND (Bachani et al., 2013). This may be interpreted as a protective action of anti-tat through the neutralization of tat-mediated neurotoxicity. In a prospective cohort of HIV-2, 37% of anti-tat seronegative patients and 23% of anti-tat seropositive patients progressed to AIDS (Rodriguez et al., 2006). Unfortunately, no data was gathered on CSF anti-tat status (Rodriguez et al., 2006). Specific antibodies against HIV sometimes have an antibody-dependent cell cytotoxicity (ADCC) activity (Emskoetter et al., 1989). Intrathecal secretion of anti-HIV antibodies directed against env develops during asymptomatic stages, occurs in almost all patients and persists, whereas the reaction against pol and gag slightly decreases in the later stages (Chiodi et al., 1988a, Lolli et al., 1990, Goswami et al., 1991, Elovaara et al., 1993b, Fainardi et al., 2001). Results of assays of culture supernatants from free cells recovered from CSF showed IgG reactivity against p24 and env antigens (Amadori et al., 1988).
When the pattern of OCB is compared to that of anti-HIV IgG, numerous anti-IgG do not appear as anti-HIV bands, whereas faint IgG bands depict a strong anti-HIV specificity (Bukasa et al., 1988) (Fig. 2 ).
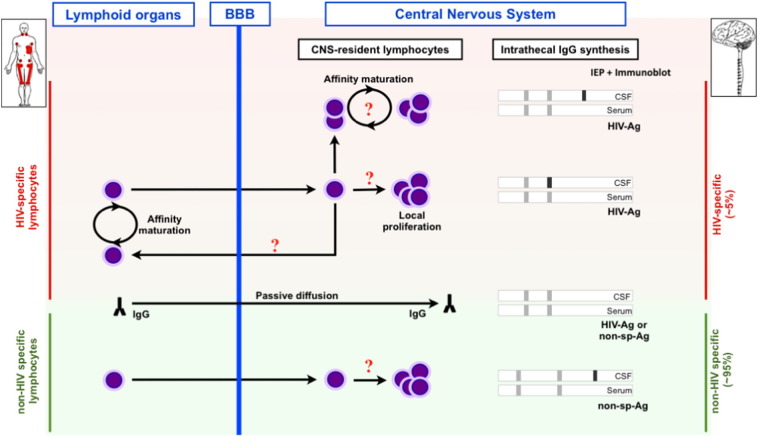
Fig. 2.
Lymphocytes traffic/maturation and intrathecal IgG synthesis associated with HIV infection in central nervous system (CNS). Upper panel. B-lymphocytes reacting against HIV are recruited in CNS where they may undergo a local proliferation; intrathecal IgG synthesis mirrors serum synthesis. Oligoclonal IgG synthesis restricted to CSF may be a consequence of local affinity maturation of B-lymphocytes. CSF IgG partly originates from passive diffusion through BBB and explains mirror pattern of OCB. Lower panel. Lymphocytes directed against non-HIV antigens are non-specifically recruited in the inflamed CNS and synthesize non-specific antibodies (i.e., anti-measles).
1.5. Intrathecal HIV-specific IgG synthesis represents a small part of all IgG synthesis occurring in the CNS
In the first study based on a quantitative ELISA assay, the proportion of intrathecal synthesis directed against HIV antigens was estimated to be 0.2 to 2.8% (Resnick et al., 1985). A more recent method uses the specific fraction (Fs) formula, which takes account of passive diffusion of specific IgG from blood to CSF (Jacobi et al., 2007). Expressed as a percentage, the Fs is the ratio of the intrathecally synthesized IgG fraction directed against a specific antigen and the total intrathecally synthesized IgG. On the other hand, the formula: 100 − Fs, expresses the percentage of non-specific intrathecally synthesized IgG. For example, an Fs value for HIV at 2% means that 2% of the total intrathecal IgG synthesis is directed against HIV, whereas 98% of IgG are not directed against it.
We applied this method for the first time in 8 patients on raw data provided by Resnick et al. (1985) and obtained an Fs against HIV of 4.3 ± 3.7% [0.48–12.7]. These low results are to be compared to the Fs obtained in various CNS disorders: 8.8% (3.5–12.5%) for HSV in HSV encephalitis (HSVE), 18.8% (11.8–27.5%) for measles in subacute sclerosis panencephalitis (SSPE) (Conrad et al., 1994, Jacobi et al., 2007) and 45% (13–73%) for VZV encephalitis (Otto et al., 2014). As a consequence, most of the intrathecal IgG synthesis associated with the immune response to CNS viral infection appears to be non-specific (up to 95% of the intrathecally produced IgG), and the range of Fs responses is even lower in HIV infection than in any other CNS infection (Fig. 1).
The frequency of HIV-specific B cells is very low in the blood of HIV-infected patients: < 0.01% of plasma blood mononuclear cells and 0.1–7% of IgG-secreting cells are reactive against HIV (review in (Doria-Rose and Connors, 2009)). Logically, since CNS is normally devoid of IgG synthesis, a far higher proportion of HIV-Ig-secreting cells may be expected from the CSF compartment during HIV infection of the CNS. Although no count is available, the low Fs value against HIV suggests that the proportion of HIV-specific cells is not large in CNS.
1.6. IgG synthesis originates in CNS-resident IgG-secreting cells but not in CSF floating cells
Flow cytometry of CSF floating cells reveals a comparable distribution of T cells and plasma cells between HIV-infected patients and controls, whereas B cells are significantly elevated (Cepok et al., 2007). Compared with controls, CD19 + B cells are elevated (~ 3% vs 0%), as are CD19 + CD138 + plasmablasts (~ 0.5% vs 0%) and CD19-CD138 + plasma cells (0.5% vs 0.8%) (Cepok et al., 2007). Plasmablasts are slightly more numerous in untreated patients (Cepok et al., 2007). CSF HIV viral load correlates with plasmablasts and B-cell recruitment in the CSF (Cepok et al., 2007). The extent of intrathecal synthesis correlates with CSF plasmablasts in all patients (Andersson et al., 1988, Cepok et al., 2007). HAART initiation is associated with the disappearance of both HIV load and regression of CSF plasmablasts (Spudich et al., 2005, Cepok et al., 2007) although the IgG index remains elevated (Abdulle et al., 2005, Yilmaz et al., 2006). Since CSF plasmablasts disappear after HAART initiation whereas CSF IgG synthesis is mostly preserved, IgG-secreting cells responsible for intrathecal IgG synthesis are probably resident within CNS inflammatory infiltrates. It is to note that the fate of parenchymal inflammatory infiltrates after HAART initiation is not fully understood.
Although floating CSF B-cells are commonly thought to be responsible for the intrathecal synthesis of IgG, such a low number of IgG-secreting cells cannot account for the bulk of synthesized IgG. Plasma cells are estimated to secrete around 0.3–2 × 104 IgG molecules·cell− 1·s− 1 (Lifter et al., 1976, Amanna and Slifka, 2010, Gomez et al., 2012) with an IgG molecular weight ≈ 160 kDa. Mean white cell count in CSFHIV is 12/mm3, and CSF subsets of plasma cells and plasmablasts are, respectively, 0.5% and 0.4–1.4% (Cepok et al., 2007). As a result of this higher estimation, the daily IgG synthesized in the CSF is in the range of ≈ 0.01 mg, which is far from the estimation based on Tourtellotte's formula (15–40 mg/d in CNS) (Singer et al., 1994), meaning that the bulk of IgG synthesis is provided by resident IgG-secreting cells residing either in the meninges or the perivascular areas.
1.7. B-cell traffic and CNS-resident IgG-secreting cell lineage are poorly understood
Infiltration of CNS by activated CD20 + CD23 + B-cells is known to occur at very low levels (0.1 B-cell/cm2 of parenchyma) for immune surveillance purposes but it never involves the perivascular areas (Anthony et al., 2003). Plasma cells are never observed in the normal brain parenchyma. During asymptomatic HIV infection, a mild focal lymphocytic infiltration occurs around the leptomeninges and around some white matter vessels. It is mainly composed of CD3 + T-cells with occasional parenchymal and perivascular B-cells (0.5 B-cell/cm2) (Anthony et al., 2003). During the course of AIDS, parenchymal B-cell infiltration decreases to a virtual absence of B-cell parenchymal and perivascular infiltration (Anthony et al., 2003), in contradiction with the persistence of intrathecal IgG synthesis. The same decrease in lymphocyte count is observed in the normal brain parenchyma outside primary brain lymphoma lesions in AIDS patients (Anthony et al., 2003). However, a sub-population of AIDS patients not associated with lymphomas demonstrates a high level of diffuse pleomorphic infiltrates with plasmacytoid B-cells in perivascular areas (0.2 B-cell/cm2 in the parenchyma and up to 15 B-cells/cm2 in perivascular areas) (Anthony et al., 2003). The preferential location of IgG-secreting cells has not been explicitly reported, nor has the correlation with IgG synthesis.
A class switch occurs mainly to IgG1 in MS (Lambin et al., 1991, Greve et al., 2001) but is less selective during HIV where the switch to both IgG1 and IgG2-4 occurs to the same extent (IgG1-4 indexes are elevated) (Lambin et al., 1991, Elovaara et al., 1993a). Data obtained from peripheral B-cells and IgG demonstrated a preferential mutation pattern of heavy chains that was characteristic of antigen-driven affinity maturation and clonal selection, as expected in a viral infection (Margolin et al., 2006).
Unfortunately, no data is available to date on the intimate clonal analysis of B-cell lineage in the CSF and brain of HIV patients, unlike in MS patients (Harp et al., 2007, Winges et al., 2007, Owens et al., 2009). CSF B-cell clonality was analyzed in a single HIV patient where 33% of CD138 + B-cells was clonal and homology to the closest germline was about 92% (82–98%, where 1% represents about 3 point mutations), in line with an antigen-driven extensive somatic mutation process (Owens et al., 2011). It is unknown whether extensive lineage analysis of IgG-VH would reveal clusters of clonally related B-cells in the CSF, cervical lymph nodes and/or blood, nor whether it would confirm or not a bidirectional exchange across the BBB, as already shown in MS (von Budingen et al., 2012). These preliminary results should be confirmed and complemented in a larger series of HIV-infected patients. Moreover, many fundamental questions remain unanswered. Enrichment of Jk2, Jk5 and VH1 genes was observed in HIV-specific B-cells recovered from blood but no data is available regarding CNS cells (Doria-Rose and Connors, 2009). Somatic hypermutation, preferential mutational targeting of RGYW/WRCY motifs in the CDR of heavy chains (which is typically targeted by the enzyme activation-induced cytidine deaminase during the affinity maturation process), and the Replacement:Silent (R:S) ratio in CDR and FDR fragments (which are typical of antigen-driven clonal selection) have not been studied in CNS B-cells to our knowledge and would be of major interest to demonstrate an intrathecal B-cell maturation process. Antigen-driven affinity maturation and clonal expansion of B-cells usually occur in primary and secondary lymphoid organs but may also occur in tertiary lymphoid organs (Humby et al., 2009, Neyt et al., 2012). Such structures are recovered from the meninges of MS patients and might account for the intrathecal IgG synthesis occurring in MS (Bonnan, 2014, Haugen et al., 2014). To date these structures have not been observed in the CNS during HIV infection, but HIV brains have never been explicitly examined. Therefore, it is still unknown whether intrathecal IgG-secreting cells are blood-borne or if they mature inside the CNS, although the association of both hypotheses is more likely.
1.8. Non-HIV-related intrathecal IgG synthesis
Serum hypergammaglobulinemia is a common finding during HIV infection and is either monoclonal, oligoclonal or polyclonal (Konstantinopoulos et al., 2007), the oligoclonal pattern being present in the serum of about 50% of patients when sensitive techniques like isoelectrofocusing are used (Laurijssens et al., 1993, del Bono et al., 1998). This synthesis is commonly attributed to HIV-induced non-specific B-cell activation due to T-cell depletion and virus-induced immune hyperactivation, but it mostly has little clinical consequence (Coker et al., 2013). Qualitative data on non-specific intrathecal synthesis have not yet been extensively examined and could be attributed to synthesis against auto-antigens, opportunistic infectious agents or against a broad non-HIV-related infectious agent. It might not even be due to antigen-driven antibodies.
From an historical point of view, the possibility of a coexistent intrathecal synthesis against a non-disease-relevant virus like measles has been considered so unlikely that older papers either excluded such patients or inappropriately used the ratio IgGCSF/IgGserum against measles to normalize data for passive diffusion (Lloyd et al., 1988, Mathiesen et al., 1988a, Van Wielink et al., 1990). Moreover, AIDS is characterized by a profound immune suppression impeding or even abolishing the capacity to develop an immune reaction against opportunistic infections. For example, intrathecal synthesis against Toxoplasma gondii may be abolished in CNS toxoplasmosis during AIDS but contradictory results have been obtained from different studies (Chiodi et al., 1988a). Specific synthesis against T. gondii is confirmed in a half of toxoplasmic encephalitis patients (Potasman et al., 1988).
1.8.1. Intrathecal synthesis against non-HIV-related infectious agents
The frequency of an MRZ pattern (measles, rubella, zoster) is thought to be absent during HIV infection and is almost absent in all acute CNS infections (review in (Bonnan, 2014)), whereas it is present in about 90% of MS patients (Bednárová et al., 2005, Jarius et al., 2009). Nevertheless, using an older methodology, a series of HIV patients was found to have intrathecal synthesis against measles in 9/12 patients and against CMV in 5/17 patients (Mathiesen et al., 1988b). In another series of 33 patients, ≥ 1 antibody against measles, HSV, VZV, CMV or T. gondii was detected by immunofixation in both sera and CSF of all patients without intrathecal synthesis, except for one patient who had a minor secretion against VZV (Chiodi et al., 1988a). Using an optimal methodology, Reiber et al. studying two HIV patients found no MRZ pattern but one displayed intrathecal synthesis against HSV (Reiber and Peter, 2001). In a series of 23 patients, intrathecal synthesis was demonstrated against HIV (22/23), rubella (7/22), HSV (10/22), VZV (12/22) and CMV (9/21) (Buffet et al., 1991), suggesting that a non-specific pattern could be common. It could be of major theoretical importance to study the MRZ pattern in this context since HIV offers a unique opportunity to investigate chronic intrathecal inflammation lasting for years or even decades.
1.8.2. Intrathecal synthesis against opportunistic infectious agents in AIDS
During toxoplasmic encephalitis in AIDS, intrathecal synthesis against T. gondii (ASI) was observed in 43–78% of patients, 78% had local oligoclonal synthesis, and a high antibody index persisted after recovery (Contini et al., 2000, Borges and Figueiredo, 2004). Free light chains showed local oligoclonal banding of Kappa type in 8/9 patients and of Lambda type in 4/9 patients, and these bands demonstrated specificity toward T. gondii antigens (Contini et al., 2000). It is to note that many articles, even recent ones, use inappropriate judgment criteria for intrathecal synthesis (Meira et al., 2013). However in AIDS patients, intrathecal synthesis against ongoing opportunistic infection may not occur and the diagnosis may be challenging. In syphilis-infected HIV patients displaying a negative CSF-VDRL, CXCL13 level in the CSF may be highly sensitive (Marra et al., 2010). Intrathecal synthesis against CMV antigen pp150 occurs in 26% (9/35) of unselected HIV patients (pre-HAART), which is in the same range of CMV lesions observed at autopsy (Weber et al., 1994). Patients who died from CMV encephalitis (pathologically confirmed) also commonly failed to mount an intrathecal Ig response (Weber et al., 1994). Therefore this synthesis may be interpreted as a scar of past CMV infection rather than as a non-specific synthesis.
1.8.3. Intrathecal synthesis against CNS auto-antigens
Anti-MOG antibodies have been reported in the CSF and serum of HIV patients (Lackner et al., 2010). The follow-up patterns of anti-MOG synthesis are variable: anti-MOG titers in the CSF after viral load clearance following the initiation of HAART may evolve either to a sharp increase, to stable titers along months or years, or over a late slow decrease of CSF titers (Lackner et al., 2010). This discordance between CSF viral clearance and anti-MOG antibodies titers suggests that, once initiated by CSF HIV infection, immunological mechanisms driving intrathecal synthesis are mainly independent from the sustain of CSF infection (Lackner et al., 2010). Many other antibodies were reported in the CSF of HIV patients: IgG or IgM against GM1, GD1a, GD1b, sulfatide, MBP, MBP 68–84 fragment, cerebellar soluble lectin (Mathiesen et al., 1989, Hagberg et al., 1992, De Gasperi et al., 1996, Gisslen et al., 2000) whereas anti-sulfatide could not be found in CSF by others (Gisslen et al., 1999b).
It should be mentioned that intrathecal synthesis of specific antibodies was not explicitly calculated in many studies, and although most of the QAlb remained low, one cannot completely exclude a passive diffusion from blood (Mathiesen et al., 1989, Hagberg et al., 1992, Gisslen et al., 1996, Gisslen et al., 2000, Lackner et al., 2010). It remains of major theoretical importance to study a possible intrathecal synthesis against auto-antigens using unbiased techniques before making definitive conclusions.
1.9. Supportive data from T-cell analysis
Perivascular infiltrates are heterogeneous and are dominated by CD8 + and CD4 + T-cells in perivascular areas, whereas B-cells are present in low proportions (Petito et al., 2003, Petito et al., 2006, Gray et al., 2013). One question is whether perivascular T-cells randomly infiltrating cells originate from blood or result locally from the antigen-driven clonal expansion of T-cells (Lin et al., 2005)? The sequencing of β-chain TCR transcripts from brain perivascular T-cells confirms a massive oligoclonality whereas transcripts recovered from normal blood donors are always unique (Lin et al., 2005). None of the identified TCR sequences were previously identified, nor were they known to react against HIV. Interestingly those clones exhibit a substantial CDR3 homology to clones directed against MBP- and tetanus toxoid-specific class II restricted T-cell clones, and against T-cell clones isolated from MS plaques or from patients with Rasmussen's encephalitis (Lin et al., 2005). A substantial CDR3 homology to MBP-specific β-chain TCR was present in 26/141 sequenced β-chains (Lin et al., 2005). Most of these clones were present in one patient who exhibited myelin pallor that was not observed in other patients. Moreover, patients exhibiting clones against MBP also share a common classical HLA DQB1*0602 and DRB1*1503 (which is only one amino acid different from *1501) (Lin et al., 2005). As a consequence, T-cells infiltrating blood vessels from the brain in pediatric patients may be autoimmune rather than antiviral cell clones. The inflammatory response is associated with a very low HIV viral load and infiltrative T cells are not associated with multinucleated giant cells. Data are lacking to confirm whether clonally expanded MBP-specific T-cell clones may be attributed to epitope spreading.
1.10. CSF cytokines and soluble markers
Besides immunoglobulins, cytokines are produced intensely in the CNS during HIV infection. Although it is unclear how locally synthesized cytokines contribute to CNS pathology, a growing set of evidence suggests that they participate in lesions (i.e., encephalitis, vacuolar myelopathy) (Tyor et al., 1993, Kobayashi et al., 1997, Saha and Pahan, 2003). We briefly review the main cytokines involved in lymphocytic traffic, macrophage activation and axonal lesion markers.
The chemokine CXCL10 is a CXCR3 ligand that is expressed by T-cells and NK-cells and acts as a major chemo-attractor in inflammatory conditions. The CXCL10 level in the CSF is consistently elevated in all HIV + patients, whatever the stage. It is involved in the lymphocyte count and promotes the recruitment of infected T-cells to the CSF (Kolb et al., 1999, Cinque et al., 2005, Spudich and Ances, 2011, Bremell et al., 2013). Paradoxically, this effect may therefore promote an amplification of CSF infection via importation of infected T-cells via a Trojan horse mechanism (Cinque et al., 2005). CSF CXCL10 levels normalize in less than 6 weeks after treatment initiation, although CSF HIV RNA remains elevated. After treatment interruption, CSF CXCL10 levels have increased by day 25–50 (Cinque et al., 2005).
CSF CXCL13 elevation is far lower during HIV infection than during neurosyphilis in HIV patients or during Lyme infection in non-HIV patients, and a very high level may be a sensitive and specific surrogate marker of associated infection (Marra et al., 2010, Bremell et al., 2013).
CCL19 and CCL21, which are associated with secondary/tertiary lymphoid organs, are elevated in the serum of HIV-infected patients but have not yet been investigated in the CSF (Damas et al., 2009).
TNFα is elevated in CSF and TNFα-positive cells are found in perivascular cells and in microglia from basal ganglia and the parenchyma of gray and white matter (Kobayashi et al., 1997). However, TNFα + cells are mainly associated with severe cases of HIV-associated cognitive motor complex (Kobayashi et al., 1997, Seilhean et al., 1997). Future trials involving monoclonal antibodies against TNFα deserve consideration (del Palacio et al., 2012).
Neopterin is a marker of macrophage activation which is elevated in the CSF of all patients, whatever the CSF viral load (Yilmaz et al., 2006, Spudich and Ances, 2011). During follow-up after treatment initiation, neopterin progressively normalized in 14% and 55% of patients at one and two years, respectively (Abdulle et al., 2002, Yilmaz et al., 2006). A long-term study of patients receiving HAART and who were perfectly controlled (CSF and serum HIV RNA < 50 copies/mL) for ≥ 3.5 years provided evidence of persistent immune activation (Eden et al., 2007). Although CSF HIV RNA was undetectable and CSF WBC was normal, neopterin was low but still abnormal in 60% of patients (Eden et al., 2007). The IgG index was still elevated in 60% of patients under HAART as compared to 70% before (range: before 0.43–1.99; under HAART 0.46–3.05) (Eden et al., 2007). In a study based on an ultrasensitive PCR technique, neopterin was lowest in the group of patients without CSF HIV RNA (Yilmaz et al., 2008, Yilmaz et al., 2013). However, it remained abnormal in 41–57% of virologically well-suppressed (< 2.5 copies/mL) patients, even after 4 years of complete viral control (Yilmaz et al., 2008, Yilmaz et al., 2013). This suggests that although complete control of intrathecal infection is apparently obtained, a low-grade immune activation persists in the CNS. In a more recent study using a single-copy assay in patients under HAART, 87% of patients had no CSF HIV RNA (Dahl et al., 2014). However, ‘blips’ of detectable CSF HIV RNA occurred in a few patients with multiple samples. Interestingly these ‘blips’ were associated with a slight and transient increase in CSF neopterin (Dahl et al., 2014), suggesting that the goal of complete viral suppression was still not attained. This persistent immunoactivation might mean that a low-level viral replication may still occur in the CSF under the limit of detection of ultrasensitive PCR techniques (2.5 copies/mL). This possibility may link persistent immunoactivation with silent and transient CSF viral escapes.
In the long-term follow-up of elite controllers (known duration of infection of 17 years), CSF concentrations of neopterin, CXCL10 and MCP1 were in the same range as those of HIV-negative control patients (Probasco et al., 2010). However, since these patients were spontaneous controllers, they probably never developed CNS HIV-infection, even before the HAART era. As a consequence, the absence of CSF inflammation in these patients may better be interpreted as a spontaneous resistance to the establishment of CSF/CNS inflammation than as proof of the vanishing of an earlier inflammatory state. It is still unknown whether prolonged persistence of intrathecal low-grade immune activation is harmful or not (Yilmaz et al., 2008).
The kinetics of CSF change has been studied in patients stopping the combination of antiretroviral therapies (Gisslen et al., 2005). Plasma HIV load first increases, followed after about 3 weeks by an increase in CSF HIV load and then by an increase in CSF white cell and damage markers (neopterin and neurofilament) (Gisslen et al., 2005). The lag between CSF viral replication and neuronal damage as measured with NFL suggests an evolving immunopathological process initiated by a surge in viral replication (Gisslen et al., 2005).
Neurofilament (NFL), a marker of axonal degeneration, was found to increase in the CSF of HIV-infected patients as immune suppression increased: 19% in patients with normal CD4 count to 93% in HAD patients (Jessen Krut et al., 2014). NFL levels are far higher in HIV dementia than in non-demented HIV patients (Gisslen et al., 2007, Jessen Krut et al., 2014). Interestingly, the increase in NFL level in the CSF always precedes the clinical presentation (Gisslen et al., 2007) and decreases a few months after treatment initiation in half of patients at month 3 and in three quarters at year 1 (Abdulle et al., 2007, Mellgren et al., 2007, Jessen Krut et al., 2014). Under HAART, CSF NFL levels decrease in most patients to normal, even after adjustment for age (Jessen Krut et al., 2014).
1.11. Intrathecal synthesis in animal models
Very few data have been obtained from animal models owing to the relative resistance of animals to HIV brain infection, so they are poor models of HIV encephalitis and not suitable for viral-induced demyelination.
HIV does not infect rodents in natural conditions. The most promising model is based on immune-depleted mice repopulated with human CD34 + hematopoietic stem cells (Gorantla et al., 2012). Although murine immunopathology differs from that of human since humanized mice lack human stromal cells (Gorantla et al., 2012), this model provides a good approach to HIV encephalitis. The animals show cognitive defects, brain macrophages are infected by HIV, and dendritic arborization of neurons surrounding infected macrophages or microglia is decreased, assuming opportunistic infection never occurs (Sas et al., 2007). No data is available on CNS IgG synthesis in this model.
Simian immunodeficiency syndrome (SIV) infection causes an AIDS-like syndrome in rhesus macaques (Macacca mulatta) (Smith et al., 1994). Infection of brain tissue is demonstrated by PCR after 1–2 days post-inoculation and is constant by 7 days, although infected cells are too rare to be observed by in situ hybridization (Milush et al., 2013). At death, intrathecal synthesis is demonstrated by an elevated IgG index in 21% and the synthesis of SIV-specific IgG in 12% (Smith et al., 1994). However, SIV-infected macaques die from non-CNS disorders shortly after inoculation (mean 220 days) and the pathological lesions of the CNS are undemonstrative (Smith et al., 1994). In a different study longitudinally analyzing CSF after inoculation, IgG index strongly increased between weeks 1 and 15 and sharply decreased or even normalized thereafter. Quinolinic acid CSF concentrations peaked at 2 weeks and then normalized (Smith et al., 1995). Animals sacrificed at week 2 had meningitis, glial nodules and perivascular cuffs of mononuclear cells (Smith et al., 1995). When rhesus macaques were infected by the viral isolate SIVmac251, classical neurological signs associated with AIDS appeared with groups of rapid or slow progressors (Sopper et al., 1998). Intrathecal IgG synthesis was not observed at 48 weeks, but virus-specific antibodies against env et gag were recovered in CSF (Sopper et al., 1998). Interestingly, rapid progressors did not demonstrate any virus-specific Ig response whereas the CSF response in slow progressors paralleled the serum response (Sopper et al., 1998). Env-specific antibody secreting cells accounted for 15% of CNS-derived plasma cells and seemed to stabilize at around 10%, corresponding to 7000 env-specific CNS plasma cells per brain (Sopper et al., 1998). High CSF viral load was associated with rapid progression and the monkeys failed to develop any intrathecal response, suggesting a protective role of intrathecal immune response (Sopper et al., 1998). However, this finding needs to be replicated. Moreover, an earlier experiment using SIVmac251 showed that intrathecal synthesis of anti-env started only 70 weeks after infection.
Interestingly, early infusion of natalizumab has been shown to reduce the viral burden of CNS macrophages by preventing HIV-infected monocytes/macrophages from entering the brain (Campbell et al., 2014) but the consequences on intrathecal synthesis were not examined.
1.12. Could intrathecal Ig synthesis associated with HIV infection be a model of some aspects of chronic autoimmune CNS inflammation?
We now set aside the direct consequences of viral infection and focus on HIV-induced chronic CNS inflammation. Three main aspects should be considered.
First, HIV-infection is sufficiently frequent to require a public health policy. In fact it is the last chronic CNS infection while all the traditional infections have already disappeared thanks to modern medicine. Neuro-syphilis and tuberculous meningitis have almost disappeared whereas neuro-borreliosis is treated early. Meningo-encephalitis from other causes is either acute and curable or quickly fatal. As a consequence, HIV infection is a unique long-lasting human model of chronic CNS inflammation.
Secondly, HIV-induced CNS inflammation has not been completely deciphered since research has focused pragmatically on virus-induced lesions. However, non-specific immune reactions are common during HIV-infection. Non-specific synthesis is also common in chronic autoimmune CNS inflammation and some cases are considered to be characteristic of chronic autoimmunity. For example, a non-specific synthesis against measles, rubella and zoster (so-called ‘MRZ reaction’) characteristically occurs during early MS in patients who never previously experienced any encephalitis, so it cannot be considered as serological scars. The proportion of patients positive for MRZ increases over time (Brecht et al., 2012), suggesting that MRZ positivity is more a non-specific function of time in chronic inflammation than an intrinsic characteristic of MS (Bonnan, 2014). Long-standing HIV-infected patients provide an ideal model of chronic inflammation that may provide decisive evidence in the field of autoimmunity.
Thirdly, the natural history of untreated HIV is an immune suppression secondary to profound CD4 cell loss leading to AIDS and frequently complicated by opportunistic infections. HAART almost completely controls HIV infection and reverses immune suppression. As a consequence, HIV infection during AIDS can also be considered to be an experimental situation of transient extreme flushing of CD4 cells from the CNS. Interestingly, a monoclonal antibody directed against VL4, natalizumab, prevents transmigration of T-cells to the CNS and is used to treat MS. Intrathecal IgG synthesis and MRZ secretion may decrease in MS patients receiving natalizumab (Warnke et al., 2014). This is a major theoretical issue since HIV infection may lower the relative risk of multiple sclerosis (Gold et al., 2015).
2. Conclusion
Intrathecal synthesis against HIV is highly prevalent during HIV infection even in non-neurologically impaired patients. Besides this IgG synthesis directed against HIV, most of this synthesis is non-specific, as observed in blood. The etiology and consequences of this chronic intrathecal reaction are unknown. The rigorous study of this chronic inflammation without any a-priori expectations and with techniques used in multiple sclerosis might be an avenue worth exploring (Table 3).
Table 3.
Pending questions.
| *Does intrathecal IgM synthesis occur during HIV infection? |
| *Is intrathecal IgG synthesis suppressed during long-term CSF HIV eradication (confirmed with ultra-sensitive PCR) or does infection trigger a sustained self-loop immune reaction in CNS? |
| *Does intrathecal non-disease-related CSF IgG synthesis (i.e., the ‘MRZ’ pattern) really occur during HIV infection as in MS? |
| *Is non-specific IgG synthesis apparently supported by antigen-driven affinity maturation? |
| *Does the synthesis against non-relevant antigens (i.e., MRZ pattern), as commonly observed in MS, also occur in HIV-infected patients? |
| *Where are the CNS-resident IgG-secreting cells mainly situated (white matter parenchyma, basal ganglia, leptomeninges)? Are plasma cells isolated or associated with lymphoid structures? |
| *Are tertiary lymphoid organs (TLO) present in the CNS of HIV-infected patients? Does IgG affinity maturation occur in the CNS before cell migration through the BBB or is traffic two-way through it? |
| *Does chronic specific/non-specific intrathecal IgG synthesis play a role in the pathophysiology of some CNS lesions? Are there any slow-rate chronic CNS lesions around IgG-secreting cell infiltrates? |
| *Do CNS infiltrates of plasma cells (and therefore local IgG synthesis) correlate with basal ganglia or cortical atrophy/lesions as observed on MRI (Chiang et al., 2007)? |
| *Some of the typical markers of oxidative injuries (p22phox, oxidized phospholipids and iron) occurring in MS are also recovered in animal models of coronavirus infection, making chronic viral infection an interesting model of progressive MS lesions (Schuh et al., 2014). Besides the mimicking of some aspects of MS pathology, the coronavirus model is also characterized by a major intrathecal IgG synthesis, as in MS pathology and in HIV infection (Dorries et al., 1986, Schwender et al., 1991). Could these oxidative markers also be recovered in chronic CNS viral infection as in human HIV? |
| *Do the sub-pial lesions observed in MS and spatially correlated with meningeal TLO also exist in association with meningeal TLO in HIV infection? Such lesions are revealed only by anti-MBP or anti-PLP immunochemistry but not by classical staining (Bø et al., 2003). Sub-pial lesions have never been described but have only been researched in AIDS patients with PML in the vicinity of macroscopic PML lesions (Moll et al., 2008). Unfortunately, PML in AIDS probably precludes any cortical immune reaction owing to a profound immune suppression. Studies on well-controlled HIV patients would be more relevant. |
| *More generally, does the intrathecal immune reaction associated with HIV infection throw any light on chronic autoimmune intrathecal disorders like MS? |
Conflict of interest
The authors declare that they have no conflict of interest.
Acknowledgment
We are indebted to Ray Cooke for copy-editing and to Christel Baigts and Aurora Medevielle for bibliographical assistance.
References
- Abdulle S., Hagberg L., Svennerholm B., Fuchs D., Gisslen M. Continuing intrathecal immunoactivation despite two years of effective antiretroviral therapy against HIV-1 infection. AIDS. 2002;16:2145–2149. doi: 10.1097/00002030-200211080-00006. [DOI] [PubMed] [Google Scholar]
- Abdulle S., Hagberg L., Gisslen M. Effects of antiretroviral treatment on blood–brain barrier integrity and intrathecal immunoglobulin production in neuroasymptomatic HIV-1-infected patients. HIV Med. 2005;6:164–169. doi: 10.1111/j.1468-1293.2005.00281.x. [DOI] [PubMed] [Google Scholar]
- Abdulle S., Mellgren A., Brew B.J., Cinque P., Hagberg L., Price R.W. CSF neurofilament protein (NFL) — a marker of active HIV-related neurodegeneration. J. Neurol. 2007;254:1026–1032. doi: 10.1007/s00415-006-0481-8. [DOI] [PubMed] [Google Scholar]
- Amadori A., De Rossi A., Gallo P., Tavolato B., Chieco-Bianchi L. Cerebrospinal fluid lymphocytes from HIV-infected patients synthesize HIV-specific antibody in vitro. J. Neuroimmunol. 1988;18:181–186. doi: 10.1016/0165-5728(88)90065-3. [DOI] [PubMed] [Google Scholar]
- Amanna I.J., Slifka M.K. Mechanisms that determine plasma cell lifespan and the duration of humoral immunity. Immunol. Rev. 2010;236:125–138. doi: 10.1111/j.1600-065X.2010.00912.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersson M.A., Bergstrom T.B., Blomstrand C., Hermodsson S.H., Hakansson C., Lowhagen G.B. Increasing intrathecal lymphocytosis and immunoglobulin G production in neurologically asymptomatic HIV-1 infection. J. Neuroimmunol. 1988;19:291–304. doi: 10.1016/0165-5728(88)90010-0. [DOI] [PubMed] [Google Scholar]
- Anthony I.C., Crawford D.H., Bell J.E. B lymphocytes in the normal brain: contrasts with HIV-associated lymphoid infiltrates and lymphomas. Brain. 2003;126:1058–1067. doi: 10.1093/brain/awg118. [DOI] [PubMed] [Google Scholar]
- Antinori A., Arendt G., Becker J.T., Brew B.J., Byrd D.A., Cherner M. Updated research nosology for HIV-associated neurocognitive disorders. Neurology. 2007;69:1789–1799. doi: 10.1212/01.WNL.0000287431.88658.8b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Awan F.M., Anjum S., Obaid A., Ali A., Paracha R.Z., Janjua H.A. In-silico analysis of claudin-5 reveals novel putative sites for post-translational modifications: insights into potential molecular determinants of blood–brain barrier breach during HIV-1 infiltration. Infect. Genet. Evol. 2014;27:355–365. doi: 10.1016/j.meegid.2014.07.022. [DOI] [PubMed] [Google Scholar]
- Bachani M., Sacktor N., McArthur J.C., Nath A., Rumbaugh J. Detection of anti-tat antibodies in CSF of individuals with HIV-associated neurocognitive disorders. J. Neurovirol. 2013;19:82–88. doi: 10.1007/s13365-012-0144-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker J.T., Maruca V., Kingsley L.A., Sanders J.M., Alger J.R., Barker P.B. Factors affecting brain structure in men with HIV disease in the post-HAART era. Neuroradiology. 2012;54:113–121. doi: 10.1007/s00234-011-0854-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bednárová J., Stourac P., Adam P. Relevance of immunological variables in neuroborreliosis and multiple sclerosis. Acta Neurol. Scand. 2005;112:97–102. doi: 10.1111/j.1600-0404.2005.00418.x. [DOI] [PubMed] [Google Scholar]
- Bø L., Vedeler C.A., Nyland H.I., Trapp B.D., Mørk S.J. Subpial demyelination in the cerebral cortex of multiple sclerosis patients. J. Neuropathol. Exp. Neurol. 2003;62:723–732. doi: 10.1093/jnen/62.7.723. [DOI] [PubMed] [Google Scholar]
- Bonnan M. Does disease-irrelevant intrathecal synthesis in multiple sclerosis make sense in the light of tertiary lymphoid organs? Front. Neurol. 2014;5:27. doi: 10.3389/fneur.2014.00027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borges A.S., Figueiredo J.F. Evaluation of intrathecal synthesis of specific IgG antibodies against Toxoplasma gondii in the diagnosis assessment of presumptive toxoplasma encephalitis in AIDS patients. Rev. Soc. Bras. Med. Trop. 2004;37:480–484. doi: 10.1590/s0037-86822004000600010. [DOI] [PubMed] [Google Scholar]
- Brecht I., Weissbrich B., Braun J., Toyka K.V., Weishaupt A., Buttmann M. Intrathecal, polyspecific antiviral immune response in oligoclonal band negative multiple sclerosis. PLoS One. 2012;7:e40431. doi: 10.1371/journal.pone.0040431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bremell D., Mattsson N., Edsbagge M., Blennow K., Andreasson U., Wikkelso C. Cerebrospinal fluid CXCL13 in lyme neuroborreliosis and asymptomatic HIV infection. BMC Neurol. 2013;13:2. doi: 10.1186/1471-2377-13-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buffet R., Agut H., Chieze F., Katlama C., Bolgert F., Devillechabrolle A. Virological markers in the cerebrospinal fluid from HIV-1-infected individuals. AIDS. 1991;5:1419–1424. doi: 10.1097/00002030-199112000-00002. [DOI] [PubMed] [Google Scholar]
- Bukasa K.S., Sindic C.J., Bodeus M., Burtonboy G., Laterre C., Sonnet J. Anti-HIV antibodies in the CSF of AIDS patients: a serological and immunoblotting study. J. Neurol. Neurosurg. Psychiatry. 1988;51:1063–1068. doi: 10.1136/jnnp.51.8.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calcagno A., Alberione M.C., Romito A., Imperiale D., Ghisetti V., Audagnotto S. Prevalence and predictors of blood–brain barrier damage in the HAART era. J. Neurovirol. 2014;20:521–525. doi: 10.1007/s13365-014-0266-2. [DOI] [PubMed] [Google Scholar]
- Campbell J.H., Ratai E.M., Autissier P., Nolan D.J., Tse S., Miller A.D. Anti-alpha4 antibody treatment blocks virus traffic to the brain and gut early, and stabilizes CNS injury late in infection. PLoS Pathog. 2014;10:e1004533. doi: 10.1371/journal.ppat.1004533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canestri A., Lescure F.X., Jaureguiberry S., Moulignier A., Amiel C., Marcelin A.G. Discordance between cerebral spinal fluid and plasma HIV replication in patients with neurological symptoms who are receiving suppressive antiretroviral therapy. Clin. Infect. Dis. 2010;50:773–778. doi: 10.1086/650538. [DOI] [PubMed] [Google Scholar]
- Cepok S., von Geldern G., Nolting T., Grummel V., Srivastava R., Zhou D. Viral load determines the B-cell response in the cerebrospinal fluid during human immunodeficiency virus infection. Ann. Neurol. 2007;62:458–467. doi: 10.1002/ana.21195. [DOI] [PubMed] [Google Scholar]
- Chiang M.C., Dutton R.A., Hayashi K.M., Lopez O.L., Aizenstein H.J., Toga A.W. 3D pattern of brain atrophy in HIV/AIDS visualized using tensor-based morphometry. Neuroimage. 2007;34:44–60. doi: 10.1016/j.neuroimage.2006.08.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiodi F., Norkrans G., Hagberg L., Sonnerborg A., Gaines H., Froland S. Human immunodeficiency virus infection of the brain. II. Detection of intrathecally synthesized antibodies by enzyme linked immunosorbent assay and imprint immunofixation. J. Neurol. Sci. 1988;87:37–48. doi: 10.1016/0022-510x(88)90052-4. [DOI] [PubMed] [Google Scholar]
- Chiodi F., Sonnerborg A., Albert J., Gaines H., Norkrans G., Hagberg L. Human immunodeficiency virus infection of the brain. I. Virus isolation and detection of HIV specific antibodies in the cerebrospinal fluid of patients with varying clinical conditions. J. Neurol. Sci. 1988;85:245–257. doi: 10.1016/0022-510x(88)90184-0. [DOI] [PubMed] [Google Scholar]
- Cinque P., Bestetti A., Marenzi R., Sala S., Gisslen M., Hagberg L. Cerebrospinal fluid interferon-gamma-inducible protein 10 (IP-10, CXCL10) in HIV-1 infection. J. Neuroimmunol. 2005;168:154–163. doi: 10.1016/j.jneuroim.2005.07.002. [DOI] [PubMed] [Google Scholar]
- Coker W.J., Jeter A., Schade H., Kang Y. Plasma cell disorders in HIV-infected patients: epidemiology and molecular mechanisms. Biomark. Res. 2013;1:8. doi: 10.1186/2050-7771-1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad A.J., Chiang E.Y., Andeen L.E., Avolio C., Walker S.M., Baumhefner R.W. Quantitation of intrathecal measles virus IgG antibody synthesis rate: subacute sclerosing panencephalitis and multiple sclerosis. J. Neuroimmunol. 1994;54:99–108. doi: 10.1016/0165-5728(94)90236-4. [DOI] [PubMed] [Google Scholar]
- Contini C., Fainardi E., Cultrera R., Seraceni S., Castellazzi M., Peyron F. Evidence of cerebrospinal fluid free kappa light chains in AIDS patients with Toxoplasma gondii encephalitis. J. Neuroimmunol. 2000;108:221–226. doi: 10.1016/s0165-5728(00)00280-0. [DOI] [PubMed] [Google Scholar]
- Dahl V., Peterson J., Fuchs D., Gisslen M., Palmer S., Price R.W. Low levels of HIV-1 RNA detected in the cerebrospinal fluid after up to 10 years of suppressive therapy are associated with local immune activation. AIDS. 2014;28:2251–2258. doi: 10.1097/QAD.0000000000000400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damas J.K., Landro L., Fevang B., Heggelund L., Froland S.S., Aukrust P. Enhanced levels of the CCR7 ligands CCL19 and CCL21 in HIV infection: correlation with viral load, disease progression and response to highly active antiretroviral therapy. AIDS. 2009;23:135–138. doi: 10.1097/QAD.0b013e32831cf595. [DOI] [PubMed] [Google Scholar]
- Davis L.E., Hjelle B.L., Miller V.E., Palmer D.L., Llewellyn A.L., Merlin T.L. Early viral brain invasion in iatrogenic human immunodeficiency virus infection. Neurology. 1992;42:1736–1739. doi: 10.1212/wnl.42.9.1736. [DOI] [PubMed] [Google Scholar]
- De Gasperi R., Angel M., Sosa G., Patarca R., Battistini S., Lamoreux M.R. Intrathecal synthesis of anti-sulfatide IgG is associated with peripheral nerve disease in acquired immunodeficiency syndrome. AIDS Res. Hum. Retrovir. 1996;12:205–211. doi: 10.1089/aid.1996.12.205. [DOI] [PubMed] [Google Scholar]
- del Bono V., Biselli R., Nisini R., Loomis-Price L.D., Loy A., Lorusso C. Isoelectricfocusing and reverse blotting as a diagnostic tool in pediatric HIV infection. J. Clin. Virol. 1998;11:203–210. doi: 10.1016/s0928-0197(98)00050-6. [DOI] [PubMed] [Google Scholar]
- del Palacio M., Alvarez S., Munoz-Fernandez M.A. HIV-1 infection and neurocognitive impairment in the current era. Rev. Med. Virol. 2012;22:33–45. doi: 10.1002/rmv.711. [DOI] [PubMed] [Google Scholar]
- Doria-Rose N.A., Connors M. Antibody-secreting B cells in HIV infection. Curr. Opin. HIV AIDS. 2009;4:426–430. doi: 10.1097/COH.0b013e32832d9fac. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorries R., Watanabe R., Wege H., ter Meulen V. Murine coronavirus-induced encephalomyelitis in rats: analysis of immunoglobulins and virus-specific antibodies in serum and cerebrospinal fluid. J. Neuroimmunol. 1986;12:131–142. doi: 10.1016/0165-5728(86)90026-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorries R., Kaiser R., ter Meulen V. Human immunodeficiency virus infection: affinity-mediated immunoblot detects intrathecal synthesis of oligoclonal IgG specific for individual viral proteins. AIDS Res. Hum. Retrovir. 1989;5:303–310. doi: 10.1089/aid.1989.5.303. [DOI] [PubMed] [Google Scholar]
- Eden A., Price R.W., Spudich S., Fuchs D., Hagberg L., Gisslen M. Immune activation of the central nervous system is still present after > 4 years of effective highly active antiretroviral therapy. J. Infect. Dis. 2007;196:1779–1783. doi: 10.1086/523648. [DOI] [PubMed] [Google Scholar]
- Eden A., Fuchs D., Hagberg L., Nilsson S., Spudich S., Svennerholm B. HIV-1 viral escape in cerebrospinal fluid of subjects on suppressive antiretroviral treatment. J. Infect. Dis. 2010;202:1819–1825. doi: 10.1086/657342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elovaara I., Iivanainen M., Valle S.L., Suni J., Tervo T., Lahdevirta J. CSF protein and cellular profiles in various stages of HIV infection related to neurological manifestations. J. Neurol. Sci. 1987;78:331–342. doi: 10.1016/0022-510x(87)90046-3. [DOI] [PubMed] [Google Scholar]
- Elovaara I., Seppala I., Poutiainen E., Suni J., Valle S.L. Intrathecal humoral immunologic response in neurologically symptomatic and asymptomatic patients with human immunodeficiency virus infection. Neurology. 1988;38:1451–1456. doi: 10.1212/wnl.38.9.1451. [DOI] [PubMed] [Google Scholar]
- Elovaara I., Seppala I., Kinnunen E., Laaksovirta H. Increased occurrence of free immunoglobulin light chains in cerebrospinal fluid and serum in human immunodeficiency virus-1 infection. J. Neuroimmunol. 1991;35:65–77. doi: 10.1016/0165-5728(91)90162-z. [DOI] [PubMed] [Google Scholar]
- Elovaara I., Albert P.S., Ranki A., Krohn K., Seppala I. HIV-1 specificity of cerebrospinal fluid and serum IgG, IgM, and IgG1-G4 antibodies in relation to clinical disease. J. Neurol. Sci. 1993;117:111–119. doi: 10.1016/0022-510x(93)90163-s. [DOI] [PubMed] [Google Scholar]
- Elovaara I., Nykyri E., Poutiainen E., Hokkanen L., Raininko R., Suni J. CSF follow-up in HIV-1 infection: intrathecal production of HIV-specific and unspecific IGG, and beta-2-microglobulin increase with duration of HIV-1 infection. Acta Neurol. Scand. 1993;87:388–396. doi: 10.1111/j.1600-0404.1993.tb04123.x. [DOI] [PubMed] [Google Scholar]
- Emskoetter T., von Laer D., Veismann S., Ermer M. Human immunodeficiency virus (HIV)-specific antibodies, neutralizing activity and antibody-dependent cellular cytotoxicity (ADCC) in the cerebrospinal fluid of HIV-infected patients. J. Neuroimmunol. 1989;24:61–66. doi: 10.1016/0165-5728(89)90099-4. [DOI] [PubMed] [Google Scholar]
- Fagnart O.C., Sindic C.J., Laterre C. Free kappa and lambda light chain levels in the cerebrospinal fluid of patients with multiple sclerosis and other neurological diseases. J. Neuroimmunol. 1988;19:119–132. doi: 10.1016/0165-5728(88)90041-0. [DOI] [PubMed] [Google Scholar]
- Fainardi E., Contini C., Benassi N., Bedetti A., Castellazzi M., Vaghi L. Assessment of HIV-intrathecal humoral immune response in AIDS-related neurological disorders. J. Neuroimmunol. 2001;119:278–286. doi: 10.1016/s0165-5728(01)00386-1. [DOI] [PubMed] [Google Scholar]
- Fischer-Smith T., Bell C., Croul S., Lewis M., Rappaport J. Monocyte/macrophage trafficking in acquired immunodeficiency syndrome encephalitis: lessons from human and nonhuman primate studies. J. Neurovirol. 2008;14:318–326. doi: 10.1080/13550280802132857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallo P., Piccinno M.G., De Rossi A., Pagni S., Amadori A., Chieco-Bianchi L. Free light chains of immunoglobulins in the cerebrospinal fluid of human immunodeficiency virus type 1-infected patients. J. Neuroimmunol. 1990;26:229–238. doi: 10.1016/0165-5728(90)90005-8. [DOI] [PubMed] [Google Scholar]
- Gisslen M., Fredman P., Norkrans G., Hagberg L. Elevated cerebrospinal fluid sulfatide concentrations as a sign of increased metabolic turnover of myelin in HIV type I infection. AIDS Res. Hum. Retrovir. 1996;12:149–155. doi: 10.1089/aid.1996.12.149. [DOI] [PubMed] [Google Scholar]
- Gisslen M., Fuchs D., Svennerholm B., Hagberg L. Cerebrospinal fluid viral load, intrathecal immunoactivation, and cerebrospinal fluid monocytic cell count in HIV-1 infection. J. Acquir. Immune Defic. Syndr. 1999;21:271–276. doi: 10.1097/00126334-199908010-00003. [DOI] [PubMed] [Google Scholar]
- Gisslen M., Lekman A., Fredman P. High levels in serum, but no signs of intrathecal synthesis of anti-sulfatide antibodies in HIV-1 infected individuals with or without central nervous system complications. J. Neuroimmunol. 1999;94:153–156. doi: 10.1016/s0165-5728(98)00244-6. [DOI] [PubMed] [Google Scholar]
- Gisslen M., Lekman A., Fredman P. Cerebrospinal fluid antibodies directed against neuron-associated gangliosides in HIV-1 infection. Infection. 2000;28:143–148. doi: 10.1007/s150100050067. [DOI] [PubMed] [Google Scholar]
- Gisslen M., Rosengren L., Hagberg L., Deeks S.G., Price R.W. Cerebrospinal fluid signs of neuronal damage after antiretroviral treatment interruption in HIV-1 infection. AIDS Res. Ther. 2005;2:6. doi: 10.1186/1742-6405-2-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gisslen M., Hagberg L., Brew B.J., Cinque P., Price R.W., Rosengren L. Elevated cerebrospinal fluid neurofilament light protein concentrations predict the development of AIDS dementia complex. J. Infect. Dis. 2007;195:1774–1778. doi: 10.1086/518043. [DOI] [PubMed] [Google Scholar]
- Gold J., Goldacre R., Maruszak H., Giovannoni G., Yeates D., Goldacre M. HIV and lower risk of multiple sclerosis: beginning to unravel a mystery using a record-linked database study. J. Neurol. Neurosurg. Psychiatry. 2015;86:9–12. doi: 10.1136/jnnp-2014-307932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez A.M., Willcox N., Molenaar P.C., Buurman W., Martinez-Martinez P., De Baets M.H. Targeting plasma cells with proteasome inhibitors: possible roles in treating myasthenia gravis? Ann. N. Y. Acad. Sci. 2012;1274:48–59. doi: 10.1111/j.1749-6632.2012.06824.x. [DOI] [PubMed] [Google Scholar]
- Gongvatana A., Harezlak J., Buchthal S., Daar E., Schifitto G., Campbell T. Progressive cerebral injury in the setting of chronic HIV infection and antiretroviral therapy. J. Neurovirol. 2013;19:209–218. doi: 10.1007/s13365-013-0162-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorantla S., Poluektova L., Gendelman H.E. Rodent models for HIV-associated neurocognitive disorders. Trends Neurosci. 2012;35:197–208. doi: 10.1016/j.tins.2011.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goswami K.K., Kaye S., Miller R., McAllister R., Tedder R. Intrathecal IgG synthesis and specificity of oligoclonal IgG in patients infected with HIV-1 do not correlate with CNS disease. J. Med. Virol. 1991;33:106–113. doi: 10.1002/jmv.1890330208. [DOI] [PubMed] [Google Scholar]
- Goudsmit J., Wolters E.C., Bakker M., Smit L., Van der Noordaa J., Hische E.A. Intrathecal synthesis of antibodies to HTLV-III in patients without AIDS or AIDS related complex. Br. Med. J. (Clin. Res. Ed.) 1986;292:1231–1234. doi: 10.1136/bmj.292.6530.1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goudsmit J., Epstein L.G., Paul D.A., van der Helm H.J., Dawson G.J., Asher D.M. Intra-blood–brain barrier synthesis of human immunodeficiency virus antigen and antibody in humans and chimpanzees. Proc. Natl. Acad. Sci. U. S. A. 1987;84:3876–3880. doi: 10.1073/pnas.84.11.3876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray F., Lescs M.C., Keohane C., Paraire F., Marc B., Durigon M. Early brain changes in HIV infection: neuropathological study of 11 HIV seropositive, non-AIDS cases. J. Neuropathol. Exp. Neurol. 1992;51:177–185. doi: 10.1097/00005072-199203000-00007. [DOI] [PubMed] [Google Scholar]
- Gray F., Scaravilli F., Everall I., Chretien F., An S., Boche D. Neuropathology of early HIV-1 infection. Brain Pathol. 1996;6:1–15. doi: 10.1111/j.1750-3639.1996.tb00775.x. [DOI] [PubMed] [Google Scholar]
- Gray F., Lescure F.X., Adle-Biassette H., Polivka M., Gallien S., Pialoux G. Encephalitis with infiltration by CD8 + lymphocytes in HIV patients receiving combination antiretroviral treatment. Brain Pathol. 2013;23:525–533. doi: 10.1111/bpa.12038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greve B., Magnusson C.G., Melms A., Weissert R. Immunoglobulin isotypes reveal a predominant role of type 1 immunity in multiple sclerosis. J. Neuroimmunol. 2001;121:120–125. doi: 10.1016/s0165-5728(01)00436-2. [DOI] [PubMed] [Google Scholar]
- Grimaldi L.M., Castagna A., Lazzarin A., Pristera R., Bianchi G., Moroni M. Oligoclonal IgG bands in cerebrospinal fluid and serum during asymptomatic human immunodeficiency virus infection. Ann. Neurol. 1988;24:277–279. doi: 10.1002/ana.410240219. [DOI] [PubMed] [Google Scholar]
- Grimaldi L.M., Castagna A., Maimone D., Martino G.V., Dolci A., Pristera R. Kappa light chain predominance in serum and cerebrospinal fluid from human immunodeficiency virus type 1 (HIV-1)-infected patients. J. Neuroimmunol. 1991;32:259–268. doi: 10.1016/0165-5728(91)90196-e. [DOI] [PubMed] [Google Scholar]
- Hagberg L., Norkrans G., Andersson M., Wachter H., Fuchs D. Cerebrospinal fluid neopterin and beta 2-microglobulin levels in neurologically asymptomatic HIV-infected patients before and after initiation of zidovudine treatment. Infection. 1992;20:313–315. doi: 10.1007/BF01710672. [DOI] [PubMed] [Google Scholar]
- Harp C., Lee J., Lambracht-Washington D., Cameron E., Olsen G., Frohman E. Cerebrospinal fluid B cells from multiple sclerosis patients are subject to normal germinal center selection. J. Neuroimmunol. 2007;183:189–199. doi: 10.1016/j.jneuroim.2006.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrington P.R., Haas D.W., Ritola K., Swanstrom R. Compartmentalized human immunodeficiency virus type 1 present in cerebrospinal fluid is produced by short-lived cells. J. Virol. 2005;79:7959–7966. doi: 10.1128/JVI.79.13.7959-7966.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassine D., Gray F., Chekroun R., Chretien F., Marc B., Durigon M. Early cerebral lesions in HIV infection. Postmortem radio-pathologic correlations in non-AIDS asymptomatic seropositive patients. J. Neuroradiol. 1995;22:148–160. [PubMed] [Google Scholar]
- Haugen M., Frederiksen J.L., Degn M. B cell follicle-like structures in multiple sclerosis-with focus on the role of B cell activating factor. J. Neuroimmunol. 2014;273:1–7. doi: 10.1016/j.jneuroim.2014.05.010. [DOI] [PubMed] [Google Scholar]
- Heaton R.K., Clifford D.B., Franklin D.R., Jr., Woods S.P., Ake C., Vaida F. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER study. Neurology. 2010;75:2087–2096. doi: 10.1212/WNL.0b013e318200d727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill J., Rom S., Ramirez S.H., Persidsky Y. Emerging roles of pericytes in the regulation of the neurovascular unit in health and disease. J. Neuroimmune Pharmacol. 2014;9:591–605. doi: 10.1007/s11481-014-9557-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong S., Banks W.A. Role of the immune system in HIV-associated neuroinflammation and neurocognitive implications. Brain Behav. Immun. 2015;45:1–12. doi: 10.1016/j.bbi.2014.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humby F., Bombardieri M., Manzo A., Kelly S., Blades M.C., Kirkham B. Ectopic lymphoid structures support ongoing production of class-switched autoantibodies in rheumatoid synovium. PLoS Med. 2009;6:e1. doi: 10.1371/journal.pmed.0060001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobi C., Lange P., Reiber H. Quantitation of intrathecal antibodies in cerebrospinal fluid of subacute sclerosing panencephalitis, herpes simplex encephalitis and multiple sclerosis: discrimination between microorganism-driven and polyspecific immune response. J. Neuroimmunol. 2007;187:139–146. doi: 10.1016/j.jneuroim.2007.04.002. [DOI] [PubMed] [Google Scholar]
- Jarius S., Eichhorn P., Jacobi C., Wildemann B., Wick M., Voltz R. The intrathecal, polyspecific antiviral immune response: specific for MS or a general marker of CNS autoimmunity? J. Neurol. Sci. 2009;280:98–100. doi: 10.1016/j.jns.2008.08.002. [DOI] [PubMed] [Google Scholar]
- Jessen Krut J., Mellberg T., Price R.W., Hagberg L., Fuchs D., Rosengren L. Biomarker evidence of axonal injury in neuroasymptomatic HIV-1 patients. PLoS One. 2014;9:e88591. doi: 10.1371/journal.pone.0088591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaiser R., Dorries R., Luer W., Poser S., Pohle H.D., Felgenhauer K. Analysis of oligoclonal antibody bands against individual HIV structural proteins in the CSF of patients infected with HIV. J. Neurol. 1989;236:157–160. doi: 10.1007/BF00314332. [DOI] [PubMed] [Google Scholar]
- Kaiser R., Dorries R., ter Meulen J., Kidenya J.J., Pollath M., Fleischer K. Serologic evidence of human immunodeficiency virus infection of the central nervous system in African patients with acquired immunodeficiency syndrome. Eur. Neurol. 1990;30:27–31. doi: 10.1159/000116621. [DOI] [PubMed] [Google Scholar]
- Kallianpur K.J., Kirk G.R., Sailasuta N., Valcour V., Shiramizu B., Nakamoto B.K. Regional cortical thinning associated with detectable levels of HIV DNA. Cereb. Cortex. 2012;22:2065–2075. doi: 10.1093/cercor/bhr285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kallianpur K.J., Shikuma C., Kirk G.R., Shiramizu B., Valcour V., Chow D. Peripheral blood HIV DNA is associated with atrophy of cerebellar and subcortical gray matter. Neurology. 2013;80:1792–1799. doi: 10.1212/WNL.0b013e318291903f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi K., Seilhan D., Uchiara T., Duyckaerts C., Hauw J. High densities of tumor necrosis factor-a in the cerebral cortex and basal ganglia in human immunodeficiency virus-1-associated cognitive/motor complex: a quantitative regional analysis study. Neuropathology. 1997;17:168–173. [Google Scholar]
- Koenig S., Gendelman H.E., Orenstein J.M., Dal Canto M.C., Pezeshkpour G.H., Yungbluth M. Detection of AIDS virus in macrophages in brain tissue from AIDS patients with encephalopathy. Science. 1986;233:1089–1093. doi: 10.1126/science.3016903. [DOI] [PubMed] [Google Scholar]
- Kolb S.A., Sporer B., Lahrtz F., Koedel U., Pfister H.W., Fontana A. Identification of a T cell chemotactic factor in the cerebrospinal fluid of HIV-1-infected individuals as interferon-gamma inducible protein 10. J. Neuroimmunol. 1999;93:172–181. doi: 10.1016/s0165-5728(98)00223-9. [DOI] [PubMed] [Google Scholar]
- Konstantinopoulos P.A., Dezube B.J., Pantanowitz L., Horowitz G.L., Beckwith B.A. Protein electrophoresis and immunoglobulin analysis in HIV-infected patients. Am. J. Clin. Pathol. 2007;128:596–603. doi: 10.1309/QWTQFGA9FXN02YME. [DOI] [PubMed] [Google Scholar]
- Lackner P., Kuenz B., Reindl M., Morandell M., Berger T., Schmutzhard E. Antibodies to myelin oligodendrocyte glycoprotein in HIV-1 associated neurocognitive disorder: a cross-sectional cohort study. J. Neuroinflammation. 2010;7:79. doi: 10.1186/1742-2094-7-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambin P., Gervais A., Levy M., Defendini E., Dubarry M., Lebon P. Intrathecal synthesis of IgG subclasses in multiple sclerosis and in acquired immunodeficiency syndrome (AIDS) J. Neuroimmunol. 1991;35:179–189. doi: 10.1016/0165-5728(91)90172-4. [DOI] [PubMed] [Google Scholar]
- Laurijssens M., Heyman P., Gillis C., De Schrijver L., Roggeman D. Oligoclonal immunoglobulins: a possible index of B cell lymphoma in AIDS. Eur. J. Cancer. 1993;29:S45. [Google Scholar]
- Lifter J., Kincade P.W., Choi Y.S. Subpopulations of chicken B lymphocytes. J. Immunol. 1976;117:2220–2225. [PubMed] [Google Scholar]
- Lin W.L., Fincke J.E., Sharer L.R., Monos D.S., Lu S., Gaughan J. Oligoclonal T cells are infiltrating the brains of children with AIDS: sequence analysis reveals high proportions of identical beta-chain T-cell receptor transcripts. Clin. Exp. Immunol. 2005;141:338–356. doi: 10.1111/j.1365-2249.2005.02845.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lloyd A., Wakefield D., Robertson P., Dwyer J.M. Antibodies to HIV are produced within the central nervous system of all subjects with all categories of HIV infection. Aust. N. Z. J. Med. 1988;18:854–860. doi: 10.1111/j.1445-5994.1988.tb01644.x. [DOI] [PubMed] [Google Scholar]
- Lolli F., Colao M.G., De Maio E., Di Pietro M., Galli E., Grippo A. Intrathecal synthesis of anti-HIV antibodies in AIDS patients. J. Neurol. Sci. 1990;99:281–289. doi: 10.1016/0022-510x(90)90162-g. [DOI] [PubMed] [Google Scholar]
- Lucey D.R., McGuire S.A., Clerici M., Hall K., Benton J., Butzin C.A. Comparison of spinal fluid beta 2-microglobulin levels with CD4 + T cell count, in vitro T helper cell function, and spinal fluid IgG parameters in 163 neurologically normal adults infected with the human immunodeficiency virus type 1. J. Infect. Dis. 1991;163:971–975. doi: 10.1093/infdis/163.5.971. [DOI] [PubMed] [Google Scholar]
- Margolin D.H., Saunders E.H., Bronfin B., de Rosa N., Axthelm M.K., Goloubeva O.G. Germinal center function in the spleen during simian HIV infection in rhesus monkeys. J. Immunol. 2006;177:1108–1119. doi: 10.4049/jimmunol.177.2.1108. [DOI] [PubMed] [Google Scholar]
- Marra C.M., Tantalo L.C., Sahi S.K., Maxwell C.L., Lukehart S.A. CXCL13 as a cerebrospinal fluid marker for neurosyphilis in HIV-infected patients with syphilis. Sex. Transm. Dis. 2010;37:283–287. doi: 10.1097/OLQ.0b013e3181d877a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marshall D.W., Brey R.L., Cahill W.T., Houk R.W., Zajac R.A., Boswell R.N. Spectrum of cerebrospinal fluid findings in various stages of human immunodeficiency virus infection. Arch. Neurol. 1988;45:954–958. doi: 10.1001/archneur.1988.00520330032007. [DOI] [PubMed] [Google Scholar]
- Marshall D.W., Brey R.L., Butzin C.A. Lack of cerebrospinal fluid myelin basic protein in HIV-infected asymptomatic individuals with intrathecal synthesis of IgG. Neurology. 1989;39:1127–1129. doi: 10.1212/wnl.39.8.1127. [DOI] [PubMed] [Google Scholar]
- Martin-Blondel G., Delobel P., Blancher A., Massip P., Marchou B., Liblau R.S. Pathogenesis of the immune reconstitution inflammatory syndrome affecting the central nervous system in patients infected with HIV. Brain. 2011;134:928–946. doi: 10.1093/brain/awq365. [DOI] [PubMed] [Google Scholar]
- Mathiesen T., Sonnerborg A., von Sydow M., Gaines H., Wahren B. IgG subclass reactivity against human immunodeficiency virus (HIV) and cytomegalovirus in cerebrospinal fluid and serum from HIV-infected patients. J. Med. Virol. 1988;25:17–26. doi: 10.1002/jmv.1890250104. [DOI] [PubMed] [Google Scholar]
- Mathiesen T., Sonnerborg A., von Sydow M., Gaines H., Wahren B. IgG subclasses reactivity against HIV and CMV in CSF and serum from HIV patients. J. Med. Virol. 1988;25:17–26. doi: 10.1002/jmv.1890250104. [DOI] [PubMed] [Google Scholar]
- Mathiesen T., Sonnerborg A., Wahren B. Detection of antibodies against myelin basic protein and increased levels of HIV-IgG antibodies and HIV antigen after solubilization of immune complexes in sera and CSF of HIV infected patients. Viral Immunol. 1989;2:1–9. doi: 10.1089/vim.1989.2.1. [DOI] [PubMed] [Google Scholar]
- McArthur J.C., Cohen B.A., Selnes O.A., Kumar A.J., Cooper K., McArthur J.H. Low prevalence of neurological and neuropsychological abnormalities in otherwise healthy HIV-1-infected individuals: results from the multicenter AIDS cohort study. Ann. Neurol. 1989;26:601–611. doi: 10.1002/ana.410260504. [DOI] [PubMed] [Google Scholar]
- McCrossan M., Marsden M., Carnie F.W., Minnis S., Hansoti B., Anthony I.C. An immune control model for viral replication in the CNS during presymptomatic HIV infection. Brain. 2006;129:503–516. doi: 10.1093/brain/awh695. [DOI] [PubMed] [Google Scholar]
- Mecha M., Carrillo-Salinas F.J., Mestre L., Feliu A., Guaza C. Viral models of multiple sclerosis: neurodegeneration and demyelination in mice infected with Theiler's virus. Prog. Neurobiol. 2013;101-102:46–64. doi: 10.1016/j.pneurobio.2012.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meira C.S., Vidal J.E., Costa-Silva T.A., Motoie G., Gava R., Hiramoto R.M. IgG4 specific to Toxoplasma gondii excretory/secretory antigens in serum and/or cerebrospinal fluid support the cerebral toxoplasmosis diagnosis in HIV-infected patients. J. Immunol. Methods. 2013;395:21–28. doi: 10.1016/j.jim.2013.06.005. [DOI] [PubMed] [Google Scholar]
- Mellgren A., Price R.W., Hagberg L., Rosengren L., Brew B.J., Gisslen M. Antiretroviral treatment reduces increased CSF neurofilament protein (NFL) in HIV-1 infection. Neurology. 2007;69:1536–1541. doi: 10.1212/01.wnl.0000277635.05973.55. [DOI] [PubMed] [Google Scholar]
- Milush J.M., Chen H.L., Atteberry G., Sodora D.L. Early detection of simian immunodeficiency virus in the central nervous system following oral administration to rhesus macaques. Front. Immunol. 2013;4:236. doi: 10.3389/fimmu.2013.00236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mokhtarian F., Huan C.M., Roman C., Raine C.S. Semliki Forest virus-induced demyelination and remyelination-involvement of B cells and anti-myelin antibodies. J. Neuroimmunol. 2003;137:19–31. doi: 10.1016/s0165-5728(03)00039-0. [DOI] [PubMed] [Google Scholar]
- Moll N.M., Rietsch A.M., Ransohoff A.J., Cossoy M.B., Huang D., Eichler F.S. Cortical demyelination in PML and MS: similarities and differences. Neurology. 2008;70:336–343. doi: 10.1212/01.WNL.0000284601.54436.e4. [DOI] [PubMed] [Google Scholar]
- Neyt K., Perros F., GeurtsvanKessel C.H., Hammad H., Lambrecht B.N. Tertiary lymphoid organs in infection and autoimmunity. Trends Immunol. 2012;33:297–305. doi: 10.1016/j.it.2012.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otto C., Hofmann J., Finke C., Zimmermann M., Ruprecht K. The fraction of varicella zoster virus-specific antibodies among all intrathecally-produced antibodies discriminates between patients with varicella zoster virus reactivation and multiple sclerosis. Fluids Barriers CNS. 2014;11:3. doi: 10.1186/2045-8118-11-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens G.P., Bennett J.L., Lassmann H., O'Connor K.C., Ritchie A.M., Shearer A. Antibodies produced by clonally expanded plasma cells in multiple sclerosis cerebrospinal fluid. Ann. Neurol. 2009;65:639–649. doi: 10.1002/ana.21641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens G.P., Gilden D., Burgoon M.P., Yu X., Bennett J.L. Viruses and multiple sclerosis. Neuroscientist. 2011;17:659–676. doi: 10.1177/1073858411386615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peluso M.J., Ferretti F., Peterson J., Lee E., Fuchs D., Boschini A. Cerebrospinal fluid HIV escape associated with progressive neurologic dysfunction in patients on antiretroviral therapy with well controlled plasma viral load. AIDS. 2012;26:1765–1774. doi: 10.1097/QAD.0b013e328355e6b2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petito C.K., Adkins B., McCarthy M., Roberts B., Khamis I. CD4 + and CD8 + cells accumulate in the brains of acquired immunodeficiency syndrome patients with human immunodeficiency virus encephalitis. J. Neurovirol. 2003;9:36–44. doi: 10.1080/13550280390173391. [DOI] [PubMed] [Google Scholar]
- Petito C.K., Torres-Munoz J.E., Zielger F., McCarthy M. Brain CD8 + and cytotoxic T lymphocytes are associated with, and may be specific for, human immunodeficiency virus type 1 encephalitis in patients with acquired immunodeficiency syndrome. J. Neurovirol. 2006;12:272–283. doi: 10.1080/13550280600879204. [DOI] [PubMed] [Google Scholar]
- Potasman I., Resnick L., Luft B.J., Remington J.S. Intrathecal production of antibodies against Toxoplasma gondii in patients with toxoplasmic encephalitis and the acquired immunodeficiency syndrome (AIDS) Ann. Intern. Med. 1988;108:49–51. doi: 10.7326/0003-4819-108-1-49. [DOI] [PubMed] [Google Scholar]
- Probasco J.C., Deeks S.G., Lee E., Hoh R., Hunt P.W., Liegler T. Cerebrospinal fluid in HIV-1 systemic viral controllers: absence of HIV-1 RNA and intrathecal inflammation. AIDS. 2010;24:1001–1005. doi: 10.1097/QAD.0b013e328331e15b. [DOI] [PubMed] [Google Scholar]
- Ragin A.B., Du H., Ochs R., Wu Y., Sammet C.L., Shoukry A. Structural brain alterations can be detected early in HIV infection. Neurology. 2012;79:2328–2334. doi: 10.1212/WNL.0b013e318278b5b4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiber H., Lange P. Quantification of virus-specific antibodies in cerebrospinal fluid and serum: sensitive and specific detection of antibody synthesis in brain. Clin. Chem. 1991;37:1153–1160. [PubMed] [Google Scholar]
- Reiber H., Peter J.B. Cerebrospinal fluid analysis: disease-related data patterns and evaluation programs. J. Neurol. Sci. 2001;184:101–122. doi: 10.1016/s0022-510x(00)00501-3. [DOI] [PubMed] [Google Scholar]
- Reiber H., Teut M., Pohl D., Rostasy K.M., Hanefeld F. Paediatric and adult multiple sclerosis: age-related differences and time course of the neuroimmunological response in cerebrospinal fluid. Mult. Scler. 2009;15:1466–1480. doi: 10.1177/1352458509348418. [DOI] [PubMed] [Google Scholar]
- Resnick L., diMarzo-Veronese F., Schupbach J., Tourtellotte W.W., Ho D.D., Muller F. Intra-blood–brain-barrier synthesis of HTLV-III-specific IgG in patients with neurologic symptoms associated with AIDS or AIDS-related complex. N. Engl. J. Med. 1985;313:1498–1504. doi: 10.1056/NEJM198512123132402. [DOI] [PubMed] [Google Scholar]
- Rodriguez S.K., Sarr A.D., Olorunnipa O., Popper S.J., Gueye-Ndiaye A., Traore I. The absence of anti-Tat antibodies is associated with risk of disease progression in HIV-2 infection. J. Infect. Dis. 2006;194:760–763. doi: 10.1086/507042. [DOI] [PubMed] [Google Scholar]
- Saha R.N., Pahan K. Tumor necrosis factor-alpha at the crossroads of neuronal life and death during HIV-associated dementia. J. Neurochem. 2003;86:1057–1071. doi: 10.1046/j.1471-4159.2003.01942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sas A.R., Bimonte-Nelson H.A., Tyor W.R. Cognitive dysfunction in HIV encephalitic SCID mice correlates with levels of interferon-alpha in the brain. AIDS. 2007;21:2151–2159. doi: 10.1097/QAD.0b013e3282f08c2f. [DOI] [PubMed] [Google Scholar]
- Schnell G., Spudich S., Harrington P., Price R.W., Swanstrom R. Compartmentalized human immunodeficiency virus type 1 originates from long-lived cells in some subjects with HIV-1-associated dementia. PLoS Pathog. 2009;5:e1000395. doi: 10.1371/journal.ppat.1000395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuh C., Wimmer I., Hametner S., Haider L., Van Dam A.M., Liblau R.S. Oxidative tissue injury in multiple sclerosis is only partly reflected in experimental disease models. Acta Neuropathol. 2014;128:247–266. doi: 10.1007/s00401-014-1263-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwender S., Imrich H., Dorries R. The pathogenic role of virus-specific antibody-secreting cells in the central nervous system of rats with different susceptibility to coronavirus-induced demyelinating encephalitis. Immunology. 1991;74:533–538. [PMC free article] [PubMed] [Google Scholar]
- Seilhean D., Kobayashi K., He Y., Uchihara T., Rosenblum O., Katlama C. Tumor necrosis factor-alpha, microglia and astrocytes in AIDS dementia complex. Acta Neuropathol. 1997;93:508–517. doi: 10.1007/s004010050646. [DOI] [PubMed] [Google Scholar]
- Simioni S., Cavassini M., Annoni J.M., Rimbault Abraham A., Bourquin I., Schiffer V. Cognitive dysfunction in HIV patients despite long-standing suppression of viremia. AIDS. 2010;24:1243–1250. doi: 10.1097/QAD.0b013e3283354a7b. [DOI] [PubMed] [Google Scholar]
- Simioni S., Cavassini M., Annoni J.M., Hirschel B., Du Pasquier R.A. HIV-associated neurocognitive disorders: a changing pattern. Future Neurol. 2011;6:81–95. [Google Scholar]
- Singer E.J., Syndulko K., Fahy-Chandon B.N., Shapshak P., Resnick L., Schmid P. Cerebrospinal fluid p24 antigen levels and intrathecal immunoglobulin G synthesis are associated with cognitive disease severity in HIV-1. AIDS. 1994;8:197–204. doi: 10.1097/00002030-199402000-00007. [DOI] [PubMed] [Google Scholar]
- Smit T.K., Brew B.J., Tourtellotte W., Morgello S., Gelman B.B., Saksena N.K. Independent evolution of human immunodeficiency virus (HIV) drug resistance mutations in diverse areas of the brain in HIV-infected patients, with and without dementia, on antiretroviral treatment. J. Virol. 2004;78:10133–10148. doi: 10.1128/JVI.78.18.10133-10148.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith M.O., Sutjipto S., Lackner A.A. Intrathecal synthesis of IgG in simian immunodeficiency virus (SIV)-infected rhesus macaques (Macaca mulatta) AIDS Res. Hum. Retrovir. 1994;10:81–89. doi: 10.1089/aid.1994.10.81. [DOI] [PubMed] [Google Scholar]
- Smith M.O., Heyes M.P., Lackner A.A. Early intrathecal events in rhesus macaques (Macaca mulatta) infected with pathogenic or nonpathogenic molecular clones of simian immunodeficiency virus. Lab. Invest. 1995;72:547–558. [PubMed] [Google Scholar]
- Sonnerborg A.B., von Sydow M.A., Forsgren M., Strannegard O.O. Association between intrathecal anti-HIV-1 immunoglobulin G synthesis and occurrence of HIV-1 in cerebrospinal fluid. AIDS. 1989;3:701–705. doi: 10.1097/00002030-198911000-00003. [DOI] [PubMed] [Google Scholar]
- Sopper S., Sauer U., Hemm S., Demuth M., Muller J., Stahl-Hennig C. Protective role of the virus-specific immune response for development of severe neurologic signs in simian immunodeficiency virus-infected macaques. J. Virol. 1998;72:9940–9947. doi: 10.1128/jvi.72.12.9940-9947.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spudich S.S., Ances B.M. Central nervous system complications of HIV infection. Top Antivir. Med. 2011;19:48–57. [PMC free article] [PubMed] [Google Scholar]
- Spudich S.S., Nilsson A.C., Lollo N.D., Liegler T.J., Petropoulos C.J., Deeks S.G. Cerebrospinal fluid HIV infection and pleocytosis: relation to systemic infection and antiretroviral treatment. BMC Infect. Dis. 2005;5:98. doi: 10.1186/1471-2334-5-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stam A.J., Nijhuis M., van den Bergh W.M., Wensing A.M. Differential genotypic evolution of HIV-1 quasispecies in cerebrospinal fluid and plasma: a systematic review. AIDS Rev. 2013;15:152–161. [PubMed] [Google Scholar]
- Subak-Sharpe I., Dyson H., Fazakerley J. In vivo depletion of CD8 + T cells prevents lesions of demyelination in Semliki Forest virus infection. J. Virol. 1993;67:7629–7633. doi: 10.1128/jvi.67.12.7629-7633.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavazzi E., Morrison D., Sullivan P., Morgello S., Fischer T. Brain inflammation is a common feature of HIV-infected patients without HIV encephalitis or productive brain infection. Curr. HIV Res. 2014;12:97–110. doi: 10.2174/1570162x12666140526114956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tourtellotte W.W., Baumhefner R.W., Potvin A.R., Ma B.I., Potvin J.H., Mendez M. Multiple sclerosis de novo CNS IgG synthesis: effect of ACTH and corticosteroids. Neurology. 1980;30:1155–1162. doi: 10.1212/wnl.30.11.1155. [DOI] [PubMed] [Google Scholar]
- Towgood K.J., Pitkanen M., Kulasegaram R., Fradera A., Kumar A., Soni S. Mapping the brain in younger and older asymptomatic HIV-1 men: frontal volume changes in the absence of other cortical or diffusion tensor abnormalities. Cortex. 2012;48:230–241. doi: 10.1016/j.cortex.2011.03.006. [DOI] [PubMed] [Google Scholar]
- Tyor W.R., Glass J.D., Baumrind N., McArthur J.C., Griffin J.W., Becker P.S. Cytokine expression of macrophages in HIV-1-associated vacuolar myelopathy. Neurology. 1993;43:1002–1009. doi: 10.1212/wnl.43.5.1002. [DOI] [PubMed] [Google Scholar]
- Van Wielink G., McArthur J.C., Moench T., Farzadegan H., McArthur J.H., Johnson R.T. Intrathecal synthesis of anti-HIV IgG: correlation with increasing duration of HIV-1 infection. Neurology. 1990;40:816–819. doi: 10.1212/wnl.40.5.816. [DOI] [PubMed] [Google Scholar]
- von Budingen H.C., Kuo T.C., Sirota M., van Belle C.J., Apeltsin L., Glanville J. B cell exchange across the blood–brain barrier in multiple sclerosis. J. Clin. Invest. 2012;122:4533–4543. doi: 10.1172/JCI63842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warnke C., Stettner M., Lehmensiek V., Dehmel T., Mausberg A.K., von Geldern G. Natalizumab Exerts a Suppressive Effect on Surrogates of B Cell Function in Blood and CSF. Mult. Scler. 2014 doi: 10.1177/1352458514556296. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- Weber T., Beck R., Stark E., Gerhards J., Korn K., Haas J. Comparative analysis of intrathecal antibody synthesis and DNA amplification for the diagnosis of cytomegalovirus infection of the central nervous system in AIDS patients. J. Neurol. 1994;241:407–414. doi: 10.1007/BF00900957. [DOI] [PubMed] [Google Scholar]
- Wildemann B., Oschmann P., Reiber H. Thieme; Stuttgart; New York: 2010. Laboratory Diagnosis in Neurology. [Google Scholar]
- Winges K.M., Gilden D.H., Bennett J.L., Yu X., Ritchie A.M., Owens G.P. Analysis of multiple sclerosis cerebrospinal fluid reveals a continuum of clonally related antibody-secreting cells that are predominantly plasma blasts. J. Neuroimmunol. 2007;192:226–234. doi: 10.1016/j.jneuroim.2007.10.009. [DOI] [PubMed] [Google Scholar]
- Yilmaz A., Fuchs D., Hagberg L., Nillroth U., Stahle L., Svensson J.O. Cerebrospinal fluid HIV-1 RNA, intrathecal immunoactivation, and drug concentrations after treatment with a combination of saquinavir, nelfinavir, and two nucleoside analogues: the M61022 study. BMC Infect. Dis. 2006;6:63. doi: 10.1186/1471-2334-6-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yilmaz A., Price R.W., Spudich S., Fuchs D., Hagberg L., Gisslen M. Persistent intrathecal immune activation in HIV-1-infected individuals on antiretroviral therapy. J. Acquir. Immune Defic. Syndr. 2008;47:168–173. doi: 10.1097/QAI.0b013e31815ace97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yilmaz A., Yiannoutsos C.T., Fuchs D., Price R.W., Crozier K., Hagberg L. Cerebrospinal fluid neopterin decay characteristics after initiation of antiretroviral therapy. J. Neuroinflammation. 2013;10:62. doi: 10.1186/1742-2094-10-62. [DOI] [PMC free article] [PubMed] [Google Scholar]