Abstract
Signaling lymphocyte activation molecule (SLAM) is one of the receptors for canine distemper virus (CDV). In this study, canine and feline cells expressing canine SLAM, designated A-72/cSLAM and CRFK/cSLAM, were established for the in vitro study of canine distemper. Recent CDV isolates, KDK-1 and 246, which belong to genotypes Asia/H1 and Asia/H2, respectively, rapidly grew and produced distinct syncytia in both the SLAM-expressing cells. The virus-neutralizing (VN) test was successfully performed using these cells, and the results indicated that sera from dogs experimentally infected with KDK-1 had higher VN titers for homologous strain KDK-1 than for heterologous strain 246 and the vaccine Onderstepoort. These newly established cells expressing canine SLAM would help virological and serological analyses of canine distemper.
Keywords: Canine distemper virus, SLAM
Canine distemper virus (CDV) is a member of the Order Mononegavirale, Family Paramyxoviridae, Genus Morbillivirus. CDV infects dogs, as well as many other Carnivores, and causes lethal disease accompanied by clinical signs such as pyrexia, anorexia, nasal discharge, conjunctivitis, diarrhea, lymphopenia and encephalitis (Appel, 1969). Both attenuated live and recombinant canary poxvirus expressing hemagglutinin (H) and fusion (F) protein vaccines have been used to protect dogs from canine distemper. However, sequence analyses of the H gene of recent field CDV isolates have revealed that they are genetically different from currently available live vaccine strains, such as Onderstepoort. In Japan, it was elucidated that at least two different CDV genotypes, Asia/H1 and Asia/H2, have been circulated among the domestic dog population (Mochizuki et al., 1999).
For in vitro studies of CDV, such as viral isolation and propagation, Madin-Darby canine kidney (MDCK) or African green monkey Vero cells have traditionally been used (Lednicky et al., 2004), but these cells do not express canine signaling lymphocyte activation molecule (SLAM), which is a major receptor in CDV infection (Tatsuo et al., 2001). Therefore, CDV field isolates usually do not replicate with a distinct cytopathic effect (CPE) in these cells. The B cell line of marmoset origin, which expresses marmoset SLAM, transformed with Epstein-Barr virus (B95a cells; Kai et al., 1993), or more recently, canine SLAM-expressing Vero cells (Seki et al., 2003, Lan et al., 2006) have been established as improved alternative cell culturing methods and are used for isolation and propagation of CDV field isolates. By using such cells, in vitro studies of CDV have become easier. However, there is concern that CDV might replicate with mutations when raised in Vero cells (Hamburger et al., 1991, Nielsen et al., 2003, Plattet et al., 2004, Plattet et al., 2005). Therefore, canine SLAM-expressing cell of dog origin was generated. In addition, since CDV can infect to the member of Felis, lions, tigers and so on (Myers et al., 1997), canine SLAM-expressing cell of cat origin was also generated and these established cells were assessed for practical use.
In order to clone the cDNA encoding canine SLAM, total RNA was extracted from the canine B-cell leukaemia cell line GL-1 (Nakaichi et al., 1996) using an RNAeasy Mini Kit (QIAGEN, Germany) and the gene encoding canine SLAM was amplified by PCR using two primers; 5′-ATGTCGACGCCGATGGATTCCAGGGGCT-3′ (canineSLAM-13F; nucleotide position at 13–32 [accession no. AF390108]), and 5′-TCGTCGACTCAGCTCTCTGGGAACGTCA-3′ (canineSLAM-1045R; nucleotide position at 1045-1026). The nucleotide sequence was analyzed, and we found that 3 amino acid sequences were different from those reported by Tatsuo et al. (2001). The amino acids located at positions of 44, 182 and 296 had changed from glutamine, isoleucine and lysine to arginine, valine and arginine, respectively. For the expression of canine SLAM, the gene encoding canine SLAM without the signal peptide was amplified with the primers, 5′-CCAGATCTGAGAGCTTGATGAATTGCCC-3′ (canineSLAM-95F; nucleotide position at 95-114) and canineSLAM-1045R, followed by digestion with restriction endonuleases BglII and SalI, and cloning into the BglII and SalI sites of the expression plasmid pDisplay (Invitrogen, USA). The constructed plasmid, pDisplay/cSLAM, encoded the fusion protein consisting of the signal peptide mouse immunoglobulin kappa-chain V-J2-C, a hemagglutinin A (HA) epitope tag and the mature canine SLAM without the signal peptide. The transient expression of canine SLAM in 293T cells was confirmed by immunoblot analysis using an MAb 12CA5 against the HA tag (Roche, USA), because of which the expressed canine SLAM had molecular masses of 45, 60 and 75 kDa (data not shown).
Stable cell lines expressing canine SLAM were prepared as follows: A-72 (ATCC Number: CRL-1542) and CRFK (ATCC Number: CCL-94) were transfected with pDisplay/cSLAM using Lipofectamine 2000 (Invitrogen, USA), and expression of canine SLAM was confirmed by indirect immunofluorescent assay with the MAb 12CA5 against the HA tag (Roche, USA). Transfected cells were selected by using 400 and 800 μg/ml of G418 (SIGMA, USA) for A-72 and CRFK cells, respectively, and G418-resistant cells were cloned by the limiting dilution method. Several clones were obtained and further examined for their capacity for syncytium formation after infection with CDV strain KDK-1 (genotype Asia/H1). Although there was some difference in syncytium formation among clones, the clones to show the largest syncytium were selected. The cells finally established were designated A-72/cSLAM and CRFK/cSLAM, respectively (Fig. 1A). These established cells, but not A-72 and CRFK cells, transcribed mRNA for canine SLAM after 30 passages (data not shown).
Fig. 1.
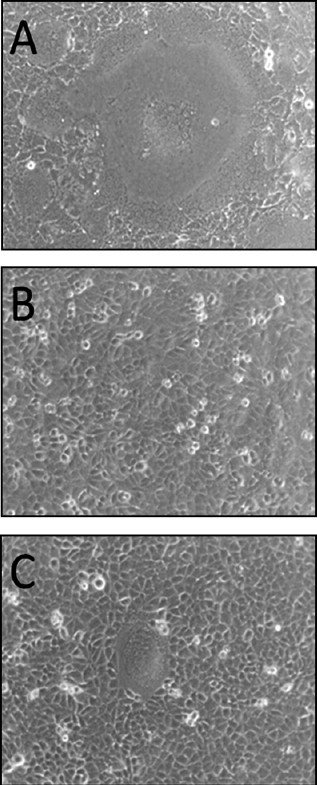
Plaques formed by CDV infection in CRFK/cSLAM cells. CRFK/cSLAM cells were infected with KDK-1 (A) or Onderstepoort (C) at low MOI, incubated for 48 h and then observed by microscopy. (B) Mock-infected CRFK/cSLAM cells.
In order to examine whether recent and vaccine CDV strains induce fusion of these established cell lines, three representative strains were inoculated on A72/cSLAM and CRFK/cSLAM, respectively. Recent isolates KDK-1 and 246 of the genotypes Asia/H1 and Asia/H2, respectively, and the vaccine strain Onderstepoort of the genotype Old America were used. Strains KDK-1 and 246 were both isolated from typical distemper dogs and were cultured in Vero cell culture (JCRB Number: JCRB 9013) six and three passages, respectively. After receiving the viruses in our laboratory, they were maintained for fewer than four passages of subculture in A-72/cSLAM cell culture. The vaccine strain Onderstepoort was also propagated in Vero cell culture; however, previous subculturing records for this strain were uncertain. All three strains produced distinct CPE on both canine SLAM-expressing cell cultures, but not on their parent cells, A-72 and CRFK cell cultures (Fig. 1). On the other hand, Onderstepoort produced distinct CPE in Vero cell culture, but KDK-1 and 246 did not.
Viral growth in the established cells was then compared among CDV strains. Cells were seeded on 35-mm dishes (5.0 × 105 cells per dish) and infected with the virus at a multiplicity of infection (MOI) of 0.01. After 1 h of adsorption, inoculum was removed and the cells were washed twice with DMEM. Then, 2 ml of DMEM containing 10% fetal calf serum (FCS) was added to the dishes, followed by incubation at 37 °C. At various intervals after infection, dishes were frozen and thawed three times and the cultured fluid was centrifuged at 2000 × g for 10 min at 4 °C. The resulting supernatant was stored at −80 °C until use. Virus titer was determined by a plaque assay using CRFK/cSLAM cells, as distinct plaques were formed when compared with assay using A-72/cSLAM cells. In the case of KDK-1, as shown in Fig. 2A, the highest infectivity titer was 6.7 × 104 PFU/ml at 48 h after infection in A-72/cSLAM cells and 1.2 × 105 PFU/ml at 72 h after infection in CRFK/cSLAM cell culture. Similarly, strain 246 grew with a highest infectivity titer 6.4 × 104 PFU/ml at 48 h after infection in A-72/cSLAM cell culture and 1.7 × 105 PFU/ml at 24 h after infection in CRFK/cSLAM cell culture (Fig. 2B). Strains KDK-1 and 246 both propagated very slowly in A-72 cell culture during the observation period, and their titers finally reached about 2.2 × 104 PFU/ml and 2.4 × 104 PFU/ml at 144 h after infection, respectively. On the other hand in CRFK cell culture, strains KDK-1 and 246 both propagated, but their growth ceased at 96 h after infection. Infectivity titers of Onderstepoort in cSLAM-expressing cell culture did not reach as high as those for strains KDK-1 and 246, despite the marked presence of CPE. However, the strain grew in the parental A-72 and CRFK cell cultures without CPE, and the highest infectivity titers were 5.3 × 103 PFU/ml and 9.0 × 103 PFU/ml at 72 h after infection in A-72 and CRFK cells, respectively (Fig. 2C). The viral growth of KDK-1 in A-72/cSLAM was very similar to that in canine SLAM-expressing VERO cells reported by Lan et al. (2006).
Fig. 2.
Comparison of viral growth. A-72/cSLAM (●), CRFK/cSLAM (▴), A-72 (○) and CRFK (▵) were infected with KDK-1, 246 or Onderstepoort at an MOI of 0.01, and mixtures of infected cells and culture supernatant were collected every 24 h. Virus titer was determined by plaque assay using CRFK/cSLAM. The results were shown as means of three independent assays.
For the virus-neutralizing (VN) test using recent CDV isolates, 75% plaque reduction neutralizing test (PRNT75) with CRFK/cSLAM cells was carried out. Four 8-week-old specific-pathogen-free (SPF) beagle puppies with no CDV antibody were divided into two groups, and KDK-1 was oronasally administered to each group once (dogs 1 and 2) or twice four weeks apart (dogs 3 and 4) (Mochizuki et al., 2002). All sera were heat-inactivated at 56 °C for 30 min before examination. Dog sera were serially two-fold diluted in DMEM, 50 μl of the diluted sera were mixed with 50 μl of virus solution containing 50 PFU of CDV, and they were incubated at 37 °C for 1 h. Fifty microliters of each mixture was then inoculated onto CRFK/cSLAM cell culture in 24-well plates, which were then incubated for 1 h at 37 °C. Inoculum was then removed by aspiration, cells were washed twice with DMEM and cells were overlaid with DMEM containing 10% FCS and 0.8% (w/v) agarose (SeaPlaque® Agarose, CAMBREX, USA). Plates were incubated for 3 to 4 days, and cells were fixed and stained with crystal violet formalin solution for plaque counting. VN titer was defined as the reciprocal of the highest serum dilution showing a 75% or more reduction in plaque number in comparison with control wells without test serum (Mochizuki et al., 2002). The results indicated that sera from dogs infected with KDK-1 possessed higher VN titers for KDK-1, ranging from 1:4000 to 1:8000, than those for Onderstepoort (1:160 to 1:640) and 246 (1:320 to 1:640) (Table 1 ). This result suggested that VN antibody induced by genotype Asia/H1 CDV infection may not readily cross-react with heterologous genotype CDV. Similarly, genetic analysis of H protein indicated that recent field isolates are more similar to one another than to the vaccine strains (Mochizuki et al., 1999). However, further studies will be required to clarify the difference of VN titer among strains.
Table 1.
Comparison of virus-neutralizing titers against CDV strains
| Dogsa | VN titer (PRNT75)b |
||
|---|---|---|---|
| KDK-1 | 246 | Onderstepoort | |
| No. 1 | 1:8000 | 1:320 | 1:160 |
| No. 2 | 1:8000 | 1:640 | 1:160 |
| No. 3 | 1:8000 | 1:320 | 1:320 |
| No. 4 | 1:4000 | 1:640 | 1:160 |
Four SPF dogs were experimentally inoculated with KDK-1 via oral route and sera were collected at 4 weeks after final inoculation. Nos. 1 and 2 were inoculated once and Nos. 3 and 4 were twice at an interval of 4 weeks (Mochizuki et al., 2002).
Since VN titer of KDK-1 strain was over 1:1280, sera were serially two-fold diluted from 1:1000 dilution.
The recent field isolates KDK-1 (Asia/H1) and 246 (Asia/H2) produced large syncytia and rapidly grew for 24–72 h after infection in both A-72/cSLAM and CRFK/cSLAM cells, but not in A-72 or CRFK cells, confirming that canine SLAM is one of the most important molecules for syncytium formation and efficient infection in cultured cells. Interestingly, three CDV strains replicated in A-72 cells, which do not express cSLAM, implying that another receptor for CDV exists on A-72 cells. In addition, the field strains also grew in CRFK cells, which suggests that the receptor homologue on CRFK cells might function as a receptor for CDV, but was less effective than that on A-72 cells.
Compared with recent isolates, Onderstepoort grew well in A-72 and CRFK cell cultures, indicating that Onderstepoort may be adapted to these cultured cells. Surprisingly, Onderstepoort did not grow well in the cSLAM-expressing A-72 and CRFK cell cultures, indicating that growth of Onderstepoort is inhibited by the expression of SLAM in the cells. Recent isolates KDK-1 and 246 may possess a mechanism to evade this type of inhibition. Further examination will be required to clarify the reason for the poor growth of Onderstepoort strain in the cSLAM-expressed cell cultures.
In conclusion, the present cSLAM-expressed cell lines of canine and feline origin are useful for virus propagation and sero-diagnosis of CDV. In fact, several CDV were isolated from six raccoon dogs and one Japanese weasel by using A-72/cSLAM cells (manuscript in preparation). In addition, because parental A-72 cells are known to support the growth of other canine virus species, such as canine coronavirus, canine adenovirus types I and II, canine herpesvirus, canine parainfluenza virus and canine parvovirus, A-72/cSLAM cells are potentially advantageous for virological studies in Canids. Furthermore, CDV strains also replicated with syncytium formation in CRFK/cSLAM cells, indicating that the cat cell line may be useful for the study of CDV infection in Felids, such as lions, tigers and so on.
Acknowledgements
We thank Dr. Nakaichi for kindly providing GL-1 cells. This study was supported by a grant-in-aid from the Ministry of Science, Sport and Culture of Japan.
References
- Appel M.J. Pathogenesis of canine distemper. Am. J. Vet. Res. 1969;30:1167–1182. [PubMed] [Google Scholar]
- Hamburger D., Griot C., Zurbriggen A., Orvell C., Vandevelde M. Loss of virulence of canine distemper virus is associated with a structural change recognized by a monoclonal antibody. Experia. 1991;47:842–845. doi: 10.1007/BF01922469. [DOI] [PubMed] [Google Scholar]
- Kai C., Ochikubo F., Okita M., Iinuma T., Mikami T., Kobune F., Yamanouchi K. Use of B95a cells for isolation of canine distemper virus from clinical cases. J. Vet. Med. Sci. 1993;55:1067–1070. doi: 10.1292/jvms.55.1067. [DOI] [PubMed] [Google Scholar]
- Lan N.T., Yamaguchi R., Inomata A., Furuya Y., Uchida K., Sugano S., Tateyama S. Comparative analyses of canine distemper viral isolates from clinical cases of canine distemper in vaccinated dogs. Vet. Microbiol. 2006;115:32–42. doi: 10.1016/j.vetmic.2006.01.010. [DOI] [PubMed] [Google Scholar]
- Lednicky J.A., Meehan T.P., Kinsel M.J., Dubach J., Hungerford L.L., Sarich N.A., Witecki K.E., Braid M.D., Pedrak C., Houde C.M. Effective primary isolation of wild-type canine distemper virus in MDCK, MV1 Lu and Vero cells without nucleotide sequence changes within the entire haemagglutinin protein gene and in subgenomic sections of the fusion and phospho protein genes. J. Virol. Methods. 2004;118:147–157. doi: 10.1016/j.jviromet.2004.02.004. [DOI] [PubMed] [Google Scholar]
- Mochizuki M., Hashimoto M., Hagiwara S., Yoshida Y., Ishiguro S. Genotypes of canine distemper virus determined by analysis of the hemagglutinin genes of recent isolates from dogs in Japan. J. Clin. Microbiol. 1999;37:2936–2942. doi: 10.1128/jcm.37.9.2936-2942.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mochizuki M., Motoyoshi M., Maeda K., Kai K. Complement-mediated neutralization of canine distemper virus in vitro: cross-reaction between vaccine Onderstepoort and field KDK-1 strains with different hemagglutinin gene characteristics. Clin. Diagn. Lab. Immunol. 2002;9:921–924. doi: 10.1128/CDLI.9.4.921-924.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers D.L., Zurbriggen A., Lutz H., Pospischil A. Distemper: not a new disease in lions and tigers. Clin. Diagn. Lab. Immunol. 1997;4:180–184. doi: 10.1128/cdli.4.2.180-184.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakaichi M., Taura Y., Kanki M., Mamba K., Momoi Y., Tsujimoto H., Nakama S. Establishment and characterization of a new canine B-cell leukemia cell line. J. Vet. Med. Sci. 1996;58:469–471. doi: 10.1292/jvms.58.469. [DOI] [PubMed] [Google Scholar]
- Nielsen L., Andersen M.K., Jensen T.D., Blixenkrone-Moller M., Bolt G. Changes in the receptor-binding haemagglutinin protein of wild-type morbilliviruses are not required for adaptation to Vero cells. Virus Genes. 2003;27:157–162. doi: 10.1023/a:1025724526378. [DOI] [PubMed] [Google Scholar]
- Plattet P., Rivals J.P., Zuber B., Brunner J.M., Zurbriggen A., Wittek R. The fusion protein of wild-type canine distemper virus is a major determinant of persistent infection. Virology. 2005;337:312–326. doi: 10.1016/j.virol.2005.04.012. [DOI] [PubMed] [Google Scholar]
- Plattet P., Zweifel C., Wiederkehr C., Belloy L., Cherpillod P., Zuebriggem A., Wittek R. Recovery of a persistent canine distemper virus expressing the enhanced green fluorescent protein from cloned cDNA. Virus Res. 2004;101:147–153. doi: 10.1016/j.virusres.2004.01.002. [DOI] [PubMed] [Google Scholar]
- Seki F., Ono N., Yamaguchi R., Yanagi Y. Efficient isolation of wild strains of canine distemper virus in Vero cells expressing canine SLAM (CD150) and their adaptability to marmoset B95a cells. J. Virol. 2003;77:9943–9950. doi: 10.1128/JVI.77.18.9943-9950.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tatsuo H., Ono N., Yanagi Y. Morbilliviruses use signaling lymphocyte activation molecules (CD150) as cellular receptors. J. Virol. 2001;75:5842–5850. doi: 10.1128/JVI.75.13.5842-5850.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]


