Abstract
The potential effect of the fucose mannose ligand (FML)-vaccine on immunotherapy of canine visceral leishmaniasis was assayed on five mongrel dogs experimentally infected with Leishmania donovani and on 21 Leishmania chagasi naturally infected dogs when seropositive to FML but completely asymptomatic. The clinical signs of the experimentally infected, symptomatic dogs only disappeared after the complete vaccination. Protection was obtained in 3/5 animals that remained asymptomatic, IDR positive and parasite free, 1 year after infection. Furthermore, the asymptomatic, FML-vaccine treated dogs showed stable anti-FML IgG1 levels, increasing IgG2 levels and 79–95% of positive DTH response, during the whole experiment. Twenty-two months after complete vaccination, no obits due to visceral leishmaniasis were recorded and 90% of these dogs were still asymptomatic, healthy and parasite free. On the other hand, 37% (17/46 dogs) kala-azar obits were recorded in a control group that received no treatment during the same period, and that was FML-seropositive and asymtpomatic at the beginning of the assay. Our results indicate that the FML-vaccine was effective in the immunotherapy against visceral leishmaniasis of asymptomatic infected dogs. Normal proportions of CD4 and CD21 lymphocytes were detected in PBMC by FACS analysis, in dogs submitted to immunotherapy, suggesting their non-infectious condition. All animals showed as well significantly increased percents of CD8 lymphocytes as expected for Quillaja saponin (QuilA) vaccine treatments.
Keywords: Canine visceral leishmaniasis, Vaccine, Kala-azar, Immunotherapy, Immunochemotherapy, Saponin, Zoonoses, Fucose mannose ligand
1. Introduction
Human visceral leishmaniasis or kala-azar is a severe and frequently lethal parasitic disease spreading all over the world. A 500,000 new human kala-azar cases are registered annually, most of them (90%) in India, Sudan, Bangladesh and Brazil. The disease is a canid zoonosis [1]. Its etiological agents, Leishmania chagasi and Leishmania infantum, are exposed on the skin of foxes, wild canids and dogs, and transmitted to humans through the sandfly’s byte. Zoonotic visceral leishmaniasis (ZVL) is then a re-emergent canid zoonoses, the epidemiological control of which involves: the elimination of seropositive infected dogs, insecticide treatment within domestic and peridomestic habitations and the systematic treatment of human cases [1]. The killing of seropositive dogs is widely practiced in Brazil and China but unacceptable in Europe. Canine surveillance programs are very laborious and expensive. They require continual vigilance [1] and sensitive serological diagnostic methods [2] to be effective. Furthermore, since many seropositive infected dogs are asymptomatic, owner compliance is complicated [1] even though the infectivity of asymptomatic dogs to sandflies has already been proved [3].
The sacrifice of seropositive animals for epidemiological control is still performed, because chemotherapy of infected dogs using pentavalent antimonial was mostly unsuccessful, even causing exacerbation of disease [3], [4], [5], [6]. Reports about an increase in survival rate [7] and a possible cure potential [8], however, stimulated dog therapy against ZVL. Treatment of infected dogs then became usual in Europe and, lately, in Brazil. However, this is still not recommended by WHO since both the human and canine treatment are performed with the same drugs and this fact can give rise to drug-resistant parasites [9]. Many relapses were described after treatment. Clinically, major signs of disease disappear although this might not indicate the complete absence of parasites. Indeed, dogs were shown to be infective to sandflies several months post-treatment [3], [10]. The presence of latent infections in dogs is then typical and important in maintaining the long-term presence of the parasite in endemic regions [2].
The development of a protective vaccine against canine visceral leishmaniasis has been recommended by WHO as a possible tool for an effective eradication of the disease [11], [12], reducing the offer of parasites to sandfly vectors and consequently the number of human kala-azar cases. However, an effective vaccine against human kala-azar is not yet available and only a few reports in literature deal with a vaccine against canine visceral leishmaniasis. Dunan et al. [13] using a L. infantum semi-purified lyophilized protein preparation (94–67 kD), paradoxically achieved, a significantly higher rate of infection in vaccinated than in control dogs, during the first year of experiment [13]. This vaccine then, while effective in murine models, developed no protection against canine kala-azar in the field [13]. Mayrink et al. [14], using a L. braziliensis lysate and BCG vaccine, in a formulation related to the Leishvacin, previously shown to be about 50% protective against tegumentar leishmaniasis in humans [15], obtained 90% protection against experimental canine kala-azar in the kennel [14] while failed to detect any significant difference between vaccinees and placebos in a well designed field assay [16]. Ramiro et al. [17] obtained 60% protection against experimental infection by L. infantum, in dogs immunized with a plasmid carrying the gene for the LACK antigen of L. infantum followed by a buster with a recombinant vaccinia virus containing the same gene. Protection in vaccinated animals over the untreated controls was disclosed by the increase of IFN-γ, IL-12 expression, lymphocyte proliferative response, IgG2 to IgG1 ratio, and by the decrease of clinical symptoms, number of parasites in target tissues, and IL-4 expression [17].
We recently described the protective effect of the fucose mannose ligand (FML)-vaccine on canine visceral leishmaniasis [18], [19], [20]. The fucose and mannose containing glycoprotein-enriched fraction was isolated from Leishmania donovani promastigotes and named FML ligand, since it strongly inhibits the in vitro infection of murine macrophages by promastigotes and amastigotes of L. donovani [21], [22]. The FML antigen is present on the surface of the parasite throughout the life cycle [22], being a potent immunogen in mice and rabbits [22], [23], [24] and a sensitive, predictive and specific antigen in serodiagnosis of human [25] and canine kala-azar [26].The FML-saponin formulation had already been shown to be safe, immunogenic and protective in Phase I–IIa trials in Balb/c (87.7% parasite load reduction), Swiss albino mice (73–93%) and CB hamsters (84%) [24], [27], [28]. In a Brazilian area endemic for both human and dog visceral leishmaniasis, recent Phase III trials of efficacy using the FML-vaccine in dogs induced 92% [18] and 95% [19] protection in naturally exposed vacinees (76 and 80% of vaccine efficacy, respectively). Protection induced by the FML-Quillaja saponin (QuilA) vaccine lasted up to 3.5 years after vaccination. At this time, vaccinees showed higher seropositivities and intradermal reactions, with no Leishmanial DNA nor parasites in bone marrow punctures. Saline controls, on the other hand showed positive PCR for Leishmania DNA, amastigotes in bone marrow and FML-serology with no intradermal reaction [19]. The FML-QuilA vaccine then, induced a significant, long-lasting and strong protective effect against canine kala-azar in the field [19].
The impressive protection achieved by the FML-vaccines in the field Phase III assays introduced the perspective of their large-scale use in dog vaccination in kala-azar endemic areas. However, while in Phase III assays, all vaccine candidates are healthy and FML-seronegative animals, the larger use of the vaccines in areas where serological control is not obligatory might involve the vaccination of asymtpomatic, seropositive or seronegative dogs that are already infected by Leishmania. It is noteworthy that, the serological window for canine visceral leishmaniasis diagnosis ranges between 90 and 120 days after infection for most well established assays [26], [29]. In all these cases, we would be vaccinating infected dogs and looking for the immunotherapeutic rather than the immunoprophylactic potential of the FML-vaccine. Considering also, the relative failure of chemotherapy against canine visceral leishmaniasis and its negative impact on the epidemiological control of the disease, the possible use of a protective vaccine in the immunotherapy of already infected dogs is highly encouraging and would have broader community acceptance than a control effort based instead on killing infected dogs.
The efficacy of the immunotherapic treatment with FML-saponin vaccine was recently proved in Balb/c mice infected with L. donovani which showed specific increases in IgG1, IgG2a and IgG2b antibodies, a DTH response to promastigote lysate and in vitro ganglion cell proliferative response against FML-antigen, and decreased IL-10 levels in serum [30]. Concomitantly, an impressive and specific decline of liver parasitic burden was detected only in vaccine-treated animals (94.7%) indicating that therapeutic FML-vaccine has a potent effect on modulation of the murine infection [30].
In this investigation, we initiated the study of the potential use of the FML-vaccine in immunotherapy as an aid in treatment of infected, symptomatic and asymptomatic dogs with canine visceral leishmaniasis.
2. Material and methods
2.1. Immunotherapy with the FML-QuilA vaccine
Five mongrel dogs (5-month-old) of the same brood were vaccinated against rabies (Rai-vac I) and canine distemper, Type 2 adenovirus, coronavirus, parainfluenza, parvovirus and leptospira (duramune DA2PP + CvK/Lci, Fort Dodge Animal Health, IW, USA). All animals were experimentally infected, by the i.v. route, with 108 amastigotes obtained from L. donovani (LD-1S/MHOM/SD/00-strain 1S) infected hamster spleens and further vaccinated at days 127, 164 and 187 after infection with 1.5 mg of FML +1 mg of QuilA saponin (Superfos Biosector a/s, QuilA purified saponin, Kvistgaard, Denmark). The FML-vaccine is registered as a Patent: INPI number: PI1100173-9 (March 18, 1997). Federal University of Rio de Janeiro. Brazil. The dogs were monthly analyzed for the following parameters: anti-FML antibody levels (total IgG, IgG1/IgG2), delayed type of hypersensitivity (DTH) against LD1S f/t promastigote lysate antigen, clinical signs lymphocyte phenotyping and parasite evaluation on bone marrow puncture smears. One year after infection, the surviving animals were sacrificed by anesthesia with thionembutal via i.v. (Abbott Laboratories, São Paulo, Brazil) and the presence of Leishmania amastigotes was assessed in Giemsa stained touch biopsies of skin, spleen, liver, lymphnodes and bone marrow smears.
2.2. Immunotherapy with the FML-vaccine in asymptomatic dogs
A group of 21 dogs from the endemic area (Araçatuba, SP, Brazil) received three vaccine doses (1.5 mg FML +1 mg saponin R) when seropositive to FML but completely asymptomatic. They also received their first annual booster. The group was heterogeneous, including individuals of the kala-azar highest susceptible breeds (one Cocker and four German shepherds [31], [32] three Filas, three poodles, two foxes, ine beagle, three dasch hounds, one pinscher [33] and three Rottweilers). During the experiment (22 months), the animals were monitored by the: FML-ELISA assay for total IgG and IgG1 and IgG2 subtypes, the intradermal reaction to L. donovani freeze–thawed promastigote lysate, lymphocyte phenotyping in PBMC, bone marrow puncture and clinical survey. Forty-six asymptomatic FML-seropositive dogs from endemic area were included in this study as the untreated controls.
2.3. FML-ELISA assay
Dog sera were tested for the presence of total anti-L. donovani antibodies by the FML-ELISA assay [26] using the FML-antigen (2 μg/well) solubilized in carbonate buffer (pH 9.6) coated on flat-bottom 96-well plates (Corning 25805-96, cat. number 430480, highly absorbent). Total antibodies were detected by using peroxidase-labeled protein-A (Kirkegaard & Perry Laboratories, Gaithersburg, MD) at a 1:16,000 dilution, in blocking buffer. The cut-off of the FML-ELISA assay, as determined by the Youden test calculation [26] is Abs 492 ηm: 0.450 (mean average of absorbance values of normal healthy serum plus two standard deviations). Furthermore, goat anti-dog IgG1 heavy chain specific (1:2000) or IgG2 (1:32,000) (Bethyl Laboratories, Inc., Montgomery, TX, USA) conjugated with horseradish-peroxidase were used for the IgG subtype determination of each sera. The IgG1 and IgG2-conjugates were titrated, in a two-fold dilution, as previously described [20]. The absorbance values at 492 ηm were compared using a 1:100 dilution of the individual serum samples. Results are expressed as mean values of triplicates.
2.4. Delayed type hypersensitivity (intradermal reaction to promastigote lysate)
This was determined by injecting dogs intradermally, in the inner aspect of the right hind leg, with 0.1 ml of L. donovani freeze–thawed antigen containing 200 μg protein in NaCl 0.9% sterile saline solution (108 stationary phase promastigotes/ml). The left hind leg received only 0.1 ml saline. Measure of the increase of intradermal reaction was performed 24, 48 and 72 h after antigen injection. Indurated areas were marked, and each time the values of the saline control were subtracted from the reaction due to the Leishmania antigen. Reactions showing diameters ≥5 mm were considered positive [18].
2.5. Flow cytometry
PBMC were obtained from 1 ml of the cephalic vein blood collected in EDTA-tubes. After diluting in FACS™ lysing solution (Becton and Dickinson, USA) cells were homogenized, incubated for 10 min at room temperature and centrifuged three-times in 5% FCS added PBS. A 96 well round bottom immunoplates were plated with 30 μl of each one of the following monoclonal antibodies diluted in FACS dil (Serotec, Oxford, UK): anti-Thy-1 (Rat-IgG2b-clone YKIX337.217) (1:800), anti-CD5 (Rat-IgG2a-clone YKIX322.3) (1:800), anti-CD4 (Rat-IgG2a-clone YKIX302.9) (1:12,500), anti-CD8 (Rat-IgG1-clone YCATE55.9) (1:100). 30 μl of anti-rat serum (1:600), 5% FCS supplemented FACS dil, PBS and FACS dil with anti-rat FITC-conjugate (1:200) were used as negative controls. Furthermore, 30 μl of the purified PBMC were added to the wells in triplicates and further incubated for 30 min at room temperature. After incubation, 140 μl of PBS were added to the wells and plates were centrifuged at 800 × g for 10 min at room temperature. Cells were homogenized and labeled with 60 μl of FITC-labeled IgG sheep anti-rat policlonal antibody diluted in FACS dil (1:100) for the anti-CD8 assay and (1:200) for the other antibodies. At this time 2 μl of the FITC-labeled mouse anti-human-CD21 (Mouse-IgG1-clone IOB1a) monoclonal antibody (Immunotech Co. Marseille, France) was used in a direct immunofluorescence procedure. The immunoplate was then incubated at room temperature for 30 min, washed with PBS by centrifugation and cells were finally fixed with the FACS FIX solution (10.0 g/l paraformaldehyde; 10.2 g/l sodium cacodylate and 6.65 g/l sodium chloride, pH 7.2), transferred to microtubes and their relative immunofluorescence was measured on a Becton Dickinson FACS Calibur apparatus (Becton and Dickinson). Each assay included an internal control for non-specific binding to FITC-conjugate sheep anti-rat IgG antibody. We also analyzed the PBMC of 20 healthy and Leishmania-seronegative dogs of a Brazilian kala-azar endemic area (Belo Horizonte) and included them as normal controls.
2.6. Statistical analysis
To test the significance of the differences between groups we used the 95% confidence interval of the averages. Mean average of continuous variables were compared by a standard t-test [34]. Proportions were compared by the χ 2-test.
3. Results
In this work we aimed to investigate the efficacy of the FML-saponin vaccines in immunotherapy of dogs with visceral leishmaniasis. The anti-FML serological response of the experimentally infected dogs submitted to immunotherapy with FML-QuilA vaccine (dogs 1–5) is represented on Fig. 1(A-D) . The IgG antibody started to increase at day 45 after infection (Fig. 1A). All animals showed responses above cut-off at day 60, when mild kala-azar symptoms appeared (Table 1 ). A rapid anti-FML IgG burst was noted after the first vaccine dose (day 127) reaching a plateau after the third dose (day 187) which was maintained during the whole experiment. On the other hand, the anti-FML IgG subtype responses, were only evident after the onset of vaccination schedule, reaching higher levels of IgG2 than of IgG1 (Fig. 1B and C). Dogs 1, 2 and 4, that were olygosymptomatic, became asymptomatic after complete vaccination (Table 1) and maintained a very potent intradermal reaction that disclosed the protective response achieved after FML-QuilA vaccination of these heavily infected animals (Fig. 1D). Despite these results, dog 2 suddenly died, asymptomatic at day 330 after infection. Dog number 5, although polysymptomatic (Table 1), survived resistant until the end of the experiment maintaining a positive, although minor IDR (Fig. 1D).The IDR reaction to the L. donovani promastigote lysate was negative or at the limit of positivity for dog 3 which was polysymptomatic (Table 1), developed the more rapid increase in IgG1 (Fig. 1B) and died of kala-azar on day 240 after infection (Table 1).
Fig. 1.
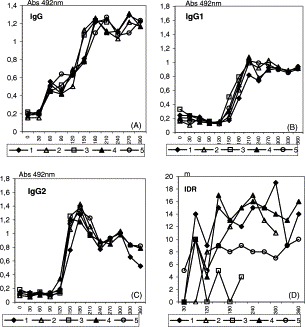
Evolution of anti-FML antibodies and intradermal reaction in infected dogs submitted to immunotherapy (A–D) with the FML-QuilA vaccine. The y-axis represents the FML-ELISA absorbency values of each dog serum (1/100) (A–C) and the thickness of skin test in millimeter (D). Control values of the saline control were subtracted from the DTH reaction due to the Leishmania antigen. Reactions showing diameters ≥5 mm were considered positive. Total antibodies were detected by using peroxidase-labeled protein-A (1:16,000). The cut-off value for IgG is Abs 492 ηm: 0.450. Goat anti-dog IgG1 heavy chain specific (1:2000) or IgG2 (1:32,000) conjugated with horseradish-peroxidase were used for the IgG subtype determination of each serum.
Table 1.
Evolution of the clinical signs of visceral leishmaniasis and parasitic burden in infected dogs submitted to immunotherapy with the FML-QuilA vaccine
| Protocol | Time (days) | Dogs |
||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | ||
| Infection | 0 | – | – | – | – | – |
| 30 | – | – | – | – | – | |
| 60 | Al | Al | Al, ap | Al | Al | |
| 90 | Al | Al | Al, ap | Al | Al | |
| Bone marrow parasites | 120 | – | – | – | – | – |
| 120 | Al | Al | Al, ap | Al | Al | |
| First vaccine dose | 127 | Al | Al | Al, ap | Al | Al |
| 150 | Al | Al | Al, ap, lw | Al | Al, ly | |
| Second vaccine dose | 164 | Al | Al | Al, ap, lw | Al | Al, ly |
| 180 | Al | Al | Al, ap, lw, sl | Al | Al, ly, sl | |
| Third vaccine dose | 187 | Al | Al | Al, ap, lw, sl | Al | Al, ly, sl |
| 210 | Al, sl | Al, sl | Al, ca, lw, gin, sl | Al | Al, sl | |
| 240 | sl | sl | Obit | – | Al, sl, Msl | |
| Bone marrow parasites | 240 | – | – | – | – | |
| 270 | – | – | – | Al, sl, Msl | ||
| 300 | – | – | – | Al, sl, Msl | ||
| Bone marrow parasites | 322 | – | – | – | – | |
| 330 | – | Obit | sl | Al, sl, Msl | ||
| 360 | – | – | Msl | |||
| Parasites in spleen, liver, lymphnode, skin, bone marrow | 360 | – | – | – | ||
Note: Five mongrel dogs (dogs 1–5) were experimentally infected with 108 amastigotes of L. donovani and treated with three monthly doses of the FML (1.5 mg) QuilA (1 mg) vaccine. Kala-azar clinical signs: Al (allopecia), ap (apathy), ca (cachexia), ex (purulent exudate), gin (gingivites), ly (lymphnode swelling), lw (loss of weight), Msl (major skin lesions), sl (small skin lesions).
The analysis of the evolution of clinical symptoms shows that the protected dogs (1, 2 and 4) became asymptomatic 2 months after the complete vaccination (Table 1). Leishmania amastigotes were absent from bone marrow smears of these animals at days 120 (before immunotherapy), 240 and 322 after infection, and from skin, spleen, liver, lymphnodes and bone marrow touch biopsies after sacrifice. Our results indicate that protection against visceral leishmaniasis was achieved in 3/5 infected dogs.
The PBMC flow cytometry analysis of the FML-QuilA-treated dogs was performed on day 240 after infection. By this time, dog 3 was dead, dog 5 was polsymptomatic, dog 4 was asymtpomatic and dogs 1 and 2 were curing from skin lesions. The animals showed close related values for CD5 and Thy-1 lymphocyte markers (Table 2 ). The low value for Thy-1 detected for dog 1 is probably an artifact. Although both average values lie outside of the respective healthy dog’s confidence interval, they are still close to normal values. CD4 percent values remained within the range of normality, as compared with normal uninfected dogs from the endemic area. It is noteworthy that the CD8 percents were significantly increased in all vaccinated dogs while CD21 percentages remained slight below the confidence interval of normal dogs (Table 2).
Table 2.
T cell phenotypes in peripheral blood mononuclear cells of dogs with visceral leishmaniasis submitted to immunotherapy with the FML-vaccine
| Treatment | Dog number | Thy (%) | CD5 (%) | CD4 (%) | CD8 (%) | CD21 (%) |
|---|---|---|---|---|---|---|
| Normal dogs of endemic area | 1 | 81.38 | 71.17 | 41.70 | 28.37 | 12.27 |
| 2 | 82.53 | 72.55 | 40.91 | 22.18 | 8.59 | |
| 3 | 76.19 | 74.51 | 48.92 | 19.67 | 11.37 | |
| 4 | 76.15 | 72.61 | 55.03 | 17.95 | 5.60 | |
| 5 | 80.73 | 78.05 | 42.59 | 26.24 | 16.02 | |
| 6 | 69.48 | 68.67 | 41.67 | 25.26 | 25.62 | |
| 7 | 82.32 | 80.01 | 49.23 | 34.99 | 8.84 | |
| 8 | 66.87 | 75.38 | 42.83 | 19.10 | 23.89 | |
| 9 | 67.89 | 64.45 | 36.79 | 27.45 | 15.84 | |
| 10 | 79.03 | 71.13 | 45.02 | 31.74 | 13.12 | |
| 11 | 75.83 | 66.07 | 45.76 | 26.12 | 19.47 | |
| 12 | 88.78 | 84.96 | 63.54 | 20.69 | 9.85 | |
| 13 | 73.40 | 66.93 | 49.29 | 22.49 | 15.12 | |
| 14 | 90.95 | 79.57 | 54.03 | 42.77 | 3.00 | |
| 15 | 80.36 | 78.72 | 39.53 | 34.07 | 6.54 | |
| 16 | 81.29 | 80.86 | 51.94 | 33.51 | 8.00 | |
| 17 | 76.48 | 66.41 | 45.10 | 23.16 | 13.78 | |
| 18 | 85.82 | 73.43 | 37.49 | 43.85 | 9.46 | |
| 19 | 82.11 | 86.75 | 36.18 | 41.53 | 6.82 | |
| 20 | 70.76 | 72.59 | 40.76 | 17.26 | 11.55 | |
| CI (95%) | 75.50–81.30 | 71.50–77.00 | 42.40–48.50 | 24.30–31.50 | 9.60–14.80 | |
| FML-QuilA immunotherapy of symptomatics (n=4) day 240 | 1 | 25.25 | 79.61 | 77.09 | 45.83 | 11.52 |
| 2 | 87.19 | 80.86 | 33.85 | 55.81 | 8.05 | |
| 4 | 94.33 | 91.13 | 35.83 | 65.17 | 4.97 | |
| 5 | 79.96 | 72.10 | 26.45 | 57.30 | 10.89 | |
| Mean average | 71.68 | 80.92 | 43.30 | 56.02 | 8.86 | |
| FML-R immunotherapy in asymptomatics seropositive (n=10) 22 months after vaccination | 1 | 75.84 | 78.63 | 46.44 | 20.55 | 13.0 |
| 2 | 91.63 | 86.55 | 66.15 | 20.44 | 9.53 | |
| 3 | 76.19 | 85.84 | 42.72 | 56.70 | 11.75 | |
| 4 | 90.27 | 83.95 | 36.71 | 46.81 | 17.59 | |
| 5 | 86.00 | 86.64 | 56.72 | 32.10 | 9.63 | |
| 6 | 90.27 | 80.00 | 35.43 | 29.28 | 11.06 | |
| 7 | 82.57 | 85.46 | 40.56 | 37.50 | 19.23 | |
| 8 | 80.02 | 81.03 | 48.83 | 30.22 | 7.33 | |
| 9 | 82.92 | 81.85 | 54.02 | 32.71 | 30.94 | |
| 10 | 77.98 | 40.82 | 21.82 | 71.76 | 10.33 | |
| Mean average | 83.36 | 79.07 | 44.94 | 38.18 | 14.03 |
Data of normal dogs correspond to the results of 20 healthy and Leishmania-seronegative dogs of a Brazilian kala-azar endemic area (Belo Horizonte). Data of immunotherapy correspond to the results of dogs 1, 2, 4 and 5 infected with L. donovani amastigotes (240 days of infection) and further treated with FML-QuilA vaccine and of 10 L. chagasi naturally infected dogs treated with FML-R vaccine when FML-seropositive and asymptomatic (22 months after vaccination).
The potential immunotherapic effect of the FML-saponin R vaccine was investigated in a group of 21 L. chagasi naturally infected dogs from endemic area that received three vaccine doses when seropositive to FML but completely asymptomatic. The IgG anti-FML absorbencies oscillated from 0.434 to 0.784 at the beginning of the experiment (cut-off value=0.433) and increased during the vaccination schedule, reaching their maximum at day 300 and maintaining the plateau until month 22 (Fig. 2A ). The IgG1 levels were high and relatively stable during the whole period (Fig. 2B) while the IgG2 antibodies, on the other hand, showed lower levels at the beginning, significantly increasing towards the end of the first year of experiment and after the first annual vaccine burst, reaching maximal values at month 22 (Fig. 2B). The DTH response was positive in 79–95% of the animals during the whole experiment (Fig. 2C). No significant variations with time were detected, either in the proportion of DTH positive reactions (P>0.05) or in the size of the skin tests (P>0.05). At month 22, no parasites were present in the dog’s bone marrow smears and the PBMC flow cytometry analysis was performed (Table 2). The mean averages of Thy-1 and CD5 population are close to the levels of normal controls and the CD4 and CD21 averages fall within the confidence interval of normal dogs. Only dog 10 showed a decline in CD4 levels being, however, asymptomatic by the time of evaluation. As detected for the FML-QuilA-treated dogs, the FML-saponin R vaccinees showed as well increased levels of CD8 lymphocytes (Table 2). During the whole period, 19 dogs remained asymptomatic while mild kala-azar symptoms were detected only in two dogs (10%) (Fig. 2F). The first one (cocker) showed lymphnode swelling, stomatites and fever at month 14. It was treated and cured with allopurinol. The second one (German shepherd) showed popliteal lymphnode enlargement with spontaneous regression at month 22. No parasites were found in smears obtained from these lymphnodes. Twenty-two months after complete vaccination, therefore, 90% of these dogs remain asymptomatic, healthy and parasite free, indicating that the FML-saponin R formulation was effective in inducing protection against visceral leishmaniasis in asymptomatic infected dogs.
Fig. 2.
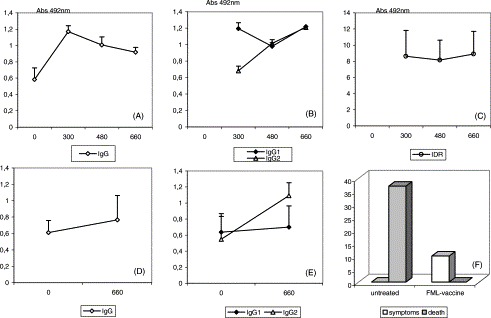
Evolution of anti-FML antibodies and intradermal reaction in infected asymptomatic dogs submitted to immunotherapy and the FML-vaccine. Total IgG (A), IgG1 and IgG2 anti-FML antibodies (B) and the intradermal reaction to L. donovani freeze–thawed promastigote lysate (C) of 21 dogs with canine visceral leishmaniasis from an endemic area, treated with three vaccine doses and the first annual booster of the FML-vaccine when FML-seropositive but completely asymptomatic. The results represent the mean and standard deviation. Total IgG (D) and IgG1 and IgG2 anti-FML-antibodies (E) of 45 control dogs, seropositive to FML and asymptomatic, from an endemic area, that did not receive any treatment. All animals were monitored for a 22 months period. The percent of obits due to canine visceral leishmaniasis and of symptomatic cases were recorded until the end of 22 months, both in the vaccinated and in the control group (F).
As a control group, we monitored 46 dogs seropositive to FML and asymptomatic that remained in the field and did not receive any treatment. The anti-FML IgG antibody level of the control group was not different from the vaccine-treated group, both at days 0 or 660 (P>0.05) (Fig. 2D). Also, while no differences were detected on the initial values for IgG1 and IgG2, between vaccinated and untreated controls (P>0.05), the absorbency values at day 660 were lower in the untreated controls (IgG1 and IgG2; P<0.05). A striking difference in survival rate of the vaccinated or untreated animals was detected confirming the protective potential of the FML-vaccine (Fig. 2F). On the month 12, 15% of the animals were symptomatic while until the end of month 22, 37% (17/46) dead of kala-azar was recorded among the untreated control dogs. This value accounts for dogs that positivated in immunofluorescent assay, showing characteristic symptomatology and either spontaneously died or were sacrificed by the control program. The number of kala-azar cases could be underestimated since 10% of the untreated controls were lost due to other pathologies or to proprietary migration, and 44% were lost or died under undetermined reasons. Only 9% (n=5) of the dogs remained alive and asymptomatics, on month 22. The day 660 absorbency values, on Fig. 2D and E, correspond to these survivors. On the other hand, no kala-azar obits were detected and 10% (2/21) of the dogs became symptomatic among the FML-vaccine-treated dogs, until month 22 (Fig. 2F) disclosing the protective effect of the immunotherapy treatment.
4. Discussion
Since no effective chemotherapy treatment for canine visceral leishmaniasis is available [3], [4], [5], [6], infected dogs remaining in the field constitute a parasite reservoir for the human infection. An effective immunotherapy treatment of these animals would represent a possible welcome alternative for the massive sacrifice of seropositive animals. The major result obtained in this study was the delay on the clinical manifestations of the disease and the absolute reduction of deaths of infected dogs with visceral leishmaniasis submitted to immunotherapy with the FML-vaccine. A second important finding of epidemiological impact was the indication that the immunotherapy-treated dogs remained healthy and probably non-infectious to sandflies, as disclosed by their normal lymphocyte subtype composition. Indeed, the animals maintained normal levels of CD4 and CD21 lymphocytes, while in canine visceral leishmaniasis both types are decreased [35], [36], [37] with the specific CD4 decline being associated to an increased infectivity to sandflies [35].
The average percent value of CD4 lymphocytes in naturally infected dogs with visceral leishmaniasis was 20.47% [35]. The CD4 levels achieved by L. infantum vaccine immunochemotherapy-treated dogs was 35% [38] The FML-QuilA vaccine-treated animals showed an average of 43.30% and the FML-saponin R of 44.94%, strictly included in the confidence interval of the normal healthy dogs. Also, while the L. infantum vaccine-treated animals showed decreased CD8 lymphocyte counts (16.4%) [38] the FML-QuilA and the FML-saponin R vaccines induced significant increases (56.02 and 38.18%) of this lymphocyte subset over the normal range. The CD8 percent increase was higher for FML-QuilA vaccine than for FML-saponin R vaccine. This difference might be due to different composition of the two mixtures of saponins or to different time of evaluation. Indeed, while FACS analysis of FML-QuilA group was performed 2 months after complete vaccination, data of FML-saponin R animals correspond to 10 months after the annual vaccine burst. Cytotoxic T cell (CD8) increase is a clear effect subsequent to saponin treatment [39]. The QS21 saponins has its glycidic moieties linked to the C-28 or C-3 carbon of the triterpen nucleus. The C-28 carbohydrate moiety and its associated normoterpen are responsible for the CTL response [39].
The immunotherapy experiment with FML-QuilA vaccine lacked the adequate control of infected and not vaccine-treated animals. The IgG antibody, clinical signs and IDR results, however, indicate that infection was successfully achieved in all five dogs experimentally infected with L. donovani. The IgG antibody levels were above cut-off at day 60 after infection. Seroconversion in canine visceral leishmaniasis occurs usually between 90 and 120 days after experimental infection [26], [40], [41], [42], [43]. Dogs 1, 2 and 4 that were olygosymptomatic, showed high and sustained IDR since day 30 after infection. This is expected in the early infection with visceral leishmaniasis [32], [44], [45]. Infected dogs develop a strong and transitory IDR response against leishmanial antigen between 120 and 210 days of infection followed by suppression of IDR with the advance of disease [44], [45], [46]. The parasite load used in this experiment (108 amastigotes i.v.) was very high indeed, in order to develop a quick and strong infection in all animals. Amastigotes, however, were absent in bone marrow puncture at 120 days of infection with this heavy inoculum, confirming the low sensitivity of direct parasitologycal methods in the early infection [32], [42]. In a previous study, amastigotes were evident in bone marrow smears, only near their dead of kala-azar (270 days after infection) [45].
Different susceptibility to visceral leishmaniasis was already described in dogs [47], [48]. The macrophage surface Nramp protein that is responsible for murine resistance to visceral leishmaniasis, tuberculosis and salmonellosis shows mutations in visceral leishmaniasis susceptible dogs [49].The mechanisms of inheritance of this genetic marker are not yet defined. In previous work, we disclosed evidence of a familiar component of susceptibility to canine visceral leishmaniasis [45]. Mongrel dogs belonging to the same brood were highly susceptible to L. donovani infection, developing fatal disease, while dogs belonging to other families were more resistant disclosing asymptomatic or oligosymptomatic infections with no obits [45]. For all these reasons, in this investigation we used animals from the same brood, aiming to reduce the genetic background variability. Therefore, the absence of disease in experimentally infected dogs 1, 2 or 4 would be related to the protective effect of the FML-QuilA vaccine rather than to an individual genetically determined resistance to visceral leishmaniasis. The FML-QuilA vaccine showed efficacy then, in the immunotherapy of 3/5 symptomatic experimentally-infected dogs that maintained a strong and positive IDR to leishmanial antigen and showed normal lymphocyte phenotype proportions with no parasites in bone marrow or in target organs. Our results indicate then, that the FML-QuilA vaccination might be capable of inducing a protective status, not only in healthy prophylactically vaccinated dog’s [18], [19] but also, in infected symptomatic dogs submitted to immunotherapy that would remain non-infectious. In agreement with our results, immunotherapy with a first generation Leishmania amazonensis vaccine (Leishvacin) led to a high cure ratio (98.1%) of human tegumentar leishmaniasis patients (Dr Mayrink’s personal communication).
The protective response against canine visceral leishmaniasis obtained after FML-vaccine treatment was higher than that reported by Guarga et al. [38] using a first generation L. infantum lysate vaccine plus glucantime treatment. Protection achieved after treatment was evident in: general clinical improvement (7/10 animals), significant reduction of the infection rates to the sandfly vector fed on the dogs and in the increase in proportion of T cell lymphocytes, especially of CD4/TcR αβ+ and CD4/CD45RA+ cells. However, lymphnode aspirates remained positive [38]. This study showed that immunochemotherapy induces positive changes in the immune response and reduces the infectiousness of dog reservoirs [38]. Working also with a first generation L. braziliensis vaccine (Leishvacin) plus BCG, combined with three series of glucantime treatment, Mayrink et al. [50] achieved protection against canine visceral leishmaniasis in five dogs that remained asymptomatic, gained weight and showed no parasites in bone marrow nor in skin biopsies during 1 year of observation [50].
Protection achieved by immunotherapy-treated dogs in the endemic area was evident in the number of survivors. Indeed, while 37% dead of kala-azar were recorded among the untreated control dogs, no obits were detected among vaccinees. The predominant subtype of immunoglobulin G in dogs might indicate as well a protective response [20], [47], [48]. We recently described the presence of higher IgG1 absorbencies in L. chagasi infected dogs, while the IgG2 subtype was predominant in pre-immune sera, and in FML-vaccinated animals [20]. In the present investigation, we detected similar levels of IgG1 and IgG2 in the immunotherapy FML-treated dogs. This is probably due to the fact that vaccination began after the onset of disease and after the development of IgG1 antibodies. Indeed, the levels of IgG1 were predominant in dog 3 that was not protected by vaccination, suppressed its IDR, developed severe clinical kala-azar and died. It seems that, while predominance of IgG2 characterizes dogs vaccinated prophylactically, similar levels of IgG1 and IgG2 would correlate with the protective status of infected dogs maintained after immunotherapy with FML-QuilA vaccine. In the group vaccinated with FML-saponin R, the IgG1 levels were high and relatively stable during the whole period, while the IgG2 antibodies significantly increased towards the end of the first year of experiment. In conclusion, the predominance of IgG2 response or equivalence to an IgG1 response indicates that protection against the disease is active. Regarding the untreated controls (n=46), we were able to analyze only five survivor animals by the end of the assay. Different from the 37% of the group that developed severe visceral leishmaniasis and died, this very reduced group of animals were asymptomatic or oligosymptomatic at month 22, showing a higher IgG2 response that disclosed a possible natural protective response against visceral leishmaniasis [47], [48] or recent infection [20].
Considering that at present, the epidemiological control campaign removes infected dogs for sacrifice, and no chemotherapy treatment has shown completely satisfactory results, the FML-saponin vaccines might represent new tools to be used as an aid in control of canine visceral leishmaniasis in the field.
Acknowledgements
This study received financial support from: National Council for Scientific and Technological Development (CNPQ); MCT/PRONEX (Brazilian Ministry of Science and Technology);“José Bonifácio” University Foundation of the Federal University of Rio de Janeiro (FUJB-UFRJ), Rio de Janeiro State Research Foundation (FAPERJ), Council for Graduate Studies-UFRJ (CEPG-UFRJ), Brazil, and Fort Dodge Animal Health, Brazil and USA. English language editing was by Dr Jack Woodall, Universidade Federal do Rio de Janeiro.
References
- 1.Tesh R. Control of zoonotic visceral leishmaniasis. Is it time to change strategies. Am. J. Trop. Med. Hyg. 1995;52:287–292. doi: 10.4269/ajtmh.1995.52.287. [DOI] [PubMed] [Google Scholar]
- 2.Palatnik-de-Sousa C.B, Santos W.R, França-Silva J.C, da Costa R.T, Barbosa Reis A, Palatnik M. Impact of canine control on the epidemiology of canine and human visceral leishmaniasis in Brazil. Am. J. Trop. Med. Hyg. 2001;65:510–517. doi: 10.4269/ajtmh.2001.65.510. [DOI] [PubMed] [Google Scholar]
- 3.Alvar J, Molina R, San Andrés M, Tesouro M, Nieto J, Vitutia M. Canine leishmaniasis: clinical, parasitological and entomological follow-up after chemotherapy. Ann. Trop. Med. Parasitol. 1994;88:371–378. doi: 10.1080/00034983.1994.11812879. [DOI] [PubMed] [Google Scholar]
- 4.Ranque J, Ranque M, Cabassu J, Cabassu H. Le diagnostic précoce de la leishmaniose canine par la ponction ganglionnaire. Réflexions à propos de soixante examens positifs obtenus en dix mois dans la région Marsellaise. Bull. Acad. Nat. Med. 1948;132:339–340. [PubMed] [Google Scholar]
- 5.Marzochi M.C.A, Coutinho S.G, Souza W.J.S, Toledo L.M, Grimaldi G, Jr., Momen H. Canine visceral leishmaniasis in Rio de Janeiro, Brazil. Clinical, parasitological, therapeutical and epidemiological findings (1977–1983) Mem. Inst. Osw. Cruz. 1985;80:349–357. doi: 10.1590/s0074-02761985000300012. [DOI] [PubMed] [Google Scholar]
- 6.Oliva G, Gradoni L, Cortese L, Orsini S, Ciaramella P, Scalone A. Comparative efficacy of meglumine antimoniate and aminosidine sulphate, alone or in combination, in canine leishmaniasis. Ann. Trop. Med. Parasitol. 1998;92:165–171. doi: 10.1080/00034989860003. [DOI] [PubMed] [Google Scholar]
- 7.Lanotte G, Rioux J.Á, Pereires J, Vollhardt Y. Ecologie des leishmanioses dans le sud de la France. 10. Les formes évolutives de la leishmaniose viscérale canine. Elaboration d’une typologie bio-clinique à finalité épidémiologique. Ann. Parasitol. 1979;54:277–295. [PubMed] [Google Scholar]
- 8.Von Reiter I, Kretzschmar A, Boch J, Krampitz H. Zur leishmaniose des hundes. Infektionsverlauf, diagnose un therapieversuche nach exp. Infection vom Beagles mit Leishmania donovani (st. Kalkutta) Berl. Münc. Tierärztl. Wschr. 1985;98:40–44. [PubMed] [Google Scholar]
- 9.Organização Mundial da Saúde. Lucha contra las leishmanioses. Serie de informes técnicos 1990;793:1–177.
- 10.Gradoni L, Maroli M, Gramiccia M, Mancianti F. Leishmania infantum infection rates in Phlebotomus perniciosus fed on naturally infected dogs under antimonial treatment. Med. Vet. Entomol. 1987;1:339–342. doi: 10.1111/j.1365-2915.1987.tb00364.x. [DOI] [PubMed] [Google Scholar]
- 11.World Health Organization-TDR. The TDR 15th programm report. Research Progress 1999–2000. New and Improved Tools; 2003. http://www.who.int/tdr/research/progress9900/tools/vdr.htm.
- 12.Modabber, F. Leishmaniasis. In: Tropical disease research progress, 1991–1992. Geneva: World Health Organization; 1993. p. 77–87.
- 13.Dunan S, Frommel D, Monjour L, Ogunkolade B.W, Cruz A, Quilici M. Vaccination trial against canine visceral leishmaniasis. Parasite Immunol. 1989;11:397–402. doi: 10.1111/j.1365-3024.1989.tb00676.x. [DOI] [PubMed] [Google Scholar]
- 14.Mayrink W, Genaro O, Franca-Silva J.C, Costa R.T, Tafuri L, Toledo V.P.C.P. Phase I and II open clinical trials of a vaccine against Leishmania chagasi infections in dogs. Mem. Inst. Osw. Cruz. MEMIOC. 1996;91:695–697. doi: 10.1590/s0074-02761996000600006. [DOI] [PubMed] [Google Scholar]
- 15.Antunes C.M, Mayrink W, Magalhães P.A, Costa A, Melo M.N, Dias M. Controlled field trials of a vaccine against new world cutaneous leishmaniasis. Int. J. Epidemiol. 1986;15:572–580. doi: 10.1093/ije/15.4.572. [DOI] [PubMed] [Google Scholar]
- 16.Genaro O, Pinto J.A, da Costa C.A, França Silva J.C, Costa R.T, Silva J.C. Phase III randomized double blind clinical trial of the efficacy of a vaccine against canine visceral leishmaniasis in urban area of Montes Claros, MG. Braz. Mem. Inst. Osw. Cruz. MEMIOC. 1996;91:166. [Google Scholar]
- 17.Ramiro M.J, Zárate J.J, Hanke T, Rodriguez D, Rodriguez J.R, Esteban M. Protection in dogs against visceral leishmaniasis caused by Leishmania infantum is achieved by immunization with a heterologous prime-boost regime using DNA vaccine and vaccinia recombinant vectors expresssing LACK. Vaccine. 2003;21:2474–2484. doi: 10.1016/s0264-410x(03)00032-x. [DOI] [PubMed] [Google Scholar]
- 18.da Silva V.O, Borja-Cabrera G.P, Correia Pontes N.N, Paraguai de Souza E, Luz K.G, Palatnik M. A Phase III trial of Efficacy of the FML-vaccine against canine kala-azar in an endemic area of Brazil(São Gonçalo do Amarante, RN) Vaccine. 2001;19:1082–1092. doi: 10.1016/s0264-410x(00)00339-x. [DOI] [PubMed] [Google Scholar]
- 19.Borja-Cabrera G.P, Correia Pontes N.N, da Silva V.O, Paraguai de Souza E, Santos W.R, Gomes E.M. Long lasting protection against canine kala-azar using the FML-QuilA saponin vaccine in an endemic area of Brazil (São Gonçalo do Amarante) Vaccine. 2002;20:3277–3284. doi: 10.1016/s0264-410x(02)00294-3. [DOI] [PubMed] [Google Scholar]
- 20.Mendes C.O, Paraguai de Souza E, Borja-Cabrera G.P, Melo Batista L.M, Santos M.A, Parra L.E. IgG1/IgG2 antibody dichotomy in sera of vaccinated or naturally infected dogs with visceral leishmaniasis. Vaccine. 2003;21(19/20):2589–2597. doi: 10.1016/s0264-410x(03)00046-x. [DOI] [PubMed] [Google Scholar]
- 21.Palatnik C.B, Borojevic R, Previato J.O, Mendonça-Previato L. Inhibition of Leishmania donovani promastigote internalization into murine macrophages by chemically defined parasite glycoconjugate. Infect. Immun. 1989;57:754–763. doi: 10.1128/iai.57.3.754-763.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Palatnik de Sousa C.B, Dutra H.S, Borojevic R. Leishmania donovani surface glycoconjugate GP36 is the major immunogen component of the fucose–mannose ligand (FML) Acta Trop. 1993;53:59–72. doi: 10.1016/0001-706x(93)90006-w. [DOI] [PubMed] [Google Scholar]
- 23.Santos W.R, Paraguai de Souza E, Palatnik M, Palatnik de Sousa C.B. Vaccination with the FML antigen (fucose mannose ligand) of Leishmania donovani in the Swiss Albino model. Vaccine. 1999;17:2554–2561. doi: 10.1016/s0264-410x(99)00058-4. [DOI] [PubMed] [Google Scholar]
- 24.Santos W.R, de Lima V.M.F, Paraguai de Souza E, Bernardo R.R, Palatnik M, Palatnik de Sousa C.B. Saponins, IL12 and BCG adjuvant in the FML-vaccine formulation against murine visceral leishmaniasis. Vaccine. 2002;21:30–43. doi: 10.1016/s0264-410x(02)00444-9. [DOI] [PubMed] [Google Scholar]
- 25.Palatnik de Sousa C.B, Gomes E.M, Paraguai de Souza E, Luz K, Palatnik M, Borojevic R. Leishmania donovani: titration of antibodies to the fucose mannose ligand as an aid in diagnosis and prognosis of visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 1995;89:390–393. doi: 10.1016/0035-9203(95)90022-5. [DOI] [PubMed] [Google Scholar]
- 26.Borja Cabrera G.P, da Silva V.O, da Costa R.T, Barbosa Reis A, Mayrink W, Genaro O. The FML-ELISA assay in diagnosis and prognosis of canine visceral leishmaniasis. Am. J. Trop. Med. Hyg. 1999;61:296–301. doi: 10.4269/ajtmh.1999.61.296. [DOI] [PubMed] [Google Scholar]
- 27.Palatnik de Sousa C.B, Paraguai de Souza E, Gomes E.M, Borojevic R. Experimental murine Leishmania donovani infection: Immunoprotection by the fucose mannose ligand (FML) Braz. J. Med. Biol. Res. 1994;27:547–551. [PubMed] [Google Scholar]
- 28.Palatnik de Sousa C.B, Moreno M.B, Paraguai de Souza E, Borojevic R. The FML vaccine (fucose–mannose ligand) protects hamsters from experimental Kala-Azar. Ciência e Cultura (J. Braz. Assoc. Adv. Sci.) 1994;46:290–296. [Google Scholar]
- 29.Badaró R, Chun P, Nakatani M, Burns J, Skeiky Y, Hougton R. TRALd —A rapid test specific for serodiagnosis of visceral leishmanioses. Acta Parasitol. Tur. 1997;21(Suppl 1):175. [Google Scholar]
- 30.Santos W.R, Aguiar I.A, Paraguai de Souza E, de Lima V.F.M, Palatnik M, Palatnik-de-Sousa C.B. Immunotherapy against murine experimental visceral leishmaniasis with the FML-vaccine. Vaccine. 2003;21(32):4668–4676. doi: 10.1016/s0264-410x(03)00527-9. [DOI] [PubMed] [Google Scholar]
- 31.Ranque JM, Quilici M, Dunan S. Les leishmanioses de la region provençale. Considerations epidemiologiques et ecologiques. Colloques Internationaux du CNRS. Ecologie des leishmanioses. Paris: Centre National de la Recherche Scientifique. 1977;239:285–93.
- 32.Abranches P, Silva-Pereira M.C.D, Conceição-Silva F.M, Santana-Gomes G.M, Janz J.G. Canine leishmaniasis: pathological and ecological factors influencing trasmission of infection. J. Parasitol. 1991;77(4):557–561. [PubMed] [Google Scholar]
- 33.da Costa R.T, Genaro O, França-Silva J.C, da Costa C.A, Mayrink W. Urban visceral leishmaniasis in Minas Gerais: a picture about the infection in dogs attended in veterinarian clinics in the metropolitan area of Belo Horizonte and Montes Claros, MG. Mem. Inst. Osw. Cruz. 1996;91(Suppl):154. [Google Scholar]
- 34.Fisher RA, Yates F. Student’s t-test of significance. In: Statistical tables for agricultural, biological and other research workers. Edinburgh: Oliver and Boyd; 1957. p. 157.
- 35.Guarga J.L, Moreno J, Lucientes J, Gracia M.J, Peribanez M.A, Alvar J. Canine leishmaniasis transmission: higher infectivity among naturally infected dogs to sandflies is associated with lower proportions of T helper cells. Res. Vet. Sci. 2000;69:249–253. doi: 10.1053/rvsc.2000.0419. [DOI] [PubMed] [Google Scholar]
- 36.Bourdoiseau G, Bonnefont C, Hoareau E, Boehringer C, Stolle T, Chabanne L. Specific IgG1 and IgG2 antibody and lymphocyte subset levels in naturally Leishmania infantum-infected treated and untreated dogs. Vet. Immunopathol. 1997;59:21–30. doi: 10.1016/s0165-2427(97)00072-x. [DOI] [PubMed] [Google Scholar]
- 37.Moreno J, Nieto J, Chamizo C, Gonzalez F, Blanco F, Barker F. The immune response and PBMC subsets in canine visceral leishmaniasis before and after chemoterapy. Vet. Immunopathol. 1999;30:181–195. doi: 10.1016/s0165-2427(99)00096-3. [DOI] [PubMed] [Google Scholar]
- 38.Guarga J.L, Moreno J, Lucientes J, Gracia M.J, Peribáñez M.A, Castillo J.Á. Evaluation of a specific immunochemotherapy for the treatment of canine visceral leishmaniasis. Vet. Immunol. Immunopathol. 2002;88:13–20. doi: 10.1016/s0165-2427(02)00128-9. [DOI] [PubMed] [Google Scholar]
- 39.Marciani D.J, Press J.B, Eynolds R.C, Pathak A.K, Pathak V, Gundy L.E. Development of semisynthetic triterpenoid saponin derivatives with immune stimulating activity. Vaccine. 2000;18:3141–3151. doi: 10.1016/s0264-410x(00)00118-3. [DOI] [PubMed] [Google Scholar]
- 40.Courtenay O, Quinnell R.J, Garcez L.M, Shaw J.J, Dye C. Infectiousness in a cohort of Brazilian dogs: why culling fails to control visceral leishmaniasis in areas of high transmission. J. Infect. Dis. 2002;186:1314–1320. doi: 10.1086/344312. [DOI] [PubMed] [Google Scholar]
- 41.Genaro O, da Costa RT, França-Silva JC, Reis AB, Silva JC, Vieira EP, et al. Evaluation of an immunochromatographic assay for the diagnosis of dogs experimentally and naturally infected with Leishmania chagasi in Brazil. Acta Parasitol Tur (Suppl I);1996:93.
- 42.Molano I, García Alonso M, Mirón C, Redondo E, Requena J.M, Soto M. A Leishmania infantum multi-component antigenic protein mixed with live BCG confers protection to dogs experimentally infected with L. infantum. Vet. Immunol. Immunopathol. 2003;92:1–13. doi: 10.1016/s0165-2427(02)00315-x. [DOI] [PubMed] [Google Scholar]
- 43.Carrera L, Fermín M.L, Tesouro M, García P, Rollán E, González J.L. Antibody rersponse in dogs experimentally infected with Leishmania infantum: infection course antigen markers. Exp. Parasitol. 1996;82:139–146. doi: 10.1006/expr.1996.0018. [DOI] [PubMed] [Google Scholar]
- 44.Pinelli E, Killick-Kendrick R, Wagennar E, Bernadina W, del Real G, Ruitenberg J. Cellular and humoral immune responses in dogs experimentally and naturally infectes with Leishmania (L.) infantum. Infect. Immunol. 1994;62:229–235. doi: 10.1128/iai.62.1.229-235.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Borja-Cabrera GP. Análise do potencial diagnóstico, prognóstico e imunoprotetor do antígeno FML (ligante de fucose manose) de Leishmania (L.) donovani, no calazar canino experimental e de área endêmica. Ph.D. thesis. Department of Experimental Pathology, Federal Fluminense University; 2000. p:79.
- 46.Cardoso L, Cabral M. Leishmania and canine leishmanioses. Rev. Port. Cien. Vet. 1998;527:121–141. [Google Scholar]
- 47.Solano-Gallego L, Riera C, Roura X, Inieste L, Gallego M, Valladares J.E. Leishmania infantum-specific IgG, IgG1 and IgG2 antibody responses in healthy and ill dogs from endemic areas. Evolution in the course of infection and after treatment. Vet. Parasitol. 2001;96:265–276. doi: 10.1016/s0304-4017(00)00446-5. [DOI] [PubMed] [Google Scholar]
- 48.Deplazes P, Smith N.C, Arnold P, Lutz H, Eckert J. Specific IgG1 and IgG2 antibody responses of dogs to Leishmania infantum and other parasites. Parasite Immunol. 1995;17:451–458. doi: 10.1111/j.1365-3024.1995.tb00914.x. [DOI] [PubMed] [Google Scholar]
- 49.Altet L, Francino O, Solano-Gallego L, Renier C, Sanchez A. Mapping and sequencing of the canine NRAMP1 gene and identification of mutations in leishmaniasis-susceptible dogs. Infect. Immun. 2002;70:2763–2771. doi: 10.1128/IAI.70.6.2763-2771.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mayrink W, Genaro O, Costa C.A, França-Silva J.C, Hermeto M.V, Oliveira Lima A. Combined immunotherapy and chemotherapy in canine visceral leishmaniasis. Preliminary findings. Mem. Inst. Osw. Cruz. 1992;87(Suppl 2):201. [Google Scholar]


