Abstract
Glycans form a highly variable constituent of our mucosal surfaces and profoundly affect our susceptibility to infection and disease. The diversity and importance of these surface glycans can be seen in individuals who lack a functional copy of the fucosyltransferase gene, FUT2. Representing around one-fifth of the population, these individuals have an altered susceptibility to many bacterial and viral infections and diseases. The mediation of host–pathogen interactions by mucosal glycans, such as those added by FUT2, is poorly understood. We highlight, with specific examples, important mechanisms by which host glycans influence infection dynamics, including by: acting as pathogen receptors (or receptor-decoys), promoting microbial stability, altering the physical characteristics of mucus, and acting as immunological markers. We argue that the effect glycans have on infection dynamics has profound implications for many aspects of healthcare and policy, including clinical management, outbreak control, and vaccination policy.
Keywords: infection susceptibility, glycosyltransferase, FUT2, viral infection, microbiota
Trends
Mucosal surfaces form a major interface between us and the external environment. These surfaces are decorated by complex sugar moieties that profoundly affect its function. Individual variation in these mucosal sugars contributes to overall infection and disease susceptibility within the population.
Recent mechanistic studies involving mouse models have revealed how the FUT2 gene mediates host–bacterial interactions, via its effect on mucosal surfaces. There is now a need to further characterise the role of mucosal sugars in bacterial and viral infection dynamics. We describe several mechanisms that highlight the importance of mucosal sugars in host defence and their contribution to disease susceptibility.
Variation in glycans within the population may play a crucial, yet underappreciated, role in viral-infection susceptibility, with the potential to inform clinical management.
Mucus Glycans: More Than Just Decoration
Mucosal infections account for over one-tenth of deaths globally, and are a major source of morbidity [1]. These are predominately infections of the respiratory tract, gastrointestinal tract, and genitourinary tract. With these sites exposed to external sources, encounters with microbial pathogens are unavoidable; however, the likelihood that an interaction will result in an infection varies substantially between individuals [2]. The ability to predict the outcome of exposure remains a central challenge in modern medicine.i
The extent to which mucosal surfaces influence the risk of infection is complex and remains poorly understood. Often, these mucosae are oversimplified as mere physical barriers; however, their molecular composition, consisting of intricate glycan structures (see Glossary) on secreted proteins and lipids, provides many additional immunological functions 3, 4. The large diversity of glycans that can be displayed by mucosal surfaces are determined both by inherited variation [5] and transcriptional regulation within epithelial cells [6]. Importantly, interindividual variation in the types of glycan present on mucosal surfaces is a major contributor to differences in susceptibility to a variety of infections 3, 5, 7, 8. Indeed, the contribution of mucosal glycans to infection susceptibility has gained much recent interest, owing to studies that have provided mechanistic insight into their function 9, 10, 11.
Mucosal constituents that are decorated with glycans are formed through a process mediated by a diverse family of glycosyltransferase enzymes, mostly acting in a template-independent manner [12]. FUT2 encodes an α(1,2)-fucosyltransferase that is expressed in mucosal tissues by multiple epithelial cell types. FUT2 facilitates the attachment of the L-fucose monosaccharide to specific O-linked glycan chains, producing α(1,2)-fucosylated glycans 5, 8. This resulting α(1,2)-fucosylated glycan is a highly versatile structure and can be further modified to form one of a number of clinically important glycans, including the A and B histo-blood group antigens on mucosal surfaces 5, 8. Mucosal histo-blood groups are analogous to those found on erythrocytes, although only those secreted by mucosal surfaces are dependent on FUT2. For example, an individual who has an A blood type will express A-type glycans on erythrocytes, but will only express A-type glycans on mucosal surfaces if they have a functional FUT2 [5]. After the glycosylation process, α(1,2)-fucosylated proteins and lipids are either secreted from mucosal epithelium into the lumen directly, or are anchored to the apical cell surface membrane. Because FUT2 controls the nature of the various α(1,2)-fucosylated glycans secreted by mucosal surfaces, it was termed the ‘secretor’ gene, although it does not regulate secretion per se [5].
As α(1,2)-fucosylated glycans act as important precursors for a range of mucosal glycans, the high frequency of nonsense SNPs within the FUT2 gene in humans [13] is perhaps surprising. In fact, approximately one-fifth of the global population harbour two nonfunctional alleles and are therefore unable to express α(1,2)-fucosylated glycans on mucosal surfaces 7, 13. While individuals with a functional FUT2 allele are termed ‘secretors’, those with loss-of-function mutations are termed ‘nonsecretors’. Interestingly, the frequency of nonsecretors varies substantially with ethnicity 7, 13, with a range of SNPs found to confer this nonsecretor phenotype. However, the widespread distribution of these traits indicates a conserved selective advantage for nonsecretors under certain circumstances [14] (Box 1 ). In addition to an absence of α(1,2)-fucosylated glycans in mucosal secretions, including the absence of histo-blood group antigens, nonsecretors also display increased levels of sialylated glycans in mucosal secretions [15], presumably as a result of reduced glycosyltransferase competition.
Box 1. FUT2 SNPs and Historical Infections.
Multiple SNPs, found in the FUT2 coding region, confer loss, or hindered function. The most common nonsense SNP in Caucasian, African, and central Asian populations is a G→A substitution at base pair 428 (rs601338); however, the most common in east Asian populations is an A→T substitution at base pair 385 (rs1047781) 7, 13. Both SNPs occur at similar frequencies in their respective populations, with estimates dating the emergence of the 428G→A mutation to at least 1.87 million years ago and the 385A→T mutation to at least 256 000 years ago [72]. The age and frequency of these mutations suggest that they are maintained in the gene pool by balancing selection, where both secretor and nonsecretor variants provide selective advantage. Available evidence suggests that differential resistance to infection is the driver of this balancing selection [14], although identification of the causative infective agent is speculative. However, this phenomenon has been observed outside of the human population, where infection-driven selection of glycosyltransferase variants was reported in a study of rabbit populations, where those with endemic rabbit haemorrhagic disease virus had glycosyltransferase SNPs at higher frequencies compared to populations without endemic virus [73].
Alt-text: Box 1
A clue as to why these loss-of-function mutations are carried at such a high frequency, and an illustration more generally of the importance of surface glycans to infection susceptibility, is the major differences in rates of bacterial- and viral-mediated diseases between secretors and nonsecretors (Table 1 , Key Table). A large number of studies have now reported significantly higher rates of viral infection in secretors, including life-threatening infections caused by HIV, influenza, and norovirus 16, 17, 18, 19, 20, 21. At the same time, secretors appear to be at a reduced risk of infections caused by bacterial pathogens, including Streptococcus pneumoniae, Neisseria meningitidis, Haemophilus influenzae, and Salmonella enterica serovar Typhimurium 9, 11, 22, 23, 24. This dichotomy in susceptibility also extends to chronic multifactorial diseases, such as chronic pancreatitis [25], and diseases of altered immune regulation, such as asthma 26, 27, type 1 diabetes [28], and psoriasis [29]. It is important to note that while all of the studies listed in Table 1 reported significant associations between secretor status and disease, sample sizes and effect sizes vary.
Table 1.
Key Table
Secretor Status Profoundly Influences Infection and Disease Susceptibilitya
| Infection | Secretors more susceptible | Nonsecretors more susceptible | Refs |
|---|---|---|---|
| Norovirus (GII.4) | ✔ | 21, 74 | |
| Rotavirus (VP8) | ✔ | 36, 37, 38 | |
| Influenza A virus | ✔ | [19] | |
| Rhinovirus | ✔ | [19] | |
| Echovirus | ✔ | [19] | |
| RSV | ✔ | [19] | |
| HIV | ✔ | 16, 17, 18 | |
| Helicobacter pylori | ✔ | 24, 75, 76 | |
| Candida albicans | ✔ | 77, 78 | |
| Streptococcus pneumoniae | ✔ | [22] | |
| Neisseria meningitidis | ✔ | [22] | |
| Haemophilus influenzae | ✔ | [23] | |
| Salmonella enterica serovar Typhimuriumb | ✔ | [11] | |
| Citrobacter rodentiumb | ✔ | [9] | |
| Campylobacter jejuni | ✔ | 41, 42 | |
| Urinary tract infection | ✔ | 79, 80 | |
| Bacteraemia (after haematopoietic stem cell transplantation) | ✔ | [81] | |
| Disease | Secretors more susceptible | Nonsecretors more susceptible | Refs |
| Non-CF bronchiectasis severityc | ✔ | [67] | |
| Asthma severity | ✔ | [82] | |
| Graft-versus-host disease | ✔ | [81] | |
| Intestinal-type gastric cancer | ✔ | [83] | |
| Primary sclerosing cholangitis | ✔ | [84] | |
| Crohn’s disease | ✔ | 53, 85 | |
| Celiac disease | ✔ | [86] | |
| Asthma | ✔ | 26, 27 | |
| Type 1 diabetes | ✔ | [28] | |
| High plasma vitamin B12 | ✔ | 87, 88, 89 | |
| Chronic pancreatitis | ✔ | [25] | |
| Psoriasis | ✔ | [29] | |
| Acute uncomplicated pyelonephritis | ✔ | [90] | |
| Behçet’s disease | ✔ | [91] |
Secretor phenotype is associated with increased susceptibility to viral infections and respiratory disease severity, but with decreased susceptibility to bacterial infections and a diverse range of chronic inflammatory diseases.
Demonstrated in Fut2–/– mice with no human epidemiological evidence.
Abbreviation: CF, cystic fibrosis.
Despite the well-described associations between FUT2 and other mucosal glycosyltransferases, and a diversity of infections and diseases, our understanding of the mechanisms behind these relationships remains poor. Infection and pathogenesis are complex processes, with mucosal glycans likely influencing susceptibility through both direct and indirect mechanisms.
Sticking Around: FUT2 and Pathogen Adherence
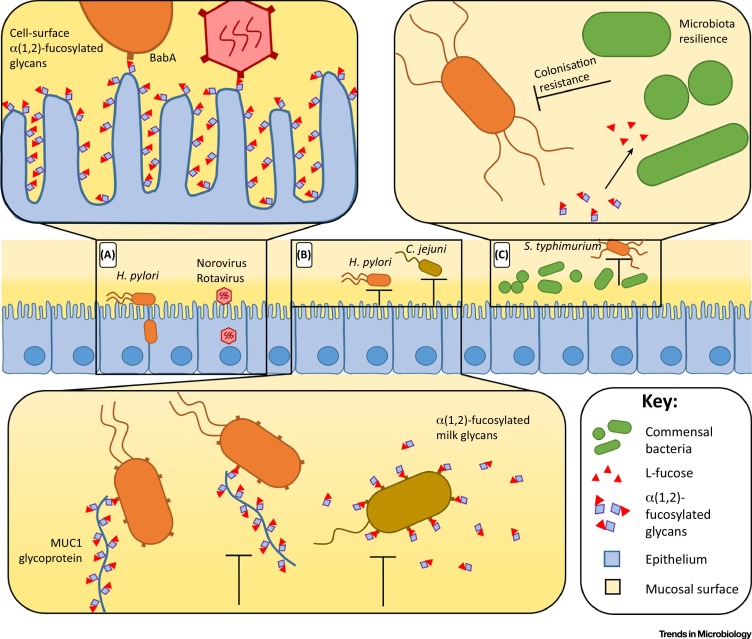
α(1,2)-fucosylated glycans influence infection susceptibility directly, through facilitating pathogen adherence. As has been reviewed in detail 30, 31, 32, multiple bacteria encode specific receptors that bind to host α(1,2)-fucosylated glycans for pathogen adherence. A well characterised example of this is in Helicobacter pylori, facilitated by the BabA adhesin. BabA has a specificity for the ‘Lewis b’ α(1,2)-fucosylated mucosal glycan, therefore BabA-expressing H. pylori is more readily able to adhere to the gastric mucosa and colonize the stomachs of secretor individuals [33] (Figure 1 A). BabA-encoding H. pylori and subsequent infection susceptibility is something of an exception, as this is the only bacterial species listed in Table 1 where susceptibility is increased in secretors (due to the specificity of BabA towards α(1,2)-fucosylated glycans). Two viruses have analogous receptors, norovirus (specifically strain GII.4) 34, 35 and rotavirus (specifically strains containing spike protein VP8) 36, 37. Both of these viruses encode different adhesins, specific to α(1,2)-fucosylated glycans. The absence of these glycans in nonsecretors therefore confers high levels of resistance 20, 21, 38 (Figure 1A).
Figure 1.
α(1,2)-Fucosylated Glycans Affect Infection Susceptibility. (A) Adhering to membrane-bound α(1,2)-fucosylated glycans facilitates pathogen infection. (B) Adhering to luminal α(1,2)-fucosylated glycans can act as a receptor decoy, reducing infection susceptibility. MUC1 glycoprotein is membrane bound, however, detaches from the epithelium following pathogen binding. (C) Commensal microbes can utilise α(1,2)-fucosylated glycans, occupying a niche space and hindering pathogen colonisation. Abbreviations: H. pylori, Helicobacter pylori; C. jejuni, Campylobacter jejuni; S. typhimurium, Salmonella enterica serovar Typhimurium.
More complex FUT2-dependent pathogen adherence pathways have also been characterised, based on glycan location. Glycosylated proteins and lipids are abundant in the gastrointestinal tract, either anchored to the cell surface, secreted into the lumen, or taken in through ingestion. Therefore, infection susceptibility, where pathogens adhere to glycans, depends on the location and anchoring of the glycan. Glycans that are not attached to the epithelium can, in fact, reduce infection susceptibility by acting as receptor decoys. For example, the cell surface mucin, MUC1, carries Lewis-b glycans and is shed from the surface of gastric epithelial cells acting as a releasable decoy to limit adhesion by BabA-expressing H. pylori to other cell surface Lewis-b-expressing molecules [39]. As a separate example, maternal secretor status affects glycosylation of glycoproteins in milk [40], and consequently maternal secretors reduce their infants’ susceptibility to Campylobacter jejuni diarrhoea [41]. This has been attributed to C. jejuni binding to α(1,2)-fucosylated milk glycans [42], which act as a receptor decoy in the infant, sequestering the pathogen away from the epithelium (Figure 1B). As these examples demonstrate, the dynamics of how glycan-mediated adherence (either membrane-bound or luminal) ultimately confers susceptibility or resistance to infection is complex.
Commensal Influence: FUT2 and the Microbiota
In addition to influencing pathogen adherence, FUT2 has been shown to also affect infection susceptibility indirectly. For example, murine studies have shown that the presence of Fut2 reduces susceptibility to S. Typhimurium, Enterococcus faecalis and Citrobacter rodentium infection through the effect of α(1,2)-fucosylated glycans on the commensal gut microbiota 9, 10, 11 (Figure 1C). Even small changes to microbiota composition can alter nutrient availability, profoundly affecting the ability of bacterial pathogens to colonise the gut [43]. Beyond such ‘colonisation resistance’, murine studies have also shown that Fut2-dependent fucosylated glycans are an important endogenous nutrient for commensal microbes, facilitating rapid host recovery following periods of stress caused by intestinal infection or inflammation [9]. These findings are supported by in silico analyses of microbial structure stability using microbiota data from Fut2 knock-out mice [44].
By contrast, a large study in healthy adult humans reported no difference in faecal microbiota composition between secretors and nonsecretors [45], contradicting previous, positive associations from a smaller cohort analysing mucosal microbiota from biopsies [46]. The use of intestinal mucosal biopsies in the smaller cohort study, where a greater host genotype effect may be expected [47], may explain this discrepancy [44].
Differences in microbiota composition and resilience may also explain the numerous diseases associated with secretor status (as detailed in Table 1). Many of these conditions (including asthma, Crohn’s disease, celiac disease, and psoriasis) are associated with intestinal microbiota composition 48, 49, 50. If secretor status can influence gut microbiology, it is reasonable to suggest that secretor status may contribute to microbiota-related disease susceptibility among predisposed individuals, as discussed elsewhere 8, 30, 31, 46, 51, 52, 53. However, given the numerous confounding environmental exposures in human populations, large cohort studies with detailed metadata are required to determine the contribution of secretor status to these complex, multifactorial diseases.
Sugar Structures: Glycans Influencing Mucus Barrier Function
The physical barrier properties of mucus define its primary function, as governed by its viscosity, permeability, and rheology 3, 4. Mucins, the major proteins that make up the mucus layer, are comprised of 70% glycans by mass [4]. Small variations in the molecular composition of mucin glycans can therefore impact the overall physical properties of mucus. While α(1,2)-fucosylated mucins constitute a large proportion of secretor mucus, interestingly, nonsecretors display higher levels of sialylated mucins [15]. Sialic acid, like fucose is often added as a terminal saccharide and has different properties to fucose.
At a molecular level, sialylated terminal glycans have a larger polar surface compared to fucose 54, 55. The higher electronegative change in sialylated mucins has therefore been proposed to increase both mucus hydration and electronegative repulsion, compared to fucosylated mucins 54, 55. Particularly in the airways, mucus hydration is an important characteristic for mucociliary clearance 4, 54. Well-described examples of impaired clearance are evident in diseases such as chronic obstructive pulmonary disease and cystic fibrosis [4]. While this requires experimental validation, higher sialic acid levels in nonsecretor airways may affect mucus clearance mechanisms. Such a difference might explain the observed findings of a study in which infection by influenza virus, respiratory syncytial virus (RSV), rhinovirus, and echovirus were all lower in nonsecretors [19]. It is interesting to speculate that altered mucus characteristics in nonsecretors may contribute to this reduced susceptibility to viral infection.
Anti-sugar Antibodies: Host Antibodies Recognise Nonself Glycans
A major contributor to mucosal immunity is the detection and neutralisation of pathogens by secreted antibodies 56, 57 – particularly natural antibodies (NAbs), which are generated in a T cell-independent way and have broad-spectrum activity against a diversity of antigens [57]. A subset of NAbs have an affinity towards glycans, recognising those specific to bacteria, but also to glycans found naturally within the human population. For example, the A and B histo-blood group antigens elicit strong NAb-specific immune responses in noncompatible individuals, best known as the primary cause of blood transfusion reactions [58]. An interesting research question is whether these same A and B antigens decorate viral particles and affect viral transmission between histo-incompatible hosts.
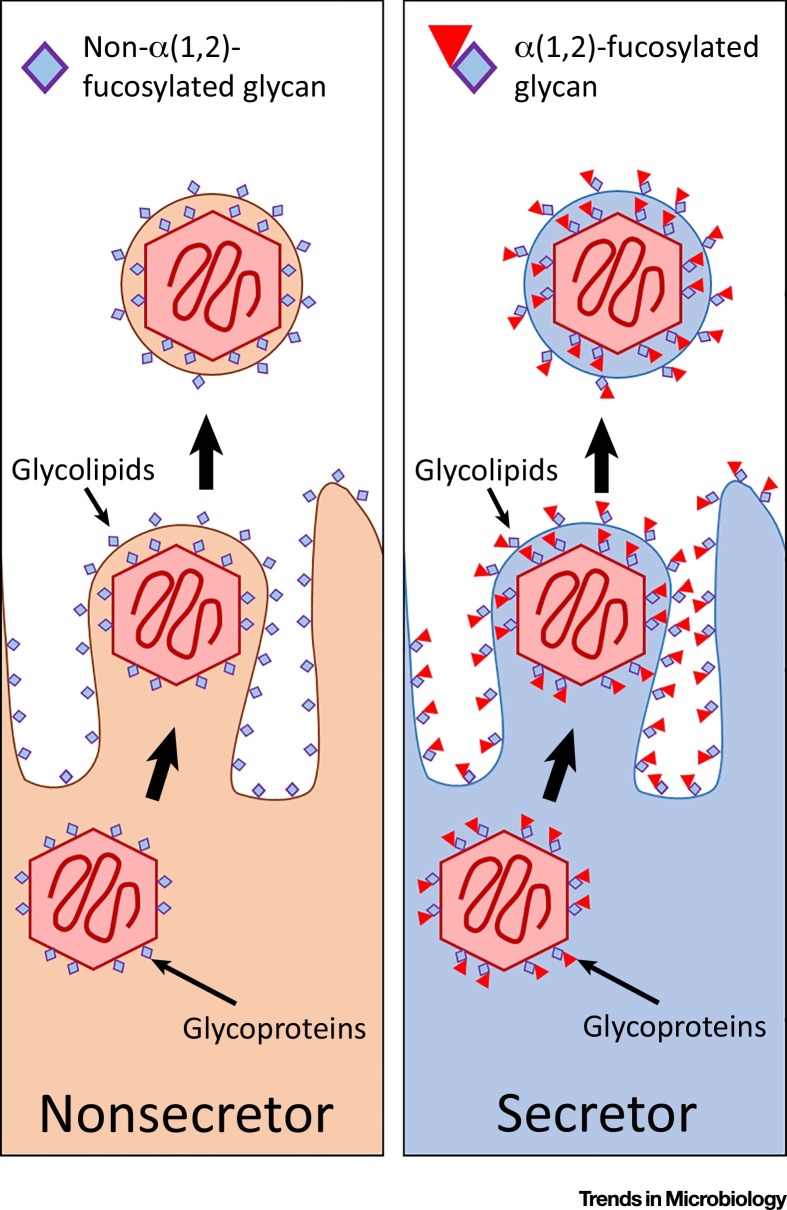
By definition, viral replication is reliant on host-cell machinery, which includes glycosylation by host glycosyltransferases. Many viruses also utilize host membranes for encapsulation. Several studies have demonstrated that viruses shed from epithelial cells displaying A or B glycans also display these glycans. Specifically, cell lines infected with HIV-1 [59] or measles virus [60] were found to produce viral particles coated in their specific blood group glycan (as depicted in Figure 2 ). While these studies were performed in vitro using transformed cell lines, they support a logical conclusion; viruses from a cell expressing particular glycosyltransferases carry a host-glycan fingerprint of this parent cell. Addition of anti-histo blood group NAbs was found to bind to these viruses and elicit antibody-mediated inactivation 59, 60. Further studies have investigated NAb-mediated viral neutralisation using other, analogous glycan structures, showing supporting results 61, 62. However, it is unlikely that nonsecretors develop NAbs with specificity to secretor glycans, as α(1,2)-fucosylated glycans exist in nonmucosal sites, which are not reliant on FUT2. Secretor status may however affect a virus’s ability to evade host antibody defences. As FUT2 mediates the display of ABO histo-blood group antigens in mucosal secretions, viruses that come from a nonsecretor would not display blood group antigens and hence would not be affected by anti-histo blood group NAbs. Further infection models are required to elucidate the contribution of viral glycosylation to infection dynamics.
Figure 2.
Viruses Are Decorated in Host Glycans. Viral replication requires the use of host cell machinery, including host glycosyltransferases. Underlying genetic factors (such as FUT2 SNPs) affect glycosylation of shed viruses. Viral glycoproteins and glycolipids (for enveloped viruses) differ between viruses shed from secretor and nonsecretor cells.
Using Glycosyltransferase Characteristics to Inform Policy and Practice
The majority of studies discussed have been observational, with stratification by glycosylation status revealing differences in infection or disease susceptibility. Information on the underlying mechanisms surrounding the contribution of glycosyltransferases remains very limited (see Outstanding Questions). However, it is becoming apparent that common SNPs in genes such as FUT2 have clinically important predictive capabilities. We are now entering an era in which whole-genome sequencing is becoming increasingly common in both research (for large cohort studies) and at an individual level (as a predictive tool for risk susceptibility). These growing data repositories provide an opportunity to explore effects of glycosylation variability on risks of infection and disease susceptibility at a population-level, as well as at an individual level.
Glycosylation status might, for example, be informative at a population level during infection outbreaks, where identifying at-risk individuals is vital for effective control management and global security. For example, a retrospective analysis of 45 people exposed to severe acute respiratory syndrome (SARS) virus in a Hong Kong hospital in 2003 found that individuals who did not express A or B histo-blood group glycans had reduced frequency of infection (odds ratio 0.18), supporting the NAb-anti-viral hypothesis [63]. Developing stratification strategies based on glycosylation compatibility could therefore inform and reduce infection spread. At an individual level, identification of common genetic infection and disease risk factors, such as FUT2 SNPs, could inform and direct an individual’s behaviour to minimise infection and disease risk, particularly when linked to other risk factors. Further, with the apparent link between glycan variation and antibody repertoire 5, 8, individual glycosylation status may inform patient stratification in clinical settings such as predicting vaccine efficacy and progression of chronic diseases.
A major growing clinical concern is the global increase in prevalence of chronic diseases caused by a diverse range of complex lifestyle, genetic, and environmental factors. Respiratory- and gastrointestinal-associated diseases often have a direct microbial component, and many other seemingly unrelated chronic diseases are also associated with our interactions with microbes, including type 2 diabetes [64], asthma [65], and rheumatoid arthritis [66], to name but a few. Secretor status is associated not only with susceptibility to many of these chronic diseases, but also their progression and severity. This relationship could inform the stratification of patient populations and the prediction of adverse events. For example, stratification of patients with noncystic fibrosis bronchiectasis by secretor status identified that chronic airway infection, lung function, and pulmonary exacerbation frequency were higher in secretor individuals [67].
The frequency of allergic diseases is also increasing rapidly, particularly in developed countries [68]. Early-life antibiotic exposure or a dysregulated microbiota has been linked with allergen sensitisation [65]. Maternal secretor status is linked with infant microbiota composition 44, 69, and nonsecretors are more susceptible to asthma [26], psoriasis [29], and early-life IgE-associated eczema [70]. While it is essential that we better understand how the influence of glycan fucosylation on the microbiome contributes to the development of these conditions, such relationships might also present opportunities for novel therapeutic strategies. For example, might breast milk, supplemented with α(1,2)-fucose, reduce allergic disease susceptibility in at-risk infants? Oral supplementation with α(1,2)-fucose has been shown to be well tolerated and shift the intestinal microbiota in healthy adults [71], highlighting its safety and efficacy as a potential prebiotic.
Concluding Remarks
We are now in the era where data repositories are available that combine genomic sequence data with clinical metadata from large cohorts [13]. The effect of genetic variation in genes that mediate mucosal glycosylation, such as SNPs in FUT2, increases our understanding of immunology at a population level, and creates opportunities to implement effective precision medicine. Testing glycan-mediated mechanisms of infection and disease susceptibility is now warranted to assess their inclusion in clinical and policy practices. Ultimately, precision medicine requires an integrated decision-making model that encompasses extrinsic factors, including diet, mental and physical health, the microbiome, and intrinsic factors, such as human genetics and subsequent phenotypes. While testing population-level variability remains challenging [2], a better understanding of the factors by which glycans mediate disease susceptibility offers the potential to significantly improve clinical outcomes.
Outstanding Questions.
How do the effects of secretor status on infection and disease susceptibility replicate across multiple studies?
Does transcriptional regulation of FUT2 change during health, infection, and disease? Might this influence severity of infection and/or disease?
Does ABO blood group type affect viral transmissibility?
Are viral particles from nonsecretors better able to evade host NAb defences?
With personal genome sequencing becoming increasingly common, might an individual’s knowledge of their infection/disease susceptibility reduce incidence or late diagnosis?
Glossary
- FUT2
the gene encoding an α(1,2)-fucosyltransferase expressed in epithelial cells of multiple mucosal tissues. This enzyme catalyses the addition of L-fucose to the terminal galactose of a glycan in an α1,2 confirmation. This α(1,2)-fucosylated glycan is termed the H antigen.
- Glycan
a polymer of covalently linked sugar monomers.
- Glycosyltransferase
enzymes that catalyse the transfer of a sugar monomer from a donor to an acceptor substrate molecule, forming a glycan. A diverse range of glycosyltransferases exist, each with a particular specificity for sugar monomers and glycosidic linkage.
- Microbiota
all commensal, symbiotic, and pathogenic microorganisms sharing a defined niche.
- Natural antibodies (NAbs)
immunoglobulins that are produced independently of T cell selection. NAbs generally have a weaker affinity, with lower specificity, compared to T cell-dependent immunoglobulins.
- Nonsecretor
an individual who has two loss-of-function mutations in the FUT2 gene and therefore fails to express α(1,2)-fucosylated glycans in mucosal secreted and cell-surface glycoproteins and glycolipids.
Resources
References
- 1.Mathers C. World Health Organization; 2008. The Global Burden of Disease: 2004 Update. [Google Scholar]
- 2.Liston A. Shaping variation in the human immune system. Trends Immunol. 2016;37:637–646. doi: 10.1016/j.it.2016.08.002. [DOI] [PubMed] [Google Scholar]
- 3.McGuckin M.A. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 2011;9:265–278. doi: 10.1038/nrmicro2538. [DOI] [PubMed] [Google Scholar]
- 4.Thornton D.J. Structure and function of the polymeric mucins in airways mucus. Annu. Rev. Physiol. 2008;70:459–486. doi: 10.1146/annurev.physiol.70.113006.100702. [DOI] [PubMed] [Google Scholar]
- 5.Cooling L. Blood groups in infection and host susceptibility. Clin. Microbiol. Rev. 2015;28:801–870. doi: 10.1128/CMR.00109-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Delmotte P. Tumor necrosis factor alpha increases the expression of glycosyltransferases and sulfotransferases responsible for the biosynthesis of sialylated and/or sulfated Lewis x epitopes in the human bronchial mucosa. J. Biol. Chem. 2002;277:424–431. doi: 10.1074/jbc.M109958200. [DOI] [PubMed] [Google Scholar]
- 7.Ferrer-Admetlla A. A natural history of FUT2 polymorphism in humans. Mol. Biol. Evol. 2009;26:1993–2003. doi: 10.1093/molbev/msp108. [DOI] [PubMed] [Google Scholar]
- 8.Dotz V., Wuhrer M. Histo-blood group glycans in the context of personalized medicine. Biochim. Biophys. Acta. 2016;1860:1596–1607. doi: 10.1016/j.bbagen.2015.12.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pickard J.M. Rapid fucosylation of intestinal epithelium sustains host-commensal symbiosis in sickness. Nature. 2014;514:638–641. doi: 10.1038/nature13823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pham T.A. Epithelial IL-22RA1-mediated fucosylation promotes intestinal colonization resistance to an opportunistic pathogen. Cell Host Microbe. 2014;16:504–516. doi: 10.1016/j.chom.2014.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goto Y. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science. 2014;345 doi: 10.1126/science.1254009. 1254009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Varki A. Sialic acids in human health and disease. Trends Mol. Med. 2008;14:351–360. doi: 10.1016/j.molmed.2008.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.The 1000 Genomes Project Consortium A global reference for human genetic variation. Nature. 2015;526:68–74. doi: 10.1038/nature15393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fumagalli M. Widespread balancing selection and pathogen-driven selection at blood group antigen genes. Genome Res. 2009;19:199–212. doi: 10.1101/gr.082768.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Thomsson K.A. MUC5B glycosylation in human saliva reflects blood group and secretor status. Glycobiology. 2005;15:791–804. doi: 10.1093/glycob/cwi059. [DOI] [PubMed] [Google Scholar]
- 16.Ali S. Secretor polymorphism and human immunodeficiency virus infection in Senegalese women. J. Infect. Dis. 2000;181:737–739. doi: 10.1086/315234. [DOI] [PubMed] [Google Scholar]
- 17.Chanzu N.M. Mucosal blood group antigen expression profiles and HIV infections: a study among female sex workers in Kenya. PLoS One. 2015;10 doi: 10.1371/journal.pone.0133049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kindberg E. A nonsense mutation (428G→A) in the fucosyltransferase FUT2 gene affects the progression of HIV-1 infection. AIDS. 2006;20:685–689. doi: 10.1097/01.aids.0000216368.23325.bc. [DOI] [PubMed] [Google Scholar]
- 19.Raza M.W. Association between secretor status and respiratory viral illness. BMJ. 1991;303:815–818. doi: 10.1136/bmj.303.6806.815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kindberg E. Host genetic resistance to symptomatic norovirus (GGII.4) infections in Denmark. J. Clin. Microbiol. 2007;45:2720–2722. doi: 10.1128/JCM.00162-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Currier R.L. Innate susceptibility to norovirus infections influenced by FUT2 genotype in a United States pediatric population. Clin. Infect. Dis. 2015;60:1631–1638. doi: 10.1093/cid/civ165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Blackwell C.C. Non-secretion of ABO antigens predisposing to infection by Neisseria meningitidis and Streptococcus pneumoniae. Lancet. 1986;2:284–285. doi: 10.1016/s0140-6736(86)92103-3. [DOI] [PubMed] [Google Scholar]
- 23.Blackwell C.C. Non-secretion of ABO blood group antigens predisposing to infection by Haemophilus influenzae. Lancet. 1986;2:687. doi: 10.1016/s0140-6736(86)90193-5. [DOI] [PubMed] [Google Scholar]
- 24.Boren T. Attachment of Helicobacter pylori to human gastric epithelium mediated by blood group antigens. Science. 1993;262:1892–1895. doi: 10.1126/science.8018146. [DOI] [PubMed] [Google Scholar]
- 25.Weiss F.U. Fucosyltransferase 2 (FUT2) non-secretor status and blood group B are associated with elevated serum lipase activity in asymptomatic subjects, and an increased risk for chronic pancreatitis: a genetic association study. Gut. 2015;64:646–656. doi: 10.1136/gutjnl-2014-306930. [DOI] [PubMed] [Google Scholar]
- 26.Kauffmann F. Associations of blood group-related antigens to FEV1, wheezing, and asthma. Am. J. Respir. Crit. Care Med. 1996;153:76–82. doi: 10.1164/ajrccm.153.1.8542166. [DOI] [PubMed] [Google Scholar]
- 27.Ronchetti F. ABO/Secretor genetic complex and susceptibility to asthma in childhood. Eur. Respir. J. 2001;17:1236–1238. doi: 10.1183/09031936.01.99109101. [DOI] [PubMed] [Google Scholar]
- 28.Smyth D.J. FUT2 nonsecretor status links type 1 diabetes susceptibility and resistance to infection. Diabetes. 2011;60:3081–3084. doi: 10.2337/db11-0638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tang H. A large-scale screen for coding variants predisposing to psoriasis. Nat. Genet. 2014;46:45–50. doi: 10.1038/ng.2827. [DOI] [PubMed] [Google Scholar]
- 30.Goto Y. Epithelial glycosylation in gut homeostasis and inflammation. Nat. Immunol. 2016;17:1244–1251. doi: 10.1038/ni.3587. [DOI] [PubMed] [Google Scholar]
- 31.Pickard J.M., Chervonsky A.V. Intestinal fucose as a mediator of host–microbe symbiosis. J. Immunol. 2015;194:5588. doi: 10.4049/jimmunol.1500395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Audfray A. Bacteria love our sugars: interaction between soluble lectins and human fucosylated glycans, structures, thermodynamics and design of competing glycocompounds. C. R. Chim. 2013;16:482–490. [Google Scholar]
- 33.Ilver D. Helicobacter pylori adhesin binding fucosylated histo-blood group antigens revealed by retagging. Science. 1998;279:373–377. doi: 10.1126/science.279.5349.373. [DOI] [PubMed] [Google Scholar]
- 34.Tan M., Jiang X. Norovirus and its histo-blood group antigen receptors: an answer to a historical puzzle. Trends Microbiol. 2005;13:285–293. doi: 10.1016/j.tim.2005.04.004. [DOI] [PubMed] [Google Scholar]
- 35.de Rougemont A. Qualitative and quantitative analysis of the binding of GII.4 norovirus variants onto human blood group antigens. J. Virol. 2011;85:4057–4070. doi: 10.1128/JVI.02077-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hu L. Cell attachment protein VP8* of a human rotavirus specifically interacts with A-type histo-blood group antigen. Nature. 2012;485:256–259. doi: 10.1038/nature10996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Huang P. Spike protein VP8* of human rotavirus recognizes histo-blood group antigens in a type-specific manner. J. Virol. 2012;86:4833–4843. doi: 10.1128/JVI.05507-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Imbert-Marcille B.M. A FUT2 gene common polymorphism determines resistance to rotavirus A of the P[8] genotype. J. Infect. Dis. 2014;209:1227–1230. doi: 10.1093/infdis/jit655. [DOI] [PubMed] [Google Scholar]
- 39.Linden S.K. MUC1 limits Helicobacter pylori infection both by steric hindrance and by acting as a releasable decoy. PLoS Pathog. 2009;5 doi: 10.1371/journal.ppat.1000617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bode L. Human milk oligosaccharides: every baby needs a sugar mama. Glycobiology. 2012;22:1147–1162. doi: 10.1093/glycob/cws074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Morrow A.L. Human milk oligosaccharides are associated with protection against diarrhea in breast-fed infants. J. Pediatr. 2004;145:297–303. doi: 10.1016/j.jpeds.2004.04.054. [DOI] [PubMed] [Google Scholar]
- 42.Ruiz-Palacios G.M. Campylobacter jejuni binds intestinal H(O) antigen (Fuc alpha 1, 2Gal beta 1, 4GlcNAc), and fucosyloligosaccharides of human milk inhibit its binding and infection. J. Biol. Chem. 2003;278:14112–14120. doi: 10.1074/jbc.M207744200. [DOI] [PubMed] [Google Scholar]
- 43.Servin A.L. Antagonistic activities of lactobacilli and bifidobacteria against microbial pathogens. FEMS Microbiol. Rev. 2004;28:405–440. doi: 10.1016/j.femsre.2004.01.003. [DOI] [PubMed] [Google Scholar]
- 44.Rausch P. Multigenerational influences of the Fut2 gene on the dynamics of the gut microbiota in mice. Front. Microbiol. 2017;8:991. doi: 10.3389/fmicb.2017.00991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Davenport E.R. ABO antigen and secretor statuses are not associated with gut microbiota composition in 1,500 twins. BMC Genomics. 2016;17:941. doi: 10.1186/s12864-016-3290-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wacklin P. Secretor genotype (FUT2 gene) is strongly associated with the composition of bifidobacteria in the human intestine. PLoS One. 2011;6 doi: 10.1371/journal.pone.0020113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Spor A. Unravelling the effects of the environment and host genotype on the gut microbiome. Nat. Rev. Microbiol. 2011;9:279–290. doi: 10.1038/nrmicro2540. [DOI] [PubMed] [Google Scholar]
- 48.Cho I., Blaser M.J. The human microbiome: at the interface of health and disease. Nat. Rev. Genet. 2012;13:260–270. doi: 10.1038/nrg3182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sanz Y. Microbiome and gluten. Ann. Nutr. Metab. 2015;67(Suppl 2):28–41. doi: 10.1159/000440991. [DOI] [PubMed] [Google Scholar]
- 50.Manichanh C. Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut. 2006;55:205–211. doi: 10.1136/gut.2005.073817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rausch P. Colonic mucosa-associated microbiota is influenced by an interaction of Crohn disease and FUT2 (Secretor) genotype. Proc. Natl. Acad. Sci. U. S. A. 2011;108:19030–19035. doi: 10.1073/pnas.1106408108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tong M. Reprograming of gut microbiome energy metabolism by the FUT2 Crohn’s disease risk polymorphism. ISME J. 2014;8:2193–2206. doi: 10.1038/ismej.2014.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.McGovern D.P. Fucosyltransferase 2 (FUT2) non-secretor status is associated with Crohn’s disease. Hum. Mol. Genet. 2010;19:3468–3476. doi: 10.1093/hmg/ddq248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Janssen W.J. Control of lung defence by mucins and macrophages: ancient defence mechanisms with modern functions. Eur. Respir. J. 2016;48:1201–1214. doi: 10.1183/13993003.00120-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rose M.C., Voynow J.A. Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol. Rev. 2006;86:245–278. doi: 10.1152/physrev.00010.2005. [DOI] [PubMed] [Google Scholar]
- 56.Kubinak J.L., Round J.L. Do antibodies select a healthy microbiota? Nat. Rev. Immunol. 2016;16:767–774. doi: 10.1038/nri.2016.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Panda S., Ding J.L. Natural antibodies bridge innate and adaptive immunity. J. Immunol. 2015;194:13–20. doi: 10.4049/jimmunol.1400844. [DOI] [PubMed] [Google Scholar]
- 58.Sazama K. Reports of 355 transfusion-associated deaths: 1976 through 1985. Transfusion. 1990;30:583–590. doi: 10.1046/j.1537-2995.1990.30790385515.x. [DOI] [PubMed] [Google Scholar]
- 59.Neil S.J.D. HIV-1 incorporates ABO histo-blood group antigens that sensitize virions to complement-mediated inactivation. Blood. 2005;105:4693. doi: 10.1182/blood-2004-11-4267. [DOI] [PubMed] [Google Scholar]
- 60.Preece A.F. Expression of ABO or related antigenic carbohydrates on viral envelopes leads to neutralization in the presence of serum containing specific natural antibodies and complement. Blood. 2002;99:2477–2482. doi: 10.1182/blood.v99.7.2477. [DOI] [PubMed] [Google Scholar]
- 61.Durrbach A. Virus recognition by specific natural antibodies and complement results in MHC I cross-presentation. Eur. J. Immunol. 2007;37:1254–1265. doi: 10.1002/eji.200636129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Guillon P. Inhibition of the interaction between the SARS-CoV Spike protein and its cellular receptor by anti-histo-blood group antibodies. Glycobiology. 2008;18:1085–1093. doi: 10.1093/glycob/cwn093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cheng Y. ABO blood group and susceptibility to severe acute respiratory syndrome. J. Am. Med. Assoc. 2005;293:1450–1451. doi: 10.1001/jama.293.12.1450-c. [DOI] [PubMed] [Google Scholar]
- 64.Hartstra A.V. Insights into the role of the microbiome in obesity and type 2 diabetes. Diabetes Care. 2015;38:159–165. doi: 10.2337/dc14-0769. [DOI] [PubMed] [Google Scholar]
- 65.Taylor S.L. Host–microbiome interactions in acute and chronic respiratory infections. Cell. Microbiol. 2016;18:652–662. doi: 10.1111/cmi.12589. [DOI] [PubMed] [Google Scholar]
- 66.Zhang X. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015;21:895–905. doi: 10.1038/nm.3914. [DOI] [PubMed] [Google Scholar]
- 67.Taylor S.L. FUT2 genotype influences lung function, exacerbation frequency and airway microbiota in non-CF bronchiectasis. Thorax. 2017;72:304–310. doi: 10.1136/thoraxjnl-2016-208775. [DOI] [PubMed] [Google Scholar]
- 68.Wang J. Allergic disease epidemiology. In: Tao A., Raz E., editors. Allergy Bioinformatics. Springer; Netherlands: 2015. pp. 15–41. [Google Scholar]
- 69.Lewis Z.T. Maternal fucosyltransferase 2 status affects the gut bifidobacterial communities of breastfed infants. Microbiome. 2015;3:13. doi: 10.1186/s40168-015-0071-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sprenger N. FUT2-dependent breast milk oligosaccharides and allergy at 2 and 5 years of age in infants with high hereditary allergy risk. Eur. J. Nutr. 2017;56:1293–1301. doi: 10.1007/s00394-016-1180-6. [DOI] [PubMed] [Google Scholar]
- 71.Elison E. Oral supplementation of healthy adults with 2′-O-fucosyllactose and lacto-N-neotetraose is well tolerated and shifts the intestinal microbiota. Br. J. Nutr. 2016;116:1356–1368. doi: 10.1017/S0007114516003354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Silva L.M. Infection-associated FUT2 (fucosyltransferase 2) genetic variation and impact on functionality assessed by in vivo studies. Glycoconj. J. 2010;27:61–68. doi: 10.1007/s10719-009-9255-8. [DOI] [PubMed] [Google Scholar]
- 73.Guillon P. Association between expression of the H histo-blood group antigen, alpha1,2fucosyltransferases polymorphism of wild rabbits, and sensitivity to rabbit hemorrhagic disease virus. Glycobiology. 2009;19:21–28. doi: 10.1093/glycob/cwn098. [DOI] [PubMed] [Google Scholar]
- 74.Lopman B.A. Norovirus infection and disease in an Ecuadorian birth cohort: association of certain norovirus genotypes with host FUT2 secretor status. J. Infect. Dis. 2015;211:1813–1821. doi: 10.1093/infdis/jiu672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Azevedo M. Infection by Helicobacter pylori expressing the BabA adhesin is influenced by the secretor phenotype. J. Pathol. 2008;215:308–316. doi: 10.1002/path.2363. [DOI] [PubMed] [Google Scholar]
- 76.Magalhaes A. Fut2-null mice display an altered glycosylation profile and impaired BabA-mediated Helicobacter pylori adhesion to gastric mucosa. Glycobiology. 2009;19:1525–1536. doi: 10.1093/glycob/cwp131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hurd E.A., Domino S.E. Increased susceptibility of secretor factor gene Fut2-null mice to experimental vaginal candidiasis. Infect. Immun. 2004;72:4279–4281. doi: 10.1128/IAI.72.7.4279-4281.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Thom S.M. Non-secretion of blood group antigens and susceptibility to infection by Candida species. FEMS Microbiol. Immunol. 1989;1:401–405. doi: 10.1111/j.1574-6968.1989.tb02428.x. [DOI] [PubMed] [Google Scholar]
- 79.Sheinfeld J. Association of the Lewis blood-group phenotype with recurrent urinary tract infections in women. N. Engl. J. Med. 1989;320:773–777. doi: 10.1056/NEJM198903233201205. [DOI] [PubMed] [Google Scholar]
- 80.Kinane D.F. ABO blood group, secretor state, and susceptibility to recurrent urinary tract infection in women. Br. Med. J. 1982;285:7–9. doi: 10.1136/bmj.285.6334.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rayes A. A genetic modifier of the gut microbiome influences the risk of graft-versus-host disease and bacteremia after hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2016;22:418–422. doi: 10.1016/j.bbmt.2015.11.017. [DOI] [PubMed] [Google Scholar]
- 82.Innes A.L. The H antigen at epithelial surfaces is associated with susceptibility to asthma exacerbation. Am. J. Respir. Crit. Care Med. 2011;183:189–194. doi: 10.1164/rccm.201003-0488OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Duell E.J. Variation at ABO histo-blood group and FUT loci and diffuse and intestinal gastric cancer risk in a European population. Int. J. Cancer. 2015;136:880–893. doi: 10.1002/ijc.29034. [DOI] [PubMed] [Google Scholar]
- 84.Folseraas T. Extended analysis of a genome-wide association study in primary sclerosing cholangitis detects multiple novel risk loci. J. Hepatol. 2012;57:366–375. doi: 10.1016/j.jhep.2012.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Franke A. Genome-wide meta-analysis increases to 71 the number of confirmed Crohn’s disease susceptibility loci. Nat. Genet. 2010;42:1118–1125. doi: 10.1038/ng.717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Parmar A.S. Association study of FUT2 (rs601338) with celiac disease and inflammatory bowel disease in the Finnish population. Tissue Antigens. 2012;80:488–493. doi: 10.1111/tan.12016. [DOI] [PubMed] [Google Scholar]
- 87.Hazra A. Common variants of FUT2 are associated with plasma vitamin B12 levels. Nat. Genet. 2008;40:1160–1162. doi: 10.1038/ng.210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Tanaka T. Genome-wide association study of vitamin B6, vitamin B12, folate, and homocysteine blood concentrations. Am. J. Hum. Genet. 2009;84:477–482. doi: 10.1016/j.ajhg.2009.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tanwar V.S. Common variant in FUT2 gene is associated with levels of vitamin B12 in Indian population. Gene. 2013;515:224–228. doi: 10.1016/j.gene.2012.11.021. [DOI] [PubMed] [Google Scholar]
- 90.Ishitoya S. Non-secretor status is associated with female acute uncomplicated pyelonephritis. BJU Int. 2002;89:851–854. doi: 10.1046/j.1464-410x.2002.02782.x. [DOI] [PubMed] [Google Scholar]
- 91.Xavier J.M. FUT2: filling the gap between genes and environment in Behçet’s disease? Ann. Rheumat. Dis. 2015;74:618–624. doi: 10.1136/annrheumdis-2013-204475. [DOI] [PubMed] [Google Scholar]