Abstract
Ethnopharmacological relevance
The Chinese Herbal Prescription JieZe-1(JZ-1), added and subtracted from Yihuang Decoction, a famous formula in the 12th year of Kangxi in Qing Dynasty, has a clear effect on Genital Herpes (GH) and no obvious adverse reactions occur clinically. JZ-1 also has preventive and therapeutic effects on Trichomonas vaginitis, Candida albicans vaginitis and GH in vitro and in vivo experiments.
Aim of study
The effect and mechanism of JZ-1 on anti-herpes simplex virus type 2(HSV-2) in vitro focusing on adhesion and penetration stages were investigated.
Materials and methods
A model of HSV-2 infection of VK2/E6E7 was developed. In order to explore JZ-1's anti-HSV-2 effect in vitro, cell morphology, ultrastructural pathology, cell viability and expression of viral glycoprotein D (gD) were assessed at 6 h, 12 h, 18 h, and 24 h of JZ-1 treatment. Then we measured the exact time required for adhesion and penetration of HSV-2 into VK2/E6E7 among a series of times at room temperature and under temperature control techniques. We treated VK2/E6E7 with JZ-1, penciclovir, or berberine and explored the mechanism of JZ-1 in blocking HSV-2 adhesion and penetration of host cells by assessing the cell ultrastructural pathology, viability, viral proteins gB, gD, VP16, ICP5, and ICP4 and host cell proteins HVEM, Nectin-1, and Nectin-2.
Results
HSV-2 can fully adhere and penetrate into VK/E6E7 within 5 mins at room temperature while it takes 60mins under temperature control techniques. JZ-1 and penciclovir showed significant anti-HSV-2 effects, with improved host cell morphologies and increased host cell viabilities observed after treatment for 24 h. The anti-HSV-2 effect of JZ-1 can be detected after treatment for 6 h while that of penciclovir was not obvious until treatment for 12 h. JZ-1 showed distinct effect on HSV-2 adhesion and penetration stages by significantly reducing the expression of viral proteins gB, gD, VP16, ICP5, and ICP4, improving cell morphology and increasing cell viability. However, these effects were not exerted via downregulated expression of membrane fusion-related proteins such as HVEM, Nectin-1, or Nectin-2. The specific anti-HSV-2 mechanism of JZ-1 need to be further explored.
Conclusion
The anti-HSV-2 effect of JZ-1 was superior to that of penciclovir and berberine in vitro, and was mainly mediated by enhancing host cell defense and blocking adhesion and penetration of HSV-2.
Keywords: Chinese herbal prescription JieZe-1, Genital herpes, Herpes simplex virus type 2, VK2/E6E7, Adhesion, Penetration
Graphical abstract
1. Introduction
GH is one of the most prevalent sexually transmitted diseases. More than 500 million people worldwide are infected with GH, and 20–23 million new infections are reported each year according to WHO (Kesharwani et al., 2017). As a member of the herpesvirus family, HSV-2 is the primary causative agent of GH (Mousavi et al., 2017). HSV-2 is a giant dsDNA virus belonging to the α-herpesvirus subfamily, which is mainly transmitted to genital mucosa through sexual behavior. HSV-2 causes genital herpes or ulcers during acute infection (Kesharwani et al., 2017, p. 2), and can migrate to cranial ganglia resulting in lifelong latent infection (Liu et al., 2018). HSV-2 infection during early pregnancy can also cause neonatal encephalitis through placental transmission, resulting in high morbidity and mortality (Vilhelmova-Ilieva et al., 2014). Epidemiological studies have shown that HSV-2 can act as an important synergistic factor for human immunodeficiency virus (HIV) and human papillomavirus (HPV) infection (Nikolic and Piguet, 2010), and increase the risk of HIV-1 infection by 2–3-fold (Mohraz et al., 2018). The prevention and treatment of GH has always been an urgent problem to be solved.
HSV-2 infection is a sequential process involving the interaction of multiple viral proteins with host cell molecules. Among more than ten glycosylated proteins in the viral envelope, only five glycoproteins (gB, gC, gD, and heterodimeric gH/gL) play an essential role in membrane fusion (Sepúlveda-Crespo et al., 2017). After contacting between HSV-2 and host cells, gD binds to specific receptors (HVEM, Nectin-1, Nectin-2, or 3-O-sulfated heparan sulfate) and sequentially recruits gB and gH/gL heterodimers. It results in activation of gH/gL heterodimers, which in turn upregulates the fusion activity of gB, allowing it to be inserted directly into cell membranes through its internal fusion loop to anchor virions to host cells (Luo et al., 2015a). Virions approach the nucleus along intracellular actin and microtubule filaments after entering the cell, and the capsids are stripped at nuclear pores to release viral dsDNA into the nucleus (Nozawa et al., 2004). After the viral genome entering the nucleus, the viral tegument protein VP16 interacts with the host cell transcription factor Oct-1, activating the host cell RNA polymerase complex (Kato et al., 2000) which in turn mediates immediate early viral gene transcription and expression of the immediate early proteins (Akhrameyeva et al., 2011). Expression of immediate early genes including RL2, RS1, UL54, US1, and US12, which are in turn translated to ICP0, ICP4, ICP27, ICP22, and ICP47, generally peaks around 2–4 h after infection (Halford et al., 2001). The immediate early proteins are the primary activators of early viral genes, and their transcription products are required for viral replication (Smith and Schaffer, 1987). Expression of early genes, such as the viral DNA polymerase, ssDNA-binding protein ICP8, and viral thymidine kinase, peaks around 5–7 h after infection (Costa et al., 1997). Expression of late gene transcripts include viral structural proteins such as gB, gC, gD, gK, VP16, VP5, and vhs peaks around 12–15 h after infection (Sampath and Deluca, 2008). The nucleocapsid is comprised of mature structural proteins and forms an envelope surrounding viral DNA to enable exit from the nucleus. Then nucleocapsid is further processed in the cytoplasm to form progeny virions that are released into the extracellular space (Desai and Kulkarni, 2015). Based on the basic biology of HSV-2 infection, anti-HSV-2 drugs can inhibit different targets. Most commonly-used anti-HSV-2 drugs act at the viral gene replication stage and have single action site. These drugs easily elicit resistance (Hopkins et al., 2018), and have little impact on latent and recurrent viral infection (Akhrameyeva et al., 2011). Therefore, the development of drugs acting at the adhesion, penetration, release, and intercellular infection stages of HSV-2 has become an important research goal.
The antiviral effect of Traditional Chinese Medicine (TCM) has been confirmed by clinical practice. It plays a vital role in the treatment of epidemic infection of severe acute respiratory syndrome coronavirus (SARS-CoV) and influenza A virus H1N1 (Ge et al., 2010; Lin et al., 2005). The anti-HSV-1 and anti-HSV-2 effects of TCM have also been verified in vitro and in vivo experiments (Li et al., 2018; Xiong et al., 2011). GH belongs to the categories of “leukorrheagial diseases” and “erosion of vulva”, which was firstly recorded in “A Handbook of Prescriptions for Emergencies” written by Ge Hong in the Eastern Jin Dynasty (Ge, 2016). There are many different TCM names of GH in later books such as “Renzhai Zhizhi Fanglun” and “Medical Prescription”, but there is no uniform standard yet. “Pudendal heat sore” is oftenly cited by most doctors as the TCM diagnosis of GH (Feng and Zhu, 2015). Symptoms of GH include vulvar erosion, ulcers, and searing pain. Disease pathogenesis begins with exogenous wind-heat toxin caused by insanitary or excessive sexual intercourse and the downward flow of dampness-heat in the liver channel. The disease is long-lasting and lingering. General therapies involve internal therapy and external therapy, acupuncture therapy is also recorded (Zhang and Li, 2012). Internal therapy strategies involve coursing wind and dissipating heat, clearing away toxins and removing dampness, calming liver and strengthening spleen, tonifying yin and replenishing the kidney with the oral preparation such as Wu Lin Decoction, Gentianae Decoction for Purging Liver-Fire, Zhibai Dihuang pill and so on (Feng and Zhu, 2015; Zhang and Li, 2012). External therapy strategies involve clearing away heat and toxins, removing dampness and relieving itching, curing leucorrhagia and ulcer with external application of Chinese herbs or Chinese medicine compounds. The treatment strategies was adjusted at different stages of the disease. The treatment at early stage of GH is to eliminate pathogenic factors. When healing is delayed, the treatment is to both strengthen body resistance and eliminate evil. During the incubation period or repeated attacks of GH, the main treatment is to strengthen healthy energy. On the basis of the ancient prescription Yihuang Decoction, a classic prescription of “Fu Qingzhu's Obstetrics and Gynecology” in Qing dynasty of CHINA (Jing, 2012; LI et al., 2017; Liu, 2010; Xin and Tan, 2016), and combined with the modern research results of Chinese herbal medicine, JZ-1 is an in-hospital preparation of Tongji Hospital (Approval Number: Z20103135) composed of Phellodendron chinense Schneid. (Phellodendri Chinensis Cortex), Ginkgo biloba L. (Ginkgo Semen), Solanum nigrum L. (Solanum Nigrum), Taraxacum mongolicum Hand.-Mazz (Taraxaci Herba), Thlaspi arvense Linn. (Herba Patriniae), Dictamnus dasycarpus Turcz. (Dictamni Cortex), Smilax glabra Roxb. (Smilacis Glabrae Rhizoma), Paeonia suffruticosa Andr. (Moutan Cortex), Mentha haplocalyx Briq. (Menthae Haplocalycis Herba), and Borneolum Syntheticum. It is mainly used to treat wet turbid zones, vulvar redness and swelling, ulcers, and other diseases such as female lower reproductive tract infection, with the effect of clearing away heat and toxins, removing dampness, curing leucorrhagia, treating ulcer, strengthening justice, and dispelling evil (Chen et al., 2009a). Clinical data showed that JZ-1 has an excellent therapeutic effect in leucorrhagia, vulva redness, and ulcer. Experimental studies in vitro and in vivo showed that JZ-1 has preventive and therapeutic effects on Trichomonas vaginitis, Candida albicans vaginitis, and GH, while it had little effect on vaginal lactic acid bacteria (Chen et al., 2009b, 2009d, 2009e, 2009c; Hong et al., 2008; Ma et al., 2011; Ping et al., 2005; Yuan et al., 2013).
In this study, we investigated the antiviral effect of JZ-1 during HSV-2 adhesion and penetration stages in vitro, and further explored the underlying antiviral mechanism of JZ-1.
2. Materials and methods
2.1. Cell culture and virus preparation
Human immortalized vaginal epithelial cells (VK2/E6E7) were purchased from the American Type Culture Collection (ATCC, CRL2616) and cultured in CnT-PR medium (CELLnTEC, Swiss) at 37 °C under a humidified atmosphere containing 5% CO2. The cells were grown in monolayers and changed fresh medium every 48 h. The cells were detached by trypsin-ethylenediaminetetraacetic (EDTA) solution (Invitrogen Company, USA) and resuspended in Dulbecco's modified Eagle's medium (DMEM)/F-12 medium (Thermo Scientific, USA) containing 10% fetal bovine serum (FBS) (Invitrogen Company, USA).
The African green monkey kidney cell line (Vero) was purchased from the Chinese Type Culture Collection (CCTCC) and cultured in DMEM (Thermo Scientific, USA) supplemented with 10% FBS at 37 °C under a humidified atmosphere containing 5% CO2. Cells were grown in monolayers and changed fresh medium every 2–3 days, detached by trypsin-EDTA solution and resuspended in DMEM containing 10% FBS.
HSV-2333 strain was purchased from Guangzhou Biotest Biotechnology Development Co., Ltd (Guangdong, China). When Vero cells reached 80–90% confluency, 250 μL of HSV-2 solution, and 3 mL of DMEM containing 2% FBS were added. The cells were cultured at 37 °C under a humidified atmosphere containing 5% CO2 for 48–72 h until more than 80% of the cells were floating. The cells were subjected to three freeze-thaw cycles. After centrifugation, supernatants were removed and stored at −80 °C. Virus-containing solutions were assessed by TCID50 and MTT methods. The titers of virus stocks were about 1010 TCID50/mL, and the survival rate of infected VK2/E6E7 was 40–60% of control group 24 h post-infection. Virus-related experiments were carried out in biosafety secondary laboratory (BSL-2).
2.2. Medicine preparation
2.2.1. Chemicals and reagents
Berberine (Sigma, USA) was stored at room temperature in the dark and diluted in CnT-PR medium to the concentrations of 12.5, 6.25, and 3.125 μmol/L before use. Penciclovir (Zhejiang Jianfeng Pharmaceutical Holdings, China) was stored at room temperature in the dark and diluted in CnT-PR medium to the concentrations of 0.3125, 0.15625, and 0.078125 mg/mL before use. The optimal antiviral concentrations of the two drugs were determined based on the results of the experiments described below (penciclovir: 0.078125 mg/mL; berberine: 6.25 μmol/L).
2.2.2. Preparation of JZ-1
JZ-1 consists of ten traditional Chinese herbs (Table 1 ): 7.13% P. chinense Schneid., 7.13% G. biloba L., 21.38% S. nigrum L., 10.69% T. mongolicum Hand.-Mazz, 21.38% T. arvense Linn., 7.13% D. dasycarpus Turcz., 10.69% S. glabra Roxb., 7.13% P. suffruticosa Andr., 7.13% M. haplocalyx Briq., and 0.2% Borneolum Syntheticum. All the crude drugs were obtained from Hubei Shengdetang Chinese Herbal Pieces Co., Ltd. (Hubei, China).
Table 1.
Composition of Chinese herbal prescription JieZe-1(JZ-1).
| Chinese Name | English Name | Latin Name | Family | Plant Part | Weight(g) |
|---|---|---|---|---|---|
| Huangbo | Phellodendri Chinensis Cortex | Phellodendron chinense Schneid. | Rutaceae | Bark | 10 |
| Baiguo | Ginkgo Semen | Ginkgo biloba L. | Ginkgoaceae | Seed | 10 |
| Longkui | Solanum Nigrum | Solanum nigrum L. | Solanaceae | Whole Plant | 30 |
| Pugongying | Taraxaci Herba | Taraxacum mongolicum Hand.-Mazz | Compositae | Whole Plant | 15 |
| Baijiangcao | Herba Patriniae | Thlaspi arvense Linn. | Cruciferae | Whole Plant | 30 |
| Baixianpi | Dictamni Cortex | Dictamnus dasycarpus Turcz. | Rutaceae | Velamen | 10 |
| Tufuling | Smilacis Glabrae Rhizoma | Smilax glabra Roxb. | Liliaceae | Rhizome | 15 |
| Mudanpi | Moutan Cortex | Paeonia suffruticosa Andr. | Ranunculaceae | Velamen | 10 |
| Bohe | Menthae Haplocalycis Herba | Mentha haplocalyx Briq. | Labiatae | Whole Plant | 10 |
| bingpian | Borneolum Syntheticum | -- | – | – | 0.3 |
JZ-1 is decocted in water and deposited in alcohol in proportion. P. chinense Schneid. was refluxed and extracted twice with 70% ethanol for 1 h each time. The extract was combined and filtered. Take P. suffruticosa Andr. and M. haplocalyx Briq. for steam distillation respectively and collect the distillates, stored them at 4 °C before use. S. nigrum L., T. mongolicum Hand.-Mazz, T. arvense Linn., G. biloba L., D. dasycarpus Turcz., S. glabra Roxb., and the residues of M. haplocalyx Briq., P. chinense Schneid., and P. suffruticosa Andr. were fried twice in water for 1.5 h each time. The decoction was filtered and concentrated to a relative density of 1.08–1.14, adding ethanol to make the alcohol content 50%. After standing for 24 h, the supernatant was taken and ethanol was added to make the alcohol content 75%. Standing still for 24 h, the supernatant was taken and added with extract of P. suffruticosa Andr. before swing. After adding the distilled liquid of P. suffruticosa Andr. and M. haplocalyx Briq., the liquid was placed in the refrigerator for 24 h. The filtrate was heated in 100 °C water bath before adding Borneolum Syntheticum methanol solution (dripping and stirring). The pH was adjusted to 7.2 ± 0.2 with NaHCO3 and the osmotic pressure was adjusted to 280–310mOsm/kgH2O by freezing point osmotic manometer. 0.5 g/ml crude drug solution was obtained.
JZ-1 was stored at room temperature in the dark and diluted in CnT-PR medium to the concentrations of 50, 25, 12.5, 6.25, 3.125, 0.15625, and 0.078125 mg/mL before use. The cytotoxic effect of JZ-1 on VK2/E6E7 was evaluated by MTT assay.
2.2.3. Detection of JZ-1 by high performance liquid chromatography (HPLC)
The main chemical constituents in JZ-1 were identified using 3D-HPLC. Reference standards for berberine, paeoniflorin, neoastilbin, astilbin, paeonol, and dictamnine were purchased from the National Institutes for Food and Drug Control (Beijing, China) and Shanghai Yuanye Bio-Technology Co, Ltd (Shanghai, China).
Standards as dry powder (5.0–7.0 mg) and JZ-1 (1.5 mL) were placed in different volumetric flasks and dissolved in 50% methanol. The samples were sonicated for 20 min, and the resulting solution was filtered through a 0.45 μm membrane filter prior to HPLC. HPLC was performed on a UltiMate 3000 HPLC system (DIONEX Corporation, Sunnyvale, CA, USA) equipped with a DAD detector and an Outstand C18 HPLC column (250 mm × 4.6 mm, 5 μm). The mobile phase included water (A) and methanolin (B) at a flow rate of 1.0 mL/min and a column temperature 26 °C. The detection wavelength was set at 230 nm. Twenty microliters of reference standards were injected. The gradient elution was as follows: 0–50 min (75–40% A, 25–60% B), 50–55 min (40–75% A, 60–25% B), 55–60 min (75% A, 25% B). Chromatographic data was collected and analyzed using Chromeleon software.
Similarities between fingerprints were analyzed by using Similarity Evaluation System for Chromatogram Fingerprint of Traditional Chinese Medicine (version 2004A).
2.3. Primary and secondary antibody
Mouse polyclonal antibodies against HSV-2 gD and gB and rabbit polyclonal antibodies against HSV-2 VP16, ICP4, and ICP5 were obtained from Abcam (UK). Rabbit monoclonal antibodies against Nectin-1, Nectin-2, and HVEM were obtained from Abcam. Alexa Fluor-conjugated goat anti-mouse and Alexa Fluor-conjugated goat anti-rabbit secondary antibodies were obtained from CST (Cell Signaling Technology Inc., USA). Goat anti-mouse secondary antibodies conjugated to infrared fluorescent dyes were obtained from LI-COR Biosciences (USA).
2.4. Time course of HSV-2 infection of VK2/E6E7 at room temperature
VK2/E6E7 were seeded in 96-well plates and incubated with HSV-2 for 1 s, 30 s, 1 min, 2 mins, 3 mins, 4 mins, 5 mins, 10 mins, 30 mins, or 24 h respectively, Virions that did not penetrate into cells were inactivated with phosphate-buffered saline (PBS) (Meilunbio, China), pH 3.0, and neutralized with PBS, pH 11.0. After discarding the supernatant and adding culture medium, the cells incubated at 37 °C under a humidified atmosphere containing 5% CO2 for 24 h. Cell survival was monitored by MTT assay.
2.5. Time course of HSV-2 penetration of VK2/E6E7 using temperature control technology
VK2/E6E7 were seeded in 96-well plates. The cells were precooled at 4 °C for 1 h, then incubated with pre-cooled HSV-2 solution at 4 °C for 3 h to allow virions adhering to cells. The cells were washed twice gently with cold D-Hank's buffered saline solution to remove the free virons and then rewarmed at 37 °C for 1 s, 10 mins, 20 mins, 30 mins, 40 mins, 50 mins, 1 h, or 24 h respectively at 37 °C. Virions that did not penetrate into cells were inactivated with PBS, pH 3.0, and neutralized with PBS, pH 11.0. After discarding the supernatant and adding culture medium, the cells were incubated at 37 °C under a humidified atmosphere containing 5% CO2 for 24 h. Cell survival was monitored by MTT assay.
2.6. HSV-2 adhesion VK2/E6E7
VK2/E6E7 were seeded in 96-well plates. The cells were precooled at 4 °C for 1 h, then a 1:1 vol mixture of pre-cooled HSV-2 solution and pre-cooled drug (or medium) was applied at 4 °C for 3 h to allow virions to adhere to host cells. The cells were washed twice gently with cold D-Hank's buffered saline solution to remove the free virons. All steps were performed on ice.
2.7. HSV-2 penetration of VK2/E6E7
VK2/E6E7 were seeded in 96-well plates. The cells were precooled at 4 °C for 1 h, then pre-cooled HSV-2 solution was applied at 4 °C for 3 h to allow virions to adhere to host cells. These steps were performed on ice. The cells were washed twice gently with Hank's buffered saline solution before applying a 1:1 vol mixture of HSV-2 solution and drug (or medium) at 37 °C for 1 h to allow virions to penetrate into host cells. Virions that did not penetrate into cells were inactivated using PBS, pH 3.0, and neutralized using PBS, pH 11.0.
2.8. MTT assay
VK2/E6E7 were seeded in 96-well plates. Once 90–95% confluent were obtained, the cells were treated as described above and incubated at 37 °C under a humidified atmosphere containing 5% CO2 for 24 h. Then, 100 μL of MTT solution (5 mg/mL) (Servicebio Technology Co., China) was added to each well, and the cells were incubated at 37 °C under a humidified atmosphere containing 5% CO2 for 4 h. The supernatant was discarded and 100 μL of dimethyl sulfoxide (DMSO) (Servicebio Technology Co., China) was added to each well. Crystals were dissolved by shaking for 15 mins and absorbance was measured at 570 nm using a microplate reader (Bio-Tek, USA). Viability was calculated using the following formula: Cell survival rate = (model group OD value-blank pore OD value)/(normal control group OD value-blank pore OD value) × 100%.
2.9. Fluorescence microscopy
VK2/E6E7 were grown in a 24-well plate containing aseptic slides and treated as described above. After treatment, the cells were washed three times with PBS and then fixed in 4% paraformaldehyde solution (Servicebio Technology Co., China) for 15 mins at room temperature. Fixed cells were permeabilized using 0.4% Triton X-100 (Servicebio Technology Co., China) for 30 mins and blocked with 10% goat serum (Servicebio Technology Co., China) for 1 h at room temperature. The cells were incubated with primary antibody then with Alexa Fluor-conjugated goat anti-mouse secondary antibody and 4’, 6-diamidino-2-phenylindole (DAPI) (Cell Signaling Technology Inc., USA). The slides were removed, covered with another slide face down and sealed with anti-fluorescence quenching agent (Servicebio Technology Co., China). The fluorescence expression of gD of HSV-2 was observed under a fluorescence microscope (Olympus, Japan).
2.10. Western blot
VK2/E6E7 were grown in 6-well plates and treated as described above. The supernatant was discarded and the cells were washed twice with PBS. The cells were lysed in RIPA buffer (Servicebio Technology Co., China) containing inhibitors cocktails of proteases (Roche, Swiss). After denaturation, the cell lysates were electrophoresed in 8–12% SDS-PAGE gels (Servicebio Technology Co., China) and transferred onto nitrocellulose membranes (Merck Milipore, Ireland). The membranes were blocked in skim milk (Servicebio Technology Co., China) for 1 h at room temperature, then incubated with primary antibody and secondary antibody. The membranes were imaged on 700 or 800 nm channels using an odyssey instrument (Odyssey LICOR, USA).
2.11. Scanning electron microscopy (SEM)
VK2/E6E7 were grown in 24-well plates containing aseptic slides and treated as described above. The supernatant was discarded and the cells were washed twice with PBS. The cells were fixed with glutaraldehyde at room temperature for 2 h and incubated at 4 °C overnight. The samples were prepared and observed by SEM (SU8100 HITACHI, Japan).
2.12. Transmission electron microscopy (TEM)
VK2/E6E7 were grown in 10 cm2 cell culture dishes and treated as described above. The supernatants were discarded and the cells were washed twice with PBS. The cells were fixed with pre-cooled glutaraldehyde solution, then gently scraped and collected into a 1.5 mL centrifuge tube. After centrifugation, the supernatant was discarded. The cells were fixed with new glutaraldehyde solution at room temperature for 2 h and incubated at 4 °C overnight. The samples were prepared and observed by TEM (HT7700 HITACHI, Japan).
2.13. Statistical analyses
Data were analyzed using GraphPad Prism5. Means and standard deviations were obtained using the statistical software SPSS22.0 and expressed as means ± standard errors of the means. Tests of normality and homogeneity of variance were conducted. Finally, single factor analysis of variance was conducted using Dunnett's test. Statistical significance was assumed for values of P < 0.05.
3. Results
3.1. HPLC profile of JZ-1
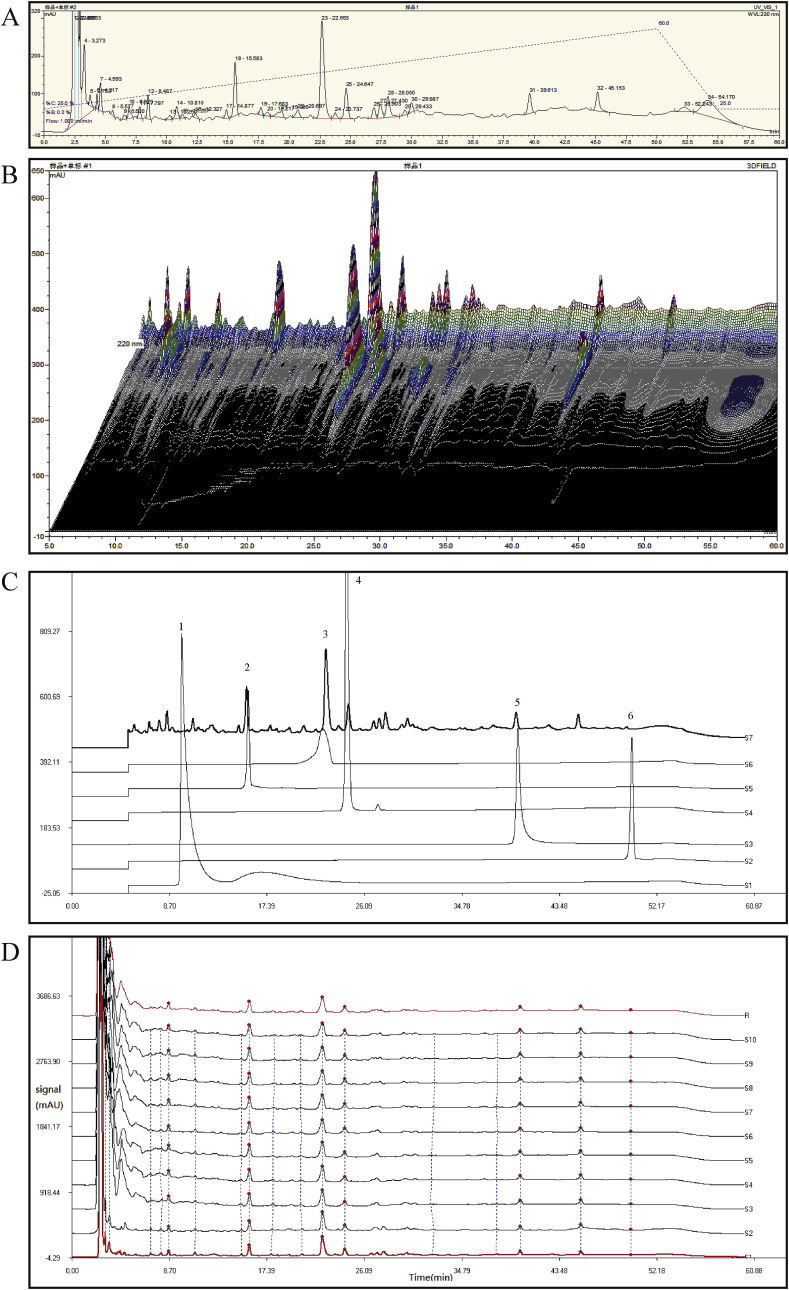
The main constituents of JZ-1 are berberine, paeoniflorin, neoastilbin, astilbin, paeonol, and dictamnine (see Fig. 1).
Fig. 1.
HPLC analysis of JZ-1. (A) The HPLC of JZ-1. (B) The 3D-HPLC of JZ-1. (C) Main chemical constituents in JZ-1 identified using HPLC. Peak number and identity: 1 berberine; 2 paeoniflorin; 3 neoastilbin; 4 astilbin; 5 paeonol; 6 dictamnine. (D) HPLC fingerprint of JZ-1 from ten batches.
3.2. Time course of HSV-2 infection of VK2/E6E7 at room temperature and using temperature control technology
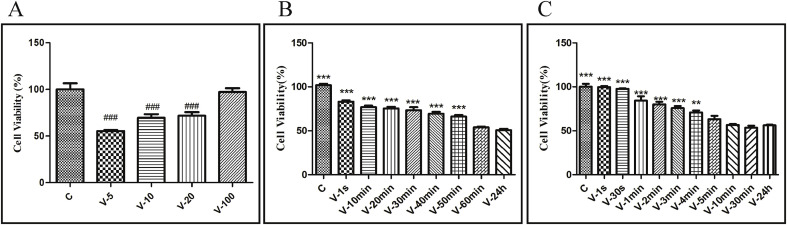
As shown in Fig. 2 A, the survival rates of cells in V-5, V-10, and V-20 groups were 40–60% of control group, suggesting that the HSV-2 preparations met the virulence criteria for a successful model of HSV-2 infection. In followed up experiments, the original HSV-2 solution is diluted to 5 times.
Fig. 2.
(A) Optimal virulence of HSV-2 on VK2/E6E7 in vitro. The original HSV-2 solution is diluted to 5, 10, 20, or 100 times respectively, and incubated with VK2/E6E7 for 24 h, the cell viability is detected by MTT assay. (B) Time for HSV-2 penetrating into VK2/E6E7 at room temperature. HSV-2 solution were incubated with VK2/E6E7 for 1 s, 30 s, 1 min, 2 mins, 3 mins, 4 mins, 5 mins, 10 mins, 30 mins, or 24 h respectively. Virions that did not penetrate into cells were inactivated with phosphate-buffered saline (PBS), pH 3.0, and neutralized with PBS, pH 11.0. Cell viability is detected by MTT assay. (C) Time for HSV-2 penetrating into VK2/E6E7 under temperature control technology. VK2/E6E7 were precooled at 4 °C for 1 h, then incubated with pre-cooled HSV-2 solution at 4 °C for 3 h. Discard the HSV-2 solution and wash the cells twice with cold D-Hank's buffered saline solution. VK2/E6E7 were rewarming at 37 °C for 1 s, 10 mins, 20 mins, 30 mins, 40 mins, 50 mins, 1 h, or 24 h respectively. Virions that did not penetrate into cells were inactivated with PBS, pH 3.0, and neutralized with PBS, pH 11.0. Cell viability is detected by MTT assay. C stands for control group. Data are expressed as mean±SEM for n = 6. ###P < 0.001 versus control group, **P < 0.01versus V-24 h group, ***P < 0.001versus V-24 h group.
As shown in Fig. 2B, compared with V-24 h group, cell viability in control, V-1 s, V-30 s, V-1 min, V-2 mins, V-3 mins, and V-4 mins groups were significantly increased. There were no significant differences comparing V-5 mins, V-10 mins, and V-30 mins groups with V-24 h group, suggesting that virions could fully adhere and penetrate into host cells within 5 mins at room temperature.
As shown in Fig. 2C, compared with V-24 h group, cell viability in control, V-1 s, V-10 mins, V-20 mins, V-30 mins, V-40 mins, and V-50 mins groups were significantly increased. There were no significant differences betweenV-60 mins and the V-24 h group, indicating that virions could fully penetrate into host cells within 60 mins of rewarming at 37 °C.
3.3. HSV-2 adhesion and penetration into VK2/E6E7
As shown in Fig. 3 A, cell viability in HSV-2 adhesion group, HSV-2 penetration group and HSV-2 infection 24 h group were significantly decreased compared with control group, suggesting that a sufficient amount of virus was infecting host cells using temperature control techniques.
Fig. 3.
Characteristics of HSV-2 adhesion and penetration into VK2/E6E7 under temperature control. (A) Effect of HSV-2 adhesion and penetration into VK2/E6E7 on cell viability under temperature control. Data are expressed as mean±SEM for n = 6. (B) Immunofluorescence expression of gD in HSV-2 adhesion and penetration into VK2/E6E7 under temperature control ( × 400). (C) Expression of membrane fusion-associated protein in HSV-2 adhesion and penetration into VK2/E6E7 under temperature control. Each bar represents means ± SEM from three wells. (D) Ultrastructural pathology of HSV-2 adherent VK2/E6E7 under temperature control. In HSV-2 adhesion group, VK2/E6E7 were precooled at 4 °C for 1 h, then a 1:1 vol mixture of pre-cooled HSV-2 solution and pre-cooled medium were applied at 4 °C for 3 h to allow virions to adhere to host cells. In HSV-2 penetration group,VK2/E6E7 were precooled at 4 °C for 1 h, then pre-cooled HSV-2 solution was applied at 4 °C for 3 h to allow virions to adhere to host cells. The cells were washed twice gently with Hank's buffered saline solution before applying a 1:1 vol mixture of HSV-2 solution and medium at 37 °C for 1 h to allow virions to penetrate into host cells. Virions that did not penetrate into cells were inactivated using PBS, pH 3.0, and neutralized using PBS, pH 11.0. In HSV-2 infection 24 h group, VK2/E6E7 and HSV-2 solution were co-incubated at 37 °C for 24 h. C stands for control group, V1 stands for HSV-2 adhesion group, V2 stands for HSV-2 penetration group, V stands for HSV-2 infection 24 h group. **P < 0.01 versus control group, ***P < 0.001 versus control group.
As shown in Fig. 3B, gD was mainly detected on the cell surface of VK2/E6E7 in adhesion group, and in the cytosol of VK2/E6E7 in penetration group, however in longtime incubation group, gD was detected in the nuclear membrane of host cells.
As shown in Fig. 3C, compared with control group, expression of gD, VP16, and ICP5 in HSV-2 adhesion and penetration groups were significantly increased, expression of HSV-2 immediate early protein ICP4 in HSV-2 adhesion and penetration groups were not significantly different compared with control group. Compared with control group, expression of gD, VP16, ICP5, and ICP4 were significantly increased in HSV-2 infection 24 h group (P < 0.001).
As shown in Fig. 3D, the ultrastructure of cells were observed by SEM. VK2/E6E7 in control group were long and fusiform, with large volumes, smooth surfaces, and few pseudopods and spinous processes. By contrast, cells in the virus adhesion group were rounded, with reduced volumes, rough surfaces, and many pseudopods and spinous processes. Attached HSV-2 virions were observed on the cell surface and cell pseudopods.
Results above demonstrated that the use of temperature control techniques ensured a sufficient amount of HSV-2 infecting VK2/E6E7 during the adhesion and penetration phases. Virions remained at the adhesion and penetration stages and did not enter nuclei to begin replication under temperature control technology, and virus adhesion can result in changes in cell morphology.
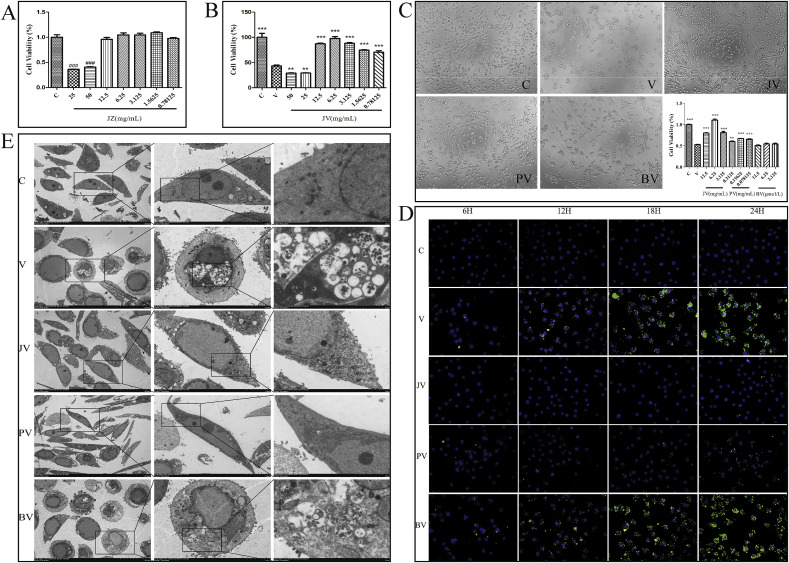
3.4. Effect of JZ-1 on HSV-2 infection of VK2/E6E7
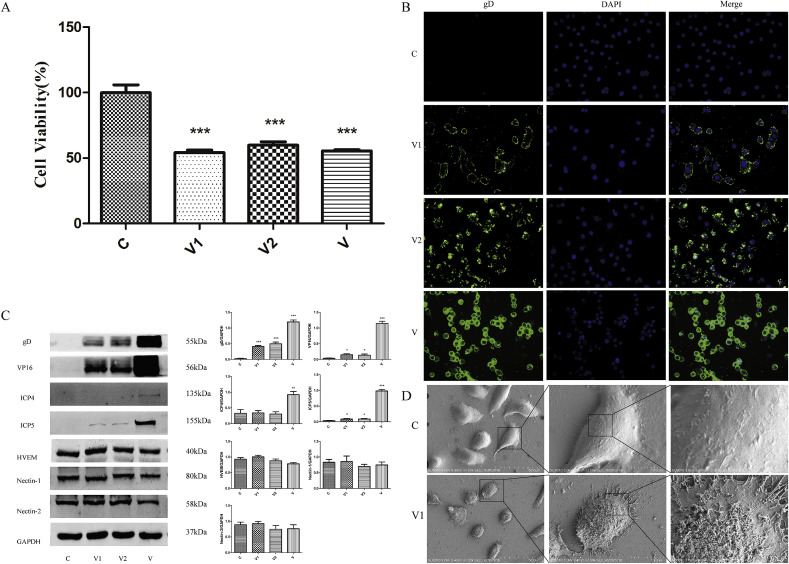
As shown in Fig. 4 A and Fig. 4B, MTT assay was used to evaluate the cytotoxic effect of JZ-1 on VK2/E6E7. A significant reduction of the cell viability was observed at concentration >25 mg/mL while there was no obvious cytotoxic effect of JZ-1 at concentration≤12.5 mg/mL. Also the anti-HSV-2 activity of JZ-1 was monitored by MTT test. JZ-1 showed significant cell protective activity at the concentration of 12.5 mg/mL, 6.25 mg/mL, 3.125 mg/mL, 1.5625 mg/mL, and 0.78125 mg/mL, while JZ-1 of 6.25 mg/mL showed the strongest antiviral effect among them. The concentration of JZ-1 used in followed up experiments were 6.25 mg/mL.
Fig. 4.
Anti-HSV-2 effect of JZ-1 in vitro after 24 h of incubation. (A) Effects of different concentrations of JZ-1 on cell viability of VK2/E6E7 after 24 h of incubation by MTT assay. (B) Antiviral effects of different concentrations of JZ-1 against HSV-2 infection by MTT assay. (C) Effect of JZ-1 on morphology and viability of HSV-2 infected VK2/E6E7 ( × 100). (D) Effect of JZ-1 on immunofluorescence expression of gD in HSV-2 infected VK2/E6E7 ( × 400). (E) Ultrastructural observation of JZ-1 intervention on HSV-2 infection of VK2/E6E7. VK2/E6E7, HSV-2 solution, and drugs (or medium) were co-incubated at 37 °C for 24 h. C stands for control group, V stands for model group, JZ sands for different concentrations of JZ-1 group, JV sands for JZ-1 treatment group, PV stands for penciclovir treatment group, BV stands for berberine treatment group. Data are expressed as mean±SEM for n = 6. **P < 0.01 versus V group, ***P < 0.001 versus V group, ###P < 0.001 versus Control group.
As shown in Fig. 4C, a large number of HSV-2-infected cells changed from fusiform to spherical shape and showed alterations in refractive index. Moreover, the cell gap widened, intercellular connections disappeared, and the number of cells was significantly decreased compared with the control group. The morphologies and cell numbers in JZ-1 and penciclovir groups were significantly improved compare to HSV-2 alone group. The cell morphologies and cell numbers in berberine group were similar to HSV-2 alone group. Compared with control group, cell viability in HSV-2 alone group was significantly decreased. Compared with HSV-2 alone group, cell viability in JZ-1 groups (12.5, 6.25, and 3.125 mg/mL) and the penciclovir groups (0.3125, 0.15625, and 0.078125 mg/mL) was significantly increased. The other groups were not statistically different from the HSV-2 alone group.
As shown in Fig. 4D, Immunofluorescence expression of gD in HSV-2 alone group was minimal at 6 h after infection and gradually increased with time. Expression of gD in JZ-1 group was lower than HSV-2 alone group at 6 h, while there were no differences compare penciclovir and berberine groups with HSV-2 alone group. Compared with HSV-2 alone group, expression of gD in JZ-1 and penciclovir groups was significantly reduced at 12 h, 18 h, and 24 h. JZ-1 showed better antiviral effect than penciclovir. There was no significant difference in gD expression between berberine group and HSV-2 alone group at 12 h, 18 h, and 24 h.
As shown in Fig. 4E, the ultrastructure of cells were observed by TEM. VK2/E6E7 in control group showed a long fusiform shape, while cells in HSV-2-treated group became rounded, with condensed and deep-stained nuclei, concentrated chromatin, mitochondrial swelling, and endoplasmic reticulum expansion. A large number of vesicle-encapsulated mature and immature virions were observed in cell membrane, cytoplasm, and nucleus. The number of virions observed in cell membrane and intracellular regions in JZ-1 and penciclovir groups were significantly lower than HSV-2 alone group. The cell morphologies and number of virions in berberine group were not significantly different from those in the HSV-2 alone group.
Observation of cell morphology by light microscopy and TEM indicates that JZ-1 and penciclovir could inhibit HSV-2 infection in vitro and reduce damage to cells, while berberine showed no such effects. The results of MTT assays also demonstrated that JZ-1 and penciclovir could significantly inhibit HSV-2 infection and improve cell viability at treatment of 24 h, consistent with the results above. Immunofluorescence expression of gD was extremely low at 6 h of infection, and increased significantly after 12 h of HSV-2 infection. Only JZ-1 showed an antiviral effect at 6 h, while penciclovir and berberine showed no such effect. However, JZ-1 and penciclovir both showed anti-HSV-2 effects in vitro at 12 h, 18 h, and 24 h, and the anti-HSV-2 effect of JZ-1 was superior to that of penciclovir. The results indicate that JZ-1 may play an antiviral role during the early stages of viral infection.
3.5. Effect of JZ-1 on HSV-2 adhesion and penetration into VK2/E6E7 using temperature control techniques
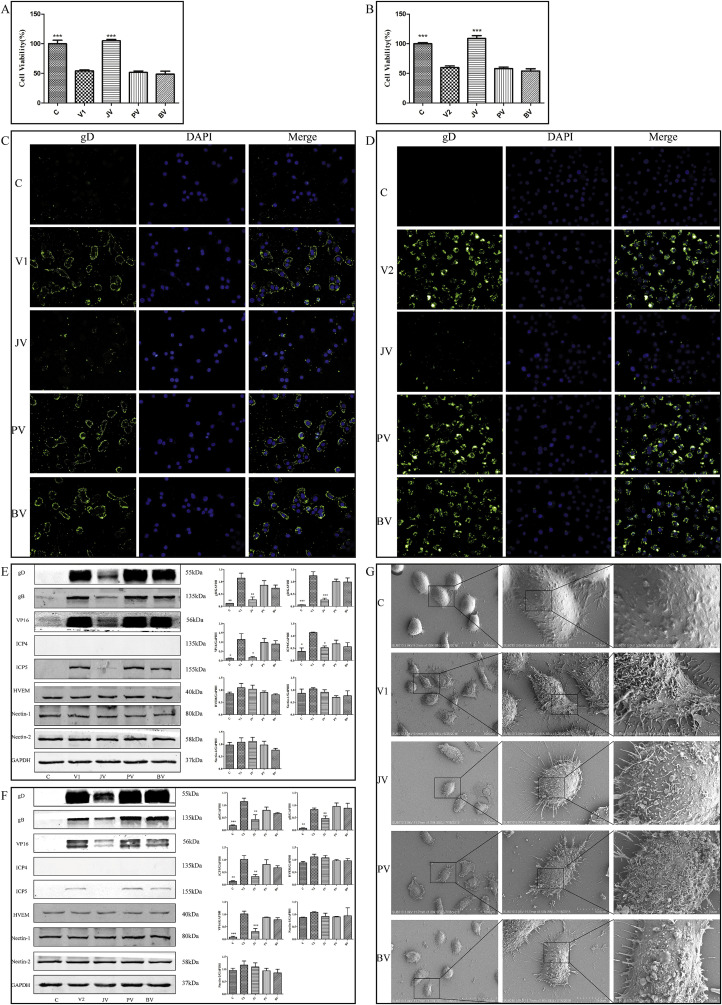
As shown in Fig. 5 A and B, viability of VK2/E6E7 in HSV-2 adhesion and penetration groups was significantly reduced compared with control group. However, cell viability in JZ-1 group was significantly increased compared with HSV-2 adhesion and penetration groups.
Fig. 5.
Effect of JZ-1 on HSV-2 adhesion and penetration into VK2/E6E7. (A) (B) Effect of JZ-1 on cell viability of HSV-2 adhesion (A) and penetration (B) into VK2/E6E7. Data are expressed as mean±SEM for n = 6. (C) (D) Effect of JZ-1 on immunofluorescence expression of gD in HSV-2 adhesion (C) and penetration (D) into VK2/E6E7 ( × 400). (E) (F) Effect of JZ-1 on the expression of membrane fusion-associated protein in HSV-2 adhesion (E) and penetration (F) into VK2/E6E7. Each bar represents means ± SEM for 3 wells. (G) Ultrastructural observation on anti-HSV-2 adhesion effect of JZ-1. In HSV-2 adhesion group, VK2/E6E7 were precooled at 4 °C for 1 h, then a 1:1 vol mixture of pre-cooled HSV-2 solution and pre-cooled drugs (or medium) were applied at 4 °C for 3 h to allow virions to adhere to host cells. In HSV-2 penetration group,VK2/E6E7 were precooled at 4 °C for 1 h, then pre-cooled HSV-2 solution was applied at 4 °C for 3 h to allow virions to adhere to host cells. The cells were washed twice gently with Hank's buffered saline solution before applying a 1:1 vol mixture of HSV-2 solution and drugs (or medium) at 37 °C for 1 h to allow virions to penetrate into host cells. Virions that did not penetrate into cells were inactivated using PBS, pH 3.0, and neutralized using PBS, pH 11.0. C stands for control group, V1 stands for HSV-2 adhesion group, V2 stands for HSV-2 penetration group, JV stands for JZ-1 treatment group, PV stands for penciclovir treatment group, BV stands for berberine treatment group.*P < 0.05 versus V group,**P < 0.01 versus V group, ***P < 0.001 versus V group.
As shown in Fig. 5C and D, significant immunofluorescence expression of gD was observed in HSV-2 adhesion and penetration groups. Expression of gD in JZ-1 group was decreased compared with HSV-2 adhesion and penetration groups. By contrast, there was no significant difference in expression of gD compare penciclovir and berberine groups with HSV-alone group, indicating that JZ-1 has a clear inhibitory effect on HSV-2 adhesion and penetration.
As shown in Fig. 5E and F, expression of the viral proteins gD, gB, VP16, and ICP5 in JZ-1 group was significantly decreased compared with HSV-2 adhesion and penetration groups. By contrast, in penciclovir and berberine groups, expression of gD, gB, VP16, and ICP5 did not differ from HSV-2 alone group. There were no significant differences in the expression of HVEM, Nectin-2, and Nectin-1 between the HSV-2 alone group and other groups.
As shown in Fig. 5G, the ultrastructure of cells were observed by SEM. VK2/E6E7 in control group were long and fusiform and had large volumes, smooth surfaces, and few pseudopods and spinous processes. By contrast, cells in HSV-2 adhesion group became rounded, with decreased volumes and increased number of pseudopods and spinous processes. More virions were observed attaching to the cell surface in HSV-2 adhesion group. However, the number of attached virions in JZ-1 group was significantly lower than HSV-2 adhesion group, and cell morphology was better in JZ-1 group than in HSV-2 adhesion group. The cell morphologies and numbers of virions in penciclovir and berberine groups were not significantly different from HSV-2 adhesion group.
The results of MTT assays and immunofluorescence microscopy indicated that JZ-1 could inhibit HSV-2 adhesion and penetration into VK2/E6E7 in vitro, while penciclovir and berberine had no such effects. Expression of the immediate early protein ICP4 indicated that virions remain at the adhesion and penetration stage and have not yet entered the nucleus using temperature control technology. Expression of the viral proteins gB, gD, VP16, and ICP5 indicated that JZ-1 has an inhibitory effect on HSV-2 adhesion and penetration into VK2/E6E7. However, there was no significant differences in the expression of HVEM, Nectin-1, and Nectin-2 among the control group, the HSV-2 infection group and the treatment groups as showed by Western blot, suggesting that HSV-2 infection and drug treatment may not alter the expression level of host cell receptors. The ultrastructure of cells observed by SEM indicates that JZ-1 inhibited adhesion of HSV-2 to VK2/E6E7 in vitro and improved health of the cells, while penciclovir and berberine showed no such effects.
4. Discussion
The incidence of GH is on the rise, bringing great pain to patients and causing increased rates of fetal abortion, permanent nerve injury, and neonatal malformation through placental transmission (Schiffer and Gottlieb, 2017). At present, nucleoside drugs such as acyclovir, ganciclovir, and penciclovir are commonly used to treat HSV-2 infections. These drugs inhibit the viral DNA polymerase via triphosphorylation, affecting viral replication (Benzekri et al., 2018). However, drug resistance rates are increasing, especially among immunosuppressed individuals (Dai et al., 2018). There is currently no effective HSV-2 vaccine (Churqui et al., 2018), so the exploration of natural plant pharmaceuticals and their active components has become a hot spot in the development of new antiviral drugs (Kesharwani et al., 2017; Yildirim et al., 2016).
“Fu Qingzhu's Obstetrics and Gynecology” was written by Fu Shan, a famous Chinese medical scientist, in the 12th year of Kangxi in Qing Dynasty (1673). It is an authoritative monograph of obstetrics and gynecology with great clinical value (Qian, 2009; Zhu et al., 2015). Fu Shan emphasized the theory of viscera (Zangfu), Qi and blood, channels and collaterals. He paid attention to the dialectics of the liver, spleen, and kidney, tonifing the qi and blood of women, and dispelling evil by strengthening justice. His medication is precise and close to the clinic, and the prescription he created is still in use today (Zhang and Zhang, 1987). Yihuang Decoction is one of the most classical prescriptions in “Fu Qingzhu's Obstetrics and Gynecology”. It is composed of P. chinense Schneid., G. biloba L., Dioscorea opposita Thunb. (Dioscoreae Rhizoma), Plantago asiatica L. (Plantaginis Semen), and Euryale ferox Salisb. (Euryales Semen) with the effect of clearing heat and dehumidifying, tonifying the kidney, and arresting leukorrhagia (Jing, 2012; Liu, 2010). Yihuang Decoction has a remarkable clinical effect on pelvic inflammation, cervicitis, and vaginitis according to clinical study (LI et al., 2017; Xin and Tan, 2016). In JZ-1 recipe, P. chinense Schneid., bitter in flavor, cold in property, acting on the kidney and urinary bladder channels with the effects of clearing away heat, removing dampness, purging fire, and detoxication, especially be good at clearing away dampness and heat in lower warmer(the term, Sanjiao, is peculiar to TCM). The therapeutic effects of P. chinense Schneid. including anti-inflammatory, antioxidant, anticancer, antimicrobial, hypotensive, antiarrhythmic, and antipyretic agents have been confirmed by modern pharmacological studies (Sun et al., 2019). P. chinense Schneid. can exert its anti-inflammatory effect by down-regulating the release of tumor necrosis factor-α (TNF-α) and interleukin 1β (IL-1β) (Cuéllar et al., 2001). G. biloba L., bitter, sweet, and pucker in flavor, natured in property, acting on the kidney and lung channels with the effect of inducing astringency, removing dampness, and curing leukorrhagia, mainly treating turbid dampness and leukorrhagia. Many experimental and clinical studies have approved the therapeutic effects of G. biloba L. including DNA repair, hepatoprotective, antioxidant, photoprotective, and anti-inflammatory activities (Mohanta and Zubaidha, 2014). The extracts of G. biloba L. could significantly decrease the release of VCAM-1 induced by TNF-α by inhibiting the NF-κB pathway (Piazza et al., 2019). P. chinense Schneid. and G. biloba L. are both used as monarch drugs (originated from Yihuang Decoction). S. nigrum L., bitter and sweet in flavor, slightly cold in property, acting on the lung and urinary bladder channels with the effects of clearing away heat, removing toxicity, detumescence, and relieving itching, mainly treating carbuncle, eczema, and dermatitis. S. nigrum L. is wildly used in TCM for the treatment of a wide range of diseases including some eye infections, seizure, epilepsy, inflammation, pain, diarrhea, ulcer, and jaundice (Campisi et al., 2019). T. mongolicum Hand.-Mazz, bitter and sweet in flavor, cold in property, acting on the liver and stomach channels with the effects of clearing away heat, removing toxicity, reducing swelling, and dissipating mass. T. mongolicum Hand.-Mazz has the effects of anti-virus, antibacterial, anti-inflammation, and enhancing immunity according to modern pharmacological studies (Jiang et al., 2017; Ling and Zheng, 1998). In vitro experiments have shown that T. mongolicum Hand.-Mazz could influence certain inflammatory mediators (TNF-α, COX-2, IL-1, iNOS) in leukocytes, astrocytes, and macrophages (González-Castejón et al., 2012). T. arvense Linn., bitter and acrid in flavor, cold in property, acting on the liver, stomach, and large intestine channels with the effects of relieving heat and detoxification, dispelling silt, and purulence. It has been reportedly used for the management of several diseases including abscess of lung, dysentery, enteritis, intestines carbuncle, conjunctivitis, constipation, hepatitis, bellyache, postpartum blood stasis, furuncle, and swollen. T. arvense Linn. could decrease the infiltration of inflammatory cells and expression of cytokines in upper genital tract of multipathogen-infected rats (Zou et al., 2015). S. nigrum L., T. mongolicum Hand.-Mazz, and T. arvense Linn. are used as minister drugs. S. glabra Roxb., sweet in flavor and natured in property, acting on the liver and stomach channels with the effects of dehumidification, detoxication, and easing joint movement. It is testified in clinic treatment that it can cure syphilitic poisoned sores, morbid leucorrhea, carbuncle toxin, limb hypertonicity, strangury due to heat, eczema pruritus, and many other human ailments (Hua et al., 2018). The extracts of S. glabra Roxb. showed immunomodulatory potential by activating macrophages and enhancing host immunity, which makes it a novel immunomodulator for application in immunological diseases (Wang et al., 2017). D. dasycarpus Turcz., bitter in flavor, cold in property, acting on the spleen and stomach channels with the effects of clearing heat and drying dampness, dispelling wind and detoxication. It has been wildly used in TCM for the treatment of rheumatism, itching, bleeding, chronic hepatitis, jaundice, and skin diseases (Lv et al., 2015). P. suffruticosa Andr., bitter and acrid in flavor, slightly cold in property, acting on the heart, liver, and kidney channels with the effects of clearing heat and cooling blood, promoting blood circulation and removing blood stasis. It is clinically used to treat gynecological diseases like endometriosis, breast hyperplasia, and uterine fibroids. It can also be used to treat digestive diseases like acute pancreatitis, ulcerative colitis, and cholecystitis (WU et al., 2019). S. glabra Roxb., D. dasycarpus Turcz., and P. suffruticosa Andr. are used to clear heat, remove toxicity, eliminate carbuncle, remove dampness, relieve itching, and treat ulcer as assistant drugs. These ingredients and guiding drugs with the functions of clearing and detoxification, aczoiling, relieving itching, and inducing resuscitation are worked together to clear away heat and toxins, remove dampness, cure leucorrhagia, and treat ulcer.
The efficacy of JZ-1 decoction against GH has been confirmed through clinical practice. Previous studies have found that JZ-1 can play a role in sustaining health and dispelling disease by affecting the innate immune system, although its specific antiviral stages and mechanisms remain unclear (Ai and Chen, 2017; Huang and Chen, 2018). To explore the antiviral effect and mechanism of JZ-1 further, we established an in vitro model of HSV-2 adhesion and penetration of VK2/E6E7.
Firstly, we explored the anti-HSV-2 effects of JZ-1, penciclovir and berberine at 6 h, 12 h, 18 h, and 24 h of treatment. Immunofluorescence microscopy analysis of gD expression showed that only JZ-1 had an antiviral effect at the treatment of 6 h. Since the late gene transcription products of HSV-2, including gB, gC, gD, gE, gG, and other viral structural proteins, are produced significantly 12–15 h after infection (Ibáñez et al., 2018; Su et al., 2017), the gD observed in host cells at 6 h of infection was mostly derived from the original virons that infected the cells, suggesting that JZ-1 plays a vital role in the early stages of viral infection. Both penciclovir and JZ-1 showed apparent antiviral effects at the treatment of 12 h, 18 h, and 24 h according to immunofluorescence microscopy analysis of gD expression, suggesting that with the extension time of HSV-2 infection, penciclovir, which acts on the replication stage of HSV-2, shows antiviral efficacy. Morphological observations and analysis of cell survival after 24 h of infection showed that cell survival rate and cell morphology (including refractive index, cell adherent state, intercellular space, and intercellular junctions) in the JZ-1 and penciclovir groups were superior to those of HSV-2 alone group. Morphological observation and virions count by TEM confirmed the anti-HSV-2 effects of JZ-1 and penciclovir in vitro. Cells in the HSV-2 alone group became significantly rounded and showed evidence of nucleus pyknosis, chromatin concentration, mitochondrial swelling, endoplasmic reticulum expansion, and other organelle damage. The morphology of cells in JZ-1 and penciclovir groups was similar to the control group, suggesting that JZ-1 can not only improve cell survival but also maintain the normal function of cells by protecting their organelles. As a nucleoside antiviral drug, the anti-HSV-2 mechanism of penciclovir is competitive inhibition of the HSV-2 polymerase, thus selectively inhibiting synthesis and replication of viral DNA (Kesharwani et al., 2017). The antiviral effect of JZ-1 was superior to penciclovir at the treatment of 12 h, 18 h, and 24 h, indicating that the antiviral effect of JZ-1 is not only exerted in the early stage of infection, but also might in the replication stage.
Adhesion and penetration of HSV-2 into host cells are sequential processes, HSV-2 can fully adhere and penetrate into host cells rapidly at room temperature (Tao, 2010), but the exact time required for these steps has not been reported yet. Firstly, we explored the time required for HSV-2 adhesion and penetration of VK2/E6E7 at room temperature in detail. Result showed that HSV-2 could completely adhere and penetrate into host cells within 5 mins at room temperature which was too short to distinguish, and drug treatments are difficult to complete within 5 mins. Since temperature can impact viral infection, we chose to use temperature control technology to delay the process of HSV-2 adhesion and penetration (Tao, 2010), hopefully allowing us to distinguish individual stages of the viral life cycle.
Using temperature control technology, VK2/E6E7 were precooled at 4 °C for 1 h, then HSV-2 solution was applied at 4 °C for 3 h to allow HSV-2 adhesion to host cells (Mukherjee et al., 2013)., The expression of gD was mostly observed on the host cell membrane, with only sparse gD intracellular expression under fluorescence microscope. This way, HSV-2 adhesion to VK2/E6E7 was successfully modeled. A large number of virions adhered to the cell membrane in the virus adhesion group were observed directly by SEM, confirming that temperature control technology can be used to restrict HSV-2 virions adhesion to cell membranes. The result of Western blot showed that expression of gD in the virus adhesion group was significantly higher than the control group using temperature control techniques, while there was no significant expression of the immediate early protein ICP4. Thus, we could exclude the possibility that virions had entered the nucleus to begin the replication cycle, ensuring that most of the virions remained at the stage of adhesion. Cell survival in the virus adhesion group was only about 50% of the control group at 24 h after temperature control treatment, indicating that even if the free virons are washed off, there were still enough virions attached to cell membrane to infect host cells and reduce their survival using temperature control technology. Technical support was provided for the study of the anti-HSV-2 adhesion effect of JZ-1 in the follow-up experiment.
We found that, unlike at room temperature, virions that had adhered to host cells could fully penetrate into host cells after rewarming at 37 °C for 1 h. It provides the possibility for the experimental operation to study the action of drugs. We precooled VK2/E6E7 at 4 °C for 1 h, then applied the HSV-2 solution at 4 °C for 3 h to allow the virions to adhere to host cells. The cells were rewarmed at 37 °C for 1 h to allow virions to penetrate into host cells (Mukherjee et al., 2013). Immunofluorescence expression of gD was mainly in the cytoplasm and rarely at cell membrane and nucleus. Thus, a model of HSV-2 penetration into VK2/E6E7 was successfully established. The result of western blot showed that the expression of gD in the virus penetration group was significantly higher than the control group, and there was no significant expression of immediate early protein ICP4 in the virus penetration group using temperature control techniques. Thus, the possibility that virions had entered the nucleus and began the replication cycle could be ruled out, with most virions remaining at the penetration stage. Cell survival at the HSV-2 penetration group was only about 50% of the control group at 24 h after temperature control treatment, indicating that there were enough virions penetrated into host cells using temperature control technology. It laid a foundation for exploring the mechanism of JZ-1's inhibitory effect on HSV-2 penetration of VK2/E6E7 in the following experiment.
Based on the characteristics of HSV-2 adhesion and penetration of VK2/E6E7 using temperature control techniques, we selected the appropriate temperature control methods and intervention times to explore the inhibitory effect of JZ-1 on HSV-2 adhesion and penetration into VK2/E6E7. Cell survival rate in the JZ-1 group was significantly higher than the virus adhesion and penetration groups at 24 h, suggesting that JZ-1 could efficiently inhibit the HSV-2 infection. Immunofluorescence expression of gD confirmed the anti-HSV-2 adhesion and penetration effect of JZ-1 based on the expression and location of viral proteins. The result of SEM demonstrated that the number of virions attached to the cell membrane in the JZ-1 group was significantly lower than the HSV-2 adhesion group. The morphologies of cells, including cell volume, shape, number of pseudopods, number of spinous processes, integrity of cell membrane, and smoothness was improved in the JZ-1 group compared with the virus adhesion group. We speculate that JZ-1 may play an anti-HSV-2 adhesion role by maintaining the normal morphological function of cells. The numbers of virions attached to the cell surface and cell morphologies in penciclovir group were closer to the virus adhesion group, suggesting that penciclovir had no effect on the adhesion stage of HSV-2.
To further explore the antagonistic effect and mechanism of JZ-1 on HSV-2 adhesion and penetration, we assessed the expression of viral envelope proteins, viral tegument protein, viral capsid protein, viral replication phase protein, and host cell receptors at the HSV-2 adhesion and penetration stages by western blot. The results showed that expression of viral envelope proteins gD and gB, viral tegument protein VP16, and viral capsid protein ICP5 in the JZ-1 group were significantly lower than the HSV-2 adhesion and penetration groups. Binding of gB to heparan sulfate proteoglycan (HSPG) on the surface of host cells can shorten the distance between HSV-2 and cells to initiate the adhesion of virus to host cells (Luo et al., 2015b). Binding of gD to HVEM, Nectin-1, and Nectin-2 can induce the fusion of virus envelope and cell membrane (Liu et al., 2016a). gB and gD are essential for mediating the infection of HSV-2 to host cells (Fujimoto et al., 2019). Our results demonstrated that JZ-1 could effectively inhibit adhesion and penetration of virions into host cells by down-regulating the expression of viral envelope proteins and then reduce its binding to cell receptors. As a late gene transcripts, HSV-2 tegument protein VP16 is not only the key factor of HSV-2 lytic mode infection but also critical for the virus to be reactivated from its latent state (Zhang et al., 2012). The reduction of VP16 expression indicates that JZ-1 can not only anti-HSV-2 infection of VK2/E6E7, but also interfere with HSV-2 intracellular replication and reinfection by inhibiting the expression of replication gene mediated by VP16 and reducing the activation of latent virus. As an immediate early protein, which is required for viral replication, ICP4 is transcribed by immediate early genes, and its expression shows entry of HSV-2 into the replication cycle (Zhang et al., 2018). There was no significant expression of ICP4 in each group, since the virons only remained at the adhesion and penetration stages and had not yet undergone replication. After binding of the viral envelope protein gD to the host cell membrane receptor, the polypeptide chain structure and protein conformations changes to induce the fusion of the virus and the cell membrane, which in turn leads to the penetration of virions into host cells (Liu et al., 2016b). There was no significant difference in the expression of the host cell receptors HVEM, Nectin-1, and Nectin-2 between the control group, the virus infection group and the treatment group, suggesting that viral infection and drug treatments did not alter the expression level of host cell receptors. Whether the inhibitory effect of JZ-1 on viral adhesion and penetration is exerted by changing the conformations of these receptors is worthy of further study. Expression of downstream proteins and terminal effector molecules after fusion of HSV-2 with cell membranes can also be further assessed to study the mechanism of the anti-HSV-2 effect of JZ-1 (Bright et al., 2012). At the same time, infection of HSV-2 to host cells are cross-regulated by multiple molecules and pathways. JZ-1 can exert its antiviral effect through a variety of signal transduction mechanisms, which needs to be further explored.
At present, the host cells mostly used to establish HSV-2 in vitro model are Vero, HeLa, and VK2/E6E7 et al. (Akhrameyeva et al., 2011; Davis et al., 1978; Yao and Rosenthal, 2011). Since the primary culture technology of human vaginal epithelial cells cannot meet the needs of in vitro research, it is a better choice using VK2/E6E7 as host cells to study the mechanism of HSV-2 infection.
As one of the main component of the monarch drug of JZ-1, berberine is also one of the leading chemical constituents in JZ-1 detected by HPLC. Using alone of berberine in our experiment showed no noticeable anti-HSV-2 effect whether in the early or late stages. But the antiviral effect of berberine might be exerted by interacting with other components of JZ-1, which needs to be further explored by observing the interaction of metabolites that could interfere with viral cycle. The results of in vitro experiments showed that the efficacy of berberine was not the equivalent of JZ-1 compound preparation, which also reflected the scientificity of the combination of monarch, minister, assistant, and guide drugs in TCM prescription.
In conclusion, our study found that JZ-1 showed a definite effect in anti-HSV-2 adhesion and penetration of vaginal epithelial cells. The antiviral effect of JZ-1 was superior to that of chemical drug penciclovir while using alone of berberine, the main component of monarch drug of JZ-1, showed no such effect. Our results revealed the anti-HSV-2 effect and part of the mechanism of JZ-1, and also provided new ideas of TCM in the prevention and treatment of viral infectious diseases.
Authors ‘contribution
The head of this project is the corresponding author Zhuo Chen, who is in charge of the organization and implementation of the project. She designed this study and participated in the writing of manuscript. The HPLC profile of JZ-1 is completed by Tong Liu. Ping Yuan have proposed suggestions for the manuscript revision. Cong Huang and Qingqing Shao provided technical help to my experiment. Jun Sun carried out technical guidance for my experiment. Guangying Huang performed guidance on the subject. Qianni Duan is me, as the main executor for this study, I have contributed to the data acquisition, statistical analysis, manuscript writing and so on. All authors have proposed suggestions for the manuscript revision, they have read and approved the final version of the manuscript.
Acknowledgments
This research was supported by the grants from Natural Science Foundation of China (Nos. 81874483, 81473718, 81273787). We wish to acknowledge Professor Xiaokuan Li and Professor Hebin Tang, School of Pharmaceutical Science, South-central University for Nationalities, for their help in technical support for HPLC profile of JZ-1.
Abbreviation
- HSV-2
Herpes simplex virus type 2
- HIV
Human immunodeficiency virus
- HPV
Human papillomavirus
- TCID50
50% tissue culture infective dose
- MTT
3-{4, 5-dimethylthiazol-2-y}-2-5-diphenytetrazolium bromide
- DMSO
Dimethyl Sulphoxide
- DMEM
Dulbecco's Modified Eagles Medium
- CnT-PR
CellnTeC-Prime Medium
- FBS
Fetal bovine serum
- PBS
Phosphate buffer saline
- D-hanks’
Hank's balanced salt solution
- EDTA
Ethylene Diamine Tetraacetic Acid
- GH
Genital herpes
- gD
Glycoprotein D
- gB
Glycoprotein B
- VP16
Viral Protein 16
- VHS
Virion host shutoff protein
- ICP5
Earliest Virus-Specific Infected Cell Polypeptides5
- ICP4
Earliest Virus-Specific Infected Cell Polypeptides42
- HVEM
Herpesvirus Entry Mediator Ligand Protein
- TCM
Traditional Chinese Medicine
- JZ-1
Chinese Herbal Prescription JieZe-1
- HPLC
High-performance liquid chromatography
- SEM
Scanning electron microscopy
- TEM
Transmission electron microscopy
References
- Ai L., Chen Z. Huazhong University of Science and Technology; 2017. An Exploration on Part of the Mechanisms of Jie Ze No.1 Decoction against Herpes Simplex Virus Type 2 Infection in Vitro (硕士论文) [Google Scholar]
- Akhrameyeva N.V., Zhang P., Sugiyama N., Behar S.M., Yao F. Development of a glycoprotein D-expressing dominant-negative and replication-defective herpes simplex virus 2 (HSV-2) recombinant viral vaccine against HSV-2 infection in mice. J. Virol. 2011;85:5036–5047. doi: 10.1128/JVI.02548-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benzekri R., Bouslama L., Papetti A., Hammami M., Smaoui A., Limam F. Anti HSV-2 activity of Peganum harmala (L.) and isolation of the active compound. Microb. Pathog. 2018;114:291–298. doi: 10.1016/j.micpath.2017.12.017. [DOI] [PubMed] [Google Scholar]
- Bright H., Perez D.L., Christy C., Cockle P., Eyles J.E., Hammond D., Khodai T., Lang S., West K., Loudon P.T. The efficacy of HSV-2 vaccines based on gD and gB is enhanced by the addition of ICP27. Vaccine. 2012;30:7529–7535. doi: 10.1016/j.vaccine.2012.10.046. [DOI] [PubMed] [Google Scholar]
- Campisi A., Acquaviva R., Raciti G., Duro A., Rizzo M., Santagati A.N. Antioxidant activities of Solanum nigrum L. Leaf extracts determined in in vitro cellular models. Foods. 2019;8 doi: 10.3390/foods8020063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z., Gao W., Ma Y.-G., Lu F.-E., Huang G.-Y. Effect of jieze No. 1 gel on vaginal levels of lactobacillus , glycogen and lactoferrin in mice. In Chinese. Chin. J. Integr. Tradit. West. Med. 2009;29:722–724. [PubMed] [Google Scholar]
- Chen Z., Kong X.-F., Wang R., Ma Y.-G., Xu L.-J., Lu F.-E., Huang G.-Y. vol. 24. 2009. Investigation on prevention effects of Jieze NO.2 on candida albicans vaginitis; pp. 788–790. (Chinese. Matern. Child Health Care China). [Google Scholar]
- Chen Z., Wang R., Kong X.-F., Lu F.-E., Ma Y.-G., Huang G.-Y. vol. 24. 2009. The experimental study of Jieze No2 on trichomonas vaginitis; pp. 241–244. (Chinese. Matern. Child Health Care China). [Google Scholar]
- Chen Z., Wang R., Ma Y.-G., Kong X.-F., Lu F.-E., Huang G.-Y. vol. 24. 2009. The effect of Jieze NO.2 jel on trichomonas vaginitis in rats; pp. 644–646. (Chinese. Matern. Child Health Care China). [Google Scholar]
- Chen Z., Wang R., Ma Y.-G., Kong X.-F., Xiong C.-L. vol. 24. 2009. Spermicidal and antifertility effects of Jieze NO. 2 jel in rabbit; pp. 3855–3858. (Chinese. Matern. Child Health Care China). [Google Scholar]
- Churqui M.P., Lind L., Thörn K., Svensson A., Savolainen O., Aranda K.T., Eriksson K. Extracts of Equisetum giganteum L and Copaifera reticulate Ducke show strong antiviral activity against the sexually transmitted pathogen herpes simplex virus type 2. J. Ethnopharmacol. 2018;210:192–197. doi: 10.1016/j.jep.2017.08.010. [DOI] [PubMed] [Google Scholar]
- Costa X.J.D., Bourne N., Stanberry L.R., Knipe D.M. Construction and characterization of a replication-defective herpes simplex virus 2 ICP8 mutant strain and its use in immunization studies in a Guinea pig model of genital disease. Virology. 1997;232:1–12. doi: 10.1006/viro.1997.8564. [DOI] [PubMed] [Google Scholar]
- Cuéllar M.J., Giner R.M., Recio M.C., Máñez S., Ríos J.L. Topical anti-inflammatory activity of some Asian medicinal plants used in dermatological disorders. Fitoterapia. 2001;72:221–229. doi: 10.1016/S0367-326X(00)00305-1. [DOI] [PubMed] [Google Scholar]
- Dai W., Wu Y., Bi J., Wang S., Li F., Kong W., Barbier J., Cintrat J.-C., Gao F., Gillet D., Su W., Jiang C. Antiviral effects of ABMA against herpes simplex virus type 2 in vitro and in vivo. Viruses. 2018;10:119. doi: 10.3390/v10030119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis W.B., Oakes J.E., Taylor J.A. Effect of treatment with 5-ethyl-2’-deoxyuridine on herpes simplex virus encephalitis in normal and immunosuppressed mice. Antimicrob. Agents Chemother. 1978;14:743–748. doi: 10.1128/AAC.14.5.743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desai D.V., Kulkarni S.S. Herpes simplex virus: the interplay between HSV, host, and HIV-1. Viral Immunol. 2015;28:546–555. doi: 10.1089/vim.2015.0012. [DOI] [PubMed] [Google Scholar]
- Feng Y., Zhu Q. Guangzhou University of Chinese Medicine; 2015. Chinese Medicine Treatment of the Clinical Curative Effect Observation of Recurrent Genital Herpes. [Google Scholar]
- Fujimoto Y., Hikita S., Takeda K., Ozaki K., Inoue H., Takakuwa H., Sonoda K., Ono E. Evaluation of the antiviral potential of the soluble forms of glycoprotein D receptors on ocular herpes caused by HSV-1 and HSV-2 infections in a transgenic mouse model. J. Med. Virol. 2019;91:820–828. doi: 10.1002/jmv.25384. [DOI] [PubMed] [Google Scholar]
- Ge H. China Press of Traditional Chinese Medicine; 2016. A Handbook of Prescriptions for Emergencies. [Google Scholar]
- Ge H., Wang Y.-F., Xu J., Gu Q., Liu H.-B., Xiao P.-G., Zhou J., Liu Y., Yang Z., Su H. Anti-influenza agents from traditional Chinese medicine. Nat. Prod. Rep. 2010;27:1758–1780. doi: 10.1039/C0NP00005A. [DOI] [PubMed] [Google Scholar]
- González-Castejón M., Visioli F., Rodriguez-Casado A. Diverse biological activities of dandelion. Nutr. Rev. 2012;70:534–547. doi: 10.1111/j.1753-4887.2012.00509.x. [DOI] [PubMed] [Google Scholar]
- Halford W.P., Kemp C.D., Isler J.A., Davido D.J., Schaffer P.A. ICP0, ICP4, or VP16 expressed from adenovirus vectors induces reactivation of latent herpes simplex virus type 1 in primary cultures of latently infected trigeminal ganglion cells. J. Virol. 2001;75:6143–6153. doi: 10.1128/JVI.75.13.6143-6153.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong W.E.I., Zhuo C., Ping X.U., Yong-gui M.A., Li-jun X.U. Effect of jieze No.1(洁泽Ⅰ号)on cervicitis caused by ureaplasma urealyticum and on ureaplasma urealyticum in vitro. Chin. J. Integr. Med. 2008;14:88–93. doi: 10.1007/s11655-008-0088-2. [DOI] [PubMed] [Google Scholar]
- Hopkins J., Yadavalli T., Agelidis A.M., Shukla D. Host enzymes heparanase and cathepsin L promote herpes simplex virus 2 release from cells. J. Virol. 2018;92 doi: 10.1128/JVI.01179-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua S., Zhang Y., Liu J., Dong L., Huang J., Lin D., Fu X. Ethnomedicine, phytochemistry and pharmacology of Smilax glabra: an important traditional Chinese medicine. Am. J. Chin. Med. 2018;46:261–297. doi: 10.1142/S0192415X18500143. [DOI] [PubMed] [Google Scholar]
- Huang C., Chen Z. Huazhong University of Science and Technology; 2018. Study on the Mechanism of the Anti-HSV-2 Effect of Chinese Herbal Prescription Jieze-1 Based on Natural Immune Toll like Receptor Signaling Pathway (博士论文) [Google Scholar]
- Ibáñez F.J., Farías M.A., Gonzalez-Troncoso M.P., Corrales N., Duarte L.F., Retamal-Díaz A., González P.A. Experimental dissection of the lytic replication cycles of herpes simplex viruses in vitro. Front. Microbiol. 2018;9:2406. doi: 10.3389/fmicb.2018.02406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang J.J., Xie Y.M., Wang Y.Y., Yuan L.I., Zhang Y.L., Sun S.L., Wang S., Zhu Y., Wen-Sheng Q.I., Chen X. Study of Jinye Baidu granule in treatment of wind-warmth lung heat disease (heat in lung-wei pattern) : a randomized,double-blind,parallel-controlled trial. China J. Chin. Mater. Med. 2017:1467–1473. doi: 10.19540/j.cnki.cjcmm.2017.0043. [DOI] [PubMed] [Google Scholar]
- Jing W. Literature studying on clinical application of Yihuang decoction. In Chinese. Guangming J. Chin. Med. 2012 [Google Scholar]
- Kato K., Daikoku T., Goshima F., Kume H., Yamaki K., Nishiyama Y. Synthesis, subcellular localization and VP16 interaction of the herpes simplex virus type 2 UL46 gene product. Arch. Virol. 2000;145:2149–2162. doi: 10.1007/s007050070045. [DOI] [PubMed] [Google Scholar]
- Kesharwani A., Polachira S.K., Nair R., Agarwal A., Mishra N.N., Gupta S.K. Anti-HSV-2 activity of Terminalia chebula Retz extract and its constituents, chebulagic and chebulinic acids. BMC Complement Altern. Med. 2017;17 doi: 10.1186/s12906-017-1620-8. 110–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LI S., Xu J., Jin S.-F. 2017. Effect of Yihuang decoction combined with chitosan membrane in the treatment of cervical HPV infection; pp. 3454–3457. (Chinese. Guangming J. Chin. Med.). [Google Scholar]
- Li W., Wang X.-H., Luo Z., Liu L.-F., Yan C., Yan C.-Y., Chen G.-D., Gao H., Duan W.-J., Kurihara H., Li Y.-F., He R.-R. Traditional Chinese medicine as a potential source for HSV-1 therapy by acting on virus or the susceptibility of host. Int. J. Mol. Sci. 2018;19 doi: 10.3390/ijms19103266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin C.-W., Tsai F.-J., Tsai C.-H., Lai C.-C., Wan L., Ho T.-Y., Hsieh C.-C., Chao P.-D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005;68:36–42. doi: 10.1016/j.antiviral.2005.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling Y., Zheng J. Research progress of a Chinese traditional drug taraxacum herb, in Chinese. Chin. J. Mod. Appl. Pharm. 1998;15:10–13. [Google Scholar]
- Liu Y., Guan X., Li C., Ni F., Luo S., Wang J., Zhang D., Zhang M., Hu Q. HSV-2 glycoprotein J promotes viral protein expression and virus spread. Virology. 2018;525:83–95. doi: 10.1016/j.virol.2018.09.004. [DOI] [PubMed] [Google Scholar]
- Liu Y., Li M., Zhang D., Zhang M., Hu Q. HSV-2 glycoprotein gD targets the CC domain of tetherin and promotes tetherin degradation via lysosomal pathway. Virol. J. 2016;13:154. doi: 10.1186/s12985-016-0610-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y., Li M., Zhang D., Zhang M., Hu Q. HSV-2 glycoprotein gD targets the CC domain of tetherin and promotes tetherin degradation via lysosomal pathway. Virol. J. 2016;13 doi: 10.1186/s12985-016-0610-7. 154–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Z. Vaginal discharge. Chin. Med. Mod. Distance Educ. China. 2010 [Google Scholar]
- Luo S., Hu K., He S., Wang P., Zhang M., Huang X., Du T., Zheng C., Liu Y., Hu Q. Contribution of N-linked glycans on HSV-2 gB to cell–cell fusion and viral entry. Virology. 2015;483:72–82. doi: 10.1016/j.virol.2015.04.005. [DOI] [PubMed] [Google Scholar]
- Luo S., Hu K., He S., Wang P., Zhang M., Huang X., Du T., Zheng C., Liu Y., Hu Q. Contribution of N-linked glycans on HSV-2 gB to cell–cell fusion and viral entry. Virology. 2015;483:72–82. doi: 10.1016/j.virol.2015.04.005. [DOI] [PubMed] [Google Scholar]
- Lv M., Xu P., Tian Y., Liang J., Gao Y., Xu F., Zhang Z., Sun J. Medicinal uses, phytochemistry and pharmacology of the genus Dictamnus (Rutaceae) J. Ethnopharmacol. 2015;171:247–263. doi: 10.1016/j.jep.2015.05.053. [DOI] [PubMed] [Google Scholar]
- Ma Y.-G., Xie W., Fang J.-G., Chen Z. vol. 30. 2011. Determination of berberine hydrochloride in jieze lotion by HPLC; pp. 1484–1486. (Chinese. Her. Med.). [Google Scholar]
- Mohanta T.K., Zubaidha Y.T.P.K. Phytochemical and medicinal importance of Ginkgo biloba L. Nat. Prod. Res. 2014;28:746–752. doi: 10.1080/14786419.2013.879303. [DOI] [PubMed] [Google Scholar]
- Mohraz M., Aghakhani A., Moayedi-Nia S., Banifazl M., Janbakhsh A., Mamishi S., Karami A., Bavand A., Mirzapour P., Ramezani A. No role of herpes simplex virus type 2 (HSV-2) infection on HIV progression in naïve HIV patients. Iran. Biomed. J. 2018;22:123–128. doi: 10.22034/ibj.22.2.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mousavi E., Makvandi M., Teimoori A., Ataei A., Ghafari S., Samarbaf-Zadeh A. Antiviral effects of Lactobacillus crispatus against HSV-2 in mammalian cell lines. J. Chin. Med. Assoc. 2017;81 doi: 10.1016/j.jcma.2017.07.010. S1726490117302551. [DOI] [PubMed] [Google Scholar]
- Mukherjee H., Ojha D., Bag P., Chandel H.S., Bhattacharyya S., Chatterjee T.K., Mukherjee P.K., Chakraborti S., Chattopadhyay D. Anti-herpes virus activities of Achyranthes aspera: an Indian ethnomedicine, and its triterpene acid. Microbiol. Res. 2013;168:238–244. doi: 10.1016/j.micres.2012.11.002. [DOI] [PubMed] [Google Scholar]
- Nikolic D.S., Piguet V. Vaccines and microbicides preventing HIV-1, HSV-2, and HPV mucosal transmission. J. Investig. Dermatol. 2010;130:352–361. doi: 10.1038/jid.2009.227. [DOI] [PubMed] [Google Scholar]
- Nozawa N., Yamauchi Y., Ohtsuka K., Kawaguchi Y., Nishiyama Y. Formation of aggresome-like structures in herpes simplex virus type 2-infected cells and a potential role in virus assembly. Exp. Cell Res. 2004;299:486–497. doi: 10.1016/j.yexcr.2004.06.010. [DOI] [PubMed] [Google Scholar]
- Piazza S., Pacchetti B., Fumagalli M., Bonacina F., Dell'Agli M., Sangiovanni E. Comparison of two Ginkgo biloba L. Extracts on oxidative stress and inflammation markers in human endothelial cells. Mediat. Inflamm. 2019;2019:12. doi: 10.1155/2019/6173893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ping X.U., Zhuo C., Lijun X.U., Fuer L.U. Spermicidal effect of jieze No. 1 in combination with nonoxynol-9 in vitro. J. Huazhong Univ. Sci. Technol. Sci. 2005;25:225–228. doi: 10.1007/BF02873584. [DOI] [PubMed] [Google Scholar]
- Qian C.C. [Distinguishing some falsifications in fuqingzhunuke (Fu Qingzhu's obstetrics and gynecology)] Zhonghua Yi Shi Za Zhi. 2009;39:232. [PubMed] [Google Scholar]
- Sampath P., Deluca N.A. Binding of ICP4, TATA-binding protein, and RNA polymerase II to herpes simplex virus type 1 immediate-early, early, and late promoters in virus-infected cells. J. Virol. 2008;82:2339–2349. doi: 10.1128/JVI.02459-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiffer J.T., Gottlieb S.L. Biologic interactions between HSV-2 and HIV-1 and possible implications for HSV vaccine development. Vaccine. 2017 doi: 10.1016/j.vaccine.2017.09.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sepúlveda-Crespo D., Ceña-Díez R., Jiménez J.L., Ángeles Muñoz-Fernández M. Mechanistic studies of viral entry: an overview of dendrimer-based microbicides as entry inhibitors against both HIV and HSV-2 overlapped infections. Med. Res. Rev. 2017;37:149–179. doi: 10.1002/med.21405. [DOI] [PubMed] [Google Scholar]
- Smith C.A., Schaffer P.A. Mutants defective in herpes simplex virus type 2 ICP4: isolation and preliminary characterization. J. Virol. 1987;61:1092–1097. doi: 10.1128/jvi.61.4.1092-1097.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su A., Qiu M., Li Y., Xu W., Song S., Wang X., Song H., Zheng N., Wu Z. BX-795 inhibits HSV-1 and HSV-2 replication by blocking the JNK/p38 pathways without interfering with PDK1 activity in host cells. Acta Pharmacol. Sin. 2017;38:402. doi: 10.1038/aps.2016.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y., Lenon G.B., Yang A.W.H. Phellodendri Cortex: a phytochemical, pharmacological, and pharmacokinetic review. Evid. Based complement. Altern. Med. 2019;2019:45. doi: 10.1155/2019/7621929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao P. Strategies for antiviral screening targeting early steps of virus infection. Virol. Sin. 2010;25:281–293. doi: 10.1007/s12250-010-3135-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vilhelmova-Ilieva N., Jacquet R., Quideau S., Galabov A.S. Ellagitannins as synergists of ACV on the replication of ACV-resistant strains of HSV 1 and 2. Antivir. Res. 2014;110:104–114. doi: 10.1016/j.antiviral.2014.07.017. [DOI] [PubMed] [Google Scholar]
- Wang M., Yang X., Zhao J., Lu C., Zhu W. Structural characterization and macrophage immunomodulatory activity of a novel polysaccharide from Smilax glabra Roxb. Carbohydr. Polym. 2017;156:390–402. doi: 10.1016/j.carbpol.2016.09.033. [DOI] [PubMed] [Google Scholar]
- WU H., DI S., DAI D., TONG X. Exploration about the clinical application and dosage of Paeonia suffruticosa Andr. In Chinese. Jilin J. Chin. Med. 2019:726–729. [Google Scholar]
- Xin J., Tan T.-H. Clinical observation on treating female mycoplasma infection with the Yihuang decoction plus doxycycline. In Chinese. Clin. J. Chin. Med. 2016;8:112–113. [Google Scholar]
- Xiong H.-R., Luo J., Hou W., Xiao H., Yang Z.-Q. The effect of emodin, an anthraquinone derivative extracted from the roots of Rheum tanguticum, against herpes simplex virus in vitro and in vivo. J. Ethnopharmacol. 2011;133:718–723. doi: 10.1016/j.jep.2010.10.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao X.-D., Rosenthal K.L. Herpes simplex virus type 2 virion host shutoff protein suppresses innate dsRNA antiviral pathways in human vaginal epithelial cells. J. Gen. Virol. 2011;92:1981–1993. doi: 10.1099/vir.0.030296-0. [DOI] [PubMed] [Google Scholar]
- Yildirim A., Duran G.G., Duran N., Jenedi K., Bolgul B.S., Miraloglu M., Muz M. Antiviral activity of hatay propolis against replication of herpes simplex virus type 1 and type 2. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2016;22:422–430. doi: 10.12659/MSM.897282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan Q., Zhuo C., Yonggui M., Fuer L., Suhua C., Guangying H. Nonoxynol-9 berberine plural gel has little effect on expression of SLPI, SP-D and lactoferrin in mice's vagina. Iran. J. Reprod. Med. 2013;11:565–576. [PMC free article] [PubMed] [Google Scholar]
- Zhang M., Deng X., Guan X., Geng L., Fu M., Zhang B., Chen R., Hu H., Hu K., Zhang D., Li M., Liu Y., Gong S., Hu Q. Herpes simplex virus type 2 infection-induced expression of CXCR3 ligands promotes CD4+ T cell migration and is regulated by the viral immediate-early protein ICP4. Front. Immunol. 2018;9:2932. doi: 10.3389/fimmu.2018.02932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang N., Li H. Guangzhou University of Chinese Medicine; 2012. The Research on Literature and Order of Prescriptios and Herbs, Syndromes and Treatment of Genital Herpes. [Google Scholar]
- Zhang R., Wang Y., Song B., Han Z.Q., Xu Y.M. Recombinant cell lines expressing shRNA targeting herpes simplex virus 2 VP16 inhibit virus replication. Intervirology. 2012;55:426–434. doi: 10.1159/000335663. [DOI] [PubMed] [Google Scholar]
- Zhang S., Zhang W. Characteristics of development of obstetrical thought in traditional Chinese medicine as viewed from Fu Qing Zhu Nu Ke, a book on obstetrics and gynecology. Zhonghua Yi Shi Za Zhi. 1987;17:149. (Chi) [PubMed] [Google Scholar]
- Zhu J.Q., Che Y.X., Center I.M. Mining analysis on composition and medication of menstruation prescriptions in Fu Qingzhu's Obstetrics and Gynecology. In Chinese. China J. Chin. Mater. Med. 2015;40:1610–1613. [PubMed] [Google Scholar]
- Zou W., Wen X., Zheng Y., Xiao Z., Luo J., Chen S., Wang Y., Cheng Z., Xiang D., Nie Y. Metabolomic study on the preventive effect of patrinia scabiosaefolia fisch on multipathogen induced pelvic inflammatory disease in rats. Evid. Based Complement. Alternat. Med. 2015;2015:9. doi: 10.1155/2015/170792. [DOI] [PMC free article] [PubMed] [Google Scholar]