Abstract
Transmissible gastroenteritis virus (TGEV) is the causative agent of porcine transmissible gastroenteritis (TGE), characterized by high mortality and severely retarded growth in piglets that dramatically affects the porcine industry. Previously, we have identified two shRNA-expressing plasmids pEGFP-U6/P1 and pEGFP-U6/P2 that target RNA-dependent RNA polymerase (RdRP) gene of TGEV with more than 95% of virus inhibition in vitro. In this study, inhibition of the TGEV replication by pEGFP-U6/P1 and pEGFP-U6/P2 was tested in mini-pigs. SPF mini-pigs at 25 days old were injected with the shRNA-expressing plasmids and then infected with TGEV. The results from the analyses of clinical signs, histopathology, indirect immunofluorescence (IIF) and RT-PCR show that the two shRNA-expressing plasmids could significantly decrease the quantity of TGEV in different organs and protect mini-pigs from TGEV infection. These findings illustrate the prospect for TGEV-specific shRNAs to be new anti-TGEV agents.
Keywords: TGEV, shRNA, Mini-pig
1. Introduction
Since RNA interference (RNAi) was confirmed as an ancient and invaluable tool for silencing genes in the animal and plant (Fire et al., 1998, Hamilton and Baulcombe, 1999, Hannon, 2002), it has been extensively used to study interference with virus replication (McCaffrey et al., 2003, Vaucheret et al., 2001). So far, the replication of various viruses including many coronaviruses has been effectively inhibited in vitro or in vivo (Chang et al., 2007, Dieckhoff et al., 2008, Jia et al., 2008, Li et al., 2005, Liu et al., 2006), however, no report yet have shown that the replication of coronaviruses in swine could be interfered by RNAi.
Porcine transmissible gastroenteritis virus (TGEV), a member of the family Coronaviridae, is the etiological agent of porcine transmissible gastroenteritis (TGE). Swine of all ages and species were susceptible to TGEV, and infected animals usually show symptoms of vomiting, diarrhea and severe dehydration. As the mortality of infected piglets under 3 weeks can be up to 100% (Garwes, 1988, Mullan et al., 1994) and survived elder pigs usually show growth retardation and low rate of reward to feeding, the annual economic loss due to outbreak of TGE was enormous around the world. Furthermore, TGE usually developed very fast in vivo. Neonatal piglets infected with TGEV may suffer from gastroenteritis within 20 h post-infection and death may happen in 1–4 days (Schwegmann-Wessels and Herrler, 2006). However, unlike the foot-and-mouth disease virus (FMDV), there are no quick and potent anti-TGEV therapeutic agents available yet (Brim et al., 1995, de los Santos et al., 2005, Saif, 2004). Previously, two shRNA-expressing plasmids pEGFP-U6/P1 and pEGFP-U6/P2, targeting the RNA-dependent RNA polymerase (RdRP) gene of TGEV, had been constructed and found to be with high efficiency of viral inhibition as demonstrated in vitro (Zhou et al., 2007). In the present study, we delivered the two plasmids into mini-pigs to further evaluate their antiviral potential in vivo.
2. Materials and methods
2.1. Selection of anti-TGEV shRNA-expressing plasmids
Here we selected two TGEV-specific shRNA-expressing plasmids pEGFP-U6/P1 and pEGFP-U6/P2 based on a previous in vitro study (Zhou et al., 2007). Meanwhile, we used the shRNA-expressing plasmid pEGFP-U6/T containing an unrelated siRNA duplex as a non-specific toxicity control. Inserted shRNA sequences of the plasmids above were designed as follows: pEGFP-U6/P1: 5′-GTACTGGGATCGCACATATTTCAAGACGATATGTGCGATCCCAGTACTTTTTT-3′; pEGFP-U6/P2: 5′-GGCAAGAGCTCGTACAGTATTCAAGACGTACTGTACGAGCTCTTGCCTTTTTT-3′; pEGFP-U6/T: 5′-G ACTTCATAAGGCGCATGCTTCAAGACGGCATGCGCCTTATGAAGTCTTTTTT-3′. Just like the method in vitro (Zhou et al., 2007), an enhanced green fluorescent protein (EGFP) gene, used to visualize the major location and expression of introduced plasmids, was still inserted in each.
2.2. Plasmids delivery and TGEV infection
The TGEV, named as TGEs-1, isolated from a diarrhea piglet in 2000 was a gift from Mr. Yongqing Qian (Shanghai Academy of Agricultural Sciences). The TGEs-1 virus was propagated in swine testicle (ST) cells as described previously (Zhou et al., 2007). Twelve full-sib SPF mini-pigs at 25 days old (1.9–2.2 kg) raised in SPF animal house in our institute were divided randomly into 6 groups as shown in Table 1 , in which the groups 1, 2, 3, 4 and 5 were served as non-infection, viral infection alone, plasmid toxicity control, pEGFP-U6/P1 interference and pEGFP-U6/P2 interference group, respectively. For the group 6, a plasmid mixture (pEGFP-U6/P1 + P2) consisting of an equal amount of pEGFP-U6/P1 and pEGFP-U6/P2 was injected. All the shRNA-expressing plasmids were diluted with 0.1 M PBS (pH 7.4) and inoculated into mini-pigs via the precava (superior vena cava) after optimizing the dose of injection plasmids by respectively introducing 1, 2, 3 and 4 mg/10 ml/pig of pEGFP-U6/P1. 24 h after injection, all mini-pigs except those in groups 1 and 3, were infected with 10 ml TGEs-1-infected cell culture (104 CCID50) orally. The experiment was performed twice, and data from each were pooled for analyses.
Table 1.
Plasmids delivery and virus infection regimen.
| Group |
||||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| Plasmid | PBS | PBS | pEGFP-U6/T | pEGFP-U6/P1 | pEGFP-U6/P2 | pEGFP-U6/P1 + P2 |
| Vector dosage | 10 ml/pig | 10 ml/pig | 3 mg/10 ml/pig | 3 mg/10 ml/pig | 3 mg/10 ml/pig | 3 mg/10 ml/pig |
| TGEV | PBS | TGEs-1 | PBS | TGEs-1 | TGEs-1 | TGEs-1 |
| Viral dosage | 10 ml/pig | 10 ml/pig | 10 ml/pig | 10 ml/pig | 10 ml/pig | 10 ml/pig |
2.3. Main clinical signs, histopathology and IIF analyses
After TGEV infection, we monitored daily the clinical signs of mini-pigs, including appetite, body weight and defecation. All mini-pigs were slaughtered by euthanasia at 3 d.p.i. Major organs, including liver, lung, kidney, spleen, jejunum, ileum and mesenteric lymph node were collected aseptically for tissue sections and RT-PCR analysis. Paraffin-embedded tissue sections were prepared for histopathology analysis, while frozen tissue sections were used for EGFP gene expression and indirect immunofluorescence (IIF) analyses, during which swine anti-TGEV polyclonal antibodies and goat anti-swine IgG conjugated by Texas Red (Santa Cruz) were used to visualize TGEV-infected cells.
2.4. RT-PCR analysis
Total RNAs in each tissue sample were extracted and purified using an RNAultra kit (Tiangen) according to the manufacturer's instructions. The first strand cDNA was synthesized and amplified using a RT-PCR Kit AMV 3.0 (TaKaRa) according to the manufacturer's protocol. Primers for TGEV N gene and swine β-actin gene were designed as followed: 5′-GAGCAGTGCCAAGCATTACCC-3′ (N forward), 5′-GACTTCTATCTGGTCGCCATCTTC-3′ (N reverse), 5′-CGTCCACCGCAAATGCTTC-3′ (β-actin forward) and 5′-AACCGACTGCTGTCACCTTCAC-3′ (β-actin reverse). The product amplified from TGEV N gene is 109 bp in length and that from swine β-actin gene (a loading control) is 217 bp.
3. Results
3.1. shRNA-expressing plasmids were transcribed in mini-pigs
Based on in vitro study, both pEGFP-U6/P1 and pEGFP-U6/P2 targeting RdRP gene of TGEV had over 95% inhibition on viral replication, therefore they were further studied in vivo by injection into mini-pigs. Location and transcription activity of the plasmids in mini-pigs were evaluated by the expression of EGFP in different tissues under a fluorescence microscope (Olympus). After 1, 2, 3 and 4 mg/10 ml/pig of pEGFP-U6/P1 were respectively injected into four SPF mini-pigs, the EGFP was mainly found in spleen, mesenteric lymph node, jejunum and ileum (data not shown). Especially, in the jejunum and ileum, the EGFP was abundant when injection of 3 or 4 mg/10 ml/pig of pEGFP-U6/P1 was performed. Thus, 3 mg/10 ml/pig of plasmids were selected as the dose used in this study.
3.2. shRNA-expressing plasmids suppressed symptoms of TGE and protected organs from TGEV destruction in mini-pigs
To investigate whether TGEV-specific shRNAs could protect mini-pigs from TGEV infection, all the mini-pigs were treated as the regimen shown in Table 1, and the clinical symptoms and histopathological changes were analyzed. Mini-pigs in all of the groups were healthy with a good appetite after the injection of plasmids or PBS. 24 h later, all the mini-pigs except those in group 1 and group 3 were infected with TGEs-1 orally. 26 h after infection, the four mini-pigs in group 2 began to show typical clinical symptoms of TGE: flabbiness and thinness, followed by diarrhea and dehydration (3 of 4 mini-pigs). The situation lasted till they were euthanized 3 d.p.i., while all of those in the other groups were in good condition without any TGE-like symptoms.
According to histopathological analysis, three of the organ samples including the lung, spleen and mesenteric lymph node from groups 2 to 6 did not show obvious differences from those in group 1 (data not shown), however, the other organs such as the liver, kidney and small intestine in treated groups, as indicated in Fig. 1 , showed pathological changes at different degree after transfection and/or infection, especially those in group 2 were seriously damaged. The representative pathological changes included liver swelling, granular degeneration of hepatocytes (Fig. 1A and E), granular degeneration of renal tubular epithelial cells, glomerular and/or renal tubular swelling in kidney (Fig. 1B and F), intestinal villi hyperemia, atrophy and destruction (Fig. 1C and G), and lymphocytes proliferation in the villi and lamina propria (Fig. 1D and H). We classified these pathological changes from mild to severe with the indication of (i) “−”, (ii) “±”, (iii) “+”, (iv) “++”, (v) “+++” and (vi) “++++” as shown in Table 2 . Based on careful analysis of different organs, histopathological changes in groups 3–6 are usually milder than which to the same organs in group 2 (Table 2 and Fig. 1), especially to the intestinal villi (Fig. 1C and G), which was the major targeting organ of TGEV. Taken together, the clinical symptom and histopathology analyses indicated that pEGFP-U6/P1, pEGFP-U6/P2 as well as their mixture could protect mini-pig organs from TGEV destruction. However, the synergy demonstrated by a mixture of pEGFP-U6/P1 and pEGFP-U6/P2 in this study was screened by the effective inhibition showed by pEGFP-U6/P1 and pEGFP-U6/P2 alone, and further investigation was required in the next optimization test.
Fig. 1.
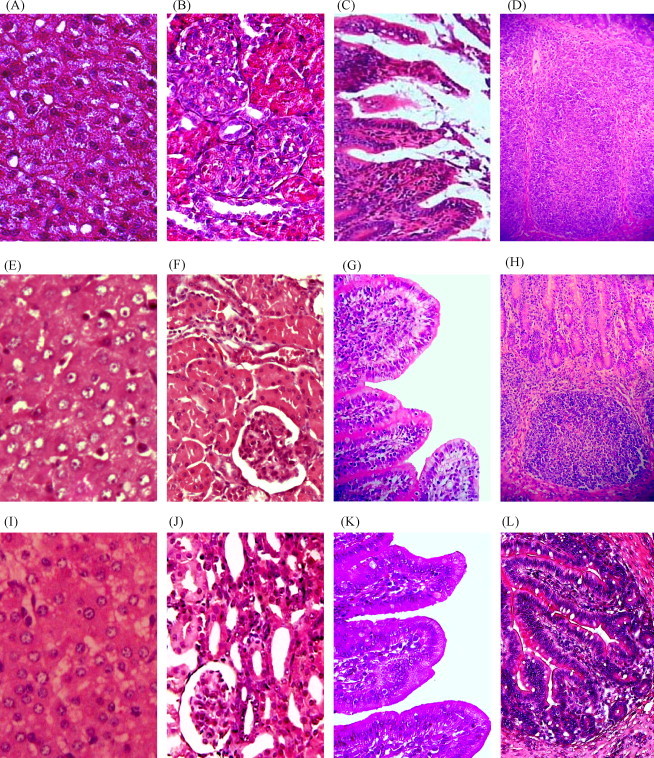
shRNA-expressing plasmids protect mini-pig organs from TGEV destruction. All histology sections were stained with H&E. Panels A to D show tissue sections from mini-pigs in group 2, panels E to H show tissue sections from mini-pigs in group 5, and panels I to L show tissue sections from mini-pigs in group 1. (A) Liver with severe swelling and granular degeneration of hepatocytes (200×). (B) Kidney with serious granular degeneration of renal tubular epithelial cells, severe glomerular and renal tubular swelling (200×). (C) Jejunum with villi severe atrophied and destructed (100×). (D) Ileum with severe lymphocytes proliferation in the lamina propria (100×). (E) Liver with low-grade swelling and granular degeneration of hepatocytes, corresponding to the “+” in Table 2 (200×). (F) Kidney with normal glomeruli but low-grade renal tubular swelling, corresponding to the “+” in Table 2 (200×). (G) Jejunum with intact villi but low-grade hyperemia and lymphocytes proliferation, corresponding to the “+” in Table 2 (100×). (H) Ileum with lymphocytes proliferation in the lamina propria, corresponding to the “++” in Table 2 (100×). (I) Liver with hepatocytes in the normal state (200×). (J) Kidney with glomeruli and tubules in the normal state (200×). (K) Jejunum with villi in the normal state (100×). (L) Ileum with lamina propria in the normal state (100×).
Table 2.
The quantity of mini-pigs with organ damage after different treatment and a score of damage degree of mini-pig organs in each group.
| Organ damage | Small intestine |
Liver |
Kidney |
|||||
|---|---|---|---|---|---|---|---|---|
| Villus |
Lamina propria |
Pig quantity | Damage degree | Pig quantity | Damage degree | |||
| Pig quantity | Damage degree | Pig quantity | Damage degree | |||||
| Group 1 | 4 | − | 4 | − | 1/3 | ±/− | 4 | − |
| Group 2 | 3/1 | ++++/+++ | 3/1 | ++++/+++ | 2/2 | +++/++ | 3/1 | ++++/+++ |
| Group 3 | 2/2 | ±/− | 1/2/1 | ++/+/− | 1/2/1 | +/±/− | 2/2 | +/− |
| Group 4 | 2/2 | ±/− | 1/3 | ++/+ | 1/2/1 | +/±/− | 1/1/1/1 | ++/+/±/− |
| Group 5 | 1/3 | +/− | 2/1/1 | ++/+/± | 1/1/2 | +/±/− | 1/1/2 | +/±/− |
| Group 6 | 1/1/2 | +/±/− | 1/2/1 | ++/+/− | 1/1/1/1 | ++/+/±/− | 1/1/2 | +/±/− |
Pig quantity: the quantity of mini-pigs with organ damage after treatment; damage degree: the organ damage degree in each mini-pig. Each number in the “pig quantity” was in accordance to the score in “damage degree” under the same organ in the same order.
3.3. shRNA-expressing plasmids inhibited TGEV replication in vivo
To understand the distribution pattern of TGEV in different organs and the effect of TGEV-specific shRNA on viral replication, IIF was performed on frozen tissue sections and viral RNA was analyzed by RT-PCR. TGEV was observed widely in mini-pig mesenteric lymph node, liver, jejunum, ileum and kidney in group 2 (Fig. 2F–J). Especially, in the mesenteric lymph node, jejunum and ileum, large amount of viral antigens were detected. In contrast, no TGEV antigen was found in the organs of mini-pigs from the other groups (Fig. 2A–E). In addition, RT-PCR analysis showed that all the organs from mini-pigs in groups 4–6 were negative for the TGEV N gene (Fig. 3A), while those in group 2 were positive (Fig. 3B). In conclusion, the analyses of IIF and RT-PCR indicated that both shRNA-expressing plasmids pEGFP-U6/P1 and pEGFP-U6/P2 could remarkably inhibit TGEV replication in mini-pigs.
Fig. 2.
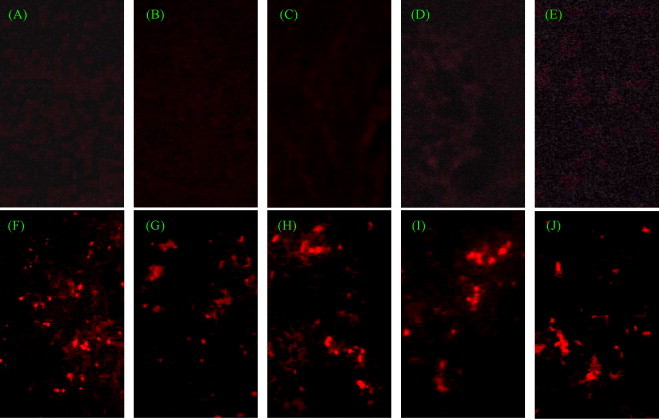
shRNA-expressing plasmids significantly reduced TGEV antigen in different organs. All sample frozen tissue sections were stained with Texas Red (original magnification, 400×). Mesenteric lymph node (A), liver (B), jejunum (C), ileum (D) and kidney (E) were sampled from the mini-pig in group 4. Red spots indicated the TGEV-infected cells. Mesenteric lymph node (F), liver (G), jejunum (H), ileum (I) and kidney (J) were sampled from the mini-pigs in group 2, served as the viral infection controls.
Fig. 3.
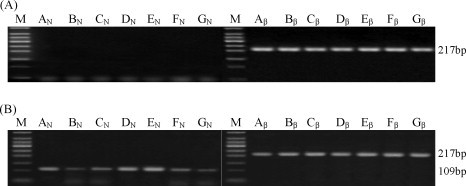
shRNA-expressing plasmids remarkably inhibited TGEV RNA replication in different organs. (A) All the organs were sampled from the mini-pigs in group 5; (B) all the organs were sampled from the mini-pigs in group 2; M: 50 bp DNA ladder marker; A: mesenteric lymph node; B: lung; C: liver; D: jejunum; E: ileum; F: kidney; G: spleen. Aβ, Bβ, Cβ, Dβ, Eβ, Fβ, Gβ are products of β-actin gene expressed in these organs; AN, BN, CN, DN, EN, FN, GN are products of the N gene of TGEV in these organs. The TGEV N gene fragments, amplified successfully in all samples in group 2, were undetectable in group 5.
4. Discussion
RdRP is an essential protein encoded by RNA viruses, and is conserved in TGEV genome. We had demonstrated that two TGEV-specific plasmids pEGFP-U6/P1 and pEGFP-U6/P2, both targeting the RdRP gene, could effectively down-regulate TGEV replication in vitro. In this study, we further confirmed a potential effect of the two plasmids on TGEV replication in vivo.
As a coronavirus, TGEV has a restricted host range and tissue tropism. The IIF and RT-PCR analyses (Fig. 2, Fig. 3) indicated that TGEV can replicate in several organs in pigs, but they mainly distribute in swine small intestine, for the amino peptidase N (APN), a TGEV major receptor in pigs, is abundantly expressed in the apical membrane of the small intestine epithelial cells, and which has a bigger quantity and higher activity in small intestine than other organs. Through the APN most of infectious TGEV invade host small intestine epithelial cells and initiate viral replication (Delmas et al., 1992, Rossen et al., 1994, Weingartl and Derbyshire, 1994). In our study, after the plasmids injected into mini-pigs via the precava, the reporter gene EGFP expressed abundantly in small intestine cells (data not shown). It indicated that the plasmids were efficiently delivered into small intestine cells, the target of TGEV, by the method of precava injection. This may be a major reason why a small dose of shRNA-expressing plasmids (3 mg/piglet) could well protect mini-pigs from clinical symptoms and histopathology induced by TGEV infection (Delmas et al., 1992, Rossen et al., 1994, Weingartl and Derbyshire, 1995). The data also suggested that precava injection may be a good choice for treating animals with intestinal diseases as this route uses relatively short distance to reach small intestine from the precava.
Based on RT-PCR analysis, TGEV propagation in all samples, including the lung, were effectively inhibited by transfected specific plasmids, but there was no obvious green fluorescence or red immunofluorescence in which compared to that in jejunum or ileum. Based on our observations, the background fluorescence in the lung was more abundant than that in the other samples or pure cultured cells (data not shown). Moreover, plasmid expression in lung was poorer than that in intestine (see Section 2 in discussion). So, we concluded that the high background fluorescence in lung hampered the detection of fluorescence emitted by the plasmids. Fortunately, TGEV propagation was also less efficient in lung (the reason discussed in Section 2 above) than that in small intestine (Fig. 3B). Therefore, though the expression of shRNA in lung was not highly active, it was enough to inhibit the same poor propagation of TGEV in lung in the present study.
Some gene therapists found that RNAi not only could effectively inhibit hepatitis B virus replication in mice, but also make the mice suffer from liver toxicity (Couzin, 2006, Grimm and Kay, 2007). Based on these, a plasmid toxicity control was also considered in the present study. After the analysis of histopathology, some damages in the liver, kidney and lymphocytes proliferation in intestinal lamina propria were also found in some of the mini-pigs injected with plasmids and/or TGEV, indicating that plasmids would induce immune responses in mini-pigs, too. Therefore, changing the pattern of siRNA or shRNA-expressing vector would be considered in the future (Couzin, 2006, McBride et al., 2008). Taken together, our data shows a tremendous potential for shRNA-expressing vectors to enable rapid and effective treatments for TGEV and other viruses with intestinal tropism in mini-pigs.
Contributor Information
Xiuguo Hua, Email: hxg@sjtu.edu.cn.
Zhibiao Yang, Email: zbyang@sjtu.edu.cn.
References
- Brim T.A., VanCott J.L., Lunney J.K., Saif L.J. Cellular immune responses of pigs after primary inoculation with porcine respiratory coronavirus or transmissible gastroenteritis virus and challenge with transmissible gastroenteritis virus. Vet. Immunol. Immunopathol. 1995;48:35–54. doi: 10.1016/0165-2427(94)05416-P. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang Z., Babiuk L.A., Hu J. Therapeutic and prophylactic potential of small interfering RNAs against severe acute respiratory syndrome: progress to date. BioDrugs. 2007;21:9–15. doi: 10.2165/00063030-200721010-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couzin J. RNAi safety comes under security. Science. 2006;312:1121. doi: 10.1126/science.312.5777.1121a. [DOI] [PubMed] [Google Scholar]
- Delmas B., Gelfi J., L’Haridon R., Vogel L.K., Sjöström H., Norén O., Laude H. Aminopeptidase N is a major receptor for the entero-pathogenic coronavirus TGEV. Nature. 1992;357:417–420. doi: 10.1038/357417a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de los Santos T., Wu Q., deAvila Botton S., Grubman M.J. Short hairpin RNA targeted to the highly conserved 2B nonstructural protein coding region inhibits replication of multiple serotypes of foot-and-mouth disease virus. Virology. 2005;335:222–231. doi: 10.1016/j.virol.2005.03.001. [DOI] [PubMed] [Google Scholar]
- Dieckhoff B., Petersen B., Kues W.A., Kurth R., Niemann H., Denner J. Knockdown of porcine endogenous retrovirus (PERV) expression by PERV-specific shRNA in transgenic pigs. Xenotransplantation. 2008;15(1):36–45. doi: 10.1111/j.1399-3089.2008.00442.x. [DOI] [PubMed] [Google Scholar]
- Fire A., Xu S., Montgomery M.K., Kostas S.A., Driver S.E., Mello C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998;391:806–811. doi: 10.1038/35888. [DOI] [PubMed] [Google Scholar]
- Garwes D.J. Transmissible gastroenteritis. Vet. Res. 1988;122(19):462–463. doi: 10.1136/vr.122.19.462. [DOI] [PubMed] [Google Scholar]
- Grimm D., Kay M.A. Therapeutic application of RNAi: is mRNA targeting finally ready for prime time? J. Clin. Invest. 2007;117:3633–3641. doi: 10.1172/JCI34129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton A.J., Baulcombe D.C. A species of small antisense RNA in posttranscriptional gene silencing in plants. Science. 1999;286:950–952. doi: 10.1126/science.286.5441.950. [DOI] [PubMed] [Google Scholar]
- Hannon G.J. RNA interference. Nature. 2002;418:244–251. doi: 10.1038/418244a. [DOI] [PubMed] [Google Scholar]
- Jia H., Ge X., Guo X., Yang H., Yu K., Chen Z., Chen Y., Cha Z. Specific small interfering RNAs-mediated inhibition of replication of porcine encephalomyocarditis virus in BHK-21 cells. Antiviral Res. 2008;79(2):95–104. doi: 10.1016/j.antiviral.2007.12.003. [DOI] [PubMed] [Google Scholar]
- Li B.J., Tang Q., Cheng D., Qin C., Xie F.Y., Wei Q., Xu J., Liu Y., Zheng B.J., Woodle M.C., Zhong N., Lu P.Y. Using siRNA in prophylactic and therapeutic regimens against SARS coronavirus in Rhesus macaque. Nat. Med. 2005;11:944–951. doi: 10.1038/nm1280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu M., Ding H., Zhao P., Qin Z.L., Gao J., Cao M.M., Luan J., Wu W.B., Qi Z.T. RNA interference effectively inhibits mRNA accumulation and protein expression of hepatitis C virus core and E2 genes in human cells. Biosci. Biotechnol. Biochem. 2006;70:2049–2055. doi: 10.1271/bbb.60001. [DOI] [PubMed] [Google Scholar]
- McBride J.L., Boudreau R.L., Harper S.Q., Staber P.D., Monteys A.M., Martins I., Gilmore B.L., Burstein H., Peluso R.W., Polisky B., Carter B.J., Davidson B.L. Artificial miRNAs mitigate shRNA-mediated toxicity in the brain: implications for the therapeutic development of RNAi. Proc. Natl. Acad. Sci. U.S.A. 2008;105:5868–5873. doi: 10.1073/pnas.0801775105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCaffrey A.P., Nakai H., Pandey K., Huang Z., Salazar F.H., Xu H., Wieland S.F., Marion P.L., Kay M.A. Inhibition of hepatitis B virus in mice by RNA interference. Nat. Biotechnol. 2003;21:639–644. doi: 10.1038/nbt824. [DOI] [PubMed] [Google Scholar]
- Mullan B.P., Davies G.T., Cutler R.S. Simulation of the economic impact of transmissible gastroenteritis on commercial pig production in Australia. Aust. Vet. J. 1994;71:151–154. doi: 10.1111/j.1751-0813.1994.tb03370.x. [DOI] [PubMed] [Google Scholar]
- Rossen J.W., Bekker C.P., Voorhout W.F., Strous G.J., van der Ende A., Rottier P.J. Entry and release of transmissible gastroenteritis coronavirus are restricted to apical surface of polarized epithelial cells. J. Virol. 1994;68:7966–7973. doi: 10.1128/jvi.68.12.7966-7973.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saif L.J. Animal coronavirus vaccines: lessons for SARS. Dev. Biol. (Base I) 2004;119:129–140. [PubMed] [Google Scholar]
- Schwegmann-Wessels C., Herrler G. Sialic acids as receptor determinants for coronaviruses. Glycoconj. J. 2006;23:51–58. doi: 10.1007/s10719-006-5437-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaucheret H., Béclin C., Fagard M. Post-transcriptional gene silencing in plants. J. Cell Sci. 2001;114:3083–3091. doi: 10.1242/jcs.114.17.3083. [DOI] [PubMed] [Google Scholar]
- Weingartl H.M., Derbyshire J.B. Evidence for a putative second receptor for porcine transmissible gastroenteritis virus on the villous enterocytes of newborn pigs. J. Virol. 1994;68:7253–7259. doi: 10.1128/jvi.68.11.7253-7259.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weingartl H.M., Derbyshire J.B. Cellular receptors for transmissible gastroenteritis virus on porcine enterocytes. Adv. Exp. Med. Biol. 1995;380:325–329. doi: 10.1007/978-1-4615-1899-0_53. [DOI] [PubMed] [Google Scholar]
- Zhou J.F., Hua X.G., Cui L., Zhu J.G., Miao D.N., Zou Y., He X.Z., Su W.G. Effective inhibition of porcine transmissible gastroenteritis virus replication in ST cells by shRNAs targeting RNA-dependent RNA polymerase gene. Antiviral Res. 2007;74:36–42. doi: 10.1016/j.antiviral.2006.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]


