Highlights
-
•
The number of animal disease outbreaks is increasing in the world.
-
•
During a disease outbreak, proper disposal of animal mortalities is critical.
-
•
Composting eliminates both bacterial and viral pathogens.
-
•
Composting was successfully used during the 2014–2015 AI outbreak in the U.S.
-
•
Research is needed to show effectiveness of composting in eliminating microbial DNA.
Keywords: Biosecure, Composting, Disease, Mortality, Livestock, Outbreak
Abstract
Despite the development of new vaccines and the application of rigorous biosecurity measures, animal diseases pose a continuing threat to animal health, food safety, national economy, and the environment. Intense livestock production, increased travel, and changing climate have increased the risk of catastrophic animal losses due to infectious diseases. In the event of an outbreak, it is essential to properly manage the infected animals to prevent the spread of diseases. The most common disposal methods used during a disease outbreak include burial, landfilling, incineration and composting. Biosecurity, transportation logistics, public perception, and environmental concerns limit the use of some of these methods. During a disease outbreak, the large number of mortalities often exceeds the capacity of local rendering plants and landfills. Transporting mortalities to disposal and incineration facilities outside the production operation introduces biosecurity risks. Burying mortalities is limited by the size and availability of suitable sites and it has the risk of pathogen survival and contamination of groundwater and soil. Portable incinerators are expensive and have the potential to aerosolize infectious particles. Composting, on the other hand, has been recognized as a biosecure disposal method. Research showed that it eliminates bacterial pathogens such as Escherichia coli O157: H7, Salmonella spp., as well as viruses including highly pathogenic avian influenza, foot-and-mouth disease, Newcastle disease, and porcine epidemic diarrhea. This paper summarizes the lessons learned during the major animal disease outbreaks including the 2010 foot-and-mouth disease, 2016 highly pathogenic avian influenza, and recent African swine fever outbreaks. The purpose of this review is to critically discuss the biosecurity of composting as a mortality disposal method during the outbreaks of infectious animal diseases.
1. Introduction
Despite the development of new vaccines and the application of rigorous biosecurity measures, animal diseases pose a continuing threat to animal health, food safety, the national economy and the environment (Wilkinson et al., 2011). Intense livestock production, increased travel, and changing climate have increased the risk of catastrophic animal losses due to infectious diseases (Gilroyed et al., 2016, Nrcs, 2015). Management of these diseases is essential in view of the threats they pose to health, farm incomes, and the viability of animal agriculture.
The 2014–2015 highly pathogenic avian influenza (HPAI) outbreak, the largest animal-health emergency in the U.S., resulted in the loss of nearly 50 million birds and cost the poultry industry $3.3 billion (Spackman et al., 2016). An additional $610 million was spent by the government in emergency efforts to contain the spread of the disease (McKenna, 2015). After its first appearance in April 2013, the spread of Porcine Epidemic Diarrhea virus (PEDv) in the U.S. caused a loss of 8 million animals (mostly pre-weaned piglets) in one year (NPPC, 2014). Porcine reproductive and respiratory syndrome (PPRS) is considered to be one of the most important diseases affecting the swine industry. Holtkamp et al. (2013) estimated the total cost of PRSS in the U.S. at $664 million annually, which is about 19% more than the annual cost estimated in 2005. Foot-and-mouth disease (FMD) outbreaks in the U.K. (2001) and in South Korea & Japan (2010–2011) caused the loss of thousands of animals. During the 2001 FMD outbreak in the United Kingdom, which had 2000 reported cases, dramatic photos of cattle burning in open pyres resulted in widespread public opposition to open burning. In South Korea, disease eradication efforts resulted in culling 20 percent of the country’s livestock population and the creation of over 4583 burial sites. This widespread carcass burial raised environmental concerns. In Japan, invasion of the disease into a high-density area led to difficulties in finding suitable burial sites, which resulted in delays in disease eradication efforts and required the Japanese government to implement a vaccinate-to-kill method (Flory and Peer, 2017, Kim et al., 2017). During an outbreak, being ready to properly manage the infected animals is essential to prevent the spread of the disease. A study of a hypothetical FMD outbreak in California concluded that delaying responding to the outbreak from 7 days to 22 days increases the estimated economic impact from $3 billion to $69 billion with 2000 additional cattle slaughtered for each extra hour of delay after 21 days (Carpenter et al., 2011).
In the event of an animal disease outbreak, proper measures have to be put in practice to control on-site pathogen proliferation and to prevent site-to-site transmission. This includes rapid culling of the animals exposed to the pathogens and proper disposal of mortalities and potentially contaminated materials. The most common disposal methods used during outbreaks include burial, landfilling, incineration and composting (Gwyther et al., 2011). However, biosecurity, transportation logistics, public perception, and environmental concerns limit the use of some of these methods (Benson et al., 2008). During a disease outbreak, the capacity of local rendering plants and landfills is often exceeded by the large number of mortalities. The 2001 FMD outbreak in the United Kingdom required the disposal of 6.5 million animals and created major difficulties to manage mortalities (Guan et al., 2010). Transporting mortalities to disposal facilities outside the production operation is a challenge that introduces new biosecurity risks (Gilroyed et al., 2016). Burying mortalities is limited by the size and availability of suitable sites and it has the risk of pathogen survival and contamination of groundwater and soil (Bonhotal et al., 2009). In South Korea, most of the buried mortalities were found to be undecomposed even after 3 years (Kim et al., 2017). Incineration can also be used but transporting mortalities to dedicated facilities is often undesirable. Portable incinerators are expensive, require an operating air permit in some states, and have the potential to aerosolize infectious particles (Ducey et al., 2017). Composting has been widely used by animal producers to manage daily mortalities. When first introduced as a diseased mortality disposal method, there were concerns regarding the availability of carbon source, generation of leachate and nuisance odors, and lack of research addresses the suitability of composting as a biosecure mass mortality disposal method. Since then, some of these concerns have been extensively discussed in the literature and addressed in recent studies. Previously two review papers regarding the disposal of animal mortalities during disease outbreaks were published. Wilkinson (2007) published a review paper which summarized the available information on the biosecurity of mortality composting and reports of the use of composting in emergency animal disease outbreaks. This paper was prepared more than a decade ago and the 2010–2011 foot-and-mouth disease outbreaks and the 2015–2016 avian influenza outbreaks, which had huge economic, animal health, and environmental consequences were not included in this paper. Gwyther et al. (2011) published another review paper and discussed the major routine disposal routes used throughout the world and the biosecurity and environmental credentials of each. This paper provides valuable information about different mortality disposable methods but it does not necessarily focus on composting. It also does not cover the recent avian influenza and African swine fever outbreaks, where composting has been used as the main mortality management method. In this review paper, an overview of the disposal methods used during major animal disease outbreaks are summarized. Each section was followed by a summary of the research that has been done to evaluate the effectiveness of composting in eliminating disease-causing pathogens. The purpose of this review is to critically discuss the biosecurity of composting as a mortality disposal method during the outbreaks of infectious animal diseases.
2. Animal mortality composting
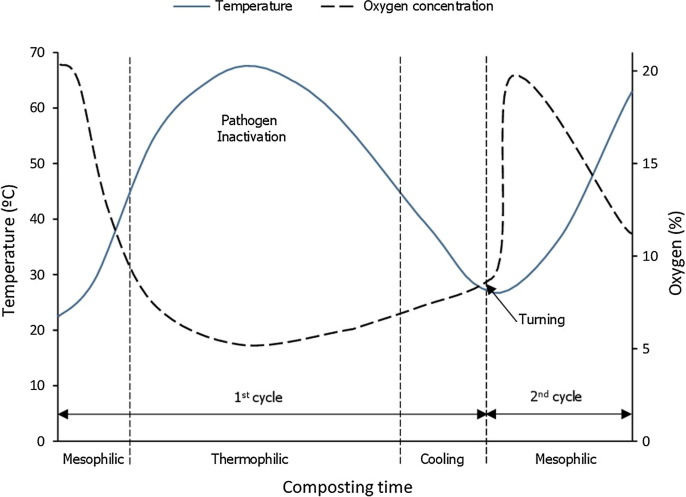
Biosecurity agencies in the U.S., Canada, Australia, and New Zealand have identified composting as a preferred disposal method for both routine and emergency management of animal mortalities (Wilkinson, 2007). Keener et al. (2000) described the composting process as an above-ground burial in a biofilter with heat inactivation of pathogens. There are two heating cycles. Fig. 1 . shows the temperature development and stages of the process.
Fig. 1.
Temperature development and stages of mortality composting (modified from NRAES, 1999, Sánchez et al., 2017).
Inactivation of pathogens is driven by both temperature and time. Exposure to an average temperature of 55–60 °C for a couple of days is sufficient to eliminate nearly all pathogenic viruses, bacteria, fungi, and protozoa (Epstein, 1997, Haug, 1993, Kalbasi et al., 2005). U.S. Environmental Protection Agency’s (USEPA) Class A and Class B time-temperature criteria are often used to assess pathogen reduction during animal mortality composting. Class A criteria require a composting temperature of 55 °C or higher for at least 3 consecutive days. Class B criteria call for compost temperature of at least 40 °C for 5 or more consecutive days with the temperature exceeding 55 °C for at least 4 h during this period. These criteria have been shown to reduce pathogenic bacteria (e.g., Salmonella and Campylobacter) found in sewage sludge (Glanville et al., 2016, USEPA, 2003). The Class A and B criteria are also used to assess viral pathogen elimination. Guan et al. (2009) reported that compost temperatures of 40–50 °C were sufficient to eliminate both avian influenza and Newcastle disease viruses in less than 3 days. Elving et al. (2012) showed that HPAI viruses present in fresh poultry manure can be inactivated at mesophilic temperatures (35 °C) within the first 24 h of composting. Results found by Vitosh-Sillman et al. (2017) indicated that PEDV in pig mortalities can also be inactivated in the first day of composting at a temperature of 37 °C. Guan et al. (2012) showed that a compost temperature of 41 °C for a day was able to inactivate bovine viral diarrhea virus (BVDV) and its RNA. Because of its close relation to BVDV, similar results were anticipated for the swine fever virus.
Even though composting is a promising method in response to a variety of infectious diseases, its application may be limited for Mycobacterium avium subsp. paratuberculosis, which causes Johne’s disease in cattle. Tkachuk et al. (2013) conducted an in vitro experiment to examine the viability of M.avium subsp. paratuberculosis after being exposed to 80 °C for 90 days. The pathogen remained viable after being exposed to the temperatures significantly higher than the ones typically achieved during composting. Another example is bovine spongiform encephalopathy (BSE). Composting was reported to be unlikely to destroy all BSE prions because of the variability in compost conditions (Xu et al., 2011). Adding feathers to the compost mix was shown to encourage the growth of microbial communities that degrade prions (Xu et al., 2013).
While composting is sufficient to eliminate most of the pathogens, its ability to degrade DNA to the point where the encoded genetic information is not viable remains undetermined. This risks introducing microbial DNA that carries pathogenic factors such as antibiotic-resistant genes (antibiotic resistant genes) to agricultural environments (Ducey et al., 2017). Once they are introduced, microbial DNA can be transformed into pathogenic organisms, which can potentially pose health risks to animals and humans or into non-pathogenic organisms, which may be served as a reservoir for future transfer into pathogenic organisms (Ducey et al., 2017). Suyama and Kawaharasaki (2013) reported that purified DNA survived steam autoclave conditions (121 °C and 0.1 MPa) for over 60 min before reaching undetectable levels. More studies are needed regarding the fate of antibiotic-resistant genes during the composting process (Cai et al., 2018).
The composting process is aerobic in nature, requiring sufficient oxygen to sustain the bioconversion. Oxygen availability and sufficient pile moisture are critical to successful composting. The oxygen concentration of the compost pile should not be lower than 5% (by volume). For optimal composting, a C:N ratio of between 25:1–30:1 (w/w), moisture content within the range of 50–60% (by weight), porosity of 35 to 45%, and oxygen levels higher than 10% are recommended (Ahn et al., 2008, Benson et al., 2008, Wilkinson, 2007). At the beginning of the process, oxygen levels can be easily kept above 10%, but as the degradation process progresses, the oxygen levels decrease. Compost materials are regularly turned to increase aeration and exposure of the anaerobic zone in the core to air. The timing of the turning is typically based on temperature monitoring of the compost material. It is important to time turning correctly especially during diseased mortality composting. Turning the compost piles too early can result in the release of pathogens into the air (Glanville et al., 2006).
The cover material is equally important to successful composting. The cover material retains heat, absorbs excess moisture, and provides a barrier that helps discourage insects and scavengers. Sawdust is acknowledged as an excellent cover material due to its ability to retain heat and absorb excess moisture. Unfortunately, sawdust is in high demand for many other uses, making it increasingly expensive and hard to obtain in recent years (Glanville, 2008). Alternative cover materials that are easier to obtain include woodchips, corn silage, and ground cornstalks or straw. Use of unground cornstalks or straw was reported to cause bridging, poor heat retention, downwind odor and heavy fly infestation (Glanville et al., 2006). Since these materials tend to be less absorptive and have poorer insulation properties compared to sawdust, their use requires more care during wet and cold weather conditions.
Using roofed bins simplifies management of the composting sites. Roofs prevent excessive moisture and leachate release and walls improve heat retention during cold weather and help to avoid problems with scavenging insects and animals (Glanville, 2008). However, bin systems are not well suited for emergency disposal since they are typically sized for average daily loss rates. Windrow composting systems can be used for emergency disposal. They can be sized to fit varying quantities and can be constructed quickly using on-farm equipment and materials (Glanville and Harmon, 2008). Windrow composting was successfully used to dispose of poultry mortalities during a number of outbreaks including the 2002 Virginia, 2004 Maryland and Delaware, 2004 and 2009 British Columbia, and 2014–2016 U.S. avian influenza outbreaks.
3. Major animal disease outbreaks in the world and relevant research
Animal disease outbreaks and managing the disposal of infectious animal mortalities are a global concern (Koziel et al., 2017). Koziel et al. (2018) summarized the epidemiologically significant outbreaks between 2005 and 2016 and reported that even though the disease type and the number of cases vary from year to year, the overall trend of total cases is increasing, which indicates that there is a need for preparedness and effective emergency disposal methods. Composting has been used during large-scale disease outbreaks and several studies have successfully demonstrated the efficacy of this disposal method in deactivating both bacterial and viral pathogens from poultry and livestock mortalities and waste. A summary of the outbreaks affecting livestock and poultry production all around the world and relevant research is provided below. Table 1 presents a summary of major animal disease outbreaks and Table 2 summarizes the relevant research in this field.
Table 1.
Major animal disease outbreaks and disposal methods.
| Outbreak | # of animals affected | Main disposal method | Highlights | References |
|---|---|---|---|---|
| Highly pathogenic avian influenza | ||||
| 1997 Hong Kong | 1.3 million | Landfill | – Caused the death of 6 people – The country’s entire poultry population was culled |
Lee et al., 2005, Chan, 2009 |
| 2002 Virginia (U.S.) | 4.7 million | Composting, incineration, landfill, controlled slaughter | – Cost $211 million to eradicate the disease – Rendering was not used due to biosecurity concerns – 43 thousand birds were composted using in-house and Ag-Bag systems |
Bendfeldt et al., 2005, Wilkinson, 2007 |
| 2003 Geldersei Valley (Netherlands) | 25 million | Incineration | – One person died and 80 people were infected by the HPAI H7N7 subtype – Culled birds were shipped in sealed trucks to incineration plants |
WHO, 2003, Swayne and Akey, 2005, Alexander, 2007, Elbers et al., 2009 |
| 2004 British Columbia | 17 million | Composting, incineration, burial | – Cost 380 million Canadian dollars 50% of the mortalities composted inside the barns – 5 days later windrows were moved outside and covered with plastic sheets |
Spencer et al., 2004, Wilkinson, 2007, Pasick et al., 2009 |
| 2004 Thailand | 62 million | Burial | – First HPAI (H5N1) outbreak reported in Thailand – 12 people died from the disease |
Parry, 2004, Tiensin et al., 2005, Nature Reports, 2018 |
| 2014–2015 U.S. | 50 million | Composting, burial, incineration & landfill | – One of the most impactful animal health events in the United States’ history – 85% of carcasses were composted |
USDA APHIS (2017), USEPA (2018) |
| 2016 U.S. Indiana | 400 thousand | Composting | – Animal mortalities were composted inside the barns – Depopulation was done by the producers with the assistance of inmate volunteers |
BOAH, 2016, Gelski, 2016, Brown et al., 2018 |
| Foot-and-mouth disease outbreaks | ||||
| 1870–1929 U.S. | 172 thousand | Not reported | – It affected cattle, sheep, and swine in 22 states – North America stayed FMD-free since 1929 through the use of stringent biosecurity measures |
Pendell et al., 2007, EDEN, 2009 |
| 1967 U.K. | 400 thousand | Burial & incineration | – On-farm burial was the major disposal method but was problematic near water resources and lime use impeded degradation | Scudamore et al., 2002, EDEN, 2009 |
| 1997 Taiwan | 3.8 million | Burial & incineration | – The depopulation was a massive task, which required substantial manpower from military – 80% of mortalities were buried – Groundwater contamination in the burial sites |
EDEN, 2009, Hseu and Chen, 2017 |
| 2001 U.K. & Netherlands | 6.5 million | Burial, burning, landfilling & rendering | – Disease had gone unreported for 3 weeks and the delay caused the disease to become an epidemic across the U.K. – On-farm burial was restricted to protect groundwater – Burning was suspended due to public health concerns |
Scudamore et al., 2002, Scudamore and Harris, 2002, Hseu and Chen, 2017 |
| 2010–2011 South Korea | 3.5 million | Burial | – Outbreak aggravated by delays in culling – Farmers faced difficulties in securing burial sites – Some animals were buried alive because the government ran out of the euthanasia drugs |
Park et al., 2013, Ko et al., 2017, Ki et al., 2018 |
| 2011 Japan | 290 thousand | Burial | – Finding enough space to bury animals was a challenge | Hayama et al. (2015) |
| Porcine epidemic diarrhea | ||||
| 2013 U.S, endemic in China, Korea, Vietnam, Japan, and the Philippines | 3% of the swine population in the U.S. | Not reported | – PED is not nationally or internationally reported – There is no PED vaccine in the U.S. |
USDA, 2013, Paarlberg, 2014 |
| Porcine reproductive and respiratory syndrome | ||||
| 2006 China | 2 million | Not reported | – This disease is reported in 20% to 40% of sow herds | Keffaber, 1989, Li et al., 2012, Nguyen, 2013 |
Table 2.
Summary of studies using composting as a pathogen inactivation method.
| Pathogen | Type of mortality/compost material | Cover and base material | Max T reached (°C) | Time of inactivation (days) | Reference |
|---|---|---|---|---|---|
| AI H5N2 and EDS-76 | Poultry | Oat straw and goat manure | 58.3 | 20 | Senne et al. (1994) |
| AI H7N1 | Poultry manure | Straw | 67.0 | 1 | Elving et al. (2012) |
| AI H7N1 and NDV | Poultry | Corn silage and poultry manure | 65.0 | 3 | Guan et al. (2009) |
| FMD | Swine | Wood shavings and poultry manure | 70.0 | 10 | Guan et al. (2010) |
| NDV | Cattle | Barley straw and cattle manure | 64.8 | 7 | Xu et al. (2009) |
| PEDV | Swine | Wood shavings | 57.0 | 1 | Vitosh-Sillman et al. (2017) |
| Salmonella and Campylobacter | Sewage sludge | Not reported | 55 | 3 | USEPA (2003) |
| Bovine Viral Diarrhea Virus | Manure and cattle | Not reported | 41 | 1 | Guan et al. (2012) |
3.1. Avian influenza (AI)
Avian influenza, also known as bird flu, is a respiratory disease caused by viruses infecting all avian species and can be divided into two groups based on their capacity of generating disease and the severity of the illness they cause: low pathogenic avian influenza (LPAI) and highly pathogenic avian influenza (HPAI) (Capua and Alexander, 2004, Keaten and Hutchinson, 2017). While LPAI viruses cause mild symptoms in poultry, infections by HPAI (H5N1 or H7N9) can lead to the death of more than 90% of the flock. HPAI presents a major public health concern because some HPAI strains are reportedly capable of causing severe illnesses in humans (CFSPH, 2014). Infections in human often happen due to direct contact with infected poultry. The first human infection occurred in 1997 during an HPAI outbreak in Hong Kong and resulted in 6 fatal cases (Keaten and Hutchinson, 2017, WHO, 2018a). Between 2003 and 2018, 860 H5N1 infections in human were reported, which caused the death of 454 people worldwide (WHO, 2018b). An outbreak of AI in domestic poultry can significantly affect global food security as poultry products are the main protein sources in many countries (Keaten and Hutchinson, 2017).
HPAI virus does not naturally occur in the U.S. However, the disease has been introduced in the country multiple times through migratory waterfowl and other potential sources such as infected poultry products, equipment, and people (APHIS-USDA, 2011). In a global perspective, the majority of HPAI cases are located in Asia as its vast territory accommodates innumerous wintering areas for migratory birds. Between 2010 and 2016, China, Vietnam, India, and Taiwan combined were the focus of more than 30% of the world’s reported AI outbreaks (Chatziprodromidou et al., 2018).
3.1.1. Avian influenza outbreaks
The 1997 HPAI outbreak in Hong Kong was of major significance to public health agencies all around the world as it was the first documented case of direct transmission of H5N1 AI virus from poultry to humans, causing the death of 6 of 18 infected people (Lee et al., 2005, Nature Reports, 2018). When the epidemiological investigations associated the deaths with H5N1 infection, the Hong Kong government decided to cull the country’s entire poultry population, which was reported to be 1.3 million chickens at the time (Chan, 2009).
The 2002 Virginia (U.S.) outbreak affected 197 poultry farms and cost $211 million to eradicate the disease. Initially, on-site burial was used for mortality disposal but soon stopped due to complaints about well water contamination. About 65% of the mortalities were sent to landfills which were expensive and problematic due to the lack of available trucks. Two flocks (43 thousand birds) were composted in-house (Bendfeldt et al., 2005, Wilkinson, 2007). Since the windrows were not constructed with the oversight of individuals familiar with composting, the compost material dried out within a month and the composting process was not successful. A commercial bag composting system (Ag-Bag), which could compost 50 tons of carcasses inside a plastic bag was investigated. The bag was sealed except for a few vent holes. Plastic pipes were placed inside the bag and air was forced through every minute by a blower (Brglez and Hahn, 2008). It was difficult to manage moisture and the material had to be re-composted upon its removal from the Ag-Bag system.
In 2003, an HPAI outbreak led the Dutch authorities to make the decision of culling approximately 25 million commercial birds in the densely populated poultry area of the Geldersei Valley, Netherlands (Alexander, 2007, Elbers et al., 2009). The severity of the incident escalated when it was reported that the virus responsible for the incident (H7N7) crossed the species barrier and caused the death of one person and more than 80 people presented symptoms of infection (WHO, 2003). The preferred method of disposal was incineration and it involved an extensive operation to ship the culled birds in sealed trucks from the farms to incineration plants that were able to comply with air-quality emission standards (Swayne and Akey, 2005).
During the 2004 Maryland and Delaware outbreaks, birds were composted inside the poultry houses and the AI virus was eliminated within 14 days (Malone et al., 2004). In 2004, the Canadian poultry industry was heavily impacted by a major AI outbreak that was started by an H7N3 LPAI strain, which went through a virulence shift to HPAI. The incident, which is regarded as Canada’s first reported outbreak of HPAI, resulted in the depopulation of 17 million birds and had an associated gross economic cost of more than 380 million Canadian dollars (Pasick et al., 2009). The birds were culled by sealing the barns and flooding them with carbon dioxide (CO2) gas. The disposal strategy relied on three main methods: incineration, burial and composting. Initially, most of the infected mortalities were placed in biosecure containers and transported to off-farm locations, where they were buried or incinerated. As the outbreak progressed, on-farm composting became the main disposal method and approximately half of the infected animals were composted (Wilkinson, 2007). Compost windrows were built inside the barns. Five days later the windrows were moved outdoors. The secondary composting was done outdoors on a layer of heavy-duty plastic sheet. For aeration, drainage pipes were placed across the plastic sheet layers. The windrows were covered with a layer of vapor barrier, wood shavings and another layer of plastic sheet (Spencer et al., 2004, Wilkinson, 2007).
In 2004, the AI subtype H5N1 was responsible for causing a major outbreak in Thailand. This was the first time an HPAI infection was reported in Thailand and over 62 million birds, including commercial and backyard birds, were killed either by the virus or culled to contain the spread of the disease (Tiensin et al., 2005). It was reported that 12 people died due to direct exposure to the virus (Nature Reports, 2018). Some of the birds had to be buried alive (Parry, 2004).
Between 2014 and 2015, the United States faced an HPAI outbreak. It remains the largest animal health emergency in the U.S. to date. The disease spread through 21 states and resulted in a loss of approximately 7.5 million turkeys and 42.1 million laying hens (Johnson et al., 2016, Ramos et al., 2017, Spackman et al., 2016). Iowa and Minnesota were the states most affected. Iowa lost about 32 million birds (95% of chickens) while the loss in Minnesota was 9 million birds (65% of turkeys) (Johnson et al., 2016). A total of 56 trading partners, including China, Russia, and South Korea, imposed bans or partial bans on trading U.S. poultry products. The U.S. government spent $879 million to clean-up the outbreak: $610 million for depopulation, cleaning and disinfecting $200 million for market value indemnification, and $69 million for overtime, supplies, travel, etc. (Johnson et al., 2016). The disposal of such a large number of animal mortalities was a challenging task. Composting was the main disposal method and was used to dispose of 85% percent of the poultry mortalities (USEPA, 2018). Animals were mostly composted inside the poultry houses but outdoor composting was also used for egg layer facilities or double-decker houses that do not provide adequate indoor floor space or ceiling clearance for using a loader to build windrows inside the facilities. Burial, incineration, and landfill were the other methods used during the outbreak. Due to environmental concerns, both incineration and burial were not applied on a large scale and landfilling were made possible only after extensive negotiation with private owned landfills that initially refused to accept infected birds (Windhorst, 2015). This outbreak prompted the USDA to take action to prepare for events of this nature in the future. Among the several areas identified by the agency as a priority, two stand out: biosecurity and disposal of mortalities. The first has been addressed by developing training material for the response teams as producers reported biosecurity infractions committed by personnel contracted by the USDA. The second is focused on promoting composting as a preferred method of disposal and training USDA personnel in large-scale composting to provide guidance to producers in the event of an outbreak (GAO, 2017). The USDA also established a Composting Technical Committee and developed the HPAI Composting Protocol to promote consistency in the composting process (USDA, 2017).
Following the 2014–2015 outbreak, a combined outbreak of HPAI and LPAI occurred in Indiana which led to the depopulation of more than 400 thousand birds (BOAH, 2016). The event was not a resurgence or continuation of the 2014–2015 outbreak as the virus strains identified in both cases were different (Gelski, 2016). Depopulation was done by the producers and industry representatives with the assistance of inmate volunteers from the Indiana Department of Correction (IDOC). The mortalities were composted inside the barns (Brown et al., 2018). The USDA APHIS HPAI Response Plan: The Red Book was updated in May 2017 (APHIS-USDA, 2017). It reflects the knowledge and lessons learned during the 2014–2015 and 2016 AI outbreaks in the U.S. The Livestock Mortality Composting Protocol was subsequently published in August 2017. It summarizes the key elements for successful composting, labor, equipment, supply and material requirements, protocol, and troubleshooting (USDA, 2017).
3.1.2. Research on composting avian influenza infected mortalities
The effectiveness of composting in deactivating HPAI strains has been extensively studied and their findings lead to the use of composting as the main disposal method during the 2004 and the 2014–2015 outbreaks in North America. In 1994, a study conducted by Senne et al. (1994) evaluated the performance of composting in deactivating exotic avian viruses and adenovirus. The study consisted of composting two groups of chickens. One was infected with HPAI virus and the other with the adenovirus that causes egg drop syndrome 76 (EDS-76). At the end of the first composting cycle, which had a duration of 10 days, it was reported that the HPAI had been completely inactivated but the EDS-76 adenovirus was still detectable. Subsequently, the compost was turned and by the end of the new 10-day cycle, both viruses were completely inactivated. The daily average temperature of the upper layer remained above 50 °C for 7 days in the first cycle and 8 days in the second.
Besides the animal mortalities, other materials such as the feed, bedding, and manure are potential sources of spread of pathogens. In the event of an outbreak, they also must be disposed of properly and in a safe manner. Ideally, it is more practical to dispose of these materials with the mortalities and, as reported by Guan et al. (2009), the presence of active microorganisms in these materials can be beneficial for the degradation process in composting systems. In terms of pathogen inactivation, a study conducted by Elving et al. (2012) has shown that HPAI virus (H7N1) present in fresh poultry manure can be inactivated within the first 24 h of composting. This is a significant achievement since HPAI strain has been reported to survive more than a year in soil amended with manure (Elving et al., 2012). In this study, which was performed using a laboratory-scale reactor, it was found that although the peak temperatures ranged from 42 °C to 67 °C, thermal inactivation of H7N1 can also happen at mesophilic temperatures (35 °C). This discovery is particularly important because the peak temperatures usually occur only in the core of the pile and the same peaks are rarely observed at the surface of the compost material.
Another important finding was made by Guan et al. (2009) when studying the survival of AI and New Castle Disease (ND) viruses. It was reported that microbial activity plays a crucial role not only in inactivating pathogens but also in degrading viral RNA. A passively aerated static composting system was used to compost chicken mortalities along with manure, feed, and eggs, which were placed in either mesh-bags or sealed vials. While both virus and viral RNA were inactivated by day 10 in mesh-bags, viral RNA was still detected by day 21 in sealed vials.
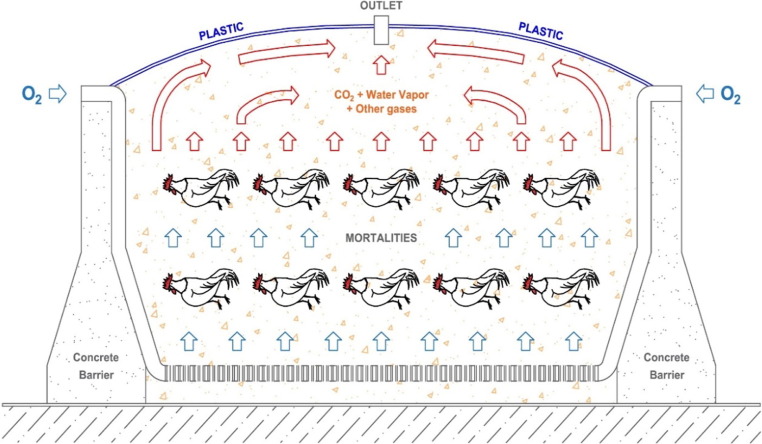
Plastic-wrapped composting systems were first used during the AI outbreak in British Columbia (Spencer et al., 2004). A plastic sheet used to cover the windrows in order to reduce the risk of spreading viruses into the surrounding environment by bird and insect activity, wind, and precipitation. To oxygenate the pile, the plastic-wrapped compost pile was passively aerated through 10-cm diameter slotted plastic drainage tubing passing through the base. Fig. 2 shows the schematic of a composting system similar to the one used during the British Columbia outbreak. After the successful implementation of this system in British Columbia, its performance for biosecure emergency disposal of disease-related swine mortalities was evaluated by Glanville et al. (2016). One of the concerns was the compost material had a very limited ventilation area (about 0.25% of a conventional compost system) due to the plastic sheet cover. To address this concern, Glanville et al. (2016) evaluated the system’s ability to sustain an adequate oxygen supply and to remove large quantities of water and decomposition gases. Another concern was monitoring the progress and completion of the process without removing the cover. Volatile organic compounds (VOCs) were used as biomarkers of decaying mortalities (Akdeniz et al., 2011, Akdeniz et al., 2010a, Akdeniz et al., 2010b, Akdeniz et al., 2009). Other researchers also recognized the importance of predicting the maturity of the biosecure composting systems. Xu et al. (2011) composted cattle mortalities and it was reported that bovine-specific DNA is a good indicator of tissue degradation.
Fig. 2.
Schematic of the plastic-wrapped composting system.
3.2. Foot-and-mouth disease (FMD)
Foot-and-mouth disease (FMD) is a highly contagious viral disease that can affect all cloven-hoofed animals including cows, pigs, sheep, goats, deer, and other animals with divided, or split hooves. It does not affect horses, cats, or dogs and it is not related to the hand, foot, and mouth disease, a common childhood illness in humans. The FMD virus is spread in aerosols and on fomites such as contaminated tires, boots, and clothing. There are seven types and more than 60 subtypes of the virus. To be effective, vaccines must be closely matched to the virus strain causing the disease. Animals with FMD typically develop a fever and lesions in the mouth, on the mammary glands, and around the hooves. Other signs include depression, anorexia, excessive salivation, and reluctance to move. FMD is a concern all around the world because it can spread quickly and cause significant economic losses. A single detection of FMD would affect export markets and suppliers, causing billions of dollars in lost trade (APHIS-USDA, 2018a, NCBA, 2018). The disposal of mortalities and infected materials has shown to cause problems in previous FMD outbreaks and, although composting has never been used in past outbreaks, more recent studies have shown that it could be a viable alternative during an outbreak.
3.2.1. FMD outbreaks
The United States has experienced FMD outbreaks nine times between 1870 and 1929. The most serious outbreak started in Michigan in 1914 and spread to the Chicago stockyards by 1915. It affected 22 states. About 172,000 cattle, sheep, and swine were culled during the eradication program (Pendell et al., 2007). The last outbreak occurred in California. The animals were infected after being fed swill with meat scraps from a tourist steamship coming from Argentina. North America stayed FMD-free since 1929 through the use of stringent biosecurity measures (EDEN, 2009).
In 1967, the United Kingdom had an FMD outbreak. Although the disease was reported quickly, more than 400,000 animals had to be slaughtered (EDEN, 2009). On-farm burial was the most used disposal method and the preferred disposal method for rapid disposal and elimination of virus load. However, some disposal sites were not suitable for burial since they were too close to the water resources. Also, the lime that had been added to the burial pits was found to inhibit the natural degradation process (Scudamore et al., 2002). On-farm burning was another method used during the outbreak (Scudamore et al., 2002).
In 1997, a sow at a farm in Taiwan was diagnosed with a strain of FMD that infected swine only. The disease spread very quickly and caused the loss of over 3.8 million swine at an estimated cost of US $6.9 billion (EDEN, 2009). The depopulation was a massive task, which required substantial manpower from the military. At peak capacity, 200,000 pigs were culled daily. More than 97% of the pigs were culled with an electric shock. The rest of them were culled using other methods such as poisoning or drowning. Approximately 80% of the mortalities were disposed of by burying in large municipal landfills. Lime was applied during the construction and management of the burial sites to eliminate pathogens. To investigate groundwater quality, Taiwanese governmental agencies analyzed 3,723 groundwater samples during two years using a budget of US $1.5 million. Groundwater at the burial sites was found to be contaminated, particularly at the sites without an impermeable cloth and those located close to the wells. Burial was avoided in water resource protection areas, which considerably impeded the disposal of the mortalities. One month after the outbreak started, industrial incinerators were running around the clock to dispose of the mortalities (Hseu and Chen, 2017).
In 2001, the U.K. had another FMD outbreak. A total of 2026 cases of FMD were confirmed in 7 months. Almost 6.5 million animals were culled to stop the spread of the disease. Disease had gone unreported for at least three weeks and the delay caused the disease to become an epidemic across the U.K. (Scudamore and Harris, 2002). Similar to the 1967 outbreak, on-farm burial and on-farm burning were initially employed to dispose of mortalities. On-farm burial was restricted by legislation to protect groundwater supplies. Due to health concerns regarding the smoke and emissions produced, burning was heavily criticized by local communities. The outbreak could only be managed by using mass burial in engineered sites and landfilling where available. Approximately 61,000 tons of mortalities were disposed of at four burial sites (Scudamore et al., 2002). To minimize the risk of environmental pollution, groundwater vulnerability maps were used to locate suitable mass burial sites (Hseu and Chen, 2017). During the course of the outbreak, a disposal hierarchy was developed to address environmental and public health concerns. According to this hierarchy, rendering and incineration ranked first, licensed landfill next, followed by mass burial or on-farm burial, which were the least preferred options. However, it was not possible to overcome the outbreak without building mass burial sites (Scudamore et al., 2002, Scudamore and Harris, 2002). A month after the U.K. outbreak, the Netherlands had an FMD outbreak. The rendering and culling capacity was not sufficient. As a solution, suppressive vaccination was used to halt the possible spread of the virus. The vaccinated animals could be removed alive and killed in central places 14 days after vaccination. Using slaughterhouses as central culling places made freezing parts of the mortalities possible. When rendering capacity became available, the frozen mortalities were removed from the freezers and sent to the rendering facilities (de Klerk, 2002).
An outbreak of FMD caused by serotype O virus occurred in South Korea during November 2010–April 2011. The virus was likely brought into the country due to a farmer’s trip to Southeast Asia since the virus was similar to FMD serotype O circulating in Southeast Asia. The outbreak affected 3700 farms. Some farmers who were required to cull their animals did not do it in a timely manner, which contributed to a spike in new infections in the 38th-64th days of the outbreak. The rates declined rapidly after the culling and completion of a national vaccination program. Most culled animals were disposed of by burial. About 3.48 million animals (cattle, pigs, goats, and deer) were buried at 4,583 burial sites (Ko et al., 2017, Park et al., 2013). Korean farmers faced difficulties in securing burial sites (Ki et al., 2018). It was reported that some animals were buried alive because the government ran out of euthanasia drugs. This brought the attention of animal rights groups, including the People for the Ethical Treatment of Animals (PETA) (Wagner and Choi, 2011). South Korea requested vaccines from the U.S. Once vaccination was expanded nationwide, a vaccination-to-live policy was started to be implemented. South Korea is still not free of the FMD virus. In March 2018, South Korea confirmed a case of rare type-A on a pig farm. Following the outbreak in South Korea, Japan had an FMD outbreak. During the outbreak, a total of 292 outbreaks were confirmed and about 290,000 animals were culled, of which 210,000 animals were from detected farms and 80,000 were from vaccinated farms. Since the outbreak happened in an area with a high density of livestock farms, finding enough suitable land for burying mortalities became a challenge. Mortalities had to be transported to burial sites away from the farms with careful implementation of biosecurity measures (Hayama et al., 2015).
3.2.2. Research on composting FMD infected animal mortalities
Composting can be used to dispose of large animals. Guan et al. (2010) demonstrated that composting can be safely used to heat treat FMD virus contaminated livestock mortalities. Three pigs infected with FMD virus through the oral and intranasal routes were composted in a mixture of chicken manure and wood shavings in a facility with biocontainment level 3. Compost temperatures reached 50 °C and 70 °C by days 10 and 19, respectively. The FMD virus was inactivated by day 10 and the viral RNA was degraded in the skin and internal organ tissues by day 21. Xu et al. (2009) tested the potential of using composting to dispose of cattle mortalities and manure during an FMD outbreak. A biosecure composting system that was passively aerated and constructed with large barley straw bales was developed. Due to biosecurity reasons, instead of FMD, vaccine strain NDV was used to infect mortalities. The whole composting process lasted 147 days and the temperatures reached to 55 °C by day 6. Virus inactivation achieved by day 7. A later study conducted by Xu et al. (2010) suggested a few changes in the setting used by Xu et al. (2009). By simply raising the mortalities higher from the bottom of the bin and utilizing manure with a high moisture content (60%), it was possible to improve heat retention and achieve more uniformity in temperature. The mortalities were composted for 230 days and a temperature of 55 °C was achieved by day 7 and remained above it for 70 days in all locations inside the bins. Kim et al. (2017) composted excavated mortalities from a 3-year old foot-and-mouth disease burial site in South Korea. The quantitative characterization of potentially pathogenic bacteria using the next-generation sequencing (NGS) showed that the burial-composting sequential system can be used to reduce the microbial risk to human health.
3.3. Porcine epidemic diarrhea (PED)
The porcine epidemic diarrhea is caused by a coronavirus similar to the transmissible gastroenteritis virus (TGEV). Diarrhea is a major symptom. The other signs include vomiting, dehydration, and weight loss. There is no immunological protection in piglets so the mortality and morbidity rates are very high (80–100%) (Weng et al., 2016). It was first reported in the U.K. in 1971. In the following years, the virus spread throughout Europe. After the 1980 s, the number of PED outbreaks decreased in Europe. However, PED has become an endemic disease in Asian countries including China, Korea, Vietnam, Japan, and the Philippines. It was diagnosed in the U.S. for the first time in Iowa, in 2013. The outbreak is estimated to have reduced swine population by 3% and resulted in a loss of approximately $1 billion to both producers and consumers (Paarlberg, 2014). Within a year, PED outbreaks occurred in Canada and Mexico (Song et al., 2015). PED is not a World Organization for Animal Health (OIE) listed disease. It is not nationally or internationally reportable. There is no PED vaccine in the U.S. Vaccines are available in South Korea, Japan, and China (USDA, 2013).
Findings of a study conducted by Vitosh-Sillman et al. (2017) indicated that virus inactivation in composting is a result of the combination of time and a high temperature of the compost cycle. Their experiment examined PED virus (PEDv) in pig mortalities and consisted of two trials, one in a laboratory setting to analyze the viral degradation rate under controlled conditions and another in which infected pig mortalities were composted in windrow sections. The results obtained in the laboratory trial showed that a temperature of 37 °C was enough to inactivate the virus in a period of 24 h. Although this result might seem inconsistent when compared to studies conducted with other viruses, this was attributed to the fact that, when compared to non-enveloped viruses such as FMD, the PED virus is less environmentally persistent. A mean compost temperature of 55 °C for 4 h was enough to achieve the degradation of PEDv RNA.
3.4. Porcine reproductive and respiratory syndrome (PRRS)
Porcine reproductive and respiratory syndrome (PRRS) is one of the most economically important swine infectious diseases in the world. Every year, outbreaks are reported in about 20% to 40% of sow herds causing $664 million in productivity losses in the U.S. (Holtkamp, 2018). It was first recognized in 1987 in the U.S. (Keffaber, 1989). The first case of the PRRS outbreak in Asia was reported in China in 1995. In 2006, China was hit by a highly virulent strain of PRRS virus (Li et al., 2012). 445 cases were reported, which affected more than two million pigs. The outbreak spread to Vietnam in 2007 (Nguyen, 2013). Following Vietnam, the disease rapidly spread in Southeast Asia, affecting many countries including Bhutan, Cambodia, Laos, Malaysia, Myanmar, the Philippines, Thailand, and Singapore. South Korea and Russia were also reported to be affected (An et al., 2011). PRRS virus (PRRSv) can remain infectious for an extended time during winter months since it has a long half-life time (T1/2). PRRSv half-life in manure was estimated at 120.5 h at 4 °C, 24.5 h at 20 °C, 1.7 h at 40 °C, 8.5 min at 60 °C, and 0.59 min at 80 °C. These data suggest that submitting transport trailers to a temperature of 50 °C for 8 h would decrease PRRSv from 106 TCID50/ml to less than 101 TCID50/ml (Linhares et al., 2012).
3.5. African swine fever (ASF)
African swine fever (ASF) was first described in Kenya in the 1920s. It is a highly contagious hemorrhagic disease of swine with extremely high morbidity and mortality rates. It is not a threat to human health. Unlike classic swine fever (hog cholera), which is caused by a different virus, there are no vaccines against ASF. The control of the disease is based on the implementation of sanitary measures including early detection and proper disposal of mortalities and waste. The disease is endemic in Sardinia and many African countries. Spain and Portugal eradicated ASF in the mid-1990s. The disease was also eradicated from the Caribbean following outbreaks from 1977 to 1980. However, the spread of ASF through Eastern Europe and Eurasia (Russia and the Caucasus region) and the recent introduction into Asia are a concern. It has never been reported in the U.S., Canada, Australia, or New Zealand (APHIS-USDA, 2018b). On August 1st, 2018, for the first time China reported to the World Organisation for Animal Health-OIE (Office International des Epizooties) an ASF outbreak on a domestic swine farm in Liaoning Province. Later, the disease spread to other regions in China. On January 14, 2019, the Ministry of Agriculture and Rural Affairs of China reported that the disease has reached 24 provinces and regions, and 916 thousand pigs have been culled since the first outbreak in August (The Pig Site, 2019). Inactivation of ASF virus (ASFv) during composting has never been tested. However, Turner et al. (1998) reported that when swine slurry was heat-treated, ASFv levels reduced to below detectable levels within 90 s at 56 °C.
4. Conclusions
Burial is the most commonly used method to manage mass mortalities. Very often finding suitable sites to burry mortalities is a challenge and mortalities remain undecomposed for years. Applying lime to prevent the contamination of groundwater inhibits the natural degradation process and mortalities stay undecomposed for longer. During the 2004 HPAI outbreak in British Columbia, initially, most of the infected mortalities were placed inside biosecure containers and transported to off-farm locations, where they were buried or incinerated. As the outbreak progressed, a biosecure composting system was employed to heat treat mortalities. This system was tested by a number of researchers (Guan et al., 2010, Xu et al., 2010, Glanville et al., 2016). During the 2014–2015 AI outbreak, some windrows were built in barns and they remained in the barns for the full 30-day cycle. Once the compost material went through two heating cycles, it could be moved outside and it was no longer considered infected. Poultry mortalities were not ground/crushed/macerated during construction of the windrows to prevent disease transmission (USDA, 2017). After the 2014–2015 and the 2016 outbreaks, the “Livestock Mortality Composting Protocol” was published by the USDA. Although composting has been shown to eliminate HPAI, FMD, PED, and PRRS viruses, no studies have been reported regarding African swine fever. More studies are needed to show the biosecurity of composting in eliminating infectious diseases and especially microbial DNA, which is often referred to be the reason for reoccurring diseases.
Acknowledgment
This project was supported by Agriculture and Food Research Initiative Competitive Grant no. 12463377 from the USDA National Institute of Food and Agriculture. The funding agency has no role in the collection and interpretation of the information; in the writing of the report; and in the decision to submit the article for publication.
References
- Ahn H.K., Richard T.L., Glanville T.D. Optimum moisture levels for biodegradation of mortality composting envelope materials. Waste Manag. 2008;28:1411–1416. doi: 10.1016/j.wasman.2007.05.022. [DOI] [PubMed] [Google Scholar]
- Akdeniz N., Koziel J.A., Ahn H.K., Glanville T.D., Crawford B.P. Field scale evaluation of volatile organic compound production inside biosecure swine mortality composts. Waste Manag. 2010;30:1981–1988. doi: 10.1016/j.wasman.2010.05.022. [DOI] [PubMed] [Google Scholar]
- Akdeniz N., Koziel J.A., Ahn H.K., Glanville T.D., Crawford B.P., Raman D.R. Laboratory scale evaluation of volatile organic compound emissions as indication of swine carcass degradation inside biosecure composting units. Bioresour. Technol. 2010;101:71–78. doi: 10.1016/j.biortech.2009.07.076. [DOI] [PubMed] [Google Scholar]
- Akdeniz N., Koziel J.A., Ahn H.K., Glanville T.D., Crawford B.P., Raman D.R. Air sampling and analysis method for volatile organic compounds (VOCs) related to field-scale mortality composting operations. J. Agric. Food Chem. 2009;57:5658–5664. doi: 10.1021/jf900801w. [DOI] [PubMed] [Google Scholar]
- Akdeniz N., Koziel J.A., Glanville T.D., Ahn H., Crawford B.P. Bioresource Technology Air sampling methods for VOCs related to field-scale biosecure swine mortality composting. Bioresour. Technol. 2011;102:3599–3602. doi: 10.1016/j.biortech.2010.10.100. [DOI] [PubMed] [Google Scholar]
- Alexander D.J. Summary of Avian Influenza Activity in Europe, Asia, Africa, and Australasia, 2002–2006 (Resumen de la actividad de la influenza aviar en Europa, Asia, África y Australasia entre los años 2002–2006) Author (s): Dennis. J. Alexander Source : Avia. Avian Dis. 2007;51:161–166. doi: 10.1637/7602-041306R.1. [DOI] [PubMed] [Google Scholar]
- An T.-Q., Tian Z.-J., Leng C.-L., Peng J.-M., Tong G.-Z. Highly pathogenic porcine reproductive and respiratory syndrome virus. Emerg. Infect. Dis. 2011;17:1782–1784. doi: 10.1093/bib/5.2.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- APHIS-USDA, 2018a. Animal disease information [WWW Document]. URL https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information (accessed 1.28.19).
- APHIS-USDA, 2018b. African Swine Fever [WWW Document]. URL https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/swine-disease-information/african-swine-fever (accessed 1.17.19).
- APHIS-USDA, 2017. HPAI Response Plan: The Red Book. [WWW Document]. URL https://www.aphis.usda.gov/animal_health/emergency_management/downloads/hpai_response_plan.pdf (accessed 1.22.19).
- APHIS-USDA, 2011. Protect your birds from Avian Influenza (Bird Flu) [WWW Document]. URL https://www.aphis.usda.gov/publications/animal_health/2011/ProtectYourBirdFromAI_Eng.pdf (accessed 1.22.19).
- Bendfeldt E.S., Peer R.W., Flory G.A. Lessons learned from avian influenza outbreaks in Virginia 1983 and 2002. In: Hutchinson M., editor. Proceedings of the Symposium on Composting Mortalities and Slaughterhouse Residuals. University of Maine Cooperative Extension; Waldoboro ME: 2005. pp. 6–9. [Google Scholar]
- Benson E.R., Malone G.W., Alphin R.L., Johnson K., Staicu E. Immunology, health and disease: Application of in-house mortality composting on viral inactivity of Newcastle disease virus. Poult. Sci. 2008;87:627–635. doi: 10.3382/ps.2007-00308. [DOI] [PubMed] [Google Scholar]
- BOAH, 2016. 2016 HPAI Information - affected Indiana counties [WWW Document]. Indiana State Board Anim. Heal. URL https://www.in.gov/boah/2745.htm (accessed 11.9.18).
- Bonhotal J., Waste C., Hall R. 3rd International Symposium: Management of Animal Carcasses, Tissue and Related Byproducts. Davis, California. 2009. Environmental effects of mortality disposal. [Google Scholar]
- Brglez B., Hahn J. Methods for disposal of poultry carcasses. In: Swayne D.E., editor. Avian Influenza. Blackwell Publishing; 2008. p. 587. [Google Scholar]
- Brown J.A., Patel R., Maitlen L., Oeding D., Gordon K., Clayton J.L., Richards S., Pontones P., Brewer J., Blosser S., Duwve J. Public health response to an Avian Influenza A(H7N8) virus outbreak in commercial turkey flocks-Indiana 2016. MMWR Morb Mortal Wkly. 2018;67:1339–1341. doi: 10.15585/mmwr.mm6748a2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai M., Ma S., Hu R., Tomberlin J.K., Thomashow L.S., Zheng L., Li W., Yu Z., Zhang J. Rapidly mitigating antibiotic resistant risks in chicken manure by Hermetia illucens bioconversion with intestinal microflora. Environ. Microbiol. 2018;20:4051–4062. doi: 10.1111/1462-2920.14450. [DOI] [PubMed] [Google Scholar]
- Capua I., Alexander D.J. Avian influenza : recent developments Avian influenza : recent developments. Avian Pathol. 2004;33:393–404. doi: 10.1080/03079450410001724085. [DOI] [PubMed] [Google Scholar]
- Carpenter T.E., Brien J.M.O., Hagerman A.D., Mccarl B.A. Epidemic and economic impacts of delayed detection of foot-and-mouth disease : a case study of a simulated outbreak in California. J. Vet. Diagn. Invest. 2011;33:26–33. doi: 10.1177/104063871102300104. [DOI] [PubMed] [Google Scholar]
- CFSPH, 2014. Avian Influenza. 10.1159/000151603. [DOI]
- Chan P.K.S. A review on human influenza A H5N1 infections in Hong Kong. Sci. China. Ser. C Life Sci. 2009;52:412–418. doi: 10.1007/s11427-009-0063-y. [DOI] [PubMed] [Google Scholar]
- Chatziprodromidou I.P., Arvanitidou M., Guitian J., Apostolou T., Vantarakis G., Vantarakis A. Global avian influenza outbreaks 2010–2016: A systematic review of their distribution, avian species and virus subtype. Syst. Rev. 2018;7:1–12. doi: 10.1186/s13643-018-0691-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Klerk P.F. Carcass disposal: lessons from The Netherlands after the foot and mouth disease outbreak of 2001. Rev. Sci. Tech. 2002;21:789–796. doi: 10.20506/rst.21.3.1376. [DOI] [PubMed] [Google Scholar]
- Ducey T.F., Collins J.C., Ro K.S., Woodbury B.L., Grif D.D. Hydrothermal carbonization of livestock mortality for the reduction of pathogens and microbially-derived DNA. Front. Environ. Sci. 2017;11 doi: 10.1007/s11783-017-0930-x. [DOI] [Google Scholar]
- EDEN, 2009. Foot and Mouth Disease [WWW Document]. Ext. Disaster Educ. Netw. URL https://eden.lsu.edu/educate/resources/foot-and-mouth-disease-archived-eden-topic-page/?tab=FrontPage (accessed 1.19.19).
- Elbers, A.A.R.W., Fabri, T.H.F., Vries, T.S. De, Wit, J.J. De, Pijpers, A., Koch, G., 2009. The Highly Pathogenic Avian Influenza A (H7N7) Virus Epidemic in the Netherlands in 2003 : Lessons Learned from the First Five Outbreaks Case Reports- The Highly Pathogenic Avian Influenza A (H7N7) Virus Epidemic in the Netherlands in 2003-Lessons Lea 48, 691–705. [DOI] [PubMed]
- Elving, J., Emmoth, E., Albihn, A., Vinnerås, B., Ottoson, J., 2012. Composting for Avian Influenza Virus Elimination 3280–3285. 10.1128/AEM.07947-11. [DOI] [PMC free article] [PubMed]
- Epstein E. Techonomic Publishing AG; Basel: 1997. The Science of Composting. [Google Scholar]
- Flory G., Peer R. Mesophilic static pile composting of animal carcasses. Biocycle. 2017:65–68. [Google Scholar]
- GAO, 2017. AVIAN INFLUENZA: USDA Has Taken Actions to Reduce Risks but Needs a Plan to Evaluate Its Efforts.
- Gelski, J., 2016. USDA detects bird flu in Indiana [WWW Document]. Food Bus. News. URL https://www.foodbusinessnews.net/articles/6941-u-s-d-a-detects-bird-flu-in-indiana (accessed 11.9.18).
- Gilroyed B.H., Conrad C., Hao X., Mcallister T.A., Stanford K., Reuter T. Composting for biocontained cattle mortality disposal and associated greenhouse gas and leachate emissions. J. Environ. Qual. 2016 doi: 10.2134/jeq2015.06.0314. [DOI] [PubMed] [Google Scholar]
- Glanville, T., Harmon, J., 2008. Composting for Emergency Disposal of Poultry and Livestock Mortalities [WWW Document]. Iowa State Univ. Ext. URL http://www.abe.iastate.edu/cattlecomposting/files/2013/05/Emergency-Mortality-Composting-Presentation.pdf (accessed 1.10.19).
- Glanville, T.D., 2008. Composting Swine Mortalities in Iowa Composting Swine Mortalities in Iowa [WWW Document]. Iowa State Univ. Ext. URL https://store.extension.iastate.edu/Product/Composting-Swine-Mortalities-in-Iowa-PDF (accessed 1.10.09).
- Glanville T.D., Ahn H., Akdeniz N., Crawford B.P., Koziel J.A. Performance of a plastic-wrapped composting system for biosecure emergency disposal of disease-related swine mortalities. Waste Manag. 2016;48:483–491. doi: 10.1016/j.wasman.2015.11.006. [DOI] [PubMed] [Google Scholar]
- Glanville T.D., Richard T.L., Ahn H.K., Richard T.L., Harmon J.D., Reynolds D.L., Akinc S. 2006 ASABE Annual International Meeting. St. Joseph, MI. 2006. Environmental impacts of emergency livestock mortality composting – leachate release and soil contamination (Paper Number: 064049) [Google Scholar]
- Guan J., Chan M., Brooks B.W., Spencer J.L., Algire J. Comparing Escherichia coli O157: H7 phage and bovine viral diarrhea virus as models for destruction of classical swine fever virus in compost. Compost. Sci. Util. 2012;20:18–23. [Google Scholar]
- Guan J., Chan M., Grenier C., Brooks B.W., Spencer J.L., Kranendonk C., Copps J., Clavijo A. Degradation of foot-and-mouth disease virus during composting of infected pig carcasses. Can. J. Vet. Res. 2010;74:40–44. [PMC free article] [PubMed] [Google Scholar]
- Guan J., Chan M., Grenier C., Wilkie D.C., Brooks B.W., Spencer J.L. Survival of avian influenza and newcastle disease viruses in compost and at ambient temperatures based on virus isolation and real-time reverse transcriptase PCR. Avian Dis. Dig. 2009;4 doi: 10.1637/8592.1. e8–e8. [DOI] [PubMed] [Google Scholar]
- Gwyther C.L., Williams A.P., Golyshin P.N., Edwards-Jones G., Jones D.L. The environmental and biosecurity characteristics of livestock carcass disposal methods: A review. Waste Manag. 2011;31:767–778. doi: 10.1016/j.wasman.2010.12.005. [DOI] [PubMed] [Google Scholar]
- Haug R.T. CRC Press; Boca Raton, FL: 1993. The Practical Handbook of Compost Engineering. [Google Scholar]
- Hayama Y., Kimura Y., Yamamoto T., Kobayashi S., Tsutsui T. Potential risk associated with animal culling and disposal during the foot-and-mouth disease epidemic in Japan in 2010. Res. Vet. Sci. 2015;102:228–230. doi: 10.1016/j.rvsc.2015.08.017. [DOI] [PubMed] [Google Scholar]
- Holtkamp, D., 2018. Lessons learned from PRRS outbreak investigations [WWW Document]. URL https://thepigsite.com/news/2017/11/lessons-learned-from-prrs-outbreak-investigations-1 (accessed 1.19.19).
- Holtkamp D.J., Kliebenstein J.B., Neumann E., Zimmerman J.J. Assessment of the economic impact of porcine reproductive and respiratory syndrome virus on United States pork producers. J. Swine Heal. Prod. 2013;21:72–84. [Google Scholar]
- Hseu Z.Y., Chen Z.S. Experiences of mass pig carcass disposal related to groundwater quality monitoring in Taiwan. Sustain. 2017;9:1–11. doi: 10.3390/su9010046. [DOI] [Google Scholar]
- Johnson K.K., Seeger R.M., Marsh T.L. Local economies and highly pathogenic avian influenza. Choices. 2016;31:1–10. [Google Scholar]
- Kalbasi A., Mukhtar S., Hawkins S.E., Auvermann B.W. Carcass composting for management of farm mortalities: A review. Compost Sci. Util. 2005;13:180–193. doi: 10.1080/1065657X.2005.10702239. [DOI] [Google Scholar]
- Keaten J.E., Hutchinson M. Efficacy and efficiency of poultry carcass composting using different mechanical mixing equipment for avian influenza outbreaks. Int. J. One Heal. 2017;3:19–27. doi: 10.14202/IJOH.2017.19-27. [DOI] [Google Scholar]
- Keener H.M., Elwell D.L., Monnin M.J. Procedures for sizing structures and windrows for composting animal mortalities. Appl. Eng. Agric. 2000;16:681–692. [Google Scholar]
- Keffaber K. Reproductive failure of unknown etiology. Am. Assoic. Swine Pr. Newsl. 1989;1:1–10. [Google Scholar]
- Ki B., Mi Y., Min J., Wook H., Cho K. Characterization of odor emissions and microbial community structure during degradation of pig carcasses using the soil burial-composting method. Waste Manag. 2018;77:30–42. doi: 10.1016/j.wasman.2018.04.043. [DOI] [PubMed] [Google Scholar]
- Kim S., Kwon H., Park S., Jeon H., Park J., Park J. Pilot-scale bio-augmented aerobic composting of excavated foot-and-mouth disease carcasses. Sustainability. 2017;9:1–13. doi: 10.3390/su9030445. [DOI] [Google Scholar]
- Ko C.R., Seol S.S., Kim G. Political response to foot-and-mouth disease: A review of Korean news. Sustain. 2017;9:463. doi: 10.3390/su9030463. [DOI] [Google Scholar]
- Koziel J.A., Ahn H., Glanville T.D., Frana T.S., Van Leeuwen J.H., Nguyen L.T. Lab-scale evaluation of aerated burial concept for treatment and emergency disposal of infectious animal carcasses. Waste Manag. 2018;76:715–726. doi: 10.1016/j.wasman.2018.03.009. [DOI] [PubMed] [Google Scholar]
- Koziel J.A., Frana T.S., Ahn H., Glanville T.D., Nguyen L.T., Van Leeuwen J.H. Efficacy of NH3 as a secondary barrier treatment for inactivation of Salmonella Typhimurium and methicillin-resistant Staphylococcus aureus in digestate of animal carcasses : Proof-of-concept. PLoS One. 2017;12:1–17. doi: 10.1371/journal.pone.0176825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee C., Suarez D.L., Tumpey T.M., Sung H., Kwon Y., Lee Y., Choi J., Joh S., Kim M., Lee E., Park J., Lu X., Katz J.M., Spackman E., Swayne D.E., Kim J. Characterization of highly Pathogenic H5N1 avian influenza A viruses isolated from South Korea. J. Virol. 2005;79:3692–3702. doi: 10.1128/JVI.79.6.3692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L., Zhao Q., Ge X., Teng K., Kuang Y., Chen Y., Guo X., Yang H. Chinese highly pathogenic porcine reproductive and respiratory syndrome virus exhibits more extensive tissue tropism for pigs. Virol. J. 2012;9:1. doi: 10.1186/1743-422X-9-203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linhares D.C.L., Torremorell M., Joo H.S., Morrison R.B. Infectivity of PRRS virus in pig manure at different temperatures. Vet. Microbiol. 2012;160:23–28. doi: 10.1016/j.vetmic.2012.05.009. [DOI] [PubMed] [Google Scholar]
- Malone G., Cloud S., Alphin R., Carr L., Tablante N. Proceedings of the 39th National Meeting on Poultry Health and Processing. Ocean City, MD. 2004. Delmarva in-house composting experiences; pp. 27–29. [Google Scholar]
- McKenna M. Bird flu cost the US $3.3 billion and worse could be coming. Natl. Geogr. Phenom. 2015 [Google Scholar]
- Nature Reports, 2018. Avian Flu Timelines - human cases of avian flu [WWW Document]. URL https://www.nature.com/avianflu/timeline/human_cases.html (accessed 6.9.18).
- NCBA-National Cattlemen’s Beef Association, 2018. Fact sheet: Industry economics [WWW Document].
- Nguyen, T., 2013. PRRS control in the region [WWW Document]. Conf.OIE. URL https://pdfs.semanticscholar.org/509e/f908651fb4d23e2fbb458999b35918e707c5.pdf (accessed 1.22.19).
- NPPC, 2014. NPCC wants focus on research, testing, biosecurity in USDA’s PEDV reporting plan [WWW Document]. URL http://nppc.org/nppc-wants-focus-on-research-testing-biosecurity-in-usdas-pedv-reporting-plan/ (accessed 1.15.19).
- NRAES, 1999. Field guide to on-farm composting. Natural Resource, Agriculture and Engineering Service, Ithaca, NY.
- NRCS, U., 2015. New Emergency Animal Mortality Management (368) Conservation Practice Standard and Highly Pathogenic Avian Influenza-Webinar [WWW Document]. URL http://www.conservationwebinars.net/webinars/emergency-animal-mortality-standard-and-hpai (accessed 1.15.19).
- Paarlberg, P.L., 2014. Updated estimated economic welfare impacts of porcine epidemic diarrhea virus (PEDV) (No. 14–4).
- Park, J., Lee, K., Ko, Y., Kim, S., Lee, H., Shin, Y., Sohn, H., Park, J., Yeh, J., Lee, Y., Kim, M., Joo, Y., Yoon, H., Yoon, S., Cho, I., 2013. Control of foot-and-mouth disease during 2010–2011 epidemic, South Korea, Emerg Infect Dis. 19, 655–659. [DOI] [PMC free article] [PubMed]
- Parry J. Death toll mounts in avian flu outbreak. BMJ. 2004;328:243. doi: 10.1136/bmj.328.7434.243-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasick J., Berhane Y., Hooper-McGrevy K. Avian influenza: the Canadian experience. Rev. Sci. Tech. 2009;28:349–358. doi: 10.20506/rst.28.1.1875. [DOI] [PubMed] [Google Scholar]
- Pendell D.L., Leatherman J., Schroeder T.C., Alward G.S. Western Agricultural Economics Association Annual Meeting. Portland, Oregon. 2007. The economic impacts of a foot-and-mouth disease outbreak: a regional analysis. [Google Scholar]
- Ramos, S., Maclachlan, M., Melton, A., 2017. Impacts of the 2014-2015 Highly Pathogenic Avian Influenza Outbreak on the U.S. Poultry Sector.
- Sánchez Ó.J., Ospina D.A., Montoya S. Compost supplementation with nutrients and microorganisms in composting process. Waste Manag. 2017;69:136–153. doi: 10.1016/j.wasman.2017.08.012. [DOI] [PubMed] [Google Scholar]
- Scudamore J.M., Harris D.M. Control of foot and mouth disease: lessons from the experience of the outbreak in Great Britain in 2001. Rev. Sci. Tech. 2002;21:699–710. doi: 10.20506/rst.21.3.1351. [DOI] [PubMed] [Google Scholar]
- Scudamore J.M., Trevelyan G.M., Tas M.V., Varley E.M., Hickman a W G. Carcass disposal: lessons from Great Britain following the foot and mouth disease outbreaks of 2001. Rev. Sci. Tech. 2002;21:775–787. doi: 10.20506/rst.21.3.1377. [DOI] [PubMed] [Google Scholar]
- Senne D.A., Panigrahy B., Morgan R.L. Effect of composting poultry carcasses on survival of exotic avian viruses: highly pathogenic avian influenza (HPAI) virus and adenovirus of egg drop syndrome-76. Avian Dis. 1994;38:733–737. doi: 10.2307/1592108. [DOI] [PubMed] [Google Scholar]
- Song D., Moon H., Kang B. Porcine epidemic diarrhea: a review of current epidemiology and available vaccines. Clin. Exp. Vaccine Res. 2015;4:166. doi: 10.7774/cevr.2015.4.2.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spackman E., Pantin-jackwood M.J., Kapczynski D.R., Swayne D.E., Suarez D.L. H5N2 Highly Pathogenic Avian Influenza Viruses from the US 2014–2015 outbreak have an unusually long pre-clinical period in turkeys. BMC Vet. Res. 2016;1–9 doi: 10.1186/s12917-016-0890-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer J.L., Rennie B., Guan J. Emphasis on biosecurity for composting poultry and manure during an outbreak of highly pathogenic avian influenza in British Columbia. Can. Anim. Heal. Bull. 2004:21–23. [Google Scholar]
- Suyama T., Kawaharasaki M. Decomposition of waste DNA with extended autoclaving under unsaturated steam. Biotechniques. 2013;55:296–299. doi: 10.2144/000114113. [DOI] [PubMed] [Google Scholar]
- Swayne, D., Akey, B., 2005. Avian influenza control strategies in the United States of America. Proc. Wageningen Front. Int. Work. Avian Influenza Prev. Control 8, 113.
- Tiensin Thanawat, Chaitaweesub Prasit, Thaweesak Songserm A.C., Hoonsuwan Wirongrong, Buranathai Chantanee, Tippawon Parakamawongsa S.P., Amonsin Alongkorn, Gilbert Marius, Mirjam Nielen A.S. Emerg. Infect; Dis: 2005. Highly Pathogenic Avian Influenza H5N1, Thailand, 2004; p. 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The Pig Site, 2019. African swine fever death toll exceeds 900,000 pigs in China [WWW Document]. URL http://www.thepigsite.com/swinenews/45866/african-swine-fever-death-toll-exceeds-900000-pigs-in-china/ (accessed 1.22.19).
- Tkachuk V.L., Krause D.O., Mcallister T.A., Buckley K.E., Reuter T., Hendrick S., Ominski H. Assessing the inactivation of mycobacterium avium subspecies paratuberculosis during composting of livestock carcasses. Appl. Environ. Microbiol. 2013;79:3215–3224. doi: 10.1128/AEM.03768-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner C., Williams S.M., Burton C.H., Farrent J.W., Wilkinson P.J. Laboratory scale inactivation of pig viruses in pig slurry and design of a pilot plant for thermal inactivation. Water Sci. Technol. 1998;38:79–86. doi: 10.1016/S0273-1223(98)00500-9. [DOI] [Google Scholar]
- USDA, 2017. Livestock Mortality Composting Protocol.
- USDA, 2013. Porcine Epidemic Diarrhea (PED) [WWW Document]. URL https://www.usda.gov/sites/default/files/documents/pedv-summary-actions.pdf (accessed 2.1.19).
- USEPA, 2018. Carcass Management During Avian Influenza Outbreaks [WWW Document]. United States Environ. Prot. Agency. URL https://www.epa.gov/homeland-security-waste/carcass-management-during-avian-influenza-outbreaks#main-content (accessed 6.9.18).
- USEPA, 2003. Environmental Regulations and Technology Control of Pathogens and Vector Attraction in Sewage Sludge Control of Pathogens and Vector Attraction. Cincinnati, OH.
- Vitosh-Sillman S., Loy J.D., Brodersen B., Kelling C., Eskridge K., Millmier Schmidt A., Schmidt A.M., Millmier Schmidt A., Schmidt A.M. Effectiveness of composting as a biosecure disposal method for porcine epidemic diarrhea virus (PEDV)-infected pig carcasses. Porc. Heal. Manag. 2017;3:22. doi: 10.1186/s40813-017-0068-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner, A., Choi, J., 2011. South Korea Foot-and-Mouth: Over a million animals culled [WWW Document].
- Weng L., Weersink A., Poljak Z., de Lange K., von Massow M. An economic evaluation of intervention strategies for Porcine Epidemic Diarrhea (PED) Prev. Vet. Med. 2016;134:58–68. doi: 10.1016/j.prevetmed.2016.09.018. [DOI] [PubMed] [Google Scholar]
- WHO, 2018a. Influenza (Avian and other zoonotic) [WWW Document]. URL https://www.who.int/news-room/fact-sheets/detail/influenza-(avian-and-other-zoonotic)) (accessed 1.17.19).
- WHO, 2018b. Cumulative number of confirmed human cases for avian influenza A (H5N1) reported to WHO , 2003-2018 [WWW Document]. URL https://www.who.int/influenza/human_animal_interface/2018_12_13_tableH5N1.pdf?ua=1 (accessed 1.17.19).
- WHO, 2003. Emergencies preparedness, response. Avian influenza in the Netherlands [WWW Document]. URL http://www.who.int/csr/don/2003_04_24/en/ (accessed 6.9.18).
- Wilkinson K., Grant W.P., Green L.E., Hunter S., Jeger M.J., Lowe P., Medley G.F., Mills P., Phillipson J., Poppy G.M., Waage J. Infectious diseases of animals and plants : an interdisciplinary approach. Phil. Trans. R. Soc. B. 2011;366:1933–1942. doi: 10.1098/rstb.2010.0415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson K.G. The biosecurity of on-farm mortality composting. J. Appl. Microbiol. 2007;102:609–618. doi: 10.1111/j.1365-2672.2006.03274.x. [DOI] [PubMed] [Google Scholar]
- Windhorst H.-W. Avian Influenza Outbreaks in the USA in 2014 and 2015. Lohmann Tierzucht. 2015;50(2) [Google Scholar]
- Xu S., Reuter T., Gilroyed B.H., Tymensen L., Hao Y., Hao X., Belosevic M., Leonard J.J., McAllister T.A. Microbial communities and greenhouse gas emissions associated with the biodegradation of specified risk material in compost. Waste Manag. 2013;33:1372–1380. doi: 10.1016/j.wasman.2013.01.036. [DOI] [PubMed] [Google Scholar]
- Xu W., Reuter T., Inglis G.D., Larney F.J., Alexander T.W., Guan J., Stanford K., Xu Y., McAllister T.A. A biosecure composting system for disposal of cattle carcasses and manure following infectious disease outbreak. J. Environ. Qual. 2009;38:437. doi: 10.2134/jeq2008.0168. [DOI] [PubMed] [Google Scholar]
- Xu W., Reuter T., Xu Y., Hsu Y., Stanford K., Mcallister T.A. Bioresource Technology Field scale evaluation of bovine-specific DNA as an indicator of tissue degradation during cattle mortality composting. Bioresour. Technol. 2011;102:4800–4806. doi: 10.1016/j.biortech.2011.01.037. [DOI] [PubMed] [Google Scholar]
- Xu W., Xu Y., Jin L., Xu W., Stanford K., Xu W., Reuter T., Gilroyed B., Larney F.J., McAllister T.A., Gilroyed B. An improved design for biocontained composting of cattle mortalities. Compost Sci. Util. 2010;18:32–41. doi: 10.1080/1065657X.2010.10736931. [DOI] [Google Scholar]