Abstract
Twenty traditional Chinese medicines (TCM) were evaluated for their antimicrobial activity against four common oral bacteria. TCMs were tested for sensitivity against Streptococcus mitis, Streptococcus sanguis, Streptococcus mutans and Porphyromonas gingivalis. Aliquots of suspension of each bacterial species were inoculated onto a horse blood agar plate with TCMs soaked separately on 6 mm paper disks. The plates were incubated for 48 h anaerobically and the mean diameters of growth inhibition of three different areas obtained. 0.2% (w/v) chlorhexidine was used as a positive control. Broth microdilution assay was used to determine minimum inhibitory concentration and minimum bactericidal concentration. Fructus armeniaca mume was effective against all four bacteria. Thirteen TCMs demonstrated antimicrobial activity against Porphyromonas gingivalis, including Cortex magnoliae officinalis, Cortex phellodendri, Flos caryophylli, Flos lonicerae japonicae, Fructus armeniaca mume, Fructus forsythiae suspensae, Herba cum radice violae yedoensitis, Herba menthae haplocalycis, Pericarpium granati, Radix et rhizoma rhei, Radix gentianae, Ramulus cinnamomi cassia and Rhizoma cimicifugae. Cortex phellodendri showed antimicrobial activity against Streptococcus mutans, while Radix et rhizoma rhei was effective against Streptococcus mitis and Streptococcus sanguis. Fructus armeniaca mume had inhibitory effects against Streptococcus mitis, Streptococcus sanguis, Streptococcus mutans and Porphyromonas gingivalis in vitro.
Key words: antimicrobial effect, Chinese medicine, bacteria, oral biofilm
The oral cavity contains a variety of bacteria that may cause dental caries, periodontal disease, postoperative infections after surgery or trauma or superinfections in immunocompromised patients. As new infections and bacterial resistance are constantly emerging, the search for novel drugs or chemicals to combat oral infection is always warranted.
Oral bacteria usually aggregate to form a biofilm or plaque on tooth surfaces. Plaque formation involves initial colonization and multiplication by pioneer species, followed by secondary colonization by other species and finally becoming a climax community1. The ‘pioneer species’ of plaque are Streptococcus oralis, Streptococcus mitis and Streptococcus sanguis. Specific bacteria are also closely related to specific dental diseases, for example, Streptococcus mutans and Porphyromonas gingivalis are associated with dental caries and periodontal disease, respectively11.
Traditional Chinese medicines (TCM) have been used in China to treat infectious diseases for more than 4000 years. In the past decade, many new pathogenic microorganisms emerged and there were alterations to existing infections, such as Severe Acute Respiratory Syndrome (SARS), which was difficult to control and caused significant mortality. There is an urgent need to screen for natural products to identify a bank of antimicrobial materials that may be used against these infections, either alone or in combination. The use of natural products may decrease the opportunity for the development of drug resistance in microorganisms as they are surrounded by a group of antimicrobial chemicals acting together. In the oral cavity, a number of pathogenic organisms cause caries and periodontal diseases. Oral bacteria provide a good starting point for investigating the antimicrobial activities of TCM because the oral cavity is a strategic place for the development of infective disease and in vivo and clinical studies are easy to carry out. Unlike Western medicine, TCM works as a formula of herbs that is tailored to the individual patient and their specific condition. They are designed using one or two main ingredients that target the illness, with the addition of many other ingredients to adjust the formula to suit the patient's condition4. The advantage of using several TCMs together can also be considered a disadvantage because the treatment does not consist of a single drug. Some TCM formulae have been used effectively. One example is Realgar-Indigo naturalis for the treatment for promyelocytic leukaemia; a recent clinical trial showed a complete remission rate of 97%8. The mechanism of action has recently been investigated at the molecular, cellular and organism levels14. TCMs possess a variety of biological properties and can be developed to produce many effective drugs. Certain TCMs have antibacterial properties16. Few studies have been performed to screen these TCMs to see whether they are effective against bacteria forming an oral biofilm.
All TCMs consist of natural products, some of which are cheap and easy to obtain, although others come from endangered animal and plant species. If any TCMs are effective in inhibiting the bacteria commonly found in the oral biofilm, they might reduce the oral biofilm and aid in the prevention of dental caries and periodontal disease. They could have an extensive impact on the prevention of oral disease and improve oral health.
Existing antimicrobial mouth rinses, such as chlorhexidine and Listerine®, are inedible and may be harmful if ingested. There is a need to search for less harmful agents that can produce a similar antimicrobial effect.
Chlorhexidine gluconate is one of the most common conventional oral antimicrobial agents. It is effective against a broad spectrum of bacteria, yet it is artificial and inedible. Owing to overuse of antimicrobial agents, some bacteria start to develop resistance towards them. It is therefore necessary to develop new antimicrobial agents to replace or enrich the existing choice of oral antimicrobial agents. The potential advantage of using TCM rather than artificial oral antimicrobial agents is that they are natural substances and some are edible. It may also allow the combination of different TCM rather than a single oral antimicrobial agent, which lowers the chance of cross-resistance in bacteria. Further research is needed to confirm this. A number of TCM herbs are already used in oral healthcare products, such as toothpaste, to improve oral health, which shows that TCM has great potential to be developed as a product for day-to-day use.
In this study, the authors investigate the antimicrobial activity of some TCMs that have been used to treat symptoms related to infection (e.g. fever, inflammation, cough) on several bacteria found in the oral biofilm to identify potential agents to control oral infections and diseases.
Methods
Organism and culture condition
Type cultures of S. mitis (ATCC 15914), S. sanguis (ATCC 10556), S. mutans (ATCC 35668) and P. gingivalis (ATCC 33277) from the Oral BioScience Laboratories of the Faculty of Dentistry, the University of Hong Kong, were used in the study16. Frozen isolates were thawed and their identity reconfirmed using standard methodology. They were inoculated onto horse blood agar (HBA) and incubated for 3 days at 37 °C. The bacteria cultures were harvested and suspended in phosphate buffered saline at a concentration of 1 × 106 cells/ml (0.5 MacFarland Standard Units) for sensitivity studies.
Identification and preparation of TCM extracts
TCM were purchased from local Chinese medicine store and were identified morphologically, histologically and chemically using standard Chinese herbal identification procedures3.
Voucher specimens including the identification and classification of all the TCMs to be tested were stored in the Hard Tissue Laboratory, University of Hong Kong. TCM extracts were prepared using standard protocols for aqueous extraction5. Briefly, 4 ml distilled water was added to 10 g of TCM powder. The mixture was boiled under stirring on a hot plate for 4 h. Distilled water was added occasionally to prevent drying. At the end of boiling, distilled water was added to make up the volume of the mixture to 4 ml. The mixture was cooled, centrifuged and filtered. This produced 2.5 g/ml of one TCM extract.
The 20 TCM drugs chosen for the study were Cortex magnoliae officinalis (hou po), Cortex phellodendri (huang bai), Flos caryophylli (ding xiang), Flos chrysanthemi morifolii (ju hua), Flos lonicerae japonicae (jin yin hua), Fructus armeniaca mume (wu mei), Fructus forsythiae suspensae (lian qiao), Herba cum radice asari (xi xin), Herba cum radice houttuyniae cordatae (yu xing cao), Herba cum radice violae yedoensitis (zi hua di ding), Herba ephedrae (ma huang), Herba portulacae oleraceae (ma chi xian), Herba menthae haplocalycis (bo he), Pericarpium granati (shi liu pi), Radix et rhizoma rhei (da huang), Radix gentianae (long dan cao), Radix isatidis (ban lan gan), Ramulus cinnamomi cassia (gui zhi), Rhizoma cimicifugae (sheng ma) and Semen raphani (lai fu zi). Chlorhexidine gluconate, a common oral antiseptic, at a concentration of 0.2% (w/v) was used as a positive control for all experiments.
Agar diffusion assay
The standard agar diffusion assay for sensitivity testing9, 10 was performed according to a standard protocol designed by Samaranayake et al.12. Briefly, 20 μl aliquots of suspension of each bacterial species were inoculated on an HBA plate using a glass rod, then 6 mm diameter paper disks soaked in 10 μl of each of the TCM extracts at a concentration of 2.5 g/ml were placed concentrically on the HBA plate. Disks soaked in 10 μl of 0.2% (w/v) chlorhexidine were used as positive controls and were also placed in the HBA plates. The HBA plates were incubated for 48 h anaerobically at 37 °C. Naked eye measurements of the growth inhibition zone, if any, were evaluated using calibrated computer software. The diameters of growth inhibition in three different areas were measured and the mean diameter of growth inhibition was calculated for each organism. The experiment was repeated on three separate occasions.
Broth microdilution susceptibility test
The TCMs, Rhizoma coptidis and Galla chinensis, which showed potent antimicrobial activity against the four tested bacteria in the agar diffusion assay screening test, were selected for minimum inhibitory concentration (MIC) determination using the standard broth microdilution assay2, 6, 7.
In brief, bacteria were cultured anaerobically in HBA for 3–7 days at 37 °C. The bacterial cultures were harvested and suspended in an appropriate medium (depending on the species) at a concentration of 1 × 106 cells/ml (0.5 MacFarland Standard Units). Broth microdilution assay was performed in polystyrene, 96-well microtiter plates as previously described13. Each plate included a positive control (bacteria without the drug) and negative control (medium only) and serial two-fold dilution of the drug. P. gingivalis suspensions were prepared in ‘P. gingivalis broth’ whereas other bacterial species were prepared in brain heart infusion broth. Bacterial inoculums were prepared as described previously and 100 μl of 1 × 106 cells/ml bacterial suspensions were added to each well. Subsequently, microtiter plates were incubated for 48 h at 37 °C in an anaerobic chamber. Visual inspection was used to determine the MIC. After MIC determination, 20 μl of suspension from each well was inoculated in blood agar plates for 48 h for observation. The lowest drug concentration that yielded no growth was documented as minimum bactericidal concentration (MBC). For P. gingivalis all the procedures took the slow growth of the organism into account.
Results
TCMs that passed all three tests are classified as TCMs that have an inhibitory effect towards specific bacteria. Those that showed only a weak inhibitory effect in one or two tests, but not all, are classified as partially effective in the inhibition of specific bacteria.
Initial biofilm forming bacteria
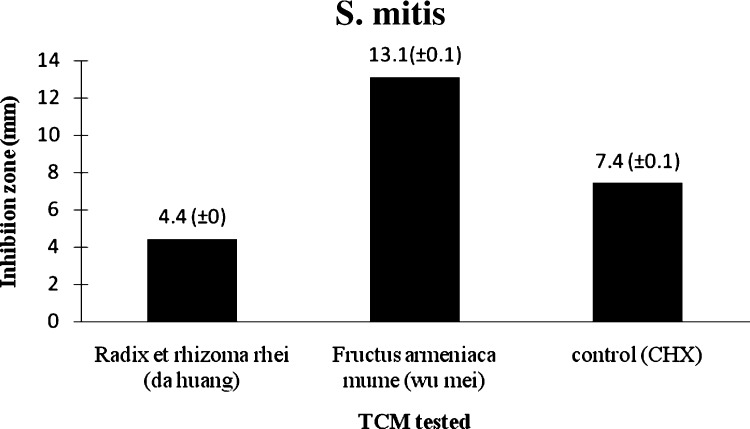
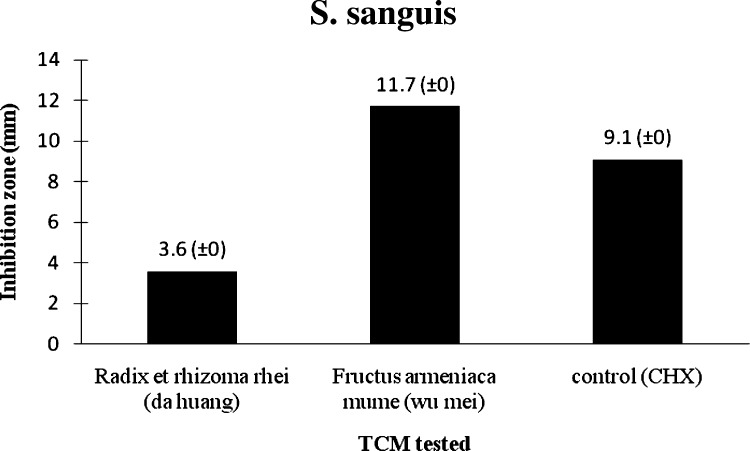
The inhibition zone measured for chlorhexidine against S. mitis was 7.4 mm compared with those of Radix et rhizoma rhei (da huang) and Fructus armeniaca mume (wu mei) which were 4.4 mm and 11.3 mm, respectively (Table 1 ; Fig. 1, Fig. 5 ). The average inhibition zone diameter measured for chlorhexidine against S. sanguis was 9.1 mm, compared with 3.6 mm and 11.7 mm for Radix et rhizoma rhei (da huang) and Fructus armeniaca mume (wu mei), respectively (Table 1; Fig. 2, Fig. 5 ).
Table 1.
Sensitivity of bacteria to the TCM tested.
| S. mutans | S. mitis | S. sanguis | P. gingivalis | |
|---|---|---|---|---|
| Cortex magnoliae officinalis (hou po) | – | – | – | 4.3 (±0.2)* |
| Cortex phellodendri (huang bai) | 8.0 (±0.1)* | - | P | 38.3 (±0.3)* |
| Flos caryophylli (ding xiang) | – | - | - | 9.8 (±0)* |
| Flos chrysanthemi morifolii (ju hua) | – | - | P | P |
| Flos lonicerae japonicae (jin yin hua) | – | – | P | 5.5 (±0.4)* |
| Fructus armeniaca mume (wu mei) | 9.0 (±0)* | 13.1(±0.1)* | 11.7 (±0)* | 10.6 (±0.1)* |
| Fructus forsythiae suspensae (lian qiao) | – | – | – | 7.4 (±0.1)* |
| Herba cum radice asari (xi xin) | – | – | – | – |
| Herba cum radice houttuyniae cordatae (yu xing cao) | – | – | P | – |
| Herba cum radice violae yedoensitis (zi hua di ding) | – | – | - | 9.0 (±0)* |
| Herba ephedrae (ma huang) | – | – | P | P |
| Herba portulacae oleraceae (ma chi xian) | – | – | – | – |
| Herba menthae haplocalycis (bo he) | – | – | – | 2.3 (±0.3)* |
| Pericarpium granati (shi liu pi) | – | – | – | 12.9 (±0)* |
| Radix et rhizoma rhei (da huang) | – | 4.4 (±0)* | 3.6(±0)* | 31.7 (±0.1)* |
| Radix gentianae (longdancao) | – | – | – | 11.4 (±0.1)* |
| Radix isatidis (ban lan gan) | – | – | – | - |
| Ramulus cinnamomi cassiae (gui zhi) | – | – | – | 8.5 (±0)* |
| Rhizoma cimicifugae (sheng ma) | – | – | – | 13.0 (±0.2)* |
| Semen raphani (lai fu zi) | – | – | – | – |
| Control | 12.1 (±0)* | 7.4 (±0.1)* | 9.1 (±0)* | 23.7 (±0.1)* |
(P) Stands for partial inhibitory effect.
(–) Indicates for no inhibitory effect.
Values in parenthesis indicate standard deviation
Indicate significant differences (p < 0.05).
Fig. 1.
Sensitivity of S. mitis to two out of 20 TCM tested.
Fig. 5.
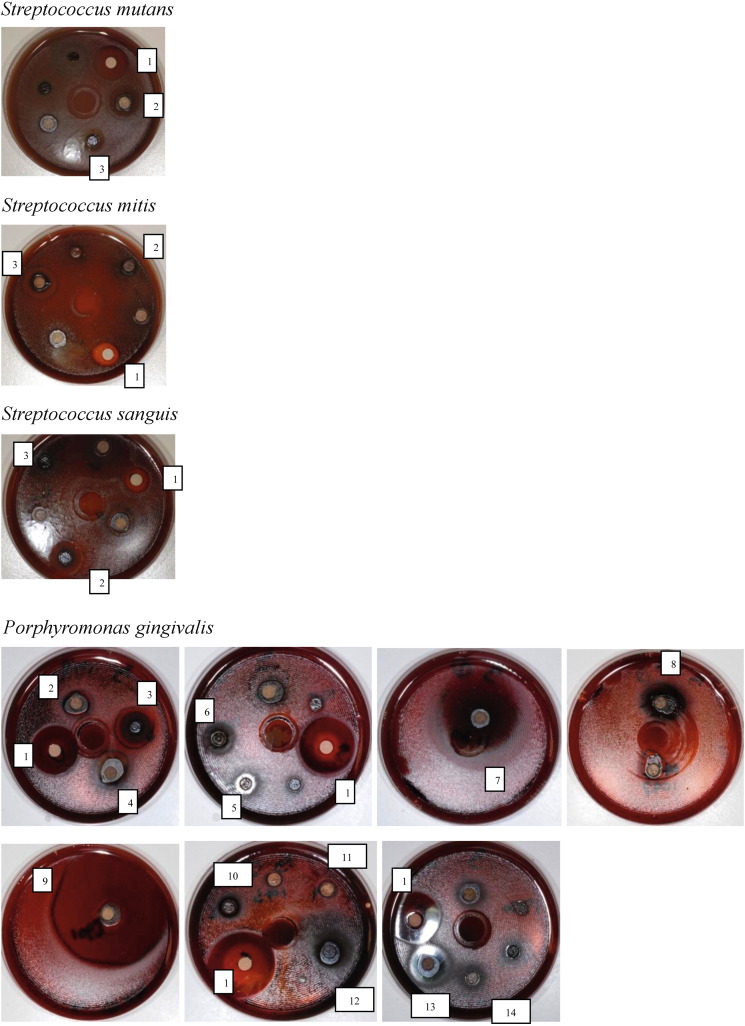
Results for all bacteria. S. mutans: (1) Chlorhexidine, (2) Cortex phellodendri, (3) Fructus armeniaca mume. S. mitis: (1) Chlorhexidine, (2) Radix et rhizoma rhei, (3) Fructus armeniaca mume. S. sanguis: (1) Chlorhexidine, (2) Fructus armeniaca mume, (3) Radix et rhizoma rhei. P. gingivalis: (1) Chlorhexidine, (2) Cortex magnoliae officinalis, (3) Fructus armeniaca mume, (4) Radix gentianae longdancao, (5) Ramulus cinnamomi cassiae, (6) Herba cum radice violae yedoensitis, (7) Radix et rhizoma rhei, (8) Rhizoma cimicifugae, (9) Cortex phellodendri, (10) Flos lonicerae japonicae, (11) Cortex magnoliae officinalis, (12) Fructus forsythiae suspensae, (13) Pericarpium granati, (14) Herba menthae haplocalycis.
Fig. 2.
Sensitivity of S. sanguis to two out of 20 TCM tested.
Radix et rhizoma rhei (da huang) demonstrated a comparable effect to chlorhexidine against S. mitis, while Fructus armeniaca mume (wu mei) showed signs of a stronger effect than chlorhexidine (9.1 mm) against S. mitis and S. sanguis.
Caries causing bacteria
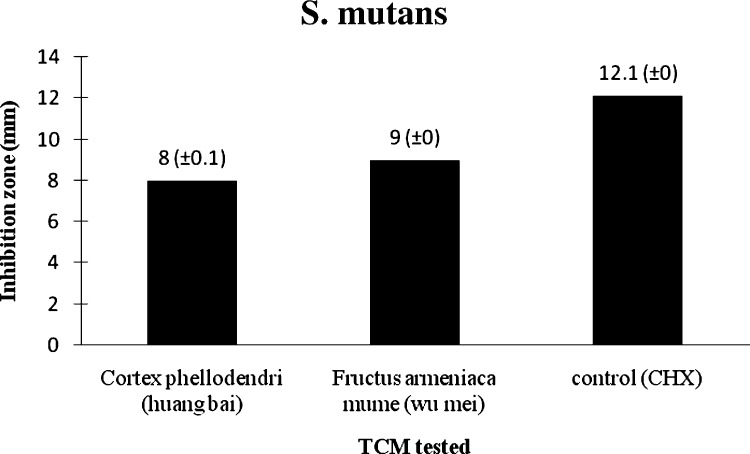
Two of 20 TCM extracts tested demonstrated consistent antimicrobial activity with zones of growth against S. mutans. The average inhibition zone diameter for Cortex phellodendri (huang bai) and Fructus armeniaca mume (wu mei) to S. mutans, was 8 mm and 9 mm, respectively (Table 1; Fig. 3, Fig. 5 ).
Fig. 3.
Sensitivity of S. mutans to two out of 20 TCM tested.
Periodontal disease causing bacteria
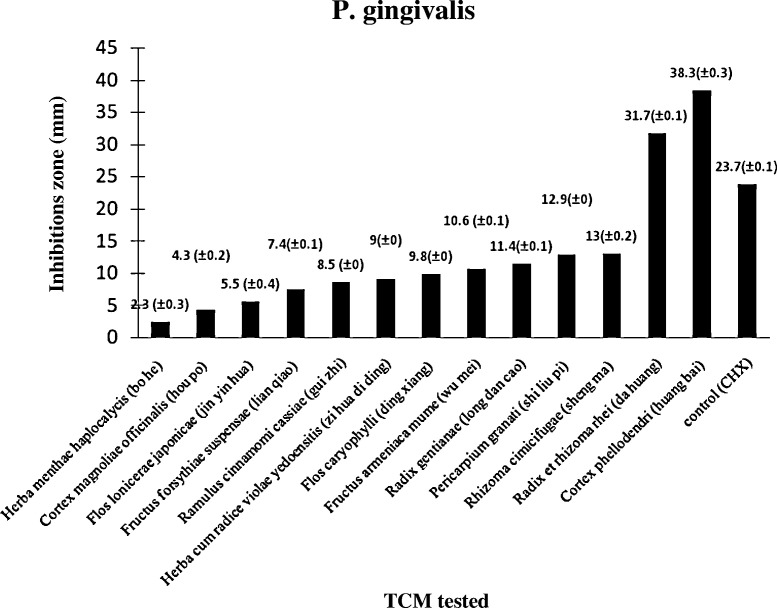
Thirteen of the 20 drugs demonstrated consistent antimicrobial activity with zones of growth inhibition ranging from 2 to 39 mm against P. gingivalis. The sequences of drug in ascending orders of effectiveness towards P. gingivalis were Herba menthae haplocalycis (bo he) (2.3 mm), Cortex magnoliae officinalis (hou po) (4.3 mm), Flos lonicerae japonicae (jin yin hua) (5.5 mm), Fructus forsythiae suspensae (lian qiao) (7.4 mm), Ramulus cinnamomi cassia (gui zhi) (8.5 mm), Herba cum radice violae yedoensitis (zi hua di ding) (9.0 mm), Flos caryophylli (ding xiang) (9.8 mm), Fructus armeniaca mume (wu mei) (10.6 mm) and Radix gentianae (long dan cao) (11.4 mm), Pericarpium granati (shi liu pi) (12.9 mm), Rhizoma cimicifugae (sheng ma) (13.0 mm), Radix et rhizoma rhei (da huang) (31.7 mm) and Cortex phellodendri (huang bai) (38.3 mm).
Radix et rhizoma rhei (da huang) (31.7 mm) and Cortex phellodendri (huang bai) (38.3 mm) show a significant antimicrobial effect compared with chlorhexidine (23.7 mm) against P. gingivalis (Table 1; Fig. 4, Fig. 5 ).
Fig. 4.
Sensitivity of P. gingivalis to 13 out of 20 TCM tested.
Partial effects
For the remaining TCMs, Cortex phellodendri (ding xiang), Flos chrysanthemi morifolii (ju hua), Flos lonicerae japonicae (jin yin hua), Herba cum radice houttuyniae cordatae (yu xing cao) and Herba ephedrae (ma huang) showed weak antimicrobial effects against S. sanguis. While Flos chrysanthemi morifolii (ju hua) and Herba ephedrae (ma huang) demonstrated weak antimicrobial effects against P. gingivalis (Table 1).
Statistical analysis
Data were analyzed with statistical analysis computer software (SPSS 15.0 for Windows©, Chicago, USA). A one-way ANOVA was used to compare the effects of different TCMs. Differences were considered significant when the p-value was less than 0.05. All the TCM results that were shown to be effective against individual bacteria in this experiment proved to be significant (Table 1).
MIC
The MIC values for Fructus armeniaca mume (wu mei) against S. mutans, S. mitis and S. sanguis were 0.0781 g/ml. The MIC value against P. gingivalis was 0.0003 g/ml (Table 2 ). The MBC values for Fructus armeniaca mume (wu mei) against S. mutans and S. mitis were 0.1563 g/ml. Its MBC value against S. sanguis was 0.0781 g/ml and 0.0003 g/ml against P. gingivalis (Table 2).
Table 2.
MIC and MBC of Fructus armeniaca mume (wu mei) against oral microorganisms.
| Species | MIC (g/ml) | MBC (g/ml) |
|---|---|---|
| S. mutans | 0.0781 | 0.1563 |
| S. mitis | 0.0781 | 0.1563 |
| S. sanguis | 0.0781 | 0.0781 |
| P. gingivalis | 0.0003 | 0.0003 |
Discussion
In this study, 20 TCM extracts were evaluated for their antimicrobial activities against four common bacterial species present in the oral cavity, which are important in biofilm formation (S. mitis, S. sanguis), as a cause of dental caries (S. mutans) or as a cause of periodontal disease (P. gingivalis).
This research showed that some TCM are active against oral bacteria. This is one of the first studies to show this possible association. Further research is needed to substantiate this and to evaluate the principles of TCM when applied to antibacterial treatment.
Fructus armeniaca mume (wu mei) was shown to be very effective against all four bacteria; this study is one of the first to show its effects specifically on oral bacteria. Fructus armeniaca mume (wu mei), also known as Prunus mume or Japanese Apricot, is a common fruit in Asia, which is eaten in its raw form and used as a herbal medicine after it has been cooked and aged. As a TCM, Fructus armeniaca mume (wu mei) is taken internally to relieve cough and used externally for the removal of warts and corns.15 Its antibacterial effect on some bacteria is known, but no research has been carried out on its action against oral bacteria. It has low toxicity compared with inedible oral antiseptic mouth rinses (e.g. chlorhexidine), costs little and is widely available. If it is effective in inhibiting the bacteria commonly found in the oral biofilm, it may be a safe agent for the effective reduction of the oral biofilm and the prevention of dental caries and periodontal disease.
The authors compared the antimicrobial activity of TCMs with one effective oral antiseptic, chlorhexidine. The authors showed that they have comparable effects. This research is the first study that investigates TCMs specifically on oral bacteria. TCMs are promising agents in the development of new antibacterials for oral microorganisms. This study is a key step in the discovery of new drugs/treatments using TCM. Further research is needed to identify the active components related to the antibacterial action, to determine the range of action, and to investigate the mechanisms involved. The antibacterial actions include the MIC and MBC. It is also necessary to test the antibacterial action on other oral bacteria and fungi. TCM extracts that are less effective against oral bacteria, have potential for further investigation because the active components inside the extracts may only present in very low concentration, so although they may be very effective, the overall antibacterial effect of the extracts is small.
Further research will focus on four strategies. The active components in the antimicrobial action of Fructus armeniaca mume should be identified and the mechanisms investigated. It contains a number of acids (e.g. citric acid, malic acid, oxalic acid, succinic acid, fumaric acid, tartaric acid, picric acid); these might create a low pH environment that may account for its antibacterial effect. Several herbs that have an antibacterial action can be combined. This may allow more effective control of microorganisms, and it may be possible to combat the newly emerging resistant microorganisms. TCM with similar properties to Fructus armeniaca mume should be screened for their antimicrobial actions. In vivo and clinical studies should be carried out to optimize the clinical use of Fructus armeniaca mume in mouth rinses, toothpaste, chewing gum and other oral products.
In this study, Fructus armeniaca mume was found active against all four oral bacteria. This result is unexpected as Fructus armeniaca mume is used for cough suppression in TCM. This shows that only investigating herbs with strong suggestions of antimicrobial action is not enough as some important sources of antibicrobial action may be missed. In conclusion, Fructus armeniaca mume had inhibitory effects on S. mitis, S. sanguis, S. mutans and P. gingivalis in vitro. It is possible that its acidic content accounts for its wide antibacterial spectrum.
Funding
This study was supported by the University Research Grant no.: 10207346.15633.08003.323.01, the University of Hong Kong.
Competing interests
The author(s) declare that they have no competing interests.
Ethical approval
Not required.
Authors’ contributions
MY performed TCM collection, method development, validation, result analysis and manuscript drafting. CS performed MIC and MBC data collection. RW and UH participated in the design of the study, selection of TCM for testing and manuscript drafting. RW, UH and LS conceived the idea of the study and participated in its design and coordination. All authors approved the final manuscript.
Acknowledgement
The authors thank Ms. J. Yau on her technical assistance.
References
- 1.Auschill T.M., Hein N. Effect of two antimicrobial agents on early in situ biofilm formation. J Clin Periodontol. 2005;32:147–152. doi: 10.1111/j.1600-051X.2005.00650.x. [DOI] [PubMed] [Google Scholar]
- 2.Banaszczak E.W., Bylka W., Matawska I., Goœliñska O., Muszyñski Z. Antimicrobial activity of Viola tricolor herb. Fitoterapia. 2005;76:458–461. doi: 10.1016/j.fitote.2005.03.005. [DOI] [PubMed] [Google Scholar]
- 3.Blaszczyk T., Krzyzanowska J., Lamer-Zarawska E. Screening for antimycotic properties of 56 traditional Chinese drugs. Phytother Res. 2000;14:210–212. doi: 10.1002/(sici)1099-1573(200005)14:3<210::aid-ptr591>3.0.co;2-7. [DOI] [PubMed] [Google Scholar]
- 4.Eisenberg D.M. Trends in alternative medicine use in the United States. JAMA. 1998;280:1569–1575. doi: 10.1001/jama.280.18.1569. [DOI] [PubMed] [Google Scholar]
- 5.Hu J.P., Takahashi N., Yamada T. Coptidis Rhizoma inhibits growth and proteases of oral bacteria. Oral Dis. 2000;6:297–302. doi: 10.1111/j.1601-0825.2000.tb00142.x. [DOI] [PubMed] [Google Scholar]
- 6.Katsura H., Tsukiyama R.I., Suzuki A., Kobayashi M. In vitro antimicrobial activities of Bakuchiol against oral microorganisms. Antimicrob Agents Chemother. 2001:3009–3013. doi: 10.1128/AAC.45.11.3009-3013.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Levison M. Antibacterial therapy: in vitro testing, pharmaco-dynamics, pharmacology, new agents. Infect Dis Clin North Am. 1995;9:483–495. [PubMed] [Google Scholar]
- 8.Liu Y.F. Long term follow-up confirms the benefit of all-trans retinoic acid and arsenic trioxide as front line therapy for newly diagnosed acute promyelocytic leukaemia. Blood. 2006;88:1052–1061. [Google Scholar]
- 9.Ogundare A.O., Olorunfemi O.B. Antimicrobial flavonids from the stem bark of Erythrina burtii. Fitoterapia. 2005;76:469–472. doi: 10.1016/j.fitote.2005.04.006. [DOI] [PubMed] [Google Scholar]
- 10.Rahman M.M., Parvin S., Haque M.E., Islam M.E., Mosaddik M.A. Antimicrobial and cytotoxic constituents from seeds of Annona squamosa. Fitoterapia. 2005;76:484–489. doi: 10.1016/j.fitote.2005.04.002. [DOI] [PubMed] [Google Scholar]
- 11.Samaranayake L.P. 3rd ed. Churchill Livingstone; 2006. Essential Microbiology for Dentistry. p 34. [Google Scholar]
- 12.Samaranayake Y.H., Ye J., Yau J.Y.Y., Cheung B.P.K., Samaranayake L.P. In vitro method to study antifungal perfusion in Candida biofilms. JCM. 2005;43:818–825. doi: 10.1128/JCM.43.2.818-825.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tsui V.W.K., Wong R.W.K., Rabie A.B.M. The inhibitory effects of naringin on the growth of periodontal pathogens in vitro. Phytother Res. 2008;22:401–406. doi: 10.1002/ptr.2338. [DOI] [PubMed] [Google Scholar]
- 14.Wang L., Zhou G.B., Liu P., Song J.H., Liang Y., Uan X.J., Xu F., Wang B.S., Mao J.H., Shen Z.X., Chen S.J., Chen Z. Dissection of mechanisms of Chinese medicinal formula Realgar-Indigo naturalis as an effective treatment for promyelocytic leukemic. PNAS. 2008;105:4826–4831. doi: 10.1073/pnas.0712365105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xiang P. 2nd ed. Shanghai Scientific Technology Publisher; Shanghai: 2006. Zhongyao Dacidian. pp. 657. [Google Scholar]
- 16.Zhu Y.P. Harwood Acedemic Publishers; 1998. Chinese Materia Medica (Chemistry, pharmacy + application) pp. 17. [Google Scholar]