Abstract
Background
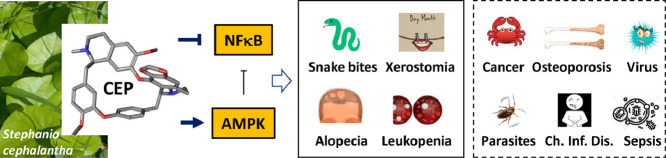
Cepharanthine (CEP) is a drug used in Japan since the 1950s to treat a number of acute and chronic diseases, including treatment of leukopenia, snake bites, xerostomia and alopecia. It is the only approved drug for Human use in the large class of bisbenzylisoquinoline alkaloids. This natural product, mainly isolated from the plant Stephania cephalantha Hayata, exhibits multiple pharmacological properties including anti-oxidative, anti-inflammatory, immuno-regulatory, anti-cancer, anti-viral and anti-parasitic properties.
Purpose
The mechanism of action of CEP is multifactorial. The drug exerts membrane effects (modulation of efflux pumps, membrane rigidification) as well as different intracellular and nuclear effects. CEP interferes with several metabolic axes, primarily with the AMP-activated protein kinase (AMPK) and NFκB signaling pathways. In particular, the anti-inflammatory effects of CEP rely on AMPK activation and NFκB inhibition.
Conclusion
In this review, the historical discovery and development of CEP are retraced, and the key mediators involved in its mode of action are presented. The past, present, and future of CEP are recapitulated. This review also suggests new opportunities to extend the clinical applications of this well-tolerated old Japanese drug.
Keywords: Cepharanthine, Alkaloids, Natural products, Inflammation, Cancer, Stephania
Abbreviations: AMPK, adenosine 5′-monophosphate (AMP)-activated protein kinase; BBIQ, bisbenzylisoquinoline; CEP, cepharanthine; NFκB, nuclear factor κB
Graphical abstract
Introduction
Cepharanthine (CEP, Fig. 1 ) is a natural product and an approved drug used for more than 70 years in Japan to treat a variety of acute and chronic diseases, such as venomous snakebites, alopecia, malaria and a few other pathologies, without major side effects. A variety of biological effects responsible for the clinical activities have been attributed to CEP. In this review, I shall try to summarize the recent findings accumulated with this compound and discuss its therapeutic potential. The history of the drug discovery and development is also highlighted here. The clinical efficacy of CEP has been discussed in previous reviews (Furusawa and Wu, 2007, Rogosnitzky and Danks, 2011) but an update of its properties and applications is useful.
Fig. 1.
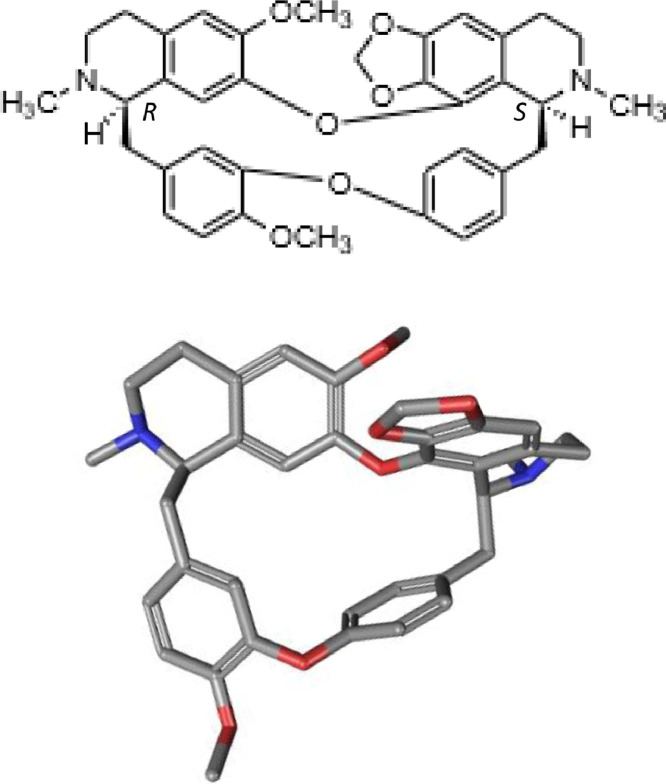
Chemical structure and conformation of cepharanthine (C37H38N2O6).
Origin, discovery and development of CEP
CEP is isolated from plants of the genus Stephania which is one of the biggest genera belonging to moonseed family Menispermaceae. The name Stephania may come from the Greek stefanos, a crown or garland. This alkaloid is found in several Stephania species, principally S. rotunda Lour and S. cephalantha Hayata (known as « Tamasaki-tsuzurafuji » in Japanese; « jin qian diao wu gui » [金 线 吊 乌 龟] in Chinese (Hong et al., 2015); « Bình vôi hoa đầu » in Vietnamese), but also in S. sasakii Hayata (known as « Koto-tsuzurafuji » in Japanese), S. suberosa Forman, S. epigaea (Lv et al., 2013) and S. japonica (Fig. 2 ). Stephania rotunda is a creeper commonly found in the mountainous areas of Cambodia and throughout Southeast Asia (Cambodia, Vietnam, Laos, and India). The plant Stephania cephalantha is native to China and Taiwan, where it was used as an indigenous medicine or Chinese herbal medicine. A botanist of Taipei Imperial University, Bunzo Hayata, reported the use of the plant for the first time in 1914. The correct botanical name is S. cephalantha although the literature principally mentions the plant as S. cepharantha. The vast majority of publications refer to S. cepharantha and rarely S. cephalantha. It is a very common misspelling. Stephania are wild plants, rich in alkaloids, commonly used in traditional food and medicine in Asia for the treatment of fevers.
Fig. 2.
(a) Stephania japonica JB Koishikawa, photo of the plant leaves (Japan, 2018). (b) The caudex of Stephania rotunda (Pierre Fabre Botanical Institute, Cambounet-sur-Sor, France). (c and d) Stephania cephalantha Hayata (China), from the Paris Herbarium, at the National Museum of Natural History, Paris, France (http://coldb.mnhn.fr/catalognumber/mnhn/p/p02467444 and p02384134).
More than 600 alkaloids have been isolated from the genus Stephania and about 40 different alkaloids were characterized from S. cephalantha extracts (Semwal et al., 2010). The chemical composition of extracts varies from one region to another and according to the harvest period. CEP is generally more abundant in the tubers of the plant than the roots and is also found in the leaves and seeds of the plant, which is cultivated in Japan (Desgrouas et al., 2014a). It is important to mention that in certain countries, in France in particular, the use of extracts of S. cepharantha is forbidden due to the risk of potential substitution with other toxic plants having similar Chinese names (notably Aristolochia fanghi, containing aristolochic acids which are nephrotoxic compounds) (Semwal et al., 2010). The principle of pharmacovigilance applied to herbal medicinal products (or phytovigilance) is now well taken in account by the international health agencies (Lehmann and Pabst, 2016). CEP has also been detected in Radix Tinosporae, an herb used in traditional Chinese medicine for the treatment of various inflammatory diseases (Xiang et al., 2016, Liu et al., 2017). This is perhaps not entirely surprising because Tinosporae Radix is the dried root tuber of Tinospora sagittata (Oliv.) Gagnep., which also belongs to the family Menispermaceae.
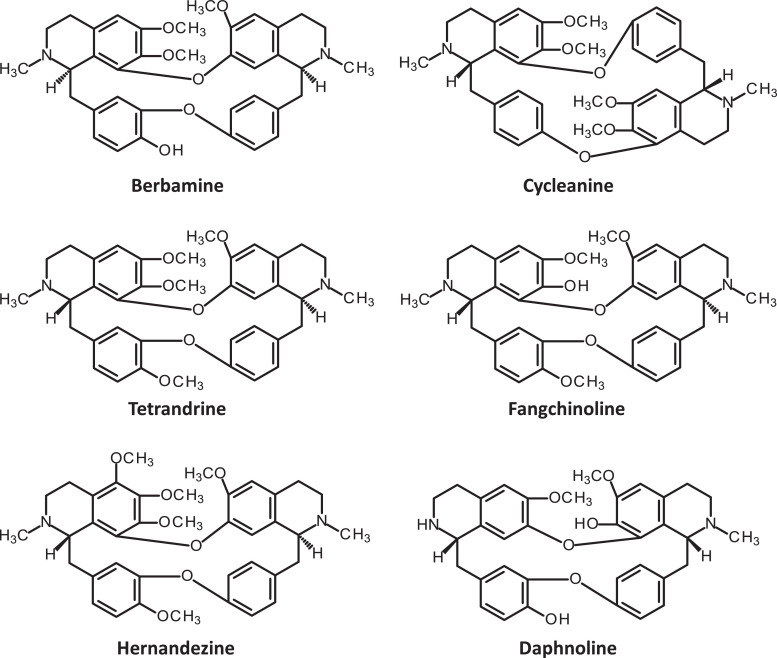
The pharmacist Heisaburo Kondo purified the active ingredient in 1934 from S. cepharantha and named it cepharanthine after its binomial nomenclature (Fig. 3 ). In 1938, a US patent application by Kondo and collaborators referred to the preparation of alkaloids from S. cepharantha and the isolation of CEP itself. From a chemical point of view, CEP is biscoclaurine alkaloid, also known as 12-O-methyl cepharanoline and it belongs to a large family of structurally close bisbenzylisoquinoline (BBIQ) cyclic alkaloids which includes fangchinoline, tetandrine, hernandezine, berbamine, daphnoline, cycleanine (Fig. 4 ), and many other BBIQ cyclic and non-cyclic, cited in Table 1 (note the unlimited originality of their names). These compounds bear two coclaurine units joined head-to-tail or head-to-head, but the chemical diversity is huge and about 500 BBIQs are known in this class (Baghdikian et al., 2013, Weber and Opatz, 2019). In CEP, the two BIQ units are joined head-to-head and the alkaloid has an elliptic macrocyclic structure (Fig. 1).
Fig. 3.
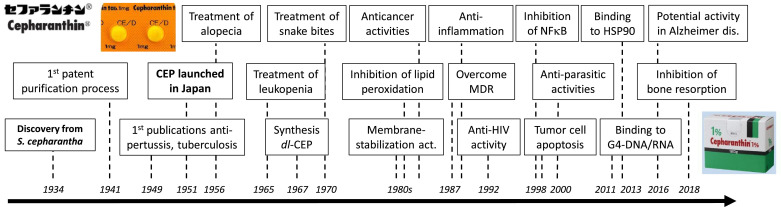
History of CEP discovery and development. Eighty years of evolution of CEP, to mention its main clinical applications and biochemical properties.
Fig. 4.
Structure of six bisbenzylisoquinoline (BBIQ) derivatives.
Table 1.
Bisbenzylisoquinoline (BBIQ) alkaloids.
| Main BBIQs | Plants | Citationsa | References |
|---|---|---|---|
| Tetrandrine | Stephania tetrandra | 910 | Liu et al. (2016b) |
| Cepharanthine | Stephania cephalantha, S. rotunda | 331 | |
| Berbamine | Berberis amurensis, B. vulgaris, B. libanotica, B. aristata, Rhizoma Coptidis, Rhizoma Coptis | 188 | |
| Neferine | Nelumbo nucifera, Plumula nelumbinis | 141 | |
| Fangchinoline | Stephania tetrandra | 115 | Mérarchi et al. (2018) |
| Dauricine | Menispermum dauricum | 110 | Liu et al. (2018b) |
| Other BBIQs | 352 | Weber and Opatz (2019) | |
| antioquine, aromoline, baluchistine, berbacolorflammine, berbibuxine, calafatimine, cocsoline, cocsulinine, colorflammine, curacautine, curine, cycleahomine, cycleanine, cycleatjehine, daphnine, daphnoline, daurisoline, dihydrosecocepharanthine, dihydrothalictrinine, faralaotrine, funiferine, gilletine, hernandezine, johnsonine, krukovine, liensinine, limacine, limacusine, macolidine, macoline, malekulatine, norpanurensine, obaberine, oblongamine, osornine, oxandrine, phaeantharine, racemosidine, repandine, sinomenine, temuconine, thalbadenzine, thalfine, thalibrunine, thalictine, thalictrinine, thalidasine, thalidezine, thalmethine, thalmine,, tiliageine, tubocurine | |||
Number of citations in the PubMed database (May 2019).
Optimized methods for the analysis of these alkaloids in S. cephalantha extracts have been reported (Yuan et al., 2013, Liu et al., 2016a, Xiao et al., 2019), as well as green extraction technologies (using ethanol–water mixtures) to purify CEP from S. rotunda tuber (Desgrouas et al., 2014b). The content of CEP in S. cephalantha is 2.9% (Tomita et al., 1968). The natural product, extracted from the plant, is used for medicinal applications. The total synthesis of CEP, first reported in 1967 (Tomita et al., 1967, Tomita et al., 1968), is not cost-effective. It should be noted that recently, the absolute configuration of CEP has been reassigned from (1R,1′S) to (1R,1′R) using quantum theory (Ren et al., 2019). The pharmaceutically used form of CEP is a dihydrochloride salt. There are several patents specific to CEP, such as (i) a US patent describing the extraction and purification of CEP from rhizomes of S. sasakii Hayata published in 1941; (ii) an European patent application covering the preparation of anti-HIV composition containing CEP in 1992; (iii) A Japanese patent about the preparation CEP for the treatment and prevention of intestinal diseases in 1996; (iv) A Chinese patent describing the method of preparation of CEP hydrochloride filed in 2012; (v) a US patent disclosing the pharmaceutical preparation of CEP and tetrandrine salts and (vi) the production of CEP at the scale of 5–10 kg under good manufacturing practice conditions mentioned in a recent US patent.
CEP was discovered in 1934 and then rapidly tested in Human, based on the traditional use of the medicinal plant. A paper published in 1949 mentions that in 1937 CEP, given in tablets or in liquid, contributed to reduce considerably (from 41% to 22%) the average mortality in patients with severe pulmonary tuberculosis at the Yokohama Sanatorium in Japan (Hasegawa, 1949). At that time, the antitubercular activity of CEP was largely promoted, but later the drug was abandoned, superseded by more active products (Al-Humadi et al., 2017). Nevertheless, the initial successful clinical use of CEP for the treatment of tuberculosis has encouraged its use in other pathological indications (e.g., pertussis, leprosy and lupus vulgaris) and associated research activities to elucidate its mode of action. The biochemical and biological properties, and clinical applications of CEP are reviewed below.
Anti-oxidative properties – free radicals scavenging
Reactive oxygen radicals (ROS), generated metabolically or induced by radiations or chemicals, can engender cell damages, in particular, lesions of nucleic acids. Accumulated DNA damages can be deleterious and toxic to cells, via DNA strand breaks and/or the accumulation of mutations and other types of molecular rearrangements. CEP can protect nuclear DNA from the damages induced by endogenous oxidants, most likely via its antioxidant properties (Halicka et al., 2008). The scavenging properties of CEP also provide a protection of DNA from the radicals generated endogenously during oxidative metabolism. The drug can also reduce the generation of radicals through activation of NADH oxidase (Matsuno et al., 1990). CEP has effective ferrous ion (Fe2+) chelating capacity, similar to EDTA for example. The anti-lipid peroxidation activity of CEP is also due to its direct radical scavenging activity (Kogure et al., 2003). One of the two amine moieties in the CEP molecule is responsible for the radical scavenging activity (Kogure et al., 1999). Many BBIQ alkaloids show antioxidant activities.
Anti-inflammatory properties - NFκB inhibition
The anti-inflammatory effects of CEP are in large part at the origin of the extensive use of S. cephalantha extracts in folk medicine for the treatment of rheumatism, lumbago, nephritis edema, dysentery and other inflammatory diseases. The capacity of CEP to reduce the production of oxidants leads subsequently to the mitigation of the transcription factor NFκB which is a potent regulator of inflammatory molecules. CEP reduces NFκB expression. The anti-inflammatory potential of CEP can be useful in a variety of pathological situations. For example, the capacity of CEP to inhibit inflammation and oxidation leads to protective effect against muscle and kidney injuries induced by limb ischemia/reperfusion (Chang et al., 2016). CEP was also found to ameliorate diabetic nephropathy in a rat model, through inhibition of NFκB and decreased levels of IL-1β and TNF-α (Samra et al., 2016). The drug mitigates pro-inflammatory cytokine response in lung injury induced by hemorrhagic shock/resuscitation in rats (Kao et al., 2015). The release of the pro- inflammatory cytokines TNF-α, IL-1β and IL-6 was found to be reduced after treatment with CEP in cell and mouse models of LPS-induced inflammation and the protective effects occur, at least in part, via NFκB inhibition. Indeed, in a mouse model of lipopolysaccharide (LPS)-induced mastitis, CEP inhibited the phosphorylation of NFκB p65 subunit and the degradation of its inhibitor IκBα (Ershun et al., 2014), thereby inhibiting the translocation of NFκB from cytosol to nucleus (Paudel et al., 2016). However, recently CEP was found to prevent the phosphorylation of JAK2 and STAT1, but did not interfere with the NFκB pathway (Aota et al., 2018). In this cell model, CEP inhibited the production of the chemokine CXCL10 induced by IFN-γ, via inhibition of the JAK2/STAT1 signaling pathway (Lv et al., 2013). Inhibition of NFκB is a mechanism common to several BBIQs. In particular, tetrandrine was shown to inhibit IκBα and NFκB p65 phosphorylation and this effect account, at least in part, for its anti- inflammatory properties (Gao et al., 2016, Xu et al., 2016). Berbamine, another BBIQ, also exerts anti-inflammatory effects via inhibition of NFκB and MAPK signaling (Jia et al., 2017). Neferine also inhibits the activation of the NFκB signaling pathway and prevents its nuclear translocation (Liu et al., 2018a).
Inhibition of drug efflux transporters
The overexpression of drug transporters on the membrane of cancer cells is a major cause of multi-drug resistance to cancer chemotherapy. Among the many transporters, ABCC10 (also known as MRP7) can be inhibited by a variety of drugs including certain kinase inhibitors (lapatinib, erlotinib, imatinib) and different natural products such as CEP. This natural product can reverse completely the resistance to paclitaxel of cells expressing ABCC10, increasing the intracellular accumulation of the anticancer agent and inhibiting its efflux (Zhou et al., 2009). The synergy between CEP and paclitaxel has led to the design of co-loaded polymeric nanoparticules that proved active against gastric cancer models in vitro and in vivo (Yu et al., 2016). ABCC10 is a broad-specificity transporter of xenobiotics, including many antitumor drugs (taxanes, epothilone B, vinca alkaloids, cytarabine, tamoxifen). Therefore, CEP can promote the anticancer activity of most of these compounds. But CEP can also interfere with other transporters, in particular ABCB1 (also known as MDR1 or P-gp) and ABCC1 (MRP1). CEP can be used to reverse resistance to many cytotoxic agents and thus to sensitize tumors to certain chemotherapeutic substances (Kathawala et al., 2014). It potently enhances the sensitivity to cytotoxic agents such as doxorubicin in K562 leukemia cells, not only via a reversal of ABCB1-mediated multi-drug resistance (MDR) but also increasing the accumulation of doxorubicin in nuclei (Ikeda et al., 2005). The suppression of ABCB1 function by CEP would derive from an inhibition of the PI3K/Akt signaling pathway. Inhibition of PI3K/Akt pathway by CEP leads to a down-regulation of ABCB1 expression and then MDR-reversion in ovarian cancer cells (Huang et al., 2017). But a direct interaction of CEP with specific drug transporters at the plasma membrane or via an indirect lipid-mediated effect cannot be excluded. For example, the related BBIQ neferine is believed to bind directly to ABCB1 (Kadioglu et al., 2017).
Binding to proteins: Hsp90 and others
The heat shock protein 90 kDa (Hsp90) has attracted significant interest as a viable drug target, particularly for cancer. Inhibitors targeting the ATPase domain of Hsp90 demonstrate potent anti-proliferative effects, but have failed clinical trials due to associated toxicities. Interestingly, it has been reported that CEP interacts with the middle domain of Hsp90α more strongly than the 3 other biscoclaurine alkaloids berbamine, isotetrandrine and cycleanine (Haginaka et al., 2013). The interaction of CEP with Hsp90 and other molecular chaperone proteins, deserves further studies. CEP also inhibited another ATPase, namely Na(+),K(+)-ATPase, although in this case the inhibition by CEP is much less potent than inhibition by cycleanine (Satoh et al., 2003).
Interaction with cell membranes
Early Japanese studies pointed out that CEP is a membrane-interacting and -stabilizing agent (Shinoda et al., 1990). The drug reduces the plasma membrane fluidity suggesting a direct interaction with the lipid bilayer. The amphipathic character of CEP facilitates its interaction with cell membranes, increasing permeability and interaction with lipophilic compounds. The membrane effects would be at the origin of the inhibition of snake venom-induced hemolysis by CEP (Morikawa et al., 1992) and the drug-induced enhancement of fatty acid synthesis in rat white adipocytes (Ohkura et al., 2001). The drug does not directly act on the insulin-involved glucose metabolism but by changing the cell membrane condition, it modifies the cell response to insulin (Ohkura and Hori, 2001). The membrane effects of CEP might also be implicated in the capacity of the drug to enhance the thermosensitivity of cancer cells, acting as a mild thermosensitizer (Yamamoto et al., 1999). The membrane stabilization effect of CEP is a prime element of its mode of action. Intracellular membranes are also affected. CEP can inhibit the endolysosomal trafficking of free-cholesterol and low-density lipoproteins, leading to the dissociation of mTOR from lysosomes and inhibition of its downstream signaling. This effect may directly contribute to the drug-induced inhibition of angiogenesis and tumor growth (Lyu et al., 2017).
Binding to G-quadruplex nucleic acid structures
Two independent studies have reported that CEP can bind and stabilize G-quadruplex formed by G- rich sequences in DNA and RNA. G-quadruplex DNA are DNA secondary structures formed from planar stacks of guanine tetrads stabilized by Hoogsteen hydrogen bonding. Certain small molecules have the capacity to stabilize these structures. CEP was found to stabilize a G-quadruplex DNA existing in the 3′-flanking region of the STAT3 gene, thereby decreasing the expression of STAT3 in cultured cardiomyocytes (Lin et al., 2011). G-quadruplexes also exist in RNA, in particular telomeric RNA to regulate telomerase activity. CEP was found to bind tightly to two types of G-quadruplex telomeric RNA sequences, with an affinity higher than that of its analogs fangchinoline and tetandrine (Cui et al., 2012). This G-quadruplex stabilization property may contribute to the anticancer activity of CEP.
Vasodilatation effect
The cutaneous microcirculatory effects of CEP have been investigated. The drug induces notable and transient vasodilatation with vasomotion. Vasodilation and improvement of peripheral circulation was also noted in early study suggesting a beneficial use to treat sickle-cell patients (Ohnishi, 1983). The vasodilatator effects of CEP improved cutaneous microcirculation in a rabbit model (Asano et al., 1987). This effect on the microcirculation, coupled with the potent anti-inflammatory activity, speak for the use of CEP to treat certain dermatological and orofacial inflammatory lesions, such as oral lichen planus (Moritani et al., 2018). In early studies, the vasodilatator effects of CEP was also evidenced using isolated vessels from rats and CEP proved more efficient than its analogs isotetrandrine and berbamine (Kamiya et al., 1993). Similarly, the related BBIQ alkaloid curine has been described as a vasodilator (Medeiros et al., 2011).
Anti-proliferative activities – induction of tumor cell death
CEP inhibits the proliferation of different cell types in vitro, including many cancer cell lines, T-cell lines and peripheral mononuclear cells (PBMC). The IC50s are usually in the 1–10 μM range and the drug is non-mutagenic. CEP can trigger apoptosis, via the activation of caspases and DNA fragmentation. The drug affects the cell cycle, generally arresting cells in the G1 and S phases. A mechanistic study using hepatocellular carcinoma HuH-7 cells revealed that CEP induces chromatin condensation and nuclear fragmentation, likely resulting from an oxidative stress and activation of stress activated kinase JNK1/2, MAPK p38 and ERK (Biswas et al., 2006). Induction of apoptosis by CEP has been observed in many cell types: dendritic cells (Uto et al., 2016), primary effusion lymphoma (Takahashi-Makise et al., 2009), leukemia cells (Wu et al., 2002), melanoma cells (Zhu et al., 2017), cholangiocarcinoma (Seubwai et al., 2010), ovarian cancer cells (Payon et al., 2019), myeloma cells (Kikukawa et al., 2008), osteosarcoma cells (Chen et al., 2012) and breast cancer cells (Gao et al., 2017). In colorectal cancer cells, cell cycle arrest and apoptosis may be mediated through up-regulation of p21Waf1/Cip1, down-regulation of cyclin A and Bcl-2 and induction of ROS production (Rattanawong et al., 2018). In fact, the mechanism of apoptosis caused by CEP is complex, likely cell-type dependent, and plurimodal. A direct interaction with mitochondria in cells has been reported, inhibiting Ca2+-induced swelling, depolarization, cytochrome c and Ca2+ releases (Nagano et al., 2003). CEP-induced mitochondrial dysfunctions were noted in several cancer cell types (Hua et al., 2015).
Both drug-induced apoptosis and autophagy were noted in breast cancer cells, possibly linked to perturbation of the AKT/mTOR signaling pathway (Gao et al., 2017). Law and coworkers reported that CEP induces autophagic cell death via an activation of the AMPK kinase activity (Law et al., 2014). The biscoclaurine hernandezine, which is structurally close to CEP, can trigger cell death via an activation of the AMPK-mTOR signaling cascade and its cytotoxic action is independent of apoptosis (Law et al., 2016). Similar data were reported with CEP, and this capacity to enhance autophagy likely contributes to its anticancer activity (Law et al., 2014). This is significant considering that most chemotherapy-resistant cancers have defective apoptotic pathways. The direct activation of AMPK by CEP mays be beneficial not only to trigger cancer cell death but also in other pathologies, in particular metabolic disorders, considering the implication of AMPK in glucose and lipid metabolisms. AMPK activation is a key property of CEP and related biscoclaurine alkaloids.
There are numerous anticancer in vitro studies with CEP but fewer studies in vivo. Interestingly, a modest but significant antitumor activity has been reported in a xenograft model of SaOS2 osteosarcoma cells. Treatment of tumor-bearing mice with CEP at 20 mg/kg/day for 20 days led to a reduction of tumor volume and a diminished expression of STAT3 in the residual tumor (Chen et al., 2012). A significant inhibition of tumor growth and infiltration was also observed after CEP treatment of immunodeficient mice inoculated with a primary effusion lymphoma. The antitumor activity would result from the capacity of CEP to abrogate the NFκB activity by blocking phosphorylation of p65 subunit, inhibiting thus nuclear translocation of NFκB and its binding to DNA (Takahashi-Makise et al., 2009, Seubwai et al., 2010). In a xenograft model of choroidal melanoma, CEP (25 mg/kg, 5 times/week for 4 weeks) reduced tumor growth and the effect was associated with an activation (phosphorylation) of JNK1/2 (Zhu et al., 2017).
Inhibition of cell migration – anti-metastatic and anti-atherosclerosis properties
Migration and proliferation are common characteristics of cancer cells and vascular smooth muscle cells (VSMC). In both situations, CEP has been shown to inhibit the motility of the cells and their growth. The anti-migratory effect of CEP on VSMCs is due to its inhibitory effect on matrix metalloproteinase (MMP)-9 expression, preventing the degradation of extracellular matrix components. This effect, coupled with attenuation of inflammation and lipid peroxidation, confers to CEP anti-atherosclerosis properties (Paudel et al., 2016). A CEP-induced suppression of MMP-9 production was also observed in human salivary gland acinar cells, via a down-regulation of NFκB (Azuma et al., 2002). The inhibition of MMP-9 by CEP prevents the destruction of the acinar structure in the salivary glands of patients with Sjögren's syndrome (Azuma et al., 2006). Similar properties are invoked to account for the anti-metastatic effects of CEP. The drug suppresses the metastatic potential of cholangiocarcinoma cells by reducing cell motility and invasion, via a mechanism potentially involving suppression of adhesion molecules (ICAM-1) and matrix metalloproteinase (MMP)-2 (Uthaisar et al., 2012). Other biscoclaurine alkaloids structurally close to CEP, such as fangchinoline (Chen et al., 2017) and tetrandrine (Zhang et al., 2018) were found to exhibit anti-metastatic effects as well. Interestingly, the anti-metastatic activity of fangchinoline in human gastric cancer cells was also found to implicate the suppression of MMP-2 and MMP-9, increase of TIMP1 and TIMP2 genes, and inhibition of AKT phosphorylation (Chen et al., 2017). Fangchinoline inhibits metastasis and migration of melanoma and glioma cells in vitro, as well as it suppresses in a dose-dependent manner the adhesion, migration, and invasion human gastric cancer cells (Mérarchi et al., 2018). Tetrandrine inhibits hepatocellular carcinoma cell invasion and migration by repressing the autophagy-dependent Wnt/β-catenin and metastatic tumor antigen 1 (MTA1) signaling pathways (Zhang et al., 2018).
Inhibition of angiogenesis
The formation of tubule networks of human umbilical vein endothelial cells (HUVEC) and human dermal microvascular endothelial cells (HMVEC) is a classical in vitro model to evaluate the anti-angiogenic effects of drugs. CEP inhibits VEGF-promoted tubule formation in HUVECs, an effect that is likely associated with the inhibition of VEGF and IL-8 expression involved in the blockade of NFκB activity (Harada et al., 2009). CEP was also found to inhibit angiogenesis in HUVEC and in zebrafish in a cholesterol-dependent manner, as mentioned above (Lyu et al., 2017).
Anti-cancer activity of CEP in drug combinations
Owing to its capacity to modulate drug transporters such as PgP and MRP7, CEP can be combined with various drugs. Synergistic effects have been observed with different anticancer drugs, such as the tetramethylnaphthalene anti-viral derivative TMNAA (Toyama et al., 2012), the nitrosourea nimustine (Kono et al., 2002), the anthracycline doxorubicin (Nakajima et al., 2004), the taxanes paclitaxel and docetaxel (Malofeeva et al., 2012), the recently approved new pan-EGFR inhibitor dacomitinib (Tang et al., 2018), and other anticancer agents (Ita et al., 2008). CEP enhances the antitumor activity of cisplatin in lung and breast cancer xenografts in mice (Mukai et al., 2003). Its capacity to downregulate the mRNA expression of MDR1 and P-gp, through activation of c-Jun and enhancement of p21 expression, leads to an increase sensitivity of the tumor to cisplatin treatment (Zhou et al., 2017). Interestingly, CEP not only promotes the anticancer activity of cisplatin, via an inhibition of P-gp but it can also reduce its renal toxicity through an induced expression of metallothionein (Sogawa et al., 2013). In K562/MDR leukemia cells, CEP was found to be very effective for the reversal of the resistance to imatinib (Mukai et al., 2003). Recently, it was shown that the combination of CEP and 5-fluorouracil additively induced apoptotic and necrotic cell death, via an upregulation of the expression of BAK and cleaved PARP in HT-29 human colorectal cancer cells harboring mutant p53, both in vitro and in vivo (Unson et al., 2019). Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) functions as a selective apoptosis-inducing ligand in cancer cells with normal cells remaining unaffected. CEP enhances TRAIL-mediated apoptosis through a down-regulation of survivin expression, not by direct inhibition of survivin mRNA expression but via a downregulation of a deubiquitin protein that modulates survivin expression in renal carcinoma cells (Shahriyar et al., 2018).
Radiosensitization
Radiotherapy is often used for the treatment of oral squamous carcinoma, the most common epithelial malignancy in the oral cavity, frequently observed in patients who smoke and drink heavily. CEP was found to improve the response to radiotherapy in oral squamous carcinoma cell lines. The drug exhibited a strong inhibitory effect on DNA double-strand break repair after radiation and the combined therapy markedly increased tumor cell apoptosis. The combination of CEP plus radiation caused growth inhibition and tumor regression of tumors in mice (Harada et al., 2012). CEP inhibits the production of irradiation-induced IL-6 and IL-8, which are downstream targets of NFκB (Tamatani et al., 2007). A similar radiation sensitization by CEP was also reported using the human cervical adenocarcinoma HeLa cell line. In this case, CEP efficiently decreased ionizing radiation-induced STAT3 and COX-2 activation. In vivo administration of CEP combined with radiation in HeLa xenografts enhanced tumor growth delay and apoptosis, with reduced expression of STAT3, Bcl-2, c-Myc, and COX-2 (Fang et al., 2013).
Treatment of radiation-induced leukopenia and thrombocytopenia
Administration of CEP to accelerate the recovery from leukopenia after X-ray irradiation (or mitomycin treatment) in cancer therapy is one of the classical and well-established use of the alkaloid. The beneficial effect of CEP on leukopenia was first noted in 1965 and then firmly demonstrated in the early 1980s (Oyaizu et al., 2001). The drug can be used to treat but also to prevent from leukopenia, especially neutropenia, in patients treated with anticancer drugs. A large multi-center clinical study demonstrated that CEP is safe and significantly effective for the management of leukopenia caused by radiotherapy, protecting leukocytes from the damaging effects of radiations and showing no effect on erythrocyte and platelet counts, according to this retrospective study (Kanamori et al., 2016). However, other studies indicated that CEP has the capacity not only to limit the platelet destruction process but also to facilitate the platelet production process, via a drug-induced increase of the number of colony-forming unit of megakaryocytes (Sato et al., 2001). Thus, CEP may prolong the platelet lifespan.
Immuno-modulation – treatment of sepsis, chronic immune thrombocytopenia
CEP therapy, alone or in combination with prednisolone, improves symptoms of chronic immune thrombocytopenia (Takahata et al., 2012). The drug was found to be effective in halting the progression of immune thrombocytopenic purpura in a patient with multiple myeloma (Tabata et al., 2012). A link can be established between the capacity of CEP to inhibit the platelet activation and its capacity to alter phospholipid dynamics and functions. This is coherent with early studies showing that inhibition of platelet aggregation by CEP is accomplished during the early, membrane-related activation process. A recent study conducted in a pediatric population also underlined the useful effect of CEP for the management of chronic immune thrombocytopenia (Yamazaki et al., 2017).
CEP can reduce the production of IL-6 and TNF-α in LPS-stimulated dendritic cells (Uto et al., 2011). The potent anti-inflammatory activity of CEP speaks for the potential use of the drug for treating systemic inflammatory response syndromes such as sepsis. Along these lines, CEP was found to attenuate acute lung injury and septic shock after smoke inhalation in sheep (Murakami et al., 2003). Treatment with CEP was also found to delay lethal shock induced by an infection with a mutant of Salmonella typhimurium (Maruyama et al., 2000). A later study revealed that CEP could have a preventive effect on the lethal action of endotoxin or TNFα-induced lethal shock, through a suppression of nitric oxide production in macrophages (Sakaguchi et al., 2007). As a potent immunoregulatory agent, CEP may have a strong potential against various autoimmune diseases and allergy.
Inhibition of bone resorption
CEP has been shown to inhibit osteoclast formation in a model of estrogen deficiency-induced osteoporosis in vivo, without affecting bone formation. Apparently, the attenuation of osteoclastogenesis results from the impairment of the JNK and PI3K-AKT signaling pathways (Zhou et al., 2018). CEP inhibits the phosphorylation of JNK (a member of the MAPK family) and suppresses RANKL-induced phosphorylation of PI3K and AKT, thereby impairing osteoclastogenesis. In another recent study, CEP was also found to inhibit the expression of several osteoclast-differentiation marker genes and to inhibit RANKL-induced osteoclast formation (Lin et al., 2018). These two recent works suggest that CEP might be useful to prevent osteoporosis. Tetrandrine was also shown to attenuate bone erosion in a model of collagen-induced arthritis (Jia et al., 2018).
Antiviral properties
The capacity of CEP to suppress the production of inflammatory cytokines and chemokines in cells is at the origin of its antiviral activity. The anti-HIV activity of CEP was first mentioned in a patent application from 1991. The drug can inhibit HIV-1 replication in latently infected U1 cells, through inhibition of NFκB which is a potent inducer of HIV-1 gene expression. It is a highly potent inhibitor of HIV-1 replication in chronically infected monocytic cells (Okamoto et al., 1998). This property, coupled with its capacity to cross the blood-brain barrier and induce neural cell death, make CEP a potential agent against HIV-1 encephalopathy (Okamoto et al., 2001). The anti-HIV-1 activity not only relies on NFκB inhibition but also CEP-induced inhibition of the HIV-1 entry process by reducing plasma membrane fluidity (Matsuda et al., 2014). Synthetic derivatives of CEP endowed with more potent anti-HIV-1 activities have been described. Replacement of the 12-O-methyl group of CEP with a 12-O-ethylpiperazinyl substituent reinforces the activity against HIV-1 replication (Baba et al., 2001). The drug can also affect the replication of other viruses, in particular it was found to be active against both wild-type and lamivudine-resistant clinical isolates of the hepatitis B virus (Zhou et al., 2012) and early studies demonstrated an activity against Herpes zoster. CEP exhibits strong anti-HSV1 activity (Liu et al., 2004) and proved active against human T-lymphotropic virus type 1 (HTLV-1)-infected T-cell lines when combined with the tetramethylnaphthalene derivative TMNAA (Toyama et al., 2012). Finally, an activity against severe acute respiratory syndrome (SARS)-related coronaviruses has been also reported (Zhang et al., 2005).
Interestingly, molecular docking simulations predicted that CEP could bind to protein targets of the Ebola virus (Uzochukwu et al., 2016). The US company Texas Biomed is developing a more bioavailable, or more easily absorbed, formulation of CEP, which might be combined with chloroquine to combat the Ebola virus.
Treatment of alopecia
CEP is used for the treatment of alopecia areata, also known as spot baldness, an autoimmune disorder which can be treated with topical corticosteroids and/or minoxidil (Hon and Leung, 2011). The first publications describing the therapy of alopecia with CEP date from 1956 in French and 1969 in Japanese (Fig. 3). The anti-alopecia activity of CEP was promoted already in the mid-1950s and remains largely cited today. But over the past 50 years, very limited scientific evidences have been published to confirm and demonstrate the efficacy of CEP to treat alopecia. CEP can protect against aminoglycoside-induced hair cell death (Ou et al., 2009) and it can stimulate cell proliferation and delayed initiation of differentiation and keratinization of the cultured murine cells the hair apparatus (Tanigaki-Obana, 1992). It can also promote the proliferation of cultured hair cells from the murine skin (Nakaoji et al., 1997). The capacity of CEP to stimulate hair growth may result from the drug-induced production of IGF-1 which is an anagen-prolonging growth factor, in dermal papilla cells (Inui and Itami, 2013). A more recent study describing the use of topical lipoid CEP (a lotion at 0.1% CEP) showed only a minor (and not statistically significant) effect on the density, growth rate and thickness of hairs (Inui et al., 2016). Clearly, the use of CEP for the treatment of alopecia areata is not supported by sufficient pharmacological and clinical evidence. However, the drug or an extract of S. cephalantha can be found in different non-medical topical lotions commercialized in Japan.
Treatment of snake bites
This is another historical use of CEP: the treatment of mamushi bites, a specific pit viper (Gloydius blomhoffii) found throughout Japan. It is also seen in China and Korea. Mamushi bites cause severe swelling (causing compression of peripheral arteries and/or compartment syndrome of the extremities) and pain that spreads gradually from the bite site. In general, the platelet count gradually decreases due to the platelet aggregation activity of the venom. Both antiserum and CEP are widely used for the treatment of mamushi snakebites. After the bite, the rapid use of an antivenom is essential to neutralize both the hemorrhagic and lethal activities of the venom. CEP has been proposed as a possible alternative therapy to antivenom, to lessen the inflammation and the pain caused by the snake bite (Hifumi et al., 2011). In the severe cases of bites, the antivenom is the major treatment recommended. The use of CEP alone is not recommended as it cannot neutralize the circulating venom (Hifumi et al., 2013). In 2000, Abe and coworkers showed that CEP inhibits two species of phospholipase B (α and β) purified from the venom of the giant hornet (Vespa mandarinia) (Abe et al., 2000).
Antiparasitic activities
CEP has been shown to be a trypanothione reductase inhibitor (Fournet et al., 1998), an enzyme considered as a robust target to find drugs against Leishmaniasis and the Chagas disease. But the antiparasitic activity of CEP is very limited (Fournet et al., 2000). However, CEP potently enhances the sensitivity of the malaria pathogen Plasmodium falciparum to chloroquine (Haruki et al., 2000). CEP has a modest direct inhibitory effect on the trophozoite stage of parasite growth, but acts as a modulator of resistance, capable of prolonging the clinical utility of chloroquine (Tamez et al., 2005). The cepharanthine-chloroquine association is synergistic (Chea et al., 2007). The combination of CEP with chloroquine (or amodiaquine) improved significantly the survival of mice and extended the delay for parasitic recrudescence (Desgrouas et al., 2014c). CEP blocks P. falciparum development in ring stage, affecting multiple metabolic pathways, such as cell-cell interactions, glycolysis and isoprenoid pathways (Desgrouas et al., 2014d). These data are consistent with the use of Stephania extracts in traditional medicine for the treatment of malaria-related fever.
Other activities
Other types of bioactivity have been attributed to CEP, linked to its anti-inflammatory and anti-oxidant properties. Notably, a potent anti-atherosclerotic effect through attenuation of inflammation, lipid peroxidation and vascular smooth muscle cell migration and proliferation, has been described (Paudel et al., 2016). Similarly, CEP might be useful to halt the progression of diabetic nephropathy, caused by an elevation of oxidative stress and proinflammatory cytokines in situations of hyperglycemia (Samra et al., 2016). CEP was also found to selectively inhibit the binding of amyloid-β oligomers to Ephrin type-B receptor 2 (EphB2) and A-β oligomers are believed to be key molecules involved in the pathogenesis of Alzheimer's disease (Suzuki et al., 2016). NFκB is also considered a promising target for inflammation-based Alzheimer's disease therapy and many phytochemicals able to interfere with the NFκB pathway are considered for the treatment of this most prevalent form of dementia (Seo et al., 2018).
CEP might also be useful to treat human autoimmune thyroid diseases, via its capacity to block T-cell activation by thyroglobulin peptides (Li et al., 2016). In this later case, CEP was first identified using a virtual in silico screening procedure from a library of approved drugs and its capacity to inhibit peptide binding to a HLA-DR variant protein and to block T-cells activation in vitro and in vivo were confirmed experimentally (Li et al., 2016). This discovery opens a new clinical field to explore the use of CEP for the treatment of different autoimmune disorders such as arthritis or Sjögren's syndrome. And last, more exotically, CEP was tested as an antifouling compound for marine paints but was found to be less active than other alkaloids such as camptothecin (Feng et al., 2018).
CEP has probably other unanticipated targets and properties. In particular, an action on certain ion channels may be envisioned considering the activity of related products. Recently, tetrandrine was found to be a potent and selective inhibitor of the Ether-à-go-go-1 (Eag1) ion channel that plays important roles in tumor proliferation, malignant transformation, invasion and metastasis (Wang et al., 2019). However, the structural analogy between the two BBIQ is not sufficient to suspect an activity. For example, the BBIQs berbamine and fangchinoline were found to bind to the water transporting protein aquaporin-4 expressed in the CNS whereas CEP and cycleanine were found to be inactive (Tradtrantip et al., 2012).
Discussion and perspective
CEP is a well-established alkaloid, known for 85 years and the only cyclic BBIQ used in the clinic to treat Human diseases. But surprisingly it has not been extensively studied. For example, a search through of the PubMed database gave 331 references for CEP vs. 910 for the related BBIQ tetrandrine which is not used clinically (Table 1). As presented here, CEP was discovered in 1934, launched in Japan in 1951 and remains used in the clinic today for the treatment of a variety of diseases and symptoms (Fig. 3). It was initially considered as a promising drug for the treatment of tuberculosis, but today the alkaloid is principally used for its anti-inflammatory, immuno-modulatory properties, notably for the treatment of anemia, alopecia and snake bites. The drug can be administered in large dose and over a long term, either orally or intravenously. It is important to underline that this plant natural product is safe, well tolerated, inducing very limited undesirable effects. Side effects are said to be insignificant and rarely appearing.
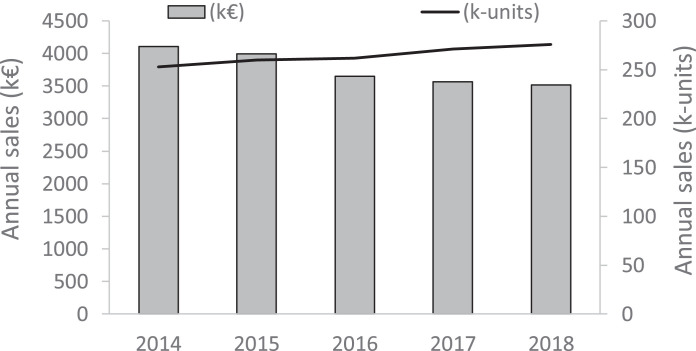
Today CEP remains almost exclusively prescribed in Japan (99% Japan, 1% South Korea), principally in the form of 1 mg tablets orally administered, although the bioavailability of CEP by the oral route is limited (Deng et al., 2017). Each tablet (100 mg) contains 1% CEP mixed with lactose, talc, calcium stearate, magnesium stearate etc. (mainly from KAKEN SHOYAKU, Japan). The usual oral dosage for the treatment of alopecia areata in adults is 1.5–2 mg CEP per day divided in 2–3 doses taken after meals. The dosage is generally higher (3–6 mg/day) for the treatment of leukopenia and usually 1 mg/day for the treatment of bronchial asthma. There is also a liquid form, corresponding to 1 ml or 2 ml vials with 0.5% CEP in a fluid solution (2% benzyl alcohol, 1% NaCl and HCl acid) for injection, but this form is more rarely used. Over the past five years, the pharmaceutical sales of CEP-containing products remained relatively stable, with a slight increase of the volume of products (+8% from 2014 to 2018) but a decrease of the associated economic impact (−14% over the same 5-years period), as illustrated in Fig. 5 . CEP is an inexpensive drug. The sale volumes are limited, corresponding to a very niche and national market. But the drug is well introduced in Japan, being authorized for 68 years now.
Fig. 5.
Pharmaceutical sales of CEP for years 2014–18 (from IMS Health, now IQVIA). The graph shows both the annual sales in k€ (left axis) and in k-units (right axis). Numbers for year 2018 are estimates based on the June 2018 MTA (Moving Annual Total) data.
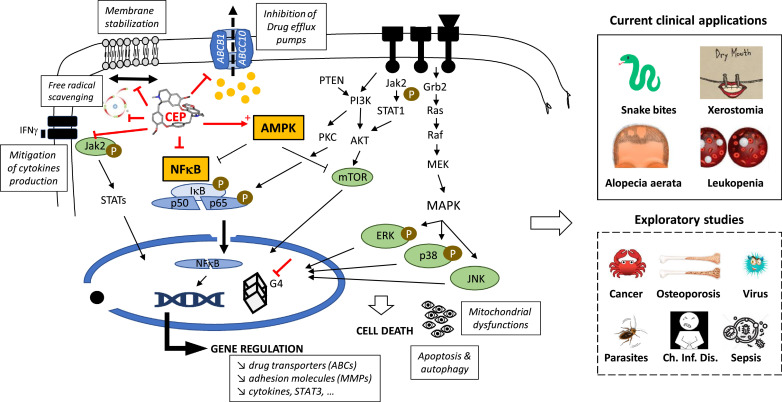
In 2011 Rogosnitzky and Danks concluded that CEP is a fascinating agent with a complex set of physiological actions and an array of clinical benefits (Rogosnitzky and Danks, 2011). Eight years later, the same conclusion can be reinforced. New properties have been discovered, such as the capacity of CEP to bind to quadruplex nucleic acids and to inhibit of bone resorption. It is difficult to summarize its multimodal mechanism of actions but a tentative scheme can be delineated (Fig. 6 ). The mode of action is complex, implicating effects at the plasma membrane (inhibition of drug efflux pumps, membrane rigidification), nuclear effects (G4 stabilization; modulation of gene expression) and cytoplasmic activities, in particular a regulation of the AMPK and NFκβ signaling pathways. With no doubt, the AMPK-NFκβ axis is central to the mode of action of CEP. Accumulating evidence from studies with various drug types indicate that AMPK activation promotes anti-inflammatory responses in cells exposed to stress/danger signals mainly through inhibition of NFκB signaling. There are many examples showing that activation of AMPK is linked to inhibition of NFκB and suppression of the downstream biological effects, such as autophagy activation (Zhai et al., 2018). Moreover, AMPK exerts also a protective role on the antioxidant defense system. Therefore, AMPK activation by CEP can also contribute to the antioxidant effects. Several studies promote the development of AMPK-targeted therapeutics to treat inflammation and related diseases. In this context, the use of CEP as an AMPK activator/NFκB inhibitor should be encouraged. It is worth noting also that the AMPK-NFκβ axis has been invoked to explain the bioactivity of other BBIQs, in particular the anti-inflammatory activity of neferine (Guolan et al., 2018) and the induction of apoptosis by isolensinine (Zhang et al., 2015). AMPK, the so-called “cellular energy regulator”, might well be the pivotal regulator of CEP bioactivity. But it some cases, the Ras/Erk signaling axis seems to be predominantly implicated, notably to regulate cells growth. Interestingly, the related BBIQ krukovine also represses the growth and proliferation of cancer cells by inactivating AKT signaling pathway and downregulating the RAF-ERK signaling pathway (Lai et al., 2018).
Fig. 6.
Main signaling pathways and sites of action of CEP in cells. The multifaceted mechanism of action of CEP leads to several clinical applications (treatment of snake bites, alopecia aerata, xerostomia, leukopenia). Other potential applications of CEP are also explored for the treatment of cancer, osteoporosis, virus and parasites infections, sepsis and chronic inflammatory diseases.
More generally, CEP can be qualified as a protector and a promotor. The drug protects cells, from lipid oxidation, from oxidative DNA damages, from pro-inflammatory cytokines production. At the same time, it promotes the activity of combined drugs and radiations for the treatment of cancer and malaria for examples. One third of the publications on CEP refers to cancer, through the study of drug- induced cell death (apoptosis, autophagy) and drug combinations. But these are mechanistic studies essentially; it is unlikely that the drug can be repositioned in cancer therapy. However, it can open the door to the use and development of other BBIQ, such as tetrandrine from Stephania tetrandra Radix which is perhaps a more promising anticancer agent than CEP (Liu et al., 2016b, Bhagya and Chandrashekar, 2018). CEP itself could be used more largely for the treatment of cancer-related conditions such as xerostomia, anemia or leukopenia.
New horizons for CEP have been evoked in this review, such as a repositioning for the treatment of bone disorders in particular osteoporosis. It really makes sense because NFκB is a robust therapeutic target in inflammatory-associated bone diseases (Lin et al., 2017). CEP activates AMPK by targeting NFκB to suppress autophagy. As such, the drug could also be considered to prevent the development of pulmonary arterial hypertension (Zhai et al., 2018). Other therapeutic perspectives can be proposed. As a well-tolerated, orally available inhibitor of NFκB-mediated inflammation, CEP certainly merits further investigation for efficacy in clinical trials for disorders characterized by inflammation and membrane injury molecular pathways. In particular, CEP might be considered for the treatment of Duchenne muscular dystrophy (DMD) which is a neuromuscular disorder causing progressive muscle degeneration. In DMD, NFκB is activated and drives inflammation and muscle degeneration while inhibiting muscle regeneration. The absence of dystrophin makes skeletal muscle more susceptible to injury, resulting in breaches of the plasma membrane and chronic inflammation in DMD. Novel NFκB inhibitors are currently evaluated as new treatments for DMD patients. It would also make sense to evaluate the potential of CEP in this indication, and in other chronic inflammatory diseases. Along the same vein, CEP might be useful to limit the progression of osteoarthritis, a chronic degenerative joint disease incredibly prevalent in older age. CEP would not repair the already degraded cartilage, but the drug might avoid damage extension to the surrounding tissue, by limiting the production of pro-inflammatory cytokines and chemokines (Herrero-Beaumont et al., 2019). In particular, osteoarthritis patients suffering from a cancer and treated with a regulator of the PD1/PDL1 immune checkpoint might face an aggravation of their osteoarthritis due to a treatment-induced elevated expression of inflammatory cytokine in macrophages (Liu et al., 2019). The use of CEP might thus be useful to limit osteoarthritis progression. A new life for an old drug? Maybe. CEP is not a « one pill fits all » medication but certainly an under-explored drug which should be reconsidered. Hopefully this review will contribute to the renaissance of this well-established alkaloid.
Conflict of interest
I confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Acknowledgments
The photography of the plant Stephania japonica (Fig. 1a) was kindly provided by Prof. Frédéric Dupont, Faculty of Pharmacy, University of Lille, France. The photo of the caudex of S. rotunda (Fig. 1b) was given by Mr Florian Beaucourt (Pierre Fabre Botanical Institute, Cambounet-sur-Sor, France). The photos of Stephania cephalantha (Fig. 1c-d) were provided by Prof. Sovanmoly Hul, Muséum National d'Histoire Naturelle, Paris (France), Collection: Plantes vasculaires (P), specimens P02467444 & P02384134. I thank Mr Bruno David, Mr François Sautel (Pierre Fabre Research Institute) and Prof. Jean-François Goossens (Faculty of Pharmacy, University of Lille) who provided the contacts. The CEP market data in Fig. 5 were graciously collected by Mr François Noailles (Pierre Fabre SA, France). I am grateful to these colleagues for the contribution to the figures.
References
- Abe T., Sugita M., Fujikura T., Hiyoshi J., Akasu M. Giant hornet (Vespa mandarinia) venomous phospholipases: the purification, characterization and inhibitory properties by biscoclaurine alkaloids. Toxicon. 2000;38:1803–1816. doi: 10.1016/s0041-0101(00)00109-4. [DOI] [PubMed] [Google Scholar]
- Al-Humadi H.W., Al-Saigh R.J., Al-Humadi A.W. Addressing the challenges of tuberculosis: a brief historical account. Front. Pharmacol. 2017;8:689. doi: 10.3389/fphar.2017.00689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asano M., Ohkubo C., Sasaki A., Sawanobori K., Nagano H. Vasodilator effects of cepharanthine, a biscoclaurine alkaloid, on cutaneous microcirculation in the rabbit. J. Ethnopharmacol. 1987;20:107–120. doi: 10.1016/0378-8741(87)90083-3. [DOI] [PubMed] [Google Scholar]
- Aota K., Yamanoi T., Kani K., Azuma M. Cepharanthine inhibits IFN-γ-Induced CXCL10 by suppressing the JAK2/STAT1 signal pathway in human salivary gland ductal cells. Inflammation. 2018;41:50–58. doi: 10.1007/s10753-017-0662-x. [DOI] [PubMed] [Google Scholar]
- Azuma M., Aota K., Tamatani T., Motegi K., Yamashita T., Ashida Y., Hayashi Y., Sato M. Suppression of tumor necrosis factor alpha-induced matrix metalloproteinase 9 production in human salivary gland acinar cells by cepharanthine occurs via down-regulation of nuclear factor kappaB: a possible therapeutic agent for preventing the destruction of the acinar structure in the salivary glands of Sjögren's syndrome patients. Arthritis Rheum. 2002;46:1585–1594. doi: 10.1002/art.10315. [DOI] [PubMed] [Google Scholar]
- Azuma M., Ashida Y., Tamatani T., Motegi K., Takamaru N., Ishimaru N., Hayashi Y., Sato M. Cepharanthin, a biscoclaurine alkaloid, prevents destruction of acinar tissues in murine Sjögren's syndrome. J. Rheumatol. 2006;33:912–920. [PubMed] [Google Scholar]
- Baba M., Okamoto M., Kashiwaba N., Ono M. Anti-HIV-1 activity and structure-activity relationship of cepharanoline derivatives in chronically infected cells. Antivir. Chem. Chemother. 2001;12:307–312. doi: 10.1177/095632020101200506. [DOI] [PubMed] [Google Scholar]
- Baghdikian B., Mahiou-Leddet V., Bory S., Bun S.S., Dumetre A., Mabrouki F., Hutter S., Azas N., Ollivier E. New antiplasmodial alkaloids from Stephania rotunda. J. Ethnopharmacol. 2013;145:381–385. doi: 10.1016/j.jep.2012.10.052. [DOI] [PubMed] [Google Scholar]
- Bhagya N., Chandrashekar K.R. Tetrandrine and cancer - an overview on the molecular approach. Biomed. Pharmacother. 2018;97:624–632. doi: 10.1016/j.biopha.2017.10.116. [DOI] [PubMed] [Google Scholar]
- Biswas K.K., Tancharoen S., Sarker K.P., Kawahara K., Hashiguchi T., Maruyama I. Cepharanthine triggers apoptosis in a human hepatocellular carcinoma cell line (HuH-7) through the activation of JNK1/2 and the downregulation of Akt. FEBS Lett. 2006;580:703–710. doi: 10.1016/j.febslet.2005.12.048. [DOI] [PubMed] [Google Scholar]
- Chang Y.K., Huang S.C., Kao M.C., Huang C.J. Cepharanthine alleviates liver injury in a rodent model of limb ischemia-reperfusion. Acta Anaesthesiol. Taiwan. 2016;54:11–15. doi: 10.1016/j.aat.2015.11.004. [DOI] [PubMed] [Google Scholar]
- Chea A., Hout S., Bun S.S., Tabatadze N., Gasquet M., Azas N., Elias R., Balansard G. Antimalarial activity of alkaloids isolated from Stephania rotunda. J. Ethnopharmacol. 2007;112:132–137. doi: 10.1016/j.jep.2007.02.005. [DOI] [PubMed] [Google Scholar]
- Chen Z., Huang C., Yang Y.L., Ding Y., Ou-Yang H.Q., Zhang Y.Y., Xu M. Inhibition of the STAT3 signaling pathway is involved in the antitumor activity of cepharanthine in SaOS2 cells. Acta Pharmacol. Sin. 2012;33:101–108. doi: 10.1038/aps.2011.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z., He T., Zhao K., Xing C. Anti-metastatic activity of fangchinoline in human gastric cancer AGS cells. Oncol. Lett. 2017;13:655–660. doi: 10.3892/ol.2016.5457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui X., Lin S., Zhou J., Yuan G. Investigation of non-covalent interaction of natural flexible cyclic molecules with telomeric RNA G-quadruplexes by electrospray ionization mass spectrometry. Rapid Commun. Mass. Spectrom. 2012;26:1803–1809. doi: 10.1002/rcm.6295. [DOI] [PubMed] [Google Scholar]
- Deng Y., Wu W., Ye S., Wang W., Wang Z. Determination of cepharanthine in rat plasma by LC-MS/MS and its application to a pharmacokinetic study. Pharm. Biol. 2017;55:1775–1779. doi: 10.1080/13880209.2017.1328446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desgrouas C., Taudon N., Bun S.S., Baghdikian B., Bory S., Parzy D., Ollivier E. Ethnobotany, phytochemistry and pharmacology of Stephania rotunda Lour. J. Ethnopharmacol. 2014;154:537–563. doi: 10.1016/j.jep.2014.04.024. [DOI] [PubMed] [Google Scholar]
- Desgrouas C., Baghdikian B., Mabrouki F., Bory S., Taudon N., Parzy D., Ollivier E. Rapid and green extraction, assisted by microwave and ultrasound of cepharanthine from Stephania rotunda Lour. Sep. Pur. Tech. 2014;123:9–14. [Google Scholar]
- Desgrouas C., Dormoi J., Chapus C., Ollivier E., Parzy D., Taudon N. In vitro and in vivo combination of cepharanthine with anti-malarial drugs. Malar. J. 2014;13:90. doi: 10.1186/1475-2875-13-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desgrouas C., Chapus C., Desplans J., Travaille C., Pascual A., Baghdikian B., Ollivier E., Parzy D., Taudon N. In vitro antiplasmodial activity of cepharanthine. Malar. J. 2014;13:327. doi: 10.1186/1475-2875-13-327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ershun Z., Yunhe F., Zhengkai W., Yongguo C., Naisheng Z., Zhengtao Y. Cepharanthine attenuates lipopolysaccharide-induced mice mastitis by suppressing the NF-κB signaling pathway. Inflammation. 2014;37:331–337. doi: 10.1007/s10753-013-9744-6. [DOI] [PubMed] [Google Scholar]
- Fang Z.H., Li Y.J., Chen Z., Wang J.J., Zhu L.H. Inhibition of signal transducer and activator of transcription 3 and cyclooxygenase-2 is involved in radiosensitization of cepharanthine in HeLa cells. Int. J. Gynecol. Cancer. 2013;23:608–614. doi: 10.1097/IGC.0b013e31828a05fd. [DOI] [PubMed] [Google Scholar]
- Feng D.Q., He J., Chen S.Y., Su P., Ke C.H., Wang W. The plant alkaloid camptothecin as a novel antifouling compound for marine paints: laboratory bioassays and field trials. Mar. Biotechnol. 2018;20:623–638. doi: 10.1007/s10126-018-9834-4. [DOI] [PubMed] [Google Scholar]
- Fournet A., Inchausti A., Yaluff G., Rojas De Arias A., Guinaudeau H., Bruneton J., Breidenbach M.A., Karplus P.A., Faerman C.H. Trypanocidal bisbenzylisoquinoline alkaloids are inhibitors of trypanothione reductase. J. Enzyme Inhib. 1998;13:1–9. doi: 10.3109/14756369809035823. [DOI] [PubMed] [Google Scholar]
- Fournet A., Rojas de Arias A., Ferreira M.E., Nakayama H., Torres de Ortiz S., Schinini A., Samudio M., Vera de Bilbao N., Lavault M., Bonté F. Efficacy of the bisbenzylisoquinoline alkaloids in acute and chronic Trypanosoma cruzi murine model. Int. J. Antimicrob. Agents. 2000;13:189–195. doi: 10.1016/s0924-8579(99)00117-x. [DOI] [PubMed] [Google Scholar]
- Furusawa S., Wu J. The effects of biscoclaurine alkaloid cepharanthine on mammalian cells: implications for cancer, shock, and inflammatory diseases. Life Sci. 2007;80:1073–1079. doi: 10.1016/j.lfs.2006.12.001. [DOI] [PubMed] [Google Scholar]
- Gao L.N., Feng Q.S., Zhang X.F., Wang Q.S., Cui Y.L. Tetrandrine suppresses articular inflammatory response by inhibiting pro-inflammatory factors via NF-κB inactivation. J. Orthop. Res. 2016;34:1557–1568. doi: 10.1002/jor.23155. [DOI] [PubMed] [Google Scholar]
- Gao S., Li X., Ding X., Qi W., Yang Q. Cepharanthine induces autophagy, apoptosis and cell cycle arrest in breast cancer cells. Cell. Physiol. Biochem. 2017;41:1633–1648. doi: 10.1159/000471234. [DOI] [PubMed] [Google Scholar]
- Guolan D., Lingli W., Wenyi H., Wei Z., Baowei C., Sen B. Anti-inflammatory effects of neferine on LPS-induced human endothelium via MAPK, and NF-κβ pathways. Pharmazie. 2018;73:541–544. doi: 10.1691/ph.2018.8443. [DOI] [PubMed] [Google Scholar]
- Haginaka J., Kitabatake T., Hirose I., Matsunaga H., Moaddel R. Interaction of cepharanthine with immobilized heat shock protein 90α (Hsp90α) and screening of Hsp90α inhibitors. Anal. Biochem. 2013;434:202–206. doi: 10.1016/j.ab.2012.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halicka D., Ita M., Tanaka T., Kurose A., Darzynkiewicz Z. Biscoclaurine alkaloid cepharanthine protects DNA in TK6 lymphoblastoid cells from constitutive oxidative damage. Pharmacol. Rep. 2008;60:93–100. [PMC free article] [PubMed] [Google Scholar]
- Harada K., Ferdous T., Itashiki Y., Takii M., Mano T., Mori Y., Ueyama Y. Cepharanthine inhibits angiogenesis and tumorigenicity of human oral squamous cell carcinoma cells by suppressing expression of vascular endothelial growth factor and interleukin-8. Int. J. Oncol. 2009;35:1025–1035. doi: 10.3892/ijo_00000417. [DOI] [PubMed] [Google Scholar]
- Harada T., Harada K., Ueyama Y. The enhancement of tumor radioresponse by combined treatment with cepharanthine is accompanied by the inhibition of DNA damage repair and the induction of apoptosis in oral squamous cell carcinoma. Int. J. Oncol. 2012;41:565–572. doi: 10.3892/ijo.2012.1501. [DOI] [PubMed] [Google Scholar]
- Haruki K., Bray P.G., Ono M., Ward S.A. Potent enhancement of the sensitivity of Plasmodium falciparum to chloroquine by the bisbenzylisoquinoline alkaloid cepharanthin. Antimicrob. Agents Chemother. 2000;44:2706–2708. doi: 10.1128/aac.44.10.2706-2708.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa S., Takahashi K. The effect of cepharanthine on pertussis. Jpn. J. Exp. Med. 1949;20:229–234. [PubMed] [Google Scholar]
- Herrero-Beaumont G., Pérez-Baos S., Sánchez-Pernaute O., Roman-Blas J.A., Lamuedra A., Largo R. Targeting chronic innate inflammatory pathways, the main road to prevention of osteoarthritis progression. Biochem. Pharmacol. 2019 doi: 10.1016/j.bcp.2019.02.030. in press. [DOI] [PubMed] [Google Scholar]
- Hifumi T., Yamamoto A., Morokuma K., Ogasawara T., Kiriu N., Hasegawa E., Inoue J., Kato H., Koido Y., Takahashi M. Surveillance of the clinical use of mamushi (Gloydius blomhoffii) antivenom in tertiary care centers in Japan. Jpn. J. Infect. Dis. 2011;64:373–376. [PubMed] [Google Scholar]
- Hifumi T., Yamamoto A., Morokuma K., Okada I., Kiriu N., Ogasawara T., Hasegawa E., Kato H., Inoue J., Koido Y., Takahashi M. Clinical efficacy of antivenom and cepharanthine for the treatment of Mamushi (Gloydius blomhoffii) bites in tertiary care centers in Japan. Jpn. J. Infect. Dis. 2013;66:26–31. doi: 10.7883/yoken.66.26. [DOI] [PubMed] [Google Scholar]
- Hon K.L., Leung A.K. Alopecia areata. Recent Pat. Inflamm. Allergy Drug Discov. 2011;5:98–107. doi: 10.2174/187221311795399291. [DOI] [PubMed] [Google Scholar]
- Hong L., Guo Z., Huang K., Wei S., Liu B., Meng S., Long C. Ethnobotanical study on medicinal plants used by Maonan people in China. J. Ethnobiol. Ethnomed. 2015;30:11–32. doi: 10.1186/s13002-015-0019-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua P., Sun M., Zhang G., Zhang Y., Tian X., Li X., Cui R., Zhang X. Cepharanthine induces apoptosis through reactive oxygen species and mitochondrial dysfunction in human non-small-cell lung cancer cells. Biochem. Biophys. Res. Commun. 2015;460:136–142. doi: 10.1016/j.bbrc.2015.02.131. [DOI] [PubMed] [Google Scholar]
- Huang C.Z., Wang Y.F., Zhang Y., Peng Y.M., Liu Y.X., Ma F., Jiang J.H., Wang Q.D. Cepharanthine hydrochloride reverses P-glycoprotein-mediated multidrug resistance in human ovarian carcinoma A2780/Taxol cells by inhibiting the PI3K/Akt signaling pathway. Oncol. Rep. 2017;38:2558–2564. doi: 10.3892/or.2017.5879. [DOI] [PubMed] [Google Scholar]
- Ikeda R., Che X.F., Yamaguchi T., Ushiyama M., Zheng C.L., Okumura H., Takeda Y., Shibayama Y., Nakamura K., Jeung H.C., Furukawa T., Sumizawa T., Haraguchi M., Akiyama S., Yamada K. Cepharanthine potently enhances the sensitivity of anticancer agents in K562 cells. Cancer Sci. 2005;96:372–376. doi: 10.1111/j.1349-7006.2005.00057.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inui S., Itami S. Induction of insulin-like growth factor-I by cepharanthine from dermal papilla cells: a novel potential pathway for hair growth stimulation. J. Dermatol. 2013;40:1054–1055. doi: 10.1111/1346-8138.12269. [DOI] [PubMed] [Google Scholar]
- Inui S., Tohyama C., Itami S. Acceleration of hair growth rate by topical liposomal cepharanthine in male androgenetic alopecia. Hair Ther. Transplant. 2016;6:145. [Google Scholar]
- Ita M., Halicka H.D., Tanaka T., Kurose A., Ardelt B., Shogen K., Darzynkiewicz Z. Remarkable enhancement of cytotoxicity of onconase and cepharanthine when used in combination on various tumor cell lines. Cancer Biol. Ther. 2008;7:1104–1108. doi: 10.4161/cbt.7.7.6172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia X.J., Li X., Wang F., Liu H.Q., Zhang D.J., Chen Y. Berbamine exerts anti-inflammatory effects via inhibition of NF-κB and MAPK signaling pathways. Cell Physiol. Biochem. 2017;41:2307–2318. doi: 10.1159/000475650. [DOI] [PubMed] [Google Scholar]
- Jia Y., Miao Y., Yue M., Shu M., Wei Z., Dai Y. Tetrandrine attenuates the bone erosion in collagen-induced arthritis rats by inhibiting osteoclastogenesis via spleen tyrosine kinase. FASEB J. 2018;32:3398–3410. doi: 10.1096/fj.201701148RR. [DOI] [PubMed] [Google Scholar]
- Kadioglu O., Law B.Y.K., Mok S.W.F., Xu S.W., Efferth T., Wong V.K.W. Mode of action analyses of neferine, a bisbenzylisoquinoline alkaloid of lotus (Nelumbo nucifera) against multidrug-resistant tumor cells. Front. Pharmacol. 2017;8:238. doi: 10.3389/fphar.2017.00238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamiya T., Sugimoto Y., Yamada Y. Vasodilator effects of bisbenzylisoquinoline alkaloids from Stephania cepharantha. Planta Med. 1993;59:475–476. doi: 10.1055/s-2006-959738. [DOI] [PubMed] [Google Scholar]
- Kanamori S., Hiraoka M., Fukuhara N., Oizumi Y., Danjo A., Nakata K., Owaki K., Nishimura Y. Clinical efficacy of cepharanthin for radiotherapy-induced leukopenia - a nationwide, multicenter, and observational study. Gan To Kagaku Ryoho. 2016;43:1075–1079. [PubMed] [Google Scholar]
- Kao M.C., Yang C.H., Sheu J.R., Huang C.J. Cepharanthine mitigates pro-inflammatory cytokine response in lung injury induced by hemorrhagic shock/resuscitation in rats. Cytokine. 2015;76:442–448. doi: 10.1016/j.cyto.2015.09.008. [DOI] [PubMed] [Google Scholar]
- Kathawala R.J., Wang Y.J., Ashby C.R., Jr, Chen Z.S. Recent advances regarding the role of ABC subfamily C member 10 (ABCC10) in the efflux of antitumor drugs. Chin. J. Cancer. 2014;33:223–230. doi: 10.5732/cjc.013.10122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kikukawa Y., Okuno Y., Tatetsu H., Nakamura M., Harada N., Ueno S., Kamizaki Y., Mitsuya H., Hata H. Induction of cell cycle arrest and apoptosis in myeloma cells by cepharanthine, a biscoclaurine alkaloid. Int. J. Oncol. 2008;33:807–814. [PubMed] [Google Scholar]
- Kogure K., Goto S., Abe K., Ohiwa C., Akasu M., Terada H. Potent antiperoxidation activity of the bisbenzylisoquinoline alkaloid cepharanthine: the amine moiety is responsible for its pH-dependent radical scavenge activity. Biochim. Biophys. Acta. 1999;1426:133–142. doi: 10.1016/s0304-4165(98)00146-9. [DOI] [PubMed] [Google Scholar]
- Kogure K., Tsuchiya K., Abe K., Akasu M., Tamaki T., Fukuzawa K., Terada H. Direct radical scavenging by the bisbenzylisoquinoline alkaloid cepharanthine. Biochim. Biophys. Acta. 2003;1622:1–5. doi: 10.1016/s0304-4165(03)00095-3. [DOI] [PubMed] [Google Scholar]
- Kono K., Takahashi J.A., Ueba T., Mori H., Hashimoto N., Fukumoto M. Effects of combination chemotherapy with biscoclaurine-derived alkaloid (Cepharanthine) and nimustine hydrochloride on malignant glioma cell lines. J. Neurooncol. 2002;56:101–108. doi: 10.1023/a:1014548618440. [DOI] [PubMed] [Google Scholar]
- Lai H., Wang Y., Duan F., Li Y., Jiang Z., Luo L., Liu L., Leung E.L.H., Yao X. Krukovine suppresses KRAS-mutated lung cancer cell growth and proliferation by inhibiting the RAF-ERK pathway and inactivating AKT pathway. Front. Pharmacol. 2018;9:958. doi: 10.3389/fphar.2018.00958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law B.Y., Chan W.K., Xu S.W., Wang J.R., Bai L.P., Liu L., Wong V.K. Natural small-molecule enhancers of autophagy induce autophagic cell death in apoptosis-defective cells. Sci. Rep. 2014;4:5510. doi: 10.1038/srep05510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law B.Y., Mok S.W., Chan W.K., Xu S.W., Wu A.G., Yao X.J., Wang J.R., Liu L., Wong V.K. Hernandezine, a novel AMPK activator induces autophagic cell death in drug-resistant cancers. Oncotarget. 2016;7:8090–8104. doi: 10.18632/oncotarget.6980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehmann H., Pabst J.Y. Phytovigilance: a medical requirement and a legal obligation. Ann. Pharm. Fr. 2016;74:49–60. doi: 10.1016/j.pharma.2015.06.004. [DOI] [PubMed] [Google Scholar]
- Li C.W., Menconi F., Osman R., Mezei M., Jacobson E.M., Concepcion E., David C.S., Kastrinsky D.B., Ohlmeyer M., Tomer Y. Identifying a small molecule blocking antigen presentation in autoimmune thyroiditis. J. Biol. Chem. 2016;291:4079–4090. doi: 10.1074/jbc.M115.694687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin S., Li S., Chen Z., He X., Zhang Y., Xu X., Xu M., Yuan G. Formation, recognition and bioactivities of a novel G-quadruplex in the STAT3 gene. Bioorg. Med. Chem. Lett. 2011;21:5987–5991. doi: 10.1016/j.bmcl.2011.07.121. [DOI] [PubMed] [Google Scholar]
- Lin T.H., Pajarinen J., Lu L., Nabeshima A., Cordova L.A., Yao Z., Goodman S.B. NF-κB as a therapeutic target in inflammatory-associated bone diseases. Adv. Protein Chem. Struct. Biol. 2017;107:117–154. doi: 10.1016/bs.apcsb.2016.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin X., Song F., Zhou L., Wang Z., Wei C., Xu J., Zhao J., Liu Q. Cepharanthine suppresses osteoclast formation by modulating the nuclear factor-κB and nuclear factor of activated T-cell signaling pathways. J. Cell. Biochem. 2018 doi: 10.1002/jcb.27495. in press. [DOI] [PubMed] [Google Scholar]
- Liu X., Wang Y., Zhang M., Li G., Cen Y. Study on the inhibitory effect of cepharanthine on herpes simplex type-1 virus (HSV-1) in vitro. Zhong Yao Cai. 2004;27:107–110. [PubMed] [Google Scholar]
- Liu Y., Xie D., Kang Y., Wang Y., Yang P., Guo J., Huang J. Microwave-assisted extraction followed by solid-phase extraction for the chromatographic analysis of alkaloids in Stephania cepharantha. J. Chromatogr. Sci. 2016;54:670–676. doi: 10.1093/chromsci/bmv191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu T., Liu X., Li W. Tetrandrine, a Chinese plant-derived alkaloid, is a potential candidate for cancer chemotherapy. Oncotarget. 2016;7:40800–40815. doi: 10.18632/oncotarget.8315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y., Zhou W., Mao Z., Liao X., Chen Z. Analysis of six active components in Radix tinosporae by nonaqueous capillary electrophoresis with mass spectrometry. J. Sep. Sci. 2017;40:4628–4635. doi: 10.1002/jssc.201700815. [DOI] [PubMed] [Google Scholar]
- Liu X.Y., Xu H.X., Li J.K., Zhang D., Ma X.H., Huang L.N., Lü J.H., Wang X.Z. Neferine protects endothelial glycocalyx via mitochondrial ROS in lipopolysaccharide-induced acute respiratory distress syndrome. Front. Physiol. 2018;9:102. doi: 10.3389/fphys.2018.00102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu P., Chen X., Zhou H., Wang L., Zhang Z., Ren X., Zhu F., Guo Y., Huang X., Liu J., Spencer P.S., Yang X. The isoquinoline alkaloid dauricine targets multiple molecular pathways to ameliorate Alzheimer-like pathological changes in vitro. Oxid. Med. Cell. Longev. 2018;2018 doi: 10.1155/2018/2025914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S., Mi J., Liu W., Xiao S., Gao C. Blocking of checkpoint receptor PD-L1 aggravates osteoarthritis in macrophage-dependent manner in the mice model. Int. J. Immunopathol. Pharmacol. 2019;33:1–10. doi: 10.1177/2058738418820760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lv J.J., Xu M., Wang D., Zhu H.T., Yang C.R., Wang Y.F., Li Y., Zhang Y.J. Cytotoxic bisbenzylisoquinoline alkaloids from Stephania epigaea. J. Nat. Prod. 2013;76:926–932. doi: 10.1021/np400084t. [DOI] [PubMed] [Google Scholar]
- Lyu J., Yang E.J., Head S.A., Ai N., Zhang B., Wu C., Li R.J., Liu Y., Yang C., Dang Y., Kwon H.J., Ge W., Liu J.O., Shim J.S. Pharmacological blockade of cholesterol trafficking by cepharanthine in endothelial cells suppresses angiogenesis and tumor growth. Cancer Lett. 2017;409:91–103. doi: 10.1016/j.canlet.2017.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malofeeva E.V., Domanitskaya N., Gudima M., Hopper-Borge E.A. Modulation of the ATPase and transport activities of broad-acting multidrug resistance factor ABCC10 (MRP7) Cancer Res. 2012;72:6457–6467. doi: 10.1158/0008-5472.CAN-12-1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maruyama H., Kikuchi S., Kawaguchi K., Hasunuma R., Ono M., Ohbu M., Kumazawa Y. Suppression of lethal toxicity of endotoxin by biscoclaurine alkaloid cepharanthin. Shock. 2000;13:160–165. doi: 10.1097/00024382-200013020-00011. [DOI] [PubMed] [Google Scholar]
- Matsuda K., Hattori S., Komizu Y., Kariya R., Ueoka R., Okada S. Cepharanthine inhibited HIV-1 cell-cell transmission and cell-free infection via modification of cell membrane fluidity. Bioorg. Med. Chem. Lett. 2014;24:2115–2117. doi: 10.1016/j.bmcl.2014.03.041. [DOI] [PubMed] [Google Scholar]
- Matsuno T., Orita K., Edashige K., Kobuchi H., Sato E.F., Inouye B., Inoue M., Utsumi K. Inhibition of active oxygen generation in guinea-pig neutrophils by biscoclaurine alkaloids. Biochem. Pharmacol. 1990;39:1255–1259. doi: 10.1016/0006-2952(90)90271-l. [DOI] [PubMed] [Google Scholar]
- Medeiros M.A., Pinho J.F., De-Lira D.P., Barbosa-Filho J.M., Araújo D.A., Cortes S.F., Lemos V.S., Cruz J.S. Curine, a bisbenzylisoquinoline alkaloid, blocks L-type Ca²⁺ channels and decreases intracellular Ca²⁺ transients in A7r5 cells. Eur. J. Pharmacol. 2011;669:100–107. doi: 10.1016/j.ejphar.2011.07.044. [DOI] [PubMed] [Google Scholar]
- Mérarchi M., Sethi G., Fan L., Mishra S., Arfuso F., Ahn K.S. Molecular targets modulated by fangchinoline in tumor cells and preclinical models. Molecules. 2018;23:10. doi: 10.3390/molecules23102538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morikawa K., Oseko F., Morikawa S. Inhibition of proliferation and differentiation of human B-lymphocytes by a biscoclaurine alkaloid. Int. J. Immunopharmacol. 1992;14:941–949. doi: 10.1016/0192-0561(92)90094-2. [DOI] [PubMed] [Google Scholar]
- Moritani N.H., Hara E.S., Kubota S. New functions of classical compounds against orofacial inflammatory lesions. Medicines. 2018;5:4. doi: 10.3390/medicines5040118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukai M., Che X.F., Furukawa T., Sumizawa T., Aoki S., Ren X.Q., Haraguchi M., Sugimoto Y., Kobayashi M., Takamatsu H., Akiyama S. Reversal of the resistance to STI571 in human chronic myelogenous leukemia K562 cells. Cancer Sci. 2003;94:557–563. doi: 10.1111/j.1349-7006.2003.tb01482.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murakami K., Cox R.A., Hawkins H.K., Schmalstieg F.C., McGuire R.W., Jodoin J.M., Traber L.D., Traber D.L. Cepharanthin, an alkaloid from Stephania cepharantha, inhibits increased pulmonary vascular permeability in an ovine model of sepsis. Shock. 2003;20:46–51. doi: 10.1097/01.shk.0000065768.72937.62. [DOI] [PubMed] [Google Scholar]
- Nagano M., Kanno T., Fujita H., Muranaka S., Fujiwara T., Utsumi K. Cepharanthine, an anti-inflammatory drug, suppresses mitochondrial membrane permeability transition. Physiol. Chem. Phys. Med. NMR. 2003;35:131–143. [PubMed] [Google Scholar]
- Nakajima A., Yamamoto Y., Taura K, Hata K., Fukumoto M., Uchinami H., Yonezawa K., Yamaoka Y. Beneficial effect of cepharanthine on overcoming drug-resistance of hepatocellular carcinoma. Int. J. Oncol. 2004;24:635–645. [PubMed] [Google Scholar]
- Nakaoji K., Nayeshiro H., Tanahashi T., Su Y., Nagakura N. Bisbenzylisoquinoline alkaloids from Stephania cepharantha and their effects on proliferation of cultured cells from the murine hair apparatus. Planta Med. 1997;63:425–428. doi: 10.1055/s-2006-957726. [DOI] [PubMed] [Google Scholar]
- Ohkura K., Hori H. Modification of cell response to insulin by membrane-acting agents in rat white adipocytes: analysis of structural features by computational simulation. Bioorg. Med. Chem. 2001;9:3023–3033. doi: 10.1016/s0968-0896(01)00199-7. [DOI] [PubMed] [Google Scholar]
- Ohnishi S.T. Inhibition of the in vitro formation of irreversibly sickled cells by cepharanthine. Br. J. Haematol. 1983;55:665–671. doi: 10.1111/j.1365-2141.1983.tb02849.x. [DOI] [PubMed] [Google Scholar]
- Okamoto M., Ono M., Baba M. Potent inhibition of HIV type 1 replication by an antiinflammatory alkaloid, cepharanthine, in chronically infected monocytic cells. AIDS Res. Hum. Retroviruses. 1998;14:1239–1245. doi: 10.1089/aid.1998.14.1239. [DOI] [PubMed] [Google Scholar]
- Okamoto M., Ono M., Baba M. Suppression of cytokine production and neural cell death by the anti-inflammatory alkaloid cepharanthine: a potential agent against HIV-1 encephalopathy. Biochem. Pharmacol. 2001;62:747–753. doi: 10.1016/s0006-2952(01)00692-x. [DOI] [PubMed] [Google Scholar]
- Ou H.C., Cunningham L.L., Francis S.P., Brandon C.S., Simon J.A., Raible D.W., Rubel E.W. Identification of FDA-approved drugs and bioactives that protect hair cells in the zebrafish (Danio rerio) lateral line and mouse (Mus musculus) utricle. J. Assoc. Res. Otolaryngol. 2009;10:191–203. doi: 10.1007/s10162-009-0158-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oyaizu H., Adachi Y., Yasumizu R., Ono M., Ikebukuro K., Fukuhara S., Ikehara S. Protection of T cells from radiation-induced apoptosis by cepharanthin. Int. Immunopharmacol. 2001;1:2091–2099. doi: 10.1016/s1567-5769(01)00127-8. [DOI] [PubMed] [Google Scholar]
- Paudel K.R., Karki R., Kim D.W. cepharanthine inhibits in vitro VSMC proliferation and migration and vascular inflammatory responses mediated by RAW264.7. Toxicol. In Vitro. 2016;34:16–25. doi: 10.1016/j.tiv.2016.03.010. [DOI] [PubMed] [Google Scholar]
- Payon V., Kongsaden C., Ketchart W., Mutirangura A., Wonganan P. Mechanism of cepharanthine cytotoxicity in human ovarian cancer cells. Planta Med. 2019;85:41–47. doi: 10.1055/a-0706-7503. [DOI] [PubMed] [Google Scholar]
- Rattanawong A., Payon V., Limpanasittikul W., Boonkrai C., Mutirangura A., Wonganan P. Cepharanthine exhibits a potent anticancer activity in p53-mutated colorectal cancer cells through upregulation of p21Waf1/Cip1. Oncol. Rep. 2018;39:227–238. doi: 10.3892/or.2017.6084. [DOI] [PubMed] [Google Scholar]
- Ren J., Zhao D., Wu S-J., Wang J., Jia Y-J., Li W-X., Zhu H-J., Cao F., Li W., Pittman C.U., He X-J. Reassigning the stereochemistry of bioactive cepharanthine using calculated versus experimental chiroptical spectroscopies. Tetrahedron. 2019;75:1194–1202. [Google Scholar]
- Rogosnitzky M., Danks R. Therapeutic potential of the biscoclaurine alkaloid, cepharanthine, for a range of clinical conditions. Pharmacol. Rep. 2011;63:337–347. doi: 10.1016/s1734-1140(11)70500-x. [DOI] [PubMed] [Google Scholar]
- Sakaguchi S., Furusawa S., Wu J., Nagata K. Preventive effects of a biscoclaurine alkaloid, cepharanthine, on endotoxin or tumor necrosis factor-alpha-induced septic shock symptoms: involvement of from cell death in L929 cells and nitric oxide production in raw 264.7 cells. Int. Immunopharmacol. 2007;7:191–197. doi: 10.1016/j.intimp.2006.09.008. [DOI] [PubMed] [Google Scholar]
- Samra Y.A., Said H.S., Elsherbiny N.M., Liou G.I., El-Shishtawy M.M., Eissa L.A. Cepharanthine and Piperine ameliorate diabetic nephropathy in rats: role of NF-κB and NLRP3 inflammasome. Life Sci. 2016;157:187–199. doi: 10.1016/j.lfs.2016.06.002. [DOI] [PubMed] [Google Scholar]
- Sato T., Morita I., Fujita H., Ono M., Kimishima A., Tomiyama J., Murota S. Pharmacological characterization of cepharanthin in chronic idiopathic thrombocytopenic purpura. Platelets. 2001;12:156–162. doi: 10.1080/09537100120039334. [DOI] [PubMed] [Google Scholar]
- Satoh K., Nagai F., Ono M., Aoki N. Inhibition of Na(+),K(+)-ATPase by the extract of Stephania cephararantha HAYATA and bisbenzylisoquinoline alkaloid cycleanine, a major constituent. Biochem. Pharmacol. 2003;66:379–385. doi: 10.1016/s0006-2952(03)00210-7. [DOI] [PubMed] [Google Scholar]
- Semwal D.K., Badoni R., Semwal R., Kothiyal S.K., Singh G.J., Rawat U. The genus Stephania (Menispermaceae): chemical and pharmacological perspectives. J. Ethnopharmacol. 2010;132:369–383. doi: 10.1016/j.jep.2010.08.047. [DOI] [PubMed] [Google Scholar]
- Seo E.J., Fischer N., Efferth T. Phytochemicals as inhibitors of NF-κB for treatment of Alzheimer's disease. Pharmacol. Res. 2018;129:262–273. doi: 10.1016/j.phrs.2017.11.030. [DOI] [PubMed] [Google Scholar]
- Seubwai W., Vaeteewoottacharn K., Hiyoshi M., Suzu S., Puapairoj A., Wongkham C., Okada S., Wongkham S. Cepharanthine exerts antitumor activity on cholangiocarcinoma by inhibiting NF-kappaB. Cancer Sci. 2010;101:1590–1595. doi: 10.1111/j.1349-7006.2010.01572.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahriyar S.A., Woo S.M., Seo S.U., Min K.J., Kwon T.K. Cepharanthine enhances TRAIL-mediated apoptosis through STAMBPL1-mediated downregulation of survivin expression in renal carcinoma cells. Int. J. Mol. Sci. 2018;19:10. doi: 10.3390/ijms19103280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinoda K., Adachi I., Ueno M., Horikoshi I. Effects of cepharanthine on liposomal permeability and size. Yakugaku Zasshi. 1990;110:186–190. doi: 10.1248/yakushi1947.110.3_186. [DOI] [PubMed] [Google Scholar]
- Sogawa N., Hirai K., Sogawa C., Ohyama K., Miyazaki I., Tsukamoto G., Asanuma M., Sasaki A., Kitayama S. Protective effect of cepharanthin on cisplatin-induced renal toxicity through metallothionein expression. Life Sci. 2013;92:727–732. doi: 10.1016/j.lfs.2013.01.031. [DOI] [PubMed] [Google Scholar]
- Suzuki K., Aimi T., Ishihara T., Mizushima T. Identification of approved drugs that inhibit the binding of amyloid β oligomers to ephrin type-B receptor 2. FEBS Open Bio. 2016;6:461–468. doi: 10.1002/2211-5463.12056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabata R., Tabata C., Tazoh A., Nagai T. Low dose cepharanthine ameliorates immune thrombocytopenic purpura associated with multiple myeloma. Int. Immunopharmacol. 2012;13:242–244. doi: 10.1016/j.intimp.2012.04.015. [DOI] [PubMed] [Google Scholar]
- Takahashi-Makise N., Suzu S., Hiyoshi M., Ohsugi T., Katano H., Umezawa K., Okada S. Biscoclaurine alkaloid cepharanthine inhibits the growth of primary effusion lymphoma in vitro and in vivo and induces apoptosis via suppression of the NF-kappaB pathway. Int. J. Cancer. 2009;125:1464–1472. doi: 10.1002/ijc.24521. [DOI] [PubMed] [Google Scholar]
- Takahata M., Hashino S., Fujimoto K., Endo T., Kobayashi N., Kurosawa M., Iwasaki H., Miyake T., Kohda K., Maekawa I., Sasagawa H., Tsustumi Y., Miyagishima T., Tanaka J., Imamura M., Teshima T. Clinical efficacy of high-dose cepharanthine for idiopathic thrombocytopenic purpura: retrospective multicenter analysis. Rinsho Ketsueki. 2012;53:1983–1990. [PubMed] [Google Scholar]
- Tamatani T., Azuma M., Motegi K., Takamaru N., Kawashima Y., Bando T. Cepharanthin-enhanced radiosensitivity through the inhibition of radiation-induced nuclear factor-kappaB activity in human oral squamous cell carcinoma cells. Int. J. Oncol. 2007;31:761–768. [PubMed] [Google Scholar]
- Tamez P.A., Lantvit D., Lim E., Pezzuto J.M. Chemosensitizing action of cepharanthine against drug-resistant human malaria, Plasmodium falciparum. J. Ethnopharmacol. 2005;98:137–142. doi: 10.1016/j.jep.2005.01.015. [DOI] [PubMed] [Google Scholar]
- Tang Z.H., Cao W.X., Guo X., Dai X.Y., Lu J.H., Chen X., Zhu H., Lu J.J. Identification of a novel autophagic inhibitor cepharanthine to enhance the anti-cancer property of dacomitinib in non-small cell lung cancer. Cancer Lett. 2018;412:1–429. doi: 10.1016/j.canlet.2017.10.001. [DOI] [PubMed] [Google Scholar]
- Tanigaki-Obana N., Ito M. Effects of cepharanthine and minoxidil on proliferation, differentiation and keratinization of cultured cells from the murine hair apparatus. Arch. Dermatol. Res. 1992;284:290–296. doi: 10.1007/BF00372583. [DOI] [PubMed] [Google Scholar]
- Tomita M., Fujitani K., Aoyagi Y. Synthesis of dl-cepharanthine. Tetrahedron Lett. 1967;13:1201–1206. doi: 10.1016/s0040-4039(00)90667-1. [DOI] [PubMed] [Google Scholar]
- Tomita M., Fujitani K., Aoyagi Y., Kajita Y. Studies on the alkaloids of menispermaceous plants. CCXLIV. Synthesis of dl-cepharanthine. Chem. Pharm. Bull. 1968;16:217–226. doi: 10.1248/cpb.16.217. [DOI] [PubMed] [Google Scholar]
- Toyama M., Hamasaki T., Uto T., Aoyama H., Okamoto M., Hashmoto Y., Baba M. Synergistic inhibition of HTLV-1-infected cell proliferation by combination of cepharanthine and a tetramethylnaphthalene derivative. Anticancer Res. 2012;32:2639–2645. [PubMed] [Google Scholar]
- Tradtrantip L., Zhang H., Anderson M.O., Saadoun S., Phuan P.W., Papadopoulos M.C., Bennett J.L., Verkman A.S. Small-molecule inhibitors of NMO-IgG binding to aquaporin-4 reduce astrocyte cytotoxicity in neuromyelitis optica. FASEB J. 2012;26:2197–2208. doi: 10.1096/fj.11-201608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unson S., Kongsaden C., Wonganan P. Cepharanthine combined with 5-fluorouracil inhibits the growth of p53-mutant human colorectal cancer cells. J. Asian Nat. Prod. Res. 2019;29:1–16. doi: 10.1080/10286020.2018.1564136. [DOI] [PubMed] [Google Scholar]
- Uthaisar K., Seubwai W., Srikoon P., Vaeteewoottacharn K., Sawanyawisuth K., Okada S., Wongkham S. Cepharanthine suppresses metastatic potential of human cholangiocarcinoma cell lines. Asian Pac. J. Cancer Prev. 2012;13:149–154. [PubMed] [Google Scholar]
- Uto T, Nishi Y, Toyama M, Yoshinaga K, Baba M. Inhibitory effect of cepharanthine on dendritic cell activation and function. Int. Immunopharmacol. 2011;11:1932–1938. doi: 10.1016/j.intimp.2011.08.003. [DOI] [PubMed] [Google Scholar]
- Uto T., Toyama M., Yoshinaga K., Baba M. Cepharanthine induces apoptosis through the mitochondria/caspase pathway in murine dendritic cells. Immunopharmacol. Immunotoxicol. 2016;38:238–243. doi: 10.3109/08923973.2016.1173059. [DOI] [PubMed] [Google Scholar]
- Uzochukwu I.C., Olubiyi O.O., Ezebuo F.C., Obinwa I.C., Ajaegbu E.E., Eze P.M., Orji C.E., Onuoha M.C., Ejiofor I.I., Ikegbunam M.N. Ending the ebola virus scourge: a case for natural products. Pharm. Res. 2016;1 [Google Scholar]
- Wang X., Chen Y., Li J., Guo S., Lin X., Zhang H., Zhan Y., An H. Tetrandrine, a novel inhibitor of ether-à-go-go-1 (Eag1), targeted to cervical cancer development. J. Cell. Physiol. 2019;234:7161–7173. doi: 10.1002/jcp.27470. [DOI] [PubMed] [Google Scholar]
- Weber C., Opatz T. Bisbenzylisoquinoline alkaloids. Alkaloids Chem. Biol. 2019;81:1–114. doi: 10.1016/bs.alkal.2018.07.001. [DOI] [PubMed] [Google Scholar]
- Wu J., Suzuki H., Akhand A.A., Zhou Y.W., Hossain K., Nakashima I. Modes of activation of mitogen-activated protein kinases and their roles in cepharanthine-induced apoptosis in human leukemia cells. Cell. Signal. 2002;14:509–515. doi: 10.1016/s0898-6568(01)00278-9. [DOI] [PubMed] [Google Scholar]
- Xiang Q., Hashi Y., Chen Z. Simultaneous detection of eight active components in Radix Tinosporae by ultra high performance liquid chromatography coupled with electrospray tandem mass spectrometry. J. Sep. Sci. 2016;39:2036–2042. doi: 10.1002/jssc.201600042. [DOI] [PubMed] [Google Scholar]
- Xiao J., Pan Y., Zhang L., Wang X., Han Y., Sun L., Chen G., Li N. High performance liquid chromatography determination and optimization of the extraction process for the total alkaloids from traditional herb Stephania cepharantha Hayata. Molecules. 2019;24:388. doi: 10.3390/molecules24030388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Liu D., Yin Q., Guo L. Tetrandrine suppresses β-glucan-induced macrophage activation via inhibiting NF-κB, ERK and STAT3 signaling pathways. Mol. Med. Rep. 2016;13:5177–5184. doi: 10.3892/mmr.2016.5187. [DOI] [PubMed] [Google Scholar]
- Yamamoto M., Kuroda M., Honda O., Ono E., Asaumi J.I., Shibuya K., Kawasaki S., Joja I., Takemoto M., Kanazawa S., Hiraki Y. Cepharanthin enhances thermosensitivity without a resultant reduction in the thermotolerance of a murine mammary carcinoma. Int. J. Oncol. 1999;15:95–99. doi: 10.3892/ijo.15.1.95. [DOI] [PubMed] [Google Scholar]
- Yamazaki T., Shibuya A., Ishii S., Miura N., Ohtake A., Sasaki N., Araki R., Ota Y., Fujiwara M., Miyajima Y., Uetake K., Hamahata K., Kato K., Kawakami K., Toyoda H., Moriguchi N., Okada M., Nishi M., Ogata Y., Takimoto T., Ohga S., Ohta S., Amemiya S. High-dose cepharanthin for pediatric chronic immune thrombocytopenia in Japan. Pediatr. Int. 2017;59:303–308. doi: 10.1111/ped.13151. [DOI] [PubMed] [Google Scholar]
- Yu H.H., Mi W.N., Liu B., Zhao H.P. In vitro and in vivo effect of paclitaxel and cepharanthine co-loaded polymeric nanoparticles in gastric cancer. J. BUON. 2016;21:125–134. [PubMed] [Google Scholar]
- Yuan Z., Xiao X., Li G. Dynamic pH junction high-speed counter-current chromatography coupled with microwave-assisted extraction for online separation and purification of alkaloids from Stephania cepharantha. J. Chromatogr A. 2013;1317:203–210. doi: 10.1016/j.chroma.2013.07.063. [DOI] [PubMed] [Google Scholar]
- Zhai C., Shi W., Feng W., Zhu Y., Wang J., Li S., Yan X., Wang Q., Zhang Q., Chai L., Li C., Liu P., Li M. Activation of AMPK prevents monocrotaline-induced pulmonary arterial hypertension by suppression of NF-κB-mediated autophagy activation. Life Sci. 2018;208:87–95. doi: 10.1016/j.lfs.2018.07.018. [DOI] [PubMed] [Google Scholar]
- Zhang C.H., Wang Y.F., Liu X.J., Lu J.H., Qian C.W., Wan Z.Y., Yan X.G., Zheng H.Y., Zhang M.Y., Xiong S., Li J.X., Qi S.Y. Antiviral activity of cepharanthine against severe acute respiratory syndrome coronavirus in vitro. Chin. Med. J. 2005;118:493–496. [PubMed] [Google Scholar]
- Zhang X., Wang X., Wu T., Li B., Liu T., Wang R., Liu Q., Liu Z., Gong Y., Shao C. Isoliensinine induces apoptosis in triple-negative human breast cancer cells through ROS generation and p38 MAPK/JNK activation. Sci. Rep. 2015;5 doi: 10.1038/srep12579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z., Liu T., Yu M., Li K., Li W. The plant alkaloid tetrandrine inhibits metastasis via autophagy-dependent Wnt/β-catenin and metastatic tumor antigen 1 signaling in human liver cancer cells. J. Exp. Clin. Cancer Res. 2018;37:7. doi: 10.1186/s13046-018-0678-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Hopper-Borge E., Shen T., Huang X.C., Shi Z., Kuang Y.H., Furukawa T., Akiyama S., Peng X.X., Ashby C.R.Jr, Chen X., Kruh G.D., Chen Z.S. Cepharanthine is a potent reversal agent for MRP7(ABCC10)-mediated multidrug resistance. Biochem. Pharmacol. 2009;77:993–1001. doi: 10.1016/j.bcp.2008.12.005. [DOI] [PubMed] [Google Scholar]
- Zhou Y.B., Wang Y.F., Zhang Y., Zheng L.Y., Yang X.A., Wang N., Jiang J.H., Ma F., Yin D.T., Sun C.Y., Wang Q.D. In vitro activity of cepharanthine hydrochloride against clinical wild-type and lamivudine-resistant hepatitis B virus isolates. Eur. J. Pharmacol. 2012;683:10–15. doi: 10.1016/j.ejphar.2012.02.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P., Zhang R., Wang Y., Xu D., Zhang L., Qin J., Su G., Feng Y., Chen H., You S., Rui W., Liu H., Chen S., Chen H., Wang Y. Cepharanthine hydrochloride reverses the mdr1 (P-glycoprotein)-mediated esophageal squamous cell carcinoma cell cisplatin resistance through JNK and p53 signals. Oncotarget. 2017;8:111144–111160. doi: 10.18632/oncotarget.22676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou C.H., Meng J.H., Yang Y.T., Hu B., Hong J.Q., Lv Z.T., Chen K., Heng B.C., Jiang G.Y., Zhu J., Cheng Z.H., Zhang W., Cao L., Wang W., Shen W.L., Yan S.G., Wu H.B. Cepharanthine prevents estrogen deficiency-induced bone loss by inhibiting bone resorption. Front. Pharmacol. 2018;9:210. doi: 10.3389/fphar.2018.00210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Q., Guo B., Chen L., Ji Q., Liang H., Wen N., Zhang L. Cepharanthine exerts antitumor activity on choroidal melanoma by reactive oxygen species production and c-Jun N-terminal kinase activation. Oncol. Lett. 2017;13:3760–3766. doi: 10.3892/ol.2017.5945. [DOI] [PMC free article] [PubMed] [Google Scholar]