Abstract
Chloroquine (CQ) and its hydroxyl analogue hydroxychloroquine (HCQ) are weak bases with a half-century long use as antimalarial agents. Apart from this antimalarial activity, CQ and HCQ have gained interest in the field of other infectious diseases. One of the most interesting mechanisms of action is that CQ leads to alkalinisation of acid vesicles that inhibit the growth of several intracellular bacteria and fungi. The proof of concept of this effect was first used to restore intracellular pH allowing antibiotic efficacy for Coxiella burnetii, the agent of Q fever, and doxycycline plus HCQ is now the reference treatment for chronic Q fever. There is also strong evidence of a similar effect in vitro against Tropheryma whipplei, the agent of Whipple's disease, and a clinical trial is in progress. Other bacteria and fungi multiply in an acidic environment and encouraging in vitro data suggest that this concept may be generalised for all intracellular organisms that multiply in an acidic environment. For viruses, CQ led to inhibition of uncoating and/or alteration of post-translational modifications of newly synthesised proteins, especially inhibition of glycosylation. These effects have been well described in vitro for many viruses, with human immunodeficiency virus (HIV) being the most studied. Preliminary in vivo clinical trials suggest that CQ alone or in combination with antiretroviral drugs might represent an interesting way to treat HIV infection. In conclusion, our review re-emphasises the paradigm that activities mediated by lysosomotropic agents may offer an interesting weapon to face present and future infectious diseases worldwide.
Keywords: Chloroquine, Hydroxychloroquine, Lysosomotropic agents, Alkalinisation, Q fever, Whipple's disease, HIV
1. Introduction: general effects of chloroquine (CQ) and hydroxychloroquine (HCQ)
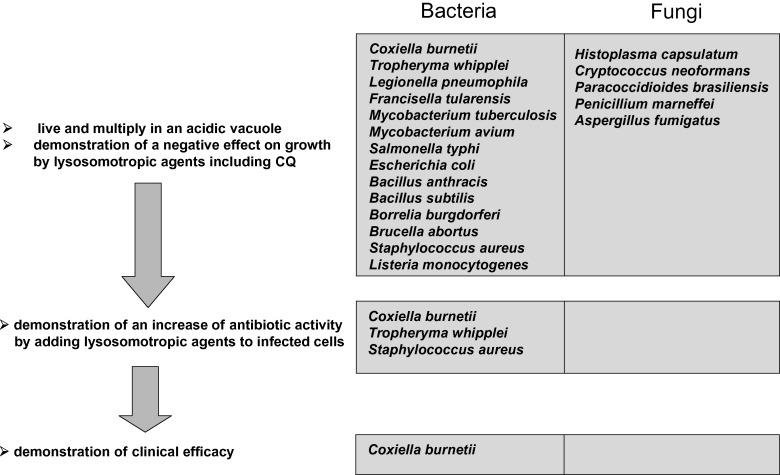
CQ is a 9-aminoquinoline known since 1934, which emerged during the first part of the 20th century as an effective quinine substitute and the drug of choice against malaria [1]. It proved to be among the most successful antimalarial drugs on a worldwide scale owing to its wide deployment coinciding with the geographical distribution of Plasmodium and its high intrinsic antiparasitic efficacy and low toxicity. Concomitant with a gradual decrease in its use for therapy and prophylaxis of Plasmodium-induced disease worldwide, related to the emergence of CQ-resistant parasites, CQ and its hydroxyl analogue HCQ have gained interest in the field of other infectious diseases [2]. The mechanism of action of CQ is multiple, differing according to the pathogen, and has not been well elucidated for all microorganisms. CQ and HCQ enter cells as non-protonated forms where they become protonated according to the Henderson–Hasselbach law, i.e. in a manner inversely proportional to the pH [2]. They therefore concentrate within acidic organelles, including endosomes, lysosomes and Golgi vesicles, in which they increase the pH [3]. Over the last decade, two main mechanisms of action of CQ have been well described, i.e. alkalinisation of acid vesicles in cells infected by intracellular bacteria and fungi, and alteration of post-translational modifications of newly synthesised proteins in cells infected by viruses. The proof of concept of the use of CQ as an anti-infectious agent, other than an antimalarial agent, has been fully demonstrated for the first time in vitro and in vivo with the model of chronic Q fever. This concept was initially based on cellular biology findings, mainly by manipulation of the pH of acidic vacuoles where Coxiella burnetii, the agent of Q fever, live and multiply. Demonstration of a negative effect on growth of C. burnetii by lysosomotropic agents [4] was the first step, followed by demonstration that alkalinising C. burnetii-containing vacuoles could restore the intracellular activity of antibiotics (Fig. 1 ) [5]. This paradigm was later used to demonstrate the clinical efficacy of an association of doxycycline and HCQ in the treatment of chronic Q fever endocarditis and this regimen is now the reference treatment and is to date the only model of confirmed clinical efficacy (Fig. 1) [6]. There is strong evidence of such an effect in other intracellular bacteria, especially for Tropheryma whipplei, the agent of Whipple's disease, which multiply in phagosomes, since agents that increase the intravacuolar pH decrease bacterial viability [7]. By analogy with C. burnetii, HCQ restores the intracellular activity of doxycycline in vitro [8] and a clinical trial using this regimen is under evaluation. Several other bacteria and fungi live and multiply in acidic vacuoles and preliminary in vitro data are encouraging for the usefulness of CQ in such infections. For viruses, CQ led to an inhibition of low-pH-dependent entry steps or alteration of post-translational modifications of newly synthesised proteins, especially via inhibition of glycosylation. These effects have been well described in vitro for many viruses, with human immunodeficiency virus type-1 (HIV-1) being the most studied. Moreover, preliminary in vivo clinical trials have suggested that CQ alone or in combination with antiretroviral drugs might represent an interesting way to treat HIV infection.
Fig. 1.
Concept on the use of chloroquine (CQ) and hydroxychloroquine for intracellular bacteria and fungi: the Coxiella burnetii paradigm.
Here we review available in vitro and in vivo data on the effects of CQ/HCQ on bacterial, fungal and viral infections, with the concept that manipulation of the intracellular pH in cells and modification of glycosylation of proteins by lysosomotropic agents instead of antimicrobial compounds is a powerful approach as new therapeutic strategies for the prevention and therapeutic management of several infectious diseases, including some of great public health concern worldwide. CQ/HCQ also have anti-inflammatory properties, however these will not be discussed in this review.
2. CQ/HCQ efficacy against bacterial infections
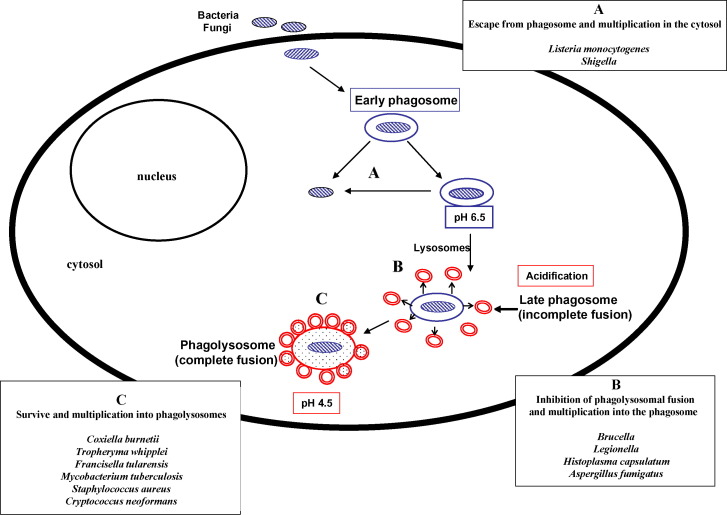
The intracellular location of several bacteria and fungi has been known for decades as a critical point to explain failure of antibiotic treatment to eradicate these pathogens from host cells [9]. Intracellular pathogens evade the first-line antimicrobial defence, which includes attack by phagocytes (Fig. 2 ). After being internalised by the cell, usually there is formation of a phagosome that rapidly fuses with lysosomes. The bacteria are then killed by oxygen-dependent and oxygen-independent killing mechanisms, which leads to acidification of the phagolysosome (pH 4.5) and acidic activation of lysosomal enzymes. Intracellular pathogens may evade this lysosomal pathway by several mechanisms: (i) lifestyle in lysosome free-cells such as erythrocytes (Bartonella spp.); (ii) escape from the phagosome before fusion with lysosomes and multiplication in the cytosol (Rickettsia and Shigella) (Fig. 2); (iii) inhibition of phagolysosomal fusion and multiplication in the phagosome (Chlamydia, Ehrlichia, Legionella, Salmonella, Yersinia, Brucella, Mycobacterium, Francisella, Histoplasma capsulatum and Aspergillus fumigatus) (Fig. 2); and (iv) survival and multiplication in phagolysosomes (C. burnetii, T. whipplei, Staphylococcus aureus, Candida albicans and Cryptococcus neoformans) (Fig. 2) [5], [9], [10]. There are many arguments suggesting that the low pH environment within phagosomal compartments of the cell is critical for many intracellular pathogens to access cellular iron for growth [11], [12]. CQ treatment of different cells, including macrophages, could inhibit the growth of several of these intracellular bacteria by neutralising the phagolysosomal pH (Table 1 ; Fig. 1, Fig. 2). Thus, two main mechanisms can explain intracellular bacterial inhibition by CQ: pH-dependent iron deprivation [11]; and direct toxicity by increasing the phagolysosomal pH, which is harmful for the growth of several intracellular pathogens such as C. burnetii [13] and T. whipplei [7].
Fig. 2.
Strategy for replication of intracellular bacteria and fungi known to be inhibited by chloroquine and/or hydroxychloroquine.
Table 1.
Bacteria, fungi and viruses inhibited by chloroquine and/or hydroxychloroquine (in vitro data)
| Bacteria | Reference | Fungi | Reference | Virus | Reference |
|---|---|---|---|---|---|
| Coxiella burnetii | [5], [13] | Histoplasma capsulatum | [24] | HIV | [2], [29], [30], [31], [32] |
| Tropheryma whipplei | [7], [8] | Cryptococcus neoformans | [15], [25] | SARS-CoV | [33], [34] |
| Legionella pneumophila | [11] | Paracoccidioides brasiliensis | [26] | Influenza viruses | [35], [36], [37], [38] |
| Francisella tularensis | [12] | Penicillium marneffei | [15], [27] | Flavivirus, including yellow fever virus | [39] |
| Mycobacterium tuberculosis | [14] | Aspergillus fumigatus | [28] | Rubella virus | [40], [41] |
| Mycobacterium avium | [15] | HAV | [42] | ||
| Salmonella Typhi | [16] | HBV | [43], [44] | ||
| Escherichia coli | [17] | HCV | [45] | ||
| Bacillus anthracis | [18] | Arenavirus | [46] | ||
| Bacillus subtilis | [19] | ||||
| Borrelia burgdorferi | [20] | Lymphocytic choriomeningitis virus | [47] | ||
| Brucella abortus | [21] | Rabies virus | [48] | ||
| Staphylococcus aureus | [22] | Varicella–Zoster virus | [49] | ||
| Listeria monocytogenes | [23] | Respiratory syncytial virus | [50] | ||
| Sindbis virus | [51] | ||||
| Herpes simplex viruses | [41], [52], [53] | ||||
| Epstein–Barr virus | [54] | ||||
| Polioviruses | [55], [56], [57] | ||||
| Newcastle disease virus | [58] | ||||
| Borna disease virus | [59] | ||||
| Vesicular stomatitis virus | [37], [60], [61], [62] | ||||
| Vaccinia virus | [63] | ||||
| Murine RNA tumour virus | [64] | ||||
| FMDV | [65] | ||||
| Mayaro virus | [66] | ||||
| Feline calicivirus | [67] | ||||
| African swine fever virus | [68] | ||||
| Bovine leukaemia virus | [69] | ||||
| Canine parvovirus | [70] | ||||
| Minute Virus of Mice | [71] |
HIV, human immunodeficiency virus; SARS-CoV, SARS coronavirus; HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus; FMDV, foot and mouth disease virus.
2.1. In vitro activity of HCQ/CQ against C. burnetii
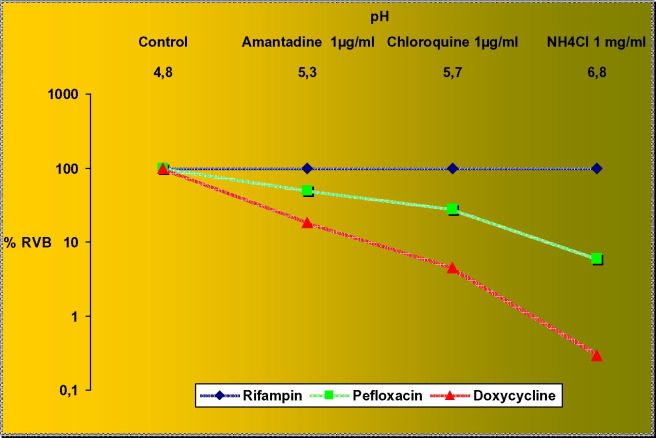
Coxiella burnetii is the agent of Q fever and is a strict intracellular bacterium that is able to survive in phagolysosomes where a low pH (pH 4.5) is necessary for its metabolism [4], [72], [73]. Q fever includes acute manifestations (mainly pneumonitis and hepatitis) and chronic forms (mainly endocarditis) [74]. Usually a regimen of doxycycline 200 mg per day for 3 weeks is recommended for patients with acute Q fever. Presently, this treatment of acute Q fever is not sufficient to prevent the development of chronic Q fever [75]. Thus, reliable antibiotic therapy for chronic infection is a more challenging problem since antibiotics are not bactericidal in vitro against C. burnetii [74]. When the pH of C. burnetii-containing phagolysosomes was raised using basic lysosomotropic agents such as CQ, methylamine and ammonium chloride, bacterial multiplication was inhibited, showing a direct negative effect on growth by lysosomotropic agents, including CQ [4], [13]. An original killing assay model developed by Maurin et al. [5] demonstrated that doxycycline, pefloxacin and rifampicin did not show any significant bactericidal activity. Conversely, it was shown that in vitro intracellular antibiotic activity was restored and was correlated with modification of the pH by lysosomotropic agents (Fig. 3 ). The lack of bactericidal activity was probably due to inactivation by the low pH of the phagolysosomes in which C. burnetii survives. Addition of a lysosomotropic alkalinising agent, i.e. CQ, to antibiotics improved the activities of doxycycline and pefloxacin, which then became bactericidal [5], [13]. The model in which bacteria actively multiply only at acidic pH is a good paradigm to demonstrate that lack of bactericidal effect of antibiotics is due to intraphagolysosomal acidity.
Fig. 3.
Phagolysosomal alkalinisation and bactericidal effect of antibiotics (adapted from Maurin et al. [5]). Colours are those of universal colours used in pH paper. RVB, residual viable bacteria. [Awaiting permission from J Infect Dis.].
2.2. In vivo activity of HCQ/CQ against C. burnetii
These in vitro findings have now been evaluated in many in vivo studies of the treatment of patients with chronic Q fever endocarditis using a combination of doxycycline and HCQ (at 1 μg/mL in serum) for 18–36 months. Indeed, prior to this treatment, patients were treated with a long-term tetracycline and quinolone regimen for at least 4 years, with a high percentage of relapses [6]. This regimen was compared with a combination of doxycycline and hydroxychloroquine sulphate in patients suffering from Q fever endocarditis [6]. Of 14 patients treated with a doxycycline and quinolone combination, 1 died, 7 relapsed (3 were re-treated and 4 switched to the new regimen), 1 was still being treated and 5 were considered cured using this regimen only. The mean duration of therapy for cure in this group was 55 months (median 60 months) [6]. Twenty-one patients received the doxycycline and HCQ regimen: 1 patient died of a surgical complication, 2 were still being treated, 17 were cured and 1 was currently being evaluated. Two patients treated for 12 months but none of the patients treated for >18 months relapsed [6]. The mean duration of treatment in this group was 31 months (median 26 months). This regimen allowed a reduction in the duration of therapy to 18 months for many patients and also reduced the relapse rate to <5% [6]. This regimen is now the current therapy for the treatment of chronic Q fever. CQ used at therapeutic dosages may have some deleterious effects, including the risk of retinopathy, necessitating a regular ophthalmological examination [74]. CQ levels in serum should be monitored to ensure that they are maintained at 1 ± 0.2 mg/L. Similarly, an HIV-infected patient with Q fever endocarditis was successfully treated with valvular replacement and a combination of doxycycline and HCQ [76]. This combined treatment is probably also indicated in cases of C. burnetii vascular graft infection, as reported recently [77]. Similarly, a retrospective study of patients diagnosed as having Q fever during 1985–2000 evaluated the risk of developing endocarditis according to the regimen of antibiotics given to the patients [75]. When these regimens were compared, 6 (75%) of 8 patients who did not receive treatment developed a chronic infection, 5 of 10 developed a chronic infection when receiving doxycycline alone for 2 weeks to 6 months, and none of the 12 who received doxycycline and HCQ for 1–15 months developed chronic infection. The regimen containing HCQ was found to be significantly superior in preventing Q fever endocarditis compared with doxycycline alone (P = 0.009) [75]. No significant differences were found between treatment with doxycycline alone and no treatment [75]. It is now established that development of Q fever endocarditis may be prevented by searching for minor valvulopathies with echocardiography following diagnosis of acute Q fever [78] and by treatment with a combination of doxycycline and HCQ for 1 year [79]. Finally, four cases of Q fever osteoarticular infection (two tenosynovitis and two spondylodiscitis complicated by paravertebral abscess) were eventually cured using the combination of doxycycline and HCQ [80].
2.3. Tropheryma whipplei
Whipple's disease was invariably fatal before the advent of antibiotics. However, current therapeutic recommendations are not based on therapeutic trials or adjusted according to the susceptibility of T. whipplei to various antimicrobial agents, since the bacteria was only isolated in 2000 [81]. Following isolation of the bacteria, it has been shown that vacuole acidification is critical to the survival of T. whipplei in phagosomes, since agents that increase the intravacuolar pH decrease bacterial viability (Fig. 1) [7]. By analogy with C. burnetii, we have demonstrated that doxycycline alone was not bactericidal against T. whipplei in an in vitro cell model and that alkalinisation with HCQ may restore activity [8]. A regimen based on this observation (doxycycline and HCQ) has thus far been the only successful bactericidal regimen against T. whipplei in vitro. Whether this regimen will work in a general clinical setting remains to be established, but it has been successful in four of our patients: two with classic Whipple's disease and two with blood culture-negative endocarditis [82].
2.4. Staphylococcus aureus
Staphylococcus aureus is a facultative intracellular bacterium that resides within phagolysosomes [83], [84], [85], [86]. The intracellular location of certain strains of S. aureus serves as a reservoir of bacteria that is thought to be important in therapy of recurrent infections in humans and in chronic staphylococcal mastitis in dairy cows [87]. Although aminoglycosides are bactericidal for extracellular staphylococci, they are ineffective in reducing the intracellular form of the microorganism [88]. It was hypothesised that diminished susceptibility of intracellular S. aureus may be related to the acidic pH within phagolysosomes [88]. It has been demonstrated that alkalinising S. aureus-containing vacuoles could restore the intracellular activity of aminoglycosides [89]. Furthermore, intracellular killing of S. aureus correlated well with increased lysosomal pH due to lysosomotropic alkalinising agents [89]. Recently, it has been demonstrated that CQ and ammonium chloride significantly enhanced intracellular killing by levofloxacin [22]. The bactericidal activity of levofloxacin was partially restored when the pH was neutralised from 5.0 to 7.4 [22]. The bactericidal activity of moxifloxacin, abolished in the intracellular salt medium, was partially restored when the pH was raised from 5.0 to 7.4 [22]. Similarly, alkalinisation of phagolysosomes significantly enhanced intracellular killing by moxifloxacin [90]. In a model of bovine mastitis due to S. aureus, it has been clearly demonstrated that low intraphagolysosomal pH affects the ability of an antibiotic to kill intracellular bacteria, since the activity of rifampicin was enhanced at pH 5.0 [91]. Similar reasoning probably explains why rifampicin, which both penetrates within eukaryotic cells [92] and is more active at acidic pH [87], displays bactericidal activity against intracellular S. aureus [93].
2.5. Other bacteria with in vitro data on the effectiveness of CQ/HCQ
2.5.1. Legionella pneumophila
Legionella pneumophila is a strict intracellular bacterium that multiplies in human mononuclear phagocytes and is responsible for Legionnaire's disease [94]. Cellular iron metabolism is of critical importance to L. pneumophila since its multiplication is dependent upon the availability of intracellular iron. It has been demonstrated that CQ and ammonium chloride inhibit the intracellular multiplication of L. pneumophila by limiting the availability of iron to the bacterium [11]. Thus, CQ may interfere with intracellular iron metabolism by recycling iron from ferritin by blocking degradation of ferritin by acid proteases [11].
2.5.2. Francisella tularensis
Francisella tularensis bv. tularensis and F. tularensis bv. palearctica are facultative intracellular bacteria responsible for tularaemia. It has been demonstrated that F. tularensis finds a successful niche for replication in an acidified vacuole where iron is concentrated [12]. Growth of F. tularensis in murine macrophages has been shown to be dramatically inhibited in vitro by CQ in a dose-dependent manner [12]. Intracellular localisation in an acidic vesicle, which facilitates the availability of iron essential for Francisella growth, is a survival tactic of this bacterium and iron depletion is one mechanism that macrophages use to inhibit its growth [12].
2.5.3. Mycobacterium spp.
CQ has been reported to inhibit the intracellular multiplication of Mycobacterium tuberculosis both in human monocyte-derived macrophages and mouse peritoneal macrophages [11]. It seems possible that CQ inhibits M. tuberculosis intracellular multiplication by raising intracellular pH and limiting the availability of iron to this bacterium, as it does for L. pneumophila. Similarly, it has been demonstrated that addition of CQ results in a significant reduction of the intracellular growth of Mycobacterium avium in bone marrow-derived macrophages [11].
2.5.4. Miscellaneous
In vitro activity of CQ and/or HCQ has been demonstrated for other bacteria, including Salmonella enterica serovar Typhi, Escherichia coli, Bacillus anthracis, Bacillus subtilis, Borrelia burgdorferi, Brucella abortus and Listeria monocytogenes (Table 1; Fig. 1). These bacteria may be good candidates for clinical use of CQ/HCQ.
3. CQ/HCQ efficacy against fungal infections
Recent in vitro studies indicate that CQ may also have interesting activity against fungal diseases (Fig. 1; Table 1), including mainly H. capsulatum [95] and C. neoformans [96]. Intracellular H. capsulatum is adapted to survive within the mammalian phagolysosome and resides within a membrane-bound phagosome that does not fully acidify. Histoplasma capsulatum is able to maintain a phagosomal pH of 6.5 [97] by inhibition of phagolysosomal fusion [98] and by expression of a unique endogenous H+-ATPase that buffers the phagosomal pH [99]. It has been demonstrated that CQ induces an antihistoplasmal state in macrophages by restricting the pH-dependent release of iron within the phagolysosome [24]. Similarly, CQ has been shown to kill C. neoformans, but by a mechanism independent of iron deprivation [15], [25]. Indeed, unlike H. capsulatum, C. neoformans is able to maintain a phagolysosome milieu at ca. pH 5.1 [100] and addition of CQ increases the phagolysosomal pH allowing inhibition of growth at alkaline pH [25]. It has been reported that A. fumigatus has the ability to inhibit fusion of the phagosome with the lysosome and that CQ may increase killing of this fungus in macrophages by a pH-dependent mechanism [28]. CQ has been shown to inhibit the intramacrophagic growth of Penicillium marneffei [15], an opportunistic fungus that causes disseminated infection in acquired immune deficiency syndrome (AIDS) patients by increasing the intravacuolar pH and disrupting pH-dependent metabolic processes [27]. The decrease in the intracellular iron concentration results in impaired functionality of several cellular enzymes with a subsequent deleterious effect on critical steps such as replication of cellular DNA or gene expression [27]. Finally, the effect of CQ on multiplication of Paracoccidioides brasiliensis has been studied in human monocytes and in a murine paracoccidioidomycosis model [26]. CQ was demonstrated to be able to kill P. brasiliensis grown in human monocytes. The effect of CQ was reversed by FeNTA, an iron compound that is soluble at neutral to alkaline pH, but not by holotransferrin, which releases iron only in an acidic environment. Thus, CQ inhibits P. brasiliensis survival in human monocytes by iron deprivation [26].
4. CQ/HCQ efficacy against viral infections (Table 1)
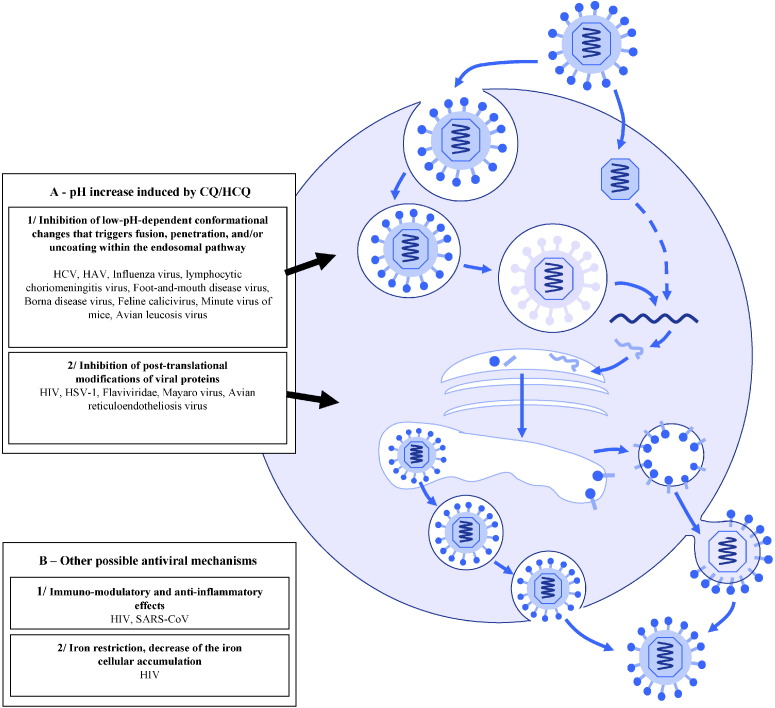
4.1. Mechanisms of antiviral activity (Fig. 4)
Fig. 4.
Viruses inhibited by chloroquine (CQ) and/or hydroxychloroquine (HCQ). HCV, hepatitis C virus; HAV, hepatitis A virus; HIV, human immunodeficiency virus; HSV-1, herpes simplex virus type-1; SARS-CoV, severe acute respiratory syndrome-associated coronavirus.
The pH increase induced by CQ/HCQ within acidic organelles, including endosomes, lysosomes and Golgi vesicles, is involved in its antiviral activity via two main mechanisms.
First, these drugs might be responsible for inhibition of viruses requiring a pH-dependent step for entry into their host cell. Indeed, many viruses have a low-pH-dependent conformational change that triggers fusion, penetration and/or uncoating, and for these viruses endocytosis is crucial due to acidification that occurs within the endosomal pathway [101]. Thus, in this mechanism the antiviral effect is dependent on the extent to which the virus uses endosomes for entry [59]. For instance, Shibata et al. [35] found that CQ might prevent the uncoating of influenza B virus by increasing the lysosomal pH above the critical value required for inducing fusion between the virus envelope and the lysosomal membrane. CQ was also found to inhibit uncoating of the hepatitis A virus (HAV) [42].
Second, CQ/HCQ might inhibit post-translational modifications of the virus envelope glycoproteins by proteases and glycosyltransferases within the trans-Golgi network and endoplasmic vesicles. Indeed, some of these enzymes require a low pH for their activity and CQ/HCQ might therefore lead to decreased viral infectivity through impaired envelope maturation. Flaviviridae are examples of viruses for which CQ could act as an antiviral by inhibiting their envelope maturation pathway through alteration of the proteolytic processing of the prM protein [39].
Also, a putative mechanism for anti-HIV-1 activity is through alteration of the glycosylation pattern and amino acid charge within several regions of the gp120 viral envelope protein [2], [102]. For instance, a reduction in the number of potential N-linked glycosylation sites within the V3 region of gp120, which might provide for altered immune escape and broadening of the antibody repertoire, has been observed [102]. Savarino et al. [2] previously found that CQ decreased the infectivity of newly produced HIV-1 as well as the ability of HIV-1-infected cells to form syncitia, and this was associated with structural changes in gp120. Recent data further suggest that CQ may be responsible for inhibition of the biosynthesis of sialic acid. Indeed, it was recently observed that this drug inhibited cellular enzymes involved in sialic acid biosynthesis [103]. This might represent a major antiviral mechanism, since sialic acids are a component of HIV-1 envelope glycoproteins.
CQ/HCQ may also have indirect antiviral effects. Indeed, CQ was found to be effective in preventing the spread of severe acute respiratory syndrome (SARS)-associated coronavirus (CoV) in cell culture by interfering with terminal glycosylation of the cellular receptor, angiotensin-converting enzyme 2 (ACE2) [33]; and sialic acids, biosynthesis of which might be inhibited by CQ/HCQ, are component of receptors of SARS-CoV and orthomyxoviruses [36].
In addition to these two main mechanisms, other possible mechanisms such as immunomodulatory and anti-inflammatory properties have been suggested, however these will not be discussed in the present review.
4.2. Activity of HCQ/CQ against viruses
In vitro activity of HCQ/CQ has been reported for a wide range of viruses, most frequently in experiments aiming to study the cycle replication pathways using CQ, especially the mechanism by which viruses penetrate host cells (Table 1; Fig. 4).
Inhibitory concentrations of CQ fell within the 0.5–10 μmol/L range, depending on antiviral CQ doses, the viruses that were targeted and the assays used for assessment of the antiviral effect. Importantly, these concentrations are in the range that is clinically achieved in plasma during malaria therapy, varying from 1.6 μmol/L to 12.5 μmol/L [104]. The most studied in vitro effect of CQ/HCQ has been against HIV. Moreover, in vivo studies quasi-exclusively concerned HIV-1. This is probably due to the high morbidity and mortality related to HIV-1 worldwide and the need for low-cost antiretroviral therapies in resource-poor countries.
4.3. In vitro anti-HIV activity of HCQ/CQ
CQ/HCQ have largely been shown to inhibit HIV replication in vitro. Importantly, this inhibition was observed in several cell line models, but also in lymphocytes and monocytes from peripheral blood [29]. An anti-HIV effect has been demonstrated either in the presence of high concentrations of CQ/HCQ prior to infection of HIV-1-permissive cells [29], [30], [102] or during incubation of HIV-infected cells with CQ concentrations similar to those found in peripheral blood from individuals chronically treated with CQ [31]. Thus, these data suggest both a preventive and curative effect of this drug against HIV. The concentration inhibiting 50% of viral replication (IC50) ranged between 1 μmol/L and 10 μmol/L for various HIV strains and culture cells in the studies of Savarino et al. [2]. Boelaert et al. [105] described an additional inhibition of HIV-1 replication with hydroxyurea plus didanosine (ddI) or zidovudine, with a CQ IC50 of 0.4–0.9 μmol/L for the cell lines and 0.2–0.9 μmol/L for the primary cells; no CQ-induced toxicity or apoptosis was noted. Interestingly, according to in vitro data, CQ/HCQ also appears to be active against HIV-2, which mostly circulates in West Africa, as well as against HIV-1 of different subtypes [2]. This deserves attention since HIV-2 strains have established or suggested natural resistance to several antiretroviral drugs, such as non-nucleoside reverse transcriptase inhibitors and likely some protease inhibitors (PIs) [106].
4.4. In vivo anti-HIV activity of HCQ/CQ
Anti-HIV-1 activity of CQ/HCQ has been observed in a few in vivo studies since 1995 [107], [108], [109], [110], [111]. Two small, phase II, randomised, double-blind studies, including 40 and 72 patients with CD4 cell counts of 200–500 cells/mm3, compared reduction of plasma HIV-1 RNA levels in individuals treated with HCQ versus either placebo or zidovudine [107], [108]. In both trials, more than two-thirds of patients were antiretroviral-naive. In the first trial, 8 weeks of treatment with 800 mg HCQ per day resulted in a significant mean 0.6 log10 reduction of HIV-1 load (P = 0.022), whereas no significant decrease was observed in the placebo arm (20 patients in each arm) [107]. Concomitantly, the percentage of CD4+ lymphocytes remained stable in the HCQ group, whereas it significantly decreased in the placebo arm (P = 0.032). In the second trial, 35 and 37 patients received HCQ or zidovudine, respectively, for 16 weeks [108]. HIV-1 load was significantly reduced in both groups (by 0.4 log10 copies/mL and 0.6 log10 copies/mL, respectively) and, interestingly, 0 of 35 patients in the HCQ group versus 8 of 37 patients in the zidovudine group showed an increase in HIV-1 RNA levels. Two other recent non-controlled studies have been reported. In Singapore, 22 patients with HIV-1 load <100 000 copies/mL and CD4 cell count >150 cells/mm3 received HCQ (200 mg), hydroxyurea and ddI twice daily for 48 weeks, resulting in a 1.3 log10 decrease in plasma HIV-1 RNA levels [109]. HIV-1 RNA levels were further reduced compared with baseline (mean decrease 1.6 log10 copies/mL) in all 14 patients who completed a 144-week course of therapy, with drug resistance mutations detected in 4 patients at this time point [110]. In another study in India, 18 patients with CD4 counts >350/mm3 received lamivudine, hydroxyurea and CQ (250 mg) twice daily for 6 months [111]. HIV-1 load reduction was significant (−2.0 log10), reaching undetectable levels in 10 patients, and the median rise in CD4 count was 78 cells/mm3. Altogether, these in vivo data in patients with non-severe immunosuppression (CD4 cell count >200/mm3) at least suggest that HIV-1 resistance to CQ/HCQ alone or in combination with antiretroviral drugs might not develop easily [36]. In contrast, addition of CQ to a zidovudine and ddI regimen provided no significant improvement in viro-immunological parameters in 21 HIV-1-infected children in a recent study from Thailand [112].
It is interesting to note that a 243-fold accumulation of CQ in colostrum cells of African mothers taking 100 mg of CQ per day has been observed. This suggests that this drug could be potentially active as an adjuvant to post-natal antiretroviral prophylaxis of mother-to-child transmission by decreasing HIV-1 load in milk in geographical areas where vertical transmission is of great concern [113], [114].
5. Additive or synergistic effects of HCQ/CQ with antiretroviral drugs
In several settings, CQ/HCQ should be used in combination with other antiviral drugs, questioning whether associations may be additive or synergistic. The additive effect of CQ and zidovudine has been shown, and might also exist in association with ddI or hydroxyurea [105]. Furthermore, the combined effect of CQ and PIs is synergistic in a dose-independent manner [2]. Savarino et al. showed that CQ in combination with PIs carries out a combined inhibitory effect on P-glycoprotein and multidrug resistance protein 1, which is involved in efflux of PIs, a major class of antiretrovirals. Interestingly, the synergism between CQ and PIs was associated with a decreased threshold of susceptibility to PIs in resistant isolates [2]. Of note, CQ is a major substrate of cytochrome CYP3A4 [116], and nevirapine (a non-nucleoside HIV-1 reverse transcriptase inhibitor) and HIV PIs are well recognised CYP3A4 inducers and inhibitors, respectively [115]. Thus, these latter drugs might subsequently decrease or increase the levels/effects of CQ.
5.1. Activity of HCQ/CQ on SARS-CoV and orthomyxoviruses
It has been shown that endosomal transport is needed for human coronavirus HCoV-229E and that cells treated with CQ displayed reduced expression of viral antigens [114]. More recently, CQ was found to have strong antiviral effects on SARS-CoV infection in cell cultures when they were treated either before or after exposure to the virus (even 3–5 h following infection), suggesting both prophylactic and therapeutic effects [33], [34]. In Keyaerts et al.'s study [34], CQ inhibited viral replication with a 50% effective concentration (EC50) of 8.8 μmol/L. Of note, the dose inducing 50% cytostatic activity was much higher (261.3 μmol/L). Potential mechanisms of action of CQ against coronaviruses are through underglycosylation of ACE2, which has been identified as a functional cellular receptor of SARS-CoV spike protein [33], [117]. Alteration by CQ of the SARS-CoV spike protein is controversial [33], [118]. A pH-related reduction of the transduction of SARS-CoV pseudotype viruses has also been suggested [33], [119], [120]. Of note, Biot et al. [121] recently reported the design and synthesis of hydroxyferroquine derivatives with antimalarial, anti-HIV and anti-SARS-CoV activities.
The activity of CQ against Orthomyxoviridae (influenza A and B viruses) has been described for several decades [35], [37], [122] and in vitro assays on avian influenza virus strains are ongoing [36]. Shibata et al.'s [35] results suggested that CQ prevents the uncoating of influenza B virus. Ooi et al. [38] found that the IC50 values of CQ against influenza A viruses H1N1 and H3N2 were 3.6 μmol/L and 0.84 μmol/L, respectively.
5.2. Activity of HCQ/CQ on hepatitis viruses
In a recent study, Blanchard et al. [45] found that pre-treating target cells with CQ inhibited hepatitis C virus (HCV) clone JFH-1 propagation in cell culture, which suggests that HCV, like flaviviruses and pestiviruses, enters cells through clathrin-mediated endocytosis and fusion within an acidic endosomal compartment. CQ was also found to be active against hepatitis B virus (HBV) and duck HBV [43], [44], [123], contrasting with another study in which infection of human hepatocyte cultures with HBV was found to be unaffected by CQ [124]. Seven patients with histologically proven chronic active hepatitis B have been treated with 150–450 mg of CQ for a median of 12 months [125]. In all patients, alanine aminotransferase (ALT) returned to normal values and prothrombin time improved. Interestingly, ALT increased in three patients following inadvertent CQ withdrawal and returned to prior levels on re-administration. In four patients, a repeat liver biopsy 1 year later revealed inactive cirrhosis. Recently, an enhancement by CQ of human CD8+ T-cell cell response to HBV antigen has been observed [126]. Inhibition by CQ of hepatitis A virus (HAV) uncoating and replication has also been described [42], [60].
5.3. Toxicity and therapeutic range
A major advantage of CQ/HCQ is their limited and preventable toxicity. Long experience of the use of these drugs in the treatment of malaria has already demonstrated the safety of short-term administration to humans. Moreover, CQ/HCQ have been widely used for chronic administration in rheumatic diseases, chronic Q fever and for antimalarial prophylaxis for up to several years with only a low incidence of adverse effects even during pregnancy [114], [127], [128], [129]. The main adverse effect reported in long-term administration of these drugs was macular retinopathy due to the cumulative dose, which could be prevented with regular visual monitoring during the course of treatment [6], [130].
6. Conclusion
In summary, CQ/HCQ have several advantages as antimicrobial agents, including multiple potential mechanisms and a broad spectrum of activity at clinically achievable plasma concentrations, together with well known and limited toxicity and low cost. Two major concepts have emerged to explain the activity of CQ/HCQ, namely alkalinisation of phagolysosomes for intracellular bacteria and fungi, and inhibition of entry steps and protein glycosylation for viruses. The pioneer and the only model of an infectious disease that could be treated by manipulation of intracellular pH by a lysosomotropic agent was chronic Q fever. This review re-emphasises that the C. burnetii paradigm and activities mediated by lysosomotropic agents could be generalised for other intracellular pathogens living in acidic vacuoles or that require a low pH for multiplication. This may offer an interesting weapon to face present and future infectious diseases worldwide.
Funding: None.
Competing interests: None declared.
Ethical approval: Not required.
References
- 1.Wellems T.E., Plowe C.V. Chloroquine-resistant malaria. J Infect Dis. 2001;184:770–776. doi: 10.1086/322858. [DOI] [PubMed] [Google Scholar]
- 2.Savarino A., Lucia M.B., Rastrelli E. Anti-HIV effects of chloroquine: inhibition of viral particle glycosylation and synergism with protease inhibitors. J Acquir Immune Defic Syndr. 2004;35:223–232. doi: 10.1097/00126334-200403010-00002. [DOI] [PubMed] [Google Scholar]
- 3.O’Neill P.M., Bray P.G., Hawley S.R., Ward S.A., Park B.K. 4-Aminoquinolines—past, present, and future: a chemical perspective. Pharmacol Ther. 1998;77:29–58. doi: 10.1016/s0163-7258(97)00084-3. [DOI] [PubMed] [Google Scholar]
- 4.Hackstadt T., Williams J.C. Biochemical stratagem for obligate parasitism of eukaryotic cells by Coxiella burnetii. Proc Natl Acad Sci USA. 1981;78:3240–3244. doi: 10.1073/pnas.78.5.3240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maurin M., Benoliel A.M., Bongrand P., Raoult D. Phagolysosomal alkalinization and the bactericidal effect of antibiotics: the Coxiella burnetii paradigm. J Infect Dis. 1992;166:1097–1102. doi: 10.1093/infdis/166.5.1097. [DOI] [PubMed] [Google Scholar]
- 6.Raoult D., Houpikian P., Tissot Dupont H., Riss J.M., Arditi-Djiane J., Brouqui P. Treatment of Q fever endocarditis: comparison of two regimens containing doxycycline and ofloxacin or hydroxychloroquine. Arch Intern Med. 1999;159:167–173. doi: 10.1001/archinte.159.2.167. [DOI] [PubMed] [Google Scholar]
- 7.Ghigo E., Capo C., Aurouze M. Survival of Tropheryma whipplei, the agent of Whipple's disease, requires phagosome acidification. Infect Immun. 2002;70:1501–1506. doi: 10.1128/IAI.70.3.1501-1506.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Boulos A., Rolain J.M., Raoult D. Antibiotic susceptibility of Tropheryma whipplei in MRC5 cells. Antimicrob Agents Chemother. 2004;48:747–752. doi: 10.1128/AAC.48.3.747-752.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maurin M., Raoult D. Optimum treatment of intracellular infection. Drugs. 1996;52:45–59. doi: 10.2165/00003495-199652010-00004. [DOI] [PubMed] [Google Scholar]
- 10.Maurin M., Raoult D. Intracellular organisms. Int J Antimicrob Agents. 1997;9:61–70. doi: 10.1016/s0924-8579(97)00028-9. [DOI] [PubMed] [Google Scholar]
- 11.Byrd T.F., Horwitz M.A. Chloroquine inhibits the intracellular multiplication of Legionella pneumophila by limiting the availability of iron. A potential new mechanism for the therapeutic effect of chloroquine against intracellular pathogens. J Clin Invest. 1991;88:351–357. doi: 10.1172/JCI115301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fortier A.H., Leiby D.A., Narayanan R.B. Growth of Francisella tularensis LVS in macrophages: the acidic intracellular compartment provides essential iron required for growth. Infect Immun. 1995;63:1478–1483. doi: 10.1128/iai.63.4.1478-1483.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Raoult D., Drancourt M., Vestris G. Bactericidal effect of doxycycline associated with lysosomotropic agents on Coxiella burnetii in P388D1 cells. Antimicrob Agents Chemother. 1990;34:1512–1514. doi: 10.1128/aac.34.8.1512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Crowle A.J., May M.H. Inhibition of tubercle bacilli in cultured human macrophages by chloroquine used alone and in combination with streptomycin, isoniazid, pyrazinamide, and two metabolites of vitamin D3. Antimicrob Agents Chemother. 1990;34:2217–2222. doi: 10.1128/aac.34.11.2217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boelaert J.R., Appelberg R., Gomes M.S. Experimental results on chloroquine and AIDS-related opportunistic infections. J Acquir Immune Defic Syndr. 2001;26:300–301. doi: 10.1097/00042560-200103010-00017. [DOI] [PubMed] [Google Scholar]
- 16.Horowitz H., Carbonaro C.A. Inhibition of the Salmonella typhi oral vaccine strain, Ty21a, by mefloquine and chloroquine. J Infect Dis. 1992;166:1462–1464. doi: 10.1093/infdis/166.6.1462-b. [DOI] [PubMed] [Google Scholar]
- 17.Wiseman D. The effect of pH on the inhibitory activity of chloroquine against Escherichia coli. J Pharm Pharmacol. 1972;24(Suppl.):162P. [PubMed] [Google Scholar]
- 18.Artenstein A.W., Opal S.M., Cristofaro P. Chloroquine enhances survival in Bacillus anthracis intoxication. J Infect Dis. 2004;190:1655–1660. doi: 10.1086/424853. [DOI] [PubMed] [Google Scholar]
- 19.Smith K.T., Dawes I.W. The preferential inhibition of Bacillus subtilis spore outgrowth by chloroquine. Arch Microbiol. 1989;152:251–257. doi: 10.1007/BF00409659. [DOI] [PubMed] [Google Scholar]
- 20.Brorson O., Brorson S.H. An in vitro study of the susceptibility of mobile and cystic forms of Borrelia burgdorferi to hydroxychloroquine. Int Microbiol. 2002;5:25–31. doi: 10.1007/s10123-002-0055-2. [DOI] [PubMed] [Google Scholar]
- 21.Detilleux P.G., Deyoe B.L., Cheville N.F. Effect of endocytic and metabolic inhibitors on the internalization and intracellular growth of Brucella abortus in Vero cells. Am J Vet Res. 1991;52:1658–1664. [PubMed] [Google Scholar]
- 22.Nguyen H.A., Grellet J., Paillard D., Dubois V., Quentin C., Saux M.C. Factors influencing the intracellular activity of fluoroquinolones: a study using levofloxacin in a Staphylococcus aureus THP-1 monocyte model. J Antimicrob Chemother. 2006;57:883–890. doi: 10.1093/jac/dkl079. [DOI] [PubMed] [Google Scholar]
- 23.Prada-Delgado A., Carrasco-Marin E., Pena-Macarro C. Inhibition of Rab5a exchange activity is a key step for Listeria monocytogenes survival. Traffic. 2005;6:252–265. doi: 10.1111/j.1600-0854.2005.00265.x. [DOI] [PubMed] [Google Scholar]
- 24.Newman S.L., Gootee L., Brunner G., Deepe G.S., Jr. Chloroquine induces human macrophage killing of Histoplasma capsulatum by limiting the availability of intracellular iron and is therapeutic in a murine model of histoplasmosis. J Clin Invest. 1994;93:1422–1429. doi: 10.1172/JCI117119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Levitz S.M., Harrison T.S., Tabuni A., Liu X. Chloroquine induces human mononuclear phagocytes to inhibit and kill Cryptococcus neoformans by a mechanism independent of iron deprivation. J Clin Invest. 1997;100:1640–1646. doi: 10.1172/JCI119688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dias-Melicio FPetruzzielloL.A., Moreira A.P., Calvi S.A., Soares A.M. Chloroquine inhibits Paracoccidioides brasiliensis survival within human monocytes by limiting the availability of intracellular iron. Microbiol Immunol. 2006;50:307–314. doi: 10.1111/j.1348-0421.2006.tb03798.x. [DOI] [PubMed] [Google Scholar]
- 27.Taramelli D., Tognazioli C., Ravagnani F., Leopardi O., Giannulis G., Boelaert J.R. Inhibition of intramacrophage growth of Penicillium marneffei by 4-aminoquinolines. Antimicrob Agents Chemother. 2001;45:1450–1455. doi: 10.1128/AAC.45.5.1450-1455.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jahn B., Langfelder K., Schneider U., Schindel C., Brakhage A.A. PKSP-dependent reduction of phagolysosome fusion and intracellular kill of Aspergillus fumigatus conidia by human monocyte-derived macrophages. Cell Microbiol. 2002;4:793–803. doi: 10.1046/j.1462-5822.2002.00228.x. [DOI] [PubMed] [Google Scholar]
- 29.Sperber K., Kalb T.H., Stecher V.J., Banerjee R., Mayer L. Inhibition of human immunodeficiency virus type 1 replication by hydroxychloroquine in T cells and monocytes. AIDS Res Hum Retroviruses. 1993;9:91–98. doi: 10.1089/aid.1993.9.91. [DOI] [PubMed] [Google Scholar]
- 30.Ferreira D.F., Santo M.P., Rebello M.A., Rebello M.C. Weak bases affect late stages of Mayaro virus replication cycle in vertebrate cells. J. Med. Microbiol. 2000;49(4):313–318. doi: 10.1099/0022-1317-49-4-313. [DOI] [PubMed] [Google Scholar]
- 31.Pardridge W.M., Yang J., Diagne A. Chloroquine inhibits HIV-1 replication in human peripheral blood lymphocytes. Immunol Lett. 1998;64:45–47. doi: 10.1016/s0165-2478(98)00096-0. [DOI] [PubMed] [Google Scholar]
- 32.Chiang G., Sassaroli M., Louie M., Chen H., Stecher V.J., Sperber K. Inhibition of HIV-1 replication by hydroxychloroquine: mechanism of action and comparison with zidovudine. Clin Ther. 1996;18:1080–1092. doi: 10.1016/s0149-2918(96)80063-4. [DOI] [PubMed] [Google Scholar]
- 33.Vincent M.J., Bergeron E., Benjannet S. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Keyaerts E., Vijgen L., Maes P., Neyts J., Van R.M. In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochem Biophys Res Commun. 2004;323:264–268. doi: 10.1016/j.bbrc.2004.08.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shibata M., Aoki H., Tsurumi T. Mechanism of uncoating of influenza B virus in MDCK cells: action of chloroquine. J Gen Virol. 1983;64:1149–1156. doi: 10.1099/0022-1317-64-5-1149. [DOI] [PubMed] [Google Scholar]
- 36.Savarino A., Di T.L., Donatelli I., Cauda R., Cassone A. New insights into the antiviral effects of chloroquine. Lancet Infect Dis. 2006;6:67–69. doi: 10.1016/S1473-3099(06)70361-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Miller D.K., Lenard J. Antihistaminics, local anesthetics, and other amines as antiviral agents. Proc Natl Acad Sci USA. 1981;78:3605–3609. doi: 10.1073/pnas.78.6.3605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ooi E.E., Chew J.S., Loh J.P., Chua R.C. In vitro inhibition of human influenza A virus replication by chloroquine. Virol J. 2006;3:39. doi: 10.1186/1743-422X-3-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Randolph V.B., Winkler G., Stollar V. Acidotropic amines inhibit proteolytic processing of flavivirus prM protein. Virology. 1990;174:450–458. doi: 10.1016/0042-6822(90)90099-d. [DOI] [PubMed] [Google Scholar]
- 40.Petruzziello R., Orsi N., Macchia S., Rieti S., Frey T.K., Mastromarino P. Pathway of rubella virus infectious entry into Vero cells. J Gen Virol. 1996;77(Pt 2):303–308. doi: 10.1099/0022-1317-77-2-303. [DOI] [PubMed] [Google Scholar]
- 41.Nakhasi H.L., Zheng D., Callahan L., Dave J.R., Liu T.Y. Rubella virus: mechanism of attenuation in the vaccine strain (HPV77) Virus Res. 1989;13(3):231–243. doi: 10.1016/0168-1702(89)90018-x. [DOI] [PubMed] [Google Scholar]
- 42.Bishop N.E. Examination of potential inhibitors of hepatitis A virus uncoating. Intervirology. 1998;41:261–271. doi: 10.1159/000024948. [DOI] [PubMed] [Google Scholar]
- 43.Offensperger W.B., Offensperger S., Walter E., Blum H.E., Gerok W. Inhibition of duck hepatitis B virus infection by lysosomotropic agents. Virology. 1991;183:415–418. doi: 10.1016/0042-6822(91)90157-7. [DOI] [PubMed] [Google Scholar]
- 44.Civitico G., Wang Y.Y., Luscombe C. Antiviral strategies in chronic hepatitis B virus infection: II. Inhibition of duck hepatitis B virus in vitro using conventional antiviral agents and supercoiled-DNA active compounds. J Med Virol. 1990;31:90–97. doi: 10.1002/jmv.1890310205. [DOI] [PubMed] [Google Scholar]
- 45.Blanchard E., Belouzard S., Goueslain L. Hepatitis C virus entry depends on clathrin-mediated endocytosis. J Virol. 2006;80:6964–6972. doi: 10.1128/JVI.00024-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Glushakova S.E., Lukashevich I.S. Early events in arenavirus replication are sensitive to lysosomotropic compounds. Arch Virol. 1989;104:157–161. doi: 10.1007/BF01313817. [DOI] [PubMed] [Google Scholar]
- 47.Borrow P., Oldstone M.B. Mechanism of lymphocytic choriomeningitis virus entry into cells. Virology. 1994;198(1):1–9. doi: 10.1006/viro.1994.1001. [DOI] [PubMed] [Google Scholar]
- 48.Tsiang H., Superti F. Ammonium chloride and chloroquine inhibit rabies virus infection in neuroblastoma cells Brief report. Arch Virol. 1984;81:377–382. doi: 10.1007/BF01310010. [DOI] [PubMed] [Google Scholar]
- 49.Pontesilli O., Carotenuto P., Levin M.J., Suez D., Hayward A.R. Processing and presentation of cell-associated varicella–zoster virus antigens by human monocytes. Clin Exp Immunol. 1987;70:127–135. [PMC free article] [PubMed] [Google Scholar]
- 50.Lindemans C.A., Coffer P.J., Schellens I.M., de Graaff P.M., Kimpen J.L., Koenderman L. Respiratory syncytial virus inhibits granulocyte apoptosis through a phosphatidylinositol 3-kinase and NF-κB-dependent mechanism. J Immunol. 2006;176:5529–5537. doi: 10.4049/jimmunol.176.9.5529. [DOI] [PubMed] [Google Scholar]
- 51.Cassell S., Edwards J., Brown D.T. Effects of lysosomotropic weak bases on infection of BHK-21 cells by Sindbis virus. J Virol. 1984;52:857–864. doi: 10.1128/jvi.52.3.857-864.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Singh A.K., Sidhu G.S., Friedman R.M., Maheshwari R.K. Mechanism of enhancement of the antiviral action of interferon against herpes simplex virus-1 by chloroquine. J Interferon Cytokine Res. 1996;16:725–731. doi: 10.1089/jir.1996.16.725. [DOI] [PubMed] [Google Scholar]
- 53.Koyama A.H., Uchida T. Inhibition of multiplication of herpes simplex virus type 1 by ammonium chloride and chloroquine. Virology. 1984;138(2):332–335. doi: 10.1016/0042-6822(84)90356-8. [DOI] [PubMed] [Google Scholar]
- 54.Miller N., Hutt-Fletcher L.M. Epstein–Barr virus enters B cells and epithelial cells by different routes. J Virol. 1992;66:3409–3414. doi: 10.1128/jvi.66.6.3409-3414.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zeichhardt H., Wetz K., Willingmann P., Habermehl K.O. Entry of poliovirus type 1 and Mouse Elberfeld (ME) virus into HEp-2 cells: receptor-mediated endocytosis and endosomal or lysosomal uncoating. J Gen Virol. 1985;66:483–492. doi: 10.1099/0022-1317-66-3-483. [DOI] [PubMed] [Google Scholar]
- 56.Kronenberger P., Vrijsen R., Boeye A. Chloroquine induces empty capsid formation during poliovirus eclipse. J Virol. 1991;65:7008–7011. doi: 10.1128/jvi.65.12.7008-7011.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Madshus I.H., Olsnes S., Sandvig K. Mechanism of entry into the cytosol of poliovirus type 1: requirement for low pH. J Cell Biol. 1984;98(4):1194–1200. doi: 10.1083/jcb.98.4.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yoshida T., Takao S., Kiyotani K., Sakaguchi T. Endoproteolytic activation of Newcastle disease virus fusion proteins requires an intracellular acidic environment. Virology. 1989;170:571–574. doi: 10.1016/0042-6822(89)90449-2. [DOI] [PubMed] [Google Scholar]
- 59.Gonzalez-Dunia D., Cubitt B., de la Torre J.C. Mechanism of Borna disease virus entry into cells. J Virol. 1998;72:783–788. doi: 10.1128/jvi.72.1.783-788.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Miller D.K., Lenard J. Inhibition of vesicular stomatitis virus infection by spike glycoprotein. Evidence for an intracellular, G protein-requiring step. J Cell Biol. 1980;84(2):430–437. doi: 10.1083/jcb.84.2.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fredericksen B.L., Whitt M.A. Attenuation of recombinant vesicular stomatitis viruses encoding mutant glycoproteins demonstrate a critical role for maintaining a high pH threshold for membrane fusion in viral fitness. Virology. 1998;240:349–358. doi: 10.1006/viro.1997.8921. [DOI] [PubMed] [Google Scholar]
- 62.Dille B.J., Johnson T.C. Inhibition of vesicular stomatitis virus glycoprotein expression by chloroquine. J Gen Virol. 1982;62:91–103. doi: 10.1099/0022-1317-62-1-91. [DOI] [PubMed] [Google Scholar]
- 63.Janeczko R.A., Rodriguez J.F., Esteban M. Studies on the mechanism of entry of vaccinia virus in animal cells. Arch Virol. 1987;92:135–150. doi: 10.1007/BF01310068. [DOI] [PubMed] [Google Scholar]
- 64.Pazmino N.H., Yuhas J.M., Tennant R.W. Inhibition of murine RNA tumor virus replication and oncogenesis by chloroquine. Int J Cancer. 1974;14:379–385. doi: 10.1002/ijc.2910140312. [DOI] [PubMed] [Google Scholar]
- 65.Carrillo E.C., Giachetti C., Campos R. Early steps in FMDV replication: further analysis on the effects of chloroquine. Virology. 1985;147:118–125. doi: 10.1016/0042-6822(85)90232-6. [DOI] [PubMed] [Google Scholar]
- 66.Ferreira D.F., Santo M.P., Rebello M.A., Rebello M.C. Weak bases affect late stages of Mayaro virus replication cycle in vertebrate cells. J Med Microbiol. 2000;49:313–318. doi: 10.1099/0022-1317-49-4-313. [DOI] [PubMed] [Google Scholar]
- 67.Stuart A.D., Brown T.D. Entry of feline calicivirus is dependent on clathrin-mediated endocytosis and acidification in endosomes. J Virol. 2006;80:7500–7509. doi: 10.1128/JVI.02452-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Geraldes A., Valdeira M.L. Effect of chloroquine on African swine fever virus infection. J Gen Virol. 1985;66(Pt 5):1145–1148. doi: 10.1099/0022-1317-66-5-1145. [DOI] [PubMed] [Google Scholar]
- 69.Mager A., Masengo R., Mammerickx M., Letesson J.J. T cell proliferative response to bovine leukaemia virus (BLV): identification of T cell epitopes on the major core protein (p24) in BLV-infected cattle with normal haematological values. J Gen Virol. 1994;75:2223–2231. doi: 10.1099/0022-1317-75-9-2223. [DOI] [PubMed] [Google Scholar]
- 70.Basak S., Turner H. Infectious entry pathway for canine parvovirus. Virology. 1992;186:368–376. doi: 10.1016/0042-6822(92)90002-7. [DOI] [PubMed] [Google Scholar]
- 71.Ros C., Burckhardt C.J., Kempf C. Cytoplasmic trafficking of minute virus of mice: low-pH requirement, routing to late endosomes, and proteasome interaction. J Virol. 2002;76(24):12634–12645. doi: 10.1128/JVI.76.24.12634-12645.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hackstadt T., Williams J.C. pH dependence of the Coxiella burnetii glutamate transport system. J Bacteriol. 1983;154:598–603. doi: 10.1128/jb.154.2.598-603.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mege J.L., Maurin M., Capo C., Raoult D. Coxiella burnetii: the ‘query’ fever bacterium. A model of immune subversion by a strictly intracellular microorganism. FEMS Microbiol Rev. 1997;19:209–217. doi: 10.1111/j.1574-6976.1997.tb00298.x. [DOI] [PubMed] [Google Scholar]
- 74.Maurin M., Raoult D. Q fever. Clin Microbiol Rev. 1999;12:518–553. doi: 10.1128/cmr.12.4.518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Fenollar F., Fournier P.E., Carrieri M.P., Habib G., Messana T., Raoult D. Risks factors and prevention of Q fever endocarditis. Clin Infect Dis. 2001;33:312–316. doi: 10.1086/321889. [DOI] [PubMed] [Google Scholar]
- 76.Madariaga M.G., Pulvirenti J., Sekosan M., Paddock C.D., Zaki S.R. Q fever endocarditis in HIV-infected patient. Emerg Infect Dis. 2004;10:501–504. doi: 10.3201/eid1003.030971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Senn L., Franciolli M., Raoult D. Coxiella burnetii vascular graft infection. BMC Infect Dis. 2005;5:109. doi: 10.1186/1471-2334-5-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Fenollar F., Thuny F., Xeridat B., Lepidi H., Raoult D. Endocarditis after acute Q fever in patients with previously undiagnosed valvulopathies. Clin Infect Dis. 2006;42:818–821. doi: 10.1086/500402. [DOI] [PubMed] [Google Scholar]
- 79.Landais C., Fenollar F., Thuny F., Raoult D. From acute Q fever to endocarditis: serological follow-up strategy. Clin Infect Dis. 2007;44:1337–1340. doi: 10.1086/515401. [DOI] [PubMed] [Google Scholar]
- 80.Landais C., Fenollar F., Constantin A. Q fever osteoarticular infection: four new cases and a review of the literature. Eur J Clin Microbiol Infect Dis. 2007;26:341–347. doi: 10.1007/s10096-007-0285-5. [DOI] [PubMed] [Google Scholar]
- 81.Raoult D., Birg M.L., La Scola B. Cultivation of the bacillus of Whipple's disease. New Engl J Med. 2000;342:620–625. doi: 10.1056/NEJM200003023420903. [DOI] [PubMed] [Google Scholar]
- 82.Fenollar F., Puechal X., Raoult D. Whipple's disease. New Engl J Med. 2007;256:55–66. doi: 10.1056/NEJMra062477. [DOI] [PubMed] [Google Scholar]
- 83.Rogers D.E., Tompsett R. The survival of staphylococci within human leukocytes. J Exp Med. 1952;95:209–230. doi: 10.1084/jem.95.2.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kapral F.A., Shayegani M.G. Intracellular survival of staphylococci. J Exp Med. 1959;110:123. doi: 10.1084/jem.110.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Craven N., Williams M.R., Field T.R., Bunch K.J., Mayer S.J., Bourne S.J. The influence of extracellular and phagolysosomal pH changes on the bactericidal activity of bovine neutrophils against Staphylococcus aureus. Vet Immunol Immunopathol. 1986;13:97–110. doi: 10.1016/0165-2427(86)90052-8. [DOI] [PubMed] [Google Scholar]
- 86.Styrt B., Klempner M.S. Modification of interactions between neutrophils and staphylococci by lysosomotropic week bases. Infect Immun. 1985;50:415–419. doi: 10.1128/iai.50.2.415-419.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Yancey R.J., Sanchez M.S., Ford C.W. Activity of antibiotics against Staphylococcus aureus within polymorphonuclear neutrophils. Eur J Clin Microbiol Infect Dis. 1991;10:107–113. doi: 10.1007/BF01964421. [DOI] [PubMed] [Google Scholar]
- 88.Lam C., Mathison G.E. Effect of low intraphagolysosomal pH on antimicrobial activity of antibiotics against ingested staphylococci. J Med Microbiol. 1983;16:309–316. doi: 10.1099/00222615-16-3-309. [DOI] [PubMed] [Google Scholar]
- 89.Maurin M., Raoult D. Phagolysosomal alkalinization and intracellular killing of Staphylococcus aureus by amikacin. J Infect Dis. 1994;169:330–336. doi: 10.1093/infdis/169.2.330. [DOI] [PubMed] [Google Scholar]
- 90.Nguyen H.A., Grellet J., Dubois V., Saux M.C., Quentin C. Factors compromising the activity of moxifloxacin against intracellular Staphylococcus aureus. J Antimicrob Chemother. 2007;59:755–758. doi: 10.1093/jac/dkm004. [DOI] [PubMed] [Google Scholar]
- 91.Sanchez M.S., Ford C.W., Yancey R.J., Jr. Evaluation of antibiotic effectiveness against Staphylococcus aureus surviving within the bovine mammary gland macrophage. J Antimicrob Chemother. 1988;21:773–786. doi: 10.1093/jac/21.6.773. [DOI] [PubMed] [Google Scholar]
- 92.Tulkens P.M. Intracellular distribution and activity of antibiotics. Eur J Clin Microbiol Infect Dis. 1991;10:100–106. doi: 10.1007/BF01964420. [DOI] [PubMed] [Google Scholar]
- 93.Mandell G.L., Vest T.K. Killing of intraleukocytic Staphylococcus aureus by rifampin: in vitro and in vivo studies. J Infect Dis. 1972;125:486–490. doi: 10.1093/infdis/125.5.486. [DOI] [PubMed] [Google Scholar]
- 94.Stout J.E., Yu V.L. Legionellosis. N Engl J Med. 1997;337:682–687. doi: 10.1056/NEJM199709043371006. [DOI] [PubMed] [Google Scholar]
- 95.Weber S.M., Levitz S.M., Harrison T.S. Chloroquine and the fungal phagosome. Curr Opin Microbiol. 2000;3:349–353. doi: 10.1016/s1369-5274(00)00102-8. [DOI] [PubMed] [Google Scholar]
- 96.Harrison T.S., Chen J., Simons E., Levitz S.M. Determination of the pH of the Cryptococcus neoformans vacuole. Med Mycol. 2002;40:329–332. doi: 10.1080/mmy.40.3.329.332. [DOI] [PubMed] [Google Scholar]
- 97.Eissenberg L.G., Goldman W.E., Schlesinger P.H. Histoplasma capsulatum modulates the acidification of phagolysosomes. J Exp Med. 1995;177:1605–1611. doi: 10.1084/jem.177.6.1605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Strasser J.E., Newman S.L., Ciraolo G.M., Morris R.E., Howell M.L., Dean G.E. Regulation of the macrophage vacuolar ATPase and phagosome–lysosome fusion by Histoplasma capsulatum. J Immunol. 1999;162:6148–6154. [PubMed] [Google Scholar]
- 99.Schafer M.P., Dean G.E. Cloning and sequence analysis of an H(+)-ATPase-encoding gene from the human dimorphic pathogen Histoplasma capsulatum. Gene. 1993;136:295–300. doi: 10.1016/0378-1119(93)90483-j. [DOI] [PubMed] [Google Scholar]
- 100.Levitz S.M., Nong S.H., Seetoo K.F., Harrison T.S., Speizer R.A., Simons E.R. Cryptococcus neoformans resides in an acidic phagolysosome of human macrophages. Infect Immun. 1999;67:885–890. doi: 10.1128/iai.67.2.885-890.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Sieczkarski S.B., Whittaker G.R. Dissecting virus entry via endocytosis. J Gen Virol. 2002;83:1535–1545. doi: 10.1099/0022-1317-83-7-1535. [DOI] [PubMed] [Google Scholar]
- 102.Naarding M.A., Baan E., Pollakis G., Paxton W.A. Effect of chloroquine on reducing HIV-1 replication in vitro and the DC-SIGN mediated transfer of virus to CD4+ T-lymphocytes. Retrovirology. 2007;4:6. doi: 10.1186/1742-4690-4-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kwiek J.J., Haystead T.A., Rudolph J. Kinetic mechanism of quinone oxidoreductase 2 and its inhibition by the antimalarial quinolines. Biochemistry. 2004;43:4538–4547. doi: 10.1021/bi035923w. [DOI] [PubMed] [Google Scholar]
- 104.Ducharme J., Farinotti R. Clinical pharmacokinetics and metabolism of chloroquine. Focus on recent advancements. Clin Pharmacokinet. 1996;31:257–274. doi: 10.2165/00003088-199631040-00003. [DOI] [PubMed] [Google Scholar]
- 105.Boelaert J.R., Sperber K., Piette J. The additive in vitro anti-HIV-1 effect of chloroquine, when combined with zidovudine and hydroxyurea. Biochem Pharmacol. 2001;61:1531–1535. doi: 10.1016/s0006-2952(01)00576-7. [DOI] [PubMed] [Google Scholar]
- 106.Witvrouw M., Pannecouque C., Switzer W.M., Folks T.M., De C.E., Heneine W. Susceptibility of HIV-2, SIV and SHIV to various anti-HIV-1 compounds: implications for treatment and postexposure prophylaxis. Antivir Ther. 2004;9:57–65. [PubMed] [Google Scholar]
- 107.Sperber K., Louie M., Kraus T. Hydroxychloroquine treatment of patients with human immunodeficiency virus type 1. Clin Ther. 1995;17:622–636. doi: 10.1016/0149-2918(95)80039-5. [DOI] [PubMed] [Google Scholar]
- 108.Sperber K., Chiang G., Chen H. Comparison of hydroxychloroquine with zidovudine in asymptomatic patients infected with human immunodeficiency virus type 1. Clin Ther. 1997;19:913–923. doi: 10.1016/s0149-2918(97)80045-8. [DOI] [PubMed] [Google Scholar]
- 109.Paton N.I., Aboulhab J., Karim F. Hydroxychloroquine, hydroxycarbamide, and didanosine as economic treatment for HIV-1. Lancet. 2002;359:1667–1668. doi: 10.1016/S0140-6736(02)08557-4. [DOI] [PubMed] [Google Scholar]
- 110.Paton N.I., Aboulhab J. Hydroxychloroquine, hydroxyurea and didanosine as initial therapy for HIV-infected patients with low viral load: safety, efficacy and resistance profile after 144 weeks. HIV Med. 2005;6:13–20. doi: 10.1111/j.1468-1293.2005.00259.x. [DOI] [PubMed] [Google Scholar]
- 111.Joshi S.R., Butala N., Patwardhan M.R., Daver N.G., Kelkar D. Low cost anti-retroviral options: chloroquine based ARV regimen combined with hydroxyurea and lamivudine: a new economical triple therapy. J Assoc Physicians India. 2004;52:597–598. [PubMed] [Google Scholar]
- 112.Engchanil C., Kosalaraksa P., Lumbiganon P. Therapeutic potential of chloroquine added to zidovudine plus didanosine for HIV-1 infected children. J Med Assoc Thai. 2006;89:1229–1236. [PubMed] [Google Scholar]
- 113.Kourtis A.P., Lee F.K., Abrams E.J., Jamieson D.J., Bulterys M. Mother-to-child transmission of HIV-1: timing and implications for prevention. Lancet Infect Dis. 2006;6:726–732. doi: 10.1016/S1473-3099(06)70629-6. [DOI] [PubMed] [Google Scholar]
- 114.Savarino A., Boelaert J.R., Cassone A., Majori G., Cauda R. Effects of chloroquine on viral infections: an old drug against today's diseases? Lancet Infect Dis. 2003;3:722–727. doi: 10.1016/S1473-3099(03)00806-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Malaty L.I., Kuper J.J. Drug interactions of HIV protease inhibitors. Drug Saf. 1999;20(2):147–169. doi: 10.2165/00002018-199920020-00005. [DOI] [PubMed] [Google Scholar]
- 116.Projean D., Baune B., Farinotti R. In vitro metabolism of chloroquine: identification of CYP2C8, CYP3A4, and CYP2D6 as the main isoforms catalyzing N-desethylchloroquine formation. Drug Metab Dispos. 2003;31:748–754. doi: 10.1124/dmd.31.6.748. [DOI] [PubMed] [Google Scholar]
- 117.Li W., Moore M.J., Vasilieva N. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Gallagher T.M., Escarmis C., Buchmeier M.J. Alteration of the pH dependence of coronavirus-induced cell fusion: effect of mutations in the spike glycoprotein. J Virol. 1991;65:1916–1928. doi: 10.1128/jvi.65.4.1916-1928.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Simmons G., Reeves J.D., Rennekamp A.J., Amberg S.M., Piefer A.J., Bates P. Characterization of severe acute respiratory syndrome-associated coronavirus (SARS-CoV) spike glycoprotein-mediated viral entry. Proc Natl Acad Sci USA. 2004;101:4240–4245. doi: 10.1073/pnas.0306446101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Yang Z.Y., Huang Y., Ganesh L. pH-dependent entry of severe acute respiratory syndrome coronavirus is mediated by the spike glycoprotein and enhanced by dendritic cell transfer through DC-SIGN. J Virol. 2004;78:5642–5650. doi: 10.1128/JVI.78.11.5642-5650.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Biot C., Daher W., Chavain N. Design and synthesis of hydroxyferroquine derivatives with antimalarial and antiviral activities. J Med Chem. 2006;49:2845–2849. doi: 10.1021/jm0601856. [DOI] [PubMed] [Google Scholar]
- 122.Yoshimura A., Kuroda K., Kawasaki K., Yamashina S., Maeda T., Ohnishi S. Infectious cell entry mechanism of influenza virus. J Virol. 1982;43:284–293. doi: 10.1128/jvi.43.1.284-293.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Hirschman S.Z., Garfinkel E. Inhibition of hepatitis B DNA polymerase by intercalating agents. Nature. 1978;271:681–683. doi: 10.1038/271681a0. [DOI] [PubMed] [Google Scholar]
- 124.Hagelstein J., Fathinejad F., Stremmel W., Galle P.R. pH-independent uptake of hepatitis B virus in primary human hepatocytes. Virology. 1997;229:292–294. doi: 10.1006/viro.1996.8376. [DOI] [PubMed] [Google Scholar]
- 125.Kouroumalis E.A., Koskinas J. Treatment of chronic active hepatitis B (CAH B) with chloroquine: a preliminary report. Ann Acad Med Singapore. 1986;15:149–152. [PubMed] [Google Scholar]
- 126.Accapezzato D., Visco V., Francavilla V. Chloroquine enhances human CD8+ T cell responses against soluble antigens in vivo. J Exp Med. 2005;202:817–828. doi: 10.1084/jem.20051106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Rolain J.M., Mallet M.N., Raoult D. Correlation between serum levels of doxycycline and serology evolution in patients treated for Coxiella burnetii endocarditis. J Infect Dis. 2003;9:1322–1325. doi: 10.1086/379082. [DOI] [PubMed] [Google Scholar]
- 128.Rolain J.M., Boulos A., Mallet M.N., Raoult D. Correlation between ratio of serum doxycycline concentration to MIC and rapid decline of antibody levels during treatment of Q fever endocarditis. Antimicrob Agents Chemother. 2005;49:2673–2676. doi: 10.1128/AAC.49.7.2673-2676.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Klinger G., Morad Y., Westall C.A. Ocular toxicity and antenatal exposure to chloroquine or hydroxychloroquine for rheumatic diseases. Lancet. 2001;358:813–814. doi: 10.1016/S0140-6736(01)06004-4. [DOI] [PubMed] [Google Scholar]
- 130.Bernstein H.N. Ophthalmologic considerations and testing in patients receiving long-term antimalarial therapy. Am J Med. 1983;75:25–34. doi: 10.1016/0002-9343(83)91267-6. [DOI] [PubMed] [Google Scholar]