Abstract
Cancer prevention and treatment using traditional Chinese medicines have attracted increasing interest. This study characterizes antioxidant activity and phenolic compounds of traditional Chinese medicinal plants associated with anticancer, comprising 112 species from 50 plant families. The improved ABTS•+ method was used to systematically assess the total antioxidant capacity (Trolox equivalent antioxidant capacity, TEAC) of the medicinal extracts. The TEAC values and total phenolic content for methanolic extracts of herbs ranged from 46.7 to 17,323 μmol Trolox equivalent/100 g dry weight (DW), and from 0.22 to 50.3 g of gallic acid equivalent/100 g DW, respectively. A positive, significant linear relationship between antioxidant activity and total phenolic content (all R2 values≥0.95) showed that phenolic compounds were the dominant antioxidant components in the tested medicinal herbs. Major types of phenolic compounds from most of the tested herbs were preliminarily identified and analyzed, and mainly included phenolic acids, flavonoids, tannins, coumarins, lignans, quinones, stilbenes, and curcuminoids. These medicinal herbs exhibited far stronger antioxidant activity and contained significantly higher levels of phenolics than common vegetables and fruits. Traditional Chinese medicinal plants associated with anticancer might be potential sources of potent natural antioxidants and beneficial chemopreventive agents.
Keywords: Antioxidant activity, Phenolic compounds, Anticancer, Traditional Chinese Medicines, ABTS, Phenolic acids, Flavonoids, Tannins, Quinones, Free radical
Introduction
Numerous physiological and biochemical processes in the human body may produce oxygen-centered free radicals and other reactive oxygen species as byproducts. Overproduction of such free radicals can cause oxidative damage to biomolecules (e.g. lipids, proteins, DNA), eventually leading to many chronic diseases, such as atherosclerosis, cancer, diabetes, aging, and other degenerative diseases in humans Halliwell, 1994, Niki, 1997, Poulson et al., 1998. Plants (fruits, vegetables, medicinal herbs, etc.) may contain a wide variety of free radical scavenging molecules, such as phenolic compounds (e.g. phenolic acids, flavonoids, quinones, coumarins, lignans, stilbenes, tannins), nitrogen compounds (alkaloids, amines, betalains), vitamins, terpenoids (including carotenoids), and some other endogenous metabolites, which are rich in antioxidant activity Larson, 1988, Shahidi and Naczk, 1995, Cotelle et al., 1996, Velioglu et al., 1998, Zheng and Wang, 2001, Cai et al., 2003. Epidemiological studies have shown that many of these antioxidant compounds possess anti-inflammatory, antiatherosclerotic, antitumor, antimutagenic, anticarcinogenic, antibacterial, or antiviral activities to a greater or lesser extent Halliwell, 1994, Mitscher et al., 1996, Owen et al., 2000, Sala et al., 2002. The intake of natural antioxidants has been associated with reduced risks of cancer, cardiovascular disease, diabetes, and other diseases associated with ageing, but there is still considerable controversy in this area Hertog et al., 1995, Kuo, 1997, Mclarty, 1997, Yang et al., 2001, Sun et al., 2002.
There are as yet no extremely effective drugs to treat most cancers; moreover many cancer treatments are very expensive. During the 1960s the National Cancer Institute (USA) began to screen plant extracts with antitumor activity (Monks et al., 2002). Natural compounds isolated from medicinal plants, as rich sources of novel anticancer drugs, have been of increasing interest since then. Traditional Chinese medicine has been used for pharmaceutical and dietary therapy for several millennia. The earliest medical monograph of China now extant, Yellow Emperor’s Canon of Medicine which was compiled around 475–221 B.C., used a term meaning “tumor” (Xu et al., 2000). Formal documentation of “cancer” appeared in the 12th century A.D. Chinese medicine has its own traditional theory of prevention and treatment of diseases including cancer with prescriptions containing several medicinal herbs, seldom with a single medicinal herb. A number of anticancer Chinese medicinal herbs and many relevant prescriptions have been screened and used for treating and preventing various cancers during long-term folk practice. Nowadays therapies with anticancer medicinal plants are still widely practised particularly in the countryside and remote mountainous regions, and even in the major cities of China.
Four recently published monographs about anticancer Chinese medicines Zhang and Gong, 1998, Ji et al., 1999, Xu et al., 2000, Bo et al., 2002 summarized and documented more than 400 species of traditional Chinese medicinal herbs (TCM) associated with anticancer, on the basis of long-term practical experience of TCM for anticancer purpose. Most of the documented anticancer herbal medicines and the relevant prescriptions have been verified in clinical reports and experimental research to be effective for treatment and prevention of cancer to a greater or lesser extent. The 112 traditional Chinese medicinal plants tested in this study were selected from those listed in these four references. The tested medicinal plants are commonly used as herbal medicines for anticancer purposes in China.
Although the tested traditional Chinese medicinal plants in this study are associated with anticancer, further scientific evidence is still required in many cases to verify the effects. Traditional Chinese medicine has a long history, but it has been mainly practiced through clinical treatment and its chemical and pharmacological bases are not well understood in most cases. Because of rapid development and employment of modern analytical equipment and technology, natural active components with anticancer activity or anticarcinogenic and antimutagenic effects have been studied and identified from Chinese medicinal plants Lien and Li, 1985, Ohigashi et al., 1994, Tsai, 2001, Xiao et al., 2000. They belong to various categories based on their chemical structures. The most commonly found active constituents include terpenes (sesquiterpenes, diterpenes, triterpenes), alkaloids, coumarins, lignans, quinones, flavonoids, tannins, stilbenes, curcuminoids, polysaccharides, etc. Some of them are alleged to have antioxidant activity Larson, 1988, Ho et al., 1994, Ng et al., 2000, Xiao et al., 2000, Mau et al., 2002. Antioxidant activity is a fundamental property important for human life. Many of the biological functions, including antimutagenicity, anticarcinogenicity, and antiaging, among others, may originate from this property Mclarty, 1997, Niki, 1997, Yang et al., 2001.
It is important to select and employ a stable and rapid method to assay antioxidant activity, because the determination of hundreds of samples is time-consuming. Several methods have been developed to assay free radical scavenging capacity and total antioxidant activity of plant extracts. The most common and reliable method involves the determination of the disappearance of free radicals using a spectrophotometer, such as 2,2-azinobis (3-ethylbenzothiazoline-6-sulfonic) acid radical (ABTS•+) and 1,1-diphenyl-2-picrylhydrazyl radical (DPPH•). Miller et al. (1993) first developed the ABTS•+ method and Brand-Williams et al. (1995) established the DPPH• method. Reduction of ABTS•+ or DPPH• by a radical species (R) (ABTS•+ or DPPH•+R• → ABTS-H DPPH-R) or by an antioxidant (A) (ABTS•+ or DPPH•+A → ABTS-H or DPPH-H + A•) causes a loss of absorbance at 734 nm or 515 nm, respectively. The two original methods have been modified and improved in recent years. Specially, Re et al. (1999) improved the traditional ABTS•+ method based on the activation of metmyoglobin with hydrogen peroxide in the presence of ABTS•+ to produce the radical cation. The improved method directly generated the blue/green ABTS•+ chromophore through the reaction between ABTS and potassium persulfate. This new method has been successfully developed and applied in this study for assaying total antioxidant activity of medicinal plant extracts on a large scale.
Information on antioxidant activity and phenolic compounds of traditional Chinese medicinal plants associated with anticancer is scarce. So far no large-scale systematical survey of the antioxidant activity and phenolic compounds of these medicinal herbs has been made. Therefore, the main objectives of this study were: 1) to assay antioxidant activity of the 112 selected Chinese medicinal herbs associated with anticancer using the improved ABTS•+ method, and to determine their total phenolic content; 2) to statistically investigate the inter-relationship between phenolic content and antioxidant activity; 3) to identify and analyze major phenolic antioxidant components from these medicinal herbs.
Materials and methods
Collection of traditional Chinese medicinal plants associated with anticancer
The traditional Chinese medicinal plants associated with anticancer in this study were selected and collected from those listed in the four references Zhang and Gong, 1998, Ji et al., 1999, Xu et al., 2000, Bo et al., 2002. In total, 112 species in 50 plant families (Table 1) from various locations in China were purchased and collected with 200–250 g per sample, from a well-known trade market for Chinese herbal medicines in Qichun, Hubei, China, and obtained from the Hubei Institute of Traditional Chinese Medicine. These medicinal plants were traditionally harvested and processed and naturally dried. Their roots, stems or barks, leaves, flowers, fruits/seeds, and even whole plants can be used for medicine. Planting, harvesting, processing, drying, and storage of the traditional Chinese medicines from various places are usually conducted according to strict traditional procedures. The scientific names and medicinally used parts are detailed in Table 1. Also, 14 common vegetables and fruits as reference plants (dietary plants) in Table 2 were purchased from a local supermarket in Hong Kong and immediately freeze-dried. They were imported from Mainland China, USA, New Zealand, Japan, and Southern Asia.
Table 1.
Antioxidant activity, total phenolic content, and major phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancera
| Family and scientific name | Medicinally used parts | TEAC (μmol trolox/100g DW)b |
Total phenolic content (g/100g DW)c |
Major types (representative components) of phenolic compounds | ||
|---|---|---|---|---|---|---|
| Methanolic extracts | Aqueous extracts | Methanolic extracts | Aqueous extracts | |||
| Anacardiaceae | ||||||
| Rhus chinensis Mill. or R. potaninii Maxim. | Gall | 17323.0 | 17673.8 | 50.3 | 50.2 | Hydrolyzable tannins (gallotannin), phenolic acids (gallic acid) |
| Aristolochiaceae | ||||||
| Aristolochia mollissima Hance | Whole plant | 88.7 | 122.3 | 0.42 | 0.58 | Phenolic acids (vanillic acid) |
| Berberidaceae | ||||||
| Epimedium brevicornum Maxim. | Aerial parts | 420.6 | 330.2 | 2.78 | 1.24 | Flavonols (epimedins, quercetin and their glycosides), lignans |
| Bignoniaceae | ||||||
| Campsis radicans (L.) Seem. | Flower | 446.2 | 317.9 | 2.17 | 1.63 | Flavonoids (quercetin, apigenin, cyandin-3-rutoside), phenolic acids (ferulic acid) |
| Boraginaceae | ||||||
| Lithospermum erythrorhizon Sieb. et Zucc. | Root | 413.9 | 153.4 | 0.92 | 0.52 | Naphthoquinones (shikonin, acetylskikonin, alkannan) |
| Campanulaceae | ||||||
| Lobelia chinensis Lour. | Whole plant | 57.5 | 97.8 | 0.39 | 0.60 | Flavonol glycosides |
| Caprifoliaceae | ||||||
| Lonicera japonica Thunb. | Floral bud | 589.1 | 418.8 | 3.63 | 1.51 | Phenolic acids (chlorgenic acid), flavones (luteolin, luteolin-7-glucoside) |
| Caryophyllaceae | ||||||
| Dianthus superbus L. | Aerial parts | 91.3 | 99.3 | 0.59 | 0.69 | Not identified |
| Celastraceae | ||||||
| Tripterygium wilfordii Hook. F. | Root | 1858.3 | 966.9 | 6.58 | 4.42 | Phenolic terpenes (triptophenolide, triptophenolide methyl ether) |
| Combretaceae | ||||||
| Terminalia chebula Retz. | Fruit | 4403.0 | 5517.2 | 13.16 | 15.18 | Tannins (ellagitannins), phenolic acids (gallic acid) |
| Compositae | ||||||
| Arctium lappa L. | Fruit | 513.3 | 106.9 | 3.74 | 0.82 | Phenolic acids (chlorogenic acid), Lignans (arctigenin, arctiin) |
| Artemisia annua L. | Aerial parts | 582.3 | 629.4 | 3.51 | 3.44 | Flavones (luteolin, luteolin-7-glucoside), Flavonols (kaempferol, quercetin, rutin), coumarins (coumarin, 6,7-dimethoxy-coumarin) |
| Artemisia argyi Lévl. et Vant. | Leaves | 328.5 | 378.9 | 1.94 | 1.29 | Flavones (apigenin, eupatilin, chrysoeriol) |
| Artemisia capillaris Thunb. | Seedling | 395.2 | 501.4 | 2.63 | 1.65 | Coumarins (6,7-dimethoxy-coumarin), phenolic acids (chlorogenic acid, caffeic acid) |
| Aster tataricus L. | Root | 81.8 | 83.0 | 0.60 | 0.46 | Flavonols (quercetin, quercitrin) |
| Atractylodes lancea (Thunb.) DC. | Rhizome | 89.3 | 52.8 | 0.71 | 0.24 | Not identified |
| Carthamus tinctorius L. | Inflorescence | 318.0 | 207.4 | 2.12 | 1.32 | Flavonoids (chalcones: carthamin, carthamone), lignans |
| Centipeda minima (L.) A. Br. et Aschers. | Aerial parts | 72.8 | 134.8 | 0.29 | 0.76 | Flavonol glycosides |
| Chrysanthemum indicum L. | Inflorescence | 303.6 | 292.6 | 2.01 | 1.04 | Flavones (acaciin, luteolin) and flavone glycosides, coumarins |
| Chrysanthemum morifolium Ramat. | Inflorescence | 731.6 | 492.8 | 4.76 | 3.16 | Flavones (acacetin, luteolin-7-glucoside) |
| Cirsium setosum (Willd.) Kitam. | Root | 257.8 | 146.5 | 0.71 | 0.47 | Flavonols (rutin), phenolic acids (caffeic acid, chlorogenic acid) |
| Eclipta prostrata L. | Aerial parts | 54.3 | 148.1 | 0.29 | 0.82 | Coumarins (wedelolactone), tannins |
| Inula Britannica L. | Inflorescence | 356.3 | 379.4 | 2.37 | 1.23 | Flavonols (quercetin and its glycosides), phenolic acids (caffeic acid, chlorogenic acid) |
| Taraxacum mongolicum Hand.–Mazz. | Whole plant | 142.0 | 281.0 | 0.50 | 1.06 | Flavonols (rutin) |
| Xanthium sibiricum Patr. | Fruit | 138.6 | 116.2 | 1.02 | 0.47 | Phenolic acids (cinnamic acid), phenolic volatile oils |
| Convolvulaceae | ||||||
| Cuscuta chinensis Lam. | Seed | 141.4 | 194.0 | 1.32 | 1.36 | Flavonols (quercetin, astragalin), furofuran lignans, coumarins, tannins |
| Cornaceae | ||||||
| Cornus officinalis Sieb. et Zucc. | Fruit | 651.8 | 578.6 | 2.30 | 0.96 | Phenolic acids (gallic acid), flavonols (kaempferol, quercetin and their glycosides), tannins (cornus-tannins) |
| Crassulaceae | ||||||
| Sedum sarmentosum Bge. | Aerial parts | 86.7 | 113.7 | 0.87 | 0.65 | Not identified |
| Cruciferae | ||||||
| Isatis indigotica Fort. | Root | 63.3 | 80.7 | 0.45 | 0.36 | Anthraquinones |
| Cucurbitaceae | ||||||
| Gynostemma pentaphyllum (Thunb.) Mak. | Whole plant | 84.6 | 43.9 | 0.44 | 0.24 | Flavonols (rutin) |
| Trichosanthes kirilowii Maxim. | Peel | 69.8 | 113.8 | 0.50 | 0.75 | Not identified |
| Gramineae | ||||||
| Imperata cylindrica (L.) Beauv. var. major. (Nees). C. E. Hubb | Root | 98.2 | 87.5 | 0.65 | 0.25 | Not identified |
| Juglandaceae | ||||||
| Juglans regia L. | Seed | 279.1 | 75.5 | 0.74 | 0.40 | Tannins |
| Labiatae | ||||||
| Lycopus lucidus Turcz. | Aerial parts | 226.7 | 334.8 | 0.93 | 0.88 | Flavones (acaciin, linarin), phenolic acids (chlorogenic acid) |
| Prunella vulgaris L. | Inflorescence | 910.1 | 1045.1 | 4.73 | 4.05 | Flavonols (rutin), anthocyanins (cyanidin, delphinidin), phenolic acids (caffeic acid), tannins |
| Salvia miltiorrhiza Bge. | Root | 761.5 | 867.6 | 4.26 | 4.91 | Phenanthraquinones (tanshinone I, tanshinone IIA and IIB), flavonoids (baicalin), phenolic acids (protocatechuic acid) |
| Scutellaria baicalensis Georgi | Root | 1323.5 | 507.0 | 8.17 | 2.57 | Flavones (baicalein, baicalin, chrysin, wogonin) |
| Scutellaria barbata D. Don | Whole plant | 155.3 | 223.5 | 1.37 | 1.43 | Flavones (baicalein, baicalin apigenin) |
| Lauraceae | ||||||
| Cinnamomum cassia Presl | Bark | 570.6 | 415.8 | 2.83 | 1.87 | Phenolic acids (cinnamic acid, protocatechuric acid), coumarin, tannins |
| Leguminosae | ||||||
| Acacia catechu (L.) Willd. | Branch, stem | 11197.1 | 10049.0 | 45.70 | 41.94 | Hydrolyzable and condensed tannins, flavan-3-ols ((+)-catechin, epicatechin) |
| Astragalus complanatus R. Br. | Seed | 119.5 | 111.0 | 0.98 | 0.69 | Flavonols (astragalin, myricetin, kaemferol glycosides) |
| Astragalus mongholicus Bge. | Root | 120.8 | 158.9 | 0.77 | 0.89 | Flavonoids (astragalin, calycosin, formononetin) |
| Caesalpinia sappan L. | Heartwood | 1301.0 | 629.8 | 7.92 | 4.58 | Chalcones, flavonols (quercetin), tannins |
| Cassia tora L. | Seed | 290.7 | 70.0 | 1.20 | 0.34 | Anthraquinones (chrysophanol, emodin, aloe-emodin, rhein) |
| Glycyrrhiza uralensis Fisch. | Root | 545.9 | 124.1 | 2.92 | 0.89 | Flavanones (dihydroflavones: liquiritin, liquirigenin), licopyranocoumarins |
| Psoralea corylifolia L. | Fruit | 541.3 | 109.3 | 2.43 | 0.67 | Coumarins (psorulen, psoralidin), flavonoids (bavachin, bavachinin, bavachalcone) |
| Sophora flavescens Ait. | Root | 413.1 | 121.8 | 2.08 | 0.60 | Flavonoids (kushenol, kurarinone, kuraridin, formononetin) |
| Sophora japonica L. | Flower | 1850.3 | 1978.6 | 5.95 | 6.07 | Flavonols (rutin, quercetin, kaempferol) |
| Sophora tonkinensis Gapnep. | Root | 137.7 | 93.8 | 1.55 | 0.54 | Flavonoids (sophoraclin, genistein) |
| Spatholobus suberectus Dunn | Stem | 1995.2 | 1047.4 | 8.63 | 4.80 | Flavonoids (formononetin, daidzein, ononin, epicatechin), phenolic acid (protocatechuic acid) |
| Liliaceae | ||||||
| Smilax glabra Roxb. | Root | 137.3 | 75.5 | 0.74 | 0.30 | Flavan-3-ols (epicatechin), tannins |
| Loranthaceae | ||||||
| Viscum coloratum (Kom.) Nakai | Stem, branch | 559.8 | 351.5 | 2.68 | 1.49 | Flavonols (avicalarin, quercetin), flavanols ((+)-catechin) |
| Magnoliaceae | ||||||
| Magnolia officinalis Rehd. et Wils. | Bark | 727.5 | 218.0 | 3.15 | 1.35 | Neolignans (magnolol, isomagnolol), tannins |
| Schisandra chinensis (Turcz.) Baill. | Fruit | 253.6 | 58.4 | 1.05 | 0.19 | Lignans (schizandrins, schizatherins, wulignan) |
| Moraceae | ||||||
| Morus alba L. | Fruit | 132.6 | 391.2 | 1.22 | 1.96 | Flavonols (rutin), stilbenes (oxyresveratrol) |
| Oleaceae | ||||||
| Forsythia suspensa (Thunb.) Vahl. | Fruit | 640.5 | 506.8 | 4.29 | 1.29 | Lignans (forsythigenol, forsythin), coumarins (6,7-dimethoxy-coumarin), flavonols (rutin) |
| Ligustrum lucidum Ait. | Fruit | 234.4 | 370.2 | 1.76 | 2.65 | Phenolic (secoiridoid) glycosides (nuzhenide, oleuropein) |
| Onocleaceae | ||||||
| Matteuccia struthiopteris (L.) Todaro | Rhizome | 2176.4 | 759.3 | 10.08 | 2.87 | m-benzo-triphenol derivatives (filicic acids), tannins |
| Orchidaceae | ||||||
| Dendrobium nobile Lindl. | Stem | 490.2 | 99.5 | 2.22 | 0.65 | Phenanthraquinones (denbinobin) |
| Palmae | ||||||
| Areca catechu L. | Seed | 2674.0 | 1997.3 | 11.83 | 7.91 | Condensed tannins (leucoanthocyanidins), flavanols (catechin) |
| Plantaginaceae | ||||||
| Plantago asiatica L. | Seed | 47.7 | 122.7 | 0.30 | 0.85 | Flavones (plantaginin) |
| Polygalaceae | ||||||
| Polygala tenuifolia Willd. | Root | 133.8 | 85.1 | 0.82 | 0.99 | Lignans (chinensin), phenolic acids (cinnamic acid) |
| Fagopyrum cymosum (Trev.) Meisn. | Rhizome | 1628.7 | 637.6 | 6.67 | 1.69 | Phenolic acids (p-coumaric acid, ferulic acid), flavonols (rutin), tannins |
| Polygonum aviculare L. | Whole plant | 856.8 | 432.0 | 4.13 | 1.57 | Flavonols (avicularin, quercitin), phenolic acids (gallic acid, caffeic acid), anthraquinones (emodin), tannins |
| Polygonum cuspidatum Sieb. et Zucc. | Rhizome | 1849.0 | 1055.3 | 8.08 | 4.54 | Anthraquinones (emodin, rhein, physcion, anthraglycosides), stilbenes (resveratrol), phenolic acids (coumaric acid), tannins |
| Polygonum multiflorum Thunb. | Root | 1811.9 | 871.0 | 5.67 | 4.18 | Anthraquinones (chrysophanol, emodin, rhein, physcion), tannins, stilbenes (resveratrol) |
| Polygonum orientale L. | Fruit | 255.3 | 83.8 | 1.23 | 0.45 | Flavonols (quercetin), flavanones (taxifolin) |
| Rheum officinale Baill. | Root | 2108.6 | 1512.3 | 8.37 | 5.07 | Anthraquinones (emodin, chrysophanol, rhein, physcion and their glycosides), phenolic acids (gallic acid), hydrolyzable tannins |
| Polypodiaceae | ||||||
| Drynaria fortunei (Kunze) J. Sm. | Root | 128.7 | 124.9 | 0.84 | 0.73 | Flavanones (dihydroflavonone: naringin) |
| Pyrrosia lingua (Thunb.) Farwell | Leaf | 233.0 | 229.3 | 1.65 | 2.04 | Phenolic acids (caffeic acid), anthraquinones, tannins |
| Portulacaceae | ||||||
| Portulaca oleracea L. | Aerial parts | 102.7 | 141.0 | 0.44 | 0.60 | Flavonols (kaempferol, quercetin), flavones (apigenin, luteolin) |
| Pteridaceae | ||||||
| Pteris multifida Pior. | Whole plant | 103.0 | 136.7 | 0.72 | 0.78 | Flavonols (quercetin), tannins |
| Punicaceae | ||||||
| Punica granatum L. | Peel | 6240.2 | 6992.2 | 22.56 | 20.66 | Tannins (ellagitannins), flavonols (quercetin), phenolic acids (gallic acid, chlorogenic acid) |
| Ranunculaceae | ||||||
| Aconitum carmichaeli Debx. | Root | 67.6 | 115.0 | 0.50 | 0.72 | Phenolic alkaloids (demethylsalsoline) |
| Aconitum kusnezoffii Reichb. | Root | 149.6 | 219.4 | 1.29 | 0.95 | Not identified |
| Cimicifuga dahurica (Thurcz.) Maxim. | Rhizome | 191.4 | 208.7 | 0.76 | 1.66 | Phenolic acids (ferulic acid, caffeic acid), tannins |
| Clematis chinensis Osbeck | Root | 95.8 | 98.7 | 0.55 | 0.47 | Coumarins (pyranocoumarin), other phenolics (syringaresinol) |
| Coptis chinensis Franch. | Root | 325.4 | 281.8 | 1.48 | 1.38 | Phenolic acids (ferulic acid, chlorogenic acid), phenolic alkaloids (magnoflorine) |
| Paeonia lactiflora Pall. | Root, seed | 407.3 | 150.9 | 1.14 | 0.70 | Flavonols (astragalin), tannins (gallotannin), stilbenes (resveratrol) |
| Paeonia suffruticosa Andr. | Root-bark | 876.1 | 423.2 | 2.90 | 1.25 | Phenolic acids (gallic acid), hydrolyzable tannins (gallotannin), other phenolics (paeonol) |
| Pulsatilla chinensis (Bge.) Regel | Root | 319.3 | 200.2 | 1.77 | 0.48 | Lignans ((+)-pinoresinol) |
| Rosaceae | ||||||
| Agrimonia pilosa Ledeb. | Aerial parts | 698.6 | 937.4 | 3.66 | 4.22 | m-benzo-triphenols (agrimol A, B), flavonoids (luteolin, apigenin, quercetin, rutin), phenolic acids (gallic acid), tannins, coumarins |
| Crataegus pinnatifida Bge. | Fruit | 884.3 | 823.5 | 4.28 | 1.01 | Flavonoids (epicatechin, quercetin, vitexin, quercitrin), phenolic acids (chlorogenic acid) |
| Rosa chinensis Jacq. | Flower | 6206.9 | 4607.9 | 18.75 | 16.82 | Flavonoids (quercetin, catechin, anthocyanins), phenolic acids (gallic acid), tannins |
| Rubus chingii Hu. | Fruit | 946.1 | 817.0 | 4.54 | 4.02 | Phenolic acids (gallic acid), tannins (ellagic acid) |
| Sanguisorba officinalis L. | Root | 4446.6 | 3526.6 | 15.87 | 14.38 | Phenolic acids (gallic acid), hydrolyzable and condensed tannins, flavanols (flavan-3-ols: (+)-catechin, gallocatechin) |
| Rubiaceae | ||||||
| Gardenia jasminoides Ellis | Fruit | 132.6 | 163.7 | 1.00 | 1.08 | Phenolic acids (chlorogenic acid), flavones (gardenins) |
| Hedyotis diffusa Willd. | Whole plant | 113.8 | 160.4 | 0.90 | 0.64 | Phenolic acids (p-coumaric acid), flavonols (kaempferol glycosides) |
| Rubia cordifolia L. | Root | 623.2 | 331.8 | 2.11 | 2.76 | Anthraquinones (purpurin, alizarin, munjistin, and their glycosides) |
| Uncaria macrophylla Wall. | Branch with hooks | 500.6 | 435.5 | 2.39 | 2.02 | Flavonoids (hyperin, catechins), tannins |
| Rutaceae | ||||||
| Citrus aurantium L. | Immature fruit | 703.6 | 701.0 | 5.07 | 4.41 | Flavanones (hesperidin, naringenin, naringin) |
| Phellodendron amurense Rupr. | Bark | 334.2 | 303.3 | 1.68 | 1.74 | Phenolic alkaloids (phellodendrine, magnoflorine) |
| Zanthoxylum nitidum (Roxb.) DC. | Root, stem | 191.2 | 158.6 | 1.27 | 0.70 | Phenolic alkaloids (nitidine, dihydronitidine), lignans |
| Saururaceae | ||||||
| Houttuynia cordata Thunb. | Aerial parts | 359.3 | 215.3 | 2.19 | 0.52 | Flavonols (quercetin, quercitrin, rutin), phenolic acids (chlorogenic acid) |
| Scrophulariaceae | ||||||
| Picrorhiza scrophulariiflora Pennell | Rhizome | 518.0 | 483.6 | 3.92 | 3.65 | Phenolic acids (ferulic acid, vanillic acid, coumaric acid) |
| Rehmannia glutinosa Libosch. | Root | 74.5 | 97.2 | 0.53 | 0.62 | Phenolic acids (ferulic acid, caffeic acid) |
| Simarubaceae | ||||||
| Brucea javanica (L.) Merr. | Fruit | 93.6 | 144.7 | 0.57 | 0.87 | Flavonoids (quercetin and luteolin glycosides), phenolic acids (vanillic acid) |
| Solanaceae | ||||||
| Hyoscyamus niger L. | Seed | 153.3 | 139.5 | 0.87 | 0.58 | Not identified |
| Lycium barbarum L. | Root bark | 188.8 | 157.3 | 0.93 | 0.76 | Phenolic acids (cinnamic acid) |
| Lycium barbarum L. | Fruit | 490.8 | 389.4 | 2.58 | 0.70 | Coumarins (scopoletin) |
| Solanum nigrum L. | Aerial parts | 46.7 | 103.5 | 0.22 | 0.90 | Phenolic acids (chlorogenic acid) |
| Stemonaceae | ||||||
| Stemona sessilifolia (Miq.) Miq. | Root | 139.4 | 118.2 | 1.26 | 1.13 | Not identified |
| Theaceae | ||||||
| Camellia sinensis (L.) Kuntze | Young leaf or flush | 5268.6 | 3610.9 | 17.4 | 13.60 | Flavanols (catechin, epicatechin), flavonols (kaempferol and quercetin glycosides), condensed tannins |
| Umbelliferae | ||||||
| Angelica sinensis (Oliv.) Diels | Root | 101.7 | 104.7 | 0.58 | 0.48 | Simple phenols (vanillin, p-cresol), phenolic acids (ferulic acid) |
| Bupleurum scorzonerifolium Willd. | Root | 75.3 | 79.3 | 0.47 | 0.43 | Flavonols (quercetin, rutin, isoquercetin) |
| Foeniculum vulgare Mill. | Fruit | 105.9 | 150.6 | 0.60 | 0.92 | Phenolic acids (cinnamic acid, vanillic acid), coumarins (6,7-dihydroxycoumarin) |
| Valerianaceae | ||||||
| Patrinia scabiosaefolia Fisch. ex Link. | Aerial parts | 90.7 | 94.7 | 0.89 | 0.59 | Tannins |
| Verbenaceae | ||||||
| Vitex trifolia L. var. simplicifolia Cham. | Fruit | 75.6 | 149.1 | 0.41 | 1.03 | Not identified |
| Violaceae | ||||||
| Viola yedoensis Makino | Whole plant | 643.2 | 537.5 | 2.40 | 0.96 | Phenolic acids (p-hydroxybenzoic acid, p-coumaric acid), flavonols (kaempferol glycosides) |
| Zingiberaceae | ||||||
| Curcuma longa L. | Rhizome | 132.1 | 54.5 | 0.80 | 0.27 | Curcuminoids (curcumin, bisdemethoxycurcumin, demethoxycurcumin), phenolic volatile oils |
| Zingiber officinale Rosc. | Rhizome | 254.0 | 82.4 | 0.74 | 0.38 | Phenolic volatile oils (gingerol analogues: gingerols, shogaol) |
| Alpinia officinarum Hance | Rhizome | 932.8 | 786.8 | 4.76 | 3.71 | Flavonoids (galangin, kaempferol, kaempferide), tannins, phenolic volatile oils |
| Mean | 941.1 | 787.9 | 3.89 | 3.01 | ||
All values were mean of triplicates.
TEAC, Trolox equivalent antioxidant capacity. Data expressed as micromoles of Trolox equivalents per 100 g dry weight (DW).
Data expressed as grams of gallic acid (GAE) equivalents per 100 g dry weight (DW).
Table 2.
Antioxidant activity and total phenolic content of methanolic extracts from common vegetables and fruits (dietary plants)a
| Scientific and common name | TEAC (μmol trolox/100 g DW)b | Total phenolic content (g GAE/100g DW)c |
|---|---|---|
| Actinida chinensis (kiwifruit) | 48.9 | 0.22 |
| Allium fistulosum (spring onion) | 43.4 | 0.42 |
| Allium sativum (garlic) | 55.0 | 0.20 |
| Brassica oleracea (broccoli) | 101.4 | 0.63 |
| Citrus sinensis (orange) | 67.2 | 0.51 |
| Curcumis sativus (cucumber) | 28.7 | 0.16 |
| Daucus carota (carrot) | 38.7 | 0.22 |
| Lactuca sativa (Chinese lettuce) | 128.4 | 0.78 |
| Lycopersicon esculentum (tomato) | 149.4 | 0.42 |
| Malus pumila (Fuji apple) | 92.7 | 0.48 |
| Malus pumila (Washington red apple) | 133.2 | 0.75 |
| Pyrus communis (pear) | 21.5 | 0.12 |
| Solanum melongena (eggplant) | 166.9 | 1.08 |
| Spinacia oleracea (spinach) | 167.1 | 0.90 |
| Mean | 88.9 | 0.49 |
All values were mean of triplicates.
TEAC, Trolox equivalent antioxidant capacity. Data expressed as micromoles of Trolox equivalents per 100 g dry weight (DW).
Data expressed as grams of gallic acid (GAE) equivalents per 100 g dry weight (DW).
Chemicals and reagents
2,2-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), potassium persulfate, sodium carbonate, ferric chloride, tert-butylhydroquinone, and dimethyl sulfoxide (DMSO) were purchased from Sigma/Aldrich (St. Louis, MO), Trolox (6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid) and bovine serum albumin (BSA) from Fluka Chemie AG (Buchs, Switzerland), and Folin-Ciocalteu reagent, HPLC grade organic reagents and HCl from BDH (Dorset, England). All authentic standards of various phenolic compounds (hydroxybenzoic acids, hydroxycinnamic acids, flavones, flavonols, flavanones, flavanols, isoflavones, coumarins, lignans, quinones, stilbenes, etc.) were obtained from Sigma/Aldrich and Fluka.
Extract preparation
The collected samples were ground to fine powder (710 μm) by a Kenwood Multi-Mill (Kenwood Ltd., UK) and passed through a sieve (24 mesh). The ground samples were dried to constant weight in desiccant at room temperature (∼23°C). (1) Water extraction: 5 g of the powdered sample was extracted with 100 mL ultra-filtered water at 80°C for 20 min in a water bath shaker (Shaking Bath 5B-16) (Techne, Ltd., UK). After cooling the extract was centrifuged at 5,000 rpm for 10 min and filtered by a Millipore filter with a 0.45-μM nylon membrane under vacuum at 23°C. The filtrate was stored at 4°C until use within 24 hr. (2) Methanol extraction: 5 g sample was extracted with 100 mL 80% methanol at 35°C for 24 hr in a Shaking Bath 5B-16. Other procedures were the same as in the water extraction method.
Total antioxidant capacity assay
Antioxidant capacity assay was carried out using a Spectronic Genesys 5 spectrophotometer by the improved ABTS•+ method as described by Re et al. (1999). ABTS•+ radical cation was generated by reacting 7 mM ABTS and 2.45 mM potassium persulfate after incubation at room temperature (23°C) in dark for 16 hr. The ABTS•+ solution was diluted with 80% ethanol to an absorbance of 0.700 ± 0.050 at 734 nm. The filtered sample was diluted with ultrafiltered water or 80% ethanol so as to give 20–80% inhibition of the blank absorbance with 0.1 mL of sample. ABTS•+ solution (3.9 mL; absorbance of 0.700 ± 0.050) was added to 0.1 mL of the tested samples and mixed thoroughly. The reactive mixture was allowed to stand at room temperature for 6 min and the absorbance was immediately recorded at 734 nm. Trolox standard solution (final concentration 0–15 μM) in 80% ethanol was prepared and assayed at the same conditions. The absorbance of the resulting oxidized solution was compared to that of the calibrated Trolox standard. Results were expressed in terms of Trolox equivalent antioxidant capacity (TEAC, μmol Trolox equivalents per 100 g dry weight of plant).
Total phenolics
Total phenolic content was estimated using the Folin-Ciocalteu colorimetric method described previously Zheng and Wang, 2001, Liu et al., 2002 with a little modification. Briefly, the appropriate dilutions of the filtered extracts were oxidized with 0.2 N Folin-Ciocalteu reagent and then the reaction was neutralized with saturated sodium carbonate (75 g/L). The absorbance of the resulting blue color was measured at 760 nm with a Spectronic Genesys 5 spectrophotometer (Milton Roy, NY) after incubation for 2 hr at 23°C. Quantification was done on the basis of the standard curve of gallic acid. Results were expressed as gram of gallic acid equivalent (GAE) per 100 g of dry weight (DW).
Estimation of tannins
Tannins are natural polyphenols with molecular weight ranging from 500 to 3000–4000, usually classified into condensed tannins (proanthocyanidins) and hydrolyzable tannins (gallo- and ellagitannins). Although several methods of determining condensed and hydrolyzable tannins (e.g. vanillin assay and sodium nitrite assay) have been developed, their repeatability and sensitivity are sometimes problematic Shahidi and Naczk, 1995, Mueller-Harvey, 2001, Schofield et al., 2001. There are no very good methods to estimate total tannin content. Protein (BSA) precipitation assay, a traditional method, was applied in this study to estimate total tannin content. The medicinal extracts were precipitated with BSA and, after centrifugation, the precipitates (tannins) reacted with FeCl3 becoming blue/green/violet coloured complexes which could be measured at 510 nm by spectrophotometer. The detailed procedures followed the methods described in Hagerman and Butler (1978) and Shahidi and Naczk (1995). Results were expressed as gram of tannic acid equivalents per 100 g of dry weight or as percentage (W/W). Measurement of total tannin content was conducted only for the medicinal herbs with higher total phenolic content (over 5.0 g GAE/100 g DW) in this study. Hydrolyzable and condensed tannins were discriminated using acidic treatment (0.5 N HCl) at 100°C. The former could be hydrolyzed without precipitates and the latter was precipitated without hydrolysis.
HPLC analysis
HPLC analysis was performed using a Hewlett-Packard HPLC System (HP 1100 series, Waldbronn, Germany), consisting of a binary pump, and a diode-array detector (DAD). Conditions for analytical HPLC were: Nucleosil 100-C18 column (5 μm, 250 × 4 mm) with Nucleosil 5 C18 guard column (5 μm, 4 × 4 mm) (Agilent Technologies, USA). Phenolic compounds in the medicinal herb extracts were analyzed using the reference HPLC method (Sakakibara et al., 2003) with gradient elution (solution A, 50 mM sodium phosphate in 10% methanol, pH 3.3, and solution B, 70% methanol): 0 min, 100% A; 15 min, 70% A; 45 min, 65%; 65 min, 60% A; 70 min, 50% A; and 95 min, 0% A. In order to improve separation of the phenolic compounds with weaker polarity (much later retention time), the improved HPLC method was with the following gradient elution program (solution A, formic acid or acetic acid in H2O, pH 2.5–3.0, and solution B 100% methanol): 0 min, 5% B; 15 min, 30% B; 40 min, 40% B; 60 min, 50% B; 65 min, 55% B; and 90 min, 100% B. Flow rate was 1.0 mL/min and injection volume was 20-μL. Detection was at 250 nm for hydroxybenzoic acids, isoflavones, and quinones; 280 nm for flavanones, flavanols, lignans, and tannins (proanthocyanidins or leucoanthocyanidins, gallotannins or ellagitannins); 320 nm for hydroxycinnamic acids, flavones, coumarins, and stilbenes; 370 nm for flavonols and chalcones; 510 nm for anthocyanins; and 420 nm for curcuminoids.
Hydrolysis
Acidic or alkaline hydrolysis is an ordinary chemical treatment for the simplified identification of phenolic compounds. Many phenolic compounds, such as flavonoids, are normally present as glycosides and sometimes as aglycones in plants. The methanolic extracts of some medicinal herbs were subjected to acidic hydrolysis (2 N HCl) to break the glycoside linkages, according to the methods reported by Xiao et al. (2000) and Sakakibara et al. (2003). The hydrolysates were analyzed by HPLC to identify the relevant aglycones so as to ascertain their phenolic types.
Data analysis
All determinations were conducted in triplicate and all results were calculated as mean ± standard deviation (SD) in this study. Statistical analysis was performed using Statistica for Windows (version 6.0) (Statsoft Inc., Tulsa, OK).
Results
Antioxidant activity and total phenolic content
The antioxidant activity assay was carried out to assess the ability of the medicinal extracts to scavenge free radicals in vitro (expressed as TEAC value) by the improved ABTS•+ method (Re et al., 1999) in this study. This method was not only a rapid and reliable test of total antioxidant capacity but also an advantageous assay applicable to both hydrophilic and lipophilic antioxidants/systems. The roots/rhizomes, stems or barks, leaves, flowers/inflorescences, fruits/seeds and even whole plants of TCM are commonly prepared and consumed as a boiled medicinal tea/soup or medicinal bolus to treat/prevent various cancers. Both aqueous and methanolic extracts of all tested Chinese medicinal herbs were used for comparison.
A total of 112 medicinal plant species from 50 families were collected, extracted, and tested. Their TEAC values (Table 1) indicated extremely large variation in antioxidant activity. Total antioxidant capacity of the aqueous extracts ranged from 43.9 to 17,674 μmol Trolox equivalents per 100 g dry weight (μmol TEAC/100 g DW), and the averaged TEAC value was 787.9 μmol/100 g DW. The TEAC values for the methanolic extracts ranged from 46.7 to 17,323 μmol/100 g DW, and the mean value was 941.1 μmol/100 g DW. Chinese Galls from Rhus chinensis/R. potaninii contained the highest antioxidant concentration (aqueous/methanolic: 17,674/17,323 μmol TEAC/100 g), followed by dried extracts (Catechu) from branches and stems of Acacia catechu (aqueous/methanolic: 10,049/11,197 μmol TEAC/100 g). Of the 112 medicinal herb species, it was found that 18 species (16.1%) contained more than 1,000.1 μmol TEAC/100 g, and 21 species (18.7%) were below 100 μmol TEAC/100 g, while the majority (65.2%) had medium amounts of antioxidants, i.e., 49 species (43.8%) between 100.1 and 500 μmol TEAC/100 g and 24 species (21.4%) between 500.1 and 1,000.0 μmol TEAC/100 g (Fig. 1) .
Fig. 1.
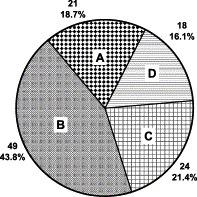
Distribution (percentage) of 112 medicinal herbs associated with anticancer among different ranges of antioxidant activity (TEAC value). A: <100.0 μmol TEAC/100 g DW; B: 100.1−500.0 μmol/100 g; C: 500.1−1,000.0 μmol/100 g; and D: >1,000.1 μmol/100 g (TEAC value of methanol extracts).
It was also found that the total phenolic content of 112 medicinal herbs had significant variation, ranging from 0.19 to 50.20 g of gallic acid equivalents (GAE)/100 g dry weight (DW) with average value of 2.93 g/100 g DW for aqueous extracts, and from 0.22 to 50.30 g/100 g DW with average value of 3.87 g/100 g DW for methanolic extracts (Table 1). Chinese Galls from Rhus chinensis/R. potaninii contained the most phenolic compounds (aqueous/methanolic: 50.20/50.30 g/100 g), followed by dried extracts (Catechu) from branches and stems of Acacia catechu (aqueous/methanolic: 41.94/45.70 g/100 g). Of the 112 medicinal herb species, most (79 species, 70.5%) contained total phenolic content between 0.51 and 5.00 g/100 g, whereas only 14 species (12.5%) had below 0.50 g/100 g of phenolics, and 19 species (17.0%) had over 5.01 g/100 g of phenolics.
In order to provide a point of reference, we measured the antioxidant activity and total phenolic content of 14 common vegetables and fruits (dietary plants). Their total antioxidant capacity ranged from 21.5 to 168.9 μmol TEAC/100 g DW with average value of 88.9 μmol TEAC/100 g DW for methanolic extracts, while their total phenolic content varied from 0.12 to 1.08 g GAE/100 g DW with average value of 0.49 g GAE/100 g DW for methanolic extracts (Table 2). On average, there was about 10-fold difference of total antioxidant capacity, and about 8-fold difference of total phenolic content between the tested medicinal herbs and dietary plants. If not including two extreme cases (Chinese Galls and Catechu), the mean TEAC value and the average total phenolic content value of other 110 medicinal herbs were also very high (698.8 μmol TEAC/100 g and 3.08 g GAE/100 g). The results revealed that the tested medicinal herbs demonstrated remarkably higher antioxidant activity and contained significantly more phenolics than common vegetables and fruits (dietary plants).
Since most Chinese medicinal herbs are prepared for consumption of herbal tea or soup, the aqueous extraction applied in this study was necessary. With comparison of antioxidant activity and total phenolic content of the medicinal herbs, although the mean values of antioxidant activity and total phenolic content of aqueous extracts were lower than those of methanolic extracts, the TEAC values of aqueous extracts of 41 herbal species were higher than those of methanolic extracts, and total phenolic contents of aqueous extracts of 29 herbal species were higher than those of methanolic extracts (Table 1). Hot water (80 °C) extraction was a useful method with extracting efficiency of 83.7% for antioxidant activity and of 77.4% for total phenolic content, as compared with 80% methanolic extraction. The correlation coefficients (R 2) of antioxidant activity and total phenolic content between aqueous and methanolic extracts were 0.967 and 0.961 for all tested medicinal herbs, and 0.891 and 0.878 for 110 medicinal herbs (not including two extreme cases), respectively, indicating that these two extraction methods were basically consistent.
Relationship between antioxidant activity and total phenolic content
The correlation coefficients between antioxidant activity and total phenolic content of the medicinal plants were calculated (Fig. 2) . The number of tested samples and the range of parameter values normally influence the calculation of correlation coefficients (R 2). More samples and suitable range of parameter values can provide reasonable R 2 values and representative correlation. The overall relationship between antioxidant activity and total phenolic content for all tested medicinal herbs (112 species) was a positive and highly significant linear correlation (methanolic/aqueous: R 2 = 0.964/0.953). The higher total phenolic content of the medicinal herbs resulted in higher total antioxidant capacity. If not including two extreme cases (Chinese Galls and Catechu), antioxidant activity was also highly significantly correlated to total phenolic content (methanolic/aqueous: R 2 = 0.958/0.950). The results suggested that the phenolic compounds contributed significantly to the antioxidant capacity of the medicinal herbs.
Fig. 2.
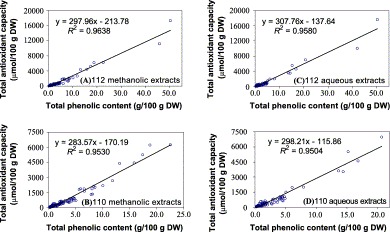
Relationship between total antioxidant capacity (μmol TEAC/100 g DW) and total phenolic content (g gallic acid equivalents/100 g DW) of methanolic (A, B) and aqueous (C, D) extracts from 112/110 medicinal herbs associated with anticancer.
Preliminary identification and analysis of phenolic compounds
Chemically, phenolic compounds (phenolics) can be defined as substances possessing an aromatic ring bearing one or more hydroxy substituents, including their functional derivatives Shahidi and Naczk, 1995, Strack, 1997. Because of the diversity and complexity of the natural mixtures of phenolic compounds in hundreds of herb extracts, it is rather difficult to characterize every compound and elucidate its structure, but it is not difficult to identify major groups and important types of phenolic compounds. Many medicinal herbs had been studied and to some extent their phenolic chemistry was known (Xiao et al., 2000). HPLC analysis methods for most kinds of phenolic compounds in plants have been developed Krizsán et al., 1996, Andrade et al., 1998, Bouchet et al., 2000, Sakakibara et al., 2003. Especially, a library of the analytical characteristics of 100 phenolic standards established by Sakakibara et al. (2003) could provide important reference data (Retention times, UV and visible λ max, spectra shapes).
In this study, major types and their representative constituents of phenolic compounds in the tested medicinal herbs were identified and ascertained by cochromatography with 65 standard samples and by comparison with literature data Ji et al., 1999, Xiao et al., 2000, Sakakibara et al., 2003, and with the aid of certain classical chemical methods (e.g. hydrolysis). Because of the large number of samples, this investigation was just preliminary identification of phenolic compounds to find out major types of phenolic compounds and their representative constituents and to ascertain their distribution. Major types and representative components of phenolics identified from most medicinal herbs are summarized in Table 1, mainly including phenolic acids, flavonoids, tannins, coumarins, lignans, quinones, stilbenes, curcuminoids, etc. The retention times (Rt) of various phenolic types identified in this study were approximately in the following ranges: 6.8–30.2 min for phenolic acids; 9.0–28.4 min for flavan-3-ols (catechins); 22.0–40.4 min for anthocyanins; 19.5–45.3 min for coumarins and stilbenes; 19.0–48.1 min for glycoside forms of flavones, isoflavones, flavonols, and flavanones, and 23.5–73.9 min for their aglycone forms; 42.7–73.9 min for chalcones; 51.5–87.1 min for quinones; and 36.2–88.7 min for lignans (71.8–88.7 min for lignans of Schisandra chinensis). Fig. 3 displays typical HPLC chromatogram of parts of phenolic standards. Several selected HPLC profiles of the medicinal herbs are shown in Fig. 4 . However, main phenolic compounds in 9 medicinal herbs could not yet be identified in this study. More phenolic constituents will be identified in future work with the aid of additional techniques.
Fig. 3.
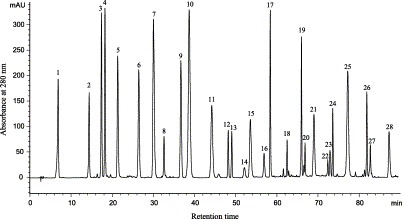
Typical HPLC chromatogram of parts of phenolic standards at 280 nm analyzed with the improved method. Peaks: 1, gallic acid; 2, catechin; 3, vanillic acid; 4, caffeic acid; 5, cyanidin-3-O-glucoside; 6, ferulic acid; 7, coumarin; 8, resveratrol; 9, myricitrin; 10, rutin; 11, quercitrin; 12, diosmin; 13, daidzein; 14, glycitein; 15, quercetin; 16, emodin-8-O-glucoside; 17, luteolin; 18, hesperetin; 19, kaempferol; 20, baicalein; 21, alizarin; 22, galangin; 23, flavonol; 24, chalcone; 25, rhein; 26, emodin; 27, quininzarin; 28, physcion.
Fig. 4.
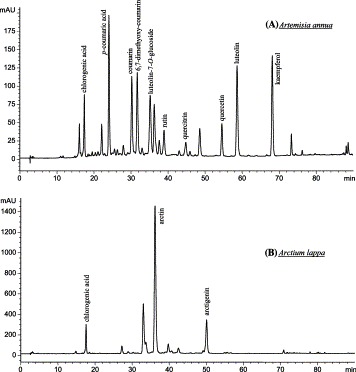
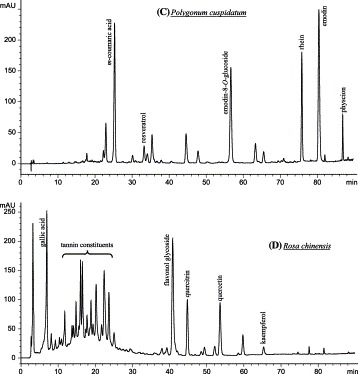
HPLC profiles of methanolic extracts from four selected medicinal herbs: (A) phenolic acids, coumarins and flavonoids from aerial parts of Artemisia annua; (B) lignans and phenolic acid from fruits of Arctium lappa; (C) anthraquinones, stilbene and phenolic acid from rhizomes of Polygonum cuspidatum (tannins removed); and (D) flavonoids, phenolic acid and tannins from flowers of Rosa chinensis. Elution monitored at 250 nm (C), 280 nm (B and D), and 370 nm (A).
Phenolic acids
Phenolic acids are a major class of phenolic compounds, widely occurring in plant kingdom. Predominant phenolic acids included hydroxybenzoic acids (e.g. gallic acid, p-hydroxybenzoic acid, protocatechuic acid, vanillic acid), and hydroxycinnamic acids (e.g. ferulic acid, caffeic acid, coumaric acid, chlorogenic acid, cinnamic acid). They were readily identified using HPLC-DAD by comparison with authentic standards. It was found that more than 70 species of the tested medicinal herbs contained phenolic acids. Table 1 only lists over 30 species with higher concentration of phenolic acids (Table 1). The HPLC profiles in Fig. 4A-D show that four selected medicinal herbs contained phenolic acids.
Flavonoids
It was found that flavonoids were the largest class of phenolics in the tested medicinal herbs in this study. Flavonoids are widely distributed in the plant kingdom, and have the basic skeleton of diphenylpropanes (C6 + C3 + C6) with different oxidation of the central pyran ring. They are subclassified as flavones (basic structure, B ring binds to the 2-position), flavonols (having a hydroxyl group at the 3-position), flavanones (dihydroflavones) and flavanonols (dihydroflavonols) (2–3 bond is saturated), and flavanols (flavan-3-ols and flavan-3,4-diols) (C-ring is 1-pyran), chalcones (C-ring is opened), isoflavonoids (mainly isoflavones) (B ring binds to the 3-position), anthocyanins (anthocyanidins) (C-ring is 1-pyran, and 1–2 and 3-4 bonds are unsaturated), and biflavonoids (dimer of flavones, flavonols, and flavanones) Rice-Evans et al., 1996, Iwashina, 2000, Xiao et al., 2000. Flavonoids in plants are mainly present as glycosides with a sugar moiety/more sugar moieties linked through an OH group (O-glycosides) or through carbon-carbon bonds (C-glycosides), but some flavonoids are present as aglycones.
Various kinds of flavonoids identified in the tested medicinal herbs were listed in Table 1, as possible as according to the above classification. The preliminarily identified results indicated that the most common flavones were luteolin, apigenin, baicalein and their glycosides, mainly distributing in the Labiatae, Compositae, etc, such as roots of Scutellaria baicalensis, inflorescences of Chrysanthemum morifolium, aerial parts of Artemisia annua (Fig. 4A). Quercetin, kaempferol, myricetin, and their glycosides were the predominant flavonols, occurring in many medicinal herbs associated with anticancer, e.g. flowers of Sphora japonica and Rosa chinensis (Fig. 4D), aerial parts of Artemisia annua (Fig. 4A), rhizomes of Alpinia officinarum, fruits (hawthorn) of Crataegus pinnatifida. Flavanols, such as catechins (flavan-3-ols), were also widespread in the tested medicinal herbs, e.g. Camellia sinensis (tea) and Acacia catechu (catechu) that contained large amounts of catechins. Flavanones and flavanonols, such as naringenin, hesperetin, liquiritin, and their glycosides, were mainly found in the medicinal herbs of Rutaceae (e.g. citrus fruits), Rosaceae, Leguminosae, etc. Isoflavones were mostly detected in the medicinal herbs of Leguminosae, such as roots of Astragalus mongholicus. Anthocyanins, including anthocyanidins (e.g. cyanidin, delphinidin, malvidin, peonidin, etc.) and their glycosides, were mainly found in flowers of the medicinal herbs, such as inflorescences of Prunella vulgaris.
Tannins
Tannins were identified as another large class of phenolics in the tested medicinal herbs (Table 1). More than 32 of the species contained tannin constituents preliminarily examined using protein precipitation method. Of these, 19 species contained considerable higher proportions of tannins, such as Chinese Galls (47 %), Acacia catechu (catechu) (41.2%), Punica granatum (19.3%), and Sanguisorba officinalis (13.5%). Tannins are generally subdivided into hydrolyzable and condensed tannins. Hydrolyzable tannins contain a central core of polyhydric alcohol such as glucose, and hydroxyl groups, which are esterified either partially or wholly by gallic acid (gallotannins) or hexahydroxy-diphenic acid (ellagitannins) Chung et al., 1998, Mueller-Harvey, 2001. Famous examples of gallotannins were Chinese tannins (50–70% of dry weight) from Chinese Galls (Xiao et al., 2000). In this study, gallotannins from sample of Chinese Galls were determined below 50% of dry weight, likely due to samples collected from different sites and using different measurement methods. Several ellagitannins were isolated from fruits of Terminalia chebula and peels of Punica granatum. Condensed tannins are structurally more complex, and more widely spread among the plants than hydrolyzable tannins. They are mainly the oligomers and polymers of flavan-3-ols (catechins), also known as proanthocyanidins Xiao et al., 2000, Schofield et al., 2001. Some authors (Chung et al., 1998) considered that the polymerized products of flavan-3,4-diols also belong to the category of condensed tannins, also called leucoanthocyanidins. For instance, it was observed that Camellia sinensis and Areca catechu contained proanthocyanidins and leucoanthocyanidins, respectively. Some plant species may produce complex mixtures containing both hydrolyzable and condensed tannins. It was found that the tannins identified in Acacia catechu, Sanguisorba officinalis, Rosa chinensis, and Punica granatum in this study belonged to the complex mixtures (Table 1). The HPLC profile (Fig. 4D) of methanolic extracts from Rosa chinensis exhibits typically separated peaks (R t 8–25 min) of tannin constituents. However, detailed tannin constituents will require further identification by MS and NMR.
Coumarins and lignans
Coumarins and lignans are lactones of cis-o-hydroxycinnamic acid derivatives, belonging to the phenolics with the basic skeleton of C6 + C3 (Xiao et al., 2000). Coumarins are present in plants in the free form and as glycosides. Lignans are mainly present in plants in the free form and as glycosides in the minority. In this study, it was detected that coumarins and lignans occurred in the medicinal herbs of Compositae, Leguminosae, Magnoliaceae, Rutaceae, Oleaceae, Convolvulaceae, and Ranunculaceae families (Table 1, Fig. 4A and B). Major coumarin constituents included simple coumarins (Artemisia annua), furanocoumarins (Psoralea corylifolia), pyranocoumarins (Citrus aurantium), isocoumarins (Agrimonia pilosa), and others (wedelolactone from Eclipta prostrata). Main lignan constituents were lignanolides (Arctium lappa), cyclolignanolides (Polygala tenuifolia), bisepoxylignans (Forsythia suspensa), neolignans (Magnolia officinalis), and others (Schisandra chinensis).
Quinones
Quinones are also an important class of phenolics, occurring only in the medicinal herbs, but not in common vegetables and fruits tested in this study. Natural quinones mainly have four classes, i.e. anthraquinones, phenanthraquinones, naphthoquinones, and benzoquinones. Of these, anthraquinones are the most common in the medicinal herbs Strack, 1997, Xiao et al., 2000. This investigation found that quinones were distributed in eleven species of medicinal herbs from eight families, such as Polygalaceae, Rubiaceae, Boraginaceae, Latiatae, Leguminosae, Rubiaceae, etc (Table 1). For example, several anthraquinones and their glycosides were identified in the rhizomes and roots from Polygonum cuspidatum (Fig. 4C), Polygonum multiflorum, and Rheum officinale in the Polygalaceae, including rehein, emodin, chrysophanol, physcion, anthraglycosides, etc.
Stilbenes
Stilbenes are 1,2-diarylethenes and are synthesized from cinnamic acid derivatives, also belonging to phenolics. Stilbenes along with isoflavones and lignans are phytoestrogens which protect against hormone-dependent tumors. They are distributed in higher plants, in monomeric form (e.g. resveratrol, oxyresveratrol) and as dimeric, trimeric and polymeric stilbenes, or as glycosides (Cassidy et al., 2000). We identified the monomeric stilbenes using HPLC by authentic standards in four species of medicinal herbs, i.e. trans-resveratrol in Polygonum cuspidatum and Polygonum multiflorum (Table 1). It was reported that dimeric stilbenes and stilbene glycosides had been identified from these three species Qiu et al., 1996, Xiao et al., 2002.
Curcuminoids
Curcuminoids are ferulic acid derivatives, such as curcumin, which contain two ferulic acid molecules linked by a methylene, with a β–diketone structure in a highly conjugated system. Curcuminoids and ginerol analogues are natural phenolic compounds from plants of the family Zingiberaceae. Curcuminoids-containing Curcuma longa and ginerol analogues-containing Zingiber officinale are not only used as Chinese traditional medicines but also as ordinary spices or natural color agents. These phenolic compounds have been studied well Cooper et al., 1994, Kikuzaki et al., 1994. With comparison of the reference data, three curcuminoids and major ginerol analogues in rhizomes of Curcuma longa and Zingiber officinale were detected by HPLC (Table 1).
Others
Because phenolic alkaloids, phenolic terpenoids (including aromatic volatile oils), special phenolic glycosides, and m-benzo-triphenol derivatives contain one or more aromatic rings bearing one or more hydroxy groups, they also belong to phenolic compounds structurally. In this study, these mixed classes of phenolics known in the medicinal herbs were also summarized in Table 1, according to The Chemistry of Traditional Chinese Medicines (Xiao et al., 2000), although we did not analyze and ascertain them. Additionally, simple phenols (only C6 skeleton) were identified in the aromatic volatiles of some medicinal herbs, such as vanillin and p-cresol from Angelica sinensis.
Discussion
Many assay methods for antioxidant activity in vitro and in vivo have been developed, but only a few rapid and reliable methods applicable to antioxidant activity assay for a huge number of plant extract samples exist Miller et al., 1993, Brand-Williams et al., 1995, Aruoma and Cuppett, 1994. Total antioxidant capacity assay, such as the ABTS•+ and DPPH• methods, is most common for antioxidant activity for large-scale examination. The improved ABTS•+ method established by Re et al. (1999) was successfully used in this study to systematically assess the total antioxidant capacity of the medicinal herb extracts on a large scale, being simple, fast, reliable, inexpensive, and also very adaptable to both hydrophilic and lipophilic antioxidants/systems. This effective and efficient method can be used for systematic screening of medicinal herbs and dietary plants for their relative antioxidant content.
Previous studies found that there was a direct relationship between antioxidant activity and total phenolic content in selected herbs and vegetables and fruits. Phenolic compounds had a major contribution to antioxidant activity Velioglu et al., 1998, Zheng and Wang, 2001, Sun et al., 2002. However, their results were based on the determination and statistical analysis of a small number of samples. This study systematically investigated 112 medicinal herbs. Statistical results of this larger number of samples revealed that antioxidant activity of the tested medicinal herbs was significantly correlated to total phenolic content (all R 2 values ≥0.950, Fig. 2), strongly confirming that phenolic compounds were the dominant antioxidant components in the traditional Chinese medicinal herbs associated with anticancer.
Antioxidant activity of phenolic compounds was correlated to their chemical structures. Structure-activity relationship of some phenolic compounds (e.g. flavonoids, phenolic acids, tannins) has been studied Rice-Evans et al., 1996, Lien et al., 1999, Son and Lewis, 2002. In general, free radical scavenging and antioxidant activity of phenolics (e.g. flavonoids, phenolic acids) mainly depends on the number and position of hydrogen-donating hydroxyl groups on the aromatic ring of the phenolic molecules, and is also affected by other factors, such as glycosylation of aglycones, other H-donating groups (-NH, -SH), etc. For example, flavonol aglycones such as quercetin, myricetin, and kaemperol, containing multiple hydroxyl groups, had higher antioxidant activity than their glycosides such as rutin, myricitrin, astragalin. The glycosylation of flavonoids reduced their activity. There were many reports on antioxidant components, generally focusing on flavonoids and phenolic acids Rice-Evans et al., 1996, Nakatani, 2000, Zheng and Wang, 2001, but relatively fewer on other phenolic antioxidant components. Through preliminary identification and analysis of phenolic compounds, we summarized that typical phenolic compounds in the tested medicinal herbs were of several major types, including phenolic acids, flavonoids, tannins, coumarins, lignans, quinones, stilbenes, and curcuminoids. Some representative constituents of major phenolic types were also identified in Table 1. However, it is believed that thousands of phenolic compounds occur in the medicinal herbs. For instance, more than 4,000 kinds of flavonoids and hundreds of coumarins and lignans have been reported as naturally occurring compounds Iwashina, 2000, Xiao et al., 2000. Therefore, further chemical identification of phenolic compounds in the tested medicinal herbs will be required to reveal the possible structure-activity relationship of various types of phenolics.
One of the main findings in this study was that the medicinal herbs demonstrated much stronger antioxidant activity and contained significantly more phenolics than common vegetables and fruits which are considered as good natural sources of dietary antioxidants. The traditional Chinese medicinal plants associated with anticancer might be a potential source of powerful natural antioxidants and beneficial chemopreventive agents. Chinese medicine emphasizes disease prevention and whole-body homeostasis instead of disease-oriented therapy. There is sometimes no strict borderline between medicinal treatment and dietary manipulation. Some medicinal herbs associated with anticancer, such as green tea (Camellia sinensis), fruits of Lycium barbarum, Juglans regia, and Crataegus pinnatifida, rhizomes of Zingiber officinale, and leaves and stems of Portulaca oleracea, are ordinarily used for pharmaceutical purposes and also as health foods and teas/soups in China. The beverages made from the tested medicinal herbs (e.g. Prunella vulgaris, Chrysanthemum indicum, Chrysanthemum morifolium, Lonicera japonica) are popular in China. The healthy alcoholic drinks are usually steeped with Chinese medicinal herbs, e.g. fruits of Lycium barbarum, roots of Panax ginseng, Angelica sinensis, and Glycyrrhiza uralensis. Another popular form of dietary therapy is daily consumption of Chinese soups prepared with plant materials including some tested medicinal herbs.
Many studies have revealed that intake of natural antioxidants is correlated with low incidence of cancer, heart disease, diabetes, and other diseases associated with ageing, but there are still controversial opinions Hertog et al., 1995, Mclarty, 1997, Albanes and Hartman, 1999. Phenolics in vegetables, fruits, spices, and medicinal herbs might prevent cancer through antioxidant action and/or the modulation of several protein functions. Phenolics may inhibit carcinogenesis by affecting the molecular events in the initiation, promotion, and progression stages (Yang et al., 2001). Phenolics demonstrated agonism and/or antagonism of carcinogenesis-related receptors such as arylhydrocarbon receptor, epidermal growth factor, and estrogen receptor β. They modulated the secretion of protein kinases in tumor cell proliferation, and induced the expression of anticarcinogenic enzymes or inhibited induction of cancer-promoting enzymes Ho et al., 1994, Wadsworth and Koop, 1999, Owen et al., 2000, Sakakibara et al., 2003. Some phenolics possessed potent antioxidant activity and also had anticancer/anticarcinogenic/antimutagenic activities, such as phenolic acids (e.g. chlorogenic acid, caffeic acid, ferulic acid), flavonoids (e.g. vitexin, quercetin, wogonin, genistein, catechins, isoflavones), quinones (e.g. emodin, rhein, aloe-emodin), coumarins (e.g. 7-hydroxy-coumarin), stilbenes (e.g. resveratrol), curcuminoids (e.g. curcumin), lignans, etc. Ho et al., 1994, Gao et al., 2000, Owen et al., 2000, Xiao et al., 2000, Yang et al., 2001, Tapiero et al., 2002. Moreover, some tannin molecules (e.g., tea polyphenols) have anticancer or anticarcinogenic or antimutagenic activity Chung et al., 1998, Kaur et al., 1998. Chung et al. (1998) pointed out in their review that the anticarcinogenic or antimutagenic potential of tannins might be related to their antioxidative properties. Therefore, phenolic antioxidants in the tested medicinal herbs and their antioxidative properties might play a role in preventing and treating cancer. However, some studies have not shown any relationship between consumption of natural dietary antioxidants and lower incidence of chronic disease such as cancer. Some evidence from in vitro studies and clinical studies is controversial. For example, observational epidemiological data for some sites are fairly consistent and supportive of a beneficial association between β-carotene and human cancer, but the preventive trial-based findings do not substantiate a beneficial effect of β-carotene supplementation on cancer incidence or mortality (Albanes and Hartman, 1999). The effects of an antioxidant may be tissue/site specific. The evidence for a protective effect for vitamin C appears to be strongest for cancers of the stomach, upper aerodigestive tract, and pancreas, but there is little evidence to support a protective relationship between either vitamin C and cancers of the colon, breast, or prostate (Mclarty, 1997). Although dietary flavonoids have been observed to inhibit cancer cell growth in vitro (Kuo, 1997), a seven-country study containing 16 cohorts showed that the average flavonoid intake did not seem to be a determinant of cancer mortality (Hertog et al., 1995).
The current controversies mainly concern individual dietary antioxidants such as vitamins (A, C, E), carotenoids (β-carotene), flavonoids, and tannins, as there are very few studies on complex phenolic antioxidant compounds in medicinal plants. Nobody appears to have reported the relationship between phenolic antioxidants and anticancer activity of the anticancer-related medicinal plants. From this study, it was found that the phenolic compounds in anticancer-associated traditional Chinese medicinal plants (TCMs) were significantly different in content and type from those (mainly flavonoids and phenolic acids) in dietary plants. The anticancer-associated TCMs not only contained much higher levels of phenolics than dietary plants, but also had many more kinds of phenolics, including tannins, coumarins, lignans, quinones, stilbenes, curcuminoids, and others (phenolic alkaloids, phenolic terpenoids, special phenolic glycosides), except flavonoids and phenolic acids. The protective effects of natural antioxidants may result from the action of complex/unknown/lesser-known antioxidants or from a concerted action of the cocktail of antioxidants present in dietary/medicinal plants. The association between the consumption of natural antioxidants and lowered cancer risk needs to be observed consistently in different studies. Therefore, it is required to further scientifically measure anticancer activity of the Chinese medicinal plants associated with anticancer. It is also necessary to make a thorough investigation of the relationship between anticancer and antioxidant activity of their phenolic compounds on a large scale to reveal whether their relationship is positive or negative. In addition to the analyzed phenolic compounds in this study, other types of anticancer constituents possibly occurring in the tested medicinal samples also warrant further analysis.
From a Chinese medicine viewpoint, tumor therapy is usually based on the theory of heat and toxin clearing. Nearly two fifths of the medicinal herbs tested in this study belong to the category of herbal medicines with heat and toxin clearing attributes. This category exhibits a wide range of biological effects in addition to antioxidant activity, such as antibacterial, antiviral, anti-inflammatory, and antineoplastic activities which may be related to some phenolic compounds (Shen and Li, 2002). It was reported that some medicinal herbs associated with anticancer, such as Viola yedoensis, Prunella vulgaris, and Paeonia suffruticosa, had anti-HIV activity (Collins et al., 1997). Notably, several common medicinal herbs with antiviral effect, such as Isatis indigotica, Forsythia suspense, Lonicera japonica, Artemisia capillaries, Prunella vulgaris, and Taraxacum mongolicum, form a Chinese Medicine prescription currently popular in China for the effective prevention and treatment of SARS (Severe Acute Respiratory Syndrome). Chinese Medicine (CM) treats diseases including tumors with formulae (conventionally 4–12 herbs) prescribed by CM practitioners, very seldom with a single medicinal herb. The therapeutic effects of the prescription are often interpreted in a magical or mystical way in traditional Chinese medicine. This is likely due to the synergistic effects caused by the combination of the medicinal plants from the prescription. Also, application of Chinese medicinal formulas may change some of the bioactive constituents from single medicinal plant extract. Therefore, elucidation of bioactive components and their biological functionality in the traditional Chinese medicinal herbs associated with anticancer and their medicinal formulas will contribute to a more rational scientific understanding.
In conclusion, the results of the present work showed that the improved ABTS•+ method was successfully applied to evaluate the total antioxidant capacity of medicinal herbs on a large scale. Large variation was found in total antioxidant capacity (∼370-fold) and total phenolic content (∼230-fold) among 112 tested medicinal herbs. The traditional Chinese medicinal plants associated with anticancer demonstrated significantly stronger antioxidant activity and contained much more phenolic compounds than common vegetables and fruits. A positive and significant correlation existed between antioxidant activity and total phenolic content, revealing that phenolic compounds were the dominant antioxidant components in the tested medicinal herbs. Phenolic acids, flavonoids, tannins, coumarins, lignans, quinones, stilbenes, and curcuminoids were identified as major types of phenolic compounds. The investigation of the inter-relationship between phenolic compounds and antioxidant/anticancer activity will be a promising field to understand and elucidate possible mechanisms for the functionality of traditional Chinese medicines for cancer prevention and treatment.
Acknowledgements
This research was supported by a University of Hong Kong Faculty of Science Research Collaboration Grant.
References
- Albanes D., Hartman T.J. Antioxidants and cancer: evidence from human observational studies and intervention trials. In: Papas A.M., editor. Antioxiant Status, Diet, Nutrition, and Health. CRC Press; Boca Raton, Florida: 1999. pp. 497–544. [Google Scholar]
- Andrade P.B., Seabra R.M., Valentão P., Areias F. Simultaneous determination of flavonoids, phenolic acids, and coumarins in seven medicinal species by HPLC/diode-array detector. Journal of Liquid Chromatography and Related Technologies. 1998;21(18):2813–2820. [Google Scholar]
- Aruoma O.I., Cuppett S.L. AOCS Press; Illinois: 1994. Antioxidant Methodology: In Vivo and In Vitro Concepts. [Google Scholar]
- Bo Q.M., Wu Z.Y., Shun Q.S., Bao X.S., Mao Z.S., Ha S.Q., Lu S.Y., Huang J.M. Shanghai Science and Technology Literature Press; Shanghai: 2002. A Selection of the Illustrated Chinese Anti-Cancer Herbal Medicines. [Google Scholar]
- Bouchet N., Lévesque J., Pousset J.L. HPLC isolation, identification and quantification of tannins from Guiera senegalensis. Phytochem Analysis. 2000;11(1):52–56. [Google Scholar]
- Brand-Williams W., Cuvelier M.E., Berset C. Use of a free radical method to evaluate antioxidant activity. Lebensmittel Wissenschaft and Technologie. 1995;28(1):25–30. [Google Scholar]
- Cai Y.Z., Sun M., Corke H. Antioxidant activity of betalains from plants of the Amaranthaceae. Journal of Agricultural and Food Chemistry. 2003;51(8):2288–2294. doi: 10.1021/jf030045u. [DOI] [PubMed] [Google Scholar]
- Cassidy A., Hanley B., Lamuela-Raventos R.M. Isoflavones, lignans and stilbenes—origins, metabolism and potential importance to human health. Journal of The Science of Food and Agriculture. 2000;80(7):1044–1062. [Google Scholar]
- Chung K.T., Wong T.Y., Huang Y.W., Lin Y. Tannins and human health: a review. Critical Reviews in Food Science and Nutrition. 1998;38(6):421–464. doi: 10.1080/10408699891274273. [DOI] [PubMed] [Google Scholar]
- Collins R.A., Ng T.B., Fong W.P., Wan C.C., Yeung H.W. A comparison of human immunodeficiency virus type 1 inhibition by partially purified aqueous extracts of Chinese medicinal herbs. Life Sciences. 1997;60(23):345–351. doi: 10.1016/s0024-3205(97)00227-0. [DOI] [PubMed] [Google Scholar]
- Cooper T.H., Clark J.G., Guzinski J.A. Analysis of curcuminoids by high-performance liquid chromatography. In: Ho C.T., Osawa T., Huang M.T., Rosen R.T., editors. Food Phytochemicals for Cancer Prevention II: Teas, Spices, and Herbs. American Chemical Society; Washington, DC: 1994. pp. 231–236. [Google Scholar]
- Cotelle N., Bernier J.L., Catteau J.P., Pommery J., Wallet J.C., Gaydou E.M. Antioxidant properties of hydroxy-flavones. Free Radical Biology and Medicine. 1996;20(1):35–43. doi: 10.1016/0891-5849(95)02014-4. [DOI] [PubMed] [Google Scholar]
- Gao X.M., Xu Z.M., Li Z.W. People's Health Publishing House; Beijing: 2000. Traditional Chinese Medicines. [Google Scholar]
- Hagerman A.E., Butler L.G. Protein precipitation method for the quantitative determination of tannins. Journal of Agricultural and Food Chemistry. 1978;26(4):809–812. [Google Scholar]
- Halliwell B. Free radicals, antioxidants, and human disease: curiosity, cause, or consequence? Lancet. 1994;344(8924):721–724. doi: 10.1016/s0140-6736(94)92211-x. [DOI] [PubMed] [Google Scholar]
- Hertog M.G.L., Kromhout D., Aravanis C., Blackburn H., Buzina R., Fidanza F., Giampaoli S., Jansen A., Menotti A., Nedeljkovic S., Pekkarinen M., Simic B.S., Toshima H., Feskens E.J.M., Hollman P.C.H., Katan M.B. Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Archives of Internal Medicine. 1995;155(11):281–286. [PubMed] [Google Scholar]
- Ho C.T., Osawa T., Huang M.T., Rosen R.T. American Chemical Society; Washington, DC: 1994. Food Phytochemicals for Cancer Prevention II: Teas, Spices, and Herbs. [Google Scholar]
- Iwashina T. The structure and distribution of the flavonoids in plants. Journal of Plant Research. 2000;113(1111):287–299. [Google Scholar]
- Ji Y.B., He S.W., Ma Y.L., Li J., Yang C., Liu L.L. Heilongjiang Science and Technology Publishing House; Ha'erbin, China: 1999. Pharmacological Action and Application of Anticancer Traditional Chinese Medicines. [Google Scholar]
- Kaur S., Grover I.S., Singh M., Kaur S. Antimutagenicity of hydrolysable tannins from Terminalia chebula in Salmonella typhimurium. Mutation Research. 1998;419:169–179. doi: 10.1016/s1383-5718(98)00130-2. [DOI] [PubMed] [Google Scholar]
- Kikuzaki H., Kawasaki Y., Nakatani N. Structure of antioxidative coumpounds in ginger. In: Ho C.T., Osawa T., Huang M.T., Rosen R.T., editors. Food Phytochemicals for Cancer Prevention II: Teas, Spices, and Herbs. American Chemical Society; Washington, DC: 1994. pp. 237–243. [Google Scholar]
- Krizsán K., Szókán G., Tóth Z.A., Hollósy F., László M., Khlafulla A. HPLC analysis of anthraquinone derivatives in madder root (Rubia tinctorum) and their cell cultures. Journal of Liquid Chromatography and Related Technologies. 1996;19(14):2295–2314. [Google Scholar]
- Kuo S.M. Dietary flavonoid and cancer prevention: evidence and potential mechanism. Critical Reviews in Oncogenesis. 1997;8(1):47–69. doi: 10.1615/critrevoncog.v8.i1.30. [DOI] [PubMed] [Google Scholar]
- Larson R.A. The antioxidants of higher plants. Phytochemistry. 1988;27(4):969–978. [Google Scholar]
- Lien E.J., Li W.Y. Oriental Healing Arts Institute; Long Beach, CA: 1985. Structure-activity Relationship Analysis of Chinese Anticancer Drugs and Related Plants. [Google Scholar]
- Lien E.J., Ren S.J., Bui H.Y.H., Wang R.B. Quantitative structure-activity relationship analysis of phenolic antioxidants. Free Radical Biology and Medicine. 1999;26(3–4):285–294. doi: 10.1016/s0891-5849(98)00190-7. [DOI] [PubMed] [Google Scholar]
- Liu M., Li X.Q., Weber C., Lee C.Y., Brown J., Liu R.H. Antioxidant and antiproliferative activities of raspberries. Journal of Agricultural and Food Chemistry. 2002;50(10):2926–2930. doi: 10.1021/jf0111209. [DOI] [PubMed] [Google Scholar]
- Mau J.L., Lin H.C., Chen C.C. Antioxidant properties of several medicinal mushrooms. Journal of Agricultural and Food Chemistry. 2002;50(21):6072–6077. doi: 10.1021/jf0201273. [DOI] [PubMed] [Google Scholar]
- Mclarty J.W. Antioxidants and cancer: the epidemiologic evidence. In: Garewal H.S., editor. Antioxidants and Disease Prevention. CRC Press; New York: 1997. pp. 45–66. [Google Scholar]
- Miller N.J., Rice-Evans C.A., Davies M.J., Gopinathan V., Milner A. A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clinical Science. 1993;84(4):407–412. doi: 10.1042/cs0840407. [DOI] [PubMed] [Google Scholar]
- Mitscher L.A., Telikepalli H., McGhee E., Shankel D.M. Natural antimutagenic agents. Mutation Research. 1996;350(1):142–143. doi: 10.1016/0027-5107(95)00099-2. [DOI] [PubMed] [Google Scholar]
- Monks N.R., Bordignon S.A.L., Ferraz A., Machado K.R., Faria D.H., Lopes R.M., Mondin C.A., de Souza I.C.C., Lima M.F.S., da Rocha A.B., Schwartsmann G. Anti-tumor screening of Brazilian plants. Pharmaceutical Biology. 2002;40(8):603–616. [Google Scholar]
- Mueller-Harvey I. Analysis of hydrolysable tannins: a review. Animal Feed, Science and Technology. 2001;91:3–20. [Google Scholar]
- Nakatani N. Phenolic antioxidants from herbs and spices. Biofactors. 2000;13(1–4):141–146. doi: 10.1002/biof.5520130123. [DOI] [PubMed] [Google Scholar]
- Ng T.B., Liu F., Wang Z.T. Antioxidative activity of natural products from plants. Life Sciences. 2000;66(8):709–723. doi: 10.1016/s0024-3205(99)00642-6. [DOI] [PubMed] [Google Scholar]
- Niki E. Free radicals, antioxidants, and cancer. In: Ohigashi H., Osawa T., Terao J., Watanabe S., Yoshikawa T., editors. Food Factors for Cancer Prevention. Springer; Tokyo: 1997. pp. 55–57. [Google Scholar]
- Ohigashi H., Murakami A., Koshimizu K. Antitumor promoters from edible plants. In: Ho C.T., Osawa T., Huang M.T., Rosen R.T., editors. Food Phytochemicals for Cancer Prevention II: Teas, Spices, and Herbs. American Chemical Society; Washington, DC: 1994. pp. 251–261. [Google Scholar]
- Owen R.W., Giacosa A., Hull W.E., Haubner R., Spiegelhalder B., Bartsch H. The antioxidant/anticancer potential of phenolic compounds isolated from olive oil. European Journal of Cancer. 2000;36(10):1235–1247. doi: 10.1016/s0959-8049(00)00103-9. [DOI] [PubMed] [Google Scholar]
- Poulson H.E., Prieme H., Loft S. Role of oxidative DNA damage in cancer initiation and promotion. European Journal of Cancer Prevention. 1998;7(1):9–16. [PubMed] [Google Scholar]
- Qiu F., Komatsu K., Kawasaki K., Saito K., Yao X.S., Kano Y. A novel stilbene glucoside, oxyresveratrol 3′-O-beta-glucopyranoside, from the root bark of Morus alba. Planta Medica. 1996;62(6):559–561. doi: 10.1055/s-2006-957972. [DOI] [PubMed] [Google Scholar]
- Re R., Pellegrini N., Proteggente A., Pannala A., Yang M., Rice-Evans C.A. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine. 1999;26(9–10):1231–1237. doi: 10.1016/s0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- Rice-Evans C.A., Miller N.J., Paganga G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Biology and Medicine. 1996;20(7):933–956. doi: 10.1016/0891-5849(95)02227-9. [DOI] [PubMed] [Google Scholar]
- Sakakibara H., Honda Y., Nakagawa S., Ashida H., Kanazawa K. Simultaneous determination of all polyphenols in vegetables, fruits, and teas. Journal of Agricultural and Food Chemistry. 2003;51(3):571–581. doi: 10.1021/jf020926l. [DOI] [PubMed] [Google Scholar]
- Sala A., Recio M.D., Giner R.M., Manez S., Tournier H., Schinella G., Rios J.L. Anti-inflammatory and antioxidant properties of Helichrysum italicum. Journal of Pharmacy and Pharmacology. 2002;54(3):365–371. doi: 10.1211/0022357021778600. [DOI] [PubMed] [Google Scholar]
- Schofield D.M., Mbugua D.M., Pell A.N. Analysis of condensed tannins: a review. Animal Feed Science and Technology. 2001;91:21–40. [Google Scholar]
- Shahidi F., Naczk M. Technomic Pub. Co; Basel, Switzerland: 1995. Food Phenolics: Sources, Chemistry, Effects and Applications. [Google Scholar]
- Shen Y.J., Li Y.K. Shanghai Science and Technology Publishing House; Shanghai: 2002. Pharmacology of Traditional Chinese Medicines. [Google Scholar]
- Son S., Lewis B.A. Free radical scavenging and antioxidant activity of caffeic acid amide and ester analogues: structure-activity relationship. Journal of Agricultural and Food Chemistry. 2002;50(3):468–472. doi: 10.1021/jf010830b. [DOI] [PubMed] [Google Scholar]
- Strack D. Phenolic metabolism. In: Dey P.M., Harbourne J.B., editors. Plant Biochemistry. Academic Press; San Diego, CA: 1997. pp. 387–416. [Google Scholar]
- Sun J., Chu Y.F., Wu X.Z., Liu R.H. Antioxidant and antiproliferative activities of common fruits. Journal of Agricultural and Food Chemistry. 2002;50(25):7449–7454. doi: 10.1021/jf0207530. [DOI] [PubMed] [Google Scholar]
- Tapiero H., Tew K.D., Ba N., Mathé G. Polyphenols: do they play a role in the prevention of human pathologies? Biomedicine and Pharmacotherapy. 2002;56(4):200–207. doi: 10.1016/s0753-3322(02)00178-6. [DOI] [PubMed] [Google Scholar]
- Tsai T.H. Analytical approaches for traditional Chinese medicines exhibiting antineoplastic activity. Journal of Chromatography B. 2001;764(1–2):27–48. doi: 10.1016/s0378-4347(01)00277-8. [DOI] [PubMed] [Google Scholar]
- Velioglu Y.S., Mazza G., Gao L., Oomah B.D. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. Journal of Agricultural and Food Chemistry. 1998;46(10):4113–4117. [Google Scholar]
- Wadsworth T.L., Koop D.R. Effects of the wine polyphenolics quercetin and resveratrol on pro-inflammatory cytokine expression in RAW 264.7 macrophages. Biochemical Pharmacology. 1999;57(8):941–949. doi: 10.1016/s0006-2952(99)00002-7. [DOI] [PubMed] [Google Scholar]
- Xiao C.H., Yang S.S., Hong X.K. Shanghai Science and Technology Publishing House; Shanghai: 2000. The Chemistry of Traditional Chinese Medicines. [Google Scholar]
- Xiao K., Xuan L.J., Xu Y.M., Bai D.L., Zhong D.X., Wu H.M., Wang Z.H., Zhang N.X. Dimeric stilbene glycosides from Polygonum cuspidatum. European Journal of Organic Chemistry. 2002;3:564–568. [Google Scholar]
- Xu G.J., Wang Q., Yu B.Y., Pang M.J., Han L., Chen T., Wang T.Z., Zeng J.H., Li P. Fujian Science and Technology Publishing House; Fuzhou, China: 2000. Coloured Illustrations of Antitumor Chinese Traditional And Herbal Drugs (2nd) [Google Scholar]
- Yang C.S., Landau J.M., Huang M.T., Newmark H.L. Inhibition of carcinogenesis by dietary polyphenolic compounds. Annual Review Nutrition. 2001;21:381–406. doi: 10.1146/annurev.nutr.21.1.381. [DOI] [PubMed] [Google Scholar]
- Zhang Q.M., Gong H.M. People's Health Publishing House; Beijing: 1998. Clinical Practice of Anticancer Traditional Chinese Medicines. [Google Scholar]
- Zheng W., Wang S.Y. Antioxidant activity and phenolic compounds in selected herbs. Journal of Agricultural and Food Chemistry. 2001;49(11):5165–5170. doi: 10.1021/jf010697n. [DOI] [PubMed] [Google Scholar]


