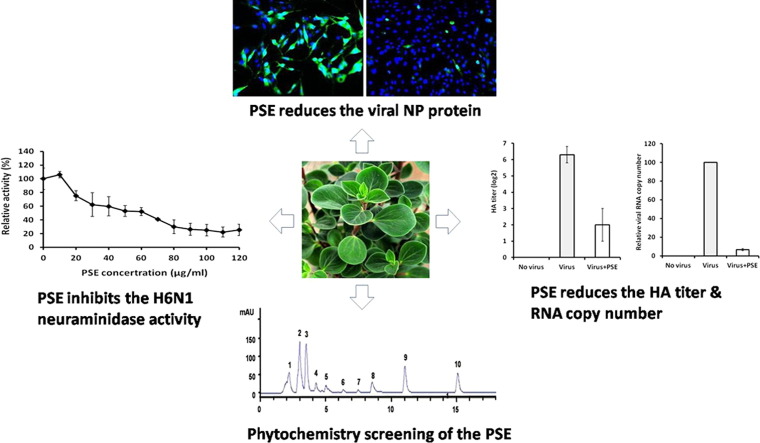
Abstract
Ethnopharmacological relevance
Peperomia sui Lin and Lu (Peperomia sui), a well-known Taiwanese folk medicine, has a broad range of biological effects, especially in treatment of upper respiratory tract diseases. However, no previous study has explored the activity of Peperomia sui against influenza virus infections. This study was carried out to evaluate the anti-influenza virus activity and the potential virucidal effect of the ethanolic extract of Peperomia sui (PSE).
Methods
The anti-H6N1 avian influenza viral activity of PSE against the influenza virus A/Chicken/TW/0518/2011 (H6N1) in chicken fibroblast DF-1 cells was evaluated by cell viability assay, hemagglutination assay, neuraminidase activity assay, indirect immunofluorescence assay and quantitative RT-PCR assay.
Results
PSE significantly increased the viability of cells that were infected by the H6N1 virus. PSE also suppressed the synthesis of viral nucleoprotein (NP), and inhibited the growth of the virus in DF-1 cells. Further, PSE inhibited the neuraminidase activity of H6N1 virus.
Conclusions
The findings of this study provide important information for the exploitation and utilization of Peperomia sui in treatment of influenza infection.
Abbreviations: CC50, 50% cytotoxic concentration; CPE, cytopathic effect; DAPI, (4′,6-diamidino-2-phenylindole); DMEM, Dulbecco׳s modified Eagle׳s medium; EC50, half maximal effective concentration; FITC, fluorescein isothiocyanate; HA, hemagglutinin; IC50, half-maximum inhibitory concentration; IFA, indirect immunofluorescence assay; MEM, minimal essential medium; MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; MU-NANA, 20-(4-Methylumbelliferyl)-a-D-N-acetylneuraminic acid; NA, neuraminidase; PBS, phosphate-buffered saline; qRT–PCR, quantitative reverse transcription–PCR
Keywords: Influenza virus, Peperomia sui, Antiviral activity, Hemagglutination
Graphical abstract
1. Introduction
Influenza is a highly contagious and epidemic respiratory disease caused by type A influenza virus, which has many subtypes with respect to two surface glycoproteins, haemagglutinin (HA) and neuraminidase (NA). Four influenza pandemics have occurred since 1918 ( Webby and Webster, 2003, Lessler et al., 2010). In addition to the currently circulating human viruses of H1N1 and H3N2 subtypes, avian influenza viruses of H5N1, H7N7 and H7N9 subtypes also caused lethal human infections (Subbarao et al., 1998, Fouchier et al., 2004, Zhu et al., 2013). A very recent report showed that the virus of H6N1 subtype is able to cause human infection (Wei et al., 2013). Four anti-influenza viral drugs, amantadine, rimantadine, zanamivir, and oseltamivir are used for treatment of influenza virus infection. These drugs may reduce the length of the illness and the severity of symptoms, but adverse side effects and an emergence of viruses resistant to these drugs are reported (Deyde et al., 2007, Meijer et al., 2009). Therefore, new drugs with greater efficacy and better tolerability than existing treatments are urgently needed for treatment of influenza virus infection.
Traditional herbal medicines have been used to treat infectious diseases for thousands of years in China. Recently, several types of these herbal medicines have been used for treatment of influenza infection (Hayashi et al., 1995, Liu et al., 2010, Zhang et al., 2011, Wang et al., 2013). Peperomia sui Lin and Lu (Peperomia sui), an endemic species in Taiwan, has been used as a folk medicine commonly for the treatment of cough, cold, fever, headache, bronchitis, sore throats, nasal congestion, chronic bronchitis and cancers. The chemical compositions of Peperomia sui was reported (Cheng et al., 2003), but no biological study of Peperomia sui has been reported. In this study, we examined the anti-influenza virus activity of Peperomia sui.
2. Materials and methods
2.1. Preparation of the ethanolic extract of Peperomia sui (PSE)
The whole plants of Peperomia sui were collected from Taiwan Seed Improvement and Propagation Station (TSIPS), Council of Agriculture, Taichung, Taiwan, in August 2010. The plant was identified by Dr. Chi-Luan Wen worked at TSIPS. Herbarium voucher specimens were deposited in the TSIPS. The dried whole plant of Peperomia sui was ground into powder and 100 g of the powder was extracted with 500 ml ethanol at room temperature for 24 h with occasion shaking. After three times extraction, the supernatant was collected and lyophilized, and then the lyophilized powder (12.29 g, 12.29% yield) was stored at -80 °C before use.
2.2. Viruses and cells
Avian influenza virus A/Chicken/TW/0518/2011 (H6N1) was isolated from broiler chickens using the method described previously (Lee et al., 2006). DF-1 cells (a chicken fibroblast cell line; ATCC CRL-12203) were grown in Dulbecco׳s Modified Eagle Medium (DMEM) (Life Technologies Corp., Carlsbad, California, USA) containing 10% fetal bovine serum at 37 °C in the CO2 incubator.
2.3. Cytotoxic activity of PSE on DF-1 cells
DF-1 cells were grown in 96-wells plates (5×103 cells per well) at 37 °C for 24 h. The media was replaced with that containing various concentrations of PSE and the cells were further incubated for 72 h. The cell viability was determined by MTT [3-(4,5-dimethylthiazol-2-yl)- 2,5-diphenyl tetrazolium bromide] assays as described (Mosmann, 1983). In brief, the medium was removed from the 96-wells plates, and the MTT solution was added to each well. After incubation at 37 °C for 2 h, the MTT solution was removed and 100 μL DMSO was added to dissolve formazan crystals. Absorbance at 540 nm of the solution was measured with subtraction of the background measurement at 655 nm. The 50% cytotoxic concentration (CC50) was calculated by regression analysis in Microsoft Excel.
2.4. Hemagglutination (HA) test
The titer of H6N1 virus was determined by the standard HA assay. Two-fold serial dilutions of 25 μL of the virus suspension are made across a plastic U-bottomed 96-well plate. A further 25 μL of PBS was added into each well, and 25 μL of 1% (v/v) chicken red blood cells (RBCs) was dispensed to each well. The plate was tapped gently and incubated at room temperature for 40 min to allow the RBCs to settle. The HA titer of the virus was the reciprocal of highest dilution that gave complete agglutination.
2.5. Cytopathic effect (CPE) inhibition assay of PSE
DF-1 cells were grown in 12-wells plates (5×104 cells per well) at 37 °C for 24 h. The medium was replaced with serum-free medium (1 ml per well) containing 0.3 HA unit of H6N1 virus, 1 μg/ml of Trypsin and various concentrations of PSE. After 1.5 h of incubation, the culture supernatant was removed and the cells were washed three times with phosphate buffered saline (PBS) and the cells were cultured with DMEM containing 10% fetal bovine serum, 1 μg/ml of trypsin and various concentrations of PSE. After 72 h incubation, the CPE was observed with a microscopy, and the viability of cells was determined by MTT assays. The half maximal effective concentration (EC50) was calculated by regression analysis in Microsoft Excel.
2.6. Indirect immunofluorescence assay (IFA)
DF-1 cells were seeded onto the 15 mm glass coverslips in 6-wells plates (1.5×105 cells per well). The cells infected with H6N1 virus in the presence (70 μg/ml) or absence of PSE. Forty-eight hours post infection, the cells on coverslip were fixed with 4% paraformaldehyde for 30 min. After washed three times with PBS and blocking with 2% bovine serum albumin in PBS for 20 min, the cells were reacted with FITC-conjugated mouse monoclonal antibody against influenza A virus nucleoprotein (NP) (Abcam, Cambridge, England) diluted 1:1000 in PBS. After washed three times with PBS, the cells were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) and visualized under a fluorescence microscope.
2.7. Quantitative RT-PCR (qRT-PCR)
DF-1 cells infected with H6N1 virus in the presence or absence of PSE at 37 °C for 72 h. The cells were lysed by three freeze-and-thaw cycles and the total RNA in the lysate was isolated using Trizol reagent (Life Technologies, Carlsbad, California, USA). The sequences of primers used for qRT-PCR of the HA gene of the H6N1 virus were: 5′–CCT CGT ATG GAT AGT AAA AA-3′ (forward) and 5′–GCT TTC AGT TCC CAT TCT AA (reverse). The qRT-PCR was conducted using SYBR® Green RT-PCR Reagents Kit (Life Technologies) and the reaction was performed on an Eco Real-Time PCR System (Illumina Inc., San Diego, California, USA). The PCR condition was 1 cycle of 52 °C for 30 min; 1 cycle of 95 °C for 10 min; 40 cycles of 95 °C 15 s, 55 °C for 30 s and 72 °C for 30 s. The relative copy number of viral RNA was determined by the instrument software.
2.8. Neuraminidase activity assay
The neuraminidase activity of H6N1 virus was determined using the NA-Star Influenza Neuraminidase Inhibitor Resistance Detection Kit (Applied Biosystems, Foster city, California, USA) according to the manufacturer’s protocol. This assay used a chemiluminescent substrate to measure the activity of neuraminidases. In brief, H6N1 viruses were incubated with various concentrations of PSE for 10 min at 37 °C, and then the neuraminidase substrate, 4-MU-NANA [2-(4-methylumbelliferyl)-A-d-nacetylneuraminic acid sodium] was added and incubated for 2 h in the dark. The luminescence signal indicating neuraminidase activity was determined with fluorescence spectrophotometer (excitation 365 nm, emission 460 nm).
2.9. High-performance liquid chromatography analysis
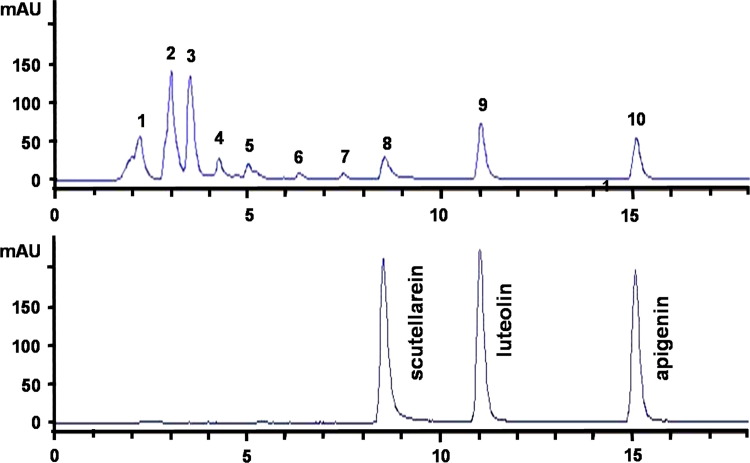
The HPLC fingerprint of PSE was performed using a reverse phase column (LiChrospher® 100 RP-18, 5 μm, 250 mm×4 mm) that was linked to Agilent 1100 System with MWD UV detector (Agilent Technologies, Santa Clara, California, USA). The column temperature was maintained at room temperature. UV detection of samples was performed at a wavelength of 350 nm, and the injection volume was 20 μL. The gradient mobile phase used for analysis was mixed by solvent A (0.1% phosphoric acid solution) and solvent B (acetonitrile). A flow rate of 1 ml/min under the initial condition of 75:25 (A/B) was used. The solvent B was brought to 40% for 15 min, up to 100% for 5 min, and switch to 25% for 5 min. PSE was completely separated by the above gradient elution. HPLC fingerprint chromatogram was established for the quantification and quality control of the isolated sample, which showed scutellarein at 8.5 min, luteolin at 11.0 min, apigenin at 15.0 min. The standard chemicals of scutellarein, luteolin, and apigenin were purchased from Sigma-Aldrich (St. Louis, Missouri, USA).
3. Results
3.1. Cytotoxic and antiviral activity of PSE on DF-1 cells
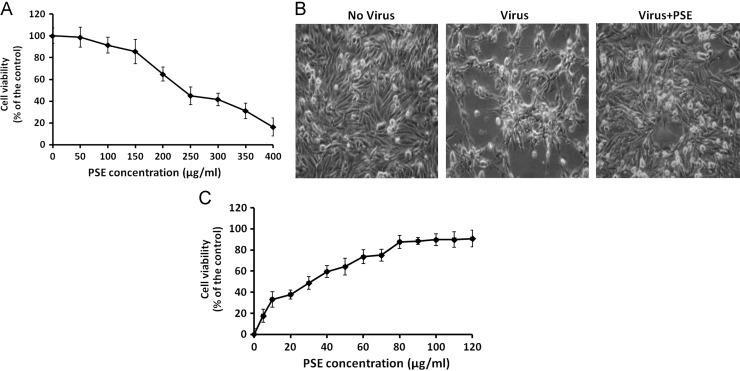
The potential cytotoxicity of PSE on the DF-1 cells was evaluated by MTT assays. The result showed that treatment with PSE reduced the viability of DF-1 cells and the CC50 of PSE on DF-1 cells was 262.8 μg/ml ( Fig. 1A). At concentration of 100 μg/ml or less, PSE had no significant cytotoxicity on DF-cells. Next, the antiviral activity of PSE was evaluated. Microscopic examination showed that DF-1 cells infected with H6N1 virus showed cytopathic effects including rounding, detachment and death of the cells (Fig. 1B). Treatment of DF-1 cells with 100 μg/ml PSE significantly reduced the CPE produced by the virus (Fig. 1B). MTT assays revealed that PSE dose-dependently increased the viability of virus-infected cells, and the EC50 value was 34.8 μg/ml (Fig. 1C). At 70 μg/ml concentration (approximately two fold of EC50 value), PSE effectively increased the viability of the cells and exhibited no obvious cytotoxicity. This concentration of PSE was therefore selected for further experiments.
Fig. 1.
(A) The cytotoxic effect of PSE on DF-1 cells. The cells were treated with various concentration of PSE and the cell viability was determined by MTT assays. (B) The anti-viral effect of PSE. DF-1 cells were infected with H6N1 virus in the absence or presence of PSE. The morphological investigation showed that PSE reduced the CPE caused by H6N1 virus on DF-1 cells. (C) PSE increased the viability of DF-1 cells infected with H6N1 virus. DF-1 cells were infected with H6N1 virus with various concentrations of PSE and the cell viability was determined by MTT assays. Each value in (A) and (C) is the result of mean±S.D. of three independent experiments.
3.2. PSE reduces the expression of the viral NP protein
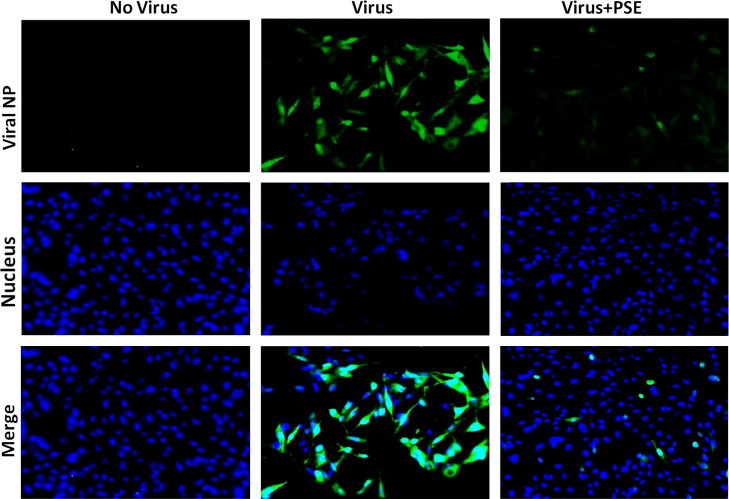
To examine whether PSE impaired the synthesis of viral protein, DF-1 cells were infected with H6N1 virus in the presence or absence of PSE. The expression of the viral NP protein was monitored by IFA using a FITC-labeled monoclonal antibody against the NP protein. As depicted in Fig. 2, a strong green fluorescence signal was observed in cytoplasm of the majority of virus-infected cells without PSE treatment. In contrast, only a weak fluorescence signal was observed in a small number of the virus-infected cells that were treated with PSE. This result suggested that treatment with the PSE inhibited the expression of the viral NP protein in DF-1 cells.
Fig. 2.
PSE reduces expression of the viral nucleoprotein (NP). DF-1 cells were infected with H6N1 virus and the level of viral NP protein expressed in DF-1 cells was examined by immunostaining. Left column: DF-1 cells not infected with virus. Middle column: DF-1 cells infected with virus. Right column: DF-1 cells infected with virus in the presence of PSE. The green fluorescence of FITC indicates the presence of the viral NP protein while blue fluorescence of DAPI indicates the nucleus of the cell.
3.3. PSE inhibits the growth of H6N1 virus
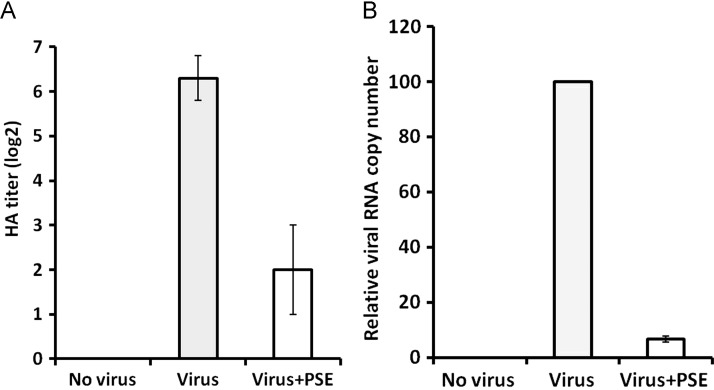
The effect of PSE treatment on viral growth was investigated by HA tests and qRT-PCR. The result of HA tests showed that the virus propagating from cells untreated with PSE had a mean HA titer of 6.3log 2 at 72 h post infection ( Fig. 3A). In contrast, the virus from cells treated with PSE (70 μg/ml) had a mean titer of only 2.0 log2 (Fig. 3A). Significant difference (p<0.01) was found between the untreated and treated groups. No HA titer was detected in the control group (Fig. 3A). This result indicated that treatment with PSE inhibited the growth of H6N1 virus.
Fig. 3.
PSE inhibits the propagation of H6N1 virus. (A) The HA titer of virus propagating in cells untreated (gray bar) or treated (white bar) with PSE was determined by HA tests. (B) The relative copy number of viral RNA in cells untreated (gray bar) or treated (white bar) with PSE was determined by qRT-PCR. Each value in (A) and (B) is the result of mean±S.D. of three independent experiments.
The result of qRT-PCR showed that treatment with PSE (70 μg/ml) drastically reduced the copy number of viral RNA synthesis at 72 h post infection. When the relative copy number of viral RNA in the untreated cells was set to 100, the copy number of viral RNA in PSE-treated cells was only 6.6 (Fig. 3B). Significant difference (p<0.01) was found between the untreated and treated groups. Taken together, these results indicated that PSE effectively inhibited the growth of H6N1 virus in DF-1 cells.
3.4. PSE inhibits the H6N1 neuraminidase activity
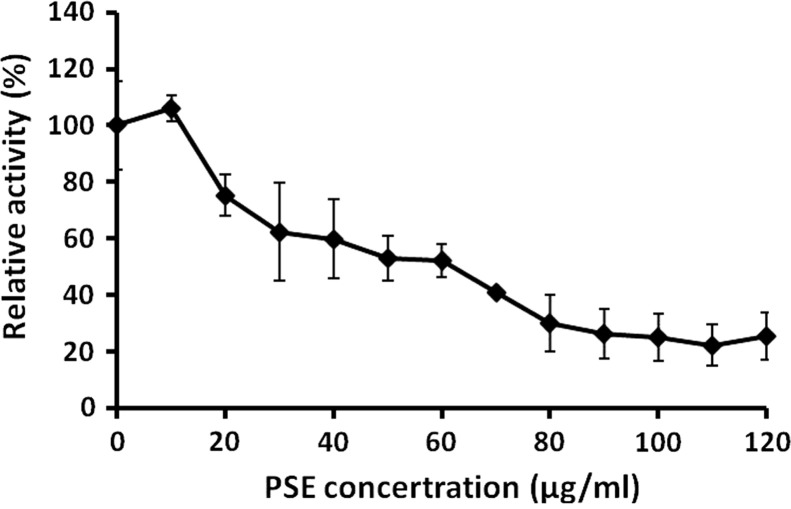
Neuraminidase (NA) has been regarded as one of the most important targets to screen the drugs of anti-influenza viruses A and B (Liu et al., 2005). The effect of PSE on the viral NA activity was investigated. The result showed that PSE reduced NA activity dose-dependently ( Fig. 4). PSE at concentration of 120 μg/mL (the maximum concentration tested) reduced the H6N1 NA activity to 24.3±11% of the control. These results indicated that the PSE might suppress the virus release step through inhibition of neuraminidase.
Fig. 4.
PSE inhibits the neuraminidase activity of influenza virus. H6N1 virus was incubated with various concentrations of PSE, and the NA activity of the virus was determined. Each value is the result of mean±S.D. of three independent experiments.
3.5. Phytochemistry screening of the PSE
In order to quality control and exploiting the active components of PSE, the HPLC fingerprint chromatogram was established. The chromatogram of the PSE showed ten major peaks ( Fig. 5), and three of them were identified to be scutellarein (retention time=8.5 min, peak 8), luteolin (retention time=11.2 min, peak 9) and apigenin (retention time=15.1 min, peak 10).
Fig. 5.
The HPLC fingerprint chromatogram of PSE. Ten major peaks were observed and three of them were identified as scutellarein (retention time=8.5 min, peak 8), luteolin (retention time=11.2 min, peak 9) and apigenin (retention time=15.1 min, peak 10).
4. Discussion
Influenza is a highly contagious disease that can cause high morbidity and mortality in an epidemic. The clinical utility of anti-influenza viral drugs has been limited by side effects and emergence of resistant strain (Deyde et al., 2007, Beigel and Bray, 2008, Meijer et al., 2009). Hence, new approaches for the control of infection with influenza viruses must be explored. Traditional Chinese medicinal herbs may be a potential alternative medicine of choice for treatment of this disease. Recently, a number of clinical trials of traditional Chinese medicines for influenza have been conducted (Wang et al., 2011, Nabeshima et al., 2012, Jiang et al., 2013;). Data presented in this study show that PSE is a potent inhibitor of the viral growth in chicken fibroblast DF-1 cells. Further, PSE had low toxicity and low effective concentration against H6N1 influenza virus. The CC50 and EC50 value of PSE were 262.8 μg/ml and 34.8 μg/ml, respectively, and the selected index (SI) value was 7.6 (SI=CC50/EC50).
Peperomia sui has been commonly used in folk remedies to treat respiratory tract disorders in Taiwan. This is the first study that demonstrates the anti-influenza viral activity of PSE in vitro against the H6N1 virus. The results using immunofluorescence staining showed that the amount of the NP protein was greatly reduced by PSE treatment. This result suggested that inhibition of the viral protein synthesis may be one of the anti-viral mechanisms of PSE.
It is well documented that NA plays an important role in the release of newly assembled virus particles from infected cells by cleavage of target cell receptor sialic acid moieties (Gamblin and Skehel, 2010). Our results showed that PSE effectively inhibited the neuraminidase activity and this inhibition may also play a role in the anti-viral activity of PSE. Overall, our observations strongly suggest that PSE may affect various steps of influenza viral infectious cycle.
A previous phytochemical study shows that over 34 compounds have been isolated and identified from methanol extract of Peperomia sui, consisting of surinone, suranone, acylresorcinol, eugenol, phytol, peperomin, sitosterol, vanillic acid, sesamin, salidroside, betulinic acid, hinokinin, etc. (Cheng et al., 2003). In the current study, three flavonoid compounds, scutellarein, luteolin and apigenin, were identified in PSE. Scutellarein has been shown to be a strong inhibitor of the severe acute respiratory syndrome (SARS) virus helicase, and may serve as an important resource for the future development of anti-SARS medications (Keum and Jeong, 2012). Apigenin and luteolin are previously shown to inhibit influenza A virus neuraminidases ( Liu et al., 2008, Grienke et al., 2012), and they can effectively reduce neuraminidase activity of H5N1 virus (Sithisarn et al., 2013). In this study, the PSE-mediated inhibition of the growth of influenza virus H6N1 might also be due to anti-influenza viral effects from apigenin and luteolin. However, additional studies are necessary to identify the possible correlation between biological activities and chemical composition of PSE to ensure the appropriate medicinal use of this medicinal herb.
We conclude that the ethanolic extract of Peperomia sui has low toxicity and exhibits strong anti-influenza virus activity in vitro. This activity may be through the suppression of synthesis of viral protein and inhibition of viral neuraminidase activity. Our results provide evidence that constituents and extracts of Peperomia sui may be considered as an alternative agent for treatment of influenza virus infections. However, further study is required to evaluate whether PSE treatment also results in antiviral activity in vivo.
Acknowledgments
The authors would like to thank the Taichung Veterans General Hospital (TCVGH1017305C) and the Taichung Veterans General Hospital and National Chung Hsing University (TCVGH-NCHU 1017612) for financially supporting this research.
Contributor Information
Shih-Lan Hsu, Email: h2326@vghtc.gov.tw.
Poa-Chun Chang, Email: pcchang@mail.nchu.edu.tw.
References
- Beigel J., Bray M. Current and future antiviral therapy of severe seasonal and avian influenza. Antivir. Res. 2008;78:91–102. doi: 10.1016/j.antiviral.2008.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng M.J., Lee S.J., Chang Y.Y., Wu S.H., Tsai I.L., Jayaprakasam B., Chen I.S. Chemical and cytotoxic constituents from Peperomia sui. Phytochemistry. 2003;63:603–608. doi: 10.1016/s0031-9422(03)00183-3. [DOI] [PubMed] [Google Scholar]
- Deyde V.M., Xu X., Bright R.A., Shaw M., Smith C.B., Zhang Y., Shu Y., Gubareva L.V., Cox N.J., Klimov A.I. Surveillance of resistance to adamantanes among influenza A(H3N2) and A(H1N1) viruses isolated worldwide. J. Infect. Dis. 2007;196:249–257. doi: 10.1086/518936. [DOI] [PubMed] [Google Scholar]
- Fouchier R.A., Schneeberger P.M., Rozendaal F.W., Broekman J.M., Kemink S.A., Munster V., Kuiken T., Rimmelzwaan G.F., Schutten M., Van Doornum G.J., Koch G., Bosman A., Koopmans M., Osterhaus A.D. Avian influenza A virus (H7N7) associated with human conjunctivitis and a fatal case of acute respiratory distress syndrome. Proc. Natl. Acad. Sci. USA. 2004;101:1356–1361. doi: 10.1073/pnas.0308352100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gamblin S.J., Skehel J.J. Influenza hemagglutinin and neuraminidase membrane glycoproteins. J. Biol. Chem. 2010;285:28403–28409. doi: 10.1074/jbc.R110.129809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grienke U., Schmidtke M., von Grafenstein S., Kirchmair J., Liedl K.R., Rollinger J.M. Influenza neuraminidase: a druggable target for natural products. Nat. Prod. Rep. 2012;29:11–36. doi: 10.1039/c1np00053e. [DOI] [PubMed] [Google Scholar]
- Hayashi K., Kamiya M., Hayashi T. Virucidal effects of the steam distillate from Houttuynia cordata and its components on HSV-1, influenza virus, and HIV. Planta Med. 1995;61:237–241. doi: 10.1055/s-2006-958063. [DOI] [PubMed] [Google Scholar]
- Jiang L., Deng L., Wu T. Chinese medicinal herbs for influenza. Cochrane Database Syst. Rev. 2013:3. doi: 10.1002/14651858.CD004559.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keum Y.S., Jeong Y.J. Development of chemical inhibitors of the SARS coronavirus: viral helicase as a potential target. Biochem. Pharmacol. 2012;84:1351–1358. doi: 10.1016/j.bcp.2012.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee M.S., Chang P.C., Shien J.H., Cheng M.C., Chen C.L., Shieh H.K. Genetic and pathogenic characterization of H6N1 avian influenza viruses isolated in Taiwan between 1972 and 2005. Avian Dis. 2006;50:561–571. doi: 10.1637/7640-050106R.1. [DOI] [PubMed] [Google Scholar]
- Lessler J., dos Santos T., Aguilera X., Brookmeyer R., Group P.I.T.W., Cummings D.A. H1N1pdm in the Americas. Epidemics. 2010;2:132–138. doi: 10.1016/j.epidem.2010.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu A., Cao H., Du G. Drug screening for influenza neuraminidase inhibitors. Sci. Chin. Life Sci. 2005;48:1–5. doi: 10.1360/062004-69. [DOI] [PubMed] [Google Scholar]
- Liu A.L., Liu B., Qin H.L., Lee S.M., Wang Y.T., Du G.H. Anti-influenza virus activities of flavonoids from the medicinal plant Elsholtzia rugulosa. Planta Med. 2008;74:847–851. doi: 10.1055/s-2008-1074558. [DOI] [PubMed] [Google Scholar]
- Liu F.Z., Shi H., Shi Y.J., Liu Y., Jin Y.H., Gao Y.J., Guo S.S., Cui X.L. Pharmacodynamic experiment of the antivirus effect of Houttuynia cordata injection on influenza virus in mice. Yao Xue Xue Bao. 2010;45:399–402. [PubMed] [Google Scholar]
- Meijer A., Lackenby A., Hungnes O., Lina B., van-der-Werf S., Schweiger B., Opp M., Paget J., van-de-Kassteele J., Hay A., Zambon M. Oseltamivir-resistant influenza virus A (H1N1), Europe, 2007-08 season. Emerg. Infect. Dis. 2009;15:552–560. doi: 10.3201/eid1504.081280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- Nabeshima S., Kashiwagi K., Ajisaka K., Masui S., Takeoka H., Ikematsu H., Kashiwagi S. A randomized, controlled trial comparing traditional herbal medicine and neuraminidase inhibitors in the treatment of seasonal influenza. J. Infect. Chemother. 2012;18:534–543. doi: 10.1007/s10156-012-0378-7. [DOI] [PubMed] [Google Scholar]
- Sithisarn P., Michaelis M., Schubert-Zsilavecz M., Cinatl J., Jr. Differential antiviral and anti-inflammatory mechanisms of the flavonoids biochanin A and baicalein in H5N1 influenza A virus-infected cells. Antivir. Res. 2013;97:41–48. doi: 10.1016/j.antiviral.2012.10.004. [DOI] [PubMed] [Google Scholar]
- Subbarao K., Klimov A., Katz J., Regnery H., Lim W., Hall H., Perdue M., Swayne D., Bender C., Huang J., Hemphill M., Rowe T., Shaw M., Xu X., Fukuda K., Cox N. Characterization of an avian influenza A (H5N1) virus isolated from a child with a fatal respiratory illness. Science. 1998;279:393–396. doi: 10.1126/science.279.5349.393. [DOI] [PubMed] [Google Scholar]
- Wang C., Cao B., Liu Q.Q., Zou Z.Q., Liang Z.A., Gu L., Dong J.P., Liang L.R., Li X.W., Hu K., He X.S., Sun Y.H., An Y., Yang T., Cao Z.X., Guo Y.M., Wen X.M., Wang Y.G., Liu Y.L., Jiang L.D. Oseltamivir compared with the Chinese traditional therapy maxingshigan-yinqiaosan in the treatment of H1N1 influenza: a randomized trial. Ann. Intern. Med. 2011;155:217–225. doi: 10.7326/0003-4819-155-4-201108160-00005. [DOI] [PubMed] [Google Scholar]
- Wang X., Xu X., Li Y., Li X., Tao W., Li B., Wang Y., Yang L. Systems pharmacology uncovers Janus functions of botanical drugs: activation of host defense system and inhibition of influenza virus replication. Integr. Biol. (Camb) 2013;5:351–371. doi: 10.1039/c2ib20204b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webby R.J., Webster R.G. Are we ready for pandemic influenza? Science. 2003;302:1519–1522. doi: 10.1126/science.1090350. [DOI] [PubMed] [Google Scholar]
- Wei S.H., Yang J.R., H.S. W., M.C. C., Lin J.S., Lin C.Y., Liu Y.L., Lo Y.C., Yang C.H., J.H. C., M.C. L., W.C. C., Liao C.H., M.S. L., Huang W.T., P.-J. C., Liu M.T., F.C. C. Human infection with avian influenza A H6N1 virus: an epidemiological analysis. The Lancet Respiratory Medicine. 2013;(13):70221–70222. doi: 10.1016/S2213-2600(13)70221-2. (Early Online Publication14 November 2013) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H., Song J., Shi J. Screen anti-influenza virus serum marker of Lonicera japonica by proteomics technology. Zhongguo Zhong Yao Za Zhi. 2011;36:1071–1074. [PubMed] [Google Scholar]
- Zhu H., Wang D., Kelvin D.J., Li L., Zheng Z., Yoon S.W., Wong S.S., Farooqui A., Wang J., Banner D., Chen R., Zheng R., Zhou J., Zhang Y., Hong W., Dong W., Cai Q., Roehrl M.H., Huang S.S., Kelvin A.A., Yao T., Zhou B., Chen X., Leung G.M., Poon L.L., Webster R.G., Webby R.J., Peiris J.S., Guan Y., Shu Y. Infectivity, transmission, and pathology of human-isolated H7N9 influenza virus in ferrets and pigs. Science. 2013;341:183–186. doi: 10.1126/science.1239844. [DOI] [PubMed] [Google Scholar]