Highlights
-
•
Deoxynivalenol (DON) is a common mycotoxin found in pig feed.
-
•
PRRS is economically important for North American swine industry.
-
•
MLV vaccine is widely used to control PRRS.
-
•
DON-contaminated diet could inhibit vaccination efficiency of PRRSV MLV.
Keywords: Pig, DON mycotoxin, PRRSV, Vaccination
Abstract
Cereal commodities are frequently contaminated with mycotoxins produced by the secondary metabolism of fungal infection. Among these contaminants, deoxynivalenol (DON), also known as vomitoxin, is the most prevalent type B trichothecene mycotoxin worldwide. Pigs are very sensitive to the toxic effects of DON and are frequently exposed to naturally contaminated feed. Recently, DON naturally contaminated feed has been shown to decrease porcine reproductive and respiratory syndrome virus (PRRSV) specific antibody responses following experimental infection. The objective of this study was to determine the impact of DON naturally contaminated feed on the immune response generated following vaccination with PRRSV live attenuated vaccine. Eighteen pigs were randomly divided into three experimental groups of 6 animals based on DON content of the diets (0, 2.5 and 3.5 mg DON/kg). They were fed these rations one week prior to the vaccination and for all the duration of the immune response evaluation. All pigs were vaccinated intra-muscularly with one dose of Ingelvac® PRRSV modified live vaccine (MLV). Blood samples were collected at day −1, 6, 13, 20, 27 and 35 post vaccination (pv) and tested for PRRSV RNA by RT-qPCR and for virus specific antibodies by ELISA. Results showed that ingestion of DON-contaminated diets significantly decreased PRRSV viremia. All pigs fed control diet were viremic while only 1 (17%) and 3 (50%) out of 6 pigs were viremic in the groups receiving 3.5 and 2.5 mg of DON/kg, respectively. Subsequently, all pigs fed control diet developed PRRSV specific antibodies while only viremic pigs that were fed contaminated diets have developed PRRSV specific antibodies. These results suggest that feeding pigs with DON-contaminated diet could inhibit vaccination efficiency of PRRSV MLV by severely impairing viral replication.
1. Introduction
Animal feeds are frequently contaminated with various mycotoxins produced by the secondary metabolism of diverse fungal contaminants in response to stress [1]. Among them, Fusarium spp. are the most prevalent mycotoxin producing fungi in temperate regions [2]. Trichothecenes, including deoxynivalenol (DON) and T-2 toxin, zearalenone and fumonisin B1, are toxicologically significant Fusarium spp. mycotoxins [3]. DON, also known as vomitoxin, is the most prevalent mycotoxin in grain [4] and because of the high percentage of cereal in pig diets, swine are frequently exposed to this toxin. In this animal, dietary concentrations between 2 and 5 mg DON/kg are associated with feed refusal and reduced weight gain, whereas concentrations over 20 mg DON/kg cause abdominal distress, diarrhea, vomiting and even shock or death [5]. High contamination levels are rare in modern agricultural practice, instead chronic exposure to low doses of DON is more frequent [6]. DON possesses also immunomodulatory properties [7]; in mouse, low concentrations exert pro-inflammatory effects by inducing cytokines and chemokines expression in mononuclear phagocytes, as a consequence of mitogen-activated protein kinases (MAPK) activation [8]. In the same model, dietary exposure to DON upregulates serum IgA and leads to decreased serum concentrations of IgM and IgG [9]. In pigs, DON has also been shown to activate MAPK in the intestine [10]. However, studies in primary porcine macrophages provide evidence for a lack of COX-2 and IL-6 activation by DON in this cell type, suggesting a distinct mode of action in this species [11]. Unlike mice, several investigations on pigs indicate only marginal or no effects of DON on IgA level [4]. Nonetheless, other studies reported an increase of specific-IgA accompanied with a decrease of specific IgG and cytokines activation following immunization with ovalbumin in DON-fed piglets [12], [13]. More recently, DON naturally contaminated diet has also been shown to decrease porcine reproductive and respiratory syndrome (PRRS) virus-specific antibody responses following experimental infection [14].
Economically, PRRS is the most important viral disease in swine livestock worldwide [15]. Causative agent of PRRS is a small enveloped positive-sense single-stranded RNA virus classified in the order Nidovirales, family Arteriviridae, genus Arterivirus, which also includes lactate dehydrogenase-elevating virus of mice, simian hemorrhagic fever virus and equine arteritis virus [16]. PRRSV causes common clinical signs such as anorexia, fever, and lethargy. In sows, PRRSV is responsible for reproductive failure, characterized by late-term abortions, increased numbers of stillborn fetuses, and/or premature, weak pigs. Furthermore, PRRSV is responsible for respiratory problems in growing and finishing pigs [17], [18]. Measures currently used to control PRRS include management practices such as whole herd depopulation/repopulation or herd closure, constraining bio-security measures, surveillance of herd status and vaccination [19]. Modified live vaccines (MLV) against PRRSV have been widely used and have shown some efficacy in reducing clinical disease severity, as well as viremia duration and virus shedding [20]. Given the impact of DON on the pig immune response and wide spread use of PRRS MLV vaccine for the control of this economically devastating disease, the objective of this study was to determine the effect of DON naturally contaminated feed on the immune response generated following vaccination with PRRS MLV.
2. Materials and methods
2.1. Animals
The experiment was conducted at the Faculté de médecine vétérinaire, Université de Montréal. Animal care procedures followed the guidelines of the Canadian Council on Animal Care and the protocol was approved by the institutional animal care committee (Protocol #14-Rech-1751). Eighteen commercial crossbred piglets, PCR and serum-negative for PRRSV were purchased locally at 4 weeks of age. After one week of acclimation on a commercial ration, piglets were randomly divided into 3 experimental groups of 6 animals, housed separately and fed ad libitum naturally contaminated diets containing 0 (control diet), 2.5 or 3.5 mg/kg of DON for the duration of the experiment.
2.2. Experimental diets
The experimental diets used in this study were formulated according to the energy and amino acid requirements for piglets as previously described [14]. Dietary contents of mycotoxins were analyzed in the final diet through ultra-performance liquid chromatography/electrospray ionization tandem mass spectrometry as previously described [14].
2.3. PRRSV vaccination
Before the beginning of the study, animals were weighed to assure the homogeneity of the experimental groups. No significant difference in body weight was found between experimental groups with a one-way ANOVA model using the parametric Tukey test (P > 0.05) (data not shown). After 1 week of acclimation with experimental diet, all animals were vaccinated intramuscularly (im) into the neck muscles using a 20G 1 in. needle with Ingelvac® PRRS MLV vaccine (lot #245-F31) as recommended by the manufacturer (Boehringer Ingelheim Vetmedica, St. Joseph, MO, USA).
2.4. Body weight and blood collection
Pigs were weekly monitored for body weight before vaccination and for 35 days post-vaccination (pv). Average daily gain (ADG) was calculated for each week of the experiment by subtracting the body weight from the previous week of the body weight at the measured time and divided by 7 days.
Blood samples were collected at days −1, 6, 13, 20, 27 and 35 pv to evaluate PRRSV viremia by RT-qPCR and to measure specific antibody response by ELISA. Serum samples were stored frozen at −80 °C for further analysis.
2.5. PRRSV quantification
Sera were analyzed for the presence of PRRSV RNA viral genome using RT-qPCR assay as previously described [21]. QIAamp Viral RNA kit (Qiagen) was used to isolate viral RNA from serum samples according to the manufacturer's instructions. A commercial PRRSV RT-qPCR diagnostic kit (NextGen, Tetracore Inc., Gaithersburg, MD, USA) was used for PRRSV quantification as recommended by the manufacturer. The quantification of PRRSV was determined by comparing sample results with a standard curve based on the amount of serially diluted PRRSV IAF-Klop reference strain produced in MARC-145 cells and titrated as TCID50/mL in the MARC-145-infected cell [21]. The PRRSV RT-qPCR results were expressed in TCID50/mL of serum.
2.6. PRRSV specific antibodies
Sera were assayed for virus-specific antibody by ELISA using the Herdchek PRRS X3 diagnostic kits (IDEXX Laboratories, Portland, Maine, USA). Serum were diluted 1/40 in a diluent supplied by the manufacturer and the assay was performed following the manufacturer's instructions. A sample-to-positive (S:P) ratio equal or greater than 0.4 was considered positive.
2.7. Virus neutralizing antibody titer
Serum samples were heat inactivated (56 °C, 30 min) and serially diluted before the titration. The serial dilutions of serum samples were mixed with equal volume PRRSV VR-2332 vaccinal strain containing 100 TCID50 of the virus. After incubation at 37 °C for 2 h, the mixtures were transferred to MARC-145 monolayers in 96-well plates and incubated for an additional 72 h at 37 °C in a humidified atmosphere containing 5% CO2. Cells were then examined for cytopathic effects (CPE). CPE was used to determine the end-point titers that were calculated as the reciprocal of the highest serum dilution required to neutralize 100 TCID50 of PRRSV.
2.8. Statistical analysis
Results are expressed as mean ± SEM. All statistical analyses were performed using GraphPad Prism software (version 5.03, GraphPad Prism software Inc., San Diego, CA). Data were statistically analyzed using a one-way ANOVA with Dunnett's multiple comparison test, using animal receiving control diet as control group. For PRRSV-specific antibody response, pair-wise mean comparisons between control and DON treated animals were made using Welch's unpaired t test. P < 0.05 was considered to reflect statistically significant differences.
3. Results
3.1. Growth performance
ADG was evaluated each week of the experiment. DON naturally contaminated diet had no significant effect on ADG during the week prior to vaccination (Fig. 1 at day −1 post vaccination (dpv)). Results also showed that contaminated diet with DON at 3.5 mg/kg significantly decreased ADG (P < 0.05) after vaccination with a loss of 32%, 24%, 12% and 18% of kg/day at day 6, 13, 27 and 34, respectively, when compared to control group (Fig. 1).
Fig. 1.
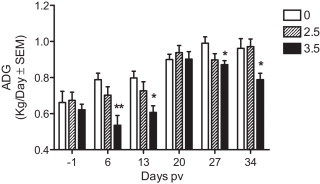
Week by week average daily gain (ADG) before and after vaccination. ADG was calculated for each week of the experiment by subtracting pigs’ body weights of the previous week with the body weight at the measured time and divided by 7 days. * Indicates difference between DON fed groups and control for each time point (P < 0.05), ** (P < 0.01).
3.2. Viremia
Presence of PRRSV mRNA in sera was evaluated by RT-qPCR prior and after vaccination, at day −1, 6, 13, 20 pv. All piglets were PCR negative prior to vaccination (data not shown). All pigs fed control diet were viremic at day 6 pv, while none and 3 out of 6 pigs were viremic in the groups receiving 3.5 and 2.5 mg of DON/kg of the diet, respectively (Fig. 2A). At day 13 pv, the viral burden was significantly lower (P < 0.05) in both groups fed DON-contaminated diets compared to the group fed control diet (Fig. 2B). At day 20 pi, all piglets had very low PRRSV titers or were PCR negative and no significant differences were observed between experimental groups (Fig. 2C).
Fig. 2.
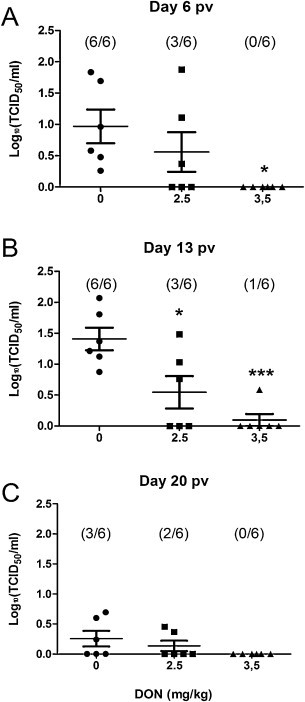
Effect of DON naturally contaminated diets on PRRSV viremia following vaccination with PRRS MLV vaccine. Blood was collected at day 6 (A), 13 (B), and 20 (C) pv and serum tested for the presence of PRRSV RNA by RT-qPCR. Data are expressed in TCID50/mL. * Indicates difference between DON fed groups and control for each time point (P < 0.05), *** (P < 0.001).
3.3. Antibody response
Presence of PRRSV-specific IgG was evaluated using a commercial ELISA kit (Herdcheck PRRS X3) at day −1, 13, 20, 27 and 34 pv. All piglets were serum-negative prior to vaccination (data not shown). At day 13 pv, 5 out of 6 pigs fed control diet had seroconverted, while none and 3 out of 6 pigs had seroconverted in groups receiving 3.5 and 2.5 mg of DON/kg of the diet, respectively (Fig. 3A). Antibody titers were significantly higher (P < 0.05) in group fed control diet compared to the group fed DON-contaminated diet at 3.5 mg/kg for all evaluated days (Fig. 3A–D). From day 27 pv, all pigs fed control diet developed PRRSV specific antibodies while only viremic pigs, i.e. 1 and 3 that were fed 3.5 and 2.5 mg of DON/kg of the diet respectively, developed PRRSV specific antibodies (Fig. 3C and D).
Fig. 3.
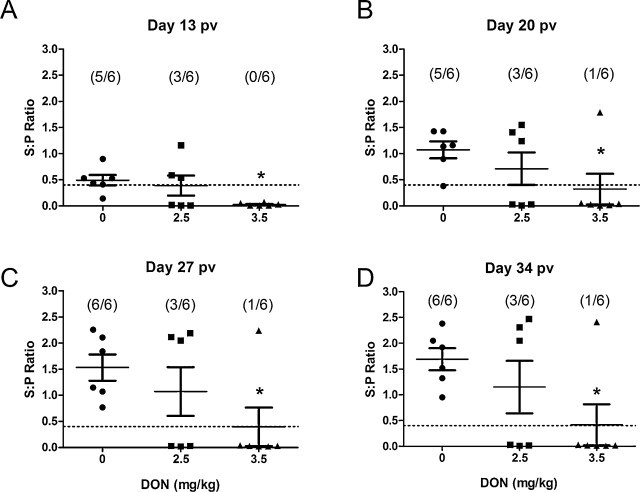
Effect of DON naturally contaminated diets on PRRS-specific antibody titer. Blood was collected at day (A) 13, (B) 20, (C) 27 and (D) 34 pv and sera were tested for the presence of specific PRRSV antibodies using a commercial ELISA kit (HerdChek-PRRS®, IDEXX). Data are expressed in sample to positive (S:P) ratio. S:P ratio equal or greater than 0.4 was considered positive. The dash bar represents value of negative–positive cut-off s/p ratio. * Indicates difference between DON fed groups and control for each time point (P < 0.05).
3.4. Neutralizing antibody response
Presence of PRRSV-neutralizing antibodies was evaluated at day 34 pv, using a PRRSV microneutralizing assay in MARC-145 cells. Results showed that the majority (5 out of 6) of pigs fed control diet mounted a neutralizing antibody response compared to 1 and 3 in pigs fed 3.5 and 2.5 mg of DON/kg of the diet, respectively (Fig. 4 ). PRRSV-neutralizing antibody response was significantly lower in pigs fed DON-contaminated diet at 3.5 mg/kg.
Fig. 4.
Effect of DON naturally contaminated diets on PRRSV neutralizing antibody titer. Blood was collected at day 34 pv and sera were tested for the presence of PRRSV neutralizing antibodies to VR-2332 strain. Data are expressed as reciprocal dilution titer. The dash bar represents the limit of detection. * Indicates difference between DON fed groups and control (P < 0.05).
4. Discussion
Contamination of cereal by mycotoxins produced by Fusarium spp. is a serious problem in animal nutrition worldwide, especially in pigs [22]. Main toxicological effects of DON-contaminated feed are decreased body weight gain and voluntary feed intake [23], [24]. Here, DON naturally contaminated feed had no significant impact on ADG prior to vaccination. Even though one other study, also showed no significant effect of DON on ADG [13], these results must be analyzed carefully because chronic effects of DON on ADG might be observed after 3 weeks of diet consumption [25]. However the ADG decreased significantly after vaccination in the group fed 3.5 mg/kg of DON. Decreased ADG in pig has also been observed soon after vaccination [26], [27]. The present results show that diets contaminated with DON interact with PRRS attenuated vaccine and increases the loss of weight gain after vaccination. Similar effects have been previously observed after experimental infection with PRRSV in pigs fed DON naturally contaminated diet [14].
PRRS MLV vaccine has shown some protective efficacy against PRRSV clinical disease induced by the strains that are genetically related to the vaccine [28]. However, this vaccine elicits relatively weak neutralizing antibody and cell-mediated immune responses. PRRSV-specific antibodies appear approximately two weeks, and peak around four weeks after vaccination [29]. The majority of the antibodies are directed against viral nucleocapsid proteins (N) which have no neutralizing activity [29]. Generation of neutralizing antibodies is delayed in PRRSV infection and usually appears three to four weeks after vaccination [30]. Typically, serum neutralizing antibody titers are unusually low in comparison to those induced by other viruses [20]. The present results showed that DON naturally contaminated feed significantly decreased the antibody response generated following PRRS MLV vaccination. Vaccine failures are not uncommon in the field [31] and can be virus related due to a lack of cross-protection between the vaccine and field strains [32], [33] or immune related due to inefficient immune response [34]. Here, PRRS vaccine failure appears to be caused by an inefficient immune response following the ingestion of feed naturally contaminated with DON. Indeed, contamination of feed with DON has been previously implicated in vaccine failure due to the effects of DON on the immune system [12], [35], [36]. Moreover, ingestion of DON naturally contaminated feed have been previously shown to decrease PRRSV-specific antibody titers after experimental PRRSV infection [14]. In the case of PRRS MLV, live PRRSV replication is required to provide immunological protection against PRRSV infection [37]. The present results showed that ingestion of DON at different concentrations (2.5 and 3.5 mg/kg) severely decreases the replication of the attenuated vaccine strain in vaccinated pigs. This suggests that the effect of DON on the immune response generated by the MLV vaccine is more related to its impact on the replication of vaccinal virus in swine. DON has been shown previously to inhibit PRRSV replication in MARC-145 and porcine alveolar macrophages (PAM) cell models [38]. In that study, it was suggested that the early activation of pro-inflammatory genes and apoptosis following DON exposure was detrimental to PRRSV replication. In studies with concomitant viral infections, previous porcine respiratory coronavirus (PRCV) [39] and porcine circovirus type 2 (PCV2) [40], two potent inducer of endogenous IFN, have also been shown to decrease significantly PRRSV replication following experimental infection. Involvement of pro-inflammatory genes in the inhibition of PRRSV replication following DON-contaminated feed ingestion remains to be determined.
In conclusion, the present study showed for the first time an adverse effect of DON naturally contaminated feed on the immune response generated by a modified live vaccine. Live viral vaccines are among the most effective strategies for the induction of lifelong immunity and many of these vaccines are routinely used to provide protection against many human [41] and animal viral diseases [42]. It is difficult to predict if the present findings can be applied to other live viral vaccine, because of the small size of groups used in this study but also the impact of DON-contaminated feed might be virus-specific. In the particular case of PRRS vaccine, the immune response was blunted by an impairment of virus replication following ingestion of DON-contaminated feed. Further studies are needed to describe the exact mechanism by which DON-contaminated feed impairs the replication of PRRSV vaccinal strains.
Conflict of interest statement
All the authors, Christian Savard, Carl A. Gagnon and Younes Chorfi do not have any financial or personal relationships with other people or organizations that could inappropriately influence (bias) their work. They do not have any potential conflicts of interest including employment, consultancies, stock ownership, honoraria, paid expert testimony, patent applications/registrations, and grants or other funding.
Acknowledgements
The authors gratefully thank the Swine and Poultry Infectious Diseases Research Centre (FRQNT grant #170946), for funding this project. C Savard received postdoctoral fellowships from the Canadian Swine Health Board (CSHB) and from the Fonds de recherche du Québec—Nature et technologies (FRQNT). C.A. Gagnon was financially supported by a discovery grant (#312113-2010) of the Natural Sciences and Engineering Research Council of Canada (NSERC).
References
- 1.Kouadio J.H., Dano S.D., Moukha S., Mobio T.A., Creppy E.E. Effects of combinations of Fusarium mycotoxins on the inhibition of macromolecular synthesis, malondialdehyde levels, DNA methylation and fragmentation, and viability in Caco-2 cells. Toxicon. 2007;49:306–317. doi: 10.1016/j.toxicon.2006.09.029. [DOI] [PubMed] [Google Scholar]
- 2.Binder E.M., Tan L.M., Chin L.J., Handl J., Richard J. Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim Feed Sci Technol. 2007;137:265–282. [Google Scholar]
- 3.Glenn A.E. Mycotoxigenic Fusarium species in animal feed. Anim Feed Sci Technol. 2007;137:213–240. [Google Scholar]
- 4.Doll S., Danicke S. The Fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) in animal feeding. Prev Vet Med. 2011;102:132–145. doi: 10.1016/j.prevetmed.2011.04.008. [DOI] [PubMed] [Google Scholar]
- 5.Bryden W.L. Mycotoxin contamination of the feed supply chain: implications for animal productivity and feed security. Anim Feed Sci Technol. 2012;173:134–158. [Google Scholar]
- 6.Streit E., Schwab C., Sulyok M., Naehrer K., Krska R., Schatzmayr G. Multi-mycotoxin screening reveals the occurrence of 139 different secondary metabolites in feed and feed ingredients. Toxins (Basel) 2013;5:504–523. doi: 10.3390/toxins5030504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pestka J.J., Smolinski A.T., Deoxynivalenol: toxicology and potential effects on humans. J Toxicol Environ Health B Crit Rev. 2005;8:39–69. doi: 10.1080/10937400590889458. [DOI] [PubMed] [Google Scholar]
- 8.Zhou H.R., Islam Z., Pestka J.J. Rapid, sequential activation of mitogen-activated protein kinases and transcription factors precedes proinflammatory cytokine mRNA expression in spleens of mice exposed to the trichothecene vomitoxin. Toxicol Sci. 2003;72:130–142. doi: 10.1093/toxsci/kfg006. [DOI] [PubMed] [Google Scholar]
- 9.Pestka J.J. Deoxynivalenol-induced IgA production and IgA nephropathy-aberrant mucosal immune response with systemic repercussions. Toxicol Lett. 2003;140–141:287–295. doi: 10.1016/s0378-4274(03)00024-9. [DOI] [PubMed] [Google Scholar]
- 10.Lucioli J., Pinton P., Callu P., Laffitte J., Grosjean F., Kolf-Clauw M. The food contaminant deoxynivalenol activates the mitogen activated protein kinases in the intestine: interest of ex vivo models as an alternative to in vivo experiments. Toxicon. 2014;66:31–36. doi: 10.1016/j.toxicon.2013.01.024. [DOI] [PubMed] [Google Scholar]
- 11.Doll S., Schrickx J.A., Danicke S., Fink-Gremmels J. Deoxynivalenol-induced cytotoxicity, cytokines and related genes in unstimulated or lipopolysaccharide stimulated primary porcine macrophages. Toxicol Lett. 2009;184:97–106. doi: 10.1016/j.toxlet.2008.10.024. [DOI] [PubMed] [Google Scholar]
- 12.Pinton P., Accensi F., Beauchamp E., Cossalter A.M., Callu P., Grosjean F. Ingestion of deoxynivalenol (DON) contaminated feed alters the pig vaccinal immune responses. Toxicol Lett. 2008;177:215–222. doi: 10.1016/j.toxlet.2008.01.015. [DOI] [PubMed] [Google Scholar]
- 13.Grenier B., Loureiro-Bracarense A.P., Lucioli J., Pacheco G.D., Cossalter A.M., Moll W.D. Individual and combined effects of subclinical doses of deoxynivalenol and fumonisins in piglets. Mol Nutr Food Res. 2011;55:761–771. doi: 10.1002/mnfr.201000402. [DOI] [PubMed] [Google Scholar]
- 14.Savard C., Pinilla V., Provost C., Gagnon C.A., Chorfi Y. In vivo effect of deoxynivalenol (DON) naturally contaminated feed on porcine reproductive and respiratory syndrome virus (PRRSV) infection. Vet Microbiol. 2014;174:419–426. doi: 10.1016/j.vetmic.2014.10.019. [DOI] [PubMed] [Google Scholar]
- 15.Holtkamp D.J., Kliebenstein J.B., Neumann E.J., Zimmerman J.J., Rotto H.F., Yoder T.K. Assessment of the economic impact of porcine reproductive and respiratory syndrome virus on United States pork producers. J Swine Health Prod. 2013;21:72–84. [Google Scholar]
- 16.Cavanagh D. Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch Virol. 1997;142:629–633. [PubMed] [Google Scholar]
- 17.Dorr P.M., Gebreyes W.A., Almond G.W. Porcine reproductive and respiratory syndrome virus: age and management system disease modeling for pathogenic co-infection. J Swine Health Prod. 2007;15:258–263. [Google Scholar]
- 18.Chand R.J., Trible B.R., Rowland R.R.R. Pathogenesis of porcine reproductive and respiratory syndrome virus. Curr Opin Virol. 2012;2:256–263. doi: 10.1016/j.coviro.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 19.Corzo C.A., Mondaca E., Wayne S., Torremorell M., Dee S., Davies P. Control and elimination of porcine reproductive and respiratory syndrome virus. Virus Res. 2010;154:185–192. doi: 10.1016/j.virusres.2010.08.016. [DOI] [PubMed] [Google Scholar]
- 20.Kimman T.G., Cornelissen L.A., Moormann R.J., Rebel J.M.J., Stochofe-Zurwieden N. Challenges for porcine reproductive and respiratory syndrome virus (PRRSV) vaccinology. Vaccine. 2009;27:3704–3718. doi: 10.1016/j.vaccine.2009.04.022. [DOI] [PubMed] [Google Scholar]
- 21.Gagnon C.A., del Castillo J.R., Music N., Fontaine G., Harel J., Tremblay D. Development and use of a multiplex real-time quantitative polymerase chain reaction assay for detection and differentiation of Porcine circovirus-2 genotypes 2a and 2b in an epidemiological survey. J Vet Diagn Invest. 2008;20:545–558. doi: 10.1177/104063870802000503. [DOI] [PubMed] [Google Scholar]
- 22.Pieters M.N., Freijer J., Baars B.J., Fiolet D.C., van Klaveren J., Slob W. Risk assessment of deoxynivalenol in food: concentration limits, exposure and effects. Adv Exp Med Biol. 2002;504:235–248. doi: 10.1007/978-1-4615-0629-4_25. [DOI] [PubMed] [Google Scholar]
- 23.Rotter B.A., Thompson B.K., Lessard M., Trenholm H.L., Tryphonas H. Influence of low-level exposure to Fusarium mycotoxins on selected immunological and hematological parameters in young swine. Fundam Appl Toxicol. 1994;23:117–124. doi: 10.1006/faat.1994.1087. [DOI] [PubMed] [Google Scholar]
- 24.Prelusky D.B., Gerdes R.G., Underhill K.L., Rotter B.A., Jui P.Y., Trenholm H.L. Effects of low-level dietary deoxynivalenol on haematological and clinical parameters of the pig. Nat Toxins. 1994;2:97–104. doi: 10.1002/nt.2620020302. [DOI] [PubMed] [Google Scholar]
- 25.Chaytor A.C., See M.T., Hansen J.A., de Souza A.L., Middleton T.F., Kim S.W. Effects of chronic exposure of diets with reduced concentrations of aflatoxin and deoxynivalenol on growth and immune status of pigs. J Anim Sci. 2011;89:124–135. doi: 10.2527/jas.2010-3005. [DOI] [PubMed] [Google Scholar]
- 26.Opriessnig T., Pallares F.J., Nilubol D., Vincent A.L., Thacker E.L., Vaughn E.M. Genomic homology of ORF 5 gene sequence between modified live vaccine virus and porcine reproductive and respiratory syndrome virus challenge isolates is not predictive of vaccine efficacy. J Swine Health Prod. 2005;13:246–253. [Google Scholar]
- 27.Ellingson J.S., Wang Y., Layton S., Ciacci-Zanella J., Roof M.B., Faaberg K.S. Vaccine efficacy of porcine reproductive and respiratory syndrome virus chimeras. Vaccine. 2010;28:2679–2686. doi: 10.1016/j.vaccine.2009.12.073. [DOI] [PubMed] [Google Scholar]
- 28.Charerntantanakul W. Porcine reproductive and respiratory syndrome virus vaccines: immunogenicity, efficacy and safety aspects. World J Virol. 2012;1:23–30. doi: 10.5501/wjv.v1.i1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Darwich L., Diaz I., Mateu E. Certainties, doubts and hypotheses in porcine reproductive and respiratory syndrome virus immunobiology. Virus Res. 2010;154:123–132. doi: 10.1016/j.virusres.2010.07.017. [DOI] [PubMed] [Google Scholar]
- 30.Mateu E., Diaz I. The challenge of PRRS immunology. Vet J. 2008;177:345–351. doi: 10.1016/j.tvjl.2007.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Opriessnig T., McKeown N.E., Harmon K.L., Meng X.J., Halbur P.G. Porcine circovirus type 2 infection decreases the efficacy of a modified live porcine reproductive and respiratory syndrome virus vaccine. Clin Vaccine Immunol. 2006;13:923–929. doi: 10.1128/CVI.00074-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Martelli P., Cordioli P., Alborali L.G., Gozio S., De Angelis E., Ferrari L. Protection and immune response in pigs intradermally vaccinated against porcine reproductive and respiratory syndrome (PRRS) and subsequently exposed to a heterologous European (Italian cluster) field strain. Vaccine. 2007;25:3400–3408. doi: 10.1016/j.vaccine.2006.12.050. [DOI] [PubMed] [Google Scholar]
- 33.Zuckermann F.A., Garcia E.A., Luque I.D., Christopher-Hennings J., Doster A., Brito M. Assessment of the efficacy of commercial porcine reproductive and respiratory syndrome virus (PRRSV) vaccines based on measurement of serologic response, frequency of gamma-IFN-producing cells and virological parameters of protection upon challenge. Vet Microbiol. 2007;123:69–85. doi: 10.1016/j.vetmic.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 34.Labarque G., Van Gucht S., Van Reeth K., Nauwynck H., Pensaert M. Respiratory tract protection upon challenge of pigs vaccinated with attenuated porcine reproductive and respiratory syndrome virus vaccines. Vet Microbiol. 2003;95:187–197. doi: 10.1016/s0378-1135(03)00157-3. [DOI] [PubMed] [Google Scholar]
- 35.Choi B.K., Jeong S.H., Cho J.H., Shin H.S., Son S.W., Yeo Y.K. Effects of oral deoxynivalenol exposure on immune-related parameters in lymphoid organs and serum of mice vaccinated with porcine parvovirus vaccine. Mycotoxin Res. 2013;29:185–192. doi: 10.1007/s12550-013-0161-3. [DOI] [PubMed] [Google Scholar]
- 36.Chen F., Ma Y., Xue C., Ma J., Xie Q., Wang G. The combination of deoxynivalenol and zearalenone at permitted feed concentrations causes serious physiological effects in young pigs. J Vet Sci. 2008;9:39–44. doi: 10.4142/jvs.2008.9.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Murtaugh M.P., Genzow M. Immunological solutions for treatment and prevention of porcine reproductive and respiratory syndrome (PRRS) Vaccine. 2011;29:8192–8204. doi: 10.1016/j.vaccine.2011.09.013. [DOI] [PubMed] [Google Scholar]
- 38.Savard C., Pinilla V., Provost C., Segura M., Gagnon C.A., Chorfi Y. In vitro effect of deoxynivalenol (DON) mycotoxin on porcine reproductive and respiratory syndrome virus replication. Food Chem Toxicol. 2014;65:219–226. doi: 10.1016/j.fct.2013.12.043. [DOI] [PubMed] [Google Scholar]
- 39.Buddaert W., Van Reeth K., Pensaert M. In vivo and in vitro interferon (IFN) studies with the porcine reproductive and respiratory syndrome virus (PRRSV) Adv Exp Med Biol. 1998;440:461–467. doi: 10.1007/978-1-4615-5331-1_59. [DOI] [PubMed] [Google Scholar]
- 40.Chang H.W., Jeng C.R., Liu J.J., Lin T.L., Chang C.C., Chia M.Y. Reduction of porcine reproductive and respiratory syndrome virus (PRRSV) infection in swine alveolar macrophages by porcine circovirus 2 (PCV2)-induced interferon-alpha. Vet Microbiol. 2005;108:167–177. doi: 10.1016/j.vetmic.2005.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gans H.A. The status of live viral vaccination in early life. Vaccine. 2012;31:2531–2537. doi: 10.1016/j.vaccine.2012.09.043. [DOI] [PubMed] [Google Scholar]
- 42.Shams H. Recent developments in veterinary vaccinology. Vet J. 2005;170:289–299. doi: 10.1016/j.tvjl.2004.07.004. [DOI] [PubMed] [Google Scholar]


