Abstract
Our previous study demonstrated that chickens immunized subcutaneously with an Eimeria recombinant profilin protein vaccine emulsified in a Quil A/cholesterol/DDA/Carbopol (QCDC) adjuvant developed partial protection against experimental avian coccidiosis compared with animals immunized with profilin alone. Because in ovo vaccination is presently used in commercial applications worldwide throughout the poultry industry, the current study was undertaken to investigate chicken embryo vaccination with profilin plus QCDC adjuvant. Eighteen day-old embryos were immunized with isotonic saline (control), profilin alone, QCDC alone, or profilin plus QCDC, and orally challenged with live Eimeria maxima at 7 days post-hatch. Body weight gain, fecal oocyst output, and intestinal cytokine transcript levels were assessed as measures of protective immunity. While immunization with profilin alone or QCDC alone did not alter body weight gain of infected chickens compared with the saline control group, vaccination with profilin plus QCDC increased body weight gain such that it was equal to the uninfected controls. Immunization with profilin plus QCDC also reduced fecal oocyst shedding compared with unimmunized controls, although in this case QCDC failed to provide an adjuvant effect since no difference was observed between the profilin-only and profilin/QCDC groups. Finally, increased levels of transcripts encoding IL-1β, IL-15, and IFN-γ were seen in the intestinal tissues of animals given profilin plus QCDC compared with the profilin-only or QCDC-only groups. In summary, this study demonstrates an adjuvant effect of QCDC on body weight gain and intestinal cytokine responses following in ovo vaccination of chickens with an Eimeria profilin vaccine.
Keywords: Profilin, Coccidiosis, Immunity, Cytokine, Parasite
1. Introduction
Coccidiosis is an economically important disease in poultry that is caused by several species of Eimeria apicomplexan protozoa that colonize the intestinal mucosa. Infected animals exhibit a variety of clinical manifestations, including inefficient feed utilization, impaired body weight gain, and, in severe cases, mortality [1]. Prophylactic medication has been successfully used to control avian coccidiosis, but alternative strategies are sought due to the increasing emergence of drug-resistant parasites in commercial production settings [2], [3]. Although live parasite vaccines have been developed, they pose the risk of unintended infection under the immunosuppressive conditions associated with high-density commercial rearing conditions [4], [5]. As an alternative, noninfectious subunit protein and DNA vaccines have been generated. In particular, subcutaneous immunization of young broiler chickens with a recombinant profilin protein induced high levels of interferon (IFN)-γ production by splenic T cells [6]. Additionally, in ovo vaccination of 18-day-old embryos with DNA plasmids encoding the profilin gene provided partial protection against challenge infection with live Eimeria [7]. Profilin is a component of Eimeria parasites that activates Toll-like receptor 11, and is involved in actin-dependent gliding motility, parasite migration across biological barriers, and host cell invasion [8], [9], [10].
In spite of these encouraging results, immunization with recombinant proteins has, in general, shown limited success in stimulating broad-spectrum protective immunity against multiple coccidia species due to their low antigenicity and restricted expression during the parasite life cycle [11], [12]. Coadministration of avian recombinant cytokines as vaccine adjuvants along with Eimeria protein vaccines enhances their immunogenicity [13], [14], [15], [16]. However, cytokine adjuvants are not as effective as conventional veterinary adjuvants and identification of additional immunostimulators for use in commercial poultry vaccination is needed. QCDC is a new adjuvant complex composed of Quil A, cholesterol, dimethyl dioctadecyl ammonium bromide (DDA) and Carbopol that enhances immune responses against multiple types of vaccines in a variety of veterinary settings [17], [18]. Our prior study demonstrated that broiler chickens immunized subcutaneously with recombinant profilin in combination with QCDC developed partial protection against experimental avian coccidiosis compared with animals given profilin alone [18]. Because in ovo vaccination of chickens is currently used in commercial production facilities and offers the advantages of inducing early and heightened immunity compared with post-hatch immunization [19], [20], the present study was conducted to evaluate the adjuvant effect of QCDC during embryo vaccination with profilin against avian coccidiosis.
2. Materials and methods
2.1. Chickens and in ovo immunization
Embryonated eggs of inbred broiler chickens (Moyer's Chicks, Inc., Quakertown, PA) were incubated for 18 days and candled to select well-developed embryos. The eggs were injected with 100 μl containing sterile phosphate buffered saline (PBS, pH 7.4), QCDC alone, profilin alone (50 μg), or profilin (50 μg) plus QCDC using the Intelliject system (AviTech, Easton, MD) (Fig. 1 ). The final concentrations of QCDC components used alone or with profilin were 12.0 μg/ml of Quil A (E.M. Sergeant Pulp & Chemical Co., Clifton, NJ), 12.0 μg/ml of cholesterol (FabriChem, Trumbull, CT), 0.6 μg/ml of DDA (Fluka, Buchs, Switzerland), and 0.75 mg/ml of Carbopol® 974P (Lubrizol, Wickliffe, OH). In brief, each egg was cleaned and positioned in a holder under the injection needle with the large end on top. With the help of a vacuum system, the needle penetrated the shell past the air cell, delivered the inoculum into the amniotic cavity and was disinfected after each inoculation. In addition, the system was designed to avoid creating negative pressure inside of the egg, thus reducing the risk of cross-contamination [12]. At 3 days post-immunization, the chickens were hatched and provided with feed and water ad libitum. Chickens were kept in brooder pens in an Eimeria-free facility for 7 days post-hatch and transferred into large hanging cages (2 birds/cage) at a separate location where they were infected with Eimeria maxima and kept until the end of the experimental period. All experiments were performed according to guidelines established by the Beltsville Agriculture Research Center Small Animal Care Committee.
Fig. 1.
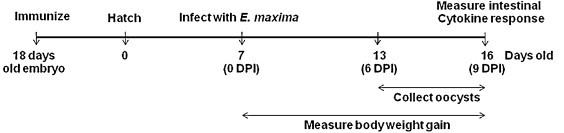
Schematic outline of the experimental design.
2.2. Expression and purification of recombinant profilin
The profilin gene was originally cloned by immunoscreening an E. acervulina cDNA library using a rabbit antiserum against E. acervulina merozoites [6]. The 1086-base pair profilin cDNA was subcloned into the pMAL plasmid (New England Biolabs, Ipswich, MA) with an NH2-terminal maltose-binding protein epitope tag and a Factor Xa protease cleavage site between maltose-binding protein and profilin. Transformed Escherichia coli DH5α bacteria were grown to mid-log phase, induced with 1.0 mM of isopropyl-β-d-thiogalactopyranoside for 3 h at 37 °C, collected by centrifugation and disrupted by sonication on ice (Misonix, Farmingdale, NY). The recombinant profilin protein was isolated on an amylose affinity column (New England Biolabs) according to the manufacturer's instructions, digested with Factor Xa to release profilin from the solid phase and repassed through the amylose column to remove contaminating maltose-binding protein.
2.3. Eimeria infection and measurements of body weight and fecal oocyst shedding
At 7 days post-hatch, chickens (20/group) were orally infected with 1.0 × 104 sporulated oocysts of E. maxima as described [21], [22]. Body weights were measured at 0 and 9 days post-infection. For determination of fecal oocyst shedding, birds (12/group) were placed in oocyst collection cages and fecal samples were collected between 6 and 9 days post-infection. Oocyst numbers were determined using a McMaster chamber according to the formula: total oocysts/bird = [oocyst count × dilution factor × (fecal sample volume/counting chamber volume)]/2 [23], [24].
2.4. Quantification of cytokine mRNA levels
Intestinal jejunum tissues were obtained from chickens (4/group) at 9 days post-infection, cut longitudinally, and washed 3 times with ice-cold Hanks’ balanced salt solution containing 100 U/ml of penicillin and 100 μg/ml of streptomycin (Sigma, St. Louis, MO). The mucosal layer was carefully removed using a surgical scalpel and total RNA was extracted using TRIzol (Invitrogen, Carlsbad, CA). Five micrograms of total RNA were treated with 1.0 U of DNase I and 1.0 μl of 10× reaction buffer (Sigma) and incubated for 15 min at room temperature. One μl of stop solution was added to inactivate DNase I and the mixture was heated at 70 °C for 10 min. RNA was reverse-transcribed using the StrataScript first-strand synthesis system (Stratagene, La Jolla, CA) according to the manufacturer's recommendations. Quantitative RT-PCR oligonucleotide primers for chicken interleukin (IL)-1β, IL-15, and IFN-γ and the GAPDH internal control are listed in Table 1 . Amplification and detection were carried out using equivalent amounts of total RNA using the Mx3000P system and Brilliant SYBR Green qPCR master mix (Stratagene). Standard curves were generated using log10 diluted standard RNA and the levels of individual transcripts were normalized to those of GAPDH by the Q-gene program [25]. Each sample was analysed in triplicate. To normalize individual replicates, the logarithmic-scaled threshold cycle (C t) values were transformed to linear units of normalized expression prior to calculating means and SEM for the references and individual targets, followed by the determination of mean normalized expression using the Q-gene program [18], [26], [27], [28].
Table 1.
Oligonucleotide primers used for quantitative RT-PCR.
| Type | mRNA Target | Primer sequences | PCR Product Size (bp) | Genbank accession no. |
|---|---|---|---|---|
| Reference | GAPDH | F: 5′-GGTGGTGCTAAGCGTGTTAT-3′ R: 5′-ACCTCTGTCATCTCTCCACA-3′ |
264 | K01458 |
| Pro-inflammatory | IL-1β | F: 5′-TGGGCATCAAGGGCTACA-3′ R: 5′-TCGGGTTGGTTGGTGATG-3′ |
244 | Y15006 |
| Th1 | IL-15 | F: 5′-TCTGTTCTTCTGTTCTGAGTGATG-3′ R: 5′-AGTGATTTGCTTCTGTCTTTGGTA-3′ |
243 | AF139097 |
| IFN-γ | F: 5′-AGCTGACGGTGGACCTATTATT-3′ R: 5′-GGCTTTGCGCTGGATTC-3′ |
259 | Y07922 |
2.5. Statistical analyses
All data was expressed as mean ± SEM values of 4–12 chickens/group with triplicates/sample. Comparisons of the mean values were performed by one-way analysis of variance followed by the Tukey's HSD test using SPSS software (SPSS 15.0 for Windows, Chicago, IL). Differences between groups were considered statistically significant at P < 0.05.
3. Results
3.1. Effect of vaccination with profilin plus QCDC on body weight gain
Animals that had been vaccinated with profilin alone or with QCDC alone and infected with E. maxima displayed reduced body weight gains between 0 and 9 days post-infection compared with uninfected controls (P < 0.05) (Fig. 2 ). In fact, the weight gains of the profilin-only (461.2 ± 13.4 g) and QCDC-only (457.6 ± 15.9 g) groups were identical to the PBS control group (410.4 ± 12.5 g). In contrast, animals immunized with profilin plus QCDC had significantly greater body weight gains (486.1 ± 13.9 g) compared with the PBS controls.
Fig. 2.
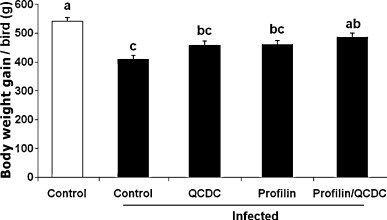
Effects of vaccination with profilin plus QCDC on body weight gain. Eighteen day-old embryos were immunized with PBS (control), QCDC alone (QCDC), profilin alone (Profilin), or profilin plus QCDC (Profilin/QCDC). Chickens were uninfected or orally infected with 1.0 × 104 sporulated oocysts of E. maxima at 7 days post-hatch, and body weight gains between 0 and 9 days post-infection were calculated. Each bar represents the mean ± SEM (n = 12). Bars not sharing the same letters are significantly different according to the Tukey's HSD test (P < 0.05).
3.2. Effect of vaccination with profilin plus QCDC on fecal oocyst shedding
Fecal oocyst shedding was decreased in the profilin alone (1.46 ± 0.08 × 107) and profilin plus QCDC (1.22 ± 0.04 × 107) groups compared with the PBS control group (2.57 ± 0.24 × 107) and with the QCDC-only group (2.33 ± 0.13 × 107) (P < 0.05) (Fig. 3 ). However, no difference in oocyst shedding was observed between the profilin alone and profilin plus QCDC groups.
Fig. 3.
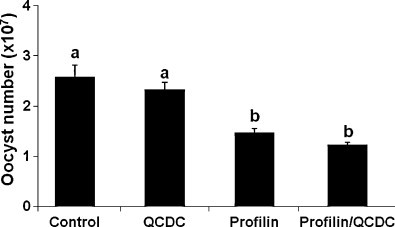
Effects of vaccination with profilin plus QCDC on fecal oocyst shedding. Eighteen day-old embryos were immunized with PBS (control), QCDC alone (QCDC), profilin alone (Profilin), or profilin plus QCDC (Profilin/QCDC). Chickens were uninfected or orally infected with 1.0 × 104 sporulated oocysts of E. maxima at 7 days post-hatch, and fecal oocyst numbers were determined between days 6 and 9 post-infection. Each bar represents the mean ± SEM (n = 12). Bars not sharing the same letters are significantly different according to the Tukey's HSD test (P < 0.05).
3.3. Effect of vaccination with profilin plus QCDC on intestinal cytokine transcript levels
The levels of mRNA transcripts encoding IL-1β, IL-15 and IFN-γ in the intestinal jejunum were increased in the profilin plus QCDC group compared with the PBS control, QCDC alone, and profilin alone groups (P < 0.05) (Fig. 4 ).
Fig. 4.
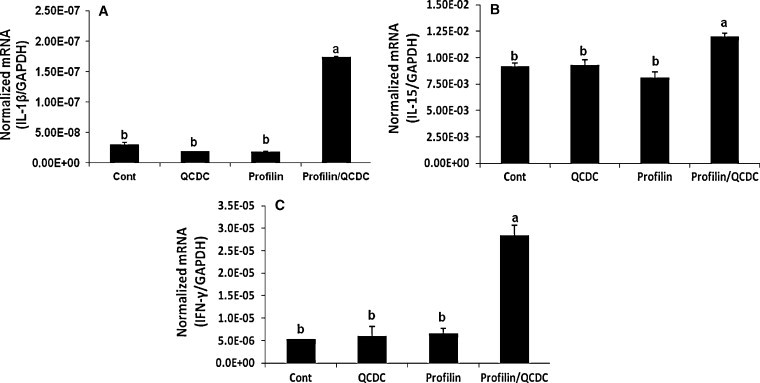
Effect of vaccination with profilin plus QCDC on intestinal cytokine transcript levels. Eighteen day-old embryos were immunized with PBS (control), QCDC alone (QCDC), profilin alone (Profilin), or profilin plus QCDC (Profilin/QCDC). The levels of transcripts encoding IL-1β (A), IL-15 (B), and IFN-γ (C) were quantified by RT-PCR and normalized to GAPDH transcript levels at 9 days post-infection. Each bar represents the mean ± SEM from triplicate samples/bird (n = 4). Bars not sharing the same letters are significantly different according to the Tukey's HSD test (P < 0.05).
4. Discussion
Novel vaccination strategies using recombinant proteins from mucosal pathogens have successfully induced protective immunity against a variety of infectious diseases, including avian coccidiosis [6], [17], [18], [22]. In our previous in vivo study, chickens immunized post-hatch with profilin plus QCDC showed increased body weight gain following E. acervulina infection compared with animals given PBS or profilin alone [18]. However, subcutaneous immunization with profilin alone or with profilin plus QCDC did not affect fecal oocyst output compared with PBS controls, and the levels of intestinal mRNAs for IL-1β, IL-15, and IFN-γ were equal in the profilin alone and profilin/QCDC groups. In the current study, we sought to determine whether in ovo immunization with profilin plus QCDC would provide beneficial effects on post-infection weight gain, oocyst shedding, and cytokine expression, as well as to extend the analysis to E. maxima, another parasite species common to commercial production facilities. Our results demonstrated that vaccination with profilin plus QCDC increased chicken body weight gain compared with the profilin- and QCDC-only groups, such that it was equal to the uninfected controls. Immunization with profilin plus QCDC also reduced oocyst output compared with unimmunized controls, although in this case QCDC failed to provide an adjuvant effect since no difference was observed between the profilin-only and profilin/QCDC groups. Finally, animals given profilin plus QCDC had increased intestinal levels of transcripts encoding IL-1β, IL-15 and IFN-γ compared with the profilin-only or with the QCDC-only groups. The reason why profilin plus QCDC had no significant effect on parasite shedding compared with profilin alone is unclear, but this observation is in agreement with published reports indicating the absence of a correlation between Eimeria-induced body weight loss and fecal oocyst shedding [26], [29].
The adjuvant effect of QCDC has been demonstrated using veterinary vaccines against E. coli, the feline leukemia virus, bovine viral diarrhea virus, Mycoplasma hyopneumoniae, avian and canine influenza viruses, canine coronavirus, and bovine rotavirus [17]. In humans, Quil A-like saponin adjuvants, such as QS-21, are currently being used in clinical trials [30], [31]. DDA stimulates predominantly Th1 cells when co-administered systemically or locally with antigen, and has no reported toxic effects in humans [32], [33], [34], [35]. Chickens immunized with Eimeria merozoite antigens in combination with DDA displayed longer lasting immunity compared with a Corynebacterium parvum adjuvant [36]. Carbopol, a mucoadhesive acrylic polymer, has been extensively investigated due to its high viscosity at low concentrations and low toxicity [37]. Carbopol has been used to enhance the efficacy of recombinant canarypox viral vectors for equine herpes virus vaccination [38], but no prior studies have examined the effects of Carbopol on poultry vaccines.
The positive effect of immunization with profilin plus QCDC on body weight gain suggests that the mode of action of this vaccine/adjuvant complex may influence intestinal physiology, increase nutrient absorption, and/or prevent Eimeria cytotoxicity [18], [28], [39]. Furthermore, the increased levels of proinflammatory and Th1 cytokines in the profilin/QCDC group imply that local immune defense mechanisms are also activated under these experimental conditions. In this regard, the importance of cell-mediated immunity in conferring protection against avian coccidiosis is well documented [18], [24], [28], [39] and other studies have verified a beneficial role of cell-mediated immunity for gut proinflammatory, Th1 and Th2 cytokines following Eimeria infection [39], [40], [41], [42]. One of the mechanisms by which adjuvants stimulate immunity is through increased cytokine expression, and several adjuvants have been shown to increase the levels of cytokine gene transcripts, including those examined in this study [18,43].
In summary, our results demonstrate an adjuvant effect of QCDC on body weight gain and intestinal cytokine responses following in ovo vaccination of chickens with the Eimeria profilin vaccine. Future studies to elucidate the molecular and cellular immune mechanisms mediated by QCDC in various clinical conditions will be of benefit to the commercial poultry industry.
Acknowledgments
This project was partially supported by ARS CRIS project 1265-32000-086 and a formal agreement established between ARS and Pfizer Animal Health and the World Class University Program (R33-10013) of the Ministry of Education, Science and Technology of South Korea. The authors thank Margie Nichols and Stacy Torreyson for their significant contribution to this research.
Footnotes
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
References
- 1.Lillehoj H.S., Lillehoj E.P. Avian coccidiosis. A review of acquired intestinal immunity and vaccination strategies. Avian Dis. 2000;44:408–425. [PubMed] [Google Scholar]
- 2.Williams R.B. Anticoccidial vaccines for broiler chickens: pathways to success. Avian Pathol. 2002;31:317–353. doi: 10.1080/03079450220148988. [DOI] [PubMed] [Google Scholar]
- 3.Pogonka T., Klotz C., Kovacs F., Lucius R. A single dose of recombinant Salmonella typhimurium induces specific humoral immune responses against heterologous Eimeria tenella antigens in chicken. Int J Parasitol. 2003;33:81–88. doi: 10.1016/s0020-7519(02)00251-5. [DOI] [PubMed] [Google Scholar]
- 4.Williams R.B., Carlyle W.W., Bond D.R., Brown I.A. The efficacy and economic benefits of Paracox, a live attenuated anticoccidial vaccine, in commercial trials with standard broiler chickens in the United Kingdom. Int J Parasitol. 1999;29:341–355. doi: 10.1016/s0020-7519(98)00212-4. [DOI] [PubMed] [Google Scholar]
- 5.Williams R.B., Catchpole J. A new protocol for a challenge test to assess the efficacy of live anticoccidial vaccines for chickens. Vaccine. 2000;18:1178–1185. doi: 10.1016/s0264-410x(99)00387-4. [DOI] [PubMed] [Google Scholar]
- 6.Lillehoj H.S., Choi K.D., Jenkins M.C., Vakharia V.N., Song K.D., Han J.Y. A recombinant Eimeria protein inducing interferon-gamma production: comparison of different gene expression systems and immunization strategies for vaccination against coccidiosis. Avian Dis. 2000;44:379–389. [PubMed] [Google Scholar]
- 7.Song K.D., Lillehoj H.S., Choi K.D., Yun C.H., Parcells M.S., Huynh J.T. A DNA vaccine encoding a conserved Eimeria protein induces protective immunity against live Eimeria acervulina challenge. Vaccine. 2000;19:243–252. doi: 10.1016/s0264-410x(00)00169-9. [DOI] [PubMed] [Google Scholar]
- 8.Yarovinsky F., Zhang D., Andersen J.F., Bannenberg G.L., Serhan C.N., Hayden M.S. TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science. 2005;308:1626–1629. doi: 10.1126/science.1109893. [DOI] [PubMed] [Google Scholar]
- 9.Plattner F., Yarovinsky F., Romero S., Didry D., Carlier M.F., Sher A. Toxoplasma profilin is essential for host cell invasion and TLR11-dependent induction of an interleukin-12 response. Cell Host Microbe. 2008;3:61–63. doi: 10.1016/j.chom.2008.01.001. [DOI] [PubMed] [Google Scholar]
- 10.Hedhli D., Dimier-Poisson I., Judge J.W., Rosenberg B., Mévélec M.N. Protective immunity against Toxoplasma challenge in mice by coadministration of T. gondii antigens and Eimeria profilin-like protein as an adjuvant. Vaccine. 2009;27:2274–2281. doi: 10.1016/j.vaccine.2009.01.100. [DOI] [PubMed] [Google Scholar]
- 11.Jang S.I., Lillehoj H.S., Lee S.H., Lee K.W., Park M.S., Bauchan G.R. Immunoenhancing effects of Montanide™ ISA oil-based adjuvants on recombinant coccidia antigen vaccination against Eimeria acervulina infection. Vet Parasitol. 2010;172:221–228. doi: 10.1016/j.vetpar.2010.04.042. [DOI] [PubMed] [Google Scholar]
- 12.Ding X., Lillehoj H.S., Quiroz M.A., Bevensee E., Lillehoj E.P. Protective immunity against Eimeria acervulina following in ovo immunization with a recombinant subunit vaccine and cytokine genes. Infect Immun. 2004;72:6939–6944. doi: 10.1128/IAI.72.12.6939-6944.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Min W., Lillehoj H.S., Burnside J., Weining K.C., Staeheli P., Zhu J.J. Adjuvant effects of IL-1β, IL-2, IL-8, IL-15, IFN-α, IFN-γ, TGF-β4 and lymphotactin on DNA vaccination against Eimeria acervulina. Vaccine. 2001;20:267–274. doi: 10.1016/s0264-410x(01)00270-5. [DOI] [PubMed] [Google Scholar]
- 14.Lillehoj H.S., Ding X., Dalloul R.A., Sato T., Yasuda A., Lillehoj E.P. Embryo vaccination against Eimeria tenella and E. acervulina infections using recombinant proteins and cytokine adjuvants. J Parasitol. 2005;91:666–673. doi: 10.1645/GE-3476. [DOI] [PubMed] [Google Scholar]
- 15.Lillehoj H.S., Ding X., Marco A.Q., Bevensee E., Lillehoj E.P. Resistance to intestinal coccidiosis following DNA immunization with the cloned 3-1E Eimeria gene plus IL-2, IL-15, and IFN-γ. Avian Dis. 2005;49:112–117. doi: 10.1637/7249-073004R. [DOI] [PubMed] [Google Scholar]
- 16.Miyamoto T., Min W., Lillehoj H.S. Kinetics of interleukin-2 production in chickens infected with Eimeria tenella. Comp Immunol Microbiol. 2002;25:149–158. doi: 10.1016/s0147-9571(01)00034-0. [DOI] [PubMed] [Google Scholar]
- 17.Dominowski PJ, Mannan RM, Krebs RL,Thompson JR, Childers TA, Olsen MK, et al. Novel adjuvant compositions. U.S. patent application number 20090324641 (http://www.freepatentsonline.com/y2009/0324641.html).
- 18.Lee S.H., Lillehoj H.S., Jang S.I., Lee K.W., Yancey R.J., Dominowski P. Effects of novel adjuvant complex/Eimeria profilin vaccine on intestinal host immune responses against live E. acervulina challenge infection. Vaccine. 2010;28:6498–6504. doi: 10.1016/j.vaccine.2010.06.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ding X., Lillehoj H.S., Dalloul R.A., Min W., Sato T., Yasuda A. In ovo vaccination with the Eimeria tenella EtMIC2 gene induces protective immunity against coccidiosis. Vaccine. 2005;23:3733–3740. doi: 10.1016/j.vaccine.2005.01.144. [DOI] [PubMed] [Google Scholar]
- 20.Sharma J.M. Embryo vaccination of chickens with turkey herpesvirus: characteristics of the target cell of early viral replication in embryonic lung. Avian Pathol. 1987;16:367–379. doi: 10.1080/03079458708436407. [DOI] [PubMed] [Google Scholar]
- 21.Lee S.H., Lillehoj H.S., Park D.W., Jang S.I., Morales A., García D. Protective effect of hyperimmune egg yolk IgY antibodies against Eimeria tenella and Eimeria maxima infections. Vet Parasitol. 2009;163:123–126. doi: 10.1016/j.vetpar.2009.04.020. [DOI] [PubMed] [Google Scholar]
- 22.Jang S.I., Lillehoj H.S., Lee S.H., Lee K.W., Park M.S., Cha S.R. Eimeria maxima recombinant Gam82 gametocyte antigen vaccine protects against coccidiosis and augments humoral and cell-mediated immunity. Vaccine. 2010;28:2980–2985. doi: 10.1016/j.vaccine.2010.02.011. [DOI] [PubMed] [Google Scholar]
- 23.Lee S.H., Lillehoj H.S., Park D.W., Hong Y.H., Lin J.J. Effects of Pediococcus- and Saccharomyces-based probiotic (MitoMax®) on coccidiosis in broiler chickens. Comp Immunol Microbiol. 2007;30:261–268. doi: 10.1016/j.cimid.2007.02.002. [DOI] [PubMed] [Google Scholar]
- 24.Lee S.H., Lillehoj H.S., Dalloul R.A., Park D.W., Hong Y.H., Lin J.J. Influence of Pediococcus-based probiotic on coccidiosis in broiler chickens. Poult Sci. 2007;86:63–66. doi: 10.1093/ps/86.1.63. [DOI] [PubMed] [Google Scholar]
- 25.Muller P.Y., Janovjak H., Miserez A.R., Dobbie Z. Processing of gene expression data generated by quantitative real-time RT-PCR. Biotechniques. 2002;32:1372–1379. [PubMed] [Google Scholar]
- 26.Lee S.H., Lillehoj H.S., Cho S.M., Park D.W., Hong Y.H., Chun H.K. Immunomodulatory properties of dietary plum on coccidiosis. Comp Immunol Microbiol. 2008;31:389–402. doi: 10.1016/j.cimid.2007.06.005. [DOI] [PubMed] [Google Scholar]
- 27.Lee S.H., Lillehoj H.S., Hong Y.H., Jang S.I., Lillehoj E.P., Ionescu C. In vitro effects of plant and mushroom extracts on immunological function of chicken lymphocytes and macrophages. Br Poult Sci. 2010;51:213–221. doi: 10.1080/00071661003745844. [DOI] [PubMed] [Google Scholar]
- 28.Lee S.H., Lillehoj H.S., Jang S.I., Kim D.K., Ionescu C., Bravo D. Effect of dietary curcuma, capsicum, and lentinus on enhancing local immunity against Eimeria acervulina infection. J Poult Sci. 2010;47:89–95. [Google Scholar]
- 29.Lillehoj H.S., Okamura M. Host immunity and vaccine development to coccidia and Salmonella infections in chickens. J Poult Sci. 2003;40:151–193. [Google Scholar]
- 30.Waite D.C., Jacobson E.W., Ennis F.A., Edelman R., White B., Kammer R. Three double-blind randomized trials evaluating the safety and tolerance of different formulations of the saponin adjuvant QS-21. Vaccine. 2001;19:3957–3967. doi: 10.1016/s0264-410x(01)00142-6. [DOI] [PubMed] [Google Scholar]
- 31.Gilewski T., Adluri S., Ragupathi G., Zhang S., Yao T.J., Panageas K. Vaccination of high-risk breast cancer patients with mucin-1 (MUC1) keyhole limpet hemocyanin conjugate plus QS-21. Clin Cancer Res. 2001;6:1693–1701. [PubMed] [Google Scholar]
- 32.Hilgers L.A., Snippe H. DDA as an immunological adjuvant. Res Immunol. 1992;143:494–503. doi: 10.1016/0923-2494(92)80060-x. [DOI] [PubMed] [Google Scholar]
- 33.Lindblad E.B., Elhay M.J., Silva R., Appelberg R., Andersen P. Adjuvant modulation of immune responses to tuberculosis subunit vaccines. Infect Immun. 1997;65:623–629. doi: 10.1128/iai.65.2.623-629.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Veronesi R., Correa A., Alterio D. Single dose immunisation against tetanus promising results in human trials. Rev Inst Med Trop. 1970;12:46–54. [PubMed] [Google Scholar]
- 35.Chambers J.D., Thomas C.R., Hobbs J.R. Induction of specific transplantation tolerance in man by autoblast immunisation. Blut. 1980;41:229–236. [Google Scholar]
- 36.Lillehoj H.S., Lindblad E.B., Nichols M. Adjuvanticity of dimethyl diooctadecyl ammonium bromide, complete Freund's adjuvant and Corynebacterium parvum with respect to host immune response to coccidial antigens. Avian Dis. 1993;37:731–740. [PubMed] [Google Scholar]
- 37.Minke J.M., Audonnet J.C., Jessett D.M., Fischer L., Guigal P.M., Coupier H. Canarypox as a vector for influenza and EHV-1 genes: challenges and rewards. Proceedings of the 2nd international veterinary vaccines and diagnostic conference; Oxford; 2000. p. 36. [Google Scholar]
- 38.Minke J.M., Fischer L., Baudu P., Guigal P.M., Sindle T., Mumford J.A. Use of DNA and recombinant canarypox viral (ALVAC) vectors for equine herpes virus vaccination. Vet Immunol Immunopathol. 2006;111:47–57. doi: 10.1016/j.vetimm.2006.01.008. [DOI] [PubMed] [Google Scholar]
- 39.Lee S.H., Lillehoj H.S., Cho S.M., Park D.W., Hong Y.H., Lillehoj E.P. Protective effects of dietary safflower (Carthamus tinctorius) on experimental coccidiosis. J Poult Sci. 2008;46:155–162. [Google Scholar]
- 40.Hong Y.H., Lillehoj H.S., Lee S.H., Dalloul R.A., Lillehoj E.P. Analysis of chicken cytokine and chemokine gene expression following Eimeria acervulina and Eimeria tenella infections. Vet Immunol Immunopathol. 2006;114:209–223. doi: 10.1016/j.vetimm.2006.07.007. [DOI] [PubMed] [Google Scholar]
- 41.Lillehoj H.S., Min W., Choi K.D., Babu U.S., Burnside J., Miyamoto T. Molecular, cellular, and functional characterization of chicken cytokines homologous to mammalian IL-15 and IL-2. Vet Immunol Immunopathol. 2001;82:229–244. doi: 10.1016/s0165-2427(01)00360-9. [DOI] [PubMed] [Google Scholar]
- 42.Lillehoj H.S., Kim C.H., Keeler C.L., Zhang S. Immunogenomic approaches to study host immunity to enteric pathogens. Poult Sci. 2007;86:1491–1500. doi: 10.1093/ps/86.7.1491. [DOI] [PubMed] [Google Scholar]


