Graphical abstract
Keyword: Isatis tinctoria L., Hairy root cultures, Flavonoids, Ultraviolet radiation, Oxidative stress, Biosynthetic gene expression
Highlights
-
•
Ultraviolet radiation was applied for flavonoid augmentation in hairy root cultures.
-
•
Ultraviolet-B treatment could increase flavonoid yield by 16.51-fold over control.
-
•
Ultraviolet-B treatment could enhance antioxidant activity and reinforce cell wall.
-
•
Oxidative stress might contribute to the activation of flavonoid biosynthetic genes.
-
•
Chalcone synthase gene is quite sensitive to ultraviolet-B radiation.
Abstract
Search of cost-effective strategies that can enhance the accumulation of phytochemicals of pharmaceutical interest in plant in vitro cultures is an essential task. For the first time, Isatis tinctoria L. hairy root cultures were exposed to ultraviolet radiation (ultraviolet-A, ultraviolet-B, and ultraviolet-C) in an attempt to promote the production of pharmacologically active flavonoids. Results showed that the maximum flavonoid accumulation (7259.12 ± 198.19 μg/g DW) in I. tinctoria hairy root cultures treated by 108 kJ/m2 dose of UV-B radiation increased 16.51-fold as compared with that in control (439.68 ± 8.27 μg/g DW). Additionally, antioxidant activity enhancement and cell wall reinforcement were found in the treated I. tinctoria hairy root cultures, indicating the positive-feedback responses to oxidative stress mediated by ultraviolet-B radiation. Moreover, the expression of chalcone synthase gene was tremendously up-regulated (up to 405.84-fold) in I. tinctoria hairy root cultures following ultraviolet-B radiation, which suggested chalcone synthase gene might play a crucial role in flavonoid augmentation. Overall, the present work provides a feasible approach for the enhanced production of biologically active flavonoids in I. tinctoria hairy root cultures via the simple supplementation of ultraviolet-B radiation, which is useful for the biotechnological production of these high-added value compounds to fulfil the ever-increasing demand in pharmaceutical fields.
1. Introduction
Isatis tinctoria L. (Brassicaceae family), an economically important crop, is widely distributed in Europe and Eastern Asia (Hamburger, 2002). The dried roots of I. tinctoria (Radix isatidis) have received great attention for the treatment of severe acute respiratory syndrome (SARS) and novel swine-origin influenza A (H1N1) (Lin et al., 2005; Wang et al., 2011). The notable pharmacological activity of I. tinctoria is mainly related to phenylpropanoids present in the extracts, particularly flavonoids and lignans (Nguyen et al., 2017). Nevertheless, the phytochemical profile in the field cultivation of I. tinctoria is highly affected by environmental, geographic, and climatic variations, which has resulted in the inconsistent treatment efficacy of this herbal medicine (Chen et al., 2015).
Currently, plant in vitro culture technology has been emerged an attractive alternative to the field cultivation of medicinal crops, which allows for the sustainable and standard production of valuable phytochemicals without occupation of agricultural lands (Dias et al., 2016). Among various types of plant in vitro cultures, hairy root cultures belonging to differentiated organs are preferred over plant cells, callus, and suspension cultures, due to genetic/biochemical stability, high growth rate independent of phytohormone, and biosynthetic capacity comparable to the parent plant (Rimando and Duke, 2013). In this context, I. tinctoria hairy root cultures (ITHRCs) have been established as a reliable biological platform for the efficient production of bioactive flavonoids (Gai et al., 2015).
It is well known that plant in vitro cultures are grown in an aseptic space with appropriate conditions of light, humidity, and temperature that are lack of environmental stresses, thus always leading to the accumulation of defense phytochemicals in low levels (Narayani and Srivastava, 2017). Generally, flavonoids are thought to be phytoanticipins that can be inducibly synthesized by biotic and abiotic stresses, which can improve plant survival under unfavorable environmental conditions (Fini et al., 2011; Giampieri et al., 2018). Based on this principle, application of external elicitors is likely to boost flavonoid biosynthesis in ITHRCs by inducing plant defense responses. In view of the bio-safety of products, atoxic elicitors should be adopted for the improvement of flavonoid yield in ITHRCs.
Notably, ultraviolet (UV) radiation is a physical elicitor that does not introduce any taints in plant in vitro culture system (Schreiner et al., 2016). Moreover, UV radiation has been acknowledged as an effective elicitor that can boost up the accumulation of various secondary metabolites such as flavonoids, phenolics, alkaloids, carotenoids, glucosinolates, and terpenoids in fruits, vegetables, and herbs (Matsuura et al., 2013; Schreiner et al., 2012), which is beneficial for the high consumption of these health-beneficial phytochemicals in nutritional and pharmaceutical industries. In view of this, it is recommendable to use UV radiation as a “clean” elicitor for promoting flavonoid production in ITHRCs.
In the present study, the elicitation effects of UV-A, UV-B, and UV-C with successive radiation doses on flavonoid accumulation in ITHRCs were initially compared. Subsequently, the profiles of eight flavonoids before and after elicitation by the selected UV-B with the optimal radiation dose were evaluated. Afterwards, antioxidant activity, cell wall modification, and biosynthetic gene expression were systematically investigated, which aimed to understanding the response mechanism of ITHRCs underlying UV-B elicitation. It is worth mentioning that there are no reports on the application of UV radiation to elevate the production of pharmacologically active flavonoids in ITHRCs.
2. Experimental
2.1. Hairy root cultures
I. tinctoria hairy roots were obtained by Agrobacterium rhizogenes LBA9402 mediated transformation of petiole explants in the Key Laboratory of Forest Plant Ecology, Ministry of Education, Northeast Forestry University, and all experiments in this study were conducted using an I. tinctoria hairy root line V (ITHRLV) due to the highest flavonoid productivity (Gai et al., 2015). The stock culture of ITHRL V was maintained on phytohormone-free MS/2-based solid medium supplemented with 30 g/L sucrose at 25 °C in the dark. According to the previous report (Gai et al., 2015), ITHRCs were initiated by culturing 1.125 g ITHRL V (fresh weight, FW) into 250 mL Erlenmeyer flask containing 150 mL of MS/2 liquid medium supplemented with 30.6 g/L sucrose, and incubated on a rotary shaker (120 rpm) at 24.71 °C in the dark.
2.2. ITHRCs treated by UV radiation
In order to enhance flavonoid production without affecting biomass yield of hairy roots, ITHRCs cultured under the aforementioned conditions for 24 days (the optimal duration for harvest) were adopted for UV radiation treatments. Prior to elicitation experiments, a series of flasks containing ITHRCs (24 day-old) with 150 mL of fresh culture media were placed in a laminar flow cabinet, and the sterile membranes of these flasks were taken off for exposing hairy root cultures to UV radiation. The UV light lamps (UV-A: 40 W, λ max = 365 nm; UV-B: 40 W, λ max = 313 nm; UV-C: 40 W, λ max = 254 nm; Beijing Institute of Electric Light Source, China) were mounted on fixtures at the top of hairy root cultures, and the distance of lamps from cultures was adjusted to obtain a constant radiation intensity (3 W/m2) measured with a DRC-100X photometer (Spectronics, USA). The UV radiation dose (kJ/m2) in this work was calculated by multiplying the fixed output value (3 W/m2) with the exposure duration. During elicitation experiments, three groups of ITHRCs (24 day-old) were individually subjected to UV-A, UV-B, and UV-C with successive time points of 0, 1, 2, 4, 6, 7, 8, 9, 10, 11, 13, and 15 h, which were equivalent to UV radiation doses of 0, 10.8, 21.6, 43.2, 64.8, 86.4, 97.2, 108, 118.8, 140.4, and 162 kJ/m2, respectively. For control, a group of ITHRCs (24 day-old) underwent the same conditions in the dark. After elicitation experiments, hairy roots were collected, rinsed by distilled water, and divided into three parts: one being dried in a vacuum oven for the liquid-solid extraction of flavonoids, one being handled quickly in fresh state for the evaluation of antioxidant enzyme activity, and one being frozen immediately with liquid nitrogen and stored at −80 °C for the extraction of total RNA. Also, the culture media were collected for the liquid-liquid extraction of flavonoids.
2.3. Flavonoid extraction and liquid chromatographic tandem mass spectrometry (LC–MS/MS) analysis
The dried hairy root samples were ground into fine powders using a mortar and pestle. The complete extraction of flavonoids from the resulting powders was carried out according to the method previously described (Gai et al., 2015). The flavonoids in culture media were extracted twice by phase partitioning with ethyl acetate, and the organic phase was collected and condensed to dryness using a rotary evaporator under vacuum. All extracts from roots and media were re-dissolved in acetonitrile (20 mL) and filtered through a nylon filter (0.45 μm) prior to LC–MS/MS analysis.
According to the previous report (Gai et al., 2015), the identification and quantification of eight target flavonoid derivatives was conducted by a LC–MS/MS method with the precursor–product ion combinations of m/z 609.1 → 300.0 (rutin), m/z 609.5 → 301.4 (neohesperidin), m/z 591.4 → 283.1 (buddleoside), m/z 255.9 → 119.0 (liquiritigenin), m/z 301.0 → 151.0 (quercetin), m/z 315.0 → 300.1 (isorhamnetin), m/z 285.3 → 183.1 (kaempferol), and m/z 255.4 → 118.9 (isoliquiritigenin). The content of each analyte was calculated by the corresponding calibration curve, and expressed as microgram per gram of the dry weight (DW) of root samples.
2.4. Antioxidant activity determination
Activities of four typical antioxidant enzymes i.e. superoxide dismutases (SOD), catalase (CAT), ascorbate peroxidase (APX), and glutathione reductase (GR) in fresh hairy root samples were measured according to the method described by Arbona et al. (2003). Enzymatic activity was expressed as unit per mg of protein that was detected in enzyme extracts. Non-enzymatic antioxidant activities of extracts from ITHRCs were determined using DPPH radical-scavenging assay and β-carotene/linoleic acid bleaching test reported by Yao et al. (2013). Antioxidant activity of non-enzymatic sample was expressed as the IC50 value that was the concentration of extracts required to scavenge 50% of DPPH radicals or inhibit 50% of β-carotene bleaching ratio.
2.5. Fourier transform infrared (FTIR) analysis
Prior to FTIR analysis, the dry hairy root samples (1%) together with KBr (spectroscopic grade) were ground into fine powders using a mortar and pestle, and pressed into transparent discs. The measurement of FTIR spectra in the wavelength range of 4000–400 cm−1 was conducted on an Affinity-1 spectrophotometer (Shimadzu, Japan). The data were corrected via the elimination of KBr background, and recorded as plots of transmittance (%) versus wavelength (cm−1).
2.6. Scanning electron microscopy (SEM) observation
The hairy root samples were initially processed to dryness tissues using the method described by Marsh et al. (2014). Prior to SEM observation, the dry root samples were mounted on an aluminium specimen holder with double stick tape, and evenly sputtered with a thin layer of gold. The micrograph examination of samples was conducted on a Quanta-200 environmental SEM system (FEI Company, USA), and photographed under high vacuum at an accelerating voltage of 12.5 kV.
2.7. Quantitative real-time PCR (qRT-PCR) analysis
Total RNA was extracted from frozen hairy root samples using a MiniBEST Plant RNA Extraction Kit (TaKaRa, Dalian, China), and RNA was reverse-transcribed to cDNA using a PrimeScript™ RT reagent Kit (TaKaRa, Dalian, China). Specific primers of the associated genes involved in flavonoid biosynthetic pathway (Table 1 ) were designed based on the reported transcriptome sequences (Chen et al., 2013; Tang et al., 2014). The reaction solution for qRT-PCR assay was prepared with a SYBR Premix Ex Taq™ II Kit (TaKaRa, Dalian, China) following the manufacturer’s guidelines. The qRT-PCR amplification of all tested genes was performed on a Stratagene Mx3000P Real-Time PCR system (Agilent Technologies, Santa Clara, CA, USA) using the following cycling procedure: initial denaturation step of 95 °C for 3 min, followed by 40 cycles with denaturation at 95 °C for 15 s, annealing at 55 °C for 30 s, and extension at 72 °C for 20 s. Ubiquntin was used as the internal reference gene, and the relative expression level of each target gene was quantified according to the previously reported ΔΔCT method (Livak and Schmittgen, 2001).
Table 1.
Specific primers for flavonoid biosynthetic genes in I. tinctoria.
| Gene names | Primer sequences (5′ to 3′) |
|---|---|
| PALa | Forward: TCACTCCTTCACTCCCTCTC |
| Reverse: TGATTCCCGCCATCTTGAAG | |
| C4Hb | Forward: AATCGACACGGTTCTTGGAC |
| Reverse: GCTTAGCGTCGTTGAGGTTC | |
| 4CLc | Forward: TTGCCGATGTTCCACATCTA |
| Reverse: GAGCAGCACCAGATTTCACA | |
| CHSd | Forward: GGGATAAGCGACTGGAACTC |
| Reverse: GAAACCAAACAAGACACCCC | |
| CHIe | Forward: CTTCCATCCTCTTCGCTCTC |
| Reverse: CAAGACTCAACCTAGCTCCAG | |
| F3′Hf | Forward: TCAAACACGTTGCACCAAAT |
| Reverse: GACTGCTTCTCCATCCAAGC | |
| FSg | Forward: CCACCTCATATCAGCGATCAAG |
| Reverse: GATTACAGGGAGGTGAACGAG | |
| Ubiquntin | Forward: ACCCTCACGGGGAAGACCATC |
| Reverse: ACCACGGAGACGGAGGACAAG |
PAL is expressed as phenylalanine ammonia lyase gene.
C4H is expressed as cinnamate-4-hydroxylase gene.
4CL is expressed as 4-coumarate coenzyme A ligase gene.
CHS is expressed as chalcone synthase gene.
CHI is expressed as chalcone isomerase gene.
F3′H is expressed as flavonoid 3′-hydroxylase gene.
FS is expressed as flavonol synthase gene.
2.8. Statistical analysis
All experiments were conducted in triplicate, and results were given as averages ± standard deviations. All statistical analyses were carried out using the SPSS statistical software 17.0 (SPSS Inc, Chicago, USA). One-way analysis of variance with Tukey’s test was used to determine significant differences between multiple groups of data at P values <0.05.
3. Results and discussion
3.1. Flavonoid accumulation in ITHRCs treated by different types of UV radiation
It is well-known that UV radiation is a part of the sunlight reaching Earth surface, which is conventionally divided into three classes: UV-A (λmax = 365 nm), UV-B (λmax = 313 nm), and UV-C (λmax = 254 nm). Generally, plants are able to specifically perceive diverse types of UV photons and trigger different signal transduction pathways, which can induce distinct effects on plant secondary metabolisms, and eventually result in accumulating different levels of secondary metabolites (Matsuura et al., 2013). Additionally, UV radiation dose is a critical parameter that can strongly influence the accumulation of certain secondary metabolites in a specific plant in vitro culture (Schreiner et al., 2016). It is well documented that the mild UV radiation can pre-dispose plant cells/organs to a state of low alert via stimuli-specific signaling pathways, which is beneficial for boosting up the biosynthesis of defense secondary metabolites (Anjum et al., 2017; Hideg et al., 2013; Pandey and Pandey-Rai, 2014). In view of this, an appropriate UV radiation intensity of 3 W/m2 was adopted according to the relevant literatures (Anjum et al., 2017; Pandey and Pandey-Rai, 2014). In this work, the UV radiation dose (kJ/m2) was calculated by multiplying 3 W/m2 with the exposure time.
Results of preliminary experiments showed that all three types of UV radiation (UV-A, UV-B, and UV-C) could causes hairy roots serious browning (indication of death) when ITHRCs were exposed to UV radiation more than 15 h under the intensity of 3 W/m2. As reported previously, ITHRCs cultured at day 24 exhibited the maximum productivity of flavonoids and biomass (Gai et al., 2015). Based on the foregoing, UV-A, UV-B, and UV-C with successive radiation doses of 0–162 kJ/m2 (equivalent to exposure time of 0–15 h) were adopted to treat 24 day-old ITHRCs for comprehensively comparing the elicitation effects on flavonoid accumulation on one hand, and in an attempt to further enhance flavonoid production without affecting biomass yield on the other hand.
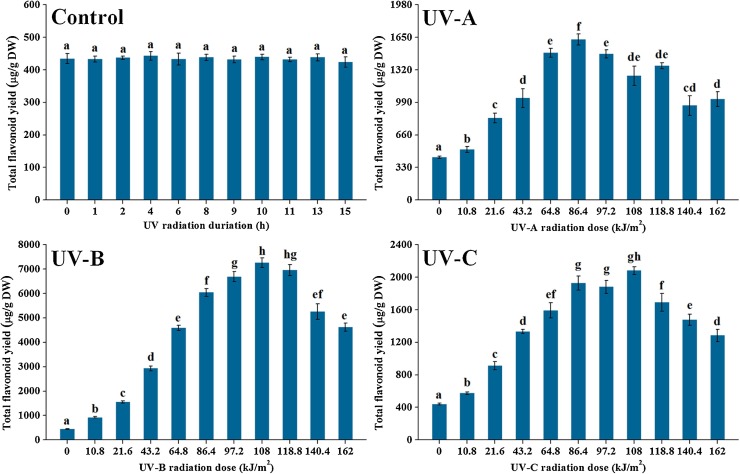
It was clearly observed from Fig. 1 that different types of UV radiation showed distinct influences on flavonoid accumulation in ITHRCs. And the optimal elicitation effects of UV-A, UV-B, and UV-C were found at radiation doses of 86.4, 108, and 108 kJ/m2, where the flavonoid yield increased 3.69-, 16.51-, and 4.73-fold relative to controls, respectively. As expected, the application of UV treatment was indeed feasible for the augmented production of flavonoids in ITHRCs. Moreover, it is worth noting that UV-B exhibited superiority in elevating flavonoid yield as compared with UV-A and UV-C. The flavonoid yield in ITHRCs challenged by UV-B was observed to increase significantly from 0 to 86.4 kJ/m2, reach its maximum level at 108 kJ/m2, and decrease gradually afterwards. Generally, UV-B is capable of causing two contrary stresses on plant metabolism, which is closely related to the radiation dose (Hideg et al., 2013). In detail, exposure of plants to UV-B radiation under mild dose can lead to the reversible and elastic eustress that contributes to the activation of plant defense secondary metabolisms for self-protection, and when the exposure exceeds a tolerance-limit dose, plants will suffer from the irreversible and plastic distress that results in metabolic damage or plant cell/organ death in extreme cases. As inferred, the optimal UV-B radiation dose (108 kJ/m2) obtained in this work might maintain the balance between eustress and distress in ITHRCs, which is conducive to maximally inducing flavonoid accumulation.
Fig. 1.
Elicitation effects of UV-A, UV-B, and UV-C with successive radiation doses (0, 10.8, 21.6, 43.2, 64.8, 86.4, 97.2, 108, 118.8, 140.4, and 162 kJ/m2) on total flavonoid yield in 24 day-old ITHRCs. Mean ± SD values not sharing the same lowercase letters are significantly different (P < 0.05).
3.2. Flavonoid derivative profiles in ITHRCs under the optimal UV-B treatment
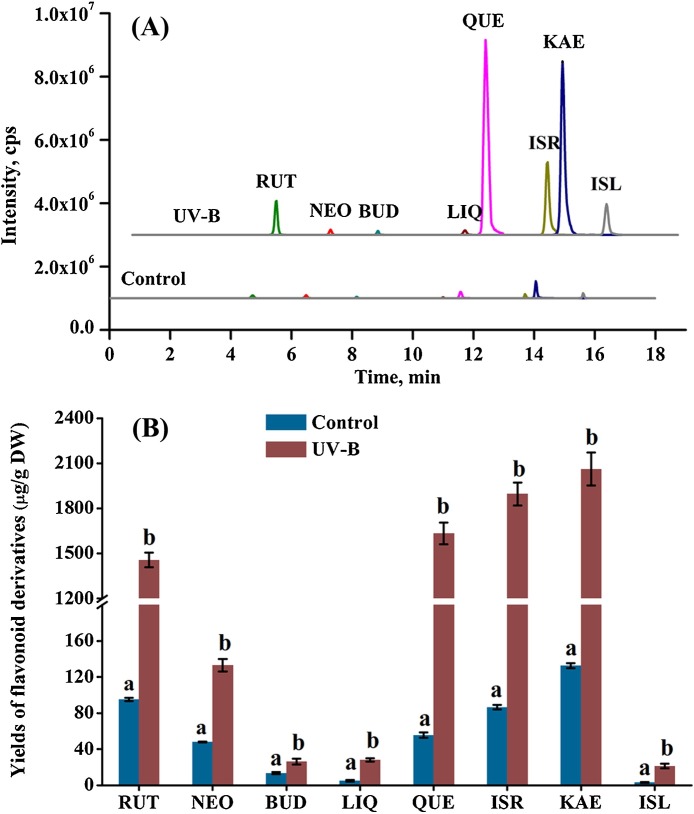
To investigate the changes in profiles of eight flavonoid derivatives (rutin, neohesperidin, buddleoside, liquiritigenin, quercetin, isorhamnetin, kaempferol, and isoliquiritigenin) following the optimal UV-B treatment (108 kJ/m2 of radiation dose), extracts from the treated ITHRCs and control were determined by LC–MS/MS with selected reaction monitoring (SRM) mode (Fig. 2 A), and the yields of all target compounds calculated are presented in Fig. 2B. In detail, the contents of rutin, neohesperidin, buddleoside, liquiritigenin, quercetin, isorhamnetin, kaempferol, and isoliquiritigenin in the treated ITHRCs were 15.33-, 2.76-, 1.96-, 5.67-, 29.38-, 21.91-, 15.54-, and 6.87-fold higher than those in control, respectively. The enhancement of all target compounds can be appreciated via the comparison of LC–MS/MS chromatograms (Fig. 2A). Taken as a whole, the total flavonoid yield in ITHRCs treated by the optimal UV-B radiation was 7259.12 ± 198.19 μg/g DW, which remarkably increased 16.51-fold as against that in control (439.68 ± 8.27 μg/g DW). The significant augmentation of flavonoids was ascribed to the multiple functions of flavonoids in plant photoprotection, such as UV attenuators, antioxidants, auxin transport modulators, etc. (Agati et al., 2011).
Fig. 2.
(A) Representative LC–MS/MS chromatograms of extracts form UV-B treated ITHRCs and control; (B) yields of eight flavonoid derivatives in extracts form UV-B treated ITHRCs and control. RUT, rutin; NEO, neohesperidin; BUD, buddleoside; LIQ, liquiritigenin; QUE, quercetin; ISR, isorhamnetin; KAE, kaempferol; ISL, isoliquiritigenin. Mean ± SD values not sharing the same lowercase letters are significantly different (P < 0.05).
Interestingly, UV-B radiation was observed to tremendously increase accumulation of the more hydroxylated flavonoids in ITHRCs, i.e. rutin (15.33-fold), quercetin (29.38-fold), isorhamnetin (21.91-fold), and kaempferol (15.54-fold). It is well known that the massive production of reactive oxygen species (ROS) is a common event in plants undergoing UV-B stress, which can cause oxidative damages to various functional macromolecules (nucleic acids, lipids, proteins, etc.) in host plant cells (Hideg et al., 2013). Phenolic hydroxyl-substituted flavonoids have been widely known as effective ROS scavengers, which is ascribed to the reduction potentials of these compounds for locating and neutralizing active oxygen radicals (Agati et al., 2012; Giampieri et al., 2018). Accordingly, it is inferred that the significant augmentation of rutin, quercetin, isorhamnetin, and kaempferol in UV-B treated ITHRCs might be attributed to the multiple hydroxyl groups existing in these compounds that could effectively scavenge the highly toxic ROS.
3.3. Antioxidant activities in ITHRCs following UV-B treatment
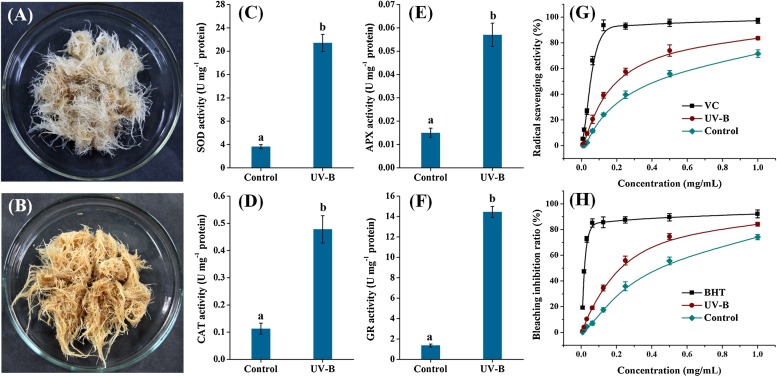
As mentioned above, the oxidative stress mediated by ROS has been flagged as a characteristic event in plants exposed to UV-B radiation (Hideg et al., 2013). In this work, an obvious indication of oxidative stress was observed in UV-B treated ITHRCs that the elicited root tissues (Fig. 3 B) exhibited browning color as compared with control (Fig. 3A). However, more conclusive evidences should be provided for verifying the occurrence of oxidative stress in ITHRCs following UV-B elicitation. Factually, the existence of an efficient antioxidant defense system mainly comprised of antioxidant enzymes and non-enzymatic antioxidant metabolites in higher plants can contribute to detoxifying the detrimental effects of ROS-mediated oxidative stress (Gill and Tuteja, 2010). In this regard, activities of four typical antioxidant enzymes (SOD, CAT, APX, and GR) and antioxidant capacities of non-enzymatic extracts were measured as proxies for the evaluation of oxidative stress in ITHRCs following UV-B treatment.
Fig. 3.
Phenotype comparison of control (A) and UV-B treated ITHRCs (B); enzyme activity comparison of SOD (C), CAT (D), APX (E), and GR (F) in control and UV-B treated ITHRCs; antioxidant activity comparison of extracts from control and UV-B treated ITHRCs evaluated by DPPH radical scavenging assay (E) and β-carotene/linoleic acid bleaching test (F). Mean ± SD values not sharing the same lowercase letters are significantly different (P < 0.05).
In comparison with control, activities of SOD (Fig. 3C), CAT (Fig. 3D), APX (Fig. 3E), and GR (Fig. 3F) significantly increased in UV-B treated ITHRCs, indicating that these antioxidant enzymes were indeed activated to counter UV-B induced alterations in the cellular redox homeostasis. Additionally, extracts from UV-B treated ITHRCs showed superiority in antioxidant efficacies determined by DPPH radical-scavenging assay (Fig. 3G) and β-carotene/linoleic acid bleaching test (Fig. 3H). To be exact, the UV-B elicited samples exhibited better anti-radical activity and lipid peroxidation inhibition capacity, corresponding to lower IC50 values (0.18 mg/mL and 0.22 mg/mL) than those in control (0.43 mg/mL and 0.39 mg/mL), respectively. In this work, the significant increase of total flavonoids, especially the multiple hydroxyl-substituted flavonoids (rutin, quercetin, isorhamnetin, and kaempferol), might contribute to the enhancement in antioxidant activity of UV-B treated samples. Meanwhile, UV-B radiation can stimulate the excessive production of other antioxidant metabolites (ascorbic acid, glutathione, praline, tocopherol, carotenoid, etc.) in plants (Gill and Tuteja, 2010), which might also contribute to the higher antioxidant property of UV-B treated samples. Overall, the enhancement of both antioxidant enzyme activities and non-enzymatic antioxidant properties of extracts was a positive-feedback response to counteract oxidative stress, which provided an indirect evidence of the occurrence of oxidative stress in UV-B treated ITHRCs.
3.4. Cell wall modifications of ITHRCs in response to UV-B treatment
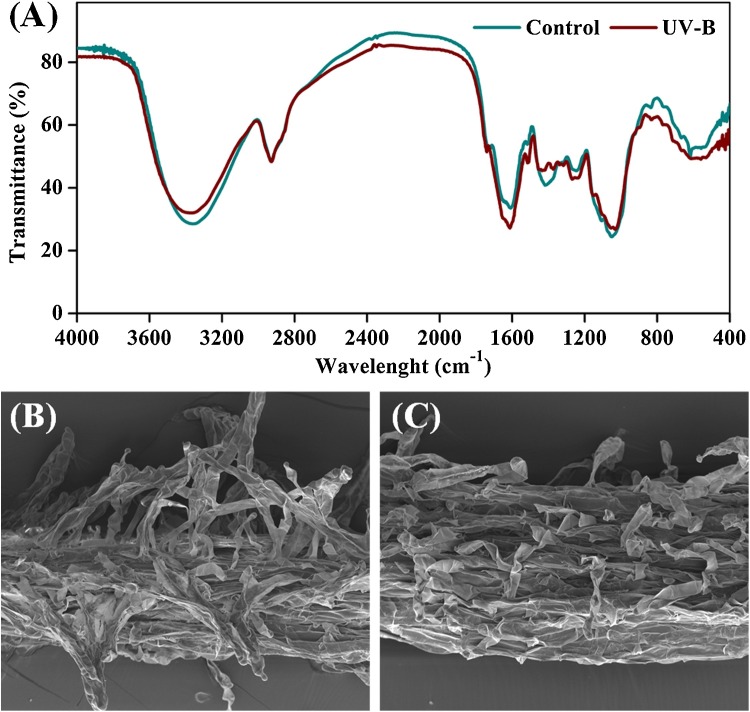
Generally, plant cell wall is an important defense barrier against a variety of biotic and abiotic stresses (Miedes et al., 2014), and ROS formed under stressful conditions can promote cell wall reinforcement (cross-linking of lignin polymers) thus improving plant defense ability conversely (Zhao et al., 2005). Accordingly, FTIR analysis was adopted to compare the functional group modifications of lignin in cell walls between UV-B treated root samples and control. As shown in Fig. 4 A, the significant enhancement in intensities of lignin characteristic peaks at 1258 cm−1 (guaiacyl/syringyl ring and C—O stretching vibration), 1512 cm−1 (aromatic skeletal vibration), and 1606 cm−1 (symmetric aromatic ring C C stretching vibration) were observed in UV-B treated sample as compared with the control spectrum. So, it is inferred that the enrichment of lignin in the treated root tissues might be induced by the oxidative stress following UV-B radiation. Moreover, it is worth mentioning that lignin is an aromatic polymer that can provide the strength and density of cell walls (Vanholme et al., 2010). Therefore, the enhanced lignification of cell walls within the treated root samples also suggested an attempt to attenuate UV-B radiation for self-protection. Additionally, SEM examination was applied to compare the morphology structure modifications of samples between UV-B treatment and control. It is clearly observed from Fig. 4B that the surface of control samples possessed abundant branching root hairs with the intact appearance of long unicellular tubes. On the contrary, the root hairs in the surface of UV-B treated sample appeared severely crimped and disrupted (Fig. 4C), which suggested an obvious indication of wound as the destructive consequence of UV-B stress.
Fig. 4.
(A) FTIR spectrum analysis between control and UV-B treated sample; SEM micrographs of control (B) and UV-B treated sample (C).
Factually, UV-B radiation is capable of triggering multiple cellular signaling pathways in plants as diverse as ROS, wound/defense, DNA damage, and photomorphogenic signaling, which can eventually activate the expression of genes associated with the biosynthesis of UV-protective secondary metabolites (Matsuura et al., 2013). In view of this, the actual occurrence of ROS-mediated oxidative stress and wound event in ITHRCs following UV-B elicitation might contribute to stimulating the transcription of genes involved in flavonoid biosynthetic pathway. Therefore, molecular studies should be carried out to further clarify the transcriptional profiles of flavonoid biosynthetic genes in ITHRCs responsive to UV-B elicitation.
3.5. Biosynthetic gene expression in ITHRCs underlying UV-B treatment
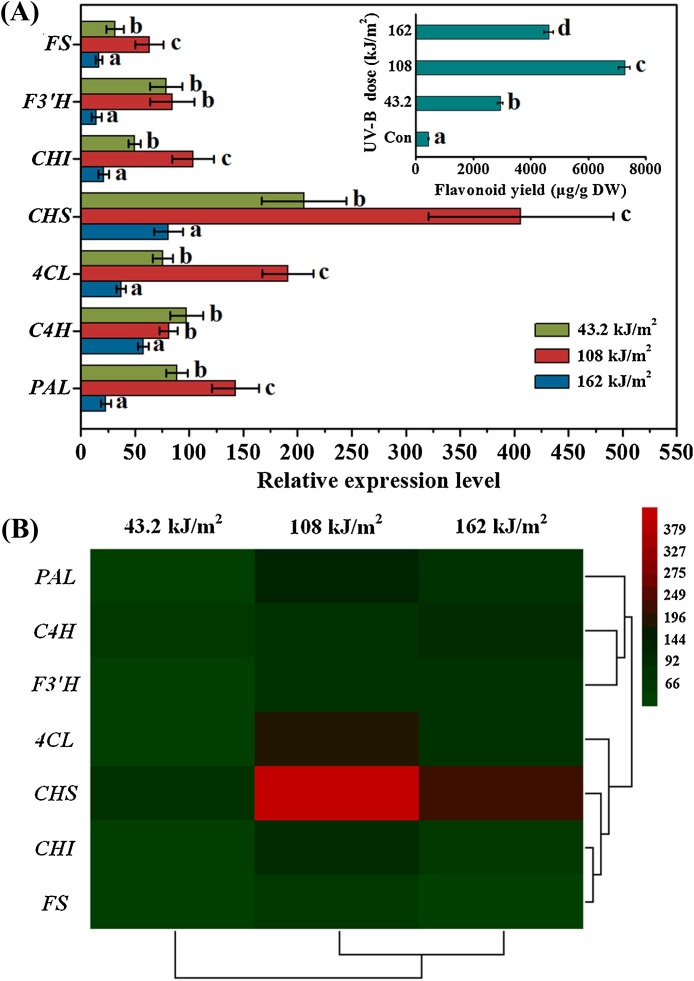
To further shed light on the molecular response mechanism in ITHRCs underlying UV-B treatment, the expression of seven genes encoding enzymes involved in flavonoid biosynthetic pathway including phenylalanine ammonia lyase (PAL), cinnamate-4-hydroxylase (C4H), 4-coumarate coenzyme A ligase (4CL), chalcone synthase (CHS), chalcone isomerase (CHI), flavonoid 3′-hydroxylase (F3′H), and flavonol synthase (FS) were determined by qRT-PCR. In this work, samples collected from UV-B treated ITHRCs under different radiation doses (43.2, 108, and 162 kJ/m2) were taken for qRT-PCR analysis.
As shown in Fig. 5 A, transcription levels of all investigated genes were significantly up-regulated in ITHRCs undergoing UV-B radiation. Moreover, the expression profiles of PAL, 4CL, CHS, CHI, F3′H, and FS genes were found to be positively correlated with the accumulation pattern of flavonoid, which suggested that the boosted flavonoid biosynthesis was a result of the simultaneous up-regulation of these biosynthetic genes. Additionally, it is clearly observed from Fig. 5B that CHS gene exhibited the greatest transcriptional abundance (up to 405.84-fold) at the optimal radiation doses (108 kJ/m2) necessary for the maximal flavonoid production, which indicated that this gene might be more crucial and sensitive for inducing flavonoid biosynthesis in ITHRCs responsive to UV-B elicitation.
Fig. 5.
Transcriptional profiles (A) and hierarchical analyses (B) of seven flavonoid biosynthetic genes in ITHRCs treated by different UV-B radiation doses (43.2, 108, and 162 kJ/m2). PAL, phenylalanine ammonia lyase; C4H, cinnamate-4-hydroxylase; 4CL, 4-coumarate coenzyme A ligase; CHS, chalcone synthase; CHI, chalcone isomerase; F3′H, flavonoid 3′-hydroxylase; FS, flavonol synthase. Mean ± SD values not sharing the same lowercase letters are significantly different (P < 0.05).
Factually, CHS is the gatekeeper of flavonoid biosynthesis that plays a crucial role in regulating the flavonoid pathway, because it is capable of catalyzing the condensation of one mole of p-coumaroyl coenzyme A (CoA) with three moles of malonyl CoA to yield naringenin chalcone, which is the basic skeleton (C6-C3-C6) for the subsequent flavonoid metabolism (Dao et al., 2011). Thus, control of CHS gene expression is an effective way to regulate flavonoid biosynthesis. It was reported that the suppression of CHS gene in tomato using RNAi silencing technology can reduce the total flavonoid levels up to a few percent of wild-type values (Schijlen et al., 2007). Additionally, studies have shown that CHS gene expression is highly influenced by UV and blue light (Jenkins et al., 2001; Wade et al., 2001). Loyall et al. (2000) found that UV-B radiation could up-regulate the expression of CHS gene thus leading to the enhanced accumulation of protective flavonoids in parsley cell cultures. And, oxidative stress was thought to act as a central regulating element in the event of UV-mediated signaling pathway to CHS gene. Similarly, the significant up-regulation of CHS gene in UV-B treated ITHRCs could be ascribed to the actual occurrence of ROS-mediated oxidative stress. Overall, the tremendous induction of CHS transcription here suggested that this gene might play a vital role in flavonoid augmentation in ITHRCs underlying UV-B treatment.
4. Conclusions
The outcomes of this work demonstrated that the simple supplementation of UV-B radiation could effectively enhance the accumulation of pharmacologically active flavonoids in ITHRCs. The optimal UV-B treatment could increase the flavonoid yield by 16.51-fold over control, which offered a promising way for the industrial production of these valuable compounds. Moreover, antioxidant activity enhancement and cell wall reinforcement evidenced the actual occurrence of oxidative stress in ITHRCs following UV-B elicitation, which might contribute to the transcriptional activation of genes involved in flavonoid biosynthetic pathway, especially CHS gene (up to 405.84-fold). Overall, the obtained results in this work not only highlight an efficient elicitation strategy to improve the production level of pharmaceutically important phytochemicals in plant in vitro cultures, but also contribute to the understanding of plant defense responses to stress.
Acknowledgements
The authors gratefully acknowledge the financial supports by National Key R&D Program of China (2017YFD0600205), Fundamental Research Funds for the Central Universities (2572017DA04 and 2572017ET03), Heilongjiang Province Science Foundation for Youths (QC2017012), Scientific Research Start-up Funds for Talents Introduction of Northeast Forestry University (1020160010 and YQ2017-03), and Double First-rate Special Funds (41112432 and 41112460).
References
- Agati G., Biricolti S., Guidi L., Ferrini F., Fini A., Tattini M. The biosynthesis of flavonoids is enhanced similarly by UV radiation and root zone salinity in L. vulgare leaves. J. Plant Physiol. 2011;168:204–212. doi: 10.1016/j.jplph.2010.07.016. [DOI] [PubMed] [Google Scholar]
- Agati G., Azzarello E., Pollastri S., Tattini M. Flavonoids as antioxidants in plants: location and functional significance. Plant Sci. 2012;196:67–76. doi: 10.1016/j.plantsci.2012.07.014. [DOI] [PubMed] [Google Scholar]
- Anjum S., Abbasi B.H., Doussot J., Favre-Réguillon A., Hano C. Effects of photoperiod regimes and ultraviolet-C radiations on biosynthesis of industrially important lignans and neolignans in cell cultures of Linum usitatissimum L. (Flax) J. Photochem. Photobiol. B Biol. 2017;167:216–227. doi: 10.1016/j.jphotobiol.2017.01.006. [DOI] [PubMed] [Google Scholar]
- Arbona V., Flors V., Jacas J., García-Agustín P., Gómez-Cadenas A. Enzymatic and non-enzymatic antioxidant responses of Carrizo citrange a salt-sensitive citrus rootstock, to different levels of salinity. Plant Cell Physiol. 2003;44:388–394. doi: 10.1093/pcp/pcg059. [DOI] [PubMed] [Google Scholar]
- Chen J., Dong X., Li Q., Zhou X., Gao S., Chen R., Sun L., Zhang L., Chen W. Biosynthesis of the active compounds of Isatis indigotica based on transcriptome sequencing and metabolites profiling. BMC Genom. 2013;14:857. doi: 10.1186/1471-2164-14-857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen G., Wang S., Huang X., Hong J., Du L., Zhang L., Ye L. Environmental factors affecting growth and development of Banlangen (Radix Isatidis) in China. Afr. J. Plant Sci. 2015;9:421–426. [Google Scholar]
- Dao T.T.H., Linthorst H.J.M., Verpoorte R. Chalcone synthase and its functions in plant resistance. Phytochem. Rev. 2011;10:397–412. doi: 10.1007/s11101-011-9211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dias M.I., Sousa M.J., Alves R.C., Ferreira I.C. Exploring plant tissue culture to improve the production of phenolic compounds: a review. Ind. Crop. Prod. 2016;82:9–22. [Google Scholar]
- Fini A., Brunetti C., Di Ferdinando M., Ferrini F., Tattini M. Stress-induced flavonoid biosynthesis and the antioxidant machinery of plants. Plant Signal. Behav. 2011;6:709–711. doi: 10.4161/psb.6.5.15069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gai Q.Y., Jiao J., Luo M., Wei Z.F., Zu Y.G., Ma W., Fu Y.J. Establishment of hairy root cultures by Agrobacterium rhizogenes mediated transformation of Isatis tinctoria L. for the efficient production of flavonoids and evaluation of antioxidant activities. PLoS One. 2015;10:e0119022. doi: 10.1371/journal.pone.0119022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giampieri F., Gasparrini M., Forbes-Hernandez T.Y., Mazzoni L., Capocasa F., Sabbadini S., Alvarez-Suarez J.M., Afrin S., Rosati C., Pandolfini T., Molesini B., Sánchez-Sevilla J.F., Amaya I., Mezzetti B., Battino M. Overexpression of the anthocyanidin synthase gene in strawberry enhances antioxidant capacity and cytotoxic effects on human hepatic cancer cells. J. Agric. Food Chem. 2018;66:581–592. doi: 10.1021/acs.jafc.7b04177. [DOI] [PubMed] [Google Scholar]
- Gill S.S., Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010;48:909–930. doi: 10.1016/j.plaphy.2010.08.016. [DOI] [PubMed] [Google Scholar]
- Hamburger M. Isatis tinctoria–from the rediscovery of an ancient medicinal plant towards a novel anti-inflammatory phytopharmaceutical. Phyto Chem. Rev. 2002;1:333–344. [Google Scholar]
- Hideg É., Jansen M.A., Strid Å. UV-B exposure, ROS, and stress: inseparable companions or loosely linked associates? Trends Plant Sci. 2013;18:107–115. doi: 10.1016/j.tplants.2012.09.003. [DOI] [PubMed] [Google Scholar]
- Jenkins G.I., Long J.C., Wade H.K., Shenton M.R., Bibikova T.N. UV and blue light signalling: pathways regulating chalcone synthase gene expression in Arabidopsis. New Phytol. 2001;151:121–131. doi: 10.1046/j.1469-8137.2001.00151.x. [DOI] [PubMed] [Google Scholar]
- Lin C.W., Tsai F.J., Tsai C.H., Lai C.C., Wan L., Ho T.Y., Hsieh C.C., Chao P.D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005;68:36–42. doi: 10.1016/j.antiviral.2005.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livak K.J., Schmittgen T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆CT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Loyall L., Uchida K., Braun S., Furuya M., Frohnmeyer H. Glutathione and a UV light–induced glutathione S-transferase are involved in signaling to chalcone synthase in cell cultures. Plant Cell. 2000;12:1939–1950. doi: 10.1105/tpc.12.10.1939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsh Z., Yang T., Nopo-Olazabal L., Wu S., Ingle T., Joshee N., Medina-Bolivar F. Effect of light, methyl jasmonate and cyclodextrin on production of phenolic compounds in hairy root cultures of Scutellaria lateriflora. Phytochemistry. 2014;107:50–60. doi: 10.1016/j.phytochem.2014.08.020. [DOI] [PubMed] [Google Scholar]
- Matsuura H.N., de Costa F., Yendo A.C.A., Fett-Neto A.G. In: Biotechnology for Medicinal Plants. Chandra S., Lata H., Varma A., editors. Springer-Verlag; Berlin Heidelberg: 2013. Photoelicitation of bioactive secondary metabolites by ultraviolet radiation: mechanisms, strategies, and applications; pp. 171–190. [Google Scholar]
- Miedes E., Vanholme R., Boerjan W., Molina A. The role of the secondary cell wall in plant resistance to pathogens. Front. Plant Sci. 2014;5:a358. doi: 10.3389/fpls.2014.00358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narayani M., Srivastava S. Elicitation: a stimulation of stress in in vitro plant cell/tissue cultures for enhancement of secondary metabolite production. Phytochem. Rev. 2017 doi: 10.1007/s11101-017-9534-0. [DOI] [Google Scholar]
- Nguyen T., Jamali A., Grand E., Morreel K., Marcelo P., Gontier E., Dauwe R. Phenylpropanoid profiling reveals a class of hydroxycinnamoyl glucaric acid conjugates in Isatis tinctoria leaves. Phytochemistry. 2017;144:127–140. doi: 10.1016/j.phytochem.2017.09.007. [DOI] [PubMed] [Google Scholar]
- Pandey N., Pandey-Rai S. Short term UV-B radiation-mediated transcriptional responses and altered secondary metabolism of in vitro propagated plantlets of Artemisia annua L. Plant Cell Tissue Organ Cult. 2014;116:371–385. [Google Scholar]
- Rimando A.M., Duke S.O. Human health and transgenic crops symposium introduction. J. Agric. Food Chem. 2013;61:11693–11694. doi: 10.1021/jf404690h. [DOI] [PubMed] [Google Scholar]
- Schijlen E.G., de Vos C.R., Martens S., Jonker H.H., Rosin F.M., Molthoff J.W., Tikunov Y.M., Angenent G.C., van Tunen A.J., Bovy A.G. RNA interference silencing of chalcone synthase, the first step in the flavonoid biosynthesis pathway, leads to parthenocarpic tomato fruits. Plant Physiol. 2007;144:1520–1530. doi: 10.1104/pp.107.100305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiner M., Mewis I., Huyskens-Keil S., Jansen M.A.K., Zrenner R., Winkler J.B., O’Brien N., Krumbein A. UV-B-induced secondary plant metabolites-potential benefits for plant and human health. Crit. Rev. Plant Sci. 2012;31:229–240. [Google Scholar]
- Schreiner M., Mewis I., Neugart S., Zrenner R., Glaab J., Wiesner M., Jansen M.A. In: III-Nitride Ultraviolet Emitters. Kneissl M., Rass J., editors. Springer-Verlag; Berlin Heidelberg: 2016. UV-B Elicitation of secondary plant metabolites; pp. 387–414. [Google Scholar]
- Tang X., Xiao Y., Lv T., Wang F., Zhu Q., Zheng T., Yang J. High-throughput sequencing and de novo assembly of the Isatis indigotica transcriptome. PLoS One. 2014;9:e102963. doi: 10.1371/journal.pone.0102963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanholme R., Demedts B., Morreel K., Ralph J., Boerjan W. Lignin biosynthesis and structure. Plant Physiol. 2010;153:895–905. doi: 10.1104/pp.110.155119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wade H.K., Bibikova T.N., Valentine W.J., Jenkins G.I. Interactions within a network of phytochrome, cryptochrome and UV-B phototransduction pathways regulate chalcone synthase gene expression in Arabidopsis leaf tissue. Plant J. 2001;25:675–685. doi: 10.1046/j.1365-313x.2001.01001.x. [DOI] [PubMed] [Google Scholar]
- Wang Y., Yang Z., Zhao S., Qin S., Guan W., Zhao Y., Lin Q., Mo Z. Screening of anti-H1N1 active constituents from Radix Isatidis. J. Guangzhou Univ. Trad. Chin. Med. 2011;4:419–422. [Google Scholar]
- Yao X.H., Zhang D.Y., Zu Y.G., Fu Y.J., Luo M., Gu C.B., Li C.Y., Mu F.S., Efferth T. Free radical scavenging capability: antioxidant activity and chemical constituents of Pyrola incarnata Fisch. leaves. Ind. Crop. Prod. 2013;49:247–255. [Google Scholar]
- Zhao J., Davis L.C., Verpoorte R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005;23:283–333. doi: 10.1016/j.biotechadv.2005.01.003. [DOI] [PubMed] [Google Scholar]