Abstract
Objective
To review and compare initial high resolution computed tomography (HRCT) findings in patients with metapneumovirus pneumonia and severe acute respiratory syndrome (SARS-Coronovirus).
Materials and methods
4 cases of metapneumovirus pneumonia (mean age of 52.3 years) in an institutional outbreak (Castle Peak Hospital) in 2008 and 38 cases of SARS-coronovirus (mean age of 39.6 years) admitted to Tuen Mun hospital during an epidemic outbreak in 2003 were included. HRCT findings of the lungs for all patients were retrospectively reviewed by two independent radiologists.
Results
In the metapneumovirus group, common HRCT features were ground glass opacities (100%), consolidation (100%), parenchymal band (100%), bronchiectasis (75%). Crazy paving pattern was absent. They were predominantly subpleural and basal in location and bilateral involvement was observed in 50% of patients. In the SARS group, common HRCT features were ground glass opacities (92.1%), interlobular septal thickening (86.8%), crazy paving pattern (73.7%) and consolidation (68%). Bronchiectasis was not seen. Majority of patient demonstrated segmental or lobar in distribution and bilateral involvement was observed in 44.7% of patients. Pleural effusion and lymphadenopathy were of consistent rare features in both groups.
Conclusion
Ground glass opacities, interlobular septal thickening and consolidations were consistent HRCT manifestations in both metapneumovirus infection and SARS. The presence of bronchiectasis (0% in SARS) may point towards metapneumovirus while crazy paving pattern is more suggestive of SARS.
Keywords: Metapneumovirus, Severe acute respiratory syndrome, SARS-Coronovirus, Atypical pneumonia
1. Introduction
Viral pneumonia is common in both paediatric and adult populations, with influenza virus type A and B accounting for the majority of cases in immunocompetent hosts, while cytomegalovirus, herpesvirus as well as adenovirus dominant in immunocompromised hosts [1]. In 2003, an epidemic of severe acute respiratory distress syndrome (SARS) worldwide had led to the recognition of coronavirus, with its characteristic high infectivity and associated severe morbidity [2]. In 2008, an outbreak of pneumonia in a local regional institution (Castle Peak Hospital) in Hong Kong had raised initial concern and suspicion of SARS return in view of similar clinical and radiological features, only later viral serology revealed identification of metapneumovirus. Indeed, the clinical and radiological findings in viral pneumonias are variable and frequently overlapping [1]. Specific-organism diagnosis often cannot be made on the basis of imaging features alone. Therefore, recognition of various radiological manifestations of viral pneumonias together with clinical history is imperative for narrowing the differential diagnosis. In this article, we reviewed the frequency and compared the differences of thoracic parenchymal findings on high resolution computed tomography (HRCT) between SARS and metapneumovirus pneumonia in the initial course of the disease.
2. Materials and methods
The clinical and radiological features of patients from both the SARS group in 2003 and the metapneumovirus group in May 2008 were retrospectively reviewed. Patients’ demographics and details of HRCT protocol are included in Table 1 .
Table 1.
Patients’ demographics and HRCT protocol of the metapneumovirus and SARS group.
| Metapneumovirus | SARS | |
|---|---|---|
| Hospital | Castle Peak Hospital | Tuen Mun Hospital |
| Year of outbreak | 2008 | 2003 |
| No. of subjects | 4 | 38 |
| Gender | 4 men | 23 women, 15 Men |
| Age range (years) | 47–62 | 19–77 |
| Mean ± 2 SD (years) | 52.3 ± 6.7 | 39.6 ± 14.5 |
| HRCT machine | Multislice 16-heads, Brillance 16, Philips | Single slice CT machine (Picker PQ6000) |
| Plain HRCT parameter | 0.75 × 16 volumetric; 1-s scanning time per section; 120 kV, 100 mA | 1 mm section thickness, 10 mm gap; 1-s scanning time per section; 120 kV, 150 mA, pitch of 1 |
| Scan range | Lung apex to diaphragm, axial plane, at end inspiration | |
| Mean time of HRCT after admission | 3.5 days | 1.7 days |
2.1. Patient selection
Patient list and data were retrieved from computer database using radiology information system and clinical management system. For the SARS group, all patients who were diagnosed with SARS in 2003 by either one of the three laboratory tests: enzyme-linked immunosorbent assay (ELISA), immunofluorescence or polymerase chain reaction (PCR); and had undergone HRCT scan of the thorax shortly after onset of symptoms (<10 days) were included. Those who were diagnosed to have SARS but did not have HRCT study performed or HRCT performed 10 days after onset of symptoms were excluded. A total of 55 patients were identified with SARS and treated in our institution (Tuen Mun Hospital) during 2003. 17 patients were excluded as either CT study was not performed or performed 10 days after onset of symptoms. Hence, only 38 patients (23 females, 15 males) were included in our study, with a mean age of 39.6 years ± 14.5 years [SD] (range 19–77 years old). For the metapneumovirus group, all patients who were diagnosed to have metapneumovirus infection in 2008 by laboratory test nasopharyngeal aspirate RT-PCR polymerase or immunoflorescence staining and had undergone a HRCT scan of the thorax shortly (<10 days) after onset of symptoms were included. Those who were diagnosed to have metapneumovirus infection but did not have HRCT study performed were excluded. A total of 4 patients were identified with metapneumovirus infection from another local institution (Castle Peak Hospital) during May 2008 and were all (4 males) included in our study, with a mean age of 52.3 years ± 6.7 years [SD] (range 47–62 years old).
2.2. High resolution CT imaging of thorax
For patients in the SARS group, non-contrast HRCT scan of the thorax were performed at a mean time of 1.7 days after onset of symptoms, using single slice CT machine (Picker PQ6000) with the following parameters: 1.0 mm section thickness, 10-mm gap, 1-s scanning time per section, 120 kV, 150 mA, pitch of 1. The scanning range was from lung apex to diaphragm in axial plane taken at end inspiration.
For patients in the metapneumovirus group, non-contrast HRCT scan of the thorax was performed at a mean time of 3.5 days after onset of symptoms, using multislice CT machine (16-heads, Brillance 16, Philips) with the following parameters: volumetric 0.75 × 16, 1-s scanning time per section, 120 kV, 100 mA. The scanning range was from lung apex to diaphragm in axial plane taken at end inspiration.
2.3. Image interpretation
All CT images from both groups of patients were reviewed directly from the work station by two independent radiologists, who were aware that all patients had proven pulmonary viral infection but were otherwise blinded to other clinical information. Images were interpreted using lung window setting (window width 1000–1500 HU, window level −700 HU). The CT images were assessed for the presence of the followings: (a) ground glass opacities (defined as area of hazy increased attenuation without obscuration of underlying vascular markings), (b) nodules (centrilobular, perilymphatic or random in distribution), (c) linear densities (interlobular septal line, intralobular septal line, parenchymal bands), (d) crazy paving, (e) consolidations (opacities obscuring underlying vessels), (f) bronchiectasis, (g) pleural effusion, (h) lymphadenopathy (defined as lymph node with a short axis dimension of >0.9 cm). The overall anatomic distribution (segmental, lobar or patchy), zonal predominance (upper, middle or lower zones) and extent (focal or multifocal) of the lesions were also recorded. In order to standardize measurement method and to minimize measurement error, no additional magnification was used. Discrepancy of observation, if any, was resolved after subsequent consensus.
2.4. Data analysis
Interobserver agreement for the measured data was evaluated and expressed with the κ statistic. Agreement was excellent with κ > 0.80; good, κ = 0.61–0.80; moderate, κ = 0.41–0.60; fair, κ = 0.21–0.40; poor, κ < 0.20.
3. Results
The overall comparative results were summarized in Table 2 (with graph).
Table 2.
Summary of HRCT findings in the metapneumovirus and SARS group.
| Metapneumovirus |
SARS |
|||
|---|---|---|---|---|
| Number | % | Number | % | |
| Total no of patients | 4 | 38 | ||
| Ground glass | 4 | 100 | 35 | 92.10 |
| Centrilobular nodules | 3 | 75 | 3 | 8.00 |
| Septal lines | 1 | 25 | 33 | 86.80 |
| Bronchiectasis | 3 | 75 | 0 | 0 |
| Parenchymal bands | 4 | 100 | 16 | 42.20 |
| Crazy paving | 0 | 0 | 28 | 73.80 |
| Consolidation | 4 | 100 | 26 | 68.40 |
| Effusion | 0 | 0 | 1 | 2.60 |
| Lymphadenopathy | 0 | 0 | 4 | 10.50 |
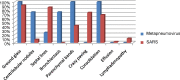 | ||||
In the metapneumovirus group of patients, commonest HRCT features were: ground glass opacities (n = 4, 100%), consolidation (n = 4, 100%), parenchymal band (n = 4, 100%), bronchiectasis (n = 3, 75%), interlobular septal thickening (n = 3, 75%), centrilobular nodules (n = 2, 50%), intralobular septal thickening (n = 1, 25%) (Fig. 1, Fig. 2, Fig. 3 ). They were predominantly subpleural and basal in location showing segmental lobar distribution in pattern. Bilateral involvement was observed in 50% of the patients. Pleural effusion, lymphadenopathy, cavitation and crazy pattern were not identified (0%).
Fig. 1.

49 year-old man with metapneumovirus pneumonia. Lung window axial plain HRCT of the thorax at the level of the heart shows band atelectasis, consolidation and bronchiectasis in the right lower lobe.
Fig. 2.

51 year-old man with metapneumovirus pneumonia. Lung window axial plain HRCT thorax at the level of the heart shows band atelectasis, consolidation, and bronchiectasis are detected in anterior aspect of right lower lobe and subpleural aspect of left lower lobe.
Fig. 3.

49 year-old man with metapneumovirus pneumonia. Lung window axial plain HRCT of the thorax at the level of the lung apices demonstrate glass opacity and atelectasis at the right lung apex.
In the SARS group of patients (n = 38), commonest HRCT features were: ground glass opacities (n = 35, 92.1%), interlobular septal thickening (n = 33, 86.8%), crazy paving pattern (n = 28, 73.7%), consolidation (n = 26, 68%), intralobular septal thickening (n = 16, 42.1%) and parenchymal band (n = 16, 42.1%) (Fig. 4, Fig. 5 ). Bilateral involvement was observed in 44.7% of patients, with the majority showing segmental in distribution. Nodules, lymph node and pleural effusion were rare manifestations, occurring in 8%, 10% and 3% respectively only. None of the patients demonstrated associated bronchiectasis or cavitation.
Fig. 4.
(a and b) 36 year-old woman with SARS infection. Axial lung window HRCT thorax image at the level of the heart shows crazy paving pattern in the medial and central aspect of the right lower lobe.
Fig. 5.

30 year-old man with SARS infection. Plain lung window axial HRCT thorax demonstrates crazy paving pattern and consolidation in the right upper lobe.
Interobserver agreement was excellent, with an overall mean κ value of 0.90. Both groups of patients consistently demonstrated the presence of ground glass opacities, interlobular and intralobular septal thickening, consolidations and parenchymal band. Crazy paving pattern occurred in 73.7% in the SARS group but 0% in the metapneumovirus group. Bronchiectasis occurred in 75% of metapneumovirus group while 0% in SARS patients.
4. Discussion
Respiratory viral infection is seen with increasing frequency worldwide, and is also likely responsible for a large number of cases of pneumonia previously classified as idiopathic. Severe acute respiratory distress syndrome (SARS), being caused by coronavirus SARS-CoV with a characteristic high infectivity, hosted center stage worldwide in 2003, causing a global fatality of approximately 11% [3] and with associated severe morbidity. An epidemic of metapneumovirus chest infection occurred in 2008, affecting 4 patients in a local institution (Castle Peak Hospital) in Hong Kong. Human metapneumovirus is a paramyxovirus first described in 2001 but has been present in human population for at least 50 years [4]. It infects people of all ages causing mild to severe respiratory infections most active in late winter and early spring – a period when many other respiratory viruses are also circulating [4], [5], [6]. Its clinical impact and epidemiology is very similar to other respiratory viral infections and differentiation is often difficult on the bases of clinical signs alone [2]. Indeed, the initial period of metapneumovirus outbreak had raised concern of SARS return due to similar clinical and radiological features.
Most respiratory viral infections produce nonspecific acute symptoms such as fever, nonproductive cough, dyspnea and hypoxemia [7]. Conventional chest radiography is usually the first imaging technique performed but with lack of sensitivity [8]. Past studies demonstrated that HRCT was the most sensitive imaging method for detecting early lung changes in acute pulmonary diseases [7], [9], [10], [11], [12]. Characteristic CT findings in SARS include ground glass opacification or consolidation, thickening of the interlobular or intralobular interstitium, and crazy paving appearance [3], [13], [14], [15], [16], [17], [18], with a peripheral and lower lobe predominance in the early course of the disease, progressing to diffuse and bilateral involvement in the latter stage [13], [15], [18]. On the other hand, characteristic CT findings in metapneumovirus infection include patchy areas of ground glass attenuation, small nodules, and multifocal areas of consolidation in a bilateral asymmetrical distribution [4], [5], [6], [7]. Despite so, these findings are nonspecific and can be found in other types of bacterial or viral/atypical pneumonia such as Mycoplasma, Chlamydia or Legionella [18], [19], [20], [21]. Ground glass attenuation, in particular, is a pattern of low specificity yet most commonly seen in respiratory viral infection, regardless of cause [1].
Findings in our study demonstrated that ground glass opacities, consolidation and interlobular/intralobular septal thickening were of consistent common initial CT features in both metapneumovirus and SARS groups, distributing in a subpleural/peripheral and basal lung locations in the early course of the disease, which were also in agreement with findings from various past literatures [18], [22]. The presence of centrilobular nodules, parenchymal band and consolidation are significantly more frequently present in metapneumovirus infections. Pleural effusion, lymphadenopathy and cavitation, in the contrary, were of consistent rare features in both groups. Bronchiectasis however occurred much more often in the metapneumovirus group while crazy paving pattern and septal lines occurred significant more frequently in the SARSs group. These two features, therefore, can be used as markers in differentiation between the two groups of patients during initial phase of the disease. Despite this seemingly positive finding, one should remember that these are nevertheless nonspecific and can be found in other types of viral pneumonia. In particular, bronchiectasis, although not a feature in initial stage of SARS, can occur during the healing phase in relation to fibrosis formation [15], [22], [23]. Correlation with clinical history and other parameters is therefore important in search for a diagnosis.
One of the major limitations in our study was the small number of data size available in the metapneumovirus group, which the results might not be truly reflecting the common findings in all patients. Secondly, the recruited patients in both groups were heterogeneous in that the severity of clinical symptoms and clinical outcome were not taken into account, which could lead to potential false positive results or overestimation of the involvement.
In conclusion, the main goal of our study was to describe and compare the HRCT features between SARS and metapneumovirus in early course of the disease. The overlapping CT features indicates difficulty in obtaining high accuracy in differentiation between the two groups, as well as between other respiratory viral infections or even from other diseases such as acute extrinsic allergic alveolitis, bronchioltis obliterans with organizing pneumonia or chronic eosinophilic pneumonia [1]. Predictive values are also likely to be low. Nevertheless, these findings, when present, should alert clinician to the possibility of respiratory viral infection and prompt the referring physician to search for the diagnosis and treatment plan. Clinical information is indispensable and microbiological test remains the gold standard in making diagnosis.
References
- 1.Kim E.A., Lee K.S., Primack S.L. Viral pneumonias in adults: radiological and pathological findings. Radiographics. 2002;22:137–149. doi: 10.1148/radiographics.22.suppl_1.g02oc15s137. [DOI] [PubMed] [Google Scholar]
- 2.Lee N., Hui D.S.C., Wu A. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1986–1994. doi: 10.1056/NEJMoa030685. [DOI] [PubMed] [Google Scholar]
- 3.Paul N.S., Roberts H., Butany J. Radiologic pattern of disease in patients with severe acute respiratory syndrome: the Toronto experience. Radiographics. 2004;24:553–563. doi: 10.1148/rg.242035193. [DOI] [PubMed] [Google Scholar]
- 4.Ebihara T., Endo R., Kikuta H. Human metapneumovirus infection in Japanese children. J Clin Microbiol. 2004;42(1):126–132. doi: 10.1128/JCM.42.1.126-132.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Falsey A.R., Erdman D., Anderson L.J., Walsh E.E. Human metapneumovirus infections in young and elderly adults. J Infect Dis. 2003;187(1):785–790. doi: 10.1086/367901. [DOI] [PubMed] [Google Scholar]
- 6.Williams J.V., Harris P.A., Tollefson S.J. Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N Engl J Med. 2004;350(5):443–450. doi: 10.1056/NEJMoa025472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Franquet T., Rodriguez S., Martino R., Giménez A., Salinas T., Hidalgo A. Thin-section CT findings in hematopoietic stem cell transplantation recipients with respiratory virus pneumonia. AJR Am J Roentgenol. 2006;187:1085–1090. doi: 10.2214/AJR.05.0439. [DOI] [PubMed] [Google Scholar]
- 8.Anuradha Rao T.N., Paul N., Chung T. Values of CT in assessing probable severe acute respiratory syndrome. AJR Am J Roentgenol. 2003;181:317–319. doi: 10.2214/ajr.181.2.1810317. [DOI] [PubMed] [Google Scholar]
- 9.Kotloff R.M., Ahya V.N., Crawford S.W. Pulmonary complications of solid and hematopoietic stem cell transplantation. Am J Respir Crit Care Med. 2004;170:22–48. doi: 10.1164/rccm.200309-1322SO. [DOI] [PubMed] [Google Scholar]
- 10.Franquet T., Müller N.L., Giménez A., Martínez S., Madrid M., Domingo P. Infectious pulmonary nodules in immunocompromised patients: usefulness of computed tomography in predicting their etiology. J Comput Assist Tomogr. 2003;27:461–468. doi: 10.1097/00004728-200307000-00001. [DOI] [PubMed] [Google Scholar]
- 11.Logan P.M., Primack S.L., Staples C., Miller P.R., Müller N.L. Acute lung disease in the immunocompromised host: diagnostic accuracy of the chest radiograph. Chest. 1995;108:1283–1287. doi: 10.1378/chest.108.5.1283. [DOI] [PubMed] [Google Scholar]
- 12.Heussel C.P., Kauczor H.U., Heussel G., Fisher B., Mildenberger P., Thelen M. Early detection of pneumonia in febrile neutropenic patients: use of thin-section CT. AJR Am J Roentgenol. 1997;169:1347–1353. doi: 10.2214/ajr.169.5.9353456. [DOI] [PubMed] [Google Scholar]
- 13.Antonio G.E., Wong K.T., Hui D.S.C. Imaging of severe acute respiratory syndrome in Hong Kong. AJR Am J Roentgenol. 2003;181:11–17. doi: 10.2214/ajr.181.1.1810011. [DOI] [PubMed] [Google Scholar]
- 14.Nicolaou S., Al-Nakshabandi N.A., Müller N.L. SARS: imaging of severe acute respiratory syndrome. AJR Am J Roentgenol. 2003;18:1247–1249. doi: 10.2214/ajr.180.5.1801247. [DOI] [PubMed] [Google Scholar]
- 15.Paul N.S., Chung T., Konen E. Prognostic significance of the radiographic pattern of disease in patients with severe acute respiratory syndrome. AJR Am J Roentgenol. 2004;182:493–498. doi: 10.2214/ajr.182.2.1820493. [DOI] [PubMed] [Google Scholar]
- 16.Hsieh S.C., Chan W.P., Chien J.C.W. Radiographic appearance and clinical outcome correlates in 26 patients with severe acute respiratory syndrome. AJR Am J Roentgenol. 2004;182:1119–1122. doi: 10.2214/ajr.182.5.1821119. [DOI] [PubMed] [Google Scholar]
- 17.Müller N.L., FitzGerald J.M. Severe acute respiratory syndrome (SARS) Thorax. 2003;58:919. doi: 10.1136/thorax.58.11.919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wong K.T., Antonio G.E., Hui D.S.C. Severe acute respiratory syndrome: radiographic appearances and pattern of progression in 138 patients. Radiology. 2003;228:401–406. doi: 10.1148/radiol.2282030593. [DOI] [PubMed] [Google Scholar]
- 19.Garbino J., Gerbase M.W., Wunderli W. Respiratory viruses and severe lower respiratory tract complications in hospitalized patients. Chest. 2004;125:1033–1039. doi: 10.1378/chest.125.3.1033. [DOI] [PubMed] [Google Scholar]
- 20.Goodman L.R., Goren R.A., Teptick S.K. Radiographic evaluation of pulmonary infection. Med Clin N Am. 1980;64:553–574. doi: 10.1016/s0025-7125(16)31609-1. [DOI] [PubMed] [Google Scholar]
- 21.Macfarlane J.T., Miller A.C., Roderick Smith W.H., Morris A.H., Rose D.H. Comparative radiographic features of community acquired legionnaires disease, pneumococcal pneumonia, mycoplasma pneumonia, and psittacosis. Thorax. 1984;39:28–33. doi: 10.1136/thx.39.1.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Müller N.L., Ooi G.C., Khong P.K., Zhou L.J., Tsang K.W.T., Nicolaou S. High-resolution CT findings of severe acute respiratory syndrome at presentation and after admission. AJR Am J Roentgenol. 2004;182:39–44. doi: 10.2214/ajr.182.1.1820039. [DOI] [PubMed] [Google Scholar]
- 23.Antonio G.E., Wong K.T., Hui D.S.C. Thin-section CT in patients with severe acute respiratory syndrome following hospital discharge: preliminary experience. Radiology. 2003;228:810–815. doi: 10.1148/radiol.2283030726. [DOI] [PubMed] [Google Scholar]



