Abstract
Background
Chronic Obstructive Pulmonary Disease (COPD) exacerbations are associated with viral infections. We wished to determine if respiratory viral infection of children in the community was associated with hospital admissions of patients with exacerbations of COPD.
Methods
We collected data over a 45-month period from the Northern Ireland Regional Virus Laboratory and from a general hospital in the same locality. We studied the relationship between upper respiratory infections in children and COPD admissions. We also examined the role of school holidays.
Results
Correlations were seen between the frequency of all viral infections in children and the number of adult COPD hospitalizations . Subgroup analysis showed distinct relationships with epidemics of; influenza A , influenza B , adenovirus , respiratory syncytial virus and hospital admissions of patients with COPD. There were significantly fewer COPD admissions in the week after the start of a school holiday period .
Conclusions
When children are hospitalized with viral respiratory infection there is an associated rise in adult COPD admissions. This suggests exacerbations of COPD are associated with epidemics of respiratory viruses. When children are on school holidays there is a reduction in COPD admissions in the community. This provides further support for respiratory viruses in the pathogenesis of COPD exacerbations.
Keywords: Chronic obstructive pulmonary disease, Respiratory viruses, Children, Hospital admissions, Exacerbation, School holidays
Abbreviations: °C, degrees celsius; COPD, chronic obstructive pulmonary disease; CI, confidence interval; FCE, finished consultant episode; G, standard gravity; ICD, international classification of disease; Min, minute(s); PBS, phosphate buffered saline; rs, Spearman's correlation; SPSS, statistical package for the social sciences; μl, microlitre
Introduction
Chronic Obstructive Pulmonary Disease (COPD) is a major cause of morbidity and mortality.1 It contributes to a large number of acute admissions to hospital. There are seasonal peaks in these episodes.2 This trend in admissions is set to rise and represents a major expenditure in the health services.2, 3 Molecular techniques for the detection of respiratory viruses have recently been developed. Viruses have been identified in 39–56% of COPD exacerbations.4, 5, 6 In asthma, correlations have been found between the seasonal patterns of upper respiratory infections in children and adult hospitalization.7 We hypothesized that a similar association could be obtained between paediatric viral respiratory tract infections and adult admissions with COPD. We obtained data from the regional laboratory. Respiratory samples are routinely screened for viral pathogens on children admitted to hospital. This should reflect the prevalence of upper respiratory infection in children attending hospital at that time. Adult admissions with COPD were recorded from a general hospital in the greater Belfast area.
Methods
Subjects
Childhood virus identification data was obtained from the Northern Ireland Regional Virus Laboratory. The children were all under 15 years of age. Each virus identified represents one patient during one hospital attendance. We screened for duplicate samples and these were removed prior to analysis. Adult hospital admissions with COPD (ICD code J44.0 or 496.0) for the same time period were identified from the Mater Hospital, an urban general hospital in Belfast. Patients were admitted to hospital following a decision to admit by a senior Doctor in the emergency department. Those patients who were coded on discharge as having other co morbid conditions contributing to their admission (e.g. left ventricular failure) were excluded from the analysis. This information was collected for each week from January 1, 1996 to September 30, 1999.
Viral detection
Respiratory viruses were detected by a combination of immunofluorescence and virus culture. Those specimens negative by immunofluorescence were re-tested by virus culture. Respiratory specimens were suspended in 5 ml of PBS, centrifuged at 2600g for 5 min and the supernatant discarded. The sample was resuspended in 100 μl of PBS and the cells were spotted onto a multiwell slide, air dried, fixed in acetone at 4 °C for 10 min and stained with 10 μl of direct fluorescent-labelled monoclonal antibody (Chemicon, Harrow, England, UK). Antibodies to adenovirus, respiratory syncytial virus, parainfluenza type 1, parainfluenza type 2, parainfluenza type 3, influenza A and influenza B were used. Following 30 min incubation in a 37 °C humidified chamber, the slide was washed in PBS for 5 min, dried, mounted with a glass coverslip using glycerol and examined under a Zeiss® IV epifluorescence microscope, set at ×40 magnification (Zeiss Microscopy, Hamburg, Germany). Samples negative by immunofluorescence were cultured using a standard protocol as previously described.8 Briefly, we inoculated multiple cell lines in suspension on a microtitre plate. These were incubated in a sealed box with 5% CO2 at 37 °C for up to 7 days. The plates were read daily for evidence of cytopathic effect using an inverted Olympus® microscope at ×40 magnification (Olympus, Hamburg, Germany).
School holidays
School holiday dates were obtained from the Department of Education for Northern Ireland. Weeks in which there were 3 or more days of schools holidays were denoted as a holiday period (for subsequent analysis).
Statistical analysis
Cosine and Sine terms were used to pick up sinusoidal variation in COPD admissions throughout the year. School holidays were included in the Poisson regression as a dummy (0/1) variable and also lagged by 1 and 2 weeks, this variable was put into the prediction only when school holidays played a role (i.e. only during a school holiday period). Relationships were assessed using Spearman's rank correlation. Individual viruses were correlated with COPD admissions. The Cosine and Sine terms along with the school holidays variable were included in this analysis.
Poisson regression was applied to individual respiratory viruses to calculate the relative risk (95% confidence intervals) of COPD admissions in the same week as well as 1 and 2 weeks later. This was also performed with the Cosine and Sine terms with the school holiday variable included in the analysis. Statistical methods used are similar to those previously described.9 Statistical analyses were performed using the SPSS (version 11) package and Poisson regression on Stata 8.0.
Results
Respiratory viral infection in children
A total of 1243 children were studied, 54.5% were male. The median age of this patient group was 0.43 years, with a (interquartile range 0.1–0.7) range of newborn to 15 years. Respiratory viruses were detected in 2279 specimens. The samples submitted for testing were most commonly respiratory secretions (2160), followed by sputum (89), swabs (28) and bronchoalveolar lavage (2). The relative frequency of viruses identified were as follows: respiratory syncytial virus 1871 (82%); influenza A, 184 (8.1%); influenza B, 19 (0.8%); adenovirus, 56 (2.5%); parainfluenza 1, 39 (1.7%); parainfluenza 2, 6 (0.3%); parainfluenza 3, 104 (4.6%).
Adult admissions with COPD
There were 1217 admissions at the Mater hospital with exacerbations of COPD, 48.9% were male. The median age was 68.5 (interquartile range 65.7–71.5) years. Table 1 shows the variables including COPD admissions and the viruses studied. The numbers of weeks of the study that a particular virus was identified are listed along with the number of episodes per week. Fig. 1 illustrates COPD admissions and the number of viruses seen in children for each week during the study.
Table 1.
The incidence of admissions and viruses detected in this study.
| Variable | Number (%) of weeks virus identified | Incidence (per week) |
|---|---|---|
| COPD admissions | — | 6.2±2.7 |
| Respiratory syncytial virus | 109 (56) | 5 (1–25) |
| Influenza A | 58 (30) | 2 (1–4) |
| Influenza B | 12 (6) | 1 (1–2) |
| Adenovirus | 45 (23) | 1 (1–1) |
| Parainfluenza 1 | 29 (15) | 1 (1–1) |
| Parainfluenza 2 | 5 (3) | 1 (1–1) |
| Parainfluenza 3 | 51 (26) | 2 (1–3) |
Column 1 shows the variables including COPD admissions and the viruses studied. Column 2 shows the number of weeks of the study that a particular virus was identified. Column 3 shows the number of episodes per week; median (IQR).
Figure 1.
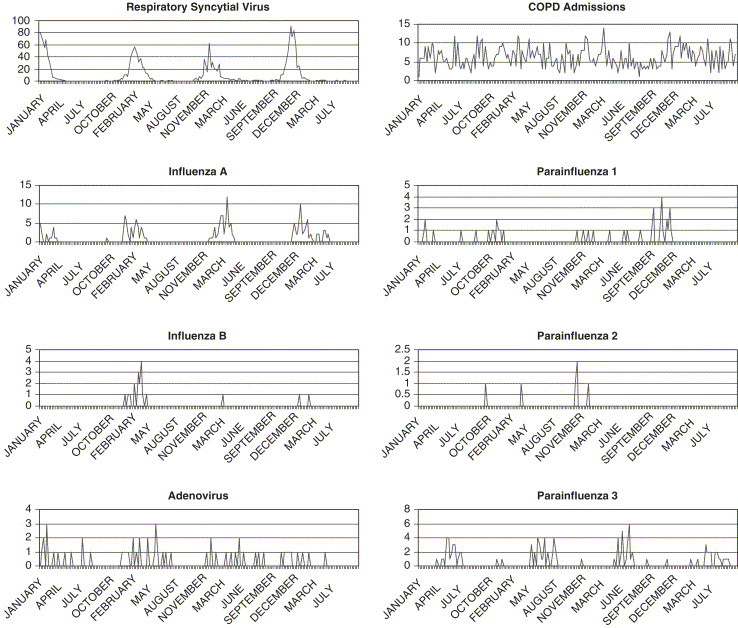
COPD admissions and virus detection during the study.
Correlations
Correlations between the total numbers of respiratory viruses as well as those for individual viruses are shown in Table 2 . A significant correlation was seen between the total number of viruses and COPD hospitalization. Subgroup analysis showed a relationship between influenza A , influenza B respiratory syncytial virus , adenovirus and COPD admissions. Fig. 2 illustrates COPD admissions and total numbers of respiratory viruses seen in children for each week during the study.
Table 2.
Correlations (Spearman's) between respiratory viruses and COPD admissions.
| Respiratory virus | rs value | Significance ( 2-tailed ) |
|---|---|---|
| All viruses | 0.231* | 0.001 |
| Respiratory syncytial virus | 0.231* | 0.001 |
| Influenza A | 0.248* | 0.0005 |
| Influenza B | 0.145* | 0.042 |
| Adenovirus | 0.175* | 0.014 |
| Parainfluenza 1 | −0.002 | 0.98 |
| Parainfluenza 2 | −0.038 | 0.59 |
| Parainfluenza 3 | −0.076 | 0.29 |
* Correlation is significant.
Figure 2.
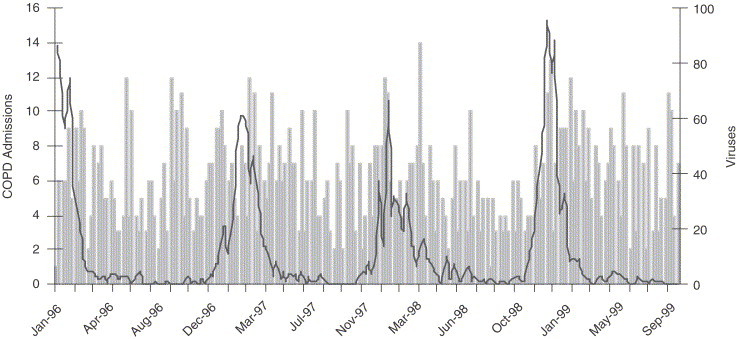
COPD admissions and total numbers of respiratory viruses seen in children. The admissions are illustrated by bars, each bar represents 1 week and the left Y-axis shows the range. Viruses detected in children are shown as a solid line; the right Y-axis shows the range.
School holidays
Following Poisson regression analysis of school holidays on numbers of COPD admissions, there were significantly fewer COPD admissions for the week after the start of a holiday period, , relative risk 0.85 (95% CI 0.73–0.98) in comparison to numbers of admissions during term time. Thus, the week after the start of a school holiday period there were 15% fewer COPD admissions. There was no increase in admissions seen for up to 3 weeks following the end of a school holiday period.
Regression analysis
Individual viruses were also correlated with COPD admissions, to determine if the detection of particular viruses could be associated with increased COPD admissions in the following weeks. Table 3 shows significant correlations with respiratory syncytial virus, influenza A, influenza B and adenovirus however not with total numbers of viruses. COPD admissions were increased for up to 2 weeks after respiratory syncytial virus and influenza A. When the Cosine and Sine terms with the school holidays variable were applied to this analysis, no significant correlations were seen.
Table 3.
The relative risk of viral infection of children and COPD admission.
| Predictor | Poisson regression coefficient |
||
|---|---|---|---|
| Relative risk (95% confidence interval) |
|||
| Same week | 1 weeks time | 2 weeks time | |
| Virus total | 1.09 (0.95, 1.26) | 1.11 (0.96, 1.28) | 1.11 (0.96, 1.28) |
| Respiratory syncytial virus | 1.17 (1.05, 1.31)** | 1.15 (1.02, 1.29)* | 1.18 (1.05, 1.33)** |
| Influenza A | 1.25 (1.11, 1.40)*** | 1.14 (1.01, 1.29)* | 1.18 (1.05, 1.33)** |
| Influenza B | 1.27 (1.03, 1.57)* | 1.20 (0.97, 1.49) | 1.20 (0.97, 1.49) |
| Adenovirus | 1.20 (1.05, 1.36)** | 1.02 (0.89, 1.17) | 1.07(0.94, 1.22) |
| Parainfluenza 1 | 1.01 (0.86, 1.18) | 1.05 (0.90, 1.23) | 1.05 (0.90, 1.23) |
| Parainfluenza 2 | 0.93 (0.65, 1.35) | 0.96 (0.67, 1.38) | 0.80 (0.54, 1.19) |
| Parainfluenza 3 | 0.93 (0.81, 1.06) | 0.90 (0.79, 1.02) | 0.94 (0.84, 1.08) |
; ; .
This table illustrates Poisson regression coefficients for relative risk of COPD admission per week (as well as the following 2 weeks) with respect to the detection of the named respiratory viruses.
Discussion
We have identified several correlations between viral respiratory tract infections in children and acute hospital admissions of patients with exacerbations of COPD. Viral infection is known to cause acute exacerbations and hospital admission.10 Respiratory syncytial virus was the most common pathogen identified. Until recently respiratory syncytial virus was infrequently detected in COPD exacerbations.4, 5 However, in the last 4 years several studies have implicated this virus in adults. Borg et al.11 identified respiratory syncytial virus infection in 28% of COPD patients, they attributed this higher incidence to a more sensitive real-time PCR assay. Furthermore Falsey et al. demonstrated respiratory syncytial virus infection was associated with 11.4% of hospitalizations for COPD patients.12
Viral infection in this group of children is likely to reflect the infection profile of the entire community. The spread of common respiratory viruses through a community is facilitated by transfer amongst young children.7 The reduction in COPD admissions seen in the week after the start of school holidays may reflect the protective effect of holidays. There is less contact between children reducing the spread of respiratory pathogens.
Significant correlations were obtained for those viruses (respiratory syncytial virus, influenza A, influenza B, and adenovirus) which are known to precipitate exacerbations of COPD.4, 5, 6 There were peaks in COPD admissions where no viruses were identified. These may be related to rhinovirus and human metapnemovirus infection. We did not test for human rhinovirus during this period as it is fastidious and requires molecular detection. Coronavirus similarly was not identified. Human metapneumovirus was only discovered after the study was completed. This underscores the possibility of other endemic viruses which have not yet been identified, but which may contribute to the peaks in numbers of acute exacerbations in adults.13
The correlation between COPD admissions and the detection of respiratory syncytial virus 2 weeks previously reflects the longer incubation period of respiratory syncytial virus infection. By comparison influenza B was associated with a rise in COPD admissions in the same week; this virus has a shorter incubation period. It is notable that the regression analysis did not identify any particular virus as having an increased relative risk with respect to subsequent COPD admissions when the analysis was performed on data in which seasonal variation in COPD admissions had been removed. Previous investigators have demonstrated no seasonally related differences in disease characteristics for COPD exacerbations,14 thus in removing seasonal variation prior to analysis the true effects of viral detection on COPD admissions may be been masked.
Thompson et al.15 have published data linking influenza and respiratory syncytial virus infection with increased mortality in the elderly. There are increased admissions in older people in winter. Fleming showed that acute respiratory infections are associated with hospitalization and increased mortality.16 This is reflected in the main findings between viruses in children and adult COPD admissions which have been highlighted in this study. It reinforces the health service implications of winter infection and increased exacerbations of COPD. It is also noteworthy that the peak death rate is in patients diagnosed with acute respiratory disease.16 Influenza has been linked with increased health care use by children and the elderly. There is an excess mortality particularly in the elderly.17 Several studies have demonstrated the cost-effectiveness and positive economic aspects of influenza vaccination in children18, 19 and associated reductions in respiratory morbidity amongst their household contacts.20 This study supports previous evidence that COPD patients benefit from influenza vaccination.
In summary, this study underscores the importance of viral infections in the pathogenesis of COPD. It relates children's infections to hospitalizations of adults with COPD. It highlights the relationship between influenza A and B, adenovirus and respiratory syncytial virus with COPD exacerbations.
Acknowledgements
John Whitmore (Mater Hospital Information Technology department) provided COPD admission data. Dr. Chris Patterson (Department of Epidemiology & Public Health, Queen's University Belfast) assisted with the statistical analysis of data.
References
- 1.The Burden of Lung Disease. A statistics report from the British Thoracic Society. Website, 2001.
- 2.Andersson F., Borg S., Jansson S.A., Jonsson A.C., Ericsson A., Prutz C. The costs of exacerbations in chronic obstructive pulmonary disease (COPD) Respir Med. 2002;96(9):700–708. doi: 10.1053/rmed.2002.1334. [DOI] [PubMed] [Google Scholar]
- 3.Faulkner M.A., Hilleman D.E. The economic impact of chronic obstructive pulmonary disease. Expert Opin Pharmacother. 2002;3(3):219–228. doi: 10.1517/14656566.3.3.219. [DOI] [PubMed] [Google Scholar]
- 4.Rohde G., Wiethege A., Borg I., Kauth M., Bauer T.T., Gillissen A. Respiratory viruses in exacerbations of chronic obstructive pulmonary disease requiring hospitalisation: a case-control study. Thorax. 2003;58(1):37–42. doi: 10.1136/thorax.58.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Seemungal T., Harper-Owen R., Bhowmik A., Moric I., Sanderson G., Message S. Respiratory viruses, symptoms, and inflammatory markers in acute exacerbations and stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001;164(9):1618–1623. doi: 10.1164/ajrccm.164.9.2105011. [DOI] [PubMed] [Google Scholar]
- 6.Beckham J.D., Cadena A., Lin J., Piedra P.A., Glezen W.P., Greenberg S.B. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330. doi: 10.1016/j.jinf.2004.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Johnston S.L., Pattemore P.K., Sanderson G., Smith S., Campbell M.J., Josephs L.K. The relationship between upper respiratory infections and hospital admissions for asthma: a time-trend analysis. Am J Respir Crit Care Med. 1996;154(3 Pt 1):654–660. doi: 10.1164/ajrccm.154.3.8810601. [DOI] [PubMed] [Google Scholar]
- 8.O’Neill H.J., Russell J.D., Wyatt D.E., McCaughey C., Coyle P.V. Isolation of viruses from clinical specimens in microtitre plates with cells inoculated in suspension. J Virol Methods. 1996;62(2):169–178. doi: 10.1016/s0166-0934(96)02102-7. [DOI] [PubMed] [Google Scholar]
- 9.Katsouyanni K., Schwartz J., Spix C., Touloumi G., Zmirou D., Zanobetti A. Short term effects of air pollution on health: a European approach using epidemiologic time series data: the APHEA protocol. J Epidemiol Community Health. 1996;50(Suppl 1):S12–S18. doi: 10.1136/jech.50.suppl_1.s12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Greenberg S.B. Viral respiratory infections in elderly patients and patients with chronic obstructive pulmonary disease. Am J Med. 2002;112(Suppl 6A):28S–32S. doi: 10.1016/S0002-9343(01)01061-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Borg I., Rohde G., Loseke S., Bittscheidt J., Schultze-Werninghaus G., Stephan V. Evaluation of a quantitative real-time PCR for the detection of respiratory syncytial virus in pulmonary diseases. Eur Respir J. 2003;21(6):944–951. doi: 10.1183/09031936.03.00088102. [DOI] [PubMed] [Google Scholar]
- 12.Falsey A.R., Hennessey P.A., Formica M.A., Cox C., Walsh E.E. Respiratory syncytial virus infection in elderly and high-risk adults. N Engl J Med. 2005;352(17):1749–1759. doi: 10.1056/NEJMoa043951. [DOI] [PubMed] [Google Scholar]
- 13.van den Hoogen B.G., de Jong J.C., Groen J., Kuiken T., de Groot R., Fouchier R.A. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat Med. 2001;7(6):719–724. doi: 10.1038/89098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.de la Iglesia M.F., Pellicer V.C., Ramos P.V., Nicolas M.R., Pita F.S., Diz-Lois M.F. Chronic obstructive pulmonary disease and the seasons of the year. Arch Bronconeumol. 2000;36(2):84–89. [PubMed] [Google Scholar]
- 15.Thompson W.W., Shay D.K., Weintraub E., Brammer L., Cox N., Anderson L.J. Mortality associated with influenza and respiratory syncytial virus in the United States. J Am Med Assoc. 2003;289(2):179–186. doi: 10.1001/jama.289.2.179. [DOI] [PubMed] [Google Scholar]
- 16.Fleming D.M. The contribution of influenza to combined acute respiratory infections, hospital admissions, and deaths in winter. Commun Dis Public Health. 2000;3(1):32–38. [PubMed] [Google Scholar]
- 17.Menec V.H., Black C., MacWilliam L., Aoki F.Y. The impact of influenza-associated respiratory illnesses on hospitalizations, physician visits, emergency room visits, and mortality. Can J Public Health. 2003;94(1):59–63. doi: 10.1007/BF03405054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cohen G.M., Nettleman M.D. Economic impact of influenza vaccination in preschool children. Pediatrics. 2000;106(5):973–976. doi: 10.1542/peds.106.5.973. [DOI] [PubMed] [Google Scholar]
- 19.Luce B.R., Zangwill K.M., Palmer C.S., Mendelman P.M., Yan L., Wolff M.C. Cost-effectiveness analysis of an intranasal influenza vaccine for the prevention of influenza in healthy children. Pediatrics. 2001;108(2):E24. doi: 10.1542/peds.108.2.e24. [DOI] [PubMed] [Google Scholar]
- 20.Hurwitz E.S., Haber M., Chang A., Shope T., Teo S., Ginsberg M. Effectiveness of influenza vaccination of day care children in reducing influenza-related morbidity among household contacts. J Am Med Assoc. 2000;284(13):1677–1682. doi: 10.1001/jama.284.13.1677. [DOI] [PubMed] [Google Scholar]


