Abstract
Despite successful vaccination programs and effective treatments for some viral infections, humans are still losing the battle with viruses. Persisting human pandemics, emerging and re-emerging viruses, and evolution of drug-resistant strains impose continuous search for new antiviral drugs. A combination of detailed information about the molecular organization of viruses and progress in molecular biology and computer technologies has enabled rational antivirals design. Initial step in establishing efficacy of new antivirals is based on simple methods assessing inhibition of the intended target. We provide here an overview of biochemical and cell-based assays evaluating the activity of inhibitors of clinically important viruses.
Keywords: Virus, Inhibitor, Assay, High-throughput screening, Method, Replication, Entry, Assembly, In vitro, Cell-based
Highlights
-
•
This paper reviews methods for screening new compounds targeting clinically most relevant groups of viruses.
-
•
It covers also methods that have not been developed for antivirals screening, but are applicable for this purpose.
-
•
It covers targets of distinct groups of viruses including DNA, RNA and retroviruses.
1. Introduction
Viral pandemics remain serious threats to humankind. Every year, known viruses such as HIV-1 and hepatitis B virus newly infect millions of people across the globe. In addition, recent outbreaks of Ebola virus, influenza virus, severe acute respiratory syndrome (SARS) coronavirus and Middle East respiratory syndrome-coronavirus (MERS-CoV) serve as a reminder of the silent danger. The World Health Organization reported in 2015 that over 1.34 million causalities per year are connected with hepatitis B and almost 500,000 with hepatitis C. Occasional epidemic outbreaks of other viruses are also striking. These include outbreaks of noroviruses, flaviviruses (Zika and dengue viruses), new strains of influenza viruses, the re-emergence of West Nile virus in Italy and the United States. Despite relatively long pauses, viruses such as influenza virus can re-emerge and cause global pandemic health problems.
Unlike cellular genomes, which consist of double-stranded (ds) DNA, viral genomes can be formed by a broad variety of nucleic acids (NAs), including ds or single-stranded (ss) circular or linear DNA or positive-, negative- or sometimes ambi-sense RNA. As viral life cycles are dependent on cellular factors and cellular metabolic and signaling pathways, the number of possible antiviral drug targets is limited. However, almost all viruses encode unique proteins and enzymes that may serve as specific targets for antiviral therapy. The overarching goal of modern drug development efforts is to design compounds that specifically inhibit viral targets or cellular targets essential for virus replication.
The purpose of this review is to provide insight into the broad variety of cell-based and biochemical assays used for identification and evaluation of antivirotics, including high-throughput screening (HTS) methods. An overview of available inhibitors and vaccines, which has been reviewed elsewhere [e.g. in (De Clercq and Li, 2016)], is beyond the scope of this paper.
1.1. Morphology
To package their genomes into particles of very limited size, viruses encode few genes. Virions consist of an RNA or DNA genome that is protected by an outer shell called a capsid (also nucleocapsid) formed by a lattice of capsid proteins. In enveloped viruses, the capsid is additionally surrounded by a lipid bilayer spiked with viral proteins. The size of animal viruses ranges from approximately 25 nm to over 300 nm (Cohen et al., 2011). The capsids and virions of RNA viruses adopt various shapes. Viral capsids may be icosahedral (e.g. Picornaviridae, Astroviridae, Reoviridae, Togaviridae), bullet-shaped (Rhabdoviridae), helical (Coronaviridae), helical filamentous (Filoviridae, Orthomyxoviridae, Paramyxoviridae), or filamentous (Arenaviridae, Bunyaviridae). The retroviral capsid core may be conical, spherical, or rod-shaped.
The morphology of DNA viruses is similarly diverse, ranging from icosahedral (enveloped - Hepadnaviridae, Herpesvridae; nonenveloped - Adenoviridae, Parvovirdae, Polyomaviridae and Papillomaviridae) to rod-shaped (Baculoviridae) or pleomorphic (Poxviridae).
1.2. Structure
The viral life cycle is the process of viral replication in a host cell. First, viruses enter the host cell and replicate their genomes. Following translation of viral proteins by the host cell machinery, viruses package their genomic material into protective proteinaceous capsids and exit the cell to infect another host cell. Nonenveloped viruses consist only of a protein capsid shell enclosing the viral genome and enzymes, while the capsid shell of enveloped viruses is enclosed in a lipid envelope derived from the host cell membrane. Regardless of whether the virus is enveloped, its surface must display (glyco)proteins suitable for specific interactions with host cell receptors. In contrast to the surface proteins of nonenveloped viruses, those of enveloped viruses usually serve another function in addition to host cell recognition and attachment. For example, they may enable fusion of the viral and cellular membranes, usually through an interaction with a secondary receptor(s) or co-receptor(s). The fusion domains of viral surface glycoproteins thus can lower the kinetic barrier for the energetically demanding membrane fusion.
The viral particle must be sufficiently stable to protect the genome until its delivery into the host cell. Simultaneously, it must be metastable, to allow its disassembly and release of the genome for replication in the infected cell at the appropriate time. The energetic barrier that prevents viral disassembly outside the cell is lowered upon infection by structural changes in the viral components. These changes may be induced by binding of a cellular ligand or by changes in the environment, such as pH change upon entering a specific cellular compartment. Numerous viruses preassemble immature particles that undergo irreversible (usually proteolytic) transitions into mature structures of fully infectious virions. Mutual interactions of viral capsid proteins are typically different from those of cellular proteins, which predominantly create binary interactions. Viral structural proteins interact with multiple neighboring partners to form multicomponent macromolecular structures [reviewed in (Cheng and Brooks, 2015, Jayaraman et al., 2016)]. The economy of the packaged viral genomes due to the limited capsid size implies formation of only a few types of structural proteins, which are usually symmetrically organized [reviewed in (Prasad and Schmid, 2012, Raguram et al., 2017)].
2. Common stages in the viral life cycle
In the initial stage of infection, viruses must overcome several obstacles. The first is the cellular plasma membrane with an actin cortex. Then, they must traffic through dense cytoplasm to reach their final destination for replication and assembly. These pathways are specific for different viruses and often are dictated by the size of the particle and its structure.
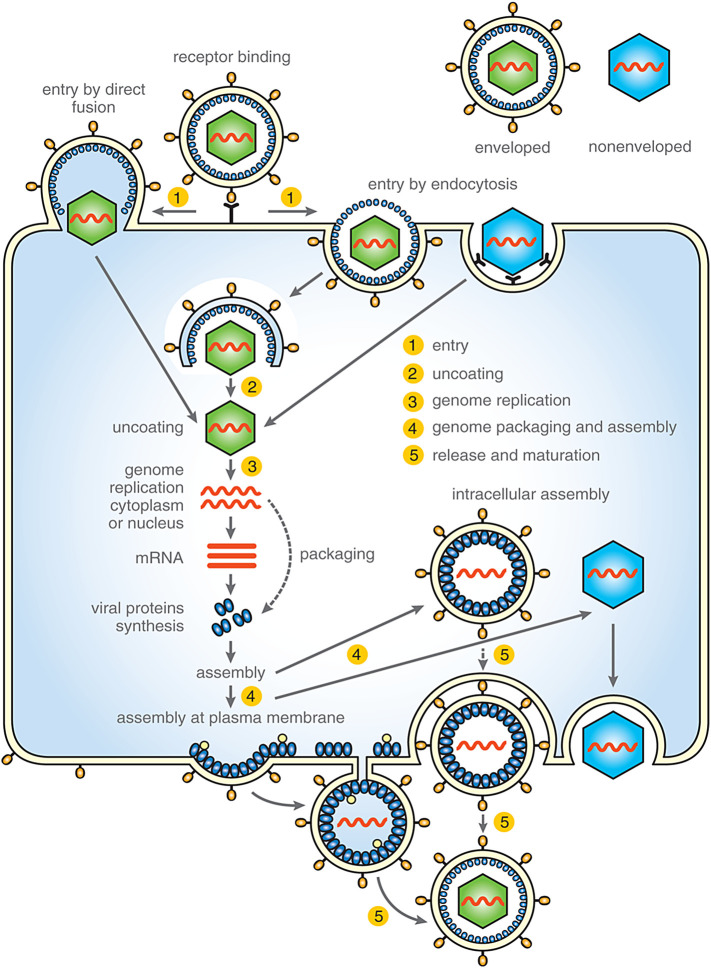
The viral life cycle can be divided into several common stages, including entry, uncoating, genome replication, genome packaging and assembly, release, and maturation (Fig. 1 ).
Fig. 1.
Simplified scheme of common stages of viral life cycle targeted by antiviral drugs. These stages including: 1) attachment and entry, 2) uncoating, 3) genome replication, 4) genome packaging and assembly of viral particle and 5) virus release and maturation.
2.1. Entry into the cell
In general, the first phase of viral infection is specific recognition of the target host cell and binding to a surface receptor displayed on the cell membrane. This process is common to both enveloped and nonenveloped viruses. In enveloped viruses, the binding is mediated by viral surface components, typically oligomers of integral glycoproteins. Nonenveloped viruses bind receptors through sites or projections on the capsid surface. Viruses can use either a single receptor (e.g. TIM-1 for hepatitis A virus, GM1 for SV40, CD155 for poliovirus, low-density lipoprotein receptor for human rhinovirus) or multiple receptors with equivalent roles (e.g., Nectin-1/2 or HVEM for herpes simplex virus 1/2, ACE or L-SIGN for SARS coronavirus). For other viruses (e.g. HIV, HCV, adenoviruses, rotaviruses, picornaviruses, and some herpesviruses), the presence of at least two cytoplasmic membrane components is required. For example, HIV-1 binds to the primary receptor CD4 and one of two co-receptors (CCR5 or CXCR4) [reviewed in (Cossart and Helenius, 2014, Grove and Marsh, 2011)].
To infect a cell, viruses must overcome the plasma membrane to deliver their genetic material into the cytosol. Viruses enter cells by two main mechanisms. A majority of animal viruses, both enveloped and nonenveloped, enter cells by one or two types of endocytosis, such as clathrin-mediated endocytosis (e.g. VSV, influenza A virus, rhinovirus), caveolin-mediated uptake (echovirus, polyoma virus), clathrin/caveolin-independent endocytosis such as caveolar or lipid raft-mediated (e.g. SV40, polyomavirus mouse), or macropinocytosis (e.g. vaccinia virus, respiratory syncytial virus, Ebola virus, HIV-1) (Blaas, 2016, Cossart and Helenius, 2014, Fields et al., 2013, Kirkham and Parton, 2005, Marechal et al., 2001, Mayor and Pagano, 2007, Mercer and Helenius, 2009, Parton and Simons, 2007, Rasmussen and Vilhardt, 2015, Saeed et al., 2010). These endocytic mechanisms enable the virus to be transported by the cell’s machinery through the plasma membrane and to pass through the dense actin cortex. Cellular entry of some viruses is coupled with receptor-mediated signaling, resulting in activation of molecules that facilitate virus entry by cytoskeleton reorganization, induction of long-distance transport of the virus-containing vesicles, or actin cortex disassembly. The second type of entry is used by some enveloped viruses (paramyxo-, herpes-, and retroviruses), which upon cell surface receptor binding, infect the cell by direct fusion of viral and plasma membranes at neutral pH.
Upon internalization, most viruses become trapped and enclosed in vesicles that pinch off the inner side of the plasma membrane and transport their cargo into the cytoplasm. On their way through the cell, these transport vesicles undergo maturation by fusing with other vesicles. To fulfill their task of genome replication, viruses have to escape from these endosomes. The “great escape” is triggered by activation of a fusion or penetration mechanism, such as changes in conditions in the endosomal interior [e.g. pH, ionic environment (e.g. calcium ion concentration), oxido-reducing conditions], changes in membrane composition, and other physico-chemical cues (Blaas, 2016, Cossart and Helenius, 2014, Inoue and Tsai, 2013). Depending on the requirements for a particular virus, these events can occur in early endosomes (pH 6.5–6.0; e.g. hepatitis C virus, vesicular stomatitis virus), late endosomes (pH 6.0–5.0; e.g. influenza A virus, dengue virus, SARS coronavirus), recycling endosomes, macropinosomes, the endoplasmic reticulum, the Golgi apparatus, or lysosomes (pH 5.0–4.5) (Blaas, 2016, Cossart and Helenius, 2014, Grove and Marsh, 2011, Inoue and Tsai, 2013). For the majority of animal viruses, the activation of these fusion or penetration mechanisms occurs through conformational changes and structural rearrangements in viral surface proteins and/or the whole virion shell that may destabilize the capsid core. Structural rearrangements in enveloped viruses usually mediate fusion of viral and endosomal membranes. In nonenveloped viruses, the structural changes uncover amphipathic or hydrophobic domains that may induce pore formation or disruption of endosomal membranes. To deliver genetic material to the replication site, these mechanisms ultimately release viral capsid structures from endosomal vesicles into the cytosol either by fusing with the endosomal membrane (enveloped viruses) or by penetrating the endosomal membrane (nonenveloped viruses).
2.2. Uncoating
Uncoating is the partial or complete disassembly of the protective capsid shell and/or lipid envelope to liberate the viral genome. For many viruses, this process is closely connected with conformational changes induced by the virus binding to the cell surface receptor; a low pH environment; or changes in oxido-reducing conditions, ion concentrations, or other factors. For enveloped viruses, uncoating involves a loss of the viral membrane by fusion either with the plasma membrane or with intracellular vesicles, followed by stepwise uncoating of the protective capsid shell. In nonenveloped viruses, the uncoating process typically involves conformational changes that result in the weakening of intermolecular interactions, loss of structural proteins, proteolytic cleavage, and so on (Fields and Knipe, 2013).
Depending on the virus, uncoating can take place at the plasma membrane, in the cytoplasm, during endocytosis in early or late endosomes, in lysosomes, in the nucleus, or at the nuclear pore complex (NPC).
The ssRNA genome of retroviruses, which is fully enclosed inside a protective capsid shell, must be reverse transcribed into dsDNA and released from the capsid. Although it is accepted that HIV-1 uncoating is linked to reverse transcription and nuclear import (Ambrose and Aiken, 2014) and is controlled by host factors (e.g. cyclophilin A, TRIM5α), the precise molecular mechanisms that trigger the uncoating remain unknown. Several hypotheses have been proposed, including breakage of the capsid shell due to increased inner pressure caused by accumulation of the reverse transcription product (Rankovic et al., 2017), the requirement of intact microtubules and dynein and kinesin motors (Lukic et al., 2014), and phosphorylation of capsid shell protein by the host cell kinase MELK (Takeuchi et al., 2017).
2.3. Entry into the nucleus
In contrast to the majority of RNA viruses, which replicate in the cytoplasm, most DNA viruses (with the exception of large DNA viruses including Poxviridae, Asfarviridae, and Mimiviridae) and several negative stranded RNA viruses enter the nucleus to replicate their genomes (Kobiler et al., 2012, Koonin and Yutin, 2010). Passive diffusion into the nucleus is suitable for molecules smaller than 9 nm in diameter, but larger structures must enter through the nuclear pore complex (NPC), which can accommodate molecules of up to 39 nm (Pante and Kann, 2002). Translocation through the NPC is tightly regulated and requires the presence of a nuclear localization signal (NLS) on the passing molecule and nuclear import receptors (importins or karyopherins) (Cautain et al., 2015).
Due to the diversity of viral particle sizes (from 25 nm to over 300 nm) and structures, viruses have evolved several strategies to export their DNA or RNA genome into the nucleoplasm. With regard to the NPC, these pathways can be divided into NPC-dependent and NPC-independent mechanisms. Due to size limitations, only a few, very small viral capsid particles, such as hepatitis B virus (HBV; 32–36 nm diameter), can pass through the NPC (Cohen et al., 2011, Rabe et al., 2003) in an importin-dependent manner. HBV then disassembles at the nuclear side of the NPC (the nuclear basket), releasing its genome into the nucleoplasm (Fay and Panté, 2015). The capsid shells or ribonucleoprotein complexes (RNPs) of some larger viruses, such as influenza A virus and HIV-1, disassemble within the cytoplasm. The viral genome, released from the shell and complexed with NLS-containing components, is then translocated through the NPC (Ambrose and Aiken, 2014, Campbell and Hope, 2015, Cohen et al., 2011, Fay and Panté, 2015, Hutchinson and Fodor, 2013). Another mechanism, used by herpes simplex virus (HSV) and adenoviruses, involves cellular (importins, Nup) or viral protein-mediated attachment (docking) of the capsid shell at the cytoplasmic side of the NPC. This facilitates the passage of genomic DNA into the NPC either by ejection from an almost intact particle (through the capsid portal in HSV-1) or upon complete disassembly of the capsid shell (in adenoviruses) (Kremer and Nemerow, 2015, Ojala et al., 2000, Pasdeloup et al., 2009). NPC-independent mechanisms are used by some retroviruses (e.g. MLV) that enter the nucleus during the mitotic phase of the cell division cycle when the nuclear envelope is dissolved (Matreyek and Engelman, 2013, Roe et al., 1993). Another mechanism, described for parvoviruses, involves partial disruption of the nuclear envelope (Cohen and Pante, 2005, Fay and Panté, 2015).
2.4. Replication
Viral genomes can be encoded by various types of NAs, as summarized in Table 1 for DNA viruses and Table 2 for RNA viruses. By convention, ssDNA of equivalent polarity to mRNA is designated as the positive (+) strand. The complementary ssNAs are of minus polarity (−). The majority of DNA viruses replicate in the nucleus, where cellular DNA replication and transcription also occur. In contrast, RNA viruses usually replicate in the cytoplasm.
Table 1.
Basic characteristics of DNA viruses.
| DNA viruses |
Genom |
Replication |
||
|---|---|---|---|---|
| Virus family | DNA type/topology | Polarity | Enzyme | Intracellular site |
| Adenoviridae | ds/linear | Both | Viral DNA polymerase | Nucleus |
| Anelloviridae | ss/circular | (−) | Cellular DNA polymerase | Nucleus |
| Asfarviridae | ds/linear | Both | Viral DNA polymerase | Cytoplasm |
| Circoviridae | ss/circular | (−) or ambisense | Cellular DNA polymerase | Nucleus |
| Hepadnaviridae | ds/linear | Both | Viral reverse transcriptase | Nucleus/cytoplasm |
| Herpesviridae | ds/linear | Both | Viral DNA polymerase | Nucleus |
| Iridoviridae | ds/linear | Both | Viral DNA polymerase | Nucleus/cytoplasm |
| Papillomaviridae | ds/circular | Both | Cellular DNA polymerase | Nucleus |
| Parvoviridae | ss/linear | Either | Cellular DNA polymerase | Nucleus |
| Polyomaviridae | ds/circular | Both | Cellular DNA polymerase | Nucleus |
| Poxviridae | ds/linear | Both | Viral DNA polymerase | Cytoplasm |
Table 2.
Basic characteristics of RNA viruses.
| RNA viruses |
Genom |
Replication |
||
|---|---|---|---|---|
| Virus family | RNA type/segment/topology | Polarity | Enzyme | Intracellular site |
| Arenaviridae | ssRNA/2/linear | Ambisense | Virion RNA polymerase | Cytoplasm |
| Arteriviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Astroviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Birnaviridae | dsRNA/2/linear | both | Virion RNA polymerase | Cytoplasm |
| Bornaviridae | ssRNA/1/linear | (−) | Virion RNA polymerase | Nucleus |
| Bunyaviridae | ssRNA/3/linear | (−) or ambisense | Virion RNA polymerase | Cytoplasm |
| Caliciviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Coronaviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Deltavirus | ssRNA/1/circular | (−) | RNA polymerase II | Nucleus |
| Filoviridae | ssRNA/1/linear | (−) | Virion RNA polymerase | Cytoplasm |
| Flaviviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Hepeviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Nodaviridae | ssRNA/2/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Orthomyxoviridae | ssRNA/6-8/linear | (−) | Virion RNA polymerase | Nucleus |
| Paramyxoviridae | ssRNA/1/linear | (−) | Virion RNA polymerase | Cytoplasm |
| Picornaviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
| Reoviridae | dsRNA/10-12/linear | both | Virion RNA polymerase | Cytoplasm |
| Retroviridae | ssRNA/2/linear | (+) | Virion reverse transcriptase | Nucleus/cytoplasm |
| Rhabdoviridae | ssRNA/1/linear | (−) | Virion RNA polymerase | Cytoplasm |
| Togaviridae | ssRNA/1/linear | (+) | Viral RNA polymerase | Cytoplasm |
2.4.1. Replication of RNA viruses
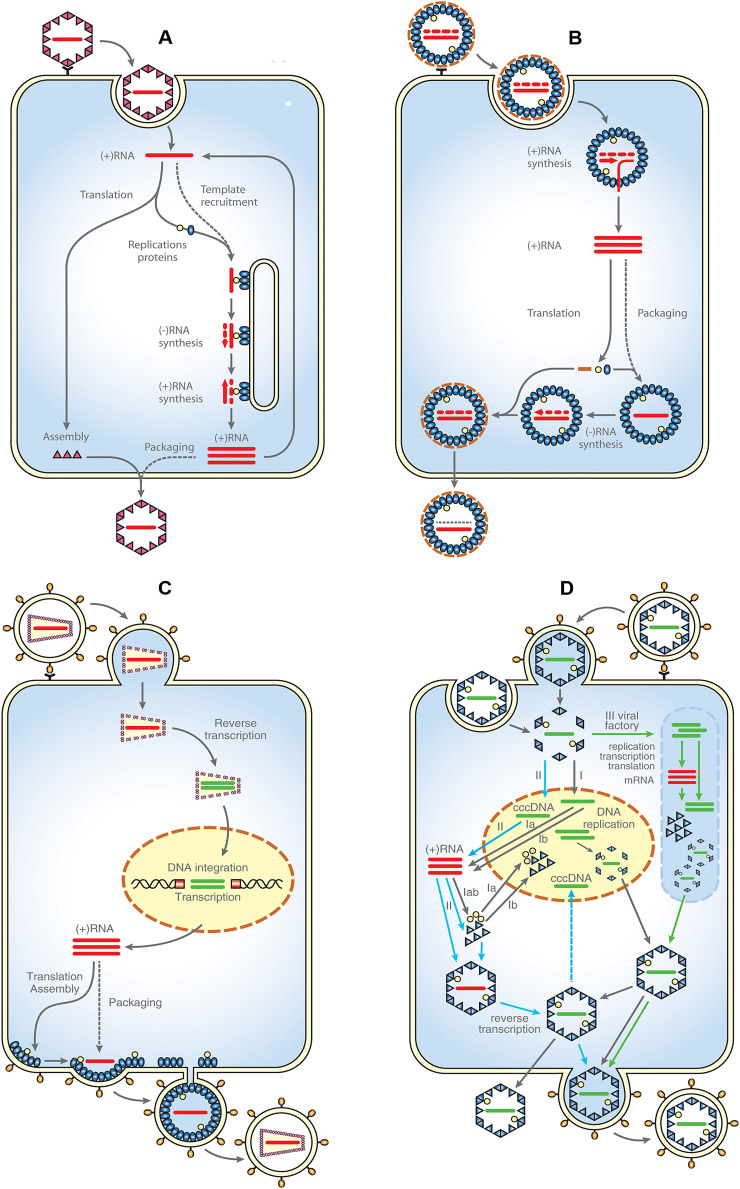
RNA viruses are the only “organisms” that store their genetic information in the form of RNA. Replication of their genomes is accomplished either by RNA-dependent RNA synthesis (Fig. 2A, B) [reviewed in (Ferron et al., 2017, Lu and Gong, 2017, Menéndez-Arias and Andino, 2017, Pietilä et al., 2017, Tao and Ye, 2010)] or through RNA-dependent DNA synthesis (reverse transcription) followed by DNA integration, replication, and transcription (Fig. 2C). As some of these enzymatic activities are not commonly found in uninfected host cells, RNA viruses must encode enzymes to aid replication (Table 2). RNA viruses are divided according to the Baltimore classification into dsRNA viruses (Birna- and Reoviridae); positive-sense ssRNA viruses (Corona-, Flavi-, and Picornaviriade); negative-sense ssRNA viruses (Filo-, Rhabdo-, Paramyxo-, and Orthomyxoviriade), and ambisense RNA viruses (Arena- and some members of Bunyaviridae) with both positive- and negative-sense RNAs (Nguyen and Haenni, 2003) (see Table 2).
Fig. 2.
Simplified scheme of replication of ss (+)RNA viruses (A), dsRNA and (-)RNA viruses (B), retroviruses (C) and DNA viruses (D) (the schemes A-C were modified from: (Ahlquist, 2006).
A: Following endocytosis, the genome of (+)RNA viruses may directly serve as mRNA for translation of virus encoded proteins. Among them, there are proteins of RNA-replication machinery that recruit (+)RNA into a membrane-associated replication complex. The genomic RNA is replicated by using (–)RNA template, which is transcribed in a low copy number Amplified (+)RNA is then packaged into newly assembled virions that exit the host cell either through secretion or cell lysis.
B: Upon the virus attachment, a core containing both gRNA and virus encoded RNA polymerase is delivered into the cytoplasm by endocytosis. Cytoplasmic transcription of the (–)RNA template provides (+)RNA that serves as mRNA for translation of viral proteins. In dsRNA viruses, the (+)RNA is packaged into new cores which undergo maturation by synthesizing (–)RNA (dotted strand) and acquiring surface proteins. In the (–)RNA viruses, the (+)RNA strand is transcribed into genomic (–)RNA in the cytoplasm and then packaged. The new virions egress the cell either through secretion or cell lysis.
C: Following fusion of viral and cell plasma membrane, the retroviral core is released into cytosol, RNA genome is transcribed into dsDNA by viral reverse transcriptase concomitantly with uncoating of the viral core, ds DNA is transferred into nucleus where it is integrated into host cell genome by viral integrase, following translation of retroviral structural and enzymatic polyproteins, the unspliced gRNA is packaged into the immature particles that usually assemble at the plasma membrane and the viral particles bud from the cell.
D: Three mechanisms (I.-III.) of DNA viruses replication are shown: (I): Following entry and uncoating, the DNA genome is transported to the nucleus; products of early genes (regulatory proteins, transcription factors) regulate the synthesis of viral enzymes (e.g. DNA polymerase) required for genome replication; expression of late genes encoding structural capsid proteins in the cytosol, they are then transported into nucleus where packaging and pre-assembly take place; preassembled procapsids exit the nucleus and leave the cell (e.g. Herpesviruses). (II) unique replication cycle of Hepatitis B virus (HBV): following entry, the viral particle is internalized by endocytosis and the nucleocapsid is released into the cytoplasm; the genome (relaxed circular rcDNA) is transported into the nucleus, where it is converted to a covalently closed circular form (cccDNA); which serves as a template for transcription of pregenomic RNA (pgRNA) for translation of the core protein and the viral polymerase and three subgenomic mRNAs used for translation of regulatory and envelope proteins; following viral transcription and translation, the HBV core proteins self-assemble in the cytoplasm into viral nucleocapsid with concurrent incorporation of pgRNA and HBV polymerase, pgRNA is reversely transcribed into a rcDNA within the nucleocapsid; nucleocapsid containing rcDNA can either re-enter the nucleus to amplify cccDNA, or can be enveloped by HBV envelope proteins in the endoplasmic reticulum. The particles are then secreted from the infected hepatocyte by exocytosis. (III) Upon entering the cell, the replication, transcription and translation take place entirely in the cytoplasm, within discrete juxtanuclear sites called virus factories (e.g. poxviruses)
Adapted from: Ahlquist, P., 2006. Parallels among positive-strand RNA viruses, reverse-transcribing viruses and double-stranded RNA viruses. Nat Rev Microbiol 4(5), 371-382.
The nature of the genome not only dictates the mechanism of replication, but also has other important consequences. The genome of (+)RNA viruses may serve directly as mRNA for production of viral proteins. Therefore, the mere introduction of genetic material (e.g. in exocytic vesicles) may result in productive infection. The RNA polymerases that copy the genetic material of RNA viruses are error-prone, which provides considerable genetic flexibility and the propensity to evolve drug-resistant mutants. These features are amplified in viruses with segmented genomes that undergo reassortment.
In viruses with segmented genomes (Orthomyxoviridae, Arenaviridae and Bunyaviridae), each segment is transcribed in an autonomous transcription-replication unit by viral RNA polymerase that binds to the 5′ end cap structure. Of note, the genomic RNA (gRNA) is capped by a unique mechanism called cap-snatching (Ferron et al., 2017), in which the cap is cleaved from cellular mRNA and transferred onto the viral gRNA by a subunit of viral RNA polymerase (Pflug et al., 2017). Some dsRNA viruses (Birnaviridae and Reoviridae) also contain segmented genomes. Upon replication, these viruses must ensure stoichiometric incorporation of single copies of each gRNA segment into new particles. This is guided by specific packaging signals on each segment of gRNA that interact with positively charged domains in the capsid proteins [reviewed in (Isel et al., 2016, Pohl et al., 2016)]. Although different models of packaging have been proposed for various segmented genome viruses, a common feature is co-assembly of the capsid proteins with the gRNA and RNA polymerase.
2.4.2. Replication of DNA viruses
The genomes of DNA viruses come in a considerable variety of sizes and shapes, from small ss to large ds molecules that may be linear or circular. The size range of these genomes (from 1.8 kb to 1200 kb) reflects the necessity for some viruses to encode specific proteins required for viral replication. Small-genome DNA viruses (polyoma-, papilloma-, and parvoviruses) use only host cell enzymes for replication and transcription. The only exceptions are some hepadnaviruses (e.g. hepatitis B virus) that despite having small genomes (approximately 3 kb), encode their own specific DNA polymerase/reverse transcriptase that reverse transcribes pregenomic RNA (pgRNA) into genomic viral DNA (Fig. 2D, II) (Beck and Nassal, 2007).
Viruses with intermediate-size genomes (up to 35 kb) (e.g. adenoviruses) encode their own DNA polymerase for genome replication, but they usually utilize cellular RNA polymerases II and III for transcription (Fields and Knipe, 2013). Viruses with large genomes (larger than 100 kb) (e.g. herpesviruses) encode proteins for replication, including DNA polymerase and DNA helicase-primase, as well as some enzymes necessary for biosynthesis of deoxyribonucleotide triphosphates (dNTPs) and several transcription factors (Boehmer and Nimonkar, 2003). Poxviruses (e.g. vaccinia virus), are another type of virus with large dsDNA genomes, and their replication, transcription, and translation take place entirely in the cytoplasm within discrete juxtanuclear sites called virus factories (Moss, 2013), (Fig. 2D, III). These viruses encode all enzymes and specific factors necessary for genomic replication and transcription. With their own replicative machinery, large-genome DNA viruses can replicate in nondividing cells. In contrast, the replication of small-genome DNA viruses, which depends on cellular DNA polymerases, must occur simultaneously with the S-phase of the cell cycle (e.g. parvoviruses), or must express some viral protein/oncogene to re-program the host cell-cycle regulatory proteins p53 or retinoblastoma protein (pRb), triggering entry into the S-phase (e.g. polyomaviruses and papillomaviruses). By affecting the G1/S checkpoint (controlled by p53 and pRb), the viruses ensure the production of host enzymes required for viral replication (Fields and Knipe, 2013).
Production of viral proteins of DNA viruses with intermediate and large size genomes is divided into early and late phases. In the early (prereplicative) phase, nonstructural proteins required for DNA replication are translated. The late phase, during which the late structural proteins needed for assembly are translated, begins after viral DNA replication (Fig. 2D, I).
2.5. Assembly
Depending on the species, viruses assemble either in contact with the cellular membrane or independent of the membrane in either the nucleus or cytoplasm. The membrane-independent route is used by nonenveloped viruses and a few enveloped viruses (e.g. orthomyxoviruses, herpesviruses, and some retroviruses) that acquire the membrane envelope after intracellular assembly during budding from the cell.
2.5.1. Assembly in the nucleus
The limiting step for nuclear assembly is the size of NPCs, which are large enough to transport RNA and import proteins required for assembly into the nucleus; however, NPC size limits the transport of larger assemblies. Therefore, some viruses form assembly intermediates in the nucleus. These structures are then exported to the cytoplasm, where they come together to form viral particles. Nuclear export is specific and depends on the presence of nuclear export signals (NES) in the transported proteins. One example of a nonenveloped icosahedral virus that assembles in the nucleus is adenovirus, the assembly of which has been studied intensely due to its potential use in gene therapies [reviewed in (Ahi and Mittal, 2016)]. Recent data suggest that upon accumulation of multiple copies of adenoviral dsDNA genomes, coordinated assembly and packaging occur by two interlinked mechanisms that involve both capsid proteins and core components (Condezo and San Martín, 2017). The assembly occurs in the so-called peripheral replicative zone with the assistance of scaffolding proteins that facilitate formation of adenoviral particles but are excluded from mature viruses. Adenoviruses are finally released upon lysis of the infected host cell.
Orthomyxoviruses and herpesviruses are enveloped viruses that assemble their nucleoproteins in the nucleus. Herpesviruses package their dsDNA genome as head-to-tail concatemers and assemble icosahedral procapsids on scaffold proteins in the nucleus [reviewed in (Heming et al., 2017)]. However, subsequent steps of herpesvirus assembly proceed in the cytoplasm. The preassembled procapsid is too large to pass through the NPC, but it exits the nucleus by viral protein-driven vesicular transport across the nuclear inner membrane leaflet. Thus, the herpesvirus acquires an initial envelope from the inner nuclear membrane [for review see (Fields et al., 2013, Hellberg et al., 2016)]. Next, the herpesviral membrane fuses with the outer nuclear membrane, and the naked particle is released from the nucleus into the cytosol. Here, the virus acquires tegument (a protein layer between the capsid and the envelope) and other proteins. Final herpesvirus envelopment occurs at the Golgi membrane containing the viral glycoproteins (Fields and Knipe, 2013). Preassembled orthomyxoviral ribonucleoproteins, upon their export from the nucleus, are driven to the plasma membrane to which they attach through electrostatic interactions of the matrix protein M1 with membrane phosphatidylserine. The virions then assemble simultaneously with budding during which they also acquire HA, NA and M2 membrane proteins. Poxviruses undergo an even more complicated pathway. They are enveloped with multiple membranes acquired from ER/intermediate compartments and Golgi or early endosomes (Moss, 2015, Moss, 2016). These membranes also provide the poxviruses with their membrane proteins.
2.5.2. Assembly in the cytoplasm
Most viruses assemble upon interaction of their structural proteins with cellular membranes. The target membrane for assembly differs according to the virus type. Flaviviruses assemble at the surface of the ER and then upon their budding into the ER lumen, the immature particles are then transported into the TGN. Some viruses, such as coronaviruses, assemble at the ER–Golgi intermediate compartment [reviewed in (Ujike and Taguchi, 2015)]. The assembly of bunyaviruses occurs concurrently with replication of gRNA segments in virus factories located along the Golgi (Guu et al., 2012, Strandin et al., 2013). The presence of membrane glycoproteins at the Golgi membrane determines the sites where assembling bunyavirus particles bud into the Golgi lumen, similar to other enveloped viruses.
Numerous viruses, including paramyxoviruses (Cox and Plemper, 2017), orthomyxoviruses (Pohl et al., 2016), alphaviruses (Jose et al., 2009), rhabdoviruses (Okumura and Harty, 2011), and most retroviruses (Freed, 2015), assemble underneath the cytoplasmic membrane, which facilitates assembly by providing a scaffolding function. The virus then acquires a lipid envelope through budding across the plasma membrane. During this process, it also gains the envelope glycoproteins (Env) that are anchored in the plasma membrane by hydrophobic transmembrane domains. Env reaches the plasma membrane by a cellular secretory pathway upon synthesis in the ER and subsequent glycosylation in the Golgi. Usually, a specific interaction between viral structural proteins and glycoproteins is required. This may be either direct or mediated through the interaction with matrix protein (e.g. in (–)RNA viruses such as ortho- and paramyxoviruses or rhabdoviruses). Most retroviruses, including HIV (so-called morphogenetic C-type), also assemble at the plasma membrane of the host cell. The interaction of the structural polyprotein precursor (Gag) with the plasma membrane is usually facilitated by a bipartite signal in the N-terminal domain of Gag (i.e. matrix protein) comprising both the basic patch and N-terminally linked myristoyl residue (added co-translationally to Gag). In contrast to C-type assembly at the membrane, morphogenetic B/D-type retroviruses assemble at pericentriolar sites where Gag polyproteins are transported along microtubules by dynein molecular motors (Sfakianos et al., 2003, Vlach et al., 2008). For both morphogenetic pathways, the packaging of gRNA facilitates assembly.
(+)RNA viruses have adopted a mechanism of extensive rearrangement of intracellular membranes to provide a milieu for virus replication and assembly. This mechanism effectively protects dsRNA intermediates from degradation by the host cell machinery (Delgui and Colombo, 2017, Jackson, 2014). This process has been well-documented for poliovirus, in which the newly formed membranous structures exclusively serve for virus production. Various types of vesicles that are formed upon viral infection have different roles in viral replication (Rossignol et al., 2015).
Some nonenveloped mammalian viruses, such as reoviruses, assemble in the cytosol in so-called viroplasms or viral factories into icosahedral particles consisting of three concentric layers encircling segments of genomic dsDNA (Benavente and Martinez-Costas, 2006, Jones, 2000, Shah et al., 2017). Rotavirus seems to be the only exemption in the reovirus family, as it enters the endoplasmic reticulum to gain its outer protein shell (Trask et al., 2012).
2.6. Maturation
Numerous viruses assemble from polyprotein precursors that must be specifically cleaved by a viral protease to generate infectious particles. This mechanism, which is an irreversible step in the virus life cycle, ensures equimolar packaging of structural proteins and proportional co-assembly of viral enzymes in the form of precursors. Upon proteolytic release, the liberated proteins may undergo different trafficking pathways or fulfill various functions in the virus. Maturation changes the energy of interaction forces among those interfaces required for intracellular assembly of immature particles to those suitable for viral stability in the environment and for disassembly and uncoating of genetic material for replication [reviewed in (Veesler and Johnson, 2012)].
In poliovirus, the autocatalytic and subsequent viral protease-mediated cleavage of P1 precursor protein allows formation of pentamers that interact with gRNA. Additional cleavage of VP0, yielding VP2 and VP4, is required to form infectious poliovirus particles. In retroviruses, proteolytic processing is initiated by the autocatalytic liberation of viral protease, which subsequently cleaves the polyproteins to trigger major structural rearrangements in the virus and release of other viral enzymes (reverse transcriptase and integrase) and structural proteins.
In herpesviruses, maturation involves proteolytic processing of the scaffolding protein and recruitment of tegument proteins that stabilize the particle and mediate interactions with the membrane during envelopment (Gibson, 2008, Tandon and Mocarski, 2012, Tandon et al., 2015).
Adenoviruses undergo a maturation process that involves processing of six structural components of the core and one non-structural precursor that initiates replication of gDNA (Gaba et al., 2017). One interesting feature of adenoviral maturation is that DNA is required as a co-factor for protease activity. This means that maturation occurs only in particles that have packaged gDNA (Mangel and San Martín, 2014).
Flaviviruses form icosahedral particles upon budding into the neutral milieu of the ER. The particles then translocate to the more acidic environment of the trans-Golgi network. This pH shift results in disassembly of the immature lattice and extensive rearrangement of the flaviviral particle. This is connected with the exposure of a viral structural glycoprotein (prM) that is specifically processed by the cellular protease furin in the trans-Golgi network. The liberated globular heads of prM remain attached at the low pH, but are released when the virus enters neutral body fluids (Rey et al., 2017).
2.7. Release
One frequently used mechanism of release of nonenveloped and some enveloped viruses is lysis of the infected cell. This is the simplest release mechanism, although the transition to lysis from latent infection is delicately regulated (Aneja and Yuan, 2017, Levings and Roth, 2013, Schmiedel et al., 2016). However, viruses that are usually lysogenic may also use alternative release pathways (Bird and Kirkegaard, 2015a). These include non-lytic spread of viruses mediated by vesicles, which has been observed for poliovirus (Bird and Kirkegaard, 2015b, Jackson et al., 2005), coxsackievirus (Alirezaei et al., 2012), and hepatitis A virus (Feng et al., 2013). Another possibility is that vesicles released from a cell infected with (+)RNA viruses contain naked viral gRNA. This (+)gRNA-containing vesicle functions itself as an infectious agent as it is transferred to another cell (Bird and Kirkegaard, 2015a).
The standard way for enveloped viruses to leave the host cell is budding, which includes membrane extrusion and separation of the viral and cellular membranes (so-called pinching off). The lipid envelope layer acquired during viral particle budding through the plasma membrane protects the virus particle. There are three basic mechanisms of budding: i) mediated by envelope glycoproteins, such as the E protein of coronaviruses; ii) independent of glycoproteins, in which the viral structural proteins trigger the extrusion of the plasma membrane, such as retroviral budding in which strong interactions between the Gag N-terminal domain (matrix protein) and the plasma membrane induce bulging of the membrane; and iii) requiring interaction between the viral glycoproteins and the capsid for membrane extrusion, as in alphaviruses.
The final step that results in the separation of virus from the cell surface is pinching off the particle. This is a controlled process that involves both viral protein domains and cellular factors. During this process, viruses apparently use cellular machinery similar to that used during the last step of cell division (the release of the daughter cell) called endosomal sorting complex required for transport (ESCRT) [reviewed in (Campsteijn et al., 2016, Hurley, 2015, Olmos and Carlton, 2016, Scourfield and Martin-Serrano, 2017)]. Direct interactions of numerous viral domains with ESCRT complex subunits have been identified (Bieniasz, 2006). In retroviruses, short specific amino acid sequences (PTAP or PSAP) interact with the ESCRT components, and the interaction of HIV with the ESCRT proteins Nedd4 and Alix is well-known (Freed, 2002, Fujii et al., 2007, Gomez and Hope, 2005). However, this mechanism has also been adopted by other viruses that interact with the same ESCRT components, such as filoviruses (Han et al., 2015, Jasenosky and Kawaoka, 2004, Liu and Harty, 2010). Interactions with ESCRT proteins have also been reported for vesicular stomatitis virus (Chen et al., 2010, Luyet et al., 2008), rhabdovirus (Taylor et al., 2007), arenaviruses (Ziegler et al., 2016), picornaviruses (Feng et al., 2013), paramyxoviruses (Park et al., 2016), and hepatitis C virus, a representative of the flavivirus family (Falcon et al., 2017).
The typical release pathways used by viruses may vary under some conditions. For example, during chronic infection, numerous viruses use alternative cell-to-cell transmission that may help the virus avoid host neutralization (Hulo et al., 2017). Syncytium formation, an HIV-induced cell fusion, was recognized in the early 1990s (Callahan, 1994). Another type of cell-to-cell transmission through tight junctions was shown for HIV (Hübner et al., 2009, Wang et al., 2017) and murine leukemia virus (Sherer et al., 2010). Receptors on tight junctions that specifically recognize hepatitis C virus (Carloni et al., 2012, Ploss et al., 2009) and reovirus (Barton et al., 2001) have been identified. Some viruses are able to modify cellular pathways to reprogram both the synthesis and metabolism of lipids and membrane compartmentalization for their transmission and to evade cellular defense mechanisms (Mazzon and Mercer, 2014). Despite a general understanding that poliovirus spreads through cellular lysis, it was recently found that it may also be transferred between cells by an autophagy-dependent mechanism, called autophagosome-mediated exit without lysis (Bird and Kirkegaard, 2015b, Bird et al., 2014, Lai et al., 2016, Richards and Jackson, 2012). Similar mechanisms have been described for varicella-zoster virus and human cytomegalovirus (Grose et al., 2016, Meier and Grose, 2017). Poxviruses encode several proteins that block the apoptotic cellular response to the presence of their dsDNA in the cytoplasm. This allows cell-to-cell passage of poxviruses by a mechanism known as apoptotic mimicry (Amara and Mercer, 2015, Nichols et al., 2017). In this process, enveloped viruses expose surface phosphatidylserine to mimic apoptotic bodies. These cells are then macropinocytosed by either dendritic cells or macrophages (Mercer and Helenius, 2010).
3. Methods
Among the plethora of compounds designed to inhibit infectious viruses, only a few (< 100) have been approved for clinical use (De Clercq, 2004, De Clercq and Li, 2016). Nevertheless, some effective antiviral drugs have been on the market for several decades, such as the anti-influenza A virus drug amantadine, marketed under the trade name Symmetrel (by Dupont), which was approved in 1966. In 1982, Burroughs-Wellcome introduced acyclovir as an inhibitor of herpesviruses. Its remarkable specificity is connected with its activation by viral thymidine kinase-catalyzed phosphorylation. However, due to development of drug resistance in a number of viruses, especially RNA viruses, there is a continuous need to design and test new inhibitors, preferably targeting different steps of viral life cycles. Here, we provide insights into the world of biochemical and cell-based assays that were developed to test antivirotics targeting various steps in the viral life cycle.
3.1. Assays and methods for screening and evaluating viral entry inhibitors
Different types of assays, including cell-cell fusion assays, cell-virus fusion assays with pseudotyped viral particles, and in vitro biochemical assays have been developed to screen inhibitors of viral entry. In enveloped viruses, entry is initiated by fusion of the viral envelope with the target cellular membrane. The entry mechanism has been well-described for HIV-1, in which it is mediated by Env glycoprotein, consisting of transmembrane gp41 and surface gp120 subunits. Binding of gp120 to its cellular receptor, CD4, and to one of two co-receptors, CXCR4 or CCR5, triggers a refolding of gp41 that promotes fusion of the viral and cellular membranes. The refolding involves oligomerization of the extracellular N- and C-terminal heptad repeat (HR) domains of gp41, which leads to the formation of a 6-helical bundle [reviewed in (Melikyan, 2008)].
Jiang et al. established an in vitro system to quantify formation of the HIV-1 gp41 6-helical bundle (Jiang et al., 1999). In their system, the bundle is formed by mixing equimolar concentrations of peptides derived from the N- and C-HR regions of gp41. ELISA using the monoclonal antibody NC-1, which specifically recognizes and binds an epitope formed on the gp41 6-helix bundle but not the individual peptides, enables screening for compounds that prevent formation of fusion-active gp41. For HTS of HIV-1 fusion inhibitors, this method was modified to use a fluorescence-linked immunosorbent assay (FLISA), in which the C-HR peptide was replaced with FITC-conjugated C-HR peptide (Boyer-Chatenet et al., 2003).
Cell-based assays are routinely used to screen viral entry inhibitors. High throughput formats have been developed for screening of HIV-1 fusion inhibitors. Cell-cell fusion assays involve two types of cells: effector cells that stably (Bradley et al., 2004) or inducibly (Herschhorn et al., 2011, Ji et al., 2006) express HIV-Env glycoprotein and target cells that express CD4 and either CXCR4 or CCR5. Co-cultivation of these cells leads to HIV-1 Env-mediated cell membrane fusion, resulting in formation of multinucleated syncytia. Membrane fusion induces expression of a reporter protein such as luciferase (Herschhorn et al., 2011, Ji et al., 2006) or β-galactosidase (Bradley et al., 2004). One such assay enables determination of both the efficiency and specificity of fusion inhibitors (Herschhorn et al., 2011). This approach uses effector cells that express both HIV-1 Env and the Renilla luciferase (R-Luc) reporter protein using inducible tetracycline-controlled transactivator (tTA) and target cells that express the HIV-1 receptor (CD4) and co-receptor (CCR5) and contain the firefly luciferase (F-Luc) reporter gene under the control of a tTA-responsive promoter. Upon fusion of the effector and target cells, tTA enters the target cells and activates the expression of the F-Luc reporter. The inhibition of fusion of cellular membranes is determined as a decrease in F-Luc luminescence, and the inhibitor specificity is measured as the R-Luc activity (Herschhorn et al., 2011).
Based on an HIV-1 cell-cell fusion method that uses a computer-controlled digital image analysis system for automatic quantitation (Lu et al., 2003), a modified method was developed to screen inhibitors targeting MERS-CoV S protein (Lu et al., 2014). To test potential fusion inhibitors, effector cells stably expressing the MERS-CoV spike protein S2S and EGFP were used to mediate fusion with target cells expressing the DPP4 receptor (Lu et al., 2014).
Virion-based fusion assays are another category of cell-based fusion assays. One such approach is based on production of chimeric HIV-1 virions carrying β-lactamase–Vpr chimeric protein (BlaM-Vpr). Chimeric HIV released into the cell culture media is isolated by ultracentrifugation and used to infect target cells. Entry of the virions into the cytoplasm is detected by cleavage of a fluorescent substrate by β-lactamase. The fluorescence shift corresponds to the fusion efficacy and is measurable by fluorescence microscopy, flow cytometry, or fluorometric plate reader (Cavrois et al., 2002, Cavrois et al., 2004, Cavrois et al., 2014). Modification of this assay by constructing pseudotyped HIV-1 virions in which HIV-1 Env was replaced with Ebola virus glycoprotein (GP) has also been described (Yonezawa et al., 2005). Tscherne et al. developed an approach to monitor viral entry using the BlaM reporter (Tscherne et al., 2010). Their assay uses influenza virus-like particles (VLPs) bearing the influenza membrane proteins hemagglutinin and neuraminidase. BlaM tagged with influenza matrix protein (M1) is incorporated into the VLPs and delivered into target cells. Upon release, BlaM can be detected by flow cytometry, microscopically, or fluorometrically.
A rapid cell-based HTS method was developed to assess SARS-CoV entry inhibitors (Zhou et al., 2011). This Dual Envelope Pseudovirion (DEP) assay employs two HIV pseudoviruses: one encodes an envelope protein from the target virus and firefly luciferase and the second encodes a control, unrelated viral envelope protein and Renilla luciferase. Reporter expression is determined with the Dual-Glo Luciferase Assay System (Promega). Inclusion of the unrelated envelope protein greatly reduced false positive hits (Zhou et al., 2011). The method was further used to screen compounds that inhibit entry of filoviruses, including Ebola virus (Zhou et al., 2015). A similar approach employing four pseudotyped HIV viruses, carrying Marburg virus glycoprotein, hemagglutinin and neuraminidase isolated from A/Goose/Qinghai/59/05 (H5N1) influenza virus, Ebolavirus Zaire envelope glycoprotein, and Lassa virus envelope glycoprotein, has been used for entry inhibitor screening (Cheng et al., 2017). This screening identified multiple compounds with potent inhibitory activity against entry of both Marburg and Ebola viruses in human cancer cell lines, and confirmed their anti-Ebola activity in human primary cells (Cheng et al., 2017). Other pseudotyped viral assays have been used to screen entry inhibitors of SARS-CoV, Ebola, Hendra, and Nipah viruses. To establish infection, the glycoproteins of these viruses must be processed by the host intracellular lysosomal protease cathepsin L (CatL). HTS resulted in identification of several inhibitors that block the cleavage of viral glycoprotein but not CatL itself (Elshabrawy et al., 2014).
Until recently, the development of anti-HBV therapeutics had been limited by the lack of an in vitro infection system. Several aspects of the HBV life cycle have been elucidated using in vitro production of HBV particles after transfection of human hepatoma cell lines (HepG2) with recombinant HBV DNA and by establishment of hepatoma cell lines with the entire HBV genome integrated, such as the HepG2.2.15 cell line (Ladner et al., 1997, Sells et al., 1987, Sureau et al., 1986). In addition to differentiated human (PHHs) (Gripon et al., 1988) and Tupaia belangeri (Glebe et al., 2003) hepatocytes, the HepaRG cell line, which exhibits hepatocyte-like characteristics, also supports HBV replication (Gripon et al., 2002). The identification of sodium taurocholate cotransporting polypeptide (NTCP) as an HBV receptor by Yan et al. opened possibilities to use NTCP-complemented HepG2 cells not only for studies of HBV replication mechanisms but also for HTS of inhibitors (Yan et al., 2012).
In an infection competition assay, HBV particles were isolated and used to infect HepaRG and PHHs cells that had been pre-incubated with HBV envelope protein-derived peptides to test their potential activity to block HBV infection. Twelve days after infection, viral RNAs were quantified by Northern blot (Gripon et al., 2002, Gripon et al., 2005). Using this assay, researchers identified a peptide that specifically prevents HBV and HDV entry into HepaRG and PHHs cell lines (Gripon et al., 2005), primary cultures of tupaia hepatocytes (Glebe et al., 2005), and cells in vivo (Lütgehetmann et al., 2012, Petersen et al., 2008, Volz et al., 2013). Recently, a Phase IIa clinical trial of a first-in-class entry inhibitor (myrcludex-B) that functions as an NTCP inhibitor was promisingly completed (Bogomolov et al., 2016).
Development of cell culture systems producing HCV pseudoparticles (HCVpp) and infectious HCV particles (HCVcc) has shed light on HCV interactions and enabled discovery of antiviral drugs and vaccines (Colpitts et al., 2016). HCVpp consist of HCV E1 and E2 glycoproteins enveloping a retroviral particle that packages GFP mRNA during assembly (Bartosch et al., 2003). The entry efficiency of HCVpp can be quantified by FACS analysis as the number of GFP-positive cells. Use of this screening system led to discovery of a triazine derivative that blocks the entry of HCV (Baldick et al., 2010).
Production of HCVcc is a robust system to generate infectious HCV in naïve cells (Kato et al., 2006, Zhong et al., 2005). The anti-HCV activity of hundreds of compounds approved for a wide variety of indications was determined immunochemically with anti-HCV E2 antibody in 96-well plate format. Over thirty compounds displayed anti-HCV activities, most of which were directed against HCV entry (Gastaminza et al., 2010).
3.2. Assays and methods for screening viral capsid disassembly/uncoating inhibitors
The majority of viruses enter cells by endocytosis. Unfortunately, there are no suitable experimental techniques for endosome handling, making it difficult to study early steps in the viral life cycle such as uncoating and capsid disassembly. Viruses that enter cells by direct membrane fusion, such as HIV-1, are an exception. There are several methods available to monitor and quantify HIV uncoating. As some of these were reviewed recently by Campbell and Hope (Campbell and Hope, 2015), we will discuss them very briefly. Two main techniques are used in these assays: ultracentrifugation or utilization of HIV-1 specific cellular restriction factors. The “in vitro core-stability assay” is based on ultracentrifugation of released HIV-1 virions through a detergent layer, where the viral membrane dissolves, into a sucrose gradient, where the viral cores are concentrated (Shah and Aiken, 2011). The “fate of capsid” assay uses ultracentrifugation through a sucrose cushion to separate the HIV-1 core from a whole cell lysate prepared shortly after infection (Stremlau et al., 2006). The “CsA-washout assay” involves specific cellular factors (TRIM-cyclophilin) that restrict HIV-1 infection by binding to the capsid core and cyclosporine A (CsA), which can effectively turn off restriction (Hulme et al., 2011). Recently, a novel entry/uncoating assay (EURT), an alternative to BlaM-Vpr (described in Section 3.1), was reported (Da Silva Santos et al., 2016). It quantifies the protein product of a virion-packaged mRNA reporter upon uncoating.
A method to monitor the uncoating/disassembly of the capsid of influenza A virus, which enters the cell by endocytosis, also is based on ultracentrifugation (Stauffer et al., 2016). Purified virions are separated using velocity gradient centrifugation through a two-layer glycerol gradient. Similar to the “in vitro core stability” assay for HIV, the sedimenting viruses migrate through the detergent-containing layer of the gradient, which dissolves the membrane to release the viral core. Moreover, modification of the detergent-containing glycerol layer—for example, by lowering pH, changing ionic strength, or adding putative viral uncoating factors—enables study of the factors or compounds that affect viral uncoating in vitro.
3.3. Assays and methods for screening and evaluating viral genome replication inhibitors
3.3.1. Polymerases
Depending on the virus type, either DNA or RNA polymerases replicate the viral genome. Thus, these enzymes play a key role in viral life cycles. Reverse transcriptase, an RNA-dependent DNA polymerase of retro- and hepadnaviruses, is unique among nucleic acid polymerases. Despite the different mechanisms of viral replication, polymerases, which are essential for all viruses, are excellent targets for antiviral therapies.
Polymerase inhibitors represent the vast majority of clinically approved antiviral drugs, followed by protease inhibitors, immunostimulators, entry inhibitors, and integrase inhibitors [reviewed in (De Clercq and Li, 2016)]. Polymerases continue to be a preferred target for newly designed inhibitors (Clercq, 2008, De Clercq and Li, 2016). Polymerase inhibitors may be nucleoside and nucleotide analogs, pyrophosphate analogs, or nonnucleosides, such as allosteric inhibitors (De Clercq and Li, 2016, Öberg, 2006). Non-specific approaches such as plaque assays were initially used to monitor the effectiveness of polymerase inhibitors (Tino et al., 1993, Tisdale et al., 1993). The activity of these inhibitors also can be evaluated based on their ability to prevent the cytopathic effects of the virus (Zhang et al., 2017). More straightforward assays involve measurement of incorporated radio-labeled nucleotides, which directly reflects the polymerase activity (Coates et al., 1992, Hirashima et al., 2006, Joyce, 2010); these include HTS methods using homopolymeric polycytidylic acid and [3H]-GTP (Amraiz et al., 2016). Alternatively, the pyrophosphate released during the polymerase reaction can be measured luminometrically by combining the primer extension with the commercial PiPer assay (Malvezzi et al., 2015). The pyrophosphate anion also can be detected with DNA-attached magnetic nanoparticles (Tong et al., 2015) or quantum dots (Chai et al., 2015). Frequently used fluorescence methods avoid the use of radiochemicals. These methods exploit a fluorescent label, such as dsDNA binding dyes like SYBR green or PicoGreen (Driscoll et al., 2014, Holden et al., 2009, Zipper et al., 2004), or FRET between two nucleotides (Schwartz and Quake, 2009, Weiss, 2000). Numerous kits for quantification of products of both DNA and RNA polymerases, including reverse transcriptase, are commercially available (Bustin, 2000). Quantitative real-time PCR is a preferred method to monitor the activity of DNA polymerases (cellular as well as viral) in the presence of inhibitors (Beadle et al., 2016, Zweitzig et al., 2012), and quantitative real-time reverse-transcription PCR has become standard for screening inhibitors of viral RNA polymerases (Lee et al., 2015, Okon et al., 2017, Pelliccia et al., 2017, Zhang et al., 2017). White et al. described a HTS method using a microfluidic apparatus combined with digital PCR for single-cell analysis (White et al., 2013). In their method, a fluorescently labeled template also serves as a primer, due to stem formation. It emits a measurable polarization signal both upon binding of the polymerase and extension (Mestas et al., 2007). The assay has been used for HTS of inhibitors of poliovirus RNA polymerase (Campagnola et al., 2011).
When available, viral genomes modified with reporter genes can be used for cell-based luminescent or fluorescent screening of viral inhibitors (Beadle et al., 2016, Edwards et al., 2015, Fenaux et al., 2016, Feng et al., 2014, Lo et al., 2017, Madhvi et al., 2017, Wang et al., 2015c). This approach can be extended to screen inhibitors of other enzymes. In the aptamer-based approach, a DNA template encodes RNA of interest joined to a fluorescence module and ribozyme sequence. When transcribed, the fluorescence module is released and detected (Höfer et al., 2013).
HCV uses an RNA-dependent RNA polymerase (RdRp). A unique cell-based assay for monitoring HCV RNA polymerase (NS5B) activity, based on the innate immune signaling molecule retionic acid-inducible gene I product (RIG-I), has been developed (Ranjith-Kumar et al., 2011). The RIG-I-like receptors are cytosolic proteins that recognize viral RNA and induce production of proinflammatory cytokines and interferon-activated genes (Jensen and Thomsen, 2012). RIG-I triggers cytokine production stimulated by various viral RNAs from different families, including Flaviviridae, Paramyxoviridae, Rhabdoviridae, Orthomyxoviridae, and Arenaviridae, as well as Ebola virus RNA (Jensen and Thomsen, 2012). The assay is based on recognition of HCV RNA produced by active NS5B by RIG-I, followed by RIG-I-mediated activation of firefly luciferase expression controlled by the interferon b promoter. Renilla luciferase expression is used for normalization of transfection efficacy.
3.3.2. Methyltransferases
The viral gRNAs of some RNA viruses are modified at the 5′ ends with cap structures, which may be either acquired from the host cell mRNA (e.g. in influenza virus) or newly synthesized (e.g. flaviviruses). The methylation of viral gRNA mimics that of cellular mRNA cap structures to enhance the chances of the virus to escape from cellular defense mechanisms and also to increase the efficiency of translation of viral proteins. Virus-specific methyltransferases are thus a promising therapeutic target.
Virus-encoded methyltransferases have been identified and characterized in flaviviruses such as Zika virus (Coloma et al., 2016, Coutard et al., 2017, Duan et al., 2017, Munjal et al., 2017, Zhao et al., 2017), West Nile virus, and dengue virus (Dong et al., 2012); rhabdoviruses such as vesicular stomatitis virus (VSV) (Rahmeh et al., 2009); coronaviruses such as SARS (Wang et al., 2015b); and roniviruses (Zeng et al., 2016). In flaviviruses, the N-terminal part of NS5 methyltransferase catalyzes cap formation via both guanine N-7 and ribose 2′-OH methylations at the 5′ end of gRNA, and the C-terminus of the enzyme is responsible for the RNA polymerase activity (Ray et al., 2006, Zhao et al., 2015). The C-terminal part of the SARS nsp14 protein exhibits guanine N7-methyltransferase activity, forming the gRNA cap, while the 3′-5′ exoribonuclease activity of the N-terminus enhances the fidelity of viral replication (Case et al., 2016, Minskaia et al., 2006). In VSV, the methyltransferase activity resides in the conserved region VI of the multifunctional large polymerase protein (Li et al., 2005, Ma et al., 2014).
Some cellular methyltransferases regulate viral infections, as shown for herpes simplex virus, for which epigenetic control is involved in viral latency (Cliffe and Wilson, 2017). Inhibition of human histone methyltransferases such as histone-lysine N-methyltransferase (EZH2/1) (Arbuckle et al., 2017) or lysine-specific demethylase-1 (LSD1) induces antiviral signaling pathways (Liang et al., 2009). The inhibitory effect of histone demethylase activity has been demonstrated for human cytomegalovirus (Gan et al., 2017) and herpes simplex virus (Hill et al., 2014, Liang et al., 2013).
The activity of purified recombinant methyltransferases can be determined by measuring the methylation by-product S-adenosyl homocysteine (SAH) using commercially available kits. In one such assay, conversion of S-adenosyl methionine (SAM) to SAH is monitored luminometrically via luciferase reaction, in which measurable ATP is generated through a sequence of reactions with MTase-Glo™ reagent (Promega). The EPIgeneous™ methyltransferase kit (CisBio Bioassays) is based on competition of produced SAH with fluorescently labeled SAH (SAH-d2 tracer) for binding to a terbium cryptate-labeled anti-SAH antibody. The decrease in FRET between the tracer and antibody is then evaluated. ELISA using anti-5-methylcytosine antibody can be used to quantify methylation of immobilized cytosine-rich DNA substrate (e.g. EpiQuik™ DNA Methyltransferase Activity/Inhibition Assay Kit; Epigentek). This method was modified with homogenous time resolved FRET (Degorce et al., 2009) to screen a library of inhibitors against SARS-CoV nsp14 (Aouadi et al., 2017).
Flaviviral and human cap N7-MTases have been screened with radioactive assays using 3H-labeled SAM and GpppAC4 or 7mGpppAC4 synthetic RNAs. The 3H methyl transferred onto DEAE filter-bound RNA can be measured by scintillation after multiple washings to remove unincorporated 3H-SAM (Coutard et al., 2017). Alternatively, in vitro transcription can be carried out using 3H-SAM. The radioactively labeled products are then resolved by thin-layer chromatography and developed using a phosphorimager (Li et al., 2007).
A yeast cell-based method was established based on the finding that coronavirus methyltransferase can functionally replace a methyltransferase essential for yeast viability (Chen et al., 2009, Sun et al., 2014). In this method, Sun et al. constructed recombinant yeasts producing the viral methyltransferase instead of the yeast one (Sun et al., 2014). This strain was used for a HTS of methyltransferase inhibitor activity that negatively correlated with the cell density after 20 h incubation.
3.3.3. Helicases
Virus-encoded ATPase-driven helicases have been identified in numerous human pathogens. Helicases from several (+)RNA viruses have been characterized, including NS3 helicases from flaviviruses such as dengue virus, HCV, West Nile virus, yellow fever virus, and Japanese encephalitis virus (Cao et al., 2016, Gu and Rice, 2016, Jain et al., 2016, Lin et al., 2017, Nedjadi et al., 2015, Wu et al., 2005). SARS and MERS coronaviruses encode nsp13 with helicase activity (Adedeji and Lazarus, 2016, Hao et al., 2017, Seybert et al., 2000). In Semliki Forest virus, a representative member of the togavirus family, helicase activity is encoded by the N-terminal domain of nsP2 protein. Helicases are also common in some DNA viruses, including poxviruses. These include the vaccinia virus helicase-primase D5 (Bayliss and Smith, 1996, Hutin et al., 2016), E1 protein of bovine papilloma virus 1 (Yang et al., 1993), and the large tumor antigen of SV40 (Stahl et al., 1986). Helicases appear to be attractive targets for antiviral drugs (Briguglio et al., 2011, Frick, 2003, Reynolds et al., 2015), but development of such compounds is challenged by cytotoxicity and bioavailability issues (Kwong et al., 2005).
Traditional methods for monitoring the activity of RNA helicases use radioactively labeled dsRNA substrates and follow the unwinding reaction by electrophoretic separation (nondenaturing PAGE) of the ss reaction products, which are detected autoradiographically (Adedeji et al., 2012, Utama et al., 2000). To determine whether the inhibitor affects binding of helicase to nucleic acid, a standard gel mobility shift assay is usually used (Adedeji et al., 2012).
Helicase activity is fueled by ATP hydrolysis; thus, inhibition of ATPase activity became another possible antiviral strategy. ATPase activity can be determined either by the decrease in ATP or formation of ADP (using commercially available fluorescent anti-ADP antibodies) or inorganic pyrophosphate. Phosphates released by ATP hydrolysis can form a molybdophosphate complex that can be measured colorimetrically using malachite green, quinaldine red, or rhodamine B (Baykov et al., 1988, Debruyne, 1983, Miyata et al., 2010) or by light-scattering (Oshima et al., 1996). The colorimetric methods can be miniaturized for HTS (Zuck et al., 2005). The absorbance-based assay was also converted into a HTS method based on fluorescence quenching by a colored quinaldine red complex (Miyata et al., 2010). An alternative method employs a europium–tetracycline complex for luminescent determination of inorganic phosphate (Schäferling and Wolfbeis, 2007). A more complex coupled enzyme colorimetric assay (with maltose phosphorylase, glucose oxidase, and horseradish peroxidase) was used for HTS of ATPase inhibitors (Avila et al., 2006). Assays for luminescent and fluorescent screening of ATPase activity, including an immunochemical method using fluorescently labeled anti-ADP antibodies, have been reviewed by Shadrick and colleagues (Shadrick et al., 2013). In a radioactive method using [γ-32P]ATP, the AMP product was separated from unreacted ATP by thin-layer chromatography on polyethyleneimine-cellulose and visualized autoradiographically (Adedeji et al., 2012).
Several research groups have described fluorescence assays to identify inhibitors targeting SARS-CoV helicase (nsp13) (Adedeji et al., 2012, Cho et al., 2015, Özeş et al., 2014). The substrate is usually a dsDNA oligonucleotide consisting of a fluorescently labeled strand annealed to the complementary strand carrying a quencher. This approach was also adapted for real-time determination of the RNA helicase activity of HCV (Tani et al., 2010). In this assay, an ssDNA capture strand complementary to the strand carrying the quencher was used to prevent reannealing of the unwound duplex. Recently, a colorimetric assay for monitoring helicase activity using DNA-conjugated gold nanoparticles was developed (Deka et al., 2017). This method is based on shifts in the optical properties of nanoparticles due to DNA unwinding and allows simple screening of inhibitor activity. The dsDNA melting curves can be determined spectrophotometrically (at 524 nm and 260 nm) or even by the naked eye. Another fluorescence HTS of dengue virus NS3 helicase inhibitors measures the unwinding of a double-labeled molecular beacon (Basavannacharya and Vasudevan, 2014). Other approaches involve graphene oxide-based fluorescence monitoring of viral helicase activity [reviewed in (Jang et al., 2013)]. A G-quadruplex-based method for label-free determination of HCV helicase NS3 activity measures changes in the luminescence of transition metal complexes with DNA upon helicase-mediated quadruplex melting (Leung et al., 2015).
3.3.4. Kinases
Both protein tyrosine kinases and protein serine/threonine (S/T) kinases have been found in viruses. Tyrosine kinase function is well-understood in connection with oncogenic retroviruses, [for review on retroviral oncogenes, see (Vogt, 2012)]. In contrast to tyrosine kinases from oncogenic viruses, such as Rous sarcoma virus src tyrosine kinase, viral S/T kinases share little homology with cellular enzymes. They are exclusively encoded by large DNA viruses (e.g. herpesviruses), in which they play important roles in viral virulence, helping the virus to escape defense mechanisms such as those regulated by cytokine signaling pathways (Jacob et al., 2011, Sato et al., 2017). Their autophosphorylation and transphosphorylation activities mimic those of cellular cyclin-dependent kinases such as cdc2. For example, viral kinases can phosphorylate translation elongation factor 1 delta (EF-1δ) (Jacob et al., 2011, Kawaguchi and Kato, 2003). All herpesviruses encode the S/T protein kinase UL13, and US3 S/T kinases have been described in the alphaherpesvirus subfamily (Kato, 2016, Kawaguchi and Kato, 2003).
In addition to protein kinases, HSV encodes a thymidine kinase. Unlike cellular thymidine kinase, HSV thymidine kinase has a wide substrate specificity that includes pyrimidine and purine phosphonate analogs (e.g. acyclovir, ganciclovir, penciclovir) (De Clercq and Li, 2016). In the body, ganciclovir is phosphorylated by cellular kinases and penciclovir and acyclovir by virus-encoded thymidine kinases to the active nucleoside triphosphate forms of the drugs that inhibit viral DNA synthesis (De Clercq and Li, 2016, Kokoris and Black, 2002).
Some cellular protein kinases appear to support viral replication. For example, polo-like kinases induce early stages in the influenza virus life cycle (Pohl et al., 2017), and human protein kinase C regulates the assembly of the ribonucleoprotein complexes in influenza virus (Mondal et al., 2017). Inhibitors of Abelson tyrosine-protein kinase 2 are active against SARS and MERS coronaviruses (Coleman et al., 2016). Several in vitro approaches can be used to determine kinase activity as well as the activity of kinase inhibitors.
Analyses of the cellular phosphoproteome, sometimes accompanied by phosphopeptide enrichment, have become standard to determine kinase activity (Lea and Simeonov, 2011, Meyer et al., 2017, Vyse et al., 2017). These assays can be used to assess the impact of inhibitors on the overall phosphoproteome in mammalian cells. Although these methods provide complex information about the overall array of kinases and phosphatases in the cell (Olsen et al., 2006), they are not applicable to screening inhibitors of a single viral kinase. For these purposes, in vitro assays with recombinant kinases have been developed [for review see (Ma et al., 2008)], including some HTS fluorescence methods (Zegzouti et al., 2009) that can replace radioactive techniques using [γ32P]ATP (Sanghera et al., 2009). Mass spectrometric analysis also can be used to identify in vitro kinase inhibitors. For these analyses, synthetic peptides, proteins, or phosphatase- and heat-treated tissue samples (to dephosphorylate the proteins and inactivate all enzymes, respectively) are subjected to kinase treatment in the presence of inhibitors (Huang et al., 2007, Meyer et al., 2017, Xue et al., 2012).
Recombinant kinase activity in the presence of inhibitors can be quantified as ATP consumption or ADP production in the phosphorylation reaction by numerous commercial kits. Other methods monitor binding of inhibitors to phage-displayed kinases in ligand competition assays (Fabian et al., 2005).
Fluorescence methods including FRET, fluorescence polarization or intensity endpoint measurement, and lifetime imaging of fluorescence (Li et al., 2008) including fluorescence biosensors (Zhang and Allen, 2007) have been reviewed elsewhere. Sulfonamido-oxine labeled peptides can be used as chromophores that bind Mg2 + upon phosphorylation and emit chelation-enhanced fluorescence (Devkota et al., 2013, Luković et al., 2008). Kinase-catalyzed phosphorylation of fluorescent peptides promotes their binding to metal-coated nanoparticles, which decreases their mobility and enhances measurable fluorescence polarization (Lea and Simeonov, 2011, Sportsman et al., 2004). TbIII complexes, in which phosphotyrosine induces fluorescence emission (Wang et al., 2015a), may be used to evaluate protein tyrosine kinase activity (Akiba et al., 2015, Sumaoka et al., 2016). Fluorescence polarization methods also can be useful for drug screening [reviewed in (Hall et al., 2016)]. Other alternatives are immunochemical methods that use antibodies specific to the phosphorylated amino acids, such as phosphotyrosine (Li et al., 2001, Youngren et al., 1997) or phosphoserine/phosphothreonine. These antibodies can be used to detect protein/peptide phosphorylation by Western blotting, ELISA, or immunoprecipitation of phosphorylated proteins for further mass spectrometry-based analysis (Grønborg et al., 2002, Zhang et al., 2002). An elegant approach that limits the false-positive hits in screening of specific kinase inhibitors is based on an in situ proximity ligation assay using both an antibody against the target protein and an anti-phosphotyrosine antibody (Leuchowius et al., 2010). Both antibodies are coupled with oligonucleotides, which when brought together due to antibody binding, can be enzymatically ligated and replicated through rolling circle amplification to form a long linear tandem repeat of sequences detectable by a complementary fluorescent oligonucleotide.
In addition to protein kinases, some lipid kinases have been targeted by antivirals. One example is sphingosine kinase 1, which affects replication of dengue virus (Aloia et al., 2017). Its activity can be determined by measuring the production of 32P-labeled sphingosine-1-phosphate from sphingosine and [γ32P]ATP (Clarke et al., 2016, Pitman et al., 2012).
3.3.5. Integrase
Retroviral integrase inhibitors are a new type approved new type of inhibitors imposed by the emergence of drug-resistant mutants. HIV integrase activities, integrase inhibitors, and drug resistance have been discussed in detail elsewhere (Andrake and Skalka, 2015, Anstett et al., 2017, Hajimahdi and Zarghi, 2016, Liao et al., 2010, Podany et al., 2017, Thierry et al., 2016). Methods to assess the two major activities of integrase—end processing of the reverse transcription product and its joining to target chromosomal DNA—have been reviewed in detail by several groups (Engelman and Cherepanov, 2014, Marchand et al., 2001, Merkel et al., 2009). Initial methods used radioactively labeled DNA oligonucleotides comprising the terminal cis-acting sequences of linear viral DNA required for integration. The joining of the processed strand to the other strand (self-integration) or to supplemented target DNAs can be analyzed by PAGE (Katz et al., 1990, Katzman et al., 1989). A less time-consuming, non-radioactive method involves time-resolved fluorescence anisotropy measurement using a 21-meric oligonucleotide fluorescently labeled on the terminal GT dinucleotide. This assay monitors the binding of integrase to the substrate as well as the subsequent 3′-processing reaction, which both change the anisotropy (Guiot et al., 2006). Alternatively, the yields of both the processing and joining reactions can be measured upon separation of the radioactively labeled product from the rest of the DNA molecule using adsorption to PEI-cellulose (Muller et al., 1993). A real-time HTS method measures fluorescence emission resulting from removal of the 3′-terminal dinucleotide, labeled with a quencher, by integrase (He et al., 2007). Han et al. described a fluorescence method to screen molecules that inhibit binding of integrase to viral DNA (Han et al., 2013). Methods evaluating the integrase strand transfer reaction have been modified to a high-throughput format using magnetic beads (He et al., 2008) or streptavidin-coated microplates (John et al., 2005). A method to assess strand transfer by time-resolved FRET with a europium-streptavidin-labeled substrate has been optimized for 384- and 1536-well plate formats (Wang et al., 2005).
3.4. Assay and methods for screening and evaluating viral assembly inhibitors
The HBV capsid protein is the building block of the viral core, surrounding the viral nucleic acid (pre-genomic RNA, pgRNA) and reverse transcriptase. The HBV core is icosahedral, formed by 240 copies of capsid protein dimers. In vitro HTS of HBV core assembly inhibitors using a modified HBV capsid protein has been described (Stray et al., 2006). The capsid protein was modified by deleting the nucleic acid binding domain, which is dispensable for capsid assembly, and the N-terminal assembly domain alone was used in the assay. To fluorescently label the HBV capsid protein, all cysteine residues dispensable for assembly were replaced with alanines. A unique cysteine residue (C150) was C-terminally joined to the assembly domain and labeled with fluorescent BODIPY-FL dye (C150BO). During assembly, capsid proteins dimerize, bringing C150BO residues close together and resulting in C150BO fluorescence self-quenching. Following incubation of the labeled HBV protein with inhibitors, fluorescence was measured in black 96-well microtiter plates.
Development of in vitro assembly systems has contributed greatly to current understanding of the structure of retroviral particles and mechanisms of virion formation. These systems also became the base for several high throughput assays for screening assembly inhibitors. During the last 20 years, a number of in vitro assembly assays have been established, mainly for HIV-1 (Campbell et al., 2001, Campbell and Rein, 1999, Ehrlich et al., 1992, Gross et al., 2000, Lanman et al., 2002), Mason-Pfizer monkey virus (Bohmova et al., 2010, Klikova et al., 1995, Rumlova-Klikova et al., 2000, Rumlova-Klikova et al., 1999, Ulbrich et al., 2006), Rous sarcoma virus (Campbell and Vogt, 1995, Campbell and Vogt, 1997, Purdy et al., 2008, Purdy et al., 2009), and murine leukemia virus (Dolezal et al., 2016, Hadravova et al., 2012, Cheslock et al., 2003). HTS assays for inhibitors of HIV-1 assembly include several methods using purified HIV-1 CA or CA-NC proteins. One of them, the turbidimetric assay, is based on the observation that direct dilution of the HIV-1 capsid protein (CA) into a high-salt solution (1.6–2.2 M NaCl) leads to the formation of tubular structures. As the tube formation is accompanied by an increase in light scattering, assembly can be detected as an increase in turbidity, and the rate of turbidity change is proportional to the rate of the assembly (Lanman et al., 2002). Other method published by Lemke et al., exploits the affinity of nucleocapsid (NC) to a short TG-rich deoxyribooligonucleotide, d(TG25), which is used as a scaffold (Lemke et al., 2012). This arrangement enables CA to assemble at much lower protein and salt concentrations than in the turbidimetric assay (Lanman et al., 2002). Biotin-labeled d(TG25) bound on the surface of neutravidin-coated microtiter well plates nucleates assembly of complexes of CA-NC and soluble fluorescein-labeled d(TG25). Fluorescence is measured after washing to remove the unbound and unassembled material from the captured assembly products (Lemke et al., 2012). Similarly, the FAITH assay uses a dually labeled oligonucleotide (tqON). However, in this case, the ssDNA oligonucleotide tqON is labeled with the reporter dye fluorescein (FAM) as well as Black Hole Quencher (BHQ); thus, it does not emit any fluorescence. The assembly reaction is triggered by mixing HIV-1 CA-NC or a Gag-truncated assembly-competent version with tqON. Following incubation, during which tqON is incorporated into the particles, exonuclease is added to degrade unbound tqON, while co-assembled tqON is protected from cleavage. Degradation of free unbound tqON with ExoI results in separation of FAM from its quencher, and the emitted fluorescence is measured (Hadravova et al., 2015).
Phage display has been employed to screen peptide inhibitors of HIV-1 assembly (Sticht et al., 2005). A commercial library of M13-derived phages presenting random 12-amino-acid peptides was analyzed for specific binding to purified CA or CA-NC proteins. The specifically bound phages were sequenced, and corresponding peptides were chemically synthesized and re-tested in an in vitro assembly assay (Gross et al., 2000).
Late in the retroviral life cycle, gRNA is incorporated into the nascent particle during assembly at the plasma membrane. NC contains two zinc-finger domains that are responsible for specific binding of gRNA. To screen for compounds that would prevent NC-RNA/DNA interactions, a HTS system consisting of two sequential screens was developed (Breuer et al., 2012). The primary screen uses fluorescence polarization (FP), while the secondary one uses differential scanning fluorimetry (DSF). The combination and order of these two techniques were selected to first identify compounds that disrupt interactions between DNA and NC, and then identify the compounds binding to NC during the secondary screen.
A rapid and simple turbidimetric method was developed to screen inhibitors of assembly of HCV core protein (Fromentin et al., 2007). For the in vitro assembly reaction, two components, an N-terminal part of the HCV core protein corresponding to the minimal assembly competent domain and RNA corresponding to the full-length 5′UTR of HCV, were used. Assembly of HCV nucleocapsid-like particles was initiated by mixing the purified protein and nucleic acid, and the assembly process was monitored by measuring turbidity at 350 nm.
3.5. Assay and methods for screening and evaluating viral maturation inhibitors
Numerous viruses, including picornaviruses, retroviruses, alphaviruses, and flaviviruses, encode proteases that are essential for their virulence. The majority of viral proteases specifically cleave viral polyprotein precursors to liberate the functional proteins of the virion. Some viral proteases, such as the papain-like proteases of coronaviruses, also reprogram cellular signaling pathways, including ubiquitination mechanisms and interferon controlled responses, to prevent degradation of viral components (Clementz et al., 2010, Frieman et al., 2009, Randow and Lehner, 2009, Xing et al., 2013). Numerous viruses, including papillomaviruses (Bronnimann et al., 2016, Buck et al., 2005) and retroviruses (Hallenberger et al., 1992), use also host cell proteases, mainly furin, to trim their envelope and surface proteins. This triggers conformational changes required for interaction with cellular receptors and membrane fusion in enveloped viruses. Some viruses use proteases other than furin to modulate their infectivity, as shown for viruses entering airway epithelial cells [reviewed in (Laporte and Naesens, 2017)] such as influenza virus (Böttcher-Friebertshäuser et al., 2010, Kühn et al., 2016), Newcastle disease virus (Gotoh et al., 1992), and respiratory syncytial virus (Sugrue et al., 2001). Extensive research efforts have yielded detailed information about HIV-1 protease and its inhibitors [reviewed in (De Clercq, 2004, Konvalinka et al., 2015, Midde et al., 2016)]. Large amounts of data also are available for HCV (Foote et al., 2011, Pawlotsky et al., 2007, Razonable, 2011) and SARS-CoV 3CL protease inhibitors (Pillaiyar et al., 2016), although there is not yet an inhibitor of the latter target approved for clinical use. Numerous approved drugs are synthetic peptides derived from the natural proteolytic substrates of target viruses modified to improve the in vivo effects related to bioavailability, stability, and so on.
Numerous in vitro assays to monitor the activity of proteases and their inhibitors, including commercial kits, have been developed. Classical methods use synthetic peptides that mimic the target sites of the protease. Although some of the methods described here were not originally designed to screen the activity of viral proteases in the presence of inhibitors, they can be adapted for this purpose by simply changing the peptide sequence to the target site of the protease of interest. The cleavage yield is usually monitored either colorimetrically (Ding and Yang, 2015, Zhou et al., 2014) or as a change in fluorescence triggered by the release of fluorescent labels such as 7-amido-4-methylcoumarin (AMC) or rhodamine (Grant et al., 2002). Fluorogenic substrates have been used to determine the activity of coronavirus proteases and screen inhibitors (Kuo et al., 2004, Lee et al., 2014, Park et al., 2017, Song et al., 2014, Tomar et al., 2015, Wang et al., 2016, Yang et al., 2005, Zhao et al., 2008). Some recently described arrangements employ nanoparticles (Feltrup and Singh, 2012, Khalilzadeh et al., 2016, Udukala et al., 2016, Wang et al., 2014, Zeng et al., 2015) or quantum dot bioconjugates (Lee and Kim, 2015, Li et al., 2014, Medintz et al., 2006) with immobilized fluorescently or luminescently labeled peptide substrates. Alternatively, cleavage products may be monitored by analysis of proteolytic products by mass spectrometric methods (Hu et al., 2015, Joshi et al., 2017, Lathia et al., 2011, Rumlová et al., 2003), analytical HPLC (Teruya et al., 2016), or electrochemical methods based on the difference in penetration of substrate and cleavage products through the membrane of a polyion-selective sensor (Gemene and Meyerhoff, 2011, Han et al., 1996). To study the specificity of inhibitor binding and to extend the research to rational design of inhibitors, X-ray or NMR structures of proteases in complex with the inhibitor may be determined, as reported in numerous cases for the proteases of HIV-1 [reviewed in (Ghosh et al., 2016)], HCV (Yilmaz et al., 2016), and MERS (St. John et al., 2015).
Cell-based assays can provide additional information, including the capability of the inhibitor to pass through the cell membrane and its stability in the cytoplasm. A general determination of infectivity, such as a plaque cytotoxicity assay in the presence of protease inhibitor, may be used for confirmation. One elegant example exploits the cytotoxicity of HIV protease. Cells are transfected with a protease precursor fused to GFP. In the absence of inhibitor, HIV-1 protease is autocatalytically activated and cleaves a broad variety of cellular proteins, resulting in activation of apoptosis and cell death (Cummins and Badley, 2010, Rumlova et al., 2014). This toxic effect, when suppressed by active inhibitors, results in production of a GFP signal in surviving cells (Lindsten et al., 2001). Another elegant approach for HIV and coxsackievirus B3 proteases, which both undergo autocatalytic cleavage, employs constructs in which the protease gene is inserted between sequences encoding the DNA-binding domain and the domain that activates transcription of the GAL1-lacZ reporter gene (Dasmahapatra et al., 1992, Murray et al., 1993). The protease-mediated cleavage separates the DNA-binding domain from the trans-activating domain and results in failure of reporter gene transcription. This approach has also been modified with GFP as a reporter gene (Hilton and Wolkowicz, 2010). Co-expression of cleavage-activated luciferase substrate and MERS-CoV protease permits both live-cell imaging and quantification of the enzyme activity (Kilianski et al., 2013).
4. Conclusion
The need to develop new antiviral compounds will likely persist over the long term, although there has been enormous progress in molecular biology methods, especially RNA silencing, bioinformatics, imaging, and structural biology techniques. Viruses present challenging targets for drug development due their flexibility and adaptability caused by the error-prone copying of their genomes, which can result in emergence of drug-resistant mutants. Viral integration into the host genome and inhibitor toxicity are other obstacles. Here, we provide an overview of in vitro methods, including cell-based assays, that may be suitable for screening of antivirotics that interfere with the key steps of viral life cycles and target either virus or cell-encoded proteins required for the infectivity.
Acknowledgements
This work was supported by GA ČR (CZ) GA17-25602S to MR, GA17-24281S to TR and the Ministry of Education, Youth and Sport of the Czech Republic (OPPC CZ.2.16/3.1.00/24503), through its “National Program of Sustainability I” NPU LO 1601.
Contributor Information
Michaela Rumlová, Email: michaela.rumlova@vscht.cz.
Tomáš Ruml, Email: tomas.ruml@vscht.cz.
References
- Adedeji A.O., Lazarus H. Biochemical characterization of Middle East respiratory syndrome coronavirus helicase. mSphere. 2016;1(5) doi: 10.1128/mSphere.00235-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adedeji A.O., Singh K., Calcaterra N.E., DeDiego M.L., Enjuanes L., Weiss S., Sarafianos S.G. Severe acute respiratory syndrome coronavirus replication inhibitor that interferes with the nucleic acid unwinding of the viral helicase. Antimicrob. Agents Chemother. 2012;56(9):4718–4728. doi: 10.1128/AAC.00957-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahi Y.S., Mittal S.K. Components of adenovirus genome packaging. Front. Microbiol. 2016;7:1503. doi: 10.3389/fmicb.2016.01503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akiba H., Sumaoka J., Tsumoto K., Komiyama M. Click conjugation of a binuclear terbium(III) complex for real-time detection of tyrosine phosphorylation. Anal. Chem. 2015;87(7):3834–3840. doi: 10.1021/ac5045466. [DOI] [PubMed] [Google Scholar]
- Alirezaei M., Flynn C.T., Wood M.R., Whitton J.L. Pancreatic acinar cell-specific autophagy disruption reduces coxsackievirus replication and pathogenesis in vivo. Cell Host Microbe. 2012;11(3):298–305. doi: 10.1016/j.chom.2012.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aloia A.L., Calvert J.K., Clarke J.N., Davies L.T., Helbig K.J., Pitson S.M., Carr J.M. Investigation of sphingosine kinase 1 in interferon responses during dengue virus infection. Clin. Translational Immunol. 2017;6(7):e151. doi: 10.1038/cti.2017.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amara A., Mercer J. Viral apoptotic mimicry. Nat. Rev. Microbiol. 2015;13(8):461–469. doi: 10.1038/nrmicro3469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambrose Z., Aiken C. HIV-1 uncoating: connection to nuclear entry and regulation by host proteins. Virology. 2014;454–455:371–379. doi: 10.1016/j.virol.2014.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amraiz D., Zaidi N.-U.-S.S., Fatima M. Development of robust in vitro RNA-dependent RNA polymerase assay as a possible platform for antiviral drug testing against dengue. Enzym. Microb. Technol. 2016;92:26–30. doi: 10.1016/j.enzmictec.2016.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrake M.D., Skalka A.M. Retroviral integrase: then and now. Annual Rev. Virol. 2015;2(1):241–264. doi: 10.1146/annurev-virology-100114-055043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aneja K.K., Yuan Y. Reactivation and lytic replication of Kaposi's sarcoma-associated herpesvirus: an update. Front. Microbiol. 2017;8(613) doi: 10.3389/fmicb.2017.00613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anstett K., Brenner B., Mesplede T., Wainberg M.A. HIV drug resistance against strand transfer integrase inhibitors. Retrovirology. 2017;14(1):36. doi: 10.1186/s12977-017-0360-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aouadi W., Eydoux C., Coutard B., Martin B., Debart F., Vasseur J.J., Contreras J.M., Morice C., Quérat G., Jung M.-L., Canard B., Guillemot J.-C., Decroly E. Toward the identification of viral cap-methyltransferase inhibitors by fluorescence screening assay. Antivir. Res. 2017;144:330–339. doi: 10.1016/j.antiviral.2017.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbuckle J.H., Gardina P.J., Gordon D.N., Hickman H.D., Yewdell J.W., Pierson T.C., Myers T.G., Kristie T.M. Inhibitors of the histone methyltransferases EZH2/1 induce a potent antiviral state and suppress infection by diverse viral pathogens. MBio. 2017;8(4) doi: 10.1128/mBio.01141-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avila C., Hadden M.K., Ma Z., Kornilayev B.A., Ye Q.-Z., Blagg B.S.J. High-throughput screening for Hsp90 ATPase inhibitors. Bioorg. Med. Chem. Lett. 2006;16(11):3005–3008. doi: 10.1016/j.bmcl.2006.02.063. [DOI] [PubMed] [Google Scholar]
- Baldick C.J., Wichroski M.J., Pendri A., Walsh A.W., Fang J., Mazzucco C.E., Pokornowski K.A., Rose R.E., Eggers B.J., Hsu M., Zhai W., Zhai G., Gerritz S.W., Poss M.A., Meanwell N.A., Cockett M.I., Tenney D.J. A novel small molecule inhibitor of hepatitis C virus entry. PLoS Pathog. 2010;6(9):e1001086. doi: 10.1371/journal.ppat.1001086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barton E.S., Forrest J.C., Connolly J.L., Chappell J.D., Liu Y., Schnell F.J., Nusrat A., Parkos C.A., Dermody T.S. Junction adhesion molecule is a receptor for reovirus. Cell. 2001;104(3):441–451. doi: 10.1016/s0092-8674(01)00231-8. [DOI] [PubMed] [Google Scholar]
- Bartosch B., Dubuisson J., Cosset F.L. Infectious hepatitis C virus pseudo-particles containing functional E1-E2 envelope protein complexes. J. Exp. Med. 2003;197(5):633–642. doi: 10.1084/jem.20021756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basavannacharya C., Vasudevan S.G. Suramin inhibits helicase activity of NS3 protein of dengue virus in a fluorescence-based high throughput assay format. Biochem. Biophys. Res. Commun. 2014;453(3):539–544. doi: 10.1016/j.bbrc.2014.09.113. [DOI] [PubMed] [Google Scholar]
- Baykov A.A., Evtushenko O.A., Avaeva S.M. A malachite green procedure for orthophosphate determination and its use in alkaline phosphatase-based enzyme immunoassay. Anal. Biochem. 1988;171(2):266–270. doi: 10.1016/0003-2697(88)90484-8. [DOI] [PubMed] [Google Scholar]
- Bayliss C.D., Smith G.L. Vaccinia virion protein I8R has both DNA and RNA helicase activities: implications for vaccinia virus transcription. J. Virol. 1996;70(2):794–800. doi: 10.1128/jvi.70.2.794-800.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beadle J.R., Valiaeva N., Yang G., Yu J.-H., Broker T.R., Aldern K.A., Harden E.A., Keith K.A., Prichard M.N., Hartman T., Buckheit R.W., Chow L.T., Hostetler K.Y. Synthesis and antiviral evaluation of octadecyloxyethyl benzyl 9-[(2-Phosphonomethoxy)ethyl]guanine (ODE-Bn-PMEG), a potent inhibitor of transient HPV DNA amplification. J. Med. Chem. 2016;59(23):10470–10478. doi: 10.1021/acs.jmedchem.6b00659. [DOI] [PubMed] [Google Scholar]
- Beck J., Nassal M. Hepatitis B virus replication. World J. Gastroenterol. 2007;13(1):48–64. doi: 10.3748/wjg.v13.i1.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benavente J., Martinez-Costas J. Early steps in avian reovirus morphogenesis. Curr. Top. Microbiol. Immunol. 2006;309:67–85. doi: 10.1007/3-540-30773-7_3. [DOI] [PubMed] [Google Scholar]
- Bieniasz P.D. Late budding domains and host proteins in enveloped virus release. Virology. 2006;344(1):55–63. doi: 10.1016/j.virol.2005.09.044. [DOI] [PubMed] [Google Scholar]
- Bird S.W., Kirkegaard K. Escape of non-enveloped virus from intact cells. Virology. 2015;479:444–449. doi: 10.1016/j.virol.2015.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bird S.W., Kirkegaard K. Nonlytic spread of naked viruses. Autophagy. 2015;11(2):430–431. doi: 10.4161/15548627.2014.994372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bird S.W., Maynard N.D., Covert M.W., Kirkegaard K. Nonlytic viral spread enhanced by autophagy components. Proc. Natl. Acad. Sci. 2014;111(36):13081–13086. doi: 10.1073/pnas.1401437111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaas D. Viral entry pathways: the example of common cold viruses. Wiener Medizinische Wochenschrift (1946) 2016;166:211–226. doi: 10.1007/s10354-016-0461-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehmer P., Nimonkar A. Herpes virus replication. IUBMB Life. 2003;55(1):13–22. doi: 10.1080/1521654031000070645. [DOI] [PubMed] [Google Scholar]
- Bogomolov P., Alexandrov A., Voronkova N., Macievich M., Kokina K., Petrachenkova M., Lehr T., Lempp F.A., Wedemeyer H., Haag M., Schwab M., Haefeli W.E., Blank A., Urban S. Treatment of chronic hepatitis D with the entry inhibitor myrcludex B: First results of a phase Ib/IIa study. J. Hepatol. 2016;65(3):490–498. doi: 10.1016/j.jhep.2016.04.016. [DOI] [PubMed] [Google Scholar]
- Bohmova K., Hadravova R., Stokrova J., Tuma R., Ruml T., Pichova I., Rumlova M. Effect of dimerizing domains and basic residues on in vitro and in vivo assembly of Mason-Pfizer monkey virus and human immunodeficiency virus. J. Virol. 2010;84(4):1977–1988. doi: 10.1128/JVI.02022-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böttcher-Friebertshäuser E., Freuer C., Sielaff F., Schmidt S., Eickmann M., Uhlendorff J., Steinmetzer T., Klenk H.-D., Garten W. Cleavage of influenza virus hemagglutinin by airway proteases TMPRSS2 and HAT differs in subcellular localization and susceptibility to protease inhibitors. J. Virol. 2010;84(11):5605–5614. doi: 10.1128/JVI.00140-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley J., Gill J., Bertelli F., Letafat S., Corbau R., Hayter P., Harrison P., Tee A., Keighley W., Perros M., Ciaramella G., Sewing A., Williams C. Development and automation of a 384-well cell fusion assay to identify inhibitors of CCR5/CD4-mediated HIV virus entry. J. Biomol. Screen. 2004;9(6):516–524. doi: 10.1177/1087057104264577. [DOI] [PubMed] [Google Scholar]
- Breuer S., Chang M.W., Yuan J., Torbett B.E. Identification of HIV-1 inhibitors targeting the nucleocapsid protein. J. Med. Chem. 2012;55(11):4968–4977. doi: 10.1021/jm201442t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briguglio I., Piras S., Corona P., Carta A. Inhibition of RNA helicases of ssRNA(+) virus belonging to Flaviviridae, Coronaviridae and Picornaviridae families. Int. J. Med. Chem. 2011;2011:213135. doi: 10.1155/2011/213135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bronnimann M.P., Calton C.M., Chiquette S.F., Li S., Lu M., Chapman J.A., Bratton K.N., Schlegel A.M., Campos S.K. Furin cleavage of L2 during Papillomavirus Infection: minimal dependence on Cyclophilins. J. Virol. 2016;90(14):6224–6234. doi: 10.1128/JVI.00038-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buck C.B., Thompson C.D., Pang Y.-Y.S., Lowy D.R., Schiller J.T. Maturation of Papillomavirus capsids. J. Virol. 2005;79(5):2839–2846. doi: 10.1128/JVI.79.5.2839-2846.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bustin S.A. Absolute quantification of mRNA using real-time reverse transcription polymerase chain reaction assays. J. Mol. Endocrinol. 2000;25(2):169–193. doi: 10.1677/jme.0.0250169. [DOI] [PubMed] [Google Scholar]
- Callahan L. HIV-1 virion-cell interactions: an electrostatic model of pathogenicity and syncytium formation. AIDS Res. Hum. Retrovir. 1994;10(3):231–233. doi: 10.1089/aid.1994.10.231. [DOI] [PubMed] [Google Scholar]
- Campagnola G., Gong P., Peersen O.B. High-throughput screening identification of poliovirus RNA-dependent RNA polymerase inhibitors. Antivir. Res. 2011;91(3):241–251. doi: 10.1016/j.antiviral.2011.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell E.M., Hope T.J. HIV-1 capsid: the multifaceted key player in HIV-1 infection. Nat. Rev. Microbiol. 2015;13(8):471–483. doi: 10.1038/nrmicro3503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell S., Rein A. In vitro assembly properties of human immunodeficiency virus type 1 Gag protein lacking the p6 domain. J. Virol. 1999;73(3):2270–2279. doi: 10.1128/jvi.73.3.2270-2279.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell S., Vogt V.M. Self-assembly in-vitro of purified Ca-Nc proteins from Rous-sarcoma virus and human-immunodeficiency-virus type-1. J. Virol. 1995;69(10):6487–6497. doi: 10.1128/jvi.69.10.6487-6497.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell S., Vogt V.M. In vitro assembly of virus-like particles with Rous sarcoma virus Gag deletion mutants: identification of the p10 domain as a morphological determinant in the formation of spherical particles. J. Virol. 1997;71(6):4425–4435. doi: 10.1128/jvi.71.6.4425-4435.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell S., Fisher R.J., Towler E.M., Fox S., Issaq H.J., Wolfe T., Phillips L.R., Rein A. Modulation of HIV-like particle assembly in vitro by inositol phosphates. Proc. Natl. Acad. Sci. U. S. A. 2001;98(19):10875–10879. doi: 10.1073/pnas.191224698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campsteijn C., Vietri M., Stenmark H. Novel ESCRT functions in cell biology: spiraling out of control? Curr. Opin. Cell Biol. 2016;41:1–8. doi: 10.1016/j.ceb.2016.03.008. [DOI] [PubMed] [Google Scholar]
- Cao X., Li Y., Jin X., Li Y., Guo F., Jin T. Molecular mechanism of divalent-metal-induced activation of NS3 helicase and insights into Zika virus inhibitor design. Nucleic Acids Res. 2016;44(21):10505–10514. doi: 10.1093/nar/gkw941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carloni G., Crema A., Valli M.B., Ponzetto A., Clementi M. HCV infection by cell-to-cell transmission: choice or necessity? Curr. Mol. Med. 2012;12(1):83–95. doi: 10.2174/156652412798376152. [DOI] [PubMed] [Google Scholar]
- Case J.B., Ashbrook A.W., Dermody T.S., Denison M.R. Mutagenesis of S-adenosyl-l-methionine-binding residues in coronavirus nsp14 N7-methyltransferase demonstrates differing requirements for genome translation and resistance to innate immunity. J. Virol. 2016;90(16):7248–7256. doi: 10.1128/JVI.00542-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cautain B., Hill R., de Pedro N., Link W. Components and regulation of nuclear transport processes. FEBS J. 2015;282(3):445–462. doi: 10.1111/febs.13163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavrois M., De Noronha C., Greene W.C. A sensitive and specific enzyme-based assay detecting HIV-1 virion fusion in primary T lymphocytes. Nat. Biotechnol. 2002;20(11):1151–1154. doi: 10.1038/nbt745. [DOI] [PubMed] [Google Scholar]
- Cavrois M., Neidleman J., Bigos M., Greene W.C. Fluorescence resonance energy transfer-based HIV-1 virion fusion assay. Methods in Molecular Biology (Clifton, N.J.) 2004;263:333–344. doi: 10.1385/1-59259-773-4:333. [DOI] [PubMed] [Google Scholar]
- Cavrois M., Neidleman J., Greene W.C. HIV-1 fusion assay. Bio-Protocol. 2014;4(16) doi: 10.21769/bioprotoc.1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chai L.J., Zhou J., Feng H., Lin J.J., Qian Z.S. A reversible fluorescence nanoswitch based on carbon quantum dots nanoassembly for detection of pyrophosphate ion. Sensors Actuators B Chem. 2015;220:138–145. [Google Scholar]
- Chen, Y., Cai, H., Pan, J.a., Xiang, N., Tien, P., Ahola, T., Guo, D., 2009. Functional screen reveals SARS coronavirus nonstructural protein nsp14 as a novel cap N7 methyltransferase. Proc. Natl. Acad. Sci. 106(9), 3484-3489. [DOI] [PMC free article] [PubMed]
- Chen H., Liu X., Li Z., Zhan P., De Clercq E. TSG101: a novel anti-HIV-1 drug target. Curr. Med. Chem. 2010;17(8):750–758. doi: 10.2174/092986710790514444. [DOI] [PubMed] [Google Scholar]
- Cheng S., Brooks C.L. Protein-protein interfaces in viral capsids are structurally unique. J. Mol. Biol. 2015;427(22):3613–3624. doi: 10.1016/j.jmb.2015.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng H., Schafer A., Soloveva V., Gharaibeh D., Kenny T., Retterer C., Zamani R., Bavari S., Peet N.P., Rong L. Identification of a coumarin-based antihistamine as an anti-filoviral entry inhibitor. Antivir. Res. 2017;145:24–32. doi: 10.1016/j.antiviral.2017.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheslock S.R., Poon D.T., Fu W., Rhodes T.D., Henderson L.E., Nagashima K., McGrath C.F., Hu W.S. Charged assembly helix motif in murine leukemia virus capsid: an important region for virus assembly and particle size determination. J. Virol. 2003;77(12):7058–7066. doi: 10.1128/JVI.77.12.7058-7066.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho J.B., Lee J.M., Ahn H.C., Jeong Y.J. Identification of a novel small molecule inhibitor against SARS coronavirus helicase. J. Microbiol. Biotechnol. 2015;25(12):2007–2010. doi: 10.4014/jmb.1507.07078. [DOI] [PubMed] [Google Scholar]
- Clarke J.N., Davies L.K., Calvert J.K., Gliddon B.L., Al Shujari W.H., Aloia A.L., Helbig K.J., Beard M.R., Pitson S.M., Carr J.M. Reduction in sphingosine kinase 1 influences the susceptibility to dengue virus infection by altering antiviral responses. J. General Virol. 2016;97(1):95–109. doi: 10.1099/jgv.0.000334. [DOI] [PubMed] [Google Scholar]
- Clementz M.A., Chen Z., Banach B.S., Wang Y., Sun L., Ratia K., Baez-Santos Y.M., Wang J., Takayama J., Ghosh A.K., Li K., Mesecar A.D., Baker S.C. Deubiquitinating and interferon antagonism activities of coronavirus papain-like proteases. J. Virol. 2010;84(9):4619–4629. doi: 10.1128/JVI.02406-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clercq E.D. Emerging antiviral drugs. Expert Opinion on Emerging Drugs. 2008;13(3):393–416. doi: 10.1517/14728214.13.3.393. [DOI] [PubMed] [Google Scholar]
- Cliffe A.R., Wilson A.C. Restarting lytic gene transcription at the onset of herpes simplex virus reactivation. J. Virol. 2017;91(2):e01419–01416. doi: 10.1128/JVI.01419-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coates J.A., Cammack N., Jenkinson H.J., Jowett A.J., Jowett M.I., Pearson B.A., Penn C.R., Rouse P.L., Viner K.C., Cameron J.M. (-)-2'-deoxy-3'-thiacytidine is a potent, highly selective inhibitor of human immunodeficiency virus type 1 and type 2 replication in vitro. Antimicrob. Agents Chemother. 1992;36(4):733–739. doi: 10.1128/aac.36.4.733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen S., Pante N. Pushing the envelope: microinjection of minute virus of mice into Xenopus oocytes causes damage to the nuclear envelope. J. General Virol. 2005;86(Pt 12):3243–3252. doi: 10.1099/vir.0.80967-0. [DOI] [PubMed] [Google Scholar]
- Cohen S., Au S., Panté N. How viruses access the nucleus. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 2011;1813(9):1634–1645. doi: 10.1016/j.bbamcr.2010.12.009. [DOI] [PubMed] [Google Scholar]
- Coleman C.M., Sisk J.M., Mingo R.M., Nelson E.A., White J.M., Frieman M.B. Abelson kinase inhibitors are potent inhibitors of severe acute respiratory syndrome coronavirus and middle east respiratory syndrome coronavirus fusion. J. Virol. 2016;90(19):8924–8933. doi: 10.1128/JVI.01429-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coloma J., Jain R., Rajashankar K.R., García-Sastre A., Aggarwal A.K. Structures of NS5 methyltransferase from Zika virus. Cell Rep. 2016;16(12):3097–3102. doi: 10.1016/j.celrep.2016.08.091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colpitts C.C., El-Saghire H., Pochet N., Schuster C., Baumert T.F. High-throughput approaches to unravel hepatitis C virus-host interactions. Virus Res. 2016;218:18–24. doi: 10.1016/j.virusres.2015.09.013. [DOI] [PubMed] [Google Scholar]
- Condezo G.N., San Martín C. Localization of adenovirus morphogenesis players, together with visualization of assembly intermediates and failed products, favor a model where assembly and packaging occur concurrently at the periphery of the replication center. PLoS Pathog. 2017;13(4):e1006320. doi: 10.1371/journal.ppat.1006320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cossart P., Helenius A. Endocytosis of viruses and bacteria. Cold Spring Harb. Perspect. Biol. 2014;6(8):a016972. doi: 10.1101/cshperspect.a016972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coutard B., Barral K., Lichiere J., Selisko B., Martin B., Aouadi W., Lombardia M.O., Debart F., Vasseur J.J., Guillemot J.C., Canard B., Decroly E. Zika Virus methyltransferase: structure and functions for drug design perspectives. J. Virol. 2017;91(5) doi: 10.1128/JVI.02202-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox R.M., Plemper R.K. Structure and organization of paramyxovirus particles. Curr. Opin. Virol. 2017;24:105–114. doi: 10.1016/j.coviro.2017.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cummins N.W., Badley A.D. Mechanisms of HIV-associated lymphocyte apoptosis: 2010. Cell Death Dis. 2010;1:e99. doi: 10.1038/cddis.2010.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Da Silva Santos C., Tartour K., Cimarelli A. A novel entry/uncoating assay reveals the presence of at least two species of viral capsids during synchronized HIV-1 infection. PLoS Pathog. 2016;12(9):e1005897. doi: 10.1371/journal.ppat.1005897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasmahapatra B., DiDomenico B., Dwyer S., Ma J., Sadowski I., Schwartz J. A genetic system for studying the activity of a proteolytic enzyme. Proc. Natl. Acad. Sci. U. S. A. 1992;89(9):4159–4162. doi: 10.1073/pnas.89.9.4159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Clercq E. Antiviral drugs in current clinical use. J. Clin. Virol. 2004;30(2):115–133. doi: 10.1016/j.jcv.2004.02.009. [DOI] [PubMed] [Google Scholar]
- De Clercq E., Li G. Approved antiviral drugs over the past 50 years. Clin. Microbiol. Rev. 2016;29(3):695–747. doi: 10.1128/CMR.00102-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debruyne I. Inorganic phosphate determination: colorimetric assay based on the formation of a rhodamine B-phosphomolybdate complex. Anal. Biochem. 1983;130(2):454–460. doi: 10.1016/0003-2697(83)90615-2. [DOI] [PubMed] [Google Scholar]
- Degorce F., Card A., Soh S., Trinquet E., Knapik G.P., Xie B. HTRF: a technology tailored for drug discovery –a review of theoretical aspects and recent applications. Current Chemical Genomics. 2009;3:22–32. doi: 10.2174/1875397300903010022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deka J., Mojumdar A., Parisse P., Onesti S., Casalis L. DNA-conjugated gold nanoparticles based colorimetric assay to assess helicase activity: a novel route to screen potential helicase inhibitors. Sci. Rep. 2017;7:44358. doi: 10.1038/srep44358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delgui L.R., Colombo M.I. A novel mechanism underlying the innate immune response induction upon viral-dependent replication of host cell mRNA: a mistake of + sRNA viruses' replicases. Front. Cell. Infect. Microbiol. 2017;7(5) doi: 10.3389/fcimb.2017.00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devkota A.K., Warthaka M., Edupuganti R., Tavares C.D.J., Johnson W.H., Ozpolat B., Cho E.J., Dalby K.N. High-throughput screens for eEF-2 kinase. J. Biomol. Screen. 2013;19(3):445–452. doi: 10.1177/1087057113505204. [DOI] [PubMed] [Google Scholar]
- Ding X., Yang K.-L. Quantitative serine protease assays based on formation of copper(ii)-oligopeptide complexes. Analyst. 2015;140(1):340–345. doi: 10.1039/c4an01731e. [DOI] [PubMed] [Google Scholar]
- Dolezal M., Hadravova R., Kozisek M., Bednarova L., Langerova H., Ruml T., Rumlova M. Functional and structural characterization of novel type of linker connecting capsid and nucleocapsid protein domains in murine leukemia virus. J. Biol. Chem. 2016;291(39):20630–20642. doi: 10.1074/jbc.M116.746461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong H., Chang D.C., Hua M.H.C., Lim S.P., Chionh Y.H., Hia F., Lee Y.H., Kukkaro P., Lok S.-M., Dedon P.C., Shi P.-Y. 2′-O methylation of internal adenosine by flavivirus NS5 methyltransferase. PLoS Pathog. 2012;8(4):e1002642. doi: 10.1371/journal.ppat.1002642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driscoll M.D., Rentergent J., Hay S. A quantitative fluorescence-based steady-state assay of DNA polymerase. FEBS J. 2014;281(8):2042–2050. doi: 10.1111/febs.12760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duan W., Song H., Wang H., Chai Y., Su C., Qi J., Shi Y., Gao G.F. The crystal structure of Zika virus NS5 reveals conserved drug targets. EMBO J. 2017;36(7):919–933. doi: 10.15252/embj.201696241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards M.R., Pietzsch C., Vausselin T., Shaw M.L., Bukreyev A., Basler C.F. High-throughput minigenome system for identifying small-molecule inhibitors of ebola virus replication. ACS Infect. Dis. 2015;1(8):380–387. doi: 10.1021/acsinfecdis.5b00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrlich L.S., Agresta B.E., Carter C.A. Assembly of recombinant human immunodeficiency virus type 1 capsid protein in vitro. J. Virol. 1992;66(8):4874–4883. doi: 10.1128/jvi.66.8.4874-4883.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elshabrawy H.A., Fan J., Haddad C.S., Ratia K., Broder C.C., Caffrey M., Prabhakar B.S. Identification of a broad-spectrum antiviral small molecule against severe acute respiratory syndrome coronavirus and ebola, hendra, and nipah viruses by using a novel high-throughput screening assay. J. Virol. 2014;88(8):4353–4365. doi: 10.1128/JVI.03050-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelman A., Cherepanov P. Retroviral integrase structure and DNA recombination mechanism. Microbiol. Spectrum. 2014;2(6) doi: 10.1128/microbiolspec.MDNA3-0024-2014. [DOI] [PubMed] [Google Scholar]
- Fabian M.A., Biggs W.H., Treiber D.K., Atteridge C.E., Azimioara M.D., Benedetti M.G., Carter T.A., Ciceri P., Edeen P.T., Floyd M., Ford J.M., Galvin M., Gerlach J.L., Grotzfeld R.M., Herrgard S., Insko D.E., Insko M.A., Lai A.G., Lelias J.-M., Mehta S.A., Milanov Z.V., Velasco A.M., Wodicka L.M., Patel H.K., Zarrinkar P.P., Lockhart D.J. A small molecule-kinase interaction map for clinical kinase inhibitors. Nat. Biotechnol. 2005;23(3):329–336. doi: 10.1038/nbt1068. [DOI] [PubMed] [Google Scholar]
- Falcon V., Acosta-Rivero N., Gonzalez S., Duenas-Carrera S., Martinez-Donato G., Menendez I., Garateix R., Silva J.A., Acosta E., Kouri J. Ultrastructural and biochemical basis for hepatitis C virus morphogenesis. Virus Genes. 2017;53(2):151–164. doi: 10.1007/s11262-017-1426-2. [DOI] [PubMed] [Google Scholar]
- Fay N., Panté N. Nuclear entry of DNA viruses. Front. Microbiol. 2015;6(467) doi: 10.3389/fmicb.2015.00467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feltrup T.M., Singh B.R. Development of a fluorescence internal quenching correction factor to correct BoNT/A endopeptidase kinetics using SNAPtide. Anal. Chem. 2012;84(24):10549–10553. doi: 10.1021/ac302997n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenaux M., Lin X., Yokokawa F., Sweeney Z., Saunders O., Xie L., Lim S.P., Uteng M., Uehara K., Warne R., Gang W., Jones C., Yendluri S., Gu H., Mansfield K., Boisclair J., Heimbach T., Catoire A., Bracken K., Weaver M., Moser H., Zhong W. Antiviral nucleotide incorporation by recombinant human mitochondrial RNA polymerase is predictive of increased in vivo mitochondrial toxicity risk. Antimicrob. Agents Chemother. 2016;60(12):7077–7085. doi: 10.1128/AAC.01253-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng Z., Hensley L., McKnight K.L., Hu F., Madden V., Ping L., Jeong S.-H., Walker C., Lanford R.E., Lemon S.M. A pathogenic picornavirus acquires an envelope by hijacking cellular membranes. Nature. 2013;496(7445):367–371. doi: 10.1038/nature12029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng J.Y., Cheng G., Perry J., Barauskas O., Xu Y., Fenaux M., Eng S., Tirunagari N., Peng B., Yu M., Tian Y., Lee Y.-J., Stepan G., Lagpacan L.L., Jin D., Hung M., Ku K.S., Han B., Kitrinos K., Perron M., Birkus G., Wong K.A., Zhong W., Kim C.U., Carey A., Cho A., Ray A.S. Inhibition of hepatitis C virus replication by GS-6620, a potent C-nucleoside monophosphate prodrug. Antimicrob. Agents Chemother. 2014;58(4):1930–1942. doi: 10.1128/AAC.02351-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferron F., Weber F., de la Torre J.C., Reguera J. Transcription and replication mechanisms of Bunyaviridae and Arenaviridae L proteins. Virus Res. 2017;234:118–134. doi: 10.1016/j.virusres.2017.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fields B.N., Knipe D.M., Howley P.M. 6th ed. Wolters Kluwer Health/Lippincott Williams & Wilkins; Philadelphia: 2013. Fields virology. [Google Scholar]
- Foote B.S., Spooner L.M., Belliveau P.P. Boceprevir: a protease inhibitor for the treatment of chronic Hepatitis C. Ann. Pharmacother. 2011;45(9):1085–1093. doi: 10.1345/aph.1P744. [DOI] [PubMed] [Google Scholar]
- Freed E.O. Viral late domains. J. Virol. 2002;76(10):4679–4687. doi: 10.1128/JVI.76.10.4679-4687.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freed E.O. HIV-1 assembly, release and maturation. Nat. Rev. Microbiol. 2015;13(8):484–496. doi: 10.1038/nrmicro3490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frick D.N. Helicases as antiviral drug targets. Drug News & Perspectives. 2003;16(6):355–362. doi: 10.1358/dnp.2003.16.6.829307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frieman M., Ratia K., Johnston R.E., Mesecar A.D., Baric R.S. Severe acute respiratory syndrome coronavirus papain-like protease ubiquitin-like domain and catalytic domain regulate antagonism of IRF3 and NF-κB signaling. J. Virol. 2009;83(13):6689–6705. doi: 10.1128/JVI.02220-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fromentin R., Majeau N., Laliberte Gagne M.E., Boivin A., Duvignaud J.B., Leclerc D. A method for in vitro assembly of hepatitis C virus core protein and for screening of inhibitors. Anal. Biochem. 2007;366(1):37–45. doi: 10.1016/j.ab.2007.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii K., Hurley J.H., Freed E.O. Beyond Tsg101: the role of Alix in 'ESCRTing' HIV-1. Nat. Rev. Microbiol. 2007;5(12):912–916. doi: 10.1038/nrmicro1790. [DOI] [PubMed] [Google Scholar]
- Gaba A., Ayalew L., Makadiya N., Tikoo S. Proteolytic cleavage of bovine adenovirus 3-encoded pVIII. J. Virol. 2017;91(10) doi: 10.1128/JVI.00211-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gan X., Wang H., Yu Y., Yi W., Zhu S., Li E., Liang Y. Epigenetically repressing human cytomegalovirus lytic infection and reactivation from latency in THP-1 model by targeting H3K9 and H3K27 histone demethylases. PLoS One. 2017;12(4):e0175390. doi: 10.1371/journal.pone.0175390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gastaminza P., Whitten-Bauer C., Chisari F.V. Unbiased probing of the entire hepatitis C virus life cycle identifies clinical compounds that target multiple aspects of the infection. Proc. Natl. Acad. Sci. U. S. A. 2010;107(1):291–296. doi: 10.1073/pnas.0912966107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gemene K.L., Meyerhoff M.E. Detection of protease activities by flash chronopotentiometry using a reversible polycation-sensitive polymeric membrane electrode. Anal. Biochem. 2011;416(1):67–73. doi: 10.1016/j.ab.2011.04.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh A.K., Osswald H.L., Prato G. Recent progress in the development of HIV-1 protease inhibitors for the treatment of HIV/AIDS. J. Med. Chem. 2016;59(11):5172–5208. doi: 10.1021/acs.jmedchem.5b01697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson W. Structure and formation of the cytomegalovirus virion. Curr. Top. Microbiol. Immunol. 2008;325:187–204. doi: 10.1007/978-3-540-77349-8_11. [DOI] [PubMed] [Google Scholar]
- Glebe D., Aliakbari M., Krass P., Knoop E.V., Valerius K.P., Gerlich W.H. Pre-s1 antigen-dependent infection of Tupaia hepatocyte cultures with human hepatitis B virus. J. Virol. 2003;77(17):9511–9521. doi: 10.1128/JVI.77.17.9511-9521.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glebe D., Urban S., Knoop E.V., Çaǧ N., Krass P., Grün S., Bulavaite A., Sasnauskas K., Gerlich W.H. Mapping of the Hepatitis B Virus attachment site by use of infection-inhibiting preS1 lipopeptides and tupaia hepatocytes. Gastroenterology. 2005;129(1):234–245. doi: 10.1053/j.gastro.2005.03.090. [DOI] [PubMed] [Google Scholar]
- Gomez C., Hope T.J. The ins and outs of HIV replication. Cell. Microbiol. 2005;7(5):621–626. doi: 10.1111/j.1462-5822.2005.00516.x. [DOI] [PubMed] [Google Scholar]
- Gotoh B., Ohnishi Y., Inocencio N.M., Esaki E., Nakayama K., Barr P.J., Thomas G., Nagai Y. Mammalian subtilisin-related proteinases in cleavage activation of the paramyxovirus fusion glycoprotein: superiority of furin/PACE to PC2 or PC1/PC3. J. Virol. 1992;66(11):6391–6397. doi: 10.1128/jvi.66.11.6391-6397.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant S.K., Sklar J.G., Cummings R.T. Development of novel assays for proteolytic enzymes using rhodamine-based fluorogenic substrates. J. Biomol. Screen. 2002;7(6):531–540. doi: 10.1177/1087057102238627. [DOI] [PubMed] [Google Scholar]
- Gripon P., Diot C., Theze N., Fourel I., Loreal O., Brechot C., Guguen-Guillouzo C. Hepatitis B virus infection of adult human hepatocytes cultured in the presence of dimethyl sulfoxide. J. Virol. 1988;62(11):4136–4143. doi: 10.1128/jvi.62.11.4136-4143.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gripon P., Rumin S., Urban S., Le Seyec J., Glaise D., Cannie I., Guyomard C., Lucas J., Trepo C., Guguen-Guillouzo C. Infection of a human hepatoma cell line by hepatitis B virus. Proc. Natl. Acad. Sci. 2002;99(24):15655–15660. doi: 10.1073/pnas.232137699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gripon P., Cannie I., Urban S. Efficient inhibition of Hepatitis B Virus infection by acylated peptides derived from the large viral surface protein. J. Virol. 2005;79(3):1613–1622. doi: 10.1128/JVI.79.3.1613-1622.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grønborg M., Kristiansen T.Z., Stensballe A., Andersen J.S., Ohara O., Mann M., Jensen O.N., Pandey A. A mass spectrometry-based proteomic approach for identification of serine/threonine-phosphorylated proteins by enrichment with phospho-specific antibodies: identification of a novel Protein, frigg, as a protein kinase A substrate. Mol. Cell. Proteomics. 2002;1(7):517–527. doi: 10.1074/mcp.m200010-mcp200. [DOI] [PubMed] [Google Scholar]
- Grose C., Buckingham E.M., Carpenter J.E., Kunkel J.P. Varicella-zoster virus infectious cycle: ER stress, autophagic flux, and amphisome-mediated trafficking. Pathogens (Basel, Switzerland) 2016;5(4) doi: 10.3390/pathogens5040067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross I., Hohenberg H., Wilk T., Wiegers K., Grattinger M., Muller B., Fuller S., Krausslich H.G. A conformational switch controlling HIV-1 morphogenesis. EMBO J. 2000;19(1):103–113. doi: 10.1093/emboj/19.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grove J., Marsh M. The cell biology of receptor-mediated virus entry. J. Cell Biol. 2011;195(7):1071–1082. doi: 10.1083/jcb.201108131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu M., Rice C.M. The spring α-helix coordinates multiple modes of HCV (Hepatitis C Virus) NS3 helicase action. J. Biol. Chem. 2016;291(28):14499–14509. doi: 10.1074/jbc.M115.704379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guiot E., Carayon K., Delelis O., Simon F., Tauc P., Zubin E., Gottikh M., Mouscadet J.F., Brochon J.C., Deprez E. Relationship between the oligomeric status of HIV-1 integrase on DNA and enzymatic activity. J. Biol. Chem. 2006;281(32):22707–22719. doi: 10.1074/jbc.M602198200. [DOI] [PubMed] [Google Scholar]
- Guu T.S., Zheng W., Tao Y.J. Bunyavirus: structure and replication. Adv. Exp. Med. Biol. 2012;726:245–266. doi: 10.1007/978-1-4614-0980-9_11. [DOI] [PubMed] [Google Scholar]
- Hadravova R., de Marco A., Ulbrich P., Stokrova J., Dolezal M., Pichova I., Ruml T., Briggs J.A., Rumlova M. In vitro assembly of virus-like particles of a gammaretrovirus, the murine leukemia virus XMRV. J. Virol. 2012;86(3):1297–1306. doi: 10.1128/JVI.05564-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadravova R., Rumlova M., Ruml T. FAITH - fast assembly inhibitor test for HIV. Virology. 2015;486:78–87. doi: 10.1016/j.virol.2015.08.029. [DOI] [PubMed] [Google Scholar]
- Hajimahdi Z., Zarghi A. Progress in HIV-1 integrase inhibitors: a review of their chemical structure diversity. Iran. J. Pharmaceut. Res. 2016;15(4):595–628. [PMC free article] [PubMed] [Google Scholar]
- Hall M.D., Yasgar A., Peryea T., Braisted J.C., Jadhav A., Simeonov A., Coussens N.P. Fluorescence polarization assays in high-throughput screening and drug discovery: a review. Methods Appl. Fluorescence. 2016;4(2):022001. doi: 10.1088/2050-6120/4/2/022001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallenberger S., Bosch V., Angliker H., Shaw E., Klenk H.-D., Garten W. Inhibition of furin-mediated cleavage activation of HIV-1 glycoprotein gpl60. Nature. 1992;360(6402):358–361. doi: 10.1038/360358a0. [DOI] [PubMed] [Google Scholar]
- Han I.S., Ramamurthy N., Yun J.H., Schaller U., Meyerhoff M.E., Yang V.C. Selective monitoring of peptidase activities with synthetic polypeptide substrates and polyion-sensitive membrane electrode detection. FASEB J. 1996;10(14):1621–1626. doi: 10.1096/fasebj.10.14.9002554. [DOI] [PubMed] [Google Scholar]
- Han Y.-S., Xiao W.-L., Quashie P.K., Mesplède T., Xu H., Deprez E., Delelis O., Pu J.-X., Sun H.-D., Wainberg M.A. Development of a fluorescence-based HIV-1 integrase DNA binding assay for identification of novel HIV-1 integrase inhibitors. Antivir. Res. 2013;98(3):441–448. doi: 10.1016/j.antiviral.2013.04.001. [DOI] [PubMed] [Google Scholar]
- Han Z., Madara J.J., Liu Y., Liu W., Ruthel G., Freedman B.D., Harty R.N. ALIX rescues budding of a Double PTAP/PPEY L-Domain Deletion Mutant of Ebola VP40: a role for ALIX in Ebola Virus Egress. J. Infect. Dis. 2015;212(Suppl. 2):S138–145. doi: 10.1093/infdis/jiu838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hao W., Wojdyla J.A., Zhao R., Han R., Das R., Zlatev I., Manoharan M., Wang M., Cui S. Crystal structure of Middle East respiratory syndrome coronavirus helicase. PLoS Pathog. 2017;13(6):e1006474. doi: 10.1371/journal.ppat.1006474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He H.Q., Ma X.H., Liu B., Zhang X.Y., Chen W.Z., Wang C.X., Cheng S.H. High-throughput real-time assay based on molecular beacons for HIV-1 integrase 3'-processing reaction. Acta Pharmacol. Sin. 2007;28(6):811–817. doi: 10.1111/j.1745-7254.2007.00561.x. [DOI] [PubMed] [Google Scholar]
- He H.Q., Ma X.H., Liu B., Chen W.Z., Wang C.X., Cheng S.H. A novel high-throughput format assay for HIV-1 integrase strand transfer reaction using magnetic beads. Acta Pharmacol. Sin. 2008;29(3):397–404. doi: 10.1111/j.1745-7254.2008.00748.x. [DOI] [PubMed] [Google Scholar]
- Hellberg T., Passvogel L., Schulz K.S., Klupp B.G., Mettenleiter T.C. Nuclear Egress of Herpesviruses: the prototypic vesicular nucleocytoplasmic transport. Adv. Virus Res. 2016;94:81–140. doi: 10.1016/bs.aivir.2015.10.002. [DOI] [PubMed] [Google Scholar]
- Heming J.D., Conway J.F., Homa F.L. Herpesvirus capsid assembly and DNA packaging. Adv. Anat. Embryol. Cell Biol. 2017;223:119–142. doi: 10.1007/978-3-319-53168-7_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herschhorn, A., Finzi, A., Jones, D.M., Courter, J.R., Sugawara, A., Smith, A.B., III, Sodroski, J.G., 2011. An inducible cell-cell fusion system with integrated ability to measure the efficiency and specificity of HIV-1 entry inhibitors. PLoS One 6(11), e26731. [DOI] [PMC free article] [PubMed]
- Hill, J.M., Quenelle, D.C., Cardin, R.D., Vogel, J.L., Clement, C., Bravo, F.J., Foster, T.P., Bosch-Marce, M., Raja, P., Lee, J.S., Bernstein, D.I., Krause, P.R., Knipe, D.M., Kristie, T.M., 2014. Inhibition of LSD1 reduces herpesvirus infection, shedding, and recurrence by promoting epigenetic suppression of viral genomes. Sci. Transl. Med. 6(265), 265ra169-265ra169. [DOI] [PMC free article] [PubMed]
- Hilton B.J., Wolkowicz R. An assay to monitor HIV-1 protease activity for the identification of novel inhibitors in T-cells. PLoS One. 2010;5(6):e10940. doi: 10.1371/journal.pone.0010940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirashima S., Suzuki T., Ishida T., Noji S., Yata S., Ando I., Komatsu M., Ikeda S., Hashimoto H. Benzimidazole derivatives bearing substituted biphenyls as Hepatitis C Virus NS5B RNA-Dependent RNA polymerase inhibitors: structure − activity relationship studies and identification of a potent and highly selective inhibitor JTK-109. J. Med. Chem. 2006;49(15):4721–4736. doi: 10.1021/jm060269e. [DOI] [PubMed] [Google Scholar]
- Höfer K., Langejürgen L.V., Jäschke A. Universal aptamer-based real-time monitoring of enzymatic RNA synthesis. J. Am. Chem. Soc. 2013;135(37):13692–13694. doi: 10.1021/ja407142f. [DOI] [PubMed] [Google Scholar]
- Holden M.J., Haynes R.J., Rabb S.A., Satija N., Yang K., Blasic J.R. Factors affecting quantification of total DNA by UV spectroscopy and picogreen fluorescence. J. Agric. Food Chem. 2009;57(16):7221–7226. doi: 10.1021/jf901165h. [DOI] [PubMed] [Google Scholar]
- Hu J., Liu F., Ju H. Peptide code-on-a-microplate for protease activity analysis via MALDI-TOF mass spectrometric quantitation. Anal. Chem. 2015;87(8):4409–4414. doi: 10.1021/acs.analchem.5b00230. [DOI] [PubMed] [Google Scholar]
- Huang S.-Y., Tsai M.-L., Chen G.-Y., Wu C.-J., Chen S.-H. A systematic MS-based approach for identifying in vitro substrates of PKA and PKG in Rat Uteri. J. Proteome Res. 2007;6(7):2674–2684. doi: 10.1021/pr070134c. [DOI] [PubMed] [Google Scholar]
- Hübner W., McNerney G.P., Chen P., Dale B.M., Gordon R.E., Chuang F.Y.S., Li X.-D., Asmuth D.M., Huser T., Chen B.K. Quantitative 3D Video microscopy of HIV transfer across T cell virological synapses. Science. 2009;323(5922):1743–1747. doi: 10.1126/science.1167525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hulme A.E., Perez O., Hope T.J. Complementary assays reveal a relationship between HIV-1 uncoating and reverse transcription. Proc. Natl. Acad. Sci. U. S. A. 2011;108(24):9975–9980. doi: 10.1073/pnas.1014522108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hulo C., Masson P., de Castro E., Auchincloss A.H., Foulger R., Poux S., Lomax J., Bougueleret L., Xenarios I., Le Mercier P. The ins and outs of eukaryotic viruses: Knowledge base and ontology of a viral infection. PLoS One. 2017;12(2):e0171746. doi: 10.1371/journal.pone.0171746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurley J.H. ESCRTs are everywhere. EMBO J. 2015;34(19):2398–2407. doi: 10.15252/embj.201592484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutchinson E.C., Fodor E. Transport of the influenza virus genome from nucleus to nucleus. Viruses. 2013;5(10):2424–2446. doi: 10.3390/v5102424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutin S., Ling W.L., Round A., Effantin G., Reich S., Iseni F., Tarbouriech N., Schoehn G., Burmeister W.P. Domain organization of vaccinia virus Helicase-Primase D5. J. Virol. 2016;90(9):4604–4613. doi: 10.1128/JVI.00044-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue T., Tsai B. How viruses use the endoplasmic reticulum for entry, replication, and assembly. Cold Spring Harb. Perspect. Biol. 2013;5(1):a013250. doi: 10.1101/cshperspect.a013250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isel, C., Munier, S., Naffakh, N., 2016. Experimental approaches to study genome packaging of influenza A viruses. Viruses 8(8). [DOI] [PMC free article] [PubMed]
- Jackson W.T. Poliovirus-induced changes in cellular membranes throughout infection. Curr. Opin. Virol. 2014;9:67–73. doi: 10.1016/j.coviro.2014.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson, W.T., Giddings, T.H., Jr., Taylor, M.P., Mulinyawe, S., Rabinovitch, M., Kopito, R.R., Kirkegaard, K., 2005. Subversion of cellular autophagosomal machinery by RNA viruses. PLoS Biol. 3(5), e156. [DOI] [PMC free article] [PubMed]
- Jacob T., Van den Broeke C., Favoreel H.W. Viral Serine/Threonine protein kinases. J. Virol. 2011;85(3):1158–1173. doi: 10.1128/JVI.01369-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain R., Coloma J., Garcia-Sastre A., Aggarwal A.K. Structure of the NS3 helicase from Zika virus. Nat. Struct. Mol. Biol. 2016;23(8):752–754. doi: 10.1038/nsmb.3258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang H., Ryoo S.-R., Lee M.J., Han S.W., Min D.-H. A new helicase assay based on graphene oxide for anti-viral drug development. Mol. Cell. 2013;35(4):269–273. doi: 10.1007/s10059-013-0066-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jasenosky L.D., Kawaoka Y. Filovirus budding. Virus Res. 2004;106(2):181–188. doi: 10.1016/j.virusres.2004.08.014. [DOI] [PubMed] [Google Scholar]
- Jayaraman B., Smith A.M., Fernandes J.D., Frankel A.D. Oligomeric viral proteins: small in size, large in presence. Crit. Rev. Biochem. Mol. Biol. 2016;51(5):379–394. doi: 10.1080/10409238.2016.1215406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen S., Thomsen A.R. Sensing of RNA viruses: a review of innate immune receptors involved in recognizing RNA virus invasion. J. Virol. 2012;86(6):2900–2910. doi: 10.1128/JVI.05738-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji C., Zhang J., Cammack N., Sankuratri S. Development of a novel dual CCR5-dependent and CXCR4-dependent cell-cell fusion assay system with inducible gp160 expression. J. Biomol. Screen. 2006;11(1):65–74. doi: 10.1177/1087057105282959. [DOI] [PubMed] [Google Scholar]
- Jiang S., Lin K., Zhang L., Debnath A.K. A screening assay for antiviral compounds targeted to the HIV-1 gp41 core structure using a conformation-specific monoclonal antibody. J. Virol. Methods. 1999;80(1):85–96. doi: 10.1016/s0166-0934(99)00041-5. [DOI] [PubMed] [Google Scholar]
- John, S., Fletcher, T.M., 3rd, Jonsson, C.B., 2005. Development and application of a high-throughput screening assay for HIV-1 integrase enzyme activities. J. Biomol. Screen. 10(6), 606-614. [DOI] [PubMed]
- Jones R.C. Avian reovirus infections. Revue scientifique et technique (International Office of Epizootics) 2000;19(2):614–625. doi: 10.20506/rst.19.2.1237. [DOI] [PubMed] [Google Scholar]
- Jose J., Snyder J.E., Kuhn R.J. A structural and functional perspective of alphavirus replication and assembly. Future Microbiol. 2009;4:837–856. doi: 10.2217/fmb.09.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi S., Chen L., Winter M.B., Lin Y.-L., Yang Y., Shapovalova M., Smith P.M., Liu C., Li F., LeBeau A.M. The rational design of therapeutic peptides for Aminopeptidase N using a substrate-based approach. Sci. Rep. 2017;7(1424) doi: 10.1038/s41598-017-01542-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyce C.M. Techniques used to study the DNA polymerase reaction pathway. Biochim. Biophys. Acta. 2010;1804(5):1032–1040. doi: 10.1016/j.bbapap.2009.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato A. Molecular mechanism by which Us3 protein kinase regulates the pathogenicity of herpes simplex virus type-1. Uirusu. 2016;66(1):83–90. doi: 10.2222/jsv.66.83. [DOI] [PubMed] [Google Scholar]
- Kato T., Date T., Murayama A., Morikawa K., Akazawa D., Wakita T. Cell culture and infection system for hepatitis C virus. Nat. Protoc. 2006;1(5):2334–2339. doi: 10.1038/nprot.2006.395. [DOI] [PubMed] [Google Scholar]
- Katz R.A., Merkel G., Kulkosky J., Leis J., Skalka A.M. The avian retroviral IN protein is both necessary and sufficient for integrative recombination in vitro. Cell. 1990;63(1):87–95. doi: 10.1016/0092-8674(90)90290-u. [DOI] [PubMed] [Google Scholar]
- Katzman M., Katz R.A., Skalka A.M., Leis J. The avian retroviral integration protein cleaves the terminal sequences of linear viral DNA at the in vivo sites of integration. J. Virol. 1989;63(12):5319–5327. doi: 10.1128/jvi.63.12.5319-5327.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawaguchi Y., Kato K. Protein kinases conserved in herpesviruses potentially share a function mimicking the cellular protein kinase cdc2. Rev. Med. Virol. 2003;13(5):331–340. doi: 10.1002/rmv.402. [DOI] [PubMed] [Google Scholar]
- Khalilzadeh B., Shadjou N., Afsharan H., Eskandani M., Nozad Charoudeh H., Rashidi M.-R. Reduced graphene oxide decorated with gold nanoparticle as signal amplification element on ultra-sensitive electrochemiluminescence determination of caspase-3 activity and apoptosis using peptide based biosensor. BioImpacts. 2016;6(3):135–147. doi: 10.15171/bi.2016.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilianski A., Mielech A.M., Deng X., Baker S.C. Assessing activity and inhibition of middle east respiratory syndrome coronavirus papain-like and 3C-Like proteases using luciferase-based biosensors. J. Virol. 2013;87(21):11955–11962. doi: 10.1128/JVI.02105-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirkham M., Parton R.G. Clathrin-independent endocytosis: new insights into caveolae and non-caveolar lipid raft carriers. Biochim. Biophys. Acta. 2005;1746(3):349–363. doi: 10.1016/j.bbamcr.2005.11.007. [DOI] [PubMed] [Google Scholar]
- Klikova M., Rhee S.S., Hunter E., Ruml T. Efficient in-vivo and in-vitro assembly of retroviral capsids from gag precursor proteins expressed in bacteria. J. Virol. 1995;69(2):1093–1098. doi: 10.1128/jvi.69.2.1093-1098.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobiler O., Drayman N., Butin-Israeli V., Oppenheim A. Virus strategies for passing the nuclear envelope barrier. Nucleus. 2012;3(6):526–539. doi: 10.4161/nucl.21979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokoris M.S., Black M.E. Characterization of herpes simplex virus type 1 thymidine kinase mutants engineered for improved ganciclovir or acyclovir activity. Protein Sci. 2002;11(9):2267–2272. doi: 10.1110/ps.2460102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konvalinka J., Kräusslich H.-G., Müller B. Retroviral proteases and their roles in virion maturation. Virology. 2015;479:403–417. doi: 10.1016/j.virol.2015.03.021. [DOI] [PubMed] [Google Scholar]
- Koonin E.V., Yutin N. Origin and evolution of eukaryotic large nucleo-cytoplasmic DNA viruses. Intervirology. 2010;53(5):284–292. doi: 10.1159/000312913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kremer E.J., Nemerow G.R. Adenovirus tales: from the cell surface to the nuclear pore complex. PLoS Pathog. 2015;11(6):e1004821. doi: 10.1371/journal.ppat.1004821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kühn N., Bergmann S., Kösterke N., Lambertz R.L.O., Keppner A., van den Brand J.M.A., Pöhlmann S., Weiß S., Hummler E., Hatesuer B., Schughart K. The proteolytic activation of (H3N2) Influenza A Virus Hemagglutinin is facilitated by different type II transmembrane serine proteases. J. Virol. 2016;90(9):4298–4307. doi: 10.1128/JVI.02693-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo C.J., Chi Y.H., Hsu J.T., Liang P.H. Characterization of SARS main protease and inhibitor assay using a fluorogenic substrate. Biochem. Biophys. Res. Commun. 2004;318(4):862–867. doi: 10.1016/j.bbrc.2004.04.098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwong A.D., Rao B.G., Jeang K.T. Viral and cellular RNA helicases as antiviral targets. Nat. Rev. Drug Discov. 2005;4(10):845–853. doi: 10.1038/nrd1853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ladner S.K., Otto M.J., Barker C.S., Zaifert K., Wang G.H., Guo J.T., Seeger C., King R.W. Inducible expression of human hepatitis B virus (HBV) in stably transfected hepatoblastoma cells: a novel system for screening potential inhibitors of HBV replication. Antimicrob. Agents Chemother. 1997;41(8):1715–1720. doi: 10.1128/aac.41.8.1715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai J.K.F., Sam I.C., Chan Y.F. The autophagic machinery in enterovirus infection. Viruses. 2016;8(2):32. doi: 10.3390/v8020032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanman J., Sexton J., Sakalian M., Prevelige P.E., Jr. Kinetic analysis of the role of intersubunit interactions in human immunodeficiency virus type 1 capsid protein assembly in vitro. J. Virol. 2002;76(14):6900–6908. doi: 10.1128/JVI.76.14.6900-6908.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laporte M., Naesens L. Airway proteases: an emerging drug target for influenza and other respiratory virus infections. Curr. Opin. Virol. 2017;24:16–24. doi: 10.1016/j.coviro.2017.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lathia U.S., Ornatsky O., Baranov V., Nitz M. Multiplexed protease assays using element-tagged substrates. Anal. Biochem. 2011;408(1):157–159. doi: 10.1016/j.ab.2010.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lea W.A., Simeonov A. Fluorescence polarization assays in small molecule screening. Expert Opin. Drug Discovery. 2011;6(1):17–32. doi: 10.1517/17460441.2011.537322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H., Kim Y.-P. Fluorescent and bioluminescent nanoprobes for in vitro and in vivo detection of matrix metalloproteinase activity. BMB Rep. 2015;48(6):313–318. doi: 10.5483/BMBRep.2015.48.6.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H., Mittal A., Patel K., Gatuz J.L., Truong L., Torres J., Mulhearn D.C., Johnson M.E. Identification of novel drug scaffolds for inhibition of SARS-CoV 3-Chymotrypsin-like protease using virtual and high-throughput screenings. Bioorg. Med. Chem. 2014;22(1):167–177. doi: 10.1016/j.bmc.2013.11.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J.-C., Tseng C.-K., Wu Y.-H., Kaushik-Basu N., Lin C.-K., Chen W.-C., Wu H.-N. Characterization of the activity of 2′-C-methylcytidine against dengue virus replication. Antivir. Res. 2015;116:1–9. doi: 10.1016/j.antiviral.2015.01.002. [DOI] [PubMed] [Google Scholar]
- Lemke C.T., Titolo S., von Schwedler U., Goudreau N., Mercier J.F., Wardrop E., Faucher A.M., Coulombe R., Banik S.S., Fader L., Gagnon A., Kawai S.H., Rancourt J., Tremblay M., Yoakim C., Simoneau B., Archambault J., Sundquist W.I., Mason S.W. Distinct effects of two HIV-1 capsid assembly inhibitor families that bind the same site within the N-terminal domain of the viral CA protein. J. Virol. 2012;86(12):6643–6655. doi: 10.1128/JVI.00493-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leuchowius K.-J., Jarvius M., Wickström M., Rickardson L., Landegren U., Larsson R., Söderberg O., Fryknäs M., Jarvius J. High content screening for inhibitors of protein interactions and post-translational modifications in primary cells by proximity ligation. Mol. Cell. Proteomics. 2010;9(1):178–183. doi: 10.1074/mcp.M900331-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung K.-H., He H.-Z., He B., Zhong H.-J., Lin S., Wang Y.-T., Ma D.-L., Leung C.-H. Label-free luminescence switch-on detection of hepatitis C virus NS3 helicase activity using a G-quadruplex-selective probe †Electronic supplementary information (ESI) available: Compound characterisation and supplementary data. See DOI: 10.1039/c4sc03319a Click here for additional data file. Chem. Sci. 2015;6(4):2166–2171. doi: 10.1039/c4sc03319a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levings R.L., Roth J.A. Immunity to bovine herpesvirus 1: I. Viral lifecycle and innate immunity. Anim. Health Res. Rev. 2013;14(1):88–102. doi: 10.1017/S1466252313000042. [DOI] [PubMed] [Google Scholar]
- Li M., Youngren J.F., Manchem V.P., Kozlowski M., Zhang B.B., Maddux B.A., Goldfine I.D. Small molecule insulin receptor activators potentiate insulin action in insulin-resistant cells. Diabetes. 2001;50(10):2323. doi: 10.2337/diabetes.50.10.2323. [DOI] [PubMed] [Google Scholar]
- Li J., Fontaine-Rodriguez E.C., Whelan S.P.J. Amino acid residues within conserved domain VI of the vesicular stomatitis virus large polymerase protein essential for mRNA cap methyltransferase activity. J. Virol. 2005;79(21):13373–13384. doi: 10.1128/JVI.79.21.13373-13384.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J., Chorba J.S., Whelan S.P. Vesicular stomatitis viruses resistant to the methylase inhibitor sinefungin upregulate RNA synthesis and reveal mutations that affect mRNA cap methylation. J. Virol. 2007;81(8):4104–4115. doi: 10.1128/JVI.02681-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Xie W., Fang G. Fluorescence detection techniques for protein kinase assay. Anal. Bioanal. Chem. 2008;390(8):2049–2057. doi: 10.1007/s00216-008-1986-z. [DOI] [PubMed] [Google Scholar]
- Li X., Deng D., Xue J., Qu L., Achilefu S., Gu Y. Quantum dots based molecular beacons for in vitro and in vivo detection of MMP-2 on tumor. Biosens. Bioelectron. 2014;61:512–518. doi: 10.1016/j.bios.2014.05.035. [DOI] [PubMed] [Google Scholar]
- Liang Y., Vogel J.L., Narayanan A., Peng H., Kristie T.M. Inhibition of the histone demethylase LSD1 blocks α-herpesvirus lytic replication and reactivation from latency. Nat. Med. 2009;15(11):1312–1317. doi: 10.1038/nm.2051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang Y., Quenelle D., Vogel J.L., Mascaro C., Ortega A., Kristie T.M. A novel selective LSD1/KDM1A inhibitor epigenetically blocks herpes simplex virus lytic replication and reactivation from latency. MBio. 2013;4(1):e00512–e00558. doi: 10.1128/mBio.00558-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao, C., Marchand, C., Burke, T.R., Jr., Pommier, Y., Nicklaus, M.C., 2010. Authentic HIV-1 integrase inhibitors. Future Med. Chem. 2(7), 1107-1122. [DOI] [PMC free article] [PubMed]
- Lin C.T., Tritschler F., Lee K.S., Gu M., Rice C.M., Ha T. Single-molecule imaging reveals the translocation and DNA looping dynamics of hepatitis C virus NS3 helicase. Protein Sci. 2017;26(7):1391–1403. doi: 10.1002/pro.3136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindsten, K., Uhlíková, T.á., Konvalinka, J., Masucci, M.G., Dantuma, N.P., 2001. Cell-based fluorescence assay for human immunodeficiency virus Type 1 protease activity. Antimicrob. Agents Chemother. 45(9), 2616-2622. [DOI] [PMC free article] [PubMed]
- Liu Y., Harty R.N. Viral and host proteins that modulate filovirus budding. Futur. Virol. 2010;5(4):481–491. doi: 10.2217/FVL.10.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S., Boyer-Chatenet L., Lu H., Jiang S. Rapid and automated fluorescence-linked immunosorbent assay for high-throughput screening of HIV-1 fusion inhibitors targeting gp41. J. Biomol. Screen. 2003;8(6):685–693. doi: 10.1177/1087057103259155. [DOI] [PubMed] [Google Scholar]
- Lo M.K., Jordan R., Arvey A., Sudhamsu J., Shrivastava-Ranjan P., Hotard A.L., Flint M., McMullan L.K., Siegel D., Clarke M.O., Mackman R.L., Hui H.C., Perron M., Ray A.S., Cihlar T., Nichol S.T., Spiropoulou C.F. GS-5734 and its parent nucleoside analog inhibit Filo-, Pneumo-, and Paramyxoviruses. Sci. Rep. 2017;7:43395. doi: 10.1038/srep43395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu G., Gong P. A structural view of the RNA-dependent RNA polymerases from the Flavivirus genus. Virus Res. 2017;234:34–43. doi: 10.1016/j.virusres.2017.01.020. [DOI] [PubMed] [Google Scholar]
- Lu H., Zhao Q., Xu Z., Jiang S. Automatic quantitation of HIV-1 mediated cell-to-cell fusion with a digital image analysis system (DIAS): application for rapid screening of HIV-1 fusion inhibitors. J. Virol. Methods. 2003;107(2):155–161. doi: 10.1016/s0166-0934(02)00213-6. [DOI] [PubMed] [Google Scholar]
- Lu L., Liu Q., Zhu Y., Chan K.-H., Qin L., Li Y., Wang Q., Chan J.F.-W., Du L., Yu F., Ma C., Ye S., Yuen K.-Y., Zhang R., Jiang S. Structure-based discovery of Middle East respiratory syndrome coronavirus fusion inhibitor. Nat. Commun. 2014;5(3067) doi: 10.1038/ncomms4067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lukic Z., Dharan A., Fricke T., Diaz-Griffero F., Campbell E.M. HIV-1 uncoating is facilitated by dynein and kinesin 1. J. Virol. 2014;88(23):13613–13625. doi: 10.1128/JVI.02219-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luković E., González-Vera J.A., Imperiali B. Recognition-domain focused (RDF) chemosensors: versatile and efficient reporters of protein kinase activity. J. Am. Chem. Soc. 2008;130(38):12821–12827. doi: 10.1021/ja8046188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lütgehetmann M., Mancke L.V., Volz T., Helbig M., Allweiss L., Bornscheuer T., Pollok J.M., Lohse A.W., Petersen J., Urban S., Dandri M. Humanized chimeric uPA mouse model for the study of hepatitis B and D virus interactions and preclinical drug evaluation. Hepatology. 2012;55(3):685–694. doi: 10.1002/hep.24758. [DOI] [PubMed] [Google Scholar]
- Luyet P.P., Falguieres T., Pons V., Pattnaik A.K., Gruenberg J. The ESCRT-I subunit TSG101 controls endosome-to-cytosol release of viral RNA. Traffic (Copenhagen, Denmark) 2008;9(12):2279–2290. doi: 10.1111/j.1600-0854.2008.00820.x. [DOI] [PubMed] [Google Scholar]
- Ma H., Deacon S., Horiuchi K. The challenge of selecting protein kinase assays for lead discovery optimization. Expert Opin. Drug Discovery. 2008;3(6):607–621. doi: 10.1517/17460441.3.6.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Y., Wei Y., Zhang X., Zhang Y., Cai H., Zhu Y., Shilo K., Oglesbee M., Krakowka S., Whelan S.P.J., Li J. mRNA cap methylation influences pathogenesis of vesicular stomatitis virus in vivo. J. Virol. 2014;88(5):2913–2926. doi: 10.1128/JVI.03420-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madhvi A., Hingane S., Srivastav R., Joshi N., Subramani C., Muthumohan R., Khasa R., Varshney S., Kalia M., Vrati S., Surjit M., Ranjith-Kumar C.T. A screen for novel hepatitis C virus RdRp inhibitor identifies a broad-spectrum antiviral compound. Sci. Rep. 2017;7(5816) doi: 10.1038/s41598-017-04449-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malvezzi S., Sturla S.J., Tanasova M. Quantification of pyrophosphate as a universal approach to determine polymerase activity and assay polymerase inhibitors. Anal. Biochem. 2015;478:1–7. doi: 10.1016/j.ab.2015.03.002. [DOI] [PubMed] [Google Scholar]
- Mangel W.F., San Martín C. Structure, function and dynamics in adenovirus maturation. Viruses. 2014;6(11):4536–4570. doi: 10.3390/v6114536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchand C., Neamati N., Pommier Y. In vitro human immunodeficiency virus type 1 integrase assays. Methods Enzymol. 2001;340:624–633. doi: 10.1016/s0076-6879(01)40446-0. [DOI] [PubMed] [Google Scholar]
- Marechal V., Prevost M.C., Petit C., Perret E., Heard J.M., Schwartz O. Human immunodeficiency virus type 1 entry into macrophages mediated by macropinocytosis. J. Virol. 2001;75(22):11166–11177. doi: 10.1128/JVI.75.22.11166-11177.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matreyek K.A., Engelman A. Viral and cellular requirements for the nuclear entry of retroviral preintegration nucleoprotein complexes. Viruses. 2013;5(10):2483–2511. doi: 10.3390/v5102483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayor S., Pagano R.E. Pathways of clathrin-independent endocytosis. Nat. Rev. Mol. Cell Biol. 2007;8(8):603–612. doi: 10.1038/nrm2216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazzon M., Mercer J. Lipid interactions during virus entry and infection. Cell. Microbiol. 2014;16(10):1493–1502. doi: 10.1111/cmi.12340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medintz I.L., Clapp A.R., Brunel F.M., Tiefenbrunn T., Tetsuo Uyeda H., Chang E.L., Deschamps J.R., Dawson P.E., Mattoussi H. Proteolytic activity monitored by fluorescence resonance energy transfer through quantum-dot-peptide conjugates. Nat. Mater. 2006;5(7):581–589. doi: 10.1038/nmat1676. [DOI] [PubMed] [Google Scholar]
- Meier J.L., Grose C. Variable effects of autophagy induction by trehalose on herpesviruses depending on conditions of infection. Yale J. Biol. Med. 2017;90(1):25–33. [PMC free article] [PubMed] [Google Scholar]
- Melikyan G.B. Common principles and intermediates of viral protein-mediated fusion: the HIV-1 paradigm. Retrovirology. 2008;5:111. doi: 10.1186/1742-4690-5-111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menéndez-Arias L., Andino R. Viral polymerases. Virus Res. 2017;234:1–3. doi: 10.1016/j.virusres.2017.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercer J., Helenius A. Virus entry by macropinocytosis. Nat. Cell Biol. 2009;11(5):510–520. doi: 10.1038/ncb0509-510. [DOI] [PubMed] [Google Scholar]
- Mercer J., Helenius A. Apoptotic mimicry: phosphatidylserine-mediated macropinocytosis of vaccinia virus. Ann. N. Y. Acad. Sci. 2010;1209:49–55. doi: 10.1111/j.1749-6632.2010.05772.x. [DOI] [PubMed] [Google Scholar]
- Merkel G., Andrake M.D., Ramcharan J., Skalka A.M. Oligonucleotide-based assays for integrase activity. Methods (San Diego, Calif.) 2009;47(4):243–248. doi: 10.1016/j.ymeth.2008.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mestas S.P., Sholders A.J., Peersen O.B. A fluorescence polarization based screening assay for nucleic acid polymerase elongation activity. Anal. Biochem. 2007;365(2):194–200. doi: 10.1016/j.ab.2007.03.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer N.O., O’Donoghue A.J., Schulze-Gahmen U., Ravalin M., Moss S.M., Winter M.B., Knudsen G.M., Craik C.S. Multiplex substrate profiling by mass spectrometry for kinases as a method for revealing quantitative substrate motifs. Anal. Chem. 2017;89(8):4550–4558. doi: 10.1021/acs.analchem.6b05002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Midde N.M., Patters B.J., Rao P.S.S., Cory T.J., Kumar S. Investigational protease inhibitors as antiretroviral therapies. Expert Opin. Investig. Drugs. 2016;25(10):1189–1200. doi: 10.1080/13543784.2016.1212837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minskaia E., Hertzig T., Gorbalenya A.E., Campanacci V., Cambillau C., Canard B., Ziebuhr J. Discovery of an RNA virus 3′ → 5′ exoribonuclease that is critically involved in coronavirus RNA synthesis. Proc. Natl. Acad. Sci. U. S. A. 2006;103(13):5108–5113. doi: 10.1073/pnas.0508200103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyata Y., Chang L., Bainor A., McQuade T.J., Walczak C.P., Zhang Y., Larsen M.J., Kirchhoff P., Gestwicki J.E. High-throughput screen for Escherichia coli heat shock protein 70 (Hsp70/DnaK): ATPase assay in low volume by exploiting energy transfer. J. Biomol. Screen. 2010;15(10):1211–1219. doi: 10.1177/1087057110380571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mondal A., Dawson A.R., Potts G.K., Freiberger E.C., Baker S.F., Moser L.A., Bernard K.A., Coon J.J., Mehle A. Influenza virus recruits host protein kinase C to control assembly and activity of its replication machinery. elife. 2017;6 doi: 10.7554/eLife.26910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss B. Poxvirus DNA replication. Cold Spring Harb. Perspect. Biol. 2013;5(9) doi: 10.1101/cshperspect.a010199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss B. Poxvirus membrane biogenesis. Virology. 2015;479-480:619–626. doi: 10.1016/j.virol.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss B. Membrane fusion during poxvirus entry. Semin. Cell Dev. Biol. 2016;60:89–96. doi: 10.1016/j.semcdb.2016.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller B., Jones K.S., Merkel G.W., Skalka A.M. Rapid solution assays for retroviral integration reactions and their use in kinetic analyses of wild-type and mutant Rous sarcoma virus integrases. Proc. Natl. Acad. Sci. U. S. A. 1993;90(24):11633–11637. doi: 10.1073/pnas.90.24.11633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munjal A., Khandia R., Dhama K., Sachan S., Karthik K., Tiwari R., Malik Y.S., Kumar D., Singh R.K., Iqbal H.M.N., Joshi S.K. Advances in developing therapies to combat Zika virus: current knowledge and future perspectives. Front. Microbiol. 2017;8(1469) doi: 10.3389/fmicb.2017.01469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray M.G., Hung W., Sadowski I., Das Mahapatra B. Inactivation of a yeast transactivator by the fused HIV-1 proteinase: a simple assay for inhibitors of the viral enzyme activity. Gene. 1993;134(1):123–128. doi: 10.1016/0378-1119(93)90185-6. [DOI] [PubMed] [Google Scholar]
- Nedjadi T., El-Kafrawy S., Sohrab S.S., Despres P., Damanhouri G., Azhar E. Tackling dengue fever: current status and challenges. Virol. J. 2015;12:212. doi: 10.1186/s12985-015-0444-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen M., Haenni A.L. Expression strategies of ambisense viruses. Virus Res. 2003;93(2):141–150. doi: 10.1016/s0168-1702(03)00094-7. [DOI] [PubMed] [Google Scholar]
- Nichols B.D., De Martini W., Cottrell J. Poxviruses utilize multiple strategies to inhibit apoptosis. Viruses. 2017;9(8) doi: 10.3390/v9080215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Öberg B. Rational design of polymerase inhibitors as antiviral drugs. Antivir. Res. 2006;71(2):90–95. doi: 10.1016/j.antiviral.2006.05.012. [DOI] [PubMed] [Google Scholar]
- Ojala P.M., Sodeik B., Ebersold M.W., Kutay U., Helenius A. Herpes simplex virus type 1 Entry into host cells: reconstitution of capsid binding and uncoating at the nuclear pore complex in vitro. Mol. Cell. Biol. 2000;20(13):4922–4931. doi: 10.1128/mcb.20.13.4922-4931.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okon A., Matos de Souza M.R., Shah R., Amorim R., da Costa L.J., Wagner C.R. Anchimerically activatable antiviral protides. ACS Med. Chem. Lett. 2017;8(9):958–962. doi: 10.1021/acsmedchemlett.7b00277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okumura A., Harty R.N. Rabies virus assembly and budding. Adv. Virus Res. 2011;79:23–32. doi: 10.1016/B978-0-12-387040-7.00002-0. [DOI] [PubMed] [Google Scholar]
- Olmos Y., Carlton J.G. The ESCRT machinery: new roles at new holes. Curr. Opin. Cell Biol. 2016;38:1–11. doi: 10.1016/j.ceb.2015.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen J.V., Blagoev B., Gnad F., Macek B., Kumar C., Mortensen P., Mann M. Global, in vivo, and site-specific phosphorylation dynamics in signaling networks. Cell. 2006;127(3):635–648. doi: 10.1016/j.cell.2006.09.026. [DOI] [PubMed] [Google Scholar]
- Oshima M., Goto N., Susanto J.P., Motomizu S. Determination of phosphate as aggregates of ion associates by light-scattering detection and application to flow injection. Analyst. 1996;121(8):1085–1088. [Google Scholar]
- Özeş A.R., Feoktistova K., Avanzino B.C., Baldwin E.P., Fraser C.S. Real-time fluorescence assays to monitor duplex unwinding and ATPase activities of helicases. Nat. Protoc. 2014;9(7):1645–1661. doi: 10.1038/nprot.2014.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pante N., Kann M. Nuclear pore complex is able to transport macromolecules with diameters of about 39 nm. Mol. Biol. Cell. 2002;13(2):425–434. doi: 10.1091/mbc.01-06-0308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park A., Yun T., Vigant F., Pernet O., Won S.T., Dawes B.E., Bartkowski W., Freiberg A.N., Lee B. Nipah Virus C protein recruits Tsg101 to promote the efficient release of virus in an ESCRT-dependent pathway. PLoS Pathog. 2016;12(5):e1005659. doi: 10.1371/journal.ppat.1005659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J.-Y., Yuk H.J., Ryu H.W., Lim S.H., Kim K.S., Park K.H., Ryu Y.B., Lee W.S. Evaluation of polyphenols from Broussonetia papyrifera as coronavirus protease inhibitors. J. Enzyme Inhibition Med. Chem. 2017;32(1):504–512. doi: 10.1080/14756366.2016.1265519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parton R.G., Simons K. The multiple faces of caveolae. Nat. Rev. Mol. Cell Biol. 2007;8(3):185–194. doi: 10.1038/nrm2122. [DOI] [PubMed] [Google Scholar]
- Pasdeloup D., Blondel D., Isidro A.L., Rixon F.J. Herpesvirus capsid association with the nuclear pore complex and viral dna release INVOLVE THE Nucleoporin CAN/Nup214 and the Capsid Protein pUL25. J. Virol. 2009;83(13):6610–6623. doi: 10.1128/JVI.02655-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pawlotsky J.M., Chevaliez S., McHutchison J.G. The Hepatitis C Virus life cycle as a target for new antiviral therapies. Gastroenterology. 2007;132(5):1979–1998. doi: 10.1053/j.gastro.2007.03.116. [DOI] [PubMed] [Google Scholar]
- Pelliccia S., Wu Y.-H., Coluccia A., La Regina G., Tseng C.-K., Famiglini V., Masci D., Hiscott J., Lee J.-C., Silvestri R. Inhibition of dengue virus replication by novel inhibitors of RNA-dependent RNA polymerase and protease activities. J. Enzyme Inhibition Med. Chem. 2017;32(1):1091–1101. doi: 10.1080/14756366.2017.1355791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen J., Dandri M., Mier W., Lutgehetmann M., Volz T., von Weizsacker F., Haberkorn U., Fischer L., Pollok J.M., Erbes B., Seitz S., Urban S. Prevention of hepatitis B virus infection in vivo by entry inhibitors derived from the large envelope protein. Nat. Biotechnol. 2008;26(3):335–341. doi: 10.1038/nbt1389. [DOI] [PubMed] [Google Scholar]
- Pflug A., Lukarska M., Resa-Infante P., Reich S., Cusack S. Structural insights into RNA synthesis by the influenza virus transcription-replication machine. Virus Res. 2017;234:103–117. doi: 10.1016/j.virusres.2017.01.013. [DOI] [PubMed] [Google Scholar]
- Pietilä M.K., Hellström K., Ahola T. Alphavirus polymerase and RNA replication. Virus Res. 2017;234:44–57. doi: 10.1016/j.virusres.2017.01.007. [DOI] [PubMed] [Google Scholar]
- Pillaiyar T., Manickam M., Namasivayam V., Hayashi Y., Jung S.-H. An overview of severe acute respiratory syndrome–coronavirus (SARS-CoV) 3CL protease inhibitors: peptidomimetics and small molecule chemotherapy. J. Med. Chem. 2016;59(14):6595–6628. doi: 10.1021/acs.jmedchem.5b01461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitman M.R., Pham D.H., Pitson S.M. Isoform-selective assays for sphingosine kinase activity. In: Pébay A., Turksen K., editors. Sphingosine-1-Phosphate: Methods and Protocols. Humana Press; Totowa, NJ: 2012. pp. 21–31. [DOI] [PubMed] [Google Scholar]
- Ploss A., Evans M.J., Gaysinskaya V.A., Panis M., You H., de Jong Y.P., Rice C.M. Human occludin is a hepatitis C virus entry factor required for infection of mouse cells. Nature. 2009;457(7231):882–886. doi: 10.1038/nature07684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Podany A.T., Scarsi K.K., Fletcher C.V. Comparative clinical pharmacokinetics and pharmacodynamics of HIV-1 integrase strand transfer inhibitors. Clin. Pharmacokinet. 2017;56(1):25–40. doi: 10.1007/s40262-016-0424-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pohl M.O., Lanz C., Stertz S. Late stages of the influenza A virus replication cycle-a tight interplay between virus and host. J. General Virol. 2016;97(9):2058–2072. doi: 10.1099/jgv.0.000562. [DOI] [PubMed] [Google Scholar]
- Pohl, M.O., von Recum-Knepper, J., Rodriguez-Frandsen, A., Lanz, C., Yanguez, E., Soonthornvacharin, S., Wolff, T., Chanda, S.K., Stertz, S., 2017. Identification of Polo-like kinases as potential novel drug targets for influenza A virus. Sci. Rep. 7(1), 8629. [DOI] [PMC free article] [PubMed]
- Prasad B.V.V., Schmid M.F. Principles of virus structural organization. Adv. Exp. Med. Biol. 2012;726:17–47. doi: 10.1007/978-1-4614-0980-9_3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purdy J.G., Flanagan J.M., Ropson I.J., Rennoll-Bankert K.E., Craven R.C. Critical role of conserved hydrophobic residues within the major homology region in mature retroviral capsid assembly. J. Virol. 2008;82(12):5951–5961. doi: 10.1128/JVI.00214-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purdy J.G., Flanagan J.M., Ropson I.J., Craven R.C. Retroviral capsid assembly: a role for the CA dimer in initiation. J. Mol. Biol. 2009;389(2):438–451. doi: 10.1016/j.jmb.2009.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabe B., Vlachou A., Pante N., Helenius A., Kann M. Nuclear import of hepatitis B virus capsids and release of the viral genome. Proc. Natl. Acad. Sci. U. S. A. 2003;100(17):9849–9854. doi: 10.1073/pnas.1730940100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raguram A., Sasisekharan V., Sasisekharan R. A chiral pentagonal polyhedral framework for characterizing virus capsid structures. Trends Microbiol. 2017;25(6):438–446. doi: 10.1016/j.tim.2016.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahmeh A.A., Li J., Kranzusch P.J., Whelan S.P. Ribose 2'-O methylation of the vesicular stomatitis virus mRNA cap precedes and facilitates subsequent guanine-N-7 methylation by the large polymerase protein. J. Virol. 2009;83(21):11043–11050. doi: 10.1128/JVI.01426-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randow F., Lehner P.J. Viral avoidance and exploitation of the ubiquitin system. Nat. Cell Biol. 2009;11(5):527–534. doi: 10.1038/ncb0509-527. [DOI] [PubMed] [Google Scholar]
- Ranjith-Kumar C.T., Wen Y., Baxter N., Bhardwaj K., Cheng Kao C. A cell-based assay for RNA synthesis by the HCV polymerase reveals new insights on mechanism of polymerase inhibitors and modulation by NS5A. PLoS One. 2011;6(7):e22575. doi: 10.1371/journal.pone.0022575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rankovic S., Varadarajan J., Ramalho R., Aiken C., Rousso I. Reverse transcription mechanically initiates HIV-1 Capsid disassembly. J. Virol. 2017;91(12) doi: 10.1128/JVI.00289-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasmussen I., Vilhardt F. Macropinocytosis is the entry mechanism of amphotropic murine leukemia virus. J. Virol. 2015;89(3):1851–1866. doi: 10.1128/JVI.02343-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray D., Shah A., Tilgner M., Guo Y., Zhao Y., Dong H., Deas T.S., Zhou Y., Li H., Shi P.-Y. West Nile Virus 5′-Cap structure is formed by sequential guanine N-7 and Ribose 2′-O methylations by nonstructural protein 5. J. Virol. 2006;80(17):8362–8370. doi: 10.1128/JVI.00814-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Razonable R.R. Antiviral drugs for viruses other than human immunodeficiency virus. Mayo Clin. Proc. 2011;86(10):1009–1026. doi: 10.4065/mcp.2011.0309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rey F.A., Stiasny K., Heinz F.X. Flavivirus structural heterogeneity: implications for cell entry. Current Opinion Virol. 2017;24:132–139. doi: 10.1016/j.coviro.2017.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynolds K.A., Cameron C.E., Raney K.D. Melting of duplex DNA in the absence of ATP by NS3 helicase domain through specific interaction with a single-strand/double-strand junction. Biochemistry. 2015;54(27):4248–4258. doi: 10.1021/acs.biochem.5b00214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards A.L., Jackson W.T. Intracellular vesicle acidification promotes maturation of infectious poliovirus particles. PLoS Pathog. 2012;8(11):e1003046. doi: 10.1371/journal.ppat.1003046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roe T., Reynolds T.C., Yu G., Brown P.O. Integration of murine leukemia virus DNA depends on mitosis. EMBO J. 1993;12(5):2099–2108. doi: 10.1002/j.1460-2075.1993.tb05858.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossignol E.D., Yang J.E., Bullitt E. the role of electron microscopy in studying the continuum of changes in membranous structures during poliovirus infection. Viruses. 2015;7(10):5305–5318. doi: 10.3390/v7102874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rumlová M., Ruml T., Pohl J., Pichová I. Specific in vitro cleavage of Mason–Pfizer monkey virus capsid protein: evidence for a potential role of retroviral protease in early stages of infection. Virology. 2003;310(2):310–318. doi: 10.1016/s0042-6822(03)00128-4. [DOI] [PubMed] [Google Scholar]
- Rumlova M., Krizova I., Keprova A., Hadravova R., Dolezal M., Strohalmova K., Pichova I., Hajek M., Ruml T. HIV-1 protease-induced apoptosis. Retrovirology. 2014;11:37. doi: 10.1186/1742-4690-11-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rumlova-Klikova M., Pichova I., Hunter E., Ruml T. Conditions resulting in formation of properly assembled retroviral capsids within inclusion bodies of Escherichia coli. Collect. Czechoslov. Chem. Commun. 1999;64(8):1348–1356. [Google Scholar]
- Rumlova-Klikova M., Hunter E., Nermut M.V., Pichova I., Ruml T. Analysis of Mason-Pfizer monkey virus gag domains required for capsid assembly in bacteria: role of the N-terminal proline residue of CA in directing particle shape. J. Virol. 2000;74(18):8452–8459. doi: 10.1128/jvi.74.18.8452-8459.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saeed M.F., Kolokoltsov A.A., Albrecht T., Davey R.A. Cellular entry of ebola virus involves uptake by a macropinocytosis-like mechanism and subsequent trafficking through early and late endosomes. PLoS Pathog. 2010;6(9):e1001110. doi: 10.1371/journal.ppat.1001110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanghera J., Li R., Yan J. Comparison of the luminescent ADP-Glo assay to a standard radiometric assay for measurement of protein kinase activity. Assay Drug Dev Technol. 2009;7(6):615–622. doi: 10.1089/adt.2009.0237. [DOI] [PubMed] [Google Scholar]
- Sato Y., Koshizuka T., Ishibashi K., Hashimoto K., Ishioka K., Ikuta K., Yokota S.I., Fujii N., Suzutani T. Involvement of herpes simplex virus type 1 UL13 protein kinase in induction of SOCS genes, the negative regulators of cytokine signaling. Microbiol. Immunol. 2017;61(5):159–167. doi: 10.1111/1348-0421.12483. [DOI] [PubMed] [Google Scholar]
- Schäferling M., Wolfbeis O.S. Europium tetracycline as a luminescent probe for nucleoside phosphates and its application to the determination of kinase activity. Chem. Eur. J. 2007;13(15):4342–4349. doi: 10.1002/chem.200601509. [DOI] [PubMed] [Google Scholar]
- Schmiedel D., Tai J., Levi-Schaffer F., Dovrat S., Mandelboim O. Human Herpesvirus 6B downregulates expression of activating ligands during lytic infection to escape elimination by natural killer cells. J. Virol. 2016;90(21):9608–9617. doi: 10.1128/JVI.01164-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz J.J., Quake S.R. Single molecule measurement of the “speed limit” of DNA polymerase. Proc. Natl. Acad. Sci. U. S. A. 2009;106(48):20294–20299. doi: 10.1073/pnas.0907404106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scourfield E.J., Martin-Serrano J. Growing functions of the ESCRT machinery in cell biology and viral replication. Biochem. Soc. Trans. 2017;45(3):613–634. doi: 10.1042/BST20160479. [DOI] [PubMed] [Google Scholar]
- Sells M.A., Chen M.L., Acs G. Production of hepatitis B virus particles in Hep G2 cells transfected with cloned hepatitis B virus DNA. Proc. Natl. Acad. Sci. U. S. A. 1987;84(4):1005–1009. doi: 10.1073/pnas.84.4.1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seybert A., Hegyi A., Siddell S.G., Ziebuhr J. The human coronavirus 229E superfamily 1 helicase has RNA and DNA duplex-unwinding activities with 5′-to-3′ polarity. RNA (New York, N.Y.) 2000;6(7):1056–1068. doi: 10.1017/s1355838200000728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sfakianos J.N., LaCasse R.A., Hunter E. The M-PMV cytoplasmic targeting-retention signal directs nascent Gag polypeptides to a pericentriolar region of the cell. Traffic (Copenhagen, Denmark) 2003;4(10):660–670. doi: 10.1034/j.1600-0854.2003.00125.x. [DOI] [PubMed] [Google Scholar]
- Shadrick W.R., Ndjomou J., Kolli R., Mukherjee S., Hanson A.M., Frick D.N. Discovering new medicines targeting helicases: challenges and recent progress. J. Biomol. Screen. 2013;18(7):761–781. doi: 10.1177/1087057113482586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah V.B., Aiken C. In vitro uncoating of HIV-1 cores. J. Vis. Exp. 2011;57 doi: 10.3791/3384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah P.N.M., Stanifer M.L., Hohn K., Engel U., Haselmann U., Bartenschlager R., Krausslich H.G., Krijnse-Locker J., Boulant S. Genome packaging of reovirus is mediated by the scaffolding property of the microtubule network. Cell. Microbiol. 2017;19(12):e12765. doi: 10.1111/cmi.12765. [DOI] [PubMed] [Google Scholar]
- Sherer N.M., Jin J., Mothes W. Directional spread of surface-associated retroviruses regulated by differential virus-cell interactions. J. Virol. 2010;84(7):3248–3258. doi: 10.1128/JVI.02155-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song Y.H., Kim D.W., Curtis-Long M.J., Yuk H.J., Wang Y., Zhuang N., Lee K.H., Jeon K.S., Park K.H. Papain-like protease (PLpro) inhibitory effects of cinnamic amides from < i > tribulus terrestris </i > fruits. Biol. Pharm. Bull. 2014;37(6):1021–1028. doi: 10.1248/bpb.b14-00026. [DOI] [PubMed] [Google Scholar]
- Sportsman J.R., Gaudet E.A., Boge A. Immobilized metal ion affinity-based fluorescence polarization (IMAP): advances in kinase screening. Assay Drug Dev Technol. 2004;2(2):205–214. doi: 10.1089/154065804323056549. [DOI] [PubMed] [Google Scholar]
- St. John S.E., Tomar S., Stauffer S.R., Mesecar A.D. Targeting zoonotic viruses: structure-based inhibition of the 3C-like protease from bat coronavirus HKU4 – the likely reservoir host to the human coronavirus that causes Middle East Respiratory Syndrome (MERS) Bioorg. Med. Chem. 2015;23(17):6036–6048. doi: 10.1016/j.bmc.2015.06.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stahl H., Dröge P., Knippers R. DNA helicase activity of SV40 large tumor antigen. EMBO J. 1986;5(8):1939–1944. doi: 10.1002/j.1460-2075.1986.tb04447.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stauffer S., Nebioglu F., Helenius A. In vitro disassembly of influenza A virus capsids by gradient centrifugation. J. Vis. Exp. 2016;109:e53909. doi: 10.3791/53909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sticht J., Humbert M., Findlow S., Bodem J., Muller B., Dietrich U., Werner J., Krausslich H.G. A peptide inhibitor of HIV-1 assembly in vitro. Nat. Struct. Mol. Biol. 2005;12(8):671–677. doi: 10.1038/nsmb964. [DOI] [PubMed] [Google Scholar]
- Strandin T., Hepojoki J., Vaheri A. Cytoplasmic tails of bunyavirus Gn glycoproteins-Could they act as matrix protein surrogates? Virology. 2013;437(2):73–80. doi: 10.1016/j.virol.2013.01.001. [DOI] [PubMed] [Google Scholar]
- Stray S.J., Johnson J.M., Kopek B.G., Zlotnick A. An in vitro fluorescence screen to identify antivirals that disrupt hepatitis B virus capsid assembly. Nat. Biotechnol. 2006;24(3):358–362. doi: 10.1038/nbt1187. [DOI] [PubMed] [Google Scholar]
- Stremlau M., Perron M., Lee M., Li Y., Song B., Javanbakht H., Diaz-Griffero F., Anderson D.J., Sundquist W.I., Sodroski J. Specific recognition and accelerated uncoating of retroviral capsids by the TRIM5alpha restriction factor. Proc. Natl. Acad. Sci. U. S. A. 2006;103(14):5514–5519. doi: 10.1073/pnas.0509996103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugrue R.J., Brown C., Brown G., Aitken J., McL. Rixon, H.W. Furin cleavage of the respiratory syncytial virus fusion protein is not a requirement for its transport to the surface of virus-infected cells. J. Gen. Virol. 2001;82(6):1375–1386. doi: 10.1099/0022-1317-82-6-1375. [DOI] [PubMed] [Google Scholar]
- Sumaoka J., Akiba H., Komiyama M. Selective sensing of tyrosine phosphorylation in peptides using terbium(III) complexes. Int. J. Analytical Chem. 2016;2016:3216523. doi: 10.1155/2016/3216523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y., Wang Z., Tao J., Wang Y., Wu A., Yang Z., Wang K., Shi L., Chen Y., Guo D. Yeast-based assays for the high-throughput screening of inhibitors of coronavirus RNA cap guanine-N7-methyltransferase. Antivir. Res. 2014;104:156–164. doi: 10.1016/j.antiviral.2014.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sureau C., Romet-Lemonne J.-L., Mullins J.I., Essex M. Production of hepatitis B virus by a differentiated human hepatoma cell line after transfection with cloned circular HBV DNA. Cell. 1986;47(1):37–47. doi: 10.1016/0092-8674(86)90364-8. [DOI] [PubMed] [Google Scholar]
- Takeuchi H., Saito H., Noda T., Miyamoto T., Yoshinaga T., Terahara K., Ishii H., Tsunetsugu-Yokota Y., Yamaoka S. Phosphorylation of the HIV-1 capsid by MELK triggers uncoating to promote viral cDNA synthesis. PLoS Pathog. 2017;13(7):e1006441. doi: 10.1371/journal.ppat.1006441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tandon R., Mocarski E.S. Viral and host control of cytomegalovirus maturation. Trends Microbiol. 2012;20(8):392–401. doi: 10.1016/j.tim.2012.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tandon R., Mocarski E.S., Conway J.F. The A, B, Cs of herpesvirus capsids. Viruses. 2015;7(3):899–914. doi: 10.3390/v7030899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tani H., Fujita O., Furuta A., Matsuda Y., Miyata R., Akimitsu N., Tanaka J., Tsuneda S., Sekiguchi Y., Noda N. Real-time monitoring of RNA helicase activity using fluorescence resonance energy transfer in vitro. Biochem. Biophys. Res. Commun. 2010;393(1):131–136. doi: 10.1016/j.bbrc.2010.01.100. [DOI] [PubMed] [Google Scholar]
- Tao Y.J., Ye Q. RNA virus replication complexes. PLoS Pathog. 2010;6(7):e1000943. doi: 10.1371/journal.ppat.1000943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor G.M., Hanson P.I., Kielian M. Ubiquitin depletion and dominant-negative VPS4 inhibit rhabdovirus budding without affecting alphavirus budding. J. Virol. 2007;81(24):13631–13639. doi: 10.1128/JVI.01688-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teruya K., Hattori Y., Shimamoto Y., Kobayashi K., Sanjoh A., Nakagawa A., Yamashita E., Akaji K. Structural basis for the development of SARS 3CL protease inhibitors from a peptide mimic to an aza-decaline scaffold. Pept. Sci. 2016;106(4):391–403. doi: 10.1002/bip.22773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thierry E., Deprez E., Delelis O. Different pathways leading to integrase inhibitors resistance. Front. Microbiol. 2016;7:2165. doi: 10.3389/fmicb.2016.02165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tino J.A., Clark J.M., Field A.K., Jacobs G.A., Lis K.A., Michalik T.L., McGeever-Rubin B., Slusarchyk W.A., Spergel S.H. Synthesis and antiviral activity of novel isonucleoside analogs. J. Med. Chem. 1993;36(9):1221–1229. doi: 10.1021/jm00061a013. [DOI] [PubMed] [Google Scholar]
- Tisdale M., Kemp S.D., Parry N.R., Larder B.A. Rapid in vitro selection of human immunodeficiency virus type 1 resistant to 3'-thiacytidine inhibitors due to a mutation in the YMDD region of reverse transcriptase. Proc. Natl. Acad. Sci. U. S. A. 1993;90(12):5653–5656. doi: 10.1073/pnas.90.12.5653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomar S., Johnston M.L., St. John S.E., Osswald H.L., Nyalapatla P.R., Paul L.N., Ghosh A.K., Denison M.R., Mesecar A.D. Ligand-induced dimerization of middle east respiratory syndrome (MERS) coronavirus nsp5 Protease (3CL(pro)): implications for nsp5 regulation and the development of antivirals. J. Biol. Chem. 2015;290(32):19403–19422. doi: 10.1074/jbc.M115.651463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong L.-I., Chen Z.-Z., Jiang Z.-Y., Sun M.-M., Li L., Liu J., Tang B. Fluorescent sensing of pyrophosphate anion in synovial fluid based on DNA-attached magnetic nanoparticles. Biosens. Bioelectron. 2015;72:51–55. doi: 10.1016/j.bios.2015.04.087. [DOI] [PubMed] [Google Scholar]
- Trask S.D., McDonald S.M., Patton J.T. Structural insights into the coupling of virion assembly and rotavirus replication. Nat. Rev. Microbiol. 2012;10(3):165–177. doi: 10.1038/nrmicro2673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tscherne D.M., Manicassamy B., Garcia-Sastre A. An enzymatic virus-like particle assay for sensitive detection of virus entry. J. Virol. Methods. 2010;163(2):336–343. doi: 10.1016/j.jviromet.2009.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Udukala D.N., Wang H., Wendel S.O., Malalasekera A.P., Samarakoon T.N., Yapa A.S., Abayaweera G., Basel M.T., Maynez P., Ortega R., Toledo Y., Bossmann L., Robinson C., Janik K.E., Koper O.B., Li P., Motamedi M., Higgins D.A., Gadbury G., Zhu G., Troyer D.L., Bossmann S.H. Early breast cancer screening using iron/iron oxide-based nanoplatforms with sub-femtomolar limits of detection. Beilstein J. Nanotechnol. 2016;7:364–373. doi: 10.3762/bjnano.7.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ujike M., Taguchi F. Incorporation of spike and membrane glycoproteins into coronavirus virions. Viruses. 2015;7(4):1700–1725. doi: 10.3390/v7041700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulbrich P., Haubova S., Nermut M.V., Hunter E., Rumlova M., Ruml T. Distinct roles for nucleic acid in in vitro assembly of purified Mason-Pfizer monkey virus CANC proteins. J. Virol. 2006;80(14):7089–7099. doi: 10.1128/JVI.02694-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Utama A., Shimizu H., Morikawa S., Hasebe F., Morita K., Igarashi A., Hatsu M., Takamizawa K., Miyamura T. Identification and characterization of the RNA helicase activity of Japanese encephalitis virus NS3 protein. FEBS Lett. 2000;465(1):74–78. doi: 10.1016/s0014-5793(99)01705-6. [DOI] [PubMed] [Google Scholar]
- Veesler D., Johnson J.E. Virus maturation. Annu. Rev. Biophys. 2012;41:473–496. doi: 10.1146/annurev-biophys-042910-155407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlach J., Lipov J., Rumlova M., Veverka V., Lang J., Srb P., Knejzlik Z., Pichova I., Hunter E., Hrabal R., Ruml T. D-retrovirus morphogenetic switch driven by the targeting signal accessibility to Tctex-1 of dynein. Proc. Natl. Acad. Sci. U. S. A. 2008;105(30):10565–10570. doi: 10.1073/pnas.0801765105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogt P.K. Retroviral oncogenes: a historical primer. Nat. Rev. Cancer. 2012;12(9):639–648. doi: 10.1038/nrc3320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volz T., Allweiss L., ḾBarek M.B., Warlich M., Lohse A.W., Pollok J.M., Alexandrov A., Urban S., Petersen J., Lütgehetmann M., Dandri M. The entry inhibitor Myrcludex-B efficiently blocks intrahepatic virus spreading in humanized mice previously infected with hepatitis B virus. J. Hepatol. 2013;58(5):861–867. doi: 10.1016/j.jhep.2012.12.008. [DOI] [PubMed] [Google Scholar]
- Vyse S., Desmond H., Huang P.H. Advances in mass spectrometry based strategies to study receptor tyrosine kinases. IUCrJ. 2017;4(Pt 2):119–130. doi: 10.1107/S2052252516020546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Klock H., Yin H., Wolff K., Bieza K., Niswonger K., Matzen J., Gunderson D., Hale J., Lesley S., Kuhen K., Caldwell J., Brinker A. Homogeneous high-throughput screening assays for HIV-1 integrase 3β-processing and strand transfer activities. J. Biomol. Screen. 2005;10(5):456–462. doi: 10.1177/1087057105275212. [DOI] [PubMed] [Google Scholar]
- Wang H., Udukala D.N., Samarakoon T.N., Basel M.T., Kalita M., Abayaweera G., Manawadu H., Malalasekera A., Robinson C., Villanueva D., Maynez P., Bossmann L., Riedy E., Barriga J., Wang N., Li P., Higgins D.A., Zhu G., Troyer D.L., Bossmann S.H. Nanoplatforms for highly sensitive fluorescence detection of cancer-related proteases. Photochem. Photobiol. Sci. 2014;13(2):231–240. doi: 10.1039/c3pp50260k. [DOI] [PubMed] [Google Scholar]
- Wang Y.-W., Liu S.-B., Yang Y.-L., Wang P.-Z., Zhang A.-J., Peng Y. A Terbium(III)-complex-based on–off fluorescent chemosensor for phosphate anions in aqueous solution and its application in molecular logic gates. ACS Appl. Mater. Interfaces. 2015;7(7):4415–4422. doi: 10.1021/am5089346. [DOI] [PubMed] [Google Scholar]
- Wang Y., Sun Y., Wu A., Xu S., Pan R., Zeng C., Jin X., Ge X., Shi Z., Ahola T., Chen Y., Guo D. Coronavirus nsp10/nsp16 methyltransferase can be targeted by nsp10-derived peptide in vitro and in vivo to reduce replication and pathogenesis. J. Virol. 2015;89(16):8416–8427. doi: 10.1128/JVI.00948-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z., Zhao F., Gao Q., Liu Z., Zhang Y., Li X., Li Y., Ma W., Deng T., Zhang Z., Cen S. Establishment of a high-throughput assay to monitor influenza A virus RNA transcription and replication. PLoS One. 2015;10(7):e0133558. doi: 10.1371/journal.pone.0133558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F., Chen C., Tan W., Yang K., Yang H. Structure of main protease from human coronavirus NL63: insights for wide spectrum anti-coronavirus drug design. Sci. Rep. 2016;6(22677) doi: 10.1038/srep22677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X., Tang S., Song X., Rong L. Mathematical analysis of an HIV latent infection model including both virus-to-cell infection and cell-to-cell transmission. J. Biol. Dyn. 2017;11(sup2):455–483. doi: 10.1080/17513758.2016.1242784. [DOI] [PubMed] [Google Scholar]
- Weiss S. Measuring conformational dynamics of biomolecules by single molecule fluorescence spectroscopy. Nat. Struct. Mol. Biol. 2000;7(9):724–729. doi: 10.1038/78941. [DOI] [PubMed] [Google Scholar]
- White A.K., Heyries K.A., Doolin C., VanInsberghe M., Hansen C.L. High-throughput microfluidic single-cell digital polymerase chain reaction. Anal. Chem. 2013;85(15):7182–7190. doi: 10.1021/ac400896j. [DOI] [PubMed] [Google Scholar]
- Wu J., Bera A.K., Kuhn R.J., Smith J.L. Structure of the Flavivirus helicase: implications for catalytic activity, protein interactions, and proteolytic processing. J. Virol. 2005;79(16):10268–10277. doi: 10.1128/JVI.79.16.10268-10277.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing Y., Chen J., Tu J., Zhang B., Chen X., Shi H., Baker S.C., Feng L., Chen Z. The papain-like protease of porcine epidemic diarrhea virus negatively regulates type I interferon pathway by acting as a viral deubiquitinase. J. Gen. Virol. 2013;94(7):1554–1567. doi: 10.1099/vir.0.051169-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue L., Wang W.-H., Iliuk A., Hu L., Galan J.A., Yu S., Hans M., Geahlen R.L., Tao W.A. Sensitive kinase assay linked with phosphoproteomics for identifying direct kinase substrates. Proc. Natl. Acad. Sci. U. S. A. 2012;109(15):5615–5620. doi: 10.1073/pnas.1119418109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan H., Zhong G., Xu G., He W., Jing Z., Gao Z., Huang Y., Qi Y., Peng B., Wang H., Fu L., Song M., Chen P., Gao W., Ren B., Sun Y., Cai T., Feng X., Sui J., Li W. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. elife. 2012;1:e00049. doi: 10.7554/eLife.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L., Mohr I., Fouts E., Lim D.A., Nohaile M., Botchan M. The E1 protein of bovine papilloma virus 1 is an ATP-dependent DNA helicase. Proc. Natl. Acad. Sci. U. S. A. 1993;90(11):5086–5090. doi: 10.1073/pnas.90.11.5086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H., Xie W., Xue X., Yang K., Ma J., Liang W., Zhao Q., Zhou Z., Pei D., Ziebuhr J., Hilgenfeld R., Yuen K.Y., Wong L., Gao G., Chen S., Chen Z., Ma D., Bartlam M., Rao Z. Design of wide-spectrum inhibitors targeting coronavirus main proteases. PLoS Biol. 2005;3(10):e324. doi: 10.1371/journal.pbio.0030324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yilmaz N.K., Swanstrom R., Schiffer C.A. Improving viral protease inhibitors to counter drug resistance. Trends Microbiol. 2016;24(7):547–557. doi: 10.1016/j.tim.2016.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yonezawa A., Cavrois M., Greene W.C. Studies of ebola virus glycoprotein-mediated entry and fusion by using pseudotyped human immunodeficiency virus type 1 virions: involvement of cytoskeletal proteins and enhancement by tumor necrosis factor alpha. J. Virol. 2005;79(2):918–926. doi: 10.1128/JVI.79.2.918-926.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youngren J.F., Goldfine I.D., Pratley R.E. Decreased muscle insulin receptor kinase correlates with insulin resistance in normoglycemic Pima Indians. Am. J. Physiol. Endocrinol. Metab. 1997;273(2):E276. doi: 10.1152/ajpendo.1997.273.2.E276. [DOI] [PubMed] [Google Scholar]
- Zegzouti H., Zdanovskaia M., Hsiao K., Goueli S.A. ADP-Glo: A bioluminescent and homogeneous ADP monitoring assay for kinases. Assay Drug Dev Technol. 2009;7(6):560–572. doi: 10.1089/adt.2009.0222. [DOI] [PubMed] [Google Scholar]
- Zeng T., Zhang T., Wei W., Li Z., Wu D., Wang L., Guo J., He X., Ma N. Compact, programmable, and stable biofunctionalized upconversion nanoparticles prepared through peptide-mediated phase transfer for high-sensitive protease sensing and in vivo apoptosis imaging. ACS Appl. Mater. Interfaces. 2015;7(22):11849–11856. doi: 10.1021/acsami.5b01446. [DOI] [PubMed] [Google Scholar]
- Zeng C., Wu A., Wang Y., Xu S., Tang Y., Jin X., Wang S., Qin L., Sun Y., Fan C., Snijder E.J., Neuman B.W., Chen Y., Ahola T., Guo D. Identification and characterization of a ribose 2′-O-methyltransferase encoded by the ronivirus branch of nidovirales. J. Virol. 2016;90(15):6675–6685. doi: 10.1128/JVI.00658-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J., Allen M.D. FRET-based biosensors for protein kinases: illuminating the kinome. Mol. BioSyst. 2007;3(11):759–765. doi: 10.1039/b706628g. [DOI] [PubMed] [Google Scholar]
- Zhang H., Zha X., Tan Y., Hornbeck P.V., Mastrangelo A.J., Alessi D.R., Polakiewicz R.D., Comb M.J. Phosphoprotein analysis using antibodies broadly reactive against phosphorylated motifs. J. Biol. Chem. 2002;277(42):39379–39387. doi: 10.1074/jbc.M206399200. [DOI] [PubMed] [Google Scholar]
- Zhang Z., Yang E., Hu C., Cheng H., Chen C.Y., Huang D., Wang R., Zhao Y., Rong L., Vignuzzi M., Shen H., Shen L., Chen Z.W. Cell-based high-throughput screening assay identifies 2′,2′-difluoro-2′-deoxycytidine gemcitabine as a potential antipoliovirus agent. ACS Infect. Dis. 2017;3(1):45–53. doi: 10.1021/acsinfecdis.6b00116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Q., Li S., Xue F., Zou Y., Chen C., Bartlam M., Rao Z. Structure of the main protease from a global infectious human coronavirus, HCoV-HKU1. J. Virol. 2008;82(17):8647–8655. doi: 10.1128/JVI.00298-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y., Soh T.S., Lim S.P., Chung K.Y., Swaminathan K., Vasudevan S.G., Shi P.-Y., Lescar J., Luo D. Molecular basis for specific viral RNA recognition and 2′-O-ribose methylation by the dengue virus nonstructural protein 5 (NS5) Proc. Natl. Acad. Sci. 2015;112(48):14834–14839. doi: 10.1073/pnas.1514978112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao B., Yi G., Du F., Chuang Y.-C., Vaughan R.C., Sankaran B., Kao C.C., Li P. Structure and function of the Zika virus full-length NS5 protein. Nat. Commun. 2017;8:14762. doi: 10.1038/ncomms14762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong J., Gastaminza P., Cheng G., Kapadia S., Kato T., Burton D.R., Wieland S.F., Uprichard S.L., Wakita T., Chisari F.V. Robust hepatitis C virus infection in vitro. Proc. Natl. Acad. Sci. U. S. A. 2005;102(26):9294–9299. doi: 10.1073/pnas.0503596102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Agudelo J., Lu K., Goetz D.H., Hansell E., Chen Y.T., Roush W.R., McKerrow J., Craik C.S., Amberg S.M., Simmons G. Inhibitors of SARS-CoV entry - identification using an internally-controlled dual envelope pseudovirion assay. Antivir. Res. 2011;92(2):187–194. doi: 10.1016/j.antiviral.2011.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z., Peng L., Wang X., Xiang Y., Tong A. A new colorimetric strategy for monitoring caspase 3 activity by HRP-mimicking DNAzyme-peptide conjugates. Analyst. 2014;139(5):1178–1183. doi: 10.1039/c3an02028b. [DOI] [PubMed] [Google Scholar]
- Zhou Y., Vedantham P., Lu K., Agudelo J., Carrion R., Nunneley J.W., Barnard D., Pöhlmann S., McKerrow J.H., Renslo A.R., Simmons G. Protease inhibitors targeting coronavirus and filovirus entry. Antivir. Res. 2015;116:76–84. doi: 10.1016/j.antiviral.2015.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler C.M., Eisenhauer P., Bruce E.A., Weir M.E., King B.R., Klaus J.P., Krementsov D.N., Shirley D.J., Ballif B.A., Botten J. The lymphocytic choriomeningitis virus matrix protein PPXY late domain drives the production of defective interfering particles. PLoS Pathog. 2016;12(3):e1005501. doi: 10.1371/journal.ppat.1005501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zipper H., Brunner H., Bernhagen J., Vitzthum F. Investigations on DNA intercalation and surface binding by SYBR Green I, its structure determination and methodological implications. Nucleic Acids Res. 2004;32(12):e103. doi: 10.1093/nar/gnh101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuck P., O’Donnell G.T., Cassaday J., Chase P., Hodder P., Strulovici B., Ferrer M. Miniaturization of absorbance assays using the fluorescent properties of white microplates. Anal. Biochem. 2005;342(2):254–259. doi: 10.1016/j.ab.2005.04.029. [DOI] [PubMed] [Google Scholar]
- Zweitzig D.R., Riccardello N.M., Sodowich B.I., O’Hara S.M. Characterization of a novel DNA polymerase activity assay enabling sensitive, quantitative and universal detection of viable microbes. Nucleic Acids Res. 2012;40(14):e109. doi: 10.1093/nar/gks316. [DOI] [PMC free article] [PubMed] [Google Scholar]