Abstract
Purpose
To evaluate the efficacy and safety of neural vision enhancement technology (NVC, NeuroVision, Inc.) to improve visual acuity and contrast sensitivity function in eyes with low myopia.
Setting
Singapore Eye Research Institute, Singapore, Singapore.
Methods
This noncomparative interventional case series comprised 20 Asian adults between 19 and 53 years of age with low myopia (cycloplegic spherical equivalence [SE] from −0.5 diopter [D] to −1.5 D in the worst eye; astigmatism not exceeding 0.5 D in either eye; uncorrected visual acuity [UCVA] ≤0.7 logMAR) who had NVC treatment. The main outcome measures were distance UCVA, uncorrected contrast sensitivity, refraction, accommodative amplitude, and safety.
Results
All eyes had improvement in UCVA and contrast sensitivity. After treatment, the mean distance UCVA improved by a mean of 2.1 lines on the Early Treatment Diabetic Retinopathy Study logMAR chart. The mean contrast sensitivity improved over a range of spatial frequencies on sine-wave contrast sensitivity chart testing (1.5 to 18 cycles per degree). Follow-up data up to 12 months posttreatment showed that the gains were retained. Treatment did not alter refraction (mean spherical equivalent) or accommodative amplitudes. No adverse effects were reported.
Conclusion
Preliminary evidence suggests NVC treatment is safe and improves UCVA and uncorrected contrast sensitivity in adult patients with low myopia.
The term perceptual learning describes a process whereby practicing certain visual tasks leads to an improvement in visual performance. Several studies of this phenomenon suggest that learning occurs as a result of modification of neural processes at the primary visual cortex in adults.1, 2, 3, 4, 5 The human visual system consists of a highly sophisticated optical processing system that begins when the cornea and lens conduct an optical image onto the retina and leads to a hierarchy of progressive levels of visual processing, starting from light detection and transduction in the eye by photoreceptors through several stages of spatial integration. Each stage forms receptive fields of increasing complexity.
Contrast is one of the most important parameters activating cortical cells involved in vision processing.6 Responses of individual neurons to repeated presentations of the same stimulus are highly variable, with signal and noise levels imposing a fundamental limit on the reliable detection and discrimination of visual signals by individual cortical neurons.7, 8, 9, 10 Neural interactions determine the sensitivity for contrast at each spatial frequency, and the combination of neural activities make up an individual's contrast sensitivity function (CSF).11 The relationship between neuronal responses and perception are mainly determined by the signal-to-noise ratio (SNR) of the neuronal activity, and the brain pools responses across many neurons to average out noisy activity of single cells, improving SNR and leading to significantly improved visual performance.12
Several studies show that the noise of individual cortical neurons can be modulated by appropriate choice of stimulus conditions.13, 14 Polat et al.13, 14, 15, 16, 17 showed that contrast sensitivity at low levels can be increased through control of stimulus parameters. The typical building block of these visual stimulations is the Gabor patch ( Figure 1), which efficiently activates and matches the shape of the receptive field in the visual cortex. Polat et al.13 found that contrast sensitivity in adult human subjects at low levels can be increased by a factor of 2 through specific control of the Gabor patch parameters. This stimulation-control technique is called lateral masking, in which collinearly oriented flanking Gabors are displayed in addition to the target Gabor image. When subjects are practicing contrast modulation under a very precise and subject-specific stimuli regimen, a significant improvement in contrast sensitivity is achieved. It is these neural modifications that are the basis of brain plasticity, which relates to the ability of the nervous system to adapt to changed conditions, sometimes after injury or stroke, but more commonly in acquiring new skills.1 Brain plasticity has been shown in many basic tasks,18 with evidence pointing to physical modifications in the adult cortex during repetitive performance.1 Several studies show that the plasticity of neural interactions resulting from repetitive performance of specific visual tasks lead to improved visual performance, with retention up to 3 years of retesting. An increased range of neuronal excitatory interactions and reduced inhibition19 were observed in subjects with normal vision and in monkeys. These studies4, 14 pointed to activity-dependent plasticity of the visual cortex, where the specific connections activated throughout repetitive performance are modified, leading to improved performance, thus underpinning the concept of perceptual learning.
Figure 1.

Manipulation of Gabor stimuli.
Work on perceptual learning by Polat and others has been adopted for clinical use in the form of a computerized, Internet-based perceptual learning program developed by NeuroVision, Inc. The NeuroVision (NVC) correction technology probes specific neuronal interactions, using a set of patient-specific stimuli that improve neuronal efficiency and induce improvement of CSF due to a reduction in noise and increase in signal strength, resulting in improved spatial resolution or visual acuity. Initially developed for the treatment of adult amblyopia, NVC has been shown to be effective in inducing a sustained improvement in CSF and visual acuity in patients who were part of a prospective randomized clinical trial of adult unilateral amblyopia.20 In this U.S. Food and Drug Administration (FDA) monitored prospective masked controlled study (FDA 510(K) approval given in August 2001), 54 adult amblyopic patients (mean age 35 years) were randomized to amblyopic NVC treatment or a placebo vision-training program. Pretreatment visual acuity in both study arms was 0.42 logMAR, and this improved by 2.5 lines to 20/30 in the NVC treatment group, with no improvement in the control group. This increase in acuity was corroborated by a commensurate increase in CSF to within the normal range. These improvements in acuity and CSF were sustained after 12 months.
The efficacy of NVC treatment may be ascribed to the fact that amblyopia is characterized by several functional abnormalities in spatial vision, including reduction in visual acuity and CSF21 that occur as a consequence of optical defects such as anisometropia22 or strabismus, which NVC treatment directly addresses. The reduction in CSF, pronounced at high spatial frequencies, is believed to result from a low SNR. A low SNR limits performance on letter identification.23 Generally, it is known that CSF (especially at higher spatial frequencies) is closely related to resolution (visual acuity). Patients with amblyopia also have abnormal neural interactions,21 reduced excitation, and increased inhibition, an effect that underlies deficient contrast response. Finally, there is mounting evidence that neural plasticity persists in adult amblyopia22, 24, 25, 26, 27 as in several studies of visual improvement in adult amblyopia in which visual loss in the good eye resulted in improved vision in the amblyopic eye. This challenges the current dogma that adult amblyopia is irreversible and untreatable.
The NVC treatment has also been adapted to treat nonamblyopic cases of visual blur or optical defocus in individuals with low myopia. In simple myopia, in which a “front-end” optical defect in emmetropization develops after the critical period, there is often a mismatch between the optical defect and the neural connections formed during early childhood. The neuronal connectivity has developed normally and is capable of processing images efficiently; however, the visual input is subnormal and limited by optical defocus. The visibility of high spatial frequencies is perceived as low contrast even when their physical contrast is high. Thus, CSF is reduced at the high spatial frequencies, resembling the amblyopic CSF, which, as a consequence, degrades visual acuity. In particular, reduced signal strength is expected to degrade letter identification, as demonstrated by Solomon and Pelli's study.23 In this study, the level of noise was systematically increased; this was followed by a parallel degradation in letter recognition. Activation of neurons in the visual cortex is directly related to signal strength (contrast); when the effective contrast is low, neurons are weakly activated. Thus, the SNR is low, which limits performance on letter identification (visual acuity), as indicated in the previously mentioned study.
We performed a prospective noncontrolled preliminary clinical study as a prelude to a formal randomized clinical trial to evaluate the efficacy and safety of NVC correction technology in improving uncorrected visual acuity (UCVA) and CSF in adults with low myopia.
Subjects and methods
Study Population
Study subjects consisted of 20 adults with low myopia of cycloplegic spherical equivalence (SE) within the range of −0.5 diopter (D) to −1.5 D in the worst eye and with astigmatism not exceeding 0.5 D in either eye. Inclusion criteria included a stable refractive state with no increase beyond 0.5 D over the past 6 months, UCVA better than 0.7 logMAR, and best spectacle-corrected visual acuity (BSCVA) better than 0.05 logMAR (Early Treatment Diabetic Retinopathy Study [ETDRS] logMAR charts). Exclusion criteria included ocular condition or cause of reduced visual acuity other than simple myopia and/or astigmatism, myopia-related ocular complications, diabetes mellitus, previous ocular surgery, pregnancy, and altered cognitive or emotional state that might impair the subject's ability to perform treatment.
Written informed consent was obtained from all study subjects before the study was initiated. The study and protocol conformed to the tenets of the Declaration of Helsinki and were approved by the Singapore Eye Research Institute Review Board. Patients were chosen from healthy volunteers at the Outram Medical Campus.
Procedures
Study phases comprised a baseline screening and enrollment visit, NVC evaluation and initiation of treatment, an end-of-treatment (EOT) examination, and follow-up visits for up to 1 year.
Baseline Screening and Enrollment
Complete medical and ophthalmic histories were taken. This was followed by a baseline examination that included manifest subjective and objective refractions, cycloplegic subjective and objective refractions, accommodative amplitude (RAF rule), distance BSCVA and UCVA using 4 m distance ETDRS charts with a self-illuminating light box with a background luminance of 50 to 100 cd/m2, uncorrected and corrected CSF using a wall-mounted sine-wave contrast test (SWCT) chart (Stereo Optical Co.) at 3 m with controlled room lighting (140 cd/m2, within the range of 68 to 240 cd/m2 specified by the manufacturer), near visual acuity, slitlamp biomicroscopy, and fundus evaluation by binocular indirect ophthalmoscopy.
NeuroVision Treatment
NeuroVision technology involves automated proprietary algorithms delivered to treatment workstations. The NeuroVision Data Center was located in a secured hosting facility in Singapore and is composed of a cluster of computer servers and databases, storing all relevant clinical and treatment data. For the treatment of low myopia, the NVC LM1 algorithm was used. Algorithms are patient-specific and adaptive to subject response, thus providing analysis of each subject's dysfunctions, resulting in individualized treatment algorithms. Improved visual function is achieved through repetitive performance of interactive visual perception tasks (VPTs), in which subject-specific stimuli are administered on a treatment workstation. The NVC treatment may be clinic based or home based and adapted to the performance of each subject. In this study, patients were offered the option of home-based or clinic-based treatment.
The primary stimulus used for treatment is the Gabor patch. Gabors are local gray-level gratings with spatial frequencies of 1.5 to 12.0 cycles per degree (cpd) modulated from a background luminance of 40 cdm−2 (Figure 1). They are widely used in visual neurosciences and have been shown to efficiently describe the shape of receptive fields of neurons in the primary visual cortex. In all experiments, the standard deviation of the Gabor is maintained equal to the wavelength (σ = λ). A Philips multiscan 107P color monitor was used to project the interactive VPTs using a standard personal computing system (HP Compaq EVO D220 or equivalent with Intel P4 processor, 256 MB SDRAM or more, NVIDIA Vanta 16 MB 4X AGP Graphics Card or better, and Windows XP operating system).
The effective size of the monitor screen is 24 cm × 32 cm, which at a viewing distance of 150 cm subtends an angle of 9 degrees × 12 degrees. Subjects have treatment sessions in a dark cubicle or room in which the only ambient light is from the display screen. They wear headphones to hear auditory responses and to reduce external ambient noise. For home treatments, study investigators ensured similar conditions prevailed.
Initial Performance Evaluation Sessions
Before initiation of the training exercises, 2 evaluation sessions were performed to measure the subject's specific basic spatial functions such as contrast sensitivity and spatial interactions, the latter representing degrees of cortical suppression and facilitation. From these sessions, initial treatment training parameters were selected.
Training Sessions
Training sessions involved determination of contrast threshold attained, which was measured by a procedure in which the study subject is given a VPT and is required to choose between 2 forced-choice alternatives. A typical VPT comprises 2 consecutive displays; 1 of the pair displays the target Gabor the subject should identify. The target is presented in the first or second of 2 images, each lasting 80 to 320 ms, at an interval of 500 ms. The subject is seated 1.5 m from the screen and uses a 2-button mouse to activate the presentation of each pair of images at his or her own pace. With subjects instructed to keep both eyes open at all times, detection of the target Gabor stimuli in the first of the 2 images requires a “left click” response with the mouse, while detection of the target GS in the second of the 2 images requires a “right” click for the correct response. Subjects are informed of a wrong response by an auditory response. Depending on the level of UCVA, at the start of treatment, standardized myopic undercorrection in the form of training glasses of −1.0 D or −0.5 D or no glasses are prescribed for the treatment sessions. These glasses are subsequently reduced in power or removed as visual acuity improves. In some instances, to focus treatment visual training on the worst eye, a neutral density filter to fog vision over the better eye is used in the training glasses. Subjects are presented with a series of Gabor stimuli with the following parameters dynamically controlled: number of Gabors, spatial arrangement, global and local orientation, target–flankers separation, exposure time, contrast, spatial frequency (Figure 1).
Treatment sessions involved contrast detection tasks with Gabors, with and without flanking collinear high contrast patches. Through the treatment sessions, the spatial frequency and orientation of the stimuli were changed, starting with lower spatial frequencies and progressively moving to the higher ones, with varied orientations at each size. Thresholds for the contrast-detection task were measured with a one-up/three-down staircase (with steps of 0.1 log units), which was used to estimate the stimulus strength at the 79% accuracy level.
After each treatment session was completed, the session results were automatically transmitted to the NeuroVision Data Center, where they were automatically analyzed. If the subject was progressing (ie, the performance was within a certain range), the next session parameters were generated toward increased levels of difficulty. Otherwise, repetition might occur, but with a new set of parameters enabling the subject to perform fundamental tasks within normal ranges. In this way, subsequent sessions were determined on an individual response basis using the automated expert system algorithms and were dependent on the study subject's performance during the previous session in relation to a standard performance of emmetropic subjects with normal vision.
The total number of treatment sessions was determined by the patient's visual response during the course of treatment. Treatment sessions were approximately 30 minutes in duration and were administered 2 to 3 times a week. After every 5 sessions, the UCVA and CSF were tested to continuously monitor the subject's progress; treatments were continued until no further visual improvement occurred. Maximum improvement is subject dependent, typically achieved in approximately 20 to 30 sessions over a course of 3 months.
Constant monitoring of each study subject by the algorithms at the NeuroVision Data Center ensures real-time feedback with regard to the individual subject's compliance and performance. Study noncompliance, which could result in study termination, included subjects absent for more than 2 weeks and subjects who were performing fewer than 9 treatment sessions per month.
Outcomes Assessment
All study measurements and assessments were performed in an unmasked manner by institutional research optometrists at the Singapore Eye Research Institute Myopia Research Clinic who were not directly involved with the study. The primary outcome measure was UCVA measured by ETDRS charts and the secondary outcome measure, CSF using SWCT charts.
End-of-Treatment Decision
Subjects would have treatment termination according to any of the following EOT criteria:
-
1.
Failure of treatment defined as no improvement in the UCVA of 0.1 logMAR or more than 40% of the required improvement from baseline in either eye at any of the first 3 visual acuity tests (ie, after 15 sessions).
-
2.
Improvement in visual acuity ceases: UCVA no longer improves more than 0.05 logMAR in either eye in the last 3 visual acuity tests from visual acuity test 4 onward.
-
3.
Subject has completed 40 treatment sessions.
Following the EOT decision, the subject was scheduled for the EOT examination, which was a repetition of the baseline examination minus the cycloplegic refraction. If the EOT manifest refraction differed from the baseline manifest refraction by more than 0.5 D, cycloplegic refraction was performed. Subjects were requested to complete a quality of vision–quality of life (QOV–QOL) questionnaire upon termination of treatment.
Follow-up Visits
Subjects visited the clinic every 3 months for up to 1 year posttreatment. At each visit, the subject repeated the EOT examination with cycloplegic refraction performed at the 1-year visit. Subjects were requested to complete a final QOV–QOL questionnaire at each follow-up visit.
Results
Twenty Asian adults (10 men, 10 women) with a mean age of 32.5 years (range 19 to 53 years) were recruited for the study. The mean subjective cycloplegic SE was −1.08 D in all eyes, with −1.26 D in the worst eye, and +0.90 D in the best eye. Of the 20 subjects, all of whom completed treatment, 16 were available for the 6-month follow-up visit and 11 returned for the 12-month visit; a major reason for the loss to follow-up related to the ongoing severe acute respiratory syndrome (SARS) crisis in Singapore that occurred during the course of the study. The SARS outbreak precluded study patients from returning to the clinic due to severe infection control and isolation restrictions on patient mobility between hospital institutions during this period.
Improvement in Uncorrected Visual Acuity Immediately After Treatment
At the baseline examination, the mean distance UCVA in study patients was 0.31 logMAR (95% confidence interval [CI], 0.00-0.70). At the EOT examination, the mean UCVA had improved to 0.10 logMAR (95% CI, 0.12-0.40), a mean improvement of 2.1 logMAR lines ( Figure 2). All eyes had gains in UCVA, but to a varying extent (range 0.2 to 5.2 logMAR lines and 1 to 26 letters). Further analysis of the data showed that although all eyes showed an improvement in UCVA, the eyes with worse UCVA at baseline had greater improvement than eyes with better baseline UCVA ( Table 1).
Figure 2.
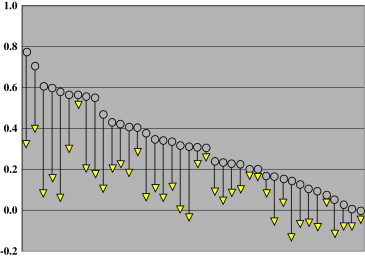
Improvement in UCVA immediately after treatment in 40 eyes of the 20 patients before and after NVC treatment. Open circles indicate the initial pretreatment UCVA in each eye. The yellow triangles indicare the level of UCVA at termination of treatment in each corresponding eye.
Table 1.
Improvement in UCVA as a function of the baseline UCVA (logMAR units).
| Improvement |
|||
|---|---|---|---|
| Baseline UCVA | Eyes (n) | Mean | Range |
| Better than 0.2 | 14 | 0.11 | 0.02–0.28 |
| 0.2 to 0.4 | 12 | 0.20 | 0.04–0.36 |
| Worse than 0.4 | 14 | 0.31 | 0.04–0.50 |
UCVA = uncorrected visual acuity
Retention of Vision Improvement at 6 and 12 Months
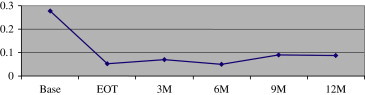
Sixteen subjects completed 6 months of follow-up after EOT. Ninety percent of the visual improvement in UCVA was maintained (from 0.30 logMAR before treatment to 0.08 logMAR immediately after treatment and 0.10 logMAR after 6 months) at 0.10 logMAR ( Figure 3). Eleven subjects completed the full 12 months of follow-up, with retention of 85% of the UCVA improvement (from 0.265 logMAR before treatment to 0.045 logMAR immediately after treatment and 0.08 logMAR after 12 months) ( Figure 4).
Figure 3.
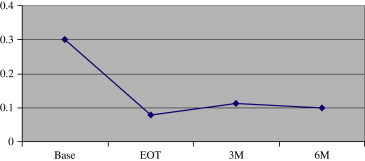
Vision retention 6 months after treatment (n = 16) (Base = baseline; EOT = end of treatment; M = month).
Figure 4.
Vision retention at 12 months after treatment (n = 11) (Base = baseline; EOT = end of treatment; M = month).
Improvement in Contrast Sensitivity Function
Mean CSF improved posttreatment over a range of spatial frequencies by the SWCT charts. This improvement was sustained at the 12-month visit ( Figure 5 and Table 2).
Figure 5.
Contrast sensitivity function before and after treatment (mean of 11 patients with 12-month follow-up).
Table 2.
Contrast sensitivity function values at baseline, EOT, and 12 months posttreatment.
| Contrast Sensitivity Function |
|||||
|---|---|---|---|---|---|
| Exam | 1.5 cpd | 3 cpd | 6 cpd | 12 cpd | 18 cpd |
| Baseline | 44 | 62 | 45 | 17 | 5 |
| EOT | 76 | 106 | 103 | 38 | 13 |
| 12 mo post | 66 | 109 | 95 | 33 | 13.5 |
cpd = cycles per degree; EOT = end of treatment; 12 mo post = 12 months post reatment
Dependency on Glasses or Contact Lenses
Subjects were given subjective QOV questionnaires at the baseline and at the end of the treatment. Analysis of the answers showed that almost all subjects (93.3%) reported less dependence on glasses or contact lenses after the treatment, with many of them (46.7%) not using glasses or contact lenses at all ( Figure 6, Figure 7).
Figure 6.
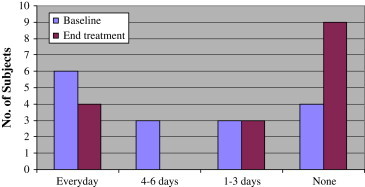
Dependency on glasses/contact lenses (“How many times a week do you wear your glasses/contact lenses?”).
Figure 7.
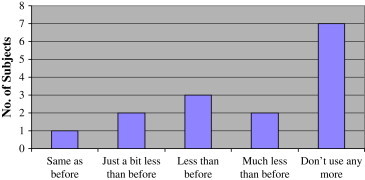
Dependency on glasses/contact lenses after treatment (“Following treatment, do you use your glasses/contact lenses less than before?”).
Effect on Refractive Error and Accommodative Amplitude
The mean SE was not significantly altered after treatment. The mean baseline cycloplegic SE and mean baseline manifest SE were −1.078 D and −1.109 D, respectively. The EOT mean manifest SE was −1.043 D. Accommodative amplitude also did not alter significantly posttreatment (mean amplitude 8.61 D at baseline and 8.73 D EOT).
Complications
No complications occurred during the study. No treated eye had a drop in UCVA or BCVA, and no subject reported adverse effects. Manifest and cycloplegic refractions remained essentially unchanged for the duration of the study.
Discussion
The study reports the efficacy and safety of NVC vision correction technology, a noninvasive perceptual learning computerized program to enhance the UCVA and uncorrected CSF in individuals with low myopia (<−1.5 D).
The mean improvement of 2.1 lines in logMAR UCVA was of sufficient magnitude clinically. If we assume measurement noise to be a difference of 1 logMAR unit, the measured improvement of a mean of 2.1 logMAR units would appear to be real and beyond measurement variability. For patients with relatively good UCVA with initially low myopia, a recorded improvement is clinically important.
In addition, this visual improvement was supported by the subjects' subjective impression as the QOV questionnaire showed less dependence on optical aids after treatment.
The increase in CSF further corroborated the visual acuity improvement (Table 2). It supports the original hypothesis of how the treatment works, and we are not aware of any other currently available treatment that can improve CSF.
Also important is the consistent retention of effect of up to 12 months. In retrospect, this is not that surprising in view of the “memory effect” of higher cortical learning programs. After all, one never quite forgets how to ride a bicycle. That compliance plays a role in the efficacy of treatment may also explain why some study subjects responded less effectively to treatment, and this remains a limitation to treatment efficacy. Variability in efficacy may also be a reflection of different individuals' “final cortical potential” that could be achieved through the perceptual learning process, which in turn is dependent on the state of inherent neural plasticity.
This study supports mounting evidence that neural plasticity is retained in the adult brain. Despite the prevailing belief that adult brain cells do not grow and that whatever limited plasticity exists in the adult brain does not involve structural remodeling, recent cortical imaging studies using 2-photon laser scanning microscopy techniques are challenging traditional concepts. Lee et al.28 confirmed the presence of dynamic structural remodeling and dendritic arbor growth occurring in GABA-positive nonpyramidal interneurons in the visual cortex of mice. Holtmaat et al.29 recently reported dendritic spine growth in the mouse adult neocortex as a direct response to novel sensory experience by whisker trimming, providing evidence that sensory input might modulate structural and functional synaptic changes in specific neocortical circuits.
As with all learning processes, the results will vary with the time and effort put in as well as the innate limits of each individual. Compliance and concentration issues are important. Follow-up studies may be designed to address these issues and find the optimal “exposure dosing.” However, individual effort and motivation will be expected to be different and have a resultant impact on variability of final result.
Limitations of this study include a small sample size, the absence of a comparative control group, and relatively subjective efficacy parameters. Care was taken to reduce the memory effect of vision testing, which included rotating logMAR charts used over a 12-month period and the use of sine-wave grating CSF charts as apposed to letter-based charts.
In conclusion, this study of NVC treatment in subjects with low myopia supports the hypothesis of perceptual learning in its ability to improve UCVA and uncorrected CSF. The results are highly encouraging and warrant further studies.
Beyond this study, we have begun a randomized placebo-controlled trial of adult subjects with low myopia recruited from military personnel in the Singapore Armed Forces and have also initiated studies to assess NVC treatment efficacy in children with early myopia progression. Studies of the utility of this treatment in enhancing vision and contrast sensitivity of postrefractive patients, patients with early presbyopia, and patients with mild to moderate degrees of foveal pathology are also in progress. These studies will further evaluate this perceptual learning technology and its prospective role in the enhancement of visual potential.
Biography

First author:
Donald T.H. Tan, FRCS, FRCOphth
Singapore Eye Research Institute; the Department of Ophthalmology, Yong Loo Lin School of Medicine, National University of Singapore; and Singapore National Eye Centre, Singapore, Singapore
Footnotes
Neither author has a financial or proprietary interest in any material or method mentioned.
References
- 1.Sagi D., Tanne D. Perceptual learning: learning to see. Curr Opin Neurobiol. 1994;4:195–199. doi: 10.1016/0959-4388(94)90072-8. [DOI] [PubMed] [Google Scholar]
- 2.Dosher B.A., Lu Z.-L. Perceptual learning reflects external noise filtering and internal noise reduction through channel reweighting. Proc Natl Acad Sci USA. 1998;95:13988–13993. doi: 10.1073/pnas.95.23.13988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dosher B.A., Lu Z.-L. Mechanisms of perceptual learning. Vision Res. 1999;39:3197–3221. doi: 10.1016/s0042-6989(99)00059-0. [DOI] [PubMed] [Google Scholar]
- 4.Gilbert C.D., Sigman M., Crist R.E. The neural basis of perceptual learning. Neuron. 2001;31:681–697. doi: 10.1016/s0896-6273(01)00424-x. [DOI] [PubMed] [Google Scholar]
- 5.Crist R.E., Li W., Gilbert C.D. Learning to see: experience and attention in primary visual cortex. Nat Neurosci. 2001;4:519–525. doi: 10.1038/87470. [DOI] [PubMed] [Google Scholar]
- 6.Albrecht D.G., Hamilton D.B. Striate cortex of monkey and cat: contrast response function. J Neurophysiol. 1982;48:217–237. doi: 10.1152/jn.1982.48.1.217. [DOI] [PubMed] [Google Scholar]
- 7.Tolhurst D.J., Movshon J.A., Dean A.F. The statistical reliability of signals in single neurons in cat and monkey visual cortex. Vision Res. 1983;23:775–785. doi: 10.1016/0042-6989(83)90200-6. [DOI] [PubMed] [Google Scholar]
- 8.Shadlen M.N., Newsome W.T. Noise, neural codes and cortical organization. Curr Opin Neurobiol. 1994;4:569–579. doi: 10.1016/0959-4388(94)90059-0. [DOI] [PubMed] [Google Scholar]
- 9.Geisler W.S., Albrecht D.G. Bayesian analysis of identification performance in monkey visual cortex: nonlinear mechanisms and stimulus certainty. Vision Res. 1995;35:2723–2730. doi: 10.1016/0042-6989(95)00029-y. [DOI] [PubMed] [Google Scholar]
- 10.Shadlen M.N., Newsome W.T. The variable discharge of cortical neurons: implications for connectivity, computation, and information coding. J Neurosci. 1998;18:3870–3896. doi: 10.1523/JNEUROSCI.18-10-03870.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Polat U. Functional architecture of long-range perceptual interactions. Spatial Vis. 1999;12:143–162. doi: 10.1163/156856899x00094. [DOI] [PubMed] [Google Scholar]
- 12.Geisler W.S., Albrecht D.G. Visual cortex neurons in monkeys and cats: detection, discrimination, and identification. Vis Neurosci. 1997;14:897–919. doi: 10.1017/s0952523800011627. [DOI] [PubMed] [Google Scholar]
- 13.Polat U., Sagi D. Lateral interactions between spatial channels: suppression and facilitation revealed by lateral masking experiments. Vision Res. 1993;33:993–999. doi: 10.1016/0042-6989(93)90081-7. [DOI] [PubMed] [Google Scholar]
- 14.Polat U., Sagi D. The architecture of perceptual spatial interactions. Vision Res. 1994;34:73–78. doi: 10.1016/0042-6989(94)90258-5. [DOI] [PubMed] [Google Scholar]
- 15.Polat U., Sagi D. Spatial interactions in human vision: from near to far via experience- dependent cascades of connections. Proc Natl Acad Sci USA. 1994;91:1206–1209. doi: 10.1073/pnas.91.4.1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Polat U., Mizobe K., Pettet M.W. Collinear stimuli regulate visual responses depending on cell's contrast threshold. Nature. 1998;391:580–584. doi: 10.1038/35372. [DOI] [PubMed] [Google Scholar]
- 17.Kasamatsu T., Polat U., Pettet M.W., Norcia A.M. Colinear facilitation promotes reliability of single-cell responses in cat striate cortex. Exp Brain Res. 2001;138:163–172. doi: 10.1007/s002210100675. [DOI] [PubMed] [Google Scholar]
- 18.Polat U., Sagi D. Plasticity of spatial interactions in early vision. In: Julesz B., Kovács I., editors. Maturational Windows and Adult Cortical Plasticity; Santa Fe Institute Studies in the Science of Complexity. Addison-Wesley; Reading, MA: 1995. vol. XXIV:111–125. [Google Scholar]
- 19.Zenger B., Sagi D. Isolating excitatory and inhibitory nonlinear spatial interactions involved in contrast detection. Vision Res. 1996;36:2497–2513. doi: 10.1016/0042-6989(95)00303-7. [DOI] [PubMed] [Google Scholar]
- 20.Polat U., Ma-Naim T., Belkin M., Sagi D. Improving vision in adult amblyopia by perceptual learning. Proc Natl Acad Sci USA. 2004;101:6692–6697. doi: 10.1073/pnas.0401200101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Polat U., Sagi D., Norcia A.M. Abnormal long-range spatial interactions in amblyopia. Vision Res. 1997;37:737–744. doi: 10.1016/s0042-6989(96)00154-x. [DOI] [PubMed] [Google Scholar]
- 22.Zhou Y., Huang C., Xu P. Perceptual learning improves contrast sensitivity and visual acuity in adults with anisometropic amblyopia. Vision Res. 2006;46:739–750. doi: 10.1016/j.visres.2005.07.031. [DOI] [PubMed] [Google Scholar]
- 23.Solomon J.A., Pelli D.G. The visual filter mediating letter identification. Nature. 1994;369:395–397. doi: 10.1038/369395a0. [DOI] [PubMed] [Google Scholar]
- 24.Simmers A.J., Gray L.S. Improvement of visual function in an adult amblyope. Optom Vis Sci. 1999;76:82–87. doi: 10.1097/00006324-199902000-00014. [DOI] [PubMed] [Google Scholar]
- 25.Fronius M., Cirina L., Cordey A., Ohrloff C. Visual improvement during psychophysical training in an adult amblyopic eye following visual loss in the contralateral eye. Graefes Arch Clin Exp Ophthalmol. 2005;243:278–280. doi: 10.1007/s00417-004-1014-8. [DOI] [PubMed] [Google Scholar]
- 26.Kaarniranta K., Kontkanen M. Visual recovery of the amblyopic eye in an adult patient after loss of the dominant eye. Acta Ophthalmol Scand. 2003;81:539. doi: 10.1034/j.1600-0420.2003.00127.x. [letter] [DOI] [PubMed] [Google Scholar]
- 27.Rahi J.S., Logan S., Borja M.C. Prediction of improved vision in the amblyopic eye after visual loss in the non-amblyopic eye. Lancet. 2002;360:621–622. doi: 10.1016/S0140-6736(02)09775-1. [DOI] [PubMed] [Google Scholar]
- 28.Lee W.C., Huang H., Feng G. Dynamic remodeling of dendritic arbors in GABAergic interneurons of adult visual cortex. PLoS Biol. 2006;4(2):e29. doi: 10.1371/journal.pbio.0040029. http://biology.plosjournals.org/archive/1545-7885/4/2/pdf/10.1371_journal.pbio.0040029-S.pdf http://biology.plosjournals.org/perlserv/?request=get-document&doi=10.1371/journal.pbio.0040126 Available at: correction 4(5):e126. Available at: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Holtmaat A., Wilbrecht L., Knott G.W. Experience-dependent and cell-type-specific spine growth in the neocortex. Nature. 2006;441:979–983. doi: 10.1038/nature04783. [letter] [DOI] [PubMed] [Google Scholar]


