Abstract
Viral infections are the most frequent triggers of wheeze and asthma and yet their role in the development of symptoms remains controversial. Pre-existing airway abnormalities contribute to early virus-induced symptoms which usually remit in early childhood, whereas an interaction with airway inflammation causes exacerbations in asthma. However, the distinction between these two groups and the reason why some but not other children wheeze with viral infections is still debated. The effect of early infections on the developing immune system is also complex. The successful maturation of the T-cell response from a predominantly type 2 (atopic predisposition) at birth to a predominantly type 1 (optimal viral immunity) response, is influenced by genetic factors and the number of infections, as both are known to affect outcome. The relative parts played by predisposition and immunomodulation by early infections in later development of asthma are still controversial. These contentions are gradually being resolved by detailed prospective studies.
Keywords: virus, wheeze, asthma, child
INTRODUCTION
The association between virus infections and acute wheezing episodes has been recognised for decades.1 Indeed, wheezing with colds is the most common form of wheezing at all ages.2 Although the nature of the debate has evolved over the years, many of the ‘old chestnuts’ are still relevant more than 30 years on, including if (or when) episodic virus-induced wheezing in young children should be considered to be asthma. The discussion continues as to whether or not severe early virus infections play a part in the development of asthma by immune modulation and/or airway damage, or whether children who present with virus-induced wheezing have a pre-existing predisposition. The subject has become more complicated since it has been recognised that early infections may protect against asthma, the so-called ‘hygiene hypothesis’.3., 4.
Since acute attacks related to virus infections are responsible for a great deal of distress to wheezy infants and children with asthma and treatment of this condition is less than satisfactory, it is clearly important to understand the mechanism by which respiratory viruses trigger wheezing. Also, as the prevalence of asthma has increased in recent years in Westernised societies, it is necessary to understand the role of virus infections in the development of asthma, as this could have implications for immunisation programmes and expensive antiviral strategies, as well as the development of new approaches to therapy. This article will attempt to put these issues into the context of recently published evidence and reviews of the subject.
VIRUSES AND ACUTE WHEEZING EXACERBATIONS
Viruses have been implicated, using culture, serology or polymerase chain reaction, in over 80–85% of children with acute wheezing exacerbations or asthma.5 In infants, the most common culprits are respiratory syncytial virus (RSV) and para-influenza virus5 but rhinoviruses account for the majority of episodes in children over 2 years.6., 7.
Effective host defence against infections, including viruses, involves the production of TH-1 type cytokines such as IFN-γ and IL-2 by CD4+ T lymphocytes. Infants are born with a tendency to respond to infection by producing cytokines with a TH-2 profile (IL-4, IL-10), so have an impaired TH-1 response.3., 8. Since the immune response to infections also involves CD8+ lymphocytes, it is probably more appropriate to label the responses as types 1 and 2.3 With increasing maturity, the immune response switches from a predominantly type 2 to a predominantly type 1 response by the effect of repeated infections on antigen-presenting cells which produce IL-12. In the absence of infections or with an atopic predisposition, this switch from a type 2 to a predominantly type 1 response is impaired (Fig. 1 ).
Figure 1.
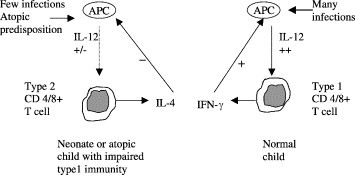
Maturation of T-cell immunity: the effect of age, multiple infections and predisposition on the development of a type 1 or type 2 profile (based on ref. 3). IL, interleukin; IFN-γ, interferon-gamma.
MECHANISM OF VIRUS-INDUCED WHEEZE
All infants experience infection with RSV and all children have recurrent rhinovirus infections. Why only a minority develop airways obstruction (wheeze) is still a matter of debate. Some viruses, for example RSV and the influenza viruses, actually infect the lower airways, so have the potential to cause airway obstruction by an acute inflammatory process. Rhinovirus RNA has been found in bronchoalveolar lavage and biopsy samples of inoculated volunteers but the significance for this is still not clear.9 A number of mechanisms have been identified that could contribute to airway obstruction and the increased bronchial responsiveness seen in association with an acute viral infection in asthma and, to a lesser extent, in normal subjects (Table 1 and Fig. 2 ). These have recently been reviewed.3., 9., 10., 11. Much of the data have been obtained with experimental viral infections in adults who had mild asthma but not necessarily with a history of virus-induced exacerbations, or who were normal. The extent of each process may vary according to the host’s immune response, the presence or absence of any underlying airway inflammation and the particular virus infection involved. There is likely to be an interaction, additive or synergistic, between the acute viral inflammation and the asthma pathology. Of particular interest is the possibility that pre-existing airway inflammation could interfere with the host’s antiviral defence by skewing the response towards type 2, so exaggerating damage to the airway.11
Table 1.
Potential mechanisms of virus-induced wheeze (based on ref 1).
| Acute inflammation | Increased bronchial responsiveness (BHR) |
| 1. Changes in small airway function | 1. Reduced lung function |
| ∗Oedema and wall thickening | ∗Airway wall changes |
| ∗Airway obstruction | ∗Bronchoconstriction |
| 2. Alteration neural control | 2. Increase in cholinergic responsiveness |
| (evidence mainly in animal models) | ∗BHR prevented by atropine |
| ∗Neural reflex | ∗Modulation substance P |
| ∗Interference M2 receptor function | 3. Induced neurogenic inflammation |
| ∗Sensory C fibres | 4. Potentiated cholinergic neurotransmission |
| ∗Disruption of epithelial barrier | ∗Increased sensitivity to constrictor agent |
| ∗Reduction neutral endopeptidase | ∗Increased maximal response |
| 3. Neutrophil recruitment and activation | |
| ∗IL-8 and LTB4 production | Interaction with airway inflammation |
| 4. Monocytes and macrophages | 1. Increased response to allergen (Fig. 2) |
| ∗Release acute phase cytokines, e.g. TNF-α | ∗Exaggerated early phase reaction |
| 5. Mediators | ∗Enhanced late response |
| ∗LTC4 and PGF2 | ∗Increased allergen penetration |
| 6. Decreased β-adrenergic sensitivity | 2. Potentiation inflammatory cascade |
| ∗Granulocytes decreased response isoprenaline | ∗Eosinophil recruitment etc. (Fig. 2) |
| 7. Upregulation ICAM-1 receptor | 3. Inefficient antiviral response due to type |
| ∗Increased viral adhesion | 2 T-cell cytokine predominance (Fig. 1) |
Figure 2.
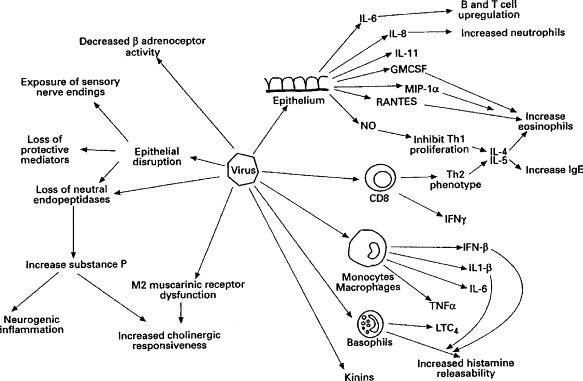
Summary of effects of a virus infection on the respiratory tract (reproduced from Thorax 1997; 52: 380–389, by kind permission of the BMJ Publishing Group). IL, interleukin; INF, interferon; TN, tumour necrosis factor; L, leukotriene; NO, nitric oxide.
EPIDEMIOLOGY OF VIRAL WHEEZE
Different wheezing phenotypes have been identified according to the age of onset and duration of wheezing in a large birth cohort follow-up study; the Tucson study.12., 13. In these influential studies, there was no report of the relation of symptoms to virus infections but the scheme provides a basis to consider the possible different underlying reasons for virus-associated wheeze in different clinical subgroups. Age cut-off points were chosen somewhat arbitrarily and may prove to be an oversimplification. The different phenotypes are related to various risk factors and these will co-exist to varying extents in different individuals, so giving rise to a spectrum of clinical presentations.14 Nonetheless, virus-induced wheezing can be categorised broadly into the clinical subgroups, with their own associated risk factors, suggested by Martinez et al. in the Tucson study (Table 2 ).12
Table 2.
Wheezing phenotypes and associated clinical characteristics.
| Transient wheeze | Episodic wheeze | Wheeze (Asthma*) |
||||||
| Persistent | Late onset | |||||||
| Age | ||||||||
| Onset | <1 year | >18 months | <1–3 years | >3 years | ||||
| Remission | <4 years | <13 years | >6 years | >6 years | ||||
| Risk factors | Maternal smoking | ? β2ADR genotype | Atopy | Atopy | ||||
| β2ADR genotype | Family history | Family history | ||||||
| Factors related to viral wheeze | Mechanical (airway size or tone) | Airway tone/dysfunction | Airway/tone dysfunction | Impaired type 1 response | ||||
| Impaired type 1 response | ||||||||
| Early airway inflammation | Airway inflammation | |||||||
| BR | ||||||||
| Birth | Increased | Normal | Normal | Normal | ||||
| Childhood | Increased | Normal | Increased | Increased | ||||
| Lung function | ||||||||
| Birth | Decreased | Normal | Normal | Normal | ||||
| Childhood | Decreased | Normal | Decreased | Decreased | ||||
| Response to Rx | ||||||||
| Bronchodilators | No | Yes | Yes | Yes | ||||
| Inhaled CS | No | No | Yes | Yes | ||||
| Symptom pattern | Episodic | Episodic | Intermittent or continuous | Intermittent or continuous | ||||
| Triggers | Viruses | ? Only | Viruses | Multiple | ||||
| virus infections | Multiple | +/− viruses | ||||||
BR, bronchial responsiveness; β2ADR, beta-2 adrenergic; CS, corticosteroid.
Associated with a family history of asthma and allergy.
Transient wheeze
It is known that children, as a group, who experience wheezing in the first 3–4 years of life but not thereafter (‘transient wheeze’) have reduced lung function and increased bronchial responsiveness12., 15., 16. prior to any respiratory illness. This predisposing airway dysfunction (reduced airway calibre and/or altered mechanics) is also associated with maternal smoking during pregnancy.12 However, transient wheeze also occurs in those whose mothers have never smoked, suggesting additional pre-natal environmental or genetic influences. For example, polymorphisms of the beta-2 receptor gene have recently been shown to influence neonatal lung function15., 17. and bronchial responsiveness.15 The association of symptoms with virus infections in those with transient wheeze was not established in the original study but it is often assumed, due to the common observation that very young children with recurrent virus-induced wheeze grow out of their symptoms in the first few years. Also viruses can be detected in the majority of acute episodes in young as well as older children.7
Transient wheeze (i.e. that remitting before the 4th birthday) may be more than one condition. Typically, these young children respond poorly to bronchodilator or corticosteroid therapy until about 18 months of age, in keeping with a mechanical basis for their symptoms. Some continue to exhibit episodic viral wheeze without changing symptom pattern, and begin to respond unequivocally to bronchodilators. As we know that infants have functioning beta receptors and can be protected from induced bronchoconstriction from 1 month of age,18 it is possible that there are two independent airway defects, one related to reduced airway calibre (and possibly maternal smoking) and the other to altered tone, both of which could result in reduced lung function at birth. There is the possibility of a genetic basis for this dysfunction of airway tone as an increase in response to a bronchodilator was associated with a polymorphism of the beta-2 adrenoceptor at amino acid 16.19 The same genotype was related to reduced lung function and increased bronchial responsiveness at birth, which in turn were found in those with transient wheeze.15 It is likely that there is overlap between transient wheeze and the group of episodic wheezers described below, in which a disorder of airway tone is also suggested.
Episodic (viral) wheeze of childhood
This condition is rather ill defined and may correspond to a category in the Tucson study which remitted between age 6 and 11 years.13 These children showed increased daily peak flow variability, suggesting an abnormality of airway dynamics unrelated to atopy. In contrast to the atopic persistent wheezers characteristic of classical asthma, these children improved through later childhood. Also from the Tucson follow-up study, children with RSV infection in early life continued to be at increased risk of episodic wheezing up to the age of 13 years but the numbers diminished with age.20 These children showed reduced pre- but not post-bronchodilator lung function. Again, neither of these studies took into account the pattern of symptoms, but they could represent the same sort of school-age children with wheezing solely related to viral infections, whose symptoms did not respond to inhaled beclomethasone.21 Taken together, these studies suggest a group of children who wheeze in association with recurrent viral infection, independently of atopy, with an abnormality of airway tone. Further support for a genetic basis for this type of wheezing, solely related to infections, is provided by family studies of children with wheezy bronchitis.22 How exactly a virus infection induces reversible bronchoconstriction in this situation is not clear. It is unlikely to be mediated via a chronic eosinophilic airway inflammatory process because of the lack of response to inhaled corticosteroids21 and because no eosinophils were found in bronchoalveolar lavage in children with symptoms only associated with virus infections (Fig. 3 ).23
Figure 3.
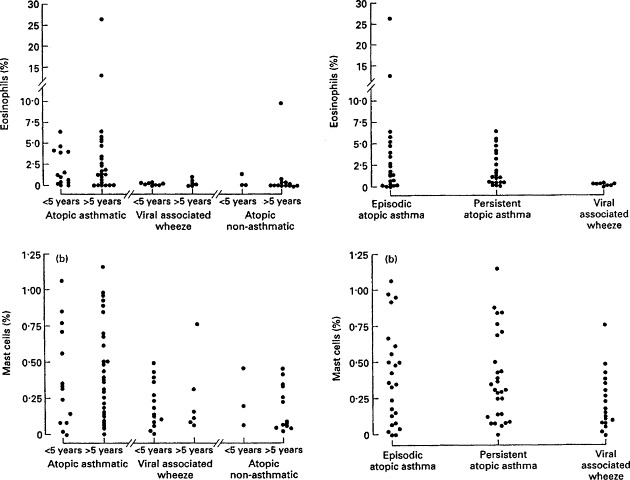
Comparison of eosinophil differential counts in bronchoalveolar lavage (BAL) fluid in different clinical subgroups, stratified according to age and atopy (reproduced from Stevenson EC et al. Bronchoalveolar lavage findings suggest two different forms of childhood asthma. Clin Exp Allergy 1997; 27: 1027–1035. By kind permission of Blackwell Science).
Virus-induced attacks in those with atopic asthma9., 10., 11., 24.
Most, but not all, children with asthma develop attacks in association with viral infections, most commonly upper respiratory tract rhinovirus. A number of mechanisms have been implicated (Table 2, Fig. 2). Infants who subsequently developed persistent wheezing showed a more marked eosinophilic response to their initial viral infection compared with those who had transient wheeze.8 This indicates that a difference between the two groups in the immune response to a virus infection occurs even in infancy, even though the clinical picture may be identical.
In those with asthma, the consequences of an acute viral infection include the necessary harmful effects of virus elimination, in addition to a possible interaction with and amplification of airway inflammation (Table 2, Fig. 2).24 Also, the type 2 skewed immune response could result in delayed viral clearance and prolonged viral inflammation. Although the sequence of events is not clearly understood, these processes would contribute to the virus-induced airways obstruction and increased bronchial responsiveness.
THE ROLE OF EARLY VIRAL INFECTIONS IN THE DEVELOPMENT OF ASTHMA AND ALLERGY
There is a paradox concerning the role of viruses and atopic disease. On the one hand, there are indications of a protective effect of early viral infections, and on the other hand, there is the view that respiratory viral infections increase the likelihood of asthma. This topic has been the subject of extensive recent reviews.3., 8., 9., 11., 25.
Is there any evidence that RSV bronchiolitis damages the growing lung or alters the immune response?
The debate as to whether severe early respiratory viral infections can damage the growing lung and enhance atopic sensitisation has continued for at least two decades. The argument has mainly focused on children with hospitalised RSV bronchiolitis. However, in these discussions, definitions are extremely important. Both the terms ‘bronchiolitis’ and ‘asthma’ are still used loosely in ways that can be very misleading.26., 27. If RSV is to be implicated in the inception of recurrent wheezing, it should be the first wheezing episode in a child < 1 year with typical severe airways obstruction, and not refer to any RSV-infected wheezy episode up to the age of 3 years. If immunomodulation by RSV infection is being postulated (see below), any subsequent wheeze should not be called ‘asthma’ unless associated with atopy or a type 2 skewed immune response.
A large number of studies have shown that recurrent wheeze is markedly increased following RSV bronchiolitis for at least 5 years.28 This increase in wheezing episodes occurred up to the age of 13 years in children with RSV-positive infections in infancy treated in the community, suggesting that severity is not a necessary factor.20 A feature of children with ‘post-bronchiolitic wheeze’ is reduced lung function and increased bronchial responsiveness compared with controls at follow-up.28 The question is whether this is due to a pre-existing abnormality causing the predisposition to wheeze or whether structural airway damage is secondary to the initial RSV infection. The finding that lung function and bronchial responsiveness are increased in children with transient wheeze prior to the first wheezy episode12., 15. strongly supports the predisposition concept.
The next contentious issue is whether or not RSV infection can influence allergic sensitisation by immunomodulation and so increase the likelihood of atopy and therefore of asthma developing. When strict entry criteria were used to define RSV bronchiolitis, by excluding children >1 year at entry and those in whom the RSV infection was not confirmed at the first wheezy episode, a review of all eligible studies concluded that post-bronchiolitic wheezing was not associated with any increased risk of atopy nor with a family history of atopy,29 making immunomodulation by RSV infections unlikely. The one glaring exception is the study by Sigurs et al. in which RSV bronchiolitis was the strongest independent risk factor for asthma and atopic sensitisation in their multivariate analysis.30 The authors defined asthma as the occurrence of at least three episodes of wheezing, which in itself does not exclude continuing recurrent viral wheeze on any pathological grounds, but it would also include those with asthma. No surprise then that ‘asthma’ by this definition was predicted by RSV infection. This study used matched controls that were enrolled contemporaneously (i.e. during a RSV epidemic). Surprisingly few of them (2%) had wheez at follow-up and neither had anyone with a family history of asthma who did not have RSV bronchiolitis. So another interpretation of their findings is that the failure to develop bronchiolitis during an RSV epidemic is a negative risk factor for the development of asthma and allergy.3 If the selection of controls was biased against development of asthma, any subsequent immunological differences in the RSV group would be similarly skewed.
Normally, the host response to a virus infection is a predominantly type 1 response with secretion of IFN-γ and IL-2 (Fig. 1). The strongest evidence that there is a causal link between RSV bronchiolitis and the development of asthma or the induction of atopic sensitisation comes from animal studies. Firstly, in guinea pigs and rats, type 2 responses have been demonstrated following RSV infections.3., 25. However, this type of response has mainly been seen when formalin-inactivated viruses have been used. Studies in mice have shown that viral infections interact with inhaled allergens to promote the development of airway inflammation and atopic sensitisation.25 In human infants, a type 2 skewed response has been indicated by low IFN-γ and relatively raised IL-4 levels in peripheral monocyte blood cells from RSV-infected infants.31 This could represent increased susceptibility to RSV infection in those with a pre-existing impaired type 1 response, or that the type 2 skewed response could be the result of the infection. The latter is supported by the finding of increased IL-10 by stimulated monocytes during convalescence from RSV bronchiolitis in children who went on to have recurrent episodes of wheezing.32 The number of subsequent wheezing episodes correlated with the convalescent IL-10 values. Since no differences were found during the acute phase, it suggests a virus-induced change in cytokine response. Whether this translates into atopic asthma still has to be determined.
The opposing view is indicated by other studies. One showed a lower cord blood IL-12 suggesting impaired type 1 immunity in those with subsequent bronchiolitis (Fig. 1), and another study showed that reduced type 1 cytokines were associated with more severe RSV disease.32 The re-interpretation of the follow-up study of infants with RSV of Sigurs et al.30 (see above) is compatible with the line of argument that a pre-existing type 2 skewed immunity occurs in those developing RSV bronchiolitis. This line of argument is re-inforced by the finding of an association between IL-4 polymorphisms and severe RSV bronchiolitis.33 Furthermore, although adults with atopic asthma are not at increased risk of rhinovirus infection, they have been shown to experience more frequent and severe lower respiratory tract symptoms.34 As a unifying explanation, it has been suggested that genetic variations in CD14 and other pattern recognition receptors may influence both clinical expression of viral disease and the development of allergy.3
It is questionable whether RSV infection is unique among viruses in its association with a subsequent increase in recurrent wheeze. Studies that have compared the outcome in infants after hospitalised RSV-positive and -negative lower respiratory tract infections or bronchiolitis have found no difference between the two.3., 35. Using polymerase chain reaction, rhinovirus has been implicated in 29% of cases of bronchiolitis,36 so it is possible that there is nothing specific about RSV apart from the fact that it is the most common cause of severe lower respiratory tract infections in infancy.
Overall, there is convincing evidence for the pre-existence of a type 2 predisposition (or failure of maturation towards type 1, Fig. 1) leading to more severe RSV or other infections in those destined to develop asthma. However, airway damage causing an additional lung function deficit cannot be excluded. Induction, or further exaggeration, of a type 2 response in some children also remains a possibility.
Upper respiratory viral infections and bronchial responsiveness
Transient increases in lung function and bronchial responsiveness have been shown in animal models and normal human volunteers with both clinical and artificially induced upper respiratory viral infections.37 In asthma, this increase is more marked with both an increase in sensitivity and maximal response to the constrictor agent detected in some subjects.24 Many of the mechanisms associated with acute viral inflammation (Table 2, Fig. 2) would be expected to increase bronchial responsiveness, which in turn is likely to be part of the virus-induced asthma exacerbation. Virus and host characteristics probably determine the size of the response, as reduced lung function and the presence of atopy increases the effect. In a recent study of virus-induced lower airway changes in adults without asthma who were inoculated with coronavirus, those with a history of viral wheeze showed a greater degree of cold symptoms, greater falls in FEV1 and larger increases in bronchial responsiveness than those without such a history.38 As no effect of atopy was seen and there were no differences in baseline measurements, an as yet unidentified additional factor is suggested.
It is not infrequently argued that RSV infection in infants can induce a prolonged state of increased bronchial responsiveness, which is responsible for the subsequent increase in recurrent wheeze.37 However, this has never been demonstrated. Furthermore, increased bronchial responsiveness has been found before the first viral infection in those with transient wheeze.12., 15.
Evidence that early viral infections protect against later development of asthma
This subject has aroused much interest in the past few years because of the findings of an effect of birth order and family size, as surrogate markers of infection load, on the development of allergy.3., 23., 36. A protective role of infections has also been seen in developing countries, where an inverse relationship between evidence of respiratory infections (serological or actual infections such as measles) and later development of atopy has been found.25 One possibility is that these findings could have resulted from a genetic bias in confined communities. This is unlikely as there was a similarly reduced prevalence of atopic disease in East Germany compared with the genetically similar population of West Germany.39 The East German children were assumed to have been exposed to more infections because of the much greater use of early daycare, which in itself has been associated with less subsequent asthma.39 There is, however, a paradox as several studies show that parental reports of lower respiratory illnesses are positively associated with later asthma.3 A little discussed reason for possible confusion is the variation in the definitions of asthma that have been used. When immune modulation is being considered as a factor in the development of asthma, two not uncommon definitions are flawed: occurrence of more than three wheezy episodes and wheeze beyond 3 years. Firstly, children with ‘transient’ wheeze often experience more than three episodes and therefore could, by this definition, be wrongly categorised as asthma. Secondly, children with recurrent wheeze following RSV infection, which is independent of atopy, may not remit until 6–13 years.20 A further explanation for the apparent paradox is put forward by Mallia et al.3 Daycare use and large family size reflect an increased viral load which includes gastrointestinal viruses and is independent of the host response. In contrast, parental reporting of symptoms reflects the host response. Children destined to have asthma have an impaired type 1 response to virus infections and are therefore at risk of more frequent and severe symptoms, which are more likely to be reported. In an adult study, designed to avoid reporting bias, asthma and controls had the same number of upper respiratory infections, assessed by polymerase chain reaction but those with asthma reported more lower respiratory symptoms.34 In support of this line of reasoning, Illi et al. showed that repeated viral infections in early life, other than those of the lower respiratory tract, protected against asthma.39
As an explanation for this ‘Hygiene hypothesis’, it has been proposed that viral infections in infancy that do not induce an acute wheezing episode promote development of a type 1 T-cell response (Fig. 1).3., 8. In the absence of infections, the default type 2 skewed response present at birth persists, with a resulting increased risk of sensitisation to aeroallergens. This theory is based on a study which showed a difference in immune response to their first respiratory infection between infants who subsequently developed persistent wheeze (asthma) and those with transient wheeze, independently of type of virus implicated.40
Prediction of later asthma
Most young children who wheeze initially present with episodes related to viral infections. Only a small proportion (in the order of 15%) continue to wheeze in later childhood and are considered to have asthma. For the first few years of life, they are clinically indistinguishable from children with ‘transient’ wheeze. It has been a long-standing aim of clinicians to predict those who will have asthma later. In theory, anti-inflammatory treatment given early could modify the outcome. From population studies, risk factors, such as personal atopy or a family history of asthma, can be used to provide relative risks of developing asthma with reasonable accuracy,41 but for use in the individual, this approach is insufficiently sensitive. Since a type 2 skewed response (increased IgE, relative eosinophilia, raised s-ECP) to infection can be seen in infants destined to have persistent wheeze,40 information obtained from a blood test during the initial episode has the potential for predictive use. However, this information is unlikely to be available routinely. The finding of RSV-specific IgE during RSV bronchiolitis correlated with subsequent wheezing episodes up to the age of 7 years but not with symptoms or atopy thereafter.42
CHRONIC INFECTIONS
One way that virus infections could perpetuate symptoms is through a chronic or latent infection. Double-stranded DNA viruses have the ability to persist in airway epithelial cells long after the acute infection has resolved.43 During these latent infections, viral genes are expressed without viral replication. Latent adenovirus has been shown in both human and animal airway epithelium. Ninety-four percent of children with steroid-resistant asthma but no controls, were found to have adenovirus antigens in their airways.44
TREATMENT OF WHEEZING RELATED TO VIRAL INFECTIONS
Little attention has been paid to symptoms specifically related to virus infections. The exception is acute RSV bronchiolitis, which is beyond the scope of this article.
Acute episodes only associated with viral infections
Wheeze solely triggered by a virus should be considered separately from asthma as the mechanisms are likely to be different (Table 1, Fig. 3).
Acute attacks
These occur largely but not exclusively in pre-school children.21 There is evidence from clinical trials that beta agonists or anticholinergic agents provide only a marginal benefit in children much below the age of 2 years,45 but are effective over this age, perhaps indicating a change of pathogenesis of virus-induced symptoms with age. Oral corticosteroids are recommended in moderate to severe symptoms at all ages, although evidence of efficacy <2 years is very limited.45 Leukotriene receptor antagonists have recently been shown to be of value in treating pre-school children following an acute wheezing episode associated with an RSV infection (the term ‘bronchiolitis’ is very confusing in this context), but no comparison with any alternative approaches is yet available.26
Prevention of viral wheeze
The 2003 BTS guidelines for asthma management do not consider the treatment for prevention of attacks of wheeze occurring only in association with viral infections. A recent meta-analysis showed no benefit from the continuous use of preventer (anti-inflammatory) medication in this situation at any age.46
Acute virus-induced attacks occurring in children with chronic asthma
Surprisingly, the prevention of virus-induced attacks in children with asthma has not been studied systematically. From clinical observation, it would seem that virus-induced attacks are more resistant to treatment than interval symptoms. Not infrequently, children have their maintenance preventer therapy increased because of continuing virus-associated symptoms without good evidence of benefit. This area needs further investigation.
PRACTICE POINTS
-
•
80–85% of acute wheezing episodes are associated with a viral infection.
-
•
Pre-existing abnormalities of lung function occur in episodic viral wheeze.
-
•
Increased viral load protects against asthma by immunomodulation.
-
•
Children with a predominant type 2 response (atopic) have more severe lower respiratory infections.
RESEARCH DIRECTIONS
-
•
Longitudinal birth cohort studies relating immune status to response to infection and respiratory outcome.
-
•
Therapeutic trials considering efficacy of anti-inflammatory agents against virus-induced symptoms in asthma.
-
•
Investigate why some children wheeze and others do not in response to virus infections.
-
•
Therapy directed against viral wheeze using recent knowledge mechanisms of rhinovirus pathology.
References
- 1.McIntosh K., Ellis E.F., Hoffman L.S. The association of viral and bacterial respiratory infections with exacerbations of wheezing in young asthmatic children. J. Pediatr. 1973;82:578–590. doi: 10.1016/S0022-3476(73)80582-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dodge R.R., Burrows B. The prevalence and incidence of asthma and asthma-like symptoms in a general population sample. Am. Rev. Respir. Dis. 1980;122:575–576. doi: 10.1164/arrd.1980.122.4.567. [DOI] [PubMed] [Google Scholar]
- 3.Mallia P., Johnston S.L. Respiratory viruses: do they protect from or induce asthma? Allergy. 2002;57:1118–1129. doi: 10.1034/j.1398-9995.2002.02169.x. [DOI] [PubMed] [Google Scholar]
- 4.von Mutius E., Illi S., Hirsch T. Frequency of infections and risk of asthma, atopy and airway hyperresponsiveness in children. Eur. Respir. J. 1999;14:4–11. doi: 10.1034/j.1399-3003.1999.14a03.x. [DOI] [PubMed] [Google Scholar]
- 5.Johnston S.L., Pattemore P.K., Sanderson G. Community study of role of viral infections in exacerbations of asthma in 9–11 year old children. BMJ. 1995;310:1225–1229. doi: 10.1136/bmj.310.6989.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pattemore P.K., Johnston S.L., Bardin P.G. Viruses as precipitants of asthma symptoms. I. Epidemiology. Clin. Exp. Allergy. 1992;22:325–326. doi: 10.1111/j.1365-2222.1992.tb03094.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rakes G.P., Arruda E., Ingram J.M. Rhinovirus and respiratory syncytial virus in wheezing children requiring emergency care. IgE and eosinophil analyses. Am. J. Respir. Crit. Care Med. 1999;159:785–789. doi: 10.1164/ajrccm.159.3.9801052. [DOI] [PubMed] [Google Scholar]
- 8.Martinez F.D. Development of wheezing disorders and asthma in preschool children. Pediatrics. 2002;109(Suppl. 2):362–367. [PubMed] [Google Scholar]
- 9.Balfour-Lynn IM, Openshaw P. Viral infection. In: Silverman M (ed.). Childhood Asthma and other Wheezing Disorders. London: Arnold, 2002; pp. 205–221.
- 10.Corne J.M., Holgate S.T. Mechanisms of virus induced exacerbations of asthma. Thorax. 1997;52:380–389. doi: 10.1136/thx.52.4.380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Message S.D., Johnston S.L. Viruses in asthma. Br. Med. Bull. 2002;61:29–43. doi: 10.1093/bmb/61.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Martinez F.D., Wright A.L., Taussig L.M. Asthma and wheezing in the first six years of life. The Group Health Medical Associates. N. Engl. J. Med. 1995;332:129–143. doi: 10.1056/NEJM199501193320301. [DOI] [PubMed] [Google Scholar]
- 13.Stein R.T., Holberg C.J., Morgan W.J. Peak flow variability, methacholine responsiveness and atopy as markers for detecting different wheezing phenotypes in childhood. Thorax. 1997;52:946–952. doi: 10.1136/thx.52.11.946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Silverman M., Wilson N. Wheezing phenotypes in childhood. Thorax. 1997;52:936–937. doi: 10.1136/thx.52.11.936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilson N.M., Lampril J.R., Mak C.W. Symptoms, lung function and β2-adrenoceptor polymorphisms in a birth cohort followed to 10 years. Thorax. 2002;57(Suppl. iii):21. doi: 10.1002/ppul.20049. (Abstract 567) [DOI] [PubMed] [Google Scholar]
- 16.Clarke J.R., Salmon B., Silverman M. Bronchial responsiveness in the neonatal period as a risk factor for wheezing in infancy. Am. J. Respir. Crit. Care Med. 1995;151:1434–1440. doi: 10.1164/ajrccm.151.5.7735597. [DOI] [PubMed] [Google Scholar]
- 17.Turner S, Siew-Kim K, Laing I et al. Association between beta-2 adrenoceptor Arg16Gly, airway responsiveness at one month and lung function in later childhood. Eur Respir J 2001; 18(Suppl. 33): abstract 2S.
- 18.Prendiville A., Green S., Silverman M. Bronchial responsiveness to histamine in wheezy infants. Thorax. 1987;42:92–99. doi: 10.1136/thx.42.2.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Martinez F.D., Graves P.E., Baldini M. Association between genetic polymorphisms of the beta2-adrenoceptor and response to albuterol in children with and without a history of wheezing. J. Clin. Invest. 1997;100:3184–3188. doi: 10.1172/JCI119874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Stein R.T., Sherrill D., Morgan W.J. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet. 1999;354:541–545. doi: 10.1016/S0140-6736(98)10321-5. [DOI] [PubMed] [Google Scholar]
- 21.Doull I.J., Lampe F.C., Smith S. Effect of inhaled corticosteroids on episodes of wheezing associated with viral infection in school age children: randomised double blind placebo controlled trial. BMJ. 1997;315:858–862. doi: 10.1136/bmj.315.7112.858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Christie G.L., Helms P.J., Ross S.J. Outcomes for children of parents with atopic asthma and transient childhood wheezy bronchiltis. Thorax. 1997;52:953–957. doi: 10.1136/thx.52.11.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Stevenson E.C., Turner G., Heaney L.G. Bronchoalveolar lavage findings suggest two different forms of childhood asthma. Clin. Exp. Allergy. 1997;27:1027–1035. doi: 10.1111/j.1365-2222.1997.tb01254.x. [DOI] [PubMed] [Google Scholar]
- 24.Folkerts G., Busse W.W., Nijkamp F.P. Virus-induced airway hyperresponsiveness and asthma. Am. J. Respir. Crit. Care Med. 1998;157:1078–1120. doi: 10.1164/ajrccm.157.6.9707163. [DOI] [PubMed] [Google Scholar]
- 25.Openshaw P.J., Hewitt C. Protective and harmful effects of viral infections in childhood on wheezing disorders and asthma. Am. J. Respir. Crit. Care Med. 2000;162:540–543. doi: 10.1164/ajrccm.162.supplement_1.maic-11. [DOI] [PubMed] [Google Scholar]
- 26.Bisgaard H. A randomised trial of Monteleukast in respiratory syncitial post-bronchiolitis., Am J Respir Crit Care Med 2003; 167: 379–383. [DOI] [PubMed]
- 27.Montalbano M.M., Lemanske R.F. Infections and asthma in children. Curr. Opin. Pediatr. 2002;1:334–337. doi: 10.1097/00008480-200206000-00010. [DOI] [PubMed] [Google Scholar]
- 28.Pullan C.R., Hey E.N. Wheezing asthma and pulmonary dysfunction 10 years after infection with respiratory syncytial virus in infancy. BMJ (Clin. Res. Ed.) 1982;284:1665–1669. doi: 10.1136/bmj.284.6330.1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kneyber M.C.J., Steyerberg E.W., de Groot R. Long-term effects of respiratory syncytial virus (RSV) bronchiolitis in infants and young children: a quantitative review. Acta. Paediatr. 2000;89:654–660. doi: 10.1080/080352500750043945. [DOI] [PubMed] [Google Scholar]
- 30.Sigurs N., Bjarnason R., Sigurbergsson F. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am. J. Respir Crit. Care Med. 2000;161:1501–1507. doi: 10.1164/ajrccm.161.5.9906076. [DOI] [PubMed] [Google Scholar]
- 31.Roman M., Calhoun W.J., Hinton K.L. Respiratory syncytial virus infection in infants is associated with predominant Th-2-like response. Am. J. Respir. Crit. Care Med. 1997;156:190–195. doi: 10.1164/ajrccm.156.1.9611050. [DOI] [PubMed] [Google Scholar]
- 32.Bont L., Kimpen J.L. Immunological mechanisms of severe respiratory syncytial virus bronchiolitis. Intens. Care Med. 2002;28:616–621. doi: 10.1007/s00134-002-1256-z. [DOI] [PubMed] [Google Scholar]
- 33.Hoebee B., Rietveld E., Bont L. Association of severe respiratory syncytial virus bronchiolitis with interleukin-4 and interleukin-4 receptor alpha polymorphisms. J. Infect. Dis. 2003;187:2–11. doi: 10.1086/345859. [DOI] [PubMed] [Google Scholar]
- 34.Corne J.M., Marshall C., Smith S. Frequency, severity, and duration of rhinovirus infections in asthmatic and non-asthmatic individuals: a longitudinal cohort study. Lancet. 2002;359:831–834. doi: 10.1016/S0140-6736(02)07953-9. [DOI] [PubMed] [Google Scholar]
- 35.Mok J.Y., Simpson H. Outcome of acute lower respiratory tract infection in infants: preliminary report of seven-year follow-up study. BMJ (Clin. Res. Ed.) 1982;285:333–337. doi: 10.1136/bmj.285.6338.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Papadopoulos N.G., Psarras S. Rhinoviruses in the pathogenesis of asthma. Curr. Allergy Asthma Rep. 2003;3:137–145. doi: 10.1007/s11882-003-0026-5. [DOI] [PubMed] [Google Scholar]
- 37.Kimpen J.L. Viral infections and childhood asthma. Am. J. Respir. Crit. Care Med. 2000;162:S108–S112. doi: 10.1164/ajrccm.162.supplement_2.ras-11. [DOI] [PubMed] [Google Scholar]
- 38.Mckean M.C., Leech M., Lambert P.C. A model of viral wheeze in nonasthmatic adults: symptoms and physiology. Eur. Respir. J. 2001;18:23–32. doi: 10.1183/09031936.01.00073101. [DOI] [PubMed] [Google Scholar]
- 39.Illi S., von Mutius E., Lau S. Early childhood infectious diseases and the development of asthma up to school age: a birth cohort study. BMJ. 2001;322:390–395. doi: 10.1136/bmj.322.7283.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Martinez F.D., Stern D.A., Wright A.L. Differential immune responses to acute lower respiratory illness in early life and subsequent development of persistent wheezing and asthma. J. Allergy Clin. Immunol. 1998;102:915–920. doi: 10.1016/s0091-6749(98)70328-8. [DOI] [PubMed] [Google Scholar]
- 41.Castro-Rodriguez J.A., Holberg C.J., Wright A.L. A clinical index to define risk of asthma in young children with recurrent wheezing. Am. J. Respir. Crit. Care Med. 2000;162:1403–1406. doi: 10.1164/ajrccm.162.4.9912111. [DOI] [PubMed] [Google Scholar]
- 42.Welliver R.C., Ogra P.L. RSV, IgE, and wheezing. J. Pediatr. 2001;139:903–905. doi: 10.1067/mpd.2001.119006. [DOI] [PubMed] [Google Scholar]
- 43.Hogg J.C. Role of latent viral infections in chronic obstructive pulmonary disease and asthma. Am. J. Respir. Crit. Care Med. 2001;164:S71–S75. doi: 10.1164/ajrccm.164.supplement_2.2106063. [DOI] [PubMed] [Google Scholar]
- 44.Macek V., Sorli J., Kopriva S. Persistent adenoviral infection and chronic airway obstruction in children. Am. J. Respir. Crit. Care Med. 1994;150:7–10. doi: 10.1164/ajrccm.150.1.8025775. [DOI] [PubMed] [Google Scholar]
- 45.Silverman M. Wheezing disorders in infants and young children. In: Silverman M (ed.). Childhood Asthma and Other Wheezing Disorders. London: Arnold, 2002; pp. 307–332.
- 46.McKean M, Ducharme F. Inhaled Steroids for Episodic Viral Wheeze of Childhood. Cochrane Database System Reviews 2000. [DOI] [PMC free article] [PubMed]


