Abstract
Since their introduction as pets several decades ago, ferrets have become an increasingly popular household pet. Great strides have been made in improving their diet and understanding common diseases (eg, insulinoma, hyperadrenocorticism, lymphoma) that affect them. With the frequency with which these conditions are seen, it sometimes is easy to forget that ferrets can be affected by other diseases. Some of these diseases, such as cryptococcosis, are known, but may be increasing in incidence and range, whereas others, such as hypothyroidism and pure red cell aplasia, may be underrecognized or underreported. This review highlights new and emerging diseases not already well reviewed in the literature.
Keywords: Ferrets, Cryptococcosis, Hypothyroidism, Influenza, Pure red cell aplasia
Key points
-
•
Cryptococcosis has been described in 17 ferrets worldwide, with 12 of these cases being reported since 2002, and has only recently been reported in the United States and Europe.
-
•
Influenza is both a zoonotic and anthroponotic disease; therefore, recognition of influenza infection in ferrets by veterinarians can aid in increased surveillance in humans, as was seen in the 2009 pandemic H1N1.
-
•
Although treatment of hypothyroidism in ferrets is simple and similar to that for dogs and cats, the clinical signs are ambiguous and include obesity, lethargy, decreased activity, excessive sleeping, and in some ferrets hind-end weakness.
-
•
Pure red cell aplasia (PRCA) is a nonregenerative anemia that should be considered when presented with an anemic ferret. Prompt and aggressive therapy with long-term immunosuppressive medications and blood transfusions as needed are vital in the successful treatment of this condition.
Several emerging diseases have been recently reviewed, with little to no new information arising since the publication of these comprehensive reviews. The reader is encouraged to seek the following resources for additional information on those emerging conditions in previous editions of Veterinary Clinics of North America—Exotic Animal Practice:
-
•Disseminated idiopathic myofasciitis:
- Ramsell KD, Garner MM. Disseminated idiopathic myofasciitis in ferrets. Vet Clin North Am Exot Anim Pract 2010;13(3):561–75.
-
•Mycobacteriosis:
- Pollock C. Mycobacterial infection in the ferret. Vet Clin North Am Exot Anim Pract 2012;15(1):121–9.
-
•Systemic coronavirus:
- Murray J, Kiupel M, Maes RK. Ferret coronavirus-associated diseases. Vet Clin North Am Exot Anim Pract 2010;13(3):543–60.
Cryptococcosis
Etiology and Epidemiology
Cryptococcus spp are capsulated, dimorphic, basidiomycetous fungi that are found in soil, air, and trees.1 Although there are 39 recognized species, only a few species are found to cause disease in humans and animals.2 The most medically relevant species are Cryptococcus neoformans and Cryptococcus gattii. C neoformans is split into 2 distinct variants: var. neoformans (previously called serotype D) and var. grubii (previously called serotype A).3 This species is classically identified as causing meningitis and meningoencephalitis in immunosuppressed people, has a worldwide distribution, and is found in pigeon droppings.1 C gattii (previously called Cryptococcus neoformans var. gattii) is also synonymously called Cryptococcus bacillisporus in some texts. It is endemic in tropical and subtropical climates, although it is being found in more areas, and is an emerging pathogen in immunocompetent humans in North America.2, 4 Additional species that can cause disease in immunosuppressed humans include Cryptococcus albidus and Cryptococcus laurentii, but to date these species have not been documented to cause disease in ferrets.
This fungus has a special life cycle that involves a yeast form and a filamentous form. The most medically relevant form is the basiospore, which is the infectious form in mammals. The natural route of infection is via inhalation of the basiospores or actual yeast cells. In the host, the yeast appears as round-ovoid structures with a polysaccharide capsule.1 This thick capsule helps prevent desiccation1 and, in the correct environment, allows the organism to stay viable for up to 2 years.2 In addition, this capsule in C neoformans also functions as its major virulence factor because it helps to evade the host’s immune system.2, 5 Because entry is via inhalation, the nasal cavity and lungs can be infected first, followed by hematogenous spread to the central nervous system (CNS).2
Cryptococcosis in ferrets
Since it was first reported in the literature in the United Kingdom in 1954,6 there have been 16 reported cases of cryptococcosis in ferrets. Before 2000 there had been 4 reported cases: 1 in the United Kingdom, 1 in Australia, and 2 in North America. Since 2000 there have been several cases in Australia, 3 in North America, and 1 in Spain.
As with cryptococcosis in humans, cats, and dogs,2, 5, 7 the 2 most common species reported in ferrets is C neoformans and C gattii (Table 1 ). Of the 13 cases that reported the infectious species, 6 ferrets were infected with C neoformans 6, 7, 8, 9, 10, 11 and 6 were infected with C gatti.7, 12, 13, 14 In the 3 cases that the variant was listed for C neoformans, it was always var. grubii.7, 10, 11 The infectious species was not explicitly listed in 2 ferrets15; they were included in a report of a 2003 outbreak of Cryptococcus in Canada, where other animals and humans were infected with both C neoformans and C gattii (see Table 1).
Table 1.
Cryptococcal species and variant, country of infection, and patient signalment in 16 ferrets with cryptococcosis
| Signalment | Cryptococcus Species and Variant | Country | Ref. | |
|---|---|---|---|---|
| 1. | Mature M | C neoformans | UK | 6 |
| 2. | 3-y-old MI | C neoformans | Australia | 8 |
| 3. | 6-y-old FS | Not stated | USA | 16 |
| 4. | 3-y-old M | C neoformans | USA (NY) | 9 |
| 5. | 5-y-old MC | C gattiia | Australia | 7, 12 |
| 6. | 6-y-old MC | C gattiia | Australia | 7 |
| 7. | 2-y-old MC | C gattiia | Australia | 7 |
| 8. | 2–3-y-old FS | C neoformans var. grubii | Australia | 7 |
| 9. | 3.5-y-old MC | Not stated | Australia | 7 |
| 10. | 1.75-y-old MC | C gattii VGIIb | Canada | 7 |
| 11. | 3-y-old MI | C gattii VGIIb | Canada | 7 |
| 12. | Not stated | Not stated | Canada | 15 |
| 13. | Not stated | Not stated | Canada | 15 |
| 14. | 1.5-y-old MC | C neoformans var. grubii | USA | 10 |
| 15. | 4-y-old MC | C neoformans var. grubii | USA | 11 |
| 16. | 17-mo-old MC | C gattii serotype B VGI/AFLP4 | Spain | 13, 14 |
Abbreviations: FS, female spayed; M, male; MC, male castrated; MI, male intact.
Called C bacillisporus in publication, which is synonymous with C gattii.
Although the original publication (Malik, 2002) states both cases from Canada as C neoformans var. grubii, a more recent publication15 and personal communication with Malik indicate that these cases were incorrectly speciated and actually had C gattii VGII, not C neoformans var. grubii. This table reflects the most updated information.
Age and sex
Of the 16 reported cases of cryptococcosis, the sex was reported in 14 cases. There were more cases reported in male ferrets: 12 males6, 7, 9, 10, 11, 12, 13, 14, 16 and only 2 females.7, 16 In addition, there are more cases reported in spayed/castrated ferrets7, 10, 11, 12, 13, 14, 16 than in intact ferrets.6, 7, 8 The ages of these ferrets were reported in 13 cases and ranged from 17 months to 6 years.6, 7, 8, 9, 10, 11, 13, 14, 16
Risk factors
In humans, it has been well documented that immunosuppression is a risk factor for the development of cryptococcosis associated with C neoformans.5 In cats, however, underlying immunosuppressive disease is not commonly found.2 In the reported cases of ferrets there was one with concurrent lymphosarcoma,16 potentially causing immunosuppression. Three ferrets were also treated with systemic steroids that may have caused an altered immune status. In one case, dexamethasone SP was used to manage hind-end paresis.9 When the ferret was euthanized 5 weeks later, it had systemic cryptococcosis. In a case of chorioretinitis, the ferret was treated with oral prednisone.13, 14 This ferret was also treated with fluconazole at the same time, but when he was euthanized 7 months after treatment he had signs of systemic cryptococcosis.
Because Cryptococcus is found in soil, bird droppings, and trees, ferrets with outdoor access are at additional risk. Reported exposures include living outdoors in a hutch,6, 8 contact with pigeons and finches,8 digging outside,7 leash walks outside,15 and outdoor play areas.13, 14 The incubation period can range from 2 to 13 months, so previous travel to an endemic area is another potential risk factor.2
There are 3 cases of ferrets with cryptococcosis living with other ferrets or humans that had the same species of Cryptococcus, either as asymptomatic carriers or with disease themselves. In Australia, two ferrets were infected with the same species of Cryptococcus: one with a nasal infection and the other with a retropharyngeal infection.7, 12 A ferret in Spain that died of disseminated cryptococcosis lived with ferrets and humans who were asymptomatic carriers of the same strain of Cryptococcus (based on nasal swabs).13 It is likely that the ferrets and humans in the household were exposed to the same source of Cryptococcus. Cryptococcus is not a zoonotic disease because the organism does not aerosolize from infected tissue, and therefore does not spread between humans and animals.2
Clinical Findings
Presenting complaints and physical examination findings for ferrets with cryptococcosis vary depending on the organ system affected. In cats, upper respiratory tract signs are most common, whereas in dogs disseminated disease affecting the CNS, eyes, urinary system, and nasal cavity are common. Clinical signs similar those found in cats and dogs and reported in ferrets include respiratory,7, 11, 12, 15 ocular,13, 14 neurologic,9 musculoskeletal,7 lymphatic,7, 10, 13, 14 and gastrointestinal signs10, 11 as well as acute death (Table 2 ).6, 8
Table 2.
Clinical signs and physical examination findings by system in 16 ferrets with cryptococcosis
| System | Clinical Signs and Physical Examination Findings |
|---|---|
| Musculoskeletal | Swollen limbs or digits7 |
| Respiratory | Nasal discharge, coughing, sneezing, dyspnea, swelling over nasal bridge7, 11, 12, 15 |
| Ocular | Chorioretinitis, acute bilateral blindness13, 14 |
| Gastrointestinal | Regurgitation, retching, gagging7, 10, 11 |
| Neurologic | Posterior paresis9 |
| Lymphadenopathy | Popliteal, cervical, submandibular7, 10, 13, 14 |
Diagnostic Testing
Clinical laboratory
As with other species, changes in the complete blood count (CBC) and chemistry panel in ferrets with cryptococcosis is generally nonspecific but is an important first step toward finding underlying disease.2, 11 In dogs and cats a mild to moderate regenerative anemia, leukocytosis with a monocytosis, and eosinophilia can be seen, as well as changes in the chemistry panel reflecting specific organs infected.2 In the literature only 3 ferrets had CBCs and serum biochemistries performed. Reported abnormalities have included monocytosis,10 lymphopenia,11 lactic acidosis,11 hypoglycemia,11 hypocalcemia,10, 11 hypophosphatemia,10 hypoalbuminemia,10, 11 and hyperglobulinemia,11 although in one ferret the CBC and serum biochemistry was within the normal range.13, 14 This ferret had an additional protein electrophoresis run, which indicated an elevation in the α- and β-globulins.13
Diagnostic imaging
Depending on the organ system affected, radiographs, ultrasonography, and/or magnetic resonance imaging (MRI) can be helpful diagnostic tools. A pulmonary bronchointerstitial pattern,11 diffuse alveolar disease,7 and/or pleural effusion7 were noted on thoracic radiographs in ferrets that presented for coughing and dyspnea. Cryptococcal infections were found in the lungs at necropsy.7, 11 A cylindrical intra-abdominal mass was noted on abdominal radiographs in one ferret that was found to have an infected jejunum and mesenteric lymph node on necropsy.7 Another ferret had an abdominal ultrasonogram indicating mesenteric lymphadenopathy, which was found to be an infected portal lymph node on biopsy.11 One ferret that presented for acute blindness developed neurologic signs of incontinence and pain 7 months into the treatment regimen and had MRI performed. This animal had a mass compressing the thoracic region of the spinal cord and had cryptococcal infection in the spinal cord on necropsy.13, 14 When indicated by clinical signs or physical examination findings, diagnostic imaging was helpful in pointing to a diagnosis of cryptococcosis.
Serologic and cerebrospinal fluid testing
The latex cryptococcal antigen agglutination test (LCAT) detects the polysaccharide capsular antigen, and can be used on cerebrospinal fluid (CSF) and serum.17 This test detects all known cryptococcal serotypes.2 In humans, commercial kits have high sensitivity and specificity, but false-negative results can occur when disease is localized.2 Antigen was detected in the CSF of one ferret that had chorioretinitis, but it could not be titrated.13 Serum LCAT was positive in 4 ferrets in which the test was used,7, 10, 12, 13 and this was used to monitor response to treatment in cryptococcal infections of the left forelimb7 and mandibular lymph nodes.10 Because titer results from different LCAT kits can vary, it is important to use the same kit when monitoring response to treatment.2
Cytology
Cytology of affected tissue can lead to a quick presumptive diagnosis of Cryptococcus. Diff Quick, Giemsa, Wright, new methylene blue, and Gram stain can all be used, and visualization at low power is usually the best. The presence of spherical capsulate yeast on aspirates or impression smears of enlarged lymph nodes, masses, or draining tracts has been seen in several ferrets.7, 10, 12, 13 In addition to the presence of the yeast, pyogranulomatous inflammation can also be seen in animals with an intact immune system.
Biopsy
If possible, collection of a piece of affected tissue for biopsy is the most important diagnostic step. If tissue is collected, impression smears should be made for cytologic analysis, part of the sample should be used for culture, and the rest should be submitted for histopathology.2 In the reported ferret cases, identification of the yeast was made on the biopsy of an enlarged lymph node in several cases,7, 11, 13, 14 as well as a biopsy of an infected digit7 and a nasal biopsy.12
Immunohistochemistry
If tissue is not available for fungal culture and routine histopathology is unable to confirm the presence of cryptococci, immunohistochemistry staining of tissue is available.2, 18 Caution should be exercised if this is the sole method being used to determine the infective species. Previous immunohistochemical testing methods performed in ferrets have been reported to be inaccurate, leading to an improper identification of the cryptococcal species.11
Fungal culture
Culturing for Cryptococcus is important because it can lead to correct identification of the infectious species and variant. Culture also has the benefit of a sensitivity spectrum, which can aid in choosing an appropriate antimicrobial agent.12, 13 Cryptococcus is cultured on a medium of Sabouraud dextrose agar.2 Cultures have been performed on aspirates of enlarged lymph nodes10, 13 and a nasal swelling in ferrets.12 Therefore, if Cryptococcus is suspected and aspirates or tissue is obtained for cytology or biopsy, a sample should also be submitted for culture.
Polymerase chain reaction
More recently, DNA-based methods have been used to correctly identify the cryptococcal species and variant. DNA polymerase chain reaction (PCR) testing has been used to confirm the diagnosis and identify the infective species and variant in 2 ferrets. PCR extraction was successful on infected lung tissue via necropsy11 and a lymph node13, 14 collected via biopsy. In addition, PCR confirmed identical strains in 2 ferrets with cryptococcosis from the same household.7
Pathology
Granulomatous or pyogranulomatous inflammation is typically seen in animals with cryptococcosis that have intact immune systems. In cats, the nose, lungs, spinal cord, and eyes are often affected, whereas dogs commonly have disseminated disease and/or infection in the CNS and eyes.2 Necropsy results were available for 8 ferrets. Four of these animals had evidence of disseminated disease.6, 7, 11, 13, 14 Six ferrets had infected lymph nodes6, 7, 11, 16 and, as expected owing to its entry via inhalation, 5 ferrets had evidence of disease in their lungs.7, 8, 11, 13 Cryptococcus has a predilection for the CNS, and infection was noted in 3 ferrets.9, 11, 13, 14 Two ferrets each had disease in their liver6, 7 and spleen,6, 11 whereas 1 ferret each had signs of disease in the intestine,7 thymus,13 eyes,13 mediastinum,7 and peritoneum.6
Treatment
Managing immunosuppression
Identification and treatment of any underlying disease causing immunosuppression is important in managing cryptococcosis. Successful treatment is limited if the cause of immunosuppression cannot be controlled.5 In humans, cancer and high-dose corticosteroid treatment have been linked with cryptococcosis.5 Concurrent neoplasia may have played a role in the one ferret that was reported to have both cryptococcosis and lymphosarcoma.9
Surgical debulking
If possible, surgical debulking or removal of the affected area can be an effective treatment. This approach was used successfully in one ferret with a digit infection7 and in one with a nasal infection.12
Antifungal agents
In humans with cryptococcosis, most initial isolates are susceptible to the major classes of antifungal agents including amphotericin B, flucytosine, and the azoles.5 Different antifungal medications are warranted depending on the severity of disease and the organ system affected. A variety of antifungal agents, doses, and frequencies have been used to treat cryptococcosis in ferrets, with variable success (Table 3 ).
Table 3.
Treatments and outcomes in 16 ferrets with cryptococcosis
| Treatment | Route, Dose, Interval | Duration | Outcome | Ref. | |
|---|---|---|---|---|---|
| 1. | None | Presented deceased | 6 | ||
| 2. | None | Presented deceased | 8 | ||
| 3. | None | <3 mo died of lymphosarcoma | 16 | ||
| 4. | Dexamethasone SP | 0.15–0.5 mg/kg q 8–48 h | Tapered over 30 d | 4 wk PT: improved CS 5 wk PT: euthanized because of cardiac disease; post mortem indicated CNS cryptococcosis |
9 |
| 5. | Surgical debulking | 171 d PT: nasal disease resolved 3 y PT: euthanized because of abdominal mass; post mortem showed no signs of cryptococcosis |
7, 12 | ||
| Itraconazole | 25 mg PO every 24 h | 21 d | |||
| 33 mg PO every 24 h | 21–171 d | ||||
| 25 mg PO every 24 h | 171 d to 3 y | ||||
| 6. | Itraconazole | 33 mg PO every 24 h | Unknown | Initial improvement Euthanized Post mortem: cryptococcus in enlarged and contralateral LN |
7 |
| 7. | None | Died after sedation | 7 | ||
| 8. | None | Died during induction | 7 | ||
| 9. | Itraconazole | 25 mg/kg PO every 24 h | 6 mo | 5 wk PT improved 6 mo PT antigen titer 0 |
7 |
| 10. | Surgical excision affected digit | 4 mo PT no disease recurrence | 7 | ||
| 11. | Fluconazole | 12 mg PO every 12 h | ≥4 mo | 4 mo PT doing well but dragging limb | 7 |
| 12. | None | Not reported | 15 | ||
| 13. | None | Not reported | 15 | ||
| 14. | Itraconazole | 15 mg/kg PO every 24 h | 43 wk | 3 wk PT decreased size of LN, stopped regurgitating 6 wk PT normal sized LN 40 wk PT antigen titer 0 |
10 |
| 15. | Itraconazole | 10 mg/kg PO every 12 h | 4 d | Died 4 d PT Post mortem showed disseminated cryptococcosis |
11 |
| Amphotericin B | 0.25 mg/kg IV every 24 h | 4 d | |||
| 16. | Fluconazole | 10 mg/kg PO every 24 h | 6 mo | 4 mo PT clinical signs improved but antigen titer unchanged 7 mo PT euthanized because of CNS signs Post mortem showed disseminated cryptococcosis |
13, 14 |
| Prednisone | 0.5 mg/kg PO every 24 h | 6 mo | |||
| Diclofenac | OU every 4–8 h | 5.5 mo |
Abbreviations: CNS, central nervous system; CS, clinical signs; IV, intravenously; LN, lymph nodes; OU, in both eyes; PO, by mouth; PT, post treatment.
Amphotericin B (AMB) acts by binding to sterols (primarily ergosterol) in the cell membrane of fungal organisms, and alters the permeability of this membrane.19 In humans AMB has cured countless cases of disseminated cryptococcosis that would have been fatal before the advent of its use.5 This medication can be expensive, must be administered parenterally, and can cause nephrotoxicity.19 Newer, lipid-based products are less nephrotoxic and penetrate better into tissues.19 AMB in combination with itraconazole has been used in one ferret with disseminated cryptococcosis, but the ferret succumbed to disease 4 days after initiating treatment.11
Flucytosine inhibits protein, RNA, and DNA synthesis in fungal cells, and is useful against strains of Cryptococcus and Candida.20 This medication reaches high levels in the CNS, but there is a high incidence of resistance, so it must be used with another antifungal agent such as AMB.5 This combination is used in humans,5 dogs, and cats.2 Side effects of this medication can include bone marrow suppression and gastrointestinal (GI) disease in humans and animals.2, 20 There are no reports on the use of flucytosine in the treatment of cryptococcosis in ferrets.
The azole antifungal agents include both imidazoles (ketoconazole, miconazole) and triazoles (fluconazole and itraconazole). These agents work by altering the cellular membranes of susceptible fungi.21 Azoles are fungistatic and rely on an intact immune system to remove the infectious agent, so they may not be effective as a single modality in immunosuppressed patients.2 In human cases of cryptococcal meningitis, ketoconazole was found to be ineffective, and miconazole is no longer used owing to its toxicity.5 There are no reports of the use of ketoconazole or miconazole in the treatment of cryptococcosis in ferrets.
Itraconazole is effective against Cryptococcus, but has poor penetration into the CSF and the aqueous humor.21 However, it has been used successfully in the management of meningeal and ocular cryptococcosis in humans, dogs, and cats, potentially because of the compromised blood-brain or blood-aqueous barriers during infection.2, 5 Itraconazole has good bioavailability if administered with food, and in most species has the additional benefit of once-daily dosing.21 Side effects seem to be dose related, and include hepatotoxicity, GI disturbances, and vasculitis.21 Fluconazole is effective against Cryptococcus and has good penetration into the CSF, eye, and peritoneal fluid.22 It is has a high bioavailability and rapid absorption when administered orally, and has minimal side effects that can include anorexia and, rarely, hepatotoxicity.22 However, resistance to fluconazole has recently been reported in C gattii in North America.2 Both itraconazole and fluconazole have been used singly in cats with mild to moderate cryptococcosis (without CNS involvement) and in dogs with localized disease. Combination use with AMB is still recommended for disseminated disease or CNS involvement.2 Itraconazole has been used in the treatment of 4 ferrets with localized disease7, 10, 12 and 1 with disseminated systemic disease.11 Fluconazole has been used in the treatment of 1 ferret with ocular cryptococcosis13, 14 and 1 that had localized disease to the hind limb (see Table 3).7
Monitoring
Definitive treatment of cryptococcosis can be difficult to determine. It is not uncommon for animals and humans to remain on long-term antifungals, sometimes for the lifetime of the patient.5 Malik, who has written numerous articles about cryptococcosis in veterinary patients (including ferrets), has seen recurrence 10 years after successful therapy, and therefore recommends antifungal administration until cryptococcal antigen titers reach zero.2 This monitoring plan was used in the successful treatment of a 1.5-year-old ferret that was treated with itraconazole for a total of 43 weeks until his LCAT reached zero.10 Malik additionally recommends monitoring titers every 3 to 6 months after the titer reaches zero, to catch recurrence early.
If patients are on potentially nephrotoxic (ie, AMB) or hepatotoxic (ie, itraconazole) antifungal agents, routine monitoring of kidney and/or liver values is recommended.
Prognosis
In humans, prognosis depends on the ability to control the patient’s underlying disease.5 Similarly, in cats, feline leukemia virus (FeLV)-positive status is associated with a poor long-term prognosis.2 In cats there also seems to be a negative outcome with CNS disease, whereas in dogs the outcome may depend on where they live.2 Because of the paucity of reported cases of cryptococcosis in ferrets, risk factors associated with prognosis cannot be assessed, but those ferrets with localized disease have had a more favorable outcome than those with disseminated disease.
Of the 16 reported cases of ferrets with cryptococcosis, 2 presented deceased,6, 8 2 did not have the outcome reported,15 2 died before treatments could be administered,7 and 1 died of lymphosarcoma with Cryptococcus as an incidental finding on postmortem examination.16 In addition, 1 ferret was treated with dexamethasone for neurologic disease, only for cryptococcosis to be found on postmortem examination.9
A total of 8 ferrets received treatment for cryptococcosis. One ferret with localized disease was treated successfully with only surgical excision of the affected digit.7 Another ferret with localized nasal cryptococcosis was successfully treated with surgical excision and oral itraconazole for 3 years postoperatively.12 When he died 3 years after his initial diagnosis, he had no signs of cryptococcosis.
Three ferrets with localized disease were successfully treated with medical management alone. One ferret with localized infection of the forelimb was treated with oral itraconazole and showed improvement 5 weeks into treatment. He was treated for a total of 6 months, at which time LCAT was negative.7 One ferret with localized disease of the hind leg was treated with oral fluconazole for 4 months until he was systemically well but still dragging his hind leg.7 The final ferret with localized disease to a submandibular lymph node was treated with oral itraconazole, and showed clinical improvement after 3 weeks. He was treated for a total of 43 weeks and his LCAT titer was negative after 40 weeks.10
Three ferrets with cryptococcosis were treated medically but died or were euthanized because of their disease. One ferret with cryptococcosis of the right retropharyngeal lymph node was treated with oral itraconazole, and although he improved initially he was euthanized because of skin disease. On postmortem examination this animal had evidence of Cryptococcus in both retropharyngeal lymph nodes.7 A ferret that presented for respiratory disease was treated with intravenous AMB and oral itraconazole for 4 days before he died. Postmortem examination revealed disseminated cryptococcosis.11 One ferret with cryptococcal associated chorioretinitis was treated with oral fluconazole and prednisone. This animal showed some initial improvement in his overall health, but remained blind and was euthanized when he developed signs of CNS disease 7 months after treatment. On postmortem examination the ferret had disseminated cryptococcosis.13, 14
Influenza
Etiology and Epidemiology
Influenza is a contagious respiratory illness caused by influenza viruses.23 These RNA viruses are in the Orthomyxoviridae family and are classified into 3 species termed A, B, and C, with species A being the most medically relevant to ferrets.24 Influenza type A viruses are further divided into subtypes, based on 2 proteins that exist on the surface of the virus: hemagglutinin (HA) and neuraminidase (NA). There are 17 known HA subtypes and 10 known NA subtypes. Many different combinations of HA and NA proteins are possible.25 For the purposes of this article, the focus is on the H1N1 subtype during the influenza season 2009 to 2010. This virus was also called “novel H1N1” or “swine flu,” which emerged in the spring of 2009 and caused the first global outbreak of disease caused by a novel influenza virus (influenza pandemic) in more than 40 years.26 H1N1 was first seen in humans in March 2009 and then became a pandemic by June 2009. As of April 2011, this virus has caused natural disease in at least 10 animal species.27
Geographic distribution and age
During 2009 to 2010 there were several reports of influenza in ferrets. There were 2 reports of naturally occurring influenza in pet ferrets in Pennsylvania,28 Nebraska, and Oregon29 and one report in a colony of ferrets30 in Iowa. In addition, there was one black-footed ferret with influenza in a California zoologic setting.27 During this pandemic, the youngest ferrets affected were 1 year of age while the oldest was 8 years.29
Zoonosis and anthroponosis
Influenza is both a zoonotic and anthroponotic disease. In the 1930s the zoonotic spread of influenza from ferrets was first reported.31 More recently, ferrets have been instrumental in influenza research and as sentinels for this viral infection in humans. Ferrets are used as animal models for influenza infection because they display flulike symptoms and immune responses similar to those of humans.32 In Pennsylvania in 2009, the diagnosis of influenza in several pet ferrets led to enhanced surveillance for pandemic H1N1 in the region. This vigilance helped to increase the identification of humans infected with this same strain.28 Experimentally, reassortment of different strains of influenza in ferrets has been proved via coinfection with both a human and avian strain of the virus. Therefore, a ferret exposed to both a pathogenic and seasonal strain of influenza could act as mediator for the formation of a pathogenic form of the seasonal influenza.33
Anthroponosis was suspected in several cases of influenza in ferrets in 2009. In a group of ferrets with pandemic H1N1, the owners reported flulike symptoms from 6 to 9 days before clinical signs in the ferrets.29 In a California zoo 3 animals contracted influenza A H1N1, including a black-footed ferret. It was suspected but not confirmed that the virus was contracted from humans interacting with the animals.28
Risk factors
Influenza infection in older people, young children, and those with immunosuppressive conditions are at high risk for serious flu complications,24 and this may also be true for ferrets because in a report of influenza in 13 ferrets, the 1 ferret that died during the outbreak was 8 years old.29
Exposure to swine and birds has been associated with influenza infections in humans. Influenza viruses that normally circulate in pigs are called variant viruses when they are found in people. In humans, certain influenza strains have been associated with prolonged exposure to pigs at agricultural fairs.34 Avian influenza occurs naturally among anseriformes and domestic poultry, but can infect other avian and animal species, including sporadic human infections.35 During an outbreak of influenza in a colony of ferrets living on a farm in Iowa, investigation of their housing revealed that they were 0.25 miles away from a swine operation and were exposed to multiple avian species. There was concern for aerosol transmission from the swine farm or locally housed birds.30
Clinical Presentation
In humans and animals, influenza is characterized by respiratory signs and a fever. In the naturally occurring cases of influenza in 2009 to 2010, the most common presenting complaints were coughing, sneezing, oculonasal discharge, and lethargy.27, 28, 29, 30 The physical examination findings included dyspnea, oculonasal discharge, and a fever between 104° and 105°F (40°–40.6°C).28, 29
Ferrets are used frequently in a research setting for influenza investigation. Therefore, a more complete picture of the clinical disease has been described in the laboratory setting. In one study that compared the clinical signs and recovery period of ferrets experimentally infected with the pandemic H1N1 influenza versus other strains of this virus, they found that weight loss could be found as early as the first day of infection.24 In comparison with other strains, ferrets infected with the pandemic H1N1 had the longest period of weight loss, the longest duration of fever, the most profound lethargy, and the most prominent sneezing and nasal discharge. The more intense clinical signs were thought to be due to the profound cytokine production and broader tissue tropism of this pandemic strain.24
Diagnostic Testing
In ferrets with suspected influenza, the initial database included a CBC, serum biochemistry, and diagnostic imaging. Of the ferrets reported during the 2009 to 2010 pandemic, CBC changes included anemia28 and biochemistry changes included an increased alanine aminotransferase, increased alkaline phosphatase, and a low total protein, potassium, and chloride.28 Thoracic radiographs were only performed in a few ferrets, and were mostly unremarkable.28
Influenza is both an anthroponotic and zoonotic disease. Therefore, identification of the influenza strain is very important. Suspected cases should have samples collected for confirmation and speciation via PCR and/or immunohistochemical staining. Samples for real-time reverse transcriptase (rRT)-PCR submission include nasal, pharyngeal, and conjunctival swabs, bronchoalveolar lavage fluid, and swabs from the lungs on postmortem specimens. Depending on the ferret’s location, samples may be submitted to the state animal health laboratory. During the pandemic, rRT-PCR samples were helpful in identifying the influenza species in pet, farm, and zoologic ferrets.27, 28, 30 Because this is a noninvasive test, it can easily be used on humans and other ferrets in contact with the ill ferret.
If postmortem samples are available, immunohistochemical (IHC) staining can assist in species identification. IHC was particularly helpful in the outbreak of naturally occurring influenza in a farm setting where 8% of the ferrets became clinically ill with influenza.30 Postmortem samples showed bronchointerstitial pneumonia with necrosuppurative bronchiolitis, and through IHC the investigators were able to identify the influenza as the pandemic H1N1 strain that was of swine lineage in origin.30
Additional diagnostic testing includes serum collection for antibody detection. In ferrets that were experimentally infected with H1N1, antibody responses correlated with the severity of the influenza infections.24 During the outbreak of natural influenza in a colony of farm ferrets, hemagglutination inhibition antibody was high in infected ferrets.30
Treatment and Monitoring
The basis of influenza treatment involves supportive care and prevention of secondary bacterial infection. Supportive care includes fluid therapy, either subcutaneously or intravenously, if severely dehydrated or febrile. Weight loss was a common clinical sign noted in ferrets experimentally infected with influenza, so support feeding is also important.24 Ferrets are strict carnivores with a rapid GI transit time, so a convalescent diet should include a meal that is palatable, and high in fat and protein. Many recovery diets for feline patients are suitable, such as Hill’s A/D, Oxbow’s Carnivore Care, or Lafeber’s Emeraid Carnivore. Coverage with a broad-spectrum antibiotic is warranted if a secondary bacterial infection is suspected. Parenteral antibiotics can be used in ferrets that are too sick or nauseated for oral antibiotics. The use of antiviral drugs in the treatment of influenza is difficult, because this virus mutates and resistance is common.36 There are variable results in the use of antiviral drugs in experimentally induced influenza in ferrets, which seem to depend on the strain of the virus and the time of administration after infection.36, 37 In the ferrets with pandemic H1N1 influenza, many were successfully treated with subcutaneous fluids, parenteral antibiotics, and support feeding.27, 29, 30 It must be noted that influenza is zoonotic and contagious to other ferrets, so appropriate precautions must be taken for all staff and owners involved in treating the ill ferret.
In most cases, monitoring is straightforward during the treatment period. Response to initial supportive care including monitoring of weight gain, hydration status, and temperature are important. In the case of the black-footed ferret with H1N1, he was in a zoologic park setting with exposure to humans. Therefore, his influenza status was monitored via PCR testing. When his clinical signs resolved after treatment with parenteral antibiotics, his influenza PCR was negative.27
Prognosis
Depending on the viral strain and concurrent disease, the prognosis for ferrets with influenza is favorable. The ferrets experimentally infected with pandemic H1N1 had severe illness, but the strains were cleared from the ferrets within 7 days after infection. This same clearance was noted in other strains of the more common seasonal influenza strains.24 During an outbreak in Nebraska and Oregon, only 1 ferret died and 12 others recovered with supportive care. The ferret that died was 8 years old and may have had concurrent disease.29 In an influenza outbreak in a colony of 1000 ferrets, there was a morbidity rate of 8% and a mortality rate of 0.6%.30
Hypothyroidism
Although one of the most common endocrinopathies seen in dogs, hypothyroidism has only recently been described in 7 ferrets.38 Impaired production or secretion by the thyroid results in a deficiency of circulating thyroid hormone and associated clinical signs.
Etiology and Pathogenesis
At present, the pathogenesis of hypothyroidism in ferrets is not known. In dogs, primary hypothyroidism is most common, with 2 abnormalities observed that affect the thyroid gland with approximately equal occurrence: lymphocytic thyroiditis and idiopathic thyroid atrophy. Lymphocytic thyroiditis involves infiltration and destruction of the glandular parenchyma by lymphocytes, plasma cells, and macrophages. This condition is believed to be immune mediated, but the exact pathogenesis has not been elucidated. The presence of antithyroglobulin varies between breeds, and whether it occurs before or as a result of follicular damage is unknown. Idiopathic thyroid atrophy is characterized by the loss of the thyroid parenchyma and replacement with adipose connective tissue, with no observable inflammation or fibrosis. Primary and metastatic neoplasia can uncommonly also cause hypothyroidism.39 In cats, acquired hypothyroidism is almost always the result of treatment for hyperthyroidism. Only one published case of feline hypothyroidism currently exists, which was confirmed to be lymphocytic thyroiditis using histopathology.40
Clinical Signs
Clinical signs are often vague, but the most consistent ones are obesity, lethargy, decreased activity, and excessive sleeping. Rear-leg weakness has also been noted in some ferrets. Common clinical signs noted in dogs (eg, hair loss, hypercholesterolemia) were not consistently noted in the 7 ferrets diagnosed with hypothyroidism. Hypoglycemia was also not noted unless associated with a concurrent insulinoma.38
Differential Diagnoses
The vague, nonspecific signs of lethargy and decreased activity are present in any myriad of diseases seen in ferrets, especially their most common endocrinopathies, insulinomas, and hyperadrenocorticism. Owners may also mistake the decreased activity as normal changes associated with aging. Ferrets that are on long-term steroid therapy to manage their insulinoma-induced hypoglycemia will often become obese and have a pot-bellied appearance. Rear-leg weakness may be associated with hypoglycemia, myasthenia gravis, or thromboembolic disease.
Diagnostic Testing
As in dogs and cats, eliminating the possibility of sick euthyroid syndrome is essential to diagnosing hypothyroidism. Diagnostic testing to rule out concurrent disease should be performed along with evaluation of the thyroid.
Experimentally, intravenous injection of 1 IU of thyroid-stimulating hormone (TSH) (Dermathycin) per ferret was sufficient to evaluate thyroid function in adult male ferrets, and showed significant increase in circulating thyroxine (T4) after 2 hours. Measured tri-iodothyronine (T3) values did not change for at least 6 hours, making measurement of circulating T4 an easier and more feasible parameter to measure in clinical practice.41
Unfortunately, Dermathycin is no longer available for purchase in the United States. More recently, however, T4 levels were measured using human recombinant TSH (Thyrogen) in 11 neutered ferrets, and successful stimulation of the thyroid axis was achieved. Prestimulation values for T4 in neutered male and female ferrets were determined to be 29.9 ± 5.8 ng/mL and 21.8 ± 3.3 ng/mL, respectively. Ferrets were stimulated using 100 μg Thyrogen intramuscularly, and euthyroid ferrets were found to have an increase of 1.4 times basal levels after 4 hours.38 A concurrent study involving 25 laboratory and pet neutered ferrets using the same protocol was also released in 2012. The investigators noted a median poststimulation T4 level at 34.8% above prestimulatory levels, and found the mean plasma T4 of euthyroid ferrets to be 21.3 nmol/L.42
Treatment
Oral levothyroxine at 50 to 100 μg every 12 hours was able to return serum T4 to normal resting levels in test studies. Treatment also led to increased activity, weight loss, and increased rear-leg function. Ferrets with sick euthyroid syndrome were not responsive to levothyroxine therapy.38
Prognosis
Prognosis with T4 supplementation has thus far been excellent for the 7 animals studied.38 As the pathogenesis of hypothyroidism in ferrets is unknown, the long-term prognosis has yet to be determined, as has whether adjustments to the dose of levothyroxine will need to be adjusted over time.
Pure red cell aplasia
Pure red cell aplasia (PRCA) is a nonregenerative anemia characterized by a marked deficiency of erythroid precursors despite normal numbers of granulocytes and megakaryocytes in the bone marrow.43 Though a rare condition, PRCA may be underreported in ferrets because other more common causes of anemia, such as hyperestrogenism and lymphoma, carry a guarded prognosis. Therefore owners may elect to provide palliative symptomatic care or euthanize the animal rather than have the anemia fully worked up.
Etiology and Pathogenesis
Variable mechanisms have been implicated in the causes of the bone marrow hypoplasia, including the production of antibodies against the erythroid precursors, inhibition of erythropoiesis owing to alterations in cytokine production, production of antibodies against erythropoietin, and induction of antibodies during an infection against antigens that cross-react with an epitope on erythroid precursors.44
Congenital and acquired types of PRCA have been described, with the majority of cases being acquired. Congenital PRCA, also known as Diamond-Blackfan anemia, is a heritable disease that develops during early infancy in humans and is often associated with other various congenital abnormalities such as craniofacial dysmorphism, cardiac defects, urogenital malformations, cleft palate, neck and thumb abnormalities, and generalized growth failure.44, 45 A congenital case of PRCA has also been described in a 3-month-old dog based on the criteria of a progressive normochromic and normocytic anemia in infancy and an absence of erythroid maturation in the bone marrow.46 In the author’s (S.C.) practice, a 5-month-old ferret (See Table 4 - Case A) was presented for a severe and progressive anemia with confirmed erythroid hypoplasia of the bone marrow. This ferret may possibly represent a case of congenital PRCA; however, the ferret had been vaccinated just 3 weeks before presentation, and it is unknown if this animal had an inherited condition or if the PRCA developed as a consequence of the vaccination.
Table 4.
PRCA signalment, treatment, and outcome
| Signalment | Initial PCV (%) | Concurrent Disease | Treatment | Response to Treatment/Outcome | Ref./Year |
|---|---|---|---|---|---|
| 8-mo-old female | 8 | Multiple blood transfusions Prednisolone 2 mg/kg PO every 12 h Cyclosporine 4 mg/kg PO every 12 h (added on 2 wk later) Cyclosporine 6 mg/kg PO every 12 h (dose increased after recurrence of clinical signs on day 86 of treatment) Azathioprine 1 mg/kg PO every 24 h (given for less than a week) Erythropoietin 40 U SC every 48 h × 3 injections |
No evidence of regeneration noted after 2 wk Marked regenerative response. PCV increased to 25% and increase in reticulocytes within 2 wk. PCV increased to 37% and reticulocytosis noted within 5 d Likely did not contribute to increase in PCV Likely did not contribute to increase in PCV |
53 | |
| 5-mo-old male (Case A) | 18 | Vaccinated with distemper 3 wk before presentationa | Multiple blood transfusions Prednisolone 1 mg/kg PO every 12 h Cyclosporine 2.2 mg/kg PO every 12 h (added on as second immunosuppressive drug owing to lack of response to prednisolone) Cyclophosphamide 10 mg/kg PO |
PCV continued to drop to 10% over 15 d Patient was on medication for less than a week before passing away Patient passed away 5 d after receiving single dose |
2004 |
| 4-y-old male (Case B) | 26 | Severe lymphocytic leukocytosis 3 wk prior with a PCV of 38%. Leukocytosis improved with a three week course of Clavamox 12.5 mg/kg PO 12 h (suspected to be reactive lymphocytes due to immune stimulation) | Prednisolone 2 mg/kg PO every 12 h Tapered prednisolone in 4-wk increments down to 1 mg/kg PO every 24 h Cyclophosphamide 5 mg/kg PO every 12 h (added on, in addition to increasing prednisolone back to 2 mg/kg PO every 12 h) |
PCV increased to 41% within 4 wk Patient became anemic again (32%) 10 wk into treatment (week 6 of taper) Patient euthanized 6 d later when patient presented in lateral recumbency. Biliary hyperplasia and hepatocellular degeneration noted on necropsy. Numerous hemosiderin laden macrophages noted in the bone marrow and all cell lines were present at the time of death |
2009 |
| 6 y-old female (Case C) | 27 | Normal PCV just 6 wk prior. Month-long history of diarrhea, mild fever, and splenomegaly | Prednisolone 2 mg/kg PO every 12 h | PCV 46% within 4 wk. Complete remission Patient became diabetic 5 mo later; was tapered off the prednisolone and started on insulin. No recurrence of anemia |
2012 |
| 6-y-old male (Case D)b | 17 | Insulinoma and hyperadrenocortisim (confirmed with adrenal panel). Was 38% on preoperative blood work just 4 d prior | Multiple blood transfusions Was already on Prednisolone 0.75 mg/kg PO every 12 h for management of hypoglycemia. Unable to wean off prednisolone because of persistent hypoglycemia after surgery Multiple blood transfusions Patient passed away before immunosuppressive therapy could be initiated |
Adrenalectomy and insulinoma nodulectomy performed because hyperestrogenism was the suspected cause for the anemia and thrombocytopenia. Patient’s PCV increased to 51% after 4 wk PCV remained normal for 9 wk Presented 13 wk after surgery. Patient arrested during sedation to collect bone marrow aspirate and core biopsy |
2012 |
|
11 |
Insulinoma; cachexia of unknown cause. No recurrence of hyperestrogenism based on repeated adrenal panel just 2 wk prior |
Cases A–D are ferrets that presented to the author’s (S.C.) clinic. All ferrets had erythroid hypoplasia on cytologic analysis of the bone marrow.
Abbreviations: PCV, packed cell volume; PO, by mouth; PRCA, pure red cell aplasia; SC, subcutaneously.
It is unknown whether vaccination was the trigger for this ferret’s PRCA.
Initial presentation of anemia believed to be caused by hyperestrogenism secondary to hyperadrenocorticism because the ferret had a concurrent thrombocytopenia and the PCV improved several weeks after adrenalectomy. Recurrence of anemia noted 13 weeks later was believed to be caused by PRCA, because no evidence of hyperestrogenism was noted 2 weeks prior on an adrenal sex hormone panel and normal numbers of megakaryocytes and granulocytes were noted in the bone marrow.
Of those cases of acquired PRCA, primary and secondary forms of PRCA have been documented in dogs,47, 48, 49 cats,48, 50 and humans.44, 51 Primary PRCA is an autoimmune disorder, and is thought to be a variant form of immune-mediated hemolytic anemia (IMHA) whereby antibodies destroy erythroblasts in the bone marrow.48, 49 In these cases, the cause of antibody production is often idiopathic.
Secondary PRCA occurs after a systemic disease (eg, viral infections, various neoplasms) or treatment with certain drugs or vaccines.44, 47, 48, 49, 50, 51 Cats infected with FeLV subgroup C can develop a progressive, fatal PRCA,50 and parvoviral infections in dogs48, 49 have been associated with cases of PRCA. In humans, parvovirus B19 directly infects the erythroid precursors, resulting in premature apoptosis.44 Aleutian disease is a parvovirus that affects ferrets; however, to date it has not been implicated in the development of PRCA. Formation of antibodies against erythropoietin after treatment with recombinant human erythropoietin is thought to be the cause of some cases of PRCA in dogs.49, 52
In the published case of an 8-month-old ferret, extensive testing was performed to find an underlying cause of the erythroid hyperplasia, but none was found.53 Based on the response to immunosuppressive medications, an immune-mediated mechanism was thought to be involved, and the conclusion was made that this was a case of primary PRCA. In the author’s (S.C.) practice, 4 additional ferrets have been diagnosed with PRCA based on erythroid hypoplasia of the bone marrow. The previously described 5-month-old ferret (Case A) had been vaccinated for canine distemper just 3 weeks before presentation, and it is unknown whether erythroid hypoplasia in the bone marrow was an idiosyncratic reaction to the vaccine or if this ferret had a primary or congenital form of PRCA. The other 3 ferrets diagnosed with PRCA were mature (older than 4 years) and had other systemic illness preceding or concurrent with their severe anemia, including chronic episodes of diarrhea in one ferret (Case C), insulinoma and hyperadrenocorticism in the second (Case D), and severe leukocytosis and generalized weight loss of unknown cause in the third (Case B) (Table 4 ).
Clinical Signs
Similar to cases seen in dogs47, 49 and cats,50 ferrets with PCRA usually present for a progressive weakness and lethargy ranging from several days to a few months in duration, although some were alert and responsive on admission despite their moderate to severe anemia. On physical examination, extreme pallor of the mucous membranes is the primary abnormality noted, just as seen in canine49 and feline50 cases. Cardiac murmurs have been reported in both dogs49 and cats,50 but were not noted in the published ferret case53 or in any of the ferrets in the author’s (S.C.) practice. The 3 older ferrets presented to this clinic were in fair to poor body condition, presumably resulting from their concurrent illnesses.
Differential Diagnoses
Differentials for PRCA encompass other more common causes of anemia. Anemia due to blood loss, secondary to trauma and gastric ulceration, is a common occurrence in ferrets. Gastric ulceration induced by stress and Helicobacter mustelidae can lead to significant melena and chronic anemia. Coagulopathies can also result in significant blood loss, and include certain types of rodenticide toxicities, disseminated intravascular coagulation, thrombocytopenia, and severe hepatic failure, resulting in deficits of coagulation factors. Anemia can also be a result of decreased red cell production, with lymphoma and renal disease as possible causes. Although anemia of chronic disease was considered as a possible differential for the ferrets with concurrent disease, the progressive decline in the packed cell volume (PCV) despite improvement or resolution of their underlying condition makes this less likely.
Hyperestrogenemia can result in a life-threatening anemia attributable to a pancytopenia. The anemia is a result of decreased production of red blood cells and increased blood loss secondary to thrombocytopenia. Bone marrow suppression from hyperestrogenism has been documented in intact females in prolonged estrus, spayed females with an ovarian remnant, and rarely in neutered males and females with hyperadrenocorticism.54, 55
Anecdotally, immune-mediated hemolytic anemia (IMHA) has been noted in ferrets, although there are no published cases in the literature.54 Dogs and cats with IMHA typically have a strong regenerative response, indicating that the destruction is targeted at mature red cells rather than the erythroblasts in the bone marrow.47 Although a strong regenerative response is noted in most cases of IMHA, a nonregenerative IMHA characterized by erythroid maturation arrest and pathologic changes in the bone marrow (eg, dysmyelopoiesis, myelonecrosis, myelofibrosis) has been described in dogs and cats.48
Diagnostic Testing
A severe anemia without leukopenia or thrombocytopenia is the primary clinical finding in animals with PRCA. The normal hematocrit of ferrets is 46% to 61% and is higher than in other species, so a mild or moderate anemia may go unrecognized unless a clinician is aware of this difference.54 The anemia is typically normocytic and normochromic with a poor reticulocyte response to the anemia, indicating that the anemia is nonregenerative.43 In ferrets, normal reticulocyte counts can be as high as 10%, with counts higher than 12% during a regenerative response.54
Severe anemia (<18%) was noted in 2 of the ferrets (see Table 4, Cases A and D) in the author’s (S.C.) practice and in the one published case report.53 The number of leukocytes and platelets were not affected except in one patient that had a lymphocytic leukocytosis three weeks prior suggestive of immune stimulation (Case B - see Table 4).
For those ferrets that were severely anemic, serum biochemistries were not performed. Serum biochemistries performed on the ferrets that were only moderately anemic on initial presentation had no significant abnormalities, other than hypoglycemia in one ferret that had an insulinoma.
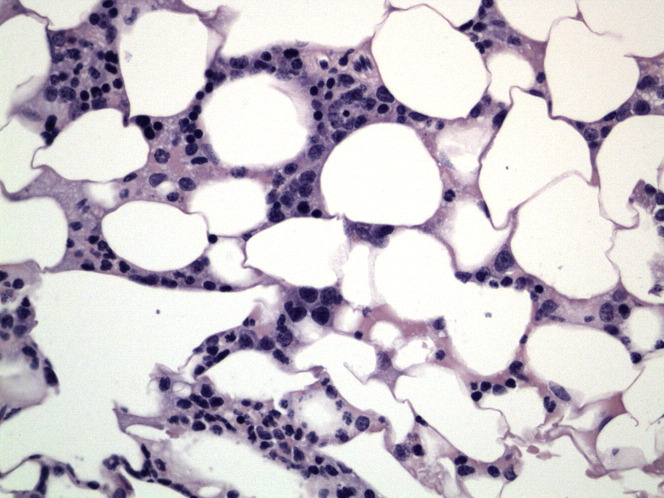
A bone marrow aspirate or bone core biopsy is indicated in any nonregenerative anemia.54 Ideally both are performed, because aspirates are ideal for evaluating cell morphology, whereas core biopsies are preferred for evaluating marrow cellularity.56 A CBC should also be performed at the same time as sample collection for an accurate interpretation of the bone marrow (Fig. 1 ).56 Samples can be collected in ferrets from the proximal femur, humerus, and iliac crest.57 Analysis of the bone marrow reveals extreme depletion of the erythroid precursors in the presence of normal granulopoiesis and thrombopoiesis.49, 50, 53 This selective hypoplasia is believed to be a result of immune-mediated destruction of erythroblasts. This disease differs from the typical appearance of IMHA, whereby there are normal to increased numbers of erythroblasts in the bone marrow. The singular loss of only the erythroid cell line and no changes in the myeloid or megakaryocytic cell lines results in a marked increase in the myeloid-to-erythroid ratio. The myeloid-to-erythroid ratio in ferrets has been reported to range from 1:3 to 3:1.5.58 In the one published case report, the ferret had a myeloid-to-erythroid ratio of 4.6:1.53
Fig. 1.
Ferret bone marrow with erythroid hypoplasia. (original magnification ×40).
(Courtesy of Catherine Pfent, DVM, Texas A&M College of Veterinary Medicine, College Station, TX.)
An antinuclear antibody test (ANA) can be performed to assess for autoantibodies as part of the diagnostic workup of certain autoimmune diseases. However, it is uncertain whether this test is effective in ferrets because ANA titers were negative in the one published case study.53
Treatment
The mainstay of treatment is to provide whole blood transfusions to support the ferret while using immunosuppressive drug therapy to cease the destruction of the immature red blood cells.
Blood transfusion
For ferrets that are clinical for their anemia, blood transfusions are indicated to provide symptomatic support so that immunosuppressive therapy has an opportunity to take effect. There are no known blood types in ferrets, so risk from transfusion reaction is lower than for other species.59 In the published case report, the anemic ferret received multiple transfusions over the course of treatment, including blood from 3 different donors for the third transfusion.53 Multiple blood transfusions from different individuals have also been administered to anemic ferrets in the author's (S.C.) practice, with no observed adverse effects. However, the recipient is always closely monitored during a transfusion for adverse reactions, such as spikes in temperature, tachycardia, tachypnea, pruritus, and vomiting. If noted, administration of the blood is temporarily stopped or given more slowly. The recipient can also be premedicated with diphenhydramine (2 mg/kg intramuscularly, intravenously, or orally) as a preventative for a transfusion reaction.60
Large healthy males make the best donors, as they are able to donate a larger volume of blood. Approximately 1% of the donor's body weight can be collected safely. Because a relatively large volume is being collected, the donor should be sedated to minimize risks of venipuncture from the anterior vena cava. Isoflurane is commonly used; however, this can cause a significant drop in PCV because of splenic sequestration of red blood cells.61 In their practice, the authors have sedated donor ferrets with subcutaneous or intramuscular injections of ketamine (10 mg/kg) and midazolam (1 mg/kg) to minimize the reduction in PCV. The neck of the donor ferret is shaved and sterilely prepped before venipuncture. A 23-gauge butterfly catheter is inserted into the thoracic cavity just cranial to the first rib and directed toward the opposite rear limb. The blood is collected into a syringe prefilled with Acid-Citrate-Dextrose (ACD) anticoagulant (1 part ACD to 9 parts blood). Supplemental subcutaneous fluids and an injection of iron dextran (10 mg/kg intramuscularly) are administered to the donor ferret during recovery.
If a donor is not available, oxyglobin (hemoglobin glutamer 200 [bovine]) can be used if the ferret is in an acute crisis from its anemia.62 However, its use is limited for stable patients with chronic anemia because of oxyglobin's short half-life of approximately 18 to 43 hours.63 Oxyglobin had been previously withdrawn from the market in 2010, but production has resumed in early 2013 and it is now available in the United Kingdom and Republic of Ireland (http://www.dechra.eu/dechra-news.aspx?PID=199&Action=1&Year=2013&NewsId=1299; accessed January 29, 2013).
Immunosuppressive therapy
Corticosteroids are often given as the first treatment in cases of PRCA for humans,44, 51 dogs,49 and cats.50 However, relapse is not uncommon (up to 80% in human cases), especially when the medication is tapered after remission.51 Prednisolone at immunosuppressive doses (2 mg/kg orally every 12–24 hours) have been used in ferrets. In the published case report, no evidence of regeneration (reticulocyte count of 0 cells/μL) was noted after 2 weeks of prednisolone (2 mg/kg orally every 12 hours) therapy alone.53 Improvement on prednisolone (2 mg/kg orally every 12 hours) was seen in 2 of the 4 cases (Case B and C) seen in the author’s practice. Increases in PCV to near normal levels were noted within 4 to 5 weeks. However, relapse of the anemia was seen in 1 of the ferrets (Case B) while the prednisolone was being tapered. A third ferret (Case D) was already on prednisolone (0.75 mg/kg orally every 12 hours) for management of insulinomas when it presented for its anemia, though not at an immunosuppressive dose. In this particular ferret, his initial presentation of anemia was believed to be caused by hyperestrogenism secondary to confirmed hyperadrenocorticism, because the patient had a concurrent thrombocytopenia and its PCV increased from 18% to 51% within 4 weeks after surgery. The ferret’s PCV remained in normal ranges until recurrence of the anemia was noted 13 weeks later. This second incidence was believed to be caused by PRCA, because no evidence of hyperestrogenism was noted 2 weeks prior on an adrenal sex hormone panel. Erythroid hypoplasia of the bone marrow was confirmed at necropsy. The PCV of the final ferret (Case A) continued to drop from 18% down to 10% over 2 weeks while receiving prednisolone, but it was most likely receiving a suboptimal dose (1 mg/kg orally every 12 hours) (see Table 4).
Because of the high rate of reoccurrence, prednisolone is rarely used alone for the treatment of PRCA. Response to cyclosporine A is considered excellent in humans.51 In the published ferret case, a regenerative response and improvement in the anemia were noted 2 weeks after the administration of cyclosporine (4 mg/kg orally every 12 hours) as a second immunosuppressive medication.53 A recurrence of the nonregenerative anemia was noted on day 86 of treatment, approximately 2 weeks after the trough plasma concentration of cyclosporine had decreased. The cyclosporine dosage was increased (6 mg/kg orally every 12 hours), and a significant increase in both reticulocyte number and trough plasma concentration of cyclosporine was noted within 5 days. Administration of both prednisolone and cyclosporine were continued for a total of 14 months, at which point the ferret appeared to be in complete remission and remained in remission for up to 21 months after cessation of all medications.53
In the published report, azathioprine (1 mg/kg orally every 24 hours) was initiated when the ferret came out of remission on day 86. However, because azathioprine has delayed immunosuppressive effects, it most likely did not play a role in the ferret’s regenerative response and was discontinued after less than a week.53 Cyclophosphamide, an alkylating agent used to treat various autoimmune conditions and cancer, has been used in dogs49 and cats50 for the treatment of PRCA. Although cyclophosphamide is used in chemotherapeutic protocols for lymphoma in ferrets,64 its use for treatment of PRCA in ferrets has not been documented. Antithymocyte globulin, alemtuzumab, rituximab, and intravenous immunoglobulin have been used in the treatment of acquired PRCA in humans, but their use in veterinary medicine has not been reported.51 Erythropoietin has been used in cases of canine and feline anemia associated with end-stage renal failure, but it has questionable benefits in patients with normal renal function.53, 65 In addition, the use of erythropoietin has been implicated in some cases of acquired PRCA in other species.49, 52 Although erythropoietin was administered to the ferret in the one published case report, it was not thought to be a factor in that ferret’s remission. In other species, erythropoietin is effective only after administration for several weeks, and the ferret had a marked reticulocytosis within 5 days of initiating treatment with erythropoietin. The dosage of the ferret’s cyclosporine had been increased at the same time and was thought to be the more likely cause of the regenerative response.53
Prognosis
In other species, prognosis depends on the inciting cause of PRCA. In people, cases of secondary PRCA due to medications are often transient and reversible when the medications are discontinued.44 Most dogs (10 out of 13) responded well to immunosuppressive medications, although initial response to therapy had a median of 38 days (range 22–87 days) and complete remission had a median time of 118 days (range 58–187 days).49 A rapid response (1.5–5 weeks) was noted in 6 of 7 cats; however, a relapse and/or refractoriness to treatment was noted if the cats were not treated promptly and aggressively with long-term immunosuppressive medication.50 In addition, cats that develop PRCA after an infection with FeLV subgroup C do not respond to immunosuppressive therapy, and usually succumb to their anemia.50
The prognosis in ferrets is unknown because of the paucity of reports, but complete remission has been reported in one ferret treated with multiple blood transfusions and immunosuppressive therapy.53 In the author’s (S.C.) practice, 1 ferret (Case C) responded extremely well to prednisolone alone, had complete remission, and has not had a recurrence of its anemia 1 year later. A partial and short-term response was noted in 1 ferret (Case B), but the nonregenerative anemia returned while in the process of weaning off the prednisolone. It is unknown whether the ferret with concurrent insulinoma and hyperadrenocorticism (Case D) had a delay in the appearance of his PRCA because it was on long-term steroids, albeit not at an immunosuppressive dose. The case in the juvenile ferret (Case A) had no response to immunosuppressive therapy and multiple blood transfusions, which may have been due to suboptimal dosing of the prednisolone (see Table 4). Based on these cases, ferrets possibly respond in a similar manner to cats, for which prompt and aggressive long-term immunosuppressive therapy is required to prevent recurrence of erythroblast destruction and refractoriness to medication.
Summary
There are several emerging diseases described in ferrets that occur with unknown frequency or may be overshadowed by those conditions that are more commonly seen. Based on current published data, it is unknown whether these emerging conditions occur sporadically or are increasing in incidence. Being cognizant of these diseases will allow for earlier detection and identification of affected ferrets. In turn, this can lead to more effective and successful treatment of affected patients in clinical practice. Diagnosis and reporting of cases is also crucial in expanding our understanding of these rarer diseases, and will facilitate elucidation of their incidence, etiology, and pathology.
References
- 1.Hirsh D., Biberstein E. 2nd edition. Blackwell Publishing; Oxford (United Kingdom): 2004. Yeast—cryptococcus, malassezia, and candida: veterinary microbiology. [Google Scholar]
- 2.Sykes J., Malik R. 4th edition. WB Saunders; Philadelphia: 2011. Cryptococcosis. Infectious diseases of the dog and cat. [Google Scholar]
- 3.Franzot S.P., Salkin I.F., Casadevall A. Cryptococcus neoformans var. grubii: separate varietal status for Cryptococcus neoformans serotype A isolates. J Clin Microbiol. 1999;37:838–840. doi: 10.1128/jcm.37.3.838-840.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Duncan C., Schwantje H., Stephen C. Cryptococcus gattii in wildlife of Vancouver Island, British Columbia, Canada. J Wildl Dis. 2006;42:175–178. doi: 10.7589/0090-3558-42.1.175. [DOI] [PubMed] [Google Scholar]
- 5.Mitchell T.G., Perfect J.R. Cryptococcosis in the era of AIDS—100 years after the discovery of Cryptococcus neoformans. Clin Microbiol Rev. 1995;8:515–548. doi: 10.1128/cmr.8.4.515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Skulski G., Symmers W.S. Actinomycosis and torulosis in the ferret (Mustela furo L) J Comp Pathol. 1954;64:306. doi: 10.1016/s0368-1742(54)80032-4. [DOI] [PubMed] [Google Scholar]
- 7.Malik R., Alderton B., Finlaison D. Cryptococcosis in ferrets: a diverse spectrum of clinical disease. Aust Vet J. 2002;80:749–755. doi: 10.1111/j.1751-0813.2002.tb11343.x. [DOI] [PubMed] [Google Scholar]
- 8.Lewington J. Isolation of Cryptococcus neoformans from a ferret. Aust Vet J. 2008;58:124. doi: 10.1111/j.1751-0813.1982.tb00619.x. [DOI] [PubMed] [Google Scholar]
- 9.Greenlee P., Stephens E. Meningeal cryptococcosis and congestive cardiomyopathy in a ferret. J Am Vet Med Assoc. 1984;184:840. [PubMed] [Google Scholar]
- 10.Hanley C.S., MacWilliams P., Giles S. Diagnosis and successful treatment of Cryptococcus neoformans variety grubii in a domestic ferret. Can Vet J. 2006;47:1015. [PMC free article] [PubMed] [Google Scholar]
- 11.Eshar D., Mayer J., Parry N.M. Disseminated, histologically confirmed Cryptococcus spp infection in a domestic ferret. J Am Vet Med Assoc. 2010;236:770–774. doi: 10.2460/javma.236.7.770. [DOI] [PubMed] [Google Scholar]
- 12.Malik R., Martin P., McGill J. Successful treatment of invasive nasal cryptococcosis in a ferret. Aust Vet J. 2000;78:158–159. doi: 10.1111/j.1751-0813.2000.tb10582.x. [DOI] [PubMed] [Google Scholar]
- 13.Morera N., Juan-Sallés C., Torres J.M. Cryptococcus gattii infection in a Spanish pet ferret (Mustela putorius furo) and asymptomatic carriage in ferrets and humans from its environment. Med Mycol. 2011;49:779–784. doi: 10.3109/13693786.2011.564216. [DOI] [PubMed] [Google Scholar]
- 14.Ropstad E.O., Leiva M., Peña T. Cryptococcus gattii chorioretinitis in a ferret. Vet Ophthalmol. 2011;14:262–266. doi: 10.1111/j.1463-5224.2011.00885.x. [DOI] [PubMed] [Google Scholar]
- 15.Lester S.J., Kowalewich N.J., Bartlett K.H. Clinicopathologic features of an unusual outbreak of cryptococcosis in dogs, cats, ferrets, and a bird: 38 cases (January to July 2003) J Am Vet Med Assoc. 2004;225:1716–1722. doi: 10.2460/javma.2004.225.1716. [DOI] [PubMed] [Google Scholar]
- 16.Erdman S.E., Moore F.M., Rose R. Malignant lymphoma in ferrets: clinical and pathological findings in 19 cases. J Comp Pathol. 1992;106:37–47. doi: 10.1016/0021-9975(92)90066-4. [DOI] [PubMed] [Google Scholar]
- 17.Malik R., McPetrie R., Wigney D. A latex cryptococcal antigen agglutination test for diagnosis and monitoring of therapy for cryptococcosis. Aust Vet J. 1996;74:358–364. doi: 10.1111/j.1751-0813.1996.tb15445.x. [DOI] [PubMed] [Google Scholar]
- 18.Krockenberger M., Canfield P., Kozel T. An immunohistochemical method that differentiates Cryptococcus neoformans varieties and serotypes in formalin-fixed paraffin-embedded tissues. Med Mycol. 2001;39:523–533. doi: 10.1080/mmy.39.6.523.533. [DOI] [PubMed] [Google Scholar]
- 19.Plumb D. Amphotericin B desoxycholate/amphotericin B lipid-based. In: Plumb D., editor. Veterinary drug handbook. 7th edition. Wiley-Blackwell; Ames (IA): 2011. pp. 62–67. [Google Scholar]
- 20.Plumb D. Flucytosine. In: Plumb D., editor. Veterinary drug handbook. 7th edition. Worldwide Print Distribution; Ames (IA): 2011. pp. 432–434. [Google Scholar]
- 21.Plumb D. Itraconazole. In: Plumb D., editor. Veterinary drug handbook. 7th edition. Wiley-Blackwell; Ames (IA): 2011. pp. 558–561. [Google Scholar]
- 22.Plumb D. Fluconazole. In: Plumb D., editor. Veterinary drug handbook. 7th edition. Wiley-Blackwell; Ames (IA): 2011. pp. 430–432. [Google Scholar]
- 23.CDC. Seasonal influenza: flu basics. Available at: http://www.cdc.gov/flu/about/disease/index.htm. Accessed January 3, 2013.
- 24.Huang S.S., Banner D., Fang Y. Comparative analyses of pandemic H1N1 and seasonal H1N1, H3N2, and influenza B infections depict distinct clinical pictures in ferrets. PLoS One. 2011;6:e27512. doi: 10.1371/journal.pone.0027512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.CDC. Influenza type A viruses and subtypes. In: CDC, editor. 2011. Available at: http://www.cdc.gov/flu/avianflu/influenza-a-virus-subtypes.htm. Accessed January 3, 2013.
- 26.CDC. 2009-2010 influenza (flu) season. Available at: http://www.cdc.gov/flu/pastseasons/0910season.htm. Accessed January 3, 2013.
- 27.Schrenzel M.D., Tucker T.A., Stalis I.H. Pandemic (H1N1) 2009 virus in 3 wildlife species, San Diego, California, USA. Emerg Infect Dis. 2011;17:747. doi: 10.3201/eid1704.101355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Campagnolo E., Moll M., Tuhacek K. Concurrent 2009 pandemic influenza A (H1N1) virus infection in ferrets and in a community in Pennsylvania. Zoonoses Public Health. 2013;60:117–124. doi: 10.1111/j.1863-2378.2012.01503.x. [DOI] [PubMed] [Google Scholar]
- 29.Swenson S.L., Koster L.G., Jenkins-Moore M. Natural cases of 2009 pandemic H1N1 influenza A virus in pet ferrets. J Vet Diagn Invest. 2010;22:784–788. doi: 10.1177/104063871002200525. [DOI] [PubMed] [Google Scholar]
- 30.Patterson A.R., Cooper V.L., Yoon K.J. Naturally occurring influenza infection in a ferret (Mustela putorius furo) colony. J Vet Diagn Invest. 2009;21:527–530. doi: 10.1177/104063870902100417. [DOI] [PubMed] [Google Scholar]
- 31.Smith W., Stuart-Harris C. Influenza infection of man from the ferret. Lancet. 1936;228:121–123. [Google Scholar]
- 32.Govorkova E.A., Rehg J.E., Krauss S. Lethality to ferrets of H5N1 influenza viruses isolated from humans and poultry in 2004. J Virol. 2005;79:2191–2198. doi: 10.1128/JVI.79.4.2191-2198.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jackson S., Van Hoeven N., Chen L.M. Reassortment between avian H5N1 and human H3N2 influenza viruses in ferrets: a public health risk assessment. J Virol. 2009;83:8131–8140. doi: 10.1128/JVI.00534-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.CDC. Influenza A (H3N2) variant virus. Available at: http://www.cdc.gov/flu/swineflu/h3n2v-cases.htm. Accessed January 3, 2013.
- 35.CDC. Information on avian influenza. Available at: http://www.cdc.gov/flu/avianflu/index.htm. Accessed January 3, 2013.
- 36.Govorkova E.A., Ilyushina N.A., Boltz D.A. Efficacy of oseltamivir therapy in ferrets inoculated with different clades of H5N1 influenza virus. Antimicrob Agents Chemother. 2007;51:1414–1424. doi: 10.1128/AAC.01312-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pearson R., Gorham J. Viral disease models. In: Fox J., editor. Biology and diseases of the ferret. 2nd edition. Lippincott Williams & Wilkins; Baltimore (MD): 1998. pp. 487–497. [Google Scholar]
- 38.Wagner R. Hypothyroidism in ferrets. Association of Exotic Mammal Veterinarians 11th Annual Conference. Oakland (CA), October 25, 2012. p. 29–31.
- 39.Scott-Moncrieff J.C. Hypothyroidism. In: Ettinger S.J., editor. Textbook of Veterinary Internal Medicine. Elsevier; 2004. pp. 1535–1544. [Google Scholar]
- 40.Rand J.S., Levine J., Best S.J. Spontaneous adult-onset hypothyroidism in a cat. J Vet Intern Med. 1993;7:272–276. doi: 10.1111/j.1939-1676.1993.tb01019.x. [DOI] [PubMed] [Google Scholar]
- 41.Heard D., Collins B., Chen D. Thyroid and adrenal function tests in adult male ferrets. Am J Vet Res. 1990;51:32–35. [PubMed] [Google Scholar]
- 42.Mayer J. Use of recombinant human TSH hormone for a thyrotropin stimulation test in euthyroid ferrets. Association of Exotic Mammal Veterinarians 11th Annual Conference. Oakland (CA), October 25, 2012.
- 43.Weiss D.J., Tvedten H. Erythrocyte disorders. In: Willard M.D., Tvedten H., editors. Small animal clinical diagnosis by laboratory methods. 5th edition. Elsevier Saunders; St Louis (MO): 2012. pp. 38–62. [Google Scholar]
- 44.Perkins S.L. Pediatric red cell disorders and pure red cell aplasia. Am J Clin Pathol. 2004;122:S70–S86. doi: 10.1309/3WWXLGK5U2MC0X9B. [DOI] [PubMed] [Google Scholar]
- 45.Ball S. Diamond Blackfan anemia. Hematology Am Soc Hematol Educ Program. 2011;2011:487–491. doi: 10.1182/asheducation-2011.1.487. [DOI] [PubMed] [Google Scholar]
- 46.Moore A., Day M., Graham M. Congenital pure red blood cell aplasia (Diamond-Blackfan anaemia) in a dog. Vet Rec. 1993;132:414. doi: 10.1136/vr.132.16.414. [DOI] [PubMed] [Google Scholar]
- 47.Stokol T., Blue J.T., French T.W. Idiopathic pure red cell aplasia and nonregenerative immune-mediated anemia in dogs: 43 cases (1988-1999) J Am Vet Med Assoc. 2000;216:1429–1436. doi: 10.2460/javma.2000.216.1429. [DOI] [PubMed] [Google Scholar]
- 48.Weiss D.J. Bone marrow pathology in dogs and cats with non-regenerative immune-mediated haemolytic anaemia and pure red cell aplasia. J Comp Pathol. 2008;138:46–53. doi: 10.1016/j.jcpa.2007.10.001. [DOI] [PubMed] [Google Scholar]
- 49.Weiss D.J. Primary pure red cell aplasia in dogs: 13 cases (1996-2000) J Am Vet Med Assoc. 2002;221:93–95. doi: 10.2460/javma.2002.221.93. [DOI] [PubMed] [Google Scholar]
- 50.Stokol T., Blue J. Pure red cell aplasia in cats: 9 cases (1989-1997) J Am Vet Med Assoc. 1999;214:75. [PubMed] [Google Scholar]
- 51.Sawada K., Fujishima N., Hirokawa M. Acquired pure red cell aplasia: updated review of treatment. Br J Haematol. 2008;142:505–514. doi: 10.1111/j.1365-2141.2008.07216.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Randolph J.E., Scarlett J., Stokol T. Clinical efficacy and safety of recombinant canine erythropoietin in dogs with anemia of chronic renal failure and dogs with recombinant human erythropoietin-induced red cell aplasia. J Vet Intern Med. 2004;18:81–91. doi: 10.1892/0891-6640(2004)18<81:ceasor>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 53.Malka S., Hawkins M.G., Zabolotzky S.M. Immune-mediated pure red cell aplasia in a domestic ferret. J Am Vet Med Assoc. 2010;237:695–700. doi: 10.2460/javma.237.6.695. [DOI] [PubMed] [Google Scholar]
- 54.Morrisey J.K., Kraus M.S. Cardiovascular and other diseases. In: Quesenberry K.E., Carpenter J.W., editors. Ferrets, rabbits, and rodents: clinical medicine and surgery. 3rd edition. Elsevier Saunders; St Louis (MO): 2012. pp. 62–77. [Google Scholar]
- 55.Chen S. Advanced diagnostic approaches and current medical management of insulinomas and adrenocortical disease in ferrets (Mustela putorius furo) Vet Clin North Am Exot Anim Pract. 2010;13:439–452. doi: 10.1016/j.cvex.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 56.Grindem C.B., Neel J.A., Juopperi T.A. Cytology of bone marrow. Vet Clin North Am Small Anim Pract. 2002;32:1313–1374. doi: 10.1016/s0195-5616(02)00052-9. [DOI] [PubMed] [Google Scholar]
- 57.Morrisey J., Ramer J. Ferrets. Clinical pathology and sample collection. Vet Clin North Am Exot Anim Pract. 1999;2:553. doi: 10.1016/s1094-9194(17)30110-x. [DOI] [PubMed] [Google Scholar]
- 58.Williams B.H. Disorders of rabbit and ferret bone marrow. In: Fudge A.M., editor. Laboratory medicine - avian and exotic pets. W.B. Saunders Company; Philadelphia: 2000. pp. 276–284. [Google Scholar]
- 59.Manning D., Bell J. Lack of detectable blood groups in domestic ferrets: implications for transfusion. J Am Vet Med Assoc. 1990;197:84. [PubMed] [Google Scholar]
- 60.Plumb D. Diphenhydramine HCl. In: Plumb D., editor. Veterinary drug handbook. 7th edition. Wiley-Blackwell; Ames (IA): 2011. pp. 338–340. [Google Scholar]
- 61.Marini R., Callahan R., Jackson L. Distribution of technetium 99m-labeled red blood cells during isoflurane anesthesia in ferrets. Am J Vet Res. 1997;58:781. [PubMed] [Google Scholar]
- 62.Lichtenberger M. Transfusion medicine in exotic pets. Clin Tech Small Anim Pract. 2004;19:88–95. doi: 10.1053/j.ctsap.2004.01.006. [DOI] [PubMed] [Google Scholar]
- 63.Plumb D. Hemoglobin glutamer-200 (Bovine) In: Plumb D., editor. Veterinary drug handbook. 7th edition. Wiley-Blackwell; Ames (IA): 2005. pp. 492–494. [Google Scholar]
- 64.Antinoff N., Williams B.H. Neoplasia. In: Quesenberry K.E., Carpenter J.W., editors. Ferrets, rabbits, and rodents: clinical medicine and surgery. 3rd edition. Elsevier Saunders; St Louis (MO): 2012. pp. 103–121. [Google Scholar]
- 65.Plumb D. Epoetin alfa, epoetin beta, erythropoietin, EPO, r-HuEPO. In: Plumb D., editor. Veterinary drug handbook. 7th edition. Wiley-Blackwell; Ames (IA): 2011. pp. 384–386. [Google Scholar]