Abstract
Chloroquine is a 9-aminoquinoline known since 1934. Apart from its well-known antimalarial effects, the drug has interesting biochemical properties that might be applied against some viral infections. Chloroquine exerts direct antiviral effects, inhibiting pH-dependent steps of the replication of several viruses including members of the flaviviruses, retroviruses, and coronaviruses. Its best-studied effects are those against HIV replication, which are being tested in clinical trials. Moreover, chloroquine has immunomodulatory effects, suppressing the production/release of tumour necrosis factor α and interleukin 6, which mediate the inflammatory complications of several viral diseases. We review the available information on the effects of chloroquine on viral infections, raising the question of whether this old drug may experience a revival in the clinical management of viral diseases such as AIDS and severe acute respiratory syndrome, which afflict mankind in the era of globalisation.
Chloroquine is a 9-aminoquinoline that has been known since 1934. Specifically synthesised to be used as an antimalarial agent, chloroquine was subsequently shown to have immunomodulatory properties that have encouraged its application in the treatment of autoimmune diseases such as rheumatoid arthritis. For this specific pathology, chloroquine and its hydroxy-analogue hydroxychloroquine have represented a valid contribution to the available pharmacological tools, since they proved able to slow down the progress of the disease while showing limited toxicity.1
Unfortunately, chloroquine is being gradually dismissed from antimalarial therapy and prophylaxis, due to the continuous emergence of chloroquine-resistant Plasmodium falciparum strains. However, the tolerability, low cost, and immunomodulatory properties of chloroquine/hydroxychloroquine are associated with biochemical effects that suggest a potential use in viral infections, some of whose symptoms may result from the inflammatory response.2, 3 We raise the question of whether this old drug whose parent compound, quinine, was isolated in the late 19th century from the bark of the tropical cinchona tree, may experience a revival in the clinical management of viral diseases of the era of globalisation.
General biochemical and cellular effects of chloroquine
Both chloroquine and hydroxychloroquine are weak bases that are known to affect acid vesicles leading to dysfunction of several enzymes. Extracellularly, chloroquine/hydroxychloroquine is present mostly in a protonated form that, due to its positive charge, is incapable of crossing the plasma membrane. However, the non-protonated portion can enter the intracellular compartment, where, in turn, it becomes protonated in a manner inversely proportional to the pH, according to the Henderson-Hasselbach law. It is thus not surprising that chloroquine/hydroxychloroquine is concentrated within acidic organelles such as the endosome, Golgi vesicles, and the lysosomes, where the pH is low and most chloroquine/hydroxychloroquine molecules are positively charged.4 Chloroquine/hydroxychloroquine is extruded to the extracellular medium mostly by exocytosis and/or through the action of the multidrug resistance protein MRP-1, a cell surface drug transporter belonging to the ATP-binding cassette family, which also includes the more thoroughly studied P-glycoprotein.5, 6, 7
It is well established that weak bases, by increasing the pH of lysosomal and trans-Golgi network (TGN) vesicles, disrupt several enzymes including acid hydrolases and inhibit the post-translational modification of newly synthesized proteins. The chloroquine-mediated rise in endosomal pH modulates iron metabolism within human cells by impairing the endosomal release of iron from ferrated transferrin, thus decreasing the intracellular concentration of iron. This decrease in turn affects the function of several cellular enzymes involved in pathways leading to replication of cellular DNA and to expression of different genes.8, 9
General mechanisms of viral inhibition by chloroquine
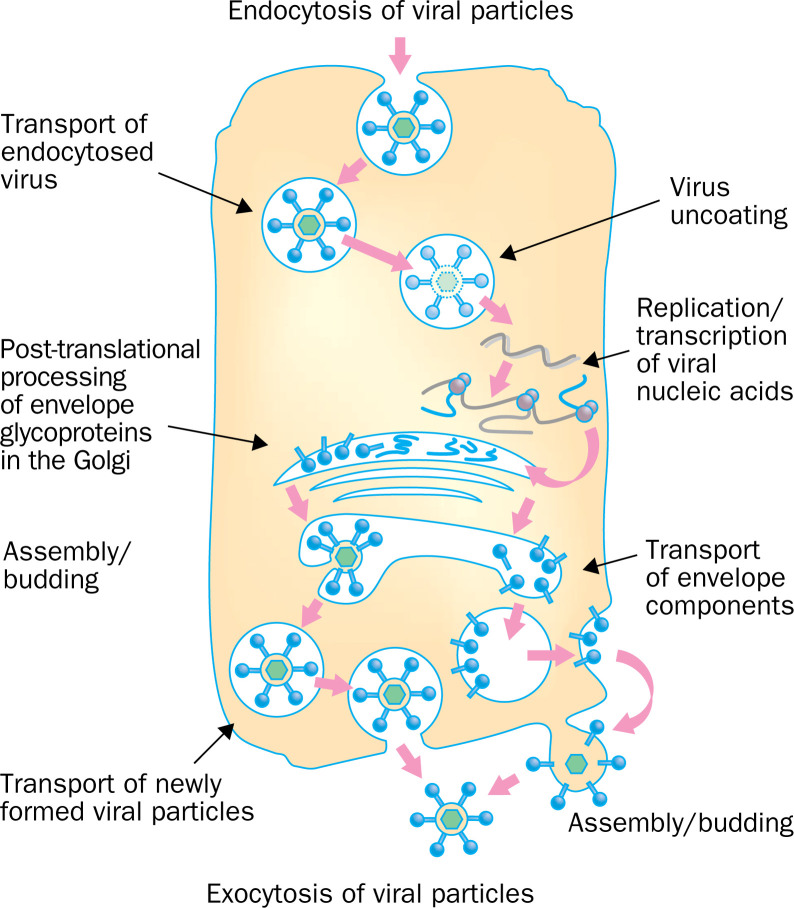
Chloroquine/hydroxychloroquie can impair the replication of several viruses by interacting with the endosome-mediated viral entry or the late stages of replication of enveloped viruses (figure 1 ).
Figure 1.
Steps of the replication of different viruses affected by chloroquine (marked by red rectangles). Chloroquine inhibits the replication of different viruses either at the early or late stages of viral replication.
Endosome-mediated viral entry interaction
Some viruses enter their target cells by endocytosis. This process targets the virus to the lysosomal compartment where the low pH, along with the action of enzymes, disrupts the viral particle, thus liberating the infectious nucleic acid and, in several cases, enzymes necessary for viral replication. Chloroquine has been shown to inhibit different viruses requiring a pH-dependent step for entry, such as the Borna disease virus,10 the minute virus of mice MVMp,11 and the avian leucosis virus.12 Of particular interest for human pathology is the report that chloroquine inhibits uncoating of the hepatitis A virus, thus blocking its entire replication cycle.13
Replication of enveloped viruses interaction
For some enveloped viruses, post-translational modification of the envelope glycoproteins occurs within the endoplasmic and TGN vesicles. This process involves proteases and glycosyl-transferases, some of which require a low pH. In line with the pH-dependence of these events, chloroquine was seen to inhibit budding of Mayaro virus particles,14 and to induce accumulation of non-infectious herpes simplex virus 1 particles in the TGN.15 Chloroquine also inhibits the replication of members of the Flaviviridae family by affecting the normal proteolytic processing of the flavivirus prM protein to M.16 As a result, viral infectivity is impaired. Finally, chloroquine induces the production of non-infectious retrovirus particles, as shown with the avian reticuloendotheliosis virus REV-A and with HIV-1.17 The mechanism of inhibition seems to be inhibition of glycosylation of the envelope glycoproteins, as will be discussed below.
Effects of chloroquine on the immune system
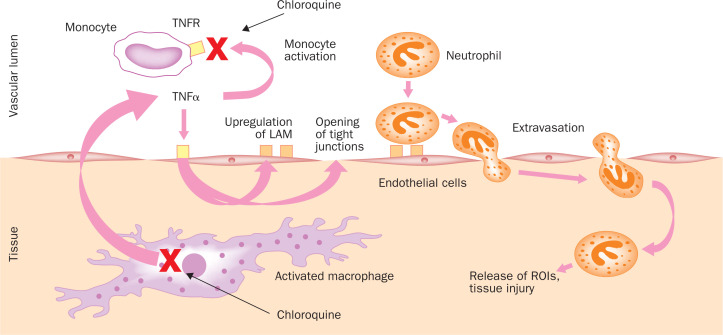
The accumulation of chloroquine/hydroxychloroquine in lymphocytes and macrophages results in anti-inflammatory properties, and has led to its clinical use in conditions such as rheumatoid arthritis, lupus erythematosus, and sarcoidosis, the last being characterised by an overproduction of tumour necrosis factor α (TNFα) by the alveolar macrophages.18 Chloroquine/hydroxychloroquine reduces the secretion of these proinflammatory cytokines and in particular TNFα, as shown in a murine macrophage cell line,19 and in primary cells such as mouse peritoneal macrophages,20 human peripheral blood mononuclear cells,21 and human whole blood.22 Several mechanisms have been evoked to explain the chloroquine/hydroxychloroquine-induced inhibition of TNFα production by monocyte-macrophages: disruption of cellular iron homeostasis,23 inhibition of TNFα mRNA expression,20 inhibition at a pretranslational stage by a non-lysosomotropic mechanism,24 or at a post-translational stage by blocking the conversion from cell-associated pro-TNFα to a soluble mature form.19 Apart from inhibiting TNFα production by stimulated monocyte-macrophages, chloroquine also decreases the surface expression of TNFα receptors in human monocytic cell lines and, hence, the receptor-mediated TNFα signalling.25 The results of such an impairment of TNFα-mediated signalling are shown in figure 2 .
Figure 2.
Effects of chloroquine on the immune system. TNFα is produced by activated monocytes/macrophages. Among its multiple functions it helps to activate resting monocytes and favours extravasation of neutrophils by opening tight junctions between human vascular endothelial cells and upregulating leucoyte adhesion molecules (LAM).26 Chloroquine diminishes TNFα production and downregulates the TNFα receptors 1 and 2 (TNFR) on the monocyte cell surface, which eventually results in decreasedmonocyte activation as well as decreased leucocyte extravasation. Red crosses mark the steps directly inhibited by chloroquine.
Safety considerations
The studies reviewed here show that chloroquine/ hydroxychloroquine has in-vitro antiviral effects and anti-inflammatory properties that may be of interest in those viral infections associated with inflammation and/or immune activation. Before analysing the potential effects of a drug on a disease, safety criteria have to be met, to estimate the risk/benefit ratio.
Chloroquine/hydroxychloroquie has a well-studied toxicity profile. The half-century-long use of this drug in the therapy of malaria demonstrates the safety of acute administration of chloroquine to human beings. The use of chloroquine/hydroxychloroquine in rheumatic diseases and for antimalarial prophylaxis showed a low incidence of adverse events during chronic administration of this drug for periods of up to a few years. In these cases, the most serious toxic effect is a macular retinopathy, which depends on the cumulative dose rather than on the daily dose, and permanent damage may be prevented with regular visual monitoring during treatment.27, 28, 29 A recent study30 provided encouraging results on the safety of a high dosage of the drug (up to 500 mg of chloroquine base per day) even during pregnancy.
We conclude that chloroquine/hydroxychloroquine administration presents limited and well-preventable toxicity and may thus result in a low risk/benefit balance at least when it is used in life-threatening conditions.
Henceforth, we will discuss the potential usefulness of this old drug in the treatment of two infectious diseases posing a serious threat to public health in the era of globalisationie, AIDS and severe acute respiratory syndrome (SARS). These diseases are both caused by enveloped RNA viruses, and share some clinical manifestations that are likely to be mediated by immune reactions of the host.
Effects on HIV infection
Anti-HIV effects of chloroquine
Under testing conditions intended to mimic as best as possible clinical situations, chloroquine/hydroxychloroquine is capable of inhibiting HIV in vitro. This ability was shown either by overloading the cells with high concentrations of chloroquine/hydroxychloroquine before the infection,17, 31 so as to mimic the drug build-up taking place in the tissues of patients subject to chronic treatment, or by keeping HIV-infected cells under constant incubation with concentrations of chloroquine detected in whole blood of individuals chronically treated with this drug.32, 33 The anti-HIV activity of chloroquine has been shown not only in cell line models, but also in peripheral blood lymphocytes and monocytes31, 33—ie, cell culture models in which cellular uptake of chloroquine is closer to the conditions occurring in vivo. Under these conditions, it was possible to obtain levels of inhibition of viral replication above 90%. That hydroxychloroquine has some antiviral activity in vivo has been reported by two phase II clinical trials.34, 35 The first trial was a small randomised, doubleblind, placebo-controlled pilot study on 40 patients (20 patients per arm), 27 of them being antiretroviral treatmentnaive. Hydroxychloroquine administration for 8 weeks resulted in a mean 0·6 log reduction in plasma HIV-1 RNA copy numbers (p=0·02) as well as in a decrease in interleukin 6 concentrations, whereas placebo did not have any effects on both HIV-1 RNA and interleukin 6.34 The second trial was also a small randomised, double-blinded trial, comparing the effectiveness of hydroxychloroquine with that of zidovudine monotherapy for 16 weeks in 72 patients, 64 of whom being antiretroviral treatment-naive (35 in the hydroxychloroquine arm and 37 in the zidovudine arm). Hydroxychloroquine again significantly reduced the plasma HIV-1 RNA copy numbers/mL (baseline 39 456 [31 000]; post-treatment 16 434 [11 373]; mean log reduction 0·4; p=0·02), though less than zidovudine (baseline 42 709 [33 050]; post-treatment 11 228 [7459]; mean log reduction 0·6; p=0·01). Since eight of 37 people in the zidovudine group, but none of the 35 individuals in the hydroxychloroquine group, showed an increase in the HIV-1 RNA levels and in the cultured virus levels during therapy, these data are consistent with the hypothesis that resistance to hydroxychloroquine may not easily develop as opposed to the well-known development of resistance to monotherapy with other antiretrovirals such as zidovudine.35 The results of larger clinical trials will be necessary for an accurate analysis of any possible discrepancies between the effects of chloroquine in vitro and in HIV-infected individuals.
Some of us have recently shown that chloroquine, at nontoxic, clinically achievable concentrations, has in-vitro activity against primary isolates belonging to different HIV-1 and HIV-2 clades.33 The mechanism of the anti-HIV effects of chloroquine/hydroxychloroquine is a reduction in the infectivity of newly produced virions (reviewed in Savarino et al2). The antiviral effects of chloroquine are associated with the reduced production of the heavily glycosylated epitope 2G12, which is located on the gp120 envelope glycoprotein surface and is fundamental for virus infectivity.33 These effects are likely to be attributed to the increased pH in TGN, which impairs the function of glycosyl-transferases involved in the post-translational processing of the HIV glycoproteins.2, 17, 33 HIV glycosylation may therefore represent a new target for antiretroviral therapy. As viral envelope glycosylation is mediated by cellular enzymes, its inhibition may explain the broad spectrum of the in-vitro anti-HIV activity of chloroquine against all major subtypes of HIV-1 and HIV-2.33 The effect of chloroquine/hydroxychloroquine on cellular rather than viral enzymes may also result in a low propensity to resistance development.
Effects of chloroquine in combination with other antiretrovirals
As chloroquine/hydroxychloroquine probably inhibits viral replication by a mechanism different from those of currently used antiretroviral drugs, its application has been studied in combination with other antiretroviral drugs. The use of chloroquine in combination with other antiretrovirals is theoretically supported by the observation that chloroquine also shows anti-HIV activity in vitro towards isolates from patients in therapeutic failure with a multidrug-resistant profile.36
First, hydroxychloroquine has an additive in vitro anti-HIV effect to that of zidovudine.37 Second, chloroquine exerts in vitro an additive anti-HIV-1 effect on the combinations of hydroxyurea plus didanosine or hydroxyurea plus zidovudine in T-cell lines, monocytes, and primary T-cells.2, 38, 39 The didanosine/hydroxyurea/hydroxychloroquine combination, especially attractive for developing countries due to its low cost, was tested clinically in Singapore in an open study. Of the initial 22 patients who started the study, six were withdrawn due to non-compliance. In the remaining 16 patients, HIV-1 RNA plasma levels decreased by a mean of 1·3 log at week 12 and 48.40 In a smaller study, with didanosine/hydroxyurea/chloroqiune, HIV-1 RNA remained lowered by a mean of 2·5 log after more than 96 weeks.41 These open pilot studies do not yet allow a determination of the contribution of chloroquine/hydroxychloroquine to the viral load drop. However, it can be concluded that the addition of chloroquine/hydroxychloroquine to hydroxyurea and didanosine is potentially safe, thus encouraging the design of larger studies with multiple arms.
Prevention of HIV transmission via breastfeeding
It is possible to hypothesise that chloroquine may find a potential application in prevention of mother-to-child transmission (MTCT) of HIV through breastfeeding, a problem still far from being solved in resource-poor countries.42 One of us reported a 243-fold accumulation of chloroquine in colostrum cells of Burkinabà mothers taking 100 mg of chloroquine daily and hypothesised that such a high degree of chloroquine accumulation in mammary cells actively replicating HIV-1 may allow a decrease in milk viral load and/or infectivity of milk and, hence, may lower the risk of breastfeeding-related transmission,43 which accounts for one-third to one-half of HIV-1 MTCTs.44 Therefore the CHARGE (Chloroquine Administration to Reduce HIV-1 Genome Exposure) study was set up, being a placebocontrolled pilot study in breastfeeding mothers (having received, together with their infant, peripartum nevirapine), that will assess whether daily chloroquine administration compared with placebo administration to the mother during the early months of breastfeeding may result in decreased HIV-1 RNA milk levels and/or decreased ex vivo infectivity of virions isolated from milk.
Decrease of excessive immune activation
The chloroquine/hydroxychloroquine-induced suppression of the synthesis of proinflammatory cytokines such as TNFα may be beneficial in decreasing the inappropriate immune activation characteristic of HIV infection. This may be particularly useful to people with HIV/AIDS in the developing world, who, probably due to concomitant infections, present higher levels of TNFα production and of immune activation than those from the developed world.45, 46, 47 Chloroquine/hydroxychloroquine should be studied, particularly in areas with high prevalence of HIV-1C, such as southern Africa. Indeed, when TNFα is used for in-vitro stimulation, the rate of transcriptional activation of HIV-1 is higher for HIV-1C than for HIV-1B and HIV-1 CRF_1AE. Such an enhanced capacity to respond to TNFα may be related to the extra NF-kappaB binding site(s) present in the long terminal repeat of the HIV-1C genome.48
Hypothesis: the case of SARS
Based on the effects of chloroquine/hydroxychloroquine on several enveloped viruses and on immune activation, we raise the hypothesis that this drug might be of some use for the clinical management of SARS. At present, any attempt to treat this disease with known antiviral drugs—namely ribavirin and oseltamivir—has been inconclusive.49 Corticosteroids may be of some benefit in controlling the inflammatory response at the lung level50 but may also cause uncontrolled immunodepression resulting in pulmonary superinfection.
The causative agent of SARS has recently been described as a new coronavirus.51, 52 Recent studies support the idea that coronaviridae infect their target cells by an endocytic pathway and that chloroquine might inhibit their replication.53, 54 Cells infected with the human coronavirus HCoV-229E and treated with nocodazole (a microtubuledepolymerising agent that blocks transport from early to late endosomes) produced decreased amounts of HCoV-229E antigens.53 This result indicates that endosomal transport is needed for HCoV-229E infection. Cells treated with chloroquine expressed decreased amounts of HCoV-229E antigens.53 Preliminary data obtained from our group confirm these reports and show that chloroquine potently inhibits the replication of a canine coronavirus at therapeutically reachable concentrations (C Buonavoglia et al, University of Bari, Italy; unpublished). Although the SARS coronavirus is distinct with unique characteristics, it is tempting to ask whether chloroquine might affect SARS coronavirus replication as well.
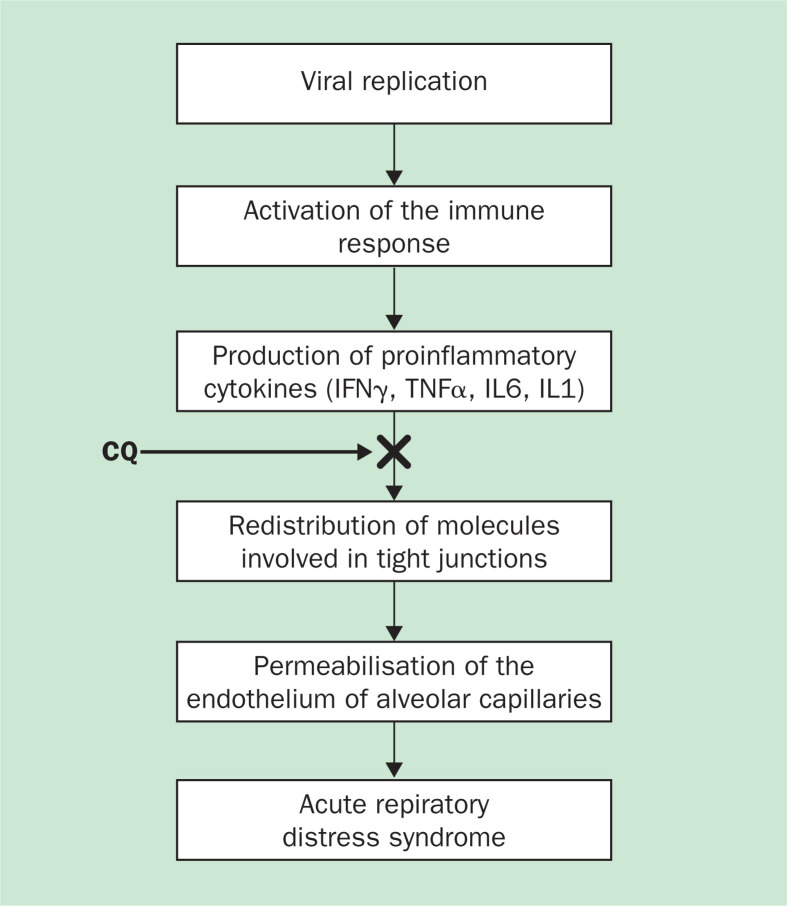
The anti-inflammatory properties of chloroquine/hydroxychloroquine should also be considered. The clinical worsening of individuals with SARS in week 2 is apparently unrelated to uncontrolled SARS coronavirus replication but may be related to immunopathological damage.55 A model taking into account the role of proinflammatory cytokines could help interpret this event (figure 3 ). This view is derived from the effects of the porcine respiratory coronavirus (PRCV), which shares with the SARS coronavirus the ability to cause a disease with similar histopathological features and symptomatology.57 PRCV induces severe lung damage through immune-mediated mechanisms—ie, probably through an increase in the concentrations of proinflammatory cytokines such as TNFα and interleukin 6, whose role in inducing lung damage has been proved using adenoviral vectors in animal models.58 On these grounds, we think that the associations between TNFα and interleukin 6 concentrations and disease severity should also be tested in stored samples from human patients with SARS. If confirmatory results are obtained, then, it would be reasonable to consider chloroquine/hydroxychloroquine to suppress TNFα and interleukin 6 production. For this purpose, efforts to develop an animal model for SARS would be welcome. Such a model would help to clarify the immunemediated component of the symptoms of the disease as well as in testing of chloroquine and other immunomodulatory drugs. These studies could also lay the groundwork for the suggestion that chloroquine be considered for the treatment of other viral infections which involve immunopathology.
Figure 3.
Hypothetical model for the potential effects of chloroquine (CQ) on the immunopathogenesis of severe acute respiratory syndrome (SARS). Proinflammatory cytokines are thought to be important in acute respiratory distress syndrome (ARDS).56 We hypothesise that chloroquine (black arrow), by inhibiting TNFα and interleukin 6 (IL6) production, might block the subsequent cascade of events, which leads to ARDS.
Conclusions
At present it is difficult to answer the question of whether old chloroquine will be able to live a “second youth”. Due to its main effect—ie, raising endosomal pH—the drug has an exceptionally broad spectrum of antimicrobial activity that could be exploited in many infections. Results obtained in the prophylaxis of Q fever indicate that chloroquine/hydroxychloroquine can be successfully used in the clinical management of infections other than malaria.59 As regards viral diseases, what is clear is that the drug has antiviral and immunomodulatory effects that warrant particular consideration.
The effects on HIV infection are the best studied among the antiviral effects of chloroquine/hydroxychloroquine and are being tested in clinical trials. The anti-HIV effect of chloroquine/hydroxychloroquine is modest by itself but is, at least in vitro, additive or synergistic when combined with selected antiretrovirals. These in-vitro results warrant in-vivo confirmation in HIV-positive individuals by comparing the long-term antiviral effect of highly active antiretroviral therapy regimens with and without chloroquine or hydroxychloroquine. If such studies provide encouraging results, they may lead to a strategy whereby co-administration of chloroquine/hydroxychloroquine allows lower doses of antiretrovirals, lessening cost and possibly toxicity. Costlessening would of course be particularly welcome in developing countries. Moreover, the potential ability of chloroquine to inhibit the replication of drug-resistant viral isolates could be important in the treatment of drugexperienced HIV-positive patients who have developed multiple resistance to antiretroviral drugs and thus have limited therapeutic options. In the developing countries where malaria is endemic, the concomitant administration of chloroquine/hydroxychloroquine to a highly active antiretroviral therapy regimen may result (at least where P falciparum is still susceptible to chloroquine) in a decreased incidence of malaria episodes that have adverse effects in HIV-infected people, especially during pregnancy.60 Moreover, the anti-inflammatory properties of chloroquine may temper the noxious immune hyperactivation that is characteristic of HIV/AIDS.61
Due to its broad spectrum of antiviral activity as well as to its suppressive effects on the production/release of TNFα and interleukin 6, chloroquine/hydroxychloroquine may also find a place in the treatment of other viral infections characterized by symptoms associated with inflammatory processes and/or immune-hyperactivation. We believe that further study should be devoted to the inhibitory effects of chloroquine on the infectivity of flaviviruses, as one of the members of this family, the hepatitis C virus, is of great importance for human pathology and often co-infects individuals with HIV-1. Flaviviridae also include several arthropod-borne viruses such as the yellow fever virus and the West Nile virus, which has recently caused an epidemic in North America.
Finally, we want to share with the scientific community the speculative hypothesis that chloroquine/hydroxychloroquine, due to its antiviral and anti-inflammatory properties, may have some effect on SARS. We emphasise the need of testing in cell cultures infected with SARS coronavirus the effects of chloroquine, as well as those ofother substances possessing in-vitro activity against members of the coronaviridae family. We should remember that the possibility of new outbreaks of SARS cannot be excluded. In the absence of effective inhibitors of SARS coronavirus, the possibility of an inhibition, at least in vitro, of the replication of this virus would represent a breakthrough in the knowledge of SARS.
Search strategy and selection criteria
We did literature reviews using relevant Medline search terms, in various combinations—“chloroquine”, “virus”, “retrovirus”, “TNFα”, “cytokines”, “endothelium”, “macrophages”, “HIV” “SARS”, “NF-kappa B”, “coronavirus”, “proinflammatory cytokines”, and “ARDS”—screened articles for relevance, and reviewed references. Because of space restrictions we (1) cited review articles in place of original manuscripts where possible, (2) excluded those articles whose content was not in line with the bulk of the published information without giving convincing explanations, and (3) ranked the articles on the basis of relevance, date of publication, and journal impact factor. We also searched the internet for relevant web pages, discarded those edited by people/institutions not directly involved in research, and reviewed references. References are not intended to provide a comprehensive listing, but were chosen to best highlight the critical issues.
Acknowledgments
Acknowledgments
We are grateful to Special Project AIDS, Istituto Superiore di Sanitα, Rome, Italy, for financial support.
Conflicts of interest
We have no conflicts of interest.
References
- 1.Canadian rheumatology association Canadian Consensus Conference on hydroxychloroquine. J Rheumatol. 2000;27:2919–2921. [PubMed] [Google Scholar]
- 2.Savarino A, Gennero L, Sperber K, Boelaert JR. The anti-HIV-1 activity of chloroquine. J Clin Virol. 2001;20:131–135. doi: 10.1016/s1386-6532(00)00139-6. [DOI] [PubMed] [Google Scholar]
- 3.Boelaert JR, Piette J, Sperber K. The potential place of chloroquine in the treatment of HIV-1–infected patients. J Clin Virol. 2001;20:137–140. doi: 10.1016/s1386-6532(00)00140-2. [DOI] [PubMed] [Google Scholar]
- 4.Ohkuma S, Poole B. Cytoplasmic vacuolation of mouse peritoneal macrophages and the uptake into lysosomes of weakly basic substances. J Cell Biol. 1981;90:656–664. doi: 10.1083/jcb.90.3.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pescarmona GP, Morra E, Aldieri E, Ghigo D, Bosia A. Movements of vesicles in eukaryotic cells: role of intravesicle protons as a fuel and modulation of their concentration by drugs or metabolic changes. MRS Bull. 1998;489:212–217. [Google Scholar]
- 6.Vezmar M, Georges E. Reversal of MRP-mediated doxorubicin resistance with quinoline-based drugs. Biochem Pharmacol. 2000;59:1245–1252. doi: 10.1016/s0006-2952(00)00270-7. [DOI] [PubMed] [Google Scholar]
- 7.Vezmar M, Georges E. Direct binding of chloroquine to the multidrug resistance protein (MRP): possible role for MRP in chloroquine drug transport and resistance in tumor cells. Biochem Pharmacol. 1998;56:733–742. doi: 10.1016/s0006-2952(98)00217-2. [DOI] [PubMed] [Google Scholar]
- 8.Byrd TF, Horwitz MA. Chloroquine inhibits the intracellular multiplication of Legionella pneumophila by limiting the availability of iron. A potential new mechanism for the therapeutic effect of chloroquine against intracellular pathogens. J Clin Invest. 1991;88:351–357. doi: 10.1172/JCI115301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Legssyer R, Josse C, Piette J, Ward RJ, Crichton RR. Changes in function of iron-loaded alveolar macrophages after in vivo administration of desferrioxamine and/or chloroquine. J Inorg Biochem. 2003;94:36–42. doi: 10.1016/s0162-0134(02)00633-5. [DOI] [PubMed] [Google Scholar]
- 10.Gonzalez-Dunia D, Cubitt B, de la Torre JC. Mechanism of Borna disease virus entry into cells. J Virol. 1998;72:783–788. doi: 10.1128/jvi.72.1.783-788.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ros C, Burckhardt CJ, Kempf C. Cytoplasmic trafficking of minute virus of mice: low-pH requirement, routing to late endosomes, and proteasome interaction. J Virol. 2002;76:12634–12645. doi: 10.1128/JVI.76.24.12634-12645.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Diaz-Griffero F, Hoschander SA, Brojatsch J. Endocytosis is a critical step in entry of subgroup B avian leukosis viruses. J Virol. 2000;76:12866–12876. doi: 10.1128/JVI.76.24.12866-12876.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bishop NE. Examination of potential inhibitors of hepatitis A virus uncoating. Intervirology. 1998;41:261–271. doi: 10.1159/000024948. [DOI] [PubMed] [Google Scholar]
- 14.Ferreira DF, Santo MP, Rebello MA, Rebello MC. Weak bases affect late stages of Mayaro virus replication cycle in vertebrate cells. J Med Microbiol. 2000;49:313–318. doi: 10.1099/0022-1317-49-4-313. [DOI] [PubMed] [Google Scholar]
- 15.Harley CA, Dasgupta A, Wilson DW. Characterization of herpes simplex virus-containing organelles by subcellular fractionation: role for organelle acidification in assembly of infectious particles. J Virol. 2001;75:1236–1251. doi: 10.1128/JVI.75.3.1236-1251.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Randolph VB, Winkler G, Stollar V. Acidotropic amines inhibit proteolytic processing of flavivirus prM protein. Virology. 1990;174:450–458. doi: 10.1016/0042-6822(90)90099-d. [DOI] [PubMed] [Google Scholar]
- 17.Tsai WP, Nara PL, Kung HF, Oroszlan S. Inhibition of human immunodeficiency virus infectivity by chloroquine. AIDS Res Hum Retroviruses. 1990;6:481–489. doi: 10.1089/aid.1990.6.481. [DOI] [PubMed] [Google Scholar]
- 18.Baughman RP, Lower EE, du Bois RM. Sarcoidosis. Lancet. 2003;361:1111–1118. doi: 10.1016/S0140-6736(03)12888-7. [DOI] [PubMed] [Google Scholar]
- 19.Jeong J-Y, Jue D-M. Chloroquine inhibits processing of tumor necrosis factor in lipopolysaccharidestimulated RAW 264·7 macrophages. J Immunol. 1997;158:4901–4907. [PubMed] [Google Scholar]
- 20.Bondeson J, Sundler R. Antimalarial drugs inhibit phospholipase A2 activation and induction of interleukinβ and tumor necrosis factor α in macrophages. General Pharmacol. 1998;30:357–366. doi: 10.1016/s0306-3623(97)00269-3. [DOI] [PubMed] [Google Scholar]
- 21.Van den Borne BE, Dijkmans BA, de Rooij HH, le Cessie S, Verweij CL. Chloroquine and hydroxychloroquine equally affect tumor necrosis factor-α, interleukin 6 and interferon-γ production by peripheral blood mononuclear cells. J Rheumatol. 1997;24:55–60. [PubMed] [Google Scholar]
- 22.Karres I, Kremer J-P, Dietl I, Steckholzer U, Jochum M, Ertel W. Chloroquine inhibits proinflammatory cytokine release into human whole blood. Am J Physiol. 1998;274:R1058–R1064. doi: 10.1152/ajpregu.1998.274.4.R1058. [DOI] [PubMed] [Google Scholar]
- 23.Picot S, Peyron F, Donadille A, Vuillez J-P, Barbe G, Ambroise-Thomas P. Chloroquine-induced inhibition of the production of TNF, but not of IL-6, is affected by disruption of iron metabolism. Immunology. 1993;80:127–133. [PMC free article] [PubMed] [Google Scholar]
- 24.Weber SM, Levitz SM. Chloroquine interferes with lipopolysaccharide-induced TNF-α gene expression by a nonlysosomotropic mechanism. J Immunol. 2000;165:1534–1540. doi: 10.4049/jimmunol.165.3.1534. [DOI] [PubMed] [Google Scholar]
- 25.Jeong J-Y, Choi JW, Jeon K-I, Jue D-M. Chloroquine decreases cell-surface expression of tumour necrosis factor receptors in human histiocytic U-937 cells. Immunol. 2002;105:83–91. doi: 10.1046/j.0019-2805.2001.01339.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nooteboom A, Hendriks T, Otteholler I, van der Linden CJ. Permeability characteristics of human endothelial monolayers seeded on different extracellular matrix proteins. Mediators Inflamm. 2000;9:235–241. doi: 10.1080/09629350020025755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bernstein HN. Ocular safety of hydroxychloroquine. Ann Ophthalmol. 1991;23:292–296. [PubMed] [Google Scholar]
- 28.Bernstein HN. Ophthalmologic considerations and testing in patients receiving long-term antimalarial therapy. Am J Med. 1983;75:25–34. doi: 10.1016/0002-9343(83)91267-6. [DOI] [PubMed] [Google Scholar]
- 29.Herman K, Leys A, Spileers W. (Hydroxy)-chloroquine retinal toxicity: two case reports and safety guidelines. Bull Soc Belge Ophtalmol. 2002;284:21–29. [PubMed] [Google Scholar]
- 30.Klinger G, Morad Y, Westall CA. Ocular toxicity and antenatal exposure to chloroquine or hydroxychloroquine for rheumatic diseases. Lancet. 2001;358:813–814. doi: 10.1016/S0140-6736(01)06004-4. [DOI] [PubMed] [Google Scholar]
- 31.Sperber K, Kalb TH, Stecher VJ, Banerjee R, Mayer L. Inhibition of human immunodeficiency virus type 1 replication by hydroxychloroquine in T cells and monocytes. AIDS Res Hum Retroviruses. 1993;9:91–98. doi: 10.1089/aid.1993.9.91. [DOI] [PubMed] [Google Scholar]
- 32.Pardridge WM, Yang J, Diagne A. Chloroquine inhibits HIV-1 replication in human peripheral blood lymphocytes. Immunol Lett. 1998;64:45–47. doi: 10.1016/s0165-2478(98)00096-0. [DOI] [PubMed] [Google Scholar]
- 33.Savarino A, Gennero L, Chen HC. Anti-HIV effects of chloroquine: mechanisms of inhibition and spectrum of activity. AIDS. 2001;15:2221–2229. doi: 10.1097/00002030-200111230-00002. [DOI] [PubMed] [Google Scholar]
- 34.Sperber K, Louie M, Kraus T. Hydroxychloroquine treatment of patients with human immunodeficiency virus type 1. Clin Ther. 1995;17:622–636. doi: 10.1016/0149-2918(95)80039-5. [DOI] [PubMed] [Google Scholar]
- 35.Sperber K, Chiang G, Chen H. Comparison of hydroxychloroquine with zidovudine in asymptomatic patients infected with HIV-1. Clin Ther. 1997;19:913–923. doi: 10.1016/s0149-2918(97)80045-8. [DOI] [PubMed] [Google Scholar]
- 36.Debiaggi M, Bruno R, Sacchi P, Filice G. Antiviral activity of chloroquine against HIV-1 strains resistant to antiretroviral drugs. Antiviral Res (in press)
- 37.Chiang G, Sassaroli M, Louie M, Chen H, Stecher VJ, Sperber K. Inhibition of HIV-1 replication by hydroxychloroquine: mechanism of action and comparison with zidovudine. Clin Ther. 1996;18:1080–1092. doi: 10.1016/s0149-2918(96)80063-4. [DOI] [PubMed] [Google Scholar]
- 38.Boelaert JR and Sperber K. Antiretroviral therapy. Lancet. 1998;352:1224–1225. doi: 10.1016/S0140-6736(05)60566-1. [DOI] [PubMed] [Google Scholar]
- 39.Boelaert JR, Sperber K, Piette J. Chloroquine exerts an additive in vitro anti-HIV-1 effect, when combined to zidovudine and hydroxyurea. Biochem Pharmacol. 2001;61:1531–1535. doi: 10.1016/s0006-2952(01)00576-7. [DOI] [PubMed] [Google Scholar]
- 40.Paton NI, Aboulhab J, Karim F. Hydroxychloroquine, hydroxycarbamide, and didanosine as economic treatment for HIV-1. Lancet. 2002;359:1667–1668. doi: 10.1016/S0140-6736(02)08557-4. [DOI] [PubMed] [Google Scholar]
- 41.Boelaert JR, Dom GM, Huitema AD, Beijnen JH, Lange JM. The boosting of didanosine by allopurinol permits a halving of the didanosine dosage. AIDS. 2002;16:2221–2223. doi: 10.1097/00002030-200211080-00016. [DOI] [PubMed] [Google Scholar]
- 42.Mofenson LM, McIntyre JA. Advances and research directions in the prevention of mother-to-child HIV-1 transmission. Lancet. 2000;355:2237–2244. doi: 10.1016/S0140-6736(00)02415-6. [DOI] [PubMed] [Google Scholar]
- 43.Boelaert JR, Yaro S, Augustijns P. Chloroquine accumulates in breast milk cells. Potential impact as adjuvant to antiretroviral prophylaxis for postnatal mother-to-child transmission of HIV-1. AIDS. 2001;15:2205–2206. doi: 10.1097/00002030-200111090-00024. [DOI] [PubMed] [Google Scholar]
- 44.DeCock KM, Fowler MG, Mercier E. Prevention of mother-to-child transmission of HIV-1 in resource-poor countries: translating research into policy and practice. JAMA. 2000;283:1175–1182. doi: 10.1001/jama.283.9.1175. [DOI] [PubMed] [Google Scholar]
- 45.Corbett EL, Steketee RW, ter Kuile FO, Latif AS, Kamali A, Hayes RJ. HIV-1/AIDS and the control of other infectious diseases in Africa. Lancet. 2002;359:2177–2187. doi: 10.1016/S0140-6736(02)09095-5. [DOI] [PubMed] [Google Scholar]
- 46.Clerici M, Butto S, Lukwiya M. Immune activation in Africa is environmentally driven and is associated with upregulation of CCR5. AIDS. 2000;14:2083–2092. doi: 10.1097/00002030-200009290-00003. [DOI] [PubMed] [Google Scholar]
- 47.Bentwich Z, Maartens G, Torten D, Lal AA, Lal RB. Concurrent infections and HIV pathogenesis. AIDS. 2000;14:2071–2081. doi: 10.1097/00002030-200009290-00002. [DOI] [PubMed] [Google Scholar]
- 48.Montano MA, Nixon CP, Ndung'u T. Elevated tumor necrosis factor-α activation of human immunodeficiency virus subtype 1 in southern Africa is associated with an NF-kB enhancer gain-of-function. J Infect Dis. 2000;181:76–81. doi: 10.1086/315185. [DOI] [PubMed] [Google Scholar]
- 49.Poutanen SM, Low DE, Henry B. Identification of severe acute respiratory syndrome in Canada. N Engl J Med. 2003;348:1995–2005. doi: 10.1056/NEJMoa030634. [DOI] [PubMed] [Google Scholar]
- 50.So LK, Lau AC, Yam LY. Development of a standard treatment protocol for severe acute respiratory syndrome. Lancet. 2003;361:1615–1617. doi: 10.1016/S0140-6736(03)13265-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Peiris JS, Lai ST, Poon LL. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319–1325. doi: 10.1016/S0140-6736(03)13077-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Drosten C, Gunther S, Preiser W. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967–1976. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- 53.Blau D, Holmes K. Human Coronavirus HCoV-229E enters susceptible cells via the endocytic pathway. In: Lavi E, editor. The Nidoviruses, Coronaviruses and Arteriviruses. Kluwer; New York: 2001. pp. 193–197. [DOI] [PubMed] [Google Scholar]
- 54.Nauwynck HJ, Duan X, Favoreel HW, Van Oostveldt P, Pensaert MB. Entry of porcine reproductive and respiratory syndrome virus into porcine alveolar macrophages via receptor-mediated endocytosis. J Gen Virol. 1999;80(Pt 2):297–305. doi: 10.1099/0022-1317-80-2-297. [DOI] [PubMed] [Google Scholar]
- 55.Peiris JS, Chu CM, Cheng VC. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shanley TP, Warner RL, Ward PA. The role of cytokines and adhesion molecules in the development of inflammatory injury. Mol Med Today. 1995;1:40–45. doi: 10.1016/1357-4310(95)80019-0. [DOI] [PubMed] [Google Scholar]
- 57.Nicholls JM, Poon LL, Lee KC. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003;361:1773–1778. doi: 10.1016/S0140-6736(03)13413-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Van Reeth K, Van Gucht S, Pensaert M. In vivo studies on cytokine involvement during acute viral respiratory disease of swine: troublesome but rewarding. Vet Immunol Immunopathol. 2002;87:161–168. doi: 10.1016/S0165-2427(02)00047-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fenollar F, Fournier PE, Carrieri MP, Habib G, Messana T, Raoult D. Risks factors and prevention of Q fever endocarditis. Clin Infect Dis. 2001;33:312–316. doi: 10.1086/321889. [DOI] [PubMed] [Google Scholar]
- 60.Ladner J, Leroy V, Karita E, van de Perre P, Dabis F. Malaria, HIV and pregnancy. AIDS. 2003;17:275–356. doi: 10.1097/00002030-200301240-00025. [DOI] [PubMed] [Google Scholar]
- 61.Savarino A, Bottarel F, Malavasi F, Dianzani U. Role of CD38 in HIV-1 infection: an epiphenomenon of T-cell activation or an active player in virus/host interactions? AIDS. 2000;14:1079–1089. doi: 10.1097/00002030-200006160-00004. [DOI] [PubMed] [Google Scholar]