Abstract
The Iberian lynx, (Lynx pardinus), is the most endangered felid in the world. To determine whether sympatric carnivores are reservoirs of pathogens posing a disease risk for the lynx, evidence of exposure to 17 viral, bacterial and protozoan agents was investigated in 176 carnivores comprising 26 free-living lynx, 53 domestic cats, 28 dogs, 33 red foxes (Vulpes vulpes), 24 Egyptian mongooses (Herpestes ichneumon), 10 common genets (Genetta genetta) and 2 Eurasian badgers (Meles meles) in the areas inhabited by the last two populations of Iberian lynx, both in Andalusia (South-Western Spain).
The results indicated that the lynx had low rates of contact with viral pathogens, with one seropositive finding each for feline leukemia virus, parvovirus and canine adenovirus-1, whereas contact with bacteria and protozoa appeared more frequent. Active infections with parvovirus, Ehrlichia spp., Mycobacterium bovis, Leptospira interrogans and Cytauxzoon spp. were confirmed. In contrast, 53% of the domestic cats were exposed to some infectious agent (prevalence range 4.5–11.4%). Antibodies to canine distemper virus and parvovirus were frequently found in dogs (32% and 42%, respectively) and foxes (30% and 12%). Past or present infections with parvovirus, Ehrlichia spp., Chlamydophila spp., M. bovis, Salmonella enterica, L. interrogans, Toxoplasma gondii, and Neospora caninum were also detected in these and other species surveyed.
Questionnaires to owners revealed that 14% of the dogs but none of the cats had been vaccinated, and no cat had been neutered. Based on the apparent absence of acquired immunity of the lynx against infectious agents, the frequent detection of agents among sympatric carnivores, and the reported lack of immunocompetence of the Iberian lynx, a disease outbreak among the local abundant carnivores may pose a serious disease risk for lynx conservation.
Keywords: Domestic carnivores, Felid, Feline leukemia virus, Maintenance host, Transmission, Spillover
Introduction
Epidemics can be a serious conservation threat for free-living populations of endangered species (Thorne and Williams, 1988, Roelke-Parker et al., 1996) and can cause mortality, reduce host fitness and/or alter dispersal and movement patterns of infected animals (Scott, 1988). As any of these outcomes may have dire consequences for the persistence of small populations, monitoring the prevalence of disease should be a priority in conservation (Scott, 1988).
Menaced species are less likely to sustain infectious agents because intra-species interactions are infrequent, and the epidemiology of a disease is driven not by intra-species pathogen transmission but by transmission from reservoir species that are able to maintain a relatively high pathogen population (Power and Mitchell, 2004). For example, high-density domestic dog populations are a more likely source of canine distemper virus (CDV) for infected African carnivores (Cleaveland et al., 2000). Moreover, because most individuals in an endangered population are seldom exposed to a pathogen, there is little acquired immunity, so when an epidemic occurs it tends to infect a large proportion of the population and mortality levels may be high (McCallum and Dobson, 1995).
The Iberian lynx (Lynx pardinus) is the most endangered felid in the world (Nowell and Jackson, 1996) with only about 160–200 individuals remaining (Palomares et al., 2003, Guzmán et al., 2004). In spite of the serious consequences that an epidemic might have on the two remaining lynx populations, very little is known about diseases affecting the species. There is little evidence of contact with microparasites other than Toxoplasma gondii (Peña et al., 2006, Roelke et al., 2008). However, the lynx is susceptible to bovine tuberculosis (TB) (Pérez et al., 2001, Aranaz et al., 2004), and is known to host the protozoal blood parasite Cytauxzoon spp. (Luaces et al., 2005). Roelke et al. (2008) speculated that the lack of evidence of exposure of Iberian lynx to pathogens such as feline immunodeficiency virus (FIV), feline leukemia virus (FeLV) or CDV might make the lynx vulnerable to outbreaks of these diseases in the future. The lynx has reportedly experienced demographic bottlenecks and associated losses of genetic diversity that may exacerbate such vulnerability (Johnson et al., 2004). Inbreeding was recently proposed as a cause of the loss of effectiveness of the immune system in the lynx (Peña et al., 2006).
The Iberian lynx coexists with other abundant wild and domestic carnivore species that may potentially act as reservoirs of disease. Many urban areas are close to or within areas inhabited by the lynx, so favouring disease transmission between domestic animals and wildlife (Bradley and Altizer, 2007, Whiteman et al., 2007). However, there is no information about infectious diseases affecting other carnivores in these areas other than TB (Martín-Atance et al., 2006). Sympatric carnivores act as reservoirs of helminth and arthropod species parasitizing the Iberian lynx (Millán and Casanova, 2007, Millán et al., 2007b).
Our aims in this study were to describe the prevalence of selected disease agents in the Iberian lynx and sympatric carnivore species, and to determine whether these carnivores and/or other species might act as reservoir hosts.
Materials and methods
Study areas
The study was carried out in the areas inhabited by the last two populations of Iberian lynx, both in Andalusia (South-Western Spain, Fig. 1 ). Sierra Morena (38°13′ N, 4°10′W; 1125 km2; 120–140 lynx) is a hilly area with private hunting estates within protected areas. There are many villages in and around the lynx areas. Ranch houses are frequent, and numerous chalets (Viñas), some permanently inhabited, are scattered in the South-Eastern part of the area. Doñana (37°0′N, 6°30′W; 2000 km2; 30–50 lynx) includes the Doñana National Park (DNP) and Doñana Natural Park (Dnp). It borders along 4 km with the village of Matalascañas, which receives some 100,000 holidaymakers each summer. There are other villages in and around lynx areas, and abundant chalets are also located close to the West side of DNP.
Fig. 1.

Map of the study areas where Iberian lynx and other sympatric carnivores were studied for exposure against infectious and protozoan pathogens. Approximate lynx distributions areas are adapted from Guzmán et al. (2004). Locations of samples with exact known origin are shown with symbols representing the different species surveyed. Sample size is given into the symbols.
Dogs and cats are widespread in both lynx areas. Shepherding and guard dogs and free-roaming cats are found in private residences and ranch houses. Hunting dogs, including large dog packs used for big game (‘rehalas’), often move across the Sierra Morena hunting estates and into Dnp during the hunting season. Feral dogs and cats can be frequently seen in both areas, including within DNP. Wild carnivores, especially the red fox (Vulpes vulpes) and the Egyptian mongoose (Herpestes ichneumon), but also the common genet (Genetta genetta), and the Eurasian badger (Meles meles) are common in both areas (see Palomo and Gisbert, 2002, and references therein for estimated densities, mainly for the Doñana area).
Sampling and laboratory analyses
From June 2004 to June 2006, we sampled 176 carnivores, including 26 free-living lynx, 53 cats, and 28 dogs (in both areas), and also (but only in Doñana) 33 foxes, 24 mongooses, 10 genets, and 2 badgers. We examined animals dead (mostly road-killed or hunted) or alive (Table 1 ).
Table 1.
Sample size and origin of the animals sampled for diseases surveillance in the Iberian lynx distribution areas (Spain) during 2004–2006
| Sierra Morena |
Doñana |
Total | |||||
|---|---|---|---|---|---|---|---|
| In vivo | Post-mortem | Total | In vivo | Post-mortem | Total | ||
| Iberian lynx Lynx pardinus | 9 | 3 | 11 | 13 | 5 | 15 | 26 |
| Domestic cat Felis catus | 20 | 10 | 30 | 10 | 13 | 23 | 53 |
| Dog Canis familiaris | 19 | 19 | 9 | 9 | 28 | ||
| Red fox Vulpes vulpes | 17 | 21 | 33 | 33 | |||
| Egyptian mongoose Herpestes ichneumon | 21 | 9 | 24 | 24 | |||
| Common genet Genetta genetta | 7 | 3 | 10 | 10 | |||
| Eurasian badger Meles meles | 2 | 2 | 2 | ||||
Some animals were surveyed alive and later subjected to post-mortem examination.
The wild carnivores and feral cats were caught in box-traps, and anaesthetised with a combination of ketamine (Imalgène, Merial) with medetomidine (Domtor, Salud Animal-Pfizer) before sampling. We did not sedate dogs and only blood was taken. Blood was obtained by cephalic or jugular venepuncture, collected in serum separator tubes and allowed to clot, and then centrifuged at 50 g for 15 min. The serum was removed and frozen at −20 °C. One millilitre of whole blood was placed in lithium-heparin-coated tubes. Swab samples using specific media for viruses, Chlamydia and bacteria were taken from the oro-pharyngeal cavity, ocular conjunctiva and anus. Thin smears from tonsil scrapings were prepared on glass slides for immunofluorescence analysis, fixed by immersion in acetone for 10 min and frozen at −40 °C. Urine and faeces were obtained whenever possible, kept in sterile tubes, and frozen at −20 °C.
Dead animals were subjected to detailed post-mortem examination. Samples of the main tissues and blood from cardiac puncture were obtained and frozen at −80 °C. Some feral cats and foxes, captured during predator control programmes in DNP, were anaesthetised, routinely sampled, humanely euthanised with sodium thiopental (B. Braun Medical), and necropsied. Evidence of active or past contact with 17 agents was studied using a variety of methods (Table 2 ). Vaccinated dogs were not included with respect to the agent(s) against which they were vaccinated.
Table 2.
Summary of methodologies used to detect evidence of contact with disease agents in carnivores in the Iberian lynx distribution areas during 2004–2006, showing samples analysed, threshold for serology (if quantitative), sequences used for polymerase chain reaction (PCR), conjugates used for immunofluorescence, and media used for culture
| Agenta | Sampleb | Assayc | Detectsd | Name of commercial kits, positive cut-off (serology), sequences (PCR), conjugates (FA), and media (culture) | Manufacturers (in parenthesis) and/or references |
|---|---|---|---|---|---|
| FCV | Serum | cELISA | c | F1008-AB02 Feline Calicivirus ELISA | Eurovet Veterinaria |
| FHV-1 | Serum | cELISA | c | F107-AB02 Feline Herpes ELISA | Eurovet Veterinaria |
| Oro-pharyngeal swab | FA | ai | Feline serum anti-FHV-1 | Eurovet Veterinaria | |
| 210-41-FHV/FVR | VMRD | ||||
| FCoV | Serum | cELISA | c | Ingezim Corona Felino | Ingenasa |
| Enteric mucosa, mesenteric LN, faeces | FA | ai | Feline serum anti-FCoV | Ingenasa | |
| Anti-felineIgG FITC conjugate | VMRD | ||||
| RIC | ai | FCoVMegacor (FB995) | VMRD | ||
| FeLV | Serum | cELISA | c | Ingezim FeLV | Ingenasa |
| Serum | ELISA-d | ai | Ingezim FeLV-Das | Ingenasa | |
| FIV | Serum | cELISA | c | Ingezim FIV | Ingenasa |
| PV | Serum | cELISA | c | Ingezim CPV | Ingenasa |
| Intestine, intestinal content, faeces | PCR | ai | BAD 1017A | Eurokit S.L. | |
| VPF (5’-GAT GGC ACC TCC GGC AAA GA-3’) | Reed et al. (1988) | ||||
| VPR (5’-TTT CTA GGT GCT AGT TGA G-3’) | |||||
| ELISA-d | ai | Ingezim CPV/FPV-Das | Ingenasa | ||
| IPo | ai | Rabbit IgG anti-feline panleukopenia virus 29989 ImmunoPure biotinylated protein A. 21123 ImmunoPure Avidin, Horseradish peroxidase conjugate | Pierce Chemical Co., Palacín, 1984, Rakich et al., 1986 | ||
| FA | ai | Feline serum anti-FPV | Ingenasa | ||
| Anti-feline IgG FITC conjugate | VMRD | ||||
| CDV | Serum | cELISA | c | Ingezim Moquillo | Ingenasa |
| Tonsils, retro-pharyngeal and/or submandibular LN | FA | ai | Canine serum anti-CDV | Ingenasa | |
| Anti-canine IgG FITC conjugate | VMRD | ||||
| PCR | ai | Ingene CDV | Ingenasa | ||
| CAV-1 | Serum | cELISA | c | Canine hepatitis IgG ELISA | Eurovet Veterinaria |
| Tonsils, retro-pharyngeal and/or submandibular LN | FA | ai | Canine serum anti-CAV-1 | Eurovet Veterinaria | |
| Anti-canine IgG FITC conjugate | VMRD | ||||
| EH | Serum | cELISA | c | Ingezim Ehrlichia | Ingenasa |
| Spleen | FA | ai | Canine serum positive to Ehrlichia canis. | VMRD | |
| Ref-211-P-EC Rabbit anti-dog IgG Fc fragment antibody, FITC conjugated | Rockland Immunochemicals | ||||
| CH | Serum | cELISA | c | Chlamydia antibodies | Eurovet Veterinaria |
| Conjunctival swab | RIC | ai | Clearview Chlamydia MF | Unipath | |
| LI | Serum | MAL | c | 1:100 | Faine (1982) |
| Kidney and/or urine | Staining | ai | Argentic stain | León-Vizcaíno et al. (1987) | |
| IPo + IFA | ai | Ig Rabbit anti Leptospira interrogans serovar Goat Ig anti-rabbit IgG: GAR/Ig/FITC or /IPo | Nordic Immunological Laboratories, León-Vizcaíno et al. (1987) | ||
| Culture | ai | EMJH medium | Difco Laboratories Baranton and Postic (1989) | ||
| BB | Tonsil smears | FA | ai | Serum anti Borrelia burgdorferi | Institut Pasteur Production VMRD, Baranton and Postic (1989) |
| Anti-canine IgG FITC | |||||
| SE | Multiorgan | Culture | ai | Selenite-cystine and Rappaport-Vassiliadis + Salmonella-Shigella agar and brilliant green agar | Millán et al., 2004 |
| MB | Serum | cELISA | c | Protein MBP70; ⩾30% | Martín-Atance et al., 2006 |
| Multiorgan | Culture | ai | Middlebrook’s 7H11 and Lowenstein-Jensen media | Corner and Nicolacopoulos, 1988, Gallager and Horwill, 1977 | |
| PCR | ai | TB1-F (5´-GAA CAA TCC GCA GTT GAC AA-3´) | Böddinghauss et al., 1990, Liébana et al., 1996 | ||
| TB1-R (5´-AGC ACG CTG TCA ATC ATG TA-3´) | |||||
| TG | Serum | MAT | c | 1:25 | Dubey and Desmonts (1987) |
| Faeces | FA | ai | Toxo-Troll ‘F’ 75 411 | BioMérieux Industry | |
| NC | Serum | ELISA + IFA | c | >40% | VMRD |
| CF | Entire blood | PCR | ai | 7549 (5´-GTCAGGATCCTGGGTTGATCCTGCCAG-3´) | Reichard et al., 2005, Millán et al., 2007a |
| 7548 (5´-GACTGAATTCGACTTCTCCTTCCTTTAAG-3´) |
FCV, feline calicivirus; FHV-1, feline herpesvirus-1; FCoV, feline coronavirus; FeLV, feline leukemia virus; FIV, feline immunodeficiency virus; PV, feline parvovirus subgroup virus; CDV, canine distemper virus; CAV-1, canine adenovirus-1; EH, Ehrlichia spp.; CH, Chlamydophila spp.; LI, Leptospira interrogans (serovars Australis, Autumnalis, Ballum, Bataviae, Bratislava, Canicola, Grippotyphosa, Hardjo, Hebdomadis, Icterogaemorrhagiae, Pomona, Saxkoebing, Sejroë, Tarassovi); BB, Borrelia burgdorferi; SE, Salmonella enterica; MB, Mycobacterium bovis; TG, Toxoplasma gondii; NC, Neospora caninum; CF, Cytauxzoon felis.
LN, lymph node.
ELISA, enzyme-linked immunosorbent assay (c, competitive; d, direct); FA, direct immunofluorescence; IFA, indirect immunofluorescence assay; IPo, immunoperoxidase; LA, latex aglutination; MAL, microaglutination-lisis; MAT, modified agglutination test; RIC, rapid immunochromatography.
ai, active infection; c, contact with the agent.
Questionnaires to domestic animal owners
We asked owners to complete a questionnaire asking for the total number of domestic animals on their property, any vaccination programmes carried out, and details of diseases suffered by their animals. The aim was to collect information about the health care that dogs and cats receive from their owners in the study areas. We also recorded the occurrence of sterilisation of cats in order to get an indication of the relative turnover of the population.
Statistics
For statistical analyses, lynx were separated into two age classes: (1) young (including juveniles living in the natal area, and subadults during the modal natal dispersal period), and (2) adults (>2 years old). The other species were divided in two age classes (juveniles and adults). Statistical analyses were performed with Statistica 6.0 (StatSoft, Inc.). Prevalence differences between areas and ages were tested using Fisher’s exact test.
Results
With the exception of feline herpesvirus (FHV)-1 and Borrelia burgdorferi, evidence of current or past contact with all the studied agents were detected in at least one of the studied species (Fig. 2 ).
Fig. 2.
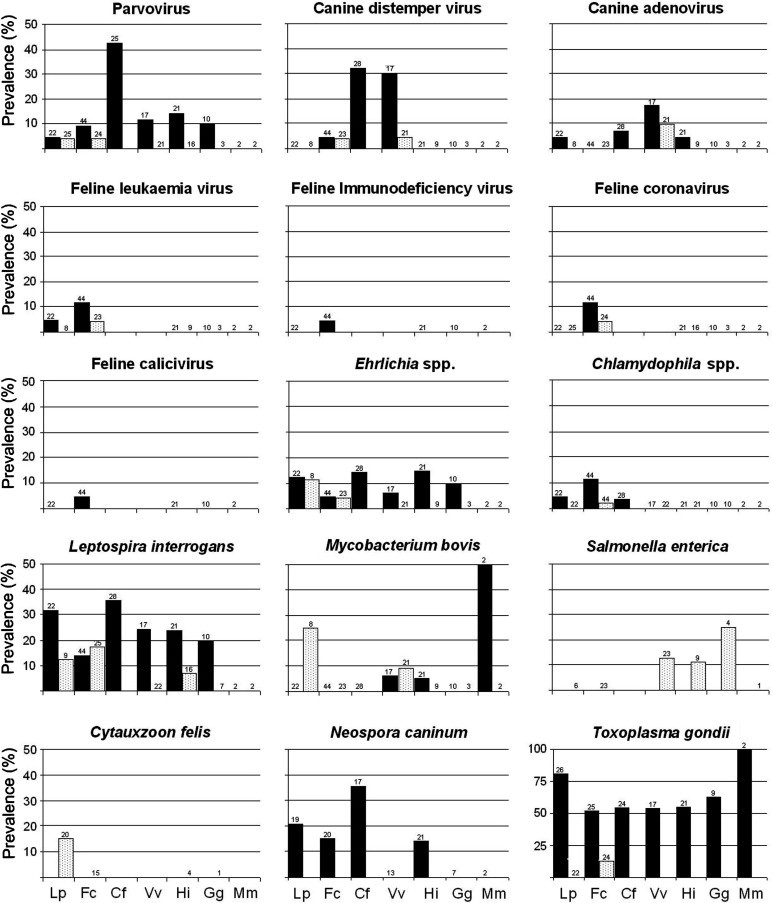
Results of the survey for pathogens in Iberian lynx (Lp), domestic cat (Fc), dog (Cf), red fox (Vv), Egyptian mongoose (Hi), common genet (Gg) and Eurasian badger (Mm) in lynx areas. Black columns: evidences of contact with the agent; white columns: detection of active infections. Sample size is shown in the top of the column (absence of data means that no animal was analysed). See Table 2 and text for further details.
Contact with pathogens in the Iberian lynx
The Iberian lynx showed low rates of contact with viral agents (Fig. 2). Antibodies against parvovirus (PV), FeLV, and canine adenovirus-1 (CAV-1) were each detected in one lynx (4.5%, 95% confidence intervals = 0.2–20%) in Sierra Morena. Antigens from PV were detected in the faeces of the lynx with antibodies against the virus. Bacteria and protozoa were more frequently indicated than viruses. The highest seroprevalence was observed for T. gondii (80.7%, 61–92), followed by Leptospira interrogans (31.8%, 15–54; serovars Icterohemorragiae [N = 4]; Canicola [1], Ballum [1], Sejroë [1]), Neospora caninum (21%, 7.5–44), Ehrlichia spp. (13.6%, 3.8–34), and Chlamydophila spp. (4.5%, 0.2–20). Prevalence through direct detection was Mycobacterium bovis: 25%, 4.6–63; Cytauxzoon felis: 15%, 4.2–37; and Ehrlichia spp. and L. interrogans: in both cases 12.5%, 0.6–50. Infection by Mycobacterium bovis (two positives from three lynx sampled, one with a generalised infection) and Cytauxzoon spp. (3/9) were only detected in lynx from Sierra Morena, though differences between areas were not statistically significant. Excretion of T. gondii in lynx faeces was not confirmed.
Significant age-related differences were only found for T. gondii, with juvenile lynx having lower prevalence (33%) compared to older age groups (86%; Fisher’s exact test, P = 0.024). One of the cases of TB was found in an individual of about 4 months of age. Cytauxzoon felis was only detected in three young males.
Contact with pathogens in sympatric carnivores
We detected 14/17 agents in cats, dogs and foxes (Fig. 2). The only difference between study areas was observed for the seroprevalence of feline PV subgroup in dogs, with a higher prevalence in Doñana (78%) than in Sierra Morena (23%; Fisher’s exact P = 0.012).
We found evidence of contact with at least one pathogen in 53% of the surveyed domestic cats (seroprevalence range 4.5–11.4%). We also found that 26% of necropsied cats had an active infection with PV, FeLV, CDV, feline coronavirus (FCoV), Ehrlichia spp. or Chlamydophila spp. Two cats belonging to the same ranch in Sierra Morena presented titres to CDV (the dog sampled in this ranch was seronegative). Antibodies to CDV and PV were frequent in dogs (32%, 17–52; and 42%, 22–60, respectively) and foxes (30%, 12–54; and 12%, 2.1–35). We detected CDV in one fox and one domestic cat culled in DNP.
Ehrlichia spp. was present in all the species studied except the badger. The seroprevalence of L. interrogans spp. was ⩾10% in these species, and was detected by culture in the domestic cat and mongoose (overall, we detected the serovars Icterohemorragiae [n = 24], Canicola [6], Ballum [4], Sejroë [3], Australis [1]). Positive cultures for Salmonella enterica were obtained from the fox, genet, and mongoose. Titres to Mycobacterium spp. were found in the two studied badgers, one mongoose and one fox. Mycobacterium bovis was isolated from organs samples in two foxes. Cytauxzoon spp. was not detected in any of the analysed blood samples from the domestic cat, mongoose or genet. The seroprevalence of T. gondii was >50% in all species, and T. gondii oocysts were only detected in the faeces of cats (17%). Antibodies to N. caninum were found in the dog, cat and mongoose.
The required sample size for age-related statistical analyses was only obtained for cats and foxes. Evidence of contact with FeLV and L. interrogans were more frequently found in juveniles (13% and 43%, respectively) than in adult cats (3% and 12%; Fisher’s exact P = 0.03 in both cases). All cats shedding T. gondii oocysts were adults. In foxes, contact with CDV, CAV-1, PV, and M. bovis was only apparent in adults. However, significant differences were only found for CDV (adults: 43%; juveniles: 0%; Fisher’s exact P = 0.02).
Questionnaires to owners
Twenty-five owners responded to the questionnaire, representing a total population of 29 dogs and 75 cats. Seven owners (16%) vaccinated their animals against rabies (13 dogs [45%] and one cat [1.3%]). Only three owners (12%) vaccinated their dogs against other agents. Three dogs (10%) were vaccinated against PV, and one (3.4%) had received a pentavalent vaccine but before the owner had moved to the study area. No cat had been given any vaccine. Only one case of disease in one domestic cat was mentioned by an owner (conjunctivitis). No cat had been neutered on any property.
Discussion
Observed prevalences
The survey showed that Iberian lynx do not have frequent contact with viral agents, which agrees with a previous serosurvey (Roelke et al., 2008), and the findings in other lynx species (see, for example, Ryser-Degiorgis et al., 2005). The solitary social system of the Iberian lynx (Ferreras et al., 1997) may decrease the risk of intra-species transmission of viral agents, and lynx probably come in contact with these agents mainly during inter-species contacts. An alternative explanation for the observed low seroprevalence may be the low rates of survival of lynx infected by viruses.
In contrast, the prevalence of viral agents in sympatric carnivores was higher. The domestic cat’s social behaviour and high population size probably explain the detection of 11 of the studied pathogens in this species. The seroprevalence of FeLV may be higher than indicated, since during the pathogenesis of feline leukemia, a proportion of infected cats remain latent, yielding ELISA negative results (Cotter, 1998). Seroprevalence of FeLV was higher among young cats, indicating that cats may be infected by their mothers and/or that many cats may die at a young age and few infected animals reach an adult age. Either way, the results showed that FeLV is actively circulating among the cat population within lynx areas.
In the case of FIV, due to the pathogenesis of the virus, the presence of antibodies implied that the seropositive individual was infected and thus a source of infection for other animals (Sellon, 1998). We also found that PV was broadly distributed in the carnivore population. The tests we ran did not allow us to determine the strain of PV, canine or feline. In fact, current taxonomy defines canine PV (CPV) and feline panleukopenia virus as a single taxonomic entity (Tattersall, 2006) and CPV was found to be responsible for about 5–10% of feline parvovirosis in cats in Germany (Truyen, 1999). Steinel et al. (2000) found CPV-2b sequences in five captive cheetahs (Acinonyx jubatus), and concluded that the observed distribution of feline PV antigenic types in captive large cats suggested a transmission from domestic dogs. The overall carnivore population studied here might well share this virus with the lynx. On the other hand, titres to CDV were found in a remarkable proportion of dogs and foxes. The higher seroprevalence we detected in adult foxes when compared to juveniles indicates that the agent is probably enzootic in the fox population in Doñana. Interestingly, we detected two seropositive cats, and the genome of CDV in a third one, which offers new evidence that interspecific transmission of CDV occurs (Harder and Osterhaus, 1997).
Bacteria and protozoa were frequent in lynx and the other species. The direct and indirect analyses conducted in the present study evidenced a wide distribution of L. interrogans. This spirochete is often found in carnivores, both wild and domestic (Richardson and Gauthier, 2003). The more frequent serovars were those that have rodents as a reservoir. The reservoir of serovar Canicola is the dog, and this serovar was already reported in wild canids (Khan et al., 1991) but, as far as we know, was not in any other wild carnivore. On the other hand, S. enterica presented prevalences in the range of these reported in wild carnivores in Northern Spain (Millán et al., 2004). The most probable source of Salmonella and Leptospira is infected water, scraps or rodents.
During the present survey, M. bovis was cultured from organ samples from two lynx. Bovine TB was already known to affect the Iberian lynx (see, for example, Pérez et al., 2001), which is probably an incidental host (Martín-Atance et al., 2006). In addition, evidence of exposure was found in three species of scavenger carnivores (fox, badger and mongoose) in Doñana. Wild ungulates display high prevalences of TB in both areas (Aranaz et al., 2004, Vicente et al., 2006), which makes them the most likely maintenance host. In fact, wild boar (Sus scrofa) and red deer (Cervus elaphus) are believed to be TB reservoirs in Mediterranean Spain (Gortazar et al., 2006). The route of infection for the lynx is unknown, although (at low proportions) ungulates are part of the lynx diet (Delibes, 1980, Gil-Sánchez et al., 2006). Other potential sources of infection that must be investigated are contaminated water or soil. Since felines are very susceptible to infection by M. bovis, repeatedly contacts with the agent are probably not necessary for the lynx to develop fatal infections. The finding of a juvenile lynx with a generalised infection supports this hypothesis.
Evidence of contact with Ehrlichia spp. was often found, including in three lynx. There is scarce information about Ehrlichia spp. in wild felids although Filoni et al. (2006) found antibodies against the agent in 1/18 pumas (Puma concolor). Ehrlichia canis and E. platys have been described in dog and foxes in Europe (Neer, 1998, Fishman et al., 2004) but, in Spain, although reactivity against agents implicated in feline ehrlichiosis have been reported in cats (see, for example, Ortuño et al., 2005) the presence of E. canis has not so far been confirmed.
Antibodies to Chlamydophila spp. were also detected in one lynx. It is not known whether C. felis or other Chlamydiae (e.g. C. psittaci or C. abortus) were detected in our survey. Few attempts have been made to detect this agent in wild felids; Paul-Murphy et al. (1994), for example, failed to detect it in pumas.
The seroprevalence of T. gondii in lynx was higher than that reported by Roelke et al. (2008), probably because the technique we used was more sensitive. High seroprevalences of T. gondii are commonly observed in free-living lynx species (Ryser-Degiorgis et al., 2006). Older lynx presented higher seroprevalences than juveniles, which may indicate that adult animals have an increasing probability of being exposed to T. gondii during their life-time, as has been observed in other lynx species (e.g. Ryser-Degiorgis et al., 2006). We did not detect oocysts in lynx faecal samples. Although it is accepted that all species of felids can act as a definitive hosts for T. gondii, Iberian lynx have not been shown to excrete oocysts demonstrating their role as a definitive host. Infection of the Iberian lynx probably occurs after exposure to infected prey, most probably rabbits (Almería et al., 2004). Lynx may also become infected transplacentally, through milk or by ingestion of food or water contaminated by oocysts from cat faeces (see Roelke et al., 2008). In fact, 17% of cats sharing a habitat with lynx showed oocysts in their faeces, which could explain the high seroprevalence of T. gondii infection observed in all the analysed species.
Antibodies to N. caninum are reported here for the first time in both the Iberian lynx and the mongoose. Recently, Sedlak and Bartova (2006) reported antibodies in the captive Eurasian lynx (Lynx lynx). Canids are the only proven definitive hosts of N. caninum (Gondim, 2006) and the highest seroprevalence of N. caninum antibodies in the present study was observed in dogs, which may be of epidemiological significance as the seropositive dogs might well have shed oocysts. We did not observe antibodies in the foxes we examined, although the presence of parasite DNA of N. caninum in the fox has been reported in Spain (Almería et al., 2002).
Cytauxzoon spp. was recently found infecting the Iberian lynx (Luaces et al., 2005) and, in agreement with that work, we only found Cytauxzoon spp. in lynx from Sierra Morena. Possible causes for the absence of the agent in Doñana included the absence of the intermediate host or the possibility of extinction of the protozoa in that area (see Millán et al., 2007a for further details). No domestic cat was found to be infected, but due to the acute course of cytauxzoonosis in cats (Kier and Greene, 1998), the probability of finding an infected individual is very low. The mongoose and genet were not found to be parasitised, probably due to the high specificity of this organism for felids.
Pathological significance of the detected agents
Very little is known about the pathological significance of the studied agents for the Iberian lynx. In most cases, only inferences from studies in other hosts can be made. For example, FeLV has been demonstrated to be fatal in the bobcat (Lynx rufus) (Sleeman et al., 2001), and it is considered a potential emerging disease in large cats (Kennedy-Stoskopf, 1999). Infection with FIV causes feline acquired immunodeficiency disease syndrome (AIDS), which is characterised by a loss of immunological capability, allowing subsequent opportunistic infections, and death from micro-organisms that would not normally harm the animal. However, so far there has been no verifiable disease association for any of the wild felid species for which FIV strains have been found (Troyer et al., 2005).
Mortality due to FPL has been observed in the bobcat (Wassmer et al., 1988) and cougar (Roelke et al., 1993). Some isolates of FCoV are known to induce feline infectious peritonitis (FIP), while others cause very mild to fatal enteritis (Addie and Jarrett, 1998). FIP is the single most important infectious cause of death in young cats (Fehr et al., 1996), and FCoV have been isolated from captive cheetahs that died during an epizootic (Pfeifer et al., 1983). In fact, only TB has been reported to be lethal for the Iberian lynx (Pérez et al., 2001); this chronic disease may not cause substantial mortality but may extract individuals from the population with the subsequent loss of population size and genetic variability. In contrast, whether or not Cytauxzoon spp. may cause disease in the Iberian lynx has still to be clarified (see Millán et al., 2007a). For other diseases (e.g. ehrlichiosis, chlamydiosis, neosporosis), little information is available regarding their potential pathogenicity in wild felids, and the relevance of the infections for lynx conservation needs to be elucidated. Since Ehrlichia spp. was widely distributed among the studied carnivores and produces a severe syndrome, it might well affect the lynx population dynamics.
It is generally believed that wild carnivores act as asymptomatic carriers of Salmonella (Millán et al., 2004) and Leptospira (Greene et al., 1998), but where there is immunosuppression by a concomitant agent (such as FIV), clinical cases may occur. Toxoplasma gondii, according to the high seroprevalence in adult lynx, may be enzootic in the lynx population, and is probably not a relevant cause of morbidity or mortality although fatal toxoplasmosis has been reported in bobcats (Dubey et al., 1987).
Are sympatric carnivores acting as reservoir of diseases for the Iberian lynx?
Often, the presence of infectious agents in other lynx species has been associated with the presence of the disease in domestic carnivores (Sleeman et al., 2001, Ryser-Degiorgis et al., 2006), and indeed dogs were considered to be the most likely source of infection of CDV for carnivores in Africa. Iberian lynx kill other carnivores (Palomares et al., 1996), which probably provides opportunities for direct transmission of pathogens such as FIV, FeLV or CDV. In addition, other agents can survive for a time in the environment (e.g. PV, FCoV, Toxoplasma, or Neospora), increasing the probability of reaching a new susceptible host.
Evidence of contact with canine agents such as CAV-1, Leptospira Canicola or N. caninum, the detection of PV or L. interrogans in most of the studied species, or the cases of CD in cats, indicates that inter-specific transmission is likely in lynx areas. However, to confirm this hypothesis, it will be necessary to fully identify and compare the strains of PV or FeLV, or the species of Chlamydophila spp. or Ehrlichia spp. present among carnivores in these areas. For example, if lynx are susceptible to CPV, the high prevalence of PV detected in the dog and fox may pose a risk for lynx conservation. It would also be necessary to clarify whether the positive result for CAV-1 is due to a cross-reaction or whether this canine virus is actually able to infect the lynx. In the case of Cytauxzoon spp., it has not so far been determined whether one or more strains or species infect the Iberian lynx and, in addition, the intermediate host has not been still identified in Spain (Millán et al., 2007a). On the other hand, our results indicate that other sympatric animals (and not only carnivores) such as wild ungulates (TB), rodents (L. interrogans) or rabbits (T. gondii) are probably also acting as maintenance host for pathogens, and this must be taken into consideration.
Management implications
Although vaccines are available against many of the canine agents that displayed high prevalences among the studied dogs, and also against feline pathogens, they appear to be seldom used. According to the results from the questionnaires, there were on average three cats per owner, and neutering was never used to control cat populations. This implies that new potential hosts are frequently available for agent maintenance, as reflected by the high seroprevalence of FeLV in young cats. The lack of vaccination and neutering of domestic animals will assist in creating a rich environment for disease agents.
Conclusions
Our results indicate that the Iberian lynx is far from safe from disease risks. With a high diversity of disease agents in the populations of sympatric carnivores, and the apparent lack of acquired immunity and immunocompetence, we believe that a disease outbreak among the local abundant carnivores may pose a serious disease risk for lynx conservation. Management actions are necessary to avoid a potential disease outbreak among sympatric carnivores that might affect the two remaining Iberian lynx populations. A campaign to control domestic carnivore populations (by vaccination, euthanasia, and/or neutering) would reduce the incidence of circulating pathogens. In addition, we believe that diseases have to be taken into account when planning lynx translocations or reintroductions (Wild et al., 2006). Disease-related issues should be carefully considered in endeavouring to optimise the conservation efforts targeted at endangered species such as the Iberian lynx.
Conflict of interest statement
None of the authors of this paper has a financial or personal relationship with other people or organisations that could inappropriately influence or bias the content of the paper.
Acknowledgments
This work was supported by the ‘Programa de Actuaciones para la Conservación del Lince Ibérico en Andalucía II’, Consejería de Medio Ambiente, Junta de Andalucía; and Project CGL2004-00346/BOS of the Dirección General de Investigación of the Ministerio de Educación y Ciencia. We wish to thank personnel from Junta de Andalucía, Estación Biológica de Doñana, Centro de Análisis y Diagnóstico, Doñana National Park, and El Acebuche. P. Acevedo-Gisguaje kindly helped us in creating the figures. A. Rodríguez was supported by Consejería de Innovación (Junta de Andalucía).
References
- Addie D.D., Jarrett O. Feline coronavirus infections. In: Greene C.E., editor. Infectious Diseases of Dog and Cat. W.B. Saunders; Pennsylvania: 1998. pp. 63–75. [Google Scholar]
- Almería S., Ferrer D., Pabón M., Castella J., Mañas S. Red foxes (Vulpes vulpes) are a natural intermediate host of Neospora caninum. Veterinary Parasitology. 2002;107:287–294. doi: 10.1016/s0304-4017(02)00162-0. [DOI] [PubMed] [Google Scholar]
- Almería S., Calvete C., Pagés A., Gauss C., Dubey J.P. Factors affecting the seroprevalence of Toxoplasma gondii infection in wild rabbits (Oryctolagus cuniculus) from Spain. Veterinary Parasitology. 2004;123:265–270. doi: 10.1016/j.vetpar.2004.06.010. [DOI] [PubMed] [Google Scholar]
- Aranaz A., de Juan L., Montero N., Sánchez C., Galka M., Delso C., Álvarez J., Romero B., Bezos J., Vela A.I., Briones V., Mateos A., Domínguez L. Bovine tuberculosis (Mycobacterium bovis) in wildlife in Spain. Journal of Clinical Microbiology. 2004;42:2602–2608. doi: 10.1128/JCM.42.6.2602-2608.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baranton G., Postic D. Méthodes de laboratoire. Institute Pasteur; Paris, France: 1989. (Leptospirose–Borreliose de Lyme). [Google Scholar]
- Böddinghauss B., Rogall T., Flohr T., Blücker H., Büttger E.C. Detection and identification of Mycobacteria by amplification of rRNA. Journal of Clinical Microbiology. 1990;28:1751–1759. doi: 10.1128/jcm.28.8.1751-1759.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley C.A., Altizer S. Urbanization and the ecology of wildlife diseases. Trends in Ecology and Evolution. 2007;22:95–102. doi: 10.1016/j.tree.2006.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cleaveland S., Appel M.G.J., Chalmers W.S.K., Chillingworth C., Kaare M., Dye C. Serological and demographic evidence for domestic dogs as a source of canine distemper virus infection for Serengeti wildlife. Veterinary Microbiology. 2000;72:217–227. doi: 10.1016/s0378-1135(99)00207-2. [DOI] [PubMed] [Google Scholar]
- Corner L.A., Nicolacopoulos C. Comparison of media used for the primary isolation of Mycobacterium bovis by veterinary and medical diagnostic laboratories. Australian Veterinary Journal. 1988;65:202–205. doi: 10.1111/j.1751-0813.1988.tb14457.x. [DOI] [PubMed] [Google Scholar]
- Cotter S.M. Feline leukemia virus infection. In: Greene C.E., editor. Infectious Diseases of Dog and Cat. W.B. Saunders; Pennsylvania: 1998. pp. 78–91. [Google Scholar]
- Delibes M. Feeding ecology of the Spanish lynx in the Coto Doñana. Acta Theriologica. 1980;25:309–324. [Google Scholar]
- Dubey J.P., Desmonts G. Serological responses of equids fed Toxoplasma gondii oocysts. Equine Veterinary Journal. 1987;19:337–339. doi: 10.1111/j.2042-3306.1987.tb01426.x. [DOI] [PubMed] [Google Scholar]
- Dubey J.P., Quinn W.J., Weinandy D. Fatal neonatal toxoplasmosis in a bobcat (Lynx rufus) Journal of Wildlife Diseases. 1987;23:324–327. doi: 10.7589/0090-3558-23.2.324. [DOI] [PubMed] [Google Scholar]
- Faine, S., 1982. Guidelines for the Control of Leptospirosis. WHO Offset Publications No. 67, World Health Organization, Geneva, Switzerland.
- Fehr D., Bolla S., Herrewegh A., Horzinek M., Lutz H. Detection of feline coronavirus using RT-PCR, basis for the study of the pathogenesis of feline infectious peritonitis (FIP) Schweiz Archiv für Tierheilkunde. 1996;138:74–79. [PubMed] [Google Scholar]
- Ferreras P., Beltrán J.F., Aldama J.J., Delibes M. Spatial organization and land tenure system of the endangered Iberian lynx (Lynx pardinus, Temminck, 1824) Journal of Zoology. 1997;243:163–189. [Google Scholar]
- Filoni C., Catao-Dias J.L., Bay G., Durigon E.L., Jorge R.S., Lutz H., Hofmann-Lehmann R. First evidence of feline herpesvirus, calicivirus, parvovirus, and Ehrlichia exposure in Brazilian free-ranging felids. Journal of Wildlife Diseases. 2006;42:470–477. doi: 10.7589/0090-3558-42.2.470. [DOI] [PubMed] [Google Scholar]
- Fishman Z., Gonen L., Harrus S., Strauss-Ayali D., King R., Baneth G. A serosurvey of Hepatozoon canis and Ehrlichia canis antibodies in wild red foxes (Vulpes vulpes) from Israel. Veterinary Parasitology. 2004;119:21–26. doi: 10.1016/j.vetpar.2003.08.012. [DOI] [PubMed] [Google Scholar]
- Gallager J., Horwill D.M. A selective oleic acid albumin agar medium for the cultivation of Mycobacterium bovis. Journal of Hygiene (Cambridge) 1977;79:155–160. doi: 10.1017/s0022172400052943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gil-Sánchez J.M., Ballesteros-Duperon E., Bueno-Segura J.F. Feeding ecology of the Iberian lynx (Lynx pardinus) in eastern Sierra Morena (Southern Spain) Acta Theriologica. 2006;51:85–90. [Google Scholar]
- Gondim L.F.P. Neospora caninum in wildlife. Trends in Parasitology. 2006;22:247–252. doi: 10.1016/j.pt.2006.03.008. [DOI] [PubMed] [Google Scholar]
- Gortazar C., Acevedo P., Ruiz-Fons F., Vicente J. Disease risks and overabundance of game species. European Journal of Wildlife Research. 2006;52:81–87. [Google Scholar]
- Greene C.E., Miller M.A., Brown C.A. Leptospirosis. In: Greene C.E., editor. Infectious Diseases of Dog and Cat. W.B. Saunders; Pennsylvania: 1998. pp. 302–310. [Google Scholar]
- Guzmán, J.N., García, F.J., Garrote, G., Pérez de Ayala, R., Iglesias, C. 2004. El lince ibérico (Lynx pardinus) en España y Portugal. Censo-diagnóstico de sus poblaciones. Madrid, Dirección General para la Biodiversidad, Ministerio de Medio Ambiente.
- Harder T.C., Osterhaus A.D.M.E. Canine distemper virus – a morbillivirus in search of new hosts? Trends in Microbiology. 1997;5:120–124. doi: 10.1016/S0966-842X(97)01010-X. [DOI] [PubMed] [Google Scholar]
- Johnson W.E., Godoy J.A., Palomares F., Delibes M., Fernandes M., Revilla E., O’Brien S.J. Phylogenetic and phylogeographic analysis of Iberian lynx populations. Journal of Heredity. 2004;95:19–28. doi: 10.1093/jhered/esh006. [DOI] [PubMed] [Google Scholar]
- Kennedy-Stoskopf S. Emerging viral infections in large cats. In: Fowler M.E., Miller R.E., editors. vol. 4. W.B. Saunders; Philadelphia: 1999. pp. 401–410. (Zoo and Wild Animal Medicine Current Therapy). [Google Scholar]
- Khan M.A., Goyal S.M., Diesch S.L., Mech L.D., Fritts S.H. Seroepidemiology of leptospirosis in Minnesota wolves. Journal of Wildlife Diseases. 1991;27:248–253. doi: 10.7589/0090-3558-27.2.248. [DOI] [PubMed] [Google Scholar]
- Kier A.B., Greene C.E. Cytauxzoonosis. In: Greene C.E., editor. Infectious Diseases of Dog and Cat. W.B. Saunders; Pennsylvania: 1998. pp. 517–519. [Google Scholar]
- León-Vizcaíno L., Hermoso de Mendoza M., Garrido G. Incidence of abortions caused by leptospirosis in sheep and goats in Spain. Comparative Immunology, Microbiology and Infectious Diseases. 1987;10:149–153. doi: 10.1016/0147-9571(87)90009-9. [DOI] [PubMed] [Google Scholar]
- Liébana E., Aranaz A., Francis B., Cousins D. Assessment of genetic markers for species differentiation within the Mycobacterium tuberculosis complex. Journal of Clinical Microbiology. 1996;34:933–938. doi: 10.1128/jcm.34.4.933-938.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luaces I., Aguirre E., García-Montijano M., Velarde J., Tesouro M.A., Sánchez C., Galka M., Fernández P., Sáinz A. First report of an intraerythrocytic small piroplasm in wild Iberian lynx (Lynx pardinus) Journal of Wildlife Diseases. 2005;41:810–815. doi: 10.7589/0090-3558-41.4.810. [DOI] [PubMed] [Google Scholar]
- Martín-Atance P., León-Vizcaíno L., Palomares F., Revilla E., González-Candela M., Calzada J., Cubero-Pablo M.J., Delibes M. Antibodies to Mycobacterium bovis in wild carnivores from Doñana National Park (Spain) Journal of Wildlife Diseases. 2006;42:704–708. doi: 10.7589/0090-3558-42.3.704. [DOI] [PubMed] [Google Scholar]
- McCallum H., Dobson A. Detecting disease and parasite threats to endangered species and ecosystems. Trends in Ecology and Evolution. 1995;10:190–194. doi: 10.1016/s0169-5347(00)89050-3. [DOI] [PubMed] [Google Scholar]
- Millán J., Casanova J.C. Helminth parasites of the endangered Iberian lynx (Lynx pardinus) and sympatric carnivores. Journal of Helminthology. 2007;81:377–380. doi: 10.1017/S0022149X07869203. [DOI] [PubMed] [Google Scholar]
- Millán J., Adúriz G., Moreno B., Juste R.A., Barral M. Salmonella isolates from wild birds and mammals in the Basque Country (Spain) Revue Scientifique et Technique de l’Office Internationale des Epizooties. 2004;23:905–911. doi: 10.20506/rst.23.3.1529. [DOI] [PubMed] [Google Scholar]
- Millán J., Naranjo V., Rodríguez A., Pérez de la Lastra J.M., Mangold A.J., de la Fuente J. Prevalence of infection and 18S rRNA gene sequences of Cytauxzoon species in Iberian lynx (Lynx pardinus) in Spain. Parasitology. 2007;134:995–1001. doi: 10.1017/S003118200700248X. [DOI] [PubMed] [Google Scholar]
- Millán J., Ruiz-Fons F., Márquez F.J., Viota M., López-Bao J.V., Martín-Mateo M.P. Ectoparasites of the endangered Iberian lynx Lynx pardinus and sympatric wild and domestic carnivores in Spain. Medical and Veterinary Entomology. 2007;21:248–254. doi: 10.1111/j.1365-2915.2007.00696.x. [DOI] [PubMed] [Google Scholar]
- Neer T.M. Ehrlichiosis. In: Greene C.E., editor. Infectious Diseases of Dog and Cat. W.B. Saunders; Pennsylvania: 1998. pp. 153–161. [Google Scholar]
- Nowell, K., Jackson, P., 1996. Wild Cats. Status Survey and Conservation Action Plan. IUCN/SSC Cat Specialist Group. IUCN, Gland, Switzerland.
- Ortuño A., Gauss C.B., García F., Gutiérrez J.F. Serological evidence of Ehrlichia spp. exposure in cats from north-eastern Spain. Journal of Veterinary Medicine Series B. 2005;52:246–248. doi: 10.1111/j.1439-0450.2005.00849.x. [DOI] [PubMed] [Google Scholar]
- Palacín, A., 1984. Técnicas inmunohistoquímicas. Aspectos teórico-prácticos. ATOM, S.A., Barcelona, Spain.
- Palomares F., Ferreras P., Fedriani J.M., Delibes M. Spatial relationships between Iberian lynx and other carnivores in an area of south-western Spain. Journal of Applied Ecology. 1996;33:5–13. [Google Scholar]
- Palomares, F., Delibes, M., Ferreras, P., Aldama, J., Revilla, E., Calzada, J., Fernández, N., 2003. Estructura de la metapoblación de linces de Doñana. In: In Memoriam al Professor Dr. Isidoro Ruiz Martínez. Jaén University, Jaén, Spain, pp. 506–526.
- Palomo, L.J., Gisbert, J., 2002. Atlas de los mamíferos terrestres de España. Dirección General de Conservación de la Naturaleza-SECEM-SECEMU, Madrid, Spain.
- Paul-Murphy J., Work T., Hunter D., McFie E., Fjelline D. Serologic survey and serum biochemical reference ranges of the free-ranging mountain lion (Felis concolor) in California. Journal of Wildlife Diseases. 1994;30:205–215. doi: 10.7589/0090-3558-30.2.205. [DOI] [PubMed] [Google Scholar]
- Peña L., García P., Jiménez M.A., Benito A., Alenza M.D.P., Sánchez B. Histopathological and immunohistochemical findings in lymphoid tissues of the endangered Iberian lynx (Lynx pardinus) Comparative Immunology Microbiology and Infectious Diseases. 2006;29:114–126. doi: 10.1016/j.cimid.2006.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pérez J., Calzada J., León-Vizcaíno L., Cubero M.J., Velarde J., Mozos E. Tuberculosis in an Iberian lynx (Lynx pardina) Veterinary Record. 2001;148:414–415. doi: 10.1136/vr.148.13.414. [DOI] [PubMed] [Google Scholar]
- Pfeifer M.L., Evermann J.F., Roelke M.E., Gallina A.M., Ott R.L., McKeirnan A.J. Feline infectious peritonitis in a captive cheetah. Journal of the American Veterinary Medical Association. 1983;183:1317–1319. [PubMed] [Google Scholar]
- Power A.G., Mitchell C.E. Pathogen spillover in disease epidemics. American Naturalist. 2004;164:79–89. doi: 10.1086/424610. [DOI] [PubMed] [Google Scholar]
- Rakich P.M., Prasse K.W., Lukert P.D., Cornelius L.M. Immunohistochemical detection of canine adenovirus in paraffin sections of liver. Veterinary Pathology. 1986;23:478–484. doi: 10.1177/030098588602300419. [DOI] [PubMed] [Google Scholar]
- Reed A.P., Jones E.V., Miller T.J. Nucleotide sequence and genome organization of canine parvovirus. Journal of Virology. 1988;62:266–276. doi: 10.1128/jvi.62.1.266-276.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichard M.V., van den Bussche R.A., Meinkoth J.H., Hoover J.P., Kocan A.A. A new species of Cytauxzoon from Pallas’ cats caught in Mongolia and comments on the systematics and taxonomy of piroplasmids. Journal of Parasitology. 2005;91:420–426. doi: 10.1645/GE-384R. [DOI] [PubMed] [Google Scholar]
- Richardson D.J., Gauthier J.L. A serosurvey of leptospirosis in Connecticut peridomestic wildlife. Vector Borne and Zoonotic Diseases. 2003;3:187–193. doi: 10.1089/153036603322662174. [DOI] [PubMed] [Google Scholar]
- Roelke M.E., Forrester D.J., Jacobson E.R., Kollias G.V., Scott F.W., Barr M.C., Evermann J.F., Pirtle E.C. Seroprevalence of infectious disease agents in free-ranging Florida panthers (Felis concolor coryi) Journal of Wildlife Diseases. 1993;29:36–49. doi: 10.7589/0090-3558-29.1.36. [DOI] [PubMed] [Google Scholar]
- Roelke M., Johnson W.E., Millán J., Palomares F., Revilla E., Rodríguez A., Calzada J., Ferreras P., León-Vizcaíno L., Delibes M., O’Brien S.J. Exposure to disease agents in the endangered Iberian lynx (Lynx pardinus) European Journal of Wildlife Research. 2008;54:171–178. doi: 10.1007/s10344-007-0122-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roelke-Parker M.E., Munson L., Packer C., Kock R., Cleaveland S., Carpenter M., O’Brien S.J., Pospischil A., Hofmann-Lehmann R., Lutz H., Mwamengele G.L.M., Mgasa M.N., Machange G.A., Summers B.A., Appel M.J.G. A canine distemper virus epidemic in Serengeti lions (Panthera leo) Nature. 1996;379:441–445. doi: 10.1038/379441a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryser-Degiorgis M.P., Hofmann-Lehmann R., Leutenegger C.M., af Segerstad C.H., Morner T., Mattsson R., Lutz H. Epizootiologic investigations of selected infectious disease agents in free-ranging Eurasian lynx from Sweden. Journal of Wildlife Diseases. 2005;41:58–66. doi: 10.7589/0090-3558-41.1.58. [DOI] [PubMed] [Google Scholar]
- Ryser-Degiorgis M.P., Jakubek E.B., af Segerstad C.H., Brojer C., Morner T., Jansson D.S., Lunden A., Uggla A. Serological survey of Toxoplasma gondii infection in free-ranging Eurasian lynx (Lynx lynx) from Sweden. Journal of Wildlife Diseases. 2006;42:182–187. doi: 10.7589/0090-3558-42.1.182. [DOI] [PubMed] [Google Scholar]
- Scott M.E. The impact of infectious disease on animal populations, implications for conservation biology. Conservation Biology. 1988;2:40–65. [Google Scholar]
- Sedlak K., Bartova E. The prevalence of Toxoplasma gondii IgM and IgG antibodies in dogs and cats from the Czech Republic. Veterinarni Medicina. 2006;51:555–558. [Google Scholar]
- Sellon R.K. Feline immunodeficiency virus infection. In: Greene C.E., editor. Infectious Diseases of Dog and Cat. W.B. Saunders; Pennsylvania: 1998. pp. 92–104. [Google Scholar]
- Sleeman J.M., Keane J.M., Johnson J.S., Brown R.J., Woude S.V. Feline leukemia virus in a captive bobcat. Journal of Wildlife Diseases. 2001;37:194–200. doi: 10.7589/0090-3558-37.1.194. [DOI] [PubMed] [Google Scholar]
- Steinel A ., Munson L., van Vuuren M., Truyen U. Genetic characterization of feline parvovirus sequences from various carnivores. Journal of General Virology. 2000;81:345–350. doi: 10.1099/0022-1317-81-2-345. [DOI] [PubMed] [Google Scholar]
- Tattersall P. The evolution of parvovirus taxonomy. In: Kerr J., Cotmore S.F., Bloom M.E., Linden R.M., Parrish C.R., editors. Parvoviruses. Oxford University Press; NY: 2006. pp. 5–14. [Google Scholar]
- Thorne E.T., Williams E.S. Disease and endangered species, the black-footed ferret as a recent example. Conservation Biology. 1988;2:66–74. [Google Scholar]
- Troyer J.L., Pecon-Slattery J., Roelke M.E., Johnson W., VandeWoude S., Vázquez-Salat N., Brown M., Frank L., Woodroffe R., Winterbach C., Winterbach H., Hemson G., Bush M., Alexander K.A., Revilla E., O’Brien S.J. Seroprevalence and genomic divergence of circulating strains of feline immunodeficiency virus among Felidae and Hyaenidae species. Journal of Virology. 2005;79:8282–8294. doi: 10.1128/JVI.79.13.8282-8294.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truyen U. Emergence and recent evolution of canine parvovirus. Veterinary Microbiology. 1999;69:47–50. doi: 10.1016/s0378-1135(99)00086-3. [DOI] [PubMed] [Google Scholar]
- Vicente J., Höfle U., Garrido J.M., Fernández-de-Mera I.G., Juste R., Barral M., Gortazar C. Wild boar and red deer display high prevalences of tuberculosis-like lesions in Spain. Veterinary Research. 2006;37:107–119. doi: 10.1051/vetres:2005044. [DOI] [PubMed] [Google Scholar]
- Wassmer D.A., Guenther D.D., Layne J.N. Ecology of the bobcat in south-central Florida. Bulletin of the Florida State Museum, Biological Sciences. 1988;3:159–228. [Google Scholar]
- Whiteman C.W., Matushima E.R., Cavalcanti-Confalonieri U.E., Correia-Palha M.D., Lima da Silva A.S., Monteiro V.C. Human and domestic animal populations as a potential threat to wild carnivore conservation in a fragmented landscape from the Eastern Brazilian Amazon. Biological Conservation. 2007;138:290–296. [Google Scholar]
- Wild M.A., Shenk T.M., Spraker T.R. Plague as a mortality factor in Canada lynx (Lynx canadensis) reintroduced to Colorado. Journal of Wildlife Diseases. 2006;42:646–650. doi: 10.7589/0090-3558-42.3.646. [DOI] [PubMed] [Google Scholar]


