Graphical abstract
Keywords: Vaccines, Autoimmunity, Autoimmune/inflammatory syndrome induced by adjuvants, Aluminium, Human papilloma virus, Vaccinomics, Vaccine safety, Adjuvant
Abstract
Vaccines and autoimmunity are linked fields. Vaccine efficacy is based on whether host immune response against an antigen can elicit a memory T-cell response over time. Although the described side effects thus far have been mostly transient and acute, vaccines are able to elicit the immune system towards an autoimmune reaction. The diagnosis of a definite autoimmune disease and the occurrence of fatal outcome post-vaccination have been less frequently reported. Since vaccines are given to previously healthy hosts, who may have never developed the disease had they not been immunized, adverse events should be carefully accessed and evaluated even if they represent a limited number of occurrences.
In this review of the literature, there is evidence of vaccine-induced autoimmunity and adjuvant-induced autoimmunity in both experimental models as well as human patients. Adjuvants and infectious agents may exert their immune-enhancing effects through various functional activities, encompassed by the adjuvant effect. These mechanisms are shared by different conditions triggered by adjuvants leading to the autoimmune/inflammatory syndrome induced by adjuvants (ASIA syndrome).
In conclusion, there are several case reports of autoimmune diseases following vaccines, however, due to the limited number of cases, the different classifications of symptoms and the long latency period of the diseases, every attempt for an epidemiological study has so far failed to deliver a connection. Despite this, efforts to unveil the connection between the triggering of the immune system by adjuvants and the development of autoimmune conditions should be undertaken. Vaccinomics is a field that may bring to light novel customized, personalized treatment approaches in the future.
1. Introduction
Vaccines have been a preventive treatment option available for over 200 years. They have been proven to be effective in preventing infections that previously had high morbidity and mortality. An example of this is the eradication of small pox, which was mainly attributed to successful vaccination programs. Preventing a high burden disease has since proven to be a cost effective measure and, as such, vaccines have become a part of multiple national health programs. These promising results led to the development of more and more vaccines and to the study of its applicability in other fields such as cancer prevention and treatment.
Vaccines are drugs administered to healthy individuals, and much like other drugs, vaccines are associated with adverse events. Usually the described adverse events are transient and acute, but may rarely present with hypersensitivity and induction of autoimmunity that may be severe and fatal. These adverse events play an important role in the life of the vaccinated patients.
Immune mediated diseases arise from various different sources; these include environmental, genetic, hormonal and immune defects. The combination of these defects can be described as the mosaic of autoimmunity [1]. Patient background can be used as a clue to determine the response that may be elicited following drug administration.
It has been proven that infectious agents may elicit an autoimmune disease in a prone subject through various mechanisms, including, but not limited to, molecular mimicry, epitope spreading and polyclonal activation [2].
Scientific findings suggest that autoimmunity may be triggered by vaccine adjuvants, of which aluminum compounds (aluminum hydroxide and phosphate) have been the most studied and the most widely used. Adjuvants are molecules, which, in combination with antigens, enhance immunological response. This enables an easier and more effective recognition of “non self”, which in turn permits the triggering of adaptive and innate immune responses [3].
Recently a new syndrome was described: “Autoimmune/inflammatory syndrome induced by adjuvants” (ASIA). This embodies a spectrum of reactions, which are usually mild but may also be severe. These reactions are attributed to adjuvant stimulation, which can include chronic exposure to silicone, tetramethylpentadecane, pristane, aluminum, infectious components and other adjuvants. All of these environmental factors have been found to induce autoimmunity and inflammatory manifestations by themselves both in animal models and in humans. The mechanisms of this disease will be described in further detail [4].
This review will focus on general mechanism of vaccines, adjuvant-induced autoimmunity, and on vaccines and the specific autoimmune diseases that they may trigger.
2. General mechanisms of vaccines and adjuvant induced autoimmunity
2.1. Adjuvants role in infections and autoimmunity
Adjuvants approved to date for human vaccines are: aluminum, MF59 in some viral vaccines, MPL, AS04, AS01B and AS02A against viral and parasitic infections, virosomes for hepatitis B virus (HBV), human papilloma virus (HPV), hepatitis A virus (HAV), and cholera toxin for cholera.
Adjuvants may be composed of several different compounds. Currently, oil based adjuvants, virosomes, toll-like receptors (TLRs) related adjuvants, MPL, adjuvants made of unmethylated CpG dinucleotides and tuftsin have all been described.
It is of great interest the understanding of the mechanisms related to the adjuvant effect, as well as to aluminum salts. Aluminum acts through multiple pathways, which do not depend solely on TLRs signaling. Each of these pathways leads to an enhanced host immune response [5].
There are many oil based adjuvants. One is incomplete Freund adjuvant (IFA), which contains water in oil emulsion. Another is complete Freund adjuvant (CFA), which is the same as IFA, except that it also contains killed Mycobacteria in addition to water in oil emulsion. Usually, CFA is used for primary vaccination and IFA for boosting. Recent oil based adjuvants that have been developed are MF59 (Novartis®), AS03 (GlaxoSmithKline®), Advax™ which are based on inulin compounds (Vaxine™ Pty) and Qs-21/ISCOMs, which are immune stimulating complexes composed of cholesterol and phospholipid with or without antigen (Table 1 ).
Table 1.
Types of adjuvants in development or use.
| Type of Adjuvants | Name of compound | Vaccines in test or use |
|---|---|---|
| Related to Toll like receptors (TLRs) | Aluminum hydroxide and phosphate | PCV7, PCV13, MenC, HPV, HAV, Hib; tetanus vaccine |
| IC31 | Influenza [14] | |
| ASO4 (MPL + QS-21), ASO2A (MPL + Alum), CPG 7907, and GM-CSF | Papilloma virus, hepatitis B, malaria [15] | |
| RD-529, ISS, Flagellin TLR agonists |
||
| Oil based emulsions | CFA, IFA, MF59TM montanide, adjuvant 65, lipovant, QS-21 [16] ISCOMs, ADVAX™, algammulin | Influenza |
| Xenobiotic adjuvants | Unmethylated CpG dinucleotides [17] | Hepatitis B, allergens, tumor cells |
| Tuftsin auto adjuvant | Tuftsin | Influenza, malaria, autoimmune encephalomyelitis, restoration of innate immune system (HIV patients), SLE [18], [19], [20], [21] |
CFA: complete freund adjuvant; IFA: incomplete freund adjuvant; PCV: pneumococcal conjugated vaccine; MenC: meningitis C; HPV: human papiloma Virus; HAV: hepatitis A virus; Hib: haemophilus influenza type b.
Virosomes are adjuvants that contain a membrane-bound hemagglutinin and neuraminidase obtained from the influenza virus. Both components facilitate the uptake into antigen presenting cells (APC) and mimic the natural immune response [6].
Leucocyte membranes have membrane bound pattern recognition receptors (PRRs) called TLRs, which are responsible for detecting most (although certainly not all) antigen-mediated infections. Their activation leads to adaptive immune responses. For this reason, many adjuvants that are used today are directed to PRRs. These adjuvants are called TLRs related adjuvants [7].
MPL is a series of 4'monophosphoryl lipid A obtained from the purification of a modified lipopolysaccharide (LPS) of Salmonella Minnesota.
Bacterial deoxyribonucleic acid (DNA) is immunostimulatory due to Unmethylated CpG dinucleotides. Vertebrate DNA has relatively low amounts of unmethylated CpG compared to Bacterial DNA. The adjuvant effect of CpG is enhanced when conjugated to protein antigens. This adjuvant is being tested in vaccines directed at infectious agents, allergens and tumor cells [8], [9], [10].
Another type of adjuvant is tuftsin. Tuftsin is an auto adjuvant, which is a natural self-immunostimulating tetrapeptide (Thr–Lys–Pro–Arg). This tetrapeptide is a fraction of the IgG heavy chain molecule produced by enzymatic cleavage in the spleen [11]. Its functions include: binding to receptors on neutrophils and macrophages to stimulate their phagocytic activity, increasing tumor necrosis factor alpha (TNFα) release from human Kupffer cells enhancing secretion of IL1 by activating macrophages, activation of macrophages expressing nitric oxide (NO) synthase to produce NO and enhancement of murine natural cell mediated cytotoxicity in vitro [11], [12], [13].
In summary, it is an adjuvant with minor side effects with a promising effect in restoring innate immune mediated response.
2.1.1. Mechanisms of adjuvanticity
Adjuvants may exert their immune enhancing effects according to five immune functional activities:
-
1.
Translocation of antigens to the lymph nodes where they can be recognized by T cells.
-
2.
Antigen protection enabling longer exposure.
-
3.
Enhanced local reaction at the injection site.
-
4.
Induction of the release of inflammatory cytokines.
-
5.
Interaction with PRRs, specifically TLRs [22].
-
a
Adjuvant effect
The term “adjuvant effect” refers to the co-administration of an antigen with a microbial specific factor to enhance an antigen-specific immune response in vivo. The microbial components of adjuvants activate APCs to produce pro-inflammatory cytokines (“non-specific” signal 2) and to up-regulate molecules essential for antigen presentation. These molecules include major histocompatibility complex (MHC) class II (antigen-specific signal 1) and B7-1/2. These innate immune events allow a more effective presentation to the adaptive immune system, resulting in an augmented activation and clonal expansion of T cells [23].
In accordance to this effect, if self-antigens are used, an autoimmune response can be elicited [24]. It has been shown that auto-reactive T-cells that surpass tolerance mechanisms can be triggered by exogenous adjuvants to become auto-aggressive [25].
Infectious agents are able to naturally generate their adjuvant effect and can induce autoimmunity [26]. An example of this is the causality between viral infection and myocarditis. Half the cases of myocarditis are preceded by an acute viral infection. Infectious myocarditis in humans can be reproduced in experimental murine models of myocarditis [27]. It has also been shown that the autoimmune reaction elicited by an infectious agent can be effective in treating cancer. An example of this is that bladder administration of BCG (bacille Calmette–Guérin) has been shown to be effective against superficial bladder cancer development [28]. It can be inferred that the adjuvant effect can be used against specific tumor derived molecules, so that these molecules can be recognized as “non self”.
2.1.2. Innate immune pattern recognition of pathogens and adjuvants
PRR-PAMP (Pattern Recognition Receptor—Pathogen-Associated Molecular Patterns) interactions activate the APCs to promote antigen-specific lymphocytic responses [29].
The definition of PAMPs has now broadened, in that the recognized structures do not need to be pathogens. Thus the concept of “microbe-associated molecular patterns” (MAMPs) and of “danger/damage-associated molecular patterns” (DAMPs) were defined based on the notion that the endogenous host molecules signal danger or damage to the immune system [30].
2.1.3. Innate immune response mediates the adjuvant effect
TLRs are single-transmembrane PRRs localized on cell surface and endosomal membranes. From all the PRRs, these are the most studied. TLRs play a crucial role in innate immune response to “non self” and are biosensors of tissue damage. The interaction between the four known TLRs adapters: MyD88, TIRAP/Mal, TRAM and TRIF, in TLR signaling, shape the innate immune response.
Besides PRRs the innate immune system also detects proteolytic enzymes generated during infection [31].
Merging the response to different PRRs signaling may be the pathway for developing customized responses to different aggressions [32].
-
b
Experimental models of adjuvants
Many animals have been used in experimental models of adjuvant-related autoimmune conditions [33]. These include primates, salmons, rabbits and swine; however, the most common are murine models.
Murine models include autoimmune prone strains, models of autoimmune disease and autoimmune resistant strains (Table 2 ).
Table 2.
Experimental models of adjuvant autoimmunity
| Experimental models | Strain | Disease model or related signs and symptoms | Adjuvant | |
|---|---|---|---|---|
| Murine | Rats | DA (dark agouti) rats | Rheumatoid arthritis | Mineral oil (CFA, pristane, squalene,avridine) [34], [35] |
| Arthritis | Collagen [36] | |||
| Sprague Dawley rats | Arthritis | CFA [37] | ||
| MMF | Aluminum [38] | |||
| Mice | BALB/c | Plasmacytomas | Mineral oil, pristane [39] | |
| Sclerosing lipogranulomas | SC injection of mineral oil [40] | |||
| SLE-related autoantibodies | Pristane, CFA, squalene [41] | |||
| C57BL/6 | Antiphospholipid-like syndrome | CFA, IFA [42] | ||
| NZB/NZWF1 | SLE, lupus like GLN | CFA, alum [43] | ||
| Salmons | Impaired growth rate, decreased carcass quality, spinal deformities, uveitis, inflammatory reactions in the abdominal cavity, RF, ANA, ANCA, immune-complex GLN and chronic granulomatous inflammation | Vaccines with adjuvants such as oils [44] | ||
| Rabbits | Inflammation at injection site | Vaccine: CFA, IFA, montanide [45] | ||
| Swine | Granulomatous inflammation Adverse local reactions |
Mineral oils [46] | ||
| Primates | Rhesus macaque | Potential delayed acquisition of neonatal reflexes | aluminum contained in pre clinical vaccine testing [47] |
C57BL/6 (transgenic factor V Leiden-mutated C57/BL6-back-crossed mice); RF: rheumatoid factor; ANA: antinuclear autoantibodies; ANCA: anti-cytoplasmic autoantibodies; GLN: glumerulonephritis; SLE: systemic lupus erythematous; MMF: macrophagic myofasciitis.
An interesting model is that described by Lujan et al. The authors described that a commercial sheep, inoculated repetitively with aluminum-containing adjuvants vaccinations, developed an acute neurological episode with low response to external stimuli and acute meningoencephalitis few days after immunization. An excitatory phase, followed by weakness, extreme cachexia, tetraplegia and death appeared. This was suggested to be part of the spectrum of ASIA syndrome. Moreover, the biopsy of the nervous tissue of experimental animals indicated the presence of alum [48].
-
c
Toxicity of aluminum adjuvants
Aluminum nanoparticles have both a unique capacity of surpassing the blood brain barrier (BBB) and of eliciting immune inflammatory responses. These are probably the reasons why Aluminums’ most sensitive target is the brain, and also why documented side effects are mostly neurologic or neuropsychiatric [49], [50].
Aluminum is present in nature, not only as a vaccine adjuvant, but also in food, water and cosmetics. It has been described as a neurotoxin because even when a relatively small amount of Aluminium reaches the brain [49], is can act as a genotoxin [51], a prooxidant [52], it can be proinflammatory [51], act as an immunotoxin [5] and also as an endocrine disruptor [53]. Aluminum interferes with many essential cellular processes. Memory, concentration, speech deficits, impaired psychomotor control, reduced seizure tolerance and altered behaviour are manifestations of aluminium neurotoxicity. Moreover, Alzheimer’s [54], amyotrophic lateral sclerosis, Parkinsonism dementia [55], multiple sclerosis [56], and neurological impairments in children have been linked to aluminum neurotoxicity [57].
Brain susceptibility to aluminum compounds is possibly due to the brain's high metabolic requirement, to the fact that it possesses a large area of biological membranes and to the relatively low concentration of antioxidants [54].
Aluminum adjuvants exert their immunostimulatory effect through many different pathways that activate both the innate and adaptive immune systems. One of the most significant is the activation of the NLRP3 inflammasome pathway [58]. NLPR3 activation has been shown to trigger type 2 diabetes. By using NLPR3 knockout mice it has been demonstrated that the absence of inflammasome components leads to a better maintenance of glucose homeostasis and higher insulin sensitivity [59]. On the other hand, activation of the inflammasome and its downstream components: pro-inflammatory cytokines IL-1β and IL-18 are strongly implicated in the development of several central nervous system (CNS) disorders [60].
The vast majority of people are consuming higher amounts of aluminum through dietary and parenteral intake than what expert authorities consider safe. Upper limits set by US food and drug administrations (FDA) for aluminum in vaccines are set at no more than 850 μg/dose. These values were not based on toxicity studies, but on the minimum amount needed for aluminum to exert its effect as an adjuvant [51]. The quantities of aluminum to which infants, in their first year of age are exposed, have been considered safe by the FDA. However the scientific basis for this recommendation does not take into account aluminum persistence in the body. The concern about aluminum in dietary intake has been reinforced by the Food and Agriculture (FAO) WHO Expert Committee, which lowered the provisional tolerable weekly intake of aluminum from 7 mg/kg/bw (490 mg/week, for an average 70 kg human) to 1 mg/kg/bw (70 mg/week) [61].
The amount of dietary intake of aluminum has risen in urban societies to up to 100 mg/day considering the widespread use of processed convenience foods. However, only about 0.25% of dietary aluminum is absorbed into systemic circulation and most of it is thereafter eliminated through the kidneys [54]. Absorption of aluminum by the skin from ointments and cosmetics containing aluminum has been shown. Moreover, the presence of aluminum in breast tissue was associated with breast cancer [62].
Aluminum compounds persist for up to 8–11 years post vaccination in human body. This fact, combined with repeated exposure, may account for a hyper activation of the immune system and subsequent chronic inflammation [63].
The clinical and experimental evidence collected so far identify at least three main risks associated with aluminum in vaccines:
-
1.
It can persist in the body.
-
2.
It can trigger pathological immunological responses.
-
3.
It can pass through the BBB into the CNS where it can trigger immuno-inflammatory processes, resulting in brain inflammation and long-term neural dysfunction.
2.2. Allergy and autoimmunity caused by metals
There is a link between allergies and autoimmunity since both are the result of an abnormal immune response [3], [4].
Metals such as mercury, aluminum, nickel and gold are known to induce immunotoxic effects in humans. The immunologic effects of these metals include immunomodulation, allergies and autoimmunity. They may act either as immunosuppressants or as immune adjuvants.
Metals bind firmly to cells and proteins and thus have the ability to modify autologous epitopes (hapetenization). T-cells then recognize the proteins as foreign and trigger an autoimmune response [64].
Hypersensitivity caused by metals may be referred to as Type IV delayed hypersensitivity. The reaction is considered delayed because the first symptoms appear 24–48 h after exposure, because it is mostly T-cell mediated and the gold standard for diagnosis of delayed type hypersensitivity is patch testing [65].
In mercury-sensitized patients, even mercury concentrations within the normal range might provoke neuroallergic reactions in the brain [66].
Identifying metal sensitivity and removal of the sensitizing metals, such as dental amalgam, have been proved successful by showing symptom improvement in patients with previous autoimmune diseases. These diseases included fibromyalgia, autoimmune thyroid diseases and orofacial granulomatosis [67], [68], [69], [70] (Table 3 ).
Table 3.
Metals reported side effects.
| Metal | Derivatives | Main cause of exposure | Side effects |
|---|---|---|---|
| Mercury | Methyl mercury | Skin ointments Dental amalgam fillings |
Kidney disease [71]; peripheral neuropathy; multiple sclerosis [72]; ANA positivity [73] |
| Polluted fish | |||
| Thimerosal and phenyl mercury | Antiseptics/preservatives in eye drops vaccines | Flu like symptoms Eyelid eczema and edema |
|
| Gold | Colloidal gold [74] | Treatment for RA | Nephropathy |
| Nickel [75], [76] | Food Jewelry Tobacco |
allergic and autoimmune symptoms; scleroderma-related autoantibodies and cutaneous sclerosis | |
| Aluminum [4], [77] | Food Vaccines |
Neurotoxic; delayed type hypersensitivity; ASIA syndrome; chronic fatigue syndrome; macrophagic myofasciitis |
RA: rheumatoid arthritis.
2.3. Genetics and vaccinology
The timeline regarding the field of vaccinology has been divided in two generations, the first regarding the administration of inactivated pathogens in whole or live attenuated forms (e.g., Bacillus Calmette Guerin (BCG), plague, pertussis, polio, rabies, and smallpox) and the second regarding vaccines assembled from purified microbial cell components, also referred as subunit vaccines (e.g., polysaccharides, or protein antigens) [78]. This latter approach relies on recombinant DNA technology and polysaccharide chemistry.
There are obstacles to conventional vaccine development methods such as non-cultivable in vitro pathogens (e.g., hepatitis C, papilloma virus types 16 and 18, and Mycobacterium leprae), antigen hypervariability (e.g., serogroup B meningococcus, gonococcus, malaria), opportunistic pathogens (e.g., Staphylococcus aureus) and rapid evolving pathogens such as Human immunodeficiency virus (HIV) [79].
Vaccine research gained a new perspective as the genomics field emerged over the last decades. Bacterial genomes have been sequenced and analyzed making it possible to choose the best candidate vaccine antigens by using the concept of reverse vaccinology [80].
The main known factors influencing the observed heterogeneity for immune responses induced by vaccines are gender, age, ethnicity, co-morbidity, immune system, and genetic background. The interaction between genetic and environmental components will dictate the response to vaccines.
Studying the vaccine and the host will enable the development of customized treatment options.
The combination of genetics, epidemiology and genomics in vaccine design has been denominated “vaccinomics” [81].
The importance of genetic influence is supported by twins and siblings studies, which show familial aggregation. This suggests that genomics is crucial in inter-individual variations in vaccine immune responses [82].
Both Human leukocyte antigen (HLA) and non-HLA gene markers have been identified as markers for immune response to vaccines. Multiple studies have shown connections between HLA gene polymorphisms and non-responsiveness to the HBV vaccine [83].
HLA region is divided in three sub regions: Class I is associated with the induction and maintenance of cell-mediated immune response, class II is associated with presentation of exogenous antigens to helper T CD4+ cells and class III, where immune non HLA related genes are located. Normal human tissue has at least 12HLA antigens, and although new recombinant haplotypes may occur, it is inherited mostly intact from progenitors [84].
HLA allelic differences are associated with different responses to vaccines, either by hyper or hypo responsiveness. We can infer that a similar response may be associated with different safety in relation to the development of autoimmune reactions to vaccines, particularly in the patients with genetic predisposition to an enhanced response to vaccine inoculation [85]. Furthermore, patients that share the same HLA, for instance siblings, have been diagnosed with ASIA following similar environmental stimuli [86], [87].
2.4. Autoantibodies induced by vaccines
Autoantibodies help to diagnose certain autoimmune diseases, however, they can also be found in healthy individuals. Thus, autoimmune diseases cannot be diagnosed based solely on antibody detection [88].
Inoculation of vaccines triggers autoimmune responses that result in the development of autoantibodies. Many studies have been carried out in animals, healthy subjects and patients with autoimmune diseases to understand if this development is of clinical significance [89], [90], [91], [92]. A difference in eliciting the production of autoantibodies in healthy humans has been observed between adjuvanted and non-adjuvanted influenza vaccines [93]. The annual influenza vaccine has been the most heavily researched vaccine, along with HPV and Pneumococcal vaccines as far as their relationship with patients who have previously been diagnosed with an autoimmune disease [94], [95], [96]. Autoantibody induction after HPV vaccination was also shown in adolescent girls with systemic lupus erythematosus (SLE) [97].
Although induction of autoantibodies was proven following vaccine administration, there have been no proven relation with disease diagnosis in either of the specific groups studied so far [92], [98].
It has been widely demonstrated that autoantibodies can develop years before the manifestation of a full-blown autoimmune disease [99].
Moreover, the development of a specific autoantibody is also genetically determined, and the link between genetic, autoantibodies and vaccines may become an even more intriguing area of research [100].
2.5. Siliconosis and autoimmune (auto-inflammatory) syndrome induced by adjuvants (ASIA)
Silicones are synthetic polymers that can be used as fluids, emulsions, resins and elastomers making them useful in diverse fields. They were thought to be biologically inert substances and were incorporated in a multitude of medical devices such as joint implants, artificial heart valves, catheters, drains and shunts. Of all the silicone-containing products, the most famous are most likely breast implants. Silicon is one of the substances suspected to induce ASIA [5].
It is currently believed that exposure alone is not enough to trigger the disease but that it requires the presence of additional risk factors (e.g., genetic susceptibility, other environmental factors) [4].
Silicone exerts local tissue reactions. Some of these reactions are considered para-physiological, such as capsular tissue formation around an implant. Other reactions are viewed as abnormal, like when capsular contractures and allergic reactions to silicone or platinum (catalyst used in silicone polymerization found in minute concentrations in implants) occur [101]. Cutaneous exposure to silicone with cosmetics or baby bottles could potentially sensitize patients [102].
There is also a systemic component of silicone exposure related to diffusion of silicone through the elastomer envelope, commonly termed “bleeding”. It may arouse systemic effects as it degrades and fragments in tissue, it can also spread throughout the body and lead to the development of cancer or autoimmune phenomena [103].
Patients with ruptured implants complain more frequently of pain and chronic fatigue when compared to patients with intact implants [104].
Anti-silicone antibodies were found to be present in human sera more frequently in patients who have undergone silicone breast implants, however, their pathological significance remains uncertain [105]. The same was seen for other antibodies such as autoantibodies directed against dsDNA, ssDNA, SSB/La, silicone and collagen II, which were found to be present in increased levels in patients after exposure to silicone [106].
It has also been shown that the formation of autoantibodies is directly related to implant duration.
Several autoimmune diseases have been linked to silicone exposure including rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), polymyositis, systemic sclerosis (SSc) and fibromyalgia. Although ASIA symptoms may arise 24 years after the onset of exposure to silicone implants [107], most of the follow-up periods are short and concluding evidence is yet to come regarding this causality.
2.6. Vaccines and autoimmune diseases
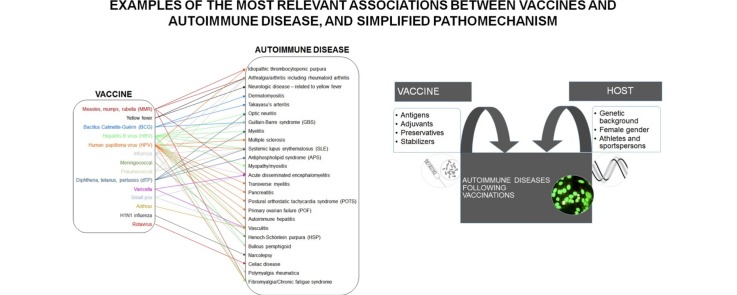
There have been published case reports, epidemiologic and research studies that suggest a connection between several vaccines and certain autoimmune conditions, notwithstanding that, overall the benefits of vaccination outweigh the risks.
3. The vaccines
3.1. Measles, mumps, rubella (MMR) vaccine
Thrombocytopenia has been reported as the main adverse event following MMR vaccine. After MMR vaccine the onset of immune thrombocytopenic purpura (ITP) usually occurred within 6 weeks at a risk rate of 1:22,000–25,000 MMR vaccine doses, while the incidence of ITP following infections is 1:6000 for measles and 1:3000 for rubella [108]. As the risk of thrombocytopenia is higher in patients who experience natural infection with measles, mumps or rubella than in those receiving the vaccine, vaccination is encouraged. Arthralgia complaints have also been reported and they may present as transient arthralgia, acute arthritis and rarely chronic arthritis [109].
Some risk factors have been found to be associated with the development of arthritis in vaccinated patients such as: female gender, older age, prior seronegativity and specific HLA alleles [110].
3.2. Yellow fever (YF) vaccine
YF vaccine is only advisable to people in, or going to endemic areas.
The risk of developing YF vaccine-associated neurologic disease (YEL-AND) is inversely proportional to age [111]. This is why children aged <6 months cannot be vaccinated and <8 months, except during epidemics [112]. Vaccination is not advisable to people >60 years because of possible higher risk of severe adverse effects (SAEs) even though the incidence remains low [113].
3.3. Bacillus Calmette-Guérin (BCG) vaccine
Besides being a vaccine for Mycobacterium tuberculosis (TB), the BCG has proved effective as immunotherapy for bladder cancer. Although the mechanism is yet to be fully understood, it is thought that BCG binds to fibronectin forming complexes that enable the recognition as “non-self” by the innate immune response of Th1 cells. Ultimately the pathways result in the apoptosis of tumor cells [114].
Because of its effect in treating non-muscle-invasive urothelial carcinoma, as well as superficial bladder tumors, it was expected that BCG could play a role in treating other types of cancer, despite data having not corroborated this hypothesis so far. Adverse events vary according to the site and method of administration. Intradermal administration of BCG has been reported to elicit arthritis [115], dermatomyositis [116] and Takayasu’s arteritis (TA) [117] among others. Intravesical treatment for bladder cancer can cause reactive arthritis (ReA) [118]. The risk relies on a systemic reaction composed of an early infective phase (PCR positive and response to anti-TB treatment) and a late hypersensitivity reaction [119].
3.4. Hepatitis B virus vaccine (HBVacc)
HBV is a DNA virus of the Hepadnaviridae family, responsible for acute and chronic liver disease.
HBV vaccines are considered the first efficient vaccines against a major human cancer. HBV vaccines have reduced the risk of developing chronic infection and they also have proved to reduce the incidence of liver cancer in children [120].
The vaccine has been associated mainly with autoimmune neuromuscular disorders. They include, but are not limited to: optic neuritis, Guillain-Barre syndrome (GBS), myelitis and multiple sclerosis (MS), systemic lupus erythematosus (SLE), arthritis, vasculitis, antiphospholipid syndrome (APS) and myopathy [121].
HBV vaccine is the most common immunization associated with acute myelitis.
There are studies that indicate that the pathogenicity behind such vaccine and autoimmunity might be based on cross-reactivity between HBV antigen (HBsAg) epitopes, yeast antigens, as well as other adjuvants contained in the vaccine itself [122].
3.5. Human papilloma virus (HPV) vaccine
Up to 90% of cervical cancer deaths, occur in developing countries that lack the ability to fully implement the Papanicolau (Pap) screening programs.
HPV poses a special challenge in vaccine safety. HPV is necessary for the development of cervical cancer. However, most women infected with HPV will not develop the disease since 70% of infections will resolve within a year and up to 90% within 2 years without specific treatment. Over the course of decades, cancer may result in a small proportion of the remaining infected women. Death rate from cervical cancer in 9–20 year old girls is zero and long-term benefits are yet to be proven. In this specific case, short term risks to healthy subjects can prove to pose a heavier burden than cervical cancer [123].
There are at least 100 types of HPV strains, 15 of which have been pathologically associated with cancer. Two vaccines, Gardasil™ and Cervarix™, are commercially available against HPV. Both contain the L1 capsid proteins of several HPV strains as antigens. Gardasil™ contains serotypes 16, 18, 6, 11. These antigens are combined with aluminum (Al) hydroxyphosphate sulphate as an adjuvant. Cervarix™ contains a combination of the oil-based adjuvant monophosphoryl lipid A (MPL) and Al hydroxide (ASO4) as adjuvant and is directed at strains 16 and 18 [124].
There have been several reports of post-licensure adverse events, some of which have even been fatal [125]. Compared to other vaccines, an unusually high proportion of adverse drug reactions has been reported associated with HPV vaccines [126].
In 2008, Australia reported an annual ADR rate of 7.3/100,000, the highest since 2003. This increase was almost entirely due to ADRs reported following the commencement of the national HPV vaccination program for females aged 12–26 years in April 2007 (705 out of a total of 1538 ADRs records). The numbers only decreased after the cessation of the catch-up schedule. Although the percentage of convulsions attributable to the HPV vaccine decreased, the overall report remained comparable between 2007 and 2009 (51% and 40% respectively). These reports do not prove the association, but show that there is a higher frequency of ADRs related to HPV vaccines reported worldwide, and that they fit a consistent pattern (i.e., nervous system-related disorders rank the highest in frequency) that deserves further investigation [126], [127], [128].
Indeed, several autoimmune diseases have been linked to HPV immunization. Examples include GBS, MS, Acute disseminated encephalomyelitis (ADEM), Transverse Myelitis (TM), postural orthostatic tachycardia syndrome (POTS), SLE, primary ovarian failure (POF), pancreatitis, vasculitis, immune thrombocytopenic purpura (ITP) and Autoimmune hepatitis (AH) [123].
3.6. Influenza
Influenza is an acute viral infection that affects the respiratory tract and is caused by influenza type A–C viruses of the Orthomyxoviridae family [129].
H1N1 mortality rates in the 2009 outbreak showed high risk in those aged 70 years and older, presence of chronic diseases and delayed admission. Risk of infection was lower in those who had been vaccinated for seasonal influenza with 2008/9 trivalent inactivated vaccine [130].
Studies have demonstrated that influenza vaccine is safe and immunogenic in patients with SLE or rheumatoid arthritis (RA), diminishing the risk of respiratory infections [129].
It has been shown that adjuvanted vaccine had more local reactions but did not increase systemic adverse reactions [131].
Molecular mimicry has been suggested as a mechanism to explain an autoimmune response following influenza vaccination. However, a causal relationship between influenza vaccines and induction of autoimmune diseases remains unproved [129].
Diseases or symptoms reported after influenza vaccination include mostly neurological syndromes such as GBS [REF]. Nonetheless, influenza vaccines should be recommended for patients with MS, because influenza infection is associated with increased risk of exacerbations.
That being said, influenza vaccinations showed increased risk of autoimmune responses suggestive of ASIA [132], vasculitis [133] and APS [134] among others.
3.7. Meningococcal vaccines
Meningococcal disease is caused by Neisseria meningitidis. One of the following five serogroups causes almost every invasive disease: A–C, Y, and W-135. Vaccines available so far for its prevention encompass either pure polysaccharide vaccines that use purified bacterial capsular polysaccharides as antigens, or protein/polysaccharide conjugate vaccines, which use the polysaccharide molecule plus diphtheria or tetanus toxoid as T-cell-stimulating antigens.
N. meningitidis serogroup B (MenB) MenB glycoconjugate vaccines are not immunogenic and hence, vaccine design has focused on sub-capsular antigens [135].
MenB capsular polysaccharide is composed of a linear homopolymer of α(2 → 8) N-acetyl-neuroaminic acid (polysialic acid; PSA).
MenB PSA and PSA found on neural cell adhesion molecules are structurally identical. As a result of this, it has been proposed that infection with MenB or vaccination with PSA may be associated with subsequent autoimmune or neurological disease [136].
No evidence of increased autoimmunity was found to be associated with meningococcal serogroup B infection [136]. Regarding vaccination, the inoculation does not cause autoimmune diseases but may unmask autoimmune phenomena in genetically predisposed individuals. Local reactions are more frequent in individuals vaccinated with quadrivalent meningococcal conjugate vaccines compared to plain polysaccharide vaccines. The intramuscular administration of the conjugate vaccine (versus subcutaneous for that of polysaccharide) may, in part, explain the higher reactivity [137].
Diseases previously associated with meningococcal vaccines are GBS [138], Henoch-Schönlein Purpura (HSP) [139] and Bullous pemphigoid (BP) [140].
3.8. Pneumococcal vaccine
Streptococcus pneumoniae (Pneumococcus) is the main cause of bacterial community-acquired pneumonia and meningitis in western countries, as well as the cause of more than 800,000 children deaths in developing countries [141], [142].
There are three anti-pneumococcal vaccines commercially available. Two of these are conjugated to a protein carrier (PCV7 and PCV13) and one is not conjugated (PPV23). PPV23 was licensed in 1983 and consists of the capsular polysaccharides of twenty-three different Streptococcus pneumoniae serotypes (1–5, 6B, 7F, 8, 9N, 9 V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19F, 19A, 20, 22F, 23F, and 33F). It does not elicit immunological memory because the immune response it triggers is T-cell independent. It is usually administered to the elderly (above 65 years), as it is believed to be less effective in children.
PCV7 is composed of the most frequent serotypes 4, 6B, 9 V, 14,18C, 19F, and 23F. PCV13 is directed at serotypes 1, 3–5, 6A, 6B, 7F, 9 V, 14, 18C, 19A, 19F, and 23F. Contrary to PPV23 both PCV7 and PCV13 have an aluminum adjuvant in their composition that elicits a T-cell mediated response [143].
Ever since vaccines were introduced in the healthcare system, prevalence, fatality and admissions for invasive pneumococcal disease have decreased significantly [144].
Vaccine adverse events vary depending on whether the vaccine is adjuvanted or not. In a non adjuvanted vaccine, local reactions are present in 9% of people vaccinated intra muscularly and in 24% of those immunized sub-cutaneously [145]. In conjugated vaccines, this percentage rises to 50% [146]. Systemic reactions such as fever, irritability, decreased appetite and sleep disturbances occur in 80–85% of recipients of PCV or PPV. Symptoms like arthralgia, arthritis, myalgia, paresthesia and fatigue are more frequent in patients post PPV. This may be related to the fact that the vaccines are administered to different age groups.
Autoimmune risk following PPV vaccine is very low. Only 14 case reports were found after PPV vaccine. Six of these referred to reactivation of a previous autoimmune disorder. Studies directed to access vaccine safety in subjects with autoimmune diseases showed immunization was safe [147], [148].
3.9. Tetanus vaccine
Tetanus toxoid (TT) is a potent exotoxin produced by the bacteria Clostridium tetani. The toxin has a predominant effect on inhibitory neurons, inhibiting release of γ-aminobutiric acid (GABA). When spinal inhibitory interneurons are affected the symptoms appear [149]. The vaccine against C. tetani contains deactivated tetanus toxoid plus an adjuvant (usually aluminium hydroxide).
The most studied and prevalent disease associated with TT is antiphospholipid syndrome (APS), but CNS complications have also been reported such as optic neuritis, acute myelitis and encephalomyelitis [150].
In mice, the immune response to TT depends on genetic background and to the specific adjuvant used for immunization. Naive BALB/c mice, immunized with TT, developed antibodies directed to TT, dsDNA and β2GPI and were extremely sick [90].
4. The diseases
4.1. Anti-phospholipid syndrome (APS)
APS is an autoimmune disease characterized by the occurrence of thrombotic events. Patients suffering from this condition have recurrent fetal loss, thromboembolic phenomena, thrombocytopenia as well as neurological, cardiac and dermatological involvement [151].
The serological marker of APS is the presence of anti-phospholipid antibodies (aPL), which bind negatively charged phospholipids, platelets and endothelial cells mainly through the plasma protein beta-2-glycoprotein-I (b2GPI). The presence of IgG and IgM anti-cardiolipin antibodies (aCL) and lupus anticoagulant is associated with thrombosis in patients with APS [151].
β2GPI was identified as the most important antigen in APS. β2GPI has several properties in vitro which define it as an anticoagulant (e.g., inhibition of prothrombinase activity, adenosine diphosphate-induced platelet aggregation, platelet factor IX production) [152]. Passive transfer of anti-β2GPI antibodies induce experimental APS in naïve mice and thrombus formation in ex vivo model [153].
Evidence suggests that the molecular mimicry mechanism between β2GPI and TT is one of the possible causes for APS.
Besides TT, APS has also been reported following HBV and influenza virus vaccination, although data are scarce [154], [155].
4.2. Systemic lupus erythematosus (SLE)
SLE is a multisystem autoimmune disease characterized by the production of a variety of autoantibodies. IgG isotype antibodies to double-stranded DNA (dsDNA) are thought to be diagnostic markers and their presence correlates with disease pathogenesis. Several factors including genetic, hormonal, environmental and immune defects are involved in the induction of autoantibodies in this disease [156].
Post vaccination manifestations of SLE or lupus like syndrome have been reported and range from autoantibody induction to full blown clinical disease. Reports have been published associating SLE to HBV, MMR, dTP, HPV, influenza, BCG, pneumococcal and small pox vaccinations [157].
Vaccination in SLE diagnosed patients is associated with disease exacerbation and decreased antibody response, which may be due to the underlying disease and the frequent use of immunosuppressive drugs [158].
A temporal link between SLE and HBV vaccination is the only relation that has been demonstrated [159].
Several studies have demonstrated an increased prevalence of HPV in individuals with lupus compared to the general population, which has increased awareness for the need to vaccinate this high-risk population [160]. To do so, the association between immunization with HPV vaccines and SLE like symptoms, as well as the higher incidence of flares in known Lupus patients must be taken into account.
4.3. Vasculitis
Vasculitis is the name given to a group of autoimmune mediated diseases, which involve blood vessels of different types and sizes. They can be categorized according to several disease features indluding: the type of vessel affected, organ distribution, genetic predisposition and clinical manifestation [161].
4.3.1. Large vessels vasculitis
So far, 18 cases of large vessel vasculitis have been detected. This includes 15 cases of giant cell arteritis (GCA) following influenza vaccination, 2 cases of Takayasu disease (TD), and one case of large cell arteritis involving subclavian and renal arteries following HBV vaccines.
Two of these patients had previous received the diagnosis of ankylosing spondylitis and polymyalgia rheumatica (PMR)-like illness [162].
4.3.2. Medium vessels vasculitis
One case of polyarteritis nodosa (PAN) following the administration of Tetanus and BCG vaccine is described. All other cases of PAN in adults follow the administration of HBV vaccine [163], [164], [165].
Case reports of medium vessels vasculitis – both polyarteritis nodosa and Kawasaki disease (KD) – have also been published in pediatric patients. KD has been described one day after the second dose of HBV vaccine and following yellow fever vaccine [166], [167]. Two cases of pediatric patients with PAN have been reported two months after receiving the HBV vaccine [164], [165].
4.3.3. Small vessels vasculitis: ANCA-associated vasculitis
Eosinophilic granulomatosis with polyangiitis (EGPA) after tetanus vaccination [163] and following HBV vaccine [168] have been reported. There are also 3 cases of microscopic polyangiitis (MPA) and 6 cases of granulomatosis with polyangiitis (GPA) following influenza vaccines in the literature [169], [170].
4.3.4. Immune complex small vessels vasculitis
Henoch Schönlein purpura (HSP) is the most common vasculitis of childhood. It is generally benign and self-limited. It is mediated by IgA immune complex deposition in various tissues as well as in small-sized blood vessels. Genetic risk factors play an important role in the pathogenesis of the disease: it is associated with HLA-DRB*01, 07 and 11. HSP was associated with seasonal influenza, influenza A (H1N1), pneumococcal and meningococcal disease, hepatitis A virus (HAV), HBV, anti-human papilloma virus (HPV) vaccines, and following multiple combinations of vaccines, such as typhoid, cholera and yellow fever [139], [171], [172], [173].
Leukocytoclastic vasculitis has been associated with several vaccines, including influenza vaccine [174], HAV vaccine [175], HBV vaccine [176], pneumococcal vaccine [177], varicella [178], rubella, smallpox [179] and the anthrax vaccine [180].
Dermal vasculitis with pan uveitis has also been described following MMR vaccine [181].
4.4. Rheumatoid arthritis (RA)
RA is the most prevalent chronic inflammatory arthritis affecting the synovial membrane of multiple diarthrodial joints. Although its etiology has not been completely clarified, deregulation of the immune system is evident with a preponderance of inflammatory cytokines and immune cells within the joints.
RA has an estimated heritability of 60%, leaving a substantial proportion of risk to environmental factors. Immunizations have previously been proposed as potential environmental triggers for RA. In the Norfolk Arthritis Register database, 19 of the first 588 patients reported receiving a tetanus vaccination within 6 weeks prior to the onset of arthritis. Similarly, a transient rise in RF titer was recorded in 10 out of 245 military recruits 2–3 weeks after receiving concomitant immunization against tetanus, typhoid, paratyphoid, mumps, diphtheria, polio and smallpox. However, only 2 showed a persistent elevation in titer and none developed arthritis [182].
Several mechanisms have been proposed to explain the putative association between vaccination and the initiation of RA, the most prominent of which are molecular mimicry and non-specific immune system activation [182].
Vaccines who have been associated with RA include rubella vaccine in which reactive arthritis occurs in 5% of recipients. Controlled studies failed to show persistent arthritis or arthralgia in these patients [110].
Patients following HBV vaccine showed an increase of arthritis in a VAERS study, but this was not seen in a large retrospective epidemiological study [183].
Data so far suggest that vaccines carry an insignificant role in the pathogenesis of RA.
4.4.1. Vaccines in the therapy of RA
Several mechanisms are being studied to produce vaccines mainly targeting inflammatory cytokines as “antigens” such as TNF, aiming to induce high titers of endogenous neutralizing anti-cytokine antibodies with the goal of breaking natural Th tolerance to auto antigens. Other cytokines, namely IL-1 IL-6, MIF, RANTES, IL-18, MCP-1 are also being tested [184].
Another vaccine related therapy uses autologous T cell lines to induce a specific immune response by the host's T cells directed against the autoimmune (vaccine) T cells [185]. This strategy has been successful in mouse models and has shown encouraging results in a small pilot study of 15 RA patients, where 10 patients showed a clinical response, defined by ACR 50 improvement criteria [186].
4.5. Undifferentiated connective tissue disease (UCTD)
UCTD is a clinical condition characterized by signs, symptoms and laboratory tests suggestive of a systemic autoimmune disease but that does not fulfill the criteria for any defined connective tissue disease (CTD).
Such patients with clinical manifestations suggestive of systemic connective tissue disease but not fulfilling any existing criteria are quite frequent: 12-20% of the patients initially asking for a rheumatologic evaluation may at least temporarily be diagnosed as affected by ‘undefined’ or ‘undifferentiated’ connective tissue disease.
Comparing studies on these diseases is unfeasible because of the inexistence of defined criteria for diagnosis [187].
Within 5 years of follow-up, patients usually evolve to defined CTDs, which include SLE, systemic sclerosis (SSc), primary Sjögren's syndrome (pSS), mixed connective tissue disease (MCTD), systemic vasculitis, poly-dermatomyositis (PM/DM) and RA. Maintaining an undefined profile for 5 years makes evolving into CTDs less probable and the diagnosis of “stable UCTD” reliable [188].
Disease etiology is a concern and it has been associated with Vitamin D deficiency and silicone implants, both of which lead to an imbalance in proinflammatory and anti-inflammatory cytokines [189].
Vaccines have also been associated with this disease, namely the HBV vaccine [190].
Etiopathogenesis of UCTD is unknown and it has been suggested it might fall on ASIA spectrum since symptomatic similarities are striking and UCTD etiopathogenesis has been associated with adjuvants [122].
4.6. Alopecia areata (AA)
AA is an autoimmune disease, characterized by one or more well demarcated oval and round non-cicatricial patches of hair loss. The disease may affect any hair bearing part of the body and has a great impact on a patient’s self-esteem and quality of life.
Depending on ethnicity and location, AA is the most prevalent skin disease. AA prevalence varies and is estimated to be between 0.1–0.2% in the United States and 3.8% in Singapore [191], [192].
As with any other autoimmune disease, the development of AA encompasses genetic and environmental factors. Environmental factors associated with AA development are emotional and/or physical stress, infections and vaccines [193].
Secondary syphilis is one of the most well studied examples, however Epstein Barr Virus [194] and Herpes Zoster [195] infections have also been related to the development of the disease.
As far as vaccines go, HBV vaccine has been associated with AA development. In one study of 60 patients, 48 developed AA after vaccination with HBV vaccine. Of those 48 patients, 16 were re-challenged, and the reappearance of disease was witnessed [196]. In mice this association failed to be established [197]. One case of AA was witnessed following Tetanus Toxoid, as well as two case reports following HPV and MMR vaccine [198], [199], [200].
4.7. Immune thrombocytopenic purpura (ITP)
ITP is an autoimmune disease defined by a platelet count of less than 105 platelets/μL without overlapping diseases. It can present with or without anti-platelet–antibodies. Thrombocytopenia is relatively common and the overall probability of developing ITP was 6,9% in a cohort of 260 patients. It was also found that 12% of patients developed an overlapping AID other than ITP [201].
The etiology of the disease is yet to be fully understood but it has been detected following infectious diseases, such as Helicobacter pylori, hepatitis C virus (HCV), novel influenza A infection, rotavirus infection and human immunodeficiency virus (HIV) [202].
ITP onset has also been reported, although rarely, as a severe adverse event following vaccine administration. This was more often observed after measles–mumps–rubella (MMR), hepatitis A and B, diphtheria–tetanus–acellular pertussis (DTaP), and varicella vaccinations [203].
Molecular mimicry has been suggested as a possible mechanism for the development of ITP, namely following Helicobacter Pylori infection. Its eradication has been shown to increase platelet count and diminish the levels of anti-CagA antibody in a subset of H. Pylori infected subjects with ITP [204].
These data point towards a beneficial role of H. pylori eradication in chronic ITP.
Two cases of ITP following anti-rabies vaccine have been reported and one after HPV vaccine. Reactivation of ITP was reported two weeks after a tick-borne encephalitis vaccination [202]. The most consistent association with ITP is with the MMR vaccine [205]. However, it should be emphasized that the number of cases are fewer than expected without vaccination.
4.8. Type 1 diabetes (T1D)
T1D is due to antigen specific reactions against insulin producing beta cells of the pancreas. Much like other autoimmune diseases, T1D results from a combination of genetic, environmental, hormonal and immunological factors. Environmental factors such as pathogens, diet, toxins, stress and vaccines are believed to be involved in the beginning of the autoimmune process [206].
Although the mechanisms by which viral infections cause autoimmune diabetes have not been fully clarified, there is some evidence to suggest a role for natural infections in the pathogenesis of T1D mellitus in susceptible individuals [207].
It has been hypothesized that vaccination could trigger T1D in susceptible individuals. Although post-vaccination T1D may be biologically plausible, cumulative evidence has not supported an increased risk of T1D following any vaccine [208].
Several experimental data have suggested that, depending on the timing, vaccination might exert a protecting or aggravating effect on the occurrence of diabetes [209].
A study suggests that Haemophillus influenza type b vaccine might be a risk factor in the induction of islet cell and anti-GAD antibodies measured at one year of age [210] but there are previous studies that show no association between Hib and T1D [211].
In a cohort of American military officers diagnosed with T1D, there was no association found between vaccination and T1D diagnosis [212].
Available data about a relation between the mumps vaccine and T1D are still incomplete and their interpretation is difficult because of miscellaneous confounding factors associated with the development of T1D [213].
Association between Hemagglutinin 1 Neuraminidase 1 (H1N1) vaccines and T1D is so far unproven [214].
In humans, it has been hypothesized that early-age BCG vaccination is associated with the risk of T1D. The few studies conducted to date provided no consistent evidence of an association. There are, however, studies showing a possible temporary boost of the immune function after vaccination [215]. Studies also show that among BCG-vaccinated children who test positive for islet autoantibodies, there is a higher cumulative risk of T1D [216].
In animal experiments it has been observed that BCG seems to have a protective effect against diabetes, however researchers have yet to translate this benefit to humans [217].
In all, studies results do not support any strong association between vaccination and T1D.
4.9. Narcolepsy
Narcolepsy is a sleep disorder described as excessive sleepiness with abnormal sleep pattern characterized by uncontrollable rapid eye movement (REM) events which occur at any time during the day. These event and may or may not be accompanied by a loss of muscle tone (cataplexy) [218].
A plethora of data indicates that narcolepsy is caused by the lack of orexin (also known as hypocretin), an important neurotransmitter, which is involved in the regulation of the sleep cycle. In Narcolepsy patients, a loss of orexin producing neurons in the hypothalamus and low levels of orexin in the cerebrospinal fluid (CSF) has been reported [218].
Narcolepsy has been shown to have an autoimmune background. Antibodies against Tribbles 2 (Trib2) have been found in these patients, which may be related to the pathogenesis of disease. An experimental model of narcolepsy in mice has been made by passive transfer of total IgG from narcolepsy patients into the animal's brains through intra ventricular injection [219].
Environmental factors like Influenza A virus and streptococcal infections have been associated with disease onset. Interestingly, fever by itself without the diagnosis of an infectious etiology was found to be a risk factor for narcolepsy [220].
Several groups have studied and found an increase in the incidence of narcolepsy diagnosis following the introduction of influenza vaccination, specifically, ASO3-adjuvanted Pandemrix™ vaccine. This association was shown in Finland especially in 4–19 year-olds, but also in case reports from other countries [221]. Other studies failed to find an association.
The actual infection with H1N1 has been associated with disease development in China, however no such relationship has been noted in Europe [220].
The above-mentioned associations are specifically related to the ASO3-adjuvanted Pandemrix™ vaccine. The same association has not been reported for other H1N1 adjuvanted or non-adjuvanted vaccines.
The major difference between the ASO3 and the MF59 adjuvants is the presence of the α-tocopherol.
α-tocopherol is unique in that it can achieve the highest and longest antibody response by producing an enhanced antigen-specific adaptive immune response. In vitro it was shown that α-tocopherol could increase the production of orexin as well as increase the proteosome activity. This increased production of orexin fragments may facilitate antigen presentation to MHC class II, thus triggering an autoimmune process [220].
All these data together support the relationship between the H1N1 Pandemrix™ vaccine and the development of narcolepsy.
4.10. Celiac disease
Gluten induced enteropathy, gluten sensitive enteropathy, or more commonly called celiac disease (CD) is a life-long autoimmune condition mainly of the gastrointestinal tract, specifically affecting the small intestine.
The abnormal immune response crates autoantigens which are directed towards Tissue transglutaminase (tTG). The two main autoantibodies and the most widespread serological markers to screen for the disease are anti tTG and anti endomysium. Two additional auto-antibodies, namely: anti deaminated gliadin peptide and anti-neoepitope tTG were found recently to be reliable for CD screening as well [222].
CD is an autoimmune disease induced by well-known nutritional environmental factors. The non-dietary ones are less studied and established. Several infectious disease have been linked to its development, the so-called infectome [193].
A clear cause-effect relation is yet to be established for most of the pathogens associated with CD. What has been shown, however, is that in countries with low economic status, inferior hygiene conditions and higher infectious load, CD prevalence is lower [223].
An epidemiologic relationship was established in 2006 between rotavirus infection and CD. Data showed that in genetically predisposed individuals, rotavirus infection was related to childhood CD development [224].
In subsequent research studies, a celiac peptide was recognized and proved to share homology with rotavirus major neutralizing protein VP7 and with the CD autoantigen tTG. The antibodies directed against the viral protein VP7 were shown to predict the onset of CD and induce typical features of CD in the intestinal epithelial cell-line T84 [225].
It has also been suggested that rotavirus vaccine alters B and T behavior, as the percentage of B-cells was higher in the vaccinated infants [226].
Rotavirus vaccine as an inducer of CD is still in discussion and warrants further study.
4.11. Polymyalgia rheumatica (PMR)
PMR is an autoimmune inflammatory rheumatic disease characterized by raised inflammatory markers with pain and morning stiffness of shoulders and pelvic girdles and synovitis of proximal joints and extra-articular synovial structures. Its diagnosis is clinical and it is typically a disease of the elderly occurring mainly in subjects above 70. Etiopathogenesis of PMR remains unknown, but genetic and environmental factors play a role [227].
A close temporal relationship has been ascertained concerning epidemics of Mycoplasma pneumoniae, Chlamydia pneumonia, Parvovirus B19 and peaks of cases of PMR and giant cell arteritis, however this is not clearly proven [228].
Cases of PMR following vaccination have rarely been reported. However, it is believed that post vaccination PMR may be underreported due to its symptomatic similarities with the transient effects of vaccines, namely: arthralgia, myalgia and low-grade fever. This leads to failure in establishing a chronological relationship when the disease is diagnosed.
Most of the reported cases are associated with seasonal influenza vaccine (Inf-V). Often, the time interval between vaccine administration and symptoms onset varies from one day, to three months. Three cases were reported with associated Giant Cell arthritis. A case report of relapsing PMR after four years of remission following tetanus vaccination has also been reported [229], [230].
4.12. Acute disseminated encephalomyelitis (ADEM)
Acute disseminated encephalomyelitis (ADEM) is an inflammatory demyelinating disease of the central nervous system (CNS).
ADEM is usually poly-symptomatic with encephalopathy (i.e., behavioral change or altered level of consciousness). It affects mostly children and young adults and has higher prevalence in males. Its incidence is 0.6–0.8 per 100 000 per year [231].
Although there is no concrete evidence of a clear pathogenic association, ADEM has been associated with immunization or previous viral infection. Post-vaccination ADEM accounts for only 5-10 percent of all cases, while post-infectious ADEM accounts for 66 percent of all cases of ADEM [232].
The hypothesis that better describes these associations is molecular mimicry. T-cells targeting human herpesvirus-6 (HHV-6), coronavirus, influenza virus and Epstein-Barr virus (EBV) have been shown to cross-react with myelin basic protein (MBP) antigens. Anti-MBP T-cells were detected in patients following vaccination with simple rabies vaccine [233], [234], [235].
In a post experimental therapy for Alzheimer’s disease with a vaccine that contained aggregates of synthetic Aβ42 fragments of amyloid precursor protein, ADEM was shown to develop in mice [236]. The experimental model of MS, EAE mice, may be induced with injection of Aβ42, but only when the latter is administered together with the complete Freund's adjuvant [237]. This observation points to the importance and central role of the adjuvants in induction of ADEM and autoimmunity in general [238].
The overall incidence of post vaccination ADEM is estimated to be 0.1–0.2 per 100 000 and a higher risk has been reported following immunization against measles. Other vaccines accountable for post-vaccination ADEM include vaccines against the varicella zoster, the rubella, the smallpox and the influenza viruses [239]. Surprisingly, certain vaccines such as anti-tetanus vaccine were shown to have a negative correlation with ADEM (statistically significant decreased risk) [240].
HBV immunization has been studied as a possible cause for ADEM but was later associated with clinically isolated syndrome (CIS) (a first time occurring demyelinating episode that may, or not develop to MS) and complete conversion to MS [241].
As far as case reports are concerned, ADEM was associated with vaccination with influenza, hepatitis A and B, MMR, HPV and tetanus [121], [242], [243].
4.13. Bullous dermatoses
Bullous dermatoses are characterized by the presence of blisters and autoantibodies against structural components of the skin: desmosomal proteins (in pemphigus), adhesion molecules of the dermal-epidermal junction (in pemphigoid diseases), and epidermal/ tissue transglutaminase (in dermatitis herpetiformis).
The most frequent autoimmune bullous diseases are bullous pemphigoid (BP) and pemphigus vulgaris (PV). BP is more frequently observed in the elderly, while the age of onset of PV is between 40 and 60 years. Neither of the diseases have any gender preference [244].
BP and PV etiology is, so far, poorly understood. Both diseases have been associated with various environmental factors, which include emotional and/or physical stress, infections and vaccinations [244].
Genetic predisposition has also been studied with overexpression of certain HLA class II alleles. These include HLA-DQB1*0301, DRB1*04, DRB1*1101, and DQB1*0302. These alleles have been found to be more prevalent in BP patients than in the general population [245]. PV is associated with certain HLA class II loci such as HLA-DR4 and HLADR14 alleles (DRB1*0401 and DRB1*0402, which is prevalent in Ashkenazi Jews, Iranian and Sardinian patients). Other loci include DRB1*1401 (common among Japanese and Italian patients) and two DQB1 alleles (DQB1*0302 and DQB1*0503), which are strongly associated with PV.
BP and PV patients’ sera were found to have significantly higher prevalence of antibodies to hepatitis B virus, hepatitis C virus, helicobacter pylori, toxoplasma gondii and cytomegalovirus [244].
As far as vaccination is concerned, BP developed in patients following influenza, diphtheria, tetanus, pertussis, hepatitis B, BCG, polio and herpes zoster vaccines [140], [246], [247] Furthermore, reactivation of BP following influenza vaccination was reported in one case report [248].
New onset PV was associated with: influenza vaccine, hepatitis B vaccine, anthrax vaccine, typhoid booster and rabies vaccination. In addition, exacerbation of PV after vaccination was also reported following influenza vaccine and tetanus vaccine [121].
4.14. Idiopathic inflammatory myopathies (IIM)
IIM compose a group of skeletal muscles diseases in which myositis without a recognized cause occurs. IIM is usually subdivided in 4 entities: dermatomyositis (DM), polymyositis (PM), inclusion body myositis (sIBM) non-specific myositis (NSM) and immune mediated necrotizing myopathy (IAM) [249].
IIM prevalence is around 1.1 × 10−6 cases, with a bimodal age of distribution that peaks in childhood and again between 45 and 55 years. DM is the most common inflammatory myopathy while PM is the least frequent.
Despite exhibiting similar clinical symptoms, the subsets of IIM exhibit significant immunopathological variation. DM begins with the activation of the complement and formation of membrane attack complexes (MAC). In PM and sIBM the fundamental process is related to CD8+ T cells mediated cytotoxicity [249].
It is unclear what breaks the tolerance and drives the immune response to induce IIM. So far, DM, PM and sIBM have been linked to vaccination. Several cases have been reported in the literature associating different vaccines with the development of idiopathic inflammatory myopathies. 119 cases of IIM had been reported to VAERS database up to June 2013. Out of these 119 cases, 33 were classified as PM, 85 as DM and an only one as a sIBM. DM has been reported after almost any vaccine, however only a few studies have attempted to clarify the possible relationship between DM and vaccination. PM is a frequent misdiagnosed disorder. Some reports have associated previous immunization, especially hepatitis B vaccine with PM [250]. Despite being recently differentiated from other IIM, sIBM has already been related to HBV vaccine [250]. Some vaccines associated with myositis are MMR vaccine, smallpox vaccine, Poliomyelitis (IPV), diphtheria and tetanus toxoid, influenza, HPV and BCG [250].
4.15. Fibromyalgia syndrome (FMS)
FMS is an entity that is related to the inability of the CNS to modulate pain.
The conditioned pain modulation process in the CNS appears to be compromised among many FMS patients, which might explain the enhanced pain sensation experienced by these patients [251].
The etiology of FMS is yet to be unveiled. Genetic predisposition, physical trauma (particularly to the cervical spine), emotional stress (to various stressors) as well as a variety of infections have been linked with FMS.
Vaccines have been associated with the triggering of FMS namely rubella and Lyme disease vaccines [252]. There are several reports of fibromyalgia-like disease after vaccination, specifically HPV (Martinez-Lavin Journal of Clinical rheumatology 2014). The medical community and regulatory agencies should be aware of these possible adverse effects aiming at defining their magnitude.
4.16. Chronic fatigue syndrome (CFS)/myalgic encephalomyelitis (ME)/systemic exertion intolerance disease (SEID)
Chronic fatigue syndrome (CFS) is a disease characterized by disabling fatigue, headaches, concentration difficulties and memory deficits (90%). Other symptoms such as sore throat (85%), tender lymph nodes (80%), skeletal muscle pain and feverishness (75%), sleep disruption (70%), psychiatric problems (65%) and rapid pulse (10%) are often observed. It more frequently affects women and has a prevalence of 0.2-2.6% [253].
Although disease etiology is still unknown, there are several pathogens, such as Epstein–Barr virus (EBV), which have been associated with CFS. Patients often have higher titers of IgM to the EBV viral capsid antigen. Cytomegalovirus and human herpes virus 6 antibodies were also detected more often in CFS patients, although other reports failed to replicate these results. Parvovirus B19 infection has also been suggested as a trigger to CFS [253], [254], [255].
Vaccine inoculation has also been appointed as a probable cause. Vaccinations against rubella, Q fever and hepatitis B were found to be associated with higher risk of developing CFS while meningococcal vaccine, poliovirus and influenza vaccine were not. Surprisingly, staphylococcus toxoid vaccine appeared to have a protective effect [121], [256], [257].
4.17. ASIA syndrome
Defined in 2011 by Shoenfeld and Agmon-Levin ASIA syndrome is characterized by hyperactive immune response to adjuvants [4].
As previously stated, ASIA incorporates four known medical conditions: Siliconosis, GWS, MMF, and post-vaccination phenomena [4]. Recently, the sick building syndrome (SBS) was proposed as a candidate for the ASIA spectrum [258]. All of these diseases satisfy several criteria for FMS and SEID [252].
-
a
Macrophagic myofasciitis (MMF)
MMF has been described as an emerging condition of unknown cause characterized by a pathognomonic lesion in muscle biopsy mixing large macrophages with submicron to micron-sized agglomerates of nanocrystals in their cytoplasm and lymphocytic infiltrates. These lesions were related to aluminum deposits in muscle following immunization with aluminum containing vaccines [63].
MMF lesion is now universally recognized as indicative of a long-lasting persistence of aluminum adjuvant at the site of prior intramuscular immunization. The long-lasting MMF lesion should be considered as a biomarker of aluminum bio persistence in a given individual.
Patients with MMF have higher reported myalgia with incidence being up to 90%. Its etiology is not clear but genuine muscle weakness is rare and the diagnosis of fibromyalgia is also rare. Higher prevalence of chronic fatigue syndrome (CFS) in patients with MMF has been reported as well.
Cognitive impairment has been associated with MMF: in one series of 105 MMF patients, up to 97% had attention and memory complaints and neuropsychological tests were abnormal in 89% [259].
-
b
Gulf War syndrome (GWS)
GWS is a clinical entity specifically related to a certain time and place in history. It was described among veterans of the military conflict occurring in 1990–1991 in the Persian Gulf.
The syndrome is characterized by chronic fatigue, musculoskeletal symptoms, malaise and cognitive impairment. It clinically overlaps with Post Traumatic Stress Disorder (PTSD), FMS, CFS and other functional disorders [260].
The unique conditions that have been associated so far with disease development are the exposure to extreme climate in the Persian Gulf, exposure to various chemicals (pesticides, depleted uranium), stress provoked by prolonged waiting without actual combat and the intense exposure to vaccinations of the soldiers for fear of biological weaponry [260].
Comparing Gulf War veterans and veterans of the Bosnian conflict, multiple vaccinations administered to servicemen in the Gulf War was identified as a unique exposure [261].
The mechanism through which vaccination exposure may lead to the development of functional symptoms is not completely understood. The possibility that a shift from Th1 to Th2 type reactions could be of pathogenic significance was raised and is supported by an increased frequency of allergic reactions, low natural killer cell activity and low levels of interferon γ and IL-2 in these patients [262].
One study with GWS patients showed a connection between anti-squalene antibodies and symptoms development. This was refuted by a larger study that found no association between anti-squalene antibodies and chronic multi-symptom illness [263].
-
c
ASIA registry
A registry is a collection of data related to patients with the same specific characteristic. It is often the first approach in the study of an area of inquiry. In rare diseases, registries are often the way to get a sufficiently sized sample of patients which can be used either for epidemiological or research purposes.
ASIA syndrome may be underreported because of unawareness and failure to connect the syndrome with the exposure. This registry was created to fully understand the clinical aspects of disease and compare patients from all over the world in order to have fully validated criteria for disease diagnosis and also to define demographic and environmental history of disease.
The ASIA Syndrome registry website can be found on the following link: https://ontocrf.costisa.com/en/web/asia. Only cases reported by physicians are accepted.
5. Vaccination in autoimmune diseases.
5.1. Autoimmune rheumatic diseases (Table 4)
Table 4.
Most common autoimmune inflammatory rheumatic diseases (AIIRDs) and non-inflammatory autoimmune rheumatic diseases (ARDs).
| AIIRDs | ARDs |
|---|---|
| Rheumatoid arthritis Ankylosing spondylitis Reactive arthritis Connective tissue diseases Polymyalgia rheumatica |
Degenerative spine diseases Osteoarthritis Osteoporosis Fibromyalgia |
To make an informed decision in medicine, there is always a need to weigh the pros and cons. ARDs may play an important role in deciding whether vaccination is or is not appropriate to a patient. In these cases, patients are immunosuppressed on account of their diagnosis and even more so if they are under specific immunomodelatory medication [4].
If the efficacy of vaccination is reduced, there is a potential for development of disease flares following vaccination. In the case of live vaccines, its inoculation may even be enough to trigger disease in the host.
For these specific reasons, live vaccines are generally contraindicated in patients receiving immunosuppressant medication. There is a need for screening and treatment of Latent Tuberculosis Infection (LTBI) before starting anti-TNF-alpha therapy. The same is true for vaccination. Preferably, even recommended vaccination (see Table 5 ) should be administered before the initiation of Disease-Modifying Anti-rheumatic Drugs (DMARDs) because these may reduce vaccine efficacy [264].
Table 5.
Vaccination recommendation in ARDs [265].
| Vaccines | Recommended | Not recommended | Special 5emarks | |
|---|---|---|---|---|
| Live | BCG | X | ||
| Herpes zoster | Previous contact with varicella (vaccine/infection) | Highly immunosuppressed patientsa | Single dose >50 y | |
| Yellow fever | Endemic areas [266] | Routine immunization not recommended | ||
| MMR | X | |||
| Non-live | Influenza | X | Allergy to egg or the vaccine itself; GBS up to 6 weeks after vaccination | Annual Rituximab: before starting/6 mts after 1st infusion/4 wks before next dose [148], [267] |
| Pneumococcal | X | 1 Initial dose + 1 booster (5 y later) | ||
| DTaP and DT | X | DTaP every 10 y Tetanus Igb if exp |
||
| Meningococcal | X | Low data support [268] | ||
| Hep A | X | |||
| Hep B | Neg HBsAg in serum | |||
| HPV | Adolescents and young women | Preferably before initiating sexual activity | ||
| Hib | X |
X – for all ARDs patients; MMR: measles, mumps and rubella; Hib: haemophilus Influenza type B; DTaP: diphteria, tetanus and pertussis; DT: diphteria and tetanus; Hep: hepatitis; Igb: immunoglobulin; y: years; mts: months; wks: weeks; HPV: human papilloma virus; GBS: guillain barré syndrome; exp: exposure; Neg HBsAG: negative hepatitis B antigen.
Highly immunosuppressed patients: high doses of corticosteroids (>20 mg of prednisone per day or equivalent) for 2 weeks or longer, pulse therapy, cytotoxic or alkylating agents, synthetic DMARDs at doses above those recommended, or immunobiological therapy [264].
5.2. Autoimmune inflammatory rheumatic diseases (AIIRD)
Immunosuppression equals high risk of infection and lower vaccine efficacy.
Taking into account safety concerns and efficacy, the EULAR recommendations for immunizations in AIIRD patients are:
-
•
Assess vaccination status in initial investigation.
-
•
Administer vaccines in a stable disease phase.
-
•
Live attenuated vaccines are to be avoided especially if immunosuppressive agents are being administered. BCG is not recommended.
-
•
Administer vaccines ideally before starting DMARDs and anti-TNFα agents.
-
•
Influenza and 23-valent polysaccharide pneumococcal vaccination is recommended.
-
•
Tetanus toxoid vaccination is recommended following recommendations of general population, in case of major and/or contaminated wounds in patients receiving rituximab in the previous 24 weeks Tetanus Ig is indicated.
-
•
HPV and Herpes Zoster should be considered.
-
•
In hyposplenic/asplenic patients, influenza, pneumococcal, Haemophilus Influenza b and Meningococcal C are advisable.
-
•
Hepatitis A and B is recommended in patients at risk.
-
•
Travel patients should be immunized according to general population guidelines except for live attenuated vaccines, which are to be avoided [148].
6. Conclusions and future perspectives
Vaccines have many beneficial effects in combating infectious diseases and preventing mortality and morbidity. They have also proved to be effective cancer treatments by immunomodulation, as demonstrated by the intravesical administration of BCG to treat superficial bladder cancer [28].
Vaccines are however, linked to autoimmunity. Beneficial outcomes, like the adjuvant effect are based on immunity triggering and enhanced immunity mechanisms. These same responses account for autoimmunity exertion. Vaccines induce the production of autoantibodies, but their pathologic effect is yet to be unveiled.
Although vaccines are widely considered safe, there are subjects with predispositions to whom vaccines pose a bigger threat. An example is the fact that animal models with autoimmune predispositions develop autoimmune disease following adjuvant exposure.
As many as 1% of recipients of aluminum containing adjuvants may be sensitized to future exposure [269].
Silicon-induced inflammatory fibro proliferative response is irrefutable and well documented. The presence of anti-silicone antibodies and silicone-associated autoimmune phenomena seems very plausible.
ASIA syndrome and aluminum safety studies show that the use of aluminum containing “placebo” in control groups in vaccine safety studies should be carefully evaluated. New studies must be performed using a proper placebo to adequately test vaccine safety. Another evident failure in vaccine safety studies are the short-term periods which are evaluated. Continued immune system activation has been observed to be a potential mechanism of disease. A disease which is poorly understood so far.
Vaccine recommendations should be reassessed frequently in different subsets of the population. This does not invalidate the need for vaccines, however, the lower the possibility of exerting adverse events, the easier it will be for the potential benefits to outweigh the risks.
Vaccinomics represents a major breakthrough in vaccine development and can lead to the development of targeted vaccines to peptides most likely to be immunogenic [81]. A predictable response to vaccine can be achieved by differentiating the host variability. This can be achieved namely in genetics and pathogen variability. Developing a vaccine accordingly will lead to increased specificity in treatment and leave less room for adverse events. By using immunomodulation, vaccinomics can also give rise to novel therapies for autoimmune diseases.
There are several reports of cases of autoimmunity diseases following vaccines but despite in vitro positive results and due to both the limited number of cases and the long latency period of the diseases, every attempt for an epidemiological study has failed to deliver a connection.
Classification as ASIA syndrome, in detriment of classic specific autoimmune diseases, could be the key to finding effective preventative therapeutic strategies. It will enable the study of bigger patient clusters with earlier diagnoses.
Future studies that could help clarify the association between vaccinations, adjuvants and autoimmunity should ideally have a different design, more long-term data and should include autoimmune phenomena as well as large-scale epidemiological studies of autoimmune diseases.
References
- 1.Shoenfeld Y., Isenberg D.A. The mosaic of autoimmunity. Immunol. Today. 1989;10(4):123–126. doi: 10.1016/0167-5699(89)90245-4. http://www.ncbi.nlm.nih.gov/pubmed/2665774 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 2.Kivity S., Agmon-Levin N., Blank M. Infections and autoimmunity—friends or foes? Trends Immunol. 2009;30(8):409–414. doi: 10.1016/j.it.2009.05.005. http://www.ncbi.nlm.nih.gov/pubmed/19643667 [Internet] (cited 24.02.15) [DOI] [PubMed] [Google Scholar]
- 3.Perricone C., Colafrancesco S., Mazor R.D. Autoimmune/inflammatory syndrome induced by adjuvants (ASIA) 2013: unveiling the pathogenic, clinical and diagnostic aspects. J. Autoimmun. 2013;47:1–16. doi: 10.1016/j.jaut.2013.10.004. http://www.ncbi.nlm.nih.gov/pubmed/24238833 [Internet] (cited 14.01.15) [DOI] [PubMed] [Google Scholar]
- 4.Shoenfeld Y., Agmon-Levin N. ASIA—autoimmune/inflammatory syndrome induced by adjuvants. J. Autoimmun. 2011;36(1):1–16. doi: 10.1016/j.jaut.2010.07.003. http://www.ncbi.nlm.nih.gov/pubmed/20708902 [Internet] (cited 23.02.15) [DOI] [PubMed] [Google Scholar]
- 5.Israeli E., Agmon-Levin N., Blank M. Adjuvants and autoimmunity. Lupus. 2009;18(13):1217–1225. doi: 10.1177/0961203309345724. http://www.ncbi.nlm.nih.gov/pubmed/19880572 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 6.Glück R. Adjuvant activity of immunopotentiating reconstituted influenza virosomes (IRIVs) Vaccine. 1999;17(13–14):1782–1787. doi: 10.1016/s0264-410x(98)00440-x. http://www.ncbi.nlm.nih.gov/pubmed/10194840 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 7.Beutler B. Interfaces questions and possibilities in Toll-like receptor signalling. Nature. 2004;430(6996):257–263. doi: 10.1038/nature02761. http://www.ncbi.nlm.nih.gov/pubmed/15241424 [Internet] (cited 23.03.15) [DOI] [PubMed] [Google Scholar]
- 8.Creticos P.S., Schroeder J.T., Hamilton R.G. Immunotherapy with a ragweed-toll-like receptor 9 agonist vaccine for allergic rhinitis. N. Engl. J. Med. 2006;355(14):1445–1455. doi: 10.1056/NEJMoa052916. http://www.ncbi.nlm.nih.gov/pubmed/17021320 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 9.Alexeev V., Mucci T., Igoucheva O. Immunotherapeutic strategies for the treatment of malignant melanoma. G. Ital. Dermatol. Venereol. 2008;143(2):139–149. http://www.ncbi.nlm.nih.gov/pubmed/18833040 [Internet] (cited 22.04.15) [PubMed] [Google Scholar]
- 10.Kirkwood J.M., Tawbi H.A., Tarhini A.A. Does pegylated interferon alpha-2b confer additional benefit in the adjuvant treatment of high-risk melanoma? Nat. Clin. Pract. Oncol. 2015;6(2):70–71. doi: 10.1038/ncponc1297. http://www.ncbi.nlm.nih.gov/pubmed/19092800 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 11.Siemion I.Z., Kluczyk A. Tuftsin on the 30-year anniversary of Victor Najjar’s discovery. Peptides. 1999;20(5):645–674. doi: 10.1016/s0196-9781(99)00019-4. http://www.ncbi.nlm.nih.gov/pubmed/10465518 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 12.Phillips J.H., Babcock G.F., Nishioka K. Tuftsin a naturally occurring immunopotentiating factor. I. In vitro enhancement of murine natural cell-mediated cytotoxicity. J. Immunol. 1981;126(3):915–921. http://www.ncbi.nlm.nih.gov/pubmed/6893996 [Internet] (cited 22.04.15) [PubMed] [Google Scholar]
- 13.Dagan S., Tzehoval E., Fridkin M. Tuftsin and tuftsin conjugates potentiate immunogenic processes: effects and possible mechanisms. J. Biol. Response Mod. 1987;6(6):625–636. http://www.ncbi.nlm.nih.gov/pubmed/3502474 [Internet] (cited 22.04.15) [PubMed] [Google Scholar]
- 14.Riedl K., Riedl R., von Gabain A. The novel adjuvant IC31 strongly improves influenza vaccine-specific cellular and humoral immune responses in young adult and aged mice. Vaccine. 2008;26(27–28):3461–3468. doi: 10.1016/j.vaccine.2008.04.029. http://www.ncbi.nlm.nih.gov/pubmed/18495302 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 15.Pichichero M.E. Improving vaccine delivery using novel adjuvant systems. Hum. Vaccin. 2015;4(4):262–270. doi: 10.4161/hv.4.4.5742. http://www.ncbi.nlm.nih.gov/pubmed/18398303 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 16.Ghochikyan A., Mkrtichyan M., Petrushina I. Prototype Alzheimer’s disease epitope vaccine induced strong Th2-type anti-Abeta antibody response with Alum to Quil A adjuvant switch. Vaccine. 2006;24(13):2275–2282. doi: 10.1016/j.vaccine.2005.11.039. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2081151&tool=pmcentrez&rendertype=Abstract [Internet] (cited 22.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zimmermann S., Egeter O., Hausmann S. CpG oligodeoxynucleotides trigger protective and curative Th1 responses in lethal murine leishmaniasis. J. Immunol. 1998;160(8):3627–3630. http://www.ncbi.nlm.nih.gov/pubmed/9558060 [Internet] (cited 22.04.15) [PubMed] [Google Scholar]
- 18.Fridkin M., Tsubery H., Tzehoval E. Tuftsin-AZT conjugate: potential macrophage targeting for AIDS therapy. J. Pept. Sci. 2005;11(1):37–44. doi: 10.1002/psc.587. http://www.ncbi.nlm.nih.gov/pubmed/15635725 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 19.Liu X., Guo J., Han S. Enhanced immune response induced by a potential influenza A vaccine based on branched M2e polypeptides linked to tuftsin. Vaccine. 2012;30(46):6527–6533. doi: 10.1016/j.vaccine.2012.08.054. http://www.ncbi.nlm.nih.gov/pubmed/22959982 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 20.Pawan K., Ivanov B.B., Kabilan L. Construction of a synthetic immunogen: use of the natural immunomodulator polytuftsin in malaria vaccines against RESA antigen of Plasmodium falciparum. Vaccine. 1994;12(9):819–824. doi: 10.1016/0264-410x(94)90291-7. http://www.ncbi.nlm.nih.gov/pubmed/7526572 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 21.Lukács K., Szabó G., Sonkoly I. Stimulating effect of tuftsin and its analogues on the defective monocyte chemotaxis in systemic lupus erythematosus. Immunopharmacology. 1984;7(3–4):171–178. doi: 10.1016/0162-3109(84)90034-1. http://www.ncbi.nlm.nih.gov/pubmed/6469603 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 22.Schijns V.E. Immunological concepts of vaccine adjuvant activity. Curr. Opin. Immunol. 2000;12(4):456–463. doi: 10.1016/s0952-7915(00)00120-5. http://www.ncbi.nlm.nih.gov/pubmed/10899018 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 23.O'Hagan D.T., Valiante N.M. Recent advances in the discovery and delivery of vaccine adjuvants. Nat. Rev. Drug Discov. 2003;2(9):727–735. doi: 10.1038/nrd1176. http://www.ncbi.nlm.nih.gov/pubmed/12951579 [Internet] (cited 30.03.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rangachari M., Kuchroo V.K. Using EAE to better understand principles of immune function and autoimmune pathology. J. Autoimmun. 2013;45:31–39. doi: 10.1016/j.jaut.2013.06.008. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3963137&tool=pmcentrez&rendertype=Abstract [Internet] (cited 11.02.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pasare C., Medzhitov R. Toll pathway-dependent blockade of CD4 + CD25 + T cell-mediated suppression by dendritic cells. Science. 2003;299(5609):1033–1036. doi: 10.1126/science.1078231. http://www.ncbi.nlm.nih.gov/pubmed/12532024 [Internet] (cited 10.03.15) [DOI] [PubMed] [Google Scholar]
- 26.Fairweather D., Frisancho-Kiss S., Rose N.R. Viruses as adjuvants for autoimmunity: evidence from Coxsackievirus-induced myocarditis. Rev. Med. Virol. 2015;15(1):17–27. doi: 10.1002/rmv.445. http://www.ncbi.nlm.nih.gov/pubmed/15386590 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 27.Cihakova D., Rose N.R. Pathogenesis of myocarditis and dilated cardiomyopathy. Adv. Immunol. 2008;99:95–114. doi: 10.1016/S0065-2776(08)00604-4. http://www.ncbi.nlm.nih.gov/pubmed/19117533 [Internet] (cited 20.04.15] [DOI] [PubMed] [Google Scholar]
- 28.Lamm D.L., Thor D.E., Harris S.C. Bacillus calmette–guerin immunotherapy of superficial bladder cancer. J. Urol. 1980;124(1):38–40. doi: 10.1016/s0022-5347(17)55282-9. http://www.ncbi.nlm.nih.gov/pubmed/6997513 [Internet] (cited 16.03.15] [DOI] [PubMed] [Google Scholar]
- 29.Palm N.W., Medzhitov R. Pattern recognition receptors and control of adaptive immunity. Immunol. Rev. 2009;227(1):221–233. doi: 10.1111/j.1600-065X.2008.00731.x. http://www.ncbi.nlm.nih.gov/pubmed/19120487 [Internet] (cited 12.03.15] [DOI] [PubMed] [Google Scholar]
- 30.Kono H., Rock K.L. How dying cells alert the immune system to danger. Nat. Rev. Immunol. 2008;8(4):279–289. doi: 10.1038/nri2215. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2763408&tool=pmcentrez&rendertype=Abstract [Internet] (cited 10.07.14] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shpacovitch V., Feld M., Hollenberg M.D. Role of protease-activated receptors in inflammatory responses, innate and adaptive immunity. J. Leukoc. Biol. 2008;83(6):1309–1322. doi: 10.1189/jlb.0108001. http://www.ncbi.nlm.nih.gov/pubmed/18347074 [Internet] (cited 17.04.15] [DOI] [PubMed] [Google Scholar]
- 32.Nhu Q.M., Shirey K., Teijaro J.R. Novel signaling interactions between proteinase-activated receptor 2 and Toll-like receptors in vitro and in vivo. Mucosal. Immunol. 2010;3(1):29–39. doi: 10.1038/mi.2009.120. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2851245&tool=pmcentrez&rendertype=Abstract [Internet] (cited 22.04.15] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cruz-Tapias P., Agmon-Levin N., Israeli E. Autoimmune (auto-inflammatory) syndrome induced by adjuvants (ASIA)—animal models as a proof of concept. Curr. Med. Chem. 2013;20(32):4030–4036. doi: 10.2174/09298673113209990253. http://www.ncbi.nlm.nih.gov/pubmed/23992328 [Internet] [cited 31.03.15] [DOI] [PubMed] [Google Scholar]
- 34.Carlson B.C., Jansson A.M., Larsson A. The endogenous adjuvant squalene can induce a chronic T-cell-mediated arthritis in rats. Am. J. Pathol. 2000;156(6):2057–2065. doi: 10.1016/S0002-9440(10)65077-8. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1850095&tool=pmcentrez&rendertype=Abstract [Internet] [cited 22.04.15] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kleinau S., Erlandsson H., Klareskog L. Percutaneous exposure of adjuvant oil causes arthritis in DA rats. Clin. Exp. Immunol. 1994;96(2):281–284. doi: 10.1111/j.1365-2249.1994.tb06554.x. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1534890&tool=pmcentrez&rendertype=Abstract [Internet] (cited 22.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Griffiths M.M., Sawitzke A.D., Harper D.S. Exacerbation of collagen-induced arthritis in rats by rat cytomegalovirus is antigen-specific. Autoimmunity. 1994;18(3):177–187. doi: 10.3109/08916939409007994. http://www.ncbi.nlm.nih.gov/pubmed/7858103 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 37.Bersani-Amado C.A., Barbuto J.A., Jancar S. Comparative study of adjuvant induced arthritis in susceptible and resistant strains of rats. I. Effect of cyclophosphamide. J. Rheumatol. 1990;17(2):149–152. http://www.ncbi.nlm.nih.gov/pubmed/2138672 [Internet] (cited 22.04.15) [PubMed] [Google Scholar]
- 38.Authier F.-J., Sauvat S., Christov C. AlOH3-adjuvanted vaccine-induced macrophagic myofasciitis in rats is influenced by the genetic background. Neuromuscul. Disord. 2006;16(5):347–352. doi: 10.1016/j.nmd.2006.02.004. http://www.ncbi.nlm.nih.gov/pubmed/16616846 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 39.Anderson P.N., Potter M. Induction of plasma cell tumours in BALB-c mice with 2,6,10,14-tetramethylpentadecane (pristane) Nature. 1969;222(5197):994–995. doi: 10.1038/222994a0. http://www.ncbi.nlm.nih.gov/pubmed/5789334 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 40.Di B., enedetto G., Pierangeli M., Scalise A. Paraffin oil injection in the body: an obsolete and destructive procedure. Ann. Plast. Surg. 2002;49(4):391–396. doi: 10.1097/00000637-200210000-00010. http://www.ncbi.nlm.nih.gov/pubmed/12370645 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 41.Satoh M., Kuroda Y., Yoshida H. Induction of lupus autoantibodies by adjuvants. J. Autoimmun. 2003;21(1):1–9. doi: 10.1016/s0896-8411(03)00083-0. http://www.ncbi.nlm.nih.gov/pubmed/12892730 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 42.Katzav A., Kivity S., Blank M. Adjuvant immunization induces high levels of pathogenic antiphospholipid antibodies in genetically prone mice: another facet of the ASIA syndrome. Lupus. 2012;21(2):210–216. doi: 10.1177/0961203311429550. http://www.ncbi.nlm.nih.gov/pubmed/22235055 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 43.Bassi N., Luisetto R., Del Prete D. Induction of the ASIA syndrome in NZB/NZWF1 mice after injection of complete Freund’s adjuvant (CFA) Lupus. 2012;21(2):203–209. doi: 10.1177/0961203311429553. http://www.ncbi.nlm.nih.gov/pubmed/22235054 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 44.Haugarvoll E., Bjerkås I., Szabo N.J. Manifestations of systemic autoimmunity in vaccinated salmon. Vaccine. 2010;28(31):4961–4969. doi: 10.1016/j.vaccine.2010.05.032. http://www.ncbi.nlm.nih.gov/pubmed/20553770 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 45.Fodey T.L., Delahaut P., Charlier C. Comparison of three adjuvants used to produce polyclonal antibodies to veterinary drugs. Vet. Immunol. Immunopathol. 2008;122(1–2):25–34. doi: 10.1016/j.vetimm.2007.10.016. http://www.ncbi.nlm.nih.gov/pubmed/18063100 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 46.Straw B.E., MacLachlan N.J., Corbett W.T. Comparison of tissue reactions produced by Haemophilus pleuropneumoniae vaccines made with six different adjuvants in swine. Can. J. Comp. Med. 1985;49(2):149–151. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1236138&tool=pmcentrez&rendertype=Abstract [Internet] (cited 22.04.15) [PMC free article] [PubMed] [Google Scholar]
- 47.Hewitson L., Houser L.A., Stott C. Delayed acquisition of neonatal reflexes in newborn primates receiving a thimerosal-containing hepatitis B vaccine: influence of gestational age and birth weight. AJ. Toxicol. Environ. Health A. 2010;73(19):1298–1313. doi: 10.1080/15287394.2010.484709. http://www.ncbi.nlm.nih.gov/pubmed/20711932 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 48.Luján L., Pérez M., Salazar E. Autoimmune/autoinflammatory syndrome induced by adjuvants (ASIA syndrome) in commercial sheep. Immunol. Res. 2013;56(2–3):317–324. doi: 10.1007/s12026-013-8404-0. http://www.ncbi.nlm.nih.gov/pubmed/23579772 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 49.Khan Z., Combadière C., Authier F.-J. Slow CCL2-dependent translocation of biopersistent particles from muscle to brain. BMC Med. 2013;11:99. doi: 10.1186/1741-7015-11-99. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3616851&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shaw C.A., Petrik M.S. Aluminum hydroxide injections lead to motor deficits and motor neuron degeneration. J. Inorg. Biochem. 2009;103(11):1555–1562. doi: 10.1016/j.jinorgbio.2009.05.019. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2819810&tool=pmcentrez&rendertype=Abstract [Internet] (cited 22.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lukiw W.J., Percy M.E., Kruck T.P. Nanomolar aluminum induces pro-inflammatory and pro-apoptotic gene expression in human brain cells in primary culture. J. Inorg. Biochem. 2005;99(9):1895–1898. doi: 10.1016/j.jinorgbio.2005.04.021. http://www.ncbi.nlm.nih.gov/pubmed/15961160 [Internet] (cited 14.04.15) [DOI] [PubMed] [Google Scholar]
- 52.Exley C. The pro-oxidant activity of aluminum. Free Radic Biol. Med. 2004;36(3):380–387. doi: 10.1016/j.freeradbiomed.2003.11.017. http://www.ncbi.nlm.nih.gov/pubmed/15036357 [Internet] (cited 10.04.15) [DOI] [PubMed] [Google Scholar]
- 53.Singla N., Dhawan D.K. Regulatory role of zinc during aluminium-induced altered carbohydrate metabolism in rat brain. J. Neurosci. Res. 2012;90(3):698–705. doi: 10.1002/jnr.22790. http://www.ncbi.nlm.nih.gov/pubmed/22108899 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 54.Tomljenovic L. Aluminum and Alzheimer’s disease: after a century of controversy, is there a plausible link? J. Alzheimers Dis. 2011;23(4):567–598. doi: 10.3233/JAD-2010-101494. http://www.ncbi.nlm.nih.gov/pubmed/21157018 [Internet] (cited 10.04.15) [DOI] [PubMed] [Google Scholar]
- 55.Perl D.P. Exposure to aluminium and the subsequent development of a disorder with features of Alzheimer's disease. J. Neurol. Neurosurg. Psychiatry. 2006;77(7):811. doi: 10.1136/jnnp.2006.090613. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2117484&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Exley C., Mamutse G., Korchazhkina O. Elevated urinary excretion of aluminium and iron in multiple sclerosis. Mult Scler. 2006;12(5):533–540. doi: 10.1177/1352458506071323. http://www.ncbi.nlm.nih.gov/pubmed/17086897 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 57.Shaw C.A., Seneff S., Kette S.D. Aluminum-induced entropy in biological systems: implications for neurological disease. J. Toxicol. 2014;2014:1. doi: 10.1155/2014/491316. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4202242&tool=pmcentrez&rendertype=Abstract [Internet] (cited 24.03.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Exley C., Siesjö P., Eriksson H. The immunobiology of aluminium adjuvants: how do they really work? Trends Immunol. 2010;31(3):103–109. doi: 10.1016/j.it.2009.12.009. http://www.cell.com/article/S1471490609002488/fulltext [Internet] (cited 20.04.15) [DOI] [PubMed] [Google Scholar]
- 59.Wen H., Gris D., Lei Y. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 2011;12(5):408–415. doi: 10.1038/ni.2022. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4090391&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chakraborty S., Kaushik D.K., Gupta M. Inflammasome signaling at the heart of central nervous system pathology. J. Neurosci. Res. 2010;88(8):1615–1631. doi: 10.1002/jnr.22343. http://www.ncbi.nlm.nih.gov/pubmed/20127816 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 61.FAO/WHO (Food and Agriculture Organization/World Health Organization) Summary and conclusions of the sixty-seventh meeting of the Joint FAO/WHO Expert Committee on Food Additives. JECFA. 2015 ftp://ftp.fao.org/ag/agn/jecfa/jecfa67_final.pdf [Internet] (cited 30.06.15) [Google Scholar]
- 62.Pineau A., Fauconneau B., Sappino A.-P. If exposure to aluminium in antiperspirants presents health risks, its content should be reduced. J. Trace Elem. Med. Biol. 2014;28(2):147–150. doi: 10.1016/j.jtemb.2013.12.002. http://www.ncbi.nlm.nih.gov/pubmed/24418462 [Internet] (cited 06.06.15) [DOI] [PubMed] [Google Scholar]
- 63.Gherardi R.K., Coquet M., Cherin P. Macrophagic myofasciitis lesions assess long-term persistence of vaccine-derived aluminium hydroxide in muscle. Brain. 2001;124:1821–1831. doi: 10.1093/brain/124.9.1821. http://www.ncbi.nlm.nih.gov/pubmed/11522584 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 64.Wang Y., Dai S. Structural basis of metal hypersensitivity. Immunol. Res. 2013;55(1–3):83–90. doi: 10.1007/s12026-012-8351-1. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4040395&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Stejskal V., Hudecek R., Stejskal J. Diagnosis and treatment of metal-induced side-effects. Neuro Endocrinol. Lett. 2006;27(Suppl. 1):7–16. http://www.ncbi.nlm.nih.gov/pubmed/17261999 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 66.Clausen J. Mercury and multiple sclerosis. Acta Neurol. Scand. 1993;87(6):461–464. doi: 10.1111/j.1600-0404.1993.tb04137.x. http://www.ncbi.nlm.nih.gov/pubmed/8356875 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 67.Prochazkova J., Sterzl I., Kucerova H. The beneficial effect of amalgam replacement on health in patients with autoimmunity. Neuro Endocrinol. Lett. 2004;25(3):211–218. http://www.ncbi.nlm.nih.gov/pubmed/15349088 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 68.Stejskal V., Ockert K., Bjørklund G. Metal-induced inflammation triggers fibromyalgia in metal-allergic patients. Neuro Endocrinol. Lett. 2013;34(6):559–565. http://www.ncbi.nlm.nih.gov/pubmed/24378456 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 69.Hybenova M., Hrda P., Procházková J. The role of environmental factors in autoimmune thyroiditis. Neuro Endocrinol. Lett. 2015;31(3):283–289. http://www.ncbi.nlm.nih.gov/pubmed/20588228 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 70.Tomka M., Machovcová A., Pelclová D. Orofacial granulomatosis associated with hypersensitivity to dental amalgam. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011;112(3):335–341. doi: 10.1016/j.tripleo.2011.03.030. http://www.ncbi.nlm.nih.gov/pubmed/21684771 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 71.Turk J.L., Baker H. Nephrotic syndrome due to ammoniated mercury. Br. J. Dermatol. 1968;80(9):623–624. http://www.ncbi.nlm.nih.gov/pubmed/5676001 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 72.Ingalls T.H. Endemic clustering of multiple sclerosis in time and place, 1934–1984. Confirmation of a hypothesis. Am. J. Forensic Med. Pathol. 1986;7(1):3–8. doi: 10.1097/00000433-198603000-00002. http://www.ncbi.nlm.nih.gov/pubmed/3728417 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 73.Somers E.C., Ganser M.A., Warren J.S. Mercury exposure and antinuclear antibodies among females of reproductive age in the United States: NHANES. Environ. Health Perspect. 2015 doi: 10.1289/ehp.1408751. http://www.ncbi.nlm.nih.gov/pubmed/25665152 [Internet] (cited 10.05.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Palosuo T., Provost T.T., Milgrom F. Gold nephropathy: serologic data suggesting an immune complex disease. Clin. Exp. Immunol. 1976;25(2):311–318. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1541350&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [PMC free article] [PubMed] [Google Scholar]
- 75.Al-Mogairen S.M., Meo S.A., Al-Arfaj A.S. Nickel-induced allergy and contact dermatitis: does it induce autoimmunity and cutaneous sclerosis? An experimental study in Brown Norway rats. Rheumatol. Int. 2010;30(9):1159–1164. doi: 10.1007/s00296-009-1117-y. http://www.ncbi.nlm.nih.gov/pubmed/19784841 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 76.US Environmental Protection Agency, Health Assessment Document for Nickel, Washington, DC (1986).
- 77.Shaw C.A., Tomljenovic L. Aluminum in the central nervous system (CNS): toxicity in humans and animals, vaccine adjuvants, and autoimmunity. Immunol. Res. 2013;56(2–3):304–316. doi: 10.1007/s12026-013-8403-1. http://www.ncbi.nlm.nih.gov/pubmed/23609067 [Internet] (cited 14.04.15) [DOI] [PubMed] [Google Scholar]
- 78.Plotkin S.A. Six revolutions in vaccinology. Pediatr. Infect. Dis. J. 2015;24(1):1–9. doi: 10.1097/01.inf.0000148933.08301.02. http://www.ncbi.nlm.nih.gov/pubmed/15665703 [Internet] (cited 264.04.15) [DOI] [PubMed] [Google Scholar]
- 79.Rappuoli R. From Pasteur to genomics: progress and challenges in infectious diseases. Nat. Med. 2004;10(11):1177–1185. doi: 10.1038/nm1129. http://www.ncbi.nlm.nih.gov/pubmed/15516917 [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pulendran B., Li S., Nakaya H.I. Systems vaccinology. Immunity. 2010;33(4):516–529. doi: 10.1016/j.immuni.2010.10.006. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3001343&tool=pmcentrez&rendertype=Abstract [Internet] (cited 23.024.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Poland G.A., Kennedy R.B., McKinney B.A. Vaccinomics, adversomics, and the immune response network theory: individualized vaccinology in the 21st century. Semin Immunol. 2013;25(2):89–103. doi: 10.1016/j.smim.2013.04.007. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3752773&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Jacobson R.M., Ovsyannikova I.G., Targonski P.V. Studies of twins in vaccinology. Vaccine. 2007;25(16):3160–3164. doi: 10.1016/j.vaccine.2007.01.048. http://www.ncbi.nlm.nih.gov/pubmed/17284336 [Internet] (cited 20.04.15) [DOI] [PubMed] [Google Scholar]
- 83.Ovsyannikova I.G., Poland G.A. Vaccinomics current findings, challenges and novel approaches for vaccine development. AAPS J. 2011;13(3):438–444. doi: 10.1208/s12248-011-9281-x. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3160164&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Shiina T., Hosomichi K., Inoko H. The HLA genomic loci map: expression, interaction, diversity and disease. J. Hum. Genet. 2009;54(1):15–39. doi: 10.1038/jhg.2008.5. http://www.ncbi.nlm.nih.gov/pubmed/19158813 [Internet] (cited 28.01.15) [DOI] [PubMed] [Google Scholar]
- 85.Posteraro B., Pastorino R., Di Giannantonio P. The link between genetic variation and variability in vaccine responses: systematic review and meta-analyses. Vaccine. 2014;32(15):1661–1669. doi: 10.1016/j.vaccine.2014.01.057. http://www.ncbi.nlm.nih.gov/pubmed/24513009 [Internet] (cited 26.03.14) [DOI] [PubMed] [Google Scholar]
- 86.Meier L.G., Barthel H.R., Seidl C. Development of polyarthritis after insertion of silicone breast implants followed by remission after implant removal in 2HLA-identical sisters bearing rheumatoid arthritis susceptibility genes. J. Rheumatol. 1997;24(9):1838–1841. http://www.ncbi.nlm.nih.gov/pubmed/9292814 [Internet] (cited 30.06.15) [PubMed] [Google Scholar]
- 87.Anaya J.-M., Reyes B., Perdomo-Arciniegas A.M. Autoimmune/auto-inflammatory syndrome induced by adjuvants (ASIA) after quadrivalent human papillomavirus vaccination in Colombians: a call for personalised medicine. Clin. Exp. Rheumatol. 2015 http://www.ncbi.nlm.nih.gov/pubmed/25962455 [Internet] (cited 11.05.15) [PubMed] [Google Scholar]
- 88.Avcin T., Ambrozic A., Kuhar M. Anticardiolipin and anti-beta(2) glycoprotein I antibodies in sera of 61 apparently healthy children at regular preventive visits. Rheumatology (Oxford) 2001;40(5):565–573. doi: 10.1093/rheumatology/40.5.565. http://www.ncbi.nlm.nih.gov/pubmed/11371668 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 89.Karali Z., Basaranoglu S.T., Karali Y. Autoimmunity and hepatitis A vaccine in children. J. Investig. Allergol. Clin. Immunol. 2015;21(5):389–393. http://www.ncbi.nlm.nih.gov/pubmed/21905502 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 90.Blank M., Krause I., Fridkin M. Bacterial induction of autoantibodies to beta2-glycoprotein-I accounts for the infectious etiology of antiphospholipid syndrome. J. Clin. Invest. 2002;109(6):797–804. doi: 10.1172/JCI12337. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=150905&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Koppang E.O., Bjerkås I., Haugarvoll E. Vaccination-induced systemic autoimmunity in farmed Atlantic salmon. J. Immunol. 2008;181(7):4807–4814. doi: 10.4049/jimmunol.181.7.4807. http://www.ncbi.nlm.nih.gov/pubmed/18802084 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 92.Toplak N., Kveder T., Trampus-Bakija A. Autoimmune response following annual influenza vaccination in 92 apparently healthy adults. Autoimmun. Rev. 2008;8(2):134–138. doi: 10.1016/j.autrev.2008.07.008. http://www.ncbi.nlm.nih.gov/pubmed/18700173 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 93.Korošec P., Petrovec M., Lainščscak M. High non-specific T lymphocyte response to the adjuvanted H1N1 vaccine in comparison with the H1N1/H3N2/B-Brisbane vaccine without adjuvant. Scand. J. Immunol. 2012;76(5):497–504. doi: 10.1111/j.1365-3083.2012.02763.x. http://www.ncbi.nlm.nih.gov/pubmed/22862739 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 94.Pasoto S.G., Ribeiro A.C., Viana V.S.T. Short and long-term effects of pandemic unadjuvanted influenza A(H1N1) pdm09 vaccine on clinical manifestations and autoantibody profile in primary Sjögren’s syndrome. Vaccine. 2013;31(14):1793–1798. doi: 10.1016/j.vaccine.2013.01.057. http://www.ncbi.nlm.nih.gov/pubmed/23395584 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 95.Elkayam O., Paran D., Burke M. Pneumococcal vaccination of patients with systemic lupus erythematosus: effects on generation of autoantibodies. Autoimmunity. 2005;38(7):493–496. doi: 10.1080/08916930500285725. http://www.ncbi.nlm.nih.gov/pubmed/16373254 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 96.Mok C.C., Ho L.Y., Fong L.S. Immunogenicity and safety of a quadrivalent human papillomavirus vaccine in patients with systemic lupus erythematosus: a case-control study. Ann. Rheum. Dis. 2013;72(5):659–664. doi: 10.1136/annrheumdis-2012-201393. http://www.ncbi.nlm.nih.gov/pubmed/22589375 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 97.Soybilgic A., Onel K.B., Utset T. Safety and immunogenicity of the quadrivalent HPV vaccine in female systemic lupus erythematosus patients aged 12 to 26 years. Pediatr. Rheumatol. Online J. 2013;11:29. doi: 10.1186/1546-0096-11-29. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3751269&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Martinuc P., orobic J., Avcin T., Bozic B. Anti-phospholipid antibodies following vaccination with recombinant hepatitis B vaccine. Clin. Exp. Immunol. 2005;142(2):377–380. doi: 10.1111/j.1365-2249.2005.02923.x. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1809502&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Arbuckle M.R., McClain M.T., Rubertone M.V. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N. Engl. J. Med. 2003;349(16):1526–1533. doi: 10.1056/NEJMoa021933. http://www.ncbi.nlm.nih.gov/pubmed/14561795 [Internet] (cited 22.06.15) [DOI] [PubMed] [Google Scholar]
- 100.Perricone C., Agmon-Levin N., Ceccarelli F. Genetics and autoantibodies. Immunol. Res. 2013;56(2–3):206–219. doi: 10.1007/s12026-013-8396-9. http://www.ncbi.nlm.nih.gov/pubmed/23564181 [Internet] (cited 30.06.15) [DOI] [PubMed] [Google Scholar]
- 101.Hajdu S.D., Agmon-Levin N., Shoenfeld Y. Silicone and autoimmunity. Eur. J. Clin. Invest. 2011;41(2):203–211. doi: 10.1111/j.1365-2362.2010.02389.x. http://www.ncbi.nlm.nih.gov/pubmed/20880074 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 102.Santucci B., Valenzano C., de Rocco M. Platinum in the environment: frequency of reactions to platinum-group elements in patients with dermatitis and urticaria. Contact Dermatitis. 2000;43(6):333–338. doi: 10.1034/j.1600-0536.2000.043006333.x. http://www.ncbi.nlm.nih.gov/pubmed/11140383 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 103.Lidar M., Agmon-Levin N., Langevitz P. Silicone and scleroderma revisited. Lupus. 2012;21(2):121–127. doi: 10.1177/0961203311430703. http://www.ncbi.nlm.nih.gov/pubmed/22235041 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 104.Vermeulen R.C.W., Scholte H.R. Rupture of silicone gel breast implants and symptoms of pain and fatigue. J. Rheumatol. 2003;30(10):2263–2267. http://www.ncbi.nlm.nih.gov/pubmed/14528527 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 105.Bekerecioglu M., Onat A.M., Tercan M. The association between silicone implants and both antibodies and autoimmune diseases. Clin. Rheumatol. 2008;27(2):147–150. doi: 10.1007/s10067-007-0659-1. http://www.ncbi.nlm.nih.gov/pubmed/17610103 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 106.Zandman-Goddard G., Blank M., Ehrenfeld M. A comparison of autoantibody production in asymptomatic and symptomatic women with silicone breast implants. J. Rheumatol. 1999;26(1):73–77. http://www.ncbi.nlm.nih.gov/pubmed/9918243 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 107.Cohen Tervaert J.W., Kappel R.M. Silicone implant incompatibility syndrome (SIIS): a frequent cause of ASIA (Shoenfeld’s syndrome) Immunol. Res. 2013;56(2–3):293–298. doi: 10.1007/s12026-013-8401-3. http://www.ncbi.nlm.nih.gov/pubmed/23576058 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 108.De M., attia D., Del V., ecchio G.C., Russo G. Management of chronic childhood immune thrombocytopenic purpura: AIEOP consensus guidelines. Acta Haematol. 2010;123(2):96–109. doi: 10.1159/000268855. http://www.ncbi.nlm.nih.gov/pubmed/20029174 [Internet] (cited 28.04.15) [DOI] [PubMed] [Google Scholar]
- 109.Tingle A.J., Allen M., Petty R.E. Rubella-associated arthritis. I. Comparative study of joint manifestations associated with natural rubella infection and RA 27/3 rubella immunisation. Ann. Rheum. Dis. 1986;45(2):110–114. doi: 10.1136/ard.45.2.110. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1001829&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ray P., Black S., Shinefield H. Risk of chronic arthropathy among women after rubella vaccination. Vaccine safety datalink team. JAMA. 1997;278(7):551–556. http://www.ncbi.nlm.nih.gov/pubmed/9268275 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 111.Gotuzzo E., Yactayo S., Córdova E. Efficacy and duration of immunity after yellow fever vaccination: systematic review on the need for a booster every 10 years. Am. J. Trop. Med. Hyg. 2013;89(3):434–444. doi: 10.4269/ajtmh.13-0264. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3771278&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Weekly epidemiological record (2013).
- 113.Rafferty E., Duclos P., Yactayo S. Risk of yellow fever vaccine-associated viscerotropic disease among the elderly: a systematic review. Vaccine. 2013;31(49):5798–5805. doi: 10.1016/j.vaccine.2013.09.030. http://www.ncbi.nlm.nih.gov/pubmed/24079979 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 114.Gandhi N.M., Morales A., Lamm D.L. Bacillus calmette–guérin immunotherapy for genitourinary cancer. BJU Int. 2013;112(3):288–297. doi: 10.1111/j.1464-410X.2012.11754.x. http://www.ncbi.nlm.nih.gov/pubmed/23517232 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 115.Kodali V.R., Clague R.B. Arthritis after BCG vaccine in a healthy woman. J. Int. Med. 1998;244(2):183–184. doi: 10.1046/j.1365-2796.1998.0371a.x. http://www.ncbi.nlm.nih.gov/pubmed/10095807 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 116.Kåss E., Straume S., Munthe E. Dermatomyositis after B.C.G. vaccination. Lancet. 1978;1(8067):772. doi: 10.1016/s0140-6736(78)90885-1. http://www.ncbi.nlm.nih.gov/pubmed/76775 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 117.Kothari S.S. Aetiopathogenesis of Takayas’s arteritis and BCG vaccination: the missing link? Med. Hypotheses. 1995;45(3):227–230. doi: 10.1016/0306-9877(95)90109-4. http://www.ncbi.nlm.nih.gov/pubmed/8569543 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 118.Bernini L., Manzini C.U., Giuggioli D. Reactive arthritis induced by intravesical BCG therapy for bladder cancer: our clinical experience and systematic review of the literature. Autoimmun. Rev. 2013;12(12):1150–1159. doi: 10.1016/j.autrev.2013.06.017. http://www.ncbi.nlm.nih.gov/pubmed/2381656 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 119.Schattner A., Gilad A., Cohen J. Systemic granulomatosis and hypercalcaemia following intravesical bacillus calmette–guérin immunotherapy. J. Int. Med. 2002;251(3):272–277. doi: 10.1046/j.1365-2796.2002.00957.x. http://www.ncbi.nlm.nih.gov/pubmed/11886488 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 120.Chang M.H., Chen C.J., Lai M.S. Universal hepatitis B vaccination in Taiwan and the incidence of hepatocellular carcinoma in children. Taiwan childhood hepatoma study group. N. Engl. J. Med. 1997;336(26):1855–1859. doi: 10.1056/NEJM199706263362602. http://www.ncbi.nlm.nih.gov/pubmed/9197213 [Internet] (cited 06.03.15) [DOI] [PubMed] [Google Scholar]
- 121.Agmon-Levin N., Paz Z., Israeli E. Vaccines and autoimmunity. Nat. Rev. Rheumatol. 2009;5(11):648–652. doi: 10.1038/nrrheum.2009.196. http://www.ncbi.nlm.nih.gov/pubmed/19865091 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 122.Perricone C., Shoenfeld Y. Hepatitis B vaccination and undifferentiated connective tissue disease: another brick in the wall of the autoimmune/inflammatory syndrome induced by adjuvants (Asia) J. Clin. Rheumatol. 2013;19(5):231–233. doi: 10.1097/RHU.0b013e31829d56f1. http://www.ncbi.nlm.nih.gov/pubmed/23884184 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 123.Tomljenovic L., Shaw C.A. In: Adverse Reactions to Human Papillomavirus (HPV) Vaccines. 1st ed. Shoenfeld Y., Agmon-Levin N., Tomljenovic L., editors. Wiley Press; New York: 2015. [Google Scholar]
- 124.De C., arvalho N., Teixeira J., Roteli-Martins C.M. Sustained efficacy and immunogenicity of the HPV-16/18 AS04-adjuvanted vaccine up to 7.3 years in young adult women. Vaccine. 2010;28(38):6247–6255. doi: 10.1016/j.vaccine.2010.07.007. http://www.ncbi.nlm.nih.gov/pubmed/20643092 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 125.Lee S. Detection of human papillomavirus L1 gene DNA fragments in postmortem blood and spleen after GardasilReg. vaccination—a case report. Adv. Biosci. Biotechnol. 2012;3(8):1214–1224. http://www.scirp.org/journal/PaperInformation.aspx?PaperID=25840 [Internet] [Google Scholar]
- 126.Tomljenovic L., Shaw C.A. Human papillomavirus (HPV) vaccine policy and evidence-based medicine: are they at odds? Ann. Med. 2013;45(2):182–193. doi: 10.3109/07853890.2011.645353. http://www.ncbi.nlm.nih.gov/pubmed/22188159 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 127.Mahajan D., Roomiani I., Gold M.S. Annual report: surveillance of adverse events following immunisation in Australia. Commun. Dis. Intell. Q. Rep. 2009;34(3):259–276. doi: 10.33321/cdi.2010.34.26. http://www.ncbi.nlm.nih.gov/pubmed/21090181 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 128.Lawrence G., Gold M.S., Hill R. Annual report: surveillance of adverse events following immunisation in Australia. Commun. Dis. Intell. Q. Rep. 2007;32(4):371–387. doi: 10.33321/cdi.2008.32.37. http://www.ncbi.nlm.nih.gov/pubmed/19374268 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 129.Agmon-Levin N., Kivity S., Shoenfeld Y. Influenza vaccine and autoimmunity. Isr. Med. Assoc. J. 2015;11(3):183–185. http://www.ncbi.nlm.nih.gov/pubmed/19544711 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 130.Echevarría-Zuno S., Mejía-Aranguré J.M., Mar-Obeso A.J. Infection and death from influenza A H1N1 virus in Mexico: a retrospective analysis. Lancet. 2009;374(9707):2072–2079. doi: 10.1016/S0140-6736(09)61638-X. http://www.ncbi.nlm.nih.gov/pubmed/19913290 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 131.Bischoff A.L., Følsgaard N.V., Carson C.G. Altered response to A(H1N1) pnd09 vaccination in pregnant women: a single blinded randomized controlled trial. PLoS One. 2013;8(4):e56700. doi: 10.1371/journal.pone.0056700. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3630160&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Cerpa-Cruz S., Paredes-Casillas P., Landeros N., avarro E. Adverse events following immunization with vaccines containing adjuvants. Immunol. Res. 2013;56(2–3):299–303. doi: 10.1007/s12026-013-8400-4. http://www.ncbi.nlm.nih.gov/pubmed/23576057 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 133.Duggal T., Segal P., Shah M. Antineutrophil cytoplasmic antibody vasculitis associated with influenza vaccination. Am. J. Nephrol. 2013;38(2):174–178. doi: 10.1159/000354084. http://www.ncbi.nlm.nih.gov/pubmed/23941822 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 134.Blank M., Israeli E., Shoenfeld When Y.A.P.S. (Hughes syndrome) met the autoimmune/inflammatory syndrome induced by adjuvants (ASIA) Lupus. 2012;21(7):711–714. doi: 10.1177/0961203312438115. http://www.ncbi.nlm.nih.gov/pubmed/22635209 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 135.Findlow J. Meningococcal group B vaccines. Hum. Vaccin Immunother. 2013;9(6):1387–1388. doi: 10.4161/hv.24689. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3901838&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Gottfredsson M., Reynisson I.K., Ingvarsson R.F. Comparative long-term adverse effects elicited by invasive group B and C meningococcal infections. Clin. Infect. Dis. 2011;53(9):e117–e124. doi: 10.1093/cid/cir500. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3189164&tool=pmcentrez&rendertype=Abstract [Internet] (cited 11.03.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Borja-Tabora C., Montalban C., Memish Z.A. Immune response, antibody persistence, and safety of a single dose of the quadrivalent meningococcal serogroups A, C, W-135, and Y tetanus toxoid conjugate vaccine in adolescents and adults: results of an open, randomised, controlled study. BMC Infect. Dis. 2013;13:116. doi: 10.1186/1471-2334-13-116. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3599520&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Haber P., Sejvar J., Mikaeloff Y. Vaccines and Guillain-Barré syndrome. Drug Saf. 2009;32(4):309–323. doi: 10.2165/00002018-200932040-00005. http://www.ncbi.nlm.nih.gov/pubmed/19388722 [Internet] (cited 14.03.15) [DOI] [PubMed] [Google Scholar]
- 139.Goodman M.J., Nordin J.D., Belongia E.A. Henoch-Schölein purpura and polysaccharide meningococcal vaccine. Pediatrics. 2010;126(2):e325–e329. doi: 10.1542/peds.2009-3195. http://www.ncbi.nlm.nih.gov/pubmed/20624811 [Internet] (cited 14.03.15) [DOI] [PubMed] [Google Scholar]
- 140.Valdivielso-Ramos M., Velázquez D., Tortoledo A. Infantile bullous pemphigoid developing after hexavalent, meningococcal and pneumococcal vaccinations. An. Pediatr. (Barcelona) 2011;75(3):199–202. doi: 10.1016/j.anpedi.2011.04.009. http://www.ncbi.nlm.nih.gov/pubmed/21676664 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 141.Jit M. The risk of sequelae due to pneumococcal meningitis in high-income countries: a systematic review and meta-analysis. J. Infect. 2010;61(2):114–124. doi: 10.1016/j.jinf.2010.04.008. http://www.ncbi.nlm.nih.gov/pubmed/20433866 [Internet] (cited 25.03.15) [DOI] [PubMed] [Google Scholar]
- 142.O'Brien K.L., Wolfson L.J., Watt J.P. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet. 2009;374(9693):893–902. doi: 10.1016/S0140-6736(09)61204-6. http://www.ncbi.nlm.nih.gov/pubmed/19748398 [Internet] (cited 22.10.15) [DOI] [PubMed] [Google Scholar]
- 143.FDA. Pneumococcal 13-valent Conjugate Vaccine (Diphtheria CRM197 Protein) Prevnar 13®, http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm201667.htm [Internet] (cited 28.04.15).
- 144.Whitney C.G., Farley M.M., Hadler J. Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. N. Engl. J. Med. 2003;348(18):1737–1746. doi: 10.1056/NEJMoa022823. http://www.ncbi.nlm.nih.gov/pubmed/12724479 [Internet] (cited 07.04.15) [DOI] [PubMed] [Google Scholar]
- 145.Cook I.F., Pond D., Hartel G. Comparative reactogenicity and immunogenicity of 23 valent pneumococcal vaccine administered by intramuscular or subcutaneous injection in elderly adults. Vaccine. 2007;25(25):4767–4774. doi: 10.1016/j.vaccine.2007.04.017. http://www.ncbi.nlm.nih.gov/pubmed/17512098 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 146.Bryant K.A., Block S.L., Baker S.A. Safety and immunogenicity of a 13-valent pneumococcal conjugate vaccine. Pediatrics. 2010;125(5):866–875. doi: 10.1542/peds.2009-1405. http://www.ncbi.nlm.nih.gov/pubmed/20435707 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 147.Mori S., Ueki Y., Akeda Y. Pneumococcal polysaccharide vaccination in rheumatoid arthritis patients receiving tocilizumab therapy. Ann. Rheum. Dis. 2013;72(8):1362–1366. doi: 10.1136/annrheumdis-2012-202658. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3711492&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Van A., ssen S., Agmon-Levin N., Elkayam O. EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum. Dis. 2011;70(3):414–422. doi: 10.1136/ard.2010.137216. http://www.ncbi.nlm.nih.gov/pubmed/21131643 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 149.Cook T.M., Protheroe R.T., Handel J.M. Tetanus a review of the literature. Br. J. Anaesth. 2015;87(3):477–487. doi: 10.1093/bja/87.3.477. http://www.ncbi.nlm.nih.gov/pubmed/11517134 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 150.Topaloglu H., Berker M., Kansu T. Optic neuritis and myelitis after booster tetanus toxoid vaccination. Lancet. 1992;339(8786):178–179. doi: 10.1016/0140-6736(92)90241-t. http://www.ncbi.nlm.nih.gov/pubmed/1346027 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 151.Cervera R., Piette J.-C., Font J. Antiphospholipid syndrome: clinical and immunologic manifestations and patterns of disease expression in a cohort of 1000 patients. Arthritis Rheum. 2002;46(4):1019–1027. doi: 10.1002/art.10187. http://www.ncbi.nlm.nih.gov/pubmed/11953980 [Internet] (cited 25.01.15) [DOI] [PubMed] [Google Scholar]
- 152.Schwarzenbacher R., Zeth K., Diederichs K. Crystal structure of human beta2-glycoprotein I: implications for phospholipid binding and the antiphospholipid syndrome. EMBO J. 1999;18(22):6228–6239. doi: 10.1093/emboj/18.22.6228. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1171686&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Blank M., Cohen J., Toder V. Induction of anti-phospholipid syndrome in naive mice with mouse lupus monoclonal and human polyclonal anti-cardiolipin antibodies. Proc. Natl. Acad. Sci. U. S. A. 1991;88(8):3069–3073. doi: 10.1073/pnas.88.8.3069. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=51386&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Huh J.Y., Yi D.Y., Hwang S.G. Characterization of antiphospholipid antibodies in chronic hepatitis B infection. Kor. J. Hematol. 2011;46(1):36–40. doi: 10.5045/kjh.2011.46.1.36. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3065625&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Tarján P., Sipka S., Lakos G. Influenza vaccination and the production of anti-phospholipid antibodies in patients with systemic lupus erythematosus. Scand. J. Rheumatol. 2015;35(3):241–243. doi: 10.1080/03009740500474552. http://www.ncbi.nlm.nih.gov/pubmed/16766374 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 156.Colafrancesco S., Agmon-Levin N., Perricone C. Unraveling the soul of autoimmune diseases: pathogenesis, diagnosis and treatment adding dowels to the puzzle. Immunol. Res. 2013;56(2–3):200–205. doi: 10.1007/s12026-013-8429-4. http://www.ncbi.nlm.nih.gov/pubmed/23733136 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 157.Orbach H., Agmon-Levin N., Zandman-Goddard G. Vaccines and autoimmune diseases of the adult. Discov. Med. 2010;9(45):90–97. http://www.ncbi.nlm.nih.gov/pubmed/20193633 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 158.Chatham W.W., Wallace D.J., Stohl W. Effect of belimumab on vaccine antigen antibodies to influenza, pneumococcal, and tetanus vaccines in patients with systemic lupus erythematosus in the BLISS-76 trial. J. Rheumatol. 2012;39(8):1632–1640. doi: 10.3899/jrheum.111587. http://www.ncbi.nlm.nih.gov/pubmed/22707609 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 159.Agmon-Levin N., Zafrir Y., Paz Z. Ten cases of systemic lupus erythematosus related to hepatitis B vaccine. Lupus. 2009;18(13):1192–1197. doi: 10.1177/0961203309345732. http://www.ncbi.nlm.nih.gov/pubmed/1988056 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 160.Lyrio L.D.C., Grassi M.F.R., Santana I.U. Prevalence of cervical human papillomavirus infection in women with systemic lupus erythematosus. Rheumatol. Int. 2013;33(2):335–340. doi: 10.1007/s00296-012-2426-0. http://www.ncbi.nlm.nih.gov/pubmed/22451033 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 161.Jennette J.C., Falk R.J., Bacon P.A. 2012 revised international chapel hill consensus conference nomenclature of vasculitides. Arthritis Rheum. 2012;65(1):1–11. doi: 10.1002/art.37715. http://www.ncbi.nlm.nih.gov/pubmed/23045170 [Internet] (cited 06.02.15) [DOI] [PubMed] [Google Scholar]
- 162.Zaas A., Scheel P., Venbrux A. Large artery vasculitis following recombinant hepatitis B vaccination: 2 cases. J. Rheumatol. 2015;28(5):1116–1120. http://www.ncbi.nlm.nih.gov/pubmed/11361200 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 163.Guillevin L., Guittard T., Boura R. Etiology and precipitating factors of necrotizing angiitis with respiratory manifestations. 5 case reports. Ann. Med. Int. (Paris) 1983;134(7):625–628. http://www.ncbi.nlm.nih.gov/pubmed/6141756 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 164.Ventura F., Antunes H., Brito C. Cutaneous polyarteritis nodosa in a child following hepatitis B vaccination. Eur. J. Dermatol. 2015;19(4):400–401. doi: 10.1684/ejd.2009.0695. http://www.ncbi.nlm.nih.gov/pubmed/19467971 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 165.De Carvalho J.F., Pereira R.M.R., Shoenfeld Y. Systemic polyarteritis nodosa following hepatitis B vaccination. Eur. J. Int. Med. 2008;19(8):575–578. doi: 10.1016/j.ejim.2007.06.035. http://www.ncbi.nlm.nih.gov/pubmed/19046721 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 166.Miron D., Fink D., Hashkes P.J. Kawasaki disease in an infant following immunisation with hepatitis B vaccine. Clin. Rheumatol. 2003;22(6):461–463. doi: 10.1007/s10067-003-0785-3. http://www.ncbi.nlm.nih.gov/pubmed/14677029 [Internet] (cited 21.04.15) [DOI] [PubMed] [Google Scholar]
- 167.Schmöeller D., Keiserman M.W., Staub H.L. Yellow fever vaccination and Kawasaki disease. Pediatr. Infect. Dis. J. 2009;28(11):1037–1038. doi: 10.1097/INF.0b013e3181bbc571. http://www.ncbi.nlm.nih.gov/pubmed/19859023 [Internet] (cited 28.04.15) [DOI] [PubMed] [Google Scholar]
- 168.Beretta L., Caronni M., Vanoli M. Churg–strauss vasculitis with brain involvement following hepatitis B vaccination. Clin. Exp. Rheumatol. 2015;19(6):757. http://www.ncbi.nlm.nih.gov/pubmed/11791655 [Internet] (cited 28.04.15) [PubMed] [Google Scholar]
- 169.Birck R., Kaelsch I., Schnuelle P. ANCA-associated vasculitis following influenza vaccination: causal association or mere coincidence? J. Clin. Rheumatol. 2009;15(6):289–291. doi: 10.1097/RHU.0b013e3181b55fe4. http://www.ncbi.nlm.nih.gov/pubmed/19734734 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 170.Spaetgens B., van Paassen P., Tervaert J.W.C. Influenza vaccination in ANCA-associated vasculitis. Nephrol. Dial Transplant. 2009;24(10):3258. doi: 10.1093/ndt/gfp398. http://www.ncbi.nlm.nih.gov/pubmed/19666908 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 171.Jariwala S., Vernon N., Shliozberg J. Henoch-Schönlein purpura after hepatitis A vaccination. Ann. Allergy Asthma Immunol. 2015;107(2):180–181. doi: 10.1016/j.anai.2011.05.006. http://www.ncbi.nlm.nih.gov/pubmed/21802028 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 172.Pimentel M.I.F., Vasconcellos de E.C.F.E., Cerbino-Neto J. Henoch-Schönlein purpura following influenza A H1N1 vaccination. Rev. Soc. Bras. Med. Trop. 2015;44(4):531. doi: 10.1590/s0037-86822011000400029. http://www.ncbi.nlm.nih.gov/pubmed/21860908 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 173.Watanabe T. Henoch-Schönlein purpura following influenza vaccinations during the pandemic of influenza A (H1N1) Pediatr. Nephrol. 2015;26(5):795–798. doi: 10.1007/s00467-010-1722-8. http://www.ncbi.nlm.nih.gov/pubmed/21120537 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 174.Famularo G., Nicotra G.C., Minisola G. Leukocytoclastic vasculitis after influenza vaccination. J. Clin. Rheumatol. 2006;12(1):48–50. doi: 10.1097/01.rhu.0000200341.38947.43. http://www.ncbi.nlm.nih.gov/pubmed/16484885 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 175.Bani-Sadr F., Gueit I., Humbert G. Vasculitis related to hepatitis A vaccination. Clin. Infect. Dis. 1996;22(3):596. doi: 10.1093/clinids/22.3.596. http://www.ncbi.nlm.nih.gov/pubmed/8853003 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 176.Masse I., Descoffres M.C. Hypersensitivity vasculitis after hepatitis B vaccination. Presse Med. 2015;27(20):965–966. http://www.ncbi.nlm.nih.gov/pubmed/9767839 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 177.Fox B.C., Peterson A. Leukocytoclastic vasculitis after pneumococcal vaccination. Am. J. Infect. Control. 2015;26(3):365–366. doi: 10.1016/s0196-6553(98)80021-7. http://www.ncbi.nlm.nih.gov/pubmed/9638297 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 178.Gerecitano J., Friedman-Kien A., Chazen G.D. Allergic reaction to varicella vaccine. Ann. Int. Med. 1997;126(10):833–834. doi: 10.7326/0003-4819-126-10-199705150-00036. http://www.ncbi.nlm.nih.gov/pubmed/9148672 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 179.Somer T., Finegold S.M. Vasculitides associated with infections, immunization, and antimicrobial drugs. Clin. Infect. Dis. 1995;20(4):1010–1036. doi: 10.1093/clinids/20.4.1010. http://www.ncbi.nlm.nih.gov/pubmed/7795045 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 180.Muñiz A.E. Lymphocytic vasculitis associated with the anthrax vaccine: case report and review of anthrax vaccination. J. Emerg. Med. 2003;25(3):271–276. doi: 10.1016/s0736-4679(03)00201-4. http://www.ncbi.nlm.nih.gov/pubmed/14585454 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 181.Sedaghat M., Zarei-Ghanavati S., Shokoohi S. Panuveitis and dermal vasculitis following MMR vaccination. East Mediterr. Health J. 2015;13(2):470–474. http://www.ncbi.nlm.nih.gov/pubmed/17684869 [Internet] (cited 26.04.15) [PubMed] [Google Scholar]
- 182.Symmons D.P., Chakravarty K. Can immunisation trigger rheumatoid arthritis? Ann. Rheum. Dis. 1993;52(12):843–844. doi: 10.1136/ard.52.12.843. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1005209&tool=pmcentrez&rendertype=Abstract [Internet] (cited 26.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Ray P., Black S., Shinefield H. Risk of rheumatoid arthritis following vaccination with tetanus, influenza and hepatitis B vaccines among persons 15-59 years of age. Vaccine. 2011;29(38):6592–6597. doi: 10.1016/j.vaccine.2011.06.112. http://www.ncbi.nlm.nih.gov/pubmed/21763385 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 184.Delavallée L., Duvallet E., Semerano L. Anti-cytokine vaccination in autoimmune diseases. Swiss Med. Wkly. 2010 doi: 10.4414/smw.2010.13108. http://www.ncbi.nlm.nih.gov/pubmed/21043003 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 185.Prakken B., Albani S. Exploiting T. cell crosstalk as a vaccination strategy for rheumatoid arthritis. Arthritis Rheum. 2007;56(2):389–392. doi: 10.1002/art.22317. http://www.ncbi.nlm.nih.gov/pubmed/17265472 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 186.Chen G., Li N., Zang Y.C.Q. Vaccination with selected synovial T cells in rheumatoid arthritis. Arthritis Rheum. 2007;56(2):453–463. doi: 10.1002/art.22316. http://www.ncbi.nlm.nih.gov/pubmed/17265481 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 187.Mosca M., Tani C., Carli L. Undifferentiated CTD a wide spectrum of autoimmune diseases. Best Pract. Res. Clin. Rheumatol. 2012;26(1):73–77. doi: 10.1016/j.berh.2012.01.005. http://www.ncbi.nlm.nih.gov/pubmed/22424194 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 188.Mosca M., Tani C., Carli L. Analysis of the evolution of UCTD to defined CTD after a long term follow-up. Clin. Exp. Rheumatol. 2015;31(3):471. http://www.ncbi.nlm.nih.gov/pubmed/23075608 [Internet] (cited 27.04.15) [PubMed] [Google Scholar]
- 189.Rasheed A., Lipstein-Kresch E., Kalra J. Undifferentiated connective tissue disease after silicone-gel testicular implantation. J. Clin. Rheumatol. 1995;1(5):310. doi: 10.1097/00124743-199510000-00014. http://www.ncbi.nlm.nih.gov/pubmed/19078007 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 190.Bruzzese V., Zullo A., Hassan C. Connective tissue disease following hepatitis B vaccination. J. Clin. Rheumatol. 2013;19(5):280–281. doi: 10.1097/RHU.0b013e31829d558a. http://www.ncbi.nlm.nih.gov/pubmed/23872550 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 191.Bolognia Jean, L., JLJ RRP, Dermatology, 3rd ed. (2012).
- 192.Safavi K. Prevalence of alopecia areata in the first national health and nutrition examination survey. Arch. Dermatol. 1992;128(5):702. doi: 10.1001/archderm.1992.01680150136027. http://www.ncbi.nlm.nih.gov/pubmed/1575541 [Internet] (cited 30.06.15) [DOI] [PubMed] [Google Scholar]
- 193.Bogdanos D.P., Smyk D.S., Invernizzi P. Tracing environmental markers of autoimmunity: introducing the infectome. Immunol. Res. 2013;56(2–3):220–240. doi: 10.1007/s12026-013-8399-6. http://www.ncbi.nlm.nih.gov/pubmed/23592050 [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Rodriguez T.A., Duvic M. Onset of alopecia areata after Epstein-Barr virus infectious mononucleosis. J. Am. Acad. Dermatol. 2008;59(1):137–139. doi: 10.1016/j.jaad.2008.02.005. http://www.ncbi.nlm.nih.gov/pubmed/18329131 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 195.Hayderi El L., Nikkels-Tassoudji N., Nikkels A.F. Hair loss after varicella zoster virus infection. Case Rep. Dermatol. 2015;5(1):43–47. doi: 10.1159/000348648. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3604874&tool=pmcentrez&rendertype=Abstract [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Wise R.P., Kiminyo K.P., Salive M.E. Hair loss after routine immunizations. JAMA. 1997;278(14):1176–1178. http://www.ncbi.nlm.nih.gov/pubmed/9326478 [Internet] (cited 27.04.15) [PubMed] [Google Scholar]
- 197.Sundberg J.P., Silva K.A., Zhang W. Recombinant human hepatitis B vaccine initiating alopecia areata: testing the hypothesis using the C3H/HeJ mouse model. Vet. Dermatol. 2009;20(2):99–104. doi: 10.1111/j.1365-3164.2008.00692.x. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2956183&tool=pmcentrez&rendertype=Abstract [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Sánchez-Ramón S., Gil J., Cianchetta-Sívori M. Alopecia universalis in an adult after routine tetanus toxoid vaccine. Med. Clin. (Barcelona) 2011;136(7):318. doi: 10.1016/j.medcli.2010.03.010. http://www.ncbi.nlm.nih.gov/pubmed/20451934 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 199.Tuccori M., Pisani C., Bachini L. Telogen effluvium following bivalent human papillomavirus vaccine administration: a report of two cases. Dermatology. 2012;224(3):212–214. doi: 10.1159/000337412. http://www.ncbi.nlm.nih.gov/pubmed/22584489 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 200.Razeghinejad M.R., Nowroozzadeh M.H., Sharifi M. Acute-onset madarosis following MMR vaccination. J. Pediatr. Ophthalmol. Strabismus. 2015;46(2):123–124. doi: 10.3928/01913913-20090301-16. http://www.ncbi.nlm.nih.gov/pubmed/19343978 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 201.Stasi R., Amadori S., Osborn J. Long-term outcome of otherwise healthy individuals with incidentally discovered borderline thrombocytopenia. PLoS Med. 2006;3(3):e24. doi: 10.1371/journal.pmed.0030024. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1326262&tool=pmcentrez&rendertype=Abstract [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Perricone C., Ceccarelli F., Nesher G. Immune thrombocytopenic purpura (ITP) associated with vaccinations: a review of reported cases. Immunol. Res. 2014;60(2–3):226–235. doi: 10.1007/s12026-014-8597-x. http://www.ncbi.nlm.nih.gov/pubmed/25427992 [Internet] (cited 29.04.15) [DOI] [PubMed] [Google Scholar]
- 203.Yong M., Schoonen W.M., Li L. Epidemiology of paediatric immune thrombocytopenia in the General Practice Research Database. Br. J. Haematol. 2010;149(6):855–864. doi: 10.1111/j.1365-2141.2010.08176.x. http://www.ncbi.nlm.nih.gov/pubmed/20377590 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 204.Russo G., Miraglia V., Branciforte F. Effect of eradication of Helicobacter pylori in children with chronic immune thrombocytopenia: a prospective, controlled, multicenter study. Pediatr. Blood Cancer. 2011;56(2):273–278. doi: 10.1002/pbc.22770. http://www.ncbi.nlm.nih.gov/pubmed/20830773 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 205.Rinaldi M., Perricone C., Ortega-Hernandez O.-D. Immune thrombocytopaenic purpura: an autoimmune cross-link between infections and vaccines. Lupus. 2014;23(6):554–567. doi: 10.1177/0961203313499959. http://www.ncbi.nlm.nih.gov/pubmed/24763539 [Internet] (cited 30.05.15) [DOI] [PubMed] [Google Scholar]
- 206.Tishler M., Shoenfeld Y. Vaccination may be associated with autoimmune diseases. Isr. Med. Assoc. J. 2015;6(7):430–432. http://www.ncbi.nlm.nih.gov/pubmed/15274537 [Internet] (cited 27.04.15) [PubMed] [Google Scholar]
- 207.Coppieters K.T., Wiberg A., von H., errath M.G. Viral infections and molecular mimicry in type 1 diabetes. APMIS. 2012;120(12):941–949. doi: 10.1111/apm.12011. http://www.ncbi.nlm.nih.gov/pubmed/2305117 [Internet] (cited 14.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Childhood immunizations and type 1 diabetes: summary of an Institute for Vaccine Safety Workshop. The Institute for Vaccine Safety Diabetes Workshop Panel, Pediatr. Infect. Dis. J. 18(3) (1999) 217–222. http://www.ncbi.nlm.nih.gov/pubmed/10093941 [Internet] (27.04.15). [DOI] [PubMed]
- 209.Singh B. Stimulation of the developing immune system can prevent autoimmunity. J. Autoimmun. 2000;14(1):15–22. doi: 10.1006/jaut.1999.0349. http://www.ncbi.nlm.nih.gov/pubmed/10648113 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 210.Wahlberg J., Fredriksson J., Vaarala O. Vaccinations may induce diabetes-related autoantibodies in one-year-old children. Ann. N. Y. Acad. Sci. 2003;1005:404–408. doi: 10.1196/annals.1288.068. http://www.ncbi.nlm.nih.gov/pubmed/14679101 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 211.Black S.B., Lewis E., Shinefield H.R. Lack of association between receipt of conjugate haemophilus influenzae type B vaccine (HbOC) in infancy and risk of type 1 (juvenile onset) diabetes: long term follow-up of the HbOC efficacy trial cohort. Pediatr. Infect. Dis. J. 2015;21(6):568–569. doi: 10.1097/00006454-200206000-00018. http://www.ncbi.nlm.nih.gov/pubmed/12182385 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 212.Duderstadt S.K., Rose C.E., Real T.M. Vaccination and risk of type 1 diabetes mellitus in active component U. S. Military, 2002–2008. Vaccine. 2012;30(4):813–819. doi: 10.1016/j.vaccine.2011.10.087. http://www.ncbi.nlm.nih.gov/pubmed/22075092 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 213.Difficulties in Assessing the Relationship, If Any, Between Mumps Vaccination and Diabetes Mellitus in Childhood, PubMed,NCBI, http://www.ncbi.nlm.nih.gov/pubmed/?term=Milne+LM.+2001.+Difficulties+in+assessing+the+relationship%2C+if+any%2C+between+mumps+vaccination+and+diabetes+mellitus+in+childhood.+Vaccine+19%3A1018%E2%80%9325 [Internet] (cited 27.04.15). [DOI] [PubMed]
- 214.Bardage C., Persson I., Ortqvist A. Neurological and autoimmune disorders after vaccination against pandemic influenza A (H1N1) with a monovalent adjuvanted vaccine: population based cohort study in Stockholm, Sweden. BMJ. 2015;343:d5956. doi: 10.1136/bmj.d5956. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3192001&tool=pmcentrez&rendertype=Abstract [Internet] (cited 30.03.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Parent M.E., Siemiatycki J., Menzies R. Bacille calmette–guérin vaccination and incidence of IDDM in Montreal, Canada. Diabetes Care. 1997;20(5):767–772. doi: 10.2337/diacare.20.5.767. http://www.ncbi.nlm.nih.gov/pubmed/9135940 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 216.Huppmann M., Baumgarten A., Ziegler A.-G. Neonatal bacille calmette–guerin vaccination and type 1 diabetes. Diabetes Care. 2005;28(5):1204–1206. doi: 10.2337/diacare.28.5.1204. http://www.ncbi.nlm.nih.gov/pubmed/15855590 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 217.Da Rosa L.C., Chiuso-Minicucci F., Zorzella-Pezavento S.F.G. Bacille calmette–guérin/DNAhsp65 prime-boost is protective against diabetes in non-obese diabetic mice but not in the streptozotocin model of type 1 diabetes. Clin. Exp. Immunol. 2013;173(3):430–437. doi: 10.1111/cei.12140. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3949630&tool=pmcentrez&rendertype=Abstract [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Fontana A., Gast H., Reith W. Narcolepsy: autoimmunity, effector T cell activation due to infection, or T cell independent, major histocompatibility complex class II induced neuronal loss? Brain. 2010;133:1300–1311. doi: 10.1093/brain/awq086. http://www.ncbi.nlm.nih.gov/pubmed/20403960 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 219.Katzav A., Arango M.T., Kivity S. Passive transfer of narcolepsy: anti-TRIB2 autoantibody positive patient IgG causes hypothalamic orexin neuron loss and sleep attacks in mice. J. Autoimmun. 2013;45:24–30. doi: 10.1016/j.jaut.2013.06.010. http://www.ncbi.nlm.nih.gov/pubmed/23834844 [Internet] (cited 07.04.15) [DOI] [PubMed] [Google Scholar]
- 220.Arango M.-T., Kivity S., Shoenfeld Y. Is narcolepsy a classical autoimmune disease? Pharmacol. Res. 2015;92:6–12. doi: 10.1016/j.phrs.2014.10.005. http://www.ncbi.nlm.nih.gov/pubmed/25447795 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 221.Käll A. The Pandemrix—narcolepsy tragedy: how it started and what we know today. Acta Paediatr. 2013;102(1):2–4. doi: 10.1111/apa.12012. http://www.ncbi.nlm.nih.gov/pubmed/23016673 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 222.Rozenberg O., Lerner A., Pacht A. A novel algorithm for the diagnosis of celiac disease and a comprehensive review of celiac disease diagnostics. Clin. Rev. Allergy Immunol. 2015;42(3):331–341. doi: 10.1007/s12016-010-8250-y. http://www.ncbi.nlm.nih.gov/pubmed/21279475 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 223.Plot L., Amital H., Barzilai O. Infections may have a protective role in the etiopathogenesis of celiac disease. Ann. N. Y. Acad. Sci. 2009;1173:670–674. doi: 10.1111/j.1749-6632.2009.04814.x. http://www.ncbi.nlm.nih.gov/pubmed/1975821 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 224.Stene L.C., Honeyman M.C., Hoffenberg E.J. Rotavirus infection frequency and risk of celiac disease autoimmunity in early childhood: a longitudinal study. Am. J. Gastroenterol. 2006;101(10):2333–2340. doi: 10.1111/j.1572-0241.2006.00741.x. http://www.ncbi.nlm.nih.gov/pubmed/17032199 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 225.Dolcino M., Zanoni G., Bason C. A subset of anti-rotavirus antibodies directed against the viral protein VP7 predicts the onset of celiac disease and induces typical features of the disease in the intestinal epithelial cell line T84. Immunol. Res. 2013;56(2–3):465–476. doi: 10.1007/s12026-013-8420-0. http://www.ncbi.nlm.nih.gov/pubmed/23572432 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 226.Pozo-Rubio T., de Palma G., Mujico J.R. Influence of early environmental factors on lymphocyte subsets and gut microbiota in infants at risk of celiac disease; the PROFICEL study. Nutr. Hosp. 2015;28(2):464–473. doi: 10.3305/nh.2013.28.2.6310. http://www.ncbi.nlm.nih.gov/pubmed/23822699 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 227.Mackie S.L., Mallen C.D. Polymyalgia rheumatica. BMJ. 2013;347:f6937. doi: 10.1136/bmj.f6937. http://www.ncbi.nlm.nih.gov/pubmed/24301266 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 228.Rojo-Contreras W., Olivas-Flores E.M., Gamez-Nava J.I. Cervical human papillomavirus infection in Mexican women with systemic lupus erythematosus or rheumatoid arthritis. Lupus. 2012;21(4):365–372. doi: 10.1177/0961203311425517. http://www.ncbi.nlm.nih.gov/pubmed/22020266 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]
- 229.Soriano A., Verrecchia E., Marinaro A. Giant cell arteritis and polymyalgia rheumatica after influenza vaccination: report of 10 cases and review of the literature. Lupus. 2012;21(2):153–157. doi: 10.1177/0961203311430222. http://www.ncbi.nlm.nih.gov/pubmed/22235046 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 230.Saadoun D., Cacoub P., Mahoux D. Postvaccine vasculitis: a report of three cases. Rev. Med. Int. 2015;22(2):172–176. doi: 10.1016/s0248-8663(00)00307-6. http://www.ncbi.nlm.nih.gov/pubmed/11234675 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 231.Torisu H., Kira R., Ishizaki Y. Clinical study of childhood acute disseminated encephalomyelitis, multiple sclerosis, and acute transverse myelitis in Fukuoka Prefecture, Japan. Brain Dev. 2010;32(6):454–462. doi: 10.1016/j.braindev.2009.10.006. http://www.ncbi.nlm.nih.gov/pubmed/19942388 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 232.Bennetto L., Scolding N. Inflammatory/post-infectious encephalomyelitis. J. Neurol. Neurosurg. Psychiatry. 2015;75(Suppl. 1):22–28. doi: 10.1136/jnnp.2003.034256. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1765651&tool=pmcentrez&rendertype=Abstract [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Markovic-Plese S., Hemmer B., Zhao Y. High level of cross-reactivity in influenza virus hemagglutinin-specific CD4+ T-cell response: implications for the initiation of autoimmune response in multiple sclerosis. J. Neuroimmunol. 2005;169(1–2):31–38. doi: 10.1016/j.jneuroim.2005.07.014. http://www.ncbi.nlm.nih.gov/pubmed/16150497 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 234.Tejada-Simon M., V, Zang Y.C.Q., Hong J. Cross-reactivity with myelin basic protein and human herpesvirus-6 in multiple sclerosis. Ann. Neurol. 2003;53(2):189–197. doi: 10.1002/ana.10425. http://www.ncbi.nlm.nih.gov/pubmed/12557285 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 235.O’Connor K.C., Chitnis T., Griffin D.E. Myelin basic protein-reactive autoantibodies in the serum and cerebrospinal fluid of multiple sclerosis patients are characterized by low-affinity interactions. J. Neuroimmunol. 2003;136(1–2):140–148. doi: 10.1016/s0165-5728(03)00002-x. http://www.ncbi.nlm.nih.gov/pubmed/12620653 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 236.Rosenmann H., Grigoriadis N., Karussis D. Tauopathy-like abnormalities and neurologic deficits in mice immunized with neuronal tau protein. Arch. Neurol. 2006;63(10):1459–1467. doi: 10.1001/archneur.63.10.1459. http://www.ncbi.nlm.nih.gov/pubmed/17030663 [Internet] (cited 05.04.15) [DOI] [PubMed] [Google Scholar]
- 237.Karussis D., Vourka-Karussis U., Mizrachi-Koll R. Acute/relapsing experimental autoimmune encephalomyelitis: induction of long lasting, antigen-specific tolerance by syngeneic bone marrow transplantation. Mult Scler. 1999;5(1):17–21. doi: 10.1177/135245859900500104. http://www.ncbi.nlm.nih.gov/pubmed/10096098 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 238.Vera-Lastra O., Medina G., Cruz-Dominguez M.D.P. Autoimmune/inflammatory syndrome induced by adjuvants (Shoenfeld’s syndrome): clinical and immunological spectrum. Expert Rev. Clin. Immunol. 2015;9(4):361–373. doi: 10.1586/eci.13.2. http://www.ncbi.nlm.nih.gov/pubmed/23557271 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 239.Huynh W., Cordato D.J., Kehdi E. Post-vaccination encephalomyelitis: literature review and illustrative case. J. Clin. Neurosci. 2008;15(12):1315–1322. doi: 10.1016/j.jocn.2008.05.002. http://www.ncbi.nlm.nih.gov/pubmed/18976924 [Internet] (cited 19.03.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.DeStefano F., Verstraeten T., Jackson L.A. Vaccinations and risk of central nervous system demyelinating diseases in adults. Arch. Neurol. 2003;60(4):504–509. doi: 10.1001/archneur.60.4.504. http://www.ncbi.nlm.nih.gov/pubmed/12707063 [Internet] (cited 21.04.15) [DOI] [PubMed] [Google Scholar]
- 241.Mikaeloff Y., Caridade G., Husson B. Acute disseminated encephalomyelitis cohort study: prognostic factors for relapse. Eur. J. Paediatr. Neurol. 2007;11(2):90–95. doi: 10.1016/j.ejpn.2006.11.007. http://www.ncbi.nlm.nih.gov/pubmed/17188007 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 242.Menge T., Cree B., Saleh A. Neuromyelitis optica following human papillomavirus vaccination. Neurology. 2012;79(3):285–287. doi: 10.1212/WNL.0b013e31825fdead. http://www.ncbi.nlm.nih.gov/pubmed/22722628 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 243.Altman A., Szyper-Kravitz M., Shoenfeld Y. HBV vaccine and dermatomyositis: is there an association? Rheumatol. Int. 2008;28(6):609–612. doi: 10.1007/s00296-007-0485-4. http://www.ncbi.nlm.nih.gov/pubmed/18034245 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 244.Sticherling M., Erfurt-Berge C. Autoimmune blistering diseases of the skin. Autoimmun. Rev. 2012;11(3):226–230. doi: 10.1016/j.autrev.2011.05.017. http://www.ncbi.nlm.nih.gov/pubmed/21640850 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 245.Ruocco V., Ruocco E., Lo S., chiavo A. Pemphigus: etiology, pathogenesis, and inducing or triggering factors: facts and controversies. Clin. Dermatol. 2015;31(4):374–381. doi: 10.1016/j.clindermatol.2013.01.004. http://www.ncbi.nlm.nih.gov/pubmed/23806154 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 246.Downs A.M., Lear J.T., Bower C.P. Does influenza vaccination induce bullous pemphigoid? A report of four cases. Br. J. Dermato. 1998;138(2):363. doi: 10.1046/j.1365-2133.1998.02097.x. http://www.ncbi.nlm.nih.gov/pubmed/9602897 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 247.Chacón G.R., Sinha A.A. Bullous pemphigoid after herpes zoster vaccine administration: association or coincidence? J. Drugs Dermatol. 2015;10(11):1328–1330. http://www.ncbi.nlm.nih.gov/pubmed/22052318 [Internet] (cited 27.04.15) [PubMed] [Google Scholar]
- 248.Bodokh I., Lacour J.P., Bourdet J.F. Reactivation of bullous pemphigoid after influenza vaccination. Therapie. 2015;49(2):154. http://www.ncbi.nlm.nih.gov/pubmed/7817351 [Internet] (cited 27.04.15) [PubMed] [Google Scholar]
- 249.Dalakas M.C. Pathophysiology of autoimmune polyneuropathies. Presse Med. 2013;42(6 pt 2):e181–e192. doi: 10.1016/j.lpm.2013.01.058. http://www.ncbi.nlm.nih.gov/pubmed/23642299 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 250.Stübgen J.-P. A review on the association between inflammatory myopathies and vaccination. Autoimmun. Rev. 2014;13(1):31–39. doi: 10.1016/j.autrev.2013.08.005. http://www.ncbi.nlm.nih.gov/pubmed/24001753 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 251.Yunus M.B. Fibromyalgia and overlapping disorders: the unifying concept of central sensitivity syndromes. Semin Arthritis Rheum. 2007;36(6):339–356. doi: 10.1016/j.semarthrit.2006.12.009. http://www.ncbi.nlm.nih.gov/pubmed/17350675 [Internet] (cited 02.03.15) [DOI] [PubMed] [Google Scholar]
- 252.Buskila D., Atzeni F., Sarzi-Puttini P. Etiology of fibromyalgia: the possible role of infection and vaccination. Autoimmun. Rev. 2008;8(1):41–43. doi: 10.1016/j.autrev.2008.07.023. http://www.ncbi.nlm.nih.gov/pubmed/18706528 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 253.Appel S., Chapman J., Shoenfeld Y. Infection and vaccination in chronic fatigue syndrome: myth or reality? Autoimmunity. 2007;40(1):48–53. doi: 10.1080/08916930701197273. http://www.ncbi.nlm.nih.gov/pubmed/17364497 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 254.Yao K., Crawford J.R., Komaroff A.L. Review part 2: human herpesvirus-6 in central nervous system diseases. J. Med. Virol. 2010;82(10):1669–1678. doi: 10.1002/jmv.21861. http://www.ncbi.nlm.nih.gov/pubmed/20827763 [Internet] (cited 27.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Kerr J.R. Pathogenesis of parvovirus B19 infection: host gene variability, and possible means and effects of virus persistence. J. Vet. Med. B Infect. Dis. Vet. Public Health. 2015;52(7–8):335–339. doi: 10.1111/j.1439-0450.2005.00859.x. http://www.ncbi.nlm.nih.gov/pubmed/16316396 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 256.Andersson M., Bagby J., Dyrehag L. Effects of staphylococcus toxoid vaccine on pain and fatigue in patients with fibromyalgia/chronic fatigue syndrome. Eur. J. Pain. 1998;2(2):133–142. doi: 10.1016/s1090-3801(98)90006-4. http://www.ncbi.nlm.nih.gov/pubmed/10700309 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 257.Magnus P., Brubakk O., Nyland H. Vaccination as teenagers against meningococcal disease and the risk of the chronic fatigue syndrome. Vaccine. 2009;27(1):23–27. doi: 10.1016/j.vaccine.2008.10.043. http://www.ncbi.nlm.nih.gov/pubmed/189840 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 258.Israeli E., Pardo A. The sick building syndrome as a part of the autoimmune (auto-inflammatory) syndrome induced by adjuvants. Mod. Rheumatol. 2011;21(3):235–239. doi: 10.1007/s10165-010-0380-9. http://www.ncbi.nlm.nih.gov/pubmed/21188456 [Internet] (cited 25.03.15) [DOI] [PubMed] [Google Scholar]
- 259.Cadusseau J., Ragunathan-Thangarajah N., Surenaud M. Selective elevation of circulating CCL2/MCP1 levels in patients with longstanding post-vaccinal macrophagic myofasciitis and ASIA. Curr. Med. Chem. 2014;21(4):511–517. doi: 10.2174/09298673113206660287. http://www.ncbi.nlm.nih.gov/pubmed/24083602 [Internet] (cited 17.04.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Lange G., Tiersky L.A., Scharer J.B. Cognitive functioning in Gulf War Illness. J. Clin. Exp. Neuropsychol. 2015;23(2):240–249. doi: 10.1076/jcen.23.2.240.1208. http://www.ncbi.nlm.nih.gov/pubmed/11309677 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 261.Unwin C., Blatchley N., Coker W. Health of UK servicemen who served in Persian Gulf War. Lancet. 1999;353(9148):169–178. doi: 10.1016/S0140-6736(98)11338-7. http://www.ncbi.nlm.nih.gov/pubmed/9923871 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 262.Rook G.A., Zumla A. Gulf War syndrome: is it due to a systemic shift in cytokine balance towards a Th2 profile? Lancet. 1997;349(9068):1831–1833. doi: 10.1016/S0140-6736(97)01164-1. http://www.ncbi.nlm.nih.gov/pubmed/9269228 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 263.Phillips C.J., Matyas G.R., Hansen C.J. Antibodies to squalene in US Navy Persian Gulf War veterans with chronic multisymptom illness. Vaccine. 2009;27(29):3921–3926. doi: 10.1016/j.vaccine.2009.03.091. http://www.ncbi.nlm.nih.gov/pubmed/1937978 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 264.Van Assen S., Elkayam O., Agmon-Levin N. Vaccination in adult patients with auto-immune inflammatory rheumatic diseases: a systematic literature review for the European League Against Rheumatism evidence-based recommendations for vaccination in adult patients with auto-immune inflammatory rheuma. Autoimmun. Rev. 2011;10(6):341–352. doi: 10.1016/j.autrev.2010.12.003. http://www.ncbi.nlm.nih.gov/pubmed/21182987 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 265.Heijstek M.W., Ott, de B., ruin L.M., Bijl M. EULAR recommendations for vaccination in paediatric patients with rheumatic diseases. Ann. Rheum. Dis. 2011;70(10):1704–1712. doi: 10.1136/ard.2011.150193. http://www.ncbi.nlm.nih.gov/pubmed/21813547 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 266.Mota da L.M.H., Oliveira A.C.V., Lima R.A.C. Vaccination against yellow fever among patients on immunosuppressors with diagnoses of rheumatic diseases. Rev. Soc. Bras. Med. Trop. 2015;42(1):23–27. doi: 10.1590/s0037-86822009000100006. http://www.ncbi.nlm.nih.gov/pubmed/19287931 [Internet] (cited 22.04.15) [DOI] [PubMed] [Google Scholar]
- 267.Buch M.H., Smolen J.S., Betteridge N. Updated consensus statement on the use of rituximab in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2011;70(6):909–920. doi: 10.1136/ard.2010.144998. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3086093&tool=pmcentrez&rendertype=Abstract [Internet] (cited 01.03.15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Zonneveld-Huijssoon E., Ronaghy A., Van R., ossum M.A.J. Safety and efficacy of meningococcal c vaccination in juvenile idiopathic arthritis. Arthritis Rheum. 2007;56(2):639–646. doi: 10.1002/art.22399. http://www.ncbi.nlm.nih.gov/pubmed/17265499 [Internet] (cited 27.04.15) [DOI] [PubMed] [Google Scholar]
- 269.Bergfors E., Trollfors B., Inerot A. Unexpectedly high incidence of persistent itching nodules and delayed hypersensitivity to aluminium in children after the use of adsorbed vaccines from a single manufacturer. Vaccine. 2003;22(1):64–69. doi: 10.1016/s0264-410x(03)00531-0. http://www.ncbi.nlm.nih.gov/pubmed/14604572 [Internet] (cited 26.04.15) [DOI] [PubMed] [Google Scholar]