Abstract
A robust and reliable cell culture system for serum-derived HCV (HCVser) has not been established yet because of the presence of neutralizing antibody and tropism for infection. To overcome this obstacle, we employed a lipid-mediated protein intracellular delivery reagent (PIDR) that permits internalization of proteins into cells. Although entry of HCVcc was not enhanced by the treatment with PIDR, entry of HCVser into hepatoma cell lines (Huh7 and HepG2) and immortalized primary hepatocytes (Hc and HuS/E2) was significantly enhanced by the PIDR treatment. The entry of HCVser into Huh7 cells in the presence of PIDR was resistant to the neutralization by an anti-hCD81 antibody, suggesting that PIDR is capable of internalizing HCVser in a receptor-independent manner. Interestingly, the PIDR-mediated entry of HCVser and HCVcc was enhanced by the addition of sera from chronic hepatitis C patients but not from healthy donors. In addition, neutralization of HCVcc infection by anti-E2 antibody was canceled by the treatment with PIDR. In conclusion, the PIDR is a valuable tool to get over the obstacle of neutralizing antibodies to internalize HCV into cells and might be useful for the establishment of in vitro propagation HCVser.
Keywords: Hepatitis C virus, Protein intracellular delivery, Serum-derived virus
Abbreviations: HCV, hepatitis C virus; IFN, interferon; PHH, primary human hepatocyte; PIDR, protein intracellular delivery reagent; PCR, polymerase chain reaction; VSV, vesicular stomatitis virus
1. Introduction
More than 170 million individuals worldwide are infected with hepatitis C virus (HCV), and hepatic steatosis, cirrhosis and hepatocellular carcinoma (HCC) induced by HCV infection are life-threatening [1]. Although combined-therapy with peg-interferon (IFN) and ribavirin has achieved a sustained virological response in 50% of individuals infected with HCV genotype 1 [2], a more effective therapeutic modality for HCV infection is needed [3]. To this end, further detailed analyses of HCV are needed in order to clarify not only the viral life cycle but also the pathogenesis. Although cell culture systems for HCV (HCVcc) have been established based on the JFH-1 strain isolated from a fulminant hepatitis C patient [4], such systems were unable to establish chronic infection in chimpanzees [4] or to induce cell damage and inflammation in chimeric mice xenotransplanted with human hepatocytes [5], and therefore establishment of a robust cell culture system capable of propagating serum-derived HCV (HCVser) from hepatitis C patients is required.
Although previous reports suggested a partial replication of HCVser in the primary hepatocytes (PHH) freshly isolated from human liver [6], the level of viral RNA replication was low and reconfirmation of the viral propagation was not achieved due to the difficulty of providing a stable supply of the PHH. Recently, it was shown that a three-dimensional culture system of immortalized PHH was capable of propagating the HCVser from chronic hepatitis C patients [7], [8]. HCVser in the patients was slightly amplified in these culture systems, but the levels of viral RNA replication were far lower than those of HCVcc in Huh7-derived adaptive cell lines. Part of the difficulty in establishing a cell culture system for HCVser might be attributable to: i) the existence of high titers of neutralizing antibodies in the sera of hepatitis C patients [9]; ii) the heterogeneity of HCV particles (quasispecies), which exhibit different cell tropisms for infection and replication [10]; and iii) the inconsistent expression of the putative receptors for HCV entry, including CD81, SR-BI, claudin-1 and occludin [11]. It may be necessary to overcome these obstacles before a robust and reliable in vitro cell culture system can be established for HCVser.
Polybrene has been used for the efficient infection of retrovirus [12], and spinoculation has also been employed to accelerate the entry of various viruses, including retrovirus [13] and murine coronavirus [14]. Entry of HCVcc into not only the permissive cell line Huh7.5.1 but also the non-permissive cell line PLC/PRF/5 has been shown to be enhanced by spinoculation [15], [16]. In this study, we examined the effects of these accelerating procedures for entry of HCVser and found that a cationic amphiphilic-based lipid-mediated protein intracellular delivery reagent (PIDR) [17] exhibited a potent enhancement of entry of HCVser. Our data suggest that PIDR allows complex formation with viral particles via both electrostatic and hydrophobic interactions and enhances internalization of the HCVser into cells in a receptor-independent manner.
2. Materials and methods
2.1. Sera
Sera from chronic hepatitis C patients and a cured patient possessing the anti-HCV antibodies were obtained at the Kyushu University Hospital after obtaining full informed consent from all patients. Seven serum samples from hepatitis C patients, including two window-period serum samples without any detectable anti-HCV antibodies, were obtained from the Benesis Corporation (Osaka, Japan). Human sera from healthy donors were obtained from Sigma–Aldrich Inc. (St. Louis, MO). Sera from healthy donors, chronic hepatitis patients and acute hepatitis patients were designated HDS, CHS, and AHS, respectively. The HCV-RNA titers of CHS and AHS were 7.15 ± 0.34 (range: 6.6–7.5) and 8.20 ± 0.14 (range: 8.1–8.3), respectively. The genotypes of HCV in these sera were 1a (7 patients) and 1b (11 patients).
2.2. Human liver cell lines and preparation of HCVcc
HepG2 and HEK-293T cell lines were obtained from the American Type Culture Collection (Rockville, MD). The Huh7OK1 cell line exhibits an efficient propagation of HCVcc as described previously [18]. The HepCD81 cell line stably expressing human CD81 was established as described previously [19]. HuS-E/2 was kindly provided by M. Hijikata, Kyoto University [20]. Hc (an immortalized human liver cell line) was purchased from the Applied Cell Biology Research Institute (Kirkland, WA). These cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM) (Sigma) containing 10% fetal bovine serum (FBS). The in vitro transcribed RNA of the JFH-1 strain of HCV was introduced into Huh7OK1 cells [21] and culture supernatants were collected at 7 days post-transfection and used as HCVcc. The infectivity of HCVcc was determined by focus forming assay as previously described [19].
2.3. Transfection of plasmids and intracellular delivery of proteins
The plasmids were transfected into cells by liposome-mediated transfection using TransIT-LT1 (Mirus, Madison, WI). The proteins were introduced into cells by PIDR (PULSin; Polyplus-transfection Inc., New York, NY) according to the manufacturer’s protocol. FITC-conjugated mouse IgG antibody (Invitrogen Molecular Probes, Eugene, OR) or recombinant phycoerythrin (PE; Polyplus-transfection Inc.) was introduced into cells by the PIDR as a positive control.
2.4. Quantitative reverse-transcription polymerase chain reaction (qRT-PCR)
Total RNA was prepared from cells using an RNeasy mini kit (Qiagen, Tokyo, Japan). The synthesis of first-strand cDNA and qRT-PCR was performed using TaqMan EZ RT-PCR Core Reagents and ABI Prism 7000 system (Applied Biosystems Japan, Tokyo, Japan) according to the manufacturer’s protocol. The primers for Taqman PCR were designed in a non-coding region as previously reported [22].
2.5. Infection of HCVser and HCVcc by spinoculation, polybrene and PIDR
Cells were seeded at 1 × 105 cells/well in a 48-well plate and cultured for 24 h. For spinoculation, 2 μl of HCV-positive serum or HCVcc at a multiplicity of infection (MOI) of 0.05 were inoculated into cells and immediately centrifuged at 500× g for 120 min at room temperature. For infection of HCV by polybrene and PIDR, 2 μg of polybrene or 1.5 μl of PIDR were incubated with HCV-positive serum or HCVcc diluted in 20 μl of phosphate-buffered saline (PBS) for 15 min at room temperature to allow complex formation [12]. Cells were trypsinized at 24 h post-inoculation, seeded in a 48-well plate to remove non-specific binding of HCV, and cultured for several days.
2.6. Production and infection of pseudotype vesicular stomatitis virus (VSV)
Pseudotype VSVs were generated as described previously [19]. The pseudotype VSVs, VSVpv/GFP and VSVpv/luc, bore the VSVG protein on the virion surface and replaced the G envelope gene with the green fluorescent protein (GFP) and luciferase genes, respectively. Pseudotype VSV bearing HCV E1 and E2 glycoproteins (HCVpv) was prepared as described previously [19]. These pseudotype viruses were inoculated into Huh7OK1 cells in the presence or absence of PIDR together with or without anti-VSVG polyclonal antibody (ab34774; Abcam Inc., Cambridge, MA) or CHS, and infectivity was determined at 24 h post-infection by the expression of GFP or luciferase activity after treatment with a passive lysis buffer (Promega Co., Madison, WI).
2.7. Inhibition of HCVcc and HCVser infection by the treatment with antibody against human CD81 and anti-E2 antibody (AP-33)
To determine the involvement of human CD81 in the intracellular delivery of HCV by PIDR, Huh7OK1 cells were pre-treated with 5 μg/ml of anti-human CD81 monoclonal antibody (JS-81; BD Biosciences Pharmingen, Mountain View, CA) for 1 h at 37 °C and then inoculated with HCVcc or HCVser in the presence of PIDR. Anti-E2 monoclonal antibody (AP-33) was kindly provided by A.H. Patel, University of Glasgow [23]. AP-33 was pre-mixed with HCVcc for 1 h with or without PIDR and then cells were incubated with this mixture and cultured for several days.
3. Results
3.1. Effect of spinoculation and polybrene on the entry of HCVser and HCVcc
First, we examined the effect of spinoculation on the entry of HCVser or HCVcc. Intracellular HCV-RNA titers of Huh7OK1 cells upon infection of HCVser and HCVcc with or without spinoculation at 24 h post-infection were determined (Data not shown). Although entry of HCVcc into Huh7OK1 cells was 10-fold increased by the spinoculation, no effect was observed in the entry of HCVser. Next, we examined the effect of polybrene on the entry of HCVser and HCVcc into Huh7OK1 cells. Although polybrene induced a slight increase of the entry of HCVcc, no significant effect on the entry of HCVser was observed intracellularly at 24 h post-infection (Data not shown). These results indicated that neither spinoculation nor polybrene induced an enhancement of the entry of HCVser.
3.2. Internalization of viral particles by PIDR
To determine the efficacy of intracellular delivery of proteins by PIDR, FITC-conjugated mouse IgG and recombinant PE were introduced into Huh7OK1 cells by PIDR. Both FITC-conjugated IgG and PE were efficiently internalized into Huh7OK1 cells by the treatment with PIDR but not by the lipofection (Fig. 1 A). Next, to determine the receptor-independent entry of viral particles into cells by the PIDR, the expression of GFP upon infection of a pseudotype VSV lacking VSVG (VSV∆Gpv/GFP) into Huh7OK1 cells was examined. Although VSV∆Gpv/GFP lost infectivity due to a lack of the G glycoprotein, the addition of PIDR facilitated entry of the particles (Fig. 1A). To further examine the effect of the presence of neutralization antibody on the delivery of viral particles by PIDR, the expression of GFP upon transduction of VSVpv/GFP into Huh7OK1 cells in the presence of neutralization antibody and PIDR was examined. Although VSVpv/GFP exhibited a high infectivity to Huh7OK1 cells and the infection was completely neutralized by the anti-VSVG antibody, treatment with PIDR partially recovered the infectivity of VSVpv/GFP neutralized by the antibody (Fig. 1B). Similar results were confirmed by using VSV∆Gpv/luc and VSVpv/luc carrying the luciferase gene as a reporter (Fig. 1C). These results indicate that PIDR is a useful tool to facilitate the entry of viral particles into target cells, irrespective of the authenticity of the envelope proteins of the particles or the presence of the neutralizing antibodies.
Fig. 1.
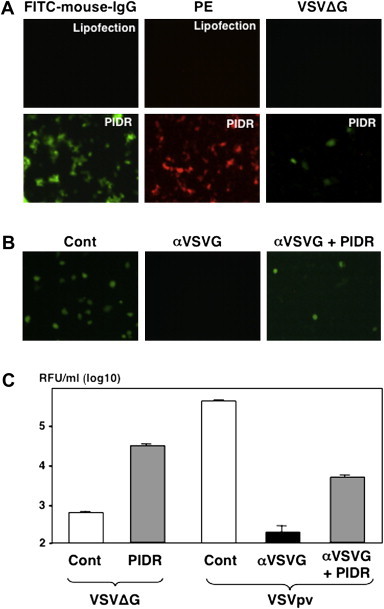
Characterization of the intracellular delivery of proteins and viral particles by PIDR. (A) FITC-conjugated mouse IgG (left panels) or recombinant PE (center panels) was introduced into Huh7OK1 cells by the treatment with PIDR or a lipofection reagent. The expression of GFP upon infection of a pseudotype VSV lacking VSVG (VSV∆Gpv/GFP) into Huh7OK1 cells in the presence (lower panel) and absence (upper panel) of PIDR was examined (right panels). (B) The effect of the presence of neutralization antibody on the delivery of viral particles by PIDR. Expression of GFP upon transduction of a VSVpv/GFP into Huh7OK1 cells in the presence of neutralization antibody and PIDR was examined. (C) The receptor-independent entry of viral particles was confirmed by using VSV∆Gpv/luc and VSVpv/luc carrying the luciferase gene as a reporter.
3.3. Effect of PIDR on the infection with HCVcc
To determine the effect of PIDR on the infection of HCV, HCVcc was inoculated into Huh7OK1 cells at an MOI of 0.05 in the presence or absence of PIDR and intracellular viral RNA was measured every 24 h. No significant difference in the infection of HCVcc was observed by the addition of PIDR (Fig. 2 A). Next, to mimic the infection of HCV in the presence of neutralization antibodies, HCVcc was mixed with CHS and inoculated into Huh7OK1 cells. Although infection of HCVcc into Huh7OK1 was completely neutralized by the incubation with CHS, addition of PIDR recovered the infectivity of HCVcc (Fig. 2B). Furthermore, to confirm the effect of PIDR on the internalization of HCVcc interacting with neutralizing antibody, HCVcc was pre-incubated with AP-33 and inoculated into Huh7OK1 cells. Although infection of HCVcc was neutralized by the treatment with AP-33 in a dose-dependent manner, the neutralization by AP-33 was canceled by the treatment with PIDR (Fig. 2C). These results suggest that PIDR has the ability to internalize HCVcc even in the presence of neutralizing antibodies.
Fig. 2.
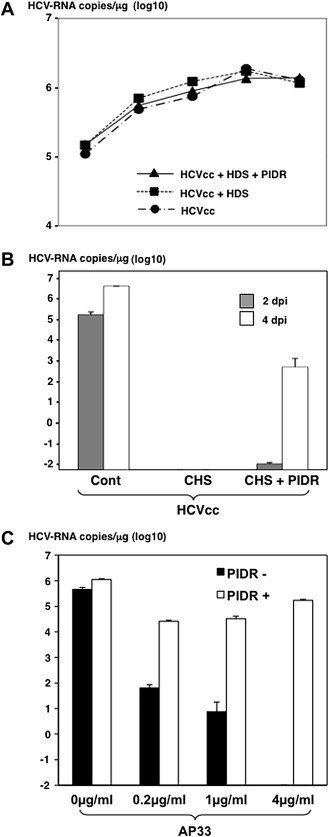
Effect of PIDR on the infection with HCVcc. (A) HCVcc was inoculated into Huh7OK1 cells at an MOI of 0.05 in the presence or absence of healthy donor sera (HDS) and PIDR and intracellular viral RNA was measured every 24 h (B) HCVcc was mixed with sera from chronic hepatitis C patients (CHS) or/and PIDR and inoculated into Huh7OK1 cells. (C) HCVcc was pre-mixed with anti-E2 monoclonal antibody (AP-33) and inoculated into Huh7OK1 cells in the presence or absence of PIDR.
3.4. Effect of PIDR on the infection of HCVser
The efficient neutralizing activities of HCV infection in CHS were comfirmed by a neutralization assay using pseudotype viruses. Infection of HCVpv bearing HCV E1 and E2 proteins but not of VSVpv bearing VSVG protein was significantly neutralized by the CHS (Fig. 3 A). Next, to determine the effect of PIDR on the entry of HCVser in the presence of neutralizing antibodies, HCVser and CHS possessing the neutralizing antibodies against HCV were inoculated into Huh7OK1 cells with or without incubation with PIDR. Huh7OK1 cells inoculated with CHS pre-incubated with PIDR exhibited significantly higher HCV-RNA titers at 24 h post-infection than those without the treatment (Fig. 3B). Furthermore, to determine the amount of HCV internalized into cells, Huh7OK1 cells inoculated with CHS treated with PIDR were trypsinized and reseeded into a new culture plate at 24 h post-infection. HCV-RNA was detected in cells inoculated CHS pre-incubated with PIDR but not in those without PIDR treatment at 24 h after reseeding (Fig. 3C). These results indicate that treatment with PIDR permits HCVser to internalize into target cells even in the presence of neutralizing antibodies.
Fig. 3.
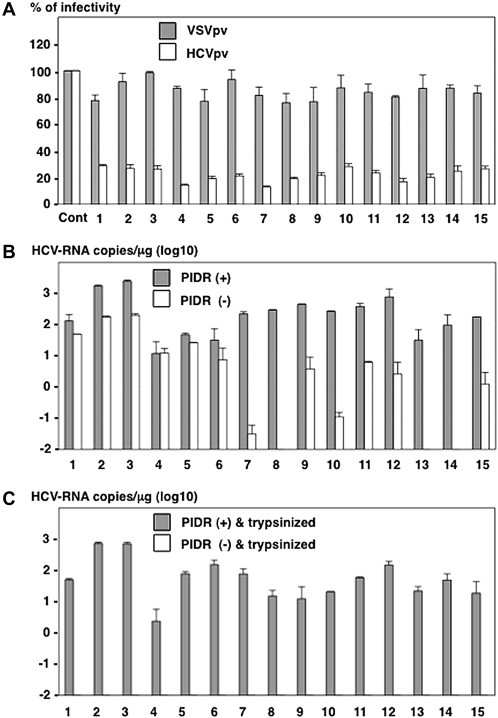
Effect of PIDR on the infection with HCVser. (A) The neutralizing effect of antibodies in the CHS was determined by a neutralization assay using the pseudotype viruses. White and gray bars indicated VSVpv and HCVpv, respectively. (B) The effect of PIDR on the entry of HCVser in the CHS into Huh7OK1 cells. HCV-RNA titers in cells 24 h post-inoculation with HCVser in the presence and absence of PIDR are indicated by gray and white bars, respectively. (C) To determine the internalization of HCVser into Huh7OK1 cells, cells inoculated with the CHS in the presence (gray bar) or absence (white bars) of PIDR were trypsinized and reseeded into a new culture plate at 24 h post-infection, and HCV-RNA titers in the cells were determined at 24 h post-inoculation.
3.5. Neutralizing antibodies in sera from chronic hepatitis C patients enhance PIDR-mediated entry of HCV
No reduction of infectivity of HCVpv and VSVpv was observed by the incubation with AHS, suggesting that AHS possesses no detectable neutralizing antibodies to HCV (Fig. 4 A). To examine the effect of neutralizing antibody on the intracellular delivery of HCVser by PIDR, AHS was incubated with the CHS carrying neutralization antibodies but no infectious HCV obtained from patients cured by the interferon therapy in the presence or absence of PIDR and inoculated into Huh7OK1 cells. Internalization of HCV in AHS was increased two-fold by the treatment with PIDR. However, intracellular viral RNA titer was slightly decreased by the incubation with CHS in the absence of PIDR, probably due to the neutralization by the antibodies, and addition of PIDR resulted in a three-fold enhancement of the entry of HCV in AHS in the presence of CHS compared with that in the absence of CHS (Fig. 4B). Next, Huh7OK1 cells were inoculated with HCVcc at an MOI of 0.05 after incubation with 0.4–40 μl/ml of HCV-negative CHS in the presence or absence of PIDR. Although infection of HCVcc was neutralized by CHS in a dose-dependent manner, addition of PIDR enhanced the infection of HCVcc in the presence of CHS in a dose-dependent manner (Fig. 4C). These results indicate that PIDR facilitates entry of HCVser in the presence of neutralizing antibody.
Fig. 4.
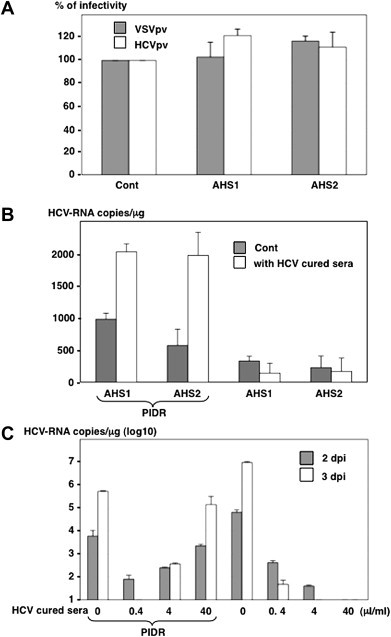
Neutralizing antibodies in the CHS enhanced the PIDR-mediated entry of HCV. (A) The absence of neutralizing antibodies in the sera from acute hepatitis C patients (AHS) was determined by a neutralization assay using the HCVpv (white bars) and VSVpv (gray bar). (B) The effect of PIDR on the entry of HCVser into Huh7OK1 cells in the presence of neutralizing antibodies. The AHS were incubated with the CHS carrying neutralization antibodies but no infectious HCV obtained from patients cured by the IFN therapy (white bars) or HDS (gray bar) in the presence (left) or absence (right) of PIDR and inoculated into Huh7OK1 cells. The HCV-RNA titers in cells were determined at 24 h post-inoculation. (C). The effect of neutralizing antibodies on the PIDR-mediated infection of HCVcc. Huh7OK1 cells were inoculated with HCVcc at an MOI of 0.05 after incubation with 0.4–40 μl/ml of HCV-negative CHS in the presence (left) or absence (right) of PIDR. Gray and white bars indicate the HCV-RNA titers at 2 and 3 days after infection, respectively.
3.6. Human CD81-independent entry of HCVser by PIDR
Next, to determine the involvement of human CD81 (hCD81), a major receptor candidate for HCV [24], on the PIDR-mediated entry of HCVser, Huh7OK1 cells were pre-treated with anti-hCD81 antibody and inoculated with HCVser treated with PIDR. Although pretreatment with anti-hCD81 antibody resulted in a significant reduction in the entry of HCVser, treatment with PIDR enhanced the entry of HCVser irrespective of the presence of the anti-human CD81antibody (Fig. 5 A). In addition, although entry of HCVser into HepG2 and HepCD81 cells was low and independent of the expression of hCD81, treatment with PIDR enhanced the entry of HCVser irrespective of the expression of human CD81 (Fig. 5B). These results suggest that the PIDR-mediated entry of HCVser is independent of the expression of hCD81 and is effective for the entry of HCVser into various cell lines other than Huh7-derived cell lines.
Fig. 5.
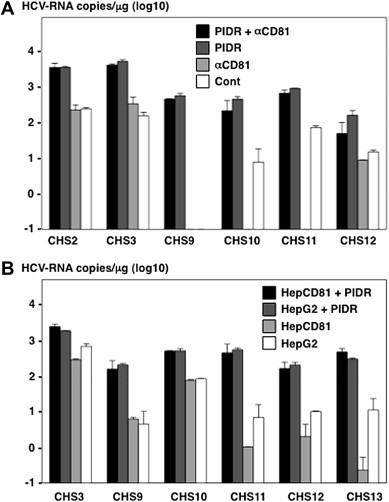
Human CD81-independent entry of HCVser by PIDR. (A) The effect of anti-hCD81 antibody on the entry of HCVser into Huh7OK1 cells in the presence or absence of PIDR. (B) The effect of PIDR on the entry of HCVser into HepG2 and HepCD81 cells. HCV-RNA titers in cells were determined at 24 h post-inoculation.
3.7. Effect of PIDR on the entry of HCVser into immortalized human hepatocytes
Recently, Aly et al. reported that immortalized human hepatocytes, HuS/E2 cells, exhibited a high susceptibility to the infection with HCVser [7]. Therefore, we examined the effect of PIDR on the entry of HCVser into immortalized human hepatocytes, including Hc and HuS/E2 cells. The addition of PIDR enhanced the entry of HCVser into both Hc and HuS/E2 cells (Fig. 6 A and B). These results indicate that PIDR has the potential to enhance the entry of HCVser into not only cancer cell lines but also immortalized hepatocytes. Next, to evaluate the long-term effect of PIDR treatment on the infectivity of HCVser, Hc cells inoculated with CHS pre-incubated with PIDR were cultured for a long period. HCV-RNA could be detected at 10, 15 and 20 days after PIDR-mediated infection (Fig. 6C). However, significant elevations of HCV-RNA titers were not seen (Data not shown).
Fig. 6.
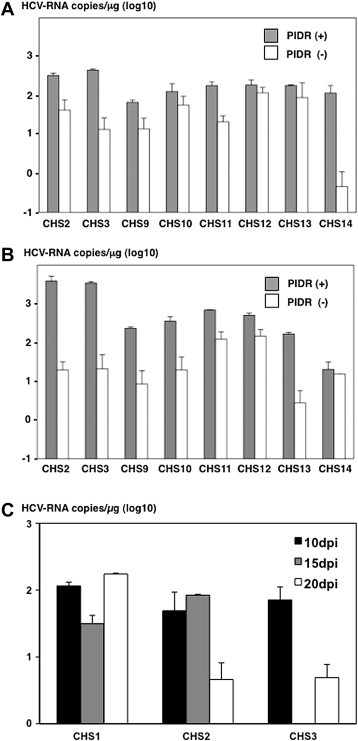
Effect of PIDR on the entry of HCVser into immortalized human hepatocytes. The effect of PIDR on the entry of HCVser into immortalized human hepatocytes, such as Hc (A) and HuS/E2 (B) cells. The HCV-RNA titers in cells were determined at 24 h post-inoculation. (C) HCV-RNA titers in Hc cells inoculated with HCVser were evaluated at 10, 15 and 20 days after PIDR-mediated infection.
4. Discussion
In this study, we examined the efficiency of intracellular deliveries of HCVser by using spinoculation, polybrene and PIDR and found that the PIDR exhibited the highest efficacy on the entry of HCVser into target cells. Especially, trypsinization and reseeding of cells dramatically reduced HCV-RNA levels in groups that were not treated with PIDR as compared to those were treated with PIDR (Fig. 3B and C), and PIDR treatment dramatically increased the internalization of HCVcc treated with CHS or AP-33 at 2 and 4 days after infection (Fig. 2B and C). These results suggest that PIDR is feasible to deliver HCV/CHS complexes into target cells that allow productive infection. In addition, PIDR facilitated the entry of HCVser into the hepatoma cell lines and immortalized human hepatocytes in an hCD81-independent manner. Furthermore, we demonstrated that the intracellular delivery of HCVser by PIDR was enhanced by the addition of anti-HCV antibodies in sera from chronic hepatitis C patients, suggesting that PIDR is an effective reagent for the intracellular delivery of HCVser into the target cells in a receptor-independent manner.
Although direct evidence of enhancement of the adsorption and penetration by the application of spinoculation and polybrene has not been demonstrated yet, sedimentation of the virus particles to the cell surface by the spinoculation and electrostatic interactions between viral particles and cells by the charged polybrene are suggested to overcome the first barrier between virus particles and cells [12], [13], [14]. PIDR is a cationic amphiphilic-based protein delivery reagent that forms a complex with proteins through electrostatic and hydrophobic interactions [17]. The complexes of protein molecules and PIDR have been shown to interact with heparan sulfate proteoglycans on the cell surface, and then to be internalized through endocytosis, after which the protein molecules are released from the complexes into the cytoplasm [17], suggesting that PIDR is capable of enhancing not only adsorption but also penetration of HCVser.
Although HCVser are composed of heterogenous viral populations and a large fraction of the viral particles was associated with lipoproteins or neutralizing antibodies [25], these particles are capable of invading into human hepatocytes and establishing a persistent infection in vivo [1]. Therefore, it is feasible to speculate that some host factors are involved in the entry of HCVser into hepatocytes in vivo. Recently, Stamataki et al. [26] suggested that peripheral blood B lymphocytes participate as a reservoir for HCV for persistent infection and as a vehicle for transinfection to hepatocytes. Although the precise mechanisms of the entry of HCV have not been clarified yet, PIDR is an efficient modality to overcome the obstacles to the entry of HCV.
Recent studies have revealed that at least four cellular molecules play crucial roles in the infection of HCV into hepatocytes in vitro: hCD81, scavenger receptor class B type I (SR-BI) [27], and tight junction proteins claudin-1 [28] and occludin [11]. In this study, the entry of HCVser by the treatment with PIDR was shown to be independent from hCD81. Although the involvement of receptor candidates other than hCD81 was not examined in this study, PIDR was shown to enhance the entry of HCVser in cell lines including Huh7, HepG2, HepCD81, Hc and HuS/E2, suggesting that PIDR is capable of enhancing the entry of HCV through a pathway independent from the expression of these receptor candidates.
Previous studies have indicated that HCV infects not only hepatocytes but also lymphoid tissues and peripheral blood mononuclear cells [29], and that the quasispecies nature of viral particles was different among tissues infected with HCV [10]. Furthermore, it was shown that the in vitro transcribed JFH-1 RNA used for the recovery of infectious particles contained 2.21 × 1011 copies/μg [30], which is much higher than the amount of viral RNA detected in the patient’s sera. The variety of cell tropisms depending on the quasispecies of HCV particles, a low viral load in sera co-existing with neutralization antibodies, and the lack of identified co-factors including functional environment of the liver might be the major obstacles to establishing cell culture systems for the propagation of HCVser. Several approaches have been taken for the establishment of an in vitro cell culture system of HCV, including the culture of human liver cells in a three-dimensional radial-flow bioreactor [31], the three-dimensional culture of immortalized primary hepatocytes [7], and the micropatterned culture of primary hepatocytes [8]. These innovative approaches to the cell culture of liver cells, in combination with PIDR which is able to overcome the first barrier of HCV propagation might contribute to a breakthrough in the establishment of a robust cell culture system of HCVser.
In this study, we demonstrated that PIDR is able to internalize HCV in a receptor-independent manner and provides a clue toward the development of a cell culture system of HCVser in the presence of neutralization antibodies. PIDR may also be useful for the study of viruses that are difficult to internalize into cells due to their low viral titers or the presence of neutralizing antibodies.
Acknowledgments
We thank H. Murase for secretarial work.
This research was supported in part by grants-in-aid from the Ministry of Health, Labor, and Welfare, and the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
References
- 1.Seeff L.-B. Natural history of chronic hepatitis C. Hepatology. 2002;36:S35–S46. doi: 10.1053/jhep.2002.36806. [DOI] [PubMed] [Google Scholar]
- 2.Manns M.-P., McHutchison J.-G., Gordon S.-C., Rustgi V.-K., Shiffman M., Reindollar R., Goodman Z.-D., Koury K., Ling M., Albrecht J.-K. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet. 2001;358:958–965. doi: 10.1016/s0140-6736(01)06102-5. [DOI] [PubMed] [Google Scholar]
- 3.Pawlotsky J.-M., Chevaliez S., McHutchison J.-G. The hepatitis C virus life cycle as a target for new antiviral therapies. Gastroenterology. 2007;132:1979–1998. doi: 10.1053/j.gastro.2007.03.116. [DOI] [PubMed] [Google Scholar]
- 4.Wakita T., Pietschmann T., Kato T., Date T., Miyamoto M., Zhao Z., Murthy K., Habermann A., Krausslich H.-G., Mizokami M., Bartenschlager R., Jake-Liang T. Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat. Med. 2005;11:791–796. doi: 10.1038/nm1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hiraga N., Imamura M., Tsuge M., Noguchi C., Takahashi S., Iwao E., Fujimoto Y., Abe H., Maekawa T., Ochi H., Tateno C., Yoshizawa K., Sakai A., Sakai Y., Honda M., Kaneko S., Wakita T., Chayama K. Infection of human hepatocyte chimeric mouse with genetically engineered hepatitis C virus and its susceptibility to interferon. FEBS Lett. 2007;581:1983–1987. doi: 10.1016/j.febslet.2007.04.021. [DOI] [PubMed] [Google Scholar]
- 6.Molina S., Castet V., Pichard-Garcia L., Wychowski C., Meurs E., Pascussi J.-M., Sureau C., Fabre J.-M., SaCunha A., Larrey D., Dubuisson J., Coste J., McKeating J., Maurel P., Fournier-Wirth C. Serum-derived hepatitis C virus infection of primary human hepatocytes is tetraspanin CD81 dependent. J. Virol. 2008;82:569–574. doi: 10.1128/JVI.01443-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Aly H.-H., Qi Y., Atsuzawa K., Usuda N., Takada Y., Mizokami M., Shimotohno K., Hijikata M. Strain-dependent viral dynamics and virus-cell interactions in a novel in vitro system supporting the life cycle of blood-borne hepatitis C virus. Hepatology. 2009;50:689–696. doi: 10.1002/hep.23034. [DOI] [PubMed] [Google Scholar]
- 8.Ploss A., Khetani S.-R., Jones C.-T., Syder A.-J., Trehan K., Gaysinskaya V.-A., Mu K., Ritola K., Rice C.-M., Bhatia S.-N. Persistent hepatitis C virus infection in microscale primary human hepatocyte cultures. Proc. Natl. Acad. Sci. U. S. A. 2010;107:3141–3145. doi: 10.1073/pnas.0915130107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Burton D.-R. Antibodies, viruses and vaccines. Nat. Rev. Immunol. 2002;2:706–713. doi: 10.1038/nri891. [DOI] [PubMed] [Google Scholar]
- 10.Sobesky R., Feray C., Rimlinger F., Derian N., Dos Santos A., Roque-Afonso A.-M., Samuel D., Brechot C., Thiers V. Distinct hepatitis C virus core and F protein quasispecies in tumoral and nontumoral hepatocytes isolated via microdissection. Hepatology. 2007;46:1704–1712. doi: 10.1002/hep.21898. [DOI] [PubMed] [Google Scholar]
- 11.Ploss A., Evans M.-J., Gaysinskaya V.-A., Panis M., You H., de Jong Y.-P., Rice C.-M. Human occludin is a hepatitis C virus entry factor required for infection of mouse cells. Nature. 2009;457:882–886. doi: 10.1038/nature07684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Landazuri N., Le Doux J.-M. Complexation of retroviruses with charged polymers enhances gene transfer by increasing the rate that viruses are delivered to cells. J. Gene Med. 2004;6:1304–1319. doi: 10.1002/jgm.618. [DOI] [PubMed] [Google Scholar]
- 13.O’Doherty U., Swiggard W.-J., Malim M.-H. Human immunodeficiency virus type 1 spinoculation enhances infection through virus binding. J. Virol. 2000;74:10074–10080. doi: 10.1128/jvi.74.21.10074-10080.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Watanabe R., Matsuyama S., Taguchi F. Receptor-independent infection of murine coronavirus: analysis by spinoculation. J. Virol. 2006;80:4901–4908. doi: 10.1128/JVI.80.10.4901-4908.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ye L., Wang X., Wang S., Luo G., Wang Y., Liang H., Ho W. Centrifugal enhancement of hepatitis C virus infection of human hepatocytes. J. Virol. Methods. 2008;148:161–165. doi: 10.1016/j.jviromet.2007.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Benedicto I., Molina-Jimenez F., Bartosch B., Cosset F.-L., Lavillette D., Prieto J., Moreno-Otero R., Valenzuela-Fernandez A., Aldabe R., Lopez-Cabrera M. The tight junction-associated protein occludin is required for a postbinding step in hepatitis C virus entry and infection. J. Virol. 2009;83:8012–8020. doi: 10.1128/JVI.00038-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Weill C.-O., Biri S., Adib A., Erbacher P. A practical approach for intracellular protein delivery. Cytotechnology. 2008;56:41–48. doi: 10.1007/s10616-007-9102-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Okamoto T., Omori H., Kaname Y., Abe T., Nishimura Y., Suzuki T., Miyamura T., Yoshimori T., Moriishi K., Matsuura Y. A single-amino-acid mutation in hepatitis C virus NS5A disrupting FKBP8 interaction impairs viral replication. J. Virol. 2008;82:3480–3489. doi: 10.1128/JVI.02253-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tani H., Komoda Y., Matsuo E., Suzuki K., Hamamoto I., Yamashita T., Moriishi K., Fujiyama K., Kanto T., Hayashi N., Owsianka A., Patel A.-H., Whitt M.-H., Matsuura Y. Replication-competent recombinant vesicular stomatitis virus encoding hepatitis C virus envelope proteins. J. Virol. 2007;81:8601–8612. doi: 10.1128/JVI.00608-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Aly H.-H., Watashi K., Hijikata M., Kaneko H., Takada Y., Egawa H., Uemoto S., Shimotohno K. Serum-derived hepatitis C virus infectivity in interferon regulatory factor-7-suppressed human primary hepatocytes. J. Hepatol. 2007;46:26–36. doi: 10.1016/j.jhep.2006.08.018. [DOI] [PubMed] [Google Scholar]
- 21.Zhong J., Gastaminza P., Cheng G., Kapadia S., Kato T., Burton D.-R., Wieland S.-F., Uprichard S.-L., Wakita T., Chisari F.-V. Robust hepatitis C virus infection in vitro. Proc. Natl. Acad. Sci. U. S. A. 2005;102:9294–9299. doi: 10.1073/pnas.0503596102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Morris T., Robertson B., Gallagher M. Rapid reverse transcription-PCR detection of hepatitis C virus RNA in serum by using the TaqMan fluorogenic detection system. J. Clin. Microbiol. 1996;34:2933–2936. doi: 10.1128/jcm.34.12.2933-2936.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Clayton R.F., Owsianska A., Aitken J., Graham S., Bhella D., Patel A.H. Analysis of antigenicity and topology of E2 glycoprotein present on recombinant hepatitis C virus-like particles. J. Virol. 2002;76:7672–7682. doi: 10.1128/JVI.76.15.7672-7682.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pileri P., Uematsu Y., Campagnoli S., Galli G., Falugi F., Petracca R., Weiner A.-J., Houghton M., Rosa D., Grandi G., Abrignani S. Binding of hepatitis C virus to CD81. Science. 1998;282:938–941. doi: 10.1126/science.282.5390.938. [DOI] [PubMed] [Google Scholar]
- 25.Lavillette D., Morice Y., Germanidis G., Donot P., Soulier A., Pagkalos E., Sakellariou G., Intrator L., Bartosch B., Pawlotsky J.-M., Cosset F.-L. Human serum facilitates hepatitis C virus infection, and neutralizing responses inversely correlate with viral replication kinetics at the acute phase of hepatitis C virus infection. J. Virol. 2005;79:6023–6034. doi: 10.1128/JVI.79.10.6023-6034.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stamataki Z., Shannon-Lowe C., Shaw J., Mutimer D., Rickinson A.-B., Gordon J., Adams D.-H., Balfe P., McKeating J.-A. Hepatitis C virus association with peripheral blood B lymphocytes potentiates viral infection of liver-derived hepatoma cells. Blood. 2009;113:585–593. doi: 10.1182/blood-2008-05-158824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Scarselli E., Ansuini H., Cerino R., Roccasecca R.-M., Acali S., Filocamo G., Traboni C., Nicosia A., Cortese R., Vitelli A. The human scavenger receptor class B type I is a novel candidate receptor for the hepatitis C virus. EMBO. J. 2002;21:5017–5025. doi: 10.1093/emboj/cdf529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Evans M.-J., von Hahn T., Tscherne D.-M., Syder A.-J., Panis M., Wolk B., Hatziioannou T., McKeating J.-A., Bieniasz P.-D., Rice C.-M. Claudin-1 is a hepatitis C virus co-receptor required for a late step in entry. Nature. 2007;446:801–805. doi: 10.1038/nature05654. [DOI] [PubMed] [Google Scholar]
- 29.Blackard J.-T., Kemmer N., Sherman K.-E. Extrahepatic replication of HCV: insights into clinical manifestations and biological consequences. Hepatology. 2006;44:15–22. doi: 10.1002/hep.21283. [DOI] [PubMed] [Google Scholar]
- 30.Kato T., Date T., Miyamoto M., Furusaka A., Tokushige K., Mizokami M., Wakita T. Efficient replication of the genotype 2a hepatitis C virus subgenomic replicon. Gastroenterology. 2003;125:1808–1817. doi: 10.1053/j.gastro.2003.09.023. [DOI] [PubMed] [Google Scholar]
- 31.Aizaki H., Nagamori S., Matsuda M., Kawakami H., Hashimoto O., Ishiko H., Kawada M., Matsuura T., Hasumura S., Matsuura Y., Suzuki T., Miyamura T. Production and release of infectious hepatitis C virus from human liver cell cultures in the three-dimensional radial-flow bioreactor. Virology. 2003;314:16–25. doi: 10.1016/s0042-6822(03)00383-0. [DOI] [PubMed] [Google Scholar]


