Abstract
Feline infectious peritonitis virus (FIPV) can cause a lethal disease in cats, feline infectious peritonitis (FIP). The antibody-dependent enhancement (ADE) of FIPV infection has been recognised in experimentally infected cats, and cellular immunity is considered to play an important role in preventing the onset of FIP. To evaluate the importance of cellular immunity for FIPV infection, monoclonal antibodies (MAbs) against feline interferon (fIFN)-γ were first created to establish fIFN-γ detection systems using the MAbs. Six anti-fIFN-γ MAbs were created. Then, the difference in epitope which those MAbs recognise was demonstrated by competitive enzyme-linked immunosorbent assay (ELISA) and IFN-γ neutralisation tests. Detection systems for fIFN-γ (sandwich ELISA, ELISpot assay, and two-colour flow cytometry) were established using anti-fIFN-γ MAbs that recognise different epitopes. In all tests, fIFN-γ production from peripheral blood mononuclear cells (PBMCs) obtained from cats experimentally infected with an FIPV isolate that did not develop the disease was significantly increased by heat-inactivated FIPV stimulation in comparison with medium alone. Especially, CD8+fIFN-γ+ cells, but not CD4+fIFN-γ+ cells, were increased. In contrast, fIFN-γ production from PBMCs isolated from cats that had developed FIP and specific pathogen-free (SPF) cats was not increased by heat-inactivated FIPV stimulation. These results suggest that cellular immunity plays an important role in preventing the development of FIP. Measurement of fIFN-γ production with the anti-fIFN-γ MAbs created in this study appeared to be useful in evaluating cellular immunity in cats.
Feline coronavirus (FCoV) is a coronavirus belonging to group 1 of the family Coronaviridae. FCoV is classified into types I and II according to the amino acid sequence of its spike protein. 1–3 Each of these types consists of two viruses: feline infectious peritonitis (FIP)-causing FIP virus (FIPV) and non-FIP-causing feline enteric coronavirus (FECV). 4 FIPV and FECV of the same type cannot be distinguished by their antigenicity or at the gene level, and differ only in their pathogenicity for cats. 5 Thus, there are types I and II FECV and FIPV in FCoV. FECV is asymptomatic in cats, 6 but FIPV infection can induce FIP, ie, FIPV is a virulence biotype of the FECV. 5 There is no detailed evidence, but it is thought that certain strains of FECV are more likely to mutate to FIPVs. 7,8 FIP is a fatal disease, characterised by a monocyte-triggered vasculitis accompanied by pyogranulomatous inflammation. 5,9
To prevent FIP, various vaccines, such as virulence-attenuated live or inactivated FIPV vaccines, have been investigated, but none have shown sufficient efficacy, and these vaccines rather enhance the development of FIP. 10–14 Intraperitoneal inoculation with FIPV induced more severe clinical signs in anti-FIPV antibody-positive kittens and kittens that received passive immunisation with serum or purified IgG from antibody-positive cats, compared to antibody-negative kittens. 15,16 These results of experimental studies suggest that antibody-dependent enhancement (ADE) of FIPV infection can be a serious obstacle to the prevention of FIP by vaccination. Potent cellular immunity was induced in FIPV-infected cats without FIP. 17–20 Cellular immunity is considered to play an important role in the prevention of FIP onset. 5 Thus, one safety concern regarding FIPV vaccine is that it may induce marked cellular immunity. 5,17–20 Therefore, evaluation of the cellular immune response is essential for the development of vaccines against FIPV infection.
Methods to evaluate cellular immunity in cats include the lymphocyte transformation test, 21 delayed hypersensitivity reaction (DTH), 19,20 measurement of IL-2 production using IL-2-dependent cell lines, 22 and measurement of interferon (IFN)-γ mRNA using the reverse transcriptase polymerase chain reaction (RT-PCR). 17,18 Measurement of the secreted IFN-γ is a method that can be easily employed to evaluate the cellular immune response. Sandwich enzyme-linked immunosorbent assay (ELISA) and ELISpot kits (R&D systems, USA) are commercially available for the measurement of secreted feline IFN (fIFN)-γ. However, these fIFN-γ-measuring kits are very expensive. In addition, they raise concerns about non-specific reactions due to the use of an anti-fIFN-γ polyclonal antibody (PAb) as a capture and detection antibody.
In other species, the tools that employed anti-IFN-γ monoclonal antibodies (MAbs) have been used for the measurement of IFN-γ. Sandwich ELISA has been used to quantify the levels of IFN-γ in tissue culture supernatants. 23 ELISpot assay has been employed to enumerate IFN-γ-secreting cells following stimulation with cognate antigen. 24 Flow cytometry has been used to characterise antigen-specific IFN-γ-secreting cells. 25,26
In this study, fIFN-γ production was measured to evaluate the importance of cellular immunity in the course of experimental FIPV infection. First, MAbs against fIFN-γ were created and examined regarding their specificity. Subsequently, peripheral blood mononuclear cells (PBMCs) obtained from FIPV-infected cats without FIP, FIP cats, and specific pathogen-free (SPF) cats were cultured with heat-inactivated FIPV. Then, fIFN-γ was detected employing sandwich ELISA, ELISpot assay, and two-colour flow cytometry with the MAbs.
Materials and methods
Expression of recombinant fIFN (rfIFN)-γ
The Escherichia coli TOP10 strain transfected with the fIFN-γ gene was provided by the Department of Veterinary Microbiology, School of Veterinary Medicine and Animal Sciences, Kitasato University. The transfected E coli were cultured, and then harvested in phosphate buffered saline (PBS). The suspension was sonicated and centrifuged. The supernatant was purified by affinity chromatography using nickel.
Production of anti-fIFN-γ MAbs
For the preparation of MAbs against fIFN-γ, 4-week-old female BALB/c mice were inoculated intraperitoneally with a mixture of 200 μg of rfIFN-γ and 109 cells of pertussis adjuvant. Mice received a booster every 2 weeks. The mice were immunised four times in total. Then, the mice were sacrificed to obtain splenic cells for fusion. Cell fusion was carried out by employing essentially the same method as described by Köhler and Milstein. 27 The fused cells were plated into 96-well flat-bottomed microplates (Greiner Bio-One, Germany) and incubated at 37°C. The supernatant was screened for the production of anti-fIFN-γ antibodies by ELISA (see below). The cells in antibody-positive wells were then cloned by limiting dilution.
The isotype of each MAb was determined using a commercial mouse MAb isotyping kit (AbD Serotec, UK) according to the manufacturer's protocol. The purification and concentration of MAbs were carried out employing a commercial antibody purification Kit (Millipore, USA) according to the manufacturer's protocol. The biotinylation of purified MAbs was carried out using a commercial biotinylation kit (Thermo Fisher Scientific, USA) according to the manufacturer's protocol.
ELISA
The supernatants of hybridoma cultures were screened for the production of anti-fIFN-γ antibodies by ELISA. ELISA plates (Toyoshima Mfg, Japan) were coated overnight at 4°C with rfIFN-γ (200 ng/100 μl/well) diluted with carbonate buffer (0.05 M, pH 9.6). After washing with PBS containing 0.02% Tween-20, each well of the plates received 100 μl of the supernatant. After 60 min incubation at 37°C, the plates were washed and peroxidase (POD)-conjugated goat anti-mouse IgG, IgM, and IgA (MP Biomedicals, LLC-Cappel Products, USA) was diluted to the optimal concentrations, and then 100 μl of the dilution was added to each well of the plates. After incubation at 37°C for 30 min, the plates were washed and each well received 100 μl of substrate solution and were incubated at 25°C for 20 min in the dark. The substrate solution was prepared by dissolving o-phenylenediamine dihydrochloride at a concentration of 0.4 mg/ml in 0.1 M citric acid and 0.2 M Na2HPO4 buffer (pH 4.8) and adding 0.2 μl/ml of 30% H2O2. The reaction was stopped with 3 N H2SO4 solution, and the optical density (OD) at 492 nm was determined.
Western immunoblotting assay
Commercial rfIFN-γ (R&D Systems, USA) was run employing 12% sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) and a transferred nitrocellulose membrane. Protein marker (Precision Plus Protein Standards) was purchased from Bio-Rad. The blots were blocked with 5% non-fat dry milk powder in TBST (20 mM Tris–HCl, pH 8.0, 0.88% NaCl, 0.05% Tween-20) for 1 h at 37°C, followed by 1 h incubation at 37°C with each anti-fIFN-γ MAb. Following washing, the membrane was incubated with POD-conjugated goat anti-mouse IgG, IgM, and IgA for 1 h at 37°C, and then visualised in substrate for 10 min.
Competitive ELISA
Each well of 96-well ELISA plates coated with rfIFN-γ received 100 μl of each purified anti-fIFN-γ MAb, and the plates were incubated at 37°C for 1 h. After washing, 100 μl of each biotinylated MAb was added and the plates were incubated at 37°C for 1 h. After washing, an optimal dilution of horseradish peroxidase (HRP)-conjugated streptavidin (Millipore, USA) was added and the plates were incubated at 37°C for 30 min. The subsequent treatment was the same as for conventional ELISA (described above). The percentage competition was determined by the formula [100(A−n)]/(A−B), where A is the OD in the absence of competing antibody, B is the OD in the presence of homologous antibody, and n is the OD in the presence of competitor.
IFN-γ neutralisation tests
The capacity of each anti-fIFN-γ MAb to neutralise the fIFN-γ antiviral activity was determined. For this, Felis catus whole fetus (fcwf)-4 cells (2×104 cells/well) were sown on flat-bottomed 96-well plates in Eagle's minimum essential medium and L-15 medium supplemented with 5% fetal bovine serum (FBS) (Trace Science, Australia) and penicillin at 100 U/ml and streptomycin at 100 μg/ml. Each anti-fIFN-γ MAb, two-fold diluted, was mixed with an equal volume of 40 laboratory units (LU)/ml of commercial rfIFN-γ for 1 h at 37°C. Each mixture was then added to the fcwf-4 cell cultures, and incubation was carried out for 24 h at 37°C. After the supernatant was removed, 100 TCID50/well of previously titrated vesicular stomatitis virus (VSV) was used for inoculation (except to the cell control). This was again incubated for 72 h at 37°C. It was decided whether or not each MAb showed neutralisation activity against fIFN-γ based on the presence of CPE.
Viruses
The type I FIPV KU-2 strain was isolated in our laboratory, and type II FIPV 79-1146 strain was supplied by Dr M C Horzinek of State University Utrecht, The Netherlands. FIP did not develop in cats oronasally inoculated with the FIPV KU-2 strain, but developed in about 50% of intraperitoneally inoculated cats (unpublished data). When cats passively immunised with anti-FCoV antibody were subcutaneously inoculated with the FIPV KU-2 strain, about 80% of them developed FIP. 28 The orally inoculated FIPV 79-1146 strain has been shown to induce FIP in about 90% of anti-FCoV antibody-negative cats. 5
Experimental animals
In this study, we used 19 cats aged 1–5 years old. Six were anti-FCoV antibody-negative SPF cats (SPFa, SPFb, SPFc, SPFd, SPFe, and SPFf). Five cats (Ia, Ib, Ic, Id, and Ie) were inoculated oronasally with the type I FIPV KU-2 strain (105 TCID50/head), and five (IIa, IIb, IIc, IId, and IIe) were inoculated oronasally with the type II FIPV 79-1146 strain (105 TCID50/head) experimentally, and these cats developed no clinical signs of FIP (FIPV-infected cats without FIP). Three cats were inoculated oronasally with the FIPV 79-1146 strain, and developed clinical signs of typical FIP (FIPa, FIPb, and FIPc). When the FCoV gene was investigated in PBMCs of FIPV-infected cats employing RT-PCR, 29 the FCoV gene was detected 4–12 days after type II FIPV inoculation but not after 18 days. In addition, the FCoV gene was detected in faeces until 18 days after type II FIPV inoculation. In contrast, the FCoV gene was not detected in PBMCs of type I FIPV-infected cats after type I FIPV inoculation, but sporadically detected in faeces for a prolonged period after type I FIPV inoculation. All FIPV-inoculated cats were judged as infected with FIPV based on an increase in the anti-FCoV antibody level in serum collected after FIPV inoculation on ELISA. These cats were maintained in a temperature-controlled, isolated facility. All experiments were performed in accordance with the Guidelines for Animal Experiments of Kitasato University.
Isolation of PBMCs
PBMCs were prepared by the density gradient centrifugation of heparinised peripheral blood samples obtained from the cats. 30 Briefly, a volume of peripheral blood was diluted with an equal volume of PBS and layered on Ficoll-Hypaque (Axis-Shield PoC AS, Norway). After centrifugation, the layer containing the PBMCs fraction was obtained and suspended in RPMI 1640 (Sigma, USA) containing 5% heat-inactivated SPF cat serum, penicillin at 100 U/ml, streptomycin at 100 μg/ml, and 50 μM 2-mercaptoethanol. PBMCs of FIPV-infected cats without FIP were collected 6 months after FIPV infection and those of FIP cats were collected when they were euthanased.
Measurement of fIFN-γ concentrations in PBMCs culture supernatants using sandwich ELISA
For measurement of the fIFN-γ level in the culture supernatants, PBMCs (5×106 cells/ml) were cultured with heat-inactivated FIPV or culture medium alone as a negative control at 37°C for 9 days. For heat-inactivated FIPV, FIPV culture fluid heated at 56°C for 30 min was used. Regarding the incubation period, 3-, 6-, 9-, and 12-day incubations were examined in a previous study, in which 9-day incubation was determined to be optimal to evaluate the fIFN-γ level in PBMCs culture supernatants (data not shown). ELISA plates (Thermo Fisher Scientific, USA) were coated with 100 μl of the unlabelled anti-fIFN-γ MAb (5 μg/ml) in carbonated buffer at 4°C overnight. After washing, the plates were blocked with a blocking buffer containing 25% Block Ace (DS Pharma Biomedical, Japan) in PBS. After another washing, 100 μl of the culture supernatants and standard samples of rfIFN-γ were added to each well and incubated at 37°C for 1 h. After a further washing, 100 μl of the biotinylated anti-fIFN-γ MAb (1 μg/ml) was added to each well and the plates were incubated at 37°C for 1 h. After washing again, an optimal dilution of HRP-conjugated streptavidin was added and the plates were incubated at 37°C for 30 min. The subsequent treatment was the same as for conventional ELISA (described above). The minimum detectable concentration was defined by the standard deviation of dose measurement at a zero dose or the background. The levels of fIFN-γ in the supernatants were interpolated from the rfIFN-γ standard calibration curve.
Counting of fIFN-γ-secreting cells using ELISpot assay
ELISpot assays were performed using a commercial Protein Detector HRP ELISpot Kit (KPL, USA). Briefly, a 96-well polyvinylidene fluoride (PVDF)-backed microplate was coated with 10 μg/ml of unlabelled capture MAb at 4°C overnight. The plate was blocked at 25°C for 1 h. The plate received 1×106/ml of PBMCs and was incubated with the heat-inactivated FIPV 79-1146 strain (105 TCID50/ml) or culture medium alone as a negative control at 37°C for 48 h. After PBMCs were removed, the plate received 2 μg/ml of biotinylated detection MAb and was incubated at 25°C for 1 h. The plate was processed according to the manufacturer's instructions. Resulting spots were counted with a stereomicroscope (Zellnet Consulting, USA). Only blue-coloured spots with fuzzy borders were scored as spot-forming cells (SFCs).
Measurement of CD4+fIFN-γ + and CD8 + fIFN-γ + cells using flow cytometry assay
PBMCs were resuspended at a cell concentration of 4×106/ml and stimulated with the heat-inactivated FIPV 79-1146 strain (105 TCID50/ml) for 48 h at 37°C. Then, the cells were cultured in the presence of the protein secretion inhibitor brefeldin A (BFA) (10 μg/106 cells, Sigma–Aldrich, USA) for 4 h at 37°C. Unstimulated cells were incubated as a negative control. PBMCs were harvested, washed in PBS containing 0.5% bovine serum albumin (BSA) and 1 mM NaN3, and incubated with fluorescein isothiocyanate (FITC)-labelled anti-feline CD4 MAb (Southern Biotechnology Associates, USA) or FITC-labelled anti-feline CD8 MAb (Southern Biotechnology Associates, USA) for 30 min at 4°C in the dark. After washing, cells were fixed and permeabilised using a Fixation/Permeabilisation kit (BD Biosciences, USA) according to the manufacturer's instructions. Permeabilised cells were incubated with biotinylated anti-fIFN-γ MAb for 1 h at 25°C. The cells were incubated with phycoerythrin (PE)-labelled streptavidin (Beckman Coulter, USA) for 1 h at 25°C in the dark. After washing, the cells were stored in fluorescence buffer prior to analysis on a flow cytometer (Cytomics FC500, Beckman Coulter, USA), and then analysed using FlowJo software (TreeStar, USA). The small lymphocyte and lymphoblast populations were gated on the basis of the cell size and granularity (forward and side scatter). For each sample 100,000 events were recorded, and the percentage of CD4+fIFN-γ + and CD8+fIFN-γ + cells population was calculated.
Statistical analysis
The results of sandwich ELISA and ELISpot assay are expressed as means±standard error of the mean (SEM). Student's t-test was employed to determine significant differences between stimulated and unstimulated PBMCs cultures in each cat. A P-value of < 0.01 or P<0.05 was considered significant.
Results
Characterisation and specificity of anti-fIFN-γ MAbs
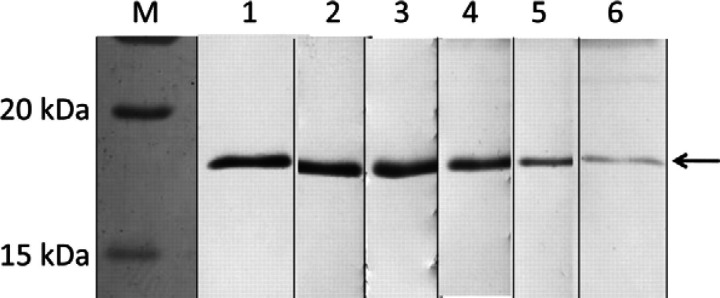
BALB/c mice were immunised with rfIFN-γ. Then, the culture supernatants of hybridomas were screened using ELISA. Finally, six anti-fIFN-γ MAbs (Noko 1-12.3, Noko 3-31.1, Noko 5-23.1, Ame 4-12.4, Ame 6-31.1, and Ame 7-19.1) were obtained. The isotypes of these MAbs were IgG2a (for Noko 3-31.1) and IgG1 (for other five MAbs). The reactivity of the MAbs against rfIFN-γ was examined by Western immunoblotting. For all six MAbs, an rfIFN-γ-specific band was observed at around 18 kDa (Fig 1).
Fig 1.
Reactivity of each MAb against rfIFN-γ using Western immunoblotting. M, marker; lane 1, Noko 1-12.3; lane 2, Noko 3-31.1; lane 3, Noko 5-23.1; lane 4, Ame 4-12.4; lane 5, Ame 6-31.1; lane 6, Ame 7-19.1. The arrow shows rfIFN-γ-specific band which is observed at around 18 kDa.
Difference in epitopes recognised by the MAbs
To examine the difference in epitopes recognised by the MAbs, competitive ELISA and fIFN-γ neutralisation tests were carried out. Competitive ELISA demonstrated 80% or higher inhibition rates for four MAbs (Noko 1-12.3, Noko 3-31.1, Noko 5-23.1, and Ame 4-12.4). The fIFN-γ neutralisation test demonstrated that three MAbs (Noko 3-31.1, Ame 6-31.1, and Ame 7-19.1) exhibited neutralising activity against fIFN-γ (Table 1). These results suggest that Noko 1-12.3, Noko 5-23.1, and Ame 4-12.4 recognise the same epitope. Noko 3-31.1 inhibited 80% or more of the binding of Noko 1-12.3, Noko 5-23.1, and Ame 4-12.4 to rfIFN-γ. However, Noko 3-31.1 differed from these MAbs in that it showed neutralising activity against fIFN-γ. Ame 6-31.1 and Ame 7-19.1 mutually inhibited 80% or more of their binding to rfIFN-γ. Both showed neutralising activity against fIFN-γ. However, they seemed to recognise different epitopes because of the different inhibition rates for Noko 3-31.1 and Noko 5-23.1. Thus, Noko 1-12.3, Noko 5-23.1, and Ame 4-12.4 recognised the same epitope, while the other MAbs recognised different epitopes.
Table 1.
Analysis of epitopes recognised by the each anti-fIFN-γ MAb using competitive ELISA and fIFN-γ neutralisation tests.
| Biotinylated anti-fIFN-γ MAb | Neutralisation activity | Epitope | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Noko 1-12.3 | Noko 5-23.1 | Ame 4-12.4 | Noko 3-31.1 | Ame 6-31.1 | Ame 7-19.1 | ||||
| Unlabelled anti-fIFN-γ MAb | Noko 1-12.3 |
 *
*
|
+ | + | + | − | − | − | A |
| Noko 5-23.1 | + |

|
+ | + | − | − | − | A | |
| Ame 4-12.4 | + | + |

|
+ | − | − | − | A | |
| Noko 3-31.1 | + | + | + |

|
76.4 | − | + | B | |
| Ame 6-31.1 | − | − | − | − |

|
+ | + | C | |
| Ame 7-19.1 | − | + | − | 69.8 | + |

|
+ | D | |
,+ and − show the percentage of competition more than 80% and less than 20%, respectively.
Detection of fIFN-γ in the culture supernatants of PBMCs using sandwich ELISA
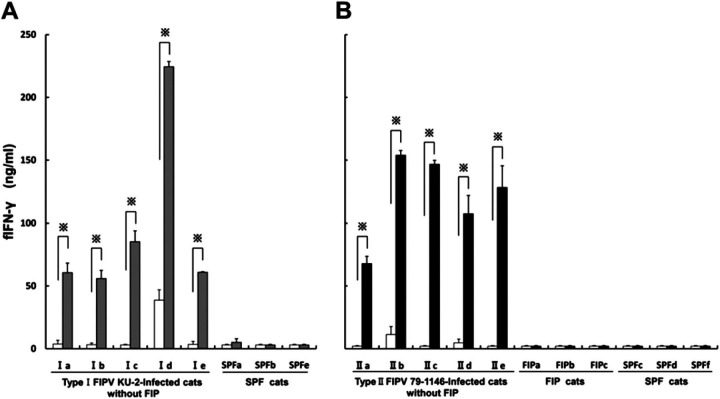
PBMCs were isolated from FIPV-infected cats without FIP, FIP cats, and SPF cats and cultured for 9 days with or without a heat-inactivated FIPV. The fIFN-γ concentrations in the culture supernatants were measured by sandwich ELISA. The combination of unlabelled capture MAb and biotinylated detection MAb, used in sandwich ELISA, was examined based on the results of competitive ELISA and IFN-γ neutralisation tests. The results demonstrated that the detectability was highest when Ame 6-31.1 was used as a capture antibody and biotinylated Ame 4-12.4 as a detection antibody. The detection range of the assay was from 0.1 to 100 ng/ml.
On unstimulated culture, the fIFN-γ concentrations in PBMCs culture supernatants of type I FIPV KU-2 strain-infected cats without FIP, Ia, Ib, Ic, Id, and Ie were 3.7±3.0, 3.1±1.3, 0.6±0.4, 38.7±8.2, and 3.4±2.4 ng/ml, respectively. On virus-stimulated culture, the values increased to 60.7±7.5, 55.9±6.4, 85.3±8.7, 224.5±4.2, and 61.0±0.4 ng/ml, respectively; significant differences were noted in all animals (P<0.01, Fig 2A). In SPF cats, SPFa, SPFb, and SPFe, the fIFN-γ levels in unstimulated culture were <0.1, 0.5±0.4, and 0.1±0.1 ng/ml, respectively. The values in virus-stimulated culture were 5.1±2.9, <0.1, and <0.1 ng/ml, respectively; significant differences were absent in three SPF cats.
Fig 2.
Measurement of the concentration of fIFN-γ in the PBMCs culture supernatants using sandwich ELISA. PBMCs were derived from 10 FIPV-infected cats without FIP (Ia–Ie and IIa–IIe were inoculated with the type I FIPV KU-2 and type II FIPV 79-1146 strains, respectively), three FIP cats (FIPa–FIPc) and six SPF cats (SPFa–SPFf). The cells were cultured with the heat-inactivated FIPV KU-2 strain (grey bar; A), FIPV 79-1146 strain (solid bar; B), or culture medium alone (open bar). Each experiment was performed in quadruplicate. The results are expressed as means±SEM. ∗ indicates a significant difference by the t-test (P<0.01).
On unstimulated culture, the fIFN-γ concentrations in PBMCs culture supernatants of type II FIPV 79-1146 strain-infected cats without FIP, IIa, IIb, IIc, IId, and IIe were <0.1, 11.3±6.4, <0.1, 4.7±2.9, and <0.1 ng/ml, respectively. On virus-stimulated culture, the values increased to 67.8±5.8, 154.0±3.7, 146.7±3.1, 107.5±14.3, and 128.2±17.3 ng/ml, respectively; significant differences were noted in all animals (P<0.01, Fig 2B). In FIP cats, FIPa, FIPb, and FIPc, the fIFN-γ levels in unstimulated culture were 0.1±0.1, <0.1, and <0.1 ng/ml, respectively. The values in virus-stimulated culture were 0.1±0.1, <0.1, and <0.1 ng/ml, respectively; significant differences were absent in three FIP cats. In SPF cats, SPFc, SPFd, and SPFf, the fIFN-γ levels in unstimulated culture were 0.2±0.1, 1.4±1.1, and 0.1±0.1 ng/ml, respectively. The values in virus-stimulated culture were 0.1±0.1, <0.1, and <0.1 ng/ml, respectively; these differences were not significant.
Counting of fIFN-γ-secreting cells among PBMCs using ELISpot assay
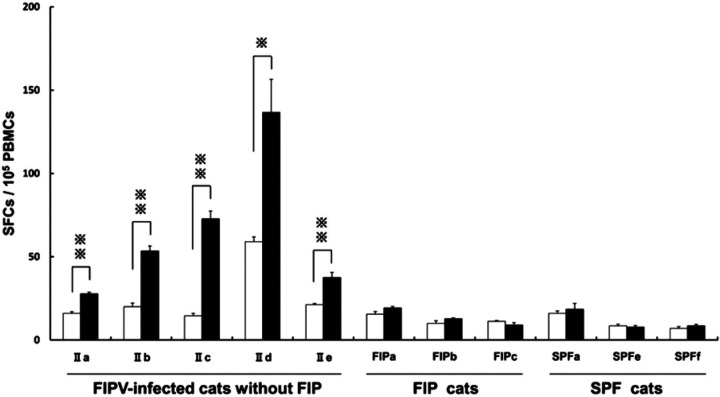
The combination of unlabelled capture MAb and biotinylated detection MAb, used for ELISpot assay, was examined. As in sandwich ELISA, detectability was highest when Ame 6-31.1 was used as a capture antibody and biotinylated Ame 4-12.4 as a detection antibody. PBMCs were isolated from FIPV-infected cats without FIP, FIP cats, and SPF cats. Of the PBMCs, fIFN-γ-secreting cells were counted in 48 h culture with or without a heat-inactivated FIPV on an ELISpot plate coated with capture MAb.
On unstimulated culture, the numbers of fIFN-γ-secreting cells among PBMCs obtained from type II FIPV-infected cats without FIP, IIa, IIb, IIc, IId, and IIe were 16.0±2.1, 20.0±4.4, 14.5±3.0, 59.0±5.1, and 21.3±1.3 SFCs/105 PBMCs, respectively. In virus-stimulated culture, the values increased to 27.8±1.9, 53.5±5.9, 72.8±9.3, 136.7±34.4, and 37.5±6.3 SFCs/105 PBMCs, respectively; significant differences were noted for all the animals (P<0.01, IIa, IIb, IIc, and IIe; P<0.05, IId; Fig 3). In FIP cats, FIPa, FIPb, and FIPc, the numbers of fIFN-γ secreting cells among unstimulated PBMCs were 15.5±3.2, 10.0±3.2, and 11.3±0.8 SFCs/105 PBMCs, respectively. The values in virus-stimulated culture were 19.3±1.8, 12.8±1.1, and 9.0±2.7 SFCs/105 PBMCs, respectively; significant differences were absent in three FIP cats. In SPF cats, SPFa, SPFe, and SPFf, the numbers of fIFN-γ secreting cells among unstimulated PBMCs were 16.0±3.0, 8.5±2.1, and 7.0±2.1 SFCs/105 PBMCs, respectively. The values in virus-stimulated culture were 18.5±7.0, 7.8±1.9, and 8.5±1.8 SFCs/105 PBMCs, respectively; these differences were not significant.
Fig 3.
Count of spot-forming fIFN-γ-secreting cells using ELISpot assay. PBMCs were obtained from five FIPV-infected cats without FIP (IIa–IIe were inoculated with the type II FIPV 79-1146 strain), three FIP cats (FIPa–FIPc), and three SPF cats (SPFa, SPFe, and SPFf). The cells were cultured with the heat-inactivated type II FIPV 79-1146 strain (solid bar) or culture medium alone (open bar). Each experiment was performed in quadruplicate. The results are expressed as means±SEM. ∗ and ∗∗, P<0.05 and P<0.01, respectively.
Detection of CD4+fIFN-γ + and CD8 + fIFN-γ + cells using two-colour flow cytometry
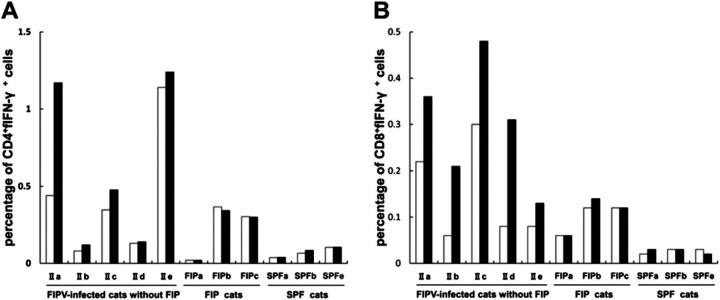
Examining biotinylated MAb used for two-colour flow cytometry demonstrated that detectability was highest for biotinylated Ame 4-12.4. PBMCs were isolated from FIPV-infected cats without FIP, FIP cats, and SPF cats and cultured for 48 h with or without a heat-inactivated FIPV. Subsequently, to measure intracellular fIFN-γ, PBMCs were cultured for 4 h in the presence of a protein secretion inhibitor, BFA. Percentages of CD4+fIFN-γ + and CD8+fIFN-γ + cells were compared in stimulated and unstimulated cells using flow cytometry.
On unstimulated culture, the percentages of CD4+fIFN-γ + cells in type II FIPV-infected cats without FIP, IIa, IIb, IIc, IId, and IIe were 0.44, 0.08, 0.35, 0.13, and 1.14%, respectively. On virus-stimulated culture, the percentages were 1.17, 0.12, 0.48, 0.14, and 1.24%, respectively (Fig 4A). The values increased 2.66-, 1.50-, 1.37-, 1.08-, and 1.09-fold, respectively. In FIP cats, FIPa, FIPb, and FIPc, the percentages of CD4+fIFN-γ + cells in unstimulated culture were 0.02, 0.37, and 0.30%, respectively. The percentages in virus-stimulated culture were 0.02, 0.34, and 0.30%, respectively. The values increased 1.00-, 0.92-, and 1.00-fold, respectively. In SPF cats, SPFa, SPFb, and SPFe, the percentages of CD4+fIFN-γ + cells in unstimulated culture were 0.04, 0.07, and 0.10%, respectively. The percentages in virus-stimulated culture were 0.04, 0.08, and 0.11%, respectively. The values increased 1.00-, 1.14-, and 1.10-fold, respectively.
Fig 4.
Calculation of the percentage of CD4+fIFN-γ + and CD8+fIFN-γ + cells in small lymphocyte and lymphoblast populations using two-colour flow cytometry. PBMCs were derived from five FIPV-infected cats without FIP (IIa–IIe were inoculated with the type II FIPV 79-1146 strain), three FIP cats (FIPa–FIPc), and three SPF cats (SPFa, SPFb, and SPFe). The cells were cultured with the heat-inactivated type II FIPV 79-1146 strain (solid bar) or culture medium alone (open bar). A and B represent the percentage of CD4+fIFN-γ + and CD8+fIFN-γ + cells, respectively.
On unstimulated culture, the percentages of CD8+fIFN-γ + cells in type II FIPV-infected cats without FIP, IIa, IIb, IIc, IId, and IIe were 0.22, 0.06, 0.30, 0.08, and 0.08%, respectively. On virus-stimulated culture, the percentages were 0.36, 0.21, 0.48, 0.31, and 0.13%, respectively (Fig 4B). The values increased 1.64-, 3.50-, 1.60-, 3.88-, and 1.63-fold, respectively. In FIP cats, FIPa, FIPb, and FIPc, the percentages of CD8+fIFN-γ + cells in unstimulated culture were 0.06, 0.12, and 0.12%, respectively. The percentages in virus-stimulated culture were 0.06, 0.14, and 0.12%, respectively. The values increased 1.00-, 1.17-, and 1.00-fold, respectively. In SPF cats, SPFa, SPFb, and SPFe, the percentages of CD8+fIFN-γ + cells in unstimulated culture were 0.02, 0.03, and 0.03%, respectively. The percentages in virus-stimulated culture were 0.03, 0.03, and 0.02%, respectively. The values increased 1.5-, 1.00-, and 0.67-fold, respectively.
Discussion
Cellular immunity is considered important for the prevention of development of FIP in FCoV infected cats. 5,17–20 Therefore, evaluation of the cellular immune response is essential for the development of vaccines against FIPV infection. We focused on IFN-γ because it has an important role as an immunoregulator. This role has been exploited to obtain important information on the specificity and efficacy of the cellular immune response. 31–33
Anti-IFN-γ MAbs are used for detecting IFN-γ production both directly and specifically. Sandwich ELISA is a simple and robust assay, and a good choice to quantitatively measure IFN-γ. ELISpot assay is designed for the detection of IFN-γ-secreting cells at the single cell level, and can be used to quantify the frequency of IFN-γ-secreting cells. Flow cytometry facilitates the detection of intracellular IFN-γ and quantification of IFN-γ-secreting cells directly. Moreover, two-colour flow cytometry, employing a combination of cell surface and intracellular IFN-γ staining, enables the characterisation of IFN-γ-secreting cells.
All six anti-fIFN-γ MAbs produced in this study specifically reacted with commercial rfIFN-γ based on Western immunoblotting assays. This revealed the specificity of anti-fIFN-γ MAbs. Furthermore, the results from competitive ELISA and IFN-γ neutralisation tests demonstrated that these anti-fIFN-γ MAbs recognised at least four different epitopes.
The combinations of MAbs used for sandwich ELISA and ELISpot assay were examined based on different epitopes recognised by the MAbs. Detectability was highest when Ame 6-31.1 and Ame 4-12.4 were used as a capture antibody and a detection antibody, respectively. Of the detection antibodies used for two-colour flow cytometry, Ame 4-12.4 showed the highest detectability. Thus, Ame 4-12.4 that recognises epitope A is an excellent detection antibody.
Recent studies in both mice and humans have demonstrated that both memory CD4+ and CD8+ T cells can rapidly produce IFN-γ following stimulation with cognate antigen. 34–37 Similar observations were also made in severe acute respiratory syndrome-associated coronavirus (SARS-CoV) infection, which belongs to Coronaviridae, like FIPV. SARS-CoV-specific memory CD4+ and CD8+ T cells from SARS-recovered donors rapidly produced IFN-γ following short-term stimulation with inactivated SARS-CoV or protein derived from SARS-CoV. 38–40 On the other hand, PBMCs derived from those who suffered from critical SARS or died of SARS apparently could not produce IFN-γ against SARS-CoV antigen stimulation. Furthermore, it has also been reported that CD8+ cytotoxic T lymphocytes (CTLs) are induced by SARS-CoV antigen stimulation in SARS-recovered donors. 41,42 It is suggested that memory CD4+ and CD8+ T cells participate in viral clearance in recovered SARS patients.
The results from sandwich ELISA and ELISpot assay in this study demonstrated that fIFN-γ production was specifically and significantly increased by heat-inactivated FIPV stimulation in PBMCs obtained from FIPV-infected cats without FIP. In addition, the results of two-colour flow cytometry demonstrated that the percentage of CD8+fIFN-γ + cells was markedly increased by heat-inactivated FIPV stimulation. Thus, stimulating PBMCs obtained from FIPV-infected cats without FIP with heat-inactivated FIPV increased fIFN-γ production particularly by FIPV-specific memory CD8+ T cells. fIFN-γ production in PBMC and in particular in FIPV-specific memory CD8+ T cells after infection of the animal, at a stage where the cat may or may not be infected and/or viraemic. It is suggested that FIPV-infected cats without FIP, but not FIP cats, generated potent cellular immunity against FIPV following infection with FIPV and that FIPV-infected cats without FIP generate a protective cellular immunity against FCoV. These results substantiate a previous report that cellular immunity plays an important role in preventing the development of FIP. 5,17–20 Detecting fIFN-γ employing sandwich ELISA, ELISpot assay, and two-colour flow cytometry using the anti-fIFN-γ MAbs created in this study appears to be useful in evaluating the cellular immunity of cats.
Recently, it was reported that the T helper (Th)1/T cytotoxic (Tc)1 epitopes were present in spike (S) and nucleocapsid (N) protein of SARS-CoV. 39–45 SSp-1, which is a 9-mer peptide, present in the C-terminal S2 domain of S protein of SARS-CoV, is known to induce strong CTL activity and is regarded as a candidate for vaccine development. 41,44,45 However, it is not yet known whether the Th1/Tc1 epitope are present in the S and/or N protein of FIPV. Hence, the identification of FIPV-specific Th1/Tc1 epitopes using anti-fIFN-γ MAbs may be very useful in FIPV vaccine development.
References
- 1. Hohdatsu T., Okada S., Koyama H. Characterization of monoclonal antibodies against feline infectious peritonitis virus type II and antigenic relationship between feline, porcine, and canine coronaviruses, Arch Virol 117, 1991, 85–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Motokawa K., Hohdatsu T., Aizawa C., Koyama H., Hashimoto H. Molecular cloning and sequence determination of the peplomer protein gene of feline infectious peritonitis virus type I, Arch Virol 140, 1995, 469–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Motokawa K., Hohdatsu T., Hashimoto H., Koyama H. Comparison of the amino acid sequence and phylogenetic analysis of the peplomer, integral membrane and nucleocapsid proteins of feline, canine and porcine coronaviruses, Microbiol Immunol 40, 1996, 425–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Pedersen N.C., Boyle J.F., Floyd K., Fudge A., Barker J. An enteric coronavirus infection of cats and its relationship to feline infectious peritonitis, Am J Vet Res 42, 1981, 368–377. [PubMed] [Google Scholar]
- 5. Pedersen N.C. A review of feline infectious peritonitis virus infection: 1963–2008, J Feline Med Surg 11, 2009, 225–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Pedersen N.C., Allen C.E., Lyons L.A. Pathogenesis of feline enteric coronavirus infection, J Feline Med Surg 10, 2008, 529–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Poland A.M., Vennema H., Foley J.E., Pedersen N.C. Two related strains of feline infectious peritonitis virus isolated from immunocompromised cats infected with a feline enteric coronavirus, J Clin Microbiol 34, 1996, 3180–3184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Vennema H., Poland A., Foley J., Pedersen N.C. Feline infectious peritonitis viruses arise by mutation from endemic feline enteric coronaviruses, Virology 243, 1998, 150–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kipar A., May H., Menger S., Weber M., Leukert W., Reinacher M. Morphologic features and development of granulomatous vasculitis in feline infectious peritonitis, Vet Pathol 42, 2005, 321–330. [DOI] [PubMed] [Google Scholar]
- 10. Barlough J.E., Stoddart C.A., Sorresso G.P., Jacobson R.H., Scott F.W. Experimental inoculation of cats with canine coronavirus and subsequent challenge with feline infectious peritonitis virus, Lab Anim Sci 34, 1984, 592–597. [PubMed] [Google Scholar]
- 11. Pedersen N.C., Black J.W. Attempted immunization of cats against feline infectious peritonitis, using avirulent live virus or sublethal amounts of virulent virus, Am J Vet Res 44, 1983, 229–234. [PubMed] [Google Scholar]
- 12. Pedersen N.C., Evermann J.F., Mckeirnan A.J., Ott R.L. Pathogenicity studies of feline coronavirus isolates 79-1146 and 79-1683, Am J Vet Res 45, 1984, 2580–2585. [PubMed] [Google Scholar]
- 13. Stoddart C.A., Barlough J.E., Baldwin C.A., Scott F.W. Attempted immunisation of cats against feline infectious peritonitis using canine coronavirus, Res Vet Sci 45, 1988, 383–388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Woods R.D., Pedersen N.C. Cross-protection studies between feline infectious peritonitis and porcine transmissible gastroenteritis viruses, Vet Microbiol 4, 1979, 11–16. [Google Scholar]
- 15. Pedersen N.C., Boyle J.F. Immunologic phenomena in the effusive form of feline infectious peritonitis, Am J Vet Res 41, 1980, 868–876. [PubMed] [Google Scholar]
- 16. Weiss R.C., Scott F.W. Antibody-mediated enhancement of disease in feline infectious peritonitis: comparisons with dengue hemorrhagic fever, Comp Immunol Microbiol Infect Dis 4, 1981, 175–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Gelain M.E., Meli M., Paltrinieri S. Whole blood cytokine profiles in cats infected by feline coronavirus and healthy non-FCoV infected specific pathogen-free cats, J Feline Med Surg 8, 2006, 389–399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kiss I., Poland A.M., Pedersen N.C. Disease outcome and cytokine responses in cats immunized with an avirulent feline infectious peritonitis virus (FIPV)-UCD1 and challenge-exposed with virulent FIPV-UCD8, J Feline Med Surg 6, 2004, 89–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Weiss R.C., Cox N.R. Delayed-type hypersensitivity skin response associated with feline infectious peritonitis in two cats, Res Vet Sci 44, 1988, 396–398. [PubMed] [Google Scholar]
- 20. Weiss R.C., Cox N.R. Evaluation of immunity to feline infectious peritonitis in cats with cutaneous viral-induced delayed hypersensitivity, Vet Immunol Immunopathol 21, 1989, 293–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Stoddart M.E., Gaskell R.M., Harbour D.A., Gaskell C.J. Virus shedding and immune responses in cats inoculated with cell culture-adapted feline infectious peritonitis virus, Vet Microbiol 16, 1988, 145–158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Bauer R.M., Olsen R.G. Parameters of production and partial characterization of feline interleukin 2, Vet Immunol Immunopathol 19, 1988, 173–183. [DOI] [PubMed] [Google Scholar]
- 23. de Antonio E. Mateu, Husmann R.J., Hansen R., et al. Quantitative detection of porcine interferon-gamma in response to mitogen, superantigen and recall viral antigen, Vet Immunol Immunopathol 61, 1998, 265–277. [DOI] [PubMed] [Google Scholar]
- 24. Surcel H.M., Troye-Blomberg M., Paulie S., et al. Th1/Th2 profiles in tuberculosis, based on the proliferation and cytokine response of blood lymphocytes to mycobacterial antigens, Immunology 81, 1994, 171–176. [PMC free article] [PubMed] [Google Scholar]
- 25. Jung T., Schauer U., Heusser C., Neumann C., Rieger C. Detection of intracellular cytokines by flow cytometry, J Immunol Methods 159, 1993, 197–207. [DOI] [PubMed] [Google Scholar]
- 26. Pitcher C.J., Quittner C., Peterson D.M., et al. HIV-1-specific CD4+ T cells are detectable in most individuals with active HIV-1 infection, but decline with prolonged viral suppression, Nat Med 5, 1999, 518–525. [DOI] [PubMed] [Google Scholar]
- 27. Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity, Nature 256, 1975, 495–497. [DOI] [PubMed] [Google Scholar]
- 28. Takano T., Kawakami C., Yamada S., Satoh R., Hohdatsu T. Antibody-dependent enhancement occurs upon re-infection with the identical serotype virus in feline infectious peritonitis virus infection, J Vet Med Sci 70, 2008, 1315–1321. [DOI] [PubMed] [Google Scholar]
- 29. Takano T., Katada Y., Moritoh S., et al. Analysis of the mechanism of antibody-dependent enhancement of feline infectious peritonitis virus infection: aminopeptidase N is not important and a process of acidification of the endosome is necessary, J Gen Virol 89, 2008, 1025–1029. [DOI] [PubMed] [Google Scholar]
- 30. Masuda K., Sakaguchi M., Saito S., et al. In vivo and in vitro tests showing sensitization to Japanese cedar (Cryptomeria japonica) pollen allergen in atopic dogs, J Vet Med Sci 62, 2000, 995–1000. [DOI] [PubMed] [Google Scholar]
- 31. Kemp K., Theander T.G., Hviid L., Garfar A., Kharazmi A., Kemp M. Interferon-gamma- and tumour necrosis factor-alpha-producing cells in humans who are immune to cutaneous leishmaniasis, Scand J Immunol 49, 1999, 655–659. [DOI] [PubMed] [Google Scholar]
- 32. Pathan A.A., Wilkinson K.A., Klenerman P., et al. Direct ex vivo analysis of antigen-specific IFN-gamma-secreting CD4 T cells in Mycobacterium tuberculosis-infected individuals: associations with clinical disease state and effect of treatment, J Immunol 167, 2001, 5217–5225. [DOI] [PubMed] [Google Scholar]
- 33. Shankar P., Russo M., Harnisch B., Patterson M., Skolnik P., Lieberman J. Impaired function of circulating HIV-specific CD8+ T cells in chronic human immunodeficiency virus infection, Blood 96, 2000, 3094–3101. [PubMed] [Google Scholar]
- 34. Harari A., Vallelian F., Meylan P.R., Pantaleo G. Functional heterogeneity of memory CD4 T cell responses in different conditions of antigen exposure and persistence, J Immunol 174, 2005, 1037–1045. [DOI] [PubMed] [Google Scholar]
- 35. van Lier R.A., ten Berge I.J., Gamadia L.E. Human CD8+ T-cell differentiation in response to viruses, Nat Rev Immunol 3, 2003, 931–939. [DOI] [PubMed] [Google Scholar]
- 36. Wherry E.J., Ahmed R. Memory CD8 T-cell differentiation during viral infection, J Virol 78, 2004, 5535–5545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Wills M.R., Okecha G., Weekes M.P., Gandhi M.K., Sissons P.J., Carmichael A.J. Identification of naive or antigen-experienced human CD8+ T cells by expression of costimulation and chemokine receptors: analysis of the human cytomegalovirus-specific CD8+ T cell response, J Immunol 168, 2002, 5455–5464. [DOI] [PubMed] [Google Scholar]
- 38. Peng H., Yang L.T., Li J., et al. Human memory T cell responses to SARS-CoV E protein, Microbes Infect 8, 2006, 2424–2431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Peng H., Yang L.T., Wang L.Y., et al. Long-lived memory T lymphocyte responses against SARS coronavirus nucleocapsid protein in SARS-recovered patients, Virology 351, 2006, 466–475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Yang L.T., Peng H., Zhu Z.L., et al. Long-lived effector/central memory T-cell responses to severe acute respiratory syndrome coronavirus (SARS-CoV) S antigen in recovered SARS patients, Clin Immunol 120, 2006, 171–178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Chen H., Hou J., Jiang X., et al. Response of memory CD8+ T cells to severe acute respiratory syndrome (SARS) coronavirus in recovered SARS patients and healthy individuals, J Immunol 175, 2005, 591–598. [DOI] [PubMed] [Google Scholar]
- 42. Zhou M., Xu D., Li X., et al. Screening and identification of severe acute respiratory syndrome-associated coronavirus-specific CTL epitopes, J Immunol 177, 2006, 2138–2145. [DOI] [PubMed] [Google Scholar]
- 43. Tsao Y.P., Lin J.Y., Jan J.T., et al. HLA-A∗0201 T-cell epitopes in severe acute respiratory syndrome (SARS) coronavirus nucleocapsid and spike proteins, Biochem Biophys Res Commun 344, 2006, 63–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Wang B., Chen H., Jiang X., et al. Identification of an HLA-A∗0201-restricted CD8+ T-cell epitope SSp-1 of SARS-CoV spike protein, Blood 104, 2004, 200–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Wang Y.D., Sin W.Y., Xu G.B., et al. T-cell epitopes in severe acute respiratory syndrome (SARS) coronavirus spike protein elicit a specific T-cell immune response in patients who recover from SARS, J Virol 78, 2004, 5612–5618. [DOI] [PMC free article] [PubMed] [Google Scholar]