Abstract
Practical relevance Lower respiratory tract infections (LRTIs) in cats can be due to bacteria, parasites, fungi and viruses. This review details the practical investigation of these infections and highlights specific therapy where possible. The aim is to avoid the all-too-frequent temptation in practice to treat cats with lower respiratory tract signs empirically for feline bronchial disease (FBD)/asthma. This is potentially hazardous as immunosuppressive therapy for FBD/asthma could exacerbate disease due to a LRTI. Empirical treatment of suspected LRTI is also difficult to recommend given the wide range of potential pathogens.
Clinical challenges Making a clinical ante-mortem diagnosis of LRTI in a cat can be challenging. Consistent historical, clinical, haematological and radiographic abnormalities are often lacking and findings may be non-specific. Astute clinical acumen, thorough investigation and high quality laboratory analysis are usually required for a diagnosis. Bronchoalveolar lavage, if feasible, and tests for lungworm should be routine in cats with lower respiratory tract signs. Lung fine needle aspiration may be useful in cases of diffuse or nodular pulmonary disease. Histopathology is rarely employed in ante-mortem investigations.
Evidence base The authors have reviewed a substantial body of literature to provide information on many of the reported bacterial, parasitic, fungal and viral pathogens, including some that occur in Asia. Attention has been given to specific therapy for each pathogen, with evidence-based comments when there is a deviation from routine recommendations.
Prerequisites for a clinical diagnosis of feline LRTI
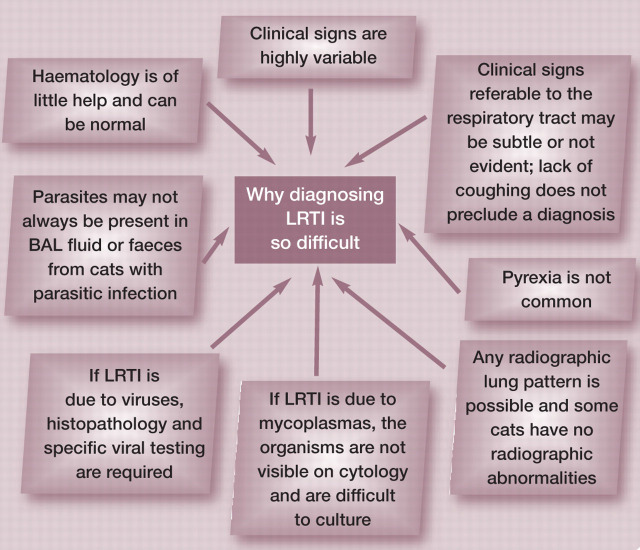
Bacteria, parasites, fungi and viruses cause lower respiratory tract infections (LRTIs) in cats. Most cats with LRTIs have pneumonia (inflammation of the lung parenchyma), although pathology is occasionally limited to the airways. 1,2 Viral causes of LRTIs are unlikely to be diagnosed without lung histopathology and specific viral detection techniques. Parasitic, bacterial and fungal LRTIs can be diagnosed by routine investigation of lower respiratory tract disease. However, obtaining a clinical diagnosis of LRTI can be challenging. Historical, clinical, haematologic and radiographic findings known to be compatible with LRTI are often non-specific or are inconsistently present. 1,3,4 In addition, some cases of LRTI may have concomitant, predisposing respiratory tract or systemic diseases.
Clinical ante-mortem diagnosis of LRTI relies on analysis of all the patient details: signalment, history, physical examination findings, radiography, cytological and microbiological assessment of broncho-alveolar lavage (BAL) fluid or pulmonary fine needle aspirates (FNAs), histopathology (if available) and response to appropriate therapy. As histopathology is rarely available, BAL (and, to a lesser extent, FNA) cytology and microbiology are the mainstays of ante-mortem investigation of LRTI. However, the cytological and microbiological analyses by the laboratory have to be of an excellent standard for these to be of high diagnostic yield. In addition, for the laboratory to achieve optimal results, high quality samples and appropriate requests from clinicians are essential. Only the veterinarian handling the case can make a diagnosis of LRTI.
Key studies to date on feline LRTIs
There have been three detailed retrospective studies on feline LRTIs: one from Switzerland by Bart et al (2000; 245 cats), 2 one from the USA by Macdonald et al (2003; 39 cats) 1 and one from Australia by Foster et al (2004; 21 cats). 3 The American 1 and Swiss 2 studies both analysed LRTIs that were histologically confirmed at necropsy. Of the two, only the American study assessed clinical findings in addition to postmortem findings. The rigid selection criteria in the American study ensured accuracy of diagnosis but, by its postmortem nature, precluded analysis of nonfatal cases. The cases in the Australian study 3 were identified by airway and/or pulmonary cytology and microbiology, in conjunction with clinical assessment, including response to appropriate therapy; histopathological confirmation was only available in a few cases as treatment was effective in most cats.
Only the veterinarian handling the case can make a diagnosis of LRTI.
In addition to these studies, there have been numerous case reports and case series describing specific feline LRTIs.
What do the studies tell us about signalment and clinical signs?
Signalment
No breed predilection has been noted for LRTIs in cats. 1,3 Foster et al found males were 2.4 times more likely to have LRTIs than females. 3 Interestingly, in a recent retrospective study on fatal feline herpesvirus type 1 (FHV-1)-associated pneumonia, all the cases were male. 5
Clinical signs
The most common presenting complaint in the Australian study was coughing (76%), and the most common abnormalities detected during physical examination were dyspnoea and/or tachypnoea (67%), and coughing or increased tracheal hypersensitivity (38%). 3
Of the cats diagnosed at necropsy in the American study, 1 only 8% coughed, indicating that while coughing cats should be investigated for LRTI, lack of coughing does not preclude a differential diagnosis of LRTI. In that same study, 36% of cats lacked any clinical signs referable to the respiratory tract and 41% of cats lacked clinical signs of systemic illness. The most common respiratory tract signs were tachypnoea or dyspnoea (49%) and nasal discharge (21%). Pyrexia was noted in 15% of the cats in the American study 1 and 24% of cats in the Australian study; 3 none of the cats that presented alive with LRTI due to Bordetella bronchiseptica were pyrexic in a study on B bronchiseptica infections. 6 Thus, pyrexia would appear to be a relatively uncommon finding in LRTIs and normothermia does not rule out LRTI.
Despite differences in selection criteria and their impact on clinical presentation, the studies indicate that cats with LRTIs may not have any signs referable to the respiratory tract and that lack of coughing does not preclude a diagnosis. Cats that do have coughing, dyspnoea or tachypnoea should be investigated for LRTI. Careful physical examination and astute clinical acumen are likely to be required if a diagnosis of LRTI is to be made in an affected cat.
Pyrexia appears to be a relatively uncommon finding in LRTIs.
Sudden death in cats and kittens.
Necropsy in a number of cases of sudden death has revealed pneumonia. Pneumonia should be on the differential diagnosis list for sudden death in cats.
Cats with LRTIs may not have any signs referable to the respiratory tract and lack of coughing does not preclude a diagnosis.
How do the studies help direct laboratory investigation?
Haematology
There has been limited investigation of changes to the erythrogram of cats with LRTIs. Foster et al reported that packed cell voume was normal in 8/12 cats and slightly decreased in 4/12 cats. 3 Leukocytosis and neutrophilia are the most common haematological abnormalities. 1,3 Leukocytosis and neutrophilia were detected in 8/10 and 9/10 cases, respectively, in the Australian study 3 and 9/18 and 7/18 of cases, respectively, in the American study. 1 Leukopenia, neutropenia and left shifts were uncommon. 1,3 It is noteworthy that 4/18 cats in the American study had leukocyte counts within the reference intervals, despite the fatal disease outcomes. Eosinophilia was present in 4/10 cats in the Australian study and only one of these had a parasitic LRTI. Other parasitic LRTIs in this study were not accompanied by an eosinophilia. 3 As such, haematology is often of little help in the diagnosis of LRTIs in cats.
Biochemistry
Serum biochemical analysis in clinical cases has been limited but serum biochemistry is unlikely to be of help in the diagnosis of LRTI. Cats with LRTIs may have increased globulin and total protein concentrations. 3
Radiography
In the Australian study, 3 all lung patterns were represented radiographically and, while 67% of cases had alveolar changes, 81% of cases had a bronchial pattern either alone or in combination with another pattern. A predominantly nodular pattern was noted in the cases of cryptococcosis, toxoplasmosis and mycoplasmal abscess. A range of radiographic patterns was also detected in the American study, and, in 3/13 (23%) cases, there were no radiographic abnormalities. 1 Lack of radiographic abnormalities was also reported in a cat with confirmed cryptococcal LRTI. 7
Non-bronchoscopic BAL.
Anaesthetise the cat with a short-acting anaesthetic agent.
Place an intravenous catheter for intravenous access, if one has not already been placed for the anaesthetic.
Pass a sterile endotracheal tube carefully into the trachea, using a laryngoscope to facilitate its passage through the larynx and minimise oropharyngeal contamination.
Position the cat in lateral recumbency, most affected side down, to increase the chance of retrieving representative samples.
Pass a sterile dog urinary catheter (6–8 French gauge) gently through the endotracheal tube until it wedges in a distal airway and then flush two 5–10 ml aliquots of warm sterile saline through the catheter.
Retrieve the lavage fluid from the catheter by syringe aspiration and perform cytology and microbiology on that fluid.
Collect any fluid that is coughed through the sterile endotracheal tube during anaesthetic recovery into a sterile container to ensure adequates sample volume for assessment (even though this is not normally processed).
Have terbutaline ‘on hand’ in case of acute bronchoconstriction (with dose [0.01 mg/kg IV, IM or SC] pre-calculated). It is preferable to maintain an intravenous catheter until full recovery to enable IV drug administration if necessary.
Lack of consistent radiographic abnormalities contributes to the difficulties in diagnosing LRTIs in cats.
Bronchoalveolar lavage
The gold standard procedure for airway sampling is BAL via bronchoscopy. Collection of airway samples by this method is detailed in numerous texts and papers. 8,9 Bronchoscopic BAL in cats with respiratory disease has a low rate of serious complications and it is possible that the overall complication rate can be reduced further by premedication with terbutaline. 9
Bronchoscopic alveolar lavage is not always possible due to lack of availability of equipment or financial or time constraints. Other simple and cheaper non-bronchoscopic methods have been described in the literature and these can be readily used in general practice. 8,10–14 The non-bronchoscopic BAL procedure used by one of the authors (SF) is outlined in the box above.
Transtracheal aspiration
Transtracheal aspiration (TTA) is rarely used in cats as, firstly, patient restraint can exacerbate respiratory signs and, secondly, airway control and ventilation in response to any bronchoconstriction or exacerbation of respiratory signs are easier under anaesthesia. However, if anaesthesia is contraindicated, then this technique can be used instead of BAL. Care must be taken that patient restraint does not result in exacerbation of respiratory signs. Local anaesthetic must be placed prior to introduction of the catheter through the cricothyroid ligament. An 18–22 G through-the-needle intravenous catheter is optimal (although rarely available in general practice) as the needle can be withdrawn after catheter placement to minimise tracheal injury. 12,15 Approximately 0.5 ml/kg of warmed sterile saline is instilled into the catheter 12 and then immediately aspirated to retrieve the lavage fluid. 12 A second flush may be required for adequate sampling. 12,15
Lung fine needle aspiration
Lung fine needle aspiration may be indicated to investigate lesions identified by thoracic radiography or ultrasonography. The specific approach and procedure chosen depend on the type of lesion (mass lesion versus diffuse disease), lesion location (central versus peripheral) and the clinical status of the patient. Aspiration can be performed either blind (for diffuse disease or discrete peripheral focal lesions) or guided by ultrasound, fluoroscopy or computed tomography (CT).
Lung fine needle aspiration might perhaps be expected to entail a higher risk than BAL. However, a number of studies in dogs and cats using 22–27 G hypodermic or spinal needles for lung or intrathoracic fine needle aspiration have demonstrated no clinically evident complications. 16–18 Interestingly, when CT monitoring was used after fine needle aspiration or core biopsy with 18–22 G diameter instruments, 43% of cases had pneumothorax, pulmonary haemorrhage or both, despite the lack of clinical manifestations. Not surprisingly, these complications were common when aerated lung was perforated. 19
The diagnostic challenge of feline LRTIs.
Cytology
Cytology can be performed on BAL (or TTA) fluid or lung fine needle aspirates (FNAs). Lung FNA cytology is performed as per any tissue aspirate cytology. In contrast, BAL/TTA fluid analysis has not been standardised. 10 Many studies and most commercial laboratories report total cell counts (which can be influenced by the amount of lavage fluid delivered and retrieved) and use cytospin preparations for cytology; cytospin preparations have been shown to be of better quality than manual smearing of pelleted cells after centrifugation, especially when there is a low total cell count. 20 When performing total cell counts and preparing cytocentrifuged smears, mucus is routinely excluded from the analytic material. However, many of the cells in the BAL fluid are trapped within the mucus strands and not performing direct smears of mucus can result in failure to detect Aelurostrongylus abstrusus. 21
When BAL fluid is collected as described earlier, the practitioner can prepare smears inhouse and perform cytology (see box below).
Reporting of differential white cell counts from airway samples is also not standardised. In two feline studies with similar smear preparation, BAL cytology was classified by the predominant inflammatory cell type, if greater than or equal to 50% of the total, or described as mixed, if no cell type predominated. 10,22
In-house BAL cytology.
Place two drops of lavage fluid containing mucus on a microscope slide.
Examine the unstained wet preparation microscopically for the presence of parasites.
Prepare multiple air-dried squash preparations of mucus from each lavage specimen and stain with Diff Quik.
Check for the presence of mucus, oropharyngeal squames, oropharyngeal bacteria, respiratory pathogens, epithelial cells and inflammatory cells.
-
Total cell counts are not performed as reference intervals have not been validated for this method of sample collection.
Choose some representative fields and count the different types of white cells to provide a differential white cell count for the smear.
Classify inflammation by the predominant cell type (if ≥50%) or describe the inflammation as ‘mixed’ if no cell type predominates.
If sufficient material is present, examine several smears from each sample.
If no mucus is present, a centrifugation technique is required for smear preparation as mucus-free samples may contain abnormal cells. 14
If bacteria are identified, Gram staining should be performed; acid-fast stains may also be necessary for some bacteria.
Airways in healthy cats are not sterile and this needs to be taken into account when interpreting airway cytology and microbiology.
In both studies epithelial cells were not considered in the calculation of relative cell counts, which is consistent with the recommendations of the American Thoracic Society for reporting cell counts in humans. 23
In cats with LRTIs, cytology performed by the method detailed 10 revealed neutrophilia in all but one. 3 The normal lower airway response to pulmonary bacterial or protozoal infection would appear to be neutrophilic unless there is concurrent immunosuppression. None of the cats with confirmed parasitic infections had an eosinophilic BAL, although other factors may have influenced this — the presence of concurrent salmonellosis in two and feline immunodeficiency virus (FIV) infection in one. 3
Bacteria consistent with the culture results should be observed cytologically in bacterial LRTIs, except in cases of mycoplasmosis; mycobacteria may also be difficult to find cytologically. 24 Mycoplasmas do not usually take up Diff Quik or Gram stain, although small Gram-negative stained ‘flecks’ have occasionally been observed in cytological preparations of BAL fluid from which mycoplasmas were cultured (P Martin, unpublished data).
One small study of feline respiratory cases (only 1/11 with histologically recognised LRTI) demonstrated that BAL fluid cytopathology performed on cytocentrifuged samples does not always correlate with the type of pulmonary disease identified on histopathology. 25 However, histopathology correlated reasonably well with cytology in another study in which a number of cats had inflammatory lower respiratory tract lesions. 26 While one cat with feline infectious peritonitis in this study had normal BAL cytology, which was not representative, cytology in three other cats (one with toxoplasmosis, two with focal bronchitis) was more sensitive than histopathology. 26 The authors concluded that cytological specimens appear more sensitive for the detection of microscopic focal lesions and Toxoplasma gondii tachyzoites, but that lesions confined to the interstitium may not be detected in bronchial washings. 26
Microbiology
Airways in healthy cats are not sterile and this needs to be taken into account when interpreting airway cytology and microbiology.
The bacteria reported as occurring in low numbers (< 2 × 10 3 colony forming units/ml) in the airways of healthy cats include Escherichia coli and species of Pasteurella, Pseudomonas, Staphylococcus, Streptococcus and Micrococcus. Anaerobic bacteria and mycoplasmas have not been isolated from the lower airways of healthy cats. 27,28
The most common bacteria causing feline LRTIs in the three retrospective studies of LRTIs were B bronchiseptica, Pasteurella species, Mycoplasma species, Streptococcus species and E coli. Other agents identified as causing infectious pneumonia in these studies were other bacteria (including mycobacteria, Salmonella typhimurium, Pseudomonas species, anaerobes), viruses (herpesvirus, coronavirus), lungworm (A abstrusus, Eucoleus aerophilus), protozoa (T gondii) and fungi (Cryptococcus species, Candida albicans, Mucor species, Aspergillus species). 1–3 Various other aetiological agents have also been identified in case reports and small case series, and described in textbooks.
Parasitology
Faecal flotation and faecal analysis by the Baermann technique should be routinely performed in cats with possible LRTIs. Other diagnostic tests for lungworm detection are also available (see later).
Histopathology
Lung histopathology is occasionally performed ante-mortem. Samples can be obtained via ultrasound- or CT-guided core biopsy or surgical excision (thoracoscopy or thoracotomy).
Bacterial LRTIs
Mycoplasmas
Mycoplasmas are the smallest free-living, self-replicating microorganisms and have no cell wall. 29 They are represented in the natural mucosal flora of cats but are not normally present in the lower airways. 27,28 Both the Swiss 2 and Australian 3 LRTI studies demonstrated the importance of mycoplasmas in feline LRTIs. Mycoplasmas are known pulmonary pathogens in other species and have been recorded as causing pyothorax, pneumonia (Fig 1), LRTIs and pulmonary abscessation in cats. 2,3,30–40 Mycoplasma felis has also been shown to directly induce pneumonia in two healthy kittens after experimental inoculation. 41
FIG 1.
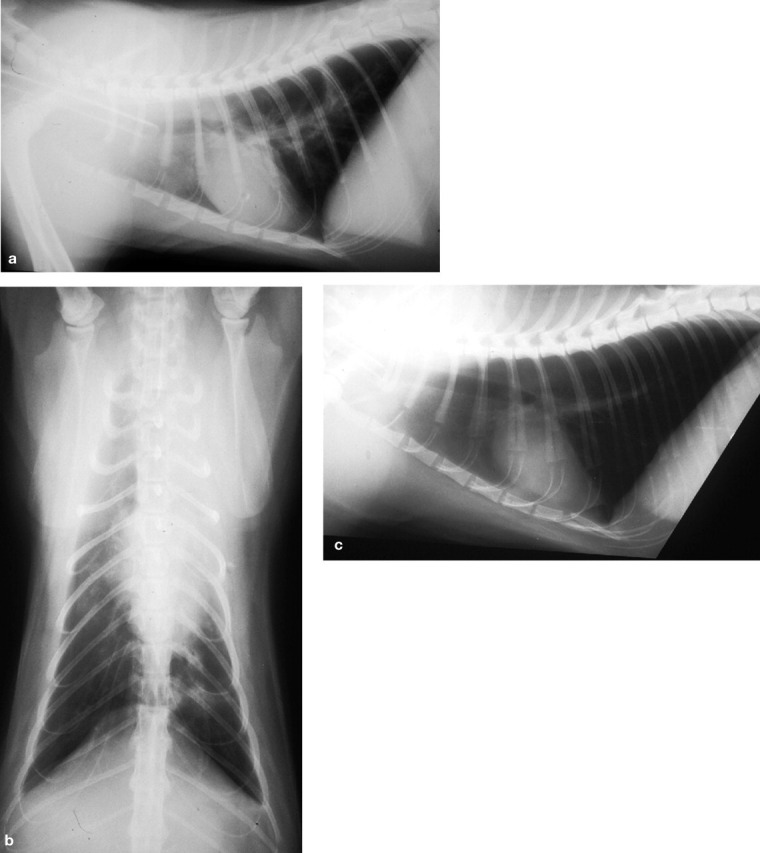
Radiographs of a cat with a mycoplasmal LRTI. (a) Right lateral thoracic radiograph showing consolidation in the cranial lung lobe area. (b) Ventrodorsal thoracic radiograph showing consolidation of the left cranial lung lobe. (c) Right lateral thoracic radiograph after 6 weeks of therapy. Note the improved aeration of the cranial lung lobe. Reprinted from Foster et al (1998), 36 with permission of John Wiley and Sons, publisher of the Australian Veterinary Journal
Despite this, mycoplasmal LRTIs are often considered to be a consequence of pre-existing pulmonary diseases such as FBD. It is possible that feline mycoplasmas may be aetiological agents rather than secondary in FBD, causing serious pathology and resulting in ongoing inflammatory bronchial disease and airway hyperresponsiveness similar to Mycoplasma pneumoniae in humans. 3,37,42–44
It is possible that feline mycoplasmas may be aetiological agents rather than secondary in feline bronchial disease, causing serious pathology and resulting in ongoing inflammatory bronchial disease and airway hyperresponsiveness, similar to Mycoplasma pneumoniae in humans.
Role of mycoplasmas in asthma.
In a review of M pneumoniae in humans, published in 2008, it was stated that ‘progress in understanding the … true role as a human pathogen has been hindered significantly over the years by its very slow replication rate, fastidious demands for laboratory cultivation and relatively low sensitivity and specificity of the earliest complement fixation serological tests … and this has meant that laboratory confirmation of mycoplasmal infection was seldom sought. A frequent but incorrect assumption was that mycoplasmal respiratory infection was uncommon, rarely significant from a clinical standpoint and limited to select age groups.' 43
Much of this could potentially be true of feline mycoplasmal LRTIs also.
Potential roles of mycoplasmas in feline FBD/asthma have yet to be investigated. It is worth considering that a role for M pneumoniae in the pathogenesis of human asthma was suggested more than 30 years ago and that support for this theory is now strong. There is increasing evidence from controlled studies to support a strong association between infection with M pneumoniae and both chronic stable asthma and acute exacerbations of asthma. 45 The role of M pneumoniae as an initiating cause of asthma is also much discussed, but less clear. 42–49
Interestingly, mice with allergic sensitisation of the lungs have Toll-like receptor 2 down-regulation, delaying M pneumoniae clearance. 50 Humans with allergic sensitisation of the lungs may also be at a greater risk of developing chronic airway colonisation by M pneumoniae, with resultant augmentation of airway hyperreactivity, and these patients may have no measurable humoral immune response. 43 Thus, there appears to be a complex relationship between mycoplasmal infection and human asthma.
Diagnosis
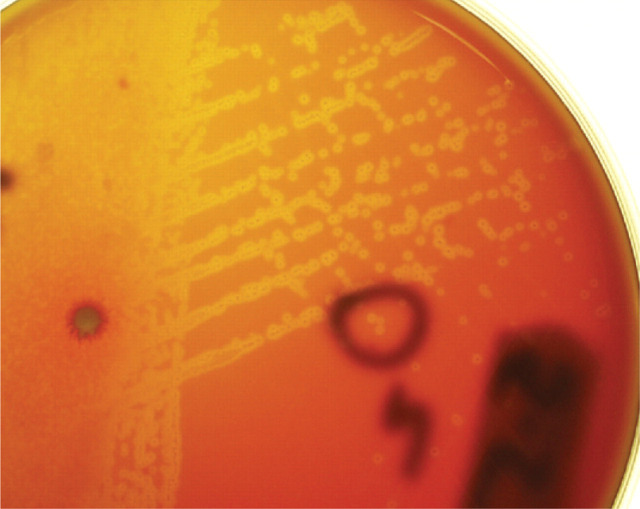
Mycoplasmas do not take up Diff Quik or Gram stain, a finding usually attributed to their lack of a cell wall. 29 Culture of mycoplasmas is difficult as they may not survive transport to a laboratory and usually require specific culture media. Diagnosis of mycoplasmosis in the Australian LRTI study was only possible because there was no transport delay and mycoplasmas were cultured from the peptone-enriched blood agar plates used (Fig 2). 3 Specific polymerase chain reaction (PCR) testing is likely to improve the sensitivity of diagnosis, as it has in human medicine. However, in practice, mycoplasmal LRTI will remain a difficult diagnosis as neither mycoplasmal culture nor mycoplasmal PCR testing is performed routinely. Clinicians need to make specific provisions (such as specialised transport swabs) and laboratory requests when investigating possible mycoplasmal LRTI. Treatment trials are often a more attractive alternative.
FIG 2.
Heavy growth of mycoplasma isolated aerobically on sheep blood agar from a feline BAL sample after 3–4 days of incubation. Note the small lucent colonies buried in the agar and surrounded by zones of haemolysis
Treatment
Treatment of mycoplasmal infections in veterinary medicine is usually empirical. Antibiotic susceptibility profiles are not available for feline mycoplasmal isolates. Mycoplasmas are generally reported to be sensitive to macrolides (erythromycin, clarithromycin, tylosin), fluoroquinolones (enrofloxacin, ciprofloxacin, pradofloxacin), tetracyclines, chloramphenicol and gentamicin. Enrofloxacin may not be as effective as doxycycline, 3 but pradofloxacin has demonstrated similar efficacy to doxycycline for upper respiratory tract (URT) infections caused by mycoplasmas. 51
Doxycycline would appear to be an appropriate and affordable initial choice of antibiotic for empirical therapy of feline LRTIs. The monohydrate preparation of doxycycline available in Australia (VibraVet; Pfizer) has not been reported to cause the oesophagitis and oesophageal strictures that have been noted in other countries with hydrochloride preparations. If using doxycycline, however, it is recommended that each dose be followed by a water or food swallow. 52
KEY POINTS: MYCOPLASMAS.
Mycoplasmas do not take up Diff Quik or Gram stain and do not respond to β-lactam antibiotics.
Specific transport swabs and culture media are required for diagnosis if PCR is not available.
Mycoplasmal infections result in neutrophilic BAL cytology with no demonstrable bacteria.
Treatment trials with doxycycline or fluoroquinolones may be required if clinical evidence is suggestive of mycoplasmosis and specific testing is not available.
Mycoplasmas are one of the ‘hot topics’ in human asthma medicine. Their role in feline asthma has yet to be explored.
It is probably worth treating all cats with lower respiratory tract disease with an appropriate antibiotic for mycoplasmas while awaiting results of cytology and culture.
It is probably worth treating all cats with lower respiratory tract disease with an appropriate antibiotic for mycoplasmas while awaiting results of cytology and culture. Additionally, if FBD/asthma is suspected and the cat is stable enough to allow trial antibiotic treatment, then this should also be considered prior to immunosuppressive or anti-inflammatory medication. It does need to be noted, however, that some antimicrobials have beneficial effects on airway inflammation unrelated to antimycoplasmal activity. 44
The duration of treatment required is not known. Mycoplasmal species that infect animals and humans are able to induce chronic disease states in which clearance of the organism is extremely difficult. Intracellular localisation, immunomodulatory effects and surface antigen variations may all contribute to this process. 43 Prior allergic sensitisation of the lungs could potentially also result in delayed organism clearance, as it does with M pneumoniae in experimental murine models. 50 Treatment of mycoplasmal URT disease in cats for 42 days has been recommended, 51 as PCR-positive results after 28 days have been demonstrated. Given that chronic persistent infection has been documented in humans, that some cases of mycoplasmal LRTI have been reported to have recurrent doxycycline-responsive coughing or need continuous treatment, 3 and the finding that 42 days may be required for treating feline URT mycoplasmal infections, 51 it may be prudent to treat mycoplasmal LRTIs with doxycycline (5 mg/kg PO q12h) for a minimum of 6 weeks.
Bordetella bronchiseptica
Bordetella bronchiseptica is a primary respiratory pathogen in cats. Naturally occurring B bronchiseptica infection may cause URT signs (sneezing, oculonasal discharge) or LRT signs (coughing, severe dypsnoea, cyanosis and even death due to bronchopneumonia). 53
Young kittens appear to be most susceptible to disease due to B bronchiseptica and fatal bronchopneumonia has been reported in kittens. 6,53,54 Lower respiratory tract infection due to B bronchiseptica has also been reported in adult cats 6,55,56 and fatal bronchopneumonia was reported in 10 cats, which were presumably adults as they were recorded as ‘cats’ and not ‘kittens'. 57 Bronchopneumonia, which was unresponsive to appropriate and intensive therapy, also occurred recently in an another adult cat (D Foster, unpublished data; Fig 3).
FIG 3.
Diff Quik-stained smear of BAL fluid from a 3.5-year-old castrated male domestic shorthair cat with a fibrinonecrotic pneumonia due to B bronchiseptica. Numerous fine rods can be seen attached to the cilia of the respiratory epithelial cells. Despite appropriate and aggressive parenteral and nebulised antibiotic therapy, this cat failed to respond to treatment. Case details courtesy of Dr Darren Foster
Virology studies have not always been performed to assess concurrent viral respiratory tract infection in field studies. Concurrent feline calicivirus (FCV) infection was demonstrated in some cases 6,58 and concurrent feline herpesvirus (FHV) in one. 1 Concurrent feline panleukopenia was associated in another report. 55 Regardless, B bronchiseptica appeared to be a significant contributing cause to bronchopneumonia and death in these cats. 1,6,55,58
Antibiotic treatment early in the course of the disease is recommended. 6 Antibiotic susceptibility for this organism indicates that it is usually resistant to penicillin, cephalosporins and ampicillin, and sensitive to a wide range of antibiotics including tetracyclines, enrofloxacin, amoxicillin—clavulanate, chloramphenicol and gentamicin. 6,59 Although most antimicrobials are able to attain concentrations in the lung parenchyma comparable to drug concentrations in the serum, concentrations achieved in the airways and bronchial secretions may be significantly lower. 60,61 This is particularly relevant to the treatment of LRTIs due to B bronchiseptica, as this bacterium attaches to the cilia on the surface of the tracheobronchial epithelium (Fig 3). 60 The β-lactam drugs, in particular, have low distribution into respiratory secretions and so cannot be recommended for the treatment of B bronchiseptica. 60 Trimethoprim-sulphonamide was used successfully in four cats with bronchopneumonia 6 but a high level of resistance to trimethoprim-sulphonamide was detected in one study. 62 Enrofloxacin was used unsuccessfully in one cat, which then responded to trimethoprim-sulphonamide, 56 and successfully in another. 3
Thus, the current recommended drugs for the treatment of bronchopneumonia due to B bronchiseptica are doxycycline, trimethoprimsulphonamide and fluoroquinolones. 53,61
Streptococcus species
Beta-haemolytic streptococci are commensal microflora of the skin, pharynx, URT and genital tract of cats. 63 Infections with Lancefield group G streptococci in kittens and occasionally in adult cats are known to result in pneumonia associated with pleuritis and cervical lymphadenitis, 64,65 fasciitis and myositis 66,67 and toxic shocklike syndrome. 68 In the American retrospective LRTI study, 1 β-haemolytic Streptococcus species were the most common bacterial cause of LRTIs (29% of cases). Streptococcus species were also a major cause of deaths in the Swiss retrospective LRTI study, 2 the majority of which were in kittens less than 12 weeks old. By contrast, Streptococcus species were not identified as a cause of LRTI in the predominantly ante-mortem, adult cat study from Australia, 3 so it is possible that pneumonia due to Streptococcus species may be severe and rapidly fatal and/or occur predominantly in kittens.
The majority of β-haemolytic streptococcal infections in cats are caused by Lancefield group G streptococci, usually S canis. 63 However, recently, an outbreak of respiratory disease due to S equi (Lancefield group C) in a large shelter caused approximately 10% mortality. 69 Clinical disease in these cats was characterised by copious purulent nasal discharge and coughing, progressing to sinusitis, dyspnoea, signs of pneumonia and death. Streptococcus pneumoniae (Lancefield group A) was reported to cause a moderate interstitial pneumonia in a kitten that died with fasciitis and myositis. 67 Streptococcus suis has also been isolated from pneumonic lungs in a 2-week-old kitten and a 2-year-old cat. 70
Lancefield group G streptococci of cats have been uniformly sensitive to penicillin, which has been regarded as the drug of choice. 64 Procaine and benzathine penicillin (long-acting penicillin) have been used both in the treatment of cases and prophylactically in cats. 63,64,71 (Penicillin doses are usually provided in terms of units. The mg/kg conversions for the various preparations of penicillin G [eg, penicillin G potassium and sodium, penicillin G procaine, penicillin G benzathine] are provided in many [if not all] editions of Plumb's Veterinary Drug Handbook.) 72
In humans, clindamycin is regarded as the drug of choice in necrotising fasciitis or toxic shock streptococcal syndromes. Clindamycin is also reported to be valuable in the treatment of streptococcal infections in cats. 63 Fluoroquinolones are not recommended for treating streptococcal infections.
LRTIs due to E coli, while relatively uncommon, appear to be associated with high mortality, especially in kittens.
Escherichia coli
Extraintestinal pathogenic E coli (ExPEC) has been reported to cause respiratory disease outbreaks in cats. Thirteen cats from an animal shelter developed acute respiratory disease and died from acute necrotising pneumonia in a week in one such outbreak. 73 Fatal pneumonia occurred in three kittens in an outbreak in another shelter. 74 In addition to the feline impact, such outbreaks also have zoonotic potential. 73,74
Comparing the three retrospective LRTI studies, E coli was isolated in 2/39 (5%) total cases (2/21 bacterial cases) in the American study 1 and 21/245 (9%) cases in the Swiss study, 2 of which 16 were septicaemic and most occurred in kittens less than 12 weeks old. Escherichia coli was not identified in the Australian study of predominantly ante-mortem cases, 3 although a fatal case of pneumonia due to E coli and A abstrusus was diagnosed in a kitten subsequent to the study (Fig 4). Lower respiratory tract infections due to E coli, while relatively uncommon, would thus appear to be associated with high mortality, especially in kittens.
FIG 4.
Lungs from a 12–16-week-old male domestic shorthair cat. Histopathology demonstrated extensive granulomatous and eosinophilic pneumonia due to E coli and A abstrusus. Photograph and case details courtesy of Dr Katrina Bosward
Pasteurella species
Pasteurella species are part of the microflora of the nasopharynx and airways of healthy cats. 27,75 Not surprisingly, these bacteria are one of the more common species to be isolated from cats with LRTIs. 1–3 It is likely that concurrent viral infections and other stresses leading to impaired defence mechanisms result in pneumonia due to Pasteurella species. 75 These bacteria are usually susceptible to the aminopenicillanic derivatives. 75
Salmonella species
Pneumonia due to Salmonella species can occur in association with systemic salmonellosis or as a localised LRTI. 3,76 Concurrent infection with A abstrusus has been reported in two cases. 3,77 While LRTIs due to Salmonella species would appear to be uncommon, it is worth noting that at least one adult cat with LRTI due to salmonellosis (and concurrent A abstrusus and Pseudomonas species infections) presented with signs typical of FBD/asthma: chronic coughing, dyspnoea and tachypnoea in an otherwise apparently healthy cat. 3 Another adult cat had no clinical history of respiratory disease, rather a chronic history of inappetence, lethargy and recent weight loss. 76 If corticosteroids had been used for non-specific symptomatic treatment in either cat, then there may have been very serious consequences.
Eugonic Fermenter 4 (Neisseria species)
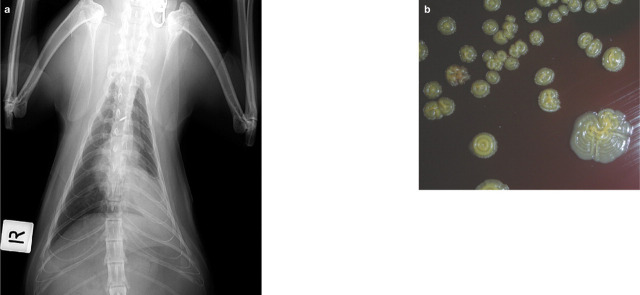
Eugonic Fermenter 4 bacteria (EF-4) have been isolated as commensal and opportunistic flora from the oral cavity of cats and occasionally cause pneumonia in cats. 75 They are ecologically and culturally similar to Pasteurella species 75 but a recent taxonomic study has shown that groups EF-4a and EF-4b belong to the genus Neisseria. 78 While pneumonia due to EF-4 bacteria is not commonly reported, it has been uniformly fatal 1,79–84 until recently (J Beatty, P Martin, unpublished data, Fig 5). Disease is usually sporadic, although an epizootic outbreak occurred in a colony with enzootic feline upper respiratory viral disease and a high rate of feline leukaemia virus (FeLV) infection. 84
FIG 5.
Imaging and culture findings in a 3-year-old castrated male domestic shorthair cat with a LRTI. EF-4 bacteria were isolated from lung FNAs. (a) Dorsoventral thoracic radiograph demonstrating an alveolar pattern of opacity in the left caudal lung lobe, consistent with lobar consolidation. (b) Characteristic yellow, crinkled colonies on the primary isolation plate. These revert to smooth colonies on subculture. Photographs and case details courtesy of Dr Julia Beatty
Affected cats have been adults presenting with acute depression, dehydration, respiratory distress, salivation 79,81,83 or sudden death. 80,82 Leukopenia with a degenerative left shift was present in two cases ante-mortem. 79 In one cat, a rare bacterium was present within granulocytes in the blood. 79 Thoracic radiographs may demonstrate lung consolidation (Fig 5a) and/or discrete focal densities. 79 Most cats with pneumonia due to EF-4 have not survived more than a few days after presentation, although one cat was ill for a week before presenting comatose and being euthanased 79 and two cats had a 2-week history of illness. 83 Penicillin treatment (of unknown dose, route or duration) was not successful in a published case of EF-4 pneumonia 84 and antibiotic therapy (no details) was also unsuccessful in another cat. 79 Diagnosis has been made by necropsy in all published cases.
Recently, a 3-year-old neutered male domestic shorthair cat presented to the Valentine Charlton Cat Centre, University of Sydney, Australia, with a consolidated lung lobe and had EF-4 cultured from lung FNA samples (Fig 5). This cat was treated with intravenous ticarcillin and subcutaneous enrofloxacin for 48 h before being discharged (against veterinary advice). The cat was then treated orally with marbofloxacin 5 mg/kg q24h for 28 days, in conjunction with amoxicillin-clavulanate 20 mg/kg q12h for the first 6 days. The cat was not returned for reassessment but was still alive 8 months after initial presentation (J Beatty, unpublished data).
Yersinia pestis
Plague exists in every continent apart from Australia. The areas most frequently associated with plague foci are adjacent to deserts and have semi-arid, cooler climates, although there are foci in areas such as Southeast Asia, which are unlikely to have that profile. 85 Practitioners in the mid- and far-western USA must consider plague as a differential diagnosis in any cat with pneumonia. 85,86
Pneumonic plague in cats can develop secondarily to haematogenous or lymphatic spread from bubonic or septicaemic forms. 85,87 However, in a large case series, 8/12 cats with pneumonia appeared to have primary pneumonic plague. 87 Cats have also been thought to be responsible for primary pneumonic plague in people who have had contact with them. 85
Although infection with Y pestis is relatively rare, its importance relates to its zoonotic potential and the fact that untreated pneumonic plague in cats and humans is uniformly fatal. 85,86 Gentamicin and tetracyclines are considered effective antibiotics against Y pestis in humans. 88,89 In cats, tetracyclines (with doxycycline preferred) are used primarily for uncomplicated cases, for the bubonic form and, prophylactically, for in-contact cats. 85,87 The recovery rate in a large cases series of cats treated with tetracyclines was 95%, 87 but administration of tetracyclines has been reported to have been associated with relapse. 85 No clinical data exist on the efficacy of fluoroquinolones in affected cats. 85
Practitioners in the mid- and far-western USA must consider plague as a differential diagnosis in any cat with pneumonia.
Mycobacterium species
Lower respiratory tract infections due to M tuberculosis, M bovis, M microti-like mycobacteria, 90–92 M avium complex (MAC), 24 M thermo-resistibile, 93 M genavense, 94 M fortuitum 95 and M simiae 96 have all been reported in cats. Of these, only M tuberculosis, M bovis and M thermoresistibile have been reported as causing localised respiratory tract infections. 90–93 Siamese and Abyssinian cats may be predisposed to mycobacteriosis. 24
Diagnosis of mycobacterial LRTI requires cytology or histopathology and culture. Bronchoalveolar lavage cytology and culture may be unhelpful in disseminated mycobacteriosis due to MAC even when clinical and radiographic signs of LRTI are present. 24
Management of mycobacterial infections is dependent on the species isolated. Mycobacteria have a well-documented capacity to develop resistance to various antimicrobial agents and combination antimicrobial therapy is recommended when treatment is being considered. 93
Rhodococcus equi
Usually considered an equine pathogen, Rhodococcus equi has occasionally been reported to cause disease, including LRTI, in cats. 97 Lower respiratory tract infection was reported in four kittens from unrelated, in-contact litters in a breeding cattery. One kitten had a moist productive cough and subsequently died. Necropsy revealed suppurative bronchopneumonia and a pure growth of R equi was cultured from the lungs. The remaining kittens were successfully treated with doxycycline with or without erythromycin. 97
Other bacteria
Other bacteria are recorded in individual cases of feline pneumonia.
A report on pulmonary lobectomy as a treatment for pneumonia documented a dual infection with Nocardia species and Corynebacterium species, and another infection due to Bacteroides species. 98
Bergeyella (Weeksella) zoohelcum, an organism which is part of the normal feline oropharyngeal flora, was linked to necrotising upper and lower respiratory tract infections in a 9-month-old cat in another report. 99
Capnocytophaga cynodegmi was reported in a cat with pulmonary carcinoma. 100
Chlamydophila felis causes mild pulmonary lesions at post-mortem examination 101 but it would not appear to be an important cause of feline pneumonia. In the largest retrospective study of feline LRTIs, chlamydial antigen was not demonstrated in any of the 245 cases. 2
Parasitic LRTIs
Aelurostrongylus abstrusus
A recent study in semi-feral Australian cats demonstrated the prevalence of A abstrusus to be 13.8%; 21 a study in Italy demonstrated a prevalence of 24%. 102 The prevalence in pet cats is likely to be considerably lower but LRTIs due to A abstrusus may well be underdiagnosed as infection is usually self-limiting, often asymptomatic, may mimic FBD/asthma and responds reasonably well to symptomatic treatment for FBD/asthma. While not usually considered a major clinical problem, A abstrusus can cause severe signs of lower respiratory tract disease, including dyspnoea and coughing. It may also predispose to infections with enteric bacteria such as Salmonella species and E coli (Fig 4), with migrating lungworm larvae possibly acting as carriers for intestinal bacteria. 3,77
Diagnosis
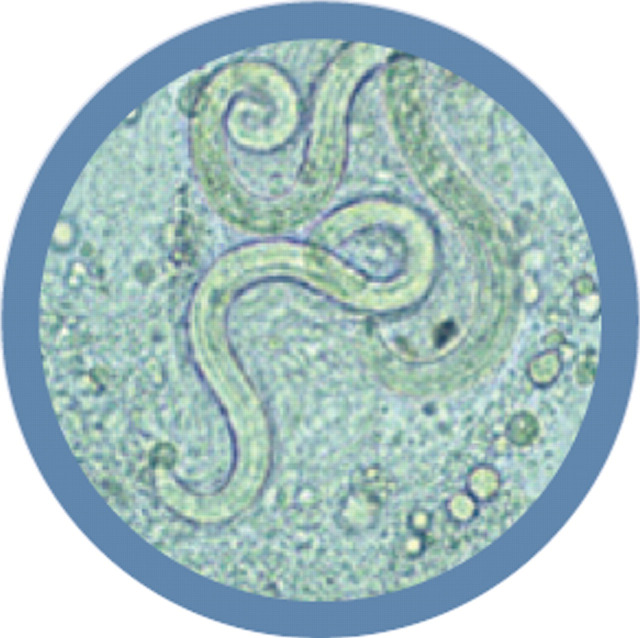
Diagnosis of A abstrusus infection can be difficult. 3,21,103 Both BAL (Fig 6) 3 and lung FNA cytology have been used for diagnosis. 104 A recent study comparing BAL examination, faecal sedimentation—flotation, histological examination and the Baermann technique (minced lung tissue and faeces) demonstrated that the Baermann technique on faeces was the most sensitive test for the detection of A abstrusus infection. 21 The combination of stereomicroscopic and cytological examination of BAL fluid improved the sensitivity of BAL diagnosis in that study, and a direct smear of any gross mucus was recommended in addition to standard cytospin preparations. 21
FIG 6.
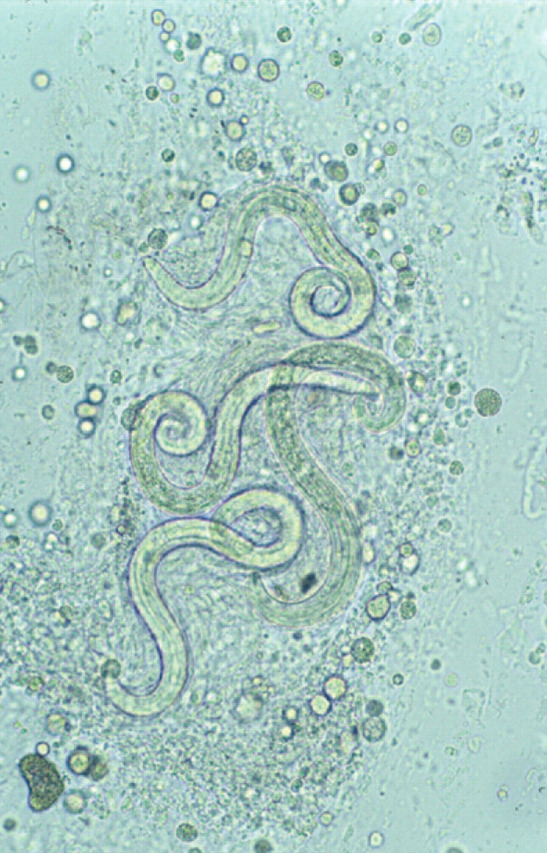
Wet preparation of BAL fluid showing larvae of A abstrusus
However, faecal analysis by the Baermann technique has limitations: it is time consuming (24–36 h), requires well-trained microscopists and is not able to diagnose infections during the prepatent period and when larvae are not being shed. 103 Another faecal analysis method, the FLOTAC technique, is considerably easier although requires specific equipment. A study performed on faeces from a single infected cat indicated that this technique yielded significantly more larvae per gram than the Baermann technique. 105 Recently, a nested PCR test performed on pharyngeal swabs has also been shown to have excellent sensitivity (96.6%) and specificity (100%), 106 and thus would appear to be the diagnostic test of choice if available.
While not usually considered a major clinical problem, A abstrusus can cause severe signs of lower respiratory tract disease, including dyspnoea and coughing.
Treatment
A single dose of ivermectin (400 μg/kg SC) is often recommended for treatment of A abstrusus infections. 4,86 This recommendation is made because a single dose of oral iver-mectin at 300 μg/kg was ineffective in two cats 107 and ivermectin administered at 200 μg/kg SC proved ineffective in another cat; 108 a dose of 400 μg/kg SC 2.5 weeks later in this particular cat was successful. 108 While the high dose appeared effective, it may be that two doses were in fact required for complete resolution, as two doses of abamectin 300 μg/kg SC, 2 weeks apart, have been successful. 3 There is one report of a cat treated successfully with a single 400 μg/kg SC dose of ivermectin, 109 but treatment failures with this dose have also been reported. 109,110 While a single dose of ivermectin 400 μg/kg SC may result in elimination of infection or decreased larval output and improved clinical signs, it cannot be recommended unless the response is monitored with Baermann faecal analysis (or a test of similar sensitivity). Two doses are required if empirical treatment is employed. It is worth noting that use of ivermectin or abamectin is ‘off-label’ and care needs to be taken in kittens. The bioavailability of ivermectin may be lower in cats than dogs and thus parenteral administration is preferable to oral. 3
Oral fenbendazole (50 mg/kg q24h for 10–20 days) is another possible treatment. 110,111 The usual licensed dose (50 mg/kg q24 h for 3 days) does not clear infections in all cats. 112,113
Topical therapy for this parasite has been reported. 110,112–114 Imidacloprid/moxidectin (Advocate; Bayer), given once, proved 100% effective in 12 cats, 112 which makes this the treatment of choice in most countries, being available, registered for cats, easy to administer and efficacious. Emodepside/praziquantel (Profender; Bayer) was 99% effective. 113 Topical selamectin has only been effective in 2/4 cats in which its use has been reported. 110,114
Eucoleus aerophilus
Eucoleus aerophilus (previously Capillaria aerophila) has a worldwide distribution. In Australia, a prevalence of 3–5% has been reported, 115,116 and a recent study demonstrated a similar prevalence (5.5%) in Italy. 117 The prevalence of clinical disease due to E aerophilus has previously been assumed to be low, 4,86 but 8/11 cats in one study had respiratory signs (most commonly, general respiratory distress, dry cough, wheezing and sneezing). 117 These findings indicate that E aerophilus is of clinical importance and should be included in the differential diagnosis of lower respiratory tract diseases in cats. In addition, it should be noted that this parasite has zoonotic potential: human capillariasis can cause severe clinical signs and mimic bronchial carcinoma. 118
KEY POINTS: LUNGWORM.
Lungworm infections due to A abstrusus and E aerophilus may be overlooked in cats.
Specific detection techniques may be required for diagnosis of infection due to A abstrusus.
Topical imidacloprid/moxidectin (Advocate; Bayer) would appear to be the treatment of choice for A abstrusus infection.
Single-dose ivermectin therapy is not recommended for treatment of A abstrusus infection.
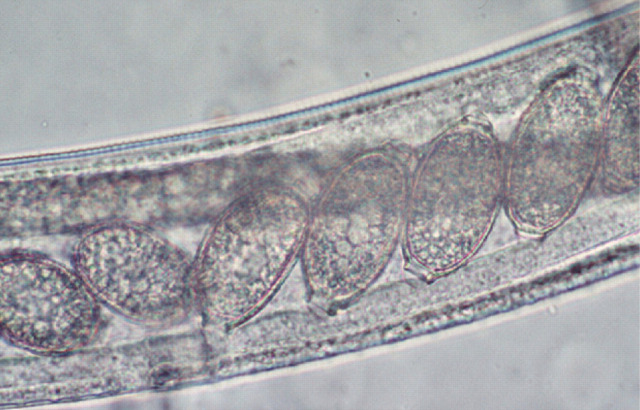
Diagnosis of infection with E aerophilus should be straightforward as the ova are passed in the faeces and routine faecal flotation is adequate for detection. However, the double-operculated ova of E aerophilus (Fig 7) may be mistaken for Trichuris species when found in faecal preparations.
FIG 7.
Wet preparation of BAL fluid showing an adult E aerophilus and its double-operculated ova with asymmetric bipolar plugs
Two doses of abamectin (Avomec; Merial) 300 μg/kg SC, 2 weeks apart, were used successfully for the treatment of E aerophilus in a case report. 119 Fenbendazole, at 50 mg/kg PO for 10–14 days, has also been suggested. 111 Treatment with topical Advocate (Bayer) or Profender (Bayer), as for A abstrusus, may be worth considering but the efficacy of these topical agents has not been reported for infections due to E aerophilus.
E aerophilus is of clinical importance and has zoonotic potential. The double-operculated ova may be mistaken for Trichuris species.
Toxoplasma gondii
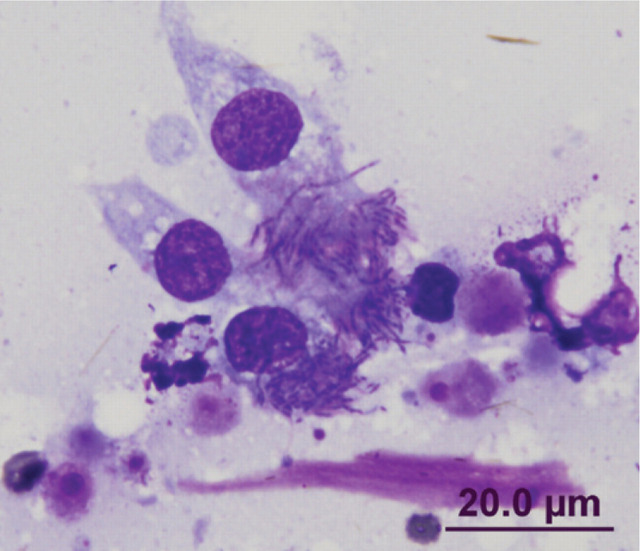
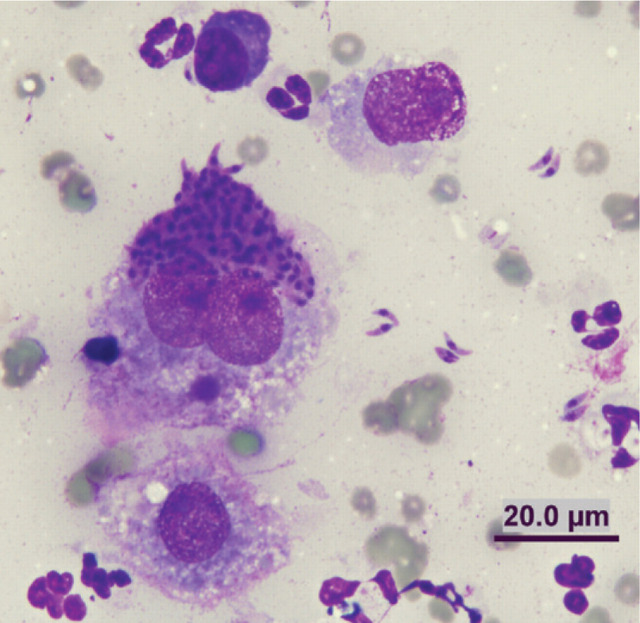
Cats are definitive hosts for the coccidian parasite T gondii. The lung appears to be a target organ in both primary and reactivated toxoplasmosis in cats. 3,26,120–132 The risk of infection is increased with immunosuppression and there is an increasing number of reports of pulmonary toxoplasmosis in cats that have been treated with ciclosporin (Fig 8). 128,130,132
FIG 8.
Diff Quik-stained smear of a lung FNA from a 7-year-old castrated male Devon Rex, which had been on ciclosporin therapy. Note the individual tachyzoites and an aggregate of tachyzoites within a macrophage. The cat was successfully treated with clindamycin and pyrimethamine; clindamycin was continued for 10 weeks, pyrimethamine for 6 weeks. Case details courtesy of Dr Katherine Briscoe
Diagnosis
Diagnosis of LRTIs due to T gondii is very difficult. Radiology and pulmonary cytology may be consistent with neoplasia. 3,125,127 Serology may be consistent with infection but does not provide a definitive diagnosis. Diagnosis is possible by BAL cytology in cats, 26,124,128,129,131,132 but lung FNA cytology and lung biopsy evaluation may fail to identify T gondii. 3,26,125 Immunohistochemistry and tissue culture have been recommended for lung biopsies and BAL specimens from human patients with acquired immunodeficiency syndrome (AIDS). 133,134
Treatment
There appears to be no published work comparing the clinical efficacy of drugs used to treat feline toxoplasmosis. A study of clindamycin efficacy in an experimental model of acute toxoplasmosis demonstrated that there was increased morbidity and mortality from hepatitis and interstitial pneumonia in both clindamycin treatment groups (12.5 mg/kg q12h PO and 11 mg/kg q24h PO) compared with both control groups (placebo once or twice daily PO). 135 Possible causes for this were discussed including the potential for clindamycin to inhibit phagocyte action. In addition, the report drew attention to the facts that clindamycin is more suppressive than curative for T gondii, that concentrations found to be inhibitory in vitro may not be achieved in vivo and that the antimicrobial effect of clindamycin is delayed in vitro for 1–3 days; 136 this last finding perhaps accounts for the lack of any demonstrable clindamycin activity against T gondii in early in vitro studies. 136–138 Clindamycin is also not effective against extracellular tachyzoites. 138
Assessment of the efficacy of clindamycin in clinical cases is difficult. One study reported it to be efficacious; 139 however, the selection criteria for that study included response to appropriate treatment or histopathological confirmation and the only case with definite histological confirmation died without treatment. Pooling several papers 123,124,126–129,132 and one unpublished case (K Briscoe, personal communication, Fig 8) of confirmed pulmonary toxoplasmosis, only one cat with mild clinical signs (but significant pulmonary pathology) responded well to clindamycin treatment alone. 127 Another cat with a 2-week history of respiratory signs and tachypnoea and dyspnoea on presentation, due to pulmonary toxoplasmosis, survived 3 months after being given clindamycin as the sole treatment, before dying (no necropsy performed). 123 However, two cats treated solely with clindamycin died. 126,128 Three cats treated with clindamycin and trimethoprim/sulphonamide 129,132 or sulfamethoxazole 128 also died, as did one treated with clindamycin and doxycycline. 124 One cat treated with clindamycin plus pyrimethamine for 16 days and then trimethoprim/sulphonamide and pyrimethamine for 26 days survived long-term, 132 as has a recent unpublished case treated with clindamycin and pyrimethamine (K Briscoe, personal communication, Fig 8). Thus, in 10 cases of pulmonary toxoplasmosis, only one survived long-term with sole clindamycin therapy and no cats survived with clindamycin and sulphonamide treatment. The only clindamycin combination therapy that was successful was clindamycin and pyrimethamine.
One cannot draw any real conclusions from such a limited number of cases. It may be that cats with pulmonary toxoplasmosis and a critical presentation have a very poor prognosis, regardless of which antimicrobial agent is used, or that clindamycin alone is poorly efficacious and possibly even detrimental in these situations. As such, based on the clinical cases reported and the experimental and theoretical data regarding clindamycin, it is difficult to recommend clindamycin alone in critical cases of pulmonary toxoplasmosis. Clindamycin (12.5 mg/kg q24h PO) and pyrimethamine (0.25–0.5 mg/kg q12h PO) may be a better option, especially in cats immunosuppressed with ciclosporin. Clindamycin should always be followed by food or a water swallow as its use has been associated with oesophageal injury. 140
KEY POINTS: PULMONARY TOXOPLASMOSIS.
Definitive diagnosis is usually BAL cytology or post-mortem histopathology.
Pulmonary toxoplasmosis is increasingly identified as a complication of ciclosporin therapy in cats.
Sole clindamycin therapy is not recommended for pulmonary toxoplasmosis.
Clindamycin (12.5 mg/kg q24h PO) and pyrimethamine (0.25–0.5 mg/kg q12h PO) combination therapy may be useful to treat pulmonary toxoplasmosis in cats immunosuppressed by ciclosporin.
Treatment with sulphonamides would also appear to be ineffective. Dubey and Carpenter reported that 17/17 cases of toxoplasmosis (signalment, presentation details and organ systems affected not specified) died despite sulphonamide treatment: 12 died or were euthanased within 30 h and the other five lived from 2–13 days. 122 Eight of these cats were treated concurrently with pyrimethamine. Pyrimethamine alone has marked in vitro activity against T gondii whereas sulfadiazine has not, but pyrimethamine and sulfadiazine have synergistic activity. 137 Pyrimethamine has greater efficacy than trimethoprim when used in combination with a sulphonamide but can cause bone marrow suppression in cats. 141 Regardless, it is probably worth considering this drug in the initial acute phases of disease.
Doxycycline, minocycline, azithromycin, clarithromycin and various combinations with pyrimethamine and sulphonamides are also outlined as possible treatment protocols; 141 azithromycin is reported to have a delayed action of onset similar to clindamycin. 137 It is perhaps also worth considering the use of triazines such as toltrazuril, which have shown promise in the treatment of equine protozoal myeloencephalitis. 142 Diclazuril and pyrimethamine in mice with experimentally induced acute toxoplasmosis had a synergistic effect on survival, 143 and toltrazuril has been shown to be effective against the intestinal developmental stages of T gondii and reasonably effective against extraintestinal stages of T gondii in cats. 144
Paragonimus species
Trematodes in the genus Paragonimus develop to maturity in the lungs. While P kellicotti is the most commonly reported pathogen of the genus in cats, there are at least 28 species and all are probably capable of developing within the cat. 145 Typically, they are found in cysts within the lungs. Each cyst may contain 1–10 flukes. 145 They can cause serious disease in the infected host and a number of these trematodes, including P westermani, cause serious disease in people. 145 Natural infection in cats with paragonimiasis in parts of Asia may be as prevalent as 45–59%. 146,147
Paragonimus kellicotti is distributed throughout the Mississippi and Great Lakes drainage systems of North America. 145 Aquatic snails and crayfish are required intermediate hosts. Infections may be subclinical or cause coughing (occasionally haemoptysis) or dyspnoea, due to inflammation, pneumothorax or secondary bacterial pneumonia. Wheezing in cats may mimic FBD/asthma. Thoracic radiographs demonstrate air-filled cysts or tissue masses averaging 1 cm, usually involving the caudal lung lobes. Diagnosis can be made by BAL or faecal analysis (sedimentation recommended and multiple faecal samples may be required). Treatment involves praziquantel (at 25 mg/kg q8h PO for 3 days) or fenbendazole (at 25–50 mg/kg q12h PO for 10–14 days). 4
There is an increasing number of reports of pulmonary toxoplasmosis in cats that have been treated with ciclosporin.
Paragonimus westermani is found in Southeast Asia. In cats with heavy natural infections, there is extensive injury to the lung parenchyma (atelectasis and fibrosis) and the pleura (thickening and fibrosis). 145 Successful treatment of a natural infection with prazi-quantel 100 mg/kg daily for 2 days has been reported. 148
Paragonimus mexicanus, found in Mexico and Central and South America, causes similar signs to P kellicotti. 145 Paragonimus heterotremus, found in China, Thailand and Laos, has been reported to have caused the death of a cat that had a natural infection with 13 flukes. 149 Paragonimus miyazakii occurs naturally in cats in Japan 150 and is reported to cause similar clinical signs to P westermani. 145 Paragonimus skrjabini occurs naturally in cats in China 147 but clinical signs do not appear to have been described. 145 Experimentally cats have also been infected with many species including P uterobilateralis, 151 P heterotremus, 152 P yunnanensis, 153 P iloktsuenensis 154 and P veocularis. 155
Cytauxzoon felis
Cytauxzoon felis, a regionally common, tickborne parasite, causes an interstitial pneumonia characterised by neutrophilic infiltrates and pulmonary oedema. Respiratory distress is seen in affected cats. 156 Prevention via ectoparasite control and confinement indoors in the tick season is recommended as treatment attempts have met with limited success and disease is often fatal. 157 Most success in treatments has been with the carbanilide compounds, diminazene or imidocarb. 157
Dirofilaria immitis
Dirofilaria immitis (heartworm) is a cardiovascular parasite that may cause coughing and dyspnoea in cats due to proliferative and inflammatory lesions in the pulmonary arteries, bronchioles and lung parenchyma (heartworm associated respiratory disease). 158 Ante-mortem diagnosis is difficult due to the low number of adult worms generally present in cats. Heartworm antigen and microfilaria testing has low sensitivity and diagnosis usually requires a high degree of clinical suspicion, serology, radiography and echocardiography.
Fungal LRTIs
Fungal causes of feline LRTIs include Cryptococcus species, Histoplasma capsulatum, Sporothrix schenkii, Aspergillus species, Mucor species, Candida species, Coccidioides immitis, Blastomyces dermatitidis, Ochroconis gallopavum, Paecilomyces lilacinus and Cladophialophora bantiana. 1–4,159–165 Siamese and Abyssinian cats appear to have an increased prevalence of cryptococcosis 161,165 and, possibly, histoplasmosis. 160 Siamese cats also featured prominently in an early description of blastomycosis cases. 160 Abyssinian and Havana brown cats have been reported to be over-represented for blastomycosis. 161
Ante-mortem clinical diagnosis of fungal LRTIs usually involves thoracic radiographs, cytology and culture. Careful cytology is required to demonstrate organisms within macrophages. Pulmonary FNA cytology is often required and fungal culture increases the likelihood of organism identification. Detection of capsular antigen in serum by the latex agglutination procedure is a sensitive diagnostic test for cryptococcosis. The test, however, is rarely included as a screening test for LRTIs and is more usually performed after diagnosis, in order to monitor treatment.
Cryptococcus species
No cases of cryptococcal pneumonia were identified in the Swiss LRTI study. 2 In the Australian study, 3 cryptococcal LRTI was only identified in a single FIV-positive cat which had the hallmarks of AIDS-like disease: opportunistic infections and, terminally, neoplasia. 119 However, in the American study, 5/39 LRTI cases had a diagnosis of cryptococcal pneumonia. 1
The lungs are considered the primary site of infection for cryptococcosis in humans. Pulmonary cryptococcal infections have been reported to be quite rare in cats, 166 although 29% of cases reviewed in one study of cryptococcosis had thoracic radiographic abnormalities and 38% had pulmonary lesions at necropsy. 161 Pulmonary involvement in one cat was identified only by BAL (thoracic radiographs were normal). 7 It is possible that pulmonary cryptococcosis may be underestimated if BAL or lung histopathology is not performed.
Itraconazole has been used successfully for the treatment of pulmonary cryptococcosis, 119 but monitoring of antigen titres is essential to document disease resolution as with other manifestations of feline cryptococcosis.
Other fungi
Blastomycosis and coccidiomycosis rarely cause respiratory disease in cats. 4,159 Cats, however, are very susceptible to histoplasmosis, most cats presenting with disseminated disease. 167 Signs are usually non-specific 167 but 38–93% of cats have been reported to have respiratory tract signs. 159 While coughing is uncommon, over half of affected cats have dyspnoea, tachypnoea and abnormal lung sounds. 167 Itraconazole is the treatment of choice for histoplasmosis. 159,167
Clinical diagnosis of fungal LRTIs usually involves thoracic radiographs, cytology and culture.
Obtaining an ante-mortem diagnosis of viral pneumonia is rare in clinical practice as histopathology and specific viral tests are required.
Viral LRTIs
Obtaining an ante-mortem diagnosis of viral pneumonia is rare in clinical practice as histopathology and specific viral tests are required.
Feline calicivirus
Feline calicivirus is usually only reported to cause interstitial pneumonia in cats under experimental conditions. 53 However, outbreaks of naturally occurring pneumonia have also occurred. 58,168 In one such outbreak, six kittens died and another six kittens, all with URT signs, were euthanased. The six euthanased kittens had histopathological evidence of pneumonia: multifocal interstitial pneumonia in five and bronchopneumonia in one that had concurrent B bronchiseptica infection. 58 Bronchointerstitial pneumonia can also occur with virulent systemic FCV infections. 169
Feline herpesvirus
Feline herpesvirus is a rare cause of pneumonia 1,53 and is usually reported in kittens or debilitated animals. 1,2,53,170 However, a recent retrospective study on FHV-induced pneumonia demonstrated fatal fibrinonecrotising pneumonia and severe necrosis of the bronchial and bronchiolar epithelium in four young adult and mature adult cats, none of which had concurrent disease identified or recorded; three were from shelters but one was from a private household or breeding cattery (combined category for the study). 5 The two oldest cats (aged 3 and 8 years), one from a shelter and one from a private household/breeding cattery, had sudden death as their only history. It may be that FHV is an underdiagnosed cause of pneumonia in older cats as few mature cats with sudden death have post-mortem histopathology performed.
Treatment with specific antiherpetic viral medication could be considered if LRTI due to FHV was suspected. Famciclovir (62.5 mg q12h to 125 mg q8h PO) has recently been associated with improvement in some adult cats with FHV-related disease, 171 but there are no reports of its use in LRTI due to FHV. Adjunctive therapies with lysine (250 mg q12h PO) and interferon alpha may also be worth considering.
Cowpox
Cowpox virus infection in cats is rare and usually leads to cutaneous lesions. 172 Until recently, cases of pulmonary infection and pneumonia have been associated with fatality. 173–175 Johnson et al, however, recently described a cat that recovered from a necrotising bronchopneumonia in which both cowpox virus and FHV were confirmed using histopathology, scanning electron microscopy and isolation of poxvirus from skin lesions. 172 Such investigations would be rare on live cats so it is possible that resolving pneumonia due to cowpox may have been underreported.
Consolidated lung lobes with mild pleural effusion would appear to be a radiographic feature and should prompt consideration of this disease in regions in which this infection is known to occur. 172 Cowpox is zoonotic and transmission to a human has occurred in at least one instance of feline cowpox pneumonia. 175
Avian influenza virus A (H5N1)
Avian influenza virus A (H5N1) was reported to have caused the death of 14 cats in a house-hold in Thailand, 176 and also caused lethal bronchointerstitial pneumonia in three cats with concurrent A abstrusus infection in Germany. 177 Outdoor cats in contact with wild birds infected with avian influenza are at risk of lethal infection. Detailed articles on feline infection, prevention and management have been published. 178,179
Swine-origin influenza A virus (H1N1)
Swine-origin influenza has also been reported in cats. 180,181 Two cats from Oregon died with moderate to severe necrotising bronchointerstitial pneumonia after H1N1 infections. 180 The cats lived in separate households, 99 km apart. Humans in both households were assumed or proven to be infected with H1N1. One of the households had five additional cats, four of which had signs of respiratory disease but recovered (one was negative for H1N1 on a nasal swab) and one of which was unaffected. Transmission of pandemic 2009 (H1N1) influenza virus from human to cat was also recently implicated in the case of an indoor domestic cat that recovered from clinical disease. 181 Polymerase chain reaction testing on a BAL sample was positive for H1N1 in the cat. Retrospective testing of the family members, two of three of whom had influenza-like signs prior to the cat's illness, was unsuccessful.
Treatment of LRTIs — some general considerations.
Perform appropriate diagnostics
Due to the diversity of causes of feline LRTIs, empirical therapy in cats with signs of lower respiratory tract disease is not recommended and appropriate cytology, parasitology and microbiology should be performed in all cases.
Consider the drug characteristics
When treating bacterial LRTIs it is important to consider not only the organism, but also the drug characteristics. Although most antimicrobials are able to attain concentrations in the lung parenchyma comparable with drug concentrations in the serum, concentrations in the airways and bronchial secretions may be significantly lower. Beta-lactams are hydrophilic and are poorly distributed in respiratory secretions and phagocytic cells. 60 Aminglycosides are inactivated in inflammatory environments after penetration into bronchial secretions and have a low degree of lipid solubility. 60 Thus, neither class is recommended for treating bronchial infections such as B bronchiseptica. Fluoroquinolones achieve high levels of distribution in respiratory tissues and intracellularly, as do the macrolides and chloramphenicol. Metronidazole also achieves a high distribution into bronchial secretions. Tetracyclines are lipophilic and tend to cross the blood—bronchus barrier reasonably well, especially doxycycline. 60
Provide supportive therapy if required
Supportive therapy may be required. Intravenous fluids may aid in maintaining euvolaemia and airway hydration. 86,187 Caution must be exercised with high volume fluids in cases of severe pneumonia as the blood—alveolar barrier may be compromised. 187 Oxygen supplementation should be provided if the SpO2 is less than 94% or if the PaO2 is less than 80 mmHg. Methods of administering oxygen include oxygen cages, nasal catheters and flow-by delivery. 187 Severe hypoxaemia may require mechanical ventilation. Use of bronchodilators such as inhaled salbutamol, oral or parenteral terbutaline or oral theophylline is controversial, 187 but bronchodilation does seem helpful in some cases of feline LRTI. 3
Lung lobectomy
Lung lobectomy is occasionally indicated for treatment when pneumonia fails to resolve with appropriate antimicrobial therapy. 86,98 Residual infection in a single lobe may be related to an underlying physical problem such as a bronchial foreign body, abscess or tumour. 86
Other viruses
The paramyxoviruses, equine morbillivirus (Hendra virus) 182 and Nipah virus, 183 have been reported to cause pneumonia experimentally in cats, though neither are primarily feline viruses.
Feline infectious peritionitis due to feline coronoviral infections may involve the pulmonary parenchyma. 1,4 Neither FIV nor FeLV directly cause overt signs of lower respiratory tract disease, 4 although FIV has been reported to cause interstitial pneumonitis in infected cats 184 and alter BAL cytology in cats experimentally infected with FIV for at least 8 months. 185 Feline foamy virus also caused histopathological lesions in the lungs after experimental infection, but did not result in clinical signs. 186
KEY CONCLUSIONS: FELINE LRTIs.
Ante-mortem diagnosis of LRTI can be very challenging.
Due to the diversity of causes of LRTIs in cats, empirical therapy in cats with signs of lower respiratory tract disease is not recommended and appropriate cytology, parasitology and microbiology should be performed in all cases.
Bronchoalveolar lavage can be performed simply in practice without bronchoscopy equipment.
If specimen transport and culture methods for mycoplasmal detection are suboptimal, empirical trial therapy with doxycycline is recommended.
Should BAL be impossible due to financial or patient constraints, then therapy should address potential bacterial and parasitic infections prior to any medium- to long-term corticosteroid administration for FBD/asthma.
Acknowledgments
The authors express appreciation to Drs Julia Beatty, Katherine Briscoe and Katrina Bosward from the Faculty of Veterinary Science, University of Sydney, Australia, and Dr Darren Foster from the Small Animal Specialist Hospital, North Ryde, Australia, for generously providing excellent case material and photos.
References
- 1. Macdonald ES, Norris CR, Berghaus RB, Griffey SM. Clinicopathologic and radiographic features and etiologic agents in cats with histologically confirmed infectious pneumonia: 39 cases (1991–2000). J Am Vet Med Assoc 2003; 223: 1142–50 [DOI] [PubMed] [Google Scholar]
- 2. Bart M, Guscetti F, Zurbriggen A, Pospischil A, Schiller I. Feline infectious pneumonia: a short literature review and a retrospective immunohistological study on the involvement of Chlamydia spp and distemper virus. Vet J 2000; 159: 220–30 [DOI] [PubMed] [Google Scholar]
- 3. Foster SF, Martin P, Allan GS, Barrs VR, Malik R. Lower respiratory tract infections in cats: 21 cases (1995–2000). J Feline Med Surg 2004; 6: 167–80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Hawkins EC. Pulmonary parenchymal diseases. In: Ettinger SJ, Feldman EC, eds. Textbook of veterinary internal medicine. 5th edn. Philadelphia: Saunders, 2000: 1061–91. [Google Scholar]
- 5. Chvala-Mannsberger S, Bagó Z, Weissenböck H. Occurrence, morphological characterization and antigen localization of felid herpesvirus-induced pneumonia in cats: a retrospective study (2000–2006). J Comp Pathol 2009; 141: 163–69 [DOI] [PubMed] [Google Scholar]
- 6. Welsh RD. Bordetella bronchiseptica infections in cats. J Am Anim Hosp Assoc 1996; 32: 153–58 [DOI] [PubMed] [Google Scholar]
- 7. Hamilton TA, Hawkins EC, DeNicola DB. Bronchoalveolar lavage and tracheal wash to determine lung involvement in a cat with cryptococcosis. J Am Vet Med Assoc 1991; 198: 655–56 [PubMed] [Google Scholar]
- 8. Hawkins EC. Bronchoalveolar lavage. In: King LG, ed. Textbook of respiratory disease in dogs and cats. St Louis: Saunders, 2004: 118–28. [Google Scholar]
- 9. Johnson LR, Drazenovich TL. Flexible bronchoscopy and bronchoalveolar lavage in 68 cats (2001–2006). J Vet Intern Med 2007; 21: 219–225 [DOI] [PubMed] [Google Scholar]
- 10. Foster SF, Martin P, Braddock JA, Malik R. A retrospective analysis of feline bronchoalveolar lavage cytology and microbiology (1995–2000). J Feline Med Surg 2004; 6: 189–98 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Foster D. Diagnosis and management of chronic coughing in cats. In Practice 1998; 20: 261–67 [Google Scholar]
- 12. Sparkes A, Wotton P, Brown P. Tracheobronchial washing in the dog and cat. In Practice 1997; 19: 257–59 [Google Scholar]
- 13. McCarthy G, Quinn PJ. The development of lavage procedures for the upper and lower respiratory tract of the cat. Irish Vet J 1986; 40: 6–9 [Google Scholar]
- 14. Moise NS, Blue J. Bronchial washings in the cat: procedure and cytologic evaluation. Compend Contin Educ Pract Vet 1983; 5: 621–27 [Google Scholar]
- 15. Syring RS. Tracheal washes. In: King LG, ed. Textbook of respiratory disease in dogs and cats. St Louis: Saunders, 2004: 128–34. [Google Scholar]
- 16. Wood EF, O'Brien RT, Young KM. Ultrasound-guided fine-needle aspiration of focal parenchymal lesions of the lung in dogs and cats. J Vet Intern Med 1998; 12: 338–42 [DOI] [PubMed] [Google Scholar]
- 17. Reichle JK, Wisner ER. Non-cardiac thoracic ultrasound in 75 feline and canine patients. Vet Radiol Ultrasound 2000; 41: 154–62 [DOI] [PubMed] [Google Scholar]
- 18. DeBerry JD, Norris CR, Samii VF, Griffey SM, Almy FS. Correlation between fine-needle aspiration cytopathology and histopathology of the lung in dogs and cats. J Am Anim Hosp Assoc 2002; 38: 327–36 [DOI] [PubMed] [Google Scholar]
- 19. Zekas LJ, Crawford JT, O'Brien RT. Computed tomography-guided fine-needle aspirate and tissue-core biopsy of intra-thoracic lesions in thirty dogs and cats. Vet Radiol Ultrasound 2005; 46: 200–4 [DOI] [PubMed] [Google Scholar]
- 20. Dehard S, Bernaerts F, Peeters D, et al. Comparison of bronchoalveolar lavage cytospins and smears in dogs and cats. J Am Anim Hosp Assoc 2008; 44: 285–94 [DOI] [PubMed] [Google Scholar]
- 21. Lacorcia L, Gasser RB, Anderson GA, Beveridge I. Comparison of bronchoalveolar lavage fluid examination and other diagnostic techniques with the Baermann technique for detection of naturally occurring Aelurostrongylus abstrusus infection in cats. J Am Vet Med Assoc 2009; 235: 43–49 [DOI] [PubMed] [Google Scholar]
- 22. Moise NS, Wiedenkeller D, Yeager AE, Blue JT, Scarlett J. Clinical, radiographic, and bronchial cytologic features of cats with bronchial disease: 65 cases (1980–1986). J Am Vet Med Assoc 1989; 194: 1467–73 [PubMed] [Google Scholar]
- 23. American Thoracic Society. Clinical role of bronchoalveolar lavage in adults with pulmonary disease. Am Rev Respir Dis 1990; 142: 481–86. [DOI] [PubMed] [Google Scholar]
- 24. Baral RM, Metcalfe SS, Krockenberger MB, et al. Disseminated Mycobacterium avium infection in young cats: overrepresentation of Abyssinian cats. J Feline Med Surg 2006; 8: 23–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Norris CR, Griffey SM, Samii VF, Christopher MM, Mellema MS. Thoracic radiography, bronchoalveolar lavage cytopathology, and pulmonary parenchymal histopathology: a comparison of diagnostic results in 11 cats. J Am Anim Hosp Assoc 2002; 38: 337–45 [DOI] [PubMed] [Google Scholar]
- 26. Greenlee PG, Roszel JF. Feline bronchial cytology: histologic/cytologic correlation in 22 cats. Vet Pathol 1984; 21: 308–15 [DOI] [PubMed] [Google Scholar]
- 27. Padrid PA, Feldman BF, Funk K, Samitz EM, Reil D, Cross CE. Cytologic, microbiologic, and biochemical analysis of bronchoalveolar lavage fluid obtained from 24 healthy cats. Am J Vet Res 1991; 52: 1300–7 [PubMed] [Google Scholar]
- 28. Randolph JF, Moise NS, Scarlett JM, Shin SJ, Blue JT, Corbett JR. Prevalence of mycoplasmal and ureaplasmal recovery from tracheobronchial lavages and of mycoplasmal recovery from pharyngeal swab specimens in cats with or without pulmonary disease. Am J Vet Res 1993; 54: 897–900 [PubMed] [Google Scholar]
- 29. Waites KB, Talkington DF. Mycoplasma pneumoniae and its role as a human pathogen. Clin Microbiol Rev 2004; 17: 697–728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Switzer WP. The genus Mycoplasma . In: Merchant I, Packer RA, eds. Veterinary bacteriology and virology. 7th edn. Ames: Iowa State University Press, 1967: 531–48. [Google Scholar]
- 31. Wong WT, Noor F. Pyothorax in the cat — a report of two cases. Kajian Vetrinar 1984; 16; 15–17. [Google Scholar]
- 32. Crisp MS, Birchard SJ, Lawrence AE, Fingeroth J. Pulmonary abscess caused by a Mycoplasma sp in a cat. J Am Vet Med Assoc 1987; 191: 340–42 [PubMed] [Google Scholar]
- 33. Pedersen NC. Mycoplasmal infections. In: Holzworth J, ed. Diseases of the cat. Philadelphia: Saunders, 1987: 308–19. [Google Scholar]
- 34. Moise NS, Dietze AE. Bronchopulmonary diseases. In: Sherding RG, ed. The cat: diseases and management. New York: Churchill Livingstone, 1989: 809. [Google Scholar]
- 35. Malik R, Love DN, Hunt GB, Canfield PJ, Taylor V. Pyothorax associated with a Mycoplasma species in a kitten. J Small Anim Pract 1991; 32: 31–34 [Google Scholar]
- 36. Foster SF, Barrs VR, Martin P, Malik R. Pneumonia associated with Mycoplasma spp in three cats. Aust Vet J 1998; 76: 460–64 [DOI] [PubMed] [Google Scholar]
- 37. Chandler JC, Lappin MR. Mycoplasmal respiratory infections in small animals: 17 cases (1988–1999). J Am Anim Hosp Assoc 2002; 38: 111–19 [DOI] [PubMed] [Google Scholar]
- 38. Gulbahar MY, Gurturk K. Pyothorax associated with a Mycoplasma sp and Arcanobacterium pyogenes in a kitten. Aust Vet J 2002; 80: 344–45 [DOI] [PubMed] [Google Scholar]
- 39. Trow AV, Rozanski EA, Tidwell AS. Primary mycoplasma pneumonia associated with reversible respiratory failure in a cat. J Feline Med Surg 2008; 10: 398–402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Swift S, Dukes-McEwan J, Fonfara S, Loureiro JF, Burrow R. Aetiology and outcome in 90 cats presenting with dyspnoea in a referral population. J Small Anim Pract 2009; 50: 466–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Tan RJS. Susceptibility of kittens to Mycoplasma felis infection. Jpn J Exp Med 1974; 41: 235–40 [PubMed] [Google Scholar]
- 42. Sabato AR, Martin AJ, Marmion BP, Kok TW, Cooper DM. Mycoplasma pneumoniae: acute illness, antibiotics and subsequent pulmonary function. Arch Dis Child 1984; 59: 1034–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Waites KB, Balish MF, Atkinson TP. New insights into the pathogenesis and detection of Mycoplasma pneumoniae infections. Future Microbiol 2008; 3: 635–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Rollins DR, Beuther DA, Martin RJ. Update on infection and antibiotics in asthma. Curr Allergy Asthma Rep 2010; 10: 67–73 [DOI] [PubMed] [Google Scholar]
- 45. Johnston SL, Martin RJ. Chlamydophila pneumoniae and Mycoplasma pneumoniae: a role in asthma pathogenesis? Am J Respir Crit Care Med 2005; 172: 1078–89 [DOI] [PubMed] [Google Scholar]
- 46. Petrovsky T. Mycoplasma pneumoniae infection and post-infection asthma. Med J Aust 1990; 152: 391 [DOI] [PubMed] [Google Scholar]
- 47. Yano T, Ichikawa Y, Komatu S, Arai S, Oizumi K. Association of Myoplasma pneumoniae antigen with initial onset of bronchial asthma. Am J Respir Crit Care Med 1994; 149: 1348–53 [DOI] [PubMed] [Google Scholar]
- 48. Sutherland ER, Brandorff JM, Martin RJ. Atypical bacterial pneumonia and asthma risk. J Asthma 2004; 41: 863–68 [DOI] [PubMed] [Google Scholar]
- 49. Sutherland ER, Martin RJ. Asthma and atypical bacterial infection. Chest 2007; 132: 1962–66 [DOI] [PubMed] [Google Scholar]
- 50. Wu Q, Martin RJ, LaFasto S, et al. Toll-like receptor 2 down-regulation in established mouse allergic lungs contributes to decreased mycoplasma clearance. Am J Respir Crit Care Med 2008; 177: 720–29 [DOI] [PubMed] [Google Scholar]
- 51. Hartmann AD, Helps CR, Lappin MR, Werckenthin C, Hartmann K. Efficacy of pradofloxacin in cats with feline upper respiratory tract disease due to Chlamydophila felis or Mycoplasma infections. J Vet Intern Med 2008; 22: 44–52 [DOI] [PubMed] [Google Scholar]
- 52. German AJ, Cannon MJ, Dye C, et al. Oesophageal strictures in cats associated with doxycycline therapy. J Feline Med Surg 2005; 7: 33–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Gaskell RM, Dawson S, Radford A. Feline respiratory disease. In: Green CE, ed. Infectious diseases of the dog and cat. St Louis: Saunders Elsevier, 2006: 145–54. [Google Scholar]
- 54. Willoughby K, Dawson S, Jones RC, et al. Isolation of B bronchiseptica from kittens with pneumonia in a breeding cattery. Vet Rec 1991; 129: 407–8 [DOI] [PubMed] [Google Scholar]
- 55. Fisk SK, Soave OA. Bordetella bronchiseptica in laboratory cats from central California. Lab Anim Sci 1973; 23: 33–35 [PubMed] [Google Scholar]
- 56. Little S. Bordetella bronchiseptica infection in a cat. Fel Pract 2000; 28: 12–15 [Google Scholar]
- 57. Snyder SB, Fisk SK, Fox JG, Soave OA. Respiratory tract disease associated with Bordetella bronchiseptica infection in cats. J Am Vet Med Assoc 1973; 163: 293–94 [PubMed] [Google Scholar]
- 58. Turnquist SE, Ostlund E. Calicivirus outbreak with high mortality in a Missouri feline colony. J Vet Diagn Invest 1997; 9: 195–98 [DOI] [PubMed] [Google Scholar]
- 59. Foley JE, Rand C, Bannasch MJ, Norris CR, Milan J. Molecular epidemiology of feline bordetellosis in two animal shelters in California, USA. Prev Vet Med 2002; 54: 141–56 [DOI] [PubMed] [Google Scholar]
- 60. Olsen JD. Rational antibiotic therapy for respiratory disorders in dogs and cats. Vet Clin North Am Small Anim Pract 2000; 30: 1337–55 [DOI] [PubMed] [Google Scholar]
- 61. Speakman AJ, Dawson S, Binns SH, Gaskell CJ, Hart CA, Gaskell RM. Bordetella bronchiseptica infection in the cat. J Small Anim Pract 1999; 40: 252–56 [DOI] [PubMed] [Google Scholar]
- 62. Speakman AJ, Binns SH, Osborn AM, et al. Characterization of antibiotic resistance plasmids from Bordetella bronchiseptica. J Antimicrob Chemother 1997; 40: 811–16 [DOI] [PubMed] [Google Scholar]
- 63. Greene CE, Prescott JF. Streptococcal and other Gram-positive bacterial infections. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier, 2006: 302–16. [Google Scholar]
- 64. Blanchard P, Wilson D. Group G streptococcal infections in kittens. In: Kirk RW, ed. Current veterinary therapy X. Philadelphia: WB Saunders, 1989: 1091–93. [Google Scholar]
- 65. Wu CC, Kiupel M, Raymond JT, de Gortari MJ, Lin TL. Group G streptococcal infection in a cat colony. J Vet Diagn Invest 1999; 11: 174–77 [DOI] [PubMed] [Google Scholar]
- 66. Sura R, Hinckley LS, Risatti GR, Smyth JA. Fatal necrotising fasciitis and myositis in a cat associated with Streptococcus canis. Vet Rec 2008; 162: 450–53 [DOI] [PubMed] [Google Scholar]
- 67. Zhang S, Wilson F, Pace L. Streptococcus pneumoniae-associated cellulitis in a two-month-old domestic shorthair kitten. J Vet Diagn Invest 2006; 18: 221–24 [DOI] [PubMed] [Google Scholar]
- 68. Taillefer M, Dunn M. Group G streptococcal toxic shock-like syndrome in three cats. J Am Anim Hosp Assoc 2004; 40: 418–22 [DOI] [PubMed] [Google Scholar]
- 69. Blum S, Elad D, Zukin N, et al. Outbreak of Streptococcus equi subsp zooepidemicus infections in cats. Vet Microbiol 2010; 144: 236–39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Devriese LA, Haesebrouck F. Streptococcus suis infections in horses and cats. Vet Rec 1992; 130: 380 [DOI] [PubMed] [Google Scholar]
- 71. Tillman PC, Dodson ND, Indiveri M. Group G streptococcal epizootic in a closed cat colony. J Clin Microbiol 1982; 16: 1057–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Plumb DC. Veterinary drug handbook. Iowa: Iowa State Press, 2002: 633–39. [Google Scholar]
- 73. Sura R, Van Kruiningen HJ, DebRoy C, et al. Extraintestinal pathogenic Escherichia coli-induced acute necrotizing pneumonia in cats. Zoonoses Public Health 2007; 54: 307–13 [DOI] [PubMed] [Google Scholar]
- 74. Highland MA, Byrne BA, Debroy C, Samitz EM, Peterson TS, Oslund KL. Extraintestinal pathogenic Escherichia coli-induced pneumonia in three kittens and fecal prevalence in a clinically healthy cohort population. J Vet Diagn Invest 2009; 21: 609–15 [DOI] [PubMed] [Google Scholar]
- 75. Greene CE, Norris Reinero C. Bacterial respiratory infections. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier, 2006: 866–82. [Google Scholar]
- 76. Rodriguez CO, Moon ML, Leib MS. Salmonella choleraesuis pneumonia in a cat without signs of gastrointestinal tract disease. J Am Vet Med Assoc 1993; 202: 953–55 [PubMed] [Google Scholar]
- 77. Barrs VR, Swinney GR, Martin P, Nicoll RG. Concurrent Aelurostrongylus abstrusus infection and salmonellosis in a kitten. Aust Vet J 1999; 77: 229–32 [DOI] [PubMed] [Google Scholar]
- 78. Vandamme P, Homes B, Bercovier H, Coenye T. Classification of Centers for Disease Control Group Eugonic Fermenter (EF)-4a and EF-4b as Neisseria animaloris sp nov and Neisseria zoodegmatis sp nov, respectively. Int J Syst Evol Microbiol 2006; 56: 1801–5 [DOI] [PubMed] [Google Scholar]
- 79. Jang SS, Demartini JC, Henrickson RV, Enright FM. Focal necrotizing pneumonia in cats associated with a Gram-negative eugonic fermenting bacterium. Cornell Vet 1973; 63: 446–54 [PubMed] [Google Scholar]
- 80. McParland PJ, O'Hagan J, Pearson GR, Neill SD. Pathological changes associated with group EF-4 bacteria in the lungs of a dog and a cat. Vet Rec 1982; 111: 336–38 [DOI] [PubMed] [Google Scholar]
- 81. Drolet R, Kenefick KB, Hakomaki MR, Ward GE. Isolation of group eugonic fermenter-4 bacteria from a cat with multifocal suppurative pneumonia. J Am Vet Med Assoc 1986; 189: 311–12 [PubMed] [Google Scholar]
- 82. Ceyssens K, Devriese LA, Maenhout T. Necrotizing pneumonia in cats associated with infection by EF-4a bacteria. J Vet Med B 1989; 36: 314–16 [DOI] [PubMed] [Google Scholar]
- 83. Corboz L, Ossent P, Gruber H. Isolation and characterization of group EF-4 bacteria from various lesions in cat, dog and badger. Zentralbl Bakteriol 1993; 279: 140–45 [DOI] [PubMed] [Google Scholar]
- 84. Weyant RS, Burris JA, Nichols DK, et al. Epizootic feline pneumonia associated with Centers for Disease Control group EF-4a bacteria. Lab Anim Sci 1994; 44: 180–83 [PubMed] [Google Scholar]
- 85. Macy D. Plague. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier, 2006: 439–46. [Google Scholar]
- 86. Cohn LA. Pulmonary parenchymal disease. In: Ettinger SJ, Feldman EC, eds. Textbook of veterinary internal medicine. 7th edn. St Louis: Saunders Elsevier, 2010: 1096–119. [Google Scholar]
- 87. Eidson M, Thilsted JP, Rollag OJ. Clinical, clinicopathologic, and pathologic features of plague in cats: 119 cases (1977–1988). J Am Vet Med Assoc 1991; 199: 1191–97 [PubMed] [Google Scholar]
- 88. Boulanger LL, Ettestad P, Fogarty JD, Dennis DT, Romig D, Mertz G. Gentamicin and tetracyclines for the treatment of human plague: review of 75 cases in New Mexico, 1985–1999. Clin Infect Dis 2004; 38: 663–69 [DOI] [PubMed] [Google Scholar]
- 89. Mwengee W, Butler T, Mgema S, et al. Treatment of plague with gentamicin or doxycycline in a randomized clinical trial in Tanzania. Clin Infect Dis 2006; 42: 614–21 [DOI] [PubMed] [Google Scholar]
- 90. Greene CE, Gunn-Moore DA. Infections caused by slow-growing mycobacteria. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier, 2006: 462–77. [Google Scholar]
- 91. O'Reilly LM, Daborn CJ. The epidemiology of Mycobacterium bovis infections in animals and man: a review. Tuber Lung Dis 1995; 76 (suppl 1): 1–46. [DOI] [PubMed] [Google Scholar]
- 92. Isaac J, Whitehead J, Adams JW, Barton MD, Coloe P. An outbreak of Mycobacterium bovis infection in cats in an animal house. Aust Vet J 1983; 60: 243–45 [DOI] [PubMed] [Google Scholar]
- 93. Foster SF, Martin P, Davis W, Allan GS, Mitchell DH, Malik R. Chronic pneumonia caused by Mycobacterium thermoresistibile in a cat. J Small Anim Pract 1999; 40: 433–38 [DOI] [PubMed] [Google Scholar]
- 94. Hughes MS, Ball NW, Love DN, et al. Disseminated Mycobacterium genavense infection in a FIV-positive cat. J Feline Med Surg 1999; 1: 23–29 [DOI] [PubMed] [Google Scholar]
- 95. Couto SS, Artacho CA. Mycobacterium fortuitum pneumonia in a cat and the role of lipid in the pathogenesis of atypical mycobacterial infections. Vet Pathol 2007; 44: 543–46 [DOI] [PubMed] [Google Scholar]
- 96. Dietrich U, Arnold P, Guscetti F, Pfyffer GE, Spiess B. Ocular manifestation of disseminated Mycobacterium simiae infection in a cat. J Small Anim Pract 2003; 44: 121–25 [DOI] [PubMed] [Google Scholar]
- 97. Gunew MN. Rhodococcus equi infection in cats. Aust Vet Pract 2002; 32: 2–5 [Google Scholar]
- 98. Murphy ST, Mathews KG, Ellison GW, Bellah JR. Pulmonary lobectomy in the management of pneumonia in five cats. J Small Anim Pract 1997; 38: 159–62 [DOI] [PubMed] [Google Scholar]
- 99. Decostere A, Devriese LA, Ducatelle R, Haesebrouck F. Bergeyella (Weeksella) zoohelcum associated with respiratory disease in a cat. Vet Rec 2002; 151: 392 [DOI] [PubMed] [Google Scholar]
- 100. Forman MA, Johnson LR, Jang S, Foley JE. Lower respiratory tract infection due to Capnocytophaga cynodegmi in a cat with pulmonary carcinoma. J Feline Med Surg 2005; 7: 227–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Hoover EA, Kahn DE, Langloss JM. Experimentally induced feline chlamydial infection (feline pneumonitis). Am J Vet Res 1978; 39: 541–47 [PubMed] [Google Scholar]
- 102. Traversa D, di Cesare A, Milillo P, Ioria R, Otranto D. Aelurostrongylus abstrusus in a feline colony from central Italy: clinical features, diagnostic procedures and molecular characterization. Parastiol Res 2008; 103: 1191–96 [DOI] [PubMed] [Google Scholar]
- 103. Traversa D, Guglielmini C. Feline aelurostrongylosis and canine angiostrongylosis: a challenging diagnosis for two emerging verminous pneumonia infections. Vet Parasitol 2008; 157: 163–74 [DOI] [PubMed] [Google Scholar]
- 104. Dürr B. [Diagnosis of Aelurostrongylus abstrusus infection by fine needle aspiration of the lungs in two cats.] Kleintierpraxis 2009; 54: 88–92German. [Google Scholar]
- 105. Gaglio G, Cringoli G, Rinaldi L, Brianti E, Giannetto S. Use of the FLOTAC technique for the diagnosis of Aelurostrongylus abstrusus in the cat. Parasitol Res 2008; 103: 1055–57 [DOI] [PubMed] [Google Scholar]
- 106. Traversa D, Iorio R, Otranto D. Diagnostic and clinical implications of a nested PCR specific for ribosomal DNA of the feline lungworm Aelurostronglus abstrusus (Nematoda, Strongylida). J Clin Microbiol 2008; 46: 1811–17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Blagburn BL, Hendrix CM, Lindsay DS, Vaughan JL. Anthelmintic efficacy of ivermectin in naturally parasitized cats. Am J Vet Res 1987; 48: 670–72 [PubMed] [Google Scholar]
- 108. Kirkpatrick CE, Megella C. Use of ivermectin in treatment of Aelurostrongylus abstrusus and Toxocara cati infections in a cat. J Am Vet Med Assoc 1987; 190; 1309–10. [PubMed] [Google Scholar]
- 109. Burgu A, Sarimehmetoglu O. Aelurostrongylus abstrusus infection in two cats. Vet Rec 2004; 154: 602–4 [DOI] [PubMed] [Google Scholar]
- 110. Grandi G, Calvi LE, Venco L, et al. Aelurostrongylus abstrusus (cat lungworm) infection in five cats from Italy. Vet Parasitol 2005; 134: 177–82 [DOI] [PubMed] [Google Scholar]
- 111. Sherding RG. Parasites of the lung. In: King LG, ed. Textbook of respiratory disease in dogs and cats. St Louis: Saunders, 2004: 548–59. [Google Scholar]
- 112. Traversa D, Di Cesare A, Milillo P, et al. Efficacy and safety of imidacloprid 10%/moxidectin 1% spot-on formulation in the treatment of feline aelurostrongylosis. Parasitol Res 2009; 105 (suppl 1): S55–S62. [DOI] [PubMed] [Google Scholar]
- 113. Traversa D, Milillo P, Di Cesare A, et al. Efficacy and safety of emodepside 2.1%/praziquantel 8.6% spot-on formulation in the treatment of feline aelurostrongylosis. Parasitol Res 2009; 105 (suppl 1): S83–S89. [DOI] [PubMed] [Google Scholar]
- 114. Reinhardt S, Ottenjann M, Schunack B, Kohn B. [Lungworm infestation (Aelurostrongylus abstrusus) in a cat.] Kleintierpraxis 2004; 49: 239–46 German. [Google Scholar]
- 115. Milstein TC, Goldsmid JM. Parasites of feral cats from southern Tasmania and their potential significance. Aust Vet J 1997; 75: 218–19 [DOI] [PubMed] [Google Scholar]
- 116. Holmes PR, Kelly JD. Capillaria aerophila in the domestic cat in Australia. Aust Vet J 1973; 49: 472–73 [DOI] [PubMed] [Google Scholar]
- 117. Traversa D, Di Cesare A, Milillo P, Iorio R, Otranto D. Infection by Eucoleus aerophilus in dogs and cats: is another extra-intestinal parasitic nematode of pets emerging in Italy? Res Vet Sci 2009; 87: 270–72 [DOI] [PubMed] [Google Scholar]
- 118. Lalosevic D, Lalosevic V, Klem I, Stanojev-Jovanovic D, Pozio E. Pulmonary capillariasis miming bronchial carcinoma. Am J Trop Med Hyg 2008; 78: 14–16 [PubMed] [Google Scholar]
- 119. Barrs VR, Martin P, Nicoll RG, Beatty JA, Malik R. Pulmonary cryptococcosis and Capillaria aerophila infection in an FIV-positive cat. Aust Vet J 2000; 78: 154–58 [DOI] [PubMed] [Google Scholar]
- 120. Parker GA, Langloss JM, Dubey JP, Hoover EA. Pathogenesis of acute toxoplasmosis in specific-pathogen-free cats. Vet Pathol 1981; 18: 786–803 [DOI] [PubMed] [Google Scholar]
- 121. Feeney DA, Sautter JH, Lees GE. An unusual case of acute disseminated toxoplasmosis in a cat. J Am Anim Hosp Assoc 1981; 17: 311–14 [Google Scholar]
- 122. Dubey JP, Carpenter JL. Histologically confirmed clinical toxoplasmosis in cats: 100 cases (1952–1990). J Am Vet Med Assoc 1993; 203: 1556–66 [PubMed] [Google Scholar]
- 123. Sardinas JC, Chastain CB, Collins BK, Schmidt D. Toxoplasma pneumonia in a cat with incongruous serological test results. J Small Anim Pract 1994; 35: 104–7 [Google Scholar]
- 124. Eddlestone SM, Hoskins JD, Hosgood G, Dubey JP. Take the Compendium challenge. [Concurrent Haemobartonella felis and Toxoplasma gondii infection in a cat.] Compend Contin Educ Pract Vet 1996; 18: 774–79 [Google Scholar]
- 125. Foster SF, Charles JA, Canfield PJ, Beatty JA, Martin P. Reactivated toxoplasmosis in a FIV-positive cat. Aust Vet Pract 1998; 28: 159–63 [Google Scholar]
- 126. Poitout F, Weiss DJ, Dubey JP. Lung aspirate from a cat with respiratory distress. Vet Clin Pathol 1998; 27: 10, 21–22. [DOI] [PubMed] [Google Scholar]
- 127. Litster AL, Mitchell G, Menrath V. Pulmonary toxoplasmosis in a FIV-negative cat. Aust Vet Pract 1999; 29: 154–58 [Google Scholar]
- 128. Bernsteen L, Gregory CR, Aronson LR, Lirtzman RA, Brummer DG. Acute toxoplasmosis following renal transplantation in three cats and a dog. J Am Vet Med Assoc 1999; 215: 1123–26 [PubMed] [Google Scholar]
- 129. Brownlee L, Sellon RK. Diagnosis of naturally occurring toxoplasmosis by bronchoalveolar lavage in a cat. J Am Anim Hosp Assoc 2001; 37: 251–55 [DOI] [PubMed] [Google Scholar]
- 130. Last RD, Suzuki Y, Manning T, Lindsay D, Galipeau L, Whitbread TJ. A case of fatal systemic toxoplasmosis in a cat being treated with cyclosporin A for feline atopy. Vet Dermatol 2004; 15: 194–98 [DOI] [PubMed] [Google Scholar]
- 131. Anfray P, Bonetti C, Fabbrini F, Magnino S, Mancianti F, Abramo F. Feline cutaneous toxoplasmosis: a case report. Vet Dermatol 2005; 16: 131–36 [DOI] [PubMed] [Google Scholar]
- 132. Barrs VR, Martin P, Beatty JA. Antemortem diagnosis and treatment of toxoplasmosis in two cats on cyclosporin therapy. Aust Vet J 2006; 84: 30–35 [DOI] [PubMed] [Google Scholar]
- 133. Derouin F, Sarfati C, Beauvais B, Iliou MC, Dehen L, Lariviere M. Laboratory diagnosis of pulmonary toxoplasmosis in patients with acquired immunodeficiency syndrome. J Clin Microbiol 1989; 27: 1661–63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Nash G, Kerschmann RL, Herndier B, Dubey JP. The pathological manifestations of pulmonary toxoplasmosis in the acquired immunodeficiency syndrome. Hum Pathol 1994; 25: 652–58 [DOI] [PubMed] [Google Scholar]
- 135. Davidson MG, Lappin MR, Rottman JR, et al. Paradoxical effect of clindamcin in experimental, acute toxoplasmosis in cats. Antimicrob Agents Chemother 1996; 40: 1352–59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Pfefferkorn ER, Nothnagel RF, Borotz SE. Parasiticidal effect of clindamycin on Toxoplasma gondii grown in cultured cells and selection of a drug-resistant mutant. Antimicrob Agents Chemother 1992; 36: 1091–96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Harris C, Salgo MP, Tanowitz HB, Wittner M. In vitro assessment of antimicrobial agents against Toxoplasma gondii. J Infect Dis 1988; 157: 14–22 [DOI] [PubMed] [Google Scholar]
- 138. Fichera ME, Bhopale MK, Roos DS. In vitro assays elucidate peculiar kinetics of clindamycin action against Toxoplasma gondii . Antimicrob Agents Chemother 1995; 39: 1530–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Lappin MR, Greene CE, Winston S, Toll SL, Epstein ME. Clinical feline toxoplasmosis. Serologic diagnosis and therapeutic management of 15 cases. J Vet Intern Med 1989; 3: 139–43 [DOI] [PubMed] [Google Scholar]
- 140. Beatty JA, Swift N, Foster DJ, Barrs VR. Suspected clindamycin-associated oesophageal injury in cats: five cases. J Feline Med Surg 2006; 8: 412–419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Dubey JP, Lappin MR. Toxoplasmosis and neosporosis. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier, 2006: 754–75. [Google Scholar]
- 142. MacKay RJ, Granstrom DE, Saville WJ, Reed SM. Equine protozoal myeloencephalitis. Vet Clin North Am Equine Pract 2000; 16: 405–25 [DOI] [PubMed] [Google Scholar]
- 143. Lindsay DS, Rippey NS, Blagburn BL. Treatment of acute Toxoplasma gondii infections in mice with diclazuril or a combination of diclazuril and pyrimethamine. J Parasitol 1995; 81: 315–18 [PubMed] [Google Scholar]
- 144. Haberkorn A. Chemotherapy of human and animal coccidioses: state and perspectives. Parasitol Res 1996; 82: 193–99 [DOI] [PubMed] [Google Scholar]
- 145. Bowman DD, Hendrix CM, Lindsay DS, Barr SC. Trematodes of the lungs. In: Feline clinical parasitology. Iowa: Iowa State University Press, 2002: 163–78. [Google Scholar]
- 146. Ming-Sheng Z, Yan-Xia Z, Ming-Hua S, et al. [Epidemiologic survey of paragonimasis in reservoir region of South-North Water Diversion Project.] Zhongguo Bingyuan Shengwuxue Zazhi 2007; 2: 256, 259. Chinese, English abstract. [Google Scholar]
- 147. ChengQuan W, ShaoJin Y, DaJun W, et al. [Investigation on the etiology of Paragonimiasis skrjabini in Xingshan County locating the Reservoir Area of Three Gorges.] Zhongguo Bingyuan Shengwuxue Zazhi 2009; 4: 36–39 Chinese, English abstract. [Google Scholar]
- 148. Cao WJ, He LY, Zhong HL, et al. Paragonimiasis: treatment with praziquantel in 40 human cases and in 1 cat. Arzneimittelforschung 1984; 34: 1203–4 [PubMed] [Google Scholar]
- 149. Miyazaki I, Vajrasthira S. Occurrence of the lung fluke Paragonimus heterotremus Chen et Hsia 1964, in Thailand. J Parasitol 1967; 53: 207 [PubMed] [Google Scholar]
- 150. Uga S, Matsumara T, Yamada T, Onishi T, Goto M, Kagei N. [On the natural infection of Paragonimus spp in cats in Hyogo Prefecture.] Jpn J Parasitology 1983; 32: 333–39 Japanese, English abstract. [Google Scholar]
- 151. Voelker J, Sachs R. Morphology of the lung fluke Paragonimus uterobilateralis occurring in Gabon, West Africa. Trop Med Parasitol 1985; 36: 210–12 [PubMed] [Google Scholar]
- 152. Hu W. [Studies on the life cycle of Paragonimus heterotremus.] Chin J Parastiol Parasit Dis 1998; 16: 347–52 Chinese, English abstract. [PubMed] [Google Scholar]
- 153. Ho LY, Chung HL, Cheng LT, Ts'ao WC, Chao SL. [Further observations on Paragonimus yunnanensis Ho, Chung et al 1959.] Acta Zoologica Sinica 1973: 19: 245–56 Chinese, English abstract. [Google Scholar]
- 154. Lee SH, Koo KH, Chai JY, Hong ST, Sohn WM. [Experimental infection of Paragonimus iloktsuenensis in albino rats, dogs and cats]. Korean J Parasitol 1989; 27: 197–202 Korean. [DOI] [PubMed] [Google Scholar]
- 155. Li YS, Chen YZ, Lin CX, et al. [Occurrence of Paragonimus veocularis in Fujian Province.] Chinese J Parastiol Parasit Dis 2000; 18: 296–300 Chinese, English abstract. [PubMed] [Google Scholar]
- 156. Snider TA, Confer AW, Payton ME. Pulmonary histopathology of Cytauxzoon felis infections in the cat. Vet Pathol 2010; 47: 698–702 [DOI] [PubMed] [Google Scholar]
- 157. Greene CE, Meinkoth J, Kocan AA. Cytauxzoonosis. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier; 2006: 716–22. [Google Scholar]
- 158. Atkins C. Heartworm disease. In: Ettinger SJ, Feldman EC, eds. Textbook of veterinary internal medicine. 7th edn. St Louis: Saunders Elsevier, 2010: 1353–80. [Google Scholar]
- 159. Norris CR. Fungal pneumonia. In: King LG, ed. Textbook of respiratory disease in dogs and cats. St Louis: Saunders, 2004: 446–56. [Google Scholar]
- 160. Holzworth J. Mycotic diseases. In: Holzworth J, ed. Diseases of the cat. Philadelphia: Saunders, 1987: 320–58. [Google Scholar]
- 161. Davies C, Troy GC. Deep mycotic infections in cats. J Am Anim Hosp Assoc 1996; 32: 380–91 [DOI] [PubMed] [Google Scholar]
- 162. Padhye AA, Amster RL, Browning M, Ewing EP. Fatal encephalitis caused by Ochroconis gallopavum in a domestic cat (Felis domesticus). J Med Vet Mycol 1994; 32: 141–45 [PubMed] [Google Scholar]
- 163. Pawloski DR, Brunker JD, Singh K, Sutton DA. Pulmonary Paecilomyces lilacinus infection in a cat. J Am Anim Hosp Assoc 2010; 46: 197–202 [DOI] [PubMed] [Google Scholar]
- 164. Evans N, Gunew M, Marshall R, Martin P, Barrs V. Focal pulmonary granuloma caused by Cladophialophora bantiana in a domestic short haired cat. Med Mycol 2011; 49: 194–97 [DOI] [PubMed] [Google Scholar]
- 165. Malik R, Wigney DI, Muir DB, Gregory DJ, Love DN. Cryptococcosis in cats: clinical and mycological assessment of 29 cases and evaluation of treatment using orally administered fluconazole. J Med Vet Mycol 1992; 30: 133–44 [DOI] [PubMed] [Google Scholar]
- 166. Malik R, Krockenberger M, O'Brien CR, Martin P, Wigney D, Medleau L. Cryptococcosis. In: Greene CE, ed. Infectious diseases of the dog and cat. St Louis: Saunders Elsevier, 2006: 584–98. [Google Scholar]
- 167. Greene CE. Histoplasmosis. In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd edn. St Louis: Saunders Elsevier, 2006: 577–84. [Google Scholar]
- 168. Love DN, Baker KD. Sudden death in kittens associated with a feline picornavirus. Aust Vet J 1972; 48: 643 [DOI] [PubMed] [Google Scholar]
- 169. Pesavento PA, MacLachlan NJ, Dillard-Telm L, Grant CK, Hurley KF. Pathologic, immunohistochemical, and electron microscopic findings in naturally occurring virulent systemic feline calicivirus infection in cats. Vet Pathol 2004; 41: 257–63 [DOI] [PubMed] [Google Scholar]
- 170. Love DN. Feline herpesvirus associated with interstitial pneumonia in kitten. Vet Rec 1971; 89: 178–181 [DOI] [PubMed] [Google Scholar]
- 171. Malik R, Lessels NS, Webb S, et al. Treatment of feline herpesvirus-1 associated disease in cats with famciclovir and related drugs. J Feline Med Surg 2009; 11: 40–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172. Johnson MS, Martin M, Stone B, Hetzel U, Kipar A. Survival of a cat with pneumonia due to cowpox virus and feline herpesvirus infection. J Small Anim Pract 2009; 50: 498–502 [DOI] [PubMed] [Google Scholar]
- 173. Hinrichs U, van de Poel H, van den Ingh TS. Necrotizing pneumonia in a cat caused by an orthopox virus. J Comp Pathol 1999; 121: 191–96 [DOI] [PubMed] [Google Scholar]
- 174. Schöniger S, Chan DL, Hollinshead M, Humm K, Smith GL, Beard PM. Cowpox virus pneumonia in a domestic cat in Great Britain. Vet Rec 2007; 160: 522–23 [DOI] [PubMed] [Google Scholar]
- 175. Schulze C, Alex M, Schirrmeier H, et al. Generalized fatal Cowpox virus infection in a cat with transmission to a human contact case. Zoonoses Public Health 2007; 54: 31–37 [DOI] [PubMed] [Google Scholar]
- 176. World Health Organization. Avian influenza A(H5N1) — update 28: reports of infection in domestic cats (Thailand), Situation (human) in Thailand, Situation (poultry) in Japan and China. World Health Organization (WHO) Global Alert and Response (GAR), 20 Feb 2004. www.who.int/csr/don/2004_02_20/en/index.html.
- 177. Klopfleisch R, Wolf PU, Uhl W, et al. Distribution of lesions and antigen of highly pathogenic avian influenza virus A/Swan/Germany/R65/06 (H5N1) in domestic cats after presumptive infection by wild birds. Vet Pathol 2007; 44: 261–68 [DOI] [PubMed] [Google Scholar]
- 178. Marschall J, Hartmann K. Avian influenza A H5N1 infections in cats. J Feline Med Surg 2008; 10: 359–65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Thiry E, Addie D, Belák S, et al. H5N1 avian influenza in cats. J Feline Med Surg 2009; 11: 615–18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180. Löhr CV, DeBess EE, Baker RJ, et al. Pathology and viral antigen distribution of lethal pneumonia in domestic cats due to pandemic (H1N1) 2009 influenza A virus. Vet Pathol 2010; 47: 378–86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181. Sponseller BA, Strait E, Jergens A, et al. Influenza A pandemic (H1N1) 2009 virus infection in domestic cat. Emerg Inf Dis 2010; 16: 534–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182. Hooper PT, Westbury HA, Russell GM. The lesions of experimental equine morbillivirus disease in cats and guinea pigs. Vet Pathol 1997; 34: 323–29 [DOI] [PubMed] [Google Scholar]
- 183. Middleton DJ, Westbury HA, Morrissy CJ, et al. Experimental Nipah virus infection in pigs and cats. J Comp Pathol 2002; 126: 124–36 [DOI] [PubMed] [Google Scholar]
- 184. Cadoré JL, Steiner-Laurent S, Greenland T, Mornex JF, Loire R. Interstitial lung disease in feline immunodeficiency virus (FIV) infected cats. Res Vet Sci 1997; 62: 287–88 [DOI] [PubMed] [Google Scholar]
- 185. Hawkins EC, Kennedy-Stoskopf S, Levy JK, et al. Effect of FIV infection on lung inflammatory cell populations recovered by bronchoalveolar lavage. Vet Immunol Immunopathol 1996; 51; 21–28. [DOI] [PubMed] [Google Scholar]
- 186. German AC, Harbour DA, Helps CR, Gruffydd-Jones TJ. Is feline foamy virus really apathogenic? Vet Immunol Immunopathol 2008; 123: 114–18 [DOI] [PubMed] [Google Scholar]
- 187. Brady CA. Bacterial pneumonia in dogs and cats. In: King LG, ed. Textbook of respiratory disease in dogs and cats. St Louis: Saunders, 2004: 412–21. [Google Scholar]