Abstract
The goal of the present study was to investigate the functional implications of sexual dimorphism in the pattern of transporters along the rodent nephron as reported by Veiras et al. (J Am Soc Nephrol 28: 3504–3517, 2017). To do so, we developed sex-specific computational models of water and solute transport along the superficial nephrons from male and female rat kidneys. The models account for the sex differences in the abundance of apical and basolateral transporters, single nephron glomerular filtration rate, and tubular dimensions. Model simulations predict that ~70% and 60% of filtered Na+ is reabsorbed by the proximal tubule of male and female rat kidneys, respectively. The lower fractional Na+ reabsorption in female kidneys is due primarily to their smaller transport area, lower Na+/H+ exchanger activity, and lower claudin-2 abundance, culminating in significantly larger fractional delivery of water and Na+ to the downstream nephron segments in female kidneys. Conversely, the female distal nephron exhibits a higher abundance of key Na+ transporters, including Na+-K+-Cl− cotransporters, Na+-Cl− cotransporters, and epithelial Na+ channels. The higher abundance of transporters accounts for the enhanced water and Na+ transport along the female, relative to male, distal nephron, resulting in similar urine excretion between the sexes. Consequently, in response to a saline load, the Na+ load delivered distally is greater in female rats than male rats, overwhelming transport capacity and resulting in higher natriuresis in female rats.
Keywords: epithelial transport, sex differences
INTRODUCTION
In the past few years, an explosion of data has emerged concerning sex differences in kidney structure and function (22, 24). The kidney mass of a female rat is approximately one-half that of an age-matched male rat (19, 24). The glomerulus population is similar in both sexes (22). The single nephron glomerular filtration rate (SNGFR) in female rat kidneys is lower than that in male rat kidneys (22, 23), whereas urinary output is not substantially different between the sexes (24). Given these morphological and hemodynamic differences between the sexes, how does nephron transport differ between the sexes to yield similar urine output? In addition, how do these differences reflect the reserve renal capacity that female rats require to face challenges in pregnancy and lactation?
Veiras et al. (31) compared the abundance, covalent modifications, and regulators of electrolyte transporters, channels, and claudins, collectively referred to as transporters, in male and female rodents (31) and reported sexually dimorphic patterns in transporter abundance (see Fig. 1). Their findings demonstrated that, compared with male rat nephrons, female rat nephrons exhibit, in the proximal tubule, greater Na+/H+ exchanger 3 (NHE3) phosphorylation a higher distribution of NHE3 at the base of the microvilli, and lower abundance of Na+-Pi cotransporter 2 (NaPi2), aquaporin-1 (AQP1), and claudin-2. These differences are associated with lower Na+ and reabsorption and increased volume flow from the proximal tubule (31). In contrast, transporters along the distal nephrons exhibit higher abundance of total and phosphorylated Na+-Cl− cotransporter (NCC), claudin-7, and cleaved forms of epithelial Na+ channel (ENaC) α- and γ-subunits (31). These differences are associated with a lower fasting plasma K+ concentration in female compared with male rats. No correlation was found between transporter abundance and the estrous cycle stage (31). A caveat is that the above findings primarily indicate sex differences in transporter abundance or pool size, which are not assumed to directly translate into transporter activity that requires localization to the plasma membrane in an active configuration, for example, associated with phosphorylation [for apical Na+-K+-Cl− cotransporter isoform 2 (NKCC2) and NCC], dephosphorylation (NHE3), or cleavage (ENaC subunits). However, Fig. 1 shows that the relative abundance of total and phosphorylated forms of NHE3, NCC, and NKCC2 in female rats are greater by a similar amount versus these forms in male rats (defined as 1), supporting the use of relative abundance.
Fig. 1.
Profile of renal transporters along the female nephron plotted relative to mean male abundance defined as 1. NHE3, Na+/H+ exchanger isoform 3; NaPi2, Na+-Pi cotransporter 2; NKA α1, Na+-K+-ATPase α1-catalytic subunit; AQP, aquaporin water channel subunit; NKCC2, Na+-K+-Cl− cotransporter isoform 2; NCC, Na+-Cl− cotransporter; pS71 and -pT53, phosphorylation sites associated with activation; ENaC, epithelial Na+ channel; fl, full-length form; cl, cleaved forms; ROMK, renal outer medullary K+ channel. For the box and whisker plots, the ends of the box are the upper and lower quartiles such that the box spans the interquartile range, the median is marked by the vertical line inside the box, and the whiskers outside the box extend to the highest and lowest observations. The nephron cartoon was loosely adapted from Ref. 20.
The principal goal of the present study was to assess the extent to which individual sex-specific differences, in morphology or transporter activities, contribute to the observed differences in solute and water transport along the nephron in male versus female rats. A comprehensive understanding of the implications of these sex differences would allow us to explain the different male and female responses to physiological, pathophysiological, and pharmaceutical challenges. To accomplish this goal, we formulated sex-specific computational models of renal epithelial transport and conducted simulations to predict solute and water transport along the superficial nephron of the male and female rat kidney under physiological, pathophysiological, and artificial conditions. Existing epithelial transport models have, until recently, been built exclusively for male animals (e.g., Refs. 9–12, 14–16, 32, and 33). To analyze epithelial transport in both sexes of the rat, Li et al. (17) developed the first sex-specific computational epithelial transport models for the proximal convoluted tubule of the rat. Model simulations predicted that the substantially lower fractional volume reabsorption in the female proximal tubule (about one-half of the male proximal tubule) can be attributed to their smaller transport area and lower AQP1 expression level. Here, we extend the sex-specific proximal convoluted tubule models (17) to represent superficial nephrons of male and female rat kidneys.
METHODS
We followed the approach of Li et al. (17) and developed sex-specific epithelial cell-based models of a superficial nephron of the male and female rat kidney. The models represent functionally distinct segments as follows: proximal convoluted tubule, proximal straight tubule (S3 segment), descending limb, thick ascending limb, distal convoluted tubule, connecting tubule, and collecting duct. Each nephron segment is represented as a rigid tubule lined by a layer of epithelial cells, with apical and basolateral transporters that vary according to cell type (Fig. 2). The model accounts for the following 15 solutes: Na+, K+, Cl−, , H2CO3, CO2, NH3, , , , H+, , H2CO2, urea, and glucose. The models account for sex differences in the expression levels of apical and basolateral transporters (31), in SNGFR (22, 23), and in tubular dimensions (24). The model is formulated for the steady state and predicts luminal fluid flow, hydrostatic pressure, luminal fluid solute concentrations and, with the exception of the descending limb segment, cytosolic solute concentrations, membrane potential, and transcellular and paracellular fluxes.
Fig. 2.
Schematic diagram of the superficial nephron. The model accounts for the transport of water and 15 solutes. The diagram displays only the main Na+, K+, and Cl− transporters. PCT, proximal convoluted tubule; mTAL, medullary thick ascending limb; cTAL, cortical thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CCD, cortical collecting duct; OMCD, outer medullary collecting duct; IMCD, inner medullary collecting duct. Reprinted from Ref. 16.
RESULTS
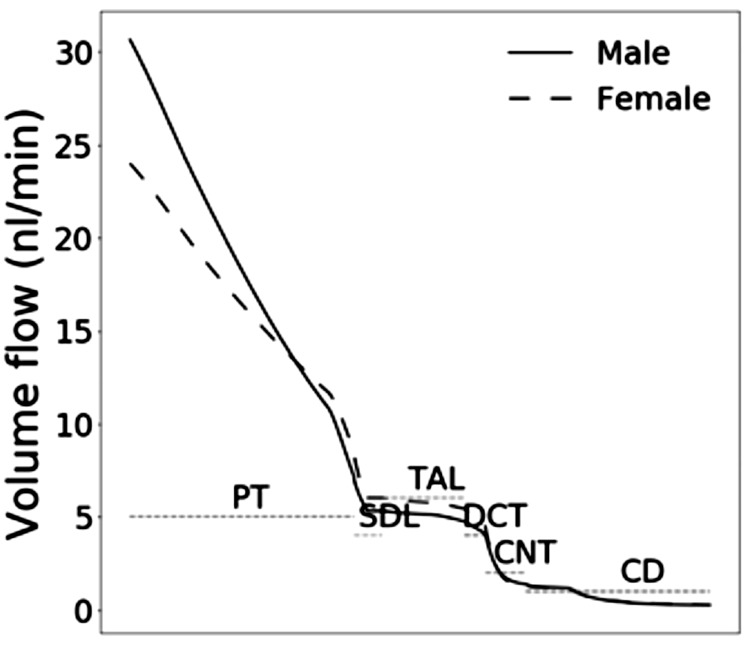
Using baseline parameters, we computed steady-state solutions to the model equations. Figure 3 shows the delivery of key solutes [Na+, K+, Cl−, , , urea, and titratable acid (TA)] and fluid to the inlets of individual nephron segments predicted by the male and female nephron models. Recall that SNGFR in the female nephron is assumed to be 80% that of the male nephron (24 vs. 30 nL/min). Solute and fluid transport along individual nephron segments is shown in Fig. 4; accumulated transport along the nephron is shown in Fig. 5.
Fig. 3.
Delivery of key solutes (A–G) and fluid (H) to the beginning of individual nephron segments in male and female rats. PT, proximal tubule; SDL, short descending limb; mTAL, medullary thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CCD, cortical collecting duct; TA, titratable acid. Insets: reproductions of distal segment values.
Fig. 4.
Net transport of key solutes (A–G) and fluid (H) along individual nephron segments, in male and female rats. Transport is taken positive out of a nephron segment. PT, proximal tubule; SDL, short descending limb; TAL, thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CD, collecting duct; TA, titratable acid. Insets: reproductions of distal segment values.
Fig. 5.
Accumulative transport of key solutes (A–G) and fluid (H) up to and including differing nephron segments in male and female rats. PT, proximal tubule; SDL, short descending limb; TAL, thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CD, collecting duct; TA, titratable acid.
The model predicts that proximal tubules of male and female nephrons reabsorb 70% and 57%, respectively, of filtered Na+, primarily via NHE3 and Na+-K+-ATPase, and 68% and 54% of filtered Cl−. The fractional reabsorption values are substantially lower in female nephrons because of their smaller transport area and lower NHE3 activity. The majority of the remaining Na+ and Cl− is reabsorbed along the thick ascending limbs. Net Na+ and Cl− transport is predicted to be 9.3% and 7.3% higher, respectively, in the model female thick ascending limb, despite its smaller transport area, because of the assumption of higher NKCC2, KCC, and Na+-K+-ATPase activities. Furthermore, the lower NHE3 activity in the proximal tubule in female nephrons does not extend to the thick ascending limb. As a result, some of the filtered that escapes the proximal tubule is reabsorbed by the thick ascending limb. Urine excretion fractions are predicted to be 0.9% and 0.7% for Na+ and Cl−, respectively, in male proximal tubules and 1% and 0.4% for Na+ and Cl−, respectively, in female proximal tubules. Urine Na+ excretion is comparable in male and female kidneys; Na+ excretion fraction differs between the sexes because of the lower filtration rate in female kidneys (Fig. 5).
Similarly, the model predicts that 69% and 54% of filtered K+ is reabsorbed along the male and female proximal tubules, respectively; the majority of the remaining K+ is also reabsorbed along the thick ascending limb. It is noteworthy that the model predicts a higher Na+ flow but lower K+ flow in the short descending limb in female versus male kidneys, even though Na+ and K+ reabsorption is in tandem. This is because plasma K+ concentration is assumed to be lower in female (3.9 mM) than male (4.9 mM) kidneys (31), which implies lower filtered K+ in female kidneys. Downstream of Henle's loop, the model distal convoluted tubules and connecting tubules are assumed to vigorously secrete K+. Consequently, 16% and 24% of the filtered load of K+ is excreted in urine in male and female rats, respectively. In both sexes, the proximal tubule is a major site of secretion via substitution of H+ in the NHE3 transporter. In contrast, a substantial fraction of is reabsorbed along the thick ascending limbs by substituting for K+ in the Na+-K+-2Cl− cotransporter.
The model predicts that 76% and 64% of the filtered volume is reabsorbed along the proximal tubules in male and female kidneys, respectively (Fig. 4H). Water reabsorption in the proximal tubule also drives paracellular urea reabsorption in that segment (Fig. 4F). More water is reabsorbed downstream, albeit at a slower rate. The model represents a kidney in antidiuretic state, with an inner medullary collecting duct that is highly urea and water permeable. As a result, 40% and 52% of the filtered urea is reabsorbed along the collecting duct in male and female kidneys, respectively.
The tubular fluid concentrations of key solutes, pH, and osmolality are shown in Fig. 6, tubular fluid flow is shown in Fig. 7, and urine composition is shown in Table 1. Tubular fluid solute concentrations are predicted to be similar in the two sexes along the proximal segments, primarily because of the leaky epithelium, but begin to diverge downstream of the thick ascending limbs. Urinary Na+ concentration and K+ concentration are similar in both sexes, but urinary Cl− concentration is two times as high in male compared with female rats. That difference is made up for, to a large extent, by the higher urinary TA and urea in female rats. Veiras et al. (31) also reported higher TA excretion but did not measure urea. Urine flow is similar in male and female rats, predicted to be 0.26 and 0.27 nl·min−1·nephron−1, respectively, or ~0.94 μl·min−1·kidney−1, assuming 36,000 nephrons in both sexes.
Fig. 6.
Tubular fluid solute concentrations (A–G), pH (H), and osmolality (I) in male and female rats. Tubular fluid solute concentrations are similar in the two sexes along the proximal segments but diverge downstream of the thick ascending limbs. PT, proximal tubule; SDL, short descending limb; TAL, thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CD, collecting duct; TA, titratable acid. Brackets denote concentration.
Fig. 7.
Tubular fluid flow in male and female rats. PT, proximal tubule; SDL, short descending limb; TAL, thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CD, collecting duct. The single nephron glomerular filtration rate in the female rat is 80% that of the male rat. However, proximal tubular outflow is slightly higher in female rats because of its lower water reabsorption. That excess fluid is reabsorbed along the distal segments so that urine output is similar in the two sexes.
Table 1.
Predicted urine output and composition
| Urine Values | Male | Female |
|---|---|---|
| Na+ concentration, mM | 150 | 139 |
| K+ concentration, mM | 91 | 82 |
| Cl− concentration, mM | 93 | 43 |
| Urea concentration, mM | 235 | 251 |
| pH | 5.7 | 5.4 |
| Osmolality, mosmol/kgH2O | 811 | 780 |
| Flow, nL/min/nephron | 0.26 | 0.27 |
Responses to Na+ load.
Veiras et al. (31) reported that female rats excrete an acute Na+ load faster than male rats. Specifically, given the same saline bolus volume relative to body weight, diuretic and natriuretic responses were more rapid in female rats, with female rats excreting ~30% and male rats <15% of the saline bolus at 3 h. Although not determined empirically, we assume that both sexes eventually excrete the excess saline and return to homeostasis. We conducted simulations to determine whether the baseline model transport parameters would predict the same sex difference. To simulate a saline load, we increase SNGFR by 50% in both male and female nephron models (1). All other parameters remained unchanged.
The enhanced filtered Na+ load increases proximal tubular Na+ by 50% in both male and female rats (Fig. 8A). The model predicts some degree of glomerulotubular balance (Fig. 8B). Despite the enhanced proximal reabsorption, Na+ delivery to the thick ascending limb in female and male rats is higher, by 100% and 93%, respectively, than baseline. Consequently, Na+ reabsorption along the thick ascending limb is also predicted to increase by 22% in both male and female rats. Water transport primarily follows Na+; the model predicts 25% and 19% increases in proximal tubular water reabsorption in male and female rats, respectively, during saline volume expansion.
Fig. 8.
Delivery of key solutes (A–G) and fluid (H) to the beginning of individual nephron segments in male and female rats. After a Na+ load, the single nephron glomerular filtration rate increases by 50%, resulting in elevated volume and solute deliveries to all nephron segments. Net urine volume and NaCl excretion are predicted to be similar in male and female rats, with higher fractional excretion rates in female rats. PT, proximal tubule; SDL, short descending limb; mTAL, medullary thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CCD, cortical collecting duct; TA, titratable acid. Insets: reproductions of distal segment values.
Na+ load is predicted to substantially impact K+ transport along the thick ascending limb and connecting tubule (Fig. 9B). In male and female rats, K+ reabsorption increases by 111% and 117%, respectively, along the thick ascending limb. K+ is secreted along the connecting tubule, and that secretion is predicted to increase markedly by 4.5- and 13-fold in male and female rats, respectively.
Fig. 9.
Net transport of key solutes (A–G) and fluid (H) along individual nephron segments in male and female rats. Transport is taken positive out of a nephron segment. After a Na+ load, the single nephron glomerular filtration rate and filtered load increase by 50%. Reabsorption of Na+ and Cl− increases in all segments, but primarily along the proximal tubule (PT). K+ reabsorption markedly increases along the PT and thick ascending limb (TAL), and K+ secretion by the connecting tubule (CNT) increases by severalfold. SDL, short descending limb; DCT, distal convoluted tubule; CD, collecting duct; TA, titratable acid. Insets: reproductions of distal segment values.
Taken together, the above transport processes result in urine flow of 3.9 and 3.4 nL/min, respectively, in male and female rats and urinary Na+ excretion of 932 and 882 pmol/min, respectively. Assuming that a female rat weighs about one-half that of the male rat (at 10−12 wk, Envigo.com), then the saline bolus volume simulated in the female model is one-half that of the male model. Thus, the female-to-male ratio of Na+ excretion (denoted ENa) as a fraction of saline load is given by (female ENa/male ENa)/(female saline volume/male saline volume) = (882/932)/0.5 = 1.89. Thus, the female model nephron predicted a fractional Na+ excretion, relative to the saline load, that was almost two times that of the male model nephron, consistent with findings by Veiras et al. (31). Similarly, the female-to-male ratio of fractional volume excretion has been predicted to be 1.74.
Sensitivity study.
A number of model parameters were assumed to differ between male and female rat nephrons, including size, SNGFR, and transporter activities. With these two sets of parameters, the models predict markedly different transport in some nephron segments, especially the proximal tubule, but similar urine outputs between the two sexes (see Figs. 3 and 4). To assess the effect of each of these sex differences, we conducted a sensitivity study in which we set individual model parameters in the female model to male values and computed urinary output. Key results are shown in Fig. 10.
Fig. 10.
Sensitivity of key model predictions to sex differences in parameters. Individual model parameters in the female model were set to male values. PCT, proximal convoluted tubule; S3, proximal straight tubule; NHE3, Na+/H+ exchanger isoform 3; Pf, transcellular water permeability; TAL, thick ascending limb; NKCC2, Na+-K+-Cl− cotransporter isoform 2; NKA, Na+-K+-ATPase; DCT, distal convoluted tubule; NCC, Na+-Cl− cotransporter; CNT, connecting tubule; ENaC, epithelial Na+ channel; CCD, cortical collecting duct; OMCD, outer medullary collecting duct; IMCD, inner medullary collecting duct; PK, apical K+ permeability; SNGFR, single nephron glomerular filtration rate.
To account for the fact that the female rat kidney is about one-half the size of the male kidney, we assume that nephron segmental lengths and radii in female kidneys are 80–85% that of male kidneys. To assess the impact of the much smaller effective transport area in the female nephron, we conducted a simulation in which we set the dimensions of female model nephron to male values. Other parameters remained unchanged. With a larger transport capacity, the female model predicted that urinary excretion of Na+, K+, and volume would decrease by 55%, 52%, and 55%, respectively, and that urine pH would decrease from its baseline value of 5.4–4.6 (a 15% reduction).
The baseline model assumes that SNGFR is 20% lower in the female model nephron compared with the male model nephron. When SNGFR of the female nephron model was set to 30 nL/min, as in the male nephron, with all other model parameters remaining at their baseline values, the model predicted that this enhanced SNGFR far exceeded the transport capacity of the female nephron. Consequently, urinary excretion of Na+, K+, and volume increased by 8.8-, 3.3-, and 5.0-fold, respectively.
Based on the findings by Veiras et al. (31), which indicated enhanced abundance of a number of Na+ transporters along the distal nephron segments in the female kidney, we assumed higher activities of these transporters in the female model. Those higher transporter activities compensate for the lesser transporter area in the female nephron. When these transporter activity levels were lowered to male levels, urinary outputs increased (see Fig. 10). Our results indicate that model results are particularly sensitive to activity levels of thick ascending limb NKCC2 and Na+-K+-ATPase. When NKCC2 activity was reduced to the male level, with other parameters unchanged, urinary excretion rates in the female model for Na+, K+, and volume were predicted by increase by 26%, 26%, and 27%, respectively, above baseline. When Na+-K+-ATPase activity along the thick ascending limb was reduced to the male level (again with other parameters and Na+-K+-ATPase activity along other nephron segments unchanged), Na+, K+, and volume increased by 44%, 44%, and 38%, respectively, above baseline. Lowering Na+-K+-ATPase activity to the male level along the distal convoluted tubule yielded a smaller effect: Na+, K+, and volume increased by 18%, 18%, and 16%, respectively, above baseline.
Taken together, the sensitivity results indicate that the lower filtrate rates and smaller effective transport area in the female nephron have major effects on urinary outputs. Our analysis further indicates that, for the female nephron model to generate urinary excretions similar to those in the male nephron model, a number of transporter activities must be adjusted in a highly coordinated manner.
DISCUSSION
The influence of sex hormones and chromosomes goes beyond the development of female- and male-specific traits and reproductive organs; sex hormones play an important role in regulating the structure and function of almost all mammalian organs and systems, including the kidneys. Indeed, in vivo and in vitro studies in experimental animals, as well as clinical observations, have revealed major sex differences in renal structure and functions under various conditions (21, 24, 27). For instance, sex hormones are known to regulate organic cation and anion transporters (18, 30) and Na+-dependent glucose cotransporter (SGLT)1 and SGLT2 (25, 26) in rodents. The recent study by Veiras et al. (31) adds to this knowledge by revealing sexually dimorphic patterns in transporter channels and claudin abundance in rodents, in terms of the abundance, covalent modifications, and regulators in rats and mice. What are the functional implications of these findings? The principal goal of the present study was to assess the effects of these known sex differences on nephron function. To that end, we developed the first and only sex-specific epithelial transport model of the rat nephron. The model was applied to analyze the functional implications of the markedly different hemodynamics, size, and renal transporter patterns observed between male and female rats (22, 24, 31). An understanding of these functional implications allows us to better explain the different responses observed between the sexes under physiological, pathophysiological, and pharmaceutical challenges.
The sexual dimorphism in renal Na+ and water transporters (31) indicates a marked sex difference in fractional Na+ and water transport along the proximal versus distal nephron segments. Along the proximal tubule, NHE3, the primary mediator of tubular Na+ reabsorption and H+ secretion, and a major driver of reabsorption, exhibits a higher degree of phosphorylation in female compared with male rats, which is a marker for localization to the base of the microvilli and lower NHE activity (2, 8). Another indicator of NHE3 activity is the rate of reabsorption along the proximal tubule, quantified as 35% lower in female than male rats (31). Because Na+ reabsorption drives water reabsorption, this finding suggests similarly lower fluid reabsorption along the proximal tubule, consistent with greater lithium clearance measured in female animals (31). Taken together, plentiful evidence points to lower fractional reabsorption along the proximal tubule in the female rat kidney. Consistent with these observations, the model predicts substantially lower reabsorption of Na+, K+, Cl−, , and water along the proximal tubule of the female rat compared with the male rat (see Figs. 4 and 5). The lower proximal tubule NHE3 activity may represent reserve capacity that can be increased significantly during pregnancy and lactation, during which both GFR and tubular transport are increased, and those increases can happen without raising blood pressure (22). It is noteworthy that the abundance of SGLT2 has been reported to be higher in female rats (26), resulting in similar reabsorption by the proximal convoluted tubule in both sexes (results not shown).
NHE3 activity contributes to the addition of TA to the tubular lumen. Taken in isolation, the lower NHE3 activity in female rats would result in lower TA excretion. In contrast, the model predicts that tubular TA flow is higher in all female nephron segments downstream of the proximal tubule and that urine pH is lower in female (5.4) compared with male (5.7) rats. In fact, these model predictions are consistent with measures of higher TA in overnight urine from fasting female versus male rats (31). This indicates alternative pathways for acidification may be stronger in the female proximal tubule. In particular, NaPi2 abundance is reported to be 25% lower in female rats. As a result, the female model predicts significantly lower reabsorption of phosphate, which is a major buffer of luminal H+. Another consequence is the slightly higher urinary phosphate excretion in female rats (20% higher than male rats), also consistent with assessment in overnight urines (31).
Despite the lower SNGFR in female rats (Table 2), their lower proximal tubule reabsorption results in slightly enhanced delivery to the distal nephron segments (see Fig. 3). Recall that the female nephron is shorter and narrower than the male nephron. Thus, to handle a transport load similar to male nephrons, the distal nephron segments in the female rat kidney must express a higher abundance of relevant transporters. Indeed, the distal nephrons of female rats have been found to have a higher abundance of key transporters, including total and phosphorylated NCC, claudin-7, and cleaved, i.e., activated, forms of ENaC α- and γ-subunits (31). The higher transporter abundance, and the presumably higher activities, in the female nephron offset the impact of its smaller transport area. Together, these two competing factors yield similar urine excretion rates in male and female rats. Furthermore, our sensitivity results indicate that the transporter abundance profiles in male and female rats differ in a coordinated manner, such that urinary output, as well as Na+, K+, and Cl− excretion, are all similar between the two sexes. That goal cannot be accomplished by any up- or downregulation of any single, or even a few, transporters.
Table 2.
Key parameter differences between male and female rat nephron models
| Parameter | Female-to-Male Ratio | Parameter | Female-to-Male Ratio |
|---|---|---|---|
| PCT | S3 | ||
| NHE3 activity | 0.83 | 0.83 | |
| NaPi2 | 0.75 | 0.75 | |
| PNa, PCl (paracellular) | 0.4 | 0.4 | |
| Pf (transcellular) | 0.64 | 0.64 | |
| SDL | |||
| PNa, PCl (transcellular) | 0.5 | ||
| Pf (transcellular) | 2 | ||
| mTAL | cTAL | ||
| NKCC2 activity | 2 | 2 | |
| KCC activity | 1.5 | 1.5 | |
| Na+-K+-ATPase activity | 2 | 2 | |
| Na+/H+ exchanger | 0.8 | 0.8 | |
| Na+- cotransporter | 0.8 | 0.8 | |
| Na+- cotransporter | 0.8 | 0.8 | |
| PNa, PCl (paracellular) | 0.9 | ||
| DCT | CNT | ||
| NCC activity | 1.6 | ||
| Na+-K+-ATPase activity | 2 | 2 | |
| Na+/H+ exchanger | 0.85 | 0.9 | |
| Na+- cotransporter | 0.85 | 0.9 | |
| ENaC activity | 2 | 1.3 | |
| PNa, PCl (paracellular) | 1.4 | 1.4 | |
| Pf (transcellular) | 2 | 1.5 | |
| CCD | OMCD | ||
| Na+-K+-ATPase activity | 1.5 | 1.1 | |
| H+-K+-ATPase activity | 1 | 1.5 | |
| Na+/H+ exchanger | 0.9 | 0.9 | |
| Na+- cotransporter | 0.9 | 0.9 | |
| ENaC activity | 1.5 | 1.2 | |
| PNa, PCl (paracellular) | 1.4 | 1 | |
| PK (apical) | 0.7 | 1.2 | |
| Pf (transcellular) | 2 | 2 | |
| IMCD | Morphology | ||
| Na+-K+-ATPase activity | 1.2 | PCT, S3 | |
| H+-K+-ATPase activity | 1.5 | Length | 0.8 |
| K+-Cl− cotransporter | 1.2 | Diameter | 0.8 |
| NaKCl2 activity | 0.6 | Distal segments | |
| PNa, PCl (paracellular) | 0.5 | Length | 0.85 |
| PK (apical) | 2 | Diameter | 0.85 |
| PNa (apical) | 0.5 | Hemodynamics | |
| Pf (transcellular) | 2 | SNGFR | 0.8 |
| Purea changing fraction | 0.89 |
NaPi2, Na+-Pi cotransporter 2; PNa, Na+ permeability; PCl, Cl− permeability; Pf, water permeability; SDL, short descending limb; NKCC2, Na+-K+-Cl− cotransporter isoform 2; KCC, K+-Cl− cotransporter; ENaC, epithelial Na+ channel; PK, K+ permeability; NaKCl2, Na+-K+-Cl− cotransporter isoform 1; Purea, urea permeability; PCT, proximal convoluted tubule; S3, proximal straight tubule; mTAL, thick ascending limb; cTAL, cortical thick ascending limb; DCT, distal convoluted tubule; CNT, connecting tubule; CCD, cortical collecting duct; OMCD, outer medullary collecting duct; IMCD, inner medullary collecting duct; SNGFR, single nephron glomerular filtration rate.
Diuretic and natriuretic responses have been reported to be more rapid in female rats (31). Our saline challenge simulations suggest similar increases in Na+ reabsorption along the model proximal tubules of both sexes when filtered Na+ is increased, provoking similar Na+ loads delivered to downstream nephron segments in both sexes. Indeed, urinary Na+ excretion was predicted to be similar in male and female rats. Note that, in these simulations, we assumed the same fractional increases in SNGFR and filtered Na+ in the two sexes. Thus, the similar urinary Na+ excretion in male and female rats implies that the fractional Na+ excretion in female rats is almost two times that of male rats. Because Na+ reabsorption drives water reabsorption, a similar result is predicted for fractional water excretion after a saline load; lower AQP1 and claudin 2 may conceivably contribute to the more rapid diuresis and natriuresis by blunting the rate of osmotic equilibration along the proximal tubule. These results suggest that the ability of the female rat to more rapidly excrete a saline load can be attributed primarily to the lower Na+ transporter activity in its proximal tubule.
A downstream shift in Na+ reabsorption in the female rat nephron likely has significant metabolic consequences. This is because it is more energetically favorable to reabsorb Na+ across the leaky proximal tubule epithelium, facilitated by the claudin 2 route, than along the downstream thick ascending limb, distal convoluted tubule, and collecting duct epithelia, which are tighter (7). Is there major sexual dimorphism in tubular metabolic demand in the rats? Also, a pregnant or lactating rat is faced with higher intakes, expanded circulatory volume, higher GFR, and reduced renal output and thus requires significantly altered tubular function to enhance reabsorption to support the growth of offspring while still maintaining pH and K+ homeostasis. That enhanced reabsorption would likely take place in the efficient proximal tubule but would, nevertheless, require additional energy. A computational model that can aide in understanding these scenarios must predict transport-driven oxygen consumption, similar to our published (male) nephron models (4–6, 13, 16).
The present models are developed for the rat. To what extent can model predictions be generalized to other mammals and, perhaps most importantly, to humans? It is noteworthy that Veiras et al. (31) have reported significant species-specific differences (rat vs. mouse) in sexual dimorphisms in renal transporter patterns. The differences between the rat kidney and the human kidney may be greater, considering that the male rat kidney is bigger than the female rat kidney, whereas the kidneys in men and women are much more similar in size. Taken in isolation, the similar kidney size in men and women, and thus tubular dimensions and transport areas, suggests that the sex differences in renal tubular transporter abundance may be significantly less pronounced in humans. On the other hand, like female rats, women also face the challenge of circulating volume adaptation during pregnancy and lactation, and the potential for increasing reabsorption along the proximal nephron coupled to the more abundant transporters in the distal nephron may facilitate that adaptation. Hence, although results obtained using a rat kidney model can shed light on human kidney function, caution must be exercised before translating these results to humans. That analysis may be facilitated by sex-specific computational models of solute and water transport along the human nephron, based on our recent published human model (11).
The present model represents a superficial nephron. In the rat, superficial nephrons account for two-thirds of the total nephron population; the remaining juxtamedullary nephrons have higher SNGFR and loops of Henle that reach the medulla. The output of the superficial nephron model likely does not accurately predict urinary output. To remedy that deficiency, the model must be extended to include a population of nephrons with different properties, including both the superficial nephron and a number of representative juxtamedullary nephrons that reach differing levels of the medulla (as done in Refs. 10 and 14). Moreover, SNGFR is assumed known a priori in the present model; thus, the model does not represent autoregulation. Given the sex differences in autoregulatory mechanisms, such as tubuloglomerular feedback (3), the extension of the present model to represent sex-specific autoregulation (following the approach in Refs. 8a, 28, and 29) would be a worthwhile study. Such elaborations of the female nephron model would allow investigations of the impact of pregnancy and lactation on function along the female nephron.
GRANTS
This work was supported by the Canada 150 Research Chair program (to A. T. Layton) and by National Institute of Diabetes and Digestive and Kidney Diseases Grants R01-DK-106102 (to A. T. Layton) and R01-K0-83785 (to A. A. McDonough).
DISCLOSURES
No author conflict of interest, financial or otherwise, is declared by the authors.
AUTHOR CONTRIBUTIONS
R.H. performed experiments; R.H., A.A.M., and A.T.L. analyzed data; R.H., A.A.M., and A.T.L. interpreted results of experiments; R.H. prepared figures; R.H. and A.T.L. drafted manuscript; R.H., A.A.M., and A.T.L. edited and revised manuscript; R.H., A.A.M., and A.T.L. approved final version of manuscript; A.T.L. conceived and designed research.
REFERENCES
- 1.Blantz RC, Singh P, Deng A, Thomson SC, Vallon V. Acute saline expansion increases nephron filtration and distal flow rate but maintains tubuloglomerular feedback responsiveness: role of adenosine A1 receptors. Am J Physiol Renal Physiol 303: F405–F411, 2012. doi: 10.1152/ajprenal.00329.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brasen JC, Burford JL, McDonough AA, Holstein-Rathlou NH, Peti-Peterdi J. Local pH domains regulate NHE3-mediated Na+ reabsorption in the renal proximal tubule. Am J Physiol Renal Physiol 307: F1249–F1262, 2014. doi: 10.1152/ajprenal.00174.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brown RD, Hilliard LM, Head GA, Jones ES, Widdop RE, Denton KM. Sex differences in the pressor and tubuloglomerular feedback response to angiotensin II. Hypertension 59: 129–135, 2012. doi: 10.1161/HYPERTENSIONAHA.111.178715. [DOI] [PubMed] [Google Scholar]
- 4.Fry BC, Edwards A, Layton AT. Impact of nitric-oxide-mediated vasodilation and oxidative stress on renal medullary oxygenation: a modeling study. Am J Physiol Renal Physiol 310: F237–F247, 2016. doi: 10.1152/ajprenal.00334.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fry BC, Edwards A, Layton AT. Impacts of nitric oxide and superoxide on renal medullary oxygen transport and urine concentration. Am J Physiol Renal Physiol 308: F967–F980, 2015. doi: 10.1152/ajprenal.00600.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fry BC, Edwards A, Sgouralis I, Layton AT. Impact of renal medullary three-dimensional architecture on oxygen transport. Am J Physiol Renal Physiol 307: F263–F272, 2014. doi: 10.1152/ajprenal.00149.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Klahr S, Hamm L, Hammerman M, Mandel L. Renal metabolism: integrated responses. In: Handbook of Physiology. Renal Physiology. Bethesda, MD: Am Physiol Soc, 1992, vol. II, chapt. 49, p. 2263–2334. [Google Scholar]
- 8.Kocinsky HS, Girardi AC, Biemesderfer D, Nguyen T, Mentone S, Orlowski J, Aronson PS. Use of phospho-specific antibodies to determine the phosphorylation of endogenous Na+/H+ exchanger NHE3 at PKA consensus sites. Am J Physiol Renal Physiol 289: F249–F258, 2005. doi: 10.1152/ajprenal.00082.2004. [DOI] [PubMed] [Google Scholar]
- 8a.Layton AT. Feedback-mediated dynamics in a model of a compliant thick ascending limb. Math Biosci 228: 185–194, 2010. doi: 10.1016/j.mbs.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Layton AT, Edwards A, Vallon V. Renal potassium handling in rats with subtotal nephrectomy: modeling and analysis. Am J Physiol Renal Physiol 314: F643–F657, 2018. doi: 10.1152/ajprenal.00460.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Layton AT, Laghmani K, Vallon V, Edwards A. Solute transport and oxygen consumption along the nephrons: effects of Na+ transport inhibitors. Am J Physiol Renal Physiol 311: F1217–F1229, 2016. doi: 10.1152/ajprenal.00294.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Layton AT, Layton HE. A computational model of epithelial solute and water transport along a human nephron. PLOS Comput Biol, 15: e1006108, 2019. doi: 10.1371/journal.pcbi.1006108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Layton AT, Vallon V. SGLT2 inhibition in a kidney with reduced nephron number: modeling and analysis of solute transport and metabolism. Am J Physiol Renal Physiol 314: F969–F984, 2018. doi: 10.1152/ajprenal.00551.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Layton AT, Edwards A, Vallon V. Adaptive changes in GFR, tubular morphology, and transport in subtotal nephrectomized kidneys: modeling and analysis. Am J Physiol Renal Physiol 313: F199–F209, 2017. doi: 10.1152/ajprenal.00018.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Layton AT, Vallon V, Edwards A. A computational model for simulating solute transport and oxygen consumption along the nephrons. Am J Physiol Renal Physiol 311: F1378–F1390, 2016. doi: 10.1152/ajprenal.00293.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Layton AT, Vallon V, Edwards A. Modeling oxygen consumption in the proximal tubule: effects of NHE and SGLT2 inhibition. Am J Physiol Renal Physiol 308: F1343–F1357, 2015. doi: 10.1152/ajprenal.00007.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Layton AT, Vallon V, Edwards A. Predicted consequences of diabetes and SGLT inhibition on transport and oxygen consumption along a rat nephron. Am J Physiol Renal Physiol 310: F1269–F1283, 2016. doi: 10.1152/ajprenal.00543.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li Q, McDonough AA, Layton HE, Layton AT. Functional implications of sexual dimorphism of transporter patterns along the rat proximal tubule: modeling and analysis. Am J Physiol Renal Physiol 315: F692–F700, 2018. doi: 10.1152/ajprenal.00171.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ljubojević M, Balen D, Breljak D, Kusan M, Anzai N, Bahn A, Burckhardt G, Sabolić I. Renal expression of organic anion transporter OAT2 in rats and mice is regulated by sex hormones. Am J Physiol Renal Physiol 292: F361–F372, 2007. doi: 10.1152/ajprenal.00207.2006. [DOI] [PubMed] [Google Scholar]
- 19.MacKay L, MacKay E. Factors which determine renal weight. III. Sex. Am J Physiol 83: 196–201, 1927. doi: 10.1152/ajplegacy.1927.83.1.196. [DOI] [Google Scholar]
- 20.Mullins LJ, Bailey MA, Mullins JJ. Hypertension, kidney, and transgenics: a fresh perspective. Physiol Rev 86: 709–746, 2006. doi: 10.1152/physrev.00016.2005. [DOI] [PubMed] [Google Scholar]
- 21.Mulroney SE, Woda C, Johnson M, Pesce C. Gender differences in renal growth and function after uninephrectomy in adult rats. Kidney Int 56: 944–953, 1999. doi: 10.1046/j.1523-1755.1999.00647.x. [DOI] [PubMed] [Google Scholar]
- 22.Munger K, Baylis C. Sex differences in renal hemodynamics in rats. Am J Physiol 254: F223–F231, 1988. doi: 10.1152/ajprenal.1988.254.2.F223. [DOI] [PubMed] [Google Scholar]
- 23.Remuzzi A, Puntorieri S, Mazzoleni A, Remuzzi G. Sex related differences in glomerular ultrafiltration and proteinuria in Munich-Wistar rats. Kidney Int 34: 481–486, 1988. doi: 10.1038/ki.1988.206. [DOI] [PubMed] [Google Scholar]
- 24.Sabolić I, Asif AR, Budach WE, Wanke C, Bahn A, Burckhardt G. Gender differences in kidney function. Pflugers Arch 455: 397–429, 2007. doi: 10.1007/s00424-007-0308-1. [DOI] [PubMed] [Google Scholar]
- 25.Sabolić I, Skarica M, Gorboulev V, Ljubojević M, Balen D, Herak-Kramberger CM, Koepsell H. Rat renal glucose transporter SGLT1 exhibits zonal distribution and androgen-dependent gender differences. Am J Physiol Renal Physiol 290: F913–F926, 2006. doi: 10.1152/ajprenal.00270.2005. [DOI] [PubMed] [Google Scholar]
- 26.Sabolic I, Vrhovac I, Eror DB, Gerasimova M, Rose M, Breljak D, Ljubojevic M, Brzica H, Sebastiani A, Thal SC, Sauvant C, Kipp H, Vallon V, Koepsell H. Expression of Na+-d-glucose cotransporter SGLT2 in rodents is kidney-specific and exhibits sex and species differences. Am J Physiol Cell Physiol 302: C1174–C1188, 2012. doi: 10.1152/ajpcell.00450.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sandberg K, Ji H. Sex and the renin angiotensin system: implications for gender differences in the progression of kidney disease. Adv Ren Replace Ther 10: 15–23, 2003. doi: 10.1053/jarr.2003.50006. [DOI] [PubMed] [Google Scholar]
- 28.Sgouralis I, Layton AT. Autoregulation and conduction of vasomotor responses in a mathematical model of the rat afferent arteriole. Am J Physiol Renal Physiol 303: F229–F239, 2012. doi: 10.1152/ajprenal.00589.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sgouralis I, Layton AT. Theoretical assessment of renal autoregulatory mechanisms. Am J Physiol Renal Physiol 306: F1357–F1371, 2014. doi: 10.1152/ajprenal.00649.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Urakami Y, Okuda M, Saito H, Inui K. Hormonal regulation of organic cation transporter OCT2 expression in rat kidney. FEBS Lett 473: 173–176, 2000. doi: 10.1016/S0014-5793(00)01525-8. [DOI] [PubMed] [Google Scholar]
- 31.Veiras LC, Girardi ACC, Curry J, Pei L, Ralph DL, Tran A, Castelo-Branco RC, Pastor-Soler N, Arranz CT, Yu ASL, McDonough AA. Sexual Dimorphic Pattern of Renal Transporters and Electrolyte Homeostasis. J Am Soc Nephrol 28: 3504–3517, 2017. doi: 10.1681/ASN.2017030295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Weinstein AM. A mathematical model of the rat kidney: K+-induced natriuresis. Am J Physiol Renal Physiol 312: F925–F950, 2017. doi: 10.1152/ajprenal.00536.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Weinstein AM. A mathematical model of the rat nephron: glucose transport. Am J Physiol Renal Physiol 308: F1098–F1118, 2015. doi: 10.1152/ajprenal.00505.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]