Abstract
Objective
To describe the main syndrome and clinical course in a large cohort of patients with anti–Ri-associated paraneoplastic neurologic syndrome (Ri-PNS).
Methods
Twenty-year retrospective nationwide study and systematic review of the literature.
Results
Thirty-six patients with complete clinical information were identified (median age 66 years, range: 47–87 years). In this French cohort, the majority were women (78%). At onset, 4 main patterns were observed: cerebellar syndrome (39%), isolated tremor (24%), oculomotor disturbances (17%), and other symptoms (19%). Course was multistep for 78% of cases. At the time the disease reached the plateau phase (median 12 weeks, range: 1–64 weeks; 28% >3 months), 24 (67%) showed an overt cerebellar syndrome, which was isolated in 3 patients, and was most frequently (21/24 cases) part of a multisystem neurologic disease. Patients manifested a variety of movement disorders, including myoclonus (33%), dystonia (17%), either cervical or oromandibular, and parkinsonism (17%). Most patients had cancer (92%), mainly breast cancer (n = 22). Misdiagnoses concerned 22% of patients (n = 8) and included atypical parkinsonism (n = 2), MS (n = 2), Bickerstaff encephalitis (n = 1), hyperekplexia (n = 1), vestibular neuritis (n = 1), and functional neurologic disorder (n = 1). Survival at 12 months was 73% (95% CI [0.54–0.85]), at 24 months 62% (95% CI [0.41–0.78]), and at 36 months 47% (95% CI [0.25–0.65]). There was no major clinical difference between cases retrieved from the systematic review of the literature (n = 55) and the French cohort.
Conclusions
Ri-PNS is a multisystem neurologic syndrome with prominent cerebellum/brainstem involvement. Opsoclonus-myoclonus is less common than expected, and the disorder can mimic neurodegenerative diseases.
Opsoclonus-myoclonus syndrome (OMS) is considered to be the stereotypical manifestation of a paraneoplastic neurologic syndrome associated with autoantibodies targeting the intracellular Ri antigen (Ri-PNS) in patients with breast cancer.1–8 Following the original descriptions of Ri-PNS, subsequent reports described diverse neurologic presentations (including patients with peripheral nervous system disorders)9 and different oncological associations (e.g., lung cancer).10 Disease course, differential diagnosis spectrum, and long-term outcome remain to be clarified.
To improve the recognition of this disease, we report and analyze herein the clinical features of previously unpublished patients with Ri-PNS resulting from a 20-year, retrospective, nationwide study at the “Centre de référence des syndromes neurologiques paranéoplasiques et encéphalites auto-immunes” (Lyon, France) and compare this French cohort with cases identified from a systematic review of the literature.
Methods
Patients
We retrospectively included all the patients identified between January 1, 1999, and December 31, 2018, whose serum and/or CSF were found to be positive for Ri antibodies. Positivity was defined by 2 complementary detection methods: staining of nucleus and cytoplasm of neurons by indirect immunofluorescence on rat brain sections and a positive confirmation test using a cell-based assay (immunodot, or Western blotting with recombinant protein), as recommended.11 Clinical data were obtained by reviewing the case records of first admission and serial follow-up examinations. Patients with insufficient information (i.e., lacking information on clinical presentation, cancer association, and paraclinical investigations) were excluded from the analysis. Outcomes were assessed using the modified Rankin Scale (mRS); this scale ranges from 0 (no symptoms) to 6 (death). All patients provided written informed consent for the storage and use of their serum, CSF, and clinical information for research purposes. The study was approved by the Institutional Review Board of the University Claude Bernard Lyon 1 and Hospices Civils de Lyon.
Search strategy
A systematic review of the literature was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines.12 We searched PubMed for records published in English or French language between January 1, 1988, and January 1, 2018, using the following search terms: (1) the text words “Anti-Ri”; (2) the MeSH term “paraneoplastic syndromes” AND the text words “ANNA-2” OR “NOVA 1” OR “NOVA2.” In addition, the reference lists of all selected articles were perused to identify any articles missed. The following data items were extracted from the studies: demographic data, neurologic symptoms, and their mode of onset (defined as acute if developing in <1 week; subacute: between 1 week and 3 months; progressive: >3 months), paraclinical data, and type of cancer.
Comparison between the literature and the French cohort of patients
We questioned whether the clinico-demographic features of the French cases were different from those of the patients reported in the literature. To perform a comparison between the 2 groups, we included only confirmed cases who did not present with coexisting autoantibodies to have a homogeneous series of patients with an identical biomarker.
Statistical methods
Data are presented as frequencies and percentages for categorical variables and as median and range for continuous variables. Categorical data were analyzed using the Fisher exact test (2 tailed) and numerical data using the Mann-Whitney U test. Survival rates were based on Kaplan-Meier estimation with 95% CI. Statistical analyses were performed using the SPSS Statistics software (IBM Corp., Armonk, NY). p Values <0.05 were considered significant.
Data availability
Data reported in this article are available within the article or its supplementary materials. More information regarding the data is available from the corresponding author on reasonable request.
Results
Clinical features
A total of 40 patients with Ri-PNS were identified. Four patients were excluded due to insufficient clinical information, leaving a series of 36 previously unpublished cases for analysis (table e-1, links.lww.com/NXI/A225). The median age at disease onset was 66 years (range: 47–87 years), and most patients were women (78%). As the majority (78%) of cases had a multistep disease course, we separated the neurologic features present at onset (figure 1A) from those that constituted the established syndrome (figure 1B).
Figure 1. Clinical features of Ri-PNS.
Clinical features at onset (A) and in the established syndrome (B) in the French Cohort of patients with Ri-PNS (n = 36). (C) Schematic representation of the complex association of neurologic signs in Ri-Ab–associated syndrome. The interactions between clinical phenomenologies in Ri-Ab–associated syndrome in a Circos plot31 with co-occurrence of cerebellar syndrome, oculomotor disturbances, movement disorders, and opsoclonus-myoclonus. (D) Differences in the clinical spectrum in progressive (violet) and acute/subacute (light blue) forms. LE = limbic encephalitis; Ri-Ab = Ri-antibody; Ri-PNS = Ri-associated paraneoplastic neurologic syndrome; SPS = stiff-person syndrome.
At disease onset, 4 main patterns were observed: cerebellar syndrome (n = 14, 39%), isolated tremor (n = 9, 25%), oculomotor disturbances (n = 6, 17%), and other symptoms (n = 7, 19%). The cerebellar ataxia presentation (14 patients) consisted of gait ataxia associated with action tremor. All the 9 patients presenting with isolated tremor had action tremor involving the upper limbs; in 7/9 cases, it progressed subsequently into an overt cerebellar syndrome. The 6 patients with an oculomotor presentation initially complained of diplopia. Four of them had objective oculomotor deficits on neurologic examination (complete ophthalmoplegia in 2 cases, internuclear ophthalmoplegia in 1 case, and third cranial nerve palsy in 1 case). In the 7 remaining patients, a variety of other CNS signs were observed (OMS, n = 2; confusion, n = 2; stiff-person syndrome [SPS], n = 1; limbic encephalitis, n = 1; parkinsonism, n = 1).
At the time the disease reached the plateau phase (median 12 weeks, range: 1–64 weeks; 28% ≥3 months), 24 patients (67%) had symptoms and signs of a cerebellar syndrome (figure 1B and video 1). The cerebellar dysfunction rarely remained isolated (n = 3; 8%) and was most commonly associated with other symptoms/signs (figure 1C): oculomotor disturbances (n = 15; 42%), opsoclonus with or without myoclonus (n = 10; 28%), spasticity (n = 9; 25%), dystonia (n = 5; 14%), and parkinsonism (n = 4; 11%). Among all cases, the cerebellar syndrome was the most common component of the full-blown presentation. Seventeen patients with initial cerebellar dysfunction developed signs of brainstem involvement as a secondary step. When the clinical findings did not include a cerebellar syndrome (12 cases; 33% of the total population), the neurologic disorder was more heterogeneous, consisting of oculomotor disturbances (n = 2), oculomotor disturbances and parkinsonism (n = 2), recurrent episodes of confusion (n = 2), OMS associated with spastic paraparesis (n = 2), SPS (n = 2), limbic encephalitis (n = 1), and peripheral neuropathy (n = 1). The patient manifesting limbic encephalitis was a 50-year-old man who presented with psychomotor slowing, behavioral disturbances, and short-term memory loss (mini-mental state examination 17/30).
Neurological examination in a patient with Ri-PNS presenting with cerebellar ataxia and slowly progressive course.Download Supplementary Video 1 (40.4MB, mp4) via http://dx.doi.org/10.1212/000699_Video_1
Other than cerebellar ataxia, the patients manifested a variety of movement disorders, including myoclonus (n = 12, 33%), dystonia (n = 6, 17%; either cervical, n = 4, or jaw-opening [oromandibular] dystonia, n = 2), and parkinsonism (n = 6, 17%). All patients with parkinsonism showed bradykinesia and rigidity, whereas only 2 of them also presented with tremor. Parkinsonian features involved asymmetrically the upper limbs and appeared later over the course of the disease in 4/6 patients. In the other 2 patients, parkinsonism was an early manifestation and involved the lower limbs. Supranuclear gaze palsy or ophthalmoplegia associated with parkinsonism was observed in 5/6 patients. Treatment with levodopa was introduced in 2 patients without clinical improvement. All those with parkinsonism were female patients with breast cancer (video 2). Among all patients, 13 (36%) developed regional or diffuse pyramidal hypertonia (spasticity), 2 of them in the context of SPS. Sex-related clinical specificities were observed as spasticity and dystonia were present only in female patients, and only male patients presented with transient, recurrent episodes of confusion (figure 2A).
Figure 2. Sex-related specificities of Ri-PNS.
Clinical (A) and oncological (B) sex-related specificities. Ri-PNS = Ri-associated paraneoplastic neurologic syndrome.
Clinical findings in an anti–Ri-positive patient with parkinsonism and oculomotor disturbances.Download Supplementary Video 2 (37.4MB, mp4) via http://dx.doi.org/10.1212/000699_Video_2
Atypical symptoms and signs were also detected in a minority of patients (n = 5, 14%). Three (8%) developed hyponatremia due to a concomitant syndrome of inappropriate antidiuretic hormone secretion (SIADH). Two others (5%) had severe dysautonomia (including heart rate instability and recurrent cardiorespiratory arrest) and central hypoventilation (Ondine syndrome).
Misdiagnoses concerned 22% of patients (n = 8): 2 of the patients from the French cohort were initially diagnosed with MS, 2 with atypical parkinsonism, 1 with Bickerstaff encephalitis, 1 was extensively studied for a genetic form of hyperekplexia, 1 with vestibular neuritis, and 1 was considered to have a functional neurologic disorder.
Oncologic associations
Thirty-three patients (92%) developed a cancer within 5 years, most commonly breast cancer (n = 22). None of the 14 patients with breast cancer for whom human epidermal growth factor receptor 2 (HER2) status was available were HER2 positive. The type of cancer was significantly associated with the sex of the patient (p < 0.001): as expected, breast cancer was more frequent in female patients (p < 0.001), whereas atypical cancer types (other than breast and lung cancer) were more frequent in men (p < 0.001), including neuroendocrine tumors and mediastinal seminoma (figure 2B).
Among patients with cancer (n = 33), the neurologic syndrome preceded the discovery of cancer in 24 (73%) patients, diagnosis of cancer and PNS were concomitant (<1 month) in 5 (15%) patients, and the neurologic manifestations followed diagnosis of cancer in 4 patients (12%). The 3 patients without cancer were women (age range: 52–83 years), with similar clinical findings as those with tumors (cerebellar syndrome and oculomotor disturbances in 2; SPS in 1).
Paraclinical data
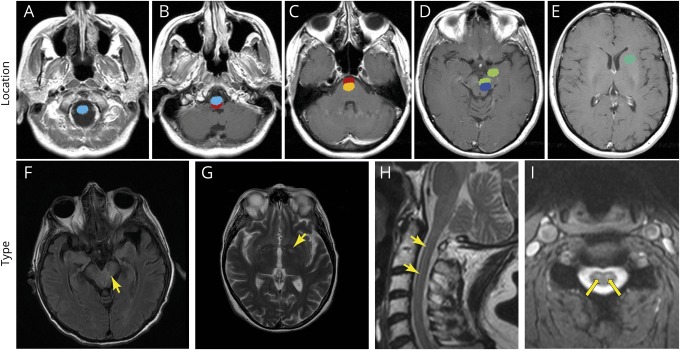
CSF analysis found pleocytosis in 36% of patients, increased protein content in 68%, and detection of oligoclonal bands in 80%, the latter being the most common CSF abnormality. Only a single CSF analysis was normal if all these 3 parameters were analyzed concomitantly. Brain MRI results were available for 33 patients (92%) and was abnormal in 6 (18%); 5 cases had T2 signal changes in the brainstem (in 1 case, the brainstem lesion coexisted with mesial temporal lobe signal abnormality, with contrast enhancement), and 1 demonstrated an isolated supratentorial lesion (left pallidum hyperintensity). The location and type of neuroradiologic abnormalities are shown in figure 3. Three patients (8%) had coexisting neural antibodies against glutamic acid decarboxylase, Hu, and glutamic acid decarboxylase/Sox-1, respectively (table e-1, links.lww.com/NXI/A225).
Figure 3. Location and type of the neuroradiologic abnormalities detected in patients with Ri-PNS.
Location (A–E) and type (F–I) of the different neuroradiologic alterations (mainly T2-weighted and fluid-attenuated inversion recovery [FLAIR] hyperintensities) detected in the French cohort of patients with Ri-PNS; each color represents a different patient (MRI abnormalities [arrows]). Note the prominent involvement of the upper cervical cord (A), brainstem structures (B–D), mesial temporal lobe (D), and basal ganglia (E). Ri-PNS = Ri-associated paraneoplastic neurologic syndrome.
Outcome and immunotherapy treatment
All patients with cancer (n = 33) received oncological treatment. In addition, 21 patients (58%) received immunotherapy: 6 (17%) received only corticosteroid bolus; 5 (14%) IV immunoglobulin (IVIG); 7 (19%) a combination of corticosteroids and IVIG; and 3 (8%) received cyclophosphamide as second-line immunotherapy. The median duration of follow-up was 13 months (range: 1–132 months). Outcome measured by the mRS was available for 22 patients. The median mRS score at disease onset was 3 (moderate disability). At 12 months, the median mRS score was 4 (moderately severe disability; the patient is unable to walk unassisted). Patient 21 showed spectacular improvement in neurologic symptoms (oculomotor disturbance and cerebellar syndrome) following breast cancer treatment with farmorubicin and cyclophosphamide. Using the Kaplan-Meier method, the overall survival at 12 months was estimated to be 73% (95% CI [0.54–0.85]), 62% at 24 months (95% CI [0.41–0.78]), and 47% at 36 months (95% CI [0.25–0.65]). There was no significant difference in overall survival according to use of immunotherapy (log-rank p = 0.615) or presence of cancer (log-rank p = 0.893).
Literature review
We screened 47 studies (41 isolated observations and 6 case series, detailed in tables e-2 and e-3, links.lww.com/NXI/A225) of the 171 records identified through the PubMed search (figure e-1, links.lww.com/NXI/A225). After removing studies that did not fulfill with the inclusion criteria, 27 studies (describing 55 cases) were included for final analysis.
Comparison between the French cohort and the literature review
Three patients were excluded from the French cohort due to the presence of coexisting antibodies, leaving 33 patients for analysis. There was no significant difference in terms of clinical or demographic characteristics, other than a significantly greater number of paraneoplastic cases among the French cohort (p = 0.023; table 1).
Table 1.
Comparison of Ri-PNS clinical features between patients reported in the literature and those from the French cohort

Discussion
This study provides several important findings concerning the clinical spectrum of patients with Ri-PNS. Most patients with Ri-PNS have a multisystem neurologic involvement, although the cerebellum and the brainstem are most commonly affected. Opsoclonus with or without myoclonus constitutes only a part of the clinical spectrum of this disease, as it appears to be less frequent than previously expected and rarely appears as an isolated manifestation. The typical progression is that of a cerebellar syndrome presenting with subacute gait ataxia, followed in a second stage by signs/symptoms of brainstem involvement, and subsequent involvement of the pyramidal system and basal ganglia. Symptom development is often slower than those of other PNS, showing a chronic course in a large proportion of patients, thus mimicking neurodegenerative conditions. The disorder predominates in females, and breast cancer is the most common cancer type overall.
The original descriptions of Ri-PNS defined OMS as the stereotyped manifestation of this disease.1–8,13–16 However, we demonstrate herein that the clinical phenotype is more heterogeneous and usually involves multiple neurologic systems, the core manifestation being a cerebellar syndrome. It is of note that OMS was present in only a quarter of the French cohort cases, and none of them presented with OMS as an isolated manifestation; the absence of OMS should therefore not exclude the diagnosis of a Ri-PNS.
According to the data presented herein, Ri-PNS is a rare disease, but we suspect that it is largely underdiagnosed. The stepwise course of the syndrome, usually subacute but chronic/progressive in almost 30% of patients, can lead to diagnostic delays or to misdiagnose the disorder as a primarily neurodegenerative disease or as a nonparaneoplastic inflammatory brain disease, thus preventing the early discovery of cancer. This is further supported by the frequency of misdiagnosis that was high in the French cohort. By reviewing critically all available cases, we were able to define a list of differential diagnoses in which Ri-PNS should be considered. The latter are characterized by a subacute/progressive ataxia-plus syndrome that develops in patients without structural or vascular brain abnormalities (table 2). In particular, in cases in which ophthalmoplegia associates with ataxia and abnormal reflexes on examination (11% of patients in the French Cohort), the presentation is strongly suggestive of Bickerstaff brainstem encephalitis (BBE), a postinfectious inflammatory brain disease. Both brain MRI and CSF tests are not able to distinguish between the 2 conditions; only a positive test for serum anti-GQ1b antibody, positive in 66% of patients with BBE, would allow a correct diagnosis.17 For example, it has been reported that a patient who presented initially with ophthalmoparesis, tetraparesis, and areflexia following a Campylobacter jejuni–related gastroenteritis, received an initial diagnosis of BBE-Guillain-Barré overlap syndrome, later proven to be an Ri-PNS associated with breast cancer.18 Aside from BBE, another relevant differential diagnosis for the Ri-PNS is represented by the group of atypical parkinsonisms, particularly progressive supranuclear palsy (PSP). Both PSP and Ri-PNS can present with supranuclear gaze palsy or ophthalmoplegia and repeated unprovoked falls, in the context of tremor, rigidity, and bradykinesia (14% of the cases in the French cohort). This presentation would likely lead to a diagnosis of probable PSP, although a sudden onset or stepwise or rapid progression should alert the clinician to look for other diagnoses, including autoimmune conditions.19 We believe that patients with Ri-PNS and chronic/progressive course could be missed if this set of criteria is applied strictly.
Table 2.
Differential diagnosis spectrum of late-onset, sporadic, progressive cerebellar-plus syndrome in patients without structural or vascular brain lesions, in which anti-Ri-PNS should be considered
Despite previous emphasis of Ri-PNS association with lung cancer,10 we demonstrate herein that, overall, breast cancer is the most frequent tumor found in patients with Ri-PNS. The analysis of the literature series further confirmed this finding. The important implication is that women with Ri antibodies, who account for 3 quarters of patients, should be investigated for the presence of breast cancer, and diagnostic surveillance should continue in cases in which cancer is not detected at the onset of the neurologic syndrome. Conversely, male patients usually harbor neuroendocrine tumors (lung or bladder) or mediastinal seminoma. A rational approach would therefore be to begin the search for an occult cancer with clinical examination and first-level tests (mammography and breast ultrasound) in women, and whole-body CT scan in men, when Ri antibodies are found. Of interest, none of the breast cancers analyzed herein demonstrated HER2 overexpression, which is usually found in approximately 20% of patients with breast cancer in general.20 Conversely, 96% of the tumors in anti–Yo-associated PNS demonstrate HER2 overexpression, strongly suggesting a different pathogenesis for the 2 diseases.20
Isolated confusion, SIADH-related hyponatremia, and severe dysautonomia were not previously reported as possible manifestations of Ri autoimmunity. This allows us to expand the clinical spectrum of the disease. We acknowledge that hyponatremia, especially if severe, could worsen the neurologic status of patients and contribute to the disturbances of gait and confusion.21 Severe dysautonomia resulted in life-threatening arrhythmias, and 1 of the patients experienced repeated episodes of cardiac arrests while he was in the intensive care unit (ICU). Dysautonomia was associated with Ondine syndrome, or central hypoventilation, characterized by normal ventilation while the patient is awake, but severe hypoventilation when asleep. Acquired central hypoventilation can result from pathologic involvement of the brainstem respiratory nuclei, as it was observed in other autoimmune conditions, including anti-Hu brainstem syndrome and anti-N-methyl-D-aspartate receptor encephalitis.22,23 Therefore, initial management and monitoring of patients in the ICU should be systematically proposed if brainstem involvement is present. Despite its phenotypical heterogeneity, there is a specific biological reason for the predilection of the autoimmune process to damage specific anatomic targets, which correspond to the different expression of the Ri antigens also called neuro-oncologic ventral antigen (NOVA)-1 and NOVA-2 in the brain.24 The neuroradiologic abnormalities detected were also in agreement with this distribution, characterized mainly by subtle alterations confined to the brainstem region. The presence of contrast enhancement at the mesial temporal lobe level in one of our patients is an interesting finding, which was previously reported in Ma2 antibody–associated syndrome.25–28
The present study highlights the grim prognosis of patients with Ri-PNS: at 1 year, most of the French patients were unable to walk unassisted, and half of them died within 3 years of disease onset. These findings are in contrast with the favorable prognosis and good functional outcome suggested by others.29 However, we did observe a case with spectacular improvement following breast cancer treatment. Therefore, despite the overall unsatisfactory effect of therapy, we believe that early cancer treatment and response evaluation to immunotherapy are reasonable options in these patients, as in other PNS.30
The present study is limited by its retrospective nature. Nevertheless, it represents the 20-year experience of a reference center, and the analysis of all cases described in the literature allowed us to redefine the main features of this disease.
In conclusion, Ri-PNS is characterized by prominent cerebellar and brainstem involvement, followed by multisystem neurologic dysfunction, with a subacute or chronic/progressive course. This could mislead diagnosis toward a neurodegenerative or an inflammatory nonparaneoplastic condition, during which the presence of underlying tumor would not be investigated. Most patients are women with an associated breast tumor, whereas male patients have a wider cancer association. During the course of the disease, several movement disorders could complicate the picture further, including parkinsonism, cervical and oromandibular dystonia, and SPS. Opsoclonus with or without myoclonus is only a part of the clinical spectrum and is rarely observed as an isolated finding. Neurologists should thus be aware of the more complex phenotype associated with Ri antibodies.
Acknowledgment
The authors thank NeuroBioTec Hospices Civils de Lyon BRC (France, AC-2013-1867, NFS96-900) for banking sera and CSF samples. They gratefully acknowledge Philip Robinson and Verena Landel for English language editing (Direction de la Recherche Clinique, Hospices Civils de Lyon).
Glossary
- BBE
Bickerstaff brainstem encephalitis
- HER2
human epidermal growth factor receptor 2
- ICU
intensive care unit
- IVIG
IV immunoglobulin
- mRS
modified Rankin Scale
- NOVA
neuro-oncologic ventral antigen
- OMS
opsoclonus-myoclonus syndrome
- PSP
progressive supranuclear palsy
- Ri-PNS
Ri-associated paraneoplastic neurologic syndrome
- SIADH
syndrome of inappropriate antidiuretic hormone secretion
- SPS
stiff-person syndrome
Appendix. Authors

Study funding
This study is supported by research grants from ANR (ANR-14-CE15-0001-MECANO) and FRM (Fondation pour la Recherche Medicale) DQ20170336751. This work has been developed within the BETPSY project, which is supported by a public grant overseen by the French National Research Agency (ANR), as part of the second “Investissements d'Avenir” program (reference ANR-18-RHUS-0012).
Disclosure
The authors have no conflicts of interest to disclose. Go to Neurology.org/NN for full disclosures.
References
- 1.Budde-Steffen C, Anderson NE, Rosenblum MK, et al. . An antineuronal autoantibody in paraneoplastic opsoclonus. Ann Neurol 1988;23:528–531. [DOI] [PubMed] [Google Scholar]
- 2.Luque FA, Furneaux HM, Ferziger R, et al. . Anti-Ri: an antibody associated with paraneoplastic opsoclonus and breast cancer. Ann Neurol 1991;29:241–251. [DOI] [PubMed] [Google Scholar]
- 3.Dropcho EJ, Kline LB, Riser J. Antineuronal (anti-Ri) antibodies in a patient with steroid-responsive opsoclonus-myoclonus. Neurology 1993;43:207–211. [DOI] [PubMed] [Google Scholar]
- 4.Bataller L, Graus F, Saiz A, Vilchez JJ; Spanish Opsoclonus-Myoclonus Study Group. Clinical outcome in adult onset idiopathic or paraneoplastic opsoclonus-myoclonus. Brain 2001;124:437–443. [DOI] [PubMed] [Google Scholar]
- 5.Prestigiacomo CJ, Balmaceda C, Dalmau J. Anti-Ri-associated paraneoplastic opsoclonus-ataxia syndrome in a man with transitional cell carcinoma. Cancer 2001;91:1423–1428. [DOI] [PubMed] [Google Scholar]
- 6.Wirtz PW, Sillevis Smitt PAE, Hoff JI, et al. . Anti-Ri antibody positive opsoclonus-myoclonus in a male patient with breast carcinoma. J Neurol 2002;249:1710–1712. [DOI] [PubMed] [Google Scholar]
- 7.Weizman DA, Leong WL. Anti-Ri antibody opsoclonus-myoclonus syndrome and breast cancer: a case report and a review of the literature. J Surg Oncol 2004;87:143–145. [DOI] [PubMed] [Google Scholar]
- 8.Armangué T, Sabater L, Torres-Vega E, et al. . Clinical and immunological features of opsoclonus-myoclonus syndrome in the era of neuronal cell surface antibodies. JAMA Neurol 2016;73:417–424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Romorini A, Frediani F, Rapuzzi S, Patruno G, Lamperti E, Nemni R. Anti-Ri antibodies and peripheral neuropathy in a man without malignancy. Eur Neurol 2004;51:177–178. [DOI] [PubMed] [Google Scholar]
- 10.Pittock SJ, Lucchinetti CF, Lennon VA. Anti-neuronal nuclear autoantibody type 2: paraneoplastic accompaniments. Ann Neurol 2003;53:580–587. [DOI] [PubMed] [Google Scholar]
- 11.Waters P, Pettingill P, Lang B. Detection methods for neural autoantibodies. Handb Clin Neurol 2016;133:147–163. [DOI] [PubMed] [Google Scholar]
- 12.Moher D, Liberati A, Tetzlaff J, Altman DG, Group TP. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Escudero D, Barnadas A, Codina M, Fueyo J, Graus F. Anti-Ri-associated paraneoplastic neurologic disorder without opsoclonus in a patient with breast cancer. Neurology 1993;43:1605–1606. [DOI] [PubMed] [Google Scholar]
- 14.Casado JL, Gil-Peralta A, Graus F, Arenas C, Lopez JM, Alberca R. Anti-Ri antibodies associated with opsoclonus and progressive encephalomyelitis with rigidity. Neurology 1994;44:1521–1522. [DOI] [PubMed] [Google Scholar]
- 15.Jongen JL, Moll WJ, Sillevis Smitt PA, Vecht CJ, Tijssen CC. Anti-Ri positive opsoclonus-myoclonus-ataxia in ovarian duct cancer. J Neurol 1998;245:691–692. [DOI] [PubMed] [Google Scholar]
- 16.Thümen A, Moser A. An uncommon paraneoplastic Ri-positive opsoclonus-myoclonus-like syndrome and Stiff-Person syndrome with elevated glutamate/GABA ratio in the cerebrospinal fluid after breast cancer. J Neurol 2010;257:1215–1217. [DOI] [PubMed] [Google Scholar]
- 17.Odaka M, Yuki N, Yamada M, et al. . Bickerstaff's brainstem encephalitis: clinical features of 62 cases and a subgroup associated with Guillain-Barré syndrome. Brain 2003;126:2279–2290. [DOI] [PubMed] [Google Scholar]
- 18.Rajabally YA, Naz S, Farrell D, Abbott RJ. Paraneoplastic brainstem encephalitis with tetraparesis in a patient with anti-Ri antibodies. J Neurol 2004;251:1528–1529. [DOI] [PubMed] [Google Scholar]
- 19.Hoglinger GU, Respondek G, Stamelou M, et al. . Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord 2017;32:853–864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rojas-Marcos I, Picard G, Chinchón D, et al. . Human epidermal growth factor receptor 2 overexpression in breast cancer of patients with anti-Yo—associated paraneoplastic cerebellar degeneration. Neuro Oncol 2012;14:506–510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pelosof LC, Gerber DE. Paraneoplastic syndromes: an approach to diagnosis and treatment. Mayo Clin Proc 2010;85:838–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Uchino A, Iizuka T, Urano Y, et al. . Pseudo-piano playing motions and nocturnal hypoventilation in anti-NMDA receptor encephalitis: response to prompt tumor removal and immunotherapy. Intern Med 2011;50:627–630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Najjar M, Taylor A, Agrawal S, et al. . Anti-Hu paraneoplastic brainstem encephalitis caused by a pancreatic neuroendocrine tumor presenting with central hypoventilation. J Clin Neurosci 2017;40:72–73. [DOI] [PubMed] [Google Scholar]
- 24.Buckanovich RJ, Posner JB, Darnell RB. Nova, the paraneoplastic Ri antigen, is homologous to an RNA-binding protein and is specifically expressed in the developing motor system. Neuron 1993;11:657–672. [DOI] [PubMed] [Google Scholar]
- 25.Vogrig A, Fouret M, Joubert B, et al. . Increased frequency of anti-Ma2 encephalitis associated with immune checkpoint inhibitors. Neurol Neuroimmunol Neuroinflamm 2019;6:e604 doi: 10.1212/NXI.0000000000000604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dalmau J. Clinical analysis of anti-Ma2-associated encephalitis. Brain 2004;127:1831–1844. [DOI] [PubMed] [Google Scholar]
- 27.Vogrig A, Ferrari S, Tinazzi M, Manganotti P, Vattemi G, Monaco S. Anti-Ma-associated encephalomyeloradiculopathy in a patient with pleural mesothelioma. J Neurol Sci 2015;350:105–106. [DOI] [PubMed] [Google Scholar]
- 28.Vogrig A, Joubert B, Maureille A, et al. . Motor neuron involvement in anti-Ma2-associated paraneoplastic neurological syndrome. J Neurol 2019;266:398–410. [DOI] [PubMed] [Google Scholar]
- 29.Shams'ili S, Grefkens J, de Leeuw B, et al. . Paraneoplastic cerebellar degeneration associated with antineuronal antibodies: analysis of 50 patients. Brain 2003;126:1409–1418. [DOI] [PubMed] [Google Scholar]
- 30.Vogrig A, Pauletto G, Belgrado E, et al. . Effect of thymectomy on refractory autoimmune status epilepticus. J Neuroimmunol 2018;317:90–94. [DOI] [PubMed] [Google Scholar]
- 31.Krzywinski M, Schein J, Birol İ, et al. . Circos: an information aesthetic for comparative genomics. Genome Res 2009;19:1639–1645. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Neurological examination in a patient with Ri-PNS presenting with cerebellar ataxia and slowly progressive course.Download Supplementary Video 1 (40.4MB, mp4) via http://dx.doi.org/10.1212/000699_Video_1
Clinical findings in an anti–Ri-positive patient with parkinsonism and oculomotor disturbances.Download Supplementary Video 2 (37.4MB, mp4) via http://dx.doi.org/10.1212/000699_Video_2
Data Availability Statement
Data reported in this article are available within the article or its supplementary materials. More information regarding the data is available from the corresponding author on reasonable request.