Abstract
Glucocerebrosidase (GCase) is a retaining β-glucosidase with acid pH optimum metabolizing the glycosphingolipid glucosylceramide (GlcCer) to ceramide and glucose. Inherited deficiency of GCase causes the lysosomal storage disorder named Gaucher disease (GD). In GCase-deficient GD patients the accumulation of GlcCer in lysosomes of tissue macrophages is prominent. Based on the above, the key function of GCase as lysosomal hydrolase is well recognized, however it has become apparent that GCase fulfills in the human body at least one other key function beyond lysosomes. Crucially, GCase generates ceramides from GlcCer molecules in the outer part of the skin, a process essential for optimal skin barrier property and survival. This review covers the functions of GCase in and beyond lysosomes and also pays attention to the increasing insight in hitherto unexpected catalytic versatility of the enzyme.
Keywords: glucocerebrosidase, lysosome, glucosylceramide, skin, Gaucher disease
1. Introduction
The cellular acid β-glucosidase (EC 3.2.1.45) was first reported to be located in lysosomes more than 50 years ago [1]. There it degrades the glycosphingolipid glucosylceramide (GlcCer), also known as glucocerebroside (Figure 1A) [2]. The enzyme, commonly named glucocerebrosidase (GCase), is active towards GlcCer molecules with different fatty acyl moieties. Deficiency of GCase causes the recessively inherited disorder Gaucher disease (GD, OMIM #230800, ORPHA355), named after the French dermatologist Ernest Gaucher, who published the first case report [3]. A hallmark of GD are lipid-laden macrophages with lysosomal GlcCer deposits, referred to as Gaucher cells [4]. Numerous mutations in the GBA gene encoding GCase have been associated with GD [5]. The genetic heterogeneity contributes to the highly variable clinical manifestation of the disorder that may involve various organs and tissues [4]. A complete absence of GCase activity is incompatible with terrestrial life due to a disturbed skin barrier [6,7]. The lethal impairment stems from the crucial extracellular role of GCase in the stratum corneum (SC). This review covers the functions of GCase in the metabolism of GlcCer inside lysosomes and beyond. First, Section 2, Section 3, Section 4 and Section 5 deal with GCase as a cellular lysosomal enzyme, and in the second part Section 6 onwards focuses on the extracellular function of GCase in the skin.
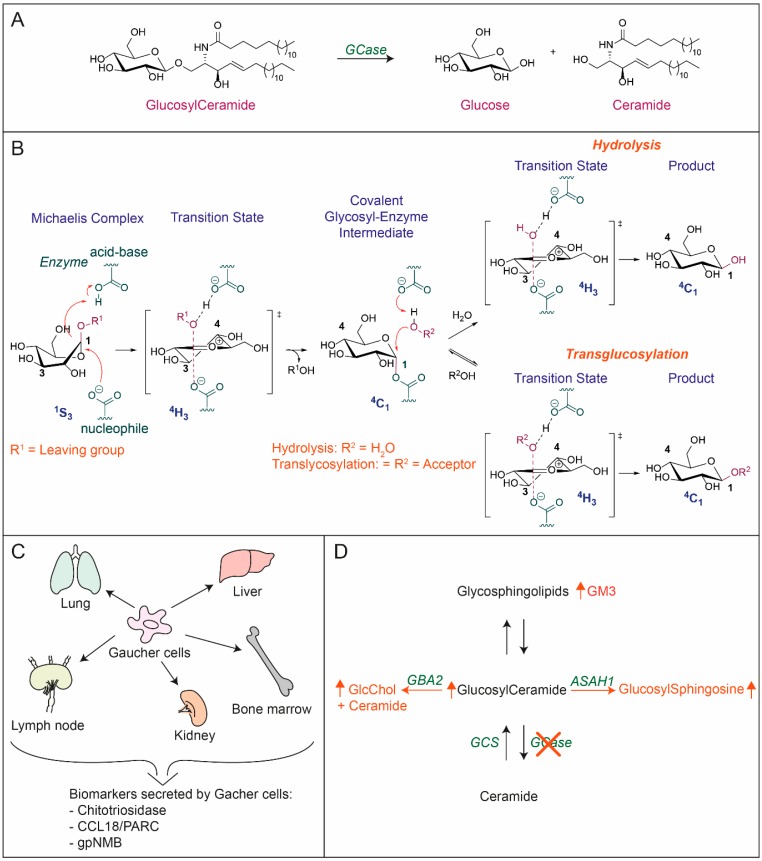
Figure 1.
(A) Structure of glucosylceramide (GlcCer) and degradation by GCase to glucose and ceramide. (B) Catalytic activity GCase: Hydrolyzation of β-glucosides and transglucosylation activity. (C) Occurrence of Gaucher cells and the biomarkers they secrete in plasma. (D) Metabolic adaptations to GCase deficiency: increase of GlcCer as a result of lack of degradation by GCase. Accumulated GlcCer is converted by ASAH1 to glucosylsphingosine, Glucosylated cholesterol (GlcChol) formed by GBA2 increases, and GM3 levels rise because increased anabolism by glycosyltransferases to complex GSLs. Enzymes are depicted in green. ASAH1: acid ceramidase, GBA2: cytosolic β-glucosidase, GCase: β-glucocerebrosidase, GCS: glucosylceramide synthase.
2. Part 1: GCase and Lysosomal Glucosylceramide Degradation
2.1. Glucosylceramide as Intermediate of Glycosphingolipids
The primary physiological substrate of GCase is GlcCer, the simplest glycosphingolipid (GSL) in which a single glucose β-glucosidic is linked to the 1-hydroxy of ceramide (Cer) [8]. Figure 2 presents an overview of the GSL metabolism. De novo formation of Cer starts on the endoplasmic reticulum (ER) with formation of 3-keto-dihydrosphingosine by the enzyme serine palmitoyl transferase (SPT) that conjugates the amino acid serine with a palmitoyl chain [9,10,11,12]. Next, the enzyme 3-ketosphinganine reductase (KSR) converts 3-keto-hydrosphingosine to dihydrosphingosine (sphinganine). Ceramide synthases (CERS) are responsible for acylation of dihydrosphingosine, thus generating diverse dihydroceramides [13,14,15]. In mammals six distinct CERS enzymes with different fatty acyl-CoA affinities have been identified. Subsequently, dihydroceramide desaturase (DES) catalyzes the conversion of dihydroceramides into ceramides 15. Ceramide is alternatively formed in the salvage pathway by acylation of sphingosine molecules released from lysosomes [16,17]. Cer can be further metabolized by conjugation of its 1-hydroxy, resulting in very diverse structures like ceramide 1-phosphate (C1P), sphingomyelin (SM), 1-O-acylceramide, galactosylceramide (GalCer), and GlcCer (reviewed in [18]). Formation of GlcCer, the key GSL of this review, involves transfer of Cer to the cytosolic surface of the Golgi apparatus where the membrane-bound glucosylceramide synthase (GCS) generates GlcCer using UDP-glucose as sugar donor and Cer as acceptor [19,20]. Next, some of the newly formed GlcCer molecules are converted back to Cer by the cytosol facing β-glucosidase GBA2 [21], but most reach via an unknown mechanism the luminal membrane of the Golgi apparatus. There, conversion to more complex GSLs like gangliosides and globosides occurs through stepwise addition of additional sugar and sulfate moieties (the biosynthesis and vast structural heterogeneity of GSL is excellently reviewed in [13,22]).
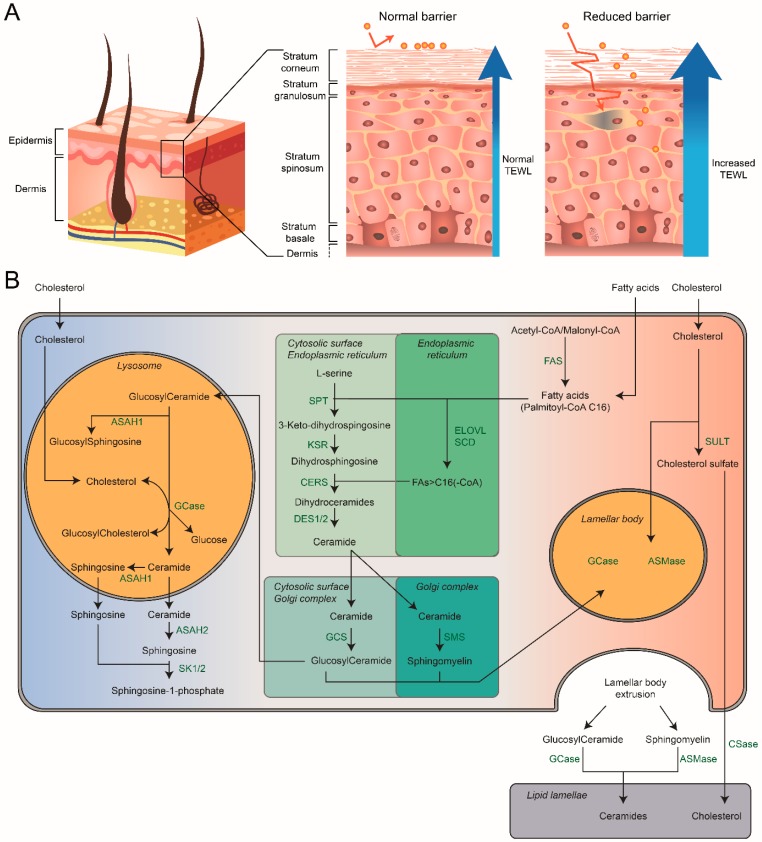
Figure 2.
Schematic overview of the human skin and the main processes involved around GCase and its related lipids. (A) Schematic overview of a cross section of the skin showing the epidermis, dermis and subcutaneous tissue. The middle illustration shows a more detailed view of the epidermis under healthy conditions. The right illustration depicts a more detailed view of the epidermis with a reduced barrier. Exogenous compounds can get into deeper layers of the epidermis when the barrier is reduced, resulting in an immune response. It also leads to an increased transepidermal water loss (TEWL). (B) Schematic overview of the main processes involved around GCase within the cell. Arrows indicate the transport or conversion of lipids; associated enzymes are listed adjacent to their abbreviations. ASAH1: acid ceramidase, ASAH2: neutral ceramidase, ASMase: acid sphingomyelinase, CERS: ceramide synthase family, CSase: cholesterol sulfatase, DES1/2: dihydroceramide desaturase 1 and 2, ELOVL: elongation of very long chain fatty acids family, FAS: fatty acid synthase, GCase: β-glucocerebrosidase, GCS: glucosylceramide synthase, KSR: 3-ketosphinganine reductase, PLA-2: phospholipase, SCD: stearoyl-CoA desaturase, SMS: sphingomyelin synthase, SPT: serine palmitoyltransferase, SULT: cholesterol sulfotransferase type 2 isoform 1b.
The major destination of newly formed GSLs is the outer leaflet of the plasma membrane. At the cell surface, GSLs fulfill a variety of important functions. GSLs interact with cholesterol molecules via hydrogen bonds and hydrophobic van der Waal’s forces and spontaneously form semi-ordered lipid microdomains, commonly referred to as lipid rafts [23,24]. Hydrophilic cis-interactions among GSL headgroups promote lateral associations with surrounding lipid and proteins. Residing in the GSL-enriched domains are proteins involved in interactions of cells with the exterior (extracellular space and other cells) and mediating the associated intracellular signaling processes [24,25,26]. The GSL composition of lipid rafts may exert modulating effects in the cell’s response to triggers. One example in this respect is the insulin receptor whose signaling is negatively influenced by neighboring gangliosides such as GM3 in lipid rafts [27,28,29]. Pharmacological reduction of GSLs results in improved glucose homeostasis in obese insulin-resistant rodents [30]. Similarly, the epidermal growth factor (EGF) receptor is influenced by the GSL composition of microdomains in which it resides [31]. GSLs at the cell surface also play direct roles in adhesion/recognition processes. For example, specific GSLs are involved in binding of pathogenic viruses, microorganisms, and bacterial toxins [32,33]. The topic was recently reviewed [34]. Glycosphingolipid-enriched lipid rafts essentially contribute to immunological functions as, for example, activation of T cells [35,36,37,38].
2.2. Lysosomal Turnover of Glycosphingolipid
GSLs may exit cells from the plasma membrane through incorporation in high-density lipoproteins [39,40]. However, most of the GSLs are internalized from the plasma membrane via endocytosis involving multi-vesicular bodies within late endosomes. Similarly, exogenous GSLs, such as constituents of lipoproteins or components of phagocytosed apoptotic cells, also reach lysosomes by endocytic processes. Upon the delivery of internalized material to lysosomes, fragmentation of GSL components takes place by step-wise removal of terminal sugars by specialized glycosidases. The process is further assisted by accessory proteins such as saposins A-D and GM2 activator protein (reviewed in [41]). The final lipid product of lysosomal fragmentation of GSLs, GalCer, and SM is in all cases Cer [41]. The lysosomal acid ceramidase (EC 3.5.1.23) subsequently splits Cer into free fatty acid and sphingosine to be exported to the cytosol [42]. The cytosolic sphingosine can then be used for the formation of Cer or can be converted by sphingosine kinases (SK1 and SK2) to sphingosine-1-phosphate (S1P) [43].
3. Glucocerebrosidase
3.1. GCase Protein and Life Cycle
The penultimate step in GSL degradation is the deglucosylation of GlcCer yielding glucose and Cer. This reaction is catalyzed by GCase, a 495 amino acid glycoprotein with four N-linked glycans [2,44]. GCase, based on its structural features, is classified in the glycoside hydrolase family GH30 (formerly in the related family GH5) [45]. The 3-D structure of GCase was resolved by crystallography [46,47]. GCase, like other GH5 and GH30 glycosidases, has an (α/β)8 TIM barrel catalytic domain. In the case of GCase this is fused with a β-structure consisting of an immunoglobulin-like fold [45]. GCase is a retaining β-glucosidase hydrolyzing a glucosidic substrate with net retention of glucose stereochemistry (Figure 1B).
Retaining beta-glucosidases generally use a two-step catalytic mechanism. The Koshland double displacement mechanism involves a catalytic nucleophile and acid/base residue [48]. A nucleophilic attack to the anomeric carbon of the glycosidic substrate is the first step. The aglycon is released assisted by a proton transfer from the acid/base residue and a covalent enzyme–glycoside complex is formed. Next, an activated water molecule deglycosylates the nucleophile, allowing a new round catalysis. The reaction involves two transient oxocarbenium ion-like states and the sugar substrate adopts different itineraries depending on its pyranose ring configuration [49]. In the case of retaining β-glucosidases like GCase, the substrate itinerary is 1S3 → 4H3 → 4C1 → 4H3 → 4C1 for the Michaelis complex → transition state → covalent intermediate → transition state → product [50,51]. In the (α/β)8 TIM barrel catalytic domain of GCase, E340 acts as nucleophile and E235 as acid/base residue [52,53].
Cyclophellitol, present in the mushroom Phellinus sp., is a potent irreversible inhibitor that binds covalently, in mechanism-based manner, to the nucleophile E340 of GCase [52,53,54]. The structurally related compounds cyclophellitol aziridine and conduritol B-epoxide inactivate GCase via the same mechanism [52,55]. Recently, superior suicide inhibitors for GCase were designed [56]. Cyclophellitol derivatives carrying a large hydrophobic substituent at C8 inactivate GCase with even higher affinity and with great specificity (not reacting with another retaining β-glucosidase like GBA2 and GBA3) [56,57]. Using cyclophellitol as scaffold, selective activity-based probes (ABPs) toward GCase were designed [52]. A reporter group (biotin or BODIPY) was attached to the C8 of cyclophellitol via a pentyl linker rendering ABPs allowing ultrasensitive and specific visualization of GCase in vitro and in vivo [58]. Subsequently, cyclophellitol aziridine ABPs with attached reporter groups via alkyl or acyl linkers were designed reacting with multiple retaining glycosidases in the same class [55,59]. Cyclophellitol aziridine ABPs labeling α-galactosidases, α-glucosidases, α-fucosidase, α-iduronidase, β-galactosidases, and β-glucuronidase, as well as cyclophellitol ABPs labelling galactocerebrosidase, were designed [60,61,62,63,64,65]. Applications of ABPs are the quantitative detection and localization of glycosidases in cells and tissues, as well as identification and characterization of glycosidase inhibitors by competitive ABP profiling [66,67].
GCase shows an acid pH optimum of hydrolytic activity, coinciding with the lysosomal pH [44]. The activity of the enzyme towards GlcCer is promoted by negatively charged lipids and saposin C, an activator protein generated in the lysosome by proteolytic processing of prosaposin [41,68]. The half-life of GCase in lysosomes is relatively short due to proteolytic degradation by cathepsins as suggested by the protective effect of leupeptin [69,70]. It was noted that unfolding and degradation of GCase is protected by occupation of the catalytic pocket [69].
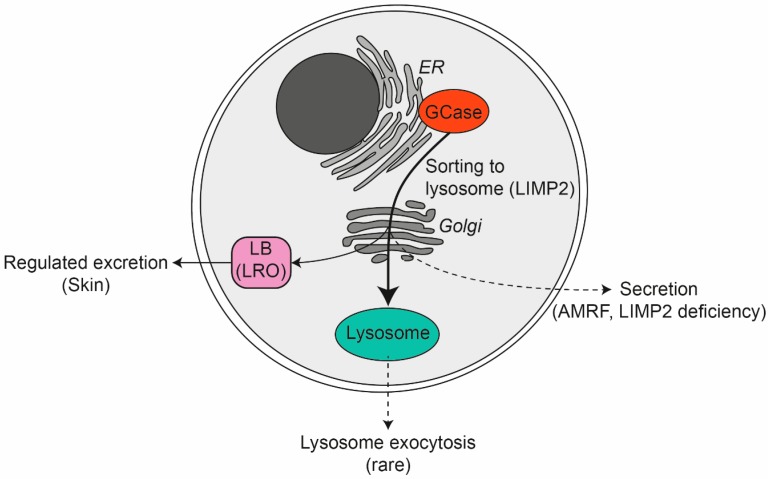
GCase fundamentally differs from other lysosomal hydrolases in the mechanism underlying sorting and transport to lysosomes [44]. While most soluble lysosomal hydrolases are transported to lysosomes by mannose-6-phosphate receptors, this is not the case for GCase. In the inherited disorders mucolipidoses II and III, where formation of mannose-6-phosphate recognition signal in N-glycans of lysosomal hydrolases is impaired and consequently these enzymes are largely secreted, the transport of GCase to lysosomes is normal. In fact, in cultured skin fibroblasts the four N-glycans of GCase do not acquire mannose-6-phosphate [71]. Following correct folding of newly formed GCase molecules in the ER, these bind to the membrane protein LIMP2 (lysosomal membrane protein 2) [72,73,74]. This binding is mediated by hydrophobic helical interfaces on both proteins [75]. Action myoclonus renal failure syndrome (AMRF) is a recessively inherited disease caused by mutations in LIMP2 [76]. In most cell types of AMRF patients, except for phagocytic cells, GCase is markedly reduced due to faulty transport to lysosomes [76,77]. More recently, progranulin (PGRN) was identified as another factor influencing GCase [78,79]. PGRN is thought to function as a chaperone facilitating the transport of GCase to lysosomes. It recruits heat shock protein 70 (HSP70) to the GCase/LIMP2 complex in the ER and thus promotes delivery of GCase to lysosomes [80]. Another protein found to interact with newly formed GCase in the ER is ERdj3 [81].
3.2. Catalytic Activity of GCase
The primary substrate of GCase is GlcCer, as is reflected by the prominent accumulation of this lipid during GCase deficiency [82,83,84]. However, it recently has become apparent that catalytic versatility of the enzymes needs consideration. Firstly, GCase was found able to hydrolyze artificial β-xylosides [20]. Secondly, several retaining β-glycosidases are reported to be able to transglycosylate when provided with a suitable aglycon acceptor (Figure 1B) [85]. Such catalytic activity has also been observed for GCase, the enzyme being able to generate glucosylated cholesterol (GlcChol) by transglucosylation [86,87,88]. This reaction occurs during cholesterol accumulation in lysosomes as occurs in Niemann–Pick disease type C (NPC) [86]. Massive accumulation of GlcChol in the liver of NPC mice was demonstrated. Inducing lysosomal cholesterol accumulation in cultured cells by their exposure to U1986663A is accompanied by formation of GlcChol [86]. Of note, under normal conditions GlcChol is primarily degraded by GCase into glucose and cholesterol. It may be envisioned that further research will reveal that there exist more β-glucosidic metabolites being substrates (and products) of GCase.
4. Gaucher Disease, Inherited Deficiency in GCase
4.1. Gaucher Disease, a Lysosomal Storage Disorder
Since degradation of GSLs is catalyzed by lysosomal glycosidases, inherited deficiencies in these enzymes cause lysosomal accumulation of their GSL substrates, so-called glycosphingolipidoses [9,41,89,90,91]. Examples of such disorders are Gaucher disease, Krabbe disease, GM2-gangliosidosis, Sandhoff disease, and GM1-gangliosidosis. The glycosphingolipidoses are clinically diverse and generally show marked heterogeneity in severity of disease that usually involves neuropathy in more severely affected patients.
Gaucher disease is a prototype glycosphingolipidosis. The first case report was published in 1882 by Ernest Gaucher concerning a female patient with unexplained massive splenomegaly without leukemia [3]. Soon it was recognized that this patient represented a distinct disease entity that was subsequently referred to as Gaucher’s disease or Gaucher disease (GD). Following the demonstration of abnormal accumulation of GlcCer in GD patients as the result of deficient GCase activity, the GBA gene encoding the acid β-glucosidase was cloned and characterized [4]. The GBA gene is located at locus 1q21 and neighbored by a pseudogene [92]. Numerous mutations in GBA have now been associated with GD. The consequences of mutations at the level of the GCase protein may markedly differ. For example, the common N370S GCase mutation among Caucasians results in near normal production of a mutant enzyme with aberrant catalytic properties [93]. The heteroallelic presence of this mutation protects against a neuronopathic disease course [4,44,94]. This GBA mutation occurs relatively frequently among Ashkenazim and was proposed to offer some advantage against an infectious disease, for example, bubonic plaque or tuberculosis [95,96,97,98,99]. In contrast, another common pan-ethnic mutation encoding L444P GCase, results in a mutant enzyme that largely misfolds in the ER and consequently only a small fraction (<10% of normal) reaches lysosomes. Homozygosity for the L444P mutation is always associated with a severe neuronopathic disease manifestation [4]. The L444P mutation is thought to have arisen repeatedly by homologous recombination of the GBA gene with its pseudogene.
The genetic heterogeneity of GD is accompanied by clinical heterogeneity of the disorder. Common symptoms manifesting in GD patients are hepatosplenomegaly, hematological abnormalities like anemia and thrombocytopenia, skeletal disease, and neuropathology. A very severe manifestation (referred to as collodion baby) involves lethal skin barrier dysfunction [4]. Discrete phenotypic variants of GD are historically discerned: type 1, the non-neuronopathic variant; type 2, the acute neuronopathic variant; type 3, the subacute neuronopathic variant and the collodion baby or neonatal variant. It was proposed to no longer adhere to this classification, but rather view GD as a continuum of phenotypes [99]. Marked intraindividual variation occurs in type 1 GD patients in the nature and degree of organ involvement and particular symptoms such as skeletal disease [4].
The correlation of GBA genotype with GD phenotype is limited in some aspects. While the presence of N370S GCase protects GD patients against neuropathology, there are several reports of monozygotic GD twins with discordant severity of visceral disease [100,101]. A very specific clinical course is associated with the presence of D409H GCase involving yet unexplained cardiac symptoms, including aortic valve, mitral valve, and ascending aorta calcifications [102,103,104].
Modifier genes, and possibly epigenetics and external factors, are considered to impact on the clinical manifestation of GCase deficiency. The transmembrane protein CLN8 (ceroid-lipofuscinosis, neuronal 8), recycling between the ER and Golgi apparatus, is a putative modifier [105]. CLN8, identified as putative modifier of GD in a genome-wide association study, has recently been reported to be involved in the transport of newly formed lysosomal enzymes between ER and Golgi [106]. Other proteins are known to directly influence the life cycle and activity of GCase. Saposin C is the lysosomal activator protein of GCase, and patients with a defective saposin C develop symptoms similar to GD patients [68]. LIMP2, encoded by the SCARB2 (scavenger receptor class B, 2) gene, transporting GCase to lysosomes was reported to be a GD modifier [107]. Polymorphisms in the UGCG gene coding for GCS catalyzing synthesis of GlcCer have also been proposed as GD modifiers [108]. Recently, microRNAs up- or downregulating GCase and downregulating LIMP2 were reported [109].
It has recently been appreciated that carrying a mutant GBA gene is not without health risk. Carriers of GD have a yet unexplained significantly increased (20-fold) risk for developing Parkinson disease (PD) and Lewy body dementia (LBD) [110,111,112]. A recent study in the United Kingdom revealed that 5–25% of patients with PD carry glucocerebrosidase gene mutations, and 10–30% of glucocerebrosidase carriers will develop PD by age 80 [113]. Of note, active GCase activity is also decreased, and corresponding glycosphingolipid substrate levels elevated, in the brain in PD without GBA1 mutations [114,115]. Abnormalities in multiple enzymes and other proteins involved in sphingolipid metabolism were observed in association with PD [114,116,117]. With increasing age, the brain of mice shows reduced GCase levels and increased amounts of lipid substrate [115]. PD is historically viewed as a “proteinopathy” with cellular protein aggregates like that of α-synuclein (αSyn). It has more recently been hypothesized that sphingolipid abnormalities may be primary disturbances that can produce protein aggregation [114]. Indeed, inactivation of GCase promotes accumulation of αSyn aggregates [118]. It was observed that insoluble alpha-synuclein positive aggregates in sporadic PD midbrain linearly correlate with loss of GCase activity [119]. Likewise, protein aggregates develop in mice with primary GBA mutations [120]. Supplementation of GCase or reduction of accumulating glycolipids prevents and reverses α-synucleinopathy [121,122]. It was furthermore observed that over-expression of aggregating αSyn causes a reduction of GCase, suggesting a potential harmful interaction between the two proteins in a self-amplifying manner [123,124,125]. In vitro experiments showed that GCase and aSyn may directly interact at lysosomal pH [126]. Different explanations have been proposed for ways by which mutant GCase may induce α-synucleinopathy (reviewed in [125,127,128]). For example, it was hypothesized that the accumulation of substrates of GCase is pathogenic; that GCase deficiency causes inhibition of autophagy and lysosomal degradative capacity and subsequently reduces turnover of αSyn; that increased αSyn levels impair the activity of GCase and vice versa; and, that GCase deficiency impairs mitochondria. Contrarily, it was proposed that mutant GCase protein may be toxic by inducing an excessive unfolded protein response in the ER or saturating the ubiquitin–proteasome pathway [129,130]. It is conceivable that multiple mechanisms may be involved in the GBA-PD pathology.
4.2. Lysosomal GlcCer Deposits in Macrophages: Gaucher Cells
The storage of GlcCer in GD patients occurs almost exclusively in macrophages residing in the spleen, liver, bone marrow, lymph nodes, and lung (Figure 1C) [131]. The lipid-laden Gaucher cells are viable, alternatively activated macrophages [132]. These cells overproduce and secrete specific proteins resulting in massively elevated plasma levels in symptomatic GD patients. These proteins are now used as biomarkers of body burden of Gaucher cells. The first identified plasma biomarker is the chitinase named chitotriosidase encoded by the CHIT1 gene [133,134]. It can be conveniently detected by the measurement of its activity towards 4-methylumbelliferyl-chitotrioside [133] and the superior substrate 4-metylumbelliferyl-4′-deoxy-chitobioside [135,136]. Plasma chitotriosidase is on average about 1000-fold elevated in type 1 GD patients. Immunohistochemistry and in situ hybridization revealed that the enzyme is produced by Gaucher cells. Common is a 24-base-pair duplication in the CHIT1 gene that excludes synthesis of active chitinase [137]. The chemokine CCL18/PARC (Chemokine (C-C motif) ligand 18; Pulmonary and activation-regulated chemokine) serves as an alternative plasma marker of Gaucher cells, being 20 to 50-fold elevated plasma of type 1 GD patients [138,139]. The chemokine is over-produced and secreted by Gaucher cells [139]. More recently the glycoprotein nonmetastatic melanoma protein B (gpNMB) was found to be overproduced by Gaucher cells [140]. A soluble fragment of gpNMB is released into plasma and is over 50-fold elevated in type 1 GD patients [140,141]. In cerebral spine fluid and brain of type 3 GD patients elevated gpNMB levels have also been observed [142]. Likewise, recently an increased level of gpNMB in the substantia nigra of PD patients was reported [143]. In mice with conditional deficiency in GCase in the white blood cell lineage Gaucher-like cells are formed. These do not produce chitotriosidase or CCL18, but gpNMB does [140,144]. Inactivation of GCase with an irreversible inhibitor was found to increase gpNMB in the brain [143]. Interestingly, zebrafish and fruit flies overproduce a chitinase during GCase deficiency [130,145].
There is compelling evidence for a direct role of Gaucher cells in GD pathology. Their presence in spleen, liver, and bone marrow is associated with splenomegaly, hepatomegaly, and hematological abnormalities, respectively [4]. The same holds for these symptoms in GD mice with induced GCase deficiency in white blood cells [144]. In GD spleens the storage lesions contain a core of mature Gaucher cells surrounded by pro-inflammatory macrophages [132]. These lesions likely contribute to the complex cytokine, chemokine, and protease abnormalities in GD patients [91,146,147]. Type 1 GD patients show low-grade inflammation and activation of both coagulation and the complement cascade [148,149]. Of note, many of the visceral symptoms of type 1 Gaucher disease patients resemble those of Niemann–Pick type A and B patients suffering from lysosomal acid sphingomyelinase (ASMase) deficiency causing lysosomal sphingomyelin storage [42]. In both disorders, lipid storage in visceral macrophages is a hallmark. In sharp contrast, while GCase is markedly reduced in most cell types of LIMP2-deficient AMRF patients, their symptoms differ from those of type 1 Gaucher patients. Likely this is due to the fact that macrophages of AMRF patients contain a high residual GCase and consequently no lipid-laden macrophages are formed [77].
4.3. Therapies of Gaucher Disease: ERT, SRT, PCT/EET
The prominence of lipid-laden macrophages in GD and their relationship to pathology has prompted the design of rational therapies aiming to prevent and/or correct the lipid-laden macrophages. The first effective treatment designed for type 1 GD is enzyme replacement therapy (ERT) aiming to supplement patient macrophages with lacking enzyme by repeated intravenous enzyme infusion [150]. Therapeutic GCase, nowadays recombinant but initially isolated from placenta, has enzymatically modified N-linked glycans with terminal mannose groups to favor uptake via the mannose receptor (or another mannose-binding lectin) at the surface of tissue macrophages. Two weekly ERTs reverse hepatosplenomegaly and hematological abnormalities in type 1 GD patients [67]. In addition, it reduces storage cells in the bone marrow [151]. Present ERT does, however, not prevent neurological symptoms due to the inability of enzyme to pass the blood brain barrier.
An alternative GD treatment is substrate reduction therapy (SRT) [152,153,154]. SRT aims to balance synthesis of GlcCer with reduced GCase activity of GD patients. Oral inhibitors of GCS (Miglustat and Eliglustat) are approved drugs. Eliglustat therapy resembles ERT in efficacy [155]. Brain-permeable inhibitors of GCS are presently designed and tested [156]. The response to treatment of GD patients is primarily monitored by clinical assessments. A retrospective evaluation revealed that reductions in plasma chitotriosidase during ERT correlate with corrections in liver and spleen volumes, improvements in hemoglobin, platelet count, and bone marrow composition [157]. Given the observed positive outcome of bone marrow transplantation in type 1 GD patients, genetic modification of hematopoietic stem cells was, and still is, seriously considered as therapeutic avenue [144].
At present there is still an unmet need for neuronopathic GD. Small compounds are actively studied as potential therapeutic agents in this respect. One envisioned approach is pharmacological chaperone therapy (PCT). Chemical chaperones are small compounds improving folding of mutant GCase in the ER, thus increasing lysosomal enzyme levels. Current studies with ambroxol, a weak inhibitor of GCase, indicate impressive reductions in spleen and liver volumes in ambroxol-treated type 1 GD patients as well as clinical improvements in type 3 GD patients [158,159,160]. Another approach is enzyme enhancement therapy with small compounds (EET). An example of this is arimoclomol, a heat shock protein amplifier, found to improve refolding, maturation, and lysosomal activity of GCase in GD fibroblasts and neuronal cells [161].
5. Metabolic Adaptations to Lysosomal GCase Deficiency
5.1. Formation of Glucosylsphingosine From Accumulating GlcCer
Important metabolic adaptations occur during GCase deficiency in lysosomes (Figure 1D) [162]. We demonstrated that part of the accumulating GlcCer is actively converted by lysosomal acid ceramidase to glucosylsphingosine (GlcSph) [163]. GlcSph is sometimes also referred to as lyso-GL1 or lyso-GB1. It was earlier observed that GlcSph is increased in the brain and spleen of GD patients [164,165]. We firstly reported an average 200-fold increased GlcSph level in plasma of symptomatic type 1 GD patients [166]. Urine of GD patients also contains increased GlcSph isoforms [167]. Pharmacological inhibition of GCase in cultured cells and zebrafish embryos causes a rapid increase in GlcSph [168]. The quantitative detection of GlcSph in biological samples was improved by o-phthaldialdehyde (OPA) derivatization and high-performance liquid chromatography [169]. Further improvement was reached by the introduction of LC-MS/MS (liquid chromatography-mass spectrometry) employing an identical (13)C-encoded glucosylsphingosine standard [168]. Measurement of elevated plasma GlcSph is now regularly used in the confirmation of GD diagnosis.
Excessive GlcSph in GD patients is believed to contribute to various symptoms. GlcSph was linked to the common reduced bone mineral density (osteopenia) in GD patients by impairing osteoblasts [170]. It is reported to promote α-synuclein aggregation, a hallmark of Parkinson disease [171]. Antigenicity of GlcCer, and possibly GlcSph, is thought to cause the common gammopathies in GD patients, gammopathies that can lead to multiple myeloma [172]. The same lipids were proposed to activate the complement cascade activation and associated local tissue inflammation [173]. GlcSph is hypothesized to diminished cerebral microvascular density in mice, based on the observed interference of the lipid with endothelial cytokinesis [174]. Earlier studies have provided evidence that GlcSph promotes lysis of red blood cells, impairs cell fission during cytokinesis, damages specific neurons, interferes with growth, and activates pro-inflammatory phospholipase A2 (see for a review [91]). In line with these observations is the occurrence of hemolysis, multinucleated macrophages, neuropathology, growth retardation, and chronic low-grade inflammation in GD patients [4]. Of note, in the brain of ageing mice reduction of active GCase in combination with increased glucosylceramide and glucosylsphingosine levels were observed [116].
The conversion of accumulating GSL in lysosomes to glycosphingoid bases (lyso-lipids) is not unique to Gaucher disease. Comparable acid ceramidase-dependent formation of sphingoid bases occurs in Krabbe disease (galactosylsphingosine), Fabry disease (globotriaosylsphingosine; lysoGb3), GM2-gangliosidosis (lysoGM1), and GM2-gangliosidoses (lysoGM2) [91,175]. In Niemann–Pick disease types A and B, the water soluble lysoSM is formed from accumulating SM [176]. As for GlcSph in GD, toxicity of excessive galactosylsphingosine in Krabbe disease and excessive lysoGb3 in Fabry disease have been proposed [91,177,178,179,180,181,182].
5.2. Excessive Gangliosides
In GD patients increases of the ganglioside GM3 (monosialodihexosylganglioside) in plasma and spleen were observed [183]. It is unknown whether this abnormality is caused by increased metabolic shuttling of newly formed GlcCer to gangliosides and/or impaired recycling of gangliosides. Not surprisingly (see Section 2.1), the elevated concentrations of GM3 in GD patients are accompanied by insulin insensitivity, without overt hyperglycemia [184].
5.3. Increased Activity of Cytosol-Faced GBA2 and GlcChol
Besides GCase, cells contain another retaining β-glucosidase that metabolizes GlcCer. The enzyme GBA2 was discovered during studies with GCase-deficient cells [21]. GBA2 is synthesized as soluble cytosolic protein that rapidly associates to the cytosolic leaflet membranes with its catalytic pocket inserted in the lipid layer. GBA2 shows prominent transglucosylase capacity and is largely responsible for the (reversible) formation of GlcChol from GlcCer and cholesterol [86]. The GBA2 gene (locus 1p13) was identified and GBA2-deficient mice have meanwhile been generated [185,186]. The animals develop normally without overt abnormality, except for incidences of male infertility [185]. GBA2-deficient zebrafish also develop normally [168]. Inhibition of GBA2 in GD and NPC patients treated with N-butyldeoxynojirimycin causes no major complications, whereas on the other hand, individuals with spastic paraplegia and cerebellar ataxia were found to be GBA2 deficient [187,188,189,190]. The physiological role of the highly conserved GBA2 is still an enigma [191].
Reducing GBA2 activity, genetically or using small compound inhibitors such AMP-DNM, has remarkable beneficial effects in NPC mice, ameliorating neuropathology and prolonging lifespan significantly [52,53]. A comparable neuro-protective effect of the iminosugar AMP-DNM was also observed in mice with Sandhoff disease, another neuropathic glycosphingolipidosis [54]. Presently zebrafish models are used to study the poorly understood interplay between GCase and GBA2-mediated metabolism of GlcCer [168]. The possible toxic effect of excessive glucosylated metabolites generated by GBA2 during GCase deficiency warrants further investigation.
6. Part 2: GCase and Glucosylceramide Metabolism Beyond the Lysosome
6.1. GCase: Other Locations Than Lysosomes
As discussed in Section 3.1, GCase does not rely on mannose-6-phosphate receptor-mediated intracellular sorting and re-uptake after secretion. The intracellular transport of GCase is tightly governed by the membrane protein LIMP2 and secretion of GCase into the extracellular space is normally prevented [77]. Immuno-electron microscopy has revealed that specific organelles are involved in trafficking of GCase-LIMP2 complexes from the Golgi apparatus to lysosomes [192]. The delivery of GCase to other locations than lysosomes warrants consideration and discussion.
6.2. Lysosome-Related Organelles
To fulfill specific physiological functions several cell types have adapted their endolysosomal apparatus and evolved specialized secretory compartments, the lysosome-related organelles (LROs) (for reviews see [193,194]). The LROs are diverse and comprise endothelial cell Weibel-Palade bodies, cytotoxic T cell lytic granules pigment cell melanosomes, and platelet dense and alpha granules. Common components of LROs are tetraspanin CD63, and GTPases RAB27A or RAB27B. The same proteins also occur in multivesicular endosomes (MVEs) that excrete intraluminal vesicles (ILVs) as exosomes upon fusion with the plasma membrane [195]. The notochord vacuole in the zebrafish is also considered to be an LRO [196,197]. Interestingly, LIMP2, the GCase transporter protein, was implicated in the formation of this LRO [198].
An established link between GSLs and LROs concerns the pigmented melanosomes in melanocytes. The formation of melanosomes requires GSLs: melanoma cells when deficient in GCS lose pigmentation due to aberrant transport of the enzyme tyrosinase synthesizing melanin [199]. Similarly, cultured melanocytes lose pigmentation when treated with a GCS inhibitor (Smit and Aerts, unpublished observations).
Keratinocytes contain a special kind of LRO, the lamellar body (LB), which justifies more detailed discussion regarding GSLs and their metabolism (see Section 8 and Section 9). Prior to this, the composition of the mammalian skin is introduced in the section below.
7. Composition of the Skin
7.1. Skin Differentiation and Barrier Formation
The mammalian skin acts as a key barrier offering protection against xenobiotics and harmful pathogens and preventing excessive water loss from the body (Figure 2A) [200]. The barrier function resides in the epidermis, the outermost part of the skin that consists of four distinct layers: the stratum basale (SB), stratum spinosum (SS), stratum granulosum (SG), and stratum corneum (SC) [201]. The innermost SB, SS, and SG are the vital parts of the epidermis (thickness: 50–100 μm) while the SC is the non-vital differentiation product (thickness: 10–20 μm). The SB contains proliferating keratinocytes that after escape from this single cell layer start to differentiate and migrate towards the SC, where the keratinocytes differentiate to terminal corneocytes. During this differentiation process the keratinocytes flatten and diminish their water content. During the flattening process cells become filled with keratin. At the interface between the SG and SC, subcellular structures like organelles and nuclei are degraded and corneocytes are formed (as reviewed in [202]).
7.2. Stratum Corneum: Hydration and Skin-pH
Proper function and features of the SC are dependent on optimal water content and acidity. The SC hydration level depends on multiple factors such as amino acids, specific sugars and salts, referred to as the natural moisturizing factor (NMF) [203]. Amino acids of the NMF are breakdown products of the major SC protein filaggrin. Mutations in the filaggrin gene FLG cause a reduced NMF level associated with dry skin [204,205]. NMF also plays a key role in maintenance of pH in the SC. At the outside of the SC the pH is 4.5–5.3 and it gradually increases to pH 6.8 in the inner SC [206]. The local pH likely modulates the activity of various enzymes in the SC, including GCase and ASMase, with optimal catalytic activity at a more acid pH, and thus also impacts on lipid structures [207].
7.3. Stratum Corneum: Composition
The SC has a “brick-and-mortar” like structure, where the corneocytes are the “bricks” embedded in a lipid matrix that is the “mortar” of the SC [206,208]. During the terminal differentiation of corneocytes, plasma membranes develop into the cornified lipid envelope, a lipid-linked crosslinked protein structure [209]. The cornified lipid envelope acts as template for the formation and organization of extracellular lipid lamellae [210,211]. The lipid matrix contains approximately on a total lipid mass basis 50% ceramides, 25% cholesterol, and 15% free fatty acids with very little phospholipid. The adequate balance of lipid components is essential for proper lipid organization and SC barrier competence [212]. Alterations in the lipid composition have been associated to various skin diseases, particularly to psoriasis, atopic dermatitis and several forms of ichthyosis [213,214,215,216,217,218].
8. Sphingolipids of the Stratum Corneum
8.1. Role of Lamellar Bodies
Keratinocytes having specific ovoid-shaped LROs with a diameter of about 200 nm are called lamellar bodies (LBs), or alternatively lamellar granules, membrane-coating granules, cementsomes, or Odland bodies [219]. LBs have a bounding membrane surrounding lipid disks. The main lipids packed in LBs are precursors of ceramides and fatty acids constituting the lamellar matrix in the SC. In the uppermost granular cells, the bounding membrane of the LB fuses into the cell plasma membrane, and the lipid disks are extruded into the intercellular space between the SC and SG. The initially extruded content of the LB is largely metabolized to ceramides and fatty acids and rearranged to form together with cholesterol the intercellular lamellae of the SC.
Keratinocytes serve as the initial factory of the permeability barrier of the skin [219]. Briefly, the generation of SC barrier lipids initiates in keratinocytes, where ceramides are de novo formed by ceramide synthase 3 (CerS3). The sphingolipid content of keratinocytes increases along with differentiation. Newly formed ceramides are rapidly modified into glucosylceramides (GlcCers) and sphingomyelins (SMs), thereby likely protecting keratinocytes from cytotoxic ceramide effects. Next, these sphingolipids are packaged into LBs [212]. The membrane protein ABCA12 (ATP-binding cassette sub-family A member 12) is essential for the presence of GlcCer in LBs [220,221,222]. Several mutations in the ABCA12 gene cause Harlequin-type ichthyosis, characterized by thickened skin over nearly the entire body at birth and causing early death. Incorporated in LBs besides lipids are also acid hydrolases including GCase, ASMase, and phospholipase A as well as proteases and antimicrobial peptides. Following exocytotic secretion of LBs, the SM and GlcCer molecules are largely enzymatically re-converted to ceramides [223,224].
8.2. Chemical Composition of Skin Sphingolipids
The sphingolipids in the skin differ in their complexity of chemical composition from those encountered in most tissues. Firstly, their sphingosine backbones are modified to yield from dihydroceramide (DS) precursors not only the regular ceramide (S) but 6-hydroxyceramide (H), phytoceramide (P) and 4,X-dihydroxysphinganine containing ceramide (T), as well [225,226,227,228]. In addition, skin ceramides have unique fatty acyl moieties. Besides regular non-hydroxylated fatty acyls of variable chain length, there are α-hydroxylated and ω-esterified structures (acylceramides) [229].
In keratinocytes, fatty acids can be elongated by elongases (mainly ELVOL1, ELVOL4, and ELVOL6) [230,231]. Very long chain fatty acids are incorporated in phospholipids and sphingolipids are packaged in LBs. Cholesterol does not require a conversion to be transported into LBs. Cholesterol can furthermore be metabolized to oxysterol or cholesterol sulfate. Oxysterol and cholesterol sulfate can both stimulate keratinocyte differentiation, additionally, cholesterol sulfate has a key role in [232,233,234,235]. Since cholesterol sulfate is highly amphiphilic it can cross the cell membrane and directly enter the SC, where it is metabolized by LB-derived steroid sulfatase to cholesterol [236,237]. Because cholesterol sulfate inhibits proteases that are involved in desquamation [238], its decrease in the upper layers of the SC results in the initiation of desquamation [239,240].
Besides the presence of regular ceramides, the scaffold of the lipid matrix in the SC is built of acylceramides, containing ω-hydroxylated very long chain fatty acids acylated at the ω-position with linoleic acid [212,228]. Also, the acylceramides are synthetized in the keratinocytes, where they and regular ceramides are glucosylated at Golgi membranes and secreted via LB secretion. Extracellularly the linoleic acid residues are replaced by glutamate residues at proteins exposed on the surface of corneocytes, thus completing the corneocyte lipid envelope [212,228,241].
9. GCase: Crucial Extracellular Role in the Skin
Inhibition of either cholesterol, phospholipid, ceramide or glucosylceramide synthesis prevents the delivery of lipids into LBs, disrupting LB formation, thereby impairing barrier homeostasis (Figure 3) [242]. LB secretion and lipid structure is abnormal in the outer epidermis of multiple skin diseases, like Atopic Dermatitis and Netherton syndrome [215,243,244]. A complete lack of GCase results in a disease phenotype (collodion baby) with fatal skin abnormalities and inhibition of GCase activity reduces the permeability barrier formation [245,246,247,248]. Gaucher mice homozygous for a null allele develop skin abnormalities that are lethal within the first day of life [6,7]. Holleran and colleagues showed increased trans-epidermal water loss (TEWL) and altered barrier function in GCase-deficient mice [248], suggesting deficient conversion of GlcCer to ceramides by GCase alters the skin barrier function. Identical changes were observed in hairless mice treated with GCase inhibitor bromoconduritol B epoxide, however, ceramide levels remained normal [246,248]. Similarly, mice deficient for prosaposin, and therefore also lacking the GBA activator protein saposin C, accumulate GlcCer in the SC and show abnormal SC lamellar membrane structures [249]. Interestingly, deficiency of LIMP2 in AMRF patients is not associated with skin abnormalities. No prominent abnormalities have also been noted in LIMP2-deficient mice. Apparently, GCase is reaching the SC sufficiently without its regular transporting protein.
Figure 3.
The life cycle of GCase in and beyond the lysosome. AMRF: Action myoclonus renal failure syndrome, ER: endoplasmic reticulum, LB: lamellar body, LIMP2: lysosomal membrane protein 2LRO: lysosome related organelle.
GlcCer and GBA appear to be co-localized in the LB [250,251,252]. GBA activity has been observed throughout the outer parts of the epidermis [253,254,255], and recently a novel in situ method with the use of activity-based probes (ABPs) confirmed predominant localization of active GBA in the extracellular space of the SC lipid matrix [256].
GD is not the only lysosomal storage disease associated with skin barrier abnormalities. In Niemann–Pick disease a deficiency in ASMase causes an impaired conversion of SM into ceramides in the SC and, therefore, into a disturbed skin barrier [248,257]. Reduction of epidermal ASMase activity by the inhibitor imipramine causes delayed permeability barrier repair after SC injury [258].
10. Atopic Dermatitis
A common skin disease is atopic dermatitis (AD, OMIM #603165). Clinical manifestation of AD involves eczematous lesions as well as erythema, xerosis, and pruritis [259,260,261]. In AD there is a complex interplay between inflammation, genetic background, and the skin barrier. Inflammation can affect the skin barrier, and subsequent entry of compounds promotes an immune response. Additionally, it was observed that AD is associated with loss of function mutations in the filaggrin gene FLG [262,263]. As discussed in Section 7.2, filaggrin is essential for SC hydration and may affect the sensitivity of the skin [264]. Even though FLG mutations have been suggested as a predisposing factor for AD, they do not influence SC ceramide synthesis [264,265,266].
10.1. SC Lipids in AD
SC lipid metabolism and composition have been substantially studied in AD, however there is some disagreement in literature about the lipid composition in the skin of AD patients. Farwanah and co-workers reported no change in non-lesional AD skin compared to control [267], although other studies report a decrease in total ceramide level, as well as an increase in ceramide (AS) and a decrease in ceramide (EOS) and (EOH), mainly in lesional AD skin compared to control [215,266,268,269,270,271]. Additionally, Di Nardo et al. have reported a decrease in ceramide/cholesterol ratio in AD skin [271].
Besides subclass composition ceramide chain length has also been studied in AD. Some report an increase of short chain ceramides (total chain length of 34 carbon atoms) in lesional AD skin that also correlated with an increased TEWL [268,272]. Moreover, levels of ω-O-acyl-ceramides correlated negatively with TEWL [268]. A reduction in ω-O-acyl-ceramide in AD compared to control was also reported by Jungersted et al. They additionally observed no statistical difference between their FLG mutant and wild-type group in relation to the ω-O-acyl-ceramide decrease [266].
Data on fatty acids in relation to AD skin are limited, but there are a few reports reporting a reduced fatty acid chain length [273,274]. A study by van Smeden et al. described an increase of shorter fatty acids, mainly saturated fatty acids with 16 and 18 carbon atoms, as well as a reduction in fatty acids with 24 carbons or more in non-lesional AD patients [273]. However, another study observed an increased level of very long fatty acid chains in non-lesional as well as lesional AD [274]. It was hypothesized that SC ceramides and fatty acids share a common synthetic pathway, and this is consistent with the observation that ceramide composition is paralleled by the chain length of fatty acids [275].
The expression of enzymes involved in the biosynthesis of fatty acids and ceramides was related to the SC lipid composition in lesional AD skin [276]. Danso et al. observed an altered expression of GBA, ASMase, and CerS3 in lesional AD skin with a corresponding increase in ceramide (AS) and (NS) and decrease in esterified ω-hydroxy CERs. Additionally, they noted increased levels of unsaturated fatty acids and reduced levels of C22–C28 fatty acids in combination with an altered expression of stearoyl CoA desaturase (SCD) and elongase 1 (ELOVL1) [276].
10.2. Potential Role for Glucosylsphingosine in AD Pathology
Deficiency of ceramides in the SC is thought to contribute to the dry and barrier-disrupted skin of patients with AD. It was proposed that this deficiency involves a tentative novel enzyme named sphingomyelin-glucosylceramide deacylase, forming sphingosylphosphorylcholine (SPC; lysoSM) and GlcSph from SM and GlcCer. Increased deacylase activity is thought to contribute to reduced formation and subsequent deficiency of ceramide in the AD skin [277]. The deacylase enzyme is considered to be distinct from acid ceramidase as based by apparent isoelectric point [278]. Increased deacylase activity was observed for involved SC and epidermis in patients with AD [279]. Unfortunately, the deacylase has so far not been isolated and characterized. At present it cannot be excluded that the intriguing observations are explained by some neutral ceramidase, a bacterial amidase, or even acid ceramidase that in lipid-laden macrophages of GD patients shows GlcCer deacylase activity.
A common symptom in AD is pruritis. It was observed that GlcSph induces scratching in mice and more recently it was demonstrated that GlcSph activates the Serotonin Receptor 2 a and b, considered to be part of a novel itch signaling pathway [280,281].
10.3. Direct Role of GCase in AD?
As discussed above, GCase expression was found to be altered in (particularly lesional) AD skin [276]. However, no abnormality in GCase activity level in AD skin was previously noted [282]. Earlier research in mice pointed to changes in location of GCase activity in mice with a skin barrier disruption [246]. Using the specific and sensitive ABP technology, the localization of active GCase molecules in AD skin has been studied. An abnormal GCase localization in (mainly lesional) AD skin was observed together with abnormal SC lipids (Boer, submitted for publication). It will be of interest to comparably study other skin diseases. It should be stressed that abnormalities in GCase are not a sole cause for AD, however, an acquired local abnormal enzyme activity might contribute to the pathology.
11. Summary and Conclusions
This review addresses the multiple functions of the enzyme GCase that degrades the ubiquitous glycosphingolipid GlcCer. In the first part of the review, the metabolism and various functions of glycosphingolipids in health and disease are discussed. The structural features and catalytic mechanism of GCase are described, as well as its remarkable life cycle involving LIMP2-mediated transport to lysosomes. The essential cellular role of GCase in turnover of GlcCer in lysosomes is illustrated by the lysosomal storage disorder Gaucher disease (GD), which results from an inherited GCase deficiency. The review describes the variable symptoms of GD patients and the presumed underlying pathophysiological mechanisms. In addition, it addresses the presently available treatments of visceral manifestations of GD. In the second part of the review, attention is focused on another, extracellular, role of GCase in the skin. In the stratum corneum, GCase converts secreted GlcCer to ceramide, an essential component of lipid lamellae contributing to the barrier properties of the skin. A major lack of GCase activity causes a lethal skin pathology, the collodion baby.
To conclude, the catalytic ability of the enzyme GCase has been exploited in evolution for two different functions: in lysosomes, it essentially contributes to cellular glycosphingolipid metabolism, and in the extracellular space of the stratum corneum, it generates an essential building block for lipid lamellae.
Author Contributions
D.E.C.B.: concept and design, drafting article, J.v.S.: critically reviewed the article, J.A.B.: critically reviewed the article, J.M.F.G.A.: concept and design, critically reviewing article. All authors have read and agreed to the published version of the manuscript.
Funding
The work on glycosphingolipids was supported by a grant from NWO to J.M.A., (BBOL-2007247202).
Conflicts of Interest
The authors declare no conflicts of interest
References
- 1.Weinreb N.J., Brady R.O., Tappel A.L. The lysosomal localization of sphingolipid hydrolases. Biochim. Biophys. Acta. 1968;159:141–146. doi: 10.1016/0005-2744(68)90251-9. [DOI] [PubMed] [Google Scholar]
- 2.Brady R.O., Kanfer J.N., Bradley R.M., Shapiro D. Demonstration of a deficiency of glucocerebroside-cleaving enzyme in Gaucher’s disease. J. Clin. Investig. 1966;45:1112–1115. doi: 10.1172/JCI105417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gaucher P.C.E. Ph.D. Thesis. Faculté de Médecine; Paris, France: 1882. De L’epithelioma Primitif de la Rate, Hypertrophie Idiopathique de la Rate Sans Leucemie. [Google Scholar]
- 4.Beutler E., Grabowski G.A. Glucosylceramide lipidosis-Gaucher disease. In: Sriver C.R., Beaudet A.L., Sly W.S., Valle D., editors. The Metabolic and Molecular Bases of Inherited Disease. 8th ed. McGraw-Hill; New York, NY, USA: 2001. [Google Scholar]
- 5.Smith L., Mullin S., Schapira A.H.V. Insights into the structural biology of Gaucher disease. Exp. Neurol. 2017;298:180–190. doi: 10.1016/j.expneurol.2017.09.010. [DOI] [PubMed] [Google Scholar]
- 6.Sidransky E., Sherer D.M., Ginns E.I. Gaucher disease in the neonate: A distinct Gaucher phenotype is analogous to a mouse model created by targeted disruption of the glucocerebrosidase gene. Pediatr. Res. 1992;32:494–498. doi: 10.1203/00006450-199210000-00023. [DOI] [PubMed] [Google Scholar]
- 7.Tybulewicz V.L., Tremblay M.L., LaMarca M.E., Willemsen R., Stubblefield B.K., Winfield S., Zablocka B., Sidransky E., Martin B.M., Huang S.P., et al. Animal model of Gaucher’s disease from targeted disruption of the mouse glucocerebrosidase gene. Nature. 1992;357:407–410. doi: 10.1038/357407a0. [DOI] [PubMed] [Google Scholar]
- 8.Van Meer G., Wolthoorn J., Degroote S. The fate and function of glycosphingolipid glucosylceramide. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003;358:869–873. doi: 10.1098/rstb.2003.1266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Merrill A.H., Jr. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chem. Rev. 2011;111:6387–6422. doi: 10.1021/cr2002917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wennekes T., Van Den Berg R.J., Boot R.G., Van Der Marel G.A., Overkleeft H.S., Aerts J.M. Glycosphingolipids—Nature, function, and pharmacological modulation. Angew. Chem. Int. Ed. Engl. 2009;48:8848–8869. doi: 10.1002/anie.200902620. [DOI] [PubMed] [Google Scholar]
- 11.Tidhar R., Futerman A.H. The complexity of sphingolipid biosynthesis in the endoplasmic reticulum. Biochim. Biophys. Acta. 2013;1833:2511–2518. doi: 10.1016/j.bbamcr.2013.04.010. [DOI] [PubMed] [Google Scholar]
- 12.Mandon E.C., Ehses I., Rother J., Van Echten G., Sandhoff K. Subcellular localization and membrane topology of serine palmitoyltransferase, 3-dehydrosphinganine reductase, and sphinganine N-acyltransferase in mouse liver. J. Biol. Chem. 1992;267:11144–11148. [PubMed] [Google Scholar]
- 13.Levy M., Futerman A.H. Mammalian ceramide synthases. IUBMB Life. 2010;62:347–356. doi: 10.1002/iub.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lahiri S., Lee H., Mesicek J., Fuks Z., Haimovitz-Friedman A., Kolesnick R.N., Futerman A.H. Kinetic characterization of mammalian ceramide synthases: Determination of K(m) values towards sphinganine. FEBS Lett. 2007;581:5289–5294. doi: 10.1016/j.febslet.2007.10.018. [DOI] [PubMed] [Google Scholar]
- 15.Fabrias G., Munoz-Olaya J., Cingolani F., Signorelli P., Casas J., Gagliostro V., Ghidoni R. Dihydroceramide desaturase and dihydrosphingolipids: Debutant players in the sphingolipid arena. Prog. Lipid Res. 2012;51:82–94. doi: 10.1016/j.plipres.2011.12.002. [DOI] [PubMed] [Google Scholar]
- 16.Chigorno V., Riva C., Valsecchi M., Nicolini M., Brocca P., Sonnino S. Metabolic processing of gangliosides by human fibroblasts in culture—Formation and recycling of separate pools of sphingosine. Eur. J. Biochem. 1997;250:661–669. doi: 10.1111/j.1432-1033.1997.00661.x. [DOI] [PubMed] [Google Scholar]
- 17.Kitatani K., Idkowiak-Baldys J., Hannun Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell. Signal. 2008;20:1010–1018. doi: 10.1016/j.cellsig.2007.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.D’Angelo G., Capasso S., Sticco L., Russo D. Glycosphingolipids: Synthesis and functions. FEBS J. 2013;280:6338–6353. doi: 10.1111/febs.12559. [DOI] [PubMed] [Google Scholar]
- 19.Hanada K., Kumagai K., Yasuda S., Miura Y., Kawano M., Fukasawa M., Nishijima M. Molecular machinery for non-vesicular trafficking of ceramide. Nature. 2003;426:803–809. doi: 10.1038/nature02188. [DOI] [PubMed] [Google Scholar]
- 20.Ichikawa S., Sakiyama H., Suzuki G., Hidari K.I., Hirabayashi Y. Expression cloning of a cDNA for human ceramide glucosyltransferase that catalyzes the first glycosylation step of glycosphingolipid synthesis. Proc. Natl. Acad. Sci. USA. 1996;93:4638–4643. doi: 10.1073/pnas.93.10.4638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Van Weely S., Brandsma M., Strijland A., Tager J.M., Aerts J.M. Demonstration of the existence of a second, non-lysosomal glucocerebrosidase that is not deficient in Gaucher disease. Biochim. Biophys. Acta. 1993;1181:55–62. doi: 10.1016/0925-4439(93)90090-N. [DOI] [PubMed] [Google Scholar]
- 22.Sandhoff R., Sandhoff K. Emerging concepts of ganglioside metabolism. FEBS Lett. 2018;592:3835–3864. doi: 10.1002/1873-3468.13114. [DOI] [PubMed] [Google Scholar]
- 23.Mukherjee S., Maxfield F.R. Membrane domains. Annu. Rev. Cell Dev. Biol. 2004;20:839–866. doi: 10.1146/annurev.cellbio.20.010403.095451. [DOI] [PubMed] [Google Scholar]
- 24.Sonnino S., Prinetti A. Membrane domains and the “lipid raft” concept. Curr. Med. Chem. 2013;20:4–21. [PubMed] [Google Scholar]
- 25.Lingwood D., Simons K. Lipid rafts as a membrane-organizing principle. Science. 2010;327:46–50. doi: 10.1126/science.1174621. [DOI] [PubMed] [Google Scholar]
- 26.Tagami S., Inokuchi Ji J., Kabayama K., Yoshimura H., Kitamura F., Uemura S., Ogawa C., Ishii A., Saito M., Ohtsuka Y., et al. Ganglioside GM3 participates in the pathological conditions of insulin resistance. J. Biol. Chem. 2002;277:3085–3092. doi: 10.1074/jbc.M103705200. [DOI] [PubMed] [Google Scholar]
- 27.Langeveld M., Aerts J.M. Glycosphingolipids and insulin resistance. Prog. Lipid Res. 2009;48:196–205. doi: 10.1016/j.plipres.2009.03.002. [DOI] [PubMed] [Google Scholar]
- 28.Kabayama K., Sato T., Saito K., Loberto N., Prinetti A., Sonnino S., Kinjo M., Igarashi Y., Inokuchi J. Dissociation of the insulin receptor and caveolin-1 complex by ganglioside GM3 in the state of insulin resistance. Proc. Natl. Acad. Sci. USA. 2007;104:13678–13683. doi: 10.1073/pnas.0703650104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yamashita T., Hashiramoto A., Haluzik M., Mizukami H., Beck S., Norton A., Kono M., Tsuji S., Daniotti J.L., Werth N., et al. Enhanced insulin sensitivity in mice lacking ganglioside GM3. Proc. Natl. Acad. Sci. USA. 2003;100:3445–3449. doi: 10.1073/pnas.0635898100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Aerts J.M., Ottenhoff R., Powlson A.S., Grefhorst A., Van Eijk M., Dubbelhuis P.F., Aten J., Kuipers F., Serlie M.J., Wennekes T., et al. Pharmacological inhibition of glucosylceramide synthase enhances insulin sensitivity. Diabetes. 2007;56:1341–1349. doi: 10.2337/db06-1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Coskun U., Grzybek M., Drechsel D., Simons K. Regulation of human EGF receptor by lipids. Proc. Natl. Acad. Sci. USA. 2011;108:9044–9048. doi: 10.1073/pnas.1105666108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nakayama H., Nagafuku M., Suzuki A., Iwabuchi K., Inokuchi J.I. The regulatory roles of glycosphingolipid-enriched lipid rafts in immune systems. FEBS Lett. 2018;592:3921–3942. doi: 10.1002/1873-3468.13275. [DOI] [PubMed] [Google Scholar]
- 33.Hanada K. Sphingolipids in infectious diseases. Jpn. J. Infect. Dis. 2005;58:131–148. [PubMed] [Google Scholar]
- 34.Aerts J., Artola M., Van Eijk M., Ferraz M.J., Boot R.G. Glycosphingolipids and infection. Potential new therapeutic avenues. Front. Cell Dev. Biol. 2019;7:324. doi: 10.3389/fcell.2019.00324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Inokuchi J.I., Inamori K.I., Kabayama K., Nagafuku M., Uemura S., Go S., Suzuki A., Ohno I., Kanoh H., Shishido F. Biology of GM3 ganglioside. Prog. Mol. Biol. Transl. Sci. 2018;156:151–195. doi: 10.1016/bs.pmbts.2017.10.004. [DOI] [PubMed] [Google Scholar]
- 36.Iwabuchi K. Gangliosides in the immune system: Role of glycosphingolipids and glycosphingolipid-enriched lipid rafts in immunological functions. Methods Mol. Biol. 2018;1804:83–95. doi: 10.1007/978-1-4939-8552-4_4. [DOI] [PubMed] [Google Scholar]
- 37.Aerts J., Kuo C.L., Lelieveld L.T., Boer D.E.C., Van Der Lienden M.J.C., Overkleeft H.S., Artola M. Glycosphingolipids and lysosomal storage disorders as illustrated by gaucher disease. Curr. Opin. Chem. Biol. 2019;53:204–215. doi: 10.1016/j.cbpa.2019.10.006. [DOI] [PubMed] [Google Scholar]
- 38.Nagata M., Izumi Y., Ishikawa E., Kiyotake R., Doi R., Iwai S., Omahdi Z., Yamaji T., Miyamoto T., Bamba T., et al. Intracellular metabolite beta-glucosylceramide is an endogenous Mincle ligand possessing immunostimulatory activity. Proc. Natl. Acad. Sci. USA. 2017;114:E3285–E3294. doi: 10.1073/pnas.1618133114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Van Den Bergh F.A., Tager J.M. Localization of neutral glycosphingolipids in human plasma. Biochim. Biophys. Acta. 1976;441:391–402. doi: 10.1016/0005-2760(76)90236-8. [DOI] [PubMed] [Google Scholar]
- 40.Ghauharali K., Kallemeijn W., Vergeer M., Motazacker M., Van Eijk M., Aerts H., Groener A. The role of ABCA1 in glycosphingolipid trafficking and efflux. Chem. Phys. Lipids. 2009;160:S15. doi: 10.1016/j.chemphyslip.2009.06.134. [DOI] [Google Scholar]
- 41.Kolter T., Sandhoff K. Lysosomal degradation of membrane lipids. FEBS Lett. 2010;584:1700–1712. doi: 10.1016/j.febslet.2009.10.021. [DOI] [PubMed] [Google Scholar]
- 42.Park J.H., Schuchman E.H. Acid ceramidase and human disease. Biochim. Biophys. Acta. 2006;1758:2133–2138. doi: 10.1016/j.bbamem.2006.08.019. [DOI] [PubMed] [Google Scholar]
- 43.Pyne S., Adams D.R., Pyne N.J. Sphingosine 1-phosphate and sphingosine kinases in health and disease: Recent advances. Prog. Lipid Res. 2016;62:93–106. doi: 10.1016/j.plipres.2016.03.001. [DOI] [PubMed] [Google Scholar]
- 44.Aerts J.M., Hollak C., Boot R., Groener A. Biochemistry of glycosphingolipid storage disorders: Implications for therapeutic intervention. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003;358:905–914. doi: 10.1098/rstb.2003.1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ben Bdira F., Artola M., Overkleeft H.S., Ubbink M., Aerts J. Distinguishing the differences in beta-glycosylceramidase folds, dynamics, and actions informs therapeutic uses. J. Lipid Res. 2018;59:2262–2276. doi: 10.1194/jlr.R086629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Brumshtein B., Greenblatt H.M., Butters T.D., Shaaltiel Y., Aviezer D., Silman I., Futerman A.H., Sussman J.L. Crystal structures of complexes of N-butyl- and N-nonyl-deoxynojirimycin bound to acid beta-glucosidase: Insights into the mechanism of chemical chaperone action in Gaucher disease. J. Biol. Chem. 2007;282:29052–29058. doi: 10.1074/jbc.M705005200. [DOI] [PubMed] [Google Scholar]
- 47.Dvir H., Harel M., McCarthy A.A., Toker L., Silman I., Futerman A.H., Sussman J.L. X-ray structure of human acid-beta-glucosidase, the defective enzyme in Gaucher disease. EMBO Rep. 2003;4:704–709. doi: 10.1038/sj.embor.embor873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Koshland D.E. Stereochemistry and the mechanism of enzymatic reactions. Biol. Rev. 1953;28:416–436. doi: 10.1111/j.1469-185X.1953.tb01386.x. [DOI] [Google Scholar]
- 49.Rye C.S., Withers S.G. Glycosidase mechanisms. Curr. Opin. Chem. Biol. 2000;4:573–580. doi: 10.1016/S1367-5931(00)00135-6. [DOI] [PubMed] [Google Scholar]
- 50.Speciale G., Thompson A.J., Davies G.J., Williams S.J. Dissecting conformational contributions to glycosidase catalysis and inhibition. Curr. Opin. Struct. Biol. 2014;28:1–13. doi: 10.1016/j.sbi.2014.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ardevol A., Rovira C. Reaction mechanisms in carbohydrate-active enzymes: Glycoside hydrolases and glycosyltransferases. Insights from ab initio quantum mechanics/molecular mechanics dynamic simulations. J. Am. Chem. Soc. 2015;137:7528–7547. doi: 10.1021/jacs.5b01156. [DOI] [PubMed] [Google Scholar]
- 52.Legler G. Glycoside hydrolases: Mechanistic information from studies with reversible and irreversible inhibitors. Adv. Carbohydr. Chem. Biochem. 1990;48:319–384. doi: 10.1016/s0065-2318(08)60034-7. [DOI] [PubMed] [Google Scholar]
- 53.Withers S.G., Aebersold R. Approaches to labeling and identification of active site residues in glycosidases. Protein Sci. 1995;4:361–372. doi: 10.1002/pro.5560040302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Atsumi S., Umezawa K., Iinuma H., Naganawa H., Nakamura H., Iitaka Y., Takeuchi T. Production, isolation and structure determination of a novel beta-glucosidase inhibitor, cyclophellitol, from Phellinus sp. J. Antibiot. 1990;43:49–53. doi: 10.7164/antibiotics.43.49. [DOI] [PubMed] [Google Scholar]
- 55.Kallemeijn W.W., Li K.Y., Witte M.D., Marques A.R., Aten J., Scheij S., Jiang J., Willems L.I., Voorn-Brouwer T.M., Van Roomen C.P., et al. Novel activity-based probes for broad-spectrum profiling of retaining beta-exoglucosidases in situ and in vivo. Angew. Chem. Int. Ed. Engl. 2012;51:12529–12533. doi: 10.1002/anie.201207771. [DOI] [PubMed] [Google Scholar]
- 56.Artola M., Kuo C.L., Lelieveld L.T., Rowland R.J., Van Der Marel G.A., Codee J.D.C., Boot R.G., Davies G.J., Aerts J., Overkleeft H.S. Functionalized cyclophellitols are selective glucocerebrosidase inhibitors and induce a bona fide neuropathic Gaucher model in zebrafish. J. Am. Chem. Soc. 2019;141:4214–4218. doi: 10.1021/jacs.9b00056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kuo C.L., Kallemeijn W.W., Lelieveld L.T., Mirzaian M., Zoutendijk I., Vardi A., Futerman A.H., Meijer A.H., Spaink H.P., Overkleeft H.S., et al. In vivo inactivation of glycosidases by conduritol B epoxide and cyclophellitol as revealed by activity-based protein profiling. FEBS J. 2019;286:584–600. doi: 10.1111/febs.14744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Witte M.D., Kallemeijn W.W., Aten J., Li K.Y., Strijland A., Donker-Koopman W.E., Van Den Nieuwendijk A.M., Bleijlevens B., Kramer G., Florea B.I., et al. Ultrasensitive in situ visualization of active glucocerebrosidase molecules. Nat. Chem. Biol. 2010;6:907–913. doi: 10.1038/nchembio.466. [DOI] [PubMed] [Google Scholar]
- 59.Jiang J., Beenakker T.J., Kallemeijn W.W., Van Der Marel G.A., Van Den Elst H., Codee J.D., Aerts J.M., Overkleeft H.S. Comparing cyclophellitol N-alkyl and N-acyl cyclophellitol aziridines as activity-based glycosidase probes. Chemistry. 2015;21:10861–10869. doi: 10.1002/chem.201501313. [DOI] [PubMed] [Google Scholar]
- 60.Willems L.I., Beenakker T.J., Murray B., Scheij S., Kallemeijn W.W., Boot R.G., Verhoek M., Donker-Koopman W.E., Ferraz M.J., Van Rijssel E.R., et al. Potent and selective activity-based probes for GH27 human retaining alpha-galactosidases. J. Am. Chem. Soc. 2014;136:11622–11625. doi: 10.1021/ja507040n. [DOI] [PubMed] [Google Scholar]
- 61.Jiang J., Kuo C.L., Wu L., Franke C., Kallemeijn W.W., Florea B.I., Van Meel E., Van Der Marel G.A., Codee J.D., Boot R.G., et al. Detection of active mammalian GH31 alpha-glucosidases in health and disease using in-class, broad-spectrum activity-based probes. ACS Cent. Sci. 2016;2:351–358. doi: 10.1021/acscentsci.6b00057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jiang J., Kallemeijn W.W., Wright D.W., Van Den Nieuwendijk A., Rohde V.C., Folch E.C., Van Den Elst H., Florea B.I., Scheij S., Donker-Koopman W.E., et al. In vitro and in vivo comparative and competitive activity-based protein profiling of GH29 alpha-l-fucosidases. Chem. Sci. 2015;6:2782–2789. doi: 10.1039/C4SC03739A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Artola M., Kuo C.L., McMahon S.A., Oehler V., Hansen T., Van Der Lienden M., He X., Van Den Elst H., Florea B.I., Kermode A.R., et al. New irreversible alpha-l-iduronidase inhibitors and activity-based probes. Chemistry. 2018;24:19081–19088. doi: 10.1002/chem.201804662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wu L., Jiang J., Jin Y., Kallemeijn W.W., Kuo C.L., Artola M., Dai W., Van Elk C., Van Eijk M., Van Der Marel G.A., et al. Activity-based probes for functional interrogation of retaining beta-glucuronidases. Nat. Chem. Biol. 2017;13:867–873. doi: 10.1038/nchembio.2395. [DOI] [PubMed] [Google Scholar]
- 65.Marques A.R., Willems L.I., Herrera Moro D., Florea B.I., Scheij S., Ottenhoff R., Van Roomen C.P., Verhoek M., Nelson J.K., Kallemeijn W.W., et al. A specific activity-based probe to monitor family GH59 galactosylceramidase, the enzyme deficient in Krabbe disease. Chembiochem. 2017;18:402–412. doi: 10.1002/cbic.201600561. [DOI] [PubMed] [Google Scholar]
- 66.Kuo C.L., Van Meel E., Kytidou K., Kallemeijn W.W., Witte M., Overkleeft H.S., Artola M.E., Aerts J.M. Activity-based probes for glycosidases: Profiling and other applications. Methods Enzymol. 2018;598:217–235. doi: 10.1016/bs.mie.2017.06.039. [DOI] [PubMed] [Google Scholar]
- 67.Lahav D., Liu B., Van Den Berg R., Van Den Nieuwendijk A., Wennekes T., Ghisaidoobe A.T., Breen I., Ferraz M.J., Kuo C.L., Wu L., et al. A fluorescence polarization activity-based protein profiling assay in the discovery of potent, selective inhibitors for human nonlysosomal glucosylceramidase. J. Am. Chem. Soc. 2017;139:14192–14197. doi: 10.1021/jacs.7b07352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tylki-Szymanska A., Groener J.E., Kaminski M.L., Lugowska A., Jurkiewicz E., Czartoryska B. Gaucher disease due to saposin C deficiency, previously described as non-neuronopathic form—No positive effects after 2-years of miglustat therapy. Mol. Genet. Metab. 2011;104:627–630. doi: 10.1016/j.ymgme.2011.09.010. [DOI] [PubMed] [Google Scholar]
- 69.Ben Bdira F., Kallemeijn W.W., Oussoren S.V., Scheij S., Bleijlevens B., Florea B.I., Van Roomen C., Ottenhoff R., Van Kooten M., Walvoort M.T.C., et al. Stabilization of glucocerebrosidase by active site occupancy. ACS Chem. Biol. 2017;12:1830–1841. doi: 10.1021/acschembio.7b00276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jonsson L.M., Murray G.J., Sorrell S.H., Strijland A., Aerts J.F., Ginns E.I., Barranger J.A., Tager J.M., Schram A.W. Biosynthesis and maturation of glucocerebrosidase in Gaucher fibroblasts. Eur. J. Biochem. 1987;164:171–179. doi: 10.1111/j.1432-1033.1987.tb11008.x. [DOI] [PubMed] [Google Scholar]
- 71.Aerts J.M., Schram A.W., Strijland A., Van Weely S., Jonsson L.M., Tager J.M., Sorrell S.H., Ginns E.I., Barranger J.A., Murray G.J. Glucocerebrosidase, a lysosomal enzyme that does not undergo oligosaccharide phosphorylation. Biochim. Biophys. Acta. 1988;964:303–308. doi: 10.1016/0304-4165(88)90030-X. [DOI] [PubMed] [Google Scholar]
- 72.Reczek D., Schwake M., Schroder J., Hughes H., Blanz J., Jin X., Brondyk W., Van Patten S., Edmunds T., Saftig P. LIMP-2 is a receptor for lysosomal mannose-6-phosphate-independent targeting of beta-glucocerebrosidase. Cell. 2007;131:770–783. doi: 10.1016/j.cell.2007.10.018. [DOI] [PubMed] [Google Scholar]
- 73.Rijnboutt S., Aerts H.M., Geuze H.J., Tager J.M., Strous G.J. Mannose 6-phosphate-independent membrane association of cathepsin D, glucocerebrosidase, and sphingolipid-activating protein in HepG2 cells. J. Biol. Chem. 1991;266:4862–4868. [PubMed] [Google Scholar]
- 74.Rijnboutt S., Kal A.J., Geuze H.J., Aerts H., Strous G.J. Mannose 6-phosphate-independent targeting of cathepsin D to lysosomes in HepG2 cells. J. Biol. Chem. 1991;266:23586–23592. [PubMed] [Google Scholar]
- 75.Zunke F., Andresen L., Wesseler S., Groth J., Arnold P., Rothaug M., Mazzulli J.R., Krainc D., Blanz J., Saftig P., et al. Characterization of the complex formed by beta-glucocerebrosidase and the lysosomal integral membrane protein type-2. Proc. Natl. Acad. Sci. USA. 2016;113:3791–3796. doi: 10.1073/pnas.1514005113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Balreira A., Gaspar P., Caiola D., Chaves J., Beirao I., Lima J.L., Azevedo J.E., Miranda M.C. A nonsense mutation in the LIMP-2 gene associated with progressive myoclonic epilepsy and nephrotic syndrome. Hum. Mol. Genet. 2008;17:2238–2243. doi: 10.1093/hmg/ddn124. [DOI] [PubMed] [Google Scholar]
- 77.Gaspar P., Kallemeijn W.W., Strijland A., Scheij S., Van Eijk M., Aten J., Overkleeft H.S., Balreira A., Zunke F., Schwake M., et al. Action myoclonus-renal failure syndrome: Diagnostic applications of activity-based probes and lipid analysis. J. Lipid Res. 2014;55:138–145. doi: 10.1194/jlr.M043802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Jian J., Zhao S., Tian Q.Y., Liu H., Zhao Y., Chen W.C., Grunig G., Torres P.A., Wang B.C., Zeng B., et al. Association between progranulin and Gaucher disease. EBioMedicine. 2016;11:127–137. doi: 10.1016/j.ebiom.2016.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Chen Y., Sud N., Hettinghouse A., Liu C.J. Molecular regulations and therapeutic targets of Gaucher disease. Cytokine Growth Factor Rev. 2018;41:65–74. doi: 10.1016/j.cytogfr.2018.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Jian J., Tian Q.Y., Hettinghouse A., Zhao S., Liu H., Wei J., Grunig G., Zhang W., Setchell K.D.R., Sun Y., et al. Progranulin recruits HSP70 to beta-glucocerebrosidase and Is therapeutic against Gaucher disease. EBioMedicine. 2016;13:212–224. doi: 10.1016/j.ebiom.2016.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Tan Y.L., Genereux J.C., Pankow S., Aerts J.M., Yates J.R., III, Kelly J.W. ERdj3 is an endoplasmic reticulum degradation factor for mutant glucocerebrosidase variants linked to Gaucher’s disease. Chem. Biol. 2014;21:967–976. doi: 10.1016/j.chembiol.2014.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Aghion H. Ph.D. Thesis. Faculte de Medecine; Paris, France: 1934. La Maladie de Gaucher dans L’enfance. [Google Scholar]
- 83.Rosenberg A., Chargaff E. A reinvestigation of the cerebroside deposited in Gaucher’s disease. J. Biol. Chem. 1958;233:1323–1326. [PubMed] [Google Scholar]
- 84.Aerts J.M., Cox T.M., Roscoe O. Brady: Physician whose pioneering discoveries in lipid biochemistry revolutionized treatment and understanding of lysosomal diseases. Blood Cells Mol. Dis. 2018;68:4–8. doi: 10.1016/j.bcmd.2016.10.030. [DOI] [PubMed] [Google Scholar]
- 85.Danby P.M., Withers S.G. Advances in enzymatic glycoside synthesis. ACS Chem. Biol. 2016;11:1784–1794. doi: 10.1021/acschembio.6b00340. [DOI] [PubMed] [Google Scholar]
- 86.Marques A.R., Mirzaian M., Akiyama H., Wisse P., Ferraz M.J., Gaspar P., Ghauharali-van der Vlugt K., Meijer R., Giraldo P., Alfonso P., et al. Glucosylated cholesterol in mammalian cells and tissues: Formation and degradation by multiple cellular beta-glucosidases. J. Lipid Res. 2016;57:451–463. doi: 10.1194/jlr.M064923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Akiyama H., Kobayashi S., Hirabayashi Y., Murakami-Murofushi K. Cholesterol glucosylation is catalyzed by transglucosylation reaction of beta-glucosidase 1. Biochem. Biophys. Res. Commun. 2013;441:838–843. doi: 10.1016/j.bbrc.2013.10.145. [DOI] [PubMed] [Google Scholar]
- 88.Akiyama H., Hirabayashi Y. A novel function for glucocerebrosidase as a regulator of sterylglucoside metabolism. Biochim. Biophys. Acta Gen. Subj. 2017;1861:2507–2514. doi: 10.1016/j.bbagen.2017.06.003. [DOI] [PubMed] [Google Scholar]
- 89.Ballabio A., Gieselmann V. Lysosomal disorders: From storage to cellular damage. Biochim. Biophys. Acta. 2009;1793:684–696. doi: 10.1016/j.bbamcr.2008.12.001. [DOI] [PubMed] [Google Scholar]
- 90.Platt F.M., d’Azzo A., Davidson B.L., Neufeld E.F., Tifft C.J. Lysosomal storage diseases. Nat. Rev. Dis. Primers. 2018;4:27. doi: 10.1038/s41572-018-0025-4. [DOI] [PubMed] [Google Scholar]
- 91.Ferraz M.J., Kallemeijn W.W., Mirzaian M., Herrera Moro D., Marques A., Wisse P., Boot R.G., Willems L.I., Overkleeft H.S., Aerts J.M. Gaucher disease and Fabry disease: New markers and insights in pathophysiology for two distinct glycosphingolipidoses. Biochim. Biophys. Acta. 2014;1841:811–825. doi: 10.1016/j.bbalip.2013.11.004. [DOI] [PubMed] [Google Scholar]
- 92.Horowitz M., Wilder S., Horowitz Z., Reiner O., Gelbart T., Beutler E. The human glucocerebrosidase gene and pseudogene: Structure and evolution. Genomics. 1989;4:87–96. doi: 10.1016/0888-7543(89)90319-4. [DOI] [PubMed] [Google Scholar]
- 93.Ohashi T., Hong C.M., Weiler S., Tomich J.M., Aerts J.M., Tager J.M., Barranger J.A. Characterization of human glucocerebrosidase from different mutant alleles. J. Biol. Chem. 1991;266:3661–3667. [PubMed] [Google Scholar]
- 94.Boot R.G., Hollak C.E., Verhoek M., Sloof P., Poorthuis B.J., Kleijer W.J., Wevers R.A., Van Oers M.H., Mannens M.M., Aerts J.M., et al. Glucocerebrosidase genotype of Gaucher patients in The Netherlands: Limitations in prognostic value. Hum. Mutat. 1997;10:348–358. doi: 10.1002/(SICI)1098-1004(1997)10:5<348::AID-HUMU3>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 95.Diamond J.M. Human genetics. Jewish lysosomes. Nature. 1994;368:291–292. doi: 10.1038/368291a0. [DOI] [PubMed] [Google Scholar]
- 96.Boas F.E. Linkage to Gaucher mutations in the Ashkenazi population: Effect of drift on decay of linkage disequilibrium and evidence for heterozygote selection. Blood Cells Mol. Dis. 2000;26:348–359. doi: 10.1006/bcmd.2000.0314. [DOI] [PubMed] [Google Scholar]
- 97.Colombo R. Age estimate of the N370S mutation causing Gaucher disease in Ashkenazi Jews and European populations: A reappraisal of haplotype data. Am. J. Hum. Genet. 2000;66:692–697. doi: 10.1086/302757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Meijer A.H., Aerts J.M. Linking smokers’ susceptibility to tuberculosis with lysosomal storage disorders. Dev. Cell. 2016;37:112–113. doi: 10.1016/j.devcel.2016.04.004. [DOI] [PubMed] [Google Scholar]
- 99.Sidransky E. Gaucher disease: Complexity in a “simple” disorder. Mol. Genet. Metab. 2004;83:6–15. doi: 10.1016/j.ymgme.2004.08.015. [DOI] [PubMed] [Google Scholar]
- 100.Lachmann R.H., Grant I.R., Halsall D., Cox T.M. Twin pairs showing discordance of phenotype in adult Gaucher’s disease. QJM. 2004;97:199–204. doi: 10.1093/qjmed/hch036. [DOI] [PubMed] [Google Scholar]
- 101.Biegstraaten M., Van Schaik I.N., Aerts J.M., Langeveld M., Mannens M.M., Bour L.J., Sidransky E., Tayebi N., Fitzgibbon E., Hollak C.E. A monozygotic twin pair with highly discordant Gaucher phenotypes. Blood Cells Mol. Dis. 2011;46:39–41. doi: 10.1016/j.bcmd.2010.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Uyama E., Takahashi K., Owada M., Okamura R., Naito M., Tsuji S., Kawasaki S., Araki S. Hydrocephalus, corneal opacities, deafness, valvular heart disease, deformed toes and leptomeningeal fibrous thickening in adult siblings: A new syndrome associated with beta-glucocerebrosidase deficiency and a mosaic population of storage cells. Acta Neurol. Scand. 1992;86:407–420. doi: 10.1111/j.1600-0404.1992.tb05109.x. [DOI] [PubMed] [Google Scholar]
- 103.Chabas A., Cormand B., Grinberg D., Burguera J.M., Balcells S., Merino J.L., Mate I., Sobrino J.A., Gonzalez-Duarte R., Vilageliu L. Unusual expression of Gaucher’s disease: Cardiovascular calcifications in three sibs homozygous for the D409H mutation. J. Med. Genet. 1995;32:740–742. doi: 10.1136/jmg.32.9.740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Abrahamov A., Elstein D., Gross-Tsur V., Farber B., Glaser Y., Hadas-Halpern I., Ronen S., Tafakjdi M., Horowitz M., Zimran A. Gaucher’s disease variant characterised by progressive calcification of heart valves and unique genotype. Lancet. 1995;346:1000–1003. doi: 10.1016/S0140-6736(95)91688-1. [DOI] [PubMed] [Google Scholar]
- 105.Zhang C.K., Stein P.B., Liu J., Wang Z., Yang R., Cho J.H., Gregersen P.K., Aerts J.M., Zhao H., Pastores G.M., et al. Genome-wide association study of N370S homozygous Gaucher disease reveals the candidacy of CLN8 gene as a genetic modifier contributing to extreme phenotypic variation. Am. J. Hematol. 2012;87:377–383. doi: 10.1002/ajh.23118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Di Ronza A., Bajaj L., Sharma J., Sanagasetti D., Lotfi P., Adamski C.J., Collette J., Palmieri M., Amawi A., Popp L., et al. CLN8 is an endoplasmic reticulum cargo receptor that regulates lysosome biogenesis. Nat. Cell Biol. 2018;20:1370–1377. doi: 10.1038/s41556-018-0228-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Velayati A., DePaolo J., Gupta N., Choi J.H., Moaven N., Westbroek W., Goker-Alpan O., Goldin E., Stubblefield B.K., Kolodny E., et al. A mutation in SCARB2 is a modifier in Gaucher disease. Hum. Mutat. 2011;32:1232–1238. doi: 10.1002/humu.21566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Alfonso P., Navascues J., Navarro S., Medina P., Bolado-Carrancio A., Andreu V., Irun P., Rodriguez-Rey J.C., Pocovi M., Espana F., et al. Characterization of variants in the glucosylceramide synthase gene and their association with type 1 Gaucher disease severity. Hum. Mutat. 2013;34:1396–1403. doi: 10.1002/humu.22381. [DOI] [PubMed] [Google Scholar]
- 109.Siebert M., Westbroek W., Chen Y.C., Moaven N., Li Y., Velayati A., Saraiva-Pereira M.L., Martin S.E., Sidransky E. Identification of miRNAs that modulate glucocerebrosidase activity in Gaucher disease cells. RNA Biol. 2014;11:1291–1300. doi: 10.1080/15476286.2014.996085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sidransky E., Nalls M.A., Aasly J.O., Aharon-Peretz J., Annesi G., Barbosa E.R., Bar-Shira A., Berg D., Bras J., Brice A., et al. Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N. Engl. J. Med. 2009;361:1651–1661. doi: 10.1056/NEJMoa0901281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Nalls M.A., Duran R., Lopez G., Kurzawa-Akanbi M., McKeith I.G., Chinnery P.F., Morris C.M., Theuns J., Crosiers D., Cras P., et al. A multicenter study of glucocerebrosidase mutations in dementia with Lewy bodies. JAMA Neurol. 2013;70:727–735. doi: 10.1001/jamaneurol.2013.1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Siebert M., Sidransky E., Westbroek W. Glucocerebrosidase is shaking up the synucleinopathies. Brain. 2014;137:1304–1322. doi: 10.1093/brain/awu002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Mullin S., Beavan M., Bestwick J., McNeill A., Proukakis C., Cox T., Hughes D., Mehta A., Zetterberg H., Schapira A.H.V. Evolution and clustering of prodromal parkinsonian features in GBA1 carriers. Mov. Disord. 2019;34:1365–1373. doi: 10.1002/mds.27775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Isacson O., Brekk O.R., Hallett P.J. Novel results and concepts emerging from lipid cell biology relevant to degenerative brain aging and disease. Front. Neurol. 2019;10:1053. doi: 10.3389/fneur.2019.01053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Rocha E.M., Smith G.A., Park E., Cao H., Brown E., Hallett P., Isacson O. Progressive decline of glucocerebrosidase in aging and Parkinson’s disease. Ann. Clin. Transl. Neurol. 2015;2:433–438. doi: 10.1002/acn3.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Hallett P.J., Huebecker M., Brekk O.R., Moloney E.B., Rocha E.M., Priestman D.A., Platt F.M., Isacson O. Glycosphingolipid levels and glucocerebrosidase activity are altered in normal aging of the mouse brain. Neurobiol. Aging. 2018;67:189–200. doi: 10.1016/j.neurobiolaging.2018.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Huebecker M., Moloney E.B., Van Der Spoel A.C., Priestman D.A., Isacson O., Hallett P.J., Platt F.M. Reduced sphingolipid hydrolase activities, substrate accumulation and ganglioside decline in Parkinson’s disease. Mol. Neurodegener. 2019;14:40. doi: 10.1186/s13024-019-0339-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Rocha E.M., Smith G.A., Park E., Cao H., Graham A.R., Brown E., McLean J.R., Hayes M.A., Beagan J., Izen S.C., et al. Sustained systemic glucocerebrosidase inhibition induces brain alpha-synuclein aggregation, microglia and complement C1q activation in mice. Antioxid. Redox Signal. 2015;23:550–564. doi: 10.1089/ars.2015.6307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Brekk O.R., Moskites A., Isacson O., Hallett P.J. Lipid-dependent deposition of alpha-synuclein and Tau on neuronal Secretogranin II-positive vesicular membranes with age. Sci. Rep. 2018;8:15207. doi: 10.1038/s41598-018-33474-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Xu Y.H., Sun Y., Ran H., Quinn B., Witte D., Grabowski G.A. Accumulation and distribution of alpha-synuclein and ubiquitin in the CNS of Gaucher disease mouse models. Mol. Genet. Metab. 2011;102:436–447. doi: 10.1016/j.ymgme.2010.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sardi S.P., Clarke J., Kinnecom C., Tamsett T.J., Li L., Stanek L.M., Passini M.A., Grabowski G.A., Schlossmacher M.G., Sidman R.L., et al. Expression of glucocerebrosidase corrects alpha-synuclein pathology and memory in a mouse model of Gaucher-related synucleinopathy. Proc. Natl. Acad. Sci. USA. 2011;108:12101–12106. doi: 10.1073/pnas.1108197108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sardi S.P., Viel C., Clarke J., Treleaven C.M., Richards A.M., Park H., Olszewski M.A., Dodge J.C., Marshall J., Makino E., et al. Glucosylceramide synthase inhibition alleviates aberrations in synucleinopathy models. Proc. Natl. Acad. Sci. USA. 2017;114:2699–2704. doi: 10.1073/pnas.1616152114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Mazzulli J.R., Xu Y.H., Sun Y., Knight A.L., McLean P.J., Caldwell G.A., Sidransky E., Grabowski G.A., Krainc D. Gaucher disease glucocerebrosidase and α-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell. 2011;146:37–52. doi: 10.1016/j.cell.2011.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Mazzulli J.R., Zunke F., Tsunemi T., Toker N.J., Jeon S., Burbulla L.F., Patnaik S., Sidransky E., Marugan J.J., Sue C.M., et al. Activation of β-glucocerebrosidase reduces pathological α-synuclein and restores lysosomal function in Parkinson’s patient midbrain neurons. J. Neurosci. 2016;36:7693–7706. doi: 10.1523/JNEUROSCI.0628-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Stojkovska I., Krainc D., Mazzulli J.R. Molecular mechanisms of α-synuclein and GBA1 in Parkinson’s disease. Cell Tissue Res. 2018;373:51–60. doi: 10.1007/s00441-017-2704-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Yap T.L., Gruschus J.M., Velayati A., Westbroek W., Goldin E., Moaven N., Sidransky E., Lee J.C. Alpha-synuclein interacts with Glucocerebrosidase providing a molecular link between Parkinson and Gaucher diseases. J. Biol. Chem. 2011;286:28080–28088. doi: 10.1074/jbc.M111.237859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Toffoli M., Smith L., Schapira A.H.V. The biochemical basis of interactions between Glucocerebrosidase and alpha-synuclein in GBA1 mutation carriers. J. Neurochem. 2020:e14968. doi: 10.1111/jnc.14968. [DOI] [PubMed] [Google Scholar]
- 128.Balestrino R., Schapira A.H.V. Parkinson disease. Eur. J. Neurol. 2020;27:27–42. doi: 10.1111/ene.14108. [DOI] [PubMed] [Google Scholar]
- 129.Maor G., Cabasso O., Krivoruk O., Rodriguez J., Steller H., Segal D., Horowitz M. The contribution of mutant GBA to the development of Parkinson disease in Drosophila. Hum. Mol. Genet. 2016;25:2712–2727. doi: 10.1093/hmg/ddw129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Cabasso O., Paul S., Dorot O., Maor G., Krivoruk O., Pasmanik-Chor M., Mirzaian M., Ferraz M., Aerts J., Horowitz M. Drosophila melanogaster mutated in its GBA1b ortholog recapitulates neuronopathic Gaucher disease. J. Clin. Med. 2019;8:1420. doi: 10.3390/jcm8091420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Bussink A.P., Van Eijk M., Renkema G.H., Aerts J.M., Boot R.G. The biology of the Gaucher cell: The cradle of human chitinases. Int. Rev. Cytol. 2006;252:71–128. doi: 10.1016/S0074-7696(06)52001-7. [DOI] [PubMed] [Google Scholar]
- 132.Boven L.A., Van Meurs M., Boot R.G., Mehta A., Boon L., Aerts J.M., Laman J.D. Gaucher cells demonstrate a distinct macrophage phenotype and resemble alternatively activated macrophages. Am. J. Clin. Pathol. 2004;122:359–369. doi: 10.1309/BG5VA8JRDQH1M7HN. [DOI] [PubMed] [Google Scholar]
- 133.Hollak C.E., Van Weely S., Van Oers M.H., Aerts J.M. Marked elevation of plasma chitotriosidase activity. A novel hallmark of Gaucher disease. J. Clin. Investig. 1994;93:1288–1292. doi: 10.1172/JCI117084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Boot R.G., Renkema G.H., Strijland A., Van Zonneveld A.J., Aerts J.M. Cloning of a cDNA encoding chitotriosidase, a human chitinase produced by macrophages. J. Biol. Chem. 1995;270:26252–26256. doi: 10.1074/jbc.270.44.26252. [DOI] [PubMed] [Google Scholar]
- 135.Aguilera B., Ghauharali-van der Vlugt K., Helmond M.T., Out J.M., Donker-Koopman W.E., Groener J.E., Boot R.G., Renkema G.H., Van Der Marel G.A., Van Boom J.H., et al. Transglycosidase activity of chitotriosidase: Improved enzymatic assay for the human macrophage chitinase. J. Biol. Chem. 2003;278:40911–40916. doi: 10.1074/jbc.M301804200. [DOI] [PubMed] [Google Scholar]
- 136.Schoonhoven A., Rudensky B., Elstein D., Zimran A., Hollak C.E., Groener J.E., Aerts J.M. Monitoring of Gaucher patients with a novel chitotriosidase assay. Clin. Chim. Acta. 2007;381:136–139. doi: 10.1016/j.cca.2007.02.042. [DOI] [PubMed] [Google Scholar]
- 137.Boot R.G., Renkema G.H., Verhoek M., Strijland A., Bliek J., De Meulemeester T.M., Mannens M.M., Aerts J.M. The human chitotriosidase gene. Nature of inherited enzyme deficiency. J. Biol. Chem. 1998;273:25680–25685. doi: 10.1074/jbc.273.40.25680. [DOI] [PubMed] [Google Scholar]
- 138.Deegan P.B., Moran M.T., McFarlane I., Schofield J.P., Boot R.G., Aerts J.M., Cox T.M. Clinical evaluation of chemokine and enzymatic biomarkers of Gaucher disease. Blood Cells Mol. Dis. 2005;35:259–267. doi: 10.1016/j.bcmd.2005.05.005. [DOI] [PubMed] [Google Scholar]
- 139.Boot R.G., Verhoek M., De Fost M., Hollak C.E., Maas M., Bleijlevens B., Van Breemen M.J., Van Meurs M., Boven L.A., Laman J.D., et al. Marked elevation of the chemokine CCL18/PARC in Gaucher disease: A novel surrogate marker for assessing therapeutic intervention. Blood. 2004;103:33–39. doi: 10.1182/blood-2003-05-1612. [DOI] [PubMed] [Google Scholar]
- 140.Kramer G., Wegdam W., Donker-Koopman W., Ottenhoff R., Gaspar P., Verhoek M., Nelson J., Gabriel T., Kallemeijn W., Boot R.G., et al. Elevation of glycoprotein nonmetastatic melanoma protein B in type 1 Gaucher disease patients and mouse models. FEBS Open Bio. 2016;6:902–913. doi: 10.1002/2211-5463.12078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Murugesan V., Liu J., Yang R., Lin H., Lischuk A., Pastores G., Zhang X., Chuang W.L., Mistry P.K. Validating glycoprotein non-metastatic melanoma B (gpNMB, osteoactivin), a new biomarker of Gaucher disease. Blood Cells Mol. Dis. 2018;68:47–53. doi: 10.1016/j.bcmd.2016.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Zigdon H., Savidor A., Levin Y., Meshcheriakova A., Schiffmann R., Futerman A.H. Identification of a biomarker in cerebrospinal fluid for neuronopathic forms of Gaucher disease. PLoS ONE. 2015;10:e0120194. doi: 10.1371/journal.pone.0120194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Moloney E.B., Moskites A., Ferrari E.J., Isacson O., Hallett P.J. The glycoprotein GPNMB is selectively elevated in the substantia nigra of Parkinson’s disease patients and increases after lysosomal stress. Neurobiol. Dis. 2018;120:1–11. doi: 10.1016/j.nbd.2018.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Dahl M., Doyle A., Olsson K., Mansson J.E., Marques A.R.A., Mirzaian M., Aerts J.M., Ehinger M., Rothe M., Modlich U., et al. Lentiviral gene therapy using cellular promoters cures type 1 Gaucher disease in mice. Mol. Ther. 2015;23:835–844. doi: 10.1038/mt.2015.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Keatinge M., Bui H., Menke A., Chen Y.C., Sokol A.M., Bai Q., Ellett F., Da Costa M., Burke D., Gegg M., et al. Glucocerebrosidase 1 deficient Danio rerio mirror key pathological aspects of human Gaucher disease and provide evidence of early microglial activation preceding alpha-synuclein-independent neuronal cell death. Hum. Mol. Genet. 2015;24:6640–6652. doi: 10.1093/hmg/ddv369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Aerts J.M., Hollak C.E. Plasma and metabolic abnormalities in Gaucher’s disease. Baillieres Clin. Haematol. 1997;10:691–709. doi: 10.1016/S0950-3536(97)80034-0. [DOI] [PubMed] [Google Scholar]
- 147.Aerts J.M., Kallemeijn W.W., Wegdam W., Joao Ferraz M., Van Breemen M.J., Dekker N., Kramer G., Poorthuis B.J., Groener J.E., Cox-Brinkman J., et al. Biomarkers in the diagnosis of lysosomal storage disorders: Proteins, lipids, and inhibodies. J. Inherit. Metab. Dis. 2011;34:605–619. doi: 10.1007/s10545-011-9308-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Vissers J.P., Langridge J.I., Aerts J.M. Analysis and quantification of diagnostic serum markers and protein signatures for Gaucher disease. Mol. Cell. Proteom. 2007;6:755–766. doi: 10.1074/mcp.M600303-MCP200. [DOI] [PubMed] [Google Scholar]
- 149.Hollak C.E., Levi M., Berends F., Aerts J.M., Van Oers M.H. Coagulation abnormalities in type 1 Gaucher disease are due to low-grade activation and can be partly restored by enzyme supplementation therapy. Br. J. Haematol. 1997;96:470–476. doi: 10.1046/j.1365-2141.1997.d01-2076.x. [DOI] [PubMed] [Google Scholar]
- 150.Brady R.O. Enzyme replacement therapy: Conception, chaos and culmination. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003;358:915–919. doi: 10.1098/rstb.2003.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Maas M., Van Kuijk C., Stoker J., Hollak C.E., Akkerman E.M., Aerts J.F., Den Heeten G.J. Quantification of bone involvement in Gaucher disease: MR imaging bone marrow burden score as an alternative to Dixon quantitative chemical shift MR imaging—Initial experience. Radiology. 2003;229:554–561. doi: 10.1148/radiol.2292020296. [DOI] [PubMed] [Google Scholar]
- 152.Aerts J.M., Hollak C.E., Boot R.G., Groener J.E., Maas M. Substrate reduction therapy of glycosphingolipid storage disorders. J. Inherit. Metab. Dis. 2006;29:449–456. doi: 10.1007/s10545-006-0272-5. [DOI] [PubMed] [Google Scholar]
- 153.Platt F.M., Jeyakumar M., Andersson U., Priestman D.A., Dwek R.A., Butters T.D., Cox T.M., Lachmann R.H., Hollak C., Aerts J.M., et al. Inhibition of substrate synthesis as a strategy for glycolipid lysosomal storage disease therapy. J. Inherit. Metab. Dis. 2001;24:275–290. doi: 10.1023/A:1010335505357. [DOI] [PubMed] [Google Scholar]
- 154.Heitner R., Elstein D., Aerts J., Van Weely S., Zimran A. Low-dose N-butyldeoxynojirimycin (OGT 918) for type I Gaucher disease. Blood Cells Mol. Dis. 2002;28:127–133. doi: 10.1006/bcmd.2002.0497. [DOI] [PubMed] [Google Scholar]
- 155.Mistry P.K., Balwani M., Baris H.N., Turkia H.B., Burrow T.A., Charrow J., Cox G.F., Danda S., Dragosky M., Drelichman G., et al. Safety, efficacy, and authorization of eliglustat as a first-line therapy in Gaucher disease type 1. Blood Cells Mol. Dis. 2018;71:71–74. doi: 10.1016/j.bcmd.2018.04.001. [DOI] [PubMed] [Google Scholar]
- 156.Shayman J.A., Larsen S.D. The development and use of small molecule inhibitors of glycosphingolipid metabolism for lysosomal storage diseases. J. Lipid Res. 2014;55:1215–1225. doi: 10.1194/jlr.R047167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Van Dussen L., Hendriks E.J., Groener J.E., Boot R.G., Hollak C.E., Aerts J.M. Value of plasma chitotriosidase to assess non-neuronopathic Gaucher disease severity and progression in the era of enzyme replacement therapy. J. Inherit. Metab. Dis. 2014;37:991–1001. doi: 10.1007/s10545-014-9711-x. [DOI] [PubMed] [Google Scholar]
- 158.Maegawa G.H., Tropak M.B., Buttner J.D., Rigat B.A., Fuller M., Pandit D., Tang L., Kornhaber G.J., Hamuro Y., Clarke J.T., et al. Identification and characterization of ambroxol as an enzyme enhancement agent for Gaucher disease. J. Biol. Chem. 2009;284:23502–23516. doi: 10.1074/jbc.M109.012393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zimran A., Altarescu G., Elstein D. Pilot study using ambroxol as a pharmacological chaperone in type 1 Gaucher disease. Blood Cells Mol. Dis. 2013;50:134–137. doi: 10.1016/j.bcmd.2012.09.006. [DOI] [PubMed] [Google Scholar]
- 160.Narita A., Shirai K., Itamura S., Matsuda A., Ishihara A., Matsushita K., Fukuda C., Kubota N., Takayama R., Shigematsu H., et al. Ambroxol chaperone therapy for neuronopathic Gaucher disease: A pilot study. Ann. Clin. Transl. Neurol. 2016;3:200–215. doi: 10.1002/acn3.292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Fog C.K., Zago P., Malini E., Solanko L.M., Peruzzo P., Bornaes C., Magnoni R., Mehmedbasic A., Petersen N.H.T., Bembi B., et al. The heat shock protein amplifier arimoclomol improves refolding, maturation and lysosomal activity of glucocerebrosidase. EBioMedicine. 2018;38:142–153. doi: 10.1016/j.ebiom.2018.11.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Aerts J.M., Ferraz M.J., Mirzaian M., Gaspar P., Oussoren S.V., Wisse P., Kuo C.L., Lelieveld L.T., Kytidou K., Hazeu M.D., et al. For Better or Worse: Adapting to Defective Lysosomal Glycosphingolipid. John Wiley & Sons, Ltd.; Chichester, UK: 2017. Lysosomal storage diseases; pp. 1–13. [Google Scholar]
- 163.Ferraz M.J., Marques A.R., Appelman M.D., Verhoek M., Strijland A., Mirzaian M., Scheij S., Ouairy C.M., Lahav D., Wisse P., et al. Lysosomal glycosphingolipid catabolism by acid ceramidase: Formation of glycosphingoid bases during deficiency of glycosidases. FEBS Lett. 2016;590:716–725. doi: 10.1002/1873-3468.12104. [DOI] [PubMed] [Google Scholar]
- 164.Raghavan S.S., Mumford R.A., Kanfer J.N. Isolation and characterization of glucosylsphingosine from Gaucher’s spleen. J. Lipid Res. 1974;15:484–490. [PubMed] [Google Scholar]
- 165.Nilsson O., Svennerholm L. Accumulation of glucosylceramide and glucosylsphingosine (psychosine) in cerebrum and cerebellum in infantile and juvenile Gaucher disease. J. Neurochem. 1982;39:709–718. doi: 10.1111/j.1471-4159.1982.tb07950.x. [DOI] [PubMed] [Google Scholar]
- 166.Dekker N., Van Dussen L., Hollak C.E., Overkleeft H., Scheij S., Ghauharali K., Van Breemen M.J., Ferraz M.J., Groener J.E., Maas M., et al. Elevated plasma glucosylsphingosine in Gaucher disease: Relation to phenotype, storage cell markers, and therapeutic response. Blood. 2011;118:e118–e127. doi: 10.1182/blood-2011-05-352971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Mirzaian M., Wisse P., Ferraz M.J., Gold H., Donker-Koopman W.E., Verhoek M., Overkleeft H.S., Boot R.G., Kramer G., Dekker N., et al. Mass spectrometric quantification of glucosylsphingosine in plasma and urine of type 1 Gaucher patients using an isotope standard. Blood Cells Mol. Dis. 2015;54:307–314. doi: 10.1016/j.bcmd.2015.01.006. [DOI] [PubMed] [Google Scholar]
- 168.Lelieveld L.T., Mirzaian M., Kuo C.L., Artola M., Ferraz M.J., Peter R.E.A., Akiyama H., Greimel P., Van Den Berg R., Overkleeft H.S., et al. Role of beta-glucosidase 2 in aberrant glycosphingolipid metabolism: Model of glucocerebrosidase deficiency in zebrafish. J. Lipid Res. 2019;60:1851–1867. doi: 10.1194/jlr.RA119000154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Groener J.E., Poorthuis B.J., Kuiper S., Helmond M.T., Hollak C.E., Aerts J.M. HPLC for simultaneous quantification of total ceramide, glucosylceramide, and ceramide trihexoside concentrations in plasma. Clin. Chem. 2007;53:742–747. doi: 10.1373/clinchem.2006.079012. [DOI] [PubMed] [Google Scholar]
- 170.Mistry P.K., Liu J., Sun L., Chuang W.L., Yuen T., Yang R., Lu P., Zhang K., Li J., Keutzer J., et al. Glucocerebrosidase 2 gene deletion rescues type 1 Gaucher disease. Proc. Natl. Acad. Sci. USA. 2014;111:4934–4939. doi: 10.1073/pnas.1400768111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Taguchi Y.V., Liu J., Ruan J., Pacheco J., Zhang X., Abbasi J., Keutzer J., Mistry P.K., Chandra S.S. Glucosylsphingosine promotes alpha-synuclein pathology in mutant GBA-associated Parkinson’s disease. J. Neurosci. 2017;37:9617–9631. doi: 10.1523/JNEUROSCI.1525-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Nair S., Branagan A.R., Liu J., Boddupalli C.S., Mistry P.K., Dhodapkar M.V. Clonal immunoglobulin against lysolipids in the origin of myeloma. N. Engl. J. Med. 2016;374:555–561. doi: 10.1056/NEJMoa1508808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Pandey M.K., Grabowski G.A., Kohl J. An unexpected player in Gaucher disease: The multiple roles of complement in disease development. Semin. Immunol. 2018;37:30–42. doi: 10.1016/j.smim.2018.02.006. [DOI] [PubMed] [Google Scholar]
- 174.Smith N.J., Fuller M., Saville J.T., Cox T.M. Reduced cerebral vascularization in experimental neuronopathic Gaucher disease. J. Pathol. 2018;244:120–128. doi: 10.1002/path.4992. [DOI] [PubMed] [Google Scholar]
- 175.Ferraz M.J., Marques A.R., Gaspar P., Mirzaian M., Van Roomen C., Ottenhoff R., Alfonso P., Irun P., Giraldo P., Wisse P., et al. Lyso-glycosphingolipid abnormalities in different murine models of lysosomal storage disorders. Mol. Genet. Metab. 2016;117:186–193. doi: 10.1016/j.ymgme.2015.12.006. [DOI] [PubMed] [Google Scholar]
- 176.Kuchar L., Sikora J., Gulinello M.E., Poupetova H., Lugowska A., Malinova V., Jahnova H., Asfaw B., Ledvinova J. Quantitation of plasmatic lysosphingomyelin and lysosphingomyelin-509 for differential screening of Niemann-Pick A/B and C diseases. Anal. Biochem. 2017;525:73–77. doi: 10.1016/j.ab.2017.02.019. [DOI] [PubMed] [Google Scholar]
- 177.Suzuki K. Twenty five years of the: “psychosine hypothesis”: A personal perspective of its history and present status. Neurochem. Res. 1998;23:251–259. doi: 10.1023/A:1022436928925. [DOI] [PubMed] [Google Scholar]
- 178.Aerts J.M., Groener J.E., Kuiper S., Donker-Koopman W.E., Strijland A., Ottenhoff R., Van Roomen C., Mirzaian M., Wijburg F.A., Linthorst G.E., et al. Elevated globotriaosylsphingosine is a hallmark of Fabry disease. Proc. Natl. Acad. Sci. USA. 2008;105:2812–2817. doi: 10.1073/pnas.0712309105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Choi L., Vernon J., Kopach O., Minett M.S., Mills K., Clayton P.T., Meert T., Wood J.N. The Fabry disease-associated lipid Lyso-Gb3 enhances voltage-gated calcium currents in sensory neurons and causes pain. Neurosci. Lett. 2015;594:163–168. doi: 10.1016/j.neulet.2015.01.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Sanchez-Nino M.D., Carpio D., Sanz A.B., Ruiz-Ortega M., Mezzano S., Ortiz A. Lyso-Gb3 activates Notch1 in human podocytes. Hum. Mol. Genet. 2015;24:5720–5732. doi: 10.1093/hmg/ddv291. [DOI] [PubMed] [Google Scholar]
- 181.Kaissarian N., Kang J., Shu L., Ferraz M.J., Aerts J.M., Shayman J.A. Dissociation of globotriaosylceramide and impaired endothelial function in alpha-galactosidase-A deficient EA.hy926 cells. Mol. Genet. Metab. 2018;125:338–344. doi: 10.1016/j.ymgme.2018.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Rombach S.M., Twickler T.B., Aerts J.M., Linthorst G.E., Wijburg F.A., Hollak C.E. Vasculopathy in patients with Fabry disease: Current controversies and research directions. Mol. Genet. Metab. 2010;99:99–108. doi: 10.1016/j.ymgme.2009.10.004. [DOI] [PubMed] [Google Scholar]
- 183.Ghauharali-van der Vlugt K., Langeveld M., Poppema A., Kuiper S., Hollak C.E., Aerts J.M., Groener J.E. Prominent increase in plasma ganglioside GM3 is associated with clinical manifestations of type I Gaucher disease. Clin. Chim. Acta. 2008;389:109–113. doi: 10.1016/j.cca.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 184.Langeveld M., Ghauharali K.J., Sauerwein H.P., Ackermans M.T., Groener J.E., Hollak C.E., Aerts J.M., Serlie M.J. Type I Gaucher disease, a glycosphingolipid storage disorder, is associated with insulin resistance. J. Clin. Endocrinol. Metab. 2008;93:845–851. doi: 10.1210/jc.2007-1702. [DOI] [PubMed] [Google Scholar]
- 185.Yildiz Y., Matern H., Thompson B., Allegood J.C., Warren R.L., Ramirez D.M., Hammer R.E., Hamra F.K., Matern S., Russell D.W. Mutation of beta-glucosidase 2 causes glycolipid storage disease and impaired male fertility. J. Clin. Investig. 2006;116:2985–2994. doi: 10.1172/JCI29224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Boot R.G., Verhoek M., Donker-Koopman W., Strijland A., Van Marle J., Overkleeft H.S., Wennekes T., Aerts J.M. Identification of the non-lysosomal glucosylceramidase as beta-glucosidase 2. J. Biol. Chem. 2007;282:1305–1312. doi: 10.1074/jbc.M610544200. [DOI] [PubMed] [Google Scholar]
- 187.Hammer M.B., Eleuch-Fayache G., Schottlaender L.V., Nehdi H., Gibbs J.R., Arepalli S.K., Chong S.B., Hernandez D.G., Sailer A., Liu G., et al. Mutations in GBA2 cause autosomal-recessive cerebellar ataxia with spasticity. Am. J. Hum. Genet. 2013;92:245–251. doi: 10.1016/j.ajhg.2012.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Martin E., Schule R., Smets K., Rastetter A., Boukhris A., Loureiro J.L., Gonzalez M.A., Mundwiller E., Deconinck T., Wessner M., et al. Loss of function of glucocerebrosidase GBA2 is responsible for motor neuron defects in hereditary spastic paraplegia. Am. J. Hum. Genet. 2013;92:238–244. doi: 10.1016/j.ajhg.2012.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Kancheva D., Atkinson D., De Rijk P., Zimon M., Chamova T., Mitev V., Yaramis A., Maria Fabrizi G., Topaloglu H., Tournev I., et al. Novel mutations in genes causing hereditary spastic paraplegia and Charcot-Marie-Tooth neuropathy identified by an optimized protocol for homozygosity mapping based on whole-exome sequencing. Genet. Med. 2016;18:600–607. doi: 10.1038/gim.2015.139. [DOI] [PubMed] [Google Scholar]
- 190.Sultana S., Reichbauer J., Schule R., Mochel F., Synofzik M., Van Der Spoel A.C. Lack of enzyme activity in GBA2 mutants associated with hereditary spastic paraplegia/cerebellar ataxia (SPG46) Biochem. Biophys. Res. Commun. 2015;465:35–40. doi: 10.1016/j.bbrc.2015.07.112. [DOI] [PubMed] [Google Scholar]
- 191.Woeste M.A., Stern S., Raju D.N., Grahn E., Dittmann D., Gutbrod K., Dormann P., Hansen J.N., Schonauer S., Marx C.E., et al. Species-specific differences in nonlysosomal glucosylceramidase GBA2 function underlie locomotor dysfunction arising from loss-of-function mutations. J. Biol. Chem. 2019;294:3853–3871. doi: 10.1074/jbc.RA118.006311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Saftig P., Klumperman J. Lysosome biogenesis and lysosomal membrane proteins: Trafficking meets function. Nat. Rev. Mol. Cell Biol. 2009;10:623–635. doi: 10.1038/nrm2745. [DOI] [PubMed] [Google Scholar]
- 193.Luzio J.P., Hackmann Y., Dieckmann N.M., Griffiths G.M. The biogenesis of lysosomes and lysosome-related organelles. Cold Spring Harb. Perspect. Biol. 2014;6:a016840. doi: 10.1101/cshperspect.a016840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Bowman S.L., Bi-Karchin J., Le L., Marks M.S. The road to lysosome-related organelles: Insights from Hermansky-Pudlak syndrome and other rare diseases. Traffic. 2019;20:404–435. doi: 10.1111/tra.12646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Delevoye C., Marks M.S., Raposo G. Lysosome-related organelles as functional adaptations of the endolysosomal system. Curr. Opin. Cell Biol. 2019;59:147–158. doi: 10.1016/j.ceb.2019.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Bahadori R., Rinner O., Schonthaler H.B., Biehlmaier O., Makhankov Y.V., Rao P., Jagadeeswaran P., Neuhauss S.C. The Zebrafish fade out mutant: A novel genetic model for Hermansky-Pudlak syndrome. Investig. Ophthalmol. Vis. Sci. 2006;47:4523–4531. doi: 10.1167/iovs.05-1596. [DOI] [PubMed] [Google Scholar]
- 197.Ellis K., Bagwell J., Bagnat M. Notochord vacuoles are lysosome-related organelles that function in axis and spine morphogenesis. J. Cell Biol. 2013;200:667–679. doi: 10.1083/jcb.201212095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Diaz-Tellez A., Zampedri C., Ramos-Balderas J.L., Garcia-Hernandez F., Maldonado E. Zebrafish scarb2a insertional mutant reveals a novel function for the Scarb2/Limp2 receptor in notochord development. Dev. Dyn. 2016;245:508–519. doi: 10.1002/dvdy.24383. [DOI] [PubMed] [Google Scholar]
- 199.Groux-Degroote S., Van Dijk S.M., Wolthoorn J., Neumann S., Theos A.C., De Maziere A.M., Klumperman J., Van Meer G., Sprong H. Glycolipid-dependent sorting of melanosomal from lysosomal membrane proteins by lumenal determinants. Traffic. 2008;9:951–963. doi: 10.1111/j.1600-0854.2008.00740.x. [DOI] [PubMed] [Google Scholar]
- 200.McLean W.H., Hull P.R. Breach delivery: Increased solute uptake points to a defective skin barrier in atopic dermatitis. J. Investig. Dermatol. 2007;127:8–10. doi: 10.1038/sj.jid.5700609. [DOI] [PubMed] [Google Scholar]
- 201.Yousef H., Alhajj M., Sharma S. Anatomy, Skin (Integument), Epidermis. StatPearls; Treasure Island, FL, USA: 2019. [PubMed] [Google Scholar]
- 202.Eckhart L., Lippens S., Tschachler E., Declercq W. Cell death by cornification. Biochim. Biophys. Acta. 2013;1833:3471–3480. doi: 10.1016/j.bbamcr.2013.06.010. [DOI] [PubMed] [Google Scholar]
- 203.Rawlings A.V., Harding C.R. Moisturization and skin barrier function. Dermatol. Ther. 2004;17:43–48. doi: 10.1111/j.1396-0296.2004.04S1005.x. [DOI] [PubMed] [Google Scholar]
- 204.Takahashi M., Tezuka T. The content of free amino acids in the stratum corneum is increased in senile xerosis. Arch. Dermatol. Res. 2004;295:448–452. doi: 10.1007/s00403-003-0448-x. [DOI] [PubMed] [Google Scholar]
- 205.Voegeli D. The role of emollients in the care of patients with dry skin. Nurs. Stand. 2007;22:64–68. doi: 10.7748/ns2007.10.22.7.62.c4626. [DOI] [PubMed] [Google Scholar]
- 206.Fluhr J.W., Elias P.M. Stratum corneum pH: Formation and function of the “acid mantle”. Exog. Dermatol. 2002;1:163–175. doi: 10.1159/000066140. [DOI] [Google Scholar]
- 207.Proksch E. pH in nature, humans and skin. J. Dermatol. 2018;45:1044–1052. doi: 10.1111/1346-8138.14489. [DOI] [PubMed] [Google Scholar]
- 208.Elias P.M. Epidermal lipids, membranes, and keratinization. Int. J. Dermatol. 1981;20 doi: 10.1111/j.1365-4362.1981.tb05278.x. [DOI] [PubMed] [Google Scholar]
- 209.Rinnerthaler M., Duschl J., Steinbacher P., Salzmann M., Bischof J., Schuller M., Wimmer H., Peer T., Bauer J.W., Richter K. Age-related changes in the composition of the cornified envelope in human skin. Exp. Dermatol. 2013;22:329–335. doi: 10.1111/exd.12135. [DOI] [PubMed] [Google Scholar]
- 210.Meguro S., Arai Y., Masukawa Y., Uie K., Tokimitsu I. Relationship between covalently bound ceramides and transepidermal water loss (TEWL) Arch. Dermatol. Res. 2000;292:463–468. doi: 10.1007/s004030000160. [DOI] [PubMed] [Google Scholar]
- 211.Elias P.M., Gruber R., Crumrine D., Menon G., Williams M.L., Wakefield J.S., Holleran W.M., Uchida Y. Formation and functions of the corneocyte lipid envelope (CLE) Biochim. Biophys. Acta. 2014;1841:314–318. doi: 10.1016/j.bbalip.2013.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Rabionet M., Gorgas K., Sandhoff R. Ceramide synthesis in the epidermis. Biochim. Biophys. Acta. 2014;1841:422–434. doi: 10.1016/j.bbalip.2013.08.011. [DOI] [PubMed] [Google Scholar]
- 213.Lampe M.A., Burlingame A.L., Whitney J., Williams M.L., Brown B.E., Roitman E., Elias P.M. Human stratum corneum lipids: Characterization and regional variations. J. Lipid Res. 1983;24:120–130. [PubMed] [Google Scholar]
- 214.Janssens M., Van Smeden J., Gooris G.S., Bras W., Portale G., Caspers P.J., Vreeken R.J., Kezic S., Lavrijsen A.P., Bouwstra J.A. Lamellar lipid organization and ceramide composition in the stratum corneum of patients with atopic eczema. J. Investig. Dermatol. 2011;131:2136–2138. doi: 10.1038/jid.2011.175. [DOI] [PubMed] [Google Scholar]
- 215.Imokawa G., Abe A., Jin K., Higaki Y., Kawashima M., Hidano A. Decreased level of ceramides in stratum corneum of atopic dermatitis: An etiologic factor in atopic dry skin? J. Investig. Dermatol. 1991;96:523–526. doi: 10.1111/1523-1747.ep12470233. [DOI] [PubMed] [Google Scholar]
- 216.Motta S., Monti M., Sesana S., Caputo R., Carelli S., Ghidoni R. Ceramide composition of the psoriatic scale. Biochim. Biophys. Acta. 1993;1182:147–151. doi: 10.1016/0925-4439(93)90135-N. [DOI] [PubMed] [Google Scholar]
- 217.Coderch L., Lopez O., De la Maza A., Parra J.L. Ceramides and skin function. Am. J. Clin. Dermatol. 2003;4:107–129. doi: 10.2165/00128071-200304020-00004. [DOI] [PubMed] [Google Scholar]
- 218.Elias P.M., Williams M.L., Crumrine D., Schmuth M. Inherited clinical disorders of lipid metabolism. Curr. Probl. Dermatol. 2010;39:30–88. doi: 10.1159/000321084. [DOI] [PubMed] [Google Scholar]
- 219.Wertz P. Epidermal lamellar granules. Skin. Pharmacol. Physiol. 2018;31:262–268. doi: 10.1159/000491757. [DOI] [PubMed] [Google Scholar]
- 220.Mitsutake S., Suzuki C., Akiyama M., Tsuji K., Yanagi T., Shimizu H., Igarashi Y. ABCA12 dysfunction causes a disorder in glucosylceramide accumulation during keratinocyte differentiation. J. Dermatol. Sci. 2010;60:128–129. doi: 10.1016/j.jdermsci.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 221.Akiyama M. The roles of ABCA12 in epidermal lipid barrier formation and keratinocyte differentiation. Biochim. Biophys. Acta. 2014;1841:435–440. doi: 10.1016/j.bbalip.2013.08.009. [DOI] [PubMed] [Google Scholar]
- 222.Akiyama M., Sugiyama-Nakagiri Y., Sakai K., McMillan J.R., Goto M., Arita K., Tsuji-Abe Y., Tabata N., Matsuoka K., Sasaki R., et al. Mutations in lipid transporter ABCA12 in harlequin ichthyosis and functional recovery by corrective gene transfer. J. Clin. Investig. 2005;115:1777–1784. doi: 10.1172/JCI24834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Schurer N.Y., Elias P.M. The biochemistry and function of stratum corneum lipids. Adv. Lipid Res. 1991;24:27–56. doi: 10.1016/b978-0-12-024924-4.50006-7. [DOI] [PubMed] [Google Scholar]
- 224.Feingold K.R. Lamellar bodies: The key to cutaneous barrier function. J. Investig. Dermatol. 2012;132:1951–1953. doi: 10.1038/jid.2012.177. [DOI] [PubMed] [Google Scholar]
- 225.Geeraert L., Mannaerts G.P., Van Veldhoven P.P. Conversion of dihydroceramide into ceramide: Involvement of a desaturase. Biochem. J. 1997;327:125–132. doi: 10.1042/bj3270125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Michel C., Van Echten-Deckert G., Rother J., Sandhoff K., Wang E., Merrill A.H., Jr. Characterization of ceramide synthesis. A dihydroceramide desaturase introduces the 4,5-trans-double bond of sphingosine at the level of dihydroceramide. J. Biol. Chem. 1997;272:22432–22437. doi: 10.1074/jbc.272.36.22432. [DOI] [PubMed] [Google Scholar]
- 227.Savile C.K., Fabrias G., Buist P.H. Dihydroceramide delta (4) desaturase initiates substrate oxidation at C-4. J. Am. Chem. Soc. 2001;123:4382–4385. doi: 10.1021/ja010088w. [DOI] [PubMed] [Google Scholar]
- 228.Mizutani Y., Kihara A., Igarashi Y. Identification of the human sphingolipid C4-hydroxylase, hDES2, and its up-regulation during keratinocyte differentiation. FEBS Lett. 2004;563:93–97. doi: 10.1016/S0014-5793(04)00274-1. [DOI] [PubMed] [Google Scholar]
- 229.Breiden B., Sandhoff K. The role of sphingolipid metabolism in cutaneous permeability barrier formation. Biochim. Biophys. Acta. 2014;1841:441–452. doi: 10.1016/j.bbalip.2013.08.010. [DOI] [PubMed] [Google Scholar]
- 230.Park Y.H., Jang W.H., Seo J.A., Park M., Lee T.R., Park Y.H., Kim D.K., Lim K.M. Decrease of ceramides with very long-chain fatty acids and downregulation of elongases in a murine atopic dermatitis model. J. Investig. Dermatol. 2012;132:476–479. doi: 10.1038/jid.2011.333. [DOI] [PubMed] [Google Scholar]
- 231.Uchida Y. The role of fatty acid elongation in epidermal structure and function. Dermatoendocrinology. 2011;3:65–69. doi: 10.4161/derm.3.2.14662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Hanley K., Ng D.C., He S.S., Lau P., Min K., Elias P.M., Bikle D.D., Mangelsdorf D.J., Williams M.L., Feingold K.R. Oxysterols induce differentiation in human keratinocytes and increase Ap-1-dependent involucrin transcription. J. Investig. Dermatol. 2000;114:545–553. doi: 10.1046/j.1523-1747.2000.00895.x. [DOI] [PubMed] [Google Scholar]
- 233.Denning M.F., Kazanietz M.G., Blumberg P.M., Yuspa S.H. Cholesterol sulfate activates multiple protein kinase C isoenzymes and induces granular cell differentiation in cultured murine keratinocytes. Cell Growth Differ. 1995;6:1619–1626. [PubMed] [Google Scholar]
- 234.Hanley K., Wood L., Ng D.C., He S.S., Lau P., Moser A., Elias P.M., Bikle D.D., Williams M.L., Feingold K.R. Cholesterol sulfate stimulates involucrin transcription in keratinocytes by increasing Fra-1, Fra-2, and Jun D. J. Lipid Res. 2001;42:390–398. [PubMed] [Google Scholar]
- 235.Elias P.M., Williams M.L., Holleran W.M., Jiang Y.J., Schmuth M. Pathogenesis of permeability barrier abnormalities in the ichthyoses: Inherited disorders of lipid metabolism. J. Lipid Res. 2008;49:697–714. doi: 10.1194/jlr.R800002-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Feingold K.R., Jiang Y.J. The mechanisms by which lipids coordinately regulate the formation of the protein and lipid domains of the stratum corneum: Role of fatty acids, oxysterols, cholesterol sulfate and ceramides as signaling molecules. Dermato Endocrinol. 2011;3:113–118. doi: 10.4161/derm.3.2.14996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Elias P.M., Crumrine D., Rassner U., Hachem J.P., Menon G.K., Man W., Choy M.H., Leypoldt L., Feingold K.R., Williams M.L. Basis for abnormal desquamation and permeability barrier dysfunction in RXLI. J. Investig. Dermatol. 2004;122:314–319. doi: 10.1046/j.1523-1747.2003.22258.x. [DOI] [PubMed] [Google Scholar]
- 238.Sato J., Denda M., Nakanishi J., Nomura J., Koyama J. Cholesterol sulfate inhibits proteases that are involved in desquamation of stratum corneum. J. Investig. Dermatol. 1998;111:189–193. doi: 10.1046/j.1523-1747.1998.00244.x. [DOI] [PubMed] [Google Scholar]
- 239.Cox P., Squier C.A. Variations in lipids in different layers of porcine epidermis. J. Investig. Dermatol. 1986;87:741–744. doi: 10.1111/1523-1747.ep12456872. [DOI] [PubMed] [Google Scholar]
- 240.Ranasinghe A.W., Wertz P.W., Downing D.T., Mackenzie I.C. Lipid composition of cohesive and desquamated corneocytes from mouse ear skin. J. Investig. Dermatol. 1986;86:187–190. doi: 10.1111/1523-1747.ep12284246. [DOI] [PubMed] [Google Scholar]
- 241.Akiyama M. Corneocyte lipid envelope (CLE), the key structure for skin barrier function and ichthyosis pathogenesis. J. Dermatol. Sci. 2017;88:3–9. doi: 10.1016/j.jdermsci.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 242.Feingold K.R. Thematic review series: Skin lipids. The role of epidermal lipids in cutaneous permeability barrier homeostasis. J. Lipid Res. 2007;48:2531–2546. doi: 10.1194/jlr.R700013-JLR200. [DOI] [PubMed] [Google Scholar]
- 243.Greene S.L., Muller S.A. Netherton’s syndrome. Report of a case and review of the literature. J. Am. Acad. Dermatol. 1985;13:329–337. doi: 10.1016/S0190-9622(85)70170-3. [DOI] [PubMed] [Google Scholar]
- 244.Traupe H. The Ichthyoses: A Guide to Clinical Diagnosis, Genetic Counseling, and Therapy. Springer; Berlin, Germany: New York, NY, USA: 1989. [Google Scholar]
- 245.Stone D.L., Carey W.F., Christodoulou J., Sillence D., Nelson P., Callahan M., Tayebi N., Sidransky E. Type 2 Gaucher disease: The collodion baby phenotype revisited. Arch. Dis. Child. Fetal Neonatal Ed. 2000;82:F163–F166. doi: 10.1136/fn.82.2.F163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Holleran W.M., Takagi Y., Menon G.K., Legler G., Feingold K.R., Elias P.M. Processing of epidermal glucosylceramides is required for optimal mammalian cutaneous permeability barrier function. J. Clin. Investig. 1993;91:1656–1664. doi: 10.1172/JCI116374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Holleran W.M., Takagi Y., Menon G.K., Jackson S.M., Lee J.M., Feingold K.R., Elias P.M. Permeability barrier requirements regulate epidermal beta-glucocerebrosidase. J. Lipid Res. 1994;35:905–912. [PubMed] [Google Scholar]
- 248.Holleran W.M., Ginns E.I., Menon G.K., Grundmann J.U., Fartasch M., McKinney C.E., Elias P.M., Sidransky E. Consequences of beta-glucocerebrosidase deficiency in epidermis. Ultrastructure and permeability barrier alterations in Gaucher disease. J. Clin. Investig. 1994;93:1756–1764. doi: 10.1172/JCI117160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Doering T., Holleran W.M., Potratz A., Vielhaber G., Elias P.M., Suzuki K., Sandhoff K. Sphingolipid activator proteins are required for epidermal permeability barrier formation. J. Biol. Chem. 1999;274:11038–11045. doi: 10.1074/jbc.274.16.11038. [DOI] [PubMed] [Google Scholar]
- 250.Freinkel R.K., Traczyk T.N. Lipid composition and acid hydrolase content of lamellar granules of fetal rat epidermis. J. Investig. Dermatol. 1985;85:295–298. doi: 10.1111/1523-1747.ep12276831. [DOI] [PubMed] [Google Scholar]
- 251.Grayson S., Johnson-Winegar A.G., Wintroub B.U., Isseroff R.R., Epstein E.H., Jr., Elias P.M. Lamellar body-enriched fractions from neonatal mice: Preparative techniques and partial characterization. J. Investig. Dermatol. 1985;85:289–294. doi: 10.1111/1523-1747.ep12276826. [DOI] [PubMed] [Google Scholar]
- 252.Madison K.C., Sando G.N., Howard E.J., True C.A., Gilbert D., Swartzendruber D.C., Wertz P.W. Lamellar granule biogenesis: A role for ceramide glucosyltransferase, lysosomal enzyme transport, and the Golgi. J. Investig. Dermatol. Symp. Proc. 1998;3:80–86. doi: 10.1038/jidsymp.1998.19. [DOI] [PubMed] [Google Scholar]
- 253.Takagi Y., Kriehuber E., Imokawa G., Elias P.M., Holleran W.M. Beta-glucocerebrosidase activity in mammalian stratum corneum. J. Lipid Res. 1999;40:861–869. [PubMed] [Google Scholar]
- 254.Schmuth M., Schoonjans K., Yu Q.C., Fluhr J.W., Crumrine D., Hachem J.P., Lau P., Auwerx J., Elias P.M., Feingold K.R. Role of peroxisome proliferator-activated receptor alpha in epidermal development in utero. J. Investig. Dermatol. 2002;119:1298–1303. doi: 10.1046/j.1523-1747.2002.19605.x. [DOI] [PubMed] [Google Scholar]
- 255.Hachem J.P., Crumrine D., Fluhr J., Brown B.E., Feingold K.R., Elias P.M. pH directly regulates epidermal permeability barrier homeostasis, and stratum corneum integrity/cohesion. J. Investig. Dermatol. 2003;121:345–353. doi: 10.1046/j.1523-1747.2003.12365.x. [DOI] [PubMed] [Google Scholar]
- 256.Van Smeden J., Dijkhoff I.M., Helder R.W.J., Al-Khakany H., Boer D.E.C., Schreuder A., Kallemeijn W.W., Absalah S., Overkleeft H.S., Aerts J., et al. In situ visualization of glucocerebrosidase in human skin tissue: Zymography versus activity-based probe labeling. J. Lipid Res. 2017;58:2299–2309. doi: 10.1194/jlr.M079376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Schmuth M., Man M.Q., Weber F., Gao W., Feingold K.R., Fritsch P., Elias P.M., Holleran W.M. Permeability barrier disorder in Niemann-Pick disease: Sphingomyelin-ceramide processing required for normal barrier homeostasis. J. Investig. Dermatol. 2000;115:459–466. doi: 10.1046/j.1523-1747.2000.00081.x. [DOI] [PubMed] [Google Scholar]
- 258.Jensen J.M., Schutze S., Forl M., Kronke M., Proksch E. Roles for tumor necrosis factor receptor p55 and sphingomyelinase in repairing the cutaneous permeability barrier. J. Clin. Investig. 1999;104:1761–1770. doi: 10.1172/JCI5307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Oyoshi M.K., He R., Kumar L., Yoon J., Geha R.S. Cellular and molecular mechanisms in atopic dermatitis. Adv. Immunol. 2009;102:135–226. doi: 10.1016/S0065-2776(09)01203-6. [DOI] [PubMed] [Google Scholar]
- 260.Wollenberg A., Rawer H.C., Schauber J. Innate immunity in atopic dermatitis. Clin. Rev. Allergy Immunol. 2011;41:272–281. doi: 10.1007/s12016-010-8227-x. [DOI] [PubMed] [Google Scholar]
- 261.David Boothe W., Tarbox J.A., Tarbox M.B. Atopic dermatitis: Pathophysiology. Adv. Exp. Med. Biol. 2017;1027:21–37. doi: 10.1007/978-3-319-64804-0_3. [DOI] [PubMed] [Google Scholar]
- 262.Leung D.Y., Bieber T. Atopic dermatitis. Lancet. 2003;361:151–160. doi: 10.1016/S0140-6736(03)12193-9. [DOI] [PubMed] [Google Scholar]
- 263.Seguchi T., Cui C.Y., Kusuda S., Takahashi M., Aisu K., Tezuka T. Decreased expression of filaggrin in atopic skin. Arch. Dermatol. Res. 1996;288:442–446. doi: 10.1007/BF02505232. [DOI] [PubMed] [Google Scholar]
- 264.Palmer C.N., Irvine A.D., Terron-Kwiatkowski A., Zhao Y., Liao H., Lee S.P., Goudie D.R., Sandilands A., Campbell L.E., Smith F.J., et al. Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis. Nat. Genet. 2006;38:441–446. doi: 10.1038/ng1767. [DOI] [PubMed] [Google Scholar]
- 265.Cole C., Kroboth K., Schurch N.J., Sandilands A., Sherstnev A., O’Regan G.M., Watson R.M., McLean W.H., Barton G.J., Irvine A.D., et al. Filaggrin-stratified transcriptomic analysis of pediatric skin identifies mechanistic pathways in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2014;134:82–91. doi: 10.1016/j.jaci.2014.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Jungersted J.M., Scheer H., Mempel M., Baurecht H., Cifuentes L., Hogh J.K., Hellgren L.I., Jemec G.B., Agner T., Weidinger S. Stratum corneum lipids, skin barrier function and filaggrin mutations in patients with atopic eczema. Allergy. 2010;65:911–918. doi: 10.1111/j.1398-9995.2010.02326.x. [DOI] [PubMed] [Google Scholar]
- 267.Farwanah H., Raith K., Neubert R.H., Wohlrab J. Ceramide profiles of the uninvolved skin in atopic dermatitis and psoriasis are comparable to those of healthy skin. Arch. Dermatol. Res. 2005;296:514–521. doi: 10.1007/s00403-005-0551-2. [DOI] [PubMed] [Google Scholar]
- 268.Ishikawa J., Narita H., Kondo N., Hotta M., Takagi Y., Masukawa Y., Kitahara T., Takema Y., Koyano S., Yamazaki S., et al. Changes in the ceramide profile of atopic dermatitis patients. J. Investig. Dermatol. 2010;130:2511–2514. doi: 10.1038/jid.2010.161. [DOI] [PubMed] [Google Scholar]
- 269.Yamamoto A., Serizawa S., Ito M., Sato Y. Stratum corneum lipid abnormalities in atopic dermatitis. Arch. Dermatol. Res. 1991;283:219–223. doi: 10.1007/BF01106105. [DOI] [PubMed] [Google Scholar]
- 270.Bleck O., Abeck D., Ring J., Hoppe U., Vietzke J.P., Wolber R., Brandt O., Schreiner V. Two ceramide subfractions detectable in Cer (AS) position by HPTLC in skin surface lipids of non-lesional skin of atopic eczema. J. Investig. Dermatol. 1999;113:894–900. doi: 10.1046/j.1523-1747.1999.00809.x. [DOI] [PubMed] [Google Scholar]
- 271.Di Nardo A., Wertz P., Giannetti A., Seidenari S. Ceramide and cholesterol composition of the skin of patients with atopic dermatitis. Acta Derm. Venereol. 1998;78:27–30. doi: 10.1080/00015559850135788. [DOI] [PubMed] [Google Scholar]
- 272.Janssens M., Van Smeden J., Gooris G.S., Bras W., Portale G., Caspers P.J., Vreeken R.J., Hankemeier T., Kezic S., Wolterbeek R., et al. Increase in short-chain ceramides correlates with an altered lipid organization and decreased barrier function in atopic eczema patients. J. Lipid Res. 2012;53:2755–2766. doi: 10.1194/jlr.P030338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Van Smeden J., Janssens M., Kaye E.C., Caspers P.J., Lavrijsen A.P., Vreeken R.J., Bouwstra J.A. The importance of free fatty acid chain length for the skin barrier function in atopic eczema patients. Exp. Dermatol. 2014;23:45–52. doi: 10.1111/exd.12293. [DOI] [PubMed] [Google Scholar]
- 274.Macheleidt O., Kaiser H.W., Sandhoff K. Deficiency of epidermal protein-bound omega-hydroxyceramides in atopic dermatitis. J. Investig. Dermatol. 2002;119:166–173. doi: 10.1046/j.1523-1747.2002.01833.x. [DOI] [PubMed] [Google Scholar]
- 275.Ohno Y., Suto S., Yamanaka M., Mizutani Y., Mitsutake S., Igarashi Y., Sassa T., Kihara A. ELOVL1 production of C24 acyl-CoAs is linked to C24 sphingolipid synthesis. Proc. Natl. Acad. Sci. USA. 2010;107:18439–18444. doi: 10.1073/pnas.1005572107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Danso M., Boiten W., Van Drongelen V., Gmelig Meijling K., Gooris G., El Ghalbzouri A., Absalah S., Vreeken R., Kezic S., Van Smeden J., et al. Altered expression of epidermal lipid bio-synthesis enzymes in atopic dermatitis skin is accompanied by changes in stratum corneum lipid composition. J. Dermatol. Sci. 2017;88:57–66. doi: 10.1016/j.jdermsci.2017.05.005. [DOI] [PubMed] [Google Scholar]
- 277.Imokawa G. A possible mechanism underlying the ceramide deficiency in atopic dermatitis: Expression of a deacylase enzyme that cleaves the N-acyl linkage of sphingomyelin and glucosylceramide. J. Dermatol. Sci. 2009;55:1–9. doi: 10.1016/j.jdermsci.2009.04.006. [DOI] [PubMed] [Google Scholar]
- 278.Ishibashi M., Arikawa J., Okamoto R., Kawashima M., Takagi Y., Ohguchi K., Imokawa G. Abnormal expression of the novel epidermal enzyme, glucosylceramide deacylase, and the accumulation of its enzymatic reaction product, glucosylsphingosine, in the skin of patients with atopic dermatitis. Lab. Investig. 2003;83:397–408. doi: 10.1097/01.LAB.0000059931.66821.92. [DOI] [PubMed] [Google Scholar]
- 279.Higuchi K., Hara J., Okamoto R., Kawashima M., Imokawa G. The skin of atopic dermatitis patients contains a novel enzyme, glucosylceramide sphingomyelin deacylase, which cleaves the N-acyl linkage of sphingomyelin and glucosylceramide. Biochem. J. 2000;350:747–756. doi: 10.1042/bj3500747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Kim H.J., Kim K.M., Noh M., Yoo H.J., Lee C.H. Glucosylsphingosine induces itch-scratch responses in mice. Biomol. Ther. 2010;18:316–320. doi: 10.4062/biomolther.2010.18.3.316. [DOI] [Google Scholar]
- 281.Afzal R., Shim W.S. Glucosylsphingosine activates serotonin receptor 2a and 2b: Implication of a novel itch signaling pathway. Biomol. Ther. 2017;25:497–503. doi: 10.4062/biomolther.2016.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Jin K., Higaki Y., Takagi Y., Higuchi K., Yada Y., Kawashima M., Imokawa G. Analysis of beta-glucocerebrosidase and ceramidase activities in atopic and aged dry skin. Acta Derm. Venereol. 1994;74:337–340. doi: 10.2340/0001555574337340. [DOI] [PubMed] [Google Scholar]